What is Tetralogy of Fallot
Tetralogy of Fallot is a birth defect of the heart, a type of cyanotic (low body oxygen) heart defect consisting of 4 specific abnormalities affecting about 1 in every 2518 babies born in the United States each year. The Centers for Disease Control and Prevention (CDC) estimates that each year about 1,660 babies in the United States are born with tetralogy of Fallot 1, making Tetralogy of Fallot the most common cyanotic congenital heart defect. Tetralogy of Fallot is classified as a cyanotic heart defect because the condition leads to cyanosis, a bluish-purple coloration to the skin, and shortness of breath due to low oxygen levels in the blood. Surgery to repair the defects in the heart is usually performed between 3 and 5 years old. In more severe forms of Tetralogy of Fallot, surgery may be indicated soon after birth – this birth defect is considered a critical congenital heart defect. In most cases the heart can be surgically corrected and the outcome is good.
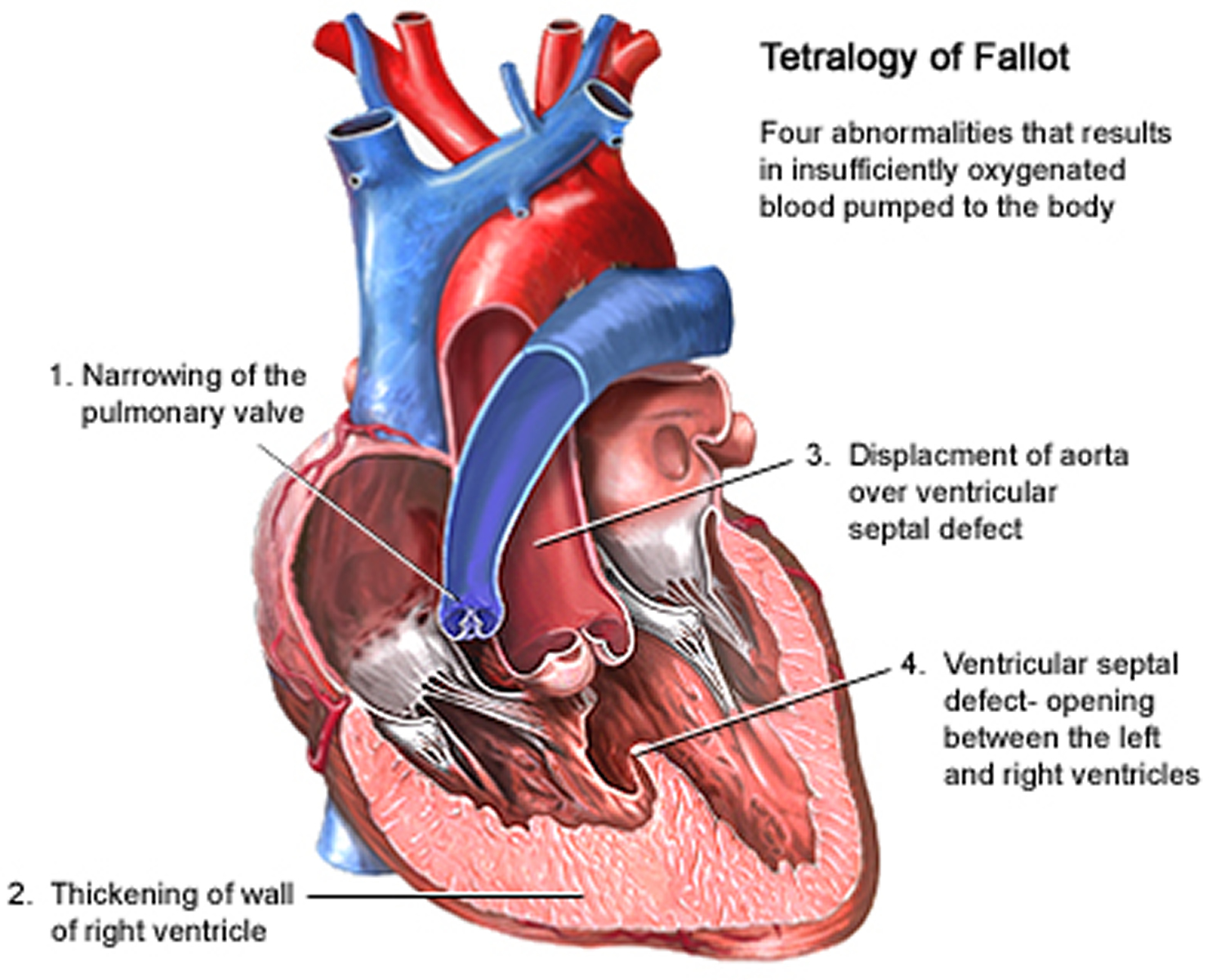
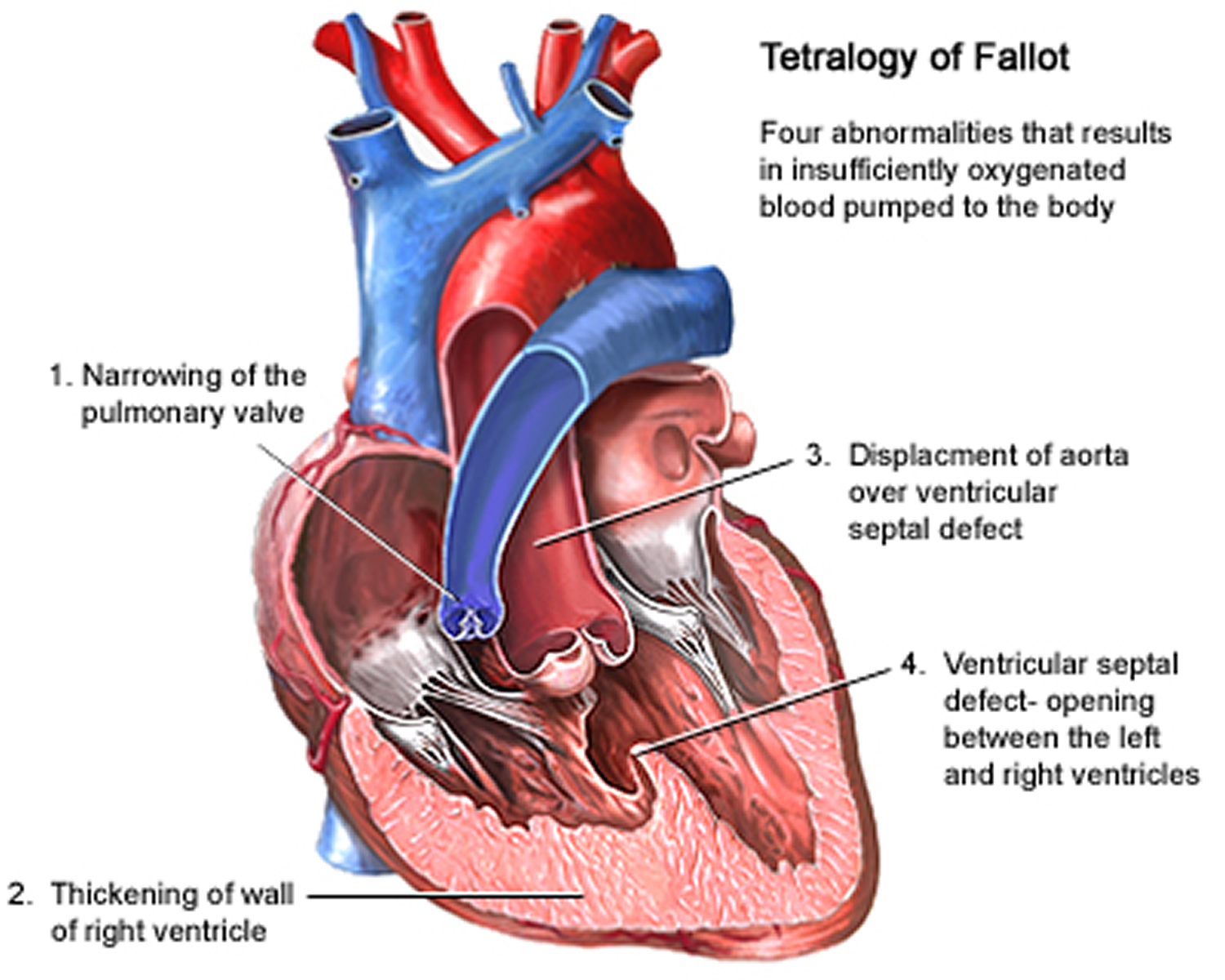
Tetralogy of Fallot is made up of the following four defects of the heart and its blood vessels (see Figure 7 below):
- Ventricular Septal Defect (VSD) – a hole between the left and right ventricle
- Pulmonary Stenosis – a narrowing of the pulmonary valve and main pulmonary artery. This condition also is called pulmonary stenosis.
- Right Ventricular Hypertrophy – where the muscle of the right ventricle is thickened
- Displaced Aorta – the aortic valves, which opens to the aorta, is enlarged and seems to open from both ventricles, rather than from the left ventricle only, as in a normal heart. In this defect, the aortic valve sits directly on top of the ventricular septal defect.
As a result of this combination of defects, oxygenated and non-oxygenated blood mixes, causing the overall amount of oxygen in the blood to be lower than normal. This may cause the baby to appear blue (known as cyanosis) at times.
Although tetralogy refers to four abnormalities, there are really only two main abnormalities (items 1 and 2 below). The other abnormalities are consequences of the process leading to points 1 and 2:
- A hole in the wall dividing the two main pumping chambers (ventricles) called a ventricular septal defect (VSD), and
- A narrowing of the pulmonary valve and main pulmonary artery. This condition also is called pulmonary stenosis. A narrow passage leaving the right ventricle in association with a small and abnormal valve at the end of that passage this partly blocks blood flow to the lung.
- The muscular wall of the lower right chamber of the heart (right ventricle) is thicker than normal. This also is called ventricular hypertrophy.
- The aorta overrides (leaves the heart in an unusual position) the ventricular septal defect (VSD).
As the aorta overlies the ventricular septal defect (VSD), it receives blood from the right ventricle and the left ventricle. This causes circulation of deoxygenated blood and central cyanosis (bluish discoloration). The high right ventricular pressure, caused by the outflow obstruction, facilitate this. Because less blood goes through the pulmonary circulation, cyanosis is independant of pulmonary hypertension.
Most babies with Tetralogy of Fallot have oxygen levels in a very safe zone for many weeks before a gradual reduction in oxygen level occurs due to slowly increasing obstruction between the heart and pulmonary artery. Sometimes the obstruction can occur in a much shorter time frame minutes. These sudden episodes of bluish skin during crying or feeding are temporary and these episodes are called “tet spells.” While they rarely cause any permanent problem, they are an important sign to show that the condition may be reaching a point where treatment is necessary.
It can be a little confusing trying to tell if your baby is just changing color normally or if a “tet spell” is occurring. It is normal for any baby to change color and become blue when crying or cold and this can be more noticeable in babies who already have lower than normal oxygen levels. In the “normal” color change the baby (1) appears well and (2) changes color only during the event (e.g., crying or feeding) the color returns to normal almost as soon as the crying stops. This is not a spell and not a problem it is normal.
Tetralogy of Fallot with pulmonary atresia: Fetal echocardiogram
3VT view demonstrates reverse filling of the duct, a key feature differentiating this subtype from the more common Tetralogy of Fallot with pulmonary stenosis.
Note that the duct is on the right side of the image, as the fetus is in breech position.
There are a number of different subtypes of tetralogy of Fallot: Tetralogy of Fallot with pulmonary stenosis (75%), Tetralogy of Fallot with pulmonary atresia (20%) and Dysplastic (absent/ring) pumonary valve syndrome (5%).
This case demonstrates the typical findings on the standard fetal echo views in a case of tetralogy of Fallot with pulmonary atresia. The key features that differentiate this from the more common pulmonary stenosis subtype are the absence of the pulmonary trunk on the RVOT view and reverse flow in the ductus appreciated on the 3VT view.
Tetralogy of Fallot with absent pulmonary valve syndrome: fetal echocardiogram. Age: Second trimester
Tet Spells are usually:
- Accompanied by deep blueness, greyness or pallor which persists for several minutes after the baby stops crying, and
- The baby can seem lethargic, floppy or unwell.
These can be best treated by simply picking up and cuddling your baby with the baby’s knees tucked up against the chest. The color generally returns to normal quickly as your baby settles. This is not an emergency, but it is a good idea to let your cardiologist know at the first convenient time in the next day or two so this can be factored into treatment planning.
It is very rare for spells to be really dangerous, but if you are worried that your baby is not returning to normal after a few minutes it is usually safest to call an ambulance.
Infants with tetralogy of Fallot or other conditions causing cyanosis can have problems including:
- A higher risk of getting an infection of the layers of the heart, called endocarditis.
- A higher risk of having irregular heart rhythms, called arrhythmia.
- Dizziness, fainting, or seizures, because of the low oxygen levels in their blood.
- Delayed growth and development.
Tetralogy of Fallot is repaired with open-heart surgery, either soon after birth or later in infancy. The timing of the surgery will depend on how narrow the pulmonary artery is. The goal of surgery is to repair the four defects of tetralogy of Fallot so the heart can work as normally as possible. Repairing the defects can greatly improve a child’s health and quality of life. Some teenagers or adults who had tetralogy of Fallot repaired in childhood need additional surgery to correct heart problems that develop over time.
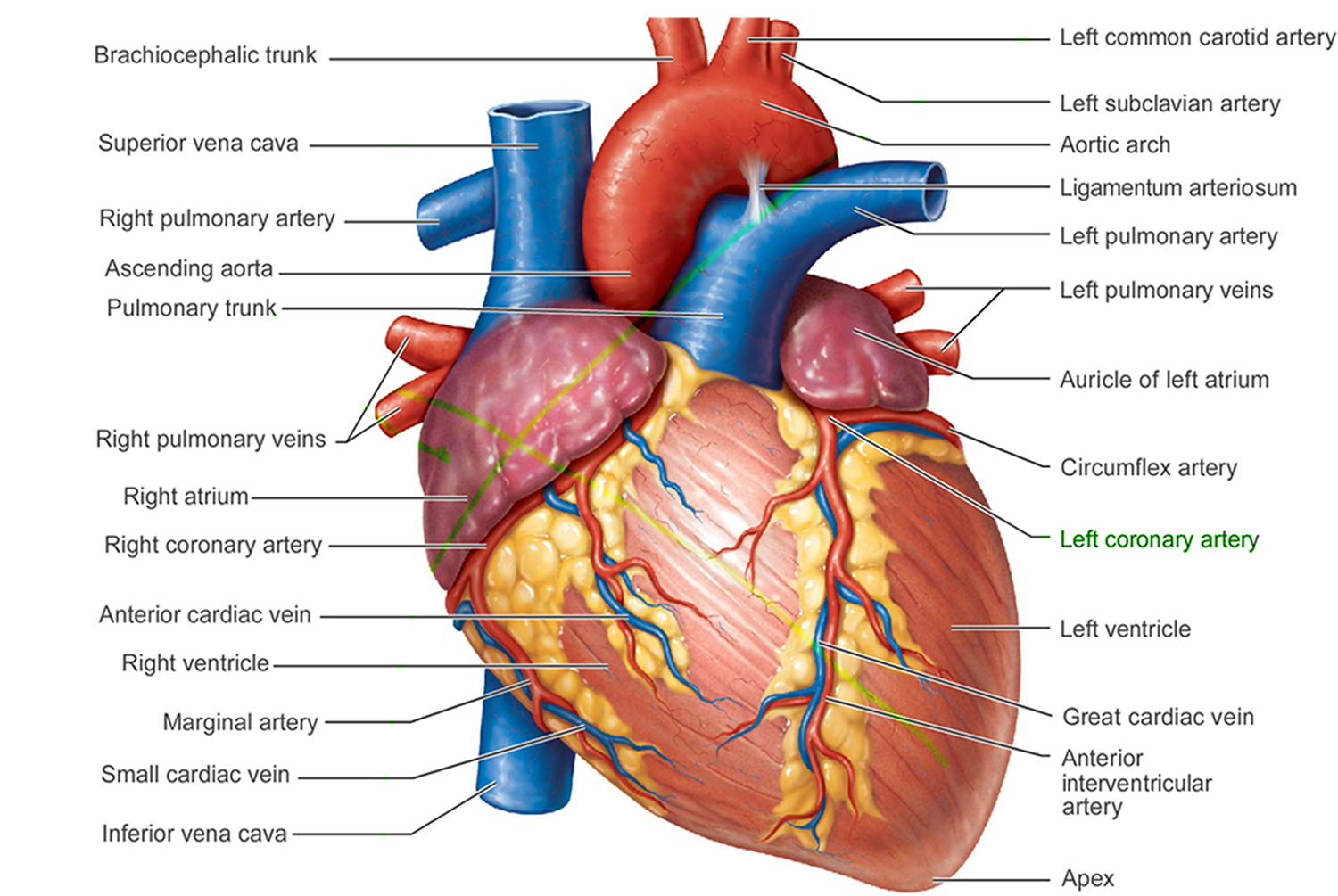
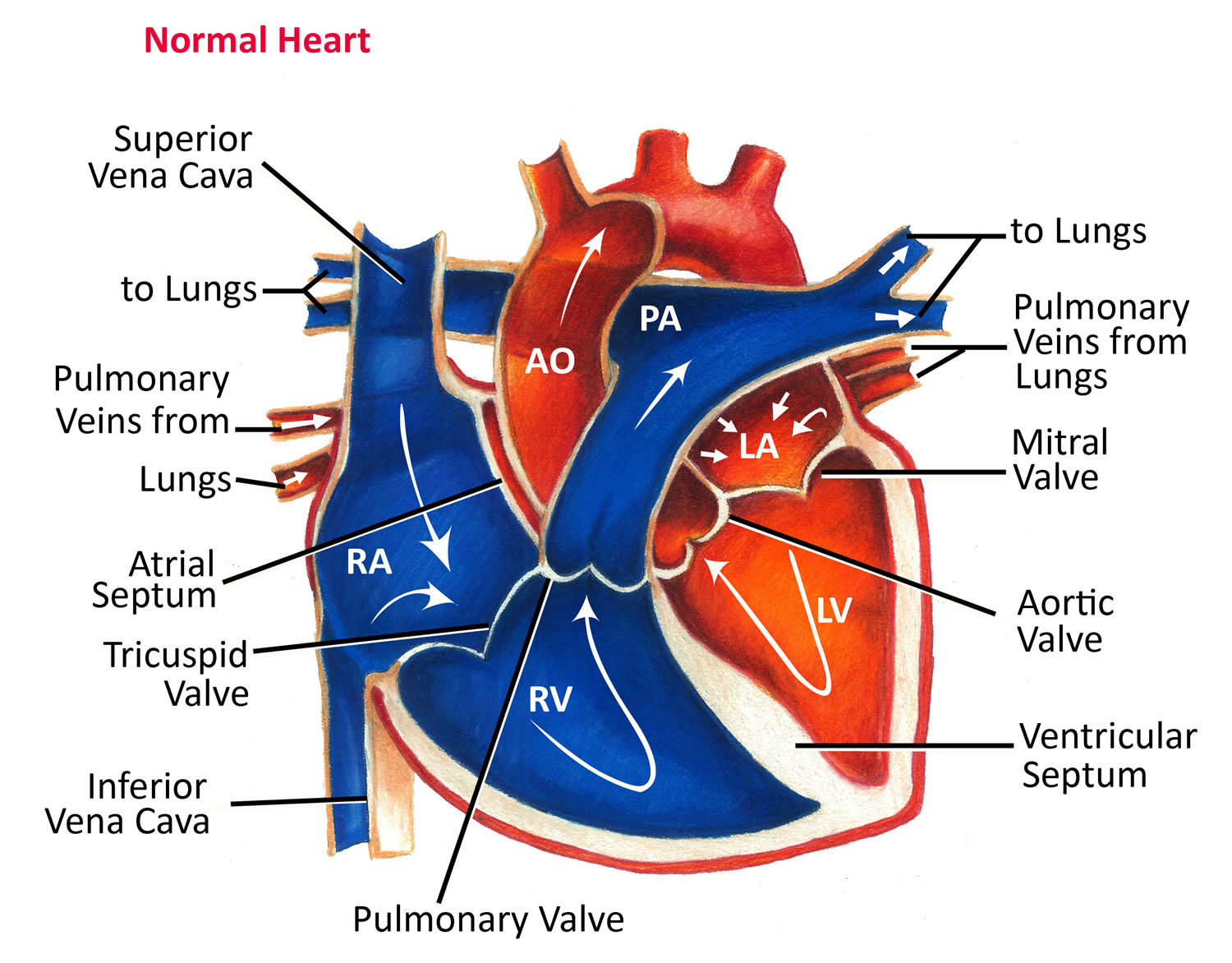
Figure 1. The anatomy of the heart
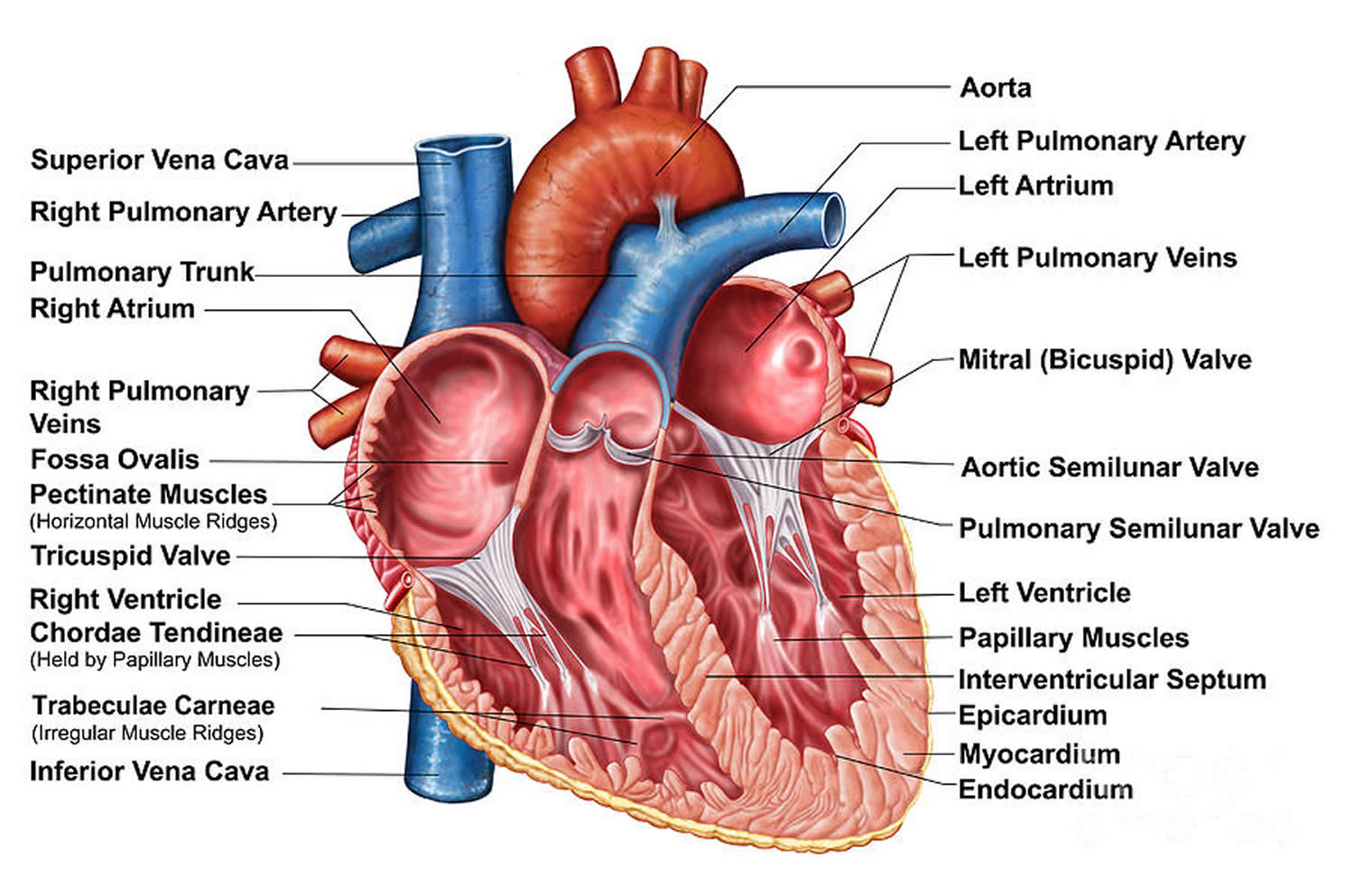
Figure 2. The anatomy of the heart chambers
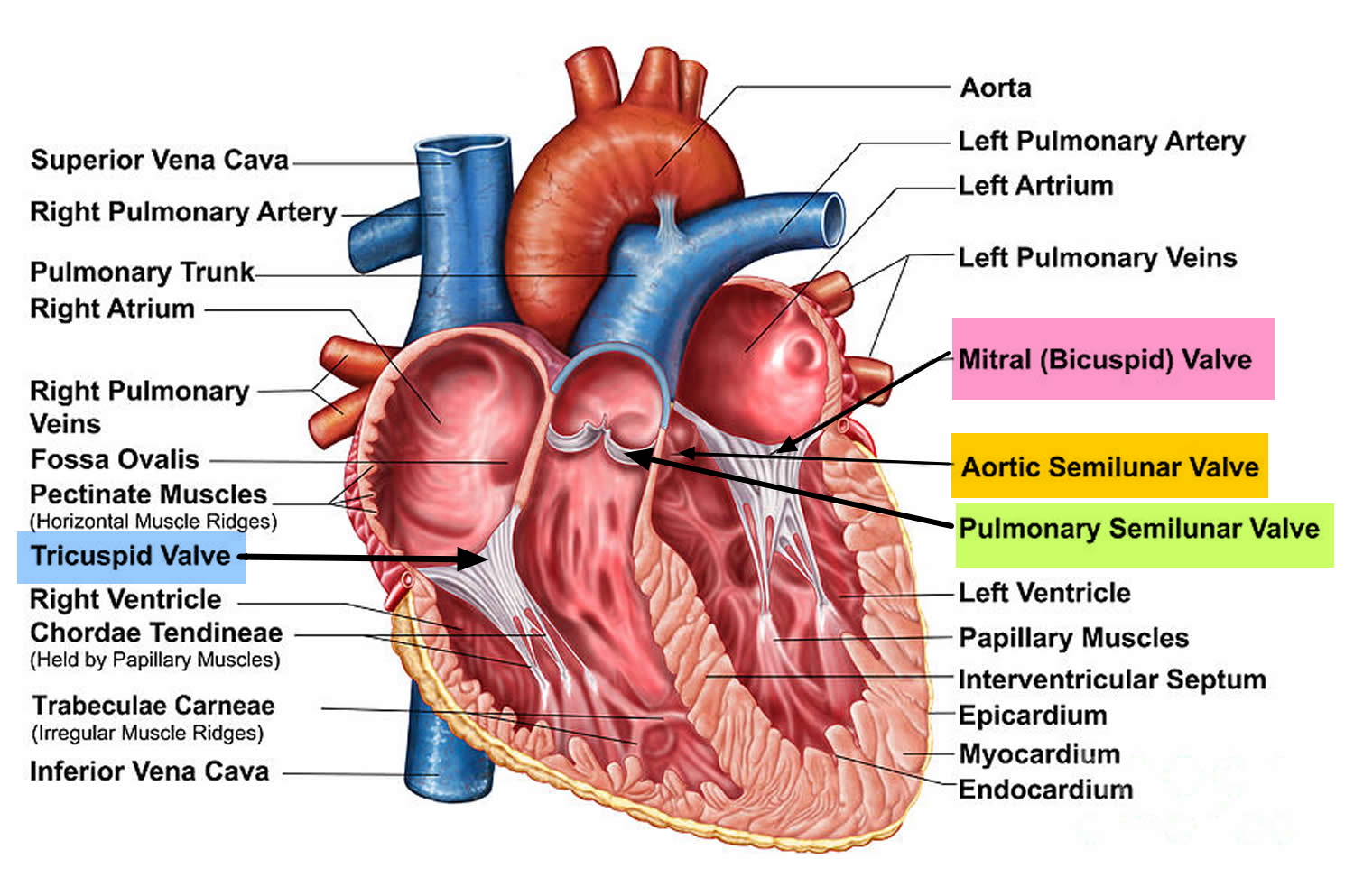
Figure 3. The anatomy of the heart valves
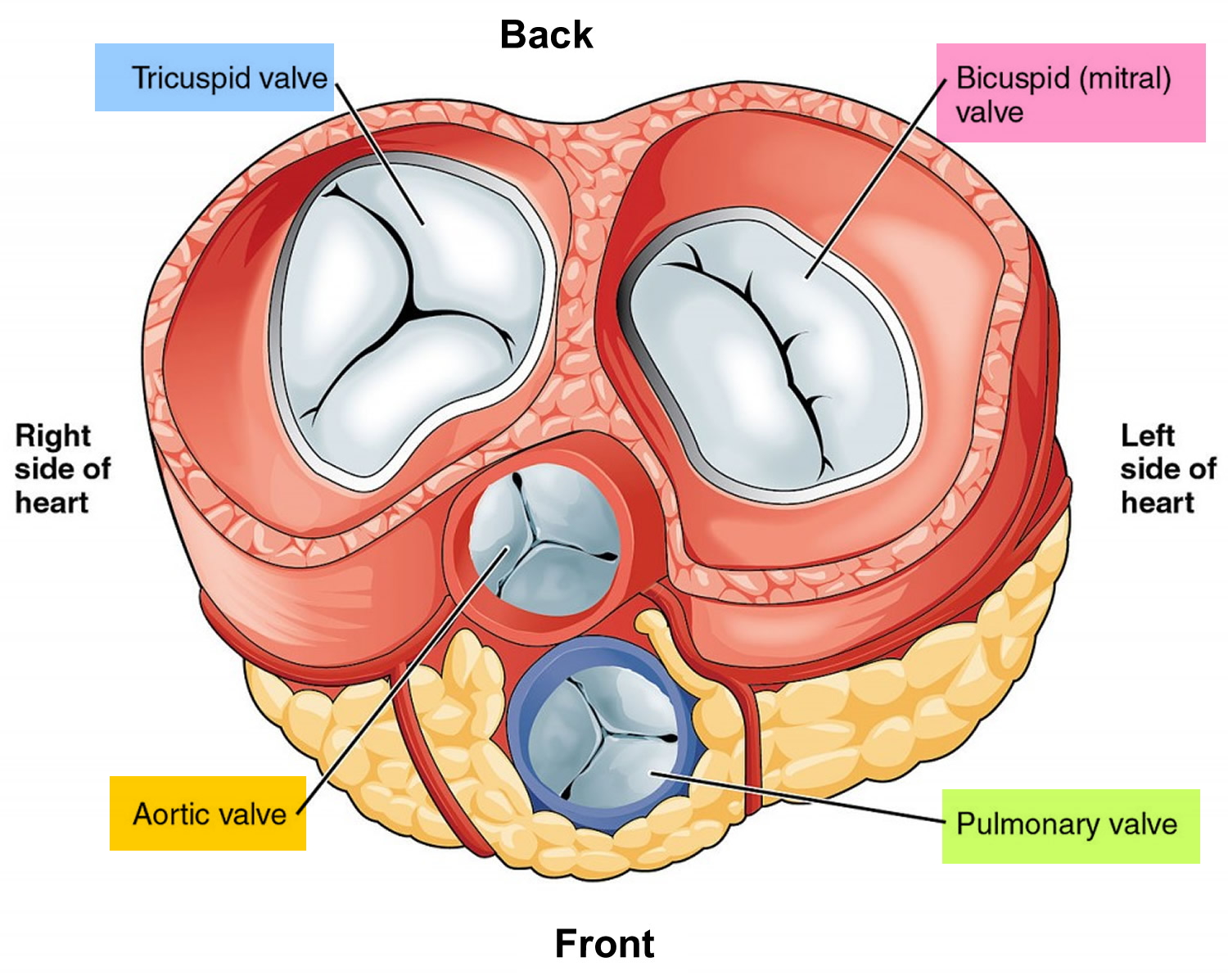
Figure 4. Top view of the 4 heart valves
Figure 5. Normal heart blood flow
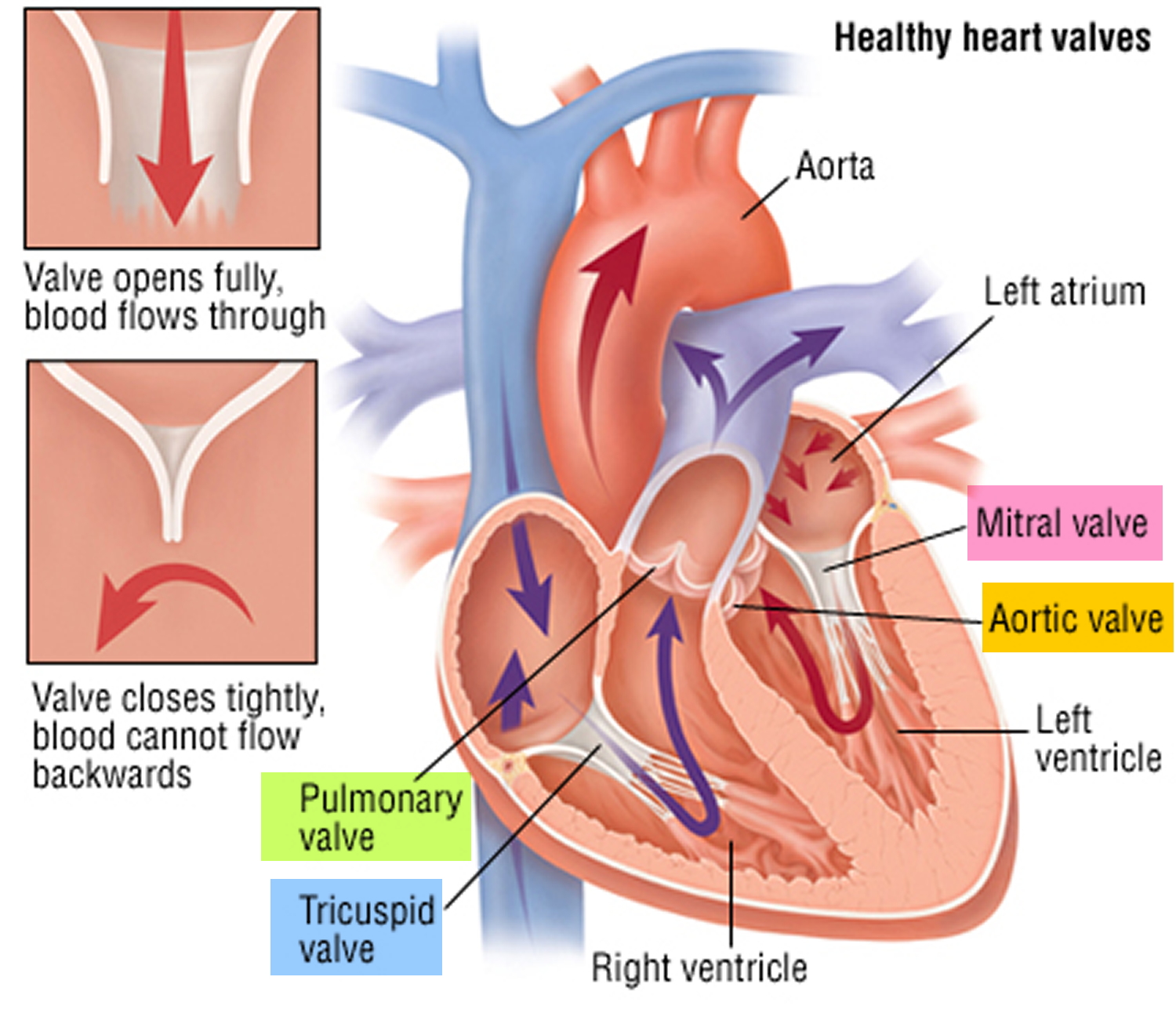
Figure 6. Heart valves function (normal)
Tetralogy of Fallot life expectancy
Over the past few decades, the diagnosis and treatment of tetralogy of Fallot have greatly improved. Most children who have this heart defect survive to adulthood. However, they’ll need lifelong medical care from specialists to help them stay as healthy as possible.
Caring for Your Child at Home
Feeding and Nutrition
Babies who have tetralogy of Fallot can tire while nursing or feeding. Small, frequent meals may be easier for your baby to handle.
Your child also may need extra nutrition. A supplement or an extra feeding can give the baby more calories, vitamins, or iron. Your child’s doctors will work with you to decide whether your baby needs extra nutrition.
Tet Spells
“Tet spells” can occur in babies whose tetralogy of Fallot hasn’t yet been repaired. Lowering your baby’s anxiety or stress can help prevent tet spells and save the baby’s energy. For example, slowly picking up your baby and speaking in a soothing voice can avoid startling him or her, which may prevent or reduce crying.
Ask your doctor how you can manage your child’s tet spells. Your doctor may suggest that you:
- Bring the child’s knees up tight against his or her chest (this is called the knee–chest position) or have your child squat down. This will increase blood flow to the lungs.
- Try to calm your child.
- Call your local emergency number for an ambulance if the symptoms don’t improve right away.
Activity Restrictions
If your child was born with tetralogy of Fallot, he or she may have some physical activity limits. The limits vary with each child. Ask your child’s doctor whether:
- Your child needs to restrict activity or exercise
- Your child can play in organized sports, especially contact sports
- You need a note for your child’s school or coaches about limiting your child’s exercise
Ongoing Medical Care
Children who have tetralogy of Fallot should have ongoing medical care. This includes making sure your child:
- Sees a pediatric cardiologist for heart checkups as directed
- Sees a pediatrician or family health care provider for routine exams
- Takes medicines as prescribed
Children who have severe heart defects, like tetralogy of Fallot, may be at slightly increased risk for infective endocarditis. Infective endocarditis is a serious infection of the inner lining of the heart chambers and valves.
Your child’s doctor or dentist may give your child antibiotics before some medical or dental procedures (such as surgery or dental cleanings) that can allow bacteria into the bloodstream. Your child’s doctor will tell you whether your child needs to take antibiotics before such procedures.
To reduce the risk of infective endocarditis, gently brush your young child’s teeth every day as soon as they begin to come in. As your child gets older, make sure he or she brushes every day and sees a dentist regularly. Talk with your child’s doctor and dentist about how to keep your child’s mouth and teeth healthy.
Consider having your child wear a medical ID bracelet or necklace. This alerts anyone caring for your child that the child has a heart defect.
Special Needs for Teenagers and Adults
As children who have heart defects grow up and become teens, they should learn how their hearts differ from normal hearts. They also should know what kind of defects they have, how they were treated, and what type of care is still needed. They should be able to recognize signs and symptoms and know how to respond.
Work with your child’s health care providers to compile a packet of medical records and information that covers all aspects of your child’s heart defect, including:
- Diagnosis
- Procedures or surgeries
- Prescribed medicines
- Recommendations about medical followup and how to prevent complications
- Health insurance
Review your current health insurance plan so you understand your coverage. Keeping your health insurance current is important. If you plan to change jobs, find out whether your new health insurance will cover care for your child’s congenital heart defect.
Transition of Care
The move from pediatric care to adult care is an important step in treatment. Talk with your teen’s health care team about creating a plan to help your teen transition to adult care. Start planning as soon as your teen is able and willing to fully take part in this process.
Following a transition plan has many benefits. It will help your teen:
- Get used to talking with health care providers
- Learn about the adult health care system
- Understand the importance of having health insurance and learn what his or her insurance covers
- Take responsibility for his or her medical care
A transition plan also can help your teen think about other important issues, such as future education and employment, birth control and pregnancy planning, and making healthy choices about nutrition, physical activity, and other lifestyle habits.
Additional Surgery and Other Complications
Some teenagers or young adults need additional surgery. For example, the pulmonary valve can narrow again over time, reducing blood flow. A surgeon may need to widen or replace the valve. The cardiologist will discuss with you and your teenager the need for any additional heart surgeries.
Over time, people who have had surgery to repair tetralogy of Fallot also may face other heart problems.
Leaking heart valves. The heart has four valves that open and close with each heartbeat. These valves ensure that blood flows only in one direction.
If a valve doesn’t seal tightly, blood can leak back into the chamber it came from. This is called backflow or regurgitation and it can lead to symptoms and complications.
The most frequent problem that occurs after tetralogy of Fallot repair is pulmonary backflow, or leaking from the pulmonary valve. Backflow from the tricuspid valve and aortic valve also can occur.
Surgery is used to repair or replace the leaking valve. In some cases, catheter-based procedures may be used to replace leaking valves.
Arrhythmias. Arrhythmias are another complication that may occur. Arrhythmias are problems with the rate or rhythm of the heartbeat.
Arrhythmias associated with tetralogy of Fallot include ventricular tachycardia, atrial fibrillation, and atrial flutter. For more information, go to the Health Topics Arrhythmia article.
Doctors use medicines to control these arrhythmias. They also may recommend medical procedures or surgery to treat arrhythmias.
Pulmonary artery branch stenosis. Over time, the pulmonary valve can narrow again. This will reduce blood flow to the lungs, making the heart work harder than it should. Several surgical techniques can be used to fix this problem.
Right ventricular aneurysms. The patch used to fix a ventricular septal defect (VSD) can cause areas of the ventricle to weaken. These areas, called aneurysms, can bulge or “balloon” out. Aneurysms make it hard for the heart to work as well as it should. Aneurysms are repaired with surgery.
Residual ventricular septal defects. Some VSDs still leak, even after they’ve been repaired. VSDs are repaired again if they’re large or are causing problems with the function of the right ventricle.
Coronary heart disease. As people who have repaired tetralogy of Fallot approach middle age, they can develop coronary heart disease (coronary artery disease). Adults who don’t have congenital heart defects also can develop coronary heart disease.
Coronary heart disease is a condition in which a waxy substance called plaque builds up in the coronary (heart) arteries. Coronary heart disease can lead to chest pain, shortness of breath, and heart attack.
For people who have repaired tetralogy of Fallot, preventing coronary heart disease is important. The procedures used to treat coronary heart disease, such as coronary artery bypass grafting, can cause problems for these people.
Other Considerations
Many women with repaired tetralogy of Fallot who become pregnant are able to have successful, full-term pregnancies. Others may have difficult pregnancies.
Women with tetralogy of Fallot who want to become pregnant (or who are pregnant) should talk with their doctors about:
- Health risks during pregnancy
- Medicines they can take during pregnancy
- Any new or worsening symptoms
If possible, these women should consult specialists who take care of pregnant women who have heart conditions, such as congenital heart defects.
Adults who were born with tetralogy of Fallot should consider job changes carefully, because health benefits may change. Some health plans have waiting periods or clauses to exclude some types of coverage. Before making any job changes, find out whether the change will affect your health insurance.
Several laws protect the employment rights of people who have health conditions, such as congenital heart defects. The Americans with Disabilities Act and the Work Incentives Improvement Act try to ensure fairness in hiring for all people, including those who have health conditions.
Tetralogy of Fallot prognosis
With the advancement of surgical technology and increased medical management, the prognosis for Tetrlogy of Fallot is quite good. Over 95% of patients with simple Tetralogy of Fallot go on to live beyond 20 years of age.
Tetralogy of Fallot complications
All babies who have tetralogy of Fallot need corrective surgery. Without treatment, your baby might not grow and develop properly.
Your baby may also be at an increased risk of serious complications, such as infective endocarditis — an infection of the inner lining of the heart or heart valve caused by a bacterial infection.
Untreated cases of tetralogy of Fallot usually develop severe complications over time, which might result in death or disability by early adulthood.
Tetralogy of Fallot causes
The causes of heart defects (such as tetralogy of Fallot) among most babies are unknown. Some babies have heart defects because of changes in their genes or chromosomes. Heart defects such as tetralogy of Fallot also are thought to be caused by a combination of genes and other risk factors, such as the things the mother or fetus come in contact with in the environment or what the mother eats or drinks or the medicines she uses.
Risk Factors for Tetralogy of Fallot
The aetiology (origins) of congenital heart defects is not understood but several factors are known to be associated:
- (1) Maternal drug abuse, alcohol abuse and radiation exposure.
- (2) Maternal infection during pregnancy particularly rubella (German measles) and some other viral illnesses
- (3) Genetic abnormalities.
- (4) Chromosomal abnormalities (septal defects are common with Trisomy 21- Down’s syndrome and DiGeorge syndrome)
- (5) Age (being older than 40)
- (6) Diabetes
- (7) Poor nutrition during pregnancy
- (8) A parent who has tetralogy of Fallot
Heredity may play a role in causing tetralogy of Fallot. An adult who has tetralogy of Fallot may be more likely than other people to have a baby with the condition.
Children who have certain genetic disorders, such as Down syndrome and DiGeorge syndrome, often have congenital heart defects, including tetralogy of Fallot.
Researchers continue to search for the causes of tetralogy of Fallot and other congenital heart defects.
Tetralogy of Fallot symptoms
Cyanosis is an important sign of tetralogy of Fallot. Cyanosis is a bluish tint to the skin, lips, and fingernails. Low oxygen levels in the blood cause cyanosis.
Babies who have unrepaired tetralogy of Fallot sometimes have “tet spells.” These spells happen in response to an activity like crying or having a bowel movement.
A tet spell occurs when the oxygen level in the blood suddenly drops. This causes the baby to become very blue. The baby also may:
- Have a hard time breathing
- Become very tired and limp
- Not respond to a parent’s voice or touch
- Become very fussy
- Pass out
In years past, when tetralogy of Fallot wasn’t treated in infancy, children would get very tired during exercise and could faint. Now, doctors repair tetralogy of Fallot in infancy to prevent these symptoms.
Another common sign of tetralogy of Fallot is a heart murmur. A heart murmur is an extra or unusual sound that doctors might hear while listening to the heart.
The sound occurs because the heart defect causes abnormal blood flow through the heart. However, not all heart murmurs are signs of congenital heart defects. Many healthy children have heart murmurs.
Babies who have tetralogy of Fallot may tire easily while feeding. Thus, they may not gain weight or grow as quickly as children who have healthy hearts. Also, normal growth depends on a normal workload for the heart and normal flow of oxygen-rich blood to all parts of the body.
Children who have tetralogy of Fallot also may have clubbing. Clubbing is the widening or rounding of the skin or bone around the tips of the fingers.
Tetralogy of Fallot diagnosis
Tetralogy of Fallot may be diagnosed during pregnancy or soon after a baby is born.
Doctors diagnose tetralogy of Fallot based on a baby’s signs and symptoms, a physical exam, and the results from tests and procedures.
Signs and symptoms of the heart defect usually occur during the first weeks of life. Your infant’s doctor may notice signs or symptoms during a routine checkup. Some parents also notice cyanosis or poor feeding and bring the baby to the doctor. (Cyanosis is a bluish tint to the skin, lips, and fingernails.)
Specialists Involved
If your child has tetralogy of Fallot, a pediatric cardiologist and pediatric cardiac surgeon may be involved in his or her care.
A pediatric cardiologist is a doctor who specializes in diagnosing and treating heart problems in children. Pediatric cardiac surgeons repair children’s heart defects using surgery.
Physical Exam
During a physical exam, the doctor may:
- Listen to your baby’s heart and lungs with a stethoscope.
- Look for signs of a heart defect, such as a bluish tint to the skin, lips, or fingernails and rapid breathing.
- Look at your baby’s general appearance. Some children who have tetralogy of Fallot also have DiGeorge syndrome. This syndrome causes characteristic facial traits, such as wide-set eyes.
Tetralogy of Fallot is a critical congenital heart defect that may be detected with newborn screening using pulse oximetry (also known as pulse ox). Pulse oximetry is a simple bedside test to estimate the amount of oxygen in a baby’s blood. Low levels of oxygen in the blood can be a sign of a critical congenital heart defect. Newborn screening using pulse oximetry can identify some infants with a critical congenital heart defect, like pulmonary atresia, before they show any symptoms.
During Pregnancy
During pregnancy, there are screening tests (also called prenatal tests) to check for birth defects and other conditions. Tetralogy of Fallot might be seen during an ultrasound (which creates pictures of the body). Some findings from the ultrasound may make the health care provider suspect a baby may have tetralogy of Fallot. If so, the health care provider can request a fetal echocardiogram to confirm the diagnosis. A fetal echocardiogram is an ultrasound of the heart of the fetus. This test can show problems with the structure of the heart and how the heart is working with this defect.
After a Baby Is Born
Tetralogy of Fallot usually is diagnosed after a baby is born, often after the infant has an episode of turning blue during crying or feeding (a tet spell). Some findings on a physical exam may make the health care provider think a baby may have tetralogy of Fallot, including bluish-looking skin or a heart murmur (a “whooshing” sound caused by blood not flowing properly through the heart). However, it is not uncommon for a heart murmur to be absent right at birth.
The health care provider can request one or more tests to confirm the diagnosis.
Diagnostic Tests and Procedures
Your child’s doctor may recommend several tests to diagnose tetralogy of Fallot. These tests can provide information about the four heart defects that occur in tetralogy of Fallot and how serious they are.
Echocardiography
The most common test is an echocardiogram. An echocardiogram is an ultrasound of the heart that can show problems with the structure of the heart and how the heart is working (or not) with this defect. Echocardiography also is useful for helping the doctor follow the child’s health over time.
Echocardiography (echo) is a painless test that uses sound waves to create a moving picture of the heart. During the test, the sound waves (called ultrasound) bounce off the structures of the heart. A computer converts the sound waves into pictures on a screen.
Echo allows the doctor to clearly see any problem with the way the heart is formed or the way it’s working.
Echo is an important test for diagnosing tetralogy of Fallot because it shows the four heart defects and how the heart is responding to them. This test helps the cardiologist decide when to repair the defects and what type of surgery to use.
Echo also is used to check a child’s condition over time, after the defects have been repaired.
EKG (Electrocardiogram)
An EKG is a simple, painless test that records the heart’s electrical activity. The test shows how fast the heart is beating and its rhythm (steady or irregular). An EKG also records the strength and timing of electrical signals as they pass through the heart.
This test can help the doctor find out whether your child’s right ventricle is enlarged (ventricular hypertrophy).
Chest X Ray
A chest x ray is a painless test that creates pictures of the structures in the chest, such as the heart and lungs. This test can show whether the heart is enlarged or whether the lungs have extra blood flow or extra fluid, a sign of heart failure.
Pulse Oximetry
For this test, a small sensor is attached to a finger or toe (like an adhesive bandage). The sensor gives an estimate of how much oxygen is in the blood.
Cardiac Catheterization
During cardiac catheterization, a thin, flexible tube called a catheter is put into a vein in the arm, groin (upper thigh), or neck. The tube is threaded to the heart.
Special dye is injected through the catheter into a blood vessel or one of the heart’s chambers. The dye allows the doctor to see the flow of blood through the heart and blood vessels on an x-ray image.
The doctor also can use cardiac catheterization to measure the pressure and oxygen level inside the heart chambers and blood vessels. This can help the doctor figure out whether blood is mixing between the two sides of the heart.
Tetralogy of Fallot treatment
Surgery is the only effective treatment for tetralogy of Fallot. Tetralogy of Fallot can be treated by surgery soon after the baby is born. During surgery, doctors widen or replace the pulmonary valve and enlarge the passage to the pulmonary artery. They also will place a patch over the ventricular septal defect to close the hole between the two lower chambers of the heart. These actions will improve blood flow to the lungs and the rest of the body.
Most infants will live active, healthy lives after surgery. However, they will need regular follow-up visits with a cardiologist (a heart doctor) to monitor their progress and check for other health conditions that might develop as they get older. As adults, they may need more surgery or medical care for other possible problems.
Beta blocker medication can be used to help with hypoxic spells while the patient awaits definitive surgical intervention.
Tetralogy of fallot surgery
Tetralogy of Fallot is repaired with open-heart surgery, either soon after birth or later in infancy. The goal of surgery is to repair the four defects of tetralogy of Fallot so the heart can work as normally as possible. Repairing the defects can greatly improve a child’s health and quality of life.
The pediatric cardiologist and cardiac surgeon will decide the best time to do the surgery. They will base their decision on your baby’s health and weight and the severity of the defects and symptoms.
Surgical therapy for Tetralogy of Fallot involves closing the ventricular septal defect (VSD) and enlarging the passage from the right ventricle to the lung artery. This surgery is often carried out at about 6 months of age but the timing can vary. Sometimes, if the cyanosis (low oxygen) is persistently too great, a smaller temporary operation called a “shunt” is performed where blood from a body artery is directed through a small tube to the lungs to add extra blood flow where it is needed. Later the shunt is removed when the major repair is carried out. After surgery there are always things that will be different to normal which need to be checked from time to time.
Types of Surgery
Complete Intracardiac Tetralogy of Fallot repair
Surgery to repair tetralogy of Fallot improves blood flow to the lungs. Surgery also ensures that oxygen-rich and oxygen-poor blood flow to the right places.
The surgeon will:
- Widen the narrowed pulmonary blood vessels. The pulmonary valve is widened or replaced. Also, the passage from the right ventricle to the pulmonary artery is enlarged. These procedures improve blood flow to the lungs. This allows the blood to get enough oxygen to meet the body’s needs.
- Repair the ventricular septal defect (VSD). A patch is used to cover the hole in the septum. This patch stops oxygen-rich and oxygen-poor blood from mixing between the ventricles.
Fixing these two defects resolves problems caused by the other two defects. When the right ventricle no longer has to work so hard to pump blood to the lungs, it will return to a normal thickness. Fixing the VSD means that only oxygen-rich blood will flow out of the left ventricle into the aorta.
The incision (cut) that the surgeon makes to reach the heart usually heals in about 6 weeks. The surgeon or a hospital staff member will explain when it’s okay to give your baby a bath, pick him or her up under the arms, and take your baby for regular shots (immunizations).
After surgery
While most babies and adults do well after intracardiac repair, long-term complications are common. Complications may include:
- Chronic pulmonary regurgitation, in which blood leaks through the pulmonary valve back into the pumping chamber (right ventricle)
- Other heart valve problems, such as blood leaking back through the tricuspid valve
- Holes in the wall between the ventricles (ventricular septal defects) that may continue to leak after repair or may need re-repair
- Enlarged right ventricle or left ventricle that isn’t working properly
- Irregular heartbeats (arrhythmias)
- Coronary artery disease
- Aortic root dilation, in which the ascending aorta enlarges
- Sudden cardiac death
Complications can continue throughout childhood, adolescence and adulthood for people with tetralogy of Fallot. Most adults with repaired tetralogy of Fallot may require another procedure or intervention during their lifetimes. It’s very important to have regular follow-up with a cardiologist trained in caring for people with congenital heart disease (pediatric cardiologist or adult congenital cardiologist) who can evaluate you and determine the appropriate timing of another intervention or procedure.
Sometimes blood flow to the lungs may still be restricted after intracardiac repair. Infants, children or adults with these complications might require additional surgeries. More commonly, there is leakage through the repaired pulmonary valve. Most adults with repaired tetralogy of Fallot may have pulmonary valve leakage (regurgitation) and may need to have the pulmonary valve replaced during their lifetimes. Your cardiologist will determine the most appropriate timing for this procedure.
Arrhythmias are common after repair and may be treated with medications, a procedure to treat the arrhythmias (ablation) or a special pacemaker device that treats life-threatening heart rhythms (implantable cardioverter-defibrillator).
In addition, as with any surgery, there’s a risk of infection, unexpected bleeding or blood clots.
Temporary or Palliative Surgery
It was common in the past to do temporary surgery during infancy for tetralogy of Fallot. This surgery improved blood flow to the lungs. A complete repair of the four defects was done later in childhood.
Now, tetralogy of Fallot usually is fully repaired in infancy. However, some babies are too weak or too small to have the full repair. They must have temporary surgery first. This surgery improves oxygen levels in the blood. The surgery also gives the baby time to grow and get strong enough for the full repair.
For temporary surgery, the surgeon places a tube between a large artery branching off the aorta and the pulmonary artery. The tube is called a shunt. One end of the shunt is sewn to the artery branching off the aorta. The other end is sewn to the pulmonary artery.
The shunt creates an additional pathway for blood to travel to the lungs to get oxygen. The surgeon removes the shunt when the baby’s heart defects are fixed during the full repair.
After temporary surgery, your baby may need medicines to keep the shunt open while waiting for the full repair. These medicines are stopped after the surgeon removes the shunt.
Ongoing care
After surgery you or your child will need lifelong care with a cardiologist trained in treating congenital heart disease (adult congenital cardiologist or pediatric cardiologist), including routine follow-up appointments to make sure that the initial operation or procedure was successful and to monitor for any new complications.
Your or your child’s doctor may conduct a physical examination and order tests in regular follow-up appointments to evaluate and monitor your or your child’s condition.
The doctor might also recommend that you or your child limit strenuous physical activity, particularly if there’s any pulmonary valve leakage or obstruction, or arrhythmias.
Sometimes, antibiotics are recommended during dental procedures to prevent infections that might cause endocarditis — an inflammation of the lining of the heart. Antibiotics are especially important for those who have had prior endocarditis, have artificial valves or have had repair with prosthetic material. Ask the doctor what’s right for you or your child.
Lifestyle and home remedies
After treatment, you might have some concerns about how best to manage your or your child’s condition, including:
- Preventing infection. A child, adolescent or adult who has severe heart defects might need to take preventive antibiotics before certain dental and surgical procedures. Your or your child’s doctor can tell you if this is necessary. Maintaining good oral hygiene and getting regular dental checkups are excellent ways to help prevent infection.
- Exercising. Parents of children or adolescents with congenital heart defects or adults who have congenital heart defects often worry about the risks of vigorous activity even after successful treatment. Although some children, adolescents or adults might need to limit the amount or type of exercise, many can lead normal or near-normal lives. Decisions about exercise need to be made on a case-by-case basis, so ask your or your child’s doctor which activities are safe for you or your child.
If you’re an adult who has congenital heart disease, you might have concerns, such as:
- Employment. Having a congenital heart defect generally won’t limit your career options. If you have serious heart rhythm problems or the potential for life-threatening complications, evaluation by a specialty team is needed to determine risk, provide therapy and counsel people regarding employment.
- Pregnancy. Most women who have congenital heart disease tolerate pregnancy without any problems. However, a severe heart defect or complications such as arrhythmias can increase the risk of complications during pregnancy. If you have congenital heart disease, discuss family planning with your doctor. Your doctor may recommend that you be seen by a doctor trained in congenital heart disease before pregnancy and that you receive care during your pregnancy from doctors trained in congenital heart disease, genetics and high-risk obstetric care. Some heart medications aren’t safe during pregnancy and might need to be stopped or adjusted before you become pregnant.
Coping and support
It can be extremely frightening to learn that you or your child has potentially life-threatening heart defects. Although support groups aren’t for everyone, talking to other adults with congenital heart disease or to other parents — especially those whose children have already gone through corrective surgery — can give you hope, encouragement and support. Ask your or your child’s doctor if there are any support groups for parents of children with heart defects or adults with congenital heart disease in your area.
If your child has a heart defect, be sure to give yourself a break at times. Ask other family members or friends to help take care of your child. When your child is in the hospital, see if you can schedule friends and family to visit with your child so that you can go home to take a shower or nap, or to spend time with your other children.
To help coordinate your or your child’s care, you might prepare a brief note with your or your child’s diagnosis, medications, surgeries and dates, and the cardiologist’s name and number. This note will provide necessary information to others who might care for your child and will help any new doctor understand your or your child’s health history.
If you change health insurance plans, be sure your new plan will cover your or your child’s care. Some plans might not allow coverage for pre-existing conditions or might require a waiting period.
References