What is choline
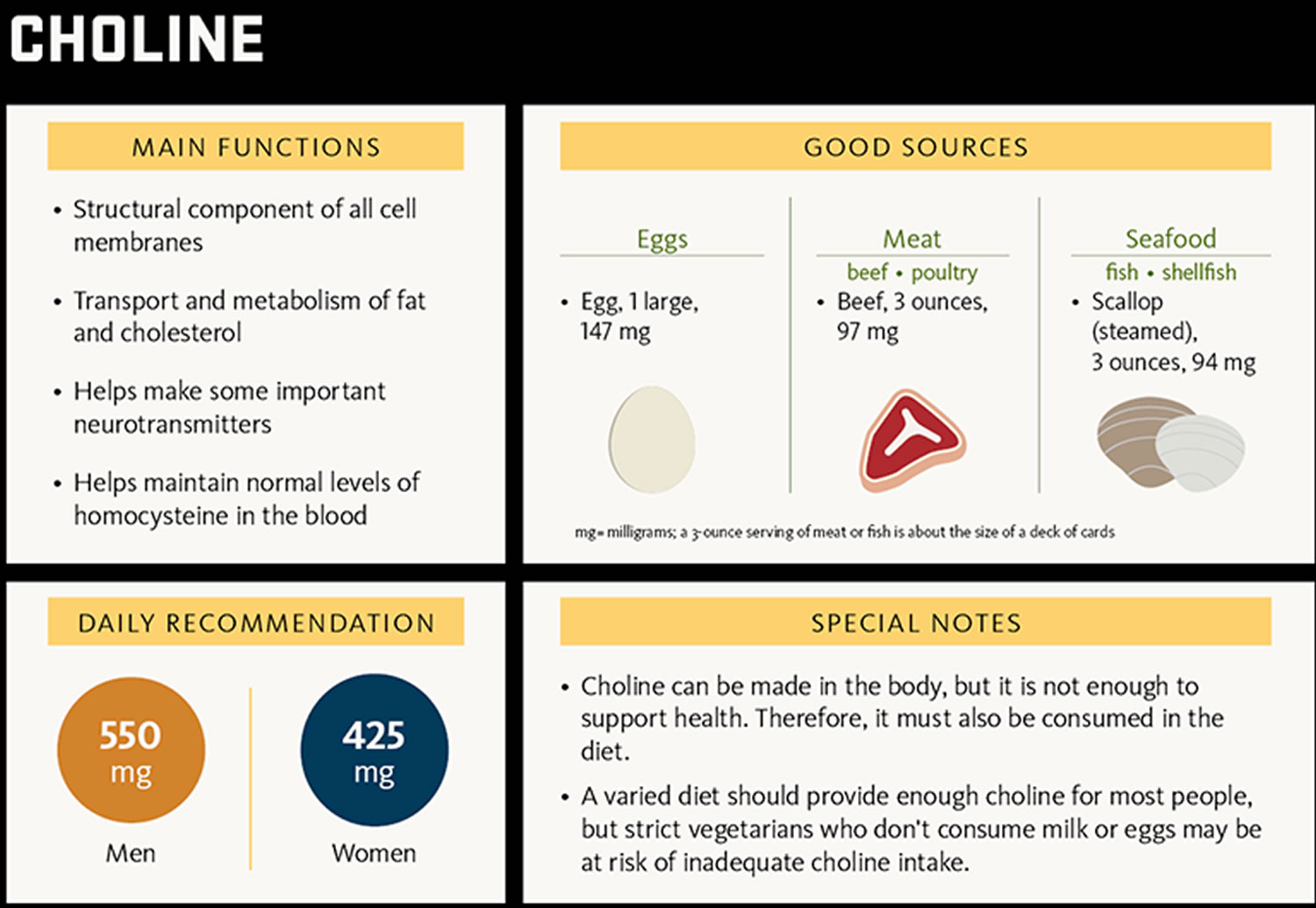
Choline (2-hydroxy-N,N,N-trimethylethanaminium) is an essential nutrient; that is, together with essential amino acids, fatty acids, vitamins and minerals, choline must be obtained from the diet to maintain health 1. Humans can synthesize small amounts of choline but not enough to support health. Therefore, choline is considered an essential nutrient and must be consumed in the diet. Choline is naturally present in some foods and available as a dietary supplement. Choline is a source of methyl groups needed for many steps in metabolism. The body needs choline to synthesize phosphatidylcholine and sphingomyelin, two major phospholipids vital for cell membranes 2. Therefore, all plant and animal cells need choline to preserve their structural integrity 3. In addition, choline is needed to produce acetylcholine, an important neurotransmitter for memory, mood, muscle control, and other brain and nervous system functions 3. Choline also plays important roles in modulating gene expression, cell membrane signaling, lipid transport and metabolism, and early brain development 3.
Humans can produce choline endogenously in the liver, mostly as phosphatidylcholine, but the amount that the body naturally synthesizes is not sufficient to meet human needs 4. As a result, humans must obtain some choline from the diet. Premenopausal women might need less choline from the diet than children or other adults because estrogen induces the gene that catalyzes the biosynthesis of choline 4. When a diet is deficient in folate, a B-vitamin that is also a methyl donor, the need for dietary choline rises because choline becomes the primary methyl donor 3.
The most common sources of choline in foods are the fat-soluble phospholipids phosphatidylcholine and sphingomyelin as well as the water-soluble compounds phosphocholine, glycerolphosphocholine, and free choline 3. When these choline-containing compounds are ingested, pancreatic and mucosal enzymes liberate free choline from about half of the fat-soluble forms and some water-soluble forms 5. Free choline, phosphocholine, and glycerophosphocholine are absorbed in the small intestine, enter the portal circulation, and are stored in the liver, where they are subsequently phosphorylated and distributed throughout the body to make cell membranes 3. The remaining fat-soluble phospholipids (phosphatidylcholine and sphingomyelin) are absorbed intact, incorporated into chylomicrons, and secreted into the lymphatic circulation, where they are distributed to tissues and other organs, including the brain and placenta 3.
Choline status is not routinely measured in healthy people. In healthy adults, the concentration of choline in plasma ranges from 7 to 20 mcmol/L 6. According to one study, the range is 7–9.3 mcmol/L in fasting adults 7. Plasma choline levels do not decline below 50% of normal, even in individuals who have not eaten for more than a week 8. This may be due to the hydrolysis of membrane phospholipids, a source of choline, to maintain plasma choline concentrations above this minimal level, or to endogenous synthesis 6.
Choline benefits
Choline might play a role: cardiovascular and peripheral artery disease, neurological disorders, and nonalcoholic fatty liver disease (NAFLD). Choline is involved in functions that overlap with those of folate and other B vitamins. Many studies do not assess the status of all B vitamins, which can confound results and obscure the true relationship between choline and the observed outcome.
Cardiovascular and peripheral artery disease
Some researchers have suggested that choline might protect cardiovascular health by reducing blood pressure, altering lipid profiles, and reducing levels of plasma homocysteine 8. Other research suggests that higher dietary choline might increase cardiovascular disease risk because some choline and other dietary ingredients, such as carnitine, are converted to trimethylamine (TMA) by intestinal bacteria. The trimethylamine (TMA) is then absorbed and converted by the liver into trimethylamine-N-oxide (TMAO), a substance that has been linked to a higher risk of cardiovascular disease 9.
Despite the hypothesis that choline might affect heart health, several large observational studies have found no significant associations between choline intakes and cardiovascular or peripheral artery disease risk. An analysis of 72,348 women in the Nurses’ Health Study and 44,504 men in the Health Professionals Follow-up Study showed no association between choline intake and risk of peripheral artery disease in men or women 10. Similarly, a prospective study in 14,430 middle-aged adults in the Atherosclerosis Risk in Communities Study found that over 14 years, risk of coronary heart disease was not significantly different in the highest choline intake quartile compared to the lowest quartile 11. Choline intakes also had no association with cardiovascular disease risk in a study of 16,165 women participating in the European Prospective Investigation into Cancer and Nutrition 12.
However, a more recent analysis of data on 80,978 women from the Nurses’ Health Study and 39,434 men from the Health Professionals Follow-Up Study found an increased risk of mortality in those consuming higher levels of choline 13. The authors suggest that the higher risk might be due to increased production of trimethylamine-N-oxide (TMAO), although they did not directly measure trimethylamine-N-oxide (TMAO). In this 2017 systematic review and meta-analysis of prospective studies 14, neither dietary choline nor betaine were associated with incident cardiovascular disease, but indicate the need for additional research into choline and cardiovascular disease mortality.
Additional research is needed to determine the relationship between choline intakes and cardiovascular and peripheral artery disease as well as the potential risks and benefits of choline supplementation to reduce the risk of these diseases.
Neurological disorders
People with Alzheimer’s disease have lower levels of the enzyme that converts choline into acetylcholine in the brain 15. In addition, because phosphatidylcholine can serve as a phospholipid precursor, it might help support the structural integrity of neurons and thus might promote cognitive function in elderly adults 16. Some experts have therefore theorized that consuming higher levels of phosphatidylcholine could reduce the progression of dementia in people with Alzheimer’s disease 15. However, little research conducted to date supports this hypothesis, as described below.
A few observational studies have shown a link between cognitive performance in adults and both higher choline intakes and plasma concentrations. In one observational study in 2,195 adults aged 70–74 years in Norway, participants with plasma free choline concentrations lower than 8.4 mcmol/L (20th percentile of concentrations in the study population) had poorer sensorimotor speed, perceptual speed, executive function, and global cognition than those with choline concentrations higher than 8.4 mcmol/L 17. A second study in 1,391 adults aged 36–83 years from the Framingham Offspring study who completed food frequency questionnaires from 1991 to 1995 and again from 1998 to 2001 found that those with higher choline intakes had better verbal memory and visual memory 18. Furthermore, higher choline intakes during the earlier period were associated with smaller white matter hyperintensity volume (a high volume is a sign of small-vessel disease in the brain).
Some small randomized intervention trials have shown that choline supplements improve cognitive performance in adults 19. However, a 2015 systematic review of 13 studies on the relationship between choline levels and neurological outcomes in adults found that choline supplements did not result in clear improvements in cognition in healthy adults 16. Similarly, a 2003 Cochrane review of 12 randomized trials in 265 patients with Alzheimer’s disease, 21 with Parkinsonian dementia, and 90 with self-identified memory problems found no clear clinical benefits of lecithin supplementation for treating Alzheimer’s disease or Parkinsonian dementia 15.
Another study published in 2017 2 found high choline intake during gestation and early postnatal development in rat and mouse models improves cognitive function in adulthood, prevents age-related memory decline, and protects the brain from the neuropathological changes associated with Alzheimer’s disease, and neurological damage associated with epilepsy, fetal alcohol syndrome, and inherited conditions such as Down and Rett syndromes. These effects of choline are correlated with modifications in histone and DNA methylation in brain, and with alterations in the expression of genes that encode proteins important for learning and memory processing, suggesting a possible epigenomic mechanism of action. Dietary choline intake in the adult may also influence cognitive function via an effect on phosphatidylcholine containing eicosapentaenoic and docosahexaenoic acids; polyunsaturated species of phosphatidylcholine whose levels are reduced in brains from Alzheimer’s disease patients, and is associated with higher memory performance, and resistance to cognitive decline. Moreover, choline is neuroprotective in a variety of experimental models of neuronal damage. Choline intake in adulthood may also be critical for normal cognitive function in people. Taken together, the available evidence strongly supports the notion that adequate choline intake during pregnancy, and throughout life, is an important determinant of brain development, cognitive performance in the adult, and resistance to cognitive decline associated with aging and neurodegenerative disease 2.
However despite these positive findings, future studies are needed to clarify the relationship between choline intakes and cognitive function and determine whether choline supplements might benefit patients with Alzheimer’s disease or other forms of dementia.
Nonalcoholic fatty liver disease
Nonalcoholic fatty liver disease (NAFLD) involves the accumulation of lipids in the livers of people who consume less than 20 g/day ethanol and who have no other known causes of steatosis 20. (A single drink [e.g., 12 oz beer, 5 oz wine, or 1.5 oz hard liquor] contains about 12–14 g alcohol.) It is the most common chronic liver disorder, present in up to 65% of overweight individuals and 90% of those with obesity 3. Although it is often benign, NAFLD can lead to steatohepatitis, fibrosis, cirrhosis, liver failure, and liver cancer 21. Choline, especially phosphatidylcholine, is essential for transporting lipids from the liver 3. Therefore, in choline deficiency, fat accumulates in the liver, which can result in nonalcoholic fatty liver disease (NAFLD) 22. Although most women of childbearing age are resistant to nonalcoholic fatty liver disease (NAFLD) because of their high estrogen levels, at least 40% have a polymorphism that makes them insensitive to activation of the gene by estrogen; adequate consumption of dietary choline is particularly important for this population 23.
Data from a single large observational study support a link between choline deficiency and risk of NAFLD. Specifically, a cross-sectional study of 56,195 Chinese adults aged 40–75 years found an inverse relationship between dietary choline intakes and risk of NAFLD based on 24-hour dietary recall 24. The risk of NAFLD was 32% lower in women in the highest quintile of choline intake (412 mg/day) compared to the lowest (179 mg/day) and 25% lower in men in the highest (452 mg/day) quintile compared to those in the lowest quintile (199 mg/day). However, choline intake was associated with NAFLD in normal-weight women only and not in those who were overweight or obese. This difference by weight status was not observed in men.
In a cross-sectional study of 664 adults and children from the Nonalcoholic Steatohepatitis Clinical Research Network, postmenopausal women who had nonalcoholic steatohepatitis (an extreme form of NAFLD involving liver inflammation and fibrosis) and a choline intake less than 50% of the AI had more severe fibrosis, but the results showed no relationship between choline intake and degree of liver steatosis 25.
Only limited data are available on the use of choline to treat NAFLD. For example, in a study of 57 adults who consumed a diet that included less than 50 mg choline per 70 kg body weight per day (<10% of the adequate intake) for up to 42 days, 37 of the participants developed liver dysfunction 22. Liver function returned to normal in 29 participants in this study after they were fed a diet containing 25%–75% of the choline adequate intake and in 8 who consumed an ad libitum diet. A pilot study in 15 adults on total parenteral nutrition (TPN) found that NAFLD resolved completely in all patients who received their usual total parenteral nutrition (TPN) regimen with an additional 2 g choline and in none of the patients who received their usual total parenteral nutrition (TPN) regimen only 26.
Adequate choline intake is needed for proper liver function and to prevent NAFLD, but more research is needed to further clarify the role of choline in preventing or treating NAFLD 27.
Choline recommended intakes
Intake recommendations for choline and other nutrients are provided in the Dietary Reference Intakes (DRIs) developed by the Food and Nutrition Board of the Institute of Medicine (IOM) 6. Dietary Reference Intakes (DRIs) is the general term for a set of reference values used for planning and assessing nutrient intakes of healthy people. These values, which vary by age and sex, include:
- Recommended Dietary Allowance (RDA): Average daily level of intake sufficient to meet the nutrient requirements of nearly all (97%–98%) healthy individuals; often used to plan nutritionally adequate diets for individuals.
- Adequate Intake (AI): Intake at this level is assumed to ensure nutritional adequacy; established when evidence is insufficient to develop an RDA.
- Estimated Average Requirement (EAR): Average daily level of intake estimated to meet the requirements of 50% of healthy individuals; usually used to assess the nutrient intakes of groups of people and to plan nutritionally adequate diets for them; can also be used to assess the nutrient intakes of individuals.
- Tolerable Upper Intake Level (UL): Maximum daily intake unlikely to cause adverse health effects.
Insufficient data were available to establish an estimated Average Requirement (EAR) for choline, so the Food and Nutrition Board established adequate intakes (AIs) for all ages that are based on the prevention of liver damage as measured by serum alanine aminostransferase levels 6. The amount of choline that individuals need is influenced by the amount of methionine, betaine, and folate in the diet; gender; pregnancy; lactation; stage of development; ability to produce choline endogenously; and genetic mutations that affect choline needs 6. Table 1 lists the current adequate intakes (AIs) for choline.
Table 1. Adequate Intakes (AIs) for Choline
| Age | Male | Female | Pregnancy | Lactation |
|---|---|---|---|---|
| Birth to 6 months | 125 mg/day | 125 mg/day | ||
| 7–12 months | 150 mg/day | 150 mg/day | ||
| 1–3 years | 200 mg/day | 200 mg/day | ||
| 4–8 years | 250 mg/day | 250 mg/day | ||
| 9–13 years | 375 mg/day | 375 mg/day | ||
| 14–18 years | 550 mg/day | 400 mg/day | 450 mg/day | 550 mg/day |
| 19+ years | 550 mg/day | 425 mg/day | 450 mg/day | 550 mg/day |
Most people in the United States consume less than the adequate intakes (AIs) for choline. An analysis of data from the 2013–2014 National Health and Nutrition Examination Survey (NHANES) found that the average daily choline intake from foods and beverages among children and teens is 256 mg for ages 2–19 28. In adults, the average daily choline intake from foods and beverages is 402 mg in men and 278 mg in women. Intakes from supplements contribute a very small amount to total choline intakes.
According to an analysis of 2007–2008 NHANES data, black males of all ages had lower mean choline intakes than their white and Hispanic counterparts, but choline intakes did not differ substantially among females of different races/ethnicities 29.
Sources of choline
Choline foods
Many foods contain choline 4. The main dietary sources of choline in the United States consist primarily of animal-based products that are particularly rich in choline—meat, poultry, fish, dairy products, and eggs 4. Cruciferous vegetables and certain beans are also rich in choline, and other dietary sources of choline include nuts, seeds, and whole grains.
About half the dietary choline consumed in the United States is in the form of phosphatidylcholine 30. Many foods also contain lecithin, a substance rich in phosphatidylcholine that is prepared during commercial purification of phospholipids; lecithin is a common food additive used as an emulsifying agent in processed foods, such as gravies, salad dressings, and margarine 8. Choline is also present in breast milk and is added to most commercial infant formulas 8. Precise estimates of the percentage absorption of the different forms of dietary choline in humans are not available 8.
Several food sources of choline are listed in Table 2.
Table 2. Foods high in choline
| Food | Milligrams (mg) per serving | Percent DV* |
|---|---|---|
| Beef liver, pan fried, 3 ounces | 356 | 65 |
| Egg, hard boiled, 1 large egg | 147 | 27 |
| Beef top round, separable lean only, braised, 3 ounces | 117 | 21 |
| Soybeans, roasted, ½ cup | 107 | 19 |
| Chicken breast, roasted, 3 ounces | 72 | 13 |
| Beef, ground, 93% lean meat, broiled, 3 ounces | 72 | 13 |
| Fish, cod, Atlantic, cooked, dry heat, 3 ounces | 71 | 13 |
| Mushrooms, shiitake, cooked, ½ cup pieces | 58 | 11 |
| Potatoes, red, baked, flesh and skin, 1 large potato | 57 | 10 |
| Wheat germ, toasted, 1 ounce | 51 | 9 |
| Beans, kidney, canned, ½ cup | 45 | 8 |
| Quinoa, cooked, 1 cup | 43 | 8 |
| Milk, 1% fat, 1 cup | 43 | 8 |
| Yogurt, vanilla, nonfat, 1 cup | 38 | 7 |
| Brussels sprouts, boiled, ½ cup | 32 | 6 |
| Broccoli, chopped, boiled, drained, ½ cup | 31 | 6 |
| Cottage cheese, nonfat, 1 cup | 26 | 5 |
| Fish, tuna, white, canned in water, drained in solids, 3 ounces | 25 | 5 |
| Peanuts, dry roasted, ¼ cup | 24 | 4 |
| Cauliflower, 1” pieces, boiled, drained, ½ cup | 24 | 4 |
| Peas, green, boiled, ½ cup | 24 | 4 |
| Sunflower seeds, oil roasted, ¼ cup | 19 | 3 |
| Rice, brown, long-grain, cooked, 1 cup | 19 | 3 |
| Bread, pita, whole wheat, 1 large (6½ inch diameter) | 17 | 3 |
| Cabbage, boiled, ½ cup | 15 | 3 |
| Tangerine (mandarin orange), sections, ½ cup | 10 | 2 |
| Beans, snap, raw, ½ cup | 8 | 1 |
| Kiwifruit, raw, ½ cup sliced | 7 | 1 |
| Carrots, raw, chopped, ½ cup | 6 | 1 |
| Apples, raw, with skin, quartered or chopped, ½ cup | 2 | 0 |
*DV = Daily Value. DVs were developed by the U.S. Food and Drug Administration (FDA) to help consumers compare the nutrient contents of products within the context of a total diet. The DV for choline is 550 mg for adults and children age 4 and older 31. However, the FDA does not require food labels to list choline content unless a food has been fortified with this nutrient. Foods providing 20% or more of the DV are considered to be high sources of a nutrient.
The U.S. Department of Agriculture’s (USDA’s) National Nutrient Database for Standard Reference 32 lists the nutrient content of many foods and provides a comprehensive list of foods containing choline arranged by choline content 33 and by food name 34.
Choline supplement
Choline is available in dietary supplements containing choline only, in combination with B-complex vitamins, and in some multivitamin/multimineral products 35. Typical amounts of choline in dietary supplements range from 10 mg to 250 mg. The forms of choline in dietary supplements include choline bitartrate, phosphatidylcholine, and lecithin. No studies have compared the relative bioavailability of choline from these different forms.
Choline is not known to have any clinically relevant interactions with medications.
Excessive choline health risks
High intakes of choline are associated with a fishy body odor, vomiting, excessive sweating and salivation, hypotension, and liver toxicity 3. Choline consumption has been shown to increase production of trimethylamine-N-oxide (TMAO), a substance that has been linked to a higher risk of cardiovascular disease, in a dose-dependent manner in adults.
The Food and Nutrition Board has established Upper Intake Levels (ULs) for choline from food and supplements based on the amounts of choline that are associated with hypotension and fishy body odor (see Table 3) 6. The ULs apply to healthy children and adults, but not to those taking high doses of choline under medical supervision. The Food and Nutrition Board was unable to establish Upper Intake Levels (ULs) for infants due to the lack of data on adverse effects in this age group.
Table 3: Tolerable Upper Intake Levels (ULs) for Choline
| Age | Male | Female | Pregnancy | Lactation |
|---|---|---|---|---|
| Birth to 6 months* | ||||
| 7–12 months* | ||||
| 1–3 years | 1,000 mg | 1,000 mg | ||
| 4–8 years | 1,000 mg | 1,000 mg | ||
| 9–13 years | 2,000 mg | 2,000 mg | ||
| 14–18 years | 3,000 mg | 3,000 mg | 3,000 mg | 3,000 mg |
| 19+ years | 3,500 mg | 3,500 mg | 3,500 mg | 3,500 mg |
*Not possible to establish; breast milk, formula, and food should be the only sources of choline for infants.
[Source 6]Choline deficiency
Choline deficiency can cause muscle damage, liver damage, and nonalcoholic fatty liver disease (NAFLD or hepatosteatosis) 3. Although most people in the United States consume less than the adequate intake (AI) of choline, frank choline deficiency in healthy, nonpregnant individuals is very rare, possibly because of the contribution of choline that the body synthesizes endogenously 36.
Groups at Risk of Choline Inadequacy
The following groups are among those most likely to have inadequate choline status.
Pregnant women
Approximately 90%–95% of pregnant women consume less choline than the adequate intake (AI) 37. Prenatal dietary supplements typically contain little if any choline 38. The risk of inadequate choline status might be greater in pregnant and lactating women who do not take folic acid supplements, those with low vitamin B12 status, and those with a common variant in methylenetetrahydrofolate dehydrogenase (an enzyme that can affect folate status), all of which reduce the body’s pool of methyl groups needed for metabolism 38.
Some evidence indicates that lower plasma or serum choline levels (e.g., serum concentration of 2.77 mmol/L in mid-pregnancy) are associated with an increased risk of neural tube defects 39, 40. However, other research found no relationship between plasma choline concentrations during pregnancy and neural tube defects in offspring 41.
People with certain genetic alterations
Genes involved in the metabolism of choline, folate, and methionine play a role in the pathways for choline production and use 42. Humans have variations in the DNA sequences for these genes (single nucleotide polymorphisms), and these single nucleotide polymorphisms can have a strong influence on demands for dietary choline. For example, one common single nucleotide polymorphism in the PEMT gene reduces endogenous synthesis of choline in women induced by estrogen 43. The prevalence of single nucleotide polymorphisms that alter requirements for dietary choline vary by race. In a study of 100 African, Asian, Caucasian, and Mexican Americans, individuals of European ancestry had a higher prevalence of four single nucleotide polymorphisms that increased the risk of organ dysfunction when these individuals consumed a low-choline diet 44.
Patients requiring total parenteral nutrition
At present, choline is not routinely added to commercial parenteral solutions for infants and adults 45. As a result, adults and infants receiving total parenteral nutrition (TPN) over the long term have low plasma choline concentrations (approximately 5 nmol/ml in adults and 5.7 nmol/ml in infants), which can result in hepatic abnormalities, including nonalcoholic fatty liver disease (NAFLD) 46. The American Society for Parenteral and Enteral Nutrition recommends the routine addition of choline to adult and pediatric parenteral nutrition formulations, and calls for the development of a commercially available parenteral product that contains choline 45.
References- Food and Nutrition Board . Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Panthotenic Acid, Biotin, and Cholin. National Academy Press; Washington, DC, USA: 1998
- Blusztajn JK, Slack BE, Mellott TJ. Neuroprotective Actions of Dietary Choline. Nutrients. 2017;9(8):815. doi:10.3390/nu9080815. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5579609/
- Zeisel SH, Corbin KD. Choline. In: Erdman JW, Macdonald IA, Zeisel SH, eds. Present Knowledge in Nutrition. 10th ed. Washington, DC: Wiley-Blackwell; 2012:405-18.
- Zeisel SH. Choline. In: Ross AC, Caballero B, Cousins RJ, Tucker KL, Ziegler TR, eds. Modern Nutrition in Health and Disease. 11th ed. Baltimore, MD: Lippincott Williams & Wilkins; 2014:416-26.
- Hollenbeck CB. An introduction to the nutrition and metabolism of choline. Cent Nerv Syst Agents Med Chem 2012;12:100-13.
- Institute of Medicine. Food and Nutrition Board. Dietary Reference Intakes: Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline. Washington, DC: National Academy Press; 1998.
- Holm PI, Ueland PM, Kvalheim G, Lien EA. Determination of choline, betaine, and dimethylglycine in plasma by a high-throughput method based on normal-phase chromatography-tandem mass spectrometry. Clin Chem 2003;49:286-94
- Zeisel SH. Choline. In: Coates PM, Betz JM, Blackman MR, et al., eds. Encyclopedia of Dietary Supplements. 2nd ed. London and New York: Informa Healthcare; 2010:136-43.
- Zheng Y, Li Y, Rimm EB, Hu FB, Albert CM, Rexrode KM, et al. Dietary phosphatidylcholine and risk of all-cause and cardiovascular-specific mortality among US women and men. Am J Clin Nutr 2016;104:173-80. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4919531/
- Bertoia ML, Pai JK, Cooke JP, Joosten MM, Mittleman MA, Rimm EB, et al. Plasma homocysteine, dietary B vitamins, betaine, and choline and risk of peripheral artery disease. Atherosclerosis 2014;235:94-101 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4102310/
- Bidulescu A, Chambless LE, Siega-Riz AM, Zeisel SH, Heiss G. Usual choline and betaine dietary intake and incident coronary heart disease: the Atherosclerosis Risk in Communities (ARIC) study. BMC Cardiovasc Disord 2007;7:20. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1934379/
- Dalmeijer GW, Olthof MR, Verhoef P, Bots ML, van der Schouw YT. Prospective study on dietary intakes of folate, betaine, and choline and cardiovascular disease risk in women. Eur J Clin Nutr 2008;62:386-94 https://www.ncbi.nlm.nih.gov/pubmed/17375117
- Zheng Y, Li Y, Rimm EB, Hu FB, Albert CM, Rexrode KM, et al. Dietary phosphatidylcholine and risk of all-cause and cardiovascular-specific mortality among US women and men. Am J Clin Nutr 2016;104:173-80 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4919531/
- Meyer KA, Shea JW. Dietary Choline and Betaine and Risk of CVD: A Systematic Review and Meta-Analysis of Prospective Studies. Nutrients. 2017;9(7):711. doi:10.3390/nu9070711. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5537826/
- Higgins JPT, Flicker L. Lecithin for dementia and cognitive impairment. Cochrane Database of Systematic Reviews 2000, Issue 4. Art. No.: CD001015. DOI: 10.1002/14651858.CD001015. http://cochranelibrary-wiley.com/doi/10.1002/14651858.CD001015/full
- Leermakers ET, Moreira EM, Kiefte-de Jong JC, Darweesh SK, Visser T, Voortman T, et al. Effects of choline on health across the life course: a systematic review. Nutr Rev 2015;73:500-22. https://academic.oup.com/nutritionreviews/article-abstract/73/8/500/1809796
- Nurk E, Refsum H, Bjelland I, Drevon CA, Tell GS, Ueland PM, et al. Plasma free choline, betaine and cognitive performance: the Hordaland Health Study. Br J Nutr 2013;109:511-9 https://www.ncbi.nlm.nih.gov/pubmed/22717142
- Poly C, Massaro JM, Seshadri S, Wolf PA, Cho E, Krall E, et al. The relation of dietary choline to cognitive performance and white-matter hyperintensity in the Framingham Offspring Cohort. Am J Clin Nutr 2011;94:1584-91 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3252552/
- Naber M, Hommel B, Colzato LS. Improved human visuomotor performance and pupil constriction after choline supplementation in a placebo-controlled double-blind study. Sci Rep 2015;5:13188. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4536529
- Veena J, Muragundla A, Sidgiddi S, Subramaniam S. Non-alcoholic fatty liver disease: need for a balanced nutritional source. Br J Nutr 2014;112:1858-72. https://www.ncbi.nlm.nih.gov/pubmed/25274101
- Corbin KD, Zeisel SH. Choline metabolism provides novel insights into nonalcoholic fatty liver disease and its progression. Curr Opin Gastroenterol 2012;28:159-65. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3601486/
- Fischer LM, daCosta KA, Kwock L, Stewart PW, Lu TS, Stabler SP, et al. Sex and menopausal status influence human dietary requirements for the nutrient choline. Am J Clin Nutr 2007;85:1275-85. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2435503/
- Resseguie ME, da Costa KA, Galanko JA, Patel M, Davis IJ, Zeisel SH. Aberrant estrogen regulation of PEMT results in choline deficiency-associated liver dysfunction. J Biol Chem 2011;286:1649-58 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3020773/
- Yu D, Shu XO, Xiang YB, Li H, Yang G, Gao YT, et al. Higher dietary choline intake is associated with lower risk of nonalcoholic fatty liver in normal-weight Chinese women. J Nutr 2014;144:2034-40. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4230213/
- Guerrerio AL, Colvin RM, Schwartz AK, Molleston JP, Murray KF, Diehl A, et al. Choline intake in a large cohort of patients with nonalcoholic fatty liver disease. Am J Clin Nutr 2012;95:892-900. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3302364/
- Buchman AL, Ament ME, Sohel M, Dubin M, Jenden DJ, Roch M, et al. Choline deficiency causes reversible hepatic abnormalities in patients receiving parenteral nutrition: proof of a human choline requirement: a placebo-controlled trial. JPEN J Parenter Enteral Nutr 2001;25:260-8. https://www.ncbi.nlm.nih.gov/pubmed/11531217
- Sherriff JL, O’Sullivan TA, Properzi C, Oddo J-L, Adams LA. Choline, Its Potential Role in Nonalcoholic Fatty Liver Disease, and the Case for Human and Bacterial Genes. Advances in Nutrition: An International Review Journal 2016;7:5-13. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4717871
- U.S. Department of Agriculture, Agricultural Research Service. Nutrient Intakes from Food and Beverages: Mean Amounts Consumed per Individual, by Gender and Age, What We Eat in America, NHANES 2013-2014. 2016 https://www.ars.usda.gov/northeast-area/beltsville-md-bhnrc/beltsville-human-nutrition-research-center/food-surveys-research-group/docs/wweia-data-tables/
- Chester DN, Goldman JD, Ahuja JK, Moshfegh AJ. Dietary Intakes of Choline: What We Eat in America, NHANES 2007-2008. Dietary Data Brief 2011;9:1-4.
- Leermakers ET, Moreira EM, Kiefte-de Jong JC, Darweesh SK, Visser T, Voortman T, et al. Effects of choline on health across the life course: a systematic review. Nutr Rev 2015;73:500-22. https://www.ncbi.nlm.nih.gov/pubmed/26108618
- U.S. Food and Drug Administration. https://www.federalregister.gov/documents/2016/05/27/2016-11867/food-labeling-revision-of-the-nutrition-and-supplement-facts-labels
- U.S. Department of Agriculture, Agricultural Research Service. USDA National Nutrient Database for Standard Reference. https://ndb.nal.usda.gov/ndb/
- https://ndb.nal.usda.gov/ndb/nutrients/report/nutrientsfrm?max=25&offset=0&totCount=0&nutrient1=421&nutrient2=&nutrient3=&subset=0&fg=&sort=c&measureby=m
- https://ndb.nal.usda.gov/ndb/nutrients/report/nutrientsfrm?max=25&offset=0&totCount=0&nutrient1=421&nutrient2=&nutrient3=&subset=0&fg=&sort=f&measureby=m
- National Institutes of Health. http://www.dsld.nlm.nih.gov/dsld/
- Hollenbeck CB. An introduction to the nutrition and metabolism of choline. Cent Nerv Syst Agents Med Chem 2012;12:100-13. https://www.ncbi.nlm.nih.gov/pubmed/22483274
- Brunst KJ, Wright RO, DiGioia K, Enlow MB, Fernandez H, Wright RJ, et al. Racial/ethnic and sociodemographic factors associated with micronutrient intakes and inadequacies among pregnant women in an urban US population. Public Health Nutr 2014;17:1960-70. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4071127/
- Caudill MA. Pre- and postnatal health: evidence of increased choline needs. J Am Diet Assoc 2010;110:1198-206. https://www.ncbi.nlm.nih.gov/pubmed/20656095
- Wu BT, Dyer RA, King DJ, Richardson KJ, Innis SM. Early second trimester maternal plasma choline and betaine are related to measures of early cognitive development in term infants. PLoS One 2012;7:e43448. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3423345/
- Shaw GM, Finnell RH, Blom HJ, Carmichael SL, Vollset SE, Yang W, et al. Choline and risk of neural tube defects in a folate-fortified population. Epidemiology 2009;20:714-9. https://www.ncbi.nlm.nih.gov/pubmed/19593156
- Mills JL, Fan R, Brody LC, Liu A, Ueland PM, Wang Y, et al. Maternal choline concentrations during pregnancy and choline-related genetic variants as risk factors for neural tube defects. Am J Clin Nutr 2014;100:1069-74. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4163794/
- Corbin KD, Zeisel SH. The nutrigenetics and nutrigenomics of the dietary requirement for choline. Prog Mol Biol Transl Sci 2012;108:159-77. https://www.ncbi.nlm.nih.gov/pubmed/22656377
- Biswas S, Giri S. Importance of choline as essential nutrient and its role in prevention of various toxicities. Prague Med Rep 2015;116:5-15. https://pmr.lf1.cuni.cz/media/pdf/pmr_2015116010005.pdf
- da Costa KA, Corbin KD, Niculescu MD, Galanko JA, Zeisel SH. Identification of new genetic polymorphisms that alter the dietary requirement for choline and vary in their distribution across ethnic and racial groups. Faseb j 2014;28:2970-8. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4062831/
- Vanek VW, Borum P, Buchman A, Fessler TA, Howard L, Jeejeebhoy K, et al. A.S.P.E.N. position paper: recommendations for changes in commercially available parenteral multivitamin and multi-trace element products. Nutr Clin Pract 2012;27:440-91. https://www.ncbi.nlm.nih.gov/pubmed/22730042
- Sentongo TA, Kumar P, Karza K, Keys L, Iyer K, Buchman AL. Whole-blood-free choline and choline metabolites in infants who require chronic parenteral nutrition therapy. J Pediatr Gastroenterol Nutr 2010;50:194-9. https://www.ncbi.nlm.nih.gov/pubmed/20038853