What is alpha lipoic acid
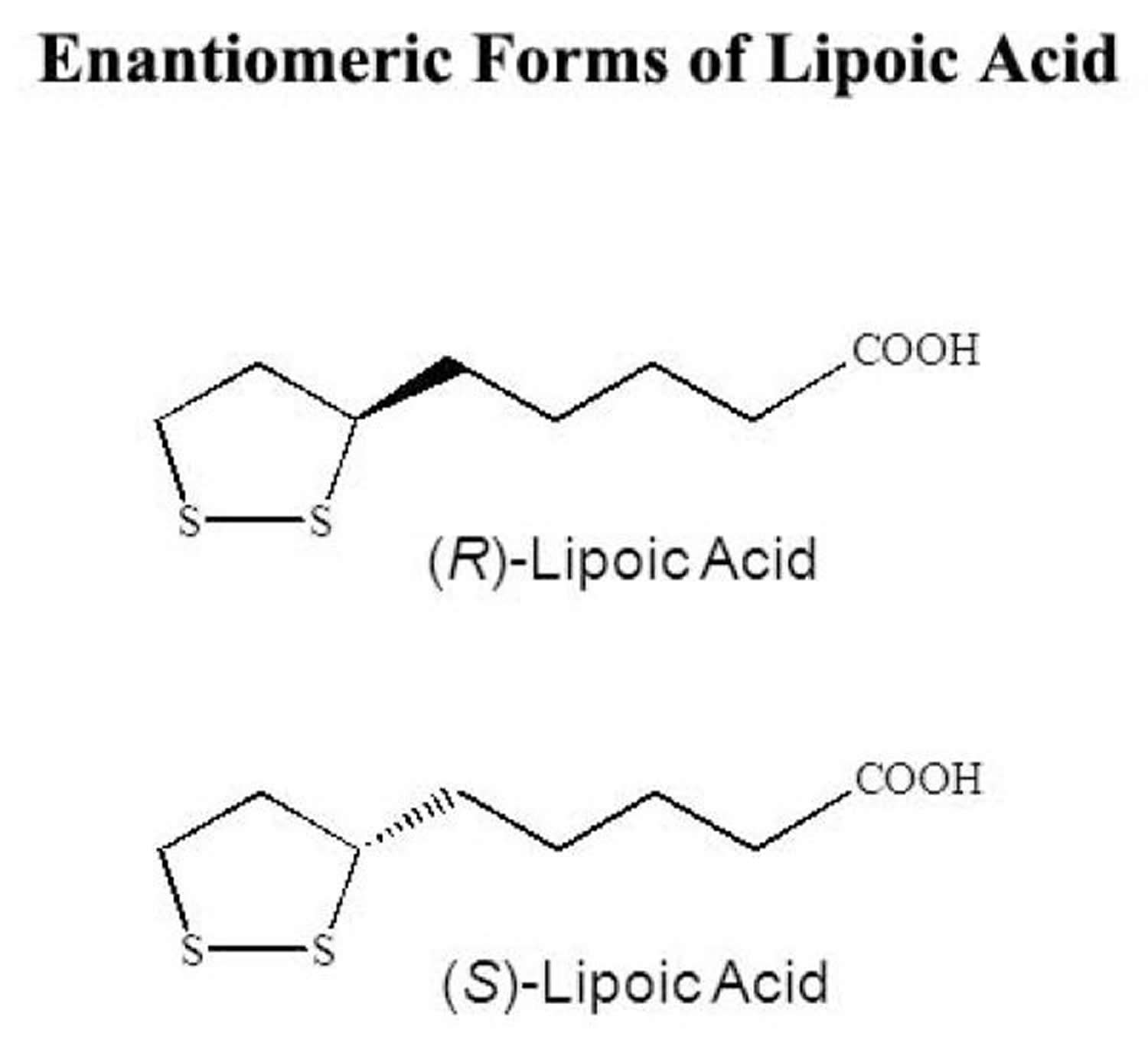
Alpha-Lipoic Acid also called thioctic acid or 1,2-dithiolane-3-pentanoic acid (pentanoic acid), is a naturally occurring short chain fatty acid which contains a thiol bond, synthesized in small amounts by plants and animals (including humans), with antioxidant and potential chemopreventive activities 1. Chemically, alpha lipoic acid is a short-chain fatty acid with a disulfide group in its dithiolane ring and a chiral carbon resulting in R and S enantiomers (see Figure 1 below). Although the majority of the commercially produced alpha lipoic acid consists of a racemic admixture, the R form, (R)-α-Lipoic acid, is the biologically active form that is endogenously produced by the body, while the S form is produced from chemical manufacture and is not biologically active 2. Only R-α-Lipoic acid is conjugated to conserved lysine residues in an amide linkage, thus making this isoform essential as a cofactor in biological systems 3. At the cellular level, alpha lipoic acid is reduced to dihydrolipoic acid (DHLA), which has a number of cellular actions including free radical scavenging and modulating oxidative stress and inflammatory pathways 2. Alpha lipoic acid, when exogenously administered, is readily absorbed from the gut and has been clinically used in Europe for the treatment of diabetic polyneuropathy 4.
Though in body synthesis appears to supply all the necessary alpha lipoic acid needed for its role in intermediary metabolism, alpha lipoic acid can also be absorbed from the diet 5. While the direct roles of alpha lipoic acid as a cofactor are well understood, less is known about the precise metabolic functions of orally supplied alpha lipoic acid.
Figure 1. Alpha lipoic acid R and S enantiomers
Alpha-lipoic acid acts as a free radical scavenger and assists in repairing oxidative damage and regenerates endogenous antioxidants, including vitamins C and E and glutathione. Alpha lipoic acid also promotes glutathione synthesis. In addition, alpha-lipoic acid exerts metal chelating capacities and functions as a cofactor for energy production in the mitochondria in various mitochondrial enzyme complexes involved in the decarboxylation of alpha-keto acids and thus serves a critical role in mitochondrial energy metabolism 6.
In addition to alpha-lipoic acid being made by your body, alpha-lipoic acid is also absorbed intact from dietary sources and can be found in almost all foods, but slightly more so in organ meats (kidney, heart, liver), spinach, broccoli, peas, brussel sprouts, and rice bran. Alpha-lipoic acid can also be made in the laboratory. Alpha-lipoic acid is biosynthesized by cleavage of linoleic acid and is a coenzyme of oxoglutarate dehydrogenase (ketoglutarate dehydrogenase complex).
Alpha lipoic acid is marketed in the US as an over-the-counter nutritional antioxidant supplement, alone or in combination with other antioxidants. In medicine, alpha lipoic acid has been shown to reduce symptoms of diabetic polyneuropathy, and several clinical trials established some efficacy and an excellent safety profile in this patient population 7.
Alpha-lipoic acid is a type of antioxidant and chemoprotective agent. Alpha-lipoic acid is an antioxidant — a substance the body can use to prevent or manage a tissue-damaging process called oxidative stress. Oxidative stress is a part of the diabetic neuropathy disease process. Alpha-lipoic acid also has been shown to reduce blood sugar levels. Alpha lipoic acid is being studied for its ability to protect normal cells from the side effects of chemotherapy and prevent peripheral neuropathy (numbness, tingling, burning, and weakness in the hands or feet) 8. A few small clinical trials have tested the treatment effect of alpha-lipoic acid given either as a supplement or intravenously. People with diabetic neuropathy had reduced pain, improvements in nerve function tests and improvements in other clinical measures of diabetic neuropathy (Han T, Bai J, Liu W, et al. A systematic review and meta-analysis of α-lipoic acid in the treatment of diabetic peripheral neuropathy. European Journal of Endocrinology. 2012;167(4):465-471.)). But long-term studies are needed.
Alpha-lipoic acid is being studied for its effect on complications of diabetes, including diabetic macular edema (an eye condition that can cause vision loss) and diabetic neuropathy (nerve damage caused by diabetes).
- In a 2011 study of 235 people with type 2 diabetes, 2 years of supplementation with alpha-lipoic acid did not help to prevent macular edema 9.
- A 2016 assessment of treatments for symptoms of diabetic neuropathy that included 2 studies of oral alpha-lipoic acid, with a total of 205 participants, indicated that alpha-lipoic acid may be helpful 10.
Alpha-lipoic acid supplements are generally considered safe when taken as recommended. However, toxicity might occur if you take this supplement when you have a significant thiamin (vitamin B-1) deficiency. Don’t use alpha-lipoic acid if you’re a heavy user of alcohol.
Alpha lipoic acid foods
Almost all foods contain alpha lipoic acid, but slightly more so in organ meats (kidney, heart, liver), spinach, broccoli, peas, brussel sprouts, and rice bran.
What is alpha lipoic acid used for
Alpha-lipoic acid for diabetic peripheral neuropathy
People with both types of diabetes develop multisystem complications 11, one of the most frequent being diabetic peripheral neuropathy. Diabetic peripheral neuropathy has an estimated prevalence in the diabetic population of between 10% and 100% depending upon the data source and ascertainment methodology 12.
Diabetic peripheral neuropathy is defined as “the presence of symptoms and/or signs of peripheral nerve dysfunction in people with diabetes after the exclusion of other causes” 13. Diabetic peripheral neuropathy may be asymptomatic and insidious at onset. The most common symptom of diabetic peripheral neuropathy is neuropathic pain, which occurs in up to 50% of people with diabetic peripheral neuropathy and is the most frequent reason for seeking medical care 14. Painful symptoms are varied and include pain, tingling, burning sensations, paresthesia, shooting or lancinating pains, aching, and contact pain (allodynia) provoked by clothing 15.
Diabetic peripheral neuropathy can be classified clinically as either focal or diffuse. Diffuse disease can affect the sensorimotor or the autonomic nervous systems or both. Sensorimotor disease can involve large or small nerve fibers 16, is usually predominantly sensory, and may be painful.
Distal symmetrical sensorimotor polyneuropathy is the most common form of diabetic peripheral neuropathy, with a reported prevalence in diabetes mellitus ranging from 28.5% to 45%, increasing with age and disease duration 17. Distal symmetrical sensorimotor polyneuropathy represents a major cause of morbidity and the leading source of diabetes-related hospitalizations and non-traumatic amputations. It is also accountable for considerable physical disability, altered quality of life, and increased mortality 15.
Diabetic peripheral neuropathy complications are also a major threat to the general well-being and quality of life of people with diabetes. Numbness caused by diabetic peripheral neuropathy, along with retinopathy and vestibular dysfunction, increase the risk of falls two- to three-fold compared to people without diabetic peripheral neuropathy 18. People with diabetic peripheral neuropathy are also seven times more likely to develop foot ulcerations 19. Foot ulcerations further predispose to active or passive soft tissue infection, which can progress to bone infection and subsequent lower extremity amputation 20. Diabetic peripheral neuropathy, peripheral vascular disease, and soft tissue and bone deformity are serious complications that make diabetes the leading cause of lower extremity amputation 21.
Diabetic peripheral neuropathy symptoms are usually assessed using patient-reported outcome measures that quantify discomfort, sleep disturbances, and quality of life 14.
The pathophysiology of diabetic peripheral neuropathy is not fully understood, and very likely to be multifactorial (genetic, environmental, behavioral, metabolic, neurotrophic, and vascular) 22. Oxidative stress generated by excess free-radical formation or errors in antioxidant protection, or both, is thought to be important in the pathogenesis 23. Good glycemic control reduces the risk of developing diabetic peripheral neuropathy, but glycemic control is not always achievable and is usually not sufficient to halt diabetic peripheral neuropathy progression 22.
Diabetic peripheral neuropathy pathophysiology can mainly be explained as neural dysfunction caused by the interplay of decreased blood flow to nerves as a result of hyperglycaemia, and increased oxidative stress, which induces local inflammatory reactions through reactive oxygen species (ROS) 24. Prolonged hyperglycemia simultaneously activates multiple pathways. It promotes the following:
- Activation of polyol and protein kinase pathways that leads to reduced nicotinamide adenine dinucleotide phosphate (NADPH) and subsequent depletion of glutathione and nitric oxide 25.
- Angiogenesis driven by the vascular endothelial growth factor pathway.
- Basement membrane thickening and endothelial proliferation (via transforming growth factor-β and nuclear factor – kappa B), which cause altered capillary permeability and local hypoxia.
- Activation of the hexosamine pathway and shunting of fructose-6-phosphate from the glycolytic pathway.
- Modified gene expression for glucose transporters and glucokinase 26.
Generation of reactive oxygen species and advanced glycosylation end-products activates the same NFkB pathway, which increases oxidative stress with additional nicotinamide adenine dinucleotide phosphate (NADPH) depletion. Oxidative stress also induces poly (ADP-ribose) polymerase activation, which sequentially results in supplementary nicotinamide adenine dinucleotide depletion, positive loop activation of the protein kinase pathway, and promotes inflammation 27. All these pathways promote mitochondrial dysfunction, which in turn is followed by apoptosis, axonal degeneration, and axonal death. Local pro-inflammatory cytokines induced by oxidative stress promote macrophage recruitment with subsequent glial failure, myelin breakdown, and impaired nerve regeneration 28.
The clinical consequences of this hyperglycemia-induced inflammatory and oxidative state are axonal dystrophy, decreased nerve conduction velocity, diminished neurovascular flow and, ultimately, small- and large-fiber neuropathy 16.
Current management of diabetic peripheral neuropathy consists of three therapeutic approaches. The main target is prevention, through control of fasting and postprandial glucose 29. Medications that target symptoms and disease-modifying treatments are used in the treatment of people with diagnosed diabetic peripheral neuropathy. Symptomatic treatments target pain; they include anticonvulsants, tricyclic antidepressants 30, serotonin and noradrenaline reuptake inhibitors 31, opioids and opioid-like drugs 32, systemic local anaesthetics 33, nonsteroidal anti-inflammatory agents 32, and non-drug therapies such as transcutaneous electrical nerve stimulation (TENS), pulsed radiofrequency sympathectomy 34, and acupuncture 35.
Disease-modifying treatments aim to prevent, slow, or reverse diabetic peripheral neuropathy progression by reduction of oxidative stress and inhibition of the polyol, hexosamine, protein kinase, advanced glycosylation product, and poly (ADP-ribose) polymerase pathways.
Alpha lipoic acid acts as a scavenger of reactive oxygen species and has antioxidant properties that could block the oxidative stress–inflammation pathways activated in diabetic peripheral neuropathy 36. Alpha lipoic acid could therefore be useful both in prevention and treatment of diabetic peripheral neuropathy 36.
Early in test tube studies showed that alpha lipoic acid and its reduced form, dihydrolipoic acid (DHLA), scavenge reactive oxygen species, including hydroxyl radicals, hypochlorous acid, and singlet oxygen 37. In animal studies also indicated that alpha lipoic acid decreases oxidative stress 38, participates in restoring endogenous cellular antioxidant levels and reducing pro-inflammatory pathways 39 and may influence the regeneration of vitamins C and E 36.
The benefit of alpha lipoic acid in people with diabetes could range beyond antioxidant and anti-inflammatory effects. The therapeutic properties of alpha lipoic acid might include the ability to restore glucose availability and increase insulin-stimulated glucose transport and non-oxidative and oxidative glucose metabolism in insulin-resistant muscle cells 40. Alpha lipoic acid has therefore been a candidate for clinical study in diabetic peripheral neuropathy.
The interaction of alpha lipoic acid with regulatory components of the insulin signaling cascade has proved functionally beneficial to skeletal muscle glucose uptake, whole-body glucose tolerance, and helpful against insulin resistance in animal models 41. Improvements in glucose disposal were also observed in human patients with type 2 diabetes receiving alpha lipoic acid either intravenously or orally 42. Several clinical trials have been conducted to measure the efficacy of racemic alpha lipoic acid in decreasing symptoms of diabetic polyneuropathies; these are the “alpha-lipoic acid in diabetic neuropathy” (Aalpha lipoic acidDIN) trials and the “symptoms of diabetic polyneuropathy” (SYDNEY) trials. alpha lipoic acid was given orally, intravenously, or i.v. with oral follow-up. A meta-analysis of four clinical trials using i.v. alpha lipoic acid, including Aalpha lipoic acidDIN, SYDNEY, and the first 3 weeks of Aalpha lipoic acidDIN III, showed a significant improvement in diabetic polyneuropathies of the feet and lower limbs in patients infused with alpha lipoic acid 600 mg/day, for three weeks 43. Diabetic patients in the Aalpha lipoic acidDIN II trial were administered alpha lipoic acid i.v. at 600 or 1200 mg/day for 5 days, then oral alpha lipoic acid for 2 years, resulting in improved indices of neuropathy 44. Patients in the Aalpha lipoic acidDIN III study received alpha lipoic acid (600 mg/day i.v.) or placebo for three weeks, followed by oral alpha lipoic acid (600 mg t.i.d.) or placebo for 6 months. The oral phase of this trial, however, was without clinically significant benefits 45. One possible conclusion from these studies was that alpha lipoic acid administered intravenously was more efficacious than oral alpha lipoic acid, which may be due to either greater bioavailability or poor solubility of the medication in the stomach acid. However, some additional studies have found that oral alpha lipoic acid is very effective. For example, the oral pilot (ORPIL) study showed a reduction in diabetic polyneuropathic symptoms after three weeks with 600 mg alpha lipoic acid three times daily 46. While the first SYDNEY trial used i.v. alpha lipoic acid 47, the SYDNEY II study used oral alpha lipoic acid at 600, 1200, or 1800 mg once daily for 5 weeks 48; consequently, both studies showed significant improvements in neuropathic endpoints.
Alpha lipoic acid and weight loss
Previous studies have suggested anti-obesity properties of alpha lipoic acid 49, 50. In animal studies, it was shown that alpha lipoic acid supplementation promotes the reduction of body weight and fat mass by decreasing food intake and enhancing energy expenditure, possibly by suppressing hypothalamic AMP-activated protein kinase (AMPK) activity 51. However, studies in humans with alpha lipoic acid supplementation are limited, and the results have been inconsistent. Some clinical trials have shown that alpha lipoic acid supplementation may help overweight or obese individuals lose weight 52, while other studies have observed no effects of alpha lipoic acid on weight 53, 54. Nevertheless, alpha lipoic acid appears to have a wide range of beneficial effects on obesity related conditions such as insulin resistance, metabolic syndrome, and type II diabetes, including their complications such as vascular damage 55.
Cumulative results in a 2017 meta-analysis showed significant reduction of body weight and BMI (body mass index) with alpha lipoic acid treatment compared to placebo, regardless if it was used for weight loss or other purposes 56. Small but significant reduction of body weight with alpha lipoic acid intervention is in line with previous open label studies that have well documented the effectiveness of alpha lipoic acid on weight loss in overweight and obese individuals 52, 57 and randomized studies 58, 59, 60, 61. These results conclude that alpha lipoic acid supplementation with diet intervention may provide more beneficial effects on body weight management in overweight and obese individuals.
Studies in the 2017 meta-analysis explored various doses of alpha lipoic acid intervention (300 mg/day to 1800 mg/day) on different intervention durations (8 weeks to 52 weeks). Only one placebo controlled study compared the effectiveness of different doses of alpha lipoic acid on body weight 50. Koh et al. 50 explored the effects of 1200 mg/day and 1800 mg/day alpha lipoic acid intervention on body weight loss. Koh et al. 50 found that the higher dose of alpha lipoic acid resulted in significant weight loss and BMI reduction throughout the study compared to placebo. The lower dose of alpha lipoic acid led to significant weight loss in the first weeks of this study, however this effect was not sustainable through the entire duration of the study. From these findings, it can argue that the effect of alpha lipoic acid on body weight is limited to the short term, especially when it is used at lower doses with an adaptation mechanism taking over later. This may have implications for future study designs, for example phasic use of the medication may be tried 62.
In summary, findings from 2017 56 and 2018 meta-analyses 63 suggest that alpha lipoic acid may be a useful supplementation for weight loss in overweight and obese individuals in reducing body weight and BMI, but has no significant effect on waist circumference. The benefits of alpha lipoic acid compared to placebo appear smaller than that of available prescription weight loss medications 64. However, alpha lipoic acid can be considered in clinical practice due to its benign side-effect profile, other beneficial effects such as in diabetic neuropathy, and lower side effects comparing to the available weight loss medications. Further research is needed to examine the effect of different doses and the long-term benefits of alpha lipoic acid on metabolic parameter in unhealthy obese individuals.
Alpha lipoic acid benefits
Effects of alpha lipoic acid on the vascular system
Vascular endothelial cells, which line the blood vessel lumen, form the physical interface between the blood and the vessel wall, preventing platelet adhesion and regulating blood vessel patency. The elasticity of the vessel wall is regulated by nitric oxide (NO), a gas produced by endothelial nitric oxide synthase (eNOS). Loss of endothelial nitric oxide synthase (eNOS) activity causes endothelial dysfunction due to nitric oxide (NO) limitation, and is characterized by reduced vasodilation, a proinflammatory milieu, and a prothrombic state. Oxidative stress has been implicated in endothelial dysfunction on the basis that antioxidants, such as ascorbate and alpha lipoic acid, improve the redox state of the plasma and endothelium-dependent nitric oxide (NO)-mediated vasodilation 65. But the question remains as to how alpha lipoic acid achieves this significant result. It is known, for instance, that the PI3K/Akt signaling pathway, cascading from the insulin receptor and stimulated by alpha lipoic acid, plays an important role in eNOS activation 66. Treating human aortic endothelial cells with alpha lipoic acid significantly increases NO synthesis 67, and alpha lipoic acid improves the loss in eNOS phosphorylation seen in aorta from aged rats through Akt 68. Furthermore, i.p. injection of alpha lipoic acid into old rats restores vasorelaxation, characterized by an increased phosphorylation of both eNOS and Akt, as well as a decrease in neutral sphingomyelinase activity and a concomitant decrease in ceramide 69. These studies using in vitro and animal models strengthen our understanding of the role of the insulin signaling pathway in vasomotor function, and underscore the health potential of alpha lipoic acid therapy. Thus far, however, only the ISalpha lipoic acidND (ISLAND) clinical trial has examined alpha lipoic acid as a potential remedy for endothelial dysfunction 70. This trial was a randomized, double-blind, placebo-controlled study comparing alpha lipoic acid to irbesartan, an angiotensin II receptor antagonist used mainly for the treatment of hypertension. Results showed that the oral administration of alpha lipoic acid (300 mg/day for 4 weeks) and/or irbesartan (150 mg/day for 4 weeks) to 14-15 patients with metabolic syndrome improved endothelial-dependent flow-mediated vasodilation, which was measured by using the noninvasive brachial artery reactivity test. However, larger and more long-term studies are necessary in order to establish the efficacy of alpha lipoic acid as a therapeutic for vascular endothelial dysfunction.
Alpha lipoic acid as a hypotensive agent
Hypertension is a risk factor for stroke, heart attack and arterial aneurysm, and a leading cause of chronic kidney failure. Even moderate elevation in arterial blood pressure correlates with shortened life expectancy. The rationale for the therapeutic use of alpha lipoic acid against hypertension stemmed from its ability to increase tissue GSH levels and prevent deleterious sulfhydryl group modification in Ca2+ channels. Feeding alpha lipoic acid to hypertensive rats normalized systolic blood pressure and cytosolic free Ca2+, and attenuated adverse renal vascular changes 71. The role of alpha lipoic acid in regenerating reduced GSH was further put forth by El Midaoui and de Champlain 72 who associated the restoration of glutathione peroxidase activity seen in alpha lipoic acid-fed rats with the normalization of aortic superoxide production and blood pressure. It was also suggested that dietary alpha lipoic acid inhibits renal and vascular overproduction of endothelin-1, a vasoconstrictor secreted by the endothelium 73. Because NO is the main vasodilator in conduit arteries and the recent finding that alpha lipoic acid improves endothelial NO synthesis 69, pharmacologists have a new rationale to investigate the role of alpha lipoic acid and high blood pressure. Clinically, alpha lipoic acid administration (in combination with acetyl-L-carnitine) showed some promise as an antihypertensive therapy by decreasing systolic pressure in high blood pressure patients and subjects with the metabolic syndrome 74. In contrast, the administration of alpha lipoic acid (300 mg/day for 4 weeks) to patients with the metabolic syndrome had no significant effect on blood pressure compared to placebo group 70.
Alpha lipoic acid as an anti-inflammatory agent
Inflammation results from the innate biological response of vascular tissues to harmful agents, such as pathogens or irritants. It is an attempt by the organism to remove the injurious stimuli, protect the surrounding tissue, and initiate the healing process. However, unabated chronic inflammation also contributes to a host of diseases, such as atherosclerosis, asthma, and rheumatoid arthritis. Elevated levels of oxidative stress play an important role in chronic inflammation. Oxidative stress-associated inflammation is thought to provoke early vascular events in atherogenesis, including the upregulation of vascular adhesion molecules and matrix metalloproteinase activity. These events require the activation of NF-kappaB, a transcription factor that induces expression of many genes involved in inflammation and endothelial cell migration. Given the oxidative nature of inflammation, therapeutic strategies aimed at mitigating oxidant production and oxidative damage have been investigated for decades in various models of inflammation.
In keeping with this strategy, alpha lipoic acid has been studied for its antioxidant properties in cytokine-induced inflammation; it is also widely known as an inhibitor of NF-kappaB 75. Results show that alpha lipoic acid lowers expression of vascular cell adhesion molecule-1 (VCAM-1) and endothelial adhesion of human monocytes 76, and inhibits NF-kappaB-dependent expression of metalloproteinase-9 in vitro 77. Similarly, alpha lipoic acid (25-100 μg/ml = 122-486 μM) prevents the upregulation of intercellular adhesion molecule-1 (ICAM-1) and vascular cell adhesion molecule-1 (VCAM-1) in spinal cords and in TNF-alpha stimulated cultured brain endothelial cells 78. Collagen-induced arthritis was attenuated by alpha lipoic acid (10-100 mg/kg i.p.) in DBA/1 mice by reduction of inflammatory cytokines like TNF-alpha, and partial inhibition of NF-kappaB binding to DNA 79. In this study, alpha lipoic acid also inhibited osteoclast formation, suggesting that alpha lipoic acid may be useful in the prevention of bone erosion and joint destruction in rheumatoid arthritis. In another study, pretreatment of collagen sheets with alpha lipoic acid (2 mg) prior to implantation decreased TNF-alpha-induced bone resorption in ICR mice 79. In experimental autoimmune encephalomyelitis (an animal model of multiple sclerosis) alpha lipoic acid-treated mice showed marked improvement in central nervous system infiltrating T-cells and macrophages, decreased demyelination and spinal cord expression of adhesion molecules (ICAM-1 and VCAM-1) 80. The downregulation of surface CD4 seen in alpha lipoic acid-treated blood mononuclear cells was proposed to account, at least in part, for the modulation of inflammatory cell infiltration into the central nervous system 81. This is because co-receptor CD4 amplifies the signal generated at the T-cell receptor by recruiting lymphocyte protein kinase Lck, which in turn triggers a cascade of events leading to T-cell activation. Interestingly, DHalpha lipoic acid did not downregulate CD4 from the surface of human peripheral blood mononuclear cells 81. As an alternative or in addition to CD4 downregulation, the immunomodulatory properties of alpha lipoic acid may involve the upregulation of cAMP in T-cells and natural killer cells 82. Cell migration and neovascularization were also inhibited by alpha lipoic acid (86 μg/day in drinking water) in c57/black mice injected with Kaposi’s sarcoma in a matrigel sponge, as well as in nude mice injected with KS cells 83. In a mouse model of bronchial asthma, dietary alpha lipoic acid significantly attenuated airway hyper-responsiveness, lowered the eosinophil count among bronchoalveolar lavage cells, and significantly improved pathologic lesion scores of the lungs 84. alpha lipoic acid inhibits TNF-alpha-induced NF-kappaB activation and adhesion molecule expression in human aortic endothelial cells via a mechanism seemingly distinct from antioxidants, such as ascorbate or reduced GSH, but consistent with the workings of a metal chelator 85. Recently, the inhibition of endotoxin-induced acute inflammation by alpha lipoic acid was associated with the stimulation of the PI3K/Akt pathway 86.
To date, the anti-inflammatory properties of alpha lipoic acid have rarely been investigated in humans. The ISalpha lipoic acidND (ISLAND) trial showed a 15% significant decrease in serum interleukin-6 levels following 4 weeks of supplementation with alpha lipoic acid (300 mg/day) 87. This finding may prove important to human health because interleukin-6 is a recognized marker of inflammation in coronary atherosclerotic plaques, and also regulates the expression of other inflammatory cytokines, such as interleukin-1 and TNF-alpha 88. However, the body of evidence is currently too limited to be conclusive.
Alpha lipoic acid dosage
Clinical trials have used different forms of administration and treatment durations. Alpha lipoic acid dosage ranges from 200 mg/day to 1800 mg/day, administered intravenously or orally. Although the maximum dose of alpha lipoic acid has not been defined, previous studies have shown that alpha lipoic acid can be used safely up to as high as 1800 mg/day 50.
However, despite the evidence attesting to its safety in moderate doses, precautions for the oral intake of alpha lipoic acid have also been voiced. Cakatay et al. 89 conducted a series of experiments in aged rats with intraperitoneal administration of racemic alpha lipoic acid (100 mg/kg body weight per day for 2 weeks) and showed that this high chronic dose (the equivalent of 5 to 10 grams per day in humans) increased plasma lipid hydroperoxide levels and oxidative protein damage 89. Alpha lipoic acid-mediated protein damage was noted in rat heart 90 and brain 91, but lipid hydroperoxide levels were beneficially decreased in both these organs. Apparently in keeping with its metal chelating abilities, this group noted that alpha lipoic acid lowered selenium levels in the serum, heart, brain, and muscle; manganese was lowered only in the heart, but increased in the brain and muscle 92. Thus, while intake of moderate doses of alpha lipoic acid have relatively few adverse side-effects, alpha lipoic acid may mediate oxidative insult at higher doses or when administered intraperitoneally. More research is therefore warranted regarding both the safety and optimal dose of alpha lipoic acid.
Alpha lipoic acid side effects
The most commonly reported side effects that were related with alpha lipoic acid in these two studies 93, 94 were gastrointestinal symptoms, such as abdominal pain, nausea, vomiting, and diarrhea 95 and dermatological symptoms, such as urticaria and itching sensation. One subject in the 1800 mg/d alpha-lipoic acid group and 3 subjects in the 1200 mg/d alpha-lipoic acid group withdrew because of itching sensation 93.
References- Alpha-lipoic acid: a multifunctional antioxidant that improves insulin sensitivity in patients with type 2 diabetes. Evans JL, Goldfine ID. Diabetes Technol Ther. 2000 Autumn; 2(3):401-13. https://www.ncbi.nlm.nih.gov/pubmed/11467343/
- Alpha-lipoic acid as a dietary supplement: molecular mechanisms and therapeutic potential. Shay KP, Moreau RF, Smith EJ, Smith AR, Hagen TM. Biochim Biophys Acta. 2009 Oct; 1790(10):1149-60. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2756298/
- Reed L. Multienzyme complexes. Accts Chem Res. 1974;7:40–46.
- The pharmacology of the antioxidant lipoic acid. Biewenga GP, Haenen GR, Bast A. Gen Pharmacol. 1997 Sep; 29(3):315-31. https://www.ncbi.nlm.nih.gov/pubmed/9378235/
- Shay KP, Moreau RF, Smith EJ, Smith AR, Hagen TM. Alpha-lipoic acid as a dietary supplement: Molecular mechanisms and therapeutic potential. Biochimica et biophysica acta. 2009;1790(10):1149-1160. doi:10.1016/j.bbagen.2009.07.026. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2756298/
- Molecular aspects of lipoic acid in the prevention of diabetes complications. Packer L, Kraemer K, Rimbach G. Nutrition. 2001 Oct; 17(10):888-95.
- Ziegler D, Ametov A, Barinov A, et al. Oral treatment with alpha-lipoic acid improves symptomatic diabetic polyneuropathy: the SYDNEY 2 trial. Diabetes Care. 2006;29:2365–2370.
- Alpha-Lipoic Acid. https://ncit.nci.nih.gov/ncitbrowser/ConceptReport.jsp?dictionary=NCI_Thesaurus&ns=NCI_Thesaurus&code=C61595
- Haritoglou C, Gerss J, Hammes HP, et al. Alpha-lipoic acid for the prevention of diabetic macular edema. Ophthalmologica. 2011;226(3):127-137. https://www.ncbi.nlm.nih.gov/pubmed/21811051
- Çakici N, Fakkel TM, van Neck JW, et al. Systematic review of treatments for diabetic peripheral neuropathy. Diabetic Medicine. 2016;33(11):1466-1476.
- World Health Organization. Global Report on Diabetes. 2016. www.who.int/diabetes/global-report/en/
- Feldman E. Epidemiology and classification of diabetic neuropathy. UpToDate®.
- Boulton AJM, Gries FA, Jervell JA. Guidelines for the diagnosis and outpatient management diabetic peripheral neuropathy. Diabetic Medicine 1998;15(6):508-14.
- Bredfeldt C, Altschuler A, Adams AS, Ports J, Bayliss E. Patient reported outcomes for diabetic peripheral neuropathy. Journal of Diabetes and Its Complications 2015;29(8):1112-8.
- Tesfaye S, Vileikyte L, Rayman G, Sindrup SH, Perkins BA, Baconja M, et al. Painful diabetic peripheral neuropathy: consensus recommendations on diagnosis, assessment and management. Diabetes/Metabolism Research and Reviews 2011;27(7):629-38.
- Edwards JL, Vincent AM Cheng HT, Feldman EL. Diabetic neuropathy: mechanisms to management. Pharmacology & Therapeutics 2008;120(1):1-34.
- Harris M, Eastman R, Cowie C. Symptoms of sensory neuropathy in adults with NIDDM in the US population. Diabetes Care 1993;16(11):1446–52.
- Agrawal Y, Carey JP, Della Santina CC, Schubert MC, Minor LB. Diabetes, vestibular dysfunction, and falls: analyses from the National Health and Nutrition Examination Survey. Otology & Neurotology 2010;31(9):1445-50.
- Amin N, Doupis J. Diabetic foot disease: From the evaluation of the “foot at risk” to the novel diabetic ulcer treatment modalities. World Journal of Diabetes 2016;7(7):153-64.
- Kim PJ, Steinberg JS. Complications of the diabetic foot. Endocrinology and Metabolism Clinics of North America 2013;42(4):833-47.
- Callaghan BC, Cheng HT, Stables CL, Smith AL, Feldman EL. Diabetic neuropathy: clinical manifestations and current treatments. Lancet Neurology 2012;11(6):521-34.
- Chen W, Zhang Y, Li X, Yang G, Liu JP. Chinese herbal medicine for diabetic peripheral neuropathy. Cochrane Database of Systematic Reviews 2013, Issue 10.
- Low PA, Nickander KK, Tritschler HJ. The roles of oxidative stress and antioxidant treatment in experimental diabetic neuropathy. Diabetes 1997;46(Suppl 2):S38-42.
- Brownlee M. The pathobiology of diabetic complications: a unifying mechanism. Diabetes 2005;54(6):1615–25.
- Uehara K, Yamagishi S, Otsuki S, Chin S, Yagihashi S. Effects of polyol pathway hyperactivity on protein kinase C activity, nociceptive peptide expression, and neuronal structure in dorsal root ganglia in diabetic mice. Diabetes 2004;53(12):3239–47.
- Kolm-Litty V, Sauer U, Nerlich A, Lehmann R, Schleicher ED. High glucose-induced transforming growth factor beta1 production is mediated by the hexosamine pathway in porcine glomerular mesangial cells. Journal of Clinical Investigation 1998;101(1):160-9.
- Vinik AI, Mehrabyan A. Diabetic neuropathies. Medical Clinics of North America 2004;88(4):947-99.
- Wang Y, Schmeichel AM, Iida H, Schmelzer JD, Low PA. Enhanced inflammatory response via activation of NF-kappaB in acute experimental diabetic neuropathy subjected to ischemia reperfusion injury. Journal of the Neurological Sciences 2006;247(1):47-52.
- Callaghan BC, Little AA, Feldman EL, Hughes RA. Enhanced glucose control for preventing and treating diabetic neuropathy. Cochrane Database of Systematic Reviews 2012, Issue 6.
- Lunn MP, Hughes RA, Wiffen PJ. Duloxetine for treating painful neuropathy, chronic pain or fibromyalgia. Cochrane Database of Systematic Reviews 2014, Issue 1.
- Allen R, Sharma U, Barlas S. Clinical experience with desvenlafaxine in treatment of pain associated with diabetic peripheral neuropathy. Journal of Pain Research 2014;7:339-51.
- Snedecor SJ, Sudharshan L, Cappelleri JC, Sadosky A, Mehta S, Botteman M. Systematic review and meta-analysis of pharmacological therapies for painful diabetic peripheral neuropathy. Pain Practice 2014;14(2):167-84.
- Challapalli V, Tremont-Lukats IW, McNicol ED, Lau J, Carr DB. Systemic administration of local anaesthetic agents to relieve neuropathic pain. Cochrane Database of Systematic Reviews 2005, Issue 4.
- Naderi Nabi B, Sedighinejad A, Haghighi M, Biazar G, Hashemi M, Haddadi S, et al. Comparison of transcutaneous electrical nerve stimulation and pulsed radiofrequency sympathectomy for treating painful diabetic neuropathy. Anesthesiology and Pain Medicine 2015;5(5):e29280.
- Zhang C, Ma YX, Yan Y. Clinical effects of acupuncture for diabetic peripheral neuropathy. Chung i Tsa Chih Ying Wen Pan [Journal of Traditional Chinese Medicine] 2010;30(1):13-4.
- Rochette L, Ghibu S, Muresan A, Vergely C. Alpha-lipoic acid: molecular mechanisms and therapeutic potential in diabetes. Canadian Journal of Physiology and Pharmacology 2015;93(12):1021-7.
- Packer L, Witt EH, Tritschler HJ. Alpha-lipoic acid as a biological antioxidant. Free Radical Biology & Medicine 1995;19(2):227-50.
- Marangon K, Devaraj S, Tirosh O, Packer L, Jialal I. Comparison of the effect of alpha-lipoic acid and alpha-tocopherol supplementation on measures of oxidative stress. Free Radical Biology & Medicine 1999;27(9-10):1114-21.
- Petersen Shay K, Moreau RF, Smith EJ, Hagen TM. Is alpha-lipoic acid a scavenger of reactive oxygen species in vivo? Evidence for its initiation of stress signaling pathways that promote endogenous antioxidant capacity. IUBMB Life 2008;60(6):362-7.
- Khanna S, Roy S, Packer L, Sen CK. Cytokine-induced glucose uptake in skeletal muscle: redox regulation and the role of alpha-lipoic acid. American Journal of Physiology 1999;276(5 Pt 2):R1327-33.
- Streeper RS, Henriksen EJ, Jacob S, Hokama JY, Fogt DL, Tritschler HJ. Differential effects of lipoic acid stereoisomers on glucose metabolism in insulin-resistant skeletal muscle. Am J Physiol. 1997;273:E185–91.
- Konrad T, Vicini P, Kusterer K, Hoflich A, Assadkhani A, Bohles HJ, Sewell A, Tritschler HJ, Cobelli C, Usadel KH. alpha-Lipoic acid treatment decreases serum lactate and pyruvate concentrations and improves glucose effectiveness in lean and obese patients with type 2 diabetes. Diabetes Care. 1999;22:280–7.
- Ziegler D, Nowak H, Kempler P, Vargha P, Low PA. Treatment of symptomatic diabetic polyneuropathy with the antioxidant alpha-lipoic acid: a meta-analysis. Diabet Med. 2004;21:114–21. https://www.ncbi.nlm.nih.gov/pubmed/14984445
- Reljanovic M, Reichel G, Rett K, Lobisch M, Schuette K, Moller W, Tritschler HJ, Mehnert H. Treatment of diabetic polyneuropathy with the antioxidant thioctic acid (alpha-lipoic acid): a two year multicenter randomized double-blind placebo-controlled trial (ALADIN II). Alpha Lipoic Acid in Diabetic Neuropathy. Free Radic Res. 1999;31:171–9. https://www.ncbi.nlm.nih.gov/pubmed/10499773
- Ziegler D, Hanefeld M, Ruhnau KJ, Hasche H, Lobisch M, Schutte K, Kerum G, Malessa R. Treatment of symptomatic diabetic polyneuropathy with the antioxidant alpha-lipoic acid: a 7-month multicenter randomized controlled trial (ALADIN III Study). ALADIN III Study Group. Alpha-Lipoic Acid in Diabetic Neuropathy. Diabetes Care. 1999;22:1296–301. https://www.ncbi.nlm.nih.gov/pubmed/10480774
- Ruhnau KJ, Meissner HP, Finn JR, Reljanovic M, Lobisch M, Schutte K, Nehrdich D, Tritschler HJ, Mehnert H, Ziegler D. Effects of 3-week oral treatment with the antioxidant thioctic acid (alpha-lipoic acid) in symptomatic diabetic polyneuropathy. Diabet Med. 1999;16:1040–3. https://www.ncbi.nlm.nih.gov/pubmed/10656234
- Ametov AS, Barinov A, Dyck PJ, Hermann R, Kozlova N, Litchy WJ, Low PA, Nehrdich D, Novosadova M, O’Brien PC, Reljanovic M, Samigullin R, Schuette K, Strokov I, Tritschler HJ, Wessel K, Yakhno N, Ziegler D. The sensory symptoms of diabetic polyneuropathy are improved with alpha-lipoic acid: the SYDNEY trial. Diabetes Care. 2003;26:770–6. https://www.ncbi.nlm.nih.gov/pubmed/12610036
- Ziegler D, Ametov A, Barinov A, Dyck PJ, Gurieva I, Low PA, Munzel U, Yakhno N, Raz I, Novosadova M, Maus J, Samigullin R. Oral treatment with alpha-lipoic acid improves symptomatic diabetic polyneuropathy: the SYDNEY 2 trial. Diabetes Care. 2006;29:2365–70. https://www.ncbi.nlm.nih.gov/pubmed/17065669
- Huerta AE, Navas-Carretero S, Prieto-Hontoria PL, Martinez JA, Moreno-Aliaga MJ. Effects of alpha-lipoic acid and eicosapentaenoic acid in overweight and obese women during weight loss. Obesity. 2015;23:313–321.
- Koh EH, Lee WJ, Lee SA, et al. Effects of alpha-lipoic Acid on body weight in obese subjects. Am J Med. 2011;124:85.e1–8.
- Prieto-Hontoria PL, Perez-Matute P, Fernandez-Galilea M, Martinez JA, Moreno-Aliaga MJ. Effects of lipoic acid on AMPK and adiponectin in adipose tissue of low- and high-fat-fed rats. Eur J Nutr. 2013;52:779–787
- Carbonelli MG, Di Renzo L, Bigioni M, Di Daniele N, De Lorenzo A, Fusco MA. Alpha-lipoic acid supplementation: a tool for obesity therapy? Curr Pharm Des. 2010;16:840–846.
- Ansar H, Mazloom Z, Kazemi F, Hejazi N. Effect of alpha-lipoic acid on blood glucose, insulin resistance and glutathione peroxidase of type 2 diabetic patients. Saudi Med J. 2011;32:584–588.
- McNeilly AM, Davison GW, Murphy MH, et al. Effect of alpha-lipoic acid and exercise training on cardiovascular disease risk in obesity with impaired glucose tolerance. Lipids Health Dis. 2011;10:217.
- Ziegler D. Thioctic acid for patients with symptomatic diabetic polyneuropathy: a critical review. Treat Endocrinol. 2004;3:173–189.
- Kucukgoncu S, Zhou E, Lucas KB, Tek C. Alpha-Lipoic Acid (ALA) as a supplementation for weight loss: Results from a Meta-Analysis of Randomized Controlled Trials. Obesity reviews : an official journal of the International Association for the Study of Obesity. 2017;18(5):594-601. doi:10.1111/obr.12528. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5523816/
- Ratliff JC, Palmese LB, Reutenauer EL, Tek C. An open-label pilot trial of alpha-lipoic acid for weight loss in patients with schizophrenia without diabetes. Clin Schizophr Relat Psychoses. 2013;8:196–200.
- Huerta AE, Navas-Carretero S, Prieto-Hontoria PL, Martinez JA, Moreno-Aliaga MJ. Effects of alpha-lipoic acid and eicosapentaenoic acid in overweight and obese women during weight loss. Obesity. 2015;23:313–321
- Koh EH, Lee WJ, Lee SA, et al. Effects of alpha-lipoic Acid on body weight in obese subjects. Am J Med. 2011;124:85.e1–8
- Kim NW, Song YM, Kim E, et al. Adjunctive alpha-lipoic acid reduces weight gain compared with placebo at 12 weeks in schizophrenic patients treated with atypical antipsychotics: a double-blind randomized placebo-controlled study. Int Clin Psychopharmacol. 2016;31:265–274.
- Mohammadi V, Khalili M, Eghtesadi S, et al. The effect of alpha-lipoic acid (ALA) supplementation on cardiovascular risk factors in men with chronic spinal cord injury: a clinical trial. Spinal Cord. 2015;53:646.
- Kucukgoncu S, Zhou E, Lucas KB, Tek C. Alpha-Lipoic Acid (ALA) as a supplementation for weight loss: Results from a Meta-Analysis of Randomized Controlled Trials. Obesity reviews : an official journal of the International Association for the Study of Obesity. 2017;18(5):594-601. doi:10.1111/obr.12528 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5523816/
- Alpha-lipoic acid supplement in obesity treatment: A systematic review and meta-analysis of clinical trials. Clinical Nutrition April 2018; Volume 37, Issue 2, Pages 419–428. https://www.clinicalnutritionjournal.com/article/S0261-5614(17)30212-1/fulltext
- Khera R, Murad MH, Chandar AK, et al. Association of Pharmacological Treatments for Obesity With Weight Loss and Adverse Events: A Systematic Review and Meta-analysis. JAMA. 2016;315:2424–2434.
- Sena CM, Nunes E, Louro T, Proenca T, Fernandes R, Boarder MR, Seica RM. Effects of alpha-lipoic acid on endothelial function in aged diabetic and high-fat fed rats. Br J Pharmacol. 2007
- Montagnani M, Ravichandran LV, Chen H, Esposito DL, Quon MJ. Insulin receptor substrate-1 and phosphoinositide-dependent kinase-1 are required for insulin-stimulated production of nitric oxide in endothelial cells. Mol Endocrinol. 2002;16:1931–42.
- Hagen TM, Moreau R, Suh JH, Visioli F. Mitochondrial decay in the aging rat heart: evidence for improvement by dietary supplementation with acetyl-L-carnitine and/or lipoic acid. Ann N Y Acad Sci. 2002;959:491–507.
- Smith AR, Hagen TM. Vascular endothelial dysfunction in aging: loss of Akt-dependent endothelial nitric oxide synthase phosphorylation and partial restoration by (R)-alpha-lipoic acid. Biochem Soc Trans. 2003;31:1447–9.
- Shay K. Petersen, Moreau RF, Smith EJ, Hagen TM. Is alpha-lipoic acid a scavenger of reactive oxygen species in vivo? Evidence for its initiation of stress signaling pathways that promote endogenous antioxidant capacity. IUBMB Life. 2008;60:362–7.
- Sola S, Mir MQ, Cheema FA, Khan-Merchant N, Menon RG, Parthasarathy S, Khan BV. Irbesartan and lipoic acid improve endothelial function and reduce markers of inflammation in the metabolic syndrome: results of the Irbesartan and Lipoic Acid in Endothelial Dysfunction (ISLAND) study. Circulation. 2005;111:343–8.
- Vasdev S, Gill V, Longerich L, Parai S, Gadag V. Salt-induced hypertension in WKY rats: prevention by alpha-lipoic acid supplementation. Mol Cell Biochem. 2003;254:319–26.
- El Midaoui A, de Champlain J. Prevention of hypertension, insulin resistance, and oxidative stress by alpha-lipoic acid. Hypertension. 2002;39:303–7.
- Takaoka M, Kobayashi Y, Yuba M, Ohkita M, Matsumura Y. Effects of alpha-lipoic acid on deoxycorticosterone acetate-salt-induced hypertension in rats. Eur J Pharmacol. 2001;424:121–9.
- McMackin CJ, Widlansky ME, Hamburg NM, Huang AL, Weller S, Holbrook M, Gokce N, Hagen TM, Keaney JF, Jr., Vita JA. Effect of combined treatment with alpha-Lipoic acid and acetyl-L-carnitine on vascular function and blood pressure in patients with coronary artery disease. J Clin Hypertens (Greenwich) 2007;9:249–55.
- Packer L, Witt EH, Tritschler HJ. alpha-Lipoic acid as a biological antioxidant. Free Radic Biol Med. 1995;19:227–50.
- Kunt T, Forst T, Wilhelm A, Tritschler H, Pfuetzner A, Harzer O, Engelbach M, Zschaebitz A, Stofft E, Beyer J. Alpha-lipoic acid reduces expression of vascular cell adhesion molecule-1 and endothelial adhesion of human monocytes after stimulation with advanced glycation end products. Clin Sci (Lond) 1999;96:75–82.
- Kim HS, Kim HJ, Park KG, Kim YN, Kwon TK, Park JY, Lee KU, Kim JG, Lee IK. Alpha-lipoic acid inhibits matrix metalloproteinase-9 expression by inhibiting NF-kappaB transcriptional activity. Exp Mol Med. 2007;39:106–13.
- Chaudhary P, Marracci GH, Bourdette DN. Lipoic acid inhibits expression of ICAM-1 and VCAM-1 by CNS endothelial cells and T cell migration into the spinal cord in experimental autoimmune encephalomyelitis. J Neuroimmunol. 2006;175:87–96.
- Lee EY, Lee CK, Lee KU, Park JY, Cho KJ, Cho YS, Lee HR, Moon SH, Moon HB, Yoo B. Alpha-lipoic acid suppresses the development of collagen-induced arthritis and protects against bone destruction in mice. Rheumatol Int. 2007;27:225–33.
- Morini M, Roccatagliata L, Dell’Eva R, Pedemonte E, Furlan R, Minghelli S, Giunti D, Pfeffer U, Marchese M, Noonan D, Mancardi G, Albini A, Uccelli A. Alpha-lipoic acid is effective in prevention and treatment of experimental autoimmune encephalomyelitis. J Neuroimmunol. 2004;148:146–53
- Marracci GH, Marquardt WE, Strehlow A, McKeon GP, Gross J, Buck DC, Kozell LB, Bourdette DN. Lipoic acid downmodulates CD4 from human T lymphocytes by dissociation of p56(Lck) Biochem Biophys Res Commun. 2006;344:963–71
- Schillace RV, Pisenti N, Pattamanuch N, Galligan S, Marracci GH, Bourdette DN, Carr DW. Lipoic acid stimulates cAMP production in T lymphocytes and NK cells. Biochem Biophys Res Commun. 2007;354:259–64
- Larghero P, Vene R, Minghelli S, Travaini G, Morini M, Ferrari N, Pfeffer U, Noonan DM, Albini A, Benelli R. Biological assays and genomic analysis reveal lipoic acid modulation of endothelial cell behavior and gene expression. Carcinogenesis. 2007;28:1008–20
- Cho YS, Lee J, Lee TH, Lee EY, Lee KU, Park JY, Moon HB. alpha-Lipoic acid inhibits airway inflammation and hyperresponsiveness in a mouse model of asthma. J Allergy Clin Immunol. 2004;114:429–35.
- Zhang WJ, Frei B. Alpha-lipoic acid inhibits TNF-alpha-induced NF-kappaB activation and adhesion molecule expression in human aortic endothelial cells. Faseb J. 2001;15:2423–32.
- Zhang WJ, Wei H, Hagen T, Frei B. Alpha-lipoic acid attenuates LPS-induced inflammatory responses by activating the phosphoinositide 3-kinase/Akt signaling pathway. Proc Natl Acad Sci U S A. 2007;104:4077–82
- Sola S, Mir MQ, Cheema FA, Khan-Merchant N, Menon RG, Parthasarathy S, Khan BV. Irbesartan and lipoic acid improve endothelial function and reduce markers of inflammation in the metabolic syndrome: results of the Irbesartan and Lipoic Acid in Endothelial Dysfunction (ISLAND) study. Circulation. 2005;111:343–8
- Ikeda U, Ito T, Shimada K. Interleukin-6 and acute coronary syndrome. Clin Cardiol. 2001;24:701–4.
- Plasma protein oxidation in aging rats after alpha-lipoic acid administration. Cakatay U, Kayali R. Biogerontology. 2005; 6(2):87-93. https://www.ncbi.nlm.nih.gov/pubmed/16034676/
- Prooxidant activities of alpha-lipoic acid on oxidative protein damage in the aging rat heart muscle. Cakatay U, Kayali R, Sivas A, Tekeli F. Arch Gerontol Geriatr. 2005 May-Jun; 40(3):231-40. https://www.ncbi.nlm.nih.gov/pubmed/15814157/
- Effect of alpha-lipoic acid supplementation on markers of protein oxidation in post-mitotic tissues of ageing rat. Kayali R, Cakatay U, Akçay T, Altuğ T. Cell Biochem Funct. 2006 Jan-Feb; 24(1):79-85. https://www.ncbi.nlm.nih.gov/pubmed/15532093/
- Postmitotic tissue selenium and manganese levels in alpha-lipoic acid-supplemented aged rats. Cakatay U, Kayali R, Kiziler AR, Aydemir B. Chem Biol Interact. 2008 Feb 15; 171(3):306-11. https://www.ncbi.nlm.nih.gov/pubmed/17996229/
- Koh EH, Lee WJ, Lee SA, et al. Effects of alpha-lipoic Acid on body weight in obese subjects. Am J Med. 2011;124:85.e1–8. https://www.amjmed.com/article/S0002-9343(10)00743-6/fulltext
- Kim NW, Song YM, Kim E, et al. Adjunctive alpha-lipoic acid reduces weight gain compared with placebo at 12 weeks in schizophrenic patients treated with atypical antipsychotics: a double-blind randomized placebo-controlled study. Int Clin Psychopharmacol. 2016;31:265–274. https://www.ncbi.nlm.nih.gov/pubmed/27276401
- Ziegler, D., Ametov, A., Barinov, A. et al. Oral treatment with alpha-lipoic acid improves symptomatic diabetic polyneuropathy: the SYDNEY 2 trial. Diabetes Care. 2006; 29: 2365–2370