What is an artificial heart
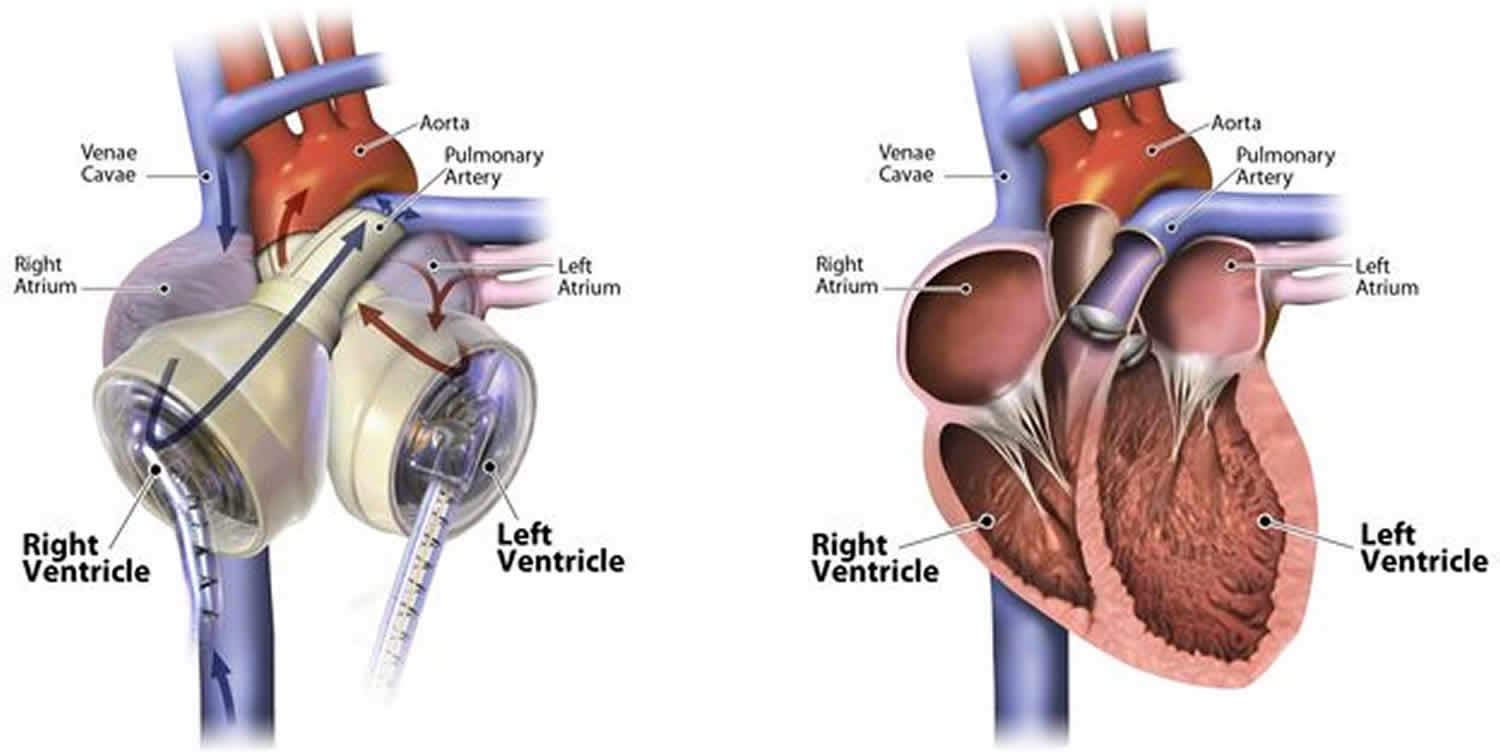
Total artificial heart replaces the function of both ventricles in people with end-stage heart failure. The total artificial heart is approved for use in patients with end-stage heart failure affecting both sides of the heart (biventricular failure). Total artificial heart is a form of mechanical circulatory support in which the patient’s native heart ventricles and heart valves are explanted and replaced by a pneumatically powered artificial heart 1. Just like a human heart, the temporary total artificial heart is pulsatile and consists of two ventricles and four valves that pump blood throughout the body. The total artificial heart is made of a special biocompatible plastic, which prevents the total artificial heart from being rejected by your body. Unlike other mechanical circulatory support (MCS) devices, such as left ventricular assist devices (LVADs) that assist the left side of the failing heart, the total artificial heart replaces both the left and right sides of the failing heart.
Similar to receiving a donor heart, the total artificial heart replaces both lower chambers of your heart (the left and right ventricles) plus the four heart valves, eliminating the symptoms and source of your heart failure. In addition, the total artificial heart offers some advantages a donor heart doesn’t: the total artificial heart is readily available when needed (no wait list), is not restricted by blood type or antibody level (no matching required) and is biocompatible with the body (no anti-rejection medications needed). In order to ensure the best outcomes with the total artificial heart, it’s important that patients undergo implant surgery promptly so that prolonged low blood flow doesn’t cause permanent damage to vital organs. The day your failing heart is replaced by the total artificial heart is the day your recovery from heart failure can begin.
Currently, the total artificial heart is approved for use in end-stage biventricular heart failure as a bridge to heart transplantation 1. If you or a loved one is suffering from advanced or end-stage heart failure (NYHA Class III or IV) and your doctor has discussed an immediate or eventual need for a heart transplant, the temporary total artificial heart may be an option for you and your doctor to consider.
The SynCardia total artificial heart (SynCardia Systems, Inc., Tucson, AZ) is the only commercially available total artificial heart in the United States approved by the U.S. Food and Drug Administration (FDA) (see Figure 1) 2. SynCardia total artificial heart provides the most definitive treatment options for patients with biventricular failure who are not candidates for isolated LVAD placement 3. More than 1,700 SynCardia total artificial heart’s have been implanted into patients 4. The SynCardia system has recently developed a smaller 50 mL total artificial heart that was designed to accommodate patients with low body surface area, as low as 1.2 m² (potentially even lower using virtual implantation) 5. This technical improvement should allow the device to be implanted in women and children, and it might be particularly useful in growing adolescents with palliated congenital heart disease 5. To date, the youngest patient to receive the total artificial heart was 9 years old. However, both the 70mL and the 50mL total artificial hearts are too large for use in babies, toddlers and small children.
The 70mL total artificial heart is approved as a bridge to transplant in the U.S., Europe and Canada for people who are:
- Suffering from end-stage, irreversible heart failure affecting both sides of their heart (biventricular failure)
- Eligible to receive a heart transplant or likely to become eligible
The 50mL total artificial heart is approved as a bridge to transplant in Europe and Canada for people who are:
- Suffering from end-stage, irreversible heart failure affecting both sides of their heart (biventricular failure)
- Eligible to receive a heart transplant or likely to become eligible
In the U.S., the 50mL total artificial heart is undergoing an FDA clinical trial for use as a bridge to transplant in both adult and pediatric patients.
More than 1,700 implants worldwide, the 70mL and 50mL total artificial hearts have been used in patients with the following conditions that have contributed to irreversible biventricular failure:
- Acute myocardial infarction
- Aortic root dissection
- Arrhythmogenic right ventricular dysplasia (ARVD)
- Cardiac tumor
- Chagas disease
- Dilated cardiomyopathy
- Donor heart rejection/failure
- Endocarditis
- End-stage congenital heart disease
- Ethylic cardiomyopathy
- Familial cardiomyopathy
- Hypertrophic cardiomyopathy
- Ischemic cardiomyopathy
- LVAD failure (device malfunction or right ventricular failure)
- Marfan syndrome
- Mixed cardiomyopathy
- Myocarditis
- Post/peripartum cardiomyopathy
- Recurrent ventricular tachycardia/fibrillation
- Restrictive cardiomyopathy (including amyloid, sarcoidosis, scleroderma and radiation/chemo-induced cardiomyopathy)
- Valvular cardiomyopathy
- Ventricular septal defect
- Viral cardiomyopathy
- And more
Figure 1. Syncardia CardioWest total artificial heart (orthotopic artificial heart which replaces the ventricles and valves of the native heart)
Surgical implantation
The Syncardia CardioWest total artificial heart consists of two polyurethane ventricles each with a stroke volume of 70 mL and occupies a volume of 400 mL within the chest 6. Given the size of the Syncardia CardioWest total artificial heart, an anterior-posterior chest diameter of at least 10 cm is required by computed tomography (CT) from the anterior border of T10 vertebra to the posterior table of the sternum 7. Each chamber contains 2 mechanical single leaflet tilting disc valves [SynHall (formerly Medtronic Hall), 27 mm inflow, 25 mm outflow] to regulate direction of flow. The two ventricles are pneumatically actuated via drive lines that percutaneously attach to an external pump.
Prior to implantation, the Dacron aortic and pulmonary grafts are sealed using CoSeal Surgical Sealant (Baxter Healthcare Co., Freemont, CA) 6. The grafts and artificial ventricles are soaked in rifampin. A standard median sternotomy is performed. Two small incisions are made in the left upper abdomen and intramuscular tunnels are created through the left rectus muscle for the total artificial heart drivelines. The drivelines are kept away from the midline to avoid injury and loss of pneumatic pressure during redo sternotomy. The patient is started on cardiopulmonary bypass. Mediastinal dissection and mobilization of the great vessels is minimized to maintain dissection planes for subsequent transplantation. The superior vena cava and inferior vena cava are cannulated via the right atrium. The aorta is cross clamped. The pulmonary artery and aorta are divided and separated at the level of the valvular commissures. The left and right ventricles are excised leaving a 1 cm rim of ventricular muscle around the mitral and tricuspid annulus. The mitral and tricuspid valve leaflets are excised. The coronary sinus is overseen. The atrial septum is inspected for a patent foramen ovale, which is closed if found. The total artificial heart atrial quick connects are sutured to the respective valve annulus. Some institutions will reinforce this suture line with a strip of felt. The aortic and pulmonary artery graft quick connects are trimmed and sutured to the respective vessels. It is important that these are carefully cut to size to avoid both stretching and kinking. The pulmonary artery graft is longer than the aortic graft in order to reach over the aortic graft and connect to the artificial right ventricle 7.
At re-entry for transplantation, surgeons have noted an intense inflammatory thickening of the pericardium that increases the difficulty of the mediastinal dissection 6. Polytetrafluoroethylene membrane (PTFE) (Preclude Pericardial Membrane, formerly called the Gore-Tex Surgical Membrane; W.L. Gore & Associates, Flagstaff, AZ) is used to fully reconstruct the pericardium. This maintains avascular tissue planes and dramatically simplifies re-entry for transplantation. Other centers have found wrapping the aortic anastomosis with a sterile tourniquet band to be helpful.
In contrast to the normal oblong cardiac silhouette, the total artificial heart has an overall spherical configuration 6. While PTFE lining of the pericardium facilitates re-entry, contracture of the pericardium about the total artificial heart can limit the space available for transplantation and require maneuvers to open the pericardium/pleura. A saline implant (Mentor smooth round, Mentor Worldwide LLC, Santa Barbara) placed at the former cardiac apex and inflated to 150-200 mL will adequately maintain this space and make such maneuvers unnecessary.
The drivelines are passed through the intramuscular tunnels in the left rectus with the Penrose drains 6. The total artificial heart ventricles are attached to their respective atrial and arterial graft quick connects. An aortic root vent is placed and low pressure/low rate pumping (LV drive pressure 40 mmHg, rate 40 bpm, 40% systole, RV drive pressure 0 mmHg) of the left ventricle is started. Routine de-airing maneuvers are done and the aortic cross clamp is removed. De-airing is confirmed by transesophageal echocardiography (TEE). The patient is often readily weaned off cardiopulmonary bypass as total artificial heart support is increased. Usual post bypass total artificial heart parameters are left drive pressure 180-200 mmHg, right drive pressure 30-60 mmHg, HR 100-120 bpm, and 50% systole. Vacuum is usually not initiated until the chest is closed or sealed.
The decision on whether to close the chest immediately is made based on the bleeding risk 6. If coagulopathy is present, there is a low threshold for packing the mediastinum and delayed sternal closure. Compression of the total artificial heart during chest closure can translate to compression of the left sided pulmonary veins, the inferior vena cava and the left bronchus. Transesophageal echocardiography is used at the end of the case to evaluate for adequate right and left atrial venous return. Should compression be identified it can generally be relieved by tethering the total artificial heart anteriorly to the left costal margin.
Anticoagulation
Long term anticoagulation is routinely used in total artificial heart patients to avoid thromboembolic complications. The initial safety and efficacy trial for the SynCardia total artificial heart as bridge to transplantation reported strokes in 12% of patients and peripheral thrombotic events in 14% patients followed from enrollment to 30 days post-heart transplantation 8.
While the approach to anticoagulation in patients with total artificial heart varies by institution, a multi-targeted antithrombotic approach including anticoagulants, antiplatelet, and rheologic agents are used.
This strategy was first introduced by Szefener at La Pitié Hospital 9. During 1,930 days of total artificial heart support with either Jarvik-7 or CardioWest total artificial heart, Szefener did not observe any strokes in patients who were treated with a combination of aspirin, dipyridamole, heparin, and pentoxifylline. Copeland and his colleagues published similar findings on thrombosis and bleeding outcomes on 99 patients with total artificial heart who were treated with aspirin, unfractionated heparin or warfarin, dipyridamole and pentoxifylline 10. Strokes were observed at a rate of 2%. gastrointestinal bleeding was observed in 4% of patients, intracranial bleeding at 2%, and late thoracic bleeding in 2%.
Anticoagulation begins post-operatively once homeostasis is achieved 11. Heparin is commonly used. As thrombocytopenia is common in critically ill patients requiring a total artificial heart (particularly in patients with prior temporary mechanical circulatory support or continuous renal replacement therapy), there is frequently a concern for heparin induced thrombocytopenia. We routinely use bivalirudin, a direct thrombin inhibitor, as our initial anticoagulation strategy. An aPTT goal of 50-70 is targeted. Once the patient is stable and tolerating oral intake well, they are bridged to warfarin anticoagulation with a target INR of 2-3. The antiplatelet medications aspirin 81 mg daily and dipyridamole 50 mg every 8 h are both started to maintain suppressed platelet function. Platelet function tests (e.g., light transmittance aggregometry) and the thromboelastogram can be used to help titrate appropriate anticoagulation and platelet suppression.
The presence of four single leaflet mechanical valves in the total artificial heart creates a constant amount of hemolysis 12. Similar to LVADs, the LDH, plasma free hemoglobin, and haptoglobin are monitored to assess the degree of hemolysis. Pentoxifylline is a rheologic agent that decreases blood viscosity, platelet adhesion, and increases red blood cell deformity and appears to improve the underlying hemolysis 11. Typical doses are 400 mg every 8 hours.
The introduction of novel oral anticoagulants holds promise for improving the management of thromboembolic and bleeding complications in total artificial heart patients.
When was first artificial heart performed?
On April 4, 1969, Dr. Denton Cooley (founder of Texas Heart Institute) and Dr. Domingo Liotta performed the first human total artificial heart implant using the Liotta Heart (an experimental device designed by Dr. Liotta) 13. The patient was a 47-year-old man with ischemic cardiomyopathy who was unable to come off cardiopulmonary bypass following remodeling ventriculoplasty. The total artificial heart successfully provided hemodynamic support, but the patient quickly developed hemolysis and progressive renal failure. The patient was bridged to transplantation after 64 hours of support but unfortunately died 32 hours later from pseudomonal sepsis. The ground breaking event was filled with controversy regarding improper consent and experimentation 14.
In 1981, Dr. Cooley performed the second human total artificial heart implant (the Akutsu III, developed by Dr. Akutsu) in a 36-year-old man with post cardiotomy shock following coronary bypass surgery 15. The postoperative course was notable for renal failure and severe hypoxia secondary to left pulmonary venous obstruction and required veno-venous extracorporeal membrane oxygenation. The patient was bridged to transplantation after 55 hours of support, but unfortunately died 1 week later from overwhelming sepsis.
Who may benefit an artificial heart?
You may benefit from a total artificial heart if you have heart failure caused by ventricles that no longer pump blood well enough, and other treatments have not worked. If you are waiting for a heart transplant, a total artificial heart can help prolong your life.
Talk to your doctor about whether you are eligible for a total artificial heart and whether the benefits of the device outweigh the risks from surgery and possible complications from living with a total artificial heart.
Who is eligible to get a total artificial heart?
You may be eligible for a total artificial heart if you have heart failure and both of your ventricles are working poorly. The Federal Drug Administration (FDA) approved the total artificial heart device as a bridge to a transplant; total artificial hearts help keep people with heart failure alive while they wait for a heart transplant. In addition, artificial hearts have been given to a few patients as part of clinical research. Researchers are working on smaller total artificial hearts that will fit infants, children, women, and smaller men who are waiting for a heart transplant, as well as devices that are alternative treatments for adults who are not eligible for a transplant.
Who is not eligible to get a total artificial heart?
A total artificial heart should not be used if you have any of these characteristics or conditions:
- Are small. The device is too large to fit in the chests of children and some adults. Researchers are testing smaller total artificial hearts in infants, children, women, and smaller men.
- Can benefit from other treatments, including medicines.
- Have heart failure on only one side of your heart. In these cases, treatment with a ventricular assist device may be more appropriate.
- Cannot take anticlotting medicines. These medicines are required as long as the total artificial heart is in place.
Is the total artificial heart different from a left ventricular assist device?
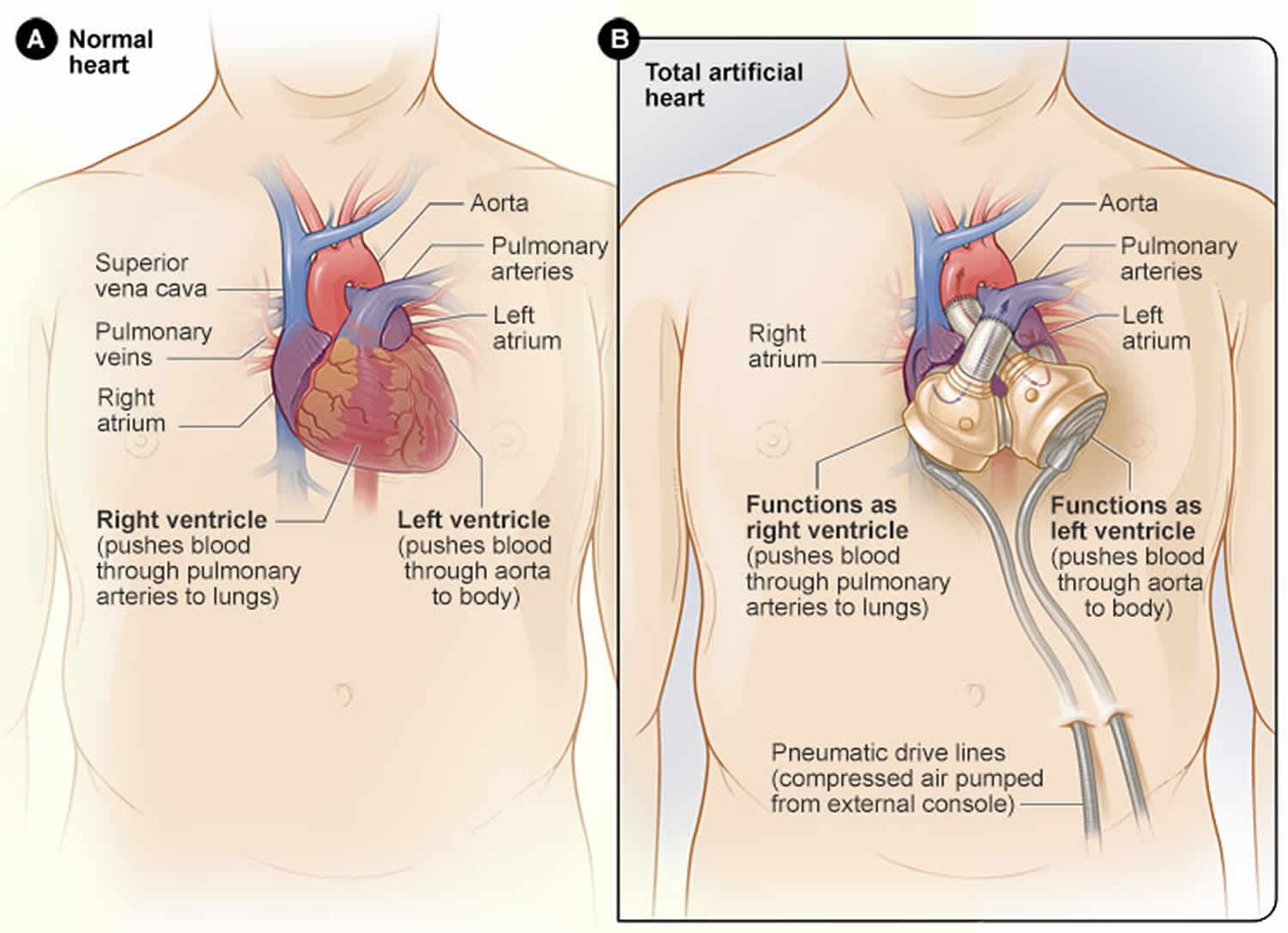
Yes. A left ventricular assist device (LVAD) is a heart pump that is implanted next to the failing heart and supports the left ventricle only. In contrast, the total artificial heart replaces, and occupies the space of, the removed failing heart and pumps blood through both the left and right ventricles.
What will happen during artificial heart surgery?
During surgery, your surgeon will remove the bottom chambers of your heart (the left and right ventricles) as well as the four heart valves and replace them with the total artificial heart, eliminating the symptoms and source of your heart failure so that you can begin your recovery. Many surgeons also take this opportunity to prepare for your eventual donor heart, which may reduce the amount of time your future transplant surgery will take.
How fast will I recover after artificial heart surgery?
Many patients describe an immediate improvement in their condition after waking up from the implant surgery, including having color return to their cheeks, being able to breathe easier and having increased vitality. Most patients are extubated (have their breathing tube removed) within 1 to 2 days and are out of bed sitting in a chair or walking around the floor shortly thereafter. Nutrition, exercise, a positive attitude and a strong support system are all important to a speedy recovery
Your physical and emotional wellbeing have a significant impact on your recovery. It’s important that you maintain a positive attitude and have a strong support system to keep you motivated.
Key components of your recovery will include a healthy diet and exercise. Your medical team will design a nutrition and exercise plan tailored to your individual needs. Following this plan is critical for building up your strength and improving your health for your future heart transplant.
It’s also important to start moving as soon as possible under the supervision of your medical team. Usually patients begin by sitting in a chair, standing and then walking around the hospital room. Eventually, you’ll walk laps around the hospital floor.
Most patients progress to regular workouts as part of their cardiac rehabilitation supervised by hospital staff. Maintaining a healthy diet during this time provides the body with the energy and nutrients it needs to recover.
Once stable, patients can be switched from the C2 Driver to the Freedom® Portable Driver, a smaller, lighter external pump for the total artificial heart. The Freedom Driver can be carried in the Backpack or Shoulder Bag, allowing patients who meet discharge criteria to leave the hospital and enjoy life at home while they wait for a matching donor heart. Life at home greatly improves the patient’s physical, mental and emotional health while eliminating most in-hospital costs for this portion of patient care.
How does an artificial heart work?
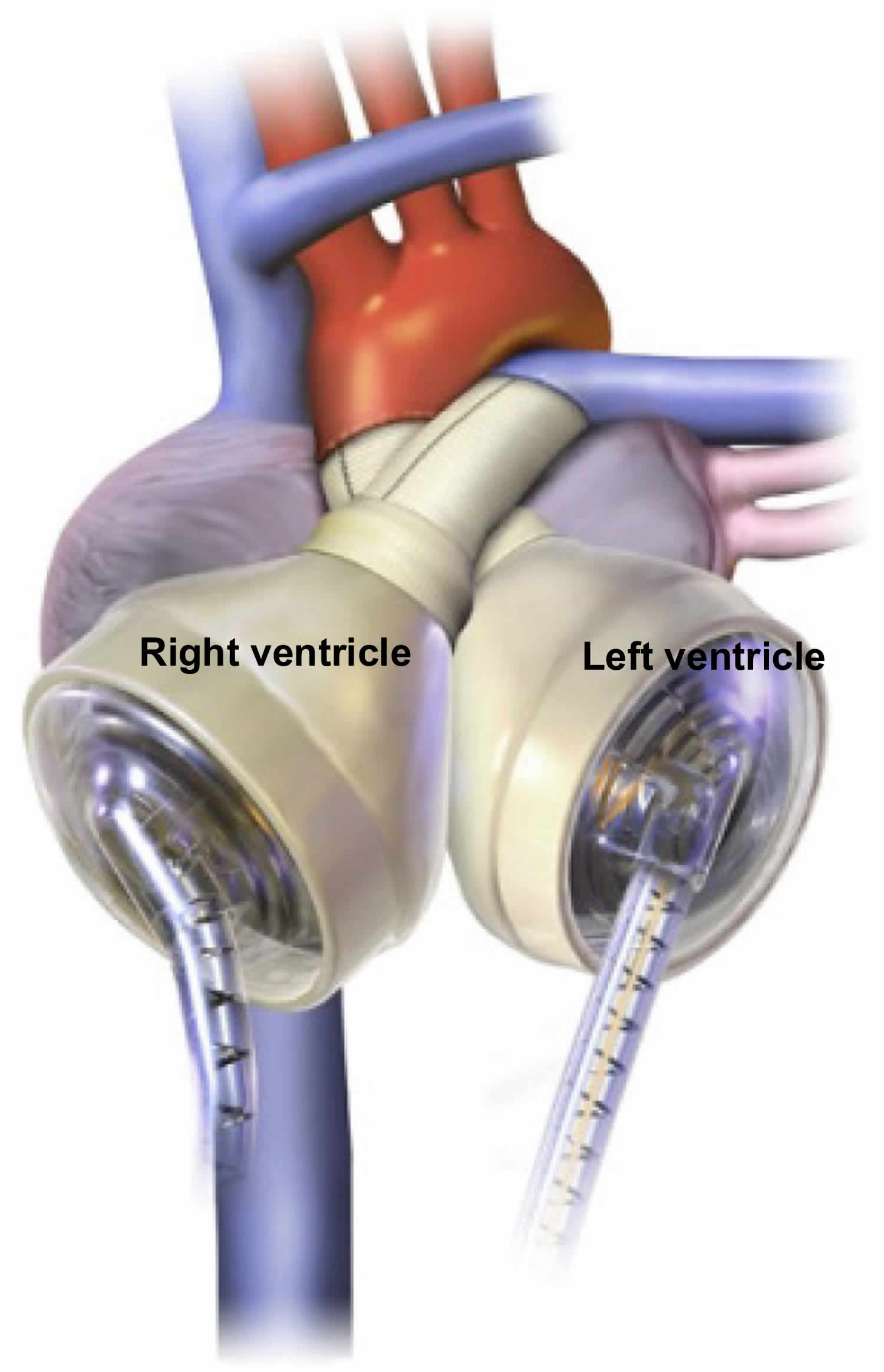
Just like a human heart, the SynCardia total artificial heart is pulsatile and consists of two ventricles and four valves that pump blood throughout the body. The total artificial heart is made of a special biocompatible plastic, which offers a high degree of fatigue resistance and strength for long-term durability.
The total artificial heart consists of two artificial ventricles made from biocompatible plastic, which prevents the total artificial heart from being rejected by the body. The ventricles are connected together by Velcro, which allows the surgeon to position the ventricles in the chest based on the patient’s anatomy.
Each ventricle has two mechanical valves: an inflow valve and an outflow valve. These valves control the direction of blood flow in and out of the ventricle.
Inside each ventricle is a flexible, balloon-like membrane known as a diaphragm. The diaphragm divides the ventricle into two chambers: a blood chamber, which holds the blood when it enters the ventricle, and an air chamber.
Two tubes called cannulae, which are partially covered in velour to promote healing, connect to the air chambers of the ventricles. The cannulae then exit the body through the abdominal wall and attach to two tubes called drivelines. These drivelines are connected to an external pump known as a driver.
The external driver produces pulses of air and vacuum that travel through the drivelines to inflate and deflate the diaphragms within each ventricle, which push blood in and out of the ventricles. The driver runs on batteries and also plugs into a standard electrical outlet.
The SynCardia total artificial heart is a fixed rate device, which means after the beat rate is set, the beats per minute will stay the same, even when your activity level changes. A typical setting is around 125 beats per minute. When you exercise, no adjustments need to be made to the device. Your body will automatically return more blood to the total artificial heart, which will then pump that increased amount of blood back out to your body.
The SynCardia total artificial heart comes in two sizes: 70mL and 50mL. The 70mL total artificial heart fits a majority of men and some women. It is capable of generating blood flow of up to 9.5 liters per minute depending on the patient. The smaller 50cc total artificial heart is designed to fit a majority of women and some adolescents. It can generate blood flow of up to 7.5 liters per minute depending on the patient. In comparison, the average human heart at rest pumps an average of 5 liters of blood per minute.
Artificial heart complications
As experience has grown with the total artificial hearts, rates for the common complications have been established. The major complications of total artificial heart implantation include strokes, infection, bleeding, thrombosis, renal failure, and chronic anemia. One center published outcomes data on 101 patients bridged with the total artificial heart supported for an average of 87 days of support 16. Strokes were reported in 7.9% of patients, 63.4% developed an infection requiring treatment, and bleeding occurred in 42.6% of patients.
The lungs and the urinary tract system were the most common sites of infection. However, mediastinitis occurred in 3% of patients, two of whom died. There was one case of methicillin resistant staphylococcus aureus (MRSA) endocarditis complicated by multiple strokes and death.
Bleeding complications varied in severity. High rates of mediastinal bleeding requiring mediastinal exploration were observed in 24.7% of patients, 44% of whom died within one month. Approximately 4% developed gastrointestinal bleeding. Fifty-eight percent had no bleeding complications.
Renal dysfunction
Post-surgical oliguric renal failure is a frequent complication following total artificial heart implantation. Severe renal dysfunction resulting in a rise in creatinine above 5 mg/dL or requiring dialysis is seen in up to 12% of patients post-operatively 17. One study found 15% of patients required renal replacement therapy who had no previous renal failure 18. Following the removal of the ventricles, B-type natriuretic peptide levels drop precipitously 19. It has been postulated that interruption of this hormonal compensatory mechanism may precipitate renal failure 19. Patients who do develop oliguric renal failure have a prompt and robust increase in urine output following nesiritide infusion without worsening in hemodynamics 19. One small study looked at routinely administering low dose nesiritide (0.05 mcg/kg/min) to all patients undergoing total artificial heart implantation at the time of ventriculectomy and demonstrated maintenance of urine output and GFR 19. While nesiritide administration appears to be beneficial in maintaining short term renal function and management of volume status, whether this results in a durable response compared with the natural history of renal recovery without the addition of nesiritide remains to be proven.
Longterm complications
While currently being investigated for use as destination therapy, the total artificial heart is currently only approved for bridge to transplantation. As a result, data is lacking regarding long-term complications. As of 2011, 47 patients had been supported with a SynCardia total artificial heart for greater than one year worldwide 20. The mean support time was 554 days. Device failure occurred in 10% of patients. Systemic infections were observed in 53% of patients, driveline infections in 27% of patients, thromboembolic events in 19% of patients, and hemorrhagic events in 14% of patients.
Chronic anemia
Severe anemia occurs following total artificial heart implantation that is multifactorial in etiology. Levinson et al. 21 demonstrated that hemolysis, similar to that seen following Left Ventricular Assist Device (LVAD) placement, occurs in patients following total artificial heart implantation. The degree of anemia seen following total artificial heart implantation is generally more severe than following LVAD placement. Mankad et al. 22 published a study comparing anemia in 36 patients who underwent total artificial heart implantation and 14 patients who underwent LVAD placement. Baseline hematocrits were similar between the two groups, and both groups experienced significant drops in hematocrit following device implantation. The anemia following total artificial heart implantation, however, was statistically lower at 2, 4, 6 and 8 weeks following device implantation. The researchers proposed multiple contributory mechanisms to the anemia. Evidence of severe hemolysis, like that which occurs in LVAD patients, was similarly found in total artificial heart patients. Ninety-six percent of total artificial heart patients had undetectable haptoglobin levels and elevated LDH (mean 1,128), and 40% of samples had detectable plasma free hemoglobin. They attributed this hemolysis to shear stress of multiple mechanical parts including the four mechanical valves and pneumatically powered diaphragms. Additionally, they proposed inflammation induced anemia to be playing a role as evidenced by elevated C-reactive protein which may be related to device materials. There was also evidence of inadequate hematopoiesis as demonstrated by a reduced reticulocyte production index. They hypothesized that this may also be mediated by inflammation. Interestingly, post-heart transplantation, the difference in hematocrit between LVAD and total artificial heart disappeared, and by three months post heart transplantation hematocrit returned to baseline in both groups. Despite the severe anemia that occur post-total artificial heart implantation, patients required a median of only 2.5 units of blood outside the post-operative period.
Artificial heart life expectancy
Total Artificial Heart is meant to be temporary, for use in end-stage biventricular heart failure as a bridge to heart transplantation 1. A single center outcomes data was published in 2014. From April 2006 through July 2012 at Virginia Commonwealth University Medical Center, 66 patients were implanted with a total artificial heart 23. Patients were supported for a median duration of 87.5 days. At the time of publication, 76% were successfully bridged to transplantation, 15% were discharged home on a portable Freedom Driver as part of a clinical trial, 11% remained on the device awaiting transplantation, and 14% died on the device. Several patients have been supported by the SynCardia total artificial heart for more than 4.5 years 24.
Some examples of activities SynCardia total artificial heart patients have been able to enjoy with the Freedom Driver include:
- Going to the mall, the beach, the zoo and county fairs with their children
- Riding a bicycle
- Playing pick-up basketball
- Golfing
- Hiking
- Lifting weights
- Tiling floors, painting walls and installing crown molding
- Trapshooting, fishing and hunting
- Taking interstate road trips
- Attending graduations and weddings
- Four-wheeling
Patients may find that when they go out in public, the backpack or the sound of the Freedom Driver can attract attention. Many patients take it as an opportunity to educate others about the total artificial heart, as well as organ donation.
Artificial heart outcomes
The original safety and efficacy trial for the SynCardia total artificial heart was responsible for first establishing the total artificial heart as a relevant and effective intervention for bridging patients dying of biventricular heart failure to heart transplantation 25. Patients were included who had class IV heart failure and hemodynamic insufficiency (hypotension, elevated central venous pressure, on multiple vasoactive medications, intra-aortic balloon pump, or cardiopulmonary bypass). Patients were only chosen who were deemed to be poor LVAD candidates. Patients were effectively bridged to transplantation in an impressive 79% of patients.
To date, there have been no head-to-head randomized control trials comparing the efficacy of total artificial heart with Left Ventricular Assist Devices (LVADs). One retrospective study published in 2001 compared the CardioWest total artificial heart with Novacor and Thoratec LVADs; patients were effectively bridged to transplantation at a rate of 75%, 57%, 38%, respectively 26. Strokes were reported at a rate of 0.03 events per patient-month in CardioWest, 0.28 events per patient-month in Novacor LVAD, and 0.08 events per patient-month in Thoratec LVAD. In this study, it was observed that the patients who had poor outcomes in the LVAD groups were more likely to have concurrent right ventricular failure. The authors concluded that CardioWest be considered first-line in unstable patients who met device size parameters.
Another larger retrospective study of 383 patients published in the Journal of Heart and Lung Transplantation in 2012 selected patients from a multicenter French database and attempted to determine whether type of device, bi-ventricular assist devices or total artificial heart, impacted rates of successful bridging to transplantation 27. This study found no statistically significant difference in rates of successful bridging to transplantation between patients treated with extracorporeal bi-ventricular assist devices, paracorporeal bi-ventricular assist devices, and CardioWest total artificial heart . There was, however, a striking difference in the rates of stroke. Compared with the strokes reported in 61% and 57% in implantable and paracorporeal biventricular devices, respectively, strokes were reported in 16% of patients bridged with CardioWest total artificial heart.
Living with artificial heart
Learn about going home after total artificial heart surgery, ongoing medical care you need, and possible complications of living with a total artificial heart. If you are on the waiting list for a heart transplant, you will stay in close contact with the transplant center.
Slowly increase your activity level
Your recovery will continue after you go home. Once you are at home, you will need to slowly increase your activity level, protect and care for your total artificial heart, and get the right nutrition and exercise.
When you go home after total artificial heart surgery, you will likely be able to do more activities than you could before the surgery. The machine with the power supply and device controls is portable, so you will probably be able to get out of bed, get dressed, and move around the house. You may even be able to drive. Your healthcare team will advise you on the level of activity that is safe for you.
Protect and care for your total artificial heart
Your healthcare team will explain how to troubleshoot your total artificial heart, change or charge the portable device’s batteries, and respond to alarms.
A total artificial heart is attached to a power source outside your body through holes in your abdomen. These holes increase the risk of bacteria getting in and causing an infection. Your doctor may tell you how often to check your temperature as part of your routine care. You may also need to take special steps before you bathe, shower, or swim. Your healthcare team will explain how to make sure that the tubes going through your skin do not get wet. They also will recommend that you avoid steam baths and dry saunas, which can overheat the total artificial heart driver.
Get the right nutrition and physical activity
While you recover from total artificial heart surgery, it is important that you get good nutrition. Talk with your healthcare team about following a proper eating plan for recovery. Read about heart-healthy eating.
Your healthcare team may recommend a supervised physical activity program. Physical activity can give your body the strength that it needs to recover. During the months or years before surgery when your heart was not working well, the muscles in your body may have weakened. Building up the muscles again will allow you to do more activities and feel less tired.
Ongoing care
You will have regular checkups with your healthcare team to check your progress and make sure that your total artificial heart is working well. There is a chance that you may have problems with your total artificial heart. In some cases, you may need surgery again.
Medicines
You will need to take anticlotting medicine to prevent dangerous blood clots as long as you have a total artificial heart. Regular blood tests will help determine the correct dose.
You also will need to take medicine to try to prevent infections. Your doctor may tell you how often to check your temperature to make sure you do not have a fever, which can be a warning sign of infection.
Make sure to take all your medicines as prescribed and report any side effects to your doctor.
Heart transplant
If you are on the waiting list for a heart transplant, you will likely be in close contact with the transplant center. Most donor hearts must be transplanted within four hours after removal from the donor.
You need to be prepared to arrive at the hospital within two hours of being notified about a donor heart.
Emotional health
Getting a total artificial heart may cause fear, anxiety, and stress. If you are waiting for a heart transplant, you may worry that the total artificial heart will not keep you alive long enough to get a new heart. You may feel overwhelmed or depressed.
These feelings are common for someone going through major heart surgery. Talk with your healthcare team about how you feel. Talking to a professional counselor also can help. If you are very depressed, your doctor may recommend medicines or other treatments that can improve your quality of life.
Support from family and friends can help relieve stress and anxiety. Let your loved ones know how you feel and what they can do to help you.
Problems with your device
There is a risk that the total artificial heart will have problems and not work properly. For example, the device may not have the correct pumping action, the power source may fail, or parts may stop working well. Talk with your healthcare team about how to spot potential problems and what to do if a problem occurs.
If you have a total artificial heart, you may want to download the product manual from the manufacturer site and visit Medical Device Recalls external link to learn about FDA recalls for these devices and their components.
References- Cook JA, Shah KB, Quader MA, et al. The total artificial heart [published correction appears in J Thorac Dis. 2017 Mar;9(3):E342]. J Thorac Dis. 2015;7(12):2172–2180. doi:10.3978/j.issn.2072-1439.2015.10.70 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4703693
- Shah KB, Smallfield MC, Tang DG, et al. Mechanical circulatory support devices in the ICU. Chest 2014;146:848-57
- Cohn WE, Timms DL, Frazier OH. Total artificial hearts: past, present, and future. Nat Rev Cardiol. 2015;12:609–617
- https://syncardia.com/clinicians/home/
- Wells D, Villa CR, Simon Morales DL. The 50/50 cc total artificial heart trial: extending the benefits of the total artificial heart to underserved populations. Semin Thorac Cardiovasc Surg Pediatr Card Surg Annu. 2017;20:16–19.
- Tang DG, Shah KB, Hess ML, et al. Implantation of the syncardia total artificial heart. J Vis Exp 2014; 89
- DeVries WC. Surgical technique for implantation of the Jarvik-7-100 total artificial heart. JAMA 1988;259:875-80
- DeVries WC, Anderson JL, Joyce LD, et al. Clinical use of the total artificial heart. N Engl J Med 1984;310:273-8
- Szefner J. Control and treatment of hemostasis in cardiovascular surgery. The experience of La Pitié Hospital with patients on total artificial heart. Int J Artif Organs 1995;18:633-48
- Copeland J, Copeland H, Nolan P, et al. Results with an anticoagulation protocol in 99 SynCardia total artificial heart recipients. ASAIO J 2013;59:216-20
- Ensor CR, Cahoon WD, Crouch MA, et al. Antithrombotic therapy for the CardioWest temporary total artificial heart. Tex Heart Inst J 2010;37:149-58
- Mankad AK, Tang DG, Clark WB, et al. Persistent anemia after implantation of the total artificial heart. J Card Fail 2012;18:433-8
- Orthotopic cardiac prosthesis for two-staged cardiac replacement. Cooley DA, Liotta D, Hallman GL, Bloodwell RD, Leachman RD, Milam JD. Am J Cardiol. 1969 Nov; 24(5):723-30.
- Law-medicine notes. The first mechanical heart transplant: informed consent and experimentation. Curran WJ. N Engl J Med. 1974 Nov 7; 291(19):1015-6.
- The total artificial heart. Cooley DA. Nat Med. 2003 Jan; 9(1):108-11.
- Copeland JG, Copeland H, Gustafson M, et al. Experience with more than 100 total artificial heart implants. J Thorac Cardiovasc Surg 2012;143:727-34
- Copeland JG, Smith RG, Arabia FA, et al. Total artificial heart bridge to transplantation: a 9-year experience with 62 patients. J Heart Lung Transplant 2004;23:823-31
- El-Banayosy A, Arusoglu L, Morshuis M, et al. CardioWest total artificial heart: Bad Oeynhausen experience. Ann Thorac Surg 2005;80:548-52
- Shah KB, Tang DG, Kasirajan V, et al. Impact of low-dose B-type natriuretic peptide infusion on urine output after total artificial heart implantation. J Heart Lung Transplant 2012;31:670-2
- Torregrossa G, Morshuis M, Varghese R, et al. Results with SynCardia total artificial heart beyond 1 year. ASAIO J 2014;60:626-34
- Levinson MM, Copeland JG, Smith RG, et al. Indexes of hemolysis in human recipients of the Jarvik-7 total artificial heart: a cooperative report of fifteen patients. J Heart Transplant 1986;5:236-48
- Mankad AK, Tang DG, Clark WB, et al. Persistent anemia after implantation of the total artificial heart. J Card Fail 2012;18:433-8.
- Implantation of the syncardia total artificial heart. Tang DG, Shah KB, Hess ML, Kasirajan V. J Vis Exp. 2014 Jul 18; 89
- https://syncardia.com/patients/patient-resources/frequently-asked-questions/
- Copeland JG, Smith RG, Arabia FA, et al. Cardiac replacement with a total artificial heart as a bridge to transplantation. N Engl J Med 2004;351:859-67
- Copeland JG 3rd, Smith RG, Arabia FA, et al. Comparison of the CardioWest total artificial heart, the novacor left ventricular assist system and the thoratec ventricular assist system in bridge to transplantation. Ann Thorac Surg 2001;71:S92-7; discussion S114-5
- Survival after biventricular mechanical circulatory support: does the type of device matter? Kirsch M, Mazzucotelli JP, Roussel JC, Bouchot O, N’loga J, Leprince P, Litzler PY, Vincentelli A, Groupe de Réflexion sur l’Assistance Mécanique. J Heart Lung Transplant. 2012 May; 31(5):501-8.