What is lipoprotein
Lipoprotein in the blood whose primary purpose is to transport cholesterol, tryglycerides, and other fats throughout the body. Because lipids, such as cholesterol and triglycerides, are insoluble in water these lipids must be transported in association with proteins in the circulation. Large quantities of fatty acids from meals must be transported as triglycerides to avoid toxicity. These lipoproteins play a key role in the absorption and transport of dietary lipids by the small intestine, in the transport of lipids from the liver to peripheral tissues, and the transport of lipids from peripheral tissues to the liver and intestine (reverse cholesterol transport). A secondary function is to transport toxic foreign hydrophobic and amphipathic compounds, such as bacterial endotoxin, from areas of invasion and infection 1.
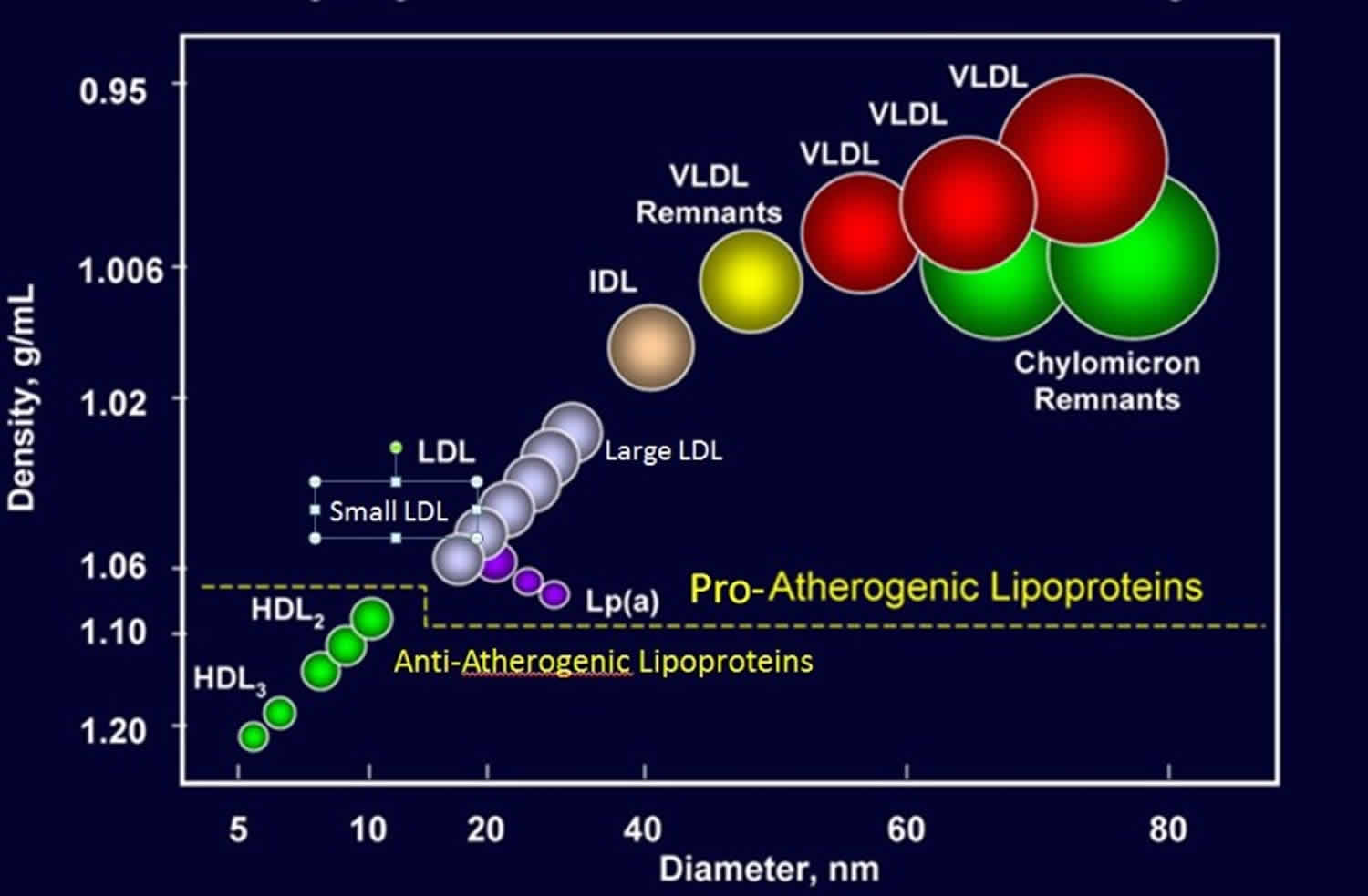
Plasma lipoproteins can be divided into seven classes based on size, lipid composition, and apolipoproteins (chylomicrons, chylomicron remnants, very-low-density lipoprotein (VLDL), intermediate-density lipoprotein (IDL), low-density lipoprotein (LDL), high-density lipoprotein (HDL), and lipoprotein (a) [Lp (a)]). Chylomicron remnants, VLDL (very-low-density lipoprotein), IDL (intermediate-density lipoprotein), LDL (low-density lipoprotein), and lipoprotein (a) are all pro-atherogenic (increases your risk of developing cardiovascular disease) while HDL (high-density lipoprotein) is anti-atherogenic (reduces your risk of developing cardiovascular disease).
Apolipoproteins have four major functions including 1) serving a structural role, 2) acting as ligands for lipoprotein receptors, 3) guiding the formation of lipoproteins, and 4) serving as activators or inhibitors of enzymes involved in the metabolism of lipoproteins.
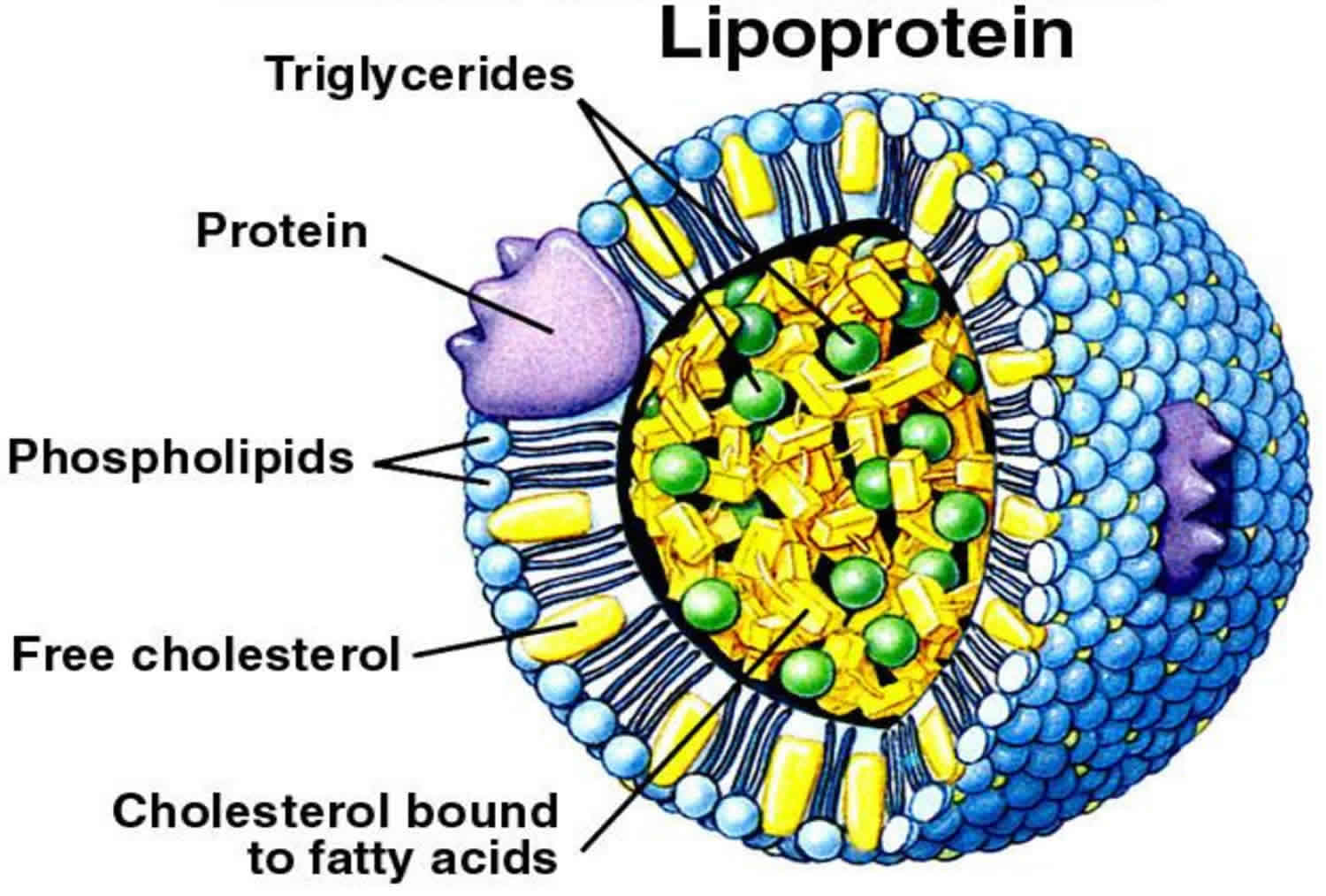
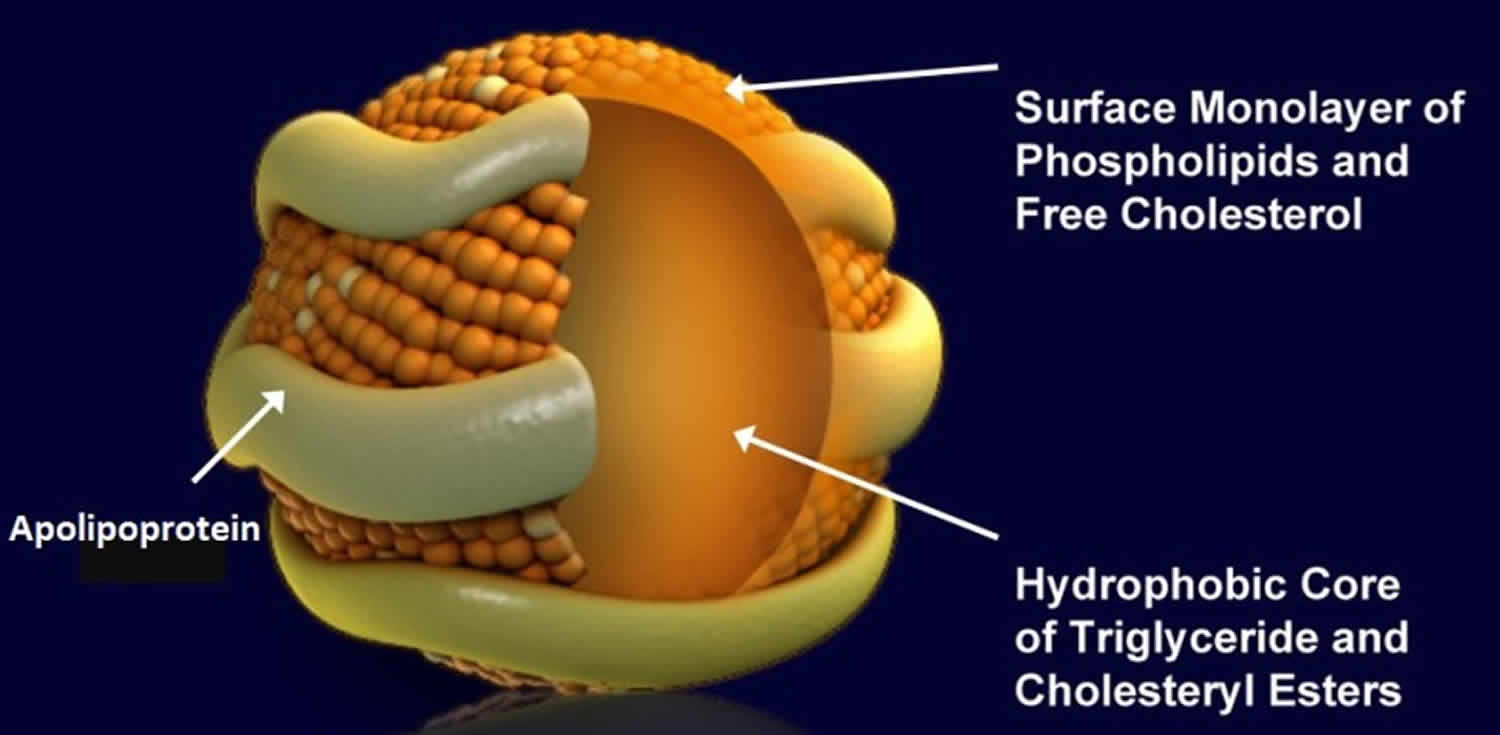
Lipoproteins are complex particles that have a central hydrophobic core of non-polar lipids, primarily cholesterol esters and triglycerides. This hydrophobic core is surrounded by a hydrophilic membrane consisting of phospholipids, free cholesterol, and apolipoproteins (Figure 1).
Figure 1. Lipoprotein structure
[Source 2 ]The origin of circulating lipoproteins is less understood than is their uptake, transport, and degradation 3. The lipoprotein lipid transport system in plasma has been described as involving two pathways: an exogenous route for the transport of cholesterol and triglycerides absorbed from dietary fat in the intestine, and an endogenous system through which cholesterol and triglycerides reach the plasma from the liver and other nonintestinal tissues.
Exogenous Lipoprotein Pathway
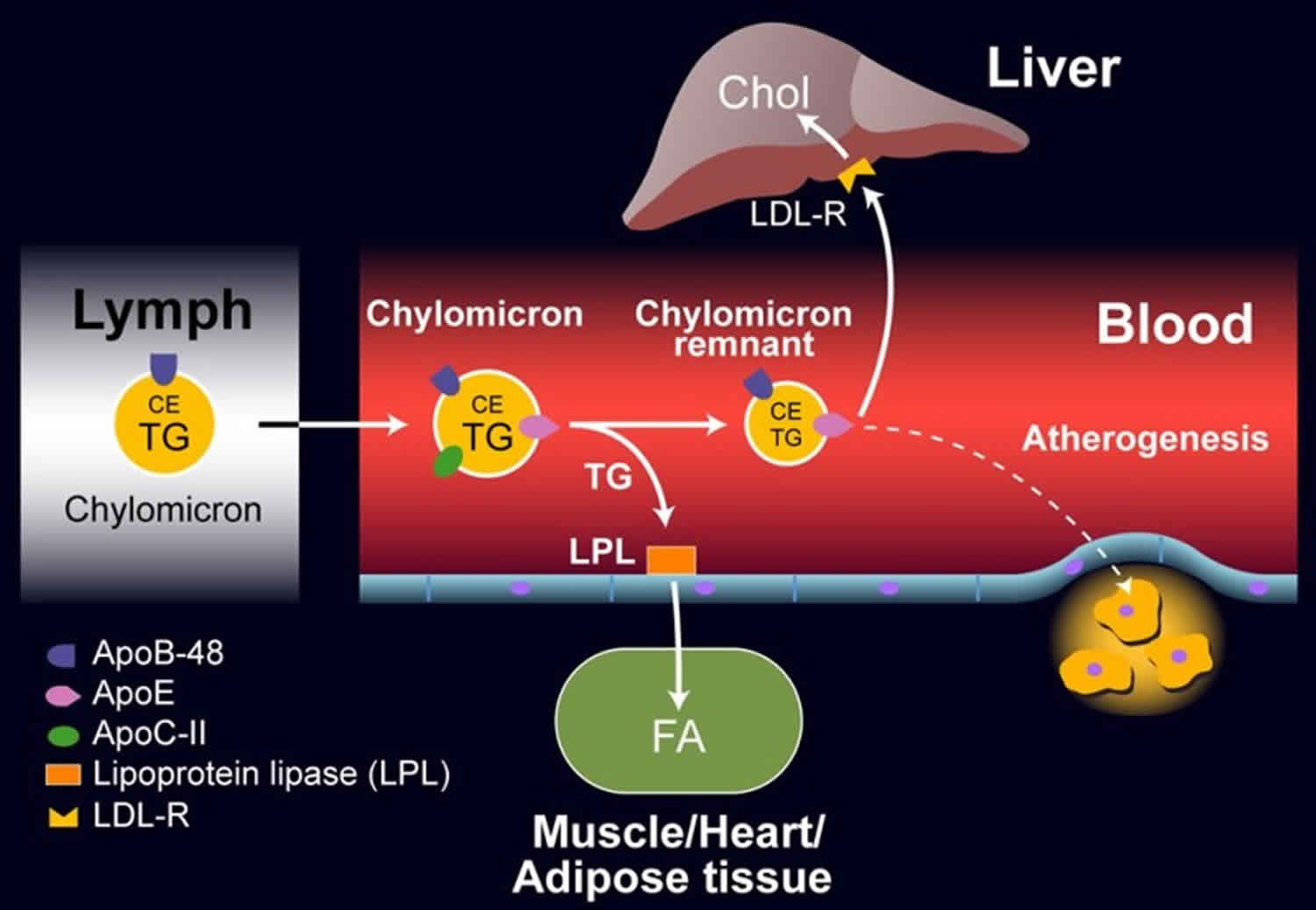
The exogenous pathway starts with the intestinal absorption of triglycerides and cholesterol from dietary sources. Its end result is the transfer of triglycerides to adipose and muscle tissue and of cholesterol to the liver. After absorption, triglycerides and cholesterol are re-esterified in the intestinal mucosal cells and then coupled with various apoproteins, phospholipids, and unesterified cholesterol into lipoprotein particles called chylomicrons. The chylomicrons in turn are secreted into intestinal lymph, enter the bloodstream through the thoracic: duct, and bind to the wall of capillaries in adipose and skeletal muscle tissue. At these binding sites the chylomicrons interact with the enzyme lipoprotein lipase, which causes hydrolysis of the triglyceride core and liberation of free fatty acids. These fatty acids then pass through the capillary endothelial cells and reach the adipocytes and skeletal muscle cells for storage or oxidation, respectively.
After removal of the triglyceride core, remnant chylomicron particles are formed. These are high in cholesterol esters and characterized by the presence of apoproteins B, CIII, and E. These remnants are cleared from the circulation by binding of their E apoprotein to a receptor present only on the surface of hepatic cells. Subsequently, the bound remnants are taken to the inside of hepatic cells by endocytosis and then catabolized by lysosomes. This process liberates cholesterol, which is then either converted into bile acids, excreted in bile, or incorporated into lipoproteins originated in the liver (VLDL).
Under normal physiologic conditions, chylomicrons are present in plasma for 1 to 5 hours after a meal and may give it a milky appearance. They are usually cleared from the circulation after a 12-hour fast.
Figure 2. Exogenous Lipoprotein Pathway
[Source 2 ]Endogenous Lipoprotein Pathway
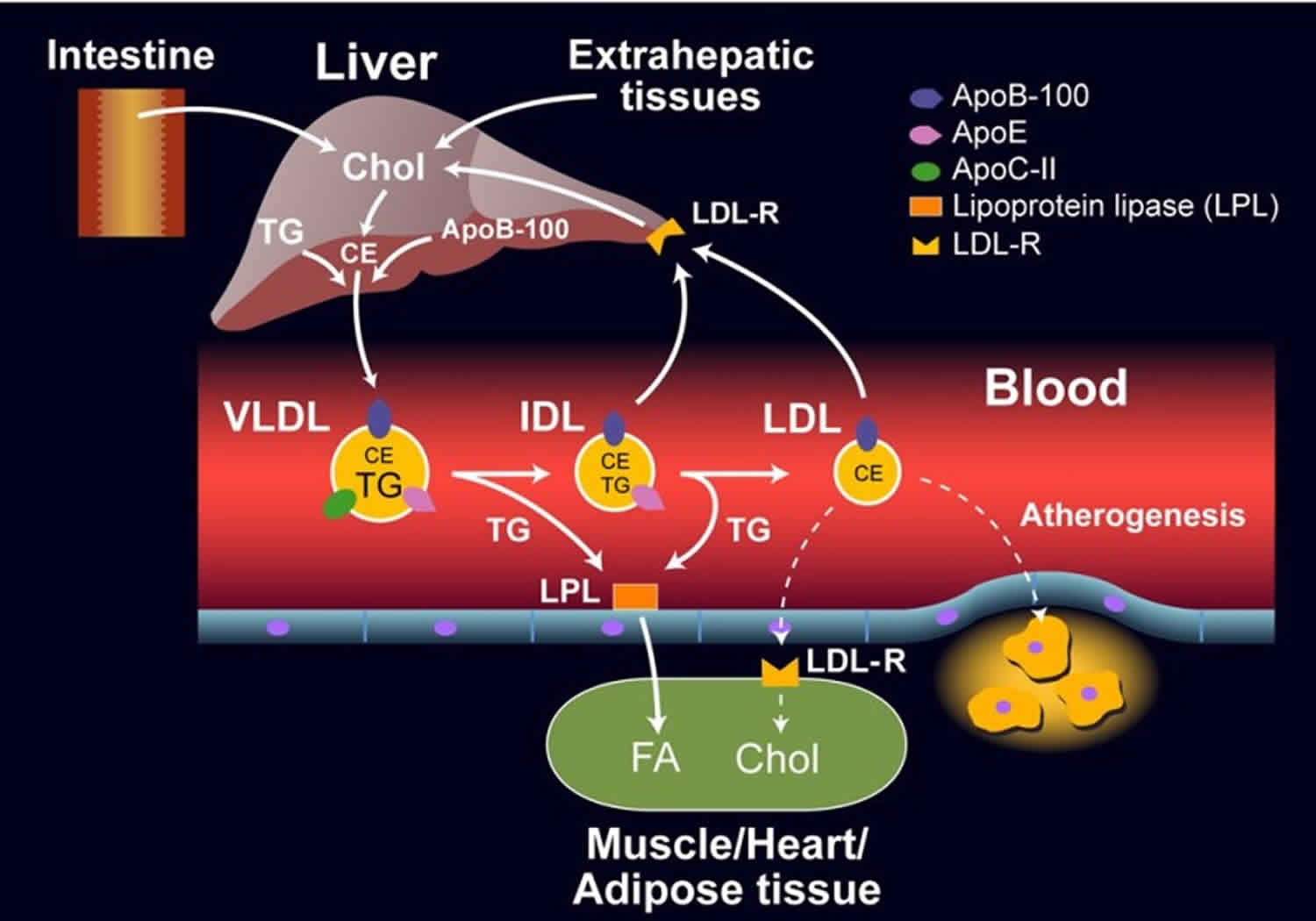
The liver constantly synthesizes triglycerides by utilizing as substrates free fatty acids and carbohydrates; these endogenous triglycerides are secreted into the circulation in the core of very-low-density lipoprotein particles (VLDL). The synthesis and secretion of very-low-density lipoprotein particles (VLDL) at cellular level occur in a process similar to that of chylomicrons, except that a different B apoprotein (B-100 instead of B-48) together with apoproteins C and E intervene in their secretion. Subsequent interaction of the VLDL particles with lipoprotein lipase in tissue capillaries leads to hydrolysis of the core triglycerides and production of smaller remnant VLDL particles rich in cholesterol esters (intermediate-density lipoproteins, IDL) and liberation of free fatty acids. Around half of these remnant particles are removed from the circulation in 2 to 6 hours as they bind tightly to hepatic cells. The rest undergo modifications with detachment of the remaining triglycerides and its substitution by cholesterol esters and removal of all the apoproteins except apoprotein B. This process results in transformation of the remnant VLDL particles into low-density lipoprotein particles (LDL) rich in cholesterol. In fact, these last particles contain around three-fourths of the total cholesterol in human plasma, although they constitute only some 7% of the total cholesterol pool. Their predominant function is to supply cholesterol to cells with LDL receptors, like those in the adrenal glands, skeletal muscle, lymphocytes, gonads, and kidneys. The quantity of cholesterol freed from LDL is said to control cholesterol metabolism in the cell through the following mechanisms: (1) increased LDL cholesterol in the cell decreases synthesis of the enzyme 3-hydroxy-3 methylglutaryl coenzyme A (HMG-CoA) reductase, which modulates the intracellular synthesis of cholesterol; (2) increased LDL cholesterol may enhance the storage of cholesterol within the cell by activation of another enzyme; and (3) increased cholesterol within the cell diminishes the synthesis of LDL receptors through a negative feedback process.
Besides the above described route for low-density lipoprotein (LDL) degradation in extrahepatic sites, a so-called scavenger cell pathway has been described. This consists of cells in the reticuloendothelial system which, by phagocytosis, dispose of the excess concentrations of this lipoprotein in plasma.
Figure 3. Endogenous Lipoprotein Pathway
Types of lipoprotein
Plasma lipoproteins are divided into seven classes based on size, lipid composition, and apolipoproteins (Table 1 and Figure 4). Plasma lipoprotein particles contain variable proportions of four major elements: cholesterol, triglycerides, phospholipids and specific proteins called apoproteins (Table 1). An alphabetical nomenclature (A, B, C, D, E.) is generally used to designate the apoproteins. The varying composition of these elements determines the density, size, and electrophoretic mobility of each particle. These factors in turn have been used for the clinical and biochemical classification of lipoprotein disorders. Schematically, lipoproteins have been described as globular or spherical units in which a nonpolar core lipid (consisting mainly of cholesterol esters and triglycerides) is surrounded by a layer containing phospholipids, apoproteins, and small amounts of unesterified cholesterol. Apoproteins, in addition to serving as carrier proteins, have other important functions such as being co-factors for enzymes involved in lipoprotein metabolism, acting as specific ligands for binding of the particles to cellular receptor sites, and intervening in the exchange of lipid constituents between lipoprotein particles.
- Chylomicrons
- Chylomicron Remnants
- Very-low-density lipoprotein (VLDL)
- Intermediate-density lipoprotein (IDL)
- Low-density lipoprotein (LDL)
- High-density lipoprotein (HDL)
- Lipoprotein (a)
Table 1. Types of Lipoprotein
| Lipoprotein | Density (g/ml) | Size (nm) | Major Lipids | Major Apoproteins |
|---|---|---|---|---|
| Chylomicrons | <0.930 | 75-1200 | Triglycerides | Apo B-48, Apo C, Apo E, Apo A-I, A-II, A-IV |
| Chylomicron Remnants | 0.930- 1.006 | 30-80 | Triglycerides Cholesterol | Apo B-48, Apo E |
| Very-low-density lipoprotein (VLDL) | 0.930- 1.006 | 30-80 | Triglycerides | Apo B-100, Apo E, Apo C |
| Intermediate-density lipoprotein (IDL) | 1.006- 1.019 | 25-35 | Triglycerides Cholesterol | Apo B-100, Apo E, Apo C |
| Low-density lipoprotein (LDL) | 1.019- 1.063 | 18- 25 | Cholesterol | Apo B-100 |
| High-density lipoprotein (HDL) | 1.063- 1.210 | 5- 12 | Cholesterol Phospholipids | Apo A-I, Apo A-II, Apo C, Apo E |
| Lipoprotein (a) | 1.055- 1.085 | ~30 | Cholesterol | Apo B-100, Apo (a) |
Figure 4. Types of Lipoprotein
[Source 2 ]Chylomicrons
Chylomicrons are large triglyceride rich particles made by the intestine, which are involved in the transport of dietary triglycerides and cholesterol to peripheral tissues and liver 2. Chylomicrons contain apolipoproteins A-I, A-II, A-IV, A-V, B-48, C-II, C-III, and E. Apo B-48 is the core structural protein and each chylomicron particle contains one Apo B-48 molecule. The size of chylomicrons varies depending on the amount of fat ingested. A high fat meal leads to the formation of large chylomicron particles due to the increased amount of triglyceride being transported whereas in the fasting state the chylomicron particles are small carrying decreased quantities of triglyceride.
Chylomicron remnants
The removal of triglyceride from chylomicrons by peripheral tissues results in smaller particles called chylomicron remnants 2. Compared to chylomicrons these particles are enriched in cholesterol and are pro-atherogenic.
Very low density lipoprotein
Very low density lipoproteins (VLDL) particles are produced by the liver and are triglyceride rich. Very low density lipoproteins (VLDL) contain apolipoprotein B-100, C-I, C-II, C-III, and E. Apo B-100 is the core structural protein and each very low density lipoprotein (VLDL) particle contains one Apo B-100 molecule. Similar to chylomicrons the size of the very low density lipoprotein particles can vary depending on the quantity of triglyceride carried in the particle. When triglyceride production in the liver is increased, the secreted very low density lipoprotein particles are large. However, very low density lipoprotein particles are smaller than chylomicrons.
Intermediate-density lipoprotein
The removal of triglycerides from very low density lipoprotein (VLDL) by muscle and adipose tissue results in the formation of intermediate-density lipoprotein (IDL) particles which are enriched in cholesterol. These particles contain apolipoprotein B-100 and E. These intermediate-density lipoprotein particles are pro-atherogenic.
Low density lipoprotein (LDL)
Low-density lipoprotein (LDL) particles are derived from very low density lipoprotein (VLDL) and intermediate-density lipoprotein (IDL) particles and they are even further enriched in cholesterol. Low-density lipoprotein (LDL) carries the majority of the cholesterol that is in the circulation. The predominant apolipoprotein is B-100 and each low-density lipoprotein (LDL) particle contains one Apo B-100 molecule. Low-density lipoprotein (LDL) consists of a spectrum of particles varying in size and density. An abundance of small dense low-density lipoprotein (LDL) particles are seen in association with hypertriglyceridemia, low high-density lipoprotein (HDL) levels, obesity, type 2 diabetes (i.e. patients with the metabolic syndrome) and infectious and inflammatory states. These small dense low-density lipoprotein (LDL) particles are considered to be more pro-atherogenic than large low-density lipoprotein (LDL) particles for a number of reasons. Small dense low-density lipoprotein (LDL) particles have a decreased affinity for the low-density lipoprotein (LDL) receptor resulting in a prolonged retention time in the circulation. Additionally, they more easily enter the arterial wall and bind more avidly to intra-arterial proteoglycans, which traps them in the arterial wall. Finally, small dense low-density lipoprotein (LDL) particles are more susceptible to oxidation, which could result in an enhanced uptake by macrophages.
High density lipoprotein (HDL)
High-density lipoproteins are a heterogeneous group of macromolecules with different physical properties and chemical components; two subclasses of HDL have been identified (HDL2 and HDL3) within which several subspecies have also been demonstrated 3. The predomination function of HDL seems to be the reverse transport of cholesterol from different tissues into the liver, where it is eventually removed, which is one potential mechanism by which high-density lipoprotein (HDL) may be anti-atherogenic. Subclass HDL2 has been reported to have a better correlation with coronary artery disease protection than total high-density lipoprotein (HDL) cholesterol. In addition, high-density lipoprotein (HDL) particles have anti-oxidant, anti-inflammatory, anti-thrombotic, and anti-apoptotic properties, which may also contribute to their ability to inhibit atherosclerosis 2. High-density lipoprotein (HDL) particles are enriched in cholesterol and phospholipids. Apolipoproteins A-I, A-II, A-IV, C-I, C-II, C-III, and E are associated with these particles. Apo A-I is the core structural protein and each high-density lipoprotein (HDL) particle may contain multiple Apo A-I molecules. High-density lipoprotein (HDL) particles are very heterogeneous and can be classified based on density, size, charge, or apolipoprotein composition (Table 2).
The serum concentration of high-density lipoprotein (HDL) and its components derives from various complex intravascular and cellular metabolic events. These events include secretion of precursor high-density lipoprotein (HDL) particles from the liver and small intestine, interaction of these particles with lipids and proteins released during the catabolism of triglyceride-rich lipoproteins, and production of cholesteryl esters (the core substance in HDL) from the action of lecithin–cholesterol acyltransferase (LCAT), an enzyme that originates in the liver. This enzyme acts on unesterified cholesterol released into plasma from cellular turnover. The cholesterol esters formed in this reaction are in turn transferred to very-low-density lipoprotein (VLDL) and subsequently appear in low-density lipoprotein (LDL). The end result is a system that allows the transfer of cholesterol through low-density lipoprotein (LDL) to peripheral cells and its return to the liver through high-density lipoprotein (HDL), and that prevents excessive accumulation of cholesterol in the body.
Table 2. Classification of high-density lipoprotein (HDL)
| Method of classification | Types of high-density lipoprotein (HDL) |
|---|---|
| Density gradient ultracentrifugation | HDL2, HDL3, Very high density HDL |
| Nuclear magnetic resonance | large, medium, and small |
| Gradient gel electrophoresis | HDL 2a, 2b, 3a, 3b, 3c |
| 2-dimensional gel electrophoresis | pre-beta 1 and 2, alpha 1, 2, 3, 4 |
| Apolipoprotein composition | A-I particles, A-I: A-II particles, A-I: E particles |
Lipoprotein-a
Lipoprotein-a is a low-density lipoprotein (LDL) particle that has apolipoprotein (a) attached to Apo B-100 via a disulfide bond. Lipoprotein (a) particle is pro-atherogenic. The physiologic function of this lipoprotein (a) is unknown 2.
Lipoprotein (a) contain Apo (a) and Apo B-100 in a 1:1 molar ratio. Like Apo B-100, apo (a) is also made by hepatocytes. Apo (a) contains multiple kringle motifs that are similar to the kringle repeats in plasminogen. The number of kringle repeats can vary and thus the molecular weight of apo (a) can range from 250,000 to 800,000. The levels of lipoprotein (a) in plasma can vary more than a 1000-fold ranging from undetectable to greater than 100mg/dl. Lipoprotein (a) levels largely reflect lipoprotein (a) production rates, which are primarily genetically regulated. Individuals with high molecular weight Apo (a) proteins tend to have lower levels of lipoprotein (a) while individuals with low molecular weight Apo (a) tend to have higher levels. It is hypothesized that the liver is less efficient in secreting high molecular weight Apo (a). The mechanism of lipoprotein (a) clearance is uncertain but does not appear to involve LDL receptors. Elevated plasma lipoprotein (a) levels are associated with an increased risk of atherosclerosis. Therapies that accelerate LDL clearance and lower LDL levels do not lower lipoprotein (a) levels (for example statin therapy). The kidney appears to play an important role in lipoprotein (a) clearance as kidney disease is associated with delayed clearance and elevations in lipoprotein (a) levels.
What are apolipoproteins
Apolipoproteins have four major functions including 1) serving a structural role, 2) acting as ligands for lipoprotein receptors, 3) guiding the formation of lipoproteins, and 4) serving as activators or inhibitors of enzymes involved in the metabolism of lipoproteins (Table 3). Apolipoproteins thus play a crucial role in lipoprotein metabolism.
Apolipoprotein A-I: Apo A-I is synthesized in the liver and intestine and is the major structural protein of HDL accounting for approximately 70% of HDL protein. It also plays a role in the interaction of HDL with ATP-binding cassette protein A1 (ABCA1), ABCG1, and class B, type I scavenger receptor (SR-B1). Apo A-I is an activator of lecithin: cholesterol acyltransferase (LCAT), an enzyme that converts free cholesterol into cholesteryl ester.
Apolipoprotein A-II: Apo A-II is synthesized in the liver and is the second most abundant protein on HDL accounting for approximately 20% of HDL protein.
Apolipoprotein A-IV: Apolipoprotein A-IV (Apo A-IV) is synthesized in the intestine during fat absorption 4. Apolipoprotein A-IV (Apo A-IV) is associated with chylomicrons and high-density lipoproteins, but is also found in the lipoprotein-free fraction. Its precise role in lipoprotein metabolism remains to be determined but studies have suggested a role for Apo A-IV in regulating food intake.
Apolipoprotein A-V: Apolipoprotein A-V (Apo A-V) is synthesized in the liver and associates with triglyceride rich lipoproteins 5. Apolipoprotein A-V (Apo A-V) is an activator of lipoprotein lipase (LPL) mediated lipolysis and thereby plays an important role in the metabolism of triglyceride rich lipoproteins.
Apolipoprotein B-48: Apo B-48 is synthesized in the intestine and is the major structural protein of chylomicrons and chylomicron remnants. There is a single molecule of apo B-48 per chylomicron particle. There is a single apolipoprotein B gene that is expressed in both the liver and intestine. The intestine expresses a protein that is approximately ½ the size of the liver due to mRNA editing. The apobec-1 editing complex is expressed in the intestine and edits a specific cytidine to an uracil in the apo B mRNA in the intestine creating a stop codon that results in the cessation of protein translation and a shorter Apo B (Apo B-48). Notably Apo B-48 is not recognized by the low-density lipoprotein (LDL) receptor.
Apolipoprotein B-100: Apo B-100 is synthesized in the liver and is the major structural component of very-low-density lipoprotein (VLDL), intermediate-density lipoprotein (IDL), and low-density lipoprotein (LDL). There is a single molecule of Apo B-100 per very-low-density lipoprotein (VLDL), intermediate-density lipoprotein (IDL), and low-density lipoprotein (LDL) particle. Apo B-100 is a ligand for the low-density lipoprotein (LDL) receptor and therefore plays an important role in the clearance of lipoprotein particles.
Apolipoprotein C: The C apolipoproteins are synthesized primarily in the liver and freely exchange between lipoprotein particles and therefore are found in association with chylomicrons, very-low-density lipoprotein (VLDL), and high-density lipoprotein (HDL).
Apo C-II is a co-factor for lipoprotein lipase (LPL) and thus stimulates triglyceride hydrolysis 6. Loss of function mutations in Apo C-II result in marked hypertriglyceridemia due to a failure to metabolize triglyceride rich lipoproteins.
Apo C-III is an inhibitor of LPL 7. Additionally, Apo C-III inhibits the interaction of triglyceride rich lipoproteins with their receptors. Recent studies have shown that loss of function mutations in Apo C-III lead to decreases in serum triglyceride levels and a reduced risk of cardiovascular disease. Interestingly, inhibition of Apo C-III expression results in a decrease in serum triglyceride levels even in patients deficient in lipoprotein lipase indicating that the ability of Apo C-III to modulate serum triglyceride levels is not dependent solely on regulating lipoprotein lipase activity.
Apolipoprotein E: Apolipoprotein E (Apo E) is synthesized in many tissues but the liver and intestine are the primary source of circulating Apo E 8. Apolipoprotein E (Apo E) exchanges between lipoprotein particles and is associated with chylomicrons, chylomicron remnants, VLDL, IDL, and a subgroup of HDL particles. There are three common genetic variants of apolipoprotein E (Apo E2, E3, and E4). Apolipoprotein E2 differs from the most common isoform, Apo E3, by a single amino acid substitution where cysteine substitutes for arginine at residue 158. Apo E4 differs from Apo E3 at residue 112, where arginine substitutes for cysteine. Apo E3 and E4 are ligands for the LDL receptor while Apo E2 is poorly recognized by the LDL receptor. Patients who are homozygous for Apo E2 can develop familial dysbetalipoproteinemia. Apo E4 is associated with an increased risk of Alzheimer’s disease and an increased risk of atherosclerosis.
Apolipoprotein (a): Apolipoprotein (a) [Apo (a)] is synthesized in the liver. This protein is a homolog of plasminogen and its molecular weight varies from 300,000 to 800,000 9. It is attached to Apo B-100 via a disulfide bond. High levels of apolipoprotein (a) [Apo (a)] are associated with an increased risk of atherosclerosis. Apolipoprotein (a) [Apo (a)] is an inhibitor of fibrinolysis and can also enhance the uptake of lipoproteins by macrophages, both of which could increase the risk of atherosclerosis. The physiologic function of Apo (a) is unknown. Interestingly this apolipoprotein is found in primates but not in other species.
Table 3. Apolipoproteins
| Apolipoprotein | Molecular weight (MW) | Primary Source | Lipoprotein Association | Function |
|---|---|---|---|---|
| Apo A-I | 28,000 | Liver, Intestine | HDL, chylomicrons | Structural protein for HDL, Activates LCAT |
| Apo A-II | 17,000 | Liver | HDL, chylomicrons | Structural protein for HDL, Activates hepatic lipase |
| Apo A-IV | 45,000 | Intestine | HDL, chylomicrons | Unknown |
| Apo A-V | 39,000 | Liver | VLDL, chylomicrons, HDL | Promotes LPL mediated TG lipolysis |
| Apo B-48 | 241,000 | Intestine | Chylomicrons | Structural protein for chylomicrons |
| Apo B-100 | 512,000 | Liver | VLDL, IDL, LDL, Lp (a) | Structural protein, Ligand for LDL receptor |
| Apo C-I | 6,600 | Liver | Chylomicrons, VLDL, HDL | Activates LCAT |
| Apo C-II | 8,800 | Liver | Chylomicrons, VLDL, HDL | Co-factor for LPL |
| Apo C-II | 8,800 | Liver | Chylomicrons, VLDL, HDL | Inhibits LPL and uptake of lipoproteins |
| Apo E | 34,000 | Liver | Chylomicron remnants, IDL, HDL | Ligand for LDL receptor |
| Apo (a) | 250,000- 800,00 | Liver | Lp (a) | Inhibits plasminogen activation |
What is lipoprotein lipase
Lipoprotein lipase (LPL) is synthesized in muscle, heart, and adipose tissue, then secreted and attached to the endothelium of the adjacent blood capillaries 10. Lipoprotein lipase hydrolyzes the triglycerides carried in chylomicrons and very-low-density lipoprotein (VLDL) to fatty acids, which can be taken up by cells. The catabolism of triglycerides results in the conversion of chylomicrons into chylomicron remnants and very-low-density lipoprotein (VLDL) into intermediate-density lipoprotein (IDL). This enzyme requires Apo C-II as a cofactor. Apo A-V also plays a key role in the activation of this enzyme. In contrast Apo C-III and Apo A-II inhibit the activity of lipoprotein lipase. Insulin stimulates lipoprotein lipase expression and lipoprotein lipase activity is reduced in patients with poorly controlled diabetes, which can impair the metabolism of triglyceride rich lipoproteins leading to hypertriglyceridemia.
Congenital lipoprotein lipase deficiency is a rare autosomal recessive disorder secondary to absence or severe diminution in the activity of lipoprotein lipase. Affected individuals are homozygous for a mutation that prevents normal expression of lipoprotein lipase activity. The parents, although clinically normal, are obligate heterozygotes. This enzymatic disorder is reflected in a massive accumulation of chylomicrons in the plasma without elevation of very-low-density lipoprotein (VLDL) (type 1 lipoprotein pattern). Triglycerides may reach levels of 2000 to 10,000 mg/dl. This disorder usually appears in childhood with recurrent bouts of abdominal pain secondary to pancreatitis. Another characteristic feature of the disorder is the occurrence of eruptive xanthomas, predominantly in the buttocks and other pressure-sensitive areas of the body, attributed to the deposition of chylomicron triglycerides in histiocytes. The elevation in chylomicrons may also cause a lipemic serum, a picture of lipemia retinalis, and hepatosplenomegaly. The diagnosis is suspected by the presence of a lipemic plasma in a young patient who has fasted for 12 hours. Overnight incubation of a test tube of such plasma in a refrigeration at 4°C demonstrates a visible white layer at the top of the tube. Failure of lipoprotein lipase levels to increase after an infusion of heparin confirms the diagnosis. The occurrence of accelerated atherosclerosis is not reported in this condition.
Clinical significance of lipoprotein
Cholesterol and triglycerides, like many other essential components of the body, attract clinical attention when present in abnormal concentrations. Increased or decreased levels usually occur because of abnormalities in the synthesis, degradation, and transport of their associated lipoprotein particles. When hyperlipidemia or hypolipidemia are defined in terms of the class or classes of increased or decreased plasma lipoproteins, the names hyperlipoproteinemia or hypolipoproteinemia are preferentially employed.
High lipoprotein
Hyperlipoproteinemia is the lipid disturbance of major relevance clinically because of its association with an increased risk of atherosclerotic cardiovascular disease. Multiple epidemiologic studies have demonstrated that increased levels of plasma total cholesterol and low-density lipoproteins are strongly and directly related to a greater incidence of coronary heart disease. Elevated plasma triglycerides and very-low-density lipoproteins are directly associated with the risk of atherosclerotic heart disease, although not as independent risk factors. In contrast, high levels of high-density lipoprotein cholesterol have been found to be a protective factor for the development of that disease, so that decreased levels constitute a risk factor.
Clinical manifestations of hyperlipoproteinemia include a greater incidence of ischemic vascular disease, acute pancreatitis, and visible accumulations of lipid deposits (xanthomas and xanthelasmas). The localization of these lesions is of great help in many instances to categorize the lipoprotein dysfunction present.
Increased concentration of plasma lipids is etiologically related mainly to genetic disorders, dietary factors (such as ingestion of excessive calories, saturated fatty acids and cholesterol), or ingestion of drugs, or it may occur as a secondary phenomenon in a large variety of diseases. In any of these instances the elevation of the different plasma lipoproteins usually occurs in a number of combinations that have led to their classification into six different patterns or phenotypes (Table 4). Genetic or acquired disorders may be related to one or more of these lipoprotein patterns, so the identification of a particular pattern gives no specific information regarding the cause of the hyperlipidemic disorder in question. A practical approach is to classify the different hyperlipidemic: states into those that mainly cause hypercholesterolemia (Table 5) or those that predominantly cause hypertriglyceridemia (Table 6). In each of these categories primary (genetic) as well as secondary disorders are included. The genetic disorders in turn could result from a single gene disturbance either of dominant or recessive inheritance, or a polygenic derangement in which multiple genes interact with environmental factors. The main characteristics of primary hyperlipoproteinemias are included in Table 7.
Table 4. Lipoprotein Patterns Resulting from Elevation of Different Plasma Lipid Fractions
| Lipoprotein pattern | Increased lipid fraction | Predominant lipoprotein |
|---|---|---|
| Type I | Triglycerides | Chylormicrons |
| Type 2a | Cholesterol | LDL |
| Type 2b | Cholesterol and triglycerides | LDL and VLDL |
| Type 3 | Triglycerides and cholesterol | Remnants |
| Type 4 | Triglycerides | VLDL |
| Type 5 | Triglycerides and cholesterol | VLDL and chylomicrons |
Table 5. Disorders Mainly Causing Hypercholesterolemia
| Primary disorders |
| Polygenic hypercholesterolemia |
| Familial hypercholesterolemia |
| Familial combined hyperlipidemia |
| Secondary disorders |
| Hyperthyroidism |
| Nephrotic syndrome |
| Dysproteinemias |
| Obstructive liver disease |
| Thiazide diuretics |
Table 6. Disorders Mainly Causing Hypertriglyceridemia
| Primary disorders |
| Familial hypertriglyceridemia |
| Familial combined hyperlipidemia |
| Congenital deficiency of lipoprotein lipase |
| Deficiency of apoprotein CII |
| Familial dysbetalipoproteinemia |
| Secondary disorders |
| Uncontrolled diabetes mellitus |
| Nephrotic syndrome |
| Dysproteinemias |
| Oral contraceptives |
| Thiazide diuretics |
| Beta-adrenergic blocking agents |
| Alcohol |
Table 7. Characteristics of Primary Hyperlipoproteinemias
| Condition | Hereditary pattern | Frequency | Biochemical defect | Plasma lipids | Elevated lipoprotein | Lipoprotein pattern | Major clinical features |
|---|---|---|---|---|---|---|---|
| Polygenic hypercholesterolemia | Multiple genes | Very common | Reduced activity hepatic LDL receptor | Increased cholesterol; TG normal | LDL | 2a | Adult onset; accelerated atherosclerosis |
| Familial hypercholesterolemia | Autosomal dominant | Common 1:500 | Deficiency of LDL receptor | Increased cholesterol; TG normal | LDL | 2a (rarely 2b) | Onset at all ages; planar, tendinous, tuberous xanthomas; accelerated atherosclerosis |
| Familial combined hyperlipidemia | Autosomal dominant | Common | Unknown | Increased cholesterol; TG normal | LDL and VLDL | Onset at all ages; accelerated atherosclerosis | |
| Familial hypertriglyceridemia | Autosomal dominant | Common | Unknown | Cholesterol normal; TG greatly increased | VLDL (rarely chylomicrons) | 4 (rarely 5) | Adult onset; eruptive xanthomas may occur; accelerated atherosclerosis; hyperglycemia; hyperuricemia; hepatosplenomegaly |
| Congenital lipoprotein lipase deficiency | Autosomal recessive | Rare | Deficiency of lipoprotein lipase | Normal cholesterol ; TG greatly increased | Chylomicrons | 1 | Juvenile onset; eruptive xanthomas; pancreatitis; lipemic plasma; lipemia retinalis; hepatosplenomegaly |
| Deficiency of apoprotein CII | Autosomal recessive | Rare | Deficiency of apoprotein CII | Normal cholesterol; TG greatly increased | Chylomicrons and VLDL | 1 or 5 | Juvenile onset; pancreatitis; lipemic plasma |
| Familial dysbetalipoproteinemia | Single mutant gene | Rare | Abnormal apoprotein E of VLDL | Increased cholesterol; TG increased | Remnants | 3 | Adult onset; palmar and/or tubereruptive xanthomas; accelerated atherosclerosis |
High alpha lipoprotein (hyperalphalipoproteinemia)
Another clinical condition associated with elevation in plasma lipoproteins is hyperalphalipoproteinemia, characterized by elevated plasma levels of high-density lipoproteins. The elevation in high-density lipoprotein (HDL) leads to slight increase in total plasma cholesterol values. Other plasma lipid components (LDL, VLDL, and triglycerides) are normal.
The majority of cases of hyperalphalipoproteinemia are genetic with either a dominant or polygenic inheritance. Secondary elevations of high-density lipoprotein (HDL) have been related to various factors such as weight reduction, regular exercise, moderate alcohol intake, estrogen administration, exposure to chlorinated hydrocarbon pesticides, and biliary cirrhosis. Patients with this condition usually do not present any distinguishing clinical features. As previously mentioned, hyperalphalipoproteinemia is associated with a decreased risk of coronary atherosclerosis and with increased longevity.
Low lipoprotein
Hypoalphalipoproteinemia probably is the most clinically significant hypolipoproteinemia in view of the fact that considerable evidence suggests that low levels of plasma HDL cholesterol are related to an increased incidence of coronary heart disease in high-risk populations.
According to the NIH Consensus Conference, the finding of high-density lipoprotein (HDL) cholesterol values below 35 mg/dl constitutes an independent risk factor for coronary artery disease. Several factors have been identified as causing a decrease in high-density lipoprotein (HDL) cholesterol. These include ill-defined genetic factors, obesity, cigarette smoking, physical inactivity, hypertriglyceridemia, oral contraceptives, beta-adrenergic blocking drugs, thiazide diuretics, and cholesterol-reducing diets.
Other hypolipoproteinemias comprise two rare disorders characterized by a decrease in the concentration of lipids in plasma and an autosomal recessive inheritance.
Abetalipoproteinernia usually appears early in childhood, and because of a defective production of apoprotein B, there is absence of chylomicrons, VLDL, and LDL in the plasma. The plasma cholesterol level is usually less than 75 mg/dl and that of triglycerides less than 15 mg/dl. The main clinical features are malabsorption of fats, peripheral neuropathy, ataxia, retinitis pigmentosa, and acanthocytosis.
Tangier disease is a condition that also manifests in childhood and is characterized by the absence of HDL from the plasma. This defect leads to the production of abnormal chylomicron remnants, which are stored as cholesterol esters in cells of the phagocytic system. Levels of plasma cholesterol are usually less than 100 mg/dl and that of triglycerides range from 100 to 250 mg/dl. The main clinical features are enlarged orange tonsils, corneal opacities, and infiltration of the bone marrow and the intestinal mucosa. Patients with this illness are at increased risk for premature atherosclerosis.
Dyslipoproteinemia, is the term utilized for conditions in which structurally abnormal lipoproteins circulate in plasma. Such a defect is seen in lecithin cholesterol acyltransferase (LCAT) deficiency. This is a rare disorder in which decreased activity of this enzyme leads to a large accumulation of unesterified cholesterol in plasma and body tissues. Laboratory findings include a variable level of total plasma cholesterol with decreased esterified cholesterol, an increase in unesterified cholesterol and increased VLDL. The structure of all the lipoproteins is abnormal. The condition usually presents in young adulthood with corneal opacities, renal insufficiency, hemolytic anemia, and premature atherosclerosis.
Lipoprotein levels
For the diagnosis of the different lipoprotein disorders, you can usually rely on simple plasma values of total cholesterol and triglycerides. These measurements should be repeated at least once, so as to obtain a more reliable estimate of the patient’s level. Obtained lipid values should be interpreted in relation to age and sex according to reference values established by the Lipid Research Clinics Program (Tables 8, 9, and 10). As stated previously, the diagnosis of hyperlipidemia is based on arbitrary reference values above the 95th percentile for the distribution of cholesterol, triglyceride or low-density lipoprotein (LDL) cholesterol for a given population. Low reference values (5th percentile for the distribution of cholesterol, LDL and HDL) identify persons with low lipid levels.
Plasma cholesterol and triglyceride values provide information concerning the lipoprotein particles that are increased. Plasma cholesterol values represent total cholesterol, which includes both unesterified cholesterol and cholesteryl esters. Isolated elevation of plasma cholesterol usually indicates that low-density lipoprotein (LDL) is increased. Isolated elevation of plasma triglyceride points to an elevation in chylomicrons, very-low-density lipoprotein (VLDL), and/or remnants.
The bedside inspection of a refrigerated fasting plasma sample, the measurement of high-density lipoprotein (HDL) cholesterol (advisable if total plasma cholesterol is persistently elevated) and the evaluation of associated clinical findings provide useful information. In all cases, a detailed history and physical examination is imperative. Evaluation and exclusion of secondary disorders, if necessary, could require additional laboratory tests as indicated in each particular case. Screening of first-degree relatives is advisable, especially if no secondary disorder is evident, as it may reveal a genetic problem. Only in selected instances will special tests or lipoprotein electrophoresis be required for exact genotypic diagnosis.
Table 8. Percentiles for Plasma Total Cholesterol (mg/dl) in the Normal White North American Population
| Men | Women | ||||||
|---|---|---|---|---|---|---|---|
| Age (yr) | 5 percentile | 75 percentile | 95 percentile | Age (yr) | 5 percentile | 75 percentile | 95 percentile |
| 0–19 | 115 | 175 | 200 | 0–19 | 120 | 175 | 200 |
| 20–24 | 125 | 185 | 220 | 20–24 | 125 | 190 | 230 |
| 25–29 | 135 | 200 | 245 | 25–34 | 130 | 195 | 235 |
| 30–34 | 140 | 215 | 255 | 35–39 | 140 | 205 | 245 |
| 35–39 | 145 | 225 | 270 | 40–44 | 145 | 215 | 255 |
| 40–44 | 150 | 230 | 270 | 45–49 | 150 | 225 | 270 |
| 45–69 | 160 | 235 | 275 | 50–54 | 165 | 240 | 285 |
| 70 + | 150 | 230 | 270 | 55 + | 170 | 250 | 295 |
Table 9. Percentiles for Plasma Triglycerides (mg/dl) in the Normal White North American Population.
| Men | Women | ||||
|---|---|---|---|---|---|
| Age (yr) | 5 percentile | 95 percentile | Age (yr) | 5 percentile | 95 percentile |
| 0–9 | 30 | 100 | 0–9 | 35 | 110 |
| 10–14 | 30 | 125 | 10–19 | 40 | 130 |
| 15–19 | 35 | 150 | 20–34 | 40 | 170 |
| 20–24 | 45 | 200 | 35–39 | 40 | 195 |
| 25–29 | 45 | 250 | 40–44 | 45 | 210 |
| 30–34 | 50 | 265 | 45–49 | 45 | 230 |
| 35–39 | 55 | 320 | 50–54 | 55 | 240 |
| 40–54 | 55 | 320 | 55–64 | 55 | 250 |
| 55–64 | 60 | 290 | 65 + | 60 | 240 |
| 65 + | 55 | 260 | |||
Table 10. Percentiles for Plasma Total Low-Density Lipoprotein Cholesterol (mg/dl) in the Normal White North American Population
| Men | Women | ||||||
|---|---|---|---|---|---|---|---|
| Age (yr) | 5 percentile | 75 percentile | 95 percentile | Age (yr) | 5 percentile | 75 percentile | 95 percentile |
| 0–19 | 65 | 105 | 130 | 0–19 | 65 | 110 | 140 |
| 20–24 | 65 | 120 | 145 | 20–24 | 55 | 120 | 160 |
| 25–29 | 70 | 140 | 165 | 25–34 | 70 | 125 | 160 |
| 30–34 | 80 | 145 | 185 | 35–39 | 75 | 140 | 170 |
| 35–39 | 80 | 155 | 190 | 40–44 | 75 | 145 | 175 |
| 40–44 | 85 | 155 | 185 | 45–49 | 80 | 150 | 185 |
| 45–69 | 90 | 165 | 205 | 50–54 | 90 | 160 | 200 |
| 70 + | 90 | 165 | 185 | 55 + | 95 | 170 | 215 |
Lipoprotein-a test
Lipoprotein-a test is used to identify an elevated level of lipoprotein (a) help evaluate a person’s risk of developing cardiovascular disease (CVD). Lipoprotein (a) test may be used in conjunction with a routine lipid profile to provide additional information about a person’s risk for cardiovascular disease.
The lipoprotein-a level is genetically determined and remains relatively constant over an individual’s lifetime. Since it is usually not affected by lifestyle changes or by most drugs, it is not the target of therapy. Instead, when lipoprotein (a) is high, the presence of this added risk factor may suggest the need for more aggressive treatment of other, more treatable risk factors such as an elevated low-density lipoprotein (LDL).
Lipoprotein (a) is a low-density lipoprotein (LDL) particle that has apolipoprotein (a) attached to Apo B-100 via a disulfide bond. Lipoprotein (a) particle is pro-atherogenic. The physiologic function of this lipoprotein (a) is unknown 2. Like low-density lipoprotein (LDL), lipoprotein (a) is considered a risk factor for cardiovascular disease. The amount of lipoprotein (a) that a person has is genetically determined and remains relatively constant over an individual’s lifetime. A high level of lipoprotein (a) is thought to contribute to a person’s overall risk of cardiovascular disease, making this test potentially useful as a cardiovascular risk marker.
Since about 50% of the people who have heart attacks have a normal cholesterol level, researchers have sought other factors that may have an influence on heart disease. It is thought that lipoprotein (a) may be one such factor. Lipoprotein (a) has two potential ways to contribute. First, since lipoprotein (a) can promote the uptake of low-density lipoprotein (LDL) into blood vessel walls, it may promote the development of atherosclerotic plaque on the walls of blood vessels. Secondly, since apo(a) has a structure that can inhibit enzymes that dissolve clots, lipoprotein (a) may promote accumulation of clots in the arteries. For these reasons, lipoprotein (a) may be more atherogenic than low-density lipoprotein (LDL).
The protein portion of lipoprotein (a) consists of:
- Apolipoprotein B (Apo B) – a protein that is involved in the metabolism of lipids and is the main protein constituent of lipoproteins such as LDL and very low-density lipoprotein (VLDL)
- Apolipoprotein(a) – A second protein component, called apolipoprotein(a), which is attached to the Apo B. Apolipoprotein(a) has an unusual structure and is thought to inhibit clots from being broken down normally. The size of the apolipoprotein(a) portion of lipoprotein (a) varies in size from person to person and tends to be smaller in Caucasians than in those of African ancestry. The significance of the variation in size in contributing to cardiovascular disease risk is complex, but there is some evidence that smaller size increases risk. Most lipoprotein (a) tests do not measure the size of apolipoprotein(a), however. They measure and report only the level of lipoprotein (a) in blood.
When is lipoprotein-a test ordered?
Lipoprotein-a test is not routinely ordered as part of a lipid profile. However, lipoprotein-a test may be ordered, along with other lipid tests, when an individual has a strong family history of cardiovascular disease at a young age that is not explained by high low-density lipoprotein (LDL) or low high-density lipoprotein (HDL).
Some health practitioners may also order these tests when:
- A person has existing heart or vascular disease, especially those individuals who have healthy lipid levels or ones that are only mildly elevated
- Someone may have an inherited predisposition for high cholesterol level
- A person has had a stroke or heart attack but has normal or only mildly elevated lipids
In rare cases, an lipoprotein (a) level may be ordered when a woman is postmenopausal to see if elevations in lipoprotein (a), tied to decreasing estrogen levels, have significantly increased her risk of developing cardiovascular disease.
A National Cholesterol Education Program (NCEP) guideline, the Adult Treatment Panel III, acknowledged the possible usefulness of lipoprotein (a), but it did not recommend widespread screening. The National Academy of Clinical Biochemistry (NACB) guidelines for emerging biomarkers of cardiovascular disease and stroke also recommend testing for individuals with a strong family history of premature atherosclerotic heart disease and/or high lipid levels (hyperlipidemia), or those with intermediate cardiovascular risk, but do not recommend general screening.
This is partially due to the fact that lipoprotein (a) levels are genetically determined and difficult to change. Niacin and estrogen (for postmenopausal women) have been shown to lower lipoprotein (a) levels a small amount, but their effect appears to be short-term and it is not known if lowering lipoprotein (a) actually lowers risk. Experts are currently not recommending drug treatments for elevated lipoprotein (a), but some are suggesting that those with elevated lipoprotein (a) should be especially vigilant about lowering their low-density lipoprotein (LDL – the “bad” cholesterol), which may help lower their overall risk.
In general, lipids should not be measured during a fever or major infection, within four weeks of an acute myocardial infarction (heart attack), a stroke, or major surgery, right after excessive alcohol intake, with severely uncontrolled diabetes, when a woman is pregnant, or during rapid weight loss.
Lipoprotein-a levels
Normal lipoprotein (a) less than or equal to 30 mg/dL
Lipoprotein (a) values >30 mg/dL may suggest increased risk of coronary heart disease. High lipoprotein (a) can occur in people with a normal lipid profile. An elevated level of lipoprotein (a) is thought to contribute to risk of heart disease independently of other lipids.
Lipoprotein-a concentrations >30 mg/dL are associated with 2- to 3-fold increased risk of cardiovascular events independent of conventional risk markers.
The level of lipoprotein (a) is genetically determined and is not easily modified by lifestyle changes or drugs. However, some non-genetic conditions may also lead to elevated lipoprotein (a). These include estrogen depletion, familial hypercholesterolemia, severe hypothyroidism, uncontrolled diabetes, chronic renal failure, and nephrotic syndrome.
There are no known problems associated with low lipoprotein (a). Many individuals have no detectable lipoprotein (a) in their blood.
Why would my doctor want to order an lipoprotein (a) more than once?
Typically, the lipoprotein (a) level is only tested once because it is usually fairly constant. Occasionally, your doctor may order a second lipoprotein (a) to confirm the initial level, especially if it was measured when you were ill, to see if your risk has increased significantly after menopause, or (rarely) to monitor the effects of treatment.
Can I have an lipoprotein (a) test done in my doctor’s office?
The lipoprotein (a) test requires specialized equipment to perform and is usually performed in a laboratory. Your sample may be sent to a reference laboratory for testing.
If lipoprotein (a) levels don’t really change over my lifetime, why would my doctor prescribe treatment?
Although lipoprotein (a) levels do not typically respond to treatment, if you have other additional cardiovascular risk factors, your doctor may want to treat these factors to help lower your overall risk.
References- Feingold KR, Grunfeld C. Lipids: a key player in the battle between the host and microorganisms. Journal of lipid research. 2012 Dec;53(12):2487-9
- Feingold KR, Grunfeld C. Introduction to Lipids and Lipoproteins. [Updated 2018 Feb 2]. In: De Groot LJ, Chrousos G, Dungan K, et al., editors. Endotext [Internet]. South Dartmouth (MA): MDText.com, Inc.; 2000-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK305896
- Cox RA, García-Palmieri MR. Cholesterol, Triglycerides, and Associated Lipoproteins. In: Walker HK, Hall WD, Hurst JW, editors. Clinical Methods: The History, Physical, and Laboratory Examinations. 3rd edition. Boston: Butterworths; 1990. Chapter 31. Available from: https://www.ncbi.nlm.nih.gov/books/NBK351
- Wang F, Kohan AB, Lo CM, Liu M, Howles P, Tso P. Apolipoprotein A-IV: a protein intimately involved in metabolism. Journal of lipid research. 2015 Aug;56(8):1403-18.
- Hubacek JA. Apolipoprotein A5 fifteen years anniversary: Lessons from genetic epidemiology. Gene. 2016 Oct 30;592(1):193-9.
- Wolska A, Dunbar RL, Freeman LA, Ueda M, Amar MJ, Sviridov DO, et al. Apolipoprotein C-II: New findings related to genetics, biochemistry, and role in triglyceride metabolism. Atherosclerosis. 2017 Dec;267:49-60.
- Taskinen MR, Boren J. Why Is Apolipoprotein CIII Emerging as a Novel Therapeutic Target to Reduce the Burden of Cardiovascular Disease? Current atherosclerosis reports. 2016 Oct;18(10):59
- Mahley RW. Apolipoprotein E: from cardiovascular disease to neurodegenerative disorders. Journal of molecular medicine. 2016 Jul;94(7):739-46.
- Nordestgaard BG, Langsted A. Lipoprotein (a) as a cause of cardiovascular disease: insights from epidemiology, genetics, and biology. Journal of lipid research. 2016 Nov;57(11):1953-75.
- Olivecrona G. Role of lipoprotein lipase in lipid metabolism. Current opinion in lipidology. 2016 Jun;27(3):233-41