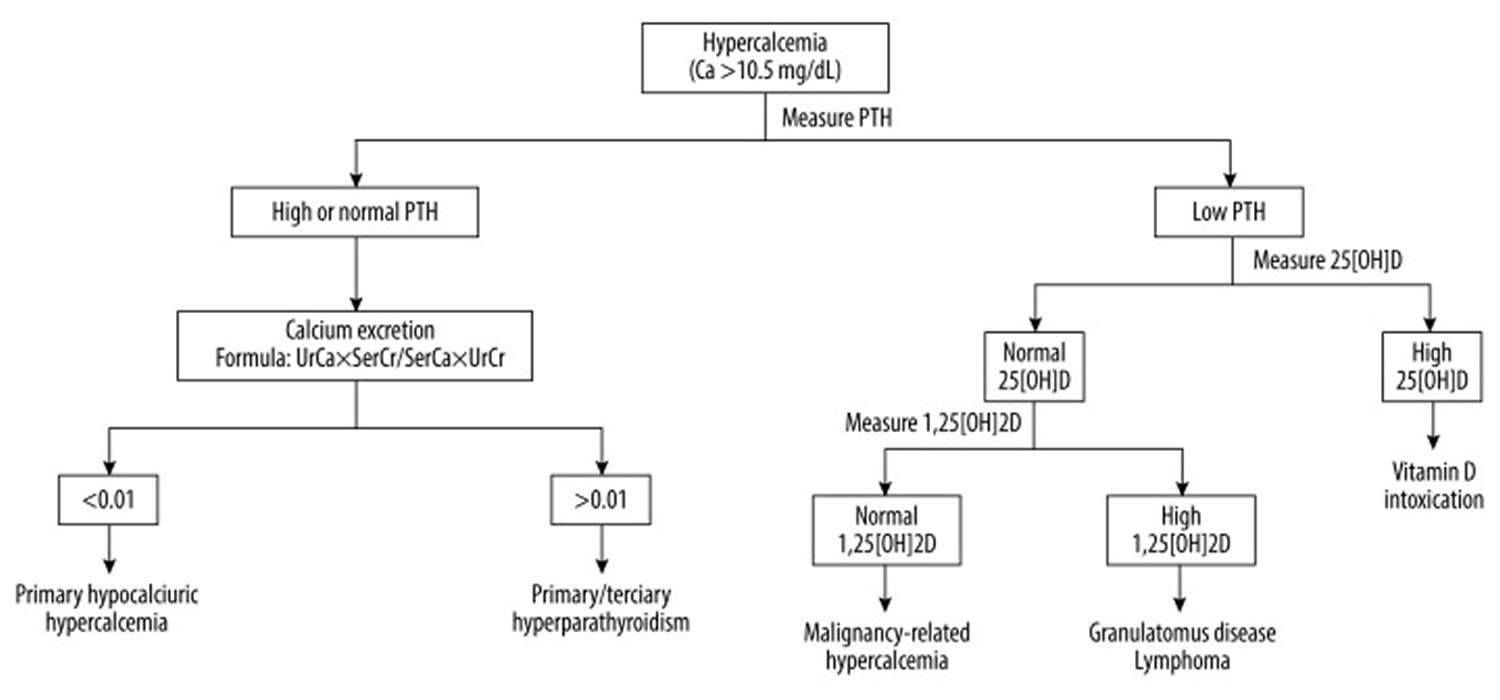
Hypercalcemia
Hypercalcemia means you have too much calcium in your blood or calcium concentration higher than 10.5 mg/dL (> 2.62 millimol/L) 1. Too much calcium in your blood can weaken your bones, create kidney stones, and interfere with how your heart and brain work. Hypercalcemia can result when too much calcium enters the extracellular fluid (body fluid that is not contained in cells) or when there is insufficient calcium excretion from your kidneys. Normal calcium values range from 8.5 to 10.2 mg/dL (2.13 to 2.55 millimol/L) and normal serum ionized calcium (Ca2+) concentration are 4.5 to 5.6 mg/dL (1.12 to 1.4 millimol/L) 2. Hypercalcemia is defined by a serum calcium concentration >10.5 mg/dL, (greater than 2.62 mmol/L) and it is classified into mild, moderate, and severe, depending on calcium values: mild hypercalcemia 10.5 to 12 mg/dL (2.62 to 2.99 mmol/L), moderate hypercalcemia 12.1 to 14 mg/dL (3.02 to 3.49 mmol/L) and severe hypercalcemia greater than 14 mg/dL (>3.5 mmol/L) 1, 3, 4, 5. Mild hypercalcemia is defined as total calcium of 10.5 to 12 mg/dL (2.62 to 2.99 mmol/L) or ionized calcium (Ca2+) of 5.6 to 8.0 mg/dL (1.4 to 2 mmol/L), is usually asymptomatic but may be associated with symptoms such as fatigue and constipation in approximately 20% of people 6. Severe hypercalcemia is defined as total calcium of greater than 14 mg/dL (>3.5 mmol/L) or ionized calcium (Ca2+) of greater than 10 mg/dL (≥2.5 mmol/L) or that develops rapidly over days to weeks, can cause nausea, vomiting, dehydration, confusion, sleepiness or drowsiness (somnolence) and coma 6.
Routinely, serum calcium concentration is evaluated by measuring total calcium. However, in situations such as thrombocytosis (a condition of having too many platelets in your blood), hyperproteinemia (a condition of having abnormally high levels of protein in your blood plasma), hypoproteinemia (a condition of having abnormally low levels of protein in your blood plasma), multiple myeloma, or macroglobulinemia, ionic calcium should be assessed, although the test for this is not widely available 7. When total calcium is measured, the value should be corrected by serum albumin concentration, as albumin is the main protein that carries the calcium ion. The correction is made by the following formula 7:
- Corrected calcium (mg/dL) = Calcium measured (mg/dL)+[0.8×(4–albumin (g/dL)]
Despite being widely used, it is not clear whether this formula needs adjustments for specific populations, such as critically ill patients 7. For these patients, ionized calcium should be tested, if the test is available 7.
Approximately 90% of cases of hypercalcemia are caused by overactive parathyroid glands also known as primary hyperparathyroidism and/or certain kinds of cancers (e.g. carcinoma of the breasts, ovaries, cervix, and esophagus, and tumors in of head or neck region) 8, 9, 10, 11, 12, 13, 14, 15.
Other rare causes of hypercalcemia include the following 7, 16, 17:
- Certain kinds of cancers
- Disease of the endocrine glands
- Taking too much of calcium and vitamin D supplements
- Being on bed rest for a long time
- Kidney failure
- Kidney transplantation
- Certain other medical disorders e.g., granulomatous diseases
- Some medications (e.g., vitamin D, retinoic acid, lithium)
- Genetic causes e.g., familial hypocalciuric hypercalcemia associated with an inactivation mutation in the calcium sensing receptor (CaSR) gene and/or a mutation in the CYP24A1 gene; part of multiple endocrine neoplasia (MEN1 and MEN2)
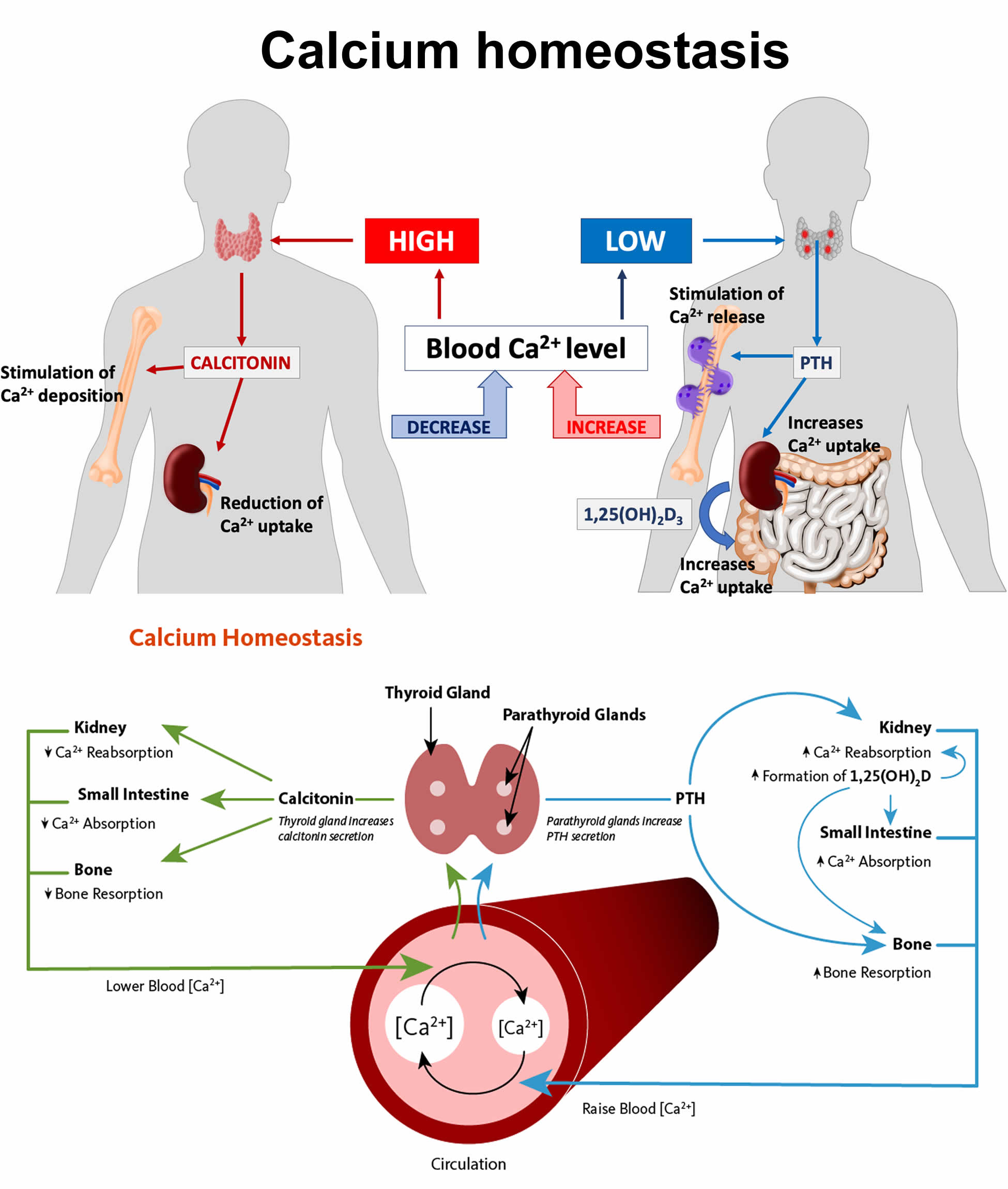
Serum calcium levels are regulated within a narrow range (8.42 to 10.42 mg/dL OR 2.1 to 2.6 mmol/L) by 3 main calcium-regulating hormones: parathyroid hormone (PTH), vitamin D, and to a lesser extent, calcitonin (the regulatory mechanisms of calcium homeostasis are summarized in Figure 1 below), through their specific effects on the bowel, kidneys, and skeleton 18, 19, 20. Imbalance in these regulations may lead to hypercalcemia or hypocalcemia. Approximately half of the total serum calcium is bound to protein (albumin), and the remaining free ionized calcium is physiologically active 20. Serum calcium levels must be corrected for the albumin level before confirming the diagnosis of hypercalcemia or hypocalcemia 21. Serum calcium is very tightly regulated and does not fluctuate with changes in dietary intakes; your body uses bone tissue as a reservoir for, and source of calcium, to maintain constant concentrations of calcium in blood, muscle, and intercellular fluids 22. Unlike your teeth, your bone undergoes continuous remodeling, with constant resorption and deposition of calcium into new bone 23. Bone remodeling is required to change bone size during growth, repair damage, maintain serum calcium levels, and provide a source of other minerals 23.
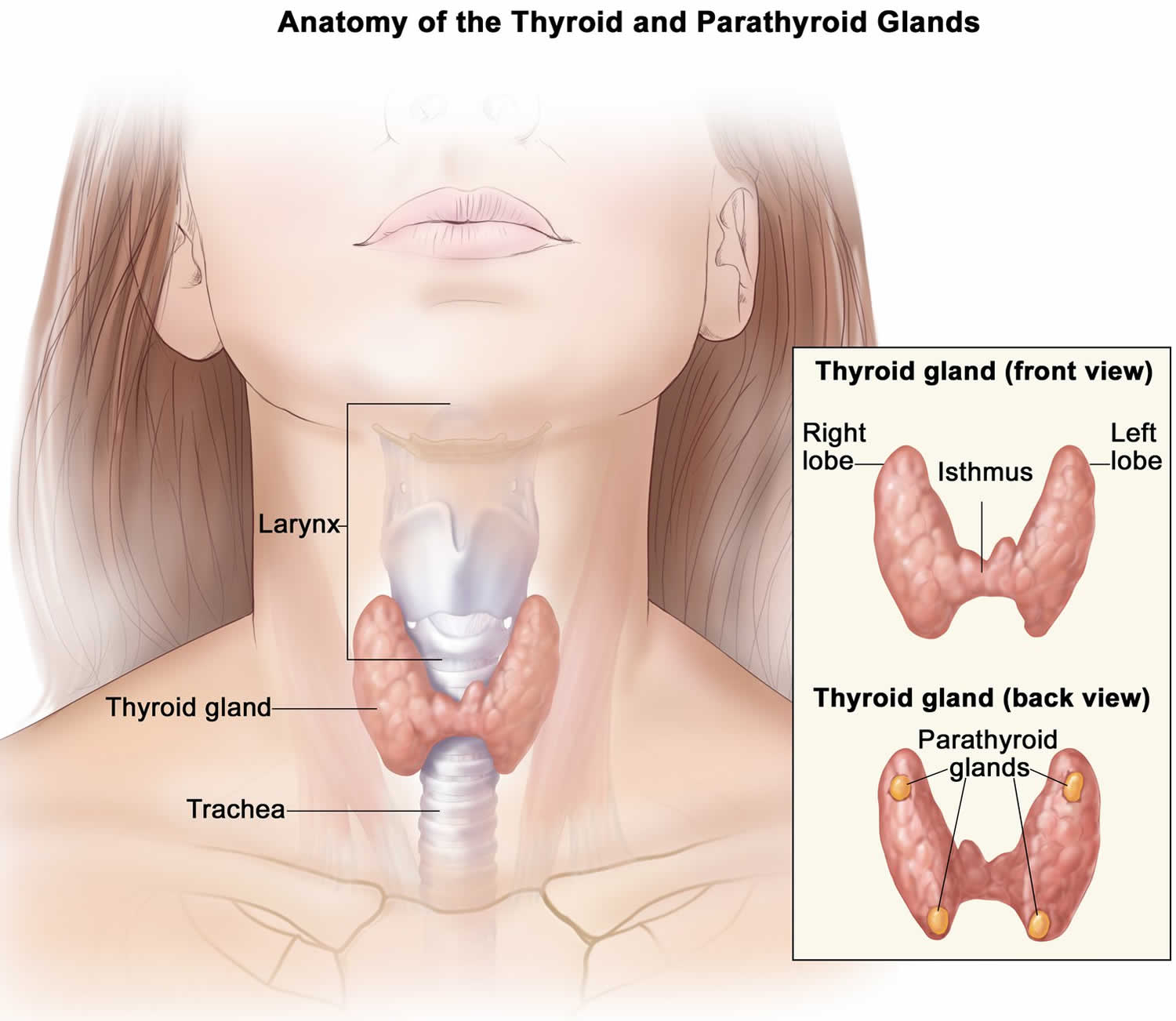
Parathyroid hormone (PTH) is produced by the parathyroid glands, four pea-sized glands that are located behind your thyroid at the bottom of your neck 18, 19. Calcium level is the main determinant for parathyroid hormone (PTH) production and release, which exerts negative feedback on the parathyroids (see Figure 1 below) 5. Parathyroid hormone (PTH) has several actions, but the most important is to defend against low blood calcium or hypocalcemia. Parathyroid cells sense decreases in serum calcium and, in response, release preformed PTH into your bloodstream. Parathyroid hormone (PTH) increases serum calcium within minutes by increasing kidney and intestinal absorption of calcium and by rapidly mobilizing calcium and phosphate from bone (bone resorption) (Figure 1). Kidney calcium excretion generally parallels sodium excretion and is influenced by many of the same factors that govern sodium transport in the proximal tubule. However, PTH enhances distal tubular calcium reabsorption independently of sodium. The consequence of this well-regulated mechanism is re-establishment of adequate calcium concentration 24.
When your blood calcium levels are low, your parathyroid glands secrete parathyroid hormone (PTH) (Figure 1), which acts mainly in your bones and renal tubules; it may also interfere in intestinal calcium absorption 5. The parathyroid hormone (PTH) helps your bones release calcium into your blood. In bones, both parathyroid hormone (PTH) and vitamin D stimulate calcium release into your blood. Additionally, parathyroid hormone (PTH) regulates kidney capacity to reabsorb calcium and through a negative feedback mechanism that inhibits parathyroid hormone (PTH) synthesis in the parathyroid glands (see Figure 1 below). In the bones, parathyroid hormone (PTH) increases the activity and number of osteoclasts, thus increasing bone reabsorption of calcium and phosphorus. In the kidneys, parathyroid hormone (PTH) stimulates tubular calcium reabsorption and phosphorus excretion and increases kidney synthesis of active vitamin D called “calcitriol” or 1,25 dihydroxyvitamin D (1,25-dihydroxycholecalciferol), which is actually a hormone, from the inactive form 25-hydroxycholecalciferol 5, 25. The main effect of calcitriol (active vitamin D) or 1,25 dihydroxyvitamin D (1,25-dihydroxycholecalciferol) is to increase calcium absorption from your intestines 26, 5. Calcitriol or 1,25 dihydroxyvitamin D (1,25-dihydroxycholecalciferol) binds to the vitamin D receptor in the epithelial cells of the duodenum causing the synthesis of calcium binding proteins that regulate active intestinal calcium absorption 26, 27. In the small intestine, vitamin D allows the absorption of calcium through an active transcellular pathway and a passive paracellular pathway. The transcellular pathway requires energy, while the paracellular pathway allows for the passage of calcium through tight junctions. Calcitriol or 1,25 dihydroxyvitamin D (1,25-dihydroxycholecalciferol) also stimulates calcium reabsorption in your kidneys.
The third mechanism regulating calcium homeostasis is calcitonin. Calcitonin is a hormone produced by thyroid parafollicular C cells, which is released into the circulation in response to an acute increase in calcium concentration 5. Calcitonin decreases calcium levels by inhibiting bone osteoclast activity and renal reabsorption of calcium. In the adult, this has a small to negligible effect on calcium homeostasis 28. Furthermore, the role of calcitonin in calcium regulation is not yet well established; for example, after thyroidectomy, in which thyroid parafollicular C cells are removed, there is no calcium disturbance 5.
Together, parathyroid hormone (PTH) and vitamin D, along with other hormones and minerals, help move calcium in or out of body tissues to keep your blood calcium at a normal level.
Parathyroid hormone (PTH) raises blood calcium levels by 29:
- Effects of parathyroid hormone (PTH) on your bones, where most of your body’s calcium is stored, to release calcium into the blood.
- In your bones, parathyroid hormone (PTH) stimulates the release of calcium in an indirect process through osteoclasts which ultimately leads to the resorption of your bones. However, before osteoclast activity, PTH directly stimulates osteoblasts which increases their expression of RANKL, a receptor activator for nuclear factor kappa-B ligand, allowing for the differentiation of osteoblasts into osteocytes. PTH also inhibits the secretion of osteoprotegerin, allowing for preferential differentiation into osteoclasts. Osteoprotegerin normally competitively binds with RANKL diminishing the ability to form osteoclasts. Osteoclasts possess the ability to remodel the bones (resorption) by dissolution and degradation of hydroxyapatite and other organic material, releasing calcium into the blood.
- Effects of parathyroid hormone (PTH) on your kidneys helping your kidneys hold on to calcium and return it to your blood instead of flushing it out in urine.
- In your kidneys, the parathyroid hormone (PTH) has 3 functions in increasing serum calcium levels. Most of the physiologic calcium reabsorption in the nephron takes place in the proximal convoluted tubule and additionally at the ascending loop of Henle. Circulating parathyroid hormone (PTH) targets the distal convoluted tubule and collecting duct, directly increasing calcium reabsorption. Parathyroid hormone (PTH) decreases phosphate reabsorption at the proximal convoluted tubule. Phosphate ions in the serum form salts with calcium that are insoluble, resulting in decreased plasma calcium. The reduction of phosphate ions, therefore, results in more ionized calcium in the blood.
- Also in your kidneys, parathyroid hormone (PTH) stimulates the production of 1-alpha-hydroxylase in the proximal convoluted tubule. This enzyme, 1-alpha-hydroxylase, is required to catalyze the synthesis of active vitamin D called “calcitriol” or 1,25 dihydroxyvitamin D (1,25-dihydroxycholecalciferol) from the inactive form 25-hydroxycholecalciferol 25. The main effect of calcitriol or 1,25 dihydroxyvitamin D (1,25-dihydroxycholecalciferol) is to increase calcium absorption from your gut 26. Calcitriol or 1,25 dihydroxyvitamin D (1,25-dihydroxycholecalciferol) binds to the vitamin D receptor in the epithelial cells of the duodenum causing the synthesis of calcium binding proteins that regulate active intestinal calcium absorption 26, 27. In the small intestine, vitamin D allows the absorption of calcium through an active transcellular pathway and a passive paracellular pathway. The transcellular pathway requires energy, while the paracellular pathway allows for the passage of calcium through tight junctions. Calcitriol or 1,25 dihydroxyvitamin D (1,25-dihydroxycholecalciferol) also stimulates calcium reabsorption in your kidneys.
- PTH indirect effects on your small intestines by helping your intestines absorb calcium from food
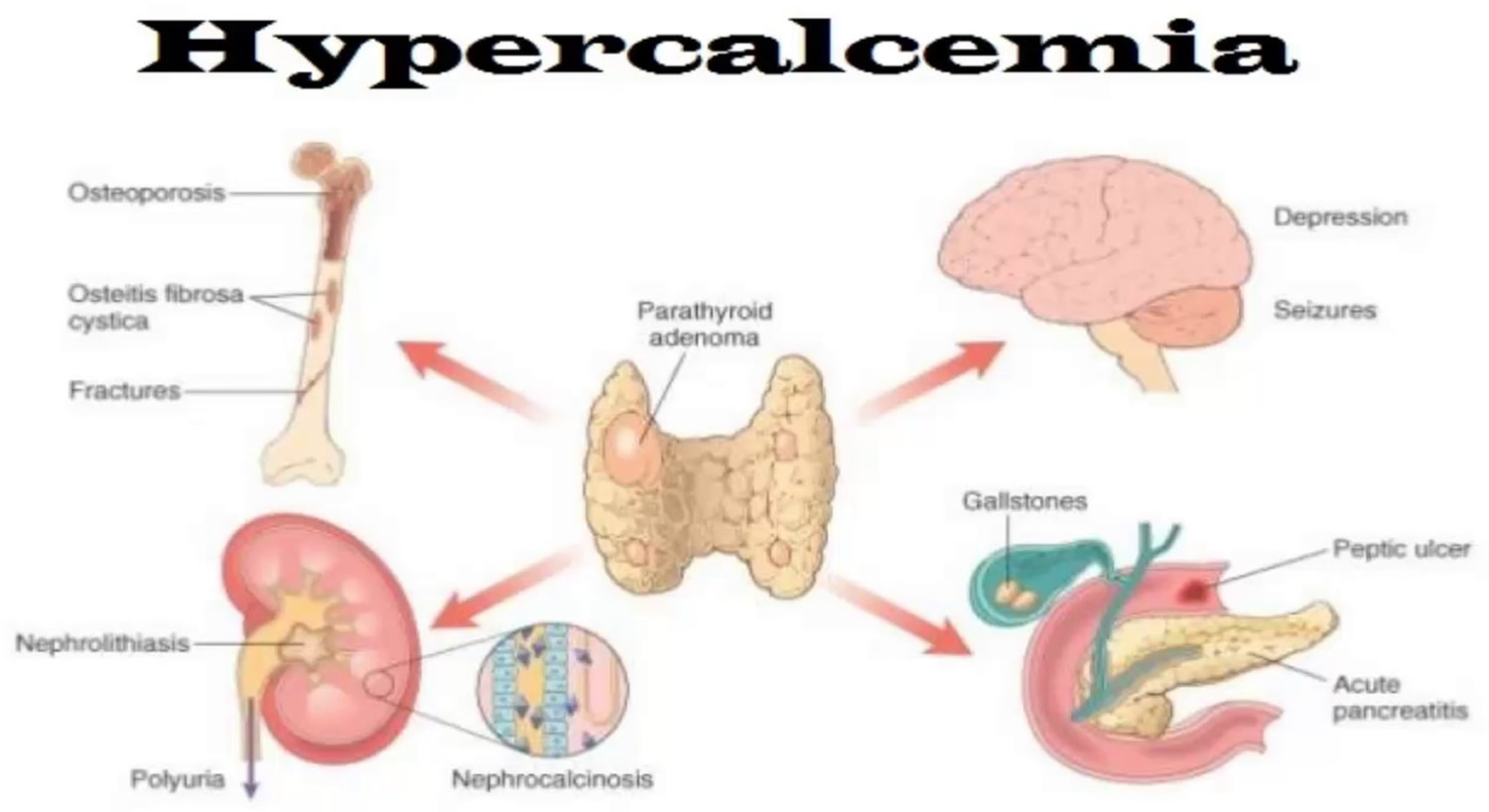
Signs and symptoms of hypercalcemia range from nonexistent to severe. The commonest presentation of hypercalcemia are “moans, bones, stones and groans” referring to depressed mood, musculoskeletal pain, renal colic and abdominal pain related to constipation or peptic ulceration 7. Symptomatic patients may also experience polyuria (production of abnormally large volumes of dilute urine) and polydipsia (excess thirst despite drinking plenty of fluids) secondary to nephrogenic diabetes insipidus and reduced level of consciousness 7. As the severity of hypercalcaemia progresses, nausea, vomiting, QT shortening potentially leading to ventricular fibrillation arrest, confusion and coma may occur 7. Physical signs of hypercalcaemia are relatively few but include band keratopathy, although this is very rare, and signs related to the cause of the hypercalcemia 7. Classical radiographic features of primary hyperparathyroidism, although rarely seen, include resorption of the distal ends of the clavicles, sub periosteal erosions on the radial borders of the middle or terminal phalanges, brown tumours and ‘pepperpot skull’ 7.
Hypercalcemia treatment depends on the underlying cause 30, 31, 32. Treatment for hypercalcemia is required if the patient is symptomatic or if the calcium level is more than 15 mg/dL (>3.74 mmol/L), even in asymptomatic patients 33. The goals of treating hypercalcemia include increased elimination from the extracellular fluid, reducing gastrointestinal absorption and decreasing bone resorption 33. Immediate therapy is directed at restoring intravascular volume and promoting calcium excretion in the urine with an infusion of 0.9% saline at twice the maintenance rate until any fluid deficit is replaced and diuresis occurs (urine output ≥ 200 mL/h to 300 mL/h) 33. Hemodialysis is the treatment of choice to rapidly decrease serum calcium in patients with heart failure or renal insufficiency. Loop diuretics should be used with caution as even though they may enhance renal excretion, paradoxical hypercalcemia can occur due to bone resorption 33.
Patients with hyperparathyroidism require surgical exploration and removal of the source of increased PTH secretion 33. Postoperatively, patients need to be monitored closely for the development of hypocalcemia and tetany. Bisphosphonates such as etidronate, pamidronate, and alendronate are the drugs of choice for hypercalcemia of malignancy as they inhibit osteoclastic activity 33.
Calcitonin can be administered subcutaneously but in most cases, the effects are mild and limited to a few days 33. Mithramycin can block the function of osteoclasts and is often administered to patients with malignancy-associated hypercalcemia. but the drug has significant renal, liver, and bone marrow toxicity.
Hypercalcemia associated with excess vitamin D can be treated with steroids as they inhibit one alpha-hydroxylase activity 33. Ketoconazole, an antifungal agent, has also been used in hypervitaminosis D as it inhibits 1-alpha-hydroxylase activity.
Hypercalcemia of immobilization can be prevented by encouraging activity as tolerated and adequate hydration.
Figure 1. Calcium homeostasis (regulation of serum calcium)
Footnotes: Calcium homeostasis (regulation of serum calcium). Parathyroid hormone (PTH) is produced by the parathyroid glands. Decreased serum calcium concentration stimulates PTH release, which increases calcium bone absorption, renal calcium reabsorption, renal hydroxylation of inactive form of vitamin D called 25-hydroxyvitamin D (25[OH]D) into active form of vitamin D called 1,25 dihydroxyvitamin D (1,25-dihydroxycholecalciferol or calcitriol), and, less importantly, intestinal calcium absorption. Vitamin D is produced by your skin after exposure to ultraviolet B radiation and a small amount comes from diet. In the skin, vitamin D2 and D3 undergo hydroxylation in the liver by 25-hydroxylase, generating 25-hydroxyvitamin D (25[OH]D). In the kidneys, 1-α-hydroxylase converts 25[OH]D into 1,25-dihydroxyvitamin D (1,25[OH]2D). Calcitriol or 1,25 dihydroxyvitamin D (1,25-dihydroxycholecalciferol) increases intestinal calcium absorption.
Figure 2. Thyroid and parathyroid glands anatomy
Footnote: The parathyroid glands are located on or near the thyroid gland in the neck.
What is calcium?
Calcium (Ca or Ca2+) is the most abundant mineral in your body that is found in bones and teeth, in some foods, added to others, available as a dietary supplement, and present in some medicines (such as gastric antacids e.g. calcium phosphate) 34. Calcium is a mineral that your body needs for numerous functions, including building and maintaining bones and teeth, blood clotting, the transmission of nerve impulses, and the regulation of the heart’s rhythm 35. About 99% of the calcium in your body is stored in your bones and teeth in the form of calcium hydroxyapatite [Ca10(PO4)6(OH)2] crystals, an inorganic matrix of calcium and phosphate 22, 23, 36, 37. While the other 1% of the calcium in your body is found in your blood and soft tissue. Calcium concentrations in your blood and fluid surrounding the cells (extracellular fluid) must be maintained within a narrow concentration range for normal physiological functioning. And your body uses your bones as a reservoir for, and source of, calcium to maintain calcium homeostasis (the state of steady or stable equilibrium of internal physical and chemical conditions) 22. Because the physiological functions of calcium are so vital for survival, your body will stimulate bone resorption (demineralization) to maintain normal blood calcium concentrations when calcium intake is inadequate 37. Thus, adequate intake of calcium is a critical factor in maintaining a healthy skeleton 38.
Calcium helps your body with:
- Building strong bones and teeth
- Clotting blood
- Sending and receiving nerve signals
- Squeezing and relaxing muscles
- Releasing hormones and other chemicals
- Keeping a normal heartbeat
Calcium is required for narrowing of blood vessels (vascular contraction) and widening of blood vessels (vasodilation), muscle function, nerve transmission, intracellular signaling and hormonal secretion, though less than 1% of total body calcium is needed to support these critical metabolic functions 22. Serum calcium is very tightly regulated and does not fluctuate with changes in dietary intakes; your body uses bone tissue as a reservoir for, and source of calcium, to maintain constant concentrations of calcium in blood, muscle, and intercellular fluids 22. Unlike your teeth, your bone undergoes continuous remodeling, with constant resorption and deposition of calcium into new bone 23. Bone remodeling is required to change bone size during growth, repair damage, maintain serum calcium levels, and provide a source of other minerals 23.
The balance between bone resorption and deposition changes with age. Bone formation exceeds resorption in periods of growth in children and adolescents, whereas in early and middle adulthood both processes are relatively equal. At birth, the body contains about 26 to 30 g calcium 39. This amount rises quickly after birth, reaching about 1,200 g (1.2 kg) in women and 1,400 g (1.4 kg) in men by adulthood 22. These levels remain constant in men, but they start to drop in women as a result of increases in bone remodeling due to decreased estrogen production at the start of menopause 22. In aging adults, particularly among postmenopausal women, bone breakdown exceeds formation, resulting in bone loss that increases the risk of osteoporosis over time 22.
Your body gets the calcium you need in two ways. One is by eating foods or supplements that contain calcium. Good sources include dairy products, which have the highest concentration per serving of highly absorbable calcium, and dark leafy greens or dried beans, which have varying amounts of absorbable calcium. Calcium supplements often contain vitamin D; taking calcium paired with vitamin D seems to be more beneficial for bone health than taking calcium alone 35.
Calcium is found in many foods. It is important to get plenty of calcium in the foods you eat. You can get recommended amounts of calcium by eating a variety of foods, including the following 40:
- Dairy products such as milk, cheese, and yogurt are the main food sources of calcium for most people in the United States.
- Fish with soft bones that you eat, such as canned sardines and salmon.
- Certain vegetables, such as kale, broccoli, and Chinese cabbage (bok choi) also contain calcium.
- Calcium is added to some breakfast cereals and beverages, including many fruit juices and milk substitutes such as soy and almond beverages, as well as some brands of tofu and ready-to-eat cereals. To find out whether these foods have calcium added, check the product labels.
- Most grains (such as breads, pastas, and unfortified cereals) do not have high amounts of calcium. However, because people eat them often, what they contribute adds up.
The other way your body gets calcium is by pulling it from your bones. This happens when the blood levels of calcium drop too low (hypocalcemia), usually when it’s been awhile since having eaten a meal containing calcium. Ideally, the calcium that is “borrowed” from the bones will be replaced at a later point. But, this doesn’t always happen. Most important, this payback can’t be accomplished simply by eating more calcium 35.
The exact amount of calcium you need depends on your age, sex and other factors 40. Growing children and teenagers need more calcium than young adults. Older women need plenty of calcium to prevent osteoporosis. People who do not eat enough high-calcium foods should take a calcium supplement.
An inverse relationship exists between calcium intake and absorption 39. Absorption of calcium from food is about 45% at intakes of 200 mg/day but only 15% when intakes are higher than 2,000 mg/day 41. Age can also affect absorption of dietary calcium 22, 23. Net absorption of dietary calcium is as high as 60% in infants and young children, who need substantial amounts to build bone, but it decreases to about 25% in adulthood and continues to decline with age 22.
Total calcium levels can be measured in serum or plasma; serum levels are typically 8.8 to 10.4 mg/dL (2. 2 to 2.6 mmol/L) in healthy people 42, 22. However, serum calcium levels do not reflect nutritional status because of their tight homeostatic control 23. Levels of ionized or free calcium (Ca2+), the biologically active form, in serum are also used to measure calcium status. The normal range of ionized calcium (Ca2+) in healthy people is 4.6 to 5.3 mg/dL (1.15 to 1.33 mmol/L) 42. Dual x-ray absorptiometry (DEXA) testing of bone mineral density (BMD) can be used to assess cumulative calcium status over the lifetime because the skeleton stores almost all calcium in the body 43.
Calcium and phosphate concentrations are linked by their ability to chemically react to form calcium phosphate. The product of concentrations of calcium and phosphate (in mEq/L) is estimated to be < 60 normally; when the product exceeds 70, precipitation of calcium phosphate crystals in soft tissue is much more likely. Calcification of vascular tissue accelerates arteriosclerotic vascular disease and may occur when the calcium and phosphate product is even lower (> 55), especially in patients with chronic kidney disease.
What does calcium do?
Calcium is a mineral your body needs to build and maintain strong bones and to carry out many important functions. Your body needs calcium for muscles to move and for nerves to carry messages between your brain and every part of your body. Calcium also helps blood vessels move blood throughout your body and helps release hormones that affect many functions in your body. Vitamin D helps your body absorb calcium.
Calcium plays a role in:
- strengthening bones and teeth
- regulating muscle functioning, such as contraction and relaxation
- regulating heart functioning
- blood clotting
- transmission of nervous system messages
- enzyme function.
Calcium acts as a cofactor for enzymes such as adenosine triphosphatase, collagenases, neutral protease, transglutaminase, and others. It also participates in the coagulation cascade, activating platelets and coagulation factors including Factor XIII 44, regulates muscle contraction, binding to troponin C on actin filaments, or calmodulin, which modulates myosin filaments 45, and stabilizes cell membranes by decreasing permeability to ions and altering membrane potential 46, 47, 14.
Structural element in bones and teeth
Calcium is a major structural element in bones and teeth. The mineral component of bone consists mainly of hydroxyapatite [Ca10(PO4)6(OH)2] crystals, which contain large amounts of calcium, phosphorus, and oxygen. Bone is a dynamic tissue that is remodeled throughout life. Bone cells called osteoclasts begin the process of remodeling by dissolving or resorbing bone. Bone-forming cells called osteoblasts then synthesize new bone to replace the bone that was resorbed. During normal growth, bone formation exceeds bone resorption. Osteoporosis may result when bone resorption chronically exceeds formation 22.
Calcium homeostasis
Calcium concentrations in the blood and fluid that surround cells are tightly controlled in order to preserve normal physiological function. A slight drop in blood calcium concentration (e.g., in the case of inadequate calcium intake) is sensed by the parathyroid glands, resulting in their increased secretion of parathyroid hormone (PTH). In the kidneys, PTH stimulates the conversion of vitamin D into its active form (1,25-dihydroxyvitamin D; calcitriol), which rapidly decreases urinary excretion of calcium but increases urinary excretion of phosphorus. Elevations in PTH also stimulates bone resorption, resulting in the release of bone mineral (calcium and phosphate) — actions that also contribute to restoring serum calcium concentrations. Increased circulating calcitriol (1,25-dihydroxyvitamin D) also triggers intestinal absorption of both calcium and phosphorus. Like PTH, calcitriol (1,25-dihydroxyvitamin D) stimulates the release of calcium from bone by activating osteoclasts (bone-resorbing cells). When blood calcium rises to normal levels, the parathyroid glands stop secreting PTH. A slight increase in blood calcium concentration stimulates the production and secretion of the peptide hormone, calcitonin, by the thyroid gland. Calcitonin inhibits PTH secretion, decreases both bone resorption and intestinal calcium absorption, and increases urinary calcium excretion (Figure 1). Finally, acute changes in blood calcium concentrations do not seem to elicit the secretion of the phosphaturic hormone fibroblast growth factor 23 (FGF-23), which is produced by bone-forming cells (osteoblasts/osteocytes) in response to increases in phosphorus intake 48. While this complex system allows for rapid and tight control of blood calcium concentrations, it does so at the expense of the skeleton 22.
Cell signaling
Calcium plays a role in mediating the constriction and relaxation of blood vessels (vasoconstriction and vasodilation), nerve impulse transmission, muscle contraction, and the secretion of hormones like insulin 22. Excitable cells, such as skeletal muscle and nerve cells, contain voltage-dependent calcium channels in their cell membranes that allow for rapid changes in calcium concentrations. For example, when a nerve impulse stimulates a muscle fiber to contract, calcium channels in the cell membrane open to allow calcium ions into the muscle cell. Within the cell, these calcium ions bind to activator proteins, which help release a flood of calcium ions from storage vesicles of the endoplasmic reticulum (ER) inside the cell. The binding of calcium to the protein troponin-c initiates a series of steps that lead to muscle contraction. The binding of calcium to the protein calmodulin activates enzymes that break down muscle glycogen to provide energy for muscle contraction. Upon completion of the action, calcium is pumped outside the cell or into the endoplasmic reticulum (ER) until the next activation 49.
Regulation of protein function
Calcium is necessary to stabilize a number of proteins, including enzymes, optimizing their activities. The binding of calcium ions is required for the activation of the seven “vitamin K-dependent” clotting factors in the coagulation cascade. The term, “coagulation cascade,” refers to a series of events, each dependent on the other that stops bleeding through clot formation.
Hypercalcemia causes
Besides building strong bones and teeth, calcium helps muscles contract and nerves transmit signals. Normally, if there isn’t enough calcium in your blood, your parathyroid glands secrete a parathyroid hormone (PTH) hormone that triggers:
- Your bones to release calcium into your blood
- Your digestive tract to absorb more calcium
- Your kidneys to excrete less calcium and activate more vitamin D, which plays a vital role in calcium absorption
Parathyroid hormone (PTH) and vitamin D help manage calcium balance in the body.
- Parathyroid hormone (PTH) is made by the parathyroid glands. These are four small glands located in the neck behind the thyroid gland.
- Vitamin D is obtained when the skin is exposed to sunlight, and from dietary sources.
This delicate balance between too little calcium in your blood and hypercalcemia can be disrupted by a variety of factors.
The most common cause of high calcium blood level is excess parathyroid hormone (PTH) released by the parathyroid glands. This excess occurs due to:
- An enlargement of one or more of the parathyroid glands.
- A growth on one of the glands. Most of the time, these growths are benign.
Women older than 50 are at highest risk of overactive parathyroid glands.
Men and women of all ages can have a high blood calcium level. However, it is most common in women over age 50 (after menopause). In most cases, this is due to an overactive parathyroid gland.
Hypercalcemia can be caused by:
- Overactive parathyroid glands (hyperparathyroidism). There are two types of hyperparathyroidism.
- Primary hyperparathyroidism is most common cause of hypercalcemia and can stem from a small, noncancerous (benign) tumor or enlargement of one or more of the four parathyroid glands causing overproduction of parathyroid hormone (PTH). This causes high calcium levels in the blood, which can cause a variety of health problems. Surgery is the most common treatment for primary hyperparathyroidism. Approximately 85% of all primary hyperparathyroidism arises because of a solitary parathyroid adenoma (noncancerous benign tumor) and 1% are due to parathyroid carcinoma (parathyroid cancer) and the remainder are due to enlargement (hyperplasia) of two or more parathyroid glands 50. Although the majority of primary hyperparathyroidism occurs randomly, parathyroid adenomas are often observed in patients with chronic renal failure or after kidney transplantation who fail to achieve of normal calcium and vitamin D homeostasis 51. Furthermore, a proportion arises on the background of a familial predisposition and some people inherit a gene that causes the disorder (Table 1) 52. Therefore, during clinical assessment of primary hyperparathyroidism, a family history should always be sought and other endocrinopathies as may be seen in, for example, multiple endocrine neoplasia (MEN1 and MEN2). Occurrence of primary hyperparathyroidism at a young age (approximately <45 years) and/or presence of multi-gland disease should also prompt consideration of multiple endocrine neoplasia (MEN).
- Multiple endocrine neoplasia type 1 (MEN1) is an autosomal-dominant tumor syndrome characterized by the occurence of tumors in multiple endocrine tissues, e.g. parathyroid gland (95 %), enteropancretic and/or neuroendocrine system (50 %) and anterior pituitary gland (40 %) are affected 53. A germline-inactivating mutation in the MEN1 gene is accompanied by a complete loss of function of its product menin. Pathways and interactions of this protein could explain some pathophysiological aspects of MEN1 53.
- Adenoma of the parathyroid gland may also accur together with medullary thyroid carcinoma (MEN2A). The syndrome develops based on protooncogene activation of RET kinase in MEN2 gene 54, 55, 56.
- Isolated benign familial hyperparathyroidism may be a consequence of a mutation in the CDKN1B (MEN4) gene. Furthermore, rare parathyroid carcinomas are associated with mutation in the CDC73/HRPT2 gene 57. Expression of additional possible hypothetical parathyroid oncogenes and/or tumor-suppressor genes, which could provide better insight into pathophysiology of primary hyperparathyroidism, remains to be found and investigated.
- Secondary hyperparathyroidism occurs due to another disease that first causes low calcium levels in the body (hypocalcemia), which then affects the parathyroid gland’s function. This causes your parathyroid glands to overwork and produce high amounts of parathyroid hormone (PTH) to maintain or restore the calcium level to the normal range. This is common in kidney disease and after certain intestinal surgeries or diseases. Factors that may result in secondary hyperparathyroidism include:
- Chronic kidney failure. Your kidneys convert vitamin D into a form that your body can use. If your kidneys work poorly, usable vitamin D may decrease and calcium levels drop. This causes parathyroid hormone levels to go up. Chronic kidney failure is the most common cause of secondary hyperparathyroidism. In some people with long-term secondary hyperparathyroidism, usually from end-stage kidney disease, the parathyroid glands enlarge. They begin to release parathyroid hormone on their own. The hormone level doesn’t go down with medical treatment and the blood calcium becomes too high. This is called tertiary hyperparathyroidism, and people with this condition may require surgery to remove parathyroid tissue.
- Severe calcium deficiency. Your body may not get enough calcium from your diet, often because your digestive system doesn’t absorb the calcium from food. This is common after intestinal surgery, including weight loss surgery.
- Severe vitamin D deficiency. Vitamin D helps maintain appropriate calcium levels in the blood. It also helps your digestive system absorb calcium from your food. Your body produces vitamin D when your skin is exposed to sunlight. You also get some vitamin D in food. If you don’t get enough vitamin D, then calcium levels may drop.
- Primary hyperparathyroidism is most common cause of hypercalcemia and can stem from a small, noncancerous (benign) tumor or enlargement of one or more of the four parathyroid glands causing overproduction of parathyroid hormone (PTH). This causes high calcium levels in the blood, which can cause a variety of health problems. Surgery is the most common treatment for primary hyperparathyroidism. Approximately 85% of all primary hyperparathyroidism arises because of a solitary parathyroid adenoma (noncancerous benign tumor) and 1% are due to parathyroid carcinoma (parathyroid cancer) and the remainder are due to enlargement (hyperplasia) of two or more parathyroid glands 50. Although the majority of primary hyperparathyroidism occurs randomly, parathyroid adenomas are often observed in patients with chronic renal failure or after kidney transplantation who fail to achieve of normal calcium and vitamin D homeostasis 51. Furthermore, a proportion arises on the background of a familial predisposition and some people inherit a gene that causes the disorder (Table 1) 52. Therefore, during clinical assessment of primary hyperparathyroidism, a family history should always be sought and other endocrinopathies as may be seen in, for example, multiple endocrine neoplasia (MEN1 and MEN2). Occurrence of primary hyperparathyroidism at a young age (approximately <45 years) and/or presence of multi-gland disease should also prompt consideration of multiple endocrine neoplasia (MEN).
- Cancer. Lung cancer and breast cancer, as well as some blood cancers, can increase your risk of hypercalcemia. Spread of cancer (metastasis) to your bones also increases your risk 58. Several mechanisms account for hypercalcemia due to malignancy:
- Humoral hypercalcemia of malignancy arises because of secretion of parathyroid hormone-related protein (PTHrP).
- Osteolytic metastases.
- A limited range of neoplasms express 1 alpha hydroxylase and are able to activate vitamin D to the active Calcitriol or 1,25 dihydroxyvitamin D (1,25-dihydroxycholecalciferol) and drive hypercalcemia. Lymphomas are the commonest tumor in this category.
- True ectopic production of PTH is very rare. Of course, a diagnosis of carcinoma does not rule out the simultaneous presence of primary hyperparathyroidism, which is common.
- Granulomatous diseases, such as tuberculosis, sarcoidosis, histoplasmosis, leprosy, coccidioidomycosis or cryptococcus, can raise blood levels of vitamin D, which stimulates your digestive tract to absorb more calcium 59. Hypercalcemia is characterized by high serum calcitriol or 1,25 dihydroxyvitamin D and elevated 24-h urine calcium. Granuloma calcifications in stricken tissues or lymph nodes are visible on CT scans 60. Hypercalcemia can prezent as a life-threatening hypercalcemic crisis, nephrocalcinosis, artery calcifications or encephalopathy 61, 62. Hypercalcemic crisis which occurs when the blood calcium levels are very high and causes neurological symptoms like confusion, extreme lethargy and even coma in some cases.
- Medical conditions. Hyperthyroidism (overactive thyroid or thyrotoxicosis), pheochromocytoma and Addison’s disease or adrenal insufficiency
- Genetic disorders. A rare genetic disorder known as familial hypocalciuric hypercalcemia causes an increase of calcium in your blood because of faulty calcium receptors in your body. Familial hypocalciuric hypercalcemia doesn’t cause symptoms or complications of hypercalcemia 63.
- Being bed-bound (or not being able to move) for a long period of time. People who have a condition that causes them to spend a lot of time sitting or lying down can develop hypercalcemia. Over time, bones that don’t bear weight release calcium into the blood.
- Severe dehydration. A common cause of mild or transient hypercalcemia is dehydration. Having less fluid in your blood causes a rise in calcium concentrations.
- Medications. Certain drugs — such as lithium, used to treat bipolar disorder — might increase the release of parathyroid hormone. Other drugs include thiazide diuretics and antacids (‘milk alkali syndrome’).
- Supplements. Taking excessive amounts of calcium, vitamin D or vitamin A supplements over time can raise calcium levels in your blood above normal.
- Hypercalcemia of pregnancy and lactation. Hypercalcemia during pregnancy is uncommon, although dangerous from the point of maternal and fetal morbidity, especially when a hypercalcemic crisis develops. Hypercalcemic crisis which occurs when the blood calcium levels are very high and causes neurological symptoms like confusion, extreme lethargy and even coma in some cases. Hypercalcemia during pregnancy is usually caused by maternal primary hyperparathyroidism. After localization of the parathyroid adenoma (noncancerous benign tumor) using ultrasonogarphy, surgery during the second trimester becomes the only curative treatment 64. Hypercalcemia during pregnancy and lactation can also be caused by excessive production of parathyroid hormone-related protein (PTHrP), which is physiologically synthetized in the placenta and mammary glands. Parathyroid hormone-related protein (PTHrP) production is under the control of peripheral serotonin (5-OH tryptamine), which is necessary for proper mammary gland function. Genetic ablation of tryptophan hydroxylase 1 (Tph1) at the start of lactation reduces parathyroid hormone-related protein (PTHrP) synthesis and decreases osteoclast activity and calcemia values in animals. Futhermore, extreme deficiencies in tryptophan hydroxylase may lead to lactation hypocalcemia. The syndrome is treatable with daily injection of 5-hydroxytryptamine 65. A hypercalcemic crisis after delivery is extremely dangerous. It has been successfully treated using saline infusions and administration of bisphosphonates 66.
Table 1. Genes implicated in primary hyperparathyroidism
| Disorder | Gene |
|---|---|
| Multiple Endocrine Neoplasia Type 1 (MEN1) | MEN1 |
| Multiple Endocrine Neoplasia Type 2 (MEN2) | RET |
| Multiple Endocrine Neoplasia Type 4 (MEN4) | CDKN1B |
| Hyperparathyroidism jaw tumor syndrome | CDC73 |
| Familial isolated hyperparathyroidism | MEN1, CDC73, CASR, CDKN1A, CDKN2B, CDKN2C |
| Neonatal severe primary hyperparathyroidism | CASR |
| Non syndromic primary hyperparathyroidism | PTH |
Primary hyperparathyroidism
Hyperparathyroidism is when your parathyroid glands create high amounts of parathyroid hormone (PTH) in the bloodstream. In primary hyperparathyroidism, an enlargement of one or more of the four parathyroid glands causes overproduction of parathyroid hormone (PTH). Too much parathyroid hormone (PTH) causes calcium levels in your blood to rise too high or hypercalcemia, which can lead to variety of health problems such as bone thinning and kidney stones. “Primary” means this disorder begins in the parathyroid glands, rather than resulting from another health problem such as kidney failure.
Primary hyperparathyroidism most often affects people between age 50 and 60. Women are affected 3 to 4 times more often than men to develop primary hyperparathyroidism, and the risk increases with age 67. The prevalence of primary hyperparathyroidism differs in different populations.
Eufrazion et al. 68 in cross-sectional study including 4,207 Brazilian subjects found the prevalence of primary hyperparathyroidism to be 0.78, of which 82% were asymptomatic. This means that a huge number of patients with primary hyperparathyroidism are yet to be identified. The incidence of primary hyperparathyroidism differs according to
populations. Griebeler et al. 69 in a sample from the United States found an incidence of 86 patients/10,000 persons/year from 1998-2010. In the United States, about 100,000 people develop primary hyperparathyroidism each year 70, 67. The prevalence is about one to seven cases per 1,000 adults. Primary hyperparathyroidism is more common in the African-American population, followed by Caucasians, in one large study performed in North America 67. Recent research has found increase trend in primary hyperparathyroidism incidence. Although the reasons are unclear, it may in part be explained by changes in osteoporosis screening guidelines 69.
Approximately 85% of all primary hyperparathyroidism arises because of a solitary parathyroid adenoma (noncancerous benign tumor) and 1% are due to parathyroid carcinoma (parathyroid cancer) and the remainder are due to enlargement (hyperplasia) of two or more parathyroid glands 50. Although the majority of primary hyperparathyroidism occurs randomly, parathyroid adenomas are often observed in patients with chronic renal failure or after kidney transplantation who fail to achieve of normal calcium and vitamin D homeostasis 51. Furthermore, a proportion arises on the background of a familial predisposition and some people inherit a gene that causes the disorder (Table 1) 52. Therefore, during clinical assessment of primary hyperparathyroidism, a family history should always be sought and other endocrinopathies as may be seen in, for example, multiple endocrine neoplasia (MEN1 and MEN2). Occurrence of primary hyperparathyroidism at a young age (approximately <45 years) and/or presence of multi-gland disease should also prompt consideration of multiple endocrine neoplasia (MEN).
Doctors usually catch primary hyperparathyroidism early through routine blood tests, before signs or symptoms of the disorder occur 71. This is usually because an elevated level of calcium or hypercalcemia is found on routine blood tests. When symptoms do occur, they’re the result of damage or dysfunction in other organs or tissues. This damage or dysfunction is due to high calcium levels in the blood and urine or too little calcium in bones.
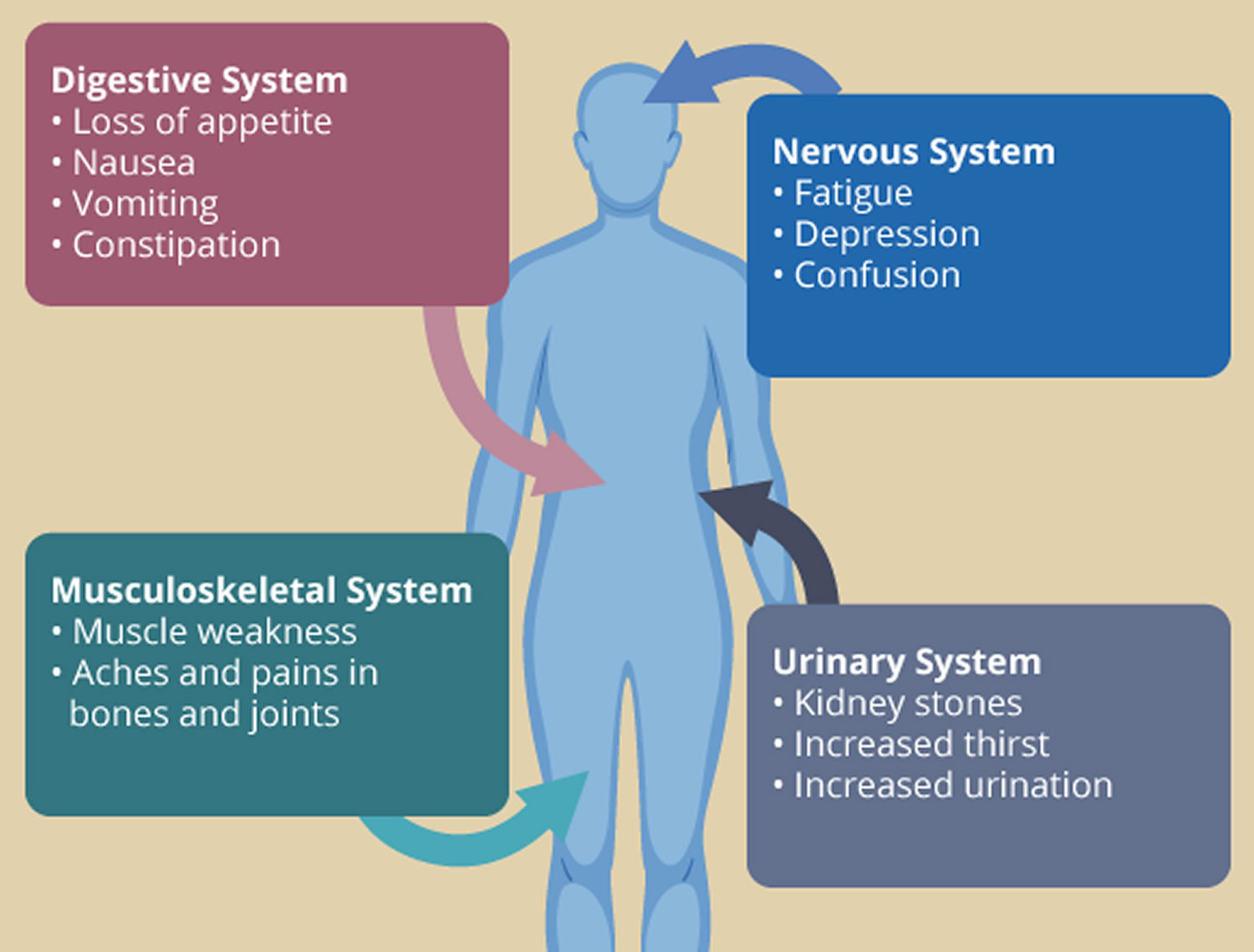
Most people with primary hyperparathyroidism have no symptoms. When symptoms appear, they’re often mild and similar to those of many other disorders. The range of signs and symptoms include:
- Muscle weakness
- Fatigue
- Tiring easily or weakness
- Depression or forgetfulness
- Aches and pains in bones and joints
People with more severe disease may have:
- Loss of appetite
- Nausea
- Vomiting
- Constipation
- Confusion
- Increased thirst
- Excessive urination
- Weak bones that break easily (osteoporosis)
- Kidney stones
- Stomach (abdominal) pain
- Frequent complaints of illness with no clear cause
Complications of hyperparathyroidism are mainly related to the long-term effect of too little calcium in your bones and too much calcium in your bloodstream. Common complications include:
- Weakened bones. High parathyroid hormone (PTH) levels trigger the bones to release more calcium than normal into the blood. The loss of calcium from the bones often results in weak, brittle bones that break easily (osteoporosis).
- Kidney stones. The small intestine may absorb more calcium from food, adding to high levels of calcium in your blood. Extra calcium that isn’t used by your bones and muscles goes to your kidneys and is flushed out in urine. Too much calcium in your blood may lead to too much calcium in your urine. Too much calcium in your urine can cause kidney stones. A kidney stone usually causes major pain as it passes from the kidneys through the urinary tract.
- Other complications. High blood calcium levels might play a part in other problems, such as heart disease, high blood pressure, and trouble concentrating. However, more research is needed to better understand how primary hyperparathyroidism affects the heart, blood vessels, and brain.
- Neonatal hypoparathyroidism. Severe, untreated hyperparathyroidism in pregnant women may cause dangerously low levels of calcium in newborns. Primary hyperparathyroidism is not common in women of childbearing age.
Figure 3. Primary hyperparathyroidism symptoms
[Source 72 ]Primary hyperparathyroidism causes
In about 8 out of 10 people with primary hyperparathyroidism, a benign, or noncancerous, tumor called an adenoma has formed in one of the parathyroid glands 73. The tumor causes the parathyroid gland to become overactive. In most other cases, extra PTH comes from two or more parathyroid adenomas or from parathyroid gland hyperplasia, a condition in which all four parathyroid glands are enlarged. People with rare inherited conditions that affect the parathyroid glands, such as multiple endocrine neoplasia type 1 (MEN1) or familial hypocalciuric hypercalcemia, are more likely to have more than one gland affected.
Rarely, primary hyperparathyroidism is caused by cancer of a parathyroid gland.
Primary hyperparathyroidism diagnosis
Doctors diagnose primary hyperparathyroidism when a blood test shows high blood calcium and parathyroid hormone (PTH) levels. Sometimes parathyroid hormone (PTH) levels are in the upper portion of the normal range, when they should drop to low-normal or below normal in response to high calcium levels. Other conditions can cause high calcium, but elevated parathyroid hormone (PTH) is the only source in primary hyperparathyroidism.
Routine blood tests can detect high blood calcium levels. High blood calcium may cause health care professionals to suspect hyperparathyroidism, even before symptoms appear.
Sometimes PTH levels are high but calcium levels are not. Doctors don’t routinely test for PTH but may do so if you have osteoporosis or another disorder that affects bone strength. In some cases, this may be the first phase of primary hyperparathyroidism, before calcium levels start to rise.
Once doctors diagnose hyperparathyroidism, a 24-hour urine collection can help find the cause. This test measures certain chemicals, such as calcium and creatinine, a waste product that healthy kidneys remove. You will collect your urine over a 24-hour period and your health care professional will send it to a lab for analysis. Results of the test may help tell primary hyperparathyroidism from hyperparathyroidism caused by a kidney disorder. The test can also rule out familial hypocalciuric hypercalcemia, a rare genetic disorder, as a cause.
Primary hyperparathyroidism treatment
After a diagnosis of primary hyperparathyroidism is made, your doctor chooses a treatment based on your age, test results, and whether you have signs and symptoms. Treatment options for primary hyperparathyroidism can include watchful waiting, surgery and medications. In general, surgical removal of adenomas or enlarged parathyroid glands (parathyroidectomy) is recommended for all patients under the age of 50, whether or not they have symptoms 70. This is because young people tend to develop more complications over time if they are untreated.
Surgery or parathyroidectomy also is suggested for people with:
- Osteoporosis or kidney stones (nephrolithiasis)
- High levels of blood calcium (above a certain level)
- Impaired kidney function
- High levels of urinary calcium
- Fragility fractures (pathologic fractures), osteoporosis or evidence of spinal compression fractures
Guidelines help doctors to decide whether or not parathyroid surgery (parathyroidectomy) should be recommended. You might be a candidate for surgery (parathyroidectomy) if you meet any of these guidelines 74:
- A serum (albumin-corrected) calcium level greater than 1 mg/dL above normal
- Bone density by DXA < -2.5 at any site (lumbar spine, hip, or forearm) indicating osteoporosis
- History of kidney stones or evidence of kidney stones (asymptomatic nephrolithiasis on imaging) or calcifications in the kidney by imaging (e.g., X-ray, ultrasound, CT scan). Evidence for stone risk by 24-hour urine with excessive calcium or hypercalciuria [defined as a 24-hour urine calcium level greater than 400 mg/dL] and other stone risk factors e.g., impaired renal function [defined as a glomerular filtration rate less than 60 mL/minute]).
- A fracture resulting from relatively little force, such as a fall from a standing or sitting position (a fragility fracture)
- Age younger than age 50 years
Doctors most often recommend parathyroid surgery, particularly if their patients meet one or more of the guidelines noted above.
It is also not inappropriate to recommend surgery in those who do not meet guidelines as long as there are no medical contraindications to surgery. In those who do not meet guidelines or do not choose surgery, the doctor will monitor the patient’s condition. If there is evidence for progressive disease (e.g., higher calcium level, lower bone density, a fracture, kidney stone), surgery would be advised. For patients who are not going to have parathyroid surgery, even though guidelines are met, doctors can prescribe medicines to control the high blood calcium or improve the bone density.
Other findings that should prompt consideration for parathyroidectomy in patients without frank, objective evidence of disease were previously debated, because there is less definitive evidence that they are caused by the primary hyperparathyroidism, and they are often multifactorial in nature. These include frailty or diminished functional capacity, gastroesophageal reflux, neurocognitive dysfunction, and (less commonly) fibromyalgia or cardiovascular disease 74, 75, 76. In patients with normocalcemic hyperparathyroidism, it is important to rule out secondary hyperparathyroidism—most commonly from vitamin D deficiency.
Complications from surgery aren’t common. Risks include:
- Damage to nerves controlling the vocal cords.
- Long-term low calcium levels requiring the use of calcium and vitamin D supplements due to removal or damage to all parathyroid glands. This means the body cannot produce enough parathyroid hormone to keep the calcium in the standard range.
Observation and/or pharmacologic management of primary hyperparathyroidism is not therapeutically or cost-effective for patients who are surgical candidates, regardless of symptomatology 74. For the patient who cannot undergo surgery, medical options tailored to the individual patient include antiresorptives for osteoporosis (bisphosphonates or denosumab) or the calcium-sensing receptor agonist cinacalcet for hypercalcemia control 77.
Medications to treat hyperparathyroidism include the following:
- Calcimimetics. A calcimimetic is a drug that mimics calcium circulating in the blood. The drug may trick the parathyroid glands into releasing less parathyroid hormone. This drug is sold as cinacalcet (Sensipar).
- Cinacalcet may be an option to treat primary hyperparathyroidism, particularly if surgery hasn’t successfully cured the disorder or a person isn’t a good surgery candidate.
- Cinacalcet and vitamin D analogs (prescription forms of vitamin D) are used to manage secondary hyperparathyroidism in chronic kidney disease. These medications help keep the balance of calcium and phosphorus minerals so that the parathyroid glands don’t have to work hard.
- The most commonly reported side effects of cinacalcet are joint and muscle pain, diarrhea, nausea, and respiratory infection.
- Hormone replacement therapy. For women who have gone through menopause and have signs of osteoporosis, hormone replacement therapy may help bones keep calcium. However, this treatment doesn’t address the underlying problems with the parathyroid glands. Prolonged use of hormone replacement therapy can increase the risk of blood clots and breast cancer. Work with your health care provider to evaluate the risks and benefits to help you decide what’s best for you.
- Some common side effects of hormone replacement therapy include breast pain and tenderness, dizziness, and headaches.
- Bisphosphonates. Bisphosphonates also prevent the loss of calcium from bones and may lessen osteoporosis caused by hyperparathyroidism. Some side effects associated with bisphosphonates include low blood pressure, a fever and vomiting. This treatment doesn’t address the underlying problems with the parathyroid glands, and the blood calcium level remains above the standard range.
Familial hypocalciuric hypercalcemia
Familial hypocalciuric hypercalcemia (FHH) is an inherited disorder of mineral homeostasis that is transmitted as an autosomal dominant trait 78. Familial hypocalciuric hypercalcemia is characterized biochemically by lifelong abnormally high levels of calcium in the blood (hypercalcemia) with low to moderate levels of calcium in urine (hypocalciuric defined as a calcium-to-creatinine clearance ratio less than 0.01 and 24-hour urinary calcium excretion less than 100 mg) and normal or mildly elevated circulating parathyroid hormone (PTH) level that is not suppressed by the hypercalcemia 79, 80, 74. Individuals with familial hypocalciuric hypercalcemia are usually asymptomatic and the disorder is considered benign and are often diagnosed by chance during routine bloodwork 81.
Weakness, fatigue, issues with concentration, and excessive thirst (polydipsia) have been reported by some people with familial hypocalciuric hypercalcemia 81. Rarely, people with familial hypocalciuric hypercalcemia experience inflammation of the pancreas (pancreatitis) or a buildup of calcium in the joints (chondrocalcinosis) 81.
In familial hypocalciuric hypercalcemia, an autosomal-dominant inherited mutation in the calcium-sensing receptor (CaSR) gene results in the inability of the parathyroid glands and kidneys to recognize alterations in serum calcium levels 81, 78. Higher calcium levels are needed to lower parathyroid hormone (PTH) secretion, and the kidneys reabsorb more calcium. Although occasionally patients with familial hypocalciuric hypercalcemia manifest symptoms of hypercalcemia and thus should be monitored clinically, they do not benefit from parathyroidectomy.
Familial hypocalciuric hypercalcemia is mainly classified into three different types depending on the genetic cause:
- Familial hypocalciuric hypercalcemia type 1 (FHH1) is the most common type of familial hypocalciuric hypercalcemia and is caused by changes (also known as pathogenic variants or mutations) in the calcium-sensing receptor (CaSR) gene. The protein made from the calcium-sensing receptor (CaSR) gene, the calcium-sensing receptor (CaSR protein), monitors and regulates the level of calcium in the blood 82.
- Familial hypocalciuric hypercalcemia type 2 (FHH2) is caused by changes in the GNA11 gene,
- Familial hypocalciuric hypercalcemia type 3 (FHH3) is caused by changes in the AP2S1 gene.
All three types of familial hypocalciuric hypercalcemia (FHH) are inherited in an autosomal dominant manner. In rare cases, familial hypocalciuric hypercalcemia (FHH) may be caused when a person’s immune system mistakenly makes antibodies that attack the CaSR protein. The autoimmune form of familial hypocalciuric hypercalcemia (autoimmune FHH) is not known to be caused by changes in a specific gene 81.
Diagnosis of familial hypocalciuric hypercalcemia (FHH) is suspected by high levels of calcium in the blood (hypercalcemia), especially when there are no other symptoms present. Further blood and urine tests may be used to rule out other possible causes. Genetic testing can confirm the diagnosis of familial hypocalciuric hypercalcemia (FHH), except in rare autoimmune cases 81.
Treatment of familial hypocalciuric hypercalcemia (FHH) is typically considered unnecessary because most people with familial hypocalciuric hypercalcemia do not have symptoms. If pancreatitis occurs, removal of the parathyroid gland (parathyroidectomy) may be recommended 81.
Hypercalcemia of malignancy
Hypercalcemia of malignancy also known as cancer-related hypercalcemia occurs in 10% to 30% of patients with cancer (e.g. carcinoma of the breasts, ovaries, cervix, and esophagus, and tumors in of head or neck region) 83, 14, 1. Hypercalcemia of malignancy represents the common life-threatening metabolic disorder with an extremely bad prognosis 84, with approximately 50% mortality at 1 month and up to 75% at 3 months after diagnosis 1. Cancer patient quality of life is also impacted by calcium levels 85. Hypercalcemia of malignancy is often underdiagnosed and treated in less than 40% of hospitalized patients 15.
The mechanisms of cancer-related hypercalcemia, in order of frequency, are the production of parathyroid hormone-related protein (PTHrp), bone metastasis (local osteolytic hypercalcemia), excessive activation of extrarenal vitamin D, and ectopic PTH secretion 86, 84.
Parathyroid hormone-related protein (PTHrp) is structurally similar to PTH; it binds to the same PTH bone receptors stimulating the synthesis of receptor activator of nuclear factor-kappa B ligand (RANKL) and activating osteoclasts, thus releasing calcium into the circulation 5. Additionally, parathyroid hormone-related protein (PTHrp) increases renal tubular reabsorption of calcium. Differently from PTH, parathyroid hormone-related protein (PTHrp) does not increase renal vitamin D hydroxylation or calcium absorption from the intestine. Cancer sites most related to parathyroid hormone-related protein (PTHrp) production are the head, neck, esophagus, lung, kidney, breast, bladder, endometrium, ovaries, and intestines 5.
The second most frequent cause of malignancy-associated hypercalcemia is bone metastases, which represents approximately 20% of cases 1. Bone destruction by tumors or metastasis was previously believed to be the primary mechanism, but it is currently known that bone destruction is a secondary mechanism. Fundamental factors responsible for development of hypercalcemia in patients with bone metastases are cytokines such as interleukin (IL)-1, IL-3, IL-6, tumor necrosis factor alpha (TNF-α) and transforming growth factor beta (TGF-β), that stimulate osteoblasts to produce receptor activator of nuclear factor-kappa B ligand (RANKL), which activate osteoclasts differentiation and bone resorption together with inability to clear calcium through the kidney 87, 88, 5. That is why bisphosphonates and/or more effective monoclonal antibody to RANKL, inhibit lysis of bone regions adjacent to the tumor 87, 89, 90.
Additionally, some types of metastases, such as breast cancer metastases, can induce local parathyroid hormone-related protein (PTHrp) production. The cancers most frequently related to this type of mechanism are Hodgkin and non-Hodgkin lymphoma, breast cancer, squamous cell carcinoma of the lung, invasive transitional cell carcinoma of the urinary bladder, and multiple myeloma 88, 91. Less frequently, ectopic PTH production can occur in lung and ovarian cancers, and some types of tumors such as lymphomas and ovarian cancer can cause extrarenal vitamin D activation 88.
Hypercalcemia from excess calcium intake
Milk-alkali syndrome is almost always caused by large amounts of calcium supplements, usually in the form of calcium carbonate 92, 93. Calcium carbonate is a common calcium supplement. It is often taken to prevent or treat bone loss (osteoporosis). Calcium carbonate is also an ingredient found in antacids (such as Tums). A high level of vitamin D in the body, such as from taking vitamin D supplements, can worsen milk-alkali syndrome. Calcium deposits in the kidneys and in other tissues can occur in milk-alkali syndrome. Milk-alkali syndrome presents with a triad of hypercalcemia, metabolic alkalosis, and acute kidney failure associated with the ingestion of calcium-rich medications and absorbable alkali 94, 95, 96, 97. Milk-alkali syndrome is estimated to be the third most common cause of hypercalcemia 98.
Milk-alkali syndrome has been reported in a 64-year-old woman secondary to chewing approximately 30 pieces of nicotine-substitute gum daily for several years after she stopped smoking 95, 99. Each piece of gum was found to contain 94 mg of elemental calcium, giving her a daily calcium intake, through this source alone, of about 2820 mg. The patient was concurrently drinking large amounts of carbonated water. Her calcium level was17.7 mg/dL on admission and normalized after 3 days with hydration, IV bisphosphonate administration, and calcitonin 99. A 59-year-old man who presented with severe hypercalcemia on 2 occasions, subsequently admitted to ingesting a household cleanser containing a large amount of calcium carbonate. Hypercalcemia resolved after the patient discontinued ingesting the product 100. Similarly, a 41-year-old woman with a known major depressive disorder presented with hypercalcemia after intentionally ingesting 250 mL of liquid calcium fertilizer containing 237 g of calcium 101. Milk- alkali syndrome has also been reported in 3 patients from Taiwan after chewing betel nuts along with oyster shell powder that was used to neutralize the bitter taste of the betel nuts 102, 103. Two patients who attempted suicide with buffered aspirin tablets developed hypercalcemia, presumably because of the associated intake of calcium carbonate 104. Hypercalcemia has also been reported in a nurse after surreptitious ingestion of large amounts of calcium carbonate with a thiazide diuretic 105. Similarly, a woman with an eating disorder developed hypercalcemia requiring hospitalization on 3 occasions after consuming a massive amount of cheese with concurrent dehydration and metabolic alkalosis caused by vomiting and thiazide diuretic use 106.
Hypercalcemia has also been reported in other rare occasions secondary to excessive non-alkali calcium absorption. For example, an 87-year-old Chinese man with allopurinol-induced toxic epidermal necrolysis developed hypercalcemia after using a large quantity of calcium alginate dressing as part of his treatment 107. In a study evaluating complications of antibiotic-eluting absorbable calcium sulfate beads in 15 patients with periprosthetic joint infection, 3 patients developed hypercalcemia during the follow-up period 108. A 31-year-old man with history of pervasive developmental disorder presented with severe hypercalcemia (serum total calcium 20.7 mg/dL) and metabolic acidosis after intentionally ingesting 300 mL of commercial moisture absorber solution containing 300 g of calcium chloride 109. Similarly, a 35-year-old man developed hypercalcemia and gastric necrosis after ingesting 3 topical hot packs in an attempt to be removed from jail. The hot packs were given to him for back pain, and each contained 90% to 97% calcium chloride 110. Hypercalcemic crisis has also been reported in a young man with extensive burns caused by a heated calcium chloride solution 111.
Hypercalcemia caused by medications
Hypercalcemia is rarely caused by medications. Common medications such as thiazide diuretics, lithium, and excessive intake of vitamin D, vitamin A, or calcium can result in hypercalcemia 112. Rarely, other medications have been associated with hypercalcemia:
- Voriconazole/itraconazole used concomitantly with all-trans-retinoic acid 113, 114
- Foscarnet 115, 116
- Discontinuation of denosumab 117, 118, 119, 120
- Omeprazole in acute interstitial nephritis 121
- Theophylline toxicity 122
- Human growth hormone 123
- Parenteral nutrition 124
- Hepatitis B vaccination 125
- Manganese toxicity 126
A 24-year-old woman with acute promyelocytic leukemia treated with all-trans-retinoic acid (ATRA) developed hypercalcemia while concurrently on voriconazole 127. Hypercalcemia was attributed to the inhibition of cytochrome P450 function by voriconazole when used concomitantly with ATRA. Similarly, a 38-year-old man with acute promyelocytic leukemia developed hypercalcemia from the interaction of all-trans-retinoic acid (ATRA) and itraconazole treatment 114. Foscarnet can lead to hypocalcemia through its chelating effect, but in 3 case reports has been associated with hypercalcemia. A 4-month-old infant with severe combined immunodeficiency disease and 2 adult patients with AIDS all developed hypercalcemia during foscarnet therapy for cytomegalovirus infection 115, 116. There are 4 case reports of denosumab discontinuation leading to rebound hypercalcemia. Hypercalcemia with elevated markers of bone remodeling has been reported in all cases, including a 10-year-old boy being treated for giant cell tumor, 2 women being treated for oncologic indications, and a 67-year-old woman being treated for osteoporosis 117, 118, 119, 120. In 1 of the women receiving oncologic doses of denosumab, FGF23 was elevated during the time of hypercalcemia 117, 118, 119, 120.
Hypercalcemia in a 31-year-old postpartum patient with acute interstitial granulomatous nephritis was attributed to use of omeprazole 121. The 1,25-OH2-D levels were not reported; however, PTH was low and angiotensin-converting enzyme level was elevated. The patient’s serum calcium and renal function responded to discontinuation of omeprazole and treatment with glucocorticoids. In 60 patients with theophylline toxicity, 11 developed hypercalcemia with normal PTH levels suggesting impaired parathyroid gland sensitivity to ionized calcium 122. Hypercalcemia developed among patients in a surgical intensive care unit receiving high doses of human growth hormone, although other factors such as parenteral nutrition and immobilization may have also had a role 123. Long-term parenteral nutrition caused hypercalcemia in several patients; this effect was resolved in 1 small study by removal of vitamin D from nutritional solutions 124. Hypercalcemia, arthritis, and lytic bone lesions occurred shortly after the third dose of recombinant hepatitis B virus vaccine in a 44-year-old man 125. Bone biopsy demonstrated increased bone resorption and his clinical symptoms responded to antiresorptive therapies. A significant increase in serum calcium also has been reported in cases with manganese poisoning 126.
Hypercalcemia of unknown mechanisms
In some reported cases of hypercalcemia, the mechanisms are unclear, either because they were published before widespread availability of accurate and helpful assays (eg, PTH, PTHrP, vitamin D metabolites), or where the mechanism could not be defined despite measurements of these factors 112. Hypercalcemia has rarely been reported in patients with adrenal insufficiency 128, 129, 130, 131, 132, 131. For example, a 47-year-old woman with a history of unilateral adrenalectomy developed hypercalcemia after long-term use of licorice as laxative and stopping her corticosteroid replacement therapy. Administration of hydrocortisone and zoledronic acid was associated with improvement in her clinical picture and normalization of serum calcium 128. Hypercalcemia associated with adrenal crisis was also described in a patient with psoriasis after abrupt withdrawal of topical and oral glucocorticoids 133. Similarly, a 45-year-old man with PTH-independent hypercalcemia was found to have adrenal histoplasmosis complicated by adrenal insufficiency 129. Hypercalcemia has been reported in isolated ACTH deficiency 130, medroxyprogesterone acetate withdrawal 131, and glucocorticoid withdrawal after surgical intervention for Cushing syndrome 132. A 57-year-old woman developed hypercalcemia secondary to adrenal insufficiency in the setting of partial hypopituitarism and pyelonephritis 131.
Transient hypercalcemia has been observed in patients with acute renal failure secondary to rhabdomyolysis, particularly during the recovery phase of the disease. In all of these cases, serum PTH and 1,25-dihydroxyvitamin D (calcitriol) levels were low 134, 135, 136, 137. Hypercalcemia in this syndrome has been attributed to remobilization of the calcium from the damaged muscles into the extracellular space 134, 135. In a retrospective cohort study of 73 burn-injury intensive care unit admissions, 22 (30%) developed hypercalcemia. Acute renal failure was more frequent in hypercalcemic patients. Hydration and mobilization resulted in normalization of calcium in most cases 138.
Hypercalcemia in granulomatous diseases
Granulomatous diseases such as tuberculosis, leprosy (Hansen disease), cryptococcosis, histoplasmosis, sarcoidosis, paracoccidioidomycosis, and granulomatosis with polyangiitis can also cause hypercalcemia by producing 1,25-dihydroxyvitamin D (calcitriol), but the exact mechanism in patients with leprosy (Hansen disease) is unclear 112, 1. Macrophages in the granulomas produce 1-α-hydroxylase, which converts 25[OH]D into its active form 1,25[OH]2D, increasing intestinal calcium absorption 139.
A 78-year-old man with borderline tuberculoid leprosy and type 1 lepra reaction developed hypercalcemia associated with normal levels of PTH and 1,25-dihydroxyvitamin D (calcitriol) 140. Similarly, another patient with leprosy complicating rheumatoid arthritis was found to have hypercalcemia in association with low levels of 1,25-dihydroxyvitamin D (calcitriol). In this case, hypercalcemia responded to treatment with dapsone and prednisone 141. A 24-year-old man with AIDS developed hypercalcemia associated with cryptococcal meningitis. Workup in this patient was remarkable for low PTH, PTHrP, and 1,25-OH2D 142. Similarly, 2 patients with AIDS and diffuse cytomegalovirus infection were found to have hypercalcemia associated with a low PTH level 143. Hypercalcemia has also been reported in 2 cases of rheumatoid arthritis (RA). A 60-year-old woman with RA developed hypercalcemia in association with suppressed PTH and increased bone resorption on bone scan 144. Similarly, a boy with juvenile rheumatoid arthritis and persistent hypercalcemia was found to have serum activity that stimulated bone resorption in vitro and was neutralized by an interleukin-1 receptor antagonist 145. Hypercalcemia has been observed in a 40-year-old man with scabies 146. Biopsy of an axillary lymph node was remarkable for nonspecific inflammation but no granuloma. 1,25-dihydroxyvitamin D (calcitriol) was not measured in this case 146.
A 34-year-old woman with a history of hypoparathyroidism developed hypercalcemia associated with Nocardia-induced pericarditis. Hypercalcemia responded to successful treatment of the infection 147. Another young woman with bone pain was found to have focal osteolysis and hypercalcemia associated with normal PTH and PTHrP 148. Treatment with glucocorticoids improved the symptoms, but the patient subsequently died of severe hypercalcemia. Histologic evaluation of these lesions was remarkable for intense osteoclastic resorption, without malignant cells 148. In 10 patients with advanced liver disease, hypercalcemia was reported in association with mild azotemia and suppressed or low-normal PTH levels 149. Hypercalcemia has also been reported in a 35-year-old woman with type 1 Gaucher disease and acute pneumonia. This was attributed to activation of osteoclasts by proinflammatory cytokines 150. Hypercalcemia associated with lymphedema in patients with systemic lupus erythematosus (SLE) has also been described in the setting of normal PTHrP levels 151. It has been suggested that autoantibodies may be responsible for hypercalcemia via cross-reacting with an activating the PTH receptor 151. Finally, a 24-year-old man with diffuse musculoskeletal pain and lymphadenopathy was found to have hypercalcemia, hyperphosphatemia, and elevated serum alkaline phosphatase activity 152. Further evaluation revealed diffuse uptake of radionuclide on a bone scan, high urinary N-telopeptide excretion, and a serum interleukin-6 (IL-6) level that was 100 times higher than normal 152. Treatment with intravenous pamidronate resulted in symptomatic and biochemical improvement 152. Many of the cases in which no specific mechanism could be identified have features suggesting increase bone resorption as the underlying etiology, similar to the presumed mechanism in cases of hypercalcemia associated with immobilization 112. By categorizing these individuals whose mechanism is not known, it is evident that there are probably many different mechanisms in these various situations all leading to osteoclast activation but most likely via different provocative intermediaries 112.
Hypercalcemia associated with parathyroid hormone-related protein (PTHrp) secretion has been described mostly in malignant tumors and is known as a major cause of humeral hypercalcemia of malignancy 153, 154, 83. However, it rarely has been reported in the setting of benign diseases (e.g., uterine leiomyoma) 155. Three patients with pilomatrixoma, a cutaneous benign appendage tumor that commonly occurs on the face and extremities, were found to have PTHrP-mediated hypercalcemia 156, 157. Hypercalcemia and elevated parathyroid hormone-related protein (PTHrp) have been reported in benign intestinal 158 and uterine leiomyomas 155, 159, benign ovarian tumor 160, renal adenoma 161, and pheochromocytomas 162, 163. Surgical removal of the benign tumor normalized the serum calcium level in these cases.
A 67-year-old man with sarcoidosis was found to have hypercalcemia with elevated levels of parathyroid hormone-related protein (PTHrp) 164. Bone marrow granulomas stained positively for parathyroid hormone-related protein (PTHrp) and treatment with glucocorticoids normalized hypercalcemia and the parathyroid hormone-related protein (PTHrp) level 164. A patient with systemic lupus erythematosus (SLE) with multiorgan involvement and diffuse lymphadenopathy presented with hypercalcemia and a high circulating parathyroid hormone-related protein (PTHrp) concentration 165. Immunohistology of biopsied lymph nodes revealed parathyroid hormone-related protein (PTHrp) expression 165. Similarly, a 39-year-old woman with SLE developed parathyroid hormone-related protein (PTHrp)-mediated hypercalcemia accompanied by ectopic calcinosis in subcutaneous tissue of both hands. Interestingly, bone scan showed intense uptake throughout the lungs 166. Hypercalcemia with elevated levels of PTHrP has also been reported in a young woman with SLE presenting with pleural effusions and lymphedematous bilateral breast enlargement 167. A 63-year-old man with HIV-associated lymphadenopathy developed recurrent hypercalcemia and was found to have positive staining for parathyroid hormone-related protein (PTHrp) and its mRNA in a cervical lymph node 168.
Parathyroid hormone-related protein (PTHrp) is produced physiologically in fetal and maternal tissues but excessive production in the placenta and mammary glands can cause hypercalcemia 169, 170. Rarely, parathyroid hormone-related protein (PTHrp)-mediated hypercalcemia has been reported in the setting of pregnancy, delivery, and lactation.
A 28-year-old pregnant woman developed severe hypercalcemia on the first postpartum day with a markedly increased serum level of parathyroid hormone-related protein (PTHrp). The serum levels of calcium and parathyroid hormone-related protein (PTHrp) both normalized after administration of saline and pamidronate 66. Another young woman presented in the 15th week of pregnancy with massive enlargement of breasts and was found to have severe hypercalcemia, suppressed PTH, and an elevated level of parathyroid hormone-related protein (PTHrp) 171. Treatment with bromocriptine suppressed parathyroid hormone-related protein (PTHrp) and normalized serum calcium levels 171. A 25-year-old pregnant woman with massive mammary hyperplasia accompanied by severe hypercalcemia responded to bilateral mastectomy 172. Immunostaining of the breast tissue revealed parathyroid hormone-related protein (PTHrp) reactivity limited to myoepithelial cells, with none evident in normal breast tissue 173. Hypercalcemia has also been reported during 2 pregnancies of a patient with low PTH levels. In the second pregnancy, parathyroid hormone-related protein (PTHrp) level remained elevated for several months after delivery 174. Another patient with hypoparathyroidism developed hypercalcemia with elevated parathyroid hormone-related protein (PTHrp) while lactating. Hypercalcemia in this case responded to the withdrawal of calcium and calcitriol supplements, whereas parathyroid hormone-related protein (PTHrp) levels remained elevated 175. There is an additional case report of similar findings in 2 pregnant patients from 1969; however, no parathyroid hormone-related protein (PTHrp) measurements were done 176. Based on these reports, patients with hypoparathyroidism on pharmacologic doses of vitamin D appear to be at increased risk for hypercalcemia during pregnancy and lactation. Calcium should be monitored closely in these patients who may need a reduction in vitamin D intake until breast feeding is completed.
Hypercalcemia caused by ectopic PTH production
Malignancy-associated hypercalcemia with elevated PTH is rare, with few reports over the past decades 112. For example, A 25-year-old man with acute myeloblastic leukemia developed hypercalcemia associated with inappropriately elevated levels of PTH on 6 occasions in conjunction with relapses of his disease 177. The in vitro studies were remarkable for production of PTH by leukemic cells 177. Hypercalcemia caused by ectopic PTH has also been reported in 2 patients with hepatocellular carcinoma 178, 179. Elevated PTH was found in hepatic venous sampling and decreased after embolization of the tumor in the first report 178. Similarly, 2 patients with small cell lung cancer were found to have hypercalcemia associated with elevated PTH levels in the tumor extract and in vitro studies 180, 181. Bronchogenic lung cancer has also been reported to cause hypercalcemia in the setting of ectopic PTH secretion. Surgical resection of the tumor resulted in a reduction in serum calcium and PTH levels in this patient 182.
Ectopic PTH secretion by ovarian tumor cells has been reported in 3 cases. A 33-year-old woman with ovarian non-small cell neuroendocrine carcinoma was found to have severe hypercalcemia with elevated serum level of PTH 183. The poorly differentiated cells of the tumor stained positive for PTH 183. Similarly, a 74-year-old woman with persistent hypercalcemia after resection of 3.5 parathyroid glands, was found to have ovarian carcinoma 184. DNA amplification and rearrangement in the regulatory region of the PTH gene was demonstrated in tumor tissue 184. A young woman with small cell carcinoma of the ovary had a similar presentation 185. Serum calcium and PTH levels normalized after surgical resection of the tumor in all 3 cases.
Hypercalcemia associated with high PTH level has been reported in 3 patients with pancreatic neuroendocrine tumors. In 2 of these cases with liver metastases, chemotherapy resulted in a decrease in the pancreatic mass, PTH, and calcium levels 186, 187. In the first case, there was no family history suggestive of multiple endocrine neoplasia type 1 (MEN1) and neck imaging did not demonstrate any parathyroid abnormalities 186. PTH and serum calcium levels responded to treatment of the neuroendocrine tumor 186. In the second case, neck exploration did not identify a parathyroid adenoma; sestamibi scanning showed uptake in the liver at the site of metastases 187. Similarly, a 41-year-old man with pancreatic neuroendocrine tumor developed hypercalcemia later during his disease 188. A PTH level was not checked, but several tumor cells were found to be positive for PTH 188. At autopsy, the parathyroid glands were normal 188. A 73-year-old woman with a clinical and biochemical profile characteristic of primary hyperparathyroidism but normal sestamibi scan was found to have a neuroendocrine tumor in the neck 189. Surgical resection of the mass resulted in normalization of the PTH level within 10 minutes 189. Additionally, papillary thyroid cancer 190, squamous cell carcinoma of tonsil 191, pheochromocytomas 192, nasopharyngeal rhabdomyosarcoma 193, and thymoma 194 have all been associated with hypercalcemia in the setting of ectopic PTH secretion.
Hypercalcemia caused by PTH and PTHrp
There have been few case reports of hypercalcemia due to tumors producing both PTH and parathyroid hormone-related protein (PTHrp) 112. Although PTH and parathyroid hormone-related protein (PTHrp) interact with a common receptor, there is no cross-reactivity in their current assays. This makes it possible to clearly distinguish between PTH-mediated, parathyroid hormone-related protein (PTHrp)-mediated, and ultra-rare situations where both appear to be etiologic in hypercalcemia. A 30-year-old man with persistent hypercalcemia and normal level of PTH was found to have a substernal parathyroid adenoma 195. He underwent parathyroidectomy with pathology remarkable for oxyphil adenoma that stained positive for PTH and parathyroid hormone-related protein (PTHrp) 195. Similarly, a 73-year-old man with a history of primary hyperparathyroidism presented with recurrent hypercalcemia 6 years after removal of a parathyroid adenoma 91. He was found to have large retroperitoneal mass. Computed tomography-guided biopsy of the mass demonstrated metastatic, poorly differentiated transitional cell carcinoma immunopositive for PTH and PTHrp 91. A poorly differentiated neuroendocrine tumor of the pancreas has also been associated with hypercalcemia and elevated levels of PTH and parathyroid hormone-related protein (PTHrp) 196. Staining of the tumor cells was positive for PTH in this case 196. Similar findings have been reported in metastatic gastric cancer 197 and neuroectodermal tumor 198.
Hypercalcemia prevention
Most causes of hypercalcemia cannot be prevented. Women over age 50 should see their health care provider regularly and have their blood calcium level checked if they have symptoms of hypercalcemia.
Talk to your health care provider about the correct dose if you are taking calcium and vitamin D supplements.
If you and your health care provider have chosen to monitor, rather than treat, your primary hyperparathyroidism, the following suggestions can help prevent complications:
- Monitor how much calcium and vitamin D you get in your diet. Restricting how much calcium you eat or drink is not recommended for people with hyperparathyroidism.
- The daily recommended amount of calcium for adults ages 19 to 50 and men ages 51 to 70 is 1,000 milligrams (mg) of calcium a day. That calcium recommendation increases to 1,200 mg a day for women age 51 and older and men age 71 and older.
- The daily recommended amount of vitamin D is 600 international units (IUs) of vitamin D a day for people ages 1 to 70 and 800 IUs a day for adults age 71 and older. Talk to your provider about dietary guidelines that are right for you.
- Drink plenty of fluids. Drink enough fluids, mostly water, to produce nearly clear urine to lessen the risk of kidney stones.
- Exercise regularly. Regular exercise, including strength training, helps maintain strong bones. Talk to your provider about what type of exercise program is best for you.
- Don’t smoke. Smoking may increase bone loss as well as increase your risk of several serious health problems. Talk to your provider about the best ways to quit.
- Avoid calcium-raising medications. Certain medications, including some diuretics and lithium, can raise calcium levels. If you take such medications, ask your provider whether another medication may be appropriate for you.
Calcium
Intake recommendations for calcium and other nutrients are provided in the Dietary Reference Intakes (DRIs) developed by the Food and Nutrition Board at the Institute of Medicine of the National Academies 199. They are listed in Table 2 in milligrams (mg) per day.
Calcium is found in many foods. It is important to get plenty of calcium in the foods you eat. You can get recommended amounts of calcium by eating a variety of foods, including the following 40:
- Dairy products such as milk, cheese, and yogurt are the main food sources of calcium for most people in the United States.
- Fish with soft bones that you eat, such as canned sardines and salmon.
- Certain vegetables, such as kale, broccoli, and Chinese cabbage (bok choi) also contain calcium.
- Calcium is added to some breakfast cereals and beverages, including many fruit juices and milk substitutes such as soy and almond beverages, as well as some brands of tofu and ready-to-eat cereals. To find out whether these foods have calcium added, check the product labels.
- Most grains (such as breads, pastas, and unfortified cereals) do not have high amounts of calcium. However, because people eat them often, what they contribute adds up.
The U.S. Department of Agriculture’s (USDA’s) FoodData Central website (https://fdc.nal.usda.gov) lists the nutrient content of many foods with Calcium arranged by nutrient content (https://ods.od.nih.gov/pubs/usdandb/Calcium-Content.pdf) and by food name (https://ods.od.nih.gov/pubs/usdandb/Calcium-Food.pdf).
People should get most of their nutrients from food, advises the federal government’s Dietary Guidelines for Americans 200. Foods contain vitamins, minerals, dietary fiber and other substances that benefit health. In some cases, fortified foods and dietary supplements may provide nutrients that otherwise may be consumed in less-than-recommended amounts. For more information about building a healthy diet, refer to the Dietary Guidelines for Americans (https://www.dietaryguidelines.gov) and the U.S. Department of Agriculture’s MyPlate (https://www.myplate.gov).
In its food guidance system, the U.S. Department of Agriculture’s MyPlate (https://www.myplate.gov) recommends that persons aged 9 years and older eat 3 cups of foods from the milk group per day 201. A cup is equal to 1 cup (8 ounces) of milk, 1 cup of yogurt, 1.5 ounces of natural cheese (such as Cheddar), or 2 ounces of processed cheese (such as American).
Milk, yogurt, and cheese are rich natural sources of calcium and are the major food contributors of this nutrient to people in the United States 199. Nondairy sources include vegetables, such as Chinese cabbage, kale, and broccoli. Spinach provides calcium, but its bioavailability is poor. Most grains do not have high amounts of calcium unless they are fortified; however, they contribute calcium to the diet because they contain small amounts of calcium and people consume them frequently. Foods fortified with calcium include many fruit juices and drinks, tofu, and cereals. Selected food sources of calcium are listed in Table 2.
Calcium absorption varies by type of food. The absorption of calcium from dairy products and fortified foods is about 30% 22. Certain compounds in plants (e.g., oxalic acid, phytic acid) can decrease calcium absorption by forming indigestible salts with calcium, decreasing its absorption 43. As a result, absorption of calcium is only 5% for spinach, whereas it is much higher, at 27%, for milk 43. In addition to spinach, foods with high levels of oxalic acid include collard greens, sweet potatoes, rhubarb, and beans 22. The bioavailability of calcium from other plants that do not contain these compounds—including broccoli, kale, and cabbage—is similar to that of milk, although the amount of calcium per serving is much lower 43. When people eat many different types of foods, these interactions with oxalic or phytic acid probably have little or no nutritional consequence. Net absorption of dietary calcium is also reduced to a small extent by intakes of caffeine and phosphorus and to a greater extent by low status of vitamin D 202, 203, 204.
Table 2. Recommended Dietary Allowances (RDAs) for Calcium
| Life Stage | Recommended Amount |
|---|---|
| Birth to 6 months | 200 mg |
| Infants 7–12 months | 260 mg |
| Children 1–3 years | 700 mg |
| Children 4–8 years | 1,000 mg |
| Children 9–13 years | 1,300 mg |
| Teens 14–18 years | 1,300 mg |
| Adults 19–50 years | 1,000 mg |
| Adult men 51–70 years | 1,000 mg |
| Adult women 51–70 years | 1,200 mg |
| Adults 71 years and older | 1,200 mg |
| Pregnant and breastfeeding teens | 1,300 mg |
| Pregnant and breastfeeding adults | 1,000 mg |
Footnote: *Adequate Intake (intake at this level is assumed to ensure nutritional adequacy; established when evidence is insufficient to develop an Recommended Dietary Allowance (RDA).
[Source 40 ]Vitamin D
Vitamin D improves your body’s ability to absorb calcium and improves bone health in other ways. The amount of vitamin D you need each day depends on your age. Average daily recommended amounts from the Food and Nutrition Board (a national group of experts) for different ages are listed below in International Units (IU). People can get some of their vitamin D from sunlight, but this might not be a good source if you live in a high latitude, if you’re housebound, or if you regularly use sunscreen or avoid the sun because of the risk of skin cancer.
Dietary sources of vitamin D include cod liver oil, trout and salmon. Many types of milk and cereal have been fortified with vitamin D.
Most people need at least 600 international units (IU) of vitamin D a day. That recommendation increases to 800 IU a day after age 70.
People without other sources of vitamin D and especially with limited sun exposure might need a supplement. Most multivitamin products contain between 600 and 800 IU of vitamin D. Up to 4,000 IU of vitamin D a day is safe for most people.
Up to 42% of adults have vitamin D deficiency, which results in compensatory, mild parathyroid hormone (PTH) elevation 80. Calcium and phosphorus levels can be normal or at the low end of normal ranges. A trial of vitamin D supplementation is both diagnostic and therapeutic and does not have any adverse effects in the setting of primary hyperparathyroidism 205
Table 3. Recommended Dietary Allowances (RDAs) for Vitamin D
| Life Stage | Recommended Amount |
|---|---|
| Birth to 12 months | 400 IU (10 mcg) |
| Children 1–13 years | 600 IU (15 mcg) |
| Teens 14–18 years | 600 IU (15 mcg) |
| Adults 19–70 years | 600 IU (15 mcg) |
| Adults 71 years and older | 800 IU (20 mcg) |
| Pregnant and breastfeeding women | 600 IU (15 mcg) |
Footnote: The amount of vitamin D contained in supplements is sometimes expressed in international units (IU) where 40 IU is equal to one microgram (1 mcg) of vitamin D.
[Source 206 ]Hypercalcemia signs and symptoms
Symptoms due to high calcium level may vary, depending on the cause, the calcium levels, and how long the problem has been present 1. You might not have signs or symptoms if your hypercalcemia is mild. Onset time is also important in the development of symptoms; acute onset is frequently associated with more symptoms than chronic onset, which allows the body to adapt to the new metabolic condition 86. Hypercalcemia is most often diagnosed at an early stage using routine blood tests. More-severe cases produce signs and symptoms related to the parts of your body affected by the high calcium levels in your blood. Usually, calcium values between 10.5 and 12 mg/dL rarely cause symptoms, values greater than 14 mg/dL are associated with more frequent and severe symptoms, and calcium values higher than 15 mg/dL (> 3.74 mmol/L) increase the risk of cardiac arrest 207.
Most patients with hypercalcemia are asymptomatic and diagnosed during routine tests 1. Only a small percentage of hypercalcemia patients present with exuberant and severe symptoms, which involve neurological, muscular, gastrointestinal, renal, and cardiovascular systems 1.
Hypercalcemia symptoms may include:
- Kidneys. Excess calcium in your blood means your kidneys have to work harder to filter it. This can cause excessive thirst (polydipsia), frequent urination (polyuria), dehydration, kidney stone (nephrolithiasis) and acute kidney failure.
- Digestive system. Hypercalcemia can cause stomach upset, nausea, vomiting, poor appetite and constipation.
- Bones. In most cases, the excess calcium in your blood was leached from your bones, which weakens them. This can cause bone pain, muscle weakness and depression. Bone pain and long-term spine changes, if the bones have become thinner or weaker.
- Muscle weakness or twitches
- Brain. Hypercalcemia can interfere with the way your brain works, resulting in confusion, somnolence, anxiety, cognitive deficits, behavior changes, lethargy and fatigue. It can also cause depression and coma.
- Heart. Rarely, severe hypercalcemia can interfere with your heart function, causing palpitations and fainting, indications of cardiac arrhythmia, and other heart problems (e.g., bradycardia, tachycardia, atrioventricular blockade, prolonged PR interval, widened QRS and shortened QT interval).
Acute hypercalcemia is usually marked by dehydration, which occurs through various mechanisms 1. Hypercalcemia causes renal afferent arteriole vasoconstriction, which decreases renal blood flow and glomerular filtration rate (GFR), further reducing the ability of the kidneys to excrete the excess calcium 208, 209, 210., 83 Also, hypercalcemia affects tubular function by inhibiting the sodium-chloride transporter and increasing sodium and chloride loss, thus leading to volume depletion 210. Additionally, hypercalcemia downregulates aquaporin 2 water channels in the collecting ducts, which contributes to polyuria 208. The volume contraction causes additional renal sodium retention by activating the renin-angiotensin-aldosterone system, and leads to reduced urinary calcium excretion, by stimulating its reabsorption in the proximal renal tubule, since the transport of these 2 ions is linked 209.
Chronic hypercalcemia presents with less exuberant symptoms. Kidney stone (nephrolithiasis), osteopenia, and osteoporosis are common clinical findings. Patients with primary hyperparathyroidism have lower bone mineral density than individuals of the same sex and age without hyperparathyroidism. Approximately 15% of hyperparathyroidism patients have osteopenia in the lumbar spine. Depression and memory loss are also associated with chronic hypercalcemia, but the causal link between these conditions and parathyroid disease is uncertain 211.
Hypercalcemia complications
Gastrointestinal
- Pancreatitis
- Peptic ulcer disease
Kidney
- Calcium deposits in the kidney (nephrocalcinosis)
- Dehydration
- High blood pressure
- Kidney failure. Severe hypercalcemia can damage your kidneys, limiting their ability to cleanse the blood and eliminate fluid.
- Kidney stones. If your urine contains too much calcium, crystals may form in your kidneys. Over time, the crystals may combine to form kidney stones. Passing a stone can be extremely painful.
Psychological
- Depression
- Difficulty concentrating or thinking
- Nervous system problems. Severe hypercalcemia can lead to confusion, dementia and coma, which can be fatal.
Skeletal
- Bone cysts
- Fractures
- Osteoporosis. If your bones continue to release calcium into your blood, you can develop the bone-thinning disease osteoporosis, which could lead to bone fractures, spinal column curvature and loss of height.
Heart
- Abnormal heart rhythm (arrhythmia). Hypercalcemia can affect the electrical impulses that regulate your heartbeat, causing your heart to beat irregularly.
These complications of long-term hypercalcemia are uncommon today.
Hypercalcemia diagnosis
Because hypercalcemia can cause few, if any, signs or symptoms, you might not know you have the disorder until routine blood tests show a high level of blood calcium. Blood tests also can reveal whether your parathyroid hormone level is high, indicating that you have hyperparathyroidism.
In most cases, hypercalcemia or elevated calcium is found by blood tests ordered for other reasons. For example, routine blood work or testing to figure out the cause of symptoms of another condition.
To determine if your hypercalcemia is caused by an underlying problem, such as cancer or sarcoidosis, your doctor might recommend imaging tests of your bones or lungs.
Your doctor may diagnose hyperparathyroidism by ordering:
- Blood tests. If blood test results show that you have high calcium levels in your blood, your health care provider will likely repeat the test. This repeated test can confirm the results after you haven’t eaten for a period of time. Many conditions can raise calcium levels. But your health care provider can diagnose hyperparathyroidism if blood tests show you also have high levels of parathyroid hormone (PTH).
- A measure of calcium levels in urine which can guide in the cause of the elevated blood calcium. Urinary calcium and urinary creatinine levels can be helpful in calculating calcium excretion by the formula:
- Calcium to creatinine clearance ratio [CaCl/CrCl] = (Urine Calcium x Serum Creatinine)/(Serum Calcium x Urine Creatinine), where Urine Calcium is mg/24 hr, Serum Calcium is mg/dL, Urine Creatinine is mg/24 hr, and Serum Creatinine is mg/dL; this is useful in differentiating primary hyperparathyroidism from familial hypocalciuric hypercalcemia (FHH), its main differential diagnosis. A calcium to creatinine clearance ratio <0.01 suggests familial hypocalciuric hypercalcemia (FHH) and a result of calcium to creatinine clearance ratio >0.01 suggests primary hyperparathyroidism 7.
- A blood test for vitamin D to see if a low vitamin D level is affecting primary hyperparathyroidism
- Imaging tests such as ultrasound or CT scan to look for kidney stones
- A bone mineral density test called a DEXA scan—a type of X-ray that checks bone strength. In some cases, patients also receive testing for genetic forms of the condition.
Figure 4. Hypercalcemia diagnostic algorithm
Abbreviations: UrCa = urinary calcium (mg/24 h); SerCa = serum calcium (mg/dL); UrCr = urinary creatinine (mg/24 h); SerCr = serum creatinine (mg/dL); PTH = parathyroid hormone; 25[OH]D = 25-hydroxyvitamin D; 1,25[OH]2D = 1,25-dihydroxyvitamin D.
[Source 1 ]Hypercalcemia workup
An accurate diagnosis is needed in hypercalcemia. Patients with kidney stones should have tests to evaluate for hypercalcemia.
- Serum calcium
- Serum PTH
- Serum PTHrP (PTH-related protein)
- Serum vitamin D level
- Urine calcium
- Electrocardiography (ECG)
Vitamin D blood test
Doctors test for vitamin D levels because low levels are common in people with primary hyperparathyroidism. In patients with primary hyperparathyroidism, the low vitamin D level can further stimulate the parathyroid glands to make even more parathyroid hormone (PTH). Also, a very low vitamin D level may cause a secondary form of hyperparathyroidism, which resolves when vitamin D levels are returned to normal.
In the blood, a form of vitamin D known as 25-hydroxyvitamin D [25(OH)D] or “calcidiol” is measured in either nanomoles per liter (nmol/L) or nanograms per milliliter (ng/mL). One nmol/L is equal to 0.4 ng/mL. So, for example, 50 nmol/L is the same as 20 ng/mL.
- Levels of 50 nmol/L (20 ng/mL) or above are adequate for most people for bone and overall health.
- Levels below 30 nmol/L (12 ng/mL) are too low and might weaken your bones and affect your health.
- Levels above 125 nmol/L (50 ng/mL) are too high and might cause health problems.
In patients with primary hyperparathyroidism, the low vitamin D level can further stimulate the parathyroid glands to make even more parathyroid hormone (PTH). Also, a very low vitamin D level may cause a secondary form of hyperparathyroidism, which resolves when vitamin D levels are returned to normal.
- A low 25-hydroxyvitamin D [25(OH)D] or “calcidiol” level suggests vitamin D deficiency from poor nutritional intake, lack of sunlight, or malabsorption.
- Low levels of 1,25-dihydroxyvitamin D [1,25(OH)2D] or “calcitriol” in association with high PTH suggest ineffective PTH from a lack of vitamin D, as observed in patients with chronic kidney failure, vitamin D–dependent rickets type 1 (VDDR-I), and pseudohypoparathyroidism 212.
Assessing vitamin D status by measuring serum 25-hydroxyvitamin D [25(OH)D] or “calcidiol” concentrations is complicated by the considerable variability of the available assays (the two most common ones involve antibodies or chromatography) used by laboratories that conduct the analyses 213, 214. As a result, a finding can be falsely low or falsely high, depending on the assay used and the laboratory. The international Vitamin D Standardization Program has developed procedures for standardizing the laboratory measurement of 25-hydroxyvitamin D [25(OH)D] or “calcidiol” to improve clinical and public health practice 215, 216, 217.
In contrast to 25-hydroxyvitamin D [25(OH)D], circulating 1,25-dihydroxyvitamin D [1,25(OH)2D] or “calcitriol” is generally not a good indicator of vitamin D status because it has a short half-life measured in hours, and serum levels are tightly regulated by parathyroid hormone, calcium, and phosphate 22. Levels of 1,25-dihydroxyvitamin D [1,25(OH)2D] do not typically decrease until vitamin D deficiency is severe 218.
Urinary cyclic adenosine monophosphate (cAMP) may help differentiate hypoparathyroidism from pseudohypoparathyroidism types 1 and 2. Urinary cAMP levels are generally elevated in hypoparathyroidism 212.
Electrocardiography
On electrocardiography (ECG), characteristic changes in patients with hypercalcemia include shortening of the QT interval. ECG changes in patients with very high serum calcium levels include the following 219, 220, 221:
- Slight prolongation of the PR and QRS intervals
- T wave flattening or inversion
- A J wave at the end of the QRS complex
- ST elevation mimicking acute myocardial infarction (heart attack)
Additional tests
After diagnosing primary hyperparathyroidism, your health care provider will likely order more tests. These can rule out possible conditions causing hyperparathyroidism, identify possible complications and determine the severity of the condition. These tests include:
- Bone mineral density test. This test is done to see if you have developed osteoporosis. The most common test to measure bone mineral density is dual-energy X-ray absorptiometry (DEXA) also called a DXA scan. This test uses low-dose x-rays to measure how many grams of calcium and other bone minerals are packed into a bone segment. During the test, you will lie on a padded table while a technician moves the scanner over your body. A bone expert or radiologist will read the scan. The DEXA provides a T-score that is used for diagnostic purposes. Clinically, a T-score of -2.5 standard deviations (SD) or below gives a diagnosis of osteoporosis in postmenopausal women and men aged ≥50 years (Table 4).
- Urine test. A 24-hour collection of urine can provide information on how well your kidneys work and how much calcium is passed in your urine. This test may help your doctor determine the severity of hyperparathyroidism or diagnose a kidney disorder causing hyperparathyroidism. If a very low calcium level is found in the urine, this may mean it’s a condition that doesn’t need treatment.
- Imaging tests of kidneys. Your doctor may order an X-ray or other imaging tests of your abdomen to determine if you have kidney stones or other kidney problems.
Bone mineral density test
Bone mineral density (BMD) at specific locations of your hip and spine is measured by dual-energy X-ray absorptiometry, also called a DXA or DEXA scan, which uses low-dose x-rays to measure bone density. During the DEXA test, you will lie on a padded table while a technician moves the scanner over your body. A bone expert or radiologist will read the scan.
The DEXA provides a T-score that is used for diagnostic purposes. Clinically, a T-score of -2.5 standard deviations (SD) or below gives a diagnosis of osteoporosis in postmenopausal women and men aged ≥50 years (Table 4).
Table 4. Bone mineral density DEXA test T-score cutoffs
| Normal bone mass | T-score equal to or above -1 standard deviation (SD) |
| Low bone mass (osteopenia) | T-score between -1 SD and -2.5 SD |
| Osteoporosis | T-score equal to or below -2.5 SD |
Risk of Fracture
The burden of osteoporosis lies primarily in the fractures that arise. Osteoporotic fractures are often precipitated by falls from standing height, although vertebral fractures can occur in the absence of a fall during daily, routine activities. About 70% of osteoporotic fractures affect women. Worldwide, 1 in 3 women and 1 in 5 men aged >50 years will suffer from a fracture.
The risk of osteoporotic fracture is influenced by bone mass, bone structure (microarchitecture, geometry), and propensity to fall (balance, mobility, muscular strength). The Fracture Risk Assessment Tool (https://frax.shef.ac.uk/FRAX/index.aspx) is an online tool that helps estimate your probability of fracture over the next 10 years, taking bone mineral density (BMD) measurement and additional risk factors into account (Table 5).
Table 5. 10-year Fracture Risk Probability
| Age (between 40 and 90 years) |
| Sex |
| Weight and height |
| Previous osteoporotic fracture |
| Parental history of hip fracture |
| Current tobacco smoking |
| Use of glucocorticoids (>3 months of doses equivalent to 5 mg/day of prednisolone) |
| Confirmed diagnosis of rheumatoid arthritis |
| Conditions associated with secondary osteoporosis (type 1 diabetes mellitus, osteogenesis imperfecta, untreated long-standing hyperparathyroidism, hypogonadism or premature menopause [<45 years], chronic malnutrition, or malabsorption and chronic liver disease) |
| Alcohol intake: ≥3 units (drinks)/day |
| Low femoral neck BMD |
Kidney imaging tests
Doctors may use one of the following imaging tests to look for kidney stones.
- Ultrasound. Ultrasound uses a device called a transducer that bounces safe, painless sound waves off organs to create an image of their structure. A specially trained technician does the procedure. A radiologist reads the images, which can show kidney stones.
- Abdominal x-ray. An abdominal x-ray is a picture of the abdomen that uses low levels of radiation and is recorded on film or on a computer. During an abdominal x-ray, you lie on a table or stand up. A technician positions the x-ray machine close to your abdomen and asks you to hold your breath so the picture won’t be blurry. A radiologist reads the x-ray, which can show the location of kidney stones in the urinary tract. Not all stones are visible on an abdominal x-ray.
- Computed tomography (CT) scans. CT scans use a combination of x-rays and computer technology to create images of your urinary tract. CT scans sometimes use a contrast medium—a dye or other substance that makes structures inside your body easier to see. Contrast medium isn’t usually needed to see kidney stones. For the scan, you’ll lie on a table that slides into a tunnel-shaped machine that takes the x-rays. A radiologist reads the images, which can show the size and location of a kidney stone.
Imaging tests before surgery
If your health care provider recommends surgery, one of these imaging tests may be used to locate the parathyroid gland or glands that are causing problems:
- Sestamibi parathyroid scan. Sestamibi is a radioactive compound that is absorbed by overactive parathyroid glands. It can be detected by a scanner that detects radioactivity. A healthy thyroid gland also absorbs sestamibi. To keep the thyroid absorption from blocking the view of the absorption in a parathyroid tumor (adenoma), you’re also given radioactive iodine. This is only absorbed by the thyroid. Using this process, the thyroid image is digitally removed so it can’t be seen.
- Computerized tomography (CT) scanning may be combined with the sestamibi scan to improve detection of any problems with the parathyroid glands.
- Ultrasound. Ultrasound uses sound waves to create images of your parathyroid glands and surrounding tissue. A small device held against your skin (transducer) sends out high-pitched sound waves and records the sound wave echoes as they reflect off internal structures. A computer converts the echoes into images on a monitor.
Hypercalcemia treatment
Treatment is aimed at the cause of hypercalcemia whenever possible. People with primary hyperparathyroidism (PHPT) may need surgery to remove the abnormal parathyroid gland. This will cure the hypercalcemia.
People with mild hypercalcemia may be able to be monitored closely over time.
Extremely high calcium levels can be a medical emergency. You might need hospitalization for treatment with IV fluids and diuretics to promptly lower the calcium level to prevent heart rhythm problems or damage to the nervous system.
Severe hypercalcemia that causes symptoms and requires a hospital stay may be treated with the following:
- Fluids through a vein: Hydration with IV fluids is the most important therapy in hypercalcemia treatment. The fluid therapy should replace sodium and water deficits and stimulate a sodium-linked calcium diuresis in the proximal renal tubule, as these 2 ions move in parallel at this part of the nephron 209, 222, 223. Heller et al 222 showed that saline alone reduces calcium by an average of 2.4 mg/dL (0.6 mmol/L); this may be enough to correct hypercalcemia in mild cases. In this trial, all patients displayed clinical improvement after venous hydration 209, 222. The type of fluid should always be individualized according to electrolytes and the presence of acid-base disorders 224. Experts usually administer 0.9% saline intravenously, starting with 1 to 2 L, followed by an infusion of 200 to 300 mL/h 1. The hydration should ensure a diuresis of 100 to 150 mL/h, a urinary flow that promotes effective calciuresis 86, 139. If the diuresis is unsatisfactory, hydration should be adjusted, always aiming at the above-mentioned diuresis.
- Drugs that stop bone breakdown and absorption by the body (bisphosphonates). The second cornerstone of hypercalcemia treatment is bisphosphonates 1. Saline with bisphosphonates is the first-line treatment, with level-one evidence for hypercalcemia 225. Most trials with bisphosphonates have only included patients with calcium levels above 12 mg/dL (>2.99 mmol/L); for this reason, hydration only is initially recommended in mild hypercalcemia. In moderate or severe hypercalcemia, or in very symptomatic patients, bisphosphonates should be added to intravenous hydration. The 2 options approved for hypercalcemia treatment are pamidronate and zoledronic acid 1. These medications have a high affinity for hydroxyapatite and are quickly deposited in the bone after administration. Bisphosphonates are internalized by osteoclasts, blocking structural proteins and inducing apoptosis of these cells. As a result, bone reabsorption is decreased, and calcium levels are consequently reduced 5, 139.
- Pamidronate was approved by the Food and Drug Administration (FDA) for clinical use in 1991. In a systematic review of bisphosphonate use in malignancy-related hypercalcemia, Saunders et al 226 showed that bisphosphonates are highly effective, with over 70% of patients reaching normocalcemia. Small studies showed no differences between 30, 60, or 90 mg pamidronate 227, 228, but a large, double-blind study suggested increased efficacy at higher doses 229. Although several authors recommend lower doses for moderate hypercalcemia, evidence suggests that higher bisphosphonate doses should be considered, regardless of initial calcium value 226. The usual pamidronate dose is between 60 and 90 mg intravenously, and infusion should be given in 2 to 4 hours. In patients with GFR <30 mL/min, extended infusion time and reduced dose should be considered 230, but there are not enough clinical trials to support this recommendation. The average time to reach normocalcemia is 4 days and the effect of the medication lasts around 18 days. The dose of zoledronic acid is 4 to 8 mg intravenously, infused over 15 min. In patients with GRF between 30 and 60 mL/min, the dose should be reduced; zoledronic acid is not recommended for patients with a GFR lower than 30 mL/min 230. The effect on calcemia can be seen in less than 3 days and it lasts approximately 32 days 5, 139, 231.
- Zoledronic acid is more potent than pamidronate in normalizing calcium levels. Major et al 232 conducted 2 concurrent randomized controlled trials that compared 4 mg and 8 mg of zoledronic acid with 90 mg of pamidronate; both zoledronic acid doses were more effective than pamidronate, with 88.4%, 86.7% and 69.7% of patients achieving normal serum calcium concentration after 10 days, respectively 232. However, unlike zoledronic acid, pamidronate can be used in association with calcitonin, and promotes a faster response 5, 84, 233.
- Calcitonin (Miacalcin). This hormone from salmon controls calcium levels in the blood. Mild nausea might be a side effect. Calcitonin has been approved by the FDA for treating osteoporosis since 1975 and has more recently become part of the therapeutic arsenal for hypercalcemia when a rapid decrease in calcium levels is needed. The action mechanism consists of decreasing osteoclast activity and renal calcium reabsorption, reducing calcium levels. In some countries, calcitonin is not widely available. Calcitonin has the advantage of a faster effect (12 to 24 h), but it downregulates calcitonin receptors in the bone and kidneys, inducing tachyphylaxis in 48 hours 230; therefore, its use in combination with pamidronate is controversial 4. Calcitonin is administered intramuscularly or subcutaneously at a dose of 4 U/kg every 12 hours, or even every 6 hours if the response is not satisfactory. The effect of isolated calcitonin in hypercalcemia treatment has been poorly studied 5, 231.
- Denosumab (Prolia, Xgeva). This drug is often used to treat people with cancer-caused hypercalcemia who don’t respond well to bisphosphonates 234, 235. Denosumab was approved by the FDA in 2010, initially for treating osteoporosis and skeletal-related events in patients with bone metastases; only in 2018 was denosumab approved for malignancy-related hypercalcemia that is unresponsive to bisphosphonates. Denosumab is a monoclonal antibody against RANKL. Blockage of RANKL prevents maturation and the function and survival of osteoclasts and therefore decreases bone resorption 236. There have been some case reports since 2012 on the effectiveness of denosumab in hypercalcemia-related malignancy 237, 238, 239, 240, 241. In a randomized, double-blind, clinical trial comparing denosumab and zoledronic acid in patients with advanced cancer and bone metastases or with multiple myeloma, Diel et al 242 showed that denosumab is more effective than zoledronic acid in delaying and preventing hypercalcemia 242. The recommended dose is a subcutaneous 120 mg denosumab weekly for the first month and then once every 4 weeks 243, 244, 245, 246, 236, 247.
- Glucocorticoids (steroids). The use of corticosteroids is limited to the treatment of some cases of malignancy-related hypercalcemia, especially in multiple myeloma, lymphomas, and breast cancer. Corticosteroids decrease the production of inflammatory mediators by the bones and therefore decrease osteoclast activity. They can also be used in vitamin D-mediated hypercalcemia because they reduce 1-α-hydroxylase activity. The recommended dose is 200 to 300 mg hydrocortisone daily or 40 to 60 mg prednisone per day for 3 to 5 days. It should be associated with another therapeutic strategy and interrupted if there is an unsatisfactory response in 10 days 5, 231.
- Calcimimetics. This type of drug can help control overactive parathyroid glands. Cinacalcet (Sensipar) has been approved for managing hypercalcemia.
- Dialysis. Hemodialysis is an alternative for patients refractory to standard management, or with severe kidney injury or congestive heart failure, in whom hydration cannot be safely performed, or if urgent reduction in calcium is needed, such as in patients with severe neurological symptoms 88, 248, 249, 250.
- Diuretic medicine, such as furosemide. In the past, furosemide was a standard of care for hypercalcemia treatment. In a review, LeGrand et al 225 found no studies since 1983 investigating furosemide dose or efficiency in hypercalcemia management. The effect of furosemide in calcium diuresis is limited and may aggravate dehydration and electrolytic disturbances; its use should therefore be limited to patients with volume overload 88.
When you were in the hospital, you were given fluids through an IV and drugs to help lower the calcium level in your blood. If you have cancer, you may have had treatment for that, as well. If your hypercalcemia is caused by a parathyroid gland problem, you may have had surgery to remove that parathyroid gland.
Surgery
Problems associated with overactive parathyroid glands often can be cured by surgery to remove the tissue that’s causing the problem. In many cases, only one of a person’s four parathyroid glands is affected. A special scanning test uses an injection of a small dose of radioactive material to pinpoint the gland or glands that aren’t working properly.
Doctors recommend surgery for people with clear symptoms or complications of primary hyperparathyroidism. In people without symptoms, doctors follow the guidelines to identify who might benefit from parathyroid surgery 73. Surgery can lead to improved bone density and can lower the chance of forming kidney stones.
You might be a candidate for surgery (parathyroidectomy) if you meet any of these guidelines 74:
- A serum (albumin-corrected) calcium level greater than 1 mg/dL above normal
- Bone density by DXA T-score equal to or below -2.5 SD at any site (lumbar spine, hip, or forearm) indicating osteoporosis
- History of kidney stones or evidence of kidney stones (asymptomatic nephrolithiasis on imaging) or calcifications in the kidney by imaging (e.g., X-ray, ultrasound, CT scan). Evidence for stone risk by 24-hour urine with excessive calcium or hypercalciuria [defined as a 24-hour urine calcium level greater than 400 mg/dL] and other stone risk factors e.g., impaired renal function [defined as a glomerular filtration rate less than 60 mL/minute]).
- A fracture resulting from relatively little force, such as a fall from a standing or sitting position (a fragility fracture)
- Age younger than age 50 years
Doctors most often recommend parathyroid surgery, particularly if their patients meet one or more of the guidelines noted above.
When performed by experienced surgeons, surgery almost always cures primary hyperparathyroidism.
Surgeons often use imaging tests before surgery to locate the overactive gland or glands to be removed. The tests used most often are sestamibi, ultrasound, and CT scans. In a sestamibi scan, you will get an injection, or shot, of a small amount of radioactive dye in your vein. The overactive parathyroid gland or glands then absorb the dye. The surgeon can see where the dye has been absorbed by using a special camera.
Surgeons use two main types of operations to remove the overactive gland or glands:
- Minimally invasive parathyroidectomy. Also called focused parathyroidectomy, surgeons use this type of surgery when they think only one of the parathyroid glands is overactive. Guided by a tumor-imaging test, your surgeon will make a small incision, or cut, in your neck to remove the gland. The small incision means you will probably have less pain and a faster recovery than people who have more invasive surgery. You can go home the same day. Your doctor may use regional or general anesthesia during the surgery.
- Bilateral neck exploration. This type of surgery uses a larger incision that lets the surgeon find and look at all four parathyroid glands and remove the overactive ones. If you have bilateral neck exploration, you will probably have general anesthesia and may need to stay in the hospital overnight.
Rarely, problems can occur after parathyroid surgery. In about 1 out of every 100 people, the nerves controlling the vocal cords (superior and inferior laryngeal nerves) are damaged during surgery, which most often results in hoarseness 251. This condition usually gets better on its own.
Low calcium levels in the blood or hypocalcemia may occur after surgery but usually return to normal in a few days or weeks. On rare occasions, not enough parathyroid tissue is left to make parathyroid hormone (PTH), which can result in hypoparathyroidism, a disorder in which the parathyroid glands in the neck do not produce enough parathyroid hormone (PTH).
Monitoring
Some people who have mild primary hyperparathyroidism may not need surgery right away, or even any surgery, and can be safely monitored.
You may want to talk with your doctor about long-term monitoring if you:
- don’t have symptoms
- have only slightly high blood calcium levels 10.5 to 12 mg/dL (2.62 to 2.99 mmol/L)
- have normal kidneys and bone density
Long-term monitoring should include regular doctor visits, a yearly blood test to measure calcium levels and check your kidney function, and a bone density test every 1 to 2 years.
If you and your doctor choose long-term monitoring, you should:
- drink plenty of water so you don’t get dehydrated
- get regular physical activity to help keep your bones strong
- avoid certain diuretics, such as thiazides
Medicines
Cinacalcet is a medicine that decreases the amount of PTH the parathyroid glands make and lowers calcium levels in the blood. Doctors may prescribe cinacalcet to treat very high calcium levels in people with primary hyperparathyroidism who can’t have surgery.
Cinacalcet does not improve bone density. If you have bone loss, your doctor may prescribe alendronate or other medications to help increase bone density.
Hypercalcemia Self-care
After you go home, follow your health care provider’s instructions about making sure your calcium level does not get high again.
You may need to drink a lot of liquids.
- Make sure you drink as much water every day as your provider recommends.
- Keep water next to your bed at night and drink some when you get up to use the bathroom.
DO NOT cut back on how much salt you eat.
Your provider may ask you to limit foods with a lot of calcium, or not to eat them at all for a while.
- Eat fewer dairy foods (such as cheese, milk, yogurt, ice cream) or DO NOT eat them at all.
- If your provider says you can eat dairy foods, DO NOT eat those that have extra calcium added. Read the labels carefully.
To further keep your calcium level from getting high again:
- DO NOT use antacids that have a lot of calcium in them. Look for antacids that have magnesium. Ask your provider which ones are OK.
- Ask your doctor what medicines and herbs are safe for you to take.
- If your doctor prescribes medicines to help keep your calcium level from getting too high again, take them the way you’re told to. Call your doctor if you have any side effects.
- Stay active when you get home. Your provider will tell you how much activity and exercise are OK.
You will probably need to get blood tests after you go home.
Keep any follow-up appointments you make with your doctor.
Hypercalcemia outlook (prognosis)
How well you do depends on the cause of your high calcium level. The outlook is good for people with mild hyperparathyroidism or hypercalcemia that have a treatable cause. Most of the time, there are no complications.
People with hypercalcemia due to conditions such as cancer or sarcoidosis may not do well. This is most often due to the disease itself, rather than the high calcium level.
- Tonon CR, Silva TAAL, Pereira FWL, Queiroz DAR, Junior ELF, Martins D, Azevedo PS, Okoshi MP, Zornoff LAM, de Paiva SAR, Minicucci MF, Polegato BF. A Review of Current Clinical Concepts in the Pathophysiology, Etiology, Diagnosis, and Management of Hypercalcemia. Med Sci Monit. 2022 Feb 26;28:e935821. doi: 10.12659/MSM.935821[↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Basso SM, Lumachi F, Nascimben F, Luisetto G, Camozzi V. Treatment of acute hypercalcemia. Med Chem. 2012 Jul;8(4):564-8. doi: 10.2174/157340612801216382[↩]
- Basso SM, Lumachi F, Nascimben F, Luisetto G, Camozzi V. Treatment of acute hypercalcemia. Med Chem. 2012 Jul;8(4):564-8. doi: 10.2174/15734061280121638[↩]
- Chang WT, Radin B, McCurdy MT. Calcium, magnesium, and phosphate abnormalities in the emergency department. Emerg Med Clin North Am. 2014 May;32(2):349-66. doi: 10.1016/j.emc.2013.12.006[↩][↩]
- Goldner W. Cancer-Related Hypercalcemia. J Oncol Pract. 2016 May;12(5):426-32. doi: 10.1200/JOP.2016.011155[↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Walker MD, Shane E. Hypercalcemia: A Review. JAMA. 2022;328(16):1624–1636. doi:10.1001/jama.2022.18331[↩][↩]
- Turner JJO. Hypercalcaemia – presentation and management . Clin Med (Lond). 2017 Jun;17(3):270-273. doi: 10.7861/clinmedicine.17-3-270[↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Sadiq NM, Naganathan S, Badireddy M. Hypercalcemia. [Updated 2022 Sep 5]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK430714/[↩]
- Zagzag, J., Hu, M.I., Fisher, S.B. and Perrier, N.D. (2018), Hypercalcemia and cancer: Differential diagnosis and treatment. CA: A Cancer Journal for Clinicians, 68: 377-386. https://doi.org/10.3322/caac.21489[↩]
- Silverberg SJ, Fitzpatrick LA, Bilezikian JP. Hyperparathyroidism. In: Becker KL, ed. Principles and practice of endocrinology and metabolism. 2d ed. Philadelphia: Lippincott, 1995:512–9.[↩]
- Bringhurst FR, Demay MB, Kronenberg HM. Hormones and disorders of mineral metabolism. In: Wilson JD, ed. Williams Textbook of endocrinology. 9th ed. philadelphia: Saunders, 1998:1155–1209.[↩]
- Wermers RA, Khosla S, Atkinson EJ, Hodgson SF, O’Fallon WM, Melton LJ 3rd. The rise and fall of primary hyperparathyroidism: a population-based study in Rochester, Minnesota, 1965-1992. Ann Intern Med. 1997 Mar 15;126(6):433-40. doi: 10.7326/0003-4819-126-6-199703150-00003[↩]
- al Zahrani A, Levine MA. Primary hyperparathyroidism. Lancet. 1997 Apr 26;349(9060):1233-8. doi: 10.1016/s0140-6736(96)06176-4[↩]
- Žofková I. Hypercalcemia. Pathophysiological aspects. Physiol Res. 2016;65(1):1-10. https://www.biomed.cas.cz/physiolres/pdf/65/65_1.pdf[↩][↩][↩]
- Radványi I, Csikós A, Balogh S. Malignitáshoz társuló hypercalcaemia korai felismerésének jelentősége [The significance of early diagnosis of cancer-related hypercalcaemia]. Orv Hetil. 2013 Sep 1;154(35):1367-73. Hungarian. doi: 10.1556/OH.2013.29665[↩][↩]
- Banu S, Batool S, Sattar S, Masood MQ. Malignant and Non-Malignant Causes of Hypercalcemia: A Retrospective Study at a Tertiary Care Hospital in Pakistan. Cureus. 2021 Jun 22;13(6):e15845. doi: 10.7759/cureus.15845[↩]
- Motlaghzadeh Y, Bilezikian JP, Sellmeyer DE. Rare Causes of Hypercalcemia: 2021 Update. J Clin Endocrinol Metab. 2021 Oct 21;106(11):3113-3128. doi: 10.1210/clinem/dgab504[↩]
- Kinder BK, Stewart AF. Hypercalcemia. Curr Probl Surg. 2002 Apr;39(4):349-448. doi: 10.1067/msg.2002.122220[↩][↩]
- Bollerslev J, Pretorius M, Heck A. Parathyroid hormone independent hypercalcemia in adults. Best Pract Res Clin Endocrinol Metab. 2018 Oct;32(5):621-638. doi: 10.1016/j.beem.2018.06.005[↩][↩]
- Murphy E, Williams GR. Hypocalcaemia. Medicine. 2009;37(9):465–468. doi: 10.1016/j.mpmed.2009.06.003[↩][↩]
- Cooper MS, Gittoes NJ. Diagnosis and management of hypocalcaemia. BMJ. 2008;336(7656):1298–302.[↩]
- Institute of Medicine (US) Committee to Review Dietary Reference Intakes for Vitamin D and Calcium; Ross AC, Taylor CL, Yaktine AL, et al., editors. Dietary Reference Intakes for Calcium and Vitamin D. Washington (DC): National Academies Press (US); 2011. Available from: https://www.ncbi.nlm.nih.gov/books/NBK56070/ doi: 10.17226/13050[↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Weaver CM. Calcium. In: Marriott BP, Birt DF, Stallings VA, Yates AA, eds. Present Knowledge in Nutrition. 11th ed. Cambridge, Massachusetts: Wiley-Blackwell; 2020:321-48.[↩][↩][↩][↩][↩][↩][↩]
- Bilezikian JP, Bandeira L, Khan A, Cusano NE. Hyperparathyroidism. Lancet. 2018 Jan 13;391(10116):168-178. doi: 10.1016/S0140-6736(17)31430-7[↩]
- Dirk Vanderschueren and others, Active Vitamin D (1,25-Dihydroxyvitamin D) and Bone Health in Middle-Aged and Elderly Men: The European Male Aging Study (EMAS), The Journal of Clinical Endocrinology & Metabolism, Volume 98, Issue 3, 1 March 2013, Pages 995–1005, https://doi.org/10.1210/jc.2012-2772[↩][↩]
- Lips P. Vitamin D physiology. Prog Biophys Mol Biol. 2006 Sep;92(1):4-8. doi: 10.1016/j.pbiomolbio.2006.02.016[↩][↩][↩][↩]
- Lieben L, Carmeliet G, Masuyama R. Calcemic actions of vitamin D: effects on the intestine, kidney and bone. Best Pract Res Clin Endocrinol Metab. 2011 Aug;25(4):561-72. doi: 10.1016/j.beem.2011.05.008[↩][↩]
- W Boron, E Boulpaep, eds. Medical Physiology. 2nd ed. Philadelphia, PA: Saunders; 2012.[↩]
- Khan M, Jose A, Sharma S. Physiology, Parathyroid Hormone. [Updated 2022 Oct 29]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK499940[↩]
- Simm PJ, Biggin A, Zacharin MR, Rodda CP, Tham E, Siafarikas A, Jefferies C, Hofman PL, Jensen DE, Woodhead H, Brown J, Wheeler BJ, Brookes D, Lafferty A, Munns CF; APEG Bone Mineral Working Group. Consensus guidelines on the use of bisphosphonate therapy in children and adolescents. J Paediatr Child Health. 2018 Mar;54(3):223-233. doi: 10.1111/jpc.13768[↩]
- Mannstadt M, Bilezikian JP, Thakker RV, Hannan FM, Clarke BL, Rejnmark L, Mitchell DM, Vokes TJ, Winer KK, Shoback DM. Hypoparathyroidism. Nat Rev Dis Primers. 2017 Aug 31;3:17055. doi: 10.1038/nrdp.2017.55. Erratum in: Nat Rev Dis Primers. 2017 Oct 05;3:17080[↩]
- Walker J. Diagnosis and management of patients with hypercalcaemia. Nurs Older People. 2015 May;27(4):22-6. doi: 10.7748/nop.27.4.22.e685[↩]
- Sadiq NM, Naganathan S, Badireddy M. Hypercalcemia. [Updated 2022 Sep 5]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK430714[↩][↩][↩][↩][↩][↩][↩][↩]
- Gilman, A. G., L. S. Goodman, and A. Gilman. (eds.). Goodman and Gilman’s The Pharmacological Basis of Therapeutics. 6th ed. New York: Macmillan Publishing Co., Inc. 1980., p. 1528[↩]
- Harvard University, Harvard School of Public Health. Calcium: What’s Best for Your Bones and Health ? https://www.hsph.harvard.edu/nutritionsource/calcium-full-story/[↩][↩][↩]
- Wawrzyniak N, Suliburska J. Nutritional and health factors affecting the bioavailability of calcium: a narrative review. Nutr Rev. 2021 Nov 10;79(12):1307-1320. doi: 10.1093/nutrit/nuaa138[↩]
- Calcium. https://lpi.oregonstate.edu/mic/minerals/calcium[↩][↩]
- Weaver CM. Calcium. In: Erdman JJ, Macdonald I, Zeisel S, eds. Present Knowledge in Nutrition. 10th ed: John Wiley & Sons, Inc.; 2012:434-446.[↩]
- Calcium. https://ods.od.nih.gov/factsheets/Calcium-HealthProfessional[↩][↩]
- Calcium. https://ods.od.nih.gov/factsheets/Calcium-Consumer[↩][↩][↩][↩]
- Fairweather-Tait SJ, Teucher B. Iron and calcium bioavailability of fortified foods and dietary supplements. Nutr Rev. 2002 Nov;60(11):360-7. doi: 10.1301/00296640260385801[↩]
- Song L. Calcium and Bone Metabolism Indices. Adv Clin Chem. 2017;82:1-46. doi: 10.1016/bs.acc.2017.06.005[↩][↩]
- Weaver CM, Heaney RP. Calcium. In: Ross AC, Caballero B, Cousins RJ, Tucker KL, Ziegler TR, eds. Modern Nutrition in Health and Disease. 11th ed. Baltimore, MD: Lippincott Williams & Wilkins; 2014:133-49.[↩][↩][↩][↩]
- Singh S, Dodt J, Volkers P, Hethershaw E, Philippou H, Ivaskevicius V, Imhof D, Oldenburg J, Biswas A. Structure functional insights into calcium binding during the activation of coagulation factor XIII A. Sci Rep. 2019 Aug 5;9(1):11324. doi: 10.1038/s41598-019-47815-z[↩]
- Kuo IY, Ehrlich BE. Signaling in muscle contraction. Cold Spring Harb Perspect Biol. 2015 Feb 2;7(2):a006023. doi: 10.1101/cshperspect.a006023[↩]
- Webb RC, Bohr DF. Mechanism of membrane stabilization by calcium in vascular smooth muscle. Am J Physiol. 1978 Nov;235(5):C227-32. doi: 10.1152/ajpcell.1978.235.5.C227[↩]
- Ariyan CE, Sosa JA. Assessment and management of patients with abnormal calcium. Crit Care Med. 2004 Apr;32(4 Suppl):S146-54. doi: 10.1097/01.ccm.0000117172.51403.af[↩]
- Wesseling-Perry K, Wang H, Elashoff R, Gales B, Jüppner H, Salusky IB. Lack of FGF23 response to acute changes in serum calcium and PTH in humans. J Clin Endocrinol Metab. 2014 Oct;99(10):E1951-6. doi: 10.1210/jc.2014-2125[↩]
- Clapham DE. Calcium signaling. Cell. 2007 Dec 14;131(6):1047-58. doi: 10.1016/j.cell.2007.11.028[↩]
- Marcocci C, Cetani F. Clinical practice. Primary hyperparathyroidism. N Engl J Med. 2011 Dec 22;365(25):2389-97. doi: 10.1056/NEJMcp1106636. Erratum in: N Engl J Med. 2012 May 31;366(22):2138.[↩][↩]
- Lumachi F, Basso SM. Pathophysiology and treatment of nonfamilial hyperparathyroidism. Recent Pat CNS Drug Discov. 2014;9(3):164-72. doi: 10.2174/1574889810666150131141054[↩][↩]
- Eastell R, Brandi ML, Costa AG, D’Amour P, Shoback DM, Thakker RV. Diagnosis of asymptomatic primary hyperparathyroidism: proceedings of the Fourth International Workshop. J Clin Endocrinol Metab. 2014 Oct;99(10):3570-9. doi: 10.1210/jc.2014-1414. Epub 2014 Aug 27. Erratum in: J Clin Endocrinol Metab. 2015 May;100(5):2137.[↩][↩]
- Agarwal SK. Multiple endocrine neoplasia type 1. Front Horm Res. 2013;41:1-15. doi: 10.1159/000345666[↩][↩]
- Arnold A, Shattuck TM, Mallya SM, Krebs LJ, Costa J, Gallagher J, Wild Y, Saucier K. Molecular pathogenesis of primary hyperparathyroidism. J Bone Miner Res. 2002 Nov;17 Suppl 2:N30-6.[↩]
- Kjaer S, Kurokawa K, Perrinjaquet M, Abrescia C, Ibáñez CF. Self-association of the transmembrane domain of RET underlies oncogenic activation by MEN2A mutations. Oncogene. 2006 Nov 9;25(53):7086-95. doi: 10.1038/sj.onc.1209698[↩]
- Lips CJ, Dreijerink KM, Höppener JW. Variable clinical expression in patients with a germline MEN1 disease gene mutation: clues to a genotype-phenotype correlation. Clinics (Sao Paulo). 2012;67 Suppl 1(Suppl 1):49-56. doi: 10.6061/clinics/2012(sup01)10[↩]
- Hendy GN, Cole DE. Genetic defects associated with familial and sporadic hyperparathyroidism. Front Horm Res. 2013;41:149-65. doi: 10.1159/000345675[↩]
- Goltzman D. Approach to Hypercalcemia. In: De Groot LJ, editor; Chrousos G, editor; Dungan K, et al., editors. Approach to Hypercalcemia. South Dartmouth (MA): MDText.com, Inc.; 2000.[↩]
- Iwakura T, Ohashi N, Tsuji N, Naito Y, Isobe S, Ono M, Fujikura T, Tsuji T, Sakao Y, Yasuda H, Kato A, Fujiyama T, Tokura Y, Fujigaki Y. Calcitriol-induced hypercalcemia in a patient with granulomatous mycosis fungoides and end-stage renal disease. World J Nephrol. 2013 May 6;2(2):44-8. doi: 10.5527/wjn.v2.i2.44[↩]
- Agrawal N, Altiner S, Mezitis NH, Helbig S. Silicone-induced granuloma after injection for cosmetic purposes: a rare entity of calcitriol-mediated hypercalcemia. Case Rep Med. 2013;2013:807292. doi: 10.1155/2013/807292[↩]
- Khasawneh FA, Ahmed S, Halloush RA. Progressive disseminated histoplasmosis presenting with cachexia and hypercalcemia. Int J Gen Med. 2013;6:79-83. doi: 10.2147/IJGM.S41520[↩]
- Yılmaz R, Kundak AA, Sezer T, Özer S, Esmeray H, Kazancı NÖ. Idiopathic infantile hypercalcemia or an extrapulmonary complication of tuberculosis? Tuberk Toraks. 2013;61(1):43-6. doi: 10.5578/tt.536[↩]
- Pearce SH, Trump D, Wooding C, Besser GM, Chew SL, Grant DB, Heath DA, Hughes IA, Paterson CR, Whyte MP, et al. Calcium-sensing receptor mutations in familial benign hypercalcemia and neonatal hyperparathyroidism. J Clin Invest. 1995 Dec;96(6):2683-92. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC185975/pdf/jcinvest00018-0148.pdf[↩]
- Dochez V, Ducarme G. Primary hyperparathyroidism during pregnancy. Arch Gynecol Obstet. 2015 Feb;291(2):259-63. doi: 10.1007/s00404-014-3526-8[↩]
- Laporta J, Keil KP, Weaver SR, Cronick CM, Prichard AP, Crenshaw TD, Heyne GW, Vezina CM, Lipinski RJ, Hernandez LL. Serotonin regulates calcium homeostasis in lactation by epigenetic activation of hedgehog signaling. Mol Endocrinol. 2014 Nov;28(11):1866-74. doi: 10.1210/me.2014-1204[↩]
- Sato K. Hypercalcemia during pregnancy, puerperium, and lactation: review and a case report of hypercalcemic crisis after delivery due to excessive production of PTH-related protein (PTHrP) without malignancy (humoral hypercalcemia of pregnancy). Endocr J. 2008 Dec;55(6):959-66. doi: 10.1507/endocrj.k08e-092[↩][↩]
- Walker MD, Bilezikian JP. Primary Hyperparathyroidism. [Updated 2021 Apr 19]. In: Feingold KR, Anawalt B, Blackman MR, et al., editors. Endotext [Internet]. South Dartmouth (MA): MDText.com, Inc.; 2000-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK278923[↩][↩][↩]
- Eufrazino C, Veras A, Bandeira F. Epidemiology of Primary Hyperparathyroidism and its Non-classical Manifestations in the City of Recife, Brazil. Clin Med Insights Endocrinol Diabetes. 2013 Dec 4;6:69-74. doi: 10.4137/CMED.S1314[↩]
- Griebeler ML, Kearns AE, Ryu E, Hathcock MA, Melton LJ 3rd, Wermers RA. Secular trends in the incidence of primary hyperparathyroidism over five decades (1965-2010). Bone. 2015 Apr;73:1-7. doi: 10.1016/j.bone.2014.12.003[↩][↩]
- Primary Hyperparathyroidism. https://www.endocrine.org/patient-engagement/endocrine-library/primary-hyperparathyroidism[↩][↩]
- Hyperparathyroidism. https://www.mayoclinic.org/diseases-conditions/hyperparathyroidism/symptoms-causes/syc-20356194[↩]
- Primary Hyperparathyroidism. https://www.niddk.nih.gov/health-information/endocrine-diseases/primary-hyperparathyroidism[↩]
- Bilezikian JP, Brandi ML, Eastell R, Silverberg SJ, Udelsman R, Marcocci C, Potts JT Jr. Guidelines for the management of asymptomatic primary hyperparathyroidism: summary statement from the Fourth International Workshop. J Clin Endocrinol Metab. 2014 Oct;99(10):3561-9. doi: 10.1210/jc.2014-1413[↩][↩]
- Campbell MJ. The Definitive Management of Primary Hyperparathyroidism: Who Needs an Operation? JAMA. 2017 Mar 21;317(11):1167-1168. doi: 10.1001/jama.2017.1620[↩][↩][↩][↩][↩]
- Udelsman R, Åkerström G, Biagini C, Duh QY, Miccoli P, Niederle B, Tonelli F. The surgical management of asymptomatic primary hyperparathyroidism: proceedings of the Fourth International Workshop. J Clin Endocrinol Metab. 2014 Oct;99(10):3595-606. doi: 10.1210/jc.2014-2000[↩]
- Udelsman R, Pasieka JL, Sturgeon C, Young JE, Clark OH. Surgery for asymptomatic primary hyperparathyroidism: proceedings of the third international workshop. J Clin Endocrinol Metab. 2009 Feb;94(2):366-72. doi: 10.1210/jc.2008-1761[↩]
- Wright JD, Tergas AI, Ananth CV, Burke WM, Hou JY, Chen L, Neugut AI, Richards CA, Hershman DL. Quality and Outcomes of Treatment of Hypercalcemia of Malignancy. Cancer Invest. 2015;33(8):331-9. doi: 10.3109/07357907.2015.1047506[↩]
- Zajickova K, Vrbikova J, Canaff L, Pawelek PD, Goltzman D, Hendy GN. Identification and functional characterization of a novel mutation in the calcium-sensing receptor gene in familial hypocalciuric hypercalcemia: modulation of clinical severity by vitamin D status. J Clin Endocrinol Metab. 2007 Jul;92(7):2616-23. doi: 10.1210/jc.2007-0123[↩][↩]
- Garner SC, Pi M, Tu Q, Quarles LD. Rickets in cation-sensing receptor-deficient mice: an unexpected skeletal phenotype. Endocrinology. 2001 Sep;142(9):3996-4005. doi: 10.1210/endo.142.9.8364[↩]
- Wu JX, Yeh MW. Asymptomatic Primary Hyperparathyroidism: Diagnostic Pitfalls and Surgical Intervention. Surg Oncol Clin N Am. 2016 Jan;25(1):77-90. doi: 10.1016/j.soc.2015.08.004[↩][↩]
- Familial hypocalciuric hypercalcemia. https://rarediseases.info.nih.gov/diseases/10828/familial-hypocalciuric-hypercalcemia[↩][↩][↩][↩][↩][↩][↩]
- Hendy GN, Canaff L, Cole DE. The CASR gene: alternative splicing and transcriptional control, and calcium-sensing receptor (CaSR) protein: structure and ligand binding sites. Best Pract Res Clin Endocrinol Metab. 2013 Jun;27(3):285-301. doi: 10.1016/j.beem.2013.02.009[↩]
- Stewart AF. Clinical practice. Hypercalcemia associated with cancer. N Engl J Med. 2005 Jan 27;352(4):373-9. doi: 10.1056/NEJMcp042806[↩][↩][↩]
- Gonciulea AR, Wang Y, Bikle DD, Sellmeyer DE. Hypercalcemia in non-Hodgkin’s lymphoma due to cosecretion of PTHrP and 1,25-dihydroxyvitamin D. Osteoporos Int. 2021 Dec;32(12):2587-2592. doi: 10.1007/s00198-021-06054-7[↩][↩][↩]
- O’Callaghan S, Yau H. Treatment of malignancy-associated hypercalcemia with cinacalcet: a paradigm shift. Endocr Connect. 2021 Jan;10(1):R13-R24. doi: 10.1530/EC-20-0487[↩]
- Klemencic S, Perkins J. Diagnosis and Management of Oncologic Emergencies. West J Emerg Med. 2019 Mar;20(2):316-322. doi: 10.5811/westjem.2018.12.37335[↩][↩][↩]
- Basso U, Maruzzo M, Roma A, Camozzi V, Luisetto G, Lumachi F. Malignant hypercalcemia. Curr Med Chem. 2011;18(23):3462-7. doi: 10.2174/092986711796642490[↩][↩]
- McCurdy MT, Shanholtz CB. Oncologic emergencies. Crit Care Med. 2012 Jul;40(7):2212-22. doi: 10.1097/CCM.0b013e31824e1865[↩][↩][↩][↩][↩]
- de Vernejoul MC. Role of Parathyroid Hormone Related Peptide (PTHrP) in Hypercalcemia of Malignancy and the Development of Osteolytic Metastases. J Clin Rheumatol. 1997 Apr;3(2 Suppl):109. doi: 10.1097/00124743-199704001-00024[↩]
- Kohno N. [The treatment for cancer with bone metastases -whether to use zoledoronate or denosumab for bone metastases-]. Clin Calcium. 2014 Aug;24(8):1229-36. Japanese.[↩]
- Eid W, Wheeler TM, Sharma MD. Recurrent hypercalcemia due to ectopic production of parathyroid hormone-related protein and intact parathyroid hormone in a single patient with multiple malignancies. Endocr Pract. 2004 Mar-Apr;10(2):125-8. doi: 10.4158/EP.10.2.125[↩][↩][↩]
- Picolos MK, Lavis VR, Orlander PR. Milk-alkali syndrome is a major cause of hypercalcaemia among non-end-stage renal disease (non-ESRD) inpatients. Clin Endocrinol (Oxf). 2005 Nov;63(5):566-76. doi: 10.1111/j.1365-2265.2005.02383.x[↩]
- Zagzag J, Hu MI, Fisher SB, Perrier ND. Hypercalcemia and cancer: Differential diagnosis and treatment. CA Cancer J Clin. 2018 Sep;68(5):377-386. doi: 10.3322/caac.21489[↩]
- Patel V, Mehra D, Ramirez B, Lindo A, Suarez M. Milk-Alkali Syndrome as a Cause of Hypercalcemia in a Gentleman With Acute Kidney Injury and Excessive Antacid Intake. Cureus. 2021 Feb 1;13(2):e13056. doi: 10.7759/cureus.13056[↩]
- Rizk MA, Abourizk N, Kovalenko I, Golubykh K, Soni B, Zaccheo M, Zanders S. Milk-Alkali Syndrome: How Electronic Medical Record Open Notes Helped to Rule Out Cancer. Am J Case Rep. 2022 Sep 3;23:e936969. doi: 10.12659/AJCR.936969[↩][↩]
- Medarov BI. Milk-alkali syndrome. Mayo Clin Proc. 2009 Mar;84(3):261-7. doi: 10.1016/S0025-6196(11)61144-0[↩]
- Orwoll ES. The milk-alkali syndrome: current concepts. Ann Intern Med. 1982 Aug;97(2):242-8. doi: 10.7326/0003-4819-97-2-242[↩]
- Abreo K, Adlakha A, Kilpatrick S, Flanagan R, Webb R, Shakamuri S. The milk-alkali syndrome. A reversible form of acute renal failure. Arch Intern Med. 1993 Apr 26;153(8):1005-10.[↩]
- Swanson CM, Mackey PA, Westphal SA, Argueta R. Nicotine-substitute gum-induced milk alkali syndrome: a look at unexpected sources of calcium. Endocr Pract. 2013 Nov-Dec;19(6):142-4. doi: 10.4158/EP13080.CR[↩][↩]
- Satyarengga M, Silver KD. Severe hypercalcaemia due to household cleaner ingestion. BMJ Case Rep. 2019 Oct 30;12(10):e231771. doi: 10.1136/bcr-2019-231771[↩]
- Kim JH, Jung HM, Kim JS, Kim JH, Han SB, Hong SB. Liquid calcium fertilizer intoxication. Clin Toxicol (Phila). 2010 Jun;48(5):466-7. doi: 10.3109/15563650.2010.487488[↩]
- Wu KD, Chuang RB, Wu FL, Hsu WA, Jan IS, Tsai KS. The milk-alkali syndrome caused by betelnuts in oyster shell paste. J Toxicol Clin Toxicol. 1996;34(6):741-5. doi: 10.3109/15563659609013839[↩]
- Lin SH, Lin YF, Cheema-Dhadli S, Davids MR, Halperin ML. Hypercalcaemia and metabolic alkalosis with betel nut chewing: emphasis on its integrative pathophysiology. Nephrol Dial Transplant. 2002 May;17(5):708-14. doi: 10.1093/ndt/17.5.708[↩]
- Reid IR. Transient hypercalcemia following overdoses of soluble aspirin tablets. Aust N Z J Med. 1985 Jun;15(3):364. doi: 10.1111/j.1445-5994.1985.tb04063.x[↩]
- Kallner G, Karlsson H. Recurrent factitious hypercalcemia. Am J Med. 1987 Mar;82(3):536-8. doi: 10.1016/0002-9343(87)90457-8[↩]
- Frame B, Jackson GM, Kleerekoper M, Rao DS, DeLorenzo AJ, Garcia M. Acute severe hypercalcemia in a la Münchausen. Am J Med. 1981 Feb;70(2):316-9. doi: 10.1016/0002-9343(81)90767-1[↩]
- Ng RW, Cheng YL. Calcium alginate dressing-related hypercalcemia. J Burn Care Res. 2007 Jan-Feb;28(1):203-4. doi: 10.1097/BCR.0B013E31802CA471[↩]
- Kallala R, Haddad FS. Hypercalcaemia following the use of antibiotic-eluting absorbable calcium sulphate beads in revision arthroplasty for infection. Bone Joint J. 2015 Sep;97-B(9):1237-41. doi: 10.1302/0301-620X.97B9.34532[↩]
- Cho K, Seo B, Koh H, Yang H. Fatal case of commercial moisture absorber ingestion. BMJ Case Rep. 2018 Apr 26;2018:bcr2018225121. doi: 10.1136/bcr-2018-225121[↩]
- Padmanabhan P, Spiller HA, Bosse GM. A case of hypercalcemia and gastric necrosis from hot pack ingestion. J Med Toxicol. 2010 Jun;6(2):139-42. doi: 10.1007/s13181-010-0063-0[↩]
- Zhang Y, Wang X, Liu L, Jiang R, Ye C, Ni L, You C, Han C. Hypercalcaemic crisis in a young man with extensive burns caused by heated CaCl2 solution: a case report. Burns. 2014 May;40(3):e12-4. doi: 10.1016/j.burns.2013.08.026[↩]
- Yasaman Motlaghzadeh and others, Rare Causes of Hypercalcemia: 2021 Update, The Journal of Clinical Endocrinology & Metabolism, Volume 106, Issue 11, November 2021, Pages 3113–3128, https://doi.org/10.1210/clinem/dgab504[↩][↩][↩][↩][↩][↩][↩]
- Bennett MT, Sirrs S, Yeung JK, Smith CA. Hypercalcemia due to all trans retinoic acid in the treatment of acute promyelocytic leukemia potentiated by voriconazole. Leuk Lymphoma. 2005 Dec;46(12):1829-31. doi: 10.1080/10428190500235298[↩]
- Cordoba R, Ramirez E, Lei SH, Lopez de la Guia A, Sanjurjo MJ, Carcas AJ, Hernandez-Navarro F. Hypercalcemia due to an interaction of all-trans retinoic acid (ATRA) and itraconazole therapy for acute promyelocytic leukemia successfully treated with zoledronic acid. Eur J Clin Pharmacol. 2008 Oct;64(10):1031-2. doi: 10.1007/s00228-008-0517-3[↩][↩]
- Rabinowicz S, Somech R, Yeshayahu Y. Foscarnet-related Hypercalcemia During CMV Treatment in an Infant With SCID: A Case Report and Review of Literature. J Pediatr Hematol Oncol. 2017 Apr;39(3):e173-e175. doi: 10.1097/MPH.0000000000000682[↩][↩]
- Gayet S, Ville E, Durand JM, Mars ME, Morange S, Kaplanski G, Gallais H, Soubeyrand J. Foscarnet-induced hypercalcaemia in AIDS. AIDS. 1997 Jul;11(8):1068-70.[↩][↩]
- Setsu N, Kobayashi E, Asano N, Yasui N, Kawamoto H, Kawai A, Horiuchi K. Severe hypercalcemia following denosumab treatment in a juvenile patient. J Bone Miner Metab. 2016 Jan;34(1):118-22. doi: 10.1007/s00774-015-0677-z[↩][↩][↩]
- Roux S, Massicotte MH, Huot Daneault A, Brazeau-Lamontagne L, Dufresne J. Acute hypercalcemia and excessive bone resorption following anti-RANKL withdrawal: Case report and brief literature review. Bone. 2019 Mar;120:482-486. doi: 10.1016/j.bone.2018.12.012[↩][↩][↩]
- Uchida T, Yamaguchi H, Kushima C, Yonekawa T, Nakazato M. Elevated levels of circulating fibroblast growth factor 23 with hypercalcemia following discontinuation of denosumab. Endocr J. 2020 Jan 28;67(1):31-35. doi: 10.1507/endocrj.EJ19-0198[↩][↩][↩]
- Koldkjær Sølling AS, Harsløf T, Kaal A, Rejnmark L, Langdahl B. Hypercalcemia after discontinuation of long-term denosumab treatment. Osteoporos Int. 2016 Jul;27(7):2383-2386. doi: 10.1007/s00198-016-3535-5[↩][↩][↩]
- Wall CA, Gaffney EF, Mellotte GJ. Hypercalcaemia and acute interstitial nephritis associated with omeprazole therapy. Nephrol Dial Transplant. 2000 Sep;15(9):1450-2. doi: 10.1093/ndt/15.9.1450[↩][↩]
- McPherson ML, Prince SR, Atamer ER, Maxwell DB, Ross-Clunis H, Estep HL. Theophylline-induced hypercalcemia. Ann Intern Med. 1986 Jul;105(1):52-4. doi: 10.7326/0003-4819-105-1-52[↩][↩]
- Knox JB, Demling RH, Wilmore DW, Sarraf P, Santos AA. Hypercalcemia associated with the use of human growth hormone in an adult surgical intensive care unit. Arch Surg. 1995 Apr;130(4):442-5. doi: 10.1001/archsurg.1995.01430040104024[↩][↩]
- Shike M, Sturtridge WC, Tam CS, Harrison JE, Jones G, Murray TM, Husdan H, Whitwell J, Wilson DR, Jeejeebhoy KN. A possible role of vitamin D in the genesis of parenteral-nutrition-induced metabolic bone disease. Ann Intern Med. 1981 Nov;95(5):560-8. doi: 10.7326/0003-4819-95-5-560[↩][↩]
- Cathebras P, Cartry O, Lafage-Proust MH, Lauwers A, Acquart S, Thomas T, Rousset H. Arthritis, hypercalcemia, and lytic bone lesions after hepatitis B vaccination. J Rheumatol. 1996 Mar;23(3):558-60.[↩][↩]
- Chandra S , Seth P , Mankeshwar J . Manganese poisoning: clinical and biochemical observations. Environ Res 1974 ;7:374 –380.[↩][↩]
- Bennett MT, Sirrs S, Yeung JK, Smith CA. Hypercalcemia due to all trans retinoic acid in the treatment of acute promyelocytic leukemia potentiated by voriconazole. Leuk Lymphoma. 2005 Dec;46(12):1829-31. doi: 10.1080/1042819050023529[↩]
- Isaia GC, Pellissetto C, Ravazzoli M, Tamone C. Acute adrenal crisis and hypercalcemia in a patient assuming high liquorice doses. Minerva Med. 2008 Feb;99(1):91-4.[↩][↩]
- Agrawal S, Goyal A, Agarwal S, Khadgawat R. Hypercalcaemia, adrenal insufficiency and bilateral adrenal histoplasmosis in a middle-aged man: a diagnostic dilemma. BMJ Case Rep. 2019 Aug 28;12(8):e231142. doi: 10.1136/bcr-2019-231142[↩][↩]
- Wong RK, Gregory R, Lo TC. A case of isolated ACTH deficiency presenting with hypercalcaemia. Int J Clin Pract. 2000 Nov;54(9):623-4.[↩][↩]
- Yamada S, Arase H, Morishita T, Tsuchimoto A, Torisu K, Torisu T, Tsuruya K, Nakano T, Kitazono T. Adrenal crisis presented as acute onset of hypercalcemia and hyponatremia triggered by acute pyelonephritis in a patient with partial hypopituitarism and pre-dialysis chronic kidney disease. CEN Case Rep. 2019 May;8(2):83-88. doi: 10.1007/s13730-018-0371-9[↩][↩][↩][↩]
- Suzuki K, Nonaka K, Ichihara K, Fukumoto Y, Miyazaki A, Yamada Y, Itoh Y, Seino Y, Moriwaki K, Tarui S. Hypercalcemia in glucocorticoid withdrawal. Endocrinol Jpn. 1986 Apr;33(2):203-9. doi: 10.1507/endocrj1954.33.203[↩][↩]
- Bhatti RS, Flynn MD. Adrenal insufficiency secondary to inappropriate oral administration of topical exogenous steroids presenting with hypercalcaemia. BMJ Case Rep. 2012 Jun 21;2012:bcr0320125983. doi: 10.1136/bcr.03.2012.5983[↩]
- Sperling LS, Tumlin JA. Case report: delayed hypercalcemia after rhabdomyolysis-induced acute renal failure. Am J Med Sci. 1996 Apr;311(4):186-8. doi: 10.1097/00000441-199604000-00007[↩][↩]
- Shrestha SM, Berry JL, Davies M, Ballardie FW. Biphasic hypercalcemia in severe rhabdomyolysis: serial analysis of PTH and vitamin D metabolites. A case report and literature review. Am J Kidney Dis. 2004 Mar;43(3):e31-5. doi: 10.1053/j.ajkd.2003.10.045[↩][↩]
- Hechanova LA, Sadjadi SA. Severe hypercalcemia complicating recovery of acute kidney injury due to rhabdomyolysis. Am J Case Rep. 2014 Sep 14;15:393-6. doi: 10.12659/AJCR.891046[↩]
- Prince RL, Hutchison BG, Bhagat CI. Hypercalcemia during resolution of acute renal failure associated with rhabdomyolysis: evidence for suppression of parathyroid hormone and calcitriol. Aust N Z J Med. 1986 Aug;16(4):506-8. doi: 10.1111/j.1445-5994.1986.tb02023.x[↩]
- Kohut B, Rossat J, Raffoul W, Lamy O, Berger MM. Hypercalcaemia and acute renal failure after major burns: An under-diagnosed condition. Burns. 2010 May;36(3):360-6. doi: 10.1016/j.burns.2009.05.001[↩]
- Muller I, Premawardhana LD. Hypercalcaemia with undetectable parathormone levels. BMJ. 2018 Nov 8;363:k4074. doi: 10.1136/bmj.k4074[↩][↩][↩][↩]
- Rao AR, Aryal A, Thakral M, Painkra B, Chakrawarty A, Dey AB. Case Report: Hypercalcemia in an Older Patient with Type I Lepra Reaction. Am J Trop Med Hyg. 2020 Oct;103(4):1439-1440. doi: 10.4269/ajtmh.20-0114[↩]
- Ryzen E, Singer FR. Hypercalcemia in Leprosy. Arch Intern Med. 1985;145(7):1305–1306. doi:10.1001/archinte.1985.00360070187031[↩]
- Bansal N, Shah R, Patel A, Vaidya G, Pantangi P, Manocha D. Hypercalcemia as a primary manifestation of cryptococcal immune reconstitution syndrome-a rare presentation. Am J Emerg Med. 2015 Apr;33(4):598.e3-4. doi: 10.1016/j.ajem.2014.08.065[↩]
- Zaloga GP, Chernow B, Eil C. Hypercalcemia and disseminated cytomegalovirus infection in the acquired immunodeficiency syndrome. Ann Intern Med. 1985 Mar;102(3):331-3. doi: 10.7326/0003-4819-102-3-331[↩]
- Mudge CS, Yoo DC, Noto RB. Rheumatoid arthritis demonstrated on PET/CT as the etiology of hypercalcemia. Med Health R I. 2012 Feb;95(2):54-6.[↩]
- Schurman SJ, Bergstrom WH, Root AW, Souid AK, Hannan WP. Interleukin 1beta mediated calciotropic activity in serum of children with juvenile rheumatoid arthritis. J Rheumatol. 1998 Jan;25(1):161-5.[↩]
- Balfour S, Masse M, Plaisance M. A case of hypercalcemia and scabies. Am J Med. 2010 Apr;123(4):e5-6. doi: 10.1016/j.amjmed.2009.09.033[↩][↩]
- Dockrell DH, Poland GA. Hypercalcemia in a patient with hypoparathyroidism and Nocardia asteroides infection: a novel observation. Mayo Clin Proc. 1997 Aug;72(8):757-60. doi: 10.1016/S0025-6196(11)63596-9[↩]
- Roux S, de Vernejoul MC, Grossin M, Bandt MD, Palazzo E, Nʼzue MK, Meyer O, Kahn MF. An unusual case of diffuse focal osteolysis with disseminated osteoclastosis. J Clin Rheumatol. 1997 Feb;3(1):58-62. doi: 10.1097/00124743-199702000-00014[↩][↩]
- Gerhardt A, Greenberg A, Reilly JJ Jr, Van Thiel DH. Hypercalcemia. A complication of advanced chronic liver disease. Arch Intern Med. 1987 Feb;147(2):274-7. doi: 10.1001/archinte.147.2.274[↩]
- Byrne CD, Bermann L, Constant C, Cox TM. Pathological bone fractures preceded by sustained hypercalcaemia in type 1 Gaucher disease. J Inherit Metab Dis. 1997 Sep;20(5):709-10. doi: 10.1023/a:1005386728894[↩]
- Berar-Yanay N, Weiner P, Magadle R. Hypercalcaemia in systemic lupus erythematosus. Clin Rheumatol. 2001;20(2):147-9. doi: 10.1007/s100670170088[↩][↩]
- Greenwald RA, Stein B, Miller F. Rapid skeletal turnover and hypercalcemia associated with markedly elevated interleukin-6 levels in a young black man. Bone. 1998 Mar;22(3):285-8. doi: 10.1016/s8756-3282(97)00283-4[↩][↩][↩]
- Burtis WJ, Brady TG, Orloff JJ, Ersbak JB, Warrell RP Jr, Olson BR, Wu TL, Mitnick ME, Broadus AE, Stewart AF. Immunochemical characterization of circulating parathyroid hormone-related protein in patients with humoral hypercalcemia of cancer. N Engl J Med. 1990 Apr 19;322(16):1106-12. doi: 10.1056/NEJM199004193221603[↩]
- Grill V, Ho P, Body JJ, Johanson N, Lee SC, Kukreja SC, Moseley JM, Martin TJ. Parathyroid hormone-related protein: elevated levels in both humoral hypercalcemia of malignancy and hypercalcemia complicating metastatic breast cancer. J Clin Endocrinol Metab. 1991 Dec;73(6):1309-15. doi: 10.1210/jcem-73-6-1309[↩]
- Dagdelen S, Kalan I, Gurlek A. Humoral hypercalcemia of benignancy secondary to parathyroid hormone-related protein secreting uterine leiomyoma. Am J Med Sci. 2008 May;335(5):407-8. doi: 10.1097/MAJ.0b013e3181520125[↩][↩]
- Yamauchi M, Yotsuyanagi T, Saito T, Ikeda K, Urushidate S, Higuma Y. Three cases of giant pilomatrixoma–considerations for diagnosis and treatment of giant skin tumours with abundant inner calcification present on the upper body. J Plast Reconstr Aesthet Surg. 2010 Jun;63(6):e519-24. doi: 10.1016/j.bjps.2009.12.006[↩]
- Kambe Y, Nakano H, Kaneko T, Aizu T, Ikenaga S, Harada K, Nakajima N, Moritsugu R, Hanada K. Giant pilomatricoma associated with hypercalcaemia and elevated levels of parathyroid hormone-related protein. Br J Dermatol. 2006 Jul;155(1):208-10. doi: 10.1111/j.1365-2133.2006.07286.x[↩]
- Herring R, Laji K. Humoral hypercalcaemia of benignancy. A case report. QJM. 2008 Apr;101(4):329-30. doi: 10.1093/qjmed/hcn010[↩]
- Tarnawa E, Sullivan S, Underwood P, Richardson M, Spruill L. Severe hypercalcemia associated with uterine leiomyoma in pregnancy. Obstet Gynecol. 2011 Feb;117(2 Pt 2):473-476. doi: 10.1097/AOG.0b013e3181fd29ae[↩]
- Knecht TP, Behling CA, Burton DW, Glass CK, Deftos LJ. The humoral hypercalcemia of benignancy. A newly appreciated syndrome. Am J Clin Pathol. 1996 Apr;105(4):487-92. doi: 10.1093/ajcp/105.4.487[↩]
- Mahoney CP, Cassady C, Weinberger E, Winters WD, Benjamin DR. Humoral hypercalcemia due to an occult renal adenoma. Pediatr Nephrol. 1997 Jun;11(3):339-42. doi: 10.1007/s004670050291[↩]
- Kimura S, Nishimura Y, Yamaguchi K, Nagasaki K, Shimada K, Uchida H. A case of pheochromocytoma producing parathyroid hormone-related protein and presenting with hypercalcemia. J Clin Endocrinol Metab. 1990 Jun;70(6):1559-63. doi: 10.1210/jcem-70-6-1559[↩]
- Mune T, Katakami H, Kato Y, Yasuda K, Matsukura S, Miura K. Production and secretion of parathyroid hormone-related protein in pheochromocytoma: participation of an alpha-adrenergic mechanism. J Clin Endocrinol Metab. 1993 Mar;76(3):757-62. doi: 10.1210/jcem.76.3.8383146[↩]
- van Raalte DH, Goorden SM, Kemper EA, Brosens LA, ten Kate RW. Sarcoidosis-related hypercalcaemia due to production of parathyroid hormone-related peptide. BMJ Case Rep. 2015 Jul 9;2015:bcr2015210189. doi: 10.1136/bcr-2015-210189. Erratum in: BMJ Case Rep. 2015;2015. pii: bcr2015210189corr1. doi: 10.1136/bcr-2015-210189corr1[↩][↩]
- Deftos LJ, Burton DW, Baird SM, Terkeltaub RA. Hypercalcemia and systemic lupus erythematosus. Arthritis Rheum. 1996 Dec;39(12):2066-9. doi: 10.1002/art.1780391217[↩][↩]
- Zhao L, Huang L, Zhang X. Systemic lupus erythematosus-related hypercalcemia with ectopic calcinosis. Rheumatol Int. 2016 Jul;36(7):1023-6. doi: 10.1007/s00296-016-3486-3[↩]
- Braude S, Graham A, Mitchell D. Lymphoedema/hypercalcaemia syndrome mediated by parathyroid-hormone-related protein. Lancet. 1991 Jan 19;337(8734):140-1. doi: 10.1016/0140-6736(91)90802-v[↩]
- Burton D, Cheung T, Fierer J, et al. PTHrP production by HIV-associated lymphadenopathy with hypercalcemia. J Invest Med 1995;43:226A[↩]
- Lippuner K, Zehnder HJ, Casez JP, Takkinen R, Jaeger P. PTH-related protein is released into the mother’s bloodstream during lactation: evidence for beneficial effects on maternal calcium-phosphate metabolism. J Bone Miner Res. 1996 Oct;11(10):1394-9. doi: 10.1002/jbmr.5650111004[↩]
- Dobnig H, Kainer F, Stepan V, Winter R, Lipp R, Schaffer M, Kahr A, Nocnik S, Patterer G, Leb G. Elevated parathyroid hormone-related peptide levels after human gestation: relationship to changes in bone and mineral metabolism. J Clin Endocrinol Metab. 1995 Dec;80(12):3699-707. doi: 10.1210/jcem.80.12.8530622[↩]
- Winter EM, Appelman-Dijkstra NM. Parathyroid Hormone-Related Protein-Induced Hypercalcemia of Pregnancy Successfully Reversed by a Dopamine Agonist. J Clin Endocrinol Metab. 2017 Dec 1;102(12):4417-4420. doi: 10.1210/jc.2017-01617[↩][↩]
- Van Heerden JA, Gharib H, Jackson IT. Pseudohyperparathyroidism secondary to gigantic mammary hypertrophy. Arch Surg. 1988 Jan;123(1):80-2. doi: 10.1001/archsurg.1988.01400250090016[↩]
- Khosla S, van Heerden JA, Gharib H, Jackson IT, Danks J, Hayman JA, Martin TJ. Parathyroid hormone-related protein and hypercalcemia secondary to massive mammary hyperplasia. N Engl J Med. 1990 Apr 19;322(16):1157. doi: 10.1056/NEJM199004193221613[↩]
- Lepre F, Grill V, Ho PW, Martin TJ. Hypercalcemia in pregnancy and lactation associated with parathyroid hormone-related protein. N Engl J Med. 1993 Mar 4;328(9):666-7. doi: 10.1056/NEJM199303043280917[↩]
- Mather KJ, Chik CL, Corenblum B. Maintenance of serum calcium by parathyroid hormone-related peptide during lactation in a hypoparathyroid patient. J Clin Endocrinol Metab. 1999 Feb;84(2):424-7. doi: 10.1210/jcem.84.2.5486[↩]
- Wright AD, Joplin GF, Dixon HG. Post-partum hypercalcaemia in treated hypoparathyroidism. Br Med J. 1969 Jan 4;1(5635):23-5. doi: 10.1136/bmj.1.5635.23[↩]
- Zidar BL, Shadduck RK, Winkelstein A, Zeigler Z, Hawker CD. Acute myelobalstic leukemia and hypercalcemia. A case of probable ectopic parathyroid hormone production. N Engl J Med. 1976 Sep 23;295(13):692-4. doi: 10.1056/NEJM197609232951302[↩][↩]
- Koyama Y, Ishijima H, Ishibashi A, Katsuya T, Ishizaka H, Aoki J, Endo K. Intact PTH-producing hepatocellular carcinoma treated by transcatheter arterial embolization. Abdom Imaging. 1999 Mar-Apr;24(2):144-6. doi: 10.1007/s002619900463[↩][↩]
- Mahoney EJ, Monchik JM, Donatini G, De Lellis R. Life-threatening hypercalcemia from a hepatocellular carcinoma secreting intact parathyroid hormone: localization by sestamibi single-photon emission computed tomographic imaging. Endocr Pract. 2006 May-Jun;12(3):302-6. doi: 10.4158/EP.12.3.302[↩]
- Yoshimoto K, Yamasaki R, Sakai H, Tezuka U, Takahashi M, Iizuka M, Sekiya T, Saito S. Ectopic production of parathyroid hormone by small cell lung cancer in a patient with hypercalcemia. J Clin Endocrinol Metab. 1989 May;68(5):976-81. doi: 10.1210/jcem-68-5-976[↩]
- Nielsen PK, Rasmussen AK, Feldt-Rasmussen U, Brandt M, Christensen L, Olgaard K. Ectopic production of intact parathyroid hormone by a squamous cell lung carcinoma in vivo and in vitro. J Clin Endocrinol Metab. 1996 Oct;81(10):3793-6. doi: 10.1210/jcem.81.10.8855839[↩]
- Weiss ES, Doty J, Brock MV, Halvorson L, Yang SC. A case of ectopic parathyroid hormone production by a pulmonary neoplasm. J Thorac Cardiovasc Surg. 2006 Apr;131(4):923-4. doi: 10.1016/j.jtcvs.2005.12.020[↩]
- Ohira S, Itoh K, Shiozawa T, Horiuchi A, Ono K, Takeuchi H, Hosoda W, Konishi I. Ovarian non-small cell neuroendocrine carcinoma with paraneoplastic parathyroid hormone-related hypercalcemia. Int J Gynecol Pathol. 2004 Oct;23(4):393-7. doi: 10.1097/01.pgp.0000139655.18062.12[↩][↩]
- Nussbaum SR, Gaz RD, Arnold A. Hypercalcemia and ectopic secretion of parathyroid hormone by an ovarian carcinoma with rearrangement of the gene for parathyroid hormone. N Engl J Med. 1990 Nov 8;323(19):1324-8. doi: 10.1056/NEJM199011083231907[↩][↩]
- Chen L, Dinh TA, Haque A. Small cell carcinoma of the ovary with hypercalcemia and ectopic parathyroid hormone production. Arch Pathol Lab Med. 2005 Apr;129(4):531-3. doi: 10.5858/2005-129-531-SCCOTO[↩]
- Doyle MA, Malcolm JC. An unusual case of malignancy-related hypercalcemia. Int J Gen Med. 2013 Dec 6;7:21-7. doi: 10.2147/IJGM.S51302[↩][↩][↩]
- Vacher-Coponat H, Opris A, Denizot A, Dussol B, Berland Y. Hypercalcaemia induced by excessive parathyroid hormone secretion in a patient with a neuroendocrine tumour. Nephrol Dial Transplant. 2005 Dec;20(12):2832-5. doi: 10.1093/ndt/gfi065[↩][↩]
- Arps H, Dietel M, Schulz A, Janzarik H, Klöppel G. Pancreatic endocrine carcinoma with ectopic PTH-production and paraneoplastic hypercalcaemia. Virchows Arch A Pathol Anat Histopathol. 1986;408(5):497-503. doi: 10.1007/BF00705303[↩][↩][↩]
- Kandil E, Noureldine S, Khalek MA, Daroca P, Friedlander P. Ectopic secretion of parathyroid hormone in a neuroendocrine tumor: a case report and review of the literature. Int J Clin Exp Med. 2011;4(3):234-40. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3182517[↩][↩]
- Iguchi H, Miyagi C, Tomita K, Kawauchi S, Nozuka Y, Tsuneyoshi M, Wakasugi H. Hypercalcemia caused by ectopic production of parathyroid hormone in a patient with papillary adenocarcinoma of the thyroid gland. J Clin Endocrinol Metab. 1998 Aug;83(8):2653-7. doi: 10.1210/jcem.83.8.5025[↩]
- Samaan NA, Ordonez NG, Ibanez ML, Goepfert H, Smith K. Ectopic parathyroid hormone production by a squamous carcinoma of the tonsil. Arch Otolaryngol. 1983 Feb;109(2):91-4. doi: 10.1001/archotol.1983.00800160025006[↩]
- Belaid R, Oueslati I, Chihaoui M, Yazidi M, Grira W, Chaker F. A Case of Von Hippel-Lindau Disease with Bilateral Pheochromocytoma and Ectopic Hypersecretion of Intact Parathyroid Hormone in an Adolescent Girl. Case Rep Endocrinol. 2020 Aug 7;2020:8824640. doi: 10.1155/2020/8824640[↩]
- Wong K, Tsuda S, Mukai R, Sumida K, Arakaki R. Parathyroid hormone expression in a patient with metastatic nasopharyngeal rhabdomyosarcoma and hypercalcemia. Endocrine. 2005 Jun;27(1):83-6. doi: 10.1385/ENDO:27:1:083[↩]
- Rizzoli R, Pache JC, Didierjean L, Bürger A, Bonjour JP. A thymoma as a cause of true ectopic hyperparathyroidism. J Clin Endocrinol Metab. 1994 Sep;79(3):912-5. doi: 10.1210/jcem.79.3.8077382[↩]
- Gurrado A, Marzullo A, Lissidini G, Lippolis A, Rubini D, Lastilla G, Testini M. Substernal oxyphil parathyroid adenoma producing PTHrP with hypercalcemia and normal PTH level. World J Surg Oncol. 2008 Feb 21;6:24. doi: 10.1186/1477-7819-6-24[↩][↩]
- VanHouten JN, Yu N, Rimm D, Dotto J, Arnold A, Wysolmerski JJ, Udelsman R. Hypercalcemia of malignancy due to ectopic transactivation of the parathyroid hormone gene. J Clin Endocrinol Metab. 2006 Feb;91(2):580-3. doi: 10.1210/jc.2005-2095[↩][↩]
- Nakajima K, Tamai M, Okaniwa S, Nakamura Y, Kobayashi M, Niwa T, Horigome N, Ito N, Suzuki S, Nishio S, Komatsu M. Humoral hypercalcemia associated with gastric carcinoma secreting parathyroid hormone: a case report and review of the literature. Endocr J. 2013;60(5):557-62. doi: 10.1507/endocrj.ej12-0406[↩]
- Strewler GJ, Budayr AA, Clark OH, Nissenson RA. Production of parathyroid hormone by a malignant nonparathyroid tumor in a hypercalcemic patient. J Clin Endocrinol Metab. 1993 May;76(5):1373-5. doi: 10.1210/jcem.76.5.7684395[↩]
- Committee to Review Dietary Reference Intakes for Vitamin D and Calcium, Food and Nutrition Board, Institute of Medicine. Dietary Reference Intakes for Calcium and Vitamin D. Washington, DC: National Academy Press, 2010.[↩][↩]
- Dietary Guidelines for Americans. https://www.dietaryguidelines.gov[↩]
- U.S. Department of Agriculture, Center for Nutrition Policy and Promotion. ChooseMyPlate.gov 2011. https://www.myplate.gov[↩]
- Wongdee K, Rodrat M, Teerapornpuntakit J, Krishnamra N, Charoenphandhu N. Factors inhibiting intestinal calcium absorption: hormones and luminal factors that prevent excessive calcium uptake. J Physiol Sci. 2019 Sep;69(5):683-696. doi: 10.1007/s12576-019-00688-3[↩]
- Wikoff D, Welsh BT, Henderson R, Brorby GP, Britt J, Myers E, Goldberger J, Lieberman HR, O’Brien C, Peck J, Tenenbein M, Weaver C, Harvey S, Urban J, Doepker C. Systematic review of the potential adverse effects of caffeine consumption in healthy adults, pregnant women, adolescents, and children. Food Chem Toxicol. 2017 Nov;109(Pt 1):585-648. doi: 10.1016/j.fct.2017.04.002[↩]
- Gallagher JC, Yalamanchili V, Smith LM. The effect of vitamin D on calcium absorption in older women. J Clin Endocrinol Metab. 2012 Oct;97(10):3550-6. doi: 10.1210/jc.2012-2020[↩]
- Grubbs EG, Rafeeq S, Jimenez C, Feng L, Lee JE, Evans DB, Perrier ND. Preoperative vitamin D replacement therapy in primary hyperparathyroidism: safe and beneficial? Surgery. 2008 Dec;144(6):852-8; discussion 858-9. doi: 10.1016/j.surg.2008.06.032[↩]
- Vitamin D. https://ods.od.nih.gov/factsheets/VitaminD-HealthProfessional[↩]
- Acharya R, Winters DM, Rowe C, Buckley N, Kafle S, Chhetri B. An unusual case of severe hypercalcemia: as dehydrated as a bone. J Community Hosp Intern Med Perspect. 2021 Jan 26;11(1):135-138. doi: 10.1080/20009666.2020.1851859[↩]
- Pawar NH, Chiam PPS, Tan JHY, Loh J, Aw DC, Baikunje S. Acute Kidney Injury, Hypercalcemia, and Osteolytic Lesions: A Familiar Triad With a Rare Cause Complicated by Posterior Reversible Encephalopathy Syndrome. Am J Kidney Dis. 2017 Nov;70(5):A12-A15. doi: 10.1053/j.ajkd.2017.07.010[↩][↩]
- Ralston SH. Medical management of hypercalcaemia. Br J Clin Pharmacol. 1992 Jul;34(1):11-20. doi: 10.1111/j.1365-2125.1992.tb04101.x[↩][↩][↩][↩]
- Benabe JE, Martinez-Maldonado M. Hypercalcemic nephropathy. Arch Intern Med. 1978 May;138(5):777-9.[↩][↩]
- Insogna KL. Primary Hyperparathyroidism. N Engl J Med. 2018 Sep 13;379(11):1050-1059. doi: 10.1056/NEJMcp1714213[↩]
- Hypocalcemia Workup. https://emedicine.medscape.com/article/241893-workup#c11[↩][↩]
- Sempos CT, Heijboer AC, Bikle DD, Bollerslev J, Bouillon R, Brannon PM, DeLuca HF, Jones G, Munns CF, Bilezikian JP, Giustina A, Binkley N. Vitamin D assays and the definition of hypovitaminosis D: results from the First International Conference on Controversies in Vitamin D. Br J Clin Pharmacol. 2018 Oct;84(10):2194-2207. doi: 10.1111/bcp.13652[↩]
- LeFevre ML; U.S. Preventive Services Task Force. Screening for vitamin D deficiency in adults: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2015 Jan 20;162(2):133-40. doi: 10.7326/M14-2450[↩]
- Sempos CT, Binkley N. 25-Hydroxyvitamin D assay standardisation and vitamin D guidelines paralysis. Public Health Nutr. 2020 May;23(7):1153-1164. doi: 10.1017/S1368980019005251[↩]
- Brooks SPJ, Sempos CT. The importance of 25-hydroxyvitamin D assay standardization and the Vitamin D Standardization Program. Journal of AOAC International 2017;100:1223-4.[↩]
- Taylor CL, Sempos CT, Davis CD, Brannon PM. Vitamin D: Moving Forward to Address Emerging Science. Nutrients. 2017 Dec 1;9(12):1308. doi: 10.3390/nu912130[↩]
- Norman AW, Henry HH. Vitamin D. In: Erdman JW, Macdonald IA, Zeisel SH, eds. Present Knowledge in Nutrition, 10th ed. Washington DC: Wiley-Blackwell, 2012.[↩]
- Durant E, Singh A. ST elevation due to hypercalcemia. Am J Emerg Med. 2017 Jul;35(7):1033.e3-1033.e6. doi: 10.1016/j.ajem.2017.02.005[↩]
- Sonoda K, Watanabe H, Hisamatsu T, Ashihara T, Ohno S, Hayashi H, Horie M, Minamino T. High Frequency of Early Repolarization and Brugada-Type Electrocardiograms in Hypercalcemia. Ann Noninvasive Electrocardiol. 2016 Jan;21(1):30-40. doi: 10.1111/anec.12303[↩]
- Schutt RC, Bibawy J, Elnemr M, Lehnert AL, Putney D, Thomas AS, Barker CM, Pratt CM. Case report: Severe hypercalcemia mimicking ST-segment elevation myocardial infarction. Methodist Debakey Cardiovasc J. 2014 Jul-Sep;10(3):193-7. doi: 10.14797/mdcj-10-3-193[↩]
- Heller SR, Hosking DJ. Renal handling of calcium and sodium in metastatic and non-metastatic malignancy. Br Med J (Clin Res Ed). 1986 Mar 1;292(6520):583-6. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1339563/pdf/bmjcred00223-0015.pdf[↩][↩][↩]
- Meyer WJ 3rd, Transbol I, Bartter FC, Delea C. Control of calcium absorption: effect of sodium chloride loading and depletion. Metabolism. 1976 Sep;25(9):989-93. doi: 10.1016/0026-0495(76)90128-1[↩]
- Hoorn EJ. Intravenous fluids: balancing solutions. J Nephrol. 2017 Aug;30(4):485-492. doi: 10.1007/s40620-016-0363-9 Erratum in: J Nephrol. 2020 Apr;33(2):387.[↩]
- LeGrand SB, Leskuski D, Zama I. Narrative review: furosemide for hypercalcemia: an unproven yet common practice. Ann Intern Med. 2008 Aug 19;149(4):259-63. doi: 10.7326/0003-4819-149-4-200808190-00007[↩][↩]
- Saunders Y, Ross JR, Broadley KE, Edmonds PM, Patel S; Steering Group. Systematic review of bisphosphonates for hypercalcaemia of malignancy. Palliat Med. 2004 Jul;18(5):418-31. doi: 10.1191/0269216304pm914ra[↩][↩]
- Gallacher SJ, Ralston SH, Fraser WD, Dryburgh FJ, Cowan RA, Logue FC, Boyle IT. A comparison of low versus high dose pamidronate in cancer-associated hypercalcaemia. Bone Miner. 1991 Dec;15(3):249-56. doi: 10.1016/0169-6009(91)90130-r[↩]
- Body JJ, Magritte A, Seraj F, Sculier JP, Borkowski A. Aminohydroxypropylidene bisphosphonate (APD) treatment for tumor-associated hypercalcemia: a randomized comparison between a 3-day treatment and single 24-hour infusions. J Bone Miner Res. 1989 Dec;4(6):923-8. doi: 10.1002/jbmr.5650040617[↩]
- Nussbaum SR, Younger J, Vandepol CJ, Gagel RF, Zubler MA, Chapman R, Henderson IC, Mallette LE. Single-dose intravenous therapy with pamidronate for the treatment of hypercalcemia of malignancy: comparison of 30-, 60-, and 90-mg dosages. Am J Med. 1993 Sep;95(3):297-304. doi: 10.1016/0002-9343(93)90282-t[↩]
- Ahmad S, Kuraganti G, Steenkamp D. Hypercalcemic crisis: a clinical review. Am J Med. 2015 Mar;128(3):239-45. doi: 10.1016/j.amjmed.2014.09.030[↩][↩][↩]
- Clines GA. Mechanisms and treatment of hypercalcemia of malignancy. Curr Opin Endocrinol Diabetes Obes. 2011 Dec;18(6):339-46. doi: 10.1097/MED.0b013e32834b4401[↩][↩][↩]
- Major P, Lortholary A, Hon J, Abdi E, Mills G, Menssen HD, Yunus F, Bell R, Body J, Quebe-Fehling E, Seaman J. Zoledronic acid is superior to pamidronate in the treatment of hypercalcemia of malignancy: a pooled analysis of two randomized, controlled clinical trials. J Clin Oncol. 2001 Jan 15;19(2):558-67. doi: 10.1200/JCO.2001.19.2.558[↩][↩]
- Minisola S, Pepe J, Piemonte S, Cipriani C. The diagnosis and management of hypercalcaemia. BMJ. 2015 Jun 2;350:h2723. doi: 10.1136/bmj.h2723[↩]
- FDA Approves XGEVA® (denosumab) For The Prevention Of Skeletal-Related Events In Patients With Multiple Myeloma. https://www.amgen.com/newsroom/press-releases/2018/01/fda-approves-xgeva-denosumab-for-the-prevention-of-skeletalrelated-events-in-patients-with-multiple-myeloma[↩]
- FDA Approves New Indication for Denosumab (Xgeva). FDA Approves New Indication for Denosumab (Xgeva) – Medscape – Dec 09, 2014.[↩]
- Thosani S, Hu MI. Denosumab: a new agent in the management of hypercalcemia of malignancy. Future Oncol. 2015;11(21):2865-71. doi: 10.2217/fon.15.232[↩][↩]
- Bech A, de Boer H. Denosumab for tumor-induced hypercalcemia complicated by renal failure. Ann Intern Med. 2012 Jun 19;156(12):906-7. doi: 10.7326/0003-4819-156-12-201206190-00026[↩]
- Boikos SA, Hammers HJ. Denosumab for the treatment of bisphosphonate-refractory hypercalcemia. J Clin Oncol. 2012 Oct 10;30(29):e299. doi: 10.1200/JCO.2012.41.7923[↩]
- Adhikaree J, Newby Y, Sundar S. Denosumab should be the treatment of choice for bisphosphonate refractory hypercalcaemia of malignancy. BMJ Case Rep. 2014 Jan 30;2014:bcr2013202861. doi: 10.1136/bcr-2013-202861[↩]
- Freeman A, El-Amm J, Aragon-Ching JB. Use of denosumab for renal cell carcinoma-associated malignant hypercalcemia: a case report and review of the literature. Clin Genitourin Cancer. 2013 Dec;11(4):e24-6. doi: 10.1016/j.clgc.2013.06.002[↩]
- Dietzek A, Connelly K, Cotugno M, Bartel S, McDonnell AM. Denosumab in hypercalcemia of malignancy: a case series. J Oncol Pharm Pract. 2015 Apr;21(2):143-7. doi: 10.1177/1078155213518361[↩]
- Diel IJ, Body JJ, Stopeck AT, Vadhan-Raj S, Spencer A, Steger G, von Moos R, Goldwasser F, Feng A, Braun A. The role of denosumab in the prevention of hypercalcaemia of malignancy in cancer patients with metastatic bone disease. Eur J Cancer. 2015 Jul;51(11):1467-75. doi: 10.1016/j.ejca.2015.04.017[↩][↩]
- Polyzos SA, Makras P, Tournis S, Anastasilakis AD. Off-label uses of denosumab in metabolic bone diseases. Bone. 2019 Dec;129:115048. doi: 10.1016/j.bone.2019.115048[↩]
- Feldenzer KL, Sarno J. Hypercalcemia of Malignancy. J Adv Pract Oncol. 2018 Jul-Aug;9(5):496-504. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6505545/[↩]
- Mirrakhimov AE. Hypercalcemia of Malignancy: An Update on Pathogenesis and Management. N Am J Med Sci. 2015 Nov;7(11):483-93. doi: 10.4103/1947-2714.170600[↩]
- Sternlicht H, Glezerman IG. Hypercalcemia of malignancy and new treatment options. Ther Clin Risk Manag. 2015 Dec 4;11:1779-88. doi: 10.2147/TCRM.S83681[↩]
- Fallah-Rad N, Morton AR. Managing hypercalcaemia and hypocalcaemia in cancer patients. Curr Opin Support Palliat Care. 2013 Sep;7(3):265-71. doi: 10.1097/SPC.0b013e3283640f5f[↩]
- Rosner MH, Dalkin AC. Onco-nephrology: the pathophysiology and treatment of malignancy-associated hypercalcemia. Clin J Am Soc Nephrol. 2012 Oct;7(10):1722-9. doi: 10.2215/CJN.02470312[↩]
- Basok AB, Rogachev B, Haviv YS, Vorobiov M. Treatment of extreme hypercalcaemia: the role of haemodialysis. BMJ Case Rep. 2018 Jun 4;2018:bcr2017223772. doi: 10.1136/bcr-2017-223772[↩]
- Loh HH, Mohd Noor N. The use of hemodialysis in refractory hypercalcemia secondary to parathyroid carcinoma. Case Rep Crit Care. 2014;2014:140906. doi: 10.1155/2014/140906[↩]
- Callender GG, Udelsman R. Surgery for primary hyperparathyroidism. Cancer. 2014 Dec 1;120(23):3602-16. doi: 10.1002/cncr.28891[↩]