Focal nodular hyperplasia
Focal nodular hyperplasia also called FNH, is a regenerative mass lesion of the liver and the second most common benign liver lesion (most common is a hemangioma) 1. In 1958, pathologist, Hugh Edmondson, MD, first described focal nodular hyperplasia (FNH) as a solid, benign hepatic mass of non-vascular origin 2. Unlike the most common liver mass which is the hemangioma, focal nodular hyperplasia is thought to be the result of increased hepatocyte number caused by hypoperfusion or hyperperfusion from anomalous arteries within the hepatic lobule 3. The majority of focal nodular hyperplasia is solitary (80%), usually smaller than 5 cm, and occurs near the liver surface 4. The isolated occurrence is the commonest but in up to 20% focal nodular hyperplasia may also be multiple (multiple FNH syndrome) and could occur with other benign nodules such as hemangiomas or adenoma. Clinical course is asymptomatic in the slightly majority of cases, as the lesion is often discovered incidentally or as a palpable abdominal mass on physical examination. The widespread use of both ultrasounds (US) and computed tomography (CT) in the clinical practice has increased the detection rate of focal nodular hyperplasia. Focal nodular hyperplasia often is confounded by comorbid conditions making establishing a diagnosis and management difficult.
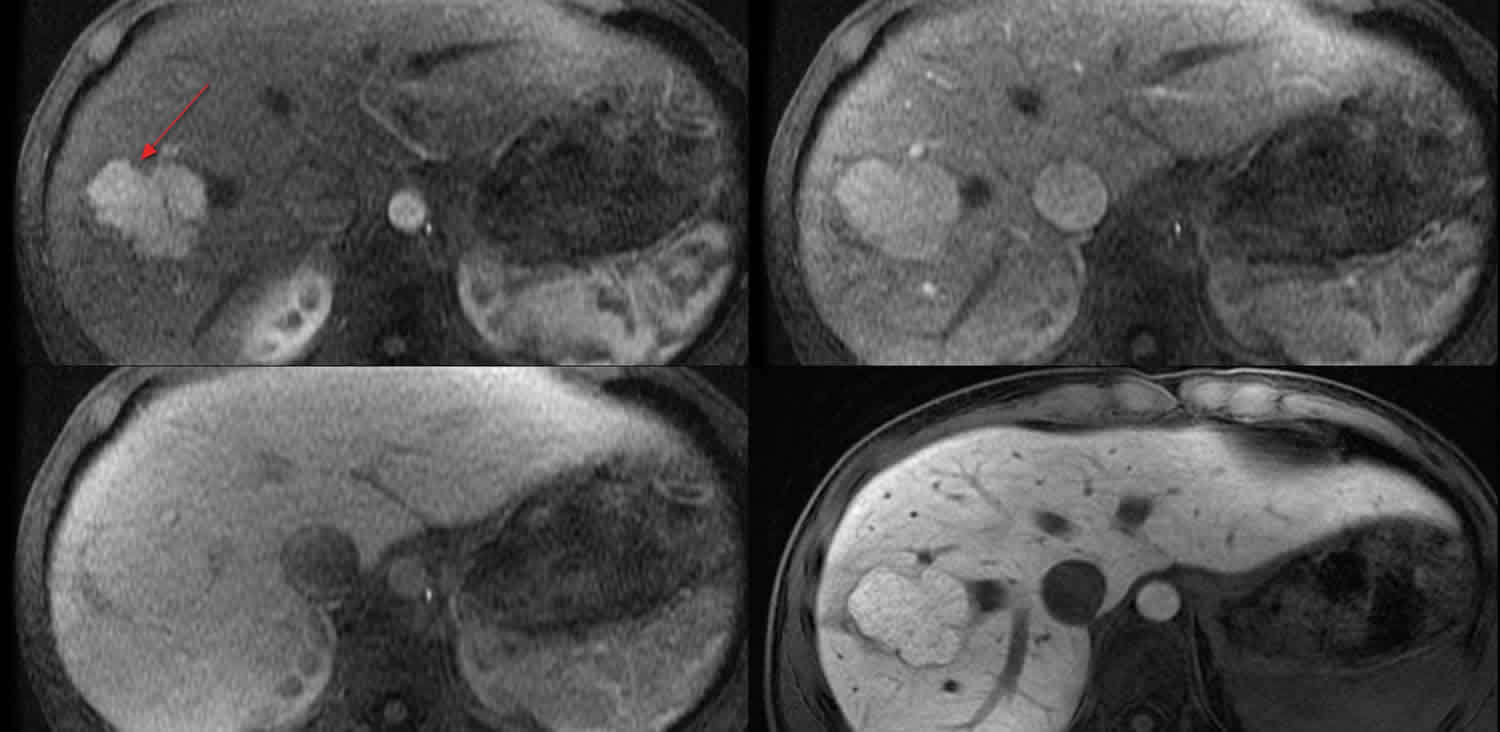
Definitive diagnosis of focal nodular hyperplasia is obtained by imaging in the majority of cases, particularly in those exhibiting a central scar at contrast CT and magnetic resonance imaging (MRI), but radiologically atypical cases occur and require the use of the liver biopsy 4. Angiography typically demonstrates a hypervascular mass with enlarged peripheral vessels and a single central feeding artery. This so-called “wheel-spoke” appearance with the vessels radiating out from the center of the tumor may help distinguish FNH from hepatic adenoma 5.
Previously, focal nodular hyperplasia was referred to by a variety of synonyms including pedunculated adenoma, solitary hyperplastic nodule, focal cirrhosis, and hepatic hamartoma 2. Because of the indeterminate classification, a standard diagnosis needed to be established. In 1994, the International Working Party of the World Congresses of Gastroenterology standardized the diagnosis term as follicular nodular hyperplasia and categorized it as a regenerative liver nodule. After that, follicular nodular hyperplasia was distinguished from neoplastic hepatic conditions 6.
Many FNHs have characteristic radiographic features on multimodality imaging, but some lesions may be atypical in appearance. Focal nodular hyperplasias are typically asymptomatic lesions, usually requiring no treatment.
Focal nodular hyperplasia can present as early as childhood but is most frequently found in young to middle-aged adults 7; however, focal nodular hyperplasia is most often found in women (80% of cases) of reproductive age 5. Although an association with oral contraceptive use has been speculated, owing to increased prevalence of these tumors in young women, studies have shown that FNH is not hormonally dependent nor affected by oral contraceptives or pregnancy 8. Although exogenous estrogens do not cause focal nodular hyperplasia, they have been shown to increase the size of these masses 9 .
The incidence of focal nodular hyperplasia is increased in females age 20 to 50, further suggesting that the condition may be linked to increased estrogen. In recent literature, oral contraceptives as a cause of developing of focal nodular hyperplasia are not proven. However, women who are on a daily oral contraceptive regimen display larger nodules that women who are not taking oral estrogen-based contraception 10. Additionally, females, regardless of oral contraceptive use, have been shown to have larger nodules than men 11.
Focal nodular hyperplasia has rarely presented in children. Most pediatric cases have been reported in patients with a history of chemotherapy, malignancy, or hematopoietic stem cell therapy. However, Baylor University Medical Center reported a case of focal nodular hyperplasia in a healthy 3-year-old female with no pertinent past medical history. In this instance, the patient underwent surgical resection of a palpable abdominal mass for what oncologists believed to be a hepatic malignancy. After histological evaluation, it was determined that this patient had focal nodular hyperplasia 12.
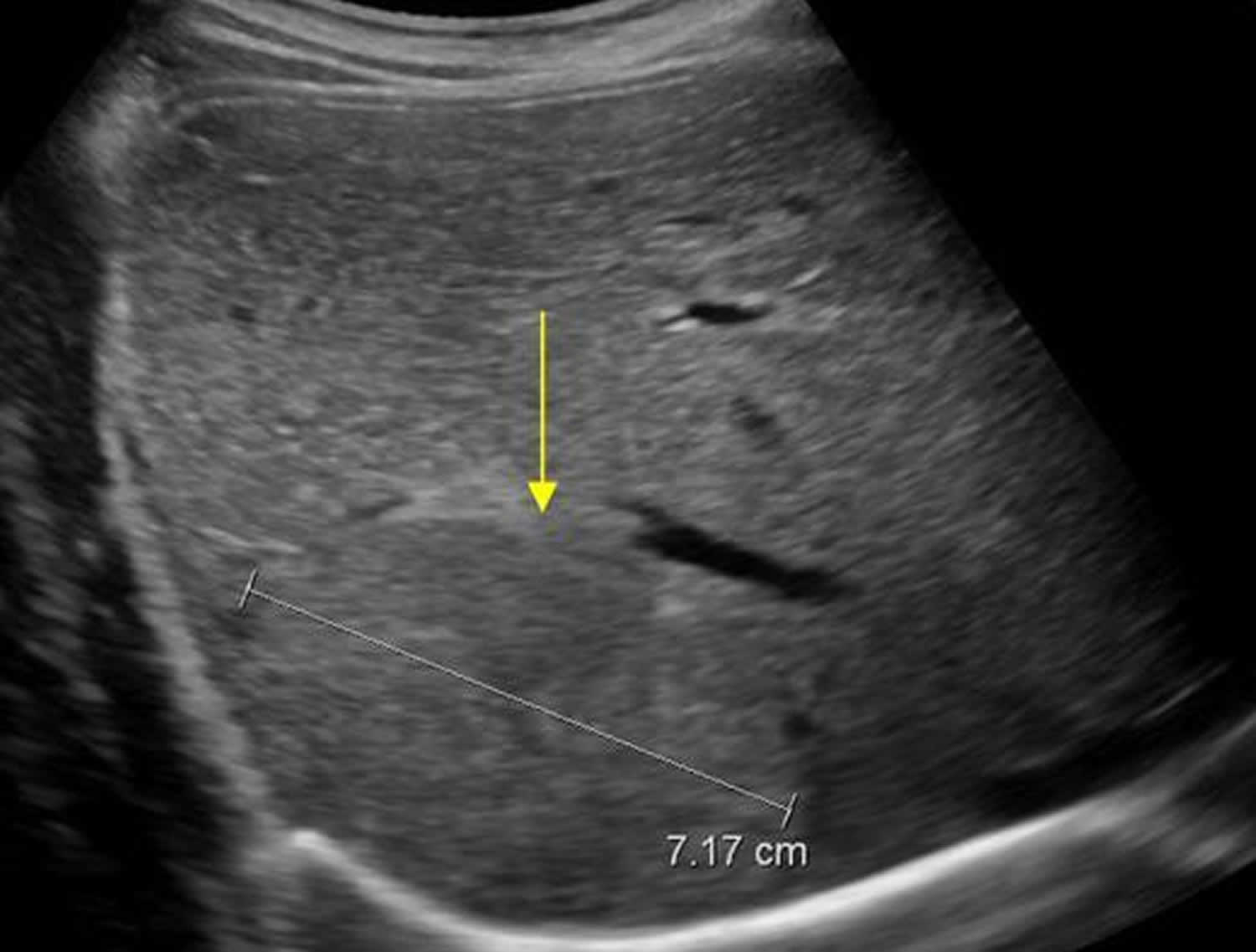
Figure 1. Focal nodular hyperplasia ultrasound
Focal nodular hyperplasia types
FNH is divided into two types 4:
- typical: 80%
- atypical: 20%
Typical focal nodular hyperplasia
Macroscopically, typical lesions demonstrate a mass which is often quite large with well-circumscribed margins but poorly encapsulated. A characteristic feature is a prominent central scar with radiating fibrous septa, but this is present in less than 50% of cases 13. A large central artery is usually present with spoke-wheel like centrifugal flow (no portal veins) 14.
Histologically the lesion is composed of abnormal nodular architecture, malformed vessels, and cholangiolar proliferation. Nearly normal hepatocytes are arranged in 1-2 cell-thick plates. Bile ductules are usually found at the interface between hepatocytes and fibrous regions 15. Kupffer cells are present 9.
There is no malignant potential 15.
Atypical focal nodular hyperplasia
An atypical focal nodular hyperplasia refers to a lesion which lacks the central scar and central artery, thus harder to distinguish from other lesions on gross inspection and imaging, or abnormal nodular architecture but with abnormal cholangiolar proliferation 9.
Atypical features also include pseudocapsule, lesion heterogeneity (more commonly seen in adenoma), non-enhancement of the central scar and intralesional fat 16.
Nodules can grow and disappear, and new nodules can appear even after resection 13.
Focal nodular hyperplasia variants
Some authors also describe division of atypical focal nodular hyperplasia into several variants which include 17:
- telangiectatic variant: most common
- mixed hyperplastic and adenomatous variant
- lesions with large cell hepatocellular atypia
Focal nodular hyperplasia causes
The exact cause of focal nodular hyperplasia has not been definitively established. However, it is thought to be caused by arterial malformations within the liver. These malformations, coupled with changes in perfusion, cause a regenerative, hyperplastic response of the normal hepatocyte. Interestingly, hepatocytes may respond with hyperplasia after both hypoperfusion and hyperperfusion 18.
Therefore, any comorbid condition that causes a predisposition to the development of arterial malformations may increase the risk of developing focal nodular hyperplasia. Hereditary hemorrhagic telangiectasia also known as Osler-Weber-Rendu syndrome can cause an increased incidence of focal nodular hyperplasia. The incidence of focal nodular hyperplasia also increases in the presence of hemangiomas 18.
Further complicating the delineation of any exact cause is the fact that focal nodular hyperplasia may develop without any comorbid vascular malformation. The research does not reveal a clear reason why this is the case. This may be due to an inadequate biopsy or structural malformations confounding histological examination and imaging. Other leading researchers in gastroenterology believe intermittent disruptions of blood flow within the hepatic lobule lead to the characteristic cellular response.
Arguably, the cornerstone distinction that differentiates focal nodular hyperplasia from other hepatic masses is its origin. A series of studies showed that the majority of hepatocytes within a focal nodular hyperplasia nodule stem from a polyclonal origin 19. Because of this, it has been classified as a benign condition. One study demonstrated an increased incidence of focal nodular hyperplasia among monozygotic twins indicating a possible genetic component 20. However, a genetic mutation responsible for predisposing individuals to focal nodular hyperplasia has not yet been identified.
Focal nodular hyperplasia associations
Focal nodular hyperplasia association with other benign lesions is commonly seen (~25%) 21:
- hepatic hemangiomas (most common) 16
- hereditary hemorrhagic telangiectasia
- arteriovenous malformations (AVM)
- anomalous venous drainage
- hepatic adenoma (possible but not proven) 22
- congenital absence of portal vein/portal vein atresia
- Budd-Chiari syndrome
- portal shunts
- idiopathic portal hypertension
- portal or pulmonary hypertension 23
Focal nodular hyperplasia histopathology
A majority of patients with focal nodular hyperplastic lesions present with solitary lesions between 4 and 8 centimeters. Only 3% of cases reach a diameter greater than ten centimeters. The hallmark feature of focal nodular hyperplasia that is evident grossly is known as a “central scar.” Histologically, the central scar consists of mature collagen surrounded by aberrant arteries, draining veins, and fibrous septae forming a pseudocapsule which distinguishes it from hepatocellular carcinoma, hepatocellular adenomas, and fibrolamellar hepatocellular carcinoma. Because of this, biopsy of the central scar is essential in determining diagnosis and ruling out malignant causes. Focal nodular hyperplastic lesions may also contain bile ducts and reticuloendothelial hepatic macrophages also known as Kuppfer cells. Additionally, these lesions lack the calcifications that are commonly seen in hepatic adenomas. Cytologically, these hepatocytes from focal nodular hyperplastic lesions are unremarkable reaffirming benign origin 24
If a biopsy sample lacks a “central scar,” bile duct canals, or calcifications that would aid in the differentiation between hepatic adenomas and focal nodular hyperplasia, immunohistochemical stainings may aid in establishing a diagnosis. Focal nodular hyperplasia displays an increased expression of glutamine synthetase located in the periphery of the nodules, which may be stained and identified 25.
Focal nodular hyperplasia symptoms
Focal nodular hyperplasia masses are either found incidentally on imaging or present due to mass effect, with right upper quadrant pain in 20% 26. Even though it is usually asymptomatic, it may present as a palpable abdominal mass that is more likely to be tender when the lesion’s diameter exceeds 10 centimeters. As focal hyperplastic nodules are solitary lesions, hepatomegaly is uncommon. Unlike hepatic adenomas, FNHs are only rarely complicated by spontaneous rupture and hemorrhage 27.
Focal nodular hyperplasia diagnosis
Diagnosis of focal nodular hyperplasia consists of biopsy or imaging consistent with the characteristics of FNH and exclusion of other similar lesions. Lab work is largely unremarkable in the setting of focal nodular hyperplasia 28. Alpha-fetoprotein levels (AFP) are generally within reference ranges, further supporting the benign clinical course associated with focal nodular hyperplasia. Additionally, serum alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), and gamma-glutamyl transpeptidase (GGT) levels may display minor elevations, but these may be confounded by comorbid conditions as they are rarely elevated in the sole presence of focal nodular hyperplasia 29.
Ultrasonography is most often the first imaging modality used in the evaluation of liver lesions including focal nodular hyperplasia, but lacks an appropriate sensitivity (twenty percent) to be utilized as the gold standard imaging modality. FNH often appears as an isoechoic or hypoechoic mass with a hyperechoic central scar relative to the liver parenchyma 30. Contrast-enhanced ultrasonography has been utilized outside the United States with some reports indicating that focal nodular hyperplasia and hepatocellular adenoma can be distinguished with the advent of early arterial phase imaging which reveals arterial morphology and filling direction 31.
Triphasic helical computed tomography with and without contrast, although not the gold standard imaging modality, is a cheap and reliable imaging modality. Pre-contrast, focal nodular hyperplasia appears hypodense or isodense with a central scar evident in approximately one-third of cases. FNH appears hyperdense during the hepatic arterial phase and isodense during the portal venous phase rendering the lesion indistinguishable from the rest of the liver parenchyma 32.
With magnetic resonance imaging, focal nodular hyperplastic lesions appear isointense to hypointense lesions on T1-weighted images and isointense to hyperintense on T2-weighted images. Similar to CT imaging, the lesion enhances on arterial phase images and is relatively obfuscated on venous and delayed phase images. Sensitivity and specificity of 99% and 100%, respectively, may be achieved with hepatobiliary contrast media consisting of administration of gadobenate dimeglumine. Thus, MRI with hepatobiliary scintigraphy is the best test to diagnose focal nodular hyperplasia 33
Focal nodular hyperplasia treatment
Given the inherent procedural risks associated with percutaneous biopsy and surgical resection and the benign nature with no malignant potential and a minuscule risk of complication (rupture, hemorrhage) of focal nodular hyperplasia, close observation is recommended with serial imaging every three to six months. However, biopsy or resection is often pursued if a patient is symptomatic, if there is a concern for underlying malignancy after an inconclusive biopsy, or if a lesion displays continued growth. The definitive therapy remains surgical resection 34.
Focal nodular hyperplasia was first described in the 1960s prior to the widespread use of oral contraceptive pills. Since then, there has been no proven increase in the incidence of focal nodular hyperplasia with the use of estrogens. However, almost all documented cases of hemorrhage or rupture have occurred in patients taking oral contraceptives. While discontinuation of oral contraceptives is not necessarily indicated, follow up imaging is advised for any patient on estrogen therapy to monitor for growth. In the pediatric population, a thorough examination of the patient’s risk factors for underlying malignancy or liver disease is warranted 35.
- Kim MJ, Han SY, Baek YH, Lee SW, Kwon HJ. A case of focal nodular hyperplasia with growth progression during pregnancy. Clin Mol Hepatol. 2014;20(4):392–397. doi:10.3350/cmh.2014.20.4.392 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4278071[↩]
- Hamad S, Mukherjee S. Focal Nodular Hyperplasia. [Updated 2019 Mar 2]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2019 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK532244[↩][↩]
- Lizardi-Cervera J, Cuéllar-Gamboa L, Motola-Kuba D. Focal nodular hyperplasia and hepatic adenoma: a review. Ann Hepatol. 2006 Jul-Sep;5(3):206-11.[↩]
- Mathieu D, Vilgrain V, Mahfouz AE, Anglade MC, Vullierme MP, Denys A. Benign liver tumors. Magn Reson Imaging Clin N Am. 1997;5:255–288.[↩][↩]
- Roncalli M, Sciarra A, Tommaso LD. Benign hepatocellular nodules of healthy liver: focal nodular hyperplasia and hepatocellular adenoma. Clin Mol Hepatol. 2016;22(2):199–211. doi:10.3350/cmh.2016.0101 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4946404[↩][↩]
- Nat L, Poantă LI. Focal nodular hyperplasia (FNH). Rom J Intern Med. 2014 Jan-Mar;52(1):45-9.[↩]
- Diagnosis of focal nodular hyperplasia: not so easy. Am J Surg Pathol. 2001 Oct;25(10):1322-5.https://journals.lww.com/ajsp/Citation/2001/10000/Diagnosis_of_Focal_Nodular_Hyperplasia__Not_So.15.aspx[↩]
- Mathieu D, Kobeiter H, Maison P, Rahmouni A, Cherqui D, Zafrani ES, et al. Oral contraceptive use and focal nodular hyperplasia of the liver. Gastroenterology. 2000;118:560–564.[↩]
- Lencioni R, Cioni D, Bartolozzi C. Focal liver lesions, detection, characterization, ablation. Springer Verlag. (2005) ISBN:3540644644[↩][↩][↩]
- Kubota T, Shimizu K, Sonoyama T, Ikeda E, Kurioka H, Ouchi T, Suyama Y, Hosokawa Y, Yamagishi H. Enlarged focal nodular hyperplasia of the liver under the influence of oral contraceptives. Hepatogastroenterology. 2001 Nov-Dec;48(42):1736-9.[↩]
- Turaihi HH, Thambi-Pillai TC, Binneboese A. Hepatic Adenoma – A Case Report. S D Med. 2017 Mar;70(3):105-107.[↩]
- Koolwal J, Birkemeier KL, Zreik RT, Mattix KD. Pedunculated focal nodular hyperplasia in a healthy toddler. Proc (Bayl Univ Med Cent). 2018 Jan;31(1):97-99.[↩]
- Bioulac-sage P, Balabaud C, Wanless IR. Diagnosis of focal nodular hyperplasia: not so easy. Am. J. Surg. Pathol. 2001;25 (10): 1322-5. https://journals.lww.com/ajsp/Citation/2001/10000/Diagnosis_of_Focal_Nodular_Hyperplasia__Not_So.15.aspx[↩][↩]
- Kumar V, Abbas AK, Fausto N et-al. Robbins and Cotran pathologic basis of disease. W B Saunders Co. (2005) ISBN:0721601871.[↩]
- Grazioli L, Morana G, Kirchin MA et-al. Accurate differentiation of focal nodular hyperplasia from hepatic adenoma at gadobenate dimeglumine-enhanced MR imaging: prospective study. Radiology. 2005;236 (1): 166-77. doi:10.1148/radiol.2361040338[↩][↩]
- Intratumoral steatosis in focal nodular hyperplasia coinciding with diffuse hepatic steatosis: CT and MRI findings with histologic correlation. Abdom Imaging. 2000 Mar-Apr;25(2):179-81. https://link.springer.com/article/10.1007%2Fs002619910039[↩][↩]
- MRI of Atypical Focal Nodular Hyperplasia of the Liver: Radiology–Pathology Correlation. AJR Am J Roentgenol. 2004 May;182(5):1227-31. https://www.ajronline.org/doi/full/10.2214/ajr.182.5.1821227[↩]
- Herman P, Pugliese V, Machado MA, Montagnini AL, Salem MZ, Bacchella T, D’Albuquerque LA, Saad WA, Machado MC, Pinotti HW. Hepatic adenoma and focal nodular hyperplasia: differential diagnosis and treatment. World J Surg. 2000 Mar;24(3):372-6.[↩][↩]
- Koehne de Gonzalez AK, Salomao MA, Lagana SM. Current concepts in the immunohistochemical evaluation of liver tumors. World J Hepatol. 2015 Jun 08;7(10):1403-11.[↩]
- Mindikoglu AL, Regev A, Levi JU, Casillas J, Schiff ER. Focal nodular hyperplasia in identical twins. Am. J. Gastroenterol. 2005 Jul;100(7):1616-9.[↩]
- MRI of atypical focal nodular hyperplasia of the liver: radiology-pathology correlation. American Journal of Roentgenology. 2004;182: 1227-1231. 10.2214/ajr.182.5.1821227 https://www.ajronline.org/doi/full/10.2214/ajr.182.5.1821227[↩]
- Laurent C, Trillaud H, Lepreux S, Balabaud C, Bioulac-Sage P. Association of adenoma and focal nodular hyperplasia: experience of a single French academic center. Comp Hepatol. 2003;2(1):6. Published 2003 Apr 23. doi:10.1186/1476-5926-2-6 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC161818[↩]
- Diagnosis of focal nodular hyperplasia: not so easy. Am J Surg Pathol. 2001 Oct;25(10):1322-5. https://journals.lww.com/ajsp/Citation/2001/10000/Diagnosis_of_Focal_Nodular_Hyperplasia__Not_So.15.aspx[↩]
- Valentino PL, Ling SC, Ng VL, John P, Bonasoni P, Castro DA, Taylor G, Chavhan GB, Kamath BM. The role of diagnostic imaging and liver biopsy in the diagnosis of focal nodular hyperplasia in children. Liver Int. 2014 Feb;34(2):227-34.[↩]
- Joseph NM, Ferrell LD, Jain D, Torbenson MS, Wu TT, Yeh MM, Kakar S. Diagnostic utility and limitations of glutamine synthetase and serum amyloid-associated protein immunohistochemistry in the distinction of focal nodular hyperplasia and inflammatory hepatocellular adenoma. Mod. Pathol. 2014 Jan;27(1):62-72.[↩]
- Vogl TJ, Lencioni R, Hammerstingl RM et-al. Magnetic Resonance Imaging in Liver Disease, Technical Approach, Diagnostic Imaging of Liver Neoplasms, Focus on a New Superparamagnetic Contrast Agent. Thieme. (2003) ISBN:1588902366.[↩]
- Ashby PF, Alfafara C, Amini A, Amini R. Spontaneous Rupture of a Hepatic Adenoma: Diagnostic Nuances and the Necessity of Followup. Cureus. 2017 Dec 20;9(12):e1975.[↩]
- Roncalli M, Sciarra A, Tommaso LD. Benign hepatocellular nodules of healthy liver: focal nodular hyperplasia and hepatocellular adenoma. Clin Mol Hepatol. 2016 Jun;22(2):199-211.[↩]
- Mneimneh W, Wadad M, Farges O, Bedossa P, Belghiti J, Paradis V. High serum level of alpha-fetoprotein in focal nodular hyperplasia of the liver. Pathol. Int. 2011 Aug;61(8):491-4.[↩]
- D’Onofrio M, Crosara S, De Robertis R, Canestrini S, Mucelli RP. Contrast-Enhanced Ultrasound of Focal Liver Lesions. AJR Am J Roentgenol. 2015 Jul;205(1):W56-66.[↩]
- Kong WT, Wang WP, Huang BJ, Ding H, Mao F, Si Q. Contrast-enhanced ultrasound in combination with color Doppler ultrasound can improve the diagnostic performance of focal nodular hyperplasia and hepatocellular adenoma. Ultrasound Med Biol. 2015 Apr;41(4):944-51.[↩]
- Scialpi M, Pierotti L, Gravante S, Piscioli I, Pusiol T, Schiavone R, D’Andrea A. Split-bolus versus triphasic multidetector-row computed tomography technique in the diagnosis of hepatic focal nodular hyperplasia: a case report. J Med Case Rep. 2014 Dec 14;8:425.[↩]
- Masand PM. Magnetic resonance imaging features of common focal liver lesions in children. Pediatr Radiol. 2018 Aug;48(9):1234-1244.[↩]
- Fodor M, Primavesi F, Braunwarth E, Cardini B, Resch T, Bale R, Putzer D, Henninger B, Oberhuber R, Maglione M, Margreiter C, Schneeberger S, Öfner D, Stättner S. Indications for liver surgery in benign tumours. Eur Surg. 2018;50(3):125-131.[↩]
- Sugito K, Uekusa S, Kawashima H, Furuya T, Ohashi K, Inoue M, Ikeda T, Koshinaga T, Tomita R, Mugishima H, Maebayashi T. The clinical course in pediatric solid tumor patients with focal nodular hyperplasia of the liver. Int. J. Clin. Oncol. 2011 Oct;16(5):482-7.[↩]