Calcium
Calcium (Ca or Ca2+) is the most abundant mineral in your body that is found in bones and teeth, in some foods, added to others, available as a dietary supplement, and present in some medicines (such as gastric antacids e.g. calcium phosphate) 1. Calcium is a mineral that your body needs for numerous functions, including building and maintaining bones and teeth, blood clotting, the transmission of nerve impulses, and the regulation of the heart’s rhythm 2. About 99% of the calcium in your body is stored in your bones and teeth in the form of calcium hydroxyapatite [Ca10(PO4)6(OH)2] crystals, an inorganic matrix of calcium and phosphate 3, 4, 5, 6. While the other 1% of the calcium in your body is found in your blood and soft tissue. Calcium concentrations in your blood and fluid surrounding the cells (extracellular fluid) must be maintained within a narrow concentration range for normal physiological functioning. And your body uses your bones as a reservoir for, and source of, calcium to maintain calcium homeostasis (the state of steady or stable equilibrium of internal physical and chemical conditions) 3. Because the physiological functions of calcium are so vital for survival, your body will stimulate bone resorption (demineralization) to maintain normal blood calcium concentrations when calcium intake is inadequate 6. Thus, adequate intake of calcium is a critical factor in maintaining a healthy skeleton 7.
Calcium is required for narrowing of blood vessels (vascular contraction) and widening of blood vessels (vasodilation), muscle function, nerve transmission, intracellular signaling and hormonal secretion, though less than 1% of total body calcium is needed to support these critical metabolic functions 3. Serum calcium is very tightly regulated and does not fluctuate with changes in dietary intakes; your body uses bone tissue as a reservoir for, and source of calcium, to maintain constant concentrations of calcium in blood, muscle, and intercellular fluids 3. Unlike your teeth, your bone undergoes continuous remodeling, with constant resorption and deposition of calcium into new bone 4. Bone remodeling is required to change bone size during growth, repair damage, maintain serum calcium levels, and provide a source of other minerals 4.
The balance between bone resorption and deposition changes with age. Bone formation exceeds resorption in periods of growth in children and adolescents, whereas in early and middle adulthood both processes are relatively equal. At birth, the body contains about 26 to 30 g calcium 8. This amount rises quickly after birth, reaching about 1,200 g (1.2 kg) in women and 1,400 g (1.4 kg) in men by adulthood 3. These levels remain constant in men, but they start to drop in women as a result of increases in bone remodeling due to decreased estrogen production at the start of menopause 3. In aging adults, particularly among postmenopausal women, bone breakdown exceeds formation, resulting in bone loss that increases the risk of osteoporosis over time 3.
Your body gets the calcium you need in two ways. One is by eating foods or supplements that contain calcium. Good sources include dairy products, which have the highest concentration per serving of highly absorbable calcium, and dark leafy greens or dried beans, which have varying amounts of absorbable calcium. Calcium supplements often contain vitamin D; taking calcium paired with vitamin D seems to be more beneficial for bone health than taking calcium alone 2.
Calcium is found in many foods. It is important to get plenty of calcium in the foods you eat. You can get recommended amounts of calcium by eating a variety of foods, including the following 9:
- Dairy products such as milk, cheese, and yogurt are the main food sources of calcium for most people in the United States.
- Fish with soft bones that you eat, such as canned sardines and salmon.
- Certain vegetables, such as kale, broccoli, and Chinese cabbage (bok choi) also contain calcium.
- Calcium is added to some breakfast cereals and beverages, including many fruit juices and milk substitutes such as soy and almond beverages, as well as some brands of tofu and ready-to-eat cereals. To find out whether these foods have calcium added, check the product labels.
- Most grains (such as breads, pastas, and unfortified cereals) do not have high amounts of calcium. However, because people eat them often, what they contribute adds up.
The other way your body gets calcium is by pulling it from your bones. This happens when the blood levels of calcium drop too low (hypocalcemia), usually when it’s been awhile since having eaten a meal containing calcium. Ideally, the calcium that is “borrowed” from the bones will be replaced at a later point. But, this doesn’t always happen. Most important, this payback can’t be accomplished simply by eating more calcium 2.
The exact amount of calcium you need depends on your age, sex and other factors 9. Growing children and teenagers need more calcium than young adults. Older women need plenty of calcium to prevent osteoporosis. People who do not eat enough high-calcium foods should take a calcium supplement.
An inverse relationship exists between calcium intake and absorption 8. Absorption of calcium from food is about 45% at intakes of 200 mg/day but only 15% when intakes are higher than 2,000 mg/day 10. Age can also affect absorption of dietary calcium 3, 4. Net absorption of dietary calcium is as high as 60% in infants and young children, who need substantial amounts to build bone, but it decreases to about 25% in adulthood and continues to decline with age 3.
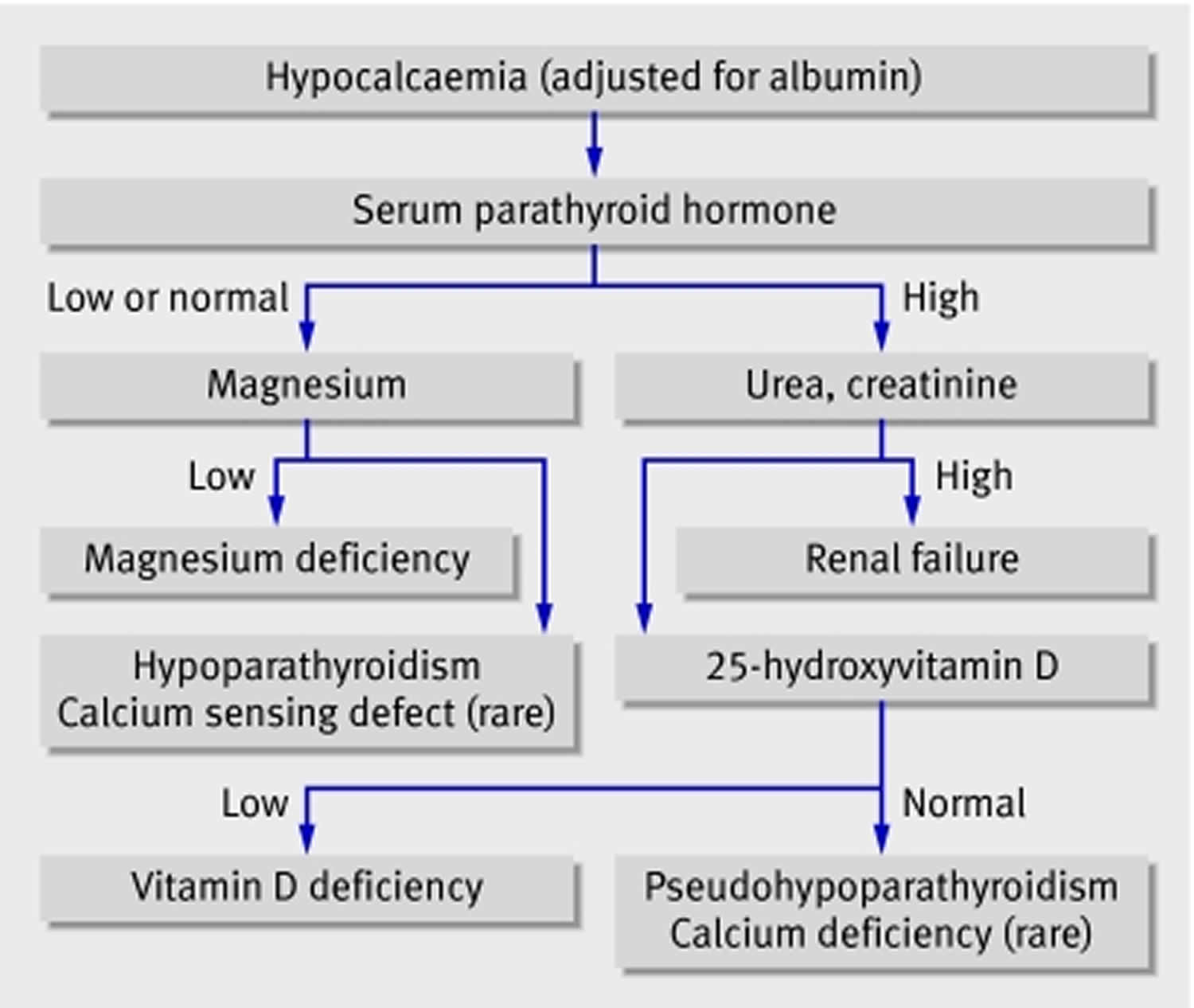
Total calcium levels can be measured in serum or plasma; serum levels are typically 8.8 to 10.4 mg/dL (2. 2 to 2.6 mmol/L) in healthy people 11, 3. However, serum calcium levels do not reflect nutritional status because of their tight homeostatic control 4. Levels of ionized or free calcium (Ca2+), the biologically active form, in serum are also used to measure calcium status. The normal range of ionized calcium (Ca2+) in healthy people is 4.6 to 5.3 mg/dL (1.15 to 1.33 mmol/L) 11. Dual x-ray absorptiometry (DEXA) testing of bone mineral density (BMD) can be used to assess cumulative calcium status over the lifetime because the skeleton stores almost all calcium in the body 12.
How your body controls blood calcium levels
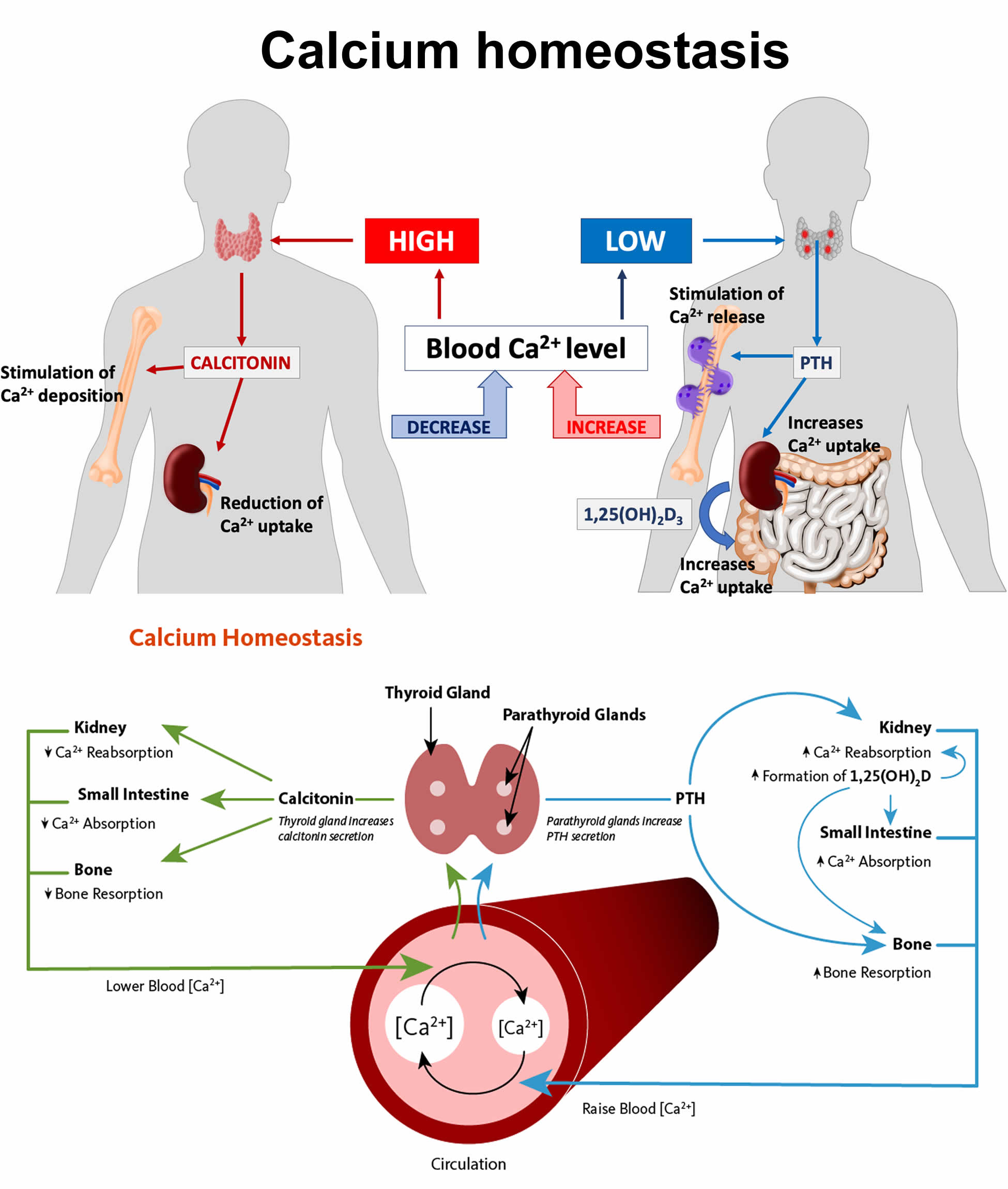
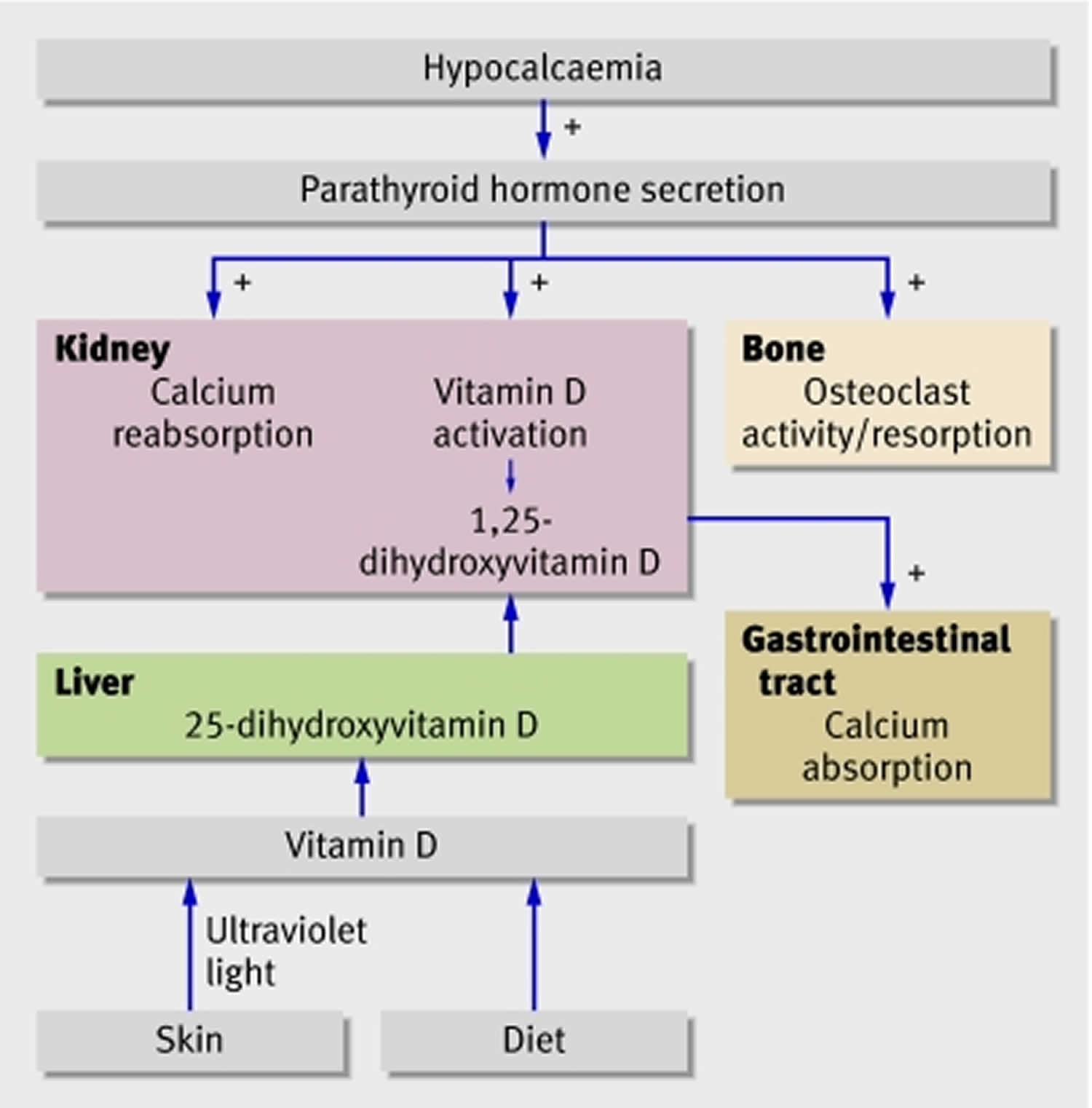
The metabolism of calcium and of phosphate is intimately related. Normally, your body controls blood calcium by adjusting the levels of several hormones. The regulation of both calcium and phosphate balance is greatly influenced by concentrations of circulating parathyroid hormone (PTH), vitamin D, and, to a lesser extent, calcitonin (Figure 1). When blood calcium levels are low, your parathyroid glands (four pea-sized glands that lie just behind the thyroid glands in your neck) secrete a hormone called parathyroid hormone (PTH) (Figure 1). The parathyroid hormone (PTH) helps your bones release calcium into the blood. Vitamin D is also important in keeping calcium levels in the normal range. Vitamin D, which is actually a hormone, helps your body absorb calcium and move it from your intestines into your blood. Together, parathyroid hormone (PTH) and vitamin D, along with other hormones and minerals, help move calcium in or out of body tissues to keep your blood calcium at a normal level.
Parathyroid hormone (PTH) is secreted by the parathyroid glands. Parathyroid hormone (PTH) has several actions, but perhaps the most important is to defend against low blood calcium or hypocalcemia. Parathyroid cells sense decreases in serum calcium and, in response, release preformed PTH into the circulation. Parathyroid hormone (PTH) increases serum calcium within minutes by increasing renal and intestinal absorption of calcium and by rapidly mobilizing calcium and phosphate from bone (bone resorption) (Figure 1). Kidney calcium excretion generally parallels sodium excretion and is influenced by many of the same factors that govern sodium transport in the proximal tubule. However, PTH enhances distal tubular calcium reabsorption independently of sodium.
Parathyroid hormone (PTH) also decreases renal phosphate reabsorption and thus increases renal phosphate losses. Kidney phosphate loss prevents the solubility product of calcium and phosphate from being exceeded in plasma as calcium concentrations rise in response to PTH.
Parathyroid hormone (PTH) also increases serum calcium by stimulating conversion of vitamin D to its most active form, calcitriol also known as 1,25-dihydroxycholecalciferol or 1,25-dihydroxyvitamin D. This form of vitamin D increases the percentage of dietary calcium absorbed by the intestine. Despite increased calcium absorption, long-term increases in PTH secretion generally result in further bone resorption by inhibiting osteoblastic function and promoting osteoclastic activity. PTH and vitamin D both function as important regulators of bone growth and bone remodeling.
Radioimmunoassays for the intact PTH molecule are still the recommended way to test for PTH. Second-generation assays for intact PTH are available. These tests measure bioavailable PTH or complete PTH. They give values equal to 50 to 60% of those obtained with the older assay. Both types of assays can be used for diagnosing primary hyperparathyroidism or monitoring hyperparathyroidism secondary to renal disease, as long as normal ranges are noted.
PTH increases urinary cAMP. Sometimes total or nephrogenous cAMP excretion is measured in diagnosis of pseudohypoparathyroidism.
Calcitoninis secreted by the thyroid parafollicular cells (C cells). Calcitonin tends to lower serum calcium concentration by enhancing cellular uptake, renal excretion, and bone formation. The effects of calcitonin on bone metabolism are much weaker than those of either PTH or vitamin D.
Figure 1. Calcium homeostasis
Calcium and phosphate concentrations are also linked by their ability to chemically react to form calcium phosphate. The product of concentrations of calcium and phosphate (in mEq/L) is estimated to be < 60 normally; when the product exceeds 70, precipitation of calcium phosphate crystals in soft tissue is much more likely. Calcification of vascular tissue accelerates arteriosclerotic vascular disease and may occur when the calcium and phosphate product is even lower (> 55), especially in patients with chronic kidney disease.
Calcium is absorbed passively (no cellular energy required) in the intestines by diffusing through the spaces between cells. It is also absorbed actively (cellular energy required) through intestinal cells by binding to a transport protein known as calbindin. The production of calbindin is dependent on vitamin D 13. Not all calcium consumed is actually absorbed in the gut. Humans absorb about 30% of the calcium in foods, but this varies depending upon the type of food consumed 14. Other factors also affect calcium absorption including the following:
- Amount consumed: the efficiency of absorption decreases as calcium intake increases.
- Age and life stage: net calcium absorption is as high as 60% in infants and young children, who need substantial amounts of the mineral to build bone.
- Absorption decreases to 15%–20% in adulthood (though it is increased during pregnancy) and continues to decrease as people age; compared with younger adults, recommended calcium intakes are higher for females older than 50 years and for both males and females older than 70 years.
- Vitamin D intake: this nutrient, obtained from food and produced by skin when exposed to sunlight of sufficient intensity, improves calcium absorption.
- Other components in food: phytic acid and oxalic acid, found naturally in some plants, bind to calcium and can inhibit its absorption. Foods with high levels of oxalic acid include spinach, collard greens, sweet potatoes, rhubarb, and beans. Among the foods high in phytic acid are fiber-containing whole-grain products and wheat bran, beans, seeds, nuts, and soy isolates. The extent to which these compounds affect calcium absorption varies. Research shows, for example, that eating spinach and milk at the same time reduces absorption of the calcium in milk. In contrast, wheat products (with the exception of wheat bran) do not appear to lower calcium absorption. For people who eat a variety of foods, these interactions probably have little or no nutritional consequence and, furthermore, are accounted for in the overall calcium Dietary Reference Intakes (DRIs), which factor in differences in absorption of calcium in mixed diets.
Some absorbed calcium is eliminated from the body in urine, feces, and sweat. This amount is affected by such factors as the following:
- Sodium and protein intakes: high sodium intake increases urinary calcium excretion. High protein intake also increases calcium excretion and was therefore thought to negatively affect calcium status. However, more recent research suggests that high protein intake also increases intestinal calcium absorption, effectively offsetting its effect on calcium excretion, so whole body calcium retention remains unchanged.
- Caffeine intake: this stimulant in coffee and tea can modestly increase calcium excretion and reduce absorption. One cup of regular brewed coffee, for example, causes a loss of only 2–3 mg of calcium. Moderate caffeine consumption (1 cup of coffee or 2 cups of tea per day) in young women has no negative effects on bone.
- Alcohol intake: alcohol intake can affect calcium status by reducing its absorption and by inhibiting enzymes in the liver that help convert vitamin D to its active form. However, the amount of alcohol required to affect calcium status and whether moderate alcohol consumption is helpful or harmful to bone is unknown.
- Phosphorus intake: the effect of this mineral on calcium excretion is minimal. Several observational studies suggest that consumption of carbonated soft drinks with high levels of phosphate is associated with reduced bone mass and increased fracture risk. However, the effect is probably due to replacing milk with soda rather than the phosphorus itself.
- Fruit and vegetable intakes: metabolic acids produced by diets high in protein and cereal grains increase calcium excretion. Fruits and vegetables, when metabolized, shift the acid/base balance of the body towards the alkaline by producing bicarbonate, which reduces calcium excretion. However, it is unclear if consuming more fruits and vegetables affects bone mineral density. These foods, in addition to reducing calcium excretion, could possibly reduce calcium absorption from the gut and therefore have no net effect on calcium balance.
Vitamin D
Vitamin D is required for optimal calcium absorption. Several other nutrients and non-nutrients influence the retention of calcium by the body and may affect calcium nutritional status 6.
Sodium
Dietary sodium (sodium chloride salt) is a major determinant of urinary calcium loss 3. High-sodium (salt) intake results in increased loss of calcium in the urine, possibly due to competition between sodium and calcium for reabsorption in the kidneys or by an effect of sodium on parathyroid hormone (PTH) secretion. Every 1-gram (g) increment in sodium (2.5 g of sodium chloride; NaCl salt) excreted by the kidneys has been found to draw about 26.3 milligrams (mg) of calcium into the urine 3. A study conducted in adolescent girls reported that a high-salt diet had a greater effect on urinary sodium and calcium excretion in White compared to Black girls, suggesting differences among ethnic groups 15. In adult women, each extra gram of salt (sodium) consumed per day is projected to produce an additional rate of bone loss of 1% per year if all of the calcium loss comes from the skeleton.
A number of cross-sectional and intervention studies have suggested that high-sodium intakes are deleterious to bone health, especially in older women 16. A two-year longitudinal study in postmenopausal women found increased urinary sodium excretion (an indicator of increased sodium intake) to be associated with decreased bone mineral density (BMD) at the hip 17. Another study in 40 postmenopausal women found that adherence to a low-sodium diet (2 g/day) for six months was associated with significant reductions in sodium excretion, calcium excretion, and amino-terminal propeptide of type 1 collagen, a biomarker of bone resorption. Yet, these associations were only observed in women with elevated baseline urinary sodium excretions 18. Finally, in a randomized, placebo-controlled study in 60 postmenopausal women, potassium citrate supplementation has been found to prevent an increase in calcium excretion induced by the consumption of a high-sodium diet (≥5,000 mg/day of elemental sodium) for four weeks 19.
Protein
Increasing dietary protein intake enhances intestinal calcium absorption, as well as urinary calcium excretion 20. The Recommended Dietary Allowance (average daily level of intake sufficient to meet the nutrient requirements of nearly all (97%–98%) healthy individuals; often used to plan nutritionally adequate diets for individuals) for protein is 46 grams (g)/day for adult women and 56 g/day for adult men; however, the average intake of protein in the US tends to be higher (about 70 g/day in adult women and over 100 g per day in adult men) 21. It was initially thought that high-protein diets may result in a negative calcium balance (when the sum of urinary and fecal calcium excretion becomes greater than calcium intake) and thus increase bone loss 22. However, most observational studies have reported either no association or positive associations between protein intake and bone mineral density in children, adults, and elderly subjects 23. The overall calcium balance appears to be unchanged by high dietary protein intake in healthy individuals 24, and current evidence suggests that increased protein intakes in those with adequate supplies of protein, calcium, and vitamin D do not adversely affect bone mineral density (BMD) or fracture risk 25.
Phosphorus
Phosphorus, which is typically found in protein-rich food, tends to increase the excretion of calcium in the urine. Diets with low calcium-to-phosphorus ratios (Ca:P ≤0.5) have been found to increase parathyroid hormone (PTH) secretion and urinary calcium excretion 26, 27. Also, the intestinal absorption and fecal excretion of calcium and phosphorus are influenced by calcium-to-phosphorus ratios of ingested food. In the intestinal lumen, calcium salts can bind to phosphorus to form complexes that are excreted in the feces. This forms the basis for using calcium salts as phosphorus binders to lower phosphorus absorption in individuals with kidney failure or disease 28. Increasing phosphorus intakes from cola soft drinks (high in phosphoric acid) and food additives (high in phosphates) may have adverse effects on bone health 29. At present, there is no convincing evidence that the dietary phosphorus levels experienced in the US adversely affect bone health. Yet, the substitution of large quantities of phosphorus-containing soft drinks for milk or other sources of dietary calcium may represent a serious risk to bone health in adolescents and adults.
Caffeine
Exposure to caffeine concentrations ≤400 mg/day have led to increased urinary calcium content in two randomized controlled trials 30, 31. However, caffeine intakes of 400 mg/day did not significantly change urinary calcium excretion over 24 hours in premenopausal women when compared to a placebo 32. A systematic review of 14 studies recently concluded that daily intake of ≤400 mg of caffeine was unlikely to interfere with calcium homeostasis, impact negatively bone mineral density, or increase the risks of osteoporosis and fracture in individuals with adequate calcium intakes 33.
What does calcium do?
Calcium is a mineral your body needs to build and maintain strong bones and to carry out many important functions. Your body needs calcium for muscles to move and for nerves to carry messages between your brain and every part of your body. Calcium also helps blood vessels move blood throughout your body and helps release hormones that affect many functions in your body. Vitamin D helps your body absorb calcium.
Structural element in bones and teeth
Calcium is a major structural element in bones and teeth. The mineral component of bone consists mainly of hydroxyapatite [Ca10(PO4)6(OH)2] crystals, which contain large amounts of calcium, phosphorus, and oxygen. Bone is a dynamic tissue that is remodeled throughout life. Bone cells called osteoclasts begin the process of remodeling by dissolving or resorbing bone. Bone-forming cells called osteoblasts then synthesize new bone to replace the bone that was resorbed. During normal growth, bone formation exceeds bone resorption. Osteoporosis may result when bone resorption chronically exceeds formation 3.
Calcium homeostasis
Calcium concentrations in the blood and fluid that surround cells are tightly controlled in order to preserve normal physiological function. A slight drop in blood calcium concentration (e.g., in the case of inadequate calcium intake) is sensed by the parathyroid glands, resulting in their increased secretion of parathyroid hormone (PTH). In the kidneys, PTH stimulates the conversion of vitamin D into its active form (1,25-dihydroxyvitamin D; calcitriol), which rapidly decreases urinary excretion of calcium but increases urinary excretion of phosphorus. Elevations in PTH also stimulates bone resorption, resulting in the release of bone mineral (calcium and phosphate) — actions that also contribute to restoring serum calcium concentrations. Increased circulating calcitriol (1,25-dihydroxyvitamin D) also triggers intestinal absorption of both calcium and phosphorus. Like PTH, calcitriol (1,25-dihydroxyvitamin D) stimulates the release of calcium from bone by activating osteoclasts (bone-resorbing cells). When blood calcium rises to normal levels, the parathyroid glands stop secreting PTH. A slight increase in blood calcium concentration stimulates the production and secretion of the peptide hormone, calcitonin, by the thyroid gland. Calcitonin inhibits PTH secretion, decreases both bone resorption and intestinal calcium absorption, and increases urinary calcium excretion (Figure 1). Finally, acute changes in blood calcium concentrations do not seem to elicit the secretion of the phosphaturic hormone fibroblast growth factor 23 (FGF-23), which is produced by bone-forming cells (osteoblasts/osteocytes) in response to increases in phosphorus intake 34. While this complex system allows for rapid and tight control of blood calcium concentrations, it does so at the expense of the skeleton 3.
Cell signaling
Calcium plays a role in mediating the constriction and relaxation of blood vessels (vasoconstriction and vasodilation), nerve impulse transmission, muscle contraction, and the secretion of hormones like insulin 3. Excitable cells, such as skeletal muscle and nerve cells, contain voltage-dependent calcium channels in their cell membranes that allow for rapid changes in calcium concentrations. For example, when a nerve impulse stimulates a muscle fiber to contract, calcium channels in the cell membrane open to allow calcium ions into the muscle cell. Within the cell, these calcium ions bind to activator proteins, which help release a flood of calcium ions from storage vesicles of the endoplasmic reticulum (ER) inside the cell. The binding of calcium to the protein troponin-c initiates a series of steps that lead to muscle contraction. The binding of calcium to the protein calmodulin activates enzymes that break down muscle glycogen to provide energy for muscle contraction. Upon completion of the action, calcium is pumped outside the cell or into the endoplasmic reticulum (ER) until the next activation 35.
Regulation of protein function
Calcium is necessary to stabilize a number of proteins, including enzymes, optimizing their activities. The binding of calcium ions is required for the activation of the seven “vitamin K-dependent” clotting factors in the coagulation cascade. The term, “coagulation cascade,” refers to a series of events, each dependent on the other that stops bleeding through clot formation.
Calcium Supplements
Calcium is available in many dietary supplements, including multivitamin/mineral products and supplements containing calcium only or calcium plus vitamin D 8. The two main forms of calcium in supplements are carbonate and citrate. Calcium carbonate is more commonly available and is both inexpensive and convenient 36. Due to its dependence on stomach acid for absorption, calcium carbonate is absorbed most efficiently when taken with food, whereas calcium citrate is absorbed equally well when taken with or without food 37. Calcium citrate is also useful for people with achlorhydria, inflammatory bowel disease, or absorption disorders 3. Other calcium forms in supplements or fortified foods include gluconate, lactate, and phosphate. Calcium citrate malate is a well-absorbed form of calcium found in some fortified juices 38.
Calcium supplements contain varying amounts of elemental calcium, multivitamin or mineral supplements commonly contain about 200 to 300 mg, and common amounts in calcium or calcium plus vitamin D supplements are 500 or 600 mg 39. Calcium carbonate is 40% calcium by weight, whereas calcium citrate is 21% calcium. Fortunately, elemental calcium is listed in the Supplement Facts panel, so consumers do not need to calculate the amount of calcium supplied by various forms of calcium supplements 39.
The percentage of calcium absorbed depends on the total amount of elemental calcium consumed at one time; as the amount increases, the percentage absorption decreases. Absorption is highest in doses ≤500 mg 40. For example, the body absorbs about 36% of a 300 mg calcium dose and 28% of a 1,000 mg dose 41. So, for example, one who takes 1,000 mg/day of calcium from supplements might split the dose and take 500 mg at two separate times during the day.
Some individuals who take calcium supplements might experience gastrointestinal side effects including gas, bloating, constipation, or a combination of these symptoms. Calcium carbonate appears to cause more of these side effects than calcium citrate 3, so consideration of the form of calcium supplement is warranted if these side effects are reported. Other strategies to alleviate symptoms include spreading out the calcium dose throughout the day and/or taking the supplement with meals.
Calcium Phosphate Supplement
The beneficial effects of calcium phosphate mainly focus on the intestinal metabolism, e.g., bile acid metabolism, fatty acid (cholesterol) excretion, and modulation of the gut microbiota 42, 43, 44, 45. Calcium from tricalcium phosphate (CaP, a water-insoluble compound at neutral pH value), is partly absorbed in the human gut; but the main part of the calcium and phosphorus is precipitated to amorphous calcium phosphate in the gut, and thus, not absorbed 46. Nevertheless, supplementation with vitamin D3 and calcium reduces the risk of hip fractures and other nonvertebral fractures among elderly women 47. Supplementation with daily 10 μg vitamin D3 significantly increases plasma 25-(OH)D concentration. The combination with daily 1 g calcium (as CaP) has a further increasing effect on the 25-(OH)D concentration. Both CaP alone and in combination with vitamin D3 have no beneficial effect on bone remodelling markers and on the metabolism of calcium, phosphorus, magnesium and iron 48.
Calcium Citrate for Kidney Stone Treatment
Kidney stones are one of the most common disorders of the urinary tract. They typically affect people aged 40 to 60 years of age and are twice as common in men than women although recent data suggest the risks are more equal. Calcium stones are the most common type of kidney stone and occur in two major forms: calcium oxalate and calcium phosphate. Kidney stones can cause severe abdominal pain and may require urgent treatment; they are one of the main causes of unscheduled admissions in urological practice. Following treatment even first time stone formers have a risk for recurrence which increased with each subsequent stone. This increased risk of recurrence of stones is mainly attributed to altered composition of urine i.e. low citrate levels. These people have a higher incidence of calcium phosphate and calcium oxalate stones. Various prevention strategies including increased fluid intake and oral citrate supplements have been tried to modify the chemical composition of the urine. Citrate therapy is believed to stop crystals from growing into stones. Oral citrate therapy increases the urinary citrate levels, which in turn binds with calcium and inhibits the crystallisation thus reduces stone formation. Despite the widespread use of oral citrate therapy for prevention and treatment of calcium oxalate stones, the evidence to support its clinical efficacy remains uncertain.
In a Cochrane review with 477 subjects, most of whom had oxalate stones. Of these, 247 participants compared potassium citrate with placebo or no intervention; 166 participants compared potassium‐sodium citrate with no intervention; and 64 participants compared potassium‐magnesium citrate with placebo. Overall, quality of the reporting of the included studies was considered moderate to poor, and there was a high risk of attrition bias in two studies.
Compared with placebo or no intervention, citrate therapy significantly reduced the stone size (4 studies, 160 participants). New stone formation was significantly lower with citrate therapy compared to control (7 studies, 324 participants). The beneficial effect on stone size stability was also evident (4 studies, 160 participants). Adverse events were reported in four studies, with the main side effects being upper gastrointestinal disturbance and one patient reported a rash. There were more gastrointestinal adverse events in the citrate group; however this was not significant (4 studies, 271 participants). There were significantly more dropouts due to adverse events with citrate therapy compared to control (4 studies, 271 participants). The need for retreatment was significantly less with citrate therapy compared to control (2 studies, 157 participants).
Authors’ conclusions: Citrate salts prevent new stone formation and reduce further stone growth in patients with residual stones that predominantly contain oxalate. The quality of reported literature remains moderate to poor; hence a well‐designed statistically powered multi‐centre random controlled trial is needed in order to answer relevant questions concerning the efficacy of citrate salts 49.
Calcium Acetate as Phosphate binders for preventing and treating bone disease in chronic kidney disease patients
People with chronic kidney disease (CKD) develop impaired excretion of the dietary phosphorus. This results in a condition known as mineral and bone disorder in chronic kidney disease (CKD‐MBD). Mineral and bone disorder in chronic kidney disease is characterized by high bone turnover, increased musculoskeletal morbidity including bone pain and muscle weakness, and vascular calcification which may contribute to the high incidence of cardiovascular disease and associated deaths. Several agents such as phosphate binders, vitamin D compounds, and calcimimetics are widely used to slow the development and progression of mineral and bone disorder in chronic kidney disease complications.
Several phosphate binders, including aluminium and calcium‐containing agents, have been widely used since 1970. The use of newer non‐calcium or aluminium‐based agents, such as sevelamer hydrochloride compounds and lanthanum carbonate is increasing although the cost is greater than the older phosphate binders. The avoidance of calcium‐based binding agents to lower phosphorus in chronic kidney disease theoretically reduces the risk of vascular calcification and cardiovascular disease. The balance between calcium‐free phosphate binders reducing clinical events in chronic kidney disease versus their cost remains controversial. Recently released KDIGO guidelines recommend restricting the use of calcium‐based binders in people with persistent or recurrent hypercalcaemia or arterial calcification, or both.
A review of 60 studies involving 7631 participants comparing phosphate binders to placebo or other phosphate binders. There was no significant reduction in all‐cause mortality (10 studies, 3079 participants) or serum calcium by phosphorus product with sevelamer hydrochloride compared to calcium‐based agents. There was a significant reduction in phosphorus (16 studies, 3126 participants) and parathyroid hormone (12 studies, 2551 participants) levels, but a significant increase in the risk of hypercalcaemia (12 studies, 1144 participants) with calcium salts compared to sevelamer hydrochloride. There was a significant increase in the risk of adverse gastrointestinal events with sevelamer hydrochloride compared to calcium salts (5 studies, 498 participants). Compared with calcium‐based agents, lanthanum significantly reduced serum calcium (2 studies, 122 participants) and the calcium by phosphorous product, but not serum phosphorus levels. The effects of calcium acetate on biochemical end‐points were similar to those of calcium carbonate. The phosphorus lowering effects of novel agents such as ferric citrate, colestilan and niacinamide were only reported in a few studies.
Conclusion of this review is that available phosphate‐binding agents have been shown to reduce phosphorus levels in comparison to placebo. However, there are insufficient data to establish the comparative superiority of novel non‐calcium binding agents over calcium‐containing phosphate binders for patient‐level outcomes such as all‐cause mortality and cardiovascular end‐points in chronic kidney disease 50. This finding is in agreement with another meta-analysis comparing the effects of calcium-based versus non-calcium-based phosphate binders on mortality, cardiovascular events and vascular calcification in patients with chronic kidney disease. Despite the trends observed, the study authors did not find a statistically significant difference in cardiovascular mortality and coronary artery calcification in patients receiving calcium-based phosphate binders compared to non-calcium-based phosphate binders. However, the data are limited by the small number of studies and the confidence intervals do not exclude a potentially important beneficial effect. Therefore, further randomized trials are required 51.
Calcium Supplements Side Effects
Is it safe to take calcium supplements ?
For most people, it is safe to eat foods containing calcium and to take calcium supplements that together do not exceed the tolerable upper intake level of 2.5 grams of calcium per day 52. This upper level for daily calcium intake in adults is the highest level that likely will not pose risks of unwanted side effects in the general population. The upper level of 2.5 grams a day is an average recommendation for all healthy people who are older than a year, regardless of gender 52.
Consuming too much calcium—in excess of 5 grams a day, or 3 grams a day in people with existing kidney problems 53 can lead to several harmful side effects. The milk-alkali syndrome, a triad of hypercalcemia, metabolic alkalosis, and renal insufficiency, was identified in 1923 as an adverse effect of peptic ulcer disease therapies involving the use of dairy products and alkaline powders 54. Most of these side effects result from people taking too many calcium supplements. Recent trends in the prevention and treatment of osteoporosis using widely available over-the-counter (OTC) calcium supplements appear to be contributing to its return 55. Rare harmful side effects from excess calcium include kidney stones 56, hypercalcemia (too much calcium in the blood), and kidney failure 57. In addition, excessive consumption of milk (which is high in calcium) and some types of antacids, especially antacids containing calcium carbonate or sodium bicarbonate (baking soda), over a long period of time can cause milk-alkali syndrome, a condition that can also lead to calcium deposits in the kidneys and other tissues and to kidney failure 53, 58, 59.
Interactions with Medications
The presence of calcium decreases iron absorption from nonheme sources (i.e., most supplements and food sources other than meat) 6. However, calcium supplementation up to 12 weeks has not been found to change iron nutritional status, probably due to a compensatory increase in iron absorption 7. Individuals taking iron supplements should take them two hours apart from calcium-rich food or supplements to maximize iron absorption. Although high calcium intakes have not been associated with reduced zinc absorption or zinc nutritional status, an early study in 10 men and women found that 600 mg of calcium consumed with a meal halved the absorption of zinc from that meal 60. Supplemental calcium (500 mg calcium carbonate) has been found to prevent the absorption of lycopene (a nonprovitamin A carotenoid) from tomato paste in 10 healthy adults randomized into a cross-over study 61.
Calcium also has the potential to interact with certain medications, and several types of medications might adversely affect calcium levels. A few examples are provided below. Individuals taking these and other medications on a regular basis should discuss their calcium status with their health care providers.
Dolutegravir
Dolutegravir (Dovato, Tivicay) is an HIV integrase inhibitor used in adults and children. Concomitant use of calcium supplements and dolutegravir can reduce blood levels of dolutegravir substantially, apparently through chelation 62, 63. The labels approved by the FDA for dolutegravir advise patients to take dolutegravir 2 hours before or 6 hours after taking calcium supplements 64, 65.
Levothyroxine
Calcium carbonate supplements can interfere with the absorption of levothyroxine (Synthroid, Levoxyl, and others), a thyroid hormone used to treat hypothyroidism and thyroid cancer 66, 67, 68. The FDA-approved label for this medication instructs patients taking calcium carbonate supplements to avoid taking levothyroxine within 4 hours of taking the supplement 69.
Lithium
Long-term use of lithium (Eskalith, Lithobid), a treatment for bipolar disorder, can lead to hypercalcemia, and use of both lithium and calcium supplements could increase this risk 70
Quinolone antibiotics
Simultaneous use of calcium supplements and quinolone antibiotics—such as ciprofloxacin (Cipro), gemifloxacin (Factive), and moxifloxacin (Avelox)—can reduce the absorption of quinolones 71, 72. Taking the antibiotic 2 hours before or 2 hours after calcium supplements prevents this effect 71.
Calcium as Medicines
Because of its ability to neutralize stomach acid, calcium carbonate is found in some over-the-counter antacid products, such as Tums® and Rolaids®. Depending on its strength, each chewable pill or soft chew provides 270 to 400 mg of elemental calcium. As noted above, calcium carbonate is an acceptable form of supplemental calcium, especially for individuals who have normal levels of stomach acid 73.
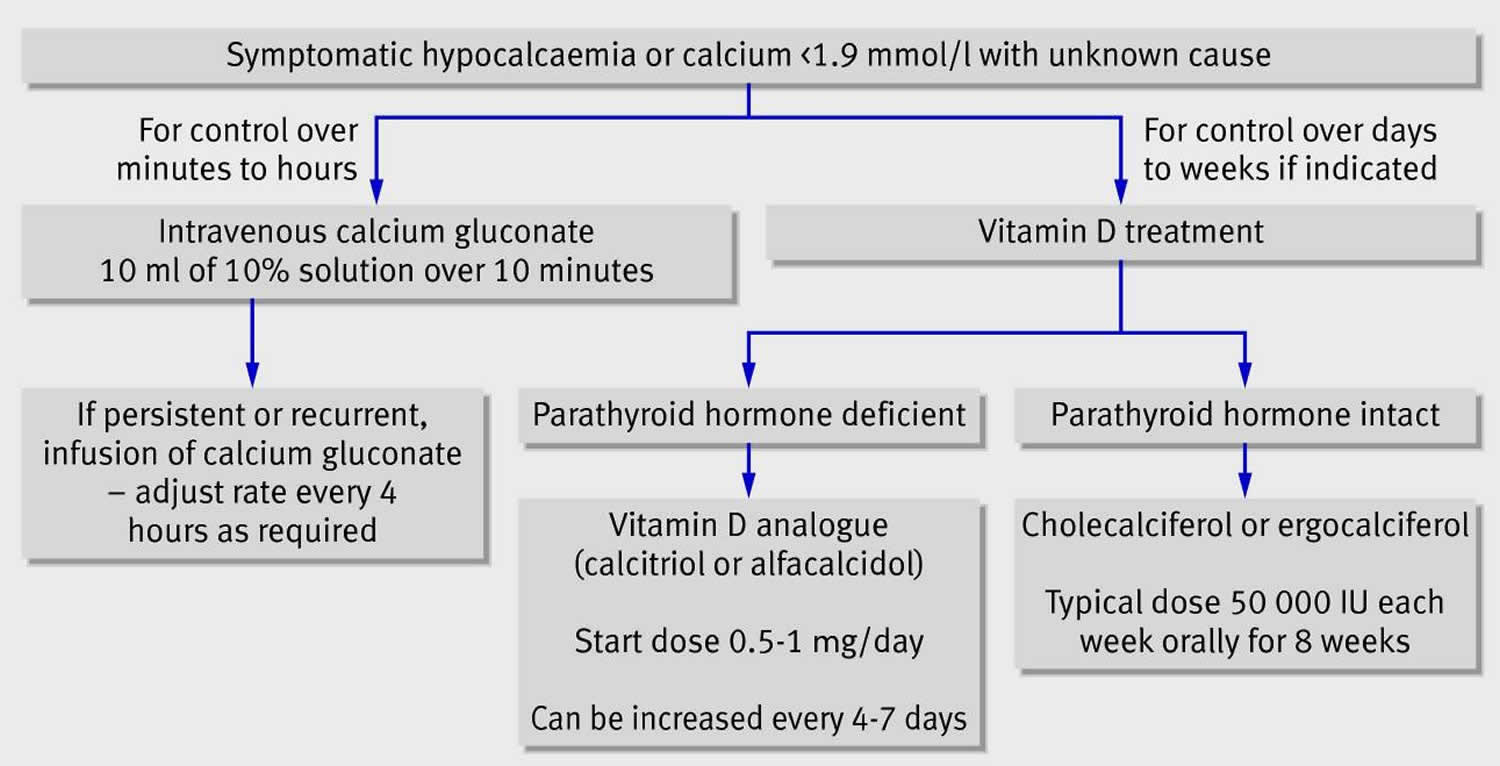
Calcium Gluconate
Calcium Gluconate injection treats too little calcium in the blood. Also treats black widow spider bites, lead colic, overdose of magnesium or certain heart medicines, and rickets. This medicine is also used for life support and life-threatening heart conditions 74.
Calcium Oxide
Materials used in the production of dental bases, restorations, impressions, prostheses, etc. Nano-thick calcium oxide armed titanium: boosts bone cells against methicillin-resistant Staphylococcus aureus (MRSA) in rabbits 75.
Calcium Phosphate
Synthetic bio-inert materials are currently used as an alternative to autogenous bone graft. Calcium hydroxyapatite (HA) and Beta tri-calcium phosphate (β-TCP), which belong to the calcium phosphate ceramics group, are biocompatible and osteo-conductive. Calcium hydroxyapatite and β-TCP are excellent bone graft substitutes for autogenous bone graft in filling voids after curettage of benign bone tumors 76. Calcium phosphate is also used as gastric antacid 1.
Calcium Sulfate
The use of calcium sulfate (plaster of Paris) has been advocated to repair bony defects because of its unique capability of stimulating osteoneogenesis. Plaster of Paris can be used as a bony alloplast, and it can be analyzed histologically. Sinus roentgenograms and technetium Tc 99m medronate bone scanning further support the use of plaster of Paris as an alloplast and assess its osteoneogenic capacity when implanted in the frontal sinus of dogs; complete bone regeneration was demonstrated in six dogs within four to six months. The use of plaster of Paris for bone reconstruction in the head and neck can be applied in surgery. The experience with plaster of Paris to date, although limited, shows it to be safe and highly encouraging as an effective bone allograft 77).
Calcium Health Benefits
Many claims are made about calcium’s potential benefits in health promotion and disease prevention and treatment.
Scientists are studying calcium to understand how it affects health. Here are several examples of what this research has shown:
Bone health and osteoporosis
Bones increase in size and mass during periods of growth in childhood and adolescence, reaching peak bone mass around age 30. The greater the peak bone mass, the longer one can delay serious bone loss with increasing age. Everyone should therefore consume adequate amounts of calcium and vitamin D throughout childhood, adolescence, and early adulthood. FDA has approved a health claim for the use of supplements containing calcium and vitamin D to reduce the risk of osteoporosis 78. However, not all research supports this claim. Additional research is needed before conclusions can be drawn about the use of calcium supplements to improve bone health and prevent fractures in older adults.
Osteoporosis, a disorder characterized by porous and fragile bones, is a serious public health problem for more than 10 million U.S. adults, 80% of whom are women. (Another 34 million have osteopenia, or low bone mass, which precedes osteoporosis.) Osteoporosis is most associated with fractures of the hip, vertebrae, wrist, pelvis, ribs, and other bones 79. An estimated 1.5 million fractures occur each year in the United States due to osteoporosis 80.
When calcium intake is low or ingested calcium is poorly absorbed, bone breakdown occurs as the body uses its stored calcium to maintain normal biological functions. Bone loss also occurs as part of the normal aging process, particularly in postmenopausal women due to decreased amounts of estrogen. Many factors increase the risk of developing osteoporosis, including being female, thin, inactive, or of advanced age; smoking cigarettes; drinking excessive amounts of alcohol; and having a family history of osteoporosis 81.
Various bone mineral density (BMD) tests are available. The T-score from these tests compares an individual’s bone mineral density (BMD) to an optimal bone mineral density (BMD) (that of a healthy 30-year old adult). A T-score of -1.0 or above indicates normal bone density, -1.0 to -2.5 indicates low bone mass (osteopenia), and lower than -2.5 indicates osteoporosis 81. Although osteoporosis affects individuals of all races, ethnicities, and both genders, women are at highest risk because their skeletons are smaller than those of men and because of the accelerated bone loss that accompanies menopause. Regular exercise and adequate intakes of calcium and vitamin D are critical to the development and maintenance of healthy bones throughout the life cycle. Both weight-bearing exercises (such as walking, running, and activities where one’s feet leave and hit the ground and work against gravity) and resistance exercises (such as calisthenics and that involve weights) support bone health.
Supplementation with calcium plus vitamin D has been shown to be effective in reducing fractures and falls (which can cause fractures) in institutionalized older adults 82. However, among community-dwelling older adults over age 50, the benefits of supplementation with these nutrients on fracture resistance are much less clear. For example, a longitudinal cohort study of 1,490 women aged 42 to 52 years at baseline who were followed for 10–12 years found that fracture risk was not significantly different in calcium supplement users (some of whom also took vitamin D supplements) and nonusers, even though supplement use was associated with less BMD loss throughout the study period 83.
A recent systematic review of 26 randomized controlled trials found that calcium supplements, with or without vitamin D, modestly but significantly reduced the risk of total and vertebral fractures, but not fractures of the hip or forearm 84. But the four trials with the lowest risk of bias, involving a total of 44,505 individuals, showed no effect of supplementation on risk of fracture at any site. A related meta-analysis of calcium intake on bone mineral density found that calcium supplementation produced only a small, initial, and non-progressive increase in bone mineral density that was unlikely to result in a clinically significant reduction in the risk of bone fractures 85. The U.S. Preventive Services Task Force (USPSTF) concluded with moderate certainty that daily doses of less than 1,000 mg calcium and less than 400 IU (10 mcg) vitamin D do not prevent fractures in postmenopausal women and that the evidence on larger doses of this combination is inadequate to assess the benefits in this population 86. The U.S. Preventive Services Task Force (USPSTF) also determined the evidence on the benefits of calcium supplementation alone or with vitamin D to be inadequate to assess its effect on preventing fractures in men and premenopausal women 86.
Some clinical trial evidence shows that supplements containing a combination of calcium and vitamin D can reduce the risk of fractures in older adults. For example, a meta-analysis of 8 randomized controlled trials in 30,970 adults older than 50 years found that 500 to 1,200 mg/day calcium and 400 to 800 IU/day (10 to 20 mcg/day) vitamin D supplementation for 1 to 7 years reduced the risk of total fractures by 15% and hip fractures by 30% 87. However, findings were negative in another systematic review and meta-analysis that included 14 randomized controlled trials of calcium supplementation and 13 trials comparing calcium and vitamin D supplements with hormone therapy, placebo, or no treatment in participants older than 50 years 88. The results showed that calcium supplementation alone had no effect on risk of hip fracture, and supplementation with both calcium and vitamin D had no effect on risk of hip fracture, nonvertebral fracture, vertebral fracture, or total fracture. Similarly, a systematic review of 11 randomized controlled trials in 51,419 adults aged 50 and older found that supplementation with vitamin D and calcium for 2 to 7 years had no impact on risk of total fractures or of hip fractures 89.
Some but not all clinical trials have found that calcium supplementation can improve bone health in older adults. A post-hoc analysis of data from a double-blind, randomized controlled trial of 1,000 mg elemental calcium in the form of calcium carbonate and 400 International Units (IU) (10 microgram [mcg]) vitamin D3 daily or placebo in 36,282 women aged 50–79 years enrolled in the Women’s Health Initiative (WHI) found that the supplementation did not prevent height loss after a mean follow-up period of 5.9 years 90. On average, women lost 1.28 mm/year of height in the supplementation group and 1.26 mm/year in the placebo group. However, a 2-year randomized controlled trial in 500 healthy postmenopausal women showed that daily intakes of 500 ml/day skimmed milk enriched to provide 900 mg calcium and 15 mcg (600 IU) vitamin D led to increased bone mineral density (BMD) at the femoral neck 91.
Several recent systematic reviews and meta-analyses have found that supplementation with calcium alone or a combination of calcium and vitamin D increases bone mineral density (BMD) in older adults. For example, a systematic review and meta-analysis included 15 randomized controlled trials in postmenopausal women (but did not include the two studies described in the previous paragraph) in 78,206 women, of which 37,412 were in the intervention group and 40,794 were in the control group 92. Supplementation with both calcium and vitamin D or consumption of dairy products fortified with both nutrients increased total bone mineral density (BMD) as well as bone mineral density (BMD) at the lumbar spine, arms, and femoral neck. However, in subgroup analyses, calcium had no effect on femoral neck BMD. Earlier systematic reviews and meta-analyses found a positive relationship between calcium and vitamin D supplementation and increased BMD in older males 93 and between higher calcium intakes from dietary sources or supplements in adults over 50 and higher BMD 94. However, whether these BMD increases were clinically significant is not clear.
In 1993, the U.S. Food and Drug Administration authorized a health claim related to calcium and osteoporosis for foods and supplements 95. In January 2010, this health claim was expanded to include vitamin D. Model health claims include the following: “Adequate calcium throughout life, as part of a well-balanced diet, may reduce the risk of osteoporosis” and “Adequate calcium and vitamin D as part of a healthful diet, along with physical activity, may reduce the risk of osteoporosis in later life” 95.
Cancer of the colon and rectum
The results of epidemiologic studies regarding the relationship between calcium intake and colorectal cancer risk have not always been consistent 52.
In the American Cancer Society’s Cancer Prevention Study 2 Nutrition Cohort, the diet, medical history, and lifestyle of more than 120,000 men and women were analyzed 96. Men and women who had the highest intakes of calcium through both their diet and supplement use had a modestly reduced risk of colorectal cancer compared with those who had the lowest calcium intakes. However, the benefit from calcium appeared to plateau, or level off, at an intake of approximately 1200 mg per day. When calcium from the diet was analyzed by itself, no reduction in colorectal cancer risk was found. However, the use of calcium supplements in any amount was associated with reduced risk. This association was strongest (a 31 percent reduction in risk) for people who took calcium supplements of 500 mg per day or more.
A stronger relationship between calcium intake and colorectal cancer risk was found when participants of the Nurses’ Health Study and the Health Professionals Follow-up Study were combined in an analysis that included more than 135,000 men and women 97. Individuals who had a calcium intake of more than 700 mg per day had a 35 percent to 45 percent reduced risk of cancer of the distal (lower) part of the colon than those who had a calcium intake of 500 mg or less per day. No association was found between calcium intake and risk of cancer of the proximal (middle and upper) part of the colon 97. Another large study of Finnish men showed a similar relationship between higher calcium intake and reduced risk of colorectal cancer 98. This study, however, did not evaluate proximal and distal colorectal cancers separately.
In a study that included more than 61,000 Swedish women, colorectal cancer risk was approximately 28 percent lower among individuals who had the highest calcium intakes (approximately 800–1000 mg per day) compared with those with the lowest calcium intakes (approximately 400–500 mg per day) 99. Data from this study also suggested that the benefit associated with calcium was limited to the distal colon 99. In a study that involved more than 34,000 postmenopausal Iowa women, high intakes of calcium (approximately 1280 mg per day or more) compared with lower calcium intakes (approximately 800 mg per day or less) from both the diet and supplements were associated with a 41 percent reduction in risk of rectal cancer 100. Reduced risks of rectal cancer were also observed for dietary calcium alone and supplemental calcium alone, but these associations were not statistically significant 100.
In an analysis involving more than 293,000 men and 198,000 women in the National Institutes of Health-American Association of Retired Persons (NIH-AARP) Diet and Health Study, high intakes of total calcium, dietary calcium, and supplemental calcium were associated with an approximately 20 percent lower risk of colorectal cancer among men and an approximately 30 percent lower risk of colorectal cancer among women 101.
Findings from two large randomized, placebo-controlled clinical trials, the Calcium Polyp Prevention Study 102, 103 and the European Cancer Prevention Organisation Intervention Study 104 showed that daily supplementation with 1200 to 2000 mg elemental calcium was associated with a reduced risk of recurrence of colorectal polyps known as adenomas in both men and women. Adenomas are thought to be the precursors of most colorectal cancers. In these trials, individuals who previously had one or more large adenomas removed during colonoscopy were randomly assigned to receive calcium supplementation or a placebo, and the rates of polyp recurrence and other factors were compared between the groups.
The Calcium Polyp Prevention Study involved 930 participants who were randomly assigned to receive 3 grams of calcium carbonate (1200 mg elemental calcium) daily for 4 years or a placebo and then receive follow-up colonoscopies approximately 9 months later and again 3 years after that. Compared with those in the placebo group, the individuals assigned to take calcium had about a 20 percent lower risk of adenoma recurrence 102, 103.
The European Cancer Prevention Organisation Intervention Study involved 665 participants who were randomly assigned to one of three treatment groups: 2 grams of elemental calcium daily (from calcium gluconolactate and calcium carbonate), 3 grams of fiber supplementation daily, or a placebo 104. The results showed that calcium supplementation was associated with a modest reduction in the risk of adenoma recurrence, but this finding was not statistically significant.
The results of another clinical trial conducted as part of the Women’s Health Initiative showed that supplementation with 1000 mg elemental calcium (from calcium carbonate) per day for an average duration of 7 years was not associated with a reduced risk of colorectal cancer 105. The calcium supplements in this trial also contained vitamin D (400 international units [IU]). During the trial, 128 cases of invasive colorectal cancer were diagnosed in the supplementation group and 126 cases were diagnosed in the placebo group.
In 2007, the World Cancer Research Fund/American Institute for Cancer Research published the most authoritative review of existing evidence relating food, nutrition, and physical activity to cancer risk. The report concluded that calcium probably has a protective effect against colorectal cancer 106.
Other cancers
The results of some studies suggest that a high calcium intake may decrease the risk of one or more types of cancer, whereas other studies suggest that a high calcium intake may actually increase the risk of prostate cancer.
In a randomized trial that included nearly 1,200 healthy, postmenopausal Nebraska women, individuals were randomly assigned to receive daily calcium supplementation alone (300–600 mg elemental calcium), calcium supplementation (300–600 mg elemental calcium) combined with vitamin D supplementation (1000 IU), or a placebo for 4 years 107. The incidence of all cancers combined was approximately 60 percent lower for women who took the calcium plus vitamin D supplements compared with women who took the placebo. A lower risk of all cancers combined was also observed for women who took calcium supplements alone, but this finding was not statistically significant. The numbers of individual types of cancer diagnosed during this study were too low to be able to draw reliable conclusions about cancer-specific protective effects.
The results of some but not all studies suggest that a high intake of calcium may increase the risk of prostate cancer. For example, the European Prospective Investigation into Cancer and Nutrition analyzed the intakes of animal foods (meat, poultry, fish, dairy products, etc.), protein, and calcium in relation to prostate cancer risk among more than 142,000 men and found that a high intake of protein or calcium from dairy products was associated with an increased risk of prostate cancer 108. Calcium from nondairy sources, however, was not associated with increased risk 108. In addition, a prospective analysis of dairy product and calcium intakes among more than 29,000 men participating in the National Cancer Institute’s Prostate, Lung, Colorectal, and Ovarian Cancer Screening Trial showed increased risks for prostate cancer associated with high dietary intakes of calcium and dairy products, particularly low-fat dairy products 109. Calcium from supplements was not associated with increased prostate cancer risk 109. In contrast, results from the NIH-AARP Diet and Health Study showed no increased risk of prostate cancer associated with total calcium, dietary calcium, or supplemental calcium intakes 102, 110.
Other studies have suggested that intakes of low-fat milk, lactose, and calcium from dairy products may reduce the risk of ovarian cancer, but this risk reduction has not been found in all studies 102, 111.
A meta-analysis included 15 epidemiological studies of calcium intake and ovarian cancer risk in 493,415 women who developed 7,453 cases of ovarian cancer 112. In this meta-analysis, ovarian cancer risk was 20% lower in participants in the highest category of dietary calcium intakes (more than 820–1,500 mg/day, depending on the study) than the lowest intake category (less than 362–800 mg/day, depending on the study) 112. However, the difference in risk was not statistically significant when both dietary and supplemental calcium intakes were considered.
An analysis from the Nurses’ Health Study that included more than 3,000 women found that higher calcium intakes (more than 800 mg per day) from dairy products—particularly low-fat or nonfat milk, yogurt, and cheese—compared with lower calcium intakes (200 mg or less per day) from dairy products was associated with a reduced risk of breast cancer among premenopausal but not postmenopausal women 113. Calcium from nondairy sources was not associated with a reduction in risk 113. Another analysis that involved more than 30,000 women in the Women’s Health Study found a reduced risk of breast cancer associated with higher (1366 mg per day or more) versus lower (less than 617 mg per day) total intakes of calcium among premenopausal but not postmenopausal women 114. In this study, higher versus lower calcium intakes from the diet, from supplements, and from total dairy products were not associated with reduced risk 114.
For breast cancer, observational studies have had mixed findings on whether higher calcium intakes are associated with a lower risk. A meta-analysis of 11 prospective cohort studies in 872,895 women who developed 26,606 cases of breast cancer over 7 to 25 years found that women with the highest calcium intakes had an 8% lower risk of breast cancer 115. However, the Women’s Health Initiative found similar incidence rates of invasive breast cancer in the supplement and placebo groups 116.
In summary, additional well-designed randomized trials are needed to determine whether dietary or supplemental calcium intakes increase, decrease, or have no effect on risk of cancer in general or of specific types of cancer, or on cancer mortality.
Cardiovascular disease
Calcium binds fatty acids, so it can reduce lipid absorption and might therefore lower cardiovascular disease risk 4, 3. However, the findings from research on the role of dietary calcium and calcium supplements in reducing cardiovascular disease have been mixed, and some evidence indicates that calcium supplements might even increase cardiovascular disease risk.
Several large observational studies have shown an association between lower calcium intakes and higher risk of hypertension, stroke, and atherosclerosis. For example, an analysis of 1999–2010 National Health and Nutrition Examination Survey (NHANES) data from 14,408 adults (mean age 54 years) with obesity found that calcium intakes were 10% lower in adults with obesity and hypertension than in those without hypertension 117. This association was strongest in women, adults aged 20–44 years, those who did not have diabetes, and, especially, women aged 20–44 years. A prospective cohort study that followed 41,514 adults aged 40 to 69 years in Australia for 13 years found a 25% lower rate of stroke in adults in the highest calcium intake quartile (mean of 1,076 mg/day) than in the lowest quartile (mean of 641 mg/day) 118. However, the study found no association between calcium intakes and risk of cardiovascular disease mortality or myocardial infarction. The risk of atherosclerosis over 10 years in a study of 5,448 adults aged 45–84 years was 27% lower in the highest quintile of calcium intake (mean of 2,157 mg/day) than in the lowest quintile (mean of 313 mg/day) 119. Furthermore, a systematic review and meta-analysis that included 27 observational studies found no consistent dose-response relationships between total, dietary, or supplemental calcium intakes and cardiovascular disease mortality 120. Evidence on dose-response relationships between calcium intakes and risk of stroke or stroke mortality was inconsistent.
A diet containing more calcium than the typical U.S. diet because of added low-fat or non-fat dairy products lowered systolic blood pressure by an average of 5.5 mmHg and diastolic blood pressure by 3.0 mmHg 121. However, this Dietary Approaches to Stop Hypertension (DASH) diet also increases intakes of other nutrients, such as potassium and magnesium, that are associated with reductions in blood pressure, so any independent contribution of calcium cannot be determined.
Some clinical trials have shown that calcium supplements are associated with decreased hypertension risk or decreased cholesterol levels, but others have had more mixed findings. A Cochrane review of 16 trials in 3,048 adults with a median follow-up period of 3.5 months found that calcium supplementation (typically 1,000 to 2,000 mg/day) reduced systolic blood pressure by 1.43 mmHg and diastolic blood pressure by 0.98 mmHg 122. Effects were greatest in adults younger than 35 years and with doses higher than 1,500 mg/day calcium. A meta-analysis of 23 randomized controlled trials in 4,071 participants showed that calcium supplements providing 162 to 2,000 mg/day (combined with vitamin D in 10 randomized controlled trials) for 2 weeks to 5 years was associated with low-density lipoprotein cholesterol levels that were 4.6 mg/dL lower and high-density lipoprotein cholesterol levels that were 1.9 mg/dL higher 123.
Findings were mixed in two analyses of data from the WHI. One analysis of results from 35,983 women aged 50 to 79 years randomly assigned to 1,000 mg/day calcium and 400 IU (10 mcg)/day vitamin D supplements or placebo for 10 years found no reduction in risk of heart failure 124. However, the calcium and vitamin D supplements were associated with 5% lower heart failure risk in participants who had no preexisting heart failure risk factors (coronary heart disease, diabetes, or hypertension). In another secondary analysis of data on 16,801 WHI participants, the supplements had no association with atrial fibrillation risk 125. Similarly, an evidence report and systematic review conducted for the USPSTF that included 11 randomized controlled trials of vitamin D, calcium, or both for 2 to 7 years in 51,419 adults aged 50 years and older found that supplementation with vitamin D alone or combined with calcium had no effect on cardiovascular disease incidence 89.
In contrast, several prospective cohort studies and randomized controlled trials have shown that calcium supplements increase the risk of cardiovascular disease. A meta-analysis of 14 randomized controlled trials (including 1 study that administered supplements providing 20 mcg [800 IU] vitamin D per day) in 28,935 healthy postmenopausal women found that calcium supplements providing 500 to 2,000 mg/day calcium for 1 to 7 years increased cardiovascular disease risk by 15% and coronary heart disease risk by 16% 126. In addition, when 132,823 adults (mean age 63 years) were followed for an average of 17.5 years, the risk of cardiovascular disease mortality was 22% higher in men with calcium supplement intakes of 1,000 mg/day or more than in those not taking calcium supplements 127. However, in women, the cardiovascular disease mortality rate was 16% lower with supplemental calcium intakes of 1,000 mg/day than with no supplemental calcium intakes.
Other studies have found no association between calcium supplements and cardiovascular disease risk or cardiovascular disease outcomes. After 24 years of follow-up of 74,245 women aged 30 to 55 years at baseline who participated in the Nurses’ Health Study, women taking more than 1,000 mg/day calcium supplements did not have a higher risk of cardiovascular disease than those taking no supplemental calcium 128.
An expert panel convened by the National Osteoporosis Foundation and American Society for Preventive Cardiology determined, on the basis of moderate-quality evidence, that calcium intakes with or without vitamin D from foods or supplements neither increase nor decrease the risk of cardiovascular disease or cardiovascular disease mortality 129. The societies therefore concluded that calcium intakes that do not exceed the Tolerable Upper Intake Level (maximum daily intake unlikely to cause adverse health effects) are safe “from a cardiovascular standpoint.”
High blood pressure
Some studies have found that getting recommended intakes of calcium can reduce the risk of developing high blood pressure (hypertension). Calcium’s effects on blood pressure might depend upon the population being studied. In hypertensive subjects, calcium supplementation appears to lower systolic blood pressure by 2–4 mmHg, whereas in normotensive subjects, calcium appears to have no significant effect on systolic or diastolic blood pressure.
One large study in particular found that eating a diet high in fat-free and low-fat dairy products, vegetables, and fruits lowered blood pressure. In the DASH (Dietary Approaches to Stop Hypertension) study, 549 people were randomized to one of three diets for eight weeks: (1) a control diet that was low in fruit, vegetables, and dairy products; (2) a diet rich in fruit (~5 servings/day) and vegetables (~3 servings/day); and (3) a combination diet rich in fruit and vegetables, as well as low-fat dairy products (~3 servings/day) 130. The combination diet represented an increase of about 800 mg of calcium/day over the control and fruit/vegetable-rich diets for a total of about 1,200 mg of calcium/day. Overall, the reduction in systolic blood pressure was greater with the combination diet than with the fruit/vegetable diet or the control diet. Among participants diagnosed with hypertension, the combination diet reduced systolic blood pressure by 11.4 mm Hg and diastolic pressure by 5.5 mm Hg more than the control diet, while the reduction for the fruit/vegetable diet was 7.2 mm Hg for systolic and 2.8 mm Hg for diastolic blood pressure compared to the control diet 131. This research suggested that calcium intake at the recommended level (1,000-1,200 mg/day) may be helpful in preventing and treating moderate hypertension 132.
A meta-analysis of 23 large observational studies conducted in different populations worldwide found a reduction in systolic blood pressure of 0.34 millimeters of mercury (mm Hg) per 100 mg of calcium consumed daily and a reduction in diastolic blood pressure of 0.15 mm Hg per 100 mg calcium 133.
Several clinical trials have demonstrated a relationship between increased calcium intakes and both lower blood pressure and risk of hypertension 134, 135, 136, although the reductions are inconsistent. In the Women’s Health Study, calcium intake was inversely associated with risk of hypertension in middle-aged and older women 137. However, other studies have found no association between calcium intake and incidence of hypertension 138. The authors of a systematic review of the effects of calcium supplements for hypertension found any link to be weak at best, largely due to the poor quality of most studies and differences in methodologies 139.
Two large systematic reviews and meta-analyses of randomized controlled trials have examined the effect of calcium supplementation on blood pressure compared to placebo in either normal blood pressure or hypertensive individuals 134, 135. Neither of the analyses reported any significant effect of supplemental calcium on blood pressure in normotensive subjects. A small but significant reduction in systolic blood pressure, but not in diastolic blood pressure, was reported in participants with hypertension. Of note, calcium supplementation in these randomized controlled trials ranged from 400 to 2,200 mg/day, with 1,000 to 1,500 mg/day being the more common dosages. A more recent meta-analysis of 13 randomized controlled studies in 485 individuals with elevated blood pressure found a significant reduction of 2.5 mm Hg in systolic blood pressure but no change in diastolic blood pressure with calcium supplementation 139. The modest effect of calcium on blood pressure needs to be confirmed in larger, high-quality, well-controlled trials before any recommendation is made regarding the management of hypertension. Finally, a review of the literature on the effect of high-calcium intake (dietary and supplemental) in postmenopausal women found either no reduction or mild and transient reductions in blood pressure 140.
Other observational and experimental studies suggest that individuals who eat a vegetarian diet high in minerals (such as calcium, magnesium, and potassium) and fiber and low in fat tend to have lower blood pressure 141, 142, 143, 144, 145. The Dietary Approaches to Stop Hypertension (DASH) study was conducted to test the effects of three different eating patterns on blood pressure: a control “typical” American diet; one high in fruits and vegetables; and a third diet high in fruits, vegetables, and low-fat dairy products. The diet containing dairy products resulted in the greatest decrease in blood pressure 130, although the contribution of calcium to this effect was not evaluated. Additional information and sample DASH menu plans are available on the National Heart, Lung, and Blood Institute Web site (https://www.nhlbi.nih.gov/education/dash-eating-plan).
Preeclampsia
Preeclampsia is a serious medical condition in which a pregnant woman develops high blood pressure and kidney problems that cause protein to spill into the urine (proteinuria), usually after 20 weeks of pregnancy, often in the third trimester 146, 147. Preeclampsia is a leading cause of sickness and death in pregnant women and their newborn babies, affecting about 5–8% of pregnancies in the United States and up to 14% of pregnancies worldwide 146, 148. Calcium supplementation during pregnancy might reduce the risk of preeclampsia, but the benefits might apply only to women with inadequate calcium intakes, and much of this evidence comes from studies with methodological weaknesses 149, 150. For women who get less than about 900 mg of calcium a day, taking calcium supplements during pregnancy (1,000 mg a day or more) reduces the risk of preeclampsia. But most women in the United States who become pregnant get enough calcium from their diets.
A Cochrane review included 27 randomized controlled trials of calcium supplements during pregnancy in 18,064 women to prevent hypertensive disorders and related problems 151. In the 13 studies—none of which administered vitamin D supplements—that evaluated high doses (at least 1,000 mg/day calcium) in 15,730 women, supplementation reduced the risk of high blood pressure by 35% and, in women with low dietary calcium intakes (less than 1,000 mg/day; 10 trials in 10,678 women), the risk of preeclampsia by 55% 151. However, the quality of this evidence was low. In 12 trials in 2,334 women, doses of less than 1,000 mg/day (usually 500 mg/day) reduced the risk of high blood pressure by 47% and of preeclampsia by 62% 151. However, most of these studies recruited women at high risk of preeclampsia and had a high risk of bias. An earlier systematic review and meta-analysis of 10 randomized controlled trials in 24,787 women also found that calcium supplementation (1,500 to 2,000 mg/day) reduced the risk of preeclampsia by 38% and, in women at increased risk of any hypertensive disorder of pregnancy, by 58% 152. But when the analysis was restricted to trials with 4,000 or more women, the effect was no longer statistically significant. An randomized controlled trial in 1,355 women in Argentina, South Africa, and Zimbabwe also found that 500 mg/day calcium supplementation starting before conception made no difference in the risk of preeclampsia 153, 154.
Several professional organizations recommend calcium supplements during pregnancy for women with low calcium intakes to reduce the risk of preeclampsia. For example, the American College of Obstetrics and Gynecology states that daily supplementation with 1,500–2,000 mg calcium might reduce the severity of preeclampsia in pregnant women who have calcium intakes of less than 600 mg/day 149. The World Health Organization (WHO) recommends 1,500–2,000 mg/day calcium for pregnant women with low dietary calcium intakes to reduce preeclampsia risk 155. The Canadian Hypertensive Disorders of Pregnancy Working Group 156, the International Society for the Study of Hypertension in Pregnancy 157 and the Society of Obstetric Medicine of Australia and New Zealand 158 have similar recommendations.
Because excessive calcium supplementation may be harmful, further research is required to verify whether calcium supplementation above the current Food and Nutrition Board recommendation (1,000 mg/day for pregnant women, ages 19-50 years) would provide greater benefits to women at high risk of preeclampsia. Finally, the lack of effect of supplemental calcium on proteinuria (reported in two trials only) suggested that calcium supplementation from mid-pregnancy might be too late to oppose the genesis of preeclampsia 159, 160. A randomized, double-blind, placebo-controlled study — the WHO Calcium and Pre-eclampsia (CAP) trial — is ongoing to evaluate the effect of calcium supplementation with 500 mg/day, starting before pregnancy and until the 20th week of pregnancy, on the risk of preeclampsia in high-risk women 153, 161.
Kidney stones
Most kidney stones are composed of calcium oxalate. Some studies have found that higher intakes of calcium from dietary supplements are linked to a greater risk of kidney stones, especially among older adults 162, 163. But calcium from foods does not appear to cause kidney stones. Current data suggest that diets providing adequate dietary calcium and low levels of animal protein, oxalate, and sodium may benefit the prevention of kidney stone recurrence in subjects with idiopathic hypercalciuria (abnormally high level of calcium in the urine) 164, 165, 166, 167.
Subjects with an abnormally high level of calcium in the urine (hypercalciuria) are at higher risk of developing kidney stones a process called nephrolithiasis 168. High urinary oxalate level is another risk factor for calcium oxalate stone formation. Most subjects with a history of kidney stones and/or idiopathic hypercalciuria have increased intestinal calcium absorption 169. Although it was initially recommended to limit dietary calcium intake in these patients, a number of prospective cohort studies have reported associations between lower total dietary calcium intake and increased risk of incident kidney stones 170, 171, 172. The prospective analyses of three large cohorts, including a total of 30,762 men and 195,865 women followed for a combined 56 years, have indicated that the risk of kidney stones was significantly lower in individuals in the highest versus lowest quintile of dietary calcium intake from dairy or nondairy sources 172. Additionally, a five-year randomized intervention study that enrolled 120 men with idiopathic hypercalciuria (mean age, 45 years) reported that those assigned to a low-calcium diet (approximately 400 mg/day) had a 51% higher risk of kidney stone recurrence compared to those on a normal-to-high calcium (1,200 mg/day), low animal-protein, low-salt diet 173.
Mechanisms by which increased dietary calcium might reduce the risk of incident kidney stones are not fully understood. An inverse relationship was reported between total calcium intake and intestinal calcium absorption in the recent cross-sectional analysis of a cohort of 5,452 postmenopausal women 169. Moreover, women with higher supplemental calcium intake and lower calcium absorption were less likely to report a history of kidney stones 169. Adequate intake of calcium with food may reduce the absorption of dietary oxalate and lower urinary oxalate through formation of the insoluble calcium oxalate salt 174, 175. A recent small intervention study in 10 non-stone-forming young adults observed that the ingestion of large amounts of oxalate did not increase the risk of calcium oxalate stone occurrence in the presence of recommended level of dietary calcium 176.
However, a randomized, double-blind, placebo-controlled trial (Women’s Health Initiative) in 36,282 postmenopausal women reported that a combination of supplemental calcium (1,000 mg/day) and vitamin D (400 IU/day) was associated with a significantly increased incidence of self-reported kidney stones during a seven-year treatment period. More controlled trials may be necessary to determine whether supplemental calcium affects kidney stone risk 177. The Nurses’ Health Study also showed a positive association between supplemental calcium intake and kidney stone formation 158. High intakes of dietary calcium, on the other hand, do not appear to cause kidney stones and may actually protect against developing them 14, 178, 171, 179, 180. However, a systematic review of observational studies and randomized controlled trials that primarily reported on bone-related outcomes failed to find an effect of calcium supplementation on stone incidence 181. A potential kidney stone risk associated with calcium supplementation may likely depend on whether supplemental calcium is co-ingested with oxalate-containing foods or consumed separately. For most individuals, other risk factors for kidney stones, such as high intakes of oxalates from food and low intakes of fluid, probably play a bigger role than calcium intake 173. Further research is needed to verify whether osteoporosis treatment drugs (e.g., biphosphonates) rather than calcium supplements might influence the risk of stone occurrence 182.
Weight loss
Although several studies have shown that getting more calcium helps lower body weight or reduce weight gain over time, most studies have found that calcium—from foods or dietary supplements—has little if any effect on body weight and amount of body fat 183, 184, 185, 186, 187.
An observational study found an association between higher calcium intakes and lower prevalence of overweight or obesity in 6,696 children (51% male, mean age 6 years) in eight European countries, of whom 2,744 were re-examined 6 years later 188. The prevalence of overweight or obesity at 6-year follow-up was lower in boys (16%) and girls (18%) in the highest tertile of calcium intake (664 mg/1,000 kcal for boys and 667 mg/1,000 kcal for girls) than in boys (26%) and girls (25%) in the lowest tertile (249 mg/1,000 kcal for both boys and girls). In contrast, a longitudinal study in 2,159 participants in Portugal evaluated at ages 13 and 21 years found no association between total dietary and supplemental calcium intake at age 13 and body mass index (BMI) at age 21 after the analysis was adjusted for energy intake 189. The study also found no associations between consumption of dairy foods (milk, yogurt, and cheese) at age 13 and BMI at age 21.
Clinical trials and meta-analyses of randomized controlled trials assessing the impact of calcium supplements or increased intakes of calcium from dairy products on prevention of weight gain or promotion of fat loss or weight loss have had mixed results 184, 185, 186, 187. For example, postmenopausal women who took 1,000 mg calcium and 400 IU (10 mcg) vitamin D daily for 3 years in the Women’s Health Initiative whose daily intakes were less than 1,200 mg calcium at baseline were 11% less likely to gain 1 kg of weight or more than those who took placebo during this period 185. A systematic review and meta-analysis of 41 randomized controlled trials that examined the effect of dairy foods or calcium supplements (at least 300 mg/day) in 4,802 adults found that higher calcium intakes from dairy foods had no impact on body weight or body fat, although they did reduce body fat when combined with an energy-restricted diet 186. In addition, calcium supplements had no effect on body weight or body fat.
To date, there is no consensus regarding the effect of calcium on body weight changes. A meta-analysis of 29 randomized controlled trials in 2,441 participants (median age, 41.4 years) found that calcium supplementation was only associated with body weight and fat loss in short-term studies (<1 year) that used energy-restricted diets 190. Another meta-analysis of 41 randomized controlled trials (4,802 participants) found little-to-no effect of increased calcium intake from supplements or dairy foods for >12 weeks on body weight and body composition 186. Finally, a meta-analysis of 33 randomized controlled trials (4,733 participants) found no overall effect of calcium supplementation (from food or supplements) for >12 weeks on body weight changes. Yet, further subgroup analyses showed weight reductions in children and adolescents (mean, -0.26 kg), in adults (mean, -0.91 kg), and in those with normal BMI (mean, -0.53 kg). Supplemental calcium did not lead to weight loss in postmenopausal women or in overweight/obese individuals 184. At present, additional research is warranted to examine the effect of calcium intake on fat metabolism, as well as its potential benefits in the management of body weight with or without caloric restriction 191.
Lead toxicity
Children who are chronically exposed to lead, even in small amounts, are more likely to develop learning disabilities, behavioral problems, and to have low IQs. Deficits in growth and neurological development may occur in the infants of women exposed to lead during pregnancy and lactation. In adults, lead toxicity may result in kidney damage and high blood pressure. Although the use of lead in paint products, gasoline, and food cans has been discontinued in the US, lead toxicity continues to be a significant health problem, especially in children living in urban areas 192.
In 2012, the US Centers for Disease Control and Prevention set the reference value for blood lead concentration at 5 micrograms per deciliter (mg/dL) to identify children at risk 193. Yet, there is no known blood lead concentration below which children are 100% safe. An early study of over 300 children in an urban neighborhood found that 49% of children ages 1 to 8 years had blood lead levels above the threshold of 10 mg/dL, indicating excessive lead exposure. In this study, only 59% of children ages 1 to 3 years and 41% of children ages 4 to 8 years met the recommended levels for calcium intakes 194.
Adequate calcium intake could be protective against lead toxicity in at least two ways. Increased dietary intake of calcium is known to decrease the gastrointestinal absorption of lead. Once lead enters the body it tends to accumulate in the skeleton, where it may remain for more than 20 years. Adequate calcium intake also prevents lead mobilization from the skeleton during bone demineralization. A study of circulating concentrations of lead during pregnancy found that women with inadequate calcium intake during the second half of pregnancy were more likely to have elevated blood lead levels, probably because of increased bone demineralization, leading to the release of accumulated lead into the blood 195. Lead in the blood of a pregnant woman is readily transported across the placenta resulting in fetal lead exposure at a time when the developing nervous system is highly vulnerable. In a randomized, double-blind, placebo-controlled study in 670 pregnant women (≤14 weeks’ gestation) with average dietary calcium intakes of 900 mg/day, daily supplementation of 1,200 mg of calcium throughout the pregnancy period resulted in 8%-14% reductions in maternal blood lead concentrations 196. Similar reductions in maternal lead concentrations in the blood and breast milk of lactating mothers supplemented with calcium were reported in earlier trials 197, 198. In postmenopausal women, factors known to decrease bone demineralization, including estrogen replacement therapy and physical activity, have been inversely associated with blood lead levels 199.
Premenstrual Syndrome (PMS)
Premenstrual Syndrome (PMS) refers to a cluster of symptoms, including but not limited to fatigue, irritability, moodiness/depression, fluid retention, and breast tenderness, that begins sometime after ovulation (mid-cycle) and subsides with the onset of menstruation (the monthly period) (100). A severe form of premenstrual syndrome (PMS) called premenstrual dysphoric disorder (PMDD) has been described in 3%-8% of women of childbearing age. Premenstrual dysphoric disorder (PMDD) interferes with normal functioning, affecting daily activities and relationships 200.
Low dietary calcium intakes have been linked to PMS in early reports, and supplemental calcium has been shown to decrease symptom severity 201. A nested case-control study within the Nurses’ Health Study 2 (NHS 2) found that women in the highest quintile of dietary (but not supplemental) calcium intake (median of 1,283 mg/day) had a 30% lower risk of developing PMS compared to those in the lowest quintile (median of 529 mg/day) 202. Similarly, women in the highest versus lowest quintile of skim or low-fat milk intake (≥4 servings/day vs. ≤1 serving/week) had a 46% lower risk of PMS 202. In a randomized, double-blind, placebo-controlled clinical trial of 466 women with moderate-to-severe premenstrual symptoms, supplemental calcium (1,200 mg/day) for three menstrual cycles was associated with a 48% reduction in total symptom scores, compared to a 30% reduction observed in the placebo group 203. Similar positive effects were reported in earlier double-blind, placebo-controlled, cross-over trials that administered 1,000 mg of calcium daily 204, 205. Recent small randomized controlled trials also reported that supplemental calcium (400-500 mg/day) for three weeks to three months reduced severity and/or frequency of symptoms in women with mild-to-moderate PMS 206, 207, 208, 209. Currently available data indicate that daily calcium intakes from food and/or supplements may have therapeutic benefits in women diagnosed with PMS or premenstrual dysphoric disorder (PMDD) 210, 211.
How much calcium do I need per day?
Intake recommendations for calcium and other nutrients are provided in the Dietary Reference Intakes (DRIs) developed by the Food and Nutrition Board at the Institute of Medicine of the National Academies 14. Dietary Reference Intake (DRI) is the general term for a set of reference values used for planning and assessing the nutrient intakes of healthy people. These values, which vary by age and gender, include:
- Recommended Dietary Allowance (RDA): average daily level of intake sufficient to meet the nutrient requirements of nearly all (97%–98%) healthy individuals.
- Adequate Intake (AI): established when evidence is insufficient to develop an RDA and is set at a level assumed to ensure nutritional adequacy.
- Estimated Average Requirement (EAR): average daily level of intake estimated to meet the requirements of 50% of healthy individuals. It is usually used to assess the adequacy of nutrient intakes in populations but not individuals.
- Tolerable Upper Intake Level (UL): maximum daily intake unlikely to cause adverse health effects.
The Food and Nutrition Board established Recommended Dietary Allowances (RDAs) for the amounts of calcium required for bone health and to maintain adequate rates of calcium retention in healthy people. They are listed in Table 1 in milligrams (mg) per day.
Many people in the United States don’t get recommended amounts of calcium from the foods they eat especially:
- Children and teens aged 4 to 18 years
- People who are Black or Asian
- Adults aged 50 years and older living in poverty
When total intakes of calcium from both food and supplements are considered, many people—particularly adolescent girls—still fall short of getting enough calcium, while some older women likely get more than the upper limit.
Certain groups of people are more likely than others to have trouble getting enough calcium, including:
- Postmenopausal women. The body absorbs and retains less calcium after menopause. Over time, this can lead to fragile bones.
- People who don’t drink milk or eat other dairy products. Dairy products are rich sources of calcium, but people with lactose intolerance, people with milk allergies, and vegans (people who don’t consume any animal products) must find other sources of calcium. Options include lactose-free or reduced-lactose dairy products; canned fish with bones; certain vegetables, such as kale, broccoli, and Chinese cabbage; calcium-fortified fruit juices and milk substitutes such as soy and almond beverages, tofu, and ready-to-eat cereals; and dietary supplements that contain calcium.
People should get most of their nutrients from food, advises the federal government’s Dietary Guidelines for Americans 212. Foods contain vitamins, minerals, dietary fiber and other substances that benefit health. In some cases, fortified foods and dietary supplements may provide nutrients that otherwise may be consumed in less-than-recommended amounts. For more information about building a healthy diet, refer to the Dietary Guidelines for Americans (https://www.dietaryguidelines.gov) and the U.S. Department of Agriculture’s MyPlate (https://www.myplate.gov).
In its food guidance system, the U.S. Department of Agriculture’s MyPlate (https://www.myplate.gov) recommends that persons aged 9 years and older eat 3 cups of foods from the milk group per day 213. A cup is equal to 1 cup (8 ounces) of milk, 1 cup of yogurt, 1.5 ounces of natural cheese (such as Cheddar), or 2 ounces of processed cheese (such as American).
Table 1. Recommended Dietary Allowances (RDAs) for Calcium
| Life Stage | Recommended Amount |
|---|---|
| Birth to 6 months | 200 mg |
| Infants 7–12 months | 260 mg |
| Children 1–3 years | 700 mg |
| Children 4–8 years | 1,000 mg |
| Children 9–13 years | 1,300 mg |
| Teens 14–18 years | 1,300 mg |
| Adults 19–50 years | 1,000 mg |
| Adult men 51–70 years | 1,000 mg |
| Adult women 51–70 years | 1,200 mg |
| Adults 71 years and older | 1,200 mg |
| Pregnant and breastfeeding teens | 1,300 mg |
| Pregnant and breastfeeding adults | 1,000 mg |
Footnote: *Adequate Intake (intake at this level is assumed to ensure nutritional adequacy; established when evidence is insufficient to develop an Recommended Dietary Allowance (RDA).
[Source 9 ]What happens if I don’t get enough calcium?
Getting too little calcium can cause several conditions, including the following:
- Osteoporosis, which causes weak, fragile bones and increases the risk of falling
- Rickets, a disease in children that causes soft, weak bones
- Osteomalacia, which causes soft bones in children and adults
What foods provide calcium?
Calcium is found in many foods. It is important to get plenty of calcium in the foods you eat. You can get recommended amounts of calcium by eating a variety of foods, including the following 9:
- Dairy products such as milk, cheese, and yogurt are the main food sources of calcium for most people in the United States.
- Fish with soft bones that you eat, such as canned sardines and salmon.
- Certain vegetables, such as kale, broccoli, and Chinese cabbage (bok choi) also contain calcium.
- Calcium is added to some breakfast cereals and beverages, including many fruit juices and milk substitutes such as soy and almond beverages, as well as some brands of tofu and ready-to-eat cereals. To find out whether these foods have calcium added, check the product labels.
- Most grains (such as breads, pastas, and unfortified cereals) do not have high amounts of calcium. However, because people eat them often, what they contribute adds up.
The U.S. Department of Agriculture’s (USDA’s) FoodData Central website (https://fdc.nal.usda.gov) lists the nutrient content of many foods with Calcium arranged by nutrient content (https://ods.od.nih.gov/pubs/usdandb/Calcium-Content.pdf) and by food name (https://ods.od.nih.gov/pubs/usdandb/Calcium-Food.pdf).
Milk, yogurt, and cheese are rich natural sources of calcium and are the major food contributors of this nutrient to people in the United States 14. Nondairy sources include vegetables, such as Chinese cabbage, kale, and broccoli. Spinach provides calcium, but its bioavailability is poor. Most grains do not have high amounts of calcium unless they are fortified; however, they contribute calcium to the diet because they contain small amounts of calcium and people consume them frequently. Foods fortified with calcium include many fruit juices and drinks, tofu, and cereals. Selected food sources of calcium are listed in Table 2.
Calcium absorption varies by type of food. The absorption of calcium from dairy products and fortified foods is about 30% 3. Certain compounds in plants (e.g., oxalic acid, phytic acid) can decrease calcium absorption by forming indigestible salts with calcium, decreasing its absorption 12. As a result, absorption of calcium is only 5% for spinach, whereas it is much higher, at 27%, for milk 12. In addition to spinach, foods with high levels of oxalic acid include collard greens, sweet potatoes, rhubarb, and beans 3. The bioavailability of calcium from other plants that do not contain these compounds—including broccoli, kale, and cabbage—is similar to that of milk, although the amount of calcium per serving is much lower 12. When people eat many different types of foods, these interactions with oxalic or phytic acid probably have little or no nutritional consequence. Net absorption of dietary calcium is also reduced to a small extent by intakes of caffeine and phosphorus and to a greater extent by low status of vitamin D 214, 33, 215.
Table 2. Calcium Rich Foods
| Food* | Milligrams (mg) per serving | Percent DV* |
|---|---|---|
| Yogurt, plain, low fat, 8 ounces | 415 | 32 |
| Orange juice, calcium fortified, 1 cup | 349 | 27 |
| Yogurt, fruit, low fat, 8 ounces | 344 | 27 |
| Mozzarella, part skim, 1.5 ounces | 333 | 26 |
| Sardines, canned in oil, with bones, 3 ounces | 325 | 25 |
| Milk, nonfat, 1 cup** | 299 | 23 |
| Soymilk, calcium fortified, 1 cup | 299 | 23 |
| Milk, whole (3.25% milk fat), 1 cup** | 276 | 21 |
| Tofu, firm, made with calcium sulfate, ½ cup*** | 253 | 19 |
| Salmon, pink, canned, solids with bones, 3 ounces | 181 | 14 |
| Cottage cheese, 1% milk fat, 1 cup | 138 | 11 |
| Tofu, soft, made with calcium sulfate, ½ cup*** | 138 | 11 |
| Soybeans, cooked, ½ cup | 131 | 10 |
| Breakfast cereals, fortified with 10% of the DV for calcium, 1 serving | 130 | 10 |
| Spinach, boiled, drained, ½ cup | 123 | 9 |
| Frozen yogurt, vanilla, soft serve, ½ cup | 103 | 8 |
| Turnip greens, fresh, boiled, ½ cup | 99 | 8 |
| Kale, fresh, cooked, 1 cup | 94 | 7 |
| Chia seeds, 1 tablespoon | 76 | 6 |
| Chinese cabbage (bok choi), raw, shredded, 1 cup | 74 | 6 |
| Beans, pinto, canned, drained, ½ cup | 54 | 4 |
| Tortilla, corn, one, 6” diameter | 46 | 4 |
| Sour cream, reduced fat, 2 tablespoons | 31 | 2 |
| Bread, whole-wheat, 1 slice | 30 | 2 |
| Kale, raw, chopped, 1 cup | 24 | 2 |
| Broccoli, raw, ½ cup | 21 | 2 |
| Apple, golden delicious, with skin, 1 medium | 10 | 0 |
Footnote: * DV = Daily Value. The U.S. Food and Drug Administration (FDA) developed DVs to help consumers compare the nutrient contents of foods and dietary supplements within the context of a total diet. The Daily Value (DV) for calcium is 1,300 mg for adults and children age 4 years and older 216. FDA requires food labels to list calcium content. Foods providing 20% or more of the DV are considered to be high sources of a nutrient, but foods providing lower percentages of the DV also contribute to a healthful diet.
** Calcium content varies slightly by fat content; the more fat in the food, the less calcium it contains.
*** Calcium content is for tofu processed with a calcium salt. Tofu processed with other salts does not provide significant amounts of calcium.
Am I getting enough calcium?
A substantial proportion of people in the United States consume less than recommended amounts of calcium. An analysis of 2007–2010 data from the National Health and Nutrition Examination Survey (NHANES) found that 49% of children aged 4–18 years and 39% of all individuals aged 4 and older consume less than the Estimated Average Requirement (average daily level of intake estimated to meet the requirements of 50% of healthy individuals; usually used to assess the nutrient intakes of groups of people and to plan nutritionally adequate diets for them; can also be used to assess the nutrient intakes of individuals) for calcium from foods and supplements 218.
Average daily intakes of calcium from foods and beverages are 1,083 mg for men aged 20 and older and 842 mg for women 219. For children aged 2–19, mean daily intakes of calcium from foods and beverages range from 965 to 1,015 mg [18]. Approximately 22% of men, 32% of women, and 4 to 8% of children take a dietary supplement containing calcium 219. Average daily calcium intakes from both foods and supplements are 1,156 mg for men, 1,009 mg for women, and 968 to 1,020 mg for children 219.
According to 2009–2012 NHANES data, rates of calcium inadequacy (intakes below the EAR) are higher among non-Hispanic Blacks and non-Hispanic Asians (47–48%) than among Hispanics (30%) and non-Hispanic Whites (24%) in the United States 220. Poverty is also associated with a higher risk of inadequacy. NHANES data from 2007 to 2014 show that the risk of inadequate calcium intakes (less than 800 to 1,100 mg) is 11.6% higher among adults aged 50 and older in households earning less than $20,000 per year than other households 221.
What happens if I don’t get enough calcium?
Insufficient intakes of calcium do not produce obvious symptoms in the short term because the body maintains calcium levels in the blood by taking it from bone. Over the long term, intakes of calcium below recommended levels have health consequences, such as causing low bone mass (osteopenia) and increasing the risks of osteoporosis and bone fractures.
Symptoms of serious calcium deficiency include numbness and tingling in the fingers, convulsions, and abnormal heart rhythms that can lead to death if not corrected. These symptoms occur almost always in people with serious health problems or who are undergoing certain medical treatments.
Calcium deficiency
Inadequate intakes of dietary calcium from food and supplements produce no obvious symptoms in the short term because circulating blood levels of calcium are tightly regulated (see Figure 1 above). However, over a longer term, dietary calcium deficiency with declining dietary calcium intake can lead on to secondary hyperparathyroidism (a disorder in which the parathyroid glands in your neck produce too much parathyroid hormone [PTH]) 222. Secondary hyperparathyroidism is an increased secretion of parathyroid hormone (PTH) due to parathyroid hyperplasia caused by triggers such as hypocalcemia, hyperphosphatemia, or decreased active vitamin D 223. The increased parathyroid hormone (PTH) secretion, in turn, causes increased calcium in the blood by acting on bones, intestines, and kidneys 223.
The parathyroid glands (four pea-sized glands that lie just behind the thyroid glands in your neck) only purpose is to make a hormone called parathyroid hormone (PTH), which helps maintain the right balance of calcium in your body. The regulation of both calcium and phosphate balance is greatly influenced by concentrations of circulating parathyroid hormone (PTH), vitamin D, and, to a lesser extent, calcitonin (Figure 1). When your blood calcium levels are low, your parathyroid glands secrete parathyroid hormone (PTH) (Figure 1). The parathyroid hormone (PTH) helps your bones release calcium into the blood. Parathyroid hormone (PTH) also increases serum calcium by stimulating conversion of vitamin D to its most active form, calcitriol also known as 1,25-dihydroxycholecalciferol or 1,25-dihydroxyvitamin D. Calcitriol also known as 1,25-dihydroxycholecalciferol (1,25-dihydroxyvitamin D), which is actually a hormone, helps your body absorb calcium by increasing the percentage of dietary calcium absorbed by your intestines. Despite increased calcium absorption, long-term increases in parathyroid hormone (PTH) secretion generally result in further bone resorption by inhibiting osteoblastic function and promoting osteoclastic activity. PTH and vitamin D both function as important regulators of bone growth and bone remodeling. Together, parathyroid hormone (PTH) and vitamin D, along with other hormones and minerals, help move calcium in or out of body tissues to keep your blood calcium at a normal level.
Parathyroid hormone (PTH) has several actions, but the most important is to defend against low blood calcium or hypocalcemia. Parathyroid cells sense decreases in serum calcium and, in response, release preformed PTH into your bloodstream. Parathyroid hormone (PTH) increases serum calcium within minutes by increasing kidney and intestinal absorption of calcium and by rapidly mobilizing calcium and phosphate from bone (bone resorption) (Figure 1). Kidney calcium excretion generally parallels sodium excretion and is influenced by many of the same factors that govern sodium transport in the proximal tubule. However, PTH enhances distal tubular calcium reabsorption independently of sodium.
Parathyroid hormone (PTH) raises blood calcium levels by 224:
- Effects of parathyroid hormone (PTH) on your bones, where most of your body’s calcium is stored, to release calcium into the blood.
- In your bones, parathyroid hormone (PTH) stimulates the release of calcium in an indirect process through osteoclasts which ultimately leads to the resorption of your bones. However, before osteoclast activity, PTH directly stimulates osteoblasts which increases their expression of RANKL, a receptor activator for nuclear factor kappa-B ligand, allowing for the differentiation of osteoblasts into osteocytes. PTH also inhibits the secretion of osteoprotegerin, allowing for preferential differentiation into osteoclasts. Osteoprotegerin normally competitively binds with RANKL diminishing the ability to form osteoclasts. Osteoclasts possess the ability to remodel the bones (resorption) by dissolution and degradation of hydroxyapatite and other organic material, releasing calcium into the blood.
- Effects of parathyroid hormone (PTH) on your kidneys helping your kidneys hold on to calcium and return it to your blood instead of flushing it out in urine.
- In your kidneys, the parathyroid hormone (PTH) has 3 functions in increasing serum calcium levels. Most of the physiologic calcium reabsorption in the nephron takes place in the proximal convoluted tubule and additionally at the ascending loop of Henle. Circulating parathyroid hormone (PTH) targets the distal convoluted tubule and collecting duct, directly increasing calcium reabsorption. Parathyroid hormone (PTH) decreases phosphate reabsorption at the proximal convoluted tubule. Phosphate ions in the serum form salts with calcium that are insoluble, resulting in decreased plasma calcium. The reduction of phosphate ions, therefore, results in more ionized calcium in the blood.
- Also in your kidneys, parathyroid hormone (PTH) stimulates the production of 1-alpha-hydroxylase in the proximal convoluted tubule. This enzyme, 1-alpha-hydroxylase, is required to catalyze the synthesis of active vitamin D called “calcitriol” or 1,25 dihydroxyvitamin D (1,25-dihydroxycholecalciferol) from the inactive form 25-hydroxycholecalciferol 225. The main effect of calcitriol or 1,25 dihydroxyvitamin D (1,25-dihydroxycholecalciferol) is to increase calcium absorption from your gut 226. Calcitriol or 1,25 dihydroxyvitamin D (1,25-dihydroxycholecalciferol) binds to the vitamin D receptor in the epithelial cells of the duodenum causing the synthesis of calcium binding proteins that regulate active intestinal calcium absorption 226, 227. In the small intestine, vitamin D allows the absorption of calcium through an active transcellular pathway and a passive paracellular pathway. The transcellular pathway requires energy, while the paracellular pathway allows for the passage of calcium through tight junctions. Calcitriol or 1,25 dihydroxyvitamin D (1,25-dihydroxycholecalciferol) also stimulates calcium reabsorption in your kidneys.
- PTH indirect effects on your small intestines by helping your intestines absorb calcium from food
A 70-kg person has approximately 1.2 kg of calcium in the body, more than 99% of which is stored as hydroxyapatite in bones 228. Less than 1% (5-6 g) of calcium is located in the intracellular (inside the cell) and extracellular (oustide the cell) compartments, with only 1.3 g located extracellularly 228. The total calcium concentration in the plasma is 4.5-5.1 mEq/L (9-10.2 mg/dL). Fifty percent of plasma calcium is ionized, 40% is bound to proteins (90% of which binds to albumin), and 10% circulates bound to anions (eg, phosphate, carbonate, citrate, lactate, sulfate). Ionized calcium is the necessary plasma fraction for normal physiologic processes. In the neuromuscular system, ionized calcium facilitates nerve conduction, muscle contraction, and muscle relaxation. Calcium is necessary for bone mineralization and is an important cofactor for hormonal secretion in endocrine organs. At the cellular level, calcium is an important regulator of ion transport and membrane integrity.
Approximately 500 mg of calcium is removed from the bones daily and replaced by an equal amount 228. Normally, the amount of calcium absorbed by the intestines is matched by urinary calcium excretion. Despite these enormous fluxes of calcium, the levels of ionized calcium remain stable because of the rigid control maintained by parathyroid hormone (PTH), vitamin D, and calcitonin through complex feedback loops (see Figure 1). These compounds act primarily at bone, renal, and gastrointestinal sites. Calcium levels are also affected by magnesium and phosphorus 229.
Insufficient intakes of calcium do not produce obvious symptoms in the short term because the body maintains calcium levels in the blood by taking it from bone. Over the long term, intakes of calcium below recommended levels have health consequences, such as causing low bone mass (osteopenia) and increasing the risks of osteoporosis and bone fractures. Chronically low calcium intakes in growing individuals may prevent the attainment of optimal peak bone mass. Once peak bone mass is achieved, inadequate calcium intake may contribute to accelerated bone loss and ultimately to the development of osteoporosis 3.
Getting too little calcium can cause several conditions, including the following:
- Osteopenia is a condition that begins as you lose bone mass and your bones get weaker. Osteopenia happens when the inside of your bones become brittle from a loss of calcium. Osteopenia is very common as you age. Total bone mass peaks around age 35. Sometimes, osteopenia is a precursor to osteoporosis. People who have osteopenia are at a higher risk of having osteoporosis.
- Osteoporosis, a condition in which bones become weak and brittle, increasing the chance they may break and increases the risk of falling.
- Rickets, a condition in children in which bones become soft and deformed because they don’t have enough calcium and phosphorus. It is caused by not having enough vitamin D in the diet or by not getting enough sunlight. In adults, this condition is called osteomalacia.
- Osteomalacia, a condition in adults in which bones become soft and deformed because they don’t have enough calcium and phosphorus. It is usually caused by not having enough vitamin D in the diet, not getting enough sunlight, or a problem with the way the body uses vitamin D. Symptoms include bone pain and muscle weakness. When the condition occurs in children, it is called rickets.
- Patients with severe low blood calcium or hypocalcemia of less than 7 mg/dL and those with an acute drop in calcium level can develop seizures or life-threatening arrhythmia 230.
Over the long term, inadequate calcium intake causes osteopenia which if untreated can lead to osteoporosis, which is characterized by fragile bones and an increased risk of falling 3. The risk of bone fractures also increases, especially in older individuals. Calcium deficiency can also cause rickets, though it is more commonly associated with vitamin D deficiency 231, 232, 233, 234. In children with rickets, the growth cartilage does not mineralize normally, which can lead to irreversible changes in the skeletal structure (Figure 3) 3. Another effect of chronic calcium deficiency is osteomalacia, or defective bone mineralization and bone softening, which can occur in adults and children 3. For rickets and osteomalacia, the requirements for calcium and vitamin D appear to be interrelated in that the lower the serum vitamin D level (measured as 25-hydroxyvitamin D [25(OH)D]), the more calcium is needed to prevent these diseases 235.
Certain groups of people are more likely than others to have trouble getting enough calcium 9:
- Postmenopausal women. The body absorbs and retains less calcium after menopause. Over time, this can lead to fragile bones.
- People who don’t drink milk or eat other dairy products. Dairy products are rich sources of calcium, but people with lactose intolerance, people with milk allergies, and vegans (people who don’t consume any animal products) must find other sources of calcium. Options include lactose-free or reduced-lactose dairy products; canned fish with bones; certain vegetables, such as kale, broccoli, and Chinese cabbage; calcium-fortified fruit juices and milk substitutes such as soy and almond beverages, tofu, and ready-to-eat cereals; and dietary supplements that contain calcium.
- People who have lactose intolerance and avoid dairy products
- Women who have an eating disorder (for example, anorexia)
- People who do not eat animal, fish, or dairy products (vegans)
- People who take certain medicines for osteoporosis
- People who have parathyroid disorders, inflammatory bowel disease, or liver or kidney disease
A substantial proportion of people in the United States consume less than recommended amounts of calcium. An analysis of 2007–2010 data from the National Health and Nutrition Examination Survey (NHANES) found that 49% of children aged 4–18 years and 39% of all individuals aged 4 and older consume less than the Estimated Average Requirement (average daily level of intake estimated to meet the requirements of 50% of healthy individuals; usually used to assess the nutrient intakes of groups of people and to plan nutritionally adequate diets for them; can also be used to assess the nutrient intakes of individuals) for calcium from foods and supplements 218.
Average daily intakes of calcium from foods and beverages are 1,083 mg for men aged 20 and older and 842 mg for women 219. For children aged 2–19, mean daily intakes of calcium from foods and beverages range from 965 to 1,015 mg 219. Approximately 22% of men, 32% of women, and 4 to 8% of children take a dietary supplement containing calcium 219. Average daily calcium intakes from both foods and supplements are 1,156 mg for men, 1,009 mg for women, and 968 to 1,020 mg for children 219.
According to 2009–2012 National Health and Nutrition Examination Survey data, rates of calcium inadequacy (intakes below the Estimated Average Requirement) are higher among non-Hispanic Blacks and non-Hispanic Asians (47–48%) than among Hispanics (30%) and non-Hispanic Whites (24%) in the United States 220. Poverty is also associated with a higher risk of inadequacy. National Health and Nutrition Examination Survey data from 2007 to 2014 show that the risk of inadequate calcium intakes (less than 800 to 1,100 mg) is 11.6% higher among adults aged 50 and older in households earning less than $20,000 per year than other households 221.
Unfortunately, getting more calcium than your body needs can cause adverse (negative) effects. This includes kidney stones, frequent urination, belly pain, nausea/vomiting, and fatigue. It is rare to get too much calcium from food alone. There is an amount of calcium that most people can take each day without developing problems. This is called the tolerable upper intake level (UL). Doctors recommend the following tolerable upper intake levels by age:
- Ages 0-6 months: 1,000 mg per day
- Ages 7-12 months: 1,500 mg per day
- Ages 1-8: 2,500 mg per day
- Ages 9-18: 3,000 mg per day
- Ages 19-50: 2,500 mg per day
- Ages 51 and older: 2,000 mg per day
- Pregnant and breastfeeding teens: 3,000 mg per day
- Pregnant and breastfeeding adults: 2,500 mg per day
In addition, research shows that high intakes of calcium may provide health benefits in lowering the risk of colon cancer, preeclampsia (pregnancy related complication in which affected women develop high blood pressure [hypertension]; they can also have abnormally high levels of protein in their urine [proteinuria]), and metabolic syndrome such as diabetes. Other research shows that high calcium intake may raise the risk of prostate cancer. Conflicting research suggests that a high intake of calcium may or may not provide a protection against heart disease 8, 6.
Figure 2. Calcium deficiency
Footnote: Parathyroid hormone (PTH) and vitamin D normally interact to protect against low calcium in blood (hypocalcemia). Problems at any level can lead to low serum calcium, but the most common problems are vitamin D deficiency and hypoparathyroidism
[Source 236 ]Figure 3. Calcium deficiency Rickets
Footnote: A 17‐year‐old patient with marked genu valgum deformities due to dietary calcium deficiency.
[Source 231 ]Calcium deficiency causes
A low blood calcium level (hypocalcemia) usually implies abnormal parathyroid function since the skeleton provides a large reserve of calcium for maintaining normal blood levels, especially in the case of low dietary calcium intake 6. Other causes of abnormally low blood calcium (hypocalcemia) concentrations include chronic kidney failure, vitamin D deficiency, and low blood magnesium levels often observed in cases of severe alcoholism 6. Magnesium deficiency can impair parathyroid hormone (PTH) secretion by the parathyroid glands and lower the responsiveness of osteoclasts to PTH 6. Therefore, magnesium supplementation is required to correct hypocalcemia in people with low serum magnesium concentrations 6. In general, kidney failure remains by far the most common cause of hypocalcemia, followed by vitamin D deficiency, magnesium deficiency and acute pancreatitis 230.
Common causes of low blood calcium level or hypocalcemia 236:
- Vitamin D deficiency as a result of limited exposure to ultraviolet light or low dietary intake
- Vitamin D deficiency as a result of malabsorption
- Hypoparathyroidism as a result of surgery
- Hypoparathyroidism as a result of autoimmune disease
- Kidney disease
Rare causes of low blood calcium level or hypocalcemia 236:
- Parathyroid hormone resistance
- Vitamin D resistance
- Autosomal dominant hypocalcemia
- Hypomagnesemia
- Sclerotic metastases
Other causes of low blood calcium level or hypocalcemia 236:
- Hungry bone syndrome after parathyroidectomy for hyperparathyroidism 237
- Infusion of phosphate or calcium chelators, such as citrate, with massive blood transfusion
- Critical illness
- After intravenous treatment with bisphosphonates, especially high dose treatment in vitamin D deficient patients.
Parathyroid Hormone (PTH) Deficiency
Parathyroid Hormone (PTH) Deficiency occurs as a result of decreased PTH secretion, which can be due to destruction of the parathyroid glands (postsurgical or autoimmune), abnormal regulation of PTH production and secretion, or abnormal development of the parathyroid gland. Post-surgical is the most common cause of hypoparathyroidism 230.
- Post-surgical: Post-surgical is the most common cause of hypoparathyroidism 230. It can occur after thyroidectomy, parathyroidectomy, or radical neck surgery. The normal parathyroid gland is made up of about 30% capillary cells. This makes the parathyroid gland very sensitive to disruption of arterial blood supply or venous drainage, which can result from mechanical, thermal, or electrical injury during thyroidectomy. The resulting hypoparathyroidism is usually transient but can also be permanent with subsequent transient or permanent hypocalcemia. Accidental removal of all 4 parathyroid glands during thyroidectomy, though not common, is also a major cause of hypoparathyroidism. Hypoparathyroidism after parathyroidectomy is usually a result of suppression of the remaining parathyroid glands by hypercalcemia from parathyroid adenoma before surgery. This is also transient in most cases but is usually permanent if all 4 glands are removed without autotransplantation of the parathyroid gland. In cases of severe hyperparathyroidism with significantly elevated PTH levels before surgery, as in tertiary hyperparathyroidism in kidney disease, the abrupt drop in PTH levels after surgery can lead to severe hypocalcemia due to unopposed osteoblast activity causing significant calcium uptake into the bones. This condition is termed “hungry bone syndrome” 238, 239, 240
- Autoimmune: Autoantibodies against the parathyroid gland are the main cause of autoimmune hypoparathyroidism which can be a manifestation of polyglandular autoimmune syndrome type 1 along with chronic mucocutaneous candidiasis and adrenal insufficiency.
Abnormal parathyroid gland development: X-linked or autosomal recessive hypoparathyroidism causes abnormal parathyroid gland development. This can be isolated or associated with complex congenital syndromes like DiGeorge syndrome. - Parathyroid gland destruction: Parathyroid gland destruction can also be due to rare causes such as infiltrative diseases of the parathyroid glands like granulomatous diseases, hemochromatosis, Wilson disease, or irradiation. Human immunodeficiency virus (HIV) infection is also a rare cause of symptomatic hypoparathyroidism. Lastly, activating mutation of calcium-sensing receptors (CaSR) decreases the setpoint of CaSR, causing hypoparathyroidism and hypocalcemia.
High Parathyroid Hormone (PTH) Levels
- Absolute or relative vitamin D deficiency: Vitamin D maintains normal calcium by enhancing intestinal calcium absorption and bone resorption. Vitamin D deficiency could be from decreased intake or malabsorption, inadequate sun exposure, liver disease, kidney disease results, and decrease conversion to its active metabolite (1,25-dihydroxy vitamin D). This can lead to decreased calcium absorption and bone resorption. The resulting hypocalcemia leads to a compensatory increase in PTH secretion (secondary hyperparathyroidism).
- Chronic kidney disease (CKD): Chronic kidney disease (CKD) leads to impaired phosphate excretion and impaired hydroxylation of 25 hydroxyvitamin D to 1,25-dihydroxy vitamin D. This drives PTH secretion and can cause secondary hyperparathyroidism. However, due to impaired vitamin D metabolism and high phosphorus level, the serum calcium remains low despite the high PTH
- Pseudohypoparathyroidism: This is a genetic disorder that causes end-organ resistance to the action of PTH and is characterized by hypocalcemia, hyperphosphatemia, and elevated PTH concentration.
Other Causes
- Pseudohypocalcemia: Serum calcium is normally bound to proteins in the blood, most prominently albumin, and therefore low albumin states can give a falsely low total serum calcium level. Ionized calcium level is usually normal in these states, and thus a correction of adding 0.8 mg/dL to serum calcium level for every 1gm drop in serum albumin below normal (4 gm/dL) is recommended.
- Acidosis or alkalosis: Calcium binding to albumin is dependent on the serum pH, and thus in states of severe acidosis, ionized calcium is increased and vice-versa decreased in severe alkalosis. There is no reliable correction factor to estimate this shift in ionized calcium level, so direct measurement of ionized calcium is recommended in these cases to guide therapy.
- Acute pancreatitis: Hypocalcemia is often seen in the setting of acute pancreatitis due to calcium deposition in the abdominal cavity as a result of ongoing inflammation 241.
- Severe sepsis or critical illness: Severe sepsis can lead to hypocalcemia through unclear mechanisms. Impaired PTH secretion, dysregulation of magnesium metabolism, and impaired calcitriol secretion have been identified as potential mechanisms, but none have been proven as of yet 242. More recent reports also indicated hypocalcemia related to severe Covid-19 infection 243.
- Hypomagnesemia or hypermagnesemia: Low serum magnesium can cause hypocalcemia due to induced PTH resistance. This usually occurs when the serum magnesium level drops below 0.8 mEq/L (1 mg/dL or 0.4 mmol/L). Decreased PTH secretion can occur in more severe hypomagnesemia 244. Severe hypermagnesemia, although rare, can also cause hypocalcemia by suppressing PTH secretion through a decreased sensitivity of calcium-sensing receptors.
- Acute hyperphosphatemia: This is an uncommon cause of hypocalcemia which is likely precipitated because of extravascular deposition of calcium phosphate products.
- Drugs: Bisphosphonates and denosumab both inhibit osteoclastic bone resorption, which can cause hypocalcemia which can be severe in some cases. Concomitant vitamin D deficiency makes hypocalcemia more likely to occur in patients treated with those drugs. Patients being treated with these agents should have their calcium and vitamin D levels checked, and low levels should be corrected before initiation of treatment. Cinacalcet is a calcimimetic agent that works by stimulating the calcium-sensing receptors (CaSR) and thus decreasing PTH secretion. It is used in the treatment of both primary and secondary hyperparathyroidism. The resulting decrease in PTH secretion can cause hypocalcemia. Cisplatin, a chemotherapeutic drug, can also cause hypocalcemia through hypomagnesemia. Foscarnet can cause hypocalcemia by forming complexes with ionized calcium, thereby reducing levels of ionized calcium. It is therefore important to regularly monitor calcium levels during treatment with all of the above-mentioned drugs 245, 246.
- Massive blood transfusion: Massive blood transfusion can cause an acute decline in ionized calcium due to calcium binding with citrate, which is used as an anticoagulant in the stored blood 247.
- Pregnancy: There are reports of hypocalcemia during pregnancy, mostly related to poor diet, hyperemesis gravidarum, or underline diseases 248.
- Osteoblastic metastasis, as in prostate cancer, can cause hypocalcemia which can be severe in rare cases. This is a result of increased calcium uptake into the bones from increased osteoblastic activity.
Groups at Risk of Calcium Deficiency
The following groups of people are more likely than others to have trouble getting enough calcium.
Postmenopausal women
Menopause leads to bone loss because decreases in estrogen production reduce calcium absorption and increase urinary calcium loss and calcium resorption from bone 3. On average, women lose approximately 1% of their bone mineral density (BMD) per year after menopause 94. Over time, these changes lead to decreased bone mass and fragile bones 3. About 30% of postmenopausal women in the United States and Europe have osteoporosis, and at least 40% of those with this condition develop at least one fragility fracture (a fracture that occurs after minor trauma, such as a fall from standing height or lower) 249. The calcium Recommended Dietary Allowance (average daily level of intake sufficient to meet the nutrient requirements of nearly all (97%–98%) healthy individuals; often used to plan nutritionally adequate diets for individuals) is 1,200 mg for women older than 50 years (vs. 1,000 mg for younger women) to lessen bone loss after menopause 3.
Women of childbearing age whose menstrual periods stop
Women of childbearing age whose menstrual periods stop (amenorrhea) because they exercise heavily, eat too little, or both. They need sufficient calcium to cope with the resulting decreased calcium absorption, increased calcium losses in the urine, and slowdown in the formation of new bone.
Amenorrhea, the condition in which menstrual periods stop or fail to initiate in women of childbearing age, results from reduced circulating estrogen levels that, in turn, have a negative effect on calcium balance. Amenorrheic women with anorexia nervosa have decreased calcium absorption and higher urinary calcium excretion rates, as well as a lower rate of bone formation than healthy women 250. The “female athlete triad” refers to the combination of disordered eating, amenorrhea, and osteoporosis. Exercise-induced amenorrhea generally results in decreased bone mass 251, 252. In female athletes and active women in the military, low bone-mineral density, menstrual irregularities, certain dietary patterns, and a history of prior stress fractures are associated with an increased risk of future stress fractures 253. Such women should be advised to consume adequate amounts of calcium and vitamin D. Supplements of these nutrients have been shown to reduce the risk of stress fractures in female Navy recruits during basic training 254.
Individuals who avoid dairy products
People with lactose intolerance, those with an allergy to milk, and those who avoid eating dairy products (including vegans) have a higher risk of inadequate calcium intakes because dairy products are rich sources of calcium 3, 255. Options for increasing calcium intakes in individuals with lactose intolerance include consuming lactose-free or reduced-lactose dairy products, which contain the same amounts of calcium as regular dairy products 3, 12. Those who avoid dairy products because of allergies or for other reasons can obtain calcium from non-dairy sources, such as some vegetables (e.g., kale, broccoli, and Chinese cabbage [bok choi]), canned fish with bones, or fortified foods (e.g., fruit juices, breakfast cereals, and tofu) 3. However, these individuals typically need to eat foods fortified with calcium or take supplements to obtain recommended amounts 256.
People with lactose intolerance
People with lactose intolerance cannot digest this natural sugar found in milk and experience symptoms like bloating, gas, and diarrhea when they drink more than small amounts at a time. They usually can eat other calcium-rich dairy products that are low in lactose, such as yogurt and many cheeses, and drink lactose-reduced or lactose-free milk.
Lactose intolerance refers to symptoms (such as bloating, flatulence, and diarrhea) that occur when one consumes more lactose, the naturally occurring sugar in milk, than the enzyme lactase produced by the small intestine can hydrolyze into its component monosaccharides, glucose and galactose 257. The symptoms vary, depending on the amount of lactose consumed, history of consumption of lactose-containing foods, and type of meal. Although the prevalence of lactose intolerance is difficult to discern 258, some reports suggest that approximately 25% of U.S. adults have a limited ability to digest lactose, including 85% of Asians, 50% of African Americans, and 10% of Caucasians 259, 260, 261.
Lactose-intolerant individuals are at risk of calcium inadequacy if they avoid dairy products. Research suggests that most people with lactose intolerance can consume up to 12 grams of lactose, such as that present in 8 ounces of milk, with minimal or no symptoms, especially if consumed with other foods; larger amounts can frequently be consumed if spread over the day and eaten with other foods. Other options to reduce symptoms include eating low-lactose dairy products including aged cheeses (such as Cheddar and Swiss), yogurt, or lactose-reduced or lactose-free milk. Some studies have examined whether it is possible to induce adaptation by consuming incremental lactose loads over a period of time 262, 263, but the evidence in support of this strategy is inconsistent 258.
Cow’s milk allergy is less common than lactose intolerance, affecting 0.6% to 0.9% of the population 264. People with this condition are unable to consume any products containing cow’s milk proteins and are therefore at higher risk of obtaining insufficient calcium.
To ensure adequate calcium intakes, lactose-intolerant individuals and those with cow’s milk allergy can choose nondairy food sources of the nutrient (such as kale, bok choy, Chinese cabbage, broccoli, collards and fortified foods) or take a calcium supplement.
Vegans and ovo-vegetarians
Vegans (vegetarians who eat no animal products) and ovo-vegetarians (vegetarians who eat eggs but no dairy products), because they avoid the dairy products that are a major source of calcium in other people’s diets.
Vegetarians might absorb less calcium than omnivores because they consume more plant products containing oxalic and phytic acids. Lacto-ovo vegetarians (who consume eggs and dairy) and nonvegetarians have similar calcium intakes 265, 266. However, vegans, who eat no animal products and ovo-vegetarians (who eat eggs but no dairy products), might not obtain sufficient calcium because of their avoidance of dairy foods 267, 141. In the Oxford cohort of the European Prospective Investigation into Cancer and Nutrition, bone fracture risk was similar in meat eaters, fish eaters and vegetarians, but higher in vegans, likely due to their lower mean calcium intake 268. It is difficult to assess the impact of vegetarian diets on calcium status because of the wide variety of eating practices and thus should be considered on a case by case basis.
Calcium deficiency prevention
Men and women between the ages of 18 and 50 need 1,000 milligrams of calcium a day. This daily amount increases to 1,200 milligrams when women turn 50 and men turn 70.
You can prevent calcium deficiency by eating a variety of foods. Good sources of calcium include the following 9:
- Milk, yogurt, and cheese are the main food sources of calcium for most people in the United States.
- Canned sardines and salmon with bones contain calcium.
- Dark green leafy vegetables, such as kale, broccoli, and Chinese cabbage (bok choi) also contain calcium.
- Calcium is added to some beverages, including many fruit juices and milk substitutes such as soy and almond beverages, as well as some brands of tofu and ready-to-eat cereals. To find out whether these foods have calcium added, check the product labels.
- Most grains (such as breads, pastas, and unfortified cereals) do not have high amounts of calcium. However, because people eat them often, what they contribute adds up.
If you find it difficult to get enough calcium from your diet, consider taking calcium supplements. However, too much calcium has been linked to kidney stones. Although yet unclear, some experts suggest that too much calcium, especially in supplements, can increase the risk of heart disease.
The Health and Medicine Division of the National Academies of Sciences, Engineering, and Medicine recommends that total calcium intake, from supplements and diet combined, should be no more than 2,000 milligrams daily for people older than 50 3.
However, many people in the United States get less than recommended amounts of calcium from food and supplements, especially 9:
- Children and teens aged 4 to 18 years
- People who are Black or Asian
- Adults aged 50 years and older living in poverty
Calcium supplements
Calcium is found in many multivitamin-mineral supplements, in calcium supplements, and in supplements that contain calcium and other nutrients such as vitamin D. Check the Supplement Facts label to determine the amount of calcium in the supplement.
The two main forms of calcium in dietary supplements are calcium carbonate and calcium citrate. Calcium carbonate is absorbed best when taken with food. Calcium citrate is well absorbed on an empty stomach or a full stomach. People with low levels of stomach acid—a condition most common in older people—absorb calcium citrate more easily than calcium carbonate.
Other forms of calcium in supplements and fortified foods include calcium sulfate, calcium ascorbate, calcium microcrystalline hydroxyapatite, calcium gluconate, calcium lactate, and calcium phosphate.
Calcium is absorbed best when you take 500 mg or less at one time. If you take 1,000 mg/day of calcium from supplements, for example, it is better to take a smaller dose twice a day than to take it all at once.
Calcium supplements might cause gas, bloating, and constipation in some people. If you have any of these symptoms, try spreading out the calcium dose throughout the day, taking the supplement with meals, or switching the form of calcium you take.
Vitamin D
Vitamin D improves your body’s ability to absorb calcium and improves bone health in other ways. People can get some of their vitamin D from sunlight, but this might not be a good source if you live in a high latitude, if you’re housebound, or if you regularly use sunscreen or avoid the sun because of the risk of skin cancer.
Dietary sources of vitamin D include cod liver oil, trout and salmon. Many types of milk and cereal have been fortified with vitamin D.
Most people need at least 600 international units (IU) of vitamin D a day. That recommendation increases to 800 IU a day after age 70.
People without other sources of vitamin D and especially with limited sun exposure might need a supplement. Most multivitamin products contain between 600 and 800 IU of vitamin D. Up to 4,000 IU of vitamin D a day is safe for most people.
Exercise
Exercise can help you build strong bones and slow bone loss. Exercise will benefit your bones no matter when you start, but you’ll gain the most benefits if you start exercising regularly when you’re young and continue to exercise throughout your life.
Combine strength training exercises with weight-bearing and balance exercises. Strength training helps strengthen muscles and bones in your arms and upper spine. Weight-bearing exercises — such as walking, jogging, running, stair climbing, skipping rope, skiing and impact-producing sports — affect mainly the bones in your legs, hips and lower spine. Balance exercises such as tai chi can reduce your risk of falling especially as you get older.
Calcium deficiency symptoms
Extracellular calcium concentrations are important for normal physiological functions, including those involving muscles, nerves, blood vessels and the release of hormones and enzymes. If your body doesn’t get enough calcium and vitamin D to support these important functions, it takes calcium from your bones. This is called losing bone mass (osteopenia). Losing bone mass makes the inside of your bones become weak and porous. This puts you at risk for the bone disease osteoporosis.
In the short term, most symptoms of calcium deficiency are subtle that you may not notice them at all. But if left unaddressed, low calcium intake can have detrimental effects on different body systems, including contributing to low bone mass (osteopenia) and risk for osteoporosis.
There typically are no symptoms in the early stages of bone loss. But once your bones have been weakened by osteoporosis, you might have signs and symptoms that include:
- Back pain, caused by a fractured or collapsed vertebra
- Loss of height over time
- A stooped posture
- A bone that breaks much more easily than expected.
Your bones are in a constant state of renewal — new bone is made and old bone is broken down. When you’re young, your body makes new bone faster than it breaks down old bone and your bone mass increases. After the early 20s this process slows, and most people reach their peak bone mass by age 30. As people age, bone mass is lost faster than it’s created.
Symptoms of serious calcium deficiency almost always occur in people with serious health problems, patients who are hospitalized or in people who are undergoing certain medical treatments where serum calcium levels drop to dangerous levels.
Classic symptoms of serious calcium deficiency include 236:
- Acute low calcium level in blood or hypocalcemia may lead to 269:
- Fainting (syncope), chronic heart failure (CHF), and angina (a type of chest pain caused by reduced blood flow to the heart) that can lead to death if not corrected.
- Neuromuscular symptoms 270. The development of neuromuscular excitability depends on both the absolute concentration of calcium and how rapidly it falls. Rapid falls in calcium, for example after the removal of a parathyroid adenoma, are often associated with symptoms; whereas patients who develop hypocalcemia gradually can be surprisingly free of symptoms, and the diagnosis may only become evident as an incidental biochemical finding. Longstanding hypocalcemia, even without neuromuscular symptoms, is associated with the development of neuropsychiatric symptoms, cataract formation, and occasionally raised intracranial pressure 271, 272:
- Numbness and tingling sensations in the perioral area or in the fingers and toes
- Muscle twitching, spasms or cramps, particularly in the back and lower extremities; may progress to carpopedal spasm (ie, tetany). Carpopedal spasm is characteristic but in severe cases can progress to tetany, seizures, and cardiac dysrhythmias. In patients without overt signs, underlying neuromuscular excitability can become evident with provocation—tapping the parotid gland over the facial nerve can induce facial muscle spasm (Chvostek’s sign). Chvostek’s sign is elicited by tapping on the face at a point just anterior to the ear and just below the zygomatic bone 273. Chvostek sign is a contraction of ipsilateral facial muscles subsequent to percussion over the facial nerve and is considered a clinical indicator of hypocalcemia 274. Positive Chvostek’s sign represents increased neuromuscular excitability caused by hypocalcemia. A positive response is represented by twitching of the ipsilateral facial muscles, suggesting neuromuscular excitability caused by hypocalcaemia. Although, classically described with hypocalcaemia due to hypoparathyroidism, it is also elicited in some young healthy children and alkalotic states as during vomiting and hyperventilation 273. Chvostek’s sign is very easy to test in clinical practice compared with Trousseau’s sign and hence its clinical significance. However, up to 10% of normal population will have a positive Chvostek sign in the absence of hypocalcemia; thus, this test, while suggestive, is not diagnostic of hypocalcemia 275. Conversely, a small study of patients with hypoparathyroidism and biochemically confirmed hypocalcemia found that 29% were negative for Chvostek’s sign, which makes this test a poor discriminator 276.
- Wheezing; may develop from bronchospasm
- Difficulty swallowing (dysphagia)
- Voice changes (due to laryngospasm)
- Neurologic symptoms 277:
- Irritability, impaired intellectual capacity, depression, and personality changes
- Fatigue
- Seizures (eg, grand mal, petit mal, focal)
- Other uncontrolled movements
- Chronic hypocalcemia may produce the following skin signs and symptoms 278:
- Coarse hair
- Brittle nails
- Psoriasis
- Dry skin
- Chronic pruritus
- Poor dentition
- Cataracts
Figure 4. Chvostek’s sign
Trousseau’s sign
Trousseau’s sign is a carpopedal spasm induced by ischemia through inflation of a sphygmomanometer cuff to a suprasystolic blood pressure. Trousseau’s sign emerged at 25 seconds after the start of cuff inflation, with a maximal spasm at 60 seconds. Trousseau sign reported sensitivity and specificity for hypocalcemia are 94% and 99%, respectively 279.
Test for the Trousseau sign by placing a blood pressure cuff on the patient’s arm and inflating to 20 mm Hg above systolic blood pressure for 3-5 minutes 276. This increases the irritability of the nerves, and a flexion of the wrist and metacarpal phalangeal joints can be observed with extension of the interphalangeal joints and adduction of the thumb (carpal spasm). The Trousseau sign is more specific than the Chvostek sign but has incomplete sensitivity.
Fatigue
Fatigue reduces your energy, your ability to do things and your ability to focus. Ongoing fatigue affects quality of life and state of mind. In the case of hypocalcemia, fatigue is the result of your cells being undernourished.
Poor oral health
If your body is leeching calcium from your bones to stay functional, it’s going to make your teeth more susceptible to decay and loosening — and possibly even periodontal disease.
Muscle pain and spasms
Calcium doesn’t just make our bones strong: It also ensures our muscles function properly. Specifically, it helps our muscles contract and relax. Put differently, hypocalcemia will, quite literally, make you weaker. You’ll also experience more aches, pains, stiffness and spasms.
Cognitive issues
If your cells are being deprived of calcium, you can encounter a range of cognitive symptoms, from brain fog to dizziness and confusion. There’s even preliminary evidence to suggest that calcium intake affects our mental health.
Numbness and tingling in your fingers
Calcium plays a vital role in many different parts of our central nervous system; if we’re deficient, we may see those nerves impacted, particularly in our extremities (hands, fingers, feet and toes).
Seizures
Nutritional deficiencies can sometimes trigger seizures.
Abnormal heart rhythm
Calcium is crucial to muscle function and your heart is a muscle. There is a well-established connection between calcium dysregulation and ventricular arrhythmias, which can be serious and potentially even deadly.
Regardless of calcium levels, you should go to the emergency room (ER) immediately if you’re experiencing chest pain (angina) along with other symptoms like shortness of breath, lightheadedness or fainting.
Calcium deficiency diagnosis
In order to diagnose calcium deficiency, initial laboratory testing involves measuring serum calcium, phosphate, magnesium, intact parathyroid hormone (PTH), 25-hydroxyvitamin D, and 1,25-dihydroxyvitamin D levels. Albumin, liver function studies, and coagulation parameters should be aslo obtained to assess liver dysfunction and hypoalbuminemia. Blood urea nitrogen (BUN) and serum creatinine should be measured, as elevated levels may indicate kidney dysfunction. An electrocardiogram (ECG) is also indicated. Imaging studies may include plain radiography or computed tomography (CT) scans. On radiographs, disorders associated with rickets or osteomalacia present with the pathognomonic Looser zones, which are better observed in the pubic ramus, upper femoral bone, and ribs. Radiography will also disclose osteoblastic metastases from certain tumors (eg, breast, prostate, lung), which can cause hypocalcemia. CT scans of the head may show basal ganglia calcification and extrapyramidal neurologic symptoms (in idiopathic hypoparathyroidism) 280.
Total calcium levels can be measured in serum or plasma; serum levels are typically 8.8 to 10.4 mg/dL (2. 2 to 2.6 mmol/L) in healthy adults 11, 3. However, serum calcium levels do not reflect nutritional status because of their tight homeostatic control 4. Furthermore, serum calcium concentration must be interpreted in relation to serum albumin. Serum calcium exists in an ionized form (∼50%) or is bound to albumin or other ions. Only ionized calcium or free calcium is biologically important. Various factors alter the ratio of ionized calcium to bound calcium, but the most important factor is the albumin concentration. Many medical conditions cause a decrease in serum albumin. Serum calcium concentrations are therefore “corrected” to a reference albumin concentration of 40 g/L, and for every 1 g/L of albumin above or below this value, the calcium is adjusted by decreasing or increasing by 0.08 mg/dL (0.02 mmol/L) 281. For example, a calcium concentration of 8.22 mg/dL (2.05 mmol/L) with an albumin concentration of 35 g/L would be corrected to 8.62 mg/dL (2.15 mmol/L) [40 g/L – 35 g/L = 5 X 0.08 mg/dL = 0.4 mg/dL], which would correct the hypocalcemic value to normal. This estimation is often used, but it can be unreliable in some situations, such as critical illness 282. If it is doubtful whether total calcium reflects ionized calcium or free calcium, the ionized value can be checked directly. Other situations in which the assessment of serum calcium may be inappropriately low include recent use of certain gadolinium contrast agents and contamination of blood samples by EDTA 236.
Levels of ionized or free calcium, the biologically active form, in serum are also used to measure calcium status and the normal range of ionized calcium in healthy adults is 4.6 to 5.3 mg/dL (1.15 to 1.33 mmol/L) 11, 3.
Hypocalcemia is defined as a total serum calcium concentration < 8.8 mg/dL (< 2.20 mmol/L) in the presence of normal plasma protein concentrations or as a serum ionized calcium concentration < 4.7 mg/dL (< 1.17 mmol/L) 283. Figure 5 shows an algorithm for investigating hypocalcemia.
In true hypocalcemia, parathyroid hormone (PTH) concentrations should be high if the parathyroids are responding appropriately to the reduced negative feedback of calcium or low if the parathyroid glands are the cause of the problem. “Normal” concentrations of parathyroid hormone (PTH) in this context are inappropriate and thus abnormal. A high concentration of parathyroid hormone (PTH) in the presence of normal kidney function suggests vitamin D deficiency or calcium malabsorption. A low parathyroid hormone (PTH) concentration usually indicates hypoparathyroidism. Parathyroid hormone (PTH) concentrations can be normal in hypoparathyroidism, but they are usually within the lower part of the reference range. Parathyroid hormone (PTH) concentrations can also be inappropriately normal in hypomagnesemia or when the extracellular calcium sensing receptor has increased sensitivity (an autosomal dominant genetic disorder) 284.
A raised serum alkaline phosphatase (ALP) suggests osteomalacia as a result of vitamin D deficiency. If the diagnosis of osteomalacia is suspected, a bone biopsy can determine the final diagnosis. The possibility of metastatic cancer, with sclerotic metastases causing rapid absorption of calcium into the skeleton, should also be excluded. Parathyroid hormone (PTH) stimulates renal phosphate clearance, so serum phosphate should be low in non-parathyroid disease but high in parathyroid hormone deficiency. Measuring phosphate has limited value because it varies diurnally and is affected by food intake. Kidney function should be measured because the kidney is central to several aspects of calcium homoeostasis.
Other tests may be needed in some situations. Serum 25-hydroxyvitamin D concentrations are useful in confirming vitamin D deficiency when it presents atypically, and it should be assessed in patients with possible pseudohypoparathyroidism. Serum magnesium is important for the synthesis and release of parathyroid hormone 285. In hypomagnesaemia, the release of parathyroid hormone is inhibited, leading to (potentially severe) hypocalcaemia. The prevalence of hypomagnesaemia in a large primary care cohort was 2% 286, but in intensive care patients it can be up to 65% 287. Poor nutrition associated with chronic alcohol use, prolonged diarrhea, and treatment with diuretics and certain chemotherapeutics (such as cisplatin) causes hypomagnesemia. Recognition of hypomagnesemia is important because in this setting it is difficult to reverse hypocalcemia without magnesium repletion.
In order to diagnose osteopenia, your doctor will do a bone density test to assess cumulative calcium status over the lifetime because the skeleton stores almost all calcium in the body 12. Bone density measures bone mass and bone strength. One type of bone density test is a dual-energy X-ray absorptiometry (DEXA or DXA). It measures bone density in your hips, spine, and wrist. The result can tell if you have osteopenia or osteoporosis, or if your bones are normal. The lower your score, the higher your risk of bone breakage. A T-score between -1 and -2.5 is labeled osteopenia. A T-score lower than -2.5 is labeled osteoporosis.
According to the U.S. Preventive Services Task Force, all women over 65 years old should have a bone density test 288. Women under 65 years old should be tested as well if they’re at high risk 288.
Figure 5. Calcium deficiency diagnostic algorithm
[Source 236 ]Parathyroid hormone (PTH)
The parathyroid hormone (PTH) level should be checked as early as possible. The parathyroid hormone (PTH) test is an antibody-mediated radioimmunoassay. Low-to-normal PTH levels occur in patients with hereditary or acquired hypoparathyroidism and in patients with severe hypomagnesemia.
Patients with ineffective PTH have elevated PTH levels. The PTH elevation is a result of hypocalcemia.
Vitamin D blood test
Doctors test for vitamin D levels because low levels are common in people with primary hyperparathyroidism.
In the blood, a form of vitamin D known as 25-hydroxyvitamin D [25(OH)D] or “calcidiol” is measured in either nanomoles per liter (nmol/L) or nanograms per milliliter (ng/mL). One nmol/L is equal to 0.4 ng/mL. So, for example, 50 nmol/L is the same as 20 ng/mL.
- Levels of 50 nmol/L (20 ng/mL) or above are adequate for most people for bone and overall health.
- Levels below 30 nmol/L (12 ng/mL) are too low and might weaken your bones and affect your health.
- Levels above 125 nmol/L (50 ng/mL) are too high and might cause health problems.
In patients with primary hyperparathyroidism, the low vitamin D level can further stimulate the parathyroid glands to make even more parathyroid hormone (PTH). Also, a very low vitamin D level may cause a secondary form of hyperparathyroidism, which resolves when vitamin D levels are returned to normal.
- A low 25-hydroxyvitamin D [25(OH)D] or “calcidiol” level suggests vitamin D deficiency from poor nutritional intake, lack of sunlight, or malabsorption.
- Low levels of 1,25-dihydroxyvitamin D [1,25(OH)2D] or “calcitriol” in association with high PTH suggest ineffective PTH from a lack of vitamin D, as observed in patients with chronic kidney failure, vitamin D–dependent rickets type 1 (VDDR-I), and pseudohypoparathyroidism 289.
Assessing vitamin D status by measuring serum 25-hydroxyvitamin D [25(OH)D] or “calcidiol” concentrations is complicated by the considerable variability of the available assays (the two most common ones involve antibodies or chromatography) used by laboratories that conduct the analyses 290, 291. As a result, a finding can be falsely low or falsely high, depending on the assay used and the laboratory. The international Vitamin D Standardization Program has developed procedures for standardizing the laboratory measurement of 25-hydroxyvitamin D [25(OH)D] or “calcidiol” to improve clinical and public health practice 292, 293, 294.
In contrast to 25-hydroxyvitamin D [25(OH)D], circulating 1,25-dihydroxyvitamin D [1,25(OH)2D] or “calcitriol” is generally not a good indicator of vitamin D status because it has a short half-life measured in hours, and serum levels are tightly regulated by parathyroid hormone, calcium, and phosphate 3. Levels of 1,25-dihydroxyvitamin D [1,25(OH)2D] do not typically decrease until vitamin D deficiency is severe 295.
Urinary cyclic adenosine monophosphate (cAMP) may help differentiate hypoparathyroidism from pseudohypoparathyroidism types 1 and 2. Urinary cAMP levels are generally elevated in hypoparathyroidism 289.
Electrocardiogram
Acute hypocalcemia causes prolongation of the QT interval, which may lead to ventricular dysrhythmias 296. It also causes decreased myocardial contractility, which can lead to heart failure, hypotension, and angina 296. Cardiomyopathy and ventricular tachycardia may be reversible with treatment.
Kidney imaging tests
Doctors may use one of the following imaging tests to look for kidney stones.
- Ultrasound. Ultrasound uses a device called a transducer that bounces safe, painless sound waves off organs to create an image of their structure. A specially trained technician does the procedure. A radiologist reads the images, which can show kidney stones.
- Abdominal x-ray. An abdominal x-ray is a picture of the abdomen that uses low levels of radiation and is recorded on film or on a computer. During an abdominal x-ray, you lie on a table or stand up. A technician positions the x-ray machine close to your abdomen and asks you to hold your breath so the picture won’t be blurry. A radiologist reads the x-ray, which can show the location of kidney stones in the urinary tract. Not all stones are visible on an abdominal x-ray.
- Computed tomography (CT) scans. CT scans use a combination of x-rays and computer technology to create images of your urinary tract. CT scans sometimes use a contrast medium—a dye or other substance that makes structures inside your body easier to see. Contrast medium isn’t usually needed to see kidney stones. For the scan, you’ll lie on a table that slides into a tunnel-shaped machine that takes the x-rays. A radiologist reads the images, which can show the size and location of a kidney stone.
Bone mineral density test
Bone mineral density (BMD) at specific locations of your hip and spine is measured by dual-energy X-ray absorptiometry, also called a DXA or DEXA scan, which uses low-dose x-rays to measure bone density. During the DEXA test, you will lie on a padded table while a technician moves the scanner over your body. A bone expert or radiologist will read the scan.
The DEXA provides a T-score that is used for diagnostic purposes. Clinically, a T-score of -2.5 standard deviations (SD) or below gives a diagnosis of osteoporosis in postmenopausal women and men aged ≥50 years (Table 3).
Table 3. Bone mineral density DEXA test T-score cutoffs
| Normal bone mass | T-score equal to or above -1 standard deviation (SD) |
| Low bone mass (osteopenia) | T-score between -1 SD and -2.5 SD |
| Osteoporosis | T-score equal to or below -2.5 SD |
Risk of Fracture
The burden of osteoporosis lies primarily in the fractures that arise. Osteoporotic fractures are often precipitated by falls from standing height, although vertebral fractures can occur in the absence of a fall during daily, routine activities. About 70% of osteoporotic fractures affect women. Worldwide, 1 in 3 women and 1 in 5 men aged >50 years will suffer from a fracture.
The risk of osteoporotic fracture is influenced by bone mass, bone structure (microarchitecture, geometry), and propensity to fall (balance, mobility, muscular strength). The Fracture Risk Assessment Tool (https://frax.shef.ac.uk/FRAX/index.aspx) is an online tool that helps estimate your probability of fracture over the next 10 years, taking bone mineral density (BMD) measurement and additional risk factors into account (Table 4).
Table 4. 10-year Fracture Risk Probability
| Age (between 40 and 90 years) |
| Sex |
| Weight and height |
| Previous osteoporotic fracture |
| Parental history of hip fracture |
| Current tobacco smoking |
| Use of glucocorticoids (>3 months of doses equivalent to 5 mg/day of prednisolone) |
| Confirmed diagnosis of rheumatoid arthritis |
| Conditions associated with secondary osteoporosis (type 1 diabetes mellitus, osteogenesis imperfecta, untreated long-standing hyperparathyroidism, hypogonadism or premature menopause [<45 years], chronic malnutrition, or malabsorption and chronic liver disease) |
| Alcohol intake: ≥3 units (drinks)/day |
| Low femoral neck BMD |
Calcium deficiency treatment
Treatment for calcium deficiency starts with changes to your diet and lifestyle. Your doctor will suggest ways for you to get more calcium and vitamin D in your diet. Nonfat and low-fat dairy products, such as yogurt, cheese, and milk, are good sources of calcium. Other sources of calcium include dried beans, salmon, spinach, and broccoli. Some foods, such as orange juice, breads, and dry cereals, may be fortified with calcium, vitamin D, or both. Very few foods naturally contain vitamin D. Fatty fish (like trout, salmon, tuna, and mackerel) and fish liver oils are among the best natural sources of vitamin D. Fortified foods provide most of the vitamin D in the diets of people in the United States 297. Check the Nutrition Facts label for the amount of vitamin D in a food or beverage. Almost all of the U.S. milk supply is fortified with about 3 mcg (120 IU) vitamin D per cup. Many plant-based alternatives such as soy milk, almond milk, and oat milk are similarly fortified. But foods made from milk, like cheese and ice cream, are usually not fortified. If you don’t get enough calcium and vitamin D from the food you eat, your doctor may suggest taking a vitamin D or calcium supplement as well.
You also should stop smoking and limit alcohol. Regular consumption of more than two alcoholic drinks a day increases the risk of osteoporosis. The exact role tobacco plays in osteoporosis isn’t clear, but it has been shown that tobacco use contributes to weak bones.
Women 50 years of age and younger and men 70 years of age and younger should get 1,000 mg of calcium per day. Women older than 50 years of age and men older than 70 years of age should get 1,200 mg of calcium per day. Women who are post-menopausal may need 1,500 mg of calcium per day. It is best to get your calcium from food.
Many countries around the world and some professional societies have somewhat different guidelines for vitamin D intakes 298. Serum concentration of 25-Hydroxyvitamin D [25(OH)D] is currently the main indicator of vitamin D status. It reflects vitamin D produced endogenously and that obtained from foods and supplements 3. The Endocrine Society states, for example, that to maintain serum 25-Hydroxyvitamin D [25(OH)D] levels above 75 nmol/L (30 ng/mL), adults might need at least 37.5 to 50 mcg (1,500–2,000 IU)/day of supplemental vitamin D, and children and adolescents might need at least 25 mcg (1,000 IU)/day 299. In contrast, the United Kingdom Scientific Advisory Committee on Nutrition recommends intakes of 10 mcg (400 IU)/day for its citizens aged 4 years and older 300.
And to prevent bone loss or osteopenia, your doctor will want you to increase your physical activity. Weight-bearing exercise can help strengthen your bones and increase your bone mass. It also can build muscle and improve your balance, which help prevent against broken bones. This is why it’s important to begin an active lifestyle at a young age. Examples of weight-bearing exercise include walking, jogging, and climbing steps. Try to do a mix of strength training and weight-bearing exercises.
If you’re at risk for falls, reduce your risk by getting rid of tripping hazards in your home. For example, remove rugs, avoid slippery surfaces, move electrical cords and wear low-heeled shoes with nonslip soles. You can install grab bars in certain places, such as your bathroom and shower. The grab bars can help you move around more easily and safely.
Your doctor may prescribe medicine(s) to help treat osteoporosis. Treatment recommendations are often based on an estimate of your risk of breaking a bone in the next 10 years using information such as the bone density test (see Tables 3 and 4 above). The Fracture Risk Assessment Tool (https://frax.shef.ac.uk/FRAX/index.aspx) is an online tool that helps estimate your probability of fracture over the next 10 years, taking bone mineral density (BMD) measurement and additional risk factors into account (https://frax.shef.ac.uk/FRAX/index.aspx).
If your risk of fracture is low, you might not need medication and instead might focus on modifying your risk factors for bone loss and falls.
Figure 6. Hypocalcemia treatment algorithm
Footnote: In an emergency setting parenteral calcium is likely to be needed. If medium or long term treatment is needed, vitamin D or one of its analogues should be given 285
[Source 236 ]Hypocalcemia treatment
The treatment of hypocalcemia depends on the cause, the severity, the presence of symptoms, and how rapidly the hypocalcemia developed 236. Most cases of hypocalcemia are clinically mild and require only supportive treatment and further laboratory evaluation. Oral calcium repletion may be indicated for outpatient treatment of mild cases. On occasion, severe hypocalcemia may result in seizures, tetany, refractory hypotension, or arrhythmias that require a more aggressive approach, including prompt management in hospital and treatment with intravenous infusions of calcium.
Asymptomatic patients with corrected serum calcium less than 1.9 mmol/L may develop serious complications and admission should be considered. Figure 6 shows an algorithm for managing acute hypocalcemia in adults that is based on clinical experience and expert recommendations 301. Calcium gluconate is the preferred form of intravenous calcium because calcium chloride is more likely to cause local irritation 236. One or two 10 ml ampoules of 10% calcium gluconate should be diluted in 50-100 ml of 5% dextrose and infused slowly over 10 minutes 236. Electrocardiographic (ECG) monitoring is recommended because arrhythmias can occur if correction is too rapid 236. Treatment can be repeated until symptoms have cleared. Often this offers only temporary relief, and continuous administration of a dilute solution of calcium may be needed to prevent recurrence of hypocalcemia. Ten 10 ml ampoules of 10% calcium gluconate in 1 litre of 5% dextrose or 0.9% saline may be given at an initial infusion rate of 50 ml/hour, with the aim of maintaining serum calcium at the lower end of the reference range 301. An infusion of 10 ml/kg of this solution over four to six hours will increase serum calcium by 0.3-0.5 mmol/l. Oral calcium supplements should be given concurrently, and—if parathyroid hormone is deficient or non-functional—calcitriol should be given (for example, 1 mcg/day).
Patients taking digoxin have increased cardiac sensitivity to fluctuations in serum calcium, so intravenous calcium administration should be more cautious in this setting, with careful electrocardiographic monitoring. Patients with hypocalcemia related to hypomagnesemia will need magnesium replacement 236.
Persistent hypocalcemia
With milder degrees of hypocalcemia, treatment depends on the underlying cause 236. In vitamin D deficiency, treatment should be with vitamin D, either ergocalciferol (vitamin D2) or cholecalciferol (vitamin D3; these two forms of vitamin D can be considered identical in this setting) 236. Most available vitamin D preparations include calcium, and two tablets of calcium with vitamin D, each containing 400 IU vitamin D, are usually given daily 236. This dose of vitamin D is relatively low, and patients with symptomatic vitamin D deficiency or those who fail to respond can be treated in the short term with higher doses, such as 50 000 IU orally once a week for eight weeks or 300 000 IU intramuscularly every three months 302.
In patients with hypoparathyroidism, the above doses of vitamin D are ineffective because parathyroid hormone (PTH) is needed for conversion to 1,25-dihydroxyvitamin D 236. Thus calcitriol or alfacalcidol is needed. Typical starting doses are 0.5 mcg of calcitriol or 1 mcg of alfacalcidol each day, with doses being increased every four to seven days to achieve a serum calcium in the lower part of the reference range 236. Once a stable calcium concentration is achieved, concentrations should be monitored every three to six months. The main long term risk is the development of nephrocalcinosis as a result of hypercalciuria. Urinary calcium excretion should be monitored annually and if high the dose of vitamin D may need to be reduced.
In hypocalcemia caused by malabsorption, the underlying problem should be treated if possible. Patients with celiac disease should receive calcium and vitamin D orally and should comply with a gluten free diet 303.
Medicines to help treat osteoporosis
Biophosphonates
Biophosphonates helps reduce your risk of breaks and fractures. Biophosphonates also increases bone density. Biophosphonates comes in oral (pill) form or intravenous (IV or injection) form. Side effects can include nausea and stomach pain. You may have heartburn-like symptoms with irritation of your esophagus (the tube that connects your mouth and stomach). These are less likely to occur if the medicine is taken properly. Intravenous forms of bisphosphonates don’t cause stomach upset but can cause fever, headache and muscle aches.
A very rare complication of bisphosphonates is a break or crack in the middle of the thighbone. A second rare complication is delayed healing of the jawbone (osteonecrosis of the jaw). This can occur after an invasive dental procedure, such as removing a tooth.
Some people cannot take biophosphonates. This includes people who have kidney disease or low levels of calcium in their blood, and women who are pregnant or nursing.
For both men and women at increased risk of fracture, the most widely prescribed osteoporosis medications are bisphosphonates. Examples include:
- Alendronate (Binosto, Fosamax). Alendronate is used to help prevent and treat osteoporosis. Alendronate helps reduce your risk of fractures by decreasing the rate of bone loss. Alendronate is available in pill form. Alendronate most common side effect is an upset stomach.
- Ibandronate (Boniva). Ibandronate helps to slow bone loss and increase bone density. It is available as a pill or injection. You have 2 options for the pill. You can take it daily or monthly. For the injection, your doctor or nurse will give you a shot every 3 months. Side effects may include lower back or side pain, shortness of breath, tightness in your chest, and bloody or cloudy urine.
- Risedronate (Actonel, Atelvia). Risedronate is used to help prevent and treat osteoporosis. Risedronate helps reduce your risk of fractures by decreasing the rate of bone loss. Risedronate is available in pill form. Risedronate most common side effect is an upset stomach.
- Zoledronic acid (Reclast, Zometa). Zoledronic acid is given through IV once a year.
Denosumab
Denosumab (Prolia, Xgeva) is used when other drugs don’t work or if you can’t tolerate other treatment options. Denosumab increases your bone density. It can be used by both women and men. Denosumab is delivered via a shot under the skin every six months. Side effects can include lower calcium levels, skin rash, or pain in the arms and legs.
Compared with bisphosphonates, denosumab produces similar or better bone density results and reduces the chance of all types of fractures.
Similar to bisphosphonates, denosumab has the same rare complication of causing breaks or cracks in the middle of the thighbone and osteonecrosis of the jaw. If you take denosumab, you might need to continue to do so indefinitely. Recent research indicates there could be a high risk of spinal column fractures after stopping the drug.
Estrogen, especially when started soon after menopause, can help maintain bone density. However, estrogen therapy can increase the risk of breast cancer and blood clots, which can cause strokes. Therefore, estrogen is typically used for bone health in younger women or in women whose menopausal symptoms also require treatment.
Raloxifene (Evista) mimics estrogen’s beneficial effects on bone density in postmenopausal women, without some of the risks associated with estrogen. Taking Raloxifene can reduce the risk of some types of breast cancer. Hot flashes are a possible side effect. Raloxifene also may increase your risk of blood clots.
In men, osteoporosis might be linked with a gradual age-related decline in testosterone levels. Testosterone replacement therapy can help improve symptoms of low testosterone, but osteoporosis medications have been better studied in men to treat osteoporosis and thus are recommended alone or in addition to testosterone.
Bone-building medications
If you have severe osteoporosis or if the more common treatments for osteoporosis don’t work well enough, your doctor might suggest trying:
- Teriparatide (Bonsity, Forteo). Teriparatide is a synthetic form of parathyroid hormone (PTH) and stimulates new bone growth. Both women and men can use it. Teriparatide is given by daily injection under the skin for up to two years. You inject it in your thigh or stomach once a day. Common side effects are nausea, stomach pain, headache, muscle weakness, fatigue, and loss of appetite.
- Abaloparatide (Tymlos) is another drug similar to parathyroid hormone (PTH). This drug can be taken for only two years.
- Romosozumab (Evenity). This is the newest bone-building medication to treat osteoporosis. It is given as an injection every month at your doctor’s office and is limited to one year of treatment.
After you stop taking any of these bone-building medications, you generally will need to take another osteoporosis drug to maintain the new bone growth.
Calcitonin
Calcitonin is a hormone that helps slow down bone loss. It is available as an injection or nasal spray. Side effects of Calcitonin injection include diarrhea, stomach pain, nausea, and vomiting. Side effects of Calcitonin nose spray include headache and irritation of your nose lining.
Calcium deficiency prognosis
The overall prognosis of hypocalcemia is good as it can be easily corrected 230. In rare cases, patients with complete parathyroidectomy require very high doses of calcium and vitamin D supplements to maintain calcium in the normal range 230. Patients who have undergone gastric bypass surgery usually have malabsorption and may also require very high doses of calcium and vitamin D to correct hypocalcemia 230.
Bone fractures, particularly in the spine or hip, are the most serious complications of osteoporosis 304. Osteoporotic fractures often lead to a reduced quality of life and premature death 305. Hip fractures are the most serious of all osteoporotic fractures since 12-20% of women suffering from a hip fracture die during the following 2 years, and 50% of those surviving lose their independence and require long-term nursing care 305. The risk of death after hip fracture is higher in men than women. Fractures at other sites can also be debilitating.
In some cases, spinal fractures can occur even if you haven’t fallen. The bones that make up your spine (vertebrae) can weaken to the point of collapsing, which can result in back pain, lost height and a hunched forward posture. Multiple and/or severe vertebral fractures can lead to loss of height and abnormal bending of the upper and middle back (thoracic spine), reducing lung function and affecting digestion. Vertebral and non-hip, non-vertebral fractures are also associated with premature death.
Can too much calcium be harmful?
Too much calcium in your blood can weaken your bones, create kidney stones, cause constipation and interfere with how your heart and brain work. It might also interfere with the body’s ability to absorb iron and zinc, but this effect is not well established. In adults, too much calcium or high levels of calcium in the blood (hypercalcemia) from dietary supplements but not food might increase the risk of kidney stones. Approximately 90% of cases of too much calcium or high levels of calcium in the blood (hypercalcemia) are caused by hyperparathyroidism or cancer 306, 307, 308.
The normal serum calcium ranges from 8.8 mg/dL to 10.8 mg/dL.
Hypercalcemia can be classified into 307.:
- Mild hypercalcemia: 10.5 to 11.9 mg/dL
- Moderate hypercalcemia: 12.0 to 13.9 mg/dL
- Hypercalecemic crisis: 14.0 to 16.0 mg/dL
Hypercalcemia or high levels of calcium in the blood has been initially reported with the consumption of large quantities of calcium supplements in combination with antacids, particularly in the days when peptic ulcers were treated with large quantities of milk, calcium carbonate (antacid), and sodium bicarbonate (absorbable alkali). This condition is termed calcium-alkali syndrome also known as milk-alkali syndrome and has been associated with calcium supplement levels from 1.5 to 16.5 g/day for 2 days to 30 years. Since the treatment for peptic ulcers has evolved and because of the widespread use of over-the-counter calcium supplements, the demographic of calcium-alkali syndrome has changed in that those at greater risk are now postmenopausal women, pregnant women, transplant recipients, patients with bulimia, and patients on dialysis, rather than men with peptic ulcers 309. Supplementation with calcium (0.6 g/day-2 g/day for two to five years) has been associated with a higher risk of adverse gastrointestinal events like constipation, cramping, bloating, pain, diarrhea 310. Mild hypercalcemia may be without symptoms or may result in loss of appetite, nausea, vomiting, constipation, abdominal pain, fatigue, frequent urination (polyuria), and hypertension 311. More severe hypercalcemia may result in confusion, delirium, coma, and if not treated, death 7.
Some studies show that people who consume high amounts of calcium might have increased risks of prostate cancer and heart disease, but more research is needed to understand these possible links 312, 313, 314.
Most people do not get amounts above the upper limits from food alone; excess intakes usually come from the use of calcium supplements. Surveys show that some older women in the United States probably get amounts somewhat above the upper limit since the use of calcium supplements is common among these women.
Health Risks from Excessive Calcium
Excessively high levels of calcium in the blood known as hypercalcemia can cause renal insufficiency, vascular and soft tissue calcification, hypercalciuria (high levels of calcium in the urine) and kidney stones 14. Although very high calcium intakes have the potential to cause hypercalcemia 315, it is most commonly associated with primary hyperparathyroidism or malignancy 14.
High calcium intake can cause constipation. It might also interfere with the absorption of iron and zinc, though this effect is not well established 14. High intake of calcium from supplements, but not foods, has been associated with increased risk of kidney stones 14, 316, 317. Some evidence links higher calcium intake with increased risk of prostate cancer, but this effect is not well understood, in part because it is challenging to separate the potential effect of dairy products from that of calcium 14. Some studies also link high calcium intake, particularly from supplements, with increased risk of cardiovascular disease 315, 318, 319, 320, 321, 322.
The Tolerable Upper Intake Levels (ULs) for calcium established by the Food and Nutrition Board are listed in Table 5 in milligrams (mg) per day. Getting too much calcium from foods is rare; excess intakes are more likely to be caused by the use of calcium supplements. NHANES data from 2003–2006 indicate that approximately 5% of women older than 50 years have estimated total calcium intakes (from foods and supplements) that exceed the UL by about 300–365 mg.
Table 5. Tolerable Upper Intake Levels (ULs) for Calcium
| Age | Male | Female | Pregnant | Lactating |
|---|---|---|---|---|
| 0-6 months | 1,000 mg | 1,000 mg | ||
| 7–12 months | 1,500 mg | 1,500 mg | ||
| 1–8 years | 2,500 mg | 2,500 mg | ||
| 9–18 years | 3,000 mg | 3,000 mg | 3,000 mg | 3,000 mg |
| 19–50 years | 2,500 mg | 2,500 mg | 2,500 mg | 2,500 mg |
| 51+ years | 2,000 mg | 2,000 mg |
Risk for prostate cancer
The analysis of a prospective cohort study (2,268 men followed for nearly 25 years) conducted in Iceland, a country with a high incidence of prostate cancer, found a positive association between the consumption of milk (at least once daily) during adolescence and developing prostate cancer later in life 323. Another large prospective cohort study in the US followed 21,660 male physicians for 28 years and found that men with daily skim or low-fat milk intake of at least 237 mL (8 oz) had a higher risk of developing prostate cancer compared to occasional consumers 324. The risk of low-grade, early-stage prostate cancer was associated with higher intake of skim milk, and the risk of developing fatal prostate cancer was linked to the regular consumption of whole milk 324. In a cohort of 3,918 male health professionals diagnosed with prostate cancer, 229 men died of prostate cancer and 69 developed metastasized prostate cancer during a median follow-up of 7.6 years 325. The risk of prostate cancer death was found to be increased in men with high (>4 servings/week) versus low (≤3 servings/month) intakes of whole milk. Yet, no increase in risk of prostate cancer-related mortality was associated with consumption of skim and low-fat milk, total milk, low-fat dairy products, full-fat dairy products, or total dairy products 325. A recent meta-analysis of 32 prospective cohort studies found high versus low intakes of total dairy product (15 studies), total milk (15 studies), whole milk (6 studies), low-fat milk (5 studies), cheese (11 studies), and dairy calcium (7 studies) to be associated with modest, yet significant, increases in the risk of developing prostate cancer 326. However, there was no increase in prostate cancer risk with nondairy calcium (4 studies) and calcium from supplements (8 studies). Moreover, high dairy intakes were not linked to fatal prostate cancer 326.
There is some evidence to suggest that milk consumption may result in higher circulating concentrations of insulin-like growth factor-1 (IGF-1), a protein known to regulate cell proliferation 327. Circulating insulin-like growth factor-1 (IGF-1) concentrations have been positively correlated to the risk of developing prostate cancer in a recent meta-analysis of observational studies 328. Milk-borne insulin-like growth factor-1 (IGF-1), as well as dairy proteins and calcium, may contribute to increasing circulating IGF-1 in milk consumers 327. In the large EPIC study, which examined the consumption of dairy products in relation to cancer in 142,520 men, the risk of prostate cancer was found to be significantly higher in those in the top versusbottom quintile of both protein and calcium intakes from dairy foods 108. Another mechanism underlying the potential relationship between calcium intake and prostate cancer proposed that high levels of dietary calcium may lower circulating concentrations of 1,25-dihydroxyvitamin D, the active form of vitamin D, thereby suppressing vitamin D-mediated cell differentiation 329. However, studies to date have provided little evidence to suggest that vitamin D status can modify the association between dairy calcium and risk of prostate cancer development and progression 330, 331, 332.
In a multicenter, double-blind, placebo-controlled trial, 672 healthy men (mean age of 61.8 years) were randomized to daily calcium supplementation (1,200 mg) for four years. While no increase in the risk for prostate cancer has been reported during a 10.3-year follow-up, calcium supplementation resulted in a significant risk reduction in the period spanning from two years after treatment started to two years after treatment ended 333. In a review of the literature published in 2009, the US Agency for Healthcare Research and Quality indicated that not all epidemiological studies found an association between calcium intake and prostate cancer 334. The review reported that 6 out of 11 observational studies failed to find statistically significant positive associations between prostate cancer and calcium intake. Yet, in five studies, daily intakes of 921 to 2,000 mg of calcium were found to be associated with an increased risk of developing prostate cancer when compared to intakes ranging from 455 to 1,000 mg/day 334. Inconsistencies among studies suggest complex interactions between the risk factors for prostate cancer, as well as reflect the difficulties of assessing the effect of calcium intake in free-living individuals. For example, the fact that individuals with higher dairy and/or calcium intakes were found to be more likely to be engaged in healthy lifestyles or more likely to seek medical attention can mitigate the statistical significance of an association with prostate cancer risk 335. Until the relationship between calcium and prostate cancer is clarified, it is reasonable for men to consume a total of 1,000 to 1,200 mg/day of calcium (diet and supplements combined), which is recommended by the Food and Nutrition Board of the Institute of Medicine 20.
What causes high blood calcium?
The most common cause of high blood calcium or hypercalcemia is excess PTH released by the parathyroid glands (primary hyperparathyroidism). This excess PTH occurs due to:
- An enlargement of one or more of the parathyroid glands.
- A growth on one of the parathyroid glands. Most of the time, these growths are benign (not a cancer).
This, in turn, causes the bones to release too much calcium into the blood. Women over the age of 50 are more likely than others to have primary hyperparathyroidism.
Certain types of cancer, most often breast cancer, lung cancer, or multiple myeloma (a type of blood cancer), can also cause high blood calcium. This usually occurs late in the course of cancer.
Less common causes of hypercalcemia include these health problems:
- Some types of infectious diseases, such as tuberculosis (TB) and sarcoidosis.
- Some types of autoimmune disease, such as sarcoidosis
- Hormone disorders, such as overactive thyroid (hyperthyroidism)
- A genetic condition called familial hypocalciuric hypercalcemia
- Chronic kidney disease or kidney failure.
Other infrequent causes of high blood calcium include:
- Some medicines, such as lithium (to treat psychiatric illness) or rarely, thiazide diuretics (water pills)
- Intake of very large amounts of calcium or large amounts of milk plus antacids. This is called milk-alkali syndrome. It most often occurs when a person is taking more than 2000 milligrams of calcium bicarbonate supplements a day along with high doses of Vitamin D.
- Intake of too much vitamin D (hypervitaminosis D) or vitamin A (hypervitaminosis A)
- Immobility–being confined to bed for at least several weeks– combined with some bone diseases, such as Paget’s disease
- Tube feeding or being fed through a vein
- Severe dehydration
High blood calcium signs and symptoms
Symptoms due to high calcium level (hypercalcemia) may vary, depending on the cause and how long the problem has been present. You might not have signs or symptoms if your hypercalcemia is mild. More-severe cases produce signs and symptoms related to the parts of your body affected by the high calcium levels in your blood.
High calcium level (hypercalcemia) affects nearly every organ system in your body, but it particularly affects the central nervous system (brain and spinal cord) and the kidneys.
Hypercalcemia can interfere with how your brain works, resulting in confusion, lethargy and fatigue. It can also cause depression. Central nervous system effects of high calcium level (hypercalcemia) include the following 336:
- Changes in how your brain works, such as feeling tired or fatigued or confused
- Muscle weakness or twitches
- Confusion
- Stupor
- Coma
Excess calcium makes your kidneys work harder to filter it. This can cause excessive thirst and frequent urination. Kidney effects include the following 336:
- Excessive urination (polyuria)
- Nephrogenic diabetes insipidus (increased thirst)
- Frequent urination at night (nocturia)
- Dehydration
- Kidney stones
- Distal renal tubular acidosis
- Acute kidney injury
- Chronic kidney disease
Gastrointestinal effects include the following 336:
- Constipation
- Nausea and vomiting
- Anorexia
- Pancreatitis
- Gastric ulcer
Excess calcium effects on bones and muscles.
- In most cases, the excess calcium in your blood was leached from your bones, which weakens them resulting in fragile bones that break more easily. This can cause bone pain and muscle weakness.
Heart. Rarely, severe hypercalcemia can interfere with your heart function, causing palpitations and fainting, indications of cardiac arrhythmia, and other heart problems. Heart effects include 336:
- Fainting (syncope)
- Arrhythmias
- Hypertension
- Bradycardia
- Shortening of QT interval
Calcium has a positive inotropic effect. Hypercalcemia also causes hypertension, presumably from renal dysfunction and direct vasoconstriction.
ECG features of hypercalcemia include 307:
- T wave flattening or inversion
- Mild prolongation of the QRS and PR intervals
- ST-elevation
- Presence of J wave at the end of the QRS complex
High blood calcium complications
High calcium level (hypercalcemia) complications can include:
- Osteoporosis. If your bones continue to release calcium into your blood, you can develop the bone-thinning disease osteoporosis, which could lead to bone fractures, spinal column curvature and loss of height.
- Kidney stones. If your urine contains too much calcium, crystals might form in your kidneys. Over time, the crystals can combine to form kidney stones. Passing a stone can be extremely painful.
- Kidney failure. Severe hypercalcemia can damage your kidneys, limiting their ability to cleanse the blood and eliminate fluid.
- Nervous system problems. Severe hypercalcemia can lead to confusion, dementia and coma, which can be fatal.
- Abnormal heart rhythm (arrhythmia). Hypercalcemia can affect the electrical impulses that regulate your heartbeat, causing your heart to beat irregularly.
High blood calcium diagnosis
Because hypercalcemia can cause few, if any, signs or symptoms, you might not know you have the disorder until routine blood tests reveal a high level of blood calcium. Blood tests can also show whether your parathyroid hormone (PTH) level is high, indicating that you have hyperparathyroidism.
People with kidney stones should have tests to evaluate for hypercalcemia.
- Serum calcium
- Serum PTH
- Serum PTHrP (PTH-related protein)
- Serum vitamin D level
- Urine calcium
To determine if your hypercalcemia is caused by a disease such as cancer or sarcoidosis, your doctor might recommend imaging tests of your bones or lungs.
How is high blood calcium treated?
Talk with your health care provider about the best treatment for your condition. Your treatment will depend on the cause of your high blood calcium. In general, the best treatment is to take care of the condition that is causing the high blood calcium. For instance, people with primary hyperparathyroidism who have symptoms usually have surgery to remove the abnormal parathyroid gland. This will cure the hypercalcemia. In many cases, only one of a person’s four parathyroid glands is affected. A special scanning test uses an injection of a small dose of radioactive material to pinpoint the gland or glands that aren’t working properly.
Until the underlying problem is resolved, treatment may include medicines to improve blood calcium levels. When blood calcium is dangerously high, people may need treatment in a hospital to return their blood calcium to a safe level.
Severe hypercalcemia that causes symptoms and requires a hospital stay may be treated with the following:
- Fluids through a vein — This is the most important therapy. Extremely high calcium levels can be a medical emergency. You might need hospitalization for treatment with IV fluids and diuretics to promptly lower the calcium level to prevent heart rhythm problems or damage to the nervous system.
- Calcitonin (Miacalcin). This hormone from salmon controls calcium levels in the blood. Mild nausea might be a side effect.
- Calcimimetics. This type of drug can help control overactive parathyroid glands. Cinacalcet (Sensipar) has been approved for managing hypercalcemia.
- Dialysis, if kidney damage is involved.
- Diuretic medicine, such as furosemide.
- Drugs that stop bone breakdown and absorption by the body (bisphosphonates). Intravenous osteoporosis drugs, which can quickly lower calcium levels, are often used to treat hypercalcemia due to cancer. Risks associated with this treatment include breakdown (osteonecrosis) of the jaw and certain types of thigh fractures.
- Denosumab (Prolia, Xgeva). This drug is often used to treat people with cancer-caused hypercalcemia who don’t respond well to bisphosphonates.
- Glucocorticoids (steroids). If your hypercalcemia is caused by high levels of vitamin D, short-term use of steroid pills such as prednisone are usually helpful.
You might not need any treatment if your blood calcium is only slightly high or you have not developed any health problems. Instead, your health care provider will continue to check your condition over time by monitoring your bones and kidneys over time to be sure they remain healthy.
High blood calcium prognosis
Hypercalcemia, when it occurs following a benign disorder, has a good prognosis but when the cause is secondary to a cancer the prognosis is poor 307. Patients with hypercalcemia associated with cancer are often symptomatic and need frequent hospitalizations 307. When hypercalcemia is due to ectopic production of PTH related protein, most patients are dead within a few months. The osteolytic metastatic lesions cause fractures, nerve compression, and paralysis.
References- Gilman, A. G., L. S. Goodman, and A. Gilman. (eds.). Goodman and Gilman’s The Pharmacological Basis of Therapeutics. 6th ed. New York: Macmillan Publishing Co., Inc. 1980., p. 1528
- Harvard University, Harvard School of Public Health. Calcium: What’s Best for Your Bones and Health ? https://www.hsph.harvard.edu/nutritionsource/calcium-full-story/
- Institute of Medicine (US) Committee to Review Dietary Reference Intakes for Vitamin D and Calcium; Ross AC, Taylor CL, Yaktine AL, et al., editors. Dietary Reference Intakes for Calcium and Vitamin D. Washington (DC): National Academies Press (US); 2011. Available from: https://www.ncbi.nlm.nih.gov/books/NBK56070/ doi: 10.17226/13050
- Weaver CM. Calcium. In: Marriott BP, Birt DF, Stallings VA, Yates AA, eds. Present Knowledge in Nutrition. 11th ed. Cambridge, Massachusetts: Wiley-Blackwell; 2020:321-48.
- Wawrzyniak N, Suliburska J. Nutritional and health factors affecting the bioavailability of calcium: a narrative review. Nutr Rev. 2021 Nov 10;79(12):1307-1320. doi: 10.1093/nutrit/nuaa138
- Calcium. https://lpi.oregonstate.edu/mic/minerals/calcium
- Weaver CM. Calcium. In: Erdman JJ, Macdonald I, Zeisel S, eds. Present Knowledge in Nutrition. 10th ed: John Wiley & Sons, Inc.; 2012:434-446.
- Calcium. https://ods.od.nih.gov/factsheets/Calcium-HealthProfessional
- Calcium. https://ods.od.nih.gov/factsheets/Calcium-Consumer
- Fairweather-Tait SJ, Teucher B. Iron and calcium bioavailability of fortified foods and dietary supplements. Nutr Rev. 2002 Nov;60(11):360-7. doi: 10.1301/00296640260385801
- Song L. Calcium and Bone Metabolism Indices. Adv Clin Chem. 2017;82:1-46. doi: 10.1016/bs.acc.2017.06.005
- Weaver CM, Heaney RP. Calcium. In: Ross AC, Caballero B, Cousins RJ, Tucker KL, Ziegler TR, eds. Modern Nutrition in Health and Disease. 11th ed. Baltimore, MD: Lippincott Williams & Wilkins; 2014:133-49.
- Bronner F. Mechanisms of intestinal calcium absorption. Journal of Cellular Biochemistry 2003; 88(2):387–393. https://www.ncbi.nlm.nih.gov/pubmed/12520541
- Committee to Review Dietary Reference Intakes for Vitamin D and Calcium, Food and Nutrition Board, Institute of Medicine. Dietary Reference Intakes for Calcium and Vitamin D. Washington, DC: National Academy Press, 2010.
- Wigertz K, Palacios C, Jackman LA, Martin BR, McCabe LD, McCabe GP, Peacock M, Pratt JH, Weaver CM. Racial differences in calcium retention in response to dietary salt in adolescent girls. Am J Clin Nutr. 2005 Apr;81(4):845-50. doi: 10.1093/ajcn/81.4.845
- Frassetto LA, Morris RC Jr, Sellmeyer DE, Sebastian A. Adverse effects of sodium chloride on bone in the aging human population resulting from habitual consumption of typical American diets. J Nutr. 2008 Feb;138(2):419S-422S. doi: 10.1093/jn/138.2.419S
- Devine A, Criddle RA, Dick IM, Kerr DA, Prince RL. A longitudinal study of the effect of sodium and calcium intakes on regional bone density in postmenopausal women. Am J Clin Nutr. 1995 Oct;62(4):740-5. doi: 10.1093/ajcn/62.4.740
- Carbone LD, Barrow KD, Bush AJ, Boatright MD, Michelson JA, Pitts KA, Pintea VN, Kang AH, Watsky MA. Effects of a low sodium diet on bone metabolism. J Bone Miner Metab. 2005;23(6):506-13. doi: 10.1007/s00774-005-0621-8
- Sellmeyer DE, Schloetter M, Sebastian A. Potassium citrate prevents increased urine calcium excretion and bone resorption induced by a high sodium chloride diet. J Clin Endocrinol Metab. 2002 May;87(5):2008-12. doi: 10.1210/jcem.87.5.8470
- Food and Nutrition Board, Institute of Medicine. Dietary Reference Intakes for Calcium and Vitamin D. Washington, D.C.; 2011. https://nap.nationalacademies.org/read/13050/chapter/1
- Fulgoni VL 3rd. Current protein intake in America: analysis of the National Health and Nutrition Examination Survey, 2003-2004. Am J Clin Nutr. 2008 May;87(5):1554S-1557S. doi: 10.1093/ajcn/87.5.1554S
- Ince BA, Anderson EJ, Neer RM. Lowering dietary protein to U.S. Recommended dietary allowance levels reduces urinary calcium excretion and bone resorption in young women. J Clin Endocrinol Metab. 2004 Aug;89(8):3801-7. doi: 10.1210/jc.2003-032016
- Calvez J, Poupin N, Chesneau C, Lassale C, Tomé D. Protein intake, calcium balance and health consequences. Eur J Clin Nutr. 2012 Mar;66(3):281-95. doi: 10.1038/ejcn.2011.196
- Kerstetter JE, O’Brien KO, Caseria DM, Wall DE, Insogna KL. The impact of dietary protein on calcium absorption and kinetic measures of bone turnover in women. J Clin Endocrinol Metab. 2005 Jan;90(1):26-31. doi: 10.1210/jc.2004-0179
- Darling AL, Millward DJ, Torgerson DJ, Hewitt CE, Lanham-New SA. Dietary protein and bone health: a systematic review and meta-analysis. Am J Clin Nutr. 2009 Dec;90(6):1674-92. doi: 10.3945/ajcn.2009.27799
- Grimm M, Müller A, Hein G, Fünfstück R, Jahreis G. High phosphorus intake only slightly affects serum minerals, urinary pyridinium crosslinks and renal function in young women. Eur J Clin Nutr. 2001 Mar;55(3):153-61. doi: 10.1038/sj.ejcn.1601131
- Kemi VE, Kärkkäinen MU, Rita HJ, Laaksonen MM, Outila TA, Lamberg-Allardt CJ. Low calcium:phosphorus ratio in habitual diets affects serum parathyroid hormone concentration and calcium metabolism in healthy women with adequate calcium intake. Br J Nutr. 2010 Feb;103(4):561-8. doi: 10.1017/S0007114509992121
- Heaney RP. Phosphorus. In: Erdman JJ, Macdonald I, Zeisel S, eds. Present Knowledge in Nutrition. 10th ed. Ames: John Wiley & Sons, Inc.; 2012:447-458.
- Calvo MS, Moshfegh AJ, Tucker KL. Assessing the health impact of phosphorus in the food supply: issues and considerations. Adv Nutr. 2014 Jan 1;5(1):104-13. doi: 10.3945/an.113.004861
- Heaney RP, Rafferty K. Carbonated beverages and urinary calcium excretion. Am J Clin Nutr. 2001 Sep;74(3):343-7. doi: 10.1093/ajcn/74.3.343
- Ribeiro-Alves MA, Trugo LC, Donangelo CM. Use of oral contraceptives blunts the calciuric effect of caffeine in young adult women. J Nutr. 2003 Feb;133(2):393-8. doi: 10.1093/jn/133.2.393
- Barger-Lux MJ, Heaney RP, Stegman MR. Effects of moderate caffeine intake on the calcium economy of premenopausal women. Am J Clin Nutr. 1990 Oct;52(4):722-5. doi: 10.1093/ajcn/52.4.722. Erratum in: Am J Clin Nutr 1991 Jan;53(1):182.
- Wikoff D, Welsh BT, Henderson R, Brorby GP, Britt J, Myers E, Goldberger J, Lieberman HR, O’Brien C, Peck J, Tenenbein M, Weaver C, Harvey S, Urban J, Doepker C. Systematic review of the potential adverse effects of caffeine consumption in healthy adults, pregnant women, adolescents, and children. Food Chem Toxicol. 2017 Nov;109(Pt 1):585-648. doi: 10.1016/j.fct.2017.04.002
- Wesseling-Perry K, Wang H, Elashoff R, Gales B, Jüppner H, Salusky IB. Lack of FGF23 response to acute changes in serum calcium and PTH in humans. J Clin Endocrinol Metab. 2014 Oct;99(10):E1951-6. doi: 10.1210/jc.2014-2125
- Clapham DE. Calcium signaling. Cell. 2007 Dec 14;131(6):1047-58. doi: 10.1016/j.cell.2007.11.028
- The National Institute of Health, Office of Dietary Supplements. Calcium. https://ods.od.nih.gov/factsheets/Calcium-HealthProfessional
- Straub DA. Calcium supplementation in clinical practice: a review of forms, doses, and indications. Nutr Clin Pract. 2007 Jun;22(3):286-96. doi: 10.1177/0115426507022003286
- Andon MB, Peacock M, Kanerva RL, De Castro JA. Calcium absorption from apple and orange juice fortified with calcium citrate malate (CCM). J Am Coll Nutr. 1996 Jun;15(3):313-6. doi: 10.1080/07315724.1996.10718604
- Office of Dietary Supplements, National Institutes of Health. https://dsld.od.nih.gov
- Institute of Medicine SCotSEoDR, Intakes,. Dietary Reference Intakes for Calcium, Phosphorus, Magnesium, Vitamin D, and Fluoride. Washington, DC: National Academies Press; 1997.
- Heaney RP, Dowell MS, Barger-Lux MJ. Absorption of calcium as the carbonate and citrate salts, with some observations on method. Osteoporos Int. 1999;9(1):19-23. doi: 10.1007/s001980050111
- Protective effects of dietary lactulose and calcium phosphate against Salmonella infection. Bovee-Oudenhoven I, Van der Meer R. Scand J Gastroenterol Suppl. 1997; 222:112-4. https://www.ncbi.nlm.nih.gov/pubmed/9145462/
- Ditscheid B, Keller S, Jahreis G. Cholesterol metabolism is affected by calcium phosphate supplementation in humans. J Nutr. 2005;135:1678–1682. https://www.ncbi.nlm.nih.gov/pubmed/15987849
- Govers MJ, Van der Meer R. Effects of dietary calcium and phosphate on the intestinal interactions between calcium, phosphate, fatty acids, and bile acids. Gut. 1993;34:365–370. doi: 10.1136/gut.34.3.365. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1374143/
- Trautvetter U, Ditscheid B, Kiehntopf M, Jahreis G. A combination of calcium phosphate and probiotics beneficially influences intestinal lactobacilli and cholesterol metabolism in humans. Clin Nutr. 2012;31:230–237. doi: 10.1016/j.clnu.2011.09.013. https://www.ncbi.nlm.nih.gov/pubmed/22019281
- Trautvetter U, Kiehntopf M, Jahreis G. Postprandial effects of calcium phosphate supplementation on plasma concentration-double-blind, placebo-controlled cross-over human study. Nutr J. 2013;12:30. doi: 10.1186/1475-2891-12-30. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3599792/
- Chapuy MC, Arlot ME, Duboeuf F, Brun J, Crouzet B, Arnaud S, Delmas PD, Meunier PJ. Vitamin D3 and calcium to prevent hip fractures in the elderly women. N Engl J Med. 1992;327:1637–1642. doi: 10.1056/NEJM199212033272305. https://www.ncbi.nlm.nih.gov/pubmed/1331788
- Nutr J. 2014; 13: 6. Published online 2014 Jan 17. doi: 10.1186/1475-2891-13-6. Effect of calcium phosphate and vitamin D3 supplementation on bone remodelling and metabolism of calcium, phosphorus, magnesium and iron. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3898568/
- Cochrane Review 6 October 2015. 10.1002/14651858.CD010057.pub2. Citrate salts for preventing and treating calcium containing kidney stones in adults. http://onlinelibrary.wiley.com/doi/10.1002/14651858.CD010057.pub2/abstract
- Cochrane Review 16 February 2011. Phosphate binders for preventing and treating bone disease in chronic kidney disease patients. http://onlinelibrary.wiley.com/doi/10.1002/14651858.CD006023.pub2/abstract
- Nephrol Dial Transplant (2009) 24 (10): 3168-3174. The effects of calcium-based versus non-calcium-based phosphate binders on mortality among patients with chronic kidney disease: a meta-analysis. https://academic.oup.com/ndt/article/24/10/3168/1818941/The-effects-of-calcium-based-versus-non-calcium
- National Cancer Institute. Calcium and Cancer Prevention. https://www.cancer.gov/about-cancer/causes-prevention/risk/diet/calcium-fact-sheet
- Caruso JB, Patel RM, Julka K, Parish DC. Health-behavior induced disease: Return of the milk-alkali syndrome. Journal of General Internal Medicine 2007; 22(7):1053–1055. https://www.ncbi.nlm.nih.gov/pubmed/17483976
- Cooke AM. Alkalosis occurring in the alkaline treatment of peptic ulcers. Quart J Med. 1932;25:527
- Life-threatening milk-alkali syndrome resulting from antacid ingestion during pregnancy. Gordon MV, McMahon LP, Hamblin PS. Med J Aust. 2005 Apr 4; 182(7):350-1. https://www.ncbi.nlm.nih.gov/pubmed/15804228/
- Wactawski-Wende J, Kotchen JM, Anderson GL, et al. Calcium plus vitamin D supplementation and the risk of colorectal cancer. New England Journal of Medicine 2006; 354(7):684–696. https://www.ncbi.nlm.nih.gov/pubmed/16481636
- Calcium. In: Dietary Reference Intakes for Calcium, Phosphorus, Magnesium, Vitamin D and Fluoride. National Academy Press, Washington, DC. https://www.nap.edu/read/5776/chapter/6
- Gordon MV, Hamblin PS, McMahon LP. Life-threatening milk-alkali syndrome resulting from antacid ingestion during pregnancy. Medical Journal of Australia 2005; 182(7):350–351. https://www.ncbi.nlm.nih.gov/pubmed/15804228
- Beall DP, Henslee HB, Webb HR, Scofield RH. Milk-alkali syndrome: A historical review and description of the modern version of the syndrome. American Journal of the Medical Sciences 2006; 331(5):233–242. https://www.ncbi.nlm.nih.gov/pubmed/16702792
- Wood RJ, Zheng JJ. High dietary calcium intakes reduce zinc absorption and balance in humans. Am J Clin Nutr. 1997 Jun;65(6):1803-9. doi: 10.1093/ajcn/65.6.1803
- Borel P, Desmarchelier C, Dumont U, Halimi C, Lairon D, Page D, Sébédio JL, Buisson C, Buffière C, Rémond D. Dietary calcium impairs tomato lycopene bioavailability in healthy humans. Br J Nutr. 2016 Dec;116(12):2091-2096. doi: 10.1017/S0007114516004335
- Song I, Borland J, Arya N, Wynne B, Piscitelli S. Pharmacokinetics of dolutegravir when administered with mineral supplements in healthy adult subjects. J Clin Pharmacol. 2015 May;55(5):490-6. doi: 10.1002/jcph.439
- Jalloh MA, Gregory PJ, Hein D, Risoldi Cochrane Z, Rodriguez A. Dietary supplement interactions with antiretrovirals: a systematic review. Int J STD AIDS. 2017 Jan;28(1):4-15. doi: 10.1177/0956462416671087
- https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/213983s000lbl.pdf
- https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/211994s000lbl.pdf
- Morini E, Catalano A, Lasco A, Morabito N, Benvenga S. L-thyroxine malabsorption due to calcium carbonate impairs blood pressure, total cholesterolemia, and fasting glycemia. Endocrine. 2019 May;64(2):284-292. doi: 10.1007/s12020-018-1798-7
- Singh N, Singh PN, Hershman JM. Effect of calcium carbonate on the absorption of levothyroxine. JAMA. 2000 Jun 7;283(21):2822-5. doi: 10.1001/jama.283.21.2822
- Schneyer CR. Calcium carbonate and reduction of levothyroxine efficacy. JAMA. 1998 Mar 11;279(10):750. doi: 10.1001/jama.279.10.750-b
- https://www.accessdata.fda.gov/drugsatfda_docs/label/2017/021342s023lbl.pdf
- Jones BJ, Twomey PJ. Requesting patterns for serum calcium concentration in patients on long-term lithium therapy. Int J Clin Pract. 2009 Jan;63(1):170-2. doi: 10.1111/j.1742-1241.2008.01918.x
- Pletz MW, Petzold P, Allen A, Burkhardt O, Lode H. Effect of calcium carbonate on bioavailability of orally administered gemifloxacin. Antimicrob Agents Chemother. 2003 Jul;47(7):2158-60. doi: 10.1128/AAC.47.7.2158-2160.2003
- Kays MB, Overholser BR, Mueller BA, Moe SM, Sowinski KM. Effects of sevelamer hydrochloride and calcium acetate on the oral bioavailability of ciprofloxacin. Am J Kidney Dis. 2003 Dec;42(6):1253-9. doi: 10.1053/j.ajkd.2003.08.027
- National Institutes of Health, Office of Dietary Supplements. Calcium. https://ods.od.nih.gov/factsheets/Calcium-HealthProfessional/
- http://www.ncbi.nlm.nih.gov/pubmedhealth/PMHT0009413/
- Sci Rep. 2016; 6: 21761. Published online 2016 Feb 22. doi: 10.1038/srep21761. Nano-thick calcium oxide armed titanium: boosts bone cells against methicillin-resistant Staphylococcus aureus. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4761977/
- Indian J Orthop. 2008 Apr-Jun; 42(2): 169–172. doi: 10.4103/0019-5413.39588. Calcium phosphate ceramics as bone graft substitutes in filling bone tumor defects. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2759621/
- Beeson WH; Arch Otolaryngol 107 (11): 664-9 (1981
- Small Entity Compliance Guide: Health Claims on Calcium and Osteoporosis; and Calcium, Vitamin D, and Osteoporosis. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/small-entity-compliance-guide-health-claims-calcium-and-osteoporosis-and-calcium-vitamin-d-and
- Osteoporosis prevention, diagnosis, and therapy. NIH Consens Statement. 2000 Mar 27-29;17(1):1-45.
- Riggs BL, Melton LJ 3rd. The worldwide problem of osteoporosis: insights afforded by epidemiology. Bone. 1995 Nov;17(5 Suppl):505S-511S. doi: 10.1016/8756-3282(95)00258-4
- National Osteoporosis Foundation. About Osteoporosis: Factors that Put You at Risk. https://www.nof.org/
- Cranney A, Horsley T, O’Donnell S, Weiler HA, Puil L, Ooi DS, Atkinson SA, Ward LM, Moher D, Hanley DA, Fang M, Yazdi F, Garritty C, Sampson M, Barrowman N, Tsertsvadze A, Mamaladze V. Effectiveness and Safety of Vitamin D in Relation to Bone Health. Evidence Report/Technology Assessment No. 158
- Bailey RL, Zou P, Wallace TC, McCabe GP, Craig BA, Jun S, Cauley JA, Weaver CM. Calcium Supplement Use Is Associated With Less Bone Mineral Density Loss, But Does Not Lessen the Risk of Bone Fracture Across the Menopause Transition: Data From the Study of Women’s Health Across the Nation. JBMR Plus. 2019 Nov 15;4(1):e10246. doi: 10.1002/jbm4.10246
- Bolland MJ, Leung W, Tai V, Bastin S, Gamble GD, Grey A, Reid IR. Calcium intake and risk of fracture: systematic review. BMJ 2015;351:h4580 doi: 10.1136/bmj.h4580.
- Tai V, Leung W, Grey A, Reid IR, Bolland MJ. Calcium intake and bone mineral density: systematic review and meta-analysis. BMJ 2015;351:h4183 doi: 10.1136/bmj.h4183
- US Preventive Services Task Force; Grossman DC, Curry SJ, Owens DK, Barry MJ, Caughey AB, Davidson KW, Doubeni CA, Epling JW Jr, Kemper AR, Krist AH, Kubik M, Landefeld S, Mangione CM, Silverstein M, Simon MA, Tseng CW. Vitamin D, Calcium, or Combined Supplementation for the Primary Prevention of Fractures in Community-Dwelling Adults: US Preventive Services Task Force Recommendation Statement. JAMA. 2018 Apr 17;319(15):1592-1599. doi: 10.1001/jama.2018.3185
- Weaver CM, Alexander DD, Boushey CJ, Dawson-Hughes B, Lappe JM, LeBoff MS, Liu S, Looker AC, Wallace TC, Wang DD. Calcium plus vitamin D supplementation and risk of fractures: an updated meta-analysis from the National Osteoporosis Foundation. Osteoporos Int. 2016 Jan;27(1):367-76. doi: 10.1007/s00198-015-3386-5. Epub 2015 Oct 28. Erratum in: Osteoporos Int. 2016 Aug;27(8):2643-6.
- Zhao JG, Zeng XT, Wang J, Liu L. Association Between Calcium or Vitamin D Supplementation and Fracture Incidence in Community-Dwelling Older Adults: A Systematic Review and Meta-analysis. JAMA. 2017 Dec 26;318(24):2466-2482. doi: 10.1001/jama.2017.19344
- Kahwati LC, Weber RP, Pan H, Gourlay M, LeBlanc E, Coker-Schwimmer M, Viswanathan M. Vitamin D, Calcium, or Combined Supplementation for the Primary Prevention of Fractures in Community-Dwelling Adults: Evidence Report and Systematic Review for the US Preventive Services Task Force. JAMA. 2018 Apr 17;319(15):1600-1612. doi: 10.1001/jama.2017.21640
- Crandall CJ, Aragaki AK, LeBoff MS, Li W, Wactawski-Wende J, Cauley JA, Margolis KL, Manson JE. Calcium plus vitamin D supplementation and height loss: findings from the Women’s Health Initiative Calcium and Vitamin D clinical trial. Menopause. 2016 Dec;23(12):1277-1286. doi: 10.1097/GME.0000000000000704
- Reyes-Garcia R, Mendoza N, Palacios S, Salas N, Quesada-Charneco M, Garcia-Martin A, Fonolla J, Lara-Villoslada F, Muñoz-Torres M. Effects of Daily Intake of Calcium and Vitamin D-Enriched Milk in Healthy Postmenopausal Women: A Randomized, Controlled, Double-Blind Nutritional Study. J Womens Health (Larchmt). 2018 May;27(5):561-568. doi: 10.1089/jwh.2017.6655
- Liu C , Kuang X , Li K , Guo X , Deng Q , Li D . Effects of combined calcium and vitamin D supplementation on osteoporosis in postmenopausal women: a systematic review and meta-analysis of randomized controlled trials. Food Funct. 2020 Dec 1;11(12):10817-10827. doi: 10.1039/d0fo00787k
- Silk LN, Greene DA, Baker MK. The Effect of Calcium or Calcium and Vitamin D Supplementation on Bone Mineral Density in Healthy Males: A Systematic Review and Meta-Analysis. Int J Sport Nutr Exerc Metab. 2015 Oct;25(5):510-24. doi: 10.1123/ijsnem.2014-0202
- Tai V, Leung W, Grey A, Reid IR, Bolland MJ. Calcium intake and bone mineral density: systematic review and meta-analysis. BMJ. 2015 Sep 29;351:h4183. doi: 10.1136/bmj.h4183
- U.S. Food and Drug Administration. Food labeling: health claims; calcium and osteoporosis, and calcium, vitamin D, and osteoporosis. https://www.fda.gov/Food/IngredientsPackagingLabeling/LabelingNutrition/ucm2006876.htm
- McCullough ML, Robertson AS, Rodriguez C, Jacobs EJ, Chao A, Carolyn J, Calle EE, Willett WC, Thun MJ. Calcium, vitamin D, dairy products, and risk of colorectal cancer in the Cancer Prevention Study II Nutrition Cohort (United States). Cancer Causes Control. 2003 Feb;14(1):1-12. doi: 10.1023/a:1022591007673
- Wu K, Willett WC, Fuchs CS, Colditz GA, Giovannucci EL. Calcium intake and risk of colon cancer in women and men. J Natl Cancer Inst. 2002 Mar 20;94(6):437-46. doi: 10.1093/jnci/94.6.437
- Pietinen P, Malila N, Virtanen M, Hartman TJ, Tangrea JA, Albanes D, Virtamo J. Diet and risk of colorectal cancer in a cohort of Finnish men. Cancer Causes Control. 1999 Oct;10(5):387-96. doi: 10.1023/a:1008962219408
- Terry P, Baron JA, Bergkvist L, Holmberg L, Wolk A. Dietary calcium and vitamin D intake and risk of colorectal cancer: a prospective cohort study in women. Nutr Cancer. 2002;43(1):39-46. doi: 10.1207/S15327914NC431_4
- Zheng W, Anderson KE, Kushi LH, Sellers TA, Greenstein J, Hong CP, Cerhan JR, Bostick RM, Folsom AR. A prospective cohort study of intake of calcium, vitamin D, and other micronutrients in relation to incidence of rectal cancer among postmenopausal women. Cancer Epidemiol Biomarkers Prev. 1998 Mar;7(3):221-5.
- Park Y, Leitzmann MF, Subar AF, Hollenbeck A, Schatzkin A. Dairy food, calcium, and risk of cancer in the NIH-AARP Diet and Health Study. Arch Intern Med. 2009 Feb 23;169(4):391-401. doi: 10.1001/archinternmed.2008.578
- Baron JA, Beach M, Mandel JS, van Stolk RU, Haile RW, Sandler RS, Rothstein R, Summers RW, Snover DC, Beck GJ, Frankl H, Pearson L, Bond JH, Greenberg ER. Calcium supplements and colorectal adenomas. Polyp Prevention Study Group. Ann N Y Acad Sci. 1999;889:138-45. doi: 10.1111/j.1749-6632.1999.tb08731.x
- Baron JA, Beach M, Mandel JS, van Stolk RU, Haile RW, Sandler RS, Rothstein R, Summers RW, Snover DC, Beck GJ, Bond JH, Greenberg ER. Calcium supplements for the prevention of colorectal adenomas. Calcium Polyp Prevention Study Group. N Engl J Med. 1999 Jan 14;340(2):101-7. doi: 10.1056/NEJM199901143400204
- Bonithon-Kopp C, Kronborg O, Giacosa A, Räth U, Faivre J. Calcium and fibre supplementation in prevention of colorectal adenoma recurrence: a randomised intervention trial. European Cancer Prevention Organisation Study Group. Lancet. 2000 Oct 14;356(9238):1300-6. doi: 10.1016/s0140-6736(00)02813-0
- Wactawski-Wende J, Kotchen JM, Anderson GL, et al. Women’s Health Initiative Investigators. Calcium plus vitamin D supplementation and the risk of colorectal cancer. N Engl J Med. 2006 Feb 16;354(7):684-96. doi: 10.1056/NEJMoa055222. Erratum in: N Engl J Med. 2006 Mar 9;354(10):1102.
- World Cancer Research Fund/American Institute for Cancer Research. Food, Nutrition, Physical Activity, and the Prevention of Cancer: A Global Perspective. 2007. American Institute for Cancer Research, Washington, DC. www.dietandcancerreport.org
- Lappe JM, Travers-Gustafson D, Davies KM, Recker RR, Heaney RP. Vitamin D and calcium supplementation reduces cancer risk: results of a randomized trial. Am J Clin Nutr. 2007 Jun;85(6):1586-91. doi: 10.1093/ajcn/85.6.1586. Erratum in: Am J Clin Nutr. 2008 Mar;87(3):794.
- Allen NE, Key TJ, Appleby PN, et al. Animal foods, protein, calcium and prostate cancer risk: the European Prospective Investigation into Cancer and Nutrition. Br J Cancer. 2008 May 6;98(9):1574-81. doi: 10.1038/sj.bjc.6604331
- Ahn J, Albanes D, Peters U, Schatzkin A, Lim U, Freedman M, Chatterjee N, Andriole GL, Leitzmann MF, Hayes RB; Prostate, Lung, Colorectal, and Ovarian Trial Project Team. Dairy products, calcium intake, and risk of prostate cancer in the prostate, lung, colorectal, and ovarian cancer screening trial. Cancer Epidemiol Biomarkers Prev. 2007 Dec;16(12):2623-30. doi: 10.1158/1055-9965.EPI-07-0601
- Park Y, Mitrou PN, Kipnis V, Hollenbeck A, Schatzkin A, Leitzmann MF. Calcium, dairy foods, and risk of incident and fatal prostate cancer: the NIH-AARP Diet and Health Study. Am J Epidemiol. 2007 Dec 1;166(11):1270-9. doi: 10.1093/aje/kwm268
- Goodman MT, Wu AH, Tung KH, McDuffie K, Kolonel LN, Nomura AM, Terada K, Wilkens LR, Murphy S, Hankin JH. Association of dairy products, lactose, and calcium with the risk of ovarian cancer. Am J Epidemiol. 2002 Jul 15;156(2):148-57. doi: 10.1093/aje/kwf022
- Song X, Li Z, Ji X, Zhang D. Calcium Intake and the Risk of Ovarian Cancer: A Meta-Analysis. Nutrients. 2017 Jun 30;9(7):679. doi: 10.3390/nu9070679
- Shin MH, Holmes MD, Hankinson SE, Wu K, Colditz GA, Willett WC. Intake of dairy products, calcium, and vitamin d and risk of breast cancer. J Natl Cancer Inst. 2002 Sep 4;94(17):1301-11. doi: 10.1093/jnci/94.17.1301
- Lin J, Manson JE, Lee IM, Cook NR, Buring JE, Zhang SM. Intakes of calcium and vitamin D and breast cancer risk in women. Arch Intern Med. 2007 May 28;167(10):1050-9. doi: 10.1001/archinte.167.10.1050
- Hidayat K, Chen GC, Zhang R, Du X, Zou SY, Shi BM, Qin LQ. Calcium intake and breast cancer risk: meta-analysis of prospective cohort studies. Br J Nutr. 2016 Jul;116(1):158-66. doi: 10.1017/S0007114516001768
- Chlebowski RT, Johnson KC, Kooperberg C, Pettinger M, Wactawski-Wende J, Rohan T, Rossouw J, Lane D, O’Sullivan MJ, Yasmeen S, Hiatt RA, Shikany JM, Vitolins M, Khandekar J, Hubbell FA; Women’s Health Initiative Investigators. Calcium plus vitamin D supplementation and the risk of breast cancer. J Natl Cancer Inst. 2008 Nov 19;100(22):1581-91. doi: 10.1093/jnci/djn360
- Chen Y, Strasser S, Cao Y, Wang KS, Zheng S. Calcium intake and hypertension among obese adults in United States: associations and implications explored. J Hum Hypertens. 2015 Sep;29(9):541-7. doi: 10.1038/jhh.2014.126
- Khan B, Nowson CA, Daly RM, English DR, Hodge AM, Giles GG, Ebeling PR. Higher Dietary Calcium Intakes Are Associated With Reduced Risks of Fractures, Cardiovascular Events, and Mortality: A Prospective Cohort Study of Older Men and Women. J Bone Miner Res. 2015 Oct;30(10):1758-66. doi: 10.1002/jbmr.2515
- Anderson JJ, Kruszka B, Delaney JA, He K, Burke GL, Alonso A, Bild DE, Budoff M, Michos ED. Calcium Intake From Diet and Supplements and the Risk of Coronary Artery Calcification and its Progression Among Older Adults: 10-Year Follow-up of the Multi-Ethnic Study of Atherosclerosis (MESA). J Am Heart Assoc. 2016 Oct 11;5(10):e003815. doi: 10.1161/JAHA.116.003815
- Chung M, Tang AM, Newberry SJ. Calcium Intake and Cardiovascular Disease Risk. Ann Intern Med. 2017 May 2;166(9):686-687. doi: 10.7326/L17-0136
- Champagne CM. Dietary interventions on blood pressure: the Dietary Approaches to Stop Hypertension (DASH) trials. Nutr Rev. 2006 Feb;64(2 Pt 2):S53-6. doi: 10.1111/j.1753-4887.2006.tb00234.x
- Cormick G, Ciapponi A, Cafferata ML, Belizán JM. Calcium supplementation for prevention of primary hypertension. Cochrane Database Syst Rev. 2015 Jun 30;2015(6):CD010037. doi: 10.1002/14651858.CD010037.pub2. Update in: Cochrane Database Syst Rev. 2021 Aug 10;8:CD010037
- Chen C, Ge S, Li S, Wu L, Liu T, Li C. The Effects of Dietary Calcium Supplements Alone or With Vitamin D on Cholesterol Metabolism: A Meta-Analysis of Randomized Controlled Trials. J Cardiovasc Nurs. 2017 Sep/Oct;32(5):496-506. doi: 10.1097/JCN.0000000000000379
- Donneyong MM, Hornung CA, Taylor KC, Baumgartner RN, Myers JA, Eaton CB, Gorodeski EZ, Klein L, Martin LW, Shikany JM, Song Y, Li W, Manson JE. Risk of heart failure among postmenopausal women: a secondary analysis of the randomized trial of vitamin D plus calcium of the women’s health initiative. Circ Heart Fail. 2015 Jan;8(1):49-56. doi: 10.1161/CIRCHEARTFAILURE.114.001738
- Boursiquot BC, Larson JC, Shalash OA, Vitolins MZ, Soliman EZ, Perez MV. Vitamin D with calcium supplementation and risk of atrial fibrillation in postmenopausal women. Am Heart J. 2019 Mar;209:68-78. doi: 10.1016/j.ahj.2018.12.006
- Myung SK, Kim HB, Lee YJ, Choi YJ, Oh SW. Calcium Supplements and Risk of Cardiovascular Disease: A Meta-Analysis of Clinical Trials. Nutrients. 2021 Jan 26;13(2):368. doi: 10.3390/nu13020368
- Yang B, Campbell PT, Gapstur SM, Jacobs EJ, Bostick RM, Fedirko V, Flanders WD, McCullough ML. Calcium intake and mortality from all causes, cancer, and cardiovascular disease: the Cancer Prevention Study II Nutrition Cohort. Am J Clin Nutr. 2016 Mar;103(3):886-94. doi: 10.3945/ajcn.115.117994
- Paik JM, Curhan GC, Sun Q, Rexrode KM, Manson JE, Rimm EB, Taylor EN. Calcium supplement intake and risk of cardiovascular disease in women. Osteoporos Int. 2014 Aug;25(8):2047-56. doi: 10.1007/s00198-014-2732-3
- Kopecky SL, Bauer DC, Gulati M, Nieves JW, Singer AJ, Toth PP, Underberg JA, Wallace TC, Weaver CM. Lack of Evidence Linking Calcium With or Without Vitamin D Supplementation to Cardiovascular Disease in Generally Healthy Adults: A Clinical Guideline From the National Osteoporosis Foundation and the American Society for Preventive Cardiology. Ann Intern Med. 2016 Dec 20;165(12):867-868. doi: 10.7326/M16-1743
- Appel LJ, Moore TJ, Obarzanek E, Vollmer WM, Svetkey LP, Sacks FM, Bray GA, Vogt TM, Cutler JA, Windhauser MM, Lin PH, Karanja N. A clinical trial of the effects of dietary patterns on blood pressure. DASH Collaborative Research Group. N Engl J Med. 1997 Apr 17;336(16):1117-24. doi: 10.1056/NEJM199704173361601
- Conlin PR, Chow D, Miller ER 3rd, Svetkey LP, Lin PH, Harsha DW, Moore TJ, Sacks FM, Appel LJ. The effect of dietary patterns on blood pressure control in hypertensive patients: results from the Dietary Approaches to Stop Hypertension (DASH) trial. Am J Hypertens. 2000 Sep;13(9):949-55. doi: 10.1016/s0895-7061(99)00284-8
- Miller GD, DiRienzo DD, Reusser ME, McCarron DA. Benefits of dairy product consumption on blood pressure in humans: a summary of the biomedical literature. J Am Coll Nutr. 2000 Apr;19(2 Suppl):147S-164S. doi: 10.1080/07315724.2000.10718085
- Cappuccio FP, Elliott P, Allender PS, Pryer J, Follman DA, Cutler JA. Epidemiologic association between dietary calcium intake and blood pressure: a meta-analysis of published data. Am J Epidemiol. 1995 Nov 1;142(9):935-45. doi: 10.1093/oxfordjournals.aje.a117741
- Allender PS, Cutler JA, Follmann D, Cappuccio FP, Pryer J, Elliott P. Dietary calcium and blood pressure: a meta-analysis of randomized clinical trials. Ann Intern Med. 1996 May 1;124(9):825-31. doi: 10.7326/0003-4819-124-9-199605010-00007
- Bucher HC, Cook RJ, Guyatt GH, Lang JD, Cook DJ, Hatala R, Hunt DL. Effects of dietary calcium supplementation on blood pressure. A meta-analysis of randomized controlled trials. JAMA. 1996 Apr 3;275(13):1016-22. doi: 10.1001/jama.1996.03530370054031
- McCarron DA, Reusser ME. Finding consensus in the dietary calcium-blood pressure debate. J Am Coll Nutr. 1999 Oct;18(5 Suppl):398S-405S. doi: 10.1080/07315724.1999.10718904
- Wang L, Manson JE, Buring JE, Lee IM, Sesso HD. Dietary intake of dairy products, calcium, and vitamin D and the risk of hypertension in middle-aged and older women. Hypertension. 2008 Apr;51(4):1073-9. doi: 10.1161/HYPERTENSIONAHA.107.107821
- Chung M, Balk EM, Brendel M, Ip S, Lau J, Lee J, Lichtenstein A, Patel K, Raman G, Tatsioni A, Terasawa T, Trikalinos TA. Vitamin D and Calcium: Systematic Review of Health Outcomes. Evidence Report/Technology Assessment No. 183. (Prepared by Tufts Evidence-based Practice Center under Contract No. 290-2007-10055-I). AHRQ Publication No. 09-E015, Rockville, MD: Agency for Healthcare Research and Quality. August 2009.
- Dickinson HO, Nicolson DJ, Cook JV, Campbell F, Beyer FR, Ford GA, Mason J. Calcium supplementation for the management of primary hypertension in adults. Cochrane Database Syst Rev. 2006 Apr 19;(2):CD004639. doi: 10.1002/14651858.CD004639.pub2
- Challoumas D, Cobbold C, Dimitrakakis G. Effects of calcium intake on the cardiovascular system in postmenopausal women. Atherosclerosis. 2013 Nov;231(1):1-7. doi: 10.1016/j.atherosclerosis.2013.08.018
- American Dietetic Association; Dietitians of Canada. Position of the American Dietetic Association and Dietitians of Canada: Vegetarian diets. J Am Diet Assoc. 2003 Jun;103(6):748-65. doi: 10.1053/jada.2003.50142
- Rouse IL, Beilin LJ, Armstrong BK, Vandongen R. Blood-pressure-lowering effect of a vegetarian diet: controlled trial in normotensive subjects. Lancet. 1983 Jan 1;1(8314-5):5-10. doi: 10.1016/s0140-6736(83)91557-x
- Margetts BM, Beilin LJ, Armstrong BK, Vandongen R. Vegetarian diet in the treatment of mild hypertension: a randomized controlled trial. J Hypertens Suppl. 1985 Dec;3(3):S429-31.
- Beilin LJ, Armstrong BK, Margetts BM, Rouse IL, Vandongen R. Vegetarian diet and blood pressure. Nephron. 1987;47 Suppl 1:37-41. doi: 10.1159/000184551
- Berkow SE, Barnard ND. Blood pressure regulation and vegetarian diets. Nutr Rev. 2005 Jan;63(1):1-8. doi: 10.1111/j.1753-4887.2005.tb00104.x
- Cudihy D, Lee RV. The pathophysiology of pre-eclampsia: current clinical concepts. J Obstet Gynaecol. 2009 Oct;29(7):576-82. doi: 10.1080/01443610903061751
- Preeclampsia and High Blood Pressure During Pregnancy. https://www.acog.org/womens-health/faqs/preeclampsia-and-high-blood-pressure-during-pregnancy
- Gestational Hypertension and Preeclampsia. https://www.acog.org/clinical/clinical-guidance/practice-bulletin/articles/2020/06/gestational-hypertension-and-preeclampsia
- Hypertension in pregnancy. Report of the American College of Obstetricians and Gynecologists’ Task Force on Hypertension in Pregnancy. Obstet Gynecol. 2013 Nov;122(5):1122-1131. doi: 10.1097/01.AOG.0000437382.03963.88
- Guideline: Calcium Supplementation in Pregnant Women. Geneva: World Health Organization; 2013. Available from: https://www.ncbi.nlm.nih.gov/books/NBK154180
- Hofmeyr GJ, Lawrie TA, Atallah ÁN, Torloni MR. Calcium supplementation during pregnancy for preventing hypertensive disorders and related problems. Cochrane Database Syst Rev. 2018 Oct 1;10(10):CD001059. doi: 10.1002/14651858.CD001059.pub5
- Tang R, Tang IC, Henry A, Welsh A. Limited evidence for calcium supplementation in preeclampsia prevention: a meta-analysis and systematic review. Hypertens Pregnancy. 2015 May;34(2):181-203. doi: 10.3109/10641955.2014.988353
- Hofmeyr GJ, Betrán AP, Singata-Madliki M, Cormick G, Munjanja SP, Fawcus S, Mose S, Hall D, Ciganda A, Seuc AH, Lawrie TA, Bergel E, Roberts JM, von Dadelszen P, Belizán JM; Calcium and Pre-eclampsia Study Group. Prepregnancy and early pregnancy calcium supplementation among women at high risk of pre-eclampsia: a multicentre, double-blind, randomised, placebo-controlled trial. Lancet. 2019 Jan 26;393(10169):330-339. doi: 10.1016/S0140-6736(18)31818-X
- Hofmeyr GJ, Manyame S, Medley N, Williams MJ. Calcium supplementation commencing before or early in pregnancy, for preventing hypertensive disorders of pregnancy. Cochrane Database Syst Rev. 2019 Sep 16;9(9):CD011192. doi: 10.1002/14651858.CD011192.pub3
- WHO recommendation: Calcium supplementation during pregnancy for the prevention of pre-eclampsia and its complications. Geneva: World Health Organization; 2018. Available from: https://www.ncbi.nlm.nih.gov/books/NBK535812
- Magee LA, Pels A, Helewa M, Rey E, von Dadelszen P; Canadian Hypertensive Disorders of Pregnancy Working Group. Diagnosis, evaluation, and management of the hypertensive disorders of pregnancy: executive summary. J Obstet Gynaecol Can. 2014 May;36(5):416-41. English, French. doi: 10.1016/s1701-2163(15)30588-0
- Tranquilli AL, Dekker G, Magee L, Roberts J, Sibai BM, Steyn W, Zeeman GG, Brown MA. The classification, diagnosis and management of the hypertensive disorders of pregnancy: A revised statement from the ISSHP. Pregnancy Hypertens. 2014 Apr;4(2):97-104. doi: 10.1016/j.preghy.2014.02.001
- Lowe SA, Bowyer L, Lust K, McMahon LP, Morton M, North RA, Paech M, Said JM. SOMANZ guidelines for the management of hypertensive disorders of pregnancy 2014. Aust N Z J Obstet Gynaecol. 2015 Oct;55(5):e1-29. doi: 10.1111/ajo.12399
- Hofmeyr GJ, Mlokoti Z, Nikodem VC, Mangesi L, Ferreira S, Singata M, Jafta Z, Merialdi M, Hazelden C, Villar J; WHO Calcium Supplementation for the Prevention of Pre-eclampsia Trial Group. Calcium supplementation during pregnancy for preventing hypertensive disorders is not associated with changes in platelet count, urate, and urinary protein: a randomized control trial. Hypertens Pregnancy. 2008;27(3):299-304. doi: 10.1080/10641950701826802
- Villar J, Abdel-Aleem H, Merialdi M, Mathai M, Ali MM, Zavaleta N, Purwar M, Hofmeyr J, Nguyen TN, Campódonico L, Landoulsi S, Carroli G, Lindheimer M; World Health Organization Calcium Supplementation for the Prevention of Preeclampsia Trial Group. World Health Organization randomized trial of calcium supplementation among low calcium intake pregnant women. Am J Obstet Gynecol. 2006 Mar;194(3):639-49. doi: 10.1016/j.ajog.2006.01.068
- Hofmeyr GJ, Seuc AH, Betrán AP, Purnat TD, Ciganda A, Munjanja SP, Manyame S, Singata M, Fawcus S, Frank K, Hall DR, Cormick G, Roberts JM, Bergel EF, Drebit SK, Von Dadelszen P, Belizan JM; Calcium and Pre-eclampsia Study Group. The effect of calcium supplementation on blood pressure in non-pregnant women with previous pre-eclampsia: An exploratory, randomized placebo controlled study. Pregnancy Hypertens. 2015 Oct;5(4):273-9. doi: 10.1016/j.preghy.2015.04.001
- Jackson RD, LaCroix AZ, Gass M, Wallace RB, Robbins J, Lewis CE, Bassford T, Beresford SA, Black HR, Blanchette P, Bonds DE, Brunner RL, Brzyski RG, Caan B, Cauley JA, Chlebowski RT, Cummings SR, Granek I, Hays J, Heiss G, Hendrix SL, Howard BV, Hsia J, Hubbell FA, Johnson KC, Judd H, Kotchen JM, Kuller LH, Langer RD, Lasser NL, Limacher MC, Ludlam S, Manson JE, Margolis KL, McGowan J, Ockene JK, O’Sullivan MJ, Phillips L, Prentice RL, Sarto GE, Stefanick ML, Van Horn L, Wactawski-Wende J, Whitlock E, Anderson GL, Assaf AR, Barad D; Women’s Health Initiative Investigators. Calcium plus vitamin D supplementation and the risk of fractures. N Engl J Med. 2006 Feb 16;354(7):669-83. doi: 10.1056/NEJMoa055218. Erratum in: N Engl J Med. 2006 Mar 9;354(10):1102.
- Wallace RB, Wactawski-Wende J, O’Sullivan MJ, Larson JC, Cochrane B, Gass M, Masaki K. Urinary tract stone occurrence in the Women’s Health Initiative (WHI) randomized clinical trial of calcium and vitamin D supplements. Am J Clin Nutr. 2011 Jul;94(1):270-7. doi: 10.3945/ajcn.110.003350
- Escribano J, Balaguer A, Roqué i Figuls M, Feliu A, Ferre N. Dietary interventions for preventing complications in idiopathic hypercalciuria. Cochrane Database Syst Rev. 2014 Feb 11;(2):CD006022. doi: 10.1002/14651858.CD006022.pub4
- Heilberg IP, Goldfarb DS. Optimum nutrition for kidney stone disease. Adv Chronic Kidney Dis. 2013 Mar;20(2):165-74. doi: 10.1053/j.ackd.2012.12.001
- Prezioso D, Strazzullo P, Lotti T, et al. CLU Working Group. Dietary treatment of urinary risk factors for renal stone formation. A review of CLU Working Group. Arch Ital Urol Androl. 2015 Jul 7;87(2):105-20. doi: 10.4081/aiua.2015.2.105. Erratum in: Arch Ital Urol Androl. 2016 Mar;88(1):76. Ferraro, Manuel [added].
- Duley L. The global impact of pre-eclampsia and eclampsia. Semin Perinatol. 2009 Jun;33(3):130-7. doi: 10.1053/j.semperi.2009.02.010
- Lerolle N, Lantz B, Paillard F, Gattegno B, Flahault A, Ronco P, Houillier P, Rondeau E. Risk factors for nephrolithiasis in patients with familial idiopathic hypercalciuria. Am J Med. 2002 Aug 1;113(2):99-103. doi: 10.1016/s0002-9343(02)01152-x
- Sorensen MD, Eisner BH, Stone KL, Kahn AJ, Lui LY, Sadetsky N, Stoller ML. Impact of calcium intake and intestinal calcium absorption on kidney stones in older women: the study of osteoporotic fractures. J Urol. 2012 Apr;187(4):1287-92. doi: 10.1016/j.juro.2011.11.109
- Curhan GC, Willett WC, Knight EL, Stampfer MJ. Dietary factors and the risk of incident kidney stones in younger women: Nurses’ Health Study II. Arch Intern Med. 2004 Apr 26;164(8):885-91. doi: 10.1001/archinte.164.8.885
- Curhan GC, Willett WC, Rimm EB, Stampfer MJ. A prospective study of dietary calcium and other nutrients and the risk of symptomatic kidney stones. N Engl J Med. 1993 Mar 25;328(12):833-8. doi: 10.1056/NEJM199303253281203
- Taylor EN, Stampfer MJ, Curhan GC. Dietary factors and the risk of incident kidney stones in men: new insights after 14 years of follow-up. J Am Soc Nephrol. 2004 Dec;15(12):3225-32. doi: 10.1097/01.ASN.0000146012.44570.20
- Borghi L, Schianchi T, Meschi T, Guerra A, Allegri F, Maggiore U, Novarini A. Comparison of two diets for the prevention of recurrent stones in idiopathic hypercalciuria. N Engl J Med. 2002 Jan 10;346(2):77-84. doi: 10.1056/NEJMoa010369
- Hess B, Jost C, Zipperle L, Takkinen R, Jaeger P. High-calcium intake abolishes hyperoxaluria and reduces urinary crystallization during a 20-fold normal oxalate load in humans. Nephrol Dial Transplant. 1998 Sep;13(9):2241-7. doi: 10.1093/ndt/13.9.2241
- Liebman M, Chai W. Effect of dietary calcium on urinary oxalate excretion after oxalate loads. Am J Clin Nutr. 1997 May;65(5):1453-9. doi: 10.1093/ajcn/65.5.1453
- Lange JN, Wood KD, Mufarrij PW, Callahan MF, Easter L, Knight J, Holmes RP, Assimos DG. The impact of dietary calcium and oxalate ratios on stone risk. Urology. 2012 Jun;79(6):1226-9. doi: 10.1016/j.urology.2012.01.053
- Jackson RD, LaCroix AZ, Gass M, et al. Women’s Health Initiative Investigators. Calcium plus vitamin D supplementation and the risk of fractures. N Engl J Med. 2006 Feb 16;354(7):669-83. doi: 10.1056/NEJMoa055218. Erratum in: N Engl J Med. 2006 Mar 9;354(10):1102.
- Curhan GC, Willett WC, Speizer FE, Spiegelman D, Stampfer MJ. Comparison of dietary calcium with supplemental calcium and other nutrients as factors affecting the risk for kidney stones in women. Ann Intern Med. 1997 Apr 1;126(7):497-504. doi: 10.7326/0003-4819-126-7-199704010-00001
- Bihl G, Meyers A. Recurrent renal stone disease-advances in pathogenesis and clinical management. Lancet. 2001 Aug 25;358(9282):651-6. doi: 10.1016/S0140-6736(01)05782-8
- Hall WD, Pettinger M, Oberman A, Watts NB, Johnson KC, Paskett ED, Limacher MC, Hays J. Risk factors for kidney stones in older women in the southern United States. Am J Med Sci. 2001 Jul;322(1):12-8. doi: 10.1097/00000441-200107000-00003
- Heaney RP. Calcium supplementation and incident kidney stone risk: a systematic review. J Am Coll Nutr. 2008 Oct;27(5):519-27. doi: 10.1080/07315724.2008.10719734
- Candelas G, Martinez-Lopez JA, Rosario MP, Carmona L, Loza E. Calcium supplementation and kidney stone risk in osteoporosis: a systematic literature review. Clin Exp Rheumatol. 2012 Nov-Dec;30(6):954-61.
- Dougkas A, Reynolds CK, Givens ID, Elwood PC, Minihane AM. Associations between dairy consumption and body weight: a review of the evidence and underlying mechanisms. Nutr Res Rev. 2011 Jun;24(1):72-95. doi: 10.1017/S095442241000034X
- Li P, Fan C, Lu Y, Qi K. Effects of calcium supplementation on body weight: a meta-analysis. Am J Clin Nutr. 2016 Nov;104(5):1263-1273. doi: 10.3945/ajcn.116.136242
- Caan B, Neuhouser M, Aragaki A, Lewis CB, Jackson R, LeBoff MS, Margolis KL, Powell L, Uwaifo G, Whitlock E, Wylie-Rosett J, LaCroix A. Calcium plus vitamin D supplementation and the risk of postmenopausal weight gain. Arch Intern Med. 2007 May 14;167(9):893-902. doi: 10.1001/archinte.167.9.893
- Booth AO, Huggins CE, Wattanapenpaiboon N, Nowson CA. Effect of increasing dietary calcium through supplements and dairy food on body weight and body composition: a meta-analysis of randomised controlled trials. Br J Nutr. 2015 Oct 14;114(7):1013-25. doi: 10.1017/S0007114515001518
- Shahar DR, Schwarzfuchs D, Fraser D, Vardi H, Thiery J, Fiedler GM, Blüher M, Stumvoll M, Stampfer MJ, Shai I; DIRECT Group. Dairy calcium intake, serum vitamin D, and successful weight loss. Am J Clin Nutr. 2010 Nov;92(5):1017-22. doi: 10.3945/ajcn.2010.29355
- Nappo A, Sparano S, Intemann T, Kourides YA, Lissner L, Molnar D, Moreno LA, Pala V, Sioen I, Veidebaum T, Wolters M, Siani A, Russo P. Dietary calcium intake and adiposity in children and adolescents: Cross-sectional and longitudinal results from IDEFICS/I.Family cohort. Nutr Metab Cardiovasc Dis. 2019 May;29(5):440-449. doi: 10.1016/j.numecd.2019.01.015
- Marabujo T, Ramos E, Lopes C. Dairy products and total calcium intake at 13 years of age and its association with obesity at 21 years of age. Eur J Clin Nutr. 2018 Apr;72(4):541-547. doi: 10.1038/s41430-017-0082-x
- Chen M, Pan A, Malik VS, Hu FB. Effects of dairy intake on body weight and fat: a meta-analysis of randomized controlled trials. Am J Clin Nutr. 2012 Oct;96(4):735-47. doi: 10.3945/ajcn.112.037119
- Soares MJ, Pathak K, Calton EK. Calcium and vitamin D in the regulation of energy balance: where do we stand? Int J Mol Sci. 2014 Mar 20;15(3):4938-45. doi: 10.3390/ijms15034938
- Mielke HW, Gonzales C, Powell E, Mielke PW. Evolving from reactive to proactive medicine: community lead (Pb) and clinical disparities in pre- and post-Katrina New Orleans. Int J Environ Res Public Health. 2014 Jul 21;11(7):7482-91. doi: 10.3390/ijerph110707482
- Blood Lead Reference Value. https://www.cdc.gov/nceh/lead/data/blood-lead-reference-value.htm?CDC_AA_refVal=https%3A%2F%2Fwww.cdc.gov%2Fnceh%2Flead%2Facclpp%2Fblood_lead_levels.htm
- Bruening K, Kemp FW, Simone N, Holding Y, Louria DB, Bogden JD. Dietary calcium intakes of urban children at risk of lead poisoning. Environ Health Perspect. 1999 Jun;107(6):431-5. doi: 10.1289/ehp.99107431
- Hertz-Picciotto I, Schramm M, Watt-Morse M, Chantala K, Anderson J, Osterloh J. Patterns and determinants of blood lead during pregnancy. Am J Epidemiol. 2000 Nov 1;152(9):829-37. doi: 10.1093/aje/152.9.829
- Ettinger AS, Lamadrid-Figueroa H, Téllez-Rojo MM, Mercado-García A, Peterson KE, Schwartz J, Hu H, Hernández-Avila M. Effect of calcium supplementation on blood lead levels in pregnancy: a randomized placebo-controlled trial. Environ Health Perspect. 2009 Jan;117(1):26-31. doi: 10.1289/ehp.11868
- Ettinger AS, Téllez-Rojo MM, Amarasiriwardena C, Peterson KE, Schwartz J, Aro A, Hu H, Hernández-Avila M. Influence of maternal bone lead burden and calcium intake on levels of lead in breast milk over the course of lactation. Am J Epidemiol. 2006 Jan 1;163(1):48-56. doi: 10.1093/aje/kwj010
- Hernandez-Avila M, Gonzalez-Cossio T, Hernandez-Avila JE, Romieu I, Peterson KE, Aro A, Palazuelos E, Hu H. Dietary calcium supplements to lower blood lead levels in lactating women: a randomized placebo-controlled trial. Epidemiology. 2003 Mar;14(2):206-12. doi: 10.1097/01.EDE.0000038520.66094.34
- Muldoon SB, Cauley JA, Kuller LH, Scott J, Rohay J. Lifestyle and sociodemographic factors as determinants of blood lead levels in elderly women. Am J Epidemiol. 1994 Mar 15;139(6):599-608. doi: 10.1093/oxfordjournals.aje.a117049
- Pearlstein T, Steiner M. Premenstrual dysphoric disorder: burden of illness and treatment update. J Psychiatry Neurosci. 2008 Jul;33(4):291-301. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2440788
- Bendich A. The potential for dietary supplements to reduce premenstrual syndrome (PMS) symptoms. J Am Coll Nutr. 2000 Feb;19(1):3-12. doi: 10.1080/07315724.2000.10718907
- Bertone-Johnson ER, Hankinson SE, Bendich A, Johnson SR, Willett WC, Manson JE. Calcium and vitamin D intake and risk of incident premenstrual syndrome. Arch Intern Med. 2005 Jun 13;165(11):1246-52. doi: 10.1001/archinte.165.11.1246
- Thys-Jacobs S, Starkey P, Bernstein D, Tian J. Calcium carbonate and the premenstrual syndrome: effects on premenstrual and menstrual symptoms. Premenstrual Syndrome Study Group. Am J Obstet Gynecol. 1998 Aug;179(2):444-52. doi: 10.1016/s0002-9378(98)70377-1
- Thys-Jacobs S, Ceccarelli S, Bierman A, Weisman H, Cohen MA, Alvir J. Calcium supplementation in premenstrual syndrome: a randomized crossover trial. J Gen Intern Med. 1989 May-Jun;4(3):183-9. doi: 10.1007/BF02599520
- Alvir JM, Thys-Jacobs S. Premenstrual and menstrual symptom clusters and response to calcium treatment. Psychopharmacol Bull. 1991;27(2):145-8.
- Bharati M. Comparing the Effects of Yoga & Oral Calcium Administration in Alleviating Symptoms of Premenstrual Syndrome in Medical Undergraduates. J Caring Sci. 2016 Sep 1;5(3):179-185. doi: 10.15171/jcs.2016.019
- Masoumi SZ, Ataollahi M, Oshvandi K. Effect of Combined Use of Calcium and Vitamin B6 on Premenstrual Syndrome Symptoms: a Randomized Clinical Trial. J Caring Sci. 2016 Mar 1;5(1):67-73. doi: 10.15171/jcs.2016.007
- Shehata NAA. RETRACTED: Calcium versus oral contraceptive pills containing drospirenone for the treatment of mild to moderate premenstrual syndrome: a double blind randomized placebo controlled trial. Eur J Obstet Gynecol Reprod Biol. 2016 Mar;198:100-104. doi: 10.1016/j.ejogrb.2016.01.015. Epub 2016 Jan 13. Retraction in: Eur J Obstet Gynecol Reprod Biol. 2019 Sep;240:387.
- Shobeiri F, Araste FE, Ebrahimi R, Jenabi E, Nazari M. Effect of calcium on premenstrual syndrome: A double-blind randomized clinical trial. Obstet Gynecol Sci. 2017 Jan;60(1):100-105. doi: 10.5468/ogs.2017.60.1.100
- Nevatte T, O’Brien PM, Bäckström T, Brown C, Dennerstein L, Endicott J, Epperson CN, Eriksson E, Freeman EW, Halbreich U, Ismail K, Panay N, Pearlstein T, Rapkin A, Reid R, Rubinow D, Schmidt P, Steiner M, Studd J, Sundström-Poromaa I, Yonkers K; Consensus Group of the International Society for Premenstrual Disorders. ISPMD consensus on the management of premenstrual disorders. Arch Womens Ment Health. 2013 Aug;16(4):279-91. doi: 10.1007/s00737-013-0346-y
- Whelan AM, Jurgens TM, Naylor H. Herbs, vitamins and minerals in the treatment of premenstrual syndrome: a systematic review. Can J Clin Pharmacol. 2009 Fall;16(3):e407-29.
- Dietary Guidelines for Americans. https://www.dietaryguidelines.gov
- U.S. Department of Agriculture, Center for Nutrition Policy and Promotion. ChooseMyPlate.gov 2011. https://www.myplate.gov
- Wongdee K, Rodrat M, Teerapornpuntakit J, Krishnamra N, Charoenphandhu N. Factors inhibiting intestinal calcium absorption: hormones and luminal factors that prevent excessive calcium uptake. J Physiol Sci. 2019 Sep;69(5):683-696. doi: 10.1007/s12576-019-00688-3
- Gallagher JC, Yalamanchili V, Smith LM. The effect of vitamin D on calcium absorption in older women. J Clin Endocrinol Metab. 2012 Oct;97(10):3550-6. doi: 10.1210/jc.2012-2020
- Food Labeling: Revision of the Nutrition and Supplement Facts Labels. https://www.federalregister.gov/documents/2016/05/27/2016-11867/food-labeling-revision-of-the-nutrition-and-supplement-facts-labels
- The USDA Food DataCentral. https://fdc.nal.usda.gov
- Wallace TC, McBurney M, Fulgoni VL 3rd. Multivitamin/mineral supplement contribution to micronutrient intakes in the United States, 2007-2010. J Am Coll Nutr. 2014;33(2):94-102. doi: 10.1080/07315724.2013.846806
- U.S. Department of Agriculture, Agricultural Research Service. Food Surveys Research Group: Beltsville, MD. https://www.ars.usda.gov/northeast-area/beltsville-md-bhnrc/beltsville-human-nutrition-research-center/food-surveys-research-group/docs/wweia-data-tables/
- Blumberg JB, Frei B, Fulgoni VL III, Weaver CM, Zeisel SH. Contribution of Dietary Supplements to Nutritional Adequacy in Race/Ethnic Population Subgroups in the United States. Nutrients. 2017 Nov 28;9(12):1295. doi: 10.3390/nu9121295
- Marshall K, Teo L, Shanahan C, Legette L, Mitmesser SH. Inadequate calcium and vitamin D intake and osteoporosis risk in older Americans living in poverty with food insecurities. PLoS One. 2020 Jul 8;15(7):e0235042. doi: 10.1371/journal.pone.0235042
- Potts JT Jr. Chemistry and physiology of parathyroid hormone. Clin Endocrinol (Oxf). 1976;5 Suppl:307S-315S. doi: 10.1111/j.1365-2265.1976.tb03839.x
- Silver J, Levi R. Regulation of PTH synthesis and secretion relevant to the management of secondary hyperparathyroidism in chronic kidney disease. Kidney Int Suppl. 2005 Jun;(95):S8-12. doi: 10.1111/j.1523-1755.2005.09501.x
- Khan M, Jose A, Sharma S. Physiology, Parathyroid Hormone. [Updated 2022 Oct 29]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK499940
- Dirk Vanderschueren and others, Active Vitamin D (1,25-Dihydroxyvitamin D) and Bone Health in Middle-Aged and Elderly Men: The European Male Aging Study (EMAS), The Journal of Clinical Endocrinology & Metabolism, Volume 98, Issue 3, 1 March 2013, Pages 995–1005, https://doi.org/10.1210/jc.2012-2772
- Lips P. Vitamin D physiology. Prog Biophys Mol Biol. 2006 Sep;92(1):4-8. doi: 10.1016/j.pbiomolbio.2006.02.016
- Lieben L, Carmeliet G, Masuyama R. Calcemic actions of vitamin D: effects on the intestine, kidney and bone. Best Pract Res Clin Endocrinol Metab. 2011 Aug;25(4):561-72. doi: 10.1016/j.beem.2011.05.008
- Hypocalcemia. https://emedicine.medscape.com/article/241893-overview#a5
- Sarko J. Bone and mineral metabolism. Emerg Med Clin North Am. 2005 Aug;23(3):703-21, viii. doi: 10.1016/j.emc.2005.03.01
- Goyal A, Anastasopoulou C, Ngu M, et al. Hypocalcemia. [Updated 2022 Jul 24]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK430912
- Schnitzler CM, Pettifor JM. Calcium Deficiency Rickets in African Adolescents: Cortical Bone Histomorphometry. JBMR Plus. 2019 Feb 11;3(6):e10169. doi: 10.1002/jbm4.10169
- Kooh SW, Fraser D, Reilly BJ, Hamilton JR, Gall DG, Bell L. Rickets due to calcium deficiency. N Engl J Med. 1977;297:1264–1266. doi: 10.1056/NEJM197712082972307
- Thacher TD, Fischer PR, Pettifor JM. Rickets: vitamin D and calcium deficiency. J Bone Miner Res. 2007;22:638–638. doi: 10.1359/jbmr.070118
- Pettifor JM. Nutritional rickets: deficiency of vitamin D, calcium, or both? Am J Clin Nutr. 2004 Dec;80(6 Suppl):1725S-9S. doi: 10.1093/ajcn/80.6.1725S
- Sempos CT, Durazo-Arvizu RA, Fischer PR, Munns CF, Pettifor JM, Thacher TD. Serum 25-hydroxyvitamin D requirements to prevent nutritional rickets in Nigerian children on a low-calcium diet-a multivariable reanalysis. Am J Clin Nutr. 2021 Jul 1;114(1):231-237. doi: 10.1093/ajcn/nqab048
- Cooper MS, Gittoes NJ. Diagnosis and management of hypocalcaemia. BMJ. 2008 Jun 7;336(7656):1298-302. doi: 10.1136/bmj.39582.589433.BE. Erratum in: BMJ. 2008 Jun 28;336(7659): doi: 10.1136/bmj.a334
- Chen Z, Zhao Q, Du J, Wang Y, Han R, Xu C, Chen X, Shu M. Risk factors for postoperative hypocalcaemia after thyroidectomy: A systematic review and meta-analysis. J Int Med Res. 2021 Mar;49(3):300060521996911. doi: 10.1177/0300060521996911
- Kakava K, Tournis S, Papadakis G, Karelas I, Stampouloglou P, Kassi E, Triantafillopoulos I, Villiotou V, Karatzas T. Postsurgical Hypoparathyroidism: A Systematic Review. In Vivo. 2016 May-Jun;30(3):171-9.
- Edafe O, Antakia R, Laskar N, Uttley L, Balasubramanian SP. Systematic review and meta-analysis of predictors of post-thyroidectomy hypocalcaemia. Br J Surg. 2014 Mar;101(4):307-20. doi: 10.1002/bjs.9384
- Mathur A, Nagarajan N, Kahan S, Schneider EB, Zeiger MA. Association of Parathyroid Hormone Level With Postthyroidectomy Hypocalcemia: A Systematic Review. JAMA Surg. 2018 Jan 1;153(1):69-76. doi: 10.1001/jamasurg.2017.3398
- Muhammed H, Jain A, Irfan M, Charles S, Dwivedi P, Chavan PP, Khubchandani R, Sharma A, Phatak S, Shukla AN, Shah R, Subramanian N, Pandya SC, Singh YP, Chengappa KG, Thabah M, Rajasekhar L, Shobha V, Negi VS, Dhir V, Sharma A, Misra R, Aggarwal A; SLE-SIG of IRA. Clinical features, severity and outcome of acute pancreatitis in systemic lupus erythematosus. Rheumatol Int. 2022 Aug;42(8):1363-1371. doi: 10.1007/s00296-021-04834-2
- Kelly A, Levine MA. Hypocalcemia in the critically ill patient. J Intensive Care Med. 2013 May-Jun;28(3):166-77. doi: 10.1177/0885066611411543
- Martha JW, Wibowo A, Pranata R. Hypocalcemia is associated with severe COVID-19: A systematic review and meta-analysis. Diabetes Metab Syndr. 2021 Jan-Feb;15(1):337-342. doi: 10.1016/j.dsx.2021.01.003
- Jacob J, Raghothama S, Subramanyam K, Johny D. Hypomagnesaemia causing functional hypoparathyroidism in rheumatic mitral stenosis leading to sudden cardiac arrest in a young woman. BMJ Case Rep. 2021 Jul 19;14(7):e244176. doi: 10.1136/bcr-2021-244176
- Liamis G, Milionis HJ, Elisaf M. A review of drug-induced hypocalcemia. J Bone Miner Metab. 2009;27(6):635-42. doi: 10.1007/s00774-009-0119-x
- Marlow CF, Sharma S, Babar F, Lin J. Severe Hypocalcemia and Hypomagnesemia with Denosumab in Advanced Chronic Kidney Disease: Case Report and Literature Review. Case Rep Oncol Med. 2018 Oct 14;2018:2059364. doi: 10.1155/2018/2059364
- Vasudeva M, Mathew JK, Groombridge C, Tee JW, Johnny CS, Maini A, Fitzgerald MC. Hypocalcemia in trauma patients: A systematic review. J Trauma Acute Care Surg. 2021 Feb 1;90(2):396-402. doi: 10.1097/TA.0000000000003027
- Almaghamsi A, Almalki MH, Buhary BM. Hypocalcemia in Pregnancy: A Clinical Review Update. Oman Med J. 2018 Nov;33(6):453-462. doi: 10.5001/omj.2018.85
- Cano A, Chedraui P, Goulis DG, Lopes P, Mishra G, Mueck A, Senturk LM, Simoncini T, Stevenson JC, Stute P, Tuomikoski P, Rees M, Lambrinoudaki I. Calcium in the prevention of postmenopausal osteoporosis: EMAS clinical guide. Maturitas. 2018 Jan;107:7-12. doi: 10.1016/j.maturitas.2017.10.004
- Abrams SA, Silber TJ, Esteban NV, Vieira NE, Stuff JE, Meyers R, Majd M, Yergey AL. Mineral balance and bone turnover in adolescents with anorexia nervosa. J Pediatr. 1993 Aug;123(2):326-31. doi: 10.1016/s0022-3476(05)81714-7
- Drinkwater BL, Bruemner B, Chesnut CH 3rd. Menstrual history as a determinant of current bone density in young athletes. JAMA. 1990 Jan 26;263(4):545-8.
- Marcus R, Cann C, Madvig P, Minkoff J, Goddard M, Bayer M, Martin M, Gaudiani L, Haskell W, Genant H. Menstrual function and bone mass in elite women distance runners. Endocrine and metabolic features. Ann Intern Med. 1985 Feb;102(2):158-63. doi: 10.7326/0003-4819-102-2-158
- Nattiv A. Stress fractures and bone health in track and field athletes. J Sci Med Sport. 2000 Sep;3(3):268-79. doi: 10.1016/s1440-2440(00)80036-5
- Lappe J, Cullen D, Haynatzki G, Recker R, Ahlf R, Thompson K. Calcium and vitamin d supplementation decreases incidence of stress fractures in female navy recruits. J Bone Miner Res. 2008 May;23(5):741-9. doi: 10.1359/jbmr.080102
- Boaventura RM, Mendonça RB, Fonseca FA, Mallozi M, Souza FS, Sarni ROS. Nutritional status and food intake of children with cow’s milk allergy. Allergol Immunopathol (Madr). 2019 Nov-Dec;47(6):544-550. doi: 10.1016/j.aller.2019.03.003
- Bakaloudi DR, Halloran A, Rippin HL, Oikonomidou AC, Dardavesis TI, Williams J, Wickramasinghe K, Breda J, Chourdakis M. Intake and adequacy of the vegan diet. A systematic review of the evidence. Clin Nutr. 2021 May;40(5):3503-3521. doi: 10.1016/j.clnu.2020.11.035
- Wilt TJ, Shaukat A, Shamliyan T, Taylor BC, MacDonald R, Tacklind J, Rutks I, Schwarzenberg SJ, Kane RL, and Levitt M. Lactose Intolerance and Health. No. 192 (Prepared by the Minnesota Evidence-based Practice Center under Contract No. HHSA 290-2007-10064-I.) AHRQ Publication No. 10-E004. Rockville, MD. Agency for Healthcare Research and Quality. February 2010.
- Suchy FJ, Brannon PM, Carpenter TO, Fernandez JR, Gilsanz V, Gould JB, Hall K, Hui SL, Lupton J, Mennella J, Miller NJ, Osganian SK, Sellmeyer DE, Wolf MA. NIH Consensus Development Conference Statement: Lactose Intolerance and Health. NIH Consens State Sci Statements. 2010 Feb 22–24; 27(2):1–27. https://consensus.nih.gov/2010/lactosestatement.htm
- Johnson AO, Semenya JG, Buchowski MS, Enwonwu CO, Scrimshaw NS. Correlation of lactose maldigestion, lactose intolerance, and milk intolerance. Am J Clin Nutr. 1993 Mar;57(3):399-401. doi: 10.1093/ajcn/57.3.399
- Nose O, Iida Y, Kai H, Harada T, Ogawa M, Yabuuchi H. Breath hydrogen test for detecting lactose malabsorption in infants and children. Prevalence of lactose malabsorption in Japanese children and adults. Arch Dis Child. 1979 Jun;54(6):436-40. doi: 10.1136/adc.54.6.436
- Rao DR, Bello H, Warren AP, Brown GE. Prevalence of lactose maldigestion. Influence and interaction of age, race, and sex. Dig Dis Sci. 1994 Jul;39(7):1519-24. doi: 10.1007/BF02088058
- Hertzler SR, Savaiano DA. Colonic adaptation to daily lactose feeding in lactose maldigesters reduces lactose intolerance. Am J Clin Nutr. 1996 Aug;64(2):232-6. doi: 10.1093/ajcn/64.2.232
- Pribila BA, Hertzler SR, Martin BR, Weaver CM, Savaiano DA. Improved lactose digestion and intolerance among African-American adolescent girls fed a dairy-rich diet. J Am Diet Assoc. 2000 May;100(5):524-8; quiz 529-30. doi: 10.1016/S0002-8223(00)00162-0
- Rona RJ, Keil T, Summers C, Gislason D, Zuidmeer L, Sodergren E, Sigurdardottir ST, Lindner T, Goldhahn K, Dahlstrom J, McBride D, Madsen C. The prevalence of food allergy: a meta-analysis. J Allergy Clin Immunol. 2007 Sep;120(3):638-46. doi: 10.1016/j.jaci.2007.05.026
- Marsh AG, Sanchez TV, Midkelsen O, Keiser J, Mayor G. Cortical bone density of adult lacto-ovo-vegetarian and omnivorous women. J Am Diet Assoc. 1980 Feb;76(2):148-51.
- Reed JA, Anderson JJ, Tylavsky FA, Gallagher PN Jr. Comparative changes in radial-bone density of elderly female lacto-ovovegetarians and omnivores. Am J Clin Nutr. 1994 May;59(5 Suppl):1197S-1202S. doi: 10.1093/ajcn/59.5.1197S. Erratum in: Am J Clin Nutr 1994 Dec;60(6):981.
- Janelle KC, Barr SI. Nutrient intakes and eating behavior scores of vegetarian and nonvegetarian women. J Am Diet Assoc. 1995 Feb;95(2):180-6, 189, quiz 187-8. doi: 10.1016/s0002-8223(95)00045-3
- Appleby P, Roddam A, Allen N, Key T. Comparative fracture risk in vegetarians and nonvegetarians in EPIC-Oxford. Eur J Clin Nutr. 2007 Dec;61(12):1400-6. doi: 10.1038/sj.ejcn.1602659
- Hurley K, Baggs D. Hypocalcemic cardiac failure in the emergency department. J Emerg Med. 2005 Feb;28(2):155-9. doi: 10.1016/j.jemermed.2004.06.014
- Kaye M, Somerville PJ, Lowe G, Ketis M, Schneider W. Hypocalcemic tetany and metabolic alkalosis in a dialysis patient: an unusual event. Am J Kidney Dis. 1997 Sep;30(3):440-4. doi: 10.1016/s0272-6386(97)90292-4
- Velasco PJ, Manshadi M, Breen K, Lippmann S. Psychiatric aspects of parathyroid disease. Psychosomatics. 1999 Nov-Dec;40(6):486-90. doi: 10.1016/s0033-3182(99)71186-2
- Ayuk J, Matthews T, Tayebjee M, Gittoes NJ. A blind panic. Lancet. 2001 Apr 21;357(9264):1262. doi: 10.1016/S0140-6736(00)04408-1
- Kamalanathan S, Balachandran K, Parthan G, et al. Chvostek’s sign: a video demonstration Case Reports 2012;2012:bcr2012007098 http://dx.doi.org/10.1136/bcr-2012-007098
- Hujoel IA. The association between serum calcium levels and Chvostek sign: A population-based study. Neurol Clin Pract. 2016;6(4):321–328. doi:10.1212/CPJ.0000000000000270 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5727700
- What is the Chvostek sign in patients with hypocalcemia? https://www.medscape.com/answers/241893-20150/what-is-the-chvostek-sign-in-patients-with-hypocalcemia
- Urbano FL. Signs of hypocalcemia: Chvostek’s and Trousseau’s. Hosp Physician 2000;36:43-5.
- Eraut D. Idiopathic hypoparathyroidism presenting as dementia. Br Med J. 1974 Mar 9;1(5905):429-30. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1633226/pdf/brmedj02178-0037.pdf
- Hypocalcemia Clinical Presentation. https://emedicine.medscape.com/article/241893-clinical
- Jesus JE, Landry A. Images in clinical medicine. Chvostek’s and Trousseau’s signs. N Engl J Med 2012;367:e15. doi:10.1056/NEJMicm1110569
- Hypocalcemia Workup. https://emedicine.medscape.com/article/241893-workup
- Bushinsky DA, Monk RD. Electrolyte quintet: Calcium. Lancet. 1998 Jul 25;352(9124):306-11. doi: 10.1016/s0140-6736(97)12331-5. Erratum in: Lancet 2002 Jan 19;359(9302):266.
- Slomp J, van der Voort PH, Gerritsen RT, Berk JA, Bakker AJ. Albumin-adjusted calcium is not suitable for diagnosis of hyper- and hypocalcemia in the critically ill. Crit Care Med. 2003 May;31(5):1389-93. doi: 10.1097/01.CCM.0000063044.55669.3C
- Hypocalcemia. https://emedicine.medscape.com/article/241893-overview#a1
- Lienhardt A, Bai M, Lagarde JP, Rigaud M, Zhang Z, Jiang Y, Kottler ML, Brown EM, Garabédian M. Activating mutations of the calcium-sensing receptor: management of hypocalcemia. J Clin Endocrinol Metab. 2001 Nov;86(11):5313-23. doi: 10.1210/jcem.86.11.8016
- Anast CS, Winnacker JL, Forte LR, Burns TW. Impaired release of parathyroid hormone in magnesium deficiency. J Clin Endocrinol Metab. 1976 Apr;42(4):707-17. doi: 10.1210/jcem-42-4-707
- Schimatschek HF, Rempis R. Prevalence of hypomagnesemia in an unselected German population of 16,000 individuals. Magnes Res. 2001 Dec;14(4):283-90.
- Ryzen E, Wagers PW, Singer FR, Rude RK. Magnesium deficiency in a medical ICU population. Crit Care Med. 1985 Jan;13(1):19-21. doi: 10.1097/00003246-198501000-00006
- Osteoporosis to Prevent Fractures: Screening. https://www.uspreventiveservicestaskforce.org/uspstf/recommendation/osteoporosis-screening
- Hypocalcemia Workup. https://emedicine.medscape.com/article/241893-workup#c11
- Sempos CT, Heijboer AC, Bikle DD, Bollerslev J, Bouillon R, Brannon PM, DeLuca HF, Jones G, Munns CF, Bilezikian JP, Giustina A, Binkley N. Vitamin D assays and the definition of hypovitaminosis D: results from the First International Conference on Controversies in Vitamin D. Br J Clin Pharmacol. 2018 Oct;84(10):2194-2207. doi: 10.1111/bcp.13652
- LeFevre ML; U.S. Preventive Services Task Force. Screening for vitamin D deficiency in adults: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2015 Jan 20;162(2):133-40. doi: 10.7326/M14-2450
- Sempos CT, Binkley N. 25-Hydroxyvitamin D assay standardisation and vitamin D guidelines paralysis. Public Health Nutr. 2020 May;23(7):1153-1164. doi: 10.1017/S1368980019005251
- Brooks SPJ, Sempos CT. The importance of 25-hydroxyvitamin D assay standardization and the Vitamin D Standardization Program. Journal of AOAC International 2017;100:1223-4.
- Taylor CL, Sempos CT, Davis CD, Brannon PM. Vitamin D: Moving Forward to Address Emerging Science. Nutrients. 2017 Dec 1;9(12):1308. doi: 10.3390/nu912130
- Norman AW, Henry HH. Vitamin D. In: Erdman JW, Macdonald IA, Zeisel SH, eds. Present Knowledge in Nutrition, 10th ed. Washington DC: Wiley-Blackwell, 2012.
- Hypocalcemia Workup. https://emedicine.medscape.com/article/241893-workup#c12
- Vitamin D. https://ods.od.nih.gov/factsheets/VitaminD-Consumer
- Bouillon R. Comparative analysis of nutritional guidelines for vitamin D. Nat Rev Endocrinol. 2017 Aug;13(8):466-479. doi: 10.1038/nrendo.2017.31
- Michael F. Holick and others, Evaluation, Treatment, and Prevention of Vitamin D Deficiency: an Endocrine Society Clinical Practice Guideline, The Journal of Clinical Endocrinology & Metabolism, Volume 96, Issue 7, 1 July 2011, Pages 1911–1930, https://doi.org/10.1210/jc.2011-0385
- SACN vitamin D and health report. https://www.gov.uk/government/publications/sacn-vitamin-d-and-health-report
- Thakker RV. Parathyroid disorders and diseases altering calcium metabolism. In: Warrall D, Cox T, Firth J, Benz E, eds. Oxford textbook of medicine 4th ed. Oxford: Oxford University Press, 2003
- Malabanan A, Veronikis IE, Holick MF. Redefining vitamin D insufficiency. Lancet. 1998 Mar 14;351(9105):805-6. doi: 10.1016/s0140-6736(05)78933-9
- Hopper AD, Hadjivassiliou M, Butt S, Sanders DS. Adult coeliac disease. BMJ. 2007 Sep 15;335(7619):558-62. doi: 10.1136/bmj.39316.442338.AD
- Osteoporosis. https://www.mayoclinic.org/diseases-conditions/osteoporosis/symptoms-causes/syc-20351968
- Osteoporosis. https://lpi.oregonstate.edu/mic/health-disease/osteoporosis
- Walker MD, Shane E. Hypercalcemia: A Review. JAMA. 2022 Oct 25;328(16):1624-1636. doi: 10.1001/jama.2022.18331
- Sadiq NM, Naganathan S, Badireddy M. Hypercalcemia. [Updated 2022 Sep 5]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK430714
- Zagzag J, Hu MI, Fisher SB, Perrier ND. Hypercalcemia and cancer: Differential diagnosis and treatment. CA Cancer J Clin. 2018 Sep;68(5):377-386. doi: 10.3322/caac.21489
- Patel AM, Goldfarb S. Got calcium? Welcome to the calcium-alkali syndrome. J Am Soc Nephrol. 2010 Sep;21(9):1440-3. doi: 10.1681/ASN.2010030255
- Lewis JR, Zhu K, Prince RL. Adverse events from calcium supplementation: relationship to errors in myocardial infarction self-reporting in randomized controlled trials of calcium supplementation. J Bone Miner Res. 2012 Mar;27(3):719-22. doi: 10.1002/jbmr.1484
- Moe SM. Disorders involving calcium, phosphorus, and magnesium. Prim Care. 2008 Jun;35(2):215-37, v-vi. doi: 10.1016/j.pop.2008.01.007
- Gonzalez CA, Riboli E. Diet and cancer prevention: Contributions from the European Prospective Investigation into Cancer and Nutrition (EPIC) study. Eur J Cancer. 2010 Sep;46(14):2555-62. doi: 10.1016/j.ejca.2010.07.025
- Kurahashi N, Inoue M, Iwasaki M, Sasazuki S, Tsugane AS; Japan Public Health Center-Based Prospective Study Group. Dairy product, saturated fatty acid, and calcium intake and prostate cancer in a prospective cohort of Japanese men. Cancer Epidemiol Biomarkers Prev. 2008 Apr;17(4):930-7. doi: 10.1158/1055-9965.EPI-07-2681
- Raimondi S, Mabrouk JB, Shatenstein B, Maisonneuve P, Ghadirian P. Diet and prostate cancer risk with specific focus on dairy products and dietary calcium: a case-control study. Prostate. 2010 Jul 1;70(10):1054-65. doi: 10.1002/pros.21139
- Michaelsson K, Melhus H, Warensjo Lemming E, Wold A, Byberg L. Long term calcium intake and rates of all cause and cardiovascular mortality: community based prospective longitudinal cohort study. BMJ 2013;12;346:f228. https://www.ncbi.nlm.nih.gov/pubmed/23403980?dopt=Abstract
- Tranquilli AL, Dekker G, Magee L, Roberts J, Sibai BM, Steyn W, et al. The classification, diagnosis and management of the hypertensive disorders of pregnancy: A revised statement from the ISSHP. Pregnancy Hypertens. 2014;4:97-104. https://www.ncbi.nlm.nih.gov/pubmed/26104417?dopt=Abstract
- Lowe SA, Bowyer L, Lust K, McMahon LP, Morton M, North RA, et al. SOMANZ guidelines for the management of hypertensive disorders of pregnancy 2014. Aust N Z J Obstet Gynaecol. 2015;55:e1-29. https://www.ncbi.nlm.nih.gov/pubmed/26412014?dopt=Abstract
- Bolland MJ, Avenell A, Baron JA, Grey A, MacLennan GS, Gamble GD, Reid IR.Effect of calcium supplements on risk of myocardial infarction and cardiovascular events: meta-analysis. BMJ. 2010 Jul 29;341:c3691. https://www.ncbi.nlm.nih.gov/pubmed/20671013?dopt=Abstract
- Bolland MJ, Barber PA, Doughty RN, Mason B, Horne A, Ames R, Gamble GD, Grey A, Reid IR. Vascular events in healthy older women receiving calcium supplementation: randomised controlled trial. BMJ 2008; 2;336:262-6. https://www.ncbi.nlm.nih.gov/pubmed/18198394?dopt=Abstract
- Li K, Kaaks R, Linseisen J, Rohrmann S. Associations of dietary calcium intake and calcium supplementation with myocardial infarction and stroke risk and overall cardiovascular mortality in the Heidelberg cohort of the European Prospective Investigation into Cancer and Nutrition study (EPIC-Heidelberg). Heart 2012;98:920-5. https://www.ncbi.nlm.nih.gov/pubmed/22626900?dopt=Abstract
- Xiao Q, Murphy RA, Houston DK, Harris TB, Chow WH, Park Y. Dietary and Supplemental Calcium Intake and Cardiovascular Disease Mortality: The National Institutes of Health-AARP Diet and Health Study. JAMA Intern Med. 2013 Feb 4:1-8. https://www.ncbi.nlm.nih.gov/pubmed/23381719?dopt=Abstract
- Bolland MJ, Grey A, Avenell A, Gamble GD, Reid IR. Calcium supplements with or without vitamin D and risk of cardiovascular events: reanalysis of the Women’s Health Initiative limited access dataset and meta-analysis. BMJ. 2011 Apr 19;342:d2040. https://www.ncbi.nlm.nih.gov/pubmed/21505219?dopt=Abstract
- Torfadottir JE, Steingrimsdottir L, Mucci L, Aspelund T, Kasperzyk JL, Olafsson O, Fall K, Tryggvadottir L, Harris TB, Launer L, Jonsson E, Tulinius H, Stampfer M, Adami HO, Gudnason V, Valdimarsdottir UA. Milk intake in early life and risk of advanced prostate cancer. Am J Epidemiol. 2012 Jan 15;175(2):144-53. doi: 10.1093/aje/kwr289
- Song Y, Chavarro JE, Cao Y, Qiu W, Mucci L, Sesso HD, Stampfer MJ, Giovannucci E, Pollak M, Liu S, Ma J. Whole milk intake is associated with prostate cancer-specific mortality among U.S. male physicians. J Nutr. 2013 Feb;143(2):189-96. doi: 10.3945/jn.112.168484
- Pettersson A, Kasperzyk JL, Kenfield SA, Richman EL, Chan JM, Willett WC, Stampfer MJ, Mucci LA, Giovannucci EL. Milk and dairy consumption among men with prostate cancer and risk of metastases and prostate cancer death. Cancer Epidemiol Biomarkers Prev. 2012 Mar;21(3):428-36. doi: 10.1158/1055-9965.EPI-11-1004
- Aune D, Navarro Rosenblatt DA, Chan DS, Vieira AR, Vieira R, Greenwood DC, Vatten LJ, Norat T. Dairy products, calcium, and prostate cancer risk: a systematic review and meta-analysis of cohort studies. Am J Clin Nutr. 2015 Jan;101(1):87-117. doi: 10.3945/ajcn.113.067157
- Qin LQ, He K, Xu JY. Milk consumption and circulating insulin-like growth factor-I level: a systematic literature review. Int J Food Sci Nutr. 2009;60 Suppl 7:330-40. doi: 10.1080/09637480903150114
- Rowlands MA, Gunnell D, Harris R, Vatten LJ, Holly JM, Martin RM. Circulating insulin-like growth factor peptides and prostate cancer risk: a systematic review and meta-analysis. Int J Cancer. 2009 May 15;124(10):2416-29. doi: 10.1002/ijc.24202
- Moreno J, Krishnan AV, Peehl DM, Feldman D. Mechanisms of vitamin D-mediated growth inhibition in prostate cancer cells: inhibition of the prostaglandin pathway. Anticancer Res. 2006 Jul-Aug;26(4A):2525-30.
- Brändstedt J, Almquist M, Manjer J, Malm J. Vitamin D, PTH, and calcium in relation to survival following prostate cancer. Cancer Causes Control. 2016 May;27(5):669-77. doi: 10.1007/s10552-016-0740-7
- Brändstedt J, Almquist M, Ulmert D, Manjer J, Malm J. Vitamin D, PTH, and calcium and tumor aggressiveness in prostate cancer: a prospective nested case-control study. Cancer Causes Control. 2016 Jan;27(1):69-80. doi: 10.1007/s10552-015-0684-3
- Rowland GW, Schwartz GG, John EM, Ingles SA. Protective effects of low calcium intake and low calcium absorption vitamin D receptor genotype in the California Collaborative Prostate Cancer Study. Cancer Epidemiol Biomarkers Prev. 2013 Jan;22(1):16-24. doi: 10.1158/1055-9965.EPI-12-0922-T
- Baron JA, Beach M, Wallace K, Grau MV, Sandler RS, Mandel JS, Heber D, Greenberg ER. Risk of prostate cancer in a randomized clinical trial of calcium supplementation. Cancer Epidemiol Biomarkers Prev. 2005 Mar;14(3):586-9. doi: 10.1158/1055-9965.EPI-04-0319
- Chung M, Balk EM, Brendel M, Ip S, Lau J, Lee J, Lichtenstein A, Patel K, Raman G, Tatsioni A, Terasawa T, Trikalinos TA. Vitamin D and calcium: a systematic review of health outcomes. Evid Rep Technol Assess (Full Rep). 2009 Aug;(183):1-420. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4781105
- Huncharek M, Muscat J, Kupelnick B. Dairy products, dietary calcium and vitamin D intake as risk factors for prostate cancer: a meta-analysis of 26,769 cases from 45 observational studies. Nutr Cancer. 2008;60(4):421-41. doi: 10.1080/01635580801911779
- Hypercalcemia. https://emedicine.medscape.com/article/240681-overview