Vitamin E
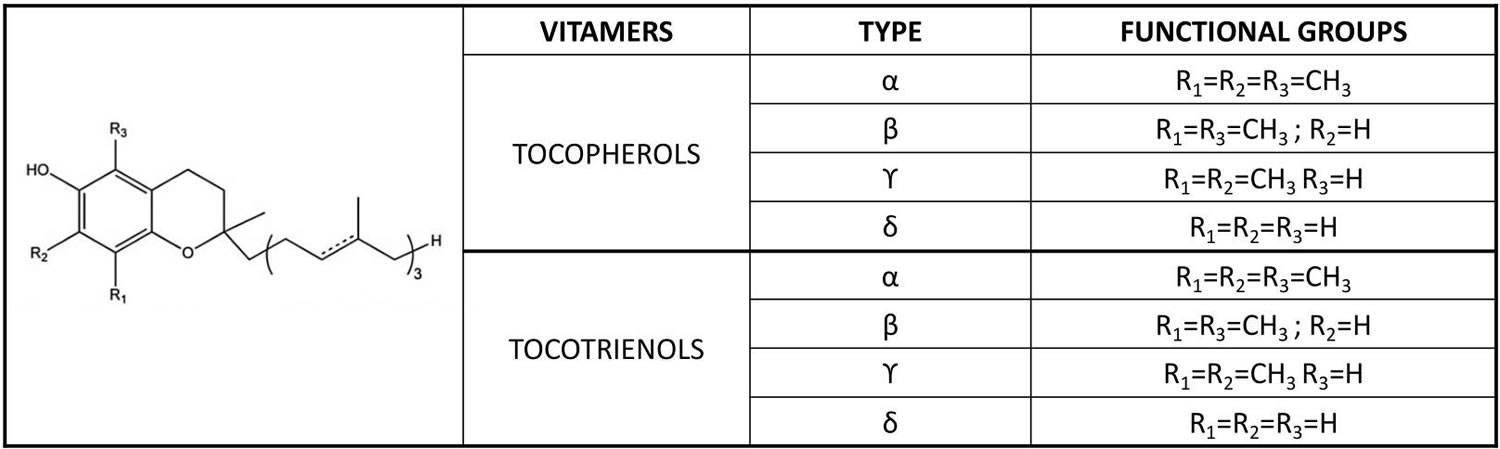
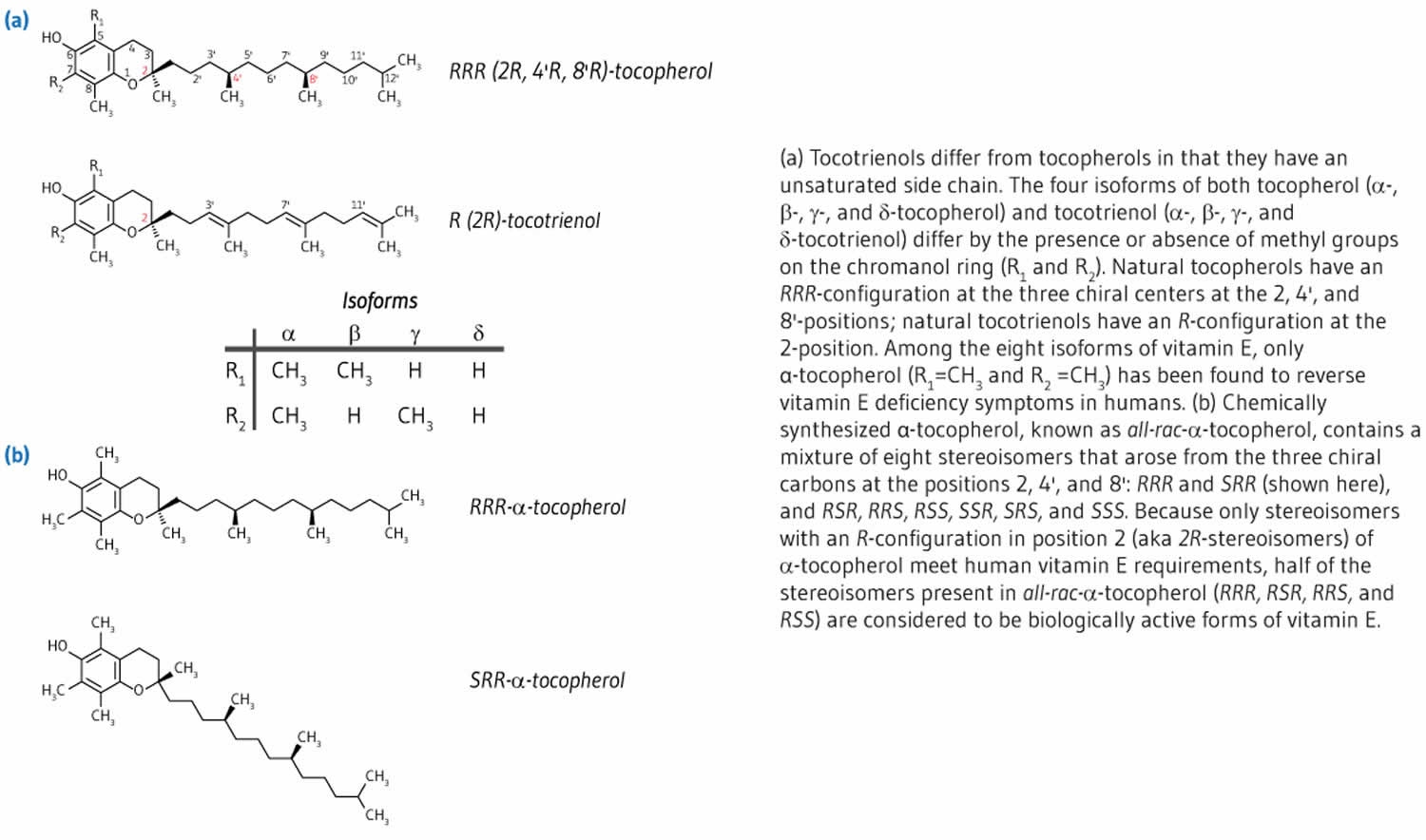
Vitamin E is a fat-soluble nutrient found in many foods, added to others, and available as a dietary supplement. “Vitamin E” is the collective name for a group of fat-soluble compounds with distinctive antioxidant activities 1. Naturally occurring vitamin E exists in eight chemical forms: four tocopherol isoforms (alpha-, beta-, gamma-, and delta-tocopherol OR α-, β-, γ-, and δ-tocopherol) and four tocotrienol isoforms (alpha-, beta-, gamma-, and delta-tocotrienol OR α-, β-, γ-, and δ-tocotrienol) that have varying levels of biological activity (see Figure 2) 1, 2. Alpha- (or α-) tocopherol is the only form that is recognized to meet human requirements and the body preferentially uses alpha-tocopherol, and only α-tocopherol supplementation can reverse vitamin E deficiency symptoms 3, 4, 5, 6, 7.
In the human liver, alpha-tocopherol (α-tocopherol) is the form of vitamin E that is preferentially bound to alpha-tocopherol transfer protein (α-TTP) and incorporated into lipoproteins that transport alpha-tocopherol (α-tocopherol) in the blood for delivery to tissues outside your liver 6. Therefore, alpha-tocopherol (α-tocopherol) is the predominant form of vitamin E found in your blood and tissues 8. In addition, alpha-tocopherol (α-tocopherol) appears to be the form of vitamin E with the greatest nutritional significance. Natural alpha-tocopherol (α-tocopherol) made by plants found in food has an RRR-configuration at the 2, 4’, and 8’-position of the alpha-tocopherol molecule wrongly referred to as d-α-tocopherol 6. Chemically synthesized all-racemic-α-tocopherol (all-rac-α-tocopherol) incorrectly labeled dl-α-tocopherol is a mixture of eight stereoisomers of alpha-tocopherol (α-tocopherol), which arose from the three chiral carbons at the 2, 4’, and 8’-positions: RRR-, RSR-, RRS-, RSS-, SRR-, SSR-, SRS-, and SSS-α-tocopherol (see Figure 2) 6. While all vitamin E stereoisomers have equal in vitro (test tube studies) antioxidant activity, only the forms in the R-conformation at position 2 (noted 2R) meet the vitamin E requirements in humans 9. Furthermore, beta-, gamma-, and delta-tocopherols, 4 tocotrienols, and several stereoisomers may also have important biologic activity 8.
In your body, vitamin E acts a fat-soluble antioxidant, helping to protect your cells from the damage caused by free radicals, which are molecules that contain an unshared electron. Unshared electrons are highly energetic and react rapidly with oxygen to form reactive oxygen species (ROS) 7. Free radicals are compounds formed when your body converts the food you eat into energy. People are also exposed to free radicals in the environment from cigarette smoke, air pollution, and ultraviolet radiation from the sun. Free radicals damage cells and might contribute to the development of cardiovascular disease and cancer 10. Vitamin E is believed to serve as a chain-breaking antioxidant that stops the oxidative degradation of fats, thus preventing free radical production and harm to the cell. Scientists are investigating whether, by limiting free-radical production and possibly through other mechanisms, vitamin E might help prevent or delay the chronic diseases associated with free radicals.
In addition to its activities as an antioxidant, vitamin E is involved in immune function and, as shown primarily by in vitro studies (test tube lab studies) of cells, cell signaling, regulation of gene expression, and other metabolic processes 8. Alpha-tocopherol inhibits the activity of protein kinase C, an enzyme involved in cell proliferation and differentiation in smooth muscle cells, platelets, and monocytes 9. Vitamin-E–packed endothelial cells lining the interior surface of blood vessels are better able to resist blood-cell components adhering to this surface. Vitamin E also increases the expression of two enzymes that suppress arachidonic acid metabolism, thereby increasing the release of prostacyclin from the endothelium, which, in turn, dilates blood vessels and inhibits platelet aggregation 9.
Your body also needs vitamin E to boost its immune system so that it can fight off invading bacteria and viruses. It helps to widen blood vessels and keep blood from clotting within them. In addition, cells use vitamin E to interact with each other and to carry out many important functions. Scientists are investigating whether, by limiting free-radical production and possibly through other mechanisms, vitamin E might help prevent or delay the chronic diseases associated with free radicals.
Aside from maintaining the integrity of cell membranes throughout the body, alpha tocopherol protects the fats in low-density lipoproteins (LDLs) from oxidation 11. Lipoproteins are particles composed of lipids and proteins that transport fats through the bloodstream. LDLs (bad cholesterol) specifically transport cholesterol from the liver to the tissues of the body. Oxidized LDLs (bad cholesterol) have been implicated in the development of cardiovascular disease 12.
Vitamin E is absorbed in the intestinal lumen, which is dependent upon various factors such as pancreatic secretions, micelle formation, and most importantly, chylomicron secretions. Chylomicron secretion is necessary for vitamin E absorption. Vitamin E is found in sunflower seeds, nuts, some oils, spinach, butternut squash, and many other food sources. Vitamin E deficiency has been linked to peripheral neuropathy in addition to spinocerebellar ataxia, skeletal myopathy and pigmented retinopathy. Interestingly, studies have reported vitamin E level in association to the development of cataracts 9.
Serum concentrations of vitamin E (alpha-tocopherol) depend on your liver, which takes up the nutrient after the various forms are absorbed from the small intestine. Yet, in the body, alpha-tocopherol (α-tocopherol) is preferentially retained in the liver by the binding to alpha-tocopherol transfer protein (α-TTP) 1, which incorporates α-tocopherol into lipoproteins for delivery to extrahepatic tissues; and other forms of vitamin E other than alpha-tocopherol (α-tocopherol) are actively broken down and excreted 13. As a result, blood and cellular concentrations of other forms of vitamin E are lower than those of alpha-tocopherol and have been the subjects of less research 14, 15. Plasma tocopherol levels vary with total plasma lipid levels. Normally, the plasma alpha-tocopherol level is 5 to 20 mcg/mL (11.6 to 46.4 mcmol/L) 16.
Vitamin E is safe for pregnancy and breastfeeding. Both vitamin K and omega-6 fatty acids requirements may increase with high doses of vitamin E.
Some food and dietary supplement labels still list vitamin E in International Units (IUs) rather than milligrams (mg). 1 IU of the natural form of vitamin E is equivalent to 0.67 mg. 1 IU of the synthetic form of vitamin E is equivalent to 0.45 mg.
International Units and Milligrams
Vitamin E is listed on the new Nutrition Facts and Supplement Facts labels in milligrams (mg) 17. The U.S. Food and Drug Administration (FDA) required manufacturers to use these new labels starting in January 2020, but companies with annual sales of less than $10 million may continue to use the old labels that list vitamin E in international units (IUs) until January 2021 18. Conversion rules are as follows:
To convert from mg to IU:
- 1 mg of alpha-tocopherol is equivalent to 1.49 IU of the natural form or 2.22 IU of the synthetic form.
To convert from IU to mg:
- 1 IU of the natural form is equivalent to 0.67 mg of alpha-tocopherol.
- 1 IU of the synthetic form is equivalent to 0.45 mg of alpha-tocopherol.
For example, 15 mg of natural alpha-tocopherol would equal 22.4 IU (15 mg x 1.49 IU/mg = 22.4 IU). The corresponding value for synthetic alpha-tocopherol would be 33.3 IU (15 mg x 2.22 IU/mg).
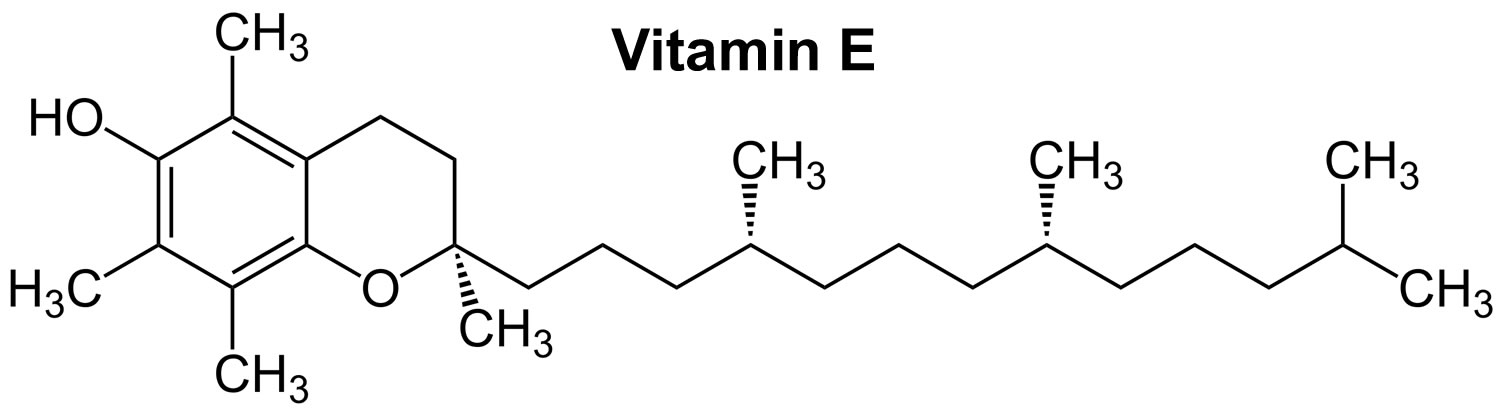
Figure 1. Vitamin E chemical structure
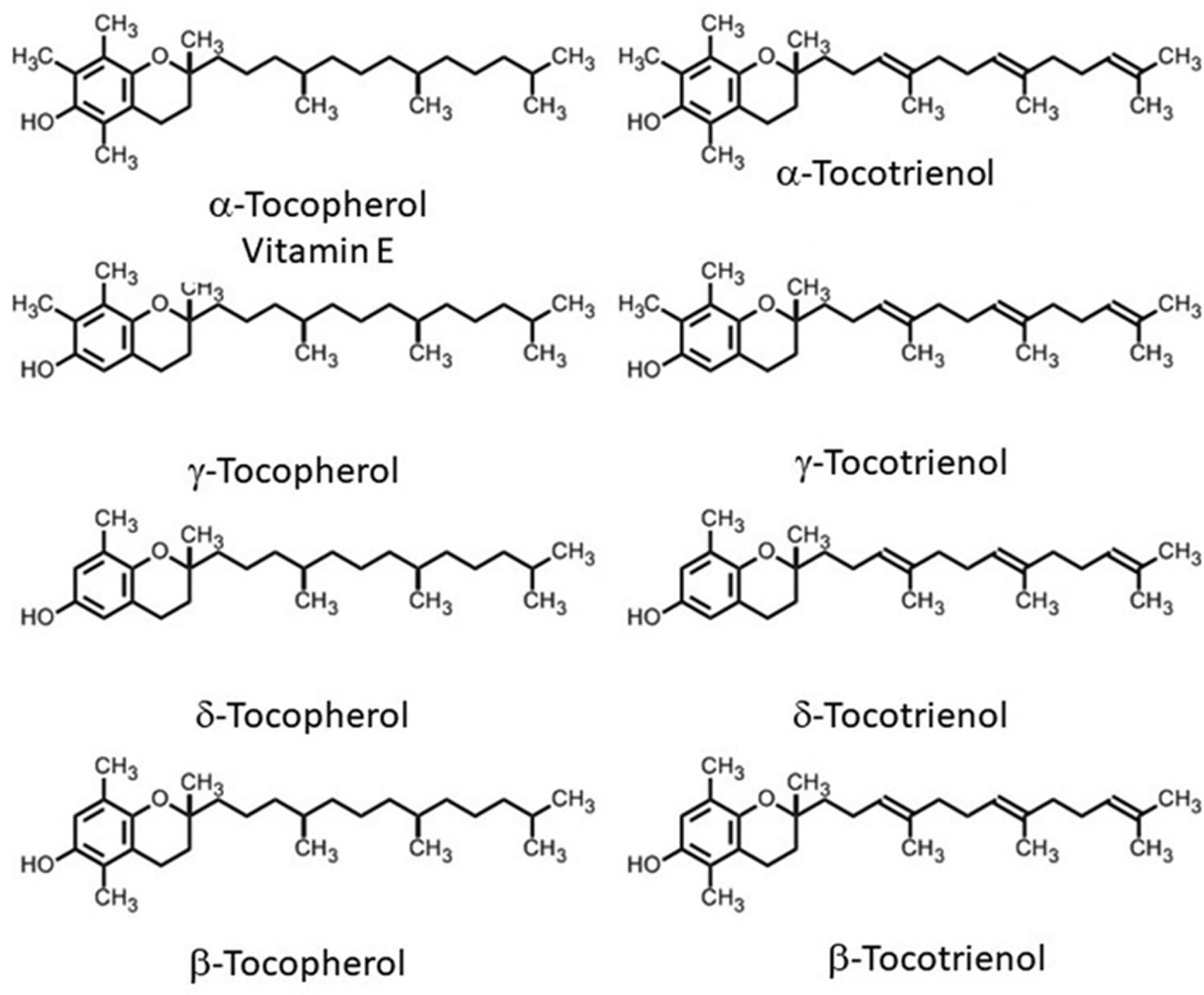
Figure 2. Vitamin E chemical structures
 Footnote: The differences in the α, β, γ and δ forms are in the number and position of the methyl groups on the chromanol ring. Only the β- and γ- forms of tocopherols or tocotrienols can be called isomers, having the same formula but a different arrangement of atoms in the molecule. The difference between the tocopherols and the tocotrienols is in the presence of three double bonds in the side chain of the latter.
[Source 4 ]
Footnote: The differences in the α, β, γ and δ forms are in the number and position of the methyl groups on the chromanol ring. Only the β- and γ- forms of tocopherols or tocotrienols can be called isomers, having the same formula but a different arrangement of atoms in the molecule. The difference between the tocopherols and the tocotrienols is in the presence of three double bonds in the side chain of the latter.
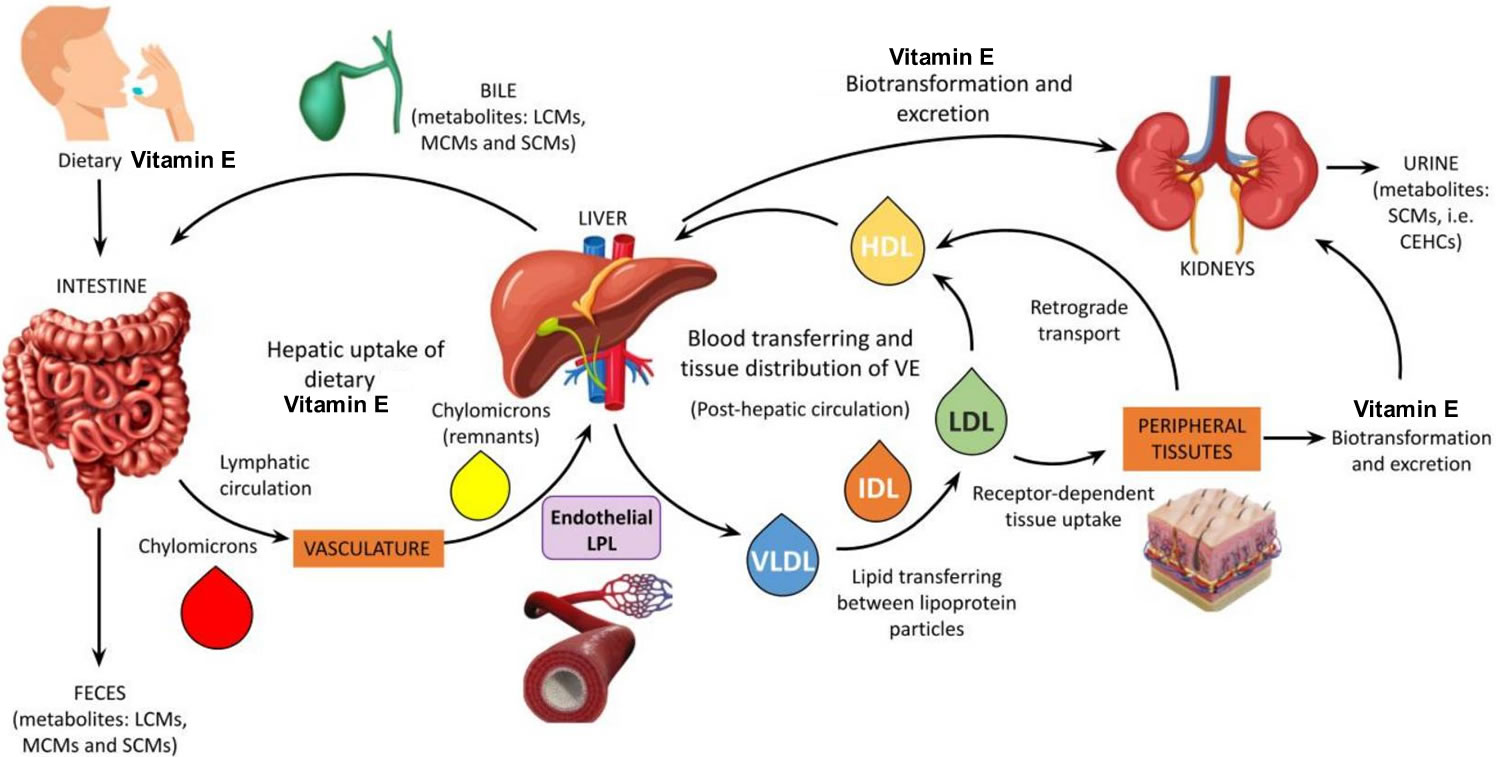
[Source 4 ]Figure 3. Vitamin E metabolism
Footnote: Schematic representation of vitamin E metabolism. The mechanism of vitamin E digestion and uptake into intestinal cells (enterocytes) is unclear but requires bile acids and pancreatic enzymes, and the packaging along with dietary fat into chylomicrons. The efficiency of vitamin E absorption increases with the amount of fat in ingested food, such that vitamin E absorption from supplements is likely to be minimal with low-fat meals 19, 20. In the circulation, all lipoproteins (i.e., VLDLs, LDLs, and HDLs) are involved in the transport and tissue distribution of alpha-tocopherol 8. Increased concentrations of fats (cholesterol and triglycerides) in the blood have been correlated to higher serum alpha-tocopherol concentrations. However, if a high blood concentration of fats is associated with a slower turnover of lipoproteins, then the distribution of alpha-tocopherol to tissues may be substantially altered 21.
Abbreviations: LCMs = long-chain-metabolites; MCMs = multi-cycling metabolites; SCMs = short-chain-metabolites; CEHCs = carboxyethyl hydroxychromans (natural vitamin E metabolites); VE = vitamin E; LPL = lipoprotein lipase.
[Source 22 ]What does Vitamin E do?
Vitamin E is a fat-soluble antioxidant that stops the production of reactive oxygen species (ROS) formed when fat undergoes oxidation. Scientists are investigating whether, by limiting free-radical production and possibly through other mechanisms, vitamin E might help prevent or delay the chronic diseases associated with free radicals.
Antioxidants protect cells from the damaging effects of free radicals, which are molecules that contain an unshared electron. Free radicals damage cells and might contribute to the development of cardiovascular disease and cancer 10. Unshared electrons are highly energetic and react rapidly with oxygen to form reactive oxygen species. The body forms reactive oxygen species endogenously when it converts food to energy, and antioxidants might protect cells from the damaging effects of reactive oxygen species. The body is also exposed to free radicals from environmental exposures, such as cigarette smoke, air pollution, and ultraviolet radiation from the sun. Reactive oxygen species are part of signaling mechanisms among cells.
The body also needs vitamin E to boost its immune system so that it can fight off invading bacteria and viruses. It helps to widen blood vessels and keep blood from clotting within them.
In addition to vitamin E activities as an antioxidant, vitamin E is involved in immune function and, as shown primarily by in vitro studies of cells, cell signaling, regulation of gene expression, and other metabolic processes 1, 23, 24. Alpha-tocopherol inhibits the activity of protein kinase C, an enzyme involved in cell proliferation and differentiation in smooth muscle cells, platelets, and monocytes 25. Vitamin-E–replete endothelial cells lining the interior surface of blood vessels are better able to resist blood-cell components adhering to this surface. Vitamin E also increases the expression of two enzymes that suppress arachidonic acid metabolism, thereby increasing the release of prostacyclin from the endothelium, which, in turn, dilates blood vessels and inhibits platelet aggregation 25. Moreover, vitamin E also helps improve nerve conduction 26, maintain the structural integrity of the hemoglobin membrane 27 and, along with vitamin A, plays a role in vision 28. The specific mechanism of action for most of vitamin E effects is still relatively unknown 29.
Vitamin E inhibits platelet adhesion by preventing oxidative changes to low-density lipoprotein (LDL) cholesterol also called bad cholesterol and inhibition of platelet aggregation by reducing prostaglandin E2. Another effect is inhibiting protein kinase C causing smooth-muscle proliferation.
Even though research has shown that vitamin E assists with the prevention of heart disease and atherosclerosis it has not been approved for this use by the United States Food and Drug Administration (FDA).
Antioxidant activity
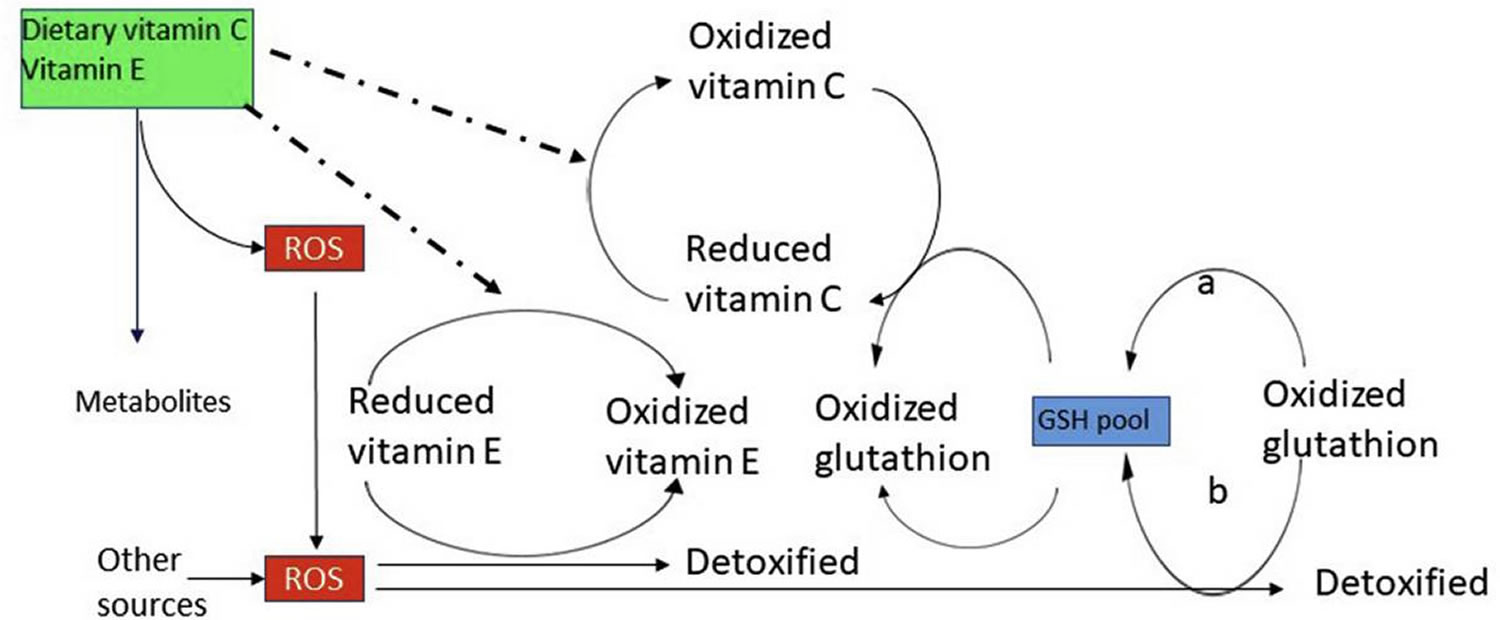
The main function of alpha-tocopherol in humans is that of a fat-soluble antioxidant. Fats, which are an integral part of all cell membranes, are vulnerable to damage through lipid peroxidation by free radicals. Alpha-tocopherol is uniquely suited to intercept peroxyl radicals and thus prevent a chain reaction of lipid oxidation (Figure 4). When a molecule of α-tocopherol neutralizes a free radical, it is oxidized and its antioxidant capacity is lost. Other antioxidants, such as vitamin C, are capable of regenerating the antioxidant capacity of α-tocopherol (Figure 4) 8.
Aside from maintaining the integrity of cell membranes throughout the body, α-tocopherol protects the fats in low-density lipoproteins (LDLs) from oxidation. Lipoproteins are particles composed of lipids and proteins that transport fats through the bloodstream. LDLs specifically transport cholesterol from the liver to the tissues of the body. Oxidized LDLs have been implicated in the development of cardiovascular disease 12.
Tocotrienols and gamma-tocopherol are thought to be better scavengers of peroxyl radicals and reactive nitrogen species, respectively, than alpha-tocopherol 30. Yet, in the body, alpha-tocopherol is preferentially retained in the liver by the binding to alpha-tocopherol transfer protein (α-TTP), which incorporates α-tocopherol into lipoproteins for delivery to extrahepatic tissues; and forms of vitamin E other than α-tocopherol are actively metabolized and excreted. Hence, while gamma-tocopherol is the most common form of vitamin E in the American diet 31, its plasma and tissue concentrations are generally significantly lower than those of alpha-tocopherol and more gamma-tocopherol is excreted in urine than alpha-tocopherol, suggesting less gamma-tocopherol is needed for use by the body 8.
Studies conducted in vitro (test tubes lab studies) and in animals have indicated that gamm-tocopherol and its major metabolite, gamma-carboxyethyl hydroxychroman (γ-CEHC), may play a role in protecting the body from free radical-induced damage in various conditions of oxidative stress and inflammation 30. Limited intervention studies highlighted in Jiang 30 have not convincingly demonstrated a potential anti-inflammatory effect of gamma-tocopherol in humans. Yet, in two recent randomized, placebo-controlled studies, the supplementation of smokers with gamma-tocopherol potentiated short-term benefits of smoking cessation (with or without nicotine replacement therapy) on vascular endothelial function 32, 33.
Numerous preclinical studies have also suggested that tocotrienols might be beneficial in the prevention of chronic diseases 34. For instance, tocotrienols especially delta-tocotrienol have shown greater anti-proliferative and pro-apoptotic effects than tocopherols in malignant cell lines 35. However, a number of factors, including dose, formulation, and type of study population, affect the bioavailability of tocotrienols and may undermine their putative efficacy in humans 36. There are currently no data available on the effectiveness of supplemental tocotrienols in humans 34.
Figure 4. Alpha-tocopherol antioxidant activity
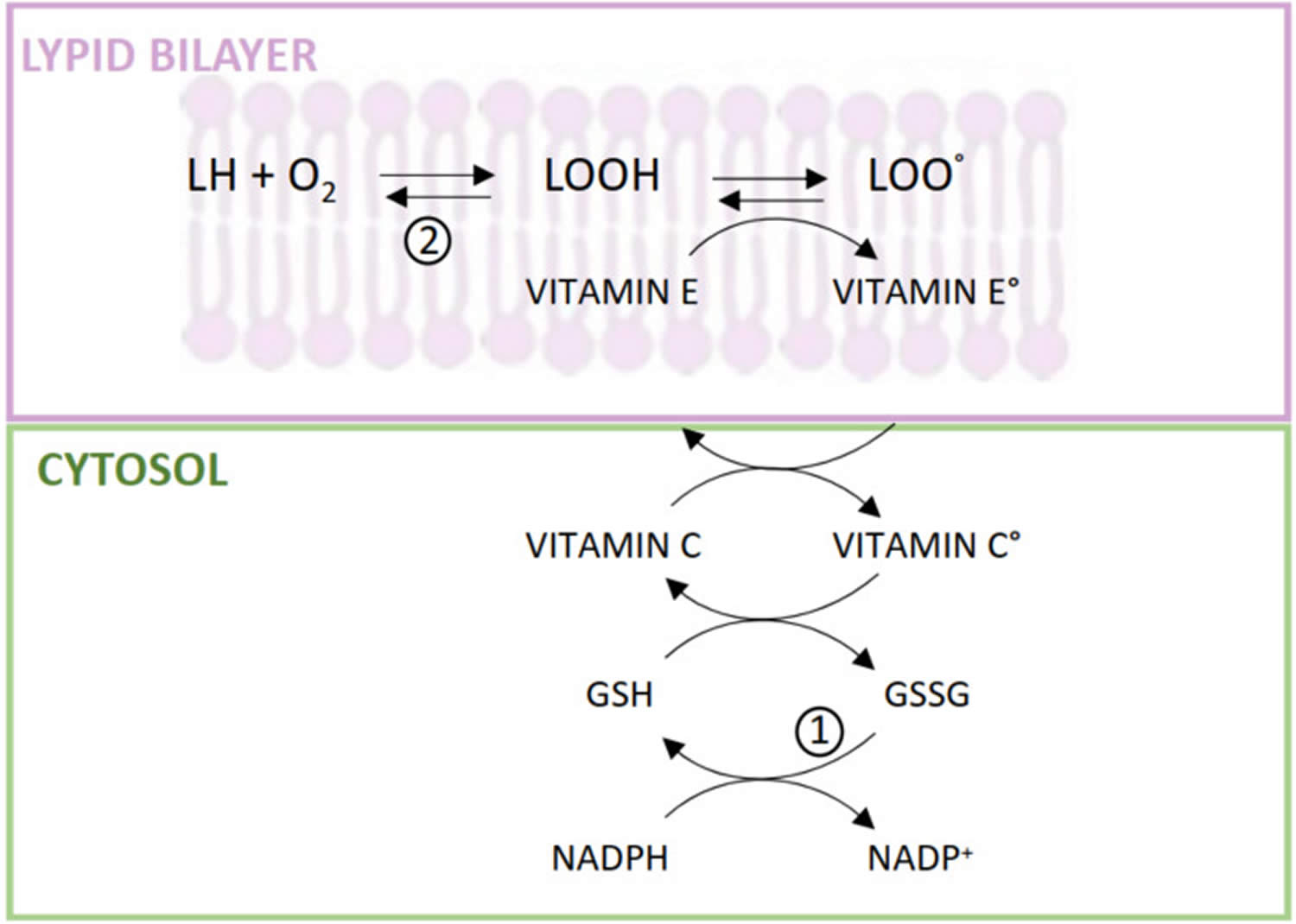
[Source 4 ]Figure 5. Vitamin E antioxidant reactions
Footnote: (1) Hexose monophosphate shunt and GSH-reductase activity; (2) Membrane GSH-peroxidase activity.
Abbreviations: L = membrane lipids; Vitamin E° = tocopheryl radical; Vitamin C°, ascorbyl radical; GSH = reduced glutathione; GSSG =oxidized glutathione; NADP+ = oxidized nicotinamide-adenine-dinucleotide phosphate; NADPH = reduced nicotinamide-adenine-dinucleotide phosphate
[Source 22 ]Immune function
Other functions of alpha-tocopherol are likely to be related to its antioxidant capacity 8. For example, alpha-tocopherol can protect the physiological properties of lipid bilayer membranes and may influence the activity of membrane proteins and enzymes 37. In cell culture studies, alpha-tocopherol was found to improve the formation of an adhesive junction known as immune synapse between naïve T lymphocytes and antigen-presenting cells (APC), which eventually prompted T cell activation and proliferation 38, 39.
The natural age-related decline of the immune function is accompanied by an increased susceptibility to infections, a poorer response to immunization, and higher risks of developing cancers and autoimmune diseases 6. Alpha-tocopherol has been shown to enhance specifically the T cell-mediated immune response that declines with advancing age 40. T cell impaired response has been partly associated with a reduced capacity of naive T cells to be activated during antigen presentation, and to produce interleukin-2 (IL-2) and proliferate as a result 39. However, very few studies have addressed the potential association between alpha-tocopherol and immune function in humans 40. In a small intervention study in older adults (mean age, 70 years), supplementation with 200 mg/day of all-rac-alpha-tocopherol (equivalent to 100 mg of RRR-α-tocopherol) for three months significantly improved natural killer (NK) cytotoxic activity, neutrophil chemotaxis, phagocytic response, and enhanced mitogen-induced lymphocyte proliferation and interleukin-2 (IL-2) production compared to baseline 41. In an earlier trial, daily supplementation of healthy older adults (≥65 years of age) with 200 mg of all-rac-alpha-tocopherol for 235 days also improved T lymphocyte-mediated immunity — as measured with the delayed-type hypersensitivity skin test and increased the production of antibodies in response to hepatitis B and tetanus vaccines 42.
Lower alpha-tocopherol doses failed to improve the delayed-type hypersensitivity response compared to a placebo in another study in healthy participants (ages, 65-80 years) 43. A randomized, placebo-controlled trial in 617 nursing home residents (≥65 years of age) reported that daily supplementation with 200 IU of synthetic alpha-tocopherol (90 mg of RRR-α-tocopherol) for one year significantly lowered the risk of contracting upper respiratory tract infections, especially the common cold, but had no effect on lower respiratory tract (lung) infections 44. More research is needed to examine whether supplemental vitamin E might enhance immune function and reduce risk of infection in older adults.
Reduction of ultraviolet (UV) radiation-induced skin damage
The primary role of vitamin E in the skin is to prevent damage induced by free radicals and reactive oxygen species (ROS); therefore, the use of vitamin E in the prevention of ultraviolet (UV) radiation-induced skin damage has been extensively studied. Ultraviolet (UV) radiation has an immunosuppressive effect on the antigen-presenting cells (APCs) within the epidermis and contributes to the likelihood of skin cancer 45. The sun is by far the strongest source of ultraviolet radiation in our environment. Solar emissions include visible light, heat and ultraviolet (UV) radiation. Just as visible light consists of different colors that become apparent in a rainbow, there are three types of ultraviolet (UV) radiation: UVC, UVB, and UVA. As sunlight passes through the atmosphere, all UVC and most UVB is absorbed by ozone, water vapor, oxygen and carbon dioxide. UVA is not filtered as significantly by the atmosphere.
The ozone layer absorbs 100% of UVC, 90% of UVB, and a minimal amount of UVA 46. For this reason, the depletion of the ozone layer increases UV transmission. UVA is associated with aging and pigmentation 46. It penetrates deep into the skin layer and produces free radical oxygen species, indirectly damaging DNA. UVA increases the number of inflammatory cells in the dermis and decreases the number of antigen-presenting cells 47. UVB causes sunburn and DNA strand breaks. UVB causes pyrimidine dimer mutations, which are associated with nonmelanoma skin cancers 48.
Although molecules in the vitamin E family can absorb light in the UVB spectrum, the “sunscreen” activity of vitamin E is considered limited since it cannot absorb UVA light or light in higher wavelengths of the UVB spectrum 49. Therefore, the primary photoprotective effect of vitamin E is attributed to its role as a lipid-soluble antioxidant.
Many studies in cell culture models (test tube lab studies) have found protective effects of vitamin E molecules on skin cells 50, 51, 52, but these models do not recreate the complex structure of skin tissues. Therefore, human studies are needed.
Studies using orally administered vitamin E have reported mixed results on its photoprotective potential. An early study of vitamin E supplementation in hairless mice found no effect of dietary α-tocopherol acetate on UV-induced carcinogenesis 53. Three other mouse studies reported inhibition of UV-induced tumors in mice fed alpha-tocopherol acetate 54, 55, 56, but one of these studies utilized vitamin E doses that were toxic to animals when combined with the UV treatment 54. Another study in mice found a reduction of UV-induced DNA damage with dietary α-tocopherol acetate, but no effects on other free radical damage were observed in the skin 57. One human study reported that subjects taking 400 IU/day of alpha-tocopherol had reduced UV-induced lipid peroxidation in the skin but concluded there was no overall photoprotective effect 58. This was supported by another human study that found that 400 IU/day of alpha-tocopherol for six months provided no meaningful protection to skin 59. Furthermore, multiple human studies have shown no effect of vitamin E on the prevention or development of skin cancers 60, 61.
In contrast to oral supplementation with alpha-tocopherol alone, multiple studies have found that the combination of vitamin C and vitamin E protects the skin against UV damage. Human subjects orally co-supplemented with vitamins C and E show increased Minimal Erythemal Dose (MED), the lowest dose of ultraviolet radiation that will produce a detectable redness 24 hours after UV exposure 62, 63. The combination of vitamin C and vitamin E was associated with lower amounts of DNA damage after UV exposure 64. Results of another study suggest a mixture of tocopherols and tocotrienols may be superior to α-tocopherol alone, as the mixture showed reduced sunburn reactions and tumor incidence after UV exposure in mice 65. However, further trials with dietary tocotrienol/tocopherol mixtures are needed in human subjects.
Topical application of vitamin E is generally effective for increasing photoprotection of the skin. In rodent models, the application of alpha-tocopherol or alpha-tocopherol acetate before UV exposure reduces UV-induced skin damage by reducing lipid peroxidation 57, 66, 67, 68, limiting DNA damage 57, 69, 70, 71, and reducing the many chemical and structural changes to skin after UV exposure 72, 73, 74, 75. Vitamin E skin applications have also been shown to reduce UV-induced tumor formation in multiple mouse studies 72, 55, 76 and to reduce the effects of photo-activated toxins in the skin 77, 78, 79, 80. Skin application of vitamin E also reduces the effects of UV radiation when applied after the initial exposure. In mice, alpha-tocopherol acetate prevents some of the redness, edema, skin swelling, and skin thickening if applied immediately after UV exposure 74, 75. A similar effect has been shown in rabbits, where applying alpha-tocopherol to skin immediately after UV increased the Minimal Erythemal Dose (the lowest dose of ultraviolet radiation that will produce a detectable redness 24 hours after UV exposure) 81. While the greatest effect was seen when vitamin E was applied immediately after UV exposure, one study showed a significant effect of application eight hours after the insult 74. In human subjects, the use of vitamin E on skin lowers peroxidation of skin surface lipids 82, decreases erythema 83, 84 and limits immune cell activation after UV exposure 85.
Like oral supplementation with vitamin C and vitamin E, skin preparations with both vitamin C and vitamin E have also been successful. Together, the application of these antioxidants to the skin of animals before UV exposure has been shown to decrease sunburned cells 86, 87, decrease DNA damage 86, 88, inhibit redness 86, 89, and decrease skin pigmentation after UV exposure 89. Similar effects have been seen in human subjects 90, 91, 92.
While a majority of studies have found benefit of topical alpha-tocopherol, there is much less evidence for the activity of esters of vitamin E in photoprotection 82. As described above, vitamin E esters require cellular metabolism to produce “free” vitamin E. Thus, skin use of vitamin E esters may provide only limited benefit or may require a delay after administration to provide significant UV protection.
Other skin functions
There is limited information concerning the effects of vitamin E supplementation on photoaging, photodamage, solar damage, or sun damage, which is commonly observed as skin wrinkling. Although vitamin E can protect mice exposed to UV from excessive skin wrinkling, this is a photoprotective effect rather than treatment of pre-existing wrinkles. Other reports using vitamin E to treat photodamage or reduce wrinkles are poorly controlled studies or unpublished observations 93, 94. An analysis of the dietary intake of Japanese women showed no correlation between vitamin E consumption and skin wrinkling 95.
Vitamin E and oils containing tocopherols or tocotrienols have been reported to have moisturizing properties, but data supporting these roles are limited. Cross-sectional studies have shown no association between vitamin E consumption and skin hydration in healthy men and women 95, 96. However, two small studies have shown topical application of vitamin E can improve skin water-binding capacity after two to four weeks of use 97, 98. Long-term studies with topical vitamin E are needed to establish if these moisturizing effects can be sustained.
Environmental pollutants like ozone can decrease vitamin E levels in the skin 99, 100, 101 and lead to free radical damage that may compound the effects of UV exposure 101. Although not well studied, topical applications of vitamin E may reduce pollution-related free radical damage 100.
Anti-inflammatory effects
Vitamin E has been considered an anti-inflammatory agent in the skin, as several studies have supported its prevention of inflammatory damage after UV exposure. As mentioned above, topical vitamin E can reduce UV-induced skin swelling, skin thickness, erythema, and edema — all signs of skin inflammation. In cultured keratinocytes, α-tocopherol and γ-tocotrienol have been shown to decrease inflammatory prostaglandin synthesis, interleukin production, and the induction of cyclooxygenase-2 (COX-2) and NADPH oxidase by UV light 102, 103, 104, as well as limit inflammatory responses to lipid hydroperoxide exposure 105. In mice, dietary gamma-tocotrienol suppresses UV-induced COX-2 expression in the skin 104. Furthermore, topical application of α-tocopherol acetate or a gamma-tocopherol derivative inhibited the induction of COX-2 and nitric oxide synthase (iNOS) following UV exposure 106. In vitro studies (test tube lab studies) have shown similar anti-inflammatory effects of alpha- and gamma-tocopherol on immune cells 107, 108, 109.
Many of these anti-inflammatory effects of vitamin E supplementation have been reported in combination with its photoprotective effects, making it difficult to distinguish an anti-inflammatory action from an antioxidant action that would prevent inflammation from initially occurring. Despite these limitations, there are many reports of vitamin E being used successfully in chronic inflammatory skin conditions, either alone 110, 111 or in combination with vitamin C 112 or vitamin D 113, therefore suggesting a true anti-inflammatory action.
Wound healing
Skin lesions have been reported in rats suffering from vitamin E deficiency, although their origin is unclear. Vitamin E levels decrease rapidly at the site of a cutaneous wound, along with other skin antioxidants, such as vitamin C or glutathione 114. Since skin antioxidants slowly increase during normal wound healing, these observations have stimulated additional studies on the effect of vitamin E on the wound healing process. However, no studies have demonstrated a positive effect of vitamin E supplementation on wound repair in normal skin. Studies have shown that α-tocopherol supplementation decreases wound closure time in diabetic mice, but no effects have been observed in normal mice 115, 116. Vitamin E increases the breaking strength of wounds pre-treated with ionizing radiation 117, but this is likely due to antioxidant functions at the wound site akin to a photoprotective effect. In contrast, intramuscular injection of α-tocopherol acetate in rats has been suggested to decrease collagen synthesis and inhibit wound repair 118.
In humans, studies with topical alpha-tocopherol have either found no effects on wound healing or appearance or have found negative effects on the appearance of scar tissue 119, 120. However, these studies are complicated by a high number of skin reactions to the vitamin E preparations, possibly due to uncontrolled formation of tocopherol radicals in the solutions used. Despite these results, vitamin E, along with zinc and vitamin C, is included in oral therapies for pressure ulcers (bed sores) and burns 121, 122.
Vitamin C interactions
A few human studies using conditions of oxidative stress have demonstrated the importance of vitamin C (ascorbic acid) in the recycling of oxidized alpha-tocopherol back to its reduced state (see Figure 4). Oxidative stress caused by cigarette smoking accelerates the depletion of plasma alpha-tocopherol in smokers compared to nonsmokers 123. In a double-blind, placebo-controlled trial in 11 smokers and 13 nonsmokers given alpha-tocopherol and gamma-tocopherol that was labeled with deuterium (hence traceable), supplementation with vitamin C reduced the rate of vitamin E loss in plasma, most probably by regenerating tocopheryl radicals back to nonoxidized forms 124.
Vitamin K interactions
One study in adults with normal blood clotting (coagulation) status found that daily supplementation with 1,000 IU (670 mg) of RRR-alpha-tocopherol for 12 weeks decreased gamma-carboxylation of prothrombin, a vitamin K-dependent factor in the coagulation cascade 125. Individuals taking anticoagulant drugs like warfarin and those who are vitamin K deficient should not take vitamin E supplements without medical supervision because of the increased risk of bleeding 126.
Vitamin E Supplements
Vitamin E supplements come in different amounts and forms. Supplements of vitamin E typically provide only alpha-tocopherol, although “mixed” products containing other tocopherols and even tocotrienols are available such as gamma-tocopherol, tocotrienols, and mixed tocopherols. Scientists do not know if any of these forms are superior to alpha-tocopherol in supplements.
Two main things to consider when choosing a vitamin E supplement are:
- The amount of vitamin E: Most once-daily multivitamin-mineral supplements provide about 13.5 mg of vitamin E, whereas vitamin E-only supplements commonly contain 67 mg or more. The doses in most vitamin E-only supplements are much higher than the recommended amounts. Some people take large doses because they believe or hope that doing so will keep them healthy or lower their risk of certain diseases.
- The form of vitamin E: Although vitamin E sounds like a single substance, it is actually the name of eight related compounds in food, including alpha-tocopherol. Each form has a different potency, or level of activity in the body.
Naturally occurring alpha-tocopherol exists in one stereoisomeric form, commonly listed as ”D-alpha-tocopherol” on food packaging and supplement labels. In contrast, synthetically produced (laboratory-made) alpha-tocopherol contains equal amounts of its eight possible stereoisomers, commonly listed as ”DL-alpha-tocopherol”; serum and tissues maintain only four of these stereoisomers 25. A given amount of synthetic alpha-tocopherol (all rac-alpha-tocopherol; commonly labeled as “DL” or “dl”) is therefore only half as active as the same amount (by weight in mg) of the natural form (RRR-alpha-tocopherol; commonly labeled as “D” or “d”). People need approximately 50% more IU of synthetic alpha tocopherol from dietary supplements and fortified foods to obtain the same amount of the nutrient as from the natural form.
- The natural vitamin E (D-alpha-tocopherol) is more potent; 1 mg vitamin E = 1 mg d-alpha-tocopherol (natural vitamin E) = 2 mg dl-alpha-tocopherol (synthetic vitamin E).
Some food and dietary supplement labels still list vitamin E in International Units (IUs) rather than mg. 1 IU of the natural form of vitamin E is equivalent to 0.67 mg. 1 IU of the synthetic form of vitamin E is equivalent to 0.45 mg.
Some vitamin E supplements provide other forms of the vitamin, such as gamma-tocopherol, tocotrienols, and mixed tocopherols. Scientists do not know if any of these forms are superior to alpha-tocopherol in supplements.
Most vitamin-E-only supplements provide ≥100 IU of the nutrient. These amounts are substantially higher than the recommended dietary allowances. The 1999–2000 National Health and Nutrition Examination Survey (NHANES) found that 11.3% of adults took vitamin E supplements containing at least 400 IU 127.
Alpha-tocopherol in dietary supplements and fortified foods is often esterified to prolong its shelf life while protecting its antioxidant properties. The body hydrolyzes and absorbs these esters (alpha-tocopheryl acetate and succinate) as efficiently as alpha-tocopherol 25.
Vitamin E interactions with medications
Vitamin E supplements have the potential to interact with several types of medications. A few examples are provided below. People taking these and other medications on a regular basis should discuss their vitamin E intakes with their healthcare providers.
Vitamin E has a few interactions with medications that are listed below:
- Anticoagulation and antiplatelet medications: due to vitamin E inhibiting platelet aggregation and disrupting vitamin K clotting factors there is a protentional increase risk of bleeding combining these two. Vitamin E can inhibit platelet aggregation and antagonize vitamin K-dependent clotting factors. As a result, taking large doses with anticoagulant or antiplatelet medications, such as warfarin (Coumadin®), can increase the risk of bleeding, especially in conjunction with low vitamin K intake. The amounts of supplemental vitamin E needed to produce clinically significant effects are unknown but probably exceed 400 IU/day 7.
- Simvastatin and niacin: Vitamin E can reduce the amount of high-density lipoprotein (HDL or “good” cholesterol) which is the opposite desired effect of taking simvastatin and/or niacin. Some people take vitamin E supplements with other antioxidants, such as vitamin C, selenium, and beta-carotene. This collection of antioxidant ingredients blunted the rise in high-density lipoprotein (HDL) cholesterol levels, especially levels of HDL, the most cardioprotective HDL component, among people treated with a combination of simvastatin (brand name Zocor®) and niacin 128.
- Chemotherapy and radiotherapy: Oncologists generally advise against the use of antioxidant supplements during cancer chemotherapy or radiotherapy because they might reduce the effectiveness of these therapies by inhibiting cellular oxidative damage in cancerous cells 129. Although a systematic review of randomized controlled trials has called this concern into question 130, further research is needed to evaluate the potential risks and benefits of concurrent antioxidant supplementation with conventional therapies for cancer.
Vitamin E health benefits
Scientists are studying vitamin E to understand how it affects health. Here are several examples of what this research has shown.
Many claims have been made about vitamin E’s potential to promote health and prevent and treat disease. The mechanisms by which vitamin E might provide this protection include its function as an antioxidant and its roles in anti-inflammatory processes, inhibition of platelet aggregation, and immune enhancement.
A primary barrier to characterizing the roles of vitamin E in health is the lack of validated biomarkers for vitamin E intake and status to help relate intakes to valid predictors of clinical outcomes 25.
Vitamin E and Coronary Heart Disease
For a time, vitamin E supplements looked like an easy way to prevent heart disease. Promising observational studies, including the Nurses’ Health Study 131 and Health Professionals Follow-Up Study 132, suggested 20 to 40 percent reductions in coronary heart disease risk among individuals who took vitamin E supplements (usually containing 400 IU or more) for least two years 132.
The results of several randomized trials have dampened enthusiasm for vitamin E’s ability to prevent heart attacks or deaths from heart disease among individuals with heart disease or those at high risk for it. In the GISSI Prevention Trial, the results were mixed but mostly showed no preventive effects after more than three years of treatment with vitamin E among 11,000 heart attack survivors 133. Results from the Heart Outcomes Prevention Evaluation (HOPE) trial also showed no benefit of four years worth of vitamin E supplementation among more than 9,500 men and women already diagnosed with heart disease or at high risk for it 134, 135. In fact, when the HOPE trial was extended for another four years, researchers found that study volunteers who took vitamin E had a higher risk of heart failure 135. In the HOPE-TOO followup study, almost 4,000 of the original participants continued to take vitamin E or placebo for an additional 2.5 years 136. HOPE-TOO study found that vitamin E provided no significant protection against heart attacks, strokes, unstable angina, or deaths from cardiovascular disease or other causes after 7 years of treatment 136. Participants taking vitamin E, however, were 13% more likely to experience, and 21% more likely to be hospitalized for, heart failure, a statistically significant but unexpected finding not reported in other large studies.
The HOPE and HOPE-TOO trials provide compelling evidence that moderately high doses of vitamin E supplements do not reduce the risk of serious cardiovascular events among men and women >50 years of age with established heart disease or diabetes 137. These findings are supported by evidence from the Women’s Angiographic Vitamin and Estrogen study, in which 423 postmenopausal women with some degree of coronary stenosis took supplements with 400 IU vitamin E (form not specified) and 500 mg vitamin C twice a day or placebo for >4 years 138. Not only did the supplements provide no cardiovascular benefits, but all-cause mortality was significantly higher in the women taking the supplements. Based on such studies, the American Heart Association has concluded that “the scientific data do not justify the use of antioxidant vitamin supplements [such as vitamin E] for cardiovascular disease risk reduction.” 139.
It’s possible that in people who already have heart disease or are high risk of heart disease, the use of drugs such as aspirin, beta blockers, and ACE inhibitors mask a modest effect of vitamin E, and that vitamin E may have benefits among healthier people. But large randomized controlled trials of vitamin E supplementation in healthy women and men have yielded mixed results.
In the Women’s Health Study, which followed 40,000 healthy women ≥45 years of age who were randomly assigned to receive either vitamin E supplements of 600 IU of natural vitamin E (402 mg) on alternate days or placebo and who were followed for an average of 10 years 140. The investigators found no significant differences in rates of overall cardiovascular events (combined nonfatal heart attacks, strokes, and cardiovascular deaths) or all-cause mortality between the groups. However, the study did find two positive and significant results for women taking vitamin E: they had a 24% reduction in cardiovascular death rates, and those ≥65 years of age had a 26% decrease in nonfatal heart attack and a 49% decrease in cardiovascular death rates 140. A later analysis found that women who took the vitamin E supplements also had a lower risk of developing serious blood clots in the legs and lungs, with women at the highest risk of such blood clots receiving the greatest benefit 141.
The most recent published clinical trial of vitamin E and men’s cardiovascular health included almost 15,000 healthy physicians ≥50 years of age who were randomly assigned to receive 400 IU synthetic alpha-tocopherol (180 mg) every other day, 500 mg vitamin C daily, both vitamins, or placebo 142. During a mean follow-up period of 8 years, intake of vitamin E (and/or vitamin C) had no effect on the incidence of major cardiovascular events, myocardial infarction, stroke, or cardiovascular morality. Furthermore, use of vitamin E was associated with a significantly increased risk of hemorrhagic stroke 142.
Other heart disease prevention trials in healthy people have not been as promising, however. The SU.VI.MAX trial found that seven years of low-dose vitamin E supplementation (as part of a daily antioxidant pill) reduced the risk of cancer and the risk of dying from any cause in men, but did not show these beneficial effects in women; the supplements did not offer any protection against heart disease in men or women 143. Discouraging results have also come from the Physicians’ Health Study II, an eight-year trial that involved nearly 15,000 middle-aged men, most of whom were free of heart disease at the start of the study. Researchers found that taking vitamin E supplements of 400 IU every other day, alone or with vitamin C, failed to offer any protection against heart attacks, strokes, or cardiovascular deaths 144.
More recent evidence suggests that vitamin E may have potential benefits only in certain subgroups of the general population: A trial of high dose vitamin E in Israel, for example, showed a marked reduction in coronary heart disease among people with type 2 diabetes who have a common genetic predisposition for greater oxidative stress 145. So we certainly have not heard the last word on vitamin E and heart disease prevention.
In general, clinical trials have not provided evidence that routine use of vitamin E supplements prevents cardiovascular disease or reduces its morbidity and mortality. However, participants in these studies have been largely middle-aged or elderly individuals with demonstrated heart disease or risk factors for heart disease. Some researchers have suggested that understanding the potential utility of vitamin E in preventing coronary heart disease might require longer studies in younger participants taking higher doses of the supplement 146. Further research is needed to determine whether supplemental vitamin E has any protective value for younger, healthier people at no obvious risk of cardiovascular disease.
Vitamin E and Cancer
Antioxidant nutrients like vitamin E protect cell constituents from the damaging effects of free radicals that, if unchecked, might contribute to cancer development. Vitamin E might also block the formation of carcinogenic nitrosamines formed in the stomach from nitrites in foods and protect against cancer by enhancing immune function 147.
Evidence to date is insufficient to support taking vitamin E to prevent cancer. In fact, daily use of large-dose vitamin E supplements (400 IU of synthetic vitamin E [180 mg]) may increase the risk of prostate cancer. Taken as a whole, observational studies have not found vitamin E in food or supplements to offer much protection against cancer in general, or against specific cancers 148, 149, 150, 151, 152, 153, 154, 155, 156, 157. Some observational studies and clinical trials, however, suggested that vitamin E supplements might lower the risk of advanced prostate cancer in smokers 150, 158, 159, 160.
Investigators had hoped that the Selenium and Vitamin E Cancer Prevention Trial (SELECT) would give more definitive answers on vitamin E and prostate cancer. SELECT’s 18,000 men were assigned to follow one of four pill regimens—vitamin E plus selenium, vitamin E plus a selenium placebo, selenium plus a vitamin E placebo, or a double placebo—and were supposed to be tracked for 7 to 12 years. But investigators halted the study halfway though, in 2008, when early analyses showed that vitamin E offered no cancer or prostate cancer prevention benefit 161. Though the trial ended, researchers continued to follow the men who had participated. In 2011, they reported a 17 percent higher risk of prostate cancer among men assigned to take vitamin E; there was no significant increased risk of prostate cancer among men who took vitamin E and selenium 162. The additional 2011 data show that the men who took vitamin E alone had a 17 percent relative increase in numbers of prostate cancers compared to men on placebo. This difference in prostate cancer incidence between the vitamin E only group and the placebos only group is now statistically significant, and not likely to be due to chance 163.
Though these results, on the face of it, sound worrisome, two other major trials of vitamin E and prostate cancer had quite different results: The Alpha Tocopherol Beta Carotene (ATBC) randomized trial, for example, followed nearly 30,000 Finnish male smokers for an average of six years 158. It found that men assigned to take daily vitamin E supplements had a 32 percent lower risk of developing prostate cancer—and a 41 percent lower risk of dying from prostate cancer—than men given a placebo. However, there are many reasons why the vitamin E supplements may not have prevented prostate cancer. Two of the most likely reasons, looking back at the Alpha-Tocopherol Beta Carotene (ATBC) Cancer Prevention trial, a study designed to test vitamin E and beta carotene for lung cancer prevention in smokers 164. In the The Alpha Tocopherol Beta Carotene trial, a reduction in prostate cancer incidence was observed, but this secondary finding may have been due to chance, as the study was not designed to determine prostate cancer risk 163. Another possible reason that men in ATBC had a reduction in prostate cancer incidence, while men on SELECT did not, is that the dose of vitamin E used in SELECT (400 IU/day) was higher than the dose used in the ATBC (50 IU/day) 163. Researchers sometimes talk about a “U-shaped response curve” where very low or very high blood levels of a nutrient are harmful but more moderate levels are beneficial; while the ATBC dose may have been preventive, the SELECT dose may have been too large to have a prevention benefit 163.
The large and long-term Physicians’ Health Study II trial, meanwhile, found that vitamin E supplements had no effect on the risk of prostate cancer or any other cancer 165.
Bear in mind that prostate cancer develops slowly, and any study looking at prostate cancer prevention needs to track men for a long time. By stopping the SELECT trial early, there’s no way to tell if vitamin E could have helped protect against prostate cancer in some men if they had continued the trial over a longer period of time. Very few cases in the SELECT Trial were of advanced prostate cancer, further limiting the interpretation of the findings.
One study of women in Iowa provides evidence that higher intakes of vitamin E from foods and supplements could decrease the risk of colon cancer, especially in women <65 years of age 166. The overall relative risk for the highest quintile of intake (>35.7 IU/day, form not specified) compared to the lowest quintile (<5.7 IU/day, form not specified) was 0.32. However, prospective cohort studies of 87,998 women in the Nurses’ Health Study and 47,344 men in the Health Professionals Follow-up Study failed to replicate these results 167. Although some research links higher intakes of vitamin E with decreased incidence of breast cancer, an examination of the impact of dietary factors, including vitamin E, on the incidence of postmenopausal breast cancer in >18,000 women found no benefit from the vitamin 168.
The American Cancer Society conducted an epidemiologic study examining the association between use of vitamin C and vitamin E supplements and bladder cancer mortality. Of the almost one million adults followed between 1982 and 1998, adults who took supplemental vitamin E for 10 years or longer had a reduced risk of death from bladder cancer 169; vitamin C supplementation provided no protection.
Should men take vitamin E or selenium supplements for cancer prevention?
No. Scientists do not understand how these supplements really work and more importantly, the interactions that these supplements have together or with foods, drugs, or other supplements. There are no clinical trials that show a benefit from taking vitamin E or selenium to reduce the risk of prostate cancer or any other cancer or heart disease 170, 171, 172, 173, 174, 175. While the men in SELECT who took both vitamin E and selenium did not have a statistically significant increase in their risk for prostate cancer, they also did not have a reduced risk of prostate cancer or any other cancer or heart disease. SELECT researchers were surprised by the findings in the men who took both vitamin E and selenium, and while the 2014 analysis suggests possible reasons for the findings, the mechanism remains unclear 163.
Evidence to date is insufficient to support taking vitamin E to prevent cancer. In fact, daily use of large-dose vitamin E supplements (400 IU) may increase the risk of prostate cancer 176.
Vitamin E and Macular Degeneration
Age-related macular degeneration (AMD) and cataracts are among the most common causes of significant vision loss in older people. Their causes are usually unknown, but the cumulative effects of oxidative stress have been postulated to play a role. If so, nutrients with antioxidant functions, such as vitamin E, could be used to prevent or treat these conditions.
Prospective cohort studies have found that people with relatively high dietary intakes of vitamin E (e.g., 20 mg/day [30 IU]) have an approximately 20% lower risk of developing age-related macular degeneration than people with low intakes (e.g., <10 mg/day [<15 IU]) 177. However, two randomized controlled trials in which participants took supplements of vitamin E (500 IU/day [335 mg] d-alpha-tocopherol in one study 178 and 111 IU/day (50 mg) dl-alpha-tocopheryl acetate combined with 20 mg/day beta-carotene in the other study 179 or a placebo failed to show a protective effect for vitamin E on age-related macular degeneration. The Age-Related Eye Disease Study (AREDS) 180, a large randomized clinical trial, found that participants at high risk of developing advanced age-related macular degeneration (i.e., those with intermediate age-related macular degeneration or those with advanced age-related macular degeneration in one eye) reduced their risk of developing advanced age-related macular degeneration by 25% by taking a daily supplement containing vitamin E (400 IU [180 mg] dl-alpha-tocopheryl acetate), beta-carotene (15 mg), vitamin C (500 mg), zinc (80 mg), and copper (2 mg) compared to participants taking a placebo over 5 years. A follow-up AREDS2 study 181 confirmed the value of this and similar supplement formulations in reducing the progression of age-related macular degeneration over a median follow-up period of 5 years.
Overall, the available evidence is inconsistent with respect to whether vitamin E supplements, taken alone or in combination with other antioxidants, can reduce the risk of developing age-related macular degeneration or cataracts. However, the formulations of vitamin E, other antioxidants, zinc, and copper used in AREDS hold promise for slowing the progression of age-related macular degeneration in people at high risk of developing advanced age-related macular degeneration.
Vitamin E and Cataracts
Several observational studies have revealed a potential relationship between vitamin E supplements and the risk of cataract formation. One prospective cohort study found that lens clarity was superior in participants who took vitamin E supplements and those with higher blood levels of the vitamin 182. In another study, long-term use of vitamin E supplements was associated with slower progression of age-related lens opacification 183. However, in the AREDS trial, the use of a vitamin E-containing (as dl-alpha-tocopheryl acetate) formulation had no apparent effect on the development or progression of cataracts over an average of 6.3 years 184. The AREDS2 study, which also tested formulations containing 400 IU (180 mg) vitamin E, confirmed these findings 185.
Vitamin E and Cognitive Function
The brain has a high oxygen consumption rate and abundant polyunsaturated fatty acids in the neuronal cell membranes. Researchers hypothesize that if cumulative free-radical damage to neurons over time contributes to cognitive decline and neurodegenerative diseases, such as Alzheimer’s disease, then ingestion of sufficient or supplemental antioxidants (such as vitamin E) might provide some protection 186. This hypothesis was supported by the results of a clinical trial in 341 patients with Alzheimer’s disease of moderate severity who were randomly assigned to receive a placebo, vitamin E (2,000 IU/day dl-alpha-tocopherol), a monoamine oxidase inhibitor (selegiline), or vitamin E and selegiline 186. Over 2 years, treatment with vitamin E and selegiline, separately or together, significantly delayed functional deterioration and the need for institutionalization compared to placebo. However, participants taking vitamin E experienced significantly more falls.
Vitamin E consumption from foods or supplements was associated with less cognitive decline over 3 years in a prospective cohort study of elderly, free-living individuals aged 65–102 years 187. However, a clinical trial in primarily healthy older women who were randomly assigned to receive 600 IU (402 mg) d-alpha-tocopherol every other day or a placebo for ≤4 years found that the supplements provided no apparent cognitive benefits 188. Another trial in which 769 men and women with mild cognitive impairment were randomly assigned to receive 2,000 IU/day vitamin E (form not specified), a cholinesterase inhibitor (donepezil), or placebo found no significant differences in the progression rate of Alzheimer’s disease between the vitamin E and placebo groups 189.
In summary, most research results do not support the use of vitamin E supplements by healthy or mildly impaired individuals to maintain cognitive performance or slow its decline with normal aging 190. More research is needed to identify the role of vitamin E, if any, in the management of cognitive impairment 191.
Vitamin E and Neurodegenerative Diseases
The brain has a high oxygen consumption rate and abundant polyunsaturated fatty acids in the neuronal cell membranes. Researchers hypothesize that if cumulative free-radical damage to neurons over time contributes to cognitive decline and neurodegenerative diseases, such as Alzheimer’s disease, then ingestion of sufficient or supplemental antioxidants (such as vitamin E) might provide some protection 192. This hypothesis was supported by the results of a clinical trial in 341 patients with Alzheimer’s disease of moderate severity who were randomly assigned to receive a placebo, vitamin E (2,000 IU/day dl-alpha-tocopherol), a monoamine oxidase inhibitor (selegiline), or vitamin E and selegiline 193. Over 2 years, treatment with vitamin E and selegiline, separately or together, significantly delayed functional deterioration and the need for institutionalization compared to placebo. However, participants taking vitamin E experienced significantly more falls.
Scientists seeking to untangle the causes of Alzheimer’s, Parkinson’s, and other diseases of the brain and nervous system have focused on the role that free radical damage plays in these diseases’ development 194. But to date, there is little evidence as to whether vitamin E can help protect against these diseases or that it offers any benefit to people who already have these diseases.
Vitamin E and Dementia
Some prospective studies suggest that vitamin E supplements, particularly in combination with vitamin C, may be associated with small improvements in cognitive function or lowered risk of Alzheimer’s disease and other forms of dementia, while other studies have failed to find any such benefit 195, 196, 197, 198. A three-year randomized controlled trial in people with mild cognitive impairment—often a precursor to Alzheimer’s disease—found that taking 2,000 IU of vitamin E daily failed to slow the progression to Alzheimer’s disease 199. Keep in mind, however, that the progression from mild cognitive impairment to Alzheimer’s disease can take many years, and this study was fairly short, so it is probably not the last word on vitamin E and dementia.
Vitamin E and Parkinson’s disease
Some, but not all, prospective studies suggest that getting higher intakes of vitamin E from diet—not from high-dose supplements—is associated with a reduced risk of Parkinson’s disease 200, 201, 202. In people who already have Parkinson’s, high-dose vitamin E supplements do not slow the disease’s progression 203. Why the difference between vitamin E from foods versus that from supplements ? It’s possible that foods rich in vitamin E, such as nuts or legumes, contain other nutrients that protect against Parkinson’s disease. More research is needed.
Vitamin E and Amyotrophic Lateral Sclerosis (ALS)
One large prospective study that followed nearly 1 million people for up to 16 years found that people who regularly took vitamin E supplements had a lower risk of dying from ALS than people who never took vitamin E supplements 204. More recently, a combined analysis of multiple studies with more than 1 million participants found that the longer people used vitamin E supplements, the lower their risk of ALS 205. Clinical trials of vitamin E supplements in people who already have ALS have generally failed to show any benefit, however 206. This may be a situation where vitamin E is beneficial for prevention, rather than treatment, but more research is needed.
Vitamin E and Fatty liver diseases
The increasing incidence of nonalcoholic fatty liver disease (NAFLD) in children and adults in industrialized countries is mainly attributed to the ongoing epidemic of obesity and type 2 diabetes mellitus. NAFLD (nonalcoholic fatty liver disease) results from the abnormal accumulation of fat (steatosis) in the liver in the absence of heavy alcohol consumption 207, 208. Although the condition is considered to be largely benign, NAFLD can progress to a more severe disease called nonalcoholic steatohepatitis (NASH) with increased risks of cirrhosis, hepatocellular carcinoma (liver cancer), and cardiovascular disease 209, 207, 208. Both environmental and genetic factors are contributing to the development of non-alcoholic fatty liver disease (NAFLD) and its progression 208. Oxidative stress is thought to be one of the possible mechanisms responsible for prompting inflammatory processes that can lead to the progression of NAFLD to NASH 207, 208, 210.
There is currently no established treatment for NAFLD and NASH other than interventions that encourage lifestyle changes and the use of medicines to control or treat metabolic disorders 208, 207, 211. In the multicenter PIVENS (PIoglitazone versus Vitamin E versus placebo for the treatment of Nonalcoholic Steatohepatitis) trial, 247 nondiabetic subjects with nonalcoholic steatohepatitis (NASH) were randomized to receive 30 mg/day of pioglitazone (an insulin-sensitizing drug), 800 IU/day (536 mg/day) of RRR-alpha-tocopherol, or a placebo for 96 weeks 212. Only vitamin E supplementation significantly increased the overall rate of improvement in histological abnormalities that characterize nonalcoholic steatohepatitis (NASH) on liver biopsies (i.e., hepatocellular ballooning, steatosis, and lobular inflammation) 213. Both active treatments improved some markers of liver function (i.e., alanine aminotransferase and aspartate aminotransferase) 213. Yet, results from another two-year, randomized controlled trial — called TONIC for Treatment Of Nonalcoholic fatty liver disease In Children — in 173 children (ages, 8-17 years) with NAFLD failed to observe any significant reduction in blood concentrations of alanine and aspartate aminotransferases either with supplemental vitamin E (536 mg/day of RRR-α-tocopherol) or with metformin (an anti-diabetic drug; 1,000 mg/day) compared to placebo 214. However, vitamin E supplementation significantly improved the overall disease activity score — used to quantify the severity of the disease. In addition, a recent meta-analysis of another six trials found that vitamin E significantly lowered circulating aminotransferase concentrations in NAFLD and NASH patients, suggesting liver function improvements 215. Finally, in a small nonrandomized, unblinded, controlled study in 42 obese children (mean age, 8 years) with NAFLD, lifestyle recommendations combined with 600 mg/day of supplemental RRR-alpha-tocopheryl acetate for six months reduced markers of oxidative stress and liver dysfunction and improved insulin sensitivity and the profile of lipid in the blood, when compared to baseline 216. No such changes in markers of oxidative stress, liver function, and glucose utilization were reported in the lifestyle intervention only group 216. Further randomized and well-controlled studies are needed to confirm these preliminary findings.
Vitamin E oil for Skin
Vitamin E is the most abundant lipophilic antioxidant found in human skin 217. In humans, levels of vitamin E in the epidermis are higher than the dermis 217. Although the predominant form of vitamin E in skin of unsupplemented individuals is alpha-tocopherol, skin may also contain measurable amounts of gamma-tocopherol 218 and other diet-derived tocopherols and tocotrienols 219.
Vitamin E first accumulates in the sebaceous glands before it is delivered to the skin surface through sebum 220. Following oral ingestion, it takes at least seven days before the vitamin E content of sebum is altered 221. There are no transport proteins specific for vitamin E in the skin. Sebum is secreted to the surface of the stratum corneum, where it concentrates in the lipid-rich extracellular matrix of this layer 218. Due to its lipophilic nature, vitamin E can also penetrate into all underlying layers of skin 222. Skin vitamin E levels are higher in individuals with increased sebum production, as well as in skin types that naturally produce more sebum (e.g., “oily’ skin on the face vs. drier skin on the arm) 222.
Exposures to UV light 223 or ozone 101 lower the vitamin E content in skin, primarily in the stratum corneum. Vitamin E concentrations in the human epidermis also decline with age 217. Since epidermal structure changes with age 224, this may be due to increased UV penetration of this layer.
Vitamin E deficiency may affect skin function, but there is little evidence from human studies. Vitamin E deficiency in rats has been reported to cause skin ulcerations 225 and changes in skin collagen cross-linking 226, but the underlying cause of these effects is unknown.
Many people believe that there are special healing qualities to vitamin E on skin. Anecdotal reports claim that vitamin E speeds wound healing and improves the cosmetic outcome of burns and other wounds. Many lay people use vitamin E on a regular basis to improve the outcome of scars and several physicians recommend topical vitamin E after skin surgery or resurfacing.
In a very small double blinded clinical trial 227 with 15 patients who had undergone skin cancer removal surgery. After the surgery, the patients were given two ointments each labeled A or B. A was a regular emollient, and the B was emollient mixed with vitamin E. The scars were randomly divided into parts A and B. Patients were asked to put the A ointment on part A and the B ointment on part B twice daily for 4 weeks. The physicians, a third blinded investigator and the patients independently evaluated the scars for cosmetic appearance on weeks 1, 4, and 12. The results of this study show that topically applied vitamin E does not help in improving the cosmetic appearance of scars and that the application of topical vitamin E may actually be detrimental to the cosmetic appearance of a scar. In 90% of the cases in this study, topical vitamin E either had no effect on, or actually worsened, the cosmetic appearance of scars. Of the patients studied, 33% developed a contact dermatitis to the vitamin E. Therefore the researchers conclude that use of topical vitamin E on surgical wounds should be discouraged 227.
Skin application
Skin application of vitamin E has been used in a wide variety of forms throughout history, ranging from the application of oils to the skin surface to the use of modern cosmetic formulations. Just as sebum provides a delivery mechanism for vitamin E to the stratum corneum, topical applications of vitamin E permeate the epidermis and dermis 228. The rate of percutaneous vitamin E absorption and factors that influence its penetration are largely unknown in humans, with a large range of concentrations and times used in various studies. It is generally assumed that solutions with vitamin E concentrations as low as 0.1% can increase vitamin E levels in the skin 229. Interestingly, vitamin E levels in the dermis increase greatly after topical application, likely accumulating in the sebaceous glands 228. However, although it is increased after topical delivery, the concentration of vitamin E in the dermis is lower than in the stratum corneum. Skin supplied only with dietary vitamin E primarily contains alpha- and gamma-tocopherol 221; by contrast, skin supplied with synthetic vitamin E topically can contain a mixture of different tocopherols and/or tocotrienols 100. In terms of penetration and absorption following topical application, tocotrienols and tocopherols accumulate in skin at varying rates, but the mechanisms governing these differences are unclear 228.
After topical application, vitamin E accumulates not only in cell membranes but also in the extracellular lipid matrix of the stratum corneum, where vitamin E contributes to antioxidant defenses. However, much of a topically applied dose of vitamin E alone will be destroyed in the skin following exposure to UV light 223. This suggests that although vitamin E is working as an antioxidant, it is unstable on its own and easily lost from the skin. Thus, improving the stability of topical applications with vitamin E is important. Products containing both vitamin C and vitamin E have shown greater efficacy in photoprotection than either antioxidant alone.
The stability of topical vitamin E solutions may also be increased by the use of vitamin E conjugates. These vitamin E derivatives are usually commercially produced esters of tocopherol (although tocotrienol esters have been formulated) that are resistant to oxidation but can still penetrate the skin layers. Vitamin E conjugates, however, do not have antioxidant functions. To be effective, the molecule conjugated to vitamin E must be removed by enzymes within a cell. Since the stratum corneum contains metabolically inactive cells and the remaining layers of the epidermis and dermis may contain a large volume of extracellular proteins, it is unclear how efficiently ester conjugates are converted to “free” vitamin E in skin. Depending on the compound and the model system used, the effectiveness of these formulations can vary greatly 230, and studies often do not compare the application of vitamin E conjugates to the application of unmodified vitamin E molecules.
Because vitamin E can absorb UV light to produce free radicals, there is the possibility that heavy sunlight exposure after topical application can cause skin reactions. However, concentrations of vitamin E between 0.1%-1.0% are generally considered safe and effective to increase vitamin E levels in the skin, but higher levels of α-tocopherol have been used with no apparent side effects 229. On the other hand, studies of dose-dependent vitamin E accumulation and effectiveness in skin protection are lacking. Some forms of vitamin E, especially ester conjugates, have led to adverse reactions in the skin, including allergic contact dermatitis and erythema. Although such reactions may be due to oxidation by-products, the emulsion creams used for topical delivery of compounds may also contribute to the observed effects 231.
Vitamin E functions in healthy skin
Photoprotection
The primary role of vitamin E in the skin is to prevent damage induced by free radicals and reactive oxygen species; therefore, the use of vitamin E in the prevention of ultraviolet (UV)-induced damage has been extensively studied. Although molecules in the vitamin E family can absorb light in the Ultraviolet B (UVB) spectrum, the “sunscreen” activity of vitamin E is considered limited since it cannot absorb Ultraviolet A (UVA) light or light in higher wavelengths of the Ultraviolet B (UVB) spectrum 49. Thus, the primary photoprotective effect of vitamin E is attributed to its role as a lipid-soluble antioxidant.
Many studies in cell culture models (in vitro studies) have found protective effects of vitamin E molecules on skin cells 51, but these models do not recreate the complex structure of skin tissues. Therefore, in vivo studies are needed.
Studies using orally administered vitamin E have reported mixed results on its photoprotective potential. An early study of vitamin E supplementation in hairless mice found no effect of dietary α-tocopherol acetate on UV-induced carcinogenesis 53. Three other mouse studies reported inhibition of UV-induced tumors in mice fed α-tocopherol acetate 55, but one of these studies utilized vitamin E doses that were toxic to animals when combined with the UV treatment 54. Another study in mice found a reduction of UV-induced DNA damage with dietary α-tocopherol acetate, but no effects on other free radical damage were observed in the skin 57. One human study reported that subjects taking 400 IU/day of α-tocopherol had reduced UV-induced lipid peroxidation in the skin but concluded there was no overall photoprotective effect 58. This was supported by another human study that found that 400 IU/day of α-tocopherol for six months provided no meaningful protection to skin 232. Furthermore, multiple human studies have shown no effect of vitamin E on the prevention or development of skin cancers 60.
In contrast to oral supplementation with α-tocopherol alone, multiple studies have found that the combination of vitamin C and vitamin E protects the skin against UV damage. Human subjects orally co-supplemented with vitamins C and E show increased Minimal Erythemal Dose (MED), the lowest dose of ultraviolet radiation that will produce a detectable redness 24 hours after UV exposure 63. The combination of the two vitamins was associated with lower amounts of DNA damage after UV exposure 64. Results of another study suggest a mixture of tocopherols and tocotrienols may be superior to α-tocopherol alone, as the mixture showed reduced sunburn reactions and tumor incidence after UV exposure in mice 65. However, further trials with dietary tocotrienol/tocopherol mixtures are needed in human subjects.
Topical application of vitamin E is generally effective for increasing photoprotection of the skin. In rodent models, the application of α-tocopherol or α-tocopherol acetate before UV exposure reduces UV-induced skin damage by reducing lipid peroxidation 66, limiting DNA damage 71, and reducing the many chemical and structural changes to skin after UV exposure 73. Vitamin E topical applications have also been shown to reduce UV-induced tumor formation in multiple mouse studies 55 and to reduce the effects of photo-activated toxins in the skin 77. Topical application of vitamin E also reduces the effects of UV radiation when applied after the initial exposure. In mice, α-tocopherol acetate prevents some of the erythema, edema, skin swelling, and skin thickening if applied immediately after UV exposure 74. A similar effect has been shown in rabbits, where applying α-tocopherol to skin immediately after UV increased the Minimal Erythemal Dose (the lowest dose of ultraviolet radiation that will produce a detectable redness 24 hours after UV exposure) 81. While the greatest effect was seen when vitamin E was applied immediately after UV exposure, one study showed a significant effect of application eight hours after the insult 74. In human subjects, the use of vitamin E on skin lowers peroxidation of skin surface lipids 82, decreases erythema 83, and limits immune cell activation after UV exposure 85.
Like oral supplementation with vitamin C and vitamin E, topical preparations with both vitamins have also been successful. Together, the application of these antioxidants to the skin of animals before UV exposure has been shown to decrease sunburned cells 86, decrease DNA damage 88, inhibit erythema 86, and decrease skin pigmentation after UV exposure 89. Similar effects have been seen in human subjects 92.
While a majority of studies have found benefit of topical α-tocopherol, there is much less evidence for the activity of esters of vitamin E in photoprotection 82. As described above, vitamin E esters require cellular metabolism to produce “free” vitamin E. Thus, topical use of vitamin E esters may provide only limited benefit or may require a delay after administration to provide significant UV protection.
Anti-inflammatory effects
Vitamin E has been considered an anti-inflammatory agent in the skin, as several studies have supported its prevention of inflammatory damage after UV exposure. As mentioned above, topical vitamin E can reduce UV-induced skin swelling, skin thickness, erythema, and edema — all signs of skin inflammation. In cultured keratinocytes, α-tocopherol and γ-tocotrienol have been shown to decrease inflammatory prostaglandin synthesis, interleukin production, and the induction of cyclooxygenase-2 (COX-2) and NADPH oxidase by UV light 103, as well as limit inflammatory responses to lipid hydroperoxide exposure 105. In mice, dietary γ-tocotrienol suppresses UV-induced COX-2 expression in the skin 104. Furthermore, topical application of α-tocopherol acetate or a γ-tocopherol derivative inhibited the induction of COX-2 and nitric oxide synthase (iNOS) following UV exposure 106. In vitro studies have shown similar anti-inflammatory effects of α- and γ-tocopherol on immune cells 108.
Many of these anti-inflammatory effects of vitamin E supplementation have been reported in combination with its photoprotective effects, making it difficult to distinguish an anti-inflammatory action from an antioxidant action that would prevent inflammation from initially occurring. Despite these limitations, there are many reports of vitamin E being used successfully in chronic inflammatory skin conditions, either alone 110 or in combination with vitamin C 112 or vitamin D 113, thus suggesting a true anti-inflammatory action.
Wound healing
As mentioned above, skin lesions have been reported in rats suffering from vitamin E deficiency, although their origin is unclear. Vitamin E levels decrease rapidly at the site of a cutaneous wound, along with other skin antioxidants, such as vitamin C or glutathione 114. Since skin antioxidants slowly increase during normal wound healing, these observations have stimulated additional studies on the effect of vitamin E on the wound healing process. However, no studies have demonstrated a positive effect of vitamin E supplementation on wound repair in normal skin. Studies have shown that α-tocopherol supplementation decreases wound closure time in diabetic mice, but no effects have been observed in normal mice 115. Vitamin E increases the breaking strength of wounds pre-treated with ionizing radiation 117, but this is likely due to antioxidant functions at the wound site akin to a photoprotective effect. In contrast, intramuscular injection of α-tocopherol acetate in rats has been suggested to decrease collagen synthesis and inhibit wound repair 118.
In humans, studies with topical alpha-tocopherol have either found no effects on wound healing or appearance or have found negative effects on the appearance of scar tissue 119, 120. However, these studies are complicated by a high number of skin reactions to the vitamin E preparations, possibly due to uncontrolled formation of tocopherol radicals in the solutions used. Despite these results, vitamin E, along with zinc and vitamin C, is included in oral therapies for pressure ulcers (bed sores) and burns 121, 122.
Other skin functions
There is limited information concerning the effects of vitamin E supplementation on photodamage, which is commonly observed as skin wrinkling. Although vitamin E can protect mice exposed to UV from excessive skin wrinkling, this is a photoprotective effect rather than treatment of pre-existing wrinkles. Other reports using vitamin E to treat photodamage or reduce wrinkles are poorly controlled studies or unpublished observations 94. An analysis of the dietary intake of Japanese women showed no correlation between vitamin E consumption and skin wrinkling 95.
Vitamin E and oils containing tocopherols or tocotrienols have been reported to have moisturizing properties, but data supporting these roles are limited. Cross-sectional studies have shown no association between vitamin E consumption and skin hydration in healthy men and women 95, 96. However, two small studies have shown topical application of vitamin E can improve skin water-binding capacity after two to four weeks of use 97, 98. Long-term studies with topical vitamin E are needed to establish if these moisturizing effects can be sustained.
Environmental pollutants like ozone can decrease vitamin E levels in the skin 99 and lead to free radical damage that may compound the effects of UV exposure 101. Although not well studied, topical applications of vitamin E may reduce pollution-related free radical damage 100.
Vitamin E oil for skin summary
Vitamin E is an integral part of the skin’s antioxidant defenses, primarily providing protection against UV radiation and other free radicals that may come in contact with the epidermis. Oral supplementation with only vitamin E may not provide adequate protection for the skin, and co-supplementation of vitamin E and vitamin C may be warranted to effectively increase the photoprotection of skin through the diet. However, topical vitamin E seems to be an effective mechanism for both delivery to the skin and providing a photoprotective effect. Additional anti-inflammatory effects of topical vitamin E have been seen in the skin, although more studies are needed to determine if vitamin E primarily works as a free-radical scavenger or can have other effects on inflammatory signaling. Vitamin E is available commercially as a variety of synthetic derivatives, but the limited cellular metabolism in skin layers makes the use of such products problematic. Use of unesterified vitamin E, similar to that found in natural sources, has provided the most consistent data concerning its topical efficacy. The vitamin E family consists of eight different tocopherols and tocotrienols, and it will be important for future studies to determine if one or more of these molecules can have unique effects on skin function.
How much vitamin E do I need?
The amount of vitamin E you need each day depends on your age. Average daily recommended amounts are listed below in milligrams (mg) and in International Units (IU). Package labels list the amount of vitamin E in foods and dietary supplements in International Units (IU). Intake recommendations for vitamin E and other nutrients are provided in the Dietary Reference Intakes (DRIs) developed by the Food and Nutrition Board (FNB) at the Institute of Medicine of The National Academies 25. Dietary Reference Intake (DRI) is the general term for a set of reference values used to plan and assess nutrient intakes of healthy people. These values, which vary by age and gender, include:
- Recommended Dietary Allowance (RDA): average daily level of intake sufficient to meet the nutrient requirements of nearly all (97%–98%) healthy people.
- Adequate Intake (AI): established when evidence is insufficient to develop an RDA and is set at a level assumed to ensure nutritional adequacy.
- Tolerable Upper Intake Level (UL): maximum daily intake unlikely to cause adverse health effects.
The Food and Nutrition Board’s vitamin E recommendations are for alpha-tocopherol alone, the only form maintained in plasma. The Food and Nutrition Board (FNB) based these recommendations primarily on serum levels of the nutrient that provide adequate protection in a test measuring the survival of red blood cells when exposed to hydrogen peroxide, a free radical. Acknowledging “great uncertainties” in these data, the Food and Nutrition Board (FNB) has called for research to identify other biomarkers for assessing vitamin E requirements.
RDAs for vitamin E are provided in milligrams (mg) and are listed in Table 1. Because insufficient data are available to develop RDAs for infants, Adequate Intake (AI) were developed based on the amount of vitamin E consumed by healthy breastfed babies.
At present, the vitamin E content of foods and dietary supplements is listed on labels in international units (IUs), a measure of biological activity rather than quantity. Naturally sourced vitamin E is called RRR-alpha-tocopherol (commonly labeled as d-alpha-tocopherol); the synthetically produced form is all rac-alpha-tocopherol (commonly labeled as dl-alpha-tocopherol). Conversion rules are as follows:
To convert from mg to IU:
- 1 mg of alpha-tocopherol is equivalent to 1.49 IU of the natural form or 2.22 IU of the synthetic form.
To convert from IU to mg:
- 1 IU of the natural form is equivalent to 0.67 mg of alpha-tocopherol.
- 1 IU of the synthetic form is equivalent to 0.45 mg of alpha-tocopherol.
Table 1 lists the RDAs for alpha-tocopherol in both mg and IU of the natural form; for example, 15 mg x 1.49 IU/mg = 22.4 IU. The corresponding value for synthetic alpha-tocopherol would be 33.3 IU (15 mg x 2.22 IU/mg).
Table 1. Recommended Dietary Allowances (RDAs) for Vitamin E (Alpha-Tocopherol)
| Life Stage | Recommended Amount |
|---|---|
| Birth to 6 months* | 4 mg (6 IU) |
| Infants 7–12 months* | 5 mg (7.5 IU) |
| Children 1–3 years | 6 mg (9 IU) |
| Children 4–8 years | 7 mg (10.4 IU) |
| Children 9–13 years | 11 mg (16.4 IU) |
| Teens 14–18 years | 15 mg (22.4 IU) |
| Adults | 15 mg (22.4 IU) |
| Pregnant teens and women | 15 mg (22.4 IU) |
| Breastfeeding teens and women | 19 mg (28.4 IU) |
Footnote: *Adequate Intake (established when evidence is insufficient to develop an RDA and is set at a level assumed to ensure nutritional adequacy)
What foods provide vitamin E?
Numerous foods provide vitamin E. Nuts, seeds, and vegetable oils are among the best sources of alpha-tocopherol, and significant amounts are available in green leafy vegetables and fortified cereals (see Table 2 for a more detailed list) 233. Most vitamin E in American diets is in the form of gamma-tocopherol from soybean, canola, corn, and other vegetable oils and food products 15.
Vitamin E is found naturally in foods and is added to some fortified foods. You can get recommended amounts of vitamin E by eating a variety of foods including the following:
- Vegetable oils like wheat germ, sunflower, and safflower oils are among the best sources of vitamin E. Corn and soybean oils also provide some vitamin E.
- Nuts (such as peanuts, hazelnuts, and, especially, almonds) and seeds (like sunflower seeds) are also among the best sources of vitamin E.
- Green vegetables, such as spinach and broccoli, provide some vitamin E.
- Food companies add vitamin E to some breakfast cereals, fruit juices, margarines and spreads, and other foods. To find out which ones have vitamin E, check the product labels.
Vitamin E from natural (food) sources is commonly listed as “d-alpha-tocopherol” on food packaging and supplement labels. Synthetic (laboratory-made) vitamin E is commonly listed as “dl-alpha-tocopherol”. The natural form is more potent. For example, 100 IU of natural vitamin E is equal to about 150 IU of the synthetic form.
Some vitamin E supplements provide other forms of the vitamin, such as gamma-tocopherol, tocotrienols, and mixed tocopherols. Scientists do not know if any of these forms are superior to alpha-tocopherol in supplements.
The U.S. Department of Agriculture’s (USDA’s) FoodData Central (https://fdc.nal.usda.gov) lists the nutrient content of many foods, including, in some cases, the amounts of alpha-, beta-, gamma-, and delta-tocopherol arranged by nutrient content (https://ods.od.nih.gov/pubs/usdandb/VitaminE-Content.pdf) and by food name (https://ods.od.nih.gov/pubs/usdandb/VitaminE-Food.pdf).
Table 2. Food Sources of Vitamin E (Alpha-Tocopherol)
| Food | Milligrams (mg) per serving | Percent DV* |
| Wheat germ oil, 1 tablespoon | 20.3 | 100 |
| Sunflower seeds, dry roasted, 1 ounce | 7.4 | 37 |
| Almonds, dry roasted, 1 ounce | 6.8 | 34 |
| Sunflower oil, 1 tablespoon | 5.6 | 28 |
| Safflower oil, 1 tablespoon | 4.6 | 25 |
| Hazelnuts, dry roasted, 1 ounce | 4.3 | 22 |
| Peanut butter, 2 tablespoons | 2.9 | 15 |
| Peanuts, dry roasted, 1 ounce | 2.2 | 11 |
| Corn oil, 1 tablespoon | 1.9 | 10 |
| Spinach, boiled, ½ cup | 1.9 | 10 |
| Broccoli, chopped, boiled, ½ cup | 1.2 | 6 |
| Soybean oil, 1 tablespoon | 1.1 | 6 |
| Kiwifruit, 1 medium | 1.1 | 6 |
| Mango, sliced, ½ cup | 0.7 | 4 |
| Tomato, raw, 1 medium | 0.7 | 4 |
| Spinach, raw, 1 cup | 0.6 | 3 |
Footnote: *DV = Daily Value. DVs were developed by the FDA to help consumers compare the nutrient content of different foods within the context of a total diet. The DV for vitamin E is 30 IU (approximately 20 mg of natural alpha-tocopherol) for adults and children age 4 and older. However, the FDA does not require food labels to list vitamin E content unless a food has been fortified with this nutrient. Foods providing 20% or more of the DV are considered to be high sources of a nutrient, but foods providing lower percentages of the DV also contribute to a healthful diet.
[Source 234 ]
Are you getting enough vitamin E?
The diets of most Americans provide less than the recommended amounts of vitamin E. Nevertheless, healthy people rarely show any clear signs that they are not getting enough vitamin E. Between 1988 and 1994, the US National Health and Nutrition Examination Survey 3 (NHANES 3) examined the dietary intake and blood concentrations of alpha-tocopherol in 16,295 adults. The study reported that about one-third of all participants had blood concentrations of alpha-tocopherol below 20 micromoles/liter (μmol/L) — a cutoff value chosen because of its initial association with an increased risk for cardiovascular disease 235. More recent data from 18,063 participants in NHANES 2003-2006 indicated an average dietary intake of alpha-tocopherol from food including enriched and fortified sources among Americans adults of 7.2 mg/day 236. This intake is well below the current recommended dietary allowance (RDA) of 15 mg/day 9. At this level of dietary intake, more than 93% of American adults do not meet the estimated average requirement (EAR) of 12 mg/day for vitamin E 236.
What happens if you don’t get enough vitamin E?
Vitamin E deficiency is very rare in healthy people. It is almost always linked to certain diseases where fat is not properly digested or absorbed. Examples include Crohn’s disease, cystic fibrosis, and certain rare genetic diseases such as abetalipoproteinemia and ataxia with vitamin E deficiency (AVED) 237, 238. Vitamin E needs some fat for the digestive system to absorb it.
Vitamin E deficiency can cause nerve and muscle damage that results in loss of feeling in the arms and legs, loss of body movement control, muscle weakness, and vision problems. Another sign of deficiency is a weakened immune system.
Vitamin E deficiency
Dietary vitamin E deficiency is common in developing countries due to malnutrition; vitamin E deficiency among adults in developed countries is uncommon and usually due to fat malabsorption, liver failure, and digestive tract diseases, which impair the absorption of dietary fats and therefore fat-soluble vitamins like vitamin E 25, 2, 239, 240. Severe vitamin E deficiency has been associated with specific genetic defects affecting the transport of alpha-tocopherol by α-tocopherol transfer protein (α-TTP) and lipoproteins 240.
Premature babies of very low birth weight (<1,500 grams) might be deficient in vitamin E. Vitamin E supplementation in these infants might reduce the risk of some complications, such as those affecting the retina, but they can also increase the risk of infections 241. In addition, a recent nested case-control study in Bangladeshi women suggested that inadequate vitamin E status during early pregnancy may be associated with an increased risk of miscarriage 242.
Because the digestive tract requires fat to absorb vitamin E, people with fat-malabsorption disorders are more likely to become deficient than people without such disorders 2, 239, 240. Vitamin E deficiency symptoms include peripheral neuropathy, ataxia, skeletal myopathy, retinopathy, and impairment of the immune response 25, 243. People with Crohn’s disease, cystic fibrosis, or an inability to secrete bile from the liver into the digestive tract, for example, often pass greasy stools or have chronic diarrhea; as a result, they sometimes require water-soluble forms of vitamin E, such as tocopheryl polyethylene glycol-1000 succinate 244.
Some people with abetalipoproteinemia, a rare inherited disorder resulting in poor absorption of dietary fat, require enormous doses of supplemental vitamin E (approximately 100 mg/kg or 5–10 g/day) 244. Vitamin E deficiency secondary to abetalipoproteinemia causes such problems as poor transmission of nerve impulses, muscle weakness, and retinal degeneration that leads to blindness 245. Ataxia and vitamin E deficiency (AVED) is another rare, inherited disorder in which the liver’s alpha-tocopherol transfer protein (α-TTP) is defective or absent 237, 238. People with AVED have such severe vitamin E deficiency that they develop nerve damage and lose the ability to walk unless they take large doses of supplemental vitamin E 245, 237, 238.
Vitamin E deficiency causes fragility of red blood cells and degeneration of neurons, particularly peripheral axons and posterior column neurons.
The main symptoms of vitamin E deficiency are hemolytic anemia and neurologic deficits. Diagnosis is based on measuring the ratio of plasma alpha-tocopherol to total plasma lipids; a low ratio suggests vitamin E deficiency. Treatment consists of oral vitamin E, given in high doses if there are neurologic deficits or if deficiency results from malabsorption.
Vitamin E deficiency causes
In developed countries, it is unlikely that vitamin E deficiency occurs due to diet intake insufficiency and the more common causes are below 246:
- In developing countries, the most common cause is inadequate intake of vitamin E.
- Premature low birth weight infants with a weight less than 1500 grams (3.3 pounds)
- Mutations in the alpha-tocopherol transfer protein (TTPA) gene causing impaired fat metabolism 247
- Disrupted fat malabsorption as the small intestine requires fat to absorb vitamin E
- Patients with cystic fibrosis fail to secrete pancreatic enzymes to absorb vitamins A, D, E, and K
- Short-bowel syndrome patients may take years to develop symptoms. Short-bowel syndrome may develop from intestinal pseudo-obstruction, surgical resection, or mesenteric vascular thrombosis. Only after 10-20 years of malabsorption do neurologic symptoms become clinically apparent.
- Chronic cholestatic hepatobiliary disease leads to a decrease in bile flow and micelle formation that is needed for vitamin E absorption. Chronic cholestatic hepatobiliary disease in infants as young as 2 years may result from this condition 248. Prior to age 1 year, neurologic function was normal in all children. Between ages 1 and 3 years, neurologic abnormalities were present in approximately 50% of the vitamin E-deficient children; after age 3 years, neurologic abnormalities were present in all vitamin E-deficient children. Areflexia was the first abnormality to develop between ages 1 and 4 years; truncal and limb ataxia, peripheral neuropathy, and ophthalmoplegia developed between ages 3 and 6 years. Neurologic dysfunction progressed to a disabling combination of findings by ages 8 to 10 years in the majority of vitamin E-deficient children. Neurologic findings are less frequent in adult patients with cholestasis secondary to cirrhosis.
- Crohn’s disease, exocrine pancreatic insufficiency, and liver disease may all not absorb fat
- Abetalipoproteinemia is a rare genetic, autosomal-recessive disease that causes an error in lipoprotein production and transportation. Infants present with steatorrhea from the time of birth. Patients have pigmented retinopathy and progressive ataxia, and they develop acanthosis of red blood cells in the first decade of life.
- Ataxia with vitamin E deficiency (AVED) also known as Familial Isolated Vitamin E Deficiency or Friedreich-like ataxia with vitamin E deficiency, is an autosomal recessive neurodegenerative disorder caused by alpha-tocopherol transfer protein (TTPA) gene mutations on chromosome 8q13 (long arm of chromosome 8) 249. Neurologic findings develop within the first decade of life, and no clinical findings distinguish deficiency from ataxia and movement disorders. Vitamin E replacement can significantly influence the outcome; therefore, screening for vitamin E deficiency is beneficial for patients with movement disorders or neuropathies that are of unknown cause.
Cigarette smoking is thought to increase the utilization of alpha-tocopherol such that smokers might be at increased risk of vitamin E deficiency compared with nonsmokers 123. Also, the 19-year follow-up analysis of the Alpha-Tocopherol, Beta-Carotene cancer (ATBC) trial in older, male smokers indicated that participants in the highest versus lowest quintile of serum alpha-tocopherol concentrations (>31 μmol/L vs. <23 μmol/L) at baseline had reduced risks of total and cause-specific mortality 250.
Ataxia with Vitamin E Deficiency (AVED)
Ataxia with vitamin E deficiency (AVED), also known as Familial Isolated Vitamin E Deficiency or Friedreich-like ataxia with vitamin E deficiency, is a rare progressive neurodegenerative disorder of autosomal recessive cerebellar ataxia (ARCA) 251, 252, 253, 254, 255, 256. The most prominent symptoms of ataxia with vitamin E deficiency (AVED) include progressive cerebellar ataxia, absence of neurologic reflexes (areflexia), peripheral neuropathy and movement disorder 254. Ataxia with vitamin E deficiency (AVED) patients may also have retinitis pigmentosa (a group of rare eye diseases that affect the retina that causes severe vision impairment such as decreased vision at night or in low light and loss of side vision (tunnel vision)), a disease of the heart muscle (cardiomyopathy) and scoliosis (a sideways curvature of the spine) 257, 258. Ataxia with vitamin E deficiency (AVED) is caused by mutations in the TTPA gene, which encodes the alpha-tocopherol transfer protein (α-TTP), which in turn binds alpha-tocopherol and transports vitamin E from liver cells to circulating lipoproteins 259. Vitamin E supplementation may prevent the worsening of the condition of patients with ataxia with vitamin E deficiency (AVED).
Ataxia with vitamin E deficiency (AVED) affects both males and females equally. Ataxia with vitamin E deficiency (AVED) is estimated to occur in fewer than 1 in 1,000,000 people 260. North African populations are most affected with AVED. Other cases have been reported in the Mediterranean region and Northern European countries. There have been cases in Asian countries such as Japan, China and the Philippines. The onset of AVED can occur during childhood or adulthood with cases reported ranging in children as young as 2 and adults as old as 52. Typically, the disease presents in individuals between ages 5 and 20 years. The disorder was first described in the medical literature in 1981.
Untreated ataxia with vitamin E deficiency (AVED) generally manifests between ages 5 and 15 years 253. The first signs and symptoms include progressive ataxia, clumsiness of the hands, loss of proprioception, and absence of neurologic reflexes (areflexia). Other features often observed are dysdiadochokinesia, dysarthria, positive Romberg sign, head titubation, decreased visual acuity, and positive Babinski sign 253. Although age of onset and disease course are more uniform within a given family, disease manifestations and their severity can vary even among siblings.
Ataxia with vitamin E deficiency (AVED) is caused by alpha-tocopherol transfer protein (TTPA) gene mutations on chromosome 8q13 (long arm of chromosome 8), which encodes the alpha-tocopherol transfer protein (α-TTP), which in turn binds alpha-tocopherol and transports vitamin E from liver cells to circulating lipoproteins 261, 262, 263, 264, 265, 266, 267, 268, 269. When the alpha-tocopherol transfer protein (TTPA) gene is damaged, vitamin E cannot be distributed throughout the body. Vitamin E is important because it protects the cells of the neurological system (neurons) from damaging molecules called free radicals. Ataxia with vitamin E deficiency (AVED) is inherited in an autosomal recessive manner 259.
Marriotti et al. 270 reported cerebellar atrophy in some individuals with AVED. Spinal sensory demyelination with neuronal atrophy and axonal spheroids and neuronal lipofuscin accumulation in the third cortical layer of the cerebral cortex, thalamus, lateral geniculate body, spinal horns, and posterior root ganglia are the commonest histopathology findings 271. The following criteria should be met to diagnose ataxia with vitamin E deficiency (AVED) 272:
- Friedreich ataxia‐like neurologic phenotype
- Markedly reduced plasma vitamin E
- Normal lipoprotein profile
- Exclusion of disease that cause fat malabsorption.
Individuals with AVED are treated with high doses of vitamin E supplements, but recovery is slow and incomplete 273. Early diagnosis and treatment can slow down or stop the progression of the disorder and in some people, even improve existing symptoms. Lifelong treatment with vitamin E will be needed. Genetic counseling is recommended for affected individuals and their families.
Vitamin E supplementation should be started as soon as possible, but the optimal dosage is still variable 272. The doses and the route of vitamin E supplementation depend on the causes of vitamin E deficiency. The treatment of choice for AVED is lifelong high‐dose oral vitamin E supplementation. The early use of vitamin E supplementation in the disease process helps to reverse some neurological symptoms like ataxia and intellectual deterioration 274.
Figure 6. Ataxia with vitamin E deficiency (AVED) pathophysiology
[Source 272 ]Ataxia with Vitamin E Deficiency (AVED) causes
Ataxia with vitamin E deficiency (AVED) is caused by changes (mutations or pathogenic variants) in the TTPA gene, which provides instructions to synthesize the alpha-tocopherol transfer protein (αTTP) . This protein controls the distribution and transportation of vitamin E from the liver to other cells and tissues throughout the body, including the brain. Individuals with AVED have non-working genes for TTPA and therefore, vitamin E cannot be properly distributed throughout the body especially to the brain where it is necessary for proper function. AVED is characterized by very low levels of vitamin E in the blood and tissues in the brain. Without normal levels of vitamin E in the tissues, an individual with AVED experiences damage to the body from lack of protection from damaging free radicals.
Vitamin E deficiency often occurs secondary to disorders that impair the absorption of vitamin E from fat including liver disorders, disorders of fat metabolism and disorders of bile secretion. These disorders include choleostasis (a syndrome of various causes characterized by impaired bile secretion), cystic fibrosis (primarily a lung disorder that results in choleostasis), primary biliary cirrhosis (a liver disorder that results in choleostasis) and abetalipoproteinemia (a digestive disorder characterized by fat malabsorption). Premature infants may have low vitamin E levels due to small amounts of vitamin E cross the placenta. In rare cases vitamin E deficiency may be caused due to poor diet. (For more information on the above disorders, choose the specific disorder name as your search term in the Rare Disease Database.)
Ataxia with vitamin E deficiency (AVED) is inherited, or passed down from parent to child, in an autosomal recessive manner. Recessive genetic disorders occur when an individual inherits a non-working gene from each parent. If an individual receives one working gene and one non-working gene for the disease, the person will be a carrier for the disease, but usually will not show symptoms. The risk for two carrier parents to both pass the non-working gene and, therefore, have a child with AVED is 25% with each pregnancy. The risk of having a child who is a carrier, like the parents, is 50% with each pregnancy. The chance for a child to receive working genes from both parents is 25%. The risk is the same for males and females.
Ataxia with Vitamin E Deficiency (AVED) signs and symptoms
Individuals usually show symptoms between 5 and 20 years of age. Symptoms and the severity of ataxia with vitamin E deficiency (AVED) may be different from person to person. Without treatment, symptoms may get worse as the person grows older.
Untreated individuals with Ataxia with Vitamin E Deficiency (AVED)
| Signs and symptoms | % of Persons with signs and symptoms | ||
|---|---|---|---|
| Cerebellar involvement | Gait impairment | 93.4% | |
| Dysarthria | 61.8% | ||
| Head titubation | 33% | ||
| Nystagmus | 5.3% | ||
| Lower motor neuron involvement | Areflexia | 94.7% | |
| Deep sensory disturbances | 67.1% | ||
| Upper motor neuron involvement | Babinski sign | 85.5% | |
| Urinary urgency | 22.4% | ||
| Pigmentary retinopathy | 2.3% | ||
| Cardiomyopathy | 1.5% | ||
Footnote: Frequency of signs and symptoms associated with untreated individuals with ataxia with vitamin E deficiency (AVED) based on findings in 132 individuals of North African heritage.
[Source 255 ]AVED affects the brain and spinal cord (central nervous system or CNS) as well as the motor and sensory nerves that connect the central nervous system (CNS) to the rest of the body (peripheral nervous system) 260. This results in ataxia, which is difficulty controlling body movements and numbness of the hands and feet (peripheral neuropathy). Individuals with AVED develop increasing weakness of the legs, which may appear as an unsteady or staggering way of walking (gait) or trembling when standing still 260. If symptoms become very severe, an individual with AVED may require a wheelchair if they cannot walk 260. Clinical symptoms like progressive ataxia with decreased reflex remain the prominent symptoms of AVED. According to the study done in North Africa, most of the individuals with AVED had mild neuropathy and were mostly combined types of neuropathies (both sensory and motor) 275.
Additional symptoms related to the central nervous system (CNS) include loss of proprioception, which is an awareness of joint position in relation to other parts of the body 260. With time, reflexes in the legs may slow down or be absent (areflexia). Involvement of the throat muscles may lead to impaired swallowing or choking and may cause difficulty in eating. Slurred speech or difficulty speaking (dysarthria) may also be present. Some affected individuals may develop a tremor or shaking of the head (titubation). Intellect and emotions are rarely affected.
In some people with AVED, psychotic episodes and intellectual decline have been described 253. In rare individuals, school performance declines secondary to loss of intellectual capacities 253.
In addition to neurological symptoms, individuals with AVED may develop symptoms affecting other systems of the body including the eyes 260. Retinitis pigmentosa (RP) is a large group of rare eye diseases that cause progressive degeneration of the membrane lining the eyes (retina). This results in visual impairment or decreased vision. A high percentage of affected individuals (e.g., 8/11 individuals in one series) experience decreased visual acuity 258. Some affected individuals may have yellow “fatty” deposits (xanthelasma) in the retina.
Affected individuals may also develop sideways curvature of the spine (scoliosis), degenerative changes of the heart muscle (cardiomyopathy) or “fatty” deposits (xanthomas) affecting the Achilles tendon around the ankle 260. Some individuals with ataxia with vitamin E deficiency (AVED) may experience a form of dystonia. Dystonia is the name for a group of movement disorders generally characterized by involuntary muscle contractions that force the body into abnormal, sometimes painful, movements and positions (postures).
Ataxia with Vitamin E Deficiency (AVED) diagnosis
A diagnosis of ataxia with vitamin E deficiency (AVED) is made upon a thorough clinical evaluation, a detailed patient history, and a variety of tests and characteristic findings (e.g., low levels of vitamin E with normal levels of lipoproteins and lipids with no evidence of fat malabsorption and abnormalities in the TTPA gene).
AVED should be suspected in an individual with the following clinical and laboratory findings and family history 253:
- Family history
- Family history is consistent with autosomal recessive inheritance (e.g., affected sibs and/or parental consanguinity). Absence of a known family history does not preclude the diagnosis.
- Clinical features
- Onset between ages 5 and 15 years
- Progressive cerebellar findings including the following:
- Gait ataxia
- Clumsiness of the hands
- Loss of proprioception (especially distal joint position and vibration sense)
- Dysdiadochokinesia
- Positive Romberg sign
- Head titubation
- Lower motor neuron involvement. Areflexia
- Upper motor neuron involvement. Positive Babinski sign
- Ophthalmologic involvement. Decreased visual acuity due to macular degeneration, pigmentary retinopathy
- Supportive laboratory findings
- Normal lipid and lipoprotein profile
- Very low plasma vitamin E (alpha-tocopherol, or α-tocopherol) concentration
- Note: There is no universal normal range of plasma vitamin E concentration, as it depends on the test method and varies among laboratories.
- In Finckh et al 276, the plasma vitamin E (alpha-tocopherol, or α-tocopherol) concentration normal range lies between 9.0 and 29.8 µmol/L. In El Euch-Fayache et al 255, the normal range is given as 16.3-34.9 µmol/L, while individuals with AVED had vitamin E levels between 0.00 and 3.76 µmol/L (mean 0.95 µmol/L). In individuals with AVED, the plasma vitamin E concentration is generally lower than 4.0 µmol/L (<1.7 mg/L) 264, 262.
- Because oxidation of alpha-tocopherol (α-tocopherol) by air may invalidate test results, the following precautions with a blood sample should be taken:
- Centrifugation of the EDTA blood soon after venipuncture
- Quick separation of plasma from blood cells after centrifugation and subsequent flash freezing of the plasma in liquid nitrogen
- Filling the space above the plasma with an inert gas (e.g., argon or nitrogen)
- Protecting the sample from light by wrapping the container in aluminum foil, or using a black or light-shielded Eppendorf tube
- Shipment of the sample to the test laboratory in dry ice
- Electrophysiologic findings
- No electrophysiologic findings (motor nerve conduction velocities, compound muscle action potentials, or nerve sensory action potentials) are specific to or diagnostic of AVED; even the presence of a severe neuropathy does not exclude the diagnosis of AVED.
- Somatosensory evoked potentials show increased central conduction time between the segment C1 (N13b) and the sensorimotor cortex (N20) and increased latencies of the N20 (median nerve) and P40 (tibial nerve) waves. The P40 wave may be missing completely 274.
- Neuroimaging
The diagnosis of ataxia with vitamin E deficiency (AVED) is established by molecular genetic testing with the identification of abnormalities in the TTPA gene 253.
Figure 7. Ataxia with vitamin E deficiency (AVED) diagnosis
[Source 272 ]Ataxia with Vitamin E Deficiency (AVED) treatment
Individuals with ataxia with vitamin E deficiency (AVED) are treated with high doses of vitamin E supplements. Early diagnosis and treatment can slow down or stop the progression of the disorder and in some people, even improve existing symptoms. The treatment of choice for ataxia with vitamin E deficiency (AVED) is lifelong high-dose oral vitamin E supplementation. With treatment, plasma vitamin E concentrations can become normal. Genetic counseling is recommended for affected individuals and their families.
One of the following vitamin E preparations is used 253:
- The chemically manufactured racemic form, all-rac-alpha-tocopherol acetate
- The naturally occurring form, RRR-alpha-tocopherol
- It is currently unknown whether affected individuals should be treated with all-rac-alpha-tocopherol acetate or with RRR-alpha-tocopherol. It is known that alpha-tocopherol transfer protein (α-TTP) stereoselectively binds and transports 2R-alpha-tocopherols 278, 279, 280. For some TTPA pathogenic variants, this stereoselective binding capacity is lost and affected individuals cannot discriminate between RRR- and SRR-alpha-tocopherol 266, 264. In this instance, affected individuals would also be able to incorporate non-2R-alpha-tocopherol stereoisomers into their bodies if they were supplemented with all-rac-alpha-tocopherol. Since potential side effects of the synthetic stereoisomers have not been studied in detail, it seems appropriate to treat with RRR-alpha-tocopherol, despite the higher cost.
When vitamin E treatment is initiated in presymptomatic individuals (e.g., younger age), manifestations of ataxia with vitamin E deficiency (AVED) do not develop 281, 255.
No large-scale therapeutic studies have been performed to determine optimal vitamin E dosage and to evaluate outcomes 253. The reported vitamin E dose ranges from 800 mg to 1,500 mg (or 40 mg/kg body weight in children) 282, 283, 281, 264, 274, 284, 285, 262. The ideal dose of vitamin E ranges from 800 mg to 1500 mg (40 mg/kg/day) in children 270, 273.
Periodic follow‐up is required during vitamin E therapy. The plasma concentration of vitamin E should be measured in regular intervals, usually, every 6 months to maintain the level of vitamin E in the high normal range especially in children. Ideally the plasma vitamin E concentration should be maintained in the high-normal range 253.
Individuals with AVED should avoid smoking and occupations requiring quick responses or good balance. Smoking reduces the total radical trapping antioxidant parameter of plasma (TRAP), which is regarded as the best prognostic marker during the supplementation of vitamin E, leading to the reduction in plasma vitamin level 286.
Symptomatic individuals
Some clinical signs and symptoms (e.g., ataxia and intellectual deterioration) can be reversed in symptomatic individuals if treatment is initiated early in the disease process 274.
In older individuals, disease progression can be stopped, but deficits in proprioception and gait unsteadiness generally remain 285, 262, 255.
Supportive Care
The goals of supportive care in those with clinical signs and symptoms of AVED are to maximize function and reduce complications. Depending on the clinical signs and symptoms, it is recommended that each individual be managed by a multidisciplinary team of relevant specialists such as neurologists, occupational therapists, physical therapists, physiatrists, orthopedists, nutritionists, speech-language pathologists, pulmonologists, and mental health specialists 253.
Pregnancy Management
Because reduced vitamin E levels are associated with low fertility and embryo resorption in mice 287 and α-tocopherol transfer protein is highly expressed in the human placenta 288, it is advisable for women with AVED to maintain vitamin E levels in the high-normal range during pregnancy 253.
Ataxia with Vitamin E Deficiency (AVED) prognosis
Ataxia with Vitamin E Deficiency (AVED) prognosis despite the supplementation of vitamin E depends on the timing of the supplementation and the age of the onset of vitamin E deficiency. Vitamin E treatment in pre‐symptomatic individuals with a history of family cases of AVED can help to prevent the individual from primary manifestations. The symptoms do not develop in the individuals with the initiation of vitamin E earlier in pre‐symptomatic individuals who are at risk 289. Older individuals usually remain deficient in proprioception and gait unsteadiness although the progression of the disease can be stopped with vitamin E supplementation 275.
Vitamin E deficiency signs and symptoms
Severe vitamin E deficiency results mainly in neurologic symptoms, including impaired balance and coordination (spinocerebellar ataxia), injury to the sensory nerves (peripheral neuropathy), muscle weakness (myopathy), and damage to the retina of the eye (retinopathy) 6. For this reason, people who develop peripheral neuropathy, ataxia, or retinitis pigmentosa of unknown causes should be screened for vitamin E deficiency 240. Severe vitamin E deficiency individuals may present with profound muscle weakness and visual-field constriction 290. Patients with severe, prolonged vitamin E deficiency may develop complete blindness, cardiac arrhythmia, and dementia 290.
The results of one randomized controlled trial in 601 patients with common forms of retinitis pigmentosa indicated that daily supplementation with 400 IU of all-rac-alpha-tocopherol (180 mg of RRR-α-tocopherol) modestly but significantly increased the loss of retinal function 291. In contrast, daily supplementation with 15,000 IU of vitamin A (4,500 μg RAE) significantly slowed the loss of retinal function over a period of four to six years, suggesting that patients with common forms of retinitis pigmentosa may benefit from long-term vitamin A supplementation but should avoid high-dose supplemental vitamin E 291.
Inherited defects in alpha-tocopherol transfer protein (α-TTP) are associated with a characteristic syndrome called Ataxia with Vitamin E Deficiency (AVED). A recent case study reported that visual impairment in a middle-age patient with AVED was caused by both retinitis pigmentosa and early-onset macular degeneration 292. Supplementation with high-dose vitamin E (800-1,200 mg/day) is used to prevent neurologic deterioration in AVED subjects 240.
The developing nervous system appears to be especially vulnerable to vitamin E deficiency. For instance, children with severe vitamin E deficiency at birth rapidly experience irreversible neurologic symptoms if not treated with vitamin E. In contrast, individuals who develop gastrointestinal disorders affecting vitamin E absorption in adulthood may not develop neurologic symptoms for 10-20 years 240. It should also be noted that neurologic symptoms caused by vitamin E deficiency have not been reported in healthy individuals who consume diets low in vitamin E 6. In addition, a recent nested case-control study in Bangladeshi women suggested that inadequate vitamin E status during early pregnancy may be associated with an increased risk of miscarriage 242.
If vitamin E deficiency is expected, a full neurological exam is recommended as well as a standard physical exam. Patients presenting early may show hyporeflexia, decreased night vision, loss/decreased vibratory sense, however, have normal cognition. A more moderate stage of this deficiency may show limb and truncal ataxia, profuse muscle weakness, and limited upward gaze. Late presentations may show cardiac arrhythmias and possible blindness with reduced cognition. Ataxia is the most common exam finding.
Patients that have abetalipoproteinemia have eye problems often including pigmented retinopathy and visual field issues. However, patients suffering from cholestatic liver disease often have personality and behavioral disorders.
Vitamin E deficiency diagnosis
A low alpha-tocopherol level or low ratio serum alpha-tocopherol to serum lipids measurement is the mainstay of diagnosis. In adults, alpha-tocopherol levels should be less than 5 mcg/mL. In an adult with hyperlipidemia, the abnormal lipids may affect the vitamin E levels and a serum alpha-tocopherol to lipids level, needing to be less than 0.8 mg/g) is more accurate. A pediatric patient with abetalipoproteinemia will have serum alpha-tocopherol levels that are not detectable.
Vitamin E deficiency treatment
Treatment addresses the underlying cause of the deficiency (fat malabsorption, fat metabolism disorders, among others) and then provide oral vitamin E supplementation 246. Also, a modification in diet can assist in vitamin E supplementation, by increasing intake of leafy vegetables, whole grains, nuts, seeds, vegetable oils and fortified cereals is highly recommended. Though normally presented in our diets, adults need 15 mg of vitamin E per day. A supplement of 15 to 25 mg/kg once per day or mixed tocopherols 200 IU can both be used. If a patient has issues with the small intestine and/or oral ingestion intramuscular vitamin E injection is necessary 293, 294, 295, 296. The recommended daily allowance of alpha-tocopherol is as follows 246, 297:
- Age 0 to 6 months: 3 mg
- Age 6 to 12 months: 4 mg
- Age 1 to 3 years: 6 mg
- Age 4 to 10 years: 7 mg
- Adults and elderly patients: 8 to 10 mg
Replacement recommendations vary by the disease and are as follows 298, 297:
- Abetalipoproteinemia: 100 to 200 IU/kg per day
- Chronic cholestasis: 15 to 25 IU/kg per day
- Cystic fibrosis: 5 to 10 IU/kg per day
- Short-bowel syndrome: 200 to 3600 IU per day
- Isolated vitamin E deficiency: 800 to 3600 IU per day
Larger doses are required in short-bowel syndrome and isolated vitamin E deficiency state.
The recommended doses of vitamin E for age group and associated causes are shown in Table 3.
Table 3. Recommended doses of vitamin E according to age group and associated causes
| Age group | Associated symptoms | Dose and route of supplementation |
|---|---|---|
| Infants and children | Cholestasis |
|
| Adults | Fat malabsorption |
|
| Any age group | Severe cholestasis/genetic disorders related to transport of vitamin E |
|
Vitamin E deficiency prognosis
If left untreated, vitamin E deficiency symptoms may worsen. However, once diagnosed, the outcome of vitamin E deficiency is very good as most symptoms will resolve quickly. However, as vitamin E deficiency becomes more pronounced, the therapy will be more restricted. Patients who are at risk for vitamin E deficiency should undergo a thorough neurologic examination, as well as periodic testing of serum vitamin E levels 245. The results of a systematic review by Sherf-Dagan et al suggest that patients who undergo weight loss surgery (bariatric surgery), particularly malabsorptive procedures, are at greater risk for the development of vitamin E deficiency 301.
Vitamin E Side Effects and Toxicity
Research has not found any adverse effects from consuming vitamin E in food 25. However, high doses of alpha-tocopherol supplements can cause hemorrhage, especially in patients already on anticoagulation or antiplatelet therapy and interrupt blood coagulation in animals, and in vitro data suggest that high doses inhibit platelet aggregation. Two clinical trials have found an increased risk of hemorrhagic stroke in participants taking alpha-tocopherol; one trial included Finnish male smokers who consumed 50 mg/day for an average of 6 years 302 and the other trial involved a large group of male physicians in the United States who consumed 400 IU every other day for 8 years 303. Because the majority of physicians in the latter study were also taking aspirin, this finding could indicate that vitamin E has a tendency to cause bleeding.
Bleeding episodes can occur anywhere in the body, and serious life-threatening hemorrhagic strokes have been reported. Other vitamin E toxicity complications include gastrointestinal manifestations, weakness, fatigue, and emotional lability. The treatment for vitamin E toxicity includes discontinuation of vitamin E supplementation and consideration of vitamin K therapy if serious bleeding occurs. To prevent vitamin E toxicity, supplementation of vitamin E should be kept to a lower dosage.
The Food and Nutrition Board at the Institute of Medicine of The National Academies has established Tolerable Upper Intake Levels (ULs – the maximum daily intake unlikely to cause adverse health effects) for vitamin E based on the potential for hemorrhagic effects (see Table 4). The Tolerable Upper Intake Levels (ULs) apply to all forms of supplemental alpha-tocopherol, including the eight stereoisomers present in synthetic vitamin E. Doses of up to 1,000 mg/day (1,500 IU/day of the natural form or 1,100 IU/day of the synthetic form) in adults appear to be safe, although the data are limited and based on small groups of people taking up to 3,200 mg/day of alpha-tocopherol for only a few weeks or months. Long-term intakes above the Tolerable Upper Intake Level (UL) increase the risk of adverse health effects 25. Vitamin E Tolerable Upper Intake Levels (ULs) for infants have not been established.
Table 4. Tolerable Upper Intake Levels (ULs) for Vitamin E
| Age | Male | Female | Pregnancy | Lactation |
| 1–3 years | 200 mg (300 IU) | 200 mg (300 IU) | ||
| 4–8 years | 300 mg (450 IU) | 300 mg (450 IU) | ||
| 9–13 years | 600 mg (900 IU) | 600 mg (900 IU) | ||
| 14–18 years | 800 mg (1,200 IU) | 800 mg (1,200 IU) | 800 mg (1,200 IU) | 800 mg (1,200 IU) |
| 19+ years | 1,000 mg (1,500 IU) | 1,000 mg (1,500 IU) | 1,000 mg (1,500 IU) | 1,000 mg (1,500 IU) |
Can vitamin E be harmful?
Eating vitamin E in foods is not risky or harmful 304. A patient who consumes vitamin E in their diet has, on average, a level of circulating alpha-tocopherol of approximately 20 micromol/L. Patients that have additional vitamin E supplementation have levels of 30 micromol/L or greater.
In supplement form, however, high doses of vitamin E might increase the risk of bleeding (by reducing the blood’s ability to form clots after a cut or injury) and of serious bleeding in the brain (known as hemorrhagic stroke) 305. High doses of alpha-tocopherol supplements can cause hemorrhage and interrupt blood coagulation in animals and test tube studies data suggest that high doses inhibit platelet aggregation. Two clinical trials have found an increased risk of hemorrhagic stroke in participants taking alpha-tocopherol; one trial included Finnish male smokers who consumed 50 mg/day for an average of 6 years 306 and the other trial involved a large group of male physicians in the United States who consumed 400 IU (180 mg) of synthetic vitamin E every other day for 8 years 142. Because the majority of physicians in the latter study were also taking aspirin, this finding could indicate that vitamin E has a tendency to cause bleeding.
Because of this risk, the upper limit for adults is 1,500 IU/day for supplements made from the natural form of vitamin E and 1,100 IU/day for supplements made from synthetic vitamin E. The upper limits for children are lower than those for adults. Some research suggests that taking vitamin E supplements even below these upper limits might cause harm. In one study, for example, men who took 400 IU of vitamin E each day for several years had an increased risk of prostate cancer.
Two meta-analyses of randomized trials have also raised questions about the safety of large doses of vitamin E, including doses lower than the Tolerable Upper Intake Level (UL). These meta-analyses linked supplementation to small but statistically significant increases in all-cause mortality. One analysis found an increased risk of death at doses of 400 IU/day (form not specified), although the risk began to increase at 150 IU 307. In the other analysis of studies of antioxidant supplements for disease prevention, the highest quality trials revealed that vitamin E, administered singly (dose range 10 IU–5,000 IU/day; mean 569 IU [form not specified]) or combined with up to four other antioxidants, significantly increased mortality risk 308.
The implications of these analyses for the potential adverse effects of high-dose vitamin E supplements are unclear 7. Participants in the studies included in these analyses were typically middle-aged or older and had chronic diseases or related risk factors. These participants often consumed other supplements in addition to vitamin E. Some of the studies analyzed took place in developing countries in which nutritional deficiencies are common. A review of the subset of studies in which vitamin E supplements were given to healthy individuals for the primary prevention of chronic disease found no convincing evidence that the supplements increased mortality 309.
However, results from the recently published, large SELECT trial show that vitamin E supplements (400 IU/day [180 mg] as dl-alpha-tocopheryl acetate) may harm adult men in the general population by increasing their risk of prostate cancer 310. Follow-up studies are assessing whether the cancer risk was associated with baseline blood levels of vitamin E and selenium prior to supplementation as well as whether changes in one or more genes might increase a man’s risk of developing prostate cancer while taking vitamin E.
Vitamin E toxicity complications
Although the major hazardous complications of elevated vitamin E levels include bleeding, there have been others mentioned. These include thyroid problems, weakness, emotional disorder, gastrointestinal derangement, tenderness of breasts, and thrombophlebitis 305.
Vitamin E toxicity diagnosis
To detect vitamin E toxicity, serum levels of circulating alpha-tocopherol can be obtained. The average range of plasma alpha-tocopherol in a patient that eats a well-balanced diet is 20 micromoles/liter 311. A patient on vitamin E supplementation may have plasma levels of 30 micromoles/liter or greater 311. The normal lab range for circulating alpha-tocopherols is 5.7 to 19.9 mg/L 312. The levels of circulating alpha-tocopherol are very dependent on the lipid content of the blood. In patients with extremely high or extremely low cholesterol levels, the levels of circulating alpha-tocopherol are not an accurate measure of vitamin E. In a patient with average cholesterol levels, the levels of circulating alpha-tocopherol are still not an accurate measure of vitamin E. This is due to the upregulation of biliary and urinary excretion once vitamin E levels are increased in the body 313. Because of these irregularities in vitamin E metabolism, there is no set cut-off level of circulating alpha-tocopherols considered universally toxic.
In a study performed on patients with intracranial hemorrhages and taking vitamin E supplementation, alpha-tocopherol levels ranged from 23.3 micromoles/L to 46.7 micromoles/L in patients that were discovered to have intracranial hemorrhages 312. In another study that correlated the vitamin E levels and risk of bleeding in patients taking oral anticoagulation, a ratio of circulating alpha-tocopherols to total serum cholesterol concentration was used. This was thought to most accurately represent the true circulating vitamin E levels 126. Although there is a concern regarding the reliability of these circulating levels of alpha-tocopherols correlating to vitamin E levels, this is still the most widely used test in the literature regarding quantifying vitamin E when describing its effects.
Vitamin E toxicity treatment
The mainstay treatment for vitamin E toxicity is stopping the exogenous vitamin supplementation. This is effective since vitamin E toxicity does not occur unless there is an exogenous supplementation 304. If there is significant bleeding, vitamin K supplementation should be considered in patients taking vitamin E supplementation. There can be inhibition of a clotting cascade that is vitamin K dependent when there are higher concentrations of vitamin E. This can occur whether the patient is on warfarin or not. Vitamin E also impedes platelet aggregation. This can occur regardless of whether the patient takes antiplatelet agents. Therefore, giving vitamin K to patients who are actively bleeding or have a severe hemorrhage should be considered 126.
- Traber MG. Vitamin E. In: Shils ME, Shike M, Ross AC, Caballero B, Cousins R, eds. Modern Nutrition in Health and Disease. 10th ed. Baltimore, MD: Lippincott Williams & Wilkins, 2006;396-411.[↩][↩][↩][↩]
- Galli F., Azzi A., Birringer M., Cook-Mills J.M., Eggersdorfer M., Frank J., Cruciani G., Lorkowski S., Özer N.K. Vitamin E: Emerging Aspects and New Directions. Free Radic. Biol. Med. 2017;102:16–36. doi: 10.1016/j.freeradbiomed.2016.09.017[↩][↩][↩]
- Jiang Q. Natural forms of vitamin E: Metabolism, antioxidant, and anti-inflammatory activities and their role in disease prevention and therapy. Free Radic. Biol. Med. 2014;72:76–90. doi: 10.1016/j.freeradbiomed.2014.03.035[↩]
- Azzi A. Tocopherols, tocotrienols and tocomonoenols: Many similar molecules but only one vitamin E. Redox Biol. 2019 Sep;26:101259. doi: 10.1016/j.redox.2019.101259[↩][↩][↩]
- Suskind DL. Nutritional deficiencies during normal growth. Pediatr Clin North Am. 2009 Oct;56(5):1035-53. doi: 10.1016/j.pcl.2009.07.004[↩]
- Vitamin E. https://lpi.oregonstate.edu/mic/vitamins/vitamin-E[↩][↩][↩][↩][↩][↩][↩]
- Vitamin E. https://ods.od.nih.gov/factsheets/VitaminE-HealthProfessional[↩][↩][↩][↩]
- Traber MG. Vitamin E. In: Erdman JWJ, Macdonald IA, Zeisel SH, eds. Present Knowledge in Nutrition. 10th ed. Washington, D.C.: Wiley-Blackwell; 2012:214-229.[↩][↩][↩][↩][↩][↩][↩]
- National Academies of Sciences, Engineering, and Medicine. 2000. Dietary Reference Intakes for Vitamin C, Vitamin E, Selenium, and Carotenoids. Washington, DC: The National Academies Press. Chapter: 6 Vitamin E. https://doi.org/10.17226/9810[↩][↩][↩][↩][↩]
- Verhagen H, Buijsse B, Jansen E, Bueno-de-Mesquita B. The state of antioxidant affairs. Nutr Today 2006;41:244-50.[↩][↩]
- Kagan VE, Serbinova EA, Forte T, Scita G, Packer L. Recycling of vitamin E in human low density lipoproteins. J Lipid Res. 1992 Mar;33(3):385-97. https://www.jlr.org/article/S0022-2275(20)41529-9/pdf[↩]
- Trpkovic A, Resanovic I, Stanimirovic J, Radak D, Mousa SA, Cenic-Milosevic D, Jevremovic D, Isenovic ER. Oxidized low-density lipoprotein as a biomarker of cardiovascular diseases. Crit Rev Clin Lab Sci. 2015;52(2):70-85. doi: 10.3109/10408363.2014.992063[↩][↩]
- Traber MG. Vitamin E regulatory mechanisms. Annu Rev Nutr. 2007;27:347-62. doi: 10.1146/annurev.nutr.27.061406.093819[↩]
- Sen CK, Khanna S, Roy S. Tocotrienols: Vitamin E beyond tocopherols. Life Sci. 2006 Mar 27;78(18):2088-98. doi: 10.1016/j.lfs.2005.12.001[↩]
- Dietrich M, Traber MG, Jacques PF, Cross CE, Hu Y, Block G. Does gamma-tocopherol play a role in the primary prevention of heart disease and cancer? A review. J Am Coll Nutr. 2006 Aug;25(4):292-9. doi: 10.1080/07315724.2006.10719538[↩][↩]
- Merck Sharp & Dohme Corp., Merck Manual. Vitamin E (Tocopherol). https://www.merckmanuals.com/professional/nutritional-disorders/vitamin-deficiency,-dependency,-and-toxicity/vitamin-e[↩]
- Food Labeling: Revision of the Nutrition and Supplement Facts Labels. https://www.federalregister.gov/documents/2016/05/27/2016-11867/food-labeling-revision-of-the-nutrition-and-supplement-facts-labels[↩]
- Food Labeling: Revision of the Nutrition and Supplement Facts Labels and Serving Sizes of Foods That Can Reasonably Be Consumed at One Eating Occasion; Dual-Column Labeling; Updating, Modifying, and Establishing Certain Reference Amounts Customarily Consumed; Serving Size for Breath Mints; and Technical Amendments; Proposed Extension of Compliance Dates. https://www.federalregister.gov/documents/2017/10/02/2017-21019/food-labeling-revision-of-the-nutrition-and-supplement-facts-labels-and-serving-sizes-of-foods-that[↩]
- Bruno RS, Leonard SW, Park SI, Zhao Y, Traber MG. Human vitamin E requirements assessed with the use of apples fortified with deuterium-labeled alpha-tocopheryl acetate. Am J Clin Nutr. 2006 Feb;83(2):299-304. doi: 10.1093/ajcn/83.2.299[↩]
- Leonard SW, Good CK, Gugger ET, Traber MG. Vitamin E bioavailability from fortified breakfast cereal is greater than that from encapsulated supplements. Am J Clin Nutr. 2004 Jan;79(1):86-92. doi: 10.1093/ajcn/79.1.86[↩]
- Traber MG, Leonard SW, Bobe G, Fu X, Saltzman E, Grusak MA, Booth SL. α-Tocopherol disappearance rates from plasma depend on lipid concentrations: studies using deuterium-labeled collard greens in younger and older adults. Am J Clin Nutr. 2015 Apr;101(4):752-9. doi: 10.3945/ajcn.114.100966. Epub 2015 Mar 4. Erratum in: Am J Clin Nutr. 2017 Feb;105(2):543.[↩]
- Galli F, Bonomini M, Bartolini D, Zatini L, Reboldi G, Marcantonini G, Gentile G, Sirolli V, Di Pietro N. Vitamin E (Alpha-Tocopherol) Metabolism and Nutrition in Chronic Kidney Disease. Antioxidants (Basel). 2022 May 18;11(5):989. doi: 10.3390/antiox11050989[↩][↩]
- Azzi A. Molecular mechanism of alpha-tocopherol action. Free Radic Biol Med. 2007 Jul 1;43(1):16-21. doi: 10.1016/j.freeradbiomed.2007.03.013[↩]
- Zingg JM, Libinaki R, Lai CQ, Meydani M, Gianello R, Ogru E, Azzi A. Modulation of gene expression by α-tocopherol and α-tocopheryl phosphate in THP-1 monocytes. Free Radic Biol Med. 2010 Dec 15;49(12):1989-2000. doi: 10.1016/j.freeradbiomed.2010.09.034[↩]
- Institute of Medicine. Food and Nutrition Board. Dietary Reference Intakes: Vitamin C, Vitamin E, Selenium, and Carotenoids. Washington, DC: National Academy Press, 2000. https://www.nap.edu/catalog/9810/dietary-reference-intakes-for-vitamin-c-vitamin-e-selenium-and-carotenoids[↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Cynamon HA, Milov DE, Valenstein E, Wagner M. Effect of vitamin E deficiency on neurologic function in patients with cystic fibrosis. J Pediatr. 1988 Oct;113(4):637-40. doi: 10.1016/s0022-3476(88)80371-8[↩]
- Swann IL, Kendra JR. Anaemia, vitamin E deficiency and failure to thrive in an infant. Clin Lab Haematol. 1998 Feb;20(1):61-3. doi: 10.1046/j.1365-2257.1998.00099.x[↩]
- Bines JE, Truby HD, Armstrong DS, Carzino R, Grimwood K. Vitamin A and E deficiency and lung disease in infants with cystic fibrosis. J Paediatr Child Health. 2005 Dec;41(12):663-8. doi: 10.1111/j.1440-1754.2005.00755.x[↩]
- Brigelius-Flohé R. Vitamin E: the shrew waiting to be tamed. Free Radic Biol Med. 2009 Mar 1;46(5):543-54. doi: 10.1016/j.freeradbiomed.2008.12.007[↩]
- Jiang Q. Natural forms of vitamin E: metabolism, antioxidant, and anti-inflammatory activities and their role in disease prevention and therapy. Free Radic Biol Med. 2014 Jul;72:76-90. doi: 10.1016/j.freeradbiomed.2014.03.035[↩][↩][↩]
- Jiang Q, Christen S, Shigenaga MK, Ames BN. gamma-tocopherol, the major form of vitamin E in the US diet, deserves more attention. Am J Clin Nutr. 2001 Dec;74(6):714-22. doi: 10.1093/ajcn/74.6.714[↩]
- Mah E, Pei R, Guo Y, Ballard KD, Barker T, Rogers VE, Parker BA, Taylor AW, Traber MG, Volek JS, Bruno RS. γ-Tocopherol-rich supplementation additively improves vascular endothelial function during smoking cessation. Free Radic Biol Med. 2013 Dec;65:1291-1299. doi: 10.1016/j.freeradbiomed.2013.09.016[↩]
- Mah E, Pei R, Guo Y, Masterjohn C, Ballard KD, Taylor BA, Taylor AW, Traber MG, Volek JS, Bruno RS. Greater γ-tocopherol status during acute smoking abstinence with nicotine replacement therapy improved vascular endothelial function by decreasing 8-iso-15(S)-prostaglandin F2α. Exp Biol Med (Maywood). 2015 Apr;240(4):527-33. doi: 10.1177/1535370214556948[↩]
- Ahsan H, Ahad A, Iqbal J, Siddiqui WA. Pharmacological potential of tocotrienols: a review. Nutr Metab (Lond). 2014 Nov 12;11(1):52. doi: 10.1186/1743-7075-11-52[↩][↩]
- Constantinou C, Papas A, Constantinou AI. Vitamin E and cancer: An insight into the anticancer activities of vitamin E isomers and analogs. Int J Cancer. 2008 Aug 15;123(4):739-52. doi: 10.1002/ijc.23689[↩]
- Fu JY, Che HL, Tan DM, Teng KT. Bioavailability of tocotrienols: evidence in human studies. Nutr Metab (Lond). 2014 Jan 13;11(1):5. doi: 10.1186/1743-7075-11-5[↩]
- Davis S, Davis BM, Richens JL, Vere KA, Petrov PG, Winlove CP, O’Shea P. α-Tocopherols modify the membrane dipole potential leading to modulation of ligand binding by P-glycoprotein. J Lipid Res. 2015 Aug;56(8):1543-50. doi: 10.1194/jlr.M059519[↩]
- Marko MG, Ahmed T, Bunnell SC, Wu D, Chung H, Huber BT, Meydani SN. Age-associated decline in effective immune synapse formation of CD4(+) T cells is reversed by vitamin E supplementation. J Immunol. 2007 Feb 1;178(3):1443-9. doi: 10.4049/jimmunol.178.3.1443[↩]
- Molano A, Meydani SN. Vitamin E, signalosomes and gene expression in T cells. Mol Aspects Med. 2012 Feb;33(1):55-62. doi: 10.1016/j.mam.2011.11.002[↩][↩]
- Wu D, Meydani SN. Age-associated changes in immune function: impact of vitamin E intervention and the underlying mechanisms. Endocr Metab Immune Disord Drug Targets. 2014;14(4):283-9. doi: 10.2174/1871530314666140922143950[↩][↩]
- De la Fuente M, Hernanz A, Guayerbas N, Victor VM, Arnalich F. Vitamin E ingestion improves several immune functions in elderly men and women. Free Radic Res. 2008 Mar;42(3):272-80. doi: 10.1080/10715760801898838[↩]
- Meydani SN, Meydani M, Blumberg JB, Leka LS, Siber G, Loszewski R, Thompson C, Pedrosa MC, Diamond RD, Stollar BD. Vitamin E supplementation and in vivo immune response in healthy elderly subjects. A randomized controlled trial. JAMA. 1997 May 7;277(17):1380-6. doi: 10.1001/jama.1997.03540410058031[↩]
- Pallast EG, Schouten EG, de Waart FG, Fonk HC, Doekes G, von Blomberg BM, Kok FJ. Effect of 50- and 100-mg vitamin E supplements on cellular immune function in noninstitutionalized elderly persons. Am J Clin Nutr. 1999 Jun;69(6):1273-81. doi: 10.1093/ajcn/69.6.1273[↩]
- Meydani SN, Leka LS, Fine BC, Dallal GE, Keusch GT, Singh MF, Hamer DH. Vitamin E and respiratory tract infections in elderly nursing home residents: a randomized controlled trial. JAMA. 2004 Aug 18;292(7):828-36. doi: 10.1001/jama.292.7.828. Erratum in: JAMA. 2004 Sep 15;292(11):1305. Erratum in: JAMA. 2007 May 2;297(17):1882[↩]
- Latha MS, Martis J, Shobha V, Sham Shinde R, Bangera S, Krishnankutty B, Bellary S, Varughese S, Rao P, Naveen Kumar BR. Sunscreening agents: a review. J Clin Aesthet Dermatol. 2013 Jan;6(1):16-26. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3543289[↩]
- Gabros S, Nessel TA, Zito PM. Sunscreens and Photoprotection. [Updated 2023 Mar 7]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK537164[↩][↩]
- Lavker RM, Gerberick GF, Veres D, Irwin CJ, Kaidbey KH. Cumulative effects from repeated exposures to suberythemal doses of UVB and UVA in human skin. J Am Acad Dermatol. 1995 Jan;32(1):53-62. doi: 10.1016/0190-9622(95)90184-1[↩]
- Rhodes LE. Topical and systemic approaches for protection against solar radiation-induced skin damage. Clin Dermatol. 1998 Jan-Feb;16(1):75-82. doi: 10.1016/s0738-081x(97)00171-5[↩]
- Kagan V, Witt E, Goldman R, Scita G, Packer L. Ultraviolet light-induced generation of vitamin E radicals and their recycling. A possible photosensitizing effect of vitamin E in skin. Free Radic Res Commun. 1992;16(1):51-64. doi: 10.3109/10715769209049159[↩][↩]
- Kondo S, Mamada A, Yamaguchi J, Fukuro S. Protective effect of dl-alpha-tocopherol on the cytotoxicity of ultraviolet B against human skin fibroblasts in vitro. Photodermatol Photoimmunol Photomed. 1990 Aug;7(4):173-7.[↩]
- Jin GH, Liu Y, Jin SZ, Liu XD, Liu SZ. UVB induced oxidative stress in human keratinocytes and protective effect of antioxidant agents. Radiat Environ Biophys. 2007 Mar;46(1):61-8. doi: 10.1007/s00411-007-0096-1[↩][↩]
- Stewart MS, Cameron GS, Pence BC. Antioxidant nutrients protect against UVB-induced oxidative damage to DNA of mouse keratinocytes in culture. J Invest Dermatol. 1996 May;106(5):1086-9. doi: 10.1111/1523-1747.ep12339344[↩]
- Pauling L, Willoughby R, Reynolds R, Blaisdell BE, Lawson S. Incidence of squamous cell carcinoma in hairless mice irradiated with ultraviolet light in relation to intake of ascorbic acid (vitamin C) and of D, L-alpha-tocopheryl acetate (vitamin E). Int J Vitam Nutr Res Suppl. 1982;23:53-82.[↩][↩]
- Gerrish KE, Gensler HL. Prevention of photocarcinogenesis by dietary vitamin E. Nutr Cancer. 1993;19(2):125-33. doi: 10.1080/01635589309514243[↩][↩][↩]
- Burke KE, Clive J, Combs GF Jr, Nakamura RM. Effects of topical L-selenomethionine with topical and oral vitamin E on pigmentation and skin cancer induced by ultraviolet irradiation in Skh:2 hairless mice. J Am Acad Dermatol. 2003 Sep;49(3):458-72. doi: 10.1067/s0190-9622(03)00900-9[↩][↩][↩][↩]
- Burke KE, Clive J, Combs GF Jr, Commisso J, Keen CL, Nakamura RM. Effects of topical and oral vitamin E on pigmentation and skin cancer induced by ultraviolet irradiation in Skh:2 hairless mice. Nutr Cancer. 2000;38(1):87-97. doi: 10.1207/S15327914NC381_13[↩]
- Record IR, Dreosti IE, Konstantinopoulos M, Buckley RA. The influence of topical and systemic vitamin E on ultraviolet light-induced skin damage in hairless mice. Nutr Cancer. 1991;16(3-4):219-25. doi: 10.1080/01635589109514160[↩][↩][↩][↩]
- McArdle F, Rhodes LE, Parslew RA, Close GL, Jack CI, Friedmann PS, Jackson MJ. Effects of oral vitamin E and beta-carotene supplementation on ultraviolet radiation-induced oxidative stress in human skin. Am J Clin Nutr. 2004 Nov;80(5):1270-5. doi: 10.1093/ajcn/80.5.1270[↩][↩]
- Werninghaus K, Meydani M, Bhawan J, Margolis R, Blumberg JB, Gilchrest BA. Evaluation of the Photoprotective Effect of Oral Vitamin E Supplementation. Arch Dermatol. 1994;130(10):1257–1261. doi:10.1001/archderm.1994.01690100041005[↩]
- van der Pols JC, Heinen MM, Hughes MC, Ibiebele TI, Marks GC, Green AC. Serum antioxidants and skin cancer risk: an 8-year community-based follow-up study. Cancer Epidemiol Biomarkers Prev. 2009 Apr;18(4):1167-73. doi: 10.1158/1055-9965.EPI-08-1211[↩][↩]
- McNaughton SA, Marks GC, Green AC. Role of dietary factors in the development of basal cell cancer and squamous cell cancer of the skin. Cancer Epidemiol Biomarkers Prev. 2005 Jul;14(7):1596-607. doi: 10.1158/1055-9965.EPI-05-0026[↩]
- Eberlein-König B, Placzek M, Przybilla B. Protective effect against sunburn of combined systemic ascorbic acid (vitamin C) and d-alpha-tocopherol (vitamin E). J Am Acad Dermatol. 1998 Jan;38(1):45-8. doi: 10.1016/s0190-9622(98)70537-7[↩]
- Fuchs J, Kern H. Modulation of UV-light-induced skin inflammation by D-alpha-tocopherol and L-ascorbic acid: a clinical study using solar simulated radiation. Free Radic Biol Med. 1998 Dec;25(9):1006-12. doi: 10.1016/s0891-5849(98)00132-4[↩][↩]
- Placzek M, Gaube S, Kerkmann U, Gilbertz KP, Herzinger T, Haen E, Przybilla B. Ultraviolet B-induced DNA damage in human epidermis is modified by the antioxidants ascorbic acid and D-alpha-tocopherol. J Invest Dermatol. 2005 Feb;124(2):304-7. doi: 10.1111/j.0022-202X.2004.23560.x[↩][↩]
- Yamada Y, Obayashi M, Ishikawa T, Kiso Y, Ono Y, Yamashita K. Dietary tocotrienol reduces UVB-induced skin damage and sesamin enhances tocotrienol effects in hairless mice. J Nutr Sci Vitaminol (Tokyo). 2008 Apr;54(2):117-23. doi: 10.3177/jnsv.54.117[↩][↩]
- Saral Y, Uyar B, Ayar A, Naziroglu M. Protective effects of topical alpha-tocopherol acetate on UVB irradiation in guinea pigs: importance of free radicals. Physiol Res. 2002;51(3):285-90.[↩][↩]
- Yuen KS, Halliday GM. alpha-Tocopherol, an inhibitor of epidermal lipid peroxidation, prevents ultraviolet radiation from suppressing the skin immune system. Photochem Photobiol. 1997 Mar;65(3):587-92. doi: 10.1111/j.1751-1097.1997.tb08610.x[↩]
- Lopez-Torres M, Thiele JJ, Shindo Y, Han D, Packer L. Topical application of alpha-tocopherol modulates the antioxidant network and diminishes ultraviolet-induced oxidative damage in murine skin. Br J Dermatol. 1998 Feb;138(2):207-15. doi: 10.1046/j.1365-2133.1998.02062.x[↩]
- Chen W, Barthelman M, Martinez J, Alberts D, Gensler HL. Inhibition of cyclobutane pyrimidine dimer formation in epidermal p53 gene of UV-irradiated mice by alpha-tocopherol. Nutr Cancer. 1997;29(3):205-11. doi: 10.1080/01635589709514625[↩]
- McVean M, Liebler DC. Inhibition of UVB induced DNA photodamage in mouse epidermis by topically applied alpha-tocopherol. Carcinogenesis. 1997 Aug;18(8):1617-22. doi: 10.1093/carcin/18.8.161[↩]
- McVean M, Liebler DC. Prevention of DNA photodamage by vitamin E compounds and sunscreens: roles of ultraviolet absorbance and cellular uptake. Mol Carcinog. 1999 Mar;24(3):169-76. doi: 10.1002/(sici)1098-2744(199903)24:3<169::aid-mc3>3.0.co;2-a[↩][↩]
- Bissett DL, Chatterjee R, Hannon DP. Photoprotective effect of superoxide-scavenging antioxidants against ultraviolet radiation-induced chronic skin damage in the hairless mouse. Photodermatol Photoimmunol Photomed. 1990 Apr;7(2):56-62.[↩][↩]
- Ritter EF, Axelrod M, Minn KW, Eades E, Rudner AM, Serafin D, Klitzman B. Modulation of ultraviolet light-induced epidermal damage: beneficial effects of tocopherol. Plast Reconstr Surg. 1997 Sep;100(4):973-80. doi: 10.1097/00006534-199709001-00021[↩][↩]
- Trevithick JR, Shum DT, Redae S, Mitton KP, Norley C, Karlik SJ, Groom AC, Schmidt EE. Reduction of sunburn damage to skin by topical application of vitamin E acetate following exposure to ultraviolet B radiation: effect of delaying application or of reducing concentration of vitamin E acetate applied. Scanning Microsc. 1993 Dec;7(4):1269-81.[↩][↩][↩][↩][↩]
- Trevithick JR, Xiong H, Lee S, Shum DT, Sanford SE, Karlik SJ, Norley C, Dilworth GR. Topical tocopherol acetate reduces post-UVB, sunburn-associated erythema, edema, and skin sensitivity in hairless mice. Arch Biochem Biophys. 1992 Aug 1;296(2):575-82. doi: 10.1016/0003-9861(92)90613-2[↩][↩]
- Gensler HL, Aickin M, Peng YM, Xu M. Importance of the form of topical vitamin E for prevention of photocarcinogenesis. Nutr Cancer. 1996;26(2):183-91. doi: 10.1080/01635589609514474[↩]
- Beijersbergen van Henegouwen GM, Junginger HE, de Vries H. Hydrolysis of RRR-alpha-tocopheryl acetate (vitamin E acetate) in the skin and its UV protecting activity (an in vivo study with the rat). J Photochem Photobiol B. 1995 Jul;29(1):45-51. doi: 10.1016/1011-1344(95)90251-1[↩][↩]
- Schoonderwoerd SA, Beijersbergen van Henegouwen GM, Persons KC. Effect of alpha-tocopherol and di-butyl-hydroxytoluene (BHT) on UV-A-induced photobinding of 8-methoxypsoralen to Wistar rat epidermal biomacromolecules in vivo. Arch Toxicol. 1991;65(6):490-4. doi: 10.1007/BF01977362[↩]
- Slaga TJ, Bracken WM. The effects of antioxidants on skin tumor initiation and aryl hydrocarbon hydroxylase. Cancer Res. 1977 Jun;37(6):1631-5.[↩]
- Potapenko AYa, Abijev GA, Pistsov MYu, Roshchupkin DI, Vladimirov YuA, Pliquett F, Ermolayev AV, Sarycheva IK, Evstigneeva RP. PUVA-induced erythema and changes in mechanoelectrical properties of skin. Inhibition by tocopherols. Arch Dermatol Res. 1984;276(1):12-6. doi: 10.1007/BF00412556[↩]
- Roshchupkin DI, Pistsov MY, Potapenko AY. Inhibition of ultraviolet light-induced erythema by antioxidants. Arch Dermatol Res. 1979 Aug;266(1):91-4. doi: 10.1007/BF00412867[↩][↩]
- Thiele JJ, Hsieh SN, Ekanayake-Mudiyanselage S. Vitamin E: critical review of its current use in cosmetic and clinical dermatology. Dermatol Surg. 2005 Jul;31(7 Pt 2):805-13; discussion 813. doi: 10.1111/j.1524-4725.2005.31724[↩][↩][↩][↩]
- Zhai H, Behnam S, Villarama CD, Arens-Corell M, Choi MJ, Maibach HI. Evaluation of the antioxidant capacity and preventive effects of a topical emulsion and its vehicle control on the skin response to UV exposure. Skin Pharmacol Physiol. 2005 Nov-Dec;18(6):288-93. doi: 10.1159/000088014[↩][↩]
- Montenegro L, Bonina F, Rigano L, Giogilli S, Sirigu S. Protective effect evaluation of free radical scavengers on UVB induced human cutaneous erythema by skin reflectance spectrophotometry. Int J Cosmet Sci. 1995 Jun;17(3):91-103. doi: 10.1111/j.1467-2494.1995.tb00113.x[↩]
- Chung JH, Seo JY, Lee MK, Eun HC, Lee JH, Kang S, Fisher GJ, Voorhees JJ. Ultraviolet modulation of human macrophage metalloelastase in human skin in vivo. J Invest Dermatol. 2002 Aug;119(2):507-12. doi: 10.1046/j.1523-1747.2002.01844.x[↩][↩]
- Lin JY, Selim MA, Shea CR, Grichnik JM, Omar MM, Monteiro-Riviere NA, Pinnell SR. UV photoprotection by combination topical antioxidants vitamin C and vitamin E. J Am Acad Dermatol. 2003 Jun;48(6):866-74. doi: 10.1067/mjd.2003.425[↩][↩][↩][↩][↩]
- Darr D, Dunston S, Faust H, Pinnell S. Effectiveness of antioxidants (vitamin C and E) with and without sunscreens as topical photoprotectants. Acta Derm Venereol. 1996 Jul;76(4):264-8. doi: 10.2340/0001555576264268[↩]
- Lin FH, Lin JY, Gupta RD, Tournas JA, Burch JA, Selim MA, Monteiro-Riviere NA, Grichnik JM, Zielinski J, Pinnell SR. Ferulic acid stabilizes a solution of vitamins C and E and doubles its photoprotection of skin. J Invest Dermatol. 2005 Oct;125(4):826-32. doi: 10.1111/j.0022-202X.2005.23768.x[↩][↩]
- Quevedo WC Jr, Holstein TJ, Dyckman J, McDonald CJ, Isaacson EL. Inhibition of UVR-induced tanning and immunosuppression by topical applications of vitamins C and E to the skin of hairless (hr/hr) mice. Pigment Cell Res. 2000 Apr;13(2):89-98. doi: 10.1034/j.1600-0749.2000.130207.x[↩][↩][↩]
- Dreher F, Gabard B, Schwindt DA, Maibach HI. Topical melatonin in combination with vitamins E and C protects skin from ultraviolet-induced erythema: a human study in vivo. Br J Dermatol. 1998 Aug;139(2):332-9. doi: 10.1046/j.1365-2133.1998.02447.x[↩]
- Quevedo WC Jr, Holstein TJ, Dyckman J, McDonald CJ. The responses of the human epidermal melanocyte system to chronic erythemal doses of UVR in skin protected by topical applications of a combination of vitamins C and E. Pigment Cell Res. 2000 Jun;13(3):190-2. doi: 10.1034/j.1600-0749.2000.130312.x[↩]
- Murray JC, Burch JA, Streilein RD, Iannacchione MA, Hall RP, Pinnell SR. A topical antioxidant solution containing vitamins C and E stabilized by ferulic acid provides protection for human skin against damage caused by ultraviolet irradiation. J Am Acad Dermatol. 2008 Sep;59(3):418-25. doi: 10.1016/j.jaad.2008.05.004[↩][↩]
- Passi S, De Pità O, Grandinetti M, Simotti C, Littarru GP. The combined use of oral and topical lipophilic antioxidants increases their levels both in sebum and stratum corneum. Biofactors. 2003;18(1-4):289-97. doi: 10.1002/biof.5520180233[↩]
- Burke KE. Photodamage of the skin: protection and reversal with topical antioxidants. J Cosmet Dermatol. 2004 Jul;3(3):149-55. doi: 10.1111/j.1473-2130.2004.00067.x[↩][↩]
- Nagata C, Nakamura K, Wada K, Oba S, Hayashi M, Takeda N, Yasuda K. Association of dietary fat, vegetables and antioxidant micronutrients with skin ageing in Japanese women. Br J Nutr. 2010 May;103(10):1493-8. doi: 10.1017/S0007114509993461[↩][↩][↩][↩]
- Boelsma E, van de Vijver LP, Goldbohm RA, Klöpping-Ketelaars IA, Hendriks HF, Roza L. Human skin condition and its associations with nutrient concentrations in serum and diet. Am J Clin Nutr. 2003 Feb;77(2):348-55. doi: 10.1093/ajcn/77.2.348[↩][↩]
- Gehring W, Fluhr J, Gloor M. Influence of vitamin E acetate on stratum corneum hydration. Arzneimittelforschung. 1998 Jul;48(7):772-5.[↩][↩]
- Gönüllü U, Sensoy D, Uner M, Yener G, Altinkurt T. Comparing the moisturizing effects of ascorbic acid and calcium ascorbate against that of tocopherol in emulsions. J Cosmet Sci. 2006 Nov-Dec;57(6):465-73.[↩][↩]
- Weber SU, Thiele JJ, Cross CE, Packer L. Vitamin C, uric acid, and glutathione gradients in murine stratum corneum and their susceptibility to ozone exposure. J Invest Dermatol. 1999 Dec;113(6):1128-32. doi: 10.1046/j.1523-1747.1999.00789.x[↩][↩]
- Thiele JJ, Traber MG, Podda M, Tsang K, Cross CE, Packer L. Ozone depletes tocopherols and tocotrienols topically applied to murine skin. FEBS Lett. 1997 Jan 20;401(2-3):167-70. doi: 10.1016/s0014-5793(96)01463-9[↩][↩][↩][↩]
- Valacchi G, Weber SU, Luu C, Cross CE, Packer L. Ozone potentiates vitamin E depletion by ultraviolet radiation in the murine stratum corneum. FEBS Lett. 2000 Jan 21;466(1):165-8. doi: 10.1016/s0014-5793(99)01787-1[↩][↩][↩][↩]
- Wu S, Gao J, Dinh QT, Chen C, Fimmel S. IL-8 production and AP-1 transactivation induced by UVA in human keratinocytes: roles of D-alpha-tocopherol. Mol Immunol. 2008 Apr;45(8):2288-96. doi: 10.1016/j.molimm.2007.11.019[↩]
- Kato E, Sasaki Y, Takahashi N. Sodium dl-α-tocopheryl-6-O-phosphate inhibits PGE₂ production in keratinocytes induced by UVB, IL-1β and peroxidants. Bioorg Med Chem. 2011 Nov 1;19(21):6348-55. doi: 10.1016/j.bmc.2011.08.067[↩][↩]
- Shibata A, Nakagawa K, Kawakami Y, Tsuzuki T, Miyazawa T. Suppression of gamma-tocotrienol on UVB induced inflammation in HaCaT keratinocytes and HR-1 hairless mice via inflammatory mediators multiple signaling. J Agric Food Chem. 2010 Jun 9;58(11):7013-20. doi: 10.1021/jf100691g[↩][↩][↩]
- Nakagawa K, Shibata A, Maruko T, Sookwong P, Tsuduki T, Kawakami K, Nishida H, Miyazawa T. gamma-Tocotrienol reduces squalene hydroperoxide-induced inflammatory responses in HaCaT keratinocytes. Lipids. 2010 Sep;45(9):833-41. doi: 10.1007/s11745-010-3458-4[↩][↩]
- Yoshida E, Watanabe T, Takata J, Yamazaki A, Karube Y, Kobayashi S. Topical application of a novel, hydrophilic gamma-tocopherol derivative reduces photo-inflammation in mice skin. J Invest Dermatol. 2006 Jul;126(7):1633-40. doi: 10.1038/sj.jid.5700236[↩][↩]
- Meydani SN, Barklund MP, Liu S, Meydani M, Miller RA, Cannon JG, Morrow FD, Rocklin R, Blumberg JB. Vitamin E supplementation enhances cell-mediated immunity in healthy elderly subjects. Am J Clin Nutr. 1990 Sep;52(3):557-63. doi: 10.1093/ajcn/52.3.557[↩]
- Han SN, Meydani SN. Impact of vitamin E on immune function and its clinical implications. Expert Rev Clin Immunol. 2006 Jul;2(4):561-7. doi: 10.1586/1744666X.2.4.561[↩][↩]
- Jiang Q, Ames BN. Gamma-tocopherol, but not alpha-tocopherol, decreases proinflammatory eicosanoids and inflammation damage in rats. FASEB J. 2003 May;17(8):816-22. doi: 10.1096/fj.02-0877com[↩]
- Tsoureli-Nikita E, Hercogova J, Lotti T, Menchini G. Evaluation of dietary intake of vitamin E in the treatment of atopic dermatitis: a study of the clinical course and evaluation of the immunoglobulin E serum levels. Int J Dermatol. 2002 Mar;41(3):146-50. doi: 10.1046/j.1365-4362.2002.01423.x[↩][↩]
- Keller KL, Fenske NA. Uses of vitamins A, C, and E and related compounds in dermatology: a review. J Am Acad Dermatol. 1998 Oct;39(4 Pt 1):611-25. doi: 10.1016/s0190-9622(98)70011-8[↩]
- Hayakawa R, Ueda H, Nozaki T, Izawa Y, Yokotake J, Yazaki K, Azumi T, Okada Y, Kobayashi M, Usuda T, Ishida J, Kondo T, Adachi A, Kawase A, Matsunaga K. Effects of combination treatment with vitamins E and C on chloasma and pigmented contact dermatitis. A double blind controlled clinical trial. Acta Vitaminol Enzymol. 1981;3(1):31-8.[↩][↩]
- Javanbakht MH, Keshavarz SA, Djalali M, Siassi F, Eshraghian MR, Firooz A, Seirafi H, Ehsani AH, Chamari M, Mirshafiey A. Randomized controlled trial using vitamins E and D supplementation in atopic dermatitis. J Dermatolog Treat. 2011 Jun;22(3):144-50. doi: 10.3109/09546630903578566[↩][↩]
- Shukla A, Rasik AM, Patnaik GK. Depletion of reduced glutathione, ascorbic acid, vitamin E and antioxidant defence enzymes in a healing cutaneous wound. Free Radic Res. 1997 Feb;26(2):93-101. doi: 10.3109/10715769709097788[↩][↩]
- Musalmah M, Nizrana MY, Fairuz AH, NoorAini AH, Azian AL, Gapor MT, Wan Ngah WZ. Comparative effects of palm vitamin E and alpha-tocopherol on healing and wound tissue antioxidant enzyme levels in diabetic rats. Lipids. 2005 Jun;40(6):575-80. doi: 10.1007/s11745-005-1418-9[↩][↩]
- Musalmah M, Fairuz AH, Gapor MT, Ngah WZ. Effect of vitamin E on plasma malondialdehyde, antioxidant enzyme levels and the rates of wound closures during wound healing in normal and diabetic rats. Asia Pac J Clin Nutr. 2002;11 Suppl 7:S448-51. doi: 10.1046/j.1440-6047.11.s.7.6.x[↩]
- Taren DL, Chvapil M, Weber CW. Increasing the breaking strength of wounds exposed to preoperative irradiation using vitamin E supplementation. Int J Vitam Nutr Res. 1987;57(2):133-7.[↩][↩]
- Ehrlich HP, Tarver H, Hunt TK. Inhibitory effects of vitamin E on collagen synthesis and wound repair. Ann Surg. 1972 Feb;175(2):235-40. doi: 10.1097/00000658-197202000-00013[↩][↩]
- Baumann LS, Spencer J. The effects of topical vitamin E on the cosmetic appearance of scars. Dermatol Surg. 1999 Apr;25(4):311-5. doi: 10.1046/j.1524-4725.1999.08223.x[↩][↩]
- Jenkins M, Alexander JW, MacMillan BG, Waymack JP, Kopcha R. Failure of topical steroids and vitamin E to reduce postoperative scar formation following reconstructive surgery. J Burn Care Rehabil. 1986 Jul-Aug;7(4):309-12. doi: 10.1097/00004630-198607000-00002[↩][↩]
- Barbosa E, Faintuch J, Machado Moreira EA, Gonçalves da Silva VR, Lopes Pereima MJ, Martins Fagundes RL, Filho DW. Supplementation of vitamin E, vitamin C, and zinc attenuates oxidative stress in burned children: a randomized, double-blind, placebo-controlled pilot study. J Burn Care Res. 2009 Sep-Oct;30(5):859-66. doi: 10.1097/BCR.0b013e3181b487a8[↩][↩]
- Ellinger S, Stehle P. Efficacy of vitamin supplementation in situations with wound healing disorders: results from clinical intervention studies. Curr Opin Clin Nutr Metab Care. 2009 Nov;12(6):588-95. doi: 10.1097/MCO.0b013e328331a5b5[↩][↩]
- Leonard SW, Bruno RS, Ramakrishnan R, Bray T, Traber MG. Cigarette smoking increases human vitamin E requirements as estimated by plasma deuterium-labeled CEHC. Ann N Y Acad Sci. 2004 Dec;1031:357-60. doi: 10.1196/annals.1331.044[↩][↩]
- Bruno RS, Leonard SW, Atkinson J, Montine TJ, Ramakrishnan R, Bray TM, Traber MG. Faster plasma vitamin E disappearance in smokers is normalized by vitamin C supplementation. Free Radic Biol Med. 2006 Feb 15;40(4):689-97. doi: 10.1016/j.freeradbiomed.2005.10.051[↩]
- Booth SL, Golly I, Sacheck JM, Roubenoff R, Dallal GE, Hamada K, Blumberg JB. Effect of vitamin E supplementation on vitamin K status in adults with normal coagulation status. Am J Clin Nutr. 2004 Jul;80(1):143-8. doi: 10.1093/ajcn/80.1.143[↩]
- Pastori D, Carnevale R, Cangemi R, Saliola M, Nocella C, Bartimoccia S, Vicario T, Farcomeni A, Violi F, Pignatelli P. Vitamin E serum levels and bleeding risk in patients receiving oral anticoagulant therapy: a retrospective cohort study. J Am Heart Assoc. 2013 Oct 28;2(6):e000364. doi: 10.1161/JAHA.113.000364[↩][↩][↩]
- Ford ES, Ajani UA, Mokdad AH. Brief communication: the prevalence of high intake of vitamin E from the use of supplements among U.S. adults. Ann Intern Med 2005;143:116-20. https://www.ncbi.nlm.nih.gov/pubmed/16027453?dopt=Abstract[↩]
- Brown BG, Zhao XQ, Chait A, Fisher LD, Cheung MC, Morse JS, Dowdy AA, Marino EK, Bolson EL, Alaupovic P, Frohlich J, Albers JJ. Simvastatin and niacin, antioxidant vitamins, or the combination for the prevention of coronary disease. N Engl J Med. 2001 Nov 29;345(22):1583-92. doi: 10.1056/NEJMoa011090[↩]
- Lawenda BD, Kelly KM, Ladas EJ, Sagar SM, Vickers A, Blumberg JB. Should supplemental antioxidant administration be avoided during chemotherapy and radiation therapy? J Natl Cancer Inst. 2008 Jun 4;100(11):773-83. doi: 10.1093/jnci/djn148[↩]
- Block KI, Koch AC, Mead MN, Tothy PK, Newman RA, Gyllenhaal C. Impact of antioxidant supplementation on chemotherapeutic efficacy: a systematic review of the evidence from randomized controlled trials. Cancer Treat Rev. 2007 Aug;33(5):407-18. doi: 10.1016/j.ctrv.2007.01.005[↩]
- Stampfer MJ, Hennekens CH, Manson JE, Colditz GA, Rosner B, Willett WC. Vitamin E consumption and the risk of coronary disease in women. N Engl J Med. 1993;328:1444-9. https://www.ncbi.nlm.nih.gov/pubmed/8479463[↩]
- Rimm EB, Stampfer MJ. Antioxidants for vascular disease. Med Clin North Am. 2000;84:239-49. https://www.ncbi.nlm.nih.gov/pubmed/10685137[↩][↩]
- Dietary supplementation with n-3 polyunsaturated fatty acids and vitamin E after myocardial infarction: results of the GISSI-Prevenzione trial. Gruppo Italiano per lo Studio della Sopravvivenza nell’Infarto miocardico. Lancet. 1999;354:447-55. https://www.ncbi.nlm.nih.gov/pubmed/10465168[↩]
- Yusuf S, Dagenais G, Pogue J, Bosch J, Sleight P. Vitamin E supplementation and cardiovascular events in high-risk patients. The Heart Outcomes Prevention Evaluation Study Investigators. N Engl J Med. 2000;342:154-60. https://www.ncbi.nlm.nih.gov/pubmed/10639540[↩]
- Lonn E, Bosch J, Yusuf S, et al. Effects of long-term vitamin E supplementation on cardiovascular events and cancer: a randomized controlled trial. JAMA. 2005;293:1338-47. https://www.ncbi.nlm.nih.gov/pubmed/15769967[↩][↩]
- Lonn E, Bosch J, Yusuf S, Sheridan P, Pogue J, Arnold JM, Ross C, Arnold A, Sleight P, Probstfield J, Dagenais GR; HOPE and HOPE-TOO Trial Investigators. Effects of long-term vitamin E supplementation on cardiovascular events and cancer: a randomized controlled trial. JAMA. 2005 Mar 16;293(11):1338-47. doi: 10.1001/jama.293.11.1338[↩][↩]
- Brown BG, Crowley J. Is there any hope for vitamin E? JAMA. 2005 Mar 16;293(11):1387-90. doi: 10.1001/jama.293.11.1387[↩]
- Waters DD, Alderman EL, Hsia J, Howard BV, Cobb FR, Rogers WJ, Ouyang P, Thompson P, Tardif JC, Higginson L, Bittner V, Steffes M, Gordon DJ, Proschan M, Younes N, Verter JI. Effects of hormone replacement therapy and antioxidant vitamin supplements on coronary atherosclerosis in postmenopausal women: a randomized controlled trial. JAMA. 2002 Nov 20;288(19):2432-40. doi: 10.1001/jama.288.19.2432[↩]
- Kris-Etherton PM, Lichtenstein AH, Howard BV, Steinberg D, Witztum JL. Antioxidant vitamin supplements and cardiovascular disease. Circulation. 2004;110:637-41. https://www.ncbi.nlm.nih.gov/pubmed/15289389[↩]
- Lee IM, Cook NR, Gaziano JM, Gordon D, Ridker PM, Manson JE, Hennekens CH, Buring JE. Vitamin E in the primary prevention of cardiovascular disease and cancer: the Women’s Health Study: a randomized controlled trial. JAMA. 2005 Jul 6;294(1):56-65. doi: 10.1001/jama.294.1.56[↩][↩]
- Glynn RJ, Ridker PM, Goldhaber SZ, Zee RY, Buring JE. Effects of random allocation to vitamin E supplementation on the occurrence of venous thromboembolism: report from the Women’s Health Study. Circulation. 2007;116:1497-503. https://www.ncbi.nlm.nih.gov/pubmed/17846285[↩]
- Sesso HD, Buring JE, Christen WG, Kurth T, Belanger C, MacFadyen J, Bubes V, Manson JE, Glynn RJ, Gaziano JM. Vitamins E and C in the prevention of cardiovascular disease in men: the Physicians’ Health Study II randomized controlled trial. JAMA. 2008 Nov 12;300(18):2123-33. doi: 10.1001/jama.2008.600[↩][↩][↩]
- Hercberg S, Galan P, Preziosi P, et al. The SU.VI.MAX Study: a randomized, placebo-controlled trial of the health effects of antioxidant vitamins and minerals. Arch Intern Med. 2004;164:2335-42. https://www.ncbi.nlm.nih.gov/pubmed/15557412[↩]
- Sesso HD, Buring JE, Christen WG, et al. Vitamins E and C in the prevention of cardiovascular disease in men: the Physicians’ Health Study II randomized controlled trial. JAMA. 2008;300:2123-33. https://www.ncbi.nlm.nih.gov/pubmed/18997197[↩]
- Milman U, Blum S, Shapira C, et al. Vitamin E supplementation reduces cardiovascular events in a subgroup of middle-aged individuals with both type 2 diabetes mellitus and the haptoglobin 2-2 genotype: a prospective double-blinded clinical trial. Arterioscler Thromb Vasc Biol. 2008;28:341-7. https://www.ncbi.nlm.nih.gov/pubmed/18032779[↩]
- Blumberg JB, Frei B. Why clinical trials of vitamin E and cardiovascular diseases may be fatally flawed. Commentary on “The relationship between dose of vitamin E and suppression of oxidative stress in humans.” Free Radic Biol Med 2007;43:1374-6. https://www.ncbi.nlm.nih.gov/pubmed/17936183[↩]
- Weitberg AB, Corvese D. Effect of vitamin E and beta-carotene on DNA strand breakage induced by tobacco-specific nitrosamines and stimulated human phagocytes. J Exp Clin Cancer Res. 1997 Mar;16(1):11-4.[↩]
- Hunter DJ, Manson JE, Colditz GA, et al. A prospective study of the intake of vitamins C, E, and A and the risk of breast cancer. N Engl J Med. 1993;329:234-40. https://www.ncbi.nlm.nih.gov/pubmed/8292129[↩]
- Willett WC, Polk BF, Underwood BA, et al. Relation of serum vitamins A and E and carotenoids to the risk of cancer. N Engl J Med. 1984;310:430-4. https://www.ncbi.nlm.nih.gov/pubmed/6537988[↩]
- Chan JM, Stampfer MJ, Ma J, Rimm EB, Willett WC, Giovannucci EL. Supplemental vitamin E intake and prostate cancer risk in a large cohort of men in the United States. Cancer Epidemiol Biomarkers Prev. 1999;8:893-9. https://www.ncbi.nlm.nih.gov/pubmed/10548318[↩][↩]
- van Dam RM, Huang Z, Giovannucci E, et al. Diet and basal cell carcinoma of the skin in a prospective cohort of men. Am J Clin Nutr. 2000;71:135-41. https://www.ncbi.nlm.nih.gov/pubmed/10617958[↩]
- Wu K, Willett WC, Chan JM, et al. A prospective study on supplemental vitamin e intake and risk of colon cancer in women and men. Cancer Epidemiol Biomarkers Prev. 2002;11:1298-304. https://www.ncbi.nlm.nih.gov/pubmed/12433706[↩]
- Fung TT, Spiegelman D, Egan KM, Giovannucci E, Hunter DJ, Willett WC. Vitamin and carotenoid intake and risk of squamous cell carcinoma of the skin. Int J Cancer. 2003;103:110-5. https://www.ncbi.nlm.nih.gov/pubmed/12455062[↩]
- Feskanich D, Willett WC, Hunter DJ, Colditz GA. Dietary intakes of vitamins A, C, and E and risk of melanoma in two cohorts of women. Br J Cancer. 2003;88:1381-7. https://www.ncbi.nlm.nih.gov/pubmed/12778065[↩]
- Cho E, Spiegelman D, Hunter DJ, et al. Premenopausal intakes of vitamins A, C, and E, folate, and carotenoids, and risk of breast cancer. Cancer Epidemiol Biomarkers Prev. 2003;12:713-20. https://www.ncbi.nlm.nih.gov/pubmed/12917201[↩]
- Cho E, Hunter DJ, Spiegelman D, et al. Intakes of vitamins A, C and E and folate and multivitamins and lung cancer: a pooled analysis of 8 prospective studies. Int J Cancer. 2006;118:970-8. https://www.ncbi.nlm.nih.gov/pubmed/16152626[↩]
- Lee JE, Giovannucci E, Smith-Warner SA, Spiegelman D, Willett WC, Curhan GC. Intakes of fruits, vegetables, vitamins A, C, and E, and carotenoids and risk of renal cell cancer. Cancer Epidemiol Biomarkers Prev. 2006;15:2445-52. https://www.ncbi.nlm.nih.gov/pubmed/17164369[↩]
- Heinonen OP, Albanes D, Virtamo J, et al. Prostate cancer and supplementation with alpha-tocopherol and beta-carotene: incidence and mortality in a controlled trial. J Natl Cancer Inst. 1998;90:440-6. https://www.ncbi.nlm.nih.gov/pubmed/9521168[↩][↩]
- Kirsh VA, Hayes RB, Mayne ST, et al. Supplemental and dietary vitamin E, beta-carotene, and vitamin C intakes and prostate cancer risk. J Natl Cancer Inst. 2006;98:245-54. https://www.ncbi.nlm.nih.gov/pubmed/16478743[↩]
- Peters U, Littman AJ, Kristal AR, Patterson RE, Potter JD, White E. Vitamin E and selenium supplementation and risk of prostate cancer in the Vitamins and Lifestyle (VITAL) study cohort. Cancer Causes Control. 2008;19:75-87. https://www.ncbi.nlm.nih.gov/pubmed/17943452[↩]
- Lippman SM, Klein EA, Goodman PJ, et al. Effect of selenium and vitamin E on risk of prostate cancer and other cancers: the Selenium and Vitamin E Cancer Prevention Trial (SELECT). JAMA. 2009;301:39-51. https://www.ncbi.nlm.nih.gov/pubmed/19066370[↩]
- Klein EA, Thompson IM, Jr., Tangen CM, et al. Vitamin E and the risk of prostate cancer: the Selenium and Vitamin E Cancer Prevention Trial (SELECT). JAMA. 2011;306:1549-56. www.ncbi.nlm.nih.gov/pubmed/21990298[↩]
- National Cancer Institute. Selenium and Vitamin E Cancer Prevention Trial (SELECT): Questions and Answers. https://www.cancer.gov/types/prostate/research/select-trial-results-qa[↩][↩][↩][↩][↩]
- Sesso HD, Buring JE, Christen WG, et al. Vitamins E and C in the prevention of cardiovascular disease in men: the Physicians’ Health Study II randomized controlled trial. JAMA. 2008; 300(18):2123-33.[↩]
- Gaziano JM, Glynn RJ, Christen WG, et al. Vitamins E and C in the prevention of prostate and total cancer in men: the Physicians’ Health Study II randomized controlled trial. JAMA. 2009;301:52-62. https://www.ncbi.nlm.nih.gov/pubmed/19066368[↩]
- Bostick RM, Potter JD, McKenzie DR, Sellers TA, Kushi LH, Steinmetz KA, Folsom AR. Reduced risk of colon cancer with high intake of vitamin E: the Iowa Women’s Health Study. Cancer Res. 1993 Sep 15;53(18):4230-7[↩]
- Wu K, Willett WC, Chan JM, Fuchs CS, Colditz GA, Rimm EB, Giovannucci EL. A prospective study on supplemental vitamin e intake and risk of colon cancer in women and men. Cancer Epidemiol Biomarkers Prev. 2002 Nov;11(11):1298-304.[↩]
- Graham S, Zielezny M, Marshall J, Priore R, Freudenheim J, Brasure J, Haughey B, Nasca P, Zdeb M. Diet in the epidemiology of postmenopausal breast cancer in the New York State Cohort. Am J Epidemiol. 1992 Dec 1;136(11):1327-37. doi: 10.1093/oxfordjournals.aje.a116445[↩]
- Jacobs EJ, Henion AK, Briggs PJ, Connell CJ, McCullough ML, Jonas CR, Rodriguez C, Calle EE, Thun MJ. Vitamin C and vitamin E supplement use and bladder cancer mortality in a large cohort of US men and women. Am J Epidemiol. 2002 Dec 1;156(11):1002-10. doi: 10.1093/aje/kwf147[↩]
- Lippman SM, Klein EA, Goodman PJ, et al. Effect of selenium and vitamin E on risk of prostate cancer and other cancers. JAMA 2009; 301(1). Published online December 9, 2008. Print edition January 2009.[↩]
- EA Klein, IM Thompson, CM Tangen, et al. Vitamin E and the Risk of Prostate Cancer: Results of The Selenium and Vitamin E Cancer Prevention Trial (SELECT). JAMA 2011; 306(14) 1549-1556.[↩]
- Heinonen OP, Albanes D, Virtamo J, et al. Prostate cancer and supplementation with alpha-tocopherol and beta-carotene: Incidence and mortality in a controlled trial. JNCI 1998; 90(6):440-446.[↩]
- Kristal AR, Darke AK, Morris S, et al. Baseline selenium status and effects of selenium and vitamin E supplementation on prostate cancer risk. J Natl Cancer Inst 2014.[↩]
- Clark LC, Combs GF Jr., Turnbull BW, et al. Effects of selenium supplementation for cancer prevention in patients with carcinoma of the skin. A randomized controlled trial: Nutritional Prevention of Cancer Study Group. JAMA 1996; 276(24):1957-1963.[↩]
- Duffield-Lillico AJ, Reid ME, Turnbull BW, et al. Baseline characteristics and the effect of selenium supplementation on cancer incidence in a randomized clinical trial: A summary report of the Nutritional Prevention of Cancer Trial. Cancer Epidemiology, Biomarkers & Prevention 2002; 11(7):630-639.[↩]
- National Institute of Health, Office of Dietary Supplements. Vitamin E. https://ods.od.nih.gov/factsheets/VitaminE-HealthProfessional/[↩]
- Evans J. Primary prevention of age related macular degeneration. BMJ. 2007 Oct 13;335(7623):729. doi: 10.1136/bmj.39351.478924.BE[↩]
- Taylor HR, Tikellis G, Robman LD, McCarty CA, McNeil JJ. Vitamin E supplementation and macular degeneration: randomised controlled trial. BMJ. 2002 Jul 6;325(7354):11. doi: 10.1136/bmj.325.7354.11[↩]
- Teikari JM, Virtamo J, Rautalahti M, Palmgren J, Liesto K, Heinonen OP. Long-term supplementation with alpha-tocopherol and beta-carotene and age-related cataract. Acta Ophthalmol Scand. 1997 Dec;75(6):634-40. doi: 10.1111/j.1600-0420.1997.tb00620.x[↩]
- Age-Related Eye Disease Study Research Group (2001). A randomized, placebo-controlled, clinical trial of high-dose supplementation with vitamins C and E, beta carotene, and zinc for age-related macular degeneration and vision loss: AREDS report no. 8. Archives of ophthalmology (Chicago, Ill. : 1960), 119(10), 1417–1436. https://doi.org/10.1001/archopht.119.10.1417[↩]
- The Age-Related Eye Disease Study 2 (AREDS2) Research Group*. Lutein + Zeaxanthin and Omega-3 Fatty Acids for Age-Related Macular Degeneration: The Age-Related Eye Disease Study 2 (AREDS2) Randomized Clinical Trial. JAMA. 2013;309(19):2005–2015. doi:10.1001/jama.2013.4997 https://jamanetwork.com/journals/jama/fullarticle/1684847[↩]
- Leske MC, Chylack LT Jr, He Q, Wu SY, Schoenfeld E, Friend J, Wolfe J. Antioxidant vitamins and nuclear opacities: the longitudinal study of cataract. Ophthalmology. 1998 May;105(5):831-6. doi: 10.1016/s0161-6420(98)95021-7[↩]
- Jacques PF, Taylor A, Moeller S, Hankinson SE, Rogers G, Tung W, Ludovico J, Willett WC, Chylack LT Jr. Long-term nutrient intake and 5-year change in nuclear lens opacities. Arch Ophthalmol. 2005 Apr;123(4):517-26. doi: 10.1001/archopht.123.4.517[↩]
- Age-Related Eye Disease Study Research Group (2001). A randomized, placebo-controlled, clinical trial of high-dose supplementation with vitamins C and E and beta carotene for age-related cataract and vision loss: AREDS report no. 9. Archives of ophthalmology (Chicago, Ill. : 1960), 119(10), 1439–1452. https://doi.org/10.1001/archopht.119.10.1439[↩]
- Age-Related Eye Disease Study 2 (AREDS2) Research Group, Chew, E. Y., SanGiovanni, J. P., Ferris, F. L., Wong, W. T., Agron, E., Clemons, T. E., Sperduto, R., Danis, R., Chandra, S. R., Blodi, B. A., Domalpally, A., Elman, M. J., Antoszyk, A. N., Ruby, A. J., Orth, D., Bressler, S. B., Fish, G. E., Hubbard, G. B., Klein, M. L., … Bernstein, P. (2013). Lutein/zeaxanthin for the treatment of age-related cataract: AREDS2 randomized trial report no. 4. JAMA ophthalmology, 131(7), 843–850. https://doi.org/10.1001/jamaophthalmol.2013.4412[↩]
- Sano M, Ernesto C, Thomas RG, Klauber MR, Schafer K, Grundman M, Woodbury P, Growdon J, Cotman CW, Pfeiffer E, Schneider LS, Thal LJ. A controlled trial of selegiline, alpha-tocopherol, or both as treatment for Alzheimer’s disease. The Alzheimer’s Disease Cooperative Study. N Engl J Med. 1997 Apr 24;336(17):1216-22. doi: 10.1056/NEJM199704243361704[↩][↩]
- Morris MC, Evans DA, Bienias JL, Tangney CC, Wilson RS. Vitamin E and cognitive decline in older persons. Arch Neurol. 2002 Jul;59(7):1125-32. doi: 10.1001/archneur.59.7.1125[↩]
- Kang JH, Cook N, Manson J, Buring JE, Grodstein F. A randomized trial of vitamin E supplementation and cognitive function in women. Arch Intern Med. 2006 Dec 11-25;166(22):2462-8. doi: 10.1001/archinte.166.22.2462[↩]
- Petersen RC, Thomas RG, Grundman M, Bennett D, Doody R, Ferris S, Galasko D, Jin S, Kaye J, Levey A, Pfeiffer E, Sano M, van Dyck CH, Thal LJ; Alzheimer’s Disease Cooperative Study Group. Vitamin E and donepezil for the treatment of mild cognitive impairment. N Engl J Med. 2005 Jun 9;352(23):2379-88. doi: 10.1056/NEJMoa050151[↩]
- Espeland MA, Henderson VW. Preventing cognitive decline in usual aging. Arch Intern Med. 2006 Dec 11-25;166(22):2433-4. doi: 10.1001/archinte.166.22.2433[↩]
- Isaac MG, Quinn R, Tabet N. Vitamin E for Alzheimer’s disease and mild cognitive impairment. Cochrane Database Syst Rev. 2008 Jul 16;(3):CD002854. doi: 10.1002/14651858.CD002854.pub2. Update in: Cochrane Database Syst Rev. 2012;11:CD002854[↩]
- Sano M, Ernesto C, Thomas RG, Klauber MR, Schafer K, Grundman M, et al. A controlled trial of selegiline, alpha-tocopherol, or both as treatment for Alzehimer’s disease. N Engl J Med 1997;336:1216-22. https://www.ncbi.nlm.nih.gov/pubmed/9110909[↩]
- Sano M, Ernesto C, Thomas RG, Klauber MR, Schafer K, Grundman M, et al. A controlled trial of selegiline, alpha-tocopherol, or both as treatment for Alzehimer’s disease. N Engl J Med 1997;336:1216-22. https://www.ncbi.nlm.nih.gov/pubmed/9110909?dopt=Abstract[↩]
- Kamat CD, Gadal S, Mhatre M, Williamson KS, Pye QN, Hensley K. Antioxidants in central nervous system diseases: preclinical promise and translational challenges. J Alzheimers Dis. 2008;15:473-93. https://www.ncbi.nlm.nih.gov/pubmed/18997301[↩]
- Grodstein F, Chen J, Willett WC. High-dose antioxidant supplements and cognitive function in community-dwelling elderly women. Am J Clin Nutr. 2003;77:975-84. https://www.ncbi.nlm.nih.gov/pubmed/12663300[↩]
- Zandi PP, Anthony JC, Khachaturian AS, et al. Reduced risk of Alzheimer disease in users of antioxidant vitamin supplements: the Cache County Study. Arch Neurol. 2004;61:82-8. https://www.ncbi.nlm.nih.gov/pubmed/14732624[↩]
- Laurin D, Masaki KH, Foley DJ, White LR, Launer LJ. Midlife dietary intake of antioxidants and risk of late-life incident dementia: the Honolulu-Asia Aging Study. Am J Epidemiol. 2004;159:959-67. https://www.ncbi.nlm.nih.gov/pubmed/15128608[↩]
- Gray SL, Anderson ML, Crane PK, et al. Antioxidant vitamin supplement use and risk of dementia or Alzheimer’s disease in older adults. J Am Geriatr Soc. 2008;56:291-5. https://www.ncbi.nlm.nih.gov/pubmed/18047492[↩]
- Petersen RC, Thomas RG, Grundman M, et al. Vitamin E and donepezil for the treatment of mild cognitive impairment. N Engl J Med. 2005;352:2379-88. https://www.ncbi.nlm.nih.gov/pubmed/15829527[↩]
- Zhang SM, Hernan MA, Chen H, Spiegelman D, Willett WC, Ascherio A. Intakes of vitamins E and C, carotenoids, vitamin supplements, and PD risk. Neurology. 2002;59:1161-9. https://www.ncbi.nlm.nih.gov/pubmed/12391343?dopt=Citation[↩]
- Etminan M, Gill SS, Samii A. Intake of vitamin E, vitamin C, and carotenoids and the risk of Parkinson’s disease: a meta-analysis. Lancet Neurol. 2005;4:362-5. https://www.ncbi.nlm.nih.gov/pubmed/15907740[↩]
- Morens DM, Grandinetti A, Waslien CI, Park CB, Ross GW, White LR. Case-control study of idiopathic Parkinson’s disease and dietary vitamin E intake. Neurology. 1996;46:1270-4. https://www.ncbi.nlm.nih.gov/pubmed/8628465[↩]
- Effects of tocopherol and deprenyl on the progression of disability in early Parkinson’s disease. The Parkinson Study Group. N Engl J Med. 1993;328:176-83. https://www.ncbi.nlm.nih.gov/pubmed/8417384[↩]
- Ascherio A, Weisskopf MG, O’Reilly E J, et al. Vitamin E intake and risk of amyotrophic lateral sclerosis. Ann Neurol. 2005;57:104-10. https://www.ncbi.nlm.nih.gov/pubmed/15529299[↩]
- Wang H, O’Reilly EJ, Weisskopf MG, et al. Vitamin E intake and risk of amyotrophic lateral sclerosis: a pooled analysis of data from 5 prospective cohort studies. Am J Epidemiol. 2011;173:595-602. https://www.ncbi.nlm.nih.gov/pubmed/21335424[↩]
- Orrell RW, Lane RJ, Ross M. Antioxidant treatment for amyotrophic lateral sclerosis / motor neuron disease. Cochrane Database Syst Rev. 2007:CD002829. https://www.ncbi.nlm.nih.gov/pubmed/17253482[↩]
- Francque SM, Marchesini G, Kautz A, Walmsley M, Dorner R, Lazarus JV, Zelber-Sagi S, Hallsworth K, Busetto L, Frühbeck G, Dicker D, Woodward E, Korenjak M, Willemse J, Koek GH, Vinker S, Ungan M, Mendive JM, Lionis C. Non-alcoholic fatty liver disease: A patient guideline. JHEP Rep. 2021 Sep 17;3(5):100322. doi: 10.1016/j.jhepr.2021.100322[↩][↩][↩][↩]
- Kudaravalli P, John S. Nonalcoholic Fatty Liver. [Updated 2023 Apr 7]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK541033[↩][↩][↩][↩][↩]
- Dyson JK, Anstee QM, McPherson S. Non-alcoholic fatty liver disease: a practical approach to diagnosis and staging. Frontline Gastroenterol. 2014 Jul;5(3):211-218. doi: 10.1136/flgastro-2013-100403[↩]
- Basaranoglu M, Neuschwander-Tetri BA. Nonalcoholic Fatty Liver Disease: Clinical Features and Pathogenesis. Gastroenterol Hepatol (N Y). 2006 Apr;2(4):282-291. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5335683[↩]
- Musso G, Cassader M, Rosina F, Gambino R. Impact of current treatments on liver disease, glucose metabolism and cardiovascular risk in non-alcoholic fatty liver disease (NAFLD): a systematic review and meta-analysis of randomised trials. Diabetologia. 2012 Apr;55(4):885-904. doi: 10.1007/s00125-011-2446-4[↩]
- Chalasani NP, Sanyal AJ, Kowdley KV, Robuck PR, Hoofnagle J, Kleiner DE, Unalp A, Tonascia J; NASH CRN Research Group. Pioglitazone versus vitamin E versus placebo for the treatment of non-diabetic patients with non-alcoholic steatohepatitis: PIVENS trial design. Contemp Clin Trials. 2009 Jan;30(1):88-96. doi: 10.1016/j.cct.2008.09.003[↩]
- Sanyal AJ, Chalasani N, Kowdley KV, McCullough A, Diehl AM, Bass NM, Neuschwander-Tetri BA, Lavine JE, Tonascia J, Unalp A, Van Natta M, Clark J, Brunt EM, Kleiner DE, Hoofnagle JH, Robuck PR; NASH CRN. Pioglitazone, vitamin E, or placebo for nonalcoholic steatohepatitis. N Engl J Med. 2010 May 6;362(18):1675-85. doi: 10.1056/NEJMoa0907929[↩][↩]
- Lavine JE, Schwimmer JB, Van Natta ML, et al; Nonalcoholic Steatohepatitis Clinical Research Network. Effect of vitamin E or metformin for treatment of nonalcoholic fatty liver disease in children and adolescents: the TONIC randomized controlled trial. JAMA. 2011 Apr 27;305(16):1659-68. doi: 10.1001/jama.2011.520[↩]
- Ji HF. Vitamin E therapy on aminotransferase levels in NAFLD/NASH patients. Nutrition. 2015 Jun;31(6):899. doi: 10.1016/j.nut.2015.02.010[↩]
- D’Adamo E, Marcovecchio ML, Giannini C, de Giorgis T, Chiavaroli V, Chiarelli F, Mohn A. Improved oxidative stress and cardio-metabolic status in obese prepubertal children with liver steatosis treated with lifestyle combined with Vitamin E. Free Radic Res. 2013 Mar;47(3):146-53. doi: 10.3109/10715762.2012.755262[↩][↩]
- Rhie G, Shin MH, Seo JY, Choi WW, Cho KH, Kim KH, Park KC, Eun HC, Chung JH. Aging- and photoaging-dependent changes of enzymic and nonenzymic antioxidants in the epidermis and dermis of human skin in vivo. J Invest Dermatol. 2001 Nov;117(5):1212-7. doi: 10.1046/j.0022-202x.2001.01469.x. Erratum in: J Invest Dermatol 2002 Apr;118(4):741.[↩][↩][↩]
- Thiele JJ, Traber MG, Packer L. Depletion of human stratum corneum vitamin E: an early and sensitive in vivo marker of UV induced photo-oxidation. J Invest Dermatol. 1998 May;110(5):756-61. doi: 10.1046/j.1523-1747.1998.00169.x[↩][↩]
- Ikeda S, Toyoshima K, Yamashita K. Dietary sesame seeds elevate alpha- and gamma-tocotrienol concentrations in skin and adipose tissue of rats fed the tocotrienol-rich fraction extracted from palm oil. J Nutr. 2001 Nov;131(11):2892-7. doi: 10.1093/jn/131.11.2892[↩]
- Ekanayake-Mudiyanselage S, Kraemer K, Thiele JJ. Oral supplementation with all-Rac- and RRR-alpha-tocopherol increases vitamin E levels in human sebum after a latency period of 14-21 days. Ann N Y Acad Sci. 2004 Dec;1031:184-94. doi: 10.1196/annals.1331.017[↩]
- Vaule H, Leonard SW, Traber MG. Vitamin E delivery to human skin: studies using deuterated alpha-tocopherol measured by APCI LC-MS. Free Radic Biol Med. 2004 Feb 15;36(4):456-63. doi: 10.1016/j.freeradbiomed.2003.11.020[↩][↩]
- Thiele JJ, Weber SU, Packer L. Sebaceous gland secretion is a major physiologic route of vitamin E delivery to skin. J Invest Dermatol. 1999 Dec;113(6):1006-10. doi: 10.1046/j.1523-1747.1999.00794.x[↩][↩]
- Weber C, Podda M, Rallis M, Thiele JJ, Traber MG, Packer L. Efficacy of topically applied tocopherols and tocotrienols in protection of murine skin from oxidative damage induced by UV-irradiation. Free Radic Biol Med. 1997;22(5):761-9. doi: 10.1016/s0891-5849(96)00346-2[↩][↩]
- Baumann L. Skin ageing and its treatment. J Pathol. 2007 Jan;211(2):241-51. doi: 10.1002/path.2098[↩]
- Machlin LJ, Filipski R, Nelson J, Horn LR, Brin M. Effects of a prolonged vitamin E deficiency in the rat. J Nutr. 1977 Jul;107(7):1200-8. doi: 10.1093/jn/107.7.1200[↩]
- Igarashi A, Uzuka M, Nakajima K. The effects of vitamin E deficiency on rat skin. Br J Dermatol. 1989 Jul;121(1):43-9. doi: 10.1111/j.1365-2133.1989.tb01398.x[↩]
- Dermatol Surg. 1999 Apr;25(4):311-5.10.1046/j.1524-4725.1999.08223. The effects of topical vitamin E on the cosmetic appearance of scars. https://www.ncbi.nlm.nih.gov/pubmed/10417589/[↩][↩]
- Traber MG, Rallis M, Podda M, Weber C, Maibach HI, Packer L. Penetration and distribution of alpha-tocopherol, alpha- or gamma-tocotrienols applied individually onto murine skin. Lipids. 1998 Jan;33(1):87-91. doi: 10.1007/s11745-998-0183-0[↩][↩][↩]
- Thiele JJ, Ekanayake-Mudiyanselage S. Vitamin E in human skin: organ-specific physiology and considerations for its use in dermatology. Mol Aspects Med. 2007 Oct-Dec;28(5-6):646-67. doi: 10.1016/j.mam.2007.06.001[↩][↩]
- Nada A, Krishnaiah YS, Zaghloul AA, Khattab I. In vitro and in vivo permeation of vitamin E and vitamin E acetate from cosmetic formulations. Med Princ Pract. 2011;20(6):509-13. doi: 10.1159/000329883[↩]
- Kosari P, Alikhan A, Sockolov M, Feldman SR. Vitamin E and allergic contact dermatitis. Dermatitis. 2010 May-Jun;21(3):148-53.[↩]
- Werninghaus K, Meydani M, Bhawan J, Margolis R, Blumberg JB, Gilchrest BA. Evaluation of the photoprotective effect of oral vitamin E supplementation. Arch Dermatol. 1994 Oct;130(10):1257-61.[↩]
- U.S. Department of Agriculture, Agricultural Research Service. 2011. USDA National Nutrient Database for Standard Reference, Release 24. Nutrient Data Laboratory Home Page. https://www.ars.usda.gov/northeast-area/beltsville-md/beltsville-human-nutrition-research-center/nutrient-data-laboratory/[↩]
- U.S. Department of Agriculture, Agricultural Research Service. USDA National Nutrient Database for Standard Reference, Release 27. Nutrient Data Laboratory home page, 2014. https://ndb.nal.usda.gov/ndb/[↩]
- Ford ES, Sowell A. Serum alpha-tocopherol status in the United States population: findings from the Third National Health and Nutrition Examination Survey. Am J Epidemiol. 1999 Aug 1;150(3):290-300. doi: 10.1093/oxfordjournals.aje.a010001[↩]
- Fulgoni VL 3rd, Keast DR, Bailey RL, Dwyer J. Foods, fortificants, and supplements: Where do Americans get their nutrients? J Nutr. 2011 Oct;141(10):1847-54. doi: 10.3945/jn.111.142257[↩][↩]
- Martinello F., Fardin P., Ottina M., Ricchieri G.L., Koenig M., Cavalier L., Trevisan C.P. Supplemental Therapy in Isolated Vitamin E Deficiency Improves the Peripheral Neuropathy and Prevents the Progression of Ataxia. J. Neurol. Sci. 1998;156:177–179. doi: 10.1016/S0022-510X(98)00038-0[↩][↩][↩]
- Kohlschutter A., Finckh B., Nickel M., Bley A., Hübner C. First Recognized Patient with Genetic Vitamin e Deficiency Stable after 36 Years of Controlled Supplement Therapy. Neurodegener. Dis. 2020;20:583–587. doi: 10.1159/000508080[↩][↩][↩]
- Ulatowski L.M., Manor D. Vitamin E and neurodegeneration. Neurobiol. Dis. 2015;84:78–83. doi: 10.1016/j.nbd.2015.04.002[↩][↩]
- Traber MG. Vitamin E. In: Ross AC, Caballero B, Cousins RJ, Tucker KL, Ziegler TR, eds. Modern Nutrition in Health and Disease. 11th ed. Philadelphia: Lippincott Williams & Wilkins; 2014:293-304.[↩][↩][↩][↩][↩][↩]
- Brion LP, Bell EF, Raghuveer TS. Vitamin E supplementation for prevention of morbidity and mortality in preterm infants. Cochrane Database Syst Rev. 2003;(4):CD003665. doi: 10.1002/14651858.CD003665[↩]
- Shamim AA, Schulze K, Merrill RD, Kabir A, Christian P, Shaikh S, Wu L, Ali H, Labrique AB, Mehra S, Klemm RD, Rashid M, Sungpuag P, Udomkesmalee E, West KP Jr. First-trimester plasma tocopherols are associated with risk of miscarriage in rural Bangladesh. Am J Clin Nutr. 2015 Feb;101(2):294-301. doi: 10.3945/ajcn.114.094920[↩][↩]
- Kowdley KV, Mason JB, Meydani SN, Cornwall S, Grand RJ. Vitamin E deficiency and impaired cellular immunity related to intestinal fat malabsorption. Gastroenterology. 1992 Jun;102(6):2139-42. doi: 10.1016/0016-5085(92)90344-x[↩]
- Traber MG. Vitamin E. In: Shils ME, Shike M, Ross AC, Caballero B, Cousins R, eds. Modern Nutrition in Health and Disease. 10th ed. Baltimore, MD: Lippincott Williams & Wilkins, 2006;396-411[↩][↩]
- Tanyel MC, Mancano LD. Neurologic findings in vitamin E deficiency. Am Fam Physician. 1997 Jan;55(1):197-201.[↩][↩][↩]
- Kemnic TR, Coleman M. Vitamin E Deficiency. [Updated 2022 Jul 4]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK519051[↩][↩][↩]
- Kono N, Ohto U, Hiramatsu T, Urabe M, Uchida Y, Satow Y, Arai H. Impaired α-TTP-PIPs interaction underlies familial vitamin E deficiency. Science. 2013 May 31;340(6136):1106-10. doi: 10.1126/science.1233508[↩]
- Sokol RJ, Guggenheim MA, Heubi JE, Iannaccone ST, Butler-Simon N, Jackson V, Miller C, Cheney M, Balistreri WF, Silverman A. Frequency and clinical progression of the vitamin E deficiency neurologic disorder in children with prolonged neonatal cholestasis. Am J Dis Child. 1985 Dec;139(12):1211-5. doi: 10.1001/archpedi.1985.02140140045024[↩]
- Di Donato I, Bianchi S, Federico A. Ataxia with vitamin E deficiency: update of molecular diagnosis. Neurol Sci. 2010 Aug;31(4):511-5. doi: 10.1007/s10072-010-0261-1[↩]
- Wright ME, Lawson KA, Weinstein SJ, Pietinen P, Taylor PR, Virtamo J, Albanes D. Higher baseline serum concentrations of vitamin E are associated with lower total and cause-specific mortality in the Alpha-Tocopherol, Beta-Carotene Cancer Prevention Study. Am J Clin Nutr. 2006 Nov;84(5):1200-7. doi: 10.1093/ajcn/84.5.1200[↩]
- Becker AE, Vargas W, Pearson TS. Ataxia with Vitamin E Deficiency May Present with Cervical Dystonia. Tremor Other Hyperkinet Mov (N Y). 2016 May 17;6:374. doi: 10.7916/D8B85820[↩]
- Elkamil A, Johansen KK, Aasly J. Ataxia with vitamin e deficiency in norway. J Mov Disord. 2015 Jan;8(1):33-6. doi: 10.14802/jmd.14030[↩]
- Schuelke M. Ataxia with Vitamin E Deficiency. 2005 May 20 [Updated 2023 Mar 16]. In: Adam MP, Mirzaa GM, Pagon RA, et al., editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993-2023. Available from: https://www.ncbi.nlm.nih.gov/books/NBK1241[↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Zhang LW, Liu B, Peng DT. Clinical and genetic study of ataxia with vitamin E deficiency: A case report. World J Clin Cases. 2022 Aug 16;10(23):8271-8276. doi: 10.12998/wjcc.v10.i23.8271[↩][↩]
- El Euch-Fayache G, Bouhlal Y, Amouri R, Feki M, Hentati F. Molecular, clinical and peripheral neuropathy study of Tunisian patients with ataxia with vitamin E deficiency. Brain. 2014 Feb;137(Pt 2):402-10. doi: 10.1093/brain/awt339[↩][↩][↩][↩][↩]
- Hamza W, Ali Pacha L, Hamadouche T, Muller J, Drouot N, Ferrat F, Makri S, Chaouch M, Tazir M, Koenig M, Benhassine T. Molecular and clinical study of a cohort of 110 Algerian patients with autosomal recessive ataxia. BMC Med Genet. 2015 Jun 12;16:36. doi: 10.1186/s12881-015-0180-3[↩]
- Esmer C, Salazar MA, Rentería PE, Bravo OA. Clinical and molecular findings in a patient with ataxia with vitamin E deficiency, homozygous for the c.205-1G>C mutation in the TTPA gene. Bol Med Hos Infant Mex. 2013;70:313–317.[↩]
- Benomar A, Yahyaoui M, Meggouh F, Bouhouche A, Boutchich M, Bouslam N, Zaim A, Schmitt M, Belaidi H, Ouazzani R, Chkili T, Koenig M. Clinical comparison between AVED patients with 744 del A mutation and Friedreich ataxia with GAA expansion in 15 Moroccan families. J Neurol Sci. 2002 Jun 15;198(1-2):25-9. doi: 10.1016/s0022-510x(02)00057-6[↩][↩]
- Chung S, Ghelfi M, Atkinson J, Parker R, Qian J, Carlin C, Manor D. Vitamin E and Phosphoinositides Regulate the Intracellular Localization of the Hepatic α-Tocopherol Transfer Protein. J Biol Chem. 2016 Aug 12;291(33):17028-39. doi: 10.1074/jbc.M116.734210[↩][↩]
- Ataxia with Vitamin E Deficiency. https://rarediseases.org/rare-diseases/ataxia-with-vitamin-e-deficiency[↩][↩][↩][↩][↩][↩][↩]
- Ouahchi K, Arita M, Kayden H, Hentati F, Ben Hamida M, Sokol R, Arai H, Inoue K, Mandel JL, Koenig M. Ataxia with isolated vitamin E deficiency is caused by mutations in the alpha-tocopherol transfer protein. Nat Genet. 1995 Feb;9(2):141-5. doi: 10.1038/ng0295-141[↩]
- Mariotti C, Gellera C, Rimoldi M, Mineri R, Uziel G, Zorzi G, Pareyson D, Piccolo G, Gambi D, Piacentini S, Squitieri F, Capra R, Castellotti B, Di Donato S. Ataxia with isolated vitamin E deficiency: neurological phenotype, clinical follow-up and novel mutations in TTPA gene in Italian families. Neurol Sci. 2004 Jul;25(3):130-7. doi: 10.1007/s10072-004-0246-z[↩][↩][↩][↩][↩]
- Gotoda T, Arita M, Arai H, Inoue K, Yokota T, Fukuo Y, Yazaki Y, Yamada N. Adult-onset spinocerebellar dysfunction caused by a mutation in the gene for the alpha-tocopherol-transfer protein. N Engl J Med. 1995 Nov 16;333(20):1313-8. doi: 10.1056/NEJM199511163332003[↩]
- Cavalier L, Ouahchi K, Kayden HJ, Di Donato S, Reutenauer L, Mandel JL, Koenig M. Ataxia with isolated vitamin E deficiency: heterogeneity of mutations and phenotypic variability in a large number of families. Am J Hum Genet. 1998 Feb;62(2):301-10. doi: 10.1086/301699[↩][↩][↩][↩]
- Sokol RJ. Vitamin E deficiency and neurologic disease. Annu Rev Nutr. 1988;8:351-73. doi: 10.1146/annurev.nu.08.070188.002031[↩]
- Traber MG, Sokol RJ, Kohlschütter A, Yokota T, Muller DP, Dufour R, Kayden HJ. Impaired discrimination between stereoisomers of alpha-tocopherol in patients with familial isolated vitamin E deficiency. J Lipid Res. 1993 Feb;34(2):201-10.[↩][↩]
- Yokota T, Shiojiri T, Gotoda T, Arai H. Retinitis pigmentosa and ataxia caused by a mutation in the gene for the alpha-tocopherol-transfer protein. N Engl J Med. 1996 Dec 5;335(23):1770-1. doi: 10.1056/NEJM199612053352315[↩]
- Yokota T, Shiojiri T, Gotoda T, Arita M, Arai H, Ohga T, Kanda T, Suzuki J, Imai T, Matsumoto H, Harino S, Kiyosawa M, Mizusawa H, Inoue K. Friedreich-like ataxia with retinitis pigmentosa caused by the His101Gln mutation of the alpha-tocopherol transfer protein gene. Ann Neurol. 1997 Jun;41(6):826-32. doi: 10.1002/ana.410410621[↩]
- Yokota T, Uchihara T, Kumagai J, Shiojiri T, Pang JJ, Arita M, Arai H, Hayashi M, Kiyosawa M, Okeda R, Mizusawa H. Postmortem study of ataxia with retinitis pigmentosa by mutation of the alpha-tocopherol transfer protein gene. J Neurol Neurosurg Psychiatry. 2000 Apr;68(4):521-5. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1736898/pdf/v068p00521.pdf[↩]
- Mariotti C, Gellera C, Rimoldi M, et al. Ataxia with isolated vitamin E deficiency: neurological phenotype, clinical follow‐up and novel mutations in TTPA gene in Italian families. Neurol Sci. 2004;25(3):130‐137. doi: 10.1007/s10072-004-0246-z[↩][↩]
- Ulatowski L, Parker R, Warrier G, Sultana R, Butterfield DA, Manor D. Vitamin E is essential for Purkinje neuron integrity. Neuroscience. 2014;260:120‐129. doi: 10.1016/j.neuroscience.2013.12.001[↩]
- Thapa S, Shah S, Chand S, Sah SK, Gyawali P, Paudel S, Khanal P. Ataxia due to vitamin E deficiency: A case report and updated review. Clin Case Rep. 2022 Sep 6;10(9):e6303. doi: 10.1002/ccr3.6303[↩][↩][↩][↩][↩]
- Gabsi S, Gouider‐Khouja N, Belal S, et al. Effect of vitamin E supplementation in patients with ataxia with vitamin E deficiency. Eur J Neurol. 2001;8(5):477‐481. doi: 10.1046/j.1468-1331.2001.00273.x[↩][↩]
- Schuelke M, Mayatepek E, Inter M, Becker M, Pfeiffer E, Speer A, Hübner C, Finckh B. Treatment of ataxia in isolated vitamin E deficiency caused by alpha-tocopherol transfer protein deficiency. J Pediatr. 1999 Feb;134(2):240-4. doi: 10.1016/s0022-3476(99)70424-5[↩][↩][↩][↩]
- El Euch‐Fayache G, Bouhlal Y, Amouri R, Feki M, Hentati F. Molecular, clinical and peripheral neuropathy study of Tunisian patients with ataxia with vitamin E deficiency. Brain. 2014;137(Pt 2):402‐410. doi: 10.1093/brain/awt339[↩][↩]
- Finckh B, Kontush A, Commentz J, Hübner C, Burdelski M, Kohlschütter A. Monitoring of ubiquinol-10, ubiquinone-10, carotenoids, and tocopherols in neonatal plasma microsamples using high-performance liquid chromatography with coulometric electrochemical detection. Anal Biochem. 1995 Dec 10;232(2):210-6. doi: 10.1006/abio.1995.0009[↩]
- Usuki F, Maruyama K. Ataxia caused by mutations in the alpha-tocopherol transfer protein gene. J Neurol Neurosurg Psychiatry. 2000 Aug;69(2):254-6. doi: 10.1136/jnnp.69.2.254[↩]
- Weiser H, Riss G, Kormann AW. Biodiscrimination of the eight alpha-tocopherol stereoisomers results in preferential accumulation of the four 2R forms in tissues and plasma of rats. J Nutr. 1996 Oct;126(10):2539-49. doi: 10.1093/jn/126.10.2539[↩]
- Hosomi A, Arita M, Sato Y, Kiyose C, Ueda T, Igarashi O, Arai H, Inoue K. Affinity for alpha-tocopherol transfer protein as a determinant of the biological activities of vitamin E analogs. FEBS Lett. 1997 Jun 2;409(1):105-8. doi: 10.1016/s0014-5793(97)00499-7[↩]
- Leonard SW, Terasawa Y, Farese RV Jr, Traber MG. Incorporation of deuterated RRR- or all-rac-alpha-tocopherol in plasma and tissues of alpha-tocopherol transfer protein–null mice. Am J Clin Nutr. 2002 Mar;75(3):555-60. doi: 10.1093/ajcn/75.3.555[↩]
- Amiel J, Maziere JC, Beucler I, Koenig M, Reutenauer L, Loux N, Bonnefont D, Féo C, Landrieu P. Familial isolated vitamin E deficiency. Extensive study of a large family with a 5-year therapeutic follow-up. J Inherit Metab Dis. 1995;18(3):333-40. doi: 10.1007/BF00710425[↩][↩]
- Burck U, Goebel HH, Kuhlendahl HD, Meier C, Goebel KM. Neuromyopathy and vitamin E deficiency in man. Neuropediatrics. 1981 Aug;12(3):267-78. doi: 10.1055/s-2008-1059657[↩]
- Harding AE, Matthews S, Jones S, Ellis CJ, Booth IW, Muller DP. Spinocerebellar degeneration associated with a selective defect of vitamin E absorption. N Engl J Med. 1985 Jul 4;313(1):32-5. doi: 10.1056/NEJM198507043130107[↩]
- Schuelke M, Finckh B, Sistermans EA, Ausems MG, Hübner C, von Moers A. Ataxia with vitamin E deficiency: biochemical effects of malcompliance with vitamin E therapy. Neurology. 2000 Nov 28;55(10):1584-6. doi: 10.1212/wnl.55.10.1584[↩]
- Gabsi S, Gouider-Khouja N, Belal S, Fki M, Kefi M, Turki I, Ben Hamida M, Kayden H, Mebazaa R, Hentati F. Effect of vitamin E supplementation in patients with ataxia with vitamin E deficiency. Eur J Neurol. 2001 Sep;8(5):477-81. doi: 10.1046/j.1468-1331.2001.00273.x[↩][↩]
- Bruno RS, Traber MG. Vitamin E biokinetics, oxidative stress and cigarette smoking. Pathophysiology. 2006;13(3):143‐149. doi: 10.1016/j.pathophys.2006.05.003[↩]
- Traber MG, Manor D. Vitamin E. Adv Nutr. 2012 May 1;3(3):330-1. doi: 10.3945/an.112.002139[↩]
- Müller-Schmehl K, Beninde J, Finckh B, Florian S, Dudenhausen JW, Brigelius-Flohé R, Schuelke M. Localization of alpha-tocopherol transfer protein in trophoblast, fetal capillaries’ endothelium and amnion epithelium of human term placenta. Free Radic Res. 2004 Apr;38(4):413-20. doi: 10.1080/10715760310001659611[↩]
- Amiel J, Maziere JC, Beucler I, et al. Familial isolated vitamin E deficiency. Extensive study of a large family with a 5‐year therapeutic follow‐up. J Inherit Metab Dis. 1995;18(3):333‐340. doi: 10.1007/BF00710425[↩]
- Vitamin E Deficiency Clinical Presentation. https://emedicine.medscape.com/article/126187-clinical[↩][↩]
- Berson EL, Rosner B, Sandberg MA, Hayes KC, Nicholson BW, Weigel-DiFranco C, Willett W. A randomized trial of vitamin A and vitamin E supplementation for retinitis pigmentosa. Arch Ophthalmol. 1993 Jun;111(6):761-72. doi: 10.1001/archopht.1993.01090060049022[↩][↩]
- Iwasa K, Shima K, Komai K, Nishida Y, Yokota T, Yamada M. Retinitis pigmentosa and macular degeneration in a patient with ataxia with isolated vitamin E deficiency with a novel c.717 del C mutation in the TTPA gene. J Neurol Sci. 2014 Oct 15;345(1-2):228-30. doi: 10.1016/j.jns.2014.07.001[↩]
- Boltshauser E, Weber KP. Laboratory investigations. Handb Clin Neurol. 2018;154:287-298. doi: 10.1016/B978-0-444-63956-1.00017-5[↩]
- Tokgöz Y, Terlemez S, Karul A. Fat soluble vitamin levels in children with newly diagnosed celiac disease, a case control study. BMC Pediatr. 2018 Apr 9;18(1):130. doi: 10.1186/s12887-018-1107-x[↩]
- Teriaky A, Mosli M, Chandok N, Al-Judaibi B, Marotta P, Qumosani K. Prevalence of fat-soluble vitamin (A, D, and E) and zinc deficiency in patients with cirrhosis being assessed for liver transplantation. Acta Gastroenterol Belg. 2017 Apr-Jun;80(2):237-241.[↩]
- Gomez-Pomar E, Hatfield E, Garlitz K, Westgate PM, Bada HS. Vitamin E in the Preterm Infant: A Forgotten Cause of Hemolytic Anemia. Am J Perinatol. 2018 Feb;35(3):305-310. doi: 10.1055/s-0037-1607283[↩]
- Vitamin E Deficiency Medication. https://emedicine.medscape.com/article/126187-medication[↩][↩]
- Kemnic TR, Coleman M. Vitamin E Deficiency. [Updated 2020 Jul 10]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2020 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK519051[↩]
- Thébaut A, Nemeth A, Le Mouhaër J, et al. Oral Tocofersolan corrects or prevents vitamin E deficiency in children with chronic cholestasis. J Pediatr Gastroenterol Nutr. 2016;63(6):610‐615. doi: 10.1097/MPG.0000000000001331[↩]
- Perlmutter DH, Gross P, Jones HR, Fulton A, Grand RJ. Intramuscular vitamin E repletion in children with chronic cholestasis. Am J Dis Child. 1987;141(2):170‐174. doi: 10.1001/archpedi.1987.04460020060027[↩]
- Sherf-Dagan S, Buch A, Ben-Porat T, Sakran N, Sinai T. Vitamin E status among bariatric surgery patients: a systematic review. Surg Obes Relat Dis. 2021 Apr;17(4):816-830. doi: 10.1016/j.soard.2020.10.029[↩]
- Alpha-Tocopherol, Beta Carotene Cancer Prevention Study Group. The effect of vitamin E and beta carotene on the incidence of lung cancer and other cancers in male smokers. N Engl J Med 1994;330:1029-35. https://www.ncbi.nlm.nih.gov/pubmed/8127329?dopt=Abstract[↩]
- Sesso HD, Buring JE, Christen WG, Kurth T, Belanger C, MacFadyen J, et al. Vitamins E and C in the prevention of cardiovascular disease in men: the Physicians’ Health Study II randomized controlled trial. JAMA 2008;300:2123-33. https://www.ncbi.nlm.nih.gov/pubmed/18997197?dopt=Abstract[↩]
- Diab L, Krebs NF. Vitamin Excess and Deficiency. Pediatr Rev. 2018 Apr;39(4):161-179. doi: 10.1542/pir.2016-0068[↩][↩]
- Owen KN, Dewald O. Vitamin E Toxicity. [Updated 2020 Nov 20]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2020 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK564373[↩][↩]
- Alpha-Tocopherol, Beta Carotene Cancer Prevention Study Group. The effect of vitamin E and beta carotene on the incidence of lung cancer and other cancers in male smokers. N Engl J Med. 1994 Apr 14;330(15):1029-35. doi: 10.1056/NEJM199404143301501[↩]
- Miller ER 3rd, Pastor-Barriuso R, Dalal D, Riemersma RA, Appel LJ, Guallar E. Meta-analysis: high-dosage vitamin E supplementation may increase all-cause mortality. Ann Intern Med. 2005 Jan 4;142(1):37-46. doi: 10.7326/0003-4819-142-1-200501040-00110[↩]
- Bjelakovic G, Nikolova D, Gluud LL, Simonetti RG, Gluud C. Mortality in randomized trials of antioxidant supplements for primary and secondary prevention: systematic review and meta-analysis. JAMA. 2007 Feb 28;297(8):842-57. doi: 10.1001/jama.297.8.842. Erratum in: JAMA. 2008 Feb 20;299(7):765-6.[↩]
- Huang HY, Caballero B, Chang S, Alberg A, Semba R, Schneyer C, et al. Multivitamin/Mineral Supplements and Prevention of Chronic Disease. Evidence Report/Technology Assessment No. 139. (Prepared by The Johns Hopkins University Evidence-based Practice Center under Contract No. 290-02-0018). AHRQ Publication No. 06-E012. Rockville, MD: Agency for Healthcare Research and Quality. May 2008.[↩]
- Klein EA, Thompson IM Jr, Tangen CM, Crowley JJ, Lucia MS, Goodman PJ, Minasian LM, Ford LG, Parnes HL, Gaziano JM, Karp DD, Lieber MM, Walther PJ, Klotz L, Parsons JK, Chin JL, Darke AK, Lippman SM, Goodman GE, Meyskens FL Jr, Baker LH. Vitamin E and the risk of prostate cancer: the Selenium and Vitamin E Cancer Prevention Trial (SELECT). JAMA. 2011 Oct 12;306(14):1549-56. doi: 10.1001/jama.2011.1437[↩]
- Traber MG. Vitamin E inadequacy in humans: causes and consequences. Adv Nutr. 2014 Sep;5(5):503-14. doi: 10.3945/an.114.006254[↩][↩]
- Le NK, Kesayan T, Chang JY, Rose DZ. Cryptogenic Intracranial Hemorrhagic Strokes Associated with Hypervitaminosis E and Acutely Elevated α-Tocopherol Levels. J Stroke Cerebrovasc Dis. 2020 May;29(5):104747. doi: 10.1016/j.jstrokecerebrovasdis.2020.104747[↩][↩]
- Traber MG. Mechanisms for the prevention of vitamin E excess. J Lipid Res. 2013 Sep;54(9):2295-306. doi: 10.1194/jlr.R032946[↩]