What is astaxanthin
Astaxanthin (3,3′-dihydroxy-β, β′-carotene-4,4′-dione) is a xanthophyll carotenoid created by micro-algae (Haematococcus pluvialis, Chlorella zofingiensis, Chlorococcum, and Phaffia rhodozyma) in times of stress 1. Haematococcus pluvialis is a green microalga, which accumulates high astaxanthin content under stress conditions such as high salinity, nitrogen deficiency, high temperature and light 2. When the Haematococcus pluvialis algae are exposed to change in their environment such as direct sunlight, starved of food or being eaten, it produces astaxanthin to protect itself. As a potent antioxidant, astaxanthin protects the algae from free radicals and oxidative stress, keeping it alive and well when stressed. Astaxanthin accumulates up to 3.8% on the dry weight basis in Haematococcus pluvialis. Astaxanthin produced from haematococcus pluvialis is a main source for human consumption 3. Astaxanthin carotenoid is what gives many sea creatures such as salmon, krill, and crayfish their red color. Carotenoids contribute to the yellow, orange, and red colors of the skin, shell, or exoskeleton of aquatic animals. Indeed, carotenoids are the most widespread pigments found in nature, as they occur in bacteria, yeasts, mold, all green plants, and many animals, and therefore various functions have been attributed to them. From anthropocentric consideration, the most significant aspect of carotenoids is the color they impart to our food and environment.
Astaxanthin is closely related to beta-carotene, lutein, and zeaxanthin, sharing with them many of the general metabolic and physiological functions attributed to carotenoids. Mammals, including humans, lack the ability to synthesize astaxanthin or to convert dietary astaxanthin into vitamin A.
Astaxanthin is a carotenoid widely used in salmon, trout, red seabream, shrimp, lobster, fish eggs and crustacean aquaculture to provide the pink color characteristic of that species. Astaxanthin is also found in some birds, such as flamingoes, quails, and other species. Astaxanthin, similar to other carotenoids, cannot be synthesized by animals and must be provided in the diet. Salmon farming increased substantially in the 1980s, which created a large market for astaxanthin, the principal pigment of salmon. Since animals lack the ability to synthesize carotenoids, the pigments must be supplemented to feeds, usually at considerable expense to the farmer (10 to 15% of total feed costs). This application has been well documented for over two decades and is currently the major market driver for the astaxanthin pigment. Currently, chemically synthesized astaxanthin and canthaxanthin (β,β-carotene4,4′-dione) are added to salmon feeds as pigmenters, but there is considerable interest within the aquaculture industry in using natural sources of astaxanthin. Additionally, astaxanthin also plays a key role in animals as an intermediary in reproductive processes, the carotenoids are associated with reproductive organs and hence the hatching success and survival of alevins (newly spawned salmons or trouts still carrying the yolk).
Synthetic astaxanthin dominates the world market but recent interest in natural sources of the pigment has increased substantially. Common sources of natural astaxanthin are the green algae Haematococcus pluvialis, the red yeast Phaffia rhodozyma, as well as crustacean byproducts. Each natural pigment source has its limitations and they currently cannot compete economically with the synthetic additive 4. The algae Haematococcus pluvialis has a high concentration of astaxanthin (0.2 to 2%), but industrial application is limited by the lengthy autotrophic cultivation in open freshwater ponds and the requirement for disruption of the cell wall to liberate the carotenoid 4. The red yeast Phaffia rhodozyma has desirable properties as a biological source of pigment, including rapid heterotrophic metabolism and production of high cell densities in fermentors, but its content of astaxanthin in wild strains is only 200 to 300 μg/g yeast (0.02 to 0.03%) 4. Mutants have been isolated that produce >3000 μg total carotenoid per gram of yeast (>0.30%) in shake flasks after 5 d growth, and measurement of carotenoid fluorescence in individual cells indicates that levels of 10,000 to 15,000 μg/g can be obtained 4. High producers, however, are often unstable and further strain development is required.
Astaxanthin possesses an unusual antioxidant activity which has caused a surge in the nutraceutical market for the encapsulated product. Also, health benefits such as cardiovascular disease prevention, immune system boosting, bioactivity against Helycobacter pylori, and cataract prevention, have been associated with astaxanthin consumption 5. Research on the health benefits of astaxanthin is very recent and has mostly been performed in test tube study or at the pre-clinical level with humans.
Table 1. Microorganism sources of astaxanthin
| Sources | Astaxanthin (%) on the Dry Weight Basis |
| Chlorophyceae | |
| Haematococcus pluvialis | 3.8 |
| Haematococcus pluvialis (K-0084) | 3.8 |
| Haematococcus pluvialis (Local isolation) | 3.6 |
| Haematococcus pluvialis (AQSE002) | 3.4 |
| Haematococcus pluvialis (K-0084) | 2.7 |
| Chlorococcum | 0.2 |
| Chlorella zofingiensis | 0.001 |
| Neochloris wimmeri | 0.6 |
| Ulvophyceae | |
| Enteromorpha intestinalis | 0.02 |
| Ulva lactuca | 0.01 |
| Florideophyceae | |
| Catenella repens | 0.02 |
| Alphaproteobacteria | |
| Agrobacterium aurantiacum | 0.01 |
| Paracoccus carotinifaciens (NITE SD 00017) | 2.2 |
| Tremellomycetes | |
| Xanthophyllomyces dendrorhous (JH) | 0.5 |
| Xanthophyllomyces dendrorhous (VKPM Y2476) | 0.5 |
| Labyrinthulomycetes | |
| Thraustochytrium sp. CHN-3 (FERM P-18556) | 0.2 |
| Malacostraca | |
| Pandalus borealis | 0.12 |
| Pandalus clarkia | 0.015 |
What is astaxanthin used for?
Astaxanthin is a red fat-soluble pigment which does not have pro-Vitamin A activity in the human body, although some of the studies reported that astaxanthin has more potent biological activity than other carotenoids 6. Unlike many other antioxidant supplements, astaxanthin does not develop into a pro-oxidant once exhausted. The United States Food and Drug Administration (FDA) has approved the use of astaxanthin as food colorant in animal and fish feed 7. The European Commission considers natural astaxanthin as a food dye 8. Astaxanthin is used as a source of pigment in the feed for salmon, trout and shrimp 9. For dietary supplement in humans and animals, astaxanthin is obtained from seafood or extracted from Haematococcus pluvialis 10. The consumption of astaxanthin can prevent or reduce risk of various disorders in humans and animals 3.
Astaxanthin Metabolism: Absorption and Tissue Distribution
In pharmacokinetic studies, after ingestion of esterified natural astaxanthin, only unesterified astaxanthin appears in the blood 11. This is most likely due to breaking the ester bonds by digestive enzymes via their hydrolytic activity. Absorption into the intestinal lining cells (enterocytes) is thought to occur by passive diffusion and is facilitated in the presence of fat or other lipids 12. The enterocytes then incorporate the unesterified astaxanthin into chylomicrons, which transport it to the liver 13. The liver does not convert this molecule to vitamin A or otherwise biochemically transform it 14. Instead unesterified astaxanthin becomes incorporated into low-density lipoprotein (LDL) and high-density lipoprotein (HDL), which then distribute it to the tissues via the circulation 11.
When astaxanthin is fed to human subjects, detailed pharmacokinetic data are difficult to obtain for single doses of less than 10 mg, due to limitations of assay precision 11. However, there is good data to indicate a single 10-mg dose can persist in the blood for 24 hours and a 100-mg dose for 76 hours 11. Doses as low as 1 mg can significantly increase blood levels when taken once daily for four weeks 15.
Astaxanthin’s bioavailability is substantially affected by meal timing and by smoking. In a 2009 study, a single 48-mg dose was much better absorbed when taken just after a meal than on an empty stomach, and was about 40-percent less bioavailable in subjects who smoked 12.
Astaxanthin health benefits
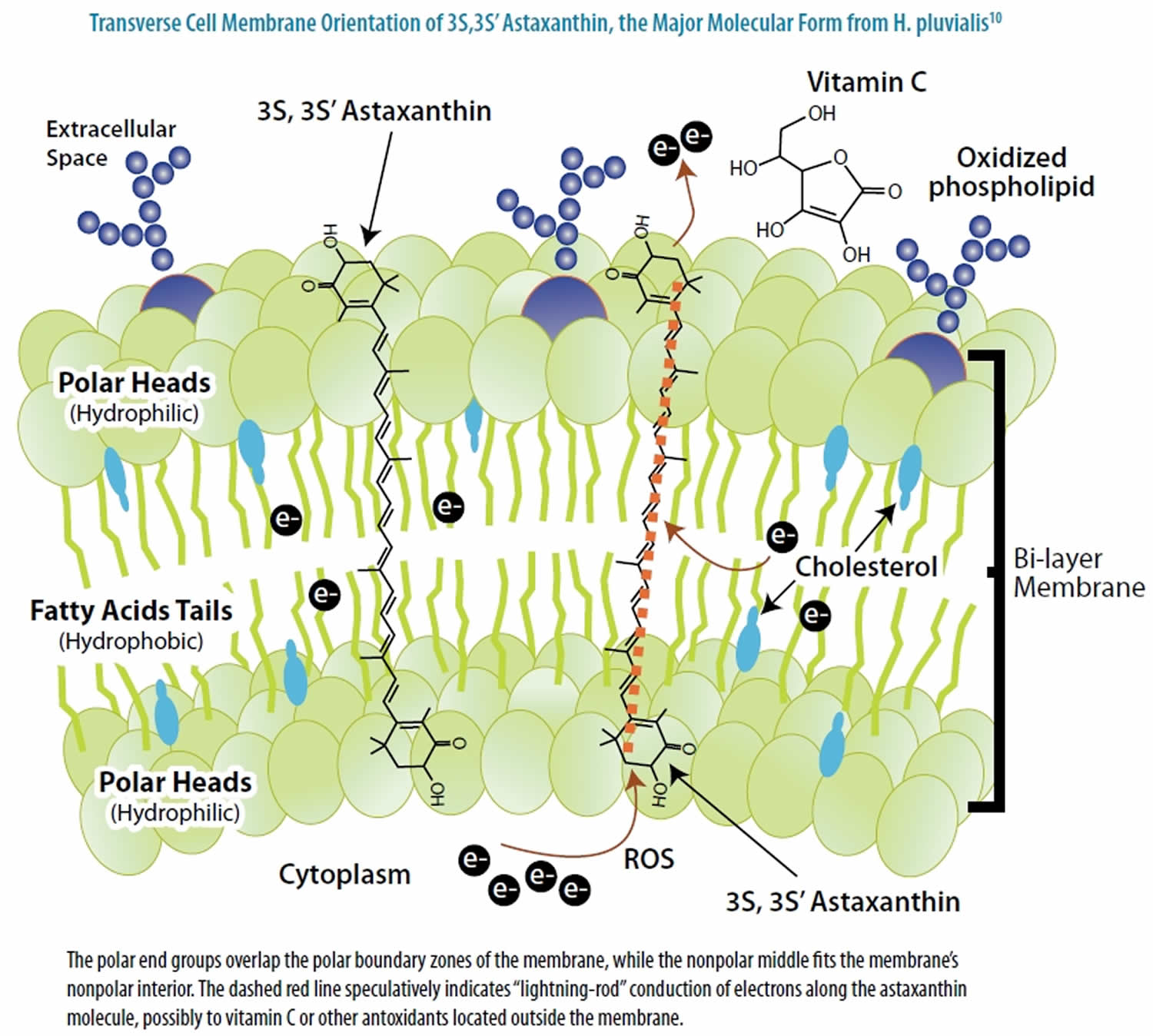
The use of astaxanthin (3,3’-dihydroxy-beta,beta-carotene-4,4’-dione) as a nutritional supplement has been rapidly growing in foods, feeds, nutraceuticals and pharmaceuticals. Astaxanthin, a xanthophyll carotenoid, is a nutrient with unique cell membrane actions and diverse clinical benefits. Astaxanthin molecule neutralizes free radicals or other oxidants by either accepting or donating electrons, and without being destroyed or becoming a pro-oxidant in the process. The astaxanthin molecule has an extended shape, with a polar structure at either end of the molecule and a nonpolar zone in the middle (Figure 1). The polar structures are ionone rings that have potent capacity for quenching free radicals or other oxidants, primarily in an aqueous environment, but possibly also in the absence of water 16. Astaxanthin’s linear, polar-nonpolar-polar molecular layout equips it to precisely insert into the membrane and span its entire width. In this position, astaxanthin can intercept reactive molecular species within the membrane’s hydrophobic interior and along its hydrophilic boundaries (see Figure 3).
The red color of Astaxanthin is due to the conjugated double bonds at the center of the compound. This type of conjugated double bond acts as a strong antioxidant by donating the electrons and reacting with free radicals to convert them to be more stable product and terminate free radical chain reaction in a wide variety of living organisms 17. Astaxanthin showed better biological activity than other antioxidants 18, because it could link with cell membrane from inside to outside (Figure 3).
Figure 1. Astaxanthin chemical structure
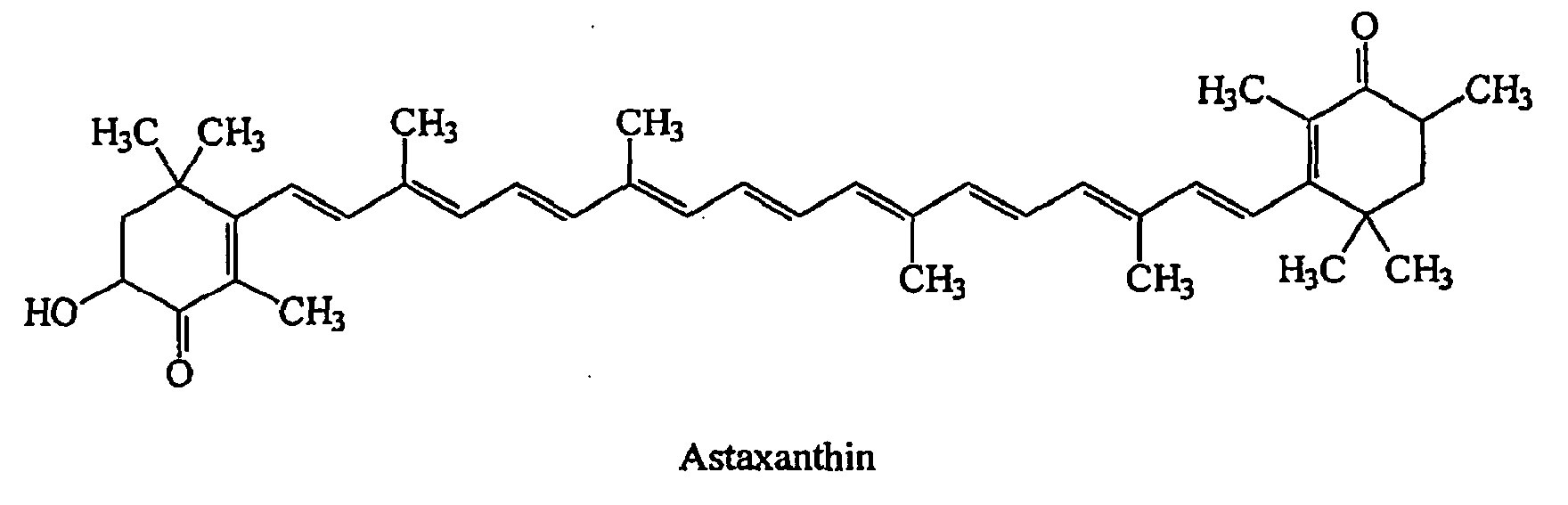 Astaxanthin has unique chemical properties based on its molecular structure. The presence of the hydroxyl (OH) and keto (CdO) moieties on each ionone ring explains some of its unique features, namely, the ability to be esterified and a higher antioxidant activity and a more polar nature than other carotenoids (Figure 1). In its free form, astaxanthin is considerably unstable and particularly susceptible to oxidation. Hence it is found in nature either conjugated with proteins (e.g., salmon muscle or lobster exoskeleton) or esterified with one or two fatty acids (monoester and diester forms), which stabilize the molecule. Various astaxanthin isomers have been characterized on the basis of the configuration of the two hydroxyl groups on the molecule. the geometrical and optical isomers of astaxanthin are distributed selectively in different tissues and that levels of free astaxanthin in the liver are greater than the corresponding concentration in the plasma, suggesting concentrative uptake by the liver. Astaxanthin, similar to other carotenoids, is a very lipophilic compound and has a low oral bioavailability.
Astaxanthin has unique chemical properties based on its molecular structure. The presence of the hydroxyl (OH) and keto (CdO) moieties on each ionone ring explains some of its unique features, namely, the ability to be esterified and a higher antioxidant activity and a more polar nature than other carotenoids (Figure 1). In its free form, astaxanthin is considerably unstable and particularly susceptible to oxidation. Hence it is found in nature either conjugated with proteins (e.g., salmon muscle or lobster exoskeleton) or esterified with one or two fatty acids (monoester and diester forms), which stabilize the molecule. Various astaxanthin isomers have been characterized on the basis of the configuration of the two hydroxyl groups on the molecule. the geometrical and optical isomers of astaxanthin are distributed selectively in different tissues and that levels of free astaxanthin in the liver are greater than the corresponding concentration in the plasma, suggesting concentrative uptake by the liver. Astaxanthin, similar to other carotenoids, is a very lipophilic compound and has a low oral bioavailability.The bonding patterns of natural astaxanthin generate many different molecular forms (isomers), each with its unique three-dimensional shape. Pertaining to its use as a dietary supplement, virtually all commercially available natural astaxanthin is predominantly in the all-trans geometric form 3S,3S’ Astaxanthin, as
occurs in Haematococcus pluvialis and as illustrated in Figure 2. This is the predominant natural astaxanthin used in all clinical trials to date.
Another complication in the chemistry of natural astaxanthin is that the molecule in its free form is relatively uncommon within the various organisms that produce it. Instead, most of this astaxanthin is either conjugated with proteins or esterified with one or two fatty acids (as astaxanthin acyl monoesters or diesters) 19. Acyl esters make up more than 99 percent of the astaxanthin from H. pluvialis and approximately 80 percent of astaxanthin in krill 19. Thus, acyl monoester and diester forms make up virtually all the astaxanthin currently available in dietary supplements.
Figure 2. Astaxanthin used in supplement
Figure 3. Astaxanthin spanning across a cell membrane
In its position spanning the membrane, astaxanthin provides versatile antioxidant actions, including 20:
- donating electrons to unpaired electrons to neutralize free radicals;
- pulling away (“abstracting”) an unpaired electron, which also can neutralize a radical;
- bonding with the radical to form an unreactive “adduct”;
- conducting electrons or electronic energy out of the membrane (Figure 3);
- neutralizing radical species of nitrogen, sulfur, or carbon, in addition to oxygen; and
- carrying very low net molecular energy, therefore providing resistance to transformation into a pro-oxidant molecule.
Astaxanthin a Unique Cell Membrane Antioxidant
Astaxanthin provides cell membranes with potent protection against free radical or other oxidative attack. Experimental studies confirm that this nutrient has a large capacity to neutralize free radical or other oxidant activity in the nonpolar (“hydrophobic”) zones of phospholipid aggregates, as well as along their polar (hydrophilic) boundary zones 21. A particularly elegant experimental study by McNulty et al conclusively established astaxanthin’s membrane protection capacity 20.
McNulty’s group 20 assembled model membranes from phosphatidylcholine and a small amount of cholesterol, at ratios similar to natural cell membranes. They then introduced varying amounts of astaxanthin, zeaxanthin, lutein, beta-carotene, and lycopene to these model membranes and monitored the packing of the phospholipids by X-ray diffraction. They also induced peroxidative processes by gently increasing the temperature of the system. With the exception of astaxanthin, the carotenoids all disrupted the phospholipid packing and exacerbated peroxidative breakdown. The greater the membrane disruption by a carotenoid, the greater was its peroxidative effect. Only astaxanthin reduced peroxidation (by 41 percent) and preserved the membrane structure. The researchers gave particular credit to astaxanthin’s unique ionone polar groups.
Astaxanthin has also protected human LDL against oxidative attack. In a Japanese study 22, astaxanthin purified from krill was provided to healthy volunteers (average age 28 years; n=24) at 0 mg/day, 1.8 mg/day, 3.6 mg/day, 14.4 mg/day, or 21.6 mg/day, for 14 days. After supplementation, venous blood was drawn and the LDL analyzed for susceptibility to oxidation (lag time after a chemical peroxidative trigger) for each subject compared to baseline. Astaxanthin significantly increased lag time, which indicated a protective effect, at the dose levels of 3.6 mg/day and higher.
Supplementation with astaxanthin may lower lipid peroxidation in vivo 23. A double-blind, randomized controlled trial investigated astaxanthin (8 mg/day) versus a placebo for three months in Finnish men ages 19-33. Plasma 12- and 15-hydroxy fatty acids, both markers of lipid peroxidation, were statistically significantly reduced in the astaxanthin group, but not in the placebo group. The reduction in 15-hydroxy fatty acid in the astaxanthin group fell just short of significance when compared to the placebo group.
Table 2. Health benefits of astaxanthin in human subjects
| Duration of Experiment | Subjects in Humans | Dosage (mg/day) | Benefits of Astaxanthin | References |
|---|---|---|---|---|
| 2 weeks | Volunteers | 1.8, 3.6, 14.4 and 21.6 | Reduction of LDL oxidation | 24 |
| Single dose | Middle aged male volunteers | 100 | Astaxanthin take up by VLDL chylomicrons | 25 |
| 8 weeks | Healthy females | 0.2 and 8 | Decreased plasma 8-hydoxy-2′-deoxyguanosine and lowered in CRP levels | 26 |
| 8 weeks | Healthy adults | 6 | Assessed by blood pressure | 27 |
| 10 days | Healthy males | 6 | Improved blood rheology | 28 |
| 12 weeks | Healthy non-smoking finnish males | 8 | Decreased oxidation of fatty acids | 29 |
| 12 months | Age related macular degeneration | 4 | Improved central retinal dysfunction in age related macular degeneration | 30 |
| 12 weeks | Middle aged/elderly | 12 | Improved Cog health battery scores | 31 |
| 12 weeks | Middle aged/elderly | 6 | Improved groton maze learning test scores | 31 |
| 8 or 6 weeks | Healthy female or male | 6 | Improved skin winkle, corneocyte layer, epidermis and dermis | 32 |
| 2 weeks | Disease (bilateral cataract) | 6 | Improved superoxide scavenging activity and lowered hydroperoxides in the human aqueous humor | 33 |
Abbreviations: LDL, Low-density lipoproteins, VLDL, Very low-density lipoprotein, CRP, C-reactive protein.
Antioxidant Effects
Significant antioxidant powers have been ascribed to astaxanthin, based primarily on experimental findings. The real breakthrough with this nutrient, however, is that it produces clinically significant antioxidant benefits in human subjects, including groups especially vulnerable to oxidative stress, such as smokers, the obese, and the overweight.
Oxidative stress can be defined as a relative excess of free radical activity over antioxidant capacity, which in human subjects can be determined using blood samples 34. Alternately, oxidative breakdown products can be measured in the blood 35. People who are overweight or obese tend to manifest greater “oxidative stress” when compared to individuals within the normal weight range 36. In a Korean double-blind randomized controlled trial 35, astaxanthin “normalized” oxidative stress in individuals with weight challenges. In this three-week study, overweight and obese individuals (body mass index [BMI] >25.0 kg/m²; n=23) were randomized to receive astaxanthin at 5 mg/day or 20 mg/day and compared to a control group (n=10) with normal body weight (BMI <25.0 kg/m²) who received no intervention 35. At baseline, the plasma levels in overweight and obese individuals were significantly higher than normal weight individuals on two oxidative biomarkers – malondialdehyde (MDA) and isoprostanes (ISP), while plasma levels in overweight and obese individuals were significantly lower on two antioxidant measures – superoxide dismutase (SOD) and total antioxidant capacity. At the three-week mark, when compared against baseline, both astaxanthin groups showed significant lowering of oxidative markers malondialdehyde and isoprostanes (p<0.01 for 5 mg/day; p<0.001 for 20 mg/day). The astaxanthin groups also had significant increases in superoxide dismutase (SOD) and total antioxidant capacity. Marked improvements on all four measures caused the overweight and obese subjects to become statistically indistinguishable from the control group, suggesting that supplementation lowered oxidative stress and improved aspects of the antioxidant defense system. The improvements were not significantly better for the 20 mg/day group than the 5 mg/day group.
In another randomized controlled trial conducted by the same group 37, heavy smokers (n=39) were randomly allocated to receive astaxanthin at 5-, 20-, or 40 mg/day for three weeks. Compared with baseline, the plasma malondialdehyde and isoprostane levels decreased, whereas superoxide dismutase (SOD) and total antioxidant capacity increased in all intervention groups over the three-week period. In particular, isoprostane levels showed a significant dose-dependent decrease after astaxanthin intake.
Astaxanthin also can protect against oxidative DNA damage. In a 2010 double-blind randomized controlled trial conducted by Park et al, 38 healthy women (ages 20-23; n=42) received either a placebo or astaxanthin at doses of 2 mg/day or 8 mg/day for eight weeks. Both astaxanthin dosages significantly lowered plasma 8-hydroxy-2’-deoxyguanosine (8-OHdG), an indicator of oxidative DNA breakdown. Plasma 8-isoprostane, a marker of lipid peroxidation, was not significantly lowered. The authors attributed this finding to a lack of sensitivity and accuracy in their assay method.
Anti-inflammatory Benefits
In the Park double-blind randomized controlled trial 38, astaxanthin also significantly lowered C-reactive protein (CRP), a biomarker of systemic inflammation 39. Although the 2-mg/day dose had a significant CRP-lowering effect, the 8-mg/day dose fell short of statistical significance. Compared with the lower dose, the 8-mg/day dose significantly increased the cytokine interferon-gamma, which may indicate an anti-inflammatory effect, but also significantly increased interleukin-6 (IL-6), which can have a pro-inflammatory effect. The clinical significance of these findings is unclear, particularly since none of these, except CRP, is a generally accepted inflammatory marker.
Astaxanthin’s effect on CRP was also investigated in a small double-blind trial that was published without peer review 40. Subjects (ages 40-60 years; n=19), with no diagnosis of cardiovasculardisease, kidney disease, diabetes, or cancer, received three softgel capsules daily supplying either astaxanthin at 12 mg/day (with 120 mcg of lutein, 195 IU of vitamin A activity [in the form of beta-carotene), and 150 IU of vitamin E) or a placebo (safflower oil) for eight weeks. The astaxanthin combination lowered CRP levels by about 20 percent (from 1.35 mg/dL to 1.07 mg/dL), which was significantly better than placebo.
An eight-week, double-blind randomized controlled trial conducted on rheumatoid arthritis subjects was published in abstract form 41. One group (n=14) received the same combination as in the previously described study – 12 mg/day astaxanthin plus 120 mcg of lutein, 195 IU vitamin A activity (from beta-carotene), and 150 IU of vitamin E, while the other group (n=7) received a placebo. The improvement in self-reported scores of pain and satisfaction for the astaxanthin group was significantly better than for the placebo group, which suggests a possible anti-inflammatory effect.
Astaxanthin has been reported to benefit other inflammatory conditions such as canker sores, carpal tunnel syndrome, and “tennis elbow,” but the evidence currently available to support these claims is insufficient.
Metabolic Syndrome
Astaxanthin improved certain blood lipids in subjects with moderately elevated serum triglycerides (TGs). Healthy non-obese subjects (BMI <28 kg/m²), ages 20-65 years (n=61) with fasting triglycerides in the range 120-200 mg/dL, were recruited into a double-blind randomized controlled trial 42. They were randomly allocated to receive astaxanthin at 6 mg/day, 12 mg/day, or 18 mg/day, or a placebo for 12 weeks. Astaxanthin, as compared to placebo, significantly elevated HDL-cholesterol at the doses of 6 mg/day and 12 mg/day. It also significantly lowered triglycerides at doses of 12 mg/day and 18 mg/day compared to placebo. There was no effect on LDL-cholesterol at any dose. Astaxanthin also significantly increased blood adiponectin levels.
Adiponectin is a hormone produced by adipose tissue, cardiac and skeletal muscle, and vessel endothelia. Serum levels of adiponectin tend to be reduced in obese and/or diabetic subjects, smokers, patients with coronary heart disease, and individuals with metabolic syndrome 43. Although the results of this study suggest a normalization of adiponectin levels, 12 weeks of supplementation had no effect on BMI.
In a small, open-label trial (16 subjects), astaxanthin did not produce clinically significant benefits on any of the criteria for metabolic syndrome 44. Further investigation is required under better controlled conditions in order to clarify astaxanthin’s utility for this condition.
Anti-Diabetic Activity
Generally, oxidative stress levels are very high in diabetes mellitus patients. It is induced by hyperglycemia, due to the dysfunction of pancreatic β-cells and tissue damage in patients. Astaxanthin could reduce the oxidative stress caused by hyperglycemia in pancreatic β-cells and also improve glucose and serum insulin levels 45. Astaxanthin can protect pancreatic β-cells against glucose toxicity. It was also shown to be a good immunological agent in the recovery of lymphocyte dysfunctions associated with diabetic rats 46. In another study, ameliorate oxidative stress in streptozotocin-diabetes rats were inhibited by the combination of astaxanthin with α-tocopherol 47. It is also inhibited glycation and glycated protein induced cytotoxicity in human umbilical vein endothelial cells by preventing lipid/protein oxidation 48. Improved insulin sensitivity in both spontaneously hypertensive corpulent rats and mice on high fat plus high fructose diets was observed after feeding with astaxanthin 49. The urinary albumin level in astaxanthin treated diabetic mice was significantly lower than the control group 45. Some of the studies demonstrated that astaxanthin prevents diabetic nephropathy by reduction of the oxidative stress and renal cell damage 50.
Effects on Circulation
As people age, their red blood cells (RBCs) can be more susceptible to oxidative attack, resulting in peroxidative damage to the red blood cell membrane phospholipids 51, impairing its oxygen-carrying capacity. In a 2011 double-blind randomized controlled trial 52, healthy subjects, ages 50-69 years (n=30), were randomly allocated to receive astaxanthin at 6 mg/day or 12 mg/day or a placebo for 12 weeks. Both astaxanthin intakes significantly lowered red blood cell hydroperoxide levels; the 12 mg/day dose did not work significantly better than the 6 mg/day dose.
Astaxanthin also improved an experimental measure of “rheology” (blood flow capacity) in healthy men 53. Venous blood was drawn with heparin to protect against coagulation, then forced using mild pressure through tiny “microchannels,” each just seven millionths of a meter wide, approximating the diameter of an red blood cell and the width of a capillary. The time required to traverse these capillary-type tubes under a set pressure was termed the transit time. A total of 20 men were selected whose blood demonstrated transit times in the range of 45-70 seconds per 100 microliters. They were then randomly allocated to receive either astaxanthin (6 mg/day) or a placebo for 10 days. Upon retest, the astaxanthin group had significantly faster transit time compared to placebo. This finding suggests astaxanthin could potentially improve microcirculation.
Anticancer Activity
The specific antioxidant dose may be helpful for the early detection of various degenerative disorders. Reactive oxygen species such as superoxide, hydrogen peroxide and hydroxyl radical are generated in normal aerobic metabolism. Singlet oxygen is generated by photochemical events whereas peroxyl radicals are produced by lipid peroxidation. These oxidants contribute to aging and degenerative diseases such as cancer and atherosclerosis through oxidation of DNA, proteins and lipids 54. Antioxidant compounds decrease mutagenesis and carcinogenesis by inhibiting oxidative damage to cells. Cell–cell communication through gap junctions is lacking in human tumors and its restoration tends to decrease tumor cell proliferation. Gap junctional communication occurs due to an increase in the connexin-43 protein via upregulation of the connexin-43 gene. Gap junctional communication was improved in between the cells by natural carotenoids and retinoids 55. Canthaxanthin and astaxanthin derivatives enhanced gap junctional communication between mouse embryo fibroblasts 56. Increased connexin-43 expression in murine fibroblast cells by β-carotene was reported 57. Astaxanthin showed significant antitumor activity when compared to other carotenoids like canthaxanthin and β-carotene 58. It also inhibited the growth of fibrosarcoma, breast, and prostate cancer cells and embryonic fibroblasts 59. Increased gap junctional intercellular communication in primary human skin fibroblasts cells were observed when treated with astaxanthin 60. Astaxanthin inhibited cell death, cell proliferation and mammary tumors in chemically induced male/female rats and mice 61. H. pluvialis extract inhibited the growth of human colon cancer cells by arresting cell cycle progression and promoting apoptosis reported by Palozza et al. 59. Nitroastaxanthin and 15-nitroastaxanthin are the products of astaxanthin with peroxynitrite, 15-nitroastaxanthin anticancer properties were evaluated in a mouse model. Epstein-Barr virus and carcinogenesis in mouse skin papillomas were significantly inhibited by astaxanthin treatment 61.
Memory and other Higher Brain Functions
Astaxanthin might improve cognitive functions. In a small, open-label trial, 10 healthy men ages 50-69, who had been complaining of forgetfulness, received astaxanthin (12 mg/day) for 12 weeks 62. On a computerized test designed to accurately detect early cognitive deterioration (“CogHealth” from CogState Ltd, Melbourne, Australia), they showed improvement on measures of reaction time, attention, and working memory.
Although this trial clearly was only preliminary, astaxanthin has shown a variety of brain benefits under experimental conditions. Unlike much of the experimental research conducted with this supplement, the following studies employed levels readily achieved by oral intake in humans.
Astaxanthin:
- significantly improved the memory performance of mice in the Morris water maze 63;
- effectively protected cultured nerve cells against hydrogen peroxide toxicity 64 and down-regulated genes linked to cell death and up-regulated genes linked to cell survival 65;
- specifically protected the mitochondria of cultured nerve cells against toxic attack 64,
- 65; and
- stimulated the proliferation of cultured nerve stem cells 66.
Effect on Vision and Eye Fatigue
Astaxanthin has been extensively researched for its benefits for vision, especially in Japan. Yuan 19 and Kajita 67 discussed double-blind and other controlled trials that were published in Japanese with English abstracts. They concluded that astaxanthin taken at 6 mg/day consistently improved visual sharpness, even in healthy subjects.
Astaxanthin also might relieve eye fatigue in persons using computer monitors. Extended work at computer monitors is linked to eyes strain and to blurred vision, often accompanied by tensing of the muscles of the shoulder and low back 68. One double-blind randomized controlled trial used instrumentation to measure eye muscle endurance, the usual basis for eye strain 69. Japanese visual display terminal workers, ages 38-53 (n=26), received astaxanthin (5 mg/day) or a placebo for four weeks. A non-visual display terminal control group received neither astaxanthin nor placebo. After supplementation, eye strain was significantly more improved in the astaxanthin group than in the placebo group.
Many individuals, as they age, suffer decline in the eye’s ability to focus on near objects (presbyopia). In an open-label Japanese trial 67, papillary constriction capacity was determined using instrumentation in presbyopic middle-aged and older subjects (ages 46-65 years; n=22). They then received astaxanthin (6 mg/day) for four weeks. Astaxanthin significantly improved pupillary constriction, and more than 60 percent of the subjects indicated on a questionnaire that they also experienced improvement in the categories of “difficulty to see near objects,” “eye strain,” “blurred vision,” and “shoulder and low-back stiffness.”
Muscle Performance and Endurance
In a double-blind randomized controlled trial 70, young healthy male students (ages 17-19 years; n=40) were subjected to fitness, strength, and endurance testing, then randomized to receive astaxanthin (4 mg/day) or a placebo for six months. Astaxanthin significantly
improved performance in the assessment designed to measure strength/endurance (maximum number of knee bends [“squats”] performed while carrying a 42.5 kg barbell) over the placebo group. The maximum number of knee bends increased by more than 50 percent in the astaxanthin group versus 19 percent in the placebo group. No benefit was detected for the assessments of strength/explosivity or overall fitness. This finding (and probably others asserted from unpublished trials) may have motivated the double-blind trials currently in progress with astaxanthin for protection against oxidative stress 71 and for improving speed and endurance in soccer players 72.
Male Fertility and Reproduction
In a double-blind randomized controlled trial, astaxanthin was evaluated for protecting sperm function and fertility 73. Thirty men were recruited, all from infertile couples in which the female partner showed no demonstrable cause of infertility. They were randomized to receive either astaxanthin (16 mg/day) or a placebo for three months. During that period they were allowed to provide semen for intrauterine insemination, and pregnancy occurrence was recorded.
By the end of three months, sperm linear velocity was significantly increased in the astaxanthin group but not in the placebo group 73. Semen oxygen radical generation (upon stimulation by the oxidant phorbol ester) was markedly decreased in the astaxanthin group. However, the most telling outcome of this trial was the pregnancy rate, which was 54.5 percent for the astaxanthin group compared to 10.5 percent for the placebo group.
Immuno-Modulation
Immune system cells are very sensitive to free radical damage. The cell membrane contains poly unsaturated fatty acids (PUFA). Antioxidants in particular astaxanthin offer protection against free radical damage to preserve immune-system defenses. There are reports on astaxanthin and its effect on immunity in animals under laboratory conditions however clinical research is lacking in humans. Astaxanthin showed higher immuno-modulating effects in mouse model when compared to β-carotene 74. Enhanced antibody production and decreased humoral immune response in older animals after dietary supplementation of astaxanthin was reported 74. Astaxanthin produced immunoglobulins in human cells in a laboratory study 75. Eight week-supplementation of astaxanthin in humans 76 resulted in increased blood levels of astaxanthin and improved activity of natural killer cells which targeted and destroyed cells infected with viruses. In this study, T and B cells were increased, DNA damage was low, and C-reactive protein (CRP) was significantly lower in the astaxanthin supplemented group.
Astaxanthin dosage
The doses of astaxanthin used in clinical trials have ranged from 1 mg/day to 40 mg/day (with the majority in the 6-12 mg range); single-dose pharmacokinetic studies used up to 100 mg per dose. Recommended dose of astaxanthin is 2–4 mg/day 5. As a dietary supplement, astaxanthin should be taken along with fats, with or immediately prior to meals, to ensure its optimal absorption 77. It is recommended to administer astaxanthin with omega-3 rich seed oils such as chia, flaxseed, fish, nutella, walnuts and almonds.
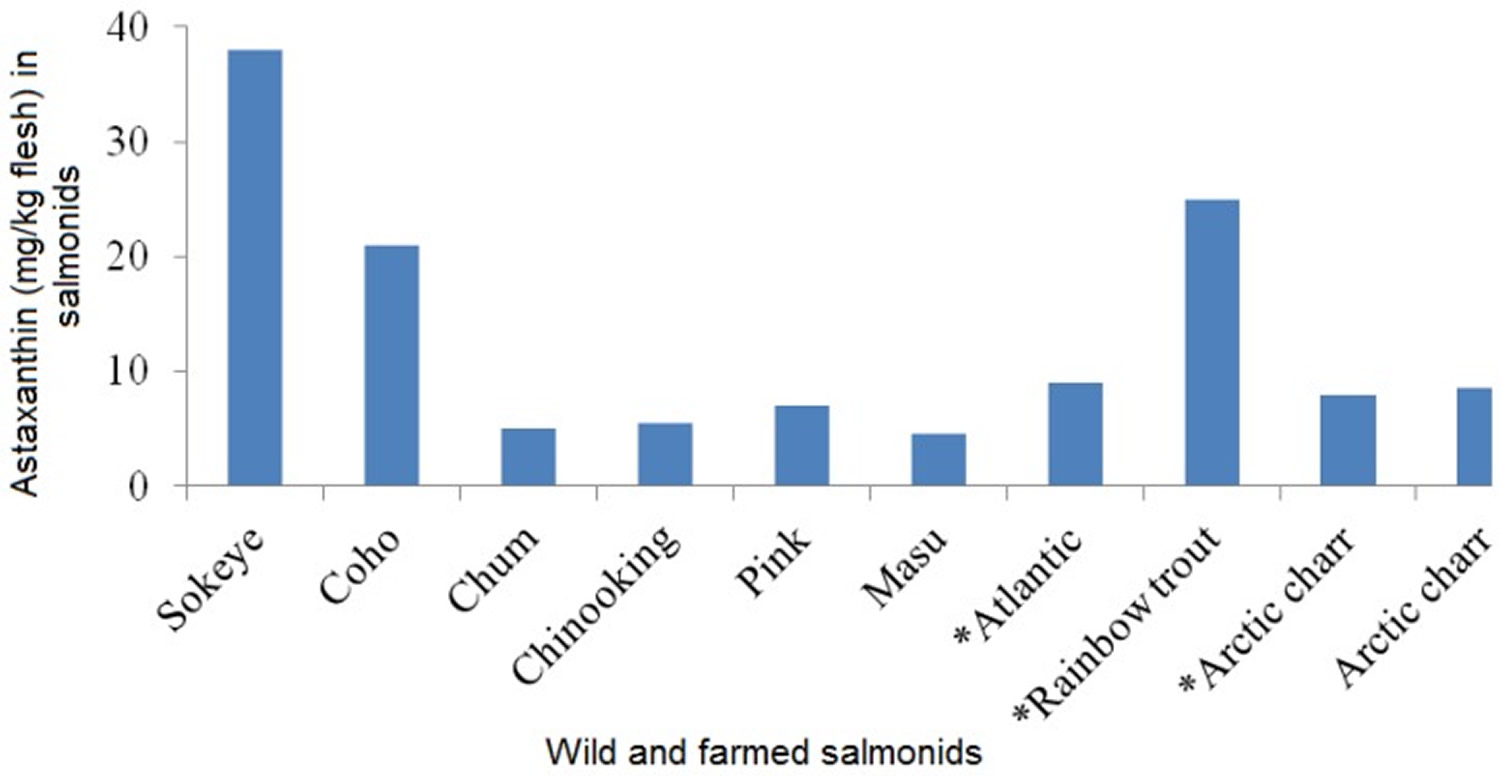
Astaxanthin content in wild and farmed salmons are shown in Figure 2. Among the wild salmonids, the maximum astaxanthin content in wild Oncorhynchus species was reported in the range of 26–38 mg/kg flesh in sockeye salmon whereas low astaxanthin content was reported in chum 78. Astaxanthin content in farmed Atlantic salmon was reported as 6–8 mg/kg flesh. Astaxanthin is available in the European (6 mg/kg flesh) and Japanese market (25 mg/kg flesh) from large trout. Shrimp, crab and salmon can serve as dietary sources of astaxanthin 78. Wild caught salmon is a good source of astaxanthin. In order to get 3.6 mg of astaxanthin you can eat 165 grams of salmon per day. Astaxanthin supplement at 3.6 mg per day can be beneficial to health as reported by Iwamoto et al. 79.
Figure 1. Astaxanthin levels (mg/kg flesh) of wild and farmed (*) salmons
Astaxanthin side effects
Astaxanthin is safe, with no side effects when it is consumed with food. It is lipid soluble, accumulates in animal tissues after feeding of astaxanthin to rats and no toxic effects were found 80. Excessive astaxanthin consumption leads to yellow to reddish pigmentation of the skin in animals.
Astaxanthin has demonstrated safety in numerous human clinical trials. In one open-label clinical study on subjects with metabolic syndrome (n=17) 44, astaxanthin (16 mg/day, for three months) significantly raised blood bilirubin, potassium and creatine kinase, although all three values remained within normal range. Also, astaxanthin significantly lowered the liver enzyme gamma-glutamyl transpeptidase (GGTP). Since the researchers noted this enzyme was abnormally elevated in 11 of the 17 subjects at baseline, this astaxanthin effect may have been beneficial.
Animal experiments have investigated astaxanthin at levels well over 120 mg/day in human equivalents 81, without causing apparent harm.
Following a request from the European Commission, the European Food Safety Authority Panel on Dietetic Products, Nutrition and Allergies (NDA) was asked to deliver a scientific opinion on the safety of astaxanthin‐rich ingredients AstaREAL A1010 and AstaREAL L10 as novel food ingredients in the context of Regulation (EC) No 258/97 82. The novel food ingredients are produced from astaxanthin‐rich alga Haematococcus pluvialis. Astaxanthin content is 5.0‐5.6 % in AstaREAL A1010 powder, 10.0‐12.0 % in AstaREAL L10 oil and 2.5‐2.7 % in AstaREAL L10 encapsulated oil. Sufficient information was provided regarding the composition, specification, manufacture and stability of the novel food ingredients. The novel food ingredients are intended to be used in fermented liquid dairy products, non‐fermented liquid dairy products, fermented soya products and fruit drinks for healthy adults at a maximum incorporation level of 1.6 mg astaxanthin per 100 g or 100 mL 82.
The European Food Safety Authority Panel considers that the composition of the ingredients and results from available studies do not indicate that the consumption of the novel food ingredients is nutritionally disadvantageous at the proposed daily intake 82.
Astaxanthin is absorbed in the human gastro-intestinal tract. Its bioavailability and distribution seem to depend on a variety of factors, including its form, its mode of consumption and the smoking habits of the consumer. Astaxanthin is also absorbed in rodents. The metabolic fate of astaxanthin involves the cleavage of the polyene chain at the C9, C9 positions and stepwise reduction in both rats and humans. On the basis of similarities in absorption and metabolic fate, the European Food Safety Authority Panel considers that rats are an acceptable species for toxicity testing of astaxanthin 82.
Based on the results of in vitro and in vivo genotoxicity studies on the biomass of Haematococcus pluvialis and other astaxanthin products, the European Food Safety Authority Panel concludes that it has no safety concerns regarding genotoxicity of the novel food ingredients 82.
In the human studies provided, which addressed safety endpoints, no clinically relevant changes or adverse effects were observed after consumption of the novel food ingredients or other astaxanthin-rich ingredients from Haematococcus pluvialis at doses ranging from 2 to 40 mg/day astaxanthin for 10 days to 3 months 82. However, as these studies were of only short duration, the European Food Safety Authority Panel considers that no conclusion can be drawn as regards long-term effects 82.
Supplemental intakes of β-carotene have been shown to increase the risk of lung cancer in smokers, and concerns were expressed that the novel food ingredients may induce similar effects. There are neither human nor animal studies that have investigated astaxanthin and smoking with regard to the risk of lung cancer. There are differences in structure, metabolism and function between astaxanthin and β-carotene. In contrast to β-carotene, astaxanthin is more polar, is not a precursor of vitamin A and is considered as an antioxidant with no indication of pro-oxidative properties. The European Food Safety Authority Panel concludes that the available data do not indicate that the novel food ingredients at the proposed level of use would increase the risk of lung cancer in smokers 82.
The Panel considers that the likelihood of adverse allergic reactions to the novel food ingredients is low.
Biomass of Haematococcus pluvialis (algal meal containing 3 % astaxanthin) and astaxanthin-rich oil obtained by solvent extraction from Haematococcus pluvialis biomass (containing ca. 5 % astaxanthin) were tested for subchronic toxicity in rats. There is no indication from these studies that the novel food ingredients would be more toxic than astaxanthin. Therefore, the Panel bases the evaluation of the novel food ingredients on astaxanthin and considers the acceptable daily intake (ADI) of 0.034 mg/kg body weight for astaxanthin derived by the European Food Safety Authority Panel on Additives and Products or Substances used in Animal Feed.
The European Food Safety Authority Panel notes that the maximum intake of 4 mg astaxanthin per day (0.06 mg/kg body weight per day for a 70 kg person) from the novel food ingredients as proposed by the applicant and the estimated mean intake based on the use levels in the proposed food categories (0.106 mg/kg body weight per day) exceed the acceptable daily intake (ADI) for astaxanthin of 0.034 mg/kg body weight per day by approximately two- and three-fold, respectively. The European Food Safety Authority Panel therefore concludes that the safety of the novel food ingredients AstaREAL A1010 and AstaREAL L10 at the proposed use and use levels has not been established.
References- Astaxanthin: a review of its chemistry and applications. Higuera-Ciapara I, Félix-Valenzuela L, Goycoolea FM. Crit Rev Food Sci Nutr. 2006; 46(2):185-96. https://www.tandfonline.com/doi/abs/10.1080/10408690590957188
- Sarada R., Ranga Rao A., Sandesh B.K., Dayananda C., Anila N., Chauhan V.S., Ravishankar G.A. Influence of different culture conditions on yield of biomass and value added products in microalgae. Dyn. Biochem. Proc. Biotechnol. Mol. Biol. 2012;6:77–85.
- Kidd P. Astaxanthin, cell membrane nutrient with diverse clinical benefits and anti-aging potential. Altern. Med. Rev. 2011;16:355–364. http://archive.foundationalmedicinereview.com/publications/16/4/355.pdf
- Eric A. Johnson & Gil-Hwan An (1991) Astaxanthin from Microbial Sources, Critical Reviews in Biotechnology, 11:4, 297-326, DOI: 10.3109/07388559109040622 https://www.tandfonline.com/doi/abs/10.3109/07388559109040622
- Ambati RR, Siew Moi P, Ravi S, Aswathanarayana RG. Astaxanthin: Sources, Extraction, Stability, Biological Activities and Its Commercial Applications—A Review. Marine Drugs. 2014;12(1):128-152. doi:10.3390/md12010128. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3917265/
- Ambati RR, Siew Moi P, Ravi S, Aswathanarayana RG. Astaxanthin: Sources, Extraction, Stability, Biological Activities and Its Commercial Applications—A Review. Marine Drugs. 2014;12(1):128-152. doi:10.3390/md12010128. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3917265
- U.S. Food and Drug Administration. GRAS Notices. https://www.accessdata.fda.gov/scripts/fdcc/index.cfm?set=GRASNotices
- Roche F. Astaxanthin As a Pigmenter in Salmon Feed, Color Additive Petition 7C02 1 1, United States Food and Drug Administration. Hoffman-La Roche Ltd.; Basel, Switzerland: 1987. Astaxanthin: Human food safety summary; p. 43.
- Higuera-Ciapara I., Felix-Valenzuela L., Goycoolea F.M. Astaxanthin: A review of its chemistry and applications. Crit. Rev. Food Sci. Nutr. 2006;46:185–196. doi: 10.1080/10408690590957188 https://www.ncbi.nlm.nih.gov/pubmed/16431409
- Guerin M., Huntley M.E., Olaizola M. Haematococcus astaxanthin: Applications for human health and nutrition. Trends Biotechnol. 2003;21:210–216. doi: 10.1016/S0167-7799(03)00078-7. https://www.ncbi.nlm.nih.gov/pubmed/12727382
- Coral-Hinostroza GN, Ytrestoyl T, Ruyter B, Bjerkeng B. Plasma appearance of unesterified astaxantin geometrical E/Z and optical R/S isomers in men given single doses of a mixture of optical 3 and 3’R/S isomers of astaxanthin fatty acyl diesters. Comp Biochem Physiol C Toxicol Pharmacol 2004;139:99-110
- Okada Y, Ishikura M, Maoka T. Bioavailability of astaxanthin in Haematococcus algal extract: the effects of timing of diet and smoking habits. Biosci Biotechnol Biochem 2009;73:1928-1932.
- Coral-Hinostroza GN, Ytrestoyl T, Ruyter B, Bjerkeng B. Plasma appearance of unesterified astaxantin geometrical E/Z and optical R/S isomers in men given single doses of a mixture of optical 3 and 3’R/S isomers of astaxanthin fatty acyl diesters. Comp Biochem Physiol C Toxicol Pharmacol 2004;139:99-110.
- Kistler A, Liechti H, Pichard L, et al. Metabolism and CYP-inducer properties of astaxanthin in man and primary human hepatocytes. Arch Toxicol 2002;75:665-675.
- Miyazawa T, Nakagawa K, Kimura F, et al. Plasma carotenoid concentrations before and after supplementation with astaxanthin in middle-aged and senior subjects. Biosci Biotechnol Biochem 2011;75:1856-1858.
- Goto S, Kogure K, Abe K, et al. Efficient radical trapping at the surface and inside the phospholipid membrane is responsible for highly potent antiperoxidative activity of the carotenoid astaxanthin. Biochim Biophys Acta 2001;1512:251-258.
- Haematococcus astaxanthin: applications for human health and nutrition. Guerin M, Huntley ME, Olaizola M. Trends Biotechnol. 2003 May; 21(5):210-6. https://www.ncbi.nlm.nih.gov/pubmed/12727382/
- Potential health-promoting effects of astaxanthin: a high-value carotenoid mostly from microalgae. Yuan JP, Peng J, Yin K, Wang JH. Mol Nutr Food Res. 2011 Jan; 55(1):150-65. https://www.ncbi.nlm.nih.gov/pubmed/21207519/
- Yuan JP, Peng J, Yin K, Wang JH. Potential health-promoting effects of astaxanthin: a high-value carotenoid mostly from microalgae. Mol Nutr Food Res 2011;55:150-165.
- McNulty HP, Byun J, Lockwood SF, et al. Differential effects of carotenoids on lipid peroxidation due to membrane interactions. X-ray diffraction analysis. Biochim Biophys Acta 2007;1768:167-174.
- Fassett RG, Coombes JS. Astaxanthin: a potential therapeutic agent in cardiovascular disease. Mar Drugs 2011;9:447-465.
- Iwamoto T, Hosoda K, Hirano R, et al. Inhibition of low-density lipoprotein oxidation by astaxanthin. J Atheroscler Throm 2000;7:216-222.
- Karppi J, Rissanen TH, Nyyssonen K, et al. Effects of astaxanthin supplementation on lipid peroxidation. Int J Vitam Nutr Res 2007;77:3-11.
- Iwamoto T., Hosoda K., Hirano R., Kurata H., Matsumoto A., Miki W., Kamiyama M., Itakura H., Yamamoto S., Kondo K. Inhibition of low-density lipoprotein oxidation by astaxanthin. J. Atheroscler. Thromb. 2000;7:216–222. https://www.ncbi.nlm.nih.gov/pubmed/11521685
- Osterlie M., Bjerkeng B., Liaaen-Jensen S. Plasma appearance and distribution of astaxanthin E/Z isomers in plasma lipoproteins of after single dose administration of astaxanthin. J. Nutr. Biochem. 2000;11:482–492. doi: 10.1016/S0955-2863(00)00104-2 https://www.ncbi.nlm.nih.gov/pubmed/11120445
- Park J.S., Chyun J.H., Kim Y.K., Line L.L., Chew B.P. Astaxanthin decreased oxidative stress and inflammation and enhanced immune response in humans. Nutr. Metab. 2010;7:1–10 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2845588/
- Spiller G.A., Dewell A. Safety of an astaxanthin rich Haemaotoccu pluvialis algal extract: A randomized clinical trial. J. Med. Food. 2003;6:51–56. doi: 10.1089/109662003765184741 https://www.ncbi.nlm.nih.gov/pubmed/12804020
- Miyawaki H., Takahashi J., Tsukahara H., Takehara I. Effects of astaxanthin on human blood rheology. J. Clin. Biochem. Nutr. 2008;43:69–74. doi: 10.3164/jcbn.2008048 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2533721/
- Karppi J., Rissanen T.H., Nyyssonen K., Kaikkonen J., Olsson A.G., Voutilainen S., Salonen J.T. Effects of astaxanthin supplementation on lipid peroxidation. Int. J. Vitam. Nutr. Res. 2007;77:3–11. doi: 10.1024/0300-9831.77.1.3
- Parisi V., Tedeschi M., Gallinaro G., Varano M., Saviano S., Piermarocchi S. Carotenoids and antioxidants in age-related maculopathy italian study: multifocal electroretinogram modifications after one year. Ophthalmology. 2008;115:324–333. doi: 10.1016/j.ophtha.2007.05.029
- Katagiri M., Satoh A., Tsuji S., Shirasawa T. Effects of astaxanthin rich Haematococcus pluvialis extact on cognitive function: Arandomised double blind, placebo-controlled study. J. Clin. Biochem. Nutr. 2012;51:102–107. doi: 10.3164/jcbn.D-11-00017
- Tominaga K., Hongo N., Karato M., Yamashita E. Cosmetic benefits of astaxanthin on humans subjects. Acta Biochim. Pol. 2012;59:43–47
- Hashimoto H., Arai K., Hayashi S., Okamoto H., Takahashi J., Chikuda M., Obara Y. Effects of astaxanthin on antioxidation in human aqueous humor. J. Clin. Biochem. Nutr. 2013;53:1–7. doi: 10.3164/jcbn.13-6 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3705160/
- Iwabayashi M, Fujioka N, Nomoto K, et al. Efficacy and safety of eight-week treatment with astaxanthin in individuals screened for increased oxidative stress burden. Anti Aging Med 2009;6:15-21.
- Choi HD, Kim JH, Chang MJ, et al. Effects of astaxanthin on oxidative stress in overweight and obese adults. Phytother Res 2011 Apr 8. doi: 10.1002/ptr.3494
- Grattagliano I, Palmieri VO, Portincasa P, et al. Oxidative stress-induced risk factors associated with the metabolic syndrome: a unifying hypothesis. J Nutr Biochem 2008;19:491-504.
- Kim JH, Chang MJ, Choi HD, et al. Protective effects of Haematococcus astaxanthin on oxidative stress in healthy smokers. J Med Food 2011;14:1469-1475.
- Park JS, Chyun JH, Kim YK, et al. Astaxanthin decreased oxidative stress and inflammation and enhanced immune response in humans. Nutr Metab (Lond) 2010;7:18. doi:10.1186/1743-7075-7-18
- Genest J. C-reactive protein: risk factor, biomarker and/or therapeutic target? Can J Cardiol 2010;26:41A-44A.
- Spiller GA. Effect of daily use natural astaxanthin on C-reactive protein. http://www.cyanotech.com/pdfs/bioastin/batl43.pdf
- Nir Y, Spiller G, Multz C. Effect of an astaxanthin containing product on rheumatoid arthritis. J Am Coll Nutr 2002;21:490
- Yoshida H, Yanai H, Ito K, et al. Administration of natural astaxanthin increases serum HDL-cholesterol and adiponectin in subjects with mild hyperlipidemia. Atherosclerosis 2010;209:520-523.
- Kajikawa Y, Ikeda M, Takemoto S, et al. Association of circulating levels of leptin and adiponectin with metabolic syndrome and coronary heart disease in patients with various coronary risk factors. Int Heart J 2011;52:17-22.
- Uchiyama A, Okada Y. Clinical efficacy of astaxanthin-containing Haematococcus pluvialis extract for the volunteers at risk of metabolic syndrome. J Clin Biochem Nutr 2008;43:390-393
- Uchiyama K., Naito Y., Hasegawa G., Nakamura N., Takahashi J., Yoshikawa T. Astaxanthin protects β-cells against glucose toxicity in diabetic db/db mice. Redox Rep. 2002;7:290–293. doi: 10.1179/135100002125000811
- Otton R., Marin D.P., Bolin A.P., Santos R.C., Polotow T.G., Sampaio S.C., De Barros M.P. Astaxanthin ameliorates the redox imbalance in lymphocytes of experimental diabetic rats. Chem. Biol. Interact. 2010;186:306–315. doi: 10.1016/j.cbi.2010.05.011
- Nakano M., Onodera A., Saito E., Tanabe M., Yajima K., Takahashi J., Nguyen V.C. Effect of astaxanthin in combination with α-tocopherol or ascorbic acid against oxidative damage in diabetic ODS rats. J. Nutr. Sci. Vitaminol. 2008;54:329–334. doi: 10.3177/jnsv.54.329
- Nishigaki I., Rajendran P., Venugopal R., Ekambaram G., Sakthisekaran D., Nishigaki Y. Cytoprotective role of astaxanthin against glycated protein/iron chelate-induced toxicity in human umbilical vein endothelial cells. Phytother. Res. 2010;24:54–59. doi: 10.1002/ptr.2867
- Bhuvaneswari S., Yogalakshmi B., Sreeja S., Anuradha C.V. Astaxanthin reduces hepatic endoplasmic reticulum stress and nuclear factor-κB-mediated inflammation in high fructose and high fat diet-fed mice. Cell Stress Chaperones. 2013 in press
- Kim Y.J., Kim Y.A., Yokozawa T. Protection against oxidative stress, inflammation, and apoptosis of high glucose- exposed proximal tubular epithelial cells by astaxanthin. J. Agric. Food Chem. 2009;57:8793–8797. doi: 10.1021/jf9019745
- Marotta F, Pavasuthipaisit K, Yoshida C, et al. Relationship between aging and susceptibility of erythrocytes to oxidative damage: in view of nutraceutical interventions. Rejuvenation Res 2006;9:227-230.
- Nakagawa K, Kiko T, Miyazawa T, et al. Antioxidant effect of astaxanthin on phospholipid peroxidation in human erythrocytes. Br J Nutr 2011;105:1563-1571.
- Miyawaki H, Takahashi J, Tsukahara H, Takehara I. Effects of astaxanthin on human blood rheology. J Clin Biochem Nutr 2008;43:69-74.
- Ryu S.K., King T.J., Fujioka K., Pattison J., Pashkow F.J., Tsimikas S. Effect of an oral astaxanthin prodrug (CDX-085) on lipoprotein levels and progression of atherosclerosis in LDLR and ApoE mice. Atherosclerosis. 2012;222:99–105. doi: 10.1016/j.atherosclerosis.2012.02.002
- Wolf G. Retinoids and carotenoids as inhibitors of carcinogenesis and inducers of cell-cell communication. Nutr. Rev. 1992;50:270–274. doi: 10.1111/j.1753-4887.1992.tb01345.x
- Hix L.M., Lockwood S.F., Bertram J.S. Upregulation of connexin 43 protein expression and increased gap junctional communication by water soluble disodium disuccinate astaxanthin derivatives. Cancer Lett. 2006;211:25–37
- Zhang L.X., Cooney R.V., Bertram J.S. Carotenoids enhance gap junctional communication and inhibit lipid peroxidation in C3H/10T1/2 cells: relationship to their cancer chemopreventive action. Carcinogenesis. 1991;12:2109–2114. doi: 10.1093/carcin/12.11.2109
- Chew B.P., Park J.S. Carotenoid action on the immune response. J. Nutr. 2004;134:257S–261S.
- Palozza P., Torelli C., Boninsegna A., Simone R., Catalano A., Mele M.C., Picci N. Growth-inhibitory effects of the astaxanthin-rich alga Haematococcus pluvialis in human colon cancer cells. Cancer Lett. 2009;283:108–117. doi: 10.1016/j.canlet.2009.03.031
- Daubrawa F., Sies H., Stahl W. Astaxanthin diminishes gap junctional intercellular communication in primary human fibroblasts. J. Nutr. 2005;135:2507–2511
- Maoka T., Tokuda H., Suzuki N., Kato H., Etoh H. Anti-oxidative, anti-tumor-promoting, and anti-carcinogenesis activities of nitroastaxanthin and nitrolutein, the reaction products of astaxanthin and lutein with peroxynitrite. Mar. Drugs. 2012;10:1391–1399. doi: 10.3390/md10061391
- Satoh A, Tsuji S, Okada Y, et al. Preliminary clinical evaluation of toxicity and efficacy of a new astaxanthin-rich Haematococcus pluvialis extract. J Clin Biochem Nutr 2009;44:280-284.
- Zhang X, Pan L, Wei X, et al. Impact of astaxanthin-enriched algal powder of Haematococcus pluvialis on memory improvement in BALB/c mice. Environ Geochem Health 2007;29:483-489.
- Lu YP, Liu SY, Sun H, et al. Neuroprotective effect of astaxanthin on H(2)O(2)-induced neurotoxicity in vitro and on focal cerebral ischemia in vivo. Brain Res 2010;1360:40-48.
- Kim JH, Choi W, Lee JH, et al. Astaxanthin inhibits H2O2-mediated apoptotic cell death in mouse neural progenitor cells via modulation of P38 and MEK signaling pathways. J Microbiol Biotechnol 2009;19:1355-1363.
- Kim JH, Nam SW, Kim BW, et al. Astaxanthin improves the proliferative capacity as well as the osteogenic and adipogenic differentiation potential in neural stem cells. Food Chem Toxicol 2010;48:1741-1745.
- Kajita M, Tsukahara H, Kato M. The effects of a dietary supplement containing astaxanthin on the accommodation function of the eye in middle-aged and older people. Med Consult New Remedies 2009;46:89-93
- Thomson WD. Eye problems and visual display terminals – the facts and the fallacies. Ophthalmic Physiol Opt 1998;18:111-119.
- Nagaki Y, Hayasaka S, Yamada T, et al. Effects of astaxanthin on accommodation, critical flicker fusion, and pattern visual evoked potential in visual display terminal workers. J Tradit Med 2002;19:170-173.
- Malmsten CL, Lignell A. Dietary supplementation with astaxanthin-rich algal meal improves strength endurance – a double blind placebo controlled study on male students. Carotenoid Sci 2008;13:20-22.
- Baralic I, Djordjevic B, Kotur-Stevuljevic J, et al. Astaxanthin supplementation prevents muscle and oxidative damage induced by training in elite young soccer players. European Database of Sports Science (EDSS). 16th Annual ECSS-Congress, Liverpool 2011.
- Radivojevic N, Dikic N, Baralic I, et al. Effects of astaxanthin supplementation on sports performance in young elite soccer players. European Database of Sports Science (EDSS). 16th Annual ECSS-Congress, Liverpool 2011
- Comhaire FH, El Garem Y, Mahmoud A, et al. Combined conventional/antioxidant “Astaxanthin” treatment for male infertility: a double blind, randomized trial. Asian J Androl 2005;7:257-262.
- Jyonouchi H., Hill R., Tomita Y., Good R. Studies of immunomodulating actions of carotenoids. I. Effects of β-carotene and astaxanthin on murine lymphocyte functions and cell surface marker expression in in vitro culture system. Nutr. Cancer. 1991;16:93–105. doi: 10.1080/01635589109514148
- Jyonouchi H., Sun S., Gross M. Effect of carotenoids on in vitro immunoglobulin production by human peripheral blood mononuclear cells: astaxanthin, a carotenoid without vitamin A activity, enhances in vitro immunoglobulin production in response to a T-dependent stimulant and antigen. Nutr. Cancer. 1995;23:171–183. doi: 10.1080/01635589509514373
- Park J.S., Chyun J.H., Kim Y.K., Line L.L., Chew B.P. Astaxanthin decreased oxidative stress and inflammation and enhanced immune response in humans. Nutr. Metab. 2010;7:1–10
- Osterlie M, Bjerkeng B, Liaaen-Jensen S. Plasma appearance and distribution of astaxanthin E/Z and R/S isomers in plasma lipoproteins of men after single dose administration of astaxanthin. J Nutr Biochem 2000;11:482-490.
- EFSA (European Food Safety Authority) Opinion of the scientific panel on additives and products or substances used in animal feed on the request from the European commission on the safety of use of colouring agents in animal human nutrition. EFSA J. 2005;291:1–40.
- Iwamoto T., Hosoda K., Hirano R., Kurata H., Matsumoto A., Miki W., Kamiyama M., Itakura H., Yamamoto S., Kondo K. Inhibition of low-density lipoprotein oxidation by astaxanthin. J. Atheroscler. Thromb. 2000;7:216–222. https://www.ncbi.nlm.nih.gov/pubmed/11521685
- Ranga Rao A., Baskaran V., Sarada R., Ravishankar G.A. In vivo bioavailability and antioxidant activity of carotenoids from micro algal biomass—A repeated dose study. Food Res. Int. 2013;54:711–717. doi: 10.1016/j.foodres.2013.07.067.
- Reagan-Shaw S, Nihal M, Ahmad N. Dose translation from animal to human studies revisited. FASEB J 2008;22:659-661
- Scientific Opinion on the safety of astaxanthin‐rich ingredients (AstaREAL A1010 and AstaREAL L10) as novel food ingredients. https://efsa.onlinelibrary.wiley.com/doi/epdf/10.2903/j.efsa.2014.3757