High Fiber Diet
Dietary fiber, also known as roughage or bulk, is the part of a plant that the body doesn’t absorb during digestion. Fibre is the part of food that is not digested in the small intestine. Dietary fibre moves largely unchanged into the large intestine or colon where it is fermented by friendly bacteria that live there. The scientific community define dietary fibre as intrinsic plant cell wall polysaccharides of vegetables, fruits and whole-grains, the health benefits of which have been clearly established, rather than synthetic, isolated or purified oligosaccharides and polysaccharides with diverse, and in some cases unique, physiological effects 1. Generally speaking, dietary fiber is the edible parts of plants or similar carbohydrates, that are resistant to digestion and absorption in the small intestine. Fiber can be soluble, which means it dissolves in water, or insoluble.
Eating a range of dietary fiber can:
- Improve the diversity of your microbiota
- Improve constipation and lactose intolerance
- Enhance immunity
- Reduce inflammation in your gut
Benefits of a high-fiber diet:
- Normalizes bowel movements. Dietary fiber increases the weight and size of your stool and softens it. A bulky stool is easier to pass, decreasing your chance of constipation. If you have loose, watery stools, fiber may help to solidify the stool because it absorbs water and adds bulk to stool.
- Helps maintain bowel health. A high-fiber diet may lower your risk of developing hemorrhoids and small pouches in your colon (diverticular disease). Studies have also found that a high-fiber diet likely lowers the risk of colorectal cancer. Some fiber is fermented in the colon. Researchers are looking at how this may play a role in preventing diseases of the colon.
- Lowers cholesterol levels. Soluble fiber found in beans, oats, flaxseed and oat bran may help lower total blood cholesterol levels by lowering low-density lipoprotein, or “bad,” cholesterol levels. Studies also have shown that high-fiber foods may have other heart-health benefits, such as reducing blood pressure and inflammation.
- Helps control blood sugar levels. In people with diabetes, fiber — particularly soluble fiber — can slow the absorption of sugar and help improve blood sugar levels. A healthy diet that includes insoluble fiber may also reduce the risk of developing type 2 diabetes.
- Aids in achieving healthy weight. High-fiber foods tend to be more filling than low-fiber foods, so you’re likely to eat less and stay satisfied longer. And high-fiber foods tend to take longer to eat and to be less “energy dense,” which means they have fewer calories for the same volume of food.
- Helps you live longer. Studies suggest that increasing your dietary fiber intake — especially cereal fiber — is associated with a reduced risk of dying from cardiovascular disease and all cancers.
Fiber includes carbohydrates called polysaccharides and resistant oligosaccharides. Recent research suggests that fiber should be categorized by its physical characteristics; how well it dissolves (solubility), how thick it is (viscosity) and how well it breaks down (fermentability). Some commonly known terms are described below:
- Soluble fiber including pectins and beta glucans is found in foods like oats, peas, beans, apples, citrus fruits, carrots, barley and psyllium. Soluble fiber dissolves in water to form a gel-like material. It can help lower blood cholesterol and glucose levels. Foods high in soluble fiber can help you feel full. They also help reduce constipation by speeding up the time it takes for feces (poop) to pass through your body.
- Insoluble fiber including cellulose is found in wheat bran and nuts. Insoluble fiber is a type of fibre that doesn’t dissolve in water. Because insoluble fiber absorbs water, it helps to soften the contents of your bowel, contributing to keep the bowels regular. Insoluble fiber promotes the movement of material through your digestive system and increases stool bulk, so it can be of benefit to those who struggle with constipation or irregular stools. Whole-wheat flour, wheat bran, nuts, beans and vegetables, such as cauliflower, green beans and potatoes, are good sources of insoluble fiber.
- Resistant starch is a soluble fiber that is highly fermentable in the gut. It gets broken down by good bacteria to produce short chain fatty acids (SCFAs). Resistant starch is naturally present in some foods such as bananas, potatoes, grains and pulses. Freekeh, a Middle Eastern grain available in some supermarkets, is another good source. In general, foods that are less highly processed contain more resistant starch. Different ways of cooking can create different amounts of resistant starch. For example, resistant starch is found in slightly undercooked (‘al dente’) pasta, cooked but cooled potatoes (including potato salad), under-ripe bananas, beans, lentils and a product called Hi-maize used in some breads and breakfast cereals.
- Prebiotics are types of carbohydrate that only our gut bacteria can feed upon. Some examples are onions, garlic, asparagus and banana
The amount of soluble and insoluble fiber varies in different plant foods. To receive the greatest health benefit, eat a wide variety of high-fiber foods.
Good sources of dietary fiber include:
- Pulses (like lentils and peas) and beans and legumes (think navy beans, small white beans, split peas, chickpeas, lentils, pinto beans)
- Fruits and vegetables, vegetables such as carrots, broccoli, green peas, and collard greens; fruits especially those with edible skin (like pears and apples with the skin on) and those with edible seeds (like berries)
- Nuts—try different kinds (pumpkin seeds, almonds, sunflower seeds, pistachios and peanuts are a good source of fiber and healthy fats, but be mindful of portion sizes, because they also contain a lot of calories in a small amount!)
- Whole grains such as:
- Quinoa, barley, bulgur, oats, brown rice and farro
- Whole wheat pasta
- Whole grain cereals, including those made from whole wheat, wheat bran and oats
Choose fiber rich foods from a variety of sources including wholegrains, fruit and vegetable, nuts and seeds, beans and pulses. When you read food labels check for the grams of fiber per serving or per 100g. Foods that are naturally high in fiber and contain at least 3 grams per 100 gram are often labeled as a “good source,” and foods labeled as “excellent source” contain more than 5 grams of fiber per serving.
Depending on your age and sex, adults should get 25 to 31 grams of fiber a day 2. Older adults sometimes don’t get enough fiber because they may lose interest in food.
- Men over the age of 50 should get at least 38 grams of fiber per day.
- Women over the age of 50 should get 25 grams per day.
- Children ages 1 to 3 should get 19 grams of fiber per day.
- Children between 4 and 8 years old should get 25 grams per day.
- Girls between 9 and 18 should get 26 grams of fiber each day. Boys of the same age range should get between 31 and 38 grams of fiber per day.
High-fiber foods are good for your health. But adding too much fiber too quickly can promote intestinal gas, abdominal bloating and cramping. Increase fiber in your diet gradually over a period of a few weeks. This allows the natural bacteria in your digestive system to adjust to the change.
Also, drink plenty of water. Fiber works best when it absorbs water, making your stool soft and bulky.
Tips for fitting in more fiber into your diet:
- For breakfast choose a high-fiber breakfast cereal — 5 or more grams of fiber a serving. Opt for cereals with “whole grain,” “bran” or “fiber” in the name. Or add a few tablespoons of unprocessed wheat bran to your favorite cereal.
- Switch to whole grains. Consume at least half of all grains as whole grains. Look for breads that list whole wheat, whole-wheat flour or another whole grain as the first ingredient on the label and have least 2 grams of dietary fiber a serving. Experiment with brown rice, wild rice, barley, whole-wheat pasta and bulgur wheat.
- Bulk up baked goods. Substitute whole-grain flour for half or all of the white flour when baking. Try adding crushed bran cereal, unprocessed wheat bran or uncooked oatmeal to muffins, cakes and cookies.
- Lean on legumes. Beans, peas and lentils are excellent sources of fiber. Add kidney beans to canned soup or a green salad. Or make nachos with refried black beans, lots of fresh veggies, whole-wheat tortilla chips and salsa.
- Eat more fruit and vegetables. Fruits and vegetables are rich in fiber, as well as vitamins and minerals. Try to eat five or more servings daily.
- Make snacks count. Fresh fruits, raw vegetables, low-fat popcorn and whole-grain crackers are all good choices. An occasional handful of nuts or dried fruits also is a healthy, high-fiber snack — although be aware that nuts and dried fruits are high in calories.
Today more than 80 percent of the U.S. population eats less than the recommended amount of vegetables, about 70 percent of the population eats more saturated fat, sodium and added sugar than is recommended. That is because the top three sources of calories in the U.S. are burgers, sandwiches and tacos; followed by desserts, sweet snacks and sugar-sweetened beverages, according to the most recent data from the National Health and Nutrition Examination Survey 3. Poor diet is classified as a diet high in saturated fat, low dietary fiber and high simple carbohydrates. This diet would be consistent with a high glycemic index and high glycemic load being higher in easily digestible and rapidly absorbable carbohydrates. In a supportive, eight year study of over 90,000 female nurses, Shuzle et al. 4 found a positive correlation between glycemic index and risk of type two diabetes. This association was significant even after adjusting for age, body mass index (BMI) and family history. Several means have been proposed to understand the physiology behind the relationship of glycemic index and diabetes. First, carbohydrates with a higher glycemic index produce higher blood glucose levels. This chronic hyperglycemia is suggested to lead to the dysfunction of beta cells in the pancreas thus decreasing insulin release. Second, due to an over abundance of energy (i.e., high glycemic load) tissues such as skeletal muscle, liver and adipose become resistant to insulin.
Although a majority of studies show a positive correlation between high glycemic foods and type 2 diabetes, several studies disagree with these findings. Meyer et al. 5 found that glycemic index had no effect on the prevalence of type 2 diabetes in older aged women. However, there was a strong inverse relationship between dietary fiber intake and type 2 diabetes when adjusted for age and BMI. Women consuming an average of 26 g/day of dietary fiber had a 22% lower risk of developing diabetes when compared to women only consuming 13 g/d. Schulze et al. 4 agreed with these findings with men and women showing a decreased risk of diabetes with the consumption of an additional 12 g of dietary fiber per day. According to these findings, it may be more significant to focus on an increased consumption of dietary fiber to prevent diabetes than glycemic index/load. It is also important to note that the inverse relationship between dietary fiber and diabetes observed by Meyer et al. 6 and Schulze et al. 4 was independent of age and body weight. Hu et al. 7 supported these findings while correcting for age, fat intake, smoking, alcohol, family history, exercise, and body weight. Therefore, it seems that dietary fiber is associated with type two diabetes, independent of other compounding factors.
According to recent research, the soluble versus insoluble fraction of fiber may give some insight on the efficacy of dietary fiber on diabetes and its mechanisms. Although some studies have been contradictory, showing no differentiation between soluble and insoluble fiber on diabetes 4, a majority of the research demonstrates a strong inverse relationship between insoluble fiber and the risk of type two diabetes. Meyer et al. 5 using healthy middle aged women, observed a strong inverse relationship between insoluble fiber and the risk of type two diabetes while soluble fiber had no effect. Montonen et al. 8 also found the same results in healthy middle aged men and women consuming increased levels of whole rye bread. Interestingly, fiber from fruits and vegetables had no effect on the risk of developing type two diabetes. Earlier studies have agreed with these findings. A large epidemiological study of 42,000 men found that dietary fiber from fruits or vegetables had no effect on the risk of diabetes. However, dietary fiber from whole cereal grains showed a significant decrease in diabetes occurrence 9. Daily intakes of fiber among all groups were similar.
Insoluble fiber only has a small effect on macronutrient absorption 10. Therefore, another mode of action must be present and several hypotheses should be discussed. Some suggest that insoluble fiber increases the passage rate of foodstuff through the gastrointestinal tract thus resulting in a decreased absorption of nutrients, namely simple carbohydrates. However, Weicket et al. 11 found that an increased intake of cereal fiber significantly improved whole body glucose disposal resulting in an 8% improvement of insulin sensitivity. This suggests that the mechanisms behind insoluble fiber are more peripheral and not limited to nutrient absorption. First, an accelerated secretion of glucose-dependent insulintropic polypeptide was observed directly after the ingestion of an insoluble fiber in healthy women 12. Glucose-dependent insulintropic polypeptide is an incretin hormone which stimulates postprandial insulin release. Second, insoluble fiber can result in a reduced appetite and food intake 13. This may lead to a decreased caloric intake and BMI. Third, short chain fatty acids, via fermentation, have been shown to reduce postpandrial glucose response 14, 15. Early research demonstrated that lipid infusions impaired glucose utilization 16 and oral acetate could decrease free fatty acids in the blood 17. According to Kelley and Mandarino 18, increases in free fatty acid in the blood can inhibit glucose metabolism through the inhibition of GLUT 4 transporters. Therefore, short chain fatty acids, by way of decreasing serum free fatty acids, may reduce blood glucose levels through competition in insulin–sensitive tissues.
As a result of the poor diet, many Americans are not getting adequate amount of nutrients. These include potassium, dietary fiber, choline, magnesium, calcium, and vitamins A, D, E, and C. Iron also is under consumed by adolescent girls and women ages 19 to 50 years. Low intakes for most of these nutrients occur within the context of unhealthy overall eating patterns, due to low intakes of the food groups—vegetables, fruits, whole grains, and dairy—that contain these nutrients. Low intakes of dietary fiber are due to low intakes of vegetables, fruits, and whole grains. Low intakes of potassium are due to low intakes of vegetables, fruits, and dairy. Low intakes of calcium are due to low intakes of dairy. 19.
Dietary fiber and whole grains contain a unique blend of bioactive components including resistant starches, vitamins, minerals, phytochemicals and antioxidants. As a result, research regarding their potential health benefits has received considerable attention in the last several decades. Epidemiological and clinical studies demonstrate that consumption of dietary fiber and whole grain intake is inversely related to obesity 20, type two diabetes 5, cancer 21 and cardiovascular disease 22.
- Eating fibre and wholegrain foods is linked to a lower risk of obesity, type 2 diabetes and heart disease, and may also reduce the risk of bowel cancer.
- Eating high fibre foods can also help prevent constipation – this in turn can help to prevent hemorrhoids.
- Because high fibre foods are filling they may also make it easier to stay at a healthy weight.
- Foods high in fibre are generally good sources of vitamins and minerals, as well as other important nutrients.
The Food and Drug Administration (FDA) has approved two health claims for dietary fiber. The first claim states that, along with a decreased consumption of fats (<30% of calories), an increased consumption of dietary fiber from fruits, vegetables and whole grains may reduce some types of cancer 23. “Increased consumption” is defined as six or more one ounce equivalents, with three ounces derived from whole grains. A one ounce equivalent would be consistent with one slice of bread, ½ cup oatmeal or rice, or five to seven crackers. The second FDA claim supporting health benefits of dietary fiber states that diets low in saturated fat (<10% of calories) and cholesterol and high in fruits, vegetables and whole grain, have a decreased risk of leading to coronary heart disease 24. For most, an increased consumption of dietary fiber is considered to be approximately 25 to 35 g/d, of which 6 g are soluble fiber.
Dietary fiber and whole grains are an abundant source of nutrients including vitamins, minerals, and a slowly digestible energy. In addition, they contain phytochemicals such as phenolics, carotenoids, lignans, beta-glucan and inulin. These chemicals, secreted by plants, are not currently classified as essential nutrients but may be important factors in human health 25. The synergistic effect of phytochemicals, increased nutrient content and digestive properties, are believed to be the mechanism behind dietary fibers beneficial effects on the treatment and prevention of obesity and diabetes 26, 27, reduced cardiovascular disease 28 and decreased incidence of certain types of cancer 29, 30.
Recent studies support this inverse relationship between dietary fiber and the development of several types of cancers including colorectal, small intestine, oral, larynx and breast 21, 31, 32. Although most studies agree with these findings, the mechanisms responsible are still unclear. Several modes of actions however have been proposed. First, dietary fiber resists digestion in the small intestine, thereby allowing it to enter the large intestine where it is fermented to produce short chain fatty acids, which have anti-carcinogenic properties 33. Second, since dietary fiber increases fecal bulking and viscosity, there is less contact time between potential carcinogens and mucosal cells. Third, dietary fiber increases the binding between bile acids and carcinogens. Fourth, increased intake of dietary fiber yield increased levels of antioxidants. Fifth, dietary fiber may increase the amount of estrogen excreted in the feces due to an inhibition of estrogen absorption in the intestines 34. Obviously, many studies support the inverse relationship of dietary fiber and the risk for coronary heart disease. However, more recent studies found interesting data illustrating that for every 10 g of additional fiber added to a diet the mortality risk of coronary heart disease decreased by 17–35% 35, 22. Risk factors for CHD include hypercholesterolemia, hypertension, obesity and type two diabetes. It is speculated that the control and treatment of these risk factors underlie the mechanisms behind dietary fiber and coronary heart disease prevention. First, soluble fibers have been shown to increase the rate of bile excretion therefore reducing serum total and LDL “bad” cholesterol 36. Second, short chain fatty acid production, specifically propionate, has been shown to inhibit cholesterol synthesis 37. Third, dietary fiber demonstrates the ability to regulate energy intake thus enhancing weight loss or maintenance of a healthier body weight. Fourth, either through glycemic control or reduced energy intake, dietary fiber has been shown to lower the risk for type two diabetes. Fifth, dietary fiber has been shown to decrease pro-inflammatory cytokines such as interleukin-18 which may have an effect on plaque stability 38. Sixth, increasing dietary fiber intake has been show to decrease circulating levels of C-Reactive protein, a marker of inflammation and a predictor for coronary heart disease 39.
Fiber supplements and fortified foods
Whole foods rather than fiber supplements are generally better. Fiber supplements — such as Metamucil, Citrucel and FiberCon — don’t provide the variety of fibers, vitamins, minerals and other beneficial nutrients that foods do.
Another way to get more fiber is to eat foods, such as cereal, granola bars, yogurt, and ice cream, with fiber added. The added fiber usually is labeled as “inulin” or “chicory root.” Inulin, a plant compound commonly extracted from chicory root that can make low-fat foods taste creamier and add sweetness. Inulin also is derived from byproducts of sugar production from beets. Soluble corn fiber, which replaces traditional sweeteners as well as adding fiber, is also turning up on ingredient lists.
However be vigilant and cautious about eating certain foods with added fiber, especially if they come with lots of calories with added sugar, salt, or fat.
Some people complain of gassiness after eating foods with added fiber.
However, some people may still need a fiber supplement if dietary changes aren’t sufficient or if they have certain medical conditions, such as constipation, diarrhea or irritable bowel syndrome. Check with your doctor before taking fiber supplements.
What is Dietary Fiber?
Dietary fiber is found mainly in plant-based foods including fruits, vegetables, wholegrains and legumes — is probably best known for its ability to prevent or relieve constipation. Dietary fiber also known as roughage or bulk, includes the parts of plant foods your body can’t digest or absorb. Generally speaking, dietary fiber is the edible parts of plants, or similar carbohydrates, that are resistant to digestion and absorption in the small intestine. Unlike other food components, such as fats, proteins or carbohydrates — which your body breaks down and absorbs — fiber isn’t digested by your body. Instead, it is completely or partially broken down (fermented) by bacteria in your large intestine. Once broken down in your large intestine, it has been suggested that dietary fibers increase the good bacteria in your gut. This improves your immune system and supports your immunity against inflammatory disorders and allergies. Furthermore, a high fiber diet can provide other health benefits as well, such as helping you to maintain a healthy weight and lowering your risk of type 2 diabetes, heart disease and some types of cancer including bowel cancer. For example, high quality randomized controlled trials have shown that eating oat bran leads to lower blood pressure and lower total cholesterol.
Dietary fiber can be separated into many different fractions. Recent research has begun to isolate these components and determine if increasing their levels in a diet is beneficial to human health. These fractions include arabinoxylan, inulin, pectin, bran, cellulose, β-glucan and resistant starch. The study of these components may give us a better understanding of how and why dietary fiber may decrease the risk for certain diseases 40.
Over the years, the definition of dietary fibre has been subject to much discussion. The most recent definitions, from about 2008 41, have general global agreement. Dietary fibre is made up of carbohydrate polymers with three or more monomeric units (MU), which are neither digested nor absorbed in the human intestine and includes: (1) Non Starch Polysaccharides (NSP) from fruits, vegetables, cereals and tubers whether intrinsic or extracted, chemically, physically and/or enzymically modified or synthetic (MU≥10); (2) resistant (non-digestible) oligosaccharides (RO) (MU 3–9); and (3) resistant starch (RS) (MU≥10). When extracted, chemically, physically and/or enzymically modified or synthetic, generally accepted scientific evidence of benefits for health must be demonstrated to consider the polymer as dietary fibre. Most definitions also include ‘associated substances’, which are non-carbohydrate such as lignin and substances which are present in cell walls linked to polysaccharides and quantified as dietary fibre by the accepted analytical methods (Table 1).
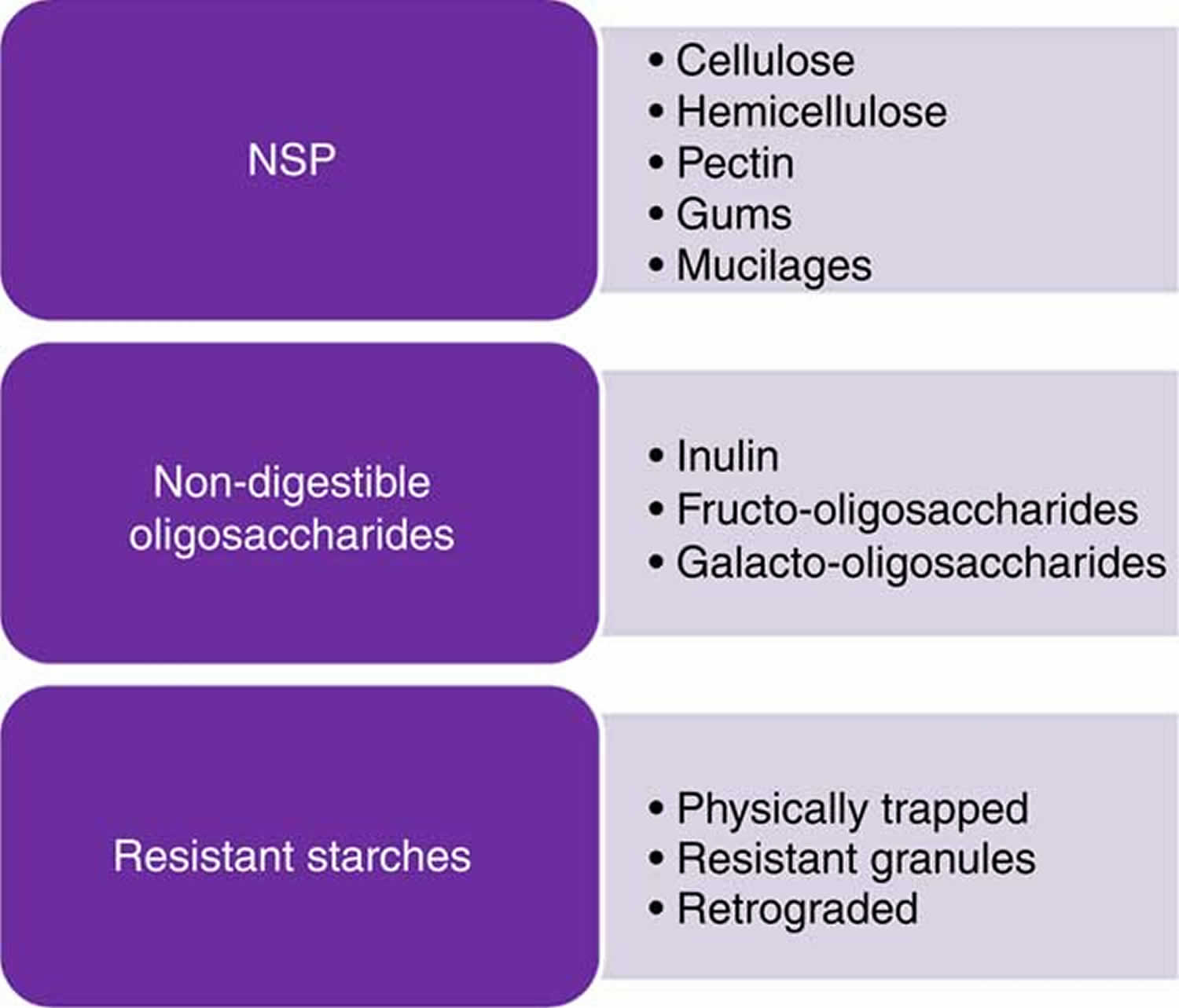
Dietary fiber is often subdivided into oligosaccharides (monosaccharide units number 3–9) resistant oligosaccharides (RO) and polysaccharides, including non-starch polysaccharides (NSP) and resistant starch (RS), with a minimum MU number of 10 42, provided in an overview in Figure 1. Dietary fiber also comprises ‘associated substances’.
Table 1. Principal definitions of dietary fiber
[Source 43 ]Footnote: MU = monosaccharide units; DF = dietary fiber; RS = resistant starch; RO = resistant oligosaccharides; DP = degree of polymerisation; NDC = non-digestible carbohydrates; NSP = Non Starch Polysaccharides. The minimum number of carbohydrate monosaccharide units to be included; the European Union (EU) definition prescribes a minimum monosaccharide units number (sometimes called ‘degree of polymerisation’) of 3, while Codex Alimentarius prescribes a minimum of 10 but leaves to the local authority the decision on whether or not to include carbohydrates with an monosaccharide units number of 3 to 9. Since many countries outside the EU have no local regulatory definition, the default is to adopt the Codex definition of monosaccharide units number≥10. As a consequence, in these countries, resistant oligosaccharides (RO) are or are not considered as dietary fibre, independently from the demonstration of a beneficial effect.
Figure 1. Some examples of potential fiber sources.
Footnote: NSP = Non Starch Polysaccharides
[Source 43 ]Unlike other food components, such as fats, proteins or carbohydrates — which your body breaks down and absorbs — fiber isn’t digested by your body. Instead, it passes relatively intact through your stomach, small intestine and colon and out of your body.
The American Association of Cereal Chemists 44, define “dietary fiber is the edible parts of plants or analogous carbohydrates that are resistant to digestion and absorption in the human small intestine with complete or partial fermentation in the large intestine. Dietary fiber includes polysaccharides, oligosaccharides, lignin, and associated plants substances. Dietary fibers promote beneficial physiological effects including laxation, and/or blood cholesterol attenuation, and/or blood glucose attenuation” 45. The World Health Organization (WHO) and Food and Agriculture Organization (FAO) agree with the American Association of Cereal Chemists International (AACCI) definition but with a slight variation. They state “Dietary fibre means carbohydrate polymers1 with ten or more monomeric units, which are not hydrolysed by the endogenous enzymes in the small intestine of humans and belong to the following categories: that dietary fiber is a polysaccharide with ten or more monomeric units which is not hydrolyzed by endogenous hormones in the small intestine” 46.
Recent research has begun to isolate these components and determine if increasing their levels in a diet is beneficial to human health. The separation of these fractions may give us a better understanding of how and why dietary fiber may decrease the risk for certain diseases.
Table 2. Components of dietary fiber according to the American Association of Cereal Chemists
| Non Starch Polysaccharides and Oligosaccharides | |
| Cellulose | |
| Hemicellulose | |
| Arabinoxylans | |
| Arabinogalactans | |
| Polyfructoses | |
| Inulin | |
| Oligofructans | |
| Galacto-oligosaccharides | |
| Gums | |
| Mucilages | |
| Pectins | |
| Analagous carbohydrates | |
| Indigestible dextrins | |
| Resistant maltodextrins | |
| Resistant potato dextrins | |
| Synthesized carbohydrates compounds | |
| Polydextrose | |
| Methyl cellulose | |
| Hydroxypropylmethyl cellulose | |
| Resistant starches | |
| Lignin substances associated with the Non Starch Polysaccharides (NSP) and lignin complex | |
| Waxes | |
| Phytate | |
| Cutin | |
| Saponins | |
| Suberin | |
| Tannin | |
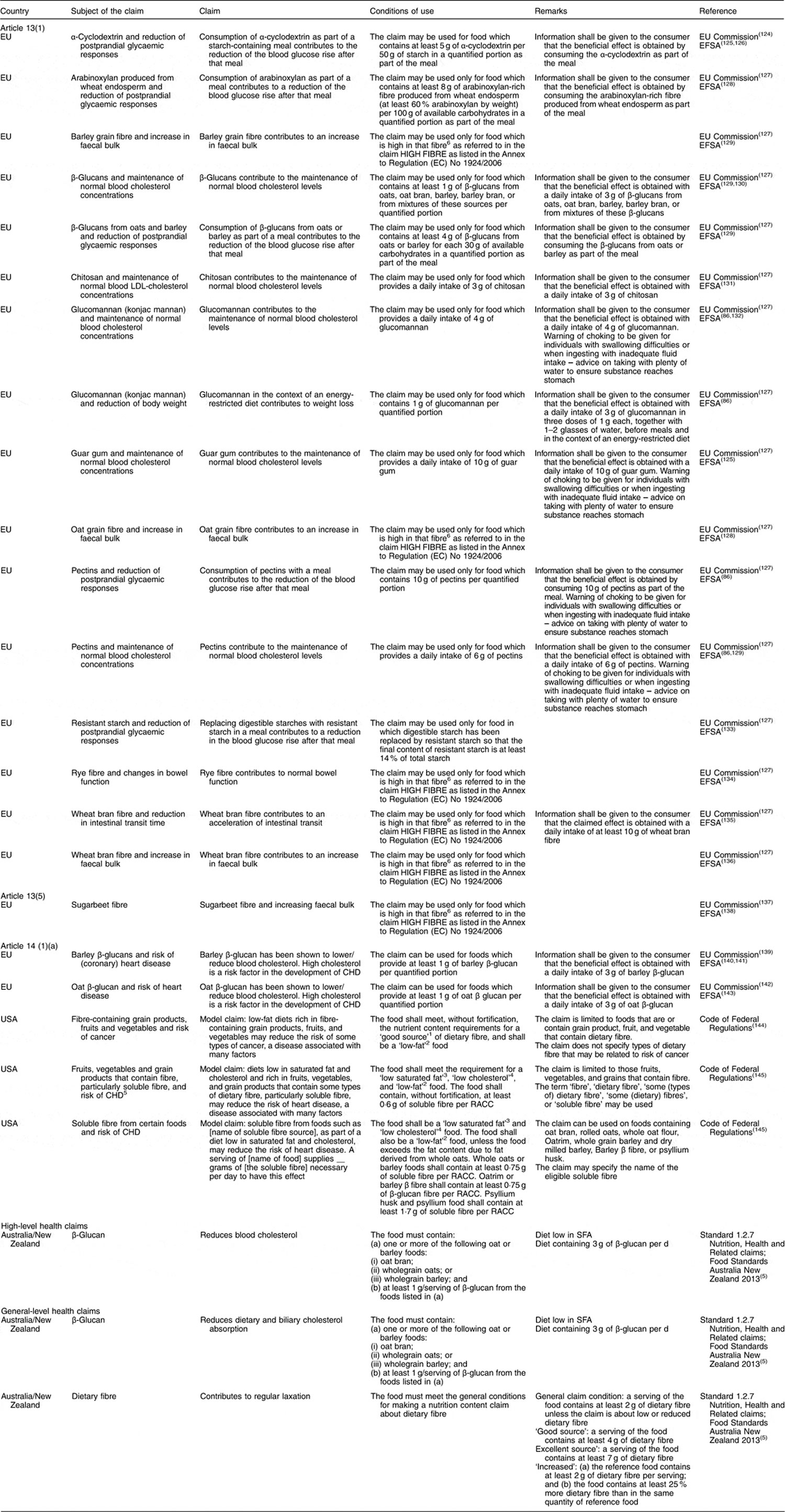
Table 3. Authorized health claims related to dietary fibre in the European Union (EU) (European Commission 2014), in the USA (Food and Drug Administration 2013) and in Australia/New Zealand (Food Standards Australia New Zealand 2013)
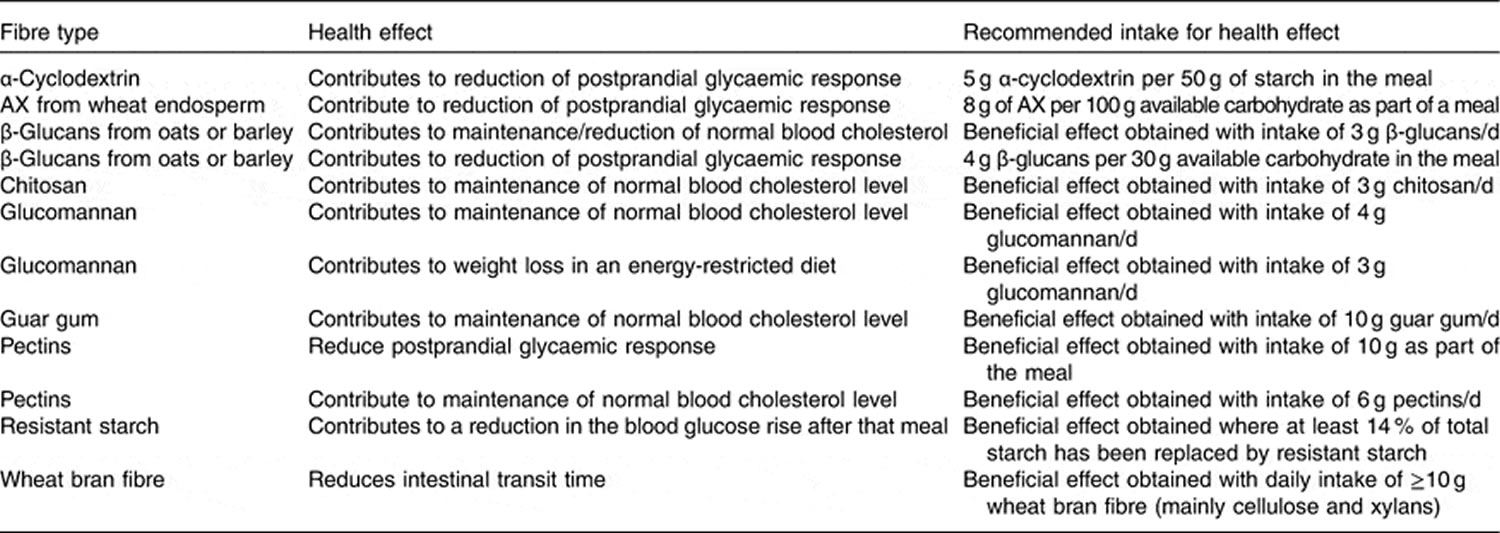
[Source 43]Table 4. Recommended for intake amount of specific fibers (subtypes of fibres) based on health claims
[Source 43]Arabinoxylan
Arabinoxylan, a constituent of hemicelluloses, is comprised of a xylose backbone with arabinose side chains. Arabinoxylan is a major component of dietary fiber in whole grains having considerable inclusions in both the endosperm and bran. In wheat, Arabinoxylan account for around 64–69% of the structural carbohydrates (complex carbohydrates, non-starch polysaccharide are resistant to digestion in the small intestine and require bacterial fermentation located in the large intestine) in the bran and around 88% in the endosperm 47. During normal wheat flour processing, a majority of the arabinoxylan is removed as a by-product. In the GI tract, arabinoxylan acts much like a soluble fiber being rapidly fermented by the microflora of the colon.
Lu et al. 48, observed an inverse relationship between the intake level of an arabinoxylan rich bread and postprandial glucose response in healthy adult subjects. When compared to the control, postprandial glucose levels were significantly lower with only 6 g of arabinoxylan rich fiber supplementation while 12 g produced the greatest benefit. Breads high in arabinoxylan also appear to control blood glucose and insulin in adults with an already impaired glucose tolerance 49. Fasting blood glucose, postprandial blood glucose and insulin were all significantly lower when adults with type two diabetes were supplemented with 15 g/d of an arabinoxylan rich fiber. The mode of action behind arabinoxylan on improving glucose tolerance is unknown. However, it is thought to be due to the high viscosity of the fiber inside the lumen of the GI tract, thereby slowing the rate of glucose absorption.
The lower glycemic index of arabinoxylan may also play a role. Breads made with a flour rich in arabinoxylan have a relatively low glycemic index of around 59. Whole wheat flour, although high in fiber, has a glycemic index of around 99 48. Arabinoxylan rich bread has a similar glycemic index to that of whole grain bread but offers some distinct advantages such as improved mouth feel and tenderness. There was no significant difference in the sensory analysis between the control and a bread containing 14% arabinoxylan rich fiber 48.
Inulin
Inulin is a polymer of fructose monomers and is present in such foods as onions, garlic, wheat, artichokes and bananas and is used to improve taste and mouthfeel in certain applications. It is also used as a functional food ingredient due to its nutritional properties. Because of this, inulin products can be used as a replacement for fat or soluble carbohydrates without affecting the taste and texture and still contribute to a foods nutritional value.
Enzymatic hydrolyses in the small intestine is minimal (<10%) since inulin consists of beta bonds. Therefore, it enters the large intestine and is almost completely metabolized by the microflora. When fermented, they tend to favor propionate production which, in turn, decreases the acetate to propionate ratio leading to decreased total serum cholesterol and LDL 37, which are important risk factors for coronary heart disease.
Inulin has also demonstrated the ability to contribute to the health of the human large intestine as a prebiotic 50. They demonstrated that inulin stimulated the growth of bifidobacteria while restricting the growth of potential pathogenic bacteria such as E. coli, Salmonella, and Listeria. This could prove to be beneficial in such disorders as ulcerative colitis and C. difficile infections. Rafter et al. 51 agreed with these findings and suggested they were the underlying mechanisms behind the observation that inulin decreased biological compounds associated with colonic cancer, including reduced colorectal cell proliferation and water induced necrosis, decreased exposure to genotoxins, and decreased interleukin-2 release.
Increased mineral absorption may also contribute to the functionality of inulin. Increased calcium absorption, by approximately 20%, was reported in adolescent girls supplemented with inulin 52. Results from Abrams et al. 53, support these findings in a longer (one year) study of pubertal boys and girls consuming an inulin supplement. Subjects in the treatment group also experienced increased bone mineral density when compared to the control. The mechanisms behind these findings are still unclear but may be due to increased calcium absorption from the colon or possibly an increased solubility in the lumen of the GI tract due to short chain fatty acids. Finally, it may increase absorption through an enhancement of vitamin D.
Inulin may also provide a way to prevent and treat obesity. Cani et al. 54 demonstrated that oligofructose, a subgroup of inulin, increased satiety in adults which led to a decrease in total energy intake. This is thought to be due to short chain fatty acids and their ability to increase appetite suppressing hormones such as glucagon-like peptide 1 (GLP-1).
Beta-glucan
Beta-glucan (β-glucan) is a linear polysaccharide of glucose monomers with β(1→4) and β(1→3) linkages and found in the endosperm of cereal grains, primarily barley and oats. β-glucan concentrations in North American oat cultivars range from 3.9% to 6.8% 55. β-glucan is water soluble and highly viscous at low concentrations 55.
The physiological benefits due to β-glucan seem to stem from their effect on lipid metabolism and postprandial glucose metabolism. Many studies agree an inverse relationship exists between consumption levels of β-glucan and cholesterol levels. Several recent studies, in both hypercholesterolemic 56 and healthy 57 subjects, found that the daily consumption of 5 g of β–glucan significantly decreased serum total and LDL cholesterol. Davidson et al. 58 found that only a daily consumption of 3.6 g β-glucan was needed to produce the same significant effects. The same relationship also has been reported to occur between β-glucan and postprandial glucose and insulin responses in both diabetic and healthy subjects. Biorklund et al. 59 found that 5 g of β-glucan from oats significantly decreased postprandial glucose and insulin levels in healthy adults. Tappy et al. 60 reported the same results in adult subjects diagnosed with type two diabetes who consumed 4.0, 6.0 or 8.4 g of β-glucan.
Most authors agree that β-glucan’s viscosity in the GI tract is the most probable mechanism in which it decreases serum cholesterol levels as well as improves post prandial glucose metabolism. This gellation property may decrease bile acid absorption by increasing intestinal viscosity and increase bile acid excretion. This subsequently results in a higher hepatic cholesterol synthesis because of the higher need for bile acid synthesis 61. The same viscosity may also delay glucose absorption into the blood thus lowering post prandial glucose and insulin levels. Nazare et al. 62 observed that 5 g of oat β–glucan added to an oat concentrate cereal significantly delayed, but did not reduce, total glucose absorption.
The production of short chain fatty acids from β-glucan may also be a probable mechanism behind its observed metabolic effects. Fermentation of oat β-glucan has been shown to yield larger amounts of propionate 63, 64. Propionate has been shown to significantly inhibit cholesterol synthesis in humans 37 and is thought to be due to the inhibition of the rate limiting enzyme HMG CoA reductase 65.
Not all research however, agrees that β-glucan can affect lipid and glucose absorption/metabolism. Keogh et al. 66 observed that treatments of 8.1 to 11.9 g/d of barley β-glucan had no effect on total or LDL cholesterol in mildly hyperlipidemic adults. Cugent-Anceau et al. 67 not only observed that 3.5 g of oat β-glucan added to soup did not alter serum lipid profiles, but also produced no change in postprandial glucose levels.
Pectin
Pectin is a linear polymer of galacturonic acid connected with α (1→4) bonds. Regions of this backbone are substituted with α (1→2) rhamnopyranose units from which side chains of neutral sugars such as galactose, mannose, glucose and xylose occur. Pectin is a water soluble polysaccharide that bypasses enzymatic digestion of the small intestine but is easily degraded by the microflora of the colon. Citrus fruit contains anywhere from 0.5% to 3.5% pectin with a large concentration located in the peel. Commercially extracted pectins are also available and are typically used in food applications which require a gelling or a thickening agent.
Inside the GI tract, pectin maintains this ability to form a gel or thicken a solution. This is thought to be the likely mechanism behind its many beneficial effects on health including dumping syndrome, improved cholesterol and lipid metabolism and diabetes prevention and control. However, pectin also contains some unique abilities that may treat or prevent other diseases/disorders such as intestinal infections, atherosclerosis, cancer and obesity.
Several recent clinical studies, demonstrated that oral pectin supplementation to children and infants reduced acute intestinal infections and significantly slowed diarrhea. This is thought to be due to a reduction in pathogenic bacteria such as Shigella, Salmonella, Klebsiella, Enterobacter, Proteus and Citrobacter. This is supported by Olano–Martin et al. 68 who observed that pectin stimulated the growth of certain strains of Bifidobacteria and Lactobacillus in vitro. These bacteria are considered to be directly related to the health of the large intestine and their concentrations depict a healthy microflora population.
The quality of fibrin is thought to be an important risk factor for atherosclerosis, stroke and coronary heart disease. Pectin has been shown to increase fibrin permeability and decrease fibrin tensile strength in hyperlipidaemic men. Although the mechanism behind this is unknown, it is thought to be due in part to acetate production. Pectin yields predominantly acetate in the colon which is thought to enter peripheral circulation and alter fibrin architecture.
Pectin may also have a potential role in the complicated area of cancer prevention. Nangia–Makker et al. 69 found that pectin was able to bind to and decrease tumor growth and cancerous cell migration in rats fed modified citrus pectin. This is thought to be a result of pectin binding to galectin-3 and inhibiting some of its functions.
Bran
Bran is the outer most layer of a cereal grain and consists of the nucellar epidermis, seed coat, pericarp and aleurone. The aleurone consists of heavy walled, cube shaped cells which are composed primarily of cellulose. It is low in starch and high in minerals, protein, and fat. However, due to its thick cellulosic walls, these nutrients are virtually unavailable for digestion in monogastric species. The AACC defines oat bran as “the food which is produced by grinding clean oat groats or rolled oats and separating the resulting oat flour by sieving bolting, and/or other suitable means into fractions such that the oat bran fraction is not more than 50% of the original starting material and has a total betaglucan content of at least 5.5% (dry-weight basis) and a total dietary fiber content of at least 16.0% (dry-weight basis), and such that at least one-third of the total dietary fiber is soluble fiber.”
Bran from a wide array of cereal grains have been shown to have an effect on postprandial glucose levels, serum cholesterol, colon cancer, and body mass. Although the efficacy of bran may change due to its source, the purpose of this section will just evaluate bran’s general effect on the parameters listed above.
In a recent study of healthy adults, 31 g of rye bran decreased peak postprandial glucose levels by 35% when compared to the control 70. This effect may be due to the high arabinoxylan content in rye bran. Arabinoxylan, as discussed previously, may increase intestinal viscosity and slow nutrient absorption. In a more lengthy study, Qureshi et al. 71 found that subjects suffering type one and two diabetes decreased their fasting glucose levels due to the daily consumption of 10 g of stabilized rice bran over two months. The results may arise due to an increased intestinal viscosity, but is more likely a result of a decreased carbohydrate/caloric intake. Koh-Banerjee et al. 72, in a larger clinical study, supports this theory in their finding that for every 20 g/d increase in consumption of bran, body weight decreased by 0.80 lbs. It should be noted that this data remained significant even after adjustment for fat and protein intake, daily activity, caloric intake and baseline weight. In an earlier study, Zhang et al. 73 observed that adults with ileostomies, consuming bread rich in rye bran, significantly increased the ileal excretion of fat, nitrogen and energy. This study suggests bran did not delay nutrient absorption in the small intestine but hindered it.
In addition to a possible effect on carbohydrate absorption and metabolism, bran also seems to have the same effect on lipids. In a long term clinical study, Jensen et al. 74 reported that an increased daily consumption of bran significantly decreased the risk of coronary heart disease in healthy adult men. This is most likely due to the data reported by Qureshi et al. 71 who found that 10 g of rice bran consumed for eight weeks was able to decrease serum total cholesterol, LDL cholesterol and triglycerides. The mechanisms behind these effects may be two fold. The reduction in cholesterol levels is likely due to an increase in bile acid synthesis. Andersson et al. 75 found that oat bran doubled the serum concentration of 7α-hydroxy-4-cholesten-3-one (α-HC), which is a metabolite in the synthesis of bile acids that is oxidized from 7α-hydroxycholesterol. The reduction in serum triglyceride levels may be a result of a decreased absorption of fat from the small intestine.
Cellulose
Cellulose is a linear chain of β(1→4) linked glucose monomers and is the structural component of cell walls in green plants and vegetables. It is water insoluble and inert to digestive enzymes in the small intestine. However, it can go through microbial fermentation to a certain degree in the large intestine in turn producing short chain fatty acid.
Natural cellulose can be divided into two groups: Crystalline and amorphous. The crystalline component, which is made up of intra and intermolecular non covalent hydrogen bonds, make cellulose insoluble in water. However, many modified celluloses such as powdered cellulose, microcrystalline cellulose and hydroxypropylmethyl cellulose have been developed and are used as food ingredients. The difference between natural and modified celluloses is the extent of crystallization and hydrogen bonding. When these hydrogen bonds are disrupted and the crystallinity is lost, the cellulose derivative becomes water soluble 75.
Little research has been conducted evaluating the effects of cellulose in humans. Therefore, studies in other models such as the rat will be discussed. The translation to human relevance is poorly understood and debatable. Cellulose pills have been made available for human consumption with the theory that cellulose may decrease a person’s caloric intake. Although no human studies could be found to support this, several animal studies using cats, dogs and rats have shown that increasing dietary cellulose can reduce daily energy intake. This is most likely a dilution factor since cellulose is virtually undigested in the small intestine and only 51% metabolized by the microflora of the colon.
Many studies have evaluated the effect of cellulose on blood glucose and insulin levels in many different models. However, the data is extremely contradictory and may depend on the subject, type of cellulose and other unknown factors. Using the rat, dog and cat, natural cellulose was shown to decrease postprandial glucose and insulin levels. However, similar studies in pigs and humans 76 demonstrated that natural cellulose had no effect on these parameters. Studies using modified celluloses showed more consistent data. Microcrystalline cellulose has shown the ability to decrease blood glucose levels in the pig and rat. Complimenting this, methylcellulose had demonstrated the same effects in humans. Lightowler and Henry 77 found that adding only 1% high viscosity hydroxypropylmethylcellulose (HV-HPMC) to mashed potatoes decreased postprandial glucose levels by 37% in healthy adults. Also, Maki et al. 78 reported an acute 35% reduction in postprandial blood glucose due to 4 g of HV-HPMC in overweight subjects.
Modified cellulose has also been reported to effect lipid metabolism. Maki et al. 79, 80 both observed a significant reduction in total and LDL cholesterol in hypercholesterolemic adults consuming 5 g/d of HV-HPMC for four weeks. Interestingly, in subjects already receiving statin drugs, HV-HPMC was able to further reduce total and LDL cholesterol.
According to this, modified celluloses may be more beneficial than natural cellulose. These modified celluloses, as described above, act like soluble fiber thus adding to the viscosity of the GI tract. Therefore, it is assumed that increased intestinal viscosity delays nutrient absorption and increases bile acid excretion.
How dietary fiber is digested
It is now well-established that dietary fibre reaches the large intestine and is fermented by the colonic microflora with the production of short chain fatty acids, hydrogen, carbon dioxide and biomass. This fermentative process dominates human large bowel function and provides a means whereby energy is obtained from carbohydrates not digested in the small bowel, through absorption of short chain fatty acids 81.
Fermentation of fiber in the colon (large intestine)
Polysaccharides (a carbohydrate (e.g. starch, cellulose) whose molecules consist of a number of sugar molecules bonded together) cannot penetrate in the bacterial cells. They are first hydrolysed (broken down by chemical reaction with water) in monosaccharides (simple sugar molecule e.g. glucose), by membranous or extra-cellular enzymes secreted by bacteria. Metabolism of these monomeric sugars continue in the bacterial cells using the Embden-Meyerhoff pathway which leads to pyruvate. Pyruvate does not appear in the large bowel because it is immediately converted in end-products. These are short chain fatty acids, mainly acetate, propionate and butyrate, and gases: carbon dioxide (CO2), hydrogen (H2), and methane (CH4) 81.
Colonic fermentation is an efficient digestive process since starch is almost totally degraded, as well as lactose, alcohol-sugars and fructans if the intake of these sugars is not too high. More than half of the usually consumed fibres are degraded in the large intestine, the rest being excreted in the stool (see Table 2). A number of factors are likely to affect the utilization of fermentable carbohydrates in the colon. Among these is solubility. The more soluble substrates, being more accessible to hydrolytic enzymes, are likely to be degraded more rapidly. Nevertheless, some soluble fibres such as alginates or carragheenans are poorly fermented. Other factors involving digestive motility and individual differences in microflora could also modulate fermentation. Furthermore, certain metabolic pathways can be modified by the repeated occurrence of some sugars (lactose, lactulose, fructans) in the colon. The mechanisms and the physiological consequences of this adaptation are not completely identified.
Table 5. Colonic fermentability of dietary fibres in humans
Dietary fibre | Fermentability (%) |
Cellulose | 20-80 |
Hemicelluloses | 60-90 |
Pectins | 100 |
Guar gum | 100 |
Ispaghula | 55 |
Wheat bran | 50 |
Resistant starch | 100 |
Inulin, oligosaccharides | 100 (if they are not in excess) |
(Source 81).
Absorption and metabolism of dietary fiber end-products
Reducing the rate of digestion of carbohydrate spreads the absorption of carbohydrate along a longer portion of the small intestine and tends to increase the amount of carbohydrate which escapes digestion in the small intestine). For example, the amount of carbohydrate from lentils entering the colon is 2.5 times as great as carbohydrate from bread. Increasing the delivery of starch to the colon has many implications which include those on the health of the colon itself and on systemic metabolism. It is believed that starch entering the colon is completely and rapidly fermented, mostly in the cecum. The fermentation of starch produces relatively more butyrate than the fermentation of dietary fibre and resistant starch produces somewhat different fermentation products than readily digested starch.
A part of the products of fermentation are utilized by bacteria yielding energy and carbon necessary for synthesis and growth of the flora. Another part is eliminated in the stool or rectal gases, but the major part is absorbed by the colonic mucosa. Absorption of short chain fatty acids is rapid and leads to accumulation of bicarbonates and increase of pH in the lumen. Butyrate is considered to be the primary nutrient for the epithelial cells lining the colon and short chain fatty acids stimulate proliferation of colonic epithelial cells and growth of the colon in general. Butyrate is the preferred substrate of colonocytes. Short chain fatty acids which are not metabolized in the mucosa are oxidized in the liver, a part of acetate being also metabolised in the peripheric tissues.
Only a fraction of gases produced during fermentation is available for absorption. Hydrogen and methane are excreted in the breath gases. A large part of gases are consumed in the colonic lumen by ‘bacteria. Acetogenic bacteria produce acetate from CO2 and H2. Methanogenic bacteria produce CH4 by reduction of CO2 with H2. Finally, sulfate reducing bacteria utilise H2 to reduce sulfates and produce sulfites or hydrogen sulfide. Unused gases are excreted through the anus.
Effects of dietary fibre on gut microflora
The composition of microflora appears to be influenced to some degree by diet, age and geographic considerations, but these factors are not thought to be particularly significant, at least as far as the commonly studied bacterial groups are concerned. Recent studies have shown, however, that the ingestion of certain oligosaccharides, such as fructo-oligosaccharides, could modify bacterial composition of the dominant flora by increasing bifidobacteria. Some studies suggest that these bifidobacteria, which are saccharolytic bacteria naturally occurring in the normal colonic flora, might be beneficial to host health. At the present time, however, this has not been conclusively established.
Ingestion of fructo-oligosaccharides have increased faecal counts of endogenous bifidobacteria by a factor of 10, without changing the total anaerobes concentration. The similarity of effects of chemically different substrates is likely due to the capacity of bifidobacteria to hydrolyze all these substrates and to metabolize the produced monomeric sugars (glucose, galactose, fructose). The exact mechanisms whereby only some substrates could stimulate preferentially the growth of bifidobactria are not known. A recent in vitro study suggested that the polymerization degree could be more determinant than the chemical nature of oligosaccharides. The metabolic consequences of the changes in faecal flora composition are unknown. Ingestion of oligosaccharides had no effect on stool weight and pH.
Effects of dietary fibre on gut function
In the gastrointestinal tract, some fibres form a matrix with fibrous characteristics. That is, some fibres, because of their ability to swell within the aqueous medium, can trap water and nutrients, especially water-soluble ones such as sugars. The physical characteristics of the gastric and small intestinal contents are altered by fibre sources. The bulk or amount of material in the gastrointestinal tract is greater because fibre is not digestible and hence remains during the transit of digesta through the small intestine. The volume increase is due to the water-holding capacity of certain fibres. The viscosity of the intestinal contents increases due to the presence of fibre sources containing viscous polysaccharides.
The changes in the physical characteristics of the intestinal contents may influence gastric emptying, dilute enzymes and absorbable compounds in the gut, prevent starch from hydrolyzing, and slow the diffusion or mobility of enzymes, substrates and nutrients to the absorptive surface. These effects result in the slower appearance of nutrients such as glucose and some lipid molecules in the plasma following a meal.
The effects of purified dietary fibres on bowel function may or may not be similar to those of intact fibres in whole foods. This is presumably due, at least in part, to interactions between fibre and starch, and the presence of fibre associated substances such as phytate and lectins which are present in the whole food. This makes it very difficult to make valid generalizations about the physiologic effects of fibre based simply on fibre analysis. For example, when considering the effect of fibre on postprandial blood glucose responses, purified viscous fibres have been found to produce a significant reduction in glycemic response in 33 of 50 studies (66%) reviewed in 1992, compared to only 3 of 14 (21%) studies with insoluble fibre (166). The effects of purified fibres appear to be directly related to their viscosity. This would suggest that the blood glucose responses of foods should be more closely related to their soluble than insoluble fibre content, however the opposite is the case. For 52 foods, the food glycemic index (as the indicator of rise in blood glucose) was weakly related to the amount of total fibre per 50g carbohydrate, and insoluble fibre explained a larger proportion of the variance in glycemic index, 17%, than soluble fibre, 9%.
Effects on carbohydrate digestion and absorption
- Gastric emptying
Dietary fibres may affect gastric emptying in several ways. First, they may slow gastric filling, due to their bulking and energetic dilution capacity, which might in turn slow gastric emptying. Secondly, when certain soluble fibres are mixed in liquid meals or in liquid/solid meals, they delay emptying of gastric liquids by increasing viscosity of gastric contents. Such an increase in the viscosity of chyma could also slow the gastric emptying of solid components of the meal. On this issue, results are very controversial. Moreover, by acting as an emulsifier, viscous fibre can stabilize the gastric chyma and prevent separation of the solid from the liquid phase, impairing selective retention of the largest particles, and thereby increasing their rate of passage into the small intestine. Besides the effects of soluble fibres, insoluble fractions may also alter gastric emptying by mechanisms depending on their water retention capacity or size of particles.
- Enzyme-substrate interaction
Available evidence suggests that fibre has little, if any, direct acute effect on the secretory function of the exocrine pancreas suggesting that the primary effect of fibre on carbohydrate digestion is exerted in the intestinal lumen. In the lumen, enzymes and substrates may be diluted with the addition of non-digestible material. Evidence from in vitro studies and from duodenal aspirates suggest that most of the tested fibres can alter the activity of pancreatic amylase (88). The inhibitory effects of fibre on pancreatic enzyme activities have been attributed to various factors including pH changes, ion-exchange properties, enzyme inhibitors and adsorption. Rather than a chemical enzyme-fibre interaction, the presence of fibre, through its particulate or viscous nature, probably impedes enzyme-substrate interaction.
The presence of fibre in a form that restricts starch gelatinization or access of the hydrolytic enzymes to starch can slow the rate of digestion of the starch. For instance, the slow rate of digestion of legumes may be related to the entrapment of starch in fibrous thick-walled cells, which prevents its complete swelling during cooking. In addition, resistance of starch to pancreatic hydrolysis may result from the presence of intact cell walls, which survive processing and cooking and insulate starch in such a manner that portions of it cannot be digested or absorbed.
- Small intestinal motility
There is evidence that viscous fibres can influence accessibility of available carbohydrates to the mucosal surface and slow their absorption. One of the major mechanisms of this action is related to the effects of dietary fibre on small intestinal motility. Small intestinal contractions create turbulences and convective currents which cause fluid circulation and mixing of luminal contents. These movements allow glucose to be brought from the centre of the lumen close to the epithelium. When it reaches proximity to the epithelium, glucose must then diffuse across the unstirred water layer. This layer is created by a gradient of progressively poorer stirring as the mucosa is approached and forms an aqueous diffusion barrier separating mixed bulk luminal contents from the brush border. Thickness of the unstirred water layer depends on small intestinal contractions and is inversely related to the magnitude of the stir rate. When there is no contraction, fluid moves through the small intestine with laminar flow comparable to that occurring in a pipe. In this flow, there is no movement in the radial direction (from the centre of the lumen toward the epithelium), and consequently the stirring is very poor and the unstirred water layer very thick. On the contrary, normal motility generates both longitudinal and radial convection currents, hence creating turbulences and stirring of luminal fluid. Beside the effects of mixing contractions on glucose movement, small intestinal motility may alter absorption by influencing transit rate which determines area and time of contact between glucose and the epithelium.
Dietary fibres which alter small intestinal motility could thus influence glucose absorption by this mechanism. Viscous fibres, such as guar gum, stimulate motility but decrease transit rate, because they resist propulsive contractions. However, though guar gum slows transit it does not affect the distribution of glucose in the human upper small intestine. It is thus unlikely that guar gum delays glucose absorption by reducing contact area. As they resist propulsion, viscous fibres should similarly resist mixing contractions, hence inhibiting the effects of motility on fluid stirring. This is probably the mechanism by which they increase thickness of the unstirred water layer, and diminish passage of glucose across the epithelium.
- Effects of dietary fibre on large bowel function
The major effects of dietary fibre occur in the colon. Here each type of dietary fibre interacts with the microflora, and the colonic mucosa and muscle to produce several possible effects. The actions of an individual fibre source depends to a large extent on its fermentability. The range of fermentability of different fibre is great and difficult to predict. Dietary fibre, however, can be roughly divided into those which are rapidly fermented, such as oligosaccharides, those which are more slowly fermented, such as gums, and those which are hardly fermented at all, such as wheat bran. The least fermentable fibres are the most likely to increase stool output. Dietary fibre which is highly fermentable is unlikely to have much effect on stool output but will affect bacterial fermentation products in the proximal colon and hence colonic and systemic physiology. Fibres which are slowly fermented may have a major influence in the distal colon even if they do not increase stool output significantly. Furthermore, the effect of each type of fibre is determined by dose.
- Stool output
The dietary fibres which have the greatest effects on stool output are in general the least fermentable. These fibres probably act by virtue of their water holding capacity. The relationship between water holding capacity and stool output is not simple. Dietary fibres with high water holding capacity are those which are the most fermentable and are lost before they reach the rectum. There are exceptions such as ispaghula which has high water holding capacity but resists fermentation. Moreover, one of the most reliable stool bulkers is wheat bran which has a water holding capacity that is as low as the rest of faecal contents on a normal low fibre diet. It appears that the most important factor for a large effect on stool output is simply for the fibre to appear in stool. The effect is then dependent on the amount of fibre present as well as its residual water holding capacity. The contribution of bacterial cells to faecal mass should not be forgotten, as the water content of bacteria is high. The effects of fibre are not restricted to increasing output. Dietary fibre has also a role in changing the consistency of the stool by increasing the water content and the plasticity, and increasing stool frequency.
- Colonic motility and transit time
Certain fibres are known to have a laxative effect, in that their presence in the colon affects its motility and modifies colonic transit time. Two major mechanisms to explain this effect depend on the physicochemical properties and fermentative fate of fibre. These mechanisms refer to stimulation by the bulking effect of fibre as well as changes in the contractile activity and secretion of the colon.
Increasing the volume of colonic contents distends the colon wall and stimulates propulsion of digesta through the activation of intramuscular mechanoreceptors. Dietary fibre can increase the faecal bulk by several mechanisms. First, the volume occupied by undegraded fibres adds to the volume of the rest of contents. This explains why the least fermentable fibres, such as wheat and corn bran, ispaghula or some algal polysaccharides, are particularly efficient laxatives. Also, these residues can trap water within their matrix, thus leading to a greater bulk. A third possible mechanism to increase intraluminal volume and stretch colonic muscle is the production of gases occurring during the fermentation of fibre.
In addition to their bulking effects, dietary fibre can reduce transit time by modulating contractile activity and water movements in the colon. Here again, they can act in several ways. First, the edges of solid particles can stimulate mechanoreceptors located in the submucosa and by that, modify the contractile pattern of the colon in favour of a greater propulsion of digesta, as has been shown with plastic particles. Fibre could also release compounds trapped in the small intestine (such as biliary salts or fatty acids) into the colon during fermentation. Such compounds have been shown to stimulate secretion and rectosigmoid motility.
Finally, a large part of fibre is fermented by microflora yielding several metabolites which can themselves influence colonic motility. For instance, short chain fatty acids stimulate contractions in the terminal ileum of humans and may also affect colonic motility as has been demonstrated with rats.
It has recently been appreciated that dietary starch bulks the stool, presumably because undigested starch provides energy for colonic bacterial growth. Thus, some of the faecal bulking effect of dietary fibre, at least in intact foods, could be due to the associated increase in starch delivery to the colon.
Types of Dietary Fiber
Recent research suggests that fiber should be categorized by its physical characteristics; how well it dissolves (solubility), how thick it is (viscosity) and how well it breaks down (fermentability). Some commonly known terms are described below:
- Soluble fiber including pectins and beta glucans is found in foods like oats, peas, beans, apples, citrus fruits, carrots, barley and psyllium. Soluble fiber dissolves in water to form a gel-like material. It can help lower blood cholesterol and glucose levels. Foods high in soluble fiber can help you feel full. They also help reduce constipation by speeding up the time it takes for feces (poop) to pass through your body. Soluble dietary fiber has been associated with lower postprandial glucose levels and increased insulin sensitivity in diabetic and healthy subjects; these effects were generally attributed to the viscous and/or gelling properties of soluble fiber 82. Soluble dietary fiber exerts physiological effects on the stomach and small intestine that modulate postprandial glycemic responses, including delaying gastric emptying 10, which accounts for ~35% of the variance in peak glucose concentrations following the ingestion of oral glucose 83, modulating gastrointestinal myoelectrical activity and delaying small bowel transit 84, 85, reducing glucose diffusion through the unstirred water layer 86, and reducing the accessibility of α-amylase to its substrates due to the increased viscosity of gut contents 87. Notably, the increased viscosity and gel-forming properties of soluble fiber are predominantly responsible for its glycemic effect, since the hypoglycemic effect can be reversed by the hydrolysis of guar gum or following ultra-high heating and homogenization 10. In addition, the intestinal absorption of carbohydrates was prolonged by soluble dietary fiber, which was partially due to altered incretin levels, including increased glucagon-like peptide 1 levels 87. In experimental clamp studies, soluble dietary fiber also influenced peripheral glucose uptake mechanisms 88, 89, including increasing skeletal muscle expression of the insulin-responsive glucose transporter type 4 (GLUT-4), which enhances skeletal muscle uptake, augments insulin sensitivity and normalizes blood glucose 89. In humans, various fatty acids stimulate the expression of peroxisome proliferator-activated receptor-γ, which increases adipocyte GLUT-4 levels 90. A more recent study to find out the health benefits of soluble fiber on type 2 diabetes 91. A total of 117 patients with type 2 diabetes between the ages of 40 and 70 were assessed. Patients were randomly assigned to one of two groups, and administered extra soluble dietary fiber (10 or 20 g/day), or to a control group (0 g/day) for one month. The 20 g/day soluble dietary fiber group exhibited significantly improved fasting blood glucose and low-density (LDL) lipoprotein “bad cholesterol” levels, as well as a significantly improved insulin resistance index. In addition, 10 and 20 g/day soluble dietary fiber significantly improved the waist and hip circumferences and levels of triglycerides and apolipoprotein A. The results of the present study suggested that increased and regular consumption of soluble dietary fiber led to significant improvements in blood glucose levels, insulin resistance and metabolic profiles 91.
- Insoluble fiber including cellulose is found in wheat bran and nuts. Insoluble fiber is a type of fibre that doesn’t dissolve in water. Because insoluble fiber absorbs water, it helps to soften the contents of your bowel, contributing to keep the bowels regular. Insoluble fiber promotes the movement of material through your digestive system and increases stool bulk, so it can be of benefit to those who struggle with constipation or irregular stools. Whole-wheat flour, wheat bran, nuts, beans and vegetables, such as cauliflower, green beans and potatoes, are good sources of insoluble fiber. Diverticulitis, an inflammation of the intestine, is one of the most common age-related disorders of the colon in Western society. Among male health professionals in a long-term follow-up study, eating dietary fiber, particularly insoluble fiber, was associated with about a 40 percent lower risk of diverticular disease 92.
- Resistant starch is a soluble fiber that is highly fermentable in the gut. While most starch is digested in the upper part of the gut, resistant starch resists digestion in the small intestine and so goes all the way to the large intestine 93. Once in the large intestine, good bacteria ferment resistant starch. This process produces short chain fatty acids (SCFAs) and gasses that help to keep the lining of the bowel healthy. Resistant starch is starch that is not easily absorbed. Different ways of cooking can create different amounts of resistant starch. For example, resistant starch is found in slightly undercooked (‘al dente’) pasta, cooked but cooled potatoes (including potato salad), cooked and cooled grains like rice, quinoa, barley and buckwheat, under-ripe bananas, beans, lentils and a product called Hi-maize used in some breads and breakfast cereals. Freekeh, a Middle Eastern grain available in some supermarkets, is another good source. In general, foods that are less highly processed contain more resistant starch. An important benefit of resistant starch is that it ferments, which produces substances that help to keep the lining of the bowel healthy. Resistant starch has been classified into five basic “types” 94.
- Type 1 (resistant starch 1) is made up of starch granules surrounded by an indigestible plant matrix.
- Type 2 (resistant starch 2) occurs in its natural form such as in an uncooked potato and high amylose maize.
- Type 3 (resistant starch 3) are crystallized starches made by unique cooking and cooling processes.
- Type 4 (resistant starch 4) is a starch chemically modified by esterification, crosslinking, or transglycosylation and is not found in nature.
- Type 5 (resistant starch 5) is a starch consisting of amylose-lipid complex.
- Prebiotics are types of carbohydrate that only our gut bacteria can feed upon. Some examples are onions, garlic, asparagus and banana.
Table 6. Types of resistant starches
| Designation | Description | Example | Reference |
| Type 1 (resistant starch 1) | Physically inaccessible starch | Coarsely ground or whole-kernel grains | 95 |
| Type 2 (resistant starch 2) | Granular starch with the B- or C-polymorph | High-amylose maize starch, raw potato, raw banana starch | 95 |
| Type 3 (resistant starch 3) | Retrograded starch | Cooked and cooled starchy foods | 96 |
| Type 4 (resistant starch 4) | Chemically modified starches | Cross-linked starch and octenyl succinate starch | 97 |
| Type 5 (resistant starch 5) | Amylose-lipid complex | Stearic acid-complexed high-amylose starch | 98 |
Footnote: 1RSI, type I resistant starch; (RS); RSII, type II resistant starch; RSIII, type III resistant starch; RSIV, type IV resistant starch; RSV; type V resistant starch.
[Source 94 ]Few studies have compared types, but one recent study by Haub et al. 99 reported that cross-linked resistant starch 4 elicited a greater glucose lowering effect than the more commonly tested resistant starch 2.
A majority of human studies involving resistant starch have shown a decrease in postprandial blood glucose and insulin levels. However, it is difficult to completely understand these effects due to differences in study design and the type of resistant starch used. Behall et al. 100 found that women consuming 0.71 g, 2.57 g or 5.06 g of resistant starch had significantly lower postprandial glucose and insulin levels when compared to the control. However, this study failed to maintain an equal amount of available carbohydrate between the treatments and control. Therefore, it is difficult to determine whether the attenuation of glucose and insulin was due to the resistant starch or the fact that there was less available carbohydrate in the meal. Similarly, Reader et al. 101 reported that 7.25 g of resistant starch added to an energy bar decreased blood glucose and insulin levels in healthy adults. But, ingredients, amount of ingredients and nutrient levels were different for each treatment. A recent study by Al-Tamimi et al. 102 on Glucose and Insulin Responses in Humans. Al-Tamimi EK, Seib PA, Snyder BS, Haub MD. J Nutr Metab. 2010; 2010. https://www.ncbi.nlm.nih.gov/pubmed/20798767/)), however removed these variables by controlling for non starch ingredients and available carbohydrates. It was reported that postprandial blood glucose and insulin levels were significantly reduced with the supplementation of 30 g of resistant starch 4.
Several studies report that longer term consumption of a resistant starch may decrease fasting cholesterol and triglyceride levels. In a five week study, Behall et al. 103 found that men consuming 34% of their energy from high amylose maize, when compared to a high amylopectin carbohydrate, had significantly reduced fasting cholesterol and triglyceride levels. Resier et al. 104 reported similar results in an isocaloric and isonutrient diet with either high amylose maize or fructose. Porikos and Van Itallie 105 suggest that an interaction exists between sucrose, and therefore most likely fructose, and saturated fatty acids in turn promoting serum triglyceride levels. Interestingly, the relationship does not seem to exist for polyunsaturated fatty acids. The likely mechanism behind the ability of resistant starch to decrease cholesterol levels is an increased intestinal viscosity. However, some studies, such as Jenkins et al. 106, report conflicting data as resistant starch 2 and resistant starch 3 had no effect on serum lipid profiles. While using the same type of resistant starch, subjects were only tested for two weeks. It may be that the resistant starch requires a longer period of time to promote an effect.
Research has also been conducted which evaluates the effect of resistant starch on fat oxidation and storage. However, data between studies are contradictory with no clear conclusions. Tagliabue et al. 107 reported that resistant starch 2, obtained from raw potatoes, was able to increase fat oxidation 5 h postprandial. However, the test diet, consisting of the resistant starch 2, had significantly less gross and metabolizable energy. Therefore, it is difficult to determine if the increased fat oxidation was due to the resistant starch 2 or a decreased caloric intake. A 10 week study by Howe et al. 108 may suggest the latter. High amylose starch, compared to a high amylopectin, produced no change on fat oxidation when an isocaloric diet was consumed. Conversely, Robertson et al. 109 reported that 30 g of resistant starch 2 added to healthy subjects habitual diet resulted in a significant decrease in subcutaneous abdominal adipose tissue non-esterfied fatty acid and glycerol release. This may be a result of increased peripheral short chain fatty acid metabolism or ghrelin secretions.
How much fiber do you need?
Depending on your age and sex, adults should get 25 to 31 grams of fiber a day 2. Older adults sometimes don’t get enough fiber because they may lose interest in food.
- Men over the age of 50 should get at least 38 grams of fiber per day.
- Women over the age of 50 should get 25 grams per day.
- Children ages 1 to 3 should get 19 grams of fiber per day.
- Children between 4 and 8 years old should get 25 grams per day.
- Girls between 9 and 18 should get 26 grams of fiber each day. Boys of the same age range should get between 31 and 38 grams of fiber per day.
The recommendations for fibre intake for adults for most European countries and for countries like Australia, New Zealand and the USA are in the order of 30–35 g/day for men and 25–32 g/day for women. Overall average intakes do not reach this level of intake for any country.
For children, recommendations vary quite markedly from country to country; for example, for those aged 10–12 years, France recommends 5+age, equivalent to 15–17 g/d, for Poland, 19 g/d, for Australia and New Zealand, 20 g/d for girls and 24 g/d for boys (for 9–13 years) and for the USA, 26 g/d for girls and 31 g/d for boys (for 9–13 years). Other countries have no official recommendation for children. Hence it is difficult to say if recommendations are being met overall, although for most countries, intakes are lower than the recommendation, with few reaching an average intake of 20 g/d for boys or 18 g/d for girls. For teenagers, recommendations are similar or slightly higher than for younger children.
Most Americans eat less than this. Getting sufficient fibre isn’t just about adding unprocessed wheat bran to breakfast cereal – it’s important to include different types of fibre from a variety of plant foods.
Sources of Dietary Fiber
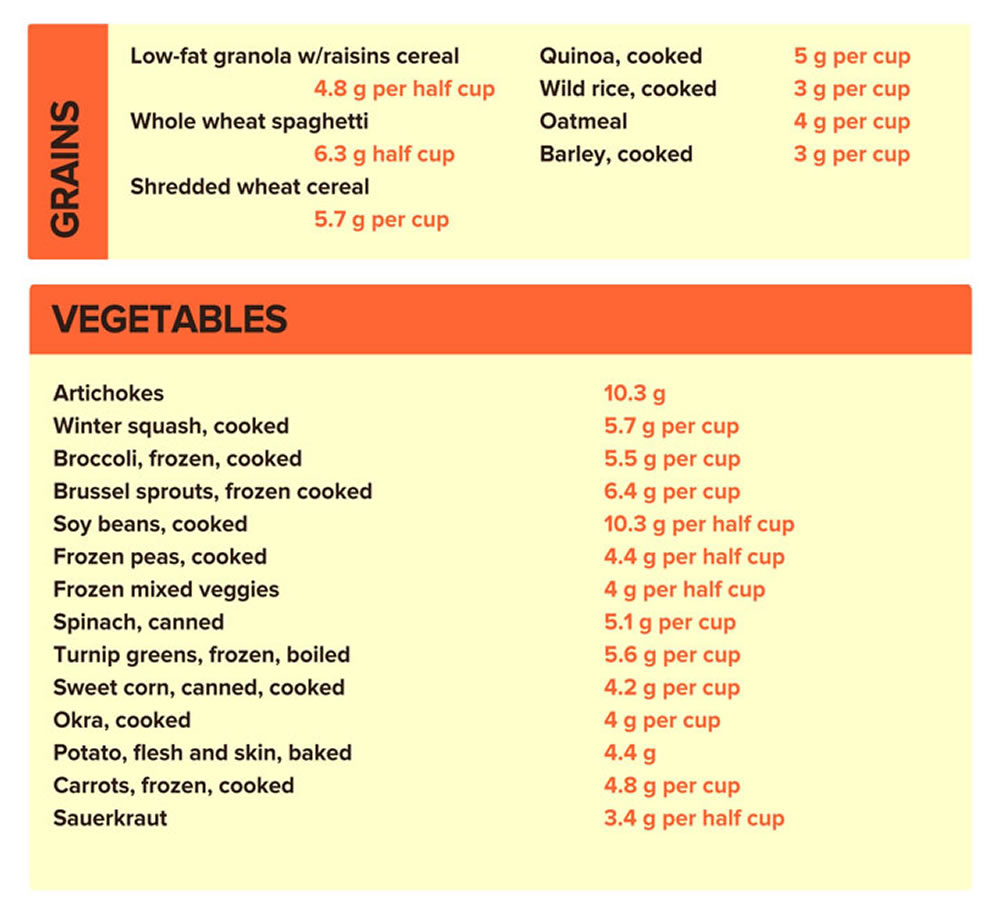
The amount of soluble and insoluble fiber varies in different plant foods. To receive the greatest health benefit, eat a wide variety of high-fiber foods.
Good sources of dietary fiber include:
- Pulses (like lentils and peas) and beans and legumes (think navy beans, small white beans, split peas, chickpeas, lentils, pinto beans)
- Fruits and vegetables, vegetables such as carrots, broccoli, green peas, and collard greens; fruits especially those with edible skin (like pears and apples with the skin on) and those with edible seeds (like berries)
- Nuts—try different kinds (pumpkin seeds, almonds, sunflower seeds, pistachios and peanuts are a good source of fiber and healthy fats, but be mindful of portion sizes, because they also contain a lot of calories in a small amount!)
- Whole grains such as:
- Quinoa, barley, bulgur, oats, brown rice and farro
- Whole wheat pasta
- Whole grain cereals, including those made from whole wheat, wheat bran and oats
Choose fiber rich foods from a variety of sources including wholegrains, fruit and vegetable, nuts and seeds, beans and pulses. When you read food labels check for the grams of fiber per serving or per 100g. Foods that are naturally high in fiber and contain at least 3 grams per 100 gram are often labeled as a “good source,” and foods labeled as “excellent source” contain more than 5 grams of fiber per serving.
There are relatively few publications which provide detail on the types of dietary fibre consumed. In adults, grain products were the largest source of fibre in all countries, providing from 32–33 % of fibre intake in the USA and Spain to 48–49 % in Ireland, the Netherlands and Sweden. Some countries provided greater breakdown of grain sources, and, in these, bread was the major source, ranging from 11 to 30 % of total fibre, with much smaller contributions from breakfast cereals, from 5 to 8 %, biscuits and pastries, from 3 to 11 %, and pasta from 1 to 4 %. Vegetables, potatoes and fruit were the next highest sources, but these varied considerably, vegetables contributing from 12 to 21 % of fibre intake, potatoes from 6 to 19 % and fruit from 8 to 23 %.
In children, a report of the National Health and Nutrition Examination Survey (NHANES) for 2003–2006 indicated that grains provided 44 % of fibre intake, while another of 2009–2010 found 33 %.
To get enough fibre every day, the U.S. Department of Health and Human Services 19 recommends that an individual eats:
- at least 4 serves of wholegrain or wholemeal foods every day (or ensure about half of your daily serves of breads and cereals are wholegrain or wholemeal varieties)
- at least 2 serves of fruit daily
- 5 serves of vegetables daily including legumes (also known as ‘pulses’)
- wholefoods rather than dietary fibre supplements as the benefits of fibre from food may be from the combination of nutrients in food working together.
Table 7. Below is an example of how an adult may meet their daily dietary fibre requirements:
| Food | Fibre Content |
| 3/4 cup whole grain breakfast cereal | 4.5g |
| 2 slices wholemeal bread | 4.5g |
| 1 apple (with skin) and 1 orange | 5.5g |
| 2 cups mixed raw vegetables | 10g |
| 1/4 cup legumes eg. baked beans | 3g |
| Total | 27.5g |
Note: Daily recommended fiber intake are highlighted in pink. (Source: U.S. Department of Health and Human Services and U.S. Department of Agriculture: Dietary Guidelines for Americans 2015-2020 19).
Tips for including more fibre in your diet
- Know which packaged foods are high in fibre by reading the nutrient panel on the pack. A food with at least 4g fibre per serve is a good source; food with at least 7g fibre per serve is an excellent source.
- Enjoy wholegrain, wholemeal or mixed grain toast instead of white.
- Use wholegrain pasta instead of white pasta.
- Try brown rice or quinoa instead of white rice with casseroles or curries.
- Use wholemeal flour to thicken sauces, gravies and stews.
- Try wholegrain or wholemeal crisp breads with toppings such as creamed corn or salsa dip.
- For breakfast choose a high-fiber breakfast cereal — 5 or more grams of fiber a serving. Opt for cereals with “whole grain,” “bran” or “fiber” in the name. Or add a few tablespoons of unprocessed wheat bran to your favorite cereal.
- Switch to whole grains. Consume at least half of all grains as whole grains. Look for breads that list whole wheat, whole-wheat flour or another whole grain as the first ingredient on the label and have at least 2 grams of dietary fiber a serving. Experiment with brown rice, wild rice, barley, whole-wheat pasta and bulgur wheat.
- Bulk up baked goods. Substitute whole-grain flour for half or all of the white flour when baking. Try adding crushed bran cereal, unprocessed wheat bran or uncooked oatmeal to muffins, cakes and cookies.
- Lean on legumes. Beans, peas and lentils are excellent sources of fiber. Add kidney beans to canned soup or a green salad. Or make nachos with refried black beans, lots of fresh veggies, whole-wheat tortilla chips and salsa.
- Eat more fruit and vegetables. Fruits and vegetables are rich in fiber, as well as vitamins and minerals. Try to eat five or more servings daily.
- Make snacks count. Fresh fruits, raw vegetables, low-fat popcorn and whole-grain crackers are all good choices. A handful of nuts or dried fruits also is a healthy, high-fiber snack — although be aware that nuts and dried fruits are high in calories.
Keep in mind that if you haven’t been eating a lot of foods high in fiber on a daily basis, it’s important to increase your intake slowly to allow your body to adjust. A sudden increase in eating foods high in fiber (especially foods with added fiber or when using supplements) can cause gas, bloating or constipation. Be sure you are drinking enough water too, because fiber needs water to move through your body, making your stool soft and bulky.
Eat more fiber. You’ve probably heard it before. But do you know why fiber is so good for your health ?
Dietary Fiber and Health Benefits
The Food and Drug Administration (FDA) has approved two health claims for dietary fiber. The first claim states that, along with a decreased consumption of fats (<30% of calories), an increased consumption of dietary fiber from fruits, vegetables and whole grains may reduce some types of cancer 23. “Increased consumption” is defined as six or more one ounce equivalents, with three ounces (85 grams) derived from whole grains. A one ounce equivalent (~ 28.35 grams) would be consistent with one slice of bread, ½ cup oatmeal or rice, or five to seven crackers.
Recent survey data indicate that more than 50% of all adult Americans are overweight or obese. In parallel with this epidemic of weight gain in the general population, the incidence rate of type 2 diabetes mellitus is rapidly rising. Recent epidemiological data indicate that diets rich in high-fiber whole grains are associated with lower risk of coronary heart disease and type 2 diabetes. These data are consistent with results from recent metabolic experiments, suggesting favorable lipid profiles and glycemic control associated with higher intake of whole grains, but not with refined grains 111.
High intake of dietary fiber has been linked to a lower risk of heart disease in a number of large studies that followed people for many years 112. In a Harvard study of over 40,000 male health professionals, researchers found that a high total dietary fiber intake was linked to a 40 percent lower risk of coronary heart disease 113. A related Harvard study of female nurses produced quite similar findings 114.
Higher fiber intake has also been linked to a lower risk of metabolic syndrome, a combination of factors that increases the risk of developing heart disease and diabetes. These factors include high blood pressure, high insulin levels, excess weight (especially around the abdomen), high levels of triglycerides, and low levels of HDL (good) cholesterol. Several studies suggest that higher intake of fiber may offer protective benefits from this syndrome 115, 116.
Dietary fiber is widely prescribed 117, either alone or in combination with lipid-lowering therapies, to reduce cholesterol levels 118. The exact mechanism by which soluble fiber lowers serum levels of low-density lipoprotein (LDL) and cholesterol is not completely understood; however, it has been suggested that soluble fiber may interfere with lipid and/or bile acid metabolism 119. A reasonable increase in dietary fiber intake (20–35 g/day) is recommended by the American Diabetes Association based on the effects of soluble fiber on plasma levels of cholesterol 120. Recent epidemiological findings have suggested that there is an association between high dietary fiber intake and a reduced risk of developing diabetes and coronary heart disease 121, 122. In particular, soluble dietary fiber has been shown to reduce insulin resistance in female non-diabetic patients 123.
In a prospective cohort study involving 74,091 US female nurses, who were free of known cardiovascular disease, cancer, and diabetes at baseline, aged 38-63 y in 1984 and were followed from 1984 to 1996; their dietary habits were assessed in 1984, 1986, 1990, and 1994 124. Women who consumed more whole grains consistently weighed less than did women who consumed less whole grains. Over 12 yrs, those with the greatest increase in intake of dietary fiber gained an average of 1.52 kg less than did those with the smallest increase in intake of dietary fiber independent of body weight at baseline and age. Women in the highest quintile of dietary fiber intake had a 49% lower risk of major weight gain than did women in the highest quintile. The researchers concluded that weight gain was inversely associated with the intake of high-fiber, whole-grain foods but positively related to the intake of refined-grain foods, which indicated the importance of distinguishing whole-grain products from refined-grain products to aid in weight control 124. This is because refined-grain products have a higher starch content but a lower fiber content (ie, greater energy density) than do whole grains. Concentrations of vitamins, minerals, essential fatty acids, and phytochemicals that are important in carbohydrate metabolism are also lower in refined grains 111.
- Serving Size
The following count as 1 ounce-equivalent (or 1 serving) of whole grains:
- 1 slice whole-grain bread (such as 100% whole-wheat bread)
- 1 cup ready-to-eat, whole-grain cereal
- 1⁄2 cup cooked whole-grain cereal, brown rice, or whole-wheat pasta
- 5 whole-grain crackers
- 3 cups unsalted, air-popped popcorn
- 1 6-inch whole-wheat tortilla
The second FDA claim supporting health benefits of dietary fiber states that diets low in saturated fat (<10% of calories) and cholesterol and high in fruits, vegetables and whole grain, have a decreased risk of leading to coronary heart disease 24. For most, an increased consumption of dietary fiber is considered to be approximately 25 to 35 g/d, of which 6 g are soluble fiber.
- Epidemiological and clinical studies demonstrate that intake of dietary fiber and whole grain is inversely related to obesity 26, type 2 diabetes 125, cancer 126 and cardiovascular disease 127.
- A study published in the Annals of Internal Medicine suggests that something as simple as aiming to eat 30 grams of fiber each day can help you lose weight, lower your blood pressure, and improve your body’s response to insulin just as effectively as a more complicated diet.
Dietary Fiber and Weight Loss
Although there are multiple factors that could contribute to obesity, the primary cause is due to an increase in the energy intake over energy expenditure (Calorie IN/Calorie OUT ratio). Therefore, limiting energy absorption is critical when treating obesity. Scientists have taken this a step further and studied the effect of other dietary aspects that may serve in weight regulation, including dietary fiber. Increasing dietary fiber consumption may decrease energy absorption by way of diluting a diet’s energy availability while maintaining other important nutrients 40.
Substantial research has been conducted to evaluate the effect of dietary fiber and body weight, most all of which show an inverse relationship between dietary fiber intake and change in body weight. Tucker and Thomas 128 supported this statement in a study consisting of 252 middle aged women. They observed that over a 20 month period participants lost an average of 4.4 lbs due to an 8 g increase in dietary fiber per 1000 kcal. This weight loss was primarily due to decreased body fat. It should be recognized that the correlation between dietary fiber and weight change was independent of many other potential factors including age, baseline fiber and fat intakes, activity level, and baseline energy intake 128.
Koh-Banerjee et al. concur with the above findings and also suggest a dose-response relationship. They reported that for every 40 g/d increase in whole grain intake, weight gain decreased by 1.1 lbs. Moreover, bran seemed to play an important role in the reduction of weight gain by 0.8 lbs per 20 g/d intake.
Dietary fiber’s ability to decrease body weight or attenuate weight gain could be contributed to several factors. First, soluble fiber, when fermented in the large intestine, produces glucagon-like peptide (GLP-1) and peptide YY (PYY) 128. These two gut hormones play a role inducing satiety. Second, dietary fiber may significantly decrease energy intake 128. Women who consumed increased levels of fiber tended to also have a decreased consumption of dietary fat. Third, dietary fiber may decrease a diets metabolizable energy, which is gross energy minus the energy lost in the feces, urine and combustible gases. Baer et al. 129 observed that an increased consumption of dietary fiber resulted in a decrease in the metabolizable energy of the diet. This may be attributed to the fact that fat digestibility decreased as dietary fiber increased. Also, as dietary fiber intake increases, the intake of simple carbohydrates tends to decrease. Although, dietary fiber still contributes to the total caloric content of a diet, it is much more resistant to digestion by the small intestine and even somewhat resistant in the large intestine.
It should also be noted that the inverse relationship between dietary fiber and metabolizable energy was independent of dietary fat. Therefore, metabolizable energy decreased as dietary fiber increased in both high and low fat diets. However, when dietary fiber was split into soluble and insoluble fiber, the results were much more inconclusive. Soluble fiber decreased metabolizable energy when added to a low fat diet but increased metabolizable energy when added to a high fat diet 129. It is not really known how dietary fat changes the effects of soluble fiber. Isken et al. 130 showed supportive data in mice consuming a high fat diet. Mice showed an increased weight gain when soluble fiber was added to a high fat diet. There are several mechanisms that may explain how soluble fiber could increase metabolizable energy or weight gain. First, bacterial populations in the large intestine increase due to an increase in soluble fiber consumption 58. This could result in increased fermentation and utilization of short chain fatty acids thereby increasing energy absorption. Second, soluble fiber enlarges in the gastrointestinal tract and forms a viscous material which delays intestinal transit time 131. Subsequently, this increase time in the gastrointestinal tract may allow for more complete digestion and absorption. Conversely, some believe this increase viscosity has an opposite effect and retards absorption 132. More research is needed in this area.
Insoluble fiber seems to have the opposite effect to that of soluble. When insoluble fiber intake was increased in mice consuming a high fat diet, body weight decreased 130. Research in sows demonstrated that insoluble fiber decreased energy digestibility while it increased with soluble fiber intake 133. The mode of action behind these findings may be due to the fact that insoluble fiber causes an increased rate of passage through the gastrointestinal tract 134. This would be expected to result in diminished digestion and absorption of nutrients.
According to the data presented above, both soluble and insoluble fiber may lead to weight loss. However, there seems to be a relationship between the type of diet (high or low fat) and the type of fiber consumed. Insoluble fiber may play a more important role for weight loss during consumption of a high fat diet. Since resistant starch is a constituent of dietary fiber and undergoes the same digestion as insoluble fiber, comparing resistant starch and insoluble fiber may give us a better understanding of how dietary fiber can be used to treat and prevent obesity. Adding resistant starch to a diet dilutes its metabolizable energy, but not to the degree of insoluble fiber 135.
Numerous studies 72, 136 have found the same inverse relationship between dietary fiber and weight gain. However, the data are more inconsistent when comparing soluble and insoluble. Thus, although increasing dietary fiber in general has a favorable effect on body weight, more research is warranted to determine the optimal dietary fibers for the purpose of weight management.
Dietary Fiber and Cholesterol
In 1999, Brown et al. 137 undertook a meta-analysis of sixty-seven trials that demonstrated a reduction of total cholesterol by 0·047 mmol/l, and LDL-cholesterol by 0·057 mmol/l with daily consumption of fibre isolates or fibre-enriched products containing 2–10 g of soluble fibre provided by pectin, guar gum, psyllium and oat bran. Similar findings, albeit using narrower trials inclusion criteria, were reported in the evidence reviews prepared for the Scientific Advisory Committee on Nutrition Carbohydrate Working Group 138. Total and LDL-cholesterol were lowered by supplementation with mixed, soluble types of dietary fibre, and total and LDL-cholesterol and fasting TAG levels were significantly lowered by supplementation with oats, oat bran or β-glucan-supplemented diets. Other types of dietary fibre supplementation did not consistently reduce blood lipids in these trials of normolipidaemic individuals. Some of this evidence forms the basis for the authorised health claims that certain fibre types, including β-glucans from oats and barley, pectin, guar gum and chitosan, may contribute to the maintenance of normal blood cholesterol concentrations.
Eating more Whole Grains linked with Lower Risk of Death
Eating at least three servings of whole grains every day could lower your risk of death, according to new research in the American Heart Association’s journal Circulation 139. Whole grains, such as whole wheat, oats and brown rice, contain dietary fiber, which may help improve blood cholesterol levels, and lower the risk of heart disease, stroke, obesity and type 2 diabetes. Dietary fiber can also make you feel full longer, so you may eat fewer calories.
Although dietary guidelines around the world have included whole grains as an essential component of healthy eating patterns, people aren’t eating enough, according to the analysis.
This analysis included 12 studies published through February 2016 and unpublished results from the National Health and Nutrition Examination Survey (NHANES) III, conducted from 1988 to 1994, and NHANES 1999-2004. Of the reviewed studies, 10 were conducted in U.S. populations, three in Scandinavian countries and one in the United Kingdom.
The combined studies involved 786,076 men and women with 97,867 total deaths, 23,597 deaths from cardiovascular disease, and 37,492 deaths from cancer.
In the first meta-analysis review of studies reporting associations between whole grain consumption and death, researchers noted that for about every serving (16 grams) of whole grains there was a:
- 7 percent decreased risk in total deaths;
- 9 percent decline in cardiovascular disease-related deaths; and
- 5 percent decline in cancer-related deaths.
The more whole grains consumed, the lower the death rate. According to researchers, when three servings (48 grams) were consumed daily the rates declined:
- 20 percent for total deaths;
- 25 percent for cardiovascular deaths; and
- 14 percent for cancer-related deaths.
Study Highlights
- Eating at least three servings of whole grains a day was associated with lower risk of death from cardiovascular disease, cancer and all causes in an analysis of nutrition studies.
- These findings further support the U.S. government’s current Dietary Guidelines for Americans, which recommends at least three daily servings of whole grains.
The American Heart Association recommends a heart-healthy dietary pattern emphasizing fruits, vegetables, whole grains and other nutritious foods and specifically that at least half of grain consumption should be whole grains. Whole grains provide many nutrients, such as fiber, B vitamins, and minerals, which are removed during the refining process.
In another recent meta-analysis using seven prospective cohort studies on high fiber diet and all-cause of death, observed an 11 % reduction in mortality risk for each 10 g/day increment of dietary fibre consumed 140. When comparing the highest (mean approximately 27 g/d) and lowest (mean approximately 15 g/d) fibre intake groups, the pooled estimate indicated a 23 % lower risk in the highest dietary fiber (cereal fibre, vegetable fibre and fruit fibre) consumers. Overall, the strongest inverse associations were observed with increasing cereal fibre consumption (8 % reduction in risk per 10 g/d), with weaker associations being observed for vegetable and fruit sources 140.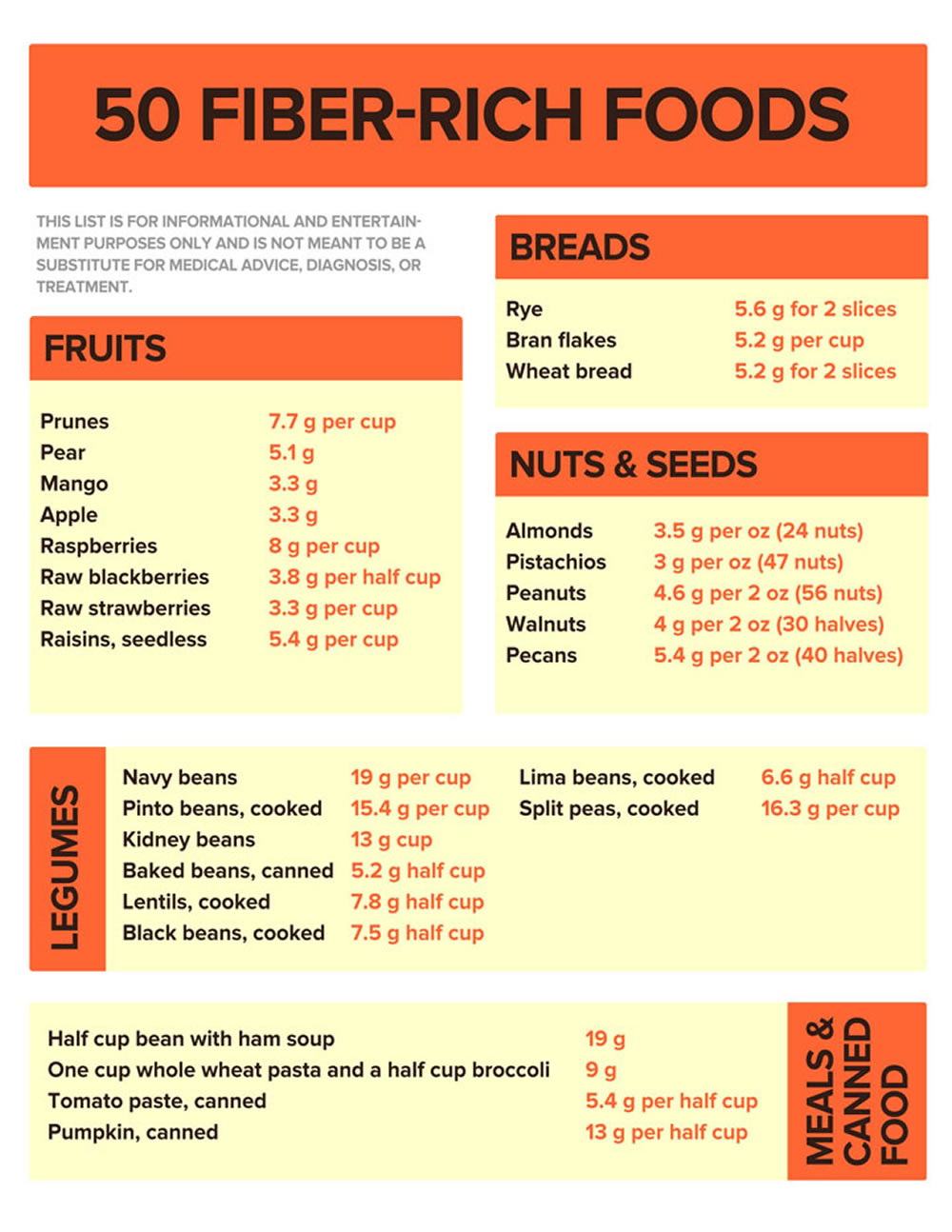
High Fiber Diets and Cancer Prevention
Recent studies support this inverse relationship between dietary fiber and the development of several types of cancers including colorectal, small intestine, oral, larynx and breast cancer 141, 142, 143. Although most studies agree with these findings, the mechanisms responsible are still unclear.
A large-scale 2016 study 144 led by researchers at Harvard School of Public Health showed findings that higher fiber intake reduces breast cancer risk, suggesting that fiber intake during adolescence and early adulthood may be particularly important. Women who eat more high-fiber foods during adolescence and young adulthood, including vegetables and fruit, may have significantly lower breast cancer risk than those who eat less dietary fiber when young 144.
Several modes of actions however have been proposed. First, dietary fiber resists digestion in the small intestine, thereby allowing it to enter the large intestine where it is fermented to produce short chain fatty acids, which have anti-carcinogenic properties 145. Second, since dietary fiber increases fecal bulking and viscosity, there is less contact time between potential carcinogens and mucosal cells. Third, dietary fiber increases the binding between bile acids and carcinogens. Fourth, increased intake of dietary fiber yield increased levels of antioxidants. Fifth, dietary fiber may increase the amount of estrogen excreted in the feces due to an inhibition of estrogen absorption in the intestines 34.
In contrary to the above positive results, several studies have largely failed to show a link between fiber and colon cancer. One of these—a Harvard study that followed over 80,000 female nurses for 16 years—found that dietary fiber was not strongly associated with a reduced risk for either colon cancer or polyps (a precursor to colon cancer) 146.
High Fiber Diets in the Treatment of Diabetes Mellitus
These data 147, 148 indicate that substantial changes in fiber content of the diabetic diet may lead to marked changes in diabetic control and that increasing dietary fiber may be a useful means of lowering plasma glucose in some diabetic patients. There is sufficient evidence to support a number of authorised health claims that certain types of dietary fibre, including arabinoxylan, β-glucans from oats and barley, and pectins if consumed within a meal, may contribute to the reduction of the blood glucose rise after that meal. Furthermore, two systematic reviews of prospective cohort studies, with dose–response meta-analyses, of the association between dietary fibre and risk of type 2 diabetes mellitus have been published recently that indicate a reduction in long-term risk 149, 150. Both reviews concluded that there is clear evidence of diminishing risk of type 2 diabetes mellitus with increasing consumption of dietary fiber (approximately 6 % reduction in risk with each additional 7 g daily consumption that fibre from cereals was associated with reduced diabetes risk. With each 7 g/d consumed, Threapleton et al. 149 reported that risk was reduced by 21 %. Both insoluble and soluble fibres were separately associated with lower risk but no associations were observed when studies reporting fibre from fruits or from vegetables were separately examined.
The high plant fiber content may be responsible for the reduction in serum cholesterol and triglyceride values. Fasting blood glucose, cholesterol and triglyceride values were significantly lower on high fiber diets than on control diets despite significantly lower insulin doses on the high fiber diets. High fiber diets were accompanied by increased insulin sensitivity and by binding of insulin by monocytes. These studies suggest that high fiber diets may have an important place in the management of patients with the maturity-onset type of diabetes. (Source 151, 152).
Diets low in fiber and high in foods that cause sudden increases in blood sugar may increase the risk of developing Type 2 Diabetes. Both Harvard studies—of female nurses and of male health professionals—found that this type of diet more than doubled the risk of type 2 diabetes when compared to a diet high in cereal fiber and low in high-glycemic-index foods 153, 154, 155. A diet high in cereal fiber was linked to a lower risk of type 2 diabetes. Other studies, such as the Black Women’s Health Study 156 and the European Prospective Investigation Into Cancer and Nutrition–Potsdam, have shown similar results.
These results suggest that in Insulin Dependent Diabetes Mellitus patients, high-carbohydrate (70%) with high-fiber (70 g) (HCHF) diets enhance peripheral glucose disposal, decrease basal insulin requirements, and lower total cholesterol without altering glycemic control or triglycerides. (Source 157).
A more recent study to find out the health benefits of soluble fiber on type 2 diabetes 91. A total of 117 patients with type 2 diabetes between the ages of 40 and 70 were assessed. Patients were randomly assigned to one of two groups, and administered extra soluble dietary fiber (10 or 20 g/day), or to a control group (0 g/day) for one month. The 20 g/day soluble dietary fiber group exhibited significantly improved fasting blood glucose and low-density (LDL) lipoprotein “bad cholesterol” levels, as well as a significantly improved insulin resistance index. In addition, 10 and 20 g/day soluble dietary fiber significantly improved the waist and hip circumferences and levels of triglycerides and apolipoprotein A. The results of the present study suggested that increased and regular consumption of soluble dietary fiber led to significant improvements in blood glucose levels, insulin resistance and metabolic profiles 91.
A high-fiber diet has many benefits, including normalizing bowel movements, helping maintain bowel integrity and health, lowering blood cholesterol levels, and helping control blood sugar levels. A high-fiber diet may also aid in achieving and maintaining a healthy weight.
High Fiber Diets in the Reducing Mortality from Cardiovascular Disease
Risk factors for cardiovascular disease include hypercholesterolemia (high cholesterol), hypertension (high blood pressure), obesity and type 2 diabetes. Recent studies found interesting data illustrating that for every 10 g of additional fiber added to a diet the mortality risk of cardiovascular disease (coronary heart disease, heart failure, peripheral vascular disease and stroke) is decreased by 17–35% 158, 159.
Increasing dietary fiber intake, which is one of the goals of nutritional counseling, deserves greater attention due to its ability to reduce total cholesterol levels and hyperglycemia in patients with impaired glucose tolerance and type 2 diabetes 100. In addition, increased fiber intake was shown to improve insulin sensitivity and reduce systemic inflammation 160, 161. Previous studies have demonstrated that high-fiber diets (30 g/day) altered biochemical parameters, reduced the severity of type 2 diabetes mellitus and decreased the occurrence of risk factors associated with cardiovascular disease 160, 162. According to Weickert et al 160, nutritional educational studies involving dietary restrictions are typically met with poor treatment compliance. Participants in a previous study were encouraged to progressively alter their eating behaviors, including increasing the frequency of meals and increasing the intake of complex carbohydrates, dietary fiber, fruits, and vegetables, as well as polyunsaturated and monounsaturated fatty acids, including fish and olive oils, respectively 160.
It is speculated that the control and treatment of these risk factors (hypercholesterolemia (high cholesterol), hypertension (high blood pressure), obesity and type 2 diabetes) underlie the mechanisms behind dietary fiber and cardiovascular disease prevention.
First, soluble fibers have been shown to increase the rate of bile excretion therefore reducing serum total cholesterol and LDL “bad” cholesterol 163. Second, short chain fatty acid production, specifically propionate, has been shown to inhibit cholesterol synthesis 37. Third, dietary fiber demonstrates the ability to regulate energy intake thus enhancing weight loss or maintenance of a healthier body weight. Fourth, either through glycemic control or reduced energy intake, dietary fiber has been shown to lower the risk for type two diabetes. Fifth, dietary fiber has been shown to decrease pro-inflammatory cytokines such as interleukin-18 which may have an effect on plaque stability 164. Sixth, increasing dietary fiber intake has been show to decrease circulating levels of C-Reactive protein, a marker of inflammation and a predictor for cardiovascular disease 165.
High Fiber Diets and Hypertension (high blood pressure)
Two reviews of randomised controlled trials of fibre and blood pressure were published in 2005 by Streppel et al. 166 and Whelton et al. 167. Both described a significant inverse relationship between fibre consumption and blood pressure, with reductions in blood pressure by 1–2 mmHg with fibre supplementation. Greater reductions were observed in older, more hypertensive populations. Whelton et al. 167 also described some evidence of a greater magnitude of reduction with fruit and vegetable sources of dietary fibre rather than grain sources, although this finding was based on a very limited number of studies (four and nine trials, respectively). More recently, randomised controlled trials with blood pressure outcomes that were of at least of 6 weeks’ duration were identified by Evans et al. 168, and pooled in random-effects meta-analyses. No overall reduction in blood pressure was found when the results of all eighteen trials were pooled, comparing high- and low-fibre intervention groups regardless of dietary fibre type. However, diets rich in β-glucans were found to reduce systolic blood pressure by 2·9 mmHg and diastolic blood pressure by 1·5 mmHg for a median difference in β-glucans of 4 g/d.
High Fiber Diets May Reduce Stroke Risk
New research in the American Heart Association journal Stroke March 28, 2013 169, 170 shows that eating more fiber may decrease your risk of first-time stroke.
Among the study’s key highlights:
- Eating foods with more fiber was linked to a lower risk of first-time stroke.
- Every seven-gram increase in total dietary fiber was associated with a 7 percent lower risk of first-time stroke.
- The results reinforce the importance of a diet that includes at least 25 grams of fiber daily.
Previous research has shown that dietary fiber may help reduce risk factors for stroke, including high blood pressure and high blood levels of low-density lipoprotein (LDL) “bad” cholesterol.
In the new study, researchers found that each seven-gram increase in total daily fiber intake was associated with a 7 percent decrease in first-time stroke risk. One serving of whole-wheat pasta, plus two servings of fruits or vegetables, provides about 7 grams of fiber, researchers said.
Greater intake of fiber-rich foods – such as whole-grains, fruits, vegetables and nuts – are important for everyone, and especially for those with stroke risk factors like being overweight, smoking and having high blood pressure.
The results were based on total dietary fiber. Researchers did not find an association with soluble fiber and stroke risk, and lacked enough data on insoluble fiber to make any conclusions.
The average daily fiber intake among U.S. adults is lower than the American Heart Association’s recommendation of at least 25 grams per day. Six to eight servings of grains and eight to 10 servings of fruits and vegetables can provide the recommended amount.
Most people do not get the recommended level of fiber, and increasing fiber may contribute to lower risk for strokes.
Whole Grains Intake and Mortality From All Causes, Cardiovascular Disease, and Cancer
Eating at least three servings of whole grains a day was associated with lower risk of death from cardiovascular disease, cancer and all causes in an analysis of nutrition studies. A Meta-Analysis of Prospective Cohort Studies 171 demonstrated inverse associations of whole grains intake with total and cause-specific mortality, and findings were particularly strong and robust for cardiovascular disease mortality.
Whole Grains consumption was inversely associated with mortality in a dose-response manner, and the association with cardiovascular disease mortality was particularly strong and robust. Further dose-response analysis showed a strong monotonic association of whole grains with total and cause-specific mortality. These observations endorse current dietary guidelines that recommend increasing whole grains intake to replace refined grains to facilitate long-term health and to help prevent premature death.
The relationship between whole grains intake and cancer outcomes is less clear. In their meta-analysis, significantly lower cancer mortality was observed only when daily whole grains consumption exceeded 30 g/d.
Huang et al 172 found an inverse association between whole grains intake and mortality from respiratory diseases, infections, and other unknown causes. Johnsen et al 173 also reported that whole grains intake was associated with lower mortality resulting from causes other than cardiovascular, cancer, diabetes mellitus and respiratory diseases. Clearly, more data are required to further elucidate the potential benefits of whole grains on other health conditions.
These findings support current dietary guidelines for whole grains consumption that recommends ≥3 servings per day for long-term health and longevity. These findings further support current Dietary Guidelines for Americans, which recommends at least 3 servings per day of whole grains intake.
Dietary fiber summary
In a simplified definition, dietary fiber is a carbohydrate that resists digestion and absorption and may or may not undergo microbial fermentation in the large intestine. This definition is essentially the basis to its correlation between consumption levels and possible health benefits. Dietary fiber consists of many different constituents, however; some are of particular interest and include arabinoxylan, inulin, β-glucan, pectin, bran and resistant starches. These individual components of dietary fiber have been shown to significantly play an important role in improving human health. Current research is paying particular attention to these elements; although further research is needed to better understand particular health claims and the mechanisms involved.
A large amount of research has reported an inverse relationship between high fiber consumption and the risk for coronary heart disease and several types of cancer. For that reason, the FDA has adopted and published the claim that increased consumption of dietary fiber can reduce the prevalence of coronary heart diseases and cancer. The mechanisms behind these findings are still unclear. However, it is thought to be attributed to several factors including increasing bile acid excretion, decreased caloric intake, increased short chain fatty acid production, carcinogen binding effects, increased antioxidants, and increased vitamins and minerals.
A heart-healthy dietary pattern emphasizing fruits, vegetables, whole grains and other nutritious foods and specifically that at least half of grain consumption should be whole grains. Whole grains provide many nutrients, such as fiber, B vitamins, and minerals, which are removed during the refining process. Based on the solid evidence from numerous previous studies that collectively document beneficial effects of whole grains, we unanimously recommend whole grain consumption to the general population as well as to people with certain diseases to help achieve better health and perhaps reduce death.
Although not as yet adopted by the FDA, dietary fiber is suggested to play a role in other conditions such as obesity and diabetes. Although some data are contradictory, a majority of studies regarding dietary fiber report a decrease of these two conditions with increased consumption of fiber.
The digestive and viscosity characteristics of dietary fiber are the likely modes of action which affect diabetes and obesity risk. These mechanisms appear to decrease nutrient absorption, therefore, decreasing metabolizable energy. Dietary fiber may also be able to decrease gross energy of a food due to its lower energy density.
Further studies are needed in certain areas of dietary fiber research. Those of particular interest are in the components of fiber such as β-glucan, arabinoxylan, resistant starches, etc. These sub fractions may give a better understanding of the health benefits of dietary fiber as well as the mechanisms behind them 174.
References- European Journal of Clinical Nutrition (2007) 61 (Suppl 1), S132–S137; doi:10.1038/sj.ejcn.1602943. FAO/WHO Scientific Update on carbohydrates in human nutrition: conclusions. http://www.nature.com/ejcn/journal/v61/n1s/full/1602943a.html
- U.S. Department of Agriculture and U.S. Department of Health and Human Services. Dietary Guidelines for Americans, 2015-2020. https://health.gov/sites/default/files/2019-09/2015-2020_Dietary_Guidelines.pdf
- American Heart Association Scientific Statement October 27, 2016 – Statement provides blueprint for healthcare providers to translate nutrition recommendations into practical food choices – http://newsroom.heart.org/news/statement-provides-blueprint-for-healthcare-providers-to-translate-nutrition-recommendations-into-practical-food-choices
- Glycemic index, glycemic load, and dietary fiber intake and incidence of type 2 diabetes in younger and middle-aged women. Schulze MB, Liu S, Rimm EB, Manson JE, Willett WC, Hu FB. Am J Clin Nutr. 2004 Aug; 80(2):348-56. https://www.ncbi.nlm.nih.gov/pubmed/15277155/
- Carbohydrates, dietary fiber, and incident type 2 diabetes in older women. Meyer KA, Kushi LH, Jacobs DR Jr, Slavin J, Sellers TA, Folsom AR. Am J Clin Nutr. 2000 Apr; 71(4):921-30. https://www.ncbi.nlm.nih.gov/pubmed/10731498/
- Carbohydrates, dietary fiber, and incident type 2 diabetes mellitus in older women. Meyer KA, Kushi LH, Jacobs DR Jr, Slavin J, Sellers TA, Folsom AR. Am J Clin Nutr. 2000 Apr; 71(4):921-30. https://www.ncbi.nlm.nih.gov/pubmed/10731498/
- Diet, lifestyle, and the risk of type 2 diabetes mellitus in women. Hu FB, Manson JE, Stampfer MJ, Colditz G, Liu S, Solomon CG, Willett WC. N Engl J Med. 2001 Sep 13; 345(11):790-7. https://www.ncbi.nlm.nih.gov/pubmed/11556298/
- Whole-grain and fiber intake and the incidence of type 2 diabetes. Montonen J, Knekt P, Järvinen R, Aromaa A, Reunanen A. Am J Clin Nutr. 2003 Mar; 77(3):622-9. https://www.ncbi.nlm.nih.gov/pubmed/12600852/
- Dietary fiber, glycemic load, and risk of NIDDM in men. Salmerón J, Ascherio A, Rimm EB, Colditz GA, Spiegelman D, Jenkins DJ, Stampfer MJ, Wing AL, Willett WC. Diabetes Care. 1997 Apr; 20(4):545-50. https://www.ncbi.nlm.nih.gov/pubmed/9096978/
- Dietary fibres, fibre analogues, and glucose tolerance: importance of viscosity. Jenkins DJ, Wolever TM, Leeds AR, Gassull MA, Haisman P, Dilawari J, Goff DV, Metz GL, Alberti KG. Br Med J. 1978 May 27; 1(6124):1392-4. https://www.ncbi.nlm.nih.gov/pubmed/647304/
- Metabolic effects of dietary fiber consumption and prevention of diabetes. Weickert MO, Pfeiffer AF. J Nutr. 2008 Mar; 138(3):439-42. https://www.ncbi.nlm.nih.gov/pubmed/18287346/
- Impact of cereal fibre on glucose-regulating factors. Weickert MO, Mohlig M, Koebnick C, Holst JJ, Namsolleck P, Ristow M, Osterhoff M, Rochlitz H, Rudovich N, Spranger J, Pfeiffer AF. Diabetologia. 2005 Nov; 48(11):2343-53. https://www.ncbi.nlm.nih.gov/pubmed/16172868/
- Insoluble cereal fiber reduces appetite and short-term food intake and glycemic response to food consumed 75 min later by healthy men. Samra RA, Anderson GH. Am J Clin Nutr. 2007 Oct; 86(4):972-9. https://www.ncbi.nlm.nih.gov/pubmed/17921373/
- Effect of neutralized and native vinegar on blood glucose and acetate responses to a mixed meal in healthy subjects. Brighenti F, Castellani G, Benini L, Casiraghi MC, Leopardi E, Crovetti R, Testolin G. Eur J Clin Nutr. 1995 Apr; 49(4):242-7. https://www.ncbi.nlm.nih.gov/pubmed/7796781/
- Barley bread containing lactic acid improves glucose tolerance at a subsequent meal in healthy men and women. Ostman EM, Liljeberg Elmståhl HG, Björck IM. J Nutr. 2002 Jun; 132(6):1173-5. https://www.ncbi.nlm.nih.gov/pubmed/12042429/
- Effect of fatty acids on glucose production and utilization in man. Ferrannini E, Barrett EJ, Bevilacqua S, DeFronzo RA. J Clin Invest. 1983 Nov; 72(5):1737-47. https://www.ncbi.nlm.nih.gov/pubmed/6138367/
- Role of acetate in the reduction of plasma free fatty acids produced by ethanol in man. Crouse JR, Gerson CD, DeCarli LM, Lieber CS. J Lipid Res. 1968 Jul; 9(4):509-12. https://www.ncbi.nlm.nih.gov/pubmed/5725882/
- Fuel selection in human skeletal muscle in insulin resistance: a reexamination. Kelley DE, Mandarino LJ. Diabetes. 2000 May; 49(5):677-83. https://www.ncbi.nlm.nih.gov/pubmed/10905472/
- U.S. Department of Health and Human Services and U.S. Department of Agriculture: Dietary Guidelines for Americans 2015-2020. https://health.gov/dietaryguidelines/
- Increasing total fiber intake reduces risk of weight and fat gains in women. Tucker LA, Thomas KS. J Nutr. 2009 Mar; 139(3):576-81. https://www.ncbi.nlm.nih.gov/pubmed/19158230/
- Dietary fiber intake and risk of breast cancer in postmenopausal women: the National Institutes of Health-AARP Diet and Health Study. Park Y, Brinton LA, Subar AF, Hollenbeck A, Schatzkin A. Am J Clin Nutr. 2009 Sep; 90(3):664-71. https://www.ncbi.nlm.nih.gov/pubmed/19625685/
- Dietary fiber intake in relation to coronary heart disease and all-cause mortality over 40 y: the Zutphen Study. Streppel MT, Ocké MC, Boshuizen HC, Kok FJ, Kromhout D. Am J Clin Nutr. 2008 Oct; 88(4):1119-25. https://www.ncbi.nlm.nih.gov/pubmed/18842802/
- FDA, authors. Code of Federal Regulations. Vol. 2 Food and Drug Administration; Silver Spring, MD, USA: 2008. Health claims: Fiber-contaning grain products, fruits and vegetables and cancer.
- FDA, authors. Code of Federal Regulations. Vol. 2 Food and Drug Administration; Silver Spring, MD, USA: 2008. Health claims: fruits, vegetables, and grain products that contain fiber, particularly soluble fiber, and risk of coronary heart disease.
- Liu R.H. Health benefits of fruit and vegetables are from additive and synergistic combinations of phytochemicals. Am. J. Clin. Nutr. 2003;78:517S–520S. – https://www.ncbi.nlm.nih.gov/pubmed/12936943
- Tucker L.A., Thomas K.S. Increasing total fiber intake reduces risk of weight and fat gains in women. J. Nutr. 2009;139:576–581. – https://www.ncbi.nlm.nih.gov/pubmed/19158230
- Weickert M.O., Pfeiffer A.F. Metabolic effects of dietary fiber consumption and prevention of diabetes. J. Nutr. 2008;138:439–442. – https://www.ncbi.nlm.nih.gov/pubmed/18287346
- Liu S., Stampfer M.J., Hu F.B., Giovannucci E., Rimm E., Manson J.E., Hennekens C.H., Willett W.C. Whole-grain consumption and risk of coronary heart disease: Results from the Nurses’ Health Study. Am. J. Clin. Nutr. 1999;70:412–419. – https://www.ncbi.nlm.nih.gov/pubmed/10479204
- Ferguson L.R., Chavan R.R., Harris P.J. Changing concepts of dietary fiber: Implications for carcinogenesis. Nutr. Cancer. 2001;39:155–169. – https://www.ncbi.nlm.nih.gov/pubmed/11759275
- Terry P., Giovannucci E., Michels K.B., Bergkvist L., Hansen H., Holmberg L., Wolk A. Fruit, vegetables, dietary fiber, and risk of colorectal cancer. J. Natl. Cancer Inst. 2001;93:525–533. – https://www.ncbi.nlm.nih.gov/pubmed/11287446
- Dietary fiber and colorectal cancer risk: the multiethnic cohort study. Nomura AM, Hankin JH, Henderson BE, Wilkens LR, Murphy SP, Pike MC, Le Marchand L, Stram DO, Monroe KR, Kolonel LN. Cancer Causes Control. 2007 Sep; 18(7):753-64. https://www.ncbi.nlm.nih.gov/pubmed/17557210/
- Prospective study of dietary fiber, whole grain foods, and small intestinal cancer. Schatzkin A, Park Y, Leitzmann MF, Hollenbeck AR, Cross AJ. Gastroenterology. 2008 Oct; 135(4):1163-7. https://www.ncbi.nlm.nih.gov/pubmed/18727930/
- Dietary fibre and colorectal cancer: a model for environment–gene interactions. Young GP, Hu Y, Le Leu RK, Nyskohus L. Mol Nutr Food Res. 2005 Jun; 49(6):571-84. https://www.ncbi.nlm.nih.gov/pubmed/15864783/
- Adlercreutz H., Hamalainen E., Gorbach S.L., Goldin B.R., Woods M.N., Brunson L.S., Dwyer J.T. Association of Diet and Sex-Hormones in Relation to Breast-Cancer. Eur. J. Cancer Clin. Oncol. 1987;23:1725–1726.
- Dietary fiber and risk of coronary heart disease: a pooled analysis of cohort studies. Pereira MA, O’Reilly E, Augustsson K, Fraser GE, Goldbourt U, Heitmann BL, Hallmans G, Knekt P, Liu S, Pietinen P, Spiegelman D, Stevens J, Virtamo J, Willett WC, Ascherio A. Arch Intern Med. 2004 Feb 23; 164(4):370-6. https://www.ncbi.nlm.nih.gov/pubmed/14980987/
- Dietary fiber and bile acid metabolism–an update. Story JA, Furumoto EJ, Buhman KK. Adv Exp Med Biol. 1997; 427():259-66. https://www.ncbi.nlm.nih.gov/pubmed/9361851/
- Amaral L., Morgan D., Stephen A.M., Whiting S. Effect of Propionate on Lipid-Metabolism in Healthy-Human Subjects. FASEB J. 1992;6:A1655.
- Meal modulation of circulating interleukin 18 and adiponectin concentrations in healthy subjects and in patients with type 2 diabetes mellitus. Esposito K, Nappo F, Giugliano F, Di Palo C, Ciotola M, Barbieri M, Paolisso G, Giugliano D. Am J Clin Nutr. 2003 Dec; 78(6):1135-40. https://www.ncbi.nlm.nih.gov/pubmed/14668275/
- Association between dietary fiber and serum C-reactive protein. Ma Y, Griffith JA, Chasan-Taber L, Olendzki BC, Jackson E, Stanek EJ 3rd, Li W, Pagoto SL, Hafner AR, Ockene IS. Am J Clin Nutr. 2006 Apr; 83(4):760-6. https://www.ncbi.nlm.nih.gov/pubmed/16600925/
- Lattimer JM, Haub MD. Effects of Dietary Fiber and Its Components on Metabolic Health. Nutrients. 2010;2(12):1266-1289. doi:10.3390/nu2121266. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3257631/
- WHO/FAO (2009) Report of the 30th session of the Codex Committee on Nutrition and Foods for Special Dietary Uses, no. ALINORM 02/32/26. http://www.fao.org/fao-who-codexalimentarius/codex-home/en/
- NG Asp (2001) Development of dietary fibre methodology. In Advanced Dietary Fibre Technology, pp. 77–86 [B McCleary and L Prosky, editors]. London: Blackwell Science Ltd.
- Stephen, A., Champ, M., Cloran, S., Fleith, M., Van Lieshout, L., Mejborn, H., & Burley, V. (2017). Dietary fibre in Europe: Current state of knowledge on definitions, sources, recommendations, intakes and relationships to health. Nutrition Research Reviews, 1-42. doi:10.1017/S095442241700004X. https://www.cambridge.org/core/journals/nutrition-research-reviews/article/dietary-fibre-in-europe-current-state-of-knowledge-on-definitions-sources-recommendations-intakes-and-relationships-to-health/B263D1D7B3440DC9D6F68E23C2B4212F
- American Association of Cereal Chemists (AACC International). Dietary Fiber. http://www.aaccnet.org/initiatives/definitions/Pages/DietaryFiber.aspx
- American Association of Cereal Chemists (AACC International). Dietary Fiber. http://www.aaccnet.org/initiatives/definitions/Pages/DietaryFiber.aspx
- Food and Agriculture Organization and World Health Organization. CODEX ALIMENTARIUS COMMISSION. Thirty second Session, Rome, Italy, 29 June – 4 July 2009. http://www.fao.org/input/download/report/710/al32_26e.pdf
- Ring S.G., Selvendran R.R. Isolation and Analysis of Cell-Wall Material from Beeswing Wheat Bran (Triticum-Aestivum) Phytochemistry. 1980;19:1723–1730.
- Arabinoxylan fiber, a byproduct of wheat flour processing, reduces the postprandial glucose response in normoglycemic subjects. Lu ZX, Walker KZ, Muir JG, Mascara T, O’Dea K. Am J Clin Nutr. 2000 May; 71(5):1123-8. https://www.ncbi.nlm.nih.gov/pubmed/10799374/
- Arabinoxylan fibre improves metabolic control in people with Type II diabetes. Lu ZX, Walker KZ, Muir JG, O’Dea K. Eur J Clin Nutr. 2004 Apr; 58(4):621-8. https://www.ncbi.nlm.nih.gov/pubmed/15042130/
- Selective stimulation of bifidobacteria in the human colon by oligofructose and inulin. Gibson GR, Beatty ER, Wang X, Cummings JH. Gastroenterology. 1995 Apr; 108(4):975-82. https://www.ncbi.nlm.nih.gov/pubmed/7698613/
- Dietary synbiotics reduce cancer risk factors in polypectomized and colon cancer patients. Rafter J, Bennett M, Caderni G, Clune Y, Hughes R, Karlsson PC, Klinder A, O’Riordan M, O’Sullivan GC, Pool-Zobel B, Rechkemmer G, Roller M, Rowland I, Salvadori M, Thijs H, Van Loo J, Watzl B, Collins JK. Am J Clin Nutr. 2007 Feb; 85(2):488-96. https://www.ncbi.nlm.nih.gov/pubmed/17284748/
- Griffin I.J., Hicks P.M.D., Heaney R.P., Abrams S.A. Enriched chicory inulin increases calcium absorption mainly in girls with lower calcium absorption. Nutr. Res. 2003;23:901–909.
- A combination of prebiotic short- and long-chain inulin-type fructans enhances calcium absorption and bone mineralization in young adolescents. Abrams SA, Griffin IJ, Hawthorne KM, Liang L, Gunn SK, Darlington G, Ellis KJ. Am J Clin Nutr. 2005 Aug; 82(2):471-6. https://www.ncbi.nlm.nih.gov/pubmed/16087995/
- Oligofructose promotes satiety in healthy human: a pilot study. Cani PD, Joly E, Horsmans Y, Delzenne NM. Eur J Clin Nutr. 2006 May; 60(5):567-72. https://www.ncbi.nlm.nih.gov/pubmed/16340949/
- Wood P.J., Weisz J., Fedec P. Potential for Beta-Glucan Enrichment in Brans Derived from Oat (Avena sativa L.) Cultivars of Different (1→3),(1→4)-Beta-D-Glucan Concentrations. Cereal Chem. 1991;68:48–51.
- Simultaneous intake of beta-glucan and plant stanol esters affects lipid metabolism in slightly hypercholesterolemic subjects. Theuwissen E, Mensink RP. J Nutr. 2007 Mar; 137(3):583-8. https://www.ncbi.nlm.nih.gov/pubmed/17311944/
- Beta-glucan incorporated into a fruit drink effectively lowers serum LDL-cholesterol concentrations. Naumann E, van Rees AB, Onning G, Oste R, Wydra M, Mensink RP. Am J Clin Nutr. 2006 Mar; 83(3):601-5. https://www.ncbi.nlm.nih.gov/pubmed/16522906/
- Davidson M.H., McDonald A. Fiber: Forms and functions. Nutr. Res. 1998;18:617–624.
- Changes in serum lipids and postprandial glucose and insulin concentrations after consumption of beverages with beta-glucans from oats or barley: a randomised dose-controlled trial. Biörklund M, van Rees A, Mensink RP, Onning G. Eur J Clin Nutr. 2005 Nov; 59(11):1272-81. https://www.ncbi.nlm.nih.gov/pubmed/16015250/
- Regulation of hepatic glucose production in healthy subjects and patients with non-insulin-dependent diabetes mellitus. Tappy L. Diabete Metab. 1995 Oct; 21(4):233-40. https://www.ncbi.nlm.nih.gov/pubmed/8529757/
- Oat beta-glucan increases bile acid excretion and a fiber-rich barley fraction increases cholesterol excretion in ileostomy subjects. Lia A, Hallmans G, Sandberg AS, Sundberg B, Aman P, Andersson H. Am J Clin Nutr. 1995 Dec; 62(6):1245-51. https://www.ncbi.nlm.nih.gov/pubmed/7491888/
- Modulation of the postprandial phase by beta-glucan in overweight subjects: effects on glucose and insulin kinetics. Nazare JA, Normand S, Oste Triantafyllou A, Brac de la Perrière A, Desage M, Laville M. Mol Nutr Food Res. 2009 Mar; 53(3):361-9. https://www.ncbi.nlm.nih.gov/pubmed/18837470/
- In vitro bile-acid binding and fermentation of high, medium, and low molecular weight beta-glucan. Kim HJ, White PJ. J Agric Food Chem. 2010 Jan 13; 58(1):628-34. https://www.ncbi.nlm.nih.gov/pubmed/20020684/
- In vitro fermentation of oat flours from typical and high beta-glucan oat lines. Kim HJ, White PJ. J Agric Food Chem. 2009 Aug 26; 57(16):7529-36. https://www.ncbi.nlm.nih.gov/pubmed/19572543/
- Regulation by dietary fats of 3-hydroxy-3-methylglutaryl-Coenzyme A reductase in rat liver. Ide T, Okamatsu H, Sugano M. J Nutr. 1978 Apr; 108(4):601-12. https://www.ncbi.nlm.nih.gov/pubmed/632948/
- Randomized controlled crossover study of the effect of a highly beta-glucan-enriched barley on cardiovascular disease risk factors in mildly hypercholesterolemic men. Keogh GF, Cooper GJ, Mulvey TB, McArdle BH, Coles GD, Monro JA, Poppitt SD. Am J Clin Nutr. 2003 Oct; 78(4):711-8. https://www.ncbi.nlm.nih.gov/pubmed/14522728/
- A controlled study of consumption of beta-glucan-enriched soups for 2 months by type 2 diabetic free-living subjects. Cugnet-Anceau C, Nazare JA, Biorklund M, Le Coquil E, Sassolas A, Sothier M, Holm J, Landin-Olsson M, Onning G, Laville M, Moulin P. Br J Nutr. 2010 Feb; 103(3):422-8. https://www.ncbi.nlm.nih.gov/pubmed/19781120/
- Olano-Martin E., Gibson G.R., Rastell R.A. Comparison of the in vitro bifidogenic properties of pectins and pectic-oligosaccharides. J. Appl. Microbiol. 2002;93:505–511. doi: 10.1046/j.1365-2672.2002.01719.x. https://www.ncbi.nlm.nih.gov/pubmed/12174051
- Nangia-Makker P., Hogan V., Honjo Y., Baccarini S., Tait L., Bresalier R., Raz A. Inhibition of human cancer cell growth and metastasis in nude mice by oral intake of modified citrus pectin. J. Natl. Cancer Inst. 2002;94:1854–1862. https://www.ncbi.nlm.nih.gov/pubmed/12488479
- Ulmius M., Johansson A., Onning G. The influence of dietary fibre source and gender on the postprandial glucose and lipid response in healthy subjects. Eur. J. Nutr. 2009;48:395–402. https://www.ncbi.nlm.nih.gov/pubmed/19415409
- Qureshi A.A., Sami S.A., Khan F.A. Effects of stabilized rice bran, its soluble and fiber fractions on blood glucose levels and serum lipid parameters in humans with diabetes mellitus Types I and II. J. Nutr. Biochem. 2002;13:175–187. https://www.ncbi.nlm.nih.gov/pubmed/11893482
- Koh-Banerjee P., Franz M.V., Sampson L., Liu S.M., Jacobs D.R., Spiegelman D., Willett W., Rimm E. Changes in whole-grain, bran, and cereal fiber consumption in relation to 8-y weight gain among men. Am. J. Clin. Nutr. 2004;80:1237–1245. https://www.ncbi.nlm.nih.gov/pubmed/15531671
- Zhang J.X., Lundin E., Hallmans G., Adlercreutz H., Andersson H., Bosaeus I., Aman P., Stenling R., Dahlgren S. Effect of Rye Bran on Excretion of Bile-Acids, Cholesterol, Nitrogen, and Fat in Human-Subjects with Ileostomies. Am. J. Clin. Nutr. 1994;59:389–394. https://www.ncbi.nlm.nih.gov/pubmed/8310990
- Jensen M.K., Koh-Banerjee P., Hu F.B., Franz M., Sampson L., Gronbaek M., Rimm E.B. Intakes of whole grains, bran, and germ and the risk of coronary heart disease in men. Am. J. Clin. Nutr. 2004;80:1492–1499. https://www.ncbi.nlm.nih.gov/pubmed/15585760
- Andersson M., Ellegard L., Andersson H. Oat bran stimulates bile acid synthesis within 8 h as measured by 7 alpha-hydroxy-4-cholesten-3-one. Am. J. Clin. Nutr. 2002;76:1111–1116. https://www.ncbi.nlm.nih.gov/pubmed/12399287
- Schwartz S.E., Levine R.A., Singh A., Scheidecker J.R., Track N.S. Sustained pectin ingestion delays gastric emptying. Gastroenterology. 1982;83:812–817. https://www.ncbi.nlm.nih.gov/pubmed/6286402
- Lightowler H.J., Henry C.J. Glycemic response of mashed potato containing high-viscocity hydroxypropylmethylcellulose. Nutr. Res. 2009;29:551–557. https://www.ncbi.nlm.nih.gov/pubmed/19761889
- Maki K.C., Davidson M.H., Witchger M.S., Dicklin M.R., Subbaiah P.V. Effects of high‑fiber oat and wheat cereals on postprandial glucose and lipid responses in healthy men. Int. J. Vitam. Nutr. Res. 2007;77:347–356. https://www.ncbi.nlm.nih.gov/pubmed/18453320
- Maki K.C., Davidson M.H., Torri S., Ingram K.A., O’Mullane J., Daggy B.P., Albrecht H.H. High-molecular-weight hydroxypropylmethylcellulose taken with or between meals is hypocholesterolemic in adult men. J. Nutr. 2000;130:1705–1710. https://www.ncbi.nlm.nih.gov/pubmed/10867040
- Maki K.C., Carson M.L., Anderson W.H.K., Geohas J., Reeves M.S., Farmer M.V., Turowski M., Miller M., Kaden V.N., Dicklin M.R., Rains T.M. Lipid-altering effects of different formulations of hydroxypropylmethylcellulose. J. Clin. Lipidol. 2009;3:159–166. https://www.ncbi.nlm.nih.gov/pubmed/21291811
- Food and Agriculture Organization. Physiological Effects of Dietary Fibre. http://www.fao.org/docrep/w8079e/w8079e0l.htm
- Therapeutic effects of psyllium in type 2 diabetic patients. Sierra M, García JJ, Fernández N, Diez MJ, Calle AP. Eur J Clin Nutr. 2002 Sep; 56(9):830-42. https://www.ncbi.nlm.nih.gov/pubmed/12209371/
- Gastric emptying in early noninsulin-dependent diabetes mellitus. Jones KL, Horowitz M, Carney BI, Wishart JM, Guha S, Green L. J Nucl Med. 1996 Oct; 37(10):1643-8. https://www.ncbi.nlm.nih.gov/pubmed/8862300/
- Action of guar gums on the viscosity of digestive contents and on the gastrointestinal motor function in pigs. Cherbut C, Albina E, Champ M, Doublier JL, Lecannu G. Digestion. 1990; 46(4):205-13. https://www.ncbi.nlm.nih.gov/pubmed/2178135/
- Effect of viscous fiber (guar) on postprandial motor activity in human small bowel. Schönfeld J, Evans DF, Wingate DL. Dig Dis Sci. 1997 Aug; 42(8):1613-7. https://www.ncbi.nlm.nih.gov/pubmed/9286225/
- Effect of gel-forming gums on the intestinal unstirred layer and sugar transport in vitro. Johnson IT, Gee JM. Gut. 1981 May; 22(5):398-403. https://www.ncbi.nlm.nih.gov/pubmed/7250752/
- Role of viscous guar gums in lowering the glycemic response after a solid meal. Leclère CJ, Champ M, Boillot J, Guille G, Lecannu G, Molis C, Bornet F, Krempf M, Delort-Laval J, Galmiche JP. Am J Clin Nutr. 1994 Apr; 59(4):914-21. https://www.ncbi.nlm.nih.gov/pubmed/7818627/
- Dietary guar gum improves insulin sensitivity in streptozotocin-induced diabetic rats. Cameron-Smith D, Habito R, Barnett M, Collier GR. J Nutr. 1997 Feb; 127(2):359-64. https://www.ncbi.nlm.nih.gov/pubmed/9039840/
- Soluble dietary fibre improves insulin sensitivity by increasing muscle GLUT-4 content in stroke-prone spontaneously hypertensive rats. Song YJ, Sawamura M, Ikeda K, Igawa S, Yamori Y. Clin Exp Pharmacol Physiol. 2000 Jan-Feb; 27(1-2):41-5. https://www.ncbi.nlm.nih.gov/pubmed/10696527/
- Troglitazone effects on gene expression in human skeletal muscle of type II diabetes involve up-regulation of peroxisome proliferator-activated receptor-gamma. Park KS, Ciaraldi TP, Lindgren K, Abrams-Carter L, Mudaliar S, Nikoulina SE, Tufari SR, Veerkamp JH, Vidal-Puig A, Henry RR. J Clin Endocrinol Metab. 1998 Aug; 83(8):2830-5. https://www.ncbi.nlm.nih.gov/pubmed/9709955/
- Chen C, Zeng Y, Xu J, et al. Therapeutic effects of soluble dietary fiber consumption on type 2 diabetes mellitus. Experimental and Therapeutic Medicine. 2016;12(2):1232-1242. doi:10.3892/etm.2016.3377. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4950069/
- Aldoori WH, Giovannucci EL, Rockett HR, Sampson L, Rimm EB, Willett WC. A prospective study of dietary fiber types and symptomatic diverticular disease in men. J Nutr. 1998;128:714-9. https://www.ncbi.nlm.nih.gov/pubmed/9521633
- Resistant starch: metabolic effects and potential health benefits. Higgins JA. J AOAC Int. 2004 May-Jun; 87(3):761-8. https://www.ncbi.nlm.nih.gov/pubmed/15287677/
- Birt DF, Boylston T, Hendrich S, et al. Resistant Starch: Promise for Improving Human Health. Advances in Nutrition. 2013;4(6):587-601. doi:10.3945/an.113.004325. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3823506/
- Englyst HN, Kingman SM, Cummings JH. Classification and measurement of nutritionally important starch fractions. Eur J Clin Nutr. 1992;46(Supp 2):S33–50. https://www.ncbi.nlm.nih.gov/pubmed/1330528
- Woo KS, Seib PA. Cross-linked resistant starch: preparation and properties. Cereal Chem. 2002;79:819–25.
- Han J-A, BeMiller JN. Preparation and physical characteristics of slowly digesting modified food starches. Carbohydr Polym. 2007;67:366–74.
- Seneviratne HD, Biliaderis CG. Action of α-amylases on amylose-lipid complex superstructures. J Cereal Sci. 1991;13:129–43.
- Different types of resistant starch elicit different glucose reponses in humans. Haub MD, Hubach KL, Al-Tamimi EK, Ornelas S, Seib PA. J Nutr Metab. 2010; 2010. https://www.ncbi.nlm.nih.gov/pubmed/20700404/
- Consumption of both resistant starch and beta-glucan improves postprandial plasma glucose and insulin in women. Behall KM, Scholfield DJ, Hallfrisch JG, Liljeberg-Elmståhl HG. Diabetes Care. 2006 May; 29(5):976-81. https://www.ncbi.nlm.nih.gov/pubmed/16644623/
- Glycemic and insulinemic response of subjects with type 2 diabetes after consumption of three energy bars. Reader DM, O’Connell BS, Johnson ML, Franz M. J Am Diet Assoc. 2002 Aug; 102(8):1139-42. https://www.ncbi.nlm.nih.gov/pubmed/12171462/
- Consumption of Cross-Linked Resistant Starch (RS4(XL
- Diets containing high amylose vs amylopectin starch: effects on metabolic variables in human subjects. Behall KM, Scholfield DJ, Yuhaniak I, Canary J. Am J Clin Nutr. 1989 Feb; 49(2):337-44. https://www.ncbi.nlm.nih.gov/pubmed/2644803/
- Blood lipids, lipoproteins, apoproteins, and uric acid in men fed diets containing fructose or high-amylose cornstarch. Reiser S, Powell AS, Scholfield DJ, Panda P, Ellwood KC, Canary JJ. Am J Clin Nutr. 1989 May; 49(5):832-9. https://www.ncbi.nlm.nih.gov/pubmed/2497634/
- Diet-induced changes in serum transaminase and triglyceride levels in healthy adult men. Role of sucrose and excess calories. Porikos KP, Van Itallie TB. Am J Med. 1983 Oct; 75(4):624-30. https://www.ncbi.nlm.nih.gov/pubmed/6624769/
- Physiological effects of resistant starches on fecal bulk, short chain fatty acids, blood lipids and glycemic index. Jenkins DJ, Vuksan V, Kendall CW, Würsch P, Jeffcoat R, Waring S, Mehling CC, Vidgen E, Augustin LS, Wong E. J Am Coll Nutr. 1998 Dec; 17(6):609-16. https://www.ncbi.nlm.nih.gov/pubmed/9853541/
- The effect of raw potato starch on energy expenditure and substrate oxidation. Tagliabue A, Raben A, Heijnen ML, Deurenberg P, Pasquali E, Astrup A. Am J Clin Nutr. 1995 May; 61(5):1070-5. https://www.ncbi.nlm.nih.gov/pubmed/7733031/
- Dietary starch composition and level of energy intake alter nutrient oxidation in “carbohydrate-sensitive” men. Howe JC, Rumpler WV, Behall KM. J Nutr. 1996 Sep; 126(9):2120-9. https://www.ncbi.nlm.nih.gov/pubmed/8814200/
- Insulin-sensitizing effects of dietary resistant starch and effects on skeletal muscle and adipose tissue metabolism. Robertson MD, Bickerton AS, Dennis AL, Vidal H, Frayn KN. Am J Clin Nutr. 2005 Sep; 82(3):559-67. https://www.ncbi.nlm.nih.gov/pubmed/16155268/
- Dietitians Association of Australia. Dietary fibre: key for a happy, healthy gut. https://daa.asn.au/smart-eating-for-you/smart-eating-fast-facts/nourishing-nutrients/dietary-fibre-a-key-ingredient-in-gut-happiness/
- Liu S. Intake of refined carbohydrates and whole grain foods in relation to risk of type 2 diabetes mellitus and coronary heart disease.J Am Coll Nutr 2002;21:298-306. https://www.ncbi.nlm.nih.gov/pubmed/12166526?dopt=Abstract
- Pereira MA, O’Reilly E, Augustsson K, et al. Dietary fiber and risk of coronary heart disease: a pooled analysis of cohort studies. Arch Intern Med. 2004;164:370-6. https://www.ncbi.nlm.nih.gov/pubmed/14980987
- Rimm EB, Ascherio A, Giovannucci E, Spiegelman D, Stampfer MJ, Willett WC. Vegetable, fruit, and cereal fiber intake and risk of coronary heart disease among men. JAMA. 1996;275:447-51. https://www.ncbi.nlm.nih.gov/pubmed/8627965
- Brown L, Rosner B, Willett WW, Sacks FM. Cholesterol-lowering effects of dietary fiber: a meta-analysis. Am J Clin Nutr. 1999;69:30-42. https://www.ncbi.nlm.nih.gov/pubmed/9925120
- McKeown NM, Meigs JB, Liu S, Wilson PW, Jacques PF. Whole-grain intake is favorably associated with metabolic risk factors for type 2 diabetes and cardiovascular disease in the Framingham Offspring Study. Am J Clin Nutr. 2002;76:390-8. https://www.ncbi.nlm.nih.gov/pubmed/12145012
- McKeown NM, Meigs JB, Liu S, Saltzman E, Wilson PW, Jacques PF. Carbohydrate nutrition, insulin resistance, and the prevalence of the metabolic syndrome in the Framingham Offspring Cohort. Diabetes Care. 2004;27:538-46. https://www.ncbi.nlm.nih.gov/pubmed/14747241
- National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III): Third report of the national cholesterol education program (ncep) expert panel on detection, evaluation, and treatment of high blood cholesterol in Adults (Adult Treatment Panel III) final report. Circulation. 2002;106:3143–3421. https://www.ncbi.nlm.nih.gov/pubmed/12485966
- Ortega RM, Palencia A, López-Sobaler AM. Improvement of cholesterol levels and reduction of cardiovascular risk via the consumption of phytosterols. Br J Nutr. 2006;96(Suppl 1):S89–S93. doi: 10.1079/BJN20061708. https://www.ncbi.nlm.nih.gov/pubmed/16923260
- Theuwissen E, Mensink RP. Water-soluble dietary fibers and cardiovascular disease. Physiol Behav. 2008;94:285–292. doi: 10.1016/j.physbeh.2008.01.001. https://www.ncbi.nlm.nih.gov/pubmed/18302966
- American Diabetes Association: Nutrition recommendations and principles for people with diabetes mellitus. Diabetes Care. 2000;23(Suppl 1):S43–S46. https://www.ncbi.nlm.nih.gov/pubmed/12017676
- Bernaud FS, Rodrigues TC. Dietary fiber-adequate intake and effects on metabolism health. Arq Bras Endocrinol Metabol. 2013;57:397–405. doi: 10.1590/S0004-27302013000600001. https://www.ncbi.nlm.nih.gov/pubmed/24030179
- Frankenfeld CL. Cardiometabolic risk factors are associated with high urinary enterolactone concentration, independent of urinary enterodiol concentration and dietary fiber intake in adults. J Nutr. 2014;144:1445–1453. doi: 10.3945/jn.114.190512. https://www.ncbi.nlm.nih.gov/pubmed/24966407
- Breneman CB, Tucker L. Dietary fibre consumption and insulin resistance-the role of body fat and physical activity. Br J Nutr. 2013;110:375–383. doi: 10.1017/S0007114512004953. https://www.ncbi.nlm.nih.gov/pubmed/23218116
- Am J Clin Nutr November 2003, vol. 78 no. 5 920-927. Relation between changes in intakes of dietary fiber and grain products and changes in weight and development of obesity among middle-aged women. http://ajcn.nutrition.org/content/78/5/920.long
- Meyer K.A., Kushi L.H., Jacobs D.R., Jr., Slavin J., Sellers T.A., Folsom A.R. Carbohydrates, dietary fiber, and incident type 2 diabetes in older women. Am. J. Clin. Nutr. 2000;71:921–930. – https://www.ncbi.nlm.nih.gov/pubmed/10731498
- Park Y., Brinton L.A., Subar A.F., Hollenbeck A., Schatzkin A. Dietary fiber intake and risk of breast cancer in postmenopausal women: The National Institutes of Health-AARP Diet and Health Study. Am. J. Clin. Nutr. 2009;90:664–671. – https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2728649/
- Streppel M.T., Ocke M.C., Boshuizen H.C., Kok F.J., Kromhout D. Dietary fiber intake in relation to coronary heart disease and all-cause mortality over 40 y: The Zutphen Study. Am. J. Clin. Nutr. 2008;88:1119–1125. – https://www.ncbi.nlm.nih.gov/pubmed/18842802
- Tucker L.A., Thomas K.S. Increasing total fiber intake reduces risk of weight and fat gains in women. J. Nutr. 2009;139:576–581. https://www.ncbi.nlm.nih.gov/pubmed/19158230
- Baer D.J., Rumpler W.V., Miles C.W., Fahey G.C. Dietary fiber decreases the metabolizable energy content and nutrient digestibility of mixed diets fed to humans. J. Nutr. 1997;127:579–586. https://www.ncbi.nlm.nih.gov/pubmed/9109608
- Isken F., Klaus S., Osterhoff M., Pfeiffer A.F.H., Weickert M.O. Effects of long-term soluble vs. insoluble dietary fiber intake on high-fat diet-induced obesity in C57BL/6J mice. J. Nutr. Biochem. 2010;21:278–284. doi: 10.1016/j.jnutbio.2008.12.012. https://www.ncbi.nlm.nih.gov/pubmed/19369060
- Schneeman B.O. Dietary fiber and gastrointestinal function. Nutr. Res. 1998;18:625–632.
- Weickert M.O., Pfeiffer A.F. Metabolic effects of dietary fiber consumption and prevention of diabetes. J. Nutr. 2008;138:439–442. https://www.ncbi.nlm.nih.gov/pubmed/18287346
- Renteria-Flores J.A., Johnston L.J., Shurson G.C., Gallaher D.D. Effect of soluble and insoluble fiber on energy digestibility, nitrogen retention, and fiber digestibility of diets fed to gestating sows. J. Anim. Sci. 2008;86:2568–2575. doi: 10.2527/jas.2007-0375. https://www.ncbi.nlm.nih.gov/pubmed/18539846
- Anderson J.W. Physiological and Metabolic Effects of Dietary Fiber. Fed. Proc. 1985;44:2902–2906. https://www.ncbi.nlm.nih.gov/pubmed/2996944
- Keenan M.J., Zhou J., McCutcheon K.L., Raggio A.M., Bateman H.G., Todd E., Jones C.K., Tulley R.T., Melton S., Martin R.J., Hegsted M. Effects of resistant starch, a non–digestible fermentable fiber, on reducing body fat. Obesity (Silver Spring) 2006;14:1523–1534. https://www.ncbi.nlm.nih.gov/pubmed/17030963
- Du H.D., van der A D.L., Boshuizen H.C., Forouhi N.G., Wareham N.J., Halkjaer J., Tjonneland A., Overvad K., Jakobsen M.U., Boeing H., et al. Dietary fiber and subsequent changes in body weight and waist circumference in European men and women. Am. J. Clin. Nutr. 2010;91:329–336. doi: 10.3945/ajcn.2009.28191. https://www.ncbi.nlm.nih.gov/pubmed/20016015
- L Brown , B Rosner , WW Willett , et al. (1999) Cholesterol-lowering effects of dietary fiber: a meta-analysis. J Hypertens 69, 30–42.
- Scientific Advisory Committee on Nutrition (2015) Carbohydrates and Health Report. London: Public Health England.
- American Heart Association – Eating more whole grains linked with lower risk of death – http://newsroom.heart.org/news/eating-more-whole-grains-linked-with-lower-risk-of-death?preview=f77a
- Y Kim & Y Je (2014) Dietary fiber intake and total mortality: a meta-analysis of prospective cohort studies. Am J Epidemiol 180, 565–573. https://academic.oup.com/aje/article-lookup/doi/10.1093/aje/kwu174
- Park Y., Brinton L.A., Subar A.F., Hollenbeck A., Schatzkin A. Dietary fiber intake and risk of breast cancer in postmenopausal women: The National Institutes of Health-AARP Diet and Health Study. Am. J. Clin. Nutr. 2009;90:664–671. – https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2728649/
- Nomura A.M., Hankin J.H., Henderson B.E., Wilkens L.R., Murphy S.P., Pike M.C., Le Marchand L., Stram D.O., Monroe K.R., Kolonel L.N. Dietary fiber and colorectal cancer risk: The multiethnic cohort study. Cancer Causes Control. 2007;18:753–764. – https://www.ncbi.nlm.nih.gov/pubmed/17557210
- Schatzkin A., Park Y., Leitzmann M.F., Hollenbeck A.R., Cross A.J. Prospective study of dietary fiber, whole grain foods, and small intestinal cancer. Gastroenterology. 2008;135:1163–1167. – https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3513331/
- Farvid MS, Eliassen AH, Cho E, Liao X, Chen WY, Willett WC. Dietary fiber intake in young adults and breast cancer risk. Pediatrics 2016: 137(3). http://pediatrics.aappublications.org/content/early/2016/01/28/peds.2015-1226
- Young G.P., Hu Y., Le Leu R.K., Nyskohus L. Dietary fibre and colorectal cancer: A model for environment—gene interactions. Mol. Nutr. Food Res. 2005;49:571–584. – https://www.ncbi.nlm.nih.gov/pubmed/15864783
- Fuchs CS, Giovannucci EL, Colditz GA, et al. Dietary fiber and the risk of colorectal cancer and adenoma in women. N Engl J Med. 1999;340:169-76. https://www.ncbi.nlm.nih.gov/pubmed/9895396
- Ann Intern Med. 1978 Apr;88(4):482-6. – High-fiber diets in the treatment of diabetes mellitus.- https://www.ncbi.nlm.nih.gov/pubmed/637427
- Va Med. 1979 Nov;106(11):852-5. – High-fiber diet: its role in the treatment of diabetes mellitus reviewed. – https://www.ncbi.nlm.nih.gov/pubmed/506434
- DE Threapleton , DC Greenwood , C Evans , et al. (2013) Dietary fibre intake and diabetes risk: a systematic review and meta-analysis of prospective studies. Proc Nutr Soc 72, E253.
- B Yao , H Fang , W Xu , et al. (2014) Dietary fiber intake and risk of type 2 diabetes: a dose–response analysis of prospective studies. Eur J Epidemiol 29, 79–88.
- Adv Exp Med Biol. 1979;119:263-73.- High carbohydrate, high fiber diets for patients with diabetes. – https://www.ncbi.nlm.nih.gov/pubmed/495284
- Diabetes Care. 1978 Mar-Apr;1(2):77-82. – Long-term effects of high-carbohydrate, high-fiber diets on glucose and lipid metabolism: a preliminary report on patients with diabetes. – https://www.ncbi.nlm.nih.gov/pubmed/729433
- Fung TT, Hu FB, Pereira MA, et al. Whole-grain intake and the risk of type 2 diabetes: a prospective study in men. Am J Clin Nutr. 2002;76:535-40. https://www.ncbi.nlm.nih.gov/pubmed/12197996
- Liu S, Willett WC, Stampfer MJ, et al. A prospective study of dietary glycemic load, carbohydrate intake, and risk of coronary heart disease in US women. Am J Clin Nutr. 2000;71:1455-61. https://www.ncbi.nlm.nih.gov/pubmed/10837285
- Schulze MB, Liu S, Rimm EB, Manson JE, Willett WC, Hu FB. Glycemic index, glycemic load, and dietary fiber intake and incidence of type 2 diabetes in younger and middle-aged women. Am J Clin Nutr. 2004;80:348-56. https://www.ncbi.nlm.nih.gov/pubmed/15277155
- Krishnan S, Rosenberg L, Singer M, et al. Glycemic index, glycemic load, and cereal fiber intake and risk of type 2 diabetes in US black women. Arch Intern Med. 2007;167:2304-9. https://www.ncbi.nlm.nih.gov/pubmed/18039988
- Am J Clin Nutr. 1991 Nov;54(5):936-43. – Metabolic effects of high-carbohydrate, high-fiber diets for insulin-dependent diabetic individuals. – https://www.ncbi.nlm.nih.gov/pubmed/1659172
- Streppel M.T., Ocke M.C., Boshuizen H.C., Kok F.J., Kromhout D. Dietary fiber intake in relation to coronary heart disease and all-cause mortality over 40 y: The Zutphen Study. Am. J. Clin. Nutr. 2008;88:1119–1125. – https://www.ncbi.nlm.nih.gov/pubmed/18842802
- Pereira M.A., O’Reilly E., Augustsson K., Fraser G.E., Goldbourt U., Heitmann B.L., Hallmans G., Knekt P., Liu S.M., Pietinen P., Spiegelman D., Stevens J., Virtamo J., Willett W.C., Ascherio A. Dietary fiber and risk of coronary heart disease—A pooled analysis of cohort studies. Arch. Intern. Med. 2004;164:370–376. – https://www.ncbi.nlm.nih.gov/pubmed/14980987
- Cereal fiber improves whole-body insulin sensitivity in overweight and obese women. Weickert MO, Möhlig M, Schöfl C, Arafat AM, Otto B, Viehoff H, Koebnick C, Kohl A, Spranger J, Pfeiffer AF. Diabetes Care. 2006 Apr; 29(4):775-80. https://www.ncbi.nlm.nih.gov/pubmed/16567814/
- Whole-grain, bran, and cereal fiber intakes and markers of systemic inflammation in diabetic women. Qi L, van Dam RM, Liu S, Franz M, Mantzoros C, Hu FB. Diabetes Care. 2006 Feb; 29(2):207-11. https://www.ncbi.nlm.nih.gov/pubmed/16443861/
- Evidence-based nutrition principles and recommendations for the treatment and prevention of diabetes and related complications. American Diabetes Association. Diabetes Care. 2002 Jan; 25(1):202-12. https://www.ncbi.nlm.nih.gov/pubmed/11772917/
- Story J.A., Furumoto E.J., Buhman K.K. Dietary fiber and bile acid metabolism—an update. Adv. Exp. Med. Biol. 1997;427:259–266.- https://www.ncbi.nlm.nih.gov/pubmed/9361851
- Esposito K., Nappo F., Giugliano F., Di Palo C., Ciotola M., Barbieri M., Paolisso G., Giugliano D. Meal modulation of circulating interleukin 18 and adiponectin concentrations in healthy subjects and in patients with type 2 diabetes mellitus. Am. J. Clin. Nutr. 2003;78:1135–1140. – https://www.ncbi.nlm.nih.gov/pubmed/14668275
- Ma Y.S., Griffith J.A., Chasan-Taber L., Olendzki B.C., Jackson E., Stanek E.J., Li W.J., Pagoto S.L., Hafner A.R., Ockene I.S. Association between dietary fiber and serum C-reactive protein. Am. J. Clin. Nutr. 2006;83:760–766. – https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1456807/
- MT Streppel , LR Arends , P Van’t Veer , et al. (2005) Dietary fiber and blood pressure: a meta-analysis of randomized placebo-controlled trials. Arch Intern Med 165, 150–156.
- SP Whelton , AD Hyre , B Pedersen , et al. (2005) Effect of dietary fiber intake on blood pressure: a meta-analysis of randomized, controlled clinical trials. J Hypertens 23, 475–481.
- CE Evans , DC Greenwood , DE Threapleton , et al. (2015) Effects of dietary fibre type on blood pressure: a systematic review and meta-analysis of randomized controlled trials of healthy individuals. J Hypertens 33, 897–911.
- American Heart Association journal Stroke March 28, 2013 – Eating More Fiber May Reduce Stroke Risk – https://www.goredforwomen.org/about-heart-disease/eating-more-fiber-may-reduce-stroke-risk/
- American Heart Association Rapid Access Journal Report May 08, 2014 – Stroke News – Eating more fruits, vegetables may cut stroke risk worldwide – http://newsroom.heart.org/news/eating-more-fruits-vegetables-may-cut-stroke-risk-worldwide
- American Heart Association journal Circulation June 14, 2016, Volume 133, Issue 24: Whole Grain Intake and Mortality From All Causes, Cardiovascular Disease, and Cancer: A Meta-Analysis of Prospective Cohort Studies – https://doi.org/10.1161/CIRCULATIONAHA.115.021101; Circulation. 2016;133:2370-2380
- Huang T, Xu M, Lee A, Cho S, Qi L. Consumption of whole grains and cereal fiber and total and cause-specific mortality: prospective analysis of 367,442 individuals. BMC Med. 2015;13:59. doi: 10.1186/s12916-015-0294-7.
- Johnsen NF, Frederiksen K, Christensen J, Skeie G, Lund E, Landberg R, Johansson I, Nilsson LM, Halkjær J, Olsen A, Overvad K, Tjønneland A. Whole-grain products and whole-grain types are associated with lower all-cause and cause-specific mortality in the Scandinavian HELGA cohort. Br J Nutr. 2015;114:608–623. doi: 10.1017/S0007114515001701.
- Nutrients. 2010 Dec; 2(12): 1266–1289. – Effects of Dietary Fiber and Its Components on Metabolic Health – https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3257631/