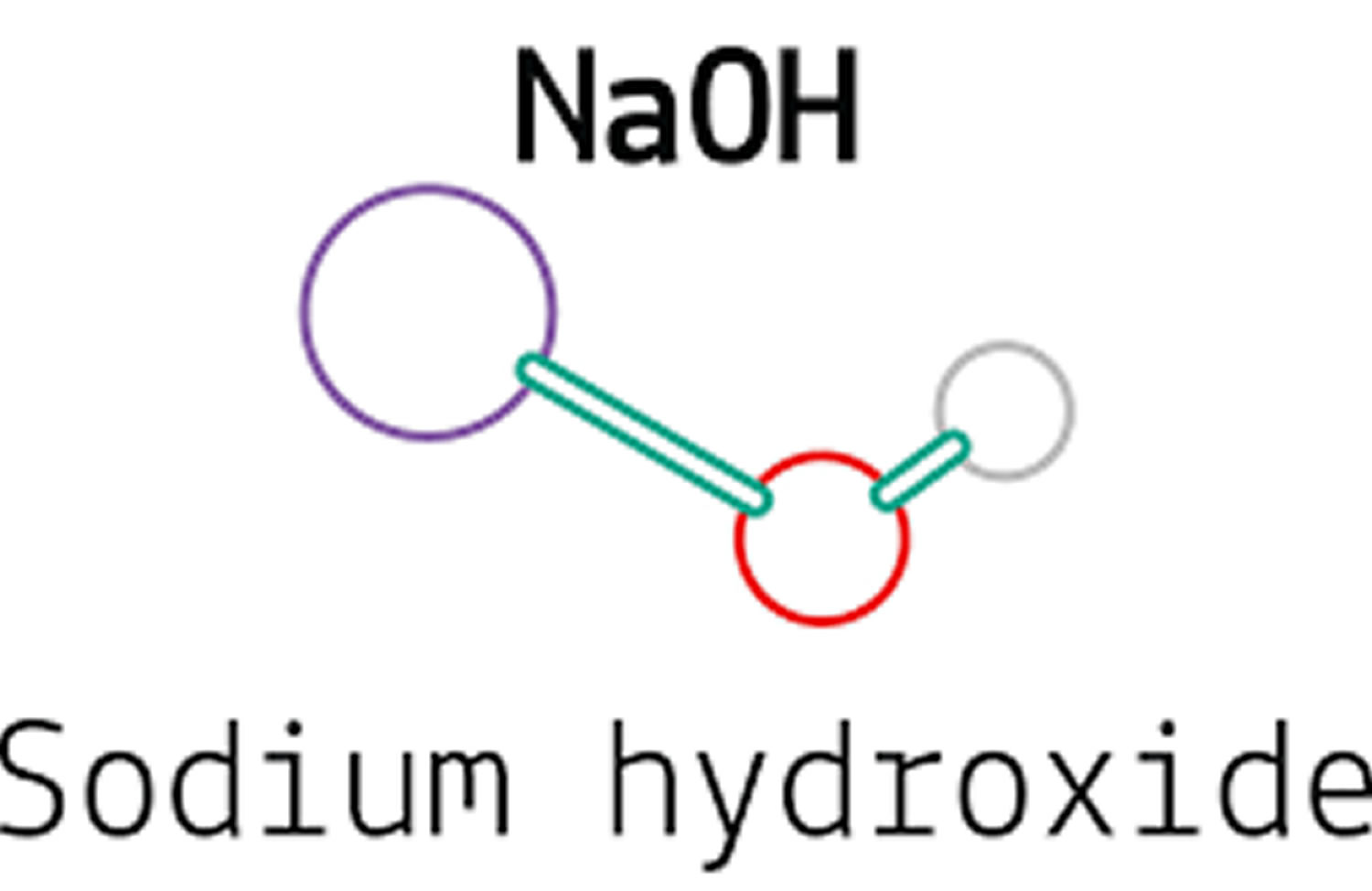
What is sodium hydroxide
Sodium hydroxide [Na(OH)] also known as caustic soda, is a highly caustic substance that is used to neutralize acids and make sodium salts. Sodium hydroxide is added to food (food additive E 524) as an acidity regulator and its function in food is essentially the same as that in food. Sodium hydroxide is approved as a food additive (E 524) 1 for use as an acidity regulator without limitation (quantum satis) in jams, jellies, marmalades, sweetened chestnut purée, other similar fruit and vegetable spreads, processed cereal-based foods and baby foods, other foods for young children, dietary foods for infants for special medical purposes and special formulas for infants. Sodium hydroxide is also approved for use in food additives, food enzymes, food flavourings and nutrients with no limitation (quantum satis) 2. Sodium hydroxide was assessed by the Joint FAO/WHO (Food and Agriculture Organization/World Health Organization) Committee on Food Additives 3 and by the Scientific Committee for Food 4, and both set an acceptable daily intake (ADI) of “not specified”.
Sodium hydroxide is a manufactured substance and sodium hydroxide [Na(OH)] is sold as a solid (cast, flakes, pearls, compounders) or as solutions with varying concentrations. The most important industrial concentration is 50 %. Sodium hydroxide is used for example for cleaning, disinfection, wood treatment and to make soap at home, but many other uses could exist. At room temperature, sodium hydroxide is a white crystalline odorless solid that absorbs moisture from the air. Sodium hydroxide [Na(OH)] solidifies at 20 °C if the concentration is higher than 52 % (by weight), which can be considered the maximum water solubility at 20 °C. When dissolved in water or neutralized with acid it liberates substantial heat, which may be sufficient to ignite combustible materials. Sodium hydroxide is very corrosive. It is generally used as a solid or a 50% solution. Other common names include caustic soda and lye. Sodium hydroxide is used to manufacture soaps, rayon, paper, explosives, dyestuffs, and petroleum products. It is also used in processing cotton fabric, laundering and bleaching, metal cleaning and processing, oxide coating, electroplating, and electrolytic extracting. It is commonly present in commercial drain and oven cleaners.
Sodium hydroxide uses
Sodium hydroxide [Na(OH)] is a strong alkaline substance that dissociates completely in water to sodium (Na+) and hydroxyl (OH–) ions. The dissolution/dissociation in water is strongly exothermic, so a vigorous reaction occurs when sodium hydroxide is added to water.
Sodium hydroxide is used to neutralize acid solutions and make sodium salts. Sodium hydroxide is used in the manufacture of rayon, mercerised cotton, soap, paper, aluminium, petroleum products and in metal cleaning, electrolytic extraction of zinc, tin plating and oxide coating. Mercerisation is a treatment for cellulosic material, typically cotton threads, that strengthens them, gives them a lustrous appearance and increases cotton’s affinity to dye, and resistance to mildew.
Sodium hydroxide solutions hydrolyze fats to form soaps, they precipitate bases and most metals (as hydroxides) from aqueous solutions of their salts.
Sodium hydroxide is a common constituent of many household and industrial cleaners including oven cleaners and beerline cleaners. Sodium hydroxide is present as a stabilizing agent in bleach. It may also be found in dishwasher detergents. Some paint strippers and drain cleaners contain sodium hydroxide. It is used as a pipe line cleaner in dairies, bars and public houses.
Sodium hydroxide has mainly industrial uses. On a global level the main uses are 5:
- Organic chemicals (18 %)
- Pulp and paper (18 %)
- Inorganic chemicals (15 %)
- Soaps, detergents and textile (12 %)
- Alumina (8 %)
- Water treatment (5 %)
- Others (25 %)
Sodium hydroxide is also used by the drink and beer industry to clean non-disposable bottles. Although main quantities are used by the industry (large enterprises) it is also widely used by small and medium sized enterprises. Sodium hydroxideis used for example for disinfection and cleaning purposes. Sodium hydroxide (up to 100 %) is also used by consumers. Sodium hydroxide is used at home for drain and pipe cleaning, wood treatment and it also used to make soap at home 6. Sodium hydroxide is also used in batteries and in oven-cleaner pads. The previously mentioned uses are only examples of uses but probably many other uses do occur because sodium hydroxide is widely available. However, significant differences in uses between countries can be expected.
Small amounts of sodium hydroxide are produced as a by-product and released into the car interior when motor car air bag systems are activated. Air bag systems are triggered when a sensor in the bumper sends an electrical charge to a gas generator containing sodium azide (70 g). A chemical reaction is initiated that produces nitrogen gas from the sodium azide.
Sodium hydroxide is present in Clinitest(R) tablets which are used by diabetics as an in vitro semiquantitive test for glycosuria.
Sodium hydroxide safety
Solid sodium hydroxide [Na(OH)] is corrosive. Depending on the concentration, solutions of sodium hydroxide are non-irritating, irritating or corrosive and they cause direct local effects on the skin, eyes and gastrointestinal tracts. Based on human data concentrations of 0.5-4.0 % were irritating to the skin, while a concentration of 8.0 % was corrosive for the skin of animals 7. Eye irritation data are available for animals. The non-irritant level was 0.2-1.0 %, while the corrosive concentration was 1.2 % or higher 7. A study with human volunteers did not indicate a skin sensitization potential of sodium hydroxide. This is supported by the extensive human experience.
The acute toxicity of sodium hydroxide depends on the physical form (solid or solution), the concentration and dose. Lethality has been reported for animals at oral doses of 240 and 400 mg/kg body weight 7. Fatal ingestion and fatal
dermal exposure has been reported for humans.
No valid animal data are available on repeated dose toxicity studies by oral, dermal, inhalation or by other routes for sodium hydroxide. However, under normal handling and use conditions (non-irritating) neither the concentration of sodium in the blood nor the pH of the blood will be increased and therefore sodium hydroxide is not expected to be systemically available in the body. It can be stated that sodium hydroxide will neither reach the fetus nor reach male and female
reproductive organs, which shows that there is no risk for developmental toxicity and no risk for toxicity to reproduction. Both in vitro and in vivo genetic toxicity tests indicated no evidence for a mutagenic activity.
Based on the available literature, there is a risk for accidental and intentional exposure to solid sodium hydroxide or to irritating or corrosive solutions of sodium hydroxide. Most of the ingestion accidents seem to be related with children and seem to occur at home. Accidental skin and eye exposure seem to be less frequently reported than ingestion in the medical literature. Dust formation is unlikely because of hygroscopic properties. Furthermore sodium hydroxide has a negligible vapor pressure and is rapidly neutralized in air by carbon dioxide and therefore dust and vapor exposure are not expected.
Toxic dose
The severity of sodium hydroxide injury will depend on a number of factors including the concentration of the sodium hydroxide agent, the duration of contact and the volume ingested. It is greatest where the pH is above 12. However, pH is not the only factor which determines the extent to which a substance can cause corrosive injury. Alkaline reserve (which is the amount of a standard acid solution needed to titrate an alkali to a specified pH usually pH 8, the pH of normal esophageal mucosa) has been found to correlate better than pH with the production of caustic esophageal injury 8. Although this method is not currently used (except internally by some manufacturers to classify products as irritant or corrosive) it appears to be a better predictor of injury than pH. Alkaline reserve may also be referred to as the titratable alkaline reserve.
Sodium hydroxide solid preparations and viscous liquids are also more likely to produce severe injury due to prolonged contact. Following ingestion of a small amount the injury is usually limited to the oropharyngeal region and the esophagus. The greater the volume the greater the risk of duodenal and gastric damage.
Several studies have been carried out in an attempt to correlate clinical effects and injury. Gaudreault et al 9 found that signs and/or symptoms do not adequately predict the presence or severity of an esophageal lesion. Crain et al 10 found that the presence of two or more signs or symptoms (vomiting, drooling, stridor) may be a reliable predictor of esophageal injury. In the study by Nuutinen et al 11 prolonged drooling and dysphagia (12-24 hours) were observed to predict esophageal scar formation with 100% sensitivity. In the study of 224 children (aged 0-14 years) by Clausen et al 12 serious complications were due to ingestion of sodium hydroxide or a dishwasher product. Children without any signs or symptoms at the first examination did not develop stricture or epiglottal edema.
The study by Christesen 13 also found that complications only developed in children who had ingested strong alkalis (sodium hydroxide, ammonia and dishwasher products). The author found that children with respiratory symptoms were at greater risk of developing complications and that liquid sodium hydroxide tended to cause more complications than the granular form. It was also concluded that asymptomatic patients are not at risk of complications and probably do not require endoscopy.
Knopp 14 reported that in patients with oral burns approximately one third had significant esophageal injury, whereas 2-15% of patients with esophageal injury had no oral burns.
Clinitest(R) tablets
Ingestion of 1 tablet is sufficient to cause esophageal stricture. However, ingestion of 47 tablets over 1 month by an adult caused only gastritis and eschar formation in the lower two thirds of the stomach with full recovery 15.
Mechanism of toxicity action
Alkalis cause liquifactive necrosis with saponification of fats and solubilisation of proteins. There is a decrease in the collagen content of tissue and saponification of cell membrane lipids and cellular death. They are also hygroscopic and will absorb water from the tissues. These effects result in adherence and deep penetration into the tissues.
Ingestion
Alkalis cause the most severe corrosive effects on the esophagus, rather than the stomach as is the case with acids. However, following deliberate ingestion of a large quantity of an alkali (as with intentional ingestion in adults) both the stomach and small intestine may be involved. This is particularly the case with liquid sodium hydroxide.
Esophageal changes can be divided into 3 stages:
- Acute necrotic phase in which cell death occurs due to coagulation of intracellular protein,
- Intense inflammatory reaction in viable tissues surrounding the necrotic area, thrombosis of vessels occurs,
- Sloughing of superficial necrotic layer 2-4 days later 16.
Strictures form due to an intense fibroblastic reaction and superficial granulation tissue formation that terminates with extensive scar formation and luminal narrowing. The newly formed collagen contracts both cirumferentially and longitudinally resulting in esophageal shortening and stricture formation.
Clinitest(R) tablets
The sodium hydroxide in these tablets reacts with the saliva and liberates heat which can produce a full thickness burn of the oesophagus. Clinitest(R) tablets also adhere to the esophagus either because of thermal coagulation or because of the carbon dioxide bubbles produced from the reaction of the citric acid and sodium hydroxide present 17. Gastric injury may also occur.
Ocular burns
Alkali burns of the eye are very serious because they cause disruption of the protective permeability barriers and rapidly penetrate the cornea and anterior chamber. They combine with cell membrane lipids which causes disruption of the cells and stromal mucopolysaccharides with concomitant tissue softening. Sodium hydroxide can pass freely through the cornea and cause damage to all layers of the cornea and to the anterior segment structures in severe cases 18.
In the acute phase the following occurs: sloughing of the corneal epithelium, necrosis of the cells of the corneal stroma and endothelium, loss of corneal mucoid, edema of the corneal stroma and ciliary processes, ischemic necrosis and edema of the conjunctiva and limbal region of the sclera and infiltration of inflammatory cells into the cornea and iris. Corneal infiltration and degeneration occurs 1-3 weeks after injury 19.
Alkalis cause rapid loss of corneal mucoprotein. They also bind to corneal mucoprotein and collagen and the eye may remain alkaline despite prolonged irrigation due to slow dissociation of hydroxyl ions from corneal proteins. A large number of animal experiments, particularly on rabbits, have been conducted to study alkali injury to the eye and it is known from these experiments that in the rabbit eye the pH of the aqueous humor can rise to 10-11 within a few minutes or even higher in severe cases. The pH slowly falls over a period of hours, except in extreme cases 19.
Injury of the endothelium causes failure of the endothelial pump which normally keeps the cornea hydrated and clear. This failure causes the corneal stroma to become edematous and susceptible to vascularisation and scarring.
When the protective sheath around collagen is damaged, collagenases produced by polymorphonuclear leucocytes infiltrate the damaged area and degrade the corneal collagen causing melting or ulceration of the stroma, formation of descemoteceles (herniation of Descemet’s membrane) and perforation of the cornea. This phase is evident 3-7 days post-injury.
The role of injury to blood vessels remains unclear, as with other chemical burns to the eye the blood vessels of the conjunctiva and episclera are seen to be thrombosed immediately after exposure.
Both alkali and acid burns cause a transient rise in intraocular pressure due to shrinkage of the eye coats. A second phase of raised intraocular pressure may occur due to prostaglandin release and a still later phase with glaucoma due to obstruction of aqueous outflow caused by inflammation or synechiae.
In severely damaged untreated burns inflammatory destruction and ulceration continues to occur for weeks or months resulting in damage ranging from dense vascular invasion to opaque scarring and corneal perforation.
During the recovery process, surviving keratocytes begin to form new collagen but a decrease in available ascorbate (caused by injury to the main source in the ciliary body) is a limiting factor in this process. This lack of ascorbate may also render the cornea more susceptible to attack by oxygen free radicals.
Sodium hydroxide dangers
The major human health hazard (and the mode of action) of sodium hydroxide is local irritation and/or corrosion on skin and eye irritation/corrosion.
Human poisoning cases indicate that a dose of 10 grams orally is fatal 20. Sodium hydroxide is toxic by oral ingestion 20. Sodium hydroxide is corrosive to all tissues. Concentrated vapors lead to serious damage to the eyes and respiratory system. Oral ingestion of sodium hydroxide, which occurs frequently in children, causes severe tissue necrosis, with stricture formation of the esophagus, often resulting in death 20. Contact with the skin may result in contact dermatitis, hair loss, as well as necrosis due to severe irritation 20. Increased incidence of esophageal carcinoma after severe intoxication with sodium hydroxide has been reported in man 20. In animal studies, long-term dermal contact with substances leading to pH changes in the skin causes the development of tumors, as a result of severe tissue irritation and reparative cell growth 20. Mutagenic for mammalian somatic cells. May cause damage to the following organs: mucous membranes, upper respiratory tract, skin, eyes 20. Tumors are not to be expected if the effects of irritation are prevented. To date, there are no relevant studies of the prenatal toxic effects of sodium hydroxide 20.
Immediate first aid: Remove patient from contact with the material. Ensure that adequate decontamination has been carried out. If patient is not breathing, start artificial respiration, preferably with a demand valve resuscitator, bag-valve-mask device, or pocket mask, as trained. Perform CPR if necessary. Immediately flush contaminated eyes with gently flowing water. Do not induce vomiting. If vomiting occurs, lean patient forward or place on the left side (head-down position, if possible) to maintain an open airway and prevent aspiration. Keep patient quiet and maintain normal body temperature. Obtain medical attention.
- If sodium hydroxide contacts the eyes, immediately wash (irrigate) the eyes with large amounts of water, occasionally lifting the lower and upper lids. Get medical attention immediately.
- If sodium hydroxide contacts the skin, immediately flush the skin with large amounts of water. Get medical attention immediately. If this chemical (or liquids containing this chemical) contacts the skin, promptly wash the contaminated skin with soap and water. If this chemical or liquids containing this chemical penetrate the clothing, immediately remove the clothing and wash the skin with soap and water. If irritation persists after washing, get medical attention.
- If a person breathes large amounts of this chemical, move the exposed person to fresh air at once. If breathing has stopped, perform artificial respiration. Keep the affected person warm and at rest. Get medical attention as soon as possible.
- If this chemical has been swallowed, get medical attention immediately.
Sodium hydroxide ingestion
In studies using rabbits, instillation by oral intubation caused within 10 seconds: erosion into the stomach muscle with 12% solutions; perforation with 28% solutions; and no damage with 1% solutions. According to Schober et al. 21 between January 1976 and October 1988 a total number of 6 cases of ingestion of sodium hydroxide was reported by the Children Surgery Department (University of Graz, Austria). The University Hospital of Santiago de Compostela (Spain) reported about 67 cases of accidental ingestion of sodium hydroxide by children between 1981 and 1990 22. Most of the accidents occurred at home and the container was located within easy reach of the children. A nationwide survey of ingestion of corrosives has been performed for the period 1984-1988 in Denmark 23. It revealed 57 admissions to hospital of children (0-14 years) due to sodium hydroxide ingestion. The authors were confident that all children with serious complications after ingestion of corrosives were included in the study.
All previously mentioned publications reported accidental ingestion of sodium hydroxide by children. Wijburg et al. 24 reviewed the records of 170 patients admitted to the Department of Otolaryngology of the University Hospital of Amsterdam in the period January 1, 1971 to December 31, 1981 with suspected caustic ingestion. Of these 170 patients about 15 patients had ingested sodium hydroxide.
A 14 year old boy took a sodium hydroxide solution (30%) in to his mouth 25. He immediately spat it out. He drank some milk and water and vomited. On arrival about 30 minutes later he had retrosternal pain and had difficulty swallowing. He was given antibiotics and steroids. Esophagoscopy was performed two days later and revealed mucosal lesions in the upper esophagus. He began to improve and was able to take mashed food orally. He then began to develop difficulty in swallowing and a X-ray on day 23 revealed a stricture at the level of the carina of trachea. On the 38th day esophagoscopy with dilatation of the stricture was performed. About 2 hour later he suffered immediate retrosternal pain. An X-ray showed perforation of the stricture. This was sewn up via a left side thoracotomy. Serious inflammatory changes were observed with mediastinal emphysema and a purulent pleuritis. A nasogastric tube and three drains were left in place. On the 44th day after ingestion profuse bleeding was observed through the nasogastric tube and drains were noted. He became shocked and the decision was made to operate. He suffered a cardiac arrest while general anaesthetic was being given. A right side thoracotomy showed a 4-5 mm rupture of the descending part of the aorta with bleeding into the left pleura. After cardiac massage, blood transfusion and repair of the rupture he stabilized. Part of the esophagus was removed due to inflammation. On day 52 another hemorrhage occurred. He was operated on again and the hemorrhage was seen to arise from the aortic rupture. The aorta wall was fragile and could not be repaired. The patient died on the operating table. A purulent mediastinitis, bilateral purulent pleuritis, lung atalectasis and pericarditis were observed at postmortem 25.
A 16 month old female refused to drink and began drooling after ingesting the residue of a sodium hydroxide solution which the mother had been using for cleaning 25. She vomited several times with the vomitus containing a small amount of blood. The pharynx was red and there was slight bleeding of the upper gums. The chest was initially clear but 90 minutes after admission inspiratory and expiratory wheezes were present and a chest X-ray suggested aspiration pneumonia. At 15 hours post-ingestion laryngoscopy and esophagoscopy were performed. The false cords and epiglottis were found to be red and edematous. The cricopharyngeus was ulcerated and bleeding. The esophageal mucosa was bleeding and circumferential second and third degree burns were present. The child required intubation and ventilation and was started on methylprednisolone and ampicillin. Ventilatory support was necessary for three weeks. Subsequent laryngoscopy revealed laryngeal edema and burns which resulted in laryngeal stenosis. An esophagoscopy at five weeks post-ingestion revealed esophageal narrowing. A barium swallow showed multiple esophageal strictures and hypoperistalsis of the proximal segment of the esophagus. The child required nine esophageal dilatations, and was eventually able to take oral feedings. She was discharged one year after the ingestion 25.
9 cases of liquid sodium hydroxide ingestion which resulted in esophageal and gastric injury 26. One person who ingested 10 g sodium hydroxide in water suffered transmural necrosis of the esophagus and stomach and died 3 days after admission to the hospital.
200 patients with suspected caustic ingestation were examined 27. No steroids were administered to the patients involved. Lesions in the esophagus were found in 93 patients. Thirty-two patients with deep circular burns had nasogastric tubes inserted immediately. Of these patients, 2 developed esophageal strictures, but subsequent dilatation was successful. No stricture formation was observed in the group of patients with noncircular lesions. This low percentage of stricture formation is due to the use of nasogastric tubes. Since neither the presence nor the severity of esophageal burns is predictable, an endoscopy should be performed in all suspected cases. In the absence of severe pharyngeal lesions, the use of a flexible fiberoptic endoscope is preferable because it also allows examination of the stomach and proximal part of the duodenum 27.
An experimental study was conducted to investigate the effects of erythropoietin (EOP) on the acute phase of esophageal burn damage induced by sodium hydroxide. A standard esophageal alkaline burn was produced by the application of 10% sodium hydroxide to the distal esophagus in an in vivo rat model 28. Fifty-six female rats were allocated into three groups: Group uninjured and untreated (baseline control, n = 8) rats were uninjured and untreated, Group sodium hydroxide (positive control, n = 24) rats were injured but untreated and Group EPO (erythropoietin-treated, n = 24) rats were injured and given subcutaneous erythropoietin (1,000 IU/kg per day), 15 min, 24, and 48 hr after administration of the sodium hydroxide solution. Six animals from Group sodium hydroxide and six from Group erythropoietin-treated were killed at 4, 24, 48, and 72 hours after application of sodium hydroxide to the esophagus. All of animals in Group uninjured and untreated were killed 4 hour after exposure to 0.9% NaCl (sodium chloride or saline). Oxidative damage was assessed by measuring levels of malondialdehyde and nitric oxide (NO), and activities of superoxide dismutase and catalase in homogenized samples of esophageal tissue. Histologic damage to esophageal tissue was scored by a single pathologist blind to groups. Malondialdehyde levels in the uninjured and untreated and erythropoietin-treated groups were significantly lower than those in the sodium hydroxide but untreated group. Superoxide dismutase and catalase activities, and nitric oxide (NO) levels in the uninjured and untreated and erythropoietin-treated groups were significantly higher than in the sodium hydroxide but untreated group. Esophageal tissue damage measured at 4, 24, 48, and 72 hours after NaOH application was significantly less in the erythropoietin-treated group than in the sodium hydroxide but untreated group. When administered early after an esophageal burn induced by 10% sodium hydroxide in this rat model, erythropoietin significantly attenuated oxidative damage, as measured by biochemical markers and histologic scoring 28.
Skin
When sodium hydroxide (caustic soda) comes into contact with the skin it does not usually cause immediate pain, but it does start to cause immediate damage. It fails to coagulate protein which would serve to prevent further penetration. Thus, upon contact with eyes, washing with water must be started within 10 seconds and continued for at least 15 minutes to prevent permanent injury. Following contact with skin, washing with water must be started immediately to prevent corrosive chemical burns.
A human skin irritation test with 0.5 % sodium hydroxide was performed using exposure periods of 15, 30 and 60 min. The treatment sites were assessed 24, 48 and 72 hours after patch removal. The results showed that after a maximum exposure of 60 min, 61 % of the volunteers (20 of 33) showed a positive skin irritation reaction 29. Sodium hydroxide skin exposure causes redness, pain, serious skin burns and blisters. Sodium hydroxide liquid causes second or third degree burns after short contact. In studies using rabbits, application to the skin of a 5% solution caused severe necrosis after 4 hours. Solutions > 30% are highly corrosive to skin.
A 20 year old patient presented 2 hours after accidentally spraying herself in the face with an oven cleaner containing 4% sodium hydroxide 25. She had removed the excess liquid but did not irrigate the area. She did not experience any pain until nearly 2 hours later. On examination she was in moderate distress with no ocular involvement. The right side of her face was erythematous and blistering in a serpiginous pattern extending from the infraorbital rim to the body and angle of her mandible. The area of the right cheek had a bronze discoloration. The body surface area involved was about 2%. The area was irrigated for 60 minutes. Despite this the burn continued to show signs of third degree burn involvement. She was transferred to a burns unit and underwent surgical debridement and skin graft. Follow up six weeks later revealed good healing and no complications.
Eyes
A 31 year man had sodium hydroxide blown into his amblyopic left eye after an explosion caused by placing solid sodium hydroxide into a plugged drain 25. He washed the eye immediately in a shower and arrived at hospital within 5 minutes. On examination the cornea was opaque and the lower two thirds of the conjunctiva were ischemic. Topical irrigation was repeated and he was transferred to the operating room where intraocular irrigation was commenced. About 100-120 mL of Ringer’s solution was used in this procedure over 90 minutes. At this time the cornea was slightly clearer. Methylprednisolone was given by retrobulbar and subconjunctival injection. Continuous slow topical irrigation was continued for a further 24 hours. On the first post-operative day visual acuity was present to light, intraocular pressure was high and a cataract was present. Topical antibiotics, systemic and topical corticosteroids and carbonic anhydrase inhibitors were given. Two weeks after the injury aspiration-irrigation of the cataract was undertaken with an improvement in visual acuity. Acetylcysteine drops were used and a soft contact lens was put in place. He was discharged three weeks after the injury but returned three days later with severe pain, hypopyon and hyphema. The cornea ulcerated and perforated 27 days after the injury. The perforation was repaired with a corneoscleral free hand graft. Despite the presence of light perception the eye was enucleated at the patient’s request 70 days after the injury 25.
A 28-year old member of an oil-well drilling crew sustained extensive splash burns of the left eye from sodium hydroxide and received emergency care from a general physician prior to being hospitalized 30. At the hospital, initial examination showed vision limited to light perception, corneal clouding to such an extent that iris markings were not discernable, necrosis of most of the bulbar conjunctiva, some sloughing in the nasal area of the cornea, blanched and necrotic cul-de-sac, and some involvement of the lids and adjacent skin. The treatment of the patient at the hospital consisted of daily debridement of necrotic areas, local atropine, antibiotics, steroids, systematic ACTH, vitamins, antacids, and proteolytic enzymes. The treatment produced some improvement with time so that usual, late sequelae such as vascular invasion and symblepharon did not occur, and the cornea cleared sufficiently within 7 weeks that vision returned to near normal 30.
A total of 23 burns of the eye due to sodium hydroxide or potassium hydroxide were admitted to the eye clinic of the RWTH Aachen in Germany from 1985 to 1992 31. In 17 cases the accident happened during work, while 6 cases occurred at home using sodium hydroxide/potassium hydroxide as drain cleaner. The alkali burns were of special interest because of the rapid and deep penetration of alkali into the ocular tissues.
From January 1984 to June 1991 a total number of 24 patients were treated for sodium hydroxide related eye injury in the Department of Ophthalmology, Postgraduate Institute of Medical Education and Research, Chandigarh, India. Over half of the patients which had ocular chemical burns were young people working in laboratories and factories.
Inhalation
For production and major uses of sodium hydroxide aerosols do normally not occur. However, for certain specific uses, e.g. cleaning ovens and disinfection of sheds, the formation of aerosols can not be excluded completely. For example the cleaning of ovens could result in an irritation of the throat due to the presence of sodium hydroxide in the air. However, it should be realized that aerosols of sodium hydroxide are not stable. They are rapidly transformed due to an uptake of carbon dioxide from the atmosphere resulting in the formation of sodium bicarbonate and sodium carbonate. The transformation of respirable sodium hydroxide aerosols into sodium carbonate aerosols can occur in seconds 32. Analytical measurements, to determine the inhalation exposure of workers during production and use, seem to be unavailable. However, in animal studies, inhalation of the aerosol can cause pulmonary edema.
The inhalation of aerosols of 5 % sodium hydroxide by a 25-year-old women resulted in irreversible obstructive lung injury after working for one day in a poorly ventilated room 26.
Sodium hydroxide toxicokinetics, metabolism and distribution
Sodium is a normal constituent of your blood and an excess is excreted in your urine. A significant amount of sodium is taken up via the food because the normal uptake of sodium via food is 3.1-6.0g per day. Exposure to sodium hydroxide could potentially increase the pH of the blood. However, the pH of the blood is regulated between narrow ranges to maintain homeostasis. Via urinary excretion of bicarbonate and via exhalation of carbon dioxide the pH is maintained at the normal pH of 7.4-7.5.
When humans are dermally (skin) exposed to low (non-irritating) concentrations, the uptake of sodium hydroxide should be relatively low due to the low absorption of ions. For this reason the uptake of sodium hydroxide is expected to be limited under normal handling and use conditions. Under these conditions the uptake of hydroxyl (OH–) ion, via exposure to sodium hydroxide, is not expected to change the pH in the blood. Furthermore the uptake of sodium, via exposure to sodium hydroxide, is much less than the uptake of sodium via food under these conditions. For this reason sodium hydroxide is not expected to be systemically available in the body under normal handling and use conditions.
An example will be given for an inhalation exposure scenario. Assume an exposure to an sodium hydroxide concentration of 2 mg/m³, which is the threshold limit value (TLV) in the USA, and a respiratory volume of 10 m3 per day. In this case the daily exposure is 20 mg sodium hydroxide. The threshold limit value (TLV) of a chemical substance is believed to be a level to which a worker can be exposed day after day for a working lifetime without adverse effects.
The amount of 20 mg sodium hydroxide is equivalent with 11.5 mg sodium which is a negligible amount compared to the daily dietary exposure of 3.1-6.0 g. The amount of 20 mg sodium hydroxide is equivalent with 0.5 mmole and if this amount would be taken up in the blood stream it would result in a concentration of 0.1 mM hydroxyl (OH–) ion (assuming 5 liter blood per human). This is a negligible amount when it is compared with the bicarbonate concentration of 24 mM of blood. This example confirms that sodium hydroxide is not expected to be systemically available in the body under normal handling and use conditions.
References- Commission Regulation (EU) No 1129/2011 of 11 November 2011 amending Annex II to Regulation (EC) No 1333/2008 of the European Parliament and of the Council by establishing a Union list of food additives. OJ L 295, 12.11.2011, p. 178.
- Commission Regulation (EU) No 1130/2011 amending Annex III to Regulation (EC) No 1333/2008 of the European Parliament and of the Council on food additives by establishing a Union list of food additives approved for use in food additives, food enzymes, food flavourings and nutrients.
- Joint FAO/WHO Expert Committee On Food Additives (JECFA), 1966. Specifications for the identity and purity of food additives and their toxicological evaluation: some antimicrobials, antioxidants, emulsifiers, stabilisers, flour-treatment agents, acids, and bases. Ninth Report of the Joint FAO/WHO Expert Committee On Food Additives. Rome, 13–20 December 1966.
- EC (European Commission), 1991, online. Opinion of the Scientific Committee for Food: First Series of Food Additives of Various Technological Functions. https://ec.europa.eu/info/departments/health-and-food-safety_en
- CMAI (2000), Fifteenth Annual World Petrochemical Conference, March 29 & 30, 2000, Houston, Texas, USA
- Keskin E et al. (1991), Eur J Pediatr Surg, 1, 335-338
- Sodium hydroxide. http://www.inchem.org/documents/sids/sids/NAHYDROX.pdf
- Hoffman RS, Howland MA, Kamerow HN and Goldfrank LR. 1989. Comparison of titratable acid/alkaline reserve and pH in potentially caustic household products. Clin Toxicol 27 (4&5):24261
- Gaudreault P, Parent M, McGuigan MA, Chicoine L and Lovejoy FH. 1983. Predicability of eosophageal injury from signs and symptoms: a study of caustic ingestions in 378 children. Pediatrics 71 (5):767-770
- Crain EE, Gershel JC and Mezey AP. 1984. Caustic ingestions. Symptoms as predictors of esophageal injury. Am J Dis Child 138:863-865
- Nuutinen M, Uhari M, Karvali T and Kouvalainen K. 1994. Consequences of caustic ingestions in children. Acta Paediatr 83:1200-1205
- Clausen JO, Nielsen TLF and Fogh A. 1994. Admission to Danish hospitals after suspected ingestion of corrosives. A nationwide survey (1984-1988) comprising children aged 0-14 years. Dan Med Bull 41:234-237
- Christesen HBT. 1995. Prediction of complications following unintentional caustic ingestion in children. Is endoscopy always necessary? Acta Paediatr 84:1177-1182
- Knopp R. 1979. Caustic ingestions. JACEP 8:329-336
- Mallory A and Schaefer JW. 1977. Clinitest ingestion. Br Med J 2:105-107
- Adam JS and Birck HG. 1982. Pediatric caustic ingestion. Ann Otol Rhinol Laryngol 91:656-658
- Burrington JD. 1975. Clinitest burns of the esophagus. Ann Thorac Surg 20 (4):400-404
- Wright P. 1982. The chemically injured eye. Trans Ophthal Soc UK 102:85-87
- Grant WM and Schuman JS. 1993. Toxicology of the eye 4th ed. Charles C Thomas, Springfield Illinois
- Sodium hydroxide. https://pubchem.ncbi.nlm.nih.gov/compound/14798
- Schober PH et al. (1989), Wiener Klin Wschr, 101, 318-322
- Casasnovas et al. (1997), Eur J Pediatr, 156, 410-414
- Clausen JO et al. (1994), Danish Medicinal Bulletin 41, 234-237
- Wijburg FA et al. (1985), Ann Otol Rhinol Laryngol, 94, 337-341
- IPCS; UK Poisons Information Document: Sodium Hydroxide (1310-73-2). Available from, as of October 5, 2011: http://www.inchem.org/documents/ukpids/ukpids/ukpid26.htm
- Organization for Economic Cooperation and Development; Screening Information Data Set for Sodium Hydroxide, (1310-73-2) p.13 (March 2002). Available from, as of October 4, 2011: http://www.inchem.org/pages/sids.html
- Wijburg FA et al; Ann Otol Rhinol Laryngol 94 (4 Part 1): 337-41; 1985. https://www.ncbi.nlm.nih.gov/pubmed/4026118
- The protective effect of erythropoietin on the acute phase of corrosive esophageal burns in a rat model. Pediatr Surg Int. 2010 Feb;26(2):195-201. doi: 10.1007/s00383-009-2480-1. Epub 2009 Sep 16. https://www.ncbi.nlm.nih.gov/pubmed/19760200
- Organization for Economic Cooperation and Development; Screening Information Data Set for Sodium Hydroxide, (1310-73-2) p.14, March 2002
- Horowitz ID; Am J Ophthalmol 61: 340-341 (1966) as cited in NIOSH; Criteria Document: Sodium Hydroxide p.29 (1975) DHEW Pub. NIOSH 76-105
- Kuckelkorn et al. (1993), Klin Monatsbl Augenheilkd, 203, 397-402
- Cooper et al. (1979), American Industrial Hygiene Association Journal, 40, 365-371