Vitamins and supplements for hair growth
Hair loss also known as alopecia is a common problem that affects up to 50 percent of men and women throughout their lives 1. Hair loss can also affect children. Hair loss can affect people of all ages. Hair loss can occur anywhere on your body, but more commonly affecting just your scalp where it can cause concerns about the cosmetic effect. Studies have shown that hair loss can be associated with low self-esteem, anxiety, depression, introversion, and feelings of unattractiveness 2, 3.
This is reinforced by attitudes in Western society, which place great value on youthful appearance and attractiveness. Some studies have shown that based on appearance alone, men with hair loss are seen as less attractive, less assertive, less likeable, and less successful than men without hair loss.
Hair loss can occur in different patterns, depending on the cause. The hair loss can be temporary or permanent.
Hair loss is not usually anything to be worried about, but occasionally it can be a sign of a medical condition.
Some types of hair loss are permanent, like male and female pattern baldness also known as androgenic alopecia or androgenetic alopecia. This type of hair loss usually runs in the family.
Hair loss occurs in:
- Men and women
- Children and adults
- People with any color or type of hair.
Hair loss can be an isolated problem or associated with another disease or condition, including:
- A family history of balding on your mother’s or father’s side. Androgenic alopecia also known as androgenetic alopecia is the modern medical term for either male pattern hair loss or female pattern baldness caused by a combination of genetic and hormonal factors 4. Androgenic alopecia represents close to 95% of all hair loss. This hair loss causes a receding hairline and lack of hair on the top of the head. This type of hair loss can be defined in two parts. First, andro- means to consist of androgens which are various hormones that control the appearance and development of masculine characteristics such as testosterone. Second is genetics, or the inheritance of genes from either the mother or father. Age added to genetics creates a time clock that signals the hair follicle to produce an enzyme named 5-alpha reductase. When testosterone is present in the hair follicle and it combines with the enzyme 5-alpha reductase, it produces dihydrotestosterone (DHT). DHT (dihydrotestosterone) attacks the hair follicle, causing it to shrink, finally causing the hair to fall out and not grow back. Hair follicle receptors are sensitive to DHT and thereby start the process of male or female pattern hair loss 5.
- Age
- Significant weight loss
- Prolonged fever
- Certain medical conditions, such as diabetes and lupus
- Stressful conditions, physical or emotional, such as illness or surgery
- Poor nutrition
- Drug treatment for cancer
- Autoimmune disease
- A localized infection, such as tinea capitis
- Severe local skin disease, such as psoriasis, seborrheic dermatitis, atopic dermatitis, pityriasis rubra pilaris, cutaneous lupus erythematosus, cutaneous T-cell lymphoma
- Generalized skin disease (erythroderma)
- Traumatic causes
- Other causes of hair loss include certain medicines (e.g., chemotherapy drugs, contraceptives, anticoagulants, anticonvulsants), low levels of iron in your blood (iron deficiency), pregnancy (after childbirth), syphilis, thyroid disease, systemic lupus erythematosus (SLE) and repeated hair twisting
- Unknown causes.
Hair loss can be temporary or permanent, depending on the cause.
- Hair loss may be localized or diffuse.
- Hair loss can affect the scalp or other parts of the body.
- Hair loss may be due to hair shedding, poor quality hair, or hair thinning.
- There may be areas of skin that are completely bald.
- There may be associated skin disease or scarring.
Local hair loss in one or more small parts of the scalp can be caused by any of the following:
- Alopecia areata (patchy hair loss; the cause is unknown)
- Traction alopecia (tight hairstyles such as cornrows or pigtails)
- Trichotillomania (repeated hair pulling or nervous hair twisting or twirling)
- Tinea capitis (ringworm or fungal infection)
Since hair loss may be an early sign of a disease, it is important to find the cause so that it can be treated.
If you suspect that you may have excessive hair loss, talk to your doctor. He or she will probably ask you some questions about your diet, any medicines you’re taking, and whether you’ve had a recent illness, and how you take care of your hair. If you’re a woman, your doctor may ask questions about your menstrual cycle, pregnancies, and menopause. Your doctor may want to do a physical exam to look for other causes of hair loss. Finally, your doctor may order blood tests to measure hormone levels, serum ferritin and thyroid function or a biopsy (taking a small sample of cells to examine under a microscope). Your doctor will usually diagnose androgenetic alopecia by examining the pattern of hair loss on the scalp.
Treatment for hair loss depends on the cause. In some cases, treating the underlying cause will correct the problem. With some conditions, such as patchy hair loss (alopecia areata), hair may regrow without treatment within a year.
If a medicine is causing your hair loss, your doctor may be able to prescribe a different medicine. Recognizing and treating an infection may help stop the hair loss. Correcting a hormone imbalance may prevent further hair loss.
Sometimes changing how you style or treat your hair can help. Getting rid of stress in your life can also help. Other treatments include changing your diet, correcting any hormone imbalances, switching medicines, treating infections, or getting shots into your scalp.
Medicines may also help slow or prevent the development of common baldness. One medicine that is used to slow hair loss, minoxidil (brand name: Rogaine), is available without a prescription. It is applied to the scalp. Both men and women can use it. Minoxidil (Rogaine) is the only US Food and Drug Administration (FDA) approved topical treatment for male or female pattern hair loss. Although minoxidil (Rogaine) is not effective in stimulating new hair growth in many males, it appears to be more effective in retarding hair loss in a substantial amount of both male and females.
Another medicine, finasteride (type II 5-alpha-reductase inhibitor), is available with a prescription. It comes in pills and is only for men. 5 alpha reductase converts testosterone into dihydrotestosterone (DHT). DHT (dihydrotestosterone) binding to the scalp hair follicle androgen receptors produces male pattern hair loss. For men, finasteride tablets reduce levels of dihydrotestosterone (DHT), which may slow hair loss and possibly help regrowth of hair 6. A daily oral finasteride dose of one milligram reduces scalp dihydrotestosterone by 64% and serum dihydrotestosterone by 68% 7. Continuous use for 3 to 6 months is required before a benefit is usually seen. It may take up to 6 months before you can tell if one of these medicines is working. When you stop taking these medicines, any beneficial effects on hair growth will be lost within 6 to 12 months of discontinuing treatment. Decreased libido and erectile problems are recognized side-effects of this treatment.
If treatment doesn’t work or is not available for your type of hair loss, you may want to consider wearing a wig, hairpiece, hair weave, or artificial hair replacement.
Despite the widespread use of supplements and vitamins for hair growth or hair loss, the safety and effectiveness of available products remain unclear 8. Studies of nutritional interventions with the highest-quality evidence showed the potential benefit of Viviscal, Nourkrin, Nutrafol, Lambdapil, Pantogar, capsaicin and isoflavone, omegas 3 and 6 with antioxidants, apple nutraceutical, total glucosides of paeony and compound glycyrrhizin tablets, zinc, tocotrienol, and pumpkin seed oil 8. Kimchi and cheonggukjang, vitamin D3, and Forti5 had low-quality evidence for hair growth 8.
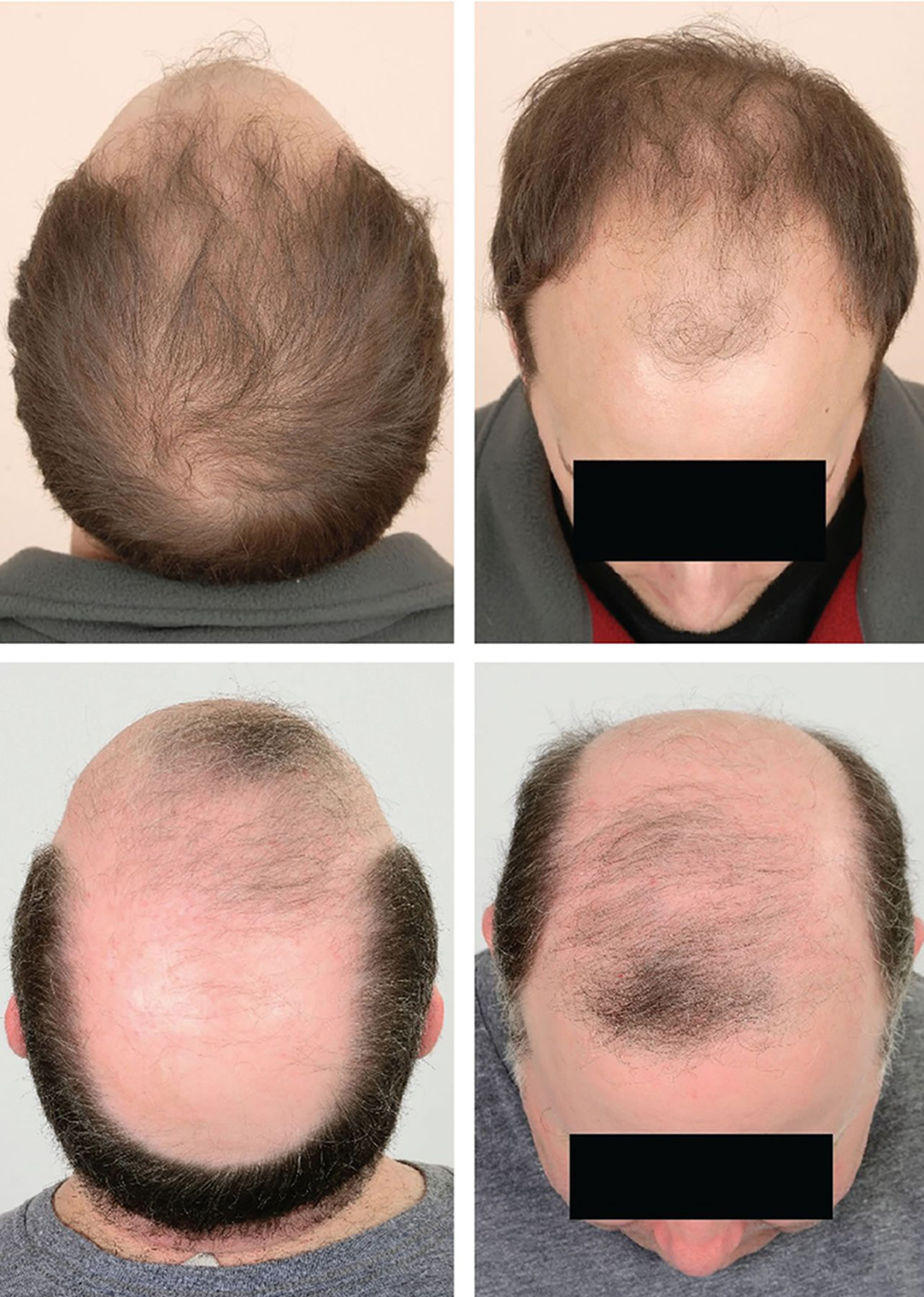
Figure 1. Male pattern hair loss
Figure 2. Female pattern hair loss
See your doctor if you are distressed by persistent hair loss in you or your child and want to pursue treatment. For women who are experiencing a receding hairline (frontal fibrosing alopecia), talk with your doctor about early treatment to avoid significant permanent baldness.
Also talk to your doctor if you notice sudden or patchy hair loss or more than usual hair loss when combing or washing your or your child’s hair. Sudden hair loss can signal an underlying medical condition that requires treatment.
Also if you are having significant, persistent hair loss or if there is redness, itching, or skin changes associated with the hair loss, seek medical advice, as there are sometimes other causes for hair loss that can be treated.
Lastly, if you have hair loss that is cosmetically concerning and other causes have been ruled out, you might consult a surgical specialist in hair replacement.
What is male pattern hair loss?
Male pattern hair loss also known as androgenic alopecia or androgenetic alopecia, is the most common type of diffuse thinning of the hair and balding that occurs in adult males.
- Male pattern hair loss is due to a combination of hormones (androgens) and a genetic predisposition.
- Male pattern hair loss is characterized by a receding hairline and hair loss on the top and front of the head.
- A similar type of hair loss in women, female pattern hair loss, results in thinning hair on the mid-frontal area of the scalp and is generally less severe than occurs in males.
What causes male pattern baldness?
Male pattern hair loss is an inherited condition, caused by a genetically determined sensitivity to the effects of dihydrotestosterone (DHT) in some areas of the scalp. DHT (dihydrotestosterone) is believed to shorten the growth, or anagen phase of the hair cycle, from a usual duration of 3–6 years to just weeks or months. This occurs together with miniaturisation of the follicles and progressively produces fewer and finer hairs. The production of DHT is regulated by an enzyme called 5-alpha reductase.
Male pattern hair loss occurs in men who are genetically predisposed to be more sensitive to the effects of DHT. Researchers now believe that the condition can be inherited from either side of the family.
Several genes are involved, accounting for differing age of onset, progression, pattern and severity of hair loss in family members. The susceptibility genes are inherited from both mother and father. At this time, genetic testing for prediction of balding is unreliable.
A few women present with male pattern hair loss because they have excessive levels of androgens as well as genetic predisposition. These women also tend to suffer from acne, irregular menses and excessive facial and body hair. These symptoms are characteristic of polycystic ovarian syndrome (PCOS) although the majority of women with PCOS do not experience hair loss. Less often, congenital adrenal hyperplasia may be responsible. Females that are losing their hair with age are more likely to present with female pattern hair loss, in which hormone tests are normal.
Is male pattern hair loss hereditary?
Yes. It is believed male pattern hair loss can be inherited from either or both parents.
How common is male pattern hair loss?
Male pattern hair loss affects nearly all men at some point in their lives. It affects different populations at different rates, probably because of genetics. Up to half of male Caucasians will experience some degree of hair loss by age 50, and possibly as many as 80% by the age of 70 years, while other population groups such as Japanese and Chinese men are far less affected.
Can male pattern hair loss be cured?
No, there is no cure. However, it tends to progress very slowly, from several years to decades. An earlier age of onset may lead to quicker progression.
Can my hairstyle cause hair loss?
Wigs, tight braids, hair weaves, and the use of hair curlers can damage hair and lead to hair loss. Hair processing (such as bleaching, coloring, and perming) can also damage hair and cause hair loss. Your hair will usually grow back once you stop stressing your hair. In certain cases, it can lead to scarring and permanent hair loss.
What treatments are available for male pattern hair loss?
Current male pattern hair loss treatment options include:
- Hair replacement / transplantation
- Cosmetics
- Micropigmentation (tattoo) to resemble shaven scalp
- Hairpieces
- Minoxidil solution
- Finasteride tablets (type II 5-alpha-reductase inhibitor)
- Dutasteride (type I and type II 5-alpha-reductase inhibitor).
A phase 2 randomized placebo-controlled study of dutasteride versus finasteride showed that the effect of dutasteride was dose dependent and 2.5mg of dutasteride was superior to 5mg finasteride in improving scalp hair growth in men between the ages of 21 and 45 years 9. It was also able to produce hair growth earlier than finasteride. This was evidenced by target area hair counts and clinical assessment at 12 and 24 weeks. In addition, a recent randomized, double blind, placebo-controlled study on the efficacy of dutasteride 0.5mg/day in identical twins demonstrated that dutasteride was able to significantly reduce hair loss progression in men with male pattern hair loss 10. A single case report showed improvement of hair loss with dutasteride 0.5mg in a woman who had failed to show any response to finasteride 11, 12.
In one phase 3 study dutasteride 0.5 mg daily showed significantly higher efficacy than placebo based on subject self-assessment and by investigator and panel photographic assessment 13. There was no major difference in adverse events between two groups the treatment and placebo groups. However, this study was limited to only 6 months. Another more recent phase 3 trial found that dutasteride 0.5 mg was statistically superior to finasteride 1 mg and placebo at 24 weeks 14.
There is some evidence that ketoconazole shampoo may also be of benefit, perhaps because it is effective in seborrheic dermatitis and dandruff 15, 16. Low-dose oral minoxidil (off label) can increase hair growth on the scalp, but may also result in generalized hypertrichosis and other adverse effects 17.
Low-level laser therapy (LLLT) is of unproven benefit in male pattern balding; the Capillus® laser cap and Hairmax® Lasercomb/Laserband are two low‐level laser therapy (LLLT) devices have been approved by the FDA for the management of androgenetic alopecia 18, 19. Minimal side effects were reported. Small number of participants reported adverse events of acne, mild paresthesia such as burning sensation, dry skin, headache, and itch 20.
Light‐emitting diode (LED) devices. In contrast with low-level laser therapy (LLLT) that delivers a single, collimated wavelength of light, light‐emitting diode (LED) devices may emit a small band of wavelengths. In particular, an all‐LED device that delivers dual dark orange (620 nm) and red light (660 nm) (Revian Red) to promote blood flow, reduce inflammation, and inhibit DHT via 5-alpha-reductase downregulation 21. In a prospective, randomized, double‐blind, controlled study, 18 male pattern hair loss subjects were treated with Revian Red cap vs. 18 male pattern hair loss subjects were treated with a sham light device for 10 min daily for 16 weeks total 22. Preliminary photographic assessments revealed increased mean hair count in the active group as compared to placebo group. Specifically, active group participants demonstrated approximately 26.3 more hairs per cm² compared to the placebo group. Overall, literature has suggested light therapy to be a safe treatment modality for androgenic alopecia (androgenetic alopecia) in both male and female patients when used independently or in combination with topical/oral therapies 20, 23. Light therapy has an excellent side effect profile, and there are no contraindications for use, although caution may be taken when administering in patients with dysplastic lesions on the scalp 24.
Platelet-rich plasma injections are also under investigation 25. Platelet‐rich plasma treatment can be administered alone or in combination with other therapies for androgenetic alopecia, although better results are obtained if platelet‐rich plasma administration is used in association with topical (such as minoxidil) or oral therapies (finasteride) 25. Further studies are required to determine the magnitude of the benefit if any.
Platelet‐rich plasma is generally indicated for patients with early‐stage androgenic alopecia, as intact hair follicles are present and a more significant hair restorative effect can be achieved 26. During the procedure, approximately 10–30 mL of blood are drawn from the patient’s vein and centrifuged for 10 min in order to separate the plasma from red blood cells. The platelet‐rich plasma, containing numerous growth factors, is then injected into the deep dermis or subcutaneous tissue at a volume of 4–8 mL per session. Mild side effects include scalp pain, headache, and burning sensation, but these effects usually subside in 10–15 minutes post‐injection and do not warrant use of topical anesthesia or pain medications 27. Vibration or cool air is typically sufficient to alleviate any significant pain that a patient may feel from the treatment. Patients can resume regular activities immediately after treatment but should avoid strenuous physical activity 24 hour post‐treatment to allow for optimal absorption of platelet‐rich plasma into tissue.
Hausauer and Jones 28 conducted a single center, blinded, randomized controlled trial investigating the efficacy of two platelet‐rich plasma regimens in 40 androgenic alopecia subjects. Participants received either subdermal platelet‐rich plasma injections with 3 monthly sessions and booster 3 months later (group 1) or 2 sessions every 3 months (group 2). Folliscope hair count and shaft caliber, global photography, and patient satisfaction questionnaires were completed at baseline, 3‐month, and 6‐month visits. The authors reported statistically significant increases in hair count and shaft caliber in both groups at 6 months. Importantly, improvements occurred more rapidly and profoundly in group 1, indicating that platelet‐rich plasma injections should be administered first monthly 28. Alves and Grimalt 29 demonstrated significant differences in mean anagen hair and telogen hair count as well as telogen and overall hair density when compared to baseline. In a review of 16 studies comprising a total of 389 patients with androgenic alopecia, the majority demonstrated efficacy in promoting successful hair growth after 3–4 sessions on a monthly basis, followed by quarterly maintenance sessions 30.
Platelet‐rich plasma is not curative for hair loss and must be continued long term for hair sustenance. However, patient satisfaction is typically very high and 60–70% of patients continue to undergo maintenance treatments. Due to the relatively recent introduction of platelet‐rich plasma injections for androgenic alopecia, there are no long‐term studies evaluating its effectiveness. Additionally, it is difficult to compare the efficacy with other remedies due to the lack of standardization in regard to platelet‐rich plasma kits, treatment fractions, and regimens, including the use of newer multi‐needle injectors.
While platelet‐rich plasma injections are considered safe when performed by a trained medical provider, these treatments are not suitable for everyone. Platelet‐rich plasma may not be appropriate for those with a history of bleeding disorders, autoimmune disease, or active infection, or those currently taking an anticoagulant medication. Although the majority of patients seem to tolerate the pain associated with scalp injections, some patients may prefer to avoid it.
Clascoterone gained FDA approval in August 2020 as the first topical antiandrogen agent to treat hormonal acne. The clascoterone molecule resembles DHT and spironolactone in molecular structure and works by antagonizing androgen receptors on dermal papillae and inhibiting DHT’s effect on hair miniaturization and dermal inflammation 31. Due to its mechanism of action, clascoterone has potential in treating androgenic alopecia. In a 6‐month dose‐ranging study, patients with androgenic alopecia who received clascoterone 7.5% twice a day showed a significant improvement in hair loss from baseline and compared to those who received placebo 32.
What is female pattern hair loss?
Female pattern hair loss also known as androgenic alopecia or androgenetic alopecia, is a distinctive form of diffuse hair loss that occurs in women. Many women are affected by female pattern hair loss. Around 40% of women by age 50 show signs of hair loss and less than 45% of women reach the age of 80 with a full head of hair.
In female pattern hair loss, there is diffuse thinning of hair on the scalp due to increased hair shedding or a reduction in hair volume, or both. It is normal to lose up to 50-100 hairs a day. Another condition called chronic telogen effluvium also presents with increased hair shedding and is often confused with female pattern hair loss. It is important to differentiate between these conditions as management for both conditions differ.
Female pattern hair loss presents quite differently from the more easily recognizable male pattern baldness, which usually begins with a receding frontal hairline that progresses to a bald patch on top of the head. It is very uncommon for women to bald following the male pattern unless there is excessive production of androgens in the body.
What causes female pattern hair loss?
Female pattern hair loss has a strong genetic predisposition. The mode of inheritance is polygenic, indicating that there are many genes that contribute to female pattern hair loss, and these genes could be inherited from either parent or both. Genetic testing to assess the risk of balding is currently not recommended, as it is unreliable.
Currently, it is not clear if androgens (male sex hormones) play a role in female pattern hair loss, although androgens have a clear role in male pattern baldness. The majority of women with female pattern hair loss have normal levels of androgens in their bloodstream. Due to this uncertain relationship, the term female pattern hair loss is preferred to ‘female androgenetic alopecia’.
The role of estrogen is uncertain. female pattern hair loss is more common after the menopause suggesting estrogens may be stimulatory for hair growth. But laboratory experiments have also suggested estrogens may suppress hair growth.
What treatments are available for female pattern hair loss?
A Cochrane systematic review published in 2012 33 concluded that minoxidil solution was effective for female pattern hair loss. Minoxidil is available as 2% and 5% solutions; the stronger preparation is more likely to irritate and may cause undesirable hair growth unintentionally on areas other than the scalp.
Hormonal treatment, i.e. anti-androgen medicines are oral medications that block the effects of androgens (e.g. spironolactone, cyproterone acetate, finasteride and flutamide) is also often tried. These medicines help stop hair loss and may also stimulate hair regrowth. Spironolactone has been shown to stop the loss of hair in 90 per cent women with androgenetic alopecia. In addition, partial hair regrowth occurs in almost half of treated women. The effects of treatment generally only last while you continue to take the medicine – stopping the medicine will mean that your hair loss will return. Spironolactone and cyproterone acetate should not be taken during pregnancy. Effective contraception must be used while you are being treated with these medicines, as it can affect a developing baby. These medicines should also not be taken if you are breast feeding.
A combination of low dose oral minoxidil (eg, 2.5 mg daily) and spironolactone (25 mg daily) has been shown to significantly improve hair growth, reduce shedding and improve hair density.
Once started, treatment needs to continue for at least six months before the benefits can be assessed, and it is important not to stop treatment without discussing it with your doctor first. Long term treatment is usually necessary to sustain the benefits.
It takes about 4 months of using minoxidil to see any obvious effect. You might have some hair loss for the first couple of weeks as hair follicles in the resting phase are stimulated to move to the growth phase. You need to keep using minoxidil to maintain its effect – once you stop treatment the scalp will return to its previous state of hair loss within 3 to 4 months. Also, be aware that minoxidil is not effective for all women, and the amount of hair regrowth will vary among women. Some women experience hair regrowth while in others hair loss is just slowed down. If there is no noticeable effect after 6 months, it’s recommended that treatment is stopped.
Always carefully follow the directions for use, making sure you use minoxidil only when your scalp and hair are completely dry. Take care when applying minoxidil near the forehead and temples to avoid unwanted excessive hair growth. Wash your hands after use.
The most common side effects of minoxidil include a dry, red and itchy scalp. Higher-strength solutions are more likely to cause scalp irritation.
Bear in mind that minoxidil is also used in tablet form as a prescription medicine to treat high blood pressure, and there is a small chance that minoxidil solution could possibly affect your blood pressure and heart function. For this reason, minoxidil is generally only recommended for people who do not have heart or blood pressure problems.
Minoxidil should not be used if you are pregnant or breast feeding.
Side effects of spironolactone can include:
- irregular periods and spotting;
- breast tenderness or lumpiness; and
- tiredness.
Side effects of cyproterone acetate can include:
- spotting and irregular periods;
- tiredness;
- weight gain;
- reduced libido; and
- depressed mood.
Cosmetic camouflages include colored hair sprays to cover thinning areas on the scalp, hair bulking fiber powder, and hair wigs. Hair transplantation for female pattern hair loss is becoming more popular although not everyone is suitable for this procedure. Your doctor can refer you to a hair transplant surgeon to assess whether hair transplant surgery may be a suitable option for you.
Hair transplant surgery involves follicular unit transplantation, where tiny clusters of hair-producing tissue (each containing up to 4 hairs) are taken from areas of the scalp where hair is growing well and surgically attached (grafted) onto thinning areas. However, if your hair is very thin all over your scalp, you may not have enough healthy hair to transplant.
Hair transplant surgery can be expensive and painful, and multiple procedures are sometimes needed. Side effects may include infection and scarring.
Low-level laser therapy (LLLT) is of unproven benefit in female pattern balding; the Capillus® laser cap and Hairmax® Lasercomb/Laserband are two low‐level laser therapy (LLLT) devices have been approved by the FDA for the management of androgenetic alopecia 34, 18, 19. In a randomized, double‐blind, placebo‐controlled trial comprising 42 female subjects with androgenetic alopecia, 24 active group subjects were treated with 655 nm low-level laser therapy (LLLT) vs. 18 placebo group subjects were treated with incandescent red lights (sham) 34. Subjects were treated on alternate days for 16 weeks, and photography and hair count assessments revealed a 37% increase in terminal hair counts in the active treatment group as compared to the control group. In a review of 11 trials, 10 demonstrated significant improvement in androgenetic alopecia compared to baseline or controls when treated with low-level laser therapy (LLLT) 20. Two of the trials demonstrated efficacy for low-level laser therapy (LLLT) in combination with topical minoxidil, and one trial showed efficacy in combination with finasteride 26. Small number of participants reported adverse events of acne, mild paresthesia such as burning sensation, dry skin, headache, and itch 20.
Overall, literature has suggested light therapy to be a safe treatment modality for androgenic alopecia (androgenetic alopecia) in both male and female patients when used independently or in combination with topical/oral therapies 20, 23. Light therapy has an excellent side effect profile, and there are no contraindications for use, although caution may be taken when administering in patients with dysplastic lesions on the scalp 24.
Platelet‐rich plasma (PRP) injections are also under investigation 25. Further studies are required to determine the magnitude of the benefit if any.
Platelet‐rich plasma is generally indicated for patients with early‐stage androgenic alopecia, as intact hair follicles are present and a more significant hair restorative effect can be achieved 26. During the procedure, approximately 10–30 mL of blood are drawn from the patient’s vein and centrifuged for 10 min in order to separate the plasma from red blood cells. The platelet‐rich plasma, containing numerous growth factors, is then injected into the deep dermis or subcutaneous tissue at a volume of 4–8 mL per session. Mild side effects include scalp pain, headache, and burning sensation, but these effects usually subside in 10–15 minutes post‐injection and do not warrant use of topical anesthesia or pain medications 27. Vibration or cool air is typically sufficient to alleviate any significant pain that a patient may feel from the treatment. Patients can resume regular activities immediately after treatment but should avoid strenuous physical activity 24 hour post‐treatment to allow for optimal absorption of platelet‐rich plasma into tissue.
Hausauer and Jones 28 conducted a single center, blinded, randomized controlled trial investigating the efficacy of two platelet‐rich plasma regimens in 40 androgenic alopecia subjects. Participants received either subdermal platelet‐rich plasma injections with 3 monthly sessions and booster 3 months later (group 1) or 2 sessions every 3 months (group 2). Folliscope hair count and shaft caliber, global photography, and patient satisfaction questionnaires were completed at baseline, 3‐month, and 6‐month visits. The authors reported statistically significant increases in hair count and shaft caliber in both groups at 6 months. Importantly, improvements occurred more rapidly and profoundly in group 1, indicating that platelet‐rich plasma injections should be administered first monthly 28. Alves and Grimalt 29 demonstrated significant differences in mean anagen hair and telogen hair count as well as telogen and overall hair density when compared to baseline. In a review of 16 studies comprising a total of 389 patients with androgenic alopecia, the majority demonstrated efficacy in promoting successful hair growth after 3–4 sessions on a monthly basis, followed by quarterly maintenance sessions 30. Platelet‐rich plasma is not curative for hair loss and must be continued long term for hair sustenance. However, patient satisfaction is typically very high and 60–70% of patients continue to undergo maintenance treatments. Due to the relatively recent introduction of platelet‐rich plasma injections for androgenic alopecia, there are no long‐term studies evaluating its effectiveness. Additionally, it is difficult to compare the efficacy with other remedies due to the lack of standardization in regard to platelet‐rich plasma kits, treatment fractions, and regimens, including the use of newer multi‐needle injectors.
While platelet‐rich plasma injections are considered safe when performed by a trained medical provider, these treatments are not suitable for everyone. Platelet‐rich plasma may not be appropriate for those with a history of bleeding disorders, autoimmune disease, or active infection, or those currently taking an anticoagulant medication. Although the majority of patients seem to tolerate the pain associated with scalp injections, some patients may prefer to avoid it.
How does hair grow?
Hair grows on most parts of the skin surface, except palms, soles, lips and eyelids. Hair thickness and length varies according to the site.
- Vellus hair is fine, light in colour, and short in length
- Terminal or androgenic hair is thicker, darker and longer
A hair shaft grows within a hair follicle at a rate of about 1 cm per month. It is due to cell division within the hair bulb at the base of the follicle. The cells produce the three layers of the hair shaft (medulla, cortex, cuticle), which are mainly made of the protein keratin (which is also the main structure of skin and nails).
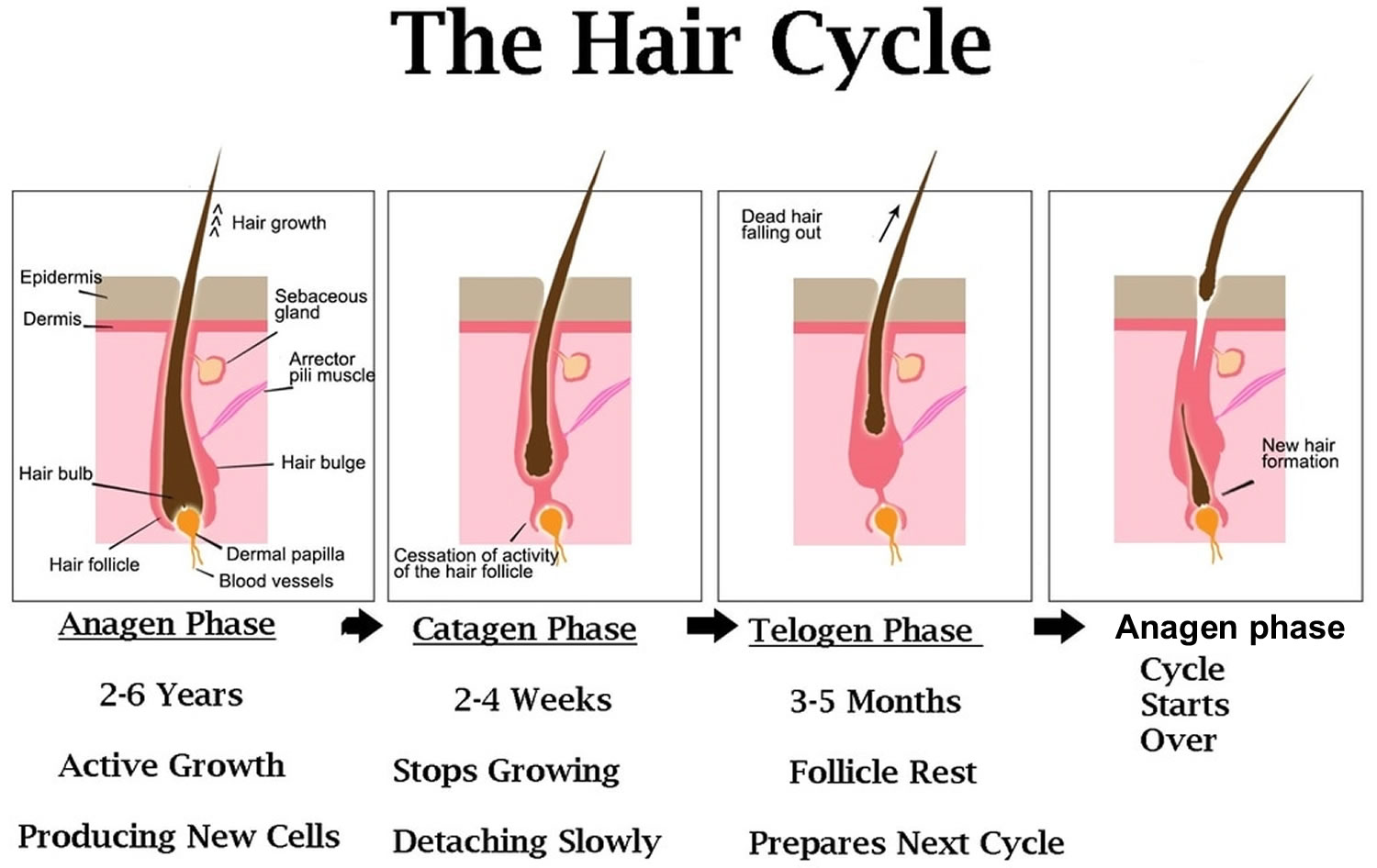
The human scalp contains about 100,000 hair follicles. These anchor the hair to the skin and contain the cells that produce new hairs (Figure 3).
Hair grows in 3 phases. However, these phases are not synchronized, and any hair may be at a particular phase at random.
The 3 main phases of the hair growth cycle are:
- The anagen or follicle growing phase starts growing the new hair (about 90% of hairs). This phase is genetically determined and can vary from 2 to 6 years (the average is just under 3 years). Most hair follicles on the scalp are in the anagen phase.
- The catagen phase (less than 10% of hairs) is a transition stage between the growing and resting phases and lasts 1-2 weeks.
- The telogen or resting phase (5% to 10% of hairs) is a mature hair with a root, which is held very loosely in the follicle. The telogen phase generally lasts about 4-5 months. About 100 telogen hairs are lost from the human scalp each day.
Because each hair follicle passes independently through the three stages of growth, the normal process of hair loss usually is unnoticeable. At any one time, approximately 85 to 90 percent of scalp hair follicles are in the anagen phase of hair growth. Hair follicles remain in anagen phase for an average of three years (range, two to six years) 35. The transitional or catagen phase of follicular regression follows, usually affecting 2 to 3 percent of hair follicles. Finally, the telogen phase occurs, during which 10 to 15 percent of hair follicles undergo a rest period for about three months. At the conclusion of this phase, the inactive or dead hair is ejected from the skin, leaving a solid, hard, white nodule at its proximal hair shaft 36. The hair growth cycle is then repeated.
Different causes of hair loss affect the hair follicles in different phases of growth. See below for the different types of hair loss (What are causes of hair loss?).
What is normal hair growth and hair loss?
Hair normally grows in cycles of two to six years. Each hair grows about one centimeter per month during a cycle. About 90 percent of your hair is growing, and about 10 percent is resting. After two to three months, the resting hair falls out and new hair starts to grow in its place. It is normal to lose up to 100 hairs each day. But, some people may have more hair loss than normal. This can be stressful, can lead to depression, and may affect self-confidence.
The scalp contains, on average, 100,000 hair follicles 37. You lose up to 100 hairs from your scalp every day. That’s normal, and in most people, those hairs grow back. A hair shaft grows within a hair follicle at a rate of about 1 – 1.5 cm per month. It is due to cell division within the hair bulb at the base of the hair follicle. The cells produce the three layers of the hair shaft (medulla, cortex, cuticle), which are mainly made of the protein keratin (which is also the main structure of skin and nails). Hair growth follows a cycle and the hair growth cycle is divided into three phases: anagen (active growing stage, about 90 % of hairs), catagen (degeneration stage, less than 10% of hairs) and telogen (resting stage, 5% to 10% of hairs). Hair is shed during the telogen phase. When telogen hairs are shed, new anagen hairs grow to replace them, beginning a new cycle 38, 39. These phases are not synchronized, and any hair may be at a particular phase at random. Hair length depends on the duration of anagen. Short hairs (eyelashes, eyebrows, hair on arms and legs) have a short anagen phase of around one month. Anagen lasts up to 6 years or longer in scalp hair. In addition to the ratio of anagen hair to telogen hair, the diameter of the hair follicles determines scalp coverage. Vellus hairs have a hair-shaft diameter of less than 0.03 mm, whereas terminal hairs have a diameter greater than 0.06 mm. The optimal hairs for scalp-hair growth and scalp coverage are anagen and terminal hairs.
Timespan of the hair growth cycle
- The anagen phase constitutes about 90% (1000 days or more) of the growth cycle. Anagen hairs are anchored deeply into the subcutaneous fat and cannot be pulled out easily.
- The catagen phase (10 days) and telogen phase (100 days) constitute only 10% of the hair growth cycle.
- During the catagen and telogen phase of the hair growth cycle, as hairs are at the shedding and rest-from-growth period, no bald spots are shown as hairs are randomly distributed over the scalp.
Anagen (active growing stage, about 90 % of hairs) stage
Your hair grows around 1 – 1.5 cm per month, faster in summer than in winter.
- The anagen stage is the growing period of a hair follicle.
- This stage typically lasts about 3 to 5 years. Asian hair can last 5-7 years
- Full length hair can be upto 100 cm long
Catagen (degeneration stage, less than 10% of hairs) stage
At the end of the anagen phase, your hair enters the catagen phase.
The catagen stage is the intermediate period of hair growth.
- Hair follicles prepare themselves for the resting phase.
- It lasts around 1-2 weeks.
- During this phase, the deeper portions of the hair follicles start to collapse.
Telogen (resting stage, 5% to 10% of hairs) stage
During the telogen phase each hair is released and falls out
- The telogen stage is the resting and shedding period of the hair cycle.
- The follicle remains inactive for 3 to 4 months.
- At the end of this period, older hairs that have finished their life will fall out and newer hairs will begin to grow.
- As compared with anagen hair, telogen hair is located higher in the skin and can be pulled out relatively easily. Normally, the scalp loses approximately 100 telogen hairs per day.
Hair loss, hair thinning and problems with hairgrowth occur when the growth cycle is interrupted/disrupted. This can be triggered by conditions such as nutritional and medical situations, illness or stress. For instance 6 weeks after intensive dieting or stress you can experience hair loss. This occurs because the growing stage (Anagen) is cut short and hairs enter the falling (Telogen) stage at the same time.
Figure 3. Hair growth cycle
What are causes of hair loss?
People typically lose 50 to 100 hairs a day. This usually isn’t noticeable because new hair is growing in at the same time. Hair loss occurs when new hair doesn’t replace the hair that has fallen out.
Hair loss is typically related to one or more of the following factors:
- Family history (heredity). The most common cause of hair loss is a hereditary condition that happens with aging. This condition is called androgenic alopecia (androgenetic alopecia), male-pattern baldness and female-pattern baldness. It usually occurs gradually and in predictable patterns — a receding hairline and bald spots in men and thinning hair along the crown of the scalp in women.
- Hormonal changes and medical conditions. A variety of conditions can cause permanent or temporary hair loss, including hormonal changes due to pregnancy, childbirth, menopause and thyroid problems. Medical conditions include alopecia areata, which is autoimmune hair loss and causes patchy hair loss, scalp infections such as ringworm, and a hair-pulling disorder called trichotillomania (traction alopecia or traumatic alopecia).
- If your thyroid gland is overactive or underactive, your hair may fall out. This hair loss usually can be helped by treating your thyroid disease.
- Hair loss may occur if male or female hormones, known as androgens and estrogens, are out of balance. Correcting the hormone imbalance may stop your hair loss.
- Many women notice hair loss about 3 months after they’ve had a baby. This loss is also related to hormones. During pregnancy, high levels of certain hormones cause the body to keep hair that would normally fall out. When the hormones return to pre-pregnancy levels, that hair falls out and the normal cycle of growth and loss starts again.
- Certain infections can cause hair loss. Fungal infections of the scalp (tinea capitis) can cause hair loss in adults and children. The infection is treated with antifungal medicines.
- Systemic diseases resulting in reversible patchy hair thinning, poor hair quality and bald patches include:
- Diabetes
- Iron deficiency
- Thyroid hormone deficiency (hypothyroidism)
- Systemic lupus erythematosus (SLE)
- Syphilis
- Severe acute or chronic illness.
- Dermatological disease resulting in reversible patchy hair thinning, poor hair quality and bald patches include:
- Localized alopecia areata
- A localized infection, such as tinea capitis
- Severe local skin disease, such as psoriasis, seborrhoeic dermatitis, atopic dermatitis, pityriasis rubra pilaris, cutaneous lupus erythematosus, cutaneous T-cell lymphoma
- Generalized skin disease (erythroderma).
- Medications and supplements. Hair loss can be a side effect of certain drugs, such as those used for cancer, arthritis, depression (antidepressants), birth control pills, vitamin A (if you take too much of it), heart problems, gout, blood thinners (anticoagulants) and high blood pressure. This type of hair loss improves when you stop taking the medicine.
- Radiation therapy to the head. The hair may not grow back the same as it was before.
- A very stressful event. Many people experience a general thinning of hair several months after a physical or emotional shock. This type of hair loss is temporary.
- Hairstyles and treatments. Excessive hairstyling or hairstyles that pull your hair tight, such as pigtails, cornrows or use tight hair rollers, the pull on your hair can cause a type of hair loss called traction alopecia. If the pulling is stopped before scarring of the scalp develops, your hair will grow back normally. However, scarring can cause permanent hair loss. Hot-oil hair treatments and chemicals used in permanents (also called “perms”) may cause inflammation (swelling) of the hair follicle cause the hair to fall out. If scarring occurs, hair loss could be permanent.
Hair loss can be subdivided into two main categories: scarring hair loss and non-scarring hair loss 40:
Non-scarring hair loss
Non-scarring hair loss falls into six major categories:
Androgenetic alopecia
Androgenetic alopecia is a pattern of hair loss that is affected by the genes and hormones (androgenic alopecia). Androgenetic alopecia is the most common form of hair loss in men and women and is a normal physiologic variant. Androgenetic alopecia is most prevalent in white men, with 30%, 40%, and 50% experiencing androgenetic alopecia at 30, 40, and 50 years of age, respectively (see Figure 1). Although androgenetic alopecia is less common in women, 38% of women older than 70 years may be affected (see Figure 2) 41. Androgenetic alopecia hair loss follows a gradual progressive course. Many patients with androgenetic alopecia have a family history of this condition. Hair thinning occurs in a sex-specific pattern.
Androgenic alopecia in men
Androgenic alopecia in men: bitemporal thinning of the frontal and vertex scalp, complete hair loss with some hair at the occiput and temporal fringes 42. Minoxidil and oral finasteride are the only treatments currently approved by the U.S. Food and Drug Administration for the treatment of androgenetic alopecia. Both of these drugs stimulate hair regrowth in some men, but are more effective in preventing progression of hair loss. Although there are a number of other treatments listed in various texts, there is not good evidence to support their use 43. Topical minoxidil (2% or 5% solution) is approved for the treatment of androgenetic alopecia in men. Hair regrowth is more robust at the vertex than in the frontal area, and will take six to 12 months to improve 42. Treatment should continue indefinitely because hair loss reoccurs when treatment is discontinued. Adverse effects include irritant and contact dermatitis. Finasteride (Propecia), 1 mg per day orally, is approved to treat androgenetic alopecia in men for whom topical minoxidil has been ineffective. Adverse effects of finasteride include decreased libido, erectile dysfunction, and gynecomastia (increase in the amount of breast gland tissue in men) 44.
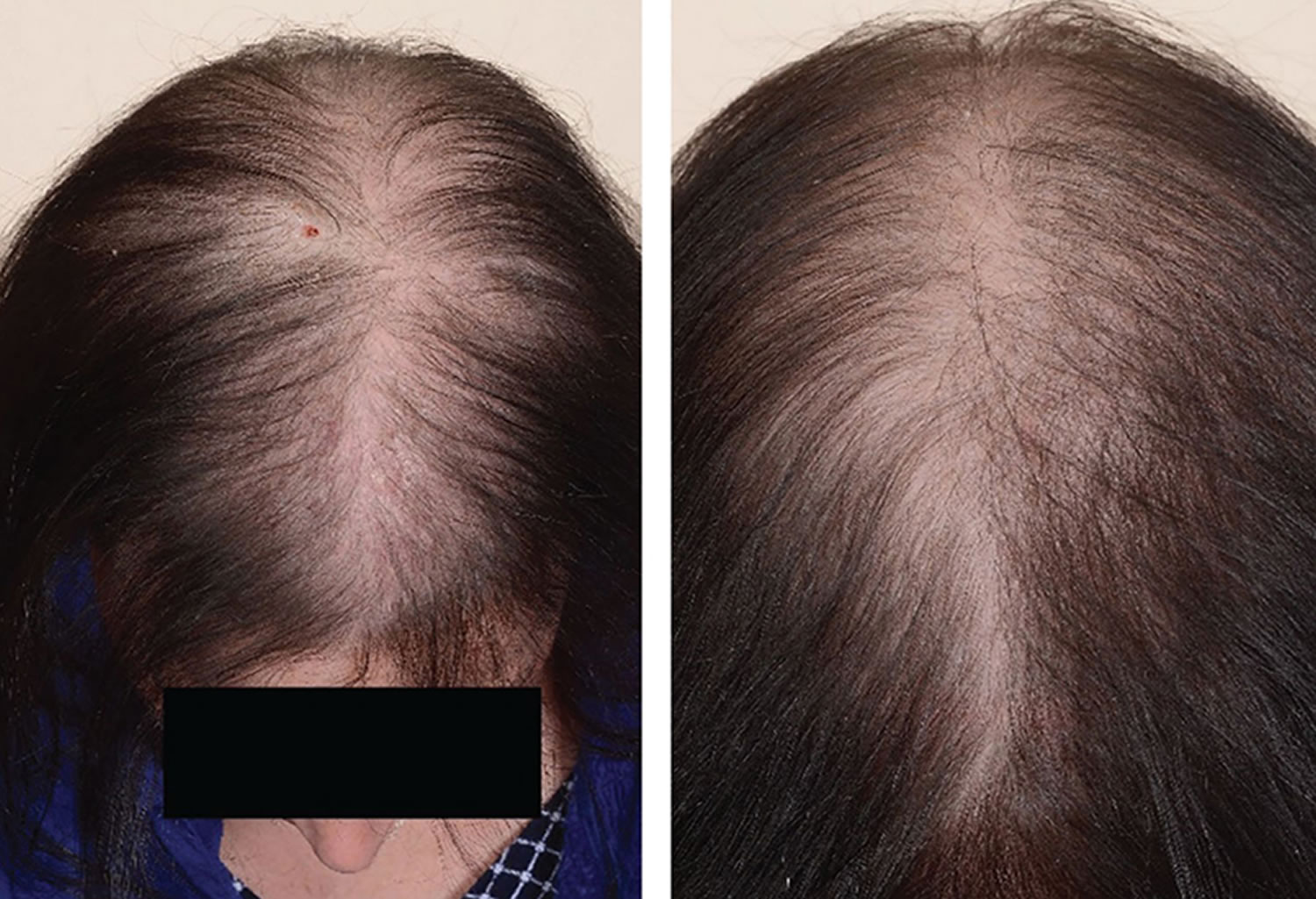
Androgenic alopecia in women
Androgenic alopecia in women: diffuse hair thinning of the vertex with sparing of the frontal hairline. Treatment involves topical minoxidil (2% solution). Adverse effects include irritant or contact dermatitis.
Alopecia areata
Alopecia areata also called autoimmune alopecia or autoimmune hair loss, is a common autoimmune skin disease, where your body’s immune system attacks your hair cells, causing patches of hair loss on the scalp, face and sometimes on other areas of your body 45. The term “alopecia” means hair loss and “areata” refers to the patchy nature of the hair loss that is typically seen with alopecia areata. Alopecia areata represent an attack on the hair roots by the body’s own immune system. Alopecia areata hair loss that can affect every part of the body, including the scalp, face, trunk, and extremities. When it affects only a portion of the body, it is called alopecia areata. When it affects an entire site, it is called alopecia totalis. When it involves the whole body, it is called alopecia universalis. The skin in these areas looks smooth. The cause is unknown, but it might be related to an autoimmune disease 46. The hair loss is usually fast, can happen at any age (mostly in young adults), and is more common in people with certain illnesses (such as diabetes and thyroid disease). In 80% of patients with a single bald patch, spontaneous regrowth occurs within a year. Even in the most severe cases of alopecia totalis and alopecia universalis, recovery may occur at some future date. This is an important difference between alopecia areata and the scarring forms of alopecia, which destroy the hair follicle and result in irreversible hair loss. Referral centers indicate that 34–50% of patients will recover spontaneously within 1 year, although most will experience multiple episodes of the alopecia, and 14–25% of patients will progress to alopecia totalis or alopecia universalis, from which full recovery is unusual (<10% of patients) 47.
Telogen effluvium
Telogen effluvium results from shifting of the hair cycle growth (anagen) phase towards the shedding (telogen) phase, you may lose 30 to 50 percent of your hair all at once. Clumps of hair come out in the shower or in hairbrush. Telogen effluvium is associated with physiologic or emotional stress. This stress may be a severe illness, injury, crash diet, or extreme mental stress. Your hair will usually grow back. Patients typically report significant hair loss and a decrease in hair volume (they commonly complain about their ponytail reducing in diameter) without well-defined alopecic patches. A pull test is typically positive 48. Telogen effluvium may result from an illness like hypothyroidism (underactive thyroid) or hyperthyroidism (overactive thyroid). Also, it can arise from stress like major surgery. A crash diet, poor feeding, and drugs can cause telogen effluvium 49. Telogen effluvium is usually self-limited and resolves within two to six months. Treatment involves removing the underlying cause and providing reassurance about the reversible nature of hair loss.
Traumatic alopecia
This is similar to traction alopecia, which results from forceful traction of the hair commonly seen in children. Also, trichotillomania is a type of traumatic alopecia in which the patient pulls on his/her hair repeatedly 50.
Tinea capitis (ringworm of the scalp)
Tinea capitis is a fungal infection of the scalp and hair shafts (scalp ringworm). It is caused primarily by the dermatophyte species Microsporum and Trichophyton 51. The fungi can penetrate the hair follicle outer root sheath and ultimately may invade the hair shaft. Tinea capitis also causes round patches of hair loss. The skin in these areas looks dry, red, and scaly. There may be swollen lymph nodes at the back of the lower head. Clinically, tinea capitis divides into inflammatory and non-inflammatory types. The non-inflammatory type usually will not be complicated by scarring alopecia. The inflammatory type may result in a kerion (painful nodules with pus) as well as scarring alopecia 52. Tinea capitis, a highly contagious infection, occurs primarily in children between 3 and 14 years of age, but it might affect any age group. It may also involve the eyelashes and eyebrows. The signs and symptoms of ringworm of the scalp may vary, but it usually appears as itchy, scaly, bald patches on the head. Tinea capitis can is treatable with systemic antifungal medications because topical antifungals do not penetrate hair follicles. The treatment is for 4 to 8 weeks. Topical treatment is not recommended, as it is ineffective 53.
- Trichophyton species: oral terbinafine (Lamisil), itraconazole (Sporanox), fluconazole (Diflucan), or griseofulvin
- Microsporum species: griseofulvin
Anagen effluvium
Anagen effluvium is a sudden loss of 80 to 90 percent of your hair that occurs during the anagen phase (growing phase) of the cell cycle due to an event that impairs the mitotic or metabolic activity of the hair follicle. Anagen effluvium often happens in people with cancer who are receiving chemotherapy or it can be an inherited or congenital condition, such as loose anagen syndrome. Patients typically present with diffuse hair loss that begins days to weeks after exposure to a chemotherapeutic agent and is most apparent after one or two months 54. In cancer patients who are receiving chemotherapeutic agents, short broken hairs and empty hair follicles may be observed. The incidence of anagen effluvium after chemotherapy is approximately 65% 55; it is most commonly associated with cyclophosphamide, nitrosoureas, and doxorubicin (Adriamycin). Other causative medications include tamoxifen, allopurinol, levodopa, bromocriptine (Parlodel), and toxins such as bismuth, arsenic, and gold. Other medical and inflammatory conditions, such as mycosis fungoides or pemphigus vulgaris, can lead to anagen effluvium 56. Anagen effluvium is usually reversible, with regrowth one to three months after cessation of the offending agent. Permanent alopecia is rare. No pharmacologic intervention has been proven effective. A large meta-analysis of clinical trials concluded that scalp cooling was the only intervention that significantly reduced the risk of chemotherapy-induced anagen effluvium 57. However, scalp cooling should be discouraged because it may minimize delivery of chemotherapeutic drugs to the scalp, leading to cutaneous scalp metastases 57. Minoxidil may help during regrowth period.
Scarring hair loss
Scarring hair loss is divided into four major types:
Tinea capitis
Tinea capitis: the inflammatory variety of tinea capitis (favus) may culminate with scarring hair loss.
Alopecia mucinosa
Alopecia mucinosa also known as follicular mucinosis: Alopecia mucinosa is a benign condition that occurs when mucinous material accumulates in the hair follicles and the sebaceous glands. The mucinous material causes an inflammatory response that hinders the growth of hair.
Alopecia neoplastica
Alopecia neoplastica: This is the metastatic infiltration of the scalp hair with malignant cells.
Frontal fibrosing alopecia
Frontal fibrosing alopecia is a form of scarring hair loss affecting the hair margin on the front of the scalp (i.e. the forehead and sideburns). This happens due to inflammation and destruction of the hair follicles. There may also be hair loss from the scalp near the ears and from the eyebrows. Sometimes hair loss can also occur from other parts of the body, but this is less common. Frontal fibrosing alopecia occurs mostly in white postmenopausal women but can occur in premenopausal women, men, and people of other ethnicities. Frontal fibrosing alopecia is thought to be a variant of another condition called lichen planopilaris. There are a number of treatments that are used for frontal fibrosing alopecia to help to slow down or halt further hair loss in some people. Unfortunately, their success is variable and some people cannot find a treatment that is effective for them. Treatments used to slow the progression of the condition include oral corticosteroids, intralesional steroid injections, anti-inflammatory antibiotics such as tetracyclines, or anti-malarial tablets (hydroxychloroquine). All these treatments aim to lower the activity of the immune system and slow down the attack on the hair follicles.
Hair loss prevention
There is no way to prevent male-pattern baldness or female-pattern baldness (androgenetic alopecia), because it is a genetic trait, meaning you inherited a gene for baldness from your parents. This type of hair loss is not preventable.
Some other causes of excessive hair loss can be prevented. These tips may help you avoid preventable types of hair loss:
- Be gentle with your hair. Use a detangler and avoid tugging when brushing and combing, especially when your hair is wet. A wide-toothed comb might help prevent pulling out hair. Avoid harsh treatments such as hot rollers, curling irons, hot-oil treatments and permanents. Limit the tension on hair from styles that use rubber bands, barrettes and braids.
- Ask your doctor about medications and supplements you take that might cause hair loss.
- Protect your hair from sunlight and other sources of ultraviolet light.
- Stop smoking. Some studies show an association between smoking and baldness in men.
- If you’re being treated with chemotherapy, ask your doctor about a cooling cap. This cap can reduce your risk of losing hair during chemotherapy.
Hair loss diagnosis
Before making a diagnosis, your doctor will likely give you a physical exam and ask about your diet, your hair care routine, and your medical and family history. You might also have tests, such as the following:
- Blood test. This might help uncover medical conditions that can cause hair loss.
- Pull test. Your doctor gently pulls several dozen hairs to see how many come out. This helps determine the stage of the shedding process.
- Scalp biopsy. Your doctor scrapes samples from the skin or from a few hairs plucked from the scalp to examine the hair roots under a microscope. This can help determine whether an infection is causing hair loss.
- Light microscopy. Your doctor uses a special instrument to examine hairs trimmed at their bases. Microscopy helps uncover possible disorders of the hair shaft.
Best vitamins and supplements for hair growth
A variety of vitamins and supplements for hair growth have appeared in the market over the past few years 58, 8, 59. Several prior studies have demonstrated vitamins, omegas 3 and 6 fatty acids, and antioxidants promote hair growth, suggesting a role of adequate nutrition in hair growth and supporting the use of dietary supplements for hair loss 60, 61, 62.
Nutrafol
Nutrafol launched in 2016 and is currently the fastest growing nutraceutical supplement for hair growth on the market 59. Nutrafol is composed of 21 ingredients, including a proprietary Synergen Complex®, which includes standardized phytoactives with clinically tested anti-inflammatory, stress-adaptogenic, antioxidant and dihydrotestosterone (DHT)-inhibiting properties 59. The phytocompounds in this complex include curcumin, piperine, ashwagandha, saw palmetto, and tocotrienols 63, 64. In addition to the components detailed above, Nutrafol also contain amino acids, marine collagen, hyaluronic acid, organic kelp, and vitamins and minerals that have been identified to play a role in the stress response as well as gut, thyroid and hair health 64. There are currently four formulations available: Nutrafol Women, Nutrafol Women’s Balance, Nutrafol Postpartum, and Nutrafol Men, which has a higher concentration of saw palmetto.
Curcumin (a yellow pigment found primarily in turmeric) is a potent anti-inflammatory and immunomodulating agent that has been shown to inhibit NF-kb and decrease tumor necrosis factor (TNF)-alpha and interleukin (IL)-1, inflammatory cytokines involved in follicular regression 64. Curcumin also inhibits androgen receptor expression, which is known to be overexpressed in follicles in androgenetic alopecia 65, 66. Co-administration with botanical piperine, found in black pepper and long pepper, enhances curcumin bioavailability and has been shown to increase plasma levels up to 154 percent after ingestion 64, 67. The stress response is known to play an important role in hair loss pathology and is intrinsically linked to alopecia areata (autoimmune hair loss) and telogen effluvium (hair loss associated with physiologic or emotional stress), with recent studies indicating that cortisol and micro-inflammation at the level of the hair follicle also plays a role in androgenic alopecia 68, 69.
Ashwagandha is a botanical that contains steroidal lactones which modulate and reduce cortisol levels. In a randomized, double-blind, placebo-controlled study of 98 patients, daily supplementation with 10% withanolide ashwaghanda showed statistically significant reductions in serum cortisol, serum C-reactive protein, blood pressure, and subjective feelings of stress compared to placebo 68. Saw palmetto extract is a natural inhibitor of both types I and II 5-alpha reductase which prevents conversion of testosterone to active DHT. A study of 100 men with mild to moderate androgenic alopecia who were treated with either 320 mg of saw palmetto or 1 mg of finasteride daily for two years revealed a significant improvement in 38 percent of patients taking saw palmetto and 68 percent of patients taking finasteride 70. Despite the increased efficacy of finasteride, saw palmetto might be a desirable alternative to avoid side effects of erectile dysfunction and falsely reducing prostate specific antigen (PSA) levels.5 While there are no reports of teratogenicity, saw palmetto is considered functionally related to finasteride, and therefore, it is considered unsafe in pregnancy. Vitamin E isoforms consist of four tocopherols and four tocotrienols, which are potent free radical scavengers 64. A randomized, placebo-controlled study of 38 patients with hair loss showed a statistically significant increase in hair counts of 38 percent from baseline compared to placebo 71. The authors concluded that the effect was most likely due to antioxidant activity, inhibition of lipid peroxidation, and oxidative stress in the scalp 71, 72.
The efficacy of Nutrafol to promote hair growth was studied in a six-month randomized, double-blind, placebo-controlled trial. Forty healthy women between the ages of 21 and 65 years old with self-perceived hair thinning were randomized into two groups, with 26 subjects receiving four capsules of Nutrafol daily and 14 subjects receiving placebo. The number of terminal and vellus hairs was analyzed based on phototrichograms of a 1 cm² area along the frontalis bone at Day 0, Day 90, and Day 180. Subjects taking Nutrafol showed an increased terminal hair count of 6.8 percent and 10.4 percent at 90 and 180 days, respectively, compared to 0.07 percent and 3.5 percent in the placebo group 64. Vellus hair counts increased by 10.1 percent and 15.7 percent at days 90 and 180 in the Nutrafol group compared to a decrease in vellus hair counts of 2.9 percent and 2.2 percent at days 90 and 180 in the placebo group 73. There was no statistically significant difference in mean hair shaft diameter between treatment and placebo groups at any point in the study 73. Investigator scores for hair growth and hair quality increased significantly from baseline to day 180. Eighty percent of subjects in the Nutrafol group reported a significant improvement in hair growth compared with 46.2 percent of placebo-treated subjects. Subjects taking Nutrafol also reported improvement in overall hair volume, noticeable new hair, hair growth rate, stress and anxiety levels, sleep quality, skin smoothness and skin health 73. The majority of subjects taking Nutrafol (73.1%) would recommend it to friends with hair loss.
A recent case series demonstrated clinical improvement in four subjects taking Nutrafol as a monotherapy 64. A 52-year-old woman who had previously failed a several month course of topical minoxidil showed increased hair density after seven months. A 45-year-old woman with early signs of diffuse pattern hair loss showed improved hair density after four months. A 37-year-old man with early pattern hair loss and a strong family history of hair loss who had previously failed minoxidil showed improved hair growth and decreased shedding. Lastly, a 38-year-old woman with early diffuse thinning of the temple areas experienced increased density after three months of daily Nutrafol use. No patient reported any side effects, and all were satisfied with their improvement 59.
Viviscal
Viviscal® is an oral supplement based on a novel marine complex formulation designed to promote hair growth in both men and women 74, 75. The key ingredients in Viviscal include a proprietary blend of shark and mollusk powder derived from sustainable marine sources (AminoMar® C marine complex), Equisteum arvense (natural occurring form of silica), Malpighia glabra (acerola cherry providing vitamin C), biotin (vitamin B7), and zinc. Other ingredients include calcium, iron, horsetail stem extract, millet seed extract, flaxseed extract, procyanidin B-2 (i.e., apple fruit extract), L-cystine and L-methionine, depending on the formulation. In addition to the original formulation, other formulations include Viviscal® Professional Strength and Viviscal® Man. Early studies evaluating a similar oral formulation of marine extracts in women with photodamaged skin demonstrated improvements in skin thickness, elasticity and erythema, as well as improvements in hair and nail brittleness after 90 days of treatment 76, 77. Following an initial open-label pilot study, several randomized, placebo-controlled trials have demonstrated the current oral marine complex supplement to be effective in promoting hair growth in both men and women 75, 78, 79, 80.
In the first randomized-controlled trial, 15 healthy women with Fitzpatrick Skin Types I to IV and self-perceived hair thinning were randomized to receive the Viviscal® Maximum Strength oral supplement or placebo twice daily for 180 days 79. A 2 cm² area of the scalp was selected for hair counts, which were performed at baseline and after 90 and 180 days of treatment. Mean number of terminal hairs in the treatment group increased from 271.0 to 571 and 609.6 at 90 and 180 days, respectively. In contrast, the mean number of terminal hairs in the placebo group decreased at 90 and 180 days. The mean number of vellus hairs did not significantly change in either group. After 90 days, more subjects in the treatment group reported improvements in overall hair volume, scalp coverage and hair thickness. Additional improvements noted at 180 days in the treatment group included increased hair shine, skin moisture retention, and skin smoothness. No adverse events were reported.
In another double-blind, placebo-controlled trial, 60 healthy women with Fitzpatrick Skin Types I to IV with self-perceived hair thinning were randomized to receive either the Viviscal® Extra Strength oral supplement or placebo twice daily for 90 days 75. Similar to the prior study, a predesignated 4 cm² area of the scalp was selected for evaluation at baseline and after 90 days. Additionally, ten terminal hairs in the target area were chosen and cut at the surface of the scalp to evaluate hair growth. Digital photographs were obtained to measure hair diameter and the diameter for ten hairs was used to obtain the mean hair diameter within the target area. At 90 days, there was a significant increase in mean number of terminal hairs from 178.3 to 235.8 in the treatment group compared to a smaller increase of 178.2 to 180.9 in the placebo group. The number of vellus hairs also increased in the treatment group from 19.6 to 21.2 compared to 19.8 to 19.9 in the placebo group. A significant decrease in the mean number of shed hairs from 27.1 to 16.5 was also observed in the treatment group (vs. 23.4 to 21.9 in the placebo group). No significant change in terminal hair diameter was observed in either group. In addition to the improvements in objective measures observed, subjects in the treatment group also had higher scores on a self-assessment questionnaire that rated overall hair quality including hair growth, hair volume, hair thickness, hair strength, eyebrow hair growth and scalp coverage as well as overall skin health. No adverse events were reported.
A subsequent double-blind, placebo-controlled trial evaluated the efficacy of the Viviscal® Professional Strength Oral Tablets, an oral formulation of the marine supplement containing procyanidin B-2 (i.e., apple fruit extract), L-cystine and L-methionine for the treatment of hair loss in women 81. Forty healthy women with Fitzpatrick Skin Types I to IV and with self-perceived hair thinning were randomly assigned to receive either the oral supplement or placebo twice daily for 180 days 81. A predesignated target area on the scalp was assessed using phototrichogram analysis to determine change in number of terminal and vellus hairs at 180 days. Terminal hair diameter and response scores to quality of life and self-assessment questionnaires were also evaluated. In the treatment group, the mean number of terminal hairs increased from 189.9 to 297.4 and 341.0 at 90 and 180 days, respectively, and mean number of vellus hairs increased from 19.9 to 20.2 and 22.8 at 90 and 180 days, respectively. Mean hair diameter also significantly improved in the treatment group from 0.06 mm to 0.07 mm and 0.067 mm at 90 and 180 days, respectively, which had not been found in previous studies of the prior formulation. There were no significant improvements in any of these parameters in the placebo group. Subjects in the treatment group reported greater scores on the quality of life and self-assessment questionnaires compared to the placebo group, indicating greater improvements in overall hair volume, scalp coverage, hair strength, nail strength, eyelash growth, skin smoothness, and overall skin health.
In another study, 96 healthy women with Fitzpatrick Skin Types I to III and self-perceived hair thinning were randomly assigned to receive the Viviscal® Oral Supplement or placebo three times daily for 180 days 78. The aim of this study was to add to the results of the initial double-blind, placebo-controlled trial by evaluating shed hair count analysis and hair diameter analysis using a phototrichogram. A predesignated area of the scalp was selected for two-dimensional digital images and trichoanalysis, which was performed at baseline, 90 days and 180 days. Shed hair was collected during shampooing and counted at each visit. Overall, mean hair shedding was significantly reduced in the treatment group from 52.1 to 42.6 and 42.7 at 90 and 180 days, respectively. In the placebo group, an initial increase in mean hair shedding was seen at 90 days, followed by a small decrease at 180 days. Mean vellus hair diameter showed a small but significant increase in the treatment group at 90 and 180 days, but no change was observed in the placebo group at either time point.
The studies discussed thus far consistently demonstrated the effectiveness of Viviscal® in promoting hair growth in women with self-perceived hair thinning 59. However, the efficacy of the oral supplement in men with hair loss had not yet been evaluated. In a double-blind, placebo-controlled trial, Ablon 80 evaluated the efficacy of Viviscal® Man, a reformulation of the original marine complex supplement for use in men. Sixty healthy men with clinically diagnosed male pattern hair loss were randomized to receive the reformulated supplement or placebo twice daily for 180 days 80. A predesignated target area on the midline scalp was chosen for two-dimensional digital images and trichoanalysis at baseline, 90 days and 180 days. A hair pull test on the right and left parietal, frontal and occipital scalp was also performed at baseline and 180 days. After treatment, subjects in the treatment group experienced significant improvement in all efficacy measures. Mean total hair increased from 162.2 to 169.1 and 174.9 at 90 and 180 days, respectively, total hair density increased from 159.7 to 166.5 and 172.2 at 90 and 180 days, respectively, and terminal hair density increased from 121.9 to 127.7 and 130.3 at 90 and 180 days, respectively 80. The hair pull test was also significantly improved in the treatment group at 180 days. No improvements in any of these parameters were observed in the placebo group. Subjects in the treatment group also reported significant improvements in several quality of life measures and self-assessment items including overall hair growth, hair volume, hair and nail strength, and overall skin healthy. No adverse events were reported.
Based on the current literature, Viviscal® appears to be effective in promoting hair growth and decreasing hair shedding in both men and women 80, 75, 81. Studies have also demonstrated that Viviscal® may offer the additional benefit of subjective improvements in the appearance and quality of the skin, nails and eyebrows 80, 81, 79. Theories behind the benefit of Viviscal® primarily involve providing adequate nutrition and vitamins that promote hair growth, as inadequate nutrition and various vitamin deficiencies have been associated with hair loss 82.
Advantages of Viviscal over current pharmaceutical therapies, such as topical minoxidil and oral finasteride, include additional improvements to skin and nail health and a more favorable side effect profile. In all of the studies performed thus far, no significant adverse events associated with the oral supplement have been reported 59. Therefore, Viviscal would likely be appropriate to use in the majority of patients. This is in contrast to many of the current pharmaceutical treatments for hair loss, such as topical minoxidil, which may cause local irritation at the application site, or oral finasteride, which has been associated in rare instances with sexual dysfunction in men and is often not favorable to use in women. Several Viviscal formulations contain biotin (vitamin B7), which may be a cause for concern. Recently, the FDA issued a warning that biotin might interfere with certain laboratory testing results, including endocrine and cardiovascular laboratory tests 83, 84. Given this warning, patients taking Viviscal® regularly should be aware of this possibility. However, Viviscal supplement contains about 100 to 240 micrograms of biotin compared to 5,000 micrograms found in most biotin supplements, which might have a smaller impact on laboratory tests compared to supplements containing higher amounts of biotin. Of note, the formulation for men does not contain any biotin 59.
Nourkrin
Nourkrin® is a newly developed hair growth product containing marine proteins extract, vitamins, minerals, acerola cherry extract, silica kieselguhr, horsetail extract and immunoglobulins 85. The active ingredients in Nourkrin are meant tonourish the hair follicles and ‘awaken’ the dormant hair follicles, thereby stopping hairloss, stimulating regrowth of new hair and strengthening the existing hair 85. However, the mechanism of action of Nourkrin is not known, but it might be related to the production of dihydrotestosterone (DHT) in the hair follicle. Dihydrotestosterone (DHT) is known to cause hair loss. Studies have been initiated to investigate this aspect. Nourkrin has been shown, in open pilot studies carried out as part of the product development process, to have a favourable effect on hair loss 85. In a randomized, double-blind, placebo-controlled study involving 60 subjects (55 males and 4 females [there were 5/60 (8.3%) withdrawals/losses to follow‐up: 3/30 (10%) in Nourkrin® group, 2/30 (6.7%) placebo group]) taking 2 capsules/day (< 80 kg in body weight) or 3 capsules/day (> 80 kg) for 6 months, the average hair growth increase was 35.7% in the group using Nourkrin and 1.5% in the placebo group 85, The results of this study show that the subjects reported favourable effects on hair gain with Nourkrin® compared with placebo and the same effect was also detectable in the open long-term (6 months or more) study.
The results obtained in the present study compare favourably with results obtained in studies with the drugs minoxidil and finasteride 85. The positive effect of Nourkrin combined with its excellent tolerability may make this product an attractive alternative treatment for people with hair-loss problems.
Biotin (vitamin B7)
Biotin also known as vitamin B7 or vitamin H, is a B-complex water-soluble vitamin that helps turn the carbohydrates, fats, and proteins in the food you eat into the energy you need 86. Biotin (vitamin B7) is a cofactor for five carboxylase enzymes (propionyl-CoA carboxylase, pyruvate carboxylase, methylcrotonyl-CoA carboxylase [MCC], acetyl-CoA carboxylase 1, and acetyl-CoA carboxylase 2) that catalyze critical steps in fatty acid synthesis, amino acid catabolism, gluconeogenesis, and mitochondrial function in hair root cells 87, 88, 89. Biotin also plays key roles in histone modifications, gene regulation (by modifying the activity of transcription factors), and cell signaling 88.
Foods that contain the most biotin include organ meats, eggs, fish, meat, seeds, nuts, and certain vegetables (such as sweet potatoes) 89. The biotin content of food can vary; for example, plant variety and season can affect the biotin content of cereal grains, and certain processing techniques (e.g., canning) can reduce the biotin content of foods 90. Although overt biotin deficiency is very rare 91, 90 and severe biotin deficiency in healthy individuals eating a normal mixed diet has never been reported 92. The human requirement for dietary biotin has been demonstrated in three different situations: prolonged intravenous feeding (parenteral) without biotin supplementation, infants fed an elemental formula devoid of biotin, and consumption of raw egg white for a prolonged period (many weeks to years) 93.
The signs and symptoms of biotin deficiency typically appear gradually and can include thinning hair with progression to loss of all hair on the body; scaly, red rash around body openings (eyes, nose, mouth, and genital area); conjunctivitis; ketolactic acidosis (which occurs when lactate production exceeds lactate clearance) and aciduria (abnormal amounts of acid in urine); seizures; skin infection (candidiasis); brittle nails; neurological findings (e.g., depression, lethargy, hallucinations, ataxia and numbness and tingling of the extremities) in adults; and hypotonia, lethargy, hearing loss, vision loss and developmental delay in infants, older children and adolescents 94, 89, 88, 92. Once vision problems, hearing loss, and developmental delay occur, they are usually irreversible, even with biotin therapy 94. The rash and unusual distribution of facial fat in people with biotin deficiency is known as “biotin deficiency facies” 95, 92. Individuals with hereditary disorders of biotin metabolism resulting in functional biotin deficiency often have similar physical findings, as well as seizures and evidence of impaired immune system function and increased susceptibility to bacterial and fungal infections 96, 97.
Therefore, biotin supplements are often promoted for hair, skin, and nail health. However, these claims are supported, at best, by only a few case reports and small studies. Only case reports are available to support claims that biotin supplements can promote hair health, and these reports were only in children 98, 99. These studies found that 3–5 mg/day biotin in children with uncombable hair syndrome (a rare disorder of the hair shaft) significantly improved hair health after 3–4 months. The evidence supporting the use of biotin supplements to support skin health is equally limited to a small number of case reports, all in infants, showing that 100 mcg to 10 mg/day resulted in dramatic improvements in rash or dermatitis as well as hair loss 100, 101.
Biotin deficiency (< 100 ng/L) and suboptimal biotin levels (100–400 ng/L) were reported in 38% and 49% of healthy women complaining of hair loss, respectively. However, 11% of these patients were later found to have a secondary cause for low biotin levels, including use of antiepileptics, isotretinoin, antibiotics, or gastrointestinal disease altering the biotin-producing gut microflora 102.
Although highly popularized in the media for its beneficial effects on hair loss, there have been no randomized controlled trials to evaluate the effect of biotin supplementation in hair loss 103. Patients taking isotretinoin or valproic acid have decreased biotinidase levels, the enzyme responsible for releasing biotin from food. As a result, isotretinoin-associated telogen effluvium and alopecia secondary to valproic acid may benefit from biotin supplementation 104, 105. A recent review identified 11 cases of hair loss secondary to biotin deficiency, from either an inherited enzyme deficiency or medication, where biotin was an effective supplementation for hair regrowth 106. Current clinical evidence supports biotin supplementation as an effective therapy for hair loss only in cases secondary to biotin deficiency; however, apart from medications, this is rare in developed countries due to well-balanced dietary intake 86.
Clinicians should exercise caution in recommending biotin as an oral supplement for hair loss because it may interfere with thyrotropin and thyroid hormone assays, resulting in artificially high or low thyroid function results 83, 84. Many immunoassays used in diagnostic tests rely on the binding of biotin with streptavidin to improve test sensitivity; high levels of serum biotin can compete with these immunoassays. Multiple cases have been reported in which patients taking high levels of biotin had laboratory results indistinguishable from Graves disease (an autoimmune disorder that can cause hyperthyroidism, or overactive thyroid), and as a result were unnecessarily treated with antithyroid medications 107, 108. Biotin may also interact with troponin, N-terminal pro-brain natriuretic peptide, and parathyroid hormone assays, underscoring the need for a comprehensive medication history at every patient visit, including the use of over-the-counter supplements 109.
Vitamin D
Vitamin D also referred to as calciferol, is a fat-soluble vitamin that plays an important role in calcium and phosphorus homeostasis, as well as immune regulation 110. The flesh of fatty fish (such as trout, salmon, tuna, and mackerel) and fish liver oils are among the best sources of vitamin D 111. Vitamin D is also produced inside your body when ultraviolet (UV) rays from sunlight strike your skin and trigger vitamin D synthesis 112. Vitamin D dysregulation may contribute to autoimmune diseases, including rheumatoid arthritis, systemic lupus erythematosus, and multiple sclerosis 113. Vitamin D receptors are intracellular receptors expressed in hair follicles, essential for normal hair cycle and differentiation of the interfollicular epidermis. Homozygous knockout of vitamin D receptor in mice resulted in the development of hair loss and near total hair loss at 8 months 114.
Meta-analyses have reported that alopecia areata patients have a higher prevalence of vitamin D deficiency and lower 25-hydroxyvitamin D serum levels compared to healthy controls 115. One study reported the presence of vitamin D deficiency in 39% of alopecia areata patients compared to 12.79% of healthy controls 116. Given this association, multiple studies have investigated the efficacy of oral vitamin D supplementation for alopecia areata treatment; however, they failed to support its benefit in alopecia totalis or alopecia universalis or its ability to potentiate squaric acid dibutylester 117, 118. A case report of presumed alopecia areata associated with reduced vitamin D receptor expression reported a complete clinical remission after topical calcipotriol ointment 50 μg/mL applied once daily for 3 months 119. Alopecia areata patients using topical 0.005% calcipotriol cream twice daily for 12 weeks had greater than 50% hair regrowth in 65% of patients, greater than 75% hair regrowth in 62.5% of patients, and complete regrowth in 27.1% 120. Twice daily topical 0.005% calcipotriol in 22 patients with patchy alopecia areata resulted in 59.1% of patients demonstrating hair growth within 4.21 ± 2.13 weeks. Patients with lower baseline serum vitamin D levels responded faster and more robustly 121. Studies using topical vitamin D in alopecia are inconsistent and limited by small sample size or lack of appropriate controls. Preliminary results suggest a potential therapeutic benefit for topical vitamin D, with minor side effects 58.
Vitamin E derivatives (Tocotrienols)
Vitamin E consists of fat-soluble compounds known as tocopherols and tocotrienols that function as antioxidants by scavenging peroxyl radicals 122. Eight months of supplementation with 50 mg of mixed tocotrienols and 23 IU of alpha-tocopherol resulted in 34.5% increased hair count in 38 patients with hair loss, compared to a 0.1% decrease with placebo. The ability of the derivatives to inhibit lipid peroxidation may limit hair follicle oxidative stress, thus preventing hair loss; however, additional studies are needed 72.
Amino acids
A variety of amino acids have been studied for the treatment of hair loss. Most notably, cystine and lysine have been evaluated in humans 58. Other amino acids, including methionine and arginine, are often included in hair nutraceuticals, but have not yet been evaluated in clinical studies 58.
Cysteine plays a central role in hair health; it forms dimers that are oxidized to produce cystine, creating disulfide bridges that provide strength and rigidity between keratin strands. In disorders with decreased cysteine, such as trichothiodystrophy (is a rare inherited disease that affects many parts of the body and the hallmark of this condition is brittle hair that is sparse and easily broken), there is a deficiency of sulfur containing amino acids, and hair is brittle 123. In homocystinuria, patients have thin and hypopigmented hair 124. Many nutraceuticals contain cysteine rather than cystine, in particular N-acetyl-L-cysteine (NAC), as it is better absorbed than any other cysteine product 125.
Oral L-cystine (70 mg) in combination with retinol was evaluated for the treatment of diffuse hair loss, with increases seen in both hair density and anagen rate 126. Oral L-cystine (unknown dose) was also studied in combination with histidine, copper, and zinc taken 4 times daily, resulting in a significant mean change in total hair count after 50 weeks (29 vs. 11% for placebo) in 24 patients with androgenic alopecia 127. Millet seed containing amino acids, silicic acid, several B vitamins, and dietary minerals including manganese in combination with L-cystine (2 mg), and calcium pantothenate (Priorin®; Bayer Inc., Mississauga, ON, Canada) taken twice daily for 6 months showed a significantly increased anagen rate in 40 female patients 128. Supplementation with L-cystine (20 mg), medicinal yeast, pantothenic acid, thiamine, keratin, and para-aminobenzoic acid (Pantogar®; Merz Pharmaceuticals GmbH, Raleigh, NC, USA) in 30 women with telogen effluvium resulted in significant improvement and normalization of the mean anagen hair rate after 6 months compared with placebo 129.
Lysine is an essential amino acid found in meat and eggs, and is thought to play a role in the absorption of iron. In patients with chronic telogen effluvium, supplementation with L-lysine (1.5 g), iron (72 mg), vitamin B12, vitamin C, biotin, and selenium (Florisene®; Lambers Healthcare Ltd, Kent, UK) resulted in a significant 39% reduction in hair shedding after 6 months, as well as a significant increase in serum ferritin levels in women who had previously failed with iron supplementation alone 130.
Methionine is another essential amino acid that is vital for both keratin and procollagen synthesis. Studies have suggested a role for L-methionine in slowing the onset of grey hair in an in vitro (test tube) model by counteracting hydrogen peroxide-mediated oxidative stress and blunting of methionine sulfoxide repair. Currently, there are no in vivo studies in human subjects demonstrating a benefit for the treatment of hair loss 131.
Arginine is a nonessential amino acid that is protective against the negative effects of hydrogen peroxide on hair proteins and hair surface lipids secondary to oxidative coloring or bleaching 132. However, there are no studies that support arginine supplementation for the treatment of hair loss. Overall, the data regarding the use of amino acids to treat hair loss is limited. All of the trials reviewed here combined amino acids with various other supplements. Therefore, no definitive conclusions can be drawn regarding the effect of amino acids on hair loss.
Caffeine
Caffeine is an alkaloid methylxanthine and functions as a phosphodiesterase inhibitor, promoting cellular proliferation. In vitro studies (test tube studies) report that caffeine counteracts the inhibitory effects of testosterone on hair growth, promotes hair shaft elongation, prolongs anagen duration, and stimulates hair matrix keratinocyte proliferation 133. Caffeine also downregulates testosterone-induced transforming growth factor (TGF)-β1 expression, a hair growth inhibitor, and increases expression of insulin-like growth factor (IGF)-1, a hair growth promoter. Female hair follicles appear to have a higher sensitivity to caffeine 134.
Research has focused on the use of topical caffeine for the treatment of androgenetic alopecia. Six months of daily caffeine shampoo use (Alpecin caffeine shampoo C1, unknown concentration, 7 mL, left on scalp for 2 min) resulted in fewer hairs extracted on the hair-pull test and hairs shed during combing, with reduced speed of hair loss progression and overall hair loss intensity 135, 136, 137. Caffeine lotion (unknown concentration) has also been tested with similar results, including decrease in hairs released during the hair-pull test and positive treatment response in 75% of patients at 2 months and 83% at 4 months 138. Comparison of 0.2% caffeine topical liquid to 5% topical minoxidil demonstrated noninferiority (n = 210) with 10.59% improvement in anagen ratio compared to 11.68% using minoxidil 139.
Topical caffeine has also been studied in combination with conventional therapies. A topical solution containing both caffeine and 2.5% minoxidil was compared to 2.5% minoxidil alone in 60 patients with androgenetic alopecia. After 120 days of treatment, the combined solution was more effective than minoxidil alone, with 58.33% of patients satisfied versus 41.37% in the minoxidil cohort 140. Another topical solution containing 1% caffeine, 5% minoxidil, and 1.5% azelaic acid was more effective for hair regrowth and decreased shedding after 32 weeks of treatment compared to 5% minoxidil alone or placebo 141. Topical caffeine shows potential for hair loss; however, studies are limited by lack of quantitative, standardized evaluation 142.
Capsaicin
Capsaicin, via activation of vanilloid receptor-1 and release of calcitonin gene-related peptide from sensory neurons, upregulates IGF-1 and inhibits TGF-β, which induces apoptosis of keratinocytes through the phosphatidylinositol 3-kinase/Akt pathway 143. Oral capsaicin 6 mg and isoflavone 75 mg daily for 5 months increased serum IGF-I in patients with androgenetic alopecia and alopecia areata in comparison to those who received placebo. Hair growth occurred in 64.5% of treated patients versus 11.8% of controls. In androgenetic alopecia specifically, 88% observed hair growth following treatment 143.
Capsaicin cream 0.075% applied daily to affected scalp in patients with extensive alopecia areata resulted in growth of vellus hairs at day 21 144. In addition, half of patients (12 alopecia areata and 2 alopecia totalis) in a small prospective trial reported hair growth after three weeks of 0.075% topical capsaicin cream 58. Comparing topical capsaicin ointment to clobetasol 0.05% ointment in 50 alopecia areata patients showed an improvement in vellus hair growth, but no significant cosmetic hair regrowth 145. Further research shows that topical 0.01% raspberry ketone, which has a structure similar to capsaicin, also upregulates IGF-I and promotes hair growth in 50% of patients 146.
Curcumin
Curcumin also called diferuloylmethane (1,7-bis(4-hydroxy-3-methoxyphenyl)-1,6-heptadiene-3,5-dione), is the active ingredient of turmeric (Curcuma longa), has been used for centuries as an anti-inflammatory agent. Curcumin downregulates cyclooxygenase-2, lipoxygenase, and inducible nitric oxide synthetase enzymes and inhibits nuclear factor-kB signaling, thereby decreasing proinflammatory cytokines such as tumor necrosis factor (TNF)-α and interleukin (IL)-1. Additionally, TNF-α and IL-1 are involved in follicular regression. Curcumin also has antioxidant, antimicrobial, antineoplastic, and antiandrogenic properties 147.
A 5% topical hexane extract of Curcuma aeruginosa (pink and blue ginger) was compared to placebo, 5% minoxidil, and combination Curcuma aeruginosa and minoxidil in 87 patients. After 6 months, no significant improvement in total area hair count was noted in any group versus placebo. On photographic review, combination therapy of Curcuma aeruginosa and 5% minoxidil showed significant improvement, while subjective assessment of hair regrowth/shedding was only significantly improved in the combination group. While this study does not support the efficacy of Curcuma aeruginosa extract alone for hair growth, it does suggest a synergistic effect when used in combination with minoxidil. In fact, Curcuma aeruginosa (pink and blue ginger) has been shown to increase epidermal penetration of minoxidil, possibly improving drug delivery in androgenetic alopecia 148, 149.
Garlic Gel
Garlic (Allium sativum) belongs to the Allium genus along with onions, scallions, shallots, leeks, and chives. These plants produce organosulfur compounds, which have antimicrobial, immunomodulatory and anti-inflammatory effects 150. In a trial of 40 alopecia areata patients, topical 5% garlic gel in combination with betamethasone was evaluated in comparison to placebo. After 3 months, good to moderate responses were observed in 95% of those treated compared to 5% with placebo. No adverse effects were reported 151. Further investigation is needed to define the effect of topical garlic for hair loss.
Marine Proteins
Marine proteins, including extracellular matrix components from sharks and mollusks, have been produced for over 15 years to enhance hair growth. Initial studies with this marine complex supplement for the treatment of androgenetic alopecia were performed in the 1990s. Scandinavian researchers first described the exceptionally healthy skin and hair of the Inuit peoples to be a result of their fish- and protein-rich diet 152. Lassus and Eskelinen 152 conducted a 6‐month, controlled, randomized, double‐blind, parallel‐group study of 20 male subjects with hereditary androgenic alopecia receiving once daily marine complex supplement vs. 20 male subjects receiving once daily fish extract. The marine complex supplement group showed a mean increase in non‐vellus hair of 38% compared with a 2% increase in the fish extract group. Moreover, 19 subjects in the marine complex supplement group showed both clinical and histological improvement, while subjects in the fish extract group did not. More recently, in a 6‐month, double‐blind clinical trial, adult male subjects with thinning hair were randomized to marine complex supplement or placebo administration twice daily 80. Subjects taking the marine complex supplement experienced decreased shedding and increased hair growth (total hair count, total hair density, and terminal hair density) at 180 days. Digital photography, trichoanalysis, and investigator assessments demonstrated significant improvements in terminal and vellus hair count. Hair pull test results were also lower in the marine complex supplement group. Similar research conducted in female patients also has shown promising results 75, 79. These studies revealed that a marine complex supplement has efficacy in treating androgenic alopecia. Based on known side effects from its active ingredients, the marine proteins supplement may have the potential to cause arthralgias, bloating, constipation, diarrhea, nausea, and allergic reaction but none of these have been seen in clinical trials 153.
Originally marketed as Hairgain® (Parexel, Norway) and later Viviscal® (Hair Nourishment System: Lifes2good, Inc., Chicago, IL, USA), marine peptide complexes are thought to enhance the proliferation of dermal papillae cells and increase levels of alkaline phosphatase, an indicator of anagen phase 81. As summarized in a recent review, eight clinical trials and seven cohort studies demonstrated the ability of proprietary marine proteins, lipids, and glycosaminoglycans to promote growth of terminal and vellus hairs, increase hair shaft diameter, and decrease hair loss 154, 155, 156, 157, 158, 159, 160, 78, 75, 80, 152, 81.
Two recent trials by Ablon and colleagues 78, 80 reported a significant improvement in hair counts and hair volume with the use of oral marine protein supplement. Using oral marine protein supplement 3 times daily in 100 females with self-perceived hair loss for 6 months demonstrated a significant increase in the mean hair diameter of vellus-like hairs, as well as a reduction in shedding 78. Sixty males with androgenetic alopecia were treated with oral marine protein supplement twice daily for 6 months, resulting in significant increases in total hair count, total hair density, and terminal hair density, as well as fewer hairs extracted on the hair-pull test 80. Marine protein supplement appears to be an effective, well-tolerated for patients with hair loss with no adverse events reported. Shellfish allergy is a contraindication to marine protein supplement use and detailed allergy history of prospective patients is warranted 58.
Melatonin
Melatonin is a neurohormone secreted by the pineal gland that regulates mammalian circadian rhythm. Melatonin is also an antioxidant synthesized in hair follicles 161. The first study 162 to report the effects of melatonin on hair growth compared topical melatonin 0.1% solution daily for 6 months in 40 patients with androgenetic alopecia or diffuse alopecia; efficacy was evaluated by trichograms to assess anagen and telogen hair. After treatment with melatonin, patients with diffuse alopecia had a significant increase in anagen hair at the occiput versus the frontal hairline 162.
A follow-up study 163 evaluated a 0.0033% topical melatonin solution in an open-label study of 15 women and 15 men with androgenetic alopecia. After treatment, the degree of alopecia severity was significantly reduced. Using the same melatonin solution for 6 months in 35 men with androgenetic alopecia, TrichoScan evaluation demonstrated a 29.2% increase in the hair count in 54.8% of patients after 3 months, and a 42.7% increase in 58.1% of patients after 6 months. Hair density improved 29.1% in 54.8% of patients, and 40.9% in 58.1% of patients after 3 and 6 months, respectively 163. Continued research using 0.0033% topical melatonin solution, including a large multicenter study, demonstrated improvements in hair texture, decreased hair loss, and a reduction in seborrheic dermatitis 58.
Onion Juice
Although the mechanism of topical onion juice (Allium cepa L.) in alopecia areata is unknown, it is thought that sulfur and phenolic compounds cause an irritant contact dermatitis, stimulating hair regrowth through antigenic competition. The effectiveness of topical crude onion juice in the treatment of alopecia areata compared to tap water was evaluated in 62 patients (45 treatment, 17 placebo) 164. At 8 weeks 87% of patients treated with onion juice demonstrated full hair regrowth versus 13% using water. The most common reported adverse effect was unpleasant odor 164.
Procyanidin
Procyanidins are a class of flavonoids found mainly in plants, including apples, barley, cocoa, cinnamon, grapes, and tea, described to have antioxidant, anti-inflammatory and antifungal capabilities 165. Procyanidins also induce anagen phase in murine hair models 166. Topical 1% procyanidin B2, derived from apple juice, resulted in a significant increase in total and terminal hair counts at 4 months and 6 months in 29 patients with androgenetic alopecia compared to placebo 165, 167. Procyanidin 0.7% used to treat 43 men with androgenetic alopecia also demonstrated a significant increase in hair counts (3.3 vs. −3.6 for placebo) after 6 months, with a total increase of 23 hairs/cm² after 12 months 168.
Oral procyanidin supplementation (400 mg) was investigated in a double-blind, placebo-controlled randomized clinical trial conducted in 250 patients with androgenetic alopecia 169. This procyanidin combination is extracted from Annurca, an apple variety native to Southern Italy with one of the highest contents of oligomeric procyanidins (specifically procyanidin B2). The supplement can be prepared with the addition of biotin, zinc, and selenomethionine (AMSbzs) or without (AMS). Both supplements lead to improvement in all hair clinical parameters, with the biotin, zinc, and selenomethionine (AMSbzs) cohort demonstrating increased hair density by 125.2%, hair weight by 42.1%, and keratin content by 40.1% at 2 months 169. Procyanidins also upregulate MAPK/extracellular signal-related kinase kinase (MEK) in hair epithelial cells and counteract hair growth inhibitory effects of TGF-β in vitro, which may account for these clinical results 170, 171. Additional studies are warranted to further define the effect of procyanidins on hair growth.
Pumpkin Seed Oil
Pumpkin (Cucurbita pepo) is a member of the squash family native to North America. Pumpkin seed oil contains phytosterols known to inhibit 5-alpha-reductase, preventing the conversion of testosterone to active dihydrotestosterone (DHT) 172. Comparing 400 mg of oral pumpkin seed oil daily to placebo for 24 weeks in 76 male patients with androgenetic alopecia demonstrated a mean increase in hair count of 40 versus 10% with placebo, with improved patient-reported satisfaction scores 173. Mean hair count among the treatment group increased by 40% while mean hair count among the placebo group increased by 10% 173. However, this Korean pumpkin seed oil supplement (Octa Sabal Plus®; Serona Company, South Korea) contains additional ingredients derived from mixed vegetables, primrose, red clover, and tomatoes, making it unclear whether the effects are due mostly to the pumpkin seed oil component 173. In addition, this study showed that pumpkin seed oil is a promising treatment for androgenetic alopecia involving the vertex, but failed to address the supplement’s effect on frontal variants. Additional studies are required to confirm these preliminary results of the effects of pumpkin seed oil on hair growth.
Rosemary Oil
Rosemary (Rosmarinus officinalis L.) is an aromatic evergreen herb with antioxidant, antibacterial, antifungal, and anti-inflammatory properties such as increasing prostaglandin E2 production, and decreasing leukotriene B4 production 174, 175. Rosemary also enhances microcapillary perfusion.
In a randomized controlled trial with 100 androgenetic alopecia patients, topical rosemary oil lotion (3.7 mg/mL) applied daily was noninferior to topical 2% minoxidil 176. 50 subjects were assigned to each treatment group, and they were observed for a 6‐month period with microphotographic assessments. Both groups experienced a significant increase in hair count at the 6‐month endpoint compared to the baseline and 3‐month endpoint 176. The most common adverse effect reported was scalp itching, more frequent with minoxidil use 176. Rosemary oil appeared to be a safe nonprescription alternative for androgenetic alopecia, and the results of this study merit further investigation.
Tea tree oil
Tea tree oil also known as Melaleuca oil, Australian tea tree oil, tea tree essential oil or tea tree, is the essential oil extracted using steam distillation of the leaves and terminal branches of Melaleuca alternifolia (tea tree), a tree native to Australia 177. Herbal medicines containing tea tree oil preparations are usually available in liquid or semi-solid forms to be applied to the skin and in liquid forms to be applied to the lining of the mouth. Tea tree oil preparations may also be found in combination with other herbal substances in some herbal medicines. Tea tree oil is used as a topical antibacterial, antifungal, antiviral, anti-inflammatory and analgesic properties due to the presence of terpineol (terpinen-4-ol) and α-terpineol 178. Tea tree (Melaleuca alternifolia) is an Australian species from the northern coast with a high content of terpineol (terpinen-4-ol) (more than 30% of gross weight) and low content of cineole (lower than 15%) 179. Terpinen-4-ol is the main antimicrobial component but other components, such as α-terpineol, also have antimicrobial activities similar to those of terpinen-4-ol 180. Some of the chemical and physical properties of tea tree oil components are shown in Tables 1 and 2 below. The concentrations of tea tree oil found in commercially available products range from 2 to 5% 181. Although the oil comprises a variable and complex mixture of over 100 components, its composition has been described as being approximately a 50/50 blend of oxygenated and nonoxygenated monoterpenes 182. The essential oil from the Melaleuca leaves, because of its antinflammatory and antibacterial properties, is widely used in the traditional medicine among the native Australian population to treat infections of the urinary tract, fingernails, skin, and acne 183. Due to tea tree oil components, such as terpinen-4-ol, α-terpineol, linalool, α-pinene, β-pinene, β-myrcene and 1,8-cineole, tea tree essential oil has demonstrated an high degree of antibacterial effect against a wide range of bacteria (Staphylococcus aureus, Staphylococcus epidermidis, Streptococcus spp, vancomycin resistant enterococci [VRE], Acinetobacter baumanni, Escherichia coli, Klebisella pulmonae), fungi (Candida spp, Malassezia spp) and protozoa (Trichomonas vaginalis) 184. Tea tree oil has also been used in a spray form to control castor bean ticks (Ixodes ricinus) and poultry red mites (Dermanyssus gallinae) in poultry houses 184. The composition of tea tree oil may change considerably during storage, with ρ-cymene levels increasing and α- and γ-terpinene levels declining 185. Light, heat, exposure to air, and moisture all affect oil stability, and tea tree oil should be stored in dark, cool, dry conditions, preferably in a vessel that contains little air.
In a double‐blind, randomized, placebo‐controlled study, subjects receiving a microemulsion of minoxidil, diclofenac, and tea tree oil, vs. minoxidil alone or placebo, demonstrated an earlier response in androgenic alopecia treatment 186.
Saw Palmetto (Serenoa repens)
Saw palmetto (Serona repens) is an extract from the berries of the saw palmetto palm tree (American dwarf tree) containing phytosterols (beta-sitosterol), fatty acids, beta-carotene, and polysaccharides. Saw palmetto is a competitive, nonselective inhibitor of both forms of 5-alpha-reductase and was advertised as a regimen for benign prostatic hyperplasia and androgenic alopecia 187, 188, 189. Saw palmetto blocks nuclear uptake of dihydrotestosterone (DHT) in target cells and decreases DHT (dihydrotestosterone) binding to androgen receptors by approximately 50%. Additionally, the extract increases 3α-hydroxysteroid-dehydrogenase activity, increasing the conversion of DHT to its weaker metabolite, androstanediol. As a result, the pharmacodynamic profile of saw palmetto differs from finasteride due to multiple sites of action 190, 191.
Twenty-six males with androgenetic alopecia treated with either 50 mg of oral beta-sitosterol and 200 mg saw palmetto or placebo daily resulted in 60% of patients with “improved” outcomes compared to 11% of controls. Gastrointestinal side effects, including loss of appetite, flatulence, and diarrhea, were experienced by 3 patients 192. Another study applying topical saw palmetto extract in lotion and shampoo for 3 months led to 35% increase in hair density 189. In another study, 100 men with androgenetic alopecia were treated with 320 mg of oral saw palmetto extract or 1 mg of oral finasteride daily for 2 years 70. Although hair growth scores were higher in the finasteride group with 68% experiencing growth at the front and vertex of scalp, 38% of patients in the saw palmetto cohort also had an increase in hair growth, mainly on the vertex 70. The investigators also noted that finasteride affected the vertex and frontal scalp, while saw palmetto primarily affected the vertex scalp 70.
In addition to systemic therapy, saw palmetto has also been studied as a topical agent. A study evaluating the hair growth effect of 3.3 mL topical saw palmetto serum applied for 4 weeks and 2 mL lotion for 24 weeks, in 50 men with androgenetic alopecia, demonstrated increased average and terminal hair counts at 12 and 24 weeks 193. Although systemic saw palmetto has not demonstrated superiority to conventional systemic therapies, it does have clinical benefits and is an attractive alternative treatment for male androgenetic alopecia patients who are not interested in oral finasteride 58.
Side effects of saw palmetto are minimal. The most common side effect is gastric discomfort. Saw palmetto may reduce PSA levels by 50% after 6–12 months of treatment, thus possibly missing early detection of prostate cancer in patients self‐medicating with Serenoa repens (saw palmetto) 189.
Zinc
Zinc is an essential trace element involved in enzyme catalysis, protein folding, and gene expression. Signs of deficiency include growth retardation, delayed puberty, diarrhea, alopecia, glossitis, nail dystrophy, and decreased immunity among others 194. Low zinc levels have been identified in patients with alopecia areata, androgenetic alopecia, and telogen effluvium 195. Proposed mechanisms for zinc-associated hair regrowth include antimicrobial, anti-inflammatory, antioxidant, and anti-5alpha-reductase activity 196, 197, 198, 199. Zinc has been studied as both a topical and oral supplement.
Zinc chelates with pyrithione to create a coordination complex that acts as an antifungal for treatment of seborrheic dermatitis 200. Comparing the efficacy of 1% pyrithione zinc shampoo used daily, 5% topical minoxidil solution used twice daily, or a combination of both, for 9 weeks in 200 androgenetic alopecia patients resulted in increased hair counts in all groups compared to placebo. However, the 1% pyrithione zinc group had only a modest improvement in hair growth, with hair counts less than half of the minoxidil group. No increase in hair count was noted between the combination therapy versus minoxidil alone 201.
Oral supplementation of zinc has been studied for androgenetic alopecia and alopecia areata. Supplementation of 50 mg of zinc sulfate daily, 100 mg of calcium pantothenate (the calcium salt of vitamin B5) daily, or a combination of the two twice weekly was compared to 2% topical minoxidil solution in 73 women with androgenetic alopecia, demonstrating positive outcomes in all groups. Although 2% minoxidil results in a greater increase in hair density, oral zinc and pantothenate supplementation creates thicker hair shafts 202.
Alopecia areata may present with concomitant zinc deficiency, and oral supplementation may be beneficial at a dose of 50 mg daily, with positive results seen in 67% of patients in one study and complete resolution seen in a child with diffuse alopecia 203, 204. In a double-blinded, cross-over study, researchers systematically evaluated oral zinc sulfate (5 mg/kg/day) for the treatment of alopecia areata in 100 patients 205. The first cohort was initially treated with zinc and then placebo, resulting in complete hair regrowth in 60% of patients after 3 months and maintenance for 3 months after cessation. Conversely, the second cohort was first treated with placebo and then zinc. After 3 months of placebo, 10% of patients completely regrew hair, and subsequent supplementation with zinc resulted in 67% of patients with complete regrowth. Side effects were mild, including gastric upset 205. Topical and oral zinc supplementation may prove to be an efficacious adjuvant for both androgenetic alopecia and alopecia areata treatment 58.
- Price VH. Treatment of hair loss. N Engl J Med. 1999 Sep 23;341(13):964-73. doi: 10.1056/NEJM199909233411307[↩]
- Elsaie, LT, Elshahid, AR, Hasan, HM, Soultan, FAZM, Jafferany, M, Elsaie, ML. Cross sectional quality of life assessment in patients with androgenetic alopecia. Dermatologic Therapy. 2020; 33:e13799. https://doi.org/10.1111/dth.13799[↩]
- Rajabi, F., Drake, L.A., Senna, M.M. and Rezaei, N. (2018), Alopecia areata: a review of disease pathogenesis. Br J Dermatol, 179: 1033-1048. https://doi.org/10.1111/bjd.16808[↩]
- Chen S, Xie X, Zhang G, Zhang Y. Comorbidities in Androgenetic Alopecia: A Comprehensive Review. Dermatol Ther (Heidelb). 2022 Oct;12(10):2233-2247. doi: 10.1007/s13555-022-00799-7[↩]
- Girijala RL, Riahi RR, Cohen PR. Platelet-rich plasma for androgenic alopecia treatment: A comprehensive review. Dermatol Online J. 2018 Jul 15;24(7):13030/qt8s43026c https://doi.org/10.5070/D3247040910[↩]
- Motofei IG, Rowland DL, Tampa M, Sarbu MI, Mitran MI, Mitran CI, et al. Finasteride and androgenic alopecia; from therapeutic options to medical implications. J Dermatolog Treat. 2020;31:415–421. doi: 10.1080/09546634.2019.1595507[↩]
- Cranwell W, Sinclair R. Male Androgenetic Alopecia. [Updated 2016 Feb 29]. In: Feingold KR, Anawalt B, Boyce A, et al., editors. Endotext [Internet]. South Dartmouth (MA): MDText.com, Inc.; 2000-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK278957[↩]
- Drake L, Reyes-Hadsall S, Martinez J, Heinrich C, Huang K, Mostaghimi A. Evaluation of the Safety and Effectiveness of Nutritional Supplements for Treating Hair Loss: A Systematic Review. JAMA Dermatol. Published online November 30, 2022. doi:10.1001/jamadermatol.2022.4867[↩][↩][↩][↩]
- Olsen EA, Hordinsky M, Whiting D, Stough D, Hobbs S, Ellis ML, Wilson T, Rittmaster RS; Dutasteride Alopecia Research Team. The importance of dual 5alpha-reductase inhibition in the treatment of male pattern hair loss: results of a randomized placebo-controlled study of dutasteride versus finasteride. J Am Acad Dermatol. 2006 Dec;55(6):1014-23. doi: 10.1016/j.jaad.2006.05.007[↩]
- Debruyne F, Barkin J, van Erps P, Reis M, Tammela TL, Roehrborn C; ARIA3001, ARIA3002 and ARIB3003 Study Investigators. Efficacy and safety of long-term treatment with the dual 5 alpha-reductase inhibitor dutasteride in men with symptomatic benign prostatic hyperplasia. Eur Urol. 2004 Oct;46(4):488-94; discussion 495. doi: 10.1016/j.eururo.2004.05.008[↩]
- Olszewska M, Rudnicka L. Effective treatment of female androgenic alopecia with dutasteride. J Drugs Dermatol. 2005 Sep-Oct;4(5):637-40.[↩]
- Saceda-Corralo D, Moustafa F, Moreno-Arrones Ó, Jaén-Olasolo P, Vañó-Galván S, Camacho F. Mesotherapy With Dutasteride for Androgenetic Alopecia: A Retrospective Study in Real Clinical Practice. J Drugs Dermatol. 2022 Jul 1;21(7):742-747. https://jddonline.com/articles/mesotherapy-with-dutasteride-for-androgenetic-alopecia-a-retrospective-study-in-real-clinical-practice-S1545961622P0742X[↩]
- Eun HC, Kwon OS, Yeon JH, Shin HS, Kim BY, Ro BI, Cho HK, Sim WY, Lew BL, Lee WS, Park HY, Hong SP, Ji JH. Efficacy, safety, and tolerability of dutasteride 0.5 mg once daily in male patients with male pattern hair loss: a randomized, double-blind, placebo-controlled, phase III study. J Am Acad Dermatol. 2010 Aug;63(2):252-8. doi: 10.1016/j.jaad.2009.09.018[↩]
- Gubelin Harcha W, Barboza Martínez J, Tsai TF, Katsuoka K, Kawashima M, Tsuboi R, Barnes A, Ferron-Brady G, Chetty D. A randomized, active- and placebo-controlled study of the efficacy and safety of different doses of dutasteride versus placebo and finasteride in the treatment of male subjects with androgenetic alopecia. J Am Acad Dermatol. 2014 Mar;70(3):489-498.e3. doi: 10.1016/j.jaad.2013.10.049[↩]
- Inui S, Itami S. Reversal of androgenetic alopecia by topical ketoconzole: relevance of anti-androgenic activity. J Dermatol Sci. 2007 Jan;45(1):66-8. doi: 10.1016/j.jdermsci.2006.08.011[↩]
- Hugo Perez BS. Ketocazole as an adjunct to finasteride in the treatment of androgenetic alopecia in men. Med Hypotheses. 2004;62(1):112-5. doi: 10.1016/s0306-9877(03)00264-0[↩]
- Jimenez-Cauhe J, Saceda-Corralo D, Rodrigues-Barata R, Hermosa-Gelbard A, Moreno-Arrones OM, Fernandez-Nieto D, Vaño-Galvan S. Effectiveness and safety of low-dose oral minoxidil in male androgenetic alopecia. J Am Acad Dermatol. 2019 Aug;81(2):648-649. doi: 10.1016/j.jaad.2019.04.054[↩]
- Leavitt M, Charles G, Heyman E, Michaels D. HairMax LaserComb laser phototherapy device in the treatment of male androgenetic alopecia: A randomized, double-blind, sham device-controlled, multicentre trial. Clin Drug Investig. 2009;29(5):283-92. doi: 10.2165/00044011-200929050-00001[↩][↩]
- Adil A, Godwin M. The effectiveness of treatments for androgenetic alopecia: A systematic review and meta-analysis. J Am Acad Dermatol. 2017 Jul;77(1):136-141.e5. doi: 10.1016/j.jaad.2017.02.054[↩][↩]
- Darwin E, Heyes A, Hirt PA, Wikramanayake TC, Jimenez JJ. Low-level laser therapy for the treatment of androgenic alopecia: a review. Lasers Med Sci. 2018 Feb;33(2):425-434. doi: 10.1007/s10103-017-2385-5[↩][↩][↩][↩][↩]
- RED light treatment for hair loss ‐ the science behind REVIAN RED. https://revian.com/how-it-works[↩]
- Kernel Networks Inc . Modulated light therapy in participants with pattern hair loss. Case Med Res. 2019. 10.31525/ct1-nct04019795[↩]
- Esmat, S.M., Hegazy, R.A., Gawdat, H.I., Abdel Hay, R.M., Allam, R.S., El Naggar, R. and Moneib, H. (2017), Low level light-minoxidil 5% combination versus either therapeutic modality alone in management of female patterned hair loss: A randomized controlled study. Lasers Surg. Med., 49: 835-843. https://doi.org/10.1002/lsm.22684[↩][↩]
- Frigo L, Luppi JS, Favero GM, Maria DA, Penna SC, Bjordal JM, Bensadoun RJ, Lopes-Martins RA. The effect of low-level laser irradiation (In-Ga-Al-AsP – 660 nm) on melanoma in vitro and in vivo. BMC Cancer. 2009 Nov 20;9:404. doi: 10.1186/1471-2407-9-404[↩][↩]
- Alves R, Grimalt R. Platelet-Rich Plasma and its Use for Cicatricial and Non-Cicatricial Alopecias: A Narrative Review. Dermatol Ther (Heidelb). 2020 Aug;10(4):623-633. doi: 10.1007/s13555-020-00408-5[↩][↩][↩]
- Nestor MS, Ablon G, Gade A, Han H, Fischer DL. Treatment options for androgenetic alopecia: Efficacy, side effects, compliance, financial considerations, and ethics. J Cosmet Dermatol. 2021 Dec;20(12):3759-3781. doi: 10.1111/jocd.14537[↩][↩][↩]
- Girijala RL, Riahi RR, Cohen PR. Platelet-rich plasma for androgenic alopecia treatment: A comprehensive review. Dermatol Online J. 2018 Jul 15;24(7):13030/qt8s43026c https://escholarship.org/uc/item/8s43026c[↩][↩]
- Hausauer AK, Jones DH. Evaluating the Efficacy of Different Platelet-Rich Plasma Regimens for Management of Androgenetic Alopecia: A Single-Center, Blinded, Randomized Clinical Trial. Dermatol Surg. 2018 Sep;44(9):1191-1200. doi: 10.1097/DSS.0000000000001567[↩][↩][↩][↩]
- Alves R, Grimalt R. Randomized Placebo-Controlled, Double-Blind, Half-Head Study to Assess the Efficacy of Platelet-Rich Plasma on the Treatment of Androgenetic Alopecia. Dermatol Surg. 2016 Apr;42(4):491-7. doi: 10.1097/DSS.0000000000000665[↩][↩]
- Hesseler MJ, Shyam N. Platelet-Rich Plasma and Its Utilities in Alopecia: A Systematic Review. Dermatol Surg. 2020 Jan;46(1):93-102. doi: 10.1097/DSS.0000000000001965[↩][↩]
- Tobin DJ. The aging hair pigmentary unit. In: Trüeb, RM , Tobin, DJ , eds.. Aging Hair. Heidelberg: Springer‐Verlag; 2010:77‐89. 10.1007/978-3-642-02636-2_9[↩]
- Sun, H.Y. and Sebaratnam, D.F. (2020), Clascoterone as a novel treatment for androgenetic alopecia. Clin Exp Dermatol, 45: 913-914. https://doi.org/10.1111/ced.14292[↩]
- van Zuuren, E.J., Fedorowicz, Z. and Carter, B. (2012), Evidence-based treatments for female pattern hair loss: a summary of a Cochrane systematic review. British Journal of Dermatology, 167: 995-1010. https://doi.org/10.1111/j.1365-2133.2012.11166.x[↩]
- Lanzafame RJ, Blanche RR, Chiacchierini RP, Kazmirek ER, Sklar JA. The growth of human scalp hair in females using visible red light laser and LED sources. Lasers Surg Med. 2014 Oct;46(8):601-7. doi: 10.1002/lsm.22277[↩][↩]
- Sperling LC, Mezebish DS. Hair diseases. Med Clin North Am. 1998 Sep;82(5):1155-69. doi: 10.1016/s0025-7125(05)70408-9[↩]
- Habif TP. Clinical dermatology: a color guide to diagnosis and therapy. 3d ed. St. Louis: Mosby, 1996.[↩]
- Hair Loss in Women. N Engl J Med 2007; 357:1620-1630. https://www.nejm.org/doi/full/10.1056/nejmcp072110[↩]
- Meephansan J, Thummakriengkrai J, Ponnikorn S, Yingmema W, Deenonpoe R, Suchonwanit P. Efficacy of topical tofacitinib in promoting hair growth in non-scarring alopecia: possible mechanism via VEGF induction. Arch Dermatol Res. 2017;309(9):729–738. doi:10.1007/s00403-017-1777-5[↩]
- Rojhirunsakool, S., & Suchonwanit, P. (2017). Parietal scalp is another affected area in female pattern hair loss: an analysis of hair density and hair diameter. Clinical, cosmetic and investigational dermatology, 11, 7–12. https://doi.org/10.2147/CCID.S153768[↩]
- Al Aboud AM, Zito PM. Alopecia. [Updated 2021 Aug 11]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2021 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK538178[↩]
- Shapiro J. Clinical practice. Hair loss in women. N Engl J Med. 2007;357(16):1620–1630. DOI: 10.1056/NEJMcp072110[↩]
- Price VH. Treatment of hair loss. N Engl J Med. 1999;341(13):964–973.[↩][↩]
- Banka N, Bunagan MJ, Shapiro J. Pattern hair loss in men: diagnosis and medical treatment. Dermatol Clin. 2013;31(1):129–140.[↩]
- Rittmaster RS. Finasteride. N Engl J Med. 1994;330(2):120–125.[↩]
- Pratt, C. H., King, L. E., Jr, Messenger, A. G., Christiano, A. M., & Sundberg, J. P. (2017). Alopecia areata. Nature reviews. Disease primers, 3, 17011. https://doi.org/10.1038/nrdp.2017.11[↩]
- Lee YB, Jun M, Lee WS. Alopecia areata and poliosis: A retrospective analysis of 258 cases. J Am Acad Dermatol. 2019 Jun;80(6):1776-1778. doi: 10.1016/j.jaad.2018.11.033[↩]
- Tosti A, Bellavista S, Iorizzo M. Alopecia areata: a long term follow-up study of 191 patients. J Am Acad Dermatol. 2006 Sep;55(3):438-41. doi: 10.1016/j.jaad.2006.05.008[↩]
- Asghar, F., Shamim, N., Farooque, U., Sheikh, H., & Aqeel, R. (2020). Telogen Effluvium: A Review of the Literature. Cureus, 12(5), e8320. https://doi.org/10.7759/cureus.8320[↩]
- Liu LY, King BA. Response to tofacitinib therapy of eyebrows and eyelashes in alopecia areata. J Am Acad Dermatol. 2019 Jun;80(6):1778-1779. doi: 10.1016/j.jaad.2018.11.037[↩]
- Chen P, Chen F, Zhou B. The risk of dermatological toxicities of combined BRAF and MEK inhibition versus BRAF inhibition alone in melanoma patients: a systematic review and meta-analysis. Cutan Ocul Toxicol. 2019 Jun;38(2):105-111. doi: 10.1080/15569527.2018.1553180[↩]
- Dei-Cas I, Carrizo D, Giri M, Boyne G, Domínguez N, Novello V, Acuña K, Dei-Cas P. Infectious skin disorders encountered in a pediatric emergency department of a tertiary care hospital in Argentina: a descriptive study. Int J Dermatol. 2019 Mar;58(3):288-295. doi: 10.1111/ijd.14234[↩]
- Souissi A, Ben Lagha I, Toukabri N, Mama M, Mokni M. Morse code-like hairs in tinea capitis disappear after successful treatment. Int J Dermatol. 2018 Dec;57(12):e150-e151. doi: 10.1111/ijd.14224[↩]
- Al Aboud AM, Crane JS. Tinea Capitis. [Updated 2021 Aug 11]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2021 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK536909[↩]
- McGarvey EL, Baum LD, Pinkerton RC, Rogers LM. Psychological sequelae and alopecia among women with cancer. Cancer Pract. 2001;9(6):283–289.[↩]
- Trüeb RM. Chemotherapy-induced alopecia. Semin Cutan Med Surg. 2009;28(1):11–14.[↩]
- Kanwar AJ, Narang T. Anagen effluvium. Indian J Dermatol Venereol Leprol. 2013;79(5):604–612.[↩]
- Thiedke CC. Alopecia in women. Am Fam Physician. 2003;67(5):1007–1014.[↩][↩]
- Hosking AM, Juhasz M, Atanaskova Mesinkovska N. Complementary and Alternative Treatments for Alopecia: A Comprehensive Review. Skin Appendage Disord. 2019 Feb;5(2):72-89. doi: 10.1159/000492035[↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Ring C, Heitmiller K, Correia E, Gabriel Z, Saedi N. Nutraceuticals for Androgenetic Alopecia. J Clin Aesthet Dermatol. 2022 Mar;15(3):26-29. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8944288[↩][↩][↩][↩][↩][↩][↩]
- Trueb RM. Berlin Heidelberg: Springer- Verlag; 2008. Diffuse hair loss. In: Blume-Peytavi UTA, Whiting DA, Treub R, eds. Hair Growth and Disorders.[↩]
- Lengg N, Heidecker B, Seifert B, Trueb RM. Dietary supplement increases anagen hair rate in women with telogen effluvium: results of a double-blind, placebo-controlled trial. Therapy. 2007;4:59–65.[↩]
- Le Floc’h, C., Cheniti, A., Connétable, S., Piccardi, N., Vincenzi, C. and Tosti, A. (2015), Effect of a nutritional supplement on hair loss in women. J Cosmet Dermatol, 14: 76-82. https://doi.org/10.1111/jocd.12127[↩]
- Ablon G, Kogan S. A Six-Month, Randomized, Double-Blind, Placebo-Controlled Study Evaluating the Safety and Efficacy of a Nutraceutical Supplement for Promoting Hair Growth in Women With Self-Perceived Thinning Hair. J Drugs Dermatol. 2018 May 1;17(5):558-565. https://jddonline.com/articles/a-six-month-randomized-double-blind-placebo-controlled-study-evaluating-the-safety-and-efficacy-of-a-S1545961618P0558X/[↩]
- Farris PK, Rogers N, McMichael A, Kogan S. A Novel Multi-Targeting Approach to Treating Hair Loss, Using Standardized Nutraceuticals. J Drugs Dermatol. 2017 Nov 1;16(11):s141-s148. https://jddonline.com/articles/a-novel-multi-targeting-approach-to-treating-hair-loss-using-standardized-nutraceuticals-S1545961617S0141X[↩][↩][↩][↩][↩][↩][↩]
- Nakamura K, Yasunaga Y, Segawa T, Ko D, Moul JW, Srivastava S, Rhim JS. Curcumin down-regulates AR gene expression and activation in prostate cancer cell lines. Int J Oncol. 2002 Oct;21(4):825-30. https://doi.org/10.3892/ijo.21.4.825[↩]
- Shishodia, S. (2013), Molecular mechanisms of curcumin action: Gene expression. BioFactors, 39: 37-55. https://doi.org/10.1002/biof.1041[↩]
- Shoba G, Joy D, Joseph T, Majeed M, Rajendran R, Srinivas PS. Influence of piperine on the pharmacokinetics of curcumin in animals and human volunteers. Planta Med. 1998 May;64(4):353-6. doi: 10.1055/s-2006-957450[↩]
- Auddy B, Hazra J, Mitra A et al. A Standardized Withania Somnifera Extract Significantly Reduces Stress-Related Parameters in Chronically Stressed Humans: A Double-Blind, Randomized, Placebo-Controlled Study. Journal of American Nutraceutical Association. 2008;11:50–56.[↩][↩]
- Thom E. Stress and the Hair Growth Cycle: Cortisol-Induced Hair Growth Disruption. J Drugs Dermatol. 2016 Aug 1;15(8):1001-4. https://jddonline.com/articles/stress-and-the-hair-growth-cycle-cortisol-induced-hair-growth-disruption-S1545961616P1001X[↩]
- Rossi A, Mari E, Scarno M, Garelli V, Maxia C, Scali E, Iorio A, Carlesimo M. Comparitive effectiveness of finasteride vs Serenoa repens in male androgenetic alopecia: a two-year study. Int J Immunopathol Pharmacol. 2012 Oct-Dec;25(4):1167-73. doi: 10.1177/039463201202500435[↩][↩][↩][↩]
- Wang Y, Park NY, Jang Y, Ma A, Jiang Q. Vitamin E γ-Tocotrienol Inhibits Cytokine-Stimulated NF-κB Activation by Induction of Anti-Inflammatory A20 via Stress Adaptive Response Due to Modulation of Sphingolipids. J Immunol. 2015 Jul 1;195(1):126-33. doi: 10.4049/jimmunol.1403149[↩][↩]
- Beoy LA, Woei WJ, Hay YK. Effects of tocotrienol supplementation on hair growth in human volunteers. Trop Life Sci Res. 2010 Dec;21(2):91-9. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3819075[↩][↩]
- Ablon G, Kogan S. A Six-Month, Randomized, Double-Blind, Placebo-Controlled Study Evaluating the Safety and Efficacy of a Nutraceutical Supplement for Promoting Hair Growth in Women With Self-Perceived Thinning Hair. J Drugs Dermatol. 2018 May 1;17(5):558-565. https://jddonline.com/articles/a-six-month-randomized-double-blind-placebo-controlled-study-evaluating-the-safety-and-efficacy-of-a-S1545961618P0558X[↩][↩][↩]
- Hornfeldt, CS. Growing evidence of the beneficial effects of a marine protein-based dietary supplement for treating hair loss. J Cosmet Dermatol. 2018; 17: 209– 213. https://doi.org/10.1111/jocd.12400 [↩]
- Ablon G. A 3-month, randomized, double-blind, placebo-controlled study evaluating the ability of an extra-strength marine protein supplement to promote hair growth and decrease shedding in women with self-perceived thinning hair. Dermatol Res Pract. 2015;2015:841570. doi: 10.1155/2015/841570[↩][↩][↩][↩][↩][↩]
- Costa A, Pereira ESP, Favaro R et al. Treating cutaneous photoaging in women with an oral supplement based on marine protein, concentrated acerola, grape seed extract and tomato extract, for 360 days. Surg Cosmet Dermatol. 2011;3:302–311.[↩]
- Lassus A, Jeskanen L, Happonen HP, Santalahti J. Imedeen for the treatment of degenerated skin in females. J Int Med Res. 1991 Mar-Apr;19(2):147-52. doi: 10.1177/030006059101900208[↩]
- Rizer RL, Stephens TJ, Herndon JH, Sperber BR, Murphy J, Ablon GR. A Marine Protein-based Dietary Supplement for Subclinical Hair Thinning/Loss: Results of a Multisite, Double-blind, Placebo-controlled Clinical Trial. Int J Trichology. 2015 Oct-Dec;7(4):156-66. doi: 10.4103/0974-7753.171573[↩][↩][↩][↩][↩]
- Glynis A. A Double-blind, Placebo-controlled Study Evaluating the Efficacy of an Oral Supplement in Women with Self-perceived Thinning Hair. J Clin Aesthet Dermatol. 2012 Nov;5(11):28-34. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3509882[↩][↩][↩][↩]
- Ablon, G. (2016), A 6-month, randomized, double-blind, placebo-controlled study evaluating the ability of a marine complex supplement to promote hair growth in men with thinning hair. J Cosmet Dermatol, 15: 358-366. https://doi.org/10.1111/jocd.12265[↩][↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Ablon G, Dayan S. A Randomized, Double-blind, Placebo-controlled, Multi-center, Extension Trial Evaluating the Efficacy of a New Oral Supplement in Women with Self-perceived Thinning Hair. J Clin Aesthet Dermatol. 2015 Dec;8(12):15-21. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4689507[↩][↩][↩][↩][↩][↩]
- Hornfeldt, CS. Growing evidence of the beneficial effects of a marine protein-based dietary supplement for treating hair loss. J Cosmet Dermatol. 2018; 17: 209– 213. https://doi.org/10.1111/jocd.12400[↩]
- FDA In Brief: FDA provides draft recommendations to diagnostic manufacturers to help reduce risks of biotin interference with certain lab tests. https://www.fda.gov/news-events/fda-brief/fda-brief-fda-provides-draft-recommendations-diagnostic-manufacturers-help-reduce-risks-biotin[↩][↩]
- FDA in Brief: FDA reminds patients, health care professionals and laboratory personnel about the potential for biotin interference with certain test results, especially specific tests to aid in heart attack diagnoses. https://www.fda.gov/news-events/fda-brief/fda-brief-fda-reminds-patients-health-care-professionals-and-laboratory-personnel-about-potential[↩][↩]
- Thom E. Nourkrin: objective and subjective effects and tolerability in persons with hair loss. J Int Med Res. 2006 Sep-Oct;34(5):514-9. doi: 10.1177/147323000603400508[↩][↩][↩][↩][↩]
- Biotin. https://ods.od.nih.gov/factsheets/Biotin-HealthProfessional[↩][↩]
- Zempleni J, Hassan YI, Wijeratne SS. Biotin and biotinidase deficiency. Expert Rev Endocrinol Metab. 2008 Nov 1;3(6):715-724. doi: 10.1586/17446651.3.6.715[↩]
- Zempleni J, Wijeratne SSK, Kuroishi T. Biotin. In: Erdman JW, Macdonald IA, Zeisel SH, eds. Present Knowledge in Nutrition. 10th ed. Washington, DC: Wiley-Blackwell; 2012:359-74.[↩][↩][↩]
- Mock DM. Biotin. In: Ross AC, Caballero B, Cousins RJ, Tucker KL, Ziegler TR, eds. Modern Nutrition in Health and Disease. 11th ed. Baltimore, MD: Lippincott Williams & Wilkins; 2014:390-8.[↩][↩][↩]
- Combs GF, Jr. Biotin. In: Combs GF, Jr., ed. The vitamins: fundamental aspects in nutrition and health. Third ed. Burlington, MA: Elsevier Academic Press; 2008:331-44.[↩][↩]
- Perry CA, West AA, Gayle A, Lucas LK, Yan J, Jiang X, Malysheva O, Caudill MA. Pregnancy and lactation alter biomarkers of biotin metabolism in women consuming a controlled diet. J Nutr. 2014 Dec;144(12):1977-84. doi: 10.3945/jn.114.194472[↩]
- Mock DM. Biotin. In: Coates PM, Betz JM, Blackman MR, et al., eds. Encyclopedia of Dietary Supplements. 2nd ed. London and New York: Informa Healthcare; 2010:43-51.[↩][↩][↩]
- Mock DM. Biotin. In: Ross AC, Caballero B, Cousins RJ, Tucker KL, Ziegler TR, eds. Modern Nutrition in Health and Disease. 11th ed.: Lippincott Williams & Wilkins; 2014:390-398.[↩]
- Wolf B. Biotinidase Deficiency. 2000 Mar 24 [Updated 2016 Jun 9]. In: Adam MP, Everman DB, Mirzaa GM, et al., editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993-2022. Available from: https://www.ncbi.nlm.nih.gov/books/NBK1322[↩][↩]
- Food and Nutrition Board, Institute of Medicine. Biotin. Dietary Reference Intakes: Thiamin, Riboflavin, Niacin, Vitamin B6, Vitamin B12, Pantothenic Acid, Biotin, and Choline. Washington, D.C.: National Academy Press; 1998:374-389.[↩]
- Elrefai S, Wolf B. Disorders of biotin metabolism. In: Rosenberg RN, Pascual JM, eds. Rosenberg’s Molecular and Genetic basis of Neurological and Psychiatric Disease. 5th ed. United States of America: Elsevier; 2015:531-539.[↩]
- Regula Baumgartner, E. and Suormala, T. (1999), Inherited defects of biotin metabolism. BioFactors, 10: 287-290. https://doi.org/10.1002/biof.5520100229[↩]
- Shelley WB, Shelley ED. Uncombable hair syndrome: observations on response to biotin and occurrence in siblings with ectodermal dysplasia. J Am Acad Dermatol. 1985 Jul;13(1):97-102. doi: 10.1016/s0190-9622(85)70150-8[↩]
- Boccaletti, V., Zendri, E., Giordano, G., Gnetti, L. and De Panfilis, G. (2007), Familial Uncombable Hair Syndrome: Ultrastructural Hair Study and Response to Biotin. Pediatric Dermatology, 24: E14-E16. https://doi.org/10.1111/j.1525-1470.2007.00385.x[↩]
- Mock DM, Baswell DL, Baker H, Holman RT, Sweetman L. Biotin deficiency complicating parenteral alimentation: diagnosis, metabolic repercussions, and treatment. J Pediatr. 1985 May;106(5):762-9. doi: 10.1016/s0022-3476(85)80350-4[↩]
- Fujimoto, W., Inaoki, M., Fukui, T., Inoue, Y. and Kuhara, T. (2005), Biotin Deficiency in an Infant Fed with Amino Acid Formula. The Journal of Dermatology, 32: 256-261. https://doi.org/10.1111/j.1346-8138.2005.tb00758.x[↩]
- Trüeb RM. Serum Biotin Levels in Women Complaining of Hair Loss. Int J Trichology. 2016 Apr-Jun;8(2):73-7. doi: 10.4103/0974-7753.188040[↩]
- Soleymani T, Lo Sicco K, Shapiro J. The Infatuation With Biotin Supplementation: Is There Truth Behind Its Rising Popularity? A Comparative Analysis of Clinical Efficacy versus Social Popularity. J Drugs Dermatol. 2017 May 1;16(5):496-500. https://jddonline.com/articles/the-infatuation-with-biotin-supplementation-is-there-truth-behind-its-rising-popularity-a-comparativ-S1545961617P0496X[↩]
- Schulpis KH, Georgala S, Papakonstantinou ED, Michas T, Karikas GA. The effect of isotretinoin on biotinidase activity. Skin Pharmacol Appl Skin Physiol. 1999 Jan-Apr;12(1-2):28-33. doi: 10.1159/000029843[↩]
- Schulpis, K.H., Karikas, G.A., Tjamouranis, J., Regoutas, S. and Tsakiris, S. (2001), Low Serum Biotinidase Activity in Children with Valproic Acid Monotherapy. Epilepsia, 42: 1359-1362. https://doi.org/10.1046/j.1528-1157.2001.47000.x[↩]
- Patel DP, Swink SM, Castelo-Soccio L. A Review of the Use of Biotin for Hair Loss. Skin Appendage Disord. 2017 Aug;3(3):166-169. doi: 10.1159/000462981[↩]
- Thyroid News and Research Articles. January 2016: Thyroid Month: Beware of Biotin. https://endocrinenews.endocrine.org/january-2016-thyroid-month-beware-of-biotin[↩]
- Kummer S, Hermsen D, Distelmaier F. Biotin Treatment Mimicking Graves’ Disease. N Engl J Med. 2016 Aug 18;375(7):704-6. doi: 10.1056/NEJMc1602096[↩]
- Li D, Radulescu A, Shrestha RT, Root M, Karger AB, Killeen AA, Hodges JS, Fan SL, Ferguson A, Garg U, Sokoll LJ, Burmeister LA. Association of Biotin Ingestion With Performance of Hormone and Nonhormone Assays in Healthy Adults. JAMA. 2017 Sep 26;318(12):1150-1160. doi: 10.1001/jama.2017.13705[↩]
- Vitamin D. https://ods.od.nih.gov/factsheets/VitaminD-HealthProfessional[↩]
- Roseland JM, Phillips KM, Patterson KY, Pehrsson PR, Taylor CL. Vitamin D in foods: An evolution of knowledge. Pages 41-78 in Feldman D, Pike JW, Bouillon R, Giovannucci E, Goltzman D, Hewison M, eds. Vitamin D, Volume 2: Health, Disease and Therapeutics, Fourth Edition. Elsevier, 2018.[↩]
- Institute of Medicine, Food and Nutrition Board. Dietary Reference Intakes for Calcium and Vitamin D. Washington, DC: National Academy Press, 2010.[↩]
- Kriegel MA, Manson JE, Costenbader KH. Does vitamin D affect risk of developing autoimmune disease?: a systematic review. Semin Arthritis Rheum. 2011 Jun;40(6):512-531.e8. doi: 10.1016/j.semarthrit.2010.07.009[↩]
- Xie Z, Komuves L, Yu QC, Elalieh H, Ng DC, Leary C, Chang S, Crumrine D, Yoshizawa T, Kato S, Bikle DD. Lack of the vitamin D receptor is associated with reduced epidermal differentiation and hair follicle growth. J Invest Dermatol. 2002 Jan;118(1):11-6. doi: 10.1046/j.1523-1747.2002.01644.x[↩]
- Tsai TY, Huang YC. Vitamin D deficiency in patients with alopecia areata: A systematic review and meta-analysis. J Am Acad Dermatol. 2018 Jan;78(1):207-209. doi: 10.1016/j.jaad.2017.07.051[↩]
- Miller R, Conic RZ, Bergfeld W, Mesinkovska NA. Prevalence of Comorbid Conditions and Sun-Induced Skin Cancers in Patients with Alopecia Areata. J Investig Dermatol Symp Proc. 2015 Nov;17(2):61-2. doi: 10.1038/jidsymp.2015.44[↩]
- Berth-Jones J, Hutchinson PE. Alopecia totalis does not respond to the vitamin-D analogue calcipotriol. J Dermatol Treat. 2009;1:293–294.[↩]
- Orecchia G, Rocchetti GA. Topical use of calcipotriol does not potentiate squaric acid dibutylester effectiveness in the treatment of alopecia areata. J Dermatol Treat. 1995;6:21–23.[↩]
- Kim DH, Lee JW, Kim IS, Choi SY, Lim YY, Kim HM, Kim BJ, Kim MN. Successful treatment of alopecia areata with topical calcipotriol. Ann Dermatol. 2012 Aug;24(3):341-4. doi: 10.5021/ad.2012.24.3.341[↩]
- Çerman AA, Solak SS, Altunay İ, Küçükünal NA. Topical Calcipotriol Therapy for Mild-to-Moderate Alopecia Areata: A Retrospective Study. J Drugs Dermatol. 2015 Jun;14(6):616-20. https://jddonline.com/articles/topical-calcipotriol-therapy-for-mild-to-moderate-alopecia-areata-a-retrospective-study-S1545961615P0616X[↩]
- Narang, T, Daroach, M, Kumaran, MS. Efficacy and safety of topical calcipotriol in management of alopecia areata: A pilot study. Dermatologic Therapy. 2017; 30:e12464. https://doi.org/10.1111/dth.12464[↩]
- Serbinova E, Kagan V, Han D, Packer L. Free radical recycling and intramembrane mobility in the antioxidant properties of alpha-tocopherol and alpha-tocotrienol. Free Radic Biol Med. 1991;10(5):263-75. doi: 10.1016/0891-5849(91)90033-y[↩]
- Faghri S, Tamura D, Kraemer KH, Digiovanna JJ. Trichothiodystrophy: a systematic review of 112 published cases characterises a wide spectrum of clinical manifestations. J Med Genet. 2008 Oct;45(10):609-21. doi: 10.1136/jmg.2008.058743[↩]
- Sacharow SJ, Picker JD, Levy HL. Homocystinuria Caused by Cystathionine Beta-Synthase Deficiency. 2004 Jan 15 [Updated 2017 May 18]. In: Adam MP, Everman DB, Mirzaa GM, et al., editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993-2022. Available from: https://www.ncbi.nlm.nih.gov/books/NBK1524[↩]
- Camfield DA. Cham: Springer; 2017. Nutritional-based nutraceuticals in the treatment of anxiety; in: Evidence-Based Herbal and Nutritional Treatments for Anxiety in Psychiatric Disorders; pp. pp 81–101.[↩]
- Hertel H, Gollnick H, Matthies C, Baumann I, Orfanos CE. Niedrig dosierte Retinol- und L-Cystin-Kombination bessern die Alopezie vom diffusen Typ nach peroraler Langzeitapplikation [Low dosage retinol and L-cystine combination improve alopecia of the diffuse type following long-term oral administration]. Hautarzt. 1989 Aug;40(8):490-5. German.[↩]
- Morganti P, Fabrizi P, James B, Bruno C. Effect of gelatin-cystine and Serenoa repens extract on free radicals level and hair growth. J Appl Cosmetol. 1998;16:57–64.[↩]
- Gehring W, Gloor M. Use of the phototrichogram to assess the stimulation of hair growth – an in vitro study of women with androgenetic alopecia. Z Hautkr. 2000;75:419–423.[↩]
- Lengg N, Heidecker B, Seifert B, Trüeb Dietary supplement increases anagen hair rate in women with telogen effluvium: results of a double-blind, placebo-controlled trial. Therapy. 2007;4:59–65.[↩]
- Rushton, D.H., Norris, M.J., Dover, R. and Busuttil, N. (2002), Causes of hair loss and the developments in hair rejuvenation. International Journal of Cosmetic Science, 24: 17-23. https://doi.org/10.1046/j.0412-5463.2001.00110.x[↩]
- Wood JM, Decker H, Hartmann H, Chavan B, Rokos H, Spencer JD, et al. Senile hair graying: H2O2-mediated oxidative stress affects human hair color by blunting methionine sulfoxide repair. FASEB J. 2009;23:2065–2075. https://faseb.onlinelibrary.wiley.com/doi/epdf/10.1096/fj.08-125435[↩]
- Oshimura E, Ino M. Effects of arginine on hair damage via oxidative coloring process. Journal of Cosmetic Science. 2004 ;55 Suppl:S155-70.[↩]
- Fischer, T.W., Hipler, U.C. and Elsner, P. (2007), Effect of caffeine and testosterone on the proliferation of human hair follicles in vitro. International Journal of Dermatology, 46: 27-35. https://doi.org/10.1111/j.1365-4632.2007.03119.x[↩]
- Fischer, T.W., Herczeg-Lisztes, E., Funk, W., Zillikens, D., Bíró, T. and Paus, R. (2014), Differential effects of caffeine on hair shaft elongation, matrix and outer root sheath keratinocyte proliferation, and transforming growth factor-β2/insulin-like growth factor-1-mediated regulation of the hair cycle in male and female human hair follicles in vitro. Br J Dermatol, 171: 1031-1043. https://doi.org/10.1111/bjd.13114[↩]
- Sisto T, Bussoletti C, Celleno L. Efficacy of a cosmetic caffeine shampoo in androgenetic alopecia management. J Appl Cosmetol. 2012;31:57–66.[↩]
- Bussoletti C, Mastropietro F, Tolani M. Use of a caffeine shampoo for the treatment of male androgenetic alopecia. J Appl Cosmetol. 2011;29:167–180.[↩]
- Bussoletti C, Tolaini MV, Celleno L. Efficacy of a cosmetic phyto-caffeine shampoo in female androgenetic alopecia. G Ital Dermatol Venereol. 2018 DOI: 10.23736/S0392-0488.18.05499-8[↩]
- Bussoletti C, Mastropietro F, Tolaini M, Celleno L. Use of a cosmetic caffeine lotion in the treatment of male androgenetic alopecia. J Appl Cosmetol. 2011;29:167–180.[↩]
- Dhurat R, Chitallia J, May TW, Jayaraaman AM, Madhukara J, Anandan S, Vaidya P, Klenk A. An Open-Label Randomized Multicenter Study Assessing the Noninferiority of a Caffeine-Based Topical Liquid 0.2% versus Minoxidil 5% Solution in Male Androgenetic Alopecia. Skin Pharmacol Physiol. 2017;30(6):298-305. doi: 10.1159/000481141[↩]
- Golpour M, Rabbani H, Farzin D, Azizi F. Comparing the effectiveness of local solution of minoxidil and caffeine 2.5% with local solution of minoxidil 2.5% in treatment of androgenetic alopecia. J Mazandaran Univ Med Sci. 2013;23:30–36.[↩]
- Pazoki-Toroudi H. The efficacy and safety of minoxidil 5% combination with azelaic acid 1/5% and caffeine 1% solution on male pattern hair loss (abstract) J Invest Dermatol. 2013;133:S84.[↩]
- Dressler, C., Blumeyer, A., Rosumeck, S., Arayesh, A. and Nast, A. (2017), Efficacy of topical caffeine in male androgenetic alopecia. JDDG: Journal der Deutschen Dermatologischen Gesellschaft, 15: 734-741. https://doi.org/10.1111/ddg.13271[↩]
- Harada N, Okajima K, Arai M, Kurihara H, Nakagata N. Administration of capsaicin and isoflavone promotes hair growth by increasing insulin-like growth factor-I production in mice and in humans with alopecia. Growth Horm IGF Res. 2007 Oct;17(5):408-15. doi: 10.1016/j.ghir.2007.04.009[↩][↩]
- Hordinsky M, Ericson M. Autoimmunity: alopecia areata. J Investig Dermatol Symp Proc. 2004 Jan;9(1):73-8. doi: 10.1111/j.1087-0024.2004.00835.x[↩]
- Ehsani, A., Toosi, S., Seirafi, H., Akhyani, M., Hosseini, M., Azadi, R., Noormohamadpour, P. and Ghanadan, A. (2009), Capsaicin vs. clobetasol for the treatment of localized alopecia areata. Journal of the European Academy of Dermatology and Venereology, 23: 1451-1453. https://doi.org/10.1111/j.1468-3083.2009.03243.x[↩]
- Harada N, Okajima K, Narimatsu N, Kurihara H, Nakagata N. Effect of topical application of raspberry ketone on dermal production of insulin-like growth factor-I in mice and on hair growth and skin elasticity in humans. Growth Horm IGF Res. 2008 Aug;18(4):335-44. doi: 10.1016/j.ghir.2008.01.005[↩]
- Fadus MC, Lau C, Bikhchandani J, Lynch HT. Curcumin: An age-old anti-inflammatory and anti-neoplastic agent. J Tradit Complement Med. 2016 Sep 9;7(3):339-346. doi: 10.1016/j.jtcme.2016.08.002[↩]
- Pumthong G, Asawanonda P, Varothai S, Jariyasethavong V, Triwongwaranat D, Suthipinittharm P, Ingkaninan K, Leelapornpisit P, Waranuch N. Curcuma aeruginosa, a novel botanically derived 5α-reductase inhibitor in the treatment of male-pattern baldness: a multicenter, randomized, double-blind, placebo-controlled study. J Dermatolog Treat. 2012 Oct;23(5):385-92. doi: 10.3109/09546634.2011.568470[↩]
- Srivilai J, Waranuch N, Tangsumranjit A, Khorana N, Ingkaninan K. Germacrone and sesquiterpene-enriched extracts from Curcuma aeruginosa Roxb. increase skin penetration of minoxidil, a hair growth promoter. Drug Deliv Transl Res. 2018 Feb;8(1):140-149. doi: 10.1007/s13346-017-0447-7[↩]
- Arreola R, Quintero-Fabián S, López-Roa RI, Flores-Gutiérrez EO, Reyes-Grajeda JP, Carrera-Quintanar L, Ortuño-Sahagún D. Immunomodulation and anti-inflammatory effects of garlic compounds. J Immunol Res. 2015;2015:401630. doi: 10.1155/2015/401630[↩]
- Hajheydari Z, Jamshidi M, Akbari J, Mohammadpour R. Combination of topical garlic gel and betamethasone valerate cream in the treatment of localized alopecia areata: a double-blind randomized controlled study. Indian J Dermatol Venereol Leprol. 2007 Jan-Feb;73(1):29-32. doi: 10.4103/0378-6323.30648[↩]
- Lassus A, Eskelinen E. A comparative study of a new food supplement, ViviScal, with fish extract for the treatment of hereditary androgenic alopecia in young males. J Int Med Res. 1992 Nov;20(6):445-53. doi: 10.1177/030006059202000601. Erratum in: J Int Med Res 1993 Jan-Feb;21(1):following 65.[↩][↩][↩]
- Efficacy of the New Viviscal Professional Strength Oral Supplement in Females With Thinning Hair. https://clinicaltrials.gov/ct2/show/NCT02302053[↩]
- Hornfeldt CS, Holland M, Bucay VW, Roberts WE, Waldorf HA, Dayan SH. The Safety and Efficacy of a Sustainable Marine Extract for the Treatment of Thinning Hair: A Summary of New Clinical Research and Results from a Panel Discussion on the Problem of Thinning Hair and Current Treatments. J Drugs Dermatol. 2015 Sep;14(9):s15-22. https://jddonline.com/articles/the-safety-and-efficacy-of-a-sustainable-marine-extract-for-the-treatment-of-thinning-hair-a-summary-S1545961615S0015X/[↩]
- Lassus A, Santalahti J, Sellmann M. Treatment of hereditary androgenic alopecia in middle aged males by combined oral and topical administration of special marine extract-compound (Viviscal) for 8 months. Nouv Dermatol Anglo Fr Int Dermatol. 1994;13:254–255.[↩]
- Majass M, Puuste O, Prästbacka B, Brorsdotter-Johansson P. Treatment of alopecia areata, alopecia totalis and alopecia universalis with oral Viviscal for 12 months. Swedish Association for Alopecia. 1996 http://www.llogo.hu/viviscal/study4.html[↩]
- Pereira J. Treatment of androgenetic alopecia with a marine-based extract of proteins and polysaccharides. Rev Bras Med. 1997;54:144–149.[↩]
- Jackson B. A 4-month study evaluating the efficacy and tolerability of an oral supplement for the treatment of thinning hair in African American women. Viviscal Prof Clin Trials Conduct Res. 2011[↩]
- Thom E. Efficacy and tolerability of Hairgain in individuals with hair loss: a placebo-controlled, double-blind study. J Int Med Res. 2001 Jan-Feb;29(1):2-6. doi: 10.1177/147323000102900101[↩]
- Bloch L. Demonstrating the efficacy of a nutraceutical for promoting hair growth using a digital photography technique with posterior image analysis. 2015 World Hair Congress, Miami.[↩]
- Fischer, T.W., Slominski, A., Tobin, D.J. and Paus, R. (2008), Melatonin and the hair follicle. Journal of Pineal Research, 44: 1-15. https://doi.org/10.1111/j.1600-079X.2007.00512.x[↩]
- Fischer, T.W., Burmeister, G., Schmidt, H.W. and Elsner, P. (2004), Melatonin increases anagen hair rate in women with androgenetic alopecia or diffuse alopecia: results of a pilot randomized controlled trial. British Journal of Dermatology, 150: 341-345. https://doi.org/10.1111/j.1365-2133.2004.05685.x[↩][↩]
- Fischer TW, Trüeb RM, Hänggi G, Innocenti M, Elsner P. Topical melatonin for treatment of androgenetic alopecia. Int J Trichology. 2012 Oct;4(4):236-45. doi: 10.4103/0974-7753.111199[↩][↩]
- Sharquie, K.E. and Al-Obaidi, H.K. (2002), Onion Juice (Allium cepa L.), A New Topical Treatment for Alopecia Areata. The Journal of Dermatology, 29: 343-346. https://doi.org/10.1111/j.1346-8138.2002.tb00277.x[↩][↩]
- Kamimura A, Takahashi T, Watanabe Y. Investigation of topical application of procyanidin B-2 from apple to identify its potential use as a hair growing agent. Phytomedicine. 2000 Dec;7(6):529-36. doi: 10.1016/S0944-7113(00)80040-9[↩][↩]
- Takahashi T, Kamiya T, Hasegawa A, Yokoo Y. Procyanidin oligomers selectively and intensively promote proliferation of mouse hair epithelial cells in vitro and activate hair follicle growth in vivo. J Invest Dermatol. 1999 Mar;112(3):310-6. doi: 10.1046/j.1523-1747.1999.00532.x[↩]
- Takahashi, T., Kamimura, A., Yokoo, Y., Honda, S. and Watanabe, Y. (2001), The first clinical trial of topical application of procyanidin B-2 to investigate its potential as a hair growing agent. Phytother. Res., 15: 331-336. https://doi.org/10.1002/ptr.800[↩]
- Takahashi, T., Kamimura, A., Kagoura, M., Toyoda, M. and Morohashi, M. (2005), Investigation of the topical application of procyanidin oligomers from apples to identify their potential use as a hair-growing agent. Journal of Cosmetic Dermatology, 4: 245-249. https://doi.org/10.1111/j.1473-2165.2005.00199.x[↩]
- Tenore GC, Caruso D, Buonomo G, D’Avino M, Santamaria R, Irace C, Piccolo M, Maisto M, Novellino E. Annurca Apple Nutraceutical Formulation Enhances Keratin Expression in a Human Model of Skin and Promotes Hair Growth and Tropism in a Randomized Clinical Trial. J Med Food. 2018 Jan;21(1):90-103. doi: 10.1089/jmf.2017.0016[↩][↩]
- Kamimura A, Takahashi T, Morohashi M, Takano Y. Procyanidin oligomers counteract TGF-beta1- and TGF-beta2-induced apoptosis in hair epithelial cells: an insight into their mechanisms. Skin Pharmacol Physiol. 2006;19(5):259-65. doi: 10.1159/000093981[↩]
- Kamimura, A. and Takahashi, T. (2002), Procyanidin B-3, isolated from barley and identified as a hair-growth stimulant, has the potential to counteract inhibitory regulation by TGF-β1. Experimental Dermatology, 11: 532-541. https://doi.org/10.1034/j.1600-0625.2002.110606.x[↩]
- Carbin BE, Larsson B, Lindahl O. Treatment of benign prostatic hyperplasia with phytosterols. Br J Urol. 1990 Dec;66(6):639-41. doi: 10.1111/j.1464-410x.1990.tb07199.x[↩]
- Cho YH, Lee SY, Jeong DW, Choi EJ, Kim YJ, Lee JG, Yi YH, Cha HS. Effect of pumpkin seed oil on hair growth in men with androgenetic alopecia: a randomized, double-blind, placebo-controlled trial. Evid Based Complement Alternat Med. 2014;2014:549721. doi: 10.1155/2014/549721[↩][↩][↩]
- Ribeiro-Santos R, Carvalho-Costa D, Cavaleiro C, Costa HS, Albuquerque TG, Castilho MC, et al. A novel insight on an ancient aromatic plant: the rosemary (Rosmarinus officinalis L.) Trends Food Sci Technol. 2015;45:355–368.[↩]
- Ezekwe N, King M, Hollinger JC. The Use of Natural Ingredients in the Treatment of Alopecias with an Emphasis on Central Centrifugal Cicatricial Alopecia: A Systematic Review. J Clin Aesthet Dermatol. 2020 Aug;13(8):23-27. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7595365[↩]
- Panahi Y, Taghizadeh M, Marzony ET, Sahebkar A. Rosemary oil vs minoxidil 2% for the treatment of androgenetic alopecia: a randomized comparative trial. Skinmed. 2015 Jan-Feb;13(1):15-21.[↩][↩][↩]
- Markham, J.L. (1999) Biological activity of tea tree oil. In Tea Tree, the Genus Melaleuca ed. Southwell, I. and Lowe, R. pp. 169 190. Amsterdam: Harwood Academic publishers.[↩]
- Carson, C. F., Riley, T. V. & Cookson, B. D. (1998). Efficacy and safety of tea tree oil as a topical antimicrobial agent. Journal of Hospital Infection 40, 175–8.[↩]
- Carson, C. F., Hammer, K. A., & Riley, T. V. (2006). Melaleuca alternifolia (Tea Tree) oil: a review of antimicrobial and other medicinal properties. Clinical microbiology reviews, 19(1), 50–62. https://doi.org/10.1128/CMR.19.1.50-62.2006[↩]
- Carson, C. F. & Riley, T. V. (1995). Antimicrobial activity of the major components of the essential oil of Melaleuca alternifolia. Journal of Applied Bacteriology 78, 264–9.[↩]
- Carson, C. F., Cookson, B. D., Farrelly, H. D. & Riley, T. V. (1995). Susceptibility of methicillin-resistant Staphylococcus aureus to the essential oil of Melaleuca alternifolia. Journal of Antimicrobial Chemotherapy 35, 421–4.[↩]
- Brophy, J.J., Davies, N.W., Southwell, I.A., Stiff, I.A., Williams, L.R. (1989) Gas chromatographic quality control for oil of Melaleuca Terpinen‐4‐ol Type (Australian Tea Tree). Journal of Agricultural and Food Chemistry 37, 1330 1335.[↩]
- EMA/HMPC. Assessment report on Melaleuca alternifolia (Maiden and Betch) Cheel, M.linarifolia Smith , M.dissitiflora F.Mueller and /or other species of Melaleuca, Aetheroleum. Eur Med Agency. 2013. https://www.ema.europa.eu/en/documents/herbal-report/draft-assessment-report-melaleuca-alternifolia-maiden-betch-cheel-m-linariifolia-smith-m/other-species-melaleuca-aetheroleum_en.pdf[↩]
- Puvača N, Čabarkapa I, Petrović A, Bursić V, Prodanović R, Soleša D, Lević J. Tea tree (Melaleuca alternifolia) and its essential oil: antimicrobial, antioxidant and acaricidal effects in poultry production. Worlds Poult Sci J. 2019;75(2):235–246. doi: 10.1017/S0043933919000229[↩][↩]
- Brophy, J. J., N. W. Davies, I. A. Southwell, I. A. Stiff, and L. R. Williams. 1989. Gas chromatographic quality control for oil of Melaleuca terpinen-4-ol type (Australian tea tree). J. Agric. Food Chem. 37:1330-1335.[↩]
- Sakr FM, Gado AM, Mohammed HR, Adam AN. Preparation and evaluation of a multimodal minoxidil microemulsion versus minoxidil alone in the treatment of androgenic alopecia of mixed etiology: a pilot study. Drug Des Devel Ther. 2013 May 30;7:413-23. doi: 10.2147/DDDT.S43481[↩]
- Kalwat JI. The use of serenoa repens (Saw Palmetto) in hair care products. Biomed J Sci Tech Res. 2019;13(1):9725–9728. 10.26717/bjstr.2019.13.002348[↩]
- Dhariwala, MY, Ravikumar, P. An overview of herbal alternatives in androgenetic alopecia. J Cosmet Dermatol. 2019; 18: 966– 975. https://doi.org/10.1111/jocd.12930 [↩]
- Murugusundram S. Serenoa Repens: Does It have Any Role in the Management of Androgenetic Alopecia? J Cutan Aesthet Surg. 2009 Jan;2(1):31-2. doi: 10.4103/0974-2077.53097[↩][↩][↩]
- Chaterjee S, Agrawala S. Saw palmetto (Serenoa repens) in androgenic alopecia – an effective phytotherapy. Nat Prod Radiance. 2003;2:302–305.[↩]
- Iehlé C, Délos S, Guirou O, Tate R, Raynaud JP, Martin PM. Human prostatic steroid 5 alpha-reductase isoforms–a comparative study of selective inhibitors. J Steroid Biochem Mol Biol. 1995 Sep;54(5-6):273-9. doi: 10.1016/0960-0760(95)00134-l[↩]
- Prager N, Bickett K, French N, Marcovici G. A randomized, double-blind, placebo-controlled trial to determine the effectiveness of botanically derived inhibitors of 5-alpha-reductase in the treatment of androgenetic alopecia. J Altern Complement Med. 2002 Apr;8(2):143-52. doi: 10.1089/acm.2002.8.143. Erratum in: J Altern Complement Med. 2006 Mar;12(2):199.[↩]
- Wessagowit, V., Tangjaturonrusamee, C., Kootiratrakarn, T., Bunnag, T., Pimonrat, T., Muangdang, N. and Pichai, P. (2016), AGA treatment with Serenoa repens. Australasian Journal of Dermatology, 57: e76-e82. https://doi.org/10.1111/ajd.12352[↩]
- Saper RB, Rash R. Zinc: an essential micronutrient. Am Fam Physician. 2009 May 1;79(9):768-72. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2820120[↩]
- Kil MS, Kim CW, Kim SS. Analysis of serum zinc and copper concentrations in hair loss. Ann Dermatol. 2013 Nov;25(4):405-9. doi: 10.5021/ad.2013.25.4.405[↩]
- Tenenbaum S, Opdyke DL. Antimicrobial properties of the pyrithione salts. VII. In vitro methods for comparing pyrithiones to standard antimicrobials. Food Cosmet Toxicol. 1969 May;7(3):223-32. doi: 10.1016/s0015-6264(69)80326-3[↩]
- Guéniche A, Viac J, Lizard G, Charveron M, Schmitt D. Protective effect of zinc on keratinocyte activation markers induced by interferon or nickel. Acta Derm Venereol. 1995 Jan;75(1):19-23. doi: 10.2340/00015555751923[↩]
- Rostan, E.F., DeBuys, H.V., Madey, D.L. and Pinnell, S.R. (2002), Evidence supporting zinc as an important antioxidant for skin. International Journal of Dermatology, 41: 606-611. https://doi.org/10.1046/j.1365-4362.2002.01567.x[↩]
- STAMATIADIS, D., BULTEAU-PORTOIS, M.-C. and MOWSZOWICZ, I. (1988), Inhibition of 5α-reductase activity in human skin by zinc and azelaic acid. British Journal of Dermatology, 119: 627-632. https://doi.org/10.1111/j.1365-2133.1988.tb03474.x[↩]
- Reeder NL, Kaplan J, Xu J, Youngquist RS, Wallace J, Hu P, Juhlin KD, Schwartz JR, Grant RA, Fieno A, Nemeth S, Reichling T, Tiesman JP, Mills T, Steinke M, Wang SL, Saunders CW. Zinc pyrithione inhibits yeast growth through copper influx and inactivation of iron-sulfur proteins. Antimicrob Agents Chemother. 2011 Dec;55(12):5753-60. doi: 10.1128/AAC.00724-11[↩]
- Berger, R.S., Fu, J.L., Smiles, K.A., Turner, C.B., Schnell, B.M., Werchowski, K.M. and Lammers, K.M. (2003), The effects of minoxidil, 1% pyrithione zinc and a combination of both on hair density: a randomized controlled trial. British Journal of Dermatology, 149: 354-362. https://doi.org/10.1046/j.1365-2133.2003.05435.x[↩]
- Siavash M, Tavakoli F, Mokhtari F. Comparing the Effects of Zinc Sulfate, Calcium Pantothenate, Their Combination and Minoxidil Solution Regimens on Controlling Hair Loss in Women: A Randomized Controlled Trial. J Res Pharm Pract. 2017 Apr-Jun;6(2):89-93. doi: 10.4103/jrpp.JRPP_17_17[↩]
- Park H, Kim CW, Kim SS, Park CW. The therapeutic effect and the changed serum zinc level after zinc supplementation in alopecia areata patients who had a low serum zinc level. Ann Dermatol. 2009 May;21(2):142-6. doi: 10.5021/ad.2009.21.2.142[↩]
- Alhaj E, Alhaj N, Alhaj NE. Diffuse alopecia in a child due to dietary zinc deficiency. Skinmed. 2007 Jul-Aug;6(4):199-200. doi: 10.1111/j.1540-9740.2007.05881.x[↩]
- Sharquie K, Noaimi A, Shwail E. Oral zinc sulphate in treatment of alopecia areata (double blind; cross-over study) J Clin Exp Dermatol Res. 2012;3:150.[↩][↩]