What is gelatin
Gelatin is a protein derived from collagen and it is obtained principally from cow and pig bones and hides and fish skin 1. Gelatin or gelatine is a translucent, colorless, brittle (when dry), flavorless. Gelatin is commonly used as a gelling agent in food, pharmaceutical drugs, vitamin capsules, photography, and cosmetic manufacturing. It is a common ingredient in foods such as jellies, sweets, yogurt and frozen desserts. It is also found in lunch meats, and is used extensively as clarifying agents in wine, juices and other beverages. Bovine and porcine gelatins, in particular, also have numerous applications throughout the pharmaceutical industry as integral components in drug capsules, plasma expanders, and stabilizers in vaccines, including MMR (measles, mumps and rubella), varicella (chicken pox), yellow fever, rabies, and some influenza vaccines. Severe allergic reactions, including anaphylaxis, have been reported following intravenous administration of modified fluid gelatins as plasma substitutes 2.
The earliest commercial production of gelatin appears to have been in Holland around 1685, followed shortly thereafter in England about 1700. The first commercial production of gelatin in the United States was in Massachusetts in 1808.
Substances containing gelatin or functioning in a similar way are called “gelatinous”. Gelatin is an irreversibly hydrolyzed form of collagen, wherein the hydrolysis results in the reduction of protein fibers into smaller peptides, which will have broad molecular weight ranges associated with physical and chemical methods of denaturation, based on the process of hydrolysis. It is found in most gummy candy, as well as other products such as marshmallows, gelatin desserts, and some ice creams, dips, and yogurts 3. Gelatin for cooking, foods and baking use comes in the form of sheets, granules, or powder. Instant types can be added to the food as they are; others need to be soaked in water before hand.
Figure 1. Gelatin

Where does gelatin come from ?
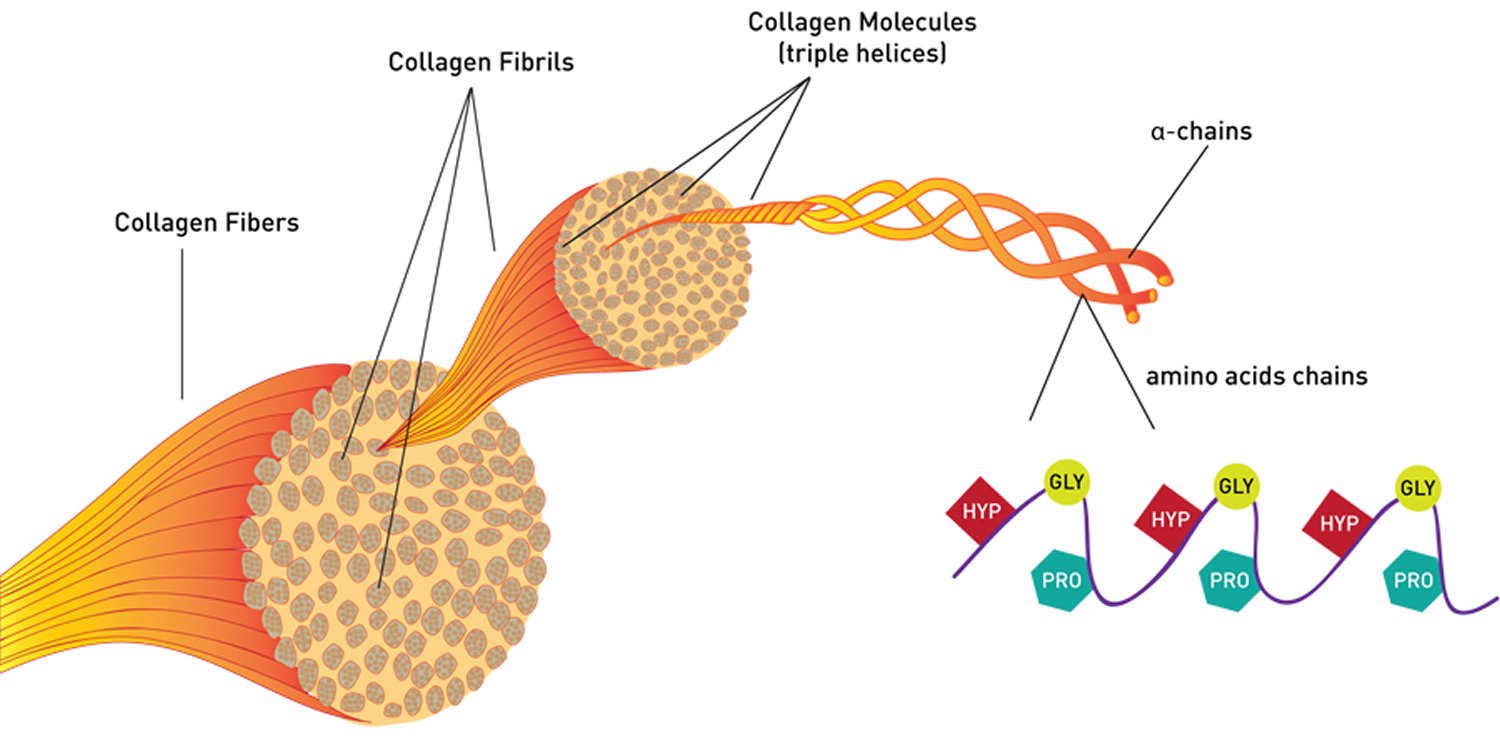
Gelatin is a mixture of peptides and proteins produced by partial hydrolysis of collagen extracted from the skin, bones, and connective tissues of animals such as domesticated cattle, chicken, pigs, and fish. During hydrolysis, the natural molecular bonds between individual collagen strands are broken down into a form that rearranges more easily. Its chemical composition is, in many aspects, closely similar to that of its parent collagen 4. Photographic and pharmaceutical grades of gelatin generally are sourced from cattle bones and pig skin. Gelatin has proline, hydroxyproline and glycine in its polypeptide chain. Glycine is responsible for close packing of the chains. Presence of proline restricts the conformation. This is important for gelation properties of gelatin 5.
Figure 2. Collagen chemical structure
Note: HYP= Hydroxyproline; GLY= Glycine; PRO= Proline. Every third amino acid residue is a Glycine residue, a small amino acid that fits perfectly inside the helix. Many of the remaining positions in the chain are filled by two amino acids: proline and a modified version of proline, hydroxyproline.
Gelatin readily dissolves in hot water and sets to a gel on cooling. When added directly to cold water, it does not dissolve well, however. Gelatin also is soluble in most polar solvents 5. Gelatin solutions show visco-elastic flow and streaming birefringence. Solubility is determined by the method of manufacture. Typically, gelatin can be dispersed in a relatively concentrated acid. Such dispersions are stable for 10–15 days with little or no chemical changes and are suitable for coating purposes or for extrusion into a precipitating bath.
The mechanical properties of gelatin gels are very sensitive to temperature variations, the previous thermal history of the gels, and the amount of time elapsing. These gels exist over only a small temperature range, the upper limit being the melting point of the gel, which depends on gelatin grade and concentration, but typically, is less than 35 °C (95 °F) and the lower limit the freezing point at which ice crystallizes. The upper melting point is below human body temperature, a factor that is important for mouthfeel of foods produced with gelatin. The viscosity of the gelatin-water mixture is greatest when the gelatin concentration is high and the mixture is kept cool at about 4 °C (39 °F). The gel strength is quantified using the Bloom test. Gelatin powder is very stable and can be stored in air-tight containers for years with no loss in quality.
Bloom test is used to measure the strength of a gel or gelatin 6. The test determines the weight in grams needed by a specified plunger (normally with a diameter of 0.5 inch) to depress the surface of the gel by 4 mm without breaking it at a specified temperature 7. The number of grams is called the Bloom value, and most gelatins are between 30 and 300 g Bloom. The higher a Bloom value, the higher the melting and gelling points of a gel, and the shorter its gelling times 7. This method is most often used on soft gels. To perform the Bloom test on gelatin, a 6.67% gelatin solution is kept for 17–18 hours at 10 °C prior to being tested.
How is gelatin made ?
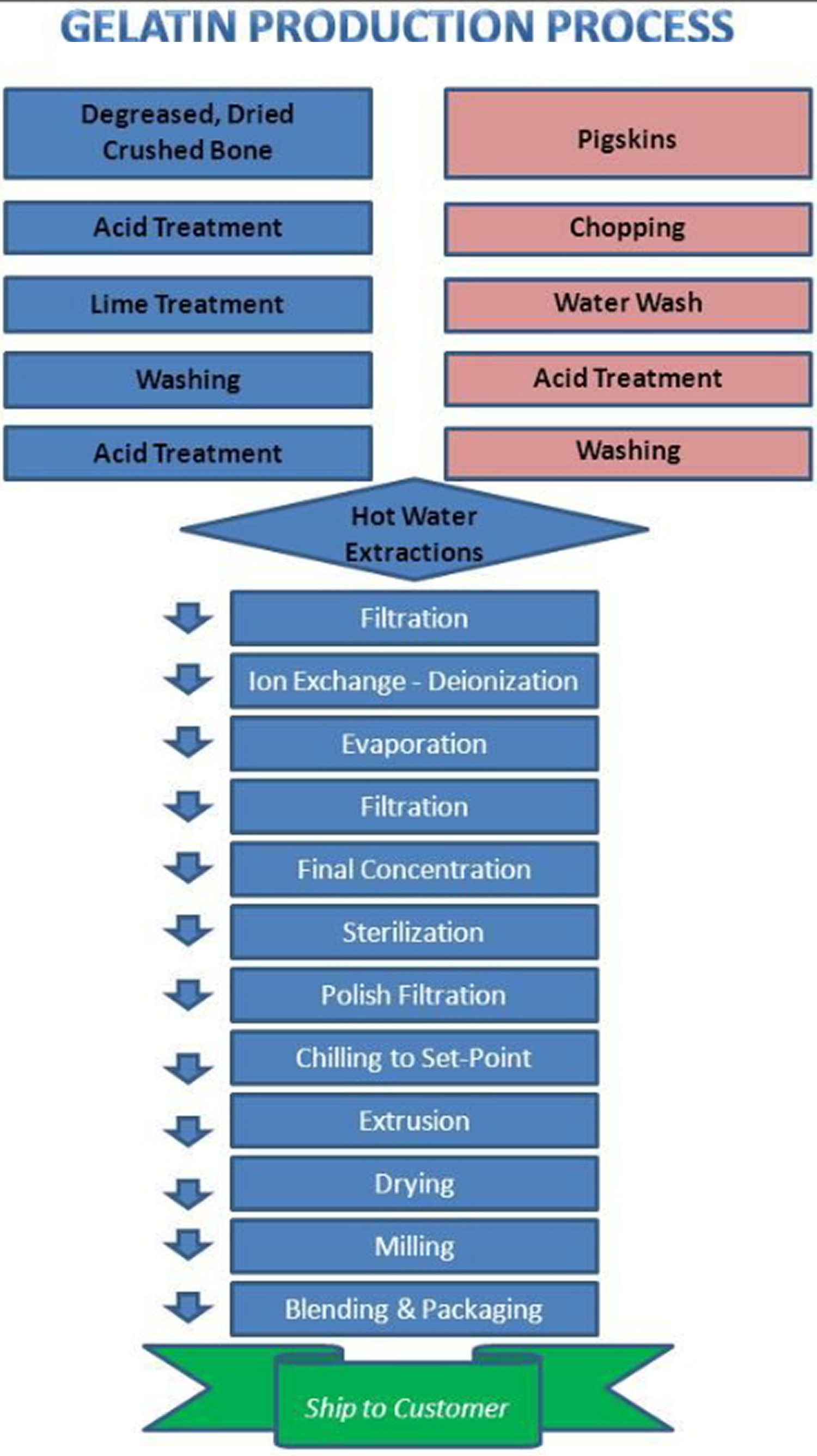
On a commercial scale, the principal raw materials used in gelatin production are cattle bones, cattle hides, and pork skins. Several alternative sources include poultry and fish. Extraneous substances, such as minerals (in the case of bone), fats and albuminoids (found in skin), are removed by chemical and physical treatment to give purified collagen. These pretreated materials are then hydrolyzed to gelatin which is soluble in hot water.
Gelatin recovered from bone is used primarily for pharmaceutical purposes. The so-called green bone from the slaughter of cattle is cleaned, degreased, dried, sorted, and crushed to a particle size of about 1-2 cm. The pieces of bone are then treated with dilute hydrochloric acid to remove mineral salts. The resulting sponge-like material is called ossein. From this point on in the manufacture of Type B gelatin, both cattle hides and ossein receive similar treatment.
Cattle hides are available from trimming operations in leather production. The hide pieces are usually dehaired chemically with a lime/sulfide solution followed by a mechanical loosening.
For the production of Type B gelatin, both ossein and cattle hide pieces are subjected to lengthy treatment with an alkali (usually lime) and water at ambient temperature. Depending on previous treatment, the nature of the material, the size of the pieces, and the exact temperature, liming takes 5-20 weeks, usually 8-12. The process is controlled by the degree of alkalinity of the lime liquor as determined by titration with acid, or by making test extractions. Ossein usually requires more liming time than cattle hides. Additional lime is added to maintain an excess, thereby compensating for any consumed. During liming some deamination of the collagen occurs, with evolution of ammonia. After conditioning, the raw material is thoroughly washed with cold water to remove excess lime; the pH adjusted with acid; and the product extracted with hot water to recover the soluble gelatin.
Porkskin is a significant raw material source for production of edible gelatin in the United States. The short time required for pre-treatment prior to extraction, and the minimization of wastewater generated are important economic factors in the manufacture of gelatin from this raw material. Supplied as either fresh or frozen, porkskins come from slaughter houses and meat processing plants already trimmed of fat, flesh and hair. Porkskins are usually dehaired by scalding with a hot dilute caustic soda solution. When porkskins are utilized for production of Type A gelatin they are washed with cold water and then soaked in cold dilute mineral acid for several hours until maximum swelling has occurred. Hydrochloric acid and sulfuric acid are most commonly employed. The remaining acid is then drained off and the material is again washed several times with cold water. The porkskins are then ready for extraction with hot water.
The pH, time, temperature, and number of extractions varies from processor to processor depending on product needs, type of equipment employed, timing of operations, and economics. Extraction procedures are closely controlled in the manufacture of both Type A and Type B gelatin since they influence both quality and quantity. Although continuous extraction is used by some processors, most methods still employ discrete batch fractions.
Extraction is normally carried out in stainless steel vessels equipped with provisions for heating and temperature control.
The number of extractions varies, 3-6 is typical. The first extraction generally takes place at 50-60°C, subsequent extractions being made with successive increases in temperature of 5-10°C. The final extraction is carried out close to the boiling point. Extracts are kept separate, analyzed, and subsequently blended to meet various customer specifications.
The initial extraction usually provides a superior product, compared with subsequent extractions. Earlier extractions have higher molecular weights, higher viscosity, higher gel strength, and lighter color. The later extractions are made at increasingly higher temperatures; the resulting product has lower molecular weight, lower gel strength, and darker color.
The dilute gelatin solutions from the various hot water extractions are filtered, deionized, and concentrated by cross flow membrane filtration and/or vacuum evaporation. The gelatin solution is then chilled and either cut into ribbons or extruded as noodles, and the gelled material is deposited as a bed onto an endless, open weave, stainless steel belt. The belt is passed through a drying chamber, which is divided into zones in each of which the temperature and humidity of the drying air is accurately controlled. Typical temperatures range from about 30°C in the initial zone up to about 70°C in the final zone. The air is usually conditioned by filtration, dehumidification and tempering.
Drying involves progressive increases in air temperature, often with exhaustion of moist air and replenishment with conditioned air. Drying time is 1-5 hours, depending on the quality and concentration of the material and the exact conditions employed. The rate of drying is carefully controlled to avoid melting and case hardening. The gelatin leaves the dryer with a moisture content of approximately 10%. The dried bed is then broken into pieces that are ground to the required particle size. Dried gelatin is tested for quality and gel strength according to standard methods developed by the Gelatin Manufacturers Institute of America.
Although gelatins are sometimes referred to as edible, pharmaceutical, photographic or technical, these terms refer only to their uses, and not to the methods of manufacture.
There are several varieties of gelatin, the composition of which depends on the source of collagen and the hydrolytic treatment used. Typical gelatin production processes are shown in Figure 1.
Figure 1. How gelatin is made
Gelatin also can be prepared in the home. Boiling certain cartilaginous cuts of meat or bones results in gelatin being dissolved into the water. Depending on the concentration, the resulting stock (when cooled) will form a jelly or gel naturally.
While many processes exist whereby collagen may be converted to gelatin, they all have several factors in common. The intermolecular and intramolecular bonds that stabilize insoluble collagen must be broken, and also, the hydrogen bonds that stabilize the collagen helix must be broken. The manufacturing processes of gelatin consists of three main stages:
- Pretreatments to make the raw materials ready for the main extraction step and to remove impurities that may have negative effects on physiochemical properties of the final gelatin product
- The main extraction step, which usually is done with hot water or dilute acid solutions as a multistage extraction to hydrolyze collagen into gelatin
- The refining and recovering treatments including filtration, clarification, evaporation, sterilization, drying, rutting, grinding, and sifting to remove the water from the gelatin solution, to blend the gelatin extracted, and to obtain dried, blended and ground final product
Pretreatments
If the raw material used in the production of the gelatin is derived from bones, dilute acid solutions are used to remove calcium and other salts. Hot water or several solvents may be used to reduce the fat content, which should not exceed 1% before the main extraction step. If the raw material consists of hides and skin; size reduction, washing, removal of hair from hides, and degreasing are necessary to prepare the hides and skins for the main extraction step.
Collagen hydrolysis is performed by one of three different methods: acid-, alkali-, and enzymatic hydrolysis. Acid treatment is especially suitable for less fully crosslinked materials such as pig skin collagen and normally requires 10 to 48 hours. Alkali treatment is suitable for more complex collagen such as that found in bovine hides and requires more time, normally several weeks. The purpose of the alkali treatment is to destroy certain chemical crosslinks still present in collagen. Within the gelatin industry, the gelatin obtained from acid-treated raw material has been called type-A gelatin and the gelatin obtained from alkali-treated raw material is referred to as type-B gelatin.
Advances are occurring to optimize the yield of gelatin using enzymatic hydrolysis of collagen. The treatment time is shorter than that required for alkali treatment, however, and results in almost complete conversion to the pure product. The physical properties of the final gelatin product are considered better.
Extraction
After preparation of the raw material, i.e., reducing cross-links between collagen components and removing some of the impurities such as fat and salts, partially purified collagen is converted into gelatin by extraction with either water or acid solutions at appropriate temperatures. All industrial processes are based on neutral or acid pH values because although alkali treatments speed up conversion, they also promote degradation processes. Acidic extraction conditions are extensively used in the industry, but the degree of acid varies with different processes. This extraction step is a multistage process, and the extraction temperature usually is increased in later extraction steps, which ensures minimum thermal degradation of the extracted gelatin.
Recovery
This process includes several steps such as filtration, evaporation, drying, grinding, and sifting. These operations are concentration-dependent and also dependent on the particular gelatin used. Gelatin degradation should be avoided and minimized, so the lowest temperature possible is used for the recovery process. Most recoveries are rapid, with all of the processes being done in several stages to avoid extensive deterioration of the peptide structure. A deteriorated peptide structure would result in a low gel strength, which is not generally desired.
Gelatin benefits
Gelatin nutrition facts
Table 1. Gelatin (unflavored)
Nutrient | Unit | PACKET 7 g | Value per 100 g | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Approximates | |||||||||||||||||||
| Energy | kcal | 30 | 429 | ||||||||||||||||
| Protein | g | 7.00 | 100.00 | ||||||||||||||||
| Total lipid (fat) | g | 0.00 | 0.00 | ||||||||||||||||
| Carbohydrate, by difference | g | 0.00 | 0.00 | ||||||||||||||||
| Fiber, total dietary | g | 0.0 | 0.0 | ||||||||||||||||
| Sugars, total | g | 0.00 | 0.00 | ||||||||||||||||
| Minerals | |||||||||||||||||||
| Calcium, Ca | mg | 0 | 0 | ||||||||||||||||
| Iron, Fe | mg | 0.00 | 0.00 | ||||||||||||||||
| Sodium, Na | mg | 10 | 143 | ||||||||||||||||
| Vitamins | |||||||||||||||||||
| Vitamin C, total ascorbic acid | mg | 0.0 | 0.0 | ||||||||||||||||
| Vitamin A, IU | IU | 0 | 0 | ||||||||||||||||
| Lipids | |||||||||||||||||||
| Fatty acids, total saturated | g | 0.000 | 0.000 | ||||||||||||||||
| Fatty acids, total trans | g | 0.000 | 0.000 | ||||||||||||||||
| Cholesterol | mg | 0 | 0 | ||||||||||||||||
Table 2. Gelatins, dry powder, unsweetened
Nutrient | Unit | Value per 100 g | envelope (1 tbsp) 7 g | package (1 oz) 28 g | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Approximates | |||||||||||||||||||
| Water | g | 13.00 | 0.91 | 3.64 | |||||||||||||||
| Energy | kcal | 335 | 23 | 94 | |||||||||||||||
| Protein | g | 85.60 | 5.99 | 23.97 | |||||||||||||||
| Total lipid (fat) | g | 0.10 | 0.01 | 0.03 | |||||||||||||||
| Carbohydrate, by difference | g | 0.00 | 0.00 | 0.00 | |||||||||||||||
| Fiber, total dietary | g | 0.0 | 0.0 | 0.0 | |||||||||||||||
| Sugars, total | g | 0.00 | 0.00 | 0.00 | |||||||||||||||
| Minerals | |||||||||||||||||||
| Calcium, Ca | mg | 55 | 4 | 15 | |||||||||||||||
| Iron, Fe | mg | 1.11 | 0.08 | 0.31 | |||||||||||||||
| Magnesium, Mg | mg | 22 | 2 | 6 | |||||||||||||||
| Phosphorus, P | mg | 39 | 3 | 11 | |||||||||||||||
| Potassium, K | mg | 16 | 1 | 4 | |||||||||||||||
| Sodium, Na | mg | 196 | 14 | 55 | |||||||||||||||
| Zinc, Zn | mg | 0.14 | 0.01 | 0.04 | |||||||||||||||
| Vitamins | |||||||||||||||||||
| Vitamin C, total ascorbic acid | mg | 0.0 | 0.0 | 0.0 | |||||||||||||||
| Thiamin | mg | 0.025 | 0.002 | 0.007 | |||||||||||||||
| Riboflavin | mg | 0.230 | 0.016 | 0.064 | |||||||||||||||
| Niacin | mg | 0.085 | 0.006 | 0.024 | |||||||||||||||
| Vitamin B-6 | mg | 0.007 | 0.000 | 0.002 | |||||||||||||||
| Folate, DFE | µg | 30 | 2 | 8 | |||||||||||||||
| Vitamin B-12 | µg | 0.00 | 0.00 | 0.00 | |||||||||||||||
| Vitamin A, RAE | µg | 0 | 0 | 0 | |||||||||||||||
| Vitamin A, IU | IU | 0 | 0 | 0 | |||||||||||||||
| Vitamin E (alpha-tocopherol) | mg | 0.00 | 0.00 | 0.00 | |||||||||||||||
| Vitamin D (D2 + D3) | µg | 0.0 | 0.0 | 0.0 | |||||||||||||||
| Vitamin D | IU | 0 | 0 | 0 | |||||||||||||||
| Vitamin K (phylloquinone) | µg | 0.0 | 0.0 | 0.0 | |||||||||||||||
| Lipids | |||||||||||||||||||
| Fatty acids, total saturated | g | 0.070 | 0.005 | 0.020 | |||||||||||||||
| Fatty acids, total monounsaturated | g | 0.060 | 0.004 | 0.017 | |||||||||||||||
| Fatty acids, total polyunsaturated | g | 0.010 | 0.001 | 0.003 | |||||||||||||||
| Cholesterol | mg | 0 | 0 | 0 | |||||||||||||||
| Other | |||||||||||||||||||
| Caffeine | mg | 0 | 0 | 0 | |||||||||||||||
Table 3. Gelatin desserts, dry mix
Nutrient | Unit | Value per 100 g | package (3 oz) 85 g | portion, amount to make 1/2 cup 21 g | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Approximates | |||||||||||||||||||
| Water | g | 1.00 | 0.85 | 0.21 | |||||||||||||||
| Energy | kcal | 381 | 324 | 80 | |||||||||||||||
| Protein | g | 7.80 | 6.63 | 1.64 | |||||||||||||||
| Total lipid (fat) | g | 0.00 | 0.00 | 0.00 | |||||||||||||||
| Carbohydrate, by difference | g | 90.50 | 76.92 | 19.00 | |||||||||||||||
| Fiber, total dietary | g | 0.0 | 0.0 | 0.0 | |||||||||||||||
| Sugars, total | g | 86.04 | 73.13 | 18.07 | |||||||||||||||
| Minerals | |||||||||||||||||||
| Calcium, Ca | mg | 3 | 3 | 1 | |||||||||||||||
| Iron, Fe | mg | 0.13 | 0.11 | 0.03 | |||||||||||||||
| Magnesium, Mg | mg | 2 | 2 | 0 | |||||||||||||||
| Phosphorus, P | mg | 141 | 120 | 30 | |||||||||||||||
| Potassium, K | mg | 7 | 6 | 1 | |||||||||||||||
| Sodium, Na | mg | 466 | 396 | 98 | |||||||||||||||
| Zinc, Zn | mg | 0.01 | 0.01 | 0.00 | |||||||||||||||
| Vitamins | |||||||||||||||||||
| Vitamin C, total ascorbic acid | mg | 0.0 | 0.0 | 0.0 | |||||||||||||||
| Thiamin | mg | 0.003 | 0.003 | 0.001 | |||||||||||||||
| Riboflavin | mg | 0.041 | 0.035 | 0.009 | |||||||||||||||
| Niacin | mg | 0.009 | 0.008 | 0.002 | |||||||||||||||
| Vitamin B-6 | mg | 0.001 | 0.001 | 0.000 | |||||||||||||||
| Folate, DFE | µg | 3 | 3 | 1 | |||||||||||||||
| Vitamin B-12 | µg | 0.00 | 0.00 | 0.00 | |||||||||||||||
| Vitamin A, RAE | µg | 0 | 0 | 0 | |||||||||||||||
| Vitamin A, IU | IU | 0 | 0 | 0 | |||||||||||||||
| Vitamin E (alpha-tocopherol) | mg | 0.00 | 0.00 | 0.00 | |||||||||||||||
| Vitamin D (D2 + D3) | µg | 0.0 | 0.0 | 0.0 | |||||||||||||||
| Vitamin D | IU | 0 | 0 | 0 | |||||||||||||||
| Vitamin K (phylloquinone) | µg | 0.0 | 0.0 | 0.0 | |||||||||||||||
| Lipids | |||||||||||||||||||
| Fatty acids, total saturated | g | 0.000 | 0.000 | 0.000 | |||||||||||||||
| Fatty acids, total monounsaturated | g | 0.000 | 0.000 | 0.000 | |||||||||||||||
| Fatty acids, total polyunsaturated | g | 0.000 | 0.000 | 0.000 | |||||||||||||||
| Cholesterol | mg | 0 | 0 | 0 | |||||||||||||||
| Other | |||||||||||||||||||
| Caffeine | mg | 0 | 0 | 0 | |||||||||||||||
Gelatin Protein content
Although gelatin is 98–99% protein by dry weight, it has little additional nutritional value, varying according to the source of the raw material and processing technique 9.
Amino acids present in gelatin are variable, due to varying sources and batches, but are approximately 9:
- Glycine 21%
- Proline 12%
- Hydroxyproline 12%
- Glutamic acid 10%
- Alanine 9%
- Arginine 8%
- Aspartic acid 6%
- Other 22%
Gelatin capsules
The use of gelatin in the manufacture of various pharmaceutical dosage forms dates back to the early 19th century and possibly earlier. Today, the commonly recognized dosage forms using gelatin are two-piece hard capsules, soft elastic gelatin capsules (Softgels), tableting, tablet coating, granulation, encapsulation and micro-encapsulation. All gelatins used meet or exceed the requirements of the current United States Pharmacopoeia and National Formulary.
Two-Piece Hard Capsules – The Manufacture of hard gelatin capsules consists of the dipping of stainless steel mold pins into a gelatin solution, drying, stripping from the pins into a collate, trimming of the caps and bodies, and joining together for shipment. The strength and flexibility, the clarity of the gelatin, and the movability of the gelatin provide unique characteristics that allow the manufacture of various sizes, colors and designs for assuring a snap closure after filling 10.
The typical hard capsule formula would be:
- Gelatin 30%
- Water 65%
- Dye 5%
- Pigment As needed
- Plasticizer As needed
Soft Elastic Gelatin Capsules – Softgels, as they are commonly known today, utilize a gelatin solution that is plasticized with propylene glycol, sorbitol, glycerin or various approved mixtures. Soft gelatin capsules are one-piece and hermetically sealed to enclose a liquid or semi-liquid fill. Soft gelatin capsules are manufacture-formed, filled and sealed in one continuous operation. The soft gelatin encapsulation process begins with the formation of two sheets called ribbons from a molten gelatin mass, each of which is passed over a die of the desired capsule size and shape. At the point where the two rotating dies meet, the capsule is formed and filled with a liquid that contains the active ingredient in a vehicle. The capsule is then washed to remove the lubricants and dried.
Functional properties of gelatin in foods
Commercial gelatins vary from 50-300 Bloom grams and, except for specialty items, are free of added colors, flavors, preservatives, and chemical additives. Gelatin is a generally recognized as safe (GRAS) food ingredient.
Two of gelatin’s most desirable properties are its melt-in-the-mouth characteristics and its ability to form thermo-reversible gels. In addition, gelatin is relatively unaffected by ionic strength and is stable over a broad pH range. Gelatin is preferred in many applications for its clarity and bland flavor.
Table 4. Gelatin in foods
Function | Application |
Gel former | Gelled desserts, lunch meats, confectionery, pate, consommé, aspics |
Whipping agent | Marshmallows, nougats, mousses, soufflés, chiffons, whipped cream |
Protective colloid | Confectionery, icings, ice creams, frozen desserts and confections |
Binding agent | Meat rolls, canned meats, confectionery, cheeses, dairy products |
Clarifying agent | Beer, wine, fruit juices, vinegar |
Film former | Coating for fruits, meats, deli items |
Thickener | Powdered drink mixes, bouillon, gravies, sauces, soups, puddings, jellies, syrups, dairy products |
Process aid | Microencapsulation of colors, flavors, oils, vitamins |
Emulsifier | Cream soups, sauces, flavorings, meat pastes, whipped cream, confectionery, dairy products |
Stabilizer | Cream cheese, chocolate milk, yogurt, icings, cream fillings, frozen desserts |
Adhesive agent | To affix nonpareils, coconut and other items to confections, to bond layered confections together, to bind frostings to baked goods, to bind seasonings to meat products. |
Confectionary
Confections are typically made from a base of sugar, corn syrup and water. Gelatin is widely used in confections because it foams, gels, or solidifies into a piece that dissolves slowly or melts in the mouth.
Confections such as gummi bears contain a relatively high percentage of gelatin. These candies dissolve more slowly thus lengthening the enjoyment of the candy while smoothing the flavor.
Gelatin is used in whipped confections such as marshmallows where it serves to lower the surface tension of the syrup, stabilize the foam thru increased viscosity, set the foam via gelation, and prevent sugar crystallization. Gelatin is used in foamed confections at a 2-7% level, depending upon the desired texture. Gummy foams use about 7% of a 175 Bloom gelatin. Marshmallow producers generally use 2.5% of a 250 Bloom Type A gelatin.
Gelatin Desserts
Gelatin desserts can be traced back to 1845 when a U.S. patent was issued use for “portable gelatin” for use in desserts. Gelatin desserts remain popular: the current U.S. market for gelatin desserts exceeds 100 million pounds annually. Today’s consumers are concerned with caloric intake. Regular gelatin desserts are easy to prepare, pleasant tasting, nutritious, available in a variety of flavors, and contain only 80 calories per half-cup serving. Sugar-free versions are a mere eight calories per serving. Gelatin desserts can be prepared using either Type A or Type B gelatin with Blooms between 175 and 275. The higher the Bloom the less gelatin required for a proper set (i.e. 275 Bloom gelatin will require about 1.3% gelatin while a 175 Bloom gelatin will require 2.0% to obtain an equal set). Sweeteners other than sucrose can be used.
Gelatin in Meats
Gelatin is used to gel aspics, head cheese, souse, chicken rolls, glazed and canned hams, and jellied meat products of all kinds. The gelatin functions to absorb meat juices and to give form and structure to products that would otherwise fall apart. Normal usage level ranges from 1 to 5% depending upon the type of meat, amount of broth, gelatin Bloom, and texture desired in the final product.
Clarification of Beverages and Juices
Gelatin has traditionally been used to clarify wine, beer and fruit juices. Fining of these beverages requires only 40 to 80 parts per million of a 100 to 200 Bloom gelatin. In practice a dilute (1-3%) gelatin solution is introduced into the top of the tank and then allowed to settle before filtration.
Special Dietary Uses
Gelatin in hydrolyzed form is used to protein fortify dietary foods. Dried, hydrolyzed gelatin contains over 92% protein. Typically it is blended with other hydrolyzed proteins to balance the nutritional aspects of the amino acids. Soups, shakes and fruit drinks are common uses for gelatin hydrolysates.
Pharmaceutical gelatins
Tablet Coating
Tablets are generally coated to reduce dusting, mask unpleasant taste, and allow for printing and color coatings for product identification. The coating process is more and more becoming aqueous-based which allows for the use of gelatin. Typical formulations for coating include a sugar, pigment, and gelatin to act as a film former. The most popular coating method utilizes a roll coating pan and the addition of the coating solution as a fine spray.
Suppositories
Glycerinated gelatin is typically used as a vehicle for suppositories for insertion into the rectum, vagina or the urethra. The firmness of the finished product is adjusted by varying the gelatin concentration in the formula. The criteria for any suppository formulation are that the base (gelatin) be nontoxic and nonirritating to mucous membranes, compatible with a variety of drugs, the base melts or dissolves in body fluids, and the base should be stable on storage. Suppositories are generally molded by compression or by fusion molding. The gelatin base affords all the desired characteristics required by either method. The two types of gelatin each provide characteristics that are advantageous for formulation dependent upon the active and the area of application.
Type A gelatin is generally adjusted to an acidic pH which is below the isoelectric point, while a Type B gelatin is also adjusted to a pH that is acidic, but in this case the pH is above the isoelectric point. Thus the cationic, anionic, and the nonionic characteristics can be used to promote compatibility with the active, compatibility with the surrounding fluids and temperatures, and enhance or control the bioavailability. The pH exists where the positive and negative charges are equivalent and the application of an electric charge has no effect. The principal chemical and physical characteristics of a gelatin solution are dependent upon the pH and the ionic strength of the media. The formulation and preparation of the gelatin can in fact modify the isoelectric point in such a way that various physical characteristics can be enhanced. The suppository dosage form is just one example of the use of varied isoelectric points that enhance the performance of the drug delivery system.
Gelatin Emulsions
Gelatin is recognized in many industries as a stabilizer, a texturizing agent, a film former and as a colloidal support media. The preparation of an oil emulsion with gelatin for use as a topical allows the oils to be stored and maintains droplet distribution and droplet size over extended periods. Depending upon the concentration and temperature, the emulsion can be free flowing, semi-solid or solid.
Microencapsulation
Gelatin is used to produce microencapsulated oils for various uses both in nutritional and pharmaceutical applications. The traditional method of encapsulation is known as coacervation in which the dispersed oil is encapsulated by gelatin at the interface between the aqueous phase and the nonaqueous phases. Common examples of this are vitamin supplements for various foods and for multi-vitamins. The size and formation of the spherical microcapsules can be controlled by various methods. Typical microcapsules range in size from 5 microns to 500 microns.
Absorbable Gelatin Sponge
Gelatin is used in the form of a sterile, water insoluble sponge (Gelfoam TM) to control bleeding during surgery. It is later absorbed over a 4 to 6 week period.
Plasmas Substitues – Solution of modified gelatin (3.0 – 5.5%) and salts are commonly used as plasma substitute during emergency surgery.
Absorbable Gelatin Film
Another application is a sterile, non-antigenic, water-insoluble absorbable gelatin film (Gelfilm TM) obtained from a specially prepared gelatin/formaldehyde solution.
Pastilles and Troches
These products are commonly used as cough drops. Like gelled confections, these products are unique in that in the mouth they are surrounded by a fluid that prevents the active ingredient from being concentrated and creating irritations.
Bacterial Growth Media
As with any pharmaceutical excipient, the level and type of bacteria found is always of concern. Pharmaceutical gelatin is highly purified and sterilized to eliminate these concerns. However, as gelatin is derived from collagen, the ability to perform as a nutrient is itself an application. Special grades of gelatin are useful in bacterial culture media. The specificity of the media to detect and count various bacterial strains is a useful diagnostic and research tool. A variety of media are available that utilize the nutrient properties of gelatin.
Is gelatin bad for you
Potential allergic reaction in vaccines
In 16% and 38% of beef and pork meat sensitized children, respectively, have IgE antibodies to gelatins that are cross-reactive 1. The presence of IgE anti-gelatin may place them at risk for potential allergic reactions following exposure to gelatin containing foods, vaccines or other medical products. American 11 and Finnish 12 groups have reported that 27% and 14–28%, respectively, of children who experienced systemic reactions after measles, mumps and rubella vaccination, had gelatin-specific IgE antibodies.
Bovine spongiform encephalopathy (mad cow disease)
Bovine spongiform encephalopathy (BSE) is a progressive neurological disorder of cattle; its symptoms are similar to a disease of sheep, called scrapie 13. Bovine spongiform encephalopathy (BSE) has been called “mad cow disease.” Bovine spongiform encephalopathy (BSE) and scrapie both result from infection with a very unusual infectious agent. As of January 2004, more than 180,000 cases of bovine spongiform encephalopathy (BSE) were confirmed in Great Britain in more than 35,000 herds of cattle. The epidemic peaked in January 1993 at almost 1,000 new cases per week. Although the origin of the disease is uncertain it may have resulted from the feeding of scrapie containing meat and bone meal to cattle or from feeding cattle meat and bone meal derived from a cow or other animal that developed the disease due to a spontaneous mutation. There is strong evidence and general agreement that the outbreak was amplified by feeding meat-and-bone meal prepared from cattle to young calves.
The nature of the infectious agent that causes bovine spongiform encephalopathy (BSE) and scrapie is unknown 13. Currently, the most accepted theory is that the agent is a modified form of a normal cell protein known as a prion. A prion is not a bacterium, parasite, or virus, and thus treatments usually used for treating or preventing bacterial infections (e.g. antibiotics) or viral infections are not effective against prions.
In cattle naturally infected with bovine spongiform encephalopathy, the BSE agent has been found in brain tissue, in the spinal cord, and in the retina of the eye. Additional experimental studies suggest that the bovine spongiform encephalopathy agent may also be present in the small intestine, tonsil, bone marrow, and dorsal root ganglia (lying along the vertebral column).
Bovine spongiform encephalopathy (BSE) belongs to a group of progressive degenerative neurological diseases known as transmissible spongiform encephalopathies (TSEs). Transmissible spongiform encephalopathy (TSE) diseases are always fatal. The TSE diseases include scrapie, which affects sheep and goats; transmissible mink encephalopathy; feline (cat) spongiform encephalopathy; and chronic wasting disease of deer and elk. There are six transmissible spongiform encephalopathy (TSE) diseases that affect people: kuru, classical Creutzfeldt-Jakob disease (CJD) and variant Creutzfeldt-Jakob disease (vCJD), Gerstmann-Sträussler-Scheinker syndrome, fatal familial insomnia, and sporadic fatal insomnia. The human diseases are very rare; for example, classical Creutzfeldt-Jakob disease (CJD) has been well studied and occurs sporadically worldwide at a rate of about one case per one million people each year.
In contrast to the classic form of CJD, the new variant or variant form (vCJD) in the United Kingdom and France affects younger persons (average age at onset: 26 years), and has different clinical features from CJD 14. People with vCJD begin with serious psychiatric problems or problems with their senses (ears, eyes or smell). This first set of symptoms is followed weeks or months later by poor muscle coordination, muscle spasms, and mental confusion. The illness lasts for at least 6 months, and on average people with vCJD die approximately 13 months after their symptoms begin. When patients’ brains are examined by autopsy, there are clear changes in brain tissue structure, including many “spongiform,” or open spongy-looking areas, abnormal clumps of prion protein called plaques, and other areas with less prominent accumulations of abnormal prion protein.
On March 20, 1996 a statement from the Spongiform Encephalopathy Advisory Committee (SEAC) of the United Kingdom indicated concern that before November 1989, when inclusion of certain cow and sheep by-products in human food was banned, the BSE agent may have been transmitted to people through contaminated food products. The SEAC said that food might account for the 10 vCJD cases described in 1996 15. The specific foods, if any that may be associated with the transmission of this agent from cattle to humans are unknown.
The vast majority of cases of bovine spongiform encephalopathy (BSE) (more than 97% as of 2003) have been reported from the United Kingdom during an epidemic. However, endemic cases have also been reported in other European countries including: the Republic of Ireland, Switzerland, France, Liechtenstein, Luxembourg, Netherlands, Portugal and Denmark. The numbers of reported cases by country are available on the web site of the Office International des Epizooties. These numbers should be interpreted with caution, however, because the intensity and methods of surveillance probably vary over time and by country. In 2003 one case was reported in Canada and one in the United States (in a cow born in Canada).
An inquiry by the British government has however, concluded that scrapie infected meat and bone meal was not the source of BSE nor was the change in the rendering practices responsible for survival of the bovine spongiform encephalopathy (BSE) agent. Rather, this inquiry has stated that bovine spongiform encephalopathy (BSE) may have originated spontaneously as a result of a genetic mutation and was amplified by the feeding of contaminated meat and bone meal to cattle.
According to the Animal Health and Plant Inspection Service (APHIS) of the United States Department of Agriculture, bovine spongiform encephalopathy (BSE) has been detected in one cow in the United States. Following complications of pregnancy this cow was slaughtered December 9, 2003. Following this discovery the USDA and FDA announced additional measures to enhance the US protections against bovine spongiform encephalopathy (BSE).
What measures has the US government taken to ensure that people are not exposed to the BSE agent in foods ?
The USDA is responsible for the health of US livestock. To prevent BSE from entering the country, the USDA Animal and Plant Health Inspection Service (APHIS) has, since 1989, prohibited the importation of live ruminants from countries where BSE is known to exist in native cattle. On December 12, 1997, APHIS stopped the importation of live ruminants and most ruminant products, including meat, meat-and-bone meal, offals, glands, etc. from all of Europe. FDA is responsible for animal feeds in the US. In August 1997, FDA prohibited the use of most mammalian protein in the manufacture of animal feeds given to ruminants. Following the discovery of one cow with BSE in the US, the USDA and FDA have announced additional measures to enhance protections against the spread of BSE in US cattle and to minimize human exposure to bovine materials that may contain the BSE agent. USDA has issued an interim final rule (Federal Register January 12, 2004 Vol. 69, Number 7) removing downer animals and specified risk materials and tissues from the human food chain; requiring additional process controls for establishments using advanced meat recovery; holding meat from cattle that have been targeted for BSE surveillance testing until the test has confirmed negative; and prohibiting the air injection stunning of cattle. In January 2004, FDA proposed additional safeguards including: excluding brain, spinal cord, gut and eyes of older animals from human food and from rendered material in animal feeds, eliminating poultry litter, cow blood and processed plate waste as feed ingredients for cattle, labeling requirements for pet food, and additional control measures to prevent cross contamination of feed and feed ingredients at feed mills. In addition, since 1990, the USDA has led an interagency surveillance program for evidence of BSE in the US. USDA has tested 20,000 animals annually for each of the last 2 years, and approximately 75 percent of these were downers at slaughter. A BSE risk assessment performed by Harvard University’s Center for Risk Analysis at the School of Public Health concluded that even if BSE were to occur in the US the measures already taken would largely prevent its spread to animals or humans, and the disease would gradually disappear over a number of years.
On March 18, 2016 the FDA finalized three previously-issued interim final rules designed to further reduce the potential risk of BSE in human food 16. The final rule clarified that “…we are amending the final rule to provide a definition of gelatin and to clarify that gelatin is not considered a prohibited cattle material under 21 CFR 189.5(a)(1) and 700.27(a)(1) as long as it is manufactured using the customary industry processes specified” 16.
The FDA Recommendations – FDA has been reviewing the currently available scientific information, including information provided on behalf of the Gelatin Manufacturers of Europe and the Gelatin Manufacturers Institute of America. FDA also considered the advisory committee’s recommendations and other available information. Based on this review, FDA proposes the following recommendations concerning the acceptability of gelatin for use in FDA-regulated products intended for human use 17:
- In order to ensure that all parties in the distribution chain take appropriate responsibility, importers, manufacturers, and suppliers should determine the tissue, species, and country source of all materials to be used in processing gelatin for human use.
- Bones and hides from cattle that shows signs of neurological disease, from any source country, should not be used as raw material for the manufacture of gelatin.
- Gelatin produced from bones and hides obtained from cattle residing in, or originating from, countries reporting BSE or from countries that do not meet the latest BSE-related standards of the Office International des Epizooties should not be used either in injectable, ophthalmic, or implanted FDA-regulated products, or in their manufacture.
- At this time, there does not appear to be a basis for objection to the use of gelatin in FDA-regulated products for oral consumption and cosmetic use by humans when the gelatin is produced from bones obtained from cattle residing in, or originating from, BSE countries, if the cattle come from BSE-free herds and if the slaughterhouse removes the heads, spines, and spinal cords directly after slaughter. Nor does there appear to be a basis for objection to gelatin for oral consumption and cosmetic use which is produced from bones from countries which have not reported BSE but which fail to meet OIE standards if the slaughterhouse removes the heads, spine, and spinal cords after slaughter. Gelatin processors should ensure that slaughterhouses that supply bovine bones for gelatin production remove heads, spines, and spinal cords as the first procedure following slaughter.
- At this time, there does not appear to be a basis for objection to the use of gelatin produced from bovine hides, from any source country, in FDA-regulated products for oral consumption and cosmetic use by humans use if processors ensure that the bovine hides have not been contaminated with brain, spinal cord, or ocular tissues of cattle residing in, or originating from, BSE countries and if they exclude hides from cattle that have signs of neurological disease (see #2).
- At this time, there does not appear to be a basis for objection to the use of gelatin produced from bovine hides and bones in FDA-regulated products for human use if the gelatin is produced from U.S.-derived raw materials or from cattle born, raised, and slaughtered in other countries that have no reported BSE cases and that meet OIE BSE standards.
- At this time, there does not appear to be a basis for objection to the use of gelatin produced from porcine skins, from any source country, in FDA-regulated products for human use. Processors should ensure that gelatin made from porcine skins is not cross-contaminated with bovine materials originating from BSE countries or from countries that do not meet OIE standards.
- Bogdanovic J, Halsey NA, Wood RA, Hamilton RG. Bovine and Porcine Gelatin Sensitivity in Milk and Meat-Sensitized Children. The Journal of allergy and clinical immunology. 2009;124(5):1108-1110. doi:10.1016/j.jaci.2009.06.021. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2784137/
- Anaphylactic reactions to modified fluid gelatins. Vervloet D, Senft M, Dugue P, Arnaud A, Charpin J. J Allergy Clin Immunol. 1983 Jun; 71(6):535-40. https://www.ncbi.nlm.nih.gov/pubmed/6189875/
- Gelatin: a valuable protein for food and pharmaceutical industries: review. Crit Rev Food Sci Nutr. 2001 Sep;41(6):481-92. http://www.tandfonline.com/doi/abs/10.1080/20014091091904
- Ward, A.G.; Courts, A. (1977). The Science and Technology of Gelatin. New York: Academic Press. ISBN 0-12-735050-0.
- Gelatin Handbook. http://www.gelatin-gmia.com/images/GMIA_Gelatin_Manual_2012.pdf
- Bloom test. Wikipedia. https://en.wikipedia.org/wiki/Bloom_(test
- Schrieber, Reinhard; Gareis, Herbert. Gelatine Handbook: Theory and Industrial Practice. Wiley. ISBN 978-3-527-61097-6.
- United States Department of Agriculture Agricultural Research Service. National Nutrient Database for Standard Reference Release 28. https://ndb.nal.usda.gov/ndb/search/list
- Francis, Frederick J., ed. (2000). “Gelatin”. Encyclopedia of Food Science and Technology (2nd ed.). John Wiley & Sons. pp. 1183–1188. ISBN 9780471192558.
- Eaton, F. 1989. Manufacture of Hard Shell Gelatin Capsules and Associated Problems, Presented at Hard Shell and Softgel Capsule Manufacturing Conference, Medical Manufacturing Techsource, March, 1989.
- Prevalence of anti-gelatin IgE antibodies in people with anaphylaxis after measles-mumps rubella vaccine in the United States. Pool V, Braun MM, Kelso JM, Mootrey G, Chen RT, Yunginger JW, Jacobson RM, Gargiullo PM, VAERS Team. US Vaccine Adverse Event Reporting System. Pediatrics. 2002 Dec; 110(6):e71. https://www.ncbi.nlm.nih.gov/pubmed/12456938/
- Allergic reactions to measles-mumps-rubella vaccination. Patja A, Mäkinen-Kiljunen S, Davidkin I, Paunio M, Peltola H. Pediatrics. 2001 Feb; 107(2):E27. https://www.ncbi.nlm.nih.gov/pubmed/11158501/
- Bovine Spongiform Encephalopathy (BSE) Questions and Answers. U.S. Food and Drug Administration. https://www.fda.gov/biologicsbloodvaccines/safetyavailability/ucm111482.htm
- Lancet, 1996, 347: 921-25, Rev.Med.Virol. 2002;12: 143-50
- Lancet 1996; 347:921-5
- Use of Materials Derived From Cattle in Human Food and Cosmetics. U.S. Food and Drug Administration. https://www.federalregister.gov/documents/2016/03/18/2016-06123/use-of-materials-derived-from-cattle-in-human-food-and-cosmetics
- The Sourcing and Processing of Gelatin to Reduce the Potential Risk Posed by Bovine Spongiform Encephalopathy (BSE) in FDA-Regulated Products for Human Use. U.S. Food and Drug Administration. https://www.fda.gov/regulatoryinformation/guidances/ucm125182.htm