What is ammonia
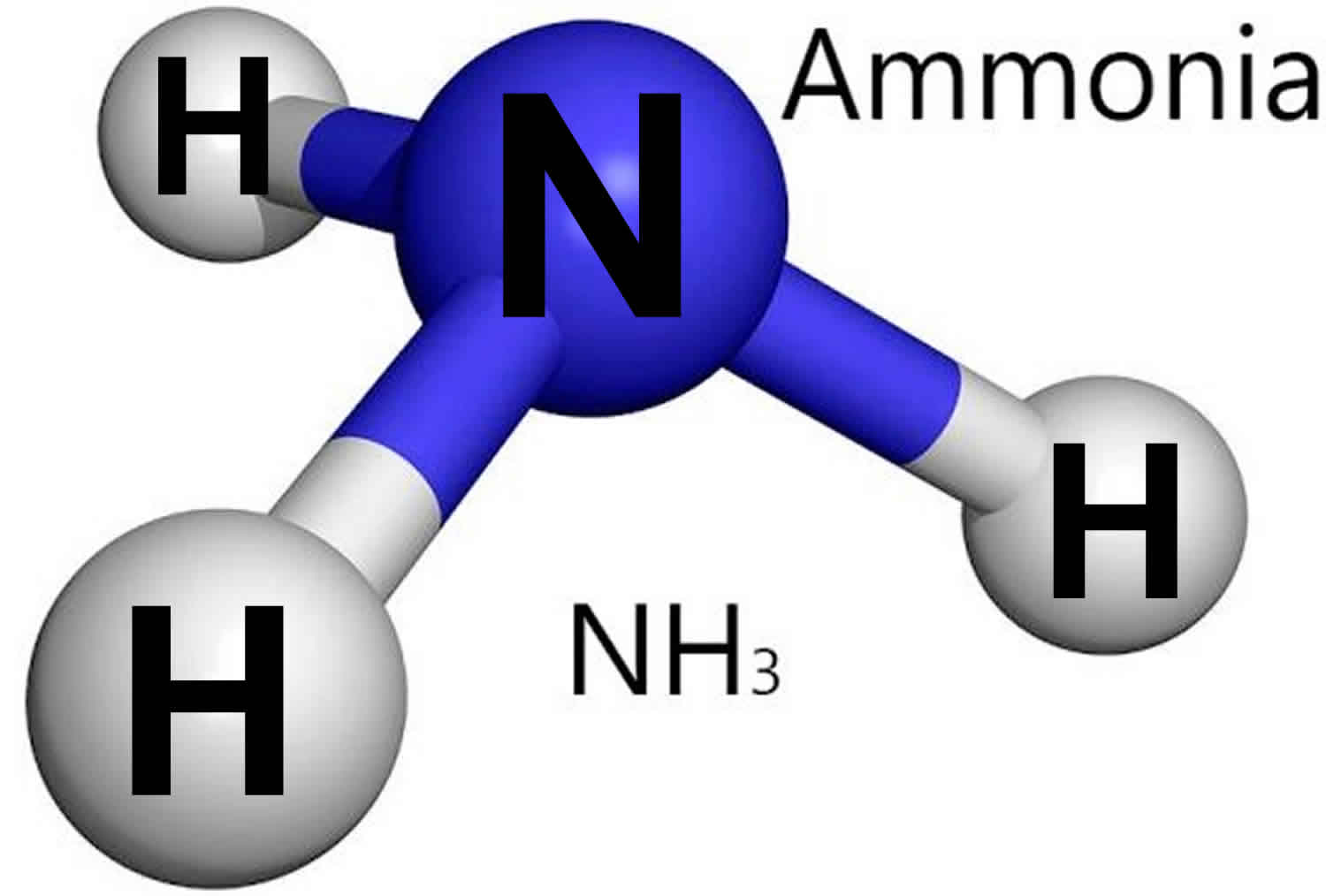
Ammonia (NH3) is a colorless acrid-smelling gas at room temperature and normal atmospheric pressure that occurs naturally in the environment, in the air, soil, and water and in plants and animals including humans. Bacteria found in the intestines can produce ammonia. Ammonia (NH3) is also produced by human activity. In nature, most ammonia probably comes from decomposing animal excreta, with the decay of organic materials from plants, dead animals, and the like also contributing significant amounts. Ammonia is also exhaled by animals. Ammonia (NH3) is an important source of nitrogen which is needed by plants and animals. Ammonia is applied directly into soil on farm fields, and is used to make fertilizers for farm crops, lawns, and plants. Many household and industrial cleaners contain ammonia.
Most people can identify ammonia very distinct odor at 35 mg/m³ in air. This odor is familiar to many people because ammonia is used in smelling salts, many household and industrial cleaners, and window-cleaning products. Ammonia can be stored and transported as a liquid at a pressure of 10 atmospheres at 25°C (77 °F). Spilled liquid ammonia boils immediately, cooling its surroundings as it vaporizes. Ammonia gas dissolves readily in water; in solution it forms liquid ammonia or aqueous ammonia and is in equilibrium with ammonium ions (NH4+). Ammonia solutions are alkaline and react with acids to form ammonium salts. Once exposed to open air, liquid ammonia quickly turns into a gas.
Gaseous ammonia is usually very pure and the purity of solutions depends on the purity of the water used, e.g., chlorinated water may give rise to chloramines and nitrogen trichloride.
Ammonia is released to the atmosphere by natural processes such as the decay of organic matter and animal excreta, or by volcanic eruptions. Ammonia can also be released to the atmosphere by human activities such as fertilizer use; spillage or leakage from storage or production facilities; or loss from waste water effluents. The average global ammonia concentration in the atmosphere ranges from 0.3 to 6 parts of ammonia per one billion parts of air (ppb), with concentrations sometimes higher in the vicinity of agricultural or industrial areas. For example, near industrial sources or manure heaps in Germany, ammonia concentrations ranged from 10.3 to 89 ppb 1. Concentrations may be orders of magnitude higher near some types of livestock areas, such as pigpens, where local atmospheric concentrations have been reported to be as high as 47 parts of ammonia per one million parts of air (ppm).
Exposure to high levels of ammonia can cause irritation and serious burns on the skin and in the mouth, throat, lungs, and eyes. At very high levels, ammonia can even cause death. The Occupational Safety and Health Administration (OSHA) has set an acceptable eight-hour exposure limit at 25 parts of ammonia per one million parts of air (ppm) and a short-term (15 minutes) exposure level at 35 ppm.
Elevated concentrations of ammonia in water are usually due to effluent discharges from sewage treatment plants or industrial processes, or runoff from fertilized fields or livestock areas. Ammonia concentrations can therefore vary widely in aquatic environments, with concentrations being lower in bodies of water that are unimpacted by residential, industrial, or farming effluents, compared to those that are impacted (where concentrations can be orders of magnitude higher). In unimpacted waterways, ammonia concentrations have been reported to range from 8.5 to 43 ppb, whereas in impacted waterways, concentrations as high as 16 ppm have been reported.
Soils usually obtain additional ammonia from natural or synthetic fertilizer application, animal excreta, decaying organic matter, or natural fixation from the atmosphere. Soils have been reported to have background concentrations of ammonia ranging from 1 to 5 ppm. Immediately following application of fertilizer or manure, however, ammonia concentrations can rise to 2–3,000 ppm, with levels dropping after 5 days to 2–850 ppm. These high ammonia concentrations are usually limited to the upper few centimeters of topsoil.
For the general population, the most likely source of exposure to elevated levels of ammonia is from the use of household cleaners containing ammonia or ammonium salts. People who live near farms, who visit farms during the application of fertilizer, or who live near cattle feedlots, poultry confinement buildings, or other areas where animal populations are concentrated can also be exposed to ammonia. Local atmospheric concentrations in these agricultural settings have been reported to range from 280 to 88,000 ppb.
There is also the possibility for exposure to ammonia via water and food ingestion. If untreated surface water is ingested, the average uptake would be 0.36 mg/day (assuming an ammonia concentration in untreated water of 0.18 mg/L and a consumption rate of 2 L/day). For most sources of drinking water, however, adsorption, nitrification, and the conversion of ammonia to chloramines upon chlorination will result in negligible levels of ammonia in most drinking water supplies. Food ingestion can also lead to an exposure to ammonia, primarily due to the use of various ammonium salts as food stabilizers; the estimated exposure from these food additives is 18 mg/day.
Populations that live or work near a hazardous waste site that contains ammonia or ammonium salts could be exposed to above-average levels of ammonia in soil, water, or air in similar concentrations as those in agricultural settings. While these exposures may occur, the half-life of ammonia in nature is probably
very short.
What happens to ammonia when it enters the environment?
- Ammonia is found throughout the environment in air, water, soil, animals, and plants.
- Ammonia does not last very long in the environment. It is rapidly taken up by plants, bacteria, and animals.
- Ammonia does not build up in the food chain, but serves as a nutrient for plants and bacteria.
How might I be exposed to ammonia?
- Everyone is exposed to low levels of naturally-occurring ammonia in air, food, water, and soil.
- You may be exposed to higher levels during use of cleaning products containing ammonia.
- You may be exposed to higher levels if you apply ammonia fertilizers or live near farms where these fertilizers have been applied.
- You may be exposed to high levels if you go into enclosed buildings that contain lots of animals (such as on farms).
How can ammonia affect my health?
No health effects have been found in humans exposed to typical environmental concentrations of ammonia. Exposure to high levels of ammonia in air may be irritating to your skin, eyes, throat, and lungs and cause coughing and burns. Lung damage and death may occur after exposure to very high concentrations of ammonia. Some people with asthma may be more sensitive to breathing ammonia than others.
Swallowing concentrated solutions of ammonia can cause burns in your mouth, throat, and stomach. Splashing ammonia into your eyes can cause burns and even blindness.
How likely is ammonia to cause cancer?
There is no evidence that ammonia causes cancer. The Department of Health and Human Services (DHHS), the Environmental Protection Agency (EPA), and the International Agency for Research on Cancer (IARC), have not classified ammonia for carcinogenicity.
How can ammonia affect children?
Children are less likely than adults to be exposed to concentrated levels of ammonia because most exposures occur at work. The effects on children are likely to be the same as for adults. We do not know if exposure to ammonia causes birth defects, or if it can pass to the fetus across the placenta or to infants via breast milk.
How can families reduce the risk of exposure to ammonia?
- Keep products that contain ammonia out of the reach of children.
- Make sure there is adequate ventilation when you use cleaners that contain ammonia, and wear proper clothing and eye protection.
- Never store cleaning solutions in containers that children might find attractive, like soda bottles.
- Avoid farm fields after they have been treated with ammonia or ammonia-containing fertilizers.
- Minimize exposure to ammonia in the workplace by wearing proper safety clothes and equipment, and by following safety rules.
You can reduce your risk of exposure to ammonia by carefully using household products and by avoiding areas where ammonia is used or produced. At home, you can reduce your risk of exposure to ammonia by careful handling of any household products that contain ammonia. For example, some cleaning products contain ammonia; so when you use them, you should be sure that rooms are adequately ventilated during the time you are using them. Avoid ammonia-containing products in glass bottles since breakage could lead to a serious exposure. You should wear proper clothing and eye protection, because ammonia can cause skin burns and damage eyes if it is splashed on them. To lower the risk of your children being exposed to ammonia, you should tell them to stay out of the room when you are using it. While use of ammonia by a child is not recommended, any use by a child should be closely supervised by an adult.
You can also reduce your risk of exposure to ammonia by avoiding areas where it is being used. Ammonia is used to fertilize crops, so you can lower your exposure to ammonia by avoiding these areas when it is being applied. You can also lower your exposure to ammonia by avoiding places where it is produced. Ammonia is found in many animal wastes, and it may be present in high concentrations in the air in livestock buildings. You can lower your exposure to ammonia by avoiding these buildings, especially if large numbers of animals are inside.
If you are a worker who uses or applies ammonia for farming, you can reduce your exposure by using it according to the instructions and wearing proper clothing and protective gear. Be sure to follow all instructions and heed any warning statements.
Is there a medical test to show whether I’ve been exposed to ammonia?
There are tests to measure ammonia in blood and urine. These tests can not definitely determine whether you have been exposed because ammonia is normally found in your bodies.
What is anhydrous ammonia
Anhydrous ammonia, a colorless gas with pungent, suffocating fumes, is used as an agricultural fertilizer and industrial refrigerant.
When handled improperly, anhydrous ammonia can be immediately dangerous to life or health. As liquid anhydrous ammonia is released from its container into the air, it expands rapidly, forming a large cloud that acts like a heavier-than-air gas for a period of time. Because the vapors hug the ground initially, the chances for humans to be exposed are greater than with other gases. Symptoms of anhydrous ammonia exposure include:
- eye, nose, and throat irritation
- breathing difficulty, wheezing, or chest pain
- pulmonary edema, pink frothy sputum
- burns, blisters and frostbite.
Exposure to anhydrous ammoniacan be fatal at high concentrations.
Anhydrous ammonia is also a key ingredient in the production of methamphetamine (meth), an illicit activity frequently occurring in makeshift laboratories. Because states require a license for purchase of anhydrous ammonia, those involved in illicit production of meth often resort to stealing it from areas where it is stored and used (e.g., farms, industrial refrigeration systems, and railroad tanker cars). These thefts often lead to releases when valves are left open as ammonia is being siphoned, ammonia is transferred inappropriately into makeshift containers, plugs are removed from ammonia lines at refrigeration facilities or the wrong hoses or fittings are attached to storage containers. This creates a health hazard for anyone in the proximity including emergency health responders.
Anhydrous ammonia quick facts
- Increased exposure risk can result from anhydrous ammonia leaks at storage facilities or from means of transportation. These leaks may require evacuation and the use of emergency personnel and procedures. Respiratory distress and skin and eye irritation may result from anhydrous ammonia exposure and high levels of exposure may result in death. Advance safety preparation and precautions and/or rapid emergency response can lessen the possibility of serious injury.
- Although most anhydrous ammonia is used for legitimate purposes, a small percentage is diverted to meth manufacturing. Since meth production is illegal, meth “cookers” may attempt to obtain a supply of ammonia by stealing it from storage facilities. This theft can result in the release of ammonia into the air due to removal of plugs or opening of valves during the transfer process or the use of inadequate containers, hoses or fittings. This presents a dangerous risk of exposure not only to those involved in the theft, but also to the emergency responders or innocent bystanders.
Who’s at risk of anhydrous ammonia?
The use of chemical fertilizer containing anhydrous ammonia can lead to exposure. Outdoor exposure to high levels of anhydrous ammonia can result from leaks and spills at production plants and storage facilities, and from pipelines, tank trucks, railcars, or ships and barges that transport ammonia.
Another possible exposure to anhydrous ammonia results from the theft of ammonia by “cookers” of methamphetamines. Of the 40,349 events reported to the Hazardous Substances Emergency Events Surveillance (HSEES) system from January 1, 2000 to June 30, 2004, a total of 1,791 (4%) were associated with illicit meth production. Of these 1,791 events at least 164 were known to have been caused by anhydrous ammonia theft for the production of methamphetamines. These 164 events resulted in 85 injured persons, mostly members of the general public and police officers. The most frequent injuries were respiratory distress and eye irritation. However, these numbers are not completely representative of the number of incidents or injuries, since only 16 states contributed data to HSEES during that time period and reporting is not mandatory for all events. In addition, law enforcement officials might not report events that could jeopardize criminal investigations.
How can anhydrous ammonia risk be reduced?
The best ways to reduce risk of serious injury from anhydrous ammonia exposure are to wear protective equipment and to know what to do in an emergency. Before entering an anhydrous ammonia release zone, emergency responders should select the proper personal protective equipment (PPE) including positive-pressure, self-contained breathing apparatus and chemical-protective clothing. The best first aid treatment for anhydrous ammonia exposure is to flush skin and eyes with water for at least 15 minutes.
Those who work with anhydrous ammonia or who respond to an ammonia release can take the following precautions:
Always keep 5 gallons of clean water in your supply tank and carry a small squeeze bottle.
Understand first aid treatment and practice what you would do in an emergency.
Wear ventless goggles, rubber gloves, and a long-sleeved shirt when working with anhydrous ammonia.
Regularly inspect equipment and have worn hoses and valves replaced.
Never allow bystanders in the area where anhydrous ammonia is being transferred or applied.
Review instructions before coupling and uncoupling lines.
What is ammonia used for?
The largest and most significant use of ammonia and ammonium compounds is the agricultural application of fertilizers. Ammonia and ammonium compounds used as fertilizer represent 89–90% of the commercially produced ammonia, with plastics, synthetic fibers and resins, explosives, and other uses accounting for most of the remainder 2. Direct uses of ammonia as fertilizer can be broken down into the following categories (percentages based on mass of nitrogen in each compound): anhydrous ammonia, 26%; urea/ammonium nitrate solutions, 23%; urea, 20%; ammonium nitrate, 4.5%;
ammonium sulfate, 2%; other forms, 3%; and multiple nutrient forms, 21% 3. Most ammonium compounds and nitric acid, which are produced from anhydrous ammonia, are used directly in the production of fertilizers.
The small proportion of commercially produced ammonia not incorporated into fertilizers is used as a corrosion inhibitor, in the purification of water supplies, as a component of household cleaners, and as a refrigerant. It is also used in the pulp and paper, metallurgy, rubber, food and beverage, textile, and leather industries. Ammonia is used in the manufacture of pharmaceuticals and explosives, and in the production of various chemical intermediates 4.
Ammonia levels
Ammonia is an essential mammalian waste product for DNA, RNA, and protein synthesis and is necessary for maintaining acid-base balance. Ammonia is produced and used endogenously in all mammalian species. Ammonia is a waste product formed primarily by bacteria in the intestines during the digestion of protein. If not processed and cleared from the body appropriately, excess ammonia can accumulate in the blood. It has been estimated that up to 17 grams of ammonia are produced in humans daily. Of these 17 grams, approximately 4 grams are produced in the gut by intestinal bacteria, where it enters the portal
circulation and is metabolized rapidly in the liver to urea. Ammonia is excreted primarily as urea and urinary ammonium compounds through the kidneys. Levels of ammonia in the blood from healthy humans range from 0.7 to 2 mg/L.
Ammonia is normally transported in the blood to the liver, where it is converted into two substances called urea and glutamine. The urea is then carried to the kidneys, where it is eliminated in the urine. If this “urea cycle” does not complete the breakdown of ammonia, ammonia builds up in the blood and can pass from the blood into the brain.
Ammonia is toxic to the brain. For example, when liver function is significantly reduced due to disorders such as cirrhosis or hepatitis, ammonia and other compounds processed by the liver can accumulate in the brain and cause a condition called hepatic encephalopathy.
Hepatic encephalopathy causes mental and neurological changes that can lead to confusion, disorientation, sleepiness, and eventually to coma and even death.
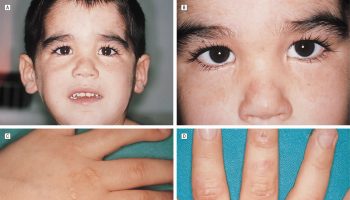
Infants and children with increased ammonia levels may vomit frequently, be irritable, and be increasingly lethargic. Left untreated, they may experience seizures, have difficulty breathing, and may lapse into a coma.
Problems with ammonia processing can arise from conditions such as:
- Severe liver disease – damage limits the ability of the liver to process ammonia; spikes in ammonia blood levels may be seen in people with stable liver disease, especially following a triggering event such as gastrointestinal bleeding or an electrolyte imbalance.
- Decreased blood flow to the liver – ammonia is less able to get to the liver to be processed.
- Reye syndrome – a rare condition that affects the blood, brain, and liver; it is characterized by a rise in ammonia levels and a fall in glucose. It affects primarily children and young adults. In most cases, it follows and appears to be triggered by a viral infection, such as the flu or chickenpox. Children who are given aspirin are at an increased risk.
- Renal failure – the kidneys are unable to effectively rid the body of urea, leading to a build-up of ammonia in the blood.
- Rare inherited defects in the urea cycle – a deficiency or defect in one or more of the enzymes necessary to complete the conversion of ammonia to urea.
Ammonia blood test
The ammonia test is used to detect an elevated level in the blood that can be caused by severe liver disease, kidney failure, Reye syndrome or a rare genetic disorder of the urea cycle, for example. The test may be used to help investigate the cause of an individual’s changes in behavior and consciousness. However, ammonia tests would probably not tell you whether you have been exposed to harmful amounts of ammonia, because ammonia is normally found in your body. If you were exposed to harmful amounts of ammonia, you would notice it immediately because of the strong, unpleasant, and irritating smell, the strong taste, and because of skin, eye, nose, or throat irritation.
Ammonia is a waste product naturally produced in your body. Ammonia primarily comes from the digestion of protein by bacteria in the intestines. If not processed by the liver and cleared from the body appropriately, excess ammonia can accumulate in the blood and pass from the blood into the brain, where it is toxic.
An ammonia test may be ordered, along with other tests such as glucose, electrolytes, and kidney and liver function tests, to help diagnose the cause of a coma or to help support the diagnosis of Reye syndrome or hepatic encephalopathy caused by various liver diseases. An ammonia level may also be ordered to help diagnose a rare urea cycle defect and to evaluate the severity of the condition.
Some healthcare practitioners use the ammonia test to monitor the effectiveness of treatment of hepatic encephalopathy, but there is not widespread agreement on its clinical utility. Since hepatic encephalopathy can be caused by the build-up of a variety of toxins in the blood and brain, blood ammonia levels correlate poorly with the severity of the condition.
A normal blood ammonia level may mean that a person’s signs and symptoms are due to a cause other than excess ammonia. However, normal concentrations of ammonia do not rule out hepatic encephalopathy. Other wastes can contribute to changes in mental function and consciousness, and brain levels of ammonia may be much higher than blood levels. This can make correlation of a person’s symptoms to ammonia blood levels difficult.
A decreased level of ammonia may be seen with some types of hypertension, such as essential hypertension (high blood pressure of unknown cause) and malignant hypertension (very high blood pressure that occurs suddenly and quickly).
Decreased levels of ammonia may also be seen with the use of some antibiotics, such as neomycin.
Ammonia tests can also be performed on arterial blood, but this method is much less frequently used. Some healthcare practitioners feel that arterial ammonia measurements are more clinically useful, but there is not widespread agreement on this.
When is ammonia blood test ordered?
An ammonia test may be ordered when a newborn has symptoms that arise in the first few days after birth, such as:
- Irritability
- Vomiting
- Lethargy
- Seizures
An ammonia test may be performed when a child develops these same symptoms about a week following a viral illness, such as influenza or chickenpox, or when a healthcare practitioner suspects that the child may have Reye syndrome.
When adults experience mental changes, disorientation, sleepiness, or lapse into a coma and may have liver disease or kidney failure, an ammonia level may be ordered to help evaluate the cause of the change in consciousness. In people with stable liver disease, an ammonia level may be ordered, along with other liver function tests, when a person suddenly becomes more acutely ill.
Is ammonia testing used to detect or monitor ammonia poisoning?
Not generally. In most cases, ammonia acts locally, burning or irritating whatever it comes in contact with, but according to the Agency for Toxic Substances and Disease Registry, ammonia does not usually act as a systemic poison. Concentrated commercial ammonia, in a liquid or vapor form, can cause more severe burns than the more dilute liquid household ammonia, but both can damage the eyes, skin, respiratory tract, and, if swallowed, the mouth, throat, and stomach.
What does high ammonia levels mean?
A significantly increased concentration of ammonia in the blood indicates that the body is not effectively processing and eliminating ammonia and it may be the cause of the person’s signs and symptoms.
In infants, an extremely high level is associated with an inherited urea cycle enzyme deficiency or defect but may also be seen with hemolytic disease of the newborn. Moderate short-lived increases in ammonia are relatively common in newborns, where the level may rise and fall without causing noticeable symptoms. (For more about urea cycle disorders, see the Related Pages tab.)
An increased ammonia level and decreased glucose level may indicate the presence of Reye syndrome in symptomatic children and teens. An increased concentration may also indicate a previously undiagnosed enzymatic defect of the urea cycle.
In children and adults, an elevated ammonia level may indicate that severe liver or kidney damage has impacted the body’s ability to clear ammonia and that the brain may be affected. Frequently, an acute or chronic illness will act as a trigger, increasing ammonia levels to the point that an affected person has difficulty clearing the ammonia.
High ammonia levels may also be seen with:
- Gastrointestinal bleeding – blood cells are hemolyzed in the intestines, releasing protein.
- Muscular exertion – muscles produce ammonia when active and absorb it when resting.
- Tourniquet use – ammonia levels can be increased in the blood sample collected.
- Use of certain drugs, including alcohol, barbiturates, diuretics, valproic acid, and narcotics
- Cigarette smoking
When my increased ammonia level has returned to normal, can it rise again?
It depends on why it was increased in the first place. If you had a temporary condition, then it is likely that the ammonia level will continue to be normal. If you have a chronic condition, then it is possible that it will increase again and your health status will likely need to be monitored. Talk to your healthcare provider about what is best for you.
Ammonia sickness
Ammonia (NH3) is found throughout the environment in the air, soil, and water, and in plants and animals, including humans. Ammonia is also found in many household and industrial cleaners. Ammonia is a corrosive substance and the main toxic effects are restricted to the sites of direct contact with ammonia (i.e., skin, eyes, respiratory tract, mouth, and digestive tract) 5. High levels of ammonia can irritate and burn the skin, mouth, throat, lungs, and eyes. Very high levels of ammonia can damage the lungs or cause death. Workers may be harmed from exposure to ammonia. The level of exposure depends upon dose, duration, and work being done. For example, if you spilled a bottle of concentrated ammonia on the floor, you would smell a strong ammonia odor; you might cough, and your eyes might water because of irritation. If you were exposed to very high levels of ammonia, you would experience more harmful effects. For example, if you walked into a dense cloud of ammonia or if your skin comes in contact with concentrated ammonia, your skin, eyes, throat, or lungs may be severely burned. These burns might be serious enough to cause permanent blindness, lung disease, or death. Likewise, if you accidentally ate or drank concentrated ammonia, you might experience burns in your mouth, throat, and stomach. There is no evidence that ammonia causes cancer. Ammonia has not been classified for carcinogenic effects by Environmental Protection Agency (EPA), the Department of Health and Human Services (DHHS), or the International Agency for Research on Cancer (IARC). Ammonia can also have beneficial effects, such as when it is used as a smelling salt. Certain ammonium salts have long been used in veterinary and human medicine.
Ammonia is used in many industries. Some examples of workers at risk of being exposed to ammonia include the following:
- Agricultural workers who use soil fertilizer
- Industrial workers who manufacture fertilizers, rubber, nitric acid, urea, plastics, fibers, synthetic resin, solvents and other chemicals
- Miners and metallurgic workers
- Workers in petroleum refining
- Workers who use a commercial refrigerant in food processing, produce ice, are near cold storage and de-icing operations.
Children are less likely than adults to be exposed to concentrated ammonia because most exposures to concentrated ammonia occur in occupational settings. Children can still be exposed in the same way as adults to ammonia gas from spills or leaks from ammonia tanks or pipelines, especially on farms where it is used as a fertilizer. Children can also be exposed to dilute ammonia solutions from household cleaners containing ammonia.
The effects of ammonia on children are likely to be the same as for adults. Ammonia is an irritant and the solution and gas can cause burns of the skin, eyes, mouth, and lungs. If a spill occurs, children may be exposed to ammonia for a longer time than adults because they may not leave the area as quickly.
There is no evidence that exposure to the levels of ammonia found in the environment causes birth defects or other developmental effects. It is not known whether ammonia can be transferred from a pregnant mother to a developing fetus through the placenta or from a nursing mother to her offspring through breast milk. One study in animals showed that exposure of mothers to very high levels of ammonia during pregnancy caused their newborn offspring to be smaller than normal, but this occurred at levels of ammonia that also affected the mothers.
The most likely and significant effects of exposure to elevated levels of ammonia are discussed below.
Ammonia summary of health effects
Respiratory Effects
Ammonia is an upper respiratory irritant in humans. Exposures to levels exceeding 50 ppm result in immediate irritation to the nose and throat; however, tolerance appears to develop with repeated exposure. Exposure to an air concentration of 250 ppm is bearable for most persons for 30–60 minutes. Acute exposure to higher levels (500 ppm) have been shown to increase respiratory minute volume. Accidental exposures to concentrated aerosols of ammonium salts or high concentrations of ammonia gas have resulted in nasopharyngeal and tracheal burns, airway obstruction and respiratory distress, and bronchiolar and alveolar edema. Ammonia vapor readily dissolves in the moisture present on the skin, eyes, oropharynx and lungs forming ammonium hydroxide which dissociates to yield hydroxyl ions. Chronic occupational exposure to low levels of airborne ammonia (<25 ppm) had little effect on pulmonary function or odor sensitivity in workers at some factories, but studies of farmers exposed to ammonia and other pollutants in livestock buildings indicated an association between exposure to pollutants, including ammonia, and an increase in respiratory symptoms (such as bronchial reactivity/hyperresponsiveness, inflammation, cough, wheezing, or shortness of breath) and/or a decrease in lung function parameters. The contribution of ammonia to these respiratory symptoms is unclear.
Dermal Effects
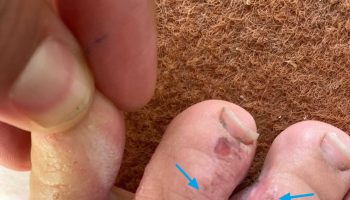
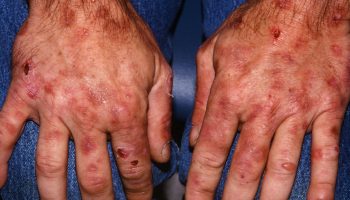
Skin is extremely sensitive to airborne ammonia or ammonia dissolved in water. The topical damage caused by ammonia is probably due mainly to its reactivity and irritation properties. Its high water solubility allows it to dissolve in moisture on these surfaces, react with fatty substances, be absorbed into deeper layers, and inflict extensive damage. Reports of skin damage in humans are numerous, but good quantitative data are lacking. The severity of the damage is proportional to the concentration and duration of exposure; flushing with water immediately after contact alleviates or prevents effects.
Dermal exposures to liquid ammonia or concentrated solutions and/or ammonia gas are frequently occupationally related and produce cutaneous burns, blisters, and lesions of varying degrees of severity. Unlike acid burns, which cause a coagulation necrosis, ammonia causes alkali burns, resulting in liquification of the tissue and deeper penetrations. Burns can be severe enough to require skin grafting, and loss of the epidermal layer increases body fluid loss and incidence of infection. While most ammonia exposures are occupational, household products containing ammonia can also cause dermal injury. Several cases of young children (2–3 years old) who bit into ammonia pellets/capsules and sustained oral and esophageal lesions have been reported in the literature.
Very limited animal data regarding dermal effects of exposure to ammonia support the findings in humans.
Ocular Effects
Reported ocular effects in humans following ammonia gas exposure increased in severity with dose and duration. Good quantitative data are lacking, but symptoms progress as follows: inflamed eyes, lacrimation, swelling of the eyelids, hyperemic conjunctiva, blurred vision, possible transient blindness, corneal abrasions, and sustained corneal damage. Ammonia is slightly irritating to human eyes in a brief exposure at concentrations of 100 ppm, and immediately irritating to the eyes and throat at 698 ppm. Exposure to an air concentration of 250 ppm is bearable for most persons for 30– 60 minutes.
Limited animal data regarding ocular effects of exposure to ammonia support the findings in humans.
Neurological Effects
Neurological effects in humans following inhalation or dermal exposure to ammonia are usually limited to blurred vision, most likely due to direct contact, but more severe exposures, which result in significant elevation of blood ammonia levels (hyperammonemia), can result in diffuse nonspecific encephalopathy, muscle weakness, decreased deep tendon reflexes, and loss of consciousness. Hyperammonemia in humans can result from certain disease states such as cirrhosis of the liver, acute liver failure, and congenital deficiencies of any of the urea cycle enzymes; hyperammonemia may lead to encephalopathy. Some have suggested that ammonia may be involved in the generation of the symptomatology and progression of Alzheimer’s disease as a result of pathological ammonia metabolism in the brain. Cerebral edema and herniation and intracranial hypertension have been noted in animal models of hyperammonemia. The mechanism of ammonia-induced encephalopathies has not been definitively elucidated. It is thought to involve the alteration of glutamate metabolism in the brain with resultant increased activation of N-methyl-D-aspartate (NMDA) receptors, which causes decreased protein kinase C-mediated phosphorylation of Na+/K+ ATPase, increased activity of Na+/K+ ATPase, and depletion of ATP. Additional evidence of altered energy levels includes changes in some TCA cycle-associated components including acetoacetate, and NAD+/NADH ratio, 2-oxoglutarate, and 3-hydroxybutarate. This reduced ATP level may be involved in ammonia-induced coma and death. A disruption in neurotransmission has also been suggested by alteration of brain tubulin, which is an essential component of the axonal transport system.
- https://www.atsdr.cdc.gov/toxprofiles/tp126-c2.pdf[↩]
- Kramer DA. 2004. Nitrogen (fixed)–ammonia. U.S. Geological survey, mineral commodity summaries. http://minerals.usgs.gov/minerals/pubs/commodity/nitrogen/nitromcs04.pdf[↩]
- Kramer DA. 2003. Nitrogen. U.S. Geological survey minerals yearbook. http://minerals.usgs.gov/minerals/pubs/commodity/nitrogen/nitromyb02.pdf[↩]
- Sax NI, Lewis RJ, eds. 1987. Hawley’s condensed chemical dictionary. New York, NY: Van Nostrand Reinhold, 1288[↩]
- Toxic Substances Portal – Ammonia. Public Health Statement for Ammonia. https://www.atsdr.cdc.gov/phs/phs.asp?id=9&tid=2[↩]