What is limbic encephalitis
Limbic encephalitis represents a group of autoimmune conditions characterized by inflammation of the limbic system and other parts of the brain. The main regions of the limbic system include the hippocampus and amygdala. The limbic areas of the brain control many functions including memory, learning, and emotions such as aggression. In addition, some of these limbic areas are susceptible to seizures, which are a common feature of limbic encephalitis.
The cardinal sign of limbic encephalitis is a severe impairment of short-term memory; however, symptoms may also include confusion, psychiatric symptoms, and seizures. The symptoms typically develop over a few weeks or months, but they may evolve over a few days. Limbic encephalitis is often associated with an underlying neoplasm (paraneoplastic limbic encephalitis); however some cases never have a neoplasm identified (non-paraneoplastic limbic encephalitis).
Initially limbic encephalitis was considered a rare disorder in association with cancer (paraneoplastic limbic encephalitis); most commonly associated with small cell lung cancer (SCLC), breast cancer, testicular tumors, teratomas, Hodgkin’s lymphoma and thymomas but can occur in the absence of malignancy also 1. About 60-75% of patients neurological symptoms precedes the diagnosis of malignancy 2.
Limbic encephalitis can have varied presentation and delayed diagnosis is common, but improvements are being made to assist in early detection. The improvements in neuroimaging studies (MRI, PET) and identification of antineuronal antibodies in patients (CSF analysis) and tests that measure the electrical activity of the brain (EEG) have facilitated to confirm a diagnosis. These antineuronal antibodies are directed against (1) intracellular or classic paraneoplastic antigens, including Hu, Ma2, CV2/collapsin response-mediator protein 5 and amphiphysin among others and (2) cell membrane antigens, including voltage-gated potassium channels (VGKC), N-methyl-D-aspartate (NMDA) receptor and others expressed in the neuropil of hippocampus and cerebellum 1, 3.
Treatment of limbic encephalitis includes removal of the neoplasm (if identified) and immunotherapy 4, 5, 6.
The Limbic System
The limbic system is sometimes called the “emotional brain” because it plays a primary role in a range of emotions, including pain, pleasure, docility, affection, and anger. It also is involved in olfaction (smell), learning and memory.
Together with parts of the cerebrum, the limbic system also functions in memory; damage to the limbic system causes memory impairment. One portion of the limbic system, the hippocampus, is seemingly unique among structures of the central nervous system—it has cells reported to be capable of mitosis. Thus, the portion of the brain that is responsible for some aspects of memory may develop new neurons, even in the elderly.
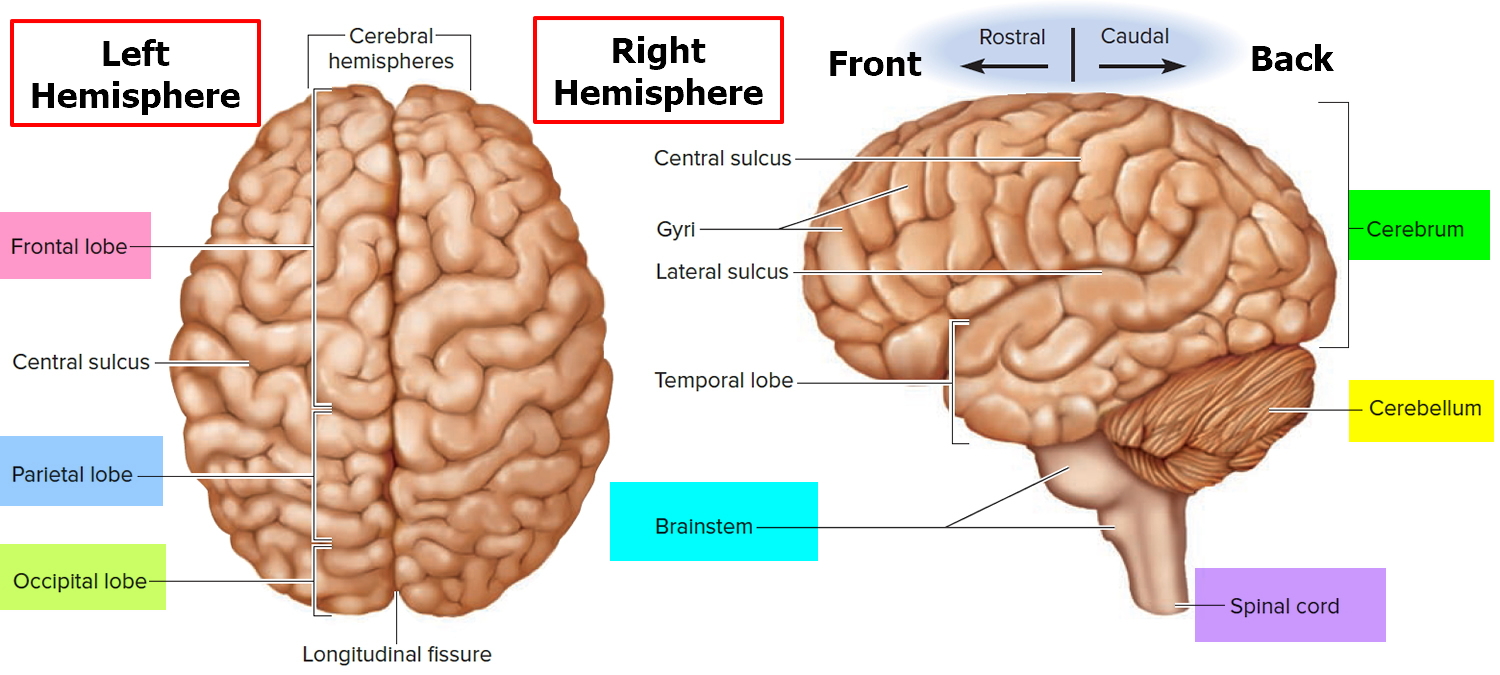
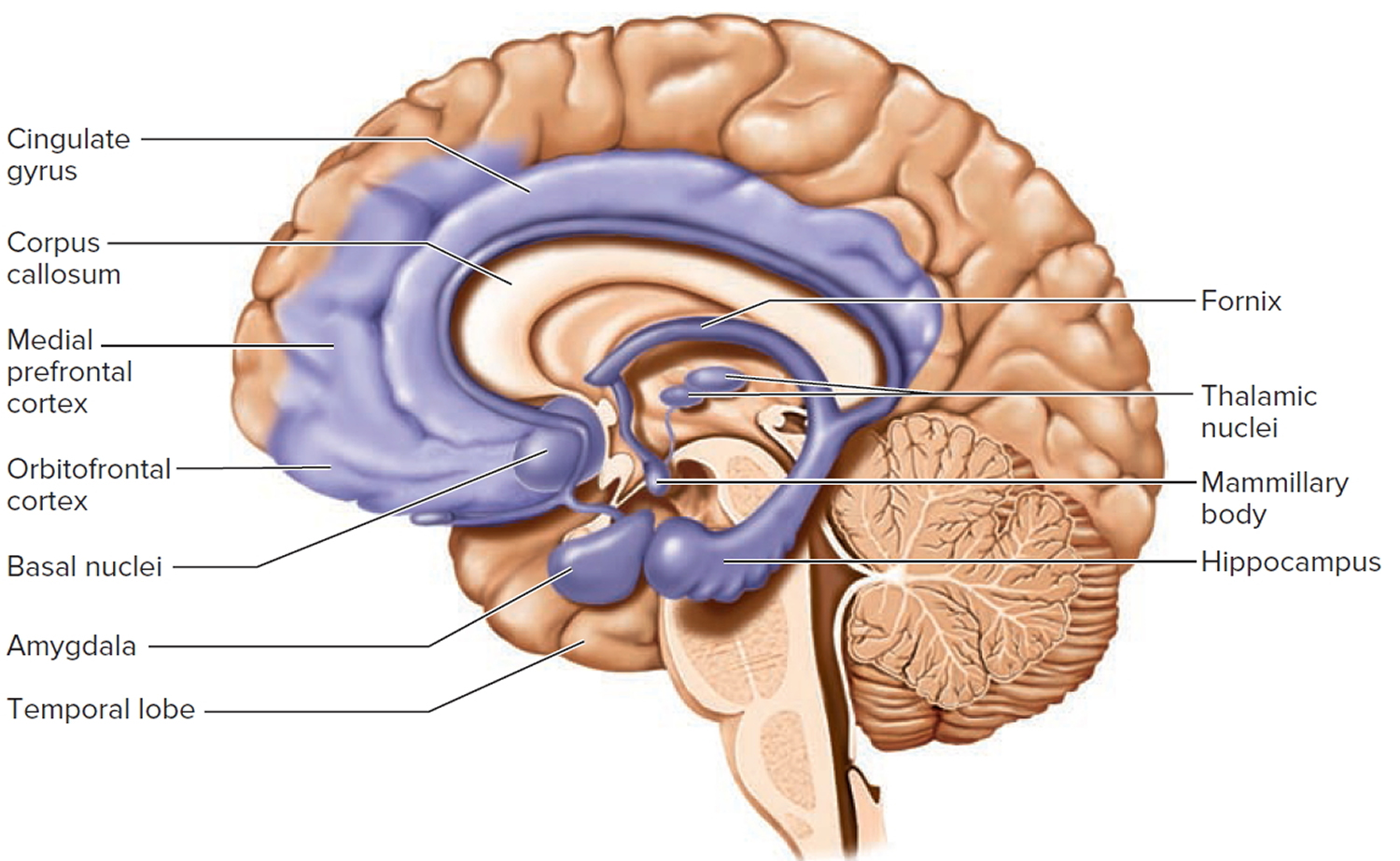
The limbic system is a ring of cortex on the medial side of each hemisphere, encircling the corpus callosum and thalamus. The main components of the limbic system are as follows:
- The so-called limbic lobe is a rim of cerebral cortex on the medial surface of each hemisphere. It includes the cingulate gyrus, which lies above the corpus callosum, and the parahippocampal gyrus, which is in the temporal lobe below. The hippocampus is a portion of the parahippocampal gyrus that extends into the floor of the lateral ventricle.
- The dentate gyrus lies between the hippocampus and parahippocampal gyrus.
- The amygdala is composed of several groups of neurons located close to the tail of the caudate nucleus.
- The septal nuclei are located within the septal area formed by the regions under the corpus callosum and the paraterminal gyrus (a cerebral gyrus).
- The mammillary bodies of the hypothalamus are two round masses close to the midline near the cerebral peduncles.
- Two nuclei of the thalamus, the anterior nucleus and the medial nucleus, participate in limbic circuits.
- The olfactory bulbs are flattened bodies of the olfactory pathway that rest on the cribriform plate.
- The fornix, stria terminalis, stria medullaris, medial forebrain bundle, and mammillothalamic tract are linked by bundles of interconnecting myelinated axons.
Its most anatomically prominent components are the cingulate gyrus, which arches over the top of the corpus callosum in the frontal and parietal lobes; the hippocampus in the medial temporal lobe; and the amygdala immediately rostral to the hippocampus, also in the temporal lobe. There are still differences of opinion on what structures to consider as parts of the limbic system, but these three are agreed upon.
Other components include the mammillary bodies and other hypothalamic nuclei, some thalamic nuclei, parts of the basal nuclei, and parts of the frontal lobe called prefrontal and orbitofrontal cortex. Limbic system components are interconnected through a complex loop of fiber tracts allowing for somewhat circular patterns of feedback among its nuclei and cortical neurons. All of these structures are bilaterally paired; there is a limbic system in each cerebral hemisphere.
The limbic system was long thought to be associated with smell because of its close association with olfactory pathways, but beginning in the early 1900s and continuing even now, experiments have abundantly demonstrated more significant roles in emotion and memory. Most limbic system structures have centers for both gratification and aversion. Stimulation of a gratification center produces a sense of pleasure or reward; stimulation of an aversion center produces unpleasant sensations such as fear or sorrow. Gratification centers dominate some limbic structures, such as the nucleus accumbens (not illustrated), while aversion centers dominate others such as the amygdala. The roles of the amygdala in emotion and the hippocampus in memory.
Figure 1. Human brain
Figure 2. Limbic system
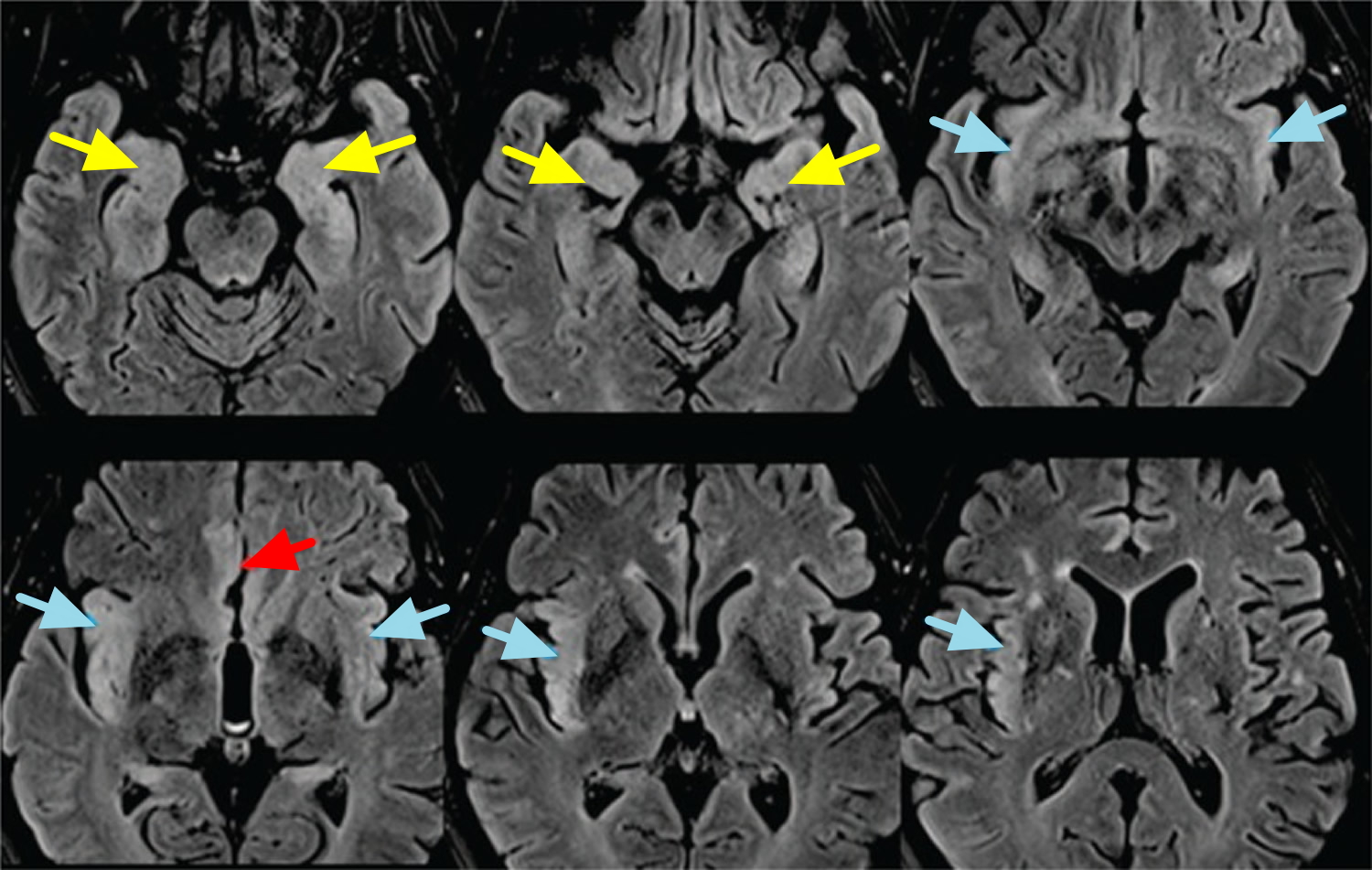
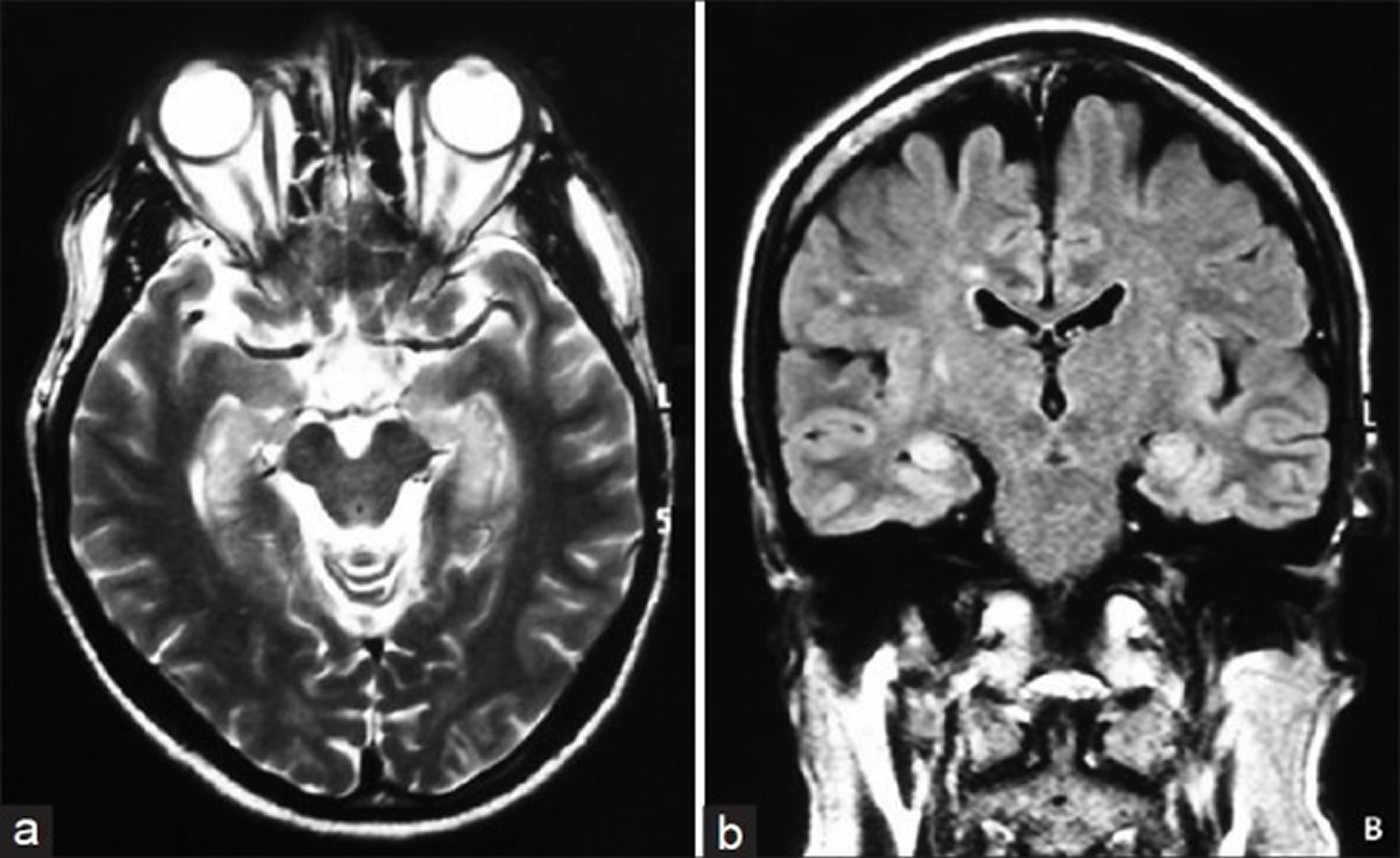
Note: Mesial temporal lobes (yellow arrows), cingulate gyrus (red arrow), and insulae (blues arrows) involvement with asymmetric (right > left) cortical thickening and FLAIR hyperintensity.
Figure 4. Limbic encephalitis MRI (autoimmune limbic encephalitis)
Note: Bilateral mesial temporal lobe increased T2 signal is demonstrated, more marked on the right, and seen extending in along the hippocampal head, body and tail, but also involving the fornicies, amygdala, and parahippocampal gyrus.
Figure 5. Limbic encephalitis MRI (autoimmune limbic encephalitis associated with antibodies against γ-aminobutyric acid B receptor [GABAB receptor])
Note: (a) T2-weighted magnetic resonance imaging showing increased signal in the mesial temporal lobes; (b) Fluid-attenuated inversion recovery magnetic resonance imaging show increased signal in the mesial temporal lobes.
Limbic encephalitis causes
Most forms of limbic encephalitis fall into two main categories:
- Infectious encephalitis – caused by direct invasion of the limbic area of the brain by a bug, usually a virus.
- Autoimmune encephalitis – caused by the person’s own immune system reacting against parts of the limbic system.
1. Infectious limbic encephalitis causes
Many infections of the brain can potentially cause inflammation of the limbic areas. A number of viruses, such as the herpes simplex virus (HSV) seem to preferentially target this area. Some people may therefore be given the diagnosis of limbic encephalitis whilst others are given the diagnosis herpes simplex encephalitis for the same condition. A clearer way for people would be to say that the person has ‘herpes simplex virus encephalitis affecting mainly the limbic areas of the brain’, but this is rather long-winded.
2. Autoimmune limbic encephalitis causes
A major role of our immune system is to recognize and eliminate infection. But sometimes parts of the immune system called ‘antibodies’ may instead react with proteins of our own body to cause autoimmune diseases. When this reaction is against proteins of the limbic areas of the brain, this is called ‘autoimmune limbic encephalitis’.
There are broadly two forms of autoimmune limbic encephalitis: paraneoplastic limbic encephalitis and non-paraneoplastic limbic encephalitis.
Diagnostic criteria for definite autoimmune limbic encephalitis
Diagnosis can be made when all four* of the following criteria have been met 7:
- Subacute onset (rapid progression of less than 3 months) of working memory deficits, seizures, or psychiatric symptoms suggesting involvement of the limbic system
- Bilateral brain abnormalities on T2-weighted fluid-attenuated inversion recovery MRI highly restricted to the medial temporal lobes†
- At least one of the following:
- CSF pleocytosis (white blood cell count of more than five cells per mm3)
- EEG with epileptic or slow-wave activity involving the temporal lobes
- Reasonable exclusion of alternative causes.
In the proposed diagnostic criteria, antibody status is not needed to consider limbic encephalitis as having a definite autoimmune origin because immune-mediated limbic encephalitis can occur without detectable autoantibodies 8, 9. Measurement of autoantibodies, however, remains important for two reasons: their presence clarifies the immunological subgroup of limbic encephalitis, with comorbidities, tumor association, and prognosis that might differ according to the autoantibody 10; and, in patients who do not satisfy the indicated criteria, detection of autoantibodies establishes the diagnosis of autoimmune limbic encephalitis (Table 1).
Table 1. Antibodies in the diagnosis of autoimmune encephalitis
| Syndrome | Diagnostic assay | Frequency of cancer | Main type of cancer | |
|---|---|---|---|---|
| Antibodies against intracellular antigens | ||||
| Hu (ANNA1)11* | Limbic encephalitis | Western blot | >95% | Small-cell lung carcinoma |
| Ma212 | Limbic encephalitis† | Western blot | >95% | Testicular seminoma |
| GAD13 | Limbic encephalitis‡ | Radioimmunoassay | 25%§ | Thymoma, small-cell lung carcinoma |
| Antibodies against synaptic receptors | ||||
| NMDA receptor14 | Anti-NMDA receptor encephalitis | Cell-based assay | Varies with age and sex | Ovarian teratoma¶ |
| AMPA receptor15 | Limbic encephalitis | Cell-based assay | 65% | Thymoma, small-cell lung carcinoma |
| GABAB receptor16 | Limbic encephalitis | Cell-based assay | 50% | Small-cell lung carcinoma |
| GABAA receptor17 | Encephalitis | Cell-based assay | <5% | Thymoma |
| mGluR518 | Encephalitis | Cell-based assay | 70% | Hodgkin’s lymphoma |
| Dopamine 2 receptor19 | Basal ganglia encephalitis | Cell-based assay | 0% | .. |
| Antibodies against ion channels and other cell-surface proteins | ||||
| LGI120 | Limbic encephalitis | Cell-based assay | 5–10% | Thymoma |
| CASPR221 | Morvan’s syndrome|| or limbic encephalitis | Cell-based assay | 20–50% | Thymoma** |
| DPPX22 | Encephalitis†† | Cell-based assay | <10% | Lymphoma |
| MOG23‡‡ | Acute disseminated encephalomyelitis | Cell-based assay | 0% | .. |
| Aquaporin 4 24‡‡ | Encephalitis | Cell-based assay | 0% | .. |
| GQ1b25 | Bickerstaff’s brainstem encephalitis | ELISA | 0% | .. |
GAD=glutamic acid decarboxylase. LGI1=leucine-rich glioma inactivated 1. CASPR2=contactin associated protein 2. DPPX=dipeptidyl-peptidase-like protein-6. MOG=myelin oligodendrocyte glycoprotein.
a) Paraneoplastic limbic encephalitis
Sometimes when the immune system starts to react with the limbic areas, this happens because the person has a tumor (neoplasm) in their body which activates the immune system. This activated immune system can, in turn, attack the brain. Doctors call this paraneoplastic limbic encephalitis as the tumor affects the brain from a distance, via the immune system.
In many cases, paraneoplastic limbic encephalitis can be diagnosed by testing for one of paraneoplastic autoantibodies in the patient’s blood and spinal fluid. Most individuals with paraneoplastic limbic encephalitis have a cancer of the lung, thymus gland, ovary, breast or testes. More rarely, other cancers can initiate the condition. The outcome is very dependent on the underlying tumor and the precise condition, often classified by the antibody. In some cases, the condition may improve or at least stabilize if the cancer is detected and treated effectively. However, unfortunately, in many cases treatment does not improve the patient’s neurological symptoms, probably because the immune system has irreversibly damaged the brain cells and the tumor cannot be controlled successfully.
b) Non-paraneoplastic limbic encephalitis
Non-paraneoplastic limbic encephalitis has only been clearly recognized in the last few years when doctors began to identify patients with symptoms similar to those with paraneoplastic limbic encephalitis but who did not have any of the marker paraneoplastic antibodies in their blood and never developed a tumor. Non-paraneoplastic limbic encephalitis is far more common than paraneoplastic limbic encephalitis.
It is becoming increasingly clear that non-paraneoplastic limbic encephalitis is caused, at least in part, by specific antibodies in the patient’s blood that target the patient’s brain, particularly the hippocampus and other limbic areas. Many of these patients improve if they are treated with drugs that suppress the immune system and reduce the levels of the antibodies. These drugs include steroids, intravenous immunoglobulins and plasma exchange.
As these autoimmune diseases have only been recently described, there is still much to be done to raise awareness amongst clinicians. Future research aims to understand the biological mechanisms by which this antibody affects the excitability of the brain, and hence causes disease. Researchers also hope to discover further antibodies which may allow other autoimmune encephalitis forms to be diagnosed.
Types of antibodies
A number of specific brain protein targets for these antibodies have been discovered over the last years and this variety may explain why people have different symptoms. The main established antibodies and their associated features are described below:
LGI1/CASPR2 (previously termed voltage-gated potassium channel complex antibodies)
These two forms of limbic encephalitis, LGI1/CASPR2 antibody encephalitis (previously termed VGKC/voltage-gated potassium channel antibody encephalitis), are associated with LGI1 (leucine-rich-glioma inactivated 1) and CASPR2 (contactin-associated protein 2) antibodies. Your immune system works to clear your bodies of foreign invaders such as infections. It does this, in part, by producing antibodies which attach to and help destroy the invaders. If the brain is the mistaken region, the antibody attack produces an autoimmune encephalitis. When the antibodies target LGI1 (leucine-rich glioma inactivated 1) or CASPR2 (contactin-associated protein 2), they cause an encephalitis often termed limbic encephalitis. Men are roughly affected twice as often as women.
LGI1/CASPR2-antibody encephalitis can be mistaken for many other diseases, including viral encephalitis or other autoimmune conditions. Most patients are investigated with:
- blood tests (patients often have a low salt level in their blood)
- lumbar puncture (LP) to help exclude infection which is often normal in LGI1/CASPR2-antibody encephalitis
- electrical recordings of the brain (EEG) to look for seizure activity
- magnetic resonance imaging (MRI) scans which often show swelling and inflammation of the brain
Patients with both conditions may have tumors which are related to their condition. This is especially common in patients with CASPR2 antibodies.
To obtain a definitive diagnosis with the above clinical features, the LGI1/CASPR2-antibodies can be measured in blood and, often, spinal fluid.
Patients who have been tested for voltage gated potassium (VGKC) channel-antibodies should have additional LGI1 and CASPR2-antibody testing. These additional tests ensure the relevance of their antibodies to their condition and largely prevent misdiagnosis of the condition.
LGI1/CASPR2-antibody encephalitis prognosis
As this disease was only relatively recently described, there is still much to learn about what happens to patients in the long-term.
Almost all patients make some improvements but very few patients are left with no residual problems after treatments. Patients are left with problems in memory, seizures and behaviour which may require medical and psychological input and, occasionally, may necessitate long-term placement in a home or other care facility. Other patients may return to their previous functioning, including getting back to work, some months or years after treatment. The time to treatment with immunosuppressive drugs is the only known factor in determining outcome.
LGI1/CASPR2-antibody encephalitis treatment
LGI1/CASPR2-antibody encephalitis is a serious disease. Patients often spend some weeks in hospital receiving specialist treatment and assessment. If the antibodies are found in a patient with the recognised clinical features, LGI1/CASPR2-antibody encephalitis can be treated by dampening down the immune reaction that is causing the inflammation using immunosuppression.
However, no single set of medications is proven to be superior to others and research into new or optimal treatments is ongoing. Nevertheless, most clinicians opt to use immunosuppression with steroids (either taken as tablets or into a vein), intravenous immunoglobulin (a blood product given into the vein in a drip) and/or plasma exchange (when some of a person’s blood is taken out from a vein, washed of various components including antibodies, and then put back into the vein in a drip). All these treatments have known side-effects and their benefits need to be weighed against possible side-effects in individual patients.
Typically, patients are given further tablet steroid treatments after discharge from hospital.
Treatment often results in fewer seizures and some improvement in confusion and memory problems. Improvements may be seen within a few days of treatment or in some cases only noticed after several months. Treatment, typically with steroid tablets, may be continued for 1-2 years. In addition, patients may receive medications which suppress seizures (anti-epileptic drugs).
Relapses of LGI1/CASPR2-antibody encephalitis
In around 90% of cases (although the exact figures are not yet known) LGI1/CASPR2-antibody encephalitis will not recur. Unfortunately, in the remaining 10% of cases it may recur with many of the same symptoms as before (such as seizures, confusion or forgetfulness). It is not yet known what leads to this recurrence but it usually warrants repeat treatments that might range from steroids into the vein (intravenous methylprednisolone) through to plasma exchange.
AMPAR and GABAB/AR antibodies
Antibodies against two other receptors in the brain, AMPA and GABAA/B, are less common causes of autoimmune limbic encephalitis. Although the majority of these patients have an underlying tumour, this is a form of Paraneoplastic Limbic Encephalitis that can often respond to treatment relatively well.
NMDAR-antibodies
Another antibody that can cause paraneoplastic limbic encephalitis is the N-methyl-D-aspartate (NMDA) receptor antibody. This disease may be associated with a growth such as a cancer in around 30% of cases. This antibody usually causes encephalitis involving several brain regions, but it can sometimes cause a pure limbic encephalitis. The N-methyl-D-aspartate (NMDA) receptor is a protein in the brain that helps control thoughts, mood and movements, and therefore antibodies against NMDA receptors are likely to have an important role in altering these functions. Anti-NMDAR encephalitis affects the brain more diffusely than purely the limbic system, and therefore it is not classified as a limbic encephalitis.
It should be noted that the brain imaging and the routine lumbar puncture results may be normal in autoimmune limbic encephalitis.
Anti-NMDAR encephalitis symptoms
At onset, the most distinctive features include prominent psychiatric symptoms, seizures, confusion and memory loss. Patients will sometimes show bizarre and often rather disturbing behaviors. They may see things which aren’t there, develop strange beliefs or appear agitated. Typically 10 to 20 days later, patients develop a movement disorder, variations in blood pressure, heart rate and temperature and may become less conscious. The movement disorder often consists of continuous writhing and twitching of face and limbs but can also be a generalized slowing-down of movement. Most patients develop several of these features, but very rarely individual patients may experience only a few of these features.
Anti-NMDAR encephalitis diagnosis
The symptoms and signs seen in patients with anti-NMDAR encephalitis can be distinctive and are prompting many clinicians to request the NMDA receptor antibody test to diagnose this condition. The disease mainly affects young people, with around 30% of cases under 18 years of age. Women are affected more often than men.
Once a patient has been diagnosed with anti-NMDAR encephalitis, an underlying tumour should be looked for. While very few men have tumors detected (typically <5%), recent reports suggest that around 20-30% of women have an underlying tumor. The most common tumor found in women is called an ovarian teratoma. This is usually a non-cancerous tumor but is thought to stimulate the production of the NMDA receptor antibody.
Anti-NMDAR encephalitis treatment
If these symptoms and signs are recognized, other causes excluded (particularly infections) and the antibody is found in the cerebrospinal (CSF) fluid, treatments should be started. Treatment consists of immune therapies and removal of a tumor, if present.
The immune therapies used to dampen down the immune system include steroids (drugs to reduce inflammation), immunoglobulins (a blood product given into the vein in a drip) and plasma exchange (when some of a person’s blood is taken out from a vein, and the plasma part of the blood which contains antibodies is separated and replaced with new plasma and then put back into the vein in a drip).
In addition, some patients are treated with other drugs which dampen down the immune system. All drugs have known side-effects but their benefits are generally felt to outweigh possible side-effects.
Prompt therapies offer a good chance of substantial recovery in the majority of patients. As they improve, there is often a reduction in the amount of NMDA receptor antibody in the person’s blood or spinal fluid when the test is repeated. Some patients are now being treated after recognition of the clinical symptoms and signs, while the antibody result is awaited, to try to expedite recovery. However, recovery is usually slow and many patients spend a few months in hospital, including time on the intensive care unit undergoing ventilation. Early in the illness, it is important that doctors realize there may be few or no signs of recovery despite treatment. Those who return to work typically only do so after a year or two but most patients have some problems with memory, mood or behavior which can be significant or subtle.
In summary, anti-NMDAR encephalitis is a recently identified autoimmune disease that causes psychiatric features, confusion, memory loss and seizures followed by a movement disorder, loss of consciousness and changes in blood pressure, heart rate and temperature. The disease can respond well to various therapies that dampen down the immune system and the removal of an underlying tumor if one is found, but improvement is often slow.
Limbic encephalitis symptoms
Although the symptoms of the condition may vary from person to person, the presenting sign of limbic encephalitis is severe impairment of short-term memory, with most patients having difficulties in recall. Epileptic seizures are common and may occur prior to symptoms of memory loss 5. A variety of symptoms may be associated with limbic encephalitis such as anterograde amnesia (the inability to store new memories after the onset of the condition), anxiety, depression, irritability, personality or behavioral change, acute confusional state, hallucinations and seizures.
Other possible symptoms may include obsessiveness, hyperthermia (increase in body temperature), weight change, sleep disturbances, endocrine dysfunction, aphasia, and apraxia 27. The symptoms associated with limbic encephalitis can develop over a few days, weeks, or months. It is important to note the neurological symptoms generally precede diagnosis of the malignancy in 60%-75% of patients that have paraneoplastic limbic encephalitis 4.
Limbic encephalitis prognosis
The prognosis for limbic encephalitis varies from person to person and is based on the underlying cause of the condition. For example, individuals that have small cell lung carcinoma (SCLC) and test positive for an antibody called anti-Hu, tend to respond poorly to treatment, whereas individuals that have an ovarian teratoma and test positive for the NMDAR (N-methyl-D-aspartate receptor) antibody respond well to treatment 4.
Given evidence that paraneoplastic limbic encephalitis may occur prior to identification of a tumor, if a tumor is not initially identified, regular tumor screening may be recommended to occur after treatment is completed 5.
Limbic encephalitis treatment
Treatment for limbic encephalitis varies depending on the underlying cause. In cases of confirmed paraneoplastic limbic encephalitis, removal or treatment of the tumor is often the first step in treatment. If the cause of limbic encephalitis is a viral infection, an antiviral drug may be prescribed. Immunotherapy is often utilized as a first line or second line treatment 28. If immunotherapy fails, medications such as rituximab or cyclophosphamide may be considered 5.
Treatments of autoimmune limbic encephalitis
The diagnosis of autoimmune encephalitis is particularly important because the disease is potentially treatable with medicines that dampen down the immune system. These medications are called immunosuppressive and include steroids, immunoglobulins (a blood product given into the vein in a drip) and plasma exchange (when some of a person’s blood is taken out from a vein, and the plasma part of the blood which contains antibodies is separated and replaced with new plasma and then put back into the vein in a drip). All these drugs have known side-effects but their benefits are generally felt to outweigh possible side-effects in these conditions. Ongoing research is trying to understand how to best target the cells which produce antibodies and hence tailor therapies in patients with autoimmune encephalitis.
References- Höftberger R, Titulaer MJ, Sabater L, Dome B, Rózsás A, Hegedus B, et al. Encephalitis and GABAB receptor antibodies: Novel findings in a new case series of 20 patients. Neurology. 2013;81:1500–6. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3888170/
- Billinton A, Ige AO, Wise A, White JH, Disney GH, Marshall FH, et al. GABA(B) receptor heterodimer-component localisation in human brain. Brain Res Mol Brain Res. 2000;77:111–24. https://www.ncbi.nlm.nih.gov/pubmed/10814837
- Benarroch EE. GABAB receptors: Structure, functions, and clinical implications. Neurology. 2012;78:578–84. https://www.ncbi.nlm.nih.gov/pubmed/22351795
- Anderson NE, Barber PA. Limbic encephalitis – a review. Journal of Clinical Neuroscience. 2008; http://www.ncbi.nlm.nih.gov/pubmed/18411052
- Asztely F, Kumlien E.. The diagnosis and treatment of limbic encephalitis. Acta Neurol Scand. Dec 2012; 126(6):365-375. http://www.ncbi.nlm.nih.gov/pubmed/22713136
- Dr Sarosh R Irani, Dr Camilla Buckley and Prof Angela Vincent. Limbic Encephalitis. The Encephalitis Society. September 2017; https://www.encephalitis.info/limbic-encephalitis
- Graus F, Titulaer MJ, Balu R, et al. A clinical approach to diagnosis of autoimmune encephalitis. The Lancet Neurology. 2016;15(4):391-404. doi:10.1016/S1474-4422(15)00401-9. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5066574
- Graus F, Saiz A, Lai M, et al. Neuronal surface antigen antibodies in limbic encephalitis: clinical-immunologic associations. Neurology. 2008;71:930–36 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2586945/
- Najjar S, Pearlman D, Zagzag D, Devinsky O. Spontaneously resolving seronegative autoimmune limbic encephalitis. Cogn Behav Neurol. 2011;24:99–105 https://www.ncbi.nlm.nih.gov/pubmed/21677575
- Alamowitch S, Graus F, Uchuya M, Reñé R, Bescansa E, Delattre JY. Limbic encephalitis and small cell lung cancer. Clinical and immunological features. Brain. 1997;120:923–28. https://www.ncbi.nlm.nih.gov/pubmed/9217677
- Alamowitch S, Graus F, Uchuya M, Reñé R, Bescansa E, Delattre JY. Limbic encephalitis and small cell lung cancer. Clinical and immunological features. Brain. 1997;120:923–28.
- Dalmau J, Graus F, Villarejo A, et al. Clinical analysis of anti-Ma2-associated encephalitis. Brain. 2004;127:1831–44
- Malter MP, Helmstaedter C, Urbach H, Vincent A, Bien CG. Antibodies to glutamic acid decarboxylase define a form of limbic encephalitis. Ann Neurol. 2010;67:470–78
- Dalmau J, Lancaster E, Martinez-Hernandez E, Rosenfeld MR, Balice-Gordon R. Clinical experience and laboratory investigations in patients with anti-NMDAR encephalitis. Lancet Neurol. 2011;10:63–74.
- Lai M, Hughes EG, Peng X, et al. AMPA receptor antibodies in limbic encephalitis alter synaptic receptor location. Ann Neurol. 2009;65:424–34
- Lancaster E, Lai M, Peng X, et al. Antibodies to the GABA(B) receptor in limbic encephalitis with seizures: case series and characterisation of the antigen. Lancet Neurol. 2010;9:67–76
- Petit-Pedrol M, Armangue T, Peng X, et al. Encephalitis with refractory seizures, status epilepticus, and antibodies to the GABAA receptor: a case series, characterisation of the antigen, and analysis of the effects of antibodies. Lancet Neurol. 2014;13:276–86
- Lancaster E, Martinez-Hernandez E, Titulaer MJ, et al. Antibodies to metabotropic glutamate receptor 5 in the Ophelia syndrome. Neurology. 2011;77:1698–701.
- Dale RC, Merheb V, Pillai S, et al. Antibodies to surface dopamine-2 receptor in autoimmune movement and psychiatric disorders. Brain. 2012;135:3453–68.
- Lai M, Huijbers MG, Lancaster E, et al. Investigation of LGI1 as the antigen in limbic encephalitis previously attributed to potassium channels: a case series. Lancet Neurol. 2010;9:776–85.
- Irani SR, Alexander S, Waters P, et al. Antibodies to Kv1 potassium channel-complex proteins leucine-rich, glioma inactivated 1 protein and contactin-associated protein-2 in limbic encephalitis, Morvan’s syndrome and acquired neuromyotonia. Brain. 2010;133:2734–48.
- Boronat A, Gelfand JM, Gresa-Arribas N, et al. Encephalitis and antibodies to dipeptidyl-peptidase-like protein-6, a subunit of Kv4. 2 potassium channels. Ann Neurol. 2013;73:120–28.
- Brilot F, Dale RC, Selter RC, et al. Antibodies to native myelin oligodendrocyte glycoprotein in children with inflammatory demyelinating central nervous system disease. Ann Neurol. 2009;66:833–42.
- McKeon A, Lennon VA, Lotze T, et al. CNS aquaporin-4 autoimmunity in children. Neurology. 2008;71:93–100.
- Shahrizaila N, Yuki N. Bickerstaff brainstem encephalitis and Fisher syndrome: anti-GQ1b antibody syndrome. J Neurol Neurosurg Psychiatry. 2013;84:576–83.
- Ariño H, Höftberger R, Gresa-Arribas N, et al. Paraneoplastic neurological syndromes and glutamic acid decarboxylase antibodies. JAMA Neurol. 2015;72:874–81 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4838033/
- Voltz R. Neuropsychological symptoms in paraneoplastic disorders. J Neurol. (2007); http://www.ncbi.nlm.nih.gov/pubmed/17503138
- Tuzun E, Dalmau J. Limbic Encephalitis and Variants: Classification, Diagnosis, and Treatment. The Neurologist. 2007