Glucose intolerance
Glucose intolerance is an umbrella term for metabolic conditions which result in higher than normal blood glucose levels – hyperglycemia. Western lifestyles have seen glucose intolerance become more common year on year.
Glucose intolerance includes anyone with either impaired fasting glucose or impaired glucose tolerance.
The World Health Organisation’s definitions for impaired fasting glucose and impaired glucose tolerance, glucose intolerance is defined as 1:
- A fasting blood glucose level of above 108 mg/dl (6.0 mmol/L) or
- A blood glucose level of over 140.4 mg/dl (7.8 mmol/L) 2 hours after consuming 75g of glucose.
Conditions which can be considered as glucose intolerance include:
- Impaired fasting glucose
- Impaired glucose tolerance
- Pre-diabetes
- Type 1 diabetes due to autoimmune B-cell destruction, usually leading to absolute insulin deficiency
- Type 2 diabetes due to a progressive loss of B-cell insulin secretion frequently on the background of insulin resistance
- Gestational diabetes mellitus (diabetes diagnosed in the second or third trimester of pregnancy that was not clearly overt diabetes prior to gestation) 2
- Specific types of diabetes due to other causes such as neonatal diabetes and maturity-onset diabetes of the young, diseases of the exocrine pancreas, and drug- or chemical-induced diabetes 3
In most cases, the diagnosis of a type of diabetes or glucose intolerance is based on the patient’s condition at the time, but not all patients have a set of symptoms that fit readily into a particular class.
When hyperglycemia is present, its severity may change in time, depending on the underlying process. Choosing an appropriate management approach to any disorders of glucose intolerance necessitates a strong understanding of the mechanisms involved in the disease process 4.
From 2009 through 2012, the prevalence of pre-diabetes in the United States was 37% in adults older than 20 years and 51% in those older than 65 years. According to American Diabetes Association, in 2015, 84.1 million Americans age 18 and older had prediabetes. People with impaired glucose tolerance are generally asymptomatic and have only increased glucose level; therefore, a large proportion of these patients remains undiagnosed.
In 2010, the worldwide prevalence of glucose intolerance was about 8% 5. In the United States, about 57 million people had impaired fasting glucose while 30 million had impaired glucose tolerance. In a study of progression from normal glucose tolerance to impaired glucose tolerance, 14% of participants with normal glucose tolerance had progressed to impaired fasting glucose and 48% to impaired glucose tolerance by 10 years. The progress from normal glucose tolerance to impaired glucose tolerance is about 4-times greater than the progression from normal glucose tolerance to impaired fasting glucose. Persons older than 65 years have higher rates of progression to abnormal 2-hour value during a glucose tolerance test than younger persons.
Zhang et al. did a systematic review on the A1C level and future risk of diabetes and found that as A1C increased from 6.0% to 6.5%, 5-year incidence of diabetes showed an increase of 25% to 50%. Conversion from glucose intolerance to diabetes varies with population characteristics and criteria used for diagnosis.
As glucose intolerance is a state with increased risk for diabetes and its complications, intervention can reduce the progression to diabetes. Interventions are more beneficial in impaired glucose tolerance with or without impaired fasting glucose in comparison to isolated impaired fasting glucose.
Glucose intolerance causes
The precise cause of glucose intolerance remains unclear 6. There appears to be an interaction between genetic and epigenetic factors with a sedentary lifestyle and poor dietary habits. Defects in both insulin resistance and insulin secretion are important in declaring the clinical syndrome. Causes of glucose intolerance may include the following 3..
Genetic defects of beta-cell function include the following:
- Mutation on chromosome 12, the hepatocyte nuclear factor (HNF-1) alpha -MODY3
- Mutation on chromosome 7p, the glucokinase gene -MODY2
- Mutation on chromosome 20, HNF-4 alpha -MODY1
- Point mutations in mitochondrial DNA
Defects in insulin action include the following:
- Structure and function of insulin receptor: postreceptor signal transduction pathways
- Type A insulin resistance
- Leprechaunism
- Rabson-Mendenhall syndrome
- Lipoatrophic diabetes
Diseases of the exocrine pancreas include the following:
- Pancreatitis
- Trauma
- Infection
- Pancreatectomy
- Pancreatic cancer
- Cystic fibrosis
- Hemochromatosis
- Fibrocalculous pancreatopathy
Note that the malnutrition-related diabetes has been eliminated from the above list, as evidence is lacking on protein deficiency as a direct cause of diabetes, and fibrocalculous pancreatopathy has been reclassified as a disease of the exocrine pancreas.
Endocrine diseases associated with excess production of insulin antagonists include the following:
- Acromegaly
- Cushing syndrome
- Glucagonoma
- Pheochromocytoma
- Hyperthyroidism
- Somatostatinoma
- Aldosteronoma
Drugs or chemical agents with adverse effects on glucose tolerance include the following:
- Thiazides
- Diazoxide
- Glucocorticoids
- Calcineurin inhibitors, such as cyclosporine and tacrolimus
- Oral contraceptives
- Beta-adrenergic agonists
- Nicotinic acid
- Thyroid hormone
- Pentamidine
- Alpha interferon
- Atypical antipsychotics, especially clozapine and olanzapine
- Antiretroviral drugs
- Vacor
Infections associated with beta-cell destruction include the following:
- Rubella
- Coxsackievirus B
- Mumps
- Cytomegalovirus
- Adenovirus
Genetic syndromes that predispose an individual to impaired glucose tolerance include the following:
- Down syndrome
- Klinefelter syndrome
- Turner syndrome
- Wolfram syndrome
- Friedreich ataxia
Pregnancy can be associated with gestational diabetes mellitus, and the risk of diabetes increases with parity.
Obesity is a powerful determinant of glucose intolerance in the general population and develops through the interaction of genetics and acquired factors such as physical inactivity and dietary habits.
Immune-mediated causes of impaired glucose tolerance include stiff person syndrome and anti-insulin receptor abnormalities. Other causes of glucose intolerance are liver disease (as in cirrhosis) and renal failure.
Glucose intolerance pathophysiology
In healthy individuals, levels of glucose in the circulation are strictly regulated. The exit and entry of glucose are predominantly regulated by insulin and glucagon respectively. During an overnight fast of 10 to 14 hours, glucose is produced primarily in the liver by glycogenolysis and gluconeogenesis. This is known as endogenous glucose production (endogenous glucose production) and is directly related to both fat-free mass and fasting plasma glucose (fasting plasma glucose) concentration 7. In the postprandial state, endogenous glucose production is suppressed by an increase in insulin concentration and a decrease in glucagon levels.
Impaired fasting glucose and impaired glucose tolerance are insulin resistance conditions with associated beta cell dysfunction. Initially, there is a compensatory increase in insulin secretion which maintains glucose levels in normal range. As the condition progresses, beta cells change, and the insulin secretion is unable to maintain glucose homeostasis, producing glucose intolerance. Under normal physiological conditions, postprandial insulin release is not in steady state but pulsatile. This pattern of pulsatile secretion is blunted in individuals with glucose intolerance, reflecting the lost ability of beta cells to sense and respond to changes in plasma glucose levels.
Due to insulin resistance, the fasting plasma glucose concentrations are higher in glucose intolerance than in those with normal glucose tolerance. In overweight or obese individuals, increased liver fat oxidation is also seen.
The two subsets of glucose intolerance, impaired fasting glucose and impaired glucose tolerance differ in the site of insulin resistance as well as the pattern of insulin secretion. impaired fasting glucose is characterized predominantly by hepatic insulin resistance and normal muscle insulin sensitivity. People with impaired fasting glucose show a defect in early phase insulin secretory response to glucose. impaired glucose tolerance is mainly associated with muscle insulin resistance. In addition to the defective early-phase insulin-secretory response, impaired glucose tolerance also shows a severe deficit in late-phase insulin secretion. Evolving data suggest a role for impaired lipolysis, abnormal incretin biology, glucagon dysregulation and inflammation in both impaired glucose tolerance and impaired fasting glucose 8.
Glucose intolerance symptoms
These patients are usually asymptomatic since they have prediabetes. The family history of diabetes and any previous history of gestational diabetes mellitus should be obtained. If they are diabetic, they will present with polyuria, polydipsia, infections and neuropathy.
The symptoms of glucose intolerance match those of type 2 diabetes:
- Feeling very thirsty
- Dry mouth
- Extreme tiredness
- Blurred vision
- Drowsiness
- Frequent need to urinate
- Loss of muscle mass
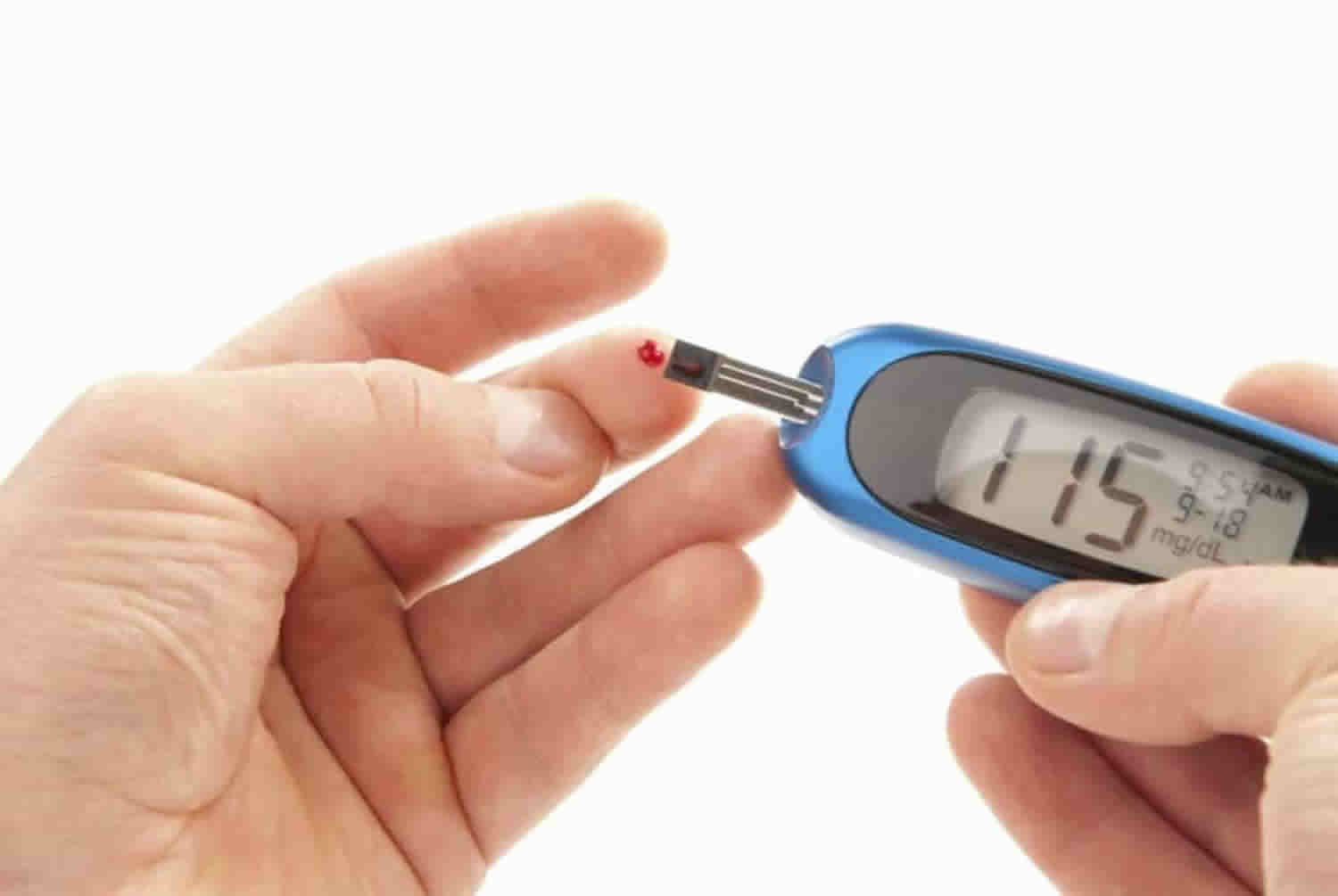
Glucose intolerance test
A number of tests can be used to diagnose forms of glucose intolerance.
Test performed to diagnose glucose intolerance include:
- Fasting plasma glucose test. The blood sample is taken after an 8 hour overnight fast. As per American Diabetes Association, fasting plasma glucose levels between 100 and 125 mg/dL (5.6 to 6.9 mmol/L) are diagnostic of impaired fasting glucose. Several organizations, including World Health Organization (WHO), diagnose impaired fasting glucose at fasting plasma glucose between 110 and 125 mg/dl (6.1 and 6.9 mmol/L).The diagnosis of diabetes requires fasting plasma glucose greater than or equal to 126 mg/dl. Fasting plasma glucose is fast and easy to perform and an established standard but has a high day to day variability.
- Oral glucose tolerance test (OGTT). In this test, plasma glucose level is measured before and 2 hours after ingestion of 75 gm of glucose. Impaired glucose tolerance is diagnosed if plasma glucose level in the 2-hour sample is between 140 to 199 mg/dL (7.8 to 11.0 mmol/L). Diabetes is diagnosed if the plasma glucose is greater than or equal to 200 mg/dl. It is also a standard test but is inconvenient and more costly than fasting plasma glucose and has major variability issues. Patients need to consume a diet with at least 150 g per day of carbohydrates for 3 to 5 days and not take any medications that can impact glucose tolerance such as steroids and thiazide diuretics.
- Glycated Hemoglobin (A1C). This test gives an average of blood glucose over last two to three months. Patients with A1C between 5.7% to 6.4% (39 to 47 mmol/mol) are diagnosed as having increased risk of diabetes or prediabetes, the latter has stigmatization and should be avoided where possible. For a diagnosis of diabetes, the A1C levels should be greater than or equal to 6.5% (48 mmol/mol). A1C is a convenient, standardized test but is costly and has many issues as discussed below including lower sensitivity.
For all of the above tests, if the person is asymptomatic testing should be repeated on another occasion to make a diagnosis of diabetes.
The disadvantage with oral glucose tolerance test (OGTT) is that it requires patient preparation and is poorly reproducible. Measuring A1C is convenient as fasting is not required. Also, it shows less variation due to pre-analytical variables and is not much affected by acute illness or stress. However, A1C should be measured using National Glycohemoglobin Standardization Program certified method which has been standardized for Diabetes Control and Complications Trial assay. In cases where there is a significant difference in plasma glucose values and A1C levels, interference due to hemoglobin variants is possible, and other glucose assays should be considered. In conditions such as sickle cell disease, pregnancy, hemodialysis, blood loss or transfusion, or erythropoietin therapy, which are known to be associated with increased red cell turnover, only plasma glucose should be used. Also, A1C is costly and may not be available everywhere. It has not been well validated in non-Caucasian populations.
Anemia due to deficiency of iron or vitamin B12 leads to spurious elevation of A1C, limiting its use in countries with high prevalence of anemia. Also, in children and elderly, the relation between A1C and fasting plasma glucose is suboptimal 9.
Fasting plasma glucose , 2-hour plasma glucose during 75-g oral glucose tolerance test (OGTT), and A1C are equally appropriate for diagnosis of glucose intolerance. There is no concordance between the results of these tests. An individual with increased fasting plasma glucose may have normal postprandial glucose or A1C measurements. While all 3 tests can be used, given how cumbersome the oral glucose tolerance test (OGTT) is, it appears that fasting plasma glucose and A1C will be most practical globally bearing in mind the caveats of the tests 10.
Screening
It is estimated that the onset of diabetes occurs 4 to 7 years before its clinical diagnosis. Screening for hyperglycemia can identify individuals who are at risk for preventable diabetes complications. Persons more than 40 years of age should be screened annually. More frequent screening is recommended in individuals with additional risk factors for diabetes. These include:
- Certain races/ethnicities (Native American, African American, Hispanics or Asian American, Pacific Islander)
- Overweight or obese persons: BMI greater than or equal to 25 kg/m2 or 23 kg/m² in Asian Americans
- First-degree relative with diabetes
- History of cardiovascular disease, hypertension, low HDL-cholesterol or hypertriglyceridemia
- Women with polycystic ovarian syndrome
- Physical inactivity
- Conditions associated with insulin resistance such as acanthosis nigricans
Women who are diagnosed with gestational diabetes mellitus should have life-long testing at least every 3 years. For all other patients, testing should begin at age 45 years, and if results are normal, testing should be repeated at a minimum of 3-year intervals.
Fasting plasma glucose and A1C are recommended screening tests. The 75-gm GTT can be used and needed for the diagnosis of impaired glucose tolerance but has poor reproducibility and needs adequate preparation.
Glucose intolerance treatment
Treatments for glucose intolerance will either require lifestyle changes or a combination of lifestyle changes and anti-diabetic medication.
Intensive lifestyle modification has been shown to effectively delay or prevent diabetes in a cost-effective manner 11. Several studies have shown that lifestyle intervention can reduce conversion to type 2 diabetes. The aim is to achieve and maintain 7% loss of initial body weight. It should include moderate-intensity physical activity (such as brisk walking) for at least 150 minutes per week, with a minimum frequency of 3 times a week.
Nonpharmacologic therapy and lifestyle modification include the following:
- Diet
- Exercise
- Counseling related to smoking cessation and alcohol intake
- Reversing drug-related, iatrogenic causation of glucose intolerance
- Substituting or adding agents that do not adversely affect glucose tolerance; reducing dosage of offending drugs
Lifestyle changes involve taking part in regular physical activity, aiming to lose weight, if appropriate, and cutting down on smoking and alcohol as necessary.
If medication is advised, most people will start on a drug, taken in tablet form, called metformin. Note that long-term metformin use may be associated with biochemical vitamin B12 deficiency; thus, consider periodic measurement of vitamin B12 levels in patients receiving metformin, particularly those with anemia or peripheral neuropathy 12.
Some people may need to take additional or alternative medication.
Pharmacologic therapy may be required in the following situations 13:
- Fasting glucose more than 126 mg/dL, postprandial glucose more than 160 mg/dL, or glycosylated hemoglobin (HbA1c) more than 7%
- Hyperglycemia (a significant risk factor in development of vascular complications)
In addition to lifestyle counseling, metformin therapy for prevention of type 2 diabetes should be considered in those with prediabetes, especially for those with a body mass index (BMI) above 35 kg/m², those younger than 60 years, women with prior gestational diabetes, and/or those with rising HBA1C despite lifestyle intervention 12.
All patients with type 1 diabetes are insulin-dependent. Treatment of severe hyperglycemia during acute decompensation in a patient with type 2 diabetes may reverse the state of glucose toxicity, further improving secretory function of beta cells in the pancreas. Type 2 diabetes can be treated effectively with oral hypoglycemic drugs, with or without the addition of insulin. The natural history of type 2 diabetes is that of progressive beta-cell deterioration, secondary failure of oral agents, and the subsequent need for insulin therapy.
Gestational diabetes mellitus is treated with insulin and/or with lifestyle change. Oral agents are contraindicated in pregnancy.
With regard to the management of impaired glucose tolerance, the current approach is aggressive lifestyle modification. The results of the Diabetes Prevention Program showed that metformin therapy and intensive lifestyle intervention reduced the risk of developing type 1 and type 2 diabetes by 31% and 58%, respectively, compared with placebo in individuals with impaired glucose tolerance 14. The Study to Prevent Non-Insulin–Dependent Diabetes Mellitus Trial demonstrated a 25% relative risk reduction in the development of diabetes, and an associated reduction in hypertension (34%) and cardiovascular events (49%) 15.
Orlistat may be beneficial in the context of obesity 11.
Medical nutritional therapy should be guided by the American Dietetic Association recommendations and individualized by weight and height, level of physical activity, and requirements for calories and nutrients 16. A high level of physical activity is desirable, as appropriate to the patient’s ability and general health. Most patients benefit from carefully planned exercise programs tailored to individual needs 17.
Long-term monitoring of affected patients includes ensuring medication compliance, identifying adverse effects, blood glucose and HbA1c monitoring, dietary consultations and measures, and exercise management.
Bariatric Surgery
Bariatric surgery should be considered in a patient with type 2 diabetes who has a BMI of more than 35 kg/m², especially if glycemic control with lifestyle and pharmacotherapy is difficult 18. Surgically induced weight loss may result in improvements in insulin sensitivity and beta-cell function, as well as changes in gut hormones 19. Better diabetic control or complete resolution of the disease (64-93%) is the end result.
A bariatric procedure is not currently recommended in the management of impaired fasting glucose and impaired glucose tolerance; however, glucose intolerance resolved in 99-100% of cases of patients who underwent bariatric surgery for a comorbid state that required such an intervention (eg, class 3 obesity).
Long-term support and medical monitoring are still important after a bariatric procedure. Various complications, including postprandial hyperinsulinemic hypoglycemia, have been reported following gastric bypass surgery.
Glucose intolerance diet
The diet recommended by the diabetes association follows general healthy eating advice. The diabetes association recommends eating a balanced diet based on whole grain foods, rich in fruit and vegetables and low in sugar, salt and saturated fat.
Reducing caloric intake is of paramount importance for those at high risk for developing type 2 diabetes. The quality of fats consumed in the diet is more important than the total quantity. Diets relatively high in monounsaturated fats can help prevent type 2 diabetes. Higher intakes of nuts, berries, yogurt and high fiber food, and tea are associated with reduced diabetes risk. Conversely, red meats and sugar-sweetened beverages are associated with an increased risk of type 2 diabetes. Both the Mediterranean diet and the dietary approaches to stop hypertension (DASH) diet are reasonable strategies.
- Definition and Diagnosis of Diabetes Mellitus and Intermediate Hyperglycaemia. https://www.who.int/diabetes/publications/Definition%20and%20diagnosis%20of%20diabetes_new.pdf[↩]
- Buchanan TA, Xiang AH. Gestational diabetes mellitus. J Clin Invest. 2005 Mar. 115(3):485-91.[↩]
- Glucose Intolerance. https://emedicine.medscape.com/article/119020-overview[↩][↩]
- Fauci AS, Braunwald E, Kasper DL, et al, eds. Harrison’s Principles of Internal Medicine. 17th ed. New York, NY: McGraw-Hill; 2008.[↩]
- Bansal N. Prediabetes diagnosis and treatment: A review. World J Diabetes. 2015 Mar 15;6(2):296-303.[↩]
- Goyal R, Jialal I. Glucose Intolerance. [Updated 2019 Apr 1]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2019 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK499910[↩]
- Ferrannini E, Gastaldelli A, Iozzo P. Pathophysiology of prediabetes. Med. Clin. North Am. 2011 Mar;95(2):327-39, vii-viii.[↩]
- Nathan DM, Davidson MB, DeFronzo RA, Heine RJ, Henry RR, Pratley R, Zinman B., American Diabetes Association. Impaired fasting glucose and impaired glucose tolerance: implications for care. Diabetes Care. 2007 Mar;30(3):753-9.[↩]
- Vikram NK, Jialal I. Use of HbA1c in the diagnosis of diabetes and prediabetes: sensitivity versus specificity. Metab Syndr Relat Disord. 2014 Jun;12(5):255-7[↩]
- Diabetes Canada Clinical Practice Guidelines Expert Committee. Punthakee Z, Goldenberg R, Katz P. Definition, Classification and Diagnosis of Diabetes, Prediabetes and Metabolic Syndrome. Can J Diabetes. 2018 Apr;42 Suppl 1:S10-S15.[↩]
- Gillies CL, Abrams KR, Lambert PC, Cooper NJ, Sutton AJ, Hsu RT, et al. Pharmacological and lifestyle interventions to prevent or delay type 2 diabetes in people with impaired glucose tolerance: systematic review and meta-analysis. BMJ. 2007 Feb 10. 334(7588):299.[↩][↩]
- [Guideline] Riddle MC, ed. American Diabetes Association standards of medical care in diabetes- 2019. Diabetes Care. 2019 Jan. 42 (suppl 1):S1-193.[↩][↩]
- DeFronzo RA. Pharmacologic therapy for type 2 diabetes mellitus. Ann Intern Med. 1999 Aug 17. 131(4):281-303.[↩]
- Report of the Expert Committee on the Diagnosis and Classification of Diabetes Mellitus. Diabetes Care. 1997 Jul. 20(7):1183-97.[↩]
- Chiasson JL. Acarbose for the prevention of diabetes, hypertension, and cardiovascular disease in subjects with impaired glucose tolerance: the Study to Prevent Non-Insulin-Dependent Diabetes Mellitus (STOP-NIDDM) Trial. Endocr Pract. 2006 Jan-Feb. 12 Suppl 1:25-30.[↩]
- Du H, van der A DL, van Bakel MM, et al. Glycemic index and glycemic load in relation to food and nutrient intake and metabolic risk factors in a Dutch population. Am J Clin Nutr. 2008 Mar. 87(3):655-61.[↩]
- Molena-Fernandes C, Bersani-Amado CA, Ferraro ZM, Hintze LJ, Nardo N Jr, Cuman RK. Effects of exercise and metformin on the prevention of glucose intolerance: a comparative study. Braz J Med Biol Res. 2015 Dec. 48 (12):1101-8.[↩]
- [Guideline] Standards of Medical Care in Diabetes-2017: Summary of Revisions. Diabetes Care. 2017 Jan. 40 (suppl 1):S4-S5.[↩]
- Hofsø D, Jenssen T, Bollerslev J, Ueland T, Godang K, Stumvoll M, et al. Beta cell function after weight loss: a clinical trial comparing gastric bypass surgery and intensive lifestyle intervention. Eur J Endocrinol. 2011 Feb. 164(2):231-8.[↩]