Winter melon
Winter melon also known as Benincasa hispida, wax gourd, ash gourd, white gourd, winter gourd, tallow gourd, ash pumpkin, winter melon, Chinese preserving melon, kundur fruit, chalkumra, and (alu) puhul, is a fruit the family Cucurbitaceae that is eaten as a vegetable when mature 1. The taste is rather bland but sweet 2. Winter melon fruit is covered in a fuzzy coating of fine hairs when young. The immature melon has thick white flesh that tastes sweet. The mature fruit sheds its hairs and forms a waxy white coating, giving the name of “wax gourd.” The gourd wax coating increases the long shelf life of it. Winter melon can grow of a length up to 80 cm and also have broad leaves and yellow flowers 3.
Winter melon is a native of South and Southeast Asia. However, it is commonly grown all over Asia, including Japan, China, Korea, Burma, Ceylon, Sri Lanka, Indonesia, and Australia 2, 4, 5.
Winter melon fruit contains 93–96% water and is rich in nutrients such as vitamin C, vitamin B2 (riboflavin), sodium (Na), and calcium (Ca) 6, 7, 8. Phenolic compounds such as astilbin, catechin, and naringenin have been isolated from winter melon fruit 9. Other bioactive compound constituents include triterpenes (alnusenol, multiflorenol, isomultiflorenol), sterols (lupeol, lupeol acetate, β-sitosterol), glycosides, saccharides, carotenes, β-sitosterin, tannins, and uronic acid 10, 11. However more studies are needed to be done to isolate and identify the bioactive chemical components contained in winter melon 12.
Several lab animal and test tube studies on the functionality of winter melon fruit extracts have reported their antioxidant, anti-inflammatory, anti-ulcerogenic, antidiabetic, and anti-Alzheimer’s efficacy (see Figure 2 below) 13, 14, 15, 16, 17, 18, 19, 20, 2. However, there have been no randomized studies on the effect of ash gourd (winter melon) on the prevention of diseases in human 21. In Chinese medicine, winter melon crust is used to treat urinary dysfunction, and the fruits are used to treat fever 21. In Ayurveda, winter melon fruits are also used to treat epilepsy, lung diseases, asthma, cough, and urinary retention 21.
Figure 1. Winter melon
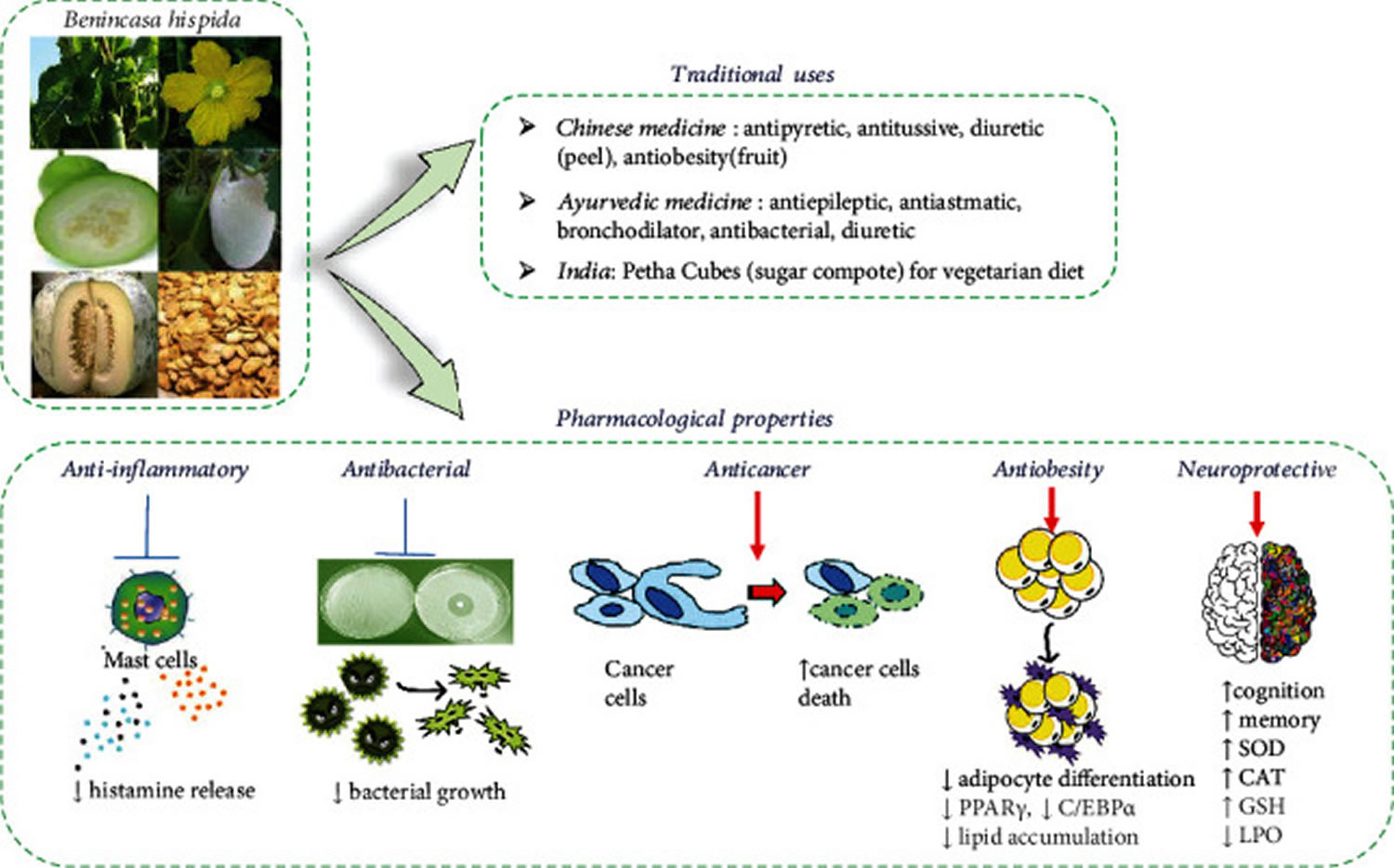
Figure 2. Winter melon uses
Footnotes: Diagram with different parts (aerial parts, flower, fruit pulp unripe, fruits, fruit pulp mature, and seeds) of Benincasa hispida (Thunb.) Cogn, traditional uses, and its most important pharmacological properties.
Abbreviations: PPARγ = peroxisome proliferator-activated receptor gamma; C/EBPα = CCAAT enhancer-binding protein alpha; CAT = catalase; SOD = superoxidase dismutase; GSH = reduced glutathione; LPO = lipid peroxidations.
[Source 21 ]Winter melon nutrition facts
Winter melon fruit contains 93–96% water, proteins (0.30-0.70/100 g), carbohydrates (1.10-4.00/100 g), fat (0.02-0.20/100 g), fiber (0.50-2.10/100 g), and ash (0.27-0.70/100 g).
Vitamins present in the edible portion (per 100 g) of winter melon are vitamin C (13 mg), thiamin (vitamin B1) (0.04 mg), riboflavin (vitamin B2) (0.11 mg), niacin (vitamin B3) (0.4 mg), and vitamin E. Major minerals in the edible portion (per 100 g) include sodium (Na) (111 mg), potassium (K) (6 mg), calcium (Ca) (19 mg), iron (Fe) (0.4 mg), and phosphorus (P) (19 mg) 22, 23, 24, 25. The fruit contains water-soluble polysaccharides 26, such as arabinogalactans 27.
The fruit pulp contains homogalacturonan, β-(1 → 4)-D-galactan, acidic arabinan 28 and natural sugars (e.g., glucose and fructose) 29. The mature fruit also contains organic acids such as malic and citric acid. The fruit also contain many volatile compounds, including (E,E)-2,4-nonadienal, (E)-2-hexenal, n-hexanal, n-hexyl formate, (E,E)-2,4-heptadienal, (Z)-3-hexenal, (E)-2-heptenal, 1-octen-3-ol, 2,5-dimethylpyrazine, 2-methyl pyrazine, 2-ethyl-5-methyl pyrazine, and 2,6-dimethylpyrazine, 2,3,5-trimethylpyrazine 30.
Winter melon seeds contain proteins 31, carbohydrates, phenolic compounds, amino acids, flavonoids, sterols 32, glycosides, alkaloids, fixed oils and fats, phenolic compounds, steroids 33 and unsaturated fatty acids 34. The peel contains alkaloids, saponins, steroids, carbohydrates, flavonoids 35, tannins, carotenoids, oxalates, and phytate 25.
The root contains proteins 31.
Table 1. Winter melon, ash gourd, wax gourd, chinese preserving melon (raw per 100 gram)
| Name | Amount | Unit |
|---|---|---|
| Water | 96.1 | g |
| Energy | 13 | kcal |
| Energy | 54 | kJ |
| Protein | 0.4 | g |
| Total lipid (fat) | 0.2 | g |
| Ash | 0.3 | g |
| Carbohydrate, by difference | 3 | g |
| Fiber, total dietary | 2.9 | g |
| Calcium (Ca) | 19 | mg |
| Iron (Fe) | 0.4 | mg |
| Magnesium (Mg) | 10 | mg |
| Phosphorus (P) | 19 | mg |
| Potassium (K) | 6 | mg |
| Sodium (Na) | 111 | mg |
| Zinc (Zn) | 0.61 | mg |
| Copper (Cu) | 0.023 | mg |
| Manganese (Mn) | 0.058 | mg |
| Selenium (Se) | 0.2 | µg |
| Vitamin C (ascorbic acid) | 13 | mg |
| Thiamin (vitamin B1) | 0.04 | mg |
| Riboflavin (vitamin B2) | 0.11 | mg |
| Niacin (vitamin B3) | 0.4 | mg |
| Pantothenic acid (vitamin B5) | 0.133 | mg |
| Vitamin B6 | 0.035 | mg |
| Folate, total | 5 | µg |
| Folic acid | 0 | µg |
| Folate, food | 5 | µg |
| Folate, DFE | 5 | µg |
| Vitamin B12 | 0 | µg |
| Vitamin A, RAE | 0 | µg |
| Retinol | 0 | µg |
| Vitamin A, IU | 0 | IU |
| Vitamin D (D2 + D3), International Units | 0 | IU |
| Vitamin D (D2 + D3) | 0 | µg |
| Fatty acids, total saturated | 0.016 | g |
| SFA 16:0 | 0.011 | g |
| SFA 18:0 | 0.005 | g |
| Fatty acids, total monounsaturated | 0.037 | g |
| MUFA 18:1 | 0.037 | g |
| Fatty acids, total polyunsaturated | 0.087 | g |
| PUFA 18:2 | 0.087 | g |
| Fatty acids, total trans | 0 | g |
| Cholesterol | 0 | mg |
| Tryptophan | 0.002 | g |
| Lysine | 0.009 | g |
| Methionine | 0.003 | g |
Abbreviations: SFA = saturated fatty acid; MUFA = monounsaturated fatty acid; PUFA = polyunsaturated fatty acid; SFA 16:0 = palmitic (hexadecanoic acid); SFA 18:0 = stearic (octadecanoic acid); MUFA 18:1 = oleic acid; PUFA 18:2 = linoleic acid
[Source 22 ]Winter melon bioactive compounds
Winter melon is rich in phenolic compounds. Several other bioactive compounds present in it are isomultiflorenyl acetate, isovitexin, 1-sinapoylglucose, multiflorenol, 5-gluten-3-β-ylacetate, alnusenol, and benzylalcolcohol-O-α-l-arabinopyranosyl-(1-6)-β-d-glucopyranoside 36. The most representative phytochemicals present in winter melon is shown Table 2.
Table 2. Winter melon chemical phytoconstituents
| Compounds | Plant parts | Locality/country | References |
|---|---|---|---|
| E-2-hexenal, n-hexanal and n-hexyl formate; however, 2,5-dimethylpyrazine, 2,6-dimethylpyrazine, 2,3,5-trimethylprazine, 2-methylpyrazine, 2-ethyl-5-methylpyrazine | Fruit | Taipei, Taiwan/China | 30 |
| Cucumisin-like protease | Sarcocarp | Kagoshima/Japan | 37 |
| Triterpenes, sterols, flavonoid C-glycoside, benzyl glycoside, alnusenol, multiflorenol | Fruit | Kyoto/Japan | 38 |
| Osmotin-like protein | Seeds | New York/USA | 39 |
| Chitinase | Seeds | New York/USA | 40 |
| Astilbin, catechin, naringenin | Fruit | Hainan/China | 36 |
| Di-2-ethylhexyl phthalate | Fruit | Hainan/China | 41 |
| W-sitosterol, V-amyrin, quercetin | Stem | Visakhapatnam/India | 42 |
| β-Carotene | Fruit | Faisalabad/Pakistan | 24 |
| Tryptophan | Fruit | Gwalior/India | 43 |
| Linoleic, palmitic, oleic, and stearic acids | Fruit | Temerloh, Pahang/Malaysia | 44 |
| Acetoin, octanal, nonanal | — | Mumbai/India | 45 |
| α-Tocopherol, δ-tocopherol, linoleic acid, β-sitosterol, campesterol, stigmasterol, Δ5-avenasterol | Fruit | Serdang, Selangor/Malaysia | 46 |
| Galactose, glucose, xylose, sorbose | Peel | Karnataka/India | 47 |
| Linoleic acid, linolenic acid | Seeds | Serdang, Selangor/Malaysia | 48 |
| Myristic acid, palmitoleic acid, oleic acid, linoleic acid, stearic acid, α-linolenic acid, palmitic acid, other saturated and unsaturated fatty acids | Seed oil | Serdang, Selangor/Malaysia | 49 |
| 3α,29-O-di-trans-cinnamoyl-D:C-friedooleana-7,9(11)-diene, oleanolic acid 28-O-β-D-xylopyranosyl-[β-D-xylopyranosyl-(1 → 4)]-(1 → 3)-α-L-rhamnopyranosyl (1 → 2)-α-L-arabinopyranoside, oleanolic acid 28-O-β-D-glucopyranosyl-(1 → 3)-β-D-xylopyranosyl-[β-D-xylopyranosyl-(1 → 4)]-(1 → 3)-α-L-rhamnopyranosyl-(1 → 2)-α-L-arabinopyranoside, multiflorenol, isomultiflorenyl acetate, stigmasterol, stigmasterol 3-O-β-D-glucopyranoside, α-spinasterol, α-spinasterol 3-O-β-D-glucopyranoside, β-sitosterol, daucosterol, arbutin, nicotinic acid, (+)-pinonesinol, ethyl β-D-glucopyranoside | Fruit | Jinghong/China | 50 |
| Phloem lectin-like protein | Exudate | Fukuoka/Japan | 51 |
| Linoleic acid, palmitic acid, oleic acid, stearic acid | Seeds | Rambagh, Allahabad/India | 52 |
| Gallic acid | Fruit | Kota Bharu/Malaysia | 53 |
| Lupeol | Seeds | Mumbai/India | 33 |
| Gallic acid, linoleic acid | Seeds | Serdang, Selangor/Malaysia | 34 |
| β-Sitosterol | Seeds | Mumbai/India | 54 |
| Ascorbic acid | Fruit | Kubang Kerian, Kelantan/Malysia | 55 |
| β-Carotein, ascorbic acid | Peel | Mysore/India | 25 |
| Polysaccharides | Fruit | Guangzhou/China | 56 |
| Gallic acid, catechin, epicatechin, rutin, quercetin, quercetin-3-D-galactoside, trans-ferrulic acid, oleanolic acid, ursolic acid | Fruit | Buzau/Romania | 19 |
Winter melon uses
Scientific reports suggest that winter melon possesses many important nutritious substances, including vitamins, natural sugars, amino acids, organic acids, and mineral elements 57, 23, 58. Winter melon fruit contains 93–96% water and is rich in nutrients such as vitamin C (ascorbic acid), vitamin B2 (riboflavin), sodium (Na), and calcium (Ca) 6, 7, 8. Phenolic compounds such as astilbin, catechin, and naringenin have been isolated from winter melon fruit 9. Other bioactive compound constituents include triterpenes (alnusenol, multiflorenol, isomultiflorenol), sterols (lupeol, lupeol acetate, β-sitosterol), glycosides, saccharides, carotenes, β-sitosterin, tannins, and uronic acid 10, 11.
In India, winter melon is used as a winter season vegetable for a wide variety of diseases. Its medicinal properties have been also recognized in the Ayurvedic system of medicine, spiritual traditions of India and Yoga 21. In Vietnam, its soup (cooked with pork short ribs) is traditionally used by breastfeeding mothers 21. In north India and almost all regions in Bangladesh, it is added with pulses like as moong which usually crushed, along with wax gourd, makes a dish locally called bori, which after sun drying is used in curry dishes and eaten with rice or chapati 59. To make wax gourd soup in China, it is used in stir-fries or added into pork or pork/beef bones, which often served in the scooped-out gourd, carved by scraping off the waxy coating. It is also cut into pieces, candied and normally eaten during the time of New Year festivals, or used as filling in Sweetheart cake. For the Moon Festival, the Chinese and Taiwanese also used it in moon cakes as a base filling. It is candied by the people of the Philippines and is used as a pastry filling for bakpia. In some savoury soups and stir-fries, it also acts as an ingredient. In Nepal, India, and Bangladesh, the tendrils, shoots, and leaves of the plant are consumed as green vegetables 23.
Winter melon is widely used in Chinese medicine, in the treatment of fever, cough accompanied by thick mucus and urinary disorders, it is used especially in bark with a very good diuretic effect 21. The fruit is recommended for overweight people who want to follow diets 21. In Ayurvedic medicine, it is used in the treatment of epilepsy, cough, lung disease, hiccups, asthma, internal bleeding, and urinary retention. In India, a fruit compote called Petha Cubes is made from the pulp of the fruit, which is recommended for vegetarians 60.
Winter melon fruit is also used in peptic ulcer, and it is also used in diabetes mellitus, urinary infection, hemorrhages from internal organs, insanity, epilepsy, and other nervous disorders in Ayurveda 61. The fruit is sweet and traditionally used as a cooling, styptic, antiperiodic, laxative, diuretic, tonic, aphrodisiac, and cardiotonic, and also in jaundice, dyspepsia, urinary calculi, blood disease (e.g., hemorrhages from internal organs), insanity, epilepsy, asthma, diabetes, vitiated conditions of pitta, fever, menstrual disorders, and balancing the body heat 53.
Antioxidant effects
Antioxidants are man-made or natural substances that may prevent or delay some types of cell damage 62. Antioxidants are found in many foods, including fruits and vegetables 62. Antioxidants are also available as dietary supplements. Table 3 shows the antioxidant effects of various parts of winter melon.
The body’s trillions of cells face constant threat from chemicals called free radicals. In very high levels, free radicals are capable of damaging cells and genetic material. The body generates free radicals as the inevitable byproducts of turning food into energy. Free radicals are also formed after exercising or exposure to cigarette smoke, air pollution, and sunlight 62. Oxidative stress is a term used for free radical diseases 63, 64. It is defined as the imbalance between free radicals and antioxidants, given that oxidants (free radicals) are more and have a destructive potential on the human body 65, 66.
The methanolic ash gourd seed extract showed a concentration-dependent (25-200 μg/mL) 2,2-diphenyl-1-picrylhydrazyl (DPPH) and hydrogen peroxide radical scavenging effects 67. Another study revealed that the ethanolic winter melon seed extract shows better DPPH and 2,2′-azino-bis (3-ethylbenzothiazoline-6-sulfonic acid (ABTS) radical scavenging along with total phenolic content (TPC) than its ethyl acetate and n-hexane extracts 48. The seed oil (0.1 mg/mL) also showed significant 2,2-diphenyl-1-picrylhydrazyl (DPPH) and 2,2′-azino-bis (3-ethylbenzothiazoline-6-sulfonic acid (ABTS) radical scavenging capacity 68. This study also determined the total phenolic content in seed oil. The aqueous extract of ash gourd plant reduced reactive oxygen species (ROS) in human umbilical vein endothelial cells (HUVECs) 69.
Polysaccharides of fruit extract showed 2,2-diphenyl-1-picrylhydrazyl (DPPH) free radicals scavenging activity with an EC50 (half-maximal effective concentration) value of 0.98 mg/mL 56. The seed oil also showed DPPH and ABTS radical scavenging capacity. However, the antioxidant activity was lower than the catechin and BHT at the same concentration (0.1 mg/mL) 49. Petroleum ether and methanol fruit extracts increased in catalase (CAT) levels in gastric ulcer rats 70. Hispidalin isolated from this herb also showed DPPH radical scavenging and inhibition of lipid peroxidation capacity 71. The aqueous fruit extract significantly increased the antioxidant status as well as levels of vitamin C concentration in gastric juice of rats 72.
Antioxidant effects of various parts of winter melon on various test models have been also observed by several authors 68, 35, 73, 49, 74, 25, 34, 56.
Table 3. Antioxidant properties of winter melon parts
| Extract/isolated compounds | Test system | Results | References |
|---|---|---|---|
| Crude oil from seeds | DPPH ABTS TPC | DPPH: EC50 = 0.1 mg/mL ABTS: EC50 = 0.1 mg/mL Significant antioxidant effect Standards: methyl ether, fatty acids | 68 |
| Seeds extract | DPPH, ABTS, total phenolic content | EC50 = 10 − 100 μg/mL Significant antioxidant effect Standards: methyl ether, fatty acids | |
| Methanolic and aqueous peel extracts | DPPH | EC50 = 10 − 100 μg/mL Concentration-dependent radical scavenging activity The methanolic extract exhibited a better antioxidant effect Standard: DPPH | 35 |
| Aqueous seeds extract | TPC, TFC DPPH, ABTS, H2O2, linoleic acid oxidation nitrite scavenging assay | TPC: EC50 = 81.3 ± 1.4 μg gallic acid/g TFC: EC50 = 486.8 ± 4.1 μg catechin/g dry mass DPPH: EC50 = 0.6 − 3 mg/mL Concentration-dependent antioxidant activity Standards: catechin 0.05-0.5 mg/mL, BHT, ascorbic acid 10 mg/mL | 73 |
| Seed oil | DPPH ABTS radical scavenging assay | DPPH: EC50 = 0.1 mg/mL The antioxidant activity of the seed oil was lower than the catechin and BHT at the same concentration. Standard: FAME | 49 |
| Hispidalin | DPPH Lipid peroxidation assay | DPPH: EC50 = 2 − 40 μg/mL EC50 = 40 μg/mL Significant DPPH radical scavenging and inhibition of lipid peroxidation capacity Standard: methyl ether | 74 |
| Methanol, ethanol, aqueous peel extracts | DPPH Reducing power assay | Significant antioxidant effect Standard: acarbose 20, 40, 60, 80, 100 μg/mL | 25 |
| Seed extract | DPPH ABTS | Significant antioxidant effect Standard: FAME | 34 |
| Polysaccharides of fruit extract | DPPH | EC50 = 0.98 mg/mL Significant antioxidant effect Standard: glucose | 56 |
Abbreviations: TPC = total phenolic contents; TFC = total flavonoid contents (TFC); ABTS = 2, 2′-azinobis (3-ethylbenzothiazoline-6-sulfonic acid); DPPH = 2,2-diphenyl-1-picrylhydrazyl free radical-scavenging ability; BHT = antioxidant butylated hydroxytoluene; FAME = fatty acid methyl ester; EC50 = the half-maximal effective concentration.
[Source 21 ]Antibacterial, antihelmintic, and larvicidal effects
Due to the excessive use of antibiotics that can lead to the development of antibiotic resistance of various strains of bacteria 75, 76, attempts have been made to use natural antibiotic alternatives 77. Most of these options include plants with antiviral and antibacterial properties that can be effective against gram-negative and gram-negative germs, which are often difficult to eradicate 78. The methanolic ash gourd whole plant extract (500 μg/disc) was found to act against Pseudomonas aeruginosa and Vibrio parahaemolyticus 79. In the latter case, the zone of inhibition was 6 mm only. Hispidalin, an isolated compound from winter melon seeds, was found to act against several bacteria (e.g., Escherichia coli, Pseudomonas aeruginosa, Staphylococcus aureus, and Salmonella enterica) and fungi (e.g., Aspergillus flavus, Penicillium chrysogenum, Fusarium Solani, and Colletotrichum gloeosporioides) 71. In this case, the minimum inhibitory concentrations (MIC = the lowest concentration of an antimicrobial that will inhibit the visible growth of a microorganism after overnight incubation) were 30-120 and 100-200 μg/mL for bacterial and fungal strains, respectively. Moreover, the aqueous, methanol, and petroleum extracts of winter melon seeds showed significant therapeutic efficacy with methanol extract being the best comparable to the antibiotic ciprofloxacin. In other study, the aqueous peel extract showed strong antibacterial activity against S. aureus (MIC = 14.5 μg/mL), Micrococcus luteus (MIC = 8.6 μg/mL), E. coli (MIC = 6.1 μg/mL), and Klebsiella pneumoniae (MIC = 13.4 μg/mL) 20. The herb shows prebiotic activity 80. Table 4 shows the antibacterial, antihelminthic and larvacidal effects of various parts of winter melon.
Ethanolic ash gourd seeds extract (20, 40 and 60 mg) showed a dose-dependent anthelmintic activity on Pheretima posthuma 81. The phloem lectin-like protein from the exudate of ash gourd exerted an inhibitory effect on the Samia ricini larvae 51.
Table 4. Antimicrobial, anthelmintic, and larvicidal effects of winter melon parts
| Extract/isolated compounds | Dose/concentration model (in vitro/in vivo) | Results/mechanisms | References |
|---|---|---|---|
| Antibacterial effects | |||
| Methanolic whole plant extract | Pseudomonas aeruginosa Vibrio parahaemolyticus In vitro Standard: DMSO | IC50 = 500 μg/disc Zone of inhibition = 6 mm | 82 |
| Hispidalin | Staphylococcus aureus, Escherichia coli, Pseudomonas aeruginosa, and Salmonella enterica; Fungi: Penicillium chrysogenum, Fusarium solani, Aspergillus flavus, Colletotrichum gloeosporioides In vitro Standard: acetoin (0.01–20 μg/μl) | Antibacterial: MIC = 30 − 120 μg/mL, Antifungal: MIC = 100 − 200 μg/mL | 74 |
| Aqueous peel extract | Staphylococcus aureus, Micrococcus luteus, Escherichia coli, Klebsiella pneumoniae In vitro Standard: DMSO 150 μL | Antibacterial: MIC = 6.1 − 14.5 μg/mL | 20 |
| Anthelmintic effect (antiparasitic to treat infections with parasitic worms) | |||
| Ethanolic seed extract | Pheretima posthuman/in vitro Standard: phenytoin sodium | IC50 = 20, 40, and 60 mg Dose-dependent anthelmintic effect | 81 |
| Larvicidal effect (insecticide that is specifically targeted against the larval life stage of an insect) | |||
| Phloem lectin-like protein from the exudate | Samia ricini larvae/in vitro Standards: Precision Plus Protein™, serum albumin | ↑ inhibitory activity against the larvae Dose: 70 μg/g | 51 |
Abbreviations: IC50 = value concentration that inhibits cell growth by 50%; MIC = minimum inhibitory concentration.
[Source 21 ]Anti-inflammatory effect
The methanolic ash gourd seeds extract (100-300 mg/kg, orally) showed dose-dependent anti-inflammatory effects on carrageenan-induced paw oedema rat (n = 6) model 67. The winter melon fruit peel methanolic extract showed an anti-inflammatory effect on egg albumin-induced inflammation in rats 83. The petroleum ether and methanolic fruit extract of winter melon (300 mg/kg, oral) showed a dose-dependent anti-inflammatory effect on cotton pellet-induced granuloma models in rats, carrageenan-induced paw oedema, and histamine-induced paw edema 74.
Anticancer effects
The winter melon fruit, seed, and root proteins (10-1000 μg/mL) exerted a concentration-dependent cytotoxic effect on Artemia salina (a species of brine shrimp) 21. The median lethal concentration (LC50) values of ash gourd fruit, seed, and root extract were 44, 41, and 50 μg/mL, respectively 31. In this study, the ash gourd root proteins inhibited the proliferation of HeLa and K-562 cells by 28.50 and 36.60%, respectively 31. Another study reveals that the whole winter melon plant methanolic extract (5-50 μg/mL) exerted a cytotoxic effect on Artemia salina (a species of brine shrimp) (LC50: 45.187 μg/mL) 79. Moreover, the aqueous winter melon seed extract (20-800 μg/mL) did not exert cytotoxic effects on HUVECs and normal fibroblast (NIH/3T3) cells. On male C57BL/6 mice, the extract showed a potent inhibitory effect on basic fibroblast growth factor- (bFGF-) induced angiogenesis 84. The aqueous extract (1–20 μg/mL) also reduced cell adhesion molecules activation by inhibiting monocyte adhesion, ROS, and nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) on high glucose (25 mM) induced HUVECs cells 85.
Table 5 shows the cytotoxic (toxic to cells) and anti-cancer effects of various parts of winter melon.
Table 5. Anticancer effects of various parts of winter melon
| Extract/isolated compounds | Model dose/concentration | Results/mechanisms | References |
|---|---|---|---|
| Aqueous seed extract | HUVECs, NIH/3T3 cells/in vitro Male C57BL/6 mice/in vivo IC50 = 20 − 800 μg/mL Standard: NNGH | No cytotoxicity on HUVECs, NIH/3T3 cells decrease bFGF-induced angiogenesis in mice | 84 |
| Fruit, seed, root proteins | HeLa, K-562 cells/in vitro IC50 of fruit, seeds root extract = 44, 40-50 μg/mL IC50 = 10 − 1000 μg/mL in Artemia salina IC50 = 10–50 μg/mL on HeLa, K-562 cells Standards: lysozime, tyrosine, carbonic anhydrase, ovalbumin, albumin | Decrease cell proliferation by 28.50-36.80% | 31 |
| Aqueous extract | HUVECs cells/in vitro IC50 = 1–20 μg/mL on high glucose (25 mM) Standards: glucose 25 mM, glucose and ABH 5 μg/ml, 20 μg/ml | Decrease cell adhesion molecules activation, Decrease ROS, NF-κB Decrease inhibiting monocyte adhesion | 85 |
| Methanolic/whole plant extract | Artemia salina/in vitro IC50 = 45.186 μg/mL Standard: DMSO, vincristine sulphate 0.91 μg/mL | Increase cytotoxic effect concentration-dependent | 79 |
Abbreviations: IC50 = value concentration that inhibits cell growth by 50%; bFGF = basic fibroblast growth factor; ROS = reactive oxygen species; NF-κB = nuclear factor kappa-light-chain-enhancer of activated B cells; NNGH = N-isobutyl-N-(4-methoxyphenylsulfonyl)-glycylhydroxamic acid.
[Source 21 ]Gastrointestinal protective effects
Fresh ash gourd juice (1-4 mL/animal, orally), ethanol (12, 24 and 48 mg/kg, orally), and petroleum ether extract (0.75, 1.5 and 3 mg/kg, orally) in swimming stress, aspirin plus restraint, serotonin-induced ulcers, and indomethacin plus histamine displayed a dose-dependent anti experimental ulcers effect in rats and mice 60.
The petroleum ether and methanol winter melon fruit extracts (300 mg/kg, orally) significantly reduced ulcer index, vascular permeability, and malondialdehyde (MDA) content, while an increase in CAT levels in comparison to the control group in pylorus ligated gastric ulcers, ethanol-induced gastric mucosal damage, and cold restraint stress- (CRS-) induced gastric ulcer rat models 70. The winter melon fruit extract (1 mL/kg, orally) also decreased ulcer index as well as malondialdehyde (MDA), superoxide dismutase (SOD), and vitamin C levels in indomethacin-induced gastric ulcer in rats 86.
The hydromethanol, ethyl acetate, and aqueous ripe winter melon fruit extracts (20 mg/kg, orally/alternative days) were treated for 14 days in ranitidine (5 mg/kg, orally) induced hypochlorhydria in rats 72. The aqueous extract showed better effects on the test animals. It increased the antioxidant status as well as levels of pepsin, vitamin C, and gastric juice chloride concentration than the other extracts 72. On the other hand, the extract of winter melon fruits with the whole plant of Fumaria vaillantii Loisel (earthsmoke) (1 : 1) (20 mg/kg, orally) was administrated in ranitidine (5 mg/kg) induced hypochlorhydria in rats as pre-and cotreatment manners. The extract significantly enhanced the concentration of pepsin, iron levels in serum, chloride level in gastric juice, and liver along with blood hemoglobin level in experimental animals 87.
A prospective pilot study on dyspeptic patients (n = 20) (baseline between 30 days and 45 days) aged between 18 and 45 years with only single dose of 200 mL winter melon fruit juice every morning in empty stomach for thirty days suggests that a significant improvement of pain, nausea, belching, retrosternal burning, and bowel habits among the patients 88. Table 6 shows the gastrointestinal -protective effects of various parts of winter melon.
Table 6. Gastrointestinal protective effects of various parts of winter melon
| Gastroprotective | Model/dose/concentration | Mechanisms | References |
|---|---|---|---|
| Antiulcer effect | |||
| Fresh juice, petroleum ether, alcoholic/fruits extract | Aspirin plus restraint, serotonin-induced ulcers, indomethacin plus histamine Swimming stress Mice/in vivo Dose: 1 ml/mouse | ↓ulcer index formed by several ulcerogenic | 60 |
| Fresh juice, ethanol, petroleum ether extracts (5% v/v) | Aspirin plus restraint, swimming stress, indomethacin plus histamine, and serotonin-induced ulcers Rats and mice/in vivo Fresh juice (1-4 mL/animal, p.o.), Dose: ethanol extract 12, 24, and 48 mg/kg, p.o. Dose: petroleum ether extract 0.75, 1.5, 3 mg/kg, p.o. | Dose-dependent anti-ulcerogenic effect The fresh juice treatment for 3 months did not change the indices (i.e., WBC, RBC counts HCT, HB, MCV, MCH urea, and sugar) No behavioural changes in experimental animals. | 60 |
| Petroleum ether, methanol/fruits extract | Pylorus ligated (PL) gastric ulcers, ethanol-induced gastric mucosal damage, cold restraint-stress- (CRS-) induced gastric ulcer Rats/in vivo Dose: 300 mg/kg, p.o. | ↓ulcer index ↓MDA ↓vascular permeability ↑ CAT | 70 |
| Fruit extract | Indomethacin-induced gastric ulcer Rats/in vivo Dose: 1 mL/kg, p.o. | ↓ulcer index, ↓MDA ↓SOD, ↓vitamin C | 89 |
| Hydromethanol, aqueous ripe fruit, ethyl acetate extracts | Ranitidine (5 mg/kg) induced hypochlorhydria Rats/in vivo Dose: 20 mg/kg, p.o./alternative days for 14 days | The aqueous extract showed better effects: ↑antioxidant status, ↑pepsin, ↑vitamin C, ↑chloride in gastric juice | 72 |
| Fruit extract with the whole plant of Fumaria vaillantii Loisel (1 : 1) | Ranitidine (5 mg/kg) induced hypochlorhydria Rats/in vivo Dose: 20 mg/kg, p.o. | ↑iron levels in serum, ↑pepsin, ↑gastric juice chloride level and liver ↑blood haemoglobin level | 87 |
| Fruit juice | Prospective pilot study Dyspeptic patients (n = 20) (baseline between 30-45 days); age 18-45 years; 200 mL single-dose every morning in empty stomach for thirty days | ↓pain ↓belching retrosternal burning ↓nausea ↓bowel habits | 88 |
| Antidiarrheal effect | |||
| Methanolic fruit extract | Castor oil-induced diarrheal, PGE2-induced, enter pooling and charcoal meal models Rats/in vivo Dose: 200, 400, and 600 mg/kg, orally by gavage | Dose-dependent antidiarrheal effect ↓PGE2- induced, enter pooling ↓gastrointestinal motility | 90 |
| Methanolic fruit extract | Castor oil, charcoal meal, and antienter pooling models in rats/in vivo Dose: 200, 400, 600 mg/kg, p.o. | ↓ activity against castor oil-induced diarrhoea; ↓PGE2 induced enter pooling ↓gastrointestinal motility | 90 |
Abbreviations: ↑(increased); ↓(decreased); WBC = white blood cells; RBC = red blood cells; HCT = hematocrit; HB = hemoglobin; MCV = mean corpuscular volume; MCH = mean corpuscular haemoglobin concentration; MDA = malondialdehyde; CAT = catalase; SOD = superoxide dismutase; PGE2 = prostaglandin E2.
[Source 21 ]Antidiarrheal effect
The winter melon fruit methanolic extract displayed potential antidiarrheal activity on the castor oil-induced diarrheal rat model 90. It was also seen to inhibit induced PGE2, enter pooling, and reduce in the motility of gastro-intestine in charcoal meal rats 90. The same extract also possessed a significant inhibitory activity against castor oil-induced diarrhoea and induced PGE2, enter pooling and gastrointestinal motility at 200, 400, and 600 mg/kg (orally) in castor oil, charcoal meal, and antienter pooling models in rats 90.
Antidiabetic effects
The methanolic winter melon
stem extract (50,100, 200 mg/kg, orally) dose-dependently lowered the blood glucose level in alloxan-induced diabetic rats 42. The chloroform fruit extract (250 and 500 mg/kg, orally) dose-dependently ameliorated the derangements in lipid metabolism in alloxan-induced diabetic albino rats after 14 days of treatment 91. The study reveals that the methanol, ethanol, and aqueous winter melon peel extracts showed significant α-amylase inhibition activity 25. The ethanol and ethyl ethanoate winter melon leaf extracts lowered the blood glucose level of the diabetic mice in a dose-dependent manner 92. Antidiabetic effects of various parts of winter melon have been shown in Table 7.
Table 7. Winter melon antidiabetic, antiobesity, lipid-lowering and neuroprotective effects
| Extract/isolated compounds | Model dose/concentration | Results/potential mechanisms | References |
|---|---|---|---|
| Lipid-lowering effect | |||
| Hexane, chloroform, ethyl acetate/aqueous fruit extract | 3T3-L1 cells/in vitro | Hexane extract: ↓adipocyte differentiation, ↓PPARγ, ↓C/EBPα, ↓leptin gene expression, ↓lipids accumulation, ↑releasing of glycerol, ↑ triglycerides | 93 |
| Antidiabetic effect | |||
| Methanolic/stem extract | Alloxan-induced diabetes Rats/in vivo Dose: 50,100, 200 mg/kg p.o. | ↓blood glucose level dose-dependent | 42 |
| Chloroform/fruits extract | Alloxan-induced diabetes Rats/in vivo Dose: 250, 500 mg/kg p.o. | Dose-dependently ameliorated the disorders in the metabolism of lipids in diabetic mice | 91 |
| Ethanol, hexane, ethyl ethanoate/leaf extract | STZ-induced diabetes Mice/in vivo Dose: 0.2-1 g/kg, i.p | Ethanol, ethyl ethanoate extracts: ↓blood glucose level | 92 |
| Antiobesity effect | |||
| Methanolic fruit extract | Mice/in vivo Dose: 0.2-1 g/kg, i.p. | Anorexic activity ↓food intake | 94 |
| Antiageing of skin | |||
| Petroleum ether, chloroform, ethyl acetate, methanol/dried fruit pulp extract | Stratum corneum of human skin and dansyl chloride fluorescence models In vitro | Cream prepared from the fruit extract showed the significant antiageing effect | 95 |
| Effects on other diseases | |||
| Fruit methanol extract | Antigen-antibody induced reaction in rats exudate cells/in vitro | ↓histamine release, anti-inflammatory effect Triterpenes, sterols, multiflorenol, alnusenol exerted better inhibitory effects | [38 |
| Methanolic fruits extract | Histamine and acetylcholine-induced bronchospasm Guinea pigs/in vivo Dose: 50, 200, 400 mg/kg, p.o. | Bronchodilator effect: dose-dependent protection against histamine and acetylcholine-induced bronchospasm | 96 |
| Petroleum ether, methanolic/fruits extract | Histamine stimulated paw oedema carrageenan- stimulated paw oedema cotton pellet stimulated granuloma Rats/in vivo Dose: 300 mg/kg, p.o. | ↓histamine release Anti-inflammatory effect | 70 |
| Juice | Isolated rat aortic ring/in vitro Cultured porcine endothelial cells/in vitro Rats/in vivo Dose: 0.4–1.6 mL/kg, i.v. | Antihypertensive effect dose-dependent ↓blood pressure ↑relaxation, ↓contraction of isolated rat aortic ring ↑NO in cultured porcine aortic endothelial cells | 97 |
| Methanolic fruit extract | Renal ischemia/reperfusion injury model Rats/in vivo Dose: 500 mg/kg/day, p.o. for 5 days | Nephroprotective ↓MDA, ↑SOD, CAT, ↑GSH | 98 |
| Ethanolic seeds extract | Ethylene glycol induced chronic Hyperoxaluria model Rats/in vivo Dose:250, 500 mg/kg, p.o. for 35 days | Nephroprotective ↓ urinary oxalate, ↓endogenous oxalate synthesis; ↓urinary protein excretion, ↓kidney oxalate and calcium; ↓elevated serum levels of sodium, creatinine, calcium, phosphorus | 99 |
| Neuroprotective effects | |||
| Fruit juice | Morphine addiction model Mice/in vivo Dose: 1 mL/mouse, p.o. | The development of morphine addiction prevented along with the suppression of opioid withdrawal symptoms | 100 |
| Methanolic fruit extract | Spontaneous motor, muscle relaxant, antihistaminic effect and barbiturate induced hypnosis models Mice, rats, and guinea pigs/in vivo Dose: 200-3000 mg/kg, p.o. | ↑ barbiturate induced hypnosis ↑ antihistaminic activity | 101 |
| Fruit methanol extract | Pentylenetetrazole, strychnine, picrotoxin, and maximal electro seizures model Rats/in vivo Dose: 0.2-1 g/kg, p.o. | Dose-dependent anticonvulsant activity | 102 |
| Methanolic fruit extract | Acetic acid-induced writhing and hot plate Model Mice/in vivo Dose: 200, 400, 600 mg/kg, p.o. | Dose-dependent analgesic effect | 103 |
| Aqueous pulp extract | Colchicine-induced Alzheimer’s model Rats/in vivo Dose: 100-450 mg/kg, p.o. | ↑SOD, ↑CAT, ↑GSH, ↓LPO dose-dependent | 24 |
| Ethanolic seed extract | Rats/in vivo Dose: 250, 500 mg/kg, p.o. | Dose-dependent analgesic and antipyretic effects | 104 |
| Methanolic fruit extract | Marble-burying and motor coordination tests Mice/in vivo Dose: 200, 400, 600 mg/kg, p.o. | Significant dose-dependent anticompulsive effect | 105 |
| Methanolic leaf extract | Acetic acid-induced writhing Mice/in vivo Dose: 50, 100, 200, 400 mg/kg, p.o. | Dose-dependent analgesic effect | 106 |
| Fruit peel methanolic extract | Egg albumin-induced inflammation in rats; acetic acid-induced writhing, formalin-induced pain, hot plate-induced, and pentylenetetrazol-induced convulsions Mice/in vivo Dose: 50, 100, 200, 400 mg/kg, p.o. | Dose-dependently (0.25-1.5 g/kg) inhibited acetic acid-induced writhing, formalin-induced pain licking, and hot plate-induced pain in mice. Significantly inhibition of egg albumin-induced inflammation in rats and pentylenetetrazol-induced convulsion in mice | 83 |
| Ethanolic seed extract | Anticonvulsant activity Mice/in vivo Dose: 250, 500 mg/kg, p.o. | Dose-dependent anticonvulsant effects | 81 |
| Methanolic fruit extract | TST and FST model Mice/in vivo Dose: 50, 100, 200 mg/kg, p.o. | Dose-dependent antidepressant effect possibly through GABAergic involvement. | 107 |
| Petroleum ether, methanolic, aqueous/fruit extracts | Motor coordination, locomotor, cognitive behaviour, anxiolytic, haloperidol-induced catalepsy, and anticonvulsant models Mice/in vivo Dose: 100, 200, 400 mg/kg, p.o. | Dose-dependent anxiolytic, analgesic, and nootropic activity | 108 |
Antiobesity and lipid-lowering effect
Lipids are fatty organic substances that are the largest source of energy for the body. The vast majority of fats are stored in solid form in various organs or skin, and a small part circulates in the blood in liquid form 109. Imbalances in lipid metabolism lead to pathophysiological changes and the appearance of chronic diseases such as cardiovascular disease, fatty liver, endocrine disorders, and diabetes 110. Methanolic fruit extract (0.2-1 g/kg) reduced food intake, suggesting anorectic activity in mice 94. Hexane fraction from the aqueous winter melon fruit extract inhibited adipocyte differentiation by blocking leptin gene expression, peroxisome proliferator-activated receptor gamma (PPARγ), and CCAAT enhancer-binding protein alpha (C/EBPα), resulting in the reduction of lipid accumulation, increased releasing of glycerol and intracellular triglycerides in 3T3-L1 cells 93.
Effects on Alzheimer’s disease
The winter melon fruit extract at a dose of 400 mg/kg (orally) showed a protective effect on colchicine-induced Alzheimer’s disease in rats, possibly through the presence of both vitamin E and β-carotene protecting rat neurons against oxidative stress 24. On the other hand, the aqueous fruit pulp extract (100-450 mg/kg, p.o.) dose-dependently increased SOD, CAT, and GSH, while reduced in LPO levels in the colchicine-induced Alzheimer’s rat model 24.
Effects on memory and cognitive behavior
The methanolic winter melon fruit extract (200, 400, or 600 mg/kg, orally) showed a significant dose-dependent anticompulsive effect in marble-burying and motor coordination test models in mice 105. The petroleum ether, methanolic, and aqueous winter melon fruit extracts (100, 200, and 400 mg/kg, p.o.) showed a dose-dependent nootropic activity in the cognitive behaviour mouse model 108. Kumar and Nirmala 111 also studied the possible nootropic (enhance memory or other cognitive functions) effects of the winter melon fruit on experimental animals.
Anticonvulsant effects
The winter melon fruit methanol extract (0.2-1 g/kg, orally) showed a dose-dependent anticonvulsant activity in pentylenetetrazole, strychnine and picrotoxin, and maximal electro seizures model 102. On the other hand, winter melon fruit peel methanolic extract exerted a dose-dependent (0.25-1.5 g/kg) anticonvulsant effect on pentylenetetrazol-induced convulsion in mouse models 83. Ethanolic winter melon seed extract (250 and 500 mg/kg, p.o.) showed a dose-dependent anticonvulsant effect in anticonvulsant activity in Swiss albino mice 81.
Antidepressant and anxiolytic effects
Methanolic winter melon fruit extract (50, 100, and 200 mg/kg, p.o.) showed a dose-dependent antidepressant-like effect in TST and FST models possibly through GABAergic involvement in Swiss mice 107.
Petroleum ether, methanolic, and aqueous winter melon fruit extracts (100, 200, and 400 mg/kg, p.o.) confirmed a dose-dependent anxiolytic activity in mice 108. Effects of various parts of winter melon on the nervous system are shown in Table 7 above.
Analgesic and antifever effects
The methanolic winter melon fruit extract (200, 400, and 600 mg/kg, orally) showed a dose-dependent analgesic effect in acetic acid-induced writhing and hot plate model in mice 103. The ethanolic winter melon seed extract (250 and 500 mg/kg, p.o.) exerted a dose-dependent analgesic and antipyretic effect in Wistar albino rats 104. Winter melon fruit peel methanolic extract also dose-dependently (0.25-1.5 g/kg) inhibited acetic acid-induced writhing, formalin-induced pain licking, and hot plate-induced pain in mice 83.
In another study, the methanolic winter melon seed extract (100-300 mg/kg, p.o.) also showed a dose-dependent analgesic effect on the rats (n = 6) model 67. The methanolic leaf extract (50-400 mg/kg, p.o.) exerted a dose-dependent analgesic effect in an acetic acid-induced writhing mouse model 106. Petroleum ether, methanolic, and aqueous winter melon fruit extracts (100, 200 and 400 mg/kg, p.o.) showed a dose-dependent analgesic effect in the mouse model 108. The ash gourd juice juice (1 mL, p.o.) prevents morphine addiction development along with the suppression of opioid withdrawal symptoms in mice 100. In experimental animals such as rats, mice, and guinea pigs, the methanolic winter melon fruit extract (200-3000 mg/kg, p.o.) significantly potentiated the barbiturate stimulated hypnosis 101.
Bronchodilatator effect
Winter melon fruit methanol extract inhibited histamine release 38. The methanolic extract (50, 200, and 400 mg/kg, orally) of winter melon exhibited significant protection in guinea pigs against the histamine and acetylcholine-induced bronchospasm 96. The methanolic winter melon fruit extract (200-3000 mg/kg, p.o.) showed significant antihistaminic activity on experimental animals (e.g., rats, mice, and guinea pigs) 101.
Antihypertensive effect
The ACE inhibitory effect of the winter melon plant may show the pharmacological basis in the treatment of high blood pressure for its long time uses in traditional Chinese medicine. The winter melon fruit juice (0.4 – 1.6 mL/kg, i.v.) dose-dependently lowered blood pressure, concentration-dependently showed relaxation of isolated rat aortic rings and produced nitric oxide (NO) from the cultured porcine aortic endothelial cells 97. Polysaccharides of winter melon fruit extract showed an antiglycation effect 56.
Kidney protective effects
Methanolic winter melon fruit extract (500 mg/kg/day, orally) for five days reduced the MDA content, while the increase in SOD, CAT, and GSH levels in renal ischemia/reperfusion injury in female Wistar albino rats 98. Winter melon seed ethanolic extract (250 and 500 mg/kg, orally) for 35 days significantly lowered the increased urinary oxalate, presenting a regulatory action on endogenous oxalate synthesis; decreased in the urinary excretion and kidney retention levels of protein, oxalate, and calcium; and reduced the increased serum levels of sodium, calcium, phosphorus, and creatinine levels in ethylene glycol induced chronic hyperoxaluria in Wistar albino rat 99.
Effects of various parts of winter melon on the kidney are shown in Table 7 above.
Antiageing of skin
A cream prepared from the dried ash gourd fruit pulp extract (petroleum ether, chloroform, ethyl acetate, and methanol) showed a significant antiageing effect on the stratum corneum of human skin and dansyl chloride fluorescence models 95.
Winter melon safety and side effects
The fresh ash gourd juice (5% v/v) treatment for 3 months did not change the total white blood cells (WBC), red blood cells (RBC), hemoglobin (HB), mean corpuscular haemoglobin (MCH), hematocrit (HCT), mean corpuscular volume (MCV), sugar, and urea levels in rats and mice 60. The treatment also caused no behavioural changes in experimental animals 60. The methanolic extract of winter melon fruit was nontoxic and did not cause the death of mice, rats, and guinea pigs in doses up to 3.0 g/kg 101. Other studies, performed in female and male rats, concluded that the standardized hydroalcoholic (70% ethanol) extract of the fruit pulp of winter melon administered orally was relatively safe when to female and male rats 112. Up to oral dose (1000 mg/kg body weight/day) level, no-observe-adverse-effect-level (NOAEL) was obtained for the extract in the 90-days toxicity study. The ethanolic seed extract up to 5000 mg/kg (orally) did not exert toxicity in rats 104. There is a report of Di-2-ethylhexyl phthalate (DEHP), a popularly used plasticizer, contaminated winter melon fruit collected from southern and northern provinces in China and is harmful to human health 41.
References- Benincasa hispida. https://www.ncbi.nlm.nih.gov/data-hub/taxonomy/102211
- Patil J. K., Patel M. R. Pharmacognostic and phytochemical investigation of Benincasa hispida (Thunb.) Cogn. fruit. Pharma Science Monitor . 2012;3:146–156.
- Salunkhe, D. K., & Kadam, S. S. (1998). Handbook of vegetable science and technology: production, compostion, storage, and processing. CRC press.
- Chopra, R.N. Glossary of Indian Medicinal Plants; CSIR: New Delhi, India, 1956.
- Nanjing University of Traditional Chinese Medicine. Dictionary of Traditional Chinese Medicine; Nanjing University of Traditional Chinese Medicine: Nanjing, China, 2006.
- Sahu, P.K.; Sharma, D.; Nair, S.K. Performance of ash gourd genotypes for earliness and yield under chhattisgarh plains, india. Plant Arch. 2015, 15, 1157–1160.
- Siong, T.E.; Noor, M.I.; Azudin, M.N.; Idris, K. Nutrient Composition of Malaysian Foods; ASEAN Sub-Committee on Protein: Kuala Lumpur, Malaysia, 1988.
- Beertsen, W.; Van den Bos, T. Alkaline phosphatase induces the mineralization of sheets of collagen implanted subcutaneously in the rat. J. Clin. Investig. 1992, 89, 1974–1980.
- Du, Q.; Zhang, Q.; Ito, Y. Isolation and identification of phenolic compounds in the fruit of benincasa hispida by HSCCC. J. Liq. Chromatogr. Relat. Technol. 2005, 28, 137–144.
- Gill, N.S.; Dhiman, K.; Bajwa, J.; Sharma, P.; Sood, S. Evaluation of free radical scavenging, anti-inflammatory and analgesic potential of benincasa hispida seed extract. IJP-Int. J. Pharmacol. 2010, 6, 652–657.
- Yoshizumi, S.; Murakami, T.; Kadoya, M.; Matsuda, H.; Yamahara, J.; Yoshikawa, M. Medicinal foodstuffs. XI. histamine release inhibitors from wax gourd, the fruits of benincasa hispida cogn. Yakugaku Zasshi J. Pharm. Soc. Jpn. 1998, 118, 188–192.
- Choi Y-E, Yang J-M, Cho J-H. Benincasa hispida Extract Promotes Proliferation, Differentiation, and Mineralization of MC3T3-E1 Preosteoblasts and Inhibits the Differentiation of RAW 246.7 Osteoclast Precursors. Applied Sciences. 2022; 12(17):8849. https://doi.org/10.3390/app12178849
- Grover, J.K.; Adiga, G.; Vats, V.; Rathi, S.S. Extracts of benincasa hispida prevent development of experimental ulcers. J. Ethnopharmacol. 2001, 78, 159–164.
- Grover, J.K.; Rathi, S.S. Benincasa hispida: An anti-inflammatory agent with cytoprotective activity (abs). Can. J. Physiol. Pharmaol. 1994, 72, 269.
- Patil, R.N.; Patil, R.Y.; Ahirwar, B.; Ahirwar, D. Evaluation of antidiabetic and related actions of some indian medicinal plants in diabetic rats. Asian Pac. J. Trop. Med. 2011, 4, 20–23.
- Rachchh, M.A.; Jain, S.M. Gastroprotective effect of benincasa hispida fruit extract. Indian J. Pharmacol. 2008, 40, 271.
- Roy, C.; Guha, D. Role of benincasa hispida linn. on brain electrical activity in colchicine induced experimental rat model of alzheimer’s disease: Possible involvements of antioxidants. World J. Pharm. Res. 2017, 6, 1439–1443.
- Mandal U, De D, Ali KM, Biswas A, Ghosh D. Effect of different solvent extracts of Benincasa hispida T. on experimental hypochlorhydria in rat. J Adv Pharm Technol Res. 2012 Jan;3(1):41-6. doi: 10.4103/2231-4040.93563
- Busuioc AC, Botezatu AD, Furdui B, Vinatoru C, Maggi F, Caprioli G, Dinica RM. Comparative Study of the Chemical Compositions and Antioxidant Activities of Fresh Juices from Romanian Cucurbitaceae Varieties. Molecules. 2020 Nov 23;25(22):5468. doi: 10.3390/molecules25225468
- Soliman WE, Khan S, Rizvi SMD, Moin A, Elsewedy HS, Abulila AS, Shehata TM. Therapeutic Applications of Biostable Silver Nanoparticles Synthesized Using Peel Extract of Benincasa hispida: Antibacterial and Anticancer Activities. Nanomaterials (Basel). 2020 Sep 30;10(10):1954. doi: 10.3390/nano10101954
- Islam MT, Quispe C, El-Kersh DM, Shill MC, Bhardwaj K, Bhardwaj P, Sharifi-Rad J, Martorell M, Hossain R, Al-Harrasi A, Al-Rawahi A, Butnariu M, Rotariu LS, Suleria HAR, Taheri Y, Docea AO, Calina D, Cho WC. A Literature-Based Update on Benincasa hispida (Thunb.) Cogn.: Traditional Uses, Nutraceutical, and Phytopharmacological Profiles. Oxid Med Cell Longev. 2021 Dec 10;2021:6349041. doi: 10.1155/2021/6349041
- U.S. Department of Agriculture (USDA) FoodData Central. https://fdc.nal.usda.gov
- Zaini N. A. M., Anwar F., Hamid A. A., Saari N. Kundur [Benincasa hispida (Thunb.) Cogn.]: a potential source for valuable nutrients and functional foods. Food Research International . 2011;44:2368–2376. doi: 10.1016/j.foodres.2010.10.024
- Roy C., Ghosh T. K., Guha D. Dose dependent activity of Benincasa hispida on colchicine induced experimental rat model of Alzheimer’s disease. International Journal of Pharmacology . 2008;4(4):237–244. doi: 10.3923/ijp.2008.237.244
- Nagarajaiah S. B., Prakash J. Chemical composition and bioactive potential of dehydrated peels of Benincasa hispida, Luffa acutangula, and Sechium edule. Journal of Herbs Spices & Medicinal Plants . 2015;21:193–202. doi: 10.1080/10496475.2014.940437
- Mazumder S., Lerouge P., Loutelier-Bourhis C., Driouich A., Ray B. Structural characterisation of hemicellulosic polysaccharides from Benincasa hispida using specific enzyme hydrolysis, ion exchange chromatography and MALDI-TOF mass spectroscopy. Carbohydrate Polymers . 2005;59:231–238. doi: 10.1016/j.carbpol.2004.09.014
- Mazumder S., Ray B., Ghosal P. K. Chemical investigation on the polysaccharides present in the mesocarp of chalkumra (Benincasa hispida) fruit. Asian Journal of Chemistry . 2001;13:1389–1395.
- Mazumder S., Morvan C., Thakur S., Ray B. Cell wall polysaccharides from chalkumra (Benincasa hispida) fruit. Part I. Isolation and characterization of pectins. Journal of Agricultural and Food Chemistry . 2004;52:3556–3562. doi: 10.1021/jf0343130
- Wills R. B. H., Wong A. W. K., Scriven F. M., Greenfield H. Nutrient composition of Chinese vegetables. Journal of Agricultural and Food Chemistry . 1984;32(2):413–416. doi: 10.1021/jf00122a059
- Wu C. M., Liou S. E., Chang Y.-H., Chiang W. Volatile compounds of the wax gourd (Benincasa hispida, Cogn) and a wax gourd beverage. Journal of Food Science . 1987;52(1):132–134. doi: 10.1111/j.1365-2621.1987.tb13988.x
- Churiyah, Darusman L. K. Bioactive Proteins from Benincasa hispida (Thunb.) Cogn. Journal of Biosciences . 2009;16(4):161–164. doi: 10.4308/hjb.16.4.161
- Qadrie ZL, Hawisa NT, Khan MW, Samuel M, Anandan R. Antinociceptive and anti-pyretic activity of Benincasa hispida (thunb.) cogn. in Wistar albino rats. Pak J Pharm Sci. 2009 Jul;22(3):287-90.
- Hemant D. U., Doshi G. M. Chromatographic studies on Benincasa hispida (thunb.) Cogn. seed extract scrutinized by HPLC and HPTLC. Pharmacognosy Journal . 2014;6:42–48.
- Bimakr M., Rahman R. A., Ganjloo A., Taip F. S., Adzahan N. M., Sarker M. Z. I. Characterization of valuable compounds from winter melon (Benincasa hispida (Thunb.) Cogn.) seeds using supercritical carbon dioxide extraction combined with pressure swing technique. Food and Bioprocess Technology . 2016;9(3):396–406. doi: 10.1007/s11947-015-1636-3
- Rana S., Suttee D. A. Phytochemical investigation and evaluation of free radical scavenging potential of Benincasa hispida peel extracts. International Journal of Current Pharmaceutical Review and Research . 2012;3:43–46.
- Du Q., Zhang Q., Ito Y. Isolation and identification of phenolic compounds in the fruit ofBenincasa hispidaby HSCCC. Journal of Liquid Chromatography and Related Technologies . 2005;28(1):137–144. doi: 10.1081/JLC-200038620
- Uchikoba T., Yonezawa H., Kaneda M. Cucumisin like protease from the sarcocarp of Benincasa hispida var. ryukyu. Phytochemistry . 1998;49(8):2215–2219. doi: 10.1016/S0031-9422(98)00135-6
- Yoshizumi S., Murakami T., Kadoya M., Matsuda H., Yamahara J., Yoshikawa M. Medicinal Foodstuffs. XI. Histamine release inhibitors from wax gourd, the fruits of Benincasa hispida Cogn. Yakugaku Zasshi . 1998;118(5):188–192. doi: 10.1248/yakushi1947.118.5_188
- Shih C. T., Wu J., Jia S., Khan A. A., Ting K. H., Shih D. S. Purification of an osmotin-like protein from the seeds of Benincasa hispida and cloning of the gene encoding this protein. Plant Science . 2001;160:817–826. doi: 10.1016/s0168-9452(00)00450-7
- Shih C.-Y. T., Khan A. A., Jia S., Wu J., Shih D. S. Purification, characterization, and molecular cloning of a Chitinase from the seeds of Benincasa hispida. Bioscience, Biotechnology, and Biochemistry . 2001;65:501–509. doi: 10.1271/bbb.65.501
- Du Q., Shen L., Xiu L., Jerz G., Winterhalter P. Di-2-ethylhexyl phthalate in the fruits of Benincasa hispida. Food Additives & Contaminants . 2006;23:552–555. doi: 10.1080/02652030500539758
- Battu G. R., Mamidipalli S. N., Parimi R., Viriyala R. K., Patchula R. P., Mood L. R. Hypoglycemic and anti-hyperglycemic effect of alcoholic extract of Benincasa hispida in normal and in alloxan induced diabetic rats. Pharmacognosy Magazine . 2007;3:101–105.
- Girdhar S, Wanjari MM, Prajapati SK, Girdhar A. Evaluation of anti-compulsive effect of methanolic extract of Benincasa hispida Cogn. fruit in mice. Acta Pol Pharm. 2010 Jul-Aug;67(4):417-21. https://www.ptfarm.pl/pub/File/Acta_Poloniae/2010/4/417.pdf
- Sew C., Zaini N., Anwar F., Hamid A., Saari N. Nutritional composition and oil fatty acids of kundur [Benincasa hispida (Thunb.) Cogn.] seed. Pakistan Journal of Botany . 2010;42:3247–3255.
- Sharma J., Chatterjee S., Kumar V., Variyar P. S., Sharma A. Analysis of free and glycosidically bound compounds of ash gourd (Benincasa hispida): Identification of key odorants. Food Chemistry . 2010;122(4):1327–1332. doi: 10.1016/j.foodchem.2010.03.099
- Anwar F., Mohammad N., Othman F., Saari N. Inter-varietal variation in the composition of seeds and seed oils from winter melon [Benincasa hispida (Thunb.) Cogn.] fruit. Pakistan Journal of Botany . 2011;43:2029–2037.
- Kumar C. C., Mythily R., Chandraju S. Extraction and mass characterization of sugars from ash gourd peels (Benincasa hispida) Rasayan Journal of Chemistry . 2012;5:280–285.
- Mandana B., Russly A. R., Farah S. T., Noranizan M. A., Zaidul I. S., Ali G. Antioxidant activity of winter melon (Benincasa hispida) seeds using conventional soxhlet extraction technique. International Food Research Journal . 2012;19:229–234.
- Bimakr M., Rahman R. A., Taip F. S., Adzahan N. M., Sarker M. Z., Ganjloo A. Supercritical carbon dioxide extraction of seed oil from winter melon (Benincasa hispida) and its antioxidant activity and fatty acid composition. Molecules . 2013;18(1):997–1014. doi: 10.3390/molecules18010997
- Han X. N., Liu C. Y., Liu Y. L., Xu Q. M., Li X. R., Yang S. L. New triterpenoids and other constituents from the fruits of Benincasa hispida (Thunb.) Cogn. Journal of Agricultural and Food Chemistry . 2013;61(51):12692–12699. doi: 10.1021/jf405384r
- Ota E., Tsuchiya W., Yamazaki T., Nakamura M., Hirayama C., Konno K. Purification, cDNA cloning and recombinant protein expression of a phloem lectin-like anti-insect defense protein BPLP from the phloem exudate of the wax gourd, Benincasa hispida. Phytochemistry . 2013;89:15–25. doi: 10.1016/j.phytochem.2013.01.013
- Rayees B., Dorcus M., Chitra S. Nutritional composition and oil fatty acids of Indian winter melon Benincasa hispida (Thunb.) seeds. International Food Research Journal . 2013;20:151–155.
- Fatariah Z., Zulkhairuazha T. Y. T., Wan Rosli W. I. Quantitative HPLC analysis of gallic acid in Benincasa hispida prepared with different extraction techniques. Sains Malaysiana . 2014;43:1181–1187.
- Doshi G., Chaskar P., Une H. Elucidation of β-sitosterol from Benincasa hispida seeds, Carissa congesta roots and Polyalthia longifolia leaves by high performance thin layer chromatography. The Pharmacogenomics Journal . 2015;7:221–227.
- Fatariah Z., Zulkhairuazha T. T. Y., Rosli W. W. I. Ascorbic acid quantification in Benincasa hispida fruit extracted using different solvents. International Food Research Journal . 2015;22:208–212.
- Jiang X., Kuang F., Kong F., Yan C. Prediction of the antiglycation activity of polysaccharides from Benincasa hispida using a response surface methodology. Carbohydrate Polymers . 2016;151:358–363. doi: 10.1016/j.carbpol.2016.05.079
- Purohit P, Palamthodi S, Lele SS. Effect of karwanda (Carissa congesta Wight) and sugar addition on physicochemical characteristics of ash gourd (Benincasa hispida) and bottle gourd (Langenaria siceraria) based beverages. J Food Sci Technol. 2019 Feb;56(2):1037-1045. doi: 10.1007/s13197-019-03570-7
- Andrias D. R., Fahmida U., Adi A. C. Nutritional potential of underutilized food crops to improve diet quality of young children in food insecure prone areas of Madura Island, Indonesia. Asia Pacific Journal of Clinical Nutrition . 2019;28:826–836. doi: 10.6133/apjcn.201912_28(4).0020
- Sabale V, Kunjwani H, Sabale P. Formulation and in vitro evaluation of the topical antiageing preparation of the fruit of Benincasa hispida. J Ayurveda Integr Med. 2011 Jul;2(3):124-8. doi: 10.4103/0975-9476.85550
- Grover J. K., Adiga G., Vats V., Rathi S. S. Extracts of Benincasa hispida prevent development of experimental ulcers. Journal of Ethnopharmacology . 2001;78:159–164. doi: 10.1016/S0378-8741(01)00334-8
- Ramesh M., Gayathri V., Rao A. V. N. A., Prabhakar M. C., Rao C. S. Pharmacological actions of fruit juice of Benincasa hispida. Fitoterapia . 1989;60:241–247.
- Antioxidants: In Depth. https://www.nccih.nih.gov/health/antioxidants-in-depth
- Chouikh A. Phytochemical profile, antioxidant, analgesic and hypolipidaemic effects of ephedra alata decne. female cones extract. Farmacia . 2020;68:1011–1020. doi: 10.31925/farmacia.2020.6.7
- Sharifi-Rad J., Dey A., Koirala N., et al. Cinnamomum species: bridging phytochemistry knowledge, pharmacological properties and toxicological safety for health benefits. Frontiers in Pharmacology . 2021;12 doi: 10.3389/fphar.2021.600139
- Sharifi-Rad J., Rodrigues C. F., Sharopov F., et al. Diet, lifestyle and cardiovascular diseases: linking pathophysiology to cardioprotective effects of natural bioactive compounds. International Journal of Environmental Research and Public Health . 2020;17(7):p. 2326. doi: 10.3390/ijerph17072326
- Tsoukalas D., Zlatian O., Mitroi M., et al. A novel nutraceutical formulation can improve motor activity and decrease the stress level in a murine model of middle-age animals. Journal of Clinical Medicine . 2021;10(4):p. 624. doi: 10.3390/jcm10040624
- Gill N., Dhiman K., Bajwa J., Sharma P., Sood S. Evaluation of free radical scavenging, antiinflammatory and analgesic potential of Benincasa hispida seed extract. International Journal of Pharmacology . 2010;6(5):652–657. doi: 10.3923/ijp.2010.652.657
- Bimakr M., Rahman R. A., Taip F. S., Adzahan N. M., Sarker M. Z., Ganjloo A. Optimization of ultrasound-assisted extraction of crude oil from winter melon (Benincasa hispida) seed using response surface methodology and evaluation of its antioxidant activity, total phenolic content and fatty acid composition. Molecules . 2012;17:11748–11762. doi: 10.3390/molecules171011748
- Moon M. K., Kang D. G., Lee Y. J., Kim J. S., Lee H. S. Effect of Benincasa hispida Cogniaux on high glucose-induced vascular inflammation of human umbilical vein endothelial cells. Vascular Pharmacology . 2009;50:116–122. doi: 10.1016/j.vph.2008.11.007
- Rachchh M. A., Jain S. M. Gastroprotective effect of Benincasa hispida fruit extract. Indian journal of pharmacology . 2008;40:271–275. doi: 10.4103/0253-7613.45154
- Sharma S., Verma H. N., Sharma N. K. Cationic Bioactive Peptide from the Seeds of Benincasa hispida. International Journal of Peptides . 2014;2014:12. doi: 10.1155/2014/156060.156060
- Mandal U., De D., Ali K. M., Biswas A., Ghosh D. Effect of different solvent extracts of Benincasa hispida T. on experimental hypochlorhydria in rat. Journal of Advanced Pharmaceutical Technology & Research . 2012;3:41–46. doi: 10.4103/2231-4040.93563
- Samad N. B., Debnath T., Jin H. L., et al. Antioxidant activity of benincasa hispidaseeds. Journal of Food Biochemistry . 2013;37(4):388–395. doi: 10.1111/j.1745-4514.2011.00643.x
- Sharma S, Verma HN, Sharma NK. Cationic Bioactive Peptide from the Seeds of Benincasa hispida. Int J Pept. 2014;2014:156060. doi: 10.1155/2014/156060
- Taheri Y, Joković N, Vitorović J, Grundmann O, Maroyi A, Calina D. The Burden of the Serious and Difficult-to-Treat Infections and a New Antibiotic Available: Cefiderocol. Front Pharmacol. 2021 Jan 14;11:578823. doi: 10.3389/fphar.2020.578823
- Ghenea AE, Cioboată R, Drocaş AI, Țieranu EN, Vasile CM, Moroşanu A, Țieranu CG, Salan AI, Popescu M, Turculeanu A, Padureanu V, Udriștoiu AL, Calina D, Cȃrţu D, Zlatian OM. Prevalence and Antimicrobial Resistance of Klebsiella Strains Isolated from a County Hospital in Romania. Antibiotics (Basel). 2021 Jul 16;10(7):868. doi: 10.3390/antibiotics10070868
- Salehi B., Capanoglu E., Adrar N., et al. Cucurbits plants: a key emphasis to its pharmacological potential. Molecules . 2019;24(10):p. 1854. doi: 10.3390/molecules24101854
- Sharifi-Rad J., Quispe C., Rahavian A., et al. Bioactive compounds as potential agents for sexually transmitted diseases management: a review to explore molecular mechanisms of action. Frontiers in Pharmacology . 2021;12 doi: 10.3389/fphar.2021.674682
- Ullah M. O., Haque M., Urmi K. F., et al. Anti-bacterial activity and brine shrimp lethality bioassay of methanolic extracts of fourteen different edible vegetables from Bangladesh. Asian Pacific Journal of Tropical Biomedicine . 2013;3:1–7. doi: 10.1016/s2221-1691(13)60015-5
- Sreenivas K. M., Lele S. S. Prebiotic activity of gourd family vegetable fibres using in vitro fermentation. Food Bioscience . 2013;1:26–30. doi: 10.1016/j.fbio.2013.01.002
- Qadrie Z., Anandan R., Mushtaque M., Kumar K., Ashraf H. Anthelmintic and anticonvulsant studies of ethanolic extract of Benincasa hispida seeds. Pharmacology . 2011;2:1298–12302.
- Ullah MO, Haque M, Urmi KF, Zulfiker AH, Anita ES, Begum M, Hamid K. Anti-bacterial activity and brine shrimp lethality bioassay of methanolic extracts of fourteen different edible vegetables from Bangladesh. Asian Pac J Trop Biomed. 2013 Jan;3(1):1-7. doi: 10.1016/S2221-1691(13)60015-5
- Parida N. K., Sahu M. R., Debata P. C., Panda P. K. Antinociceptive and antiinflammatory effects of methanolic extract of Benincasa hispida fruit peel in rodents. Asian Journal of Chemistry . 2010;22:7573–7579.
- Lee K. H., Choi H. R., Kim C. H. Anti-angiogenic effect of the seed extract of Benincasa hispida Cogniaux. Journal of Ethnopharmacology . 2005;97(3):509–513. doi: 10.1016/j.jep.2004.12.008
- Moon M. K., Kang D. G., Lee Y. J., Kim J. S., Lee H. S. Effect of Benincasa hispida Cogniaux on high glucose-induced vascular inflammation of human umbilical vein endothelial cells. Vascular Pharmacology . 2009;50:116–122. doi: 10.1016/j.vph.2008.11.00
- Shetty BV, Arjuman A, Jorapur A, Samanth R, Yadav SK, Valliammai N, Tharian AD, Sudha K, Rao GM. Effect of extract of Benincasa hispida on oxidative stress in rats with indomethacin induced gastric ulcers. Indian J Physiol Pharmacol. 2008 Apr-Jun;52(2):178-82.
- Mandal U., Ali K. M., Chatterjee K., De D., Biswas A., Ghosh D. Management of experimental hypochlorhydria with iron deficiency by the composite extract of Fumaria vaillantii L. and Benincasa hispida T. in rat. Journal of Natural Science, Biology, and Medicine . 2014;5:397–403. doi: 10.4103/0976-9668.136202
- Vinaya T. M., Aravind B. S., Sibbritt D., Tapasbrata T., Shivakumar S. The use of _Benincasa hispida_ for the treatment of uninvestigated dyspepsia: Preliminary results of a non-randomised open label pilot clinical trial. Advances in Integrative Medicine . 2015;2(3):130–134. doi: 10.1016/j.aimed.2015.09.003
- Shetty B. V., Arjuman A., Jorapur A., et al. Effect of extract of Benincasa hispida on oxidative stress in rats with indomethacin induced gastric ulcers. Indian Journal of Physiology and Pharmacology . 2008;52:178–182.
- Vrushabendra Swamy B., Rao T., Dhanapal R., Balamuralidhar V., Ashoka Babu V. Antidiarrheal evaluation of Benincasa hispida (Thunb.) Cogn. fruit extracts. Iranian Journal of Pharmacology and Therapeutics (IJPT) . 2005;4:24–27.
- Patil R. N., Patil R. Y., Ahirwar B., Ahirwar D. Evaluation of antidiabetic and related actions of some Indian medicinal plants in diabetic rats. Asian Pacific Journal of Tropical Medicine . 2011;4(1):20–23. doi: 10.1016/s1995-7645(11)60025-4
- Arbotante C., Arriola E. Investigation of the bioactive properties and hypoglycemic effects of ethanol, hexane and ethyl ethanoate extracts from kondol leaves (Benincasa hispida Cogniaux) American Journal of Clinical Pathology . 2016;146(Supplement 1):S33–S38. doi: 10.1093/ajcp/aqw163.008
- You Y., Jun W. Effects of fractions from Benincasa hispida on inhibition of adipogenesis in 3T3-L1 Preadipocytes. Journal of the Korean Society of Food Science and Nutrition . 2012;41(7):895–900. doi: 10.3746/jkfn.2012.41.7.895
- Kumar A., Vimalavathini R. Possible anorectic effect of methanol extract of Benincasa hispida (Thunb). Cogn, fruit. Indian Journal of Pharmacology . 2004;36:348–350.
- Sabale V., Kunjwani H., Sabale P. Formulation and in vitro evaluation of the topical antiageing preparation of the fruit of Benincasa hispida. Journal of Ayurveda and Integrative Medicine . 2011;2(3):124–128. doi: 10.4103/0975-9476.85550
- Kumar A., Ramu P. Effect of methanolic extract of Benincasa hispida againsthistamine and acetylcholine induced bronchospasm in guinea pigs. Indian Journal of Pharmacology . 2002;34:365–366.
- Nakashima M., Shigekuni Y., Obi T., et al. Nitric oxide-dependent hypotensive effects of wax gourd juice. Journal of Ethnopharmacology . 2011;138(2):404–407. doi: 10.1016/j.jep.2011.09.027
- Yagnik B., Jitendra V., Nurudin J., Nilesh K., Rameshvar P., Natavarlal P. Antioxidant activity of Benincasa hispida on renal ischemia/reperfusion injury. Pharmacology . 2009;1:44–49.
- Patel R., Patel S., Shah J. Anti-urolithiatic activity of ethanolic extract of seeds of Benincasa hispida (thumb) Pharmacology . 2011;3:586–591.
- Grover J. K., Rathi S. S., Vats V. Preliminary study of fresh juice of Benincasa hispida on morphine addiction in mice. Fitoterapia . 2000;71(6):707–709. doi: 10.1016/S0367-326X(00)00227-6
- Babu S. C., Ilavarasan R., Refai M. S., Thameemul-Ansari L. H., Kumar D. A. Preliminary pharmacological screening of Benincasa hispida Cogn. Journal of Natural Remedies . 2003;3 doi: 10.18311/jnr/2003/154
- Kumar A., Ramu P. Anti-convulsant activity of Benincasa hispida fruit, methanol extract. Journal of Natural Remedies . 2004;4(2):195–198.
- Hemamalini K., Varma M. V. M. Antinociceptive effects of methanolic extract of Benincasa hispida (Thunb.) Cong. fruit. Pharmacologyonline . 2007;3:327–332.
- Qadrie Z. L., Hawisa N. T., Khan M. W., Samuel M., Anandan R. Antinociceptive and anti-pyretic activity of Benincasa hispida (thunb.) cogn. in Wistar albino rats. Pakistan Journal of Pharmaceutical Sciences . 2009;22:287–290.
- Girdhar S., Wanjari M. M., Prajapati S. K., Girdhar A. Evaluation of anti-compulsive effect of methanolic extract of Benincasa hispida Cogn. fruit in mice. Acta Poloniae Pharmaceutica . 2010;67:417–421.
- Jahan F., Hossain M., Mamun A., et al. An evaluation of antinociceptive effect of methanol extracts of Desmodium gangeticum (L.) Dc. stems and Benincasa hispida (Thunb.) Cogn. leaves on acetic acid-induced gastric pain in mice. Advances in Natural and Applied Science . 2010;4:365–369.
- Dhingra D., Joshi P. Antidepressant-like activity of Benincasa hispida fruits in mice: possible involvement of monoaminergic and GABAergic systems. Journal of Pharmacology and Pharmacotherapeutics . 2012;3(1):60–62. doi: 10.4103/0976-500x.92521
- Ambikar D., Mohanta G. Effect of dried fruit extract of Benincasa hispida on brain behaviour in laboratory animals. Journal of Cell and Tissue Research . 2013;13:3519–3524.
- Amir S., Shah S. T. A., Mamoulakis C., et al. Endocrine disruptors acting on estrogen and androgen pathways cause reproductive disorders through multiple mechanisms: a review. International Journal of Environmental Research and Public Health . 2021;18(4):p. 1464. doi: 10.3390/ijerph18041464
- Ţieranu EN, Donoiu I, Istrătoaie O, Găman AE, Ţieranu ML, Ţieranu CG, Gheonea DI, Ciurea T. Rare case of single coronary artery in a patient with liver cirrhosis. Rom J Morphol Embryol. 2017;58(4):1505-1508.
- Kumar A., Nirmala V. Nootropic activity of methanol extract of Benincasa hispida fruit. Indian Journal of Pharmacology . 2003;35:p. 130.
- Shakya A., Chaudhary S. K., Bhat H. R., Ghosh S. K. Acute and sub-chronic toxicity studies of Benincasa hispida (Thunb.) cogniaux fruit extract in rodents. Regulatory Toxicology and Pharmacology . 2020;118, article 104785 doi: 10.1016/j.yrtph.2020.104785