What is a Ketogenic Diet ?
Developed in the 1920s by Hugh Conklin a faith healer in Battle Creek, Michigan, who treated children with epilepsy by depriving them of all foods by giving them nothing but water for up to 25 days, this diet induces a state that mimics carbohydrate starvation 1. As medications became available and effectively addressed seizures, the ketogenic diet fell out of favor. During the last few decades, researchers and clinicians have learned that ketogenic diet can be useful in children and adults with refractory epilepsy and a variety of other conditions. Once again, from the 1960s onwards keto diets — especially as the ‘Atkins Diet’ 2 have become widely known as one of the most common methods for obesity treatment. These very-low-carbohydrate high-fat ketogenic diets result in the substitution of ketone bodies as a source of energy (also known as nutritional ketosis). Ketosis is also produced during fasting periods, which is known as fasting ketosis. Recently, the combinations of nutritional ketosis and fasting ketosis, as well as nutritional ketosis alone, have been used as resources for weight loss management 3. Today’s ketogenic diet has been modified with scientifically proven adjustments to increase palatability and help with adherence. Recent work over the last decade or so has provided evidence of the therapeutic potential of ketogenic diets in many pathological conditions, such as diabetes, some forms of epilepsy, polycystic ovary syndrome, acne, Alzheimer’s disease, Parkinson’s disease, cancer, glaucoma, and the amelioration of respiratory and cardiovascular disease risk factors. And many Americans are using it to lose weight 4.
The keto diet (often termed ketogenic diet) is a very low-carb, high-fat diet that shares many similarities with the Atkins and low-carb diets. It lowers blood sugar and insulin levels, and shifts the body’s metabolism away from carbs and towards fat and ketones.
Ketogenic diet involves drastically reducing carbohydrate intake, and replacing it with fat. Ketogenic diets are characterized by a reduction in carbohydrates (usually to less than 50 g/day) and a relative increase in the proportions of protein and fat 5. The reduction in carbs puts your body into a metabolic state called ketosis. When this happens, your body becomes incredibly efficient at burning fat for energy. It also turns fat into ketones in the liver, which can supply energy for the brain.
This study showed that subjects with lower fat percentage, body mass index (BMI), as well as higher metabolic rate (resting energy expenditure or basal metabolic rate) have higher ketone buildups, and thus metabolize fat more efficiently 6. The researchers also found with no significant difference between ketogenic diets with 79% fat content and 90% fat content in terms of the significant buildup of ketone bodies.
The keto diet is so filling that you can lose weight without counting calories or tracking your food. One study found that people on a keto diet lost 2.2 times more weight than those on a calorie-restricted low-fat diet. Triglyceride and HDL cholesterol levels also improved. Another study found that participants on the keto diet lost 3 times more weight than those on the Diabetes USA recommended diet.
Keto diets can cause massive reductions in blood sugar and insulin levels. This, along with the increased ketones, has numerous health benefits.
When your body burns a lot of fat, the liver releases some of the partly burned fat molecules containing the last four carbon atoms into the blood; scientists call these ‘ketones’, which suppress hunger. It has been demonstrated that ketosis prevents the rise in ghrelin, a potent stimulator of appetite, which otherwise occurs as a result of diet-induced weight loss 7, that occurs after weight loss, and also increases the secretion of the hunger-suppressing hormone cholecystokinin 8. There are additional nutrient changes that contribute to hunger suppression 7. The heart and brain are the only two organs that can burn ketones, so the other possibility, not yet proven, is that ketones, like other nutrients such as glucose and fatty acids, suppress hunger directly by working on the brain. The point is that during the weight-loss phase, the patient can manufacture her own appetite suppressant. The downside to being mildly ketotic is that the ketones are volatile and, thus, can be breathed out, so the patient may develop ketotic breath. Chewing sugarless gum may help.
Although the mechanism of the efficacy of ketogenic diets has not been definitively established, it is commonly proposed that ketones suppress appetite 9 and it has been observed that study participants on ad libitum ketogenic diets spontaneously restrict their energy intake 10.
In the hypothalamus, signals from several circulating hormones and nutrients are integrated to regulate appetite and energy expenditure 11. The peripheral modulators of appetite include glucose 12, free-fatty acids 13 and hormones from the gastrointestinal tract, pancreas and adipose tissue, such as leptin, insulin, ghrelin, cholecystokinin (CCK), glucagon-like peptide 1, peptide YY and pancreatic polypeptide 14.
Following diet-induced weight loss, a number of compensatory changes occur, which encourage weight regain and restoration of energy balance. These include reductions in energy expenditure 15 and circulating leptin 16 and an increase in the orexigenic hormone ghrelin 17. It was recently reported that postprandial release of cholecystokinin (CCK), a hormone which increases satiety, was significantly reduced after diet-induced weight loss 18. However, when weight-reduced subjects were ketotic due to restriction of dietary carbohydrate, cholecystokinin (CCK) release was maintained at preweight loss concentrations, raising the possibility of an interaction between circulating ketones and hormonal mediators of appetite.
It should be noted that although the majority of randomized controlled trials comparing ad libitum ketogenic low-carbohydrate diets with low-fat diets have found greater weight loss over 6 months on the ketogenic diets, the difference is no longer observed at 12 months 19. In one of these studies, urinary ketones were significantly higher in the low-carbohydrate group compared with the low-fat group over the first 12 weeks, but no relationship was found between urinary ketones and weight loss 20. Only a minority of people have detectable urinary ketones after 3–6 months on low-carbohydrate diets 21.
Consumption of the ketogenic diet is characterized by elevated circulating levels of the ketone bodies acetoacetate, β-hydroxybutyrate and acetone, produced largely by the liver. During high rates of fatty acid oxidation, large amounts of acetyl-CoA are generated. These exceed the capacity of the tricarboxylic acid cycle and lead to the synthesis of the three ketone bodies within liver mitochondria. Plasma levels of ketone bodies rise, with acetoacetate and β-hydroxybutyrate increasing three-fold to four-fold from basal levels of 100 and 200 µmol/l, respectively 22. In the absence of glucose, the preferred source of energy (particularly of the brain), the ketone bodies are used as fuel in extrahepatic tissues. The ketone bodies are oxidized, releasing acetyl-CoA, which enters the tricarboxylic acid cycle.
In a double blind randomized crossover study on the safety and efficacy with respect to weight loss, cardiometabolic diseases of 21 days very low-calorie ketogenic diet (<800 kcal/day) 23. The study showed ketogenic diet may be used safely for a limited period up to 3 weeks to stimulate fat loss, to ensure weight loss, ectopic and visceral fat reduction, improve metabolism, without running the risk of the possibility of cardiovascular, renal and hepatic diseases 23.
However, in a small New Zealand study involving 5 endurance athletes (4 females, 1 male), who underwent a 10-week ketogenic (low-carbohydrate, high-fat) dietary intervention 24. All five athletes reported experiencing reduced energy levels initially and an inability to easily undertake high intense bouts. On the other hand, each athlete reported experiencing enhanced well-being, included improved recovery, improvements in skin conditions, reduced body fat and reduced inflammation 24.
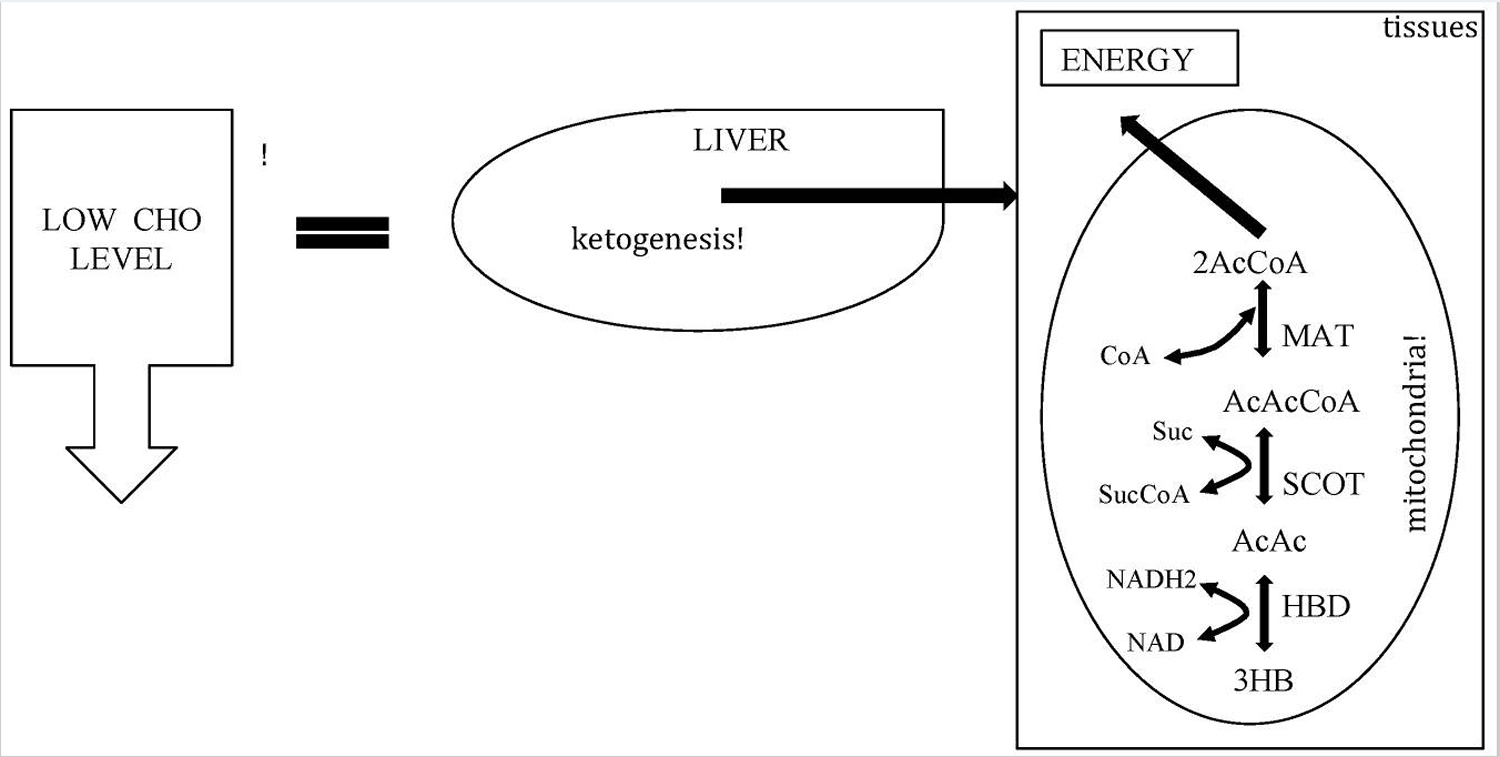
Figure 1. Metabolic pathway of ketosis and tissues ketolysis.
Footnote: CHO = carbohydrate.
[Source 25 ]What is Ketosis ?
Ketogenic diets have undoubtedly been shown to be effective, at least in the short to medium term, as a tool to fight obesity including people with diabetes who are obese 26, hyperlipidemia and some cardiovascular risk factors 27, 28, 29, but ketogenic diets also raise some concerns among physicians 30. Many of the concerns about the use of ketogenic diet as therapeutic tools could be attributed to a broad lack of knowledge about the physiological mechanisms involved. Ketogenic diets induce a metabolic condition named “physiological ketosis” by Hans Krebs, to distinguish it from the pathological diabetic ketosis 31.
Under normal diet with average amount of carbohydrates and fat, insulin activates key enzymes in the glucose metabolic pathways, which store energy derived from carbohydrates, and when there is an absence or scarcity of dietary carbohydrates the resulting reduced insulin level leads to a reduction in lipogenesis and fat accumulation. After a few days of fasting, or of drastically reduced carbohydrate consumption (below 20 g/day), glucose reserves become insufficient both for normal fat oxidation via the supply of oxaloacetate in the Krebs cycle (which gave origin to the phrase ‘fat burns in the flame of carbohydrate’) and for the supply of glucose to the central nervous system 32, 33, 34, 35.
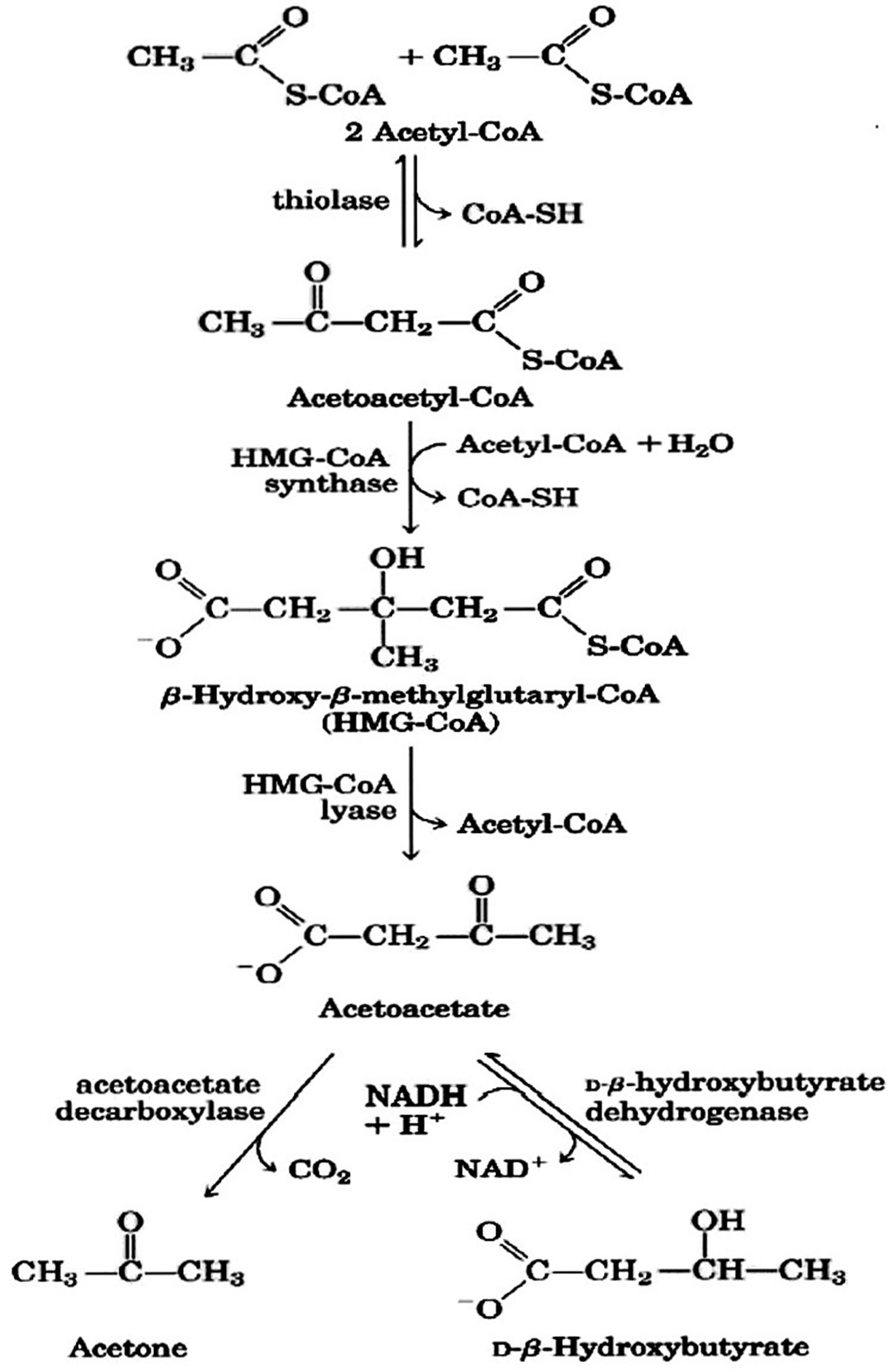
Figure 2. Ketone bodies formation from acetyl-CoA (acetyl coenzyme A).
Footnote: Ketone bodies are used by tissues as a source of energy through a pathway that involves firstly that β-hydroxybutyric acid is converted back to acetoacetate (AcAc) this is then transformed into acetoacetyl-CoA and, finally, two molecules of acetyl-CoA are formed from acetoacetyl-CoA which are used in the Krebs cycle.
[Source 25 ]The central nervous system cannot use fat as an energy source; hence, it normally utilizes glucose. After 3–4 days without carbohydrate consumption the central nervous system is ‘forced’ to find alternative energy sources, and as demonstrated by the classic experiments of Cahill and colleagues 32 this alternative energy source is derived from the overproduction of acetyl coenzyme A (CoA). This condition seen in prolonged fasting, type 1 diabetes and high-fat/low-carbohydrate diets (aka ketogenic diets) leads to the production of higher-than-normal levels of so-called ketone bodies, that is, acetoacetate, β-hydroxybutyric acid and acetone—a process called ketogenesis and which occurs principally in the mitochondrial matrix in the liver (see Figures 1 and 2) 36. It is important to underline that the liver produces ketone bodies, but is unable to utilize them because the absence of the enzyme 3-ketoacyl CoA transferase required to convert acetoacetate into acetoacetyl-CoA 37. Even though the main ketone body produced in the liver is acetoacetate, the primary circulating ketone is β-hydroxybutyrate 38. Under normal conditions of adequate dietary carbohydrate, the production of free acetoacetic acid is negligible (<0.3 mmol/l) and this compound, transported via the blood stream, is rapidly metabolized by various tissues, especially the skeletal and heart muscles. In conditions of overproduction of acetoacetic acid, it accumulates above normal levels and part of it is converted to the other two ketone bodies leading to ketonemia and ketonuria (presence of ketone bodies in the blood and urine).
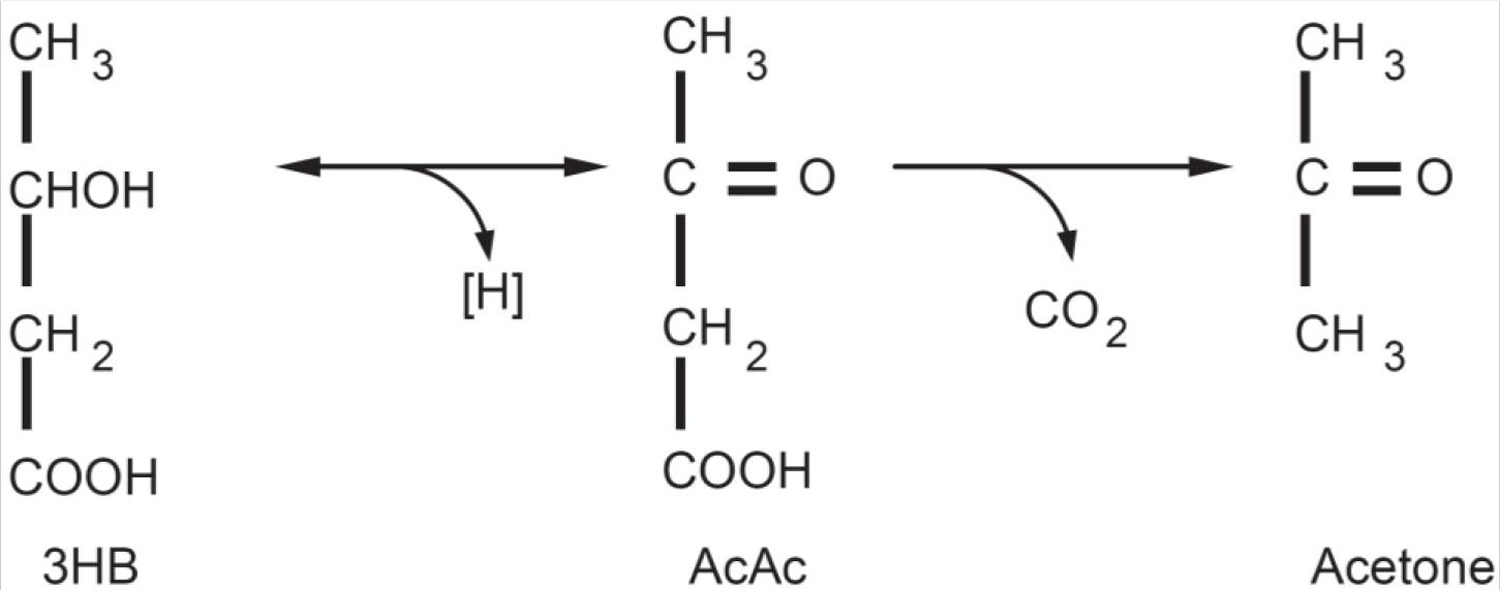
Acetone (produced by spontaneous decarboxylation of acetoacetate), being a very volatile compound, is eliminated mainly via respiration in the lungs, hence the characteristic sweet “fruity breath” breath odour of ketosis is caused by acetone, which, being a very volatile compound, is eliminated mainly via respiration in the lungs. Acetone even though it does not have metabolic functions, its presence can be useful from a clinical diagnostic point of view. Thus it is to be considered that a “fruity breath” indicates a condition of ketosis that could be physiological (fasting, low carbohydrate diet, post exercise) 38.
- It should be stressed that, as shown in Table 1 (below), blood glucose level, even though reduced, remains within physiological levels 39. In fact glucose is formed from two sources: glucogenic amino acids and from glycerol liberated via lysis from triglycerides 40, 41. The importance of the second source increases progressively during the ketosis condition. In the first days of a ketogenic diet the main source of glucose is via neoglucogenesis from amino acids, as the days goes by, the contribution of amino acids decreases whilst the amount of glucose derived from glycerol increases. As a matter of fact glycerol (released from triglyceride hydrolysis) can produce more than 16% of glucose in the liver during a ketogenic diet and about 60% after a few days of complete fasting 40. According to Bortz (1972) of the new glucose formed from protein and glycerol 38% is derived from glycerol in the lean and 79% in the obese 42.
It is of interest to underline that ketosis is a metabolic state characteristic of humans. humans are more susceptible to ketosis-induced fasting due to the greater brain/body mass (this can explain why newborns are more susceptible to ketosis) 43. Under normal conditions the concentration of ketone bodies is very low (<0.3 mmol/L) compared to glucose (approx. 4 mmol) and since glucose and ketone bodies have a similar Km (or Michaelis-Menten constant) for glucose transport to the brain, the ketone bodies begin to be utilised as an energy source by the brain when they reach a concentration of about 4 mmol/L 44 which is close to the Km for the monocarboxylate transporter 45.
The ketone bodies begin to be utilized as an energy source by the central nervous system when they reach a concentration of about 4 mmol/l (normal level is <0.3 mmol/l), which is close to the Km for the monocarboxylate transporter 44, 36. The brain will use ketone bodies whenever provided with them (i.e., whenever blood ketone body levels rise). However, continued use of some glucose appears obligatory 46 and is supplied by way of hepatic gluconeogenesis 47.
Figure 3. Ketone bodies: Acetoacetate (AcAc) is the principal ketone body. It is produced and utilized during intermediary metabolism and other ketone bodies are derived from it. Acetone is produced by the spontaneous decarboxylation of acetoacetate and is important from the clinical point of view because it is responsible for the fruity sweet odour of infant ketoacidosis. β-Hydroxybutyric acid (3HB) is produced via the reduction of acetoacetate. From a strictly biochemical point of view it is not actually a ketone body since the ketonic moiety is reduced to a hydroxyl group; it is though grouped among the ketone bodies. 3HB is relatively stable biochemically and is transported to the tissues where it is reconverted to AcAc.
[Source 25]Ketosis or ketoacidosis is a physiological state of increased ketones (ketone bodies acetoacetate or acetoacetic acid, β-hydroxybutyrate or β -hydroxybutyric acid and acetone) level in the blood, blood and urine due to relatively high lipid oxidation rates. Detecting altered concentrations of ketones in the breath, blood, and urine is crucial for the diagnosis ketosis and for monitoring rapid and dramatic changes in ketones as valuable diagnoses for lipid oxidation and metabolism 48. Several studies have clearly demonstrated that metabolic imbalance in type I diabetes has led to ketoacidosis of blood, leading to elevated ketone levels with arterial pH < 7.3 and bicarbonate < 15 mEq/L, and causing arresting of major organ functions 49. In addition to acidosis, studies have also shown that elevated ketone levels are a natural metabolic response to negative energy balance, wherein caloric intake is smaller than total energy expenditure, and the body burns stored fat to produce the needed energy 50, leading to a state of ketosis known as fasting ketosis. Fasting ketosis has been used as an indicator of the effectiveness of weight loss 51, 52, 53. Ketosis also occurs in situations where caloric intake equals total energy expenditure, specifically in a diet that contains high percentage of fat (>60%) and/or low carbohydrate. This state of ketosis has been referred to as nutritional ketosis 54, 55. Nutritional ketosis has been investigated as a treatment for epilepsy because ketones are thought to provide energy to the brain, which reduces epileptic seizures 56, 57. In addition, ketosis buildup capability resulting from a combination of nutritional ketosis and fasting ketosis has been associated with weight loss efficiency and positive health outcomes 58, 59, 60.
| Blood levels | Normal diet | Ketogenic diet | Diabetic ketoacidosis |
|---|---|---|---|
| Glucose (mg/dl) | 80–120 | 65–80 | >300 |
| Insulin (μU/l) | 6–23 | 6.6–9.4 | ≅ 0 |
| KB conc (mℳ/l) | 0.1 | 7/8 | >25 |
| pH | 7.4 | 7.4 | <7.3 |
Note (Table 1): Nutritional and Functional Ketosis are completely physiological mechanism and it was the biochemist Hans Krebs who first referred to physiological ketosis to differentiate it from the pathological ketoacidosis seen in type 1 diabetes 62. In physiological ketosis (which occurs during very-low-calorie ketogenic diets and in fasting), ketonemia reaches maximum levels of 7/8 mmol/l (it does not go higher precisely because the central nervous system efficiently uses these molecules for energy in place of glucose) and with no change in pH, whereas in uncontrolled diabetic ketoacidosis it can exceed 20 mmol/l with a concomitant lowering of blood pH 63, 64. Diabetic patients know that the detection in their urine of the ketone bodies is a danger signal that their diabetes is poorly controlled. Indeed, in severely uncontrolled diabetes, if the ketone bodies are produced in massive supranormal quantities, they are associated with ketoacidosis. In this life-threatening complication of diabetes mellitus, the acids 3-hydroxybutyric acid and acetoacetic acid are produced rapidly, causing high concentrations of protons, which overwhelm the body’s acid-base buffering system 47.
It is very important to note the difference between physiological ketosis (nutritional and functional ketosis) and pathological ketoacidosis (diabetic ketoacidosis).
- During Physiological ketosis, ketonemia (blood ketone bodies) reaches maximum levels of 7/8 mmol/L with no change in pH while in uncontrolled diabetic ketoacidosis it can exceed 20 mmol/L with a concomitant lowering of blood pH 65 (see Table 1). Blood levels of ketone bodies in healthy people do not exceed 8 mmol/L precisely because the central nervous system (brain) efficiently uses these molecules for energy in place of glucose.
- Diabetic Ketoacidosis is an acute metabolic complication of type 1 diabetes mellitus and develops when insulin levels are insufficient to meet the body’s basic metabolic requirements. Diabetic ketoacidosis is less common in type 2 diabetes mellitus, but it may occur in situations of unusual physiologic stress. It is characterized by hyperglycemia (high blood glucose), hyperketonemia (very high blood ketone bodies), and metabolic acidosis (low blood pH). Hyperglycemia causes an osmotic diuresis with significant fluid and electrolyte loss. It causes nausea, vomiting, and abdominal pain and can progress to cerebral edema, coma, and death. Diabetic ketoacidosis is diagnosed by detection of hyperketonemia and anion gap metabolic acidosis in the presence of hyperglycemia 66.
Under physiological ketosis or ketoacidosis, the liver metabolizes fatty acids to produce two water-soluble types of ketones: acetoacetic acid and beta-hydroxybutyric acid. A third type of ketone, i.e., acetone, is also produced by the enzymatic decarboxylation of acetoacetic acid. Due to its high vapor pressure, acetone crosses the membrane barrier into the alveoli of the lung and the airway. As a result, acetone is normally present in breath. Since, the ketone level in breath is reflective of ketone level in blood, breath acetone has been considered a reliable indicator of ketosis in adults consuming ketogenic meals 22 and can be used to predict plasma ketone bodies in children with epilepsy who are on a ketogenic diet 67. Most recently, breath acetone has been used as a new ketone biomarker because it is non-invasive, convenient, and an accurate reflection of the body’s ketone level 68. Breath testing for ketones has a high sensitivity and specificity and appears to be a noninvasive, convenient, and repeatable method for the diagnosis and therapeutic monitoring of ketosis 69.
Furthermore, there are other conditions, such as exercise that can actually decrease ketone levels in the blood by using ketone as an energy source in the muscles 70, 71. For the reasons described above, ketone levels are affected by several factors, such as energy balance, diet composition, diabetes (type 1 and 2) and physical activities, all of which underscore the importance of determining the accuracy of ketone levels, blood glucose and pH levels.
Different Types of Keto Diets
There are several versions of the keto diet, the standard keto diet (SKD) is the most researched and most recommended.
- Standard keto diet (SKD): This is a very low-carb, moderate-protein and high-fat diet. It typically contains 75% fat, 20% protein and only 5% carbs. A standard keto diet is an effective way to lose weight and lower risk factors for disease.
- Cyclical keto diet (CKD): This diet involves periods of higher-carb refeeds, such as 5 keto days followed by 2 high-carb days.
- Targeted keto diet (TKD): This diet allows you to add carbs around workouts.
- High-protein keto diet: This is similar to a standard ketogenic diet, but includes more protein. The ratio is often 60% fat, 35% protein and 5% carbs.
However, only the standard and high-protein ketogenic diets have been studied extensively. Cyclical or targeted ketogenic diets are more advanced methods, and primarily used by bodybuilders or athletes. The information in this article mostly applies to the standard ketogenic diet, although many of the same principles also apply to the other versions.
**The keto diet can boost insulin sensitivity and cause fat loss, leading to drastic improvement for type 2 diabetes and prediabetes.
Studies have now shown that the diet can have benefits for a wide variety of different health conditions. However, keep in mind that research into many of these areas is far from conclusive:
- Heart disease: The keto diet can improve risk factors like body fat, HDL levels, blood pressure and blood sugar.
- Cancer: The diet is currently being used to treat several types of cancer and slow tumor growth.
- Alzheimer’s disease: The diet may reduce symptoms of Alzheimer’s and slow down the disease’s progression.
- Epilepsy: Research has shown that the ketogenic diet can cause massive reductions in seizures in epileptic children.
- Parkinson’s disease: One study found that the diet helped improve symptoms of Parkinson’s disease.
- Polycystic ovary syndrome: The ketogenic diet can help reduce insulin levels, which may play a key role in polycystic ovary syndrome.
- Brain injuries: One animal study found that the diet can reduce concussions and aid recovery after brain injury.
- Acne: Lower insulin levels and eating less sugar or processed foods may help improve acne.
The Ketogenic Diet Foods
Ketogenic diets allow you to eat real foods in the form of natural fats and protein (meat, fish, poultry) but severely restrict the amount of carbohydrates (sugars and starches) you are allowed each day. The ketogenic diet is an extremely high-fat diet that requires you to eat four times as many fat calories as calories from protein or carbohydrates. A meal might include a small portion of chicken, a little bit of fruit, and a lot of fat, typically butter or cream. The diet was originally developed to treat epileptic children, but there are many variations, including the Atkins diet. You can achieve ketosis through fasting, diet, exogenous ketones, or a combination. Your brain and body begin to use ketones (derived from stored or ingested fat) for energy instead of blood sugar (glucose)—a state called ketosis.
On a standard American diet, carbohydrate intake is high (about 40-60% of calories) while fat intake, and especially saturated fat, is limited. Diets high in carbohydrate have the effect of elevating blood sugar and a hormone called insulin over time. The chronically higher levels of blood sugar and insulin result in a condition called insulin resistance or metabolic syndrome which can result in constant hunger, weight gain and a long list of disease outcomes.
In contrast, carbohydrate intake on a keto diet is only about 2-4% of calories. By reducing the number of carbohydrates a person eats, the body is forced to burn fat for energy, a process called ketosis. This ketosis is the same process that kicks in when someone is fasting — on purpose or because of starvation. Fasting has been a traditional seizure treatment for centuries. When carb intake is low, meals are delicious and satiating. Hunger goes away, and more importantly, this dietary change has some powerful and beneficial metabolic effects on the human body, in part because it lowers blood sugar and insulin levels. The bottom line here is that you can increase your energy and improve your health simply by changing the way you eat.
How Do Keto Diets Work ?
When carbohydrates (sugar and starch) are digested, they are broken down into blood sugar (glucose) in the body. More carbohydrate intake results in higher blood sugar. If you reduce carb intake and instead eat more fat and protein, it results in a switch in metabolic pathways from using sugar as a primary fuel to burning stored and dietary fat as a primary fuel.
As more fat is burned, some of it is converted in ketone bodies. As blood glucose and insulin levels drop and ketone levels rise, muscle tissue, including the heart, use the available fatty acids in the bloodstream as fuel and the brain begins using ketones as an energy source. Being in this metabolic state of “nutritional ketosis” (blood sugar is low and ketone levels are moderate) has some powerful benefits. There is strong research evidence that low carb, high fat metabolic diets are effective for treating the following medical conditions:
- Diabetes: Since carbohydrate is the prime driver of higher blood sugar, diet rich is carbohydrate is detrimental to diabetic health because it results in blood sugar spikes and crashes, which in turn causes a greater need for medication and insulin. Those high blood sugars also result in the complications of diabetes. In contrast, a ketogenic diet reduces and in many cases, eliminates the need for diabetic medications and lowers the number of insulin units needed to manage blood sugar. For people with Type 2 diabetes, ketogenic diets remove the trigger (carbohydrate intake) and reverse the underlying insulin resistance which causes the disease. As a result, long term complications are reversed or avoided. Type 1 Diabetes: Lowering carb intake and increasing fat intake is also beneficial for people with Type 1 diabetes and can help lower the number and severity of hypoglycemic episodes, reduce HbA1c scores and minimize future complications.
- Weight Loss: There is no doubt that there is strong supportive evidence that the use of ketogenic diets – low carb, high fat keto diets have been proven in study after study as the best diet for weight loss because the diet works with your biochemistry instead of against it. However the mechanisms underlying the effects of ketogenic diets on weight loss is still a subject of debate. Atkins’ original hypothesis suggested that weight loss was induced by losing energy through excretion of ketone bodies, but more recently different hypotheses have been proposed: one hypothesis is that the use of energy from protein in ketogenic diet is an “expensive” process for the body and so can lead to a “waste of calories” and therefore increased weight loss compared to other “less expensive” diets. The hypothesized mechanisms of keto diet’s weight loss effects:
- (1) Reduction in appetite due to higher satiety effect of proteins, effects on appetite control hormones and to a possible direct appetite suppressant action of the ketone bodies;
(2) Reduction in lipogenesis and increased lipolysis;
(3) Greater metabolic efficiency in consuming fats highlighted by the reduction in the resting respiratory quotient;
(4) Increased metabolic costs of gluconeogenesis and the thermic effect of proteins.
The key to correctly implement a ketogenic diet plan is to remember that you are exchanging carbohydrate containing foods with a higher fat intake and a moderate protein consumption.

Ketogenic Diet and Weight Loss
There is no doubt that there is strong supportive evidence that the use of ketogenic diets in weight-loss therapy is effective; however, there are contrasting theories regarding the mechanisms through which they work 38. Some researchers suggest that there are not in fact any metabolic advantages in low-carbohydrate diets and that weight loss results simply from reduced caloric intake, probably due to the increased satiety effect of protein 72. Others instead promote the hypothesis that there is indeed a distinct metabolic advantage, which has recently been explored in more detail, raising interest in the role of very-low-carbohydrate ketogenic diets 72 in weight loss and effects on metabolism in general 73. Adhering to these traditional concepts the US Department of Agriculture has concluded that diets, which reduce calories, will result in effective weight loss independent of the macronutrient composition, which is considered less important, even irrelevant 74, 75. In contrast with these views, the majority of studies demonstrate that subjects who follow a low-carbohydrate diet lose more weight during the first 3–6 months compared with those who follow balanced diets 76, 77, 78. One hypothesis is that the use of energy from proteins in very-low-carbohydrate ketogenic diets is an ‘expensive’ process for the body and so can lead to a ‘waste of calories’, and therefore increased weight loss compared with other ‘less-expensive’ diets 73, 79, 80. The average human body requires 60–65 g of glucose per day, and during the first phase of a diet very low in carbohydrates this is partially (16%) obtained from glycerol, with the major part derived via gluconeogenesis from proteins of either dietary or tissue origin 72. The energy cost of gluconeogenesis has been confirmed in several studies 81 and it has been calculated at ∼400–600 Kcal/day (due to both endogenous and food source proteins 79. Despite this, there is no direct experimental evidence to support this intriguing hypothesis; on the contrary, a recent study reported that there were no changes in resting energy expenditure after a very-low-carbohydrate ketogenic diets 82. A simpler, perhaps more likely, explanation for improved weight loss is a possible appetite-suppressant action of ketosis. The mechanism for this is not established but evidence supports direct action of ketone bodies together with modifications in levels of hormones, which influence appetite, such as ghrelin and leptin 83.
Here is the summary (listed in order of importance and available evidence) that the weight-loss effect of very-low-carbohydrate ketogenic diet seems to be caused by several factors:
- Reduction in appetite due to higher satiety effect of proteins 72, 84, effects on appetite control hormones 83 and to a possible direct appetite-suppressant action of the ketone bodies 85.
- Reduction in lipogenesis and increased lipolysis 86, 64.
- Reduction in the resting respiratory quotient and, therefore, greater metabolic efficiency in consuming fats 82, 87.
- Increased metabolic costs of gluconeogenesis and the thermic effect of proteins 73, 79.
Ketogenic Diet and Type 2 Diabetes
Recently, four studies have re-examined the effect of carbohydrate restriction on type 2 diabetes. One outpatient study enrolled 54 participants with type 2 diabetes (out of 132 total participants) and found that hemoglobin A1c improved to a greater degree over one year with a low-carbohydrate diet compared with a low-fat, calorie-restricted diet 88, 89. Another study enrolled 8 men with type 2 diabetes in a 5-week crossover outpatient feeding study that tested similar diets 90. The participants had greater improvement in glyco-hemoglobin (hemoglobin A1c or HbA1c) while on the low-carbohydrate diet than when on a eucaloric (normal calorie) low-fat diet. The third study was an inpatient feeding study in 10 participants with type 2 diabetes 91. After only 14 days, hemoglobin A1c improved from 7.3% to 6.8%. In the fourth study, 16 participants with type 2 diabetes who followed a 20% carbohydrate diet had improvement of hemoglobin A1c from 8.0% to 6.6% over 24 weeks 92. Only these latter three studies targeted glycemic control as a goal, and two of these were intensely-monitored efficacy studies in which all food was provided to participants for the duration of the study 90, 91. Three of the studies 91, 89, 92 mentioned that diabetic medications were adjusted but only one of them provided detailed information regarding these adjustments 92. This information is critical for patients on medication for diabetes who initiate a low-carbohydrate diet because of the potential for adverse effects resulting from hypoglycemia.
In studies that have evaluated well-formulated very-low-carbohydrate diets and documented high rates of compliance in individuals with type 2 diabetes, results have been nothing short of remarkable. Bistrian et al. 93 reported withdrawal of insulin and major weight loss in a matter of weeks in type 2 diabetes individuals who were fed a very-low-calorie and -carbohydrate diet. Gumbiner et al. 94 fed obese type 2 diabetes individuals two types of hypocaloric (650 kcal) diets for 3 weeks, they were matched for protein but one was much lower in carbohydrate content (24 vs 94 g/day). As expected, the lower-carbohydrate diet resulted in significantly greater levels of circulating ketones (∼3 mmol/l), which was strongly associated with a lower hepatic glucose output. Interestingly, there was a strong inverse correlation between circulating ketones and hepatic glucose output, suggesting that higher levels of ketones are associated with more favourable effects on glycaemic control in diabetics. More recently, Boden et al. 95 performed an in-patient study in obese type 2 diabetes individuals who were fed a low-carbohydrate (<20 g/day) diet for 2 weeks. Plasma glucose fell from 7.5 to 6.3 mmol/l, haemoglobin A1c decreased from 7.3 to 6.8% and there were dramatic improvements (75%) in insulin sensitivity.
In a longer study 96 obese type 2 diabetes individuals were prescribed a well-formulated ketogenic diet for 56 weeks, and significant improvements in both weight loss and metabolic parameters were seen at 12 weeks and continued throughout the 56 weeks as evidenced by improvements in fasting circulating levels of glucose (−51%), total cholesterol (−29%), high-density lipoprotein–cholesterol (63%), low-density lipoprotein–cholesterol (−33%) and triglycerides (−41%). It is of interest to note that in a recent study in overweight non-diabetic subjects, it was reported that during ketosis fasting glucose was not affected, but there was an elevation in post-prandial blood glucose concentration. This data suggests a different effect of ketosis on glucose homeostasis in diabetic and non-diabetic individuals 83. Other studies support the long-term efficacy of ketogenic diets in managing complications of type 2 diabetes 97, 98. Although significant reductions in fat mass often results when individuals restrict carbohydrate, the improvements in glycaemic control, haemoglobin A1c and lipid markers, as well as reduced use or withdrawal of insulin and other medications in many cases, occurs before significant weight loss occurs. Moreover, in normal caloric experiments individuals with insulin resistance showed dramatically improved markers of metabolic syndrome than diets lower in fat 99. It is interesting in this respect that a recent extremely large epidemiological study reported that diabetes risk is directly correlated, in an apparently causative manner, with sugar intake alone, independently of weight or sedentary lifestyle 100.
This very small study 101 with 28 subjects with type 2 diabetes (age 35–75 years; body mass index (BMI) >25 kg/m2; and fasting serum glucose >125 mg/dL or hemoglobin A1c >6.5% without medications, or treatment with oral hypoglycemic agents and/or insulin) to evaluate the effects of a low-carbohydrate, ketogenic diet (≤20 g carbohydrate per day and fats and oils were not restricted except that intake of trans fats was to be minimized) in overweight and obese patients with type 2 diabetes over 16 weeks. In summary, the low-carbohydrate ketogenic diet resulted in significant improvement of glycemia, as measured by fasting glucose and hemoglobin A1c, in patients with type 2 diabetes. More importantly, this improvement was observed while diabetes medications were reduced or discontinued in 17 of the 21 participants, and were not changed in the remaining 4 participants. Participants also experienced reductions in body weight, waist circumference, serum triglycerides and percent body fat but these improvements were moderate. While body weight decreased significantly (-8.5 kg) in these 21 diabetic participants, the mean weight loss was less compared with what we observed in the low-carbohydrate ketogenic diet participants of an another trial (-12.0 kg) 102. Most impressive is that improvement in hemoglobin A1c was observed 101.
In summary, individuals with metabolic syndrome, insulin resistance and type 2 diabetes (all diseases of carbohydrate intolerance) are likely to see symptomatic as well as objective improvements in biomarkers of disease risk if they follow a well-formulated very-low-carbohydrate diet. Glucose control improves not only because there is less glucose coming in, but also because systemic insulin sensitivity improves as well.
Ketogenic Diet and Cardiovascular disease
Several lines of evidence point to beneficial effects of low-carbohydrate ketogenic diet on cardiovascular risk factors. In the past, there have been doubts expressed about their long-term safety and increased effectiveness compared with ‘balanced’ diets 103, and clearly negative opinions regarding possible deleterious effects on triglycerides and cholesterol levels in the blood 104. However, the majority of recent studies seem instead to amply demonstrate that the reduction of carbohydrates to levels that induce physiological ketosis (see above ‘What is ketosis?’ section) can actually lead to significant benefits in blood lipid profiles 76, 78, 99. The low-carbohydrate ketogenic diet effect seems to be particularly marked on the level of blood triglycerides 87, 105, but there are also significant positive effects on total cholesterol reduction and increases in high-density (HDL) “good” cholesterol 87, 105, 106. Furthermore, low-carbohydrate ketogenic diet have been reported to increase the size and volume of low-density lipoprotein–cholesterol particles 106, which is considered to reduce cardiovascular disease risk, as smaller low-density lipoprotein particles have a higher atherogenicity. There are also direct diet-related effects on overall endogenous cholesterol synthesis. A key enzyme in cholesterol biosynthesis is 3-hydroxy-3-methylglutaryl–CoA reductase (the target for the drug statins), which is activated by insulin, which means that an increase in blood glucose and consequently of insulin levels will lead to increased endogenous cholesterol synthesis. A reduction in dietary carbohydrate will have the opposite effect and this, coupled with the additional inhibition by dietary cholesterol and fats on endogenous synthesis, is likely to be the mechanism via which physiological ketosis can improve lipid profiles. Hence, there are strong doubts about the negative effects of dietary fats when they are consumed as part of a low-carbohydrate ketogenic diet, on cholesterol and triglycerides blood levels, whereas there are strong pointers to the beneficial effects of low-carbohydrate ketogenic diet on these cardiovascular risk parameters 99, 105.
Ketogenic Diet and Polycystic Ovary Syndrome (PCOS)
Polycystic ovary syndrome (PCOS) is a common endocrine disorder in females, with a high prevalence (6–10%) 107; symptoms include hyperandrogenism, ovulatory dysfunction, obesity, insulin resistance and subfertility. Insulin resistance and related hyperinsulinaemia is actually a very common feature affecting about 65–70% of women with polycystic ovary syndrome 108; it is seen most frequently in obese patients, affecting 70–80%, compared with only 20–25% of lean polycystic ovary syndrome sufferers. Despite this observation, insulin resistance and hyperinsulinaemia appear to be linked to polycystic ovary syndrome independently of obesity, and modifications in the normal cellular mechanisms of insulin signalling have been demonstrated in both lean and obese patients. Furthermore, high blood levels of insulin can act by increasing androgenous hormonal stimulation of the ovarian theca cells as well as potentiating gonadotropin-stimulated ovarian androgen steroidogenesis—although recent data has suggested that the insulin-induced increase in ovarian hormone secretion is not accompanied by increased steroid metabolism 109. Hyperinsulinaemia may also affect the central actions of androgen by impairing progesterone inhibition of the gonadotropin-releasing hormone pulse generator 110. Insulin has also been shown to increase expression of adrenal steroidogenic enzyme mRNA47 as well as adrenal responsiveness to adrenocorticotropic hormone 111.
Women with PCOS frequently demonstrate many of the signs related to metabolic syndrome, such as insulin resistance, obesity, glucose intolerance, type 2 diabetes, dyslipidemia and also high levels of inflammation. Suggested treatments include those that reduce insulin resistance/hyperinsulinaemia, such as lifestyle modifications (exercise, diet and weight loss) and pharmacological treatments that include the administration of thiazolidinediones or metformin. It is evident that any interventions that improve insulinaemia and reduce body weight may also be effective in reducing hyperandrogenism, normalizing ovulation and reducing the various symptoms of polycystic ovary syndrome.
Finally, although we only have preliminary evidence of the positive effects of very-low-carbohydrate ketogenic diet in polycystic ovary syndrome 112, there are clear mechanisms that are consistent with the physiological plausibility of such dietary therapy.
Ketogenic Diet and Acne
In recent years there have been an increasing number of studies published, suggesting that at least for certain food types there is a nutritional influence on the development of acne. The negative effects seem to lie in the capacity of some foods/nutrients to stimulate proliferative pathways that in turn stimulate development of acne—suspect foods include those with a high glycaemic load and milk 61, 113, 114. Other evidence comes from several studies reporting that the prevalence of acne varies significantly between different populations and is substantially lower in non-Westernized populations that follow traditional diets 115, a common factor among these traditional diets being a low glycaemic load 116. Various studies have provided evidence that high-glycaemic-load diets are implicated in the cause of acne through their capacity to stimulate insulin, androgen bioavailability and insulin-like growth factor-1 (IGF-1) activity, whereas the beneficial effects of low-glycaemic-load diets, apart from weight and blood glucose levels, also include improved skin quality 114. The clinical and experimental evidence does in fact suggest ways in which insulin can increase androgen production and affect via induction of steroidogenic enzymes 117, the secretion by the pituitary gland of gonadotropin-releasing hormone and the production of sex hormone-binding globulin.48 Insulin is also able to reduce serum levels of IGF-binding protein-1 increasing the effect of IGF-1 118. These insulin-mediated actions can therefore influence diverse factors that underlie the development of acne such as:
- The increased proliferation of basal keratinocytes within the pilosebaceous ducts.
- An abnormal desquamation of the follicular epithelium.
- Increased androgen-mediated sebum production.
- Colonization of the stratum corneum by Propionibacterium acnes and consequent inflammation.46
In summary, there is persuasive, although not yet conclusive, clinical and physiological evidence that the ketogenic diet could be effective in reducing the severity and progression of acne and randomized clinical trials will be required to resolve the issue 61.
Ketogenic Diet and Epilepsy
At present, strong evidence exists that the ketogenic diet protects against seizures in children with difficult-to-treat epilepsy 119. Recent reports have raised the possibility that the diet may also improve the long-term outcome in such children 120, 121. In these studies, children with intractable epilepsy who remained on the ketogenic diet for more than 1 year and who experienced a good response to the diet, often had positive outcomes at long-term follow-up 3–6 years after the initiation of diet. Forty-nine percent of the children in this cohort experienced a nearly complete (≥ 90%) resolution in seizures. Surprisingly, even those children who remained on the diet for 6 months or less (most of these children terminated the diet because of an inadequate response) may have obtained a long-term benefit from exposure to the diet. Thirty-two percent of these children had a ≥ 90% decrease in their seizures and 22% became seizure free even without surgery. The diet also allowed a decrease or discontinuation of medications without a relapse in seizures. Of course, in the absence of a control group, it is not possible to be certain that the apparent good response in these children is simply the natural history of the epilepsy in the cohort studied, although these children had, by definition, intractable epilepsy before starting the diet. In any case, the results raise the possibility that the ketogenic diet, in addition to its ability to protect against seizures, may have disease-modifying activity leading to an improved long-term outcome. It is noteworthy that none of the currently marketed antiepileptic drugs has been demonstrated clinically to possess such a disease-modifying effect 122, 123. Determining whether the ketogenic diet truly alters long-term outcome will require prospective controlled trials.
Ketogenic Diet and Parkinson’s disease
One recently published clinical study tested the effects of the ketogenic diet on symptoms of Parkinson’s disease 124. In this uncontrolled study, Parkinson’s disease patients experienced a mean of 43% reduction in Unified Parkinson’s Disease Rating Scale scores after a 28-day exposure to the ketogenic diet. All participating patients reported moderate to very good improvement in symptoms. Further, as in Alzheimer’s disease, consumption of foods containing increased amounts of essential fatty acids has been associated with a lower risk of developing Parkinson’s disease 125.
Ketogenic Diet and Alzheimer’s disease
Recent studies have raised the possibility that the ketogenic diet could provide symptomatic benefit and might even be disease modifying in Alzheimer’s disease. Thus, Reger et al. 126 found that acute administration of medium-chain triglycerides improves memory performance in Alzheimer’s disease patients. Further, the degree of memory improvement was positively correlated with plasma levels of β-hydroxybutyrate produced by oxidation of the medium-chain triglycerides. If β-hydroxybutyrate is responsible for the memory improvement, then the ketogenic diet, which results in elevated β-hydroxybutyrate levels, would also be expected to improve memory function. When a patient is treated for epilepsy with the ketogenic diet, a high carbohydrate meal can rapidly reverse the antiseizure effect of the diet 127. It is therefore of interest that high carbohydrate intake worsens cognitive performance and behavior in patients with Alzheimer’s disease 128, 129.
It is also possible that the ketogenic diet could ameliorate Alzheimer’s disease by providing greater amounts of essential fatty acids than normal or high carbohydrate diets 130, 128. This is because consumption of foods or artificial supplements rich in essential fatty acids may decrease the risk of developing Alzheimer’s disease 131, 132, 133.
Keto Diet Plan
The ketogenic diet is a high-fat, adequate-protein, low-carbohydrate diet that actually originated as a tool for treating neurological diseases, such as difficult-to-control (refractory) epilepsy in children.
The ketogenic diet forces the body to burn fats rather than carbohydrates. Normally, the carbohydrates contained in food are converted into glucose, which is then transported around the body and is particularly important in fueling brain-function. However, if there is very little carbohydrate in the diet, the liver converts fat into fatty acids and ketone bodies. The ketone bodies pass into the brain and replace glucose as an energy source. An elevated level of ketone bodies in the blood, a state known as ketosis, leads to a reduction in the frequency of epileptic seizures. Over 20 studies show that this type of diet can also help you lose weight and improve health.
There are several reasons why a ketogenic diet is superior to a low-fat diet. One is the increased protein intake, which provides numerous benefits.
The increased ketones, lowered blood sugar levels and improved insulin sensitivity may also play a key role changes in metabolism, high blood sugar and impaired insulin function in pre-diabetes and type 2 diabetes. One study found that the ketogenic diet improved insulin sensitivity by a whopping 75%. The ketogenic diet can help you lose excess fat, which is closely linked to type 2 diabetes, prediabetes and metabolic syndrome.
Why high fat and moderate protein ?
Fats have little to no effect on blood sugar and insulin levels. Protein does affect both blood sugar and insulin. If you eat too much protein for your ideal body weight or lean muscle mass (usually more than 1.5-2.0 grams/kg lean body mass), it can spark gluconeogenesis and raise blood sugar and protein will also spike blood insulin levels temporarily. Higher insulin levels will interfere with ketone body production. In addition, eating a diet that is heavy on lean protein (without enough fat) can make one sick with a condition know as “rabbit starvation”. Rabbit starvation is a form of malnutrition experienced by explorers and native Americans eating exclusively from the land during the Winter season, when food is scarce. It’s caused by a diet restricted to the lean meat of animals such as rabbits, in the absence of carbohydrate and fat.
Other Beneficial Effects on Obesity
It could be argued that the ketogenic diet has beneficial effects other than simply fat and weight loss. For example, researchers recently suggested that ketones may protect from cognitive impairment caused by weight gain and obesity. Moreover there is some evidence that ketogenic diets may have positive effects on mood in overweight subjects. Even if during the very early phase of a ketogenic diet (the first 4 or 5 days) subjects may sometimes complain of lethargy this effect passes rapidly and subjects subsequently report an improved mood.
Although the prevalence of insulin resistance in obesity is not precisely known, it is quite common. Indeed the first demonstration of resistance to insulin stimulation of glucose uptake was obtained in obese subjects. A primary feature of insulin resistance is an impaired ability of muscle cells to take up circulating glucose and also the ability to slow down hepatic glucose output may be compromised. Thus, individuals with insulin resistance have a fundamental problem metabolizing dietary carbohydrate and will divert a greater proportion of dietary carbohydrate to the liver where much of it is converted to fat (i.e., de novo lipogenesis), as opposed to being oxidized for energy in skeletal muscle. Hence the beneficial effects of very low carbohydrate diets in obese subjects are not just a function of weight loss per se but also improved glycaemic control, haemoglobin A1c, and lipid markers, as well as reduced use or withdrawal of insulin and other medications in many cases, occurs before significant weight loss occurs. Moreover, in isocaloric experiments, individuals with insulin resistance showed dramatically improved markers of metabolic syndrome than diets lower in fat.
Is Keto Diet Safe for Obese People ?
In common opinion a low carbohydrate, high protein and fat diet is potentially unhealthy as it may cause a rise in LDL cholesterol and Triglycerides and this issue is of special importance in obese individuals. There are nevertheless several lines of evidence that point to beneficial effects of ketogenic diets on these cardiovascular risk factors. The majority of recent studies seem to amply demonstrate that the reduction of carbohydrates can actually lead to significant benefits in total cholesterol reduction, increases in HDL and reduction of blood triglycerides 25. Furthermore ketogenic diets have been reported to increase the size and volume of LDL-C particles which is considered to reduce cardiovascular disease risk since smaller LDL particles have a higher atherogenicity 134. There is a biochemical rational behind the effects of ketogenic diets on endogenous cholesterol synthesis. A key enyzme in cholesterol biosynthesis is HMGCoA reductase (the target for statins) which is activated by insulin, which means that an increase in blood glucose and consequently of insulin levels will lead to increased endogenous cholesterol synthesis. Thus a reduction in dietary carbohydrate together with a correct intake of cholesterol will lead to a inhibition of cholesterol biosynthesis.
Another point that must be addressed is the effect of ketogenic diet on bone metabolism. There is evidence showing that short-term ketogenic diets impair bone mass density and mechanical properties of bone in mouse. On the other side, it has been suggested recently that the metabolic complications of obesity, such as type 2 diabetes, metabolic syndrome, impaired glucose tolerance, insulin resistance, hyperglycemia, and inflammation may be associated with an increased risk of fracture and poor bone health. Moreover the amount of visceral fat (related to low-grade chronic inflammation) is associated with lower bone mineral density. Whilst there are many studies on the effects of dietary protein levels on bone metabolism in humans, the few investigations on ketogenic diet and bone metabolism were performed on epileptic children. But even in the recently published articles suggested that there is some negative effect on bone health. In any case in human subjects the effects of a very prolonged ketogenic diet are, as yet, not well investigated, for this reason, ketogenic diets may be used safely for a limited period (from 3 weeks to some months) to stimulate fat loss, improve metabolism and help the transition to a correct Mediterranean diet style.
Ketogenic Diet Risks
Regarding the overall safety of ketogenic diet on health there are differences in opinion about the research. Recently, in a recent systematic review based on limited observational studies, Noto and colleagues suggested a possible harmful effect of low carbohydrate/high protein diet (LC/HP) on health: i.e., an increase of all-cause mortality risk whilst there was no effect on cardiovascular disease mortality 135.
On the other hand, for example, a large European study demonstrated that an increase in protein content and a reduction in the glycaemic index led to better maintenance of weight loss without differences regarding adverse effects 136. The existing contradictory evidences on this matter lies in the complex interactions between low-carbohydrate diets and long-term outcomes. Moreover it is important to underline again that a ketogenic diet is not, strictly speaking, a low-carb/high-protein diet; ketogenic diet is mainly a very low carbohydrate diet with high fat and a normal amount of protein that produce a peculiar metabolic state that should not be assimilated to a low-carb/high-protein diet 25.
Some authors claims that a ketogenic diet could affect negatively glucose metabolism 137 but in the cited study the researchers performed the glucose load immediately after the cessation of the ketogenic diet. It is reasonable to suppose that after a period of very low carbohydrate diet there would be an increased glucose sensitively and for this reason is advisable to have a transition phase from ketogenic diet to a normal diet. Another very recently published study demonstrated that a long-term ketogenic diet (22 weeks) caused dyslipidemia, a pro-inflammatory state, signs of hepatic steatosis, glucose intolerance and a reduction in beta and alpha cell mass, without weight loss in mice 138. Two considerations need to be made: the first is that the induction of ketosis and the response to ketosis in man and mouse are quite different and this species metabolic differences could explain why insulin sensitivity is improved in man 139 whilst is decreased in mouse 140 after ketogenic diet. The second is that 22 weeks is a very long period for a mouse that could be compared to several years in human beings. In any case in human subjects the effects of a very prolonged ketogenic diet are, as yet, not well investigated, for this reason, ketogenic diet may be used safely for a limited period (from 3 weeks to some months) to stimulate fat loss, improve metabolism and help the transition to a correct Mediterranean diet style 141. Some authors report that a ketogenic diet induces a severe reduction of IGF-I concentration in rats 142 but, (even if we can compare rat with man) but as stated above a blunting of the IGF-1/AKT/mTOR pathway is not necessary a harmful effect.
Another point that must be addressed is the effect of ketogenic diet on bone metabolism. There is evidence showing that short-term ketogenic diets impair (suggested to be mediated by reductions in circulating IGF-1) bone mass density and mechanical properties of bone in mouse 143; whilst in humans very long term ketogenic diet in children with intractable epilepsy may lead to a progressive reduction of bone mineral content 144. The “caveat” about ketogenic diet and bone is indispensable but we have to take into account that the study of Bielohuby was performed on growing rats for 4 weeks (i.e., a very long time for humans) and thus we have to consider the above mentioned difference between ketosis in the two species, whilst the condition of a life-long ketogenic diet in intractable epilepsy is beyond the scope of this review.
On the other side, it has been suggested recently that the metabolic complications of obesity, such as type 2 diabetes, metabolic syndrome, impaired glucose tolerance, insulin resistance, hyperglycemia, and inflammation may be associated with an increased risk of fracture and poor bone health. Moreover the amount of visceral fat (related to low-grade chronic inflammation) is associated with lower bone mineral density 145. Whilst there are many studies on the effects of dietary protein levels on bone metabolism in humans the few investigations on ketogenic diet and bone metabolism were performed on epileptic children. But even if we assume a higher protein intake during a ketogenic diet (and it is not completely correct as stated above) recently published articles suggested that there is not a negative effect on bone health 146, 147, 148, 149.
Conclusions
A keto diet may provide many health benefits, especially with metabolic, neurological or insulin-related diseases. You should check with your physician if you have any concerns about starting a ketogenic diet plan with pre-existing health conditions, especially if those conditions involve kidney, bone or heart problems. People with kidney disease should definitely consult with their physician about starting a ketogenic diet.
Talk to your doctor about implementing a ketogenic diet if you have any of these conditions:
- History of pancreatitis
- Active gall bladder disease
- Impaired liver function
- Impaired fat digestion
- Poor nutritional status
- Gastric bypass surgery
- Abdominal tumors
- Decreased gastrointestinal motility; this may be in conjunction with conventional cancer treatment and associated drugs
- History of kidney failure
- Pregnancy and lactation
A period of low carbohydrate ketogenic diet may help to control hunger and may improve fat oxidative metabolism and therefore reduce body weight. Furthermore new kinds of ketogenic diets using meals that mimic carbohydrate rich foods could improve the compliance to the diet. Attention should be paid to patient’s renal function and to the transition phase from ketogenic diet to a normal diet that should be gradual and well controlled. The duration of ketogenic diet may range from a minimum (to induce the physiological ketosis) of 2–3 weeks to a maximum (following a general precautionary principle) of many months (6–12 months). Correctly understood the ketogenic diet can be a useful tool to treat obesity in the hands of the physician.
- Conklin HW. Cause and treatment of epilepsy. J Am Osteopath Assoc. 1922;26;11-14[↩]
- Atkins RC. Dr Atkins’ Diet Revolution: The High Calorie Way to Stay Thin Forever. D. McKay Co: New York, NY, USA; 1972.[↩]
- Nutr J. 2015 Apr 22;14:41. doi: 10.1186/s12937-015-0028-x. Acetone as biomarker for ketosis buildup capability–a study in healthy individuals under combined high fat and starvation diets. https://www.ncbi.nlm.nih.gov/pubmed/25897953[↩]
- The Consultant Pharmacist®, Volume 32, Number 7, July 2017, pp. 388-396(9). https://doi.org/10.4140/TCP.n.2017.388. The Ketogenic Diet: Making a Comeback.https://www.ncbi.nlm.nih.gov/pubmed/28701250[↩]
- Prostaglandins Leukot Essent Fatty Acids. 2004 Mar;70(3):309-19. The therapeutic implications of ketone bodies: the effects of ketone bodies in pathological conditions: ketosis, ketogenic diet, redox states, insulin resistance, and mitochondrial metabolism. http://www.plefa.com/article/S0952-3278(03)00221-7/fulltext[↩]
- Prabhakar A, Quach A, Zhang H, et al. Acetone as biomarker for ketosis buildup capability – a study in healthy individuals under combined high fat and starvation diets. Nutrition Journal. 2015;14:41. doi:10.1186/s12937-015-0028-x. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4471925/[↩]
- Sumithran P, Prendergast LA, Delbridge E, et al. Ketosis and appetite-mediating nutrients and hormones after weight loss. Eur J Clin Nutr 2013;67:759–64. https://www.nature.com/articles/ejcn201390[↩][↩]
- Chearskul S, Delbridge E, Shulkes A, Proietto J, Kriketos A. Effect of weight loss and ketosis on postprandial cholecystokinin and free fatty acid concentrations. Am J Clin Nutr 2008;87(5):1238–46.[↩]
- Astrup A, Meinert Larsen T, Harper A. Atkins and other low-carbohydrate diets: hoax or an effective tool for weight loss? Lancet 2004; 364: 897–899.[↩]
- Boden G, Sargrad K, Homko C, Mozzoli M, Stein TP. Effect of a low-carbohydrate diet on appetite, blood glucose levels, and insulin resistance in obese patients with type 2 diabetes. Ann Intern Med 2005; 142: 403–411.[↩]
- Schwartz MW, Woods SC, Porte D Jr., Seeley RJ, Baskin DG. Central nervous system control of food intake. Nature 2000; 404: 661–671.[↩]
- Campfield LA, Smith FJ, Rosenbaum M, Hirsch J. Human eating: evidence for a physiological basis using a modified paradigm. Neurosci Biobehav Rev 1996; 20: 133–137.[↩]
- Obici S, Feng Z, Morgan K, Stein D, Karkanias G, Rossetti L. Central administration of oleic acid inhibits glucose production and food intake. Diabetes 2002; 51: 271–275.[↩]
- Moran TH, Kinzig KP. Gastrointestinal satiety signals II. Cholecystokinin. Am J Physiol Gastrointest Liver Physiol 2004; 286: G183–G188.[↩]
- Leibel RL, Hirsch J. Diminished energy requirements in reduced-obese patients. Metabolism 1984; 33: 164–170.[↩]
- Geldszus R, Mayr B, Horn R, Geisthovel F, von zur Muhlen A, Brabant G. Serum leptin and weight reduction in female obesity. Eur J Endocrinol 1996; 135: 659–662.[↩]
- Cummings DE, Weigle DS, Frayo RS, Breen PA, Ma MK, Dellinger EP et al. Plasma ghrelin levels after diet-induced weight loss or gastric bypass surgery. N Engl J Med 2002; 346: 1623–1630.[↩]
- Chearskul S, Delbridge E, Shulkes A, Proietto J, Kriketos A. Effect of weight loss and ketosis on postprandial cholecystokinin and free fatty acid concentrations. Am J Clin Nutr 2008; 87: 1238–1246.[↩]
- Nordmann AJ, Nordmann A, Briel M, Keller U, Yancy WS Jr., Brehm BJ et al. Effects of low-carbohydrate vs low-fat diets on weight loss and cardiovascular risk factors: a meta-analysis of randomized controlled trials. Arch Intern Med 2006; 166: 285–293[↩]
- Foster GD, Wyatt HR, Hill JO, McGuckin BG, Brill C, Mohammed BS et al. A randomized trial of a low-carbohydrate diet for obesity. N Engl J Med 2003; 348: 2082–2090.[↩]
- Yancy WS Jr., Olsen MK, Guyton JR, Bakst RP, Westman EC. A low-carbohydrate, ketogenic diet versus a low-fat diet to treat obesity and hyperlipidemia: a randomized, controlled trial. Ann Intern Med 2004; 140: 769–777.[↩]
- Breath acetone is a reliable indicator of ketosis in adults consuming ketogenic meals. Musa-Veloso K, Likhodii SS, Cunnane SC. Am J Clin Nutr. 2002 Jul; 76(1):65-70. https://www.ncbi.nlm.nih.gov/pubmed/12081817/[↩][↩]
- C. Colica, G. Merra, A. Gasbarrini, A. De Lorenzo, G. Cioccoloni, P. Gualtieri, M.A. Perrone, S. Bernardini, V. Bernardo, L. Di Renzo, M. Marchetti. Eur Rev Med Pharmacol Sci 2017; 21 (9): 2274-2289. Efficacy and safety of very-low-calorie ketogenic diet: a double blind randomized crossover study. http://www.europeanreview.org/article/12711[↩][↩]
- Caryn Zinn, Matthew Wood, Mikki Williden, Simon Chatterton, and Ed Maunder. J Int Soc Sports Nutr. 2017; 14: 22. Ketogenic diet benefits body composition and well-being but not performance in a pilot case study of New Zealand endurance athletes. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5506682/[↩][↩]
- Paoli A. Ketogenic Diet for Obesity: Friend or Foe? International Journal of Environmental Research and Public Health. 2014;11(2):2092-2107. doi:10.3390/ijerph110202092. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3945587/[↩][↩][↩][↩][↩]
- Very-low-carbohydrate ketogenic diet v. low-fat diet for long-term weight loss: a meta-analysis of randomised controlled trials. Bueno NB, de Melo IS, de Oliveira SL, da Rocha Ataide T. Br J Nutr. 2013 Oct; 110(7):1178-87. https://www.ncbi.nlm.nih.gov/pubmed/23651522/[↩]
- Therapeutic role of low-carbohydrate ketogenic diet in diabetes. Al-Khalifa A, Mathew TC, Al-Zaid NS, Mathew E, Dashti HM. Nutrition. 2009 Nov-Dec; 25(11-12):1177-85. https://www.ncbi.nlm.nih.gov/pubmed/19818281/[↩]
- Beneficial effects of ketogenic diet in obese diabetic subjects. Dashti HM, Mathew TC, Khadada M, Al-Mousawi M, Talib H, Asfar SK, Behbahani AI, Al-Zaid NS. Mol Cell Biochem. 2007 Aug; 302(1-2):249-56. https://www.ncbi.nlm.nih.gov/pubmed/17447017/[↩]
- A ketogenic diet favorably affects serum biomarkers for cardiovascular disease in normal-weight men. Sharman MJ, Kraemer WJ, Love DM, Avery NG, Gómez AL, Scheett TP, Volek JS. J Nutr. 2002 Jul; 132(7):1879-85. https://www.ncbi.nlm.nih.gov/pubmed/12097663/[↩]
- Popular diets: a scientific review. Freedman MR, King J, Kennedy E. Obes Res. 2001 Mar; 9 Suppl 1():1S-40S. https://www.ncbi.nlm.nih.gov/pubmed/11374180/[↩]
- The regulation of the release of ketone bodies by the liver. Krebs HA. Adv Enzyme Regul. 1966; 4():339-54. https://www.ncbi.nlm.nih.gov/pubmed/4865971/[↩]
- Brain metabolism during fasting. Owen OE, Morgan AP, Kemp HG, Sullivan JM, Herrera MG, Cahill GF Jr. J Clin Invest. 1967 Oct; 46(10):1589-95. https://www.ncbi.nlm.nih.gov/pubmed/6061736/[↩][↩]
- Amino acid metabolism during prolonged starvation. Felig P, Owen OE, Wahren J, Cahill GF Jr. J Clin Invest. 1969 Mar; 48(3):584-94. https://www.ncbi.nlm.nih.gov/pubmed/5773094/[↩]
- Owen O.E. Ketone bodies as a fuel for the brain during starvation. Biochem. Mol. Biol. Educ. 2005;33:246–251. doi: 10.1002/bmb.2005.49403304246.[↩]
- Liver and kidney metabolism during prolonged starvation. Owen OE, Felig P, Morgan AP, Wahren J, Cahill GF Jr. J Clin Invest. 1969 Mar; 48(3):574-83. https://www.ncbi.nlm.nih.gov/pubmed/5773093/[↩]
- Pathways and control of ketone body metabolism: on the fringe of lipid biochemistry. Fukao T, Lopaschuk GD, Mitchell GA. Prostaglandins Leukot Essent Fatty Acids. 2004 Mar; 70(3):243-51. https://www.ncbi.nlm.nih.gov/pubmed/14769483/[↩][↩]
- Paoli A. Ketogenic Diet for Obesity: Friend or Foe? International Journal of Environmental Research and Public Health. 2014;11(2):2092-2107. doi:10.3390/ijerph110202092. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3945587/ [↩]
- Paoli A, Rubini A, Volek JS, Grimaldi KA. Beyond weight loss: a review of the therapeutic uses of very-low-carbohydrate (ketogenic) diets. European Journal of Clinical Nutrition. 2013;67(8):789-796. doi:10.1038/ejcn.2013.116. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3826507/[↩][↩][↩]
- Targeting energy metabolism in brain cancer: review and hypothesis. Seyfried TN, Mukherjee P. Nutr Metab (Lond). 2005 Oct 21; 2:30. https://www.ncbi.nlm.nih.gov/pubmed/16242042/[↩]
- Lipolysis and gluconeogenesis from glycerol during weight reduction with very-low-calorie diets. Vazquez JA, Kazi U. Metabolism. 1994 Oct; 43(10):1293-9. https://www.ncbi.nlm.nih.gov/pubmed/7934983/[↩][↩]
- Gluconeogenesis and energy expenditure after a high-protein, carbohydrate-free diet. Veldhorst MA, Westerterp-Plantenga MS, Westerterp KR. Am J Clin Nutr. 2009 Sep; 90(3):519-26. https://www.ncbi.nlm.nih.gov/pubmed/19640952/[↩]
- Glycerol turnover and oxidation in man. Bortz WM, Paul P, Haff AC, Holmes WL. J Clin Invest. 1972 Jun; 51(6):1537-46. https://www.ncbi.nlm.nih.gov/pubmed/5024045/[↩]
- Role of beta-hydroxybutyric acid in the central regulation of energy balance. Laeger T, Metges CC, Kuhla B. Appetite. 2010 Jun; 54(3):450-5. https://www.ncbi.nlm.nih.gov/pubmed/20416348/[↩]
- The therapeutic implications of ketone bodies: the effects of ketone bodies in pathological conditions: ketosis, ketogenic diet, redox states, insulin resistance, and mitochondrial metabolism. Veech RL. Prostaglandins Leukot Essent Fatty Acids. 2004 Mar; 70(3):309-19. https://www.ncbi.nlm.nih.gov/pubmed/14769489/[↩][↩]
- Diet-induced ketosis increases monocarboxylate transporter (MCT1) levels in rat brain. Leino RL, Gerhart DZ, Duelli R, Enerson BE, Drewes LR. Neurochem Int. 2001 May; 38(6):519-27. https://www.ncbi.nlm.nih.gov/pubmed/11248400/[↩]
- Fernstrom JD, Fernstrom MH. Nutrition and the brain. In: Gibney MJ, Macdonald IA, Roche HM, editor. Nutrition & Metabolism. Oxford, UK: Blackwell Science; 2003. pp. 145–167.[↩]
- Manninen AH. Metabolic Effects of the Very-Low-Carbohydrate Diets: Misunderstood “Villains” of Human Metabolism. Journal of the International Society of Sports Nutrition. 2004;1(2):7-11. doi:10.1186/1550-2783-1-2-7. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2129159/[↩][↩]
- Ketone bodies, potential therapeutic uses. Veech RL, Chance B, Kashiwaya Y, Lardy HA, Cahill GF Jr. IUBMB Life. 2001 Apr; 51(4):241-7. https://www.ncbi.nlm.nih.gov/pubmed/11569918/[↩]
- Point-of-care blood ketone testing: screening for diabetic ketoacidosis at the emergency department. Charles RA, Bee YM, Eng PH, Goh SY. Singapore Med J. 2007 Nov; 48(11):986-9. https://www.ncbi.nlm.nih.gov/pubmed/17975686/[↩]
- Frayn K. Metabolic Regulation: A Human Perspective (3rd Edition) United Kingdom: Wiley-Blackwell; 2010.[↩]
- Brehm BJ, Seeley RJ, Daniels SR, D’Alessio DA. A randomized trial comparing a very low carbohydrate diet and a calorie-restricted low fat diet on body weight and cardiovascular risk factors in healthy women. J Clin Endocrinol Metabol. 2003;88:1617–23. doi: 10.1210/jc.2002-021480. https://www.ncbi.nlm.nih.gov/pubmed/12679447[↩]
- Bueno NB, De Melo ISV, De Oliveira SL, Da Rocha Ataide T. Very-low-carbohydrate ketogenic diet v. low-fat diet for long-term weight loss: A meta-analysis of randomised controlled trials. Br J Nutr. 2013;110:1178–87. doi: 10.1017/S0007114513000548. https://www.ncbi.nlm.nih.gov/pubmed/23651522[↩]
- Paoli A, Rubini A, Volek J, Grimaldi K. Beyond weight loss: a review of the therapeutic uses of very-low-carbohydrate (ketogenic) diets. Eur J Clin Nutr. 2013;67:789–96. doi: 10.1038/ejcn.2013.116. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3826507/[↩]
- Vining EP. The ketogenic diet. Adv Exp Med Biol. 2002;497:225–31. doi: 10.1007/978-1-4615-1335-3_20. https://www.ncbi.nlm.nih.gov/pubmed/11993735[↩]
- Garrow JS. Energy balance and obesity in man. Amsterdam/London: North-Holland Publishing Company; 1974.[↩]
- Keene DL. A systematic review of the use of the ketogenic diet in childhood epilepsy. Pediatr Neurol. 2006;35:1–5. doi: 10.1016/j.pediatrneurol.2006.01.005. https://www.ncbi.nlm.nih.gov/pubmed/16814077[↩]
- Mitchell G, Kassovska-Bratinova S, Boukaftane Y, Robert M, Wang S, Ashmarina L, et al. Medical aspects of ketone body metabolism. Clin Invest Med. 1995;18:193–216. https://www.ncbi.nlm.nih.gov/pubmed/7554586[↩]
- Mattson MP, Wan R. Beneficial effects of intermittent fasting and caloric restriction on the cardiovascular and cerebrovascular systems. J Nutr Biochem. 2005;16:129–37. doi: 10.1016/j.jnutbio.2004.12.007. https://www.ncbi.nlm.nih.gov/pubmed/15741046[↩]
- Johnson JB, Summer W, Cutler RG, Martin B, Hyun D-H, Dixit VD, et al. Alternate day calorie restriction improves clinical findings and reduces markers of oxidative stress and inflammation in overweight adults with moderate asthma. Free Radic Biol Med. 2007;42:665–74. doi: 10.1016/j.freeradbiomed.2006.12.005. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1859864/[↩]
- Yancy WS, Olsen MK, Guyton JR, Bakst RP, Westman EC. A Low-carbohydrate, ketogenic diet versus a Low-Fat diet to treat obesity and HyperlipidemiaA randomized. Controlled trial. Ann Intern Med. 2004;140:769–77. doi: 10.7326/0003-4819-140-10-200405180-00006. https://www.ncbi.nlm.nih.gov/pubmed/15148063[↩]
- Paoli A, Grimaldi K, Toniolo L, Canato M, Bianco A, Fratter A. Nutrition and acne: therapeutic potential of ketogenic diets. Skin Pharmacol Physiol. 2012;25:111–117. https://www.ncbi.nlm.nih.gov/pubmed/22327146[↩][↩][↩]
- Krebs HA. The regulation of the release of ketone bodies by the liver. Adv Enzyme Regul. 1966;4:339–354. https://www.ncbi.nlm.nih.gov/pubmed/4865971[↩]
- Paoli A, Canato M, Toniolo L, Bargossi AM, Neri M, Mediati M, et al. The ketogenic diet: an underappreciated therapeutic option. Clin Ter. 2011;162:e145–e153. https://www.ncbi.nlm.nih.gov/pubmed/22041813[↩]
- Cahill GF., Jr Fuel metabolism in starvation. Annu Rev Nutr. 2006;26:1–22. https://www.ncbi.nlm.nih.gov/pubmed/16848698[↩][↩]
- Beyond weight loss: a review of the therapeutic uses of very-low-carbohydrate (ketogenic) diets. Paoli A, Rubini A, Volek JS, Grimaldi KA. Eur J Clin Nutr. 2013 Aug; 67(8):789-96. https://www.ncbi.nlm.nih.gov/pubmed/23801097/[↩]
- Merck Sharp & Dohme Corp., Merck Manual. Diabetic Ketoacidosis (DKA). https://www.merckmanuals.com/professional/endocrine-and-metabolic-disorders/diabetes-mellitus-and-disorders-of-carbohydrate-metabolism/diabetic-ketoacidosis-dka[↩]
- Breath acetone predicts plasma ketone bodies in children with epilepsy on a ketogenic diet. Musa-Veloso K, Likhodii SS, Rarama E, Benoit S, Liu YM, Chartrand D, Curtis R, Carmant L, Lortie A, Comeau FJ, Cunnane SC. Nutrition. 2006 Jan; 22(1):1-8. https://www.ncbi.nlm.nih.gov/pubmed/16183255/[↩]
- Breath ketone testing: a new biomarker for diagnosis and therapeutic monitoring of diabetic ketosis. Qiao Y, Gao Z, Liu Y, Cheng Y, Yu M, Zhao L, Duan Y, Liu Y. Biomed Res Int. 2014;2014:869186. https://www.ncbi.nlm.nih.gov/pubmed/24900994/[↩]
- Biomed Res Int. 2014;2014:869186. doi: 10.1155/2014/869186. Epub 2014 May 11. Breath ketone testing: a new biomarker for diagnosis and therapeutic monitoring of diabetic ketosis. https://www.ncbi.nlm.nih.gov/pubmed/24900994/[↩]
- Post-exercise ketosis and the hormone response to exercise: a review. Koeslag JH. Med Sci Sports Exerc. 1982; 14(5):327-34. https://www.ncbi.nlm.nih.gov/pubmed/6759842/[↩]
- Sasaki H, Ishikawa S, Ueda H, KIMURA Y. Response of acetone in expired Air during graded and prolonged exercise. Adv Exerc Sports Physiol. 2011;16:97–100.[↩]
- Westerterp-Plantenga MS, Nieuwenhuizen A, Tome D, Soenen S, Westerterp KR. Dietary protein, weight loss, and weight maintenance. Annu Rev Nutr. 2009;29:21–41. https://www.ncbi.nlm.nih.gov/pubmed/19400750[↩][↩][↩][↩]
- Feinman RD, Fine EJ. Nonequilibrium thermodynamics and energy efficiency in weight loss diets. Theor Biol Med Model. 2007;4:27. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1947950/[↩][↩][↩]
- Freedman MR, King J, Kennedy E. Popular diets: A scientific review. Obes Res. 2001;9 (Suppl 1:1S–40S. https://www.ncbi.nlm.nih.gov/pubmed/11374180[↩]
- Johnston BC, Kanters S, Bandayrel K, et al. Comparison of weight loss among named diet programs in overweight and obese adults: a network meta-analysis. JAMA. doi:10.1001/jama.2014.10397[↩]
- Brehm BJ, Seeley RJ, Daniels SR, D’Alessio DA. A randomized trial comparing a very low carbohydrate diet and a calorie-restricted low fat diet on body weight and cardiovascular risk factors in healthy women. J Clin Endocrinol Metab. 2003;88:1617–1623. https://www.ncbi.nlm.nih.gov/pubmed/12679447[↩][↩]
- Gardner CD, Kiazand A, Alhassan S, Kim S, Stafford RS, Balise RR, et al. Comparison of the atkins, zone, ornish, and LEARN diets for change in weight and related risk factors among overweight premenopausal women: The A TO Z weight loss study: a randomized trial. JAMA. 2007;297:969–977. https://www.ncbi.nlm.nih.gov/pubmed/17341711[↩]
- Shai I, Schwarzfuchs D, Henkin Y, Shahar DR, Witkow S, Greenberg I, et al. Weight loss with a low-carbohydrate, mediterranean, or low-fat diet. N Engl J Med. 2008;359:229–241. https://www.ncbi.nlm.nih.gov/pubmed/18635428[↩][↩]
- Fine EJ, Feinman RD. Thermodynamics of weight loss diets. Nutr Metab (Lond) 2004;1:15. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC543577/[↩][↩][↩]
- Halton TL, Hu FB. The effects of high protein diets on thermogenesis, satiety and weight loss: a critical review. J Am Coll Nutr. 2004;23:373–385. https://www.ncbi.nlm.nih.gov/pubmed/15466943[↩]
- Veldhorst MA, Westerterp-Plantenga MS, Westerterp KR. Gluconeogenesis and energy expenditure after a high-protein, carbohydrate-free diet. Am J Clin Nutr. 2009;90:519–526. https://www.ncbi.nlm.nih.gov/pubmed/19640952[↩]
- Paoli A, Grimaldi K, Bianco A, Lodi A, Cenci L, Parmagnani A. Medium term effects of a ketogenic diet and a mediterranean diet on resting energy expenditure and respiratory ratio BMC Proceedings 20126(Suppl 3): P37[↩][↩]
- Sumithran P, Prendergast LA, Delbridge E, Purcell K, Shulkes A, Kriketos A, et al. Ketosis and appetite-mediating nutrients and hormones after weight loss Eur J Clin Nutr 2013;. e-pub ahead of print 1 May 2013; doi:10.1038/ejcn.2013.90. https://www.ncbi.nlm.nih.gov/pubmed/23632752[↩][↩][↩]
- Veldhorst M, Smeets A, Soenen S, Hochstenbach-Waelen A, Hursel R, Diepvens K, et al. Protein-induced satiety: effects and mechanisms of different proteins. Physiol Behav. 2008;94:300–307. https://www.ncbi.nlm.nih.gov/pubmed/18282589[↩]
- Johnstone AM, Horgan GW, Murison SD, Bremner DM, Lobley GE. Effects of a high-protein ketogenic diet on hunger, appetite, and weight loss in obese men feeding ad libitum. Am J Clin Nutr. 2008;87:44–55. https://www.ncbi.nlm.nih.gov/pubmed/18175736[↩]
- Veldhorst MA, Westerterp-Plantenga MS, Westerterp KR. Gluconeogenesis and energy expenditure after a high-protein, carbohydrate-free diet. Am J Clin Nutr. 2009;90:519–526. https://www.ncbi.nlm.nih.gov/pubmed/19640952[↩]
- Paoli A, Cenci L, Fancelli M, Parmagnani A, Fratter A, Cucchi A, et al. Ketogenic diet and phytoextracts comparison of the efficacy of mediterranean, zone and tisanoreica diet on some health risk factors. Agro Food Ind Hi-Tech. 2010;21:24.[↩][↩][↩]
- The effects of low-carbohydrate versus conventional weight loss diets in severely obese adults: one-year follow-up of a randomized trial. Stern L, Iqbal N, Seshadri P, Chicano KL, Daily DA, McGrory J, Williams M, Gracely EJ, Samaha FF. Ann Intern Med. 2004 May 18; 140(10):778-85. https://www.ncbi.nlm.nih.gov/pubmed/15148064/[↩]
- A low-carbohydrate as compared with a low-fat diet in severe obesity. Samaha FF, Iqbal N, Seshadri P, Chicano KL, Daily DA, McGrory J, Williams T, Williams M, Gracely EJ, Stern L. N Engl J Med. 2003 May 22; 348(21):2074-81. https://www.ncbi.nlm.nih.gov/pubmed/12761364/[↩][↩]
- Effect of a high-protein, low-carbohydrate diet on blood glucose control in people with type 2 diabetes. Gannon MC, Nuttall FQ. Diabetes. 2004 Sep; 53(9):2375-82. https://www.ncbi.nlm.nih.gov/pubmed/15331548/[↩][↩]
- Effect of a low-carbohydrate diet on appetite, blood glucose levels, and insulin resistance in obese patients with type 2 diabetes. Boden G, Sargrad K, Homko C, Mozzoli M, Stein TP. Ann Intern Med. 2005 Mar 15; 142(6):403-11. https://www.ncbi.nlm.nih.gov/pubmed/15767618/[↩][↩][↩]
- Lasting improvement of hyperglycaemia and bodyweight: low-carbohydrate diet in type 2 diabetes. A brief report. Nielsen JV, Jönsson E, Nilsson AK. Ups J Med Sci. 2005; 110(2):179-83. https://www.ncbi.nlm.nih.gov/pubmed/16075898/[↩][↩][↩]
- Nitrogen metabolism and insulin requirements in obese diabetic adults on a protein-sparing modified fast. Bistrian BR, Blackburn GL, Flatt JP, Sizer J, Scrimshaw NS, Sherman M. Diabetes. 1976 Jun; 25(6):494-504. https://www.ncbi.nlm.nih.gov/pubmed/1278601/[↩]
- Gumbiner B, Wendel JA, McDermott MP. Effects of diet composition and ketosis on glycemia during very-low-energy-diet therapy in obese patients with non-insulin-dependent diabetes mellitus. Am J Clin Nutr. 1996;63:110–115. https://www.ncbi.nlm.nih.gov/pubmed/8604657[↩]
- Boden G, Sargrad K, Homko C, Mozzoli M, Stein TP. Effect of a low-carbohydrate diet on appetite, blood glucose levels, and insulin resistance in obese patients with type 2 diabetes. Ann Intern Med. 2005;142:403–411. https://www.ncbi.nlm.nih.gov/pubmed/15767618[↩]
- Dashti HM, Al-Zaid NS, Mathew TC, Al-Mousawi M, Talib H, Asfar SK, et al. Long term effects of ketogenic diet in obese subjects with high cholesterol level. Mol Cell Biochem. 2006;286:1–9. https://www.ncbi.nlm.nih.gov/pubmed/16652223[↩]
- Yancy WS, Jr, Foy M, Chalecki AM, Vernon MC, Westman EC. A low-carbohydrate, ketogenic diet to treat type 2 diabetes. Nutr Metab (Lond) 2005;2:34. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1325029/[↩]
- Nielsen JV, Joensson EA. Low-carbohydrate diet in type 2 diabetes: Stable improvement of bodyweight and glycemic control during 44 months follow-up. Nutr Metab (Lond) 2008;5:14. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2424054/[↩]
- Volek JS, Phinney SD, Forsythe CE, Quann EE, Wood RJ, Puglisi MJ, et al. Carbohydrate restriction has a more favorable impact on the metabolic syndrome than a low fat diet. Lipids. 2009;44:297–309. https://www.ncbi.nlm.nih.gov/pubmed/19082851[↩][↩][↩]
- Basu S, Yoffe P, Hills N, Lustig RH. The relationship of sugar to population-level diabetes prevalence: An econometric analysis of repeated cross-sectional data. PLoS One. 2013;8:e57873. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3584048/[↩]
- Yancy WS, Foy M, Chalecki AM, Vernon MC, Westman EC. A low-carbohydrate, ketogenic diet to treat type 2 diabetes. Nutrition & Metabolism. 2005;2:34. doi:10.1186/1743-7075-2-34.[↩][↩]
- A low-carbohydrate, ketogenic diet versus a low-fat diet to treat obesity and hyperlipidemia: a randomized, controlled trial. Yancy WS Jr, Olsen MK, Guyton JR, Bakst RP, Westman EC. Ann Intern Med. 2004 May 18; 140(10):769-77. https://www.ncbi.nlm.nih.gov/pubmed/15148063/[↩]
- Nordmann AJ, Nordmann A, Briel M, Keller U, Yancy WS, Jr, Brehm BJ, et al. Effects of low-carbohydrate vs low-fat diets on weight loss and cardiovascular risk factors: a meta-analysis of randomized controlled trials. Arch Intern Med. 2006;166:285–293. https://www.ncbi.nlm.nih.gov/pubmed/16476868[↩]
- Blackburn GL, Phillips JC, Morreale S. Physician’s guide to popular low-carbohydrate weight-loss diets Cleve Clin J Med 200168761–766.768–9, 773–4. https://www.ncbi.nlm.nih.gov/pubmed/11563479[↩]
- Sharman MJ, Kraemer WJ, Love DM, Avery NG, Gomez AL, Scheett TP, et al. A ketogenic diet favorably affects serum biomarkers for cardiovascular disease in normal-weight men. J Nutr. 2002;132:1879–1885. https://www.ncbi.nlm.nih.gov/pubmed/12097663[↩][↩][↩]
- Volek JS, Sharman MJ, Forsythe CE. Modification of lipoproteins by very low-carbohydrate diets. J Nutr. 2005;135:1339–1342. https://www.ncbi.nlm.nih.gov/pubmed/15930434[↩][↩]
- Fauser BC, Tarlatzis BC, Rebar RW, Legro RS, Balen AH, Lobo R, et al. Consensus on women’s health aspects of polycystic ovary syndrome (PCOS): the amsterdam ESHRE/ASRM-sponsored 3rd PCOS consensus workshop group Fertil Steril 20129728–38.e25. https://www.ncbi.nlm.nih.gov/pubmed/22153789[↩]
- DeUgarte CM, Bartolucci AA, Azziz R. Prevalence of insulin resistance in the polycystic ovary syndrome using the homeostasis model assessment. Fertil Steril. 2005;83:1454–1460. https://www.ncbi.nlm.nih.gov/pubmed/15866584[↩]
- Tosi F, Negri C, Perrone F, Dorizzi R, Castello R, Bonora E, et al. Hyperinsulinemia amplifies GnRH agonist stimulated ovarian steroid secretion in women with polycystic ovary syndrome. J Clin Endocrinol Metab. 2012;97:1712–1719. https://www.ncbi.nlm.nih.gov/pubmed/22419715[↩]
- Blank SK, McCartney CR, Chhabra S, Helm KD, Eagleson CA, Chang RJ, et al. Modulation of gonadotropin-releasing hormone pulse generator sensitivity to progesterone inhibition in hyperandrogenic adolescent girls–implications for regulation of pubertal maturation. J Clin Endocrinol Metab. 2009;94:2360–2366. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2708962/[↩]
- Moghetti P, Castello R, Negri C, Tosi F, Spiazzi GG, Brun E, et al. Insulin infusion amplifies 17 alpha-hydroxycorticosteroid intermediates response to adrenocorticotropin in hyperandrogenic women: apparent relative impairment of 17,20-lyase activity. J Clin Endocrinol Metab. 1996;81:881–886. https://www.ncbi.nlm.nih.gov/pubmed/8772544[↩]
- Mavropoulos JC, Yancy WS, Hepburn J, Westman EC. The effects of a low-carbohydrate, ketogenic diet on the polycystic ovary syndrome: a pilot study. Nutr Metab (Lond) 2005;2:35. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1334192/[↩]
- Cordain L, Eaton SB, Sebastian A, Mann N, Lindeberg S, Watkins BA, et al. Origins and evolution of the western diet: health implications for the 21st century. Am J Clin Nutr. 2005;81:341–354. https://www.ncbi.nlm.nih.gov/pubmed/15699220[↩]
- Smith RN, Mann NJ, Braue A, Makelainen H, Varigos GA. The effect of a high-protein, low glycemic-load diet versus a conventional, high glycemic-load diet on biochemical parameters associated with acne vulgaris: A randomized, investigator-masked, controlled trial. J Am Acad Dermatol. 2007;57:247–256. https://www.ncbi.nlm.nih.gov/pubmed/17448569[↩][↩]
- Smith R, Mann N. Acne in adolescence: a role for nutrition. Nutr Diet. 2007;64:S147–S149.[↩]
- Cordain L. Implications for the role of diet in acne. Semin Cutan Med Surg. 2005;24:84–91. https://www.ncbi.nlm.nih.gov/pubmed/16092796[↩]
- Kristiansen SB, Endoh A, Casson PR, Buster JE, Hornsby PJ. Induction of steroidogenic enzyme genes by insulin and IGF-I in cultured adult human adrenocortical cells. Steroids. 1997;62:258–265. https://www.ncbi.nlm.nih.gov/pubmed/9055386[↩]
- Powell DR, Suwanichkul A, Cubbage ML, DePaolis LA, Snuggs MB, Lee PD. Insulin inhibits transcription of the human gene for insulin-like growth factor-binding protein-1. J Biol Chem. 1991;266:18868–18876. https://www.ncbi.nlm.nih.gov/pubmed/1717456[↩]
- The efficacy of the ketogenic diet-1998: a prospective evaluation of intervention in 150 children. Freeman JM, Vining EP, Pillas DJ, Pyzik PL, Casey JC, Kelly LM. Pediatrics. 1998 Dec; 102(6):1358-63. https://www.ncbi.nlm.nih.gov/pubmed/9832569/[↩]
- The ketogenic diet: a 3- to 6-year follow-up of 150 children enrolled prospectively. Hemingway C, Freeman JM, Pillas DJ, Pyzik PL. Pediatrics. 2001 Oct; 108(4):898-905. https://www.ncbi.nlm.nih.gov/pubmed/11581442/[↩]
- The outcome of children with intractable seizures: a 3- to 6-year follow-up of 67 children who remained on the ketogenic diet less than one year. Marsh EB, Freeman JM, Kossoff EH, Vining EP, Rubenstein JE, Pyzik PL, Hemingway C. Epilepsia. 2006 Feb; 47(2):425-30. https://www.ncbi.nlm.nih.gov/pubmed/16499771/[↩]
- Current evidence indicates that antiepileptic drugs are anti-ictal, not antiepileptic. Schachter SC. Epilepsy Res. 2002 Jun; 50(1-2):67-70. https://www.ncbi.nlm.nih.gov/pubmed/12151118/[↩]
- Prevention of epilepsy after head trauma: do we need new drugs or a new approach? Benardo LS. Epilepsia. 2003; 44 Suppl 10():27-33. https://www.ncbi.nlm.nih.gov/pubmed/14511392/[↩]
- Treatment of Parkinson disease with diet-induced hyperketonemia: a feasibility study. Vanitallie TB, Nonas C, Di Rocco A, Boyar K, Hyams K, Heymsfield SB. Neurology. 2005 Feb 22; 64(4):728-30. https://www.ncbi.nlm.nih.gov/pubmed/15728303/[↩]
- Dietary fatty acids and the risk of Parkinson disease: the Rotterdam study. de Lau LM, Bornebroek M, Witteman JC, Hofman A, Koudstaal PJ, Breteler MM. Neurology. 2005 Jun 28; 64(12):2040-5. https://www.ncbi.nlm.nih.gov/pubmed/15985568/[↩]
- Effects of beta-hydroxybutyrate on cognition in memory-impaired adults. Reger MA, Henderson ST, Hale C, Cholerton B, Baker LD, Watson GS, Hyde K, Chapman D, Craft S. Neurobiol Aging. 2004 Mar; 25(3):311-4. https://www.ncbi.nlm.nih.gov/pubmed/15123336/[↩]
- Ketonemia and seizures: metabolic and anticonvulsant effects of two ketogenic diets in childhood epilepsy. Huttenlocher PR. Pediatr Res. 1976 May; 10(5):536-40. https://www.ncbi.nlm.nih.gov/pubmed/934725/[↩]
- High carbohydrate diets and Alzheimer’s disease. Henderson ST. Med Hypotheses. 2004; 62(5):689-700. https://www.ncbi.nlm.nih.gov/pubmed/15082091/[↩][↩]
- A randomized, crossover trial of high-carbohydrate foods in nursing home residents with Alzheimer’s disease: associations among intervention response, body mass index, and behavioral and cognitive function. Young KW, Greenwood CE, van Reekum R, Binns MA. J Gerontol A Biol Sci Med Sci. 2005 Aug; 60(8):1039-45. https://www.ncbi.nlm.nih.gov/pubmed/16127110/[↩]
- Potential role of polyunsaturates in seizure protection achieved with the ketogenic diet. Cunnane SC, Musa K, Ryan MA, Whiting S, Fraser DD. Prostaglandins Leukot Essent Fatty Acids. 2002 Aug-Sep; 67(2-3):131-5. https://www.ncbi.nlm.nih.gov/pubmed/12324231/[↩]
- Prognosis of Alzheimer’s disease: the Rotterdam Study. Ruitenberg A, Kalmijn S, de Ridder MA, Redekop WK, van Harskamp F, Hofman A, Launer LJ, Breteler MM. Neuroepidemiology. 2001 Aug; 20(3):188-95. https://www.ncbi.nlm.nih.gov/pubmed/11490165/[↩]
- Fish, meat, and risk of dementia: cohort study. Barberger-Gateau P, Letenneur L, Deschamps V, Pérès K, Dartigues JF, Renaud S. BMJ. 2002 Oct 26; 325(7370):932-3. https://www.ncbi.nlm.nih.gov/pubmed/12399342/[↩]
- Dietary fats and the risk of incident Alzheimer disease. Morris MC, Evans DA, Bienias JL, Tangney CC, Bennett DA, Aggarwal N, Schneider J, Wilson RS. Arch Neurol. 2003 Feb; 60(2):194-200. https://www.ncbi.nlm.nih.gov/pubmed/12580703/[↩]
- Modification of lipoproteins by very low-carbohydrate diets. Volek JS, Sharman MJ, Forsythe CE. J Nutr. 2005 Jun; 135(6):1339-42. https://www.ncbi.nlm.nih.gov/pubmed/15930434/[↩]
- Low-carbohydrate diets and all-cause mortality: a systematic review and meta-analysis of observational studies. Noto H, Goto A, Tsujimoto T, Noda M. PLoS One. 2013; 8(1):e55030. https://www.ncbi.nlm.nih.gov/pubmed/23372809/[↩]
- Diets with high or low protein content and glycemic index for weight-loss maintenance. Larsen TM, Dalskov SM, van Baak M, Jebb SA, Papadaki A, Pfeiffer AF, Martinez JA, Handjieva-Darlenska T, Kunešová M, Pihlsgård M, Stender S, Holst C, Saris WH, Astrup A, Diet, Obesity, and Genes (Diogenes) Project. N Engl J Med. 2010 Nov 25; 363(22):2102-13. https://www.ncbi.nlm.nih.gov/pubmed/21105792/[↩]
- Impaired glucose tolerance in rats fed low-carbohydrate, high-fat diets. Bielohuby M, Sisley S, Sandoval D, Herbach N, Zengin A, Fischereder M, Menhofer D, Stoehr BJ, Stemmer K, Wanke R, Tschöp MH, Seeley RJ, Bidlingmaier M. Am J Physiol Endocrinol Metab. 2013 Nov 1; 305(9):E1059-70. https://www.ncbi.nlm.nih.gov/pubmed/23982154/[↩]
- Long-term ketogenic diet causes glucose intolerance and reduced β- and α-cell mass but no weight loss in mice. Ellenbroek JH, van Dijck L, Töns HA, Rabelink TJ, Carlotti F, Ballieux BE, de Koning EJ. Am J Physiol Endocrinol Metab. 2014 Mar 1; 306(5):E552-8. https://www.ncbi.nlm.nih.gov/pubmed/24398402/[↩]
- Carbohydrate restriction has a more favorable impact on the metabolic syndrome than a low fat diet. Volek JS, Phinney SD, Forsythe CE, Quann EE, Wood RJ, Puglisi MJ, Kraemer WJ, Bibus DM, Fernandez ML, Feinman RD. Lipids. 2009 Apr; 44(4):297-309. https://www.ncbi.nlm.nih.gov/pubmed/19082851/[↩]
- Induction of ketosis in rats fed low-carbohydrate, high-fat diets depends on the relative abundance of dietary fat and protein. Bielohuby M, Menhofer D, Kirchner H, Stoehr BJ, Müller TD, Stock P, Hempel M, Stemmer K, Pfluger PT, Kienzle E, Christ B, Tschöp MH, Bidlingmaier M. Am J Physiol Endocrinol Metab. 2011 Jan; 300(1):E65-76. https://www.ncbi.nlm.nih.gov/pubmed/20943751/[↩]
- Long term successful weight loss with a combination biphasic ketogenic Mediterranean diet and Mediterranean diet maintenance protocol. Paoli A, Bianco A, Grimaldi KA, Lodi A, Bosco G. Nutrients. 2013 Dec 18; 5(12):5205-17. https://www.ncbi.nlm.nih.gov/pubmed/24352095/[↩]
- Lack of dietary carbohydrates induces hepatic growth hormone (GH) resistance in rats. Bielohuby M, Sawitzky M, Stoehr BJ, Stock P, Menhofer D, Ebensing S, Bjerre M, Frystyk J, Binder G, Strasburger C, Wu Z, Christ B, Hoeflich A, Bidlingmaier M. Endocrinology. 2011 May; 152(5):1948-60. https://www.ncbi.nlm.nih.gov/pubmed/21427215/[↩]
- Short-term exposure to low-carbohydrate, high-fat diets induces low bone mineral density and reduces bone formation in rats. Bielohuby M, Matsuura M, Herbach N, Kienzle E, Slawik M, Hoeflich A, Bidlingmaier M. J Bone Miner Res. 2010 Feb; 25(2):275-84. https://www.ncbi.nlm.nih.gov/pubmed/19653818/[↩]
- Progressive bone mineral content loss in children with intractable epilepsy treated with the ketogenic diet. Bergqvist AG, Schall JI, Stallings VA, Zemel BS. Am J Clin Nutr. 2008 Dec; 88(6):1678-84. https://www.ncbi.nlm.nih.gov/pubmed/19064531/[↩]
- Divergent effects of obesity on bone health. Gower BA, Casazza K. J Clin Densitom. 2013 Oct-Dec; 16(4):450-4. https://www.ncbi.nlm.nih.gov/pubmed/24063845/[↩]
- Effects of a high protein diet on body weight and comorbidities associated with obesity. Clifton P. Br J Nutr. 2012 Aug; 108 Suppl 2():S122-9. https://www.ncbi.nlm.nih.gov/pubmed/23107523/[↩]
- Diet-induced weight loss: the effect of dietary protein on bone. Tang M, O’Connor LE, Campbell WW. J Acad Nutr Diet. 2014 Jan; 114(1):72-85. https://www.ncbi.nlm.nih.gov/pubmed/24183993/[↩]
- The effect of a low-carbohydrate diet on bone turnover. Carter JD, Vasey FB, Valeriano J. Osteoporos Int. 2006; 17(9):1398-403. https://www.ncbi.nlm.nih.gov/pubmed/16718399/[↩]
- Effect of protein intake on bone mineralization during weight loss: a 6-month trial. Skov AR, Haulrik N, Toubro S, Mølgaard C, Astrup A. Obes Res. 2002 Jun; 10(6):432-8. https://www.ncbi.nlm.nih.gov/pubmed/12055318/[↩]