What is Omega 3 Fatty Acids ?
Omega-3 fatty acids, sometimes referred to as “n-3s,” are a group of polyunsaturated fatty acids (PUFAs) that play vital roles in innate immunity, energy homeostasis, brain development, and cognitive function 1, 2, 3. Omega-3 fatty acids (omega-3s) are essential fats, which mean your body can’t make them from scratch but you must get them from food. Foods high in Omega-3 include fish, vegetable oils, nuts (especially walnuts), flax seeds, flaxseed oil, and leafy vegetables.
There are three main omega-3s are alpha-linolenic acid (ALA), eicosapentaenoic acid (EPA), and docosahexaenoic acid (DHA).
- Eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) also known as “long-chain” omega-3s come mainly from fish and other seafood, so they are sometimes called marine omega-3s.
- Alpha-linolenic acid (ALA), the most common omega-3 fatty acid in most Western diets, is found in vegetable oils and nuts (especially walnuts), flax seeds and flaxseed oil, leafy vegetables, and some animal fat, especially in grass-fed animals. The human body generally uses ALA for energy, and conversion into EPA and DHA is very limited.
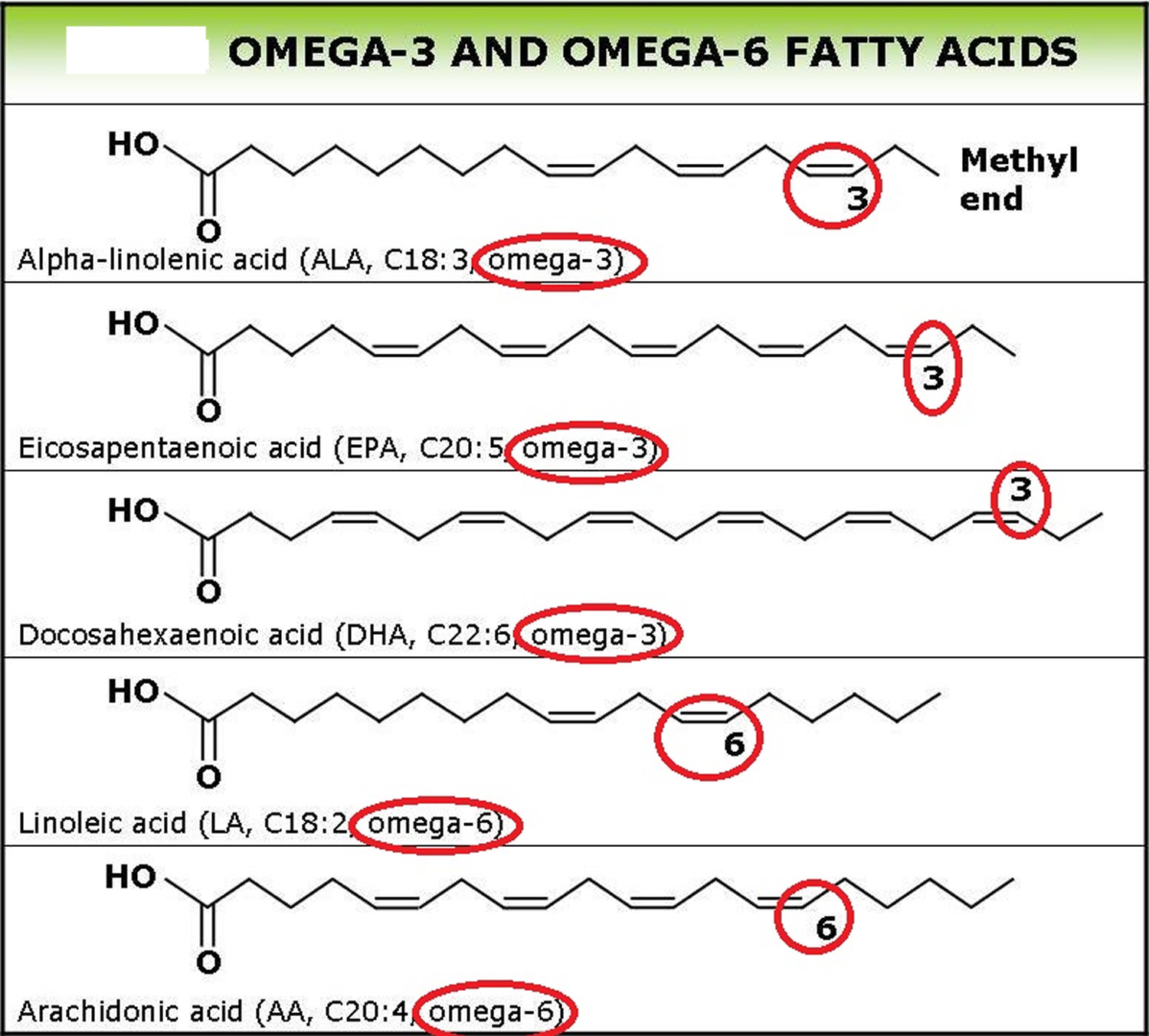
Omega-3 fatty acids (omega-3s) have a carbon–carbon double bond located three carbons from the methyl end of the chain. The human body can only form carbon–carbon double bonds after the 9th carbon from the methyl end of a fatty acid 4. Therefore, alpha-linolenic acid (ALA) and linoleic acid are considered essential fatty acids, meaning that they must be obtained from the diet 5. Alpha-linolenic acid (ALA) can be converted into eicosapentaenoic acid (EPA) and then to docosahexaenoic acid (DHA), but the conversion (which occurs primarily in the liver) is very limited, with reported rates of less than 15% 6. Therefore, consuming EPA and DHA directly from foods and/or dietary supplements is the only practical way to increase levels of these fatty acids in the body.
Alpha-linolenic acid (ALA) is present in plant oils, such as flaxseed, soybean, and canola oils 6. Eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) are present in fish, fish oils, and krill oils, but they are originally synthesized by microalgae, not by the fish. When fish consume phytoplankton that consumed microalgae, they accumulate the omega-3s in their tissues 6.
After ingestion, dietary lipids are hydrolyzed in the intestinal lumen 4. The hydrolysis products—monoglycerides and free fatty acids—are then incorporated into bile-salt– containing micelles and absorbed into enterocytes, largely by passive diffusion. The process is efficient, with an absorption rate of about 95%, which is similar to that of other ingested fats 4. Within intestinal cells, free fatty acids are primarily incorporated into chylomicrons and enter the circulation via the lymphatic system 4. Once in the bloodstream, lipoprotein particles circulate within the body, delivering lipids to various organs for subsequent oxidation, metabolism, or storage in adipose tissue 7.
Omega-3s play important roles in your body as components of the phospholipids that form the structures of cell membranes 8. DHA, in particular, is especially high in the retina, brain, and sperm 9. In addition to their structural role in cell membranes, omega-3 fatty acids (along with omega-6 fatty acids) provide energy for the body and are used to form eicosanoids. Eicosanoids are signaling molecules that have similar chemical structures to the fatty acids from which they are derived; they have wide-ranging functions in the body’s cardiovascular, pulmonary, immune, and endocrine systems 4.
Long-chain Omega-3s such as eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) and their metabolites, have anti-inflammatory and they promote resolution of inflammation 10. In addition to their effects on inflammation, circulating levels of Omega-3s, including EPA and DHA, are inversely associated with fasting and postprandial serum triglyceride (TG) concentrations, largely through attenuation of hepatic very-low-density lipoprotein (VLDL)-triglyceride production 11. Dietary supplementation with these long-chain Omega-3s has been shown consistently to reduce fasting circulating triglyceride (TG) levels and improve lipid accumulation associated with non-alcoholic fatty liver disease (NAFLD) 12.
The 2020–2025 Dietary Guidelines for Americans states that strong evidence from mostly prospective cohort studies but also some randomized controlled trials shows that eating patterns that include seafood are associated with a reduced risk of cardiovascular disease 13. In addition, consuming about 8 ounces (less for children) per week of a variety of seafood that provides about 250 mg per day EPA and DHA is associated with fewer cardiac deaths in both healthy individuals and those with preexisting cardiovascular disease. Those who are pregnant or breastfeeding consume between 8 and 12 ounces per week of a variety of seafood from choices that are lower in mercury. These women should not consume certain types of fish, such as king mackerel, shark, swordfish, and tilefish that are high in methyl mercury, and they should limit the amount of white (albacore) tuna they consume to 6 ounces a week 14. The American Academy of Pediatrics has similar advice for breastfeeding women, recommending intakes of 200–300 mg DHA per day by consuming one to two servings of fish per week to guarantee a sufficient amount of DHA in breast milk 15.
Between 2017 and 2019, the American Heart Association (AHA) released three science advisories on omega-3s 16, 17, 18. All three advisories recommend one to two servings of seafood per week to reduce the risk of congestive heart failure, coronary heart disease, ischemic stroke, and sudden cardiac death, especially when the seafood replaces less healthy foods 18. For people with existing coronary heart disease, such as a recent myocardial infarction, the AHA recommends approximately 1 g/day EPA plus DHA, preferably from oily fish; however, supplements could also be considered under the direction of a physician 17. The American Heart Association does not recommend omega-3 supplements for people who do not have a high cardiovascular disease risk.
Figure 1. Essential fatty acids
What are polyunsaturated fatty acids
Fats are essential for living organisms. Fatty acid molecules have a variable length carbon chain with a methyl terminus and a carboxylic acid head group 19. They can be categorized based on the degree of saturation of their carbon chains. Saturated fatty acids possess the maximal number of hydrogen atoms, while monounsaturated fatty acids (MUFAs) and polyunsaturated fatty acids (PUFAs) have one, or two or more, double bonds, respectively.
Figure 2. Monounsaturated Fatty Acids (MUFAs) Structure
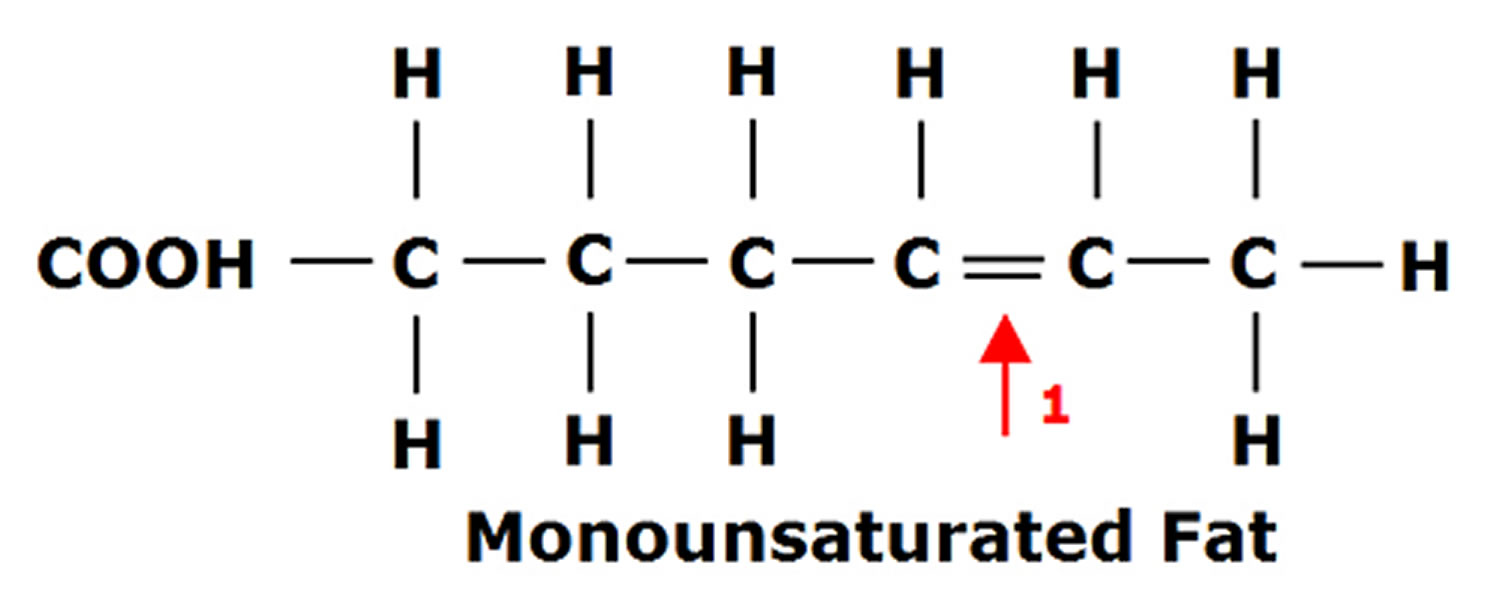
Figure 3. Polyunsaturated Fatty Acids (PUFAs) Structure
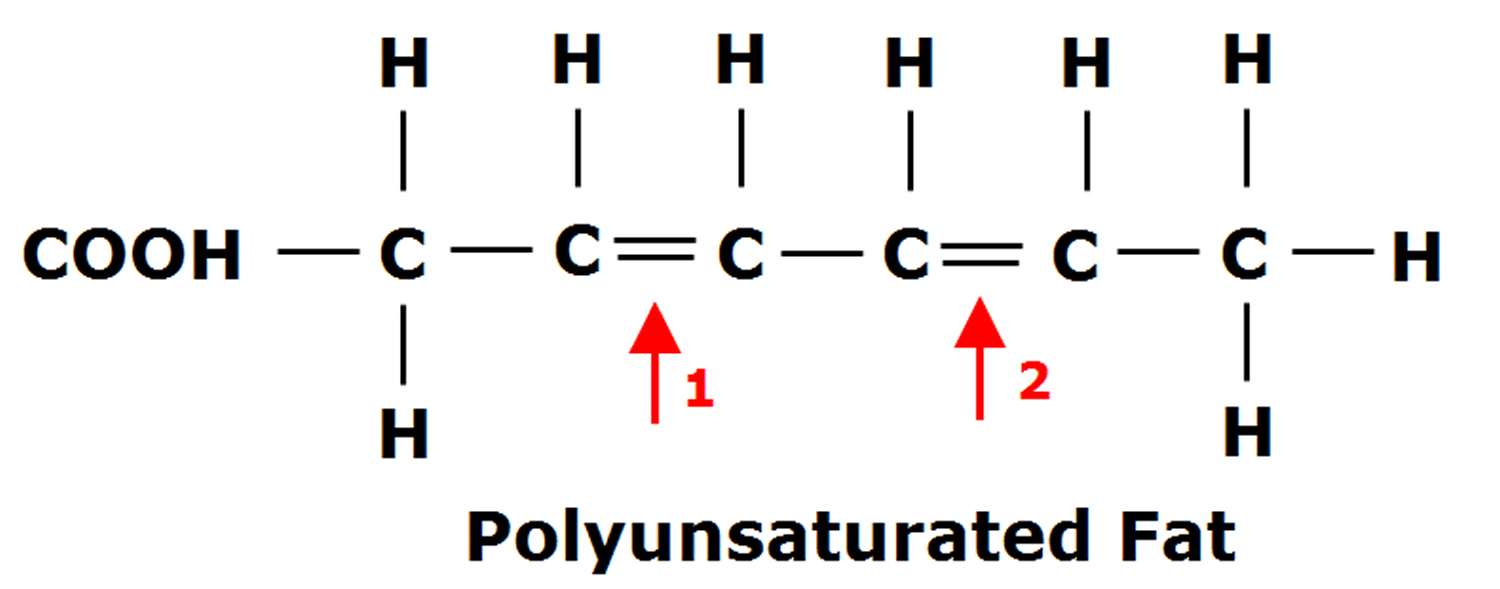
Polyunsaturated fatty acids (PUFAs) can be further subdivided on the basis of the location of the first double bond relative to the methyl terminus of the chain. For example, n-3 and n-6 fatty acids are two of the most biologically significant polyunsaturated fatty acid classes, and have their first double bond on either the third or sixth carbon from the chain terminus, respectively. The final carbon in the fatty acid chain is also known as the omega carbon, hence the common reference to these fatty acids as omega-3 or omega-6 PUFAs. The distinction between omega-6 and omega-3 fatty acids is based on the location of the first double bond, counting from the methyl end of the fatty acid molecule (see Figure 4 below). Omega-6 fatty acids are represented by Linoleic acid (LA) (18:2ω-6) and Arachidonic acid (AA) (20:4ω-6) and omega-3 fatty acids by Alpha-linolenic acid (ALA) (18:3ω-3), Eicosapentaenoic acid (EPA) (20:5ω-3) and Docosahexaenoic acid (DHA) (22:6ω-3).
Long-chain n-3 and n-6 PUFAs are synthesized from the essential fatty acids: alpha-linolenic acid (ALA) and linoleic acid, respectively. Basic structures of these two parent PUFAs are shown in Figure 3. An essential fatty acid cannot be made by the body and must be obtained through dietary sources. Animals and humans have the capacity to metabolize essential fatty acids to long-chain derivatives. Because the n-6 and n-3 pathways compete with one another for enzyme activity, the ratio of n-6 to n-3 PUFAs is very important to human health. An overabundance of fatty acids from one family will limit the metabolic production of the longer chain products of the other. The typical Western diet provides n-6 and n-3 PUFAs in a ratio ranging from 8:1 to 25:1 19, values in severe contrast with the recommendations from national health agencies of approximately 4:1 20. Lowering the n-6:n-3 ratio would reduce competition for the enzymes and facilitate the metabolism of more downstream products of ALA.
Mammalian cells cannot convert omega-6 to omega-3 fatty acids because they lack the converting enzyme, omega-3 desaturase. Omega-6 and omega-3 fatty acids are not interconvertible, are metabolically and functionally distinct, and often have important opposing physiological effects, therefore their balance in the diet is important 21.
This study showed a balanced omega-6/omega-3 ratio 1–2/1 is one of the most important dietary factors in the prevention of obesity, along with physical activity. A lower omega-6/omega-3 ratio should be considered in the management of obesity 21.
Because most diets are already very rich in n-6 PUFAs, greater focus needs to be placed on incorporating n-3 PUFAs into the diet. Dietary sources of n-3 PUFAs are readily available but in limited quantities. Many foods contain alpha-linolenic acid (ALA), including certain vegetable oils, dairy products, flaxseed, walnuts and vegetables 22. Fatty fish, such as mackerel, herring and salmon, provide an excellent source of the long-chain derivatives of ALA, eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) 20.
What are omega-3 fatty acids ?
Omega-3 fatty acids are long-chain polyunsaturated fatty acids (18–22 carbon atoms in chain length) with the first of many double bonds beginning with the third carbon atom (when counting from the methyl end of the fatty acid molecule). The three principal omega-3 fatty acids are:
- Alpha-linolenic acid (ALA) (18:3ω-3),
- Eicosapentaenoic acid (EPA) (20:5ω-3),
- Docosahexaenoic acid (DHA) (22:6ω-3).
Non-fish oil based Omega-3 Polyunsaturated Fatty Acids
The main sources of alpha-linolenic acid (ALA) in the U.S. diet are vegetable oils, particularly canola and soybean oils; flaxseed oil is richer in alpha-linolenic acid (ALA) than soybean and canola oils but is not commonly consumed. The typical North American diet provides approximately 1.4 g of ALA (alpha-linolenic acid) per day, and 0.1 g to 0.2 g of eicosapentaenoic acid (EPA), and docosahexaenoic acid (DHA) 23. Alpha-linolenic acid (ALA) can be converted, usually in small amounts, into eicosapentaenoic acid (EPA), and docosahexaenoic acid (DHA) in the body. Omega-3 EPA and Omega-3 DHA are found in seafood, including fatty fish (e.g., salmon, tuna, and trout) and shellfish (e.g., crab, mussels, and oysters). Note the differences between the Omega-3 fatty acids found in flaxseed, soybean, vegetable oils (alpha-linolenic acid [ALA]) from those in fish and seafoods (Eicosapentaenoic acid [EPA] and Docosahexaenoic acid [DHA]).
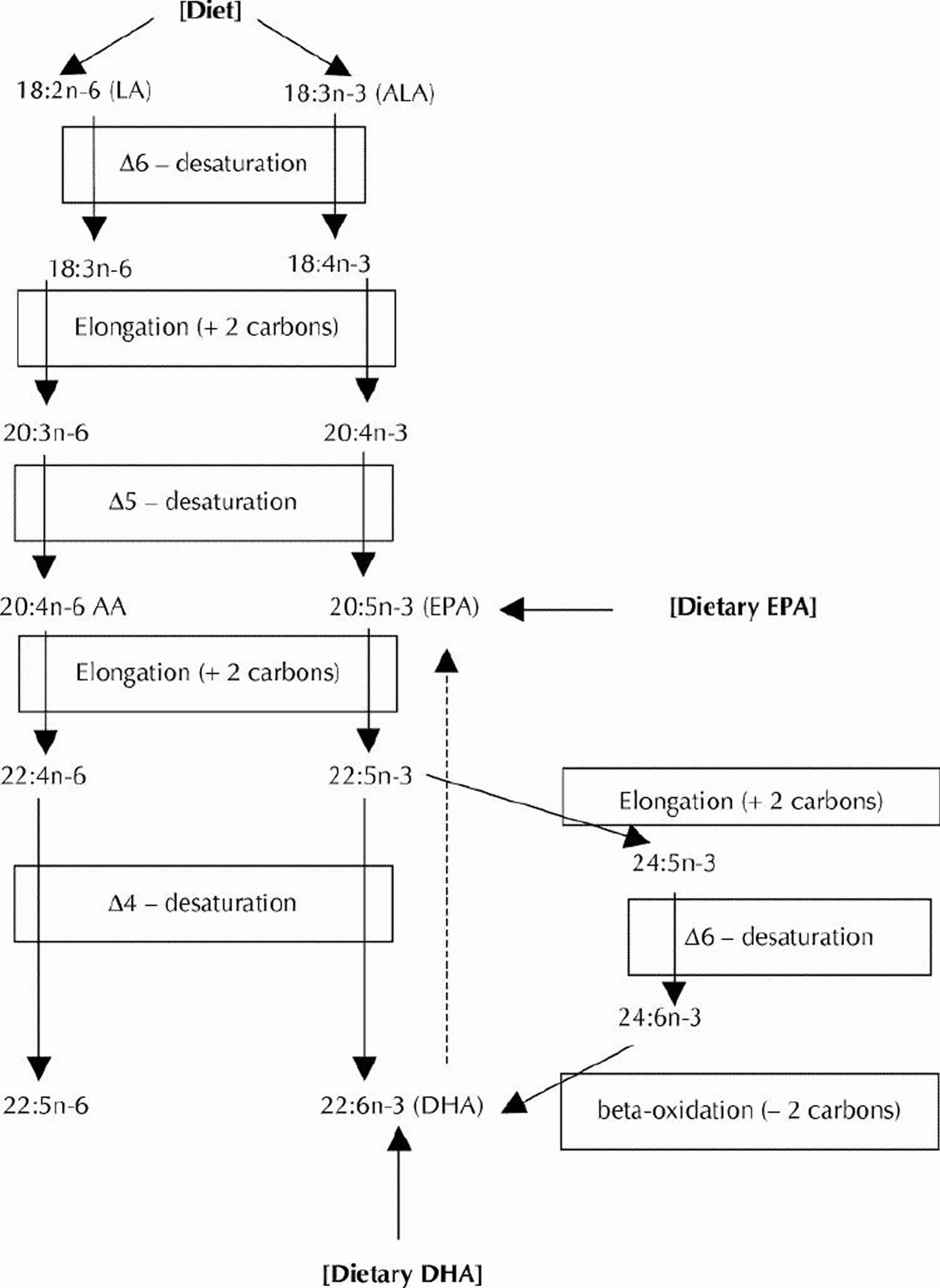
For those who do not consume fish, the omega-3 fatty acid known as alpha-linolenic acid (ALA) can be a dietary source of some metabolically derived Eicosapentaenoic acid (EPA) and Docosahexaenoic acid (DHA). The desaturation plus elongation reactions in the liver and elsewhere in the body that provide for the conversion of dietary ALA to EPA and DHA are depicted in Figure 5. The conversion of ALA to EPA and DHA occurs to a low extent (about 10%–15% efficiency) in the adult human body 24. Nonetheless, there is evidence that the benefits of the Mediterranean-type diet after myocardial infarction may be partly caused by the higher intake of ALA (commonly found in nonhydrogenated canola oil, ground flaxseed and other selected ALA-enriched foods) 25. A prospective cohort study (Nurses Health Study) revealed an inverse relation between ALA intakes and the risk of fatal ischemic heart disease among women 26. However, the Zutphen Elderly Study did not observe a beneficial effect of dietary ALA on the 10-year risk of coronary artery disease 27. The metabolic conversion of ALA to the longer chain omega-3 fatty acids (EPA and DHA) is thought to mediate any possible cardioprotective effects of dietary ALA. In contrast to the well-recognized serum triglyceride-lowering effect of EPA and DHA, most human intervention studies with ALA (e.g., using flaxseed oil) have not exhibited any lipid-lowering effects. Whereas ALA (from flaxseed oil) at a relatively high dose has been found to improve arterial compliance 28, considerably lower supplementation levels of EPA and DHA improved arterial and endothelial functioning in subjects with hypercholesterolemia 29 and subjects with type 2 diabetes mellitus 30.
Fish Oil Omega-3 Polyunsaturated Fatty Acids
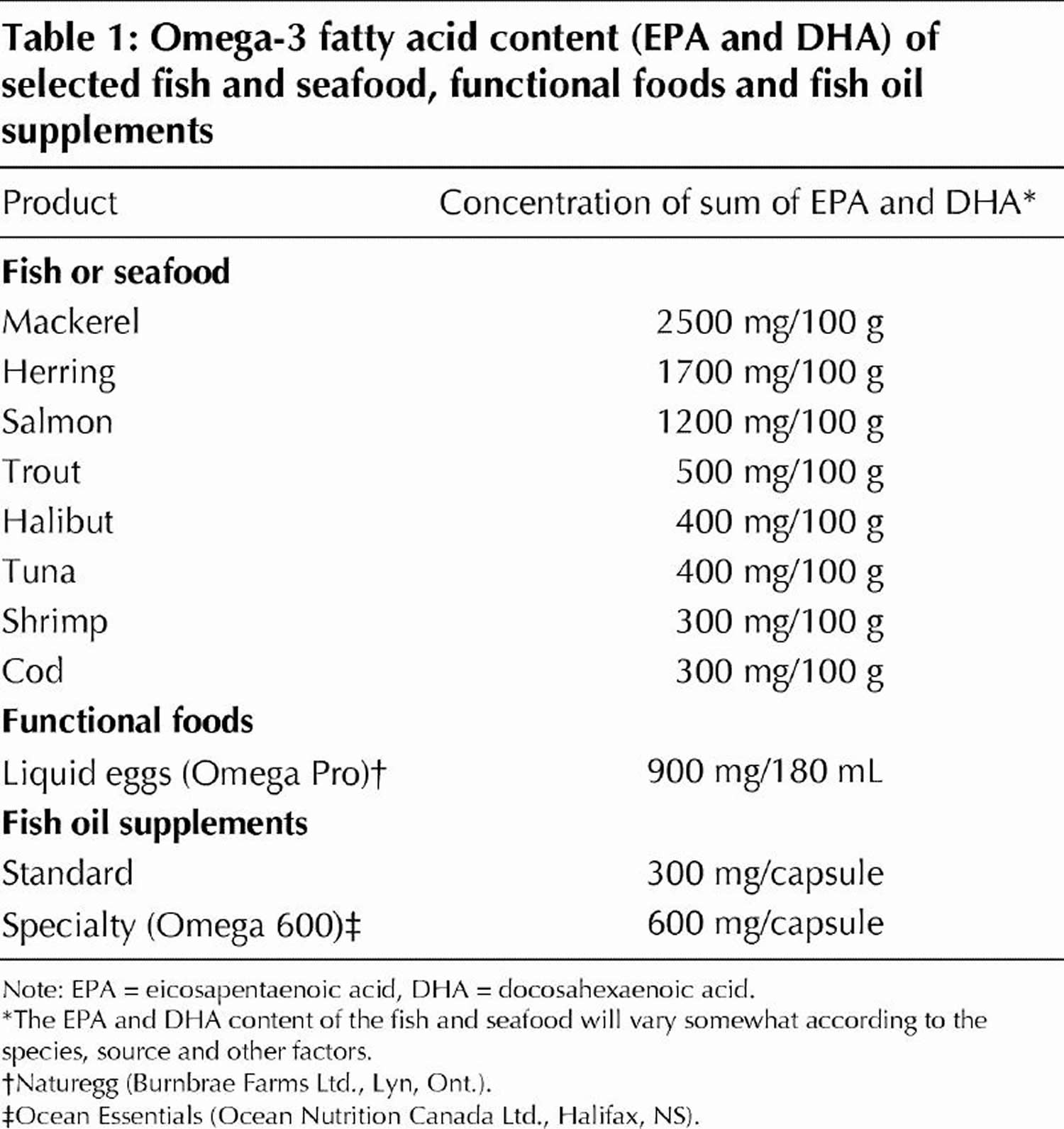
The fish-based and fish-oil–based omega-3 polyunsaturated fatty acids (also referred to as n-3 PUFA) consist of EPA (20 carbon atoms, 5 double bonds) and DHA (22 carbon atoms, 6 double bonds). The general structures for EPA and DHA are shown in Fig. 3. Commonly available dietary sources of EPA and DHA are listed in Table 1. Whereas plant foods and vegetable oils lack EPA and DHA, some do contain varying amounts of the n-3 PUFA alpha-linolenic acid (ALA), which has 18 carbon atoms and 3 double bonds (Fig. 3). Many vegetable oils are greatly enriched in omega-6 fatty acids (mainly as linoleic acid in corn, safflower, sunflower and soybean oils), but canola oil (nonhydrogenated), ground flaxseed and walnuts are rich sources of ALA.
The typical North American diet provides about 1–3 g of n-3 PUFA alpha-linolenic acid (ALA) per day but only 0.10–0.15 g of EPA (Eicosapentaenoic acid) plus DHA (Docosahexaenoic acid) per day 31, 32. The very high intake of n-6 PUFA, mostly as linoleic acid (LA) (Fig. 3) in our diet (12–15 g/day) from common vegetable oils (corn, safflower, soybean) and other sources, yields an overall n-6:n-3 dietary ratio (total omega-6 fatty acids in the diet: total omega-3 fatty acids in the diet) of about 8:1. Health Canada has recommended that this ratio be as low as 4:19 to reduce the competitive influence of high LA intakes on ALA metabolism to its longer chain products (such as EPA and DHA). Although high intakes of LA can provide some modest blood cholesterol lowering, experimental studies in animals have raised concerns regarding the enhancing effect of these high intakes on certain cancers 33. This association has not been established in human studies 34.
Figure 4. Omega-3 fatty acids and Omega-6 fatty acids structure
Figure 5. Conversion of dietary ALA to EPA and DHA via Desaturation, elongation and retroconversion of polyunsaturated fatty acids.
[Source 35]Table 1. Omega-3 Fatty Acid Foods EPA (Eicosapentaenoic acid) and DHA (Docosahexaenoic acid) – Fish and Seafood Sources
[Source 35]Table 2. EPA (Eicosapentaenoic acid) and DHA (Docosahexaenoic acid) Content of Fish Species
| Fish Species and Description | DHA per 100 g | EPA per 100 g | DHA+EPA per 100 g | DHA+EPA per 85 g (3 oz.) |
|---|---|---|---|---|
| Crustaceans, crab, Alaska king, cooked, moist heat | 0.118 | 0.295 | 0.413 | 0.351 |
| Crustaceans, crab, blue, cooked, moist heat | 0.231 | 0.243 | 0.474 | 0.403 |
| Crustaceans, crab, Dungeness, cooked, moist heat | 0.113 | 0.281 | 0.394 | 0.335 |
| Crustaceans, crab, queen, cooked, moist heat | 0.145 | 0.332 | 0.477 | 0.405 |
| Crustaceans, crayfish, mixed species, farmed, cooked, moist heat | 0.038 | 0.124 | 0.162 | 0.138 |
| Crustaceans, crayfish, mixed species, wild, cooked, moist heat | 0.047 | 0.119 | 0.166 | 0.141 |
| Crustaceans, lobster, northern, cooked, moist heat | 0.031 | 0.053 | 0.084 | 0.071 |
| Crustaceans, shrimp, mixed species, cooked, moist heat | 0.144 | 0.171 | 0.315 | 0.268 |
| Crustaceans, spiny lobster, mixed species, cooked, moist heat | 0.139 | 0.341 | 0.48 | 0.408 |
| Fish, anchovy, European, raw | 0.911 | 0.538 | 1.449 | 1.232 |
| Fish, anchovy, European, canned in oil, drained solids | 1.292 | 0.763 | 2.055 | 1.747 |
| Fish, bass, freshwater, mixed species, cooked, dry heat | 0.458 | 0.305 | 0.763 | 0.649 |
| Fish, bass, striped, cooked, dry heat | 0.75 | 0.217 | 0.967 | 0.822 |
| Fish, bluefish, cooked, dry heat | 0.665 | 0.323 | 0.988 | 0.84 |
| Fish, turbot, cooked, dry heat | 0.123 | 0.09 | 0.213 | 0.181 |
| Fish, carp, cooked, dry heat | 0.146 | 0.305 | 0.451 | 0.383 |
| Fish, catfish, channel, farmed, cooked, dry heat | 0.128 | 0.049 | 0.177 | 0.15 |
| Fish, catfish, channel, wild, cooked, dry heat | 0.137 | 0.1 | 0.237 | 0.201 |
| Fish, caviar, black and red, granular | 3.8 | 2.741 | 6.541 | 5.56 |
| Fish, cod, Atlantic, cooked, dry heat | 0.154 | 0.004 | 0.158 | 0.134 |
| Fish, cod, Pacific, cooked, dry heat | 0.173 | 0.103 | 0.276 | 0.235 |
| Fish, croaker, Atlantic, raw | 0.097 | 0.123 | 0.22 | 0.187 |
| Fish, dolphin fish, cooked, dry heat | 0.113 | 0.026 | 0.139 | 0.118 |
| Fish, drum, freshwater, cooked, dry heat | 0.368 | 0.295 | 0.663 | 0.564 |
| Fish, eel, mixed species, cooked, dry heat | 0.081 | 0.108 | 0.189 | 0.161 |
| Fish, fish portions and sticks, frozen, preheated | 0.128 | 0.086 | 0.214 | 0.182 |
| Fish, flatfish (flounder and sole species), cooked, dry heat | 0.258 | 0.243 | 0.501 | 0.426 |
| Fish, grouper, mixed species, cooked, dry heat | 0.213 | 0.035 | 0.248 | 0.211 |
| Fish, haddock, cooked, dry heat | 0.162 | 0.076 | 0.238 | 0.202 |
| Fish, halibut, Atlantic and Pacific, cooked, dry heat | 0.374 | 0.091 | 0.465 | 0.395 |
| Fish, halibut, Greenland, cooked, dry heat | 0.504 | 0.674 | 1.178 | 1.001 |
| Fish, herring, Atlantic, cooked, dry heat | 1.105 | 0.909 | 2.014 | 1.712 |
| Fish, herring, Atlantic, kippered | 1.179 | 0.97 | 2.149 | 1.827 |
| Fish, herring, Pacific, cooked, dry heat | 0.883 | 1.242 | 2.125 | 1.806 |
| Fish, lingcod, cooked, dry heat | 0.13 | 0.133 | 0.263 | 0.224 |
| Fish, mackerel, Atlantic, cooked, dry heat | 0.699 | 0.504 | 1.203 | 1.023 |
| Fish, mackerel, king, cooked, dry heat | 0.227 | 0.174 | 0.401 | 0.341 |
| Fish, mackerel, Pacific and jack, mixed species, cooked, dry heat | 1.195 | 0.653 | 1.848 | 1.571 |
| Fish, mackerel, Spanish, cooked, dry heat | 0.952 | 0.294 | 1.246 | 1.059 |
| Fish, mullet, striped, cooked, dry heat | 0.148 | 0.18 | 0.328 | 0.279 |
| Fish, ocean perch, Atlantic, cooked, dry heat | 0.271 | 0.103 | 0.374 | 0.318 |
| Fish, perch, mixed species, cooked, dry heat | 0.223 | 0.101 | 0.324 | 0.275 |
| Fish, pike, northern, cooked, dry heat | 0.095 | 0.042 | 0.137 | 0.116 |
| Fish, pike, walleye, cooked, dry heat | 0.288 | 0.11 | 0.398 | 0.338 |
| Fish, pollock, Atlantic, cooked, dry heat | 0.451 | 0.091 | 0.542 | 0.461 |
| Fish, pompano, Florida, cooked, dry heat | ?? | ?? | ?? | 0.620 est |
| Fish, rockfish, Pacific, mixed species, cooked, dry heat | 0.262 | 0.181 | 0.443 | 0.377 |
| Fish, roe, mixed species, cooked, dry heat | 1.747 | 1.26 | 3.007 | 2.556 |
| Fish, roe, mixed species, raw | 1.363 | 0.983 | 2.346 | 1.994 |
| Fish, roughy, orange, raw | 0 | 0.001 | 0.001 | 0.001 |
| Fish, sablefish, cooked, dry heat | 0.92 | 0.867 | 1.787 | 1.519 |
| Fish, sablefish, smoked | 0.945 | 0.891 | 1.836 | 1.561 |
| Fish, salmon, Atlantic, farmed, cooked, dry heat | 1.457 | 0.69 | 2.147 | 1.825 |
| Fish, salmon, Atlantic, wild, cooked, dry heat | 1.429 | 0.411 | 1.84 | 1.564 |
| Fish, salmon, Chinook, cooked, dry heat | 0.727 | 1.01 | 1.737 | 1.476 |
| Fish, salmon, chum, cooked, dry heat | 0.505 | 0.299 | 0.804 | 0.683 |
| Fish, salmon, chum, drained solids with bone | 0.702 | 0.473 | 1.175 | 0.999 |
| Fish, salmon, coho, farmed, cooked, dry heat | 0.871 | 0.408 | 1.279 | 1.087 |
| Fish, salmon, coho, wild, cooked, dry heat | 0.658 | 0.401 | 1.059 | 0.9 |
| Fish, salmon, pink, cooked, dry heat | 0.751 | 0.537 | 1.288 | 1.095 |
| Fish, salmon, sockeye, cooked, dry heat | 0.7 | 0.53 | 1.23 | 1.046 |
| Fish, sardine, Atlantic, canned in oil, drained solids with bone | 0.509 | 0.473 | 0.982 | 0.835 |
| Fish, scup, raw (Porgy—assigned to low omega-3 group) | no data | no data | no data | no data |
| Fish, sea bass, mixed species, cooked, dry heat | 0.556 | 0.206 | 0.762 | 0.648 |
| Fish, sea trout, mixed species, cooked, dry heat | 0.265 | 0.211 | 0.476 | 0.405 |
| Fish, shad, American, raw | 1.321 | 1.086 | 2.407 | 2.046 |
| Fish, shark, mixed species, raw | 0.527 | 0.316 | 0.843 | 0.717 |
| Fish, sheepshead, cooked, dry heat | 0.107 | 0.083 | 0.19 | 0.162 |
| Fish, smelt, rainbow, cooked, dry heat | 0.536 | 0.353 | 0.889 | 0.756 |
| Fish, snapper, mixed species, cooked, dry heat | 0.273 | 0.048 | 0.321 | 0.273 |
| Fish, spot, cooked, dry heat | 0.526 | 0.282 | 0.808 | 0.687 |
| Fish, sturgeon, mixed species, cooked, dry heat | 0.119 | 0.249 | 0.368 | 0.313 |
| Fish, sucker, white, cooked, dry heat | 0.371 | 0.244 | 0.615 | 0.523 |
| Fish, sunfish, pumpkin seed, cooked, dry heat | 0.092 | 0.047 | 0.139 | 0.118 |
| Fish, swordfish, cooked, dry heat | 0.681 | 0.138 | 0.819 | 0.696 |
| Fish, tilefish, cooked, dry heat | 0.733 | 0.172 | 0.905 | 0.769 |
| Fish, trout, mixed species, cooked, dry heat | 0.677 | 0.259 | 0.936 | 0.796 |
| Fish, trout, rainbow, farmed, cooked, dry heat | 0.82 | 0.334 | 1.154 | 0.981 |
| Fish, trout, rainbow, wild, cooked, dry heat | 0.52 | 0.468 | 0.988 | 0.84 |
| Fish, tuna, fresh, bluefin, cooked, dry heat | 1.141 | 0.363 | 1.504 | 1.278 |
| Fish, tuna, light, canned in oil, drained solids | 0.101 | 0.027 | 0.128 | 0.109 |
| Fish, tuna, light, canned in water, drained solids | 0.223 | 0.047 | 0.27 | 0.23 |
| Fish, tuna, skipjack, fresh, cooked, dry heat | 0.237 | 0.091 | 0.328 | 0.279 |
| Fish, tuna, white, canned in water, drained solids | 0.629 | 0.233 | 0.862 | 0.733 |
| Fish, tuna, yellowfin, fresh, cooked, dry heat | 0.232 | 0.047 | 0.279 | 0.237 |
| Fish, whitefish, mixed species, cooked, dry heat | 1.206 | 0.406 | 1.612 | 1.37 |
| Fish, whiting, mixed species, cooked, dry heat | 0.235 | 0.283 | 0.518 | 0.44 |
| Fish, wolffish, Atlantic, cooked, dry heat | 0.405 | 0.393 | 0.798 | 0.678 |
| Frog legs, raw | 0.034 | 0.02 | ||
| Mollusks, abalone, mixed species, raw | 0 | 0.049 | 0.049 | 0.042 |
| Mollusks, clam, mixed species, cooked, moist heat | 0.146 | 0.138 | 0.284 | 0.241 |
| Mollusks, conch, baked or broiled | 0.072 | 0.048 | 0.12 | 0.102 |
| Mollusks, cuttlefish, mixed species, cooked, moist heat | 0.132 | 0.078 | 0.21 | 0.179 |
| Mollusks, mussel, blue, cooked, moist heat | 0.506 | 0.276 | 0.782 | 0.665 |
| Mollusks, octopus, common, cooked, moist heat | 0.162 | 0.152 | 0.314 | 0.267 |
| Mollusks, oyster, eastern, farmed, cooked, dry heat | 0.211 | 0.229 | 0.44 | 0.374 |
| Mollusks, oyster, eastern, wild, cooked, dry heat | 0.291 | 0.26 | 0.551 | 0.468 |
| Mollusks, oyster, Pacific, cooked, moist heat | 0.5 | 0.876 | 1.376 | 1.17 |
| Mollusks, scallop, mixed species, cooked, breaded and fried | 0.103 | 0.086 | 0.18 | 0.161 |
| Mollusks, whelk, unspecified, cooked, moist heat | 0.012 | 0.008 | 0.02 | 0.017 |
Table 3. Other sources of Omega-3 Alpha-Linolenic Acid (ALA) – Non-Seafood Sources
| Source of ALA | ALA content, g |
|---|---|
| Pumpkin seeds (1 tbsp) | 0.051 |
| Olive oil (1 tbsp) | 0.103 |
| Walnuts, black (1 tbsp) | 0.156 |
| Soybean oil (1 tbsp) | 1.231 |
| Rapeseed oil (1 tbsp) | 1.302 |
| Walnut oil (1 tbsp) | 1.414 |
| Flaxseeds (1 tbsp) | 2.35 |
| Walnuts, English (1 tbsp) | 2.574 |
| Flaxseed oil (1 tbsp) | 7.249 |
| Almonds (100 g) | 0.4 |
| Peanuts (100 g) | 0.003 |
| Beans, navy, sprouted (100 g) | 0.3 |
| Broccoli, raw (100 g) | 0.1 |
| Lettuce, red leaf (100 g) | 0.1 |
| Mustard (100 g) | 0.1 |
| Purslane (100 g) | 0.4 |
| Spinach (100 g) | 0.1 |
| Seaweed, spirulina, dried (100 g) | 0.8 |
| Beans, common, dry (100 g) | 0.6 |
| Chickpeas, dry (100 g) | 0.1 |
| Soybeans, dry (100 g) | 1.6 |
| Oats, germ (100 g) | 1.4 |
| Rice, bran (100 g) | 0.2 |
| Wheat, germ (100 g) | 0.7 |
| Avocados, California, raw (100 g) | 0.1 |
| Raspberries, raw (100 g) | 0.1 |
| Strawberries, raw (100 g) | 0.1 |
| Novel sources of ALA | ALA content, g |
| Breads and pasta (100 g) | 0.1–1.6 |
| Cereals (and granola bars) (55 g) | 1.0–4.9 |
| Eggs (50 g or 1 egg) | 0.1–0.6 |
| Processed meats (100 g) | 0.5 |
| Salad dressing (14 g – 31 g) | 2.0–4.0 |
| Margarine spreads (10 g – 100 g) | 0.3–1.0 |
| Nutrition bars (50 g) | 0.1–2.2 |
Some types of omega-3s are found in foods such as fatty fish and shellfish. Another type is found in some vegetable oils. Omega-3s are also available as dietary supplements. Note the differences between the Omega-3 fatty acids found in flaxseed, soybean, vegetable oils (alpha-linolenic acid (ALA)) from those in fish and seafoods (Eicosapentaenoic acid (EPA) and Docosahexaenoic acid (DHA)).
Commonly used dietary supplements that contain omega-3s include fish oil (which provides EPA and DHA) and flaxseed oil (which provides ALA). Algae oils are a vegetarian source of DHA.
Omega-3 fatty acids are important for a number of bodily functions, including muscle activity, blood clotting, digestion, fertility, and cell division and growth. DHA is important for brain development and function. ALA is an “essential” fatty acid, meaning that people must obtain it from food or supplements because the human body cannot manufacture it.
Possible biochemical and physiological mechanisms of action for Eicosapentaenoic acid (EPA), and Docosahexaenoic acid (DHA)
The various mechanisms by which increased dietary consumption of omega-3 fatty acids from fish and fish oils is considered to favorably modify cardiovascular disease and associated disorders are outlined in Table 4. Increasing the intake of EPA and DHA results in a corresponding increase of these omega-3 fatty acids in tissue or cellular lipids and circulatory lipids 38, 39 along with a simultaneous reduction in the omega-6 fatty acids such as LA and arachidonic acid (AA) (Fig. 3). The fatty acid shifts are particularly pronounced in the cell membrane–bound phospholipid components. These changed profiles alter the physicochemical properties of cell membranes and their functioning and modify cell signalling, gene expression and biosynthetic processes, and eicosanoid formations (the eicosanoids formed via oxygenase enzymes acting on AA and EPA include prostaglandins, leukotrienes and thromboxanes.). The beneficial effects of omega-3 fatty acids on cardiovascular disease are mediated by both eicosanoid-dependent and eicosanoid-independent processes. For example, the reduced blood platelet reactivity (antithrombotic effect) observed with increased EPA and DHA intakes involves the reduced formation of the proaggregatory eicosanoid known as thromboxane A2 (TxA2). The replacement of AA (the omega-6 fatty acid and TxA2 precursor) in blood platelet membrane phospholipid by EPA and DHA yields less TxA2 upon platelet stimulation; furthermore, EPA has an inhibitory effect on the cyclo-oxygenase enzyme that connects AA to TxA2 thus leading to a lessened thrombogenic state 35.
Table 4. Cardio-protective Effects of EPA and DHA
Fish Rich in Omega-3 Fatty Acids Diet Health Benefits
Multiple prospective cohort studies support the benefit of a fish rich in omega-3 fatty acid diet 40, 41, 42.
It has long been recognized that disease patterns for the Greenland Inuit, when compared with those for the population of Denmark, exhibit a significantly lower rate of death from acute myocardial infarction despite only moderate differences in blood cholesterol levels 43. The high-fat traditional Inuit diet (Greenland; Nunavik) provides up to several grams of omega-3 fatty acid (EPA and DHA) daily in the form of marine mammals (seal, whale), wildfowl (seabirds) and various fish 43, 44. Furthermore, the higher fish intakes of the Japanese population relative to that of North America have been associated with considerably lower rates of acute myocardial infarctions, other ischemic heart disease and atherosclerosis despite only moderately lower blood cholesterol levels in the Japanese population 45. Various studies have also indicated that long-term consumption of fish (up to 2–3 servings per week) appears to be associated with lower primary and secondary heart attack rates and death from cardiovascular disease 46, 47, 48. Following dietary consumption, the levels of EPA and DHA rise considerably in mammalian cells and tissues via their esterification into the 2-position (that is, the middle carbon of the glycerol backbone in a phospholipid structure) of the membrane phospholipid components. Fatty acid analyses of serum and plasma phospholipid, a biomarker for EPA and DHA intake and physiological status, have indicated that omega-3 fatty acids in general and DHA levels in particular are inversely correlated with coronary heart disease in men 49. Among the Inuit of Nunavik, progressive increases in levels of EPA and DHA in plasma phospholipid have been found both to reflect dietary intakes of these fatty acids and to be beneficially associated with key risk factors for cardiovascular disease 39.
Intervention studies using fish oil concentrates that provide EPA and DHA at intakes of up to 2–4g/day over a few weeks have shown that these fatty acids can favourably attenuate various risk factors for cardiovascular disease (independent of any blood cholesterol-lowering effect) 38, 50, 51. These effects include an antithrombotic effect, lipid (triglyceride) lowering, reduced blood and plasma viscosity, and improvements in endothelial dysfunction 38, 50, 51, 29. Omega-3 fatty acids accumulate to a considerable extent in various sites including circulating blood platelets, the heart and serum phospholipid. The accumulation of EPA and DHA in platelets is associated with decreased platelet adhesiveness and aggregation and an overall reduction in thrombogenicity. Antiatherogenic effects of omega-3 fatty acids have also been shown in animal studies.
Human studies have revealed the potent ability of EPA and DHA to significantly reduce circulating levels of blood triglyceride 52, which is of interest because only moderate elevations in triglyceride (approaching 1.33 mmol/L or 118 mg/dL, or above) have been associated with a progressively increased risk of ischemic heart disease 53. Within 2–3 weeks of EPA and DHA supplementation, significantly reduced blood triglyceride levels with an approximate reduction of 6%–8% (or more) per gram of EPA and DHA consumed are routinely observed. In a placebo-controlled, double-blind trial, a 26% lowering in fasting triglyceride levels in postmenopausal women receiving 4 g omega-3 (EPA and DHA) daily over 28 days was recently demonstrated 54. In cases where combined statin therapy plus certain triglyceride-lowering therapies (e.g., fibric acid derivatives) may be contraindicated, statin plus omega-3 (EPA and DHA) therapy may be an attractive alternative. Supplementation with omega-3 (EPA and DHA), as given in addition to statin therapy in patients with combined hyperlipidemia, was found to reduce levels of atherogenic lipoproteins while more effectively reducing the hemostatic risk profile 55. The antiarrhythmic potential of EPA and DHA (upon accumulation in cardiac tissue) has been considered to be yet another important mechanism by which consumption of these fatty acids can reduce mortality related to cardiovascular disease (particularly sudden cardiac death) 56. This last effect is considered to be exhibited at even lower intakes of omega-3 (EPA and DHA combined) of about 1 g/day 46.
The Lyon Diet Heart Study 57 randomized 605 participants with a previous heart attack for 46 months and showed an inverse relationship between alpha-linolenic acid (ALA) intake and the risk of a second heart attack. The intervention group was advised to eat more fish, fruits, and vegetables and to use an ALA-rich margarine. The control group was advised to follow a prudent diet. There was a 68% decrease in primary end points (cardiac death and nonfatal heart attack). Secondary end points (periprocedural infarctions, unstable angina, heart failure, stroke, and pulmonary or peripheral embolisms) also decreased. Of note, at four-year follow-up, most experimental patients were still closely following the recommended diet.
The results of this study suggested that higher prenatal fish intake and exposure to elongated n−3 PUFAs were associated with lower adiposity in early childhood. In this cohort, as in other populations in North America and Western Europe, the prenatal intake of elongated n−3 fatty acids was well below recommended concentrations. A higher n−3 PUFA intake may be associated with lower rates of obesity as well as less atopy and improved neurocognitive development 58.
In summary, there is evidence for the beneficial effect of regular fish consumption (up to 2–3 times/week) both in healthy subjects and in those at considerable risk for coronary artery disease or with established coronary artery disease.
Moderate evidence has emerged about the health benefits of eating seafood. The health benefits of omega-3 dietary supplements are unclear.
Omega-3 fatty acids and Body Weight Management
Several studies have provided supporting evidence for a role of omega-3 PUFAs in body composition 59. In this twenty-seven women with type 2 diabetes without hypertriglyceridemia were randomly allocated in a double-blind parallel design to 2 months of 3 g/day of either fish oil (1.8 g n−3 (containing 1.8 g n−3 PUFAs: 1.08 g eicosapentaenoic acid and 0.72 g docosahexaenoic acid) or placebo (paraffin oil). The participants had fasting plasma glucose between 7.7 and 14.0 mmol/L, glycated hemoglobin (HbA1c) of 7–10.5%, age from 40 to 60 yrs, body mass index (BMI; in kg/m2) between 27 and 40, and plasma triacylglycerol <2.5 mmol/L. The results after 2 months of taking fish oil show body weight and energy intake measured by use of a food diary were unchanged, but their total fat mass and subcutaneous adipocyte diameter were lower in the fish oil group than in the placebo group. Insulin sensitivity was not significantly different between the 2 groups. By contrast, atherogenic risk factors, including plasma triacylglycerol, the ratio of triacylglycerol to HDL cholesterol (atherogenic index) and plasma plasminogen activator inhibitor-1, were lower in the fish oil group than in the placebo group. In addition, a subset of inflammation-related genes was reduced in subcutaneous adipose tissue after the fish oil, but not the placebo 59.
In this fish oil and weight reduction study 60, a total of 324 men and women aged 20-40 years, BMI 27.5-32.5 kg/m2 from Iceland, Spain and Ireland, were given energy-restricted diet with varying amount in fish and fish oil content for 8 weeks. Subjects were randomized to one of four groups: (1) control (sunflower oil capsules, no seafood); (2) lean fish (3 x 150 g portions of cod/week); (3) fatty fish (3 x 150 g portions of salmon/week); (4) fish oil (DHA/EPA capsules, no seafood). The macronutrient composition of the diets was similar between the groups and the capsule groups, were single-blinded. The energy-restricted diet for an average man in the study (95 kg at baseline) was 1600 kcal/day. The men in that study lost weight, but not the women. In young, overweight men, the inclusion of either lean or fatty fish, or fish oil as part of an energy-restricted diet resulted in approximately 1 kg more weight loss after 4 weeks, than did a similar diet without seafood or supplement of marine origin. The addition of seafood to a nutritionally balanced energy-restricted diet may boost weight loss 60.
In this study the addition of omega-3 fatty acids to a calorie-restricted diet to treat overweight or obesity in overweight and obese volunteer (31+/-5 years; BMI: 28.3 kg/m2) during the last 2 weeks of an 8-week energy-restricted balanced diet providing either a low (<260 mg/day ) or a high amount (>1300 mg/day; n=121) of omega-3 fatty acids. The results were the addition of omega-3 fatty acids cause less hunger and more fullness in the overweight or obese volunteers 61.
These findings support a potential role for omega-3 in appetite regulation in humans. Some intervention studies showed that omega-3 fatty acid supplementation reduced body weight and obesity in lean 62, overweight 60, 63 and obese 64 individuals. Couet et al. 62 noted a 22% increase in basal lipid oxidation with 6 grams of fish oil for 3 weeks. Omega-3 fatty acids are long term metabolic fuel partitioners with greater partitioning towards β-oxidation in men than in women.
Omega-3 fatty acids for those at increased Cardiovascular Disease
Many studies show that eating fatty fish and other types of seafood as part of a healthy eating pattern helps keep your heart healthy and helps protect you from some heart problems. This interest was spurred by epidemiological research dating back to the 1970s that found low rates of heart attack (myocardial infarction) and other coronary events among Greenland Inuit and other fish-eating populations, such as those in Japan 6. Results from observational studies have been consistent with these findings, with several systematic reviews and meta-analyses showing that higher consumption of fish and higher dietary or plasma levels of omega-3s are associated with a lower risk of heart failure, coronary disease, and fatal coronary heart disease 65.
The American Heart Association (AHA) recommends eating one to two servings of seafood per week to reduce your risk of some heart problems, especially if you consume the seafood in place of less healthy foods. For people with heart disease, the AHA recommends consuming about 1 g per day EPA plus DHA, preferably from oily fish, but supplements are an option under the guidance of a healthcare provider. The AHA does not recommend omega-3 supplements for people who do not have a high risk of cardiovascular disease.
Many studies have assessed the effects of omega-3s—primarily EPA and DHA—on cardiovascular disease and cardiovascular disease risk factors, such as high blood pressure and elevated plasma lipids.
Clinical trial data from the 1989 Diet and Reinfarction Trial, the 1999 open-label GISSI-Prevenzione trial 66 and others supported the hypothesis that long-chain omega-3s offer protection from cardiovascular disease by reducing the heart’s susceptibility to arrhythmias, lowering triglyceride levels, lowering blood pressure, and decreasing platelet aggregation 67. The authors of a systematic review that included six secondary-prevention trials and one primary-prevention trial of omega-3 supplementation published between 1966 and 2005 concluded that consumption of long-chain omega-3s from fish and fish oil supplements reduces rates of all-cause mortality, cardiac death, sudden death, and stroke 67. They noted that the evidence of benefit is stronger for secondary than for primary prevention.
Results from the Japan EPA Lipid Intervention Study in 2007 68 supported the growing body of evidence that long-chain omega-3s reduce the risk of heart disease, especially in people with a history of coronary artery disease. In this study, 18,645 people with hypercholesterolemia (total cholesterol of at least 251 mg/dL) with or without coronary artery disease received either 1.8 g/day EPA plus a statin or a statin only. After a mean of 4.6 years, the EPA group had 19% fewer major coronary events than the control group. The EPA group also experienced a significant reduction in rates of unstable angina and nonfatal coronary events but not in rates of sudden cardiac death or coronary death in comparison with the control group 68.
In an analysis of the primary prevention subgroup from this study (participants with no history of coronary artery disease), EPA supplementation had no significant effects on any outcome. However, for the secondary prevention subgroup (those with a history of coronary artery disease), the EPA group had a 28% reduction in the rate of unstable angina and a 19% reduction in that of major coronary events. A separate analysis of data from this study found that the EPA supplementation did not affect total stroke incidence but did reduce the risk of recurrent stroke by 20% in patients who had previously experienced a stroke 69.
Several subsequent clinical trials, however, had largely null findings 70, 71, 72. For example, the 2012 Outcome Reduction with an Initial Glargine Intervention (ORIGIN) trial included 12,536 patients who had diabetes or a high risk of diabetes and a high risk of cardiovascular events. Supplementation with 1 g/day omega-3s (375 mg DHA and 465 mg EPA) for about 6 years significantly lowered triglyceride levels but had no effect on risk of myocardial infarction, stroke, or death from cardiovascular causes in comparison with placebo 71. Similarly, in the 2010 Alpha Omega Trial, low-dose EPA and DHA supplementation (150 mg DHA and 226 mg EPA daily, supplied in a margarine) for 40 months also failed to reduce the rate of major cardiovascular events in comparison with placebo among 4,837 older men and women who had previously experienced a myocardial infarction and were receiving antihypertensive, antithrombotic, and/or lipid-lowering medications 72.
In recent clinical trials, scientists gained additional insight into the effects of omega-3s for the primary prevention of cardiovascular disease, including in patients with diabetes. From two 2018 trials: VITamin D and OmegA-3 TriaL (VITAL) 73 and A Study of Cardiovascular Events in Diabetes (ASCEND) 74. Both trials compared the same 1 g/day omega-3 formulation (460 mg EPA and 380 mg DHA) with placebo, but in different populations. VITAL included 25,871 men aged 50 and older and women aged 55 and older with no previous heart attacks, strokes, or cancer, whereas ASCEND included 15,480 adults aged 40 or older with diabetes but no evidence of cardiovascular disease. VITAL also tested the omega-3 supplement with and without 2,000 IU/day vitamin D.
In VITAL trial, the omega-3 supplement did not significantly reduce the rate of major cardiovascular events combined (myocardial infarction, stroke, and cardiovascular mortality) after a median of 5.3 years 73. However, participants taking the omega-3 supplement did experience a statistically significant 28% reduction in total myocardial infarction rates (including a 77% reduction among African Americans and a 40% reduction among those who consumed less than 1.5 servings of fish per week). Supplement users also had significant reductions in rates of fatal myocardial infarction, total coronary heart disease, and percutaneous coronary intervention (a procedure that widens blocked or narrowed coronary arteries). No significant reductions in stroke or death rates from cardiovascular causes were observed.
ASCEND trial had similar findings 74. After a mean follow-up of 7.4 years, the omega-3 supplement did not significantly affect the risk of a serious vascular event (composite of nonfatal myocardial infarction or stroke, transient ischemic attack, and cardiovascular death, excluding intracranial hemorrhage) or revascularization. However, omega-3 supplementation did significantly reduce the risk of cardiovascular death by 19% in comparison with placebo.
The 2019 Reduction of Cardiovascular Events with Icosapent Ethyl-Intervention Trial (REDUCE-IT) found significant cardiovascular disease benefits with Vascepa, a high-dose, prescription form of omega-3s containing EPA in the form of icosapent ethyl (IPE), an ethyl ester 75. REDUCE-IT trial included 8,179 participants with cardiovascular disease aged 45 years or older or with diabetes and at least one other risk factor aged 50 years or older. All participants had a fasting triglyceride level of 135 to 499 mg/dL even though they were receiving statin therapy, and an LDL cholesterol level of 41 to 100 mg/dL. Patients received either 4 g/day IPE or placebo for a median of 4.9 years. Icosapent ethyl (IPE) significantly reduced rates of cardiovascular events (a composite of cardiovascular death, nonfatal myocardial infarction, nonfatal stroke, coronary revascularization, and unstable angina) by 25%. Icosapent ethyl (IPE) also significantly reduced rates of other outcomes, including cardiovascular death by 20%, fatal or nonfatal stroke by 28%, and fatal or nonfatal myocardial infarction by 31%.
Possible reasons for conflicting findings
Dose probably plays a major role in the ability of omega-3 supplementation to confer significant benefits 76. The REDUCE-IT trial findings suggest that a high daily dose of icosapent ethyl (IPE), 4 g, is an effective adjunct to statin therapy in people with cardiovascular disease or a high risk of cardiovascular disease 73. The daily dose of 1 g used in many studies of omega-3 dietary supplements might affect some cardiovascular disease pathways 76 but has had no significant effect on the primary outcomes in several trials 74. Other factors, including the omega-3 form, study population, background dietary omega-3 intakes, and use of statins and other cardioprotective therapies might also explain the conflicting findings among studies 77.
Furthermore, the effects of long-chain omega-3s are not uniform across cardiovascular disease outcomes. Therefore, use of primary composite endpoints that combine multiple outcomes might dilute significant effects on individual components of those endpoints 77. This dilution occurred, for example, in both VITAL 73 and ASCEND 74.
A 2019 systematic review and meta-analysis of 13 trials included ASCEND, VITAL, and REDUCE-IT and a total of 127,477 participants 77. Omega-3 doses ranged from 0.376 to 4 g/day, and the mean treatment duration was 5 years. The authors concluded that long-chain omega-3 supplementation reduces the risk of myocardial infarction, coronary heart disease death, total coronary heart disease, cardiovascular disease death, and total cardiovascular disease, and the effects appear to be dose related. However, the findings showed no significant associations for risk of fatal and nonfatal stroke. The authors noted that REDUCE-IT reduced risk of stroke significantly 75, suggesting that a higher dose of omega-3s (4 g/day) might be needed to affect this outcome.
A 2020 Cochrane review of 86 randomized controlled trials published between 1968 and 2019 found that 0.5 g/day to more than 5 g/day omega-3s for 12 to 88 months in a total of 162,796 participants reduced serum triglyceride levels by about 15% and slightly decreased rates of cardiovascular mortality and coronary heart disease events 78. However, the supplements did not affect all-cause mortality, cardiovascular events, stroke, or arrhythmia. The authors of several earlier meta-analyses and systematic reviews, as well as a 2016 report from the Agency for Healthcare Research and Quality (AHRQ) 79, concluded that omega-3 supplements do not appear to significantly reduce the risk of most cardiovascular events 80. Many of these analyses, however, but not all 79, 81, did find that omega-3s reduce the risk of cardiac death.
Recommendations from the American Heart Association (AHA) and the Dietary Guidelines for Americans
Between 2017 and 2019, the American Heart Association (AHA) released three science advisories on omega-3s 16, 17, 18. All three advisories recommend one to two servings of seafood per week to reduce the risk of congestive heart failure, coronary heart disease, ischemic stroke, and sudden cardiac death, especially when the seafood replaces less healthy foods 18. For people with existing coronary heart disease, such as a recent myocardial infarction, the AHA recommends approximately 1 g/day EPA plus DHA, preferably from oily fish; however, supplements could also be considered under the direction of a physician 17. The American Heart Association does not recommend omega-3 supplements for people who do not have a high cardiovascular disease risk.
To manage high triglyceride levels, the American Heart Association concludes that 4 g/day prescription omega-3s (containing EPA plus DHA or EPA only) lower triglyceride levels when used alone or as adjuncts to other lipid-lowering medications 16. Although this finding pertains to high-dose prescription omega-3s, an earlier analysis of 58 trials also revealed a dose-response relationship between lower-dose dietary and supplemental omega-3 intakes and triglyceride levels 82. Each 1 g/day of omega-3 increase reduced triglyceride levels by 5.9 mg/dL, and the effect was stronger in people with higher baseline triglyceride levels.
The 2020–2025 Dietary Guidelines for Americans states that strong evidence from mostly prospective cohort studies but also some randomized controlled trials shows that eating patterns that include seafood are associated with a reduced risk of cardiovascular disease 13. In addition, consuming about 8 ounces (less for children) per week of a variety of seafood that provides about 250 mg per day EPA and DHA is associated with fewer cardiac deaths in both healthy individuals and those with preexisting cardiovascular disease. Those who are pregnant or breastfeeding consume between 8 and 12 ounces per week of a variety of seafood from choices that are lower in mercury. These women should not consume certain types of fish, such as king mackerel, shark, swordfish, and tilefish that are high in methyl mercury, and they should limit the amount of white (albacore) tuna they consume to 6 ounces a week 14. The American Academy of Pediatrics has similar advice for breastfeeding women, recommending intakes of 200–300 mg DHA per day by consuming one to two servings of fish per week to guarantee a sufficient amount of DHA in breast milk 15.
Infant health and neurodevelopment
During pregnancy and breastfeeding, eating 8 to 12 ounces per week of fish and other seafood may improve your baby’s health. However, it is important to choose fish that are higher in EPA and DHA and lower in mercury. Examples are salmon, herring, sardines, and trout. It is not clear whether taking dietary supplements containing EPA and DHA during pregnancy or breastfeeding affects a baby’s health or development. However, some studies show that taking these supplements may slightly increase a baby’s weight at birth and the length of time the baby is in the womb, both of which may be beneficial. Breast milk contains DHA. Most commercial infant formulas also contain DHA. However, the authors of a paper published by the American Academy of Family Physicians 83 and of two Cochrane reviews (one on full-term infants and one on preterm infants) 84, 85 have concluded that the evidence is insufficient to recommend the use of infant formulas that are supplemented with these fatty acids.
Numerous studies have examined the effects of maternal seafood and omega-3 intakes on infant birth weight, length of gestation, visual and cognitive development, and other infant health outcomes. High concentrations of DHA are present in the cellular membranes of the brain and retina 8 and DHA is important for fetal growth and development. The accumulation of DHA in the retina is complete by birth, whereas accumulation in the brain continues throughout the first 2 years after birth.
Observational studies indicate that maternal consumption, during pregnancy and breastfeeding, of at least 8 ounces per week of seafood that contains DHA is associated with better infant health outcomes 13. For example, in a prospective cohort study of 341 mother–child pairs in the United States, maternal fish consumption more than twice per week compared to no weekly consumption was associated with improved visual motor skills in their children at age 3 after adjustment for covariates such as maternal age, education, maternal smoking and alcohol use during pregnancy, paternal education, and fetal growth 86. In another observational cohort study in the United Kingdom in 11,875 pregnant women who reported seafood intakes ranging from none to more than 340 g (about 12 ounces) per week, lower consumption of seafood during pregnancy was associated with an increased risk of suboptimal communication skills in the offspring at ages 6 and 18 months and suboptimal verbal IQ and prosocial behavior at age 7–8 years 2. It is not possible to establish causality, however, because all of these studies were observational.
Seafood contains varying levels of methyl mercury 14. However, results from numerous studies, including a systematic review of the literature on maternal fish intake and subsequent neurodevelopmental outcomes, show that the health benefits of consuming moderate amounts of seafood during the prenatal period outweigh the risks 87.
Several randomized controlled trials have examined whether supplementation with fish oil, EPA, and/or DHA during pregnancy and early infancy is beneficial for infant health and neurodevelopment. One of these trials examined the effects of fish oil supplementation in 2,399 pregnant women on the subsequent clinical outcomes and neurodevelopment of their children 88. Pregnant women received daily supplements of either fish oil (providing 800 mg DHA and 100 mg EPA) or placebo from less than 21 weeks’ gestation until the birth of their child. Compared to the placebo group, children of mothers who received fish oil were heavier at birth and less likely to be born very preterm (less than 34 weeks’ gestation). However, assessments of 726 of the children (all 96 preterm children and 630 randomly selected full-term children) found no differences between groups in mean cognitive composite scores or mean language composite scores at age 18 months. A follow-up study of the children at age 4 years found no differences between groups in general conceptual ability score or other assessments of cognition, language, and executive functioning 89. Another study found no benefits on visual function at age 7 years when very preterm infants (less than 33 weeks’ gestation) consumed human milk with a higher DHA concentration than normal (lactating mothers took 900 mg/day DHA supplements) for the first months of life until full term 90. In a clinical trial in 420 healthy full-term infants, those who received either DHA-enriched fish oil (250 mg DHA and 60 mg EPA) or placebo daily from birth to 6 months had similar scores on neurodevelopment assessments at 18 months 91. However, infants receiving fish oil had significantly better performance on language assessments, indicating some benefit for early communication development.
The authors of a systematic review and meta-analysis of 11 randomized controlled trials concluded that the evidence neither supports nor refutes the benefits of omega-3 supplementation during pregnancy for cognitive or visual development in infants 92. Another systematic review and meta-analysis that included two randomized controlled trials in women with a previous preterm birth found no significant differences in rates of recurrent preterm birth between women who took omega-3 supplements during pregnancy and those who did not 92. Omega-3 supplementation did, however, increase latency (time from randomization to birth) by about 2 days and mean birth weight by about 103 g.
In 2016, AHRQ 93 published a review on the effects of omega-3 fatty acids on child and maternal health. This comprehensive report evaluated the findings from 95 randomized controlled trials and 48 prospective longitudinal studies and nested case-control studies. Most studies examined the effects of fish oil supplements or other DHA and EPA combinations in pregnant or breastfeeding women or of infant formula fortified with DHA plus arachidonic acid, an omega-6. The authors concluded that, except for small beneficial effects on infant birth weight and length of gestation, omega-3 supplementation or fortification has no consistent effects on infant health outcomes 93.
Cancer prevention
Some studies suggest that people who get more omega-3s from foods and dietary supplements may have a lower risk of breast cancer and perhaps colorectal cancer. But a large clinical trial found that omega-3 supplements did not reduce the overall risk of cancer, or the risk of breast, prostate, or colorectal cancers. Overall, data from observational studies show no consistent relationship between omega-3s and overall cancer risk. Additional randomized clinical trials in progress will help clarify whether omega-3s affect cancer risk.
Researchers have hypothesized that higher intakes of omega-3s from either foods or supplements might reduce the risk of cancer due to their anti-inflammatory effects and potential to inhibit cell growth factors 94. Results from observational studies however, have been inconsistent and vary by cancer site and other factors, including gender and genetic risk. For example, some studies have shown associations between higher intakes and/or blood levels of omega-3s and a decreased risk of certain cancers, including breast 95 and colorectal cancers 96. Other studies have found no associations between omega-3s and cancer risk, and some have even found associations in the opposite direction, suggesting that omega-3s might increase the risk of certain cancers such as prostate cancer 97. The first large-scale clinical trial to examine the effects of omega-3s on the primary prevention of cancer in the general population was the newly published VITAL trial 73. This clinical trial examined the effects of omega-3 fish oil supplementation (1 g/day containing 460 mg EPA and 380 mg DHA) with or without 2,000 IU/day vitamin D for a median of 5.3 years 73. The study included 25,871 men aged 50 and older and women aged 55 and older with no previous cancer, heart attacks, or strokes. Compared with placebo, the omega-3 supplement had no significant effect on cancer incidence, cancer mortality rates, or the development of breast, prostate, or colorectal cancers.
Breast cancer
Evidence from several observational studies suggests that higher intakes of omega-3s are associated with a lower risk of breast cancer, but clinical trials are needed to confirm this finding. In the prospective Singapore Chinese Health Study of 35,298 women aged 45–74 years, those in the top three quartiles of dietary omega-3 intake had a 26% lower risk of breast cancer after an average of 5.3 years of follow-up than those in the lowest quartile 98. Similarly, among 35,016 female participants aged 50–76 years in the VITamins And Lifestyle (VITAL) Cohort, those who reported current use of fish-oil supplements had a 32% lower risk of breast cancer after a mean of 6 years than those who did not take fish oil 99.
According to a systematic review of three case-control studies and five prospective studies published in 2007–2011, evidence is increasing that higher intakes of dietary and supplemental omega-3s are associated with a lower risk of breast cancer 100. Similarly, the authors of a meta-analysis of data from 21 prospective cohort studies concluded that women with the highest dietary intakes and/or tissue levels of omega-3s had a 14% lower risk of breast cancer than those with the lowest intakes and tissue levels 95. These authors also found a dose-response relationship between higher intakes of combined omega-3s and reduced breast cancer risk. Intakes of ALA and of fish, however, had no association with differences in breast cancer risk. This finding, which could be due to varying levels of omega-3s in different fish species, warrants further investigation.
Colorectal cancer
Limited evidence from observational studies suggests that greater consumption of fish and omega-3s is associated with a reduced risk of colorectal cancer 100. The authors of a meta-analysis of 19 prospective cohort studies found no significant association between fish intake and risk of colorectal cancer overall. However, a stratified analysis showed that for participants with the highest fish consumption (those who ate fish at least seven times more often per month than those with the lowest fish consumption), the risk of colorectal cancer was 22% lower than that for the lowest fish consumers 101. Results from a more recent systematic review and meta-analysis of 22 prospective cohort studies and 19 case-control studies indicate that fish consumption is inversely associated with colorectal cancer risk. In this analysis, 21 of the studies distinguished between colon cancer and rectal cancer. The risk of rectal cancer was 21% lower for participants with the highest fish intakes (as much as one serving/day) compared to those with the lowest fish intakes (as little as none), but fish consumption had no significant association with risk of colon cancer alone 96.
Results from the VITamins And Lifestyle (VITAL) Cohort study suggest that associations between fish or omega-3 intakes and colorectal cancer risk might vary by such factors as gender and genetic risk. In this study, researchers evaluated associations between colorectal cancer risk and EPA/DHA intakes from fatty fish (salmon and fresh tuna) and fish oil supplements in 68,109 Washington residents aged 50–76 102. The amount of fatty fish consumed ranged from none to 0.8 servings per week or more. Overall, EPA and DHA intakes (from either diet or supplements) and fatty fish consumption were not associated with colorectal cancer risk, but associations varied by genetic characteristics (certain inherited genetic mutations are associated with an increased risk of colorectal cancer). For individuals in the lowest two tertiles of genetic risk, higher fatty fish consumption and higher total EPA and DHA intakes were inversely associated with colorectal cancer risk. For individuals in the highest tertile of genetic risk, higher total EPA and DHA intakes were positively associated with colorectal cancer risk. Risk also varied by gender. Among men, use of fish oil supplements reduced colorectal cancer risk by an average of 34% or more depending on the frequency and duration of use, but this effect did not occur among women. Additional research is needed to clarify possible associations between fish and omega-3 intakes and colorectal cancer risk.
Prostate cancer
Several prospective and case-control studies have investigated associations between either blood levels or intakes of omega-3s and risk of low-grade or high-grade prostate cancer. Results from these studies have been inconsistent.
A few case-control and case-cohort studies have found positive associations between blood levels of long-chain omega-3s and prostate cancer risk (particularly high-grade disease that is more advanced and more likely to spread than low-grade cancer), suggesting that omega-3s might increase prostate cancer risk. In a nested case-control analysis of men aged 55–84 years participating in the Prostate Cancer Prevention Trial, serum phospholipid levels of DHA were positively associated with risk of high-grade, but not low-grade, prostate cancer 103. Serum EPA levels, however, were not associated with risk of either grade of the disease.
Similarly, results from a case-cohort study within the Selenium and Vitamin E Cancer Prevention (SELECT) trial showed that men in the highest quartile of plasma phospholipid long-chain omega-3s had a 44% higher risk of low-grade prostate cancer and a 71% higher risk of high-grade prostate cancer than those in the lowest quartile 104. An analysis of data from the European Prospective Investigation into Cancer and Nutrition cohort also found a higher prostate cancer risk in men with higher plasma levels of omega-3s 105. Among Whites participating in the Multiethnic Cohort Study, higher levels of omega-3s in erythrocyte membranes and higher ratios of omega-3s to omega-6s were both associated with an increased risk of prostate cancer. However, the results showed no associations, even with advanced or high-grade disease, for other ethnic groups or for the population as a whole 106.
Results from other observational studies using dietary intake data suggest that higher intakes of fish and/or omega-3s reduce prostate cancer risk. Both fish and omega-3 consumption were associated with a lower risk of fatal prostate cancer in a cohort of 293,464 men participating in the National Institutes of Health (NIH)-AARP Diet and Health Study 107. In the Health Professionals Follow-up Study, a prospective cohort of over 47,000 men aged 40–75 years, those who consumed fish more than three times per week had a lower risk of metastatic prostate cancer than those who consumed fish less than twice per month 108. However, men who used fish oil supplements did not have a decreased risk of prostate cancer.
A number of systematic reviews and meta-analyses of prospective studies of the effects of fish intakes, omega-3 intakes, and omega-3 blood levels on prostate cancer risk have had inconsistent findings as well. For example, circulating levels of EPA, but not DHA, were positively associated with prostate cancer risk in a meta-analysis of 5,098 men with prostate cancer and 6,649 men without prostate cancer from seven studies 109. Another meta-analysis of 12 studies that included 4,516 men with prostate cancer and 5,728 men without prostate cancer found that high serum levels of these LC omega-3s were positively associated with high-grade disease 110. In other analyses, dietary intakes of long-chain omega-3s had no effect on prostate cancer risk 111, whereas fish consumption decreased prostate cancer mortality but had no effect on prostate cancer incidence 112. A 2015 meta-analysis found no significant associations between dietary intakes or blood levels of long-chain omega-3s and total prostate cancer risk 113. The authors noted that most dietary-intake studies included in their meta-analysis found inverse associations, whereas biomarker studies of blood levels of these fatty acids found positive associations.
Overall, the evidence to date shows no consistent relationships between prostate cancer risk or mortality and omega-3 intakes or blood levels.
Other cancers
Evidence is limited for a role of omega-3s in the prevention of cancers at other sites. For example, evidence is insufficient to determine whether omega-3s affect the risk of skin cancers, including basal-cell carcinoma, squamous-cell carcinoma, and melanoma 114. Findings from the Australian Ovarian Cancer Study suggest that there is no association between total or individual omega-3 intakes from foods and ovarian cancer risk 115.
Associations between omega-3 intakes and endometrial cancer have been mixed. Some evidence indicates that dietary intakes of EPA and DHA may provide protection from the development of endometrial cancer 116. Other evidence indicates that they decrease risk in normal-weight women but have no effect or even increase risk in overweight or obese women 117.
A systematic review and meta-analysis of 9 prospective cohort and 10 case-control studies did not find an association between fish or long-chain omega-3 intakes and risk of pancreatic cancer 118. Similarly, systematic reviews and meta-analyses have not found significant associations between fish consumption and risk of gastric or esophageal cancers 119.
Age-related macular degeneration (AMD) is a major cause of vision loss among older adults. In most cases, severe vision loss is associated with advanced AMD, which consists of either central geographic atrophy (dry AMD, the most common form) or neovascular AMD (wet AMD) 120. Based on DHA’s presence as a structural lipid in retinal cellular membranes and the beneficial effects of EPA-derived eicosanoids on retinal inflammation, neovascularization, and cell survival, researchers have suggested that these omega-3s have cytoprotective effects in the retina that may help prevent the development or progression of AMD 9. Studies suggest that people who get higher amounts of omega-3s from the foods they eat may have a lower risk of developing AMD. But once someone has AMD, taking omega-3 supplements does not keep the disease from getting worse or slow down vision loss.
Results from observational studies suggest that people who consume higher amounts of fatty fish and/or dietary long-chain omega-3s have a lower risk of developing AMD. In the cross-sectional EUREYE study of 2,275 participants aged 65 years or older, those who ate fatty fish at least once per week had a 53% lower risk of neovascular AMD than those who consumed fatty fish less often 121. Results were similar in a study in 681 elderly male twins 122 and an analysis of 38,022 healthy female health professionals 120. In the latter study, women in the highest tertiles of dietary DHA plus EPA intake (median of 330 mg/day) had a 38% lower risk of developing AMD during an average of 10 years of follow-up than those in those in the lowest tertile (median intake of 80 mg/day). Higher serum and erythrocyte membrane levels of EPA (but not DHA) have also been associated with a lower risk of neovascular AMD 123.
In the Age-Related Eye Disease (AREDS) study, a dietary supplement formulation containing 15 mg beta-carotene, 400 IU vitamin E, 500 mg vitamin C, 80 mg zinc, and 2 mg copper reduced the risk of advanced AMD in people with intermediate AMD or advanced AMD in one eye 124. Data from a nested cohort study within the AREDS population indicated that participants who reported the highest omega-3 intakes were about 30% less likely to develop central geographic atrophy and neovascular AMD than other participants 125.
These findings, combined with other epidemiological evidence, formed the basis for the Age-Related Eye Disease Study 2 (AREDS 2) clinical trial that examined whether adding 350 mg DHA and 650 mg EPA to the AREDS formulation further reduced the risk of progression to advanced AMD 126. The results showed that EPA and DHA did not provide any additional benefits after a median follow-up of 5 years. These findings are in line with those from a Cochrane review 127 that included the results from AREDS2 and the Nutritional AMD Treatment 2 study 128, a 3-year randomized clinical trial of omega-3 supplements (840 mg/day DHA and 270 mg/day EPA) in patients with early age-related maculopathy and neovascular AMD. The Cochrane review authors concluded that omega-3 supplementation for up to 5 years in people with AMD does not reduce the risk of progression to advanced AMD or of moderate to severe vision loss.
Dry eye disease
Dry eye disease occurs when tears don’t provide enough moisture, causing eye discomfort and vision problems. About 14% of adults in the United States have dry eye disease, a chronic condition in which decreased tear volume and quality leads to ocular surface inflammation and damage, causing discomfort and visual impairment 129. Older women, in particular, have a higher risk of dry eye disease than other groups, possibly because of hormonal changes that affect the tear-producing glands 130. Researchers hypothesize that omega 3s—particularly EPA and DHA—might reduce the risk of dry eye disease and relieve its symptoms because of their anti-inflammatory activity, and many patients take them as adjunctive treatments to artificial tears and other medications. But a large, recent study found that the symptoms of people with dry eye disease who took fish oil supplements of 2,000 mg EPA plus 1,000 mg DHA daily for 1 year did not improve any more than those who took a placebo (a dummy pill). More research on the effects of omega-3s on dry eye disease is needed.
Some, but not all, observational studies show inverse associations between self-reported dietary consumption of omega-3s and risk of dry eye disease. For example, in a cross-sectional study of 32,470 women aged 45–84 participating in the Women’s Health Study, those in the highest quintile of total dietary omega-3 intake (mean of 1,990 mg/day) had a 17% lower risk of dry eye disease than those in the lowest quintile (mean intake of 920 mg/day) 131. The study found a similar association for DHA—women in the highest versus the lowest quintiles of DHA intake had a 12% lower risk of dry eye disease; however, the results showed no significant associations for EPA. But in another cross-sectional study of 322 postmenopausal women, total dietary omega-3 intakes were not correlated with the prevalence of dry eye disease 130.
Results from clinical trials using omega-3 supplementation, primarily EPA and DHA, have had mixed results in reducing the symptoms and signs of dry eye disease. Furthermore, there is no consensus on the optimal dose, composition, or length of omega-3 treatment for this condition 132.
The studies that have found beneficial effects from omega-3 supplementation for symptoms and signs of dry eye disease include one showing that daily supplementation with 1,000 mg omega-3s (650 mg EPA plus 350 mg DHA) for 3 months in 518 men and women (mean age about 40 years) living in northern India reduced symptoms and some signs of dry eye disease compared with placebo 133. In another clinical trial of 105 men and women, daily treatment with supplements containing 2,240 mg omega-3s (1,680 mg EPA and 560 mg DHA as re-esterified triglycerides) for 12 weeks also reduced symptoms of dry eye disease compared with placebo 134. In addition, the supplements increased tear break-up time and decreased tear osmolarity (which would be likely to reduce ocular surface damage).
However, another large, randomized, double-blind clinical trial conducted in the United States found that EPA and DHA from fish oil supplements are no better than placebo at relieving symptoms or signs of dry eye disease 129. This 12-month trial included 535 participants (about 81% female) aged 18 years or older (mean age about 58 years) with at least a 6-month history of moderate to severe dry eye disease. Among them, 349 participants received daily supplements of 3,000 mg omega-3s (2,000 mg EPA plus 1,000 mg DHA), and 186 received a placebo containing 5,000 mg olive oil. Participants could continue taking medications for dry eyes, including artificial tears and prescription anti-inflammatory eye drops, as well as omega-3 supplements as long as the total dose of EPA plus DHA was less than 1,200 mg per day. At the end of the study, symptoms were less severe than at baseline in both groups, but the results showed no significant differences between groups. Groups also showed no significant differences compared with baseline in signs of dry eye disease, including conjunctive and cornea integrity as well as tear volume and quality.
Overall, the evidence to date shows no consistent relationship between omega-3s and dry eye disease. More research is warranted to fully understand whether increased intakes of dietary or supplemental omega-3s help reduce the risk of dry eye disease and whether they are beneficial as an adjunct treatment.
Alzheimer’s disease, dementia, and cognitive function
Some, but not all, observational studies suggest that diets high in omega-3s are associated with a reduced risk of cognitive decline, Alzheimer’s disease, and dementia 135. Because DHA is an essential component of cellular membrane phospholipids in the brain, researchers hypothesize that omega-3s might protect cognitive function by helping to maintain neuronal function and cell- membrane integrity within the brain 135. This hypothesis is supported by findings from case-control studies indicating that patients with Alzheimer’s disease have lower serum levels of DHA than cognitively healthy people 136. Lower serum DHA levels are also associated with more cerebral amyloidosis (build-up of protein deposits called amyloids) in healthy older adults, whereas higher DHA is correlated with preservation of brain volume 137.
Several observational studies have examined the effects of fish, EPA, and/or DHA intakes on cognitive function in healthy older adults. In a prospective cohort study involving 210 healthy men aged 70–89, fish consumption was associated with less cognitive decline at follow-up 5 years later 138. In addition, a dose-response relationship was observed between tertiles of dietary EPA plus DHA intake and subsequent 5-year cognitive decline. Similarly, in the Rotterdam Study, a population-based prospective study of people aged 55 or older who were free from dementia at baseline, higher fish consumption among 5,386 study participants was associated with a 60% lower risk of dementia and a 70% lower risk of Alzheimer’s disease over an average of 2.1 years 139. Subsequent follow-up 6 years after baseline, however, found no associations between omega-3 intakes and incidence of dementia or Alzheimer’s disease 140. The authors suggest that the discrepancy might be explained by the short follow-up period in the first analysis and the small number of patients who developed dementia. A higher omega-3 index was associated with a greater hippocampal volume in the Women’s Health Initiative Memory Study 141 and with a larger brain volume and improved cognitive test scores in the Framingham Offspring cohort 142. A 2016 dose-response meta- analysis of 21 cohort studies found that increased intakes of fish and dietary DHA were both inversely associated with the risks of dementia and Alzheimer’s disease 143. Specifically, a 100 mg/day incremental increase in DHA intake was associated with a 14% lower risk of dementia and a 37% lower risk of Alzheimer’s disease.
Results from clinical trials, however, suggest that omega-3 supplementation does not affect cognitive function in older adults who have no cognitive impairment. In a trial in the United Kingdom, 748 cognitively healthy adults aged 70–79 years received either 500 mg DHA and 200 mg EPA or placebo daily for 24 months 144. Cognitive function did not differ significantly between the two groups, although cognitive function did not decline in either group. In the AREDS2 study, treatment with 350 mg DHA and 650 mg EPA for 5 years did not have a significant effect on cognitive function in 3,501 older adults (mean age 72.7 years) with AMD 136.
Clinical trial results also suggest that omega-3 supplementation does not benefit patients with Alzheimer’s disease, although it might help patients with mild cognitive impairment. For example, daily supplementation with 2 g DHA for 18 months did not slow the rate of cognitive decline compared to placebo in 295 participants (mean age 76 years) with mild to moderate Alzheimer’s disease 145. In the OmegaAD trial, daily supplementation with 1,700 mg DHA and 600 mg EPA for 6 months in 174 older adults with mild to moderate Alzheimer’s disease also failed to slow down the rate of cognitive decline compared to placebo 146. However, a subgroup of patients with very mild impairment experienced a significant reduction in the rate of cognitive decline. In a small trial in Malaysia, fish oil supplementation (1,290 mg DHA and 450 mg EPA daily) for 12 months improved memory—particularly short-term, working, and verbal memory—and delayed recall compared to placebo in 35 older adults with mild cognitive impairment 147.
Several systematic reviews and meta-analyses, including a Cochrane review, have assessed the effects of omega-3 supplementation on cognitive function and dementia in healthy older adults and those with Alzheimer’s disease or cognitive impairment 148, 149. Overall, the findings indicate that omega-3 supplementation does not affect cognitive function in healthy older adults or in people with Alzheimer’s disease compared to placebo. For people with mild cognitive impairment, omega-3s may improve certain aspects of cognitive function, including attention, processing speed, and immediate recall 150.
However, these findings need to be confirmed in additional clinical trials.
Rheumatoid arthritis
Rheumatoid arthritis (RA) is an autoimmune disease characterized by chronic inflammation of the joints. Rheumatoid arthritis symptoms include pain, swelling, stiffness, and functional impairments. Rheumatoid arthritis is typically treated with nonsteroidal antiinflammatory drugs (NSAIDs), corticosteroids, and disease-modifying antirheumatic drugs. Due to their antiinflammatory effects, some scientists hypothesize that omega-3s reduce some of the symptoms of rheumatoid arthritis and patients’ reliance on NSAIDs and corticosteroids. Some clinical trials have shown that taking omega-3 supplements may help manage rheumatoid arthritis when taken together with standard rheumatoid arthritis medications and other treatments. For example, people with rheumatoid arthritis who take omega-3 supplements may need less pain-relief medication, but it is not clear if the supplements reduce joint pain, swelling, or morning stiffness.
Several clinical trials, many conducted in the 1990s, have examined the use of omega-3 supplementation in patients with rheumatoid arthritis. These trials have generally shown that omega-3 supplements reduce patients’ use of antiinflammatory drugs and corticosteroids, but that they do not have consistent effects on painful and/or tender joints, joint swelling, or morning stiffness 151, 152. For example, fish oil supplementation significantly reduced NSAID use in a controlled trial in Sweden 153. In this study, 43 patients with rheumatoid arthritis received either 10 g/day fish oil (containing 1.8 g EPA and 1.2 g DHA) or placebo along with their usual rheumatoid arthritis medications. NSAID use decreased in the treatment group at 3 and 6 months, and global arthritic activity assessed by physicians improved relative to placebo at 3 months. However, patient assessments of pain, morning stiffness, and functional capacity did not differ between groups. In a 2013 clinical trial in South Korea, 81 patients with rheumatoid arthritis received either omega-3s (2.1 g EPA and 1.2 g DHA) or a sunflower oil placebo daily for 16 weeks 154. Patients were allowed to continue taking NSAIDs, glucocorticoids, and/or antirheumatic drugs throughout the study. Compared to placebo, omega-3 supplementation had no significant effects on clinical symptoms of rheumatoid arthritis, including pain and morning stiffness. In post-hoc analysis, the researchers found that the supplements reduced the amount of NSAIDs needed, but only in patients weighing more than 55 kg. In a similar study in Denmark, 51 patients received either omega-3s (2.0 g EPA and 1.2 g DHA from fish oil) or placebo daily for 12 weeks, and they continued taking rheumatoid arthritis medications 155. Compared to placebo, morning stiffness, joint tenderness, and visual pain score decreased significantly in the treatment group. However, there were no significant differences between groups in grip strength, daily activity score, or joint swelling. The amounts of NSAIDs, aspirin, and acetaminophen that patients needed did not change in either group.
Reviews and meta-analyses of studies that assessed whether fish oil and omega-3s are beneficial for rheumatoid arthritis have had inconsistent findings. Some suggest that they do not significantly affect the clinical symptoms of rheumatoid arthritis but do reduce the amounts of NSAIDs and corticosteroids that patients need 156, 151. Others indicate that omega-3s reduce joint swelling and pain, morning stiffness, and number of painful joints in addition to reducing NSAID use 152, 157. Some researchers suggest that differences in findings could be due in part to whether patient-determined use of NSAIDs is considered a measure of pain 158.
Findings to date suggest that omega-3s may be helpful as an adjunctive treatment to pharmacotherapy for reducing the symptoms of rheumatoid arthritis 158. However, more research is needed to confirm this finding.
Depression
A 2016 meta-analysis of 26 studies found a 17% lower risk of depression with higher fish intake 159. However, a 2015 Cochrane review of 26 studies found insufficient evidence to determine whether omega-3s (1,000 to 6,600 mg/day EPA, DHA, and/or other omega-3s) are beneficial for major depressive disorder in adults 160. The authors did find a small-to-modest beneficial effect on depressive symptoms, but they concluded that this effect was not clinically significant.
Inflammatory bowel disease
The authors of a systematic review of 19 randomized controlled trials concluded that the available evidence does not support the use of omega-3 supplements to treat active or inactive inflammatory bowel disease (Crohn’s disease & ulcerative colitis) 161. Similarly, the authors of a Cochrane review concluded that, based on the evidence from two large, high-quality studies, omega-3 supplements are probably not effective for maintaining remission in people who have Crohn’s disease 162. No evidence was found that supports the use of omega 3 fatty acids for maintenance of remission in ulcerative colitis 163.
ADHD
A systematic review and meta-analysis of 10 studies in children with ADHD or related neurodevelopmental disorders, such as developmental coordination disorder, found no improvements with omega-3 supplementation on measures of emotional lability, oppositional behavior, conduct problems, or aggression 164. However, in subgroup analyses of only the higher-quality studies and those with strict inclusion criteria, omega-3 supplementation (60 to 1,296 mg/day EPA and/or DHA) did significantly improve parent-rated emotional lability and oppositional behavior.
Childhood allergies
A systematic review and meta-analysis of 10 prospective cohort studies and 5 randomized clinical trials on omega-3 intakes during pregnancy and outcomes of childhood allergic disease (eczema, rhino-conjunctivitis, and asthma) found inconsistent results 165. Although the authors could not draw firm conclusions due to the heterogeneity of the studies and their results, they concluded that the overall findings were “suggestive” of a protective association between higher maternal intakes of omega-3s or fish and incidence of allergic disease symptoms in the offspring. The authors of a Cochrane review that included eight LC omega-3 supplementation trials concluded that there is limited evidence to support the use of omega-3 supplements by women during pregnancy and/or lactation for reducing the risk of allergic disease in their children 166.
Cystic fibrosis
A Cochrane review of four studies of cystic fibrosis found that omega-3 supplements (300 to 5,400 mg/day EPA and/or DHA) might improve lung function and increase blood levels of essential fatty acids in people with cystic fibrosis 167. However, the authors concluded that there is not enough evidence to recommend routine use of omega-3 supplements by people with cystic fibrosis.
Type 2 diabetes mellitus
People with type 2 diabetes are known to be at increased risk of cardiovascular disease (such as heart attack or stroke). Type 2 diabetes mellitus is the fourth leading cause of death in developed countries with a two fold excess mortality and a two to four fold increased risk of coronary heart disease and stroke. The typical dyslipidemia (abnormality in blood lipids) associated with type 2 diabetes is a combination of hypertriglyceridemia (high levels of fats (triglycerides) in the blood), low levels of HDL (high density lipoprotein) cholesterol and abnormal LDL (low density lipoprotein) composition. Low levels of HDL cholesterol and high levels of LDL cholesterol are associated with an increased risk of cardiovascular disease, while the raised levels of triglycerides are less clearly linked to an increased risk of cardiovascular disease. Several pharmacologic approaches have been used to treat diabetic dyslipidemia and standard dietary approaches focus on restriction of saturated fat and limitation of simple carbohydrate and alcohol intake. In the late 1980s, several investigators reported on the use of dietary supplementation with fish oil as a means of treating diabetic dyslipidemia. Dietary fats and oils from different sources differ considerably in their fatty acid composition. Animal fat is rich in saturated fatty acids, vegetable and marine oils are rich in polyunsaturated fatty acids. Most fish oils are of the so-called omega-3 variety (omega-3 polyunsaturated fatty acids (PUFAs)).
The review (source 168) shows that although some types of fat in the blood are reduced through omega-3 supplementation, others including LDL cholesterol (which may promote heart disease) were increased. Control of blood sugar levels was not affected by the treatment. There were no other adverse effects of the interventions noted. Clinical outcome trials of sufficient duration are required to establish conclusively the role of omega-3 PUFA in type 2 diabetes but our results do not suggest a major harmful effect on the balance of blood fats and confirm that it has no adverse affect on blood sugar control.
They concluded Omega-3 polyunsaturated fatty acids supplementation in type 2 diabetes lowers triglycerides and VLDL cholesterol, but may raise LDL cholesterol (although results were non-significant in subgroups) and has no statistically significant effect on glycemic control or fasting insulin. Trials with vascular events or mortality defined endpoints are needed.
Autism Spectrum Disorders
It has been suggested that difficulties associated with autism spectrum disorders may be explained in part by lack of omega-3 fatty acids, and that supplementation of these essential fatty acids may lead to improvement of symptoms. To date there is no high quality evidence that omega-3 fatty acids supplementation is effective for improving core and associated symptoms of autism spectrum disorder. Given the paucity of rigorous studies in this area, there is a need for large well-conducted randomised controlled trials that examine both high and low functioning individuals with autism spectrum disorder and that have longer follow-up periods 169.
Intermittent Claudication
Intermittent claudication is a pain in the calf due to a lack of blood needed to supply those muscles with oxygen during exercise or movement, ultimately resulting in the person to slow or stop movement. It is the most common presenting symptom for people with long-standing lower limb arterial disease resulting from narrowing of the arteries that supply the lower limbs with blood. This narrowing most commonly occurs through the process of atherosclerosis in which an artery wall thickens as a result of the accumulation of fatty materials such as cholesterol and triglycerides. People with mild lower limb arterial disease are advised to stop smoking, exercise, and take an antiplatelet agent to prevent heart attack or stroke. Medication to improve walking distance is only of limited value.
Omega-3 fatty acids appear to have little haematological benefit in people with intermittent claudication and there is no evidence of consistently improved clinical outcomes (quality of life, walking distance, ankle brachial pressure index or angiographic findings). Supplementation may also cause adverse effects such as nausea, diarrhoea and flatulence. Further research is needed to evaluate fully short- and long-term effects of omega-3 fatty acids on the most clinically relevant outcomes in people with intermittent claudication before they can be recommended for routine use 170.
Bipolar disorder
This systematic review (source 171 investigated the efficacy of omega-3 fatty acids for bipolar disorder. Five randomised controlled trials met inclusion criteria for the review. Only one trial provided data that could be analysed, investigating ethyl-EPA as an adjunctive treatment in a mixed outpatient population. Some positive benefits were found for depressive symptoms but not for mania, and no adverse events were reported. There is currently insufficient evidence on which to base any clear recommendations concerning omega-3 fatty acids for bipolar disorder. However, given the general health benefits and safety of omega-3, the preliminary evidence from this review provides a strong case for well-powered, high-quality trials in specific index populations.
Results from one study showed positive effects of omega-3 as an adjunctive treatment for depressive but not manic symptoms in bipolar disorder. These findings must be regarded with caution owing to the limited data available. There is an acute need for well-designed and executed randomised controlled trials in this field.
Omega-3 Supplements
- Do not use omega-3 supplements to replace conventional care or to postpone seeing a health care provider about a health problem.
- Consult your health care provider before using omega-3 supplements. If you are pregnant, trying to become pregnant, or breastfeeding; if you take medicine that affects blood clotting; if you are allergic to fish or shellfish; or if you are considering giving a child an omega-3 supplement, it is especially important to consult your (or your child’s) health care provider.
- Research indicates that omega-3 supplements don’t reduce the risk of heart disease. However, people who eat seafood one to four times a week are less likely to die of heart disease.
- For some health conditions, the evidence for benefits from seafood (fish and shellfish) is stronger than the evidence for omega-3 supplements.
- How could this happen? Here are some possibilities:
- Seafood may provide enough omega-3s; more may not be better.
- Other nutrients in seafood besides omega-3s may play a role in its benefits.
- Some of the benefits of seafood may result from people eating it in place of less healthful foods.
- There’s evidence that people who eat seafood have generally healthier lifestyles.
- How could this happen? Here are some possibilities:
- For some health conditions, the evidence for benefits from seafood (fish and shellfish) is stronger than the evidence for omega-3 supplements.
- High doses of omega-3s can reduce levels of triglycerides.
- Omega-3 supplements may help relieve symptoms of rheumatoid arthritis.
- Omega-3 supplements have not been convincingly shown to slow the progression of the eye disease age-related macular degeneration.
- For most other conditions for which omega-3 supplements have been studied, the evidence is inconclusive or doesn’t indicate that omega-3s are beneficial.
Omega-3 supplements safety
- Omega-3 fatty acid supplements usually do not have negative side effects. When side effects do occur, they typically consist of minor gastrointestinal symptoms, such as belching, indigestion, or diarrhea.
- It is uncertain whether people with fish or shellfish allergies can safely consume fish oil supplements.
- Omega-3 supplements may extend bleeding time (the time it takes for a cut to stop bleeding). People who take drugs that affect bleeding time, such as anticoagulants (“blood thinners”) or nonsteroidal anti-inflammatory drugs (NSAIDs), should discuss the use of omega-3 fatty acid supplements with a health care provider. Intakes of about 3–4 g of EPA and DHA per day have resulted in a moderate increase in bleeding times that are generally lower than those seen with aspirin therapy. On rare occasions, mild bouts of diarrhea or other minor gastrointestinal disturbances are sometimes seen with the use of encapsulated fish oil supplementation.
- Fish liver oils, such as cod liver oil, are not the same as fish oil. Fish liver oils contain vitamins A and vitamin D as well as omega-3 fatty acids. Both of these vitamins can be toxic in large doses. The amounts of vitamins in fish liver oil supplements vary from one product to another.
- There is conflicting evidence about whether omega-3 fatty acids found in seafood and fish oil might increase the risk of prostate cancer. Additional research on the association of omega-3 consumption and prostate cancer risk is under way.
In general taking fish oil supplement is quite safe. Commonly reported side effects of omega-3 supplements are usually mild. These include unpleasant taste, bad breath, heartburn, nausea, gastrointestinal discomfort, diarrhea, headache, and odoriferous sweat 172, 173.
Common gastrointestinal symptoms like nausea occurred at rates of ≈4% at dosages <3 g/d and increased to ≈20% at a dosage of 4 g/d.
The Institute of Medicine did not establish a UL for any omega-3s, although it noted that high doses of DHA and/or EPA (900 mg/day of EPA plus 600 mg/day DHA or more for several weeks) might reduce immune function due to suppression of inflammatory responses. Doses of 2–15 g/day EPA and/or DHA might also increase bleeding time by reducing platelet aggregation 8. However, according to the European Food Safety Authority, long-term consumption of EPA and DHA supplements at combined doses of up to about 5 g/day appears to be safe 174. It noted that these doses have not been shown to cause bleeding problems or affect immune function, glucose homeostasis, or lipid peroxidation. The FDA recommends not exceeding 3 g/day EPA and DHA combined, with up to 2 g/day from dietary supplements. Some doses used in clinical trials exceed these levels.
Omega-3 supplements may extend bleeding time (the time it takes for a cut to stop bleeding). People who take drugs that affect bleeding time, such as anticoagulants (“blood thinners”) or nonsteroidal anti-inflammatory drugs (NSAIDs), should discuss the use of omega-3 fatty acid supplements with a health care provider.
- Gibson, R. A., Muhlhausler, B., & Makrides, M. (2011). Conversion of linoleic acid and alpha-linolenic acid to long-chain polyunsaturated fatty acids (LCPUFAs), with a focus on pregnancy, lactation and the first 2 years of life. Maternal & child nutrition, 7 Suppl 2(Suppl 2), 17–26. https://doi.org/10.1111/j.1740-8709.2011.00299.x[↩]
- Hibbeln JR, Davis JM, Steer C, Emmett P, Rogers I, Williams C, Golding J. Maternal seafood consumption in pregnancy and neurodevelopmental outcomes in childhood (ALSPAC study): an observational cohort study. Lancet. 2007 Feb 17;369(9561):578-85. doi: 10.1016/S0140-6736(07)60277-3[↩][↩]
- Spector AA. Plasma free fatty acid and lipoproteins as sources of polyunsaturated fatty acid for the brain. J Mol Neurosci. 2001 Apr-Jun;16(2-3):159-65; discussion 215-21. doi: 10.1385/JMN:16:2-3:159[↩]
- Jones PJH, Rideout T. Lipids, sterols, and their metabolites. In: Ross AC, Caballero B, Cousins RJ, Tucker KL, Ziegler TR, eds. Modern Nutrition in Health and Disease. 11th ed. Baltimore, MD: Lippincott Williams & Wilkins; 2014.[↩][↩][↩][↩][↩]
- Jones PJH, Papamandjaris AA. Lipids: cellular metabolism. In: Erdman JW, Macdonald IA, Zeisel SH, eds. Present Knowledge in Nutrition. 10th ed. Washington, DC: Wiley-Blackwell; 2012:132-48.[↩]
- Harris WS. Omega-3 fatty acids. In: Coates PM, Betz JM, Blackman MR, et al., eds. Encyclopedia of Dietary Supplements. 2nd ed. London and New York: Informa Healthcare; 2010:577-86.[↩][↩][↩][↩]
- Lichtenstein AH, Jones PJH. Lipids: absorption and transport. In: Erdman JW, Macdonald IA, Zeisel SH, eds. Present Knowledge in Nutrition. 10th ed. Washington, DC: Wiley-Blackwell; 2012:118-31.[↩]
- Institute of Medicine, Food and Nutrition Board. Dietary reference intakes for energy, carbohydrate, fiber, fat, fatty acids, cholesterol, protein, and amino acids (macronutrients). Washington, DC: National Academy Press; 2005.[↩][↩][↩]
- SanGiovanni JP, Chew EY. The role of omega-3 long-chain polyunsaturated fatty acids in health and disease of the retina. Prog Retin Eye Res. 2005 Jan;24(1):87-138. doi: 10.1016/j.preteyeres.2004.06.002[↩][↩]
- Serhan C. N. (2014). Pro-resolving lipid mediators are leads for resolution physiology. Nature, 510(7503), 92–101. https://doi.org/10.1038/nature13479[↩]
- Oscarsson, J., & Hurt-Camejo, E. (2017). Omega-3 fatty acids eicosapentaenoic acid and docosahexaenoic acid and their mechanisms of action on apolipoprotein B-containing lipoproteins in humans: a review. Lipids in health and disease, 16(1), 149. https://doi.org/10.1186/s12944-017-0541-3[↩]
- Scorletti E, Byrne CD. Omega-3 fatty acids and non-alcoholic fatty liver disease: Evidence of efficacy and mechanism of action. Mol Aspects Med. 2018 Dec;64:135-146. doi: 10.1016/j.mam.2018.03.001[↩]
- U.S. Department of Agriculture. Dietary Guidelines for Americans, 2020-2025. 9th ed. 2020. https://www.dietaryguidelines.gov/sites/default/files/2021-03/Dietary_Guidelines_for_Americans-2020-2025.pdf[↩][↩][↩]
- Advice about Eating Fish. https://www.fda.gov/food/consumers/advice-about-eating-fish[↩][↩][↩]
- Section on Breastfeeding. Breastfeeding and the use of human milk. Pediatrics. 2012 Mar;129(3):e827-41. doi: 10.1542/peds.2011-3552[↩][↩]
- Skulas-Ray AC, Wilson PWF, Harris WS, Brinton EA, Kris-Etherton PM, Richter CK, Jacobson TA, Engler MB, Miller M, Robinson JG, Blum CB, Rodriguez-Leyva D, de Ferranti SD, Welty FK; American Heart Association Council on Arteriosclerosis, Thrombosis and Vascular Biology; Council on Lifestyle and Cardiometabolic Health; Council on Cardiovascular Disease in the Young; Council on Cardiovascular and Stroke Nursing; and Council on Clinical Cardiology. Omega-3 Fatty Acids for the Management of Hypertriglyceridemia: A Science Advisory From the American Heart Association. Circulation. 2019 Sep 17;140(12):e673-e691. doi: 10.1161/CIR.0000000000000709[↩][↩][↩]
- Siscovick, D. S., Barringer, T. A., Fretts, A. M., Wu, J. H., Lichtenstein, A. H., Costello, R. B., Kris-Etherton, P. M., Jacobson, T. A., Engler, M. B., Alger, H. M., Appel, L. J., Mozaffarian, D., & American Heart Association Nutrition Committee of the Council on Lifestyle and Cardiometabolic Health; Council on Epidemiology and Prevention; Council on Cardiovascular Disease in the Young; Council on Cardiovascular and Stroke Nursing; and Council on Clinical Cardiology (2017). Omega-3 Polyunsaturated Fatty Acid (Fish Oil) Supplementation and the Prevention of Clinical Cardiovascular Disease: A Science Advisory From the American Heart Association. Circulation, 135(15), e867–e884. https://doi.org/10.1161/CIR.0000000000000482[↩][↩][↩][↩]
- Rimm, E. B., Appel, L. J., Chiuve, S. E., Djoussé, L., Engler, M. B., Kris-Etherton, P. M., Mozaffarian, D., Siscovick, D. S., Lichtenstein, A. H., & American Heart Association Nutrition Committee of the Council on Lifestyle and Cardiometabolic Health; Council on Epidemiology and Prevention; Council on Cardiovascular Disease in the Young; Council on Cardiovascular and Stroke Nursing; and Council on Clinical Cardiology (2018). Seafood Long-Chain n-3 Polyunsaturated Fatty Acids and Cardiovascular Disease: A Science Advisory From the American Heart Association. Circulation, 138(1), e35–e47. https://doi.org/10.1161/CIR.0000000000000574[↩][↩][↩][↩]
- Salem N., Jr Introduction to polyunsaturated fatty acids. Backgrounder. 1999;3:1–8.[↩][↩]
- Clinical nutrition: 4. Omega-3 fatty acids in cardiovascular care. Holub BJ. CMAJ. 2002 Mar 5; 166(5):608-15. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC99405/[↩][↩]
- Simopoulos AP. An Increase in the Omega-6/Omega-3 Fatty Acid Ratio Increases the Risk for Obesity. Nutrients. 2016;8(3):128. doi:10.3390/nu8030128. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4808858/[↩][↩]
- Alpha-linolenic acid and cardiovascular diseases. Lanzmann-Petithory D. J Nutr Health Aging. 2001; 5(3):179-83. https://www.ncbi.nlm.nih.gov/pubmed/11458289/[↩]
- Dietary intakes of omega-6 and omega-3 polyunsaturated fatty acids and the risk of breast cancer. Thiébaut AC, Chajès V, Gerber M, Boutron-Ruault MC, Joulin V, Lenoir G, Berrino F, Riboli E, Bénichou J, Clavel-Chapelon F. Int J Cancer. 2009 Feb 15; 124(4):924-31. https://www.ncbi.nlm.nih.gov/pubmed/19035453/[↩]
- Dietary linoleic acid influences desaturation and acylation of deuterium-labeled linoleic and linolenic acids in young adult males. Emken EA, Adlof RO, Gulley RM. Biochim Biophys Acta. 1994 Aug 4; 1213(3):277-88. https://www.ncbi.nlm.nih.gov/pubmed/7914092/[↩]
- Mediterranean diet, traditional risk factors, and the rate of cardiovascular complications after myocardial infarction: final report of the Lyon Diet Heart Study. de Lorgeril M, Salen P, Martin JL, Monjaud I, Delaye J, Mamelle N. Circulation. 1999 Feb 16; 99(6):779-85. https://www.ncbi.nlm.nih.gov/pubmed/9989963/[↩]
- Dietary intake of alpha-linolenic acid and risk of fatal ischemic heart disease among women. Hu FB, Stampfer MJ, Manson JE, Rimm EB, Wolk A, Colditz GA, Hennekens CH, Willett WC. Am J Clin Nutr. 1999 May; 69(5):890-7. http://ajcn.nutrition.org/content/69/5/890.long[↩]
- alpha-Linolenic acid intake is not beneficially associated with 10-y risk of coronary artery disease incidence: the Zutphen Elderly Study. Oomen CM, Ocké MC, Feskens EJ, Kok FJ, Kromhout D. Am J Clin Nutr. 2001 Oct; 74(4):457-63. http://ajcn.nutrition.org/content/74/4/457.long[↩]
- Arterial compliance in obese subjects is improved with dietary plant n-3 fatty acid from flaxseed oil despite increased LDL oxidizability. Nestel PJ, Pomeroy SE, Sasahara T, Yamashita T, Liang YL, Dart AM, Jennings GL, Abbey M, Cameron JD. Arterioscler Thromb Vasc Biol. 1997 Jun; 17(6):1163-70. http://atvb.ahajournals.org/content/17/6/1163.long[↩]
- Dietary supplementation with marine omega-3 fatty acids improve systemic large artery endothelial function in subjects with hypercholesterolemia. Goodfellow J, Bellamy MF, Ramsey MW, Jones CJ, Lewis MJ. J Am Coll Cardiol. 2000 Feb; 35(2):265-70. https://www.ncbi.nlm.nih.gov/pubmed/10676668/[↩][↩]
- Fish oil improves arterial compliance in non-insulin-dependent diabetes mellitus. McVeigh GE, Brennan GM, Cohn JN, Finkelstein SM, Hayes RJ, Johnston GD. Arterioscler Thromb. 1994 Sep; 14(9):1425-9. http://atvb.ahajournals.org/content/14/9/1425.long[↩]
- Omega-3 fatty acid content of the US food supply. Raper NR, Cronin FJ, Exler J. J Am Coll Nutr. 1992 Jun; 11(3):304-8. https://www.ncbi.nlm.nih.gov/pubmed/1619182/[↩]
- Kris-Etherton PM, Taylor DS, Yu-Poth S, Huth P, Moriarty K, Fishell V, et al. Polyunsaturated fatty acids in the food chain in the United States. Am J Clin Nutr 2000;71(Suppl 1):179-88.[↩]
- Breast cancer risk in rats fed a diet high in n-6 polyunsaturated fatty acids during pregnancy. Hilakivi-Clarke L, Onojafe I, Raygada M, Cho E, Clarke R, Lippman ME. J Natl Cancer Inst. 1996 Dec 18; 88(24):1821-7. https://www.ncbi.nlm.nih.gov/pubmed/8961971/[↩]
- Linoleic acid intake and cancer risk: a review and meta-analysis. Zock PL, Katan MB. Am J Clin Nutr. 1998 Jul; 68(1):142-53. https://www.ncbi.nlm.nih.gov/pubmed/9665108/[↩]
- Holub BJ. Clinical nutrition: 4. Omega-3 fatty acids in cardiovascular care. Hoffer LJ, Jones PJ, eds. CMAJ: Canadian Medical Association Journal. 2002;166(5):608-615. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC99405/[↩][↩][↩]
- EPA and DHA Content of Fish Species. https://health.gov/dietaryguidelines/dga2005/report/html/table_g2_adda2.htm[↩]
- Rodriguez-Leyva D, Bassett CM, McCullough R, Pierce GN. The cardiovascular effects of flaxseed and its omega-3 fatty acid, alpha-linolenic acid. The Canadian Journal of Cardiology. 2010;26(9):489-496. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2989356/[↩]
- Fish oils and cardiovascular disease. Holub BJ. CMAJ. 1989 Nov 15; 141(10):1063. https://www.ncbi.nlm.nih.gov/pubmed/2804830/[↩][↩][↩]
- n-3 Fatty acids and cardiovascular disease risk factors among the Inuit of Nunavik. Dewailly E, Blanchet C, Lemieux S, Sauvé L, Gingras S, Ayotte P, Holub BJ. Am J Clin Nutr. 2001 Oct; 74(4):464-73. https://www.ncbi.nlm.nih.gov/pubmed/11566644/[↩][↩]
- F.B. Hu, L. Bronner, W.C. Willett, et al. Fish and omega-3 fatty acid intake and risk of coronary heart disease in women, JAMA, 287 (2002), pp. 1815-1821[↩]
- C.M. Albert, H. Campos, M.J. Stampfer, et al. Blood levels of long-chain n-3 fatty acids and the risk of sudden death. N Engl J Med, 346 (2002), pp. 1113-1118[↩]
- R.N. Lemaitre, I.B. King, D. Mozaffarian, et al. n-3 Polyunsaturated fatty acids, fatal ischemic heart disease, and nonfatal myocardial infarction in older adults: the Cardiovascular Health Study. Am J Clin Nutr, 77 (2003), pp. 319-325[↩]
- Bang HO, Dyerberg J. Lipid metabolism and ischemic heart disease in Greenland Eskimos. In: Draper HH, editor. Advances in nutrition research. New York: Plenum Publishing; 1980. p. 1-22.[↩][↩]
- Contribution of Selected Traditional and Market Foods to the Diet of Nunavik Inuit Women. Blanchet C, Dewailly E, Ayotte P, Bruneau S, Receveur O, Holub BJ. Can J Diet Pract Res. 2000 Summer; 61(2):50-59. https://www.ncbi.nlm.nih.gov/pubmed/11551348/[↩]
- Food intake patterns and 25-year mortality from coronary heart disease: cross-cultural correlations in the Seven Countries Study. The Seven Countries Study Research Group. Menotti A, Kromhout D, Blackburn H, Fidanza F, Buzina R, Nissinen A. Eur J Epidemiol. 1999 Jul; 15(6):507-15. https://www.ncbi.nlm.nih.gov/pubmed/10485342/[↩]
- N-3 fatty acids from fish and coronary artery disease: implications for public health. Schmidt EB, Skou HA, Christensen JH, Dyerberg J. Public Health Nutr. 2000 Mar; 3(1):91-8. https://www.ncbi.nlm.nih.gov/pubmed/10786728/[↩][↩]
- Effects of changes in fat, fish, and fibre intakes on death and myocardial reinfarction: diet and reinfarction trial (DART). Burr ML, Fehily AM, Gilbert JF, Rogers S, Holliday RM, Sweetnam PM, Elwood PC, Deadman NM. Lancet. 1989 Sep 30; 2(8666):757-61. https://www.ncbi.nlm.nih.gov/pubmed/2571009/[↩]
- Fish consumption and the 30-year risk of fatal myocardial infarction. Daviglus ML, Stamler J, Orencia AJ, Dyer AR, Liu K, Greenland P, Walsh MK, Morris D, Shekelle RB. N Engl J Med. 1997 Apr 10; 336(15):1046-53. https://www.ncbi.nlm.nih.gov/pubmed/9091800/[↩]
- Serum fatty acids and the risk of coronary heart disease. Simon JA, Hodgkins ML, Browner WS, Neuhaus JM, Bernert JT Jr, Hulley SB. Am J Epidemiol. 1995 Sep 1; 142(5):469-76. https://www.ncbi.nlm.nih.gov/pubmed/7677125/[↩]
- Importance of n-3 fatty acids in health and disease. Connor WE. Am J Clin Nutr. 2000 Jan; 71(1 Suppl):171S-5S. https://www.ncbi.nlm.nih.gov/pubmed/10617967/[↩][↩]
- n-3 polyunsaturated fatty acids and the cardiovascular system. Angerer P, von Schacky C. Curr Opin Lipidol. 2000 Feb; 11(1):57-63. https://www.ncbi.nlm.nih.gov/pubmed/10750695/[↩][↩]
- Fish oils and plasma lipid and lipoprotein metabolism in humans: a critical review. Harris WS. J Lipid Res. 1989 Jun; 30(6):785-807. https://www.ncbi.nlm.nih.gov/pubmed/2677200/[↩]
- Triglyceride concentration and ischemic heart disease: an eight-year follow-up in the Copenhagen Male Study. Jeppesen J, Hein HO, Suadicani P, Gyntelberg F. Circulation. 1998 Mar 24; 97(11):1029-36. https://www.ncbi.nlm.nih.gov/pubmed/9531248/[↩]
- Effect of a fish-oil concentrate on serum lipids in postmenopausal women receiving and not receiving hormone replacement therapy in a placebo-controlled, double-blind trial. Stark KD, Park EJ, Maines VA, Holub BJ. Am J Clin Nutr. 2000 Aug; 72(2):389-94. https://www.ncbi.nlm.nih.gov/pubmed/10919932/[↩]
- Effect of omega-3 fatty acids and simvastatin on hemostatic risk factors and postprandial hyperlipemia in patients with combined hyperlipemia. Nordoy A, Bonaa KH, Sandset PM, Hansen JB, Nilsen H. Arterioscler Thromb Vasc Biol. 2000 Jan; 20(1):259-65. https://www.ncbi.nlm.nih.gov/pubmed/10634827/[↩]
- The antiarrhythmic and anticonvulsant effects of dietary N-3 fatty acids. Leaf A, Kang JX, Xiao YF, Billman GE, Voskuyl RA. J Membr Biol. 1999 Nov 1; 172(1):1-11. https://www.ncbi.nlm.nih.gov/pubmed/10552009/[↩]
- M. de Lorgeril, P. Salen, J.L. Martin, I. Monjaud, J. Delaye, N. Mamelle. Mediterranean Diet, traditional risk factors, and the rate of cardiovascular complications after myocardial infarction: final report of the Lyon Diet Heart Study. Circulation, 99 (1999), pp. 779-785[↩]
- Donahue SM, Rifas-Shiman SL, Gold DR, Jouni ZE, Gillman MW, Oken E. Prenatal fatty acid status and child adiposity at age 3 y: results from a US pregnancy cohort. The American Journal of Clinical Nutrition. 2011;93(4):780-788. doi:10.3945/ajcn.110.005801. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3057547/[↩]
- Kabir M., Skurnik G., Naour N., Pechtner V., Meugnier E., Rome S., Quignard-Boulangé A., Vidal H., Slama G., Clément K., et al. Treatment for 2 mo with n 3 polyunsaturated fatty acids reduces adiposity and some atherogenic factors but does not improve insulin sensitivity in women with type 2 diabetes: A randomized controlled study. Am. J. Clin. Nutr. 2007;86:1670–1679. http://ajcn.nutrition.org/content/86/6/1670.long[↩][↩]
- Thorsdottir I., Tomasson H., Gunnarsdottir I., Gisladottir E., Kiely M., Parra M.D., Bandarra N.M., Schaafsma G., Martinéz J.A. Randomized trial of weight-loss-diets for young adults varying in fish and fish oil content. Int. J. Obes. 2007;31:1560–1566. doi: 10.1038/sj.ijo.0803643. https://www.ncbi.nlm.nih.gov/pubmed/17502874[↩][↩][↩]
- Parra D., Ramel A., Bandarra N., Kiely M., Martinez J.A., Thorsdottir I. A diet rich in long chain omega-3 fatty acids modulates satiety in overweight and obese volunteers during weight loss. Appetite. 2008;51:676–680. doi: 10.1016/j.appet.2008.06.003. https://www.ncbi.nlm.nih.gov/pubmed/18602429[↩]
- Couet C., Delarue J., Ritz P., Antoine J.M., Lamisse F. Effect of dietary fish oil on body fat mass and basal fat oxidation in healthy adults. Int. J. Obes. Relat. Metab. Disord. 1997;21:637–643. doi: 10.1038/sj.ijo.0800451. https://www.ncbi.nlm.nih.gov/pubmed/15481762[↩][↩]
- Krebs J.D., Browning L.M., McLean N.K., Rothwell J.L., Mishra G.D., Moore C.S., Jebb S.A. Additive benefits of long-chain n-3 polyunsaturated fatty acids and weight-loss in the management of cardiovascular disease risk in overweight hyperinsulinaemic women. Int. J. Obes. 2006;30:1535–1544. doi: 10.1038/sj.ijo.0803309. https://www.ncbi.nlm.nih.gov/pubmed/16552404[↩]
- Kunesova M., Braunerová R., Hlavatý P., Tvrzická E., Stanková B., Skrha J., Hilgertová J., Hill M., Kopecký J., Wagenknecht M., et al. The influence of n-3 polyunsaturated fatty acids and very low calorie diet during a short-term weight reducing regimen on weight loss and serum fatty acid composition in severely obese women. Physiol. Res. 2006;55:63–72. https://www.ncbi.nlm.nih.gov/pubmed/15857162[↩]
- Del Gobbo, Liana C et al. “ω-3 Polyunsaturated Fatty Acid Biomarkers and Coronary Heart Disease: Pooling Project of 19 Cohort Studies.” JAMA internal medicine vol. 176,8 (2016): 1155-66. https://doi.org/10.1001/jamainternmed.2016.2925[↩]
- Marchioli R, Barzi F, Bomba E, Chieffo C, Di Gregorio D, Di Mascio R, Franzosi MG, Geraci E, Levantesi G, Maggioni AP, Mantini L, Marfisi RM, Mastrogiuseppe G, Mininni N, Nicolosi GL, Santini M, Schweiger C, Tavazzi L, Tognoni G, Tucci C, Valagussa F; GISSI-Prevenzione Investigators. Early protection against sudden death by n-3 polyunsaturated fatty acids after myocardial infarction: time-course analysis of the results of the Gruppo Italiano per lo Studio della Sopravvivenza nell’Infarto Miocardico (GISSI)-Prevenzione. Circulation. 2002 Apr 23;105(16):1897-903. doi: 10.1161/01.cir.0000014682.14181.f2[↩]
- Wang C, Harris WS, Chung M, Lichtenstein AH, Balk EM, Kupelnick B, Jordan HS, Lau J. n-3 Fatty acids from fish or fish-oil supplements, but not alpha-linolenic acid, benefit cardiovascular disease outcomes in primary- and secondary-prevention studies: a systematic review. Am J Clin Nutr. 2006 Jul;84(1):5-17. doi: 10.1093/ajcn/84.1.5[↩][↩]
- Yokoyama M, Origasa H, Matsuzaki M, Matsuzawa Y, Saito Y, Ishikawa Y, Oikawa S, Sasaki J, Hishida H, Itakura H, Kita T, Kitabatake A, Nakaya N, Sakata T, Shimada K, Shirato K; Japan EPA lipid intervention study (JELIS) Investigators. Effects of eicosapentaenoic acid on major coronary events in hypercholesterolaemic patients (JELIS): a randomised open-label, blinded endpoint analysis. Lancet. 2007 Mar 31;369(9567):1090-8. doi: 10.1016/S0140-6736(07)60527-3. Erratum in: Lancet. 2007 Jul 21;370(9583):220.[↩][↩]
- Tanaka K, Ishikawa Y, Yokoyama M, Origasa H, Matsuzaki M, Saito Y, Matsuzawa Y, Sasaki J, Oikawa S, Hishida H, Itakura H, Kita T, Kitabatake A, Nakaya N, Sakata T, Shimada K, Shirato K; JELIS Investigators, Japan. Reduction in the recurrence of stroke by eicosapentaenoic acid for hypercholesterolemic patients: subanalysis of the JELIS trial. Stroke. 2008 Jul;39(7):2052-8. doi: 10.1161/STROKEAHA.107.509455. Epub 2008 May 1. Erratum in: Stroke. 2008 Sep;39(9): e149.[↩]
- Risk and Prevention Study Collaborative Group, Roncaglioni MC, Tombesi M, Avanzini F, Barlera S, Caimi V, Longoni P, Marzona I, Milani V, Silletta MG, Tognoni G, Marchioli R. n-3 fatty acids in patients with multiple cardiovascular risk factors. N Engl J Med. 2013 May 9;368(19):1800-8. doi: 10.1056/NEJMoa1205409. Erratum in: N Engl J Med. 2013 May 30;368(22):2146.[↩]
- ORIGIN Trial Investigators, Bosch J, Gerstein HC, Dagenais GR, Díaz R, Dyal L, Jung H, Maggiono AP, Probstfield J, Ramachandran A, Riddle MC, Rydén LE, Yusuf S. n-3 fatty acids and cardiovascular outcomes in patients with dysglycemia. N Engl J Med. 2012 Jul 26;367(4):309-18. doi: 10.1056/NEJMoa1203859[↩][↩]
- Kromhout D, Giltay EJ, Geleijnse JM; Alpha Omega Trial Group. n-3 fatty acids and cardiovascular events after myocardial infarction. N Engl J Med. 2010 Nov 18;363(21):2015-26. doi: 10.1056/NEJMoa1003603[↩][↩]
- Manson, J. E., Cook, N. R., Lee, I. M., Christen, W., Bassuk, S. S., Mora, S., Gibson, H., Albert, C. M., Gordon, D., Copeland, T., D’Agostino, D., Friedenberg, G., Ridge, C., Bubes, V., Giovannucci, E. L., Willett, W. C., Buring, J. E., & VITAL Research Group (2019). Marine n-3 Fatty Acids and Prevention of Cardiovascular Disease and Cancer. The New England journal of medicine, 380(1), 23–32. https://doi.org/10.1056/NEJMoa1811403[↩][↩][↩][↩][↩][↩]
- ASCEND Study Collaborative Group, Bowman L, Mafham M, Wallendszus K, Stevens W, Buck G, Barton J, Murphy K, Aung T, Haynes R, Cox J, Murawska A, Young A, Lay M, Chen F, Sammons E, Waters E, Adler A, Bodansky J, Farmer A, McPherson R, Neil A, Simpson D, Peto R, Baigent C, Collins R, Parish S, Armitage J. Effects of n-3 Fatty Acid Supplements in Diabetes Mellitus. N Engl J Med. 2018 Oct 18;379(16):1540-1550. doi: 10.1056/NEJMoa1804989[↩][↩][↩][↩]
- Bhatt DL, Steg PG, Miller M, Brinton EA, Jacobson TA, Ketchum SB, Doyle RT Jr, Juliano RA, Jiao L, Granowitz C, Tardif JC, Ballantyne CM; REDUCE-IT Investigators. Cardiovascular Risk Reduction with Icosapent Ethyl for Hypertriglyceridemia. N Engl J Med. 2019 Jan 3;380(1):11-22. doi: 10.1056/NEJMoa1812792[↩][↩]
- Kris-Etherton, P. M., Richter, C. K., Bowen, K. J., Skulas-Ray, A. C., Jackson, K. H., Petersen, K. S., & Harris, W. S. (2019). Recent Clinical Trials Shed New Light on the Cardiovascular Benefits of Omega-3 Fatty Acids. Methodist DeBakey cardiovascular journal, 15(3), 171–178. https://doi.org/10.14797/mdcj-15-3-171[↩][↩]
- Hu Y, Hu FB, Manson JE. Marine omega-3 supplementation and cardiovascular disease: an updated meta-analysis of 13 randomized controlled trials involving 127 477 participants. J Am Heart Assoc 2019;8:e013543.[↩][↩][↩]
- Abdelhamid, A. S., Brown, T. J., Brainard, J. S., Biswas, P., Thorpe, G. C., Moore, H. J., Deane, K. H., Summerbell, C. D., Worthington, H. V., Song, F., & Hooper, L. (2020). Omega-3 fatty acids for the primary and secondary prevention of cardiovascular disease. The Cochrane database of systematic reviews, 3(3), CD003177. https://doi.org/10.1002/14651858.CD003177.pub5[↩]
- Omega-3 Fatty Acids and Cardiovascular Disease: An Updated Systematic Review. https://effectivehealthcare.ahrq.gov/products/fatty-acids-cardiovascular-disease/research[↩][↩]
- Chowdhury R, Warnakula S, Kunutsor S, Crowe F, Ward HA, Johnson L, Franco OH, Butterworth AS, Forouhi NG, Thompson SG, Khaw KT, Mozaffarian D, Danesh J, Di Angelantonio E. Association of dietary, circulating, and supplement fatty acids with coronary risk: a systematic review and meta-analysis. Ann Intern Med. 2014 Mar 18;160(6):398-406. doi: 10.7326/M13-1788. Erratum in: Ann Intern Med. 2014 May 6;160(9):658.[↩]
- Zhao YT, Chen Q, Sun YX, Li XB, Zhang P, Xu Y, Guo JH. Prevention of sudden cardiac death with omega-3 fatty acids in patients with coronary heart disease: a meta-analysis of randomized controlled trials. Ann Med. 2009;41(4):301-10. doi: 10.1080/07853890802698834[↩]
- Mozaffarian D, Wu JH. Omega-3 fatty acids and cardiovascular disease: effects on risk factors, molecular pathways, and clinical events. J Am Coll Cardiol. 2011 Nov 8;58(20):2047-67. doi: 10.1016/j.jacc.2011.06.063[↩]
- O’Connor NR. Infant formula. Am Fam Physician. 2009 Apr 1;79(7):565-70. https://www.aafp.org/afp/2009/0401/p565.html[↩]
- Simmer K, Patole SK, Rao SC. Long-chain polyunsaturated fatty acid supplementation in infants born at term. Cochrane Database Syst Rev. 2011 Dec 7;(12):CD000376. doi: 10.1002/14651858.CD000376.pub3. Update in: Cochrane Database Syst Rev. 2017 Mar 10;3:CD000376[↩]
- Schulzke SM, Patole SK, Simmer K. Long-chain polyunsaturated fatty acid supplementation in preterm infants. Cochrane Database Syst Rev. 2011 Feb 16;(2):CD000375. doi: 10.1002/14651858.CD000375.pub4. Update in: Cochrane Database Syst Rev. 2016 Dec 20;12 :CD000375[↩]
- Oken, E., Radesky, J. S., Wright, R. O., Bellinger, D. C., Amarasiriwardena, C. J., Kleinman, K. P., Hu, H., & Gillman, M. W. (2008). Maternal fish intake during pregnancy, blood mercury levels, and child cognition at age 3 years in a US cohort. American journal of epidemiology, 167(10), 1171–1181. https://doi.org/10.1093/aje/kwn034[↩]
- Starling, P., Charlton, K., McMahon, A. T., & Lucas, C. (2015). Fish intake during pregnancy and foetal neurodevelopment–a systematic review of the evidence. Nutrients, 7(3), 2001–2014. https://doi.org/10.3390/nu7032001[↩]
- Makrides M, Gibson RA, McPhee AJ, Yelland L, Quinlivan J, Ryan P; DOMInO Investigative Team. Effect of DHA supplementation during pregnancy on maternal depression and neurodevelopment of young children: a randomized controlled trial. JAMA. 2010 Oct 20;304(15):1675-83. doi: 10.1001/jama.2010.1507[↩]
- Makrides M, Gould JF, Gawlik NR, Yelland LN, Smithers LG, Anderson PJ, Gibson RA. Four-year follow-up of children born to women in a randomized trial of prenatal DHA supplementation. JAMA. 2014 May 7;311(17):1802-4. doi: 10.1001/jama.2014.2194[↩]
- Molloy CS, Stokes S, Makrides M, Collins CT, Anderson PJ, Doyle LW. Long-term effect of high-dose supplementation with DHA on visual function at school age in children born at <33 wk gestational age: results from a follow-up of a randomized controlled trial. Am J Clin Nutr. 2016 Jan;103(1):268-75. doi: 10.3945/ajcn.115.114710[↩]
- Meldrum SJ, D’Vaz N, Simmer K, Dunstan JA, Hird K, Prescott SL. Effects of high-dose fish oil supplementation during early infancy on neurodevelopment and language: a randomised controlled trial. Br J Nutr. 2012 Oct 28;108(8):1443-54. doi: 10.1017/S0007114511006878[↩]
- Saccone G, Berghella V. Omega-3 long chain polyunsaturated fatty acids to prevent preterm birth: a systematic review and meta-analysis. Obstet Gynecol. 2015 Mar;125(3):663-672. doi: 10.1097/AOG.0000000000000668[↩][↩]
- Newberry SJ, Chung M, Booth M, Maglione M, Tang AM, C.E. OH, et al. Omega-3 fatty acids and maternal and child health: an updated systematic review. Evidence Report/Technology Assessment No. 224. (Prepared by the RAND Southern California Evidence-based Practice Center under Contract No. 290-2012-00006-I.) AHRQ Publication No. 16-E003-EF. Rockville, MD: Agency for Healthcare Research and Quality; 2016.[↩][↩]
- Weylandt, K. H., Serini, S., Chen, Y. Q., Su, H. M., Lim, K., Cittadini, A., & Calviello, G. (2015). Omega-3 Polyunsaturated Fatty Acids: The Way Forward in Times of Mixed Evidence. BioMed research international, 2015, 143109. https://doi.org/10.1155/2015/143109[↩]
- Zheng JS, Hu XJ, Zhao YM, Yang J, Li D. Intake of fish and marine n-3 polyunsaturated fatty acids and risk of breast cancer: meta-analysis of data from 21 independent prospective cohort studies. BMJ. 2013 Jun 27;346:f3706. doi: 10.1136/bmj.f3706[↩][↩]
- Wu S, Feng B, Li K, Zhu X, Liang S, Liu X, Han S, Wang B, Wu K, Miao D, Liang J, Fan D. Fish consumption and colorectal cancer risk in humans: a systematic review and meta-analysis. Am J Med. 2012 Jun;125(6):551-9.e5. doi: 10.1016/j.amjmed.2012.01.022[↩][↩]
- MacLean CH, Newberry SJ, Mojica WA, Khanna P, Issa AM, Suttorp MJ, Lim YW, Traina SB, Hilton L, Garland R, Morton SC. Effects of omega-3 fatty acids on cancer risk: a systematic review. JAMA. 2006 Jan 25;295(4):403-15. doi: 10.1001/jama.295.4.403. Erratum in: JAMA. 2006 Apr 26;295(16):1900[↩]
- Gago-Dominguez, M., Yuan, J. M., Sun, C. L., Lee, H. P., & Yu, M. C. (2003). Opposing effects of dietary n-3 and n-6 fatty acids on mammary carcinogenesis: The Singapore Chinese Health Study. British journal of cancer, 89(9), 1686–1692. https://doi.org/10.1038/sj.bjc.6601340[↩]
- Brasky, T. M., Lampe, J. W., Potter, J. D., Patterson, R. E., & White, E. (2010). Specialty supplements and breast cancer risk in the VITamins And Lifestyle (VITAL) Cohort. Cancer epidemiology, biomarkers & prevention : a publication of the American Association for Cancer Research, cosponsored by the American Society of Preventive Oncology, 19(7), 1696–1708. https://doi.org/10.1158/1055-9965.EPI-10-0318[↩]
- Gerber M. Omega-3 fatty acids and cancers: a systematic update review of epidemiological studies. Br J Nutr. 2012 Jun;107 Suppl 2:S228-39. doi: 10.1017/S0007114512001614[↩][↩]
- Geelen A, Schouten JM, Kamphuis C, Stam BE, Burema J, Renkema JM, Bakker EJ, van’t Veer P, Kampman E. Fish consumption, n-3 fatty acids, and colorectal cancer: a meta-analysis of prospective cohort studies. Am J Epidemiol. 2007 Nov 15;166(10):1116-25. doi: 10.1093/aje/kwm197[↩]
- Kantor, E. D., Lampe, J. W., Peters, U., Vaughan, T. L., & White, E. (2014). Long-chain omega-3 polyunsaturated fatty acid intake and risk of colorectal cancer. Nutrition and cancer, 66(4), 716–727. https://doi.org/10.1080/01635581.2013.804101[↩]
- Brasky, T. M., Till, C., White, E., Neuhouser, M. L., Song, X., Goodman, P., Thompson, I. M., King, I. B., Albanes, D., & Kristal, A. R. (2011). Serum phospholipid fatty acids and prostate cancer risk: results from the prostate cancer prevention trial. American journal of epidemiology, 173(12), 1429–1439. https://doi.org/10.1093/aje/kwr027[↩]
- Brasky, T. M., Darke, A. K., Song, X., Tangen, C. M., Goodman, P. J., Thompson, I. M., Meyskens, F. L., Jr, Goodman, G. E., Minasian, L. M., Parnes, H. L., Klein, E. A., & Kristal, A. R. (2013). Plasma phospholipid fatty acids and prostate cancer risk in the SELECT trial. Journal of the National Cancer Institute, 105(15), 1132–1141. https://doi.org/10.1093/jnci/djt174[↩]
- Dahm CC, Gorst-Rasmussen A, Crowe FL, Roswall N, Tjønneland A, Drogan D, Boeing H, Teucher B, Kaaks R, Adarakis G, Zylis D, Trichopoulou A, Fedirko V, Chajes V, Jenab M, Palli D, Pala V, Tumino R, Ricceri F, van Kranen H, Bueno-de-Mesquita HB, Quirós JR, Sánchez MJ, Luján-Barroso L, Larrañaga N, Chirlaque MD, Ardanaz E, Johansson M, Stattin P, Khaw KT, Wareham N, Wark PA, Norat T, Riboli E, Key TJ, Overvad K. Fatty acid patterns and risk of prostate cancer in a case-control study nested within the European Prospective Investigation into Cancer and Nutrition. Am J Clin Nutr. 2012 Dec;96(6):1354-61. doi: 10.3945/ajcn.112.034157[↩]
- Park, S. Y., Wilkens, L. R., Henning, S. M., Le Marchand, L., Gao, K., Goodman, M. T., Murphy, S. P., Henderson, B. E., & Kolonel, L. N. (2009). Circulating fatty acids and prostate cancer risk in a nested case-control study: the Multiethnic Cohort. Cancer causes & control : CCC, 20(2), 211–223. https://doi.org/10.1007/s10552-008-9236-4[↩]
- Bosire, C., Stampfer, M. J., Subar, A. F., Park, Y., Kirkpatrick, S. I., Chiuve, S. E., Hollenbeck, A. R., & Reedy, J. (2013). Index-based dietary patterns and the risk of prostate cancer in the NIH-AARP diet and health study. American journal of epidemiology, 177(6), 504–513. https://doi.org/10.1093/aje/kws261[↩]
- Augustsson K, Michaud DS, Rimm EB, Leitzmann MF, Stampfer MJ, Willett WC, Giovannucci E. A prospective study of intake of fish and marine fatty acids and prostate cancer. Cancer Epidemiol Biomarkers Prev. 2003 Jan;12(1):64-7.[↩]
- Crowe, F. L., Appleby, P. N., Travis, R. C., Barnett, M., Brasky, T. M., Bueno-de-Mesquita, H. B., Chajes, V., Chavarro, J. E., Chirlaque, M. D., English, D. R., Gibson, R. A., Giles, G. G., Goodman, G. E., Henning, S. M., Kaaks, R., King, I. B., Kolonel, L. N., Kristal, A. R., Neuhouser, M. L., Park, S. Y., … Endogenous Hormones, Nutritional Biomarkers and Prostate Cancer Collaborative Group (2014). Circulating fatty acids and prostate cancer risk: individual participant meta-analysis of prospective studies. Journal of the National Cancer Institute, 106(9), dju240. https://doi.org/10.1093/jnci/dju240[↩]
- Chua, M. E., Sio, M. C., Sorongon, M. C., & Morales, M. L., Jr (2013). The relevance of serum levels of long chain omega-3 polyunsaturated fatty acids and prostate cancer risk: A meta-analysis. Canadian Urological Association journal = Journal de l’Association des urologues du Canada, 7(5-6), E333–E343. https://doi.org/10.5489/cuaj.1056[↩]
- Chua, M. E., Sio, M. C., Sorongon, M. C., & Dy, J. S. (2012). Relationship of dietary intake of omega-3 and omega-6 Fatty acids with risk of prostate cancer development: a meta-analysis of prospective studies and review of literature. Prostate cancer, 2012, 826254. https://doi.org/10.1155/2012/826254[↩]
- Szymanski KM, Wheeler DC, Mucci LA. Fish consumption and prostate cancer risk: a review and meta-analysis. Am J Clin Nutr. 2010 Nov;92(5):1223-33. doi: 10.3945/ajcn.2010.29530[↩]
- Alexander, D. D., Bassett, J. K., Weed, D. L., Barrett, E. C., Watson, H., & Harris, W. (2015). Meta-Analysis of Long-Chain Omega-3 Polyunsaturated Fatty Acids (LCω-3PUFA) and Prostate Cancer. Nutrition and cancer, 67(4), 543–554. https://doi.org/10.1080/01635581.2015.1015745[↩]
- Noel SE, Stoneham AC, Olsen CM, Rhodes LE, Green AC. Consumption of omega-3 fatty acids and the risk of skin cancers: a systematic review and meta-analysis. Int J Cancer. 2014 Jul 1;135(1):149-56. doi: 10.1002/ijc.28630. Epub 2013 Dec 18. Erratum in: Int J Cancer. 2017 Jun 1;140(11):E15.[↩]
- Ibiebele TI, Nagle CM, Bain CJ, Webb PM. Intake of omega-3 and omega-6 fatty acids and risk of ovarian cancer. Cancer Causes Control. 2012 Nov;23(11):1775-83. doi: 10.1007/s10552-012-0053-4[↩]
- Arem, H., Neuhouser, M. L., Irwin, M. L., Cartmel, B., Lu, L., Risch, H., Mayne, S. T., & Yu, H. (2013). Omega-3 and omega-6 fatty acid intakes and endometrial cancer risk in a population-based case-control study. European journal of nutrition, 52(3), 1251–1260. https://doi.org/10.1007/s00394-012-0436-z[↩]
- Brasky, T. M., Rodabough, R. J., Liu, J., Kurta, M. L., Wise, L. A., Orchard, T. S., Cohn, D. E., Belury, M. A., White, E., Manson, J. E., & Neuhouser, M. L. (2015). Long-chain ω-3 fatty acid intake and endometrial cancer risk in the Women’s Health Initiative. The American journal of clinical nutrition, 101(4), 824–834. https://doi.org/10.3945/ajcn.114.098988[↩]
- Qin, B., Xun, P., & He, K. (2012). Fish or long-chain (n-3) PUFA intake is not associated with pancreatic cancer risk in a meta-analysis and systematic review. The Journal of nutrition, 142(6), 1067–1073. https://doi.org/10.3945/jn.111.156711[↩]
- Han YJ, Li J, Huang W, Fang Y, Xiao LN, Liao ZE. Fish consumption and risk of esophageal cancer and its subtypes: a systematic review and meta-analysis of observational studies. Eur J Clin Nutr. 2013 Feb;67(2):147-54. doi: 10.1038/ejcn.2012.213[↩]
- Christen, W. G., Schaumberg, D. A., Glynn, R. J., & Buring, J. E. (2011). Dietary ω-3 fatty acid and fish intake and incident age-related macular degeneration in women. Archives of ophthalmology (Chicago, Ill. : 1960), 129(7), 921–929. https://doi.org/10.1001/archophthalmol.2011.34[↩][↩]
- Augood C, Chakravarthy U, Young I, Vioque J, de Jong PT, Bentham G, Rahu M, Seland J, Soubrane G, Tomazzoli L, Topouzis F, Vingerling JR, Fletcher AE. Oily fish consumption, dietary docosahexaenoic acid and eicosapentaenoic acid intakes, and associations with neovascular age-related macular degeneration. Am J Clin Nutr. 2008 Aug;88(2):398-406. doi: 10.1093/ajcn/88.2.398[↩]
- Seddon JM, George S, Rosner B. Cigarette smoking, fish consumption, omega-3 fatty acid intake, and associations with age-related macular degeneration: the US Twin Study of Age-Related Macular Degeneration. Arch Ophthalmol. 2006 Jul;124(7):995-1001. doi: 10.1001/archopht.124.7.995[↩]
- Merle BM, Benlian P, Puche N, Bassols A, Delcourt C, Souied EH; Nutritional AMD Treatment 2 Study Group. Circulating omega-3 Fatty acids and neovascular age-related macular degeneration. Invest Ophthalmol Vis Sci. 2014 Mar 28;55(3):2010-9. doi: 10.1167/iovs.14-13916[↩]
- Age-Related Eye Disease Study Research Group (2001). A randomized, placebo-controlled, clinical trial of high-dose supplementation with vitamins C and E, beta carotene, and zinc for age-related macular degeneration and vision loss: AREDS report no. 8. Archives of ophthalmology (Chicago, Ill. : 1960), 119(10), 1417–1436. https://doi.org/10.1001/archopht.119.10.1417[↩]
- Sangiovanni, J. P., Agrón, E., Meleth, A. D., Reed, G. F., Sperduto, R. D., Clemons, T. E., Chew, E. Y., & Age-Related Eye Disease Study Research Group (2009). {omega}-3 Long-chain polyunsaturated fatty acid intake and 12-y incidence of neovascular age-related macular degeneration and central geographic atrophy: AREDS report 30, a prospective cohort study from the Age-Related Eye Disease Study. The American journal of clinical nutrition, 90(6), 1601–1607. https://doi.org/10.3945/ajcn.2009.27594[↩]
- Age-Related Eye Disease Study 2 Research Group. Lutein + zeaxanthin and omega-3 fatty acids for age-related macular degeneration: the Age-Related Eye Disease Study 2 (AREDS2) randomized clinical trial. JAMA. 2013 May 15;309(19):2005-15. doi: 10.1001/jama.2013.4997. Erratum in: JAMA. 2013 Jul 10;310(2):208.[↩]
- Lawrenson, J. G., & Evans, J. R. (2015). Omega 3 fatty acids for preventing or slowing the progression of age-related macular degeneration. The Cochrane database of systematic reviews, 2015(4), CD010015. https://doi.org/10.1002/14651858.CD010015.pub3[↩]
- Souied EH, Delcourt C, Querques G, Bassols A, Merle B, Zourdani A, Smith T, Benlian P; Nutritional AMD Treatment 2 Study Group. Oral docosahexaenoic acid in the prevention of exudative age-related macular degeneration: the Nutritional AMD Treatment 2 study. Ophthalmology. 2013 Aug;120(8):1619-31. doi: 10.1016/j.ophtha.2013.01.005[↩]
- Dry Eye Assessment and Management Study Research Group, Asbell, P. A., Maguire, M. G., Pistilli, M., Ying, G. S., Szczotka-Flynn, L. B., Hardten, D. R., Lin, M. C., & Shtein, R. M. (2018). n-3 Fatty Acid Supplementation for the Treatment of Dry Eye Disease. The New England journal of medicine, 378(18), 1681–1690. https://doi.org/10.1056/NEJMoa1709691[↩][↩]
- Ziemanski, J. F., Wolters, L. R., Jones-Jordan, L., Nichols, J. J., & Nichols, K. K. (2018). Relation Between Dietary Essential Fatty Acid Intake and Dry Eye Disease and Meibomian Gland Dysfunction in Postmenopausal Women. American journal of ophthalmology, 189, 29–40. https://doi.org/10.1016/j.ajo.2018.01.004[↩][↩]
- Miljanović, B., Trivedi, K. A., Dana, M. R., Gilbard, J. P., Buring, J. E., & Schaumberg, D. A. (2005). Relation between dietary n-3 and n-6 fatty acids and clinically diagnosed dry eye syndrome in women. The American journal of clinical nutrition, 82(4), 887–893. https://doi.org/10.1093/ajcn/82.4.887[↩]
- Hom MM, Asbell P, Barry B. Omegas and Dry Eye: More Knowledge, More Questions. Optom Vis Sci. 2015 Sep;92(9):948-56. doi: 10.1097/OPX.0000000000000655[↩]
- Bhargava, R., Kumar, P., Kumar, M., Mehra, N., & Mishra, A. (2013). A randomized controlled trial of omega-3 fatty acids in dry eye syndrome. International journal of ophthalmology, 6(6), 811–816. https://doi.org/10.3980/j.issn.2222-3959.2013.06.13[↩]
- Epitropoulos, A. T., Donnenfeld, E. D., Shah, Z. A., Holland, E. J., Gross, M., Faulkner, W. J., Matossian, C., Lane, S. S., Toyos, M., Bucci, F. A., Jr, & Perry, H. D. (2016). Effect of Oral Re-esterified Omega-3 Nutritional Supplementation on Dry Eyes. Cornea, 35(9), 1185–1191. https://doi.org/10.1097/ICO.0000000000000940[↩]
- Sydenham E, Dangour AD, Lim WS. Omega 3 fatty acid for the prevention of cognitive decline and dementia. Cochrane Database Syst Rev. 2012 Jun 13;(6):CD005379. doi: 10.1002/14651858.CD005379.pub3[↩][↩]
- Chew, E. Y., Clemons, T. E., Agrón, E., Launer, L. J., Grodstein, F., Bernstein, P. S., & Age-Related Eye Disease Study 2 (AREDS2) Research Group (2015). Effect of Omega-3 Fatty Acids, Lutein/Zeaxanthin, or Other Nutrient Supplementation on Cognitive Function: The AREDS2 Randomized Clinical Trial. JAMA, 314(8), 791–801. https://doi.org/10.1001/jama.2015.9677[↩][↩]
- Yassine HN, Feng Q, Azizkhanian I, Rawat V, Castor K, Fonteh AN, Harrington MG, Zheng L, Reed BR, DeCarli C, Jagust WJ, Chui HC. Association of Serum Docosahexaenoic Acid With Cerebral Amyloidosis. JAMA Neurol. 2016 Oct 1;73(10):1208-1216. doi: 10.1001/jamaneurol.2016.1924[↩]
- van Gelder BM, Tijhuis M, Kalmijn S, Kromhout D. Fish consumption, n-3 fatty acids, and subsequent 5-y cognitive decline in elderly men: the Zutphen Elderly Study. Am J Clin Nutr. 2007 Apr;85(4):1142-7. doi: 10.1093/ajcn/85.4.1142[↩]
- Kalmijn S, Launer LJ, Ott A, Witteman JC, Hofman A, Breteler MM. Dietary fat intake and the risk of incident dementia in the Rotterdam Study. Ann Neurol. 1997 Nov;42(5):776-82. doi: 10.1002/ana.410420514[↩]
- Engelhart MJ, Geerlings MI, Ruitenberg A, Van Swieten JC, Hofman A, Witteman JC, Breteler MM. Diet and risk of dementia: Does fat matter?: The Rotterdam Study. Neurology. 2002 Dec 24;59(12):1915-21. doi: 10.1212/01.wnl.0000038345.77753.46[↩]
- Pottala, J. V., Yaffe, K., Robinson, J. G., Espeland, M. A., Wallace, R., & Harris, W. S. (2014). Higher RBC EPA + DHA corresponds with larger total brain and hippocampal volumes: WHIMS-MRI study. Neurology, 82(5), 435–442. https://doi.org/10.1212/WNL.0000000000000080[↩]
- Tan, Z. S., Harris, W. S., Beiser, A. S., Au, R., Himali, J. J., Debette, S., Pikula, A., Decarli, C., Wolf, P. A., Vasan, R. S., Robins, S. J., & Seshadri, S. (2012). Red blood cell ω-3 fatty acid levels and markers of accelerated brain aging. Neurology, 78(9), 658–664. https://doi.org/10.1212/WNL.0b013e318249f6a9[↩]
- Zhang Y, Chen J, Qiu J, Li Y, Wang J, Jiao J. Intakes of fish and polyunsaturated fatty acids and mild-to-severe cognitive impairment risks: a dose-response meta-analysis of 21 cohort studies. Am J Clin Nutr. 2016 Feb;103(2):330-40. doi: 10.3945/ajcn.115.124081[↩]
- Dangour AD, Allen E, Elbourne D, Fasey N, Fletcher AE, Hardy P, Holder GE, Knight R, Letley L, Richards M, Uauy R. Effect of 2-y n-3 long-chain polyunsaturated fatty acid supplementation on cognitive function in older people: a randomized, double-blind, controlled trial. Am J Clin Nutr. 2010 Jun;91(6):1725-32. doi: 10.3945/ajcn.2009.29121[↩]
- Quinn, J. F., Raman, R., Thomas, R. G., Yurko-Mauro, K., Nelson, E. B., Van Dyck, C., Galvin, J. E., Emond, J., Jack, C. R., Jr, Weiner, M., Shinto, L., & Aisen, P. S. (2010). Docosahexaenoic acid supplementation and cognitive decline in Alzheimer disease: a randomized trial. JAMA, 304(17), 1903–1911. https://doi.org/10.1001/jama.2010.1510[↩]
- Freund-Levi Y, Eriksdotter-Jönhagen M, Cederholm T, Basun H, Faxén-Irving G, Garlind A, Vedin I, Vessby B, Wahlund LO, Palmblad J. Omega-3 fatty acid treatment in 174 patients with mild to moderate Alzheimer disease: OmegAD study: a randomized double-blind trial. Arch Neurol. 2006 Oct;63(10):1402-8. doi: 10.1001/archneur.63.10.1402[↩]
- Lee LK, Shahar S, Chin AV, Yusoff NA. Docosahexaenoic acid-concentrated fish oil supplementation in subjects with mild cognitive impairment (MCI): a 12-month randomised, double-blind, placebo-controlled trial. Psychopharmacology (Berl). 2013 Feb;225(3):605-12. doi: 10.1007/s00213-012-2848-0[↩]
- Yurko-Mauro, K., Alexander, D. D., & Van Elswyk, M. E. (2015). Docosahexaenoic acid and adult memory: a systematic review and meta-analysis. PloS one, 10(3), e0120391. https://doi.org/10.1371/journal.pone.0120391[↩]
- Jiao J, Li Q, Chu J, Zeng W, Yang M, Zhu S. Effect of n-3 PUFA supplementation on cognitive function throughout the life span from infancy to old age: a systematic review and meta-analysis of randomized controlled trials. Am J Clin Nutr. 2014 Dec;100(6):1422-36. doi: 10.3945/ajcn.114.095315[↩]
- Mazereeuw G, Lanctôt KL, Chau SA, Swardfager W, Herrmann N. Effects of ω-3 fatty acids on cognitive performance: a meta-analysis. Neurobiol Aging. 2012 Jul;33(7):1482.e17-29. doi: 10.1016/j.neurobiolaging.2011.12.014[↩]
- Lee YH, Bae SC, Song GG. Omega-3 polyunsaturated fatty acids and the treatment of rheumatoid arthritis: a meta-analysis. Arch Med Res. 2012 Jul;43(5):356-62. doi: 10.1016/j.arcmed.2012.06.011[↩][↩]
- Miles EA, Calder PC. Influence of marine n-3 polyunsaturated fatty acids on immune function and a systematic review of their effects on clinical outcomes in rheumatoid arthritis. Br J Nutr. 2012 Jun;107 Suppl 2:S171-84. doi: 10.1017/S0007114512001560[↩][↩]
- Sköldstam L, Börjesson O, Kjällman A, Seiving B, Akesson B. Effect of six months of fish oil supplementation in stable rheumatoid arthritis. A double-blind, controlled study. Scand J Rheumatol. 1992;21(4):178-85. doi: 10.3109/03009749209099218[↩]
- Park Y, Lee A, Shim SC, Lee JH, Choe JY, Ahn H, Choi CB, Sung YK, Bae SC. Effect of n-3 polyunsaturated fatty acid supplementation in patients with rheumatoid arthritis: a 16-week randomized, double-blind, placebo-controlled, parallel-design multicenter study in Korea. J Nutr Biochem. 2013 Jul;24(7):1367-72. doi: 10.1016/j.jnutbio.2012.11.004[↩]
- Nielsen GL, Faarvang KL, Thomsen BS, Teglbjaerg KL, Jensen LT, Hansen TM, Lervang HH, Schmidt EB, Dyerberg J, Ernst E. The effects of dietary supplementation with n-3 polyunsaturated fatty acids in patients with rheumatoid arthritis: a randomized, double blind trial. Eur J Clin Invest. 1992 Oct;22(10):687-91. doi: 10.1111/j.1365-2362.1992.tb01431.x[↩]
- MacLean CH, Mojica WA, Morton SC, Pencharz J, Hasenfeld Garland R, Tu W, et al. Effects of omega-3 fatty acids on lipids and glycemic control in type ii diabetes and the metabolic syndrome and on inflammatory bowel disease, rheumatoid arthritis, renal disease, systemic lupus erythematosus, and osteoporosis. Evidence Report/Technology Assessment no. 89 (prepared by Southern California/RAND Evidence-based Practice Center, under contract no. 290-02- 0003). Rockville, MD: Agency for Healthcare Research and Quality; 2004.[↩]
- Goldberg RJ, Katz J. A meta-analysis of the analgesic effects of omega-3 polyunsaturated fatty acid supplementation for inflammatory joint pain. Pain. 2007 May;129(1-2):210-23. doi: 10.1016/j.pain.2007.01.020[↩]
- James M, Proudman S, Cleland L. Fish oil and rheumatoid arthritis: past, present and future. Proc Nutr Soc. 2010 Aug;69(3):316-23. doi: 10.1017/S0029665110001564[↩][↩]
- Li F, Liu X, Zhang D. Fish consumption and risk of depression: a meta-analysis. J Epidemiol Community Health. 2016 Mar;70(3):299-304. doi: 10.1136/jech-2015-206278[↩]
- Appleton, K. M., Sallis, H. M., Perry, R., Ness, A. R., & Churchill, R. (2015). Omega-3 fatty acids for depression in adults. The Cochrane database of systematic reviews, 2015(11), CD004692. https://doi.org/10.1002/14651858.CD004692.pub4[↩]
- Cabré E, Mañosa M, Gassull MA. Omega-3 fatty acids and inflammatory bowel diseases – a systematic review. Br J Nutr. 2012 Jun;107 Suppl 2:S240-52. doi: 10.1017/S0007114512001626[↩]
- Lev-Tzion R, Griffiths AM, Leder O, Turner D. Omega 3 fatty acids (fish oil) for maintenance of remission in Crohn’s disease. Cochrane Database Syst Rev. 2014 Feb 28;(2):CD006320. doi: 10.1002/14651858.CD006320.pub4[↩]
- Cochcrane Review 18 July 2007 – Omega 3 fatty acids for maintenance of remission in ulcerative colitis – http://www.cochrane.org/CD006443/IBD_omega-3-fatty-acids-for-maintenance-of-remission-in-ulcerative-colitis[↩]
- Cooper RE, Tye C, Kuntsi J, Vassos E, Asherson P. The effect of omega-3 polyunsaturated fatty acid supplementation on emotional dysregulation, oppositional behaviour and conduct problems in ADHD: A systematic review and meta-analysis. J Affect Disord. 2016 Jan 15;190:474-482. doi: 10.1016/j.jad.2015.09.053[↩]
- Best KP, Gold M, Kennedy D, Martin J, Makrides M. Omega-3 long-chain PUFA intake during pregnancy and allergic disease outcomes in the offspring: a systematic review and meta-analysis of observational studies and randomized controlled trials. Am J Clin Nutr. 2016 Jan;103(1):128-43. doi: 10.3945/ajcn.115.111104[↩]
- Gunaratne, A. W., Makrides, M., & Collins, C. T. (2015). Maternal prenatal and/or postnatal n-3 long chain polyunsaturated fatty acids (LCPUFA) supplementation for preventing allergies in early childhood. The Cochrane database of systematic reviews, 2015(7), CD010085. https://doi.org/10.1002/14651858.CD010085.pub2[↩]
- Oliver, C., & Watson, H. (2016). Omega-3 fatty acids for cystic fibrosis. The Cochrane database of systematic reviews, 2016(1), CD002201. https://doi.org/10.1002/14651858.CD002201.pub5[↩]
- Cochcrane Review 23 January 2008 – Omega-3 polyunsaturated fatty acids (PUFA) for type 2 diabetes mellitus – http://www.cochrane.org/CD003205/ENDOC_omega-3-polyunsaturated-fatty-acids-pufa-for-type-2-diabetes-mellitus[↩]
- Cochcrane Review 9 November 2011 – Omega-3 fatty acids for autism spectrum disorders – http://www.cochrane.org/CD007992/BEHAV_omega-3-fatty-acids-for-autism-spectrum-disorders-asd[↩]
- Cochcrane Review 4 July 2013 – Omega-3 fatty acid supplementation for intermittent claudication – http://www.cochrane.org/CD003833/PVD_omega-3-fatty-acid-supplementation-for-intermittent-claudication[↩]
- Cochcrane Review 16 April 2008 – Omega-3 fatty acids for bipolar disorder – http://www.cochrane.org/CD005169/DEPRESSN_omega-3-fatty-acids-for-bipolar-disorder[↩]
- Mazereeuw G, Lanctot KL, Chau SA, Swardfager W, Herrmann N. Effects of omega-3 fatty acids on cognitive performance: a meta-analysis. Neurobiol Aging 2012;33:1482 e17-29. https://www.ncbi.nlm.nih.gov/pubmed/22305186?dopt=Abstract[↩]
- Lev-Tzion R, Griffiths AM, Leder O, Turner D. Omega 3 fatty acids (fish oil) for maintenance of remission in Crohn’s disease. Cochrane Database Syst Rev 2014;2:CD006320. https://www.ncbi.nlm.nih.gov/pubmed/24585498?dopt=Abstract[↩]
- EFSA Panel on Dietetic Products NaA. Scientific opinion on the tolerable upper intake level of eicosapentaenoic acid (EPA), docosahexaenoic acid (DHA) and docosapentaenoic acid (DPA). EFSA Journal 2012;10:2815.[↩]