What is body fat spot reduction
Overweight and obesity are increasingly common conditions in the United States. They are caused by the increase in the size and the amount of fat cells in the body. Doctors measure body mass index (BMI) and waist circumference (waist hip ratio) to screen and diagnose overweight and obesity. Obesity is a serious medical condition that can cause complications such as metabolic syndrome, high blood pressure, atherosclerosis, heart disease, diabetes, high blood cholesterol, cancers and sleep disorders. Treatment depends on the cause and severity of your condition and whether you have complications. Treatments include lifestyle changes, such as heart-healthy eating and increased physical activity, and Food and Drug Administration (FDA)-approved weight-loss medicines. For some people, surgery may be a treatment option.
Maintaining a healthy weight calls for keeping a balance, a balance of energy. You must balance the calories or energy that you get from food and beverages with the calories that you use to keep your body going and to be physically active.
Energy imbalances can cause overweight and obesity. An energy imbalance means that your energy IN does not equal your energy OUT. This energy is measured in calories. Energy IN is the amount of calories you get from food and drinks. Energy OUT is the amount of calories that your body uses for things such as breathing, digesting, being physically active, and regulating body temperature.
Your body uses certain nutrients such as carbohydrates or sugars, proteins, and fats from the foods you eat to:
- Make energy for immediate use to power routine daily body functions and physical activity.
- Store energy for future use by your body. Sugars are stored as glycogen in the liver and muscles. Fats are stored mainly as triglycerides in fat tissue.
The amount of energy that your body gets from the food you eat depends on the type of foods you eat, how the food is prepared, and how long it has been since you last ate.
- The same amount of energy IN and OUT over time = weight stays the same
- More energy IN than OUT over time = Weight Gain = Body Fat
- More energy OUT than IN over time = Weight Loss
Your energy IN and OUT doesn’t have to balance exactly every day: Balancing energy over time will help you to maintain a healthy weight in the long run.
- Overweight and obesity develop over time when you take in more calories than you use, or when energy IN is more than your energy OUT. This type of energy imbalance causes your body to store fat.
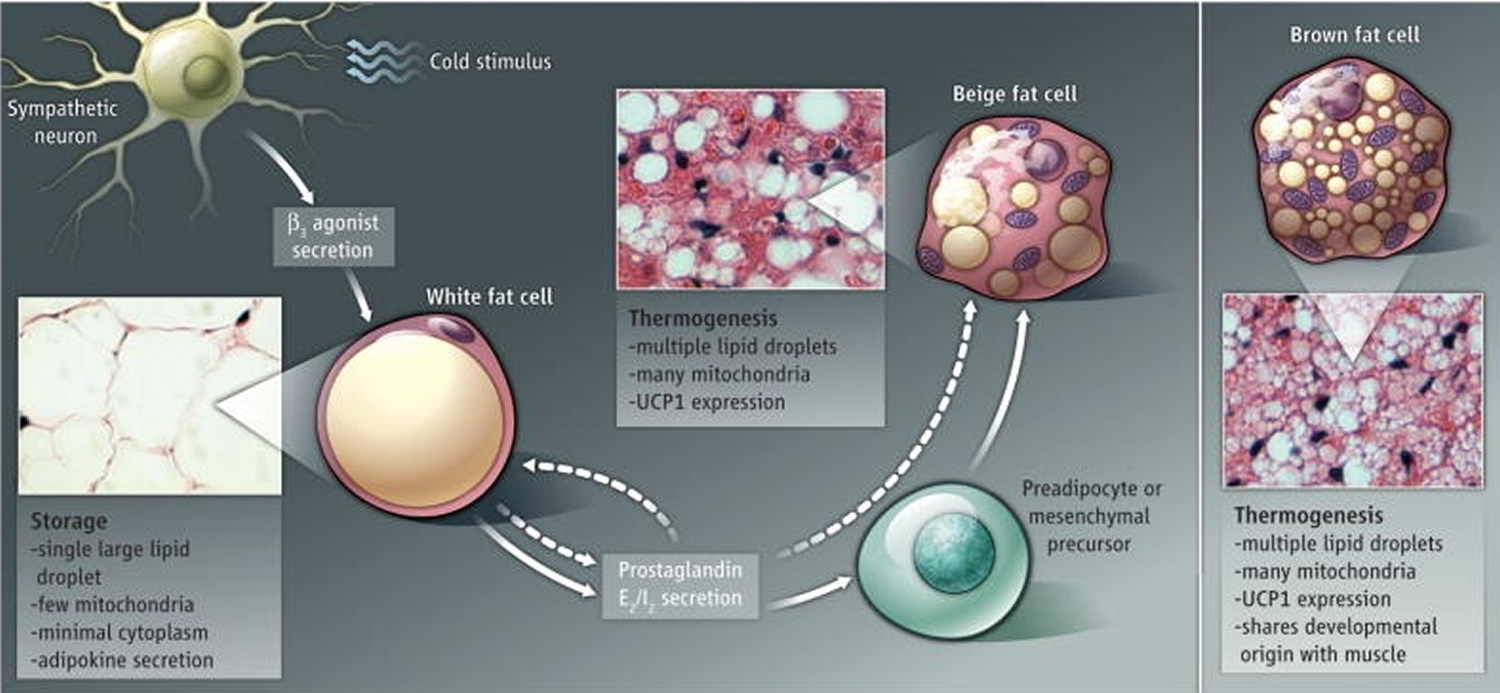
The body has three types of fat tissue—white, brown, and beige—that it uses to fuel itself, regulate its temperature in response to cold, and store energy for future use. Learn about the role of each fat type in maintaining energy balance in the body.
- White fat tissue can be found around the kidneys and under the skin in the buttocks, thighs, and abdomen. This fat type stores energy, makes hormones that control the way the body regulates urges to eat or stop eating, and makes inflammatory substances that can lead to complications. White fat store excess energy in the form of triglycerides for future need 1.
- Brown fat tissue is located in the upper back area of human infants. This fat type releases stored energy as heat energy when a baby is cold. It also can make inflammatory substances. Brown fat can be seen in children and adults. Brown fat metabolize lipid and glucose to produce heat in a process known as nonshivering thermogenesis 2. Brown adipocytes are packed with specialized mitochondria that contain Uncoupling Protein-1 (Ucp1, thermogenin) in their inner membrane. When activated, Ucp1 dissipates the electrochemical gradient used for ATP synthesis, resulting in increased heat production.
- Beige fat tissue is seen in the neck, shoulders, back, chest and abdomen of adults and resembles brown fat tissue. This fat type, which uses carbohydrates and fats to produce heat, increases when children and adults are exposed to cold.
Infants have prominent depots of brown fat that regulate body temperature 3. Although these “newborn” brown fat depots regress during childhood, appreciable amounts of brown fat persist in adults and correlate with adult leanness 4. Similarly, brown fat counteracts obesity and metabolic disease in rodents 5. Therefore, enhancing brown fat thermogenesis is an attractive target for anti-obesity therapy. Vegiopoulos et al. 6 show that white fat can be induced to form intermediate “beige” adipocytes with many of the properties of brown adipocytes, but have a completely independent developmental origin (see the Figure 1).
In rats, brown fat tissue is a major site for cold-induced thermogenesis, which is essential for protection against hypothermia 1. Brown fat tissue is also activated by overfeeding in mice as a physiological countermeasure to limit weight gain 7. Indeed, mouse strains that have increased brown fat tissue activity and/or increased numbers of brown-like (beige) fat cells within their white fat tissue are lean and healthy 8, 9. By contrast, mice lacking brown and/or beige fat activity are susceptible to obesity 10.
Figure 1. Three types of body fat tissue
Note: White to beige. Fat cells can be classified as white storage cells, brown thermogenic cells, or intermediate beige cells. Prostaglandins produced by white fat cells (in response to β3-adrenergic agonists) or from the circulation stimulate the formation of thermogenic beige cells in white fat. The origin of beige cells is unclear.
[Source 3]White fat tissue is highly adapted to store excess energy in the form of triglycerides. Conversely, brown fat tissue oxidizes chemical energy to produce heat as a defense against hypothermia and obesity. White fat tissue develops in distinct intra-abdominal depots and in the subcutaneous layer (between the fascia and muscle). The accumulation of intra-abdominal, visceral white fat tissue, rather than total adiposity, is most strongly correlated with elevated risk for metabolic dysfunction and cardiovascular disease 11, 12. By contrast, expansion of subcutaneous white fat tissue, even in the setting of obesity, has been suggested to promote insulin sensitivity in rodents and humans 13, 14. Implantation of subcutaneous white fat tissue, but not visceral white fat tissue, into the abdominal cavity of mice improves whole-body metabolism 15, 16. Moreover, subcutaneous and visceral white fat tissue express unique gene signatures 17. These data suggest that some of the distinct metabolic effects of subcutaneous and visceral white fat tissue are cell autonomous. However, the molecular mechanisms governing the fat cell–intrinsic differences of these depots have not yet been established.
Brown fat cells, having a multilocular morphology and expressing the brown fat–specific uncoupling protein-1 (Ucp1, thermogenin) protein, are found interspersed in the white fat tissue of rodents and humans. These have been called “adaptive” or “recruitable” brown fat cells, brown in white (brite) cells, or beige cells 18, 19. The development of these brown fat–like cells in WAT is dramatically enhanced during adaptation to cold or in response to treatment with β3-selective adrenergic agonists 20. The brown-like transformation of white fat tissue is most prominent in the inguinal subcutaneous depot, whereas the perigonadal adipose is less susceptible to browning 21.
It has recently become clear that constitutive adipose tissue (cBAT) shares a common developmental ancestry with skeletal muscle 22, whereas recruitable adipose tissue (rBAT), that resides within white fat or skeletal muscle, derives from a non-myogenic lineage 23. Lineage-tracing experiments have also revealed that constitutive adipose tissue (cBAT) arises from progenitors located in the embryonic dermomyotome 24 that express myogenic markers Pax7 and Myf5 25.
Is body fat spot reduction possible ?
In humans, too much abdominal white fat promotes heart disease, diabetes, and many other metabolic diseases. In contrast to the familiar white fatty tissues that specialize in lipid storage, brown fat cells burn fat to produce heat (thermogenesis). Accordingly if you want to lose weight, then you actually want more brown fat. This special type of fatty tissue burns calories, puts out heat like a furnace and helps to keep you lean. Therefore, it would be potentially therapeutic if you could transform some of your white fat into brown fat. Determining which genes control the development of white and brown fat may be the first step toward developing game changing treatments for diabetes and obesity.
Brown Fat or Brown Adipose Tissue
Brown fat tissue is composed of brown fat cells, plus abundant blood vessels and nerves 26. Since brown fat provides heat to the body in response to sympathetic nerve activity and the heat is carried via the bloodstream, this architecture makes sense. Cold exposure in rats and humans causes sympathetic nerve fibers to release norepinephrine in fat tissues 5. Norepinephrine then activates β-adrenergic receptors on brown fat cells to stimulate the breakdown of fats and heat production. In white fat depots, prolonged exposure to cold or β3-adrenergic agonists causes tissue transdifferentiation from white to beige 5. Unfortunately, β3-adrenergic agonists have not reached successful clinical application for weight loss because of technical, efficacy and safety issues 27.
Brown fat cells themselves have a very unique cellular and molecular composition. They are indeed fat cells, having all of the enzymatic machinery to synthesize and store triglyceride; in the case of brown fat cells, lipid is normally stored in multiple small fat droplets (multilocular), as opposed to white fat cells, which usually contain one giant droplet of triglyceride (unilocular). The thermogenic business of brown fat cells is carried out in the very dense mitochondria that these cells possess. In fact, brown fat cells and cardiomyocytes (heart cells) possess the highest levels of mitochondria in mammalian organisms 28. However, unlike heart cells, which utilize mitochondria to generate the extremely high amounts of ATP needed to perform cardiac contractile function, the mitochondria of brown fat cells uncouple large amounts of fuel oxidation from ATP generation. This uncoupling is caused by the presence of Uncoupling Protein-1 (Ucp1, thermogenin), which sits in the inner mitochondrial membrane and catalyzes a leak of protons from the intramembrane space into the mitochondrial matrix 29. The resulting dissipation of the mitochondrial membrane potential, along with extremely high rates of mitochondrial electron transport and fuel oxidation, results in the generation of heat and the expenditure of huge amounts of chemical energy.
Brown fat has been well-established as a crucial component in the defense against the cold in a process called nonshivering thermogenesis 2. Chronic cold exposure of rodents or humans causes an expansion and activation of brown fat 30. Cold is sensed in the central nervous system and the brown fat is activated via catecholamines secreted by sympathetic nerve terminals in the brown adipose tissue itself 30. The thermogenic function of brown fat tissue, is, in fact, only functional in response to adrenergic input. Thyroid hormones also play a key role in the sympathetic activation of nonshivering thermogenesis, in part through the stimulation of Uncoupling Protein-1 (Ucp1, thermogenin) transcription 31. Brown fat can also be activated, and can expand quite massively, by constitutive exposure to β-adrenergic agonists, either pharmacological or due to catecholamine-secreting tumors. In addition to effects on brown adipose tissue, cold exposure or catecholamines causes the emergence of Uncoupling Protein-1 (Ucp1, thermogenin)-expressing brown fat cells in classic depots of white fat 32. In the genetic absence of Uncoupling Protein-1 (Ucp1, thermogenin), mice lose their ability to produce heat by nonshivering thermogenesis and therefore exhibit a profound cold intolerance 33.
Brown fat has also been recognized for its potential and demonstrated anti-obesity properties. Certain diets in rodents that cause overfeeding (cafeteria diets) stimulate the expansion and activation of brown fat, in an apparent physiological effort to restrain weight gain and obesity, so-called diet-induced thermogenesis 34. The genetic ablation of brown fat tissue via expression of a targeted toxigene causes a propensity toward obesity and metabolic disease 35. Likewise, deletion of Uncoupling Protein-1 (Ucp1, thermogenin) in mice causes increased weight gain, when mice are housed at thermoneutrality 10. Indeed Uncoupling Protein-1 (Ucp1, thermogenin) has now clearly been demonstrated as a key factor in diet-induced thermogenesis. In addition, a large number of genetic studies in mice have shown that experimental increases in the amount and/or function of brown fat promotes a lean and healthy phenotype 36, 37, 38. Specifically, mice with higher amounts of brown type fat gain less weight, are more insulin-sensitive, have lower levels of serum-free fatty acids, and are protected from diabetes and other metabolic sequelae. Finally, stimulation of brown fat-mediated thermogenesis in adult animals by pharmacological treatment with β-adrenergic agonists or thyroid receptor agonists does reduce obesity, but thus far, these agents have had too many untoward side-effects to warrant further clinical development 27.
Brown fat tissue and thermogenesis in adult humans
The prevailing dogma that brown fat tissue is not present to significant levels in adult humans has now been refuted. Positron emission tomography (PET) is used diagnostically to detect cancer tissues/cells based on their capacity to take up large amounts of 18fluoro-labeled 2-deoxy-glucose (FDG). During the course of these imaging studies, symmetrically distributed hot spots for 18fluoro-labeled 2-deoxy-glucose (FDG) uptake were noted that had the characteristic density of adipose tissue 39. Moreover, the incorporation of 18fluoro-labeled 2-deoxy-glucose (FDG) in these tissues disappeared when patients were kept warm. Altogether, a large number of such reports had strongly suggested that these tissues were in fact brown fat tissue 40. These reports, however, were largely overlooked by the metabolism/obesity community because they were published in very specialized medical journals. Very recently, the nature of these putative brown fat depots in adult humans has been carefully examined by dedicated FDG-PET analyses, combined with tissue biopsies. Most importantly, by all criteria, these tissues are now unequivocally identified as thermogenic brown fat. Most importantly, the results of Enerback and colleagues clearly demonstrate in the same subjects that human brown fat tissue is dramatically activated by cold exposure. The human brown fat depots are localized in the supraclavicular, cervical, and paravertebral regions and express high levels of PRDM16, PGC-1α, and UCP1. Analysis of a large number of historical FDG-PET scans by Kahn and colleagues suggests an inverse correlation between brown fat tissue volume and BMI (Body mass index). Based on the amount of active brown fat tissue detected by these studies, it seems very likely that these tissues may make an important contribution to whole-body energy balance. However, because the detection of brown fat tissue by FDG-PET is so temperature-sensitive, dedicated studies to examine the relationship between obesity and brown fat tissue activity under standardized conditions are needed. Taken together, these seminal studies have revitalized interest in brown fat tissue as an important metabolic tissue in humans.
Why would you want to change white fat into brown ?
White fat stores energy as large fat droplets, while brown fat has much smaller droplets and is specialized to burn them, yielding heat. Brown fat cells are packed with energy generating powerhouses called mitochondria that contain iron—which gives them their brown color. Infants are born with rich stores of brown fat (about 5% of total body mass) on the upper spine and shoulders to keep them warm. It used to be thought that brown fat disappeared by adulthood—but it turns out you harbor small reserves in your shoulders and neck.
Brown adipocytes are packed with specialized mitochondria that contain Uncoupling Protein-1 (Ucp1, thermogenin) in their inner membrane. When activated, Uncoupling Protein-1 (Ucp1, thermogenin) dissipates the electrochemical gradient used for ATP synthesis, resulting in increased heat production. Brown fat cells metabolize lipid and glucose to produce heat in a process known as nonshivering thermogenesis 2. Although impossible to prove, good arguments can be forwarded that the acquisition of brown adipose tissue with its new protein, uncoupling protein-1 (UCP1, thermogenin), may have been the one development that gave mammals our evolutionary advantage, i.e., to survive and especially to be active during periods of nocturnal or hibernal cold, to survive the cold stress of birth, and probably also by promoting our survival on diets low in essential macronutrients, especially protein.
In mice, brown fat does something remarkable: it burns more calories when mice are overfed, protecting them from obesity. (Don’t you wish eating a plate of fries did that for you ?) Furthermore, mice genetically predisposed to have with extra brown fat are actually leaner and healthier. In humans, there is evidence that more brown fat is associated with a lower body weight.
How might you increase your brown fat production ?
Fat has been villainized; but all fat was not created equal. Our two main types of fat—brown and white—play different roles. Scientists have discovered the genetic switch that triggers the development of brown fat 2 and the second figured out how your body can recruit white fat and transform it into brown fat 41.
The team led by the University of Pennsylvania 1 figured out the switch for creating a brown fat cell—a protein called early B cell factor-2 (Ebf2). Comparing the active genes in brown and white fat cells, they discovered Ebf2 is present in larger quantities in brown fat. This protein seems to mark which genes will later be turned on to transform certain types of precursor cells into brown fat. When the team engineered mice lacking this protein, the animals had white fat cells on their upper back and spine rather than the typical brown. When the team expressed high levels of Ebf2 in white fat, these cells turned brown and consumed more oxygen—a sign they were producing more heat.
The second team, led by Harvard’s Joslin Diabetes Center 41, noted that mice have two types of brown fat: constitutive brown fat, which they have from birth and “recruitable” brown fat, scattered throughout the muscles and white fat. When researchers engineered mice lacking a protein called Type 1A BMP-receptor (BMPR1A)—which is needed for the correct development of brown fat—the mice were born with just a tiny bit of constitutive brown fat on their back. You would think that these mice would be terribly cold. Surprisingly, they kept a normal body temperature. How did they manage this feat ? The lack of brown fat apparently sends a signal via the brain to the recruitable fat cells, telling them to make the switch and transform into brown fat. The mice stayed warm and the recruited brown fat even protected them from obesity.
How To Lose Weight and Maintain It
Changing the way you approach weight loss can help you be more successful at losing it. Furthermore, maintaining long-term weight loss can also be difficult. Most people who try to lose weight focus on one thing: weight loss. However if you set goals, begin to eat healthy foods, become more physically active, and learn how to change behaviors, then you may be more successful at losing weight. Over time, these changes will become routine and part of your everyday life.
Three keys to success are setting realistic goals, following a healthy diet, and aiming for 60–90 minutes of physical activity most days of the week.
Weight Loss Goals
Setting the right goals is an important first step to losing and maintaining weight.
- Losing just 5-10 percent of your current weight over 6 months will lower your risk for heart disease and other conditions.
- Losing 1-2 pounds per week is a reasonable and safe weight loss. Losing weight at this rate will help you to keep off the weight. And it will give you the time to make new healthy lifestyle changes.
- Maintaining a modest weight loss over a longer period of time is better than losing a lot of weight and regaining it. You can think about additional weight loss after you’ve lost 10 percent of your current body weight and have kept it off for 6 months.
Set specific, realistic goals that are forgiving (less than perfect).
- To start, try walking 30 minutes, 3 days a week.
- Ask for encouragement from your health care provider(s) via telephone or e-mail; friends and family can help. You can also join a support group.
- Keep a record of your food intake and the amount of physical activity that you do. This is an easy way to track how you are doing. A record can also inspire you. For example, when it shows that you’ve been more active, you’ll be encouraged to keep it up.
- Change your surroundings to avoid overeating. For example, don’t eat while watching television. Plan to meet a friend in a nonfood setting.
- Reward your success but not with food. Instead, choose rewards that you’ll enjoy, such as a movie, music CD, an afternoon off from work, a massage, or personal time.
For help with your weight loss goals, you can use these free online tools
- Body Weight Planner. https://www.supertracker.usda.gov/bwp/index.html
To find out about how many calories you should eat to lose weight according to your weight, age, sex, height and physical activity, you can use a FREE online app Body Weight Planner 42
- ChooseMyPlate. https://www.choosemyplate.gov/
To find out about the 5 Food Groups you should have on your plate for a meal, you can use a FREE online app ChooseMyPlate 43
- SuperTracker website: https://supertracker.usda.gov
To find out What and How Much To Eat, you can use a FREE, award-winning, state-of-the-art, online diet and activity tracking tool called SuperTracker 44 from the United States Department of Agriculture Center for Nutrition Policy and Promotion 44. This free application empowers you to build a healthier diet, manage weight, and reduce your risk of chronic diet-related diseases. You can use SuperTracker 44 to determine what and how much to eat; track foods, physical activities, and weight; and personalize with goal setting, virtual coaching, and journaling.
- BMI Calculator Adults. https://www.cdc.gov/healthyweight/assessing/bmi/adult_BMI/english_bmi_calculator/bmi_calculator.html
- BMI Calculator Children. https://nccd.cdc.gov/dnpabmi/Calculator.aspx
To find out about your body mass index (BMI), you can use a FREE online BMI calculators from the Centers for Disease Control and Prevention (CDC) – for Adults 45 and for Children 46
A Healthy Eating Plan
A healthy eating plan gives your body the nutrients it needs every day and helps you to stay within your daily calorie level. This eating plan will also lower your risk for heart disease and such other conditions as high blood pressure and high blood cholesterol levels.
A healthy eating plan:
- Emphasizes fruits, vegetables, whole grains, and fat-free or low-fat milk and milk products
- Includes lean meats, poultry, fish, beans, eggs, and nuts
- Is low in saturated fats, trans fat, cholesterol, salt (sodium), and added sugars
- Controls portion sizes
Calories
Cutting back on calories is part of a healthy eating plan to lose weight. Choose foods that are lower in fats, especially saturated and trans fats, cholesterol, and added sugars. Also, pay attention to portion sizes.
To lose 1–2 pounds a week, daily intake should be reduced by 500 to 1,000 calories. In general:
- Eating plans that contain 1,000-1,200 calories each day will help most women to lose weight safely.
- Eating plans that contain 1,200-1,600 calories each day are suitable for men and may also be appropriate for women who weigh 165 pounds or more or who exercise regularly.
If you eat 1,600 calories a day but do not lose weight, then you may want to cut back to 1,200 calories. If you are hungry on either diet, then you may want to boost your calories by 100 to 200 per day. Very low calorie diets of less than 800 calories per day should not be used unless you are being monitored by your doctor.
Portion Distortion: How To Choose Sensible Servings
It’s very easy to “eat with your eyes” and misjudge what equals a serving—piling on unwanted pounds. This is especially true when you eat out, because restaurant portions are often super sized and enough for two or more people to share.
To keep portion sizes sensible:
- When eating out, choose small portions, share an entrée with a friend, or take some of the food home.
- Check a product’s Nutrition Facts label to learn how much food is considered a serving and how many calories, fat grams, and other nutrients are in the item.
- Limit portion sizes of such high-calorie foods as cookies, cakes, and other sweets; french fries; and oils.
- Use smaller plates. We eat most of what is on our plate, no matter what the size. Smaller plates can mean smaller portions.
Physical Activity
Staying physically active and eating fewer calories will help you lose weight and keep the weight off over time. Plus, physical activity has many benefits:
- Lowers the risk of heart disease; diabetes; and cancers such as breast, uterus, and colon
- Strengthens your lungs and helps them to work more efficiently
- Strengthens your muscles and keeps your joints in good condition
- May slow bone loss
- Gives you more energy
- Helps you to relax and cope better with stress
- Builds confidence
- Allows you to fall asleep more quickly and sleep more soundly
- Provides an enjoyable way to share time with friends and family
How much physical activity should you aim for ?
- For overall health and to reduce the risk of disease, aim for at least 30 minutes of moderate physical activity most days of the week.
- To help manage body weight and prevent gradual weight gain, aim for 60 minutes of moderate-to-vigorous physical activity most days of the week.
- To maintain weight loss, aim for at least 60–90 minutes of daily moderate physical activity.
You can break up the amount of time that you do physical activity, such as 15 minutes at a time. If you haven’t been physically active for some time, then don’t let that stop you. Start slowly and gradually increase your activity. For example, start walking for 10–15 minutes three times a week, then gradually build up to the recommended amount with brisk walking.
You can feel healthier by doing any of the following activities. For added fun, ask friends or family to join you.
- Walk or ride a bike in your neighborhood.
- Join a walking club at a mall or at work.
- Play golf at a local club.
- Join a dance or yoga class.
- Work in your garden.
- Use local athletic facilities.
- Join a hiking or biking club.
- Join a softball team or play other sports with coworkers, friends, and family.
- Rajakumari S, Wu J, Ishibashi J, et al. EBF2 determines and maintains brown adipocyte identity. Cell metabolism. 2013;17(4):562-574. doi:10.1016/j.cmet.2013.01.015. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3622114/[↩][↩][↩]
- Brown adipose tissue: function and physiological significance. Cannon B, Nedergaard J. Physiol Rev. 2004 Jan; 84(1):277-359. http://physrev.physiology.org/content/84/1/277.long[↩][↩][↩][↩]
- Ishibashi J, Seale P. Beige can be slimming. Science (New York, NY). 2010;328(5982):1113-1114. doi:10.1126/science.1190816. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2907667/[↩][↩]
- Cold-activated brown adipose tissue in healthy men. van Marken Lichtenbelt WD, Vanhommerig JW, Smulders NM, Drossaerts JM, Kemerink GJ, Bouvy ND, Schrauwen P, Teule GJ. N Engl J Med. 2009 Apr 9; 360(15):1500-8. http://www.nejm.org/doi/full/10.1056/NEJMoa0808718[↩]
- Transcriptional control of brown adipocyte development and physiological function–of mice and men. Seale P, Kajimura S, Spiegelman BM. Genes Dev. 2009 Apr 1; 23(7):788-97. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2763499/[↩][↩][↩]
- PRDM16 controls a brown fat/skeletal muscle switch. Seale P, Bjork B, Yang W, Kajimura S, Chin S, Kuang S, Scimè A, Devarakonda S, Conroe HM, Erdjument-Bromage H, Tempst P, Rudnicki MA, Beier DR, Spiegelman BM. Nature. 2008 Aug 21; 454(7207):961-7. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2583329/[↩]
- A role for brown adipose tissue in diet-induced thermogenesis. Rothwell NJ, Stock MJ. Nature. 1979 Sep 6; 281(5726):31-5. https://www.ncbi.nlm.nih.gov/pubmed/551265/[↩]
- [↩]
- Prdm16 determines the thermogenic program of subcutaneous white adipose tissue in mice. Seale P, Conroe HM, Estall J, Kajimura S, Frontini A, Ishibashi J, Cohen P, Cinti S, Spiegelman BM. J Clin Invest. 2011 Jan; 121(1):96-105. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3007155/[↩]
- UCP1 ablation induces obesity and abolishes diet-induced thermogenesis in mice exempt from thermal stress by living at thermoneutrality. Feldmann HM, Golozoubova V, Cannon B, Nedergaard J. Cell Metab. 2009 Feb; 9(2):203-9. https://www.ncbi.nlm.nih.gov/pubmed/19187776/[↩][↩]
- Comparison of abdominal adiposity and overall obesity in predicting risk of type 2 diabetes among men. Wang Y, Rimm EB, Stampfer MJ, Willett WC, Hu FB. Am J Clin Nutr. 2005 Mar; 81(3):555-63. http://ajcn.nutrition.org/content/81/3/555.long[↩]
- Abdominal adiposity and coronary heart disease in women. Rexrode KM, Carey VJ, Hennekens CH, Walters EE, Colditz GA, Stampfer MJ, Willett WC, Manson JE. JAMA. 1998 Dec 2; 280(21):1843-8. https://jamanetwork.com/journals/jama/fullarticle/188224[↩]
- Abdominal subcutaneous adipose tissue: a protective fat depot? Porter SA, Massaro JM, Hoffmann U, Vasan RS, O’Donnel CJ, Fox CS. Diabetes Care. 2009 Jun; 32(6):1068-75. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2681034/[↩]
- Peripheral adiposity exhibits an independent dominant antiatherogenic effect in elderly women. Tankó LB, Bagger YZ, Alexandersen P, Larsen PJ, Christiansen C. Circulation. 2003 Apr 1; 107(12):1626-31. http://circ.ahajournals.org/content/107/12/1626.long[↩]
- Studies of regional adipose transplantation reveal a unique and beneficial interaction between subcutaneous adipose tissue and the intra-abdominal compartment. Hocking SL, Chisholm DJ, James DE. Diabetologia. 2008 May; 51(5):900-2. https://www.ncbi.nlm.nih.gov/pubmed/18340430/[↩]
- Beneficial effects of subcutaneous fat transplantation on metabolism. Tran TT, Yamamoto Y, Gesta S, Kahn CR. Cell Metab. 2008 May; 7(5):410-20. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3204870/[↩]
- Evidence for a role of developmental genes in the origin of obesity and body fat distribution. Gesta S, Blüher M, Yamamoto Y, Norris AW, Berndt J, Kralisch S, Boucher J, Lewis C, Kahn CR. Proc Natl Acad Sci U S A. 2006 Apr 25; 103(17):6676-81. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1458940/[↩]
- The origins of brown adipose tissue. Enerbäck S. N Engl J Med. 2009 May 7; 360(19):2021-3. http://www.nejm.org/doi/full/10.1056/NEJMcibr0809610[↩]
- Medicine. Beige can be slimming. Ishibashi J, Seale P. Science. 2010 May 28; 328(5982):1113-4. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2907667/[↩]
- Transcriptional synergy and the regulation of Ucp1 during brown adipocyte induction in white fat depots. Xue B, Coulter A, Rim JS, Koza RA, Kozak LP. Mol Cell Biol. 2005 Sep; 25(18):8311-22.[↩]
- Murano I, Zingaretti MC, Cinti S. The adipose organ of SV129 mice contains a prevalence of brown adipocytes and shows plasticity after cold exposure. Adipocytes. 2005;1:121–130.[↩]
- Myogenic gene expression signature establishes that brown and white adipocytes originate from distinct cell lineages. Timmons JA, Wennmalm K, Larsson O, Walden TB, Lassmann T, Petrovic N, Hamilton DL, Gimeno RE, Wahlestedt C, Baar K, Nedergaard J, Cannon B. Proc Natl Acad Sci U S A. 2007 Mar 13; 104(11):4401-6. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1810328/[↩]
- Identification of inducible brown adipocyte progenitors residing in skeletal muscle and white fat. Schulz TJ, Huang TL, Tran TT, Zhang H, Townsend KL, Shadrach JL, Cerletti M, McDougall LE, Giorgadze N, Tchkonia T, Schrier D, Falb D, Kirkland JL, Wagers AJ, Tseng YH. Proc Natl Acad Sci U S A. 2011 Jan 4; 108(1):143-8. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3017184/[↩]
- Beta-catenin activation is necessary and sufficient to specify the dorsal dermal fate in the mouse. Atit R, Sgaier SK, Mohamed OA, Taketo MM, Dufort D, Joyner AL, Niswander L, Conlon RA. Dev Biol. 2006 Aug 1; 296(1):164-76. https://www.ncbi.nlm.nih.gov/pubmed/16730693/[↩]
- Inducible lineage tracing of Pax7-descendant cells reveals embryonic origin of adult satellite cells. Lepper C, Fan CM. Genesis. 2010 Jul; 48(7):424-36. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3113517/[↩]
- Anatomy of the adipose organ. Cinti S. Eat Weight Disord. 2000 Sep; 5(3):132-42. https://www.ncbi.nlm.nih.gov/pubmed/11082792/[↩]
- Beta(3)-Adrenoceptor agonists: potential, pitfalls and progress. Arch JR. Eur J Pharmacol. 2002 Apr 12; 440(2-3):99-107. https://www.ncbi.nlm.nih.gov/pubmed/12007528/[↩][↩]
- Scheffler I.E. Mitochondria. Wiley-Liss; New York: 1999.[↩]
- Uncoupling protein–a useful energy dissipator. Klingenberg M. J Bioenerg Biomembr. 1999 Oct; 31(5):419-30. https://www.ncbi.nlm.nih.gov/pubmed/10653471/[↩]
- Cold-induced recruitment of brown adipose tissue thermogenesis. Klingenspor M. Exp Physiol. 2003 Jan; 88(1):141-8. https://www.ncbi.nlm.nih.gov/pubmed/12525862/[↩][↩]
- Thyroid hormone–sympathetic interaction and adaptive thermogenesis are thyroid hormone receptor isoform–specific. Ribeiro MO, Carvalho SD, Schultz JJ, Chiellini G, Scanlan TS, Bianco AC, Brent GA. J Clin Invest. 2001 Jul; 108(1):97-105. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC209342/[↩]
- The adipose organ. Cinti S. Prostaglandins Leukot Essent Fatty Acids. 2005 Jul; 73(1):9-15. https://www.ncbi.nlm.nih.gov/pubmed/15936182/[↩]
- Mice lacking mitochondrial uncoupling protein are cold-sensitive but not obese. Enerbäck S, Jacobsson A, Simpson EM, Guerra C, Yamashita H, Harper ME, Kozak LP. Nature. 1997 May 1; 387(6628):90-4. https://www.ncbi.nlm.nih.gov/pubmed/9139827/[↩]
- Combined effects of cafeteria and tube-feeding on energy balance in the rat. Rothwell NJ, Stock MJ. Proc Nutr Soc. 1979 May 1; 38(1):5A. https://www.ncbi.nlm.nih.gov/pubmed/111255/[↩]
- Development of obesity in transgenic mice after genetic ablation of brown adipose tissue. Lowell BB, S-Susulic V, Hamann A, Lawitts JA, Himms-Hagen J, Boyer BB, Kozak LP, Flier JS. Nature. 1993 Dec 23-30; 366(6457):740-2. https://www.ncbi.nlm.nih.gov/pubmed/8264795/[↩]
- FOXC2 is a winged helix gene that counteracts obesity, hypertriglyceridemia, and diet-induced insulin resistance. Cederberg A, Grønning LM, Ahrén B, Taskén K, Carlsson P, Enerbäck S.Cell. 2001 Sep 7; 106(5):563-73. https://www.ncbi.nlm.nih.gov/pubmed/11551504/[↩]
- Adipose tissue reduction in mice lacking the translational inhibitor 4E-BP1. Tsukiyama-Kohara K, Poulin F, Kohara M, DeMaria CT, Cheng A, Wu Z, Gingras AC, Katsume A, Elchebly M, Spiegelman BM, Harper ME, Tremblay ML, Sonenberg N. Nat Med. 2001 Oct; 7(10):1128-32. https://www.ncbi.nlm.nih.gov/pubmed/11590436/[↩]
- Genetic variability affects the development of brown adipocytes in white fat but not in interscapular brown fat. Xue B, Rim JS, Hogan JC, Coulter AA, Koza RA, Kozak LP. J Lipid Res. 2007 Jan; 48(1):41-51. http://www.jlr.org/content/48/1/41.long[↩]
- Quantitative assessment of FDG uptake in brown fat using standardized uptake value and dual-time-point scanning. Alkhawaldeh K, Alavi A. Clin Nucl Med. 2008 Oct; 33(10):663-7. https://www.ncbi.nlm.nih.gov/pubmed/18806563/[↩]
- Unexpected evidence for active brown adipose tissue in adult humans. Nedergaard J, Bengtsson T, Cannon B. Am J Physiol Endocrinol Metab. 2007 Aug; 293(2):E444-52. http://ajpendo.physiology.org/content/293/2/E444.long[↩]
- Schulz TJ, Huang P, Huang TL, et al. Brown Fat Paucity Due to Impaired BMP Signaling Induces Compensatory Browning of White Fat. Nature. 2013;495(7441):379-383. doi:10.1038/nature11943. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3623555/[↩][↩]
- Body Weight Planner. https://www.supertracker.usda.gov/bwp/index.html[↩]
- ChooseMyPlate. https://www.choosemyplate.gov/[↩]
- https://supertracker.usda.gov/[↩][↩][↩]
- BMI Calculator Adults. https://www.cdc.gov/healthyweight/assessing/bmi/adult_BMI/english_bmi_calculator/bmi_calculator.html[↩]
- BMI Calculator Children. https://nccd.cdc.gov/dnpabmi/Calculator.aspx[↩]