What is carboxyhemoglobin
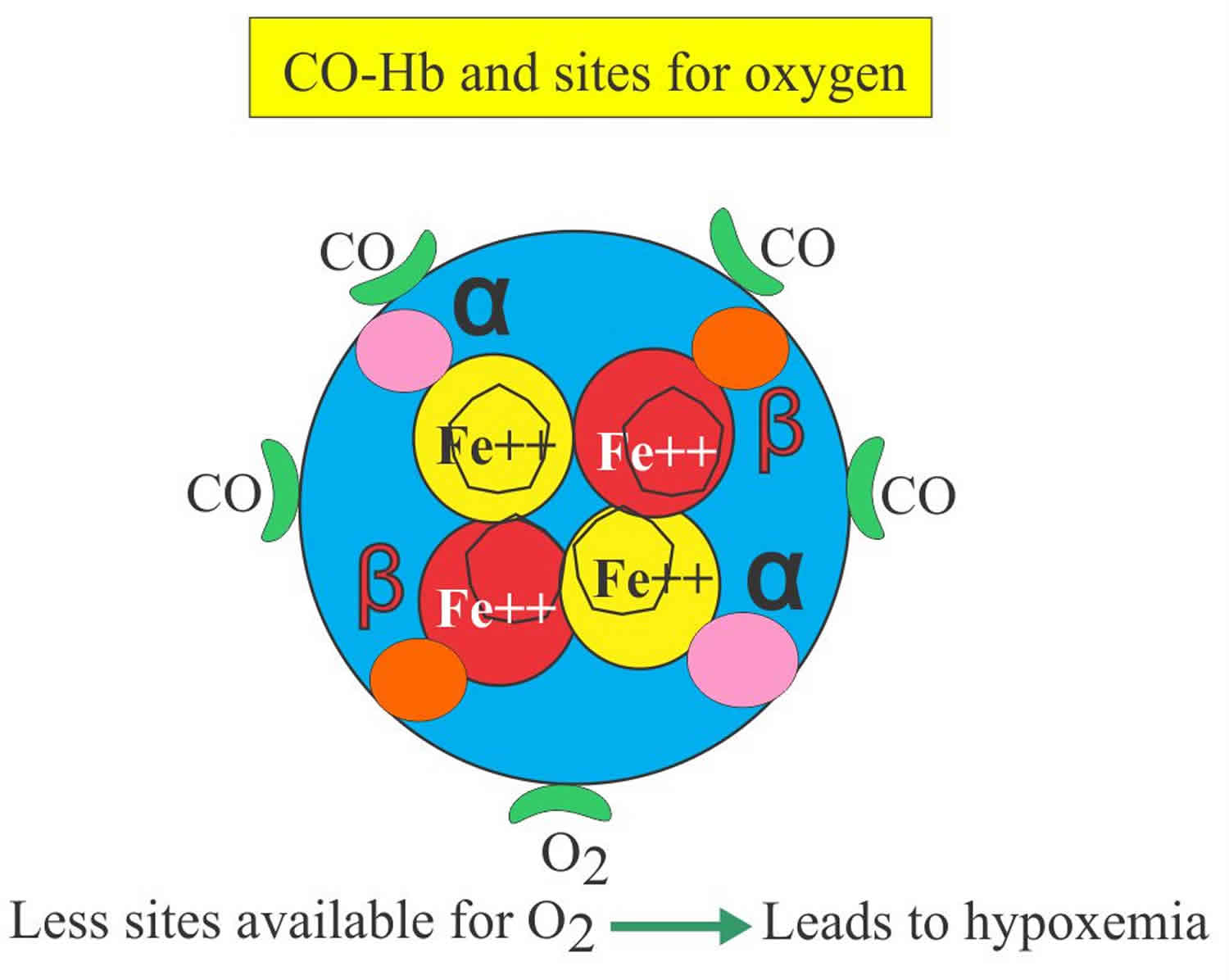
Carboxyhemoglobin (COHb) is formed by the binding of carbon monoxide (CO) to hemoglobin (Hb). High carboxyhemoglobin levels can be physiologic or pathologic. Carboxyhemoglobin is produced from hepatic metabolism of methylene chloride or as a byproduct in the process of hemoglobin degradation 1. Carboxyhemoglobin (COHb) is a stable complex of carbon monoxide (CO) that forms in red blood cells when carbon monoxide is inhaled. Carboxyhemoglobin (COHb) is unable to carry oxygen (O2), reducing oxygen (O2) availability to tissues and moving the O2 dissociation curve to the right, which causes chemical asphyxia and hypoxia 2. Carboxyhemoglobin should be measured if carbon monoxide or methylene chloride poisoning is suspected. Carboxyhemoglobin is also useful in monitoring the treatment of carbon monoxide poisoning. Carbon monoxide (CO) is an odorless, colorless gas, which can cause sudden illness and death, is produced any time a fossil fuel is burned.
The reference range of carboxyhemoglobin differs among smokers and nonsmokers, as follows:
- Nonsmokers: Up to 3% carboxyhemoglobin
- Smokers: Up to 10%-15% carboxyhemoglobin. Although the deleterious effects of carbon monoxide (CO) on the quality of blood for transfusion are known, few studies have evaluated the effect of donor smoking on the quality of packed red blood cells (RBCs) 3.
Individuals with hemolytic anemia and neonates may have carboxyhemoglobin levels of up to 5% 4.
Carbon monoxide (CO) binds to hemoglobin with 200-250 times greater affinity than oxygen 5, leading to tissue hypoxia. Carbon monoxide also causes a leftward shift of the oxyhemoglobin dissociation curve, thereby decreasing oxygen release from hemoglobin to target tissues, further exacerbating tissue hypoxia 6.
Approximately 85% of absorbed carbon monoxide binds with hemoglobin and remains in the intravascular compartment as carboxyhemoglobin. The rest of the carbon monoxide is taken up by tissues and primarily bound to myoglobin. To a lesser extent, carbon monoxide can also bind to other molecules such as cytochromes and NADPH reductase. Binding of carbon monoxide to these molecules can disrupt normal physiologic processes, including mitochondrial dysfunction 7. The brain and the heart are the most commonly affected organs in carbon monoxide poisoning.
Elimination of carbon monoxide occurs predominantly via the pulmonary circulation through competitive binding of hemoglobin by oxygen. The rate of elimination is proportionate to the degree of oxygenation, atmospheric conditions, and minute ventilation 8.
The half-life of carboxyhemoglobin in an individual breathing room air is approximately 300 minutes. This can be decreased to 80 minutes with high-flow oxygen via a nonrebreather mask 9. Carbon monoxide poisoning needs a high concentration of O2 to replace the carboxyhemoglobin. In the case of severe carbon monoxide poisoning may be treated with hyperbaric oxygen. With hyperbaric oxygen at 3 times atmospheric pressure, carboxyhemoglobin elimination half-life can be reduced even further, to about 20-30 minutes. Apart from decreasing carboxyhemoglobin half-life, hyperbaric oxygen may improve tissue oxygenation by bypassing the normal transfer of oxygen through hemoglobin 10.
The clearance of carboxyhemoglobin in methylene chloride poisoning is prolonged. The half-life of carboxyhemoglobin in this setting is approximately 13 hours because of ongoing carbon monoxide production from methylene chloride metabolism in the liver. The peak carboxyhemoglobin level can be delayed in methylene chloride poisoning 11.
Carboxyhemoglobin sources
Physiologic
Carboxyhemoglobin levels increase as a result of hemolysis. Carbon monoxide is a natural byproduct of the breakdown of protoporphyrin to bilirubin 8. In normal physiologic states, hemoglobin is metabolized by heme oxygenase into carbon monoxide, ferrous iron, and biliverdin. Heme oxygenase found in the liver and spleen is the major endogenous source of carbon monoxide, which is responsible for a small amount (< 3%) of carboxyhemoglobin found in the blood 12.
Pathologic
Elevated carboxyhemoglobin can be caused by carbon monoxide inhalation or methylene chloride toxicity, either intentional or unintentional 12.
Carbon monoxide inhalation
Carbon monoxide results from incomplete combustion of hydrocarbons. Carbon monoxide toxicity tends to occur upon exposure to a source of carbon monoxide in a poorly ventilated environment (eg, warehouses, parking garages, ice rinks, other indoor facilities). Potential sources of carbon monoxide may include motor-vehicle exhaust, ice-resurfacing machines, house fires, fireplaces, wood/charcoal camp stoves/lanterns, cooking ranges, natural gas (eg, methane, propane, kerosene) heaters, furnaces, hot water heaters, gasoline-powered equipment, and propane-powered forklifts. Compressed air for firefighters and divers has been implicated in carbon monoxide poisoning owing to faulty air compressors 13.
The most common symptoms of carbon monoxide poisoning are headache, dizziness, weakness, upset stomach, vomiting, chest pain, and confusion. carbon monoxide symptoms are often described as “flu-like.” If you breathe in a lot of carbon monoxide it can make you pass out or kill you. People who are sleeping or drunk can die from carbon monoxide poisoning before they have symptoms.
Everyone is at risk for carbon monoxide poisoning. Infants, the elderly, people with chronic heart disease, anemia, or breathing problems are more likely to get sick from carbon monoxide. Annually, there are over 40,000 cases of carbon monoxide poisoning in the United States 14. Each year, more than 400 Americans die from unintentional carbon monoxide poisoning not linked to fires, more than 20,000 visit the emergency room, and more than 4,000 are hospitalized 15.
Carbon monoxide poisoning is very unpredictable in its outcomes. Even with prompt treatment, close to 40% of patients develop residual neurocognitive impairment. Patients need to be followed up for a few months to determine if a full recovery has occurred 16.
Carbon monoxide poisoning complications:
- Amnesia
- Dementia
- Irritability
- Psychosis
- Memory loss
- Loss of executive function
- Speech deficit
- Parkinson disease
- Depression
- Cortical blindness
Methylene chloride
Methylene chloride (dichloromethane) is a solvent that can be found in paint removers. The routes of absorption can be dermal, inhalation, or oral ingestion. Methylene chloride is metabolized in the liver to carbon monoxide, which subsequently forms carboxyhemoglobin 17.
Carboxyhemoglobin test
Carboxyhemoglobin should be measured if carbon monoxide or methylene chloride poisoning is suspected. Carboxyhemoglobin is also useful in monitoring the treatment of carbon monoxide poisoning.
Carboxyhemoglobin levels can be falsely low if the patient receives oxygen prior to the test. For example, a patient who receives oxygen therapy in the ambulance prior to hospital arrival may have normal or lower-than-expected carboxyhemoglobin levels despite carbon monoxide poisoning. Although high carboxyhemoglobin levels confirm exposure to carbon monoxide, carboxyhemoglobin levels are not always predictive of symptoms or outcome 11.
An antidote for cyanide poisoning, hydroxocobalamin, is usually administered when co-intoxication with carbon monoxide is suspected. The presence of hydroxocobalamin may interfere with the co-oximetry used to detect carboxyhemoglobin, causing falsely low carboxyhemoglobin 18.
Phenobarbital, phenytoin, and progesterone enhance carbon monoxide production, which may affect the level of carboxyhemoglobin 19.
Co-intoxication with cyanide should also be considered in cases of carbon monoxide poisoning due to smoke inhalation and exposure to fires.
Carboxyhemoglobin level
- Newborn = up to 5 % saturation of total hemoglobin.
- Nonsmoker = Nonsmokers 0.5 to 3 % carboxyhemoglobin.
- Smoker Light = 2 to 5 % carboxyhemoglobin.
- Smoker heavy = 5 to 10 % carboxyhemoglobin.
- 1 to 2 packs /day 4 to 5 % carboxyhemoglobin
- >2 packs/day (heavy smoker) 8 to 9 % carboxyhemoglobin
- Critical value = >20 % carboxyhemoglobin.
- Lethal value = >50 % carboxyhemoglobin.
Carboxyhemoglobin clinical values and its effects:
| Carboxyhemoglobin % | Clinical signs and symptoms |
| 20 to 30% | Dizziness, headache, and disturbance in judgment |
| 30 to 40% | Tachycardia, hyperpnea, hypotension, and confusion |
| 50 to 60% | Coma |
| >60% | Death |
Carboxyhemoglobin treatment
The cornerstone of treatment for carbon monoxide poisoning is supplemental oxygen that should be initiated as soon as possible and continued throughout treatment 14. Patients with significant poisoning demonstrated by transient loss of consciousness, cardiac ischemia, mental status changes, tachycardia, and or hypotension, along with elevated carboxyhemoglobin levels should be treated emergently with hyperbaric oxygen. Although present in every state, only several hundred hyperbaric oxygen centers currently exist in the United States of America. The best outcomes occur when patients receive their first treatment within 6 hours of the poisoning event. Most hyperbaric physicians prescribe 3 treatments in the first 24 hours and then reassess the patient’s symptoms and response before continuing daily treatments. Despite hyperbaric therapy, up to 40% of patients can still develop chronic, neurocognitive impairment and hence, patients should be scheduled for neuropsychological evaluation approximately 1 to 2 months after recovery 20.
Alternate, more easily accessible, and useful therapies are still lacking. However, case reports and animal model studies are underway. These study therapies such as lung phototherapy.
After discharge, the patient should be followed within 4 to 8 weeks to screen for any neurological deficits. Those with intentional carbon monoxide exposure should be referred to a psychiatrist prior to discharge.
The prognosis of patients with carbon monoxide poisoning does vary on the severity, other comorbidities, and laboratory values. Individuals with documented abnormal MRI and CT findings usually have a poor prognosis. Any patient with a persistent neurological deficit also had a guarded prognosis.
- Coburn RF. Endogenous carbon monoxide production. N Engl J Med. 1970 Jan 22. 282(4):207-9.[↩]
- Carbon monoxide poisoning. Goldstein M. J Emerg Nurs. 2008 Dec; 34(6):538-42.[↩]
- Carboxyhemoglobin levels in blood donors in Bahrain. Madany IM. Sci Total Environ. 1992 May 1; 116(1-2):53-8.[↩]
- Wright GR, Shephard RJ. Physiological effects of carbon monoxide. Int Rev Physiol. 1979. 20:311-68.[↩]
- Caughey WS. Carbon monoxide bonding in hemeproteins. Ann N Y Acad Sci. 1970 Oct 5. 174(1):148-53.[↩]
- Stewart RD. The effect of carbon monoxide on humans. Annu Rev Pharmacol. 1975. 17:409-423.[↩]
- Hardy KR, Thom SR. Pathophysiology and treatment of carbon monoxide poisoning. J Toxicol Clin Toxicol. 1994. 32(6):613-29.[↩]
- Wu L, Wang R. Carbon monoxide: endogenous production, physiological functions, and pharmacological applications. Pharmacol Rev. 2005 Dec. 57(4):585-630.[↩][↩]
- Weaver LK. Clinical practice. Carbon monoxide poisoning. N Engl J Med. 2009 Mar 19. 360(12):1217-25. [↩]
- Olson KR. Carbon monoxide poisoning: mechanisms, presentation, and controversies in management. J Emerg Med. 1984. 1(3):233-43.[↩]
- Tomaszewski C. Carbon monoxide; Goldfrank’s Toxicologic Emergencies. 9th edition. ISBN 978-0-07-160593-9: Chapter 125.[↩][↩]
- Carboxyhemoglobin. https://emedicine.medscape.com/article/2085044-overview[↩][↩]
- Austin CC, Ecobichon DJ, Dussault G, Tirado C. Carbon monoxide and water vapor contamination of compressed breathing air for firefighters and divers. J Toxicol Environ Health. 1997 Dec 12. 52(5):403-23.[↩]
- Hanley ME, Patel PH. Carbon Monoxide Toxicity. [Updated 2019 Apr 18]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2019 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK430740[↩][↩]
- Carbon Monoxide Poisoning, Frequently Asked Questions. https://www.cdc.gov/co/faqs.htm[↩]
- Akcan Yildiz L, Gultekingil A, Kesici S, Bayrakci B, Teksam O. Predictors of Severe Clinical Course in Children With Carbon Monoxide Poisoning. Pediatr Emerg Care. 2018 Aug 13;[↩]
- Nager EC, O’Connor RE. Carbon monoxide poisoning from spray paint inhalation. Acad Emerg Med. 1998 Jan. 5(1):84-6.[↩]
- Livshits Z, Lugassy DM, Shawn LK, Hoffman RS. Falsely Low Carboxyhemoglobin Level after Hydroxocobalamin Therapy. N Engl J Med. September 2012. 367:1270-1271 [↩]
- Coburn RF. Enhancement by phenobarbital and diphenylhydantoin of carbon monoxide production in normal man. N Engl J Med. 1970 Sep 3. 283(10):512-5.[↩]
- Keim L, Koneru S, Ramos VFM, Murr N, Hoffnung DS, Murman DL, Cooper JS, Torres-Russotto D. Hyperbaric oxygen for late sequelae of carbon monoxide poisoning enhances neurological recovery: case report. Undersea Hyperb Med. 2018 Jan-Feb;45(1):83-87.[↩]