What is bilberry
Bilberry (Vaccinium myrtillus L.) also known as European blueberry, whortleberry, huckleberry, is any of several well-known deciduous dwarf shrubs in the genus Vaccinium (family Ericaceae), growing mostly in cool temperate regions and mountain areas of Europe, Asia and Northern America, bearing edible, nearly black berries 1. The bilberry is a small (5-9 mm in diameter) fruit, bluish black in color, with many seeds. Bilberry grows mostly in cool temperate regions and mountain areas of Europe and Asia. The bilberries are a rich source of various phenolic compounds and bilberry is one of the richest natural sources of anthocyanins 2, 3. This flavonoid subclass provides the main red, violet and blue pigments in flowers and fruits, in which they act as insect and animal attractants, possess protective roles against various biotic and abiotic stresses and also provide benefits for human health 4. Human-induced changes in abiotic environmental factors such as atmospheric CO2 and ozone (O3) levels, UV light, changes in precipitation patterns or temperature may directly affect the concentration of secondary chemicals in plants, which in turn may influence levels of herbivory or pathogen attack. For example, UV-B radiation modifies the production of secondary metabolites with photoprotective qualities 5, such as anthocyanins, isoflavonoids, and flavonol glycosides. The induction of UV-absorbing chemicals is shared with plant responses to other stresses, such as herbivore or pathogen attack, and this induction may act either positively or negatively on the levels of phytochemical production. According to the phytochemical co-evolution theory, the secondary metabolites are likely the most important mediators of plant-insect interactions. Thus, both plants and insect herbivores have evolved leading to the plant defense (i.e., plant secondary metabolites) and herbivore offense (i.e., detoxification ability) 6. The color of bilberries varies normally from blue to almost black. As rare variants, bilberry mutants with white or pink fruits have been found in nature (Figure 1 B and C). In these mutants, the taste and the size of the fruit are the same as in wild-type bilberry and the color of the berries is the only difference. Therefore, it is assumed that the change in color in these bilberry mutants would be due to the mutation in structural or regulatory genes involved in anthocyanin biosynthesis. In fruits, anthocyanins are predominantly found in the vacuoles of skin, although anthocyanins can also be found in the pulp in some berries 7. Researchers 8, 9, 10 are interested in bilberry in large part because its berries have a high concentration of antioxidants called anthocyanins, which some studies suggest may have health benefits. Bilberry is classified as a Class 1 herb by the American Herbal Products Association 11, meaning it can be safely consumed when used appropriately. No mutagenic activity has been reported, and there are no cited contraindications to its use 11. Bilberry is sold as fresh, frozen, and dried whole berries, as well as in the form of preserves, jams, and juices, and, increasingly, liquid or powdered concentrates are sold as food supplements. Bilberry extract is sold in tablets, capsules, and drops, and the berries are sold dried and as a powder. Bilberry leaves are made into teas. Bilberries and bilberry leaves have been used for medicinal purposes since the Middle Ages for a variety of conditions, including diarrhea, scurvy, infections, burns, and diabetes. During World War II, British pilots ate bilberry jam, thinking it would improve their night vision. However, the hypothesis that bilberry anthocyanins improves normal night vision is not supported by evidence from rigorous clinical studies 12. A double-blind, placebo-controlled, crossover study conducted on U.S. Navy SEAL personnel did not improved nighttime visual acuity, faster adjustment to darkness, and faster restoration of visual acuity after exposure to glare 13. Fifteen male subjects given 160 mg bilberry extract (standardized to 25-percent anthocyanins) three times daily for three weeks, showed no significant improvement in either night visual acuity or night contrast sensitivity compared to placebo 13. Today, bilberry is used as a dietary supplement for cardiovascular conditions, diarrhea, urinary tract infections, eye problems, diabetes, and other conditions. Despite bilberry widespread promotion by supplement companies and use for many diseases and health conditions, there are very few high-quality clinical trials (studies in humans) of bilberry supplements and based on these results there’s not enough scientific evidence to support the use of bilberry for any health conditions.
Figure 1. Bilberry from flower to ripe fruit
Notes: (A) Bilberry from flower to ripe fruit. Six different stages were examined in this study. Stages: 1, flower; 2, small-sized green fruits; 3, middle-sized green fruits; 4, half-expanded, just after coloring began; 5, nearly expanded, half-colored, red fruits; 6, fully colored, blue, ripe fruits. (B) The color mutation of bilberry with white berries. (C) The color mutation of bilberry with pink berries.
[Source 14]Figure 2. Bilberry
Bilberry Anthocyanins
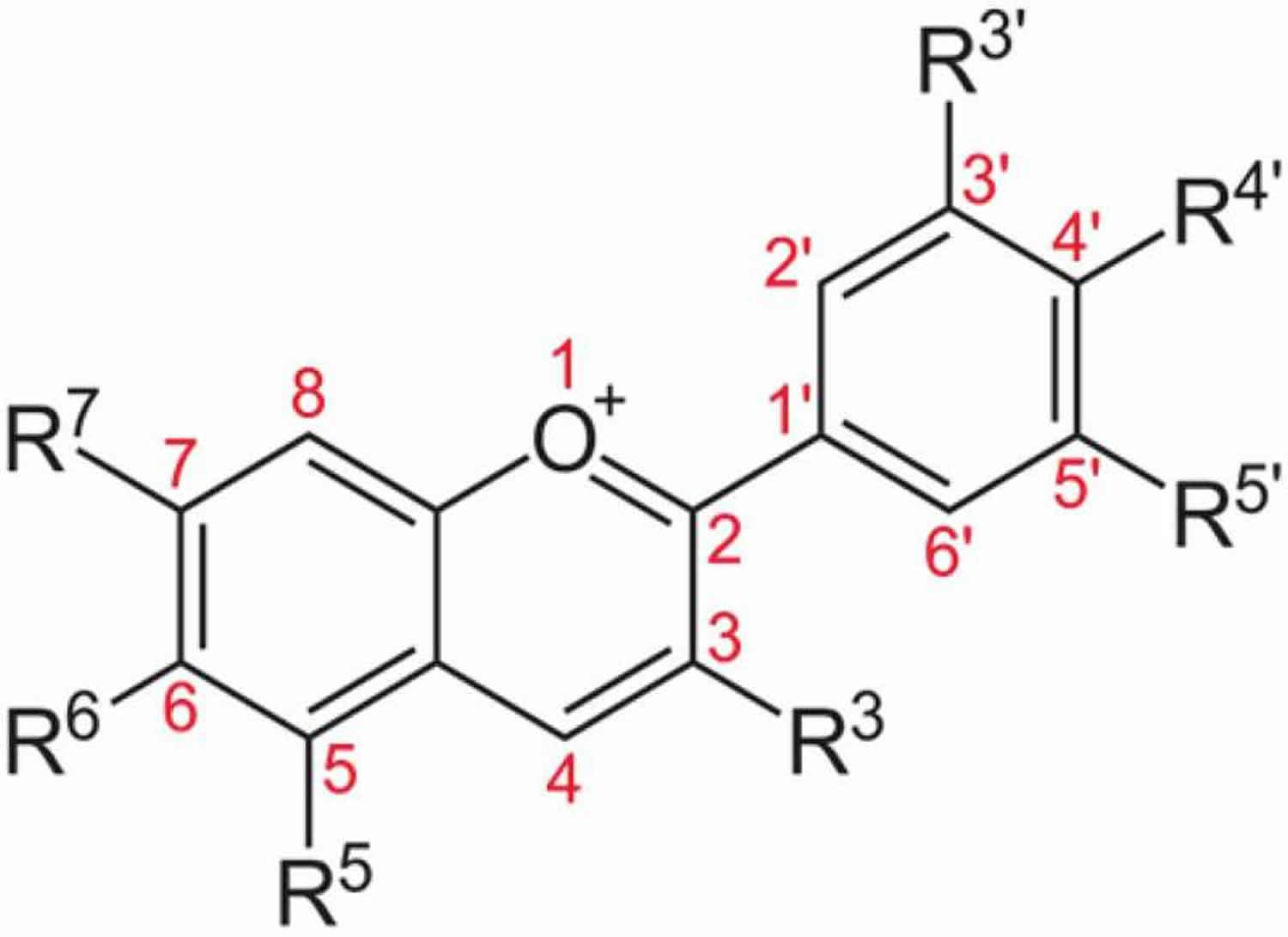
Anthocyanins are blue, red, or purple pigments found in plants, especially flowers, fruits, vegetables and tubers 15. In acidic condition, anthocyanin appears as red pigment while blue pigment anthocyanin exists in alkaline conditions. Anthocyanin is considered as one of the flavonoids although it has a positive charge at the oxygen atom of the C-ring of basic flavonoid structure. It is also called the flavylium (2-phenylchromenylium) ion. The general molecular structure of anthocyanin is shown in Figure 3. The stability of anthocyanin is dependent on pH, light, temperature, and its structure 16. The bilberry’s anthocyanins content increases as the fruit ripens, while the reverse is true of its leaf constituents 17.
Bilberry has higher anthocyanin content compared to other types of berries, such as strawberry, cranberry, elderberry, sour cherry, and raspberry 18. The total anthocyanin content of bilberry is generally in the range of 300-700 mg/100 g fresh fruit, although this range varies with cultivar, growing conditions, and degree of ripeness of the berry 19. Along with anthocyanins, 100 g of fresh bilberry contains small quantities of vitamin C (3 mg), quercetin (3 mg), and catechin (20 mg) 20.
Although the function of anthocyanin pigments in plants is thought to be mainly that of attracting animals, insects, and birds for pollination or seed dispersal, anthocyanins may help in cold tolerance as well as antimicrobial and antioxidant defense of plant tissues 21. The anthocyanin content of berries varies across species and also depends on environmental factors, such as the amount of solar radiation, temperature, and soil content of nitrogen and phosphorus. The cultivation technique is also a factor affecting the total phenolic level of berries. Anthocyanins are found mainly in the skin of the berry, and damage to the fruit skin caused during harvesting decreases anthocyanin content 11. Degrees of ripeness of berries also contribute to variation in total phenolic content—although the concentration of phenolic compounds is usually higher in unripe berries than in mature fruits, anthocyanins accumulate during bilberry maturation 11. Anthocyanins have powerful antioxidant properties, and the content of anthocyanin pigment directly correlates with antioxidant activity of plants 22. In addition to their antioxidant effects, anthocyanins have been reported to stabilize DNA, modify adipocyte gene expression, improve insulin secretion and sensitivity, and have antiapoptotic, anti-inflammatory, and antibacterial effects 23. These numerous and potentially highly beneficial effects of anthocyanins make foods rich in these compounds, such as bilberry, potential candidates as “functional foods” and phytotherapeutics 18.
Figure 3. Basic anthocyanin structure
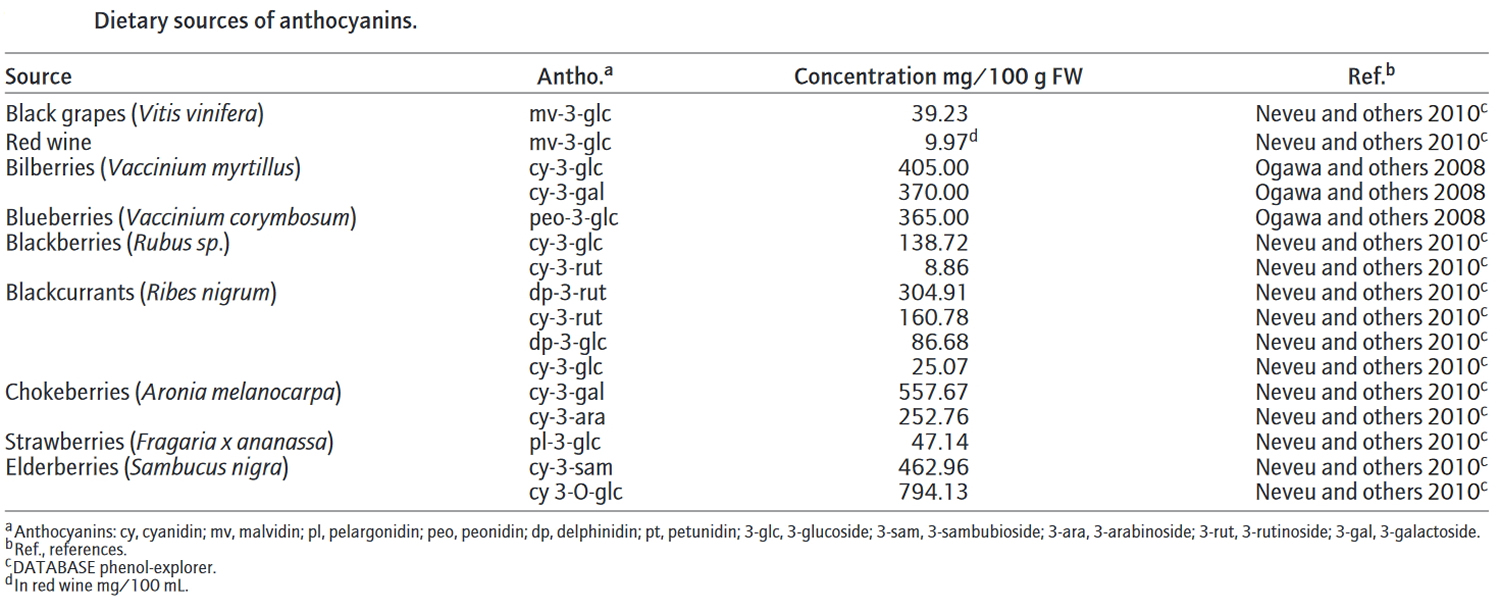
Figure 4. Dietary sources of anthocyanins
[Source 24]Table 1. Main Anthocyanins in Bilberry and Their Relative Concentrations
| Anthocyanin | Mean Content (%) |
|---|---|
| Delphinidins (total) | 15.17 |
| Delphinidin-3-O-glucoside | 5.81 |
| Delphinidin-3-O-galactoside | 5.04 |
| Delphinidin-3-O-arabinoside | 4.32 |
| Cyanidins (total) | 8.36 |
| Cyanidin-3-O-glucoside | 3.42 |
| Cyanidin-3-O-galactoside | 2.75 |
| Cyanidin-3-O-arabinoside | 2.19 |
| Petunidins (total) | 6.64 |
| Petunidin-3-O-glucoside | 3.67 |
| Petunidin -3-O-galactoside | 1.89 |
| Petunidin-3-O-arabinoside | 1.08 |
| Malvidins (total) | 5.43 |
| Malvidin-3-O-glucoside | 3.35 |
| Mavlidin-3-O-galactoside | 1.27 |
| Malvidin-3-O-arabinoside | 0.81 |
| Peonidins (total) | 1.87 |
| Peonidin-3-O-glucoside | 1.31 |
| Peonidin-3-O-galactoside | 0.34 |
| Peonidin-3-O-arabinoside | 0.22 |
In a study by Muller et al 25 on the anthocyanin content of commercially available bilberry juices and fresh fruits that were quantified by using high performance liquid chromatography with an ultra‐violet detector. Delphinidin‐3‐O‐glucopyranoside, delphinidin‐3‐O‐galactopyranoside, and cyanidin‐3‐O‐arabinopyranoside were the major anthocyanins found in bilberry juices, nectar, and fresh bilberries. Up to 1017 mg anthocyanins per 100 g fresh weight (7465 mg/100 g dry weight) were detected in bilberries. Anthocyanin concentrations of bilberry juices (1610 mg/L to 5963 mg/L) and bilberry nectar (656 mg/L to 1529 mg/L). This study has also demonstrated that commercially available juices produced from bilberries contain much higher anthocyanin concentrations than those from blueberries.
Although most attention has been focused on the antioxidant properties of anthocyanins in relation to health benefits of bilberry, the effects are likely to extend beyond simple antioxidant action to involve cell-signaling pathways, gene expression, DNA repair, and cell adhesion, as well as antineoplastic and antimicrobial effects 26. Commercial bilberry products are often standardized to a 25% anthocyanidin content (equivalent to 36% anthocyanins); but this content can vary greatly 27. Recommended daily dosages also vary greatly, for example, 20-60 g of dried berries and 160-480 mg of powdered extract 11.
Bioavailability and distribution of anthocyanins
With respect to anthocyanins from bilberry, human data are lacking, although some animal studies have been performed. In rats, the plasma total anthocyanin concentration was 26 mg/L at 1 hour after intraperitoneal administration of bilberry anthocyanins. Anthocyanins were detectable also in tissues collected at the same time, at concentrations of 12-79 μg/g tissue. Kidney had three fold higher anthocyanin content than plasma, and skin had 1.5-fold higher content than plasma 28. After oral administration (400 mg/kg) of bilberry anthocyanins to rats, a maximal plasma concentration of 2.47 mg/L was reported at 15 minutes, with bioavailability estimated at 1.2% 11. In a more recent animal study of distribution and excretion of bilberry anthocyanins 29, the plasma concentration of mice was shown to peak at 15 minutes postingestion of an ethanol extract of fresh bilberries, showing a sharp decrease thereafter, and the renal excretion of anthocyanins represented 1.88% of the dose ingested. Thirteen anthocyanins were detected in the bilberry extract, and the main anthocyanins in plasma at 60 minutes postingestion were malvidin-3-glucoside and malvidin-3- galactoside 29. In mice fed bilberry extract for 2 weeks, plasma levels reached a maximum of 0.26 μmol/L and anthocyanins were found only in liver, kidney, testes, and lung. No anthocyanins were found in brain, heart, muscle, eyes, or white fat. The researchers concluded that bilberry anthocyanins are absorbed but are taken up by specific organs 29. However, there may be differences in the tissue distribution of anthocyanins across different species, as brain uptake of anthocyanins has been reported in a rat model 28.
Bilberry vs Blueberry
The bilberry bush is a relative of the blueberry, but the bilberry tree produces single paired fruit, not clusters. Bilberry is sometimes called blueberry because both have similar appearance and are close relatives, but the true blueberry is native to the United States 30. Bilberries (Vaccinium myrtillus) are smaller than Blueberries (Vaccinium corymbosum) with a fuller, distinctive taste. The pulp inside a Bilberry is a dark red or purple color whereas a blueberry has a soft light shade of green. Bilberries are harder to grow, they require acidic, nutrient poor soil in subarctic regions. Bilberries are considered to be higher in antioxidants than Blueberries. Up to 438 mg anthocyanins per 100 g fresh weight (2762 mg/100 g dry weight) were detected in blueberries from various sources, whereas bilberries contained a maximum of 1017 mg anthocyanins per 100 g fresh weight (7465 mg/100 g dry weight). Anthocyanin concentrations of bilberry juices (1610 mg/L to 5963 mg/L) and bilberry nectar (656 mg/L to 1529 mg/L) were higher than those of blueberry juices (417 mg/L) and nectar (258 mg/L to 386 mg/L).
Bilberry extract
Bilberry extract is an antioxidant-rich supplement that contains various anthocyanins. Bilberry extract is not only believed to ameliorate ocular symptoms but has also been used in clinical trials for eye fatigue 31. To examine the effect of a dietary supplement containing bilberry extract on eye fatigue induced by acute video display terminal loads. Ozawa et al 31 enrolled two hundred eighty-one office workers aged 20–40 years that used video display terminals. Bilberry extract (480 mg/day) supplementation improved some of the objective and subjective parameters of eye fatigue induced by video display terminal loads. It can reduce reactive oxygen species, as shown in test tube experiments 32, and suppress the pathogenesis of innate retinal inflammation and diabetes in animal models.
Bilberry benefits in animal tests
- Bilberry protects against doxorubicin (a commonly used chemotherapeutic agent) induced cardiotoxicity in rats. This can be attributed, at least in part, to its antioxidant activity 33.
- Bilberry prevented the rat’s ovarian tissue from the toxic influence of cisplatin (potent chemotherapeutic agent). Although the results suggested that bilberry can reduce cisplatin ovarian toxicity, the degree of protection it provides is limited 34.
- Animal studies (rabbits and rats have demonstrated bilberry extract to be of benefit in improving vascular tone, blood flow and vasoprotection 35.
- Bilberry improved serum lipid profiles and promoted a hypocholesterolemic effect in mice, which protected against hypercholesterolemia and prevented atherosclerosis 36.
- In type-2 diabetic mice, anthocyanins in bilberry have been suggested to reduce blood glucose levels and enhance insulin sensitivity 37.
- Anthocyanin-rich bilberry extracts reduced adenoma development in ApcMin mice 38, a model of human familial adenomatous polyposis coli.
- Bilberry extracts have demonstrated anti-inflammatory properties in animals 39.
- Bilberry anthocyanins may offer therapeutic benefit in prevention of cataracts. Animal studies show diets high in anthocyanoside flavonoids retard the development of cataracts in rats 40.
- Research demonstrates that oral administration of bilberry leaf decoctions reduce hyperglycemia in dogs, even in the presence of concurrently injected glucose. This effect is attributed to the myrtillin anthocyanins, apparently the most active hypoglycemic component 41. In addition, bilberry anthocyanins enhance collagen integrity, stabilize capillary permeability, and inhibit sorbitol accumulation, thus providing protection against vascular and neurological sequelae of diabetes.
- Bilberries ameliorated type 1 pro-inflammatory responsiveness induced by high-fat diet in mice 42. Bilberries also prevented the progression of obesity associated long term increase in systolic blood pressure in mice.
- Bilberries or pure anthocyanins or their extracts have shown anti-obesity effects in mouse models or reduced obesity-associated symptoms 43, but the findings with whole berries seem not to be as consistent as with extracts 44.
- In test tube study and animal tumorigenic models have demonstrated that berry anthocyanins have cancer-preventive and -suppressive activity via antioxidant activity; antiproliferative, apoptotic, anti- angiogenic, and anti-inflammatory effects; and induce the antioxidant response element with consequent phase II enzyme induction and other cytoprotective effects 45. Other mechanisms of genoprotection may involve direct interaction of anthocyanins with DNA.
- In an animal study, the number of intestinal adenoma decreased significantly by 15-30% in rats with genetic colon adenoma that were fed a high dose of bilberry extract (10% w/w in diet, supplying approximately 5.5 g anthocyanin per kilogram per day 46. Similar findings were shown with a lower dose of a commercial bilberry extract (mirtoselect®, at a daily dose of 0.3% w/w diet, supplying approximately 0.5 g anthocyanin per kilogram per day) 47. This dosage is equal to approximately 740 g of fresh bilberries in terms of human intake.
- An extract of bilberry was shown to dose-dependently inhibit cell growth and promote induction of apoptosis in cultured breast cancer cells (MCF7-GFP-tubulin breast cancer cells) 48. At higher doses (0.5–1.0 mg/mL), the bilberry extract arrested the cell-cycle at the G(2)/M phase and inhibited microtubule polymerization 48.
- In primary cultures of rat hepatocytes (liver cells), it was found that bilberry extract protected cells against oxidative damage 49. Bilberry or anthocyanin extracts of bilberry protects rat liver microsomes against oxidative damage and apolipoprotein B against ultraviolet (UV)-induced oxidative fragmentation 50. However, animal studies show conflicting results. In a rat study, no significant change of urinary 8-OHdG (also known as 8-oxodG, and a biomarker of oxidative stress) 51, was reported after 14 weeks of supplementation with an anthocyanin-rich extract from mixed berries including bilberry 52. In other studies 53 a significant decrease of malondialdehyde (a biomarker of lipid peroxidation) in brain was seen in OXYS rats fed with bilberry extract (2 g of dried aqueous extract including 0.35 g/kg diet) 54.
- In an animal study, plasma triglycerides were decreased after feeding rats with an extract of bilberry leaves (3 g/kg/day) for 4 days 55. However, it is noted that the leaves and not the bilberries were used and the effects may not have been related to anthocyanins, which are found mainly in the deeply colored berries.
- In rats that were fed bilberry anthocyanins for 12 days prior to inducing hypertension, permeability of the blood-brain barrier was kept normal and there was limited increase in the vascular permeability of the skin and aorta wall 56.
- Cyanidin-3-glucoside has been shown to suppress the development of obesity in mice fed a high-fat diet and to regulate human adipocyte function 57. In a follow-up study, Tsuda and coworkers 58 reported that a bilberry extract added to the diet of diabetic mice (27 g/kg diet, which gave an anthocyanin content of 10 g/kg diet) lowered serum glucose and improved insulin sensitivity. There were no differences in body weight or serum adiponectin levels between the bilberryfed and the control animals, but the antidiabetic effects of the bilberry extract were associated with AMPK activation in white adipose tissue and skeletal muscle and liver, and were accompanied by increased glucose transporter 4 (GLUT 4) in white adipose and skeletal tissue and lower hepatic gluconeogenesis 58.
- Bilberry has also been reported to improve visual function in animal models 59. Bilberry extract treatment attenuated all the observed photo-induced photoreceptor degeneration, including the increased reactive oxygen species levels in the retina of photo-stressed mice 60. Furthermore, the reactive oxygen species levels in the bilberry extract-treated retinas were almost equal to those observed in the retinas of light-exposed mice treated with a dosage of N-acetyl-L-cysteine (a well-known antioxidant) previously reported to suppress photo-induced photoreceptor degeneration 61.
- In a study with cultured retinal ganglion cells, bilberry anthocyanosides inhibited chemical-induced cell damage and radical activation, and this neuroprotective effect, which may have been an antioxidant-related effect, was also seen in vivo when bilberry anthocyanosides (100 μg/eye) were injected into the vitreous of mice 62.
- A commercial bilberry extract (Myrtocyan® at 200 mg/kg daily for 5 days by intraperitoneal administration) given to rats was reported to increase triiodothyronine transport to different regions of the brain, and bilberry is reported to promote short-term memory, vision, and control of sensory input in animals 63. However, most studies on the neuroprotective effects of berries and their constituents have been performed on cultured cells and in animals, and cognition-related studies using bilberry specifically are lacking.
- Bilberry and other berry fruits, as well as purified berry phenolics, have been reported to show direct antimicrobial effects against human pathogens, including Salmonella and Staphylococcus aureus 64. Interestingly, pure phenolic compounds (as opposed to berry extracts) were found to inhibit only gram-negative bacteria (which include Salmonella species and Escherichia coli), and the effects were related to the degree of hydroxylation of the pure phenolic compounds 64.
Bilberry health benefits
Bilberries are an important source of anthocyanins in the diet, and are also rich in other phytochemicals such as procyanidins, phenolic acids and flavonoids 65. Bilberry contains 15 different anthocyanins, including 5 anthocyanidins (delphinidin, cyanidin, malvidin, petunidin, and peonidin), and 3 sugars (glucose, galactose, and arabinose) 66.
Antioxidant properties
The bilberry extract has potent antioxidant properties 67; inhibits platelet aggregation 68; and improves vascular tone, blood flow, and vasoprotection 69. Supplementation with bilberry juice resulted in significant decreases in plasma concentrations of C-reactive protein (CRP), interleukin (IL)-6, IL-15, and monokine induced by INF-γ in 31 human subjects with with elevated levels of at least one risk factor for cardiovascular disease 70. In contrast to the promising effects seen in animal studies, no effect on lipid peroxidation was seen in human volunteers after supplementation with mixed vegetables and fruits including bilberries 71. In general, most controlled human supplementation studies are performed on healthy subjects, and they have limited potential to respond in relation to biomarker changes. Better target groups for looking at antioxidant-related effects are those humans under higher oxidative stress, such as the elderly, those at elevated risk of heart disease, or diabetic patients. To date, there is only one published report of the effects of bilberry supplementation on antioxidant status and oxidative stress in human subjects 72. Subjects (n = 31) with at least one risk factor for cardiovascular disease were supplemented with 330 mL/day bilberry juice (diluted to 1 L with water) for 4 weeks. However, results showed no significant changes in biomarkers of antioxidant status or oxidative stress in these subjects, compared to 31 control subjects 72.
Two points of caution are offered here with respect to antioxidant content and action of bilberry anthocyanins. First, many of the bilberry extracts used in trials have not been characterized or standardized to anthocyanin content. Variation in anthocyanin content affects the antioxidant content of extracts. In test tube testing of the antioxidant capacity of commercial bilberry products by a research group showed surprising results 73. Using the ferric reducing/antioxidant power ability assay 74, the total antioxidant capacity of 11 commercial powdered bilberry extracts from various countries of origin were tested. Values ranged from <20 to >2000 μmol/g 73. There was a strong correlation between the ferric reducing/antioxidant power value and the anthocyanin content. Interestingly, whereas the anthocyanin content of commercial bilberry products is assumed to be standardized to 250 mg/g, or at least to be high, only 5 of the 11 commercial products had any stated anthocyanin content and 3 of the 5 had stated values of <80 mg/g. Furthermore, the actual anthocyanin content of all but one product was <60 mg/g, with concomitantly low ferric reducing/antioxidant power values 73. In one product with a stated content of 250 mg/g anthocyanins, the measured content was <10 mg/g. Second, in vitro antioxidant activity may not reflect in vivo action 75. Molecular action of antioxidant phytochemicals may be independent of or indirectly related to antioxidant activity, may change with concentration, or may manifest only in the presence of other bioactives or under particular redox conditions 76. Therefore, although anthocyanins are potent antioxidants in vitro, and fresh bilberry is a rich source of these, it cannot be assumed that all commercial bilberry products contain significant amounts of bilberry anthocyanins or that absorbed anthocyanins act directly as antioxidants in vivo. In the future, trials performed with bilberry extracts must use a standardized product of known anthocyanin content, and studies looking for health benefits should include a wide range of biomarkers of both antioxidant and nonantioxidant effects.
Cardiovascular disease
Atherosclerosis, the main underlying factor in cardiovascular disease, is an inflammatory process associated with oxidative processes in and damage to the vascular endothelium 77. Therefore, the anti-inflammatory and antioxidant effects of anthocyanins are of relevance to potential cardioprotective effects of bilberry and other berries. Antihypertensive, lipid-lowering, hypoglycemic, and antiobesity effects would also be cardioprotective 78. A controlled human supplementation trial showed decreased concentration of inflammatory biomarkers in the plasma of 31 subjects who took bilberry juice for 4 weeks 72. Most notably, significant decreases were seen in plasma levels of high-sensitivity C-reactive protein (hsCRP), a sensitive biomarker of subclinical inflammation and a predictor of cardiovascular disease, and the proinflammatory cytokine interleukin 6 (IL-6). No significant effects were seen on plasma cholesterol, triglycerides, or uric acid concentrations 72. In a controlled human study of 35 subjects who took 100 g of whole bilberries each day, platelet function, blood pressure, and high-density lipoprotein (HDL)-cholesterol were all improved 78. However, in addition to the daily bilberry supplement, the subjects took a mixture of various berries during the study. A study of 23 healthy volunteers given a mixture of grape seed, pine bark, bilberry, and red wine for 4 weeks showed an insignificant decrease in acute impairment in endothelial function caused by a high-fat meal 79. In another human study 80, mixed anthocyanins from bilberry and blackcurrant (Ribes nigram) were given as an extract (320 mg/day) for 12 weeks to 60 middle-aged dyslipidemic Chinese subjects. Results showed significant improvements in low-density lipoprotein (LDL)- “bad” cholesterol (average decrease of approximately 14%) and HDL- “good” cholesterol (average increase of approximately 14%). No significant changes were seen in plasma total cholesterol, triglycerides, or apolipoproteins, but the plasma concentration of cholesteryl ester transfer protein was significantly decreased, and the change in cholesteryl ester transfer protein correlated with the changes in lipids. An in vitro study by the same group showed that cyanidin-3-O-glucoside lowered cholesteryl ester transfer protein activity in human HepG2 cells, and the researchers concluded that the bilberry and blackberry anthocyanin mixture improved lipids in their human subjects by inhibiting cholesteryl ester transfer protein activity and changing cellular cholesterol efflux 80.
Regarding blood pressure and vascular health, bilberry fruit anthocyanins have been reported 50 to inhibit smooth muscle contraction and platelet aggregation. These are potentially antithrombotic and antihypertensive effects and have cardioprotective effects. Possible antihypertensive effects of bilberry are also suggested by the finding of inhibition of angiotensin-converting enzyme (ACE) activity in cells in vitro (Persson, Persson, and Andersson 2009). A significant, dose-dependent inhibition of ACE activity was seen in endothelial cells from human umbilical veins that had been incubated in bilberry extract (0.00625–0.1 mg/mL) for 10 minutes 81. Interestingly, individual anthocyanidins (cyanidin, delphinidin, and malvidin) had no inhibitory effect, and the researchers concluded that ACE-inhibition seemed to be dependent on the specific mixture of anthocyanins in bilberry 81.
Bilberry for eyes
Bilberry has a long history of use for eye disorders and in promoting vision. There have been numerous studies of the effects of bilberry on various aspects of vision and ocular disorders, including cataract, retinopathy, macular degeneration, and night vision 82. Many studies have shown positive effects, including improvement in retinal abnormalities, increased capillary resistance, slowing of progression of lens opacity and myopia, and improved dark adaptation. For example, in a study of 50 patients with mild senile cataract, 4 months of supplementation with bilberry anthocyanins plus vitamin E was reported to have a 97% success rate in preventing cataract progression 83. A double-blinded, placebo-controlled study reported that in six subjects who were given bilberry anthocyanins, dark adaptation at 1 hour and hours postingestion was faster (6.5 minutes) compared to six control subjects (9 minutes) 63. However these, and many other, studies, were small; used mixed supplements, such as bilberry plus other berries or bilberry combined with vitamin E; or were uncontrolled. Canter and Ernst 12 reviewed 30 published trials of bilberry-extracted anthocyanins on lowered light and night vision. Only 12 of the 30 studies were placebo controlled, and the conclusion was that there was insufficient rigorous evidence to recommend the use of bilberry for improving night vision 12. Healthy subjects with normal or above average eyesight were tested in 11 of the 12 trials. The hypothesis that bilberry anthocyanins improves normal night vision is not supported by evidence from rigorous clinical studies 12. A double-blind, placebo-controlled, crossover study conducted on U.S. Navy SEAL personnel did not improved nighttime visual acuity, faster adjustment to darkness, and faster restoration of visual acuity after exposure to glare 13. Fifteen male subjects given 160 mg bilberry extract (standardized to 25-percent anthocyanins) three times daily for three weeks, showed no significant improvement in either night visual acuity or night contrast sensitivity compared to placebo. A randomized, double-blinded, placebo-controlled study showed that symptoms of asthenopia and contrast sensitivity for 22 of the 30 subjects studied (73%) improved significantly after 4 weeks of 100 mg/day of purified anthocyanin (85% anthocyanoside oligomers) 84.
Age-related vision loss, mainly due to senile cataract and macular degeneration, affects the quality of life of virtually all elderly persons. Diabetic retinopathy is highly prevalent in those who have had diabetes for 10 or more years and is a leading cause of blindness in developed countries. There is sufficient evidence from animal and cell-culture studies and small human trials to warrant better designed and controlled human trials of bilberry in helping to address the huge clinical problem of age and diabetes-related vision loss.
Colorectal cancer
A 2009 pilot study, Thomasset and Berry et al 85 investigated mirtocyan, an anthocyanin-rich standardized bilberry extract on 25 colorectal cancer patients. An anthocyanin-standardized extract of bilberry (mirtocyan®, supplying between 0.5 and 2 g of anthocyanins per day) was given for 7 days prior to surgery 85. After 7 days, bilberry anthocyanins and their glucuronide and methyl metabolites were detected in plasma and also in tumor tissue (179 ng/g tissue at the highest bilberry extract dose), and plasma levels were found to be related to dose. The tumor tissue showed a 7% decrease in proliferation compared to pre-bilberry values, and there was a small but significant decrease in plasma insulin-like growth factor-1 (IGF-1) seen with the lowest dose 85. These preliminary data from human study provide important and clear support for further clinical studies of bilberry anthocyanins in cancer chemoprevention. In conclusion, repeated administration of bilberry anthocyanins exerts pharmacodynamic effects and generates concentrations of anthocyanins in humans resembling those seen in ApcMin mice, a model of familial adenomatous polyposis (FAP) adenomas sensitive to the chemopreventive properties of anthocyanins. Studies of doses containing <0.5 gram bilberry anthocyanins are necessary to adjudge whether they may be appropriate for development as colorectal cancer chemopreventive agents 85. There has been no follow up study since that 2009 pilot study involving patients with colorectal cancer.
Ulcerative colitis
In an open pilot trial 86 with a total follow-up of 9 weeks the effect of a daily standardized anthocyanin-rich bilberry preparation was tested in 13 patients with mild to moderate ulcerative colitis. The bilberry preparation was specifically manufactured for the purpose of that study under highly standardized conditions with dried, sieved bilberries (59.63%) and concentrated bilberry juice (25.90%) as the main components. The preparation was packed in small aluminum trays, each containing 40 g. Patients received a daily bilberry preparation dose of 160 g (4 trays per day), corresponding to 95 g dry weight (corresponding to an amount of around 600 g of fresh fruit, assuming a water content in fresh bilberries of 80–85%) for a total of six weeks. The anthocyanin content of the preparation was determined by repetitive high performance liquid chromatography (HPLC) scans, revealing highly reproducible values with a mean content of 210 mg anthocyanins per tray, corresponding to an average anthocyanin dose of 840 mg per day 86. Patients were asked to refrain from food or liquid intake within one hour prior to and after bilberry ingestion. Time and daily dose of ingestion had to be recorded in the patient’s diary. Clinical, biochemical, endoscopic and histologic parameters were assessed. At the end of the 6 week treatment interval 63.4% of patients achieved remission, the primary endpoint, while 90.9% of patients showed a response. These results clearly indicate a therapeutic potential of bilberries in ulcerative colitis. The main limitation of that study is the lack of a placebo group. There is a well-known high placebo response rate in clinical studies of ulcerative colitis. The average placebo rates of remission and response in ulcerative colitis are 13-28% has been described in a meta-analysis including 40 clinical studies in ulcerative colitis patients 87. Further studies on mechanisms and randomized clinical trials are warranted before bilberry extract can be used to treat mild to moderate ulcerative colitis.
Metabolic disease
Obesity is a strong predisposing factor for type 2 diabetes. Berry polyphenols may help prevent obesity by inhibiting digestive enzymes, such as lipase, thereby lowering fat absorption 88. Human preadipocytes were collected from subcutaneous adipose tissue, cultured, and differentiated into adipocytes before being treated with anthocyanins for 24 hours. Adiponectin, an anti-inflammatory cytokine, was upregulated, and there was downregulation of the proinflammatory cytokine IL-6 and also of the plasminogen activator inhibitor-1 (PAI-1); the anthocyanin treatment also activated adenosine monophosphate (AMP)-activated protein kinase (AMPK) in adipocytes without increasing the AMP/adenosine triphosphate (ATP) ratio 89. Together, these changes indicate a role for anthocyanins in preventing metabolic syndrome, an increasingly common condition associated with insulin resistance, hypertension, and dyslipidemia that often progresses to type 2 diabetes. Although there are some published human studies of the hypoglycemic effects of berries (e.g., cranberry, chokeberry), strong evidence from human trials is lacking 90. To our knowledge, there are no published controlled human studies with bilberry on diabetes patients. The two published human supplementation studies with bilberry 80, 72 studied subjects at elevated risk of cardiovascular disease, but they were not diabetic. It is unlikely that significant effects of bilberry would be seen in subjects with normal glucose tolerance No differences were seen in plasma glucose levels in 60 nondiabetic dyslipidemic subjects who took a mixed bilberry and blackcurrant anthocyanins supplement (120 mg/day anthocyanins) for 12 weeks 80. In the study by Karlsen et al. 72, which investigated the effect of 4 weeks of supplementation with 330 mL/day of bilberry juice in subjects with at least one risk factor for cardiovascular disease, no glucose data were shown.
Anti-inflammatory effects
Inflammation is a protective mechanism, but chronic inflammation increases oxidative stress and underlies many age-related diseases, including cardiovascular disease and cancer 91. Many studies suggest that anthocyanins, the predominant phenolic compounds found in bilberry, have anti-inflammatory effects 92. Suggested mechanisms include inhibiting proteasome activity, which controls the degradation of cellular proteins 93 and inhibiting nuclear factor κB (NF-κB) activation, which controls expression of genes involved in the inflammatory response 94. Supplementation with Medox (a commercial product of purified anthocyanins from bilberries supplying 300 mg of anthocyanins) by healthy subjects for 3 weeks was associated with a decrease in several NFκB-regulated proinflammatory chemokines and immunoregulatory cytokines 95 and a follow-up study showed decreased levels of hsCRP and inflammatory cytokines in plasma of 31 subjects who took 330 mL/day of bilberry juice for 4 weeks 96. As oxidative stress may mediate inflammation injury, the antioxidant properties of bilberry may be responsible for at least some of the anti-inflammatory effects reported. However, selective gene activation and inhibition by mechanisms other than direct antioxidant effects are likely 97.
Dymenorrhea
Women with dysmenorrhea were given bilberry extract (115 mg anthocyanosides per day) for three days before and during menstruation. A significant improvement in pelvic/lumbosacral pain, mammary tension, nausea, and lower-limb heaviness was noted 98.
Summary
Bilberry is rich in anthocyanins, especially delphinidins and cyanidins. There is supporting evidence that these compounds are bioavailable and bioactive. Molecular effects that have been demonstrated in experimental studies and animal studies. However, well-designed human trials using standardized extracts of bilberry are needed to provide the level and variety of clinical evidence that will translate current molecular insights and understanding into clear recommendations for bilberry as a possible tool to combat chronic and infectious diseases in our aging populations.
Bilberry side effects
Bilberry fruit is considered safe when consumed in amounts typically found in foods, or as an extract in recommended doses for brief periods of time. Dosages as high as 400 mg/kg body weight have been administered to rats without toxicity. Long-term oral administration in humans of doses equivalent to 180 mg/kg anthocyanins per day for six months produced no toxic effects. No mutagenic or carcinogenic effects were observed 99. Since bilberry extracts have antiplatelet aggregating properties, very high doses should be used cautiously in patients with hemorrhagic disorders and those taking anticoagulant or antiplatelet drugs. A review of studies comprising over 2,000 subjects taking bilberry extract reported only mild side effects affecting the gastrointestinal, cutaneous, or nervous system. No epidemiological studies or case reports investigating the association of exposure to bilberry extract and cancer risks in humans were identified in the available literature 100. According to Williamson and Wyandt 101, herbal preparations of bilberry should never be recommended as an alternative to diabetic medications. Potential interactions of bilberry supplements with over-the-counter and prescription medications are unknown. Bilberry leaves may be unsafe when taken orally (by mouth) in high doses or for long periods of time.
- Zorenc Z, Veberic R, Slatnar A, et al. A wild “albino” bilberry (Vaccinium myrtillus L.) from Slovenia shows three bottlenecks in the anthocyanin pathway and significant differences in the expression of several regulatory genes compared to the common blue berry type. Amancio S, ed. PLoS ONE. 2017;12(12):e0190246. doi:10.1371/journal.pone.0190246. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5741254/[↩]
- Mikulic-Petkovsek M, Schmitzer V, Slatnar A, Stampar F, Veberic R. A comparison of fruit quality parameters of wild bilberry (Vaccinium myrtillus L.) growing at different locations. J Sci Food Agric. 2014;95: 776–785. doi: 10.1002/jsfa.6897 https://www.ncbi.nlm.nih.gov/pubmed/25186189[↩]
- Veberic R, Slatnar A, Bizjak J, Stampar F, Mikulic-Petkovsek M. Anthocyanin composition of different wild and cultivated berry species. LWT-Food Sci Technol. 2015;60: 509–517.[↩]
- Falcone Ferreyra ML, Rius SP, Casati P. Flavonoids: biosynthesis, biological functions, and biotechnological applications. Front Plant Sci. 2012;3 doi: 10.3389/fpls.2012.00222 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3460232/[↩]
- Ecosystem consequences of enhanced solar ultraviolet radiation: secondary plant metabolites as mediators of multiple trophic interactions in terrestrial plant communities. Bassman JH. Photochem Photobiol. 2004 May; 79(5):382-98. https://www.ncbi.nlm.nih.gov/pubmed/15191046/[↩]
- Global change effects on plant chemical defenses against insect herbivores. Bidart-Bouzat MG, Imeh-Nathaniel A. J Integr Plant Biol. 2008 Nov; 50(11):1339-54. https://www.ncbi.nlm.nih.gov/pubmed/19017122/[↩]
- He JJ, Liu YX, Pan QH, Cui XY, Duan CQ. Different anthocyanin profiles of the skin and the pulp of Yan73 (Muscat Hamburg × Alicante Bouschet) grape berries. Molecules. 2010;15: 1141–1153. doi: 10.3390/molecules15031141 http://www.mdpi.com/1420-3049/15/3/1141[↩]
- Prior RL, Cao G, Martin A, Sofic E, McEwen J, O’Brien C, Lischner N, Ehlenfeldt M, Kalt W, Krewer G et al. Antioxidant capacity as influenced by total phenolic and anthocyanin content, maturity, and variety of Vaccinium species. J Agric Food Chem. 1998;46:2686–2693.[↩]
- Martinelli EM, Baj A, Bombardelli E. Computer-aided evaluation of liquid-chromatographic profiles for anthocyanins in Vaccinium myrtillus fruits. Anal Chim Acta. 1986;191:275–281.[↩]
- In vitro anticancer activity of fruit extracts from Vaccinium species. Bomser J, Madhavi DL, Singletary K, Smith MA. Planta Med. 1996 Jun; 62(3):212-6. https://www.ncbi.nlm.nih.gov/pubmed/8693031/[↩]
- Upton R, editor. Bilberry Fruit Vaccinium myrtillus L. Standards of Analysis, Quality Control, and Therapeutics. Santa Cruz, CA: American Herbal Pharmacopoeia and Therapeutic Compendium; 2001.[↩][↩][↩][↩][↩][↩][↩]
- Anthocyanosides of Vaccinium myrtillus (bilberry) for night vision–a systematic review of placebo-controlled trials. Surv Ophthalmol. 2004 Jan-Feb;49(1):38-50. https://www.surveyophthalmol.com/article/S0039-6257(03)00128-0/fulltext[↩][↩][↩][↩]
- Muth ER, Laurent JM, Jasper P. The effect of bilberry nutritional supplementation on night visual acuity and contrast sensitivity. Altern Med Rev 2000;5:164-173[↩][↩][↩]
- Jaakola L, Määttä K, Pirttilä AM, Törrönen R, Kärenlampi S, Hohtola A. Expression of Genes Involved in Anthocyanin Biosynthesis in Relation to Anthocyanin, Proanthocyanidin, and Flavonol Levels during Bilberry Fruit Development. Plant Physiology. 2002;130(2):729-739. doi:10.1104/pp.006957. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC166602/[↩]
- Khoo HE, Azlan A, Tang ST, Lim SM. Anthocyanidins and anthocyanins: colored pigments as food, pharmaceutical ingredients, and the potential health benefits. Food & Nutrition Research. 2017;61(1):1361779. doi:10.1080/16546628.2017.1361779. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5613902[↩]
- Laleh GH, Frydoonfar H, Heidary R, et al. The effect of light, temperature, pH and species on stability of anthocyanin pigments in four Berberis species. Pak J Nutr. 2006;5(1):90–21.[↩]
- Morazzoni P, Bombardelli E. Vaccinium myrtillus I. Fitoterapia 1996;67:3-29[↩]
- Cravotto G, Boffa L, Genzini L, Garella D. Phytotherapeutics: An evaluation of the potential of 1000 plants. J Clin Pharm Ther. 2010;35:11–48. https://www.ncbi.nlm.nih.gov/pubmed/20175810[↩][↩]
- Burdulis D, Sarkinas A, Jasutienè I, Stackivicenè E, Nikolajevas L, Janulis V. Comparative study of anthocyanin composition, antimicrobial and antioxidant activity in bilberry ( Vaccinium myrtillus L.) and blueberry ( Vaccinium corymbosum L.) fruits. Acta Pol Pharm. 2009;66:399–408. https://www.ncbi.nlm.nih.gov/pubmed/19702172[↩]
- Erlund I, Marniemi J, Hakala P, Alfthan G, Meririnne E, Aro A. Consumption of blackcurrants, lingonberries and bilberries increases serum quercetin concentrations. Eur J Clin Nutr. 2003;57:37–42 https://www.ncbi.nlm.nih.gov/pubmed/12548295[↩]
- Benzie I. F. F. Evolution of dietary antioxidants. J Comp Biochem Physiol (A) 2003;136:113–26 https://www.ncbi.nlm.nih.gov/pubmed/14527634[↩]
- Zafra-Stone S, Taharat Y, Bagchi M, Chatterjee A, Vinson J.A, Bagchi D. Berry anthocyanins as novel antioxidants in human health and disease prevention. Mol Nutr Food Res. 2007;51:675–83 https://www.ncbi.nlm.nih.gov/pubmed/17533652[↩]
- Seeram N.P. Berry fruits: Compositional elements, biochemical activities, and the impact of their intake on human health, performance, and disease. J Agric Food Chem. 2008;56:627–9. https://www.ncbi.nlm.nih.gov/pubmed/18211023[↩]
- The Case for Anthocyanin Consumption to Promote Human Health: A Review. https://onlinelibrary.wiley.com/doi/pdf/10.1111/1541-4337.12024[↩]
- Müller D, Schantz M, Richling E. High performance liquid chromatography analysis of anthocyanins in bilberries (Vaccinium myrtillus L.), blueberries (Vaccinium corymbosum L.), and corresponding juices. J Food Sci. 2012;77(4):C340–C345. https://onlinelibrary.wiley.com/doi/abs/10.1111/j.1750-3841.2011.02605.x[↩]
- Benzie I. F. F, Wachtel-Galor S. Vegetarian diets and public health: Biomarker and redox connections. Antioxid Redox Signal. 2010;13:175–91. https://www.ncbi.nlm.nih.gov/pubmed/20222825[↩]
- Lee S.W. Effects of bilberry ingestion on biomarkers of health and antioxidant content. MSc Thesis. Hong Kong: The Hong Kong Polytechnic University; 2008.[↩]
- Talavera S, Felgines C, Texier O, Besson C, Gil-Izquierdo A, Lamaison J.L, Remesy C. Anthocyanin metabolism in rats and their distribution to digestive area, kidney and brain. J Agric Food Chem. 2005;53:3902–8. https://www.ncbi.nlm.nih.gov/pubmed/15884815[↩][↩]
- Sakakibari H, Ogawa T, Koyanagi A, et al., editors. Distribution and excretion of bilberry anthocyanins in mice. J Agric Food Chem. 2009;57:7681–6 https://www.ncbi.nlm.nih.gov/pubmed/19663426[↩][↩][↩]
- Chu W, Cheung SCM, Lau RAW, et al. Bilberry (Vaccinium myrtillus L.) In: Benzie IFF, Wachtel-Galor S, editors. Herbal Medicine: Biomolecular and Clinical Aspects. 2nd edition. Boca Raton (FL): CRC Press/Taylor & Francis; 2011. Chapter 4. Available from: https://www.ncbi.nlm.nih.gov/books/NBK92770/[↩]
- Bilberry extract supplementation for preventing eye fatigue in video display terminal workers. Ozawa Y, Kawashima M, Inoue S, Inagaki E, Suzuki A, Ooe E, Kobayashi S, Tsubota K. J Nutr Health Aging. 2015 May; 19(5):548-54. https://link.springer.com/article/10.1007%2Fs12603-014-0573-6[↩][↩]
- Protective effects of bilberry and lingonberry extracts against blue light-emitting diode light-induced retinal photoreceptor cell damage in vitro. Ogawa K, Kuse Y, Tsuruma K, Kobayashi S, Shimazawa M, Hara H. BMC Complement Altern Med. 2014 Apr 2; 14():120 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3992157/[↩]
- Ashour OM, Elberry AA, Alahdal AM, et al. Protective effect of bilberry (Vaccinium myrtillus) against doxorubicin-induced oxidative cardiotoxicity in rats. Medical Science Monitor : International Medical Journal of Experimental and Clinical Research. 2011;17(4):BR110-BR115. doi:10.12659/MSM.881711.[↩]
- Pandir D, Kara O, Kara M. Protective effect of bilberry (Vaccinium myrtillus L.) on cisplatin induced ovarian damage in rat. Cytotechnology. 2014;66(4):677-685. doi:10.1007/s10616-013-9621-z. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4082771/[↩]
- Effects of Vaccinium Myrtillus anthocyanosides on arterial vasomotion. Arzneimittelforschung. 1991 Sep;41(9):905-9. https://www.ncbi.nlm.nih.gov/pubmed/1796918/[↩]
- Bilberry anthocyanin-rich extract alters expression of genes related to atherosclerosis development in aorta of apo E-deficient mice. Nutr Metab Cardiovasc Dis. 2012 Jan;22(1):72-80. doi: 10.1016/j.numecd.2010.04.011. Epub 2010 Aug 3. https://www.ncbi.nlm.nih.gov/pubmed/20678907[↩]
- Dietary anthocyanin-rich bilberry extract ameliorates hyperglycemia and insulin sensitivity via activation of AMP-activated protein kinase in diabetic mice. Takikawa M, Inoue S, Horio F, Tsuda T. J Nutr. 2010 Mar; 140(3):527-33. https://academic.oup.com/jn/article/140/3/527/4600418[↩]
- Misikangas M, Pajari AM, Paivarinta E, et al. Three Nordic berries inhibit intestinal tumorigenesis in multiple intestinal neoplasia/+ mice by modulating β-catenin signaling in the tumor and transcription in the mucosa. J Nutr 2007;137:2285–90[↩]
- Rao CN, Rao VH, Steinman B. Influence of bioflavonoids on the collagen metabolism in rats with adjuvant induced arthritis. Ital J Biochem 1981;30:54-62.[↩]
- Hess H, Knapka JJ, Newsome DA, et al. Dietary prevention of cataracts in the pink-eyed RCS rat. Lag Anim Sci 1985;35:47-53[↩]
- Bever B, Zahnd G. Plants with oral hypoglycemic action. Quart J Crude Drug Res 1979;17:139-196.[↩]
- Mykkänen OT, Huotari A, Herzig K-H, Dunlop TW, Mykkänen H, Kirjavainen PV. Wild Blueberries (Vaccinium myrtillus) Alleviate Inflammation and Hypertension Associated with Developing Obesity in Mice Fed with a High-Fat Diet. Müller M, ed. PLoS ONE. 2014;9(12):e114790. doi:10.1371/journal.pone.0114790. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4264776/[↩]
- Dietary blueberry attenuates whole-body insulin resistance in high fat-fed mice by reducing adipocyte death and its inflammatory sequelae. DeFuria J, Bennett G, Strissel KJ, Perfield JW 2nd, Milbury PE, Greenberg AS, Obin MS. J Nutr. 2009 Aug; 139(8):1510-6. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2709302/[↩]
- Purified berry anthocyanins but not whole berries normalize lipid parameters in mice fed an obesogenic high fat diet. Prior RL, Wu X, Gu L, Hager T, Hager A, Wilkes S, Howard L. Mol Nutr Food Res. 2009 Nov; 53(11):1406-18. https://www.ncbi.nlm.nih.gov/pubmed/19743407/[↩]
- Matsunaga N, Tsuruma K, Shimazawa M, Yokota S, Hara H. Inhibitory actions of bilberry anthycyanidins on angiogenesis. Phytother Res. 2010;24:S42–7. https://www.ncbi.nlm.nih.gov/pubmed/19496063[↩]
- Mutanen M, Pajari A.M, Paivarinta E, Misikangas M, Rajakangas J, Marttinen M, Oikarinen S. Berries as preventive dietary constituents-a mechanistic approach with ApcMin+ mouse. Asia Pac J Clin Nutr. 2008;17:123–5[↩]
- Cooke D, Schwartz M, Boocock D, Winterhalter P, Steward W.P, Gescher A.J, Marczylo T.H. Effect of cyanidin-3-glucoseide andan anthocyanin mixture on adenoma development in the ApcMin mouse model of intestinal carcinogenesis—relationship with tissue anthocyanin levels. Int J Cancer. 2006;119:2213–20[↩]
- Nguyen V, Tang J, Oroudjev E, Lee C.J, Marasigan C, Wilson L, Ayoub G. Cytotoxic effects of bilberry extract in MCF7-GFP-tubulin breast cancer cells. J Med Food. 2010;13:278–85. https://www.ncbi.nlm.nih.gov/pubmed/20132040[↩][↩]
- Valentova K, Ulrichova J, Cvak L, Simanek V. Cytoprotective effect of a bilberry extract against oxidative damage of rat hepatocytes. Food Chem. 2006;101:912–7.[↩]
- Upton R, editor. Bilberry Fruit Vaccinium myrtillus L. Standards of Analysis, Quality Control, and Therapeutics. Santa Cruz, CA: American Herbal Pharmacopoeia and Therapeutic Compendium; 2001[↩][↩]
- Lee K.F, Chung W.Y, Benzie I.F. Urine 8-oxo-7,8-dihydro-2′-deoxyguanosine (8-oxodG), a specific marker of oxidative stress, using direct, isocratic LC-MS/MS: Method evaluation and application in study of biological variation in healthy adults. Clin Chim Acta. 2010;411:416–22[↩]
- Lala G, Malik M, Zhao C, He J, Kwon Y, Giusti M.M, Magnuson B.A. Anthocyanin-rich extracts inhibit multiple biomarkers of colon cancer in rats. Nutr Cancer. 2006;54:84–93. https://www.ncbi.nlm.nih.gov/pubmed/16800776[↩]
- Benzie I. F. F. Lipid peroxidation: A review of causes, consequences, measurement and dietary influences. Int J Food Sci Nutr. 1996;47:233–61[↩]
- Kolosova N.G, Shcheglova T.V, Sergeeva S.V, Loskutova L.V. Long term antioxidant supplementationattenuates oxidative stress markers and cognitive deficits in senescent-accelerated OXYS rats. Neurobiol Aging. 2006;27:1289–97 https://www.ncbi.nlm.nih.gov/pubmed/16246464[↩]
- Cignarella A, Nastasi M, Cavalli E, Puglisi L. Novel lipid lowering properties of Vaccinium myrtillus L. leaves, a traditional antidiabetic treatment, in several models of rat dyslipidaemia: A comparison with clofibrate. Thromb Res. 1996;84:311–22[↩]
- Detre Z, Jellinek H, Miskulin M, Robert A.M. Studies on vascular permeability in hypertension: Action of anthocyanosides. Clin Physiol Biochem. 1986;4:143–9[↩]
- Tsuda T. Regulation of adipocyte function by anthocyanins: Possibility of preventing the metabolic syndrome. J Agric Food Chem. 2008;56:642–6[↩]
- Takikawa M, Inoue S, Horio F, Tsuda T. Dietary anthocyaninrich bilberry extract ameliorates hyperglycemia and insulin sensitivity via activation of AMP-activated protein kinase in diabetic mice. J Nutr. 2009;140:527–33[↩][↩]
- Miyake S, Takahashi N, Sasaki M, Kobayashi S, Tsubota K, Ozawa Y. Vision preservation during retinal inflammation by anthocyanin-rich bilberry extract: cellular and molecular mechanism. Lab Investig. 2012;92:102–109. doi: 10.1038/labinvest.2011.132. https://www.ncbi.nlm.nih.gov/pubmed/21894150[↩]
- Osada H, Okamoto T, Kawashima H, et al. Neuroprotective effect of bilberry extract in a murine model of photo-stressed retina. Lewin AS, ed. PLoS ONE. 2017;12(6):e0178627. doi:10.1371/journal.pone.0178627. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5453571/[↩]
- Narimatsu T, Ozawa Y, Miyake S, Nagai N, Tsubota K. Angiotensin II type 1 receptor blockade suppresses light-induced neural damage in the mouse retina. Free Radic Biol Med. 2014;71:176–85. doi: 10.1016/j.freeradbiomed.2014.03.020 https://www.sciencedirect.com/science/article/pii/S0891584914001373[↩]
- Matsunaga N, Imai S, Inokuchi Y, Shimazawa M, Yokata S, Araki Y, Hara H. Bilberry and its main constituents have neuroprotective effects against retinal neuronal damage in vitro and in vivo. Mol Nutr Food Res. 2009;53:869–77[↩]
- Zafra-Stone S, Taharat Y, Bagchi M, Chatterjee A, Vinson J.A, Bagchi D. Berry anthocyanins as novel antioxidants in human health and disease prevention. Mol Nutr Food Res. 2007;51:675–83[↩][↩]
- Puupponen-Pimià R, Nohynek L, Alakomi H.-L, Oksman-Caldentey K.-M. Theaction of berry phenolics against human intestinal pathogens. Biofactors. 2005b;23:243–51. https://www.ncbi.nlm.nih.gov/pubmed/16498212[↩][↩]
- Development of a novel adsorptive membrane chromatographic method for the fractionation of polyphenols from bilberry. Journal of Agricultural and Food Chemistry, 60 (10) (2012), pp. 2427-2433[↩]
- Ogawa K, Kuse Y, Tsuruma K, Kobayashi S, Shimazawa M, Hara H. Protective effects of bilberry and lingonberry extracts against blue light-emitting diode light-induced retinal photoreceptor cell damage in vitro. BMC Complementary and Alternative Medicine. 2014;14:120. doi:10.1186/1472-6882-14-120. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3992157/[↩]
- Morazzoni PBE. Vaccinium myrtillus. Fitoterapia. 1996;67:3–29.[↩]
- Morazoni PMMJ. Activity of myrtocyan®, an anthocyanoside complex from Vaccinium myrtillus (VMA), on platelet aggregation and adhesiveness. Fitoterapia. 1990;61:13–21.[↩]
- Colantuoni A, Bertuglia S, Magistretti MJ, Donato L. Effects of Vaccinium Myrtillus anthocyanosides on arterial vasomotion. Arzneimittelforschung. 1991;41:905–909 https://www.ncbi.nlm.nih.gov/pubmed/1796918[↩]
- Bilberry juice modulates plasma concentration of NF-κB related inflammatory markers in subjects at increased risk of CVD. European Journal of Nutrition, 2010, Volume 49, Number 6, Page 345 https://link.springer.com/article/10.1007%2Fs00394-010-0092-0[↩]
- Freese R, Vaarala O, Turpeinen A.M, Mutanen M. No difference in platelet activation or inflammation markers after diets rich or poor in vegetables, berries and apple in healthy subjects. Eur J Nutr. 2004;43:175–82. https://www.ncbi.nlm.nih.gov/pubmed/15168040[↩]
- Karlsen A, Paur I, Bøhn S.V, et al., editors. Bilberry juice modulates plasma concentration of NF-κB related inflammatory markers in subjects at increased risk of CVD. Eur J Nutr. 2010 2010 February 2;49:345–55. Epub. https://www.ncbi.nlm.nih.gov/pubmed/20119859[↩][↩][↩][↩][↩][↩]
- Lee S.W. Effects of bilberry ingestion on biomarkers of health and antioxidant content. MSc Thesis. Hong Kong: The Hong Kong Polytechnic University; 2008[↩][↩][↩]
- Benzie I. F. F, Strain J.J. Ferric reducing/antioxidant power assay: Direct measure of total antioxidant activity of biological fluids and modified version for simultaneous measurement of total antioxidant power and ascorbic acid concentration. Methods Enzymol. 1999;299:15–27. https://www.ncbi.nlm.nih.gov/pubmed/9916193[↩]
- Halliwell B. Oxidative stress and cancer: Have we moved forward? Biochem J. 2007;401:1–11 https://www.ncbi.nlm.nih.gov/pubmed/17150040[↩]
- Benzie I. F. F, Wachtel-Galor S. Vegetarian diets and public health: Biomarker and redox connections. Antioxid Redox Signal. 2010;13:175–91 https://www.ncbi.nlm.nih.gov/pubmed/20222825[↩]
- Libby P, Ridker P.M, Maseri A. Inflammation and atherosclerosis. Circulation. 2002;105:1135–43. http://circ.ahajournals.org/content/105/9/1135.long[↩]
- Erlund I, Kol R, Alfthan G, et al., editors. Favourable effects of berry consumption on platelet function, blood pressure, and HDL cholesterol. Am J Clin Nutr. 2008;87:323–31. https://www.ncbi.nlm.nih.gov/pubmed/18258621[↩][↩]
- Barringer T.A, Hatcher L, Sasser H.C. Potential benefits on impairment of endothelial function after a high-fat meal of 4 weeks of flavonoid supplementation. Evid Based Complement Alternat Med. 2008;10:1–6. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3137609/[↩]
- Qin Y, Xia M, Ma J, et al., editors. Anthocyanin supplementation improves serum LDL- and HDL-cholesterol concentrations associated with the inhibition of cholesteryl ester transfer protein in dyslipidemic subjects. Am J Clin Nutr. 2009;90:485–92 https://www.ncbi.nlm.nih.gov/pubmed/19640950[↩][↩][↩][↩]
- Persson I.A, Persson K, Andersson R.G. Effect of Vaccinium myrtillus and its polyphenols on angiotensin-converting enzyme activity in human endothelial cells. J Agric Food Chem. 2009;57:4626–9[↩][↩]
- Ghosh D, Konishi T. Anthocyanins and anthocyanin-rich extracts: Role in diabetes and eye function. Asia Pac J Clin Nutr. 2007;16:200–8. https://www.ncbi.nlm.nih.gov/pubmed/17468073[↩]
- Bravetti G.O, Fraboni E, Maccolini E. Preventive medical treatment of senile cataract with vitamin E and Vaccinium myrtillus anthocyanosides: Clinical evaluation. Ann Ottalmol Clin Ocul. 1989;115:109–16[↩]
- Lee J, Lee H.K, Kim C.Y, Choe C.M, You T.W, Seong G.J. Purified high-dose anthocyanoside oligomer administration improves nocturnal vision and clinical symptoms in myopia subjects. Br J Nutr. 2005;93:895–9 https://www.ncbi.nlm.nih.gov/pubmed/16022759[↩]
- Pilot Study of Oral Anthocyanins for Colorectal Cancer Chemoprevention. Cancer Prev Res July 1 2009 (2) (7) 625-633; DOI: 10.1158/1940-6207.CAPR-08-0201 http://cancerpreventionresearch.aacrjournals.org/content/2/7/625[↩][↩][↩][↩]
- Bilberry ingestion improves disease activity in mild to moderate ulcerative colitis – an open pilot study. Biedermann L, Mwinyi J, Scharl M, Frei P, Zeitz J, Kullak-Ublick GA, Vavricka SR, Fried M, Weber A, Humpf HU, Peschke S, Jetter A, Krammer G, Rogler G. J Crohns Colitis. 2013 May; 7(4):271-9. https://academic.oup.com/ecco-jcc/article/7/4/271/385488[↩][↩]
- A meta-analysis of the placebo rates of remission and response in clinical trials of active ulcerative colitis. Gastroenterology. 2007 Feb;132(2):516-26. Epub 2006 Dec 20. https://www.ncbi.nlm.nih.gov/pubmed/17258720[↩]
- McDougall G.J, Kulkarni N.N, Stewart D. Current developments on the inhibitory effects of berry polyphenols on digestive enzymes. Biofactors. 2008;34:73–80 https://www.ncbi.nlm.nih.gov/pubmed/19706974[↩]
- Tsuda T. Regulation of adipocyte function by anthocyanins: Possibility of preventing the metabolic syndrome. J Agric Food Chem. 2008;56:642–6. https://www.ncbi.nlm.nih.gov/pubmed/18211021[↩]
- Helmstädter A, Schuster N. Vaccinium myrtillus as an antidiabetic medicinal plant-research through the ages. Pharmazie. 2010;65:315–21 https://www.ncbi.nlm.nih.gov/pubmed/20503920[↩]
- Aggarwal B.B, Vijayalekshmi R.V, Sung B. Targeting inflammatory pathways for prevention and therapy of cancer: Short-term friend, long-term foe. Clin Cancer Res. 2009;15:425–30. http://clincancerres.aacrjournals.org/content/15/2/425.long[↩]
- Kim Y.S, Young M.R, Bobe G, Colburn N.H, Milner J.A. Bioactive food components, inflammatory targets and cancer prevention. Cancer Prev Res. 2009;2:200–8 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3449301/[↩]
- Dreiseitel A, Schreier P, Oehme A, et al., editors. Inhibition of proteasome activity by anthocyanins and anthocyanidins. Biochem Biophys Res Commun. 2008;372:57–61.[↩]
- Chen J, Uto T, Tanigawa S, Kumamoto T, Fuji M, Hou D.X. Expression profiling of genes targeted by bilberry (Vaccinium myrtillus) in macrophages through DNA array. Nutr Cancer. 2008;60:43–50[↩]
- Karlsen A, Retterstol L, Laake P, Paur I, Kjolsrud-Bohn S, Sandvik L, Blomhoff R. Anthocyanins inhibit nuclear factor-B activation in monocytes and reduce plasma concentrations of pro-inflammatory mediators in healthy adults. J Nutr. 2007;137:1951–4.[↩]
- Karlsen A, Paur I, Bøhn S.V, et al., editors. Bilberry juice modulates plasma concentration of NF-κB related inflammatory markers in subjects at increased risk of CVD. Eur J Nutr. 2010 2010 February 2;49:345–55. Epub.[↩]
- Benzie I. F. F, Wachtel-Galor S. Vegetarian diets and public health: Biomarker and redox connections. Antioxid Redox Signal. 2010;13:175–91.[↩]
- Colombo D, Vescovini R. Controlled trial of anthocyanosides from Vaccinium myrtillus in primary dysmenorrhea. G Ital Ost Ginecol 1985;7:1033-1038l.[↩]
- Eandi M. Post-marketing investigation on Tegens “preparation with respect to side effects. 1987. Cited in Morazzoni P, Bombardelli E. Vaccinium myrtillus I. Fitoterapia 1996;67:3-29.[↩]
- https://ntp.niehs.nih.gov/ntp/htdocs/chem_background/exsumpdf/bilberry_508.pdf[↩]
- Williamson, J.S. & Wyandt, C.M. (1998) An herbal update. Drug Top., 142, 66-75[↩]