What is dehydration
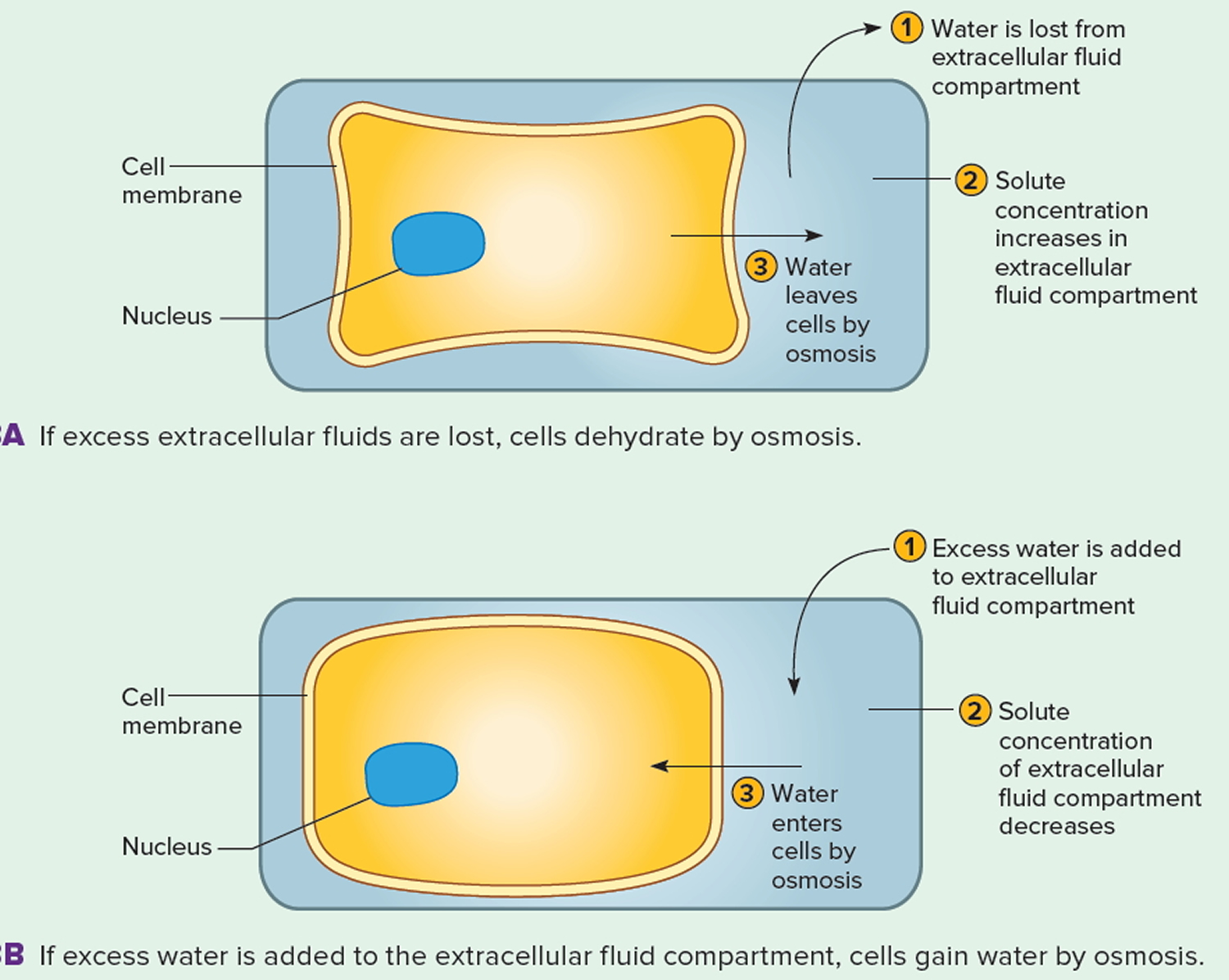
Dehydration is a state of total body water deficit due to primary water losses (e.g., sweating or gastrointestinal losses) or to inadequate water intake 1. In dehydration, water output exceeds water intake. Dehydration may develop following excessive sweating or as a result of prolonged water deprivation accompanied by continued water output 2. The extracellular fluid becomes more concentrated, and water leaves cells by osmosis (Figure 1 A). Dehydration may also accompany prolonged vomiting or diarrhea that depletes body fluids. During dehydration, the skin and mucous membranes of the mouth feel dry, and body weight drops. Severe hyperthermia may develop as the body’s temperature-regulating mechanism falters due to lack of water for sweat production. Infants are more likely to become dehydrated because their kidneys are less efficient at conserving water than those of adults. Elderly people are also especially susceptible to developing water imbalances because the sensitivity of their thirst mechanism decreases with age, and physical disabilities may make it difficult for them to obtain adequate fluids. The treatment for dehydration is to replace the lost water and electrolytes. If only water is replaced, the extracellular fluid will become more dilute than normal, causing cells to swell (Figure 1 B). This may produce a condition called water intoxication.
Figure 1. The effect of dehydration on cells
[Source 2]The severity of acute or subacute dehydration is characterized by the percentage of body weight loss 3. However, there is no generally accepted definition of different degrees of dehydration in adults 4. Mild dehydration has been defined as 1–2% loss of body weight 5, 6, 7, while severe dehydration represented a body weight loss of more than 5% 8, 9, 10. Moderate dehydration in the adult was defined as a 2–5% loss of body weight 4.
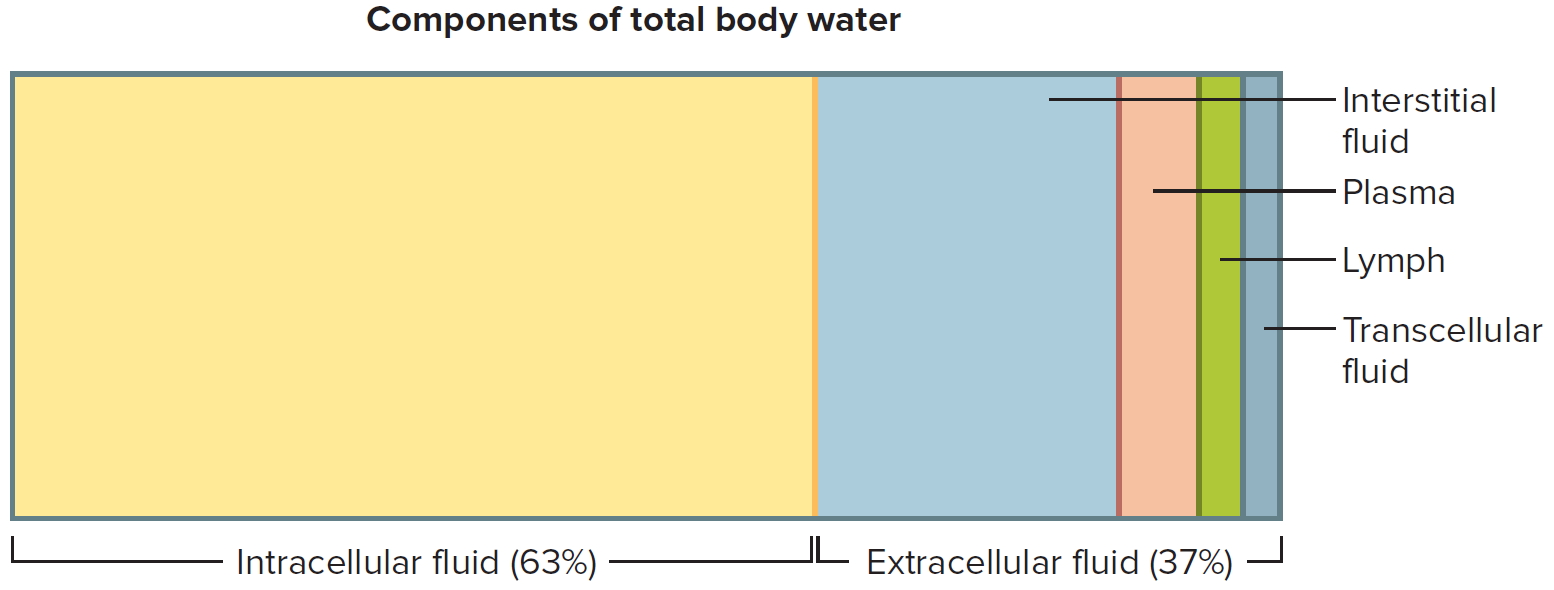
Water makes up at least two-thirds of the human body 11. About 60% of our body weight is made of water 12. The body of an average adult female is about 52% water by weight and that of an average male is about 63% water by weight. The reason for this difference is that females generally have more adipose tissue, which contains little water 2. Males generally have proportionately more muscle tissue, which contains a great deal of water. Water in the adult human body (about 40 liters), with its dissolved electrolytes, is distributed into two major compartments: an intracellular fluid compartment and an extracellular fluid compartment. This water content varies with body composition (lean and fat mass) 13. In infants and young children, water as a percentage of body weight is higher than in adults. This is mainly due to higher water content in the extracellular compartment, whereas the water content in the intracellular compartment is lower in infants than in older children and adults 12. Body composition changes rapidly during the first year of life, with a decrease in the water content of the fat-free mass and an increase in the content of protein and minerals 12.
In adults, about two-thirds of total water is in the intracellular space, whereas one-third is extracellular water 12. The intracellular fluid compartment includes all the water and electrolytes that cell membranes enclose. In other words, intracellular fluid is the fluid inside cells. In an adult it accounts for about 63% by volume of total body water.
The extracellular fluid compartment includes all the fluid outside of cells—in tissue spaces (interstitial fluid), blood vessels (plasma), and lymphatic vessels (lymph). Transcellular fluid, a type of extracellular fluid, is found in cavities separated from other extracellular fluids by epithelial or connective tissue membranes. Transcellular fluid includes cerebrospinal fluid of the central nervous system, aqueous and vitreous humors of the eyes, synovial fluid of the joints, and serous fluid in the body cavities. The fluids of the extracellular compartment constitute about 37% by volume of total body water.
A 70kg human has about 42 liters of total body water, of which 28 liters is intracellular water and 14 liters is extracellular fluid (ECF) 14. Of the latter, 3 liters is in blood plasma, 1 liter is the transcellular fluid (cerebrospinal fluid, ocular, pleural, peritoneal and synovial fluids) and 10 liters is the interstitial fluid, including lymph, which provides an aqueous medium surrounding cells 12.
Figure 2. Components of total body water
Body Fluid Composition
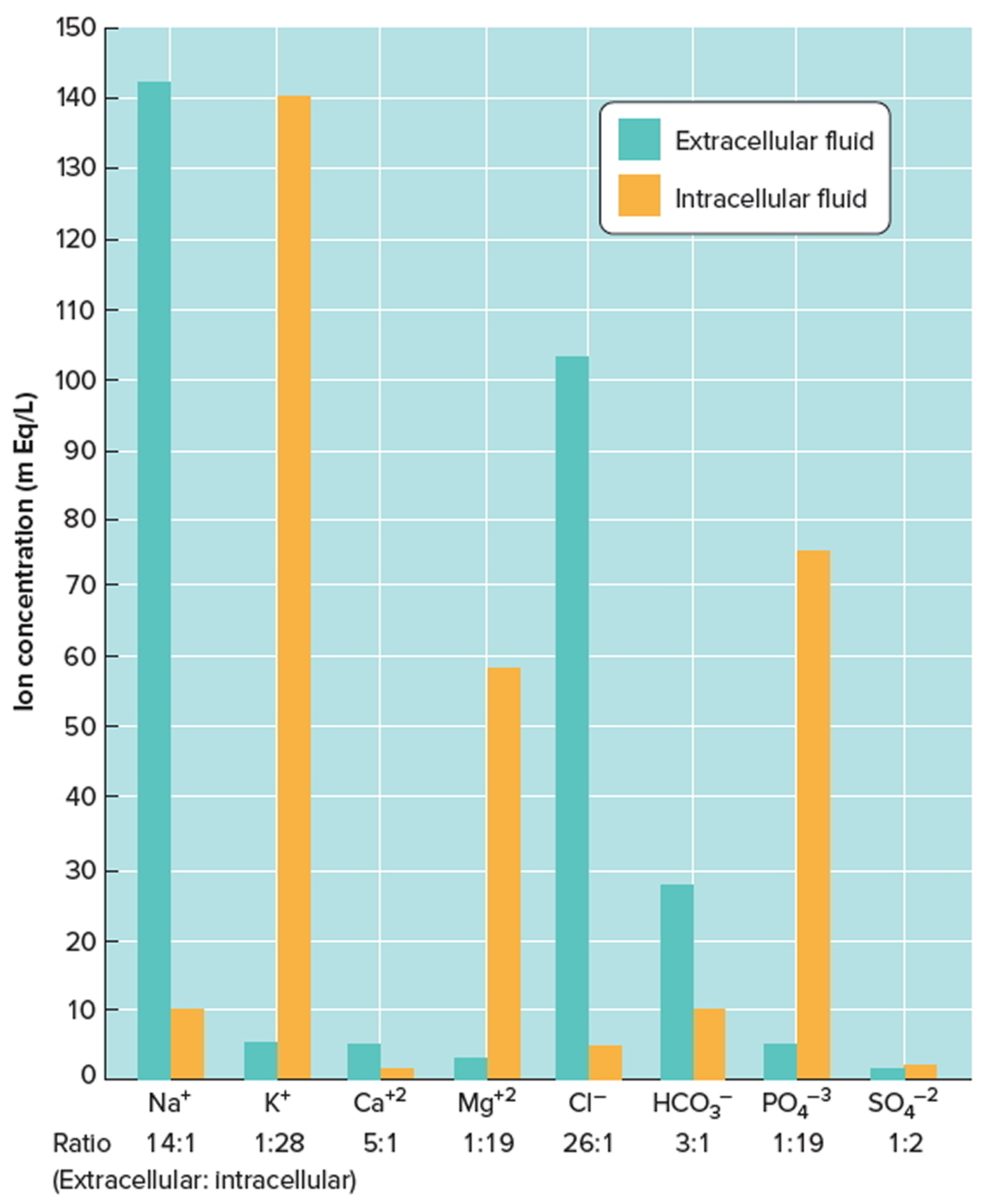
- Extracellular fluids (ECF) generally are similar in composition, including high concentrations of sodium, chloride, calcium, and bicarbonate ions and lesser concentrations of potassium, magnesium, phosphate, and sulfate ions. The blood plasma portion of extracellular fluid has considerably more protein than does either interstitial fluid or lymph.
- Intracellular fluid (ICF) has high concentrations of potassium, phosphate, and magnesium ions. It includes a greater concentration of sulfate ions and lesser concentrations of sodium, chloride, calcium, and bicarbonate ions than does extracellular fluid. Intracellular fluid also has a greater concentration of protein than does plasma. Figure 2 shows these relative concentrations.
The constancy of the amount and composition of extracellular fluid (ECF) is a necessity for the function of cells. This constancy is due to the homeostatic mechanisms that monitor and regulate its composition, osmotic pressure, pH and temperature 15. These mechanisms rely on the function of the main systems of the body, such as the circulatory, respiratory, renal and alimentary systems. The monitoring and regulation of these systems are coordinated by the nervous and endocrine systems. The composition of the intracellular fluid is maintained by solute movement across the cell membrane by passive or active transports 15.
An average person on an average day needs about 3 liters of water. But if you’re out in the hot sun, you’ll need a lot more than that. Most healthy bodies are very good at regulating water. Elderly people, young children and some special cases – like people taking certain medications – need to be a little more careful.
Water plays a large part in your normal body functions, drinking enough water is essential for physiological processes such as circulation, metabolism, temperature regulation, and waste removal 16. Water is the main constituent of cells, tissues and organs and is vital for life 17. Water as a vital nutrient, a multifunctional constituent of the human body. Every day you lose water through your breath, perspiration, urine and bowel movements.
Water is essential for cellular homeostasis because it transports nutrients to cells and removes wastes from cells 18. It is the medium in which all transport systems function, allowing exchanges between cells, interstitial fluid and capillaries 19. Water maintains the vascular volume and allows blood circulation, which is essential for the function of all organs and tissues of the body 20. Thus, the cardiovascular and respiratory systems, the digestive tract, the reproductive system, the kidney and liver, the brain and the peripheral nervous system, all depend on adequate hydration to function effectively 18. Severe dehydration therefore affects the function of many systems and is a life-threatening condition 21.
For your body to function properly, you must replenish its water supply by consuming beverages and foods that contain water. Although excessive dehydration is associated with serious health problems, even mild dehydration can cause issues, including headaches, irritability, poorer physical performance, and reduced cognitive functioning. Once the water in your body is reduced, it needs to be replaced because an imbalance between the salts and sugar in your body can affect the way you will perform. Dehydration has been classically referred to the excessive loss of body water through conditions such as diarrhea, sweating, or urinary losses, but among the lay public dehydration may also refer to the loss of both water and salt leading to a hypovolemic state 22.
How you use water to keep every system in your body functioning properly
- carrying nutrients and oxygen to your cells
- flushing bacteria from your bladder
- aiding digestion
- preventing constipation
- normalizing blood pressure
- stabilizing the heartbeat
- cushioning joints
- protecting organs and tissues
- regulating body temperature
- maintaining electrolyte (sodium) balance.
Giving your body enough fluids to carry out those tasks means that you’re staying hydrated.
If you don’t drink enough water, you risk becoming dehydrated. Warning signs of dehydration include weakness, low blood pressure, dizziness, confusion, or urine that’s dark in color.
How much water should you drink each day ? It’s a simple question with no easy answers. Studies have produced varying recommendations over the years, but in truth, your water needs depend on many factors, including your health, how active you are and where you live.
Although no single formula fits everyone, knowing more about your body’s need for fluids will help you estimate how much water to drink each day.
If your body has lost one to two percent of its entire water content, you will feel thirsty. Thirst is normally just the brain’s way of warning that you’re dehydrated because you’re not drinking enough fluid. But excessive and persistent thirst (known as polydipsia) could be a sign of an underlying problem such as diabetes.
Dehydration happens when you’ve lost too much water in your body without replacing it, preventing your body to perform its normal functions. Mild dehydration can easily be treated but if it reaches extreme levels, it can be life-threatening and will require immediate medical attention.
It’s particularly important to stay well hydrated during hot weather, while exercising and while you’re unwell with vomiting and diarrhea.
Distribution of Body Fluids
Body fluids are not uniformly distributed. Instead, they occupy regions, or compartments, of different volumes that contain fluids of varying compositions. The movement of water and electrolytes between these compartments is regulated to stabilize both the distribution and the composition of body fluids.
Fluid Compartments
The body of an average adult female is about 52% water by weight and that of an average male is about 63% water by weight. The reason for this difference is that females generally have more adipose tissue, which contains little water 2. Males generally have proportionately more muscle tissue, which contains a great deal of water. Water in the adult human body (about 40 liters), with its dissolved electrolytes, is distributed into two major compartments: an intracellular fluid compartment and an extracellular fluid compartment. The intracellular fluid compartment includes all the water and electrolytes that cell membranes enclose. In other words, intracellular fluid is the fluid inside cells. In an adult it accounts for about 63% by volume of total body water.
The extracellular fluid compartment includes all the fluid outside of cells—in tissue spaces (interstitial fluid), blood vessels (plasma), and lymphatic vessels (lymph). Transcellular fluid, a type of extracellular fluid, is found in cavities separated from other extracellular fluids by epithelial or connective tissue membranes. Transcellular fluid includes cerebrospinal fluid of the central nervous system, aqueous and vitreous humors of the eyes, synovial fluid of the joints, and serous fluid in the body cavities. The fluids of the extracellular compartment constitute about 37% by volume of total body water.
Figure 3. Intracellular and extracellular fluid electrolyte concentrations
Note: Extracellular fluids have relatively high concentrations of sodium (Na+), calcium (Ca+2), chloride (Cl−), and bicarbonate (HCO3−) ions. Intracellular fluid has relatively high concentrations of potassium (K+), magnesium (Mg+2), phosphate (PO4−3), and sulfate (SO4−2) ions.
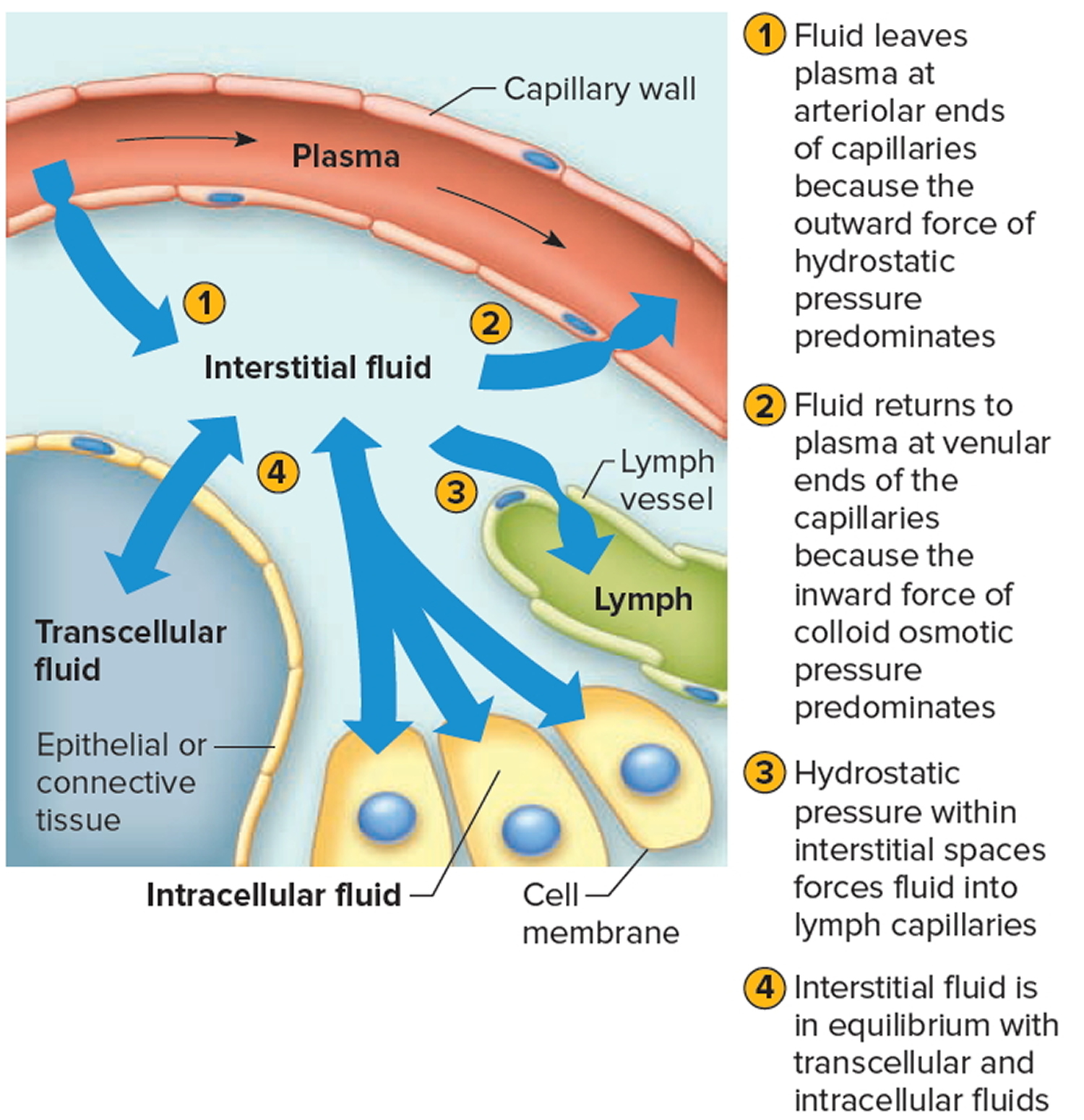
[Source 2]Movement of Fluid Between Compartments
Two major factors contribute to the movement of fluid from one compartment to another: hydrostatic pressure and osmotic pressure (Figure 1 A & B and Figure 4). For example, blood vessels, fluid leaves the plasma at the arteriolar ends of capillaries and enters the interstitial spaces because of the net outward force of hydrostatic pressure (blood pressure). Fluid returns to the plasma from the interstitial spaces at the venular ends of capillaries because of the net inward force of colloid osmotic pressure due to the plasma proteins. Likewise, tissue fluid and lymph, fluid leaves the interstitial spaces and enters the lymph capillaries due to the hydrostatic pressure of the interstitial fluid. The circulation of lymph returns interstitial fluid to the plasma.
Hydrostatic pressure in the cells and surrounding interstitial fluid is ordinarily equal and remains stable. Therefore, any net fluid movement is likely to be the result of changes in osmotic pressure.
The total solute concentration in extracellular and intracellular fluids is normally equal. However, a decrease in extracellular sodium ion concentration causes a net movement of water from the extracellular compartment into the intracellular compartment by osmosis. The cells swell. Conversely, if the extracellular sodium ion concentration increases, cells shrink as they lose water by osmosis.
Figure 4. Net movements of fluids between compartments result from differences in hydrostatic and osmotic pressures
[Source 2]Water Balance
Dehydration, water intoxication, and water retention (edema) are among the more common disorders that involve a water imbalance in body fluids.
For homeostasis, water balance and electrolyte balance must be maintained; that is, the quantities entering the body must equal the quantities leaving it 2. In other word, homeostasis requires control of both water intake and water output. Water balance exists when water intake equals water output. Therefore, the body requires mechanisms to:
- Replace lost water and electrolytes, and
- Excrete any excess water and electrolytes.
Levels of electrolytes in your body can become too low or too high. That can happen when the amount of water in your body changes, causing dehydration or overhydration. Causes include some medicines, vomiting, diarrhea, sweating or kidney problems 23. Problems most often occur with levels of sodium, potassium or calcium.
Water balance and electrolyte balance are interdependent because electrolytes are dissolved in the water of body fluids. Consequently, anything that alters the concentrations of the electrolytes will alter the concentration of the water, either by adding solutes to it or by removing solutes from it. Likewise, anything that changes the concentration of water will change concentrations of the electrolytes by concentrating or diluting them.
- Water Intake
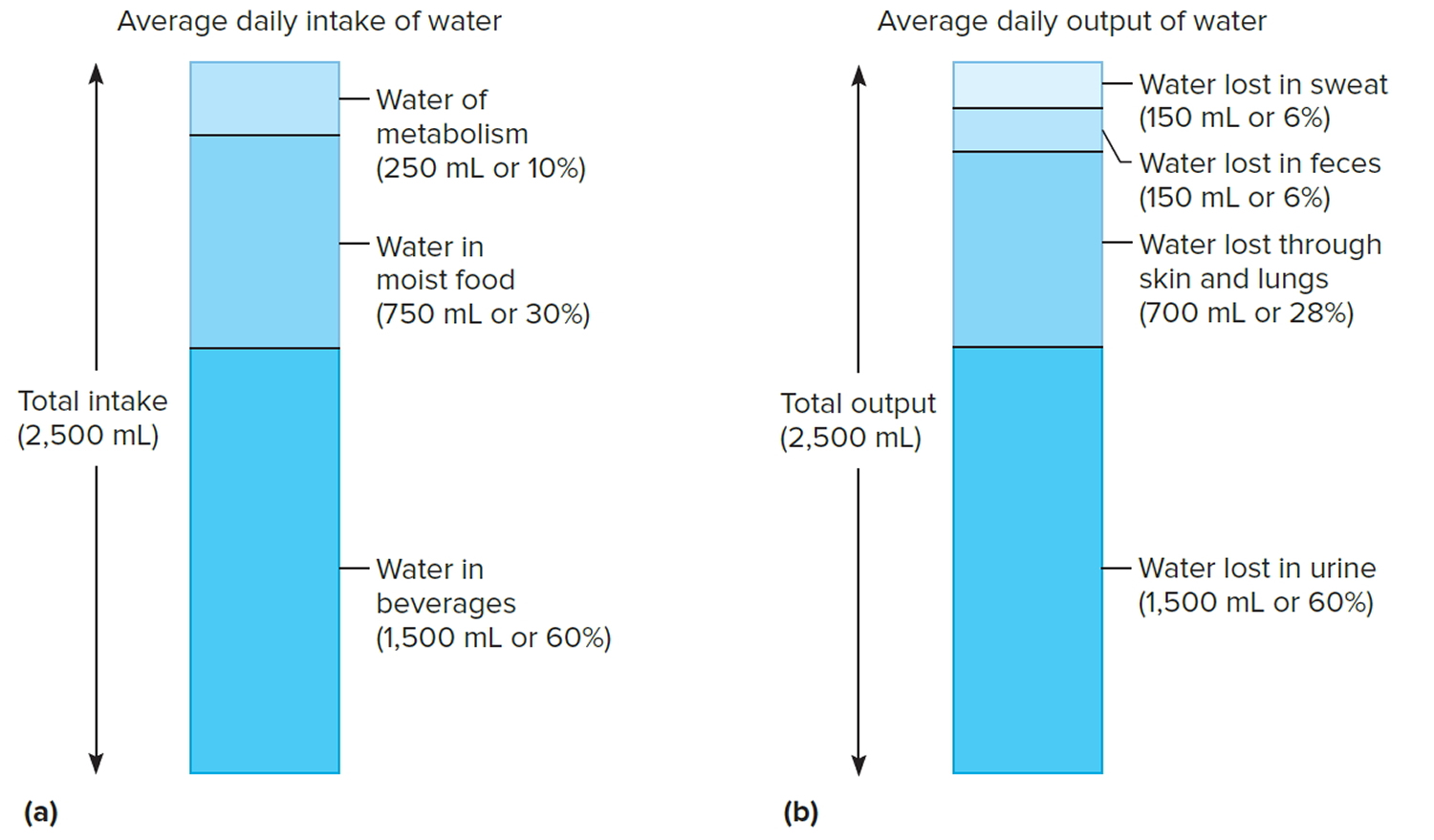
The volume of water gained each day varies among individuals. An average adult living in a moderate environment takes in about 2.5 liters. Probably 60% is obtained from drinking water or beverages, and another 30% comes from moist foods. The remaining 10% is a by-product of the oxidative metabolism of nutrients, called water of metabolism.
The primary regulator of water intake is thirst. The intense feeling of thirst derives from the osmotic pressure of extracellular fluids and both neural and hormonal input to the brain. As the body loses water, the osmotic pressure of extracellular fluids increases. Such a change stimulates osmoreceptors in the hypothalamus, and in response, the hypothalamus causes the person to feel thirsty and to seek water.
Thirst is a homeostatic mechanism, normally triggered when total body water decreases by as little as 1%. The act of drinking and the resulting distension of the stomach wall trigger impulses that inhibit the thirst mechanism. In this way, drinking stops even before the swallowed water is absorbed, preventing the person from drinking more than is required to replace the volume lost, avoiding development of an imbalance.
- Water Output
Water normally enters the body only through the mouth, but it can be lost by a variety of routes. These include obvious losses in urine, feces, and sweat (sensible perspiration), as well as evaporation of water from the skin (insensible perspiration) and from the lungs during breathing.
If an average adult takes in 2.5 liters of water each day, then 2.5 liters must be eliminated to maintain water balance. Of this volume, perhaps 60% is lost in urine, 6% in feces, and 6% in sweat. About 28% is lost by evaporation from the skin and lungs. These percentages vary with such environmental factors as temperature
and relative humidity and with physical exercise.
The primary means of regulating water output is urine production. The renal distal convoluted tubules of the nephrons and collecting ducts are the effectors of the mechanism that regulates urine volume. The epithelial linings in these structures remain relatively impermeable to water unless antidiuretic hormone (ADH) is present. Antidiuretic hormone (ADH) increases the permeability of the distal convoluted tubule and collecting duct, thereby increasing water reabsorption and reducing urine production. In the absence of antidiuretic hormone (ADH), less water is reabsorbed and more urine is produced.
Figure 5. Water balance
Note: Water balance. (a) Major sources of body water. (b) Routes by which the body loses water.
[Source 2]Electrolyte balance
Electrolyte balance exists when the quantities of electrolytes the body gains equal those lost. Homeostatic mechanisms maintain electrolyte balance. This involves keeping the associated ions in appropriate concentrations within the plasma and the interstitial fluid.
- Electrolyte Intake
The electrolytes of greatest importance to cellular functions dissociate to release sodium, potassium, calcium, magnesium, chloride, sulfate, phosphate, bicarbonate, and hydrogen ions. These electrolytes are primarily obtained from foods, but they may also be found in drinking water and other beverages. In addition, some electrolytes are by-products of metabolic reactions.
Ordinarily, a person obtains sufficient electrolytes by responding to hunger and thirst. However, a severe electrolyte deficiency may cause salt craving, which is a strong desire to eat salty foods.
- Electrolyte Output
The body loses some electrolytes by perspiring, with more lost in sweat on warmer days and during strenuous exercise. Varying amounts of electrolytes are lost in the feces. The greatest electrolyte output occurs as a result of kidney function and urine production. The kidneys alter electrolyte output to maintain the proper composition of body fluids, thereby promoting homeostasis.
Precise concentrations of positively charged ions, such as sodium (Na+), potassium (K+), and calcium (Ca+2), are required for impulse conduction along an axon, muscle fiber contraction, and maintenance of cell membrane potential. Sodium ions account for nearly 90% of positively charged ions in extracellular fluids. The kidneys and the hormone aldosterone regulate these ions. Aldosterone, which the adrenal cortex secretes, increases sodium ion reabsorption in the distal convoluted tubules of the kidneys’ nephrons and in the collecting ducts.
Aldosterone also regulates potassium ion concentration. A rising potassium ion concentration directly stimulates cells of the adrenal cortex to secrete aldosterone. This hormone enhances tubular secretion of potassium ions at the same time that it causes tubular reabsorption of sodium ions.
The calcium ion concentration dropping below normal directly stimulates the parathyroid glands to secrete parathyroid hormone. This hormone returns the concentration of calcium in extracellular fluids toward normal.
Generally the regulatory mechanisms that control positively charged ions secondarily control the concentrations of negatively charged ions. For example, chloride ions (Cl−), the most abundant negatively charged ions in extracellular fluids, are passively reabsorbed in response to the active tubular reabsorption of sodium ions. That is, the negatively charged chloride ions are electrically attracted to positively charged sodium ions and accompany them as they are reabsorbed. Water is then reabsorbed by osmosis.
Active transport mechanisms with limited transport capacities partially regulate some negatively charged ions, such as phosphate ions (PO4−3) and sulfate ions (SO4−2). Therefore, if the extracellular phosphate ion concentration is low, renal tubules reabsorb phosphate ions. On the other hand, if the renal plasma threshold is exceeded, excess phosphate is excreted in urine.
Types of Dehydration
There are three types of dehydration 24: (1) Isotonic dehydration in which net salt and water loss is equal, (2) Hypertonic dehydration characterized by loss of water in excess of salt and (3) Hypotonic dehydration, characterized by loss of salt in excess of water.
Isotonic dehydration
In isotonic dehydration, salt may be lost isotonically from the gastrointestinal tract, such as after profuse diarrhea 25. Only the extracellular fluid (ECF) volume is reduced, and treatment is the prescription of isotonic salt solutions, such as the World health Organization’s rehydration solution for the treatment of diarrhea, a solution that is widely used in developing countries.
Hypertonic dehydration
Inadequate water intake and excessive water loss are the two mechanisms responsible for the development of hypertonic dehydration 25. Insufficient water intake may be caused by defective thirst or impaired consciousness, or by a lack of available water. Large water loss may result from osmotic diuresis or diabetes insipidus. Vomiting is accompanied by a loss of hydrochloric acid, which is almost equivalent to the loss of pure water because NaHCO3 (which is finally absorbed and passes into the blood) replaces it. Sweating can represent an important hypotonic fluid loss when exercising in a hot environment.
Hypotonic dehydration
Hypotonic dehydration occurs when losses of gastrointestinal fluids (which are either hypotonic or isotonic in relation to plasma) are replaced by water, or by a solution that contains less Na+ and K+ than the fluid that has been lost 26. The reduced osmolarity of extracellular fluid (ECF) causes a shift of water into the intracellular fluid to reach osmotic balance. Hence, cell volume increases in spite of a reduction in extracellular fluid (ECF). Treatment of hypotonic dehydration may need both hypertonic saline to restore the osmolarity of body fluids and isotonic saline to compensate the loss of extracellular fluid (ECF).
Measurement of Hydration Status
A normal hydration status is the condition of healthy individuals who maintain their water balance. It is of practical importance to be able to assess the degree of hydration in individuals exposed to ambient conditions that can induce dehydration 27. In particular, elderly persons are prone to water deficit during the summer period because of blunted thirst and less efficient renal urinary concentrating mechanisms. Mild dehydration of 1 or 2% of body water can impair cognitive functions, alertness and capacity for exercise. Young infants are also prone to dehydration because they cannot express their sensation of thirst.
Body weight
The commonly used technique to measure changes in hydration status is the measurement of body weight changes that occur during short periods of time 28. When an individual is in a caloric balance, a body weight loss essentially equals water loss. Measurements of body weight must be carried out under standard conditions, preferably in the morning in the fasted state and after micturition and defecation.
Tracer techniques
Total body water can be measured by using tracer techniques, such as the use of deuterium oxide, which is a stable isotope of hydrogen 29. By determining the amount of tracer given and the equilibration concentration of the tracer in a body fluid, one can calculate the volume into which the tracer has been diluted. Tracer methods are mainly research tools and are not used in clinical practice.
Bioelectrical impedance analysis (BIA)
It is a technique that measures the resistance of body tissue and water to an electrical current that flows through the body 30. The method is easy to use, but many factors reduce the reliability and accuracy of this technique. These factors include the site placement of electrodes and problems of inadequate skin contact of electrodes, changes in plasma osmolarity and plasma sodium concentration and effect of posture. In spite of improvements in the initial technique that used a single frequency, the technological advancements, which allow impedance to be measured at numerous frequencies, have not brought about a significant improvement, and the bioelectrical impedance method remains inappropriate for measuring small changes in total body water in the range of 1 liter 31.
Plasma or serum osmolarity, plasma indices
The term osmolality (osmol/kg solvent) is also used instead of osmolarity (osmol/l solution). However, because in dilute aqueous solutions the molal concentrations (mol/kg water) closely approximate molar concentrations (mol/l solution), the terms osmolality and osmolarity are used interchangeably. Plasma or serum osmolarity is tightly controlled and rarely varies by more than 2% around a set point of 280–290 mosm/l. In well-hydrated individuals, a basal mean value of 287 mosm/l is maintained by hypothalamic osmoreceptors that control antidiuretic hormone (ADH) secretion (Figure 1 below). An osmolarity increase of 1% is sufficient to initiate a sensation of thirst and to increase the antidiuretic hormone (ADH) plasma concentration by 100% of the basal value. Therefore, measurement of changes in plasma osmolarity is the most widely used hematological index of hydration 26. However, if osmolarity is increased by solutes such as urea and glucose, which penetrate plasma membrane, antidiuretic hormone (ADH) release and thirst are not initiated. Although hypernatraemia is a sign of hypertonic dehydration, an increase in blood urea is not a valid indicator of a negative water balance, as its value depends on kidney function and protein intake.
Urine indices
Urinary indices of hydration, such as urine osmolarity 32, urine-specific gravity or 24-h urine volume, may be used, but urine variables often mirror the recent volume of fluid consumed rather than the state of hydration 33. For example, the intake of a large volume of water rapidly dilutes the plasma and the kidneys excrete diluted urine even if dehydration exists.
In conclusion, even if there is no real consensus with regard to the method by which to measure hydration status, for clinicians and general practitioners, the urine color chart can be used as an indicator 34. This is used, for example, in nursing home residents because it is a low-cost and rapid method for assessing hydration status, which can help in early intervention. Depending on the color of the urine sample that matches with the color on the chart, one can identify patients who are well hydrated or those who are poorly hydrated and who should consume fluids.
Finally, an approximation of hydration status can be obtained by measuring the sensation of thirst with a simple numerical scale. This approach is, however, of limited value in elderly individuals who have a blunted sensation of thirst.

Water Inputs
Water inputs are composed of three major sources (Table 1): the water you drink, the water you eat and the water you produce. The water you drink is essentially composed of water and other liquids with a high water content (85 to >90%). The water you eat comes from various foods with a wide range of water content (40 to >80%). The water you produce results from the oxidation of macronutrients (endogenous or metabolic water).
Table 1. Water balance in sedentary adults living in temperate climate
| Water inputs (ml/day) | Water outputs (ml/day) | ||||||
|---|---|---|---|---|---|---|---|
| Min | Max | Average | Min | Max | Average | ||
| Beverages | 1400a | 1750a | 1575 | Urine | 1200 | 2000 | 1600 |
| Foodsb | 600a | 750a | 675 | Skin | 450 | 450 | 450c |
| Subtotal | 2000d | 2500e | 2250 | Respiration | 250c | 350c | 300 |
| Metabolic water | 250 | 350 | 300 | Faeces | 100 | 300 | 200c |
| Total | 2250 | 2850 | 2550 | Total | 2000 | 3100 | 2550 |
a It is normally assumed that the contribution of food to total dietary water intake is 20–30%, whereas 70–80% are provided by beverages. This relationship is not fixed and depends on the type of beverages and on the choice of foods.
b Foods with a wide range of water content (<40 to >80%).
c (EFSA, 2008) 24.
d Average total water intakes in sedentary women (EFSA, 2008) 24.
e Average total water intakes in sedentary men (EFSA, 2008) 24.
It is normally assumed that the contribution of food to total water intake is 20–30%, whereas 70–80% is provided by beverages. This relationship is not fixed and depends on the type of beverages and on the choice of foods 24.
For an individual at rest under temperate conditions, the volume that might be drunk in a day is on an average 1.5 liters. This has to be adapted according to age, gender, climate and physical activity. The water content of food can vary within a wide range, and consequently the amount of water contributed by foods can vary between 500 ml and 1 liter a day. Endogenous or metabolic water represents about 250–350 ml a day in sedentary people.
The adequate total water intakes for sedentary adults are on an average between 2 and 2.5 liters per day (women and men, respectively) 24. In conclusion, the total water inputs for sedentary adults are on an average between 2 and 3 liters.
So how much fluid does the average, healthy adult living in a temperate climate need ?
The Institute of Medicine determined that an adequate intake (AI) for :
- Men is roughly about 13 cups (3 liters) of total beverages a day.
- The adequate intake for women is about 9 cups (2.2 liters) of total beverages a day.
You don’t need to rely only on what you drink to meet your fluid needs. What you eat also provides a significant portion of your fluid needs. On average, food provides about 20 percent of total water intake. For example, many fruits and vegetables, such as watermelon and spinach, are 90 percent or more water by weight.
Remember that sugary drinks due to their very high added sugar content can lead to weight gain and inflammation, which can increase your risk for developing diseases such as diabetes.
In general, though, drinking too much water resulting in low sodium levels in the blood, a condition called hyponatremia, is rare in healthy adults who eat an average American diet.
Hyponatremia: It’s possible to become overhydrated while exercising. This is caused by low sodium (salt) levels in the blood. It can occur if too much water is drunk over a short period of time. Hyponatremia sometimes affects athletes whose blood sodium level is reduced through sweat and then diluted by drinking large amounts of water. Symptoms of hyponatremia include nausea, vomiting and headache. In serious cases, the brain can swell, causing confusion, seizures, coma and, in rare cases, death.
Water Outputs
The main routes of water loss from the body are kidneys, skin and the respiratory tract and, at a very low level, the digestive system (Table 1).
Over a 24-h period, a sedentary adult produces 1–2 liters of urine.
Water is lost by evaporation through the skin; this is called insensible perspiration because it is an invisible water loss and it represents about 450 ml of water per day in a temperate environment.
Water is also lost by evaporation through the respiratory tract (250–350 ml per day).
Finally, a sedentary adult loses about 200 ml of water a day through faeces.
On an average, a sedentary adult loses 2–3 l of water per day. These water losses through the skin and lungs depend on the climate, air temperature and relative humidity.
When the internal body temperature rises, the only mechanism for increasing heat losses is the activation of sweat glands. Evaporation of water by way of sweat on the skin surface is a very efficient mechanism for removing heat from the body: 2.2 kJ is lost by the evaporation of 1 g of water. When exercising in a hot environment, the sweating rate can reach as much as 1–2 liters of water loss per hour 35. This can lead to dehydration and hyperosmolarity of extracellular fluid (ECF).
It is important to note that sweat is always hypotonic when compared with plasma or extracellular fluid (ECF). Sweat contains 20–50 mmol/l of Na+, whereas the extracellular Na+ concentration is 150 mmol/liter. Intense sweating therefore leads to greater water than electrolyte losses 35. The consequence is an increased extracellular osmolarity that draws water from cells into the ECF. Thus, the loss of water through sweating concerns both intracellular fluid and ECF, a situation that characterizes hypertonic dehydration. The need to drink hypotonic drinks during endurance exercise is well established. A person losing 4 liters of sweat with no fluid replacement loses about 10% of body water, but only 4% of extracellular sodium (about 120 mmol of Na+). This indicates that during exercise, fluid replacement is more important than salt replacement.
Dehydration and hyperosmolarity of extracellular fluid (ECF) can affect consciousness and are involved in the occurrence of heat stroke when internal temperature rises above critical thresholds. The latter can occur when exercising in a warm and humid environment 35.
Regulation of water balance
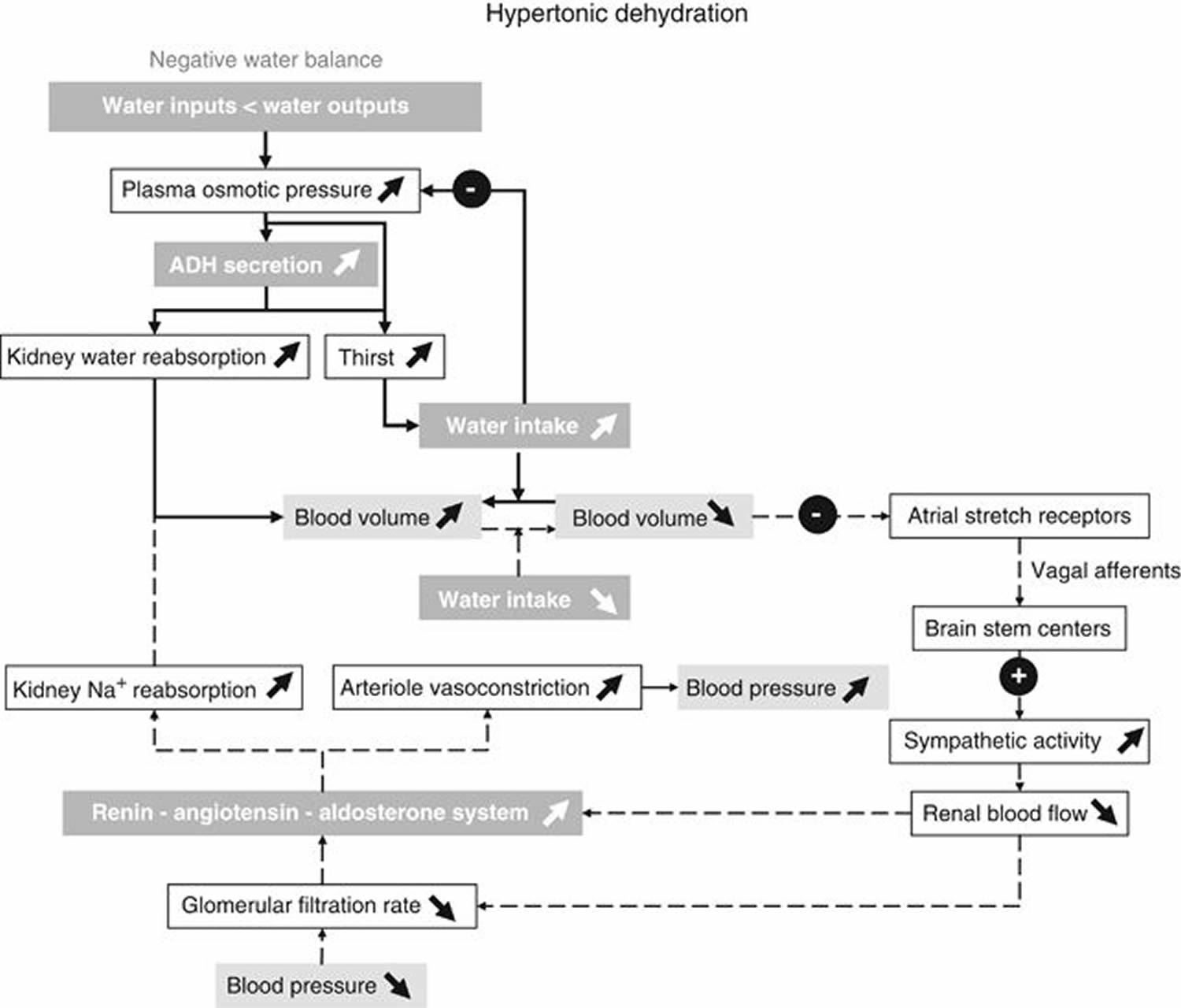
The intake of water is partially determined by thirst. When water losses exceed water intake, the osmotic pressure of extracellular fluid (ECF) increases. By activation of hypothalamic osmoreceptors, an antidiuretic hormone (ADH) is released from the posterior pituitary gland 15. Both the increased ECF osmotic pressure and antidiuretic hormone (ADH) elicit the feeling of thirst (Figure 2). The receptors that elicit thirst have an osmotic threshold higher than the osmoreceptors involved in ADH release. Thus, ADH can act on the kidneys to increase water reabsorption before thirst is elicited. Thirst is often blunted in elderly subjects who are at risk of having an insufficient water intake in conditions of elevated ambient temperature and humidity 36. Thirst is triggered by an increase in plasma and ECF osmolarity, by reductions in plasma volume at water deficits that correspond to a body weight loss of 1–3% 24. During rehydration, thirst can disappear before water balance is reached.
Figure 6. Water balance regulation in your body
[Source 12]Note: Feedback loops for water balance: main perturbations and physiological responses to hypertonic dehydration due to a negative water balance. Solid arrows show the responses induced by osmoreceptors when plasma osmotic pressure increases. Dashed arrows show the corrective mechanisms induced by insufficient water intake and a decreased blood volume to restore blood volume and blood pressure. Note that in the case of hypotonic dehydration due to a positive water balance, all perturbations and physiological responses that are induced occur in the reverse direction.
The set point of plasma osmolarity above which antidiuretic hormone (ADH) secretion is stimulated is about 280 mosm/l. Furthermore, the sensitivity of antidiuretic hormone (ADH) response to a rise in plasma osmolarity is enhanced when the circulating blood volume is lowered 15.
Kidneys are the main regulators of water losses. They have the unique property to modify the osmotic pressure of urine within a large range in response to minute changes in plasma osmotic pressure.
There are two conditions that induce the production of a large volume of urine, and therefore a large water loss. Water diuresis occurs when water is ingested in excess of body requirements. This leads to a small decrease in plasma osmolarity, with a suppression of ADH secretion. As a result, a large volume of hypotonic urine is produced. In contrast, osmotic diuresis results from a filtered load of a solute that exceeds the renal tubules’ maximum reabsorption capacity for this solute.
In summary, both deficit and excess water intakes are counterbalanced by subtle hormonal changes (ADH, aldosterone and atrial natriuretic peptide) that contribute to buffer the deleterious effects of these abnormal conditions. In the end, the final and precise regulation of water balance is dependent on thirst and on ADH release, with its predominant role in water reabsorption in the kidneys (Figure 1). Therefore, voluntary drinking of water is a key behavior for maintaining water balance. Consequently, drinking water before being thirsty is a good habit for maintaining a good body hydration status.
Factors that influence your water needs
You may need to modify your total fluid intake depending on how active you are, the climate you live in, your health status, and if you’re pregnant or breast-feeding.
- Exercise. If you exercise or engage in any activity that makes you sweat, you need to drink extra water to compensate for the fluid loss. An extra 1.5 to 2.5 cups (400 to 600 milliliters) of water should suffice for short bouts of exercise, but intense exercise lasting more than an hour (for example, running a marathon) requires more fluid intake. How much additional fluid you need depends on how much you sweat during exercise, and the duration and type of exercise.
- Intense exercise. During long bouts of intense exercise, it’s best to use a sports drink that contains sodium, as this will help replace sodium lost in sweat and reduce the chances of developing hyponatremia, which can be life-threatening. Also, continue to replace fluids after you’re finished exercising.
- Environment. Hot or humid weather can make you sweat and requires additional intake of fluid. Heated indoor air also can cause your skin to lose moisture during wintertime. Further, altitudes greater than 8,200 feet (2,500 meters) may trigger increased urination and more rapid breathing, which use up more of your fluid reserves.
- Illnesses or health conditions. When you have fever, vomiting or diarrhea, your body loses additional fluids. In these cases, you should drink more water. In some cases, your doctor may recommend oral rehydration solutions, such as Gatorade, Powerade or CeraLyte. You may also need increased fluid intake if you develop certain conditions, including bladder infections or urinary tract stones. On the other hand, some conditions, such as heart failure and some types of kidney, liver and adrenal diseases, may impair excretion of water and even require that you limit your fluid intake.
- Pregnancy or breast-feeding. Women who are pregnant or breast-feeding need additional fluids to stay hydrated. Large amounts of fluid are used especially when nursing. The Institute of Medicine recommends that pregnant women drink about 10 cups (2.3 liters) of fluids daily and women who breast-feed consume about 13 cups (3.1 liters ) of fluids a day.
If you think you are not getting enough water, these tips may help:
- Carry a water bottle for easy access when you are at work of running errands.
- Freeze some freezer safe water bottles. Take one with you for ice-cold water all day long.
- Choose water instead of sugar-sweetened beverages. This can also help with weight management. Substituting water for one 20-ounce sugar sweetened soda will save you about 240 calories. For example, during the school day students should have access to drinking water, giving them a healthy alternative to sugar-sweetened beverages.
- Choose water when eating out. Generally, you will save money and reduce calories.
- Add a wedge of lime or lemon to your water. This can help improve the taste and help you drink more water than you usually do.
Please Continue To Part 2 of Dehydration: https://healthjade.com/symptoms-signs-dehydration-part-2/
References- Leaf A. Dehydration in the elderly. N Engl J Med 311: 791–792, 1984. http://www.nejm.org/doi/full/10.1056/NEJM198409203111209
- Hole’s Essentials of Human Anatomy and Physiology 13th edition. Published by McGraw-Hill Education, 2 Penn Plaza, New York, NY 10121.
- Phillips PA, Rolls BJ, Ledingham JGG, Forsling ML, Morton JJ, Crowe MB, and Wollner L. Reduced thirst after water deprivation in healthy elderly men. N Engl J Med 311: 753–759, 1984. https://www.ncbi.nlm.nih.gov/pubmed/6472364
- Effect of water deprivation on cognitive-motor performance in healthy men and women. American Journal of Physiology – Regulatory, Integrative and Comparative Physiology Jul 2005, 289 (1) R275-R280; DOI: 10.1152/ajpregu.00501.2004. http://ajpregu.physiology.org/content/289/1/R275.long
- Ekblom B, Greenleaf CJ, Greenleaf JE, and Hermansen L. Temperature regulation during exercise dehydration in man. Acta Physiol Scand 79: 475–483, 1970. https://www.ncbi.nlm.nih.gov/pubmed/5472114
- Kleiner SM. Water: an essential but overlooked nutrient. J Am Diet Assoc 99: 200–206, 1999. https://www.ncbi.nlm.nih.gov/pubmed/9972188
- Weinberg AD, Minaker KL, and the Council of Scientific Affairs AMA. Dehydration. JAMA 274: 1552–1556, 1995. https://www.ncbi.nlm.nih.gov/pubmed/7474224
- Holtzhausen LM, Noakes TD, Kroning B, De Klerk M, Roberts M, and Emsley R. Clinical and biochemical characteristics of collapsed ultramarathon runners. Med Sci Sports Exerc 26: 1095–1101, 1994. https://www.ncbi.nlm.nih.gov/pubmed/7808242
- Nielsen B. Effects of changes in plasma volume and osmolarity on thermoregulation during exercise. Acta Physiol Scand 90: 725–730, 1974. https://www.ncbi.nlm.nih.gov/pubmed/4832088
- Noakes TD, Adams BA, Myburgh KH, Greeff C, Lotz T, and Nathan M. The danger of inadequate water intake during prolonged exercise. Eur J Appl Physiol 57: 210–219, 1988.
- NHS. Dehydration – Introduction. http://www.nhs.uk/Conditions/Dehydration/Pages/Introduction.aspx
- Water as an essential nutrient: the physiological basis of hydration. European Journal of Clinical Nutrition (2010) 64, 115–123; doi:10.1038/ejcn.2009.111; published online 2 September 2009. http://www.nature.com/ejcn/journal/v64/n2/full/ejcn2009111a.html
- Dietary Reference Intakes (2006). The Essential Guide to Nutrients Requirements. Institute of Medicine of the National Academies: Washington DC, 543 pp.
- Wang ZM, Deurenberg P, Wang W, Pietrobelli A, Baumgartner RN, Heymsfield SB (1999). Hydration of fat-free body mass: a review and critique of a classic body-composition constant. Am J Clin Nutr 69, 833–841. https://www.ncbi.nlm.nih.gov/pubmed/10232621
- Ganong WF (2005). Review of Medical Physiology, 22nd edn, LANGE-Science: New York.
- Jequier E, Constant F. Water as an essential nutrient: the physiological basis of hydration. Eur J Clin Nutr. 2010;64(2):115–123. http://www.nature.com/ejcn/journal/v64/n2/full/ejcn2009111a.html
- Lang F, Waldegger S (1997). Regulating cell volume. Am Scientist 85, 456–463.
- Häussinger D (1996). The role of cellular hydration in the regulation of cell function. Biochem J 313, 697–710. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1216967/
- Grandjean AC, Campbell SM (2004). Hydration: Fluids for Life. A monograph by the North American Branch of the International Life Science Institute. ILSI North America: Washington DC.
- Ritz P, Berrut G (2005). The importance of good hydration for day-to-day health. Nutr Rev 63, S6–S13. https://academic.oup.com/nutritionreviews/article-abstract/63/suppl_1/S6/1927737/The-Importance-of-Good-Hydration-for-Day-to-Day
- Szinnai G, Schachinger H, Arnaud MJ, Linder L, Keller U (2005). Effect of water deprivation on cognitive-motor performance in healthy men and women. Am J Physiol Regul Integr Comp Physiol 289, R275–R280. http://ajpregu.physiology.org/content/289/1/R275.long
- Parikh C, Berl T: Disorders of Water Metabolism. In: Floege J, Johnson RJ, Feehally J, eds. Comprehensive Clinical Nephrology. 4th edition ed. Philadelphia, Elsevier, 2010.
- U.S. National Library of Medicine. Fluid and Electrolyte Balance. https://medlineplus.gov/fluidandelectrolytebalance.html
- EFSA (2008). Draft dietary reference values for water. Scientific Opinion of the Panel on Dietetic Products, Nutrition and Allergies.
- Grandjean AC, Campbell SM (2004). Hydration: Fluids for Life. A monograph by the North American Branch of the International Life Science Institute. ILSI North America: Washington DC.
- Francesconi RP, Hubbard RW, Szlyk PC, Schnakenberg D, Carlson D, Leva N et al. (1987). Urinary and hematologic indexes of hypohydration. J Appl Physiol 62, 1271–1276. https://www.ncbi.nlm.nih.gov/pubmed/3571082
- Armstrong LE (2005). Hydration assessment techniques. Nut Rev 63, S40–S54. https://academic.oup.com/nutritionreviews/article-abstract/63/suppl_1/S40/1927763/Hydration-Assessment-Techniques
- Shirreffs SM (2003). Markers of hydration status. Eur J Clin Nutr 57 (Suppl 2), S6–S9. https://www.ncbi.nlm.nih.gov/pubmed/14681707
- Grandjean AC, Reimers CJ, Buyckx ME (2003). Hydration: issues for the 21st century. Nutr Rev 61, 261–271. https://www.ncbi.nlm.nih.gov/pubmed/13677588
- Pialoux V, Mischler I, Mounier R, Gachon P, Ritz P, Coudert J et al. (2004). Effect of equilibrated hydration changes on total body water estimates by bioelectrical impedance analysis. Br J Nutr 91, 153–159. https://www.ncbi.nlm.nih.gov/pubmed/14748949
- Mathie JR (2005). Second generation mixture theory equation for estimating intracellular water using bioimpedance spectroscopy. J Appl Physiol 99, 780–781. http://jap.physiology.org/content/99/2/780
- Oppliger RA, Magnes SA, Popowski LA, Gisolfi CV (2005). Accuracy of urine specific gravity and osmolality as indicators of hydration status. Int J Sport Nutr Exerc Metab 15, 236–251. https://www.ncbi.nlm.nih.gov/pubmed/16131695
- Armstrong LE, Soto JA, Hacker Jr FT, Casa DJ, Kavouras SA, Maresh CM (1998). Urinary indices during dehydration, exercise, and rehydration. Int J Sport Nutr 8, 345–355. https://www.ncbi.nlm.nih.gov/pubmed/9841955
- Mentes JC, Wakefield B, Culp K (2006). Use of a urine color chart to monitor hydration status in nursing home residents. Biol Res Nurs 7, 197–203. https://www.ncbi.nlm.nih.gov/pubmed/16552947
- Sawka MN, Cheuvront SN, Carter III R (2005). Human water needs. Nutr Rev 63, S30–S39. https://www.ncbi.nlm.nih.gov/pubmed/16028570
- Phillips PA, Rolls BJ, Ledingham JG, Forsling ML, Morton JJ, Crowe MJ et al. (1984). Reduced thirst after water deprivation in healthy elderly men. N Engl J Med 311, 753–759. https://www.ncbi.nlm.nih.gov/pubmed/6472364