Apple fruit
Apple (Malus sp., Rosaceae) is cultivated worldwide as a fruit tree, and is the most widely grown species in the genus Malus. The apple tree originated in Central Asia, where its wild ancestor, Malus sieversii, is still found today. Apples have been grown for thousands of years in Asia and Europe, and were brought to North America by European colonists.
Apples are popular because of the many ways that they can be consumed and because of their convenience and durability. Apples can be processed into sauce, slices, or juice and are favored for pastries, cakes, tarts, and pies.
The apple pulp has been processed into candies (fruit leathers) and used as a source of pectin. The apple juice can be consumed fresh, either natural or filtered, fermented into alcoholic beverages such as cider or wine, distilled into brandy, or transformed into vinegar.
Is apple a fruit ?
By definition, a fruit is the sweet and fleshy product of a tree or other plant that contains seed and can be eaten as food.
In fact apples are common fruits consumed worldwide!
Apple fruit facts
The apple fruit matures in late summer or autumn and cultivars exist with a wide range of sizes. Commercial growers aim to produce an apple that is 7.0 to 8.3 cm (2.75 to 3.25 in) in diameter, due to market preference. Some consumers, especially those in Japan, prefer a larger apple, while apples below 5.7 cm (2.25 in) are generally used for making juice and have little fresh market value. The skin of ripe apples is generally red, yellow, green, pink, or russetted although many bi- or tri-colored cultivars may be found 1. The skin may also be wholly or partly russeted i.e. rough and brown. The skin is covered in a protective layer of epicuticular wax 2. The exocarp (flesh) is generally pale yellowish-white, though pink or yellow exocarps also occur.
Commercially, apples can be stored for some months in controlled atmosphere chambers to delay ethylene-induced ripening. Apples are commonly stored in chambers with higher concentrations of carbon dioxide and high air filtration. This prevents ethylene concentrations from rising to higher amounts and preventing ripening from occurring too quickly. Ripening continues when the fruit is removed from storage. For home storage, most cultivars of apple can be held for approximately two weeks when kept at the coolest part of the refrigerator (i.e. below 5 °C). Some, including ‘Granny Smith’ and ‘Fuji’, can be stored up to a year without significant degradation.
Non-organic apples may be sprayed with 1-methylcyclopropene blocking the apples’ ethylene receptors, temporarily preventing them from ripening.
Apple nutrition facts
A typical apple serving weighs 242 grams and provides 126 calories with a moderate content of dietary fiber (Table 1). Otherwise, there is generally low content of essential nutrients.
Apples are a rich source of various phytochemicals including flavonoids (e.g., catechins, flavanols, and quercetin) and other phenolic compounds (e.g., epicatechin and procyanidins) found in the skin, core, and pulp of the apple 3. Polyphenolic compounds account for the color, flavor, taste, and metabolic activity of plant-based foods as well as the putative health benefits to humans. The concentration of polyphenols is influenced by the plant variety as well as environmental factors, including geographic region, growing season, and storage 4. The wide range of polyphenol content in whole apples and apple juice is depicted in Table 2. Typically, the greater values in the ranges shown for juices reflect the higher concentration of polyphenols in fresh juice prepared from cider apples and commercial preparations of “cloudy” juice compared to lower quantities in fresh juice made with dessert apple varieties or commercial juices that are clear 5.
Phenolic compounds, such as polyphenol oxidase, are the main driving force behind browning in apples. Polyphenol oxidase catalyzes the reaction of phenolic compounds to o-quinones causing the pigment to turn darker and therefore brown.
Ideain (cyanidin 3-O-galactoside) is an anthocyanin, a type of pigment, which is found in some red apple cultivars 6.
Phlorizin is a flavonoid that is found in apple trees, particularly in the leaves, and in only small amounts if at all in other plants, even other species of the Malus genus or related plants such as pear trees 7.
The estimated dietary intake of polyphenolic compounds varies widely depending upon methodology, consumption data, the combination of compounds (most commonly flavonoids) included in the analysis, and the food composition database used 8. In general, apple are among the top 3 or 4 dietary sources of total phenolics consumed in America and worldwide 9.
Table 1. Apple (raw with skin) nutrition facts
| Nutrient | Unit | Value per 100 g | ||||||||
| Approximates | ||||||||||
| Water | g | 85.56 | ||||||||
| Energy | kcal | 52 | ||||||||
| Energy | kJ | 218 | ||||||||
| Protein | g | 0.26 | ||||||||
| Total lipid (fat) | g | 0.17 | ||||||||
| Ash | g | 0.19 | ||||||||
| Carbohydrate, by difference | g | 13.81 | ||||||||
| Fiber, total dietary | g | 2.4 | ||||||||
| Sugars, total | g | 10.39 | ||||||||
| Sucrose | g | 2.07 | ||||||||
| Glucose (dextrose) | g | 2.43 | ||||||||
| Fructose | g | 5.9 | ||||||||
| Lactose | g | 0 | ||||||||
| Maltose | g | 0 | ||||||||
| Galactose | g | 0 | ||||||||
| Starch | g | 0.05 | ||||||||
| Minerals | ||||||||||
| Calcium, Ca | mg | 6 | ||||||||
| Iron, Fe | mg | 0.12 | ||||||||
| Magnesium, Mg | mg | 5 | ||||||||
| Phosphorus, P | mg | 11 | ||||||||
| Potassium, K | mg | 107 | ||||||||
| Sodium, Na | mg | 1 | ||||||||
| Zinc, Zn | mg | 0.04 | ||||||||
| Copper, Cu | mg | 0.027 | ||||||||
| Manganese, Mn | mg | 0.035 | ||||||||
| Selenium, Se | µg | 0 | ||||||||
| Fluoride, F | µg | 3.3 | ||||||||
| Vitamins | ||||||||||
| Vitamin C, total ascorbic acid | mg | 4.6 | ||||||||
| Thiamin | mg | 0.017 | ||||||||
| Riboflavin | mg | 0.026 | ||||||||
| Niacin | mg | 0.091 | ||||||||
| Pantothenic acid | mg | 0.061 | ||||||||
| Vitamin B-6 | mg | 0.041 | ||||||||
| Folate, total | µg | 3 | ||||||||
| Folic acid | µg | 0 | ||||||||
| Folate, food | µg | 3 | ||||||||
| Folate, DFE | µg | 3 | ||||||||
| Choline, total | mg | 3.4 | ||||||||
| Betaine | mg | 0.1 | ||||||||
| Vitamin B-12 | µg | 0 | ||||||||
| Vitamin B-12, added | µg | 0 | ||||||||
| Vitamin A, RAE | µg | 3 | ||||||||
| Retinol | µg | 0 | ||||||||
| Carotene, beta | µg | 27 | ||||||||
| Carotene, alpha | µg | 0 | ||||||||
| Cryptoxanthin, beta | µg | 11 | ||||||||
| Vitamin A, IU | IU | 54 | ||||||||
| Lycopene | µg | 0 | ||||||||
| Lutein + zeaxanthin | µg | 29 | ||||||||
| Vitamin E (alpha-tocopherol) | mg | 0.18 | ||||||||
| Vitamin E, added | mg | 0 | ||||||||
| Tocopherol, beta | mg | 0 | ||||||||
| Tocopherol, gamma | mg | 0 | ||||||||
| Tocopherol, delta | mg | 0 | ||||||||
| Vitamin D (D2 + D3) | µg | 0 | ||||||||
| Vitamin D | IU | 0 | ||||||||
| Vitamin K (phylloquinone) | µg | 2.2 | ||||||||
| Lipids | ||||||||||
| Fatty acids, total saturated | g | 0.028 | ||||||||
| 04:00:00 | g | 0 | ||||||||
| 06:00:00 | g | 0 | ||||||||
| 08:00:00 | g | 0 | ||||||||
| 10:00:00 | g | 0 | ||||||||
| 12:00:00 | g | 0 | ||||||||
| 14:00:00 | g | 0.001 | ||||||||
| 16:00:00 | g | 0.024 | ||||||||
| 18:00:00 | g | 0.003 | ||||||||
| Fatty acids, total monounsaturated | g | 0.007 | ||||||||
| 16:1 undifferentiated | g | 0 | ||||||||
| 18:1 undifferentiated | g | 0.007 | ||||||||
| 20:01:00 | g | 0 | ||||||||
| 22:1 undifferentiated | g | 0 | ||||||||
| Fatty acids, total polyunsaturated | g | 0.051 | ||||||||
| 18:2 undifferentiated | g | 0.043 | ||||||||
| 18:3 undifferentiated | g | 0.009 | ||||||||
| 18:04:00 | g | 0 | ||||||||
| 20:4 undifferentiated | g | 0 | ||||||||
| 20:5 n-3 (EPA) | g | 0 | ||||||||
| 22:5 n-3 (DPA) | g | 0 | ||||||||
| 22:6 n-3 (DHA) | g | 0 | ||||||||
| Fatty acids, total trans | g | 0 | ||||||||
| Cholesterol | mg | 0 | ||||||||
| Phytosterols | mg | 12 | ||||||||
| Amino Acids | ||||||||||
| Tryptophan | g | 0.001 | ||||||||
| Threonine | g | 0.006 | ||||||||
| Isoleucine | g | 0.006 | ||||||||
| Leucine | g | 0.013 | ||||||||
| Lysine | g | 0.012 | ||||||||
| Methionine | g | 0.001 | ||||||||
| Cystine | g | 0.001 | ||||||||
| Phenylalanine | g | 0.006 | ||||||||
| Tyrosine | g | 0.001 | ||||||||
| Valine | g | 0.012 | ||||||||
| Arginine | g | 0.006 | ||||||||
| Histidine | g | 0.005 | ||||||||
| Alanine | g | 0.011 | ||||||||
| Aspartic acid | g | 0.07 | ||||||||
| Glutamic acid | g | 0.025 | ||||||||
| Glycine | g | 0.009 | ||||||||
| Proline | g | 0.006 | ||||||||
| Serine | g | 0.01 | ||||||||
| Other | ||||||||||
| Alcohol, ethyl | g | 0 | ||||||||
| Caffeine | mg | 0 | ||||||||
| Theobromine | mg | 0 | ||||||||
| Anthocyanidins | ||||||||||
| Cyanidin | mg | 1.6 | ||||||||
| Petunidin | mg | 0 | ||||||||
| Delphinidin | mg | 0 | ||||||||
| Malvidin | mg | 0 | ||||||||
| Pelargonidin | mg | 0 | ||||||||
| Peonidin | mg | 0 | ||||||||
| Flavan-3-ols | ||||||||||
| (+)-Catechin | mg | 1.3 | ||||||||
| (-)-Epigallocatechin | mg | 0.3 | ||||||||
| (-)-Epicatechin | mg | 7.5 | ||||||||
| (-)-Epicatechin 3-gallate | mg | 0 | ||||||||
| (-)-Epigallocatechin 3-gallate | mg | 0.2 | ||||||||
| (+)-Gallocatechin | mg | 0 | ||||||||
| Flavanones | ||||||||||
| Hesperetin | mg | 0 | ||||||||
| Naringenin | mg | 0 | ||||||||
| Flavones | ||||||||||
| Apigenin | mg | 0 | ||||||||
| Luteolin | mg | 0.1 | ||||||||
| Flavonols | ||||||||||
| Kaempferol | mg | 0.1 | ||||||||
| Myricetin | mg | 0 | ||||||||
| Quercetin | mg | 4 | ||||||||
| Isoflavones | ||||||||||
| Daidzein | mg | 0 | ||||||||
| Genistein | mg | 0 | ||||||||
| Glycitein | mg | 0 | ||||||||
| Total isoflavones | mg | 0 | ||||||||
| Formononetin | mg | 0 | ||||||||
| Coumestrol | mg | 0 | ||||||||
| Proanthocyanidin | ||||||||||
| Proanthocyanidin dimers | mg | 13.2 | ||||||||
| Proanthocyanidin trimers | mg | 8 | ||||||||
| Proanthocyanidin 4-6mers | mg | 24.7 | ||||||||
| Proanthocyanidin 7-10mers | mg | 19.2 | ||||||||
| Proanthocyanidin polymers (>10mers) | mg | 28.8 | ||||||||
Table 2. Polyphenolic concentration of whole apples (freeze dried) and apple juice
| Polyphenolic compound | Whole apple (67 cultivars; n = 5 each), mg/kg dry weight | Fresh juice (combined dessert and cider apples), mg/L | Commercial juice (combined clear and cloudy juices), mg/L |
| Hydroxycinnamic acids | 50–3000 | 57–593 | 69–259 |
| 5-Caffeoylquinic acid (chlorogenic acid) | 15–2960 | ||
| p-Coumaroylquinic acid | 4–260 | ||
| Flavan-3-ols/procyanidins | 4622–25,480 | 50–393 | 14–124 |
| Procyanidins B2 | 69–2000 | ||
| Procyanidins C1 | 58–970 | ||
| (-) Epicatechin | 69–2760 | ||
| (+) Epicatechin | 10–720 | ||
| Oligomeric procyanidins | 1374–19,850 | n.d. | n.d. |
| Flavonols | 80–1660 | 0.4–27 | 4–14 |
| Dihydrochalcones | 49–434 | 10–171 | 9–87 |
| Anthocyanins (red peel) | 10–551 | n.d. | n.d. |
| Total polyphenols | 5230–27,240 | 154–970 | 110–459 |

Apple fruit benefits
Apples are a rich source of dietary phytochemicals such as flavonoids. They also contain high levels of polyphenols and other phytochemicals 13. Polyphenols in apples and their extracts (juices) have been studied in several human studies that have shown promising results related to their beneficial effects 14. For example, consumption of at least one apple a day was reported to reduce the risk of colorectal cancer 15. The study also predicted that the risk of colorectal cancer reduced by approximately 50% upon consumption of more than one apple a day. In many laboratory test tubes and animals studies on the anticancer effects of apple extracts have been evaluated, including those of phytochemical compounds in these extracts 16, 17 and apple juice fractions 18.
In previous studies, it was demonstrated that phloretin isolated from apple peels exhibits significant antihepatic tumor proliferation capacity through in animal study via the inhibition of type 2 glucose transporter (GLUT2) 17. A study further demonstrated that phloretin significantly potentiates paclitaxel-induced DNA laddering effects in a human liver cancer cell model 16. This observation indicated that phytochemical components in apples exhibit a beneficial effect on human health.
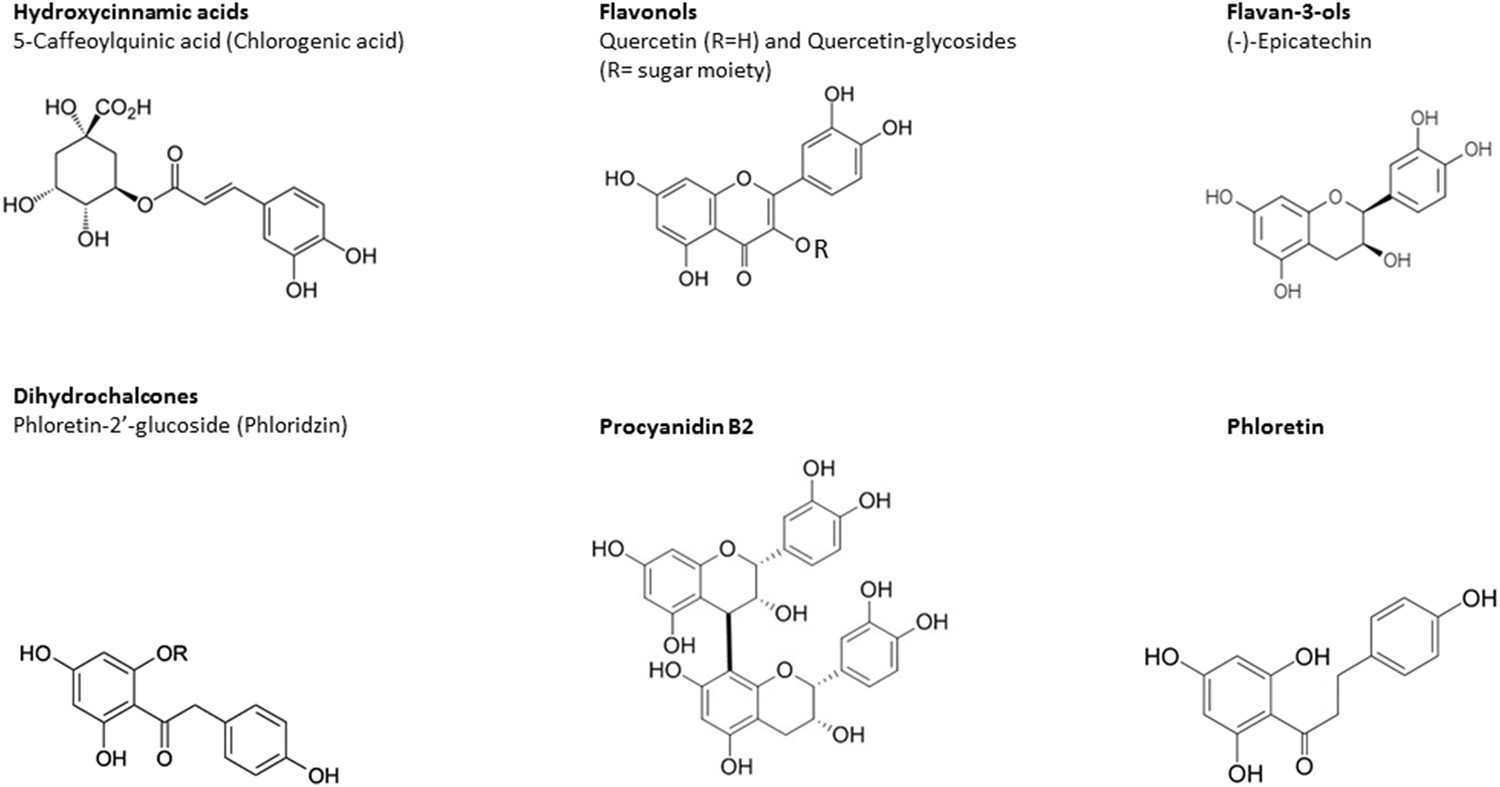
For cancer chemoprevention, dietary nutrients should be more readily available. Many studies have demonstrated the chemopreventive effects of dietary polyphenols, especially the most abundant subclasses, including flavonoids (60% of all polyphenols) and phenolic acids (30% of total polyphenols) 19. Flavonoids are divided into various groups based on their molecular structure, several of which are present in significant quantities in apple, including flavanols, flavonols, and anthocyanidins as well as dihydrochalcones and hydroxycinnamic acids 20, 21. The chemical structures of several representative polyphenols present in apple are shown in Figure 1 13.
Figure 1. Chemical structures of some selected typical bioactive compounds in apple juice
[Source 22]Apple and cardiovascular disease prevention
It is estimated that over 80 million American adults (1 in 3 and particularly adults older than 60 years) have one or more types of cardiovascular disease. These include the diet-related conditions of hypertension, coronary artery disease, myocardial infarction, angina, heart failure, and stroke. Boyer and Liu 23, early reports showed an inverse association between apple and apple-flavonoid intake and coronary mortality. A group of Finnish women consuming >71 g of apple/day experienced a 43% reduction in coronary mortality compared to women who did not eat apples 24. In men, the risk reduction was 19% in the group consuming >54 g compared to no apple intake 24. These findings were consistent with prior data showing reduced coronary mortality in elderly Dutch men (65–84 years) who consumed apples (average 69 g/day) compared to men who had little or no apple intake 25. Collectively, these studies indicate that a relatively modest intake of apple is associated with reduced risk of cardiovascular disease and associated mortality.
A clinical trial evaluated the cardiovascular protective effects of consumption of 75 g (about two medium-sized apples) of dried apple for 1 year in 146 postmenopausal women 26. The study showed that dried apple significantly lowered serum levels of total cholesterol and LDL “bad” cholesterol by 9% and 16%, respectively, at 3 months and further decreased by 13% and 24%, respectively, at 6 months, but stayed constant thereafter 26. Furthermore, consumption of dried apple also reduced lipid hydroperoxide and C-reactive protein (a blood test marker for inflammation in the body) 26. In addition, a study 27 compared the cholesterol-lowering effect of 5 different apple species, Red Delicious, Granny Smith, Fuji, Golden Delicious and Annurca apple, in mildly hypercholesterolaemic healthy subjects. The study 27 detected that Annurca apples led to the most significant outcome, reduced total cholesterol and LDL “bad” cholesterol levels by 8.3% and 14.5%, respectively, and an increased HDL “good” cholesterol level by 15.2% 27. Moreover, another study 28 compared the effects of whole fresh apple and processed apple products (apple pomace, cloudy apple juice, or clear apple juice) on lipid profiles in healthy volunteers. The result showed that whole apple, pomace, and cloudy juice lowered serum total cholesterol and LDL “bad” cholesterol; however, clear apple juice increased total cholesterol and LDL “bad” cholesterol slightly, from which it could be concluded that the fiber component was necessary for the lipid-lowering effect of apple in healthy humans. Additionally, the acute effects of apple on improving endothelial function were studied in some trials, showing that apple improved endothelial function by affecting nitric oxide metabolites 29, 30.
Antioxidant activity of apple polyphenols
Generation of oxygen radicals causes chronic diseases such as diabetes mellitus 31, retinal degeneration, neurodegenerative disorders, aging, and cancer. Several studies have demonstrated that apple polyphenols, including phloretin, exhibit promising antioxidants effects by playing a role in significant mechanisms responsible for the prevention of illnesses triggered by oxidative stress 32. For example, in a previous study on Wistar rats, diabetes was induced by a single dose of streptozotocin. Rats in the diabetic group received either apple juice (15 mL/kg) or apple peel extract (1 g/kg) for 21 days. At the end of the study, lipid profile parameters were measured in serum samples and lipid peroxidation level, antioxidant enzyme activities, and level of inflammatory markers were evaluated in pancreatic tissue samples. The study concluded that supplementation with apple juice/extract may have protective effects against deleterious complications of diabetes mellitus due to its antioxidant effects 31. In a different study on human participants, after 2 weeks of dietary intervention in 25 healthy individuals, the influence of apple and grape juices consumption on body antioxidant status was investigated. The results indicated that such a dietary consumption increased their plasma total antioxidant capacity and decreased their serum and plasma concentration of malondialdehyde 33.
Anticancer activity of apple polyphenols
In addition to its antioxidant activity, studies 34, 35, 36 have demonstrated that apple polyphenols have significant effects in affecting signaling pathways that control cell survival, growth, and proliferation both in vitro and in vivo. The results have shown that phloretin inhibited proliferation and induced apoptosis in nonsmall cell lung cancer cells (A549, Calu-1, H838, and H520) in a dose-dependent manner; phloretin also suppressed the invasion and migration of these cells. In one study, it was found that phloretin (50–150μM) significantly potentiates paclitaxel (10nM)-induced DNA laddering formation in human hepatoma (Hep G2) cells. The antitumor therapeutic efficacy of phloretin (10 mg/kg body weight) was determined by combined treatment of cells with antitumor drug (paclitaxel, 1 mg/kg body weight) in an SCID mouse model 16. Recently it was also further demonstrated that apple phloretin inhibits human colorectal cancer and liver cancer cells through inhibition of GLUT2 (Figure 2). These results further provide evidence to the hypothesis that glucose deprivation therapy has some important beneficial effects on human cancer therapy 37. To test this hypothesis, another group evaluated the antiproliferative activity of apple juices in vitro in MCF-7 and MDA-MB-231 human breast cancer cells. The study results showed that Pelingo apple juice has promising effects to inhibit breast cancer cell proliferation 38. It was demonstrated that 3-beta-trans-cinnamoyloxy-2alpha-hydroxy-urs-12-en-28-oic acid, which is one of the main components of apple peels, showed potent in vitro and in vivo antitumor activity against mammary tumor in a nude mouse xenograft model at a dose of 50 mg/kg/d without body weight loss and mortality 39.
As reviewed by Boyer and Liu 23, there were several reports prior to 2004 demonstrating that apple intake was associated with reduced risk of cancer, particularly lung cancer in cohort and case-control studies. Recent human trials have added data that suggest a protective effect of apple on risk of cancer. A hospital-based, case-control study published in 2005 included over 6000 participants from various regions in Italy and examined the association between fresh apple intake and risk of cancer 40. Data were based on food frequency questionnaire (FFQ) including interviews of dietary intake in the 2 year prior to diagnosis. It was found that consuming one or more medium-sized apples (166 g)/day was associated with a reduction in risk of cancer compared to consumption of <1 apple/day 40. Significant risk reduction was observed in cancer at several sites, including oral cavity and pharynx (18% percent reduction), esophagus (22% percent reduction), colorectal (30% percent reduction), larynx (41% percent reduction), breast (24% percent reduction), ovary (24% percent reduction), and prostate (7% percent reduction). A similar pattern of beneficial association was present when the groups were stratified by age, energy intake, vegetable consumption, smoking, and BMI.
An updated analysis of fruit and vegetable consumption and lung cancer risk from the European Prospective Investigation into Cancer and Nutrition was published in 2007 41. The data were based on dietary assessment questionnaires from a prospective cohort of 478,590 male and female participants in 23 centers from 10 European countries. Total daily intake (g/d) of “hard fruits” (apples and pears) was included in the analysis with a median follow-up time of 6.4 years. A comparison of the lowest quintile of intake (average 43 g/day) compared to the highest 2 quintiles (93.5 and 164.9 g/day, respectively) showed a significant inverse association between combined apple and pear intake and lung cancer in all participants and current smokers 41. There is no detailed breakdown of the relative proportion of apples compared to pears, but the authors noted that in general hard fruits were consumed in greater quantities than all other subgroups of fruit.
In addition to clinical data, recent animal feeding and in vitro (test tube) studies have aimed to examine the potential protective link between apple and cancer. Colorectal or colon cancer, the 4th most common cancer and the 3rd most common cause of death in Western society 42, has been the focus of many investigations. There have also been several studies that examined the potential of apple to reduce breast cancer risk. As summarized below, recent studies demonstrate the potential of apple to mitigate many metabolic processes associated with the etiology of cancer at various stages. The data are presented in the context of colon cancer, followed by breast cancer and general studies.
Barth et al. 43 used a well-established rat model of chemically induced colonic damage (using 1,2-dimethylhydrazine) to examine alterations associated with colon cancer and to test the effects of apple. Rats were fed 2 different preparations of apple juice pressed from a mixture of apples to determine if 7 week of daily consumption protected the mucosa from 1,2-dimethylhydrazine–induced genotoxic damage. Because early lesions in the colon may progress to malignancy, the presence of premalignant hyperproliferative crypts and aberrant crypts are indicators of the potential pathogenesis of cancer in this model. Apple juice preparations, including a “cloudy” (higher procyanidin and pectin) and a clear juice preparation, reduced important markers, including DNA damage and hyperproliferation and lowered the number of large aberrant crypt foci in the distal colon 43. In follow-up studies, the same authors examined the effect of isolated fractions on the above markers and concluded that the juice fraction itself was more effective than individual components of juice, including polyphenolic-rich extracts 44. The findings of these investigators and many others suggest that the whole is greater than the sum of the parts in terms of a protective effect of apple on cancer.
A different study, using rats injected with the chemical carcinogen azoxymethane, known to cause a range of morphological changes including carcinoma, found protective effects of an extract of apple procyanidins provided to the animals in their drinking water for 6 week 45. There was a significant reduction of preneoplastic lesions in the animals exposed to the apple phytochemicals, including 50% fewer aberrant crypts. The authors estimated that the amount of procyanidin ingested would be comparable to humans consuming 2 apples/d (4–10 mg procyanidin/kg bodyweight) 45.
Animal feeding and in vitro studies have shown the potential of apple in breast cancer reduction. Liu et al. 46 treated rats with a carcinogenic agent (7,12-dimethylbenz[a]anthracene) to induce mammary tumors and then fed extracts of whole apples by gavage to the animals. Daily administration of the apple extract (~272 mg of phenolics/100 g of apples) for 24 week resulted in a significant dose-dependent reduction in the number and onset of mammary tumors compared to control rats 46. Increasingly protective effects were observed in rats fed extract doses equivalent to 1, 3, or 6 apples/day with reductions in tumor incidence of 17, 39, and 44%, respectively. After 24 weeks, cumulative tumor numbers in groups receiving low, medium, or high doses of the extract were reduced by 25, 25, and 61%, respectively, and there was a dose-dependent delay in tumor onset 46.
Follow-up work with this model to further characterize the effects of the fresh apple extract demonstrated dose-dependent inhibition of markers of cell proliferation (proliferating cell nuclear antigen) and down regulation of the cell cycle in mammary tumor cells (cyclin D1 protein expression) 47. There was a concurrent and dose-dependent increase in expression of Bax, a proapoptotic protein and downward expression of Bcl-2, an antiapoptotic protein. Histological analysis showed that consumption of the apple extract also reduced the proportion of highly malignant adenocarcinoma in a dose-dependent manner from 81.3% in the control group to ~57, 50, and 23% in the low, middle, and high doses of apple extracts, respectively, over 24 weeks 47.
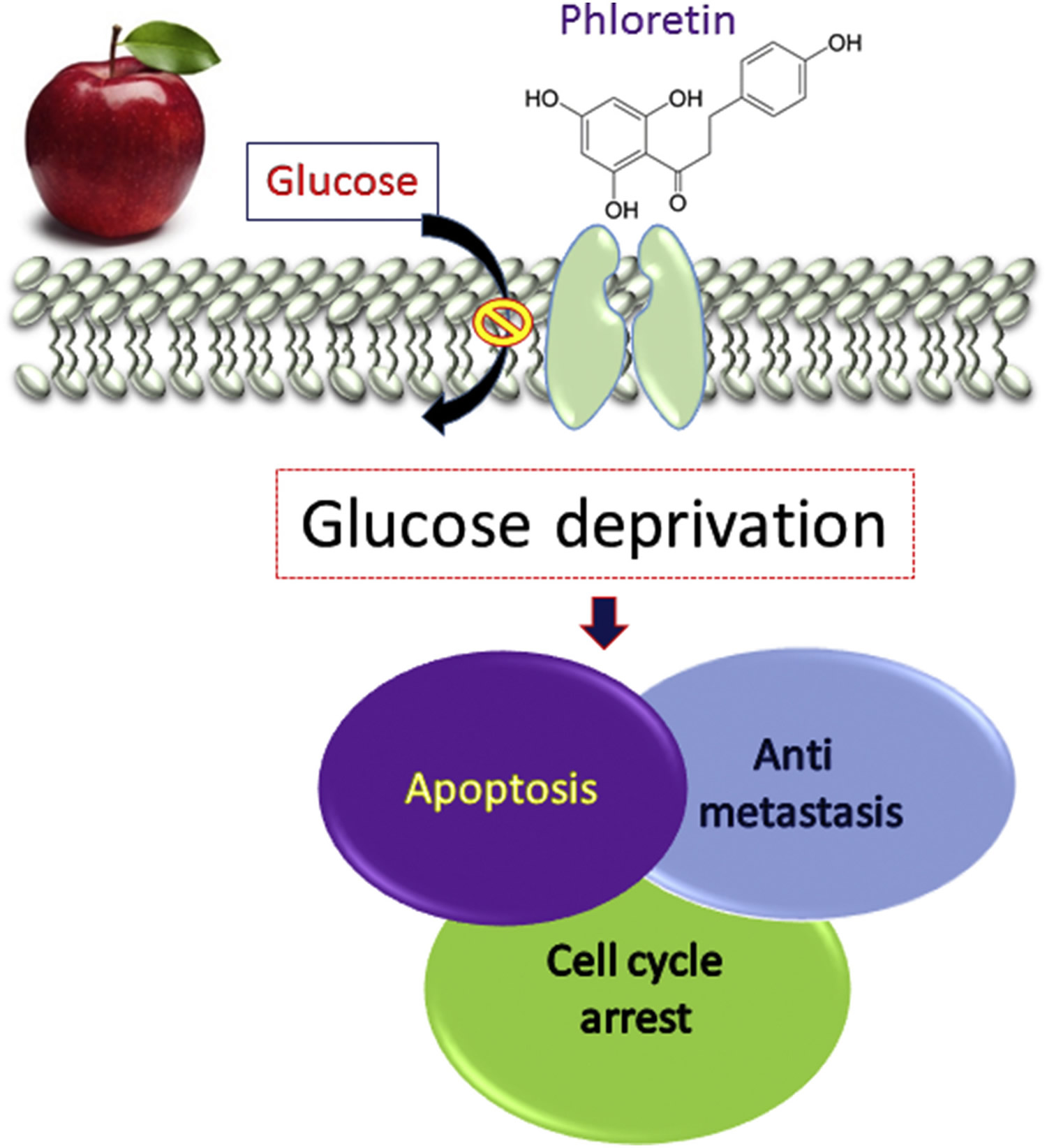
Figure 2. Apple polyphenol phloretin inhibits growth of cancer cells through inhibition of type 2 glucose transporter
Apple polyphenols inhibit cell migration and invasion
Antimetastasis effects of biocompounds in apple have been studied by our group. Our results have indicated that phloretin is an inhibitor of GLUT2 17 and that targeting GLUT2 significantly inhibited COLO 205 colon cancer cell proliferation, migration, and invasion in vitro and in vivo 48. In this study 48, p53-mediated signals were important. Inhibition of the wild-type p53 by dominant negative p53 will attenuate the phloretin-induced colon cancer migration and its related signals. In colorectal cancers, studies have demonstrated that the activation of nuclear factor-κB (NF-κB) occurs via lipopolysaccharide (LPS) binding to the Toll-like receptor 4 (TLR4). Modification of polysaccharide components in apple altered the LPS/TLR4/NF-κB pathway; consequently, supplementation of apple polysaccharide significantly inhibited the migratory ability in vitro on the LPS/TLR4/NF-κB pathway in colorectal cancer cells (HT-29 and SW620 cells) 49. In a study on liver cancer cells, the effect of apple polyphenol extract on the proliferation and invasion of rat ascites hepatoma cell line (AH109A) was examined in vitro. The apple polyphenol extract suppressed both proliferation and invasion of the hepatoma cell line in a dose-dependent manner up to 200 μg/mL. In an in vivo study, apple polyphenol also reduced the growth and metastasis of solid hepatomas and significantly suppressed the serum lipid peroxide level in rats transplanted with AH109A 50.
Apple polyphenols induced apoptotic cancer cell death
Previous results demonstrated that apple polyphenol phloretin (50–150μM) significantly potentiates paclitaxel (10nM)-induced DNA laddering formation in Hep G2 cells. It was also demonstrated that the caspases 3, 8, and 9 were involved in apoptosis, as evidenced by activity assays 16. Previous studies in this area have also demonstrated that phloretin inhibited leukemia cell growth 51 and induced apoptosis of melanoma cells through deprivation of glucose uptake by inhibition of glucose transmembrane transport 52. Using 18F-fluorodeoxyglucose micropositron emission tomography the effects of phloretin-induced suppression of liver tumor growth were demonstrated to involve regulation of glucose transportation. The 18F-fluorodeoxyglucose uptake in the phloretin-treated Hep G2 tumor-bearing mice was significantly suppressed as compared with the control mice. Effects of phloretin on glioblastoma cancer cells have been investigated via induction of apoptosis and cells’ growth cycle arrest. The identified mechanisms demonstrated increased expression of p27 and decreased expression of cdk2, cdk4, cdk6, cyclin D, and cyclin E. Moreover, the phosphatidylinositol-3-kinase/Akt and the mammalian target of rapamycin (PI3K/Akt/mTOR) signaling cascades were suppressed by phloretin in a dose-dependent manner 53. Phloretin-based combination treatment enhanced the anticancer effects of cisplatin on nonsmall cell lung cancer cell lines by suppressing the expression of Bcl-2, increasing the protein expression of cleaved caspases 3 and 9, and deregulating the expression of matrix metalloproteinase-2 and metalloproteinase-9 on gene and protein levels 34. The results suggest that inhibition of intracellular glucose uptake was the most important mechanism responsible for the cancer cell killing effects. Because many cancer cells rely on aerobic glycolysis for energy production, Xintaropoulou et al 35 targeted this pathway as a potential strategy to inhibit cancer cell growth. In that study, inhibition of five glycolysis pathway molecules (GLUT1, HKII (hexokinase II), PFKFB3 (6-phosphofructo-2-kinase/fructose-2,6-bisphosphatase 3), PDHK1 (pyruvate dehydrogenase kinase I), and LDH (lactate dehydrogenase)) using nine inhibitors (phloretin, quercetin, STF31 (Glut1 inhibitor), WZB117 (Glut1 inhibitor), 3PO (3-(3-pyridinyl)-1-(4-pyridinyl)-2-propen-1-one, glycolytic inhibitor), 3-bromopyruvate, dichloroacetate, oxamic acid, NHI-1 (lactate dehydrogenase A inhibitor)) was investigated in panels of breast and ovarian cancer cell line models. Their results indicated that growth of breast and ovarian cancer cell lines was more sensitive to the glycolytic pathway, with increased sensitivity to the inhibitors under normoxic conditions 35.
Signaling molecules and disease protection effects of phloretin
As described earlier, apple polyphenols induced anticancer activity mainly through their antioxidant activity. Such results have been confirmed by basic in vitro studies. Moreover, phloretin-induced cell cycle arrest was associated with increased expression of p27 and decreased expression of cdk2, cdk4, cdk6, cyclin D, and cyclin E 53. Inhibition of intracellular signaling pathways as well as the PI3K/Akt/mTOR and ERK/Nrf2 signaling cascades was suppressed by phloretin in a dose-dependent manner 54. In addition, many previous studies have also proposed that phloretin triggered the mitochondrial apoptosis pathway 55, 56 and generated reactive oxygen species (ROS) 57. Most of these studies were accompanied by induction of cell growth arrest and apoptosis through upregulation of proapoptotic molecules such as Bax, Bak, and poly (ADP-ribose (adenosine diphosphate-ribose)) polymerase (cleaved) and downregulation of Bcl-2. The antioxidant agents N-acetyl-l-cysteine and glutathione weakened the effect of phloretin on glioblastoma cells. In conclusion, these results demonstrate that phloretin exerts a potent chemopreventive activity in human glioblastoma cells through the generation of ROS. Such effects may have some potential applications for clinical patients. For example, in acute hepatitis patients, liver damage is induced by several damaging factors, among which viral exposure, alcohol consumption, and drug and immune system issues are most popular 58. In addition to antioxidant effects, phloretin is also able to modulate inflammatory responses. A previous study demonstrated that phloretin suppressed the activation and function of mouse dendritic cells 59. The study results showed that phloretin disturbed the multiple intracellular signaling pathways in dendritic cells induced by the TLR4 agonist LPS, including ROS, mitogen-activated protein kinases (extracellular signal-regulated kinase, c-Jun N-terminal kinase, p38 mitogen-activated protein kinase), and NF-κB, thus reducing the production of inflammatory cytokines and chemokine 59.
References- Jules Janick; James N. Cummins; Susan K. Brown; Minou Hemmat (1996). “Chapter 1: Apples”. In Jules Janick; James N. Moore. Fruit Breeding, Volume I: Tree and Tropical Fruits. https://hort.purdue.edu/newcrop/pri/chapter.pdf
- http://postharvest.tfrec.wsu.edu/pages/N2I2A
- The chemopreventive activity of apple against carcinogenesis: antioxidant activity and cell cycle control. Eur J Cancer Prev. 2014 Sep;23(5):477-80. doi: 10.1097/CEJ.0000000000000005. https://www.ncbi.nlm.nih.gov/pubmed/24366437
- Tsao R, Yang R, Xie S, Sockovie E, Khanizadeh S. Which polyphenolic compounds contribute to the total antioxidant activities of apple? J Agric Food Chem. 2005 Jun 15;53(12):4989-95. doi: 10.1021/jf048289h
- Hyson D. A. (2011). A comprehensive review of apples and apple components and their relationship to human health. Advances in nutrition (Bethesda, Md.), 2(5), 408–420. https://doi.org/10.3945/an.111.000513
- Determination of Polyphenolic Profiles of Basque Cider Apple Varieties Using Accelerated Solvent Extraction. R. M. Alonso-Salces, E. Korta, A. Barranco, L. A. Berrueta, B. Gallo and F. Vicente, J. Agric. Food Chem., 2001, volume 49, pages 3761–3767.
- Phloridzin: biosynthesis, distribution and physiological relevance in plants. Phytochemistry. 2010 Jun;71(8-9):838-43. doi: 10.1016/j.phytochem.2010.03.003. Epub 2010 Mar 29. https://www.ncbi.nlm.nih.gov/pubmed/20356611
- Chun OK, Chung SJ, Song WO. Estimated dietary flavonoid intake and major food sources of U.S. adults. J Nutr. 2007 May;137(5):1244-52. doi: 10.1093/jn/137.5.1244
- Chun OK, Kim D-O, Smith N, Schroeder D, Han J, Lee C. Daily consumption of phenolics and total antioxidant capacity from fruits and vegetables in the American diet. J Sci Food Agric. 2005;85:1715–24.
- United States Department of Agriculture Agricultural Research Service. National Nutrient Database for Standard Reference Release 28. https://ndb.nal.usda.gov/ndb/search/list
- Gerhauser C. Cancer chemopreventive potential of apples, apple juice, and apple components. Planta Med. 2008 Oct;74(13):1608-24. doi: 10.1055/s-0028-1088300
- Wojdyło A, Oszmiański J, Laskowski P. Polyphenolic compounds and antioxidant activity of new and old apple varieties. J Agric Food Chem. 2008 Aug 13;56(15):6520-30. doi: 10.1021/jf800510j
- Hyson DA. A comprehensive review of apples and apple components and their relationship to human health. Adv Nutr 2011;2:408e20.
- Jedrychowski W, Maugeri U, Popiela T, Kulig J, Sochacka-Tatara E, Pac A, Sowa A, Musial A. Case-control study on beneficial effect of regular consumption of apples on colorectal cancer risk in a population with relatively low intake of fruits and vegetables. Eur J Cancer Prev 2010;19:42e7.
- Jaganathan SK, Vellayappan MV, Narasimhan G, Supriyanto E, Dewi DEO, Narayanan ALT, et al. Chemopreventive effect of apple and berry fruits against colon cancer. World J Gastroenterol 2014;20(45):17029e36.
- Yang KC, Tsai CY, Wang YJ, Wei PL, Lee CH, Chen JH, Wu CH, Ho YS. Apple polyphenol phloretin potentiates the anticancer actions of paclitaxel through induction of apoptosis in human hep G2 cells. Mol Carcinog 2009;48:420e31.
- Wu CH, Ho YS, Tsai CY, Wang YJ, Tseng H, Wei PL, Lee CH, Liu RS, Lin SY. In vitro and in vivo study of phloretin-induced apoptosis in human liver cancer cells involving inhibition of type II glucose transporter. Int J Cancer 2009;124:2210e9.
- Sudan S, Rupasinghe HP. Flavonoid-enriched apple fraction AF4 induces cell cycle arrest, DNA topoisomerase II inhibition, and apoptosis in human liver cancer HepG2 cells. Nutr Cancer 2014;66:1237e46.
- Ramos S. Effects of dietary flavonoids on apoptotic pathways related to cancer chemoprevention. J Nutr Biochem 2007;18:427e42.
- Bouayed J, Hoffmann L, Bohn T. Total phenolics, flavonoids, anthocyanins and antioxidant activity following simulated gastro-intestinal digestion and dialysis of apple varieties: bioaccessibility and potential uptake. Food Chem 2011;128:14e21.
- van der Sluis AA, Dekker M, Verkerk R, Jongen WM. An improved, rapid in vitro method to measure antioxidant activity. Application on selected flavonoids and apple juice. J Agric Food Chem 2000;48:4116e22.
- An apple a day to prevent cancer formation: Reducing cancer risk with flavonoids. Tu, Shih-Hsin et al. Journal of Food and Drug Analysis , Volume 25, Issue 1, 119-124.
- Boyer, J., & Liu, R. H. (2004). Apple phytochemicals and their health benefits. Nutrition journal, 3, 5. https://doi.org/10.1186/1475-2891-3-5
- Knekt P, Jarvinen R, Reunanen A, Maatela J. Flavonoid intake and coronary mortality in Finland: a cohort study. BMJ. 1996 Feb 24;312(7029):478-81. doi: 10.1136/bmj.312.7029.478
- Hertog MG, Feskens EJ, Hollman PC, Katan MB, Kromhout D. Dietary antioxidant flavonoids and risk of coronary heart disease: the Zutphen Elderly Study. Lancet. 1993 Oct 23;342(8878):1007-11. doi: 10.1016/0140-6736(93)92876-u
- Daily apple versus dried plum: impact on cardiovascular disease risk factors in postmenopausal women. Chai SC, Hooshmand S, Saadat RL, Payton ME, Brummel-Smith K, Arjmandi BH. J Acad Nutr Diet. 2012 Aug; 112(8):1158-68. https://jandonline.org/article/S2212-2672(12)00625-9/fulltext
- Annurca (Malus pumila Miller cv. Annurca) apple as a functional food for the contribution to a healthy balance of plasma cholesterol levels: results of a randomized clinical trial. Tenore GC, Caruso D, Buonomo G, D’Urso E, D’Avino M, Campiglia P, Marinelli L, Novellino E. J Sci Food Agric. 2017 May; 97(7):2107-2115. https://onlinelibrary.wiley.com/doi/pdf/10.1002/jsfa.8016
- Intake of whole apples or clear apple juice has contrasting effects on plasma lipids in healthy volunteers. Ravn-Haren G, Dragsted LO, Buch-Andersen T, Jensen EN, Jensen RI, Németh-Balogh M, Paulovicsová B, Bergström A, Wilcks A, Licht TR, Markowski J, Bügel S. Eur J Nutr. 2013 Dec; 52(8):1875-89.
- Flavonoid-rich apples and nitrate-rich spinach augment nitric oxide status and improve endothelial function in healthy men and women: a randomized controlled trial. Bondonno CP, Yang X, Croft KD, Considine MJ, Ward NC, Rich L, Puddey IB, Swinny E, Mubarak A, Hodgson JM. Free Radic Biol Med. 2012 Jan 1; 52(1):95-102.
- Bioavailability of epicatechin and effects on nitric oxide metabolites of an apple flavanol-rich extract supplemented beverage compared to a whole apple puree: a randomized, placebo-controlled, crossover trial. Hollands WJ, Hart DJ, Dainty JR, Hasselwander O, Tiihonen K, Wood R, Kroon PA. Mol Nutr Food Res. 2013 Jul; 57(7):1209-17.
- Fathy SM, Drees EA. Protective effects of Egyptian cloudy apple juice and apple peel extract on lipid peroxidation, antioxidant enzymes and inflammatory status in diabetic rat pancreas. BMC Complement Altern Med 2016;16:8.
- Kim ES, Hong WK. An apple a day… does it really keep the doctor away? The current state of cancer chemoprevention. J Natl Cancer Inst 2005;97:468e70.
- Yuan L, Meng L, Ma W, Xiao Z, Zhu X, Feng JF, Yu H, Xiao R. Impact of apple and grape juice consumption on the antioxidant status in healthy subjects. Int J Food Sci Nutr 2011;62:844e50.
- Ma L, Wang R, Nan Y, Li W, Wang Q, Jin F. Phloretin exhibits an anticancer effect and enhances the anticancer ability of cisplatin on non-small cell lung cancer cell lines by regulating expression of apoptotic pathways and matrix metalloproteinases. Int J Oncol 2016;48:843e53.
- Xintaropoulou C, Ward C, Wise A, Marston H, Turnbull A, Langdon SP. A comparative analysis of inhibitors of the glycolysis pathway in breast and ovarian cancer cell line models. Oncotarget 2015;6:25677e95.
- Park SY, Kim EJ, Shin HK, Kwon DY, Kim MS, Surh YJ, Park JH. Induction of apoptosis in HT-29 colon cancer cells by phloretin. J Med Food 2007;10:581e6.
- Hong SM, Park CW, Kim SW, Nam YJ, Yu JH, Shin JH, Yun CH, Im SH, Kim KT, Sung YC, Choi KY. NAMPT suppresses glucose deprivation-induced oxidative stress by increasing NADPH levels in breast cancer. Oncogene 2016;35:3544e54.
- Schiavano GF, De Santi M, Brandi G, Fanelli M, Bucchini A, Giamperi L, Giomaro G. Inhibition of breast cancer cell proliferation and in vitro tumorigenesis by a new red apple cultivar. PLoS One 2015;10:e0135840.
- Qiao A, Wang Y, Xiang L, Wang C, He X. A novel triterpenoid isolated from apple functions as an anti-mammary tumor agent via a mitochondrial and caspase-independent apoptosis pathway. J Agric Food Chem 2015;63:185e91.
- Gallus S, Talamini R, Giacosa A, Montella M, Ramazzotti V, Franceschi S, Negri E, La Vecchia C. Does an apple a day keep the oncologist away? Ann Oncol. 2005 Nov;16(11):1841-4. doi: 10.1093/annonc/mdi361
- Linseisen J, Rohrmann S, Miller AB, Bueno-de-Mesquita HB, Büchner FL, Vineis P, Agudo A, Gram IT, Janson L, Krogh V, Overvad K, Rasmuson T, Schulz M, Pischon T, Kaaks R, Nieters A, Allen NE, Key TJ, Bingham S, Khaw KT, Amiano P, Barricarte A, Martinez C, Navarro C, Quirós R, Clavel-Chapelon F, Boutron-Ruault MC, Touvier M, Peeters PH, Berglund G, Hallmans G, Lund E, Palli D, Panico S, Tumino R, Tjønneland A, Olsen A, Trichopoulou A, Trichopoulos D, Autier P, Boffetta P, Slimani N, Riboli E. Fruit and vegetable consumption and lung cancer risk: updated information from the European Prospective Investigation into Cancer and Nutrition (EPIC). Int J Cancer. 2007 Sep 1;121(5):1103-14. doi: 10.1002/ijc.22807
- Jemal A, Siegel R, Ward E, Murray T, Xu J, Thun MJ. Cancer statistics, 2007. CA Cancer J Clin. 2007 Jan-Feb;57(1):43-66. doi: 10.3322/canjclin.57.1.43
- Barth SW, Fähndrich C, Bub A, Dietrich H, Watzl B, Will F, Briviba K, Rechkemmer G. Cloudy apple juice decreases DNA damage, hyperproliferation and aberrant crypt foci development in the distal colon of DMH-initiated rats. Carcinogenesis. 2005 Aug;26(8):1414-21. doi: 10.1093/carcin/bgi082
- Barth SW, Faehndrich C, Bub A, Watzl B, Will F, Dietrich H, Rechkemmer G, Briviba K. Cloudy apple juice is more effective than apple polyphenols and an apple juice derived cloud fraction in a rat model of colon carcinogenesis. J Agric Food Chem. 2007 Feb 21;55(4):1181-7. doi: 10.1021/jf063078t
- Gossé F, Guyot S, Roussi S, Lobstein A, Fischer B, Seiler N, Raul F. Chemopreventive properties of apple procyanidins on human colon cancer-derived metastatic SW620 cells and in a rat model of colon carcinogenesis. Carcinogenesis. 2005 Jul;26(7):1291-5. doi: 10.1093/carcin/bgi074
- Liu RH, Liu J, Chen B. Apples prevent mammary tumors in rats. J Agric Food Chem. 2005 Mar 23;53(6):2341-3. doi: 10.1021/jf058010c
- Liu JR, Dong HW, Chen BQ, Zhao P, Liu RH. Fresh apples suppress mammary carcinogenesis and proliferative activity and induce apoptosis in mammary tumors of the Sprague-Dawley rat. J Agric Food Chem. 2009 Jan 14;57(1):297-304. doi: 10.1021/jf801826w
- Lin ST, Tu SH, Yang PS, Hsu SP, Lee WH, Ho CT, Wu CH, Lai YH, Chen MY, Chen LC. Apple polyphenol phloretin inhibits colorectal cancer cell growth via inhibition of the type 2 glucose transporter and activation of p53-mediated signaling. J Agric Food Chem 2016;64:6826e37.
- Zhang D, Li YH, Mi M, Jiang FL, Yue ZG, Sun Y, Fan L, Meng J, Zhang X, Liu L, Mei QB. Modified apple polysaccharides suppress the migration and invasion of colorectal cancer cells induced by lipopolysaccharide. Nutr Res 2013;33:839e48.
- Miura D, Miura Y, Yagasaki K. Effect of apple polyphenol extract on hepatoma proliferation and invasion in culture and on tumor growth, metastasis, and abnormal lipoprotein profiles in hepatoma-bearing rats. Biosci Biotechnol Biochem 2007;71:2743e50.
- Devi MA, Das NP. In vitro effects of natural plant polyphenols on the proliferation of normal and abnormal human lymphocytes and their secretions of interleukin-2. Cancer Lett 1993;69:191e6.
- Kobori M, Shinmoto H, Tsushida T, Shinohara K. Phloretininduced apoptosis in B16melanoma 4A5 cells by inhibition of glucosetransmembranetransport.Cancer Lett1997;119:207e12.
- Liu Y, Fan C, Pu L, Wei C, Jin H, Teng Y, Zhao M, Yu AC, Jiang F, Shu J, Li F, Peng Q, Kong J, Pan B, Zheng L, Huang Y. Phloretin induces cell cycle arrest and apoptosis of human glioblastoma cells through the generation of reactive oxygen species. J Neurooncol 2016;128:217e23.
- Yang YC, Lii CK, Lin AH, Yeh YW, Yao HT, Li CC, Liu KL, Chen HW. Induction of glutathione synthesis and heme oxygenase 1 by the flavonoids butein and phloretin is mediated through the ERK/Nrf2 pathway and protects against oxidative stress. Free Radic Biol Med 2011;51:2073e81.
- Vineetha VP, Soumya RS, Raghu KG. Phloretin ameliorates arsenic trioxide induced mitochondrial dysfunction in H9c2 cardiomyoblasts mediated via alterations in membrane permeability and ETC complexes. Eur J Pharmacol 2015;754:162e72.
- Ullen A, Fauler G, Bernhart E, Nusshold C, Reicher H, Leis HJ, Malle E, Sattler W. Phloretin ameliorates 2-chlorohexadecanal-mediated brain microvascular endothelial cell dysfunction in vitro. Free Radic Biol Med 2012;53:1770e81.
- Andrisse S, Koehler RM, Chen JE, Patel GD, Vallurupalli VR, Ratliff BA, Warren DE, Fisher JS. Role of GLUT1 in regulation of reactive oxygen species. Redox Biol 2014;2:764e71.
- Zuo AR, Yu YY, Shu QL, Zheng LX, Wang XM, Peng SH, Xie YF, Cao SW. Hepatoprotective effects and antioxidant, antityrosinase activities of phloretin and phloretin isonicotinyl hydrazone. J Chin Med Assoc 2014;77:290e301.
- Lin CC, Chu CL, Ng CS, Lin CY, Chen DY, Pan IH, Huang KJ. Immunomodulation of phloretin by impairing dendritic cell activation and function. Food Funct 2014;5:997e1006.