How much water should you drink a day ?
Water plays a large part in your normal body functions, drinking enough water is essential for physiological processes such as circulation, metabolism, temperature regulation, and waste removal 1. Water is the main constituent of cells, tissues and organs and is vital for life 2. Water as a vital nutrient, a multifunctional constituent of the human body. Every day you lose water through your breath, perspiration, urine and bowel movements. For your body to function properly, you must replenish its water supply by consuming beverages and foods that contain water.
Water makes up at least two-thirds of the human body 3. About 60% of our body weight is made of water 4. This water content varies with body composition (lean and fat mass) 5. In infants and young children, water as a percentage of body weight is higher than in adults. This is mainly due to higher water content in the extracellular compartment, whereas the water content in the intracellular compartment is lower in infants than in older children and adults 4. Body composition changes rapidly during the first year of life, with a decrease in the water content of the fat-free mass and an increase in the content of protein and minerals 4.
In adults, about two-thirds of total water is in the intracellular space, whereas one-third is extracellular water 4. A 70kg human has about 42 liters of total body water, of which 28 liters is intracellular water and 14 liters is extracellular fluid (ECF) 6. Of the latter, 3 liters is in blood plasma, 1 liter is the transcellular fluid (cerebrospinal fluid, ocular, pleural, peritoneal and synovial fluids) and 10 liters is the interstitial fluid, including lymph, which provides an aqueous medium surrounding cells 4.
The constancy of the amount and composition of extracellular fluid (ECF) is a necessity for the function of cells. This constancy is due to the homeostatic mechanisms that monitor and regulate its composition, osmotic pressure, pH and temperature 7. These mechanisms rely on the function of the main systems of the body, such as the circulatory, respiratory, renal and alimentary systems. The monitoring and regulation of these systems are coordinated by the nervous and endocrine systems. The composition of the intracellular fluid is maintained by solute movement across the cell membrane by passive or active transports 7.
Under usual conditions of moderate ambient temperature (18–20 °C) and with a moderate activity level, body water remains relatively constant. This implies a precise regulation of water balance: over a 24-hour period, intake and loss of water must be equal. It has been estimated that water balance is regulated within 0.2% of body weight over a 24-h period 8.
Water Inputs
Water inputs are composed of three major sources (Table 1): the water you drink, the water you eat and the water you produce. The water you drink is essentially composed of water and other liquids with a high water content (85 to >90%). The water you eat comes from various foods with a wide range of water content (40 to >80%). The water you produce results from the oxidation of macronutrients (endogenous or metabolic water).
Table 1. Water balance in sedentary adults living in temperate climate
| Water inputs (ml/day) | Water outputs (ml/day) | ||||||
|---|---|---|---|---|---|---|---|
| Min | Max | Average | Min | Max | Average | ||
| Beverages | 1400a | 1750a | 1575 | Urine | 1200 | 2000 | 1600 |
| Foodsb | 600a | 750a | 675 | Skin | 450 | 450 | 450c |
| Subtotal | 2000d | 2500e | 2250 | Respiration | 250c | 350c | 300 |
| Metabolic water | 250 | 350 | 300 | Faeces | 100 | 300 | 200c |
| Total | 2250 | 2850 | 2550 | Total | 2000 | 3100 | 2550 |
a It is normally assumed that the contribution of food to total dietary water intake is 20–30%, whereas 70–80% are provided by beverages. This relationship is not fixed and depends on the type of beverages and on the choice of foods.
b Foods with a wide range of water content (<40 to >80%).
c (EFSA, 2008) 9.
d Average total water intakes in sedentary women (EFSA, 2008) 9.
e Average total water intakes in sedentary men (EFSA, 2008) 9.
It is normally assumed that the contribution of food to total water intake is 20–30%, whereas 70–80% is provided by beverages. This relationship is not fixed and depends on the type of beverages and on the choice of foods 9.
For an individual at rest under temperate conditions, the volume that might be drunk in a day is on an average 1.5 liters. This has to be adapted according to age, gender, climate and physical activity. The water content of food can vary within a wide range, and consequently the amount of water contributed by foods can vary between 500 ml and 1 liter a day. Endogenous or metabolic water represents about 250–350 ml a day in sedentary people.
The adequate total water intakes for sedentary adults are on an average between 2 and 2.5 liters per day (women and men, respectively) 9. In conclusion, the total water inputs for sedentary adults are on an average between 2 and 3 liters.
So how much fluid does the average, healthy adult living in a temperate climate need ?
Owing to the precise mechanisms regulating water balance, normal hydration is compatible with a wide range of fluid intake 10. Human water requirements are not based on a minimal intake as it might lead to a water deficit because of numerous factors that modify water needs (metabolism, climate, physical activity, diet and so on). Instead, water needs are based on experimentally derived intake levels that are expected to meet nutritional adequacy for members of a healthy population; this is the adequate intake determined for infants, adolescents, adults and elderly individuals 11. Numerous factors, such as high ambient temperature and humidity levels, physical activity and exercise, and heat stress in particular, influence water needs. Thus, the adequate water intake determined for standard conditions does not meet these particular requirements and adequate intake must be increased in relation to these conditions.
According to the Institute of Medicine, values for adequate intake (AI) of water coming from liquids are the following (see Table 2):
- 0.7 l/day for infants 0–6 months old, 0.6/l day for infants 7–12 months old,
- 0.9 l/day for children 1–3 years old, and 1.2 l/day for children 4–8 years old,
- Male adolescents aged 9–13 years need 1.8 l l/day, those aged 14–18 years need 2.6 l/day and
- Male adults need 3.0 l/day.
- Female adolescents aged 9–13 years need 1.6 l/day, those aged 14–18 years need 1.8 l/day and female adults need 2.2 l/day.
Table 2. Dietary reference intake values for total water in the United States (Institute of Medicine of the National Academies, Washington DC)
| Life stage group | Criterion | AI for males in l/day a | AI for females in l/day a | ||||
|---|---|---|---|---|---|---|---|
| From foods | From beverages | Total water b | From foods | From beverages | Total water b | ||
| 0–6 months | Average consumption of water from human milk | 0 | 0.7 | 0.7 | 0 | 0.7 | 0.7 |
| 7–12 months | Average consumption of water from human milk and complementary foods | 0.2 | 0.6 | 0.8 | 0.2 | 0.6 | 0.8 |
| 1–3 years | Median total water intake from NHANES III | 0.4 | 0.9 | 1.3 | 0.4 | 0.9 | 1.3 |
| 4–8 years | Median total water intake from NHANES III | 0.5 | 1.2 | 1.7 | 0.5 | 1.2 | 1.7 |
| 9–13 years | Median total water intake from NHANES III | 0.6 | 1.8 | 2.4 | 0.5 | 1.6 | 2.1 |
| 14–18 years | Median total water intake from NHANES III | 0.7 | 2.6 | 3.3 | 0.5 | 1.8 | 2.3 |
| >19 years | Median total water intake from NHANES III | 0.7 | 3.0 | 3.7 | 0.5 | 2.2 | 2.7 |
| Pregnancy 14–50 years | Median total water intake from NHANES III | 0.7 | 2.3 | 3.0 | |||
| Lactation 14–50 years | Median total water intake from NHANES III | 0.7 | 3.1 | 3.8 | |||
Abbreviations: AI, adequate intake; NHANES III, Third National Health and Nutrition Examination Survey.
a The AI is not equivalent to the recommended dietary allowances, RDA.
b Total water represents drinking water, other beverages and water from food.
[Source 12]What you eat also provides a significant portion of your fluid needs. On average, food provides about 20 percent of total water intake. For example, many fruits and vegetables, such as watermelon and spinach, are 90 percent or more water by weight. In general, though, drinking too much water resulting in low sodium levels in the blood, a condition called hyponatremia, is rare in healthy adults who eat an average American diet.
Hyponatremia: It’s possible to become overhydrated while exercising. This is caused by low sodium (salt) levels in the blood. It can occur if too much water is drunk over a short period of time. Hyponatremia sometimes affects athletes whose blood sodium level is reduced through sweat and then diluted by drinking large amounts of water. Symptoms of hyponatremia include nausea, vomiting and headache. In serious cases, the brain can swell, causing confusion, seizures, coma and, in rare cases, death.
Remember that sugary drinks due to their very high added sugar content can lead to weight gain and inflammation, which can increase your risk for developing diseases such as diabetes.
The European Food Safety Authority has been recently asked to revise the existing recommended intakes of essential substances with a physiological effect, including water, as this nutrient is essential for health and life. The values of water requirements mentioned in Table 4 are in agreement with its draft recommendations 9.
Table 3. Dietary reference intake values for total water in Europe
| Life stage group | Adequate intake of water for males (ml/day) | Adequate intake of water for females (ml/day) | ||||
|---|---|---|---|---|---|---|
| From foods a | From beverages b | Total water | From foods a | From beverages b | Total water | |
| 2–3 years | 390 | 910 | 1300 | 390 | 910 | 1300 |
| 4–8 years | 480 | 1120 | 1600 | 480 | 1120 | 1600 |
| 9–13 years | 630 | 1470 | 2100 | 570 | 1330 | 1900 |
| >14 years | 750 | 1750 | 2500 | 600 | 1400 | 2000 |
| Pregnancy | 690 | 1610 | 2300c | |||
| Lactation | 600 | 2100 | 2700d | |||
a Foods with a wide range of water content (<40 to >80%).
b It is normally assumed that the contribution of food to total dietary water intake is 20–30%, whereas 70–80% are provided by beverages. This relationship is not fixed and depends on the type of beverages and on the choice of foods.
c There are no European data available, but assuming an increase of energy intake of 15% (equivalent to 300 kcal/day), an additional total water intake of 300 ml would be adequate 9.
d Adequate water intakes for lactating women are about 700 ml/day above the adequate intakes of nonlactating women of the same age 9.
[Source 9]When we compare the dietary reference intakes (DRIs) (also called dietary reference values or DRVs), for water that have been published in the United States 12 with those proposed in discussion in Europe 9, the main differences are with regard to children aged 9–13 years and adults. This confirms a well-known fact that the establishment of dietary reference intakes is conditioned by the dietary habits of the population (food and beverage habits). This is the reason why it is necessary to have these recommendations set up by continent or by country when data are available.
In acute exercise, sweat losses can reach 1–2 liters/hour and this magnitude of fluid losses can be difficult to replace in the short term 13, 14. Long recovery periods of ad libitum (as desired) drinking are needed to recover water balance with adequate electrolyte replacement 15.
Despite varying water needs, healthy humans regulate their daily water balance with precision. However, young infants and elderly people are at a greater risk of dehydration than are adults. Parents and caregivers should be aware of signs of dehydration in infants and elderly individuals and they should encourage water intake of individuals who are at risk of dehydration.
Water Outputs
The main routes of water loss from the body are kidneys, skin and the respiratory tract and, at a very low level, the digestive system (Table 1).
Over a 24-h period, a sedentary adult produces 1–2 liters of urine.
Water is lost by evaporation through the skin; this is called insensible perspiration because it is an invisible water loss and it represents about 450 ml of water per day in a temperate environment.
Water is also lost by evaporation through the respiratory tract (250–350 ml per day).
Finally, a sedentary adult loses about 200 ml of water a day through faeces.
On an average, a sedentary adult loses 2–3 l of water per day. These water losses through the skin and lungs depend on the climate, air temperature and relative humidity.
When the internal body temperature rises, the only mechanism for increasing heat losses is the activation of sweat glands. Evaporation of water by way of sweat on the skin surface is a very efficient mechanism for removing heat from the body: 2.2 kJ is lost by the evaporation of 1 g of water. When exercising in a hot environment, the sweating rate can reach as much as 1–2 liters of water loss per hour 16. This can lead to dehydration and hyperosmolarity of extracellular fluid (ECF).
It is important to note that sweat is always hypotonic when compared with plasma or extracellular fluid (ECF). Sweat contains 20–50 mmol/l of Na+, whereas the extracellular Na+ concentration is 150 mmol/liter. Intense sweating therefore leads to greater water than electrolyte losses 16. The consequence is an increased extracellular osmolarity that draws water from cells into the ECF. Thus, the loss of water through sweating concerns both intracellular fluid and ECF, a situation that characterizes hypertonic dehydration. The need to drink hypotonic drinks during endurance exercise is well established. A person losing 4 liters of sweat with no fluid replacement loses about 10% of body water, but only 4% of extracellular sodium (about 120 mmol of Na+). This indicates that during exercise, fluid replacement is more important than salt replacement.
Dehydration and hyperosmolarity of extracellular fluid (ECF) can affect consciousness and are involved in the occurrence of heat stroke when internal temperature rises above critical thresholds. The latter can occur when exercising in a warm and humid environment 16.
Regulation of water balance
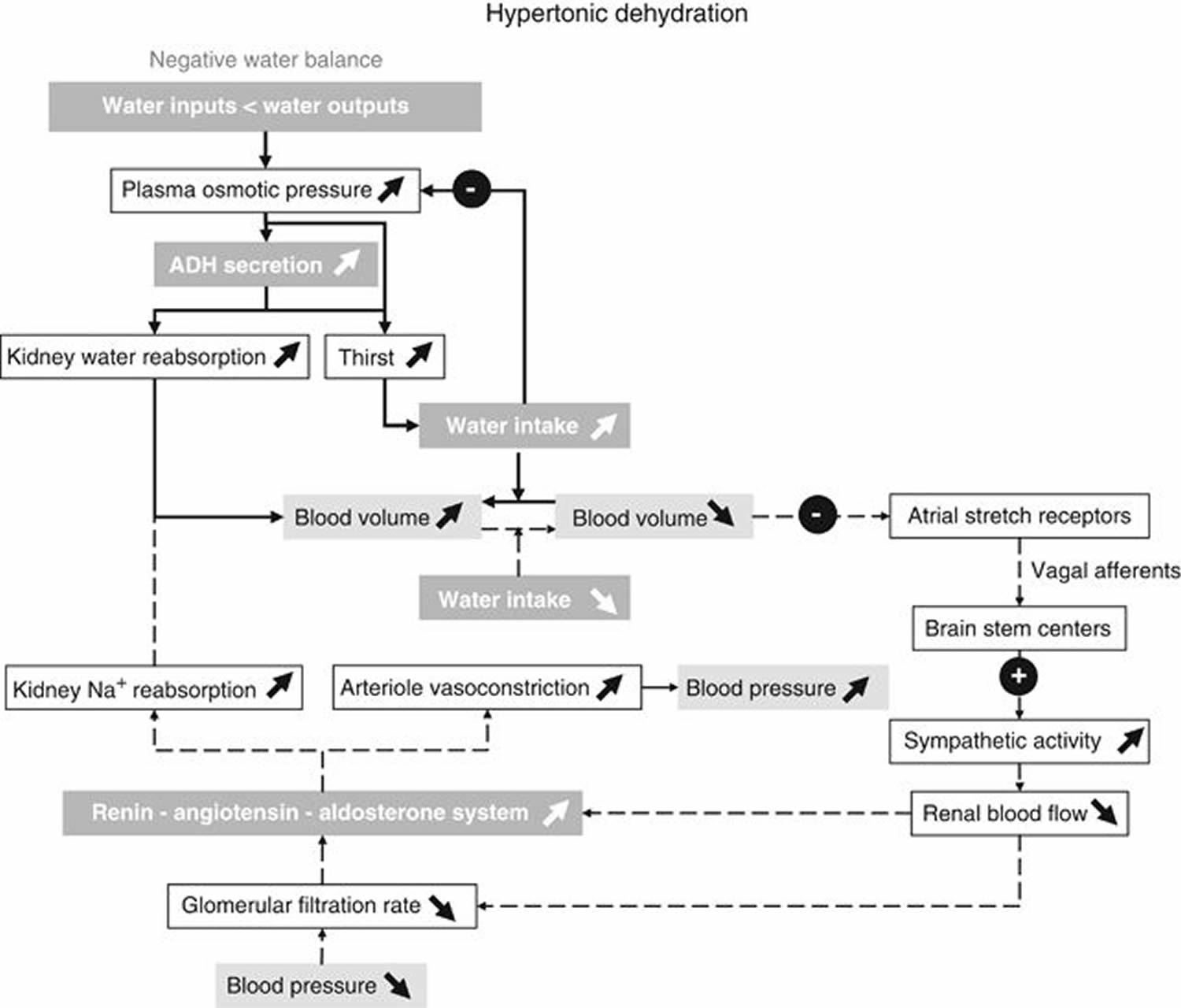
The intake of water is partially determined by thirst. When water losses exceed water intake, the osmotic pressure of extracellular fluid (ECF) increases. By activation of hypothalamic osmoreceptors, an antidiuretic hormone (ADH) is released from the posterior pituitary gland 7. Both the increased ECF osmotic pressure and antidiuretic hormone (ADH) elicit the feeling of thirst (Figure 1). The receptors that elicit thirst have an osmotic threshold higher than the osmoreceptors involved in ADH release. Thus, ADH can act on the kidneys to increase water reabsorption before thirst is elicited. Thirst is often blunted in elderly subjects who are at risk of having an insufficient water intake in conditions of elevated ambient temperature and humidity 17. Thirst is triggered by an increase in plasma and ECF osmolarity, by reductions in plasma volume at water deficits that correspond to a body weight loss of 1–3% 9. During rehydration, thirst can disappear before water balance is reached.
Figure 1. Water balance regulation in your body
[Source 4]Note: Feedback loops for water balance: main perturbations and physiological responses to hypertonic dehydration due to a negative water balance. Solid arrows show the responses induced by osmoreceptors when plasma osmotic pressure increases. Dashed arrows show the corrective mechanisms induced by insufficient water intake and a decreased blood volume to restore blood volume and blood pressure. Note that in the case of hypotonic dehydration due to a positive water balance, all perturbations and physiological responses that are induced occur in the reverse direction.
The set point of plasma osmolarity above which antidiuretic hormone (ADH) secretion is stimulated is about 280 mosm/l. Furthermore, the sensitivity of antidiuretic hormone (ADH) response to a rise in plasma osmolarity is enhanced when the circulating blood volume is lowered 7.
Kidneys are the main regulators of water losses. They have the unique property to modify the osmotic pressure of urine within a large range in response to minute changes in plasma osmotic pressure.
There are two conditions that induce the production of a large volume of urine, and therefore a large water loss. Water diuresis occurs when water is ingested in excess of body requirements. This leads to a small decrease in plasma osmolarity, with a suppression of ADH secretion. As a result, a large volume of hypotonic urine is produced. In contrast, osmotic diuresis results from a filtered load of a solute that exceeds the renal tubules’ maximum reabsorption capacity for this solute.
In summary, both deficit and excess water intakes are counterbalanced by subtle hormonal changes (ADH, aldosterone and atrial natriuretic peptide) that contribute to buffer the deleterious effects of these abnormal conditions. In the end, the final and precise regulation of water balance is dependent on thirst and on ADH release, with its predominant role in water reabsorption in the kidneys (Figure 1). Therefore, voluntary drinking of water is a key behavior for maintaining water balance. Consequently, drinking water before being thirsty is a good habit for maintaining a good body hydration status.

How you use water to keep every system in your body functioning properly
- carrying nutrients and oxygen to your cells
- flushing bacteria from your bladder
- aiding digestion
- preventing constipation
- normalizing blood pressure
- stabilizing the heartbeat
- cushioning joints
- protecting organs and tissues
- regulating body temperature
- maintaining electrolyte (sodium) balance.
Water as a building material
Water, present in each cell of your body and in the various tissues and compartments, acts first as a building material 1. This primary function leads to nutritional recommendations, as water needs are higher during the growth period of the body.
Water as a solvent, a reaction medium, a reactant and a reaction product
Water has unique properties: it is an excellent solvent for ionic compounds and for solutes such as glucose and amino acids 18. It is a highly interactive molecule and acts by weakening electrostatic forces and hydrogen bonding between other polar molecules. It has a high dielectric constant and it forms oriented solvent shells around ions, thus enabling them to move freely. Water as a macronutrient is involved in all hydrolytic reactions, for instance, in the hydrolysis of other macronutrients (proteins, carbohydrates, lipids and so on).
Water is also produced by the oxidative metabolism of hydrogen-containing substrates in the body. Theoretically, for 1 g of glucose, palmitic acid and protein (albumin), 0.6, 1.12 and 0.37 ml water, respectively, is endogenously produced, or for 100 kcal of energy, 15, 13 and 9 ml water is produced.
Water as a carrier
Water is essential for cellular homeostasis because it transports nutrients to cells and removes wastes from cells 18. It is the medium in which all transport systems function, allowing exchanges between cells, interstitial fluid and capillaries 19. Water maintains the vascular volume and allows blood circulation, which is essential for the function of all organs and tissues of the body 20. Thus, the cardiovascular and respiratory systems, the digestive tract, the reproductive system, the kidney and liver, the brain and the peripheral nervous system, all depend on adequate hydration to function effectively 18. Severe dehydration therefore affects the function of many systems and is a life-threatening condition 21.
Water and thermoregulation
Water has a large heat capacity, which contributes to limiting changes in body temperature in a warm or cold environment. Water has a large capacity for vaporization of heat, which allows a loss of heat from the body even when ambient temperature is higher than body temperature 13. When sweating is elicited, evaporation of water from the skin surface is a very efficient way to lose heat.
Water as a lubricant and shock absorber
Water, in combination with viscous molecules, forms lubricating fluids for joints; for saliva, gastric and intestinal mucus secretion in the digestive tract; for mucus in airways secretion in the respiratory system and for mucus secretion in the genito-urinary tract.
By maintaining the cellular shape, water also acts as a shock absorber during walking or running. This function is important for the brain and spinal cord, and is particularly important for the fetus, who is protected by a water cushion.
Giving your body enough fluids to carry out those tasks means that you’re staying hydrated.
If you don’t drink enough water, you risk becoming dehydrated. Warning signs of dehydration include weakness, low blood pressure, dizziness, confusion, or urine that’s dark in color.
How much water should you drink each day ? It’s a simple question with no easy answers. Studies have produced varying recommendations over the years, but in truth, your water needs depend on many factors, including your health, how active you are and where you live.
Although no single formula fits everyone, knowing more about your body’s need for fluids will help you estimate how much water to drink each day.
Factors that influence your water needs
You may need to modify your total fluid intake depending on how active you are, the climate you live in, your health status, and if you’re pregnant or breast-feeding.
- Exercise. If you exercise or engage in any activity that makes you sweat, you need to drink extra water to compensate for the fluid loss. An extra 1.5 to 2.5 cups (400 to 600 milliliters) of water should suffice for short bouts of exercise, but intense exercise lasting more than an hour (for example, running a marathon) requires more fluid intake. How much additional fluid you need depends on how much you sweat during exercise, and the duration and type of exercise.
- Intense exercise. During long bouts of intense exercise, it’s best to use a sports drink that contains sodium, as this will help replace sodium lost in sweat and reduce the chances of developing hyponatremia, which can be life-threatening. Also, continue to replace fluids after you’re finished exercising.
- Environment. Hot or humid weather can make you sweat and requires additional intake of fluid. Heated indoor air also can cause your skin to lose moisture during wintertime. Further, altitudes greater than 8,200 feet (2,500 meters) may trigger increased urination and more rapid breathing, which use up more of your fluid reserves.
- Illnesses or health conditions. When you have fever, vomiting or diarrhea, your body loses additional fluids. In these cases, you should drink more water. In some cases, your doctor may recommend oral rehydration solutions, such as Gatorade, Powerade or CeraLyte. You may also need increased fluid intake if you develop certain conditions, including bladder infections or urinary tract stones. On the other hand, some conditions, such as heart failure and some types of kidney, liver and adrenal diseases, may impair excretion of water and even require that you limit your fluid intake.
- Pregnancy or breast-feeding. Women who are pregnant or breast-feeding need additional fluids to stay hydrated. Large amounts of fluid are used especially when nursing. The Institute of Medicine recommends that pregnant women drink about 10 cups (2.3 liters) of fluids daily and women who breast-feed consume about 13 cups (3.1 liters ) of fluids a day.
If you think you are not getting enough water, these tips may help:
- Carry a water bottle for easy access when you are at work of running errands.
- Freeze some freezer safe water bottles. Take one with you for ice-cold water all day long.
- Choose water instead of sugar-sweetened beverages. This can also help with weight management. Substituting water for one 20-ounce sugar sweetened soda will save you about 240 calories. For example, during the school day students should have access to drinking water, giving them a healthy alternative to sugar-sweetened beverages.
- Choose water when eating out. Generally, you will save money and reduce calories.
- Add a wedge of lime or lemon to your water. This can help improve the taste and help you drink more water than you usually do.
Summary Daily Water Requirements
Water, a vital nutrient, has numerous critical roles in the human body. It acts as a building material; as a solvent, reaction medium, reactant and reaction product; as a carrier for nutrients and waste products; in thermoregulation and as a lubricant and shock absorber. Consequently, the optimal functioning of our body requires a good hydration level. Healthy humans regulate daily water balance remarkably well across their lifespan despite changes in biological development and exposure to stressors on hydration status.
Acute or chronic body water deficits result when intakes are reduced or losses increase, but day-to-day hydration is generally well maintained so long as food and fluid are readily available. Total water intake includes drinking water, water in beverages, and water in food. Daily water needs determined from fluid balance, water turnover, or consumption studies provide similar values for a given set of conditions.
A better estimate of daily water requirements has been established owing to the knowledge of the main physiological functions of water and the understanding of the mechanisms of water balance regulation. On an average, a sedentary adult female should drink 2.2 – 2.7 liters and adult male 3.0 – 3.7 liters of water per day, as water is the only liquid nutrient that is really essential for body hydration and is vital for the body to function properly. However, strenuous physical exercise and heat stress can greatly increase daily water needs, and the individual variability between athletes can be substantial.
The big benefits of drinking plain water
A recent study in the Journal of Human Nutrition and Dietetics provides even more evidence that we should choose water over other drinks if we want to control our weight.
The researchers found that the participants who drank the most plain water in their daily diet consumed fewer total calories, drank fewer sweetened beverages, and took in less total fat, saturated fat, sugar, salt, and cholesterol. In fact, they discovered that increasing plain water consumption by one to three cups a day could decrease calorie intake by 68 to 205 calories a day. That could add up to a lot fewer calories over time and result in significant weight loss.
These results support prior research on this topic, which has shown that drinking water before meals and that substituting water for sweetened beverages can cut down on calorie intake and improve weight control.
The CDC 22, USDA 23, American Medical Association 24, American Diabetes Association 25, American Heart Association 26 and American Academy of Pediatrics 27 recommend drinking water, either instead of other beverages or in greater volume, for weight management 28). Public health initiatives in the U.S., U.K., Australia, France, Belgium, Spain, Greece, Poland, Mexico, Israel and Taiwan are actively promoting drinking water for weight management 29, 30, 22, 23, 24, 25, 26, 27.
That means people interested in losing weight and improving their overall health could benefit from incorporating more plain water into their daily diet.
So next time you’re thirsty, instead of ignoring your kitchen sink at home or the water cooler at work, take a moment to savor the one drink that’s free, refreshing, and actually good for you: plain old water.
How much water should I drink to lose weight ?
A literature review 28 was conducted to explore the different effects of drinking water on energy intake, energy expenditure, fat oxidation and body weight. The conclusion from the review of available randomised controlled trials reported negative, null and positive effects of drinking water on energy intake, energy expenditure, fat oxidation and weight change. One 12-week randomised controlled trial 31, reports that obese adults randomly assigned to drink 500 mL water 30 min before their main meals lost significantly more weight than the control group, assigned to imagine their stomach was full before meals. The results are consistent with the weight loss effects of increasing drinking water by more than 1 L/day for overweight or obese individuals with a restricted, hypocaloric or low glycemic diet 32, 33, 34.
What is Water ?
Water is a clear, odorless, tasteless, colorless liquid that freezes into ice below 0 degrees centigrade and boils above 100 degrees centigrade. Water is essential for most animal and plant life and is an excellent solvent for many substances. The chemical formula is hydrogen oxide (H2O) 35. Water appears colorless to the naked eye in small quantities, though it is actually slightly blue in color 36. It covers 71% of Earth’s surface. Current estimates suggest that there are 1. 4 billion cubic kilometers (330 million m3) of it available on Earth, and it exists in many forms. Water appears mostly in the oceans (saltwater) and polar ice caps, but it is also present as clouds, rain water, rivers, freshwater aquifers, lakes, and sea ice. Water in these bodies perpetually moves through a cycle of evaporation, precipitation, and runoff to the sea.
Clean water is essential to human life. In many parts of the world, it is in short supply. From a biological standpoint, water has many distinct properties that are critical for the proliferation of life that set it apart from other substances. It carries out this role by allowing organic compounds to react in ways that ultimately allow replication. All known forms of life depend on water. Water is vital both as a solvent in which many of the body’s solutes dissolve and as an essential part of many metabolic processes within your body. Metabolism is the sum total of anabolism and catabolism. In anabolism, water is removed from molecules (through energy requiring enzymatic chemical reactions) in order to grow larger molecules (e. g. starches, triglycerides and proteins for storage of fuels and information). In catabolism, water is used to break bonds in order to generate smaller molecules (e. g. glucose, fatty acids and amino acids to be used for fuels for energy use or other purposes). Water is thus essential and central to these metabolic processes. Water is also central to photosynthesis and respiration. Photosynthetic cells use the sun’s energy to split off water’s hydrogen from oxygen. Hydrogen is combined with CO2 (absorbed from air or water) to form glucose and release oxygen. All living cells use such fuels and oxidize the hydrogen and carbon to capture the sun’s energy and reform water and CO2 in the process (cellular respiration). Water is also central to acid-base neutrality and enzyme function. An acid, a hydrogen ion (H+, that is, a proton) donor, can be neutralized by a base, a proton acceptor such as hydroxide ion (OH-) to form water. Water is considered to be neutral, with a pH (the negative log of the hydrogen ion concentration) of 7. Acids have pH values less than 7 while bases have values greater than 7. Stomach acid (HCl) is useful to digestion. However, its corrosive effect on the esophagus during reflux can temporarily be neutralized by ingestion of a base such as aluminum hydroxide to produce the neutral molecules water and the salt aluminum chloride. Human biochemistry that involves enzymes usually performs optimally around a biologically neutral pH of 7. 4 37.
Water is essential to good health and it’s important for your body to have plenty of water each day, yet needs vary by individual.
The average human is composed of between 55 and 60% water. Water helps you digest food, absorb nutrients from food, and then get rid of the unused waste. Water is found in foods—both solids and liquids, as well as in its natural state.
Essentially every system in your body depends on water. For example, water flushes toxins out of vital organs, carries nutrients to your cells, and provides a moist environment for ear, nose and throat tissues.
Lack of water can lead to dehydration, a condition that occurs when you don’t have enough water in your body to carry out normal functions. Even mild dehydration can drain your energy and make you tired.
Drinking Water
Drinking water comes from a variety of sources including public water systems, private wells, or bottled water 38. Ensuring safe and healthy drinking water may be as simple as turning on the tap from an Environmental Protection Agency-regulated public water system 39. Other water sources may need a water filter, a check on water fluoridation, or an inspection to ensure a septic tank is not too close to a private well. It is important to know where drinking water comes from, how it’s been treated, and if it’s safe to drink 38.
The EPA is responsible for the nation’s drinking water regulation. To learn more, visit the EPA’s Public Drinking Water Systems page 39.
Public Water Systems
The United States is fortunate to have one of the safest public drinking water supplies in the world 40.
- Providing safe drinking water is a partnership that involves EPA, the states, tribes, water systems, and water system operators. The public drinking water systems regulated by EPA and delegated states and tribes provide drinking water to 90 percent of Americans 39.
There are over 151,000 public water systems in the United States. The United States Environmental Protection Agency (EPA) 39 classifies these water systems according to the number of people they serve, the source of their water, and whether they serve the same customers year-round or on an occasional basis.
EPA has defined three types of public water systems 39:
According to EPA, public drinking water systems consist of community and non-community systems.
- A community water system supplies water to the same population year-round. It serves at least 25 people at their primary residences or at least 15 residences that are primary residences (for example, municipalities, mobile home park, sub-divisions).
- Non-community water systems are composed of transient and non-transient water systems.
1) Transient non-community water systems provide water to 25 or more people for at least 60 days/year, but not to the same people and not on a regular basis (for example, gas stations, campgrounds).
2) Non-transient non-community water systems regularly supply water to at least 25 of the same people at least six month per year, but not year-round (for example, schools, factories, office buildings, and hospitals which have their own water systems).
Public Water Systems Fast Facts
- Of the approximately 155,693 public water systems in the United States, 52,110 (33.5%) are community systems and 103,583 (66.5%) are noncommunity systems, including 84,744 transient systems and 18,839 nontransient systems 41.
- Over 286 million Americans get their tap water from a community water system 41.
- 8% of U.S. community water systems provide water to 82% of the U.S. population through large municipal water systems 41.
- Although the majority of community water systems (78%) are supplied by ground water, more people (68%) are supplied year-round by community water systems that use surface water 41.
As these systems age, deterioration can occur due to corrosion, materials erosion, and external pressures. Deteriorating water distribution systems can lead to:
- Breaches in pipes and storage facilities
- Intrusion due to water pressure fluctuation
- Main breaks
Every community water supplier must provide an annual report, sometimes called a Consumer Confidence Report, or “CCR,” to its customers. The report provides information on your local drinking water quality, including the water’s source, contaminants found in the water, and how consumers can get involved in protecting drinking water.
- View the CDC’s guide to Understanding Consumer Confidence Reports 42.
- See if your CCR is posted online (United States Environmental Protection Agency Local Drinking Water Information) 43.
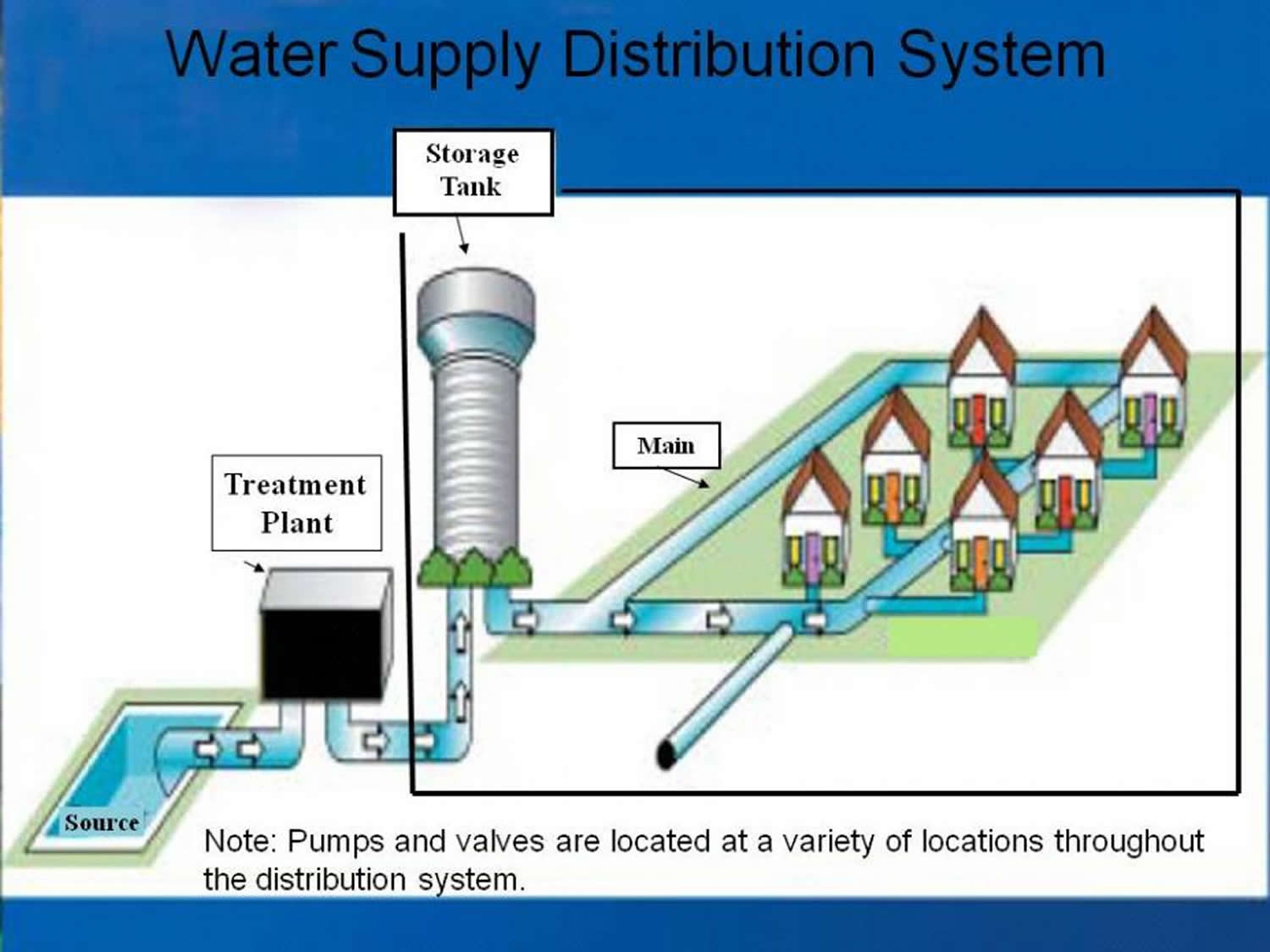
Drinking Water Distribution Systems
Water distribution systems consist of an interconnected series of components. They include:
- pipes
- storage facilities
- components that convey drinking water
Water distribution systems meet fire protection needs for:
- Cities, homes, schools, hospitals, businesses, industries, other facilities.
Public water systems depend on distribution systems to provide an uninterrupted supply of pressurized safe drinking water to all consumers. Distribution system mains carry water from either:
- the treatment plant to the consumer; or
- the source to the consumer when treatment is absent.
Distribution systems span almost one million miles in the United States. They represent the vast majority of physical infrastructure for water supplies. Distribution system wear and tear can pose intermittent or persistent health risks.
Water Quality and Testing
- Water Quality
The United States has one of the safest water supplies in the world. If you are among the 286 million Americans that get their water from a community water system (1), your tap water is regulated by the United States Environmental Protection Agency (EPA).
Drinking water varies from place to place, depending on the condition of the source water from which it is drawn and the treatment it receives, but it must meet EPA regulations.
Even though our tap water supplies are considered to be one of the safest in the world, water contamination can still occur. There are many sources of contamination, including:
- Sewage releases
- Naturally occurring chemicals and minerals (for example, arsenic, radon, uranium)
Local land use practices (for example, fertilizers, pesticides, livestock, concentrated feeding operations) - Manufacturing processes (for example, heavy metals, cyanide)
- Malfunctioning on-site wastewater treatment systems (for example, septic systems)
In addition, drinking water that is not properly treated or which travels through an improperly maintained distribution system (for example, the piping system) may also create an environment for contamination.
The presence of certain contaminants in our water can lead to health issues, including gastrointestinal illness, reproductive problems, and neurological disorders. Infants, young children, pregnant women, the elderly, and immunocompromised persons may be especially susceptible to illness.
- Water Testing
The EPA sets standards and regulations for the presence and levels of over 90 different contaminants in public drinking water, including E.coli, Salmonella, Cryptosporidium, metals such as lead, and disinfection by-products. For more information on these contaminants and maximum contaminant levels, please visit EPA’s Drinking Water Contaminants page 44.
If you are getting your water from a private ground water well, and are looking for information related to wellwater quality, please visit CDC’s Private Wells page 45.
Water Treatment
Community Water Treatment
Drinking water supplies in the United States are among the safest in the world. However, even in the U.S., drinking water sources can become contaminated, causing sickness and disease from waterborne germs, such as Cryptosporidium, E. coli, Hepatitis A, Giardia intestinalis, and other pathogens.
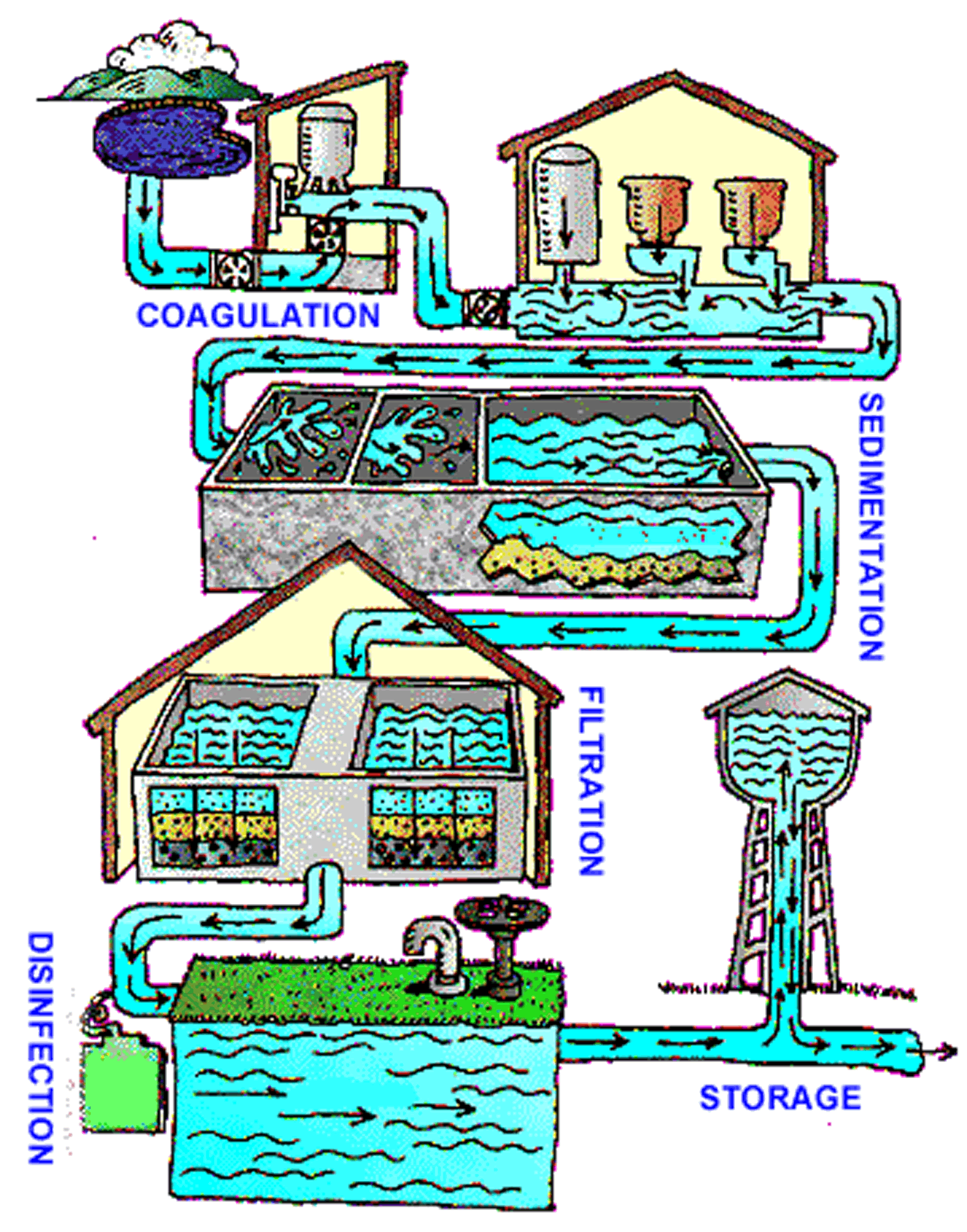
Drinking water sources are subject to contamination and require appropriate treatment to remove disease-causing agents. Public drinking water systems use various methods of water treatment to provide safe drinking water for their communities. Today, the most common steps in water treatment used by community water systems (mainly surface water treatment) include:
(Figure illustrating the water treatment cycle, showing coagulation, sedimentation, filtration, and disinfection)
- Coagulation and Flocculation
Coagulation and flocculation are often the first steps in water treatment. Chemicals with a positive charge are added to the water. The positive charge of these chemicals neutralizes the negative charge of dirt and other dissolved particles in the water. When this occurs, the particles bind with the chemicals and form larger particles, called floc.
- Sedimentation
During sedimentation, floc settles to the bottom of the water supply, due to its weight. This settling process is called sedimentation.
- Filtration
Once the floc has settled to the bottom of the water supply, the clear water on top will pass through filters of varying compositions (sand, gravel, and charcoal) and pore sizes, in order to remove dissolved particles, such as dust, parasites, bacteria, viruses, and chemicals.
- Disinfection
After the water has been filtered, a disinfectant (for example, chlorine, chloramine) may be added in order to kill any remaining parasites, bacteria, and viruses, and to protect the water from germs when it is piped to homes and businesses.
Water may be treated differently in different communities depending on the quality of the water that enters the treatment plant. Typically, surface water requires more treatment and filtration than ground water because lakes, rivers, and streams contain more sediment and pollutants and are more likely to be contaminated than ground water.
Some water supplies may also contain disinfections by-products, inorganic chemicals, organic chemicals, and radionuclides. Specialized methods for controlling formation or removing them can also be part of water treatment.
To learn more about the steps that are taken to make our water safe to drink, visit the United States Environmental Protection Agency’s (EPA) Public Drinking Water Systems webpage 44. To learn more about the 90+ contaminants EPA regulates and why, visit EPA’s Drinking Water Contaminants page 44.
Water Fluoridation
Community water fluoridation prevents tooth decay safely and effectively. Water fluoridation has been named one of 10 great public health achievements of the 20th century 46. For more information on the fluoridation process and to find details on your water system’s fluoridation, visit CDC’s Community Water Fluoridation page 47.
- Does bottled water contain fluoride ?
Bottled water products may contain fluoride, depending on the source of the water 48. Fluoride can be naturally present in the original source of the water, and many public water systems add fluoride to their water. The U.S. Food and Drug Administration (FDA) sets limits for fluoride in bottled water, based on several factors, including the source of the water. Bottled water products labeled as de-ionized, purified, demineralized, or distilled have been treated in such a way that they contain no or only trace amounts of fluoride, unless they specifically list fluoride as an added ingredient.
Water Disinfection with Chloramine and Chlorine
Water can come from a variety of sources, such as lakes and wells, which can be contaminated with germs that can make people sick. Germs can also contaminate water as it travels through miles of piping to get to a community. To prevent contamination with germs, water companies add a disinfectant—usually either chlorine 49 or chloramine 50 —that kills disease-causing germs such as Salmonella, Campylobacter, and norovirus. The type of chloramine discussed on this page that is used to treat drinking water (monochloramine) is not the same type that can form and harm the indoor air quality around swimming pools (dichloramine or trichloramine) 51.
Chlorine was first used as a drinking water disinfectant in Europe in the late 1800s. It was first used in the U.S. in 1908 in Jersey City, New Jersey 52. Chloramine has been used as a drinking water disinfectant in the U.S. in places like Cleveland, Ohio, Springfield, Illinois, and Lansing, Michigan since 1929 2. Today, chlorine and chloramine are the major disinfectants used to disinfect public water systems.
- Chlorine as a Primary Disinfectant in Drinking Water 53
Chlorine is a strong oxidant commonly used in water treatment for oxidation and disinfection. As an oxidant, chlorine is applied to control biological growth and to remove color, taste and odor compounds, iron and manganese, and other dissolved inorganic contaminants such as arsenic.
As a primary disinfectant, chlorine is applied to disinfect and to control microbial activity in the distribution system. It is also used as a secondary disinfectant after chlorine, ozone, UV irradiation, or chlorine dioxide.
Chlorine is a very effective primary disinfectant 54 :
+ Chlorine is very effective at killing most potentially harmful organisms.
+ Chlorine kills most potentially harmful organisms quickly.
+ Chlorine is the most frequently used primary disinfectant of drinking water.
Chlorination is the process of adding chlorine to drinking water to disinfect it and kill germs. Different processes can be used to achieve safe levels of chlorine in drinking water. Chlorine is available as compressed elemental gas, sodium hypochlorite solution (NaOCl) or solid calcium hypochlorite (Ca(OCl)2 53. While the chemicals could be harmful in high doses, when they are added to water, they all mix in and spread out, resulting in low levels that kill germs but are still safe to drink 44.
Chlorine levels up to 4 milligrams per liter (mg/L or 4 parts per million (ppm) are considered safe in drinking water. At this level, no harmful health effects are likely to occur 44.
Chlorinated water can taste and smell different than untreated water 44. Some people like the taste and smell of chlorinated water, and others do not. Taste and smell problems may arise depending upon the water quality and amount of chlorine in the water 44.
Chlorine was first used in the U.S. as a major disinfectant in 1908 in Jersey City, New Jersey 55. Chlorine use became more and more common in the following decades, and by 1995 about 64% of all community water systems in the United States used chlorine to disinfect their water 55.
Current studies indicate that using or drinking water with small amounts of chlorine does not cause harmful health effects and provides protection against waterborne disease outbreaks 56.
- Chloramines as a Secondary Disinfectant in Drinking Water 50
Chloramines (also known as secondary disinfection) because the monochloramine takes much longer than chlorine to kill most potentially harmful organisms 54 and because it is longer lasting than chlorine. Therefore, chloramines (monochloramine) is often used as a secondary disinfectant.
Chloramines are disinfectants used to treat drinking water and they:
- Are most commonly formed when ammonia is added to chlorine to treat drinking water.
- Provide longer-lasting disinfection as the as the water moves through pipes to consumers.
Chloramines have been used by water utilities since the 1930s. More than one in five Americans uses drinking water treated with chloramines.
The different types of chloramines are monochloramine, dichloramine, trichloramine, and organic chloramines 57.
+ When chloramines are used to disinfect drinking water, monochloramine is the most common form.
+ Dichloramine, trichloramine, and organic chloramines 58, 59 are produced when treating drinking water but at much lower levels than monochloramine.
+ Trichloramines 58 are typically associated with disinfected water used in swimming pools.
Dichloramine and trichloramine are chloramine compounds sometimes found in and around indoor swimming pools, which cause skin, eye, and respiratory problems 60. However, dichloramine and trichloramine are typically not an issue in treated drinking water, which uses monochloramine, because utilities carefully monitor the water quality 60.
Monochloramine has several advantages over chlorine as a secondary disinfectant 61 :
+ Monochloramine is more chemically stable than chlorine.
+ Monochloramine produces fewer potentially harmful regulated disinfection byproducts than chlorine.
+ Monochloramine is longer lasting than chlorine, making it useful for killing certain harmful organisms found in pipes such as those that cause Legionnaires’ disease.
Current studies indicate that using or drinking water with small amounts of chloramine does not cause harmful health effects and provides protection against waterborne disease outbreaks 62. These studies reported no observed health effects from drinking water with chloramine levels of less than 50 mg/L in drinking water 63. A normal level for drinking water disinfection can range from 1.0 to 4.0 mg/L 44.
Will chloramine affect my pets or plants ?
Chlorine and chloramine are toxic to fish, other aquatic animals, reptiles and amphibians 44. Unlike humans and other household pets, these types of animals absorb water directly into the blood stream 44. Don’t keep these animals in water that contains these disinfectants. Unlike chlorine, chloramine cannot be removed by letting water sit out for a few days. However, products are available at aquarium supply stores that can remove chloramine 44. Ask your local pet store about methods of removing disinfectants from water for these pets.
The small amount of chloramine added to water will not affect other pets (such as mammals and birds) and can be used regularly for watering and bathing animals 44.
Plants are not harmed by water treated with chloramine 44.
A Guide to Drinking Water Treatment Technologies for Household Use 64
This section is only to be used as a guide for household water treatment, not a recommendation. Before installing a household water treatment system, contact your local health department’s environmental health group for consultation.
The treatment technologies described can be used in conjunction with each other for greater pathogen reduction. The addition of coagulants, carbon, alum, and iron salts to filtration systems may aid in chemical removal from water.
- Point of Entry systems, or whole-house systems, are usually installed after the water meter.
- Point of Use water treatment systems typically treat water in batches and deliver water to a single tap, such as a kitchen sink faucet or an auxiliary faucet.
- Not all filters are created equal 65
Different water filters have different functions. Some can make your water taste better, while others can filter out harmful chemicals or germs. No single filter can keep every type of contaminant out of your drinking water and not everyone needs a water filter.
The water that comes to your tap actually contains small quantities of many other substances. Some of these are beneficial, such as the appropriate amount of a disinfectant, like chlorine, that helps keep your water safe from germs and fluoride, which helps prevent tooth decay. Other substances that might be in water can be harmful, such as lead and the germ Cryptosporidium. Filters can remove both good and bad substances from your water. Depending on your circumstances, filtering your water might not be a good idea.
There are many different types of filters available, and it can be confusing to decide whether you need one or which kind is best for you. In many areas of the world, the water is not safe to drink, so filtration is one option to protect your health. Here are some steps to consider when determining whether you want to use a water filter, and if so, what type and functions best fit your specific needs and preferences.
- One way to figure out what a water filter does is to look for an National Sanitation Foundation (NSF) certification and ratings 66 on the label. You can look up specific products in the NSF database online to see what they are certified to protect against. Some of the NSF standards applicable to water treatments are standard 41 (taste and odor), 53 (cyst reduction), 58 (reverse osmosis), and 62 (distillation) 66.
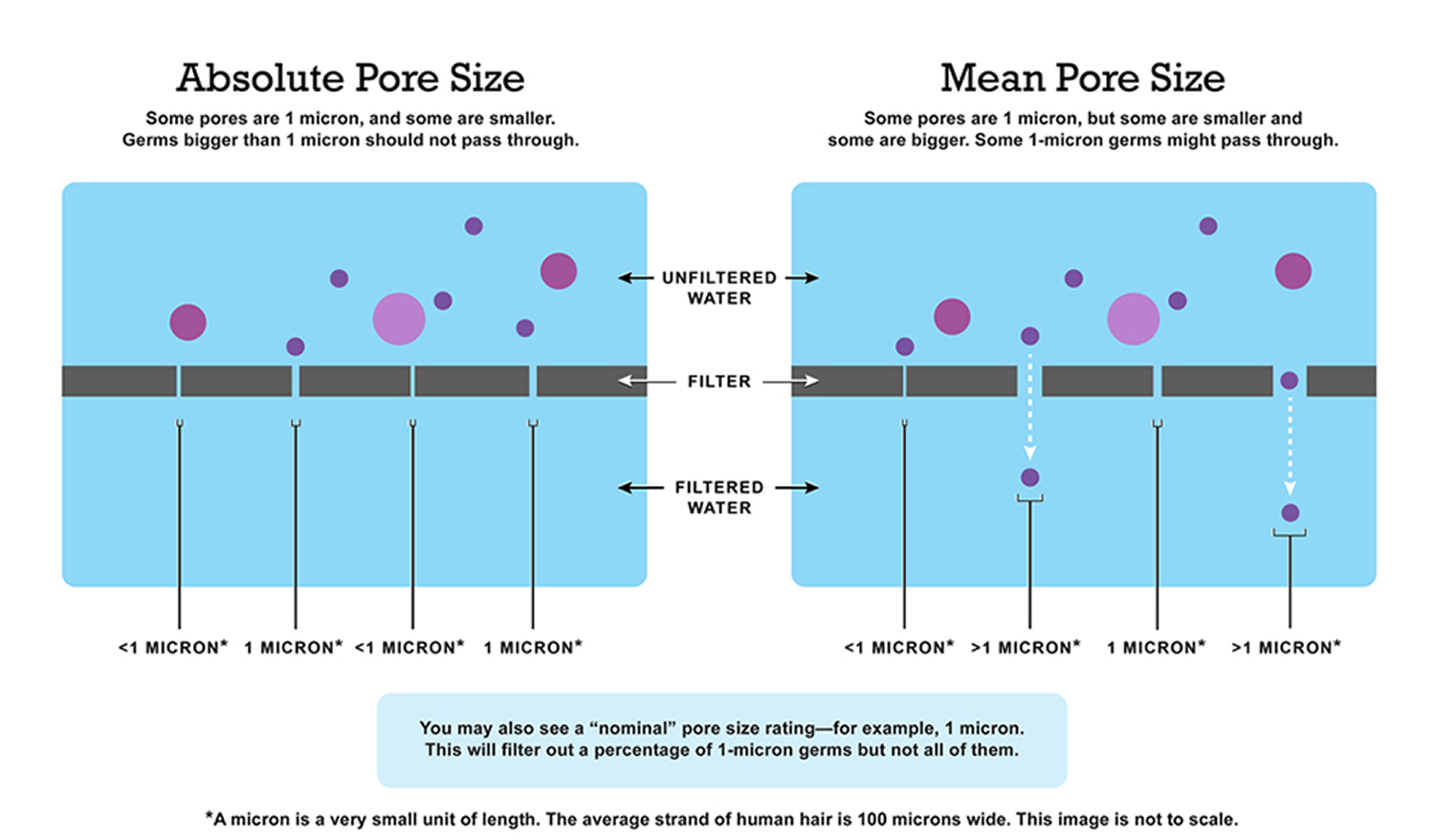
- What is the “pore size” of a filter ?The pore size is the size of the tiny holes in a filter that let water through. Think of a strainer or colander: The smaller the pores, the smaller the contaminants they keep out. If a filter has an “absolute” pore size of 1 micron, for example, each and every one of the filter’s pores is 1 micron or smaller. This means that any contaminant larger than 1 micron, like Cryptosporidium, will be caught in the filter and will not go into the filtered water. Filters with a “nominal” or “mean” pore size of 1 micron have an average pore size of 1 micron which means that some pores are smaller and some pores are larger than 1 micron, so contaminants like Cryptosporidium can travel through the larger pores and into the water you filter.
- Filtration is a physical process that occurs when liquids, gases, dissolved or suspended matter adhere to the surface of, or in the pores of, an absorbent medium. Filtration of contaminants depends highly on the amount of contaminant, size of the contaminant particle, and the charge of the contaminant particle. Depending on the household’s water needs, pretreatment before filtration may include the addition of coagulants and powdered activated carbon, adjustments in pH or chlorine concentration levels, and other pretreatment processes in order to protect the filter’s membrane surface.
Microfiltration 64
- A microfiltration filter has a pore size of approximately 0.1 micron (pore size ranges vary by filter from 0.05 micron to 5 micron);
- Microfiltration has a very high effectiveness in removing protozoa (for example, Cryptosporidium, Giardia);
- Microfiltration has a moderate effectiveness in removing bacteria (for example, Campylobacter, Salmonella, Shigella, E. coli);
- Microfiltration is not effective in removing viruses (for example, Enteric, Hepatitis A, Norovirus, Rotavirus);
- Microfiltration is not effective in removing chemicals.
Ultrafiltration 64
- An ultrafiltration filter has a pore size of approximately 0.01 micron (pore size ranges vary by filter from 0.001 micron to 0.05 micron; Molecular Weight Cut Off (MWCO) of 13,000 to 200,000 Daltons). Ultrafiltration filters remove particles based on size, weight, and charge;
- Ultrafiltration has a very high effectiveness in removing protozoa (for example, Cryptosporidium, Giardia);
- Ultrafiltration has a very high effectiveness in removing bacteria (for example, Campylobacter, Salmonella, Shigella, E. coli);
- Ultrafiltration has a moderate effectiveness in removing viruses (for example, Enteric, Hepatitis A, Norovirus, Rotavirus);
- Ultrafiltration has a low effectiveness in removing chemicals.
Nanofiltration 64
- A nanofiltration filter has a pore size of approximately 0.001 micron (pore size ranges vary by filter from 0.008 micron to 0.01 micron; Molecular Weight Cut Off (MWCO) of 200 to 2000 Daltons); Nanofiltration filters remove particles based on size, weight, and charge;
- Nanofiltration has a very high effectiveness in removing protozoa (for example, Cryptosporidium, Giardia);
- Nanofiltration has a very high effectiveness in removing bacteria (for example, Campylobacter, Salmonella, Shigella, E. coli);
- Nanofiltration has a very high effectiveness in removing viruses (for example, Enteric, Hepatitis A, Norovirus, Rotavirus);
- Nanofiltration has a moderate effectiveness in removing chemicals.
(Source 65).
Reverse Osmosis Systems
- Reverse Osmosis Systems use a process that reverses the flow of water in a natural process of osmosis so that water passes from a more concentrated solution to a more dilute solution through a semi-permeable membrane. Pre- and post-filters are often incorporated along with the reverse osmosis membrane itself.
- A reverse osmosis filter has a pore size of approximately 0.0001 micron.
- Reverse Osmosis Systems have a very high effectiveness in removing protozoa (for example, Cryptosporidium, Giardia);
- Reverse Osmosis Systems have a very high effectiveness in removing bacteria (for example, Campylobacter, Salmonella, Shigella, E. coli);
- Reverse Osmosis Systems have a very high effectiveness in removing viruses (for example, Enteric, Hepatitis A, Norovirus, Rotavirus);
- Reverse Osmosis Systems will remove common chemical contaminants (metal ions, aqueous salts), including sodium, chloride, copper, chromium, and lead; may reduce arsenic, fluoride, radium, sulfate, calcium, magnesium, potassium, nitrate, and phosphorous.
Distillation Systems
- Distillation Systems use a process of heating water to the boiling point and then collecting the water vapor as it condenses, leaving many of the contaminants behind.
- Distillation Systems have a very high effectiveness in removing protozoa (for example, Cryptosporidium, Giardia);
- Distillation Systems have a very high effectiveness in removing bacteria (for example, Campylobacter, Salmonella, Shigella, E. coli);
- Distillation Systems have a very high effectiveness in removing viruses (for example, Enteric, Hepatitis A, Norovirus, Rotavirus);
- Distillation Systems will remove common chemical contaminants, including arsenic, barium, cadmium, chromium, lead, nitrate, sodium, sulfate, and many organic chemicals.
Ultraviolet Treatment Systems (with pre-filtration)
- Ultraviolet Treatment with pre-filtration is a treatment process that uses ultraviolet light to disinfect water or reduce the amount of bacteria present.
- Ultraviolet Treatment Systems have a very high effectiveness in removing protozoa (for example, Cryptosporidium, Giardia);
- Ultraviolet Treatment Systems have a very high effectiveness in removing bacteria (for example, Campylobacter, Salmonella, Shigella, E. coli);
- Ultraviolet Treatment Systems have a high effectiveness in removing viruses (for example, Enteric, Hepatitis A, Norovirus, Rotavirus);
- Ultraviolet Treatment Systems are not effective in removing chemicals.
Choosing Home Water Filters & Other Water Treatment Systems
- Step 1: Know your water source
Your water filtering needs depend on the quality of your particular water supply, your personal preferences when it comes to taste and odor, and any special health circumstances in your household. To choose the best filter for your home water consumption needs, it is helpful first to learn more about your water source. Understanding what contaminants may already exist in your water is an important first step when deciding whether you want or need a filtration system and, if so, what type will best meet your needs.
Do you get your water from a private well or cistern ?
If you get your water from a private well, you should have your water tested by a state-certified lab at least once a year. Before you hire someone to test your water, make sure they are certified, and make sure they will test for total coliform bacteria, nitrates, total dissolved solids, and pH levels, and other contaminants common in your area. Contact your local health department for information on contaminants in your area and for suggestions on water tests you should request. You should also get your water tested if someone in your household becomes pregnant or if a child joins your household 1. Also consider testing your water if you notice changes in color, taste, or odor. Regular testing at a certified lab can provide you with information on levels of different contaminants in your water. Once you know whether any contaminants found in your water exceed the recommended levels, you can take steps to choose a filter that will reduce those contaminants.
- Private Ground Water Wells 45
Many people in the United States receive their water from private ground water wells. EPA regulations that protect public drinking water systems do not apply to privately owned wells. As a result, owners of private wells are responsible for ensuring that their water is safe from contaminants.
When rain falls, much of it is absorbed into the ground. Water that’s not used by plants moves downward through pores and spaces in the rock until it reaches a dense layer of rock. The water trapped below the ground in the pores and spaces above the dense rock barrier is called ground water, and this is the water we get when we drill wells. Another common term for ground water is “aquifer” or “ground water aquifer.”
All private wells use ground water. If polluted ground water is consumed, it could cause illness. Ground water pollution can be caused by seepage through landfills, failed septic tanks, underground fuel tanks, fertilizers and pesticides, and runoff from urban areas.
It is important that private ground water wells are checked regularly to ensure that the water is safe for drinking 45.
Do you get your water from rainwater ?
Collecting and using rainwater can be a great way to conserve resources. Some people use rainwater for watering plants, cleaning, bathing, or drinking. Rainwater collection is not allowed in all places. Some states consider rainwater the property of the state and prohibit its collection, so you should consult with your local government (for example, your environmental quality department or health department) before proceeding.
- Germs and Other Contaminants
While useful for many things, rainwater is not as pure as you might think, so you can’t assume it’s safe to drink. Rain can wash different types of contaminants into the water you collect (for example, bird poop on your roof could end up in your water barrel or tank). Rainwater can carry bacteria, parasites, viruses, and chemicals that could make you sick, and it has been linked to disease outbreaks 67, 68.
The risk of getting sick from rainwater may be different depending on your location, how frequently it rains, the season 5, and how you collect and store the rainwater. Dust, smoke, and soot from the air can be dissolved in rainwater before it lands on your roof. Roofing materials, gutters, piping, and storage materials can introduce harmful chemicals like asbestos, lead, and copper to the water, though building standards minimize some of this 69, 70, 71. Dirt and germs can be washed into collected rainwater from the roof, especially when rain follows several days of dry weather 67, 68, 72.
Rain barrels, for example, do not provide any type of disinfection or filtration. Adding chlorine or iodine to water does not protect against chemicals, and some parasites are very chlorine tolerant. Boiling the water will kill germs but will not remove chemicals. Using a simple device called a “first flush diverter” to remove the first water that comes out of the system (which may have been sitting) may help avoid some of these contaminants 71, 73.
Some cisterns, which are common in certain areas, can provide safe water supplies. Some people add purchased, treated water to the rainwater they collect in their cistern. This may make the treated water less safe. Cisterns that are intended for drinking water should be properly maintained 74. Consider adding a screen to the water inlet or emptying the rain barrel in less than 10 days to prevent mosquitoes from using the rain barrel as a breeding site 72. Water intended for drinking needs to be filtered, disinfected, and tested regularly 72, 75.
- Preventing Illness
If you collect and store rainwater for drinking, you have a private, or individual, water system and are responsible for ensuring that your water is safe. You should have your water and your system tested regularly and maintain the system properly 68, 76. When rainwater is used as a supplemental water source, homeowners should ensure that rainwater cannot enter pipes containing safe drinking water 68, 69. Contact your state or local health department for more information.
To lower your risk of getting sick, consider using rainwater only for uses such as watering plants that you don’t eat, washing items that are not used for cooking or eating, and bathing (keeping water out of your mouth and nose). If possible, avoid using rainwater for drinking, cooking, brushing your teeth, or rinsing or watering plants that you intend to eat. Instead, use municipal tap water if it is available, or purchase bottled or bulk water for these purposes.
If you have a weakened immune system, you should be especially careful when choosing your drinking water source. Discuss this with your healthcare provider.
Do you get your water from a public system ?
If so, you will get a report on your water each year. EPA requires all public water systems to send an annual report about the quality of drinking water, as well as contaminant levels, to people whose water comes from public sources. This report, called a Consumer Confidence Report (CCR), is sent by July 1 of every calendar year and will come in your water bill.
Household Water Treatment 77
Even though EPA regulates and sets standards for public drinking water, many Americans use a home water treatment unit to:
- Remove specific contaminants
- Take extra precautions because a household member has a compromised immune system
- Improve the taste of drinking water
Household water treatment systems are composed of two categories: point-of-use and point-of-entry.
- Point-of-Entry systems are typically installed after the water meter and treat most of the water entering a residence.
- Point-of-Use systems are systems that treat water in batches and deliver water to a tap, such as a kitchen or bathroom sink or an auxiliary faucet mounted next to a tap.
The most common types of household water treatment systems consist of:
- Filtration Systems
A water filter is a device which removes impurities from water by means of a physical barrier, chemical, and/or biological process.
- Water Softeners
Water Softeners use ion exchange technology for chemical or ion removal to reduce the amount of hardness (calcium, magnesium) in the water; they can also be designed to remove iron and manganese, heavy metals, some radioactivity, nitrates, arsenic, chromium, selenium, and sulfate. They do not protect against protozoa, bacteria, and viruses.
- Distillation Systems
Distillation is a process in which impure water is boiled and the steam is collected and condensed in a separate container, leaving many of the solid contaminants behind.
- Disinfection
Disinfection is a physical or chemical process in which pathogenic microorganisms are deactivated or killed. Examples of chemical disinfectants are chlorine, chlorine dioxide, and ozone. Examples of physical disinfectants include ultraviolet light, electronic radiation, and heat.
How does lead get into your tap water ?
Measures taken during the last two decades have greatly reduced exposures to lead in tap water. These measures include actions taken under the requirements of the 1986 and 1996 amendments to the Safe Drinking Water Act 78 and the U.S. Environmental Protection Agency’s (EPA’s) Lead and Copper Rule 63.
Even so, lead still can be found in some metal water taps, interior water pipes, or pipes connecting a house to the main water pipe in the street. Lead found in tap water usually comes from the corrosion of older fixtures or from the solder that connects pipes. When water sits in leaded pipes for several hours, lead can leach into the water supply 79.
How do you know if your tap water is contaminated with lead ?
The only way to know whether your tap water contains lead is to have it tested. You cannot see, taste, or smell lead in drinking water. Therefore, you must ask your water provider whether your water has lead in it. For homes served by public water systems, data on lead in tap water may be available on the Internet from your local water authority. If your water provider does not post this information, you should call and find out 79.
Does a high lead level in your tap water cause health effects ?
High levels of lead in tap water can cause health effects if the lead in the water enters the bloodstream and causes an elevated blood lead level 79.
Most studies show that exposure to lead-contaminated water alone would not be likely to elevate blood lead levels in most adults, even exposure to water with a lead content close to the EPA action level for lead of 15 parts per billion (ppb). Risk will vary, however, depending on the individual, the circumstances, and the amount of water consumed. For example, infants who drink formula prepared with lead-contaminated water may be at a higher risk because of the large volume of water they consume relative to their body size.
What can you do to reduce or eliminate lead in your tap water ?
If your tap water contains lead at levels exceeding EPA’s action level of 15 ppb, you should take action to minimize your exposure to the lead in the water.
You should begin by asking your water authority these questions 79:
1. Does my water have lead in it above EPA’s action level of 15 parts per billion (ppb) ?
- If the answer is no, no action is needed.
- If the answer is yes, also ask the next question:
2. Does the service pipe at the street (header pipe) have lead in it ?
This information is very important. It determines which of the next two actions (A or B) you should follow to protect your household’s health.
A) If the pipe in the street (header pipe) DOES NOT have lead, the lead in your tap water may be coming from fixtures, pipes, or elsewhere inside your home.
Until you eliminate the source, you should take the following steps any time you wish to use tap water for drinking or cooking, especially when the water has been off and sitting in the pipes for more than 6 hours:
a. Before using any tap water for drinking or cooking, flush your water system by running the kitchen tap (or any other tap you take drinking or cooking water from) on COLD for 1–2 minutes 79;
b. Then, fill a clean container(s) with water from this tap. This water will be suitable for drinking, cooking, preparation of baby formula, or other consumption. To conserve water, collect multiple containers of water at once (after you have fully flushed the water from the tap as described).
B) If the pipe at the street (header pipe) DOES contain lead, lead in the tap water may be coming from that pipe or connected pipes (it may also be coming from sources inside your home).
Until the lead source is eliminated, you should take the following steps any time you wish to use tap water for drinking or cooking, especially when the water has been off and sitting in the pipes for more than 6 hours. Please note that additional flushing is necessary:
a. Before using any tap water for drinking or cooking, run high-volume taps (such as your shower) on COLD for 5 minutes or more;
b. Then, run the kitchen tap on COLD for 1–2 additional minutes;
c. Fill a clean container(s) with water from this tap. This water will be suitable for drinking, cooking, preparation of baby formula, or other consumption. To conserve water, collect multiple containers of water at once (after you have fully flushed the water from the tap as described).
3. In all situations, drink or cook only with water that comes out of the tap cold. Water that comes out of the tap warm or hot can contain much higher levels of lead. Boiling this water will NOT reduce the amount of lead in your water.
4. You can also reduce or eliminate your exposure to lead in drinking water by consuming only bottled water or water from a filtration system that has been certified by an independent testing organization to reduce or eliminate lead. See resources below.
5. Children and pregnant women are especially vulnerable to the effects of lead exposure. Therefore, for homes with children or pregnant women and with water lead levels exceeding EPA’s action level of 15 ppb, CDC recommends using bottled water or water from a filtration system that has been certified by an independent testing organization to reduce or eliminate lead for cooking, drinking, and baby formula preparation. Because most bottled water does not contain fluoride, a fluoride supplement may be necessary.
Also, some bottled waters have not been tested and may not be appropriate for consumption. Contact independent testing organizations that certify bottled water. See resources below.
6. Make sure that repairs to copper pipes do not use lead solder.
Advice for lead safe water practices after plumbing work in housing with lead water lines or lead solder.
These practices include
- Testing water after plumbing work in older housing. Please contact your state lead program for information about water testing in your area.
- Inspecting the aerator on the end of the faucet and removing any debris such as metal particles.
- Flushing water lines before using the water for drinking or cooking.
If you own your home, you may also consider full replacement of lead water lines by removing the private lines running from the water meter into your home. This precaution has not been adequately studied, however, because the data available to CDC 79 included too few homes having had full replacement of lead water lines. Contact your water authority for information about replacing water service lines.
If your water has high lead levels, is it safe to take a bath or shower ?
Yes. Bathing and showering should be safe for you and your children, even if the water contains lead over EPA’s action level. Human skin does not absorb lead in water 79.
This information applies to most situations and to a large majority of the population, but individual circumstances may vary. Some situations, such as cases involving highly corrosive water, may require additional recommendations or more stringent actions. Your local water authority is always your first source for testing and identifying lead contamination in your tap water. Many public water authorities have websites that include data on drinking water quality, including results of lead testing. Links to such data can be found on the EPA website 44.
- Jequier E, Constant F. Water as an essential nutrient: the physiological basis of hydration. Eur J Clin Nutr. 2010;64(2):115–123. http://www.nature.com/ejcn/journal/v64/n2/full/ejcn2009111a.html[↩][↩]
- Lang F, Waldegger S (1997). Regulating cell volume. Am Scientist 85, 456–463.[↩]
- NHS. Dehydration – Introduction. http://www.nhs.uk/Conditions/Dehydration/Pages/Introduction.aspx[↩]
- Water as an essential nutrient: the physiological basis of hydration. European Journal of Clinical Nutrition (2010) 64, 115–123; doi:10.1038/ejcn.2009.111; published online 2 September 2009. http://www.nature.com/ejcn/journal/v64/n2/full/ejcn2009111a.html[↩][↩][↩][↩][↩][↩][↩]
- Dietary Reference Intakes (2006). The Essential Guide to Nutrients Requirements. Institute of Medicine of the National Academies: Washington DC, 543 pp.[↩]
- Wang ZM, Deurenberg P, Wang W, Pietrobelli A, Baumgartner RN, Heymsfield SB (1999). Hydration of fat-free body mass: a review and critique of a classic body-composition constant. Am J Clin Nutr 69, 833–841. https://www.ncbi.nlm.nih.gov/pubmed/10232621[↩]
- Ganong WF (2005). Review of Medical Physiology, 22nd edn, LANGE-Science: New York.[↩][↩][↩][↩]
- Grandjean AC, Campbell SM (2004). Hydration: Fluids for Life. A monograph by the North American Branch of the International Life Science Institute. ILSI North America: Washington DC.[↩]
- EFSA (2008). Draft dietary reference values for water. Scientific Opinion of the Panel on Dietetic Products, Nutrition and Allergies.[↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Manz F, Wentz A, Sichert-Hellert W (2002). The most essential nutrient: defining the adequate intake of water. J Pediatr 141, 587–592. https://www.ncbi.nlm.nih.gov/pubmed/12378203[↩]
- Sawka MN, Cheuvront SN, Carter III R (2005). Human water needs. Nutr Rev 63, S30–S39. https://academic.oup.com/nutritionreviews/article-abstract/63/suppl_1/S30/1927756/Human-Water-Needs[↩]
- National Academy of Sciences. Dietary Reference Intakes: Water, Potassium, Sodium, Chloride, and Sulfate. http://nationalacademies.org/hmd/reports/2004/dietary-reference-intakes-water-potassium-sodium-chloride-and-sulfate.aspx[↩][↩]
- Montain SJ, Latzka WA, Sawka MN (1999). Fluid replacement recommendations for training in hot weather. Mil Med 164, 502–508. https://www.ncbi.nlm.nih.gov/pubmed/10414066[↩][↩]
- Murray B (2007). Hydration and physical performance. J Am Coll Nutr 26 (Suppl 5), 542S–548S. https://www.ncbi.nlm.nih.gov/pubmed/17921463[↩]
- Maughan RJ, Shirreffs SM, Watson P (2007). Exercise, heat, hydration, and the brain. J Am Coll Nutr 26 (Suppl 5), 604S–612S. https://www.ncbi.nlm.nih.gov/pubmed/17921473[↩]
- Sawka MN, Cheuvront SN, Carter III R (2005). Human water needs. Nutr Rev 63, S30–S39. https://www.ncbi.nlm.nih.gov/pubmed/16028570[↩][↩][↩]
- Phillips PA, Rolls BJ, Ledingham JG, Forsling ML, Morton JJ, Crowe MJ et al. (1984). Reduced thirst after water deprivation in healthy elderly men. N Engl J Med 311, 753–759. https://www.ncbi.nlm.nih.gov/pubmed/6472364[↩]
- Häussinger D (1996). The role of cellular hydration in the regulation of cell function. Biochem J 313, 697–710. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1216967/[↩][↩][↩]
- Grandjean AC, Campbell SM (2004). Hydration: Fluids for Life. A monograph by the North American Branch of the International Life Science Institute. ILSI North America: Washington DC.[↩]
- Ritz P, Berrut G (2005). The importance of good hydration for day-to-day health. Nutr Rev 63, S6–S13. https://academic.oup.com/nutritionreviews/article-abstract/63/suppl_1/S6/1927737/The-Importance-of-Good-Hydration-for-Day-to-Day[↩]
- Szinnai G, Schachinger H, Arnaud MJ, Linder L, Keller U (2005). Effect of water deprivation on cognitive-motor performance in healthy men and women. Am J Physiol Regul Integr Comp Physiol 289, R275–R280. http://ajpregu.physiology.org/content/289/1/R275.long[↩]
- Centers for Disease Control and Prevention, Division of Nutrition, Physical Activity and Obesity (CDC) Rethink Your Drink. http://www.cdc.gov/healthyweight/healthy_eating/drinks.html[↩][↩]
- United States Department of Agriculture (USDA) ChooseMyPlate.gov.http://www.choosemyplate.gov/food-groups/downloads/tentips/dgtipsheet19makebetterbeveragechoices.pdf[↩][↩]
- American Medical Association (AMA) Action Plan for Healthy Eating. http://www.nwsphysicians.com/web/wp-content/uploads/2014/01/AMA-Healthy-Eating.pdf[↩][↩]
- American Diabetes Association (ADA) Lose Weight by Eating Healthy. http://www.diabetes.org/living-with-diabetes/parents-and-kids/children-and-type-2/preventing-type-2-in-children.html[↩][↩]
- American Heart Association (AHA) Replacing Sugary Drinks in Your Diet. http://www.heart.org/HEARTORG/GettingHealthy/NutritionCenter/SimpleCookingwithHeart/Rethink-Your-Drink-Reducing-Sugary-Drinks-in-Your-Diet_UCM_445267_Article.jsp[↩][↩]
- American Academy of Pediatrics Kids Should Not Consume Energy Drinks, and Rarely Need Sports Drinks. http://www.aap.org/en-us/about-the-aap/aap-press-room/Pages/Kids-Should-Not-Consume-Energy-Drinks,-and-Rarely-Need-Sports-Drinks,-Says-AAP.aspx[↩][↩]
- Nutrients. 2016 Jan; 8(1): 19. Published online 2016 Jan 2. doi: 10.3390/nu8010019. Negative, Null and Beneficial Effects of Drinking Water on Energy Intake, Energy Expenditure, Fat Oxidation and Weight Change in Randomized Trials: A Qualitative Review. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4728633/[↩][↩]
- Let’s Move, America’s Move to Raise a Healthier Generation of Kids. http://www.letsmove.gov/[↩]
- Drink Up. http://www.youarewhatyoudrink.org/[↩]
- Efficacy of water preloading before main meals as a strategy for weight loss in primary care patients with obesity: RCT. Parretti HM, Aveyard P, Blannin A, Clifford SJ, Coleman SJ, Roalfe A, Daley AJ. Obesity (Silver Spring). 2015 Sep; 23(9):1785-91. https://www.ncbi.nlm.nih.gov/pubmed/26237305/[↩]
- Water consumption increases weight loss during a hypocaloric diet intervention in middle-aged and older adults. Dennis EA, Dengo AL, Comber DL, Flack KD, Savla J, Davy KP, Davy BM. Obesity (Silver Spring). 2010 Feb; 18(2):300-7. https://www.ncbi.nlm.nih.gov/pubmed/19661958/[↩]
- Qualitative and/or quantitative drinking water recommendations for pediatric obesity treatment. Stookey JD, Del Toro R, Hamer J, Medina A, Higa A, Ng V, TinajeroDeck L, Juarez L. J Obes Weight Loss Ther. 2014 Oct 11; 4(4):232. https://www.ncbi.nlm.nih.gov/pubmed/25632375/[↩]
- Daily self-monitoring of body weight, step count, fruit/vegetable intake, and water consumption: a feasible and effective long-term weight loss maintenance approach. Akers JD, Cornett RA, Savla JS, Davy KP, Davy BM. J Acad Nutr Diet. 2012 May; 112(5):685-692.e2. https://www.ncbi.nlm.nih.gov/pubmed/22709772/[↩]
- https://www.ncbi.nlm.nih.gov/mesh/68014867[↩]
- U.S. National Library of Medicine, PubChem Open Chemistry Database. Water. https://pubchem.ncbi.nlm.nih.gov/compound/water[↩]
- Human Metabolome Database. The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. http://www.hmdb.ca/metabolites/HMDB02111[↩]
- Centers for Disease Control and Prevention. Drinking Water. https://www.cdc.gov/healthywater/drinking/[↩][↩]
- United States Environmental Protection Agency. Information about Public Water Systems. https://www.epa.gov/dwreginfo/information-about-public-water-systems[↩][↩][↩][↩][↩]
- Centers for Disease Control and Prevention. Public Water Systems. https://www.cdc.gov/healthywater/drinking/public/index.html[↩]
- United States Environmental Protection Agency. FACTOIDS: Drinking Water and Ground Water Statistics for 2007. https://nepis.epa.gov/Exe/ZyPDF.cgi/P100N2VG.PDF?Dockey=P100N2VG.PDF[↩][↩][↩][↩]
- Centers for Disease Control and Prevention. Consumer Confidence Reports (CCRs). https://www.cdc.gov/healthywater/drinking/public/understanding_ccr.html[↩]
- United States Environmental Protection Agency. Ground Water and Drinking Water. https://www.epa.gov/ground-water-and-drinking-water[↩]
- United States Environmental Protection Agency. Drinking Water Contaminants – Standards and Regulations. https://www.epa.gov/dwstandardsregulations[↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Centers for Disease Control and Prevention. Private Ground Water Wells. https://www.cdc.gov/healthywater/drinking/private/wells/index.html[↩][↩][↩]
- Centers for Disease Control and Prevention. Ten Great Public Health Achievements — United States, 1900-1999. https://www.cdc.gov/mmwr/preview/mmwrhtml/mm4850bx.htm[↩]
- Centers for Disease Control and Prevention. Community Water Fluoridation. https://www.cdc.gov/fluoridation/index.html[↩]
- Centers for Disease Control and Prevention. Bottled Water. https://www.cdc.gov/fluoridation/faqs/bottled_water.htm[↩]
- United States Environmental Protection Agency. Chlorine. https://safewater.zendesk.com/hc/en-us/sections/202366478[↩]
- United States Environmental Protection Agency. Chloramines in Drinking Water. https://www.epa.gov/dwreginfo/chloramines-drinking-water[↩][↩]
- United States Environmental Protection Agency. Quick reference guide: Stage 1 and 2 Disinfectants and Disinfection Byproducts Rules. https://www.epa.gov/ground-water-and-drinking-water/national-primary-drinking-water-regulations#Disinfectants[↩]
- United States Environmental Protection Agency. The history of drinking water treatment 2000. https://nepis.epa.gov/Exe/ZyNET.exe/P1002SMN.TXT?ZyActionD=ZyDocument&Client=EPA&Index=2000+Thru+2005&Docs=&Query=&Time=&EndTime=&SearchMethod=1&TocRestrict=n&Toc=&TocEntry=&QField=&QFieldYear=&QFieldMonth=&QFieldDay=&IntQFieldOp=0&ExtQFieldOp=0&XmlQuery=&File=D%3A%5Czyfiles%5CIndex%20Data%5C00thru05%5CTxt%5C00000019%5CP1002SMN.txt&User=ANONYMOUS&Password=anonymous&SortMethod=h%7C-&MaximumDocuments=1&FuzzyDegree=0&ImageQuality=r75g8/r75g8/x150y150g16/i425&Display=p%7Cf&DefSeekPage=x&SearchBack=ZyActionL&Back=ZyActionS&BackDesc=Results%20page&MaximumPages=1&ZyEntry=1&SeekPage=x&ZyPURL[↩]
- United States Environmental Protection Agency. Chlorine. https://iaspub.epa.gov/tdb/pages/treatment/treatmentOverview.do?treatmentProcessId=-1118142891[↩][↩]
- United States Environmental Protection Agency. Basic Information About Drinking Water Disinfection. https://www.epa.gov/sites/production/files/2015-09/documents/q5.pdf[↩][↩]
- United States Environmental Protection Agency. Safe Drinking Water Act: Consumer Confidence Reports (CCR). https://www.epa.gov/ccr[↩][↩]
- Centers for Disease Control and Prevention. Surveillance Reports for Drinking Water-associated Disease & Outbreaks. https://www.cdc.gov/healthywater/surveillance/drinking-surveillance-reports.html[↩]
- United States Environmental Protection Agency. Basic Information About Chloramines. https://www.epa.gov/sites/production/files/2015-09/documents/q1.pdf [↩]
- Dichloramine is formed when the chlorine to ammonia-nitrogen weight ratio is greater than 5:1, however, this reaction is very slow. Organic chloramines are formed when chlorine reacts with organic nitrogen compounds. Source: Optimizing Chloramine Treatment, 2nd Edition, AwwaRF, 2004[↩][↩]
- Trichloramine formation does not usually occur under normal drinking water treatment conditions. However, if the pH is lowered below 4.4 or the chlorine to ammonia-nitrogen weight ratio becomes greater than 7.6:1, then trichloramine can form. Trichloramine formation can occur at a pH between 7 and 8 if the chlorine to ammonia-nitrogen weight ratio is increased to 15:1. Source: Optimizing Chloramine Treatment, 2nd Edition, AwwaRF, 2004[↩]
- Water Research Foundation. Long-Term Effects of Disinfection Changes on Water Quality. http://www.waterrf.org/PublicReportLibrary/91169.pdf[↩][↩]
- United States Environmental Protection Agency. Basic Information About Drinking Water Disinfection. https://www.epa.gov/sites/production/files/2015-09/documents/q6.pdf[↩]
- Centers for Disease Control and Prevention. Disinfection with Chloramine. https://www.cdc.gov/healthywater/drinking/public/chloramine-disinfection.html[↩]
- United States Environmental Protection Agency. Lead and Copper Rule. https://www.epa.gov/dwreginfo/lead-and-copper-rule[↩][↩]
- Centers for Disease Control and Prevention. A Guide to Drinking Water Treatment Technologies for Household Use. https://www.cdc.gov/healthywater/drinking/home-water-treatment/household_water_treatment.html[↩][↩][↩][↩]
- Centers for Disease Control and Prevention. Choosing Home Water Filters & Other Water Treatment Systems. https://www.cdc.gov/healthywater/drinking/home-water-treatment/water-filters.html[↩][↩]
- National Sanitation Foundation. http://www.nsf.org/[↩][↩]
- J Environ Qual. 2011 Jan-Feb;40(1):13-21. Microbiological quality of roof-harvested rainwater and health risks: a review. https://www.ncbi.nlm.nih.gov/pubmed/21488488[↩][↩]
- J Amer Water Res Assoc. 2002;38(5):1301-6. Health risks associated with consumption of untreated water from household roof catchment systems. http://onlinelibrary.wiley.com/doi/10.1111/j.1752-1688.2002.tb04349.x/abstract[↩][↩][↩][↩]
- National Sanitation Foundation (NSF). Rainwater Collection. http://www.nsf.org/consumer-resources/green-living/rainwater-collection[↩][↩]
- National Sanitation Foundation (NSF). Health Effects from Rainwater Catchment System Components. http://info.nsf.org/Certified/Protocols/Listings.asp?TradeName=&Standard=P151[↩]
- Oregon Department of Consumer & Business Services, Building Codes Division (BCD). Oregon Smart Guide to Rainwater Harvesting. http://www.oregon.gov/bcd/pages/index.aspx[↩][↩]
- Virginia Department of Health. Virginia Rainwater Harvesting & Use Guidelines. http://www.vdh.virginia.gov/[↩][↩][↩]
- Water Sci Technol. 2011;63(11):2725-31. Quality assessment of rooftop runoff and harvested rainwater from a building catchment. https://www.ncbi.nlm.nih.gov/pubmed/22049771[↩]
- Georgia Department of Community Affairs. Georgia Rainwater Harvesting Guidelines.[↩]
- Virginia Water Resources Research Center. Evaluation of Rooftop Rainfall Collection-Cistern Storage Systems in Southwest Virginia.[↩]
- Water Res. 2011;45(12):3765-75. Monitoring of water quality from roof runoff: Interpretation using multivariate analysis. https://www.ncbi.nlm.nih.gov/pubmed/21561638[↩]
- Centers for Disease Control and Prevention. Water Treatment. https://www.cdc.gov/healthywater/drinking/public/water_treatment.html[↩]
- United States Environmental Protection Agency. Safe Drinking Water Act (SDWA). https://www.epa.gov/sdwa[↩]
- Centers for Disease Control and Prevention. Water. https://www.cdc.gov/nceh/lead/tips/water.htm[↩][↩][↩][↩][↩][↩][↩]