What is a sacral dimple
A sacral dimple is an indentation, present at birth, in the skin on the lower back. It’s usually located just above the crease between the buttocks. Most sacral dimples are harmless and don’t require any treatment.
Sacral dimples that are accompanied by a nearby tuft of hair, skin tag or certain types of skin discoloration are sometimes associated with a serious underlying abnormality of the spine or spinal cord. In these instances, your child’s doctor may recommend an imaging test. If an abnormality is discovered, treatment depends on the underlying cause.
Sacral dimple newborn
Sacral dimples are one of the commonest spinal cutaneous abnormalities seen in the newborn. Sacral dimples or “pits” result from incomplete closure of the neural tube during embryogenesis. The newborn or neonatal period is the first 4 weeks of extrauterine life. The skin of the newborn differs from adult in several ways. The thickness of newborn skin is 40% to 60% of that of adult skin. It has weaker intercellular attachment and produces lesser amount of sweat.
Most often, sacral dimples in newborn are benign lesions, especially as a solitary finding with no associated skin stigmata, and as such do not require further investigation or imaging studies 1, 2. Current data 3 shows that a screening ultrasound is appropriate only when other signs/lesions are found along with a sacral dimple. Further studies including MRI are required to further stratify other cutaneous findings and their association with underlying occult spinal dysraphism.
These cutaneous coccygeal and sacral dimples, most of which are below the intragluteal crease, occur in as many as 4.8% of all children 4. Yet the incidence of the “true” problematic lesions related to these dimples, such as spinal dysraphism, is only about 1 in every 2,500 births; spinal lipoma, which occurs in 1 of 4,000 births; and dermal sinuses, which occurs in 1 of 2,500 births 4.
Although problematic sacral dimple in newborn is rare 4, sacral dimple can be associated with a congenital dermal sinus tract, which is a type of closed spinal dysraphism characterized by an epithelium-lined sinus tract extending inward from the skin surface for a variable distance often into the lining of the spinal cord (thecal sac) 5. Congenital dermal sinus occurs during neurulation when the neural groove closes to form the neural tube on day 26 of gestation and results incomplete fusion of the midline mesenchymal, bony, or neural elements of the spine 6. As skin and nervous tissue are of ectodermal origin, anomalies of both may occur simultaneously. Occult spinal dysraphism is characterized by skin-covered lesions without exposed neural tissue.
Markers of occult spinal dysraphism include:
- Subcutaneous lipomas,
- Dermal sinuses,
- Tails, and
- Localized hypertrichosis (hairy patches).
A combination of 2 or more congenital midline skin lesions is the strongest marker of occult spinal dysraphism.
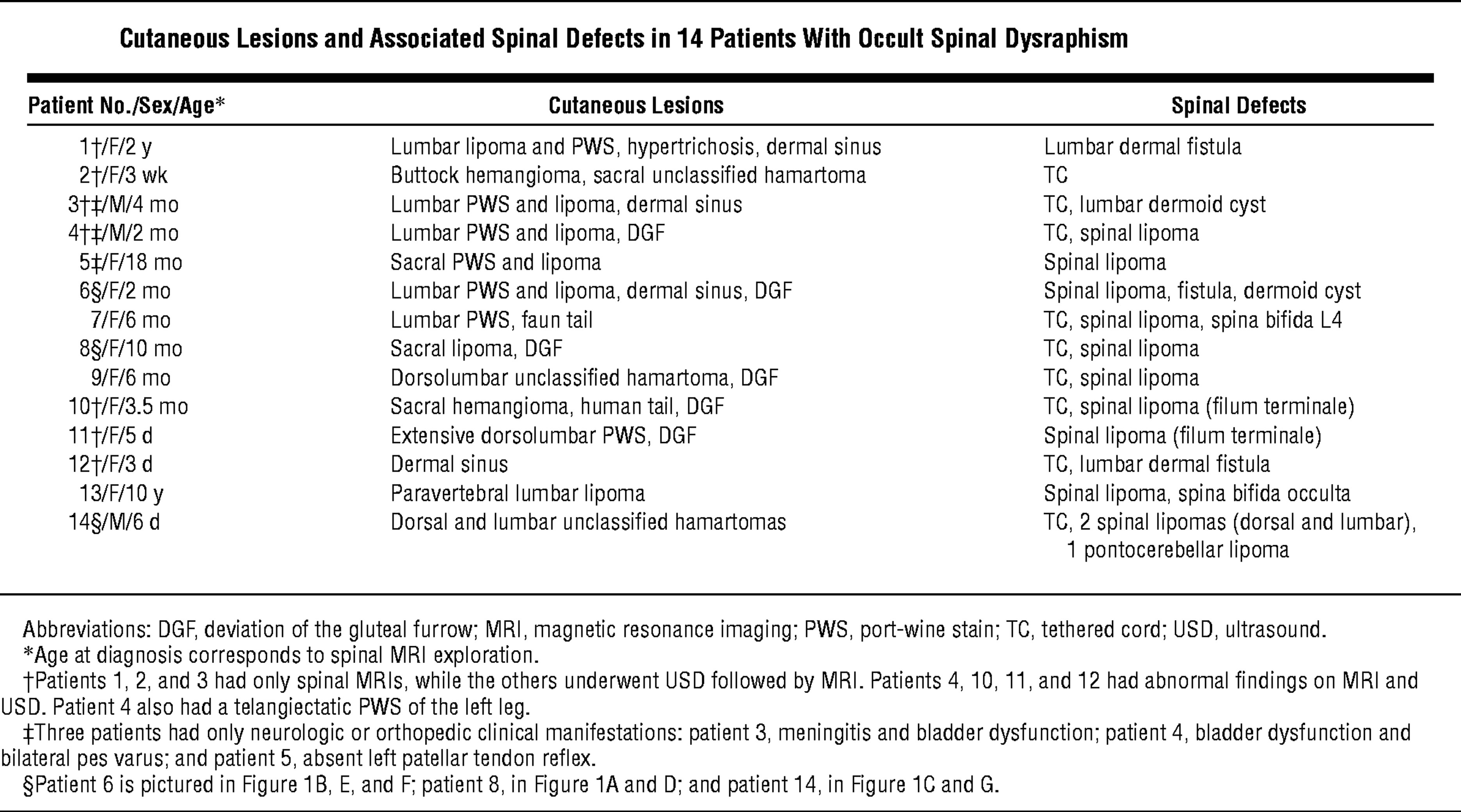
Table 1. Cutaneous lesions associated with occult spinal dysraphism
[Source 7]In the presence of such cutaneous lesions, radiologic investigations must be performed to detect a possible occult spinal dysraphism 8. Hyperpigmented lesions and aplasia cutis congenita are also reported to be associated with occult spinal dysraphism 9.
The incidence of congenital dermal sinus tract is approximately 1:2500–3000 live-births 10. These tracts may be accompanied with other pathologies such as lipomyelomeningocele, myelomeningocele, split cord malformation, tethered cord, filum abnormality and dermoid and epidermoid tumors and posterior arch defects of the vertebral column can be seen in association of dorsal dermal sinuses 11. The relationship of these findings with defects that can cause subsequent neurological sequelae and disability.
A recent study involving 1000 newborns found that sacral dimples in newborn were the most common cutaneous signs of spinal dysraphism of at 12.8% (more common than myelomeningoceles (a type of spina bifida): 0.5%, acrochordons: 0.1%, and dermoid cysts: 0.1%) 12.
In one study of 943 patients referred for cutaneous stigmata, 68% (638 patients) had a sacral dimple. Of these 638 patients, the resultant ultrasound was normal in 600 patients (where one patient had fatty filum on MRI requiring surgery), and 38 patients exhibited abnormal ultrasounds (with 4 undergoing subsequent surgical repair) 13. Based on this study by Chern et al. 13, approximately 5% of patients with cutaneous stigmata will have abnormal lumbar ultrasonography and surgery was only required in less than 1% of infants. In another study of 216 patients who were subjected to ultrasound imaging, the authors found that having multiple indications, as opposed to findings of isolated sacral dimples were, only at that point, six times more likely to yield the diagnosis of spinal dysraphism 14. Other signs, in contrast, may warrant further investigation to avoid detrimental outcomes as recently outlined in a recent case of midline hypertrichosis in a newborn 15. These findings are supported by other studies that propose further imaging only when two or more cutaneous lesions are found 16. This questions the value of imaging in detecting closed defects such as spinal lipoma, cord tethering, or fatty filum in the presence of only a sacral dimple without any other cutaneous stigmata.
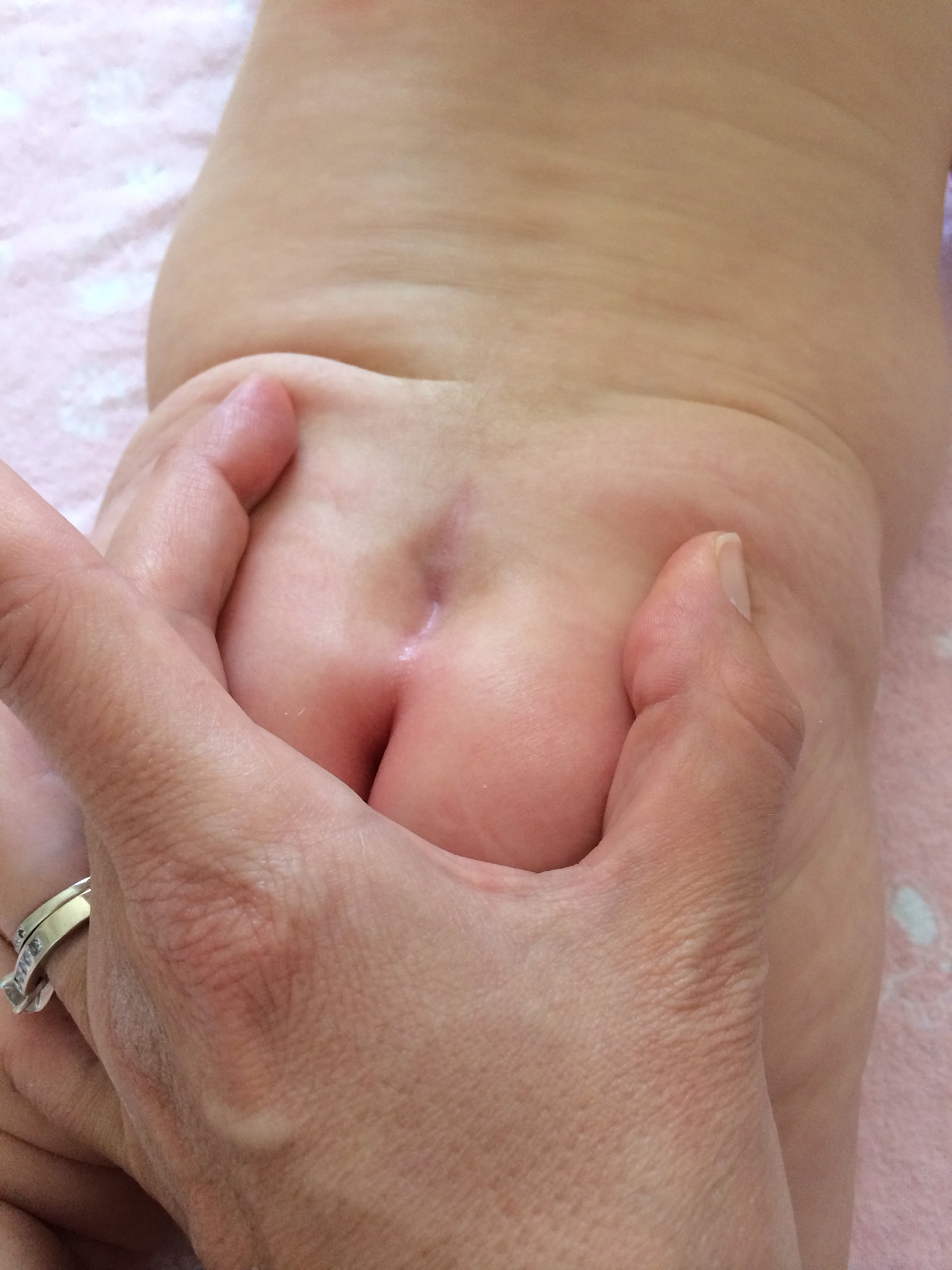
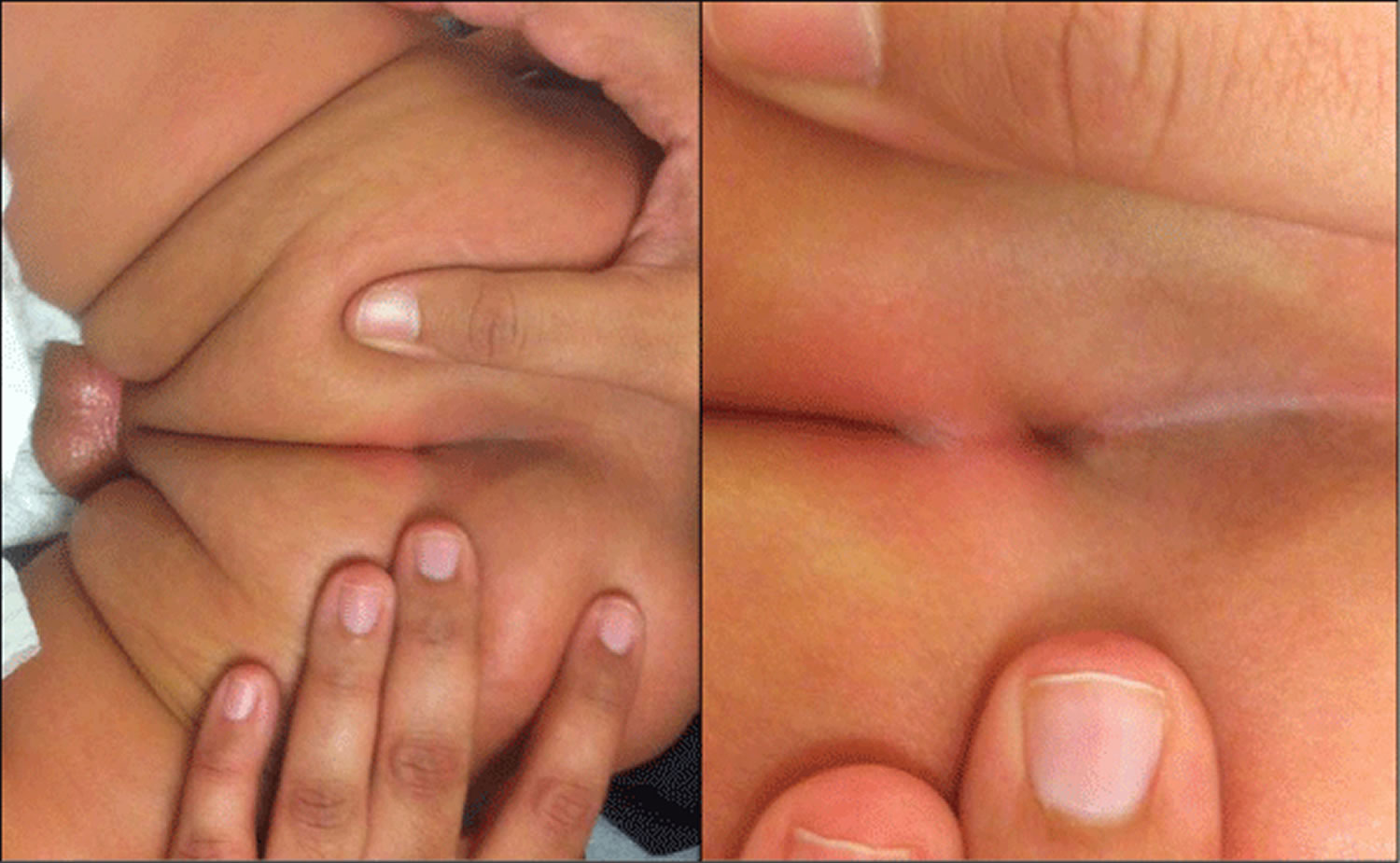
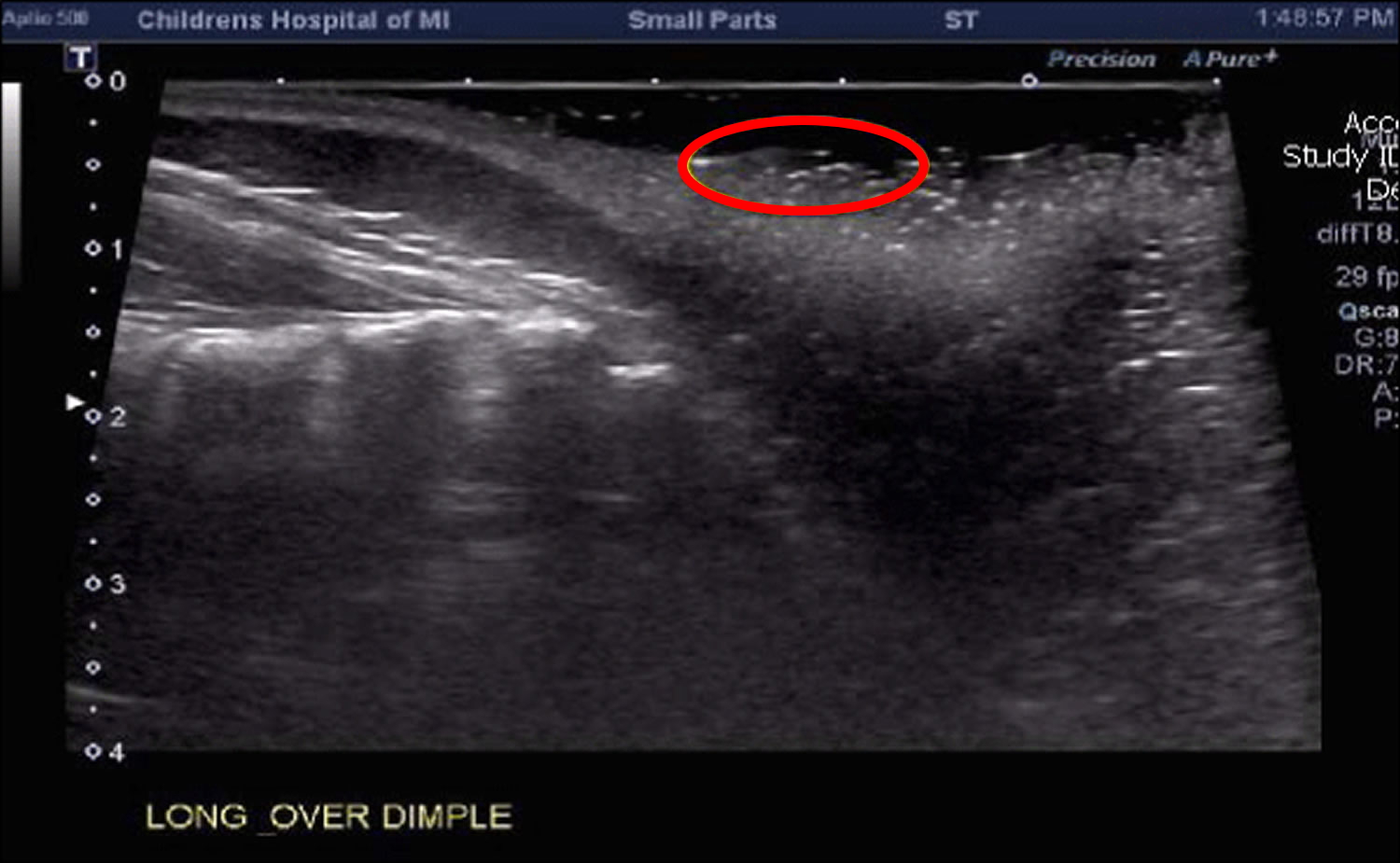
A recent report demonstrates that sacral dimples issues can occur and manifest even in adulthood-including motor weakness, incontinence, and chronic pain 17. As such, many doctors feel compelled to further evaluate the mere simple dimple in the sacro-coccygeal area, at least by ultrasound in most of these children. Clinical (Figures 1), as well as ultrasound (Figure 2) findings from a typical patient are provided.
High-resolution spinal ultrasound allows quick and noninvasive evaluation of an occult spinal dysraphism in the newborn 18. It does not require any premedication, and it costs less than MRI. Ultrasound can be applied easily and constitutes a good means of screening newborns suspected of occult spinal dysraphism because the posterior elements of the spine are nonossified in the neonate before age 5 to 6 months 19.
When to get an Ultrasound or MRI?
As a rule, lumbar MRI should always be preceded by a screening ultrasound given the disparity in absolute risk between the two modalities. With this in mind, the biggest concern then turns towards the sensitivity of ultrasound as a screening tool. A study by Sasani et al. demonstrates an overall discordance of 16.58% between ultrasound and MRI findings 20. Meanwhile, a recent comparative study using MRI as the gold standard showed that while ultrasound can be very specific for underlying occult spinal dysraphism (67- 100%) it is not nearly as sensitive (27-86%) 21. Although definitive data is still lacking, the presence of at least one additional cutaneous finding increases this sensitivity exponentially 14. In fact, several other cutaneous findings have been found to warrant prompt investigation regardless of their association with sacral dimples or not, including spinal lipomas and hairy patches as amongst the most prominent 22. The level of urgency associated with such findings should not be extended to that of isolated sacral dimples until further data concludes otherwise. A recent prospective observational study showed that ultrasound had 96% sensitivity, specificity and positive predictive value when compared with MRI in low risk skin dimples and deviated gluteal clefts. In fact, the researchers feel that simple dimples and deviated gluteal clefts do not require any imaging whatsoever 23.
Figure 1. Sacral dimple newborn – a prototypical benign sacral dimple that is located within the gluteal cleft (less than 2.5 cm above the anus) and solitary.
Figure 2. Sacral dimple ultrasound – sagittal ultrasound showing normal conus level and no underlying spinal dysraphism (red circle is approximate area of cutaneous sacral dimple)
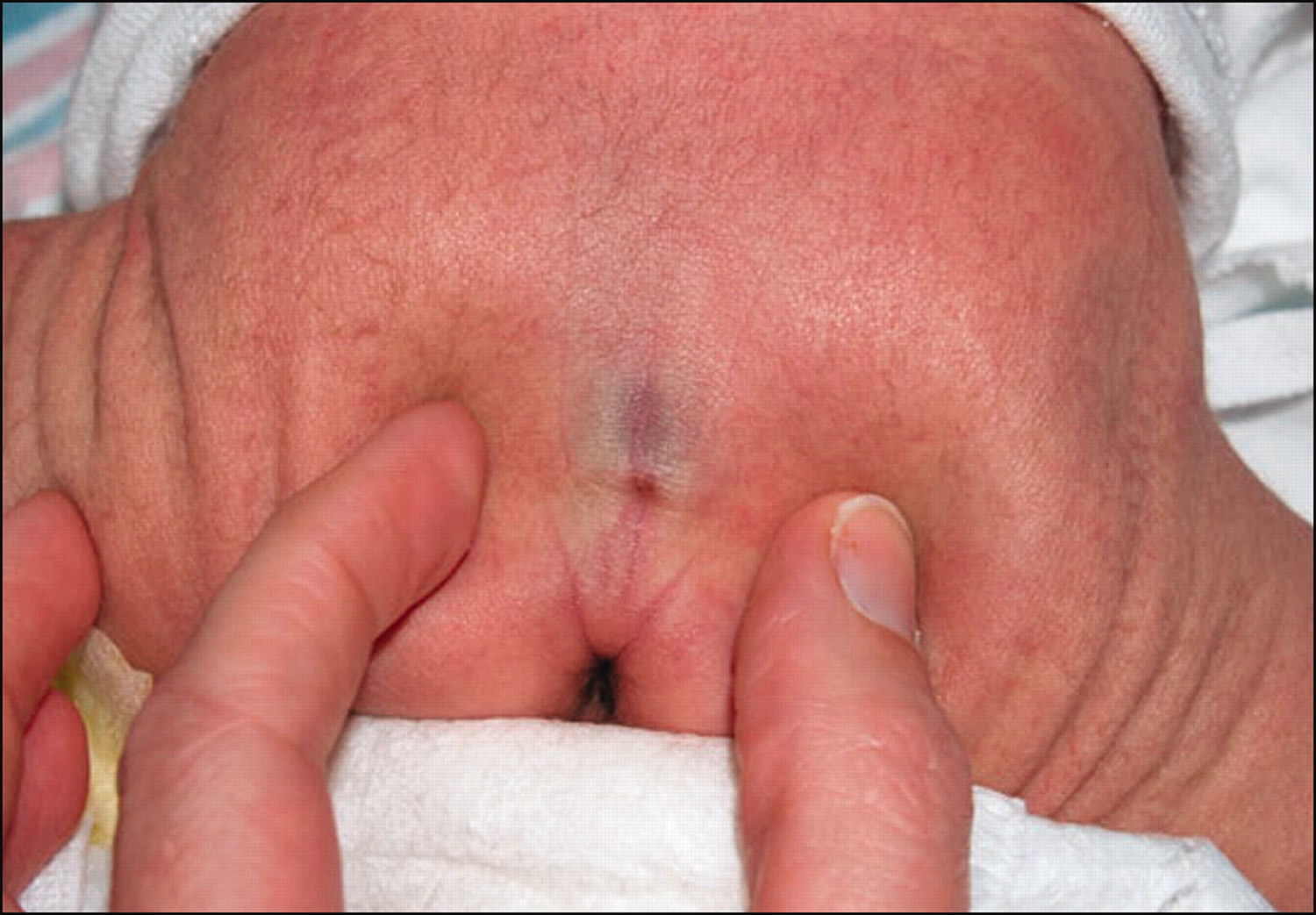
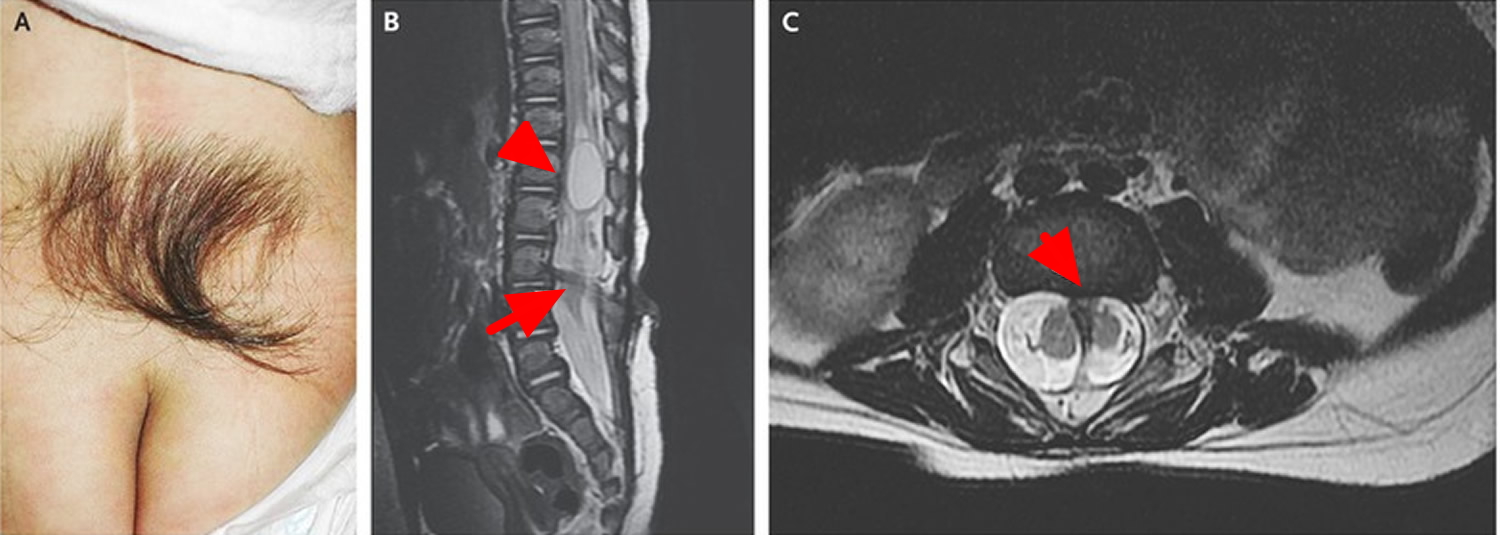
Figure 3. Spinal dysraphism
Note: A 3-year-old girl was referred for the removal of excess hair on the back, which had been present since birth, for cosmetic reasons. The physical examination revealed a tuft of coarse, dark hair in the midline of the back just above the natal cleft (Panel A). A patch of hair in the lower back may be a cutaneous marker of spinal dysraphism and should prompt consideration of underlying spinal abnormalities to prevent neurologic sequelae. Her history revealed leakage of clear fluid from the hypertrichotic region since birth. At that time, magnetic resonance imaging (MRI) had shown syringomyelia at L1 (Panel B, arrowhead), diastematomyelia with bony spurs at L3 and L4 (Panel C, arrow), and tethered cord and low-lying conus medullaris at L5 (Panel B, arrow).
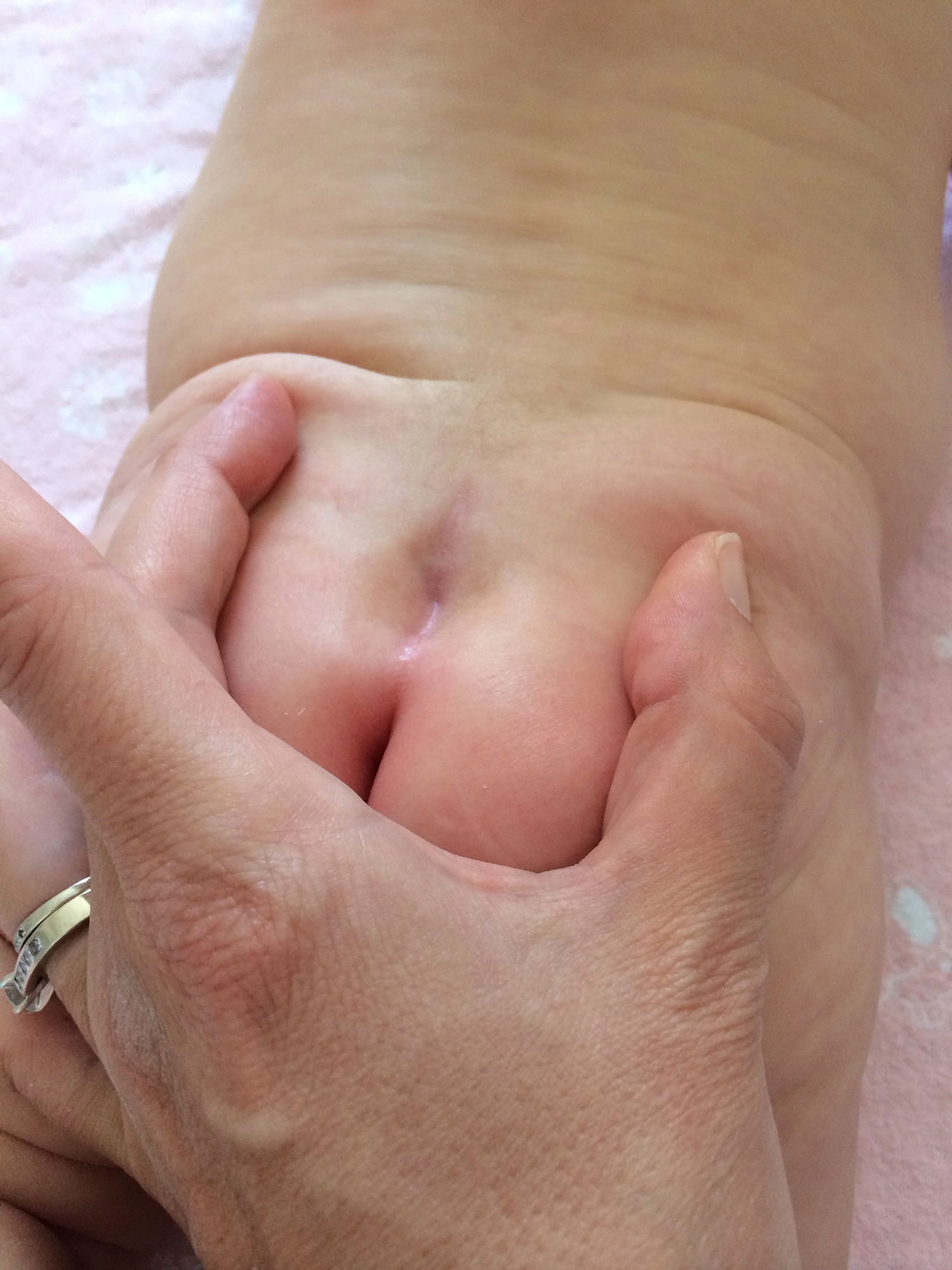
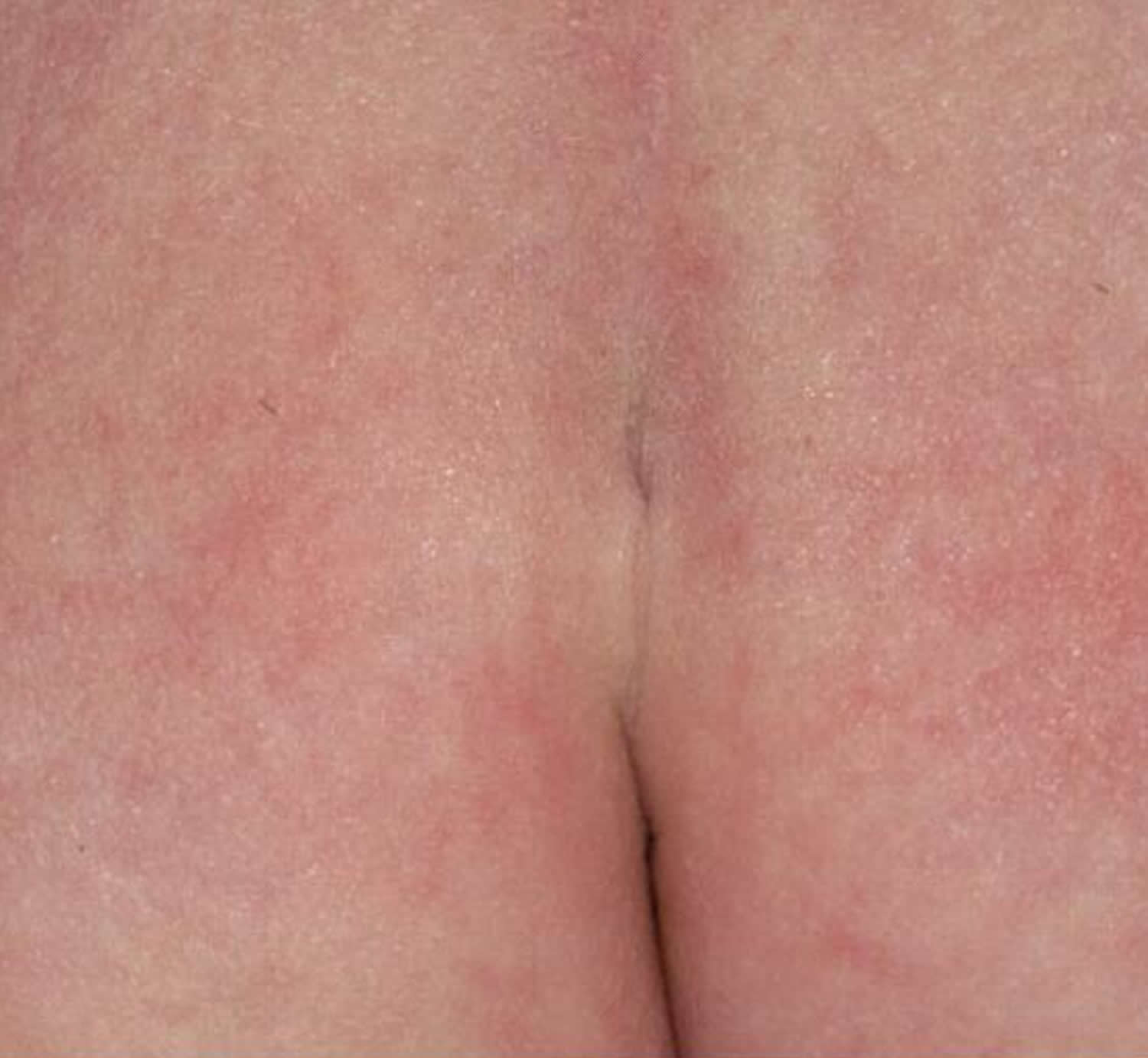
Sacral dimple in adults
Some people are born with small holes or pits near the base of the spine. The risk of significant spinal malformations in asymptomatic, healthy adults with an isolated simple sacral dimple is exceedingly low.
Figure 4. Sacral dimple in adults
Sacral dimple causes
A sacral dimple is a congenital condition, meaning it’s present at birth. There are no known causes.
Sacral dimple complications
Rarely, sacral dimples are associated with a serious underlying abnormality of the spine or spinal cord. Examples include:
- Spina bifida. A very mild form of this condition, called spina bifida occulta, occurs when the spine doesn’t close properly around the spinal cord but the cord remains within the spinal canal. In most cases, spina bifida occulta causes no symptoms.
- Tethered cord syndrome. The spinal cord normally hangs freely within the spinal canal. Tethered cord syndrome is a disorder that occurs when tissue attached to the spinal cord limits its movements. Signs and symptoms may include weakness or numbness in the legs and bladder or bowel incontinence.
The risks of these spinal problems increase if the sacral dimple is accompanied by a nearby tuft of hair, skin tag or certain types of skin discoloration.
Sacral dimple diagnosis
Sacral dimples are present at birth and are evident during an infant’s initial physical exam. In most cases, further testing is unnecessary. If the dimple is very large or is accompanied by a nearby tuft of hair, skin tag or certain types of skin discoloration, your doctor may suggest imaging tests to rule out spinal cord problems.
These tests may include:
- Ultrasound. This noninvasive procedure uses high-frequency sound waves to produce images of structures of the body.
- Magnetic resonance imaging (MRI). If more detail is needed, your doctor may recommend an MRI, which uses radio waves and a strong magnetic field to create cross-sectional images of the body.
Sacral dimple treatment
Treatment is unnecessary for a simple sacral dimple.
References- Spine ultrasounds should not be routinely performed for patients with simple sacral dimples. Acta Paediatr. 2016 Aug;105(8):890-4. doi: 10.1111/apa.13422. Epub 2016 Apr 24. https://onlinelibrary.wiley.com/doi/full/10.1111/apa.13422
- Sacral Dimple. World Neurosurg. 2017 May;101:811.e7-811.e8. doi: 10.1016/j.wneu.2017.02.074. Epub 2017 Feb 27. http://www.worldneurosurgery.org/article/S1878-8750(17)30247-4/fulltext
- Sacral Dimple–the Role and Yield of Imaging. Austin Neurosurg Open Access. 2014;1(4): 1016. http://austinpublishinggroup.com/neurosurgery/fulltext/ajns-v1-id1016.php
- The Enigmatic Sacro-Coccygeal Dimple: To Ignore or Explore? Pediatric Annals. 2014;43(3):95-100https://doi.org/10.3928/00904481-20140221-04
- Mishra SS, Panigrahi S. Thoracic congenital dermal sinus associated with intramedullary spinal dermoid cyst. J Pediatr Neurosci 2014;9:30–2 doi:10.4103/1817-1745.131478 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4040028
- Wang KC, Yang HJ, Oh CW, Kim HJ, Cho BK. Spinal congenital dermal sinus- experience of 5 cases over a period of 10 years. J Korean Med Sci. 1993;8:341–7. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3053715/pdf/8305142.pdf
- Skin Markers of Occult Spinal Dysraphism in Children. Arch Dermatol. 2004;140(9):1109-1115. doi:10.1001/archderm.140.9.1109 https://jamanetwork.com/journals/jamadermatology/fullarticle/480776
- McAtee-Smith JHebert AARapini RPGoldberg NS Skin lesions of spinal axis and spinal dysraphism: fifteen cases and a review of the literature Arch Pediatr Adolesc Med. 1994;148;740-748
- Assaad AMansy AKotb MHafez M Spinal dysraphism: experience with 250 cases operated upon Childs Nerv Syst. 1989;5;324-329
- Radmanesh F, Nejat F, El Khashab M. Dermal sinus tract of the spine. Childs Nerv Syst 2010;26:349–57 doi:10.1007/s00381-009-0962-z https://www.ncbi.nlm.nih.gov/pubmed/19662426
- Mishra SS, Panigrahi S. Thoracic congenital dermal sinus associated with intramedullary spinal dermoid cyst. Journal of Pediatric Neurosciences. 2014;9(1):30-32. doi:10.4103/1817-1745.131478. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4040028/
- Haveri FTTS, Inamadar AC. A Cross-Sectional Prospective Study of Cutaneous Lesions in Newborn. ISRN Dermatology. 2014;2014:360590. doi:10.1155/2014/360590. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3918370/
- Chern JJ, Kirkman JL, Shannon CN, Tubbs RS, Stone JD, Royal SA, et al. Use of lumbar ultrasonography to detect occult spinal dysraphism. J Neurosurg Pediatr. 2012; 9: 274-279. https://www.ncbi.nlm.nih.gov/pubmed/22380955
- McGovern M, Mulligan S, Carney O, Wall D, Moylett E. Ultrasound investigation of sacral dimples and other stigmata of spinal dysraphism. Arch Dis Child. 2013; 98: 784-786. https://www.ncbi.nlm.nih.gov/pubmed/23908189
- Chiu HY, Liao YH. Images in clinical medicine. Occult spinal dysraphism. N Engl J Med. 2014; 370: 466. http://www.nejm.org/doi/full/10.1056/NEJMicm1308004
- Guggisberg D, Hadj-Rabia S, Viney C, Bodemer C, Brunelle F, Zerah M, et al. Skin markers of occult spinal dysraphism in children: a review of 54 cases. Arch Dermatol. 2004; 140: 1109-1115. https://www.ncbi.nlm.nih.gov/pubmed/15381552
- Mete M, Umur AS, Duransoy YK, Barutcuo X011f Lu M, Umur N, Gurgen SG, et al. Congenital Dermal Sinus Tract of the Spine: Experience of 16 Patients. J Child Neurol. 2014. https://www.ncbi.nlm.nih.gov/pubmed/24563472
- Kriss VMKriss TCDesai NSWarf BC Occult spinal dysraphism in the infant: clinical and sonographic review Clin Pediatr (Phila). 1995;34;650-654
- Korsvik HEKeller MS Sonography of occult dysraphism in neonates and infants with MR imaging correlation Radiographics. 1992;12;297-306
- Sasani M, Asghari B, Asghari Y, Afsharian R, Ozer AF. Correlation of cutaneous lesions with clinical radiological and urodynamic findings in the prognosis of underlying spinal dysraphism disorders. Pediatr Neurosurg. 2008; 44: 360-370. https://www.ncbi.nlm.nih.gov/pubmed/18703880
- Chern JJ, Aksut B, Kirkman JL, Shoja MM, Tubbs RS, Royal SA, et al. The accuracy of abnormal lumbar sonography findings in detecting occult spinal dysraphism: a comparison with magnetic resonance imaging. J Neurosurg Pediatr. 2012; 10: 150-153. https://www.ncbi.nlm.nih.gov/pubmed/22747088
- Schropp C, Sörensen N, Collmann H, Krauss J. Cutaneous lesions in occult spinal dysraphism–correlation with intraspinal findings. Childs Nerv Syst. 2006; 22: 125-131. https://www.ncbi.nlm.nih.gov/pubmed/15875199
- Ben-Sira L, Ponger P, Miller E, Beni-Adani L, Constantini S. Low-risk lumbar skin stigmata in infants: the role of ultrasound screening. J Pediatr. 2009; 155: 864-869. https://www.ncbi.nlm.nih.gov/pubmed/19643444