What is cannabis oil
Cannabis oil also known as “hash oil”, is a highly potent delta-9-tetrahydrocannabinol (THC) concentrate that contains up to 90% THC 1. THC (delta-9-tetrahydrocannabinol) is the psychoactive component that produces the “high” associated with marijuana use. When taken by mouth, the main psychoactive ingredient in cannabis, THC (delta-9-tetrahydrocannabinol), is processed by the liver and changed into a different psychoactive chemical (11-hydroxy-delta-tetrahydrocannabinol). The U.S. Food and Drug Administration (FDA) has only approved two cannabinoid drugs (Dronabinol and Nabilone) that contain THC. These drugs treat nausea caused by chemotherapy and increase appetite in patients who have severe weight loss from HIV/AIDS. Dronabinol, a synthetically produced delta-9-THC, was approved in the United States in 1986 as an antiemetic to be used in cancer chemotherapy. Nabilone, a synthetic derivative of delta-9-THC, was first approved in Canada in 1982 and is now also available in the United States 2. Dronabinol (synthetic THC) is used to treat loss of appetite that causes weight loss in people with AIDS. Dronabinol is also used to treat severe nausea and vomiting caused by cancer chemotherapy. Nabilone (a synthetic substance similar to THC) is used to treat nausea and vomiting caused by cancer chemotherapy in people who have already taken other medications to treat this type of nausea and vomiting without good results. Nabiximols, a cannabis extract given as a mouth spray, was shown in a small randomized, placebo-controlled, double-blinded clinical trial in Spain to treat nausea and vomiting caused by chemotherapy 3. American Society of Clinical Oncology antiemetic guidelines updated in 2017 4 state that evidence remains insufficient to recommend medical marijuana for either the prevention or treatment of nausea or vomiting in patients with cancer who receive chemotherapy or radiation therapy. You cannot get cannabis-based medicine from a family physician – it can only be prescribed by a specialist doctor. And it is only likely to be prescribed for a small number of patients.
There is also a liquid drug that contains cannabidiol (CBD) or “hemp oil”. CBD (cannabidiol) treats two forms of severe childhood epilepsy. A prescription for medical cannabis would only be given when it was believed to be in your best interests, and when other treatments had not worked or were not suitable. Scientists are doing more research with marijuana and cannabis-based drugs to treat many diseases and conditions including cancer pain, the eye disease glaucoma, appetite loss in people with HIV or AIDS, and epilepsy in children. The FDA has not approved cannabis or cannabinoids for use as a cancer treatment. There is growing interest in treating children for symptoms such as nausea with cannabis and cannabinoids, but studies are limited. The American Academy of Pediatrics has not endorsed cannabis and cannabinoid use because of concerns about its effect on brain development.
In the 2018 United States Farm Bill, the term hemp is used to describe cultivars of the cannabis species that contain less than 0.3% THC. Hemp oil or cannabidiol (CBD) oil are products manufactured from extracts of industrial hemp (i.e., low-THC cannabis cultivars), whereas hemp seed oil is an edible fatty oil that is essentially cannabinoid-free. Some products contain other botanical extracts and/or over-the-counter analgesics, and are readily available as oral and topical tinctures or other formulations often advertised for pain management and other purposes. Hemp products containing less than 0.3% of THC (delta-9-tetrahydrocannabinol) are not scheduled drugs and could be considered as Farm Bill compliant. Hemp is not a controlled substance; however, CBD is a controlled substance.
Highly concentrated THC or cannabidiol (CBD) oil extracts are being illegally promoted as potential cancer cures 5. These oils have not been evaluated in any clinical trials for anticancer activity or safety. Because CBD is a potential inhibitor of certain cytochrome P450 enzymes, highly concentrated CBD oils used concurrently with conventional therapies that are metabolized by these enzymes could potentially increase toxicity or decrease the effectiveness of these therapies 6.
Table 1. Medicinal cannabis products
| Name/Material | Constituents/Composition | |
| Cannabis species, including C. sativa | Cannabinoids; also terpenoids and flavonoids | |
| • Hemp (aka industrial hemp) | Low THC (<0.3%); high CBD | |
| • Marijuana/marihuana. Marijuana refers to the dried leaves, flowers, stems, and seeds from the Cannabis sativa or Cannabis indica plant. | High THC (>0.3%); low CBD | |
| Nabiximols (trade name: Sativex) | Mixture of ethanol extracts of Cannabis species; contains THC and CBD in a 1:1 ratio | |
| Hemp oil/CBD oil | Solution of a solvent extract from Cannabis flowers and/or leaves dissolved in an edible oil; typically containing 1%–5% CBD | |
| Hemp seed oil | Edible, fatty oil produced from Cannabis seeds; contains no or only traces of cannabinoids | |
| Dronabinol (trade names: Marinol and Syndros) | Synthetic THC | |
| Nabilone (trade names: Cesamet and Canemes) | Synthetic THC analog | |
| Cannabidiol (trade name: Epidiolex) | Highly purified (>98%), plant-derived CBD | |
What are cannabinoids?
Cannabinoids also known as phytocannabinoids, are are a group of chemicals found in the cannabis plant (Cannabis sativa or Cannabis indica) that cause drug-like effects in your body, including the central nervous system and the immune system. Over 100 cannabinoids have been found in Cannabis. The main psychoactive cannabinoid in cannabis is delta-9-tetrahydrocannabinol (THC). Another active cannabinoid is cannabidiol (CBD).
Cannabinoids health benefits
Drugs containing cannabinoids may be helpful in treating certain rare forms of epilepsy, nausea and vomiting associated with cancer chemotherapy, and loss of appetite and weight loss associated with HIV/AIDS. In addition, some evidence suggests modest benefits of cannabis or cannabinoids for chronic pain and multiple sclerosis symptoms. Cannabis isn’t helpful for glaucoma.
At present, there is insufficient evidence to recommend inhaling cannabis as a treatment for cancer-related symptoms or cancer treatment–related symptoms or cancer treatment-related side effects; however, additional research is needed. Research on cannabis or cannabinoids for other conditions is in its early stages.
Cannabis and cannabinoids health benefits current state of evidence 7:
- There is conclusive or substantial evidence that cannabis or cannabinoids are effective:
- For the treatment of chronic pain in adults (cannabis)
- As antiemetics in the treatment of chemotherapy-induced nausea and vomiting (oral cannabinoids)
- For improving patient-reported multiple sclerosis spasticity symptoms (oral cannabinoids)
- There is moderate evidence that cannabis or cannabinoids are effective for:
- Improving short-term sleep outcomes in individuals with sleep disturbance associated with obstructive sleep apnea syndrome, fibromyalgia, chronic pain, and multiple sclerosis
(cannabinoids, primarily nabiximols)
- Improving short-term sleep outcomes in individuals with sleep disturbance associated with obstructive sleep apnea syndrome, fibromyalgia, chronic pain, and multiple sclerosis
- There is limited evidence that cannabis or cannabinoids are effective for:
- Increasing appetite and decreasing weight loss associated with HIV/AIDS (cannabis and oral cannabinoids)
- Improving clinician-measured multiple sclerosis spasticity symptoms (oral cannabinoids)
- Improving symptoms of Tourette syndrome (THC capsules)
- Improving anxiety symptoms, as assessed by a public speaking test, in individuals with social anxiety disorders (cannabidiol)
- Improving symptoms of posttraumatic stress disorder (nabilone; a single, small fair-quality trial)
- There is limited evidence of a statistical association between cannabinoids and:
- Better outcomes (i.e., mortality, disability) after a traumatic brain injury or intracranial hemorrhage
- There is limited evidence that cannabis or cannabinoids are ineffective for:
- Improving symptoms associated with dementia (cannabinoids)
- Improving intraocular pressure associated with glaucoma (cannabinoids)
- Reducing depressive symptoms in individuals with chronic pain or multiple sclerosis (nabiximols, dronabinol, and nabilone)
- There is no or insufficient evidence to support or refute the conclusion that cannabis or cannabinoids are an effective treatment for:
- Cancers, including glioma (cannabinoids)
Cancer-associated anorexia cachexia syndrome and anorexia nervosa (cannabinoids) - Symptoms of irritable bowel syndrome (dronabinol)
- Epilepsy (cannabinoids)
- Spasticity in patients with paralysis due to spinal cord injury (cannabinoids)
- Symptoms associated with amyotrophic lateral sclerosis (cannabinoids)
- Chorea and certain neuropsychiatric symptoms associated with Huntington’s disease (oral cannabinoids)
- Motor system symptoms associated with Parkinson’s disease or the levodopa-induced dyskinesia (cannabinoids)
- Dystonia (nabilone and dronabinol)
- Achieving abstinence in the use of addictive substances (cannabinoids)
- Mental health outcomes in individuals with schizophrenia or schizophreniform psychosis (cannabidiol)
- Cancers, including glioma (cannabinoids)
Stimulating appetite
The ability of cannabinoids to increase appetite has been studied:
- Delta-9-tetrahydrocannabinol (THC) taken by mouth: Despite patients’ great interest in oral preparations of cannabis to improve appetite, there is only one trial of cannabis extract used for appetite stimulation. In a randomized controlled trial, researchers compared the safety and effectiveness of orally administered cannabis extract (2.5 mg THC and 1 mg CBD), THC (2.5 mg), or placebo for the treatment of cancer-related anorexia-cachexia in 243 patients with advanced cancer who received treatment twice daily for 6 weeks. Results demonstrated that although these agents were well tolerated by these patients, no differences were observed in patient appetite or quality of life among the three groups at this dose level and duration of intervention 8. A clinical trial compared delta-9-THC (dronabinol) and a standard drug (megestrol, an appetite stimulant) in patients with advanced cancer and loss of appetite 9. Results showed that delta-9-THC did not help increase appetite or weight gain in advanced cancer patients compared with megestrol 9.
- Four controlled trials have assessed the effect of oral THC on measures of appetite, food appreciation, calorie intake, and weight loss in patients with advanced malignancies. Three relatively small, placebo-controlled trials (N = 52; N = 46; N = 65) each found that oral THC produced improvements in one or more of these outcomes 10, 11, 12. The one study that used an active control evaluated the efficacy of dronabinol alone or with megestrol acetate compared with that of megestrol acetate alone for managing cancer-associated anorexia 9. In this randomized, double-blind study of 469 adults with advanced cancer and weight loss, patients received 2.5 mg of oral THC twice daily, 800 mg of oral megestrol daily, or both. Appetite increased by 75% in the megestrol group and weight increased by 11%, compared with a 49% increase in appetite and a 3% increase in weight in the oral THC group after 8 to 11 weeks of treatment. The between-group differences were statistically significant in favor of megestrol acetate. Furthermore, the combined therapy did not offer additional benefits beyond those provided by megestrol acetate alone. The authors concluded that dronabinol did little to promote appetite or weight gain in advanced cancer patients compared with megestrol acetate.
- Inhaled cannabis: There are no published studies of the effect of inhaled cannabis on cancer patients with loss of appetite.
Pain relief
Research has been done on the effects of cannabis or cannabinoids on chronic pain, particularly neuropathic pain (pain associated with nerve injury or damage). A 2018 review looked at 47 studies (4,743 participants) of cannabis or cannabinoids for various types of chronic pain other than cancer pain and found evidence of a small benefit. Twenty-nine percent of people taking cannabis/cannabinoids had a 30 percent reduction in their pain whereas 26 percent of those taking a placebo (an inactive substance) did. The difference may be too small to be meaningful to patients. Adverse events (side effects) were more common among people taking cannabis/cannabinoids than those taking placebos. A 2018 review of 16 studies of cannabis-based medicines for neuropathic pain, most of which tested a cannabinoid preparation called nabiximols (brand name Sativex; a mouth spray containing both THC and CBD that is approved in some countries but not in the United States), found low- to moderate-quality evidence that these medicines produced better pain relief than placebos did. However, the data could not be considered reliable because the studies included small numbers of people and may have been biased. People taking cannabis-based medicines were more likely than those taking placebos to drop out of studies because of side effects. A 2015 review of 28 studies (2,454 participants) of cannabinoids in which chronic pain was assessed found the studies generally showed improvements in pain measures in people taking cannabinoids, but these did not reach statistical significance in most of the studies. However, the average number of patients who reported at least a 30 percent reduction in pain was greater with cannabinoids than with placebo.
- Vaporized cannabis with opioids: In a study of 21 patients with chronic pain, vaporized Cannabis given with morphine relieved pain better than morphine alone, while vaporized Cannabis given with oxycodone did not give greater pain relief. Further studies are needed.
- Inhaled cannabis: Randomized controlled trials of inhaled Cannabis in patients with peripheral neuropathy or other nerve pain found pain was reduced in patients who received inhaled cannabis compared with those who received placebo.
- Cannabis plant extract: A study of Cannabis extract that was sprayed under the tongue found it helped patients with advanced cancer whose pain was not relieved by strong opioids alone. In another study, patients who were given lower doses of cannabinoid spray showed better pain control and less sleep loss than patients who received a placebo. Control of cancer-related pain in some patients was better without the need for higher doses of Cannabis extract spray or higher doses of their other pain medicines. Adverse events were related to high doses of cannabinoid spray.
- Delta-9-tetrahydrocannabinol (THC) taken by mouth: Two small clinical trials of oral delta-9-tetrahydrocannabinol (THC) showed that it relieved cancer pain. In the first study, patients had good pain relief, less nausea and vomiting, and better appetite. A second study showed that delta-9-tetrahydrocannabinol (THC) could relieve pain as well as codeine. An observational study of nabilone also showed that it relieved cancer pain along with nausea, anxiety, and distress when compared with no treatment. However, neither dronabinol nor nabilone is approved by the FDA for pain relief.
A 2015 review of 23 studies (1,326 participants) on the cannabinoids dronabinol or nabilone for treating nausea and vomiting related to cancer chemotherapy found that they were more helpful than a placebo and similar in effectiveness to other medicines used for this purpose. More people had side effects such as dizziness or sleepiness, though, when taking the cannabinoid medicines.
The research on dronabinol and nabilone for treating nausea and vomiting related to cancer chemotherapy was done primarily in the 1980s and 1990s and reflects the types of chemotherapy treatments and choices of anti-nausea medicines available at that time rather than current ones.
Anxiety
Cannabis and cannabinoids have been studied in the treatment of anxiety. A small amount of evidence from studies in people suggests that cannabis or cannabinoids might help to reduce anxiety. One study of 24 people with social anxiety disorder found that they had less anxiety in a simulated public speaking test after taking cannabidiol (CBD) than after taking a placebo. Four studies have suggested that cannabinoids may be helpful for anxiety in people with chronic pain; the study participants did not necessarily have anxiety disorders.
- Inhaled cannabis: A small case series found that patients who inhaled Cannabis had improved mood, improved sense of well-being, and less anxiety. In another study, 74 patients newly diagnosed with head and neck cancer who were cannabis users were matched to 74 nonusers. The cannabis users had markedly lower anxiety or depression and less pain or discomfort than the nonusers. The cannabis users were also less tired, had more appetite, and reported greater feelings of well-being.
Posttraumatic Stress Disorder (PTSD)
Some people with PTSD have used cannabis or products made from it to try to relieve their symptoms and believe that it can help, but there’s been little research on whether it’s actually useful. In one very small study (10 people), the cannabinoid nabilone was more effective than a placebo at relieving PTSD-related nightmares. Observational studies (studies that collected data on people with PTSD who made their own choices about whether to use cannabis) haven’t provided clear evidence on whether cannabis is helpful or harmful for PTSD symptoms.
Sleep problems
Many studies of cannabis or cannabinoids in people with health problems (such as multiple sclerosis, PTSD, or chronic pain) have looked at effects on sleep. Often, there’s been evidence of better sleep quality, fewer sleep disturbances, or decreased time to fall asleep in people taking cannabis/cannabinoids. However, it’s uncertain whether the cannabis products affected sleep directly or whether people slept better because the symptoms of their illnesses had improved. The effects of cannabis/cannabinoids on sleep problems in people who don’t have other illnesses are uncertain.
Helping to decrease opioid use
There’s evidence from studies in animals that administering THC along with opioids may make it possible to control pain with a smaller dose of opioids. A 2017 review looked at studies in people in which cannabinoids were administered along with opioids to treat pain. These studies were designed to determine whether cannabinoids could make it possible to control pain with smaller amounts of opioids. There were 9 studies (750 total participants), of which 3 (642 participants) used a high-quality study design in which participants were randomly assigned to receive cannabinoids or a placebo. The results were inconsistent, and none of the high-quality studies indicated that cannabinoids could lead to decreased opioid use.
Researchers have looked at statistical data on groups of people to see whether access to cannabis (for example, through “medical marijuana laws”—state laws that allow patients with certain medical conditions to get access to cannabis)—is linked with changes in opioid use or with changes in harm associated with opioids. The findings have been inconsistent.
- States with medical marijuana laws were found to have lower prescription rates both for opioids and for all drugs that cannabis could substitute for among people on Medicare. However, data from a national survey (not limited to people on Medicare) showed that users of medical marijuana were more likely than nonusers to report taking prescription drugs.
- An analysis of data from 1999 to 2010 indicated that states with medical marijuana laws had lower death rates from overdoses of opioid pain medicines, but when a similar analysis was extended through 2017, it showed higher death rates from this kind of overdose.
- An analysis of survey data from 2004 to 2014 found that passing of medical marijuana laws was not associated with less nonmedical prescription opioid use. Thus, people with access to medical marijuana did not appear to be substituting it for prescription opioids.
Epilepsy
Cannabinoids, primarily CBD (cannabidiol), have been studied for the treatment of seizures associated with forms of epilepsy that are difficult to control with other medicines. Epidiolex (oral CBD) has been approved by the FDA for the treatment of seizures associated with two epileptic encephalopathies: Lennox-Gastaut syndrome and Dravet syndrome. Epileptic encephalopathies are a group of seizure disorders that start in childhood and involve frequent seizures along with severe impairments in cognitive development. Not enough research has been done on cannabinoids for other, more common forms of epilepsy to allow conclusions to be reached about whether they’re helpful for these conditions.
Glaucoma
Glaucoma is a group of diseases that can damage the eye’s optic nerve, leading to vision loss and blindness. Glaucoma is a leading cause of blindness in the United States. Glaucoma usually happens when the fluid pressure inside the eyes slowly rises, damaging the optic nerve. Often there are no symptoms at first. Without treatment, people with glaucoma will slowly lose their peripheral, or side vision. They seem to be looking through a tunnel. Over time, straight-ahead vision may decrease until no vision remains. Early treatment can often prevent severe loss of vision. Lowering pressure in the eye can slow progression of the disease.
Studies conducted in the 1970s and 1980s showed that cannabis or substances derived from it could lower pressure in the eye, but not as effectively as treatments already in use. One limitation of cannabis-based products is that they only affect pressure in the eye for a short period of time. A recent animal study showed that CBD, applied directly to the eye, may cause an undesirable increase in pressure in the eye.
HIV/AIDS appetite and weight loss symptoms
Unintentional weight loss can be a problem for people with HIV/AIDS. In 1992, the FDA approved the cannabinoid dronabinol for the treatment of loss of appetite associated with weight loss in people with HIV/AIDS. This approval was based primarily on a study of 139 people that assessed effects of dronabinol on appetite and weight changes. There have been a few other studies of cannabis or cannabinoids for appetite and weight loss in people with HIV/AIDS, but they were short and only included small numbers of people, and their results may have been biased. Overall, the evidence that cannabis/cannabinoids are beneficial in people with HIV/AIDS is limited.
Inflammatory bowel disease
Inflammatory bowel disease is the name for a group of conditions in which the digestive tract becomes inflamed. Ulcerative colitis and Crohn’s disease are the most common types. Symptoms may include abdominal pain, diarrhea, loss of appetite, weight loss, and fever. The symptoms can range from mild to severe, and they can come and go, sometimes disappearing for months or years and then returning.
A 2018 review looked at 3 studies (93 total participants) that compared smoked cannabis or cannabis oil with placebos in people with active Crohn’s disease. There was no difference between the cannabis/cannabis oil and placebo groups in clinical remission of the disease. Some people using cannabis or cannabis oil had improvements in symptoms, but some had undesirable side effects. It was uncertain whether the potential benefits of cannabis or cannabis oil were greater than the potential harms.
A 2018 review examined 2 studies (92 participants) that compared smoked cannabis or CBD capsules with placebos in people with active ulcerative colitis. In the CBD study, there was no difference between the two groups in clinical remission, but the people taking CBD had more side effects. In the smoked cannabis study, a measure of disease activity was lower after 8 weeks in the cannabis group; no information on side effects was reported.
Irritable bowel syndrome
Irritable bowel syndrome (IBS) is defined as repeated abdominal pain with changes in bowel movements (diarrhea, constipation, or both). It’s one of a group of functional disorders of the gastrointestinal tract that relate to how the brain and gut work together. Although there’s interest in using cannabis/cannabinoids for symptoms of IBS, there’s been little research on their use for this condition in people. Therefore, it’s unknown whether cannabis or cannabinoids can be helpful.
Movement disorders due to Tourette syndrome
A 2015 review of 2 small placebo-controlled studies with 36 participants suggested that synthetic THC capsules may be associated with a significant improvement in tic severity in patients with Tourette syndrome.
Multiple sclerosis
Several cannabis/cannabinoid preparations have been studied for multiple sclerosis symptoms, including dronabinol, nabilone, cannabis extract, nabiximols (brand name Sativex; a mouth spray containing THC and CBD that is approved in more than 25 countries outside the United States), and smoked cannabis. A review of 17 studies of a variety of cannabinoid preparations with 3,161 total participants indicated that cannabinoids caused a small improvement in spasticity (as assessed by the patient), pain, and bladder problems in people with multiple sclerosis, but cannabinoids didn’t significantly improve spasticity when measured by objective tests.
A review of 6 placebo-controlled clinical trials with 1,134 total participants concluded that cannabinoids (nabiximols, dronabinol, and THC/CBD) were associated with a greater average improvement on the Ashworth scale for spasticity in multiple sclerosis patients compared with placebo, although this did not reach statistical significance.
Evidence-based guidelines issued in 2014 by the American Academy of Neurology concluded that nabiximols is probably effective for improving subjective spasticity symptoms, probably ineffective for reducing objective spasticity measures or bladder incontinence, and possibly ineffective for reducing multiple sclerosis–related tremor. Based on two small studies, the guidelines concluded that the data are inadequate to evaluate the effects of smoked cannabis in people with multiple sclerosis.
A 2010 analysis of 3 studies (666 participants) of nabiximols in people with multiple sclerosis and spasticity found that nabiximols reduced subjective spasticity, usually within 3 weeks, and that about one-third of people given nabiximols as an addition to other treatment would have at least a 30 percent improvement in spasticity. Nabiximols appeared to be reasonably safe.
What is cannabis?
Cannabis also known as marijuana, is a plant first grown in Central Asia that is now grown in many parts of the world. Marijuana refers to the dried leaves, flowers, stems, and seeds from the Cannabis sativa or Cannabis indica plant. The leaves and flowering tops of cannabis plant contains about 540 chemical substances 13. The cannabis plant makes a resin (thick substance) that contains compounds called cannabinoids, which acts on the endocannabinoid system (see Figure 1 below). The principal cannabinoids appear to be delta-9-tetrahydrocannabinol (THC), cannabinol (CBN) and cannabidiol (CBD) 14, although the relative abundance of these and other phytocannabinoids can vary depending on a number of factors such as the cannabis strain, the soil and climate conditions, and the cultivation techniques 15. Other phytocannabinoids found in cannabis include cannabigerol (CBG), cannabichromene (CBC), tetrahydrocannabivarin (THCV) and many others 16. In the living cannabis plant, these phytocannabinoids exist as both inactive monocarboxylic acids (e.g. tetrahydrocannabinolic acid, THCA) and as active decarboxylated forms (e.g. THC); however, heating (at temperatures above 120 °C) promotes decarboxylation (e.g. THCA to THC) 17. Furthermore, pyrolysis (such as by smoking) transforms each of the hundreds of compounds in cannabis into a number of other compounds, many of which remain to be characterized both chemically and pharmacologically. Therefore, cannabis can be considered a very crude drug containing a very large number of chemical and pharmacological constituents, the properties of which are only slowly being understood.
Some cannabinoids are psychoactive (affecting your mind or mood). The use of cannabis and cannabis oil containing specific cannabinoids produces mental and physical effects such as altered sensory perception and euphoria when consumed. Cannabis is a widely used recreational drug which alters sensory perception and causes euphoria 18. Some cannabis plants contain very little THC. Under U.S. law, these plants are considered “industrial hemp” rather than marijuana. The word “marijuana” refers to parts of or products from the plant Cannabis sativa that contain substantial amounts of tetrahydrocannabinol (THC). In the United States, cannabis is a controlled substance and has been classified as a Schedule 1 agent (a drug with a high potential for abuse and no accepted medical use). By federal law, possessing cannabis (marijuana) is illegal in the United States unless it is used in approved research settings. However, a growing number of states, territories, and the District of Columbia have passed laws to legalize medical marijuana.
Hemp is a mixture of the cannabis plant with very low levels of psychoactive compounds. Hemp oil or cannabidiol (CBD) are products made from extracts of industrial hemp, while hemp seed oil is an edible fatty oil that contains only scant or no cannabinoids. Hemp is not a controlled substance, but CBD is.
Clinical trials that study cannabis for cancer treatment are limited. To do clinical trials research with plant-derived cannabis in the United States, researchers must file an Investigational New Drug (IND) application with the FDA, have a Schedule 1 license from the U.S. Drug Enforcement Administration, and have approval from the National Institute on Drug Abuse.
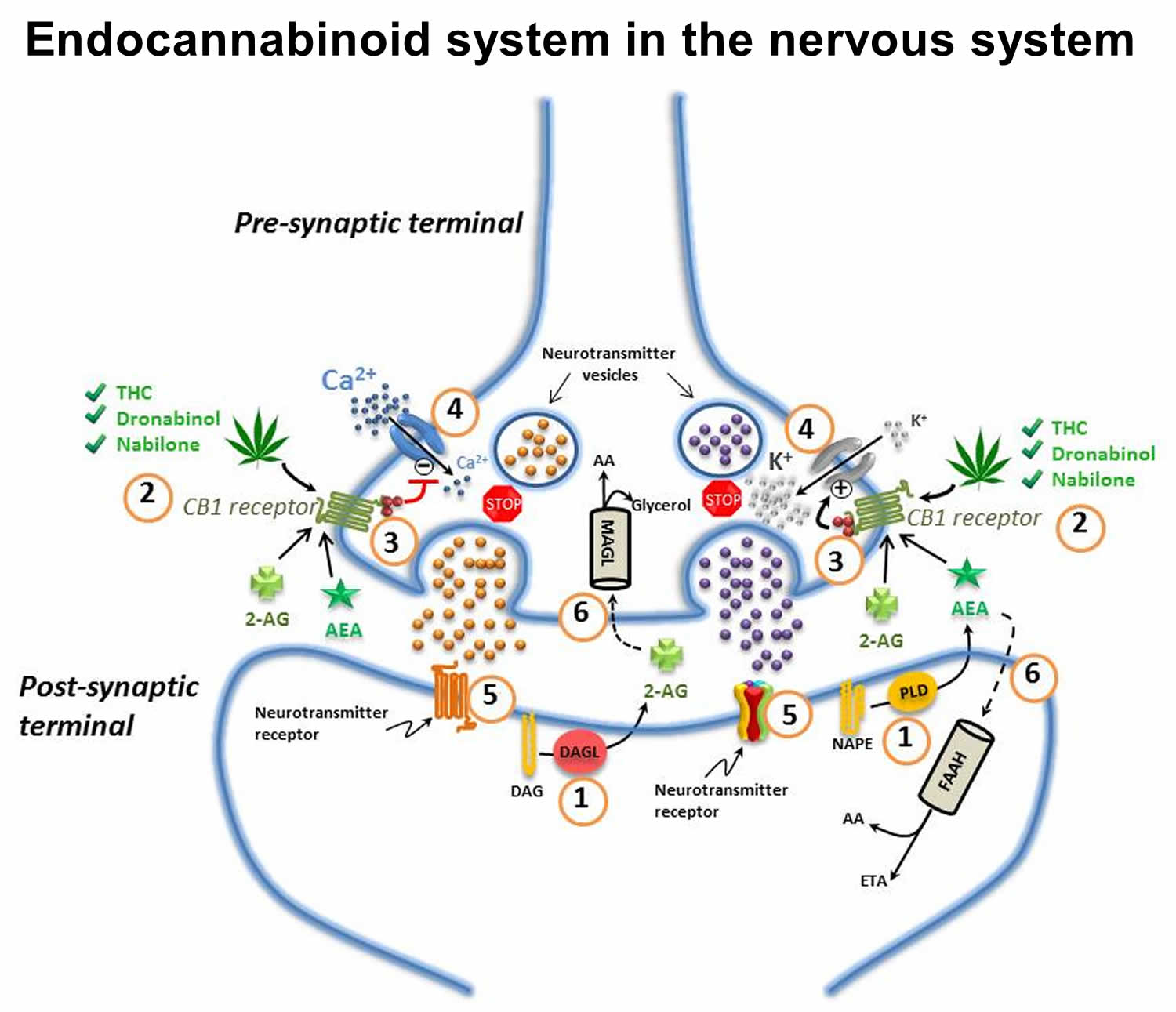
Endocannabinoid system in the nervous system
The endocannabinoid system (Figure 1) is an ancient, evolutionarily conserved, and ubiquitous lipid signaling system found in all vertebrates, and which appears to have important regulatory functions throughout the human body 19. The endocannabinoid system has been implicated in a very broad number of physiological as well as pathophysiological processes including nervous system development, immune function, inflammation, appetite, metabolism and energy, homeostasis, cardiovascular function, digestion, bone development and bone density, synaptic plasticity and learning, pain, reproduction, psychiatric disease, psychomotor behaviour, memory, wake/sleep cycles, and the regulation of stress and emotional state/mood 20, 21, 22. Furthermore, there is strong evidence that dysregulation of the endocannabinoid system contributes to many human diseases including pain, inflammation, psychiatric disorders and neurodegenerative diseases 23.
The endocannabinoid system consists mainly of: the cannabinoid 1 and 2 (CB1 and CB2) receptors; the cannabinoid receptor ligands N-arachidonoylethanolamine (“anandamide”) and 2-arachidonoylglycerol (2-AG); the endocannabinoid-synthesizing enzymes N-acyltransferase, phospholipase D, phospholipase C-β and diacylglycerol-lipase (DAGL); and the endocannabinoid-degrading enzymes fatty acid amide hydrolase (FAAH) and monoacylglycerol lipase (MAGL) (Figure 1) 20. Anandamide and 2-arachidonoylglycerol (2-AG) are considered the primary endogenous activators of cannabinoid signaling, but other endogenous molecules, which exert “cannabinoid-like” effects, have also been described. These other molecules include 2-arachidonoylglycerol ether (noladin ether), N -arachidonoyl-dopamine, virodhamine, N -homo-γ-linolenoylethanolamine and N-docosatetraenoylethanolamine 20. Other molecules such as palmitoylethanolamide (PEA) and oleoylethanolamide (OEA) do not appear to bind to cannabinoid receptors but rather to a specific isozyme belonging to a class of nuclear receptors/transcription factors known as peroxisome proliferator-activated receptors (PPARs) 24. These fatty acyl ethanolamides may, however, potentiate the effect of anandamide by competitive inhibition of FAAH, and/or through direct allosteric effects on other receptors such as the transient receptor potential vanilloid (TRPV1) channel 25. This type of effect has been generally referred to as the so-called “entourage effect” 25. The term “entourage effect” is also used in the context of the interactions between phytocannabinoids and terpenes in a physiological system.
Most tissues contain a functional endocannabinoid system with the CB1 and CB2 receptors having distinct patterns of tissue expression. The CB1 receptor is one of the most abundant G-protein coupled receptors in the central and peripheral nervous systems 26. It has been detected in the cerebral cortex, hippocampus, amygdala, basal ganglia, substantia nigra pars reticulata, internal and external segments of the globus pallidus and cerebellum (molecular layer), and at central and peripheral levels of the pain pathways including the periaqueductal gray matter, the rostral ventrolateral medulla, the dorsal primary afferent spinal cord regions including peripheral nociceptors, and spinal interneurons 27. CB1 receptor density is highest in the cingulate gyrus, the frontal cortex, the hippocampus, the cerebellum, and the basal ganglia 23. Moderate levels of CB1 receptor expression are found in the basal forebrain, amygdala, nucleus accumbens, periaqueductal grey, and hypothalamus and much lower expression levels of the receptor are found in the midbrain, the pons, and the medulla/brainstem 23. Relatively little CB1 receptor expression is found in the thalamus and the primary motor cortex 23. The CB1 receptor is also expressed in many other organs and tissues including adipocytes, leukocytes, spleen, heart, lung, the gastrointestinal tract (liver, pancreas, stomach, and the small and large intestine), kidney, bladder, reproductive organs, skeletal muscle, bone, joints, and skin 28. CB2 receptors are most highly concentrated in the tissues and cells of the immune system such as the leukocytes and the spleen, but can also be found in bone and to a lesser degree in liver and in nerve cells including astrocytes, oligodendrocytes and microglia, and even some neuronal sub-populations 29.
Besides the well-known CB1 and CB2 receptors, a number of different cannabinoids are believed to bind to a number of other molecular targets. Such targets include the third putative cannabinoid receptor GPR55 (G protein-coupled receptor 55), the transient receptor potential (TRP) cation channel family, and a class of nuclear receptors/transcription factors known as the PPARs, as well as 5-HT1A receptors, the α2 adrenoceptors, adenosine and glycine receptors 30. Modulation of these other cannabinoid targets adds additional layers of complexity to the known myriad effects of cannabinoids.
Dysregulation of the endocannabinoid system appears to be connected to a number of pathological conditions, with the changes in the functioning of the system being either protective or harmful 31. Modulation of the endocannabinoid system either through the targeted inhibition of specific metabolic pathways, and/or directed agonism or antagonism of its receptors may hold therapeutic promise 32. However, a major and consistent therapeutic challenge confronting the routine use of (THC-predominant) cannabis and psychoactive cannabinoids (e.g. THC) in the clinic has remained that of achieving selective targeting of the site of disease or symptoms and the sparing of other bodily regions such as the mood and cognitive centers of the brain 27. Despite this significant challenge, emerging evidence from clinical studies of smoked or vaporized (THC-predominant) cannabis for chronic non-cancer pain (mainly neuropathic pain) suggests that use of very low doses of THC (< 3 mg/dose) may confer therapeutic benefits with minimal psychoactive side effects 33.
Figure 1. Endocannabinoid system in the nervous system
Footnotes: (1) Endocannabinoids are manufactured “on-demand” (e.g. in response to an action potential in neurons) in the post-synaptic terminals: anandamide (AEA) is generated from phospholipase-D (PLD)-mediated hydrolysis of the membrane lipid N-arachidonoylphosphatidylethanolamine (NAPE); 2-AG from the diacylglycerol lipase (DAGL)-mediated hydrolysis of the membrane lipid diacylglycerol (DAG); (2) These endocannabinoids [anandamide (AEA) and 2-arachidonoylglycerol (2-AG)] diffuse retrogradely towards the pre-synaptic terminals and like exogenous cannabinoids such as THC (from cannabis), dronabinol, and nabilone, they bind to and activate the pre-synaptic G-protein-coupled CB1 receptors; (3) Binding of phytocannabinoid and endocannabinoid agonists to the CB1 receptors triggers Gi/Go protein signalling that, for example, inhibits adenylyl cyclase, thus decreasing the formation of cyclic AMP and the activity of protein kinase A; (4) Activation of the CB1 receptor also results in Gi/Go protein-dependent opening of inwardly-rectifying K+ channels (depicted with a “+”) causing a hyperpolarization of the pre-synaptic terminals, and the closing of Ca2+ channels (depicted with a “-“), arresting the release of stored excitatory and inhibitory neurotransmitters (e.g. glutamate, GABA, 5-HT, acetylcholine, noradrenaline, dopamine, D-aspartate and cholecystokinin) which (5) once released, diffuse and bind to post-synaptic receptors; (6) Anandamide and 2-AG re-enter the post- or pre-synaptic nerve terminals (possibly through the actions of a specialized transporter depicted by a “dashed” line) where they are respectively catabolized by fatty acid amide hydrolase (FAAH) or monoacylglycerol lipase (MAGL) to yield either arachidonic acid (AA) and ethanolamine (ETA), or arachidonic acid (AA) and glycerol.
Endocannabinoid signaling is rapidly terminated by the action of two hydrolytic enzymes: fatty acid amide hydrolase (FAAH) and monoacylglycerol lipase (MAGL) 21. FAAH is primarily localized post-synaptically 34 and preferentially degrades anandamide 35; MAGL is primarily localized pre-synaptically 34 and favors the catabolism of 2-AG 35. Signal termination is important in ensuring that biological activities are properly regulated and prolonged signaling activity, such as by the use of cannabis, can have potentially deleterious effects 36.
[Source 37 ]Cannabis pharmacology
When oral cannabis is ingested, there is a low (6%–20%) and variable oral bioavailability 38. Peak plasma concentrations of delta-9-tetrahydrocannabinol (THC) occur after 1 to 6 hours and remain elevated with a terminal half-life of 20 to 30 hours. Taken by mouth, delta-9-THC is initially metabolized in the liver to 11-OH-THC (11-hydroxy-delta-tetrahydrocannabinol), a potent psychoactive metabolite. Inhaled cannabinoids are rapidly absorbed into the bloodstream with a peak concentration in 2 to 10 minutes, declining rapidly for a period of 30 minutes and with less generation of the psychoactive 11-OH-THC metabolite.
Cannabinoids are known to interact with the hepatic cytochrome P450 enzyme system 39. In one study 40, 24 cancer patients were treated with intravenous irinotecan (600 mg, n = 12) or docetaxel (180 mg, n = 12), followed 3 weeks later by the same drugs concomitant with medicinal cannabis taken in the form of an herbal tea for 15 consecutive days, starting 12 days before the second treatment. The administration of cannabis did not significantly influence exposure to and clearance of irinotecan or docetaxel, although the herbal tea route of administration may not reproduce the effects of inhalation or oral ingestion of fat-soluble cannabinoids.
Can cannabis prevent or cause cancer?
There is no reliable evidence that cannabis can prevent cancer. There has been some research suggesting that endocannabinoids can suppress tumor growth, and in experiments where mice were given very high doses of purified THC, they seemed to have a lower risk of developing cancer. But this is not enough solid scientific evidence to suggest that cannabinoids or cannabis can cut people’s cancer risk. Right now, there simply isn’t enough evidence to prove that cannabinoids – whether natural or synthetic – can effectively treat cancer in patients, although research is ongoing. And there’s certainly no evidence that ‘street’ cannabis can treat cancer.
The evidence is a lot less clear when it comes to whether cannabis can cause cancer. This is because most people who use cannabis smoke it mixed with tobacco, a substance that scientists know causes cancer.
Cancer risk
A number of studies have yielded conflicting evidence regarding the risks of various cancers associated with cannabis smoking. A pooled analysis of three case-cohort studies of men in northwestern Africa (430 cases and 778 controls) showed a significantly increased risk of lung cancer among tobacco smokers who also inhaled cannabis 41.
A large, retrospective cohort study of 64,855 men aged 15 to 49 years from the United States found that cannabis use was not associated with tobacco-related cancers and a number of other common malignancies 42. However, the study did find that, among nonsmokers of tobacco, ever having used cannabis was associated with an increased risk of prostate cancer 42.
A population-based case-control study of 611 lung cancer patients revealed that chronic low cannabis exposure was not associated with an increased risk of lung cancer or other upper aerodigestive tract cancers and found no positive associations with any cancer type (oral, pharyngeal, laryngeal, lung, or esophagus) when adjusting for several confounders, including cigarette smoking 43.
A systematic review assessing 19 studies that evaluated premalignant or malignant lung lesions in persons 18 years or older who inhaled cannabis concluded that observational studies failed to demonstrate statistically significant associations between Cannabis inhalation and lung cancer after adjusting for tobacco use 44. In the review of the published meta-analyses, the National Academies of Sciences, Engineering, and Medicine 45 report concluded that there was moderate evidence of no statistical association between cannabis smoking and the incidence of lung cancer.
Epidemiologic studies examining one association of cannabis use with head and neck squamous cell carcinomas have also been inconsistent in their findings. A pooled analysis of nine case-control studies from the U.S./Latin American International Head and Neck Cancer Epidemiology Consortium included information from 1,921 oropharyngeal cases, 356 tongue cases, and 7,639 controls 46. Compared with those who never smoked Cannabis, Cannabis smokers had an elevated risk of oropharyngeal cancers and a reduced risk of tongue cancer. These study results both reflect the inconsistent effects of cannabinoids on cancer incidence noted in previous studies and suggest that more work needs to be done to understand the potential role of human papillomavirus infection. A systematic review and meta-analysis of nine case-control studies involving 13,931 participants also concluded that there was insufficient evidence to support or refute a positive or negative association between Cannabis smoking and the incidence of head and neck cancers 47.
With a hypothesis that chronic marijuana use produces adverse effects on the human endocrine and reproductive systems, the association between cannabis use and incidence of testicular germ cell tumors has been examined 48. Three population-based case-control studies reported an association between Cannabis use and elevated risk of testicular germ cell tumors, especially nonseminoma or mixed-histology tumors 48. However, the sample sizes in these studies were inadequate to address cannabis dose by addressing associations with respect to recency, frequency, and duration of use. In a study of 49,343 Swedish men aged 19 to 21 years enrolled in the military between 1969 and 1970, participants were asked once at the time of conscription about their use of Cannabis and were followed up for 42 years 49. This study found no evidence of a significant relation between “ever” cannabis use and the development of testicular cancer, but did find that “heavy” cannabis use (more than 50 times in a lifetime) was associated with a 2.5-fold increased risk. Limitations of the study were that it relied on indirect assessment of cannabis use; and no information was collected on Cannabis use after the conscription-assessment period or on whether the testicular cancers were seminoma or nonseminoma subtypes. These reports established the need for larger, well-powered, prospective studies, especially studies evaluating the role of endocannabinoid signaling and cannabinoid receptors in testicular germ cell tumors.
An analysis of 84,170 participants in the California Men’s Health Study was performed to investigate the association between cannabis use and the incidence of bladder cancer. During 16 years of follow-up, 89 cannabis users (0.3%) developed bladder cancer compared with 190 (0.4%) of the men who did not report cannabis use 50. After adjusting for age, race, ethnicity, and body mass index, Cannabis use was associated with a 45% reduction in bladder cancer incidence (hazard ratio, 0.55) 50.
A comprehensive Health Canada monograph on marijuana concluded that while there are many cellular and molecular studies that provide strong evidence that inhaled marijuana is carcinogenic, the epidemiologic evidence of a link between marijuana use and cancer is still inconclusive 37.
Can cannabis relieve cancer symptoms like pain or sickness?
There’s good evidence that cannabinoids may be beneficial in managing cancer pain and side effects from treatment. As far back as the 1980s, cannabinoid-based drugs including dronabinol (synthetic THC) and nabilone were used to help reduce nausea and vomiting caused by chemotherapy. But there are now safer and more effective alternatives and cannabinoids tend to only be used where other approaches fail.
In some parts of the world, medical marijuana has been legalized for relieving pain and symptoms (palliative use), including cancer pain. But one of the problems with using herbal cannabis is managing the dose. Smoking cannabis or taking it in the form of tea often provides an inconsistent dose, which may make it difficult for patients to monitor their intake. So, researchers are turning to alternative dosing methods, such as mouth sprays, which deliver a reliable and regulated dose.
Large-scale clinical trials in the UK have been testing whether a mouth spray formulation of Sativex (nabiximols) can help to control severe cancer pain that doesn’t respond to other drugs. Results from these didn’t find any difference in self-reported pain scores between the treatment and the placebo.
Cannabinoids may also have potential in combating the loss of appetite and wasting (cachexia) experienced by some people with cancer, although so far clinical evidence is lacking. One clinical trial comparing appetite in groups of cancer patients given cannabis extract, THC and a placebo didn’t find a difference between the treatments, while another didn’t show any benefit and was closed early.
Cannabis use among cancer patients
A cross-sectional survey of cancer patients seen at the Seattle Cancer Care Alliance was conducted over a 6-week period between 2015 and 2016 51. In Washington State, cannabis was legalized for medicinal use in 1998 and for recreational use in 2012. Of the 2,737 possible participants, 936 (34%) completed the anonymous questionnaire. Twenty-four percent of patients considered themselves active cannabis users. Similar numbers of patients inhaled (70%) or used edibles (70%), with dual use (40%) being common. Non–mutually exclusive reasons for cannabis use were physical symptoms (75%), neuropsychiatric symptoms (63%), recreational use/enjoyment (35%), and treatment of cancer (26%). The physical symptoms most commonly cited were pain, nausea, and loss of appetite. The majority of patients (74%) stated that they would prefer to obtain information about cannabis from their cancer team, but less than 15% reported receiving information from their cancer physician or nurse.
Data from 2,970 Israeli cancer patients who used government-issued Cannabis were collected over a 6-month period to assess for improvement in baseline symptoms 52. The most improved symptoms from baseline include the following:
- Nausea and vomiting (91.0%).
- Sleep disorders (87.5%).
- Restlessness (87.5%).
- Anxiety and depression (84.2%).
- Pruritus (82.1%).
- Headaches (81.4%).
Before treatment initiation, 52.9% of patients reported pain scores in the 8 to 10 range, while only 4.6% of patients reported this intensity at the 6-month assessment time point. It is difficult to assess from the observational data if the improvements were caused by the cannabis or the cancer treatment 52. Similarly, a study of a subset of cancer patients in the Minnesota medical Cannabis program explored changes in the severity of eight symptoms (i.e., anxiety, appetite loss, depression, disturbed sleep, fatigue, nausea, pain, and vomiting) experienced by these patients 53. Significant symptomatic improvements were noted (38.4%–56.2%) in patients with each symptom. Because of the observational and uncontrolled nature of this study, the findings are not generalizable, but as the authors suggested, may be useful in designing more rigorous research studies in the future.
A retrospective study from Israel of 50 pediatric oncology patients 54 who were prescribed medicinal Cannabis over an 8-year period reported that the most common indications include the following:
- Nausea and vomiting.
- Depressed mood.
- Sleep disturbances.
- Poor appetite and weight loss.
- Pain.
Most of the patients (N = 30) received cannabis in the form of oral oil drops, with some of the older children inhaling vaporized Cannabis or combining inhalation with oral oils. Structured interviews with the parents, and their child when appropriate, revealed that 40 participants (80%) reported a high level of general satisfaction with the use of Cannabis with infrequent short-term side effects 54.
Is cannabis addictive?
Cannabis or marijuana use can lead to the development of a substance use disorder, a medical illness in which the person is unable to stop using even though it’s causing health and social problems in their life. Severe substance use disorders are also known as addiction. Research suggests that between 9 and 30 percent of those who use marijuana may develop some degree of marijuana use disorder 55. People who begin using marijuana before age 18 are four to seven times more likely than adults to develop a marijuana use disorder 56.
Many people who use marijuana long term and are trying to quit report mild withdrawal symptoms that make quitting difficult. These include:
- grouchiness
- sleeplessness
- decreased appetite
- anxiety
- cravings.
No medications are currently available to treat marijuana use disorder, but behavioral support has been shown to be effective. Examples include therapy and motivational incentives (providing rewards to patients who remain drug-free). Continuing research may lead to new medications that help ease withdrawal symptoms, block the effects of marijuana, and prevent relapse.
Is it legal for dietary supplements or foods to contain THC or CBD?
The FDA has determined that products containing THC or CBD cannot be sold legally as dietary supplements. Foods to which THC or CBD has been added cannot be sold legally in interstate commerce. Whether they can be sold legally within a state depends on that state’s laws and regulations.
Is cannabis oil safe?
The risks of using cannabis products containing THC (delta-9-tetrahydrocannabinol) are not currently clear. That’s why clinical trials are needed before they can be used.
The main risks of THC cannabis products are:
- Psychosis – there is evidence that regular cannabis use increases your risk of developing a psychotic illness such as schizophrenia. A psychotic illness is one where you have hallucinations (seeing things that are not really there) and delusions (believing things that are not really true). Cannabis oil also increases the risk of a relapse in people who already have schizophrenia, and it can make psychotic symptoms worse.
- Dependency on the medicine – although scientists believe this risk is probably small when its use is controlled and monitored by a specialist doctor. Although cannabinoids are considered by some to be addictive drugs, their addictive potential is considerably lower than that of other prescribed agents or substances of abuse 57. The brain develops a tolerance to cannabinoids. Withdrawal symptoms such as irritability, insomnia with sleep electroencephalogram disturbance, restlessness, hot flashes, and, rarely, nausea and cramping have been observed. However, these symptoms appear to be mild compared with withdrawal symptoms associated with opiates or benzodiazepines, and the symptoms usually dissipate after a few days. Unlike other commonly used drugs, cannabinoids are stored in adipose tissue and excreted at a low rate (half-life 1–3 days), so even abrupt cessation of cannabinoid intake is not associated with rapid declines in plasma concentrations that would precipitate severe or abrupt withdrawal symptoms or drug cravings.
Because cannabinoid receptors, unlike opioid receptors, are not located in the brainstem areas controlling respiration, lethal overdoses from cannabis and cannabinoids do not occur 57. However, cannabinoid receptors are present in other tissues throughout the body, not just in the central nervous system, and adverse effects include the following:
- Tachycardia.
- Hypotension.
- Conjunctival injection.
- Bronchodilation.
- Muscle relaxation.
- Decreased gastrointestinal motility.
Cannabidiol (CBD) is an inhibitor of cytochrome P450 isoforms in vitro. Because many anticancer therapies are metabolized by these enzymes, highly concentrated CBD oils used concurrently could potentially increase the toxicity or decrease the effectiveness of these therapies 6.
Since cannabis smoke contains many of the same components as tobacco smoke, there are valid concerns about the adverse pulmonary effects of inhaled cannabis. A longitudinal study in a noncancer population evaluated repeated measurements of pulmonary function over 20 years in 5,115 men and women whose smoking histories were known 58. While tobacco exposure was associated with decreased pulmonary function, the investigators concluded that occasional and low-cumulative cannabis use was not associated with adverse effects on pulmonary function (forced expiratory volume in the first second of expiration [FEV1] and forced vital capacity [FVC]).
Several concerns have been raised about the safety of cannabis and cannabinoids:
- The use of cannabis has been linked to an increased risk of motor vehicle crashes.
- Smoking cannabis during pregnancy has been linked to lower birth weight.
- Some people who use cannabis develop cannabis use disorder, which has symptoms such as craving, withdrawal, lack of control, and negative effects on personal and professional responsibilities.
- Adolescents using cannabis are four to seven times more likely than adults to develop cannabis use disorder.
- Cannabis use is associated with an increased risk of injury among older adults.
- The use of cannabis, especially frequent use, has been linked to a higher risk of developing schizophrenia or other psychoses (severe mental illnesses) in people who are predisposed to these illnesses.
- Marijuana may cause orthostatic hypotension (head rush or dizziness on standing up), possibly raising danger from fainting and falls.
- The FDA has warned the public not to use vaping products that contain THC. Products of this type have been implicated in many of the reported cases of serious lung injuries linked to vaping.
- There have been many reports of unintentional consumption of cannabis or its products by children, leading to illnesses severe enough to require emergency room treatment or admission to a hospital. Among a group of people who became ill after accidental exposure to candies containing THC, the children generally had more severe symptoms than the adults and needed to stay in the hospital longer.
- Some long-term users of high doses of cannabis have developed a condition involving recurrent severe vomiting.
- There have been reports of contamination of cannabis/cannabinoid products with microorganisms, pesticides, or other substances.
- Some cannabis/cannabinoid products contain amounts of cannabinoids that differ substantially from what’s stated on their labels.
Generally, the more THC the product contains, the greater these risks are.
Cannabis bought illegally off the street, where the quality, ingredients and strength are not known, is the most dangerous form to use.
Cannabis and pregnancy
Cannabis use may affect fertility. Regular or heavy cannabis use has been linked to changes in the female menstrual cycle and lower sperm count, or lower sperm quality in men.
Using cannabis while pregnant may harm the unborn baby. Cannabis smoke contains many of the same harmful chemicals found in cigarette smoke.
Regularly smoking cannabis with tobacco increases the risk of a baby being born small or premature 59 and increased risk of both brain and behavioral problems in babies.
Cannabis has not been linked to birth defects, but research suggests that using cannabis regularly during pregnancy could affect a baby’s brain development as they get older.
If a pregnant woman uses marijuana, the drug may affect certain developing parts of the fetus’s brain. Children exposed to marijuana in the womb have an increased risk of problems with attention 60, memory, and problem-solving compared to unexposed children 61. Some research also suggests that moderate amounts of THC are excreted into the breast milk of nursing mothers 62. With regular use, THC can reach amounts in breast milk that could affect the baby’s developing brain. Other recent research suggests an increased risk of preterm births 63. More research is needed.
Cannabis oil dosage
Clinical studies of cannabis and cannabis-based products for therapeutic purposes are limited to studies carried out with dried cannabis that was smoked or vaporized and with synthetic or natural cannabis-based products that have received market authorization (i.e. dronabinol, nabilone, and nabiximols). With the possible exception of trials conducted with Epidiolex® (CBD-enriched oil) for epilepsy 64 and one open-label pilot clinical trial of oral THC oil for symptoms associated with post-traumatic stress disorder (PTSD) 65 there are no other clinical studies of fresh cannabis or cannabis oils for therapeutic purposes. As such, providing precise dosing guidelines for such products is not possible.
Data from an open-label longitudinal study of cannabis oil for patients with treatment-resistant chronic non-cancer pain reported that patients titrated their cannabis oil dose starting with one drop of cannabis oil per day, increasing in increments of one drop of oil per dose, three times per day, until satisfactory analgesia was achieved or until side effects appeared 66. THC concentrations ranged from 11 – 19% and 0.5 – 5.5% CBD in cannabis oil in this study.
An open-label, pilot study of add-on oral THC (25 mg/ml in olive oil) for the treatment of symptoms associated with PTSD suggested dosing begin by placing 2.5 mg THC twice daily beneath the tongue (i.e. 0.1 mL of the oil solution) 1 hour after waking up and 2 hours before going to bed 65. Maximum daily dose was 5 mg twice daily. (i.e. 0.2 mL twice daily), or a total 10 mg daily dose (i.e. 0.4 mL).
Prescription cannabinoids
Information obtained from the monograph for Marinol® (dronabinol) indicates that a daily oral dose as low as 2.5 mg THC is associated with a therapeutic effect (e.g. treatment of AIDS-related anorexia/cachexia). Naturally, dosing will vary according to the underlying disorder and the many other variables mentioned above. Dosing ranges for Marinol® (dronabinol) vary from 2.5 mg to 40 mg THC/day (maximal tolerated daily human dose = 210 mg THC/day) 67. Average daily dose of dronabinol is 20 mg and maximal recommended daily dose is 40 mg 67. Doses less than 1 mg of THC per dosing session may further avoid incidence and risks of adverse effects. Dosing ranges for Cesamet® (nabilone) vary from 0.2 mg to 6 mg/day 68. Dosing ranges for Sativex® (nabiximols) vary from one spray (2.7 mg THC and 2.5 mg CBD) to 16 sprays (43.2 mg THC and 40 mg CBD) per day 69. Information from clinical studies with Epidiolex®, an oil-based extract of cannabis containing 98% CBD, suggests a daily dosing range between 5 and 20 mg/kg/day 70.
Marinol
Dronabinol is the generic name for the oral form of synthetic THC and is marketed in the U.S. as Marinol®. It was available for sale in capsules containing 2.5, 5, or 10 mg of the drug dissolved in sesame oil. It is indicated for the treatment of severe chemotherapy-induced nausea and vomiting in cancer patients, and for AIDS-related anorexia associated with weight loss 67.
Marinol® product monograph suggests a mean of 5 mg THC/day (range: 2.5 – 20 mg THC/day) for AIDS-related anorexia associated with weight loss 67. A 2.5 mg dose may be administered before lunch, followed by a second 2.5 mg dose before supper. On the other hand, to reduce or prevent CINV, a dosage of 5 mg three times daily or four times daily is suggested. In either case, the dose should be carefully titrated to avoid the manifestation of adverse effects. Please consult the Marinol® drug product monograph for more detailed instructions 67.
Cesamet
Nabilone is the generic name for an orally administered synthetic structural analogue of THC, which is marketed as Cesamet® but also now available in generic forms (e.g. RAN-nabilone, PMS-nabilone, TEVA-nabilone, CO-nabilone, ACT-nabilone). It is available as capsules (0.25, 0.5, 1 mg) and is indicated for severe chemotherapy-induced nausea and vomiting in cancer patients 68.
The Cesamet® (nabilone) product monograph suggests administration of 1 to 2 mg of the drug, twice a day, with the first dose given the night before administration of the chemotherapeutic medication 68. A 2 mg dose of nabilone gave a mean plasma concentration of 10 ng/mL nabilone, 1 to 2 hour after administration. The second dose is usually administered 1 to 3 hours before chemotherapy. If required, the administration of nabilone can be continued up to 24 hours after the chemotherapeutic agent is given. The maximum recommended daily dose is 6 mg in divided doses. Dose adjustment (titration) may be required in order to attain the desired response, or to improve tolerability. More recent clinical trials report starting doses of nabilone of 0.5 mg at night for pain or insomnia in fibromyalgia, and for insomnia in PTSD 71. Please consult the Cesamet® drug product monograph for more detailed instructions 68.
Epidiolex
Epidiolex is the brand name for a whole-plant cannabis extract of a high CBD strain of cannabis sativa and is an oral oil-based solution product containing > 98% CBD at a concentration of 100 mg/ml. Epidiolex has received Orphan Drug Designation in the U.S. for the treatment of Lennox-Gastaut Syndrome, Dravet Syndrome and Tuberous Sclerosis Complex.
Data from an open-label clinical study of Epidiolex® for treatment-resistant childhood-onset epilepsy suggest that dosing with Epidiolex® (98 – 99% pure CBD oil) begin at a starting dose of 2 to 5 mg/kg per day divided in twice-daily dosing in addition to baseline antiepileptic drug regimen, then up-titrated by 2 to 5 mg/kg once a week until intolerance or a maximum dose of 25 mg/kg per day is reachedReference 262. In some specific situations, the study authors mention that an increase to a maximum dose of 50 mg/kg per day could be considered. In patients with drug- resistant seizures in the Dravet syndromeReference 576 or treatment-resistant Lennox-Gastaut syndromeReference 577, a dose of 20 mg/kg per day is efficacious and generally well tolerated.
Sativex
Nabiximols is the generic name for a whole-plant extract of two different, but standardized, strains of cannabis sativa giving an oro-mucosal spray product containing approximately equivalent amounts of THC (27 mg/mL) and CBD (25 mg/mL), and other cannabinoids, terpenoids, and flavonoids per 100 μl of dispensed spray. Nabiximols is marketed as Sativex and has received a notice of compliance for use as an adjunctive treatment for the symptomatic relief of spasticity in adult patients with multiple sclerosis (MS) who have not responded adequately to other therapy, and who demonstrate meaningful improvement during an initial trial of therapy. It is also marketed (with conditions) as an adjunctive treatment for the symptomatic relief of neuropathic pain in adults with MS and (with conditions) as an adjunctive analgesic in adult patients with advanced cancer who experience moderate to severe pain during the highest tolerated dose of strong opioid therapy for persistent background pain 69.
Dosing with nabiximols (Sativex®) is described in the product monograph along with a titration method for proper treatment initiation 69. Briefly, dosing indications in the drug product monograph suggest that on the first day of treatment patients take one spray during the morning (anytime between waking and noon), and another in the afternoon/evening (anytime between 4 p.m. and bedtime). On subsequent days, the number of sprays can be increased by one spray per day, as needed and tolerated. A fifteen-minute time gap should be allowed between sprays. During the initial titration, sprays should be evenly spread out over the day. If at any time unacceptable adverse reactions such as dizziness or other central nervous system-type reactions develop, dosing should be suspended or reduced or the dosing schedule changed to increase the time intervals between doses. According to the drug product monograph, the average dose of nabiximols is five sprays per day (i.e. 13 mg THC and 12.5 mg CBD) for patients with MS, whereas those patients with cancer pain tend to use an average of eight sprays per day (i.e. 21.6 mg THC and 20 mg CBD). The majority of patients appear to require 12 sprays or less; dosage should be adjusted as needed and tolerated. Administration of four sprays to healthy volunteers (total 10.8 mg THC and 10 mg CBD) was associated with a mean maximum plasma concentration varying between 4.90 and 6.14 ng/mL THC and 2.50 to 3.02 ng/mL CBD depending whether the drug was administered under the tongue or inside the cheek.
Cannabis oil side effects
After taking medical cannabis, it’s possible to develop any of the following side effects:
- decreased appetite
- diarrhea
- increased heart rate
- feeling sick
- greater weakness
- a behavioral or mood change
- dizziness
- feeling very tired
- feeling high
- hallucinations
- suicidal thoughts
Intense nausea and vomiting. Regular, long-term marijuana use can lead to some people to develop Cannabinoid Hyperemesis Syndrome 72. This causes users to experience regular cycles of severe nausea, vomiting, and dehydration, sometimes requiring emergency medical attention.
If you experience any side effects from medical cannabis, report these to your medical team.
References- Raber JC, Elzinga S, Kaplan C. Understanding dabs: contamination concerns of cannabis concentrates and cannabinoid transfer during the act of dabbing. J Toxicol Sci. 2015 Dec;40(6):797-803. doi: 10.2131/jts.40.797 https://www.jstage.jst.go.jp/article/jts/40/6/40_797/_pdf/-char/en
- Sutton IR, Daeninck P. Cannabinoids in the management of intractable chemotherapy-induced nausea and vomiting and cancer-related pain. J Support Oncol. 2006 Nov-Dec;4(10):531-5.
- Duran M, Pérez E, Abanades S, Vidal X, Saura C, Majem M, Arriola E, Rabanal M, Pastor A, Farré M, Rams N, Laporte JR, Capellà D. Preliminary efficacy and safety of an oromucosal standardized cannabis extract in chemotherapy-induced nausea and vomiting. Br J Clin Pharmacol. 2010 Nov;70(5):656-63. doi: 10.1111/j.1365-2125.2010.03743.x
- Hesketh PJ, Kris MG, Basch E, Bohlke K, Barbour SY, Clark-Snow RA, Danso MA, Dennis K, Dupuis LL, Dusetzina SB, Eng C, Feyer PC, Jordan K, Noonan K, Sparacio D, Somerfield MR, Lyman GH. Antiemetics: American Society of Clinical Oncology Clinical Practice Guideline Update. J Clin Oncol. 2017 Oct 1;35(28):3240-3261. doi: 10.1200/JCO.2017.74.4789
- FDA warns companies marketing unproven products, derived from marijuana, that claim to treat or cure cancer. https://www.fda.gov/news-events/press-announcements/fda-warns-companies-marketing-unproven-products-derived-marijuana-claim-treat-or-cure-cancer
- Jiang R, Yamaori S, Okamoto Y, Yamamoto I, Watanabe K. Cannabidiol is a potent inhibitor of the catalytic activity of cytochrome P450 2C19. Drug Metab Pharmacokinet. 2013;28(4):332-8. doi: 10.2133/dmpk.dmpk-12-rg-129
- The Health Effects of Cannabis and Cannabinoids. The Current State of Evidence and Recommendations for Research (2017). https://www.nap.edu/catalog/24625/the-health-effects-of-cannabis-and-cannabinoids-the-current-state
- Foltin RW, Fischman MW, Byrne MF. Effects of smoked marijuana on food intake and body weight of humans living in a residential laboratory. Appetite. 1988 Aug;11(1):1-14. doi: 10.1016/s0195-6663(88)80017-5
- Jatoi A, Windschitl HE, Loprinzi CL, Sloan JA, Dakhil SR, Mailliard JA, Pundaleeka S, Kardinal CG, Fitch TR, Krook JE, Novotny PJ, Christensen B. Dronabinol versus megestrol acetate versus combination therapy for cancer-associated anorexia: a North Central Cancer Treatment Group study. J Clin Oncol. 2002 Jan 15;20(2):567-73. doi: 10.1200/JCO.2002.20.2.567
- Regelson W, Butler JR, Schulz J, et al.: Delta-9-tetrahydrocannabinol as an effective antidepressant and appetite-stimulating agent in advanced cancer patients. In: Braude MC, Szara S: The Pharmacology of Marihuana. New York, NY: Raven Press, 1976, pp 763-76.
- Brisbois TD, de Kock IH, Watanabe SM, et al.: Delta-9-tetrahydrocannabinol may palliate altered chemosensory perception in cancer patients: results of a randomized, double-blind, placebo-controlled pilot trial. Ann Oncol 22 (9): 2086-93, 2011.
- Turcott JG, Del Rocío Guillen Núñez M, Flores-Estrada D, et al.: The effect of nabilone on appetite, nutritional status, and quality of life in lung cancer patients: a randomized, double-blind clinical trial. Support Care Cancer 26 (9): 3029-3038, 2018.
- Radwan MM, ElSohly MA, El-Alfy AT, Ahmed SA, Slade D, Husni AS, Manly SP, Wilson L, Seale S, Cutler SJ, et al. Isolation and pharmacological evaluation of minor cannabinoids from high-potency cannabis sativa. J Nat Prod 2015 Jun 26;78(6):1271-6.
- Yamaori S, Kushihara M, Yamamoto I, Watanabe K. Characterization of major phytocannabinoids, cannabidiol and cannabinol, as isoform-selective and potent inhibitors of human CYP1 enzymes. Biochem Pharmacol 2010 06/01;79(1873-2968; 0006-2952; 11):1691-8.
- Mehmedic Z, Chandra S, Slade D, Denham H, Foster S, Patel AS, Ross SA, Khan IA, Elsohly MA. Potency trends of delta(9)-THC and other cannabinoids in confiscated cannabis preparations from 1993 to 2008*. J Forensic Sci 2010 05/04;55(1556-4029; 0022-1198; 5):1209-17.
- Elsohly MA, Slade D. Chemical constituents of marijuana: The complex mixture of natural cannabinoids. Life Sci 2005 12/22;78(0024-3205; 0024-3205; 5):539-48.
- Huestis MA. Human cannabinoid pharmacokinetics. Chem.Biodivers. 2007 08;4(1612-1880; 1612-1872; 8):1770-804.
- Tibiriça E. (2010). The multiple functions of the endocannabinoid system: a focus on the regulation of food intake. Diabetology & metabolic syndrome, 2, 5. https://doi.org/10.1186/1758-5996-2-5
- Rodriguez de Fonseca F, del Arco I, Bermudez-Silva FJ, Bilbao A, Cippitelli A, Navarro M. The endocannabinoid system: Physiology and pharmacology. Alcohol Alcohol 2005 01;40(0735-0414; 0735-0414; 1):2-14.
- Serrano A, Parsons LH. Endocannabinoid influence in drug reinforcement, dependence and addiction-related behaviors. Pharmacol Ther 2011 12;132(1879-016; 0163-7258; 3):215-41.
- Maccarrone M, Gasperi V, Catani MV, Diep TA, Dainese E, Hansen HS, Avigliano L. The endocannabinoid system and its relevance for nutrition. Annu Rev Nutr 2010 08/21;30(1545-4312; 0199-9885):423-40.
- Aggarwal SK. Cannabinergic pain medicine: A concise clinical primer and survey of randomized-controlled trial results. Clin J Pain 2012 02/23;29(1536-5409; 0749-8047; 2):162-71.
- Hillard CJ. The endocannabinoid signaling system in the CNS: A primer. Int Rev Neurobiol 2015;125:1-47.
- O’Sullivan SE, Kendall DA. Cannabinoid activation of peroxisome proliferator-activated receptors: Potential for modulation of inflammatory disease. Immunobiology 2010 08;215(1878-3279; 0171-2985; 8):611-6.
- Hansen HS. Palmitoylethanolamide and other anandamide congeners. proposed role in the diseased brain. Exp Neurol 2010 07;224(1090-2430; 0014-4886; 1):48-55.
- Di Marzo V, Piscitelli F, Mechoulam R. Cannabinoids and endocannabinoids in metabolic disorders with focus on diabetes. Handb Exp Pharmacol 2011(0171-2004; 0171-2004; 203):75-104.
- Kraft B. Is there any clinically relevant cannabinoid-induced analgesia? Pharmacology 2012;89(1423-0313; 0031-7012; 5-6):237-46.
- Teixeira D, Pestana D, Faria A, Calhau C, Azevedo I, Monteiro R. Modulation of adipocyte biology by delta(9)-tetrahydrocannabinol. Obesity.(Silver.Spring) 2010 11;18(1930-739; 1930-7381; 11):2077-85.
- Mackie K. Signaling via CNS cannabinoid receptors. Mol Cell Endocrinol 2008 04/16;286(0303-7207; 0303-7207; 1-2):S60-5.
- De Petrocellis L, Di M,V. Non-CB1, non-CB2 receptors for endocannabinoids, plant cannabinoids, and synthetic cannabimimetics: Focus on G-protein-coupled receptors and transient receptor potential channels. J.Neuroimmune.Pharmacol. 2010 03;5(1557-1904; 1557-1890; 1):103-21.
- Miller LK, Devi LA. The highs and lows of cannabinoid receptor expression in disease: Mechanisms and their therapeutic implications. Pharmacol Rev 2011 09;63(1521-0081; 0031-6997; 3):461-70.
- Battista N, Di TM, Bari M, Maccarrone M. The endocannabinoid system: An overview. Front Behav Neurosci 2012;6(1662-5153; 1662-5153):9.
- Eisenberg E, Ogintz M, Almog S. The pharmacokinetics, efficacy, safety, and ease of use of a novel portable metered-dose cannabis inhaler in patients with chronic neuropathic pain: A phase 1a study. J Pain Palliat Care Pharmacother 2014 Sep;28(3):216-25.
- Bisogno T. Endogenous cannabinoids: Structure and metabolism. J Neuroendocrinol 2008 05;20 Suppl 1(1365-2826; 0953-8194):1-9.
- Horvath B, Mukhopadhyay P, Hasko G, Pacher P. The endocannabinoid system and plant-derived cannabinoids in diabetes and diabetic complications. Am J Pathol 2012 02;180(1525-2191; 0002-9440; 2):432-42.
- Morgan CJ, Page E, Schaefer C, Chatten K, Manocha A, Gulati S, Curran HV, Brandner B, Leweke FM. Cerebrospinal fluid anandamide levels, cannabis use and psychotic-like symptoms. Br J Psychiatry 2013 May;202(5):381-2.
- Information for Health Care Professionals: Cannabis (marihuana, marijuana) and the cannabinoids. https://www.canada.ca/en/health-canada/services/drugs-medication/cannabis/information-medical-practitioners/information-health-care-professionals-cannabis-cannabinoids.html
- Adams IB, Martin BR. Cannabis: pharmacology and toxicology in animals and humans. Addiction. 1996 Nov;91(11):1585-614.
- Yamamoto I, Watanabe K, Narimatsu S, Yoshimura H. Recent advances in the metabolism of cannabinoids. Int J Biochem Cell Biol. 1995 Aug;27(8):741-6. doi: 10.1016/1357-2725(95)00043-o. Erratum in: Int J Biochem Cell Biol 1995 Dec;27(12):1365.
- Engels FK, de Jong FA, Sparreboom A, Mathot RA, Loos WJ, Kitzen JJ, de Bruijn P, Verweij J, Mathijssen RH. Medicinal cannabis does not influence the clinical pharmacokinetics of irinotecan and docetaxel. Oncologist. 2007 Mar;12(3):291-300. doi: 10.1634/theoncologist.12-3-291
- Berthiller J, Straif K, Boniol M, Voirin N, Benhaïm-Luzon V, Ayoub WB, Dari I, Laouamri S, Hamdi-Cherif M, Bartal M, Ayed FB, Sasco AJ. Cannabis smoking and risk of lung cancer in men: a pooled analysis of three studies in Maghreb. J Thorac Oncol. 2008 Dec;3(12):1398-403. doi: 10.1097/JTO.0b013e31818ddcde
- Sidney S, Quesenberry CP Jr, Friedman GD, Tekawa IS. Marijuana use and cancer incidence (California, United States). Cancer Causes Control. 1997 Sep;8(5):722-8. doi: 10.1023/a:1018427320658
- Hashibe M, Morgenstern H, Cui Y, Tashkin DP, Zhang ZF, Cozen W, Mack TM, Greenland S. Marijuana use and the risk of lung and upper aerodigestive tract cancers: results of a population-based case-control study. Cancer Epidemiol Biomarkers Prev. 2006 Oct;15(10):1829-34. doi: 10.1158/1055-9965.EPI-06-0330 https://cebp.aacrjournals.org/content/15/10/1829.long
- Mehra R, Moore BA, Crothers K, Tetrault J, Fiellin DA. The association between marijuana smoking and lung cancer: a systematic review. Arch Intern Med. 2006 Jul 10;166(13):1359-67. doi: 10.1001/archinte.166.13.1359
- National Academies of Sciences, Engineering, and Medicine: The Health Effects of Cannabis and Cannabinoids: The Current State of Evidence and Recommendations for Research. Washington, DC: The National Academies Press, 2017.
- Marks MA, Chaturvedi AK, Kelsey K, Straif K, Berthiller J, Schwartz SM, Smith E, Wyss A, Brennan P, Olshan AF, Wei Q, Sturgis EM, Zhang ZF, Morgenstern H, Muscat J, Lazarus P, McClean M, Chen C, Vaughan TL, Wunsch-Filho V, Curado MP, Koifman S, Matos E, Menezes A, Daudt AW, Fernandez L, Posner M, Boffetta P, Lee YC, Hashibe M, D’Souza G. Association of marijuana smoking with oropharyngeal and oral tongue cancers: pooled analysis from the INHANCE consortium. Cancer Epidemiol Biomarkers Prev. 2014 Jan;23(1):160-71. doi: 10.1158/1055-9965.EPI-13-0181
- de Carvalho MF, Dourado MR, Fernandes IB, Araújo CT, Mesquita AT, Ramos-Jorge ML. Head and neck cancer among marijuana users: a meta-analysis of matched case-control studies. Arch Oral Biol. 2015 Dec;60(12):1750-5. doi: 10.1016/j.archoralbio.2015.09.009
- Lacson JC, Carroll JD, Tuazon E, Castelao EJ, Bernstein L, Cortessis VK. Population-based case-control study of recreational drug use and testis cancer risk confirms an association between marijuana use and nonseminoma risk. Cancer. 2012 Nov 1;118(21):5374-83. doi: 10.1002/cncr.27554
- Callaghan RC, Allebeck P, Akre O, McGlynn KA, Sidorchuk A. Cannabis Use and Incidence of Testicular Cancer: A 42-Year Follow-up of Swedish Men between 1970 and 2011. Cancer Epidemiol Biomarkers Prev. 2017 Nov;26(11):1644-1652. doi: 10.1158/1055-9965.EPI-17-0428
- Thomas AA, Wallner LP, Quinn VP, Slezak J, Van Den Eeden SK, Chien GW, Jacobsen SJ. Association between cannabis use and the risk of bladder cancer: results from the California Men’s Health Study. Urology. 2015 Feb;85(2):388-92. doi: 10.1016/j.urology.2014.08.060
- Pergam SA, Woodfield MC, Lee CM, Cheng GS, Baker KK, Marquis SR, Fann JR. Cannabis use among patients at a comprehensive cancer center in a state with legalized medicinal and recreational use. Cancer. 2017 Nov 15;123(22):4488-4497. doi: 10.1002/cncr.30879
- Bar-Lev Schleider L, Mechoulam R, Lederman V, Hilou M, Lencovsky O, Betzalel O, Shbiro L, Novack V. Prospective analysis of safety and efficacy of medical cannabis in large unselected population of patients with cancer. Eur J Intern Med. 2018 Mar;49:37-43. doi: 10.1016/j.ejim.2018.01.023
- Anderson SP, Zylla DM, McGriff DM, Arneson TJ. Impact of Medical Cannabis on Patient-Reported Symptoms for Patients With Cancer Enrolled in Minnesota’s Medical Cannabis Program. J Oncol Pract. 2019 Apr;15(4):e338-e345. doi: 10.1200/JOP.18.00562
- Ofir R, Bar-Sela G, Weyl Ben-Arush M, Postovsky S. Medical marijuana use for pediatric oncology patients: single institution experience. Pediatr Hematol Oncol. 2019 Aug;36(5):255-266. doi: 10.1080/08880018.2019.1630537
- Hasin DS, Saha TD, Kerridge BT, et al. Prevalence of Marijuana Use Disorders in the United States Between 2001-2002 and 2012-2013. JAMA Psychiatry. 2015;72(12):1235-1242. doi:10.1001/jamapsychiatry.2015.1858
- Winters KC, Lee C-YS. Likelihood of developing an alcohol and cannabis use disorder during youth: association with recent use and age. Drug Alcohol Depend. 2008;92(1-3):239-247. doi:10.1016/j.drugalcdep.2007.08.005
- Guzmán M. Cannabinoids: potential anticancer agents. Nat Rev Cancer. 2003 Oct;3(10):745-55. doi: 10.1038/nrc1188
- Pletcher MJ, Vittinghoff E, Kalhan R, Richman J, Safford M, Sidney S, Lin F, Kertesz S. Association between marijuana exposure and pulmonary function over 20 years. JAMA. 2012 Jan 11;307(2):173-81. doi: 10.1001/jama.2011.1961
- National Academies of Sciences, Engineering, and Medicine. 2017. The Health Effects of Cannabis and Cannabinoids: The Current State of Evidence and Recommendations for Research. Washington, DC: The National Academies Press. https://doi.org/10.17226/24625
- Goldschmidt L, Day NL, Richardson GA. Effects of prenatal marijuana exposure on child behavior problems at age 10. Neurotoxicol Teratol. 2000;22(3):325-336.
- Richardson GA, Ryan C, Willford J, Day NL, Goldschmidt L. Prenatal alcohol and marijuana exposure: effects on neuropsychological outcomes at 10 years. Neurotoxicol Teratol. 2002;24(3):309-320.
- Perez-Reyes M, Wall ME. Presence of delta9-tetrahydrocannabinol in human milk. N Engl J Med. 1982;307(13):819-820. doi:10.1056/NEJM198209233071311.
- Corsi DJ, Walsh L, Weiss D, et al. Association Between Self-reported Prenatal Cannabis Use and Maternal, Perinatal, and Neonatal Outcomes. JAMA. Published online June 18, 2019322(2):145–152. doi:10.1001/jama.2019.8734
- Thiele EA, Marsh ED, French JA, Mazurkiewicz-Beldzinska M, Benbadis SR, Joshi C, Lyons PD, Taylor A, Roberts C, Sommerville K, et al. Cannabidiol in patients with seizures associated with lennox-gastaut syndrome (GWPCARE4): A randomised, double-blind, placebo-controlled phase 3 trial. Lancet 2018 Jan 25.
- Roitman P, Mechoulam R, Cooper-Kazaz R, Shalev A. Preliminary, open-label, pilot study of add-on oral Delta9-tetrahydrocannabinol in chronic post-traumatic stress disorder. Clin Drug Investig 2014 Aug;34(8):587-91.
- Haroutounian S, Ratz Y, Ginosar Y, Furmanov K, Saifi F, Meidan R, Davidson E. The effect of medicinal cannabis on pain and quality of life outcomes in chronic pain: A prospective open-label study. Clin J Pain 2016 Feb 17.
- https://www.accessdata.fda.gov/drugsatfda_docs/label/2005/018651s021lbl.pdf
- https://www.accessdata.fda.gov/drugsatfda_docs/label/2006/018677s011lbl.pdf
- https://pdf.hres.ca/dpd_pm/00016162.PDF
- dos Santos RG, Hallak JE, Leite JP, Zuardi AW, Crippa JA. Phytocannabinoids and epilepsy. J Clin Pharm Ther 2015 Apr;40(2):135-43.
- Ware MA, Fitzcharles MA, Joseph L, Shir Y. The effects of nabilone on sleep in fibromyalgia: Results of a randomized controlled trial. Anesth Analg 2010 02/01;110(1526-7598; 0003-2999; 2):604-10.
- Galli JA, Sawaya RA, Friedenberg FK. Cannabinoid Hyperemesis Syndrome. Curr Drug Abuse Rev. 2011;4(4):241-249.