Vitamin B5
Vitamin B5 also known as pantothenic acid, is a water-soluble vitamin that is naturally present in some foods (i.e., eggs, milk, vegetables, fish, shellfish, beef, chicken, animal organs (liver and kidney), avocados, legumes, mushrooms, sweet potatoes and whole grains), added to others, and available as a dietary supplement 1. The main function of vitamin B5 or pantothenic acid is a precursor in the biosynthesis of coenzyme A (CoA) and acyl carrier protein (Figure 1) 2, 3, 4. Coenzyme A (CoA) is essential for fatty acid synthesis and degradation, transfer of acetyl and acyl groups, and a multitude of other anabolic and catabolic processes 5, 6, 7. Acyl carrier protein’s main role is in fatty acid synthesis 3. Coenzyme A (CoA) reacts with acyl groups, giving rise to thioester derivatives, such as acetyl-CoA, succinyl-CoA, malonyl-CoA, and 3-hydroxy-3-methylglutaryl (HMG)-CoA. Coenzyme A (CoA) and its acyl derivatives are required for reactions that generate energy from the degradation of dietary fat, carbohydrates, and proteins 8. In addition, coenzyme A (CoA) in the form of acetyl-CoA and succinyl-CoA is involved in the citric acid cycle, in the synthesis of essential fats, cholesterol, steroid hormones, vitamins A and D, the neurotransmitter acetylcholine, and in the fatty acid beta-oxidation pathway 8. Coenzyme A (CoA) derivatives are also required for the synthesis of the hormone, melatonin, and for a component of hemoglobin called heme. Furthermore, metabolism of a number of drugs and toxins by the liver requires coenzyme A 9.
A wide variety of plant and animal foods contain vitamin B5 (pantothenic acid) 2. About 85% of dietary vitamin B5 (pantothenic acid) is in the form of coenzyme A (CoA) or phosphopantetheine 3, 6. These forms are converted to vitamin B5 (pantothenic acid) by digestive enzymes (nucleosidases, peptidases, and phosphorylases) in the intestinal lumen and intestinal cells. Pantothenic acid (vitamin B5) is absorbed in the intestine and delivered directly into the bloodstream by active transport (and possibly simple diffusion at higher doses) 6, 2. Pantetheine, the dephosphorylated form of phosphopantetheine, however, is first taken up by intestinal cells and converted to pantothenic acid before being delivered into the bloodstream 3. The intestinal flora also produces pantothenic acid, but its contribution to the total amount of pantothenic acid that the body absorbs is not known 6. Red blood cells carry pantothenic acid throughout the body 6. Most pantothenic acid in tissues is in the form of coenzyme A (CoA), but smaller amounts are present as acyl carrier protein or free pantothenic acid 6.
Few data on vitamin B5 (pantothenic acid) intakes in the United States are available. However, a typical mixed diet in the United States provides an estimated daily intake of about 6 mg, suggesting that most people in the United States consume adequate amounts 10. Some vitamin B5 (pantothenic acid) intake information is available from other Western populations. For example, a 1996–1997 study in New Brunswick, Canada, found average daily vitamin B5 (pantothenic acid) intakes of 4.0 mg in women and 5.5 mg in men 11.
Pantothenic acid status is not routinely measured in healthy people 1. Microbiologic growth assays, animal bioassays, and radioimmunoassays can be used to measure pantothenic concentrations in blood, urine, and tissue, but urinary concentrations are the most reliable indicators because of their close relationship with dietary intake 6. With a typical American diet, the urinary excretion rate for pantothenic acid is about 2.6 mg/day 12, 5. Excretion of less than 1 mg pantothenic acid per day suggests deficiency 13, 2. Like urinary concentrations, whole-blood concentrations of pantothenic acid correlate with pantothenic acid intake, but measuring pantothenic acid in whole blood requires enzyme pretreatment to release free pantothenic acid from coenzyme A (CoA) 2. Normal blood concentrations of pantothenic acid range from 1.6 to 2.7 mcmol/L, and blood concentrations below 1 mcmol/L are considered low and suggest deficiency 2. Unlike whole-blood concentrations, plasma levels of pantothenic acid do not correlate well with changes in intake or status 2.
Vitamin B5 (pantothenic acid) deficiency is generally rare since the vitamin is present in many foods. However, vitamin B5 (pantothenic acid) deficiency can present in people with severe malnutrition 14. An individual with vitamin B5 (pantothenic acid) deficiency commonly has deficiencies in other nutrients, which can make it challenging to identify the effects that are specific to vitamin B5 (pantothenic acid) deficiency. An experimental vitamin B5 (pantothenic acid) deficiency study associated the deficiency with symptoms such as fatigue, headache, malaise, personality changes, numbness, muscle cramps, paresthesia, muscle/ abdominal cramps, nausea, and impaired muscle coordination 13.
Pantothenic acid kinase 2 (PANK2) catalyzes the initial step of phosphorylation of pantothenic acid to 4’-phosphopantothenic acid. Individuals with a mutation in their pantothenate kinase 2 (PANK2) gene are likely to have a pantothenic acid inadequacy as well. Enough PANK2 mutations reduce the activity of pantothenate kinase 2, which can potentially decrease the conversion of pantothenic acid to coenzyme A (CoA) and lead to reduced CoA levels. PANK2 gene mutations also cause pantothenate kinase-associated neurodegeneration (PKAN). A common hallmark of individuals with pantothenate kinase-associated neurodegeneration (PKAN) is an accumulation of iron in the brain that forms a pattern called the “eye of the tiger” sign 15. Pantothenate kinase-associated neurodegeneration (PKAN) disease also presents with a progressive movement disorder, and other symptoms may vary significantly from case to case. Symptoms include dysarthria, dystonia, poor balance, spasticity, and muscle rigidity. Treatment of pantothenate kinase-associated neurodegeneration (PKAN) focuses mainly on reducing symptoms. A few anecdotal reports indicate that vitamin B5 supplements can reduce symptoms, but the benefits of the general use of this supplement in PKAN are not known 16.
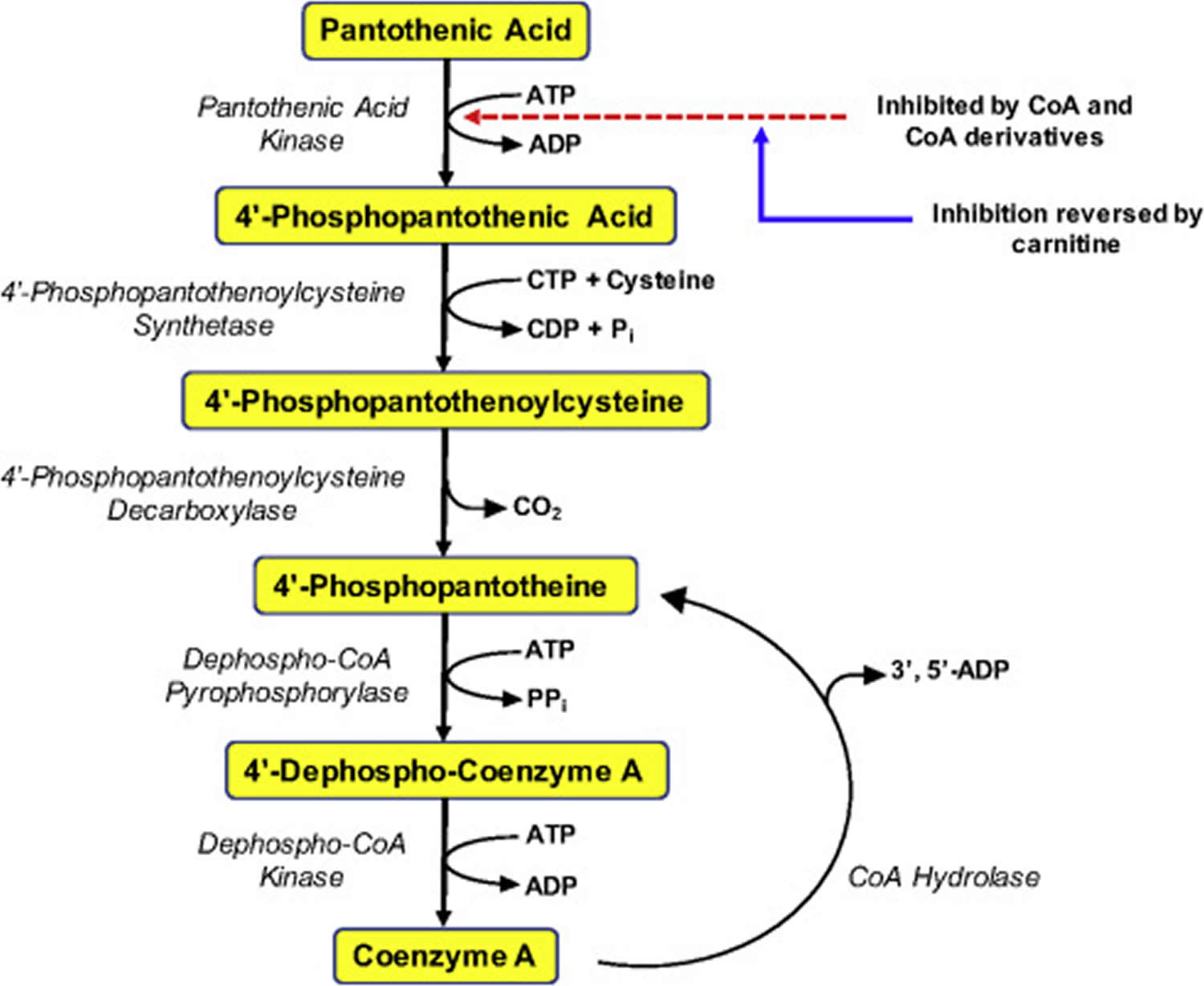
Figure 1. Coenzyme A (CoA) synthesis from vitamin B5 (pantothenic acid)
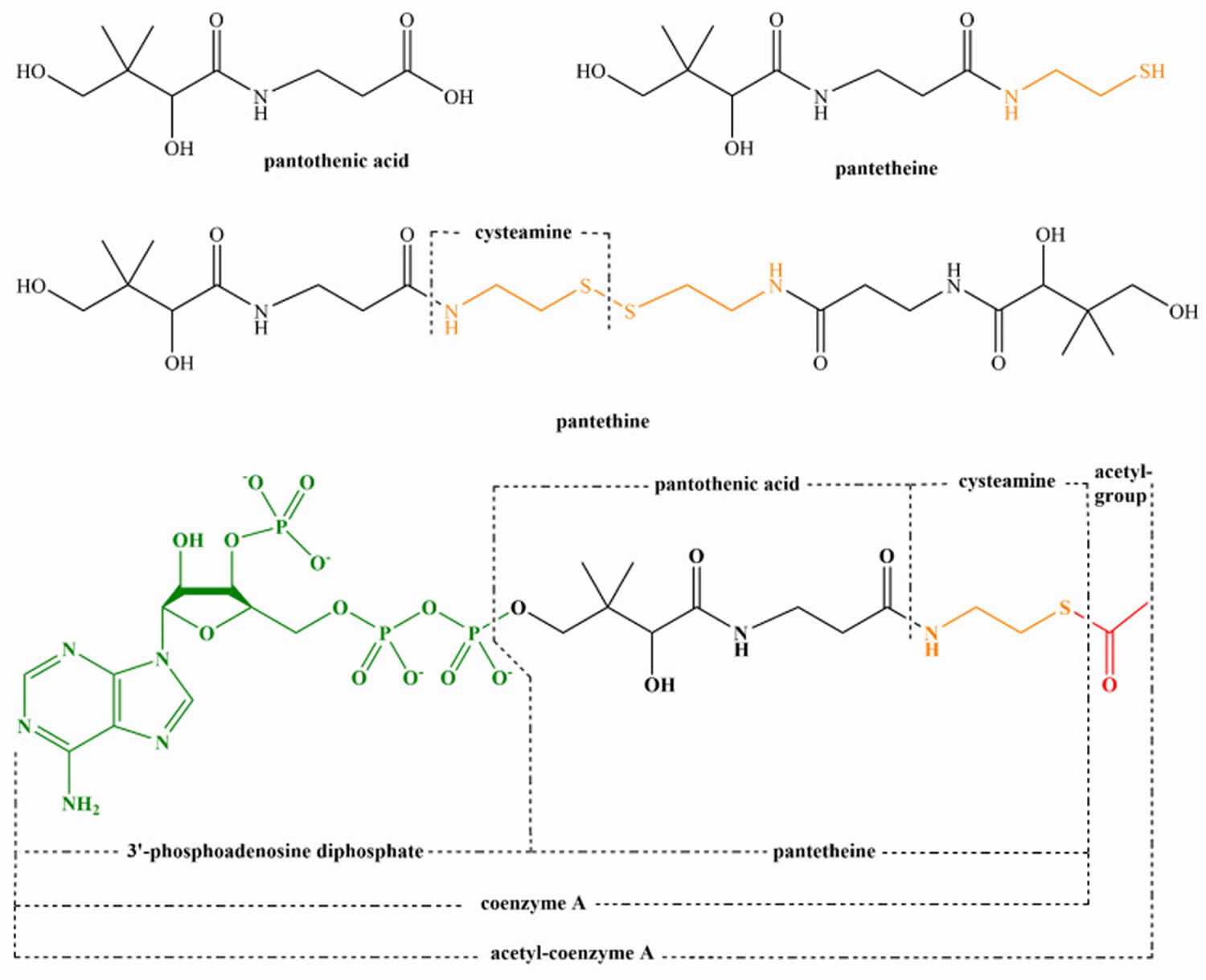
[Source 17 ]Figure 2. Structure of pantothenic acid and its derivatives (pantetheine, pantethine, and acetyl-CoA).
[Source 18 ]Vitamin B5 function
Vitamin B5 or pantothenic acid is essential for synthesis of coenzyme A (CoA) and acyl-carrier protein (ACP) and phosphopantetheine, which are crucial to fatty acid metabolism 19. Coenzyme A (CoA) plays a vital role in many catabolic and anabolic reactions. It is necessary for synthesis of fatty acids, cholesterol, acetylcholine, bile acids, and others 18. Coenzyme A (CoA) also plays a role in regulation of metabolism and gene expression. Coenzyme A (CoA) is required for processing large organic molecules, such as lipids, carbohydrates, and proteins. These reactions generate energy with formation of acylated forms of CoA, such as acetyl-CoA, succinyl-CoA, propionyl-CoA, isovaleryl-CoA, isobutyryl-CoA, α-methylbutyryl-CoA, and fatty acyl-CoA 20. The structure of pantothenic acid and its derivatives is shown in Figure 2.
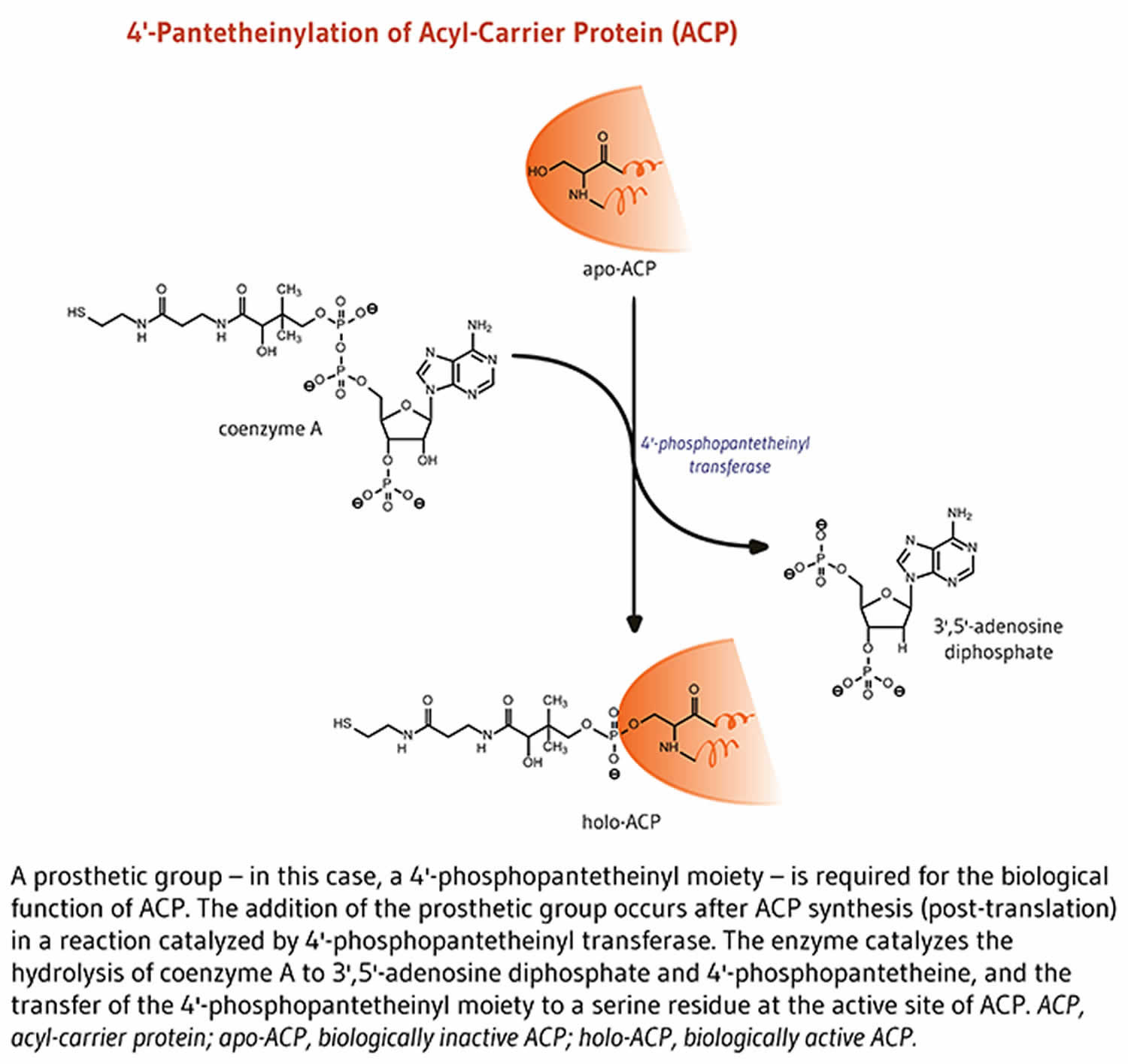
Acyl-carrier protein (ACP) is important for synthesis of fatty acids 18. Acyl-carrier protein (ACP) is expressed in the inactive form, apo-ACP. Its activation to holo-ACP requires the attachment of a prosthetic group (the 4′-phosphopantetheinyl moiety). This happens during the reaction with coenzyme A (CoA) catalyzed by 4′-phosphopantetheinyl transferase 21.
Coenzyme A
Pantothenic acid is a precursor in the biosynthesis of coenzyme A (CoA) (Figure 1), an essential coenzyme in a variety of biochemical reactions that sustain life 8. Pantothenic acid kinase 2 (PANK2) catalyzes the initial step of phosphorylation of pantothenic acid to 4’-phosphopantothenic acid. Coenzyme A (CoA) and its derivatives inhibit the synthesis of 4’-phosphopantothenic acid, but the inhibition can be reversed by carnitine, required for the transport of fatty acids into the mitochondria 22. The subsequent reactions in this biosynthetic pathway include the synthesis of the intermediate 4’-phosphopantetheine, as well as the recycling of coenzyme A to 4’-phosphopantetheine (Figure 1).
Coenzyme A (CoA) reacts with acyl groups, giving rise to thioester derivatives, such as acetyl-CoA, succinyl-CoA, malonyl-CoA, and 3-hydroxy-3-methylglutaryl (HMG)-CoA 8. Coenzyme A (CoA) and its acyl derivatives are required for reactions that generate energy from the degradation of dietary fat, carbohydrates, and proteins. In addition, coenzyme A (CoA) in the form of acetyl-CoA and succinyl-CoA is involved in the citric acid cycle, in the synthesis of essential fats, cholesterol, steroid hormones, vitamins A and D, the neurotransmitter acetylcholine, and in the fatty acid beta-oxidation pathway 8. Coenzyme A (CoA) derivatives are also required for the synthesis of the hormone, melatonin, and for a component of hemoglobin called heme 8. Furthermore, metabolism of a number of drugs and toxins by the liver requires coenzyme A (CoA) 9.
Coenzyme A was named for its role in acetylation reactions 8. Most acetylated proteins in the body have been modified by the addition of an acetate group that was donated by the coenzyme A thioester derivative, acetyl-CoA. Protein acetylation alters the overall charge of proteins, modifying their three-dimensional structure and, potentially, their function 8. For example, acetylation is a mechanism that regulates the activity of peptide hormones, including those produced by the pituitary gland 23. Also, protein acetylation, like other posttranslational modifications, has been shown to regulate the subcellular localization, the function, and the half-life of many signaling molecules, transcription factors, and enzymes. Notably, the acetylation of histones plays a role in the regulation of gene expression by facilitating transcription (i.e., mRNA synthesis), while deacetylated histones are usually associated with chromatin compaction and gene silencing. The acetylation of histones was found to result in structural changes of the chromatin, which affect both DNA-protein and protein-protein interactions. Crosstalk between acetylation marks and other posttranscriptional modifications of the histones also facilitate the recruitment of transcriptional regulators to the promoter of genes that are subsequently transcribed 24.
Finally, a number of signaling molecules are modified by the attachment of long-chain fatty acids donated by coenzyme A (CoA). These modifications are known as protein acylation and have central roles in cell-signaling pathways 9.
Acyl-carrier protein
Lipids are fat molecules essential for normal physiological function and, among other types, include sphingolipids (essential components of the myelin sheath that enhances nerve transmission), phospholipids (important structural components of cell membranes), and fatty acids 8. Fatty acid synthase (FAS) is a multi-enzyme complex that catalyzes the synthesis of fatty acids 8. Within the fatty acid synthase complex, the acyl-carrier protein (ACP) requires pantothenic acid in the form of 4′-phosphopantetheine for its activity as a carrier protein 22. A group, such as the 4’-phosphopantetheinyl moiety for acyl-carrier protein (ACP), is called a prosthetic group; the prosthetic group is not composed of amino acids and is a tightly bound cofactor required for the biological activity of some proteins (Figure 3). Acetyl-CoA, malonyl-CoA, and acyl-carrier protein (ACP) are all required for the synthesis of fatty acids in the cytosol 8. During fatty acid synthesis, the acyl groups of acetyl-CoA and malonyl-CoA are transferred to the sulfhydryl group (-SH) of the 4’-phosphopantetheinyl moiety of acyl-carrier protein (ACP). The prosthetic group is used as a flexible arm to transfer the growing fatty acid chain to each of the enzymatic centers of the type 1 fatty acid synthase complex 25. In the mitochondria, 4′-phosphopantetheine also serves as a prosthetic group for an acyl-carrier protein (ACP) homolog present in mitochondrial type 2 fatty acid synthase complex 25.
Figure 3. Acyl-carrier protein (ACP) function
[Source 8 ]10-formyltetrahydrofolate dehydrogenase
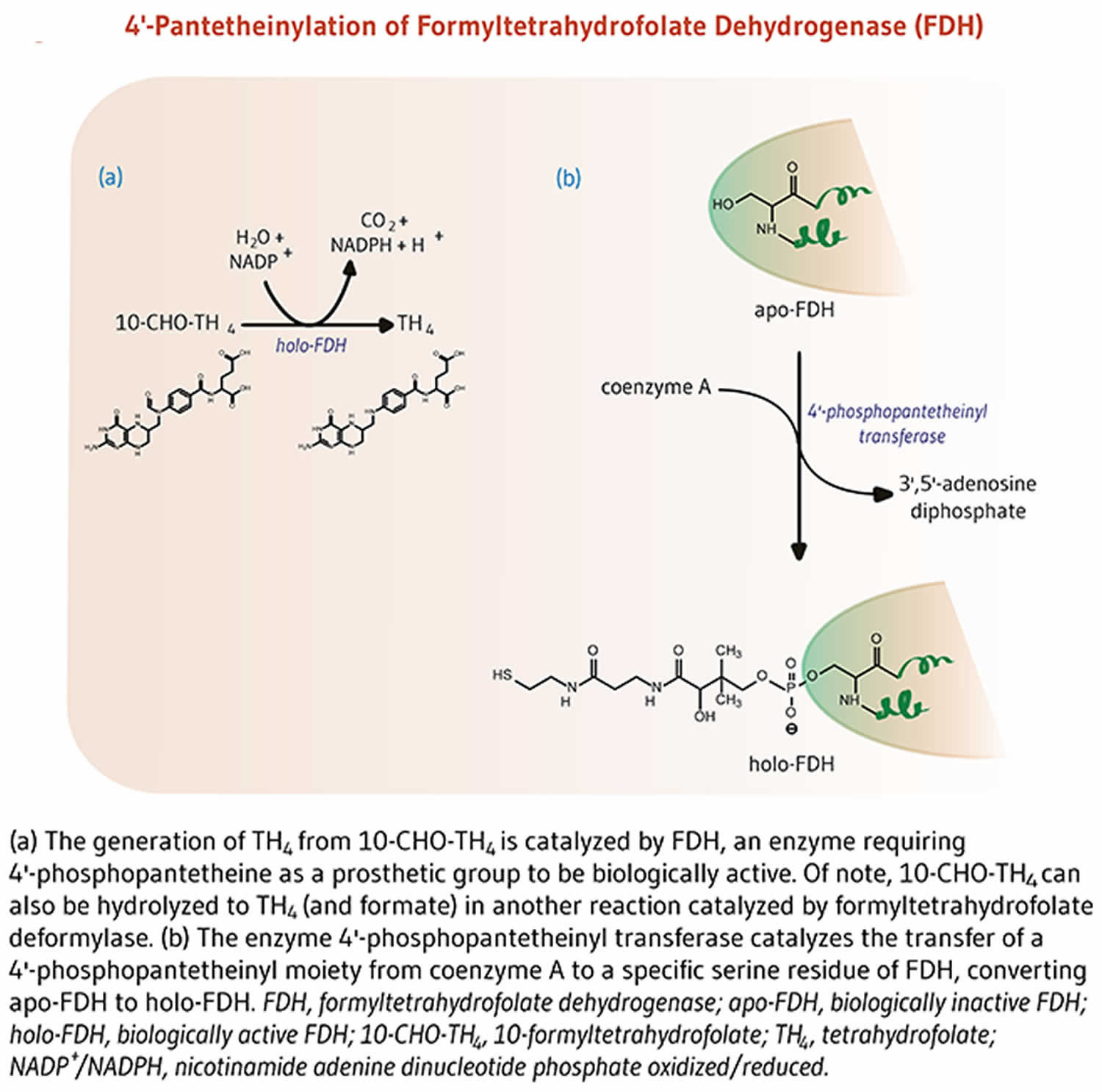
The enzyme 10-formyltetrahydrofolate dehydrogenase (FDH) catalyzes the conversion of 10-formyltetrahydrofolate to tetrahydrofolate, an essential cofactor in the metabolism of nucleic acids and amino acids (Figure 4) 8. Similar to acyl-carrier protein (ACP), 10-formyltetrahydrofolate dehydrogenase requires a 4’-phosphopantetheine prosthetic group for its biological activity 8. The prosthetic group acts as a swinging arm to couple the activities of the two catalytic domains of 10-formyltetrahydrofolate dehydrogenase 26, 27. A homolog of 10-formyltetrahydrofolate dehydrogenase in mitochondria also requires 4’-phosphopantetheinylation to be biologically active 28.
Figure 4. Formyltetrahydrofolate dehydrogenase function
[Source 8 ]Alpha-aminoadipate semialdehyde synthase
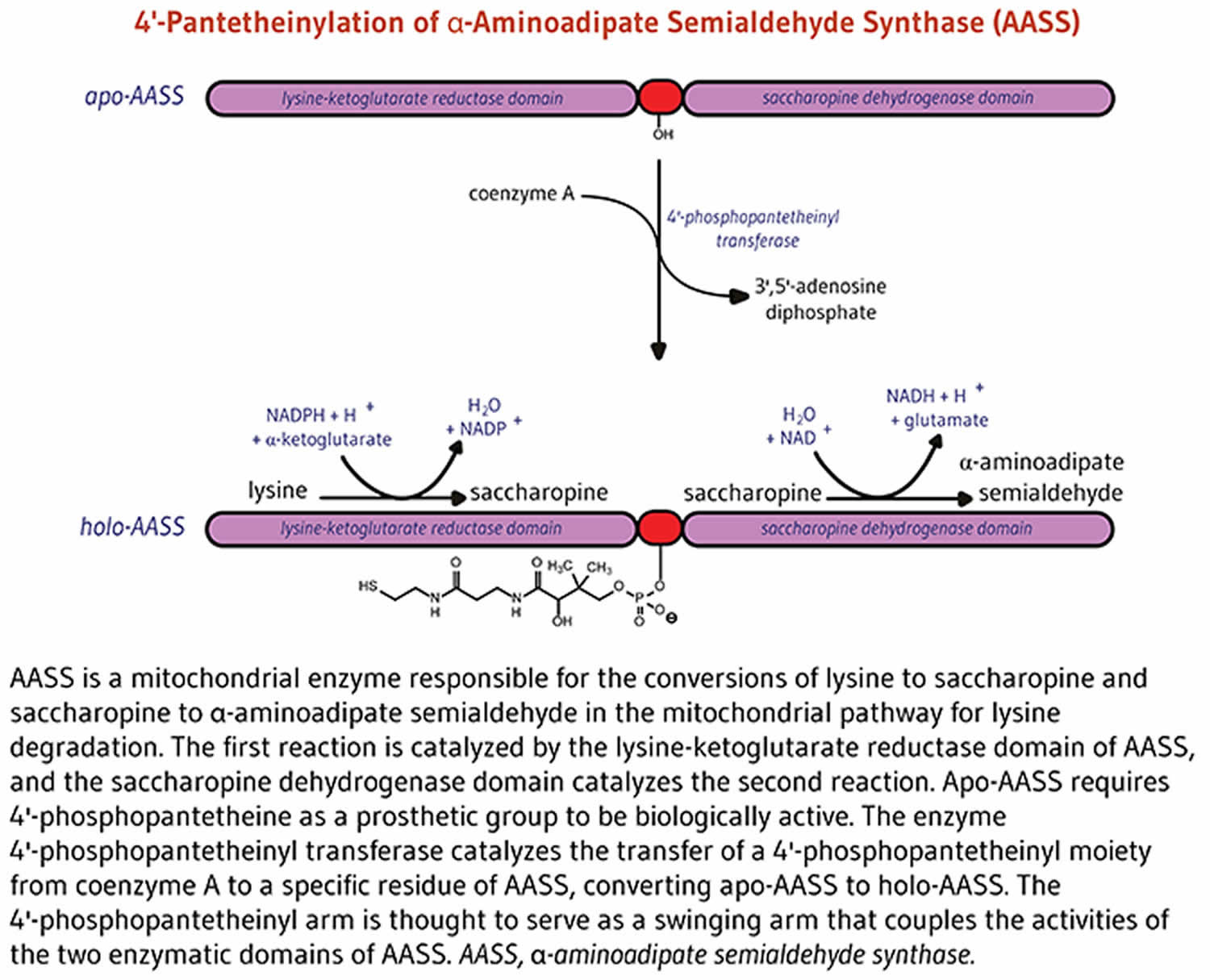
4’-phosphopantetheinylation is required for the biological activity of the apo-enzyme alpha-aminoadipate semialdehyde synthase (AASS) 8. Alpha-aminoadipate semialdehyde synthase (AASS) catalyzes the initial reactions in the mitochondrial pathway for the degradation of lysine — an essential amino acid for humans 8. Alpha-aminoadipate semialdehyde synthase (AASS) is made of two catalytic domains. The lysine-ketoglutarate reductase domain first catalyzes the conversion of lysine to saccharopine. Saccharopine is further converted to α-aminoadipate semialdehyde in a reaction catalyzed by the saccharopine dehydrogenase domain (Figure 5).
Figure 5. Alpha-aminoadipate semialdehyde synthase (AASS) function
[Source 8 ]Vitamin B5 health benefits
Scientists are studying vitamin B5 or pantothenic acid to understand how it affects health. Here is what research have shown.
High cholesterol and triglyceride levels
Because of pantothenic acid’s role in triglyceride synthesis and lipoprotein metabolism, experts have hypothesized that vitamin B5 or pantothenic acid supplementation might reduce lipid levels in patients with hyperlipidemia 29. The form of pantothenic acid called pantethine is being studied to see if it helps lower total cholesterol, low-density lipoprotein (LDL or “bad”) cholesterol, and triglyceride levels. It’s also being studied to see if it raises levels of high-density lipoprotein (HDL or “good”) cholesterol. The results of these studies so far are promising, but more research is needed to understand the effects of pantethine dietary supplements taken alone or combined with a heart-healthy diet.
Several clinical trials have shown that the form of pantothenic acid known as pantethine reduces lipid levels when taken in large amounts 30, but pantothenic acid itself does not appear to have the same effects 2. A 2005 review included 28 small clinical trials (average sample size of 22 participants) that examined the effect of pantethine supplements (median daily dose of 900 mg for an average of 12.7 weeks) on serum lipid levels in a total of 646 adults with hyperlipidemia 30. On average, the supplements were associated with triglyceride declines of 14.2% at 1 month and 32.9% at 4 months. The corresponding declines in total cholesterol were 8.7% and 15.1%, and for low-density lipoprotein (LDL) cholesterol were 10.4% and 20.1%. The corresponding increases in high-density lipoprotein (HDL) cholesterol were 6.1% and 8.4% 30.
A few additional clinical trials have assessed pantethine’s effects on lipid levels since the publication of the 2005 review. A double-blind trial in China randomly assigned 216 adults with hypertriglyceridemia (204–576 mg/dl) to supplementation with 400 U/day coenzyme A (CoA) or 600 mg/day pantethine 31. All participants also received dietary counseling. Triglyceride levels dropped by a significant 16.5% with pantethine compared with baseline after 8 weeks. Concentrations of total cholesterol and non–HDL cholesterol also declined modestly but significantly from baseline. However, these declines might have been due, at least in part, to the dietary counseling that the participants received 31.
Two randomized, blinded, placebo-controlled studies by the same research group in a total of 152 adults with low to moderate cardiovascular disease risk found that 600 mg/day pantethine for 8 weeks followed by 900 mg/day for 8 weeks plus a therapeutic lifestyle change diet resulted in small but significant reductions in total cholesterol, LDL cholesterol, and non-HDL cholesterol compared with placebo after 16 weeks 29, 32. Increasing the amount of pantethine from 600 to 900 mg/day did not increase the magnitude of reduction in the lipid measures.
Additional studies are needed to determine whether pantethine supplementation has a beneficial effect on hyperlipidemia independently of, and together with, eating a heart-healthy diet. Research is also needed to determine the mechanisms of pantethine’s effects on lipid levels.
Wound healing
The addition of calcium D-pantothenate and/or pantothenol to the medium of cultured skin fibroblasts given an artificial wound was found to increase cell proliferation and migration, thus accelerating wound healing in vitro (test tube studies) 33, 34. Likewise, in vitro (test tube studies) deficiency in pantothenic acid induced the expression of differentiation markers in proliferating skin fibroblasts and inhibited proliferation in human keratinocytes 35. The application of ointments containing either calcium D-pantothenate or pantothenol — also known as D-panthenol or dexpanthenol — to the skin has been shown to accelerate the closure of skin wounds and increase the strength of scar tissue in animals 22.
The effects of dexpanthenol on wound healing are unclear. In a placebo-controlled study that included 12 healthy volunteers, the application of dexpanthenol-containing ointment (every 12 hours for 1 to 6 days) in a model of skin wound healing was associated with an enhanced expression of markers of proliferation, inflammation, and tissue repair 36. However, the study failed to report whether these changes in response to topical dexpanthenol improved the wound-repair process compared to placebo 36. Some studies have shown no effects. Early randomized controlled trials in patients undergoing surgery for tattoo removal found that daily co-supplementation with 1 gram or 3 grams of vitamin C and 200 mg or 900 mg of pantothenic acid (vitamin B5) for 21 days did not significantly improve the wound-healing process 37, 38. Yet, in a recent randomized, double-blind, placebo-controlled study, the use of dexpanthenol pastilles (300 mg/day for up to 14 days post surgery) was found to accelerate mucosal healing after tonsillectomy in children 39.
Facial acne
A randomized, double-blind, placebo-controlled study of adults (average age of 31.8 ± 8.4 years) previously diagnosed with mild to moderate acne vulgaris was performed over over 12-weeks 40, 41. Subjects were randomized to the study agent, a pantothenic acid-based dietary supplement (2.2 g of pantothenic acid twice a day with food), or a placebo for 12 weeks 41. The primary outcome of the study was the difference in total lesion count between the study agent group versus the placebo group from baseline to endpoint. Secondary measurements included differences in mean non-inflammatory and inflammatory lesions, Investigators Global Assessment and Dermatology Life Quality Index (DLQI) scores between the two groups. The results from this study indicate that the administration of a pantothenic acid-based dietary supplement in healthy adults with facial acne lesions is safe, well tolerated and reduced total facial lesion count versus placebo after 12 weeks of administration 41. Secondary analysis shows that the study agent significantly reduced area-specific and inflammatory blemishes 41. Further randomized, placebo-controlled trials are needed to confirm these findings.
Skin conditions
The usage of vitamin B5 is prevalent within the field of dermatology. This interest has led to a study that compares the effectiveness of reduced form of vitamin B5 dexpanthenol (D-panthenol) as an alternative treatment to atopic dermatitis against a standard treatment of hydrocortisone. Overall, the study found that dexpanthenol can potentially treat mild to moderate childhood atopic dermatitis therapy 42. Other research suggests that dexpanthenol cream can be useful in managing mucocutaneous side effects that occur during isotretinoin therapy 43. Isotretinoin therapy is used as a treatment for acne, and its mucocutaneous side effects include dryness of mucous membranes, cheilitis, and xerosis.
The reduced form of vitamin B5 dexpanthenol (D-panthenol) effects are, however, likely not related to the physiological function of vitamin B5 but are mediated by its moisturizing effect, which is based on its hygroscopic property 18. Vitamin B5 dexpanthenol (D-panthenol) could be used topically as a cream, emollient, drops, gel, lotion, oil, ointment, solution, and spray in concentration of 2–5% 44. Dexpanthenol protects epithelium and promotes cellular proliferation. During the wound healing, it helps to recover the epidermal barrier function, has anti-inflammatory activity, and supports wound closure 45.
The healing properties of dexpanthenol-containing cream (5%) were confirmed on superficial skin lesions caused by application of 5% sodium lauryl sulfate solution for 4 hours. One week, twice daily dexpanthenol-containing cream (5%) application led to a significant enhancement of stratum corneum hydration, as well as reduction in skin roughness and inflammation 46. Other studies confirmed the effect of dexpanthenol containing emollient on sodium dodecyl sulfate (0.5%) induced skin barrier dysfunction. Dexpanthenol improved skin hydration and increased ceramide 3, as well as free fatty acid and cholesterol content, in the stratum corneum, and it also supported recolonization of the skin with commensal bacteria 47.
Dexpanthenol in ointment with petroleum jelly led to a significantly faster and pronounced reduction of skin lesions size and better re-epithelialization of ablative CO2 laser photo-damaged skin than the petroleum-jelly cream itself 48. Protective effect of an ointment with dexpanthenol (5%) was also seen in combination with zinc oxide in irritant diaper dermatitis in comparison to the control ointment base 49. Two-week administration of dexpanthenol 5% water-oil formulations 4 to 8 times daily restored the skin barrier of freshly tattooed skin. The disadvantage of this study is that the effect was not compared with a control group 50.
In treatment of atopic dermatitis in children, dexpanthenol (5%) ointment exerted equal effectiveness to hydrocortisone (1%) ointment and, therefore, can be used as alternative to treatment of mild and moderate atopic dermatitis 42.
Use of dexpanthenol cream (5%) on treatment of traumatic nipples of breastfeeding mothers had the same therapeutic effect in comparison with pure lanolin or 0.2% peppermint oil creams administered every 8 hours for 14 days 51.
The application of 2% dexpanthenol drops on corneal epithelial wounds after surface laser ablation only induced little effect on corneal epithelial regeneration, and, in general, the effect was of minimal clinical relevance after 2 months of use 52. However, dexpanthenol has been found to be effective in treatment of dry eye, where it exerted superior improvement in disturbances of corneal epithelium permeability comparing with dexpanthenol-free drops 53.
Dexpanthenol is also added to topical nasal decongestant (sprays and droplets) containing α-sympathomimetics to treat acute allergic or non-allergic rhinitis or after nasal surgery. A combined preparation of oxymethazoline (0.05%) with dexpanthenol (5%) showed a better efficacy than xylomethazoline (0.1%) alone in patients with acute allergic rhinitis or with post-nasal surgery. The relief in nasal congestion was significantly better, recovery time was shorter, and significant improvements in sneezing, nasal discharge, and irritation were also observed 54. Similarly, addition of dexpanthenol to xylometazoline significantly reduced nasal obstruction, rhinorrhea, hyperplasia of nasal concha, and redness of the nasal mucous membrane compared with xylometazoline alone 55, 56.
Graying of hair
Mice that are deficient in pantothenic acid (vitamin B5) developed skin irritation and graying of the fur, which is reversed by pantothenic acid administration. In humans, there is no evidence that taking pantothenic acid (vitamin B5) as supplements or using shampoos containing pantothenic acid can prevent or restore hair color 57.
How much vitamin B5 do I need?
The amount of vitamin B5 (pantothenic acid) you need depends on your age and sex. Average daily recommended amounts are listed below in milligrams (mg).
Few data on vitamin B5 or pantothenic acid intakes in the United States are available 1. However, a typical mixed diet in the United States provides an estimated daily intake of about 6 mg, suggesting that most people in the United States consume adequate amounts 10. Some intake information is available from other Western populations. For example, a 1996–1997 study in New Brunswick, Canada, found average daily pantothenic acid intakes of 4 mg in women and 5.5 mg in men 11.
Table 1. Adequate Intakes for vitamin B5 (pantothenic acid)
| Life Stage | Recommended Amount |
|---|---|
| Birth to 6 months | 1.7 mg |
| Infants 7–12 months | 1.8 mg |
| Children 1–3 years | 2 mg |
| Children 4–8 years | 3 mg |
| Children 9–13 years | 4 mg |
| Teens 14–18 years | 5 mg |
| Adults 19 years and older | 5 mg |
| Pregnant teens and women | 6 mg |
| Breastfeeding teens and women | 7 mg |
Footnote:
- Adequate Intake (AI) = Intake at this level is assumed to ensure nutritional adequacy; established when evidence is insufficient to develop an Recommended Dietary Allowance (RDA).
- Recommended Dietary Allowance (RDA) = Average daily level of intake sufficient to meet the nutrient requirements of nearly all (97%–98%) healthy individuals; often used to plan nutritionally adequate diets for individuals.
What foods have vitamin B5?
Vitamin B5 or pantothenic acid is naturally present in almost all plant- and animal-based foods 6. Vitamin B5 or pantothenic acid is also added to some foods, including some breakfast cereals and beverages (such as energy drinks) 6. Limited data indicate that the body absorbs 40%–61% (or half, on average) of pantothenic acid from foods 12.
The U.S. Department of Agriculture’s (USDA’s) FoodData Central (https://fdc.nal.usda.gov) lists the nutrient content of many foods and provides a comprehensive list of foods containing pantothenic acid arranged by nutrient content (https://www.nal.usda.gov/sites/www.nal.usda.gov/files/pantothenic_acid.pdf).
You can get recommended amounts of vitamin B5 (pantothenic acid) by eating a variety of foods, including the following:
- Beef, poultry, seafood, and organ meats
- Eggs and milk
- Vegetables such as mushrooms (especially shiitakes), avocados, potatoes, and broccoli
- Whole grains, such as whole wheat, brown rice, and oats
- Peanuts, sunflower seeds, and chickpeas.
Food processing may alter the content of vitamin B5 or pantothenic acid 58, 59. The milling of cereals, in which grains, such as wheat, rice, and corn, are dehulled and ground into smaller pieces or flours to improve palatability, reduce cooking time, and create food products, but remove grain parts rich in micronutrients, resulting in considerable losses of vitamin B5 or pantothenic acid 60, 61, 62, 63, 64. Milling reduces vitamin B5 or pantothenic acid contents, in comparison to whole cereals, by 50–55% and 64–88% in wheat and maize, respectively 60, 65, 66. Vitamin B5 or pantothenic acid losses are 50–67% and 18–25% in non-parboiled and parboiled white rice, respectively, compared to brown rice 60, 67, 63, 66.
Vitamin B5 or pantothenic acid is quite stable during thermal processing at pH levels of 5–7; losses of pantothenic acid during the preparation and cooking of foods are normally not very large 68, 58, but substantial losses of pantothenic acid can occur through leaching into the cooking liquids, such as water, soup, gravy, or drippings; when these are consumed along with the cooked food, a great part of the vitamin is retained 69, 70, 71, 72, 73. Vitamin B5 or pantothenic acid content in pork, beef, and chicken is reduced owing to steaming, braising, and, in particular, by boiling, by 15–50% solely in meat due to leaching. In the whole dish, the losses are only 10–20%. Frying decreases the vitamin level by 20%, and it only decreases by 10% when the meat is breaded 69. Similarly, a decrease in pantothenic acid in fish during cooking by different methods comes about 69, 74, 75. Steaming, boiling, baking, and frying of potatoes with the peel bring on pantothenic acid losses of 10% in all cases, but the losses might reach 30% in peeled potatoes when boiled 69, 70. In addition, in vegetables, boiling and steaming usually causes declines of 10% in the total dish, and those of 30–40% and 15%, respectively, in vegetables alone 69, 70, 76, 77. Stewing, frying, and baking lessen pantothenic acid amounts in vegetables by 10% 69, 70. Vitamin B5 or pantothenic acid losses of 24–67% in legumes during boiling are influenced by the pre-soaking method and cooking times 71, 78, 79. Boiling of rice results in a decrease of 59–66% in pantothenic acid content 60. That is why steaming is preferred to boiling, in particular, when cooked vegetables are eaten without cooking liquids 69, 76, 77. Poached, boiled, and fried eggs lose, due to cooking, 4%, 7%, and 9% of their pantothenic acid, respectively 80. In milk, pantothenic acid is stable during pasteurization, since the normal pH of milk is within the optimal pH stability range; milk generally loses less than 10% during processing 71, 58, 81.
In breadmaking, no significant difference of vitamin B5 or pantothenic acid was observed during the kneading phase, while a mild decrease of 12% was documented during baking. This indicates that pantothenic acid is more sensitive to heat than to light and oxygen 82. The roasting of peanuts at 160 °C and 180 °C decreases the amount of pantothenic acid by 24% and 92%, respectively; so, peanuts can be an excellent source if properly processed 83.
Canning leads to various reductions in vitamin B5 or pantothenic acid content: 1–43% in pork luncheon meat, depending on times and temperatures used during thermal processing 84; 20–35%, 46–78%, and 51%, in foods of animal origin (such as meats, fish, and dairy products), vegetables, and fruits and fruit juices, respectively 66. Thermal degradation kinetics of pantothenic acid in extracts of Averrhoa bilimbi fruits showed that increasing the temperature speeds up the decomposition, which was also linearly time-dependent 85. Treatment of food with ionizing radiation used as a method for its preservation has insignificant effects on pantothenic acid content 86, 87. Less vitamin B5 or pantothenic acid is in food products based on nixtamalized (i.e., alkali-treated) maize 88, 89.
Lower contents of vitamin B5 or pantothenic acid in frozen foods, compared to those in raw ones, have been reported; decreases were 18–63% in vegetables, 29–71% in legumes, 7% in fruits and fruit juices, and 4–55% in fish 90, 66, 91. After thawing frozen meat, pantothenic acid, together with other B vitamins, transfer in a drip; amounts of pantothenic acid from defrosted meat found in the drip were 7% and 33% in pork and in beef, respectively. For prevention of the loss of the vitamin, collection and use of the drip is recommended 72, 92, 93.
Regarding fortification of foods using vitamin B5 or pantothenic acid, adult human intake of that vitamin has generally been considered adequate in view of the absence of deficiency in normal populations and the fact that the daily requirement for vitamin B5 or pantothenic acid is easily fulfilled from most natural dietary sources owing to its ubiquitous distribution 60, 94. Pantothenic acid (as calcium pantothenate or sodium pantothenate or dexpanthenol) is added to various foods (such as milk-based products, breakfast cereals, and rice powders) to prevent deficiency due to incorrect nutrition or malnutrition or for certain nutritional requirements (baby foods, e.g., for non-breastfed infants; athletes’ products; low-calorie, reduced-calorie, and vitamin-rich foods) 95, 96, 97, 98, 94, 99.
Table 2. Vitamin B5 (pantothenic acid) content of selected foods
| Food | Milligrams (mg) per serving | Percent DV* |
|---|---|---|
| Beef liver, boiled, 3 ounces | 8.3 | 166 |
| Breakfast cereals, fortified with 100% of the DV | 5 | 100 |
| Shitake mushrooms, cooked, ½ cup pieces | 2.6 | 52 |
| Sunflower seeds, ¼ cup | 2.4 | 48 |
| Chicken, breast meat, skinless, roasted, 3 ounces | 1.3 | 26 |
| Tuna, fresh, bluefin, cooked, 3 ounces | 1.2 | 24 |
| Avocados, raw, ½ avocado | 1 | 20 |
| Milk, 2% milkfat, 1 cup | 0.9 | 18 |
| Mushrooms, white, stir fried, ½ cup sliced | 0.8 | 16 |
| Potatoes, russet, flesh and skin, baked, 1 medium | 0.7 | 14 |
| Egg, hard boiled, 1 large | 0.7 | 14 |
| Greek yogurt, vanilla, nonfat, 5.3-ounce container | 0.6 | 12 |
| Ground beef, 85% lean meat, broiled, 3 ounces | 0.6 | 12 |
| Peanuts, roasted in oil, ¼ cup | 0.5 | 10 |
| Broccoli, boiled, ½ cup | 0.5 | 10 |
| Whole-wheat pita, 1 large | 0.5 | 10 |
| Chickpeas, canned, ½ cup | 0.4 | 8 |
| Rice, brown, medium grain, cooked, ½ cup | 0.4 | 8 |
| Oats, regular and quick, cooked with water, ½ cup | 0.4 | 8 |
| Cheese, cheddar, 1.5 ounces | 0.2 | 4 |
| Carrots, chopped, raw, ½ cup | 0.2 | 4 |
| Cabbage, boiled, ½ cup | 0.1 | 2 |
| Clementine, raw, 1 clementine | 0.1 | 2 |
| Tomatoes, raw, chopped or sliced, ½ cup | 0.1 | 2 |
| Cherry tomatoes, raw, ½ cup | 0 | 0 |
| Apple, raw, slices, ½ cup | 0 | 0 |
Footnote: *DV = Daily Value. The Daily Value (DV) for pantothenic acid is 5 mg for adults and children age 4 years and older. The U.S. Food and Drug Administration (FDA) does not require food labels to list pantothenic acid content unless pantothenic acid has been added to the food. Foods providing 20% or more of the DV (Daily Value) are considered to be high sources of a nutrient, but foods providing lower percentages of the DV (Daily Value) also contribute to a healthful diet.
[Source 100 ]Vitamin B5 supplement
Vitamin B5 (pantothenic acid) is available in dietary supplements containing only pantothenic acid, in combination with other B-complex dietary supplements and in some multivitamin or multimineral supplements 1. Vitamin B5 (pantothenic acid) in dietary supplements is often in the form of calcium pantothenate or pantethine (a dimeric form of pantetheine) 6, 91, 101. Research has not shown that any form of pantothenic acid is better than the others. The amount of pantothenic acid in dietary supplements typically ranges from about 10 mg in multivitamin or multimineral products to up to 1,000 mg in supplements of B-complex vitamins or pantothenic acid alone 1. Pantethine is used as a cholesterol-lowering agent in Japan and is available in the US as a dietary supplement 102.
What happens if I don’t get enough vitamin B5?
Vitamin B5 or Pantothenic acid deficiency is very rare in the United States because most people in the United States get enough vitamin B5 or pantothenic acid from their diet. However, people with severe malnutrition or people with a rare inherited disorder called pantothenate kinase-associated neurodegeneration mutation (PKAN) can’t use pantothenic acid properly. These disorders can lead to symptoms of pantothenic acid deficiency. Severe vitamin B5 or pantothenic acid deficiency can cause numbness and burning of the hands and feet, headache, extreme tiredness, irritability, restlessness, sleeping problems, stomach pain, heartburn, diarrhea, nausea, vomiting, and loss of appetite.
A common hallmark of individuals with pantothenate kinase-associated neurodegeneration mutation (PKAN) (formerly called Hallervorden-Spatz syndrome) is an accumulation of iron in the brain that forms a pattern called the “eye of the tiger” sign 15, 103. Pantothenate kinase-associated neurodegeneration mutation (PKAN) also presents with a progressive movement disorder, and other symptoms may vary significantly from case to case. Symptoms include dysarthria, dystonia, dysphasia, poor balance, spasticity, and muscle rigidity 15. Dementia, severe mental retardation and severe movement disability may develop at later stages 104. Rare clinical features include rigidity, parkinsonism, choreoathetosis, seizures, optic atrophy, and pigmentary retinopathy 15. Based on age at onset and rate of progression, PKAN can be classified in two major forms. In the classic form of PKAN, onset is usually in the first decade of life. Visual impairment caused by optic atrophy or retinal degeneration have been described in some classical cases 15. Atypical PKAN is presented in the second decade of life with slow progression 15. Neurobehavioral disorders and seizure are common in atypical form 105. All PKAN cases have mutation in pantothenate kinase 2 (PANK2) gene located on the short arm of chromosome 20 (20p13) 106. PANK2 encodes a mitochondrial pantothenate kinase which is the key regulatory enzyme in coenzyme A biosynthesis 107. Treatment of pantothenate kinase-associated neurodegeneration mutation (PKAN) focuses mainly on reducing symptoms. A few anecdotal reports indicate that vitamin B5 supplements can reduce symptoms, but the benefits of the general use of this supplement in PKAN are not known 108.
Vitamin B5 deficiency
Because some vitamin B5 (pantothenic acid) is present in almost all foods, vitamin B5 (pantothenic acid) deficiency is very rare in the United States except in people with severe malnutrition 6. When someone has a pantothenic acid (vitamin B5) deficiency, it is usually accompanied by deficiencies in other nutrients, making it difficult to identify the effects that are specific to pantothenic acid deficiency 2. The only individuals known to have developed pantothenic acid deficiency were fed diets containing virtually no pantothenic acid or were taking a pantothenic acid metabolic antagonist 5.
On the basis of the experiences of prisoners of war in World War II and studies of diets lacking vitamin B5 (pantothenic acid) in conjunction with administration of an antagonist of pantothenic acid metabolism, a vitamin B5 or pantothenic acid deficiency is associated with numbness and burning of the hands and feet, headache, fatigue, extreme tiredness, irritability, restlessness, disturbed sleep, and gastrointestinal disturbances such as stomach pain, heartburn, diarrhea, nausea, vomiting, and loss of appetite 2, 6, 109, 110, 13, 9.
The following group is most likely to have inadequate vitamin B5 (pantothenic acid) status:
Pantothenate kinase-associated neurodegeneration (PKAN)
Pantothenate kinase-associated neurodegeneration (PKAN) also known as Hallervorden-Spatz syndrome is a rare inherited neurological movement disorder characterized by the progressive degeneration of specific regions in the central nervous system (neurodegenerative disorder) and buildup of iron in the brain 111. Pantothenate kinase-associated neurodegeneration (PKAN) is characterized by progressive difficulty with movement, typically beginning in childhood. Movement abnormalities include involuntary muscle spasms, rigidity, and trouble with walking that worsens over time 112, 113, 103. Many people with pantothenate kinase-associated neurodegeneration (PKAN) also develop problems with speech (dysarthria), and some develop vision loss. Additionally, affected individuals may experience a loss of intellectual function (dementia) and psychiatric symptoms such as behavioral problems, personality changes, and depression.
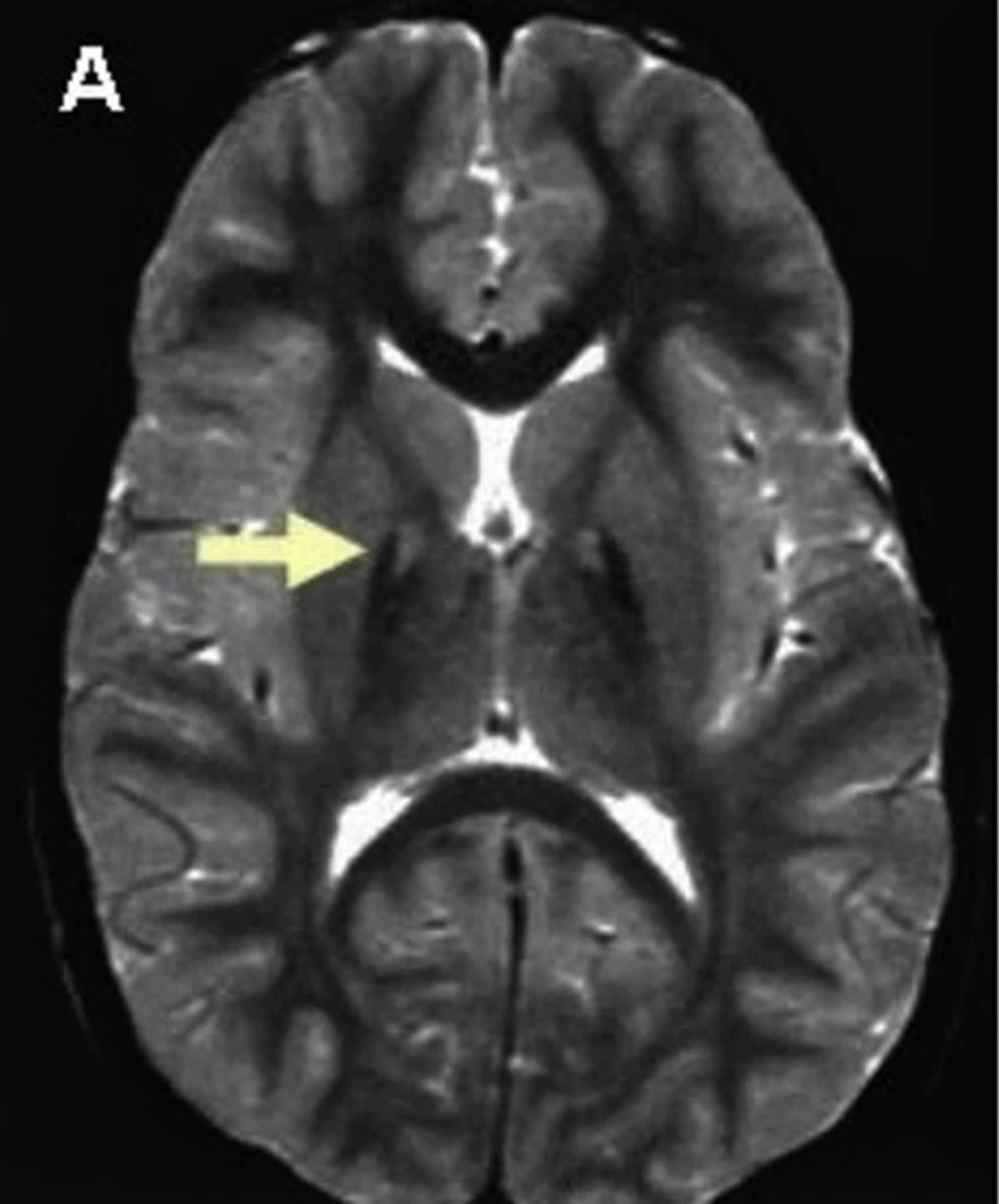
Pantothenate kinase-associated neurodegeneration (PKAN) is the most common type of neurodegeneration with brain iron accumulation (NBIA), a group of clinical disorders marked by progressive abnormal involuntary movements, alterations in muscle tone, and postural disturbances (extrapyramidal) 114, 15, 112, 115, 116, 105. Pantothenate kinase-associated neurodegeneration (PKAN) is also known as neurodegeneration with brain iron accumulation type 1 (NBIA type 1), which accounts for approximately half of the cases of neurodegeneration with brain iron accumulation (NBIA) 15. The neurodegeneration with brain iron accumulation (NBIA) disorders show radiographic evidence of iron accumulation in the brain, called the “eye-of-the-tiger sign”, which is typically seen on magnetic resonance imaging (MRI) scans of the brain in people with pantothenate kinase-associated neurodegeneration (PKAN) (Figure 5). The ‘eye of the tiger’ pattern of iron accumulation in the globus pallidus on T2 weighted magnetic resonance imaging (MRI) which is caused by iron deposition in the periphery (hypointensity) and necrosis on its core (hyperintensity) 117, 118.
Pantothenate kinase-associated neurodegeneration (PKAN) is inherited as an autosomal recessive genetic condition caused by mutations in the pantothenate kinase 2 (PANK2) gene, located on the short arm of chromosome 20 (20p13) 112. The PANK2 gene provides instructions for making an enzyme called pantothenate kinase 2 119. The pantothenate kinase 2 (PANK2) enzyme is active in specialized cellular structures called mitochondria, which are the cell’s energy-producing centers. Within mitochondria, pantothenate kinase 2 regulates the formation of a molecule called coenzyme A (CoA). Coenzyme A (CoA) is found in all living cells, where it is essential for the body’s production of energy from carbohydrates, fats, and some protein building blocks (amino acids).
PANK2 is one of four human genes that provide instructions for making versions of pantothenate kinase. The functions of these different versions probably vary among tissue types and parts of the cell. The version produced by the PANK2 gene is active in cells throughout the body, including nerve cells in the brain.
Vitamin B5 is required for the production of coenzyme A in cells. Disruption of this enzyme affects energy and lipid metabolism and may lead to accumulation of potentially harmful compounds in the brain, including iron. Currently, PANK2 is the only gene known to be associated with pantothenate kinase-associated neurodegeneration (PKAN).
Recessive genetic disorders occur when an individual inherits a non-working gene from each parent 112. If an individual receives one working gene and one non-working gene for the disease, the person will be a carrier for the disease, but usually will not show symptoms. The risk for two carrier parents to both pass the non-working gene and, therefore, have an affected child is 25% with each pregnancy. The risk to have a child who is a carrier, like the parents, is 50% with each pregnancy 112. The chance for a child to receive working genes from both parents is 25% 112. The risk is the same for males and females.
Pantothenate kinase-associated neurodegeneration (PKAN) clinical presentations include dystonia, dysarthria, and dysphasia. Dementia, severe mental retardation and severe movement disability may develop at later stages 104. Rare clinical features include rigidity, parkinsonism, choreoathetosis, seizures, optic atrophy, and pigmentary retinopathy.
Based on age at onset and rate of progression, pantothenate kinase-associated neurodegeneration (PKAN) is usually classified into two major forms: classic and atypical PKAN.
- Classic PKAN causes symptoms in the first 10 years of life, with symptoms that worsen rapidly. Visual impairment caused by optic atrophy or retinal degeneration have been described in some classical cases.
- Atypical PKAN usually occurs after the age of 10 and progresses more slowly. Neurobehavioral disorders and seizure are common in atypical form 105.
- Classic PKAN tends to have onset before 6 years of age, whereas atypical PKAN manifests at a mean age of 14 years 108. Some people have been diagnosed in infancy or adulthood, and some of those affected have characteristics that are between the two categories.
- Signs and symptoms vary, but the atypical PKAN is more likely than the classic PKAN to involve speech defects and psychiatric problems.
- Pantothenate kinase-associated neurodegeneration (PKAN) prevalence is estimated around 1 to 3 per million 108.
All individuals with PKAN have an abnormal buildup of iron in certain areas of the brain. A particular change, called the ‘eye-of-the-tiger sign’, which indicates a buildup of iron, is typically seen on magnetic resonance imaging (MRI) scans of the brain in people with this disorder.
A condition called HARP (hypoprebetalipoproteinemia, acanthocytosis, retinitis pigmentosa, and pallidal degeneration) syndrome, which was historically described as a separate syndrome, is now considered part of pantothenate kinase-associated neurodegeneration (PKAN) 103.
The manifestations of pantothenate kinase-associated neurodegeneration (PKAN) can include dystonia (contractions of opposing groups of muscles), spasticity, and pigmentary retinopathy 3, 6, 120. Its progression is rapid and leads to significant disability and loss of function 120. Treatment focuses primarily on reducing symptoms 121.
PKAN is typically diagnosed by molecular genetic testing, most often after a characteristic finding on magnetic resonance imaging (MRI), called the “eye-of-the-tiger” sign, is detected.
There is no specific treatment for individuals with pantothenate kinase-associated neurodegeneration (PKAN) 112. Treatment is directed towards the specific symptoms that appear in each individual and may include medication (such as botulinum toxin), surgery, deep brain stimulation and physical therapy. Whether pantothenate supplementation is beneficial in PKAN is not known, but some anecdotal reports indicate that supplements can reduce symptoms in some patients with atypical PKAN 108. Research is focusing on a better understanding of the underlying cause of this disorder, which may eventually help to find a more effective treatment.
Figure 6. Eye-of-the-tiger sign (MRI scan of the brain in pantothenate kinase-associated neurodegeneration (PKAN))
Footnote: T2 weighted brain magnetic resonance imaging (MRI) of a patient with pantothenate kinase-associated neurodegeneration (PKAN) which shows a central hyperintensity (bright spots) within substantia nigra and a surrounding area of hypointensity in globus pallidus (eye-of-the-tiger).
[Source 122 ]Pantothenate kinase-associated neurodegeneration (PKAN) causes
Pantothenate kinase-associated neurodegeneration (PKAN) is an autosomal recessive genetic condition caused by mutations in the pantothenate kinase 2 (PANK2) gene, located on the short arm of chromosome 20 (20p13) 106, 112. The PANK2 gene provides instructions for making an enzyme called pantothenate kinase 2 119. The pantothenate kinase 2 (PANK2) enzyme is active in specialized cellular structures called mitochondria, which are the cell’s energy-producing centers. Within mitochondria, pantothenate kinase 2 regulates the formation of a molecule called coenzyme A (CoA). Coenzyme A (CoA) is found in all living cells, where it is essential for the body’s production of energy from carbohydrates, fats, and some protein building blocks (amino acids).
PANK2 is one of four human genes that provide instructions for making versions of pantothenate kinase. The functions of these different versions probably vary among tissue types and parts of the cell. The version produced by the PANK2 gene is active in cells throughout the body, including nerve cells in the brain.
Individuals with PKAN have abnormal accumulation of iron in certain areas of the brain. This is especially seen in regions of the basal ganglia called the globus pallidus and the substantia nigra 112. The basal ganglia is a collection of structures deep within the base of the brain that assist in regulating movements. The exact relationship between iron accumulation and the symptoms of PKAN is not fully understood 112.
Pantothenate kinase-associated neurodegeneration (PKAN) signs and symptoms
The common feature among all individuals with pantothenate kinase-associated neurodegeneration (PKAN) is iron accumulation in the brain, in a pattern called the ‘eye of the tiger sign,’ along with a progressive movement disorder. Symptoms may vary greatly from case to case. In most cases, progression of the disease extends over several years, leading to death in childhood or early adulthood in classic PKAN cases 112. Some patients experience rapid deterioration and die within 1-2 years 112. Others have a slower progression or can plateau for long periods of time and continue to function into the third decade of life. Atypical individuals often retain a high level of function into later adulthood and some are known to be living in their sixties to seventies 112.
Pantothenate kinase-associated neurodegeneration (PKAN) symptoms include dystonia, (sustained muscle contractions causing repetitive movements), dysarthria (abnormal speech), muscular rigidity, poor balance, and spasticity (sudden involuntary muscle spasms), These features can result in clumsiness, gait (walking) problems, difficulty controlling movement, and speech problems. Another common feature is degeneration of the retina, resulting in progressive night blindness and loss of peripheral (side) vision.
Dystonia is characterized by involuntary muscle contractions that may force certain body parts into unusual, and sometimes painful, movements and positions. In addition, there may be stiffness in the arms and legs because of continuous resistance to muscle relaxing (spasticity) and abnormal tightening of the muscles (muscular rigidity). Spasticity and muscle rigidity usually begin in the legs and later develop in the arms. As affected individuals age, they may eventually lose control of voluntary movements. Muscle spasms combined with decreased bone mass can result in bone fractures (not caused by trauma or accident).
Dystonia affects the muscles in the mouth and throat, which may cause dysarthria and difficulty swallowing (dysphagia). The progression of dystonia in these muscles can result in loss of speech as well as tongue-biting and difficulty with eating.
Specific forms of dystonia that may occur in association with PKAN include blepharospasm and torticollis. Blepharospasm is a condition in which the muscles of the eyelids do not function properly, resulting in excessive blinking and involuntary closing of the eyelids. Torticollis is a condition in which there are involuntary contractions of neck muscles resulting in abnormal movements and positions of the head and neck.
Many of the delays in development pertain to motor skills (movement), although a small subgroup may have intellectual delays. Although intellectual impairment has often been described as a part of the condition in the past, it is unclear if this is a true feature. Intellectual testing may be hampered by the movement disorder; therefore, newer methods of studying intelligence are necessary to determine if there are any cognitive features of this condition.
The symptoms and physical findings associated with PKAN gene mutations can be distinguished between classical and atypical disease. Individuals with classical disease have a more rapid progression of symptoms. In most cases, atypical disease progresses slowly over several years. The symptoms and physical findings vary from case to case.
Classical PKAN develops in the first ten years of life (average age for developing symptoms is three and a half years). These children may initially be perceived as clumsy and later develop more noticeable problems with walking. Speech delay is also common. Eventually, falling becomes a frequent feature. Because of the limited ability to protect themselves during falls, children may have repeated injury to the face and chin. Many individuals with the classic form of PKAN require a wheelchair by their mid-teens (in some cases earlier). Most lose the ability to move/walk independently between 10 and15 years after the beginning of symptoms.
Individuals with classical PKAN are more likely to have specific eye problems. Approximately two-thirds of these patients will have retinal degeneration. This is a progressive degeneration of the nerve-rich membrane lining the eyes (retina), resulting in tunnel vision, night blindness, and loss of peripheral vision. Loss of this peripheral vision may contribute to the more frequent falls and gait disturbances in the early stages. [For more information on this retinopathy (retinitis pigmentosa), choose “retinitis pigmentosa” as your search term in the Rare Disease Database].
The atypical form of PKAN usually occurs after the age of ten years and progresses more slowly. The average age for developing symptoms is 13 years. Loss of independent ambulation (walking) often occurs 15 to 40 years after the initial development of symptoms. The initial presenting symptoms usually involve speech. Common speech problems are repetition of words or phrases (palilalia), rapid speech (tachylalia), and dysarthria. Psychiatric symptoms are more commonly observed and include impulsive behavior, violent outbursts, depression, or a tendency to rapid mood swings. While the movement disorder is a very common feature, it usually develops later. In general, atypical disease is less severe and more slowly progressive than early-onset PKAN.
In cases of neurodegeneration with brain iron accumulation (NBIA) that are not caused by PKAN, the movement-related symptoms (such as dystonia) may be very similar. Nine additional genes causing various subtypes of NBIA have been identified at this time. For those without a specific diagnosis or known cause of NBIA, symptoms are more varied because there are probably several different causes of neurodegeneration in this group. There is a subgroup of patients with moderate to severe intellectual disability. Also, seizure disorders are more common among non-PKAN individuals.
Pantothenate kinase-associated neurodegeneration (PKAN) diagnosis
The diagnosis of pantothenate kinase-associated neurodegeneration (PKAN) is made based upon a detailed patient history, a thorough clinical evaluation, and a variety of specialized tests 112. PKAN is typically suspected when the characteristic brain MRI finding called the “eye-of-the-tiger” sign, which is a dark area indicating accumulation of iron with a bright spot in the center, is observed on T2-weighted MRI. This MRI finding is not seen in other forms of neurodegeneration with brain iron accumulation (NBIA).
Molecular genetic testing for the full gene sequence of the PANK2 gene is the gold standard way to make this diagnosis 112. Approximately 95% of those affected have two identifiable mutations in the PANK2 gene and approximately 5% have only one identifiable mutation. Some PANK2 gene deletions are not detected by sequencing the gene, so for individuals without a detectable mutation or only one detectable mutation, gene deletion/duplication analysis is also recommended 112.
Pantothenate kinase-associated neurodegeneration (PKAN) treatment
There is no specific treatment for individuals with pantothenate kinase-associated neurodegeneration (PKAN) 112. Treatment is directed towards the specific symptoms that appear in each individual. Research is focusing on a better understanding of the underlying cause of this disorder, which may eventually help to find a more comprehensive treatment.
Treatment may require the coordinated efforts of a team of specialists. Physicians that the family may work with include the pediatrician or internist, neurologist, ophthalmologist, physiatrist and geneticist. A team approach to supportive therapy may include physical therapy, exercise physiology, occupation therapy, speech pathology and nutrition/feeding. In addition, many families may benefit from genetic counseling.
The most consistent forms of relief from disabling dystonia are baclofen, trihexyphenidyl, and clonazepam. These medications can be taken orally. Later in disease, a baclofen pump can be used to administer regular doses automatically into the central nervous system. Intramuscular botulinum toxin may also help treat specific regions where dystonia is problematic.
Levodopa/carbidopa does not generally appear to help patients with PKAN, although there may be exceptions. These treatments may have a role in the treatment of other causes of NBIA; however, their overall effectiveness is unknown and the responsiveness in individual cases is unpredictable.
Drugs that reduce the levels of iron in the body (iron chelation) have been attempted to treat individuals with PKAN 112. These early agents were proven ineffective and can cause anemia 112. A clinical trial of the drug deferiprone was completed for PKAN and results were published in 2019. The results suggested a possible modest slowing of disease progression, although the statistical analysis of the data was not able to prove this as significant 123, 124.
Pallidotomy and thalamotomy have been investigational attempts at controlling dystonia. These are both surgical techniques which destroy (ablate) very specific regions of the brain, the globus pallidus and thalamus, respectively. Some families have reported some immediate and temporary relief. However, most patients return to their pre-operative level of dystonia within one year of the operation 112. Deep brain stimulation (DBS) of the globus pallidus has been found to have promising results in some patients with PKAN and NBIA and is now favored over ablative procedures 112.
Individuals experiencing seizures usually benefit from standard anti-convulsive drugs 112. In addition, standard approaches to pain management are generally recommended where there is no identifiable treatment for the underlying cause of pain. Referral to pediatric palliative care specialists can be highly beneficial during later disease stages.
The association between pantothenate kinase and PKAN suggests that supplemental pantothenate (pantothenic acid, calcium pantothenate) taken orally could be beneficial. Pantothenate is another name for vitamin B5, a water soluble vitamin. Theoretically, this is most likely to assist individuals with very low levels of pantothenate kinase activity (atypical PKAN). It is hypothesized that classic PKAN results from complete absence of the enzyme pantothenate kinase, whereas atypical PKAN results from a severe deficiency, although the individuals still may have some level of enzyme activity. Clinical trials are needed to investigate the effectiveness of this treatment 112.
The benefits and limitations of any of the above treatments should be discussed in detail with a physician.
Vitamin B5 deficiency symptoms
Because vitamin B5 or pantothenic acid is widely distributed in nature and deficiency is extremely rare in humans, most information regarding the consequences of vitamin B5 deficiency has been gathered from experimental research in animals 22, 18. The most common symptoms of vitamin B5 or pantothenic acid in animals are growth problems, skin rash, gastrointestinal and nervous symptoms, such as ataxia, loss of coordination, and muscle weakness 18. Similar symptoms appeared also in human studies. Symptoms are described in more detail in Table 3. The diversity of symptoms emphasizes the numerous functions of pantothenic acid in its coenzyme forms.
Humans administered with a vitamin B5 antagonist omega-methyl pantothenic acid developed personality changes with irritability, restlessness, and quarrelsomeness 18. Similar symptoms developed in humans on a diet deficient in vitamin B5 content (8 weeks) 125. An analogous experiment was performed, as well, by Fry et al., in 1976 126, who tested the effect of a diet essentially free from pantothenic acid on human health. In that study, however, no clinical symptoms of deficiency were observed, but some subjects appeared listless and complained of fatigue at the end of diet deficient period (63 days) 126.
Lower levels of pantothenic acid were also detected in some brain regions affected by Alzheimer’s disease compared with controls. It is still unknown whether vitamin B5 depletion participates in the pathophysiology or if this is simply a consequence of the underlying neuropathological process 127.
Vitamin B5 or pantothenic acid deficiency in rats can cause damage to the adrenal glands, breeding problems and failure of embryo implementation with subsequent resorption 128. Vitamin B5 or pantothenic acid deficiency in rats throughout pregnancy has an impact on endocrine function of the placenta, which is linked to a lower production of progesterone and acetylcholine, and underdevelopment of fetuses 129. Among the reported abnormalities in rats include: cerebral and eye defects, digital hemorrhages and edema, interventricular septal defects, anomalies of the aortic arch pattern, hydronephrosis and hydroureter, clubfoot, tail defects, cleft palate, and dermal defects 128.
While vitamin B5 or pantothenic acid-deficient monkeys developed anemia due to decreased synthesis of heme, a component of hemoglobin 8. Dogs with vitamin B5 or pantothenic acid deficiency developed low blood glucose, rapid breathing and heart rates, and convulsions 8. Chickens developed skin irritation, feather abnormalities, and spinal nerve damage associated with the degeneration of the myelin sheath 8. Pantothenic acid-deficient mice showed decreased exercise tolerance and diminished storage of glucose (in the form of glycogen) in muscle and liver 8. Mice also developed skin irritation and graying of the fur, which is reversed by pantothenic acid administration 8.
Table 3. Vitamin B5 or pantothenic acid deficiency symptoms
| Species | Symptoms | Sources |
|---|---|---|
| Humans | Nervous system: headache, irritability, restlessness, quarrelsomeness, excessive fatigue, numbness, paresthesia, muscle cramps, faulty coordination associated with tremor and peculiar gait | 125 |
| Digestive track: abdominal rumbling, diarrhea, epigastric burning, regurgitation | ||
| Glands: loss of eosinophilic response to adrenocorticotropic hormone, increased sensitivity to insulin | ||
| Rodents (rats, mice, guinea pigs) | Growth: retardation, decrease in weight | 130, 131, 132, 133, 134 |
| Skin and mucosa: ruffing and discoloration of the fur, thinning of hair, alopecia, dryness of the skin with scaly desquamation, nasal discharge, watering of the eyes | ||
| Digestive track: diarrhea, duodenal changes (Lieberkühn crypts—enlargement, hyperplasia, increase in space between crypts, atrophy; villi diminution, epithelial changes to cuboid or flat, leading to ulcerations, perforation and chronic lesions), salivation | ||
| Nervous system: muscle weakness of the hind legs, convulsions, coma | ||
| Glands: adrenal lesions | ||
| Birds (ducklings and chicks) | Growth: retardation, decrease in weight | 132, 135, 136 |
| Skin: scaly dermatitis, skin lesions, scabs around beak and eyes, feather depigmentation, dermal edema | ||
| Nervous system: severe ataxia, tendency to fall and inability to rise and laying panting | ||
| Glands: lymphoid cell necrosis in the bursa of Fabricius and the thymus, and a lymphocytic paucity in the spleen | ||
| Pigs | Growth: failure to gain in weight, loss of appetite | 137, 138, 139 |
| Skin: loss of hair, roughness of the coat | ||
| Digestive track: diarrhea, severe colonic lesions | ||
| Nervous system: ataxia, lesions in sensory neurons, sudden lifting one of the limbs from the ground, unusual walk, inability to walk or stand | ||
| Respiratory system: cough and nasal secretion | ||
| Dogs | Growth: retardation Nervous system: sudden weakness, coma, rapid respiratory and heart rate, convulsions, spasticity of the hind legs | 140, 141 |
| Digestive track: decreased appetite, gastrointestinal symptoms, gastritis or enteritis | ||
| Glands: fatty liver, mottled thymusis | ||
| Blood: blood level of glucose and chlorides were lower and non-protein nitrogen was elevated | ||
| Urinary system: hemorrhagic kidney degeneration |
Vitamin B5 safety
Vitamin B5 or pantothenic acid is considered safe, even at high doses 142, 61. However, taking very high doses of vitamin B5 or pantothenic acid supplements (such as 10,000 mg to 20,000 mg/day) can cause an upset stomach and diarrhea, but the mechanism for this effect is not known 2, 61. However, there is one case report of life-threatening eosinophilic pleuropericardial effusion in an elderly woman who took a combination of 10 mg/day of biotin and 300 mg/day of pantothenic acid for two months 143. She was hospitalized with chest pain and breathing problems. Blood tests showed an inflammatory syndrome with a high eosinophil concentration (1200–1500 cells/mm³) 144. Due to the lack of reports of adverse effects when the Dietary Reference Intakes (DRI) for pantothenic acid were established in 1998, the Food and Nutrition Board of the Institute of Medicine did not establish a tolerable upper intake level (UL) for pantothenic acid 145. Pantethine is generally well tolerated in doses up to 1,200 mg/day. However, gastrointestinal side effects, such as nausea and heartburn, have been reported 102. Also, topical formulations containing up to 5% of dexpanthenol (D-panthenol) have been safely used for up to one month. Yet, a few cases of skin irritation, contact dermatitis, and eczema have been reported with the use of dexpanthenol-containing ointments 146, 147.
Vitamin B5 contraindications
Vitamin B5 (pantothenic acid) contraindications include patients with hypersensitivity or allergy to the drug or any of its derivatives. A report suggests that vitamin B5 (pantothenic acid) intake might correlate with increased cerebral amyloid-beta peptide burden in individuals with cognitive impairment 148. Although further studies are still needed to confirm the findings and discover the molecular mechanisms of this pathway, the current research suggests those with cognitive impairment to be a potential contraindication 148.
Interactions with medications or other supplements
Large doses of vitamin B5 (pantothenic acid) have the potential to compete with biotin for intestinal and cellular uptake by the human sodium-dependent multivitamin transporter (hSMVT) 149, 150.
Oral contraceptives (birth control pills) containing estrogen and progestin may increase the requirement for pantothenic acid 142. Use of pantethine in combination with cholesterol-lowering drugs called statins (HMG-CoA reductase inhibitors) or with nicotinic acid (niacin) may produce additive effects on blood lipids 102.
The following drugs have moderate interactions with vitamin B5 (pantothenic acid) 14:
- Azithromycin
- Clarithromycin
- Erythromycin base
- Erythromycin ethylsuccinate
- Erythromycin lactobionate
- Erythromycin stearate
- Roxithromycin
Furthermore, there are at least 60 other drugs that have mild interactions with vitamin B5 (pantothenic acid).
References- Pantothenic Acid. https://ods.od.nih.gov/factsheets/PantothenicAcid-HealthProfessional
- Miller JW, Rucker RB. Pantothenic acid. In: Erdman JW, Macdonald IA, Zeisel SH, eds. Present Knowledge in Nutrition. 10th ed. Washington, DC: Wiley-Blackwell; 2012:375-90.
- Sweetman L. Pantothenic acid. In: Coates PM, Betz JM, Blackman MR, et al., eds. Encyclopedia of Dietary Supplements. 2nd ed. London and New York: Informa Healthcare; 2010:604-11.
- Trumbo PR. Pantothenic acid. In: Ross AC, Caballero B, Cousins RJ, Tucker KL, Ziegler TR, eds. Modern Nutrition in Health and Disease. 11th ed. Baltimore: Lippincott Williams & Wilkins; 2014:351-357.
- Institute of Medicine. Food and Nutrition Board. Dietary Reference Intakes: Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline. Washington, DC: National Academy Press; 1998.
- Trumbo PR. Pantothenic acid. In: Ross AC, Caballero B, Cousins RJ, et al., eds. Modern Nutrition in Health and Disease. 11th ed. Baltimore, MD: Lippincott Williams & Wilkins; 2014:351-7.
- Martinez DL, Tsuchiya Y, Gout I. Coenzyme A biosynthetic machinery in mammalian cells. Biochem Soc Trans. 2014 Aug;42(4):1112-7. doi: 10.1042/BST20140124
- Pantothenic Acid. https://lpi.oregonstate.edu/mic/vitamins/pantothenic-acid
- Bauerly K, Rucker RB. Pantothenic acid. In: Zempleni J, Rucker RB, McCormick DB, Suttie JW, eds. Handbook of vitamins. 4th ed. Boca Raton: CRC Press; 2007:289-314.
- Iyenga GV, Wolfe WR, Tanner JT, Morris ER. Content of minor and trace elements, and organic nutrients in representative mixed total diet composites from the USA. Sci Total Environ. 2000 Jul 10;256(2-3):215-26. doi: 10.1016/s0048-9697(00)00494-0
- Provincial Epidemiology Service, New Brunswick Department of Health and Wellness. New Brunswick nutrition survey; 1997.
- Tarr JB, Tamura T, Stokstad EL. Availability of vitamin B6 and pantothenate in an average American diet in man. Am J Clin Nutr. 1981 Jul;34(7):1328-37. doi: 10.1093/ajcn/34.7.1328
- Hodges RE, Ohlson MA, Bean WB. Pantothenic acid deficiency in man. J Clin Invest. 1958 Nov;37(11):1642-57. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1062846/pdf/jcinvest00347-0188.pdf
- Sanvictores T, Chauhan S. Vitamin B5 (Pantothenic Acid) [Updated 2022 Oct 12]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK563233
- Dezfouli MA, Jaberi E, Alavi A, Rezvani M, Shahidi G, Elahi E, Rohani M. Pantothenate kinase 2 mutation with eye-of-the-tiger sign on magnetic resonance imaging in three siblings. Iran J Neurol. 2012;11(4):155-8. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3829266
- Kurian MA, Hayflick SJ. Pantothenate kinase-associated neurodegeneration (PKAN) and PLA2G6-associated neurodegeneration (PLAN): review of two major neurodegeneration with brain iron accumulation (NBIA) phenotypes. Int Rev Neurobiol. 2013;110:49-71. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6059649
- Chapter 16 – Pantothenic acid. Present Knowledge in Nutrition (Eleventh Edition). Volume 1: Basic Nutrition and Metabolism. 2020, Pages 273-287. https://doi.org/10.1016/B978-0-323-66162-1.00016-0
- Hrubša M, Siatka T, Nejmanová I, Vopršalová M, Kujovská Krčmová L, Matoušová K, Javorská L, Macáková K, Mercolini L, Remião F, Máťuš M, Mladěnka P, On Behalf Of The Oemonom. Biological Properties of Vitamins of the B-Complex, Part 1: Vitamins B1, B2, B3, and B5. Nutrients. 2022 Jan 22;14(3):484. doi: 10.3390/nu14030484
- Rucker R.B. Pantothenic Acid. Academic Press; Cambridge, MA, USA: 2016.
- Czumaj A., Szrok-Jurga S., Hebanowska A., Turyn J., Swierczynski J., Sledzinski T., Stelmanska E. The Pathophysiological Role of CoA. Int. J. Mol. Sci. 2020;21:9057. doi: 10.3390/ijms21239057
- Mindrebo J.T., Patel A., Misson L.E., Kim W.E., Davis T.D., Ni Q.Z., La Clair J.J., Burkart M.D. 1.04-Structural Basis of Acyl-Carrier Protein Interactions in Fatty Acid and Polyketide Biosynthesis. In: Hung-Wen L., Begley T.P., editors. Comprehensive Natural Products III. Elsevier; Amsterdam, The Netherlands: 2020.
- Miller JW, Rucker RB. Pantothenic acid. In: Erdman JWJ, Macdonald IA, Zeisel SH, eds. Present Knowledge in Nutrition. 10th ed. Ames: Wiley-Blackwell; 2012:375-390.
- Takahashi A, Mizusawa K. Posttranslational modifications of proopiomelanocortin in vertebrates and their biological significance. Front Endocrinol (Lausanne). 2013 Oct 17;4:143. doi: 10.3389/fendo.2013.00143
- Choudhary C, Weinert BT, Nishida Y, Verdin E, Mann M. The growing landscape of lysine acetylation links metabolism and cell signalling. Nat Rev Mol Cell Biol. 2014 Aug;15(8):536-50. doi: 10.1038/nrm3841
- Bunkoczi G, Pasta S, Joshi A, Wu X, Kavanagh KL, Smith S, Oppermann U. Mechanism and substrate recognition of human holo ACP synthase. Chem Biol. 2007 Nov;14(11):1243-53. doi: 10.1016/j.chembiol.2007.10.013
- Donato H, Krupenko NI, Tsybovsky Y, Krupenko SA. 10-formyltetrahydrofolate dehydrogenase requires a 4′-phosphopantetheine prosthetic group for catalysis. J Biol Chem. 2007 Nov 23;282(47):34159-66. doi: 10.1074/jbc.M707627200
- Strickland KC, Hoeferlin LA, Oleinik NV, Krupenko NI, Krupenko SA. Acyl carrier protein-specific 4′-phosphopantetheinyl transferase activates 10-formyltetrahydrofolate dehydrogenase. J Biol Chem. 2010 Jan 15;285(3):1627-33. doi: 10.1074/jbc.M109.080556
- Strickland KC, Krupenko NI, Dubard ME, Hu CJ, Tsybovsky Y, Krupenko SA. Enzymatic properties of ALDH1L2, a mitochondrial 10-formyltetrahydrofolate dehydrogenase. Chem Biol Interact. 2011 May 30;191(1-3):129-36. doi: 10.1016/j.cbi.2011.01.008
- Rumberger JA, Napolitano J, Azumano I, Kamiya T, Evans M. Pantethine, a derivative of vitamin B(5) used as a nutritional supplement, favorably alters low-density lipoprotein cholesterol metabolism in low- to moderate-cardiovascular risk North American subjects: a triple-blinded placebo and diet-controlled investigation. Nutr Res. 2011 Aug;31(8):608-15. doi: 10.1016/j.nutres.2011.08.001
- McRae MP. Treatment of hyperlipoproteinemia with pantethine: a review and analysis of efficacy and tolerability. 2005. In: Database of Abstracts of Reviews of Effects (DARE): Quality-assessed Reviews [Internet]. York (UK): Centre for Reviews and Dissemination (UK); 1995-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK71836
- Chen YQ, Zhao SP, Zhao YH. Efficacy and tolerability of coenzyme A vs pantethine for the treatment of patients with hyperlipidemia: A randomized, double-blind, multicenter study. J Clin Lipidol. 2015 Sep-Oct;9(5):692-7. doi: 10.1016/j.jacl.2015.07.003
- Evans M, Rumberger JA, Azumano I, Napolitano JJ, Citrolo D, Kamiya T. Pantethine, a derivative of vitamin B5, favorably alters total, LDL and non-HDL cholesterol in low to moderate cardiovascular risk subjects eligible for statin therapy: a triple-blinded placebo and diet-controlled investigation. Vasc Health Risk Manag. 2014 Feb 27;10:89-100. doi: 10.2147/VHRM.S57116
- Weimann BI, Hermann D. Studies on wound healing: effects of calcium D-pantothenate on the migration, proliferation and protein synthesis of human dermal fibroblasts in culture. Int J Vitam Nutr Res. 1999 Mar;69(2):113-9. doi: 10.1024/0300-9831.69.2.113
- Wiederholt, T., Heise, R., Skazik, C., Marquardt, Y., Joussen, S., Erdmann, K., Schröder, H., Merk, H.F. and Baron, J.M. (2009), Calcium pantothenate modulates gene expression in proliferating human dermal fibroblasts. Experimental Dermatology, 18: 969-978. https://doi.org/10.1111/j.1600-0625.2009.00884.x
- Kobayashi D, Kusama M, Onda M, Nakahata N. The effect of pantothenic acid deficiency on keratinocyte proliferation and the synthesis of keratinocyte growth factor and collagen in fibroblasts. J Pharmacol Sci. 2011;115(2):230-4. doi: 10.1254/jphs.10224sc
- Heise R, Skazik C, Marquardt Y, Czaja K, Sebastian K, Kurschat P, Gan L, Denecke B, Ekanayake-Bohlig S, Wilhelm KP, Merk HF, Baron JM. Dexpanthenol modulates gene expression in skin wound healing in vivo. Skin Pharmacol Physiol. 2012;25(5):241-8. doi: 10.1159/000341144
- Vaxman F, Olender S, Lambert A, Nisand G, Aprahamian M, Bruch JF, Didier E, Volkmar P, Grenier JF. Effect of pantothenic acid and ascorbic acid supplementation on human skin wound healing process. A double-blind, prospective and randomized trial. Eur Surg Res. 1995;27(3):158-66. doi: 10.1159/000129395
- Vaxman F, Olender S, Lambert A, Nisand G, Grenier JF. Can the wound healing process be improved by vitamin supplementation? Experimental study on humans. Eur Surg Res. 1996 Jul-Aug;28(4):306-14. doi: 10.1159/000129471
- Celebi S, Tepe C, Yelken K, Celik O. Efficacy of dexpanthenol for pediatric post-tonsillectomy pain and wound healing. Ann Otol Rhinol Laryngol. 2013 Jul;122(7):464-7. doi: 10.1177/000348941312200710
- Capodice J.L. Feasibility, Tolerability, Safety and Efficacy of a Pantothenic Acid Based Dietary Supplement in Subjects with Mild to Moderate Facial Acne Blemishes. J. Cosmet. Dermatol. Sci. Appl. 2012;2:132–135. doi: 10.4236/jcdsa.2012.23026
- Yang M, Moclair B, Hatcher V, Kaminetsky J, Mekas M, Chapas A, Capodice J. A randomized, double-blind, placebo-controlled study of a novel pantothenic Acid-based dietary supplement in subjects with mild to moderate facial acne. Dermatol Ther (Heidelb). 2014 Jun;4(1):93-101. doi: 10.1007/s13555-014-0052-3
- Udompataikul M, Limpa-o-vart D. Comparative trial of 5% dexpanthenol in water-in-oil formulation with 1% hydrocortisone ointment in the treatment of childhood atopic dermatitis: a pilot study. J Drugs Dermatol. 2012 Mar;11(3):366-74.
- Kuwahara, R.T. and Skinner, R.B. (2002), Granuloma Annulare Resolved With Topical Application Of Imiquimod. Pediatric Dermatology, 19: 368-371. https://doi.org/10.1046/j.1525-1470.2002.00105.x
- Proksch E., de Bony R., Trapp S., Boudon S. Topical use of dexpanthenol: A 70th anniversary article. J. Dermatol. Treat. 2017;28:766–773. doi: 10.1080/09546634.2017.1325310
- Wollina U., Kubicki J. Dexpanthenol supports healing of superficial wounds and injuries. Kosm. Med. 2006;27:240–249.
- Proksch E., Nissen H.P. Dexpanthenol enhances skin barrier repair and reduces inflammation after sodium lauryl sulphate-induced irritation. J. Dermatol. Treat. 2002;13:173–178. doi: 10.1080/09546630212345674
- Stettler H., Kurka P., Lunau N., Manger C., Bohling A., Bielfeldt S., Wilhelm K.P., Dahnhardt-Pfeiffer S., Dahnhardt D., Brill F.H., et al. A new topical panthenol-containing emollient: Results from two randomized controlled studies assessing its skin moisturization and barrier restoration potential, and the effect on skin microflora. J. Dermatol. Treat. 2017;28:173–180. doi: 10.1080/09546634.2016.1214235
- Heise R., Schmitt L., Huth L., Krings L., Kluwig D., Katsoulari K.V., Steiner T., Holzle F., Baron J.M., Huth S. Accelerated wound healing with a dexpanthenol-containing ointment after fractional ablative CO2 laser resurfacing of photo-damaged skin in a randomized prospective clinical trial. Cutan. Ocul. Toxicol. 2019;38:274–278. doi: 10.1080/15569527.2019.1597879
- Wananukul S., Limpongsanuruk W., Singalavanija S., Wisuthsarewong W. Comparison of dexpanthenol and zinc oxide ointment with ointment base in the treatment of irritant diaper dermatitis from diarrhea: A multicenter study. J. Med. Assoc. Thai. 2006;89:1654–1658.
- Olsavszky R., Nanu E.A., Macura-Biegun A., Kurka P., Trapp S. Skin barrier restoration upon topical use of two 5% dexpanthenol water-in-oil formulations on freshly tattooed skin: Results from a single-blind prospective study. Wounds Int. 2019;10:33–39.
- Shanazi M., Farshbaf Khalili A., Kamalifard M., Asghari Jafarabadi M., Masoudin K., Esmaeli F. Comparison of the Effects of Lanolin, Peppermint, and Dexpanthenol Creams on Treatment of Traumatic Nipples in Breastfeeding Mothers. J. Caring Sci. 2015;4:297–307. doi: 10.15171/jcs.2015.030
- Hamdi I.M. Effect of D-Panthenol on Corneal Epithelial Healing after Surface Laser Ablation. J. Ophthalmol. 2018;2018:6537413. doi: 10.1155/2018/6537413
- Gobbels M., Gross D. Clinical study of the effectiveness of a dexpanthenol containing artificial tears solution (Siccaprotect) in treatment of dry eyes. Klin. Monbl. Augenheilkd. 1996;209:84–88. doi: 10.1055/s-2008-1035283
- Jagade M.V., Langade D.G., Pophale R.R., Prabhu A. Oxymetazoline plus dexpanthenol in nasal congestion. Indian J. Otolaryngol. Head Neck Surg. 2008;60:393–397. doi: 10.1007/s12070-008-0125-7
- Kehrl W., Sonnemann U., Dethlefsen U. Advance in therapy of acute rhinitis—Comparison of efficacy and safety of xylometazoline in combination xylometazoline-dexpanthenol in patients with acute rhinitis. Laryngo-Rhino-Otologie. 2003;82:266–271. doi: 10.1055/s-2003-38941
- Kehrl W., Sonnemann U. Improving wound healing after nose surgery by combined administration of xylometazoline and dexpanthenol. Laryngo-Rhino-Otologie. 2000;79:151–154. doi: 10.1055/s-2000-295
- Pantothenic Acid. https://lpi.oregonstate.edu/mic/vitamins/pantothenic-acid#disease-treatment
- Berry Ottaway P. Stability of vitamins during food processing and storage. In: Skibsted L.H., Risbo J., Andersen M.L., editors. Chemical Deterioration and Physical Instability of Food and Beverages. Woodhead Publishing; Cambridge, UK: 2010.
- Walsh J.H., Wyse B.W., Hansen R.G. Pantothenic acid content of 75 processed and cooked foods. J. Am. Diet. Assoc. 1981;78:140–144. doi: 10.1016/S0002-8223(21)04766-0
- Garg M., Sharma A., Vats S., Tiwari V., Kumari A., Mishra V., Krishania M. Vitamins in Cereals: A Critical Review of Content, Health Effects, Processing Losses, Bioaccessibility, Fortification, and Biofortification Strategies for Their Improvement. Front. Nutr. 2021;8:586815. doi: 10.3389/fnut.2021.586815
- Chawla J, Kvarnberg D. Hydrosoluble vitamins. Handb Clin Neurol. 2014;120:891-914. doi: 10.1016/B978-0-7020-4087-0.00059-0
- MacDonald R., Reitmeier C., editors. Understanding Food Systems. Academic Press; Cambridge, MA, USA: 2017. Food Processing; pp. 179–225.
- Kyritsi A., Tzia C., Karathanos V.T. Vitamin fortified rice grain using spraying and soaking methods. LWT Food Sci. Technol. 2011;44:312–320. doi: 10.1016/j.lwt.2010.06.001
- Miller J.W., Rucker R.B. Present Knowledge in Nutrition. John Wiley & Sons, Inc.; Hoboken, NJ, USA: 2012. Pantothenic Acid; pp. 375–390.
- Gwirtz J.A., Garcia-Casal M.N. Processing maize flour and corn meal food products. Ann. N. Y. Acad. Sci. 2014;1312:66–75. doi: 10.1111/nyas.12299
- Schroeder H.A. Losses of vitamins and trace minerals resulting from processing and preservation of foods. Am. J. Clin. Nutr. 1971;24:562–573. doi: 10.1093/ajcn/24.5.562
- Tiozon R.N., Fernie A.R., Sreenivasulu N. Meeting human dietary vitamin requirements in the staple rice via strategies of biofortification and post-harvest fortification. Trends Food Sci. Technol. 2021;109:65–82. doi: 10.1016/j.tifs.2021.01.023
- Godoy H.T., Amaya-Farfan J., Rodriguez-Amaya D.B. Degradation of vitamins. In: Rodriguez-Amaya D.B., Amaya-Farfan J., editors. Chemical Changes During Processing and Storage of Foods. Academic Press; Cambridge, MA, USA: 2021. pp. 329–383.
- Bognár A. Tables on Weight Yield of Food and Retention Factors of Food Constituents for the Calculation of Nutrient Composition of Cooked Foods (Dishes) Bundesforschungsanstalt für Ernährung; Karlsruhe, Germany: 2002.
- Bell S., Becker W., Vásquez-Caicedo A., Hartmann B., Møller A., Butriss J. Report on Nutrient Losses and Gains Factors Used in European Food Composition Databases. Federal Research Centre for Nutrition and Food; Karlsruhe, Germany: 2006.
- Lešková E., Kubíková J., Kováčiková E., Košická M., Porubská J., Holčíková K. Vitamin losses: Retention during heat treatment and continual changes expressed by mathematical models. J. Food Compos. Anal. 2006;19:252–276. doi: 10.1016/j.jfca.2005.04.014
- Engler P.P., Bowers J.A. B-vitamin retention in meat during storage and preparation. A review. J. Am. Diet. Assoc. 1976;69:253–257. doi: 10.1016/S0002-8223(21)06708-0
- Meyer B.H., Mysinger M.A., Wodarski L.A. Pantothenic acid and vitamin B6 in beef. J. Am. Diet Assoc. 1969;54:122–125. doi: 10.1016/S0002-8223(21)12596-9
- Roe M., Church S., Pinchen H., Finglas P. Nutrient Analysis of Fish and Fish Products. Institute of Food Research; Norwich, UK: 2013. pp. 14–69. Analytical Report.
- Bodwell C., Anderson B. Nutritional Composition and Value of Meat and Meat Products. Academic Press; Cambridge, MA, USA: 1986. pp. 321–369.
- Roe M., Church S., Pinchen H., Finglas P. Nutrient Analysis of Fruit and Vegetables. Institute of Food Research; Norwich, UK: 2013. pp. 17–76. Analytical Report.
- Cheng T.S., Eitenmiller R.R. Effects of Processing and Storage on the Pantothenic-Acid Content of Spinach and Broccoli. J. Food Process. Preserv. 1988;12:115–123. doi: 10.1111/j.1745-4549.1988.tb00071.x
- Hoppner K., Lampi B. Pantothenic-Acid and Biotin Retention in Cooked Legumes. J. Food Sci. 1993;58:1084–1085. doi: 10.1111/j.1365-2621.1993.tb06119.x
- Khalil A.H., Mansour E.H. The Effect of Cooking, Autoclaving and Germination on the Nutritional Quality of Faba Beans. Food Chem. 1995;54:177–182. doi: 10.1016/0308-8146(95)00024-D
- Roe M., Church S., Pinchen H., Finglas P. Nutrient Analysis of Eggs. Institute of Food Research; Norwich, UK: 2013. pp. 1–44. Analytical Report.
- Rolls B.A., Porter J.W.G. Some effects of processing and storage on the nutritive value of milk and milk products. Proc. Nutr Soc. 1973;32:9–15. doi: 10.1079/PNS19730003
- Nurit E., Lyan B., Pujos-Guillot E., Branlard G., Piquet A. Change in B and E vitamin and lutein, β-sitosterol contents in industrial milling fractions and during toasted bread production. J. Cereal Sci. 2016;69:290–296. doi: 10.1016/j.jcs.2016.04.005
- Dunn K.R., Goddard V.R. Effect of heat upon the nutritive values of peanuts; riboflavin and pantothenic acid content. Food Res. 1948;13:512–517. doi: 10.1111/j.1365-2621.1948.tb16652.x
- Greenwood D.A., Kraybill H.R., Feaster J.F., Jackson J.M. Vitamin Retention in Processed Meat. Ind. Eng. Chem. 1944;36:922–927. doi: 10.1021/ie50418a012
- Muhamad N., Yusoff M.M., Gimbun J. Thermal degradation kinetics of nicotinic acid, pantothenic acid and catechin derived from Averrhoa bilimbi fruits. RSC Adv. 2015;5:74132–74137. doi: 10.1039/C5RA11950B
- Woodside J. Nutritional aspects of irradiated food. Stewart Postharvest Rev. 2015;11:1–6. doi: 10.2212/spr.2015.3.2
- Kilcast D. Effect of Irradiation on Vitamins. Food Chem. 1994;49:157–164. doi: 10.1016/0308-8146(94)90152-X
- Suri D.J., Tanumihardjo S.A. Effects of Different Processing Methods on the Micronutrient and Phytochemical Contents of Maize: From A to Z. Compr. Rev. Food Sci. Food Saf. 2016;15:912–926. doi: 10.1111/1541-4337.12216
- Escalante-Aburto A., Mariscal-Moreno R.M., Santiago-Ramos D., Ponce-García N. An Update of Different Nixtamalization Technologies, and Its Effects on Chemical Composition and Nutritional Value of Corn Tortillas. Food Rev. Int. 2020;36:456–498. doi: 10.1080/87559129.2019.1649693
- Gonzalez-Lopez J., Aliaga L., Gonzalez-Martinez A., Martinez-Toledo M.V. Industrial Biotechnology of Vitamins, Biopigments, and Antioxidants. John Wiley & Sons, Inc.; Hoboken, NJ, USA: 2016. Pantothenic Acid; pp. 67–101.
- Kelly GS. Pantothenic acid. Monograph. Altern Med Rev. 2011 Sep;16(3):263-74.
- Pearson A., West R., Luecke R. The vitamin and amino acid content of drip obtained upon defrosting frozen pork. Food Res. 1959;24:515–519. doi: 10.1111/j.1365-2621.1959.tb17302.x
- Pearson A.M., Burnside J.E., Edwards H.M., Glasscock R.S., Cunha T.J., Novak A.F. Vitamin losses in drip obtained upon defrosting frozen meat. Food Res. 1951;16:85–87. doi: 10.1111/j.1365-2621.1951.tb17354.x
- Woollard D.C., Indyk H.E., Christiansen S.K. The analysis of pantothenic acid in milk and infant formulas by HPLC. Food Chem. 2000;69:201–208. doi: 10.1016/S0308-8146(99)00255-1
- Liberato S.C., Pinheiro-Sant’Ana H.M. Fortification of industrialized foods with vitamins. Rev. Nutr. 2006;19:215–231. doi: 10.1590/S1415-52732006000200009
- EU Commision E. Commission Directive 2006/125/EC of 5 December 2006 on Processed Cereal-Based Foods and Baby Foods for Infants and Young Children. https://eur-lex.europa.eu/eli/dir/2006/125/oj
- Müller M.A., Medlock J., Prágai Z., Warnke I., Litta G., Kleefeldt A., Kaiser K., De Potzolli B. Ullmann’s Encyclopedia of Industrial Chemistry. John Wiley & Sons, Inc.; Hoboken, NJ, USA: 2019. Vitamins, 9. Vitamin B5; pp. 1–16.
- EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA) Scientific Opinion on Dietary Reference Values for pantothenic acid. EFSA J. 2014;12:3581. doi: 10.2903/j.efsa.2014.3581
- Lu B., Ren Y., Huang B., Liao W., Cai Z., Tie X. Simultaneous determination of four water-soluble vitamins in fortified infant foods by ultra-performance liquid chromatography coupled with triple quadrupole mass spectrometry. J. Chromatogr. Sci. 2008;46:225–232. doi: 10.1093/chromsci/46.3.225
- U.S. Department of Agriculture, Agricultural Research Service. FoodData Central. https://fdc.nal.usda.gov
- Horváth Z, Vécsei L. Current medical aspects of pantethine. Ideggyogy Sz. 2009 Jul 30;62(7-8):220-9.
- Hendler SS, Rorvik DR, eds. PDR for Nutritional Supplements. 2nd ed. Montvale: Thomson Reuters; 2008.
- Pantothenate kinase-associated neurodegeneration. https://medlineplus.gov/genetics/condition/pantothenate-kinase-associated-neurodegeneration
- Hartig MB, Hörtnagel K, Garavaglia B, Zorzi G, Kmiec T, Klopstock T, Rostasy K, Svetel M, Kostic VS, Schuelke M, Botz E, Weindl A, Novakovic I, Nardocci N, Prokisch H, Meitinger T. Genotypic and phenotypic spectrum of PANK2 mutations in patients with neurodegeneration with brain iron accumulation. Ann Neurol. 2006 Feb;59(2):248-56. doi: 10.1002/ana.20771
- Schneider SA, Hardy J, Bhatia KP. Syndromes of neurodegeneration with brain iron accumulation (NBIA): an update on clinical presentations, histological and genetic underpinnings, and treatment considerations. Mov Disord. 2012 Jan;27(1):42-53. doi: 10.1002/mds.23971
- Zhou B, Westaway SK, Levinson B, Johnson MA, Gitschier J, Hayflick SJ. A novel pantothenate kinase gene (PANK2) is defective in Hallervorden-Spatz syndrome. Nat Genet. 2001 Aug;28(4):345-9. doi: 10.1038/ng572
- Hörtnagel K, Prokisch H, Meitinger T. An isoform of hPANK2, deficient in pantothenate kinase-associated neurodegeneration, localizes to mitochondria. Hum Mol Genet. 2003 Feb 1;12(3):321-7. doi: 10.1093/hmg/ddg026
- Kurian MA, Hayflick SJ. Pantothenate kinase-associated neurodegeneration (PKAN) and PLA2G6-associated neurodegeneration (PLAN): review of two major neurodegeneration with brain iron accumulation (NBIA) phenotypes. Int Rev Neurobiol. 2013;110:49-71. doi: 10.1016/B978-0-12-410502-7.00003-X
- Glusman M. The syndrome of burning feet (nutritional melalgia) as a manifestation of nutritional deficiency. Am J Med 1947;3:211-23. doi: 10.1016/0002-9343(47)90151-4
- Hodges RE, Bean WB, Ohlson MA, et al. Human pantothenic acid deficiency produced by omega-methyl pantothenic acid. J Clin Invest 1959;38:1421-5. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC442097/pdf/jcinvest00451-0153.pdf
- Johnson M.A., Kuo Y.M., Westaway S.K., Parker S.M., Ching K.H., Gitschier J., Hayflick S.J. Mitochondrial localization of human PANK2 and hypotheses of secondary iron accumulation in pantothenate kinase-associated neurodegeneration. Ann. N. Y. Acad. Sci. 2004;1012:282–298. doi: 10.1196/annals.1306.023
- Pantothenate Kinase-Associated Neurodegeneration. https://rarediseases.org/rare-diseases/pantothenate-kinase-associated-neurodegeneration
- Pantothenate kinase-associated neurodegeneration. https://rarediseases.info.nih.gov/diseases/6564/pantothenate-kinase-associated-neurodegeneration
- Sanvictores T, Chauhan S. Vitamin B5 (Pantothenic Acid) [Updated 2022 Oct 12]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK563233
- Gregory A, Polster BJ, Hayflick SJ. Clinical and genetic delineation of neurodegeneration with brain iron accumulation. J Med Genet. 2009 Feb;46(2):73-80. doi: 10.1136/jmg.2008.061929
- Gregory A, Hayflick SJ. Pantothenate Kinase-Associated Neurodegeneration. Orphanet Encyclopedia. 2004.
- Hayflick SJ, Westaway SK, Levinson B, Zhou B, Johnson MA, Ching KH, Gitschier J. Genetic, clinical, and radiographic delineation of Hallervorden-Spatz syndrome. N Engl J Med. 2003 Jan 2;348(1):33-40. doi: 10.1056/NEJMoa020817
- Hayflick SJ, Hartman M, Coryell J, Gitschier J, Rowley H. Brain MRI in neurodegeneration with brain iron accumulation with and without PANK2 mutations. AJNR Am J Neuroradiol. 2006 Jun-Jul;27(6):1230-3. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2099458
- PANK2 gene. https://medlineplus.gov/genetics/gene/pank2
- Hayflick SJ. Defective pantothenate metabolism and neurodegeneration. Biochem Soc Trans. 2014 Aug;42(4):1063-8. doi: 10.1042/BST20140098
- Gregory A, Hayflick SJ. Pantothenate Kinase-Associated Neurodegeneration. In: Adam MP, Ardinger HH, Pagon RA, et al., eds. GeneReviews. Seattle, WA: University of Washington, Seattle; 2017.
- Pantothenate kinase-associated neurodegeneration. https://medlineplus.gov/images/PX000366_PRESENTATION.jpeg
- Olivieri NF, Sabouhanian A, Gallie BL. Single-center retrospective study of the effectiveness and toxicity of the oral iron chelating drugs deferiprone and deferasirox. PLoS One. 2019 Feb 27;14(2):e0211942. doi: 10.1371/journal.pone.0211942. Erratum in: PLoS One. 2019 Mar 13;14(3):e0214005
- Munshi MI, Yao SJ, Ben Mamoun C. Redesigning therapies for pantothenate kinase-associated neurodegeneration. J Biol Chem. 2022 Mar;298(3):101577. doi: 10.1016/j.jbc.2022.101577
- Hodges R.E., Ohlson M.A., Bean W.B. Pantothenic acid deficiency in man. J. Clin. Investig. 1958;37:1642–1657. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1062846/pdf/jcinvest00347-0188.pdf
- Fry P.C., Fox H.M., Tao H.G. Metabolic response to a pantothenic acid deficient diet in humans. J. Nutr. Sci. Vitam. 1976;22:339–346. doi: 10.3177/jnsv.22.339
- Xu J., Patassini S., Begley P., Church S., Waldvogel H.J., Faull R.L.M., Unwin R.D., Cooper G.J.S. Cerebral deficiency of vitamin B5 (d-pantothenic acid; pantothenate) as a potentially-reversible cause of neurodegeneration and dementia in sporadic Alzheimer’s disease. Biochem. Biophys. Res. Commun. 2020;527:676–681. doi: 10.1016/j.bbrc.2020.05.015
- Nelson M.M., Evans H.M. Pantothenic acid deficiency and reproduction in the rat. J. Nutr. 1946;31:497–507. doi: 10.1093/jn/31.4.497
- Chen M.-C., Song Y., Song W.O. Fetal growth retardation and death in pantothenic acid-deficient rats is due to imparired placental function. J. Nutr. Biochem. 1996;7:451–456. doi: 10.1016/0955-2863(96)00078-2
- Drell W., Dunn M.S. Production of pantothenic acid deficiency syndrome in mice with-methylpantothenic acid. Arch. Biochem. Biophys. 1951;33:110–119. doi: 10.1016/0003-9861(51)90085-9
- Pudelkewicz C., Roderuck C. Pantothenic acid deficiency in the young guinea pig. J. Nutr. 1960;70:348–352. doi: 10.1093/jn/70.3.348
- Olson R.E., Kaplan N.O. The effect of pantothenic acid deficiency upon the coenzyme A content and pyruvate utilization of rat and duck tissues. J. Biol. Chem. 1948;175:515–529. doi: 10.1016/S0021-9258(18)57172-6
- Hatano M. Pantothenic acid deficiency in rats. J. Vitam. 1962;8:143–159. doi: 10.5925/jnsv1954.8.143
- BERG BN. Duodenitis and duodenal ulcers produced in rats by pantothenic acid deficiency. Br J Exp Pathol. 1959 Aug;40(4):371-4.
- Groody T.C., Groody M.E. Feather Depigmentation and Pantothenic Acid Deficiency in Chicks. Science. 1942;95:655–656. doi: 10.1126/science.95.2478.655
- Gries C.L., Scott M.L. The pathology of thiamin, riboflavin, pantothenic acid and niacin deficiencies in the chick. J. Nutr. 1972;102:1269–1285. doi: 10.1093/jn/102.10.1269
- Wintrobe M.M., Follis R.H., Jr., Alcayaga R., Paulson M., Humphreys S. Pantothenic acid deficiency in swine with particular reference to the effects on growth and on the alimentary tract. Bul. Johns Hopkins Hosp. 1943;73:313–341.
- Follis R.H., Wintrobe M.M. A Comparison of the Effects of Pyridoxine and Pantothenic Acid Deficiencies on the Nervous Tissues of Swine. J. Exp. Med. 1945;81:539–552. doi: 10.1084/jem.81.6.539
- Nelson R.A. Intestinal transport, coenzyme A, and colitis in pantothenic acid deficiency. Am. J. Clin. Nutr. 1968;21:495–501. doi: 10.1093/ajcn/21.5.495
- Schaefer A.E., McKibbin J.M., Elvehjem C.A. Pantothenic acid deficiency studies in the dog. J. Biol. Chem. 1942;143:321–330. doi: 10.1016/S0021-9258(18)72619-7
- Silber R.H. Studies of Pantothenic Acid Deficiency in Dogs: Three Figures. J. Nutr. 1944;27:425–433. doi: 10.1093/jn/27.5.25
- Flodin N. Pharmacology of micronutrients. New York: Alan R. Liss, Inc.; 1988.
- Debourdeau PM, Djezzar S, Estival JL, Zammit CM, Richard RC, Castot AC. Life-threatening eosinophilic pleuropericardial effusion related to vitamins B5 and H. Ann Pharmacother. 2001 Apr;35(4):424-6. doi: 10.1345/aph.10213
- Debourdeau P.M., Djezzar S., Estival J.L., Zammit C.M., Richard R.C., Castot A.C. Life-threatening eosinophilic pleuropericardial effusion related to vitamins B5 and H. Ann. Pharmacother. 2001;35:424–426. doi: 10.1345/aph.10213
- Food and Nutrition Board, Institute of Medicine. Pantothenic acid. Dietary Reference Intakes: Thiamin, Riboflavin, Niacin, Vitamin B6, Vitamin B12, Pantothenic Acid, Biotin, and Choline. Washington, D.C.: National Academy Press; 1998:357-373.
- Herbst, R.A., Uter, W., Pirker, C., Geier, J. and Frosch, P.J. (2004), Allergic and non-allergic periorbital dermatitis: patch test results of the Information Network of the Departments of Dermatology during a 5-year period. Contact Dermatitis, 51: 13-19. https://doi.org/10.1111/j.0105-1873.2004.00334.x
- M. Schmuth, M.A. Wimmer, S. Hofer, A. Sztankay, G. Weinlich, D.M. Linder, P.M. Elias, P.O. Fritsch, E. Fritsch, Topical corticosteroid therapy for acute radiation dermatitis: a prospective, randomized, double‐blind study, British Journal of Dermatology, Volume 146, Issue 6, 1 June 2002, Pages 983–991, https://doi.org/10.1046/j.1365-2133.2002.04751.x
- Lee JH, Ahn SY, Lee HA, Won KS, Chang HW, Oh JS, Kim HW. Dietary intake of pantothenic acid is associated with cerebral amyloid burden in patients with cognitive impairment. Food Nutr Res. 2018 Dec 10;62. doi: 10.29219/fnr.v62.1415
- Chirapu SR, Rotter CJ, Miller EL, Varma MV, Dow RL, Finn MG. High specificity in response of the sodium-dependent multivitamin transporter to derivatives of pantothenic acid. Curr Top Med Chem. 2013;13(7):837-42. doi: 10.2174/1568026611313070006
- Said HM, Ortiz A, McCloud E, Dyer D, Moyer MP, Rubin S. Biotin uptake by human colonic epithelial NCM460 cells: a carrier-mediated process shared with pantothenic acid. Am J Physiol. 1998 Nov;275(5):C1365-71. doi: 10.1152/ajpcell.1998.275.5.C1365