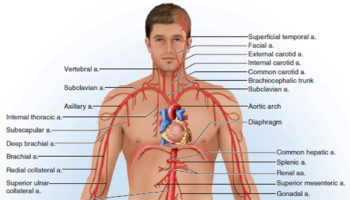
Subclavian artery
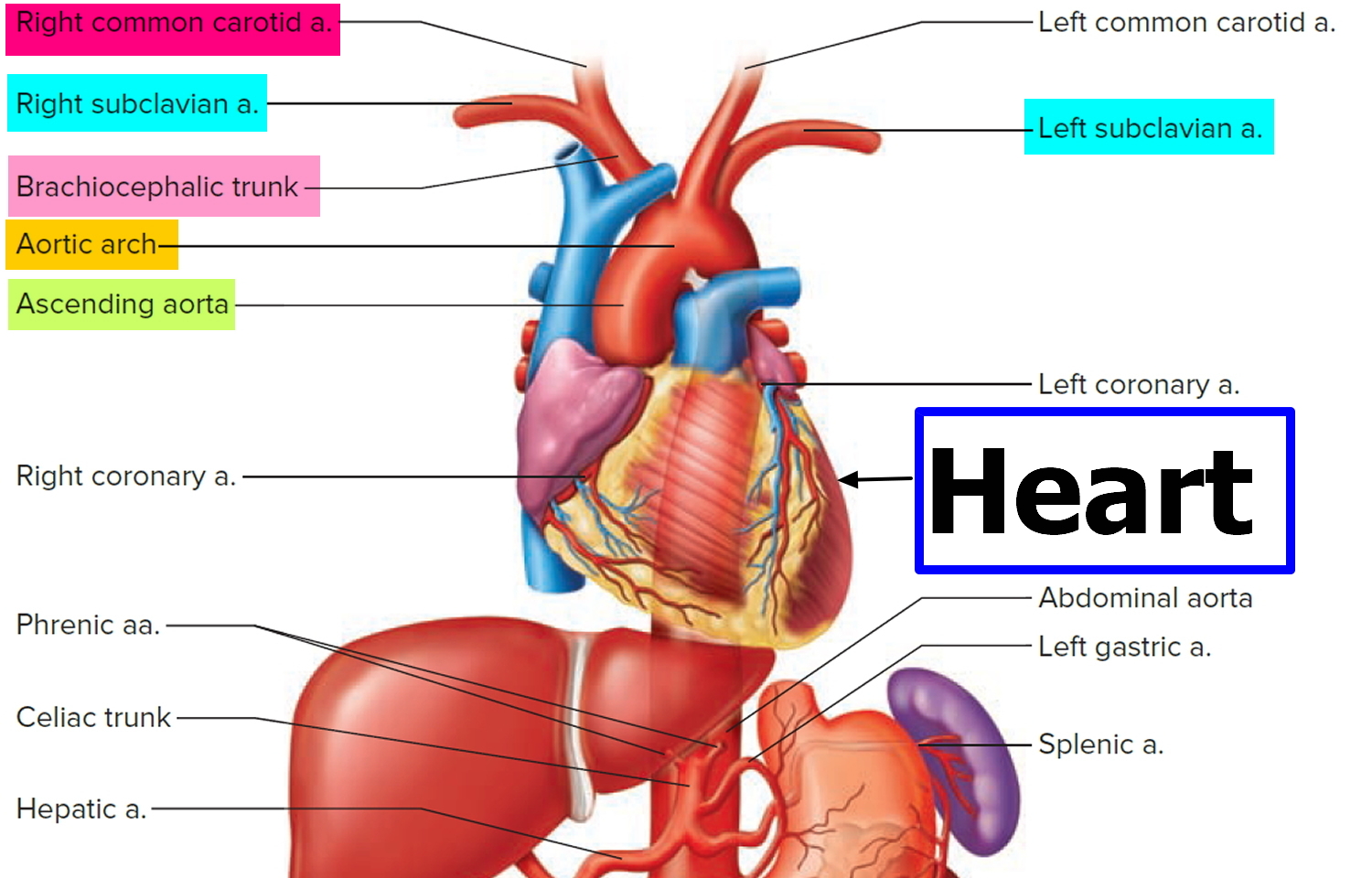
Three large arteries arise from the aortic arch to collectively supply the head, neck, shoulder, and upper limbs: the brachiocephalic trunk, the left common carotid artery, and the left subclavian artery.
The brachiocephalic trunk (brachiocephalic artery) gives rise to the right subclavian artery and the right common carotid artery.
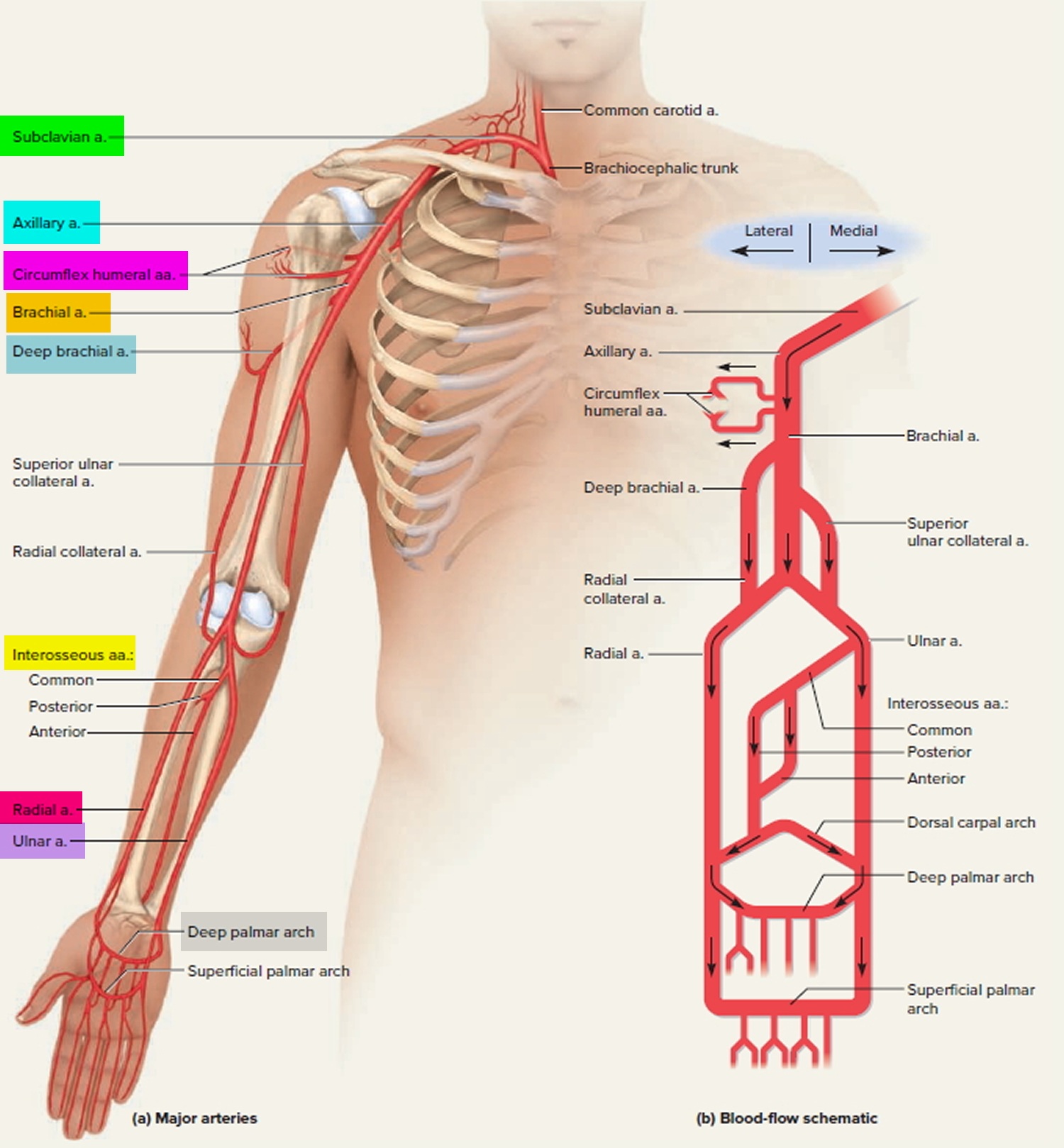
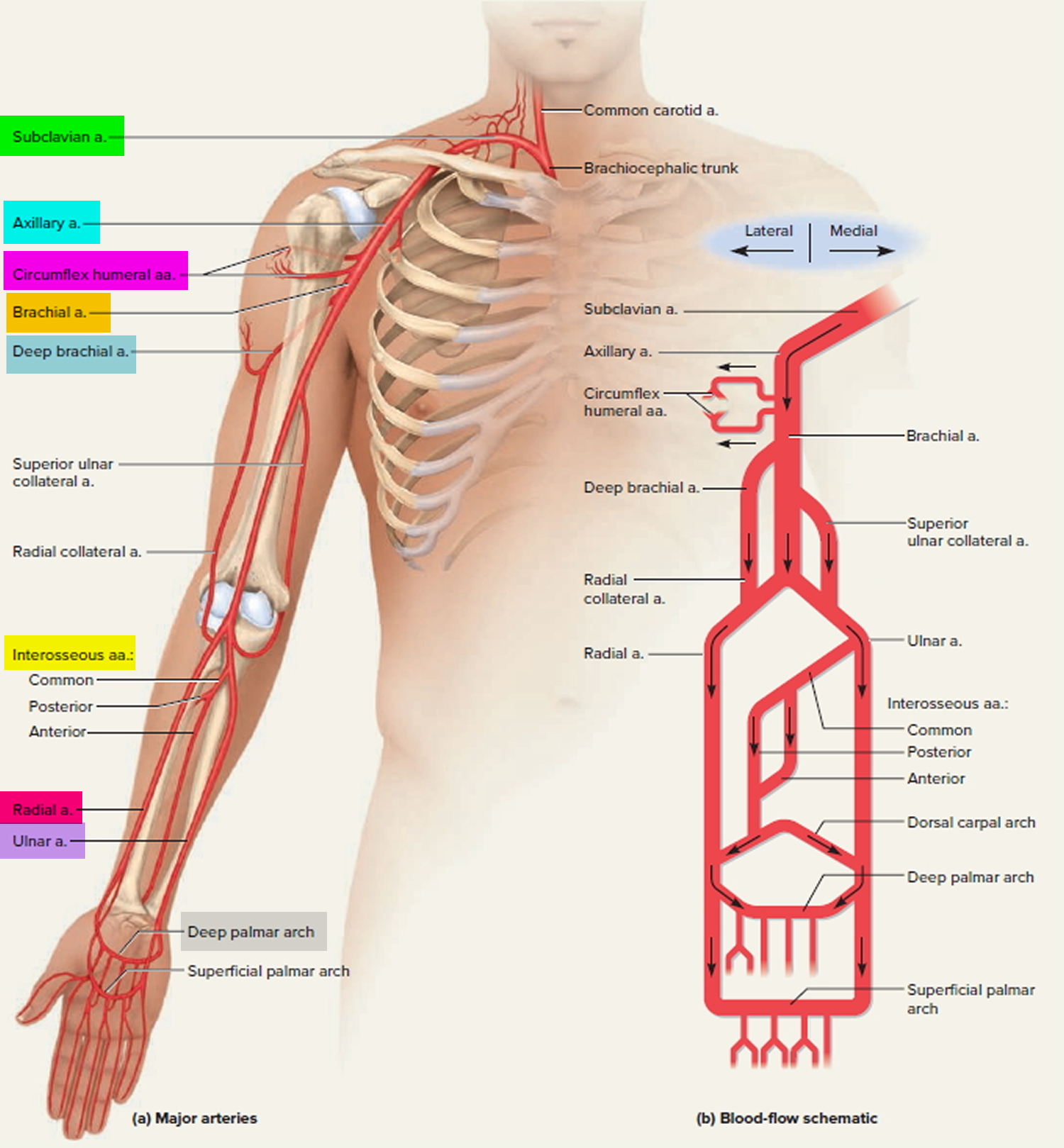
Each subclavian artery arches over the respective lung, rising as high as the base of the neck slightly superior to the clavicle. It then passes posterior to the clavicle, downward over the first rib, and ends in name only at the first rib’s lateral margin. In the shoulder, it gives off several small branches to the thoracic wall and viscera.
As the subclavian artery continues past the first rib, it is named the axillary artery. It continues through the axillary region, gives off small thoracic branches and ends, again in name only, at the neck of the humerus. Here, it gives off a pair of circumflex humeral arteries, which encircle the humerus, anastomose with each other laterally, and supply blood to the shoulder joint and deltoid muscle. Beyond this loop, the vessel is called the brachial artery.
The brachial artery continues down the medial and anterior sides of the humerus and ends just distal to the elbow, supplying the anterior flexor muscles of the brachium along the way. This artery is the most common site of blood pressure measurement with the sphygmomanometer. The deep brachial artery arises from the proximal end of the brachial and supplies the humerus and triceps brachii muscle.
About midway down the arm, the deep brachial artery continues as the radial collateral artery. The radial collateral artery descends in the lateral side of the arm and empties into the radial artery slightly distal to the elbow.
The superior ulnar collateral artery arises about midway along the brachial artery and descends in the medial side of the arm. It empties into the ulnar artery slightly distal to the elbow.
Just distal to the elbow, the brachial artery forks into the radial and ulnar arteries. The radial artery descends the forearm laterally, alongside the radius, nourishing the lateral forearm muscles. The most common place to take a pulse is at the radial artery just proximal to the thumb.
The ulnar artery descends medially through the forearm, alongside the ulna, nourishing the medial forearm muscles.
The interosseous arteries of the forearm lie between the radius and ulna. They begin with a short common interosseous artery branching from the upper end of the ulnar artery. The common interosseous quickly divides into anterior and posterior branches. The anterior interosseous artery travels down the anterior side of the interosseous membrane, nourishing the radius, ulna, and deep flexor muscles. It ends distally by passing through the interosseous membrane to join the posterior interosseous artery. The posterior interosseous artery descends along the posterior side of the interosseous membrane and nourishes mainly the superficial extensor muscles.
Two U-shaped palmar arches arise by anastomosis of the radial and ulnar arteries at the wrist. The deep palmar arch is fed mainly by the radial artery and the superficial palmar arch mainly by the ulnar artery. The arches issue arteries to the palmar region and fingers.
Figure 1. Origins of Left and Right subclavian arteries
Figure 2. Left Subclavian artery directly coming off the Aortic arch
Figure 3. Branches of subclavian artery to the shoulder and upper limb
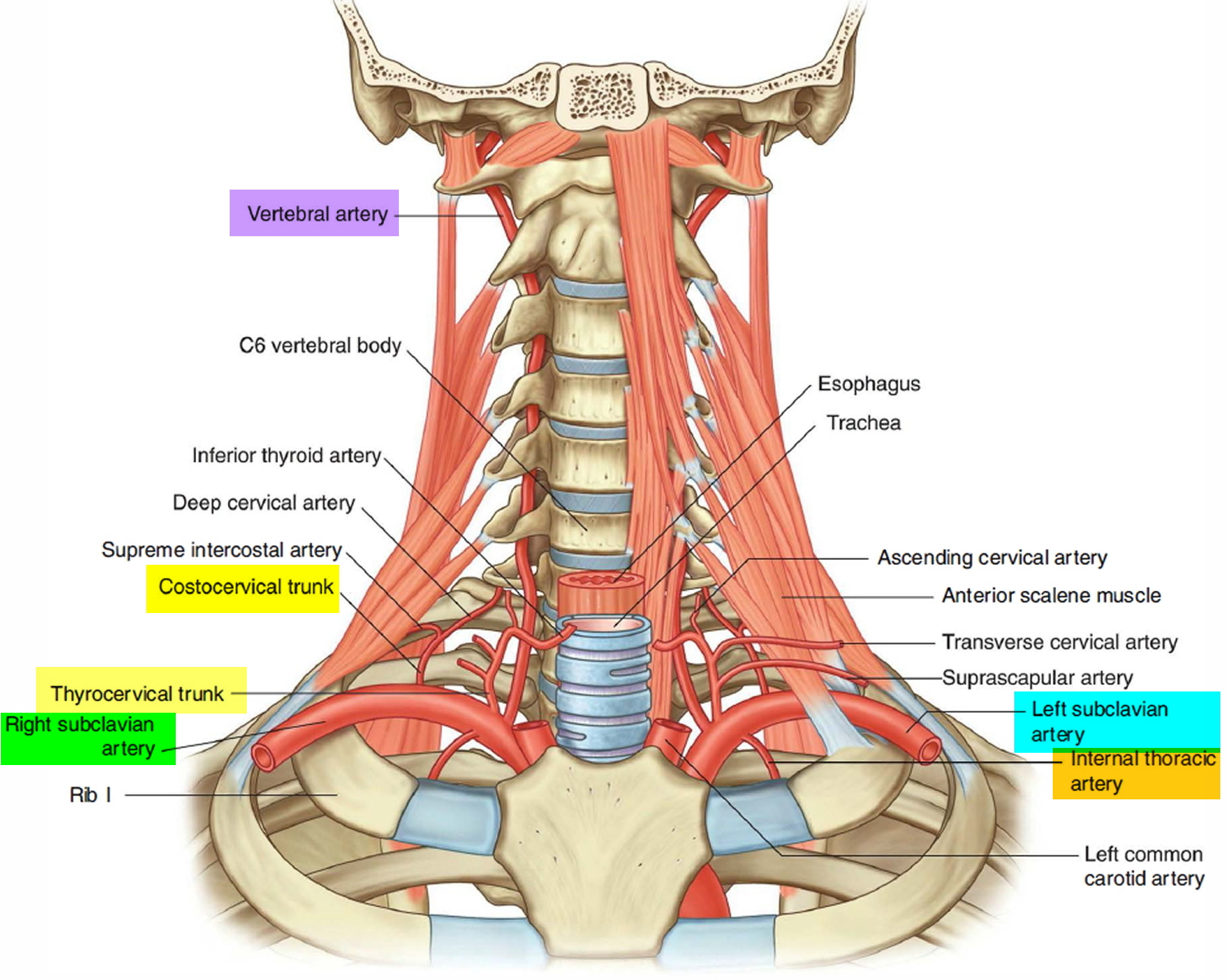
Branches of the Subclavian artery in the head and neck
All branches from the right and left subclavian arteries in the head and neck arise from the first part of the artery, except in the case of one branch (the costocervical trunk) on the right side (Figure 4). The 4 majors branches include:
- the Vertebral artery,
- the Thyrocervical trunk,
- the Internal thoracic artery, and
- the Costocervical trunk.
Figure 4. Branches of subclavian artery in the neck and head
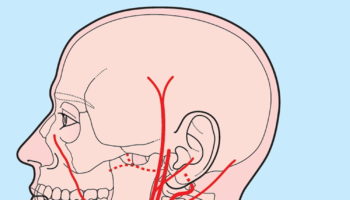
Vertebral Artery
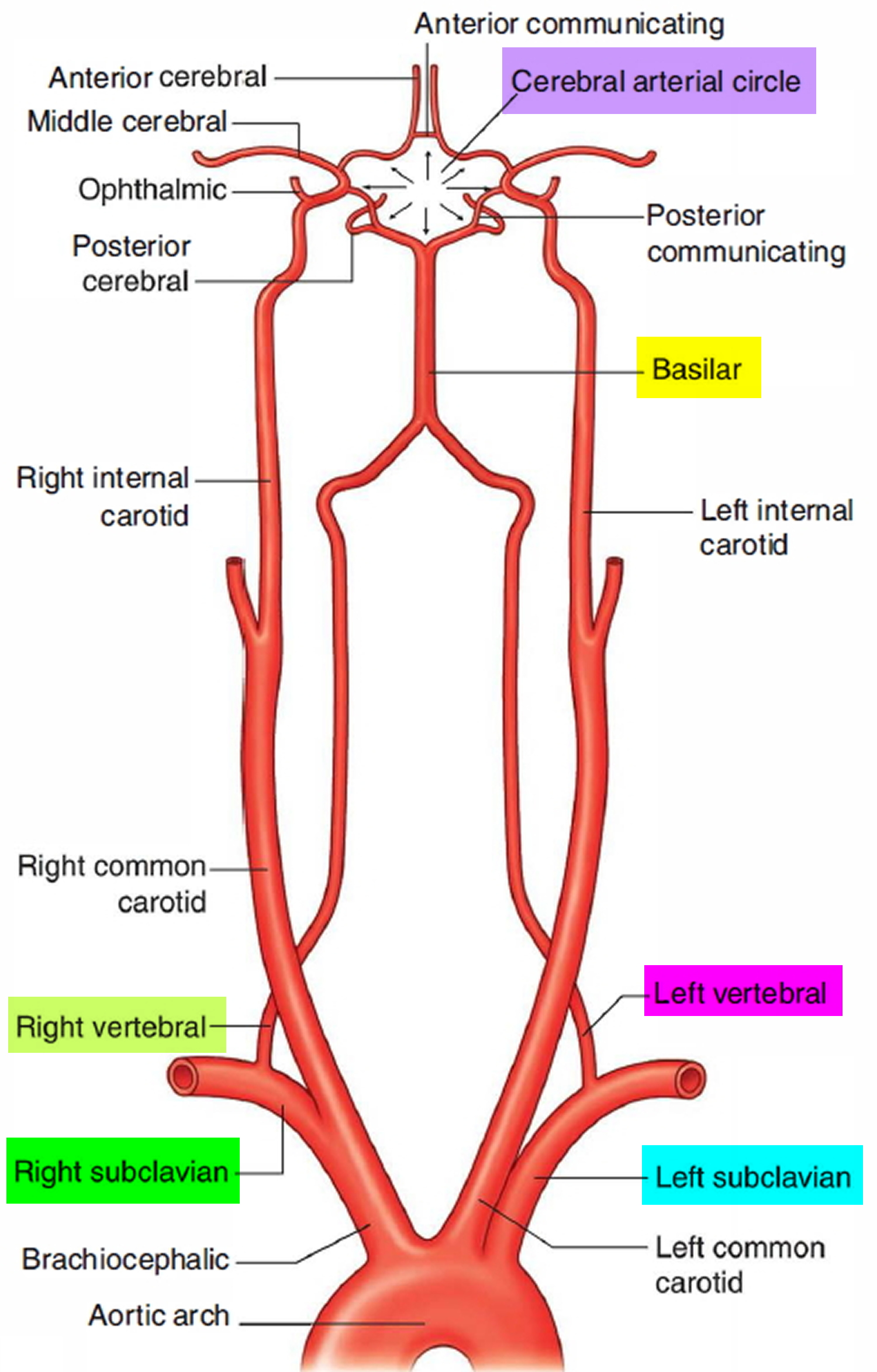
The brain receives its arterial supply from two pairs of vessels, the vertebral and internal carotid arteries (Figure 5), which are interconnected in the cranial cavity to produce a cerebral arterial circle (of Willis). The cerebral arterial circle (of Willis) is formed at the base of the brain by the interconnecting vertebrobasilar and internal carotid systems of vessels (Figure 5). This anastomotic interconnection is accomplished by an anterior communicating artery connecting the left and right anterior cerebral arteries to each other, and two posterior communicating arteries, one on each side, connecting the internal carotid artery with the posterior cerebral artery.
Each vertebral artery arises as the first branch of each subclavian artery (Figure 5) in the lower part of the neck, and passes superiorly through the transverse foramina of the upper six cervical vertebrae. On entering the cranial cavity through the foramen magnum each vertebral artery gives off a small meningeal branch. Continuing forward, the vertebral artery gives rise to three additional branches before joining with its companion vessel to form the basilar artery (Figure 5).
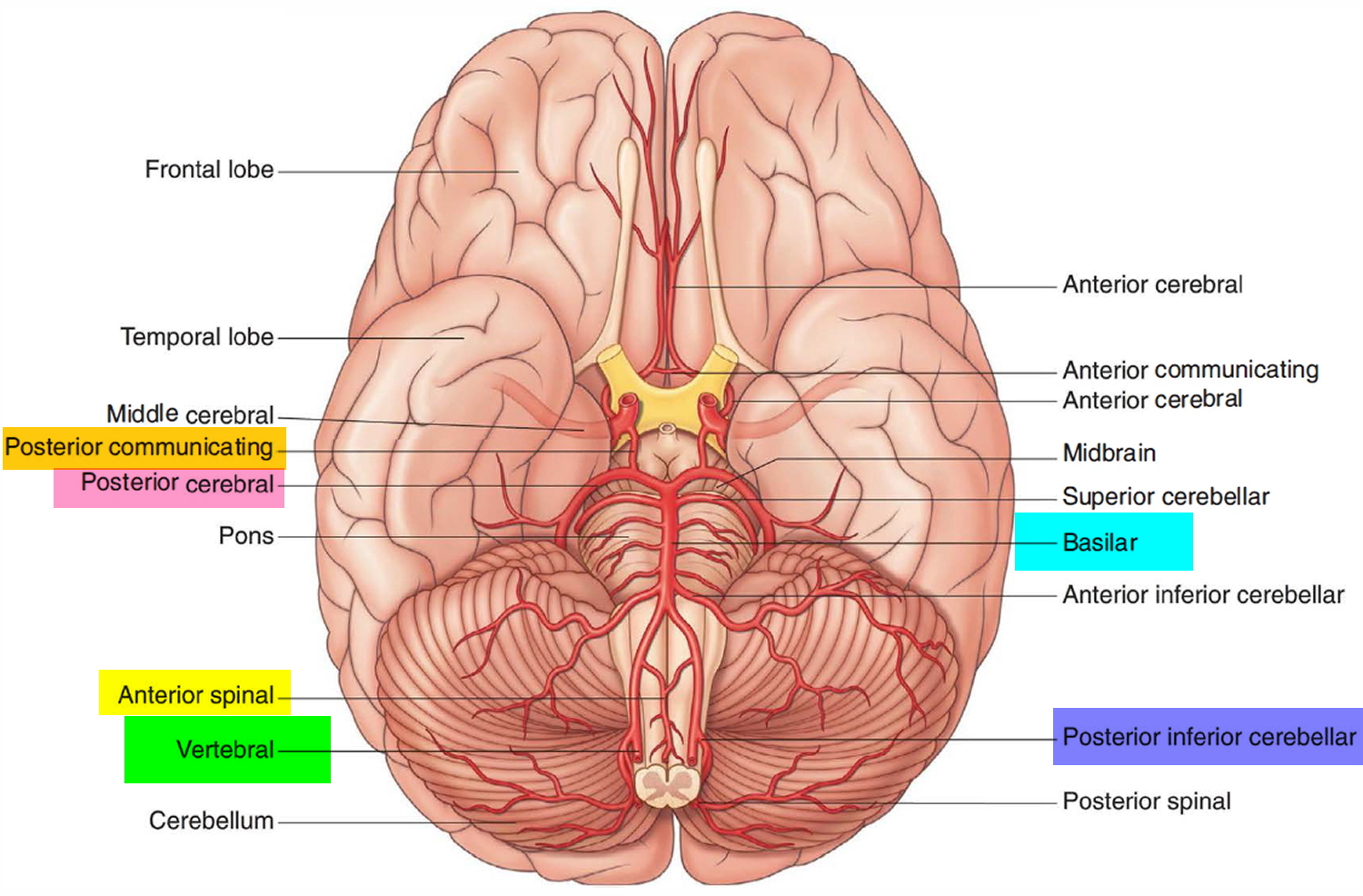
One branch joins with its companion from the other side to form the single anterior spinal artery, which then descends in the anterior median fissure of the spinal cord.
A second branch is the posterior spinal artery, which passes posteriorly around the medulla and then descends on the posterior surface of the spinal cord in the area of the attachment of the posterior roots, there are two posterior spinal arteries, one on each side (although the posterior spinal arteries can originate directly from the vertebral arteries, they more commonly branch from the posterior inferior cerebellar arteries).
Just before the two vertebral arteries join, each gives off a posterior inferior cerebellar artery.
The basilar artery travels in a rostral direction along the anterior aspect of the pons. Its branches in a caudal to rostral direction include the anterior inferior cerebellar arteries, several small pontine arteries, and the superior cerebellar arteries. The basilar artery ends as a bifurcation, giving rise to two posterior cerebral arteries.
Figure 5. Branches of subclavian artery to the brain
Figure 6. Brain blood supply
Thyrocervical trunk
The second branch of the subclavian artery is the thyrocervical trunk (Figure 4). It arises from the first part of the subclavian artery medial to the anterior scalene muscle, and divides into three branches- the inferior thyroid, the transverse cervical. and the suprascapular arteries.
Inferior thyroid artery. The inferior thyroid artery is the superior continuation of the thyrocervical trunk. It ascends, anterior to the anterior scalene muscle, and eventually turns medially, crossing posterior to the carotid sheath and its contents and anterior to the vertebral artery. Reaching the posterior surface of the thyroid gland it supplies the thyroid gland. When the inferior thyroid artery turns medially, it gives off an important branch (the ascending cervical artery), which continues to ascend on the anterior surface of the prevertebral muscles, supplying these muscles and sending branches to the spinal cord.
Transverse cervical artery. The middle branch of the thyrocervical trunk is the transverse cervical artery. This branch passes laterally, across the anterior surface of the anterior scalene muscle and the phrenic nerve, and enters and crosses the base of the posterior triangle of the neck. It continues to the deep surface of the trapezius muscle, where it divides into superficial and deep branches :
- The superficial branch continues on the deep surface of the trapezius muscle.
- The deep branch continues on the deep surface of the rhomboid muscles near the medial border of the scapula.
Suprascapular artery. The lowest branch of the thyrocervical trunk is the suprascapular artery. This branch passes laterally, crossing anterior to the anterior scalene muscle, the phrenic nerve, the third part of the subclavian artery, and the trunks of the brachial plexus. At the superior border of the scapula, it crosses over the superior transverse scapular ligament and enters the supraspinatus fossa.
Internal thoracic artery
The third branch of the subclavian artery is the internal thoracic artery (Figure 4). This artery branches from the inferior edge of the subclavian artery and descends. It passes posterior to the clavicle and the large veins in the region and anterior to the pleural cavity. It enters the thoracic cavity posterior to the ribs and anterior to the transversus thoracis muscle and continues to descend giving off numerous branches .
Costocervical trunk
The final branch of the subclavian artery in the root o f the neck is the costocervical trunk (Figure 4). It arises in a slightly different position, depending on the side:
- On the left, it arises from the first part of the subclavian artery, just medial to the anterior scalene muscle.
- On the right, it arises from the second part of the subclavian artery.
On both sides, the costocervical trunk ascends and passes posteriorly over the dome of the pleural cavity and continues in a posterior direction behind the anterior scalene muscle. Eventually it divides into two branches the deep cervical and the supreme intercostal arteries.
- The deep cervical artery ascends in the back of the neck and anastomoses with the descending branch of the occipital artery.
- The supreme intercostal artery descends anterior to first rib and divides to form the posterior intercostal arteries for the first two intercostal spaces.
Aberrant subclavian artery
Aberrant subclavian artery is a rare vascular anomaly that is present from birth 1. It usually causes no symptoms and is often discovered as an incidental finding (such as through a barium swallow or echocardiogram). Occasionally the anomaly causes swallowing difficulty (dysphagia) 2, 3. Swallowing symptoms in children may present as feeding difficulty and/or recurrent respiratory tract infection 3. When aberrant subclavian artery causes no symptoms, treatment is not needed. If the anomaly is causing significant symptoms, treatment may involve surgery 2, 3. Swallowing symptoms in children may present as feeding difficulty and/or recurrent respiratory tract infection 3. Children with symptomatic aberrant subclavian artery should be carefully evaluated for additional vascular and heart anomalies 3.
Aberrant right subclavian artery
Aberrant right subclavian artery or Lusoria artery is a relatively common congenital anomaly. It has a prevalence of up to 1.8% 4. About a third of people with this anatomic variant experience symptoms. Dysphagia (difficulty swallowing) is experienced in 90% of such cases, whereas dyspnea (difficult or laboured breathing), is less common 5. Dysphagia secondary to extrinsic esophageal compression by an aberrant right subclavian artery is known as dysphagia lusoria 4. The term, Lusoria artery, dates back to the first description of the condition in 1794 by David Bayford, who called it “lusus naturae,” meaning “freak or jest of nature” 6.
Essentially, right subclavian artery originates from the brachiocephalic artery, but in 0.4-1.8% of the general population it may arise directly from the aortic arch distal to the left subclavian artery 4, 7, 8. Aberrant right subclavian artery on its way to the right arm crosses the midline posterior to esophagus.
The anomaly may be associated with some clinical manifestations such as dyspnea, stridor (a harsh vibrating noise when breathing), dysphagia (which is called dysphagia lusoria), chest pain or fever 9, 10, 11, but majority of cases with aberrant right subclavian artery are asymptomatic.
In the symptomatic cases, particularly in the cases with high-risk dilated aneurysm, the general census is advocating surgical treatment 7, 12, 13. In asymptomatic cases, the anomaly has low importance, and there is no need for further intervention.
Even if it is asymptomatic, this anomaly should be taken into consideration during surgical procedures around esophagus, such as esophagectomy. Any unintentional injury of this artery during surgical procedures could be extremely life threatening.
Figure 7. Aberrant right subclavian artery
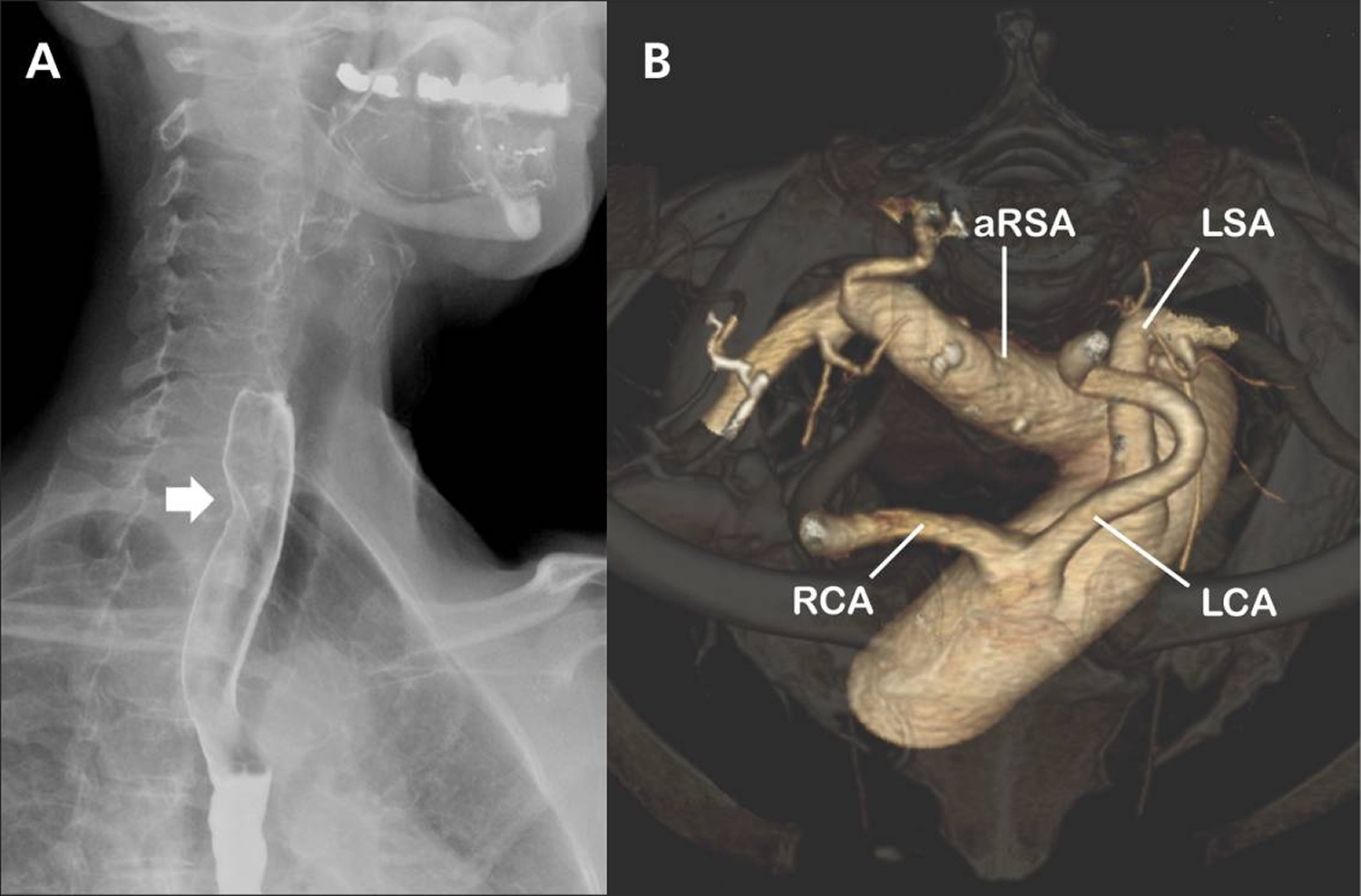
Note:
(A) A barium esophagogram (lateral projection) showing an oblique indentation of the posterior esophageal wall (arrow) that suggests extrinsic compression of the esophagus by an aberrant right subclavian artery.
(B) Volume-rendered, 3-dimensional computed tomography scan (cranial view) of the aortic arch. The right subclavian artery arises as the last branch from the aortic arch distal to the origin of the left subclavian artery. Notice the aneurysmal origin of the aberrant right subclavian artery.
RCA = right common carotid artery, LCA = left common carotid artery, LSA = left subclavian artery, aRSA = aberrant right subclavian artery.
[Source 4]Subclavian artery stenosis
Subclavian artery stenosis or occlusion has a low prevalence (1.9%) of the free-living population and is asymptomatic in most cases 14:618–623. https://www.ncbi.nlm.nih.gov/pubmed/15358030)). Additionally, subclavian artery stenosis is correlated with current and past smoking histories, systolic blood pressure (hypertension), HDL levels (low HDL “good” cholesterol levels), and the presence of peripheral artery disease. These findings suggest that bilateral brachial blood pressure measurements should routinely be performed in patients with an elevated risk profile, both to screen for subclavian artery stenosis and to avoid missing a hypertension or peripheral artery disease diagnosis because of unilateral pressure measurement in an obstructed arm.
Patients with atherosclerotic occlusive plaques in the subclavian artery are usually asymptomatic. Intervention is warranted in the symptomatic patient 15, 16. Hemodynamically significant stenosis of the subclavian artery usually presents with symptoms of upper limb ischemia on the ipsilateral side as the lesion. It may also present as subclavian steal syndrome with symptoms of vertebro-basilar insufficiency as a result of retrograde flow in the ipsilateral vertebral artery 17. The most common cause of subclavian artery stenosis is atherosclerosis but other causes include congenital abnormalities such as arteria lusoria (aberrant subclavian artery) or right sided aortic arch that can cause compression of the right subclavian artery leading to congenital subclavian steal syndrome 18.
There are several methods of treating symptomatic occlusive lesions of the proximal subclavian artery. An endovascular approach is attempted before proceeding to open subclavian artery revascularization as it is a less invasive procedure 19. Surgical revascularization is attempted either via transposition of subclavian to carotid artery, carotid – subclavian bypass using a synthetic graft or by subclavian – axillary bypass if the carotid is not feasible 20.
Angioplasty and stenting are first line interventions for symptomatic subclavian occlusive disease. A retrospective study on the long-term outcome of this endovascular intervention concluded that there was high primary success with satisfactory outcomes beyond 10 years 21. Angioplasty can however, can cause intraluminal hyperplasia and the re-stenosis rates are higher than for extrathoracic surgical revascularisation 22. Extrathoracic surgical revascularisation becomes necessary when endoluminal measures fail, or when anatomical variations make it more technically difficult.
The study conducted by Stone et al. 23 compared endovascular treatment and open surgery in subclavian occlusive disease. The study concluded that endoluminal therapy for subclavian disease is effective and safe; however, open surgery still carries a better long-term durability and should be the preferred approach in low-risk patients. Symptomatic patients who failed endovascular treatment or subsequent loss of patency by stent occlusion should be considered for surgical revascularization.
The carotid subclavian bypass is a safe and commonly used surgical procedure when endovascular intervention has failed 24. A retrospective study analysing outcomes of common carotid-subclavian artery bypass versus transposition of subclavian artery on the common carotid artery concluded that the latter should be considered the first line surgical management of proximal subclavian artery lesions. The study showed the six-year patency rate was one hundred percent for transposition of subclavian artery on the common carotid artery and sixty-six percent for carotid subclavian bypass 25.
Transposition of the subclavian artery onto the carotid artery is suitable if there is proximal stenosis of the subclavian artery, allowing for mobilisation of the subclavian artery distal to the stenosis. In the management of proximal subclavian artery stenosis it is safe and re-occlusion rates are low 26. Re-occlusion rates are lower than those for carotid-subclavian artery bypass using a graft 27:1275–1282. https://www.ncbi.nlm.nih.gov/pubmed/23384492)). This technique has been used to treat subclavian steal due to a congenital aberrant right subclavian artery 28. Another common reason for doing this operation is in preparation for stenting of thoracic aortic aneurysms, to reduce the risk of cerebral ischemia 29.
Carotid-subclavian artery bypass using a prosthetic graft is considered as a safe surgical intervention with mortality rates of 0–3% and stroke risk of 0–5% 30. These rates are comparable with those for transposition of the subclavian artery onto the carotid artery 31. A wide range of grafts may be used, including Dacron, autologous vein and PTFE. Takach et al. 32 have demonstrated the safety and effectiveness of carotid-subclavian bypass in their analysis of 287 patients treated for subclavian artery stenosis in this way. Carotid-subclavian bypass has also been used in repair of aberrant right subclavian artery aneurysm following endoluminal aortic stent graft exclusion 33.
Carotid-axillary bypass is less widely used than the other techniques. It has the advantage of avoiding the area of stenosis in cases where the subclavian artery has extensive disease and damage from previous angioplasty and stenting. It can be performed even with more distal disease in the subclavian artery 34.
It is important to consider the options available when considering surgical management of subclavian artery stenosis. In addition to atherosclerosis and damage from previous endovascular treatments, anatomical variations such arteria lusoria may complicate surgical decisions.
References- Aberrant subclavian artery. Genetic and Rare Diseases Information Center. https://rarediseases.info.nih.gov/diseases/5706/aberrant-subclavian-artery
- Pramesh CS, Saklani AP, Parmar V, Acharya S, Badwe RA. Aberrant subclavian artery causing difficulty in transhiatal esophageal dissection. Diseases of the Esophagus. 2003;16:173-176.
- Woods RK, Sharp RJ, Holcomb GW, Snyder CL, Laofland GK, Ashcraft KW, Holder TM. Vascular anomalies and tracheoesophageal compression: A single institution’s 25-year experience. Ann Thorac Surg. 2001;72:434-9.
- Dandelooy J, Coveliers JPM, Van Schil PEY, Anguille S. Dysphagia lusoria. CMAJ : Canadian Medical Association Journal. 2009;181(8):498. doi:10.1503/cmaj.081651. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2761442/
- Levitt B, Richter J. Dysphagia lusoria: a comprehensive review. Dis Esophagus. 2007;20:455–60. https://www.ncbi.nlm.nih.gov/pubmed/17958718
- Bayford D. An account of a singular case of obstructed deglutition. Memoirs Med Soc London. 1794;2:275–86.
- Hart PA, Kamath PS. Dysphagia Lusoria. Mayo Clinic Proceedings. 2012;87(3):e17. doi:10.1016/j.mayocp.2011.11.015. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3497995/
- Pramesh CS, Saklani AP, Parmar V, Acharya S, Badwe RA. Aberrant subclavian artery causing difficulty in transhiatal esophageal dissection. Dis Esophagus. 2003;16:173–6. https://www.ncbi.nlm.nih.gov/pubmed/12823224
- Abraham V, Mathew A, Cherian V, Chandran S, Mathew G. Aberrant subclavian artery: Anatomical curiosity or clinical entity. Int J Surg. 2009;7:106–9. https://www.ncbi.nlm.nih.gov/pubmed/19246267
- Saeed G, Ganster G, Friedel N. Arteria lusoria aneurysm with truncus bicaroticus: Surgical resection without restoring blood supply to the right arm. Tex Heart Inst J. 2010;37:602–7. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2953214/
- Saito T, Tamatsukuri Y, Hitosugi T, Miyakawa K, Shimizu T, Oi Y, et al. Three cases of retroesophageal right subclavian artery. J Nippon Med Sch. 2005;72:375–82. https://www.ncbi.nlm.nih.gov/pubmed/16415518
- Saeed G, Ganster G, Friedel N. Arteria Lusoria Aneurysm with Truncus Bicaroticus: Surgical Resection without Restoring Blood Supply to the Right Arm. Texas Heart Institute Journal. 2010;37(5):602-607. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2953214/
- Myers PO, Fasel JH, Kalangos A, Gailloud P. Arteria lusoria: Developmental anatomy, clinical, radiological and surgical aspects. Ann Cardiol Angeiol (Paris) 2010;59:147–54. https://www.ncbi.nlm.nih.gov/pubmed/19962688
- Shadman R., Criqui M.H., Bundens W.P., Fronek A., Denenberg J.O., Gamst A.C. Subclavian artery stenosis: prevalence, risk factors, and association with cardiovascular diseases. J Am Coll Cardiol. 2004;44(August (3
- Iared W., Mourão J.E., Puchnick A., Soma F., Shigueoka D.C. Angioplasty versus stenting for subclavian artery stenosis. Cochrane Database Syst. Rev. 2014;16(5). https://www.ncbi.nlm.nih.gov/pubmed/24833157
- Labropoulos N., Nandivada P., Bekelis K. Prevalence and impact of the subclavian steal syndrome. Ann. Surg. 2010;252(1):166–170. https://www.ncbi.nlm.nih.gov/pubmed/20531004
- Potter B.J., Pinto D.S. Subclavian steal syndrome. Circulation. 2014;129(22):2320–2323. http://circ.ahajournals.org/content/129/22/2320.long
- Stefańczyk L., Szymczyk K., Stefańczyk K., Polguj M. The presence of a right asortic arch associated with severe stenosis of the right common carotid artery and steal phenomenon. Ann. Vasc. Surg. 2015;29(8):1656. https://www.ncbi.nlm.nih.gov/pubmed/26184367
- Eisenhauer A.C. Subclavian and innominate revascularization: surgical therapy versus catheter-based intervention. Curr. Interv. Cardiol. Rep. 2000;2:101–110. https://www.ncbi.nlm.nih.gov/pubmed/11096657
- van der Vliet J.A., Palamba H.W., Scharn D.M., van Roye S.F., Buskens F.G. Arterial reconstruction for subclavian obstructive disease: a comparison of extrathoracic procedures. Eur. J. Vasc. Endovasc. Surg. 1995;4:454–458. https://www.ncbi.nlm.nih.gov/pubmed/7633992
- Mousa A.Y., AbuRahma A.F., Bozzay J., Broce M., Barsoum E., Bates M. Anatomic and clinical predictors of reintervention after subclavian artery stenting. J. Vasc. Surg. 2015;(15):S0741–S5214. https://www.ncbi.nlm.nih.gov/pubmed/25864043
- AbuRahma A.F., Bates M.C., Stone P.A., Dyer B., Armistead L., Scott Dean L., Scott Lavigne P. Angioplasty and stenting versus carotid-subclavian bypass for the treatment of isolated subclavian artery disease. J. Endovasc. Ther. 2007;14(5):698–704. https://www.ncbi.nlm.nih.gov/pubmed/17924737
- Stone P.A., Srivastiva M., Campbell J.E., Mousa A.Y. Diagnosis and treatment of subclavian artery occlusive disease. Expert Rev. Cardiovasc. Ther. 2010;8(9):1275–1282. https://www.ncbi.nlm.nih.gov/pubmed/20828350
- AbuRahma A.F., Robinson P.A., Jennings T.G. Carotid-subclavian bypass grafting with polytetrafluoroethylene grafts for symptomatic subclavian artery stenosis or occlusion: a 20-year experience. J. Vasc. Surg. 2000;32(3):411–419. https://www.ncbi.nlm.nih.gov/pubmed/10957647
- Deriu G.P., Milite D., Verlato F., Cognolato D., Frigatti P., Zaramella M., Mellone G., Greco F. Surgical treatment of atherosclerotic lesions of subclavian artery: carotid-subclavian bypass versus subclavian-carotid transposition. J. Cardiovasc. Surg. (Torino) 1998;39(6):729–734. https://www.ncbi.nlm.nih.gov/pubmed/9972889
- Duran M., Grotemeyer D., Danch M.A., Grabitz K., Schelzig H., Sagban T.A. Subclavian carotid transposition: immediate and long-term outcomes of 126 surgical reconstructions. Ann. Vasc. Surg. 2015;29(3):397–403. https://www.ncbi.nlm.nih.gov/pubmed/25463327
- Madenci A.L., Ozaki C.K., Belkin M., McPhee J.T. Carotid-subclavian bypass and subclavian-carotid transposition in the thoracic endovascular aortic repair era. J. Vasc. Surg. 2013;57(May (5
- De Vleeschauwer P., Horsch S. Subclavian steal syndrome in a congenitaly anomalous subclavian artery: a case report. Ann. Vasc. Surg. 1986;1(3):389–391. https://www.ncbi.nlm.nih.gov/pubmed/3504353
- Xydas S., Wei B., Takayama H., Russo M., Bacchetta M., Smith C.R., Stewart A. Use of carotid-subclavian arterial bypass and thoracic endovascular aortic repair to minimize cerebral ischemia in total aortic arch reconstruction. J. Thorac. Cardiovasc. Surg. 2010;139(3):717–722. https://www.ncbi.nlm.nih.gov/pubmed/20080269
- Law M.M., Colburn M.D., Moore W.S., Quinones-Baldrich W.J., Machleder H.I., Gelabert H.A. Carotid-subclavian bypass for brachiocephalic occlusive disease. Stroke. 1995;26:1565–1571. http://stroke.ahajournals.org/content/26/9/1565.long
- Madenci A.L., Ozaki C.K., Belkin M., McPhee J.T. Carotid-subclavian bypass and subclavian-carotid transposition in the thoracic endovascular aortic repair era. J. Vasc. Surg. 2013;57(5):1275–1282. https://www.ncbi.nlm.nih.gov/pubmed/23384492
- Takach T.J., Duncan J.M., Livesay J.J., Ott D.A., Cervera R.D., Cooley D.A. Contemporary relevancy of carotid-subclavian bypass defined by an experience spanning five decades. Ann. Vasc. Surg. 2011;25(7):895–901. https://www.ncbi.nlm.nih.gov/pubmed/21831586
- Daniels L., Coveliers H.M., Hoksbergen A.W., Nederhoed J.H., Wisselink W. Hybrid treatment of aberrant right subclavian artery and its aneurysms. Acta. Chir. Belg. 2010;110(3):346–349. https://www.ncbi.nlm.nih.gov/pubmed/20690521
- Salman R, Hornsby J, Wright LJ, et al. Treatment of subclavian artery stenosis: A case series. International Journal of Surgery Case Reports. 2016;19:69-74. doi:10.1016/j.ijscr.2015.12.011. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4756098/