What is butylated hydroxytoluene
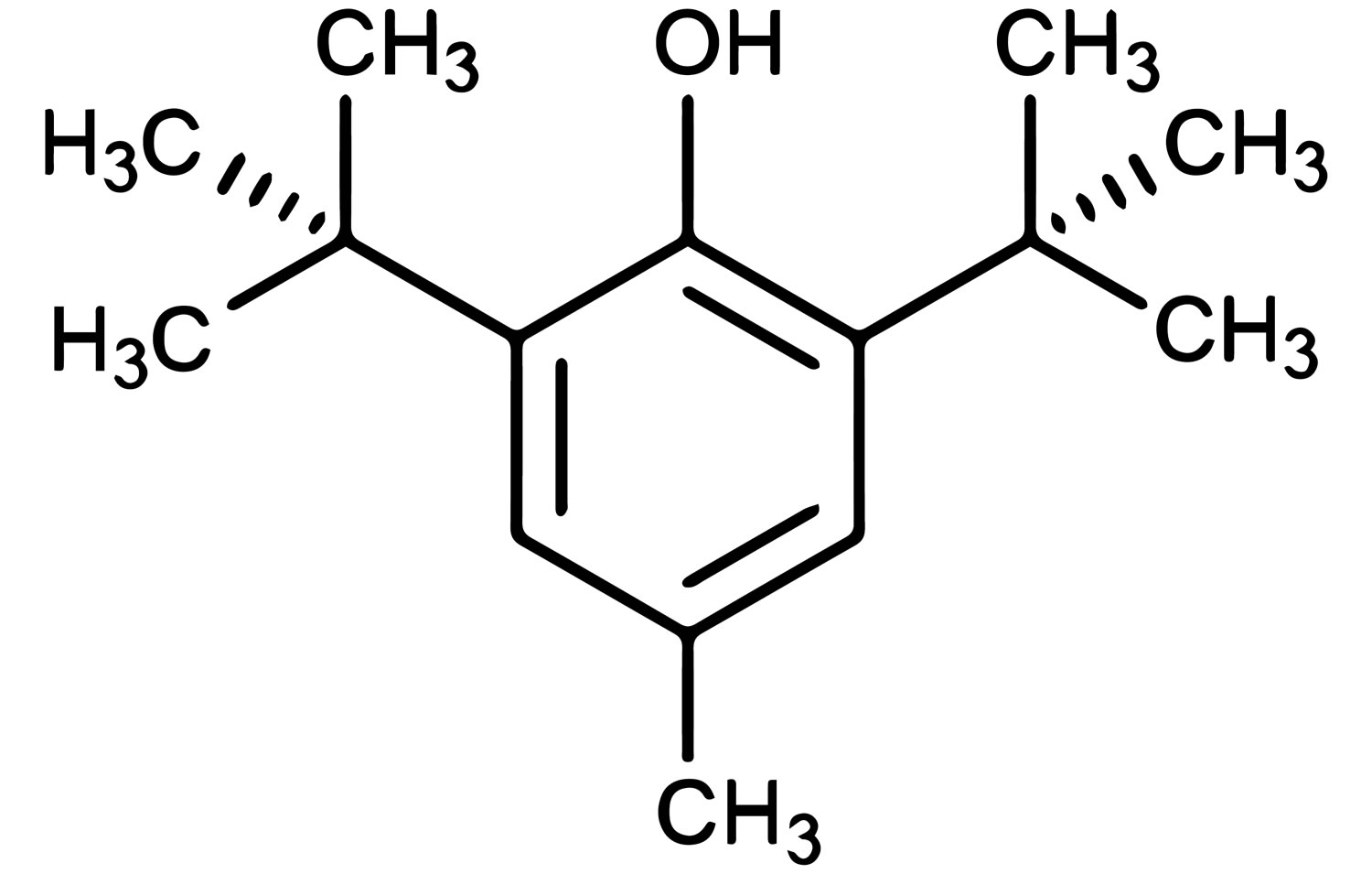
Butylated hydroxytoluene (2,6-Di-tert-butyl-4-methylphenol) also known as BHT or butylhydroxytoluene, is a synthetic α-tocopherol analogue antioxidant and preservative in the foods, cosmetics and pharmaceutical industry for scavenging hydrogen peroxide 1. Butylated hydroxytoluene (BHT) is a white, crystalline solid which is odorless or has a faint aromatic odor. Butylated hydroxytoluene is insoluble in water, and freely soluble in ethanol and fatty oils. It has a melting point of 70 °C and an octanol/water partition coefficient of 5.1 2. According to the Hazardous Substances Data Bank HSDB 3, butylated hydroxytoluene (BHT) is prepared in a multistep process by the reaction of p-cresol (4-
methylphenol) with high purity isobutylene (2-methylpropene) using an acid catalyst. Upon neutralization by addition of sodium carbonate, crystallization with isopropanol, filtration and washing with isopropanol, the substance is dried and sieved to obtain the final product.
During storage, food quality is reduced quickly because of microbial spoilage and chemical reactions 4. The most important quality deterioration in the food systems is caused by chemical processes called lipid oxidation. In this process unsaturated fatty acids reacting with molecular oxygen via a free radical chain mechanism 5. As a consequence, hydroperoxides are formed which are unstable and decompose relatively quickly into aldehydes, ketones, alcohols, acids, esthers or hydrocarbons. These secondary oxidative products are responsible for food quality, nutrition, safety, color and consumers’ acceptance 6. The variables that affect the susceptibility of lipids to oxidation are temperature, fatty acid composition, antioxidants, metals, enzyme-catalyzed reactions, and water 7. In order to slow or control food’s lipid oxidation, manufacturers use antioxiative substances. The most commonly used are butyl hydroxyanisole (BHA), butylated hydroxytoluene (BHT), tertiary-butyl hydroquinone (TBHQ), and propylgalate by directly adding them to food products 7.
Nowadays, active packaging which antioxidants are incorporated into packaging materials is an alternative way for food spoilage preservation 4. It can avoid contacting between the substances and the food as well as decrease the risk of adverse reactions 8. Active packaging extends the shelf life of food by preventing interacting of humidity or substances such as oxygen, ethylene, aroma or unusual flavors with the food 9. For example, incorporation of synthetic antioxidants, such as butylated hydroxytoluene (BHT) and butyl hydroxyanisole (BHA) in polyethylene film, showed to protect the fresh beef color from oxidation 10. However, in recent years, there has been a growing interest in the use of natural antioxidants in food packaging applications such as green tea extract with fish gelatin films 11, brown algae extract with tuna-skin gelatin 12, essential oil 13, tocopherols 14, catechin-lysozyme 15. Longan seeds and leaves have been employed as a good source of phenolic compounds, showing good antioxidant activity 16.
Figure 1. Butylated hydroxytoluene (BHT)
Butylated hydroxytoluene in food
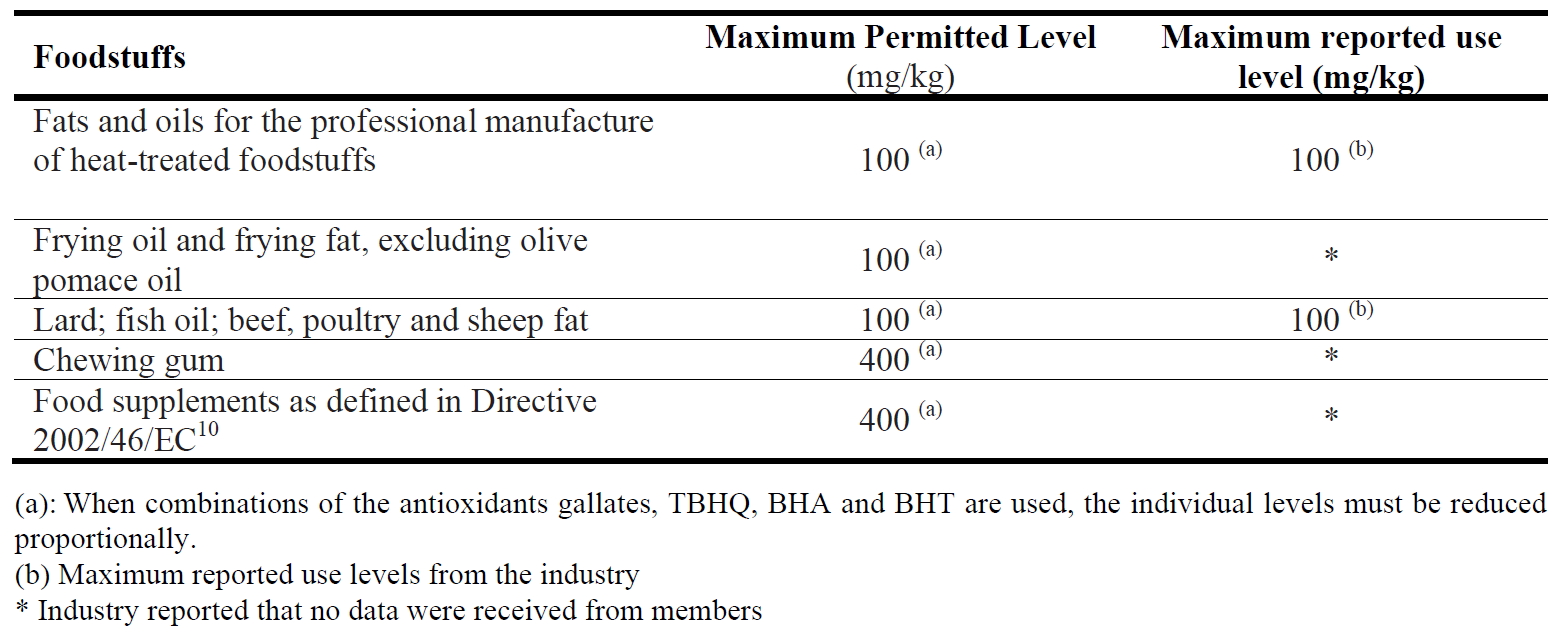
Butylated hydroxytoluene (BHT) is a synthetic antioxidant authorized for use in fats and oils, only for the professional manufacture of heat-treated food, in frying oil and frying fat (excluding olive pomace oil) and in lard, fish oil, beef, poultry and sheep fat 17. Butylated hydroxytoluene (BHT) is permitted alone or in combination with other antioxidants such as gallates, tert-butylhydroquinone (TBHQ) and butylated hydroxyanisole (BHA) in amounts up
to 100 mg/kg expressed as fat. In addition, butylated hydroxytoluene (BHT) is permitted in chewing gum alone or in combination with the aforementioned antioxidants at a maximum level of 400 mg/kg chewing gum 17.
No data on the actual levels of butylated hydroxytoluene (BHT) in foods have been found during literature searches in the databases ToxNet, PubMed and CAPlus, or on the web pages of the Food Standards Agency of Great Britain. The Danish Veterinary and Food Administration has reported a project on monitoring and control of food additives in which butylated hydroxytoluene (BHT) levels were analyzed in 122 samples of emulsified and non-emulsified sauces (dressings, ketchup etc.) and fruit- and vegetable preparations (chutney, tomato paste etc.). Butylated hydroxytoluene (BHT) was not identified in any of these samples 18.
When scrutinizing ingredient lists on various chewing gums on the Danish market, butylated hydroxytoluene (BHT) is not mentioned in the list of ingredients on every brand. Hence, it can be deduced that chewing gum may be manufactured without the use of butylated hydroxytoluene (BHT). In a survey by Bemrah et al. 19 1 out of 28 chewing gum brands was found to contain 200 mg butylated hydroxytoluene/kg, whereas the remainder contained no butylated hydroxytoluene (BHT).
Additional information on reported use levels of use for butylated hydroxytoluene was made available to the Panel by FoodDrinkEurope. For the food categories “fats and oils for the professional manufacture of heat-treated foodstuffs” and for “lard, fish oil, beef, poultry and sheep fat” the data made available are listed in Table 1. For other food categories where the use of butylated hydroxytoluene is authorized, FoodDrinkEurope either reported that no data were received from their membership or that these categories are not representatively covered by FoodDrinkEurope’s membership. Butylated hydroxytoluene (BHT) is also used in food contact materials with a specific migration limit of 3 mg/kg food (Commission Regulation 10/201111).
Table 1. Maximum Permitted Levels of butylated hydroxytoluene (BHT) in foodstuffs according to Directive No 95/2/EC and maximum reported use levels of BHT in foodstuffs used for the refined exposure assessment
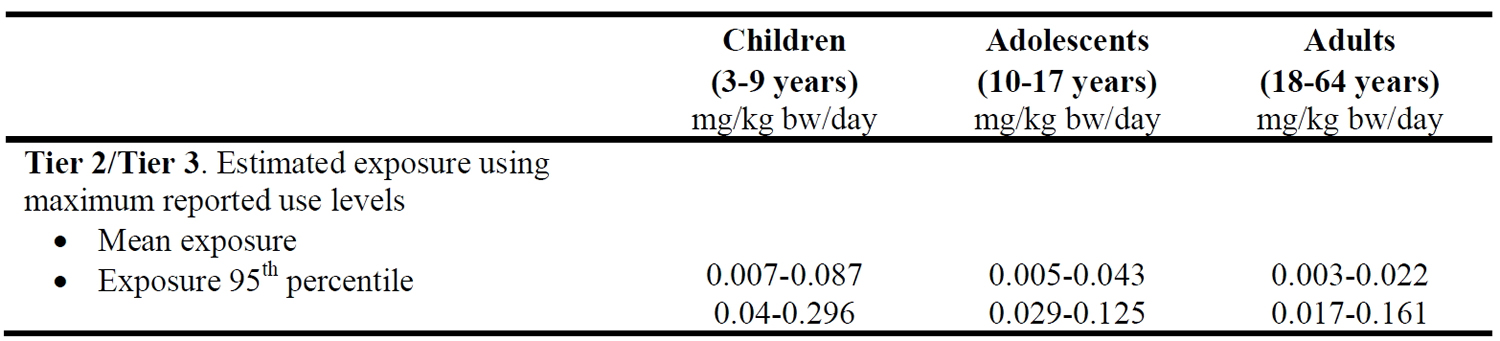
[Source 17]Table 2. Summary of anticipated exposure to butylated hydroxytoluene (BHT) from its use as food additive using maximum reported use levels for four population groups
[Source 17]Table 2 summarizes the estimated exposure to butylated hydroxytoluene (BHT) from its use as food additive of all four population groups. An exposure to butylated hydroxytoluene from the consumption of chewing gum using the Maximum Permitted Level for this food category of 400 mg/kg would require a daily intake of 7.5 g of chewing gum to reach a level of 0.05 mg butylated hydroxytoluene (BHT)/kg body weight/day. Unfortunately, very limited data were available for the consumption of this food category in the Comprehensive Database since only few consumers in some countries reported data on chewing gum consumption. It also has to be considered that not all butylated hydroxytoluene (BHT) present in chewing gum will be ingested. It was concluded that 80-99% of butylated hydroxytoluene (BHT) in chewing gum is not ingested 20, but ingestion may be higher given that higher levels of extraction of up to 50% were also found in one study 21. Assuming a 10% extraction and subsequent ingestion of butylated hydroxytoluene (BHT) from chewing gum and selecting only those surveys with a sufficiently high number of consumers, the average exposure to butylated hydroxytoluene (BHT) from chewing gum consumption is in the range of 0-0.003 mg/kg body weight/day for children, 0-0.002 mg/kg body weight/day for adolescents and 0-0.004 mg/kg body weight/day for adults (consumers only). At the 95th percentile, exposure to butylated hydroxytoluene (BHT) from chewing gum consumption is in the range of 0.004-0.008 mg/kg body weight/day for children, 0.003-0.006 mg/kg bw/day for adolescents and 0.004-0.008 mg/kg body weight/day for adults.
Using a worst case scenario of combined exposure to butylated hydroxytoluene (BHT) from food categories where its use as food additive is authorized, the European Food Safety Authority (EFSA) Panel estimated potential exposure to be in the range of 0.01-0.03 mg/kg body weight/day on average and 0.03-0.17 mg/kg body weight/day at the 95th percentile for adults. For adolescents, combined exposure to butylated hydroxytoluene (BHT) from all food categories would be on average in the range of 0.01-0.05 mg/kg body weight/day and in the range of 0.04-0.13 mg/kg body weight/day at the 95th percentile. For children, combined exposure to butylated hydroxytoluene (BHT) from all food categories would be on average in the range of 0.01-0.09 mg/kg body weight/day and in the range of 0.05-0.30 mg/kg body weight/day at the 95th percentile. Exposure to butylated hydroxytoluene (BHT) from its use in food contact materials with a specific migration limit of 3 mg/kg food and the assumption that every day throughout lifetime a person weighing 60 kg consumes 1 kg of food packed in plastics containing butylated hydroxytoluene (BHT) in the maximum permitted quantity would be increased by 0.05 mg/kg body weight/day. Assuming that children weighing 15 kg also consume 1 kg of food packed in plastics containing butylated hydroxytoluene (BHT) in the maximum permitted quantity, exposure to butylated hydroxytoluene (BHT) of children would be increased by 0.2 mg/kg body weight/day.
The European Union Scientific Committee for Food (SCF) established an Acceptable Daily Intake (ADI) for butylated hydroxytoluene (BHT) of 0-0.05 mg/kg body weight/day based on thyroid, reproduction and hematological effects in the rat. Acceptable Daily Intake (ADI) is an estimate of the amount of a food additive that can be ingested daily over a lifetime without appreciable health risk. The Joint FAO/WHO Expert Committee on Food Additives (JECFA) allocated an ADI of 0-0.3 mg/kg body weight/day for butylated hydroxytoluene (BHT) based on effects in the reproduction segments and hepatic enzyme induction seen in two separate 2-generation studies in rats. The European Food Safety Authority (EFSA) Panel concluded that butylated hydroxytoluene (BHT) is not of concern with respect to genotoxicity and that any carcinogenicity would be thresholded. After the last European Union Scientific Committee for Food (SCF) evaluation, two new 2-generation studies have been reported which were the basis for the ADI set by Joint FAO/WHO Expert Committee on Food Additives (JECFA). Both studies revealed a No-observed-adverse-effect level (NOAEL) of 25 mg/kg body weight/day. No-observed-adverse-effect level (NOAEL) is the greatest concentration or amount of a substance, found by experiment or observation, that causes no detectable adverse alteration of morphology, functional capacity, growth, development, or lifespan of the target organism under defined conditions of exposure. Overall, the European Food Safety Authority (EFSA) Panel concluded that the present database gives reason to revise the ADI of 0.05 mg/kg body weight/day. Based on the NOAEL of 25 mg/kg body weight/day and an uncertainty factor of 100, the European Food Safety Authority (EFSA) Panel derived an ADI of 0.25 mg/kg body weight/day. Since the NOAEL of 25 mg/kg body weight/day is below the BMDL10 value of 247 mg/kg body weight/day derived from the data for the incidence of hepatocellular carcinomas (liver cancers) in male rats, the European Food Safety Authority (EFSA) Panel concluded that this NOAEL also covers the hepatocellular carcinomas observed in the long-term studies with butylated hydroxytoluene (BHT). Exposure of adults to butylated hydroxytoluene (BHT) is unlikely to exceed the newly derived Acceptable Daily Intake (ADI) at the mean and at the 95th percentile. Benchmark dose (BMD) is defined as the dose that corresponds to a specific change in an adverse response compared to the response in unexposed subjects and the lower 95% confidence limit is termed the benchmark dose level (BMDL). For exposure of children to butylated hydroxytoluene (BHT) from its use as food additive, the European Food Safety Authority (EFSA) Panel noted that it is also unlikely that this ADI is exceeded at the mean, but is exceeded for some European countries (Finland, The Netherlands) at the 95th percentile.
Butylated hydroxytoluene biological and toxicological data
In one study in humans the urinary metabolites butylated hydroxytoluene (BHT)-COOH (the “acid”) and benzoyl-glycine (hippuric acid) were found after two separate oral doses of 100 mg butylated hydroxytoluene (BHT), and in another study butylated hydroxytoluene (BHT)-COOH and its glucuronide ester were the only major metabolites identified in urine after an oral dose of 1.0 g 22. In two men, excretion of a single oral dose of 40 mg/kg [14C] butylated hydroxytoluene (BHT) was 50% in the first 24 hours, followed by slower excretion for the next 10 days. In total, 63-67% of the dose was excreted in the urine after 10 days. Fecal excretion was measured in one man and was found to be 0.3% of the dose per day initially, and waning off to 0.02% of the dose per day, 31 days after dosing 23.These studies show that butylated hydroxytoluene (BHT) is rapidly absorbed from the gastrointestinal tract and distributed. Upon absorption, butylated hydroxytoluene (BHT) is generally distributed to the liver and body fat. Excretion is mainly via urine and feces. The metabolism of butylated hydroxytoluene (BHT) is complex. There may be important species differences. It is not known for example whether humans are capable of forming the quinone methides, metabolites that were found in rats and mice. In addition, biliary excretion seems not to be as significant in man as it is in rats, rabbits and dogs.
Acute oral toxicity
According to the results of acute toxicity studies with butylated hydroxytoluene (BHT) reported to JECFA 22, butylated hydroxytoluene (BHT) possesses low acute toxicity. The acute oral LD50 of butylated hydroxytoluene (BHT) was 1700-1970 mg BHT/kg body weight in rats, 2100-3200 mg butylated hydroxytoluene (BHT)/kg body weight in rabbits, 10,700 mg BHT/kg body weight in guinea pigs, 940-2100 mg BHT/kg body weight in cats, and 2000 mg BHT/kg bw in mice 24.
Short-term or subchronic exposure to butylated hydroxytoluene (BHT) affects the liver of mice, rats and chickens, also showing histopathological changes in this organ. In addition, BHT has been shown to increase the relative thyroid and adrenal weight in rats. Newer data show an α-tocopherol sparing effect in rats.None of the studies available could be used to derive a No-observed-adverse-effect level (NOAEL).
Genotoxicity
The genotoxicity of butylated hydroxytoluene (BHT) has been studied in several in vitro systems 22 and an extensive database of genotoxicity studies for butylated hydroxytoluene (BHT) was reviewed by Bomhard et al. 25 and by Williams et al. 26. These authors concluded that the majority of evidence indicated a lack of potential for butylated hydroxytoluene (BHT) to induce point mutations, chromosomal aberrations, or to interact with or damage DNA, and that butylated hydroxytoluene (BHT) does not represent a genotoxic risk.
TemaNord 2 reported that butylated hydroxytoluene (BHT) was not genotoxic in vivo in rats at oral doses up to 500 mg/kg body weight (dominant lethal assay) or in mice at doses of 1% in the feed (approximately 1000 mg/kg body weight/day) (dominant lethal and heritable translocation assays).
In general, the genotoxicity studies on butylated hydroxytoluene (BHT) show that the majority of evidence indicates a lack of potential for butylated hydroxytoluene (BHT) to induce point mutations, chromosomal aberrations, or to interact with or damage DNA. The European Food Safety Authority (EFSA) Panel recognized that positive genotoxicity results obtained in vitro with butylated hydroxytoluene (BHT) and butylated hydroxytoluene (BHT) metabolites may be due to pro-oxidative chemistry giving rise to formation of quinones and reactive oxygen species and that such a mechanism of genotoxicity is generally considered to have a threshold.
The International Agency for Research on Cancer 27 evaluated butylated hydroxytoluene (BHT) and classified it in group 3, since no evaluation could be made of the carcinogenicity of butylated hydroxytoluene (BHT) to humans, and there was
limited evidence for the carcinogenicity in experimental animals. Williams et al. 26 have reviewed data on genotoxicity and carcinogenicity of butylated hydroxytoluene (BHT) including reports which appeared subsequent to the International Agency for Research on Cancer 27 evaluation. Williams et al. 26 argued that butylated hydroxytoluene (BHT) is not genotoxic or carcinogenic; they particularly argued that the carcinogenicity in rats found in the study by Olsen et al. 28 has not been confirmed in other studies with rats, and that the effects found may be attributable to study conditions and not to the administration of butylated hydroxytoluene (BHT). They also point out that a more recent study 29 dosing Wistar rats for up to 18 months with 0.1% 2,2’-methylenebis (4-methyl-6-tertbutylphenol), an antioxidant which is essentially two molecules of butylated hydroxytoluene (BHT) and has all the attributes of butylated hydroxytoluene (BHT), did not result in a carcinogenic response.
The European Food Safety Authority (EFSA) Panel concluded that butylated hydroxytoluene (BHT) in high doses can exert tumor-promoting effects in some animal models. The data do not allow the establishment of a NOAEL for a carcinogen-promotional effect.
The association between dietary intake of butylated hydroxytoluene (BHT) and stomach cancer risk was investigated in the Netherlands Cohort Study (NLCS) that started in 1986 among 120 852 men and women aged 55-69 years. A semi-quantitative food frequency questionnaire was used to assess food consumption.
Information on butylated hydroxytoluene (BHT) content of cooking fats, oils, mayonnaise and other creamy salad dressings and dried soups was obtained by chemical analysis, a Dutch database of food additives (ALBA) and the Dutch Compendium of Foods and Diet Products. After 6.3 years of follow-up, complete data on butylated hydroxyanisole (BHA) and butylated hydroxytoluene (BHT) intake of 192 incident stomach cancer cases and 2035 sub-cohort members were available for case-cohort analysis. Mean intake of butylated hydroxytoluene (BHT) among sub-cohort members was 0.351 mg/day. No association with stomach cancer risk was observed for consumption of mayonnaise and other creamy salad dressings with butylated hydroxytoluene (BHT). A statistically non-significant decrease in stomach cancer risk was observed with increasing BHA and butylated hydroxytoluene (BHT) intake. No significant association with stomach cancer risk was found in this study, for normal dietary intake of low levels of butylated hydroxytoluene (BHT) 30.
In conclusion, lung or liver tumors have been seen in some studies in mice or rats exposed orally to butylated hydroxytoluene (BHT) as the single test substance. BMD analyses of the data reported by Brooks et al. 31 on the incidence of lung neoplasia in mice induced by butylated hydroxytoluene (BHT) revealed a BMDL10 of 38 mg/kg body weight/day, and of the data reported by Olsen et al. 28 on the incidence of hepatocellular carcinomas in male rats induced by butylated hydroxytoluene (BHT) a BMDL10 of 247 mg/kg body weight/day. The NOAEL for non-neoplastic effects in the study of Olsen et al. 28 was 25 mg/kg bw/day, based on the effects on litter size, sex ratio and pup body weight gain during the lactation period in the reproduction segment of the study.
Reproductive and developmental toxicity
Studies on reproductive toxicity have been reported in mice, rats, chickens and monkeys 22. In a study by Eriksson and Siman 32, pregnant diabetic and normal Sprague-Dawley rats were fed as required either a standard diet or a diet with 1% of butylated hydroxytoluene (BHT) (estimated to be equivalent to approximately 500 mg/kg body weight/day). The fetuses of the diabetic rats (2.70 g body weight) were smaller than the fetuses of the normal rats (3.68 g) when the mothers were fed a standard diet. The butylated hydroxytoluene (BHT) diet increased the fetal weight in the offspring of diabetic rats (3.17 g), with no change in fetuses of the normal rats (3.65 g). The placentas of diabetic rats were heavier than the placentas of normal rats; this difference was not present in the butylated hydroxytoluene (BHT)-fed rats. The butylated hydroxytoluene (BHT) treatment had no effect on the rate of resorptions, which was increased in the diabetic rats compared with the normal rats. In contrast, the increased rate of congenital malformations in the offspring of diabetic rats (19%), compared with that in the normal rats (0%), was markedly decreased by the butylated hydroxytoluene (BHT) diet (2.3%). No malformations were found in the normal rats treated with butylated hydroxytoluene (BHT). The European Food Safety Authority (EFSA) Panel considered that the only dose level of butylated hydroxytoluene (BHT) tested in this study of 500 mg/kg body weight/day can be considered a NOAEL.
Hypersensitivity, allergy and intolerance
Two patients with chronic idiopathic urticaria were subjected to double-blind, placebo-controlled, oral challenges with a series of food additives. During testing, butylated hydroxytoluene (BHT) and butylated hydroxyanisole (BHA) were identified as causative agents. Avoidance of foods containing butylated hydroxytoluene (BHT) and BHA resulted in long-term reduction in severity and frequency of urticarial episodes 33.
In a double-blind placebo controlled study by Hannuksela and Lahti 34 with challenge tests of 44 patients with chronic urticaria, 91 with atopic dermatitis and 123 with contact dermatitis, none reacted to butylated hydroxytoluene (BHT) when it was ingested in a capsule containing 50 mg butylated hydroxytoluene (BHT).
Signs of contact dermatitis after dermal exposure to butylated hydroxytoluene (BHT) and allergic reactions after oral intake of a mixture of butylated hydroxytoluene (BHT) and butylated hydroxyanisole (BHA) were occasionally reported 35. In another report (Goodman et al., 1990), two patients with chronic
idiopathic urticaria developed exacerbations when challenged with butylated hydroxytoluene (BHT)/butylated hydroxyanisole (BHA), but had less symptomsafter cons uming a butylated hydroxytoluene (BHT)/butylated hydroxyanisole (BHA)-free diet.
When patch-tested on more than 15 individuals, butylated hydroxytoluene (BHT) showed mild skin irritation; a positive skin reaction 14 days later was interpreted as sensitization 36.
Recent patch test results obtained from the medical surveillance of great numbers of workers 37 or patients 38 were all negative.
The European Food Safety Authority (EFSA) Panel noted that these limited reports do not allow any conclusions to be drawn about sensitization to butylated hydroxytoluene (BHT) following oral intake.
Human studies: case studies
Shilian and Goldstone 39 reported a case of gastritis caused by ingestion of butylated hydroxytoluene (BHT) in a 22-year-old woman who ingested 4 g of butylated hydroxytoluene (BHT) on an empty stomach two days before the onset of the gastritis. This amounts to an acute dose of about 67 mg/kg bw assuming a body weight of 60 kg. Later that evening she experienced severe epigastric cramping, generalized weakness, nausea and vomiting, followed by dizziness, confusion and a brief loss of consciousness.
A similar case study was reported by Grogan 40. A 24-year-old woman complained of lightheadedness, unsteadiness of gait and slurred speech. On examination the following findings were noted: dysarthria, wide-based gait, a positive Romberg test, slowed mentation without thought disorder and dysmetria of the left (non-dominant arm). On the evening before admission the patient ingested 80 grams of butylated hydroxytoluene (BHT) suspended in safflower oil on an empty stomach. This dose is estimated to be equivalent to about 1.3 g/kg bw, assuming a 60 kg body weight.
Special studies on hemorrhagic effects
The EU Scientific Committee for Food (SCF) 41 concluded that a series of studies has shown that some, but not all species tested show hemorrhage and/or a reduction in blood prothrombin index after dosing with butylated hydroxytoluene (BHT). The mechanisms by which butylated hydroxytoluene (BHT) brings about these effects appear to be several but the major effect is a reduction in activity of certain clotting factors, principally those which are vitamin K-dependent. The most susceptible species for hemorrhagic effects appears to be the rat, and for this species the NOAEL for transient reduction (1 week duration) of the prothrombin index was approximately 9 mg/kg body weight/day, and for persistent reduction (4 week duration), approximately 250 mg/kg body weight/day given with the diet. Chickens may also respond with hemorrhage of the liver 42.
Administration to groups of five male Slc:ddY mice of 0.5%, 1.0% or 2.0% butylated hydroxytoluene (BHT) in a purified diet (equivalent to 660, 1390 or 2860 mg/kg body weight/day) for 21 days resulted in a dose-related increase in the mortality due to massive haemorrhage in the lungs 22.
Administration to groups of five male Slc:ddY mice of 0.5%, 1.0% or 2.0% butylated hydroxytoluene (BHT) in a purified diet (equivalent to 660, 1390 or 2860 mg/kg body weight/day) for 21 days resulted in a dose-related increase in the mortality due to massive hemorrhage in the lungs 22.
Special studies on effects on the thyroid
Rats were fed 0, 500 or 5000 mg butylated hydroxytoluene (BHT)/kg of feed (estimated to be equivalent to approximately 25 or 250 mg/kg bw/day) for 8, 26 or 90 days and the uptake of 125I by the thyroid was determined. The presence of butylated hydroxytoluene (BHT) in the diet resulted in a marked increase in the uptake of 125I at all time periods studied. When rats were fed butylated hydroxytoluene (BHT) in diets containing varying amounts of iodine (0.12, 0.15 or 0.3 mg iodine/kg feed) for 30 days, there was a significant increase in thyroid weight in butylated hydroxytoluene (BHT) treated animals when compared to controls. butylated hydroxytoluene (BHT) did not change levels of T3 and T4 in the blood. The biological half-life of thyroxine was increased after 13 days on a butylated hydroxytoluene (BHT) diet but returned to normal after 75 days. Electron microscopy of the thyroid glands of rats exposed to 250 mg butylated hydroxytoluene (BHT)/kg bw/day for 28 days showed an increase in the number of follicle cells 22.
Based on this observation the European Food Safety Authority (EFSA) Panel noted that the NOAEL of this study would be 25 mg/kg body weight/day.
Although rodents and humans share a common physiology in regard to the thyroid-pituitary feedback system, a number of factors contribute to the greater sensitivity of the rat to long-term perturbation of the pituitary thyroid axis which predisposes it to a higher incidence of proliferative lesions in response to chronic TSH stimulation than human thyroid.
Both humans and rodents have nonspecific low affinity protein carriers of thyroid hormone (e.g., albumin). However, in humans, other primates, and dogs there is a high affinity binding protein, thyroxine-binding globulin (TBG), which binds T4 (and T3 to a lesser degree); this protein is not present in rodents, birds, amphibians and fish, and has a 1000-fold greater binding affinity than nonspecific low affinity protein carriers.
Although qualitatively the rat is an indicator of a potential human thyroid cancer hazard, humans appear to be quantitatively less sensitive than rodents to developing cancer from perturbations in thyroid-pituitary status. Given that the rodent is a sensitive model for measuring the carcinogenic influences of TSH and that humans appear to be less responsive, effects on rodents would represent a conservative indicator of potential risk for humans. Rodent cancer studies typically include doses that lead to toxicity, including perturbation in thyroid-pituitary function over a lifetime. The relevance of the experimental conditions to anticipated human exposure scenarios (i.e., dose, frequency and time) should be considered. In addition, chemically-induced effects that are produced by short-term disruption in thyroid-pituitary function appear to be reversible, when the stimulus is removed.
Summary
Butylated hydroxytoluene (BHT) is a synthetic antioxidant authorized as a food additive in the European Union that was previously evaluated by the European Union Scientific Committee for Food (SCF) in 1987 and the Joint FAO/WHO Expert Committee on Food Additives (JECFA) several times, the latest in 1996.
The European Food Safety Authority (EFSA) Panel concluded that the present database justifies the revision of the Acceptable Daily Intake (ADI) of 0-0.05 mg/kg body weight/day set by the European Union Scientific Committee for Food (SCF).
Based on a No-observed-adverse-effect level (NOAEL) of 25 mg/kg body weight/day from two 2-generation studies in rats and an uncertainty factor of 100, the European Food Safety Authority (EFSA) Panel derived an ADI of 0.25 mg/kg body weight/day.
Since the NOAEL of 25 mg/kg body weight/day for the reproductive effects is below the BMDL10 values for hepatocellular carcinomas in long-term studies with butylated hydroxytoluene (BHT), the Panel concluded that this NOAEL also covers this effect.
The European Food Safety Authority (EFSA) Panel noted that exposure of adults to butylated hydroxytoluene (BHT) from its use as food additive is unlikely to exceed the newly derived Acceptable Daily Intake (ADI) of 0.25 mg/kg body weight/day at the mean and for the high consumers (95th percentile). Exposure of children to butylated hydroxytoluene (BHT) from its use as food additive is also unlikely to exceed this ADI at the mean, but is exceeded for some European countries (Finland, The Netherlands) at the 95th percentile. If exposure to butylated hydroxytoluene (BHT) from its use as food contact material is also taken into account the new ADI would be exceeded by children at the mean and at the 95th percentile.
Butylated hydroxytoluene MSDS
Butylated hydroxytoluene solubility:
- less than 1 mg/mL at 68° F (20 °C).
- Insoluble in water and propane- 1,2-diol;
- Freely soluble in ethanol
- Freely soluble in toluene; soluble in methanol, isopropanol, methyl ethyl ketone, acetone, cellosolve, benzene, most hydrocarbon solvents, ethanol, petroleum ether, liquid petrolatum (white oil): 0.5% wt/wt; more sol in food oils and fats than butylated hydroxyanisol; good solubility in linseed oil. Insoluble in propylene glycol 43
- Soluble in ethanol, acetone, benzene, petroleum ether; Insoluble in alkali 44
- Soluble in naphtha; insoluble in 10% sodium hydroxide 45
- In water, 0.6 mg/L at 77° F (25 °C) 46
- In water, 0.4 mg/L at 68° F (20 °C)
- Depression of membrane-bound Na+-K+-ATPase activity induced by free radicals and by ischemia of kidney. Kako K, Kato M, Matsuoka T, Mustapha A. Am J Physiol. 1988 Feb; 254(2 Pt 1):C330-7. https://www.ncbi.nlm.nih.gov/pubmed/2831728/
- TemaNord, 2002. Food Additives in Europe 2000. Status of safety assessments of food additives presently permitted in the EU. TemaNord, 2002:560
- HSDB, 2010. 2,6-Di-t-butyl-p-cresol. Hazardous Substances Data Bank (HSDB), a database of the National Library of Medicine’s TOXNET system. https://toxnet.nlm.nih.gov/
- Sai-Ut S, Benjakul S, Rawdkuen S. Retardation of lipid oxidation using gelatin film incorporated with longan seed extract compared with BHT. Journal of Food Science and Technology. 2015;52(9):5842-5849. doi:10.1007/s13197-014-1631-0. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4554665/
- Gray JI. Measurement of lipid oxidation. A review. J Am Oil Chem Soc. 1978;55:539–546. doi: 10.1007/BF02668066
- McClements DJ, Decker EA. Lipid oxidation in oil-in-water emulsions: impact of molecular environment on chemical reactions in heterogeneous food system. J Food Sci. 2000;65:1270–1282. doi: 10.1111/j.1365-2621.2000.tb10596.x.
- Schaich KM, Shahidi F, Zhong Y, Eskin NAM. Lipid oxidation. In: Eskin NAM, Shahidi F, editors. Biochemistry of foods. 3. Amsterdam: Academic; 2013.
- Pereira de Abreu D, Paseiro P, Angulo I, Cruz J. Development of new polyolefin films with nanoclays for application in food packaging. Eur Polym J. 2007;43:2229–2243. doi: 10.1016/j.eurpolymj.2007.01.021.
- Salgado PR, Fernández GB, Drago SR, Mauri AN. Addition of bovine plasma hydrolysates improves the antioxidant properties of soybean and sunflower protein-based films. Food Hydrocoll. 2011;25:1433–1440. doi: 10.1016/j.foodhyd.2011.02.003.
- Moore ME, Han IY, Acton JC, Ogale AA, Barmore CR, Dawson PL. Effects of antioxidants in polyethylene film on fresh beef color. J Food Sci. 2003;68:99–104. doi: 10.1111/j.1365-2621.2003.tb14122.x.
- Giménez B, Moreno S, López-Caballero ME, Montero P, Gómez-Guillén MC. Antioxidant properties of green tea extract incorporated to fish gelatin films after simulated gastrointestinal enzymatic digestion. LWT Food Sci Technol. 2013;53:445–451. doi: 10.1016/j.lwt.2013.03.020.
- Functional, antioxidant and film-forming properties of tuna-skin gelatin with a brown algae extract. Haddar A, Sellimi S, Ghannouchi R, Alvarez OM, Nasri M, Bougatef A. Int J Biol Macromol. 2012 Nov; 51(4):477-83. https://www.ncbi.nlm.nih.gov/pubmed/22721871/
- Tongnuanchan P, Benjakul S, Prodpran T. Physico-chemical properties, morphology and antioxidant activity of film from fish skin gelatin incorporated with root essential oils. J Food Eng. 2013;117:350–360. doi: 10.1016/j.jfoodeng.2013.03.005.
- Barbosa-Pereira L, Manuel Cruz J, Sendon R, Bernaldo de Quirós AR, Ares A, Castro-Lopez M, Abad MJ, Maroto J, Paseiro-Losada P. Development of antioxidant active films containing tocopherols to extend the shelf life of fish. Food Control. 2013;31:236–243. doi: 10.1016/j.foodcont.2012.09.036.
- Rawdkuen S, Suthiluk P, Kamhangwong D, Benjakul S. Mechanical, physico-chemical, and antimicrobial properties of gelatin-based film incorporated with catechin-lysozyme. Chemistry Central Journal. 2012;6:131. doi:10.1186/1752-153X-6-131. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3585795/
- Isolation and characterization of ellagitannins as the major polyphenolic components of Longan (Dimocarpus longan Lour) seeds. Sudjaroen Y, Hull WE, Erben G, Würtele G, Changbumrung S, Ulrich CM, Owen RW. Phytochemistry. 2012 May; 77():226-37. https://www.ncbi.nlm.nih.gov/pubmed/22277734/
- Scientific Opinion on the re-evaluation of butylated hydroxytoluene BHT (E 321) as a food additive. https://efsa.onlinelibrary.wiley.com/doi/pdf/10.2903/j.efsa.2012.2588
- Jensen U, 2006. Overvågning og kontrol af tilsætningsstoffer. Undersøgelse af konserveringsstoffer og andre relevante tilsætningsstoffer i dressinger, saucer og lignende produkter (summary in English). Fødevare Rapport 2006:05.
- Bemrah N, Leblanc J-C and Volatier J-L, 2008. Assessment of dietary exposure in the French population to 13 selected food colours, preservatives,antioxidants, stabilizers, emulsifiers and sweeteners. Food Additives and Contaminants, Part B: Surveillance 1, 2-14.
- Nunn, CJ, 1991. Some comments on the dietary intake of butylated hydroxytoluene, Food and Chemical Toxicology 29, 73–7.
- Sato Y and Kawamura T, 1972. Antioxidants in foods II: colorimetric determination of dibutylhydroxytoluene and butylhydroxyanisole. Journal of the Food Hygiene Society of Japan 13, 53–56.
- JECFA, 1996. 833. Butylated hydroxytoluene. Toxicological evaluation of certain food additives and contaminants in food. Prepared by the forty-fourth meeting of the Joint FAO/WHO Expert Committee on Food Additives (JECFA). WHO Food Additives Series 35.
- Daniel JW, Gage JC, Jones DI and Stevens MA, 1967. Excretion of butylated hydroxy toluene (BHT) and butylated hydroxyanisole (BHA) in man. Food and Cosmetics Toxicology 5, 475-479.
- Madhavi D, Deshpande S and Salunkhe D, 1996. Food Antioxidants: Technological, Toxicological, and Health Perspectives. Marcel Dekker, New York.
- Bomhard EM, Bremmer JN and Herbold BA, 1992. Review of the mutagenicity/genotoxicity of butylated hydroxytoluene. Mutation Research 277, 187-200.
- Williams GM, Iatropoulos MJ and Whysner J, 1999. Safety assessment of butylated hydroxyanisole and butylated hydroxytoluene as antioxidant food additives. Food and Chemical Toxicology 37, 1027-1038.
- https://monographs.iarc.fr/ENG/Monographs/vol1-42/mono40.pdf
- Olsen P, Meyer O, Bille N and Wuertzen G, 1986. Carcinogenicity study on butylated hydroxytoluene in Wistar rats exposed in utero. Food and Chemical Toxicology 24, 1-12.
- Takagi A, Takada K, Sai K, Ochiai T, Matsumoto K, Sekita K, Momma J, Aida Y, Saitoh M, Naitoh K and et al., 1994. Acute, subchronic and chronic toxicity studies of a synthetic antioxidant, 2,2′-methylenebis (4-methyl-6-tert-butylphenol) in rats . Journal of Toxicological Sciences 19, 77-89.
- Botterweck AA, Verhagen H, Goldbohm RA, Kleinjans J and van den Brandt PA, 2000. Intake of butylated hydroxyanisole and butylated hydroxytoluene and stomach cancer risk: results from analyses in the Netherlands Cohort Study. Food and Chemical Toxicology 38, 599-605.
- Brooks T, Hunt P and et al., 1976. Effects of prolonged exposure of mice to butylated hydroxytoluene. Unpublished report from Shell Research, Ltd., Tunstell Lab., Sittingbourne, Kent, UK submitted to the World Health Organization by the authors.
- Eriksson UJ and Siman CM, 1996. Pregnant diabetic rats fed the antioxidant butylated hydroxytoluene show decreased occurrence of malformations in offspring. Diabetes 45, 1497-1502.
- Goodman DL, McDonnell JT, Nelson HS, Vaughan TR and Weber RW, 1990. Chronic urticaria exacerbated by the antioxidant food preservatives, butylated hydroxyanisole (BHA) and butylated hydroxytoluene (BHT). Journal of Allergy and Clinical Immunology 86, 570-575
- Hannuksela M and Lahti A, 1986. Peroral challenge tests with food additives in urticaria and atopic dermatitis. International Journal of Dermatology 25, 178-180.
- Flyvholm MA and Menne T, 1990. Sensitizing risk of butylated hydroxytoluene based on exposure and effect data. Contact Dermatitis 23, 341–345.
- Mallette FS, von Haam E, 1952. Studies on the toxicity and skin effects of compounds used in the rubber and plastics industries. I. Accelerators, activators, and antioxidants. American Medical Association Archives of Industrial Hygiene and Occupational Medicine 5, 311–317.
- Goh CL, Ho SF, 1993. Contact dermatitis from dielectric fluids in electro discharge machining. Contact Dermatitis 28, 134–138.
- Kanerva L, Jolanki R, Alanko K and Estlander T, 1999. Patch test reactions with plastic and glue allergens at an occupational dermatology clinic. Acta Dermato Venereologica 79, 296–300.
- Shilian DM and Goldstone J, 1986. Toxicity of butylated hydroxytoluene. The New England Journal of Medicine 314(10), 648-649.
- Grogan MW, 1986. Toxicity from BHT ingestion. The Western Journal of Medicine 145(2), 245–246.
- SCF, 1989. Butylated hydroxytoluene. In: Reports of the Scientific Committee for Food; Twenty-second series.
- Rao GVS, Parthasarathy KR and Sundararaj A, 2000. Haemorrhagic syndrome in butylated hydroxy toluene (BHT) toxicity in broiler chicken. Indian Veterinary Journal 77, 117-119.
- O’Neil, M.J. (ed.). The Merck Index – An Encyclopedia of Chemicals, Drugs, and Biologicals. Cambridge, UK: Royal Society of Chemistry, 2013., p. 273
- Haynes, W.M. (ed.). CRC Handbook of Chemistry and Physics. 95th Edition. CRC Press LLC, Boca Raton: FL 2014-2015, p. 3-156
- Larranaga, M.D., Lewis, R.J. Sr., Lewis, R.A.; Hawley’s Condensed Chemical Dictionary 16th Edition. John Wiley & Sons, Inc. Hoboken, NJ 2016., p. 443
- Verschueren, K. Handbook of Environmental Data on Organic Chemicals. Volumes 1-2. 4th ed. John Wiley & Sons. New York, NY. 2001, p. 702