What is white tea
White tea is tea made from new growth buds and young leaves of the plant Camellia sinensis. White tea leaves are steamed and dried almost immediately after harvesting (sometimes before even leaving the fields) or fried to inactivate oxidation and then dried. Currently there is no generally accepted definition of white tea and very little international agreement; some sources use the term to refer to tea that is merely dried with no additional processing 1, some to tea made from the buds and immature tea leaves picked shortly before the buds have fully opened and allowed to wither and dry in natural sun, while others include tea buds and very young leaves which have been steamed or fired before drying. Most definitions agree, however, that white tea is not rolled or oxidized, resulting in a flavor characterized as “lighter” than green or traditional black teas.
White tea
- The Chinese position: white tea is defined by the sub-species it is manufactured from (Camellia sinensis var. khenghe baihao) and Camellia sinensis var. fudin bai hao) found only in Fujian province and with minimal processing following traditional guidelines. White tea is a seasonal crop (spring) with special sensory and health benefits.
- Other producing countries define white tea by plucking standard i.e. only the bud or first leaves that are plucked and dried with minimal processing. In this way the delicate white leaf hairs are left intact lending the appearance of ‘white tea’. It has to be noted that, if this definition would be generally accepted, it would exclude traditional Chinese white tea varieties such as Pai Mu Tan (White Peony).
So there are a few issues: firstly the appearance (liquor of white tea is very pale yellow in color, and mild tasting in cup) and secondly the manufacture (minimal processing, just drying, no “fermentation”).
There are health claims underway, such as:
- (a) white tea is lower in caffeine than green tea,
- (b) white tea is much higher in antioxidants, esp. catechins than green tea and
- (c) white tea has a higher anti-mutagenicity action compared to green tea.
However, from compositional data available it can be stated that the first two claims are completely nonsense 1 (see Table 1 below). Currently there is a great marketing and public interest for white tea. As there is a high demand but low supply it has sometimes been suspected that more white tea is now sold than is grown!
In spite of its name, brewed white tea is pale yellow. Its name derives from the fine silvery-white hairs on the unopened buds of the tea plant, which give the plant a whitish appearance. The unopened buds are used for some types of white tea. All kinds of tea originate from Camellia sinensis, white tea is harvested primarily in China, mostly in the Fujian province, but more recently produced in Eastern Nepal, Taiwan, Northern Thailand, Galle (Southern Sri Lanka) and India.
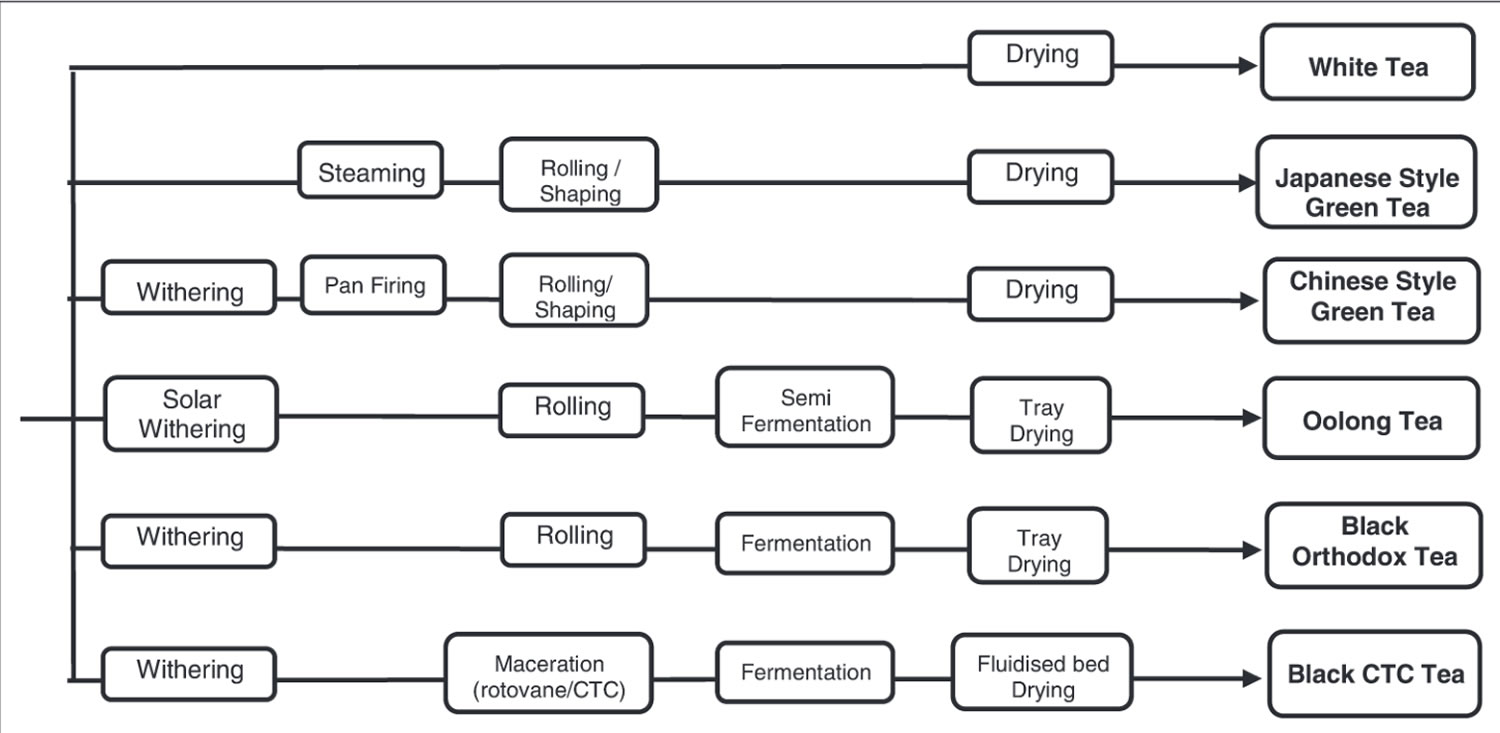
Figure 1. Tea manufacture – major steps and corresponding types of tea
[Source 1]Table 1. White tea and green tea caffeine and polyphenols content
| White Tea | Average | Green Tea | Average | |
| Caffeine | 3.35-5.74 g/100 gram | 4.85 g/100 gram | 1.67-3.90 g/100 gram | 2.90 g/100 gram |
| Total Polyphenols | 16.23-25.95 g/100 gram | 21.54 g/100 gram | 13.7-24.7 g/100 gram | 19.18 g/100 gram |
| Total Catechins | 7.94-16.56 g/100 gram | 13.22 g/100 gram | 9.89-17 g/100 gram | 12.95 g/100 gram |
| Epigallocatechin-3-gallate (EGCG) | 5.23-9.49 | 8 | 4.40-9.6 | 6.75 |
| Epigallocatechin | 0.24-2.64 | 1.11 | 1.94-4.07 | 2.84 |
| Flavonol glycosides * | 0.06-1.44 | 0.61 (1.25) | 0.64-2.02 | 1.1 (2.27) |
| * Data are calculated as aglycones, in parentheses as glycosides. | ||||
Health benefits of white tea
Tea is a natural source of proanthocyanidins which does occur in green teas in relevant amounts. Green tea is relatively rich in proanthocyanidins and lower in bisflavanols, while black tea has higher amounts of bisflavanols. This is in tune with the observation of Hashimoto et al. 2 that especially the galloylated proanthocyanidins are degraded during the so-called fermentation. There are at least 16 proanthocyanidins present in tea 3. Not much information on the contribution of proanthocyanidins to the taste of tea beverages or to the health effects is currently available.
Tea contains both kaempferol and quercetin as well as flavanols or catechins up to 30% dry weight and other acids such as gallic acid, caffeic acid and coumaric acid 4. Flavonols (quercetin, kaempferol, and myricetin) are present in form of their O-glycosides. At least 14 different glycosides have been detected in tea 5. There are mono-, di- and triglycosides present in tea, among those often the quercetin 3-rhamno-glucoside in the highest concentration. Flavonol glycosides are interesting in terms of health benefits. According to the literature there are protective effects against coronary heart disease. Scharbert et al. 5 stated that the flavonol glycosides are responsible for the astringency in black tea and not theaflavins or catechins. The flavonol glycosides content is not very much affected by the enzyme transformation (fermentation).
Flavonoids are thought to prevent the formation of reactive oxygen species by inhibiting enzymes or chelating trace metals which can mediate free radical production as well as being free radical scavengers and upregulating genetic antioxidant defences 6. Catechins are powerful bioflavonoids and green tea has been shown in vivo and in vitro to have anti-inflammatory and antioxidant activity 7.
Phenolics in tea
All teas contain high quantities of several polyphenolic components, particularly epicatechin, epicatechin gallate, epigallocatechin, and the most abundant (constituting 65% of the total catechin content) and perhaps the most bioactive component, epigallocatechin-3-gallate (EGCG) 8. Other, such as epiafzelechin and its gallate as well as acetylated catechins have also been identified 9. Tea leaves that have been processed the least contain the most catechins. Tea’s polyphenols include flavanols, flavandiols, flavonoids, and phenolic acids; these compounds may account for up to 30% of the dry weight of the tea leaves according to the literature.
Figure 2. Chemical structure of the four major catechins present in tea
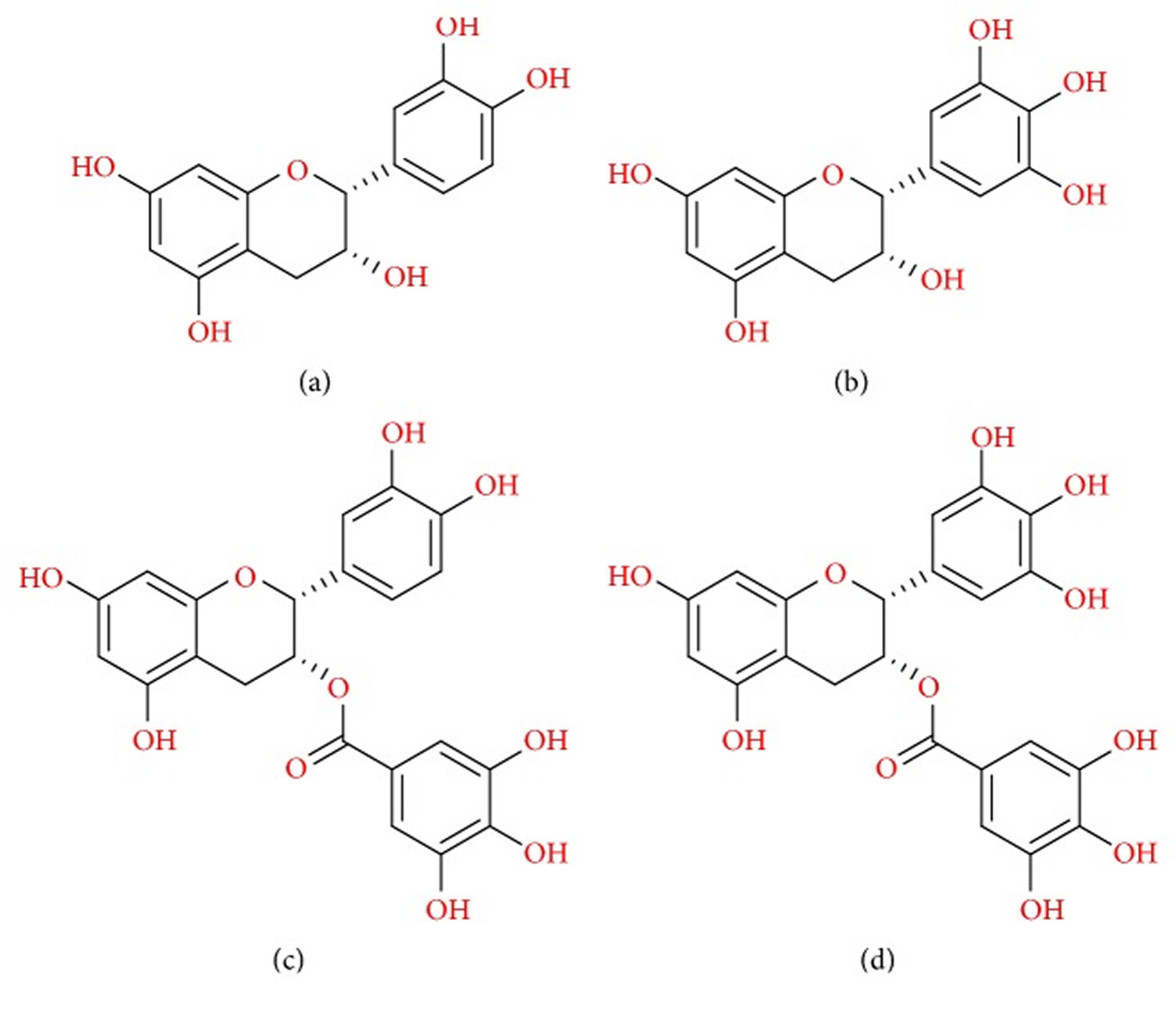
Note: (a) (−)-Epicatechin (EC), (b) (−)-epigallocatechin (EGC), (c) (−)-epicatechin gallate (ECG), and (d) (−)-epigallocatechin gallate (EGCG).
[Source 10]Epigallo-catechin-3-gallate (EGCG) is a complex molecule formed by a flavanol core (flavan-3-ols) structure with a gallocatechol group and a gallate ester 11. These two gallocatechol rings confer the potent antioxidant and chelating properties to epigallo-catechin-3-gallate (EGCG) 12. Each of the gallocatechol rings is capable of directly capturing free radicals from the environment with high efficiency 13. Previous studies have shown that epigallo-catechin-3-gallate (EGCG) possesses a stronger antioxidant capacity compared with the other green tea catechins and it is also demonstrated that epigallo-catechin-3-gallate (EGCG) is more efficient in radical scavenging than vitamins E and C 14.
Therapeutic Potential of Epigallo-catechin-3-gallate (EGCG)
For many years, the consumption of green tea has been associated with numerous health benefits 15. These properties can be directly linked with the polyphenol content of tea, more specifically with epigallo-catechin-3-gallate (EGCG). For this reason, the study of epigallo-catechin-3-gallate (EGCG) is of utmost importance because this compound seems to prevent and also be useful in the treatment of numerous diseases like cancer and cardiovascular and neurodegenerative diseases 16. Epigallo-catechin-3-gallate (EGCG) is a powerful antioxidant, anti-inflammatory, antibacterial, and antiviral agent and is capable of modulating some pathways, changing the metabolism of lipids 17.
Cancer Chemoprevention
Cancer is the end of several steps of cellular growth lesions, namely, hyperplasia, metaplasia, dysplasia, and neoplasia 18. Each of the presented conditions is a progression in the cancer formation, culminating in the malignant neoplasia known as cancer 19. Nowadays, most modern therapies currently available for treating cancer are very expensive and toxic and have low effectiveness in treating the disease 19. Therefore, it is urgent to investigate natural compounds like epigallocatechin-3-gallate (EGCG) derived from green tea for the prevention and treatment of cancer and other diseases 18. According to previous studies, epigallocatechin-3-gallate (EGCG) is a promising molecule in the prevention and treatment of cancer 20. Some anticancer properties of EGCG are attributed to its free radical scavenging properties, avoiding the damage of the cell structures induced by the free radicals 21. Besides being antioxidant, EGCG has the ability to bind and modulate the activity of several signaling molecules related to mitosis, survival, and cellular death, moderating the cellular responses present in cancer 22. Previous works demonstrated that EGCG is able to inhibit all of the processes involved in carcinogenesis: initiation, promotion, and progression 23. EGCG has the ability to bind to some proteins associated in molecular pathways that are misregulated in cancerous cells. Indeed, EGCG induces the suppression of two important transcription factors, tumor suppressor p53 and nuclear factor kappa-light-chain enhancer of activated B-cells (NF-kB), leading to regression of the tumors 24. To assist the growth of the tumor, new capillaries are needed to satisfy the oxygen and nutrient requirements of the cells 25. The growth process of new blood vessels is called angiogenesis 25. To promote formation of new capillaries, the tumor secretes signaling molecules to the surrounding tissues, especially vascular endothelial growth factor (VEGF). VEGF is directly influenced by the activity of hypoxia-inducible factor 1α (HIF-1α) and NF-kB factors, which are modulated by the presence of EGCG 25. For these reasons, EGCG is able to diminish tumor angiogenesis and stall growth 26. In addition, there is strong evidence that EGCG is capable of diminishing migration and metastasis formation of tumors 27. Previous studies report that EGCG promotes a reduction in the migration and metastasis formation of tumor cells with tumor size reduction, accomplishing a more reliable and efficient chemotherapy 28. Although the single use of EGCG in chemotherapy is unlikely due to its inefficacy in completely eliminating the disease, it would be very interesting to use EGCG as an adjuvant of the cytostatic drugs 29. This synergism that has been reported in numerous in vitro, in vivo, and preclinical studies may be useful to reduce the amount of the necessary cytostatic drugs, which will reduce the side effects 30. In addition, EGCG’s antioxidant and anti-inflammatory properties are also useful to protect against chemotherapy side effects. Finally, the health benefits of EGCG would be advantageous in enhancing the overall condition of the patients 24.
Cardiovascular Benefits
Cardiovascular diseases have a high incidence, mainly in the developed world due to a sedentary lifestyle, poor nutrition, and ambient factors 31. A diet rich in cholesterol, fat, and sugar can lead to coronary diseases like arteriosclerosis and ischemia 31. Recent studies showed that EGCG can enhance the capillary circulation dilating the capillaries, diminishing inflammation, and interfering with the lipid absorption and digestion 32. On the other hand, EGCG interferes directly with the lipid emulsion process in the lipid digestion 33. This is achieved by direct interference in the micelle formation and by inhibiting the phospholipase A2, with this enzyme being of high importance in the lipid digestion 33. The junction of the two processes can limit the absorption of lipids and consequently lower the amount of plasmatic lipids and cholesterol 33. In addition, EGCG can lower cholesterol even more, stimulating its excretion through the bile. Moreover, EGCG will further improve the lipid profile by enhancing the lipid metabolism 34. This catechin can also modulate the process of platelets formation, from macrophage recruiting to macrophage uptake of cholesterol 34. This effect is internally modulated in the macrophage and externally helped by the anti-inflammatory response caused by EGCG 35. Previous studies demonstrated that the administration of EGCG is capable of preventing the growth and also reducing the size of existing platelets. The action mechanism responsible for the anti-inflammatory property of EGCG is the direct inhibition of the phospholipase A2 35.
Neurodegenerative Diseases
The causes of neurodegenerative diseases like Parkinson’s disease and Alzheimer’s disease are still unknown, with various theories being proposed. Both diseases present clinical features, like the oxidative damage of neurons and accumulation of iron in specific brain areas 36. Another relevant aspect is the accumulation of misfolded proteins in deposits, such as the β-amyloid peptide in Alzheimer’s disease that interferes with the survival of the neurons, leading to premature apoptosis 36. Special interest has been assigned to the therapeutic role of antioxidants in such neurodegenerative diseases 37. The neuroprotective properties of the EGCG agent are related to its antioxidant, anti-inflammatory, and iron chelating properties 37. In addition, the blood-brain barrier is permeable to EGCG 38. The mechanism behind the passage of this hydrophilic compound through the blood-brain barrier remains unknown 38. In the literature, it is described that EGCG is more efficient in radical scavenging than vitamins C and E, with its iron chelating ability being useful to significantly improve the symptoms of these neurodegenerative diseases 39. According to what was mentioned above, EGCG is also a cellular modulator that interacts with various pathways. In neuronal cells, this catechin promotes cell survival responses and the inhibition of cell death signals, leading to an enhancement of neuronal health 40. Modifications in cell signaling also promote the nonamyloid α-secretase pathway, diminishing the production of Aβ-amyloid peptides 39.
Several research studies confirm that EGCG has neuroprotective properties in humans, promoting an enhancement of the degree of cognition after oral administration. These studies also confirm that EGCG induces an overall increase in the cerebral activity and calmness 41.
Infectious Diseases
Nowadays, the main strategy to fight viruses is immunization. Unfortunately, several viral infections lack one efficient vaccine, with the HIV infection being the most important. Nance et al. have shown strong HIV inhibition promoted by EGCG in cell cultures in a dose dependent manner 42. Moreover, Li et al. have also proven that EGCG inhibit reverse transcriptase and act synergistically with another reverse transcriptase inhibitor, namely, azidothymidine 43. Some studies also described that EGCG is capable of binding to CD4 cells, preventing the virus from anchoring and entering the host 42.
EGCG is also useful in the inhibition of other viruses, such as enterovirus 71, hepatitis C, adenovirus, herpes simplex virus, and influenza virus 44. One of the molecular targets that seem to be deregulated by the viral infection is the NF-kB and the MAP-kinases pathway 45. As a consequence, EGCG may induce an essential immune response, which helps to fight the viral infection. Concerning antibacterial and antifungal activities, EGCG seems to be less effective in combating infectious diseases caused by bacteria and fungi 46. The most relevant studies in the literature show that there may be some synergistic effects on EGCG association with antibiotics against multidrug-resistant strains, such as Staphylococcus aureus and Stenotrophomonas maltophilia 47. The antifungal activity of EGCG was also reported against human-pathogenic yeasts, such as Candida albicans. However, the mechanisms of action are still unclear 48.
Chronic Inflammatory Disorder
Inflammation is a body response to foreign structures to the human body and damage in the tissues 49. However, in chronic inflammatory disorders, this inflammatory response is continuously active leading to the destruction of healthy tissues causing all the above-mentioned symptoms. These conditions can be incurable and cause major discomfort to the patients 36. Rheumatoid arthritis is one chronic inflammatory disorder characterized by cellular infiltration and proliferation of the synovium, leading to the progressive destruction of the joints through the interaction between infiltrating cells and mediators 50. These injuries lead to chronic pain affecting the life quality of the patients 50. In this disease, the cartilage cells (i.e., chondrocytes) enter in apoptosis in response to oxidative stress and some inflammatory cytokines, interleukin (IL-1β) and tumor necrosis factor-α (TNF-α) 50. The same cytokines also lead to the increase of bone reabsorption and the differentiation of osteoclasts 50. In addition, IL-1β is capable of increasing the amounts of reactive oxygen species via overexpression of inducible nitric oxide synthase and increases the inflammation by overexpression of cyclooxygenase (COX-2) 51. The presence of IL-1β can also activate the expression of matrix metalloproteinases (MMPs) responsible for matrix degradation 51. TNF-α also plays an important role in bone turnover. In arthritis, there is overexpression of TNF-α, which is responsible for the differentiation and activity of osteoclasts. The long-term activation of these cells leads to bone erosion and fragility 52. The current treatment for arthritis is by the administration of methotrexate combined with analgesics and nonsteroidal anti-inflammatory drugs which can be proficient in most cases, but ineffective in some patients 53. Moreover, recent studies have shown that this treatment tends to lose efficacy over time 53. For this reason, new therapies are needed and EGCG may be a promising compound. In fact, EGCG has a high antioxidant activity and also capacity to decrease the inflammation response in the body 54. In cartilage cell cultures, EGCG showed marked inhibition of IL-1β inducible nitric oxide synthase COX-2 expression and activity 55. The expressions of both enzymes are mediated by NF-kB, which is also suppressed in the presence of EGCG 55.
Obesity
Obesity is a medical condition characterized by excess accumulation of fat in the body in an extension that may have negative effects on the overall health condition and may lead to the development of diseases, such as diabetes and arteriosclerosis 56. The main treatment of obesity is lifestyle reeducation, including diet modification 57. However, in some cases, drugs and supplements are needed to help in the process of losing weight 57. As previously stated, EGCG interferes directly with the lipid digestion by the inhibition of the phospholipase A2 and interfering with the lipid/cholesterol emulsion in the gut 58. The lipid blocking capacity of EGCG can be highly relevant in the loss of weight and weight managing protocols. In addition, EGCG is capable of enhancing the lipid metabolism, leading to more caloric burn and consequent fat loss. EGCG can also interfere with the digestion of starch by inhibition of α-amylase 59. Besides that, the ingestion of EGCG during a weight loss program is very useful because its administration is strongly linked with circulation improvement, free radical scavenging, and mood enhancement 60.
Diabetes
EGCG has been associated with the prevention and reversion of diabetes mellitus through a number of effects, such as improvement of insulin secretion, regulation of glucose uptake, inhibition of insulin resistance, and enhancement of glucose tolerance and its role in oxidative stress and inflammation 61. However, these beneficial effects in diabetes are not regulated by a single mechanism, but still EGCG appears to act through multiple signaling pathways. Green tea intake has been reported to exert beneficial intestinal effects increasing the blood EGCG levels which in turn seem to inhibit cellular glucose uptake, improving its tolerance in vivo 61. Several studies demonstrated that EGCG significantly enhances glucose tolerance in rodents with type 2 diabetes mellitus 62. Another study suggested that EGCG increases glucose-stimulated insulin secretion in db/db mice, through its potent antioxidant effect 63. At the same time, EGCG induces tyrosine phosphorylation of insulin receptors, thereby mimicking insulin in H4IIE rat hepatoma cells [130]. In H4IIE cells, EGCG downregulates genes involved in gluconeogenesis and the synthesis of fatty acids, triacylglycerol, and cholesterol and glucokinase mRNA expression was upregulated in the liver of db/db mice in a dose dependent manner 64. Moreover, Cai et al. showed that EGCG improves the insulin secretory function in rat pancreatic β-cell lines under conditions of glucotoxicity through mediation of Akt signaling pathway 65. EGCG also revealed effects on fatty acid-induced insulin resistance in skeletal muscle, through the activation of protein kinase C (PKC) or c-Jun N-terminal kinase (JNK) signaling pathways 66. Furthermore, EGCG can also enhance AMPK/ACC cascade that blocks insulin receptor substrate-1 (IRS-1) serine phosphorylation, which is essential for the glucose uptake in response to insulin stimulus 67.
Summary
White tea and tea in general contains various bioactive phytochemicals – Epigallo-catechin-3-gallate (EGCG) and Flavonoids – that could provide therapeutic effects.
However, more human clinial trials are needed in order to establish whether or not there is therapeutic effect of drinking tea (white, green, Oolong or black teas). Without such evidence, it will remain unclear whether these untested and unproven positive effects in test tube and animal studies are truly beneficial in humans. For now, there is insufficient evidence that tea oe white tea have any benefits in diabetes, metabolic syndrome, obesity or weight loss, cancer, infectious diseases, Alzheimer’s disease, Parkinson’s disease or cardiovascular diseases.
References- Characterisation of white tea–Comparison to green and black tea. J. Verbr. Lebensm. 2 (2007): 414 – 421. https://www.tu-braunschweig.de/Medien-DB/ilc/w_t.pdf
- Hashimoto, F., Nonaka, G. and Nishioka, I. (1992) Tannins and Related Compounds. CXIV. Structures of Novel Fermentation Products, Theogallinin, Theaflavonin and Desgalloyl Theaflavonin from Black Tea, and Changes of Tea Leaf Polyphenols during Fermentation. Chem Pharm Bull 40: 1383–1389.
- Engelhardt, U. H., Lakenbrink, C. and Pokorny, O. (2004) Proanthocyanidins, Bisflavanols,and Hydrolyzable Tannins in Green and Black Teas. In: Shahidi, F. and Weerasinghe, D. K. (eds) Nutraceutical Beverages. Chemistry, Nutrition, and Health Effects. ACS Symposium Series 871, pp. 254–264.
- Van Wyk BE, Wink M. Medicinal plants of the world. Pretoria, South Africa: Briza publications; 2004.
- Scharbert, S., Holzmann, N. and Hofmann, T. (2004) Identification of the astringent taste compounds in black tea infusions by combining instrumental analysis and human bioresponse. J Agric Food Chem 52: 3498–508.
- Pietta P-G. Flavonoids as antioxidants. J Natural Products. 2000;63:1035–1042. doi: 10.1021/np9904509. https://www.ncbi.nlm.nih.gov/pubmed/10924197
- Yang CS, Lee MJ, Chen L. Human salivary tea catechin levels and catechin esterase activities: implication in human cancer prevention studies. Cancer Epidem Biomar. 1999;8:83–89. http://cebp.aacrjournals.org/content/8/1/83.long
- Cardiovascular effects of green tea catechins: progress and promise. Islam MA. Recent Pat Cardiovasc Drug Discov. 2012 Aug; 7(2):88-99. https://www.ncbi.nlm.nih.gov/pubmed/22670802/
- Hashimoto, F., Nonaka, G., Nishioka, I. (1987): Tannins and Related Compounds. LVI. Isolation of Four New Acylated Flavan-3-ols from Oolong Tea. Chem Pharm Bull 35: 611–616.
- Granja A, Frias I, Neves AR, Pinheiro M, Reis S. Therapeutic Potential of Epigallocatechin Gallate Nanodelivery Systems. BioMed Research International. 2017;2017:5813793. doi:10.1155/2017/5813793. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5534279/
- Structural Properties of Green Tea Catechins. Botten D, Fugallo G, Fraternali F, Molteni C. J Phys Chem B. 2015 Oct 8; 119(40):12860-7.
- The relationship between the structure and biological actions of green tea catechins. Braicu C, Ladomery MR, Chedea VS, Irimie A, Berindan-Neagoe I. Food Chem. 2013 Dec 1; 141(3):3282-9. https://www.ncbi.nlm.nih.gov/pubmed/23871088/
- Scavenging effects of tea catechins and their derivatives on 1,1-diphenyl-2-picrylhydrazyl radical. Nanjo F, Goto K, Seto R, Suzuki M, Sakai M, Hara Y. Free Radic Biol Med. 1996; 21(6):895-902.
- Structure-activity relationship of the tocopherol-regeneration reaction by catechins. Mukai K, Mitani S, Ohara K, Nagaoka S. Free Radic Biol Med. 2005 May 1; 38(9):1243-56.
- Afzal M., Safer A. M., Menon M. Green tea polyphenols and their potential role in health and disease. Inflammopharmacology. 2015;23(4):151–161. doi: 10.1007/s10787-015-0236-1.
- Keske M. A., Ng H. L. H., Premilovac D., et al. Vascular and metabolic actions of the green tea polyphenol epigallocatechin gallate. Current Medicinal Chemistry. 2015;22(1):59–69. doi: 10.2174/0929867321666141012174553. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4909506/
- Yang C. S., Zhang J., Zhang L., Huang J., Wang Y. Mechanisms of body weight reduction and metabolic syndrome alleviation by tea. Molecular Nutrition and Food Research. 2016;60(1):160–174. doi: 10.1002/mnfr.201500428. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4991829/
- Singh B. N., Shankar S., Srivastava R. K. Green tea catechin, epigallocatechin-3-gallate (EGCG): mechanisms, perspectives and clinical applications. Biochemical Pharmacology. 2011;82(12):1807–1821. doi: 10.1016/j.bcp.2011.07.093. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4082721/
- Barr R. D., Ferrari A., Ries L., Whelan J., Bleyer W. A. Cancer in adolescents and young adults: A narrative review of the current status and a view of the future. JAMA Pediatrics. 2016;170(5):495–501. doi: 10.1001/jamapediatrics.2015.4689. https://www.ncbi.nlm.nih.gov/pubmed/26999630
- Baker K. M., Bauer A. C. Green Tea Catechin, EGCG, Suppresses PCB 102-Induced Proliferation in Estrogen-Sensitive Breast Cancer Cells. International Journal of Breast Cancer. 2015;2015 doi: 10.1155/2015/163591.163591 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4691479/
- Farhan M., Khan H. Y., Oves M., et al. Cancer therapy by catechins involves redox cycling of copper ions and generation of reactive oxygenspecies. Toxins. 2016;8(2) doi: 10.3390/toxins8020037. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4773790/
- Rahmani A. H., Al Shabrmi F. M., Allemailem K. S., Aly S. M., Khan M. A. Implications of green tea and its constituents in the prevention of cancer via the modulation of cell signalling pathway. BioMed Research International. 2015;2015 doi: 10.1155/2015/925640.925640 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4419223/
- Wang P., Aronson W. J., Huang M., et al. Green tea polyphenols and metabolites in prostatectomy tissue: implications for cancer prevention. Cancer Prevention Research (Philadelphia, Pa.) 2010;3(8):985–993. doi: 10.1158/1940-6207.capr-09-0210. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3163459/
- Yamauchi R., Sasaki K., Yoshida K. Identification of epigallocatechin-3-gallate in green tea polyphenols as a potent inducer of p53-dependent apoptosis in the human lung cancer cell line A549. Toxicology in Vitro. 2009;23(5):834–839. doi: 10.1016/j.tiv.2009.04.011.
- Shi J., Liu F., Zhang W., Liu X., Lin B., Tang X. Epigallocatechin-3-gallate inhibits nicotine-induced migration and invasion by the suppression of angiogenesis and epithelial-mesenchymal transition in non-small cell lung cancer cells. Oncology Reports. 2015;33(6):2972–2980. doi: 10.3892/or.2015.3889.
- Gu J.-W., Makey K. L., Tucker K. B., et al. EGCG, a major green tea catechin suppresses breast tumor angiogenesis and growth via inhibiting the activation of HIF-1α and NFκB, and VEGF expression. Vascular Cell. 2013;5(1, article 9) doi: 10.1186/2045-824x-5-9.
- Zapf M. A. C., Kothari A. N., Weber C. E., et al. Green tea component epigallocatechin-3-gallate decreases expression of osteopontin via a decrease in mRNA half-life in cell lines of metastatic hepatocellular carcinoma. Surgery (United States) 2015;158(4):1039–1048. doi: 10.1016/j.surg.2015.06.011. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5105906/
- Takahashi A., Watanabe T., Mondal A. Mechanism-based inhibition of cancer metastasis with (−)-epigallocatechin gallate. Biochemical and Biophysical Research Communications. 2014;443(1):1–6. doi: 10.1016/j.bbrc.2013.10.094.
- Wang X., Jiang P., Wang P., Yang C. S., Wang X., Feng Q. EGCG enhances Cisplatin sensitivity by regulating expression of the copper and cisplatin influx transporter CTR1 in ovary cancer. PLoS ONE. 2015;10(4) doi: 10.1371/journal.pone.0125402.e0125402 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4416002/
- Cai Y., Zhang J., Chen N. G., et al. Recent Advances in Anticancer Activities and Drug Delivery Systems of Tannins. Medicinal Research Reviews. 2016 doi: 10.1002/med.21422.
- Gokulakrisnan A., Jayachandran Dare B., Thirunavukkarasu C. Attenuation of the cardiac inflammatory changes and lipid anomalies by (−)-epigallocatechin-gallate in cigarette smoke-exposed rats. Molecular and Cellular Biochemistry. 2011;354(1-2):1–10. doi: 10.1007/s11010-011-0785-6.
- Jang Y. H., Lee Y. C., Park N. H., et al. Polyphenol (−)-epigallocatechin gallate protection from ischemia/reperfusion-induced renal injury in normotensive and hypertensive rats. Transplantation Proceedings. 2006;38(7):2190–2194. doi: 10.1016/j.transproceed.2006.06.101.
- Wang S., Noh S. K., Koo S. I. Green tea catechins inhibit pancreatic phospholipase A2 and intestinal absorption of lipids in ovariectomized rats. The Journal of Nutritional Biochemistry. 2006;17(7):492–498. doi: 10.1016/j.jnutbio.2006.03.004.
- Jin Y.-R., Im J.-H., Park E.-S., et al. Antiplatelet activity of epigallocatechin gallate is mediated by the inhibition of PLCγ2 phosphorylation, elevation of PGD2 production, and maintaining calcium-ATPase activity. Journal of Cardiovascular Pharmacology. 2008;51(1):45–54. doi: 10.1097/FJC.0b013e31815ab4b6.
- Koo S. I., Noh S. K. Green tea as inhibitor of the intestinal absorption of lipids: potential mechanism for its lipid-lowering effect. Journal of Nutritional Biochemistry. 2007;18(3):179–183. doi: 10.1016/j.jnutbio.2006.12.005 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1852441/
- Skovronsky D. M., Lee V. M.-Y., Trojanowski J. Q. Neurodegenerative diseases: new concepts of pathogenesis and their therapeutic implications. Annual Review of Pathology. 2006;1:151–170. doi: 10.1146/annurev.pathol.1.110304.100113
- Lee J. W., Lee Y. K., Ban J. O., et al. Green tea (−)-epigallocatechin-3-gallate inhibits β-amyloid-induced cognitive dysfunction through modification of secretase activity via inhibition of ERK and NF-κB pathways in mice. Journal of Nutrition. 2009;139(10):1987–1993. doi: 10.3945/jn.109.109785.
- Giunta B., Hou H., Zhu Y., et al. Fish oil enhances anti-amyloidogenic properties of green tea EGCG in Tg2576 mice. Neuroscience Letters. 2010;471(3):134–138. doi: 10.1016/j.neulet.2010.01.026.
- Weinreb O., Mandel S., Amit T., Youdim M. B. H. Neurological mechanisms of green tea polyphenols in Alzheimer’s and Parkinson’s diseases. Journal of Nutritional Biochemistry. 2004;15(9):506–516. doi: 10.1016/j.jnutbio.2004.05.002
- Ortiz-López L., Márquez-Valadez B., Gómez-Sánchez A., et al. Green tea compound epigallo-catechin-3-gallate (EGCG) increases neuronal survival in adult hippocampal neurogenesis in vivo and in vitro. Neuroscience. 2016;322:208–220. doi: 10.1016/j.neuroscience.2016.02.040
- Scholey A., Downey L. A., Ciorciari J., et al. Acute neurocognitive effects of epigallocatechin gallate (EGCG) Appetite. 2012;58(2):767–770. doi: 10.1016/j.appet.2011.11.016. https://www.ncbi.nlm.nih.gov/pubmed/22127270
- Nance C. L., Siwak E. B., Shearer W. T. Preclinical development of the green tea catechin, epigallocatechin gallate, as an HIV-1 therapy. Journal of Allergy and Clinical Immunology. 2009;123(2):459–465. doi: 10.1016/j.jaci.2008.12.024.
- Li S., Hattori T., Kodama E. N. Epigallocatechin gallate inhibits the HIV reverse transcription step. Antiviral Chemistry and Chemotherapy. 2011;21(6):239–243. doi: 10.3851/IMP1774
- Wang Y., Li J., Wang X., et al. (−)-Epigallocatechin-3-Gallate Enhances Hepatitis C Virus Double-Stranded RNA Intermediates-Triggered Innate Immune Responses in Hepatocytes. Scientific Reports. 2016;6 doi: 10.1038/srep21595.21595
- Friedman M. Overview of antibacterial, antitoxin, antiviral, and antifungal activities of tea flavonoids and teas. Molecular Nutrition & Food Research. 2007;51(1):116–134. doi: 10.1002/mnfr.200600173
- Steinmann J., Buer J., Pietschmann T., Steinmann E. Anti-infective properties of epigallocatechin-3-gallate (EGCG), a component of green tea. British Journal of Pharmacology. 2013;168(5):1059–1073. doi: 10.1111/bph.12009
- Gordon N. C., Wareham D. W. Antimicrobial activity of the green tea polyphenol (−)-epigallocatechin-3-gallate (EGCG) against clinical isolates of Stenotrophomonas maltophilia. International Journal of Antimicrobial Agents. 2010;36(2):129–131. doi: 10.1016/j.ijantimicag.2010.03.025
- Farhad Mollashahi N., Bokaeian M., Farhad Mollashahi L., Afrougheh A. Antifungal efficacy of green tea extract against candida albicans biofilm on tooth substrate. Journal of Dentistry (Tehran, Iran) 2015;12(8):p. 592.
- Baizabal-Aguirre V. M., Rosales C., López-Macías C., Gómez M. I. Control and resolution mechanisms of the inflammatory response. Mediators of Inflammation. 2014;2014 doi: 10.1155/2014/387567.387567
- Scott D. L., Wolfe F., Huizinga T. W. J. Rheumatoid arthritis. The Lancet. 2010;376(9746):1094–1108. doi: 10.1016/S0140-6736(10)60826-4
- Ahmed S., Rahman A., Hasnain A., Lalonde M., Goldberg V. M., Haqqi T. M. Green tea polyphenol epigallocatechin-3-gallate inhibits the IL-1β-induced activity and expression of cyclooxygenase-2 and nitric oxide synthase-2 in human chondrocytes. Free Radical Biology and Medicine. 2002;33(8):1097–1105. doi: 10.1016/S0891-5849(02)01004-3
- Morinobu A., Biao W., Tanaka S., et al. (−)-Epigallocatechin-3-gallate suppresses osteoclast differentiation and ameliorates experimental arthritis in mice. Arthritis and Rheumatism. 2008;58(7):2012–2018. doi: 10.1002/art.23594
- Firestein G. S. Evolving concepts of rheumatoid arthritis. Nature. 2003;423(6937):356–361. doi: 10.1038/nature01661.
- Singh A. K., Umar S., Riegsecker S., Chourasia M., Ahmed S. Regulation of Transforming Growth Factor β-Activated Kinase Activation by Epigallocatechin-3-Gallate in Rheumatoid Arthritis Synovial Fibroblasts: Suppression of K63-Linked Autoubiquitination of Tumor Necrosis Factor Receptor-Associated Factor 6. Arthritis and Rheumatology. 2016;68(2):347–358. doi: 10.1002/art.39447.
- Singh R., Ahmed S., Islam N., Goldberg V. M., Haqqi T. M. Epigallocatechin-3-gallate inhibits interleukin-1β-induced expression of nitric oxide synthase and production of nitric oxide in human chondrocytes: Suppression of nuclear factor κB activation by degradation of the inhibitor of nuclear factor κB. Arthritis and Rheumatism. 2002;46(8):2079–2086. doi: 10.1002/art.10443
- Moon H.-S., Lee H.-G., Choi Y.-J., Kim T.-G., Cho C.-S. Proposed mechanisms of (−)-epigallocatechin-3-gallate for anti-obesity. Chemico-Biological Interactions. 2007;167(2):85–98. doi: 10.1016/j.cbi.2007.02.008
- Thielecke F., Boschmann M. The potential role of green tea catechins in the prevention of the metabolic syndrome—a review. Phytochemistry. 2009;70(1):11–24. doi: 10.1016/j.phytochem.2008.11.011
- Raederstorff D. G., Schlachter M. F., Elste V., Weber P. Effect of EGCG on lipid absorption and plasma lipid levels in rats. Journal of Nutritional Biochemistry. 2003;14(6):326–332. doi: 10.1016/S0955-2863(03)00054-8
- Forester S. C., Gu Y., Lambert J. D. Inhibition of starch digestion by the green tea polyphenol, (−)-epigallocatechin-3-gallate. Molecular Nutrition and Food Research. 2012;56(11):1647–1654. doi: 10.1002/mnfr.201200206
- Scholey A., Downey L. A., Ciorciari J., et al. Acute neurocognitive effects of epigallocatechin gallate (EGCG) Appetite. 2012;58(2):767–770. doi: 10.1016/j.appet.2011.11.016
- Park J.-H., Bae J.-H., Im S.-S., Song D.-K. Green tea and type 2 diabetes. Integrative Medicine Research. 2014;3(1):4–10.
- Wolfram S., Raederstorff D., Preller M., et al. Epigallocatechin gallate supplementation alleviates diabetes in rodents. Journal of Nutrition. 2006;136(10):2512–2518
- Ortsäter H., Grankvist N., Wolfram S., Kuehn N., Sjöholm Å. Diet supplementation with green tea extract epigallocatechin gallate prevents progression to glucose intolerance in db/db mice. Nutrition and Metabolism. 2012;9, article 11 doi: 10.1186/1743-7075-9-11.
- Wolfram S., Raederstorff D., Preller M., et al. Epigallocatechin gallate supplementation alleviates diabetes in rodents. Journal of Nutrition. 2006;136(10):2512–2518.
- Cai E. P., Lin J.-K. Epigallocatechin gallate (EGCG) and rutin suppress the glucotoxicity through activating IRS2 and AMPK signaling in rat pancreatic β cells. Journal of Agricultural and Food Chemistry. 2009;57(20):9817–9827. doi: 10.1021/jf902618v.
- Deng Y.-T., Chang T.-W., Lee M.-S., Lin J.-K. Suppression of free fatty acid-induced insulin resistance by phytopolyphenols in C2C12 mouse skeletal muscle cells. Journal of Agricultural and Food Chemistry. 2012;60(4):1059–1066. doi: 10.1021/jf204496f
- Dong Z. Effects of food factors on signal transduction pathways. BioFactors. 2000;12(1-4):17–28. doi: 10.1002/biof.5520120104