What is mucuna pruriens
Mucuna pruriens (Fabaceae) is a tropical legume native to Africa and tropical Asia and widely naturalized and cultivated. Mucuna pruriens other common names include velvet bean, Bengal velvet bean, Florida velvet bean, Mauritius velvet bean, Yokohama velvet bean, cowhage, cowitch, lacuna bean, and Lyon bean 1. Since Mucuna pruriens is a legume, it fixes nitrogen and fertilizes soil. In Indonesia, particularly Java, the Mucuna pruriens beans are eaten and widely known as ‘Benguk’. The beans can also be fermented to form a food similar to tempe and known as Benguk tempe or ‘tempe Benguk’.
Mucuna pruriens is also a widespread fodder plant in the tropics. To that end, the whole Mucuna pruriens plant is fed to animals as silage, dried hay or dried seeds. Mucuna pruriens silage contains 11-23% crude protein, 35-40% crude fiber, and the dried beans 20-35% crude protein. Mucuna pruriens also has use in the countries of Benin and Vietnam as a biological control for problematic Imperata cylindrica grass 2. Mucuna pruriens is said to not be invasive outside its cultivated area 2. However, the plant is invasive within conservation areas of South Florida, where it frequently invades disturbed land and rockland hammock edge habitats. Mucuna pruriens is sometimes used as a coffee substitute. Cooked fresh shoots or beans can also be eaten.
The hairs lining the Mucuna pruriens seed pods contain serotonin and the protein mucunain which cause severe itching when the pods are touched 3. The calyx below the flowers is also a source of itchy spicules and the stinging hairs on the outside of the seed pods are used in itching powder 3. Scratching the exposed area can spread the itching to other areas touched. Once this happens, the subject tends to scratch vigorously and uncontrollably and for this reason the local populace in northern Mozambique refer to the beans as “mad beans” (feijões malucos).
Figure 1. Mucuna pruriens
Figure 2. Mucuna pruriens seeds
In addition to the low levels of sulfur-containing amino acids in Mucuna pruriens seeds, the presence of anti-physiological and toxic factors may contribute to a decrease in their overall nutritional quality 4. These factors include polyphenols, trypsin inhibitors, phytate, cyanogenic glycosides, oligosaccharides, saponins, lectins, and alkaloids 4. Polyphenols (or tannins) are able to bind to proteins, thus lowering their digestibility. Phenolic compounds inhibit the activity of digestive as well as hydrolytic enzymes such as amylase, trypsin, chymotrypsin, and lipase. Recently, phenolics have been suggested to exhibit health related functional properties such as anti-carcinogenic, anti-viral, anti-microbial, anti-inflammatory, hypotensive, and anti-oxidant activities.
Trypsin inhibitors belong to the group of proteinase inhibitors that include polypeptides or proteins that inhibit trypsin activity. Tannins exhibit weak interactions with trypsin, and thus also inhibit trypsin activity. Phytic acid [myoinositol-1,2,3,4,5,6-hexa(dihydrogen phosphate)] is a major component of all plant seeds, which can reduce the bioavailability of certain minerals such as zinc, calcium, magnesium, iron, and phosphorus, as well as trace minerals, via the formation of insoluble complexes at intestinal pH. Phytate-protein complexes may also result in the reduced solubility of proteins, which can affect the functional properties of proteins.
Cyanogenic glycosides are plant toxins that upon hydrolysis, liberate hydrogen cyanide. The toxic effects of the free cyanide are well documented and affect a wide spectrum of organisms since their mode of action is inhibition of the cytochromes of the electron transport system 5. Hydrogen cyanide is known to cause both acute and chronic toxicity, but the hydrogen cyanide content of Mucuna pruriens seeds is far below the lethal level. Janardhan et al. 6 have investigated the concentration of oligosaccharides in Mucuna pruriens seeds, and verbascose is reportedly the principal oligosaccharide therein 7. Fatty acid profiles reveal that lipids are a good source of the nutritionally essential linoleic and oleic acids. Linoleic acid is evidently the predominant fatty acid, followed by palmitic, oleic, and linolenic acids 8. The nutritional value of linoleic acid is due to its metabolism at tissue levels that produce the hormone-like prostaglandins. The activity of these prostaglandins includes lowering of blood pressure and constriction of smooth muscle. Phytohemagglutinins (lectins) are substances possessing the ability to agglutinate human erythrocytes.
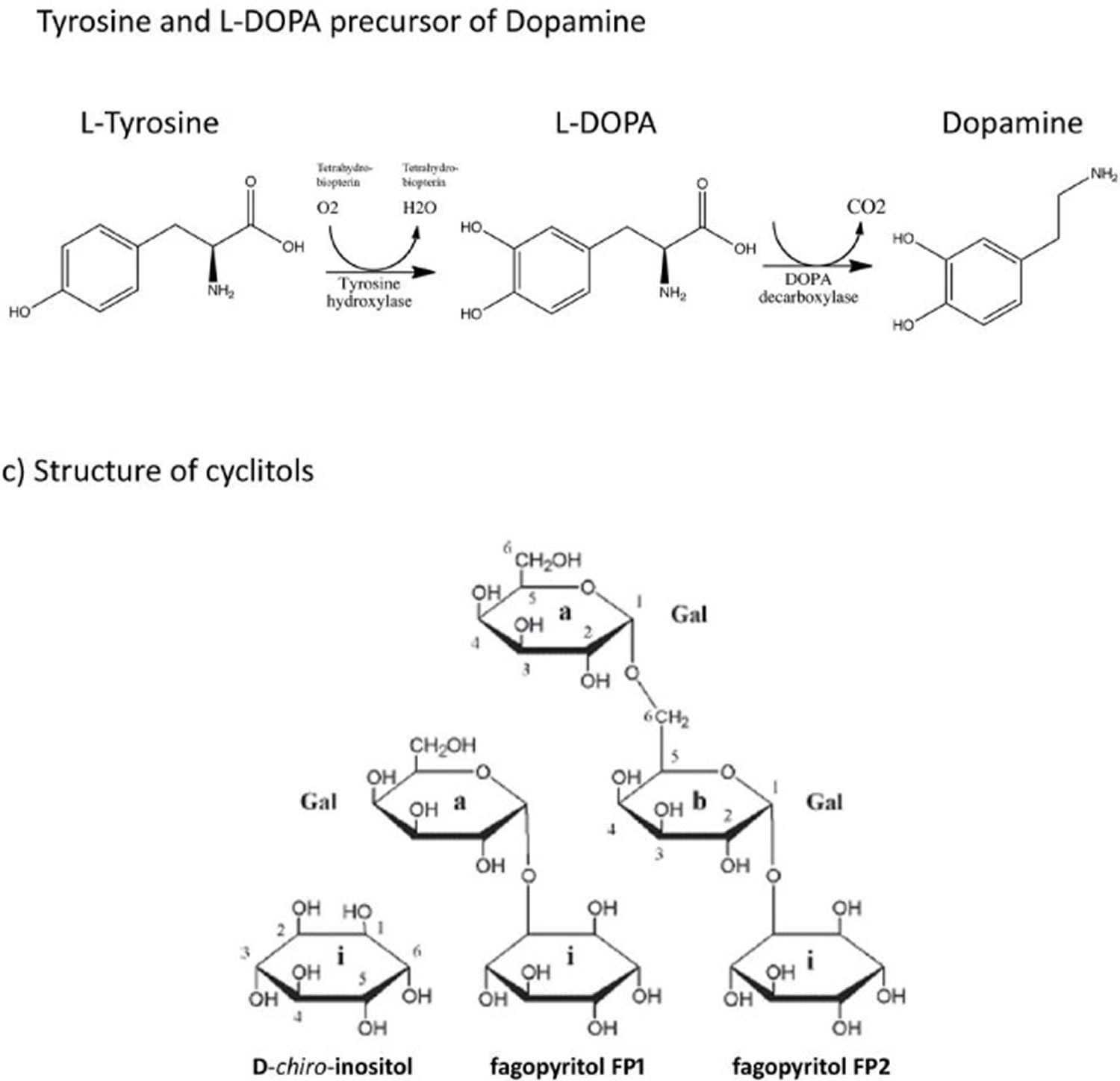
The major phenolic constituent of Mucuna pruriens beans was found to be levodopa (L-DOPA) (5%), along with minor amounts of methylated and non-methylated tetrahydroisoquinolines (0.25%) 9. However, in addition to L-dopa, 5-indole compounds, two of which were identified as tryptamine and 5-hydroxytryptamine, were also reported in Mucuna pruriens seed extracts 10. Mucunine, mucunadine, prurienine, and prurieninine are four alkaloids that have been isolated from such extracts 11. The chemical structures of some of these compounds are shown in Figure 3.
Figure 3. Mucuna pruriens bioactive compounds
[Source 4]Pharmacological effects of Mucuna pruriens extracts
All parts of the Mucuna plant possess medicinal properties 12. In vitro and in vivo studies on Mucuna pruriens extracts have revealed the presence of substances that exhibit a wide variety of pharmacological effects, including anti-diabetic, anti-inflammatory, neuroprotective and anti-oxidant properties, probably due to the presence of L-dopa, a precursor of the neurotransmitter dopamine (Misra and Wagner, 2007). It is known that the main phenolic compound of Mucuna seeds is L-dopa (approximately 5%) (Vadivel and Pugalenthi, 2008). Nowadays, Mucuna is widely studied because L-dopa is a substance used as a first-line treatment for Parkinson’s disease. Some studies indicate that L-dopa derived from Mucuna pruriens has many advantages over synthetic L-dopa when administered to Parkinson’s patients, as synthetic L-dopa can have several side effects when used for many years.
In small amounts (approximately 0.25%) L-dopa corresponds to methylated and non-methylated tetrahydroisoquinoline 9. These substances are present in the Mucuna roots, stems, leaves, and seeds. Other substances are present in different parts of the plant, among which are N,N-dimethyl tryptamine and some indole compounds 10. Alcoholic extracts of the seeds were shown to have potential anti-oxidant activity in in vivo models of lipid peroxidation induced by stress 10. On the other hand, Spencer et al. 13 have reported that the pro-oxidant and anti-oxidant actions of levodopa (L-DOPA) and its metabolites promote oxidative DNA damage and could also be harmful to tissues damaged by neurodegenerative diseases, namely parkinsonism.
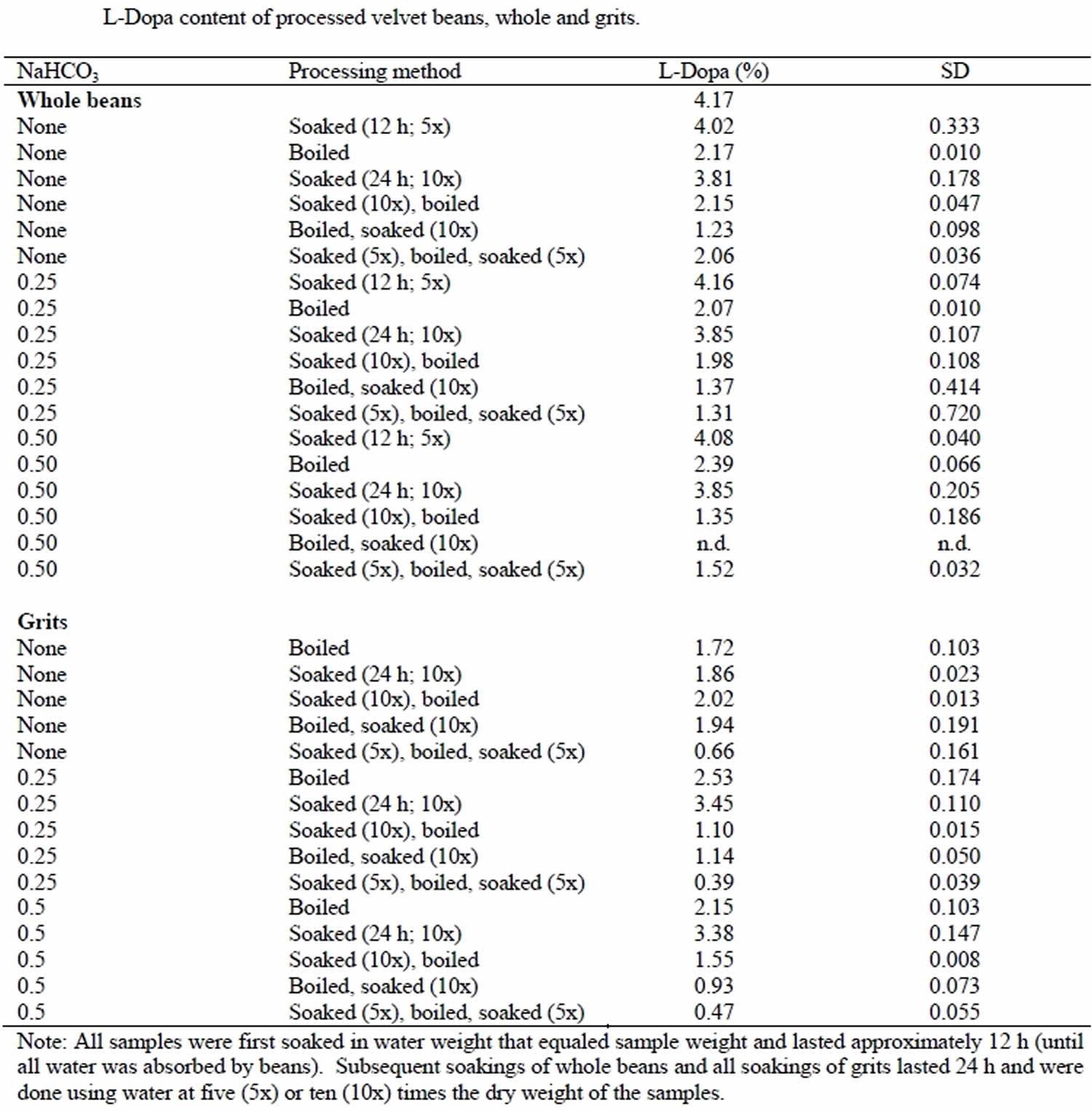
The Mucuna pruriens plant contains relatively high (3–7% dry weight) levels of levodopa (L-DOPA). The levodopa (L-DOPA) content of the raw local velvet bean varieties was 3.75, 3.90, and 4.36% for the white, speckled, and black beans, respectively 14. Some people who are sensitive to levodopa (L-DOPA) and may experience nausea, vomiting, cramping, arrhythmias, and hypotension. Up to 99% of the levodopa (L-DOPA) can be leached out of Mucuna pruriens by repeated soaking in boiling water and then cold water. Acidic water significantly increases the rate at which levodopa (L-DOPA) is leached out. Pre-boiling also contributes to better decomposition of anti-nutrients found in Mucuna pruriens through cooking 14. Soaking Mucuna pruriens grits in 1.5 L, boiling in 1.5 L and then soaking in 1.5 L water for 24 h, all in the presence of sodium bicarbonate (0.25%), extracted the most L-Dopa (90%; from 4.02 to 0.39 %) (Table 1) 14.
There is some limited evidence that Mucuna pruriens may have beneficial effects on some symptoms of Parkinson’s disease such as motor function because Mucuna pruriens seeds contain levodopa (L-DOPA) 15.
Mucuna pruriens and Parkinson disease research
- A 2018 non-inferiority, randomized, crossover, pilot study 16 with 14 Parkinson’s disease patients received Mucuna pruriens powder and levodopa+carbidopa. Daily intake of Mucuna pruriens resulted in 50% of patients discontinuing uses due to either gastrointestinal side-effects (n = 4) or worsening of motor performance (n = 3). During the levodopa+carbidopa phase no one discontinued use. For patients who tolerated Mucuna pruriens, clinical response was similar to levodopa+carbidopa.
- A 2017 randomized controlled trial 17 of 18 patients with advanced Parkinson’s disease found that Mucuna pruriens powder, at both high and low doses, is as effective and safe as levodopa/benserazide. The clinical response to Mucuna pruriens was similar to the effects of a pharmaceutical preparation of levodopa alone at similar doses, with less side effects. Mucuna pruriens could be a sustainable alternative to marketed levodopa for indigent individuals with Parkinson disease in low-income countries, provided that it is tolerated in the long term.
- A 2004 preliminary pilot study 18 of eight participants with Parkinson’s disease with a short duration L-dopa response and disabling peak dose dyskinesias found that the seed powder formulation of Mucuna pruriens contains a considerable quantity of L-dopa and has a rapid onset of action with a slightly longer duration of therapeutic response compared with standard L-dopa. Mucuna pruriens’s long-term efficacy and safety has not yet been established.
Safety
- The long-term safety of Mucuna pruriens has not yet been established.
Table 1. Levodopa (L-Dopa) content of processed Mucuna pruriens beans, whole and grits
[Source 14]Mucuna pruriens benefits
Mucuna pruriens use in traditional medicine
Mucuna pruriens is a popular Indian medicinal plant, which has long been used in traditional Ayurvedic Indian medicine, for diseases including parkinsonism 12 and as a powerful aphrodisiac 19. Mucuna pruriens have been used to treat nervous disorders and arthritis 20. The Mucuna pruriens bean, if applied as a paste on scorpion stings, is thought to absorb the poison 20.
Anti-epileptic and anti-neoplastic activity of methanol extract of Mucuna pruriens has been reported 21. A methanol extract of Mucuna pruriens seeds has demonstrated significant in vitro anti-oxidant activity, and there are also indications that methanol extracts of Mucuna pruriens may be a potential source of natural anti-oxidants and anti-microbial agents 22.
All parts of Mucuna pruriens possess valuable medicinal properties and it has been investigated in various contexts, including for its anti-diabetic, aphrodisiac, anti-neoplastic, anti-epileptic, and anti-microbial activities 12. Its anti-venom activities have been investigated by Guerranti et al. 23 and its anti-helminthic activity has been demonstrated by Jalalpure 24. Mucuna pruriens has also been shown to be neuroprotective 25, and has demonstrated analgesic and anti-inflammatory activity 26.
Mucuna pruriens seeds against snake venom poisoning
Mucuna pruriens is one of the plants that have been shown to be active against snake venom and, indeed, its seeds are used in traditional medicine to prevent the toxic effects of snake bites, which are mainly triggered by potent toxins such as neurotoxins, cardiotoxins, cytotoxins, phospholipase A2 (PLA2), and proteases 23. In Plateau State, Nigeria, the seed is prescribed as a prophylactic oral anti-snakebite remedy by traditional practitioners, and it is claimed that when the seeds are swallowed intact, the individual is protected for one full year against the effects of any snake bite 27. The mechanisms of the protective effects exerted by Mucuna pruriens seed aqueous extract (MPE), were investigated in detail, in a study involving the effects of Echis carinatus venom (EV) 23. In vivo experiments on mice showed that protection against the poison is evident at 24 hours (short-term), and 1 month (long term) after injection of Mucuna pruriens seed aqueous extract 28. Mucuna pruriens seed aqueous extract protects mice against the toxic effects of Echis carinatus venom via an immune mechanism 29. Mucuna pruriens seed aqueous extract contains an immunogenic component, a multiform glycoprotein, which stimulates the production of antibodies that cross-react with (bind to) certain venom proteins 23. This glycoprotein, called gpMuc, is composed of seven different isoforms with molecular weights between 20.3 and 28.7 kDa, and pI between 4.8 and 6.5 30.
It is likely that one or more gpMuc isoform is analogous in primary structure to venom PLA2. The presence of at least one shared epitope has been demonstrated with regard to Mucuna pruriens seeds and snake venom. These cross-reactivity data explain the mechanism of the long-term protection conferred by Mucuna pruriens, and confirm that certain plant species contain PLA2-like proteins, which are beneficial for plant growth, and are involved in important processes 31. In addition, Mucuna pruriens seeds contain protein and non-protein components that are able to directly inhibit the activity of proteases and PLA2, and are responsible for short-term protection. In fact, Mucuna pruriens seed aqueous extract contains protease inhibitors that are active against snake venom, in particular a gpMuc isoform sequence also found in a “Kunitz type” trypsin inhibitor contained in soy. Two-dimensional gel electrophoresis has been used to separate the seven gpMuc isoforms, in order to perform N-terminal analysis of each individual isoform. On the other hand, the direct inhibitory action of Mucuna pruriens seed aqueous extract is probably caused by L-dopa, the main bioactive component, which acts in synergy with other compounds.
Anti-microbial properties of Mucuna pruriens leaves
Various parts of certain plants are known to contain substances that can be used for therapeutic purposes or as precursors for the production of useful drugs 32. Plant-based anti-microbials represent a vast untapped source of medicines and further investigation of plant anti-microbials is needed. Anti-microbials of plant origin have enormous therapeutic potential. Phytochemical compounds are reportedly responsible for the anti-microbial properties of certain plants 33. While bioactive compounds are often extracted from whole plants, the concentration of such compounds within the different parts of the plant varies. Parts known to contain the highest concentration of the compounds are preferred for therapeutic purposes. Some of these active components operate individually, others in combination, to inhibit the life processes of microbes, particularly pathogens. Crude methanolic extracts of Mucuna pruriens leaves have been shown to have mild activity against some bacteria in experimental settings, probably due to the presence of phenols and tannins 34. Further studies are required in order to isolate the bioactive components responsible for the observed anti-microbial activity.
Neuroprotective effect of Mucuna pruriens seeds
In India, the seeds of Mucuna pruriens have traditionally been used as a nervine tonic, and as an aphrodisiac for male virility. The pods are anthelmintic, and the seeds are anti-inflammatory. Powdered seeds possess anti-parkinsonism properties, possibly due to the presence of L-dopa (a precursor of neurotransmitter dopamine). It is well known that dopamine is a neurotransmitter. The dopamine content in brain tissue is reduced when the conversion of tyrosine to L-dopa is blocked. L-Dopa, the precursor of dopamine, can cross the blood-brain barrier and undergo conversion to dopamine, restoring neurotransmission 35. Good yields of L-dopa can be extracted from Mucuna pruriens seeds with EtOH-H2O (1:1), using ascorbic acid as a protector 25. An n-propanol extract of Mucuna pruriens seeds yields the highest response in neuroprotective testing involving the growth and survival of DA neurons in culture. Interestingly, n-propanol extracts, which contain a negligible amount of L-dopa, have shown significant neuroprotective activity, suggesting that a whole extract of Mucuna pruriens seeds could be superior to pure L-dopa with regard to the treatment of parkinsonism.
Anti-diabetic effect of Mucuna pruriens seeds
Using a combination of chromatographic and NMR techniques, the presence of d-chiro-inositol and its two galacto-derivatives, O-α-d-galactopyranosil-(1→2)-d-chiro-inositol (FP1) and O-α-d-galactopyranosil-(1→6)-O-α-d-galactopyranosil-(1→2)-D-chiro-inositol (FP2), was demonstrated in Mucuna pruriens seeds 36. Galactopyranosyl d-chiro-inositols are relatively rare and have been isolated recently from the seeds of certain plants; they constitute a minor component of the sucrose fraction of Glycine max (Fabaceae) and lupins, and a major component of Fagopyrum esculentum (Polygonaceae) 37. Although usually ignored in phytochemical analyses conducted for dietary purposes, the presence of these cyclitols is of interest due to the insulin-mimetic effect of d-chiro-inositol, which constitutes a novel signaling system for the control of glucose metabolism 38. According to Akhtar et al., 39, Mucuna pruriens seeds used at a dose of 500 mg/kg reduced plasma glucose levels. These and other data demonstrated that the amount of seeds necessary to obtain a significant anti-diabetic effect contain a total of approximately 7 mg of d-chiro-inositol (including both free, and that derived from the hydrolysis of FP1 and FP2). The anti-diabetic properties of Mucuna pruriens seed EtOH/H2O 1:1 extract are most likely due to d-chiro-inositol and its galacto-derivatives.
Anti-oxidant activity of Mucuna pruriens
Free radicals that have one or more unpaired electrons are produced during normal and pathological cell metabolism. Reactive oxygen species (ROS) react readily with free radicals to become radicals themselves. Anti-oxidants provide protection to living organisms from damage caused by uncontrolled production of reactive oxygen species (ROS) and concomitant lipid peroxidation, protein damage and DNA strand breakage. Several substances from natural sources have been shown to contain anti-oxidants and are under study. Anti-oxidant compounds such as phenolic acids, polyphenols, and flavonoids, scavenge free radicals such as peroxide, hydroperoxide or lipid peroxyl, and thus inhibit oxidative mechanisms. Polyphenols are important phytochemicals due to their free radical scavenging and in vivo biological activities 40; the total polyphenolic content has been tested using Folin-Ciocalteau reagent. Flavonoids are simple phenolic compounds that have been reported to possess a wide spectrum of biochemical properties, including anti-oxidant, anti-mutagenic and anti-carcinogenic activity 41. The hydrogen donating ability of the methanol extract of Mucuna pruriens was measured in the presence of 1,1-diphenyl-2-picryl-hydrazyl (DPPH) radical. In a recent study, Kottai Muthu et al. 42 found that ethylacetate and methanolic extract of whole Mucuna pruriens plant (MEMP), which contains large amounts of phenolic compounds, exhibits high anti-oxidant and free radical scavenging activities. These in vitro assays indicate that this plant extract is a significant source of natural anti-oxidant, which may be useful in preventing various oxidative stresses. It has been reported 43 that methanolic extracts of Mucuna pruriens leaves have numerous biochemical and physiological activities, and contain pharmaceutically valuable compounds.
Mucuna pruriens for skin treatments
The skin is one of the main targets of several exogenous insults such as UV radiation, O3, and cigarette smoke, and all of these exert toxicity via the induction of oxidative stress 44. Several skin pathologies, such as psoriasis, dermatitis, and eczema, are related to increased oxidative stress and reactive oxygen species (ROS) production 45 and research investigating novel natural compounds with anti-oxidant proprieties is an expanding field. As mentioned above, certain plant-derived compounds have been an important source of traditional treatments for various diseases, and have received considerable attention in more recent years due to their numerous pharmacological proprieties.
Recent preliminary studies from our group have shown that human keratinocytes treated with a methanolic extract from Mucuna pruriens leaves exhibit downregulation of total protein expression. In addition, treatment with Mucuna pruriens significantly decreased the baseline levels of 4HNE present in human keratinocytes 46. This preliminary study suggests that evaluating the effect that topical Mucuna pruriens methanolic extract treatment may have on skin diseases would be worthwhile, as would further work aimed at clarifying the mechanisms involved in such effects.
Mucuna pruriens dosage
Based on the small data from a 2017 randomized controlled trial 17 of 18 patients with advanced Parkinson’s disease found that Mucuna pruriens powder, at both high dose at 17.5 mg/kg body weight and low dose at 12.5 mg/kg body weight, is as effective and safe as levodopa/benserazide.
When compared to levodopa+carbidopa, Mucuna pruriens powder low dose at 12.5 mg/kg body weight showed similar motor response with fewer dyskinesias and side effects, while Mucuna pruriens powder at high dose at 17.5 mg/kg body weight induced greater motor improvement at 90 and 180 minutes, longer ON duration, and fewer dyskinesias. Mucuna pruriens powder at high dose at 17.5 mg/kg body weight induced less side effects than levodopa+carbidopa and levodopa. No differences in cardiovascular response were recorded.
Mucuna pruriens side effects
Side effects most commonly occurred within 30–45 minutes after treatment administration and lasted <15 minutes, except for 4 patients, who reported side effects lasting >90 minutes after levodopa 17. Despite similar levodopa dose, levodopa was associated with more frequent side effects than Mucuna pruriens powder at high dose at 17.5 mg/kg body weight, and it was the only treatment associated with prolonged side effects. There was no difference among the active treatments in terms of change in blood pressure and heart rate between off and on state.
Levodopa side effects
Along with its needed effects, a medicine may cause some unwanted effects. Although not all of these side effects may occur, if they do occur they may need medical attention.
Check with your doctor as soon as possible if any of the following side effects occur:
More common
- abnormal thinking: holding false beliefs that cannot be changed by fact
- agitation
- anxiety
- clenching or grinding of teeth
- clumsiness or unsteadiness
- confusion
- difficulty swallowing
- dizziness
- excessive watering of mouth
- false sense of well being
- feeling faint
- general feeling of discomfort or illness
- hallucinations (seeing, hearing, or feeling things that are not there)
- hand tremor, increased
- nausea or vomiting
- numbness
- unusual and uncontrolled movements of the body, including the face, tongue, arms, hands, head, and upper body
- unusual tiredness or weakness
Less common
- blurred vision
- difficult urination
- difficulty opening mouth
- dilated (large) pupils
- dizziness or lightheadedness when getting up from a lying or sitting position
- double vision
- fast, irregular, or pounding heartbeat
- hot flashes
- increased blinking or spasm of eyelids
- loss of bladder control
- mental depression
- other mood or mental changes
- skin rash
- unusual weight gain or loss
Rare
- back or leg pain
- bloody or black tarry stools
- chills
- convulsions (seizures)
- fever
- high blood pressure
- inability to move eyes
- loss of appetite
- pain, tenderness, or swelling of foot or leg
- pale skin
- prolonged, painful, inappropriate penile erection
- sore throat
- stomach pain
- swelling of face
- swelling of feet or lower legs
- vomiting of blood or material that looks like coffee grounds
Some side effects may occur that usually do not need medical attention. These side effects may go away during treatment as your body adjusts to the medicine. Also, your health care professional may be able to tell you about ways to prevent or reduce some of these side effects. Check with your health care professional if any of the following side effects continue or are bothersome or if you have any questions about them:
More common
- abdominal pain
- dryness of mouth
- loss of appetite
- nightmares
- passing gas
Less common
- constipation
- diarrhea
- flushing of skin
- headache
- hiccups
- increased sweating
- muscle twitching
- trouble in sleeping
Levodopa may sometimes cause the urine, saliva, and sweat to be darker in color than usual. The urine may at first be reddish, then turn to nearly black after being exposed to air. Some bathroom cleaning products will produce a similar effect when in contact with urine containing levodopa. This is to be expected during treatment with levodopa. Also, levodopa may cause a bitter taste, or a burning sensation of the tongue.
Other side effects not listed may also occur in some patients. If you notice any other effects, check with your healthcare professional.
Call your doctor for medical advice about side effects.
Nervous system side effects
Nervous system side effects most frequently reported have included involuntary movements and mental status changes (in as many as 50% of treated patients on long-term therapy). The types of involuntary movements due to levodopa have been characterized as choreiform, dystonic and dyskinetic. Fluctuations in motor function occur frequently and often increase as the duration of therapy increases.
Choreiform movements due to levodopa therapy may occur in as many as 80% of patients treated for one year and frequently involve facial grimacing, exaggerated chewing, and twisting and protrusion of the tongue.
Several types of motor fluctuations may occur and result in “bradykinetic episodes”. Some motor fluctuations are related to the timing of levodopa dosage administration. For example, patients may experience “peak of the dose dyskinesia” and a wearing-off effect called “end of the dose akinesia”. The “wearing-off effect may result in early morning dystonia. Such motor fluctuations may be managed by increasing the frequency of dosage administration and decreasing the dose administered to achieve a smoother therapeutic effect.
Other motor fluctuations are not related to the timing of dose administration. Such fluctuations are characterized by sudden loss of levodopa effect which may last for minutes to hours and result in akinesia followed by a sudden return of levodopa effect. These “on-off” fluctuations may occur many times per day. “On-off” fluctuations may respond to more frequent dose administration.
Finally, akinesia paridoxica is a sudden episode of akinesia which occurs as patients begin to walk. Akinesia paridoxica frequently results in falls and often responds to levodopa dose reductions.
Other adverse nervous system effects due to levodopa include myoclonus, sleep disturbances (including insomnia, daytime somnolence, altered dreams and episodic nocturnal myoclonus), Meige’s syndrome (blepharospasm-oromandibular dystonia) and ocular dyskinesia. In addition, the orofacial movements induced by levodopa have occasionally been reported to cause severe dental erosion.
Some investigators have suggested that levodopa may cause brain dysfunction and may have negative effects on cognitive performance. Levodopa “drug holidays” have been proposed by some investigators as potentially beneficial (perhaps by causing dopamine receptor resensitization). However, the therapeutic value of these drug holidays is controversial.
Gastrointestinal side effects
Exacerbation of preexisting stomach ulcer disease with severe upper gastrointestinal bleeding has been reported.
Gastrointestinal side effects most commonly reported have included nausea and vomiting. Anorexia and gastrointestinal hemorrhage have been reported rarely.
Psychiatric side effects
Some authors have suggested that clozapine may be useful in the management of levodopa-induced psychotic symptoms.
Other investigators have suggested that levodopa may induce alterations in the noradrenergic systems of the CNS which may lead to panic attacks.
Psychiatric side effects have included hallucinations (particularly visual hallucinations), psychosis, confusion, anxiety, mania, hypomania, depression, rapid mood cycling, nightmares, and hypersexuality.
Neuroleptic malignant syndrome
Fever, altered consciousness, autonomic dysfunction and muscle rigidity are the hallmarks of the neuroleptic malignant syndrome. The neuroleptic malignant syndrome (NMS) is associated with a case fatality rate of about 20%. If withdrawal of dopaminergic therapy is suspected as the cause of NMS (neuroleptic malignant syndrome), dopaminergic therapy should be restarted. If a neuroleptic agent is suspected as the cause, the neuroleptic agent should be immediately discontinued. For patients with NMS (neuroleptic malignant syndrome) suspected to be due to neuroleptic therapy, consideration should be given to dantrolene (or bromocriptine) administration. Intensive monitoring and supportive care are indicated for all patients with NMS (neuroleptic malignant syndrome).
Sudden discontinuation or rapid tapering of levodopa therapy may result in acute worsening of parkinsonism or less frequently, in a syndrome resembling the neuroleptic malignant syndrome. Cases of psychologic levodopa addiction have also been reported rarely.
Cardiovascular side effects
Some authors have reported marked hemodynamic and clinical improvements in patients with congestive heart failure treated with oral levodopa. However, at least one author has reported marked hemodynamic deterioration following such treatment.
Cardiovascular side effects have included hypotension and syncope. Arrhythmias have also been reported rarely.
Dermatologic side effects
Dermatologic side effects have included a number of cases of malignant melanoma in patients taking levodopa for Parkinson’s Disease. Additionally, several cases of maculopapular skin rashes have been reported in patients taking levodopa-containing drugs.
Despite reports of melanoma occurring in levodopa-treated patients, some authors have suggested that a causal association is tenuous and other authors have suggested that levodopa may have an antitumor effect on melanoma. Nevertheless, the manufacturers of levodopa-containing drugs report that either the history of melanoma or the presence of suspicious skin lesions is a contraindication for the use of levodopa-containing drugs.
Immunologic side effects
Immunologic side effects have included rare reports of a lupus-like syndrome.
Hematologic side effects
Hematologic side effects reported rarely have included severe hemolytic and nonhemolytic anemias.
Respiratory side effects
Respiratory side effects have included dyskinesias (occasionally of life-threatening severity).
Liver side effects
Hepatic side effects have included rare cases of asterixis (without abnormalities of liver function tests). The manufacturer of levodopa-containing products reports that abnormal liver function tests may occur.
Endocrine side effects
Endocrine side effects have included elevated urinary vanillylmandelic acid levels which have occasionally led to confusion concerning the diagnosis of pheochromocytoma.
Kidney side effects
Renal side effects have included hypokalemia and hyponatremia. Chronic administration of levodopa may also slightly but significantly increase BUN without changes in the glomerular filtration rate.
References- Mucuna pruriens (L.) DC. https://npgsweb.ars-grin.gov/gringlobal/taxonomydetail.aspx?id=24652
- Mucuna pruriens http://www.tropicalforages.info/key/forages/Media/Html/entities/mucuna_pruriens.htm
- Human Surrogate Models of Histaminergic and Non-histaminergic Itch. Acta Derm Venereol. 2015 Sep;95(7):771-7. doi: 10.2340/00015555-2146. https://www.medicaljournals.se/acta/content/html/10.2340/00015555-2146
- Lampariello LR, Cortelazzo A, Guerranti R, Sticozzi C, Valacchi G. The Magic Velvet Bean of Mucuna pruriens. Journal of Traditional and Complementary Medicine. 2012;2(4):331-339. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3942911/
- Laurena A.C, Revilleza M.J.R, Mendoza E.M.T. Polyphenols phytate, cyanogenic glycosides and trypsin inhibitor activity of several Philippine indigenous food legumes. J. of Food Comp. and Analys. 1994;7:194–202.
- Janardhanan K, Gurumoorthi P, Pugalenthi M. Nutritional potential of five accessions of a South Indian tribal pulse, Mucuna pruriens var. utilis. Part I. The effect of processing methods on the contents of L-Dopa phytic acid, and oligosaccharides. Journal of Tropical and Subtropical Agro-ecosystems. 2003;1:141–152.
- Siddhuraju P, Becker K, Makkar H.P.S. Studies on the nutritional composition and antinutritional factors of three different seed material of an under-utilised tropical legume, Mucuna pruriens var. utilis. J. Agric. Food Chem. 2000;48:6048–6060.
- Mohan V.R, Janardhanan K. Chemical analysis and nutritional assessment of lesser-known pulses of the genus. Mucuna Food Chemistry. 1995;52:275–280.
- Siddhuraju P, Becker K. Rapid reversed-phase high performance liquid chromatographic method for the quantification of L-Dopa (L-3,4-dihydroxyphenylalanine), non-methylated and methylated tetrahydroisoquinoline compounds from Mucuna beans. Food Chem. 2001;72:389–394.
- Tripathi Y.B, Updhyay A.K. Antioxidant property of Mucuna pruriens. Linn. Curr. Sci. 2001;80:1377–1378.
- Mehta J.C, Majumdar D.N. Indian medicinal plants-V Mucuna pruriens bark (N.O; Papilionaceae) Ind J Pharm. 1994;6:92–94.
- Sathiyanarayanan L, Arulmozhi S. Mucuna pruriens A comprehensive review. Pharmacognosy Rev. 2007;1:157–162.
- Spencer J.P.E, Jenner P, Halliwell B. Superoxide-dependent GSH depletion by L-Dopa and dopamine Relevance to Parkinson’s disease. Neuroreport. 1995;6:1480–1484
- The effects of different processing methods of velvet beans (Mucuna pruriens) on L-dopa content, proximate composition and broiler chicken performance. Tropical and Subtropical Agroecosystems, 1 (2003): 253-260. http://www.redalyc.org/pdf/939/93911288026.pdf
- Parkinson’s Disease and Complementary Health Approaches: What the Science Says. https://nccih.nih.gov/health/providers/digest/parkinson-disease-science
- Daily intake of Mucuna pruriens in advanced Parkinson’s disease: A 16-week, noninferiority, randomized, crossover, pilot study. Parkinsonism Relat Disord. 2018 Apr;49:60-66. doi: 10.1016/j.parkreldis.2018.01.014. Epub 2018 Jan 11. https://www.prd-journal.com/article/S1353-8020(18)30014-2/fulltext
- Cilia R, Laguna J, Cassani E, et al. Mucuna pruriens in Parkinson disease: A double-blind, randomized, controlled, crossover study. Neurology. 2017;89(5):432-438. doi:10.1212/WNL.0000000000004175. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5539737/
- Katzenschlager R, Evans A, Manson A, et al. Mucuna pruriens in Parkinson’s disease: a double blind clinical and pharmacological study. Journal of Neurology, Neurosurgery, and Psychiatry. 2004;75(12):1672-1677. doi:10.1136/jnnp.2003.028761. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1738871/pdf/v075p01672.pdf
- Amin K.M.Y, Khan M.N, Zillur-Rehman S, Khan N.A. Sexual function improving effect of Mucuna pruriens in sexually normal male rats. Fitoterapia Milano. 1996;67:53–56.
- Jeyaweera D.M.A. Sri Lanka: National Science Council of Sri Lanka; 1981. Madicinal plants used in Ceylon Colombo.
- Gupta M, Mazumder U.K, Chakraborti S, Bhattacharya S, Rath N, Bhawal S.R. Antiepileptic and anticancer activity of some indigenous plants. Indian J. of Physiol. Allied Sci. 1997;51:53–56.
- Rajeshwar Y, Kumar S.G.P, Gupta M, Mazumder K.U. Studies on in vitro antioxidant activities of mhetanol extract of Mucuna pruriens (Fabaceae) seeds. European Bull of Drug Research. 2005;13:31–39.
- Guerranti R, Aguiyi J.C, Ogueli I.G, Onorati G, Neri S, Rosati F, Del Buono F, Lampariello R, Leoncini R, Pagani R, Marinello E. Protection of Mucuna pruriens seeds against Echis carinatus venom is exerted through a multiform glycoprotein whose oligosaccharide chains are functional in this role. BBRC. 2004;323:484–490.
- Jalalpure S.S, Alagawadi K.R, Mahajanashell C.S. In vitro antihelmintic property of various seed oils against Pheritima posthuma. Ind Pharm Sci. 2007;69:158–160.
- Misra L, Wagner H. Extraction of bioactive principles from Mucuna pruriens seeds. Indian J. Biochem. Biophys. 2007;44:56–60.
- Hishika R, Shastry S, Shinde S, Guptal S.S. Preliminary phytochemical and anti-inflammatory activity of seeds of Mucuna pruriens. Indian J pharmacol. 1981;13(1):97–98.
- Guerranti R, Aguiyi J.C, Errico E, Pagani R, Marinello E. Effects of Mucuna pruriens extract on activation of prothrombin by Echis carinatus venom. J Ethnopharmacol. 2001;75:175–180.
- Guerranti R, Ogueli I.G, Bertocci E, Muzzi C, Aguiyi J.C, Cianti R, Armini A, Bini L, Leoncini R, Marinello E, Pagani R. Proteomic analysis of the pathophysiological process involved in the antisnake venom effect of Mucuna pruriens extract. Proteomics. 2008;8:402–412.
- Guerranti R, Aguiyi J.C, Neri S, Leoncini R, Pagani R, Marinello E. Proteins from Mucuna pruriens and enzymes from Echis carinatus venom: characterization and cross-reactions. J Biol Chem. 277:17072–17078.
- Di Patrizi L, Rosati F, Guerranti R, Pagani R, Gerwig G.J, Kamerling J.P. Structural characterization of the N-glycans of gpMuc from Mucuna pruriens seeds. 2006
- Lee H.Y, Bahn S.C, Shin J.S, Hwang I, Back K, Doelling J.H, Ryu S.B. Multiple forms of secretory phospholipase A2 in plants. Prog. Lipid. Res. 2005;44:52–67
- Sofowora A. 1st Edn. London: John Wiley and Sons; 1982. Medicinal plants in traditional medicine in West Africa.
- Mandal P, Sinha Babu S.P, Mandal N.V. Antimicrobial activity of saponins from Acacia auriculiformis. Fitoterapia. 2005;76:462–465.
- Ogundare A.O, Olorunfemi O.B. Antimicrobial efficacy of the leale of Dioclea reflexa, Mucana pruriens, Ficus asperifolia and Tragia spathulata. Res. J. of Microbiol. 2007;2:392–396.
- Kulhalli P. Heritage Healing. 1999 Jul;:29–30.
- Donati D, Lampariello L.R, Pagani R, Guerranti R, Cinci G, Marinello E. Antidiabetic oligocyclitols in seeds of Mucuna pruriens. Phytotherapy Res. 2005;19:1057–1060.
- Horbovitz M, Brenac P, Obendorf R.L. Fagopyritol B1, O-α- D-galactopyranosyl-(1→2)-D-chiro-inositol, a galactosylcyclitol in maturing buckwheat seeds associated with desiccation tolerance. Planta. 1998;205:1–11.
- Larner J, Allan G, Kessler C, Reamer P, Gunn R, Huang L.C. Phosphoinositol glycan derived mediators and insulin resistance Prospects for diagnosis and therapy. J. Basic Clin. Physiol. Pharmacol. 1998;9:127–137.
- Akhtar M.S, Qureshi A.Q, Iqbal J. Antidiabetic evaluation of Mucuna pruriens Linn seeds. JPMA. 1990;40:147–150.
- Bravo L, Siddhuraju P, Saura-Calixto F. Effect of various processing methods on the in vitrostarch digestibility and resistant starch content of Indian pulses. J. Agric. Food. Chem. 1998;46:4667–4674.
- Beta T, Nam S, Dexter J.E, Sapirstein H.D. Phenolic content and antioxidant activity of pearled wheat and roller-milled fractions. Cereal Chem. 2005;82:390–393.
- Kumar D.S, Muthu Kottai A, Smith A.A, Manavalan R. In vitro antioxidant activity of various extracts of whole plant of Mucuna pruriens (Linn) Int. J. Pharm. Tech. Res. 2010;2:2063–2070.
- Ujowundu C.O, Kalu F.N, Emejulu A.A, Okafor O.E, Nkwonta C.G, Nwosunjoku E. Afr. J. Pharm. Pharmacol. 2010;4:811–81.
- Valacchi G, Weber S.U, Luu C, Cross C.E, Packer L. Ozone potentiates vitamin E depletion by ultraviolet radiation in the murine stratum corneum. FEBS Lett. 2000;466:165–8.
- Briganti S, Picardo M. Antioxidant activity, lipid peroxidation and skin diseases What’s new. J Eur Acad Dermatol Venereol. 2003;17:663–9.
- Lampariello L.R, Cortelazzo A, Sticozzi C, Belmonte G, Guerranti R, Di Capua A, Anzini M, Valacchi G. Alba (Italy): 2012. Jun, Proeteomic profiling and post-trasductional modifications in human keratinocytes treated with Mucona Pruriens leale extract Oxygen Club of California World Congress 2012. Oxidants and Antioxidants in Biology Cell Signalling and Nutrient-Gene Interactions; pp. 20–23. 2012.