What is Budd Chiari syndrome
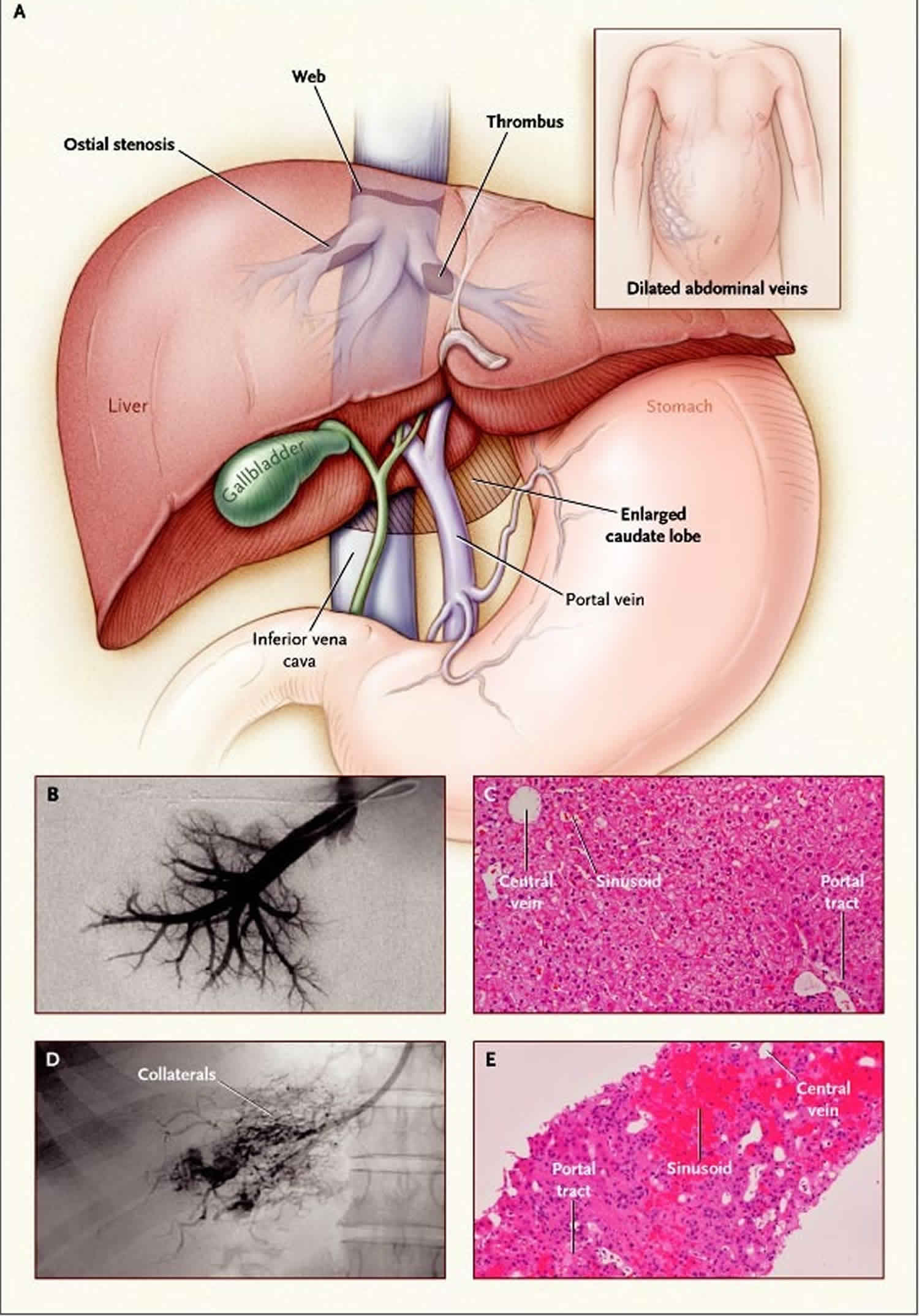
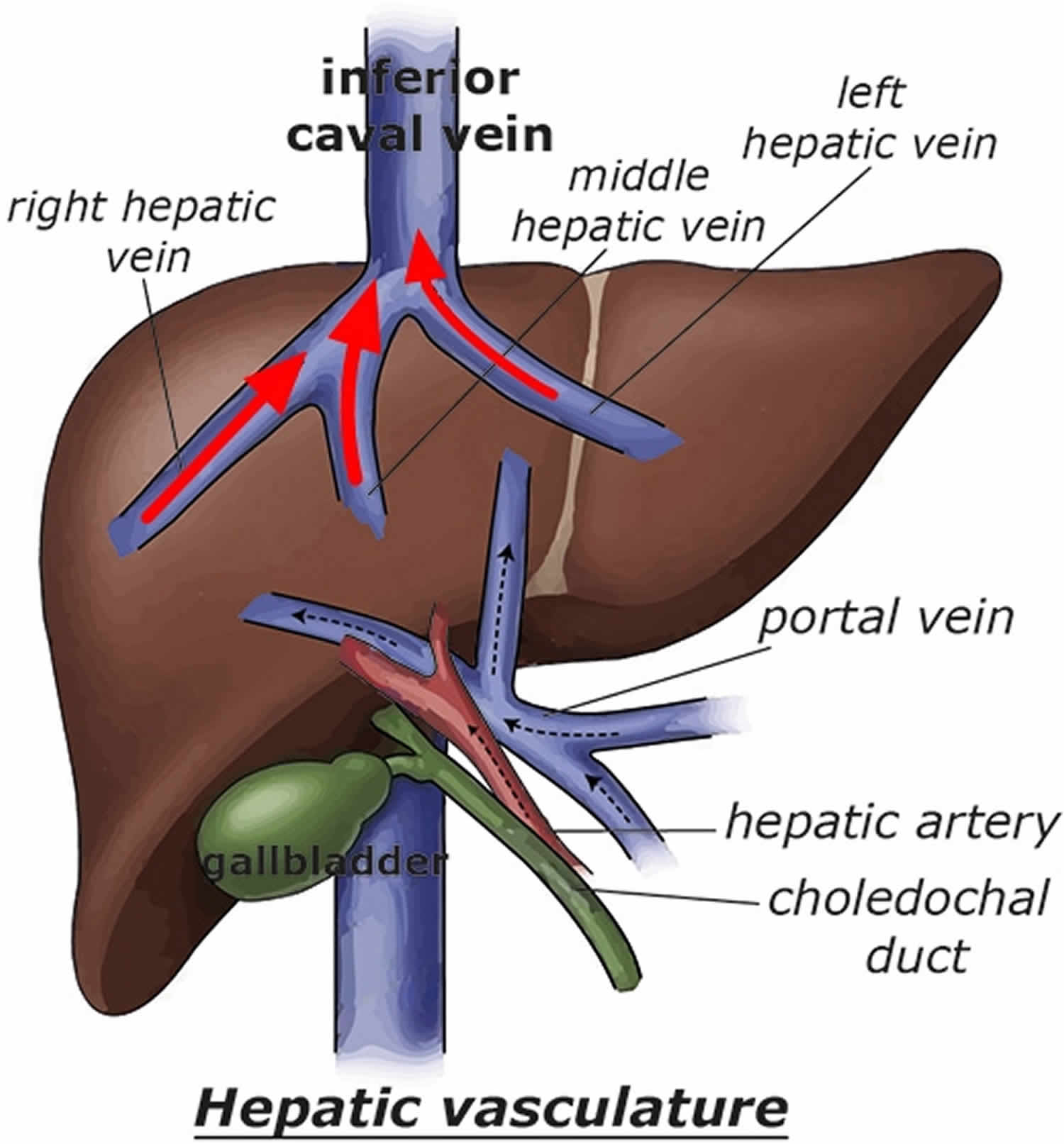
Budd-Chiari syndrome is obstruction of the hepatic vein, which carries blood away from the liver and the obstruction can originates anywhere from the small hepatic veins inside the liver to the inferior vena cava and right atrium. Hepatic vein obstruction prevents blood from flowing out of the liver and back to your heart. This blockage can cause liver damage. When the blood flow out of the liver is impeded, blood backs up in the liver, causing it to enlarge (hepatomegaly). The spleen may also enlarge (splenomegaly). This backup of blood increases blood pressure in the portal vein, which carries blood to the liver from the intestines (portal hypertension), and result in dilated, twisted veins in the esophagus (esophageal varices). Portal hypertension, leads to fluid accumulating in the abdomen (called ascites). The clot may extend to also block the inferior vena cava (the large vein that carries blood from the lower parts of the body to the heart). Varicose veins in the abdomen near the skin’s surface may develop and become visible. In some cases, scarring of the liver (cirrhosis) occurs. Other symptoms may include fatigue, abdominal pain, nausea, jaundice and bleeding in the esophagus 1.
The severity of Budd Chiari syndrome varies from case to case, depending on the site and number of affected veins 2. Obstruction of the hepatic vein most often occurs in patients which have a disorder that makes blood more likely to clot, such as those who are pregnant or who have a tumor or growth pressing on the vessel, a chronic inflammatory disease, a clotting disorder, a clot in the vessel (hepatic vein thrombosis), an infection, or a myeloproliferative disorder. In about one third of the cases, the cause of Budd-Chiari syndrome is unknown.
Budd-Chiari syndrome symptoms range from no symptoms to fulminant liver failure. Budd-Chiari syndrome diagnosis is based on ultrasonography. Treatment includes supportive medical therapy and measures to establish and maintain venous patency, such as thrombolysis, decompression with shunts, and long-term anticoagulation. In some cases liver transplantation is needed 1.
Figure 1. Hepatic vein
Budd Chiari syndrome causes
Hepatic veins and the adjacent inferior vena cava obstruction due to blood clot is the most common cause of Budd-Chiari syndrome. However, 25% arise from external compression that results in hepatic veins obstruction.
Most often, clots commonly result from the following conditions:
- Abnormal growth of cells in the bone marrow (e.g., myeloproliferative disorders such as polycythemia and paroxysmal nocturnal hemoglobinopathy)
- Arsenic
- Blood poisoning (sepsis)
- Cancers
- Chronic inflammatory or autoimmune diseases
- Connective tissue disorders
- Exposure to radiation
- Inflammatory bowel disease
- Infections (e.g., hydatid cyst, amebiasis)
- Inherited (hereditary) or acquired problems with blood clotting (e.g., protein C or S deficiency, antiphospholipid syndrome, antithrombin 3 deficiency, factor V Leiden mutation, pregnancy, oral contraceptive use)
- Oral contraceptives (birth control pills)
- Pregnancy
- Pyrrolidizine plant alkaloids (bush tea) used in some parts of the world as a drink
- Some cancer chemotherapy drugs
- Trauma
- Tumor invasion of the hepatic vein (e.g., hepatocellular or renal cell carcinoma)
- Vinyl chloride monomer
Sometimes Budd-Chiari syndrome begins during pregnancy and unmasks a previously asymptomatic hypercoagulability disorder.
The cause of hepatic veins obstruction is often unknown. In Asia and South Africa, the basic defect is often a membranous obstruction (webs) of the inferior vena cava above the liver, likely representing recanalization of a prior thrombus in adults or a developmental flaw (e.g., venous stenosis) in children. This type of obstruction is called obliterative hepatocavopathy.
Budd-Chiari syndrome usually develops over weeks or months. When it does develop over a period of time, cirrhosis and portal hypertension tend to develop.
Budd Chiari syndrome symptoms
Budd-Chiari syndrome symptoms range from no symptoms (asymptomatic) to fulminant liver failure or cirrhosis. Symptoms vary depending on whether the hepatic veins obstruction occurs acutely or over time. The classic acute presentation of Budd-Chiari syndrome is with the clinical triad of ascites, hepatomegaly, and abdominal pain, although this is non-specific 3.
Acute obstruction (in about 20%) results from an acute thrombosis of the main hepatic veins or the inferior vena cava. Acute obstruction causes fatigue, right upper quadrant pain, nausea, vomiting, mild jaundice, tender hepatomegaly, and rapid onset ascites. It typically occurs during pregnancy. Fulminant liver failure with encephalopathy is rare. Aminotransferase levels are quite high.
Chronic outflow obstruction (developing over weeks to months) is related to fibrosis of the intrahepatic veins, presumably related to inflammation. Chronic outflow obstruction may cause few or no symptoms until it progresses, or it may cause fatigue, abdominal pain, and hepatomegaly. Lower-extremity edema and ascites may result from venous obstruction, even in the absence of cirrhosis. Cirrhosis may develop, leading to variceal bleeding, massive ascites, splenomegaly, hepatopulmonary syndrome, or a combination. Complete obstruction of the inferior vena cava causes edema of the abdominal wall and legs plus visibly tortuous superficial abdominal veins from the pelvis to the costal margin.
If the liver has had time to develop collaterals and decompress, patients can be asymptomatic (≤20%) or present with few symptoms 3. As Budd-Chiari syndrome progresses, however, it can lead to liver failure and portal hypertension with corresponding symptoms (eg, encephalopathy, hematemesis).
Budd-Chiari syndrome symptoms may include:
- Abdominal swelling or stretching due to fluid in the abdomen (ascites)
- Pain in the right upper abdomen due to swollen and tender liver
- Vomiting blood
- Yellowing of the skin (jaundice)
Symptoms associated with Budd-Chiari syndrome include pain in the upper right part of the abdomen, an abnormally enlarged liver (hepatomegaly), yellowing of the skin and the whites of the eyes (jaundice), and/or accumulation of fluid in the space (peritoneal cavity) between the two layers of the membrane that lines the stomach (ascites). In some cases, there may be increased blood pressure in the veins carrying blood from the gastrointestinal (GI) tract back to the heart through the liver (portal hypertension). In some cases, impaired liver function may also develop. Liver biopsy tests show central cell deterioration, development of fibrous growths and blockage (occlusion) of the terminal liver veins. When vein blockage is severe, onset of the disorder can be sudden, and may be accompanied by severe pain. If the disease is chronic, onset may be gradual. In some cases, there may also be unusual swelling due to abnormal accumulation of fluid (edema) within the tissues of the legs.
In some cases, affected individuals may have scarring (fibrosis) and impaired functioning of the liver (cirrhosis) before or following the development of Budd-Chiari syndrome.
Budd Chiari syndrome possible complications
Hepatic vein obstruction can get worse and lead to cirrhosis and liver failure. This can be life threatening.
Budd Chiari syndrome diagnosis
Budd-Chiari syndrome is suspected in patients with hepatomegaly (enlarged liver), ascites, liver failure, or cirrhosis when there is no obvious cause (e.g., alcohol abuse, hepatitis) or when the cause is unexplained. But a high index of suspicion is needed to make the diagnosis.
Physical examination may reveal the following findings:
- Jaundice
- Ascites
- Hepatomegaly
- Splenomegaly
- Ankle edema
- Stasis ulcerations
- Prominence of collateral veins
Your doctor will order these tests:
- CT scan or MRI of the abdomen
- Doppler ultrasound of the liver veins
- Liver biopsy
- Liver function tests
- Ultrasound of the liver
Liver function tests are usually abnormal; the pattern is variable and nonspecific. The presence of risk factors for thrombosis increase the consideration of this diagnosis.
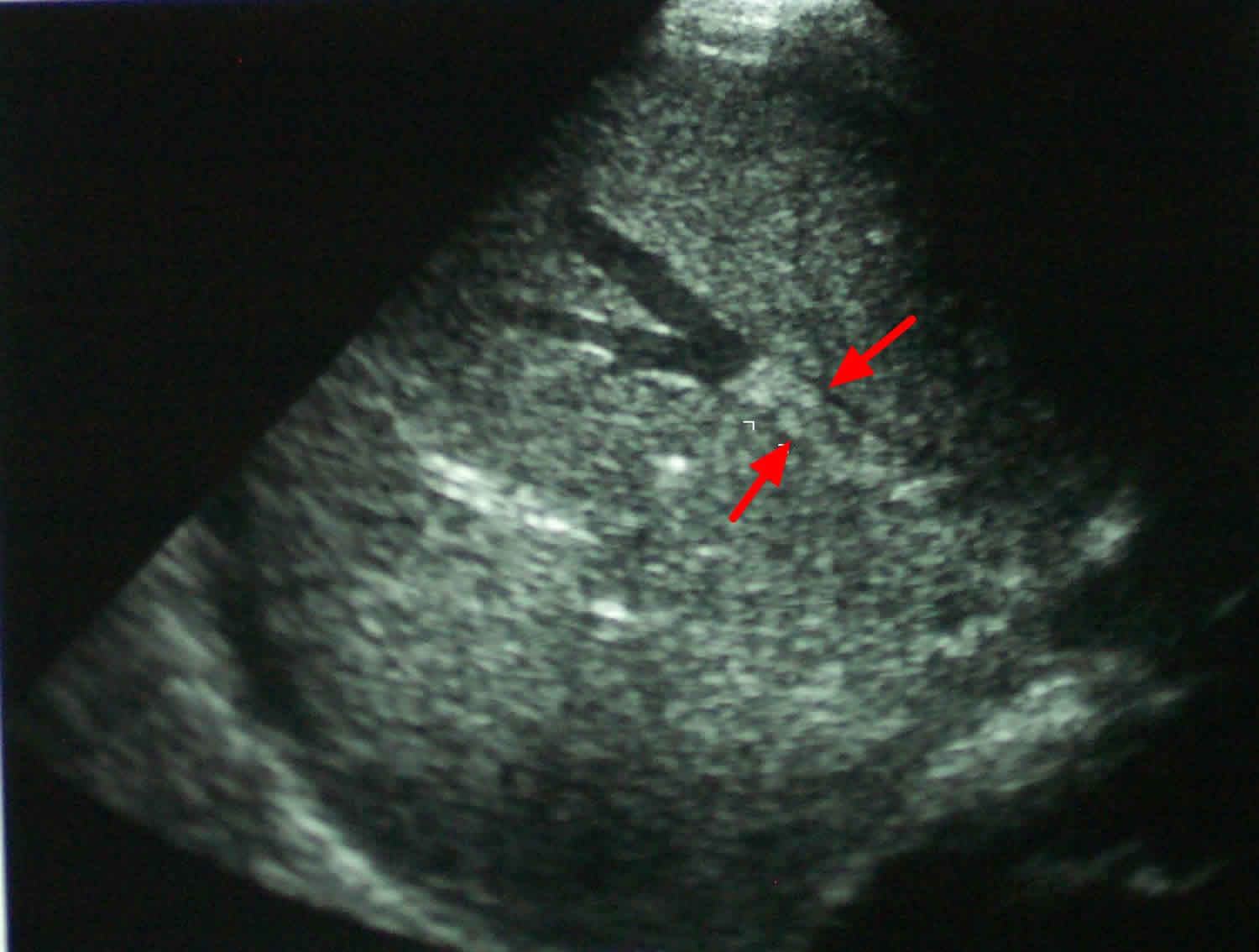
Imaging usually begins with abdominal Doppler ultrasonography, which can show the direction of blood flow and the site of obstruction. Magnetic resonance angiography and CT are useful if ultrasonography is not diagnostic. Conventional angiography (venography with pressure measurements and arteriography) is necessary if therapeutic or surgical intervention is planned.
Liver biopsy is done occasionally to diagnose the acute stages and determine whether cirrhosis has developed.
2016 European Association for the Study of the Liver (EASL) clinical practice guidelines on vascular diseases of the liver includes the following recommendations for the diagnosis of Budd-Chiari syndrome 4:
- The diagnosis of Budd-Chiari syndrome should be considered in all patients with symptomatic or asyptomatic acute or chronic liver disease.
- The first-line diagnostic study is Doppler ultrasonography; magnetic resonance imaging (MRI) and computed tomography (CT) scanning are for diagnostic confirmation. In the setting of negative imaging studies but high clinical suspicion of Budd-Chiari syndrome, reevaluate the patient with the help of an expert radiologist.
- Refer all patients with Budd-Chiari syndrome to institutions with expertise in this condition.
- Patients with Budd-Chiari syndrome should be screened for hepatocellular carcinoma (liver cancer). Referral to expert centers may be necessary to determine whether liver nodules are benign or malignant.
Figure 2. Budd Chiari syndrome ultrasound showing hepatic vein thrombus
Budd Chiari syndrome treatment
Budd Chiari syndrome treatment varies, depending on the cause of the blockage and whether the onset is acute or chronic and its severity such as fulminant liver failure versus decompensated cirrhosis or stable/asymptomatic. For example, patients with complications of portal hypertension should begin treatment on the basis of therapeutic recommendations for individuals with cirrhosis 5.
The cornerstones of Budd Chiari syndrome management are:
- Giving supportive therapy directed at complications (eg, ascites, liver failure, esophageal varices)
- Decompressing the congested liver (ie, maintaining venous outflow)
- Preventing propagation of the clot
Your doctor may recommend the following medicines:
- Blood thinners (anticoagulants)
- Clot-busting drugs (thrombolytic treatment)
- Medicines to treat the liver disease, including ascites
Although medical therapy can be instituted for short-term, symptomatic benefit 6, the use of such treatment alone is associated with a high 2-year mortality rate (80-85%). All individuals with Budd-Chiari syndrome should have close monitoring for early identification of liver deterioration 5.
Surgery may be recommended. This may involve:
- Angioplasty and stent placement
- Transjugular intrahepatic portosystemic shunt (TIPS)
- Venous shunt surgery
- Liver transplant
Aggressive interventions (e.g., thrombolysis, stents) are used when the disease is acute (e.g., within 4 weeks and in the absence of cirrhosis). Thrombolysis can dissolve acute clots, allowing recanalization and so relieving hepatic congestion. Radiologic procedures, such as angioplasty, stenting, and/or portosytemic shunts, can have a major role.
For caval webs or hepatic venous stenosis, decompression via percutaneous transluminal balloon angioplasty with intraluminal stents can maintain hepatic outflow. When dilation of a hepatic outflow narrowing is not technically feasible, transjugular intrahepatic portosystemic shunting (TIPS) and various surgical shunts can provide decompression by diverting blood flow into the systemic circulation. Portosystemic shunts are typically not used if hepatic encephalopathy is present; such shunts worsen liver function. Further, clots tend to form in shunts, especially if patients have a hematologic or thrombotic disorder.
Long-term anticoagulation is often necessary to prevent recurrence. Liver transplantation may be lifesaving in patients with fulminant disease or decompensated cirrhosis.
Anticoagulant therapy
Anticoagulation is needed in some patients, especially those with underlying hematologic disorders as the cause of Budd-Chiari syndrome.
Prothrombin time and activated partial thromboplastin time should be monitored once anticoagulation is started and should be maintained within the therapeutic range.
The European Association for the Study of the Liver (EASL) indicates that in the setting of adequately treated complications of portal hypertension, anticoagulant therapy is not contraindicated 5. A brief interruption in anticoagulation therapy may be considered in patients undergoing an invasive procedure, including paracentesis.
Thrombolytic therapy
This therapy has been used in a few cases. Agents include streptokinase, urokinase, recombinant tissue-type plasminogen activator (rt-PA), and other modalities.
Radiologic intervention
Systemic thrombolysis can be a high-risk endeavor; local thrombolysis performed by an interventional radiologist is preferable.
Other available radiologic interventions include balloon angioplasty, as well as placement of a stent or a transjugular intrahepatic portacaval shunt (TIPS) 7.
In a single-center retrospective study (1996-2012), Tripathi et al 8 reported good long-term outcomes in 67 patients with Budd-Chiari syndrome following successful transjugular intrahepatic portosystemic stent-shunt (TIPSS) using either polytertrafluoroethane (PTFE)-covered (n=40) or bare (n=27) stents. At a mean follow-up of 82 months, 15% of patients experienced post-transjugular intrahepatic portosystemic stent-shunt (TIPSS) encephalopathy; 2 patients underwent transplantation, 2 patients developed hepatocellular cancer, and 6 patients had liver-related deaths. The polytertrafluoroethane (PTFE)-covered stents had significantly better primary patency (76%) and shunt reinterventions (22%) compared to the bare stents (27% and 100%, respectively). Survival at 6 and 12 months was at 92% or above; that at 24 and 60 months was 80% or above; and 120-month survival was 72%. The investigators indicated that in symptomatic patients in whom hepatic vein patency cannot be restored, transjugular intrahepatic portacaval stent-shunt (TIPSS) should be considered as the first-line therapy 8.
In another single center retrospective study (2008-2014) of 190 patients with Budd-Chiari syndrome who underwent endovascular procedures (hepatic vein, collateral vein or IVC plasty with or without stenting, or transjugular intrahepatic portacaval stent-shunt [TIPSS]), venous recanalization and transjugular intrahepatic portosystemic stent-shunt (TIPSS) were safe and effective: 153 patients (80.5%) experienced treatment response, with 19 patients (10.0%) requiring repeat interventions and 9 patients (4.7%) with complications 9. Of the 190 patients, 147 had hepatic vein obstruction, 40 had inferior vena cava obstruction, and 3 had both. Thirty-eight patients underwent hepatic vein/stenting; 3, collateral vein stenting; 40, inferior vena cava plasty/stenting; 3, hepatic vein and inferior vena cava stenting; and 106, transjugular intrahepatic portacaval stent-shunt [TIPSS] 9.
Tripathi et al 10 reported similar findings for venous recanalization and transjugular intrahepatic portacaval stent-shunt [TIPSS] in 122 patients.
More recently, retrospective data (2011-2016) from another study with 68 patients revealed excellent efficacy and long-term outcomes of endovascular therapy (hepatic vein recanalization and accessory hepatic vein recanalization) for hepatic vein-type Budd-Chiari syndrome 11. Investigators found a 100% technical success rate, with a 95.6% clinical success rate. The mean follow-up period was 39.4 ± 13.6 months. At 1 year, the primary and secondary patency rates were 80.0% and 93.8%; at 2 years, 72.8% and 90.3%; and at 5 years, 67.9% and 91.2%, respectively. Survival was 96.9% at 1 year, 93.4% at 2 years, and 91.2% at 5 years 11.
Angioplasty
The European Association for the Study of the Liver (EASL) indicates angioplasty/stenting should be considered the first-line decompressive procedure in patients with short hepatic vein or inferior vena cava stenosis 5. For those in whom initial therapy or angioplasty/stenting is ineffective, treat with portal derivative techniques, of which transjugular intrahepatic portosystemic shunt (TIPS) using polytertrafluoroethane (PTFE)-covered stents is deemed the derivative therapy of choice by the EASL. Consider surgical shunting in the setting of TIPS unfeasibility or failure 5.
Angioplasty procedure can help relieve obstruction caused by membranous webs. In a study of 101 patients with Budd-Chiari syndrome, Li et al 12 concluded that the condition can be safely and effectively treated with percutaneous transhepatic balloon angioplasty (PTBA). The authors reported successful percutaneous transhepatic balloon angioplasty (PTBA) (performed after hepatovenography, with or without stenting) in 92 of the study’s patients, with all of the successful procedures resulting in significant symptom improvement.
Complications included acute hepatic vein thrombosis, occurring during or after the operation (n=3); sustained intraperitoneal bleeding from the transhepatic puncture track (n=2); pulmonary embolism, which occurred during the procedure (n=1); and intrahepatic hematoma (n=1) 12. All were managed nonsurgically. Primary patency rates at 6-, 12-, and 24-month follow-up were 84%, 78%, and 76%, respectively (with several patients lost to follow-up); secondary patency rates were 95%, 92%, and 84%, respectively. Despite these satisfactory midterm patient outcomes, the authors cautioned that long-term outcomes in patients treated with percutaneous transhepatic balloon angioplasty (PTBA) for Budd-Chiari syndrome require investigation 12.
Variceal treatment
Gastroscopy should be performed to help rule out the presence of esophageal and gastric varices. If present, they may be obliterated with banding or sclerotherapy. Nonselective beta blockers (eg, propranolol, nadolol) can be administered for primary prophylaxis against variceal bleeding.
Diuretic therapy
Patients with liver failure and ascites have total body sodium overload, despite typically low serum sodium concentrations. Inducing negative sodium balance can reduce the amount of ascites. Take special care when using diuretics, to avoid inducing hepatorenal syndrome or creating electrolyte and fluid disturbances through overly aggressive diuresis. Electrolyte levels should be monitored closely.
Secondary hyperaldosteronism is part of this clinical picture, making spironolactone typically the first-line diuretic. Chlorothiazide or furosemide is often added, which can provide synergy and avoid hyperkalemia.
Diet
A low-sodium diet is recommended for the control of ascites.
Paracentesis
Symptomatic treatment for Budd-Chiari syndrome includes diuretics and therapeutic paracentesis, when necessary, although paracentesis can be associated with catastrophic complications, such as bacterial peritonitis. Consequently, the benefits of therapeutic paracentesis must be carefully weighed against its risks.
Portal decompression
Decompression of the hepatic vasculature should be offered if portal hypertension is the cause of the symptoms. Either surgery or a transjugular intrahepatic portosystemic shunt (TIPS) procedure can be performed 13.
Liver transplantation
Liver transplantation should be offered if decompensated liver cirrhosis is present 14 or as salvage treatment in the setting of failed portal derivative techniques 5. Posttransplantation anticoagulation is required in most patients with Budd-Chiari syndrome 5.
In a Polish retrospective study (2000-2009) 15, the long-term clinical outcomes (eg, patient and graft survival) following liver transplantation and anticoagulation maintenance for Budd-Chiari syndrome were good in 25 patients with myeloproliferative disease and recurrent thrombosis.
Similar findings were reported in an Indian retrospective study (2011-2015) 16 of 9 patients with Budd-Chiari syndrome and chronic liver disease who underwent living donor liver transplantation. The investigators noted that prevention of recurrent thrombosis was dependent on “meticulous surgical technique, perfect and wide outflow anastomoses, and a strict anticoagulation protocol. Moreover, the use of synthetic (PTFE) graft for inferior vena cava interposition was safe, feasible, and provided good reconstruction results 16.
Follow-up and monitoring
Patients with lesions that are amenable to balloon dilatation or stents require follow-up catheterizations and, frequently, repeat dilatations or stent replacement. In addition, patients should have routine surveillance for hepatocellular carcinoma (HCC) 17.
Budd Chiari syndrome life expectancy
Without treatment, most patients with complete venous obstruction die of liver failure within 3 to 5 years. For patients with incomplete obstruction, the course varies.
The following factors, however, have been associated with a good prognosis 1:
- Younger age at diagnosis
- Low Child-Pugh score. The following formula has been proposed to calculate the prognostic index for Budd-Chiari syndrome; a score of less than 5.4 is associated with a good prognosis:
- Prognostic index = (ascites score x 0.75) + (Pugh score x 0.28) + (age x 0.037) + (creatinine level x 0.0036)
- Absence of ascites or easily controlled ascites
- Low serum creatinine level
In a systematic review of 79 studies, investigators found that although univariate analysis indicated bilirubin and creatinine levels as well as ascites might be significant prognostic factors, multivariate analyses did not always reveal achievement of statistical significance 18.
The 5-year survival rate for patients with the syndrome is 38-87% following portosystemic shunting. The actuarial 5-year survival rate following liver transplantation is 70% 19. Long-term follow-up in adults has demonstrated 10-year survival rates as high as 55%.
The prognosis is poor, however, in patients with Budd-Chiari syndrome who remain untreated, with death resulting from progressive liver failure in 3 months to 3 years from the time of diagnosis 20.
In a University of Pennsylvania retrospective study (2008-2013) 21 comprising 47 patients with Budd-Chiari syndrome, there were no significant differences in the treatment outcomes among those receiving anticoagulation therapy alone, transjugular intrahepatic portosystemic shunt (TIPS) placement alone, and TIPS in conjunction with anticoagulation. The investigators noted that the significant prognostic predictors for liver transplantation were age, presence of cirrhosis, and presence of chronic kidney disease.
Morbidity and mortality
Morbidity and mortality in Budd-Chiari syndrome are generally related to complications of liver failure and ascites but can also be impacted by the type of concomitant underlying disease, if any. Complications associated with Budd-Chiari syndrome include the following:
- Hepatic encephalopathy
- Variceal hemorrhage
- Hepatorenal syndrome
- Portal hypertension
- Complications secondary to hypercoagulable state
- Complications secondary to hepatic decompensation
Bacterial peritonitis is always of concern in the patient with ascites, especially if paracentesis is undertaken. Complications must also be considered in relation to therapies used (eg, thrombolytics). The mortality rate can be high in patients who develop fulminant hepatic failure.
Budd-Chiari syndrome can also lead to liver cancer (hepatocellular carcinoma) or oppositely, in some cases, develop secondary to it. In a retrospective study, Liu et al 22 found evidence that hepatocellular carcinoma in primary Budd-Chiari syndrome is associated with blockage of the inferior vena cava and stricture of the hepatic venous outflow tract. The investigators’ results also indicated that transcatheter arterial chemoembolization (TACE) is an effective treatment for liver cancer (hepatocellular carcinoma) in these patients, with a significant drop in alpha-fetoprotein (AFP) levels after TACE treatment. The study included 246 patients with Budd-Chiari syndrome, including 14 with hepatocellular carcinoma. Ultrasonography, computed tomography (CT) scanning, magnetic resonance imaging (MRI), and angiography were used to determine the imaging characteristics in patients with hepatocellular carcinoma 22.
References- Budd-Chiari Syndrome. https://emedicine.medscape.com/article/184430-overview
- Budd Chiari Syndrome. https://rarediseases.org/rare-diseases/budd-chiari-syndrome/
- Goel RM, Johnston EL, Patel KV, Wong T. Budd-Chiari syndrome: investigation, treatment and outcomes. Postgrad Med J. 2015 Dec. 91(1082):692-7
- European Association for the Study of the Liver. EASL clinical practice guidelines: vascular diseases of the liver. J Hepatol. 2016 Jan. 64 (1):179-202
- European Association for the Study of the Liver. EASL clinical practice guidelines: vascular diseases of the liver. J Hepatol. 2016 Jan. 64 (1):179-202.
- Horton JD, San Miguel FL, Ortiz JA. Budd-Chiari syndrome: illustrated review of current management. Liver Int. 2008 Apr. 28(4):455-66.
- Copelan A, Remer EM, Sands M, Nghiem H, Kapoor B. Diagnosis and management of Budd Chiari syndrome: an update. Cardiovasc Intervent Radiol. 2015 Feb. 38(1):1-12
- Tripathi D, Macnicholas R, Kothari C, et al. Good clinical outcomes following transjugular intrahepatic portosystemic stent-shunts in Budd-Chiari syndrome. Aliment Pharmacol Ther. 2014 Apr. 39(8):864-72
- Rathod K, Deshmukh H, Shukla A, et al. Endovascular treatment of Budd-Chiari syndrome: single center experience. J Gastroenterol Hepatol. 2017 Jan. 32(1):237-43.
- Tripathi D, Sunderraj L, Vemala V, et al. Long-term outcomes following percutaneous hepatic vein recanalization for Budd-Chiari syndrome. Liver Int. 2017 Jan. 37(1):111-20.
- Chen ZK, Fan J, Cao C, Li Y. Endovascular treatment for hepatic vein-type Budd-Chiari syndrome: effectiveness and long-term outcome. Radiol Med. 2018 Oct. 123 (10):799-807.
- Li T, Zhai S, Pang Z, et al. Feasibility and midterm outcomes of percutaneous transhepatic balloon angioplasty for symptomatic Budd-Chiari syndrome secondary to hepatic venous obstruction. J Vasc Surg. 2009 Nov. 50(5):1079-84.
- Wu X, Ding W, Cao J, Han J, Li J. Modified transjugular intrahepatic portosystemic shunt in the treatment of Budd-Chiari syndrome. Int J Clin Pract. 2010 Mar. 64(4):460-4.
- Ulrich F, Pratschke J, Neumann U, et al. Eighteen years of liver transplantation experience in patients with advanced Budd-Chiari syndrome. Liver Transpl. 2008 Feb. 14(2):144-50.
- Oldakowska-Jedynak U, Ziarkiewicz M, Ziarkiewicz-Wroblewska B, et al. Myeloproliferative neoplasms and recurrent thrombotic events in patients undergoing liver transplantation for Budd-Chiari syndrome: a single-center experience. Ann Transplant. 2014 Nov 14. 19:591-7.
- Pahari H, Chaudhary RJ, Thiagarajan S, et al. Hepatic venous and inferior vena cava morphology no longer a barrier to living donor liver transplantation for Budd-Chiari syndrome: surgical techniques and outcomes. Transplant Proc. 2016 Oct. 48(8):2732-7.
- Ren W, Qi X, Yang Z, Han G, Fan D. Prevalence and risk factors of hepatocellular carcinoma in Budd-Chiari syndrome: a systematic review. Eur J Gastroenterol Hepatol. 2013 Jul. 25(7):830-41
- Qi X, Ren W, Wang Y, Guo X, Fan D. Survival and prognostic indicators of Budd-Chiari syndrome: a systematic review of 79 studies. Expert Rev Gastroenterol Hepatol. 2015 Jun. 9 (6):865-75
- Montano-Loza AJ, Tandon P, Kneteman N, Bailey R, Bain VG. Rotterdam score predicts early mortality in Budd-Chiari syndrome, and surgical shunting prolongs transplant-free survival. Aliment Pharmacol Ther. 2009 Nov 15. 30(10):1060-9.
- Khuroo MS, Al-Suhabani H, Al-Sebayel M, et al. Budd-Chiari syndrome: long-term effect on outcome with transjugular intrahepatic portosystemic shunt. J Gastroenterol Hepatol. 2005 Oct. 20(10):1494-502
- Pavri TM, Herbst A, Reddy R, Forde KA. Budd-Chiari syndrome: A single-center experience. World J Gastroenterol. 2014 Nov 21. 20(43):16236-44
- Liu FY, Wang MQ, Duan F, Fan QS, Song P, Wang Y. Hepatocellular carcinoma associated with Budd-Chiari syndrome: imaging features and transcatheter arterial chemoembolization. BMC Gastroenterol. 2013 Jun 24. 13:105