What is hemochromatosis
Hemochromatosis is a inherited condition and the most common form of iron overload disease where there is too much iron builds up in your body (iron overload). Iron is a mineral found in many foods. Iron is important because it is part of hemoglobin, a molecule in the blood that transports oxygen from the lungs to all body tissues. However, too much iron is toxic to your body. In people with hemochromatosis, too much iron is absorbed from the gut, leading to iron overload. Too much iron (iron overload) can poison your organs and cause organ failure. Normally your body controls iron levels by absorbing just the right amount of iron from your food. It has no method of excreting excess iron – any excess is stored in organs and joints in your body. This can lead to damage to the liver, heart, joints and pancreas.
In hemochromatosis, iron can build up in most of your body’s organs, but especially in the liver, heart, joints and pancreas. Too much iron in the liver can cause an enlarged liver, liver failure, liver cancer, or cirrhosis. Cirrhosis is scarring of the liver, which causes the organ to not work well.
Too much iron in the heart can cause irregular heartbeats called arrhythmias and heart failure. Too much iron in the pancreas can lead to diabetes.
Hemochromatosis is the most common genetic disorder in America. Estimates suggest that about 1 in 10 people in the United States are hemochromatosis carriers. It is caused by an abnormal gene common in people of northern European background and is particularly common in countries where lots of people have a Celtic background. This gene is usually inherited from a parent. People who carry the abnormal gene might have haemochromatosis, or they might not.
Primary (hereditary) hemochromatosis is caused by a faulty HFE gene that affects how the body absorbs iron from food. You’re at risk of developing the condition if both of your parents have this faulty gene and you inherit one copy from each of them.
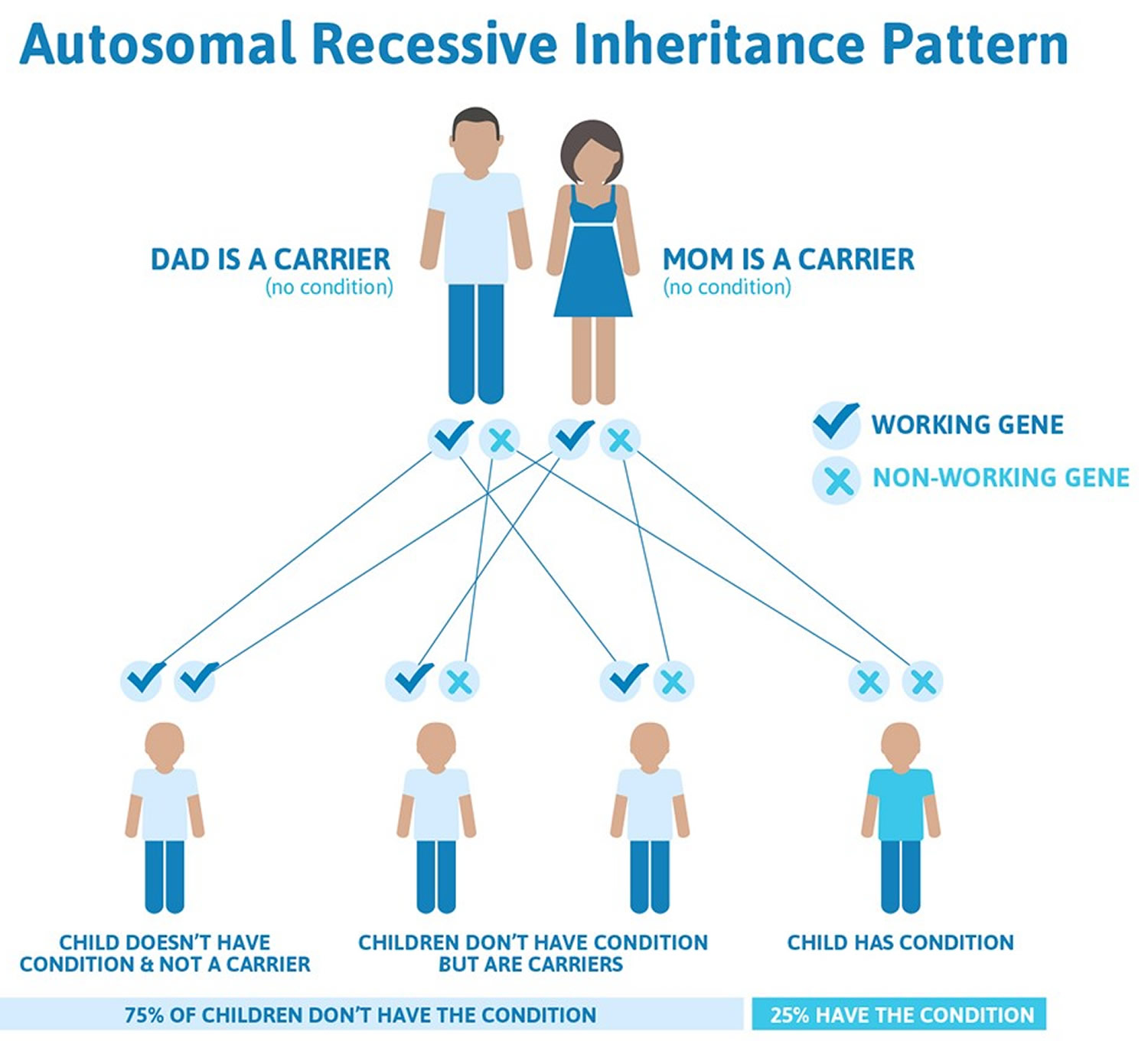
If two parents are carriers of the faulty HFE gene, then each of their children has a 1 in 4 chance of inheriting two faulty HFE genes.
You won’t get hemochromatosis if you only inherit one copy of the genetic fault, but there’s a chance you could pass the faulty gene on to any children you have.
And even if you do inherit two copies, you won’t necessarily get hemochromatosis. Only a small number of people with two copies of this genetic fault will ever develop the condition. It’s not clear exactly why this is.
Although less common, other faulty genes also can cause hemochromatosis. Researchers continue to study what changes to normal genes may cause the disease.
If someone in the family has hemochromatosis, you can have genetic testing to see if you carry the gene.
How hemochromatosis is inherited
Everyone receives two sets of genes – one from their father and one from their mother. You’re only at risk of hemochromatosis if you inherit the faulty HFE gene from both of your parents.
If you only inherit the faulty gene from one parent, you’ll be at risk of passing it on to your children – known as being a “carrier” – but you won’t develop hemochromatosis yourself.
In certain ethnic groups, such as people with a Celtic background – which is common in Ireland, Scotland and Wales – it’s quite common to be a carrier of the faulty HFE gene.
If two carriers have a baby, there’s a:
- 1 in 4 (25%) chance the baby will receive two normal versions of the HFE gene, so they won’t have hemochromatosis and won’t be a carrier
- 1 in 2 (50%) chance the baby will inherit one normal HFE gene and one faulty one, so they’ll be a carrier but won’t develop hemochromatosis
- 1 in 4 (25%) chance the baby will inherit two copies of the faulty HFE gene and will be at risk of developing hemochromatosis
But inheriting two copies of the genetic fault doesn’t mean you’ll definitely get hemochromatosis.
For unknown reasons, only a small proportion of people with two copies of the faulty HFE gene will ever develop the condition.
Symptoms of hemochromatosis
Hereditary hemochromatosis is present at birth. But, most people don’t experience signs and symptoms until later in life — usually between the ages of 50 and 60 in men and after age 60 in women. Women are more likely to develop symptoms after menopause, when they no longer lose iron with menstruation and pregnancy.
Symptoms of hemochromatosis usually start between the ages of 30 and 60.
Common symptoms include:
- feeling very tired all the time (fatigue)
- weight loss
- weakness
- joint pain
- in men, an inability to get or maintain an erection (erectile dysfunction)
- in women, irregular periods or absent periods
If hemochromatosis isn’t treated, it may even cause death.
If hemochromatosis is diagnosed and treated early on, hemochromatosis doesn’t affect life expectancy and is unlikely to result in serious problems.
But if hemochromatosis is not found until it’s more advanced, the high iron levels can damage parts of the body.
This can lead to potentially serious complications, such as:
- liver problems – including scarring of the liver (cirrhosis) or liver cancer
- diabetes – where the level of sugar in the blood becomes too high
- arthritis – pain and swelling in the joints
- heart failure – where the heart is unable to pump blood around the body properly
- loss of sex drive or erection problems
- thyroid problems
There’s currently no cure for hemochromatosis, but there are treatments that can reduce the amount of iron in the body and reduce the risk of damage.
There are two main treatments:
- Phlebotomy – a procedure to remove some of your blood; this may need to be done every week at first but can be done every few months once your iron level comes down to normal
- Chelation therapy – where you take medication to reduce the amount of iron in your body; this is only used if it’s not easy to regularly remove some of your blood
You don’t need to make any big changes to your diet to control your iron levels if you’re having treatment, but you’ll usually be advised to avoid:
- breakfast cereals containing added iron
- iron or vitamin C supplements
- drinking too much alcohol
Hemochromatosis Types
There are 4 types of hemochromatosis: primary, secondary, juvenile and neonatal hemochromatosis.
- Primary hemochromatosis (Hereditary hemochromatosis) is caused by a defect in the genes that control how much iron you absorb from food.
- Secondary hemochromatosis usually is the result of another disease or condition that causes iron overload.
- Neonatal hemochromatosis is a rare disease characterized by liver failure and death in fetuses and newborns.
- Juvenile hemochromatosis. This causes the same problems in young people that hereditary hemochromatosis causes in adults. But iron accumulation begins much earlier, and symptoms usually appear between the ages of 15 and 30. This disorder is caused by mutations in the hemojuvelin or hepcidin genes.
Most people who have primary hemochromatosis inherit it from their parents. If you inherit two hemochromatosis genes—one from each parent—you’re at risk for iron overload and signs and symptoms of the disease. The two faulty genes cause your body to absorb more iron than usual from the foods you eat.
Hemochromatosis is one of the most common genetic disorders in the United States. However, not everyone who has hemochromatosis has signs or symptoms of the disease.
Estimates of how many people develop signs and symptoms vary greatly. Some estimates suggest that as many as half of all people who have the disease don’t have signs or symptoms.
The severity of hemochromatosis also varies. Some people don’t have complications, even with high levels of iron in their bodies. Others have severe complications or die from the disease.
Certain factors can affect the severity of the disease. For example, a high intake of vitamin C can make hemochromatosis worse. This is because vitamin C helps your body absorb iron from food.
Alcohol use can worsen liver damage and cirrhosis caused by hemochromatosis. Conditions such as hepatitis also can further damage or weaken the liver.
Other names for hemochromatosis
- Acquired hemochromatosis
- Classical hemochromatosis
- Genetic hemochromatosis
- Genetic iron poisoning
- Hereditary hemochromatosis
- Iron overload disease
- Primary hemochromatosis
- Secondary hemochromatosis
- bronze diabetes
- bronzed cirrhosis
- familial hemochromatosis
- genetic hemochromatosis
- haemochromatosis
- HC
- hemochromatosis
- hereditary haemochromatosis
- HH
- HLAH
- iron storage disorder
- pigmentary cirrhosis
- Troisier-Hanot-Chauffard syndrome
- Von Recklenhausen-Applebaum disease
The Liver
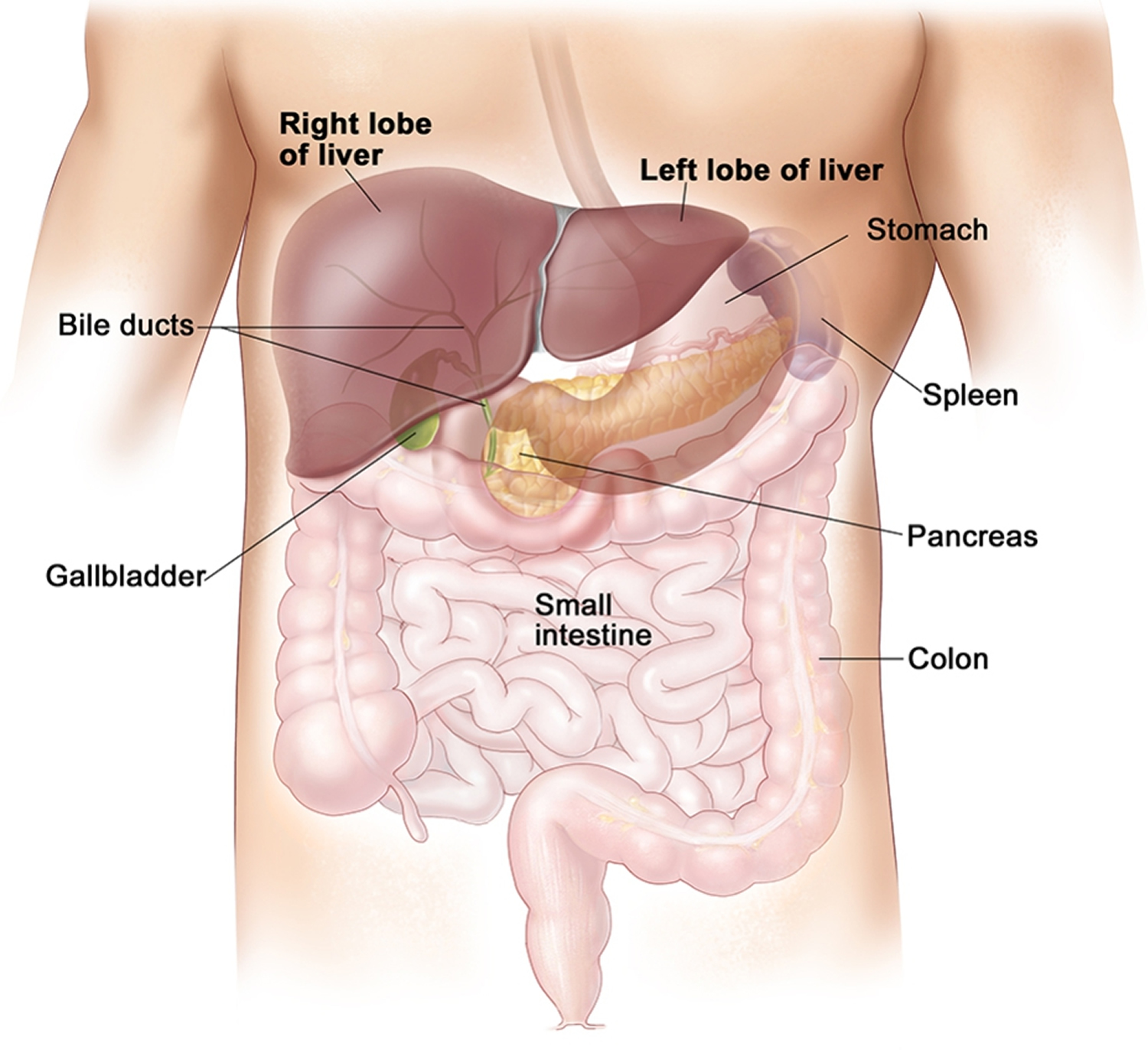
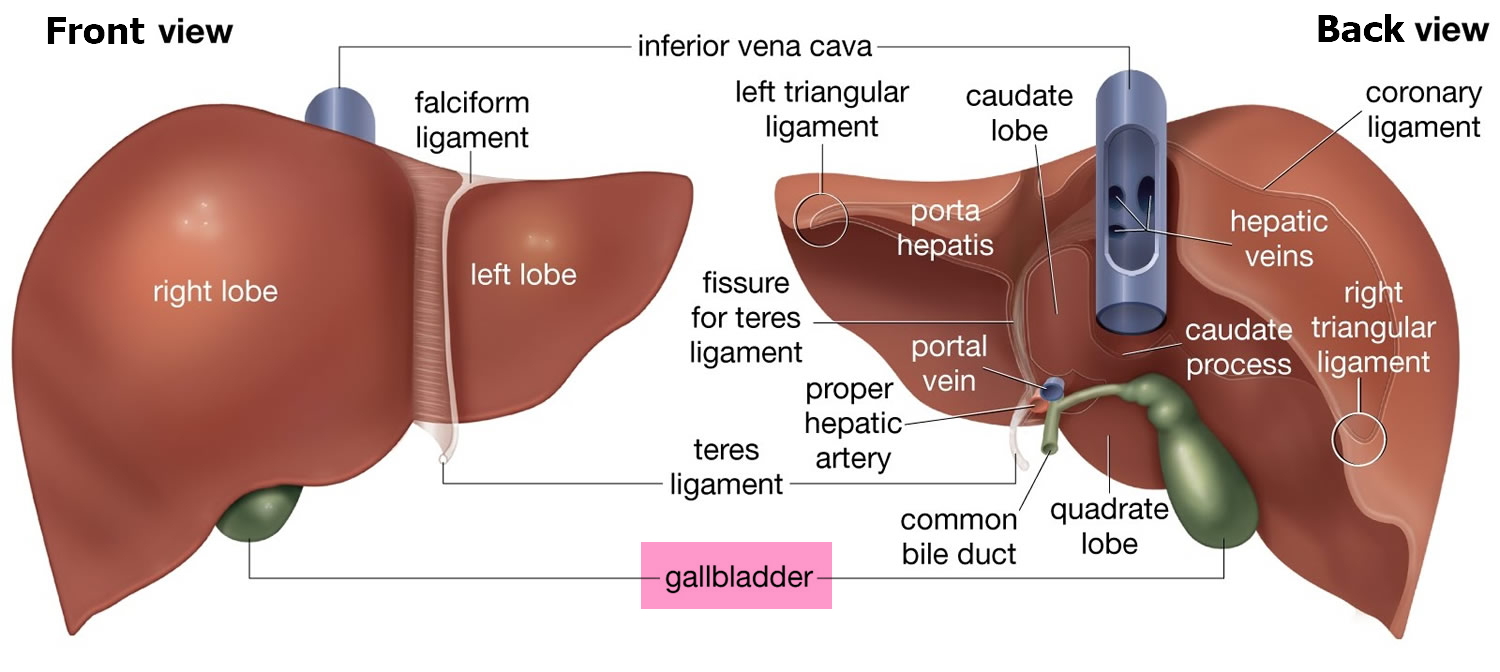
Your liver is the largest organ inside your body, weighing about 1.4 kg (3 pounds) in an average adult. The liver is in the right upper quadrant of the abdominal cavity, just inferior to the diaphragm in the right superior part of the abdominal cavity and under your right ribs just beneath your right lung – filling much of the right hypochondriac and epigastric regions and extending into the left hypochondriac region. The liver is partially surrounded by the ribs, and extends from the level of the fifth intercostal space to the lower margin of the right rib cage, which protects this highly vascular organ from blows that could rupture it. The liver is shaped like a wedge, the wide base of which faces right and the narrow apex of which lies just inferior to the level of the left nipple. The reddish-brown liver is well supplied with blood vessels.
Liver functions
Amazingly versatile, your liver performs over 500 functions. Its digestive function is to produce bile, a green alkaline liquid that is stored in the gallbladder and secreted into the duodenum. Bile salts emulsify fats in the small intestine; that is, they break up fatty nutrients into tiny particles, just as dish detergent breaks up a pool of fat drippings in a roasting pan. These smaller particles are more accessible to digestive enzymes from the pancreas. The liver also performs many metabolic functions and you cannot live without your liver:
- Picks up glucose from nutrient-rich blood returning from the alimentary canal and stores this carbohydrate as glycogen for subsequent use by the body.
- Processes fats and amino acids and stores certain vitamins.
- Detoxifies many poisons and drugs in the blood.
- Makes the blood proteins.
- It breaks down and stores many of the nutrients absorbed from the intestine that your body needs to function. Some nutrients must be changed (metabolized) in the liver before they can be used for energy or to build and repair body tissues.
- It makes most of the clotting factors that keep you from bleeding too much when you are cut or injured.
- It secretes bile into the intestines to help absorb nutrients (especially fats).
- It breaks down alcohol, drugs, and toxic wastes in the blood, which then pass from the body through urine and stool.
Almost all of these functions are carried out by a type of cell called a hepatocyte or simply a liver cell.
The liver carries on many important metabolic activities. The liver plays a key role in carbohydrate metabolism by helping maintain concentration of blood glucose within the normal range. Liver cells responding to the hormone insulin lower the blood glucose level by polymerizing glucose to glycogen. Liver cells responding to the hormone glucagon raise the blood glucose level by breaking down glycogen to glucose or by converting noncarbohydrates into glucose.
The liver’s effects on lipid metabolism include oxidizing (breaking down) fatty acids at an especially high rate; synthesizing lipoproteins, phospholipids, and cholesterol; and converting excess portions of carbohydrate molecules into fat molecules. The blood transports fats synthesized in the liver to adipose tissue for storage.
Other liver functions concern protein metabolism. They include deaminating amino acids; forming urea; synthesizing plasma proteins such as clotting factors; and converting certain amino acids into other amino acids.
The liver also stores many substances, including glycogen, iron, and vitamins A, D, and B12. In addition, macrophages in the liver help destroy damaged red blood cells and phagocytize foreign antigens. The liver also removes toxic substances such as alcohol and certain drugs from blood (detoxification).
Figure 1. Location of the human liver
Figure 2. Liver anatomy
Hereditary hemochromatosis
Hereditary hemochromatosis is a disorder that causes the body to absorb too much iron from the diet. The excess iron is stored in the body’s tissues and organs, particularly the skin, heart, liver, pancreas, and joints. Because humans cannot increase the excretion of iron, excess iron can overload and eventually damage tissues and organs. For this reason, hereditary hemochromatosis is also called an iron overload disorder.
Early symptoms of hereditary hemochromatosis are nonspecific and may include fatigue, joint pain, abdominal pain, and loss of sex drive. Later signs and symptoms can include arthritis, liver disease, diabetes, heart abnormalities, and skin discoloration. The appearance and progression of symptoms can be affected by environmental and lifestyle factors such as the amount of iron in the diet, alcohol use, and infections.
Hereditary hemochromatosis is classified by type depending on the age of onset and other factors such as genetic cause and mode of inheritance. Type 1, the most common form of the disorder, and type 4 (also called ferroportin disease) begin in adulthood. Men with type 1 or type 4 hemochromatosis typically develop symptoms between the ages of 40 and 60, and women usually develop symptoms after menopause.
Type 1 hemochromatosis is one of the most common genetic disorders in the United States, affecting about 1 million people. It most often affects people of Northern European descent. The other types of hemochromatosis are considered rare and have been studied in only a small number of families worldwide.
Type 2 hemochromatosis is a juvenile-onset disorder. Iron accumulation begins early in life, and symptoms may appear in childhood. By age 20, decreased or absent secretion of sex hormones is evident. Females usually begin menstruation in a normal manner, but menses stop after a few years. Males may experience delayed puberty or symptoms related to a shortage of sex hormones. If the disorder is untreated, heart disease becomes evident by age 30.
The onset of type 3 hemochromatosis is usually intermediate between types 1 and 2. Symptoms of type 3 hemochromatosis generally begin before age 30.
Hemochromatosis gene
Mutations in several genes, including HAMP, HFE, HJV, SLC40A1, and TFR2, can cause hereditary hemochromatosis. Type 1 hemochromatosis results from mutations in the HFE gene, and type 2 hemochromatosis results from mutations in either the HJV or HAMP gene. Mutations in the TFR2 gene cause type 3 hemochromatosis, and mutations in the SLC40A1 gene cause type 4 hemochromatosis.
The proteins produced from these genes play important roles in regulating the absorption, transport, and storage of iron. Mutations in any of these genes impair the control of iron absorption during digestion and alter the distribution of iron to other parts of the body. As a result, iron accumulates in tissues and organs, which can disrupt their normal functions.
Types 1, 2, and 3 hemochromatosis are inherited in an autosomal recessive pattern, which means both copies of the gene in each cell have mutations. Most often, the parents of an individual with an autosomal recessive condition each carry one copy of the mutated gene but do not show signs and symptoms of the condition.
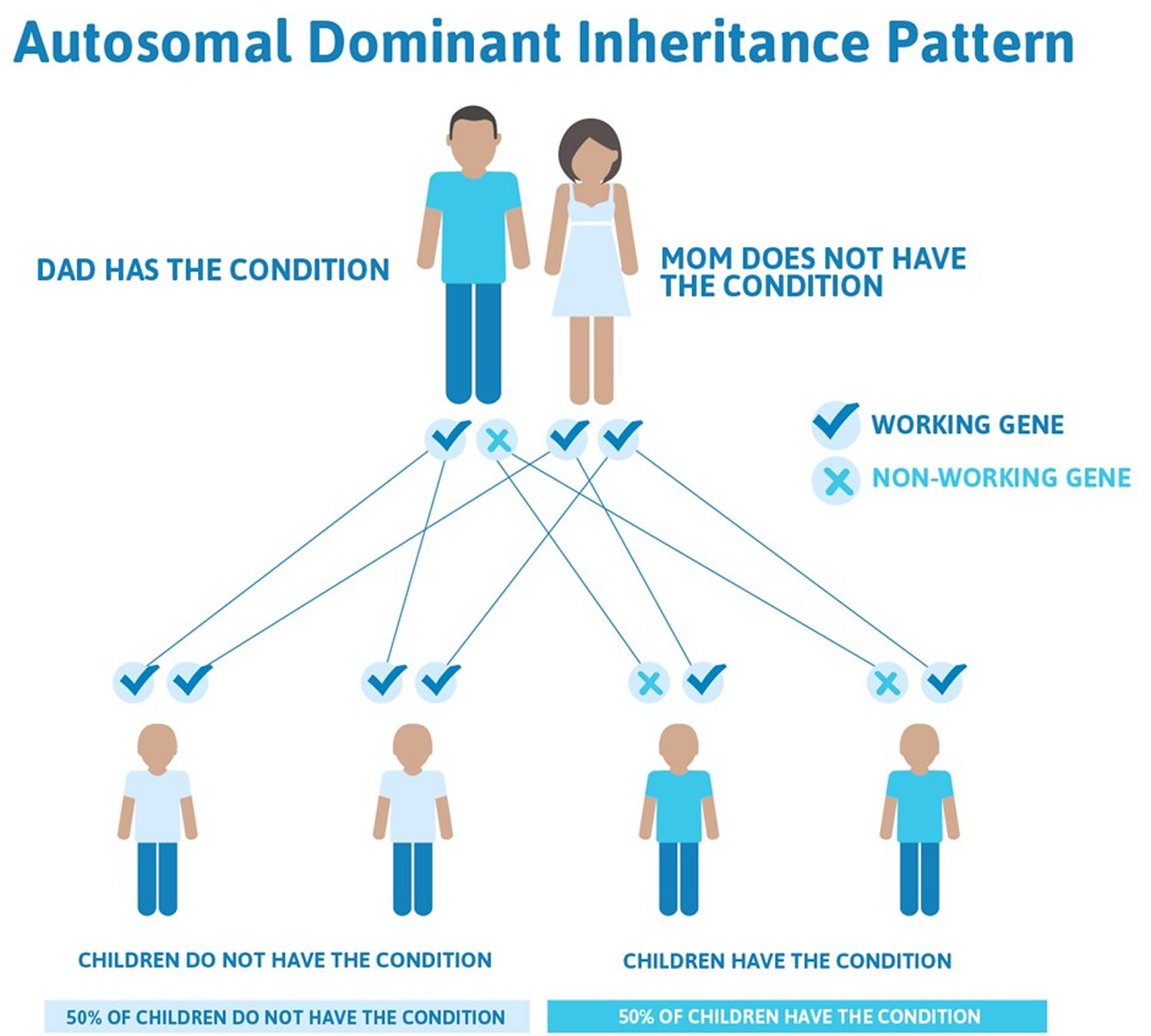
Type 4 hemochromatosis is distinguished by its autosomal dominant inheritance pattern. With this type of inheritance, one copy of the altered gene in each cell is sufficient to cause the disorder. In most cases, an affected person has one parent with the condition.
Figure 3. Hemochromatosis types 1,2 and 3 are inherited in an autosomal Recessive inheritance pattern
Figure 4. Hemochromatosis type 4 are inherited in an autosomal Dominant inheritance pattern
Hemochromatosis causes
Hemochromatosis is caused by a genetic fault that can be passed on to a child by his or her parents.
Most cases are linked to a fault in a gene called HFE, which affects your ability to absorb iron from food.
Normally, your body maintains a steady level of iron. The amount of iron absorbed from food varies according to your body’s need for it.
But people with hemochromatosis can’t control their iron level. The level gradually increases over time and iron starts to build up in their organs, damaging them in the process.
Primary Hemochromatosis
Primary hemochromatosis is caused by a defect in the genes that control how much iron you absorb from food. This form of the disease sometimes is called hereditary or classical hemochromatosis. Primary hemochromatosis is more common than the secondary form of the disease.
The genes usually involved in primary hemochromatosis are called HFE genes. Faulty HFE genes cause the body to absorb too much iron and are associated with up to 90 percent of cases 1. The HFE gene helps regulate the amount of iron absorbed from food. The two known mutations of HFE are C282Y and H63D. C282Y defects are the most common cause of primary hemochromatosis.
If you inherit two copies of the faulty HFE gene (one from each parent), you’re at risk for iron overload and signs and symptoms of hemochromatosis.
If you inherit one faulty HFE gene and one normal HFE gene, you’re a hemochromatosis “carrier.” Carriers usually don’t develop the disease. However, they can pass the faulty gene on to their children.
People inherit two copies of the HFE gene—one copy from each parent. Most people who inherit two copies of the HFE gene with the C282Y defect will have higher-than-average iron absorption. However, not all of these people will develop health problems associated with hemochromatosis. One recent study found that 31 percent of people with two copies of the C282Y defect developed health problems by their early fifties 2. Men who develop health problems from HFE defects typically develop them after age 40 1. Women who develop health problems from HFE defects typically develop them after menopause 1.
People who inherit two H63D defects or one C282Y and one H63D defect may have higher-than-average iron absorption 3. However, they are unlikely to develop iron overload and organ damage.
Rare defects in other genes may also cause primary hemochromatosis. Mutations in the hemojuvelin or hepcidin genes cause juvenile hemochromatosis, a type of primary hemochromatosis. People with juvenile hemochromatosis typically develop severe iron overload and liver and heart damage between ages 15 and 30.
Secondary Hemochromatosis
Hemochromatosis that is not inherited is called secondary hemochromatosis. The most common cause of secondary hemochromatosis is frequent blood transfusions in people with severe anemia. Anemia is a condition in which red blood cells are fewer or smaller than normal, which means they carry less oxygen to the body’s cells.
Types of anemia that may require frequent blood transfusions include:
- congenital, or inherited, anemias such as sickle cell disease, thalassemia, sideroblastic anemia and Fanconi’s syndrome
- Atransferrinemia and aceruloplasminemia—both are rare, inherited diseases
- severe acquired anemias, which are not inherited, such as aplastic anemia and autoimmune hemolytic anemia
Other factors also can cause secondary hemochromatosis, including:
- Blood transfusions
- Oral iron pills or iron injections, with or without very high vitamin C intake (vitamin C helps your body absorb iron)
- Long-term kidney dialysis
Liver diseases—such as alcoholic liver disease, nonalcoholic steatohepatitis, and chronic hepatitis C infection—may cause mild iron overload. However, this iron overload causes much less liver damage than the underlying liver disease causes.
Neonatal Hemochromatosis
Neonatal hemochromatosis is a rare disease characterized by liver failure and death in fetuses and newborns. Researchers are studying the causes of neonatal hemochromatosis and believe more than one factor may lead to the disease.
Experts previously considered neonatal hemochromatosis a type of primary hemochromatosis. However, recent studies suggest genetic defects that increase iron absorption do not cause this disease. Instead, the mother’s immune system may produce antibodies—proteins made by the immune system to protect the body from foreign substances such as bacteria or viruses—that damage the liver of the fetus. Women who have had one child with neonatal hemochromatosis are at risk for having more children with the disease 4. Treating these women during pregnancy with intravenous (IV) immunoglobulin—a solution of antibodies from healthy people—can prevent fetal liver damage 4.
Researchers supported by the National Institute of Diabetes and Digestive and Kidney Diseases recently found that a combination of exchange transfusion—removing blood and replacing it with donor blood—and IV immunoglobulin is an effective treatment for babies born with neonatal hemochromatosis 5.
Who is more likely to develop hemochromatosis?
Primary hemochromatosis mainly affects Caucasians of Northern European descent. This disease is one of the most common genetic disorders in the United States. About four to five out of every 1,000 Caucasians carry two copies of the C282Y mutation of the HFE gene and are susceptible to developing hemochromatosis 1. About one out of every 10 Caucasians carries one copy of C282Y 1.
Hemochromatosis is extremely rare in African Americans, Asian Americans, Hispanics/Latinos, and American Indians. HFE mutations are usually not the cause of hemochromatosis in these populations.
Both men and women can inherit the gene defects for hemochromatosis; however, not all will develop the symptoms of hemochromatosis. Men usually develop symptoms at a younger age than women. Women lose blood—which contains iron—regularly during menstruation; therefore, women with the gene defects that cause hemochromatosis may not develop iron overload and related symptoms and complications until after menopause.
Hemochromatosis prevention
You can’t prevent primary, or inherited, hemochromatosis. However, not everyone who inherits hemochromatosis genes develops symptoms or complications of the disease. In those who do, treatments can keep the disease from getting worse.
Treatments include therapeutic phlebotomy, iron chelation therapy, dietary changes, and other treatments.
People who have hemochromatosis (or a family history of it) and are planning to have children may want to consider genetic testing and counseling. Testing will help show whether one or both parents have faulty HFE genes. A genetic counselor also can help figure out the likelihood of the parents passing the faulty genes on to their children.
Hemochromatosis prognosis (outlook)
The outlook for people who have hemochromatosis largely depends on how much organ damage they have at the time of diagnosis. Early diagnosis and treatment of the disease are important.
Treatment may help prevent, delay, or sometimes reverse complications of the disease. Treatment also may lead to better quality of life.
For people who are diagnosed and treated early, a normal lifespan is possible. If left untreated, hemochromatosis can lead to severe organ damage and even death.
Hemochromatosis complications
If hemochromatosis isn’t diagnosed and treated early on, iron can build up in the body and cause serious problems.
Without treatment, iron may build up in the organs and cause complications, including:
- Cirrhosis, or scarring of liver tissue
- Diabetes
- Irregular heart rhythms or weakening of the heart muscle
- Erectile dysfunction
- Joint damage and pain, including arthritis
- Reproductive organ failure, such as erectile dysfunction (impotence), shrinkage of the testicles, and loss of sex drive in men, and absence of the menstrual cycle and early menopause in women
- Changes in skin color that make the skin look gray or bronze
- Underactive pituitary and thyroid glands
- Damage to the adrenal glands
The complication most often associated with hemochromatosis is liver damage. Iron buildup in the liver causes cirrhosis, which increases the chance of developing liver cancer.
For some people, complications may be the first sign of hemochromatosis. However, not everyone with hemochromatosis will develop complications.
Some of the main complications associated with the condition are outlined below.
Liver damage
The liver can be very sensitive to the effects of iron, and many people with hemochromatosis will have some degree of liver damage.
This often won’t cause any obvious symptoms at first, but can be picked up during tests for hemochromatosis.
If significant scarring of the liver (cirrhosis) occurs, you may experience:
- tiredness and weakness
- loss of appetite
- weight loss
- feeling sick
- very itchy skin
- tenderness or pain around the liver
- yellowing of the eyes and skin (jaundice)
Cirrhosis also increases your risk of developing liver cancer.
Surgery and medication can help relieve symptoms of cirrhosis, but the only way to achieve a complete cure is to have a liver transplant.
Diabetes
Diabetes is a condition in which a person’s blood sugar level becomes too high. It can occur in people with hemochromatosis if high levels of iron damage the pancreas.
The pancreas is an organ that produces insulin. Insulin is a hormone that’s used to change sugar (glucose) from your diet into energy.
If the pancreas is damaged, it may not produce enough insulin, which can lead to an increase in the level of sugar in the blood.
Symptoms can include:
- needing to urinate more often than usual, particularly at night
- feeling very thirsty
- feeling very tired
Lifestyle changes such as eating healthily and exercising regularly can help, although some people need to take medication to control their blood sugar level.
Arthritis
In severe and advanced cases of hemochromatosis, the high levels of iron can damage the joints. This is known as arthritis.
The main symptoms of arthritis are:
- joint pain
- stiff joints
- swelling (inflammation) in the joints
It may be possible to relieve the symptoms with painkillers and steroid medication.
But if significant damage has occurred, it may be necessary to replace the affected joint with an artificial one, such as a hip replacement or knee replacement.
Heart problems
If excess iron builds up in the heart, it can damage the muscles of the heart (cardiomyopathy).
This can lead to heart failure, which is where the heart has become so damaged it struggles to pump blood around the body properly.
Symptoms of heart failure include:
- shortness of breath
- extreme tiredness and weakness
- swelling in the legs, ankles and feet (edema)
Heart failure can usually be treated with medication.
Hemochromatosis symptoms
Symptoms of hemochromatosis usually begin between the ages of 30 and 60, although they can occasionally occur earlier.
The symptoms tend to develop earlier in men than in women. Women often don’t experience problems until after the menopause.
Sometimes there are no symptoms and the condition is only found during a blood test.
Early symptoms
Initial symptoms of hemochromatosis can include:
- feeling very tired all the time (fatigue)
- weight loss
- weakness
- joint pain
- in men, an inability to get or maintain an erection (erectile dysfunction)
- in women, irregular periods or absent periods
These symptoms can have many different causes and may sometimes just be put down to getting older.
Later problems
As the condition progresses, it can also cause problems such as:
- loss of sex drive (libido)
- darkening of the skin – you may look permanently tanned
- tummy (abdominal) pain and swelling
- yellowing of the skin and eyes (jaundice)
- feeling thirsty all the time and needing to pee frequently
- severe pain and stiffness in your joints, particularly in the fingers
- chest pain
- shortness of breath
- swelling of your hands and feet
- an irregular heartbeat (arrhythmia)
- in men, the testicles getting smaller
These problems are often caused by complications of hemochromatosis that can occur if the condition isn’t treated early on.
Hemochromatosis diagnosis
Hemochromatosis can usually be diagnosed with blood tests.
However, hemochromatosis is rare, and health care providers may not think to test for this disease. Thus, the disease is often not diagnosed or treated. The initial symptoms can be diverse, vague, and similar to the symptoms of many other diseases. Health care providers may focus on the symptoms and complications caused by hemochromatosis rather than on the underlying iron overload. Moreover, if a health care provider diagnoses and treats the iron overload caused by hemochromatosis before organ damage has occurred, a person can live a normal, healthy life.
Speak to your doctor about getting tested if:
- you have persistent symptoms of hemochromatosis – these symptoms can have a number of causes, and your doctor may want to rule out some of these before arranging a blood test
- a parent or sibling has been diagnosed with hemochromatosis – even if you don’t have any symptoms, you may be at risk of developing the condition at some point
The tests you may have are outlined below.
Blood tests
Several blood tests are needed to diagnose hemochromatosis.
You’ll have tests to check:
- the amount of iron in your blood – known as your transferrin saturation level. The transferrin saturation test shows how much iron is bound to the protein that carries iron in the blood. Transferrin saturation values above or equal to 45 percent are considered abnormal.
- the amount of iron stored in your body – known as your serum ferritin level. The serum ferritin test detects the amount of ferritin—a protein that stores iron—in the blood. Levels above 300 μg/L in men and 200 μg/L in women are considered abnormal. Levels above 1,000 μg/L in men or women indicate a high chance of iron overload and organ damage.
These tests will help show if you have hemochromatosis, if you’re a carrier of a genetic fault linked to the condition, or if you might have another condition that causes high iron levels.
If either test shows higher-than-average levels of iron in the body, health care providers can order a special blood test that can detect two copies of the C282Y mutation to confirm the diagnosis. If the mutation is not present, health care providers will look for other causes.
You might need any further tests or treatment.
Further tests
If blood tests show that you have hemochromatosis, you may need to have some further tests to check if the condition has caused any organ damage, particularly damage to your liver.
These tests may include:
- a blood test to check for substances that indicate a problem with your liver
- a liver biopsy – where a needle is used to remove a small sample of liver tissue under local anesthetic so it can be checked for signs of damage
- a magnetic resonance imaging (MRI) scan to check for iron in your liver and look for signs of liver damage
Liver damage is one of the main complications of hemochromatosis.
Liver biopsy. Health care providers may perform a liver biopsy, a procedure that involves taking a piece of liver tissue for examination with a microscope for signs of damage or disease. The health care provider may ask the patient to temporarily stop taking certain medications before the liver biopsy. The health care provider may ask the patient to fast for 8 hours before the procedure.
During the procedure, the patient lies on a table, right hand resting above the head. The health care provider applies a local anesthetic to the area where he or she will insert the biopsy needle. If needed, a health care provider will also give sedatives and pain medication. The health care provider uses a needle to take a small piece of liver tissue. He or she may use ultrasound, computerized tomography scans, or other imaging techniques to guide the needle. After the biopsy, the patient must lie on the right side for up to 2 hours and is monitored an additional 2 to 4 hours before being sent home.
A health care provider performs a liver biopsy at a hospital or an outpatient center. The health care provider sends the liver sample to a pathology lab where the pathologist—a doctor who specializes in diagnosing disease—looks at the tissue with a microscope and sends a report to the patient’s health care provider. The biopsy shows how much iron has accumulated in the liver and whether the patient has liver damage.
Other causes of high iron levels
A high level of iron in the body can have several causes other than hemochromatosis, including:
- long-term liver disease
- conditions that need frequent blood transfusions, such as sickle cell disease or thalassemia
- drinking beer brewed in iron containers
- excess iron intake from supplements or injections
- long-term dialysis, a treatment that replicates some of the functions of the kidneys
- rare inherited conditions that affect red blood cells, the proteins that transport iron (such as atransferrinaemia) or where iron collects in the body (such as aceruloplasminaemia)
Who should be tested for hemochromatosis?
Experts recommend testing for hemochromatosis in people who have symptoms, complications, or a family history of the disease.
Some researchers have suggested widespread screening for the C282Y mutation in the general population. However, screening is not cost-effective. Although the C282Y mutation occurs quite frequently, the disease caused by the mutation is rare, and many people with two copies of the mutation never develop iron overload or organ damage.
Researchers and public health officials suggest the following:
- Siblings of people who have hemochromatosis should have their blood tested to see if they have the C282Y mutation.
- Parents, children, and other close relatives of people who have hemochromatosis should consider being tested.
- Health care providers should consider testing people who have severe and continuing fatigue, unexplained cirrhosis, joint pain or arthritis, heart problems, erectile dysfunction, or diabetes because these health issues may result from hemochromatosis.
Hemochromatosis treatment
There’s currently no cure for hemochromatosis, but there are treatments that can reduce the amount of iron in your body.
This can help relieve some of the symptoms and reduce the risk of damage to organs such as the heart, liver and pancreas.
The main treatments and things you can do are outlined below.
Phlebotomy (venesection)
The most commonly used treatment for hemochromatosis is a procedure to remove some of your blood, known as a phlebotomy or venesection. Phlebotomy rids the body of extra iron. This treatment is simple, inexpensive, and safe.
The procedure is similar to giving blood. You lie back in a chair and a needle is used to drain a small amount of blood – usually about 500ml, or just less than a pint – from a vein in your arm.
The removed blood includes red blood cells that contain iron, and your body will use up more iron to replace them, helping to reduce the amount of iron in your body.
Health care providers will test serum ferritin levels periodically to monitor iron levels. The goal is to bring serum ferritin levels to the low end of the average range and keep them there. Depending on the lab, the level is 25 to 50 μg/L.
There are two main stages to treatment:
- induction – blood is removed on a frequent basis (usually weekly) until your iron levels are normal; this can sometimes take up to a year or more
- maintenance – blood is removed less often (usually every two or three months) to keep your iron levels under control; this is usually needed for the rest of your life.
After phlebotomy reduces serum ferritin levels to the desired level, patients may need maintenance phlebotomy treatment every few months. Some patients may need phlebotomies more often. Serum ferritin tests every 6 months or once a year will help determine how often a patient should have blood drawn. Many blood donation centers provide free phlebotomy treatment for people with hemochromatosis.
Treating hemochromatosis before organs are damaged can prevent complications such as cirrhosis, heart problems, arthritis, and diabetes. Treatment cannot cure these conditions in patients who already have them at diagnosis. However, treatment will help most of these conditions improve. The treatment’s effectiveness depends on the degree of organ damage. For example, treating hemochromatosis can stop the progression of liver damage in its early stages and lead to a normal life expectancy. However, if a patient develops cirrhosis, his or her chance of developing liver cancer increases, even with phlebotomy treatment. Arthritis usually does not improve even after phlebotomy removes extra iron.
Chelation therapy
A treatment called chelation therapy may be used in a small number of cases where regular phlebotomies aren’t possible because it’s difficult to remove blood regularly – for example, if you have very thin or fragile veins.
This involves taking medication that removes iron from your blood and releases it into your urine or poo.
A commonly used medication is deferasirox. It comes as a tablet that’s usually taken once a day.
It’s unlicensed for the treatment of hemochromatosis, which means it hasn’t undergone extensive clinical trials for this use. But your doctor may recommend it if they feel the possible benefits outweigh any risks.
Hemochromatosis diet and alcohol
You don’t need to make any big changes to your diet, such as avoiding all foods containing iron, if you have hemochromatosis.
This is unlikely to be of much extra help if you’re having one of the treatments above, and could mean you don’t get all the nutrition you need.
Given that people with hemochromatosis absorb much more iron from the food they eat compared with healthy people. People with hemochromatosis can help prevent iron overload by:
- have a generally healthy, balanced diet
- avoid breakfast cereals that have been “fortified” with extra iron
- be careful not to eat raw oysters and clams – these may contain a type of bacteria that can cause serious infections in people with high iron levels
- eating only moderate amounts of iron-rich foods, such as red meat and organ meat
- avoiding supplements that contain iron
- avoiding supplements that contain vitamin C, which increases iron absorption
People with hemochromatosis can take steps to help prevent liver damage, including:
- limiting the amount of alcoholic beverages they drink because alcohol increases their chance of cirrhosis and liver cancer
- avoiding alcoholic beverages entirely if your liver is damaged.
- Bacon BR, Adams PC, Kowdley KV, Powell LW, Tavill AS. Diagnosis and management of hemochromatosis: 2011 practice guideline by the American Association for the Study of Liver Diseases. Hepatology. 2011;54(1):328–343.
- Aguilar-Martinez P, Bismuth M, Blanc F, et al. The Southern French registry of genetic hemochromatosis: a tool for determining clinical prevalence of the disorder and genotype penetrance. Haematologica. 2010;95(4):551–556.
- Aranda N, Viteri FE, Montserrat C, Arija V. Effects of C282Y, H63D, and S65C HFE gene mutations, diet, and life-style factors on iron status in the general Mediterranean population from Tarragona, Spain. Annals of Hematology. 2010;89(8):767–773.
- Whitington PF, Kelly S. Outcome of pregnancies at risk for neonatal hemochromatosis is improved by treatment with high-dose intravenous immunoglobulin. Pediatrics. 2008;121(6):e1615–e1621.
- Rand EB, Karpen SJ, Kelly S, et al. Treatment of neonatal hemochromatosis with exchange transfusion and intravenous immunoglobulin. The Journal of Pediatrics. 2009;155(4):566–571.