What is auscultation
Auscultation is a method used to listen to the sounds of your body during a physical examination by using a stethoscope. A patient’s lungs, heart, and intestines are the most common organs heard during auscultation. The stethoscope is an instrument that does not significantly amplify sound, but, more important, acts as a selective filter of sound. Briefly, the bell filters high-frequency sounds greater than 1500 cycles per second and therefore should be used to detect low-frequency sounds 1. On the other hand, the diaphragm selectively filters low-frequency sounds 1.
Health care providers routinely listen to a person’s lungs, heart, and intestines to evaluate these things about the sounds:
- Frequency
- Intensity
- Duration
- Number
- Quality
Providers also use auscultation to listen to the heart sounds of unborn infants. This can be done with a stethoscope or with sound waves (called Doppler ultrasound).
Auscultation can also be used to hear pulses in the arms and legs.
Lung auscultation
Auscultation of the chest is part of every chest examination but it is the data collected during inspection, palpation, and percussion that alert the clinician what to listen for during auscultation in order to identify the correct diagnosis most effectively 1.
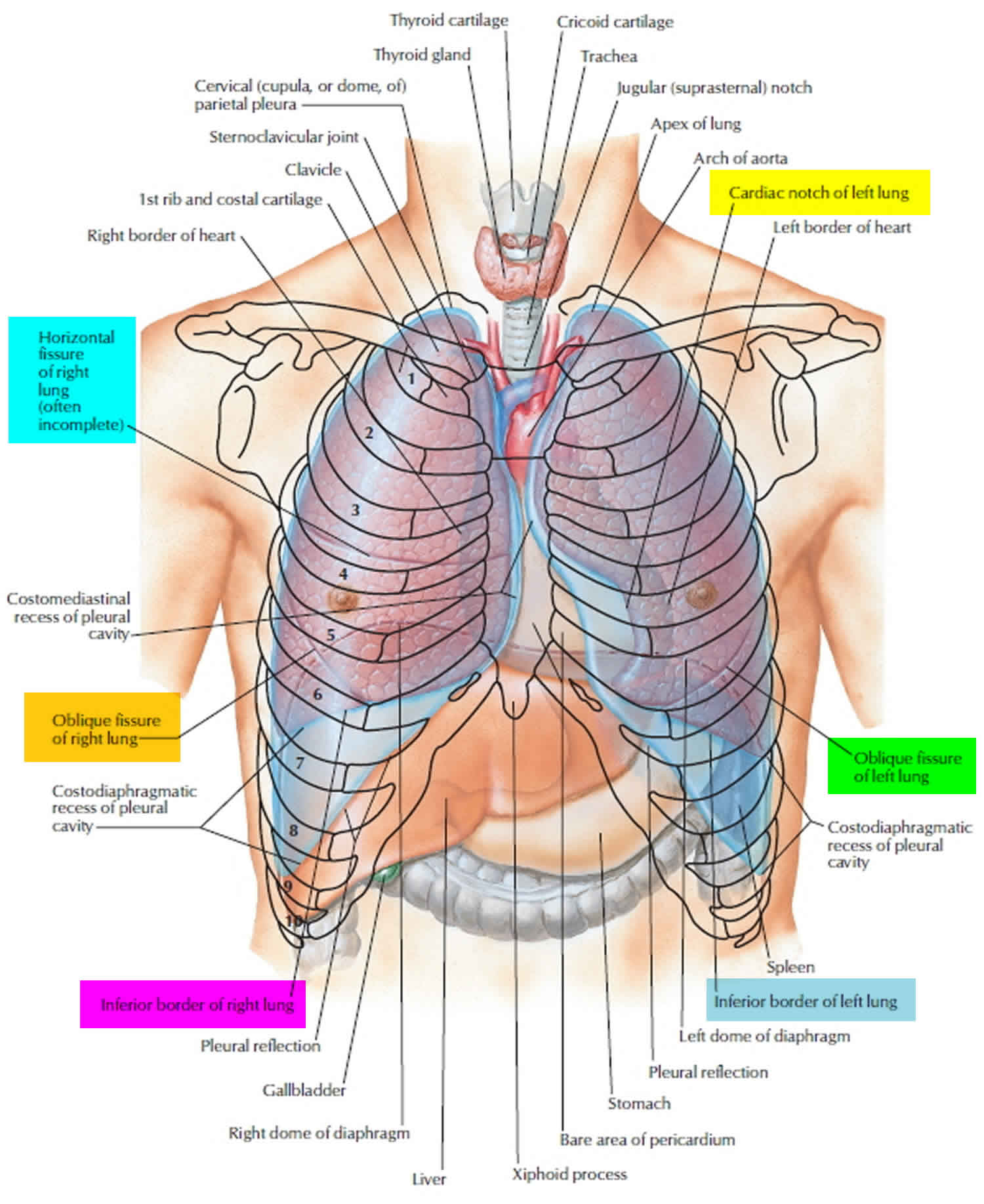
Auscultation of the lungs should be systematic and follow a stepwise approach in which the examiner surveys all the lung zones. For practical purposes, the lung can be divided into apical, middle and basilar regions during auscultation 2. The description of abnormal breathing sounds should be tagged with the location in which it was heard.
Methods of performing chest auscultation 3
- Auscultation should be done in a quiet room with the patient either sitting or standing, preferably in a sitting position. If the patient cannot assume sitting posture, roll the patient from one side to the other to examine the back.
- Always warm up the cold stethoscope by rubbing the chest piece in your hands before placing it on naked body. Auscultation should never be done through the clothing.
- Since sounds produced by breathing tend to be of relatively high pitch, the chest is ausculted with the diaphragm of the stethoscope.
- Ask the patient to take deep breaths through the open mouth.
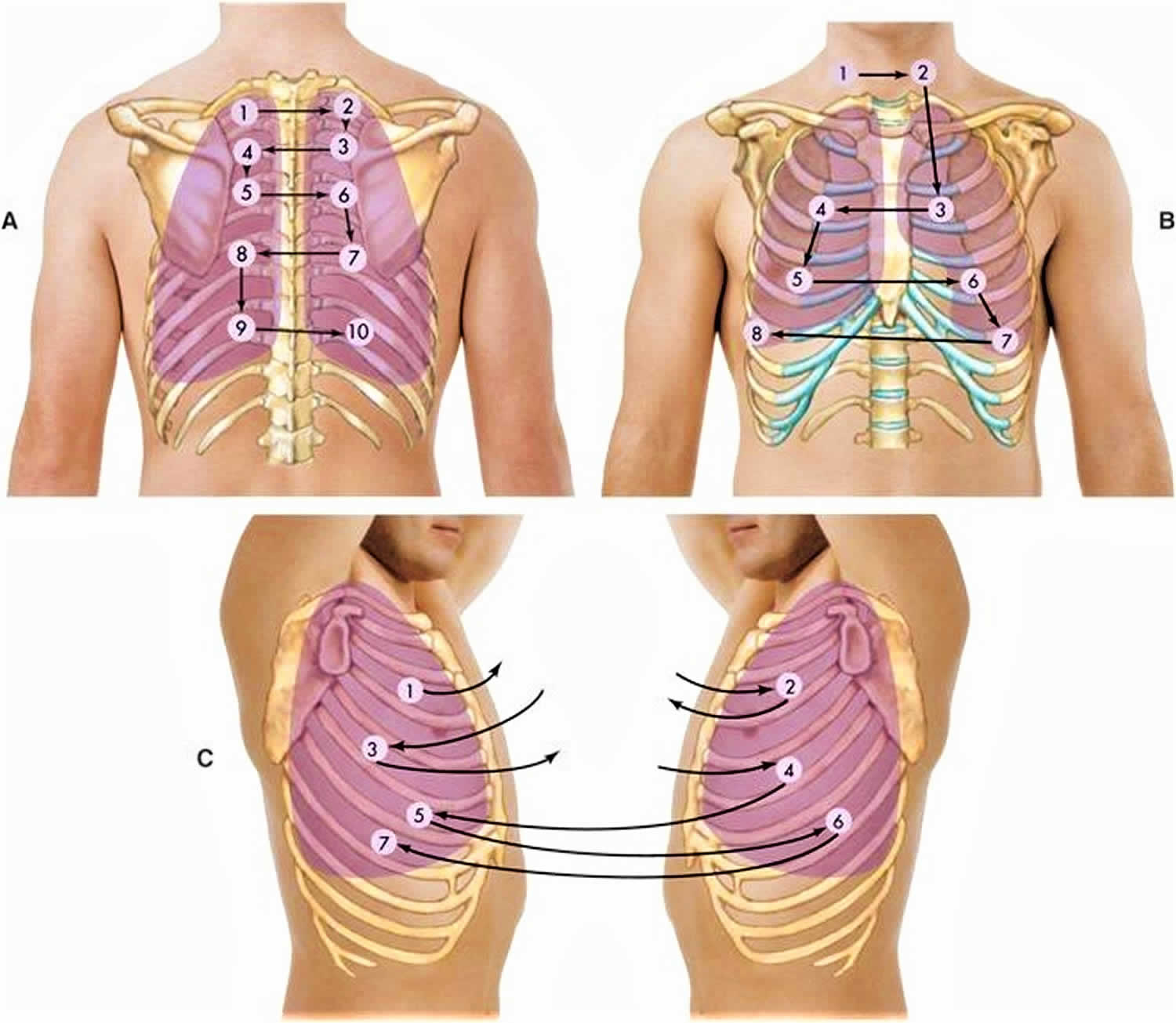
- Using the diaphragm of the stethoscope, start auscultation anteriorly at the apices, and move downward till no breath sound is appreciated. Next, listen to the back, starting at the apices and moving downward. When the posterior thorax is examined, the patient’s arms should be crossed anteriorly to move the scapulas laterally as much as possible. At least one complete respiratory cycle should be heard at each site.
- Always compare symmetrical points on each side. Comparing one side to the other is a helpful maneuver to identify the patient’s “normal.”
- Auscultation should be performed during tidal ventilation, deep forceful inspiration, and forceful expiration. It is not only intuitively obvious but rigorously proved that the intensity of breath sounds is related to flow rates; that is, the louder the sound, the greater the flow rate, all other things being equal.
- Listen for the quality of the breath sounds, the intensity of the breath sounds, and the presence of adventitious sounds.
Figure 1. Lung anatomy
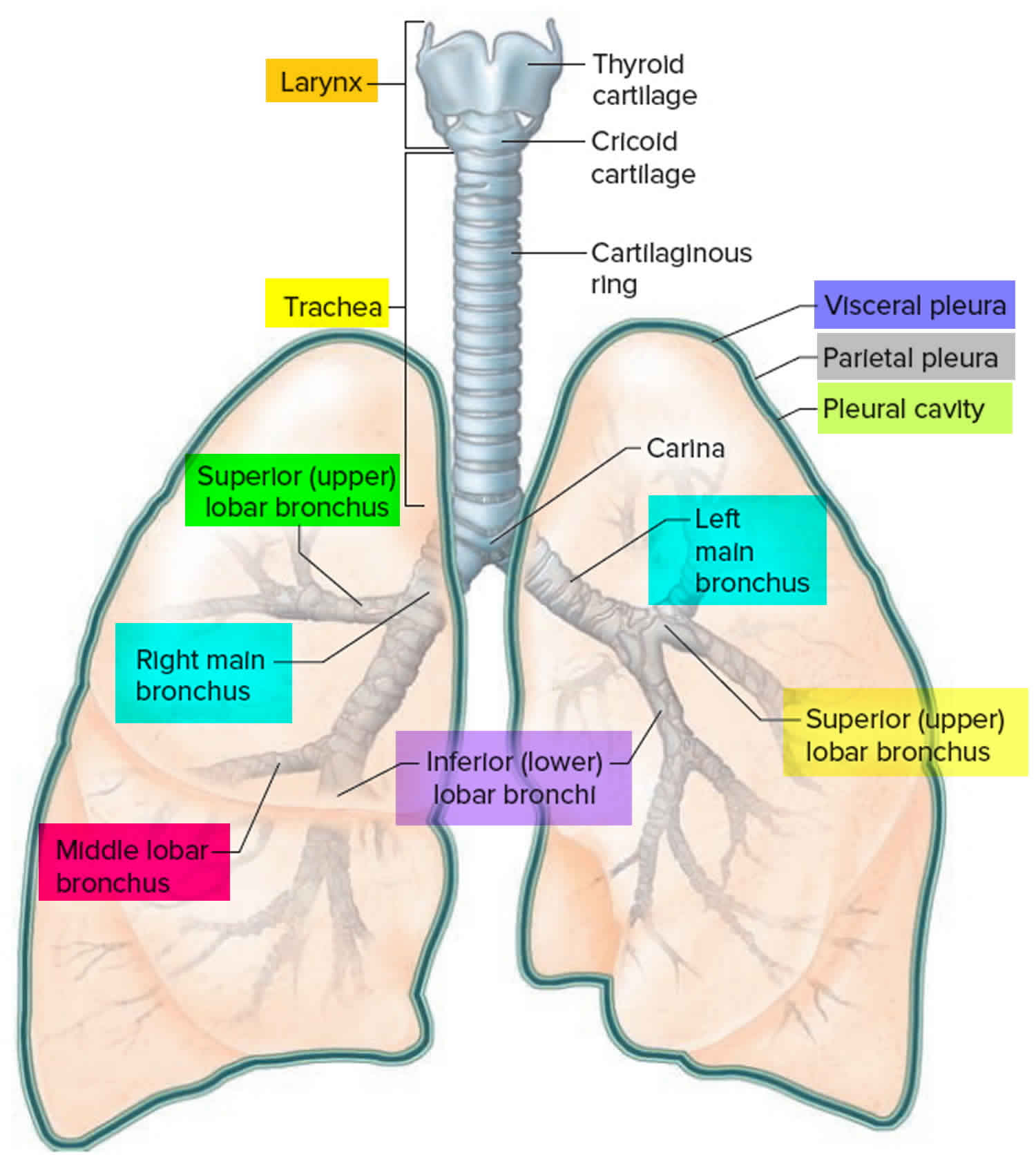
Figure 2. Bronchial tree of the lungs
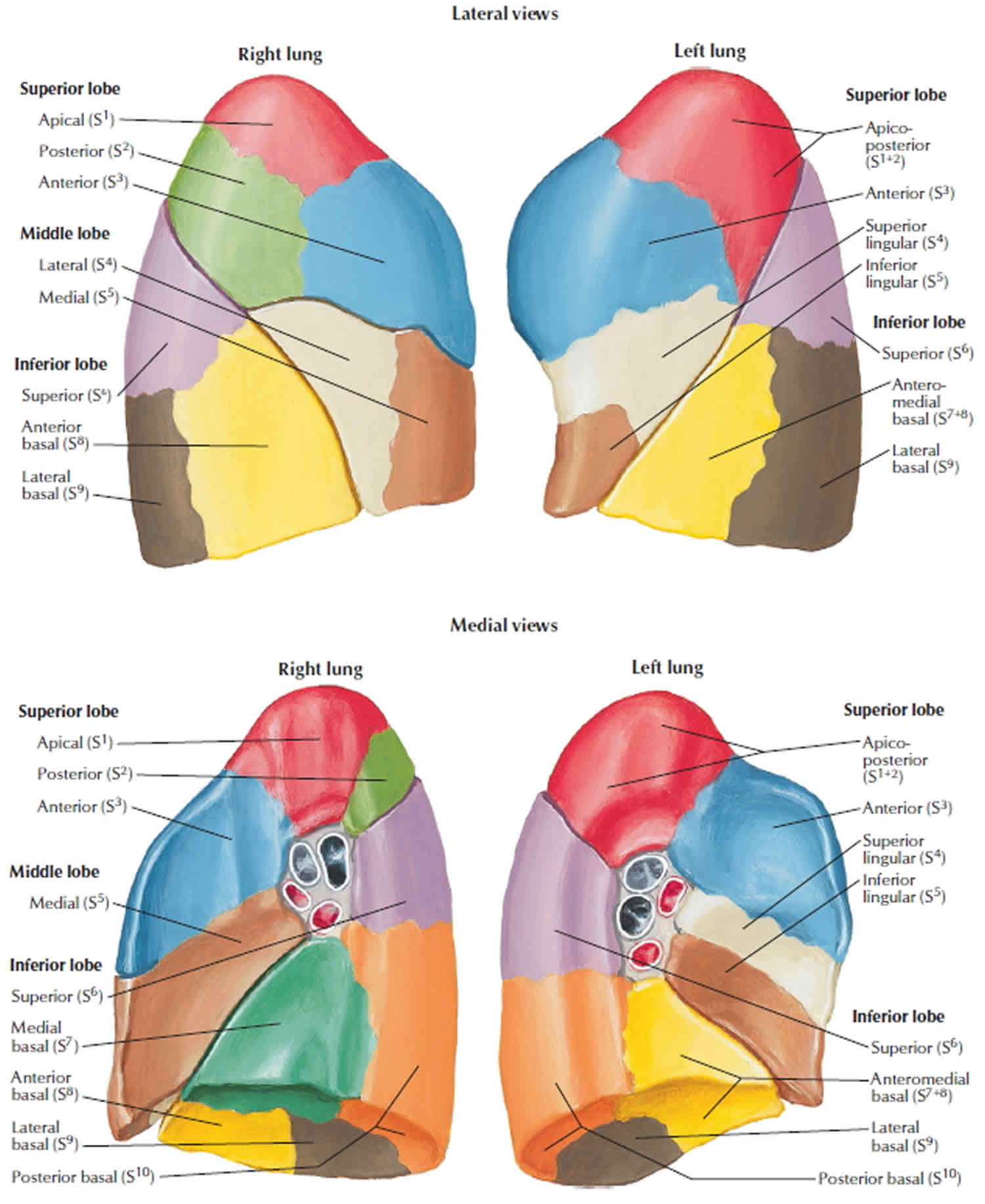
Figure 3. Lungs bronchopulmonary segments
Figure 4. Lung auscultation points
The movement of air generates normal breath sounds through the large and small airways. Normal breath sounds have a frequency of approximately 100 Hz 2. The absence of breath sounds should prompt the health care provider to consider shallow breath, abnormal anatomy or pathologic entities such as airway obstruction, bulla, hyperinflation, pneumothorax, pleural effusion or thickening, and obesity.
Tubular breath sounds are high pitched, bronchial breath sounds, seen in the following conditions: consolidation, pleural effusion, pulmonary fibrosis, distal collapse, and mediastinal tumor over a large patent bronchus.
Bronchial breath sounds may be either normal or abnormal. When they are heard on the periphery, where vesicular breath sounds are normally heard, one can imply that the airways to the lung units are open but that the lung units themselves are filled with liquid-like material. When this occurs without pleural fluid, the bronchial breath sounds are loud; when consolidation is associated with a pleural effusion, the bronchial breath sounds are present but often quite decreased in intensity. Confirmation of the presence of bronchial breath sounds can be obtained by listening for egophony (“E to A” sound). This sound is elicited by asking the patient to say the letter “E” as one listens over the suspicious area with the stethoscope. When consolidation is present, the spoken “E” sound is converted to an ausculted “A” sound, similar to that produced by a bleating goat.
In addition to assessing the quality of breath sounds, it is also important to assess the duration of the expiratory phase. Timing the duration of expiratory sound while listening with the diaphragm over the trachea during a forced expiratory volume maneuver is used to identify airways obstruction. Expiratory sound should terminate within 6 seconds. If the sound is prolonged, airways obstruction manifested by an FEV1 of less than 1.5 liters can be assumed.
Auscultatory wheezes imply the presence of slitlike openings through which a critical velocity of gas is passing. When wheezes are local, one must consider external compression of an airway. Enlarged lymph nodes and tumors do this. A lesion within the airway, such as an endobronchial malignancy or foreign body, also can produce a localized wheeze. Diffuse wheezing is present in inflammatory processes such as bronchitis (both acute or chronic), contraction of hypertrophied bronchial smooth muscle as seen in asthma, inspissated thick secretions of pneumonia, and airway collapse associated with the dynamic compression of pulmonary emphysema.
Crackles imply the snapping open of airways or alveoli. Since larger airways open first as inhalation progresses from residual volume, early inspiratory crackles imply large airways disease while late inspiratory crackles either mean small airways problems (less than 2 mm) or poorly compliant alveoli walls such as seen in congestive heart failure, pulmonary fibrosis, or other interstitial pulmonary processes.
Gurgles suggest fluid in the airways. This may be produced by excessive serous secretion in alveolar cell carcinoma, infected purulent secretion of acute or chronic bronchitis or bronchiectasis, or transudated fluid entering the airways from the alveoli as occurs in pulmonary edema.
Vesicular breath sounds/normal breath sounds
While René Théophile Hyacinthe Laënnec (inventor of the stethoscope) 4 considered normal lung sounds to originate from the flow of air in and out of alveoli, later investigations of the origin of respiratory sounds have not shown lung “vesicles” to participate in sound generation. Therefore, vesicular breath sounds is a misnomer for normal breath sounds.
Wheezes
High-pitched continuous sounds with a dominant frequency of 400 Hz or more. Suggestive of asthma, chronic obstructive pulmonary disease (COPD), airway obstruction, or mucus plug.
Ronchi
Low-pitched continuous musical sounds with a dominant frequency of about 200 Hz or less.
Crackles
A “popping” sound generated by the passage of air through the accumulated secretions within the large and medium-size airways, creating the bubbling sounds (brief, non-musical, “discontinuous” sounds). Seen in chronic obstructive pulmonary disease (COPD), Pneumonia and Heart Failure.
Pleural Rub
Occurs due to inflamed pleural surface rubbing each other during breathing. It is difficult to differentiate from fine crackles, but the sound is similar to rubbing your stethoscope against cotton.
Stridor
A loud, high-pitched, musical sound produced by upper respiratory tract obstruction. It indicates an extrathoracic upper airway obstruction (supraglottic lesions like laryngomalacia, vocal cord lesion) when heard on inspiration. It occurs in expiration if associated with intrathoracic tracheobronchial lesions (tracheomalacia, bronchomalacia, and extrinsic compression). It occurs in both phases if a lesion is fixed, for example, stenosis.
Special Maneuvers
- Pectoriloquy – Ask the patient to whisper a word such as “one-two-three” or “ninety-nine” and listen with a stethoscope. Typically, words are heard faintly. In cases of consolidation, the whispered sounds will be heard clearly and distinctly.
- Egophony is elicited by asking the patient to say “Ee,” and it will sound like an “A.” Suggestive of consolidation or pleural effusion.
Heart auscultation
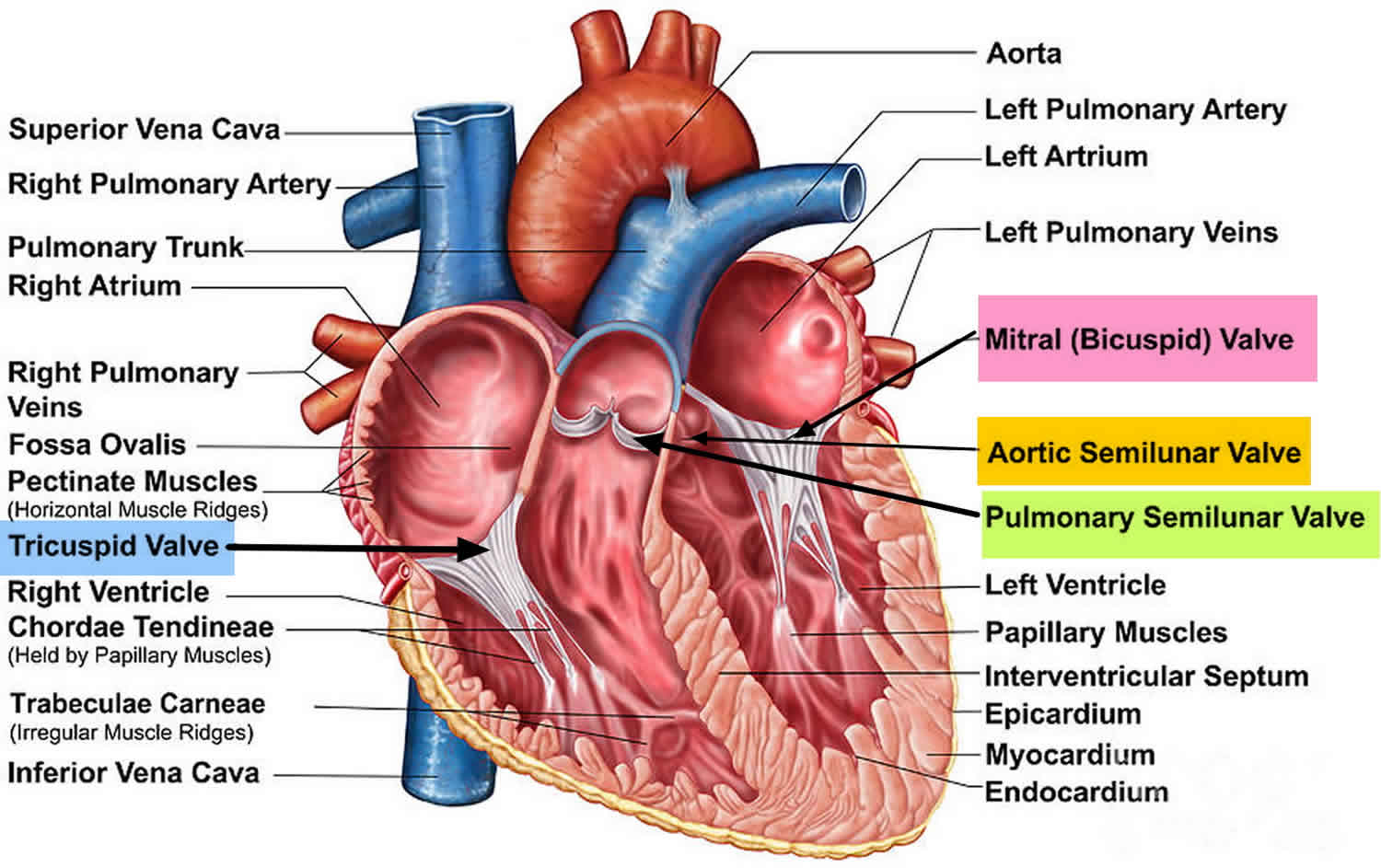
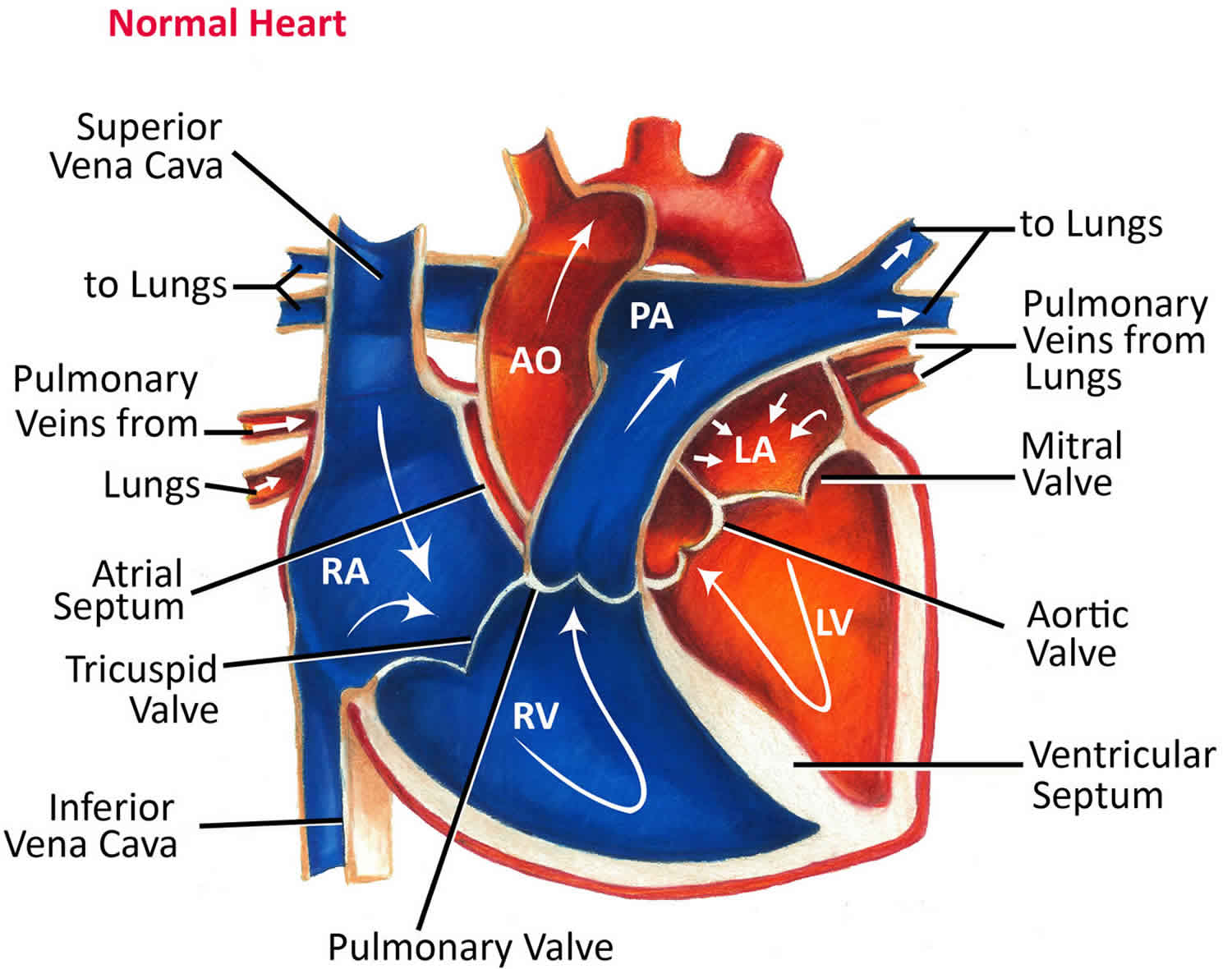
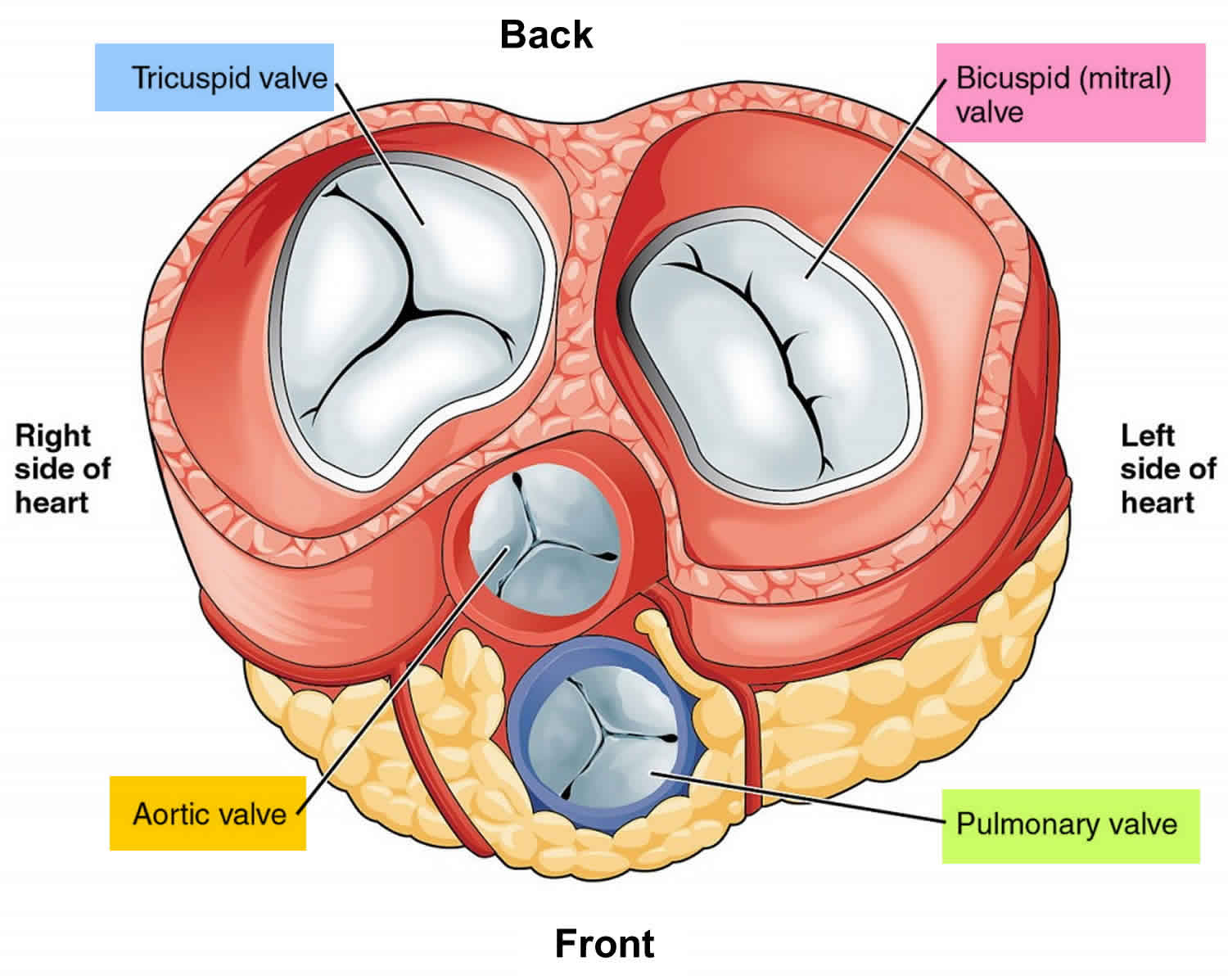
Heart sounds are discrete bursts of auditory vibrations of varying intensity (loudness), frequency (pitch), quality, and duration. There are two normal heart sounds that should be elicited in auscultation: S1 (lub) and S2 (dub). The first heart sound (S1) is composed of several high-frequency components; only the first two are normally audible. These two sets of audible vibrations are temporally related to closure of the mitral and tricuspid valves 5. The first heart sound (S1) coincides with contraction of the ventricles, thus identifying the onset of ventricular systole and the end of mechanical diastole.
Auscultation of the heart and great vessels should take place in a warm, quiet room with the patient’s chest exposed. The clinician should use a stethoscope with plastic or rubber tubing 25 to 30 cm (10 to 12 in) long. The stethoscope should be equipped with a stiff diaphragm and a shallow bell. The diaphragm of the stethoscope is used to identify high-pitched sounds, while the bell is used to identify low-pitched sounds.
The patient should be examined in the recumbent, sitting, and left lateral decubitus positions. The principal areas of interest are the primary aortic area (second and third intercostal space at the left sternal border), the tricuspid area (fourth intercostal space at the left sternal border), and the mitral area (cardiac apex) 6. Levine 7 and Harvey 8 have recommended that auscultation begin at the cardiac apex, then proceed along the left sternal border from the tricuspid area to the pulmonic area and finally to the aortic area. The clinician should also auscultate the right parasternal region, the right and left base of the neck, the right and left carotid arteries, the left axilla, and the interscapular area. These are areas to which systolic heart murmurs may radiate or from which extracardiac sounds simulating systolic heart murmurs may emanate. The clinician should alternate use of the diaphragm and bell at each location. The clinician should also listen for any additional sounds such as clicks, and heart murmurs. Before attempting to detect and characterize a heart murmur, the clinician should define the first and second heart sounds in order to locate systole accurately.
Surface anatomy of the heart
The heart rests on the diaphragm and is covered anteriorly by the body of the sternum and the 3rd-6th costal cartilages of both sides. More specifically, the upper limit of the heart reaches as high as the 3rd costal cartilage on the right side of the sternum and the 2nd intercostal space on the left side of the sternum (both 1.2cm from the sternal border). The right margin of the heart extends from the right 3rd costal cartilage to near the right 6th costal cartilage (at the 6th chondro-sternal junction). The left margin of the heart descends laterally from the 2nd intercostal space to the apex located near the midclavicular line in the 5th intercostal space. The lower margin of the heart extends from the sternal end of the right 6th costal cartilage to the apex in the 5th intercostal space near the midclavicular line (or 9cm from the midline).
The right border of the heart is formed by the right atrium. The vena cavae enter the heart at the upper and lower ends of the borders. The lower border of the heart is mainly the right ventricle but includes some left ventricle at the apex. It lies at the end of the xiphisternal joint. The lower 4/5 of the left border of the heart is formed by the left ventricle and the upper 1/5 by the left atrium. The upper border is formed by both atria. The superior vena cava enters the heart at its right end, the ascending aorta crosses the middle of the border, the pulmonary trunk bifurcates just above the left end, and the right pulmonary artery runs to the right just above the border.
The chambers of the heart can me marked out by drawing (see Figure 8):
- The coronary sulcus: separating the atria and the ventricles from the upper medial end of the 3rd left costal cartilage to the middle of the right 6th chondro-sternal joint.
- The anterior interventricular sulcus: from the 3rd left intercostal space 2.5cm to the left of the midline to a point 1.2cm medial to the apex.
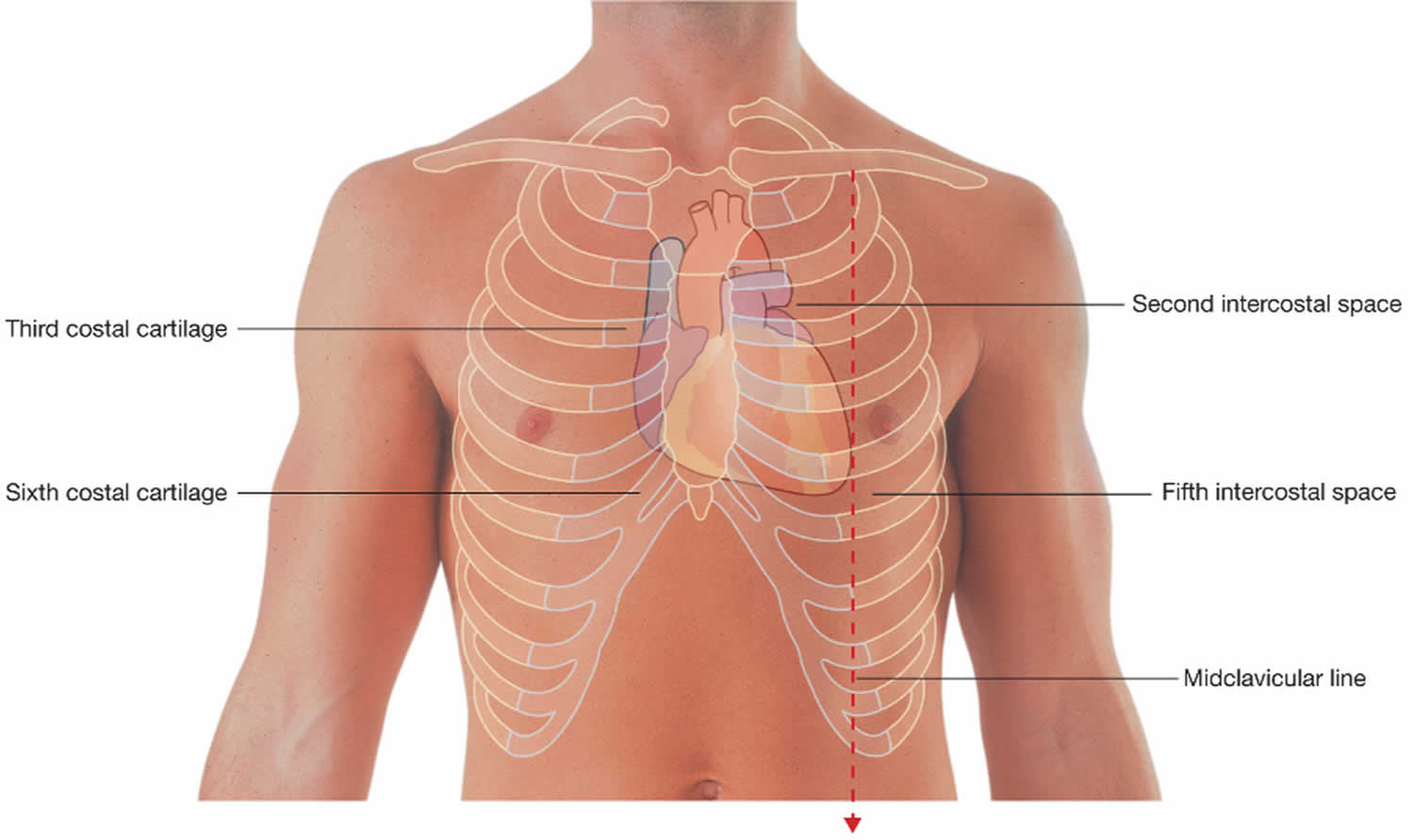
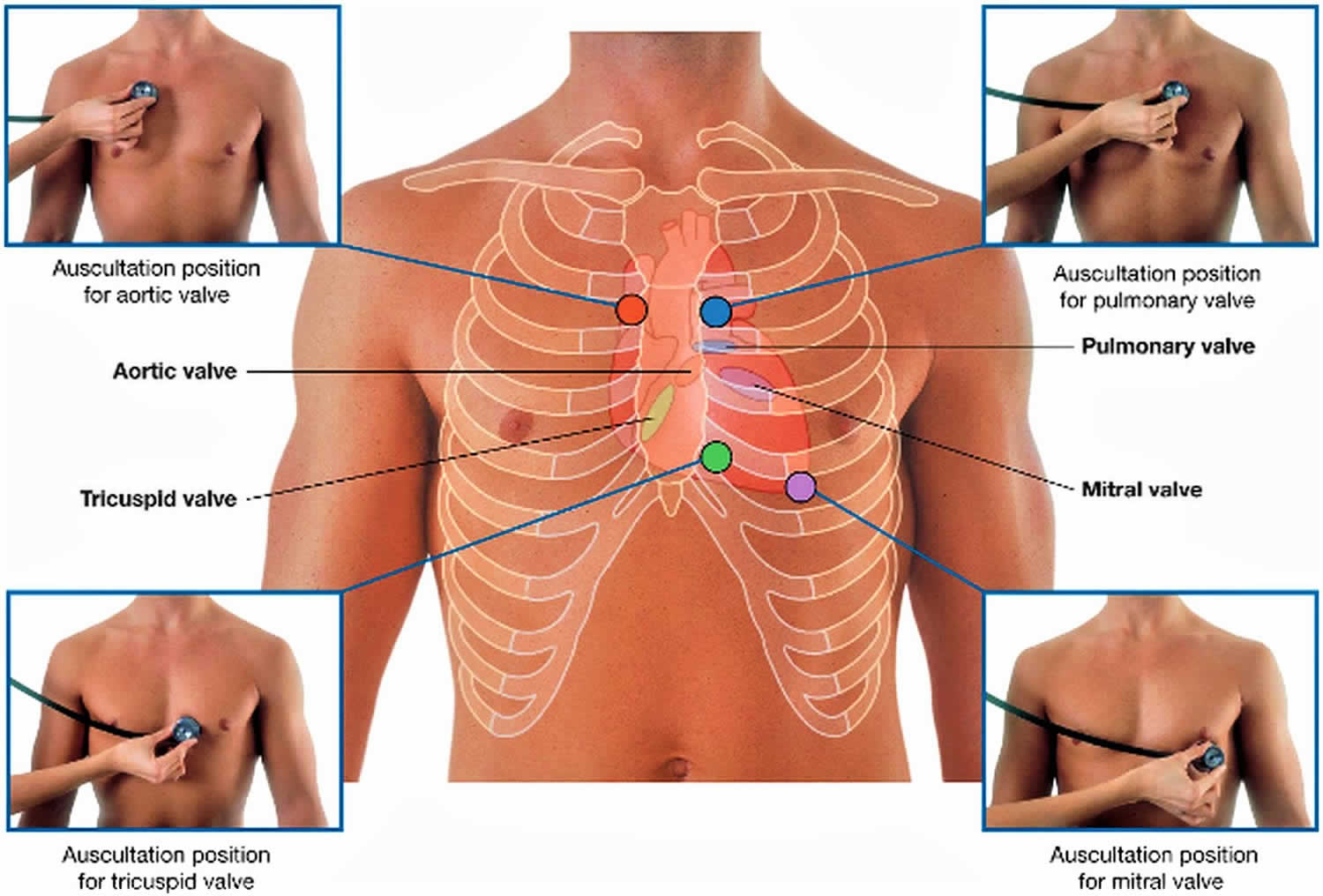
5 areas of the heart for auscultation (see Figure 9):
- Aortic Valve Area: Second right intercostal space, right sternal border
- Pulmonic Valve Area: Second left intercostal space, left sternal border
- Erb’s Point: Third left intercostal space, left sternal border
- Tricuspid Valve Area: Fouth left intercostal space, left sternal border
- Mitral Valve Area: Fifth left intercostal space, left mid-clavicular line
To listen for valve sounds, you should position your stethoscope downstream from the flow of blood through the valves. The tricuspid valve is almost vertical and centered at the 4th intercostal space just to the right of the midline. It can be heard just to the left of the lower part of the sternum near the 5th intercostal space. The mitral valve is oblique, running down and right, starting opposite the 4th costal cartilages and lying beneath the left side of the sternum. It can be heard over the apex of the heart in the left 5th intercostal space at the midclavicular line. The pulmonary valve is horizontal and centered at the 3rd left chondro-sternal joint. It is heard over the medial end of the left 2nd intercostal space. The aortic valve is oblique, running down and right, starting from the medial end of the 3rd left intercostal space. It can be heard over the medial end of the right 2nd intercostal space.
The physician listens to the heart sounds, rate, and rhythm. If murmurs are detected during this examination, it can be determined if the murmur takes place in systole (between S1 and S2) or diastole (after S2 and before the next S1). Determining the timing of murmurs can help narrow down an otherwise broad differential diagnosis. For example, a murmur determined to be systolic rules out diastolic murmur causes such as mitral stenosis and aortic regurgitation. As a general rule, systolic murmurs involve blood leaving the ventricle (e.g., mitral regurgitation, aortic stenosis, hypertrophic cardiomyopathy [HOCM]) and diastolic murmurs involve blood entering the ventricles (mitral stenosis, aortic regurgitation).
The symptomatology of various cardiac pathology may be evident on physical examination by methods other than auscultation. Palpation and simple observation can reveal alterations to the normal pressure-contraction cycling of heart. Palpation of the distal extremities for temperature and the presence of edema is a rapid bedside maneuver. One can also inspect the neck for jugular venous distention. Because the jugular veins are continuous with the right atria, their distention can be used as a rough estimation of the right atrial pressure. An elevated right atrial pressure can correlate to pathologies such as arrhythmia or poor cardiac output. One striking example occurs in congestive heart failure, where poor forward flow causes a backup into preceding cardiac chambers.
Figure 5. Heart anatomy
Figure 6. Normal heart blood flow
Figure 7. Top view of the 4 heart valves
Figure 9. Heart auscultation locations
Heart valve disease
Disease of the heart valves remains an important cause of morbidity and mortality across the world. While advances in echocardiography and the widespread availability of antibiotics have changed the prevalence, management, and especially the diagnosis of valve disease for specialists, very little has changed for generalists, who hear heart murmurs less frequently. Even though echo diagnosis is not readily available to generalists, they nevertheless feel pressured to identify murmurs and to report them, with their associated signs, in the referral letter. Similarly, for many examiners, the appeal of obscure murmurs for clinical short-case exams remains too great to resist, despite its mostly historical relevance. Yet, there are few greater pleasures in clinical medicine than having your stethoscopic diagnostic brilliance confirmed by an echo report. Furthermore, healthcare economics has prompted a renewed interest in the power of the stethoscope for diagnosing and even quantifying valve disease.
Table 1. Grading of heart murmurs
| Grade | Heart murmur |
|---|---|
| 1 | Just audible in a quiet room |
| 2 | Quiet |
| 3 | Loud, no thrill |
| 4 | Loud, with thrill |
| 5 | Very loud with thrill |
| 6 | Audible without a stethoscope |
Systolic murmurs
Systolic murmurs may be classified as ejection murmurs, regurgitant murmurs, or extracardiac sounds that simulate systolic heart murmurs 6. Ejection murmurs emanate from the semilunar valves or surrounding structures (i.e., the aortic or pulmonic root). Regurgitant murmurs are created when blood flows from a high-pressure “donor” chamber to a low-pressure “recipient” chamber. Table 2 is a summary of systolic” ejection and regurgitant heart murmurs and extracardiac sounds.
Systolic murmurs may be further subclassified as functional or organic. Functional systolic murmurs occur in the absence of cardiac structural abnormalities. They are frequently encountered in healthy individuals, but may also accompany a variety of high cardiac output states. Organic systolic murmurs evolve from structural abnormalities in the heart or great vessels. Systolic ejection murmurs may be functional or organic, but systolic regurgitant murmurs indicate organic heart disease.
Table 2. Differential Diagnosis of Systolic Murmurs 6
- Ejection murmurs
- Functional
- Still’s murmur and its adult variant
- Flow murmur emanating from the root of the pulmonary artery
- Murmur associated with high cardiac output states
- Flow murmurs associated with aortic or pulmonary valvular insufficiency
- Organic
- Valvular aortic stenosis
- Aortic sclerosis
- Discrete subvalvular aortic stenosis (web or tunnel)
- Supravalvular aortic stenosis
- Hypertrophic obstructive cardiomyopathy
- Pulmonary valvular stenosis
- Pulmonary infundibular stenosis
- Atrial septal defect
- Tetralogy of Fallot
- Functional
- Regurgitant murmurs
- Functional: none
- Organic
- Mitral regurgitation
- Rheumatic
- Papillary muscle dysfunction
- Mitral valve prolapse
- Acute
- Tricuspid regurgitation
- Chronic
- Acute
- Ventricular septal defect
- Roger’s type (small and large)
- Without pulmonary hypertension
- With pulmonary hypertension
- Slitlike
- Roger’s type (small and large)
- Mitral regurgitation
- Extracardiac sounds simulating systolic heart murmurs
- Subclavian (supraclavicular/brachiocephalic) murmur
- Internal mammary soufflé
- Carotid artery bruits
- Coarctation of the aorta
- Murmurs emanating from a dilated aortic or pulmonary artery root
- Patent ductus arteriosus with pulmonary hypertension
Ejection Murmurs
Functional systolic ejection murmurs include pulmonic flow murmurs in patients with either normal or increased pulmonary artery or aortic flow. The most common functional systolic ejection murmur in adults is probably a variant of Still’s murmur, the so-called innocent murmur of childhood. It is a short, buzzing, pure, medium-pitched, nonradiating, midsystolic murmur heard best along the upper left sternal border. It is thought to result from vibrations set in motion by the pulmonic valve. A less frequently encountered functional pulmonic flow murmur, occurring predominantly in children and adolescents, emanates from the root of the pulmonary artery. It is midsystolic and similar in location, but less uniform than Still’s murmur. It is high pitched, often blowing in nature, and is similar to the flow murmur heard in patients with the “straight-back syndrome.” Functional systolic ejection murmurs may also result from hyperdynamic blood flow over a normal pulmonic or aortic valve. Such murmurs are commonly associated with high cardiac output states such as thyrotoxicosis, anemia, infection, fever, arteriovenous fistula, beriberi, the hyperkinetic heart syndrome, or pregnancy. They may also be audible after exercise or with anxiety.
Systolic flow murmurs associated with aortic or pulmonic valvular insufficiency may also be included in this category. These murmurs are typically midsystolic. They begin in early systole, peak in early to midsystole, end well before the onset of the second heart sound, have variable intensity, and do not radiate. These murmurs are usually medium pitched and have a crescendo–decrescendo configuration. Although the vibrations that produce these murmurs emanate from both semilumar valves, the murmurs are best (often exclusively) heard at the pulmonic area of the precordium because of the proximity of the pulmonic valve to the chest wall.
The intensity of functional systolic ejection murmurs varies, but generally ranges from grades 1 to 3. The intensity typically increases during phase 3 (release phase) of the Valsalva maneuver, following a post-extrasystolic pause and after inhalation of amyl nitrite. The intensity may decrease with isometric hand grip exercise or intravenous administration of alpha-adrenergic agonists.
Organic systolic ejection murmurs include those associated with valvular aortic stenosis, aortic sclerosis, supravalvular aortic stenosis, subvalvular aortic stenosis, hypertrophic obstructive cardiomyopathy, valvular pulmonic stenosis, pulmonary infundibular stenosis, atrial septal defect, and tetralogy of Fallot.
The murmur of valvular aortic stenosis typically has a crescendo–decrescendo configuration and a low to medium pitch. The murmur is commonly described as harsh, rasping, grunting or rough. Best heard over the primary and secondary aortic area, the murmur is transmitted widely over the precordium and radiates to the carotid arteries. The intensity of the murmur is variable (usually grade 2, 3, or 4), and increases with passive leg raising, sudden squatting, 5 or 6 beats into phase 3 of the Valsalva maneuver (release phase), following a post-extrasystolic pause and after amyl nitrite administration. The murmur tends to fade during phase 2 of the Valsalva maneuver (strain phase), with isometric hand grip exercise and occasionally with intravenous administration of alpha-adrenergic agonists. In elderly persons the murmur may be more intense and high pitched over the mitral area. It commonly takes on a musical or wheezing quality and may be confused with mitral valve regurgitation. The crescendo–decrescendo configuration helps to differentiate aortic stenosis from mitral valve regurgitation. The murmur may peak in either mid or late systole. There is a tendency for the murmur to peak progressively later in systole as stenosis become more severe. Other signs that suggest severity of valvular aortic stenosis include a delayed carotid upstroke (the single best criterion on physical examination), diminished intensity or paradoxical splitting of the second heart sound, and left ventricular hypertrophy. The presence of a systolic ejection click suggests that valve mobility is reasonably well preserved: it mitigates against severe stenosis.
Aortic sclerosis results from degeneration and calcification of the aortic cusps, predominantly at their base. This abnormality produces a murmur that is identical in character to aortic valvular stenosis but does not result in a pressure gradient over the aortic valve. The murmur typically peaks in midsystole and is accompanied by a normal second heart sound and carotid pulse upstroke. The responses to physiologic and pharmacologic interventions are identical to those of functional systolic ejection murmurs. Echocardiographic studies suggest that aortic sclerosis is common in the elderly and may be the single most common cause of systolic murmur in this population.
Discrete subaortic stenosis, resulting from a fibrous web or fibromuscular tunnel, produces a murmur that is indistinguishable from aortic stenosis. An ejection click is typically absent. The absence of calcium and poststenotic dilatation of the aorta on chest x-ray or fluoroscopic examination of a patient with apparently “severe aortic stenosis” suggests the diagnosis.
Supravalvular aortic stenosis also produces a murmur indistinguishable from valvular aortic stenosis. The murmur is typically most intense over the primary aortic area and radiates to both carotid arteries, but the amplitude of the carotid and brachial pulses may be unequal (greater on the right side) due to the orientation of the jet of blood that traverses the area of stenosis. An ejection click is typically absent. Supravalvular aortic stenosis usually becomes apparent early in childhood, and may be associated with mental retardation and elfin facies.
The systolic murmur associated with hypertrophic obstructive cardiomyopathy (HOCM), also known as idiopathic hypertrophic subaortic stenosis, results from dynamic left ventricular outflow tract obstruction. Left ventricular outflow obstruction is probably related to systolic anterior motion of the anterior mitral valve leaflet caused by a Venturi (suction) effect in the outflow tract resulting from rapid ejection of blood. Recent evidence suggests that concurrent mitral valve regurgitation may contribute to the murmur in some cases. The murmur of hypertrophic obstructive cardiomyopathy (HOCM) is typically medium pitched, has a crescendo–decrescendo configuration, and is heard best along the left sternal border. There is radiation to the base of the neck, but not into the carotid arteries. The murmur is similar in quality to that of aortic valve stenosis, but is somewhat less harsh and slightly higher pitched. The intensity of the murmur is quite variable. The murmur of hypertrophic obstructive cardiomyopathy (HOCM) becomes louder during phase 2 of Valsalva’s maneuver (strain phase), with standing, after a post-extrasystolic pause, and following amyl nitrite inhalation. All of these maneuvers or interventions reduce left ventricular volume, increasing the subaortic pressure gradient. The murmur decreases in intensity with recumbency, passive leg raising, squatting, isometric hand grip, or following intravenous administration of alpha-adrenergic agonists. The murmur of hypertrophic obstructive cardiomyopathy (HOCM) is commonly accompanied by a fourth heart sound, a double or triple apical impulse, and a bisferiens (spike and dome) carotid pulse. A thrill is occasionally palpable over the left sternal border.
Valvular pulmonic stenosis is usually congenital in origin. Severe cases are apt to be encountered more in childhood than adulthood. The murmur of valvular pulmonic stenosis is midsystolic (with respect to right heart events). It begins well after the first heart sound and ends before the pulmonic component of the second heart sound (which may be delayed in severe cases), but often extend through the aortic component of the second heart sound. The murmur has a crescendo–decrescendo configuration. There is a strong tendency for the murmur to peak later in systole as stenosis becomes more severe. The murmur is best heard over the second intercostal space at the left sternal border and does not radiate. Its quality is similar to that of valvular aortic stenosis. The intensitv of the murmur is variable but is generally grade 3 or higher; it increases during phase 3 of the Valsalva maneuver (release phase), with inhalation of amyl nitrite, and occasionally with passive leg raising and following a post-extrasystolic pause. The murmur is commonly accompanied by a right ventricular lift and thrill palpable along the upper left sternal border. A fourth heart sound (right sided) may be audible at the lower left sternal border. A pulmonic ejection click is frequently audible (during expiration) over the pulmonic area except in very severe cases. Patients with congenital pulmonary valvular stenosis commonly, but not invariably, have moon facies and hyperteleorism.
Infundibular pulmonic stenosis is present in approximately 10% of cases of right ventricular outflow obstruction. It is usually accompanied by a ventricular septal defect, but may also occur as an isolated abnormality. The murmur of isolated infundibular pulmonic stenosis is identical to that of valvular pulmonic stenosis except that it is best heard lower along the left sternal border (third intercostal space) and is rarely accompanied by an ejection click.
The murmur associated with atrial septal defect (ASD) is caused by increased blood flow in the right ventricular outflow tract. Accordingly, it is identical in character to the functional pulmonic ejection murmurs previously described. The presence of fixed and wide splitting of the second heart sound, not the nonspecific pulmonic flow murmur, confirms the presence of atrial septal defect.
The murmur associated with tetralogy of Fallot (ventricular septal defect, infundibular pulmonic stenosis, right ventricular hypertrophy, and overriding aorta) emanates predominantly from the ventricular septal defect when pulmonic stenosis is mild, but from the pulmonary outflow tract when pulmonic stenosis is moderate to severe. Tetralogy of Fallot with mild pulmonic stenosis is characterized by a loud (usually grade 4 or higher), harsh, midsystolic, crescendo–decrescendo murmur. The murmur is widely audible over the precordium arid peaks in late midsystole. With moderate to severe pulmonic stenosis, the murmur peaks progressively earlier in systole as much of the blood flow is shunted through the ventricular septal defect into the aorta. In extreme cases a short early systolic aortic flow murmur may be all that is audible. Tetralogy associated with mild pulmonic stenosis may be accompanied by a delayed and diminished pulmonic component of the second heart sound. The pulmonic component is not audible when pulmonic stenosis is moderate to severe. When extreme pulmonic stenosis is present, an aortic ejection click may be audible.
Regurgitant Murmurs
Systolic regurgitant murmurs include the many variations of mitral valve regurgitation, tricuspid valve regurgitation, and ventricular septal defect. Depending on its etiology and pathogenesis, mitral regurgitation may produce four discrete auscultatory patterns. Classic mitral valve regurgitation is typified by the systolic murmur of a rheumatic mitral valve. Other variations include mitral regurgitation associated with papillary muscle dysfunction, acute mitral regurgitation, and mitral valve prolapse.
The murmur of rheumatic mitral valve regurgitation is high pitched, blowing, and best heard at the cardiac apex with radiation to the axilla. It is holosystolic, starting with the first heart sound and extending to and sometimes through the aortic component of the second heart sound. Typically plateau in configuration, the murmur occasionally has late systolic accentuation. The intensity of classic mitral valve regurgitation is quite variable. The intensity increases with squatting, isometric hand grip exercise, and intravenous administration of alpha-adrenergic agonists. It typically decreases with amyl nitrite inhalation. As a result of the volume overload state associated with valvular mitral regurgitation, a third heart sound is commonly audible at the apex. Dilated cardiomyopathy can produce a mitral regurgitation murmur similar in character to rheumatic mitral valve regurgitation.
Mitral regurgitation associated with papillary muscle dysfunction is typically a midsystolic, crescendo–decrescendo, nonradiating murmur localized to the lower left sternal border. The murmur is medium to high pitched and commonly has a musical or cooing quality. The intensity is generally less than grade 4. Mitral regurgitation murmurs associated with left ventricular dilatation (e.g., from dilated cardiomyopathy) may occasionally present in this fashion.
Mitral valve prolapse (idiopathic or secondary) is a common cause of mitral regurgitation. Classically, mitral valve prolapse is characterized by a medium-pitched late systolic murmur ushered in by a midsystolic click. The murmur is best heard over the apex and generally does not radiate. The murmur commonly, but not invariably, has a crescendo configuration as it reaches the aortic component of the second heart sound. The intensity is quite variable. The intensity increases and the murmur starts earlier in systole with maneuvers or interventions that decrease left ventricular volume (and therefore increase the severity of prolapse), for example, standing, phase 2 (strain phase) of the Valsalva maneuver, and following amyl nitrite inhalation. These interventions also cause the midsystolic click to migrate closer to the first heart sound. Maneuvers and interventions that increase left ventricular volume decrease the intensity of the murmur. These include passive leg raising, recumbency, a post-extrasystolic pause, squatting, isometric hand grip exercise, and intravenous administration of alpha-adrenergic agonists. Such maneuvers and interventions cause the click to return to its original location in midsystole. Not infrequently, the murmur of mitral valve prolapse is holosystolic (either spontaneously or following interventions to decrease left ventricular volume). Such a murmur generally retains a medium pitch, but may radiate to the axilla. It may reach an intensity of grade 5 or 6 and may have a musical whooping or honking quality.
Acute mitral valve regurgitation is most commonly caused by chordae tendineae rupture, but may also result from papillary muscle rupture, infective endocarditis, or trauma. The murmur of acute mitral regurgitation is typically decrescendo and of variable intensity (usually grade 3 or higher). It begins with the first heart sound and decreases in intensity throughout systole, occasionally terminating before the aortic component of the second heart sound. Best heard at the cardiac apex, the murmur typically radiates to the axilla and may be audible along the cervical spine or at the top of the head in selected cases. The murmur is lower pitched than that of rheumatic mitral regurgitation, often possessing a harsh quality reminiscent of valvular aortic stenosis. A third and fourth heart sound may be audible at the apex.
The murmur of tricuspid valve regurgitation is typically a high-pitched, blowing, holosystolic, plateau, nonradiating murmur best heard at the lower left sternal border. The intensity is variable, but tends to increase during inspiration (Carvallo’s sign), with passive leg raising, after a post-extrasystole pause, and following amyl nitrite inhalation. The intensity of the murmur tends to correlate positively with the severity of regurgitation. Right ventricular enlargement may displace the location of the murmur leftward. Right ventricular failure may abolish respiratory variation. When tricuspid regurgitation is caused by pulmonary hypertension, a pulmonic ejection click may be audible. Severe tricuspid regurgitation is commonly accompanied by a third heart sound emanating from the right ventricle and best heard at the lower left sternal border. Severe tricuspid regurgitation typically produces an accentuated jugular “cv” wave and may produce hepatic congestion with a pulsatile liver.
The murmur of acute tricuspid valve regurgitation (due to infective endocarditis or trauma) is similar in character to that previously described, but often possesses a decrescendo configuration (similar to acute mitral regurgitation) and may terminate before the second heart sound.
The auscultatory findings associated with ventricular septal defect (VSD) are variable, depending on a variety of morphologic and hemodynamic considerations. The systolic murmur associated with a Roger’s-type ventricular septal defect (regurgitant jet flows directly into the right ventricular outflow tract) in patients with low pulmonary vascular resistance is a low to medium pitched, holosystolic murmur with midsystolic accentuation. The murmur is heard best over the third and fourth intercostal space at the left sternal border but is widely audible over the entire precordium. The intensity of the murmur is typically grade 3 or higher. Amyl nitrite inhalation causes the murmur of an uncomplicated ventricular septal defect to decrease, whereas alpha-adrenergic agonists cause no change or an increase in intensity. A precordial thrill and third heart sound (from left ventricular volume overload) frequently accompany the murmur. The pulmonic component of the second heart sound is occasionally delayed.
If pulmonary hypertension produces equalization or reversal of shunt flow in a Roger’s-type ventricular septal defect (Eisenmenger’s complex), the systolic murmur emanating from the septal defect may disappear entirely or be replaced by an early-peaking, medium-pitched, midsystolic murmur emanating from a dilated aortic root. Other auscultatory signs of pulmonary hypertension, including a loud pulmonic component of the second heart sound, a pulmonary ejection click, the early diastolic murmur of pulmonary valve insufficiency (Graham-Steel murmur), and right-side Austin-Flint murmur, may accompany the relatively nondescript systolic murmur. Physiologic maneuvers and pharmacologic interventions have little effect on small defects when pulmonary hypertension is present. With large defects, the murmur intensity may increase with amyl nitrite inhalation, but shows no change or decreases following intravenous administration of alpha-adrenergic agonists.
Small, slit-like ventricular septal defects typically produce an early systolic murmur that ends during the early portion of midsystole.
Extracardiac Sounds
Extracardiac sounds that may simulate systolic murmurs include the innocent subclavian murmur, carotid murmurs, the internal mammary souffle, coarctation of the aorta, and murmurs associated with a dilated aortic or pulmonic trunk and patent ductus arteriosus associated with pulmonary hypertension.
The innocent subclavian murmur (supraclavicular or brachiocephalic systolic murmur) is detected most frequently in children and adolescents. It has a crescendo–decrescendo configuration, an abrupt onset and brief duration in early to midsystole, and is medium to high pitched. Its origin is the brachiocephalic artery, and it is best heard over that region, but it may be heard with attenuation over the aortic and pulmonic areas of the precordium. The murmur disappears with compression of the brachiocephalic artery or hyperextension of the ipsilateral shoulder, a maneuver accomplished by bringing the elbows well behind the back, causing the shoulder girdle muscles to be taut. This murmur is frequently mistaken for an organic systolic murmur. Consequently some children who have it are unnecessarily kept from athletic or other strenuous physical activities.
A soft, high-pitched, early to midsystolic murmur may occasionally be audible along the sternal borders in pregnant women and appears to emanate from one of the internal mammary arteries. It is termed the internal mammary souffle and is of no pathological significance.
Bruits that originate in the carotid arteries may occasionally be mistaken for systolic heart murmurs. They are consistently heard best over the carotid arteries and are heard only with attenuation over the aortic and pulmonic areas.
Coarctation of the aorta typically is associated with a systolic murmur. In 50% of the cases a bicuspid aortic valve is present. When the murmur is not caused by valvular stenosis, it is thought to result from rapid blood flow across the stenotic segment of aorta. The murmur of coarctation is medium to high pitched and peaks rather late in systole. It is heard best over the left interscapular area and faintly if at all over the aortic areas of the precordium. The typical peripheral manifestations of coarctation permit relatively easy detection when a thorough examination is performed.
Systolic murmurs may also emanate from a dilated aortic or pulmonary trunk. Most commonly, this occurs in association with hypertension in the respective vascular circuit. The murmur is similar in character to that of aortic sclerosis or mild pulmonic stenosis.
Patent ductus arteriosus produces a continuous murmur in patients with normal pulmonary vascular resistance. As pulmonary vascular resistance increases, the diastolic portion of the murmur attenuates. With equalization of pressures in the systemic and pulmonary circuits, the systolic component of the murmur remains, extends through the second heart sound, and ends in early diastole. When pulmonary hypertension produces a right-to-left shunt, the murmur emanating from the ductus disappears and is replaced by a systolic flow murmur emanating from the root of the pulmonary artery.
The cause of a systolic murmur can accurately be ascertained in most cases from the physical examination. The medical history, resting electrocardiogram, and chest x-ray may provide valuable information concerning the impact of the underlying cardiac abnormality on the patient’s cardiopulmonary status, but rarely provide specific information useful in characterizing the systolic murmur. Phonocardiography and recording of pulse tracings may be used to confirm clinical suspicions. In this regard, they are most useful as teaching tools. Echocardiography is well suited to the characterization of systolic heart murmurs. A complete examination, employing M-mode, two-dimensional, and Doppler echocardiographic techniques, can accurately identify the cause of virtually any organic systolic heart murmur (and by exclusion confirm the presence of a functional systolic ejection murmur). Limitations exist in the ability of echocardiography reliably to quantify the severity of most cardiac abnormalities that produce systolic murmurs. Despite its potential usefulness in identifying underlying causes of systolic murmurs, echocardiography is too expensive to use as a screening tool. It should be considered only when the diagnosis is in question following examination by an experienced clinician. Cardiac catheterization is rarely needed to define the cause of a systolic heart murmur, but may be of great value in assessing the severity of the cause of the murmur and determining its impact on the heart and circulation.
Diastolic murmurs
A diastolic murmur is a sound of some duration occurring during diastole. All diastolic murmurs imply some alteration of anatomy or function of the cardiovascular structures. The four most commonly encountered diastolic murmurs include aortic and pulmonary valve regurgitation, and mitral and tricuspid valve rumbles (Table 3) 10. Compared to most systolic murmurs, diastolic murmurs are usually more difficult to hear, and certain auscultatory techniques are essential for their detection.
Table 3. Diastolic murmur causes
- Aortic regurgitation
- Pulmonary valve regurgitation
- Mitral rumble
- Obstruction to flow
- Mitral stenosis (rheumatic, congenital)
- Left atrial myxoma
- Cor triatriatum
- Localized pericardial constriction
- Increased flow
- Mitral regurgitation
- Ventricular septal defect
- Patent ductus arteriousus
- Complete heart block
- Obstruction to flow
- Tricuspid rumble
- Obstruction to flow
- Tricuspid stenosis (rheumatic, Ebstein’s anomoly, carinoid)
- Right atrial myxoma
- Localized pericardial constriction
- Increased flow
- Atrial septal defect
- Tricuspid regurgitation
- Obstruction to flow
The murmur of aortic regurgitation begins with the aortic component of the second sound and is decrescendo in intensity for a variable duration of diastole. It is usually a high-frequency, “blowing” sound, most often heard best along the left lower sternal border, although occasionally only in the second right intercostal space. It may be of maximum intensity along the right sternal border. Rarely, the murmur may be isolated at the apex impulse.
For detection, first think of a blowing, high-frequency sound coming from a distance (to simulate it, purse your lips very tightly and blow). Place the diaphragm of the stethoscope along the left sternal border with very firm pressure, enough pressure to leave a slight indentation on the skin when removed. The fingers may be used to hold the stethoscope, but to avoid the extraneous noise from tremor of the finger muscles, the palm of the hand may be better. The patient should be instructed “don”t breathe” at end expiration, or told to “take a deep breath, blow it all out then relax and don”t breathe.” A command to “hold your breath” may cause the patient to take in a deep breath and hold it. If the murmur is not heard at the left lower sternal border with the patient supine, auscultation in a similar fashion should be performed at the second right intercostal space and along the right sternal border. The murmur may only be heard by listening in one of these areas with the patient sitting, leaning forward in relaxed expiratory apnea. Any bedside maneuver that transiently increases blood pressure may intensify or bring out the murmur. Hand grip or squatting can be useful. Proper timing of the cardiac cycle is essential. A heart rate of 100 or greater abbreviates diastole so that systolic and diastolic duration are nearly equal. In this situation even a loud murmur of aortic regurgitation may be mistaken for a systolic murmur. Simultaneous palpation of the carotid pulse is essential to avoid this error.
The murmur of pulmonary valve regurgitation associated with pulmonary hypertension is an early diastolic, decrescendo murmur beginning with the pulmonary component of the second sound, best heard along the upper left sternal border. Auscultatory techniques are like those for aortic regurgitation. The quality of pulmonary valve regurgitation is similar to that of aortic regurgitation, and differentiation may be difficult. The murmur of pulmonary valve regurgitation may increase in intensity with inspiration. In association with mitral regurgitation, intensity may actually decrease with inspiration. The presence of bounding pulses and a wide pulse pressure support the diagnosis of aortic regurgitation. Pulmonary valve regurgitation frequently results from severe pulmonary hypertension. When the murmur is associated with mitral stenosis and pulmonary hypertension, it usually represents trivial aortic regurgitation simply because aortic regurgitation is more common than pulmonary valve regurgitation in this clinical setting.
The murmur of pulmonary valve regurgitation without associated pulmonary hypertension, as in pulmonary valve endocarditis or congenital abnormalities of the pulmonary valve, is of lower frequency and may be middiastolic with a crescendo—decrescendo pattern of intensity.
A mitral valve rumble is a diastolic murmur of low frequency occurring in middiastole and/or late diastole (presystole). It is frequently localized to a small area at the apex impulse. The patient should be relaxed in a left lateral decubitus position and the apex impulse localized. The bell of the stethoscope should be applied with very light pressure, just enough to make contact with the skin. Concentrate on diastole and move the bell over and just adjacent to the apex impulse. Listen in middiastole and just before the first sound. If the murmur is due to mitral stenosis, there may be accentuation of the first sound and an opening snap. The opening snap is a high-frequency sound that introduces the middiastolic component of the rumble and occurs .03 to .14 second after the second sound. Maneuvers that transiently increase cardiac output, such as sit-ups, coughing, or squatting, may aid in detection. When the apex impulse is not easily located, scanning the area, listening for the point of maximum intensity of the heart sounds, can help identify the apex impulse and the area on which to concentrate for the mitral diastolic rumble.
A tricuspid valve rumble has similar characteristics as the mitral rumble, but is localized along the left lower sternal border and increases in intensity with inspiration. The bell should be placed, again with very light pressure, exploring from the third to the fifth interspaces, concentrating in diastole both during inspiration and expiration. Similar maneuvers to increase venous return may augment the murmur. The inspiratory accentuation aids in differentiation from the mitral rumble, although the latter does not usually radiate to the left sternal border. Accentuation of the first sound (tricuspid component) and a tricuspid opening snap may also be present. The presystolic component of the tricuspid valve rumble is often crescendo—decrescendo, unlike the crescendo pattern of the mitral rumble.
References- Tuteur PG. Chest Examination. In: Walker HK, Hall WD, Hurst JW, editors. Clinical Methods: The History, Physical, and Laboratory Examinations. 3rd edition. Boston: Butterworths; 1990. Chapter 46. Available from: https://www.ncbi.nlm.nih.gov/books/NBK368
- Reyes FM, Bhimji SS. Lung, Exam. [Updated 2018 Oct 27]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2018 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK459253
- Sarkar M, Madabhavi I, Niranjan N, Dogra M. Auscultation of the respiratory system. Ann Thorac Med. 2015;10(3):158-68. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4518345/
- Bishop PJ. Reception of the stethoscope and Laënnec’s book. Thorax 1981; 36: 487–492.
- Felner JM. The First Heart Sound. In: Walker HK, Hall WD, Hurst JW, editors. Clinical Methods: The History, Physical, and Laboratory Examinations. 3rd edition. Boston: Butterworths; 1990. Chapter 22. Available from: https://www.ncbi.nlm.nih.gov/books/NBK333
- Alpert MA. Systolic Murmurs. In: Walker HK, Hall WD, Hurst JW, editors. Clinical Methods: The History, Physical, and Laboratory Examinations. 3rd edition. Boston: Butterworths; 1990. Chapter 26. Available from: https://www.ncbi.nlm.nih.gov/books/NBK345
- Levine S. The systolic murmur: its clinical significance. JAMA. 1934;101:436–38.
- Harvey WP. Innocent versus significant systolic murmurs. Curr Prob Cardiol. 1966;1(8):1–55.
- Ashley EA, Niebauer J. Cardiology Explained. London: Remedica; 2004. Chapter 9, Valve disease. Available from: https://www.ncbi.nlm.nih.gov/books/NBK2220
- Crawley IS. Diastolic Murmurs. In: Walker HK, Hall WD, Hurst JW, editors. Clinical Methods: The History, Physical, and Laboratory Examinations. 3rd edition. Boston: Butterworths; 1990. Chapter 27. Available from: https://www.ncbi.nlm.nih.gov/books/NBK346