What is brachial plexus nerve injury
The brachial plexus is a group of nerves around shoulder (see Figures 1 and 2 below). A loss of movement or weakness of the arm may occur if the brachial plexus is injured or damaged. This injury is called brachial plexus injury.
Brachial plexus injuries can happen because of shoulder trauma, tumors, or inflammation. Sometimes they happen during childbirth when a baby’s shoulders become stuck during delivery and the nerves stretch or tear.
Minor brachial plexus injuries, known as stingers or burners, are common in contact sports, such as football. Babies sometimes sustain brachial plexus injuries during birth. Other conditions, such as inflammation or tumors, may affect the brachial plexus.
The most severe brachial plexus injuries usually result from auto or motorcycle accidents. Severe brachial plexus injuries can leave your arm paralyzed, with a loss of function and sensation. Surgical procedures such as nerve grafts, nerve transfers or muscle transfers can help restore function.
Symptoms of a brachial plexus injury may include a limp or paralyzed arm; lack of muscle control in the arm, hand, or wrist; and a lack of feeling or sensation in the arm or hand. Brachial plexus injuries can also occur as a result of shoulder trauma, tumors, or inflammation. There is a rare syndrome called Parsonage-Turner Syndrome, or brachial plexitis, which causes inflammation of the brachial plexus without any obvious shoulder injury. This syndrome can begin with severe shoulder or arm pain followed by weakness and numbness. In infants, brachial plexus injuries may happen during birth if the baby’s shoulder is stretched during passage in the birth canal.
Based on the location of the nerve damage, brachial plexus injuries can affect part of or the entire arm. For example, musculocutaneous nerve damage weakens elbow flexors, median nerve damage causes proximal forearm pain, and paralysis of the ulnar nerve causes weak grip and finger numbness. In some cases, these injuries can cause total and irreversible paralysis. In less severe cases, these injuries limit use of these limbs and cause pain.
The cardinal signs of brachial plexus injury are weakness in the arm, diminished reflexes, and corresponding sensory deficits.
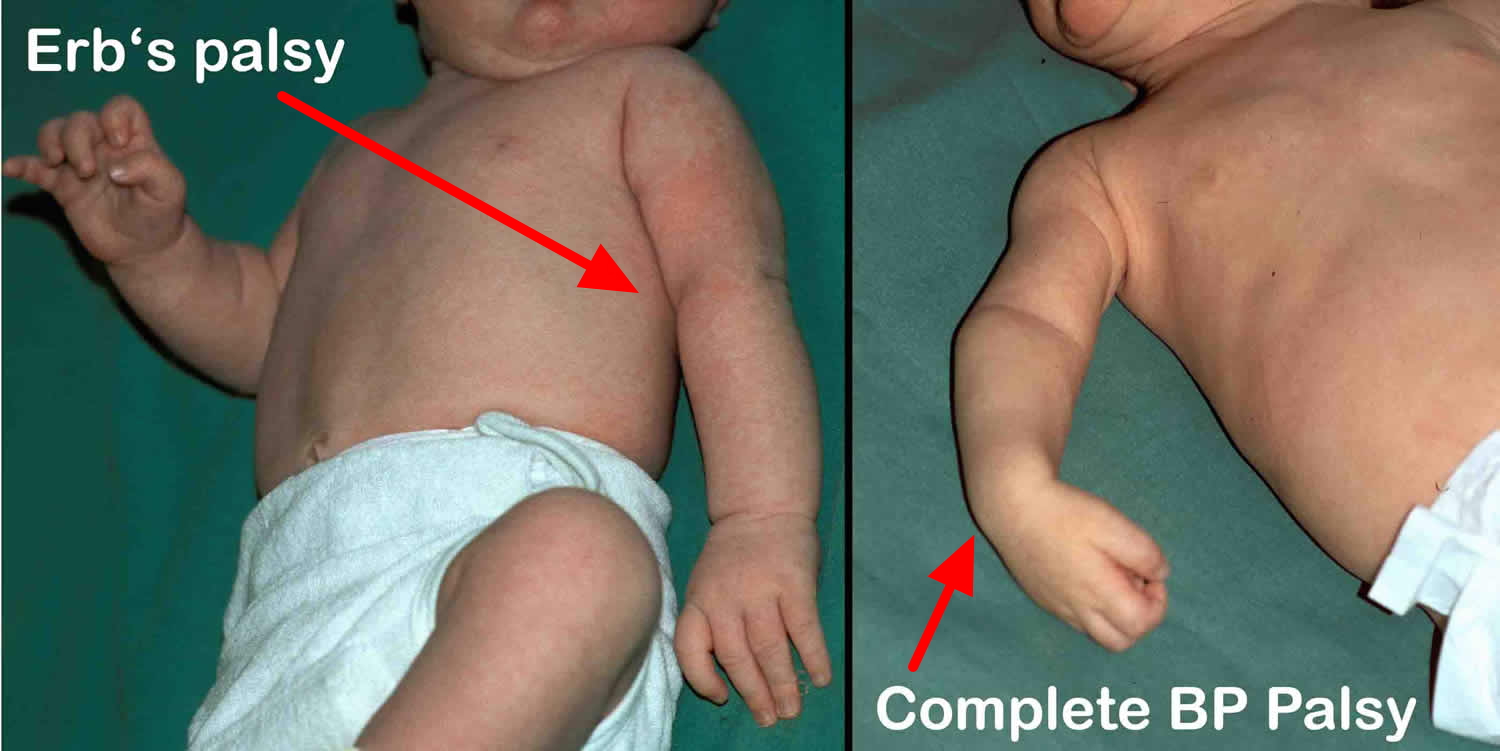
- Erb’s palsy (Erb-Duchenne palsy) caused by injury to the superior roots of the brachial plexus (C5–C6). The position of the limb, under such conditions, is characteristic: the arm hangs by the side and is rotated medially; the forearm is extended and pronated. The arm cannot be raised from the side; all power of flexion of the elbow is lost, as is also supination of the forearm — “waiter’s tip position”. There is loss of sensation along the lateral side of the arm.
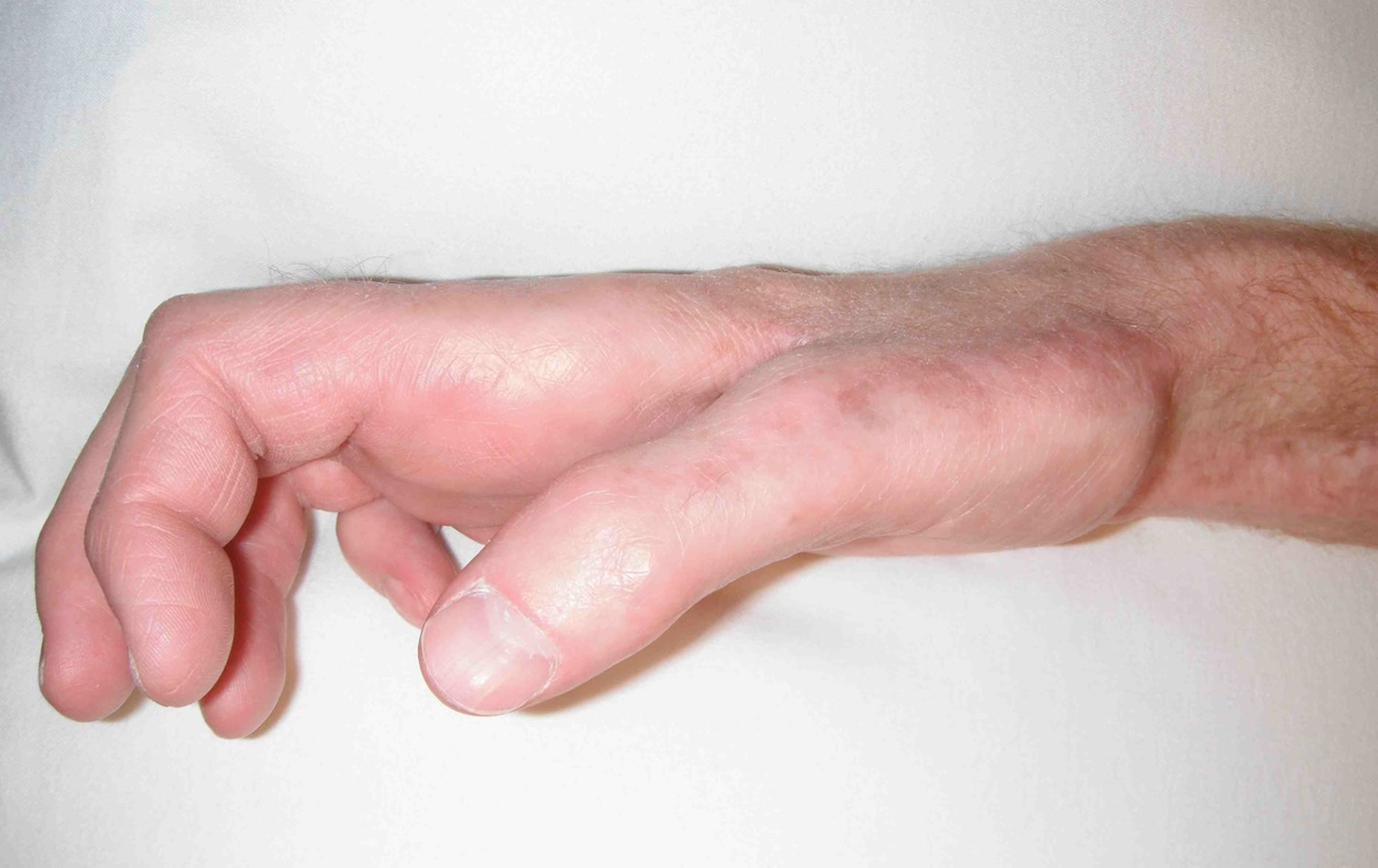
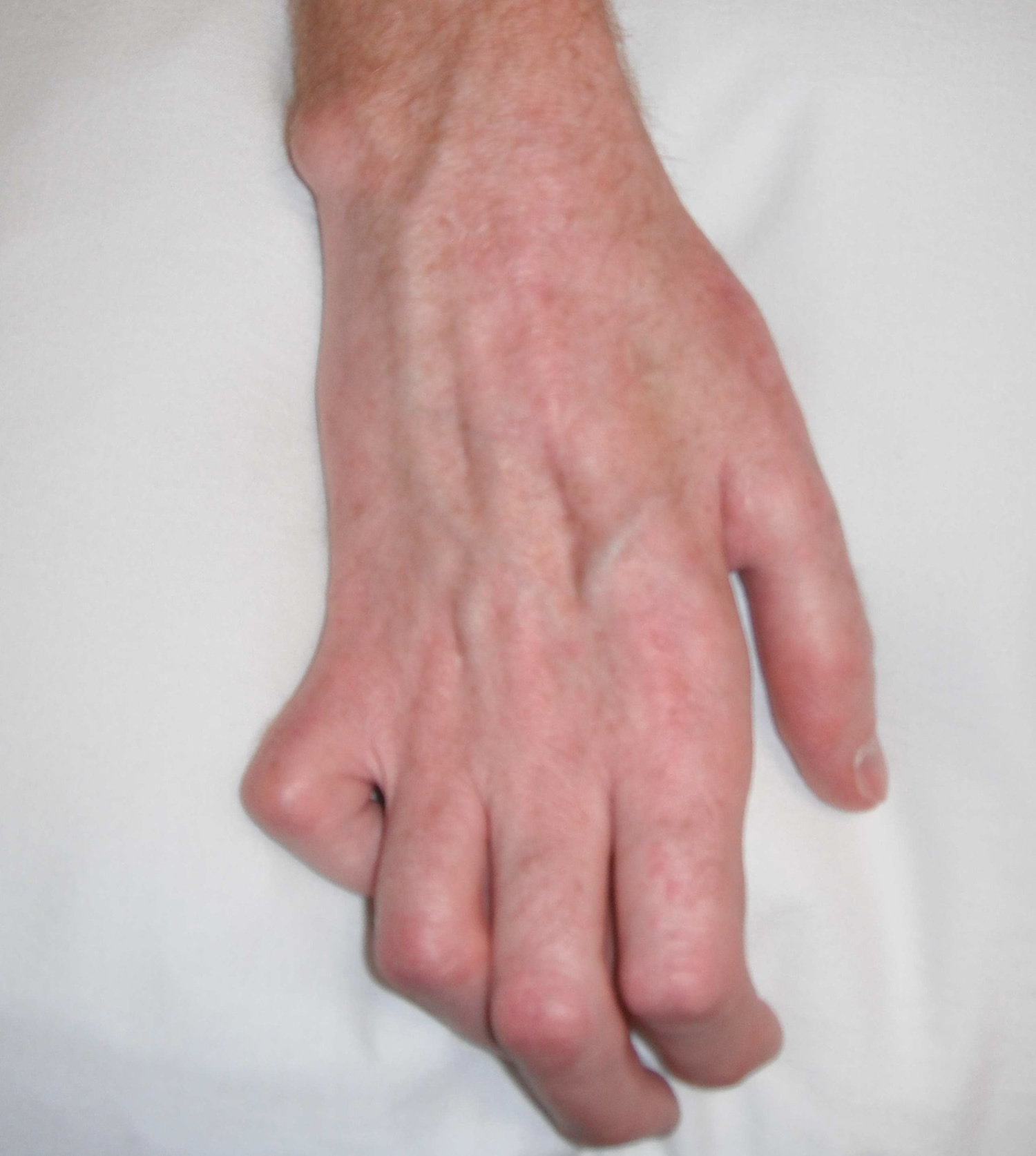
- Klumpke’s paralysis, also called Dejerine-Klumpke palsy refers to paralysis of the lower brachial plexus (C8 and T1). Klumpke’s paralysis typically involve the muscles of the forearm and hand, a characteristic sign is the clawed hand, due to loss of function of the ulnar nerve and the intrinsic muscles of the hand it supplies. Involvement of T1 may result in Horner’s syndrome, with ptosis (droop upper eyelid), enophthalmos (sinking of the eyeball into the face), anhidrosis (decreased sweating on the affected side of the face) and miosis (excessive constriction of the pupil of the eye).
The site and type of brachial plexus injury determines the prognosis. For avulsion and rupture injuries, there is no potential for recovery unless surgical reconnection is made in a timely manner. The potential for recovery varies for neuroma and neuropraxia injuries. Most individuals with neuropraxia injuries recover spontaneously with a 90-100% return of function.
Brachial plexus injuries can cause permanent weakness or disability. Even if yours seems minor, you may need medical care. See your doctor if you have:
- Recurrent burners and stingers
- Weakness in your hand or arm
- Weakness in any part of the arm following trauma
- Complete paralysis of the upper extremity following trauma
- Neck pain
- Symptoms in both arms
- Symptoms in upper and lower limbs
It’s important to be evaluated and treated within six to seven months after the injury. Delays in treatment may compromise outcomes of nerve surgeries.
The brachial plexus
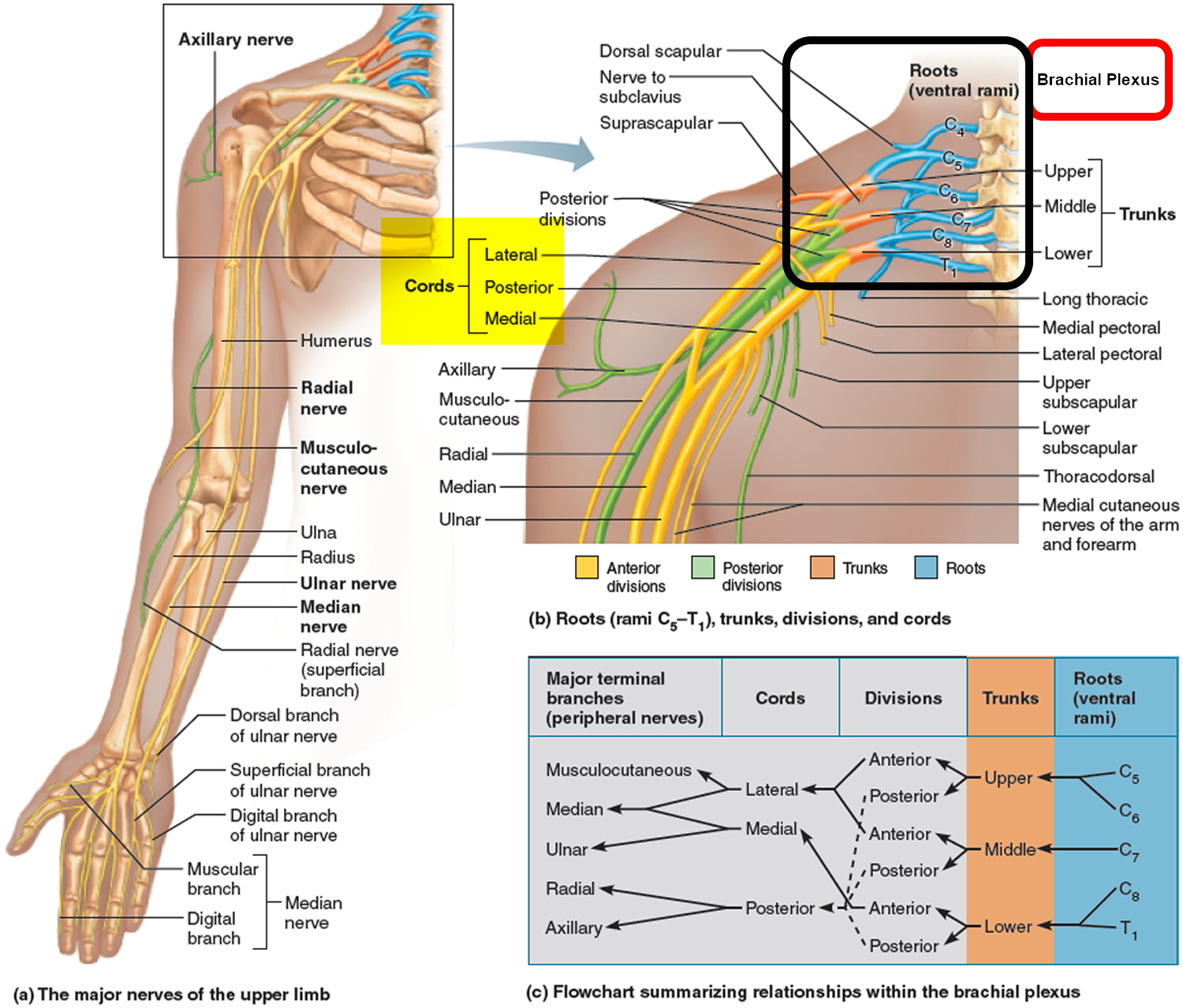
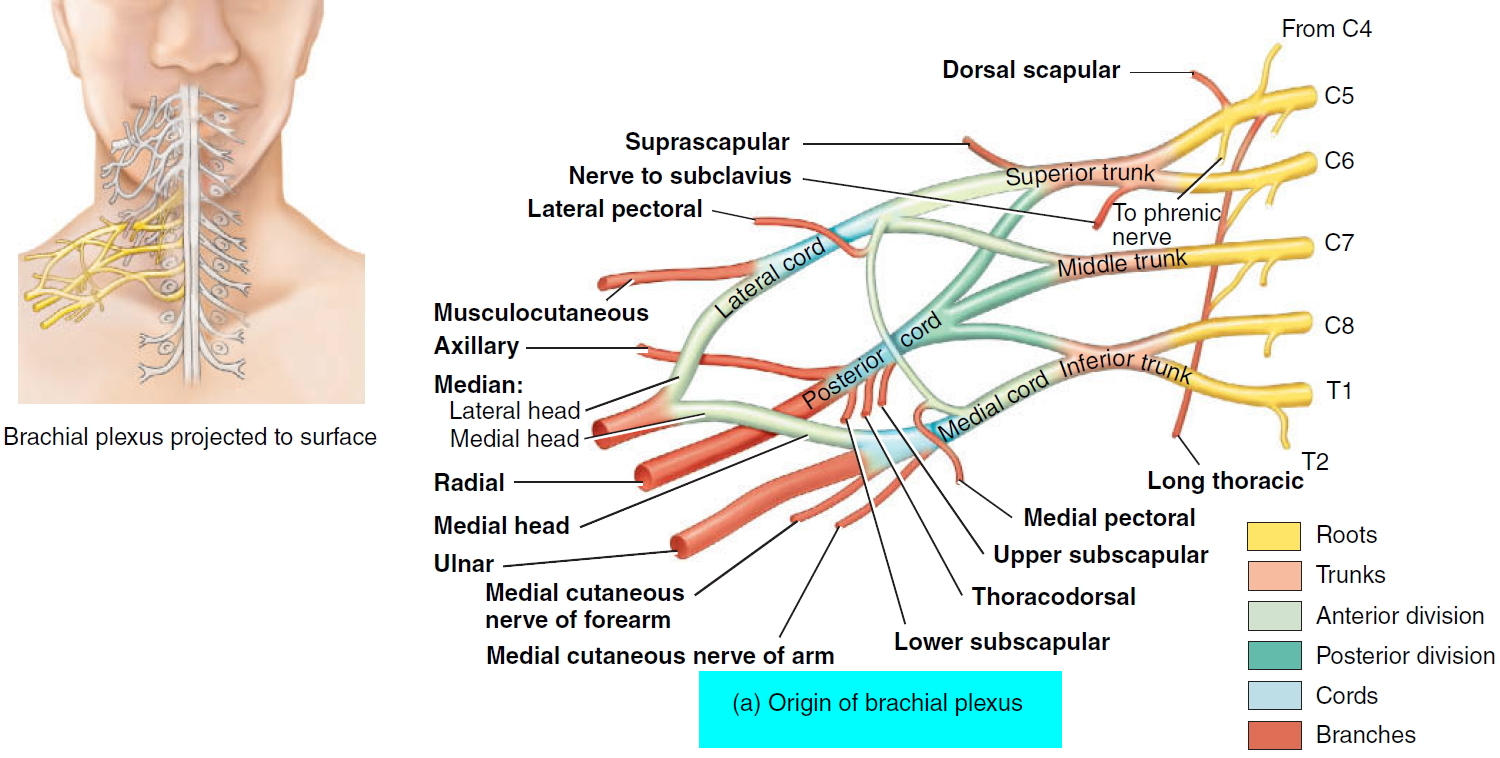
The roots (anterior rami) of spinal nerves C5–C8 and T1 form the brachial plexus, which extends inferiorly and laterally on either side of the last four cervical and first thoracic vertebrae (Figures 1 and 2). The brachial plexus passes above the first rib posterior to the clavicle and then enters the axilla (armpit).
Figure 1. Brachial plexus
Figure 2. Brachial plexus origin and nerve branches
Figure 4. Erb’s palsy (C5-C6 nerve damage) waiter’s tip position
Figure 5. Klumpke’s paralysis (C8-T1 nerve damage) claw hand
Since the brachial plexus is so complex, an explanation of its various parts is helpful. As with the cervical and other plexuses, the roots are the anterior rami of the spinal nerves. The roots of several spinal nerves unite to form trunks in the inferior part of the neck. These are the superior, middle, and inferior trunks. Posterior to the clavicles, the trunks diverge into divisions, called the anterior and posterior divisions. In the axillae (armpits), the divisions unite to form cords called the lateral, medial, and posterior cords. The cords are named for their relationship to the axillary artery, a large artery that supplies blood to the upper limb. The branches of the brachial plexus form the principal nerves of the brachial plexus.
The brachial plexus provides almost the entire nerve supply of the shoulders and upper limbs (Figures 1 and 2). Five large terminal branches arise from the brachial plexus:
- (1) The axillary nerve supplies the deltoid and teres minor muscles.
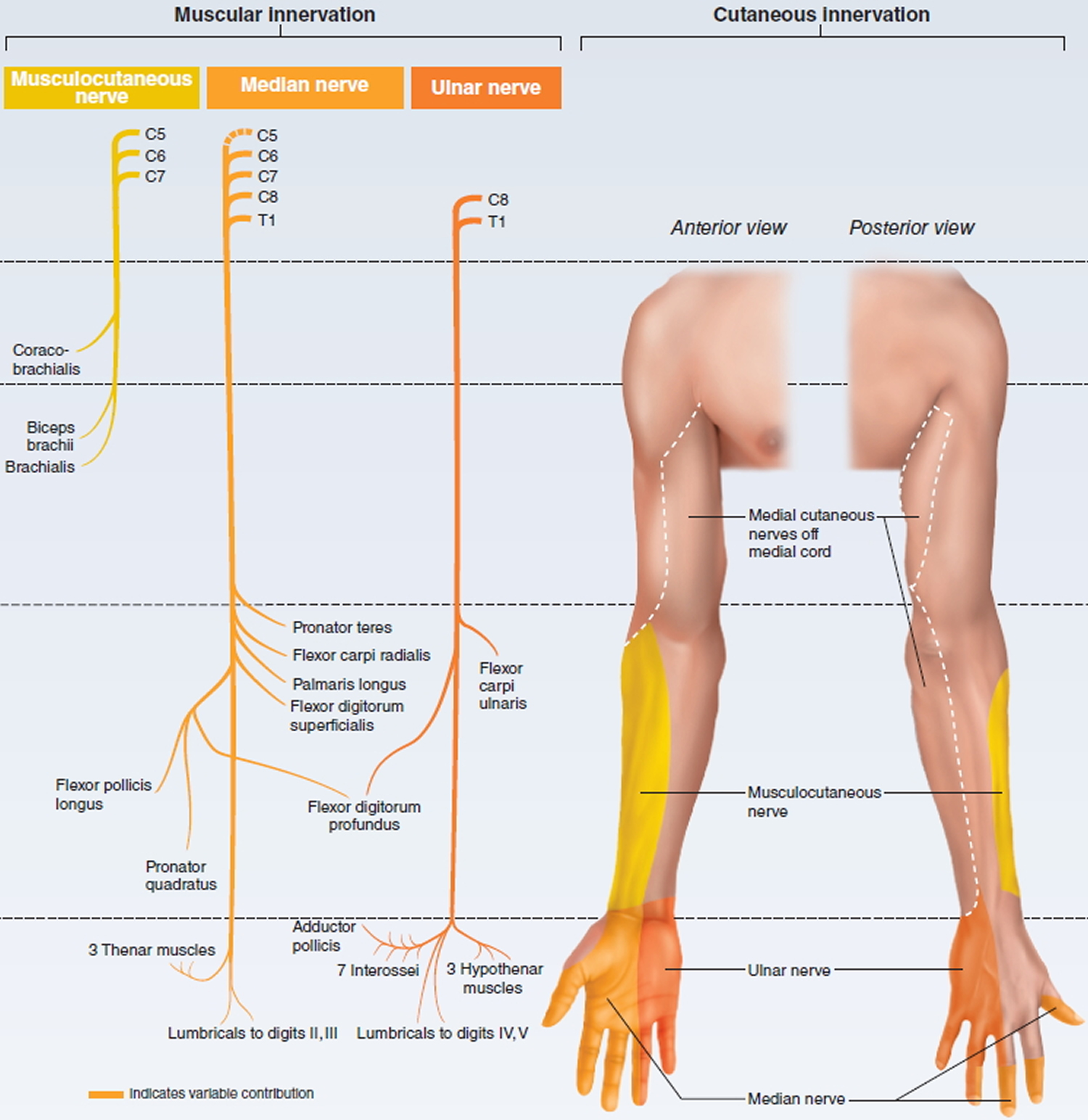
- (2) The musculocutaneous nerve supplies the anterior muscles of the arm.
- (3) The radial nerve supplies the muscles on the posterior aspect of the arm and forearm.
- (4) The median nerve supplies most of the muscles of the anterior forearm and some of the muscles of the hand.
- (5) The ulnar nerve supplies the anteromedial muscles of the forearm and most of the muscles of the hand.
Brachial plexus injury at birth
Birth brachial plexus injury also known as obstetric brachial plexus injury, is unfortunately a rather common injury in newborn children. Incidence varies between 0.15 and 3 per 1000 live births in various series and countries 1. Birth brachial plexus injury (obstetric brachial plexus palsy) is caused by traction to the brachial plexus during labour 2. In the majority of cases delivery of the upper shoulder is blocked by the mother’s pubic symphysis (shoulder dystocia). If additional traction is applied to the child’s head, the angle between the neck and the shoulder is forcefully widened, overstretching the ipsilateral brachial plexus. Injury to the superior roots of the brachial plexus (C5–C6) may result from forceful pulling away of the head from the shoulder, as might occur from excessive stretching of an infant’s neck during childbirth. The resulting traction injury may vary from neurapraxia (a temporary loss of motor and sensory function due to blockage of nerve conduction) or axonotmesis (a more severe injury where the nerve axons and their myelin sheath are damaged but the endoneurium, perineurium and epineurium remain intact) to neurotmesis (the most serious nerve injury both the nerve and the nerve sheath are disrupted. While partial recovery may occur, complete recovery is impossible) and avulsion of rootlets (most severe form of brachial plexus injury where the nerve roots are torn off the spinal cord) from the spinal cord. Most individuals with neuropraxia injuries recover spontaneously with a 90-100% return of function.
The generally accepted mechanism in cases of shoulder dystocia is traction to the neck caused by pull of the obstetrician’s hand or instruments like forceps or vacuum. In this scenario, the neck on the side of the anterior shoulder is stretched and this stretch causes a “strain” on the brachial plexus on that side, causing a varying degree of injury. This mechanism would typically cause anterior shoulder involvement, which would imply a higher incidence of right-sided injuries because left occipito anterior is the most common presentation. However, posterior shoulder involvement is seen frequently 3 and the right side is not the more common side in literature. In a cohort of 305 patients, 60% were right sided, 37 % were left sided and 3% were bilateral.
Therefore, other mechanisms have been looked at. For example, some papers have discussed whether it is really an intrauterine injury. Jennet et al. 3 have shown that almost half the cases they reviewed did not have shoulder dystocia and concluded that it could be caused by intrauterine maladaptation and not birth trauma. Others like Gherman 4 have described that birth brachial plexus injury has occurred following caesarean sections and also that shoulder dystocia does not always lead to birth brachial plexus injury. There is some electrophysiological evidence 5 to show that birth brachial plexus injury could have occurred in the intrauterine period since denervation potentials are seen on day 1 after delivery which ought not be possible in case it occurred at the moment of delivery. It has been shown that the posterior shoulder can get stuck on the Sacral promontory and cause injury through a stretch on that side 3 while the baby is in early stage of labor before any question about shoulder dystocia and traction. Bicornuate uterus has also given rise to birth brachial plexus injury with phrenic palsy 6.
In contrast, Gilbert et al. 7 found that the majority (92%) of the high-risk patients (diabetic women delivered by operative vaginal delivery with infants of 4.5 kg birth weight) did not have birth brachial plexus injury and caesarean delivery would have been unnecessary. Although macrosomia is commonly associated with birth brachial plexus injury, Rouse et al. 8 found no benefit with elective caesarean delivery in women with estimated fetal weights of 4.5 kg, unless they were also diabetic. Lindsay et al. 9 found that a policy of elective caesarean section for macrosomia would necessitate 148–258 caesarean sections to prevent a single persistent injury and avoidance of operative vaginal delivery would require 50–99 caesarean sections per injury prevented.
Thus, there is no agreement among scholars about the mechanism or active prevention and this has medico-legal implications. Lawyers for aggrieved parents routinely blame the obstetrician for the injury, but this may not be supported by evidence in the literature.
The injury caused to the roots of the plexus can be of a varied nature and can affect some or all roots. The classical injury is a C5 – C6 palsy, but all roots can be involved. The level and nature of root involvement varies from a neuropraxia to varying levels of axonotomesis to neurotomesis. In the worst injuries, even a root avulsion is possible. Clinical examination and electrophysiology with or without magnetic resonance imaging (MRI) can help in largely determining the type and extent of injury. Sunderland’s 10 well-known classification is useful to understand the nature of the injury. Broadly speaking, for the surgeon, there are three different kinds of lesions:
- Neuroma in continuity: This represents a postganglionic rupture, i.e. a Sunderland type 2, 3 or 4 injury which is healed with fibrosis and some axons may be attempting to go across the scar tissue. Occasionally, it is a conducting neuroma with useful function. However, if seen early enough, it is far better to resect and reconstruct.
- Rupture: This is a postganglionic neurotomesis, i.e. Sunderland type 5 causing a separation of proximal and distal ends often bridged by scar tissue.
- Avulsion: This is a preganglionic lesion showing avulsed ganglia in the neck or a pseudomeningocoele.
In birth brachial plexus injury spontaneous recovery is known, but there is a large subset which does not recover and needs primary or secondary surgical intervention. An extensive review of peer-reviewed publications has been done in this study, including clinical papers, review articles and systematic review of the subject. A systematic review by Pondaag et al. 11 concluded that no scientifically sound evidence exists to support the common perception of complete spontaneous recovery. In their opinion, the incidence of children requiring surgery was 20–30% rather than the 10% often mentioned in literature. Although all affected children do not require surgery in infancy, a substantial proportion of them, however, require it and are better off for it 12. Secondary surgery is needed for shoulder elbow and hand problems. Results of nerve surgery are very encouraging. Children with birth brachial plexus injury should be seen early by a hand surgeon dealing with brachial plexus injuries. Good results are possible with early and appropriate intervention even in severe cases.
Birth brachial plexus injury outlook (prognosis)
Most babies will fully recover within 3 to 9 months. Those who do not recover during this time have a poor outlook. In these cases, there may be a separation of the nerve root from the spinal cord (avulsion).
It is not clear whether surgery to fix the nerve problem can help. Nerve grafts and nerve transfers are sometimes tried in specialized centers with expertise in managing birth brachial plexus injury.
In cases of pseudoparalysis, the child will begin to use the affected arm as the fracture heals. Fractures in infants heal quickly and easily in most cases.
Birth brachial plexus injury possible complications
Complications include:
- Abnormal muscle contractions (contractures) or tightening of the muscles. These may be permanent.
- Permanent, partial, or total loss of function of the affected nerves, causing paralysis of the arm or arm weakness
Birth brachial plexus injury – Initial Treatment
Initial treatment consists of coming to a proper diagnosis, which includes a careful history taking, detailed clinical examination, including a check for associated injuries like fractures of the clavicle and humerus. Electrophysiology is recommended at 4 weeks initially to confirm the diagnosis and get a baseline reading of the involvement of various nerves and muscles as well as sensory parameters. Counseling of parents is critical and clear open communication is strongly recommended. Gentle mobilisation of all joints of the affected limb is suggested to avoid stiffness. “Gift wrapping” the baby in the typical traditional Indian style after morning bath is discouraged as it deprives the child of a chance to use that limb spontaneously. Repeat examinations are carried out every 4–6 weeks until 3 months. At this time, a decision is made about the need for surgery, which in turn depends on clinical and electrophysiological findings. MRI is considered very useful, at least in the adult palsy. However, clinical exam and electrophysiology can give adequate evidence of the status of the plexus and the indication for surgery.
Brachial plexus injury exercises
Gentle massage of the arm and range-of-motion exercises are recommended for mild cases. The infant may need to be seen by specialists if the damage is severe or the condition does not improve in the first few weeks.
Surgery may be considered if some strength has not returned to the affected muscles by the time the baby is 3 to 9 months old.
Indications for Surgery
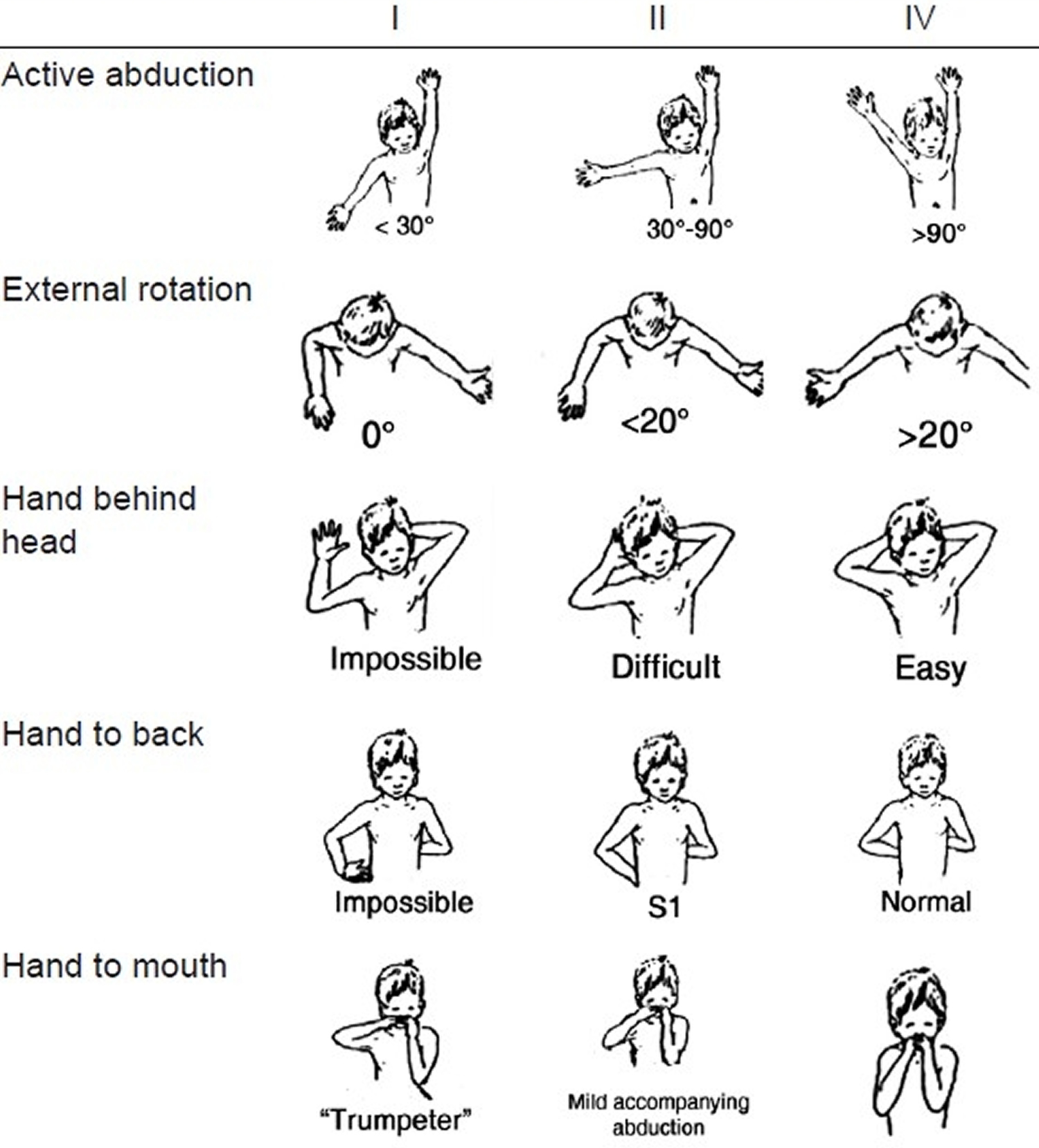
In case of global involvement, involvement of the hand or a flail upper limb, there is no real dispute about indication or timing of surgery. The dispute arises in the upper plexus lesions of Narakas Type I and II (see Table 1 below). The author follows Prof. Gilbert’s criteria of using the biceps brachii as an indicator of recovery in upper plexus lesions. Following the landmark theses by Tassin 13 it was generally agreed that a lack of antigravity biceps function at 3 months is an indication for surgery in C5-C6+/–C7 lesions. The logic behind this caveat is that the biceps is the only C5–C6 innervated muscle whose function cannot be duplicated by other muscles, and therefore is a good uncluttered indicator of C5-C6 recovery. The shoulder, for example, is moved by multiple muscles, which have various innervations and represent many neurotomes, and is therefore considered unsuitable for making this judgement call. Biceps recovery indicates the good health of C5–C6 and the upper trunk all the way to the upper arm. Clarke and his group 14 developed an alternative Active Movement Scale (AMS) in Toronto (see Table 2). They use this scoring and are prepared to wait for a much longer observation period to come to a decision. They also use the “Cookie” test around 8–9 months in children who are on the borderline of surgery and conservative treatment to make a final call regarding the indication of surgery. While there is merit in this system, the resource requirement in terms of physical and occupational therapists dedicated to this clinic, etc. exceeds that which is typically available. In addition, the system is cumbersome to administer at this point in time and may not offer a great advantage over Gilbert’s rather simpler indication.
Table 1. Narakas classification
Table 2. Toronto active movement scale of Clarke and Curtis
Waters, in a review 15, has summarized the issue with the following observation: “Infants who recover partial antigravity upper trunk muscle strength in the first 2 months of life should have a full and complete recovery over the first 1–2 years of life. Infants who do not recover antigravity biceps strength by 5–6 months of life should have microsurgical reconstruction of the brachial plexus, as successful surgery will result in a better outcome than natural history alone. Infants with partial recovery of C5–C6–C7 antigravity strength during months 3 through 6 of life will have permanent, progressive limitations of motion and strength; they also are at risk for the development of joint contractures in the affected limb.”
Thatte 12 uses Gilbert’s criteria for the decision, i.e. if a child with upper plexus lesion does not have a biceps function exceeding MRC grade 3 (antigravity movement) at 3 months of age, then surgery is recommended for nerve repair. In cases where there is visible biceps contraction but triceps co-contraction preventing elbow flexion, or biceps being 2+ but not really 3, etc., the surgeon can wait and watch with/without botox to triceps up to 6 months by which time the picture is clear. If the bicpes continues to be under MRC grade 3, then surgery is better than conservative treatment 12.
Birth brachial plexus injury – Surgical Treatment
Surgery in these indications consists of a complete exploration of the supra and infraclavicular plexus and nerve repair using microsurgical techniques based on merits, i.e. after assessing the injury and its pathophysiology, the surgeon can decide on the strategy to be used. Thatte et al. 12 have recently published a detailed paper describing the technique for exposure of the brachial plexus, which can be used for further reading. Typically, the preference in a total palsy is to hand function, while in an upper plexus it is of course to reconstruct the nerves for elbow and shoulder function. The suprascapular nerve supplying the supra and infraspinatus is a crucial nerve for shoulder abduction and external rotation and needs to be targeted with priority, either from one of the roots or separately with the spinal accessory nerve as the donor. The lower trunk/medial cord for hand and musculocutaneous nerve for biceps are the other priorities.
Intraplexus neurotisation thus uses existing healthy root stumps, which are ruptured for reconstruction by joining them with the distal target trunks, cords or nerves using nerve grafts. The source of nerve grafts is both sural nerves and the ipsilateral medial cutaneous nerve of forearm (MCNF) and superficial radial nerve (SRN), especially in global severe palsies if grafts are falling short. Not all cases may be of rupture; there can be avulsions as well, and therefore no roots may be available. Depending on the number of roots available, a policy is evolved for neurotisation.
Global palsy strategy
- Four or five roots available: Very rare; just direct them to respective trunks/cords.
- Three roots available: One each to medial, lateral and posterior cords. Spinal accessory to suprascapular nerve depending on the pathoanatomy of the upper trunk lesion.
- Two roots available: Root 1 to lower trunk/medial cord, Root 2 to lateral cord (or shared between lateral and posterior cords) and spinal accessory to suprascapular nerve. Some authorities would prefer Root 2 to posterior division of upper trunk/posterior cord and 2 or 3 intercostal nerves to biceps. Their point is getting a strong triceps and a stable shoulder with deltoid and triceps as well as the rotator cuff is important to balance a well-recovered biceps. This is logical; however, the addition of intercostal nerves adds to surgical time, blood loss and increases surgical risks and morbidity and needs to considered carefully before attempting. In well-equipped centres with blood bank support, ICU for children and experienced anaesthesiologists, it can be done if the child is fit.
- One root available: Root to lower trunk/medial cord + spinal accessory to suprascapular nerve, 2 or 3 intercostal nerves to biceps. Shoulder will need secondary transfers if possible.
- Zero roots – rare total avulsion: Opp. C7 root (posterior division) to lower trunk/medial cord and spinal accessory to musculocutaneous nerve or spinal accessory to suprascapular nerve and intercostal nerves to musculocutaneous nerve plus one intercostal nerve to long head of triceps. If the child is healthy and fit for prolonged anaesthesia-or stagger in a second session in a couple of months if there is any concern on this count.
- Upper plexus: C5–C6
- Both roots available: Typically C5 to anterior division of upper trunk and C6 to posterior division of upper trunk.
- One root available: Root to anterior division of upper trunk (or both divisions if they are of really good quality with plenty of healthy axons) and XI nerve to suprascapular nerve. Intercostal nerves can be used for additional neurotisation depending on situation and fitness.
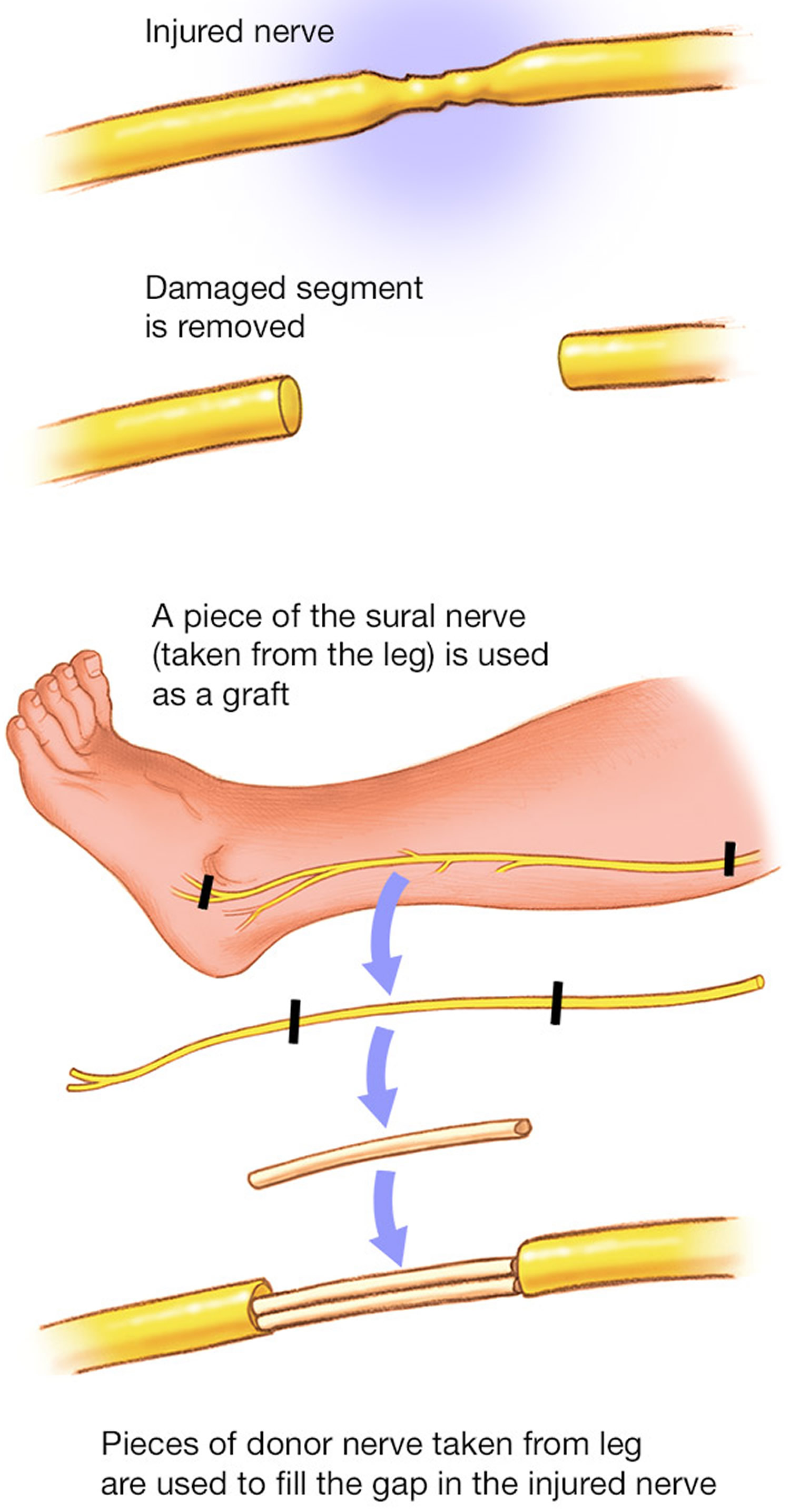
Figure 6. Nerve graft – Nerve tissue can be removed from other parts of your body to replace the damaged portions of your brachial plexus nerves.
Distal nerve transfers
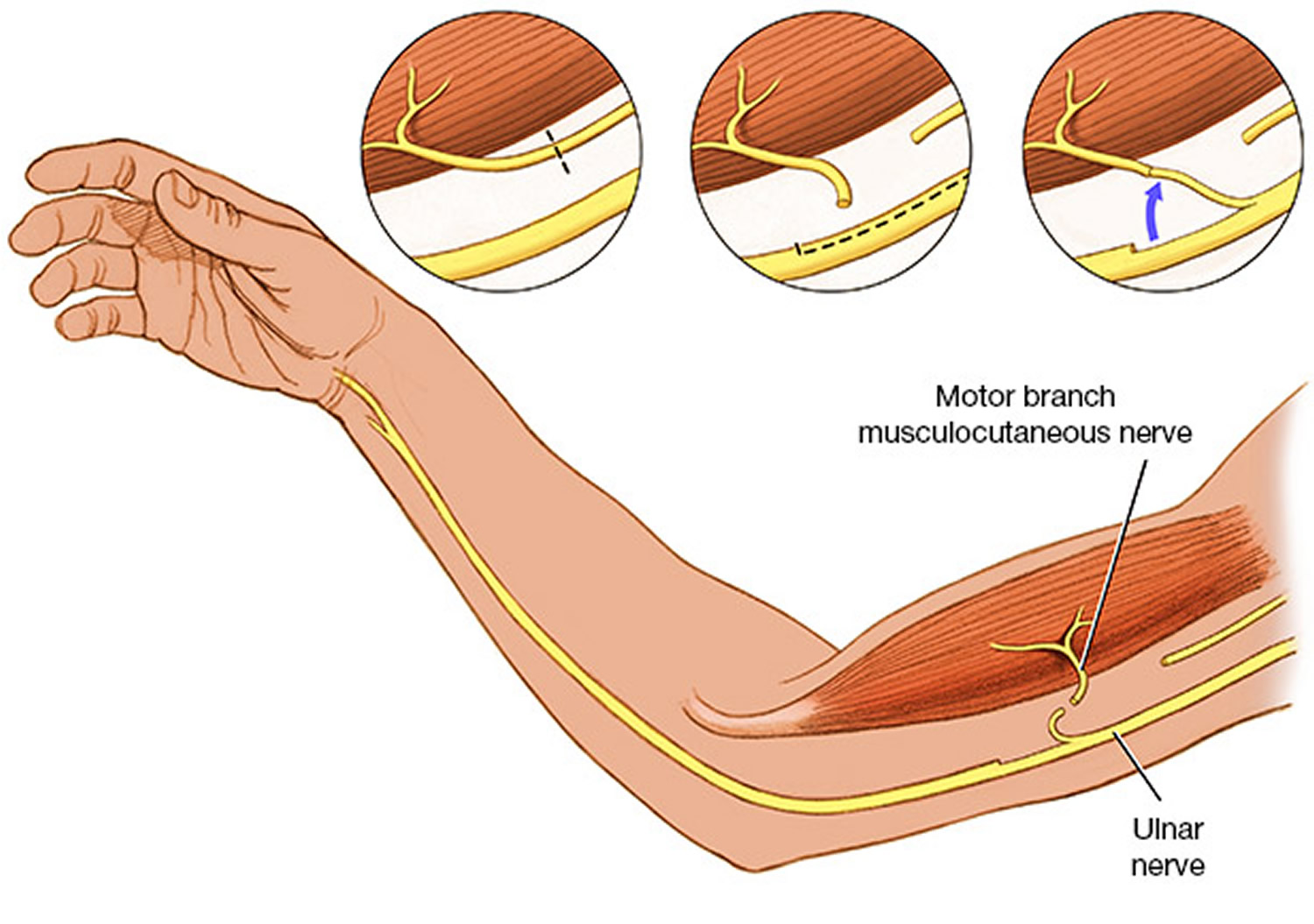
The entire protocol given above is without considering the additional use of distal nerve transfers in upper plexus lesion. Following the description by Oberlin 16 and Somsak 17 of nerve transfers for elbow flexion and deltoid reinnervation, the whole landscape in adult plexus injury changed dramatically. This philosophy is now coming in play in the birth brachial plexus injury reconstruction as well in selected cases. Oberlin 16 described the use of motor fascicles of the ulnar nerve for reinnervation of the biceps via the musculocutaneous nerve.
Subsequently, this has been modified to ulnar to biceps and median to brachialis. Somsak 17 described the use of the branch to long head of triceps for the anterior division of the axillary nerve to reinnervate the deltoid. The interesting point about distal transfers is the speed of neurotisation, since the nerve is coapted very close to the hilum of the muscle and the growing axons reach the end plates much faster.
In upper plexus lesions (C5, C6), these transfers allow for leaving the neuroma undisturbed and doing a triple transfer consisting of spinal accessory to suprascapular nerve, Oberlin and Somsak transfers to reconstruct shoulder abduction and external rotation, as well as elbow flexion. If the C7 is involved, then the triceps is often not functioning and the Somsak transfer is not possible; it also often means wrist extension is affected. In such cases, intercostal nerves can be used for the deltoid as described by Somsak 18 and intraplexal neurotisation can be used for the C7 posterior division to help reinnervate both triceps and wrist extension.
There is considerable disagreement about this. The two European doyens, Gilbert and Raimondi 19, argue that if roots are available, it is preferable to do a classical reinnervation and reserve distal transfers for later use. This allows reinnervation of all muscles supplied by those trunks/cords in a proper manner and yet leaves the distal transfers in reserve in case of unfavourable outcome. As mentioned earlier, distal transfers can be done at a late stage as they reach the muscle very rapidly. There is also no conclusive evidence in the form of a properly conducted prospective study to show that distal transfers give better results compared to classical neurotisation in the long run.
In the Indian scenario, distal transfers have a great benefit of targeting select muscles with no visible function without cutting the neuroma. Thus, for late referrals where some movements are seen, these are not lost even temporarily due to resection of the neuroma. This has social implications in this country because many parents and grandparents are not willing to risk losing existing movements (however inadequate they may be) to gain an eventual better result. Gilbert has reported similar social taboos in the Middle East.
Figure 7. Nerve transfer – Nerve transfers are particularly helpful in the most serious types of brachial plexus injuries, called avulsions, when the nerve root has been torn out of the spinal cord. They may also be used when surgeons are trying to speed up muscle recovery.
Secondary surgery
Secondary surgery is performed to treat either untreated older children or as a follow-up to primary nerve reconstruction. Secondary surgery involves surgery on shoulder, elbow as well as the hand.
Shoulder
Typically, shoulder surgery is performed to treat an internal rotation contracture combined with poor abduction. This is typically caused by co-contraction of abductors with adductors and internal with external rotators. The deltoid and supraspinatus along with the trapezius and serratus anterior are involved in various stages of shoulder abduction. The infraspinatus and teres minor are external rotators, while four big muscles, viz. pectoralis major, teres major, latissimus dorsi and subscapularis, are involved in adduction and internal rotation. In the recovering plexus (both natural and in those with nerve reconstruction), there is a tendency of mix-up of the growing axons to target muscles. This results in co-contraction of these groups. In such a battle, the four big muscles mentioned above are the invariable winners leading to an internal rotation contracture and an inability to abduct fully. If the anterior and posterior axillary folds of these children are felt during an attempted abduction, this co-contraction of one or both folds (containing the pectoralis major, teres major and latissimus dorsi, respectively) can be easily felt and helps determine the choice of muscle to release and transfer. Internal rotation contracture, if allowed to persist, leads to glenoid deformation and posterior subluxation of the humeral head. It is advisable to prevent this by early surgery in these instances to restore function and avoid deformity. Mallet’s score 20 is often used to assess the shoulder function before and after surgery (Table 3). Gilbert 21 also used a scoring system for shoulder (Table 4).
Table 3. The Mallet score
Table 4. Gilbert score for shoulder results
Elbow
Surgery is directed towards restoration of either flexion or extension. For elbow flexion, typical transfers are triceps to biceps and Steindler’s flexoroplasty. Rarely, an latissimus dorsi transfer can be theoretically used, but the author has no experience of it in birth brachial plexus injury. For extension, the transfer of choice is deltoid to triceps. The posterior half of the deltoid is detached and extended with fascia lata to attach it to the triceps insertion.
Hand
Raimondi 22 (Table 5) describes an excellent system of assessing the hand. A plethora of tendon transfers can be used to improve hand function. They are based on standard tendon transfers done for peripheral nerve lesions and very valuable insights gained through the voluminous Indian work on leprosy tendon transfers. But there are a few caveats for transfers in the hand following birth brachial plexus injury, given as follows:
- Birth brachial plexus injury cases have relatively weaker donors to achieve the desired function as the donor muscle too is often reinnervated. Full function, therefore, may not be achieved.
- Children keep recovering hand function for 3–4 years both naturally as well as after nerve surgery (unless of course you know that a particular nerve is not targeted at all).
- It is better to splint with appropriate splints in the interim.
- Start transfers only after about 3.5–4 years to get optimum results.
Table 5. Raimondi score for hand function
Brachial plexus injury causes
Damage to the upper nerves that make up the brachial plexus tends to occur when your shoulder is forced down while your neck stretches up and away from the injured shoulder. The lower nerves are more likely to be injured when your arm is forced above your head. These injuries can occur in several ways, including:
- Contact sports. Many football players experience burners or stingers, which can occur when the nerves in the brachial plexus get stretched beyond their limit during collisions with other players.
- Difficult births. Newborns can sustain brachial plexus injuries when there are problems during birth, such as a breech presentation or prolonged labor. If an infant’s shoulders get wedged within the birth canal, there is an increased risk of a brachial plexus palsy. Most often, the upper nerves are injured, a condition called Erb’s palsy. Total brachial plexus birth palsy occurs when both the upper and lower nerves are damaged.
- Trauma. Several types of trauma — including motor vehicle accidents, motorcycle accidents, falls or bullet wounds — can result in brachial plexus injuries.
- Inflammation. Inflammation may cause damage to the brachial plexus. A rare condition known as Parsonage-Turner syndrome (brachial plexitis) causes brachial plexus inflammation with no trauma and results in paralysis of some muscles of the arm.
- Tumors. Noncancerous (benign) or cancerous tumors can grow in the brachial plexus or put pressure on the brachial plexus or spread to the nerves, causing damage to the brachial plexus.
- Radiation treatment. Radiation treatment may cause damage to the brachial plexus.
Risk factors for brachial plexus injury
Participating in contact sports, particularly football and wrestling, or being involved in high-speed accidents increases your risk of brachial plexus injury.
Brachial plexus injury prevention
Although damage to your brachial plexus often can’t be prevented, you can take steps to reduce the risk of complications once an injury has occurred:
- For yourself. If you temporarily lose the use of your hand or arm, daily range-of-motion exercises and physical therapy can help prevent joint stiffness. Avoid burns or cuts, as you may not feel it if you’re experiencing numbness. If you’re an athlete who has experienced several injuries to the brachial plexus area, your doctor may suggest you wear specific padding to protect the area during sports.
- For your child. If you’re the parent of a child with a brachial plexus palsy, it’s important that you exercise your child’s joints and functioning muscles every day, beginning when your baby is about 3 weeks old. This helps prevent the joints from becoming permanently stiff and keeps your child’s working muscles strong and healthy.
Brachial plexus injury signs and symptoms
Signs and symptoms of a brachial plexus injury can vary greatly, depending on the severity and location of your injury. Usually only one arm is affected.
Less severe injuries
Minor damage often occurs during contact sports, such as football or wrestling, when the brachial plexus nerves get stretched or compressed. These are called stingers or burners, and can produce the following symptoms:
- A feeling like an electric shock or a burning sensation shooting down your arm
- Numbness and weakness in your arm
These symptoms usually last only a few seconds or minutes, but in some people may linger for days or longer.
More-severe injuries
More-severe symptoms result from injuries that seriously injure or even tear or rupture the nerves. The most serious brachial plexus injury (avulsion) occurs when the nerve root is torn from the spinal cord.
Signs and symptoms of more-severe injuries can include:
- Weakness or inability to use certain muscles in your hand, arm or shoulder
- Complete lack of movement and feeling in your arm, including your shoulder and hand
- Severe pain.
Brachial plexus injuries complications
Given enough time, many brachial plexus injuries in both children and adults heal with no lasting damage. But some injuries can cause temporary or permanent problems:
- Stiff joints. If you experience paralysis of your hand or arm, your joints can stiffen, making movement difficult, even if you regain use of your limb. For that reason, your doctor is likely to recommend ongoing physical therapy during your recovery.
- Pain. This results from nerve damage and may become chronic.
- Loss of feeling. If you lose feeling in your arm or hand, you run the risk of burning or injuring yourself without knowing it.
- Muscle atrophy. Slow-growing nerves can take several years to heal after injury. During that time, lack of use may cause the affected muscles to break down (degenerate).
- Permanent disability. How well you recover from a serious brachial plexus injury depends on a number of factors, including your age and the type, location and severity of the injury. Even with surgery, some people experience permanent disability, ranging from weakness in the hand, shoulder or arm to paralysis.
Brachial plexus diagnosis
To diagnose your condition, your doctor will review your symptoms and conduct a physical examination.
To help diagnose the extent and severity of a brachial plexus injury, you may have one or more of the following tests:
- Electromyography (EMG). During an EMG, your doctor inserts a needle electrode through your skin into various muscles. The test evaluates the electrical activity of your muscles when they contract and when they’re at rest. You may feel a little pain when the electrodes are inserted, but most people can complete the test without much discomfort.
- Nerve conduction studies. These tests are usually performed as part of the EMG, and measure the speed of conduction in your nerve when a small current passes through the nerve. This provides information about how well the nerve is functioning.
- Magnetic resonance imaging (MRI). This test uses powerful magnets and radio waves to produce detailed views of your body in multiple planes. It often can show the extent of the damage caused by a brachial plexus injury and can help assess the status of arteries that are important for the limb or for reconstruction of it. New methods of high-resolution MRI, known as magnetic resonance neurography, may be used.
- Computerized tomography (CT) myelography. Computerized tomography uses a series of X-rays to obtain cross-sectional images of your body. CT myelography adds a contrast material, injected during a spinal tap, to produce a detailed picture of your spinal cord and nerve roots during a CT scan. This test is sometimes performed when MRIs don’t provide adequate information.
- Angiogram. If your doctor suspects that the blood vessels feeding your arm might be injured, he or she might suggest an angiogram — an imaging test where contrast material is injected into an artery or vein to check the condition of your blood vessels. This information is important in planning your surgical procedure.
Brachial plexus injury treatment
Treatment depends on several factors including the severity of the injury, the type of injury, the length of time since the injury and other existing conditions.
Nerves that have only been stretched may recover without further treatment.
Your doctor may recommend physical therapy to keep your joints and muscles working properly, maintain the range of motion, and prevent stiff joints.
The healing process sometimes forms scar tissue that must be removed surgically to improve the nerve’s function. Surgical repair is often required for nerves that have significant surrounding scar tissue or that have been cut or torn.
Surgery to repair brachial plexus nerves should generally occur within six to seven months after the injury. If nerve surgery occurs more than six to seven months after the injury, the muscles may not recover their function.
Types of surgery
Nerve graft. In this procedure, the damaged part of the brachial plexus is removed and replaced with sections of nerves harvested from other parts of your body (see Figure 6 above). This helps restore your arm’s function.
Nerve transfer. When the nerve root has been torn from the spinal cord, surgeons often take a less important nerve that’s still attached to the spinal cord and connect it to the nerve that’s no longer attached to the spinal cord (see Figure 7 above).
In some cases, surgeons may perform this technique at a level close to the targeted muscle in an effort to speed up recovery rather than doing a repair (nerve graft) farther from the muscle. Sometimes doctors may perform a combination of nerve graft and nerve transfer.
Nerve tissue grows slowly, about an inch (2.5 centimeters) a month, so it can take several years to know the full benefit of surgery. During the recovery period, you must keep your joints flexible with a program of exercises. Splints may be used to keep your hand from curling inward.
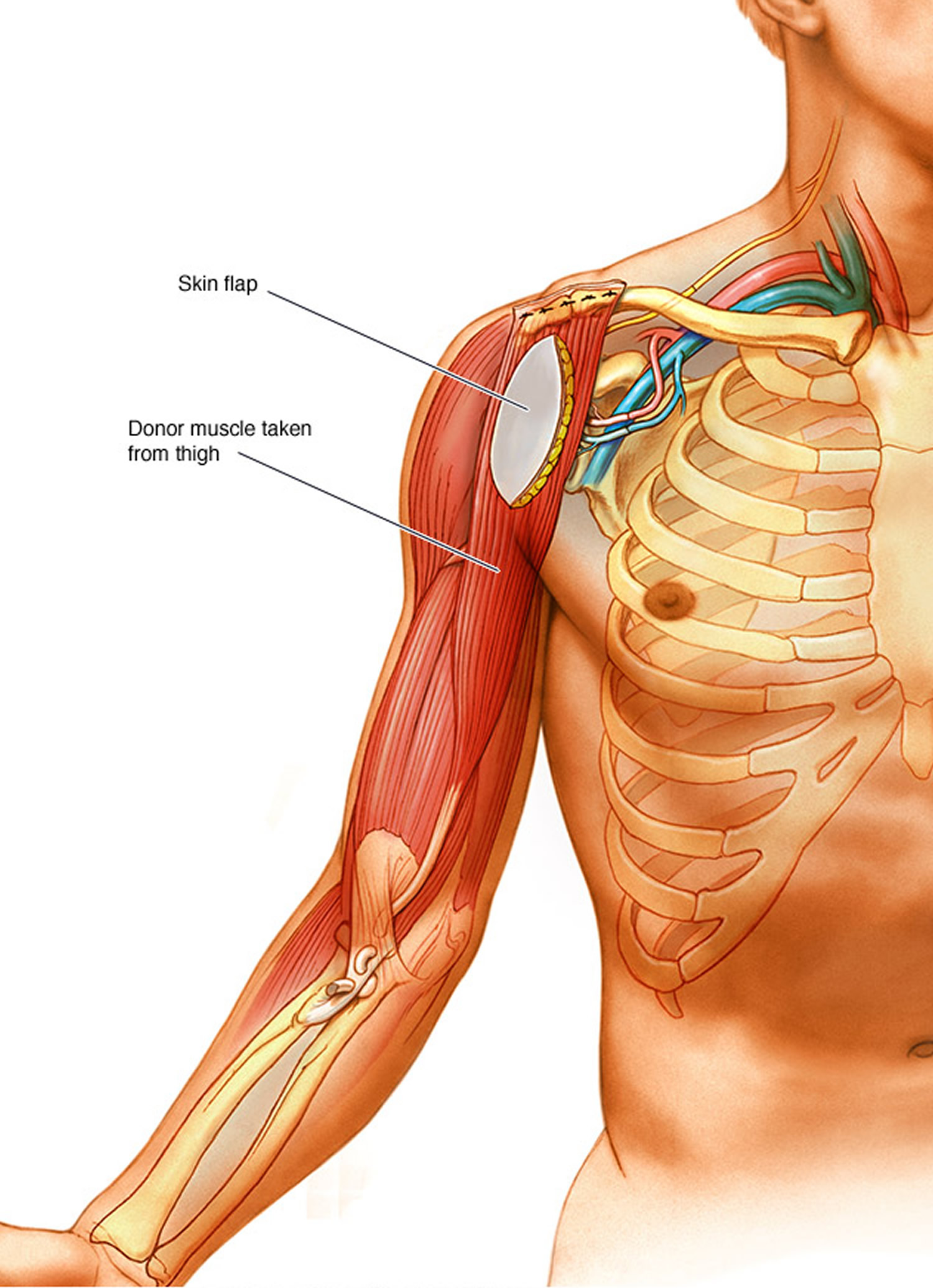
Muscle transfer. Muscle transfer is a procedure in which your surgeon removes a less important muscle or tendon from another part of your body, typically the thigh, transfers it to your arm, and reconnects the nerves and blood vessels supplying the muscle (Figure 8). If your arm muscles have atrophied from lack of use, a muscle transfer may be needed. The most commonly used donor muscle is in the inner thigh. A section of skin and tissue attached to the donor muscle may also be removed. This skin flap can help the surgeons monitor whether the muscle is getting enough blood after it’s been transferred to its new location.
Figure 8. Muscle transfer
Pain control
Pain from the most severe types of brachial plexus injuries has been described as a debilitating, severe crushing sensation or a constant burning. Narcotic medications are often used initially, but may be changed as your recovery progresses to optimize pain relief.
References- Pondaag W, Malessy M, van Dijk JG, Thomeer R. Natural history of obstetric brachial plexus palsy: A systematic review. Dev Med Child Neurol. 2004;46:138–44. http://onlinelibrary.wiley.com/doi/10.1111/j.1469-8749.2004.tb00463.x/pdf
- Metaizeau JP, Gayet C, Plenat F. (1979) Les lesions obstétricales du plexus brachial. Chir Pediatr 20:159–163. (In French).
- Jennett RJ, Tarby TJ, Kreinick CJ. Brachial plexus palsy: An old problem revisited. Am J Obstet Gynecol. 1992;166:1673–6. https://www.ncbi.nlm.nih.gov/pubmed/1309174
- Gherman RB, Murphy Goodwin T, Ouzounian JG, Miller DA, Paul RH. Brachial plexus palsy associated with cesarean section: An in utero injury? Am J Obstet Gynecol. 1997;177:1162–4. https://www.ncbi.nlm.nih.gov/pubmed/9396913
- Koenigsberger MR. Brachial plexus palsy at birth: Intrauterine or due to delivery trauma? Ann Neurol. 1980;8:228.
- Dunn DW, Engle WA. Brachial plexus palsy: Intrauterine onset. Pediatr Neurol. 1985;1:367–9. https://www.ncbi.nlm.nih.gov/pubmed/3880422
- Gilbert M, Nesbitt T, Danielsen B. Associated Factors in 1611 Cases of Brachial Plexus Injury. Obstet Gynecol. 1999;93:536–40. https://www.ncbi.nlm.nih.gov/pubmed/10214829
- Rouse DJ, Owen J, Goldenberg RL, Cliver SP. The effectiveness and costs of elective cesarean delivery for fetal macrosomia diagnosed by ultrasound. JAMA. 1996;276:1480–6. https://jamanetwork.com/journals/jama/article-abstract/410522
- Lindsay B, Kolderup LB, Laros RK, Musci TJ. Incidence of persistent birth injury in macrosomic infants: Association with mode of delivery. Am J Obstet Gynecol. 1997;177:37–42. https://www.ncbi.nlm.nih.gov/pubmed/9240580
- Sunderland S. Nerves and Nerve Injuries. London: Churchill Livingstone; 1978.
- Pondaag W, Malessy M, van Dijk JG, Thomeer R. Natural history of obstetric brachial plexus palsy: A systematic review. Dev Med Child Neurol. 2004;46:138–44. http://onlinelibrary.wiley.com/doi/10.1111/j.1469-8749.2004.tb00463.x/epdf
- Thatte MR, Mehta R. Obstetric brachial plexus injury. Indian Journal of Plastic Surgery : Official Publication of the Association of Plastic Surgeons of India. 2011;44(3):380-389. doi:10.4103/0970-0358.90805. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3263264/
- Tassin JL. Thesis. Paris: Universite Paris VII; 1983. Paralysies obstetricales du plexus brachial: Evolution spontanee, resultats des interventions reparatrices precoces.
- Clarke HM, Curtis CG. An approach to obstetrical brachial plexus injuries. Hand Clin. 1995;11:563–81. https://www.ncbi.nlm.nih.gov/pubmed/8567739
- Waters PM. Update on management of pediatric brachial plexus palsy. J Pediatr Orthop. 2005;25:116–26. https://www.ncbi.nlm.nih.gov/pubmed/15614072
- Oberlin C, Beal D, Leechavengvongs S, Salon A, Dauge MC, Sarcy JJ. Nerve transfer to biceps muscle using a part of ulnar nerve for C5/C6 avulsion of the brachial plexus. Anatomical study and report of 4 cases. J Hand Surg. 1994;19:232–7. https://www.ncbi.nlm.nih.gov/pubmed/8201186
- Leechavengvongs S, Witoonchart K, Uerpairojkit C, Thuvasethakul P. Nerve transfer to deltoid muscle using the nerve to the long head of the triceps, part II: A report of 7 cases. J Hand Surg Am. 2003;28:633–8. https://www.ncbi.nlm.nih.gov/pubmed/12877852
- Malungpaishrope K, Leechavengvongs S, Uerpairojkit C, Witoonchart K, Jitprapaikulsarn S, Chongthammakun S. Nerve transfer to deltoid muscle using the intercostal nerves through the posterior approach: An anatomic study and two case reports. J Hand Surg Am. 2007;32:218–24. https://www.ncbi.nlm.nih.gov/pubmed/17275597
- Gilbert A, Raimondi P. Presentations in Club Narakas meeting Lisbon Mallet, J. Paralysie obstandricale. Revue de Chimrgie Orthopedique et Reparatrice de L’Appareil Moteur. 2011;58(Suppl):116.
- Pagnotta A, Haerle M, Gilbert A. Long-term results on abduction and external rotation of the shoulder after latissimus dorsi transferfor sequelae of obstetric palsy. Clin Orthop Relat R. 2004;426:199–205. https://www.ncbi.nlm.nih.gov/pubmed/15346074
- Thatte MR, Agashe MV, Rao A, Rathod CM, Mehta R. Clinical outcome of shoulder muscle transfer for shoulder deformities in obstetric brachial plexus palsy: A study of 150 cases. Indian J Plast Surg. 2011;44:21–8. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3111117/
- Raimondi P. Presented at the International Meeting on Obstetric Brachial Plexus Palsy. The Netherlands: Heerlen; 1993. Evaluation of results in obstetric brachial plexus palsy: The hand.