Vascular resistance
Vascular resistance is a term used to define the resistance that must be overcome to push blood through the circulatory system and create blood flow 1. Vascular resistance offered by the peripheral circulation is known as the systemic vascular resistance (SVR), while the resistance offered by the vasculature of the lungs is known as the pulmonary vascular resistance (PVR). The systemic vascular resistance (SVR) may also be referred to as the total peripheral resistance. Vascular resistance in the circulatory system is used to create blood pressure, the flow of blood and is also a component of cardiac function. When blood vessels constrict (vasoconstriction) this leads to an increase in systemic vascular resistance (SVR). When blood vessels dilate (vasodilation), this leads to a decrease in systemic vascular resistance (SVR).
Vascular resistance is used to maintain organ perfusion. In certain disease states, such as congestive heart failure, there is a hyper-adrenergic response, causing an increase in peripheral vascular resistance. Prolonged increases in blood pressure affect several organs throughout the body. In conditions such as shock, there is a decrease in vascular resistance thus causing decreased organ perfusion which leads to organ malfunction. Peripheral vascular resistance is mediated locally by metabolites, and over a distance on a neuro-hormonal level, therefore, many different components may become altered leading to changes in peripheral vascular resistance.
Units for measuring vascular resistance are dyn·s·cm-5 or pascal seconds per cubic meter (Pa·s/m³). Pediatric cardiologists use hybrid reference units (HRU), also known as Wood units, as they were introduced by Dr. Paul Wood. To convert from Wood units to MPa·s/m³ you must multiply by 8, or to dyn·s·cm-5 you must multiply by 80.
| Measurement | Reference Range |
|---|---|
| Systemic vascular resistance | 900–1200 dyn·s/cm5 (90–120 MPa·s/m3) |
| Pulmonary vascular resistance | 100–200 dyn·s/cm5 (10–20 MPa·s/m3) |
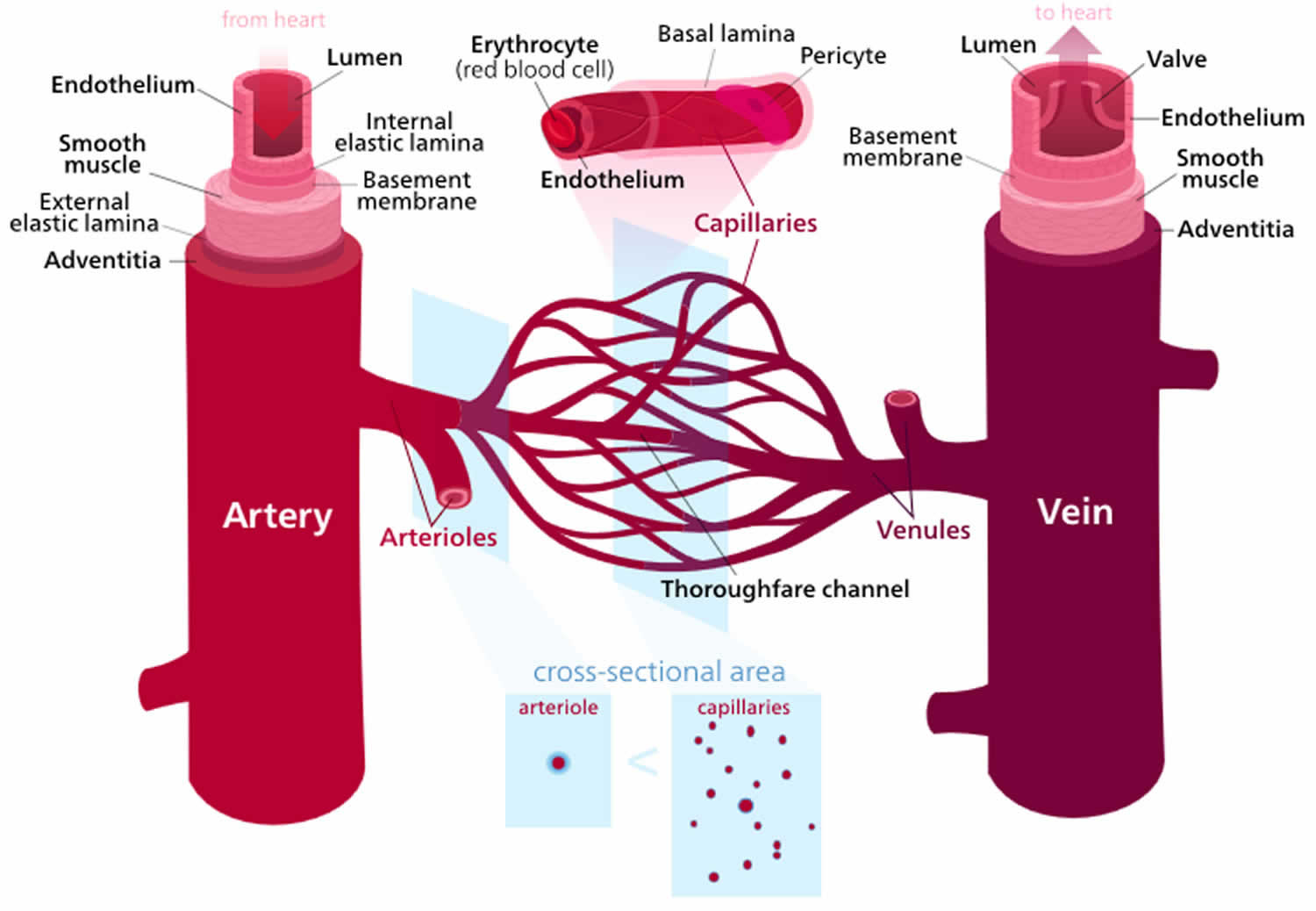
The major determinant of vascular resistance is small arteriolar (known as resistance arterioles) tone. These vessels are from 450 µm down to 100 µm in diameter. As a comparison, the diameter of a capillary is about 3 to 4 µm.
Another determinant of vascular resistance is the pre-capillary arterioles. These arterioles are less than 100 µm in diameter. They are sometimes known as autoregulatory vessels.
Regulation of vascular resistance
There are many factors that alter the vascular resistance. Many of the platelet-derived substances, including serotonin, are vasodilatory when the endothelium is intact and are vasoconstrictive when the endothelium is damaged.
Cholinergic stimulation causes release of endothelium-derived relaxing factor (EDRF) (later it was discovered that EDRF was nitric oxide) from intact endothelium, causing vasodilatation. If the endothelium is damaged, cholinergic stimulation causes vasoconstriction.
Role of adenosine
Adenosine probably doesn’t play a role in maintaining the vascular resistance in the resting state. However, it causes vasodilatation and decreased vascular resistance during hypoxia. Adenosine is formed in the myocardial cells during hypoxia, ischemia, or vigorous work, due to the breakdown of high-energy phosphate compounds (e.g., adenosine monophosphate, AMP). Most of the adenosine that is produced leaves the cell and acts as a direct vasodilator on the vascular wall. Because adenosine acts as a direct vasodilator, it is not dependent on an intact endothelium to cause vasodilatation.
Adenosine causes vasodilatation in the small and medium sized resistance arterioles (less than 100 µm in diameter). When adenosine is administered it can cause a coronary steal phenomenon, where the vessels in healthy tissue dilate as much as the ischemic tissue and more blood is shunted away from the ischemic tissue that needs it most. This is the principle behind adenosine stress testing.
Adenosine is quickly broken down by adenosine deaminase, which is present in red cells and the vessel wall.
Systemic vascular resistance calculation
Systemic vascular resistance (SVR) also referred to as the total peripheral resistance refers to the cumulative resistance of the thousands of arterioles in your body, or the lungs, respectively. Systemic vascular resistance is approximately equal to the resistance of the arterioles, since the arterioles are the chief resistance blood vessels in your body.
Systemic vascular resistance occurs when arterioles are unable to completely relieve pressure in the arteries. When the heart contracts, blood enters the arteries faster than it can leave, and the arteries stretch from the pressure. Although the blood vessels return to their normal state during diastole, the heart will contract again before all the blood has completely flowed into the arterioles. This inhibits the arterioles’ ability to fully relieve the pressure in the arteries, causing systemic vascular resistance.
- Systemic Vascular Resistance = (Mean Arterial Pressure – Central Venous Pressure) / Cardiac Output
Note: Because central venous pressure (CVP) is normally near 0 mmHg, the calculation is sometimes simplified to:
- Systemic Vascular Resistance = Mean Arterial Pressure / Cardiac Output
It is very important to note that systemic vascular resistance (SVR) can be calculated from mean arterial pressure (MAP) and cardiac output (CO), but it is not determined by either of these variables. A more accurate way to view this relationship is that at a given cardiac output (CO), if the mean arterial pressure (MAP) is very high, it is because systemic vascular resistance (SVR) is high. Mathematically, systemic vascular resistance (SVR) is the dependent variable in the above equations; however, physiologically, systemic vascular resistance (SVR) and cardiac output (CO) are normally the independent variables and mean arterial pressure (MAP) is the dependent variable
Cardiac output (CO) is defined as the amount of blood pumped by the left ventricle in unit time. Cardiac output is an indicator of the left ventricular function.
- Cardiac output = Stroke Volume x Heart Rate
Therefore, if there are 70 beats per minute, and 70 ml blood is ejected with each beat of the heart, the cardiac output is 4900 ml/minute.
The normal cardiac output is 5-6L/min and it can increase up to 5 times during exercise.
Mean arterial pressure (MAP) is a term used in medicine to describe a notional average blood pressure in an individual. It is defined as the average arterial pressure during a single cardiac cycle.
- Mean arterial pressure = (cardiac output x systemic vascular resistance) + central venous pressure
Central venous pressure is usually small enough to be neglected in this formula.
The mean arterial pressure (MAP) can also be calculated by the following formula 1:
- [(2/3) x (diastolic blood pressure)] + [(1/3) x (systolic blood pressure)]
Mean arterial pressure is normally between 65 and 110 mmHg, with a mean arterial pressure (MAP) of over 70mmHg needed for basic organ function 2.
In septic shock, a mean arterial pressure (MAP) of 65 mmHg is considered sufficient for end-organ perfusion 3.
Central venous pressure (CVP) describes the pressure of blood in the thoracic vena cava, near the right atrium of the heart. central venous pressure reflects the amount of blood returning to the heart and the ability of the heart to pump the blood into the arterial system.
Central venous pressure (CVP) is a good approximation of right atrial pressure, which is a major determinant of right ventricular end diastolic volume. However, there can be exceptions in some cases 4.
Normal central venous pressure can be measured from two points of reference:
- Sternum: 0-5 cm H2O
- Midaxillary line: 5-10 cm H2O
Central venous pressure can be measured by connecting the patient’s central venous catheter to a special infusion set which is connected to a small diameter water column. If the water column is calibrated properly the height of the column indicates the central venous pressure.
In most progressive intensive care units in the U.S., specialized monitors are available to continuously measure central venous pressure as well as other hemodynamic values.
Pulmonary vascular resistance
Pulmonary vascular resistance of healthy lung arterioles is typically about 15 to 20 percent that of the body, so pulmonary artery mean blood pressures are typically about 15-20% of aortic mean blood pressures.
As a consequence of the arteries being forced to work against peripheral resistance, there is substantial blood pressure even during diastole. Arterioles are sometimes referred to as resistance vessels because of their peripheral resistance.
Peripheral resistance and blood pressure
Blood pressure mediation is by a balance of the cardiac output and the peripheral vascular resistance. In idiopathic hypertension, most patients will have a near normal cardiac output, but their peripheral resistance is elevated. As mentioned earlier, mediation of this resistance is at the level of the arteriole. As with other tissues in the body, if there is prolonged constriction of the smooth muscle within the arterioles, this will lead to hypertrophy and thickening of the vessel. There are several mechanisms by which the systemic vascular resistance may be altered 5.
The renin-angiotensin system is mediated by the renal system. Renin is a molecule released from the juxtaglomerular apparatus in response to under perfusion; renin may also be released via activation of the sympathetic nervous system. Renin converts angiotensinogen into angiotensin 1, which subsequently converts into angiotensin 2 which acts as a vasoconstrictor on blood vessels, thus causing a rise in blood pressure 5.
The autonomic nervous system causes both vasoconstriction and vasodilation. Alpha-1 receptor activation causes vasoconstriction, and beta-2 receptor activation causes vasodilation 5.
The endothelium, itself, can modulate blood pressure. The endothelium may release nitrous oxide (vasodilation) or endothelin (vasoconstrictor) 5.
Several molecules have been found to place a role in blood pressure but have an unclear significance on the control of hypertension. Examples of these molecules are bradykinin, thromboxane, and atrial natriuretic peptide (ANP) 5.
Clinical significance
The main concerns of peripheral vascular resistance are when it is at its extremes, hypertension (too high) and hypotension (too low).
Hypertension (elevated peripheral vascular resistance) can be diagnosed when blood pressure measurements are greater than 140/90 on two separate clinical encounters. The majority of patients with hypertension are said to have essential hypertension, meaning there is no ‘underlying’ cause for the condition, and it is idiopathic. A minority of patients will have secondary hypertension, which is attributable to the underlying pathology. Examples of causes of secondary hypertension are renal disease (e.g., renal artery stenosis), endocrine conditions (e.g., Cushing’s disease), and drug-induced (e.g., oral contraceptives). Untreated hypertension can lead to chronic medical conditions consisting of coronary artery disease, renal disease, stroke, aneurysms, aortic dissection, congestive heart failure, peripheral vascular disease, and visual changes (e.g., retinal hemorrhages) 6.
Medications to lower peripheral vascular resistance include beta-blockers, diuretics, ACE-inhibitors, calcium-channel blockers, and alpha-blockers.
Hypotension is commonly associated with shock to which there are four main types. Hypovolemic shock is due to an excessive loss of blood resulting in a decreased cardiac output and increased systemic vascular resistance (SVR), as the body tries to maintain blood pressure. Cardiogenic shock is from a malfunction of the heart which results in decreased cardiac output and increased systemic vascular resistance (SVR). Neurogenic shock is from alterations in the autonomic nervous system that also results in decreased cardiac output and a decrease in systemic vascular resistance (SVR) from a loss of the sympathetic innervation. Distributive shock reduces systemic vascular resistance from anaphylaxis or septic mediators, with an increase in cardiac output 5.
Hypertensive urgency is a condition in which there is significantly elevated blood pressure (systolic BP greater than 180mmHg or diastolic BP greater than 120mmHg) without evidence of any end-organ damage. A hypertensive emergency may correlate with end-organ damage (e.g., a headache, chest pain, focal neurologic deficits, altered mental status, shortness of breath, pulmonary edema, renal failure, etc.) 7.
References- Delong C, Sharma S. Physiology, Peripheral Vascular Resistance. [Updated 2019 Feb 7]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2019 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK538308
- Moran D, Epstein Y, Keren G, Laor A, Sherez J, Shapiro Y. Calculation of mean arterial pressure during exercise as a function of heart rate. Appl Human Sci. 1995 Nov;14(6):293-5.
- Rodriguez R, Cucci M, Kane S, Fernandez E, Benken S. Novel Vasopressors in the Treatment of Vasodilatory Shock: A Systematic Review of Angiotensin II, Selepressin, and Terlipressin. J Intensive Care Med. 2018 Dec 18;:885066618818460.
- Independence of changes in right atrial pressure and central venous pressure. Bull Exp Biol Med. 2002 Oct;134(4):318-20. https://link.springer.com/article/10.1023%2FA%3A1021931508946
- Bolívar JJ. Essential hypertension: an approach to its etiology and neurogenic pathophysiology. Int J Hypertens. 2013;2013:547809.
- Gargiulo R, Suhail F, Lerma EV. Hypertension and chronic kidney disease. Dis Mon. 2015 Sep;61(9):387-95.
- Henny-Fullin K, Buess D, Handschin A, Leuppi J, Dieterle T. [Hypertensive urgency and emergency]. Ther Umsch. 2015 Jun;72(6):405-11.