What are adaptogenic herbs
The term “adaptogen” was coined in the middle of the 20th century by the Russian scientist Lazarev 1 to describe medicinal plants or substances “that increase resistance to a broad spectrum of harmful factors (stressors) of different physical, chemical, and biological natures” 2. “Adaptogen” an agent that allows an organism to adapt to unfavorable stressful conditions, which could be physical, chemical, biological, or mental conditions 1. Adaptogens are considered “metabolic regulators, which increase the ability of an organism to adapt to environmental factors and to avoid damage from such factors”3. Inherent in Lazarev’s definition is the concept that administration of the adaptogenic agent allows an organism to pre-adapt itself in a manner that allows it to be more capable of responding appropriately when diverse demands are eventually placed on it. The mode of action of adaptogens is basically associated with the stress-system (neuroendocrine-immune complex) and can be directed on the various targets of the system involved in regulation (activation and inhibition) of stimulus-response coupling. Some pioneer researchers in this field put forth specific criteria that need to be fulfilled to qualify as an adaptogens, In 1969, Brekhman and Dardymov proposed specific criteria that need to be fulfilled in order for a substance to qualify as an adaptogen (see criteria below). Adaptogens could be synthetic or natural substances. However, most researches on adaptogens have focused on natural substances (specifically plants), and the term “phytoadaptogens” is now commonly used for adaptogens of plant origin 4.
It is now accepted that true adaptogens must 1:
- Possess stress-protective effects (i.e. reduction of stress-induced damage) such as anti-fatigue, anti-infection and restorative activities;
- Present stimulating effects, following both single and multiple administration, that give rise to an increase in working capacity and mental performance against a background of fatigue and stress (such stimulating effects must be different from those of conventional CNS [central nervous system] stimulants and anabolics that deplete energy and plastic resources of the organism and are accompanied by negative side effects including drug withdrawal syndrome); and
- Be innocuous and not disturb the normal level of body functions, but rather present a normalizing influence on the pathologic state, independent of the nature of that state.
Subjecting animals and humans to a period of stress produces characteristic changes in several hormones and parameters associated with the central nervous system and the hypothalamic-pituitary-adrenal axis (HPA). Hypothalamic–pituitary–adrenal (HPA) axis changes include an increase in cortisol, a reduced sensitivity of the HPA to feedback down-regulation, and a disruption in the circadian rhythm of cortisol secretion. Central nervous system changes include the stress-induced depletion of catecholamine neurotransmitters such as norepinephrine and dopamine. An acute increase in beta-endorphin levels is also observed under stressful conditions.
To successfully combat stress and stressful situations, adaptation is required. Adaptation might be best thought of as the ability to be exposed to a stressor, while responding with either decreased or no characteristic hormonal perturbations. Adaptation also implies being prepared to and capable of rapidly reassuming homeostasis after the stressor is withdrawn. As an example, a well-trained athlete can participate in an event that would induce a large HPA perturbation (stress response) in a sedentary person, and yet the athlete will be relatively unaffected. This is a result of adaptation that has occurred during the athlete’s training process. Additionally, if athletes are exposed to stressors they were not trained for, hormonal perturbations characteristic of a stress response would be expected; however, this response might not be as great as that found in less fit individuals. Furthermore, after the stress ended, their physiology would be expected to re-establish homeostasis rapidly. This is a result of non-specific resistance to stress gained by virtue of a training-induced higher level of fitness.
The utility of plant adaptogens is analogous to the training an athlete undergoes in order to prepare for competition. Plant adaptogens cause our physiology to begin the adaptation process to stress. When a stressful situation occurs, consuming adaptogens generates a degree of generalized adaptation (or non-specific resistance) that allows our physiology to handle the stressful situation in a more resourceful manner.
Plant adaptogens are compounds that increase the ability of an organism to adapt to environmental factors and to avoid damage from such factors 5. The beneficial effects of multi-dose administration of adaptogens are mainly associated with the hypothalamic–pituitary–adrenal (HPA) axis, a part of the stress-system that is believed to play a primary role in the reactions of the body to repeated stress and adaptation. In contrast, the single dose application of adaptogens is important in situations that require a rapid response to tension or to a stressful situation. In this case, the effects of the adaptogens are associated with another part of the stress-system, namely, the sympatho–adrenal-system (SAS), that provides a rapid response mechanism mainly to control the acute reaction of the organism to a stressor 5.
It should be noted that only 5:
- Schizandra chinensis,
- Eleutherococcus senticosus (Siberian ginseng) andRhodiola rosea (“Golden root” or “Arctic root”) have been found to be fully compliant with this specific definition of adaptogen.
A review 5 on the effects of single doses of adaptogens derived from Rhodiola rosea, Schizandra chinensis and Eleutherococcus senticosus that focuses primarily on the sympatho–adrenal-system (SAS) found, single administration of these adaptogens effectively increases mental performance and physical working capacity in humans. Rhodiola rosea is the most active of the three plant adaptogens producing, within 30 min of administration, a stimulating effect that continues for at least 4–6 hours. The active principles of the three plants that exhibit single dose stimulating effects are glycosides of phenylpropane- and phenylethane-based phenolic compounds such as salidroside, rosavin, syringin and triandrin, the latter being the most active.
As an example of this process, Rhodiola rosea administration promotes a moderate increase in the amount of serum immunoreactive beta-endorphin in rats under basal conditions. This moderate increase is similar to that found when rats are adapted to exercise. When Rhodiola rosea-treated rats were subjected to a 4-hour period of non-specific stress, the expected elevation in beta-endorphin was either not observed or substantially decreased. Consequently, the characteristic perturbations of the HPA were decreased or totally prevented 6. In these rats administration of Rhodiola rosea appears to have generated non-specific resistance and prepared the rats to respond more appropriately to the eventual stressful situation.
The adaptogen concept
Resistance to stress and survival depends on adaptability and the thresholds that determine an organism’s innate tolerance to a given level of stress. The stress-induced responses of the innate and adaptive defense systems involve numerous mediators of stress signaling, including the neuroendocrine–immune complex that supports allostasis in simple and complex organisms 7. Repeated mild exposure or low doses of stress result in the increased resistance of cells and organisms to subsequent stress exposure, resulting in an adaptation that favors survival. This phenomenon of adaptation to repetitive low-level stress was first described by Hans Selye in 1936 8 using rats exposed to low temperatures, low oxygen tension, muscular exercise, adrenaline, and morphine. Several nonspecific reactions were evoked (thymus atrophy, adrenal hyperplasia, stomach ulceration, increased secretion of cortisol and catecholamines, etc.), which Selye termed the general adaptation syndrome (GAS) 8. GAS necessitates three stages. The first is the initial stress recognition or “alarm reaction” when symptoms emerge. The second stage involves the acquisition of nonspecific resistance, following which symptoms disappear. Stage 3 signals exhaustion, when the same symptoms reappear, followed by death.
What happens in your body during stress
Biochemical Changes
The responses to stressors may be pleasant or unpleasant, and they vary among people and even within the same person at different times.
Your body’s homeostatic mechanisms attempt to counteract stress. When they are successful, the internal environment remains within normal physiological limits. If stress is extreme, unusual, or long lasting, the normal mechanisms may not be enough. In 1936, Hans Selye, a pioneer in stress research, showed that a variety of stressful conditions or noxious agents elicit a similar sequence of bodily changes. These changes, called the stress response or general adaptation syndrome, are controlled mainly by the hypothalamus.
The stress response occurs in three stages:
- Stage 1: an initial Fight-or-Flight Response,
- Stage 2: a Slower Resistance reaction, and eventually
- Stage 3: Exhaustion.
Research on the stress response has shown that many of the physiological alterations associated with stress are brought about by centrally controlled biochemical changes – stressors stimulate the hypothalamus to initiate the stress response through the fight-or-flight response and the resistance reaction. During situations perceived as being acutely stressful, the 2 main pathways activated are:
- The Hypthothalamus-Pituitary-Adreno Axis and
- The Sympatho-Adreno-Medullary Axis.
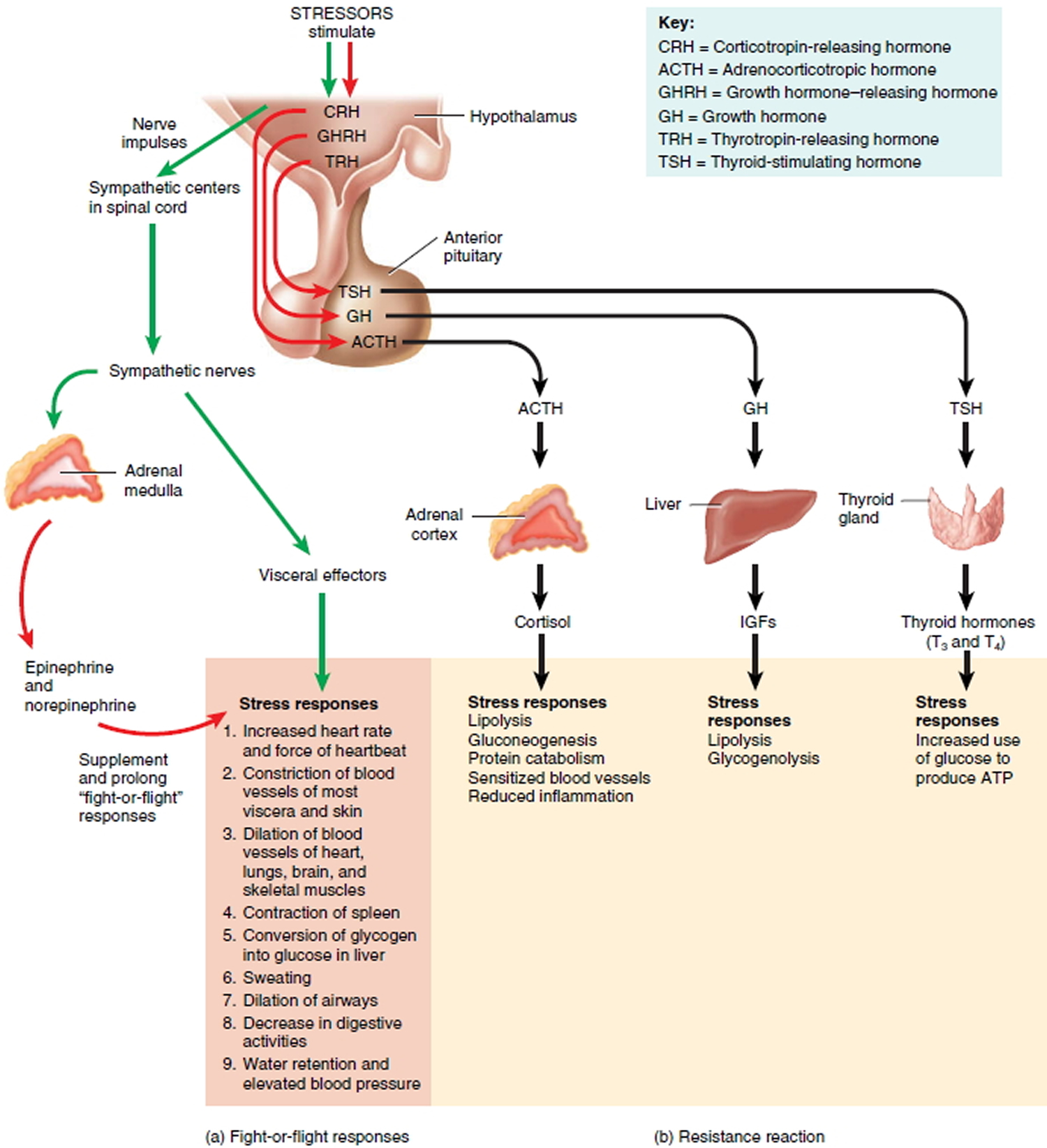
Both axes are activated by the hypothalamus secreting corticotrophin-releasing hormone (CRH), which causes the pituitary gland to release adrenocorticotropic hormone (ACTH). In the more rapidly acting of these pathways, the Sympatho-Adreno-Medullary axis, ACTH stimulates the adrenal medulla to release the catecholamines epinephrine and norepinephrine 9. These stress-induced alterations are directly linked to a number of the physiological changes that take place in the body including increases in blood pressure, heart rate, respiration, and oxygen consumption. In the slower-acting HPA axis, blood-borne ACTH acts on the adrenal cortex to release cortisol. Once in the bloodstream, cortisol induces metabolic changes in the liver, resulting in increased glucose concentrations in blood and tissues. The increased glucose produces adenosine triphosphate (ATP) to repair damaged cells and enables metabolically active cells throughout the body to respond to the stressor 9.
Figure 1. Hypthothalamus-Pituitary-Adreno Axis and Sympatho-Adreno-Medullary Axis
Note: Responses to stressors during the stress response. Red arrows (hormonal responses) and green arrows (neural responses) in (a) indicate immediate fight-or-flight reactions; black arrows in (b) indicate long-term resistance reactions.
The Fight-or-Flight Response (Stage 1 of Stress)
The fight-or-flight response, initiated by nerve impulses from the hypothalamus to the sympathetic division of the autonomic nervous system (ANS), including the adrenal medulla, quickly mobilizes the body’s resources for immediate physical activity (see Figure 1 above). It brings huge amounts of glucose and oxygen to the organs that are most active in warding off danger: the brain, which must become highly alert; the skeletal muscles, which may have to fight off an attacker or flee; and the heart, which must work vigorously to pump enough blood to the brain and muscles. During the fight-or-flight response, nonessential body functions such as digestive, urinary, and reproductive activities are inhibited. Reduction of blood flow to the kidneys promotes release of renin, which sets into motion the renin–angiotensin–aldosterone pathway. Aldosterone causes the kidneys to retain Na+, which leads to water retention and elevated blood pressure. Water retention also helps preserve body fluid volume in the case of severe bleeding.
The Resistance Reaction (Stage 2 of Stress)
The second stage in the stress response is the resistance reaction (Figure 1b). Unlike the short-lived fight-or-flight response, which is initiated by nerve impulses from the hypothalamus, the resistance reaction is initiated in large part by hypothalamic releasing hormones and is a longer-lasting response. The hormones involved are corticotropin-releasing hormone (CRH), growth hormone–releasing hormone (GHRH), and thyrotropin-releasing hormone (TRH).
Corticotropin-releasing hormone (CRH) stimulates the anterior pituitary to secrete adrenocorticotropic hormone (ACTH), which in turn stimulates the adrenal cortex to increase release of cortisol. Cortisol then stimulates gluconeogenesis by liver cells, breakdown of triglycerides into fatty acids (lipolysis), and catabolism of proteins into amino acids. Tissues throughout the body can use the resulting glucose, fatty acids, and amino acids to produce ATP (adenosine triphosphate is the energy packets necessary for cells to function) or to repair damaged cells. Cortisol also reduces inflammation.
A second hypothalamic releasing hormone, growth hormone–releasing hormone (GHRH), causes the anterior pituitary to secrete growth hormone (GH). Acting via insulin like growth factors (ILGF), GH stimulates lipolysis and glycogenolysis, the breakdown of glycogen to glucose, in the liver. A third hypothalamic releasing hormone, thyrotropin-releasing hormone (TRH), stimulates the anterior pituitary to secrete thyroid-stimulating hormone (TSH). TSH promotes secretion of thyroid hormones, which stimulate the increased use of glucose for ATP production. The combined actions of GH and TSH supply additional ATP for metabolically active cells throughout the body.
The resistance stage helps the body continue fighting a stressor long after the fight-or-flight response dissipates. This is why your heart continues to pound for several minutes even after the stressor is removed. Generally, it is successful in seeing you through a stressful episode, and your bodies then return to normal. Occasionally, however, the resistance stage fails to combat the stressor, and the body moves into the state of exhaustion.
Stage of Exhaustion (Stage 3 of Stress)
The resources of the body may eventually become so depleted that they cannot sustain the resistance stage, and exhaustion ensues. Prolonged exposure to high levels of cortisol and other hormones involved in the resistance reaction causes wasting of muscle, suppression of the immune system, ulceration of the gastrointestinal tract, and failure of pancreatic beta cells. In addition, pathological changes may occur because resistance reactions persist after the stressor has been removed.
This puts you at increased risk of numerous health problems, including:
- Anxiety
- Depression
- Digestive problems
- Headaches
- Heart disease
- Sleep problems
- Weight gain
- Memory and concentration impairment
That’s why it’s so important to learn healthy ways to cope with the stressors in your life.
Figure 2. Comparative Impact of the Acute Stress and Relaxation Responses
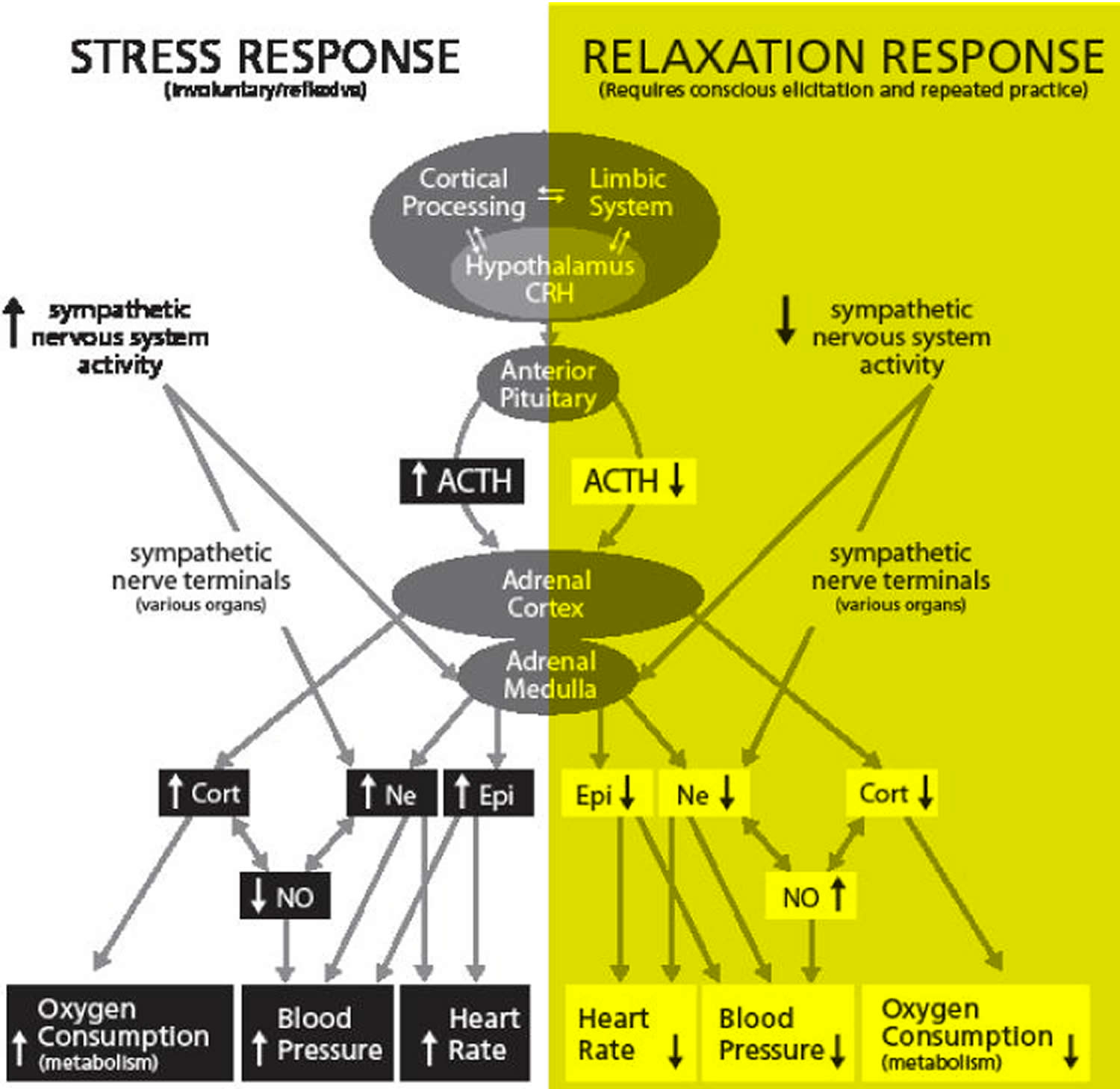
Note:
Central and Peripheral Nervous System Activities
Using blood pressure as an example, we show how acute stress and relaxation responses alter hypothalamus-pituitary-adrenal (HPA) and sympatho-adreno-medullary (SAM) axis activities. These responses introduce contrasting hormonal and signal molecule changes that in turn influence clinically significant conditions such as high blood pressure. Epi = epinephrine or adrenaline / Ne = norepinephrine or noradrenaline / SAM axis is the adrenal medulla to Ne and Epi / Cort = cortisol / NO = nitric oxide / HPA axis is the adrenal cortex to Cort (cortisol)
Why you react to life stressors the way you do ?
Your reaction to a potentially stressful event is different from anyone else’s. How you react to stressors in your life is affected by such factors as:
- Genetics. The genes that control the stress response keep most people on a fairly even keel, only occasionally priming the body for fight or flight. Overactive or underactive stress responses may stem from slight differences in these genes.
- Life experiences. Strong stress reactions sometimes can be traced to traumatic events. People who were neglected or abused as children tend to be particularly vulnerable to stress. The same is true of people who have experienced violent crime, airplane crash survivors, military personnel, police officers and firefighters.
You may have some friends who seem laid-back about almost everything and others who react strongly at the slightest stress. Most reactions to life stressors fall somewhere between those extremes.
The adaptive stress response
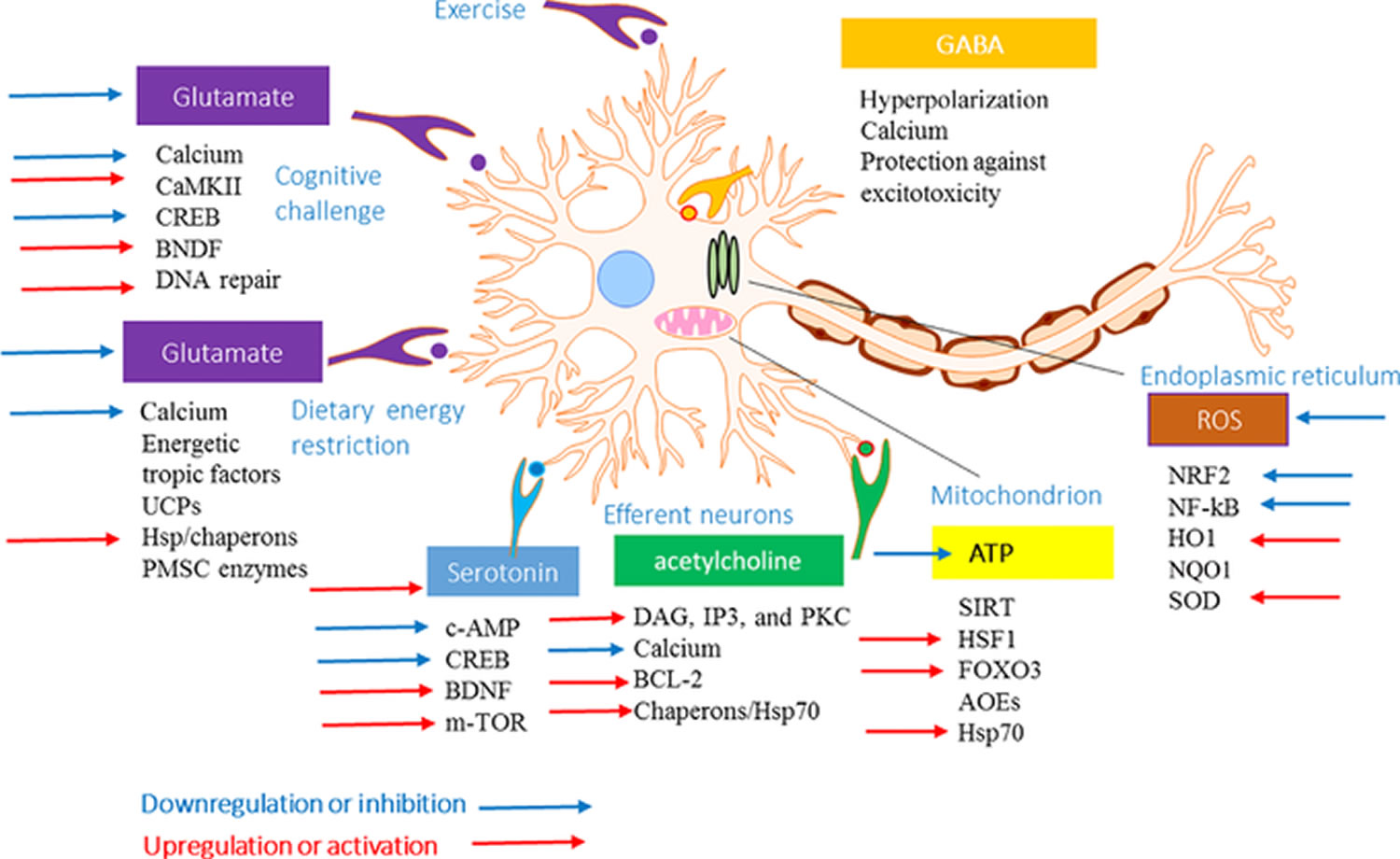
The adaptive stress response occurs in a variety of regulatory systems from the cellular level to the whole organism. At the cellular and molecular levels, intra- and extracellular signaling pathways promote upregulation of antiapoptotic proteins, neuropeptides, and antioxidant enzymes in the alarm phase 10. Figure 3 outlines seven adaptive stress response signaling pathways that protect neurons against degeneration and promote synaptic plasticity and depicts how adaptogens influence signaling to promote neuroplasticity and decrease vulnerability to neurodegeneration. In this context, botanical adaptogens are metabolic regulators that increase survival by increasing adaptability in stress.
A characteristic feature of adaptogens is that they act as eustressors (i.e., “good stressors”) and as mild stress mimetics or “stress vaccines” that induce stress-protective responses 11. Mild (survivable) stress induces a resistance or “immunity” to subsequent, more severe stress exposure 11. However, this stress-induced resistance carries no memory function, and repeated exposure to the adaptogen is required to maintain the plastic adaptive state. Another comparison could be made with repetitive physical exercise, which increases endurance and performance 12. A state of nonspecific resistance (SNSR) could be achieved either by the gradual “training” of an organism to withstand the effects of the stress or by adaptogens that mimic the stress. The repeated administration of adaptogens and the consequent adaptogenic or stress-protective response arise in a manner analogous to repeated physical exercise that leads to prolonged SNSR and increased endurance and stamina 13. The phenomenon of adaptation to stress also underpins the hormetic response, which is defined as an adaptive response characterized by a biphasic dose–response, with a low dose that is stimulatory (i.e., has a beneficial effect) and a high dose that is inhibitory (i.e., has a toxic effect) 14.
Figure 3. Adaptive stress response and effects of adaptogens
Note: Adaptive cellular stress response signaling that mediates beneficial effects of environmental challenges and adaptogens on neuroplasticity and vulnerability to neurodegeneration. A typical glutamatergic neuron in the hippocampus is depicted receiving excitatory inputs (red) from neurons activated in response to exercise, cognitive challenges, and dietary energy restriction. Examples of seven different adaptive stress response signaling pathways that protect neurons against degeneration and promote synaptic plasticity are shown. During exercise and cognitive challenges, postsynaptic receptors for glutamate, serotonin, and acetylcholine are activated to engage intracellular signaling cascades and transcription factors that induce the expression of neuroprotective proteins, including brain-derived neurotrophic factor (BDNF), mitochondrial uncoupling proteins (UCPs), and antiapoptotic proteins (e.g., BCL-2). BDNF promotes neuronal growth, in part, by activating the mammalian target of rapamycin (mTOR). Mild cellular stress resulting from reduced energy substrates and reactive oxygen species (ROS) engages adaptive stress response pathways, including those that upregulate antioxidant enzymes (AOEs) and protein chaperones. Release of GABA from interneurons in response to activity in excitatory circuits (as occurs during exercise and cognitive challenges) hyperpolarizes excitatory neurons, protecting them from Ca2+ overload and excitotoxicity. CaMKII, calcium/calmodulin kinase II; CREB, cyclic AMP response element–binding protein; DAG, diacylglycerol; FOXO3, forkhead box protein O3; HO1, heme oxygenase 1; HSF1, heat shock factor 1; IP3 PKC, inositol-trisphosphate 3 protein kinase C; NF-κB, nuclear factor κB; NQO1, NAD(P)H-quinone oxidoreductase 1; NRF2, nuclear regulatory factor 2.
[Source 15]Pharmacology and the mechanism of action of adaptogens
The pharmacologic efficacy of adaptogens and their stress-protective effects are usually investigated by testing cognitive function and physical endurance under stressful conditions 16. Further, the use of valid and specific biomarkers related to pharmacological activity is a generally accepted practice in pharmacology.
Which effectors are responsible for mediating adaptogenic effects, and what are their key molecular targets ?
A number of human and animal studies have suggested that the stress hormones cortisol and neuropeptide Y (NPY) and several important mediators of the adaptive stress response (e.g., nitric oxide, stress-activated protein kinases, heat shock proteins (HSP70 and HSP25), and the FOXO (DAF-16) transcription factor) are key players in mediating the adaptogenic effects of plant extracts (e.g., Rhodiola, Eleutherococcus, Schisandra, ginseng, Bryonia, Withania, etc.) 17. These mediators orchestrate the process of stress adaptation (including aging or disease pathology), with no single contribution that can be estimated with any degree of certainty. Figures 4 show the hypothetical mechanisms of action of adaptogens in stress-induced fatigue, depression, and aging.
Several reviews describe the possible mechanisms of action of adaptogens on the basis of the results of in vitro and ex vivo experiments using cells of both human and animal origin. HSP70 and heat shock factor-1 (HSF1) are considered to be pharmacological targets of antiaging therapies 18. However, chemicals used to induce HSP70 are typically cytotoxic and therefore cannot be used by the target patient population (e.g., elderly individuals) who are more susceptible to stress. Fortunately, plant adaptogens have been used safely over a very wide dose range (up to 3000 mg/kg of rat body weight), even with repeated administration (over several months). The adaptogens Rhodiola, Schisandra, Eleutherococcus, and their combination as ADAPT-232 (with its active constituent salidroside) stimulate the expression of HSF-1 and heat shock protein 70 (HSP72) in isolated neuroglia, provoke HSP72 release from cells 19 and promote the increased expression of HSP70 in vivo 20. Chronic Rhodiola rosea use significantly ameliorated swimming-induced fatigue by promoting glycogen levels, increasing energy generated by lipogenic enzymes, and boosting defense mechanisms inclusive of HSP70 action 21. R. rosea root extract significantly upregulates HSP70 mRNA and protects skeletal muscle cells against chemically induced oxidation 22. Further, Schizandrin B pretreatment induces a time-dependent increase in HSP25 and HSP70 expression in rat heart and protects against myocardial ischemia–reperfusion injury 23. The hepatic cytoprotective action of schizandrin B against acetaminophen-induced liver injury is also mediated, at least in part, by the induction of HSP27 and HSP70 in mice. Oral administration of schizandrin B increased HSP27 and HSP70 gene and protein expression in a time- and dose-dependent fashion 24.
Salidroside and extracts of Schisandra chinensis and R. rosea were found to be the most active inhibitors of stress-induced p-SAPK/p-JNK. It has been shown that oral supplements of rhodioloside, or extracts of Eleutherococcus senticosus, S. chinensis, or R. rosea, administered over a 7-day period to rabbits subject to restraint stress, significantly decreased their levels of stress-activated protein kinases (i.e., the phosphorylated forms of SAPK/JNK) in circulating blood 25.
Exercise can induce expression of HSP70, which acts as an antiaging agent. This upregulation contributes to the maintenance of muscle fiber integrity and facilitates muscle regeneration and recovery. On the other hand, HSP70 expression is reduced during muscle inactivity and aging. Dysfunction of HSP70 generation may drive muscle atrophy, contractile dysfunction, and reduce regenerative capacity (associated with aging). The beneficial effects of activating the biosynthesis of HSP70 in skeletal muscle have been established in animal studies, suggesting that HSP70 is a key therapeutic target for the treatment of various conditions that negatively affect skeletal muscle mass and function 26.
Figure 4. Hypothetical mechanism of action of adaptogens on the stress system in depression
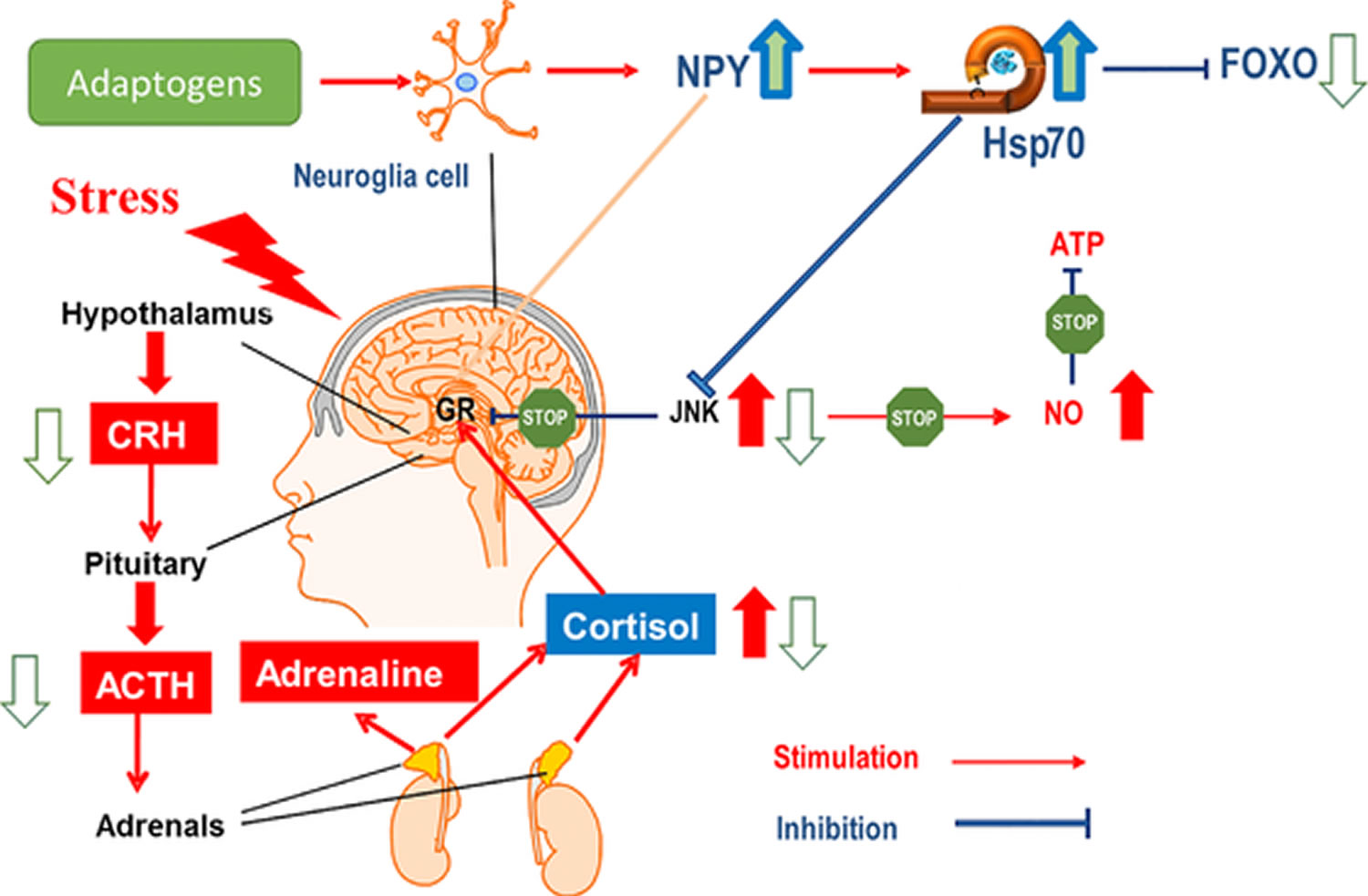 Note: Stress-induced release of CRH from the hypothalamus, followed by release of ACTH from the pituitary, simulates release of adrenal hormones and NPY in order to cope with the stress. Feedback regulation of overreaction is initiated by cortisol release from the adrenal cortex, followed by binding to glucocorticoid receptors (GR) in the brain. This signal stops the further release of brain hormones, and the stress-induced increase in cortisol decreases to normal levels in the circulatory system. While short and mild stress (eustress) is essential to life, severe stress can cause disease depression, which is associated with generation of active oxygen-containing molecules, including nitric oxide, which is known to inhibit ATP formation. Stress-induced signaling protein JNK was found to inhibit GR; consequently, this feedback normalization is blocked and cortisol content in blood of depressive patients is permanently high. This is associated with impaired memory, impaired ability to concentrate, fatigue, and other symptoms. Adaptogens suppress elevated JNK and cortisol in stress and stimulate the formation of HSP70, which is known to inhibit JNK. Consequently, nitric oxide levels no longer increase and ATP generation is not suppressed.
Note: Stress-induced release of CRH from the hypothalamus, followed by release of ACTH from the pituitary, simulates release of adrenal hormones and NPY in order to cope with the stress. Feedback regulation of overreaction is initiated by cortisol release from the adrenal cortex, followed by binding to glucocorticoid receptors (GR) in the brain. This signal stops the further release of brain hormones, and the stress-induced increase in cortisol decreases to normal levels in the circulatory system. While short and mild stress (eustress) is essential to life, severe stress can cause disease depression, which is associated with generation of active oxygen-containing molecules, including nitric oxide, which is known to inhibit ATP formation. Stress-induced signaling protein JNK was found to inhibit GR; consequently, this feedback normalization is blocked and cortisol content in blood of depressive patients is permanently high. This is associated with impaired memory, impaired ability to concentrate, fatigue, and other symptoms. Adaptogens suppress elevated JNK and cortisol in stress and stimulate the formation of HSP70, which is known to inhibit JNK. Consequently, nitric oxide levels no longer increase and ATP generation is not suppressed.What are the chemical structures of the principal active substances in adaptogenic plant extracts and their structure–function relationships ?
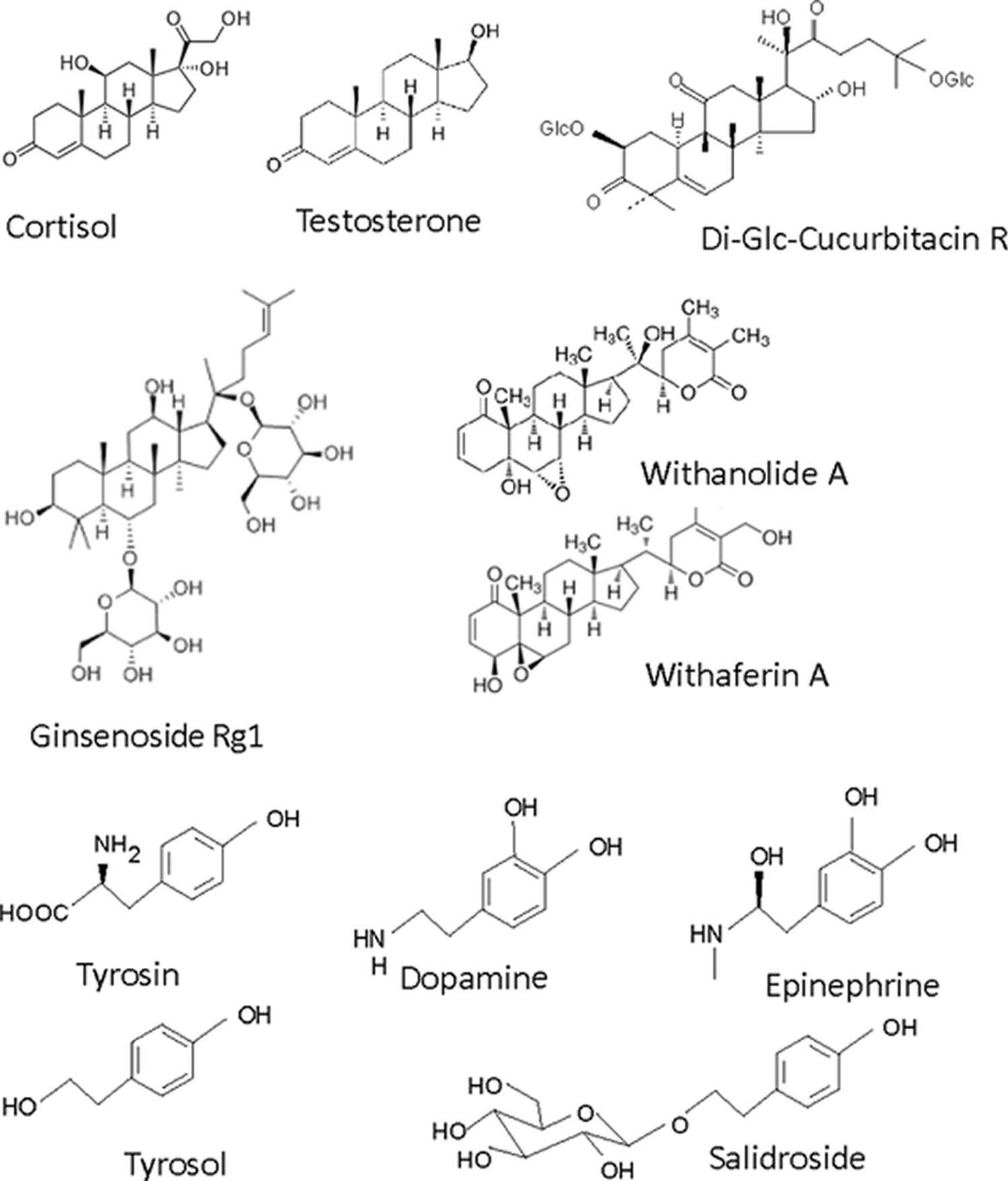
Currently, no systematic studies on the structure–function activities of purified adaptogens with their targets are available. However, the principal active ingredients of plant adaptogens (as investigated thus far) can be divided into two main chemical groups (Figure 5): (1) terpenoids, with a tetracyclic skeleton, such as cortisol and testosterone (ginsenosides, sitoindosides, cucurbitacines, and withanolides) and (2) aromatic compounds that are structurally similar to catecholamines or tyrosine, including lignans (eleutheroside E (E. senticosus) and schizandrin B (S. chinensis)), phenylpropane derivatives (syringin (E. senticosus), rosavin (R. rosea)), and phenylethane derivatives (salidroside (R. rosea)). A number of studies indicate direct interactions between ginsenosides and corticosteroid and estrogenic receptors.
Plants containing mainly tetracylic or pentacyclic terpenoids (ginseng, Withania, Rhaponticum, Bryonia, etc.) are presumed to act via the hypothalamic–pituitary–adrenal (HPA) axis, while plants (e.g., Rhodiola, Schisandra, etc.) containing predominantly phenolic compounds (phenylpropanoids, phenylethanoids, and their dimers [lignans]) are presumed to interact with elements of the efferent sympathoadrenal system (SAS).
Figure 5. Chemical structures of adaptogenic compounds of plant origin
What physiological functions and diseases can adaptogens influence ?
As our knowledge of cell and systems biology grows, new questions and challenges arise. One of these relates to specific indications for drug use in medicine. The one drug–one indication paradigm is ordinarily unsuitable for adaptogens, given their polyvalent mode of action and nonspecific effects on the immune, endocrine, and nervous systems.
Adaptogens normalize chronically increased cortisol/corticosterone in the blood and saliva of humans or animals 25 presumably owing to a direct interaction with glucocorticoid receptors. For example, it was shown that the ginsenoside Rg1 is a functional ligand of glucocorticoid receptors, and its direct interaction with glucocorticoid receptors ligand-binding sites has been demonstrated. Rg1 behaves as a partial agonist of glucocorticoid receptors (not an inhibitor). Interestingly, the ginsenoside Rb1 is a functional ligand of the ER, in particular, the β isoform 27 and may also have beneficial effects in the conditions described above.
All other mediators of the effects of adaptogens (e.g., nitric oxide, JNK, SAPK, HSP70, HSP25, and FOXO [DAF16]) play roles in chronic inflammation (common to all age-related diseases), such as that seen in muscle degeneration (sarcopenia), senile dementia, Alzheimer’s disease, atherosclerosis, cardiovascular disease, hypertension, osteoarthritis, type 2 diabetes, and obesity. Clearly, more randomized clinical trials of standardized botanicals are required if we are to implement these agents in medical practice for use in these specific indications.
Adaptogenic herbs
Current and potential uses of adaptogens in pharmacotherap are related to their treatment of mental diseases and behavioral disorders, stress-induced fatigue, and cognitive function 28, 29. However further studies are warranted if we are to understand the range of interactions between adaptogens and stress response pathways (both intracellular and extracellular) in terms of the metabolic regulation of homeostasis in stress- and age-associated disease.
Some adaptogenic plants (see Table 1) have been used in traditional Chinese medicine and Ayurveda for centuries to promote physical and mental health, improve the body’s defense mechanisms, and enhance longevity 15. However, further evidence, based on well-designed clinical trials with standardized herbal preparations, is required to support the efficacy of these traditional herbal medicines to qualify them as herbal medicinal products with well-established use in medicine. Moreover, the investigations of molecular mechanisms of action of adaptogens are required for understanding the polyvalent pharmacological activity of adaptogens. The reductionist concept of a single receptor-based view of drug action 30 would appear to be unsatisfactory for adaptogens. However, adaptogens exhibit multitarget action and the shared use of a number of different receptors, including receptors for corticosteroid, mineralocorticoid, progestin, estrogen, serotonin (5-HT), N-methyl-d-aspartate, and nicotinic acetylcholine, receptor tyrosine kinases, and many G protein–coupled receptors 31. Therefore, the possibility that numerous molecular network interactions (with feedback regulation of the neuroendocrine and immune systems) contribute to the overall pharmacological response and result in agonist-dependent antagonism is most suitable for understanding the mechanisms of action of adaptogens. Thus, the pharmacology of adaptogens is a typical example of network pharmacology 32. Network pharmacology has the potential to provide treatments for complex diseases, chronic conditions, and syndromes, inclusive of their pathophysiologic evolution, where conventional approaches have often been disappointing 33. Adaptive stress responses include several stages and involve multiple molecular networks in which receptors interact with adaptogens 32.
Table 1. Plants cited in the literature with adaptogenic properties
| Ajuga turkestanica (Regel) Briq. | Emblica officinalis Gaetrn. | Piper longum L. |
| Alstonia scholaris (L.) R. Br. | Eucommia ulmoides Oliv. | Potentilla alba L. |
| Anacyclus pyrethrum (L.) Lag. | Evolvulus alsinoides (L.) L. | Ptychopetalum olacoides Benth. |
| Andrographis paniculata (Burm.f.) Nees | Firmiana simplex (L.) W.Wight | Rhaponticum carthamoides (Willd.) Iljin |
| Aralia mandshurica Rupr. & Maxim | Gentiana pedicellata (D.Don) Wall | Rhodiola heterodonta (Hook. f. & Thomson) Boriss. |
| Argyreia nervosa (Burm. f.) Bojer | Glycyrrhiza glabra L. | Rhodiola rosea L. |
| Argyreia speciosa (L. f.) Sweet | Heteropterys aphrodisiaca Machado | Rostellularia diffusa (Willd.) Nees. |
| Asparagus racemosus Wild | Hippophae rhamnoides L. | Salvia miltiorrhiza Bunge |
| Bacopa monnieri (L.) Wettst | Holoptelea integrifolia Planch | Schisandra chinensis (Turcz.) Baill. |
| Bergenia crassifolia (L.) Fritsch | Hoppea dichotoma Willd. | Scutellaria baicalensis Georgi |
| Bryonia alba L. | Hypericum perforatum L. | Serratula inermis Poir |
| Caesalpinia bonduc (L.) Roxb | Lepidium peruvianum/Lepidium meyenii Walp. | Sida cordifolia L. |
| Centella asiatica (L.) Urb. | Ligusticum striatum DC. | Silene italica (L.) Pers. |
| Chlorophytum borivilianum Santapau & R.R.Fern. | Melilotus officinalis (L.) Pall. | Sinomenium acutum (Thunb.) Rehder & E.H.Wilson |
| Chrysactinia mexicana A. Gray | Morus alba L. | Solanum torvum SW. |
| Cicer arietinum L. | Mucuna pruriens (L.) DC. | Sutherlandia frutescens (L.) R.Br. |
| Codonopsis pilosula (Franch.) Nannf. | Nelumbo nucifera Gaertn. | Terminalia chebula Retz. |
| Convolvulus prostratus Forssk. | Ocimum sanctum L. | Tinospora cordifolia (Willd.) Miers |
| Curculigo orchioides Gaertn. | Oplopanax elatus (Nakai) Nakai | Trichilia catigua A.Juss. |
| Curcuma longa L. Curcumin | Panax ginseng C.A.Mey. | Trichopus zeylanicus Gaertn. |
| Dioscorea deltoidea Wall. ex Griseb. | Panax pseudoginseng Wall. | Turnera diffusa Willd. ex Schult. |
| Drypetes roxburghii (Wall.) Hurus. | Pandanus odoratissimus L.f. | Vitis vinifera L. |
| Echinopanax elatus Nakai | Paullinia cupana Kunth | Withania somnifera (L.) Dunal |
| Eleutherococcus senticosus (Rupr. & Maxim.) Maxim. | Pfaffia paniculata (Mart.) Kuntze |
Note: This table is an update from the reviews of Wagner et al. 34 and Panossian and Wagner 35. It includes plants that do and do not meet the formal definition of adaptogen.
[Source 15]The active components of stress-protective plants and adaptogens (see Table 2) can be formally divided into three main groups, namely, tetra(penta)cyclic terpenoids, phenyl- and phenylethyl-propanoids and derivatives, and oxylipins. On the basis of the chemical nature of their active principles, some indication of the possible mechanism of action of these plants may be derived. Thus, the extracts of Panax ginseng, Withania somnifera, Bryonia alba and Aralia mandshurica contain phytosterols, and tetracyclic and pentacyclic triterpenes, that likely exert their effect on the hypothalamus-pituitary-adrenal (HPA) axis in which cortisone plays a key role during stress. Typically these adaptogens prevent or at least decrease certain hormonal changes, such as the increased level of cortisone, that are characteristic of a stress reaction 36. Plants such as Rhodiola rosea and Schizandra chinensis accumulate phenolic secondary metabolites such as phenyl- and phenylethyl-propanoids and their dimeric lignans 37. Such compounds can play an active role in stress response in respect of achieving a state of maximum work capacity as would be required in fight-or-flight situations 38. Interestingly, Eleutherococcus senticosus contains both types of biologically active, low molecular weight compound and exhibits a very wide range of pharmacological effects 39.
Table 2. Adaptogenic herbs
| Group I: Stress-protectors | Group II: Adaptogens |
|---|---|
| Panax ginseng | Rhodiola rosea |
| Tetracyclic triterpenes and their glycosides | Phenylethyl glycosides |
| Pentacyclic triterpenes and their glycosides | Phenylpropanoids |
| Polyacetylenes | Flavonoids |
| Bryonia alba | Phenolics |
| Tetracyclic triterpenes and their glycosides | Polyphenolics |
| Pentacyclic triterpenes | Lignans |
| Sterols and their glycosides | Flavolignans |
| Oxylipins and glycolipids | |
| Withania somnifera | Schizandra chinensis |
| Tetracyclic triterpene lactones and their glycosides | Dibenzo[a,c]cyclooctadiene |
| Sterols and their glycosides | Sterols |
| Alkaloids | Organic and fatty acids, |
| Aralia mandshurica | Vitamins A,C and E |
| Pentacyclic triterpenes and their glycosides | Sterols |
| Eleutherococcus senticosus | |
| Pentacyclic and tetracyclic triterpene glycosides | Phenylpropanoids |
| Sterols | Lignans |
| Polysaccharides (heteroglycans, eleutherans) | Coumarins |
- New substances of plant origin which increase nonspecific resistance. Brekhman II, Dardymov IV. Annu Rev Pharmacol. 1969; 9():419-30. https://www.ncbi.nlm.nih.gov/pubmed/4892434/
- Brekhman, I.I. & I.V. Dardymov. 1968. New substances of plant origin which increase nonspecific resistance. Annu. Rev. Pharmacol. 8: 419–430.
- Panossian, A., G. Wikman & H. Wagner. 1999. Plant adaptogens. III. Earlier and more recent aspects and concepts on their mode of action. Phytomedicine 6: 287–300.
- Esimone C. O., Adikwu M. U., Nworu C., Okoye S. C., Odimegwu D. C. Adaptogenic potentials of Camellia sinensis leaves, Garcinia kola and Kola nitida seeds. Scientific Research and Essays. 2007;2(7):232–237.
- Stimulating effect of adaptogens: an overview with particular reference to their efficacy following single dose administration. Panossian A, Wagner H. Phytother Res. 2005 Oct; 19(10):819-38. https://www.ncbi.nlm.nih.gov/pubmed/16261511/
- Lishmanov IB, Trifonova ZV, Tsibin AN, et al. Plasma beta-endorphin and stress hormones in stress and adaptation. Biull Eksp Biol Med 1987;103:422-424. [Article in Russian]
- McEwen, B.S. 2000. Allostasis and allostatic load: implications for neuropsychopharmacology. Neuropsychopharmacology 22: 108–124.
- Selye, H. 1976. Forty years of stress research: principal remaining problems and misconceptions. Can. Med. Assoc. J. 115: 53–56.
- The concepts of stress and stress system disorders. Overview of physical and behavioral homeostasis. Chrousos GP, Gold PW. JAMA. 1992 Mar 4; 267(9):1244-52. https://www.ncbi.nlm.nih.gov/pubmed/1538563/
- Stranahan, A.M. & M.P. Mattson. 2012. Recruiting adaptive cellular stress responses for successful brain ageing. Nat. Rev. Neurosci. 13: 209–216.
- Wiegant, F.A.C., G. Limandjaja, S.A.H. de Poot, et al. 2008. Plant adaptogens activate cellular adaptive mechanisms by causing mild damage. In Adaptation Biology and Medicine: Health Potentials. Vol. 5. L. Lukyanova, N. Takeda & P.K. Singal, Eds.: 319–332. New Delhi: Narosa Publishers.
- Viru, A.A. 1981. Hormonal Mechanisms of Adaptation and Training. Leningrad: Nauka, 1–154.
- Hovhannisyan, A.S., M. Nylander, A.G. Panossian, et al. 2015. Efficacy of adaptogenic supplements on adapting to stress: a randomized, controlled trial. J. Athl. Enhancement 4: 4.
- Mattson, M.P. 2008. Hormesis defined. Ageing Res. Rev. 7: 1–7.
- Panossian, A. (2017), Understanding adaptogenic activity: specificity of the pharmacological action of adaptogens and other phytochemicals. Ann. N.Y. Acad. Sci., 1401: 49–64. doi:10.1111/nyas.13399 http://onlinelibrary.wiley.com/doi/10.1111/nyas.13399/full
- Panossian, A. & H. Wagner. 2005. Stimulating effect of adaptogens: an overview with particular reference to their efficacy following single dose administration. Phytother. Res. 19: 819–838.
- Rattan, S.I., V. Kryzch, S. Schnebert, et al. 2013. Hormesis-based anti-aging products: a case study of a novel cosmetic. Dose Response 11: 99–108.
- Perez, F.P., S.S. Moinuddin, S. Ulain, et al. 2012. Longevity pathways: HSF1 and FoxO pathways, a new therapeutic target to prevent age-related diseases. Curr. Aging Sci. 5: 87–95.
- Asea, A., P. Kaur, A. Panossian, et al. 2013. Evaluation of molecular chaperons Hsp72 and neuropeptide Y as characteristic markers of adaptogenic activity of plant extracts. Phytomedicine 20: 1323–1329.
- Panossian, A., G. Wikman, P. Kaur, et al. 2009. Adaptogens exert a stress protective effect by modulation of expression of molecular chaperons. Phytomedicine 16: 617–622.
- Lee, F.T., T.Y. Kuo, S.Y. Liou, et al. 2009. Chronic Rhodiola rosea extract supplementation enforces exhaustive swimming tolerance. Am. J. Chin. Med. 37: 557–572.
- Hernández-Santana, A., V. Pérez-López, J.M. Zubeldia, et al. A Rhodiola rosea root extract protects skeletal muscle cells against chemically induced oxidative stress by modulating heat shock protein 70 (HSP70) expression. Phytother. Res. 28: 623–628.
- Chiu, P.Y. & K.M. Ko. 2004. Schisandrin B protects myocardial ischemia–reperfusion injury partly by inducing Hsp25 and Hsp70 expression in rats. Mol. Cell. Biochem. 266: 139–144.
- Li, L., T. Zhang, L. Zhou, et al. 2014. Schisandrin B attenuates acetaminophen-induced hepatic injury through heat-shock protein 27 and 70 in mice. J. Gastroenterol. Hepatol. 29: 640–647.
- Panossian, A., M. Hambardzumyan, A. Hovhanissyan, et al. 2007. The adaptogens rhodiola and schizandra modify the response to immobilization stress in rabbits by suppressing the increase of phosphorylated stress-activated protein kinase, nitric oxide and cortisol. Drug Target Insights 2: 39–54.
- Senf, S.M. 2013. Skeletal muscle heat shock protein 70: diverse functions and therapeutic potential for wasting disorders. Front. Physiol. 4: 330.
- Leung, K.W. & A.S. Wong. 2010. Pharmacology of ginsenosides: a literature review. Chin. Med. 5: 20.
- Amsterdam, J.D. & A.G. Panossian. 2016. Rhodiola rosea L. as a putative botanical antidepressant. Phytomedicine 23: 770–783. https://www.ncbi.nlm.nih.gov/pubmed/27013349
- Panossian, A. & G. Wikman. 2009. Evidence-based efficacy of adaptogens in fatigue, and molecular mechanisms related to their stress-protective activity. Curr. Clin. Pharmacol. 4: 198–219.
- Kenakin, T.P. 2012. Pharmacology in Drug Discovery and Development: Understanding Drug Response. Elsevier Inc.
- Panossian, A., R. Hamm, G. Wikman, et al. 2013. Synergy and antagonism of active constituents of ADAPT-232 on transcriptional level of metabolic regulation in isolated neuroglia cells. Front. Neurosci. 7: 16.
- Panossian, A., R. Hamm, O. Kadioglu, et al. 2014. Mechanism of action of Rhodiola, salidroside, tyrosol and triandrin in isolated neuroglial cells: an interactive pathway analysis of the downstream effects using RNA microarray data. Phytomedicine 21: 1325–1348.
- Poornima, P., J.D. Kumar, Q. Zhao, et al. 2016. Network pharmacology of cancer: from understanding of complex interactomes to the design of multi-target specific therapeutics from nature. Pharmacol. Res. 2111: 290–302.
- Wagner, H., H. Nörr & H. Winterhoff. 1994. Plant adaptogens. Phytomedicine 1: 63–76. https://www.ncbi.nlm.nih.gov/pubmed/23195818
- Panossian, A. & H. Wagner. 2011. Adaptogens. A review of their history, biological activity, and clinical benefits. HerbalGram 90: 52–63.
- Effects of ginseng saponin administered intraperitoneally on the hypothalamo-pituitary-adrenal axis in mice. Kim DH, Moon YS, Jung JS, Min SK, Son BK, Suh HW, Song DK. Neurosci Lett. 2003 May 29; 343(1):62-6. https://www.ncbi.nlm.nih.gov/pubmed/12749998/
- Saratikov AS, Krasnov EA. Rhodiola rosea (Golden root): a valuable medicinal plant. Tomsk: Tomsk University Press; 2004. pp. 1–205.
- Lüllmann H, Mohr K, Hein L, et al. Color atlas of pharmacology. 3rd ed. Stuttgart: Georg Thieme Verlag; 2005.
- World Health Organisation. WHO Monographs on Selected Medicinal Plants. Vol. 2. Geneva: WHO; 2002.
- Panossian A, Hambardzumyan M, Hovhanissyan A, Wikman G. The Adaptogens Rhodiola and Schizandra Modify the Response to Immobilization Stress in Rabbits by Suppressing the Increase of Phosphorylated Stress-activated Protein Kinase, Nitric Oxide and Cortisol. Drug Target Insights. 2007;2:39-54. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3155223/