What is blood gas
Blood gases are a group of tests that are performed together, usually on an arterial blood sample (blood obtained from an artery instead of a vein), to measure of how much oxygen (PaO2 the partial pressure of oxygen or the amount of oxygen in the blood) and carbon dioxide (PaCO2 the partial pressure of carbon dioxide or the amount of carbon dioxide the blood) are in your blood. From these results, bicarbonate (HCO3-) can be calculated. Blood gas test also determines the acidity (pH) of your blood, usually taken from an artery (arterial blood gas), in order to evaluate your lung function and help detect an acid-base imbalance that could indicate a respiratory, metabolic or kidney disorder.
A person’s body carefully regulates blood pH, maintaining it within a narrow range of 7.35-7.45 (average pH 7.40), not allowing blood to become too acidic (acidosis or pH<7.35) nor too alkaline/basic (alkalosis or pH >7.45). The body’s regulation of acids and bases has two main components. The first component involves both metabolism and the kidneys: the cellular process of converting one substance to another for energy produces large amounts of acid that the kidneys help eliminate. The second component of regulating pH balance involves eliminating carbon dioxide (an acid when dissolved in blood) through exhalation of the lungs. This respiratory component is also the way that the body supplies oxygen to tissues. The lungs inhale oxygen, which is then dissolved in the blood and carried throughout the body to tissues.
These processes of gas exchange and acid/base balance are also closely associated with the body’s electrolyte balance. In a normal state of health, these processes are in a dynamic balance and the blood pH is stable.
There is a wide range of acute and chronic conditions that can affect kidney function, acid production, and lung function, and they have the potential to cause a pH, carbon dioxide/oxygen, or electrolyte imbalance. Examples include uncontrolled diabetes, which can lead to ketoacidosis and metabolic acidosis, and severe lung diseases that can affect CO2/O2 gas exchange. Even temporary conditions such as shock, anxiety, pain, prolonged vomiting, and severe diarrhea can sometimes lead to acidosis or alkalosis.
Blood gas analysis gives a snapshot of a person’s blood pH, oxygen (O2) and carbon dioxide (CO2) content. The following components are generally included in blood gas analysis:
- pH—a measure of the balance of acids and bases in the blood. Increased amounts of carbon dioxide and other acids can cause blood pH to decrease (become acidic). Decreased carbon dioxide or increased amounts of bases, like bicarbonate (HCO3-), can cause blood pH to increase (become alkaline).
- Partial pressure of O2 (PaO2) — Partial pressure of oxygen (PaO2) is the amount of oxygen dissolved in the plasma. A PaO2 less than 80 mm Hg is considered abnormal. However, this should be in line with the clinical situation.
- Partial pressure of CO2 (PaCO2) — measures the amount of carbon dioxide gas in the blood and is an indirect measure of exchange of carbon dioxide (CO2) with the air via the alveoli, its level is related to minute ventilation. Partial pressure of CO2 (PaCO2) is elevated in hypoventilation like in obesity hypoventilation, deep sedation, or may be low in the setting of acute hypoxia secondary to tachypnea and washout of CO2. As PaCO2 levels rise, blood pH decreases, making the blood more acidic; as PaCO2 decreases, pH rises, making the blood more basic (alkaline).
- O2 saturation (O2Sat or SaO2) — the percentage of hemoglobin that is carrying oxygen. Hemoglobin is the protein in red bloods cells that carries oxygen through blood vessels to tissues throughout the body.
- O2 content (O2CT or CaO2) — the amount of oxygen per 100 mL of blood.
- Bicarbonate (HCO3-) — the main form of CO2 in the body. It can be calculated from the pH and PaCO2. It is a measurement of the metabolic component of the acid-base balance. Bicarbonate (HCO3-) is released and reabsorbed by the kidneys in response to pH imbalances and is directly related to the pH level. As the amount of bicarbonate (HCO3-) rises in the blood, so does the pH (becomes alkaline).
- Base excess/base deficit — a calculated number that represents a sum total of the metabolic buffering agents (anions) in the blood. These anions include hemoglobin, proteins, phosphates, and HCO3- (bicarbonate, which is the dominant anion). Anions are regulated to compensate for imbalances in blood pH. The healthcare practitioner will look at the HCO3- and base excess/deficit results to evaluate the total buffering capacity of the lungs and kidneys when deciding on a treatment to correct an imbalance.
Blood gas test
Arterial whole blood is almost always used for blood gas analysis but, in some cases, as with babies, whole blood from a heelstick is collected instead. Blood may also be taken from the umbilical cord of a newborn. Since arterial blood carries oxygen to the body and blood from a vein (venous blood) carries waste products to the lungs and kidneys, the gas and pH levels will not be the same in both types of blood samples. Typically, the largest difference in reported values between venous and arterial blood is the PaO2, and consideration of sample type should be taken into account when reviewing results.
Blood may be collected from one of the following arteries:
- Radial artery in the wrist
- Femoral artery in the groin
- Brachial artery in the arm
Your health care provider may test circulation to the hand before taking a sample of arterial blood from the wrist area.
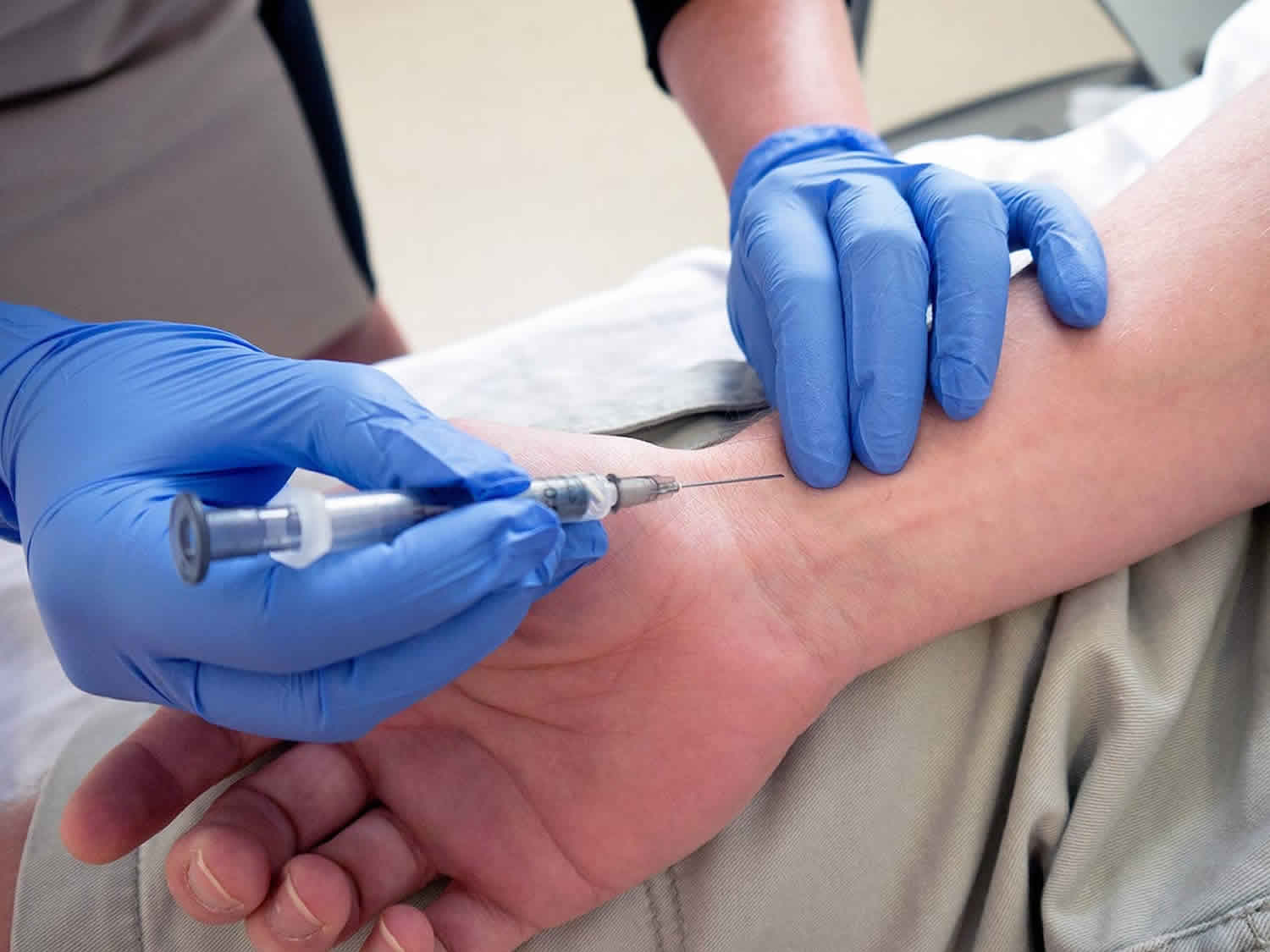
The provider inserts a small needle through the skin into the artery. The sample is quickly sent to a laboratory for analysis.
An arterial blood sample is usually collected from the radial artery in the wrist, located on the inside of the wrist, below the thumb, where the pulse can be felt. A circulation test called an Allen test will be done before the collection to make sure that there is adequate circulation in the person’s wrist. The test involves compressing both the radial and the ulnar wrist arteries, then releasing each in turn to watch for “flushing,” the pinking of the skin as blood returns to the hand. If one hand does not flush, then the other wrist will be tested. Blood can also be collected from the brachial artery in the elbow or the femoral artery in the groin, although these sample locations require special training to properly access. Blood may also be collected from an arterial catheter line but should be taken as to ensure minimal contamination. Arterial blood sample collection is usually a bit more painful than regular venipuncture. You may experience moderate discomfort and a compress may be required for some time to prevent any bleeding from the site.
Sometimes mixed venous blood taken from a central line is used in particular situations, such as in cardiac catheterization labs and by transplant services. Careful interpretation of the results is required. Peripheral venous blood, such as that taken from a vein in the arm, is of no use for oxygen status because it has decreased oxygen content due to the fact that it is composed of blood returning to the heart.
In newborns that experience difficulty in breathing right after birth, blood may be collected from both the umbilical artery and vein and tested separately.
After an arterial blood draw, pressure must be firmly applied to the site for at least 5 minutes. Since blood pumps through the artery, the puncture may take awhile to stop bleeding. If someone is taking blood thinners or aspirin, it may take as long as 10-15 minutes to stop bleeding. Following collection, the person taking the sample will verify that the bleeding has stopped and will put a wrap around the wrist, which should be left in place for about an hour.
Typically, no test preparation is needed. However, if someone is on oxygen therapy, the O2 (oxygen) may either be turned off for 20 to 30 minutes before the collection for a “Room Air” test or, if this cannot be tolerated or if the healthcare practitioner wants to check oxygen levels with the O2 on, the amount of oxygen being delivered will be recorded. This is usually expressed as fraction of inspired (inhaled) oxygen in percent (FiO2) or as liters of O2 flowing per minute.
How is blood gas test used?
Blood gas test is used to evaluate respiratory diseases and conditions that affect the lungs. Blood gas test helps determine the effectiveness of oxygen therapy. Blood gas test also provides information about the body’s acid/base balance, which can reveal important clues about lung and kidney function and the body’s general metabolic state.
Blood gas tests are typically ordered if someone is having worsening symptoms of a respiratory problem, such as difficulty breathing or shortness of breath, and a condition such as asthma or chronic obstructive pulmonary disease (COPD) is suspected. Blood gases may also be used to monitor treatment for lung diseases and to evaluate the effectiveness of supplemental oxygen therapy.
Blood gases can also be used to detect an acid-base imbalance, which can occur in kidney failure, heart failure, uncontrolled diabetes, severe infections, and drug overdose. They may be ordered along with other tests, such as electrolytes to determine if an electrolyte imbalance is present, glucose to evaluate blood sugar concentrations, and BUN (blood urea nitrogen) and creatinine tests to evaluate kidney function.
When is blood gas test ordered?
A blood gas analysis is ordered when someone has symptoms of an oxygen/carbon dioxide or pH imbalance, such as difficulty breathing, shortness of breath, nausea, or vomiting. It may also be ordered when someone is known to have respiratory, metabolic, or kidney disease and is experiencing respiratory distress.
When someone is “on oxygen” (ventilation), blood gases may be measured at intervals to monitor the effectiveness of treatment. Other treatments for lung diseases may also be monitored with blood gases.
Blood gases may also be ordered when someone has head or neck trauma, which may affect breathing, and when someone is undergoing prolonged anesthesia – particularly for cardiac bypass surgery or brain surgery – to monitor blood gases during, and for a period after, the procedure.
Checking blood gases from the umbilical cord of a newborn may uncover respiratory problems as well as determine acid/base status. Testing is usually only done if a newborn is having difficulty breathing.
Is there any other way to measure my oxygen levels?
A pulse oximeter is a noninvasive way (no needlestick or blood sample required) of continuously monitoring O2 saturation. A small clip-like device (sensor) is attached to the end of the finger or earlobe. The sensor reads light that is transmitted through the skin. Pulse oximeters are useful for monitoring trends in O2 saturation, but their accuracy can be affected by the presence of abnormal forms of hemoglobin, like carboxyhemoglobin (see below), low blood pressure due to poor perfusion (pumping of blood into an organ or tissue), and very low levels of hemoglobin due to severe anemia.
Are there any risks associated with having blood gas test?
There is very little risk when the procedure is done correctly. Veins and arteries vary in size from one person to another and from one side of the body to the other. Taking blood from some people may be more difficult than from others.
Other risks associated with blood gas test may include:
- Bleeding at the puncture site
- Blood flow problems at puncture site (rare)
- Bruising at the puncture site
- Delayed bleeding at the puncture site
- Fainting or feeling lightheaded
- Hematoma (blood accumulating under the skin)
- Infection (a slight risk any time the skin is broken)
Blood gas analysis
Blood gas analysis, performed by trained personnel, is usually done in a hospital, emergency room, surgical center, ambulance, or large laboratory setting because it should be performed immediately after sample collection and specialized equipment is required.
Normal values will vary from lab to lab. They are also dependent on elevation above sea level as a person’s blood oxygen level will be lower if he or she lives higher than sea level.
- Results from an arterial blood gas analysis are not diagnostic; they should be used in combination with the results of other tests and exams to evaluate someone for a respiratory, metabolic, or kidney problem.
Arterial blood gas normal values
Normal arterial blood gas values at sea level:
- Partial pressure of oxygen (PaO2): 75 to 100 mm Hg (10.5 to 13.5 kPa)
- Partial pressure of carbon dioxide (PaCO2): 38 to 42 mm Hg (5.1 to 5.6 kPa)
- Arterial blood pH: 7.38 to 7.42
- Oxygen saturation (SaO2): 94% to 100%
- Bicarbonate (HCO3-): 22 to 28 mEq/L
Note: mEq/L = milliequivalents per liter; mmHg = millimeters of mercury
At altitudes of 3,000 feet (900 meters) and higher, the oxygen value is lower.
Normal value ranges may vary slightly among different laboratories. Some laboratories use different measurements or may test different specimens. Talk to your doctor about the meaning of your specific test results.
Arterial blood gas abnormal values
Abnormal results of any of the blood gas components may indicate one or more of the following issues:
- A person is not getting enough oxygen (hypoxia)
- A person is not getting rid of enough carbon dioxide (hypercarbia or hypercapnia)
- There is a problem with a person’s kidney function either kidney disease or kidney failure (metabolic acidosis)
Abnormal results may be due to lung, kidney, or metabolic diseases. Head or neck injuries or other injuries that affect breathing can also lead to abnormal results.
A low partial pressure of oxygen (PaO2) suggests that a person is not getting enough oxygen, while results that are within normal range usually mean that oxygen intake is sufficient.
All other components of the blood gas analysis (pH, PaCO2, HCO3-) are interrelated and the results must be considered together. Certain combinations of results, if abnormal, may indicate a condition that is causing acidosis or alkalosis. These may include the following:
- Respiratory acidosis is characterized by a lower pH and an increased PaCO2 and is due to respiratory depression (not enough oxygen taken in and carbon dioxide removed). This can be caused by many things, including pneumonia, chronic obstructive pulmonary disease (COPD), and over-sedation from narcotics.
- Respiratory alkalosis, characterized by a raised pH and a decreased PaCO2, is due to over-ventilation caused by hyperventilating, pain, emotional distress, or certain lung diseases that interfere with oxygen exchange.
- Metabolic acidosis is characterized by a lower pH and decreased HCO3-, causing the blood to be too acidic for proper metabolic/kidney function. Causes include diabetes (diabetic ketoacidosis), shock, and kidney failure.
- Metabolic alkalosis is characterized by an elevated pH and increased HCO3- and is seen in hypokalemia, chronic vomiting (losing acid from the stomach), and sodium bicarbonate overdose.
How to interpret arterial blood gas values
The first value you should look at is the pH to determine if the patient is in normal range, above, or below. If a patient’s pH > 7.45, the patient is alkalotic. If the pH < 7.35, then the patient is acidotic.
Acidosis occurs when blood pH falls below 7.35. It can be due to increased acid or decreased base:
- Increased acid production within the body
- Consumption of substances that are metabolized to acids
- Decreased acid excretion
- Increased excretion of base
Alkalosis occurs when blood pH rises above 7.45. It can be due to decreased acid or increased base:
- Electrolyte disturbances caused by, for example, prolonged vomiting or severe dehydration
- Administration or consumption of base
- Hyperventilation (with increased excretion of acid in the form of CO2)
Any disease or condition that affects the lungs, kidneys, metabolism or breathing has the potential to cause acidosis or alkalosis.
Next, examine the PaCO2. This will determine if the changes in the blood gas are due to the respiratory system or metabolically driven. In combination with the bicarbonate (HCO3-), to fully comprehend the blood gas.
The CO2 is the respiratory component of the blood gas: if the PaCO2 is low, the bicarbonate (HCO3-) is low and the pH is high then the patient would have respiratory alkalosis. These values move in opposite directions.
On the other hand, the bicarbonate (HCO3-) is the metabolic component of the blood gas. If the bicarbonate (HCO3-) is low, the PaCO2 is low and the pH is low (pH<7.35) then the patient is in metabolic acidosis. These values move in the same direction.
In most acid-base disorders, both bicarbonate (HCO3-) and PaCO2 (partial pressure of carbon dioxide) are outside the reference range. That is because the body tries to keep the pH close to normal. Therefore, if one of these substances is abnormally high or low, the other one will change in order to bring the pH back toward its healthy range. This process is called compensation.
The table below shows the expected values for each of the four main acid-base disorders. This will enable the medical team to treat the patient adequately.
Table 1. Results seen in the four primary acid-base disorders (examples of test results associated with the above conditions are summarized below)
| Acid-Base Disorder | pH | HCO3– | PaCO2 | Common causes | Body Compensation |
|---|---|---|---|---|---|
| Metabolic acidosis | Less than 7.35 | Low | Low | Kidney failure, shock, diabetic ketoacidosis, intoxication with methanol, salicyate, ethanol | Increased breathing rate (hyperventilation) to increase CO2 elimination |
| Metabolic alkalosis | Greater than 7.45 | High | High | Chronic vomiting, low blood potassium, heart failure, cirrhosis | Slowed breathing (hypoventilation) to decrease CO2 elimination |
| Respiratory acidosis | Less than 7.35 | High | High | Narcotics, lung diseases such as asthma, COPD, airway obstruction, pneumonia, myasthenia gravis | Kidney increases retention of HCO3– and excretion of acid |
| Respiratory alkalosis | Greater than 7.45 | Low | Low | Hyperventilation, pain, anxiety, brain trauma, pneumonia, certain drugs (salicylate, catecholamines) | Kidney decreases retention of HCO3– and excretion of acid |
It is possible for a person to have more than one acid-base disorder at the same time. Examples include ingestion of aspirin (which can produce both a respiratory alkalosis and metabolic acidosis) and those with lung disease who are taking diuretics (respiratory acidosis plus metabolic alkalosis). By using formulas to calculate the expected degree of compensation, it is possible to determine if a mixed acid-base disorder is present.
If left untreated, these conditions can create an imbalance that can eventually become life-threatening.
A healthcare practitioner can provide the necessary medical intervention to regain normal acid/base balance. Treatment of acidosis and alkalosis involves identifying and treating the underlying cause(s) of the imbalance and providing whatever support is necessary to the affected person. In some cases, health practitioners may give intravenous HCO3- to people with dangerously low blood pH levels.
Common causes of Acid-Base Disorders
Respiratory acidosis
Reduced CO2 elimination
- Decreased breathing rate (respiratory drive) due to drugs or central nervous system disorders
- Impaired breathing and lung movement (respiratory mechanics) due, for example, to trauma or abnormal presence of air between the lung and the wall of the chest (pneumothorax)
- Respiratory muscle/nerve disease (myasthenia gravis, botulism, amyotrophic lateral sclerosis (ALS), Guillain-Barre syndrome)
- Airway obstruction (food or foreign object)
- Lung disease
Respiratory alkalosis
Increased CO2 elimination
- Hyperventilation due to anxiety, pain, shock
- Drugs, such as early in a overdose of aspirin (salicylates)
- Pneumonia, pulmonary (lung) congestion (due to heart failure), or embolism
- Exercise
- Fever
- Central nervous system tumor, trauma, infection (meningitis, encephalitis)
- Liver failure
Metabolic acidosis
Decreased bicarbonate (HCO3-), due to increased acid or loss of bicarbonate
- Alcoholic ketoacidosis
- Diabetic ketoacidosis
- Kidney failure
- Lactic acidosis
- Toxins – late in an overdose of salicylates (aspirin), methanol, ethylene glycol
- Gastrointestinal bicarbonate loss, such as from prolonged diarrhea
- Renal bicarbonate loss
Metabolic alkalosis
Increased bicarbonate (HCO3-), due to loss of acid or gain of bicarbonate
- Diuretics
- Prolonged vomiting
- Severe dehydration
- Diseases that cause loss of potassium
- Administration of bicarbonate, ingestion of alkali
Some acid-base disorders are also “mixed” expecially when the body tries to compensate.
Why does my lab report also list carbyoxyhemoglobin?
If your blood gases were measured using an instrument known as a co-oximeter, then your lab report may also list results for carboxyhemoglobin (COHb or HbCO) and other altered forms of hemoglobin (Hb). A co-oximeter is a blood gas analyzer that can measure concentrations of hemoglobin derivatives (like carboxyhemoglobin) in addition to the usual blood gas measurements. A co-oximeter is not always used, so these values are not reported for all blood gas analyses.
Carboxyhemoglobin is an altered form or derivative of hemoglobin that forms when carbon monoxide binds to hemoglobin. Levels of carboxyhemoglobin are often elevated with carbon monoxide poisoning, and a co-oximeter is used to measure carboxyhemoglobin levels and to monitor oxygen therapy. Hemoglobin binds to carbon monoxide about 210 times more strongly than to oxygen. Binding to carbon dioxide significantly decreases hemoglobin’s ability to carry oxygen through the body, which can lead to a serious, life-threatening condition.
Other hemoglobin derivatives include sulfhemoglobin (or sulfmethemoglobin) and methemoglobin, which may result from the ingestion of certain medicines or exposure to chemicals. These altered forms of hemoglobin, like carboxyhemoglobin, cannot function properly to carry oxygen to tissues in the body and are commonly measured by a co-oximeter.
Venous blood gas
A venous blood gas test is identical to an arterial blood gas measurement, except the blood is drawn from a venous site. This results in a slightly more acidic “normal” pH range. Most neonatologists use venous blood gas in neonates for metabolic evaluation and monitoring gas exchange, but not for monitoring oxygenation 1.
References- Tan RNGB, Pauws SC, van Loon E, Smits VEHJ, Lopriore E, Te Pas AB. Correlation and Interchangeability of Venous and Capillary Blood Gases in Non-Critically Ill Neonates. Front Pediatr. 2018;6:89. Published 2018 Apr 12. doi:10.3389/fped.2018.00089 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5932392/