Periventricular leukomalacia
Periventricular leukomalacia (PVL) is a type of brain injury that affects premature babies that is characterized by the death or damage and softening of the white matter around the ventricles (fluid-filled spaces) of the brain potentially leading to neurological and developmental problems 1, 2, 3, 4, 5, 6, 7, 8, 9. “Periventricular” refers to the area around the ventricles, the spaces in the brain containing the cerebrospinal fluid (CSF). “Leuko” refers to the brain’s white matter. “Malacia” means softening. The white matter is the paler tissue of your brain and spinal cord, consisting mainly of nerve fibers (axons or neurons) with their myelin sheaths that connect different areas of your brain to each other and to your spinal cord like electrical wire. Myelin gives the white matter its color and it also protects the nerve fibers (axons or neurons) from injury. Myelin improves the speed and transmission of electrical nerve signals along extensions of the nerve cells called axons. The white matter connects regions of your brain and transmits information between the nerve cells and the spinal cord to the rest of your body, as well as from one part of the brain to another. While all the parts of your brain work together, each part is responsible for a specific function — controlling everything from your thought, memory, emotion, touch, motor skills, vision, heart rate, breathing, temperature, hunger and every process that regulates your body. The brain and spinal cord that extends from it make up the central nervous system, or CNS. Periventricular leukomalacia (PVL) represent small “holes” in baby’s brain due to the death of small areas of brain tissue around the normal fluid-filled cavities or the ventricles of the brain (see Figure 2 and 3 below). There are case series reporting periventricular leukomalacia (PVL) in term infants, but premature babies are at the highest risk of periventricular leukomalacia 10, 11, 12. Periventricular leukomalacia is caused by a lack of oxygen or blood flow to the periventricular area of the brain, which results in the death or loss of brain tissue. The periventricular area or the area around the spaces in the brain called ventricles that contains nerve fibers to carry messages from the brain to the body’s muscles. Although babies with periventricular leukomalacia generally have no outward signs or symptoms of the disorder, they are at risk for motor disorders, delayed mental development, coordination problems, and vision and hearing impairments. Periventricular leukomalacia (PVL) may be accompanied by a hemorrhage or bleeding in the periventricular-intraventricular area (the area around and inside the ventricles), and can lead to cerebral palsy, accounting for 40–50% cerebral palsy (CP) cases 13, 14. A recent study in Canada reported that 7.06% of fullterm children with cerebral palsy are complicated with periventricular leukomalacia 15 and in China the morbidity of periventricular leukomalacia in fullterm children with cerebral palsy was reported to be 5.7% 16.
Periventricular leukomalacia tends to be suspected in babies born prematurely or at low birthweight, so babies may be checked soon after birth before any symptoms start to show. Periventricular leukomalacia is often diagnosed by ultrasound of your baby’s head – this is similar to the ultrasound scans carried out in pregnancy but the probe is held over the soft spot on the top of the head (fontanelle). Another imaging scan that might be suggested is an MRI scan – this shows the characteristic appearance of the condition (see Figure 3 below).
In addition to a complete medical history and physical examination, diagnostic procedures for periventricular leucomalacia may include:
- Cranial ultrasound. A painless test that uses sound waves to view the baby’s brain through the fontanelles, the soft openings between the skull bones. With periventricular leucomalacia, the ultrasound shows cysts or hollow places in the brain tissue. The timing of initial cranial ultrasonography can be useful in determining the timing of the insult. Cystic periventricular leukomalacia has been identified on cranial sonograms on the first day of life, indicating that the event was prenatal rather than perinatal or postnatal.
- Magnetic resonance imaging (MRI). This test uses a combination of a large magnet, radio frequencies, and a computer to produce detailed images of internal structures. MRI may show some of the early changes in the brain tissue that occur with periventricular leucomalacia.
Periventricular leukomalacia (PVL) is the second most common brain complication in preterm infants, after periventricular hemorrhage. Periventricular leucomalacia is caused by inadequate blood supply in the watershed territory of the preterm infant brain, which in premature infants are located in a periventricular area 17. This area of the brain is very susceptible to injury, especially in premature babies whose brain tissues are fragile. However, it is not clear when the trigger for periventricular leukomalacia occurs, before, during, or after birth. Periventricular leukomalacia (PVL) occurs most commonly in premature infants born at less than 32 weeks’ gestation who have a birth weight of less than 1500 g. Other factors that may be associated with periventricular leukomalacia include early rupture of membranes (amniotic sac) and infection inside the uterus. Many of these infants have a history of maternal chorioamnionitis (a serious infection of the placenta, amniotic fluid, and membranes surrounding the fetus during pregnancy, often linked to prolonged rupture of membranes or labor). Most affected infants experience cardiorespiratory problems, such as respiratory distress syndrome or pneumonia, in association with low blood pressure or patent ductus arteriosus (a congenital heart defect where a blood vessel called the ductus arteriosus that connects the aorta and pulmonary artery in the fetus doesn’t close as it should after birth, leading to extra blood flow to the lungs) during their first days of life. Bacterial infection at birth also appears to be a risk factor.
The incidence of periventricular leukomalacia ranges from 4 to 26% in premature infants in neonatal intensive care units (NICUs) 18. The incidence of periventricular leukomalacia is much higher in reports from autopsy studies of premature infants. As many as 75% of premature infants have evidence of periventricular leukomalacia on postmortem examination.
With periventricular leucomalacia, the area of damaged brain tissue can affect the nerve cells that control motor movements. As the baby grows, the damaged nerve cells cause the muscles to become spastic, or tight, and resistant to movement. Infants with periventricular leukomalacia are at risk for development of neurodevelopmental deficits 19. Babies with periventricular leucomalacia have a higher risk of developing cerebral palsy (a group of disorders that prevent the child from controlling their muscles normally), and may have intellectual impairment, learning difficulties or visual disturbances.
- Mild periventricular leukomalacia is often associated with spastic diplegia, a type of cerebral palsy that primarily affects the legs causing muscle stiffness and difficulty with movement, often leading to a “scissoring” gait where legs cross at the knees.
- Severe periventricular leukomalacia is associated with quadriplegia also known as tetraplegia is paralysis that affects all four limbs. Severe periventricular leukomalacia is also associated with a higher incidence of intelligence deficiencies and visual disturbances.
There is no specific treatment for periventricular leukomalacia. Treatment is symptomatic and supportive to improve the child’s quality of life. Children with periventricular leukomalacia should receive regular medical screenings to determine appropriate interventions.
Although there is no treatment for periventricular leucomalacia, doctors may recommend other types of care for your child, such as:
- Physical therapy
- Occupational therapy
- Speech-language therapy
- Vision therapy
Most of these therapies are provided through early Intervention programs in your community.
The outlook (prognosis) for individuals with periventricular leucomalacia depends on the severity of the brain damage, some children may have mild symptoms while others may experience significant disabilities.
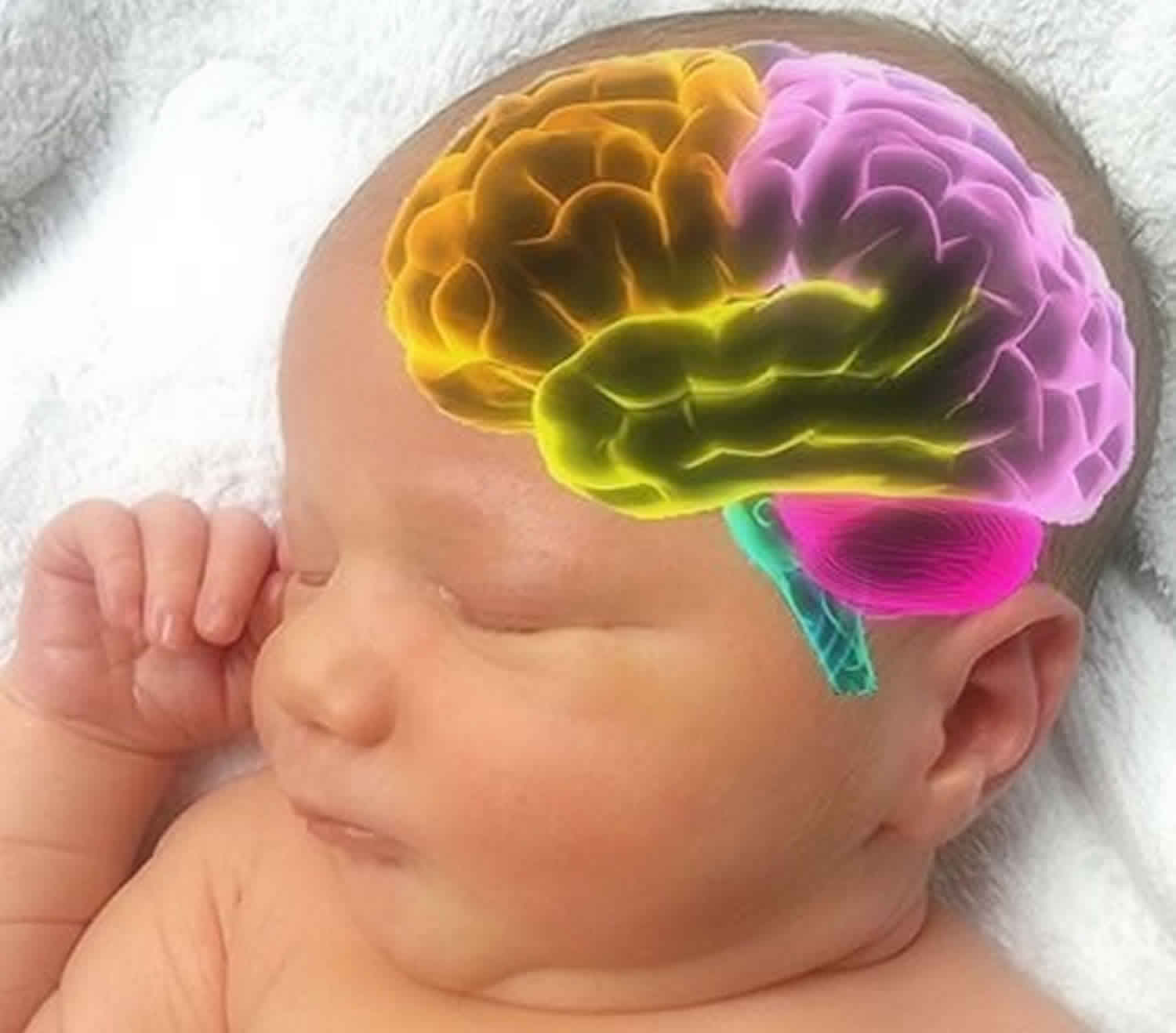
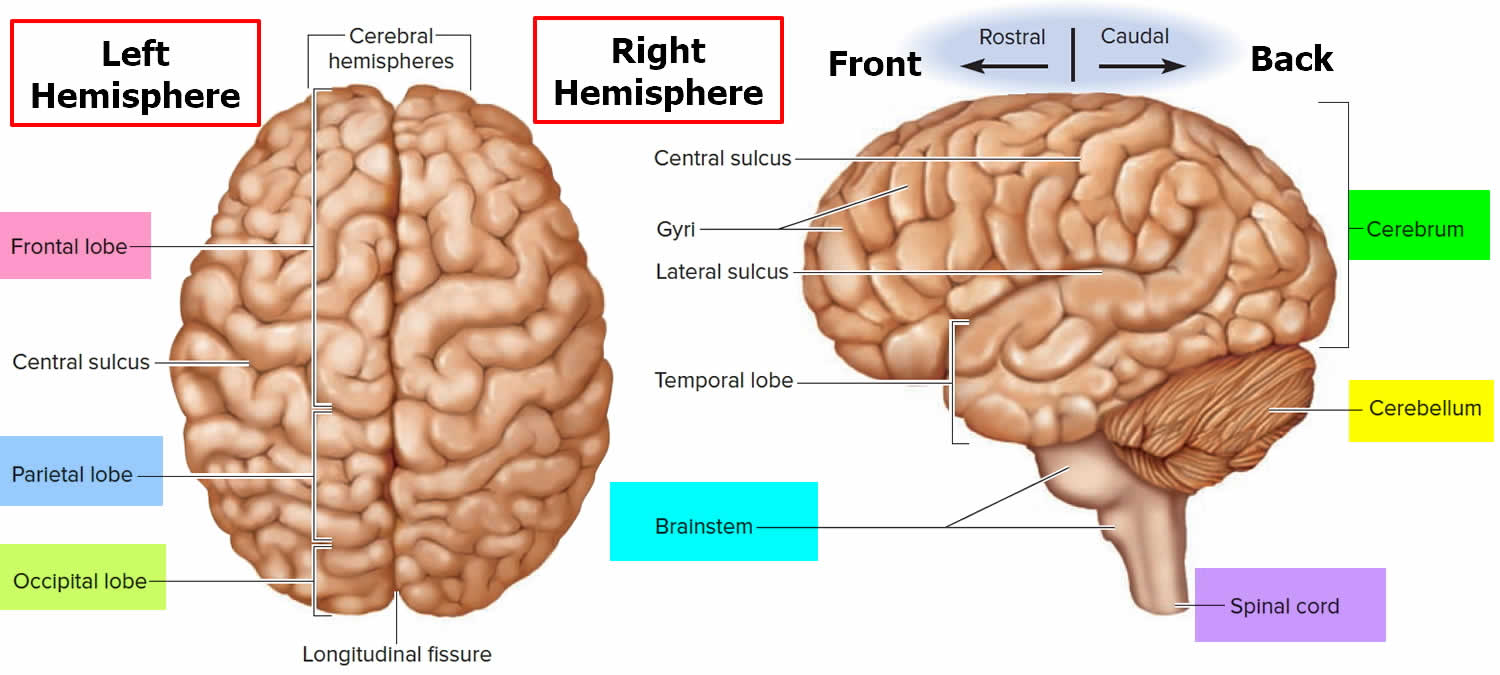
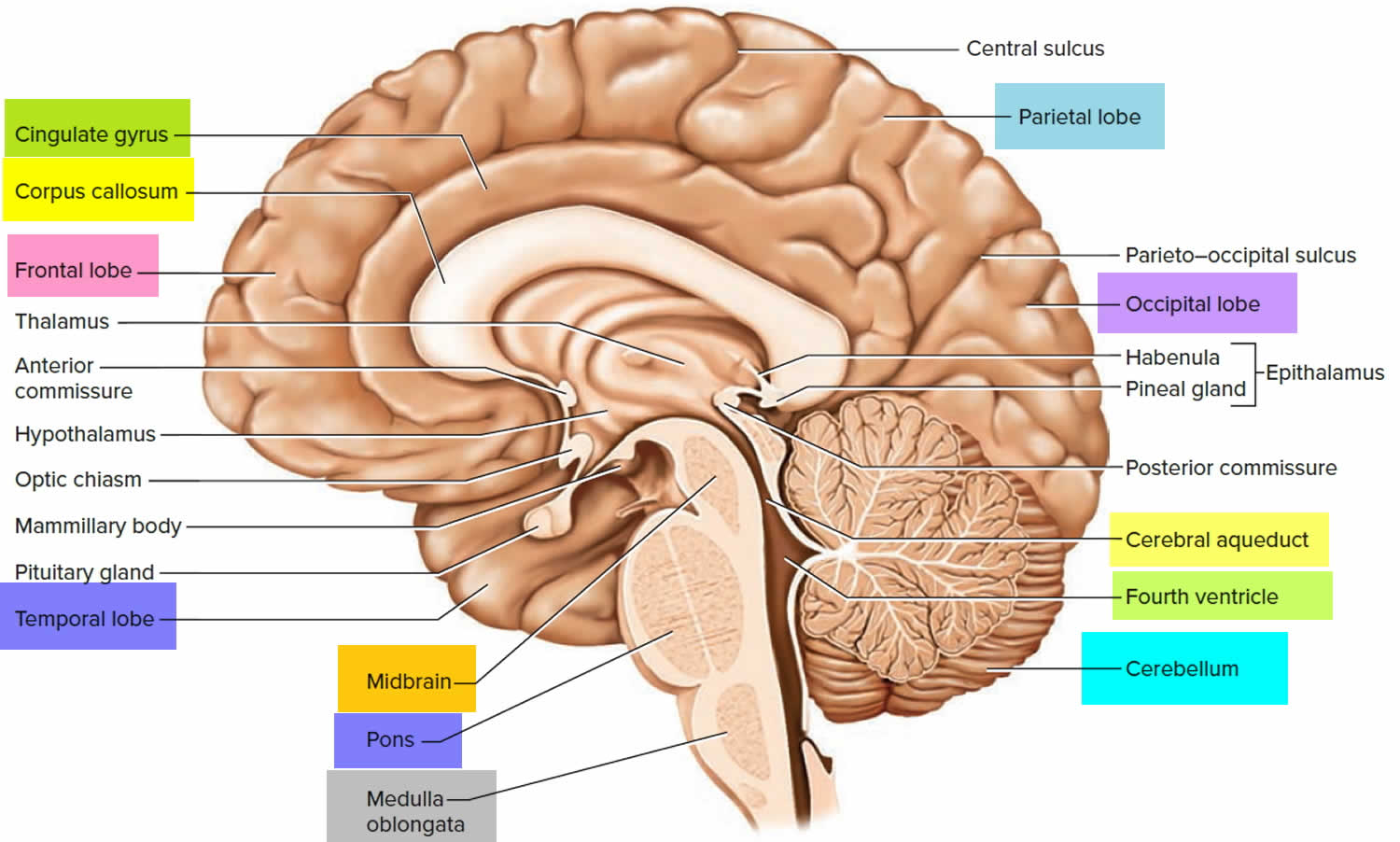
Figure 1. Brain anatomy
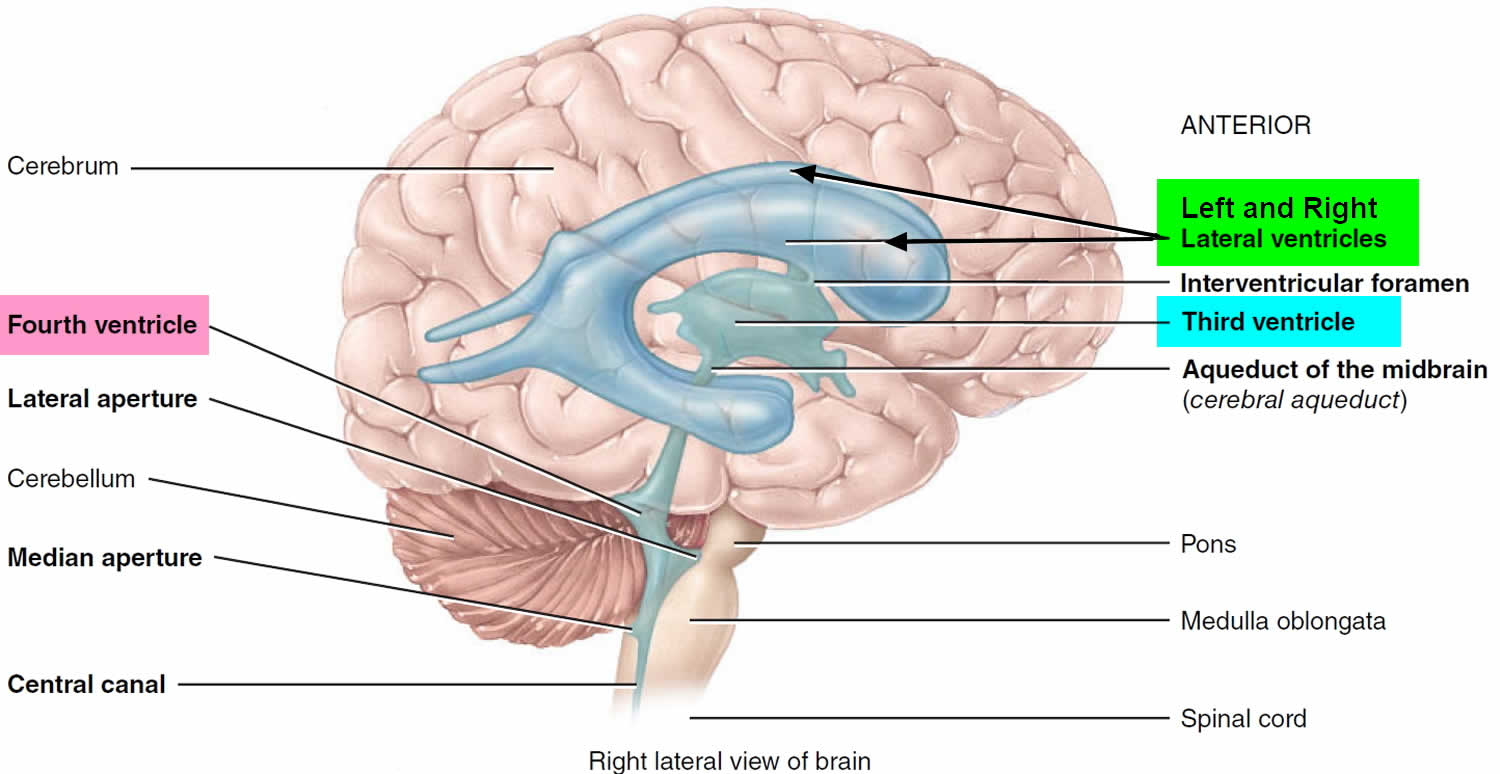
Figure 2. Ventricles of the brain
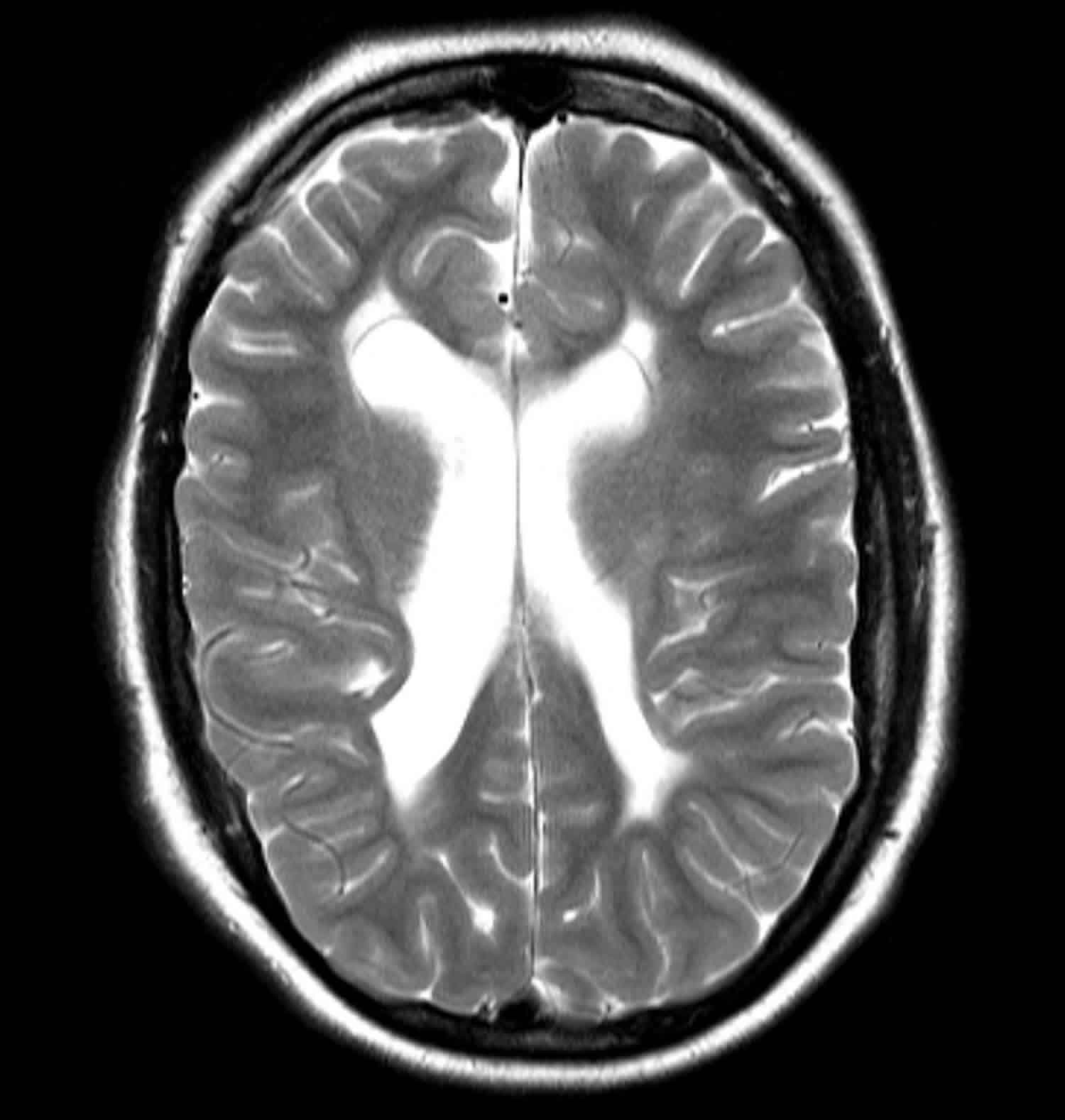
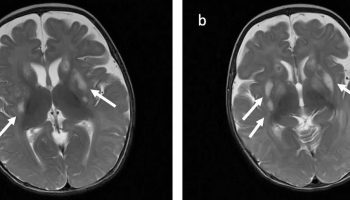
Figure 3. Periventricular leukomalacia MRI scans
Footnote: This child had a history of a perinatal insult and had significant developmental delay. The lateral ventricles are enlarged with wavy lateral contours demonstrating increase in volume with associated white matter tissue loss typical of moderate to severe periventricular leukomalacia.
Can periventricular leukomalacia cause migraines?
Migraines or headaches are not specifically described as a feature of periventricular leukomalacia. However, there are two case reports of headaches in individuals with periventricular leukomalacia. Two different studies 20, 21 of children with recurrent headaches each reported one individual with periventricular leukomalacia diagnosed after brain imaging. It is difficult to determine if headaches can be caused by periventricular leukomalacia because there are few reported cases. It is important to keep in mind that although there are often symptoms that are documented in case reports, they are based on the specific individuals that are studied and may differ from one affected person to another.
What is myelin?
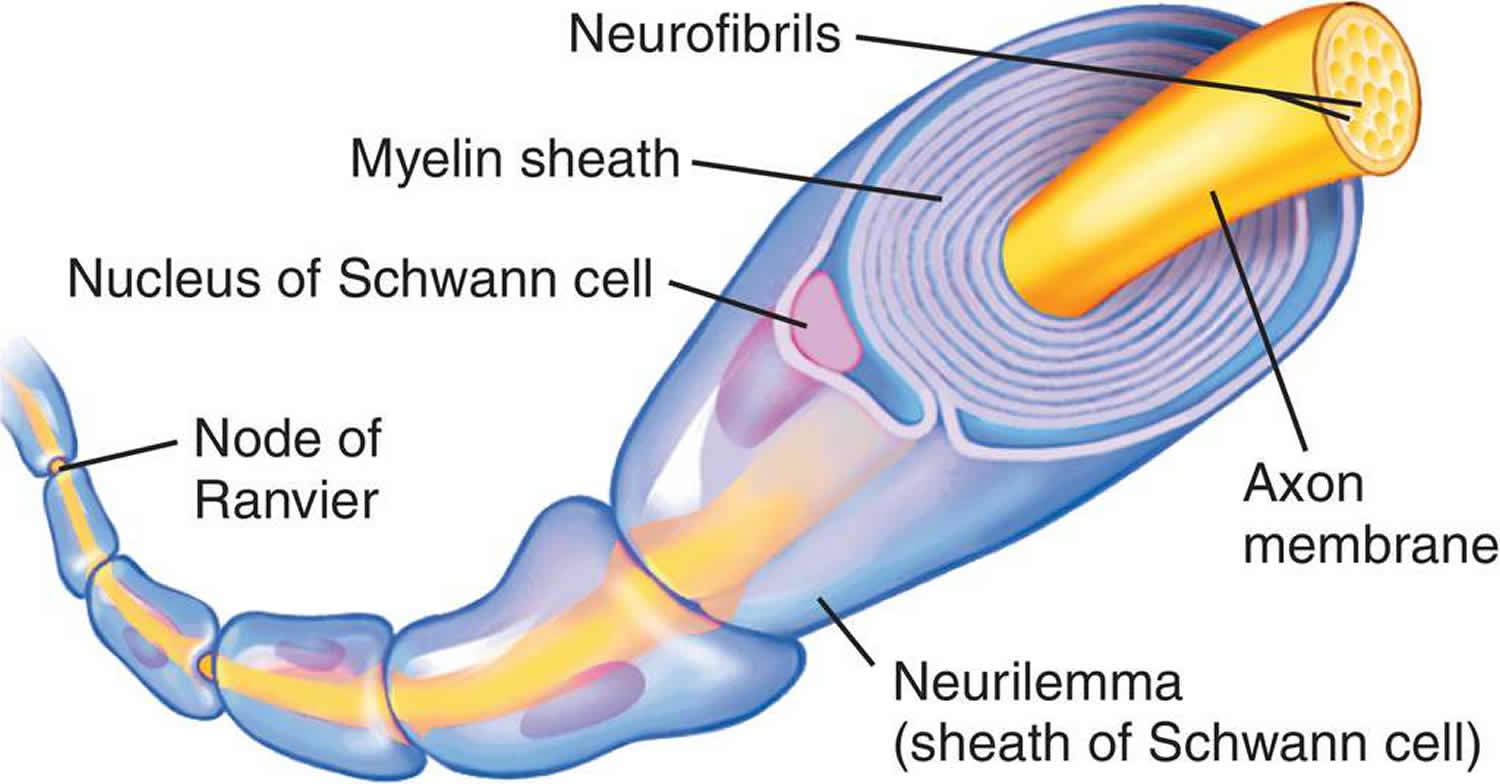
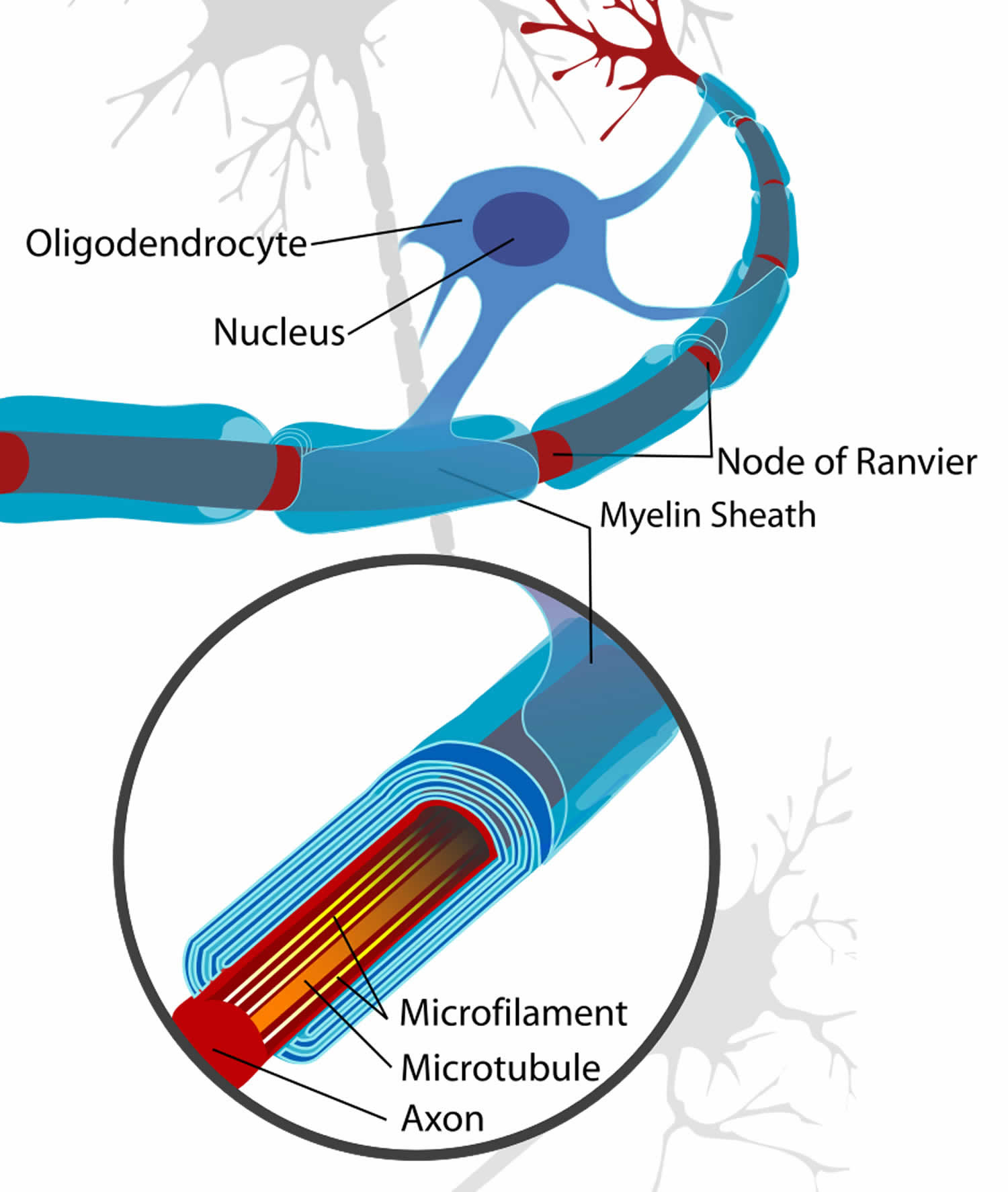
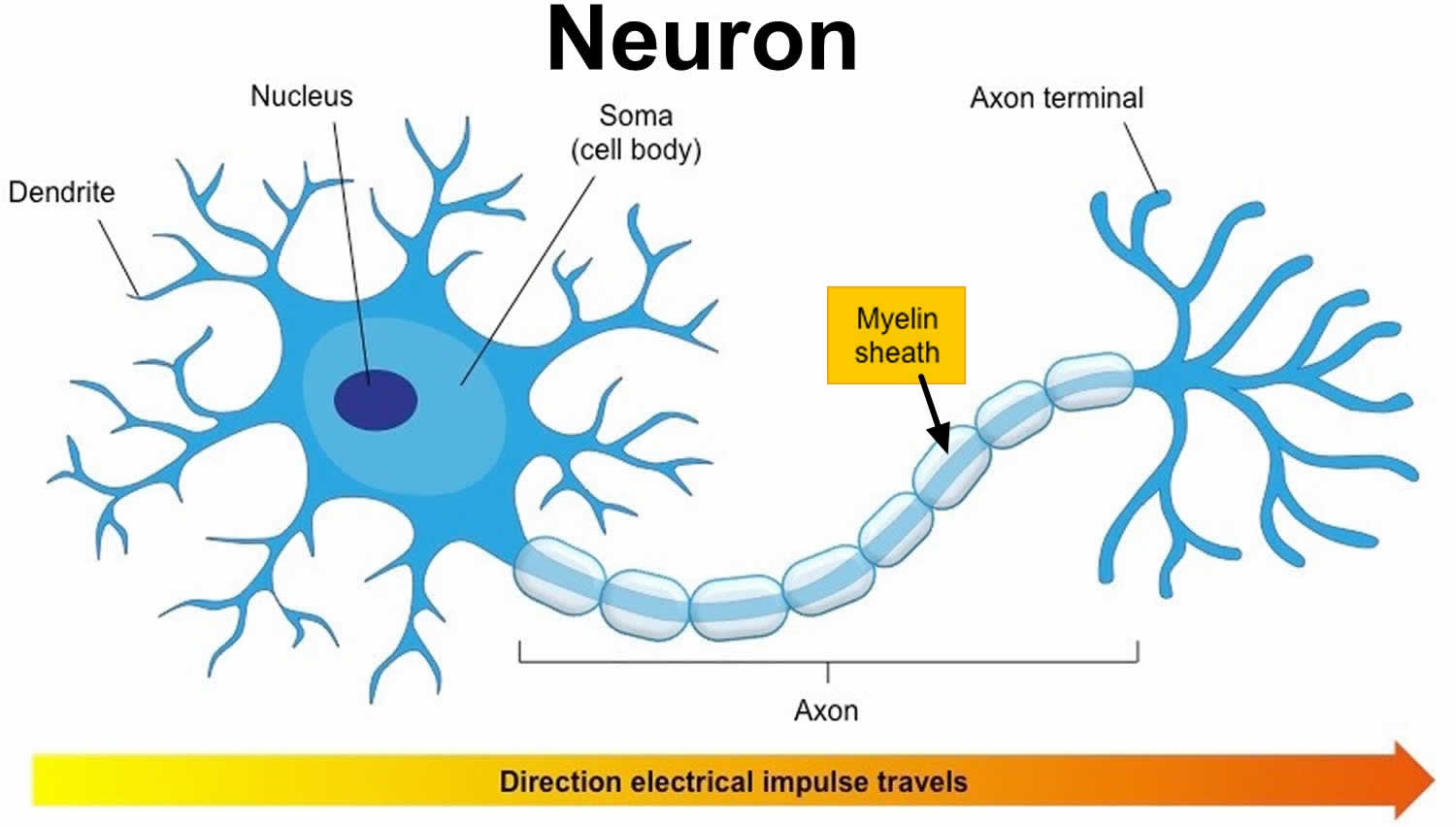
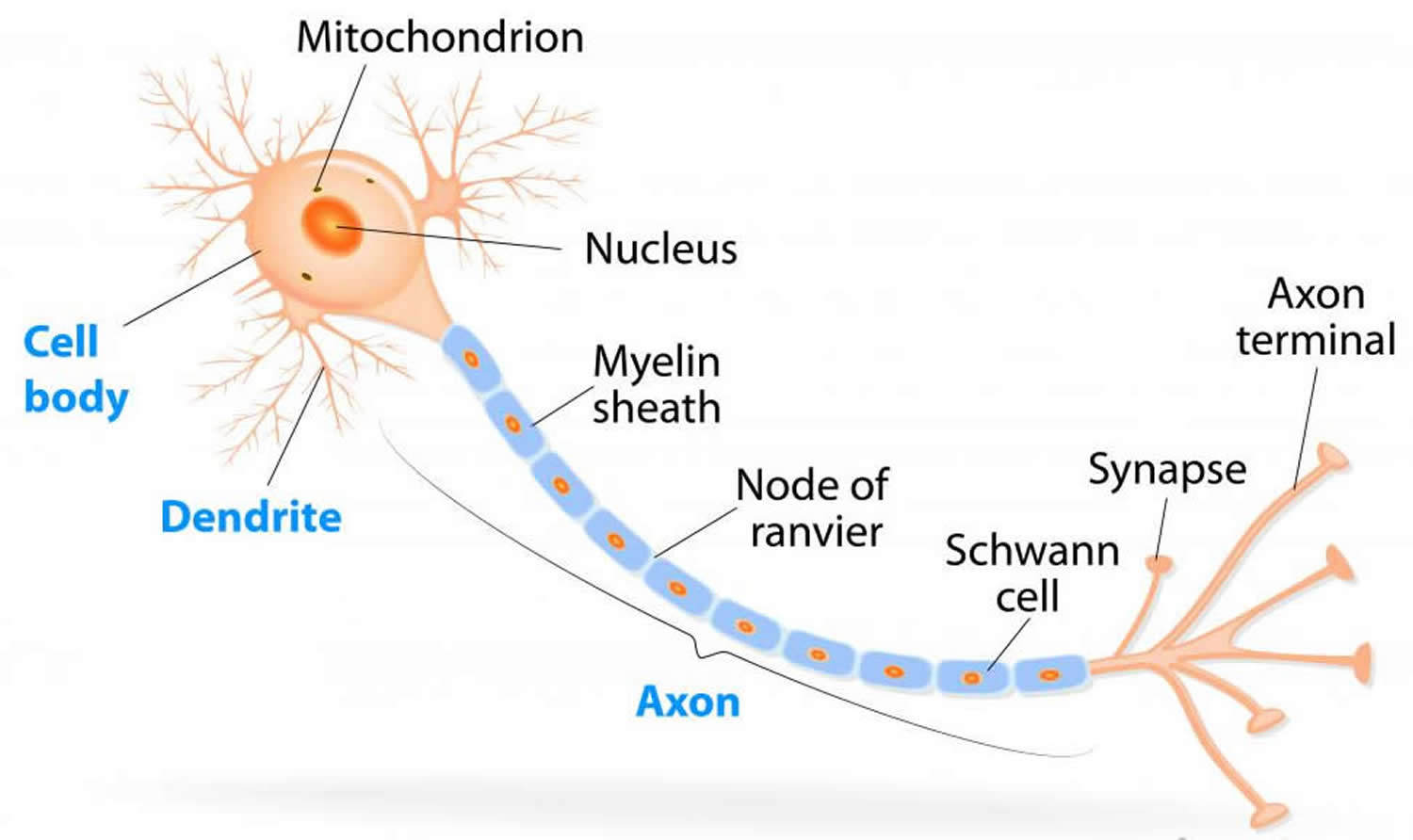
Myelin is a lipid-rich (fatty) substance and the myelin membranes originate from and are a part of the Schwann cells in the peripheral nervous system and the glial cells called oligodendrocytes in the central nervous system (brain and spinal cord). Myelin is an electrical insulator that insulates nerve cell axons to increase the speed at which information (encoded as an electrical signal) travels from one nerve cell body to another (as in the central nervous system) or from a nerve cell body to a muscle (as in the peripheral nervous system). The myelinated axon can be likened to an electrical wire (the axon) with insulating material (myelin) around it. However, unlike the plastic covering on an electrical wire, myelin does not form a single long sheath over the entire length of the axon. Rather, each myelin sheath insulates the axon over a single section and in general, each axon comprises multiple long myelinated sections separated from each other by short gaps called the nodes of Ranvier. Nodes of Ranvier are the short (~1 micron) unmyelinated regions of the axon between adjacent long (~0.2 mm – >1 mm) myelinated internodes 22. Each myelin sheath is formed by the concentric wrapping of an oligodendrocyte or Schwann cell process around the axon. Each myelin-generating cell (oligodendrocyte in the CNS or Schwann cell in the peripheral nervous system) furnishes myelin for only one segment of any given axon. The periodic interruptions where short portions of the axon are left uncovered by myelin, the nodes of Ranvier, are critical to the functioning of myelin.
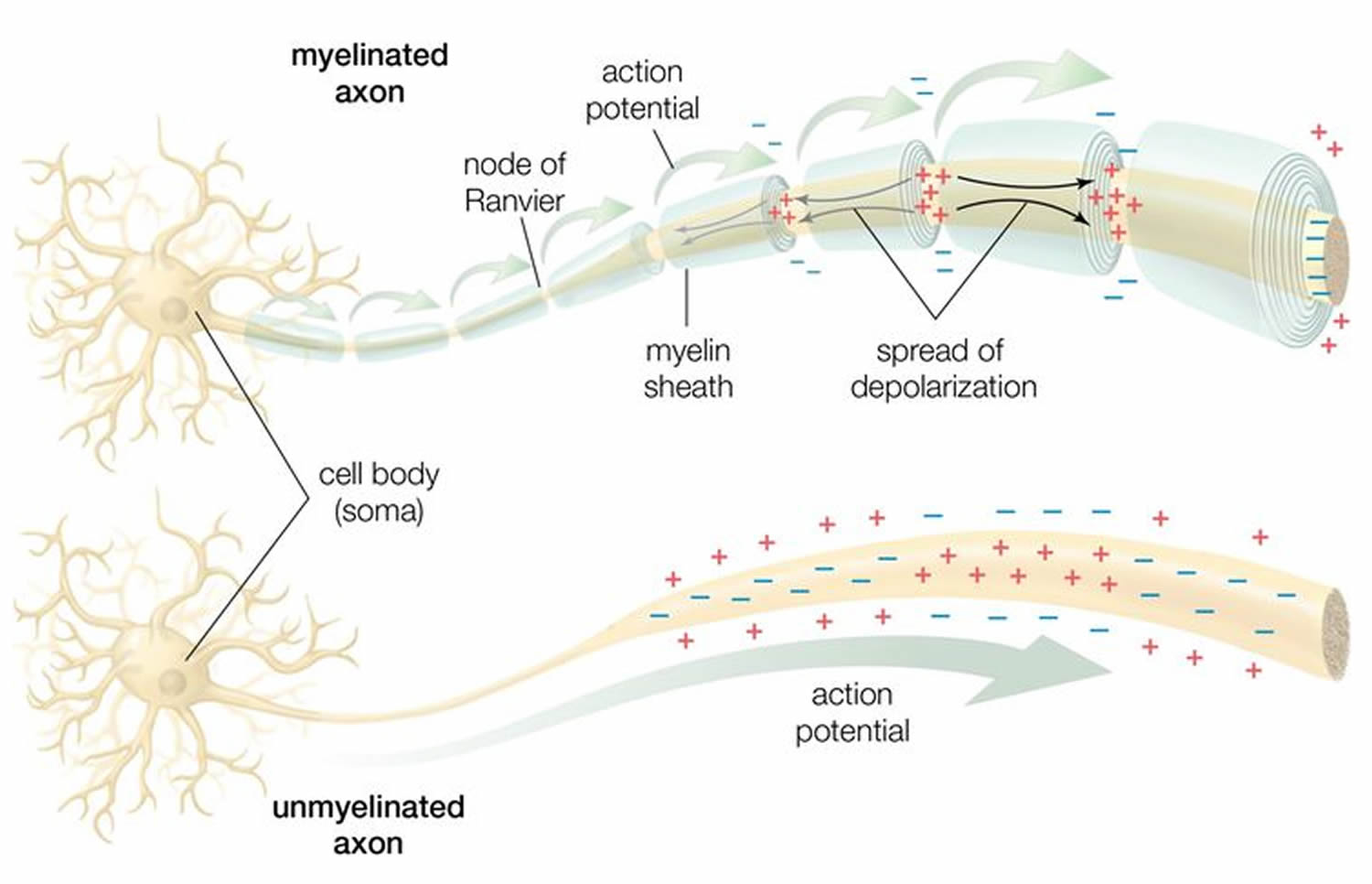
In myelinated axons, the excitable axonal membrane is exposed to the extracellular space only at the nodes of Ranvier; this is the location of sodium channels 23. When the membrane at the node of Ranvier is excited, the local circuit generated cannot flow through the high-resistance sheath and, therefore, flows out through and depolarizes the membrane at the next node, which might be 1 mm or farther away (Figure 3). The low capacitance of the myelin sheath means that little energy is required to depolarize the remaining membrane between the nodes of Ranvier, which results in local circuit spreading at an increased speed. Active excitation of the axonal membrane jumps from node to node; this form of impulse propagation is called saltatory conduction (Latin saltare, “to jump”). This saltatory conduction whereby the action potential “jumps” from one node of Ranvier, over a long myelinated stretch of the axon called the internode, before ‘recharging’ at the next node of Ranvier, and so on, until it reaches the axon terminal. Once it reaches the axon terminal, this electrical signal provokes the release of a chemical message or neurotransmitter that binds to receptors on the adjacent post-synaptic cell (e.g. nerve cell in the CNS or muscle cell in the peripheral nervous system) at specialized regions called synapses.
Furthermore, such movement of the wave of depolarization is much more rapid in myelinated nerve fibers than in unmyelinated fibers, because only the nodes of Ranvier are excited during conduction in myelinated fibers, Na+ (sodium) flux into the nerve is much less than in unmyelinated fibers, where the entire membrane is involved. An example of the advantage of myelination is obtained by comparison of two different nerve fibers, both of which conduct at 25 m/sec at 20°C. The 500-mm diameter unmyelinated giant axon of the squid requires 5,000 times as much energy and occupies about 1,500 times as much space as the 12-mm diameter myelinated nerve in the frog.
In another word, myelin speeds the transmission of electrical impulses called action potentials along myelinated axons by insulating the axon and reducing axonal membrane capacitance. Conduction velocity in myelinated fibers is proportional to the diameter, while in unmyelinated fibers it is proportional to the square root of the diameter 24. Thus, differences in energy and space requirements between the two types of fiber are exaggerated at higher conduction velocities. If nerves were not myelinated and equivalent conduction velocities were maintained, the human spinal cord would need to be as large as a good-sized tree trunk. Myelin, then, facilitates conduction while conserving space and energy 25.
This “insulating” role for myelin is essential for normal motor function (i.e. movement such as walking), sensory function (e.g. hearing, seeing or feeling the sensation of pain) and cognition (e.g. acquiring and recalling knowledge), as demonstrated by the consequences of disorders that affect it, such as the genetically determined leukodystrophies 26, the acquired inflammatory demyelinating disorder, multiple sclerosis 27 and the inflammatory demyelinating peripheral neuropathies 28. Due to its high prevalence, multiple sclerosis, which specifically affects the central nervous system (brain, spinal cord and optic nerve), is the best known disorder of myelin.
Figure 4. Myelin sheath (the Schwann cell has surrounded the nerve axon)
Figure 5. Formation of myelin sheath in the central nervous system (CNS)
Figure 6. Neuron with myelin sheath
Figure 7. How electrical impulses travel down a neuron (myelinated and unmyelinated)
What is the function of myelin?
The main purpose of myelin is to increase the speed at which electrical impulses travel along the myelinated fiber. In unmyelinated fibers, electrical impulses (action potentials) travel as continuous waves, but in myelinated fibers, they “hop” or propagate by saltatory conduction. The latter is markedly faster than the former, at least for axons over a certain diameter. Myelin decreases capacitance and increases electrical resistance across the axonal membrane (the axolemma). It has been suggested that myelin permits larger body size by maintaining agile communication between distant body parts 29.
Myelinated fibers lack voltage-gated sodium channels along the myelinated internodes, exposing them only at the nodes of Ranvier. Here, they are highly abundant and densely packed. Positively charged sodium ions can enter the axon through these voltage-gated channels, leading to depolarisation of the membrane potential at the node of Ranvier. The resting membrane potential is then rapidly restored due to positively charged potassium ions leaving the axon through potassium channels. The sodium ions inside the axon then diffuse rapidly through the axoplasm (axonal cytoplasm), to the adjacent myelinated internode and ultimately to the next (distal) node of Ranvier, triggering the opening of the voltage gated sodium channels and entry of sodium ions at this site. Although, the sodium ions diffuse through the axoplasm rapidly, diffusion is decremental by nature, thus nodes of Ranvier have to be (relatively) closely spaced, to secure action potential propagation 30. The action potential “recharges” at consecutive nodes of Ranvier as the axolemmal membrane potential depolarises to approximately +35 mV 31. Along the myelinated internode, energy-dependent sodium/potassium pumps, pump the sodium ions back out of the axon and potassium ions back into the axon, to restore the balance of ions between the intracellular (inside the cell i.e. axon in this case) and extracellular (outwith the cell) fluids.
Whilst the role of myelin as an “axonal insulator” is well-established, other functions of myelinating cells are less well known or only recently established. The myelinating cell “sculpts” the underlying axon by promoting the phosphorylation of neurofilaments, thus increasing the diameter or thickness of the axon at the internodal regions; helps cluster molecules on the axolemma (such as voltage-gated sodium channels) at the node of Ranvier 32; and modulates the transport of cytoskeletal structures and organelles such as mitochondria, along the axon 22. Recently, evidence came to light to support a role for the myelinating cell in “feeding” the axon 33. In other words, the myelinating cell seems to act as a local ‘fueling station’ for the axon, which uses a great deal of energy to restore the normal balance of ions between it and its environment 34, following the generation of action potentials.
When a peripheral fiber is severed, the myelin sheath provides a track along which regrowth can occur. However, the myelin layer does not ensure a perfect regeneration of the nerve fiber. Some regenerated nerve fibers do not find the correct muscle fibers, and some damaged motor neurons of the peripheral nervous system die without regrowth. Damage to the myelin sheath and nerve fiber is often associated with increased functional insufficiency.
Unmyelinated fibers and myelinated axons of the mammalian central nervous system do not regenerate.
Some studies have revealed that optic nerve fibers can be regenerated in postnatal rats. This regeneration depends upon two conditions: axonal die-back has to be prevented with appropriate neurotrophic factors, and neurite growth inhibitory components have to be inactivated. These studies may lead to further understanding of nerve fiber regeneration in the central nervous system.
Periventricular leukomalacia causes
It is not clear why periventricular leukomalacia occurs 3. Periventricular leukomalacia may happen when the brain receives too little oxygen. The risk for periventricular leucomalacia is higher for babies who are more premature and more unstable at birth. A major cause is thought to be changes in blood flow to the area around the ventricles of the brain. This area is fragile and prone to injury, especially before 32 weeks of gestation. Most babies who develop periventricular leukomalacia are premature, especially those born before 30 weeks gestation (less than 34 weeks gestational age with a median gestational age of 30 weeks and <1500 grams at birth). However, it is not clear when the trigger for periventricular leucomalacia occurs–before, during, or after birth.
Premature babies who have intraventricular hemorrhage (bleeding inside the brain ventricle) are also at increased risk for developing periventricular leukomalacia. Hypotension (low blood pressure), hypoxemia (abnormally low level of oxygen in the blood) and acidosis (a build-up of acid in the bloodstream) may result in ischemic brain injury and periventricular leukomalacia. Marked hypocarbia also known as hypocapnia (a decrease in alveolar and blood carbon dioxide [CO2] levels) in ventilated premature infants has been associated with increased risk of developing periventricular leukomalacia.
Other associated risk factors include the following:
- Placental vascular anastomoses, twin gestation, antepartum hemorrhage
- Chorioamnionitis and funisitis
- Sepsis around the time of delivery may also play a role in causing periventricular leukomalacia.
- Maternal cocaine abuse
The incidence of periventricular leukomalacia ranges from 4-26% in premature infants in neonatal intensive care units (NICUs). The incidence of periventricular leukomalacia is much higher in reports from autopsy studies of premature infants. As many as 75% of premature infants have evidence of periventricular leukomalacia on postmortem examination.
Periventricular leukomalacia likely occurs as a result of hypoxic-ischemic lesions resulting from impaired perfusion at the watershed areas, which in premature infants are located in a periventricular location. It is likely that infection or vasculitis also play a role in pathogenesis.
- Early: periventricular white matter necrosis
- Subacute: cyst formation
- Late: parenchymal loss and enlargement of the ventricles
Infection around the time of delivery may also play a role in causing periventricular leukomalacia. The risk for periventricular leukomalacia is higher for babies who are more premature and more unstable at birth.
Maternal infection, placental inflammation, and vasculitis are also important in the pathogenesis of periventricular leukomalacia. A link between maternal infection, preterm birth, and central nervous system (CNS) injury has been established by epidemiological studies 35. A role for infection and cytokine-induced injury in periventricular leukomalacia is strengthened by studies that demonstrate the presence of tumor necrosis factor in periventricular leukomalacia lesions 36 and in the cerebrospinal fluid (CSF) of infants with cerebral white matter injury 37.
After the initial insult, either ischemia or inflammation, injury to the immature premyelinating oligodendrocytes occurs by either free radical attack or by excitotoxicity. The preterm infant is particularly sensitive to oxygen free radical attack because of delayed development of superoxide dismutase and catalase 38.
In a 2014 report, Inomata et al suggested that combined elevations in serum levels of interleukin (IL) 6 and C-reactive protein (CRP) at birth are predictive of white matter injury in preterm infants with a fetal inflammatory response 39.
Injury to the premyelinating oligodendrocytes results in astrogliosis and microgliosis. This results in a deficit of mature, myelin-producing oligodendrocytes, which leads to cerebral hypomyelination 40.
Premature infants on mechanical ventilation may develop hypocarbia. Several studies have linked hypocarbia, particularly in the first few days of life, with the development of periventricular leukomalacia 41. Cumulative exposure during the first 7 days of life has been shown to independently increase the risk of periventricular leukomalacia in low birth weight infants 42.
Periventricular leukomalacia pathophysiology
The pathophysiology of periventricular leukomalacia is a complex process. Periventricular leukomalacia occurs because of ischemia induced injury to oligodendrocytes in the periventricular area of the developing brain. Cytokine-induced damage following maternal or fetal infection may play a role.
Periventricular leukomalacia is a white matter lesion in premature infants that results from hypotension, ischemia, and coagulation necrosis at the border or watershed zones of deep penetrating arteries of the middle cerebral artery.
Several factors related to vascular development make the periventricular region of the preterm brain uniquely sensitive to ischemic injury. First, early in development, the deep penetrating arteries that supply the watershed zone of the periventricular white matter lack the vascular anastomoses that help maintain perfusion during periods of hypotension. As the fetus matures, the number of anastomoses between the deep penetrating arteries increases, and the periventricular white matter becomes less susceptible to small decreases in blood pressure.
The second vascular developmental factor that plays a role in periventricular leukomalacia is related to cerebral autoregulation. Premature infants have impaired cerebrovascular blood flow autoregulation, increasing their susceptibility to periventricular leukomalacia and intracranial hemorrhage. Decreased blood flow affects the white matter at the superolateral borders of the lateral ventricles. The site of injury affects the descending corticospinal tracts, visual radiations, and acoustic radiations.
Maternal infection, placental inflammation, and vasculitis are also important in the pathogenesis of periventricular leukomalacia. A link between maternal infection, preterm birth, and central nervous system (brain and spinal cord) injury has been established by epidemiological studies 35. A role for infection and cytokine-induced injury in periventricular leukomalacia is strengthened by studies that demonstrate the presence of tumor necrosis factor in periventricular leukomalacia lesions 36 and in the cerebrospinal fluid (CSF) of infants with cerebral white matter injury 37.
After the initial insult, either ischemia or inflammation, injury to the immature premyelinating oligodendrocytes occurs by either free radical attack or by excitotoxicity. The preterm infant is particularly sensitive to oxygen free radical attack because of delayed development of superoxide dismutase and catalase 38.
In a 2014 report, Inomata et al 39 suggested that combined elevations in serum levels of interleukin (IL) 6 and C-reactive protein (CRP) at birth are predictive of white matter injury in preterm infants with a fetal inflammatory response.
Injury to the premyelinating oligodendrocytes results in astrogliosis and microgliosis. This results in a deficit of mature, myelin-producing oligodendrocytes, which leads to cerebral hypomyelination 40.
Premature infants on mechanical ventilation may develop hypocarbia. Several studies have linked hypocarbia, particularly in the first few days of life, with the development of periventricular leukomalacia 41. Cumulative exposure during the first 7 days of life has been shown to independently increase the risk of periventricular leukomalacia in low birth weight infants 42.
Periventricular leukomalacia prevention
Prevention of premature birth is the most important means of preventing periventricular leukomalacia.
Prior to birth, diagnosing and managing chorioamnionitis may prevent periventricular leukomalacia. In 1999, Baud et al 43, 44 reported that betamethasone administered to mothers at 24-31 weeks’ gestation, before delivery, significantly reduced the risk of periventricular leukomalacia, suggesting the possible effect of steroids on fetal inflammatory response.
Avoiding maternal cocaine abuse and avoiding maternal-fetal blood flow alterations has been suggested to minimize periventricular leukomalacia.
Following delivery of a premature infant, attempts to minimize blood pressure swings and hypotension may also be beneficial in preventing periventricular leukomalacia.
Avoidance of prolonged hypocarbia in the mechanically ventilated premature infant may be useful in the prevention of periventricular leukomalacia.
Periventricular leukomalacia symptoms
Periventricular leukomalacia (PVL) occurs most commonly in premature infants born at less than 32 weeks’ gestation who have a birth weight of less than 1500 g. Many of these infants have a history of maternal chorioamnionitis (a serious infection of the placenta, amniotic fluid, and membranes surrounding the fetus during pregnancy, often linked to prolonged rupture of membranes or labor). Most affected infants experience cardiorespiratory problems, such as respiratory distress syndrome or pneumonia, in association with low blood pressure (hypotension) or patent ductus arteriosus (a congenital heart defect where a blood vessel called the ductus arteriosus that connects the aorta and pulmonary artery in the fetus doesn’t close as it should after birth, leading to extra blood flow to the lungs) during their first days of life. Bacterial infection at birth also appears to be a risk factor.
Some children exhibit fairly mild symptoms, while others have significant deficits and disabilities. Periventricular leukomalacia may manifest as cerebral palsy (>50% in the setting of cystic periventricular leukomalacia), intellectual disability or visual disturbance.
Initially, most premature infants are asymptomatic. If symptoms occur, they are usually subtle. Symptoms may include the following:
- Decreased tone in lower extremities
- Increased tone in neck extensors
- Trouble with vision and with eye movements
- Apnea and bradycardia events
- Irritability
- Pseudobulbar palsy with poor feeding
- Clinical seizures (may occur in 10-30% of infants).
- Trouble with movement, and tight muscles
- Developmental delay that is increasingly apparent over time.
Periventricular leukomalacia (PVL) can lead to a variety of problems, including:
- Motor problems: Tight or spastic muscles, difficulty with movement, and an increased risk of cerebral palsy.
- Developmental delays: Problems with cognitive development, learning, and other developmental skills.
- Vision and hearing problems.
Initially, most premature infants are asymptomatic. Periventricular leucomalacia may not be apparent until later months. Each baby may experience symptoms differently. If symptoms occur, they are usually subtle. The most common symptom of periventricular leucomalacia is spastic diplegia, a form of cerebral palsy (a condition that affects coordination and movement) characterized by tight, contracted muscles, especially in the legs. Cerebral palsy is a very variable condition – some children may only have minor movement problems but others may have severe disabilities. Children with periventricular leukomalacia may also have visual problems and/or learning disabilities. The symptoms of periventricular leucomalacia may resemble other conditions or medical problems. Always consult your baby’s doctor for a diagnosis.
Periventricular leukomalacia diagnosis
Tests used to diagnose periventricular leukomalacia include ultrasound and MRI of the head. The traditional diagnostic hallmarks of periventricular leukomalacia are periventricular echodensities or cysts detected by cranial ultrasonography. More recently MRI studies have demonstrated a relatively common diffuse non-cystic form of periventricular leukomalacia in premature infants. Diagnosing periventricular leukomalacia is important because a significant percentage of surviving premature infants develop cerebral palsy (CP), intellectual impairment, or visual disturbances.
Cranial ultrasound
Cranial ultrasound provides a convenient, non-invasive, relatively low-cost screening examination of the hemodynamically-unstable neonate at the bedside. The examination also imparts no radiation exposure. Cranial ultrasonography is sensitive for the detection of hemorrhage, periventricular leukomalacia, and hydrocephalus. Cranial ultrasonography may be performed in the newborn intensive care unit (NICU) without the need to transport fragile infants.
On ultrasound, hyperechoic areas are firstly identified in a distinctive fashion in the periventricular area, more often at the peritrigonal area and in an area anterior and lateral to the frontal horns (periventricular white matter should be less echogenic than the choroid plexus). These are watershed areas that are sensitive to ischemic injury. Initial cranial ultrasonographic findings may be normal in patients who go on to develop clinical and delayed imaging findings of periventricular leukomalacia.
The earliest ultrasonographic appearance of periventricular leukomalacia is abnormal increased echotexture in the periventricular white matter. This is a nonspecific finding that must be differentiated from the normal periventricular halo and mild periventricular edema that may not result in permanent injury.
The abnormal periventricular echotexture of periventricular leukomalacia usually disappears at 2-3 weeks. Approximately 15% of infants experiencing periventricular leukomalacia demonstrate periventricular cysts first appearing at 2-3 weeks after the initial increased echodensities 45. Periventricular leukomalacia (PVL) is characterized by one or more cysts located in the area around the ventricle, and in severe cases even the semi-oval center or the deep subcortical white matter can be involved 46.
Follow-up scans in the more severely affected infants may reveal the development of cysts in these areas, known as cystic periventricular leukomalacia. The severity of periventricular leukomalacia is related to the size and distribution of these cysts. When cystic periventricular leukomalacia is present, it is considered the most predictive sonographic marker for cerebral palsy. However, cystic periventricular leukomalacia may remain apparent for only a few weeks in the periventricular region before the cystic areas coalesce, collapse, and then disappear, often resulting in ventriculomegaly being the visible sequel on cranial ultrasonography 47, 48, 49.
Periventricular leukomalacia classification
One of the methods used for grading of periventricular leukomalacia based on sonographic appearances is as 50:
- Grade 1: Areas of increased periventricular echogenicity without any cyst formation persisting for more than 7 days
- Grade 2: The echogenicity has resolved into small periventricular cysts
- Grade 3: Areas of increased periventricular echogenicity, that develop into extensive periventricular cysts in the occipital and frontoparietal region
- Grade 4: Areas of increased periventricular echogenicity in the deep white matter developing into extensive subcortical cysts.
Cranial magnetic resonance imaging (MRI)
As with CT scanning, MRI does not play a major role in the early evaluation of periventricular leukomalacia. MRI is most helpful in detecting infants with non-cystic periventricular leukomalacia or evaluating infants who develop clinical signs suggestive of periventricular leukomalacia 51.
MRI demonstrates the loss of white matter, abnormal signal intensity of the deep white matter, and ventriculomegaly; it also reveals thinning of the posterior body and splenium of the corpus callosum in severe cases of periventricular leukomalacia.
In a study of MRIs at term-equivalent age from 3 cohorts of 325 very preterm infants, Kidokoro et al found 33% (n=107) had some grade of brain injury (eg, periventricular leukomalacia, intraventricular/cerebellar hemorrhage) and 10% (n=33) had severe brain injury 52. The investigators noted severe brain injury and impaired growth patterns were independently associated with perinatal risk factors and delayed cognitive development 52.
Volumetric MRI scanning is also helpful in determining the extent of injury to the descending corticospinal tracts.
A relationship between the degree of injury to the descending corticospinal tracts as assessed by MRI and the severity of diplegia has been reported.
Other studies
Obtain electroencephalography (EEG) studies in preterm infants for early detection of periventricular leukomalacia. Changes in hypoxic-ischemic encephalopathy and EEG wave patterns may change over time and indicate the severity of the brain injury 53. EEG abnormalities may be apparent before anomalies seen on ultrasonography.
Spectral-domain optical coherence tomography (SD-OCT) shows promise in the evaluation of prematurity on early optic nerve development and of central nervous system development and anomalies 54.
On histologic examination, periventricular leukomalacia lesions demonstrate widespread loss of oligodendrocytes and an increase in astrocytes.
Periventricular leukomalacia treatment
Currently there are no treatments for periventricular leucomalacia. Premature babies’ heart, lung, intestine, and kidney functions are watched closely and treated in the newborn intensive care unit (NICU). This helps reduce the risk of developing periventricular leukomalacia.
Management of the problems that can result from periventricular leucomalacia will be determined by your baby’s doctor based on:
- Your baby’s gestational age, overall health, and medical history
- Extent of the condition
- Your baby’s tolerance for specific medications, procedures, or therapies
- Expectations for the course of the condition
- Your opinion or preference
Babies at risk for periventricular leucomalacia may need special care and close neurodevelopmental follow-up after discharge from the hospital. Follow-up may include pediatricians, neurologists, developmental specialists, physical therapists, occupational therapists, and speech therapists. Free radical scavengers are being investigated to determine if they have a role in preventing oligodendrocyte injury in periventricular leukomalacia.
Developmental follow-up
Premature infants with evidence of periventricular leukomalacia (PVL) require close developmental follow-up because of the high association with cerebral palsy (CP) 55.
Early intervention strategies carried out by occupational therapists or physical therapists may decrease symptoms and may increase the infant’s motor function.
Periventricular leukomalacia prognosis
Periventricular leukomalacia occurs most commonly in premature infants born at less than 32 weeks’ gestation. The long-term prognosis (outlook) for babies with periventricular leukomalacia depends upon the severity of the initial brain damage 1, 2. The types of symptoms vary widely and can range from a child who has minimal problems to a child who has severe delays and problems with movement. Periventricular leukomalacia often leads to nervous system and developmental problems in growing babies. These problems most often occur during the first to second year of life. Many infants with periventricular leukomalacia later develop signs of cerebral palsy (CP), especially tightness or increased muscle tone (spasticity) in the legs 56, 57, 58.
Mild periventricular leukomalacia is often associated with spastic diplegia, a type of cerebral palsy that primarily affects the legs causing muscle stiffness and difficulty with movement, often leading to a “scissoring” gait where legs cross at the knees. Severe periventricular leukomalacia is associated with quadriplegia also known as tetraplegia is paralysis that affects all four limbs. Severe periventricular leukomalacia is also associated with a higher incidence of intelligence deficiencies and visual disturbances.
Babies with periventricular leukomalacia are at risk for major nervous system problems. These are likely to include movements such as sitting, crawling, walking, and moving the arms. Fixation difficulties, nystagmus, strabismus, and blindness have also been associated with periventricular leukomalacia. These babies may need physical therapy. Extremely premature babies may have more problems with learning than with movement.
Varying degrees of intellectual impairment, developmental impairment, or both have been reported in association with periventricular leukomalacia. Fixation difficulties, nystagmus, strabismus, and blindness have been associated with periventricular leukomalacia. Some cases of visual dysfunction in association with periventricular leukomalacia occur in the absence of retinopathy of prematurity, suggesting damage to optic radiations as causation.
A baby who is diagnosed with periventricular leukomalacia should be monitored by a developmental pediatrician or a pediatric neurologist. The child should see the regular pediatrician for scheduled exams.
- Song J, Yue Y, Sun H, Cheng P, Xu F, Li B, Li K, Zhu C. Clinical characteristics and long-term neurodevelopmental outcomes of leukomalacia in preterm infants and term infants: a cohort study. J Neurodev Disord. 2023 Aug 7;15(1):24. doi: 10.1186/s11689-023-09489-7[
][
]
- Gotardo JW, Volkmer NFV, Stangler GP, Dornelles AD, Bohrer BBA, Carvalho CG. Impact of peri-intraventricular haemorrhage and periventricular leukomalacia in the neurodevelopment of preterms: A systematic review and meta-analysis. PLoS ONE. 2019;14(10):e0223427. doi: 10.1371/journal.pone.0223427[
][
]
- Abiramalatha T, Ramaswamy VV, Ponnala AK, Kallem VR, Murkunde YV, Punnoose AM, et al. Emerging neuroprotective interventions in periventricular leukomalacia – A systematic review of preclinical studies. Expert Opin Investig Drugs. 2022;31(3):305–330. doi: 10.1080/13543784.2022.2040479[
][
]
- Banker BQ, Larroche JC. Periventricular leukomalacia of infancy. A form of neonatal anoxic encephalopathy. Arch Neurol. 1962;7:386–410. doi: 10.1001/archneur.1962.04210050022004[
]
- Suzuki S. Case Report: Peripartum periventricular leukomalacia resulting in cerebral palsy associated with placenta previa in Japan. F1000Res. 2024 Aug 6;9:241. doi: 10.12688/f1000research.22878.[
]
- Starr R, De Jesus O, Shah SD, et al. Periventricular and Intraventricular Hemorrhage. [Updated 2023 Aug 23]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK538310[
]
- Ahya KP, Suryawanshi P. Neonatal periventricular leukomalacia: current perspectives. Res Rep Neonatol. 2018;8:1–8.[
]
- Brodsky MC. Periventricular leukomalacia: an intracranial cause of pseudoglaucomatous cupping. Arch Ophthalmol. 2001;119(4):626–662. doi: 10.1001/archopht.119.4.626[
]
- Brodsky MC. Semiology of periventricular leucomalacia and its optic disc morphology. Br J Ophthalmol. 2003;87(11):1309–1310. doi: 10.1136/bjo.87.11.1309[
]
- Lasry O, Shevell MI, Dagenais L REPACQ Consortium. Cross-sectional comparison of periventricular leukomalacia in preterm and term children. Neurology. 2010;74:1386–91. doi: 10.1212/WNL.0b013e3181dad62d[
]
- Miller SP, Shevell MI, Patenaude Y, O’Gorman AM. Neuromotor spectrum of periventricular leukomalacia in children born at term. Pediatr Neurol. 2000;23:155–9. doi: 10.1016/s0887-8994(00)00172-7[
]
- Deguchi K, Oguchi K, Matsuura N, Armstrong DD, Takashima S. Periventricular leukomalacia:Relation to gestational age and axonal injury. Pediatr Neurol. 1999;20:370–4. doi: 10.1016/s0887-8994(99)00010-7[
]
- Ali A, Yalçın R, Ünlüer-Gümüştaş A. Cranial MR characteristics of Cerebral Palsy cases and correlation of findings with clinical results. Turk J Pediatr. 2019;61(4):525-537. doi: 10.24953/turkjped.2019.04.009[
]
- Horber V, Sellier E, Horridge K, Rackauskaite G, Andersen GL, Virella D, Ortibus E, Dakovic I, Hensey O, Radsel A, Papavasiliou A, Cruz De la J, Arnaud C, Krägeloh-Mann I, Himmelmann K. The Origin of the Cerebral Palsies: Contribution of Population-Based Neuroimaging Data. Neuropediatrics. 2020 Apr;51(2):113-119. doi: 10.1055/s-0039-3402007[
]
- Marefi A, Husein N, Dunbar M, Dewey D, Letourneau N, Oskoui M, et al. Risk factors for term-born periventricular white matter injury in children with cerebral palsy: A case-control study. Neurology. 2022;99(22):e2485–e2493. doi: 10.1212/WNL.0000000000201274[
]
- Shang Q, Ma CY, Lv N, Lv ZL, Yan YB, Wu ZR, et al. Clinical study of cerebral palsy in 408 children with periventricular leukomalacia. Exp Ther Med. 2015;9(4):1336–1344. doi: 10.3892/etm.2015.2222[
]
- Periventricular Leukomalacia Imaging. https://emedicine.medscape.com/article/416016-overview[
]
- Pediatric Periventricular Leukomalacia. https://emedicine.medscape.com/article/975728-overview#a5[
]
- Pediatric Periventricular Leukomalacia. https://emedicine.medscape.com/article/975728-overview#a6[
]
- Füsun Korkmaz Alehan. Value of neuroimaging in the evaluation of neurologically normal children with recurrent headache. J Child Neurol. November 17, 2002; (11):807-809. http://www.ncbi.nlm.nih.gov/pubmed/12585718[
]
- Todd J. Schwedt, Yifan Guo, David Rothner. “Benign” imaging abnormalities in children and adolescents with headache. Headache. March 21, 2006; 46(3):387-398. http://www.ncbi.nlm.nih.gov/pubmed/16618255[
]
- Stassart RM, Möbius W, Nave K-A, Edgar JM. The Axon-Myelin Unit in Development and Degenerative Disease. Frontiers in Neuroscience. 2018;12:467. doi:10.3389/fnins.2018.00467. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6050401/[
][
]
- Waxman S G , Ritchie J M . Molecular dissection of the myelinated axon. Ann. Neurol. 1993;33:121–136.[
]
- Morell P, Quarles RH. The Myelin Sheath. In: Siegel GJ, Agranoff BW, Albers RW, et al., editors. Basic Neurochemistry: Molecular, Cellular and Medical Aspects. 6th edition. Philadelphia: Lippincott-Raven; 1999. Available from: https://www.ncbi.nlm.nih.gov/books/NBK27954[
]
- Ritchie, J. M. Physiological basis of conduction in myelinated nerve fibers. In P. Morell (ed.), Myelin, 2nd ed. New York: Plenum, 1984, pp. 117–141.[
]
- Van der Knaap MS, Bugiani M. Leukodystrophies: a proposed classification system based on pathological changes and pathogenetic mechanisms. Acta Neuropathologica. 2017;134(3):351-382. doi:10.1007/s00401-017-1739-1. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5563342/[
]
- Multiple sclerosis. Lancet. 2008 Oct 25;372(9648):1502-17. doi: 10.1016/S0140-6736(08)61620-7. https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(08)61620-7/fulltext[
]
- Chronic inflammatory demyelinating polyneuropathy. Curr Opin Neurol. 2017 Oct;30(5):508-512. doi: 10.1097/WCO.0000000000000481. https://www.ncbi.nlm.nih.gov/pubmed/28763304[
]
- What is myelin? Neuron Glia Biol. 2008 May;4(2):153-63. doi: 10.1017/S1740925X09990263. https://doi.org/10.1017/S1740925X09990263[
]
- Raine CS (1999). “Characteristics of Neuroglia”. In Siegel GJ, Agranoff BW, Albers RW, Fisher SK, Uhler MD. Basic Neurochemistry: Molecular, Cellular and Medical Aspects (6th ed.). Philadelphia: Lippincott-Raven.[
]
- Saladin KS (2012). Anatomy & physiology: the unity of form and function (6th ed.). New York, NY: McGraw-Hill[
]
- Brivio V, Faivre-Sarrailh C, Peles E, Sherman DL, Brophy PJ. Assembly of CNS Nodes of Ranvier in Myelinated Nerves Is Promoted by the Axon Cytoskeleton. Current Biology. 2017;27(7):1068-1073. doi:10.1016/j.cub.2017.01.025. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5387178/[
]
- Funfschilling U, Supplie LM, Mahad D, et al. Glycolytic oligodendrocytes maintain myelin and long-term axonal integrity. Nature. 2012;485(7399):517-521. doi:10.1038/nature11007. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3613737/[
]
- Engl E, Attwell D. Non‐signalling energy use in the brain. The Journal of Physiology. 2015;593(16):3417-3429. doi:10.1113/jphysiol.2014.282517. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4560575/[
]
- Perlman JM, Risser R, Broyles RS. Bilateral cystic periventricular leukomalacia in the premature infant: associated risk factors. Pediatrics. June 1996. 97:822-7.[
][
]
- Kadhim H, Tabarki B, Verellen G, et al. Inflammatory cytokines in the pathogenesis of periventricular leukomalacia. Neurology. 2001 May 22. 56(10):1278-84.[
][
]
- Ellison VJ, Mocatta TJ, Winterbourn CC, et al. The relationship of CSF and plasma cytokine levels to cerebral white matter injury in the premature newborn. Pediatr Res. 2005 Feb. 57(2):282-6.[
][
]
- Khwaja O, Volpe JJ. Pathogenesis of cerebral white matter injury of prematurity. Arch Dis Child Fetal Neonatal Ed. 2008 Mar. 93(2):F153-61.[
][
]
- Inomata K, Mizobuchi M, Tanaka S, et al. Patterns of increases in interleukin-6 and C-reactive protein as predictors for white matter injury in preterm infants. Pediatr Int. 2014 Dec. 56(6):851-5.[
][
]
- Volpe JJ, Kinney HC, Jensen FE, Rosenberg PA. The developing oligodendrocyte: key cellular target in brain injury in the premature infant. Int J Dev Neurosci. 2011 Jun. 29(4):423-40.[
][
]
- Okumura A, Hayakawa F, Kato T, et al. Hypocarbia in preterm infants with periventricular leukomalacia: the relation between hypocarbia and mechanical ventilation. Pediatrics. 2001 Mar. 107(3):469-75.[
][
]
- Resch B, Neubauer K, Hofer N, Resch E, Maurer U, Haas J, et al. Episodes of hypocarbia and early-onset sepsis are risk factors for cystic periventricular leukomalacia in the preterm infant. Early Hum Dev. 2012 Jan. 88(1):27-31.[
][
]
- Baud O, Foix-L’Helias L, Kaminski M, et al. Antenatal glucocorticoid treatment and cystic periventricular leukomalacia in very premature infants. N Engl J Med. 1999 Oct 14. 341(16):1190-6.[
]
- Canterino JC, Verma U, Visintainer PF, et al. Antenatal steroids and neonatal periventricular leukomalacia. Obstet Gynecol. 2001 Jan. 97(1):135-9.[
]
- Hayakawa F, Okumura A, Kato T, Kuno K, Watanabe K. Determination of timing of brain injury in preterm infants with periventricular leukomalacia with serial neonatal electroencephalography. Pediatrics. 1999 Nov;104(5 Pt 1):1077-81. doi: 10.1542/peds.104.5.1077[
]
- Back SA. White matter injury in the preterm infant: pathology and mechanisms. Acta Neuropathol. 2017;134(3):331–349. doi: 10.1007/s00401-017-1718-6[
]
- Dubowitz LM, Bydder GM, Mushin J. Developmental sequence of periventricular leukomalacia. Correlation of ultrasound, clinical, and nuclear magnetic resonance functions. Arch Dis Child. 1985 Apr;60(4):349-55. https://pmc.ncbi.nlm.nih.gov/articles/instance/1777237/pdf/archdisch00723-0067.pdf[
]
- Burdjalov V, Srinivasan P, Baumgart S, Spitzer AR. Handheld, portable ultrasound in the neonatal intensive care nursery: a new, inexpensive tool for the rapid diagnosis of common neonatal problems. J Perinatol. 2002 Sep;22(6):478-83. doi: 10.1038/sj.jp.7210782[
]
- Pierrat V, Duquennoy C, van Haastert IC, Ernst M, Guilley N, de Vries LS. Ultrasound diagnosis and neurodevelopmental outcome of localised and extensive cystic periventricular leucomalacia. Arch Dis Child Fetal Neonatal Ed. 2001 May;84(3):F151-6. https://pmc.ncbi.nlm.nih.gov/articles/instance/1721251/pdf/v084p0F151.pdf[
]
- Neonatal Hypoxic-Ischemic Encephalopathy: Multimodality Imaging Findings. Christine P. Chao, Christopher G. Zaleski, and Alice C. Patton. RadioGraphics 2006 26:suppl_1, S159-S172.[
]
- Tzarouchi LC, Astrakas LG, Zikou A, et al. Periventricular leukomalacia in preterm children: assessment of grey and white matter and cerebrospinal fluid changes by MRI. Pediatr Radiol. 2009 Dec. 39(12):1327-32.[
]
- Kidokoro H, Anderson PJ, Doyle LW, Woodward LJ, Neil JJ, Inder TE. Brain injury and altered brain growth in preterm infants: predictors and prognosis. Pediatrics. 2014 Aug. 134(2):e444-53.[
][
]
- Volpe JJ. Hypoxic-ischemic encephalopathy. Neurology of the Newborn. 5th ed. Philadelphia, PA: Saunders-Elsevier; 2008. Volume 899: chapter 9.[
]
- Tong AY, El-Dairi M, Maldonado RS, et al. Evaluation of optic nerve development in preterm and term infants using handheld spectral-domain optical coherence tomography. Ophthalmology. 2014 Sep. 121(9):1818-26.[
]
- Ment LR, Bada HS, Barnes P, Grant PE, Hirtz D, Papile LA, Pinto-Martin J, Rivkin M, Slovis TL. Practice parameter: neuroimaging of the neonate: report of the Quality Standards Subcommittee of the American Academy of Neurology and the Practice Committee of the Child Neurology Society. Neurology. 2002 Jun 25;58(12):1726-38. doi: 10.1212/wnl.58.12.1726[
]
- Tüzün F, Gencpınar P, Ozbal S, Dilek M, Ergur BU, Duman N, Ozkan H, Kumral A. Neuroprotective effect of neotrofin in a neonatal rat model of periventricular leukomalacia. Neurosci Lett. 2012;520(1):6–10. doi: 10.1016/j.neulet.2012.04.076[
]
- Blumenthal I. Periventricular leucomalacia: A review. Eur J Pediatr. 2004;163(8):435–442. doi: 10.1007/s00431-004-1477-y[
]
- Sarkar S, Shankaran S, Barks J, Do BT, Laptook AR, Das A, Ambalavanan N, Van Meurs KP, Bell EF, Sanchez PJ, Hintz SR, Wyckoff MH, Stoll BJ, Carlo WA; Eunice Kennedy Shriver National Institute of Child Health and Human Development Neonatal Research Network. Outcome of Preterm Infants with Transient Cystic Periventricular Leukomalacia on Serial Cranial Imaging Up to Term Equivalent Age. J Pediatr. 2018 Apr;195:59-65.e3. doi: 10.1016/j.jpeds.2017.12.010[
]