Joint effusion
Joint effusion is commonly referred to as the abnormal accumulation of fluid in or around a joint. Joint effusion is most commonly caused by infection, injury, and arthritis. In addition to swelling, joint effusion is associated with pain and stiffness.
Knee effusion
The knee is a hinge joint and susceptible to injury from trauma, inflammation, infection, and degenerative changes. Knee joint effusion may result from acute or chronic conditions 1. Knee effusion causes range from trauma or overuse to systemic disease. The knee is more frequently injured than other joints because it is part of a weight-bearing limb, and it does not have the stability brought by the joint congruity of the hip and ankle 2. The lifetime prevalence of knee joint effusion has been reported as high 27% 3. Knee joint effusion may occur at any age in children, but most commonly occurs in infants and teenagers.
The knee contains sac-like structures containing synovial fluid, called bursae, which are located between the skin and bony prominences. Fluid accumulation in the intra-articular space of a joint is called an effusion. Small, asymptomatic effusions can occur in healthy individuals. Larger joint effusions indicate intra-articular pathology. In the knee joint, trauma, overuse, infections, systemic causes, or changes in osteoarthritis may cause the effusion. These large effusions can also cause popliteal cysts. Acute swelling can be the result of a hemarthrosis from an injury to a ligament or fracture.
Understanding the basic anatomy and pathophysiology of knee effusions is important to make an accurate diagnosis. However, the most important information in making the diagnosis will be to correlate findings with a thorough history and physical exam.
If trauma is involved, the clinician should inquire about the date and mechanism of injury, what sport, if any, was involved, and whether a direct blow caused the injury. Historical findings suggestive of fracture include a mechanism of injury involving a high-velocity collision, the occurrence of a “pop” at the time of injury, age greater than 55 years and inability to bear weight immediately after the injury. Anterior cruciate ligament (ACL) injuries typically present after a noncontact deceleration, a “cutting” movement or hyperextension, often accompanied by a “pop,” with the inability to continue sports participation and associated knee instability.
The posterior cruciate ligament (PCL) is injured far less frequently than the anterior cruciate ligament (ACL). The typical mechanism of injury of the posterior cruciate ligament (PCL) is a blow to the anterior proximal tibia with the knee flexed, such as tripping over a hurdle or striking the dashboard in a motor vehicle accident. Patients with PCL injuries tend to report less instability and swelling than patients with ACL injuries.
Collateral ligament injuries present with effusion or localized swelling. Lateral collateral ligament injuries result from a medial-to-lateral force on the knee, while medial collateral ligament injuries result from a force in the opposite direction. The amount of pain reported depends on the severity of the injury. A severe sprain is typically less painful than a partial tear, and instability is the major complaint. Meniscal injuries can occur in isolation or in combination with ligamentous injuries. The typical mechanism of injury involves weight bearing associated with a twisting of the knee, as in cutting or squatting movements. The patient may describe postexertional swelling, clicking and locking, and pain with rotational movements.
In all cases, it should be determined how quickly swelling occurred after the injury. If an effusion evolved within four hours of injury, there is a high likelihood of major osseous, ligamentous or meniscal injury 4. In a prospective study 5 of 106 cases of hemarthrosis caused by sporting injury, 71 patients (67 percent) had complete or partial disruption of the ACL. Another prospective study 6 of acutely injured yet clinically stable hemarthrotic knees revealed that patellar dislocation and ACL disruption accounted for 35 percent and 34 percent of the diagnoses, respectively.
If swelling occurred without a history of trauma, questioning should be directed at uncovering both local and systemic disorders. Overuse injuries, or repetitive microtrauma, occasionally present with knee swelling. Important questions to ask include which recreational or occupational activity was involved, how quickly the swelling occurred or resolved, and if any self-treatment had been attempted.
Systemic disorders often cause knee effusion. A variety of infectious diseases may present as monoarticular arthritis with joint redness, effusion and pain. Infiltrative disorders such as gout and pseudogout often present in a similar fashion. Immunologic diseases such as Reiter’s syndrome, rheumatoid arthritis and rheumatic fever can also cause knee effusion. The knee is the most common joint involved in both benign and malignant tumors 7.
Infectious arthritis, if untreated, often leads to irreversible joint damage. Typical historical clues include acute onset of symptoms, history of previously abnormal joint, immunocompromised status or intravenous drug use. Physical findings such as fever, warmth and erythema over the involved joint, coupled with the absence of ligamentous or meniscal findings, suggest an infectious etiology. Radiographs are typically obtained, although they often fail to reveal any abnormality.
The most important test is joint fluid evaluation. Findings indicating infection include effusion, white blood cell (WBC) count greater than 50,000 per mm³ (50 × 109 per L), organisms present on Gram stain and positive cultures. An elevated peripheral white blood cell and erythrocyte sedimentation rate also point to an infectious process. If the effusion recurs despite appropriate therapy, evaluation for fungal infection, tuberculosis and Lyme disease should be undertaken.
Crystal-induced arthritis can present in a similar fashion as infectious arthritis. The patient often describes periodic exacerbations of joint erythema, edema and swelling in the past. A history of diuretic use, a high purine diet, increased alcohol intake or renal stones is often associated with gouty arthritis. A diagnostic evaluation of the inflamed joint fluid is essential. Joint aspirate WBC count is in the range of 3,000 to 50,000 per mm³ (3 to 50 × 109 per L), and crystals are present when the fluid is examined with a polarizing microscope. The urate crystals seen in gout appear as negative birefringent rods or needles, while the calcium pyrophosphate crystals of pseudogout are weakly positive birefringent rectangles or rhomboids 8. The presence of crystals does not rule out an infectious cause, as the two may co-exist. The fluid should always be sent for Gram stain and culture.
Red Flags
Red flags include fever, non-weight bearing, loss of distal pulses, loss of sensation distal to the knee. These red flags typically need immediate evaluation.
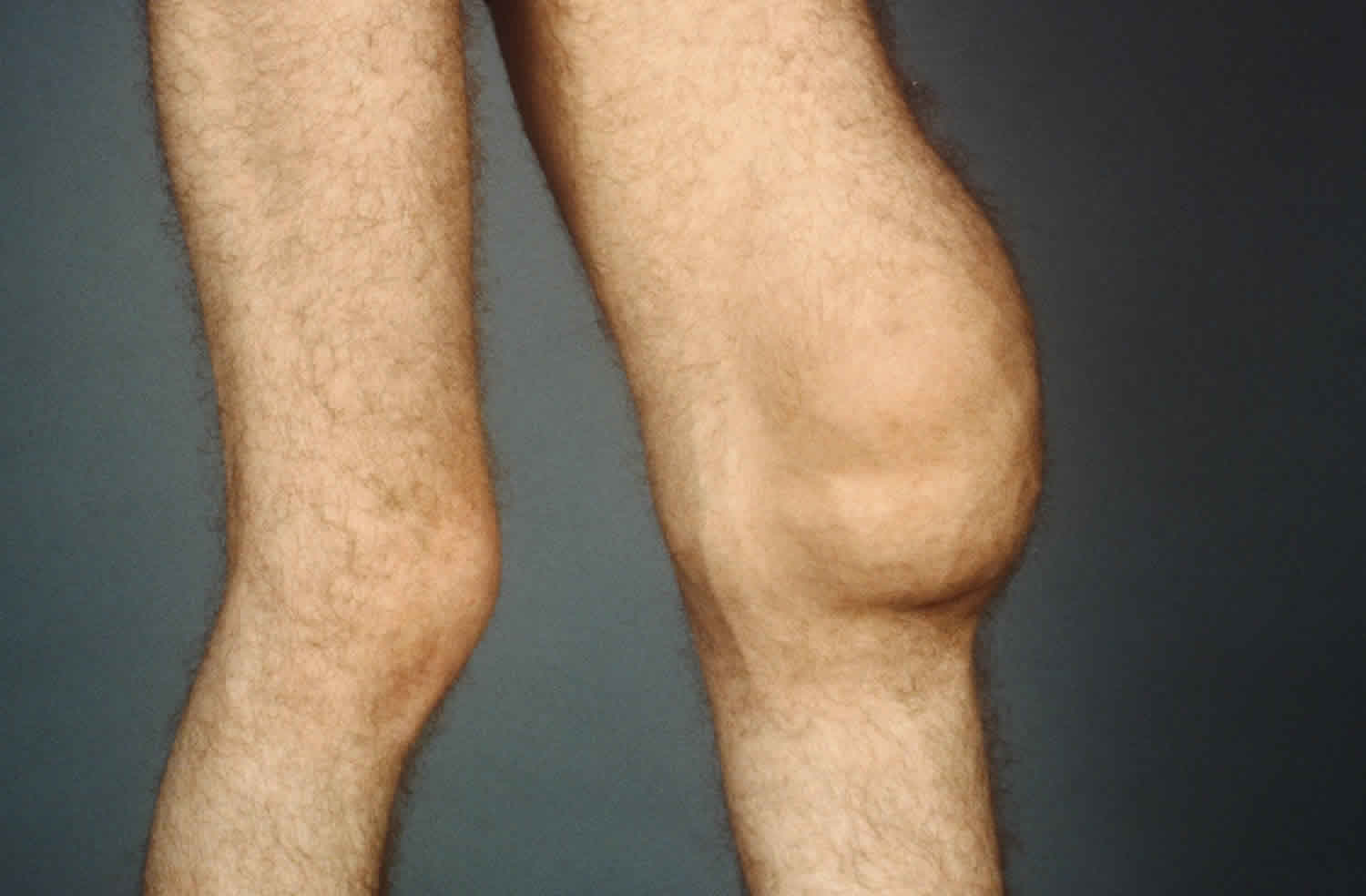
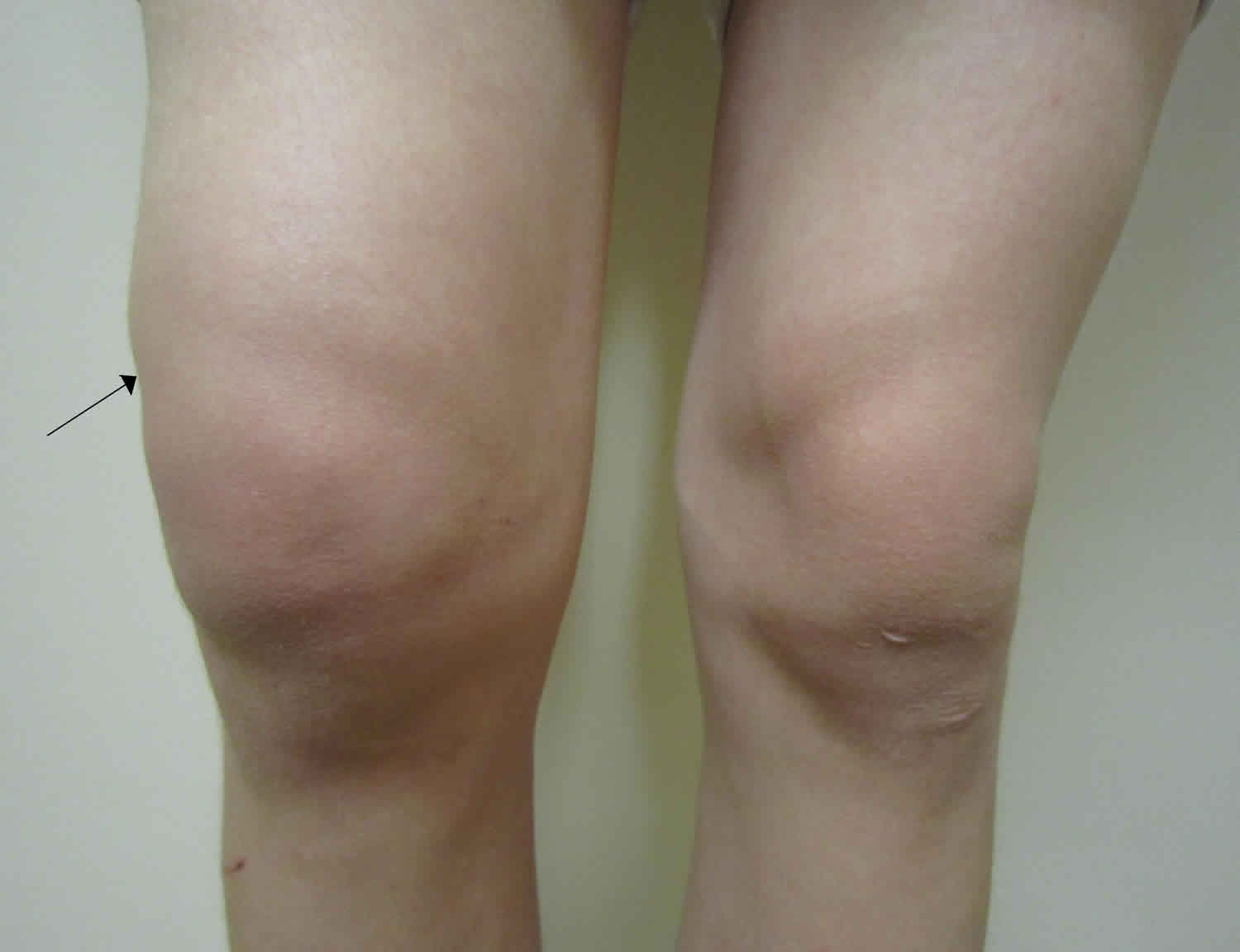
Figure 1. Knee effusion
Knee anatomy
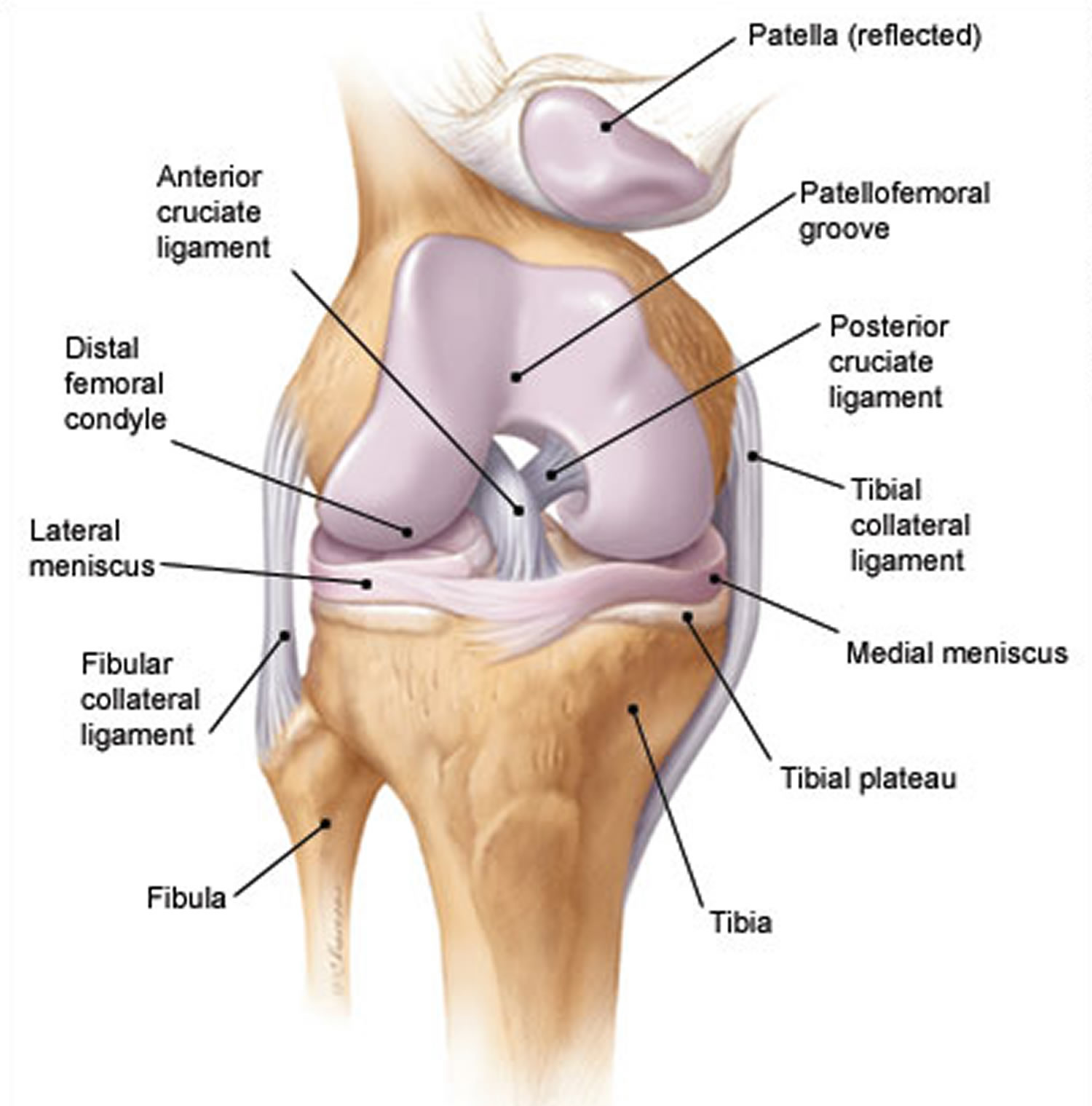
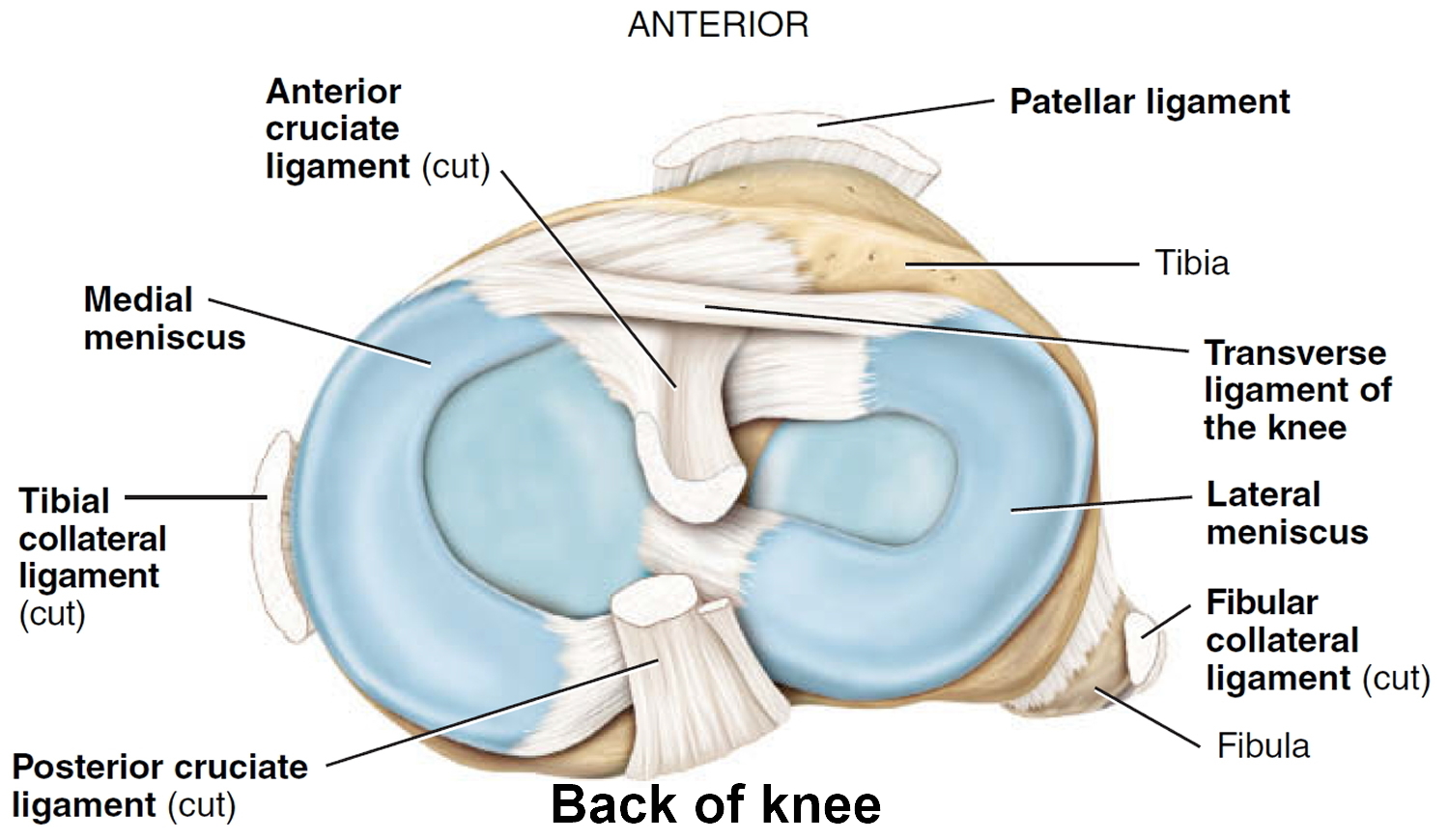
As with any musculoskeletal assessment, a precise understanding of knee pathoanatomy is essential. The knee is a compound condylar joint formed by three articulations: the first, between the medial femoral and tibial condyles, the second, between the lateral femoral and tibial condyles, and the third, between the patella and the femur. The knee moves through flexion and extension, internal and external rotation and a small degree of anterior-posterior glide.
Four major ligaments support the knee, which is the largest joint in the body. The anterior and posterior cruciate ligaments provide anterior and posterior stability, preventing dislocation of the tibia on the femur. The medial and lateral collateral ligaments provide stability to lateral and medial stresses, respectively (Figure 2).
The patella is the largest sesamoid bone in the body and provides increased mechanical advantage for knee extension. It is held in place by the medial and lateral retinaculae, and its posterior surface is covered with articular cartilage. The quadriceps muscles act on the knee through the quadriceps and patellar tendons.
The medial and lateral menisci are curved fibrocartilaginous structures located between the tibial and femoral articulating surfaces. They are connected to the joint capsule at their periphery and attached to the tibia via the coronary ligaments. The menisci act as shock absorbers within the knee joint. They also assist in lubrication of the joint and increase its elasticity. The medial meniscus is much less mobile than the lateral meniscus, accounting for its higher rate of injury 9.
Figure 2. Knee anatomy
Knee effusion causes
Any condition that may cause knee pathology can initially present as a knee effusion from a broad differential diagnosis. A knee effusion can be caused by an acute or chronic condition which may be inflammatory or non-inflammatory. The most common diagnoses in the primary care setting are osteoarthritis, trauma, and gout 10.
Knee effusions causes 11:
- Trauma
- Ligamentous injury
- Intra-articular fracture
- Patellar dislocation
- Meniscus injury
- Polyarthritis
- Reiter’s syndrome
- Juvenile rheumatoid arthritis
- Rheumatoid arthritis
- Infection
- Gonorrhea
- Lyme disease
- Tuberculosis
- Brucellosis
- Gout
- Pseudogout (calcium pyrophosphate deposition disease)
- Osteoarthritis and overuse syndrome
Tumor
- Malignant
- Hematologic
- Solid tumor
- Chondroblastoma
- Chondroblastoma
- Eosinophilic granuloma
- Giant cell tumor
- Ewing’s sarcoma
- Osteosarcoma
- Synovial sarcoma
- Benign
- Aneurysmal bone cyst
- Fibrous cortical defect
- Fibrous dysplasia
- Osteochondroma
- Osteoid osteoma
- Pigmented villonodular synovitis
Knee effusion diagnosis
The evaluation of an acutely swollen knee must begin with a very thorough history. Important questions to ask include mechanism of injury, duration, acuity of onset, aggravating symptoms, or if swelling occurred without trauma. A knee effusion with a history of recent injury may suggest a derangement such as a ligament or meniscal tear, while an atraumatic effusion would have a higher suspicion for septic arthritis. A history of previous surgery should be determined in every patient who presents with knee swelling. Systemic symptoms of inflammation or infection should be investigated as well. Patients commonly complain of swelling and stiffness with decreased range of motion 2.
Knee examination
A knee joint effusion will demonstrate swelling around the patella and distend of the suprapatellar space. Patients may have a restricted range of motion along with pain with ambulation. The exam should include observation of gait, palpation of the external knee, range of motion, joint line tenderness, McMurray tests, Thessaly test, duck walk, patellar tendon stability (ACL, PCL, valgus, varus), and a patellofemoral joint test (compression test) 12. Both the ballottement test and bulge test are done to look for knee effusion. The ballottement test is done by pressing upward on the medial aspect of the knee 2 to 3 times, then tapping the lateral patella to see if it floats outward due to effusion. Always compare the exam with the unaffected knee. In septic arthritis, the following symptoms are the only ones to occur in more than 50% of patients: joint pain, a history of joint swelling, and fever 13.
Imaging studies
The use of plain radiographs is often necessary to assess a swollen knee. In an acute injury, selecting the appropriate radiographic series is critical. In patients presenting with an acutely swollen knee, initially plain, weight-bearing radiographs in 2 planes should be ordered to look for a fracture, in case of trauma. A fabella, a sesamoid bone located inside the gastrocnemius may be seen on an x-ray. It is a radio-opaque marker for the posterior border of a knee’s synovium. The fabella sign or displacement of the fabella is seen with a synovial effusion and popliteal mass. The same lateral knee radiograph may show an increased opacity and widening of the suprapatellar bursa, which should be assessed if the fabella sign is seen. Another reason for plain radiographs is to detect erosive disease found in rheumatoid arthritis (RA), or joint space narrowing found both in osteoarthritis and rheumatoid arthritis 14.
An ultrasound of the effusion can help assess a complicated effusion from a simple effusion and can also be used to performed arthrocentesis. A Saline load test may be utilized to determine if a wound near a joint communicates with the joint. In the knee, 155 mL of saline is needed to reach 95% sensitivity.
Arthrocentesis
Arthrocentesis and subsequent synovial fluid analysis should be done in all cases of unexplained knee effusion. The aspirated fluid should be analyzed for cell counts, Gram stain, cultures, and crystal analysis. Hemarthrosis is commonly caused by joint trauma. Fat droplets (detected by polarized microscopy) also indicate an articular fracture. Other clotting disorders like hemophilia can cause hemarthrosis in the absence of trauma.
The synovial fluid aspirate should be analyzed for 15:
- Complete blood count (CBC) with differential (white blood cell [WBC], polymorphonuclear leukocytes)
- Crystal examination of synovial fluid
- Culture and Gram staining of synovial fluid
- Viscosity (rheumatoid arthritis: expect decreased viscosity and poor mucin clot formation)
- Glucose
- Low level of synovial-fluid glucose is suggestive of an infected joint, but low glucose levels are present in only about 50% of patients with septic joints and can also occur in rheumatoid arthritis
- Fasting glucose levels are usually reduced to less than half of the simultaneously obtained blood levels
- The presence of crystals cannot exclude septic arthritis with certainty 16. Septic arthritis occurs concurrently with gout or pseudogout in less than 5% of cases.
Septic arthritis
- Joint fluid appears cloudy or purulent
- Cell count with white blood cell greater than 50,000 is considered diagnostic for septic arthritis. However, lower counts may still indicate infection (not sensitive) 16
- Prosthetic joint with white blood cell greater than 1100 is considered septic
- Gram stains only identify infective organism one-third of time
- Glucose less than 50% of serum level
Non-inflammatory synovial fluid
- Contains less than 60 to 180 cells per mL, most of which should be mononuclear
- Synovial fluid is considered to be non-inflammatory if it contains less than 2000 cells/mL, but most samples of synovial fluids from pts with osteoarthritis contain less than 500 cells per ml.
- The most common cause of non-inflammatory effusions of the knee (synovial fluid white blood cell count less than 2000 cells/mcL) is osteoarthritis; other causes include osteonecrosis, Charcot arthropathy, sarcoidosis, amyloidosis, hypothyroidism, and acromegaly. Inflammatory arthritis (synovial fluid white blood cell greater than 2000 cells/mL) can be caused by infection, autoimmune disease, and crystal-induced arthritis. Aspiration of dark brown serosanguinous fluid should raise the possibility of pigmented villonodular synovitis.
Inflammatory synovial fluid
- Greater than 2000 leukocytes/mL
- Traumatic: Less than 5000 (with red blood cells)
- Toxic Synovitis: 5000 to 15,000 and less than 25% polymorphs
- Acute Rheumatic Fever: 10,000 to 15,000 and 50% polymorphs
- Juvenile rheumatoid arthritis 15,000 to 80,000 and 75% polymorphs
- Greater 50,000 leukocytes/mL;
- Although other diseases including trauma, may produce white blood cells in joint fluid, levels greater than 50,000/mm3 are usually due to infectious arthritis.
- Usually causes most intense synovial fluid leukocytosis, w/ 50,000 to 200,000 cells/mL and usually over 90% polymorphonuclear leukocytes
- Lower leukocyte counts are more common early in course of bacterial arthritis and in patients with disseminated gonococcal infection
- Non-infectious conditions such as gout, pseudogout, acute rheumatic fever, reactive arthritis, and rheumatoid arthritis can cause a markedly inflammatory synovial effusion. Finding of greater than 90%
- Polymorphonuclear leukocytes despite relatively low total leukocyte count should prompt concern about infection or crystal-induced disease. However, the presence of crystals cannot exclude septic arthritis with certainty 17.
- Septic arthritis 80,000 to 200,000 and greater than 75% polymorphs
- In synovial fluid white blood cell count and percentage of polymorphonuclear cells from arthrocentesis are the most powerful predictors for septic arthritis. The likelihood ratio is increased as the synovial fluid white blood cell count increased.
- For counts greater than 50,000/microL and for counts greater than 100,000/microL. On the same synovial fluid sample, a polymorphonuclear cell count of at least 90% suggests septic arthritis with an likelihood ratio of 3.4, while a polymorphonuclear leukocyte cell count of less than 90% lowers the likelihood 13.
Knee effusion treatment
Knee effusion treatment involves treating the underlying cause.
- Septic joint: Once the lab studies have been sent, start intravenous (IV) antibiotics for the suspected infective agent. The most common bacterial causes include staphylococci (40%), streptococci (28%), gram-negative bacilli (19%), mycobacteria (8 percent), gram-negative cocci (3%), gram-positive bacilli (1%), and anaerobes (1%). Also, orthopedic consult may be necessary. Drainage of the joint is associated with rapid recovery and low morbidity. Arthroscopy allows visualization of the joint, provides the ability to lyse adhesions, drains any purulent pockets, and can facilitate debridement of necrotic material if needed 18.
- Ligamentous injuries: The patient can be placed in a knee brace with an outpatient referral to an orthopedic surgeon.
- Fractures: These may need to be referred to a surgeon depending on severity. The Salter-Harris classification can be used for pediatric fractures involving the growth plate.
- Rheumatologic conditions: Anti-inflammatories (NSAIDs) or acetaminophen can be used, and the patient should be referred to a rheumatologist.
For acute pain and swelling, treatment should be individualized. This includes splints, cold or ice packs, partial or non-weight-bearing braces, non-steroidal anti-inflammatories (NSAIDs), or other analgesics. If the joint has a large effusion causing pain, drainage may be an effective treatment. The fluid sample should be obtained and tested/cultured before starting antibiotics. Intra-articular steroids should be held until infection, or other contraindications have been ruled out first 14.
Knee effusion prognosis
Poor outcomes may occur in septic arthritis if the patient has any of the following 19:
- Sixty years old or greater
- Affected area is the hip or shoulder joint
- Current or history of rheumatoid arthritis
- If the patient has been on appropriate therapy for 7 days and still has positive findings on synovial fluid cultures
- If greater than 7 days pass before starting treatment
Risks of delaying treatment beyond 24 to 48 hours include a permanent limitation in joint function and subchondral bone loss. Bacterial invasion can lead to permanent damage to articular cartilage. An infection in a prosthetic joint may result from a local infection that is left untreated and has spread to the prosthesis via blood flow to the area 18.
References- Gerena LA, DeCastro A. Knee Effusion. [Updated 2019 May 5]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2019 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK532279
- Gupte C, St Mart JP. The acute swollen knee: diagnosis and management. J R Soc Med. 2013 Jul;106(7):259-68.
- Baker P, Reading I, Cooper C, Coggon D. Knee disorders in the general population and their relation to occupation. Occup Environ Med. 2003 Oct;60(10):794-7.
- Sternbach GL. Evaluation of the knee. J Emerg Med. 1986;4:133–43.
- Maffulli N, Binfield PM, King JB, Good CJ. Acute haemarthrosis of the knee in athletes. A prospective study of 106 cases. J Bone Joint Surg [Br]. 1993;75:945–9.
- Visuri T, Koskenvuo M, Dahlstrom S. Hemarthrosis of the clinically stable knee due to sports and military training in young recruits: an arthroscopic analysis. Mil Med. 1993;158:378–81.
- Munn RK, Pierce ST, Sloan D, Weeks JA. Malignant joint effusions secondary to solid tumor metastasis. J Rheumatol. 1995;22:973–5.
- Bakerman S. ABC’s of interpretive laboratory data. 2d ed. Greenville, N.C.: Interpretive Laboratory Data, 1984:60–2.
- Morgan GJ. Joint examination: a study guide for medical students and house staff Hanover, N.H.: Arthritis Center Publications,1988:22–9.
- Becker JA, Daily JP, Pohlgeers KM. Acute Monoarthritis: Diagnosis in Adults. Am Fam Physician. 2016 Nov 15;94(10):810-816.
- Acute Knee Effusions: A Systematic Approach to Diagnosis. Am Fam Physician. 2000 Apr 15;61(8):2391-2400. https://www.aafp.org/afp/2000/0415/p2391.html
- Solomon DH, Simel DL, Bates DW, Katz JN, Schaffer JL. The rational clinical examination. Does this patient have a torn meniscus or ligament of the knee? Value of the physical examination. JAMA. 2001 Oct 03;286(13):1610-20.
- Margaretten ME, Kohlwes J, Moore D, Bent S. Does this adult patient have septic arthritis? JAMA. 2007 Apr 04;297(13):1478-88.
- Landewé RB, Günther KP, Lukas C, Braun J, Combe B, Conaghan PG, Dreinhöfer K, Fritschy D, Getty J, van der Heide HJ, Kvien TK, Machold K, Mihai C, Mosconi M, Nelissen R, Pascual E, Pavelka K, Pileckyte M, Puhl W, Punzi L, Rüther W, San-Julian M, Tudisco C, Westhovens R, Witso E, van der Heijde DM. EULAR/EFORT recommendations for the diagnosis and initial management of patients with acute or recent onset swelling of the knee. Ann. Rheum. Dis. 2010 Jan;69(1):12-9.
- Li SF, Henderson J, Dickman E, Darzynkiewicz R. Laboratory tests in adults with monoarticular arthritis: can they rule out a septic joint? Acad Emerg Med. 2004 Mar;11(3):276-80.
- McGillicuddy DC, Shah KH, Friedberg RP, Nathanson LA, Edlow JA. How sensitive is the synovial fluid white blood cell count in diagnosing septic arthritis? Am J Emerg Med. 2007 Sep;25(7):749-52.
- Shah K, Spear J, Nathanson LA, McCauley J, Edlow JA. Does the presence of crystal arthritis rule out septic arthritis? J Emerg Med. 2007 Jan;32(1):23-6.
- Horowitz DL, Katzap E, Horowitz S, Barilla-LaBarca ML. Approach to septic arthritis. Am Fam Physician. 2011 Sep 15;84(6):653-60.
- Kaandorp CJ, Krijnen P, Moens HJ, Habbema JD, van Schaardenburg D. The outcome of bacterial arthritis: a prospective community-based study. Arthritis Rheum. 1997 May;40(5):884-92.