How Your Body Metabolizes Sugar
Sugars are the smallest and simplest type of carbohydrate. They are easily digested and absorbed by the body.
There are two types of sugars, and most foods contain some of each kind.
| Single sugars (monosaccharides) are small enough to be absorbed directly into the bloodstream. They include: | Sugars that contain two molecules of sugar linked together (disaccharides) are broken down in your body into single sugars. They include: |
|---|---|
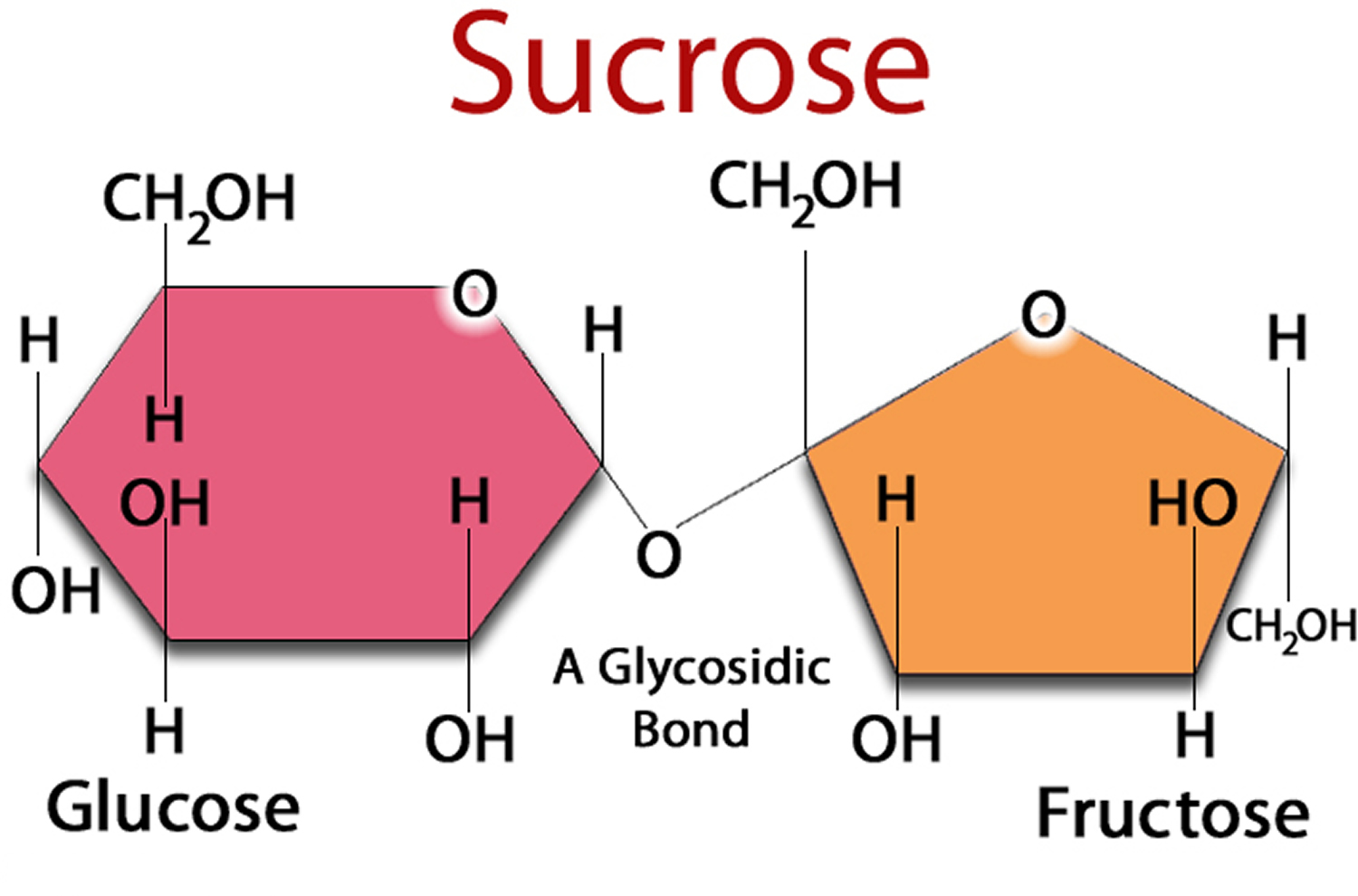
| Fructose | Sucrose (table sugar ) = glucose + fructose |
| Galactose | Lactose (milk sugar) = glucose + galactose |
| Glucose | Maltose (malt sugar) = glucose + glucose |
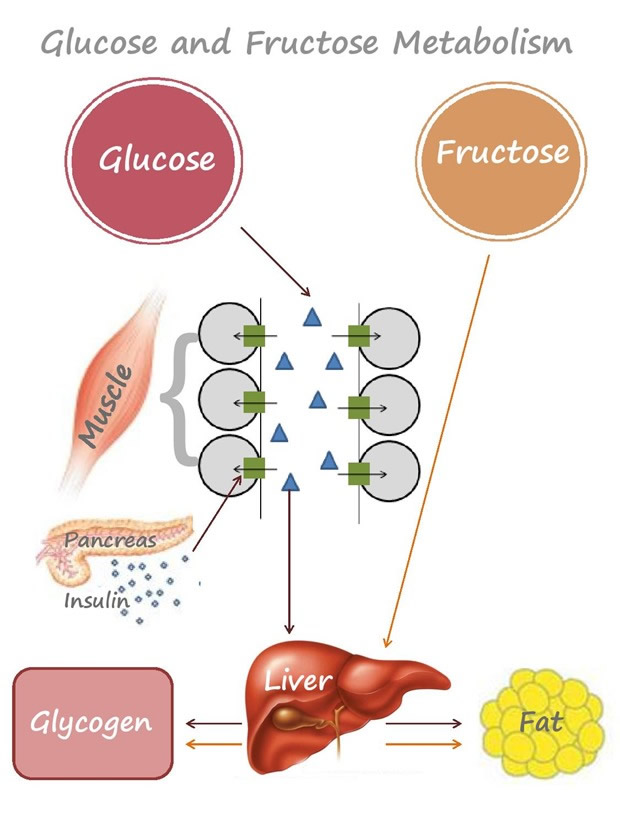
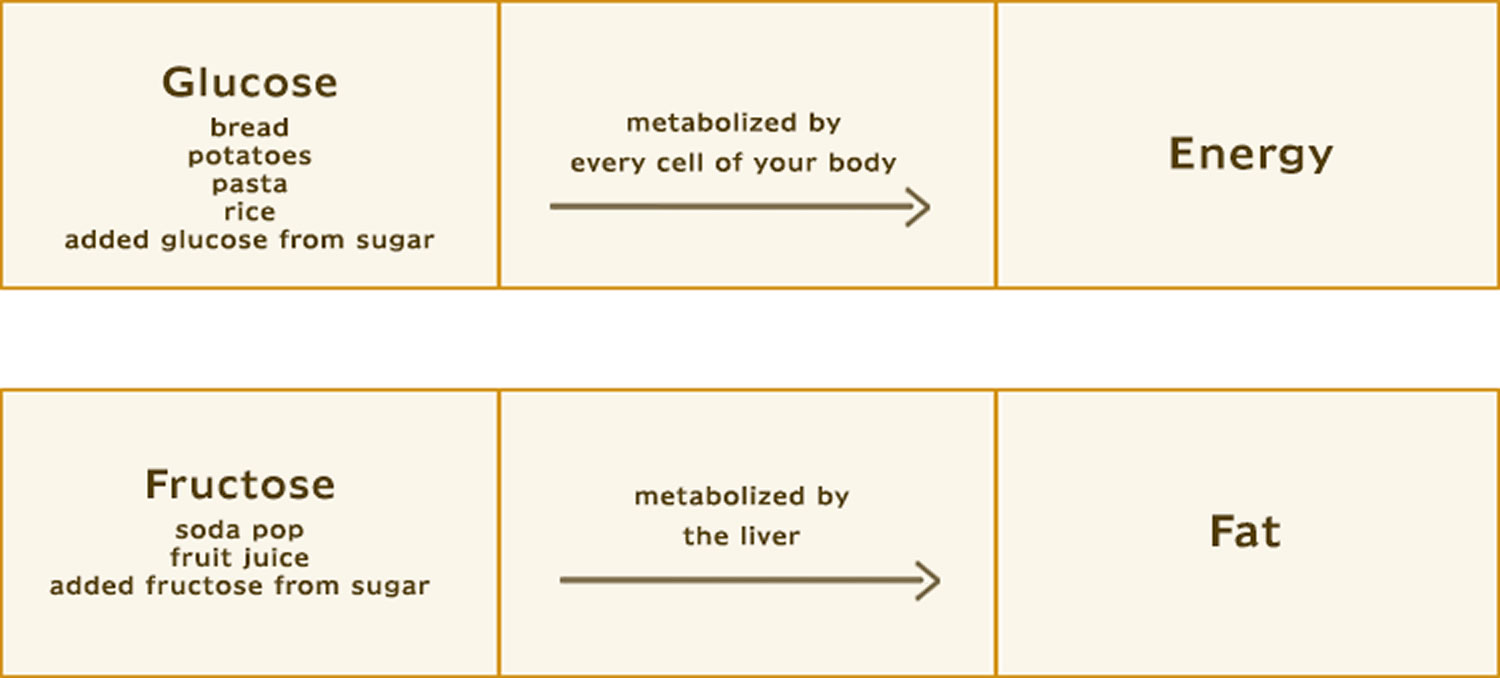
Sugar metabolism 1 is the process by which energy contained in the foods that you eat is made available as fuel for your body. The human body handles glucose and fructose — the most abundant sugars in our diet — in different ways. Virtually every cell in the body can break down glucose for energy. The body’s cells can use glucose directly for energy and most cells can also use fatty acids for energy.
About the only ones that can handle fructose are liver cells. What the liver does with fructose, especially when there is too much in the diet, has potentially dangerous consequences for the liver, the arteries, and the heart 2.
Because glucose and fructose are metabolised differently and when they are consumed in excess they may have different implications for health.
Fructose, also called fruit sugar, was once a minor part of our diet. In the early 1900s, the average American took in about 15 grams of fructose a day (about half an ounce), most of it from eating fruits and vegetables. Today we average four or five times that amount, almost all of it from the refined sugars used to make breakfast cereals, pastries, sodas, fruit drinks, and other sweet foods and beverages.
Refined table sugar, called sucrose, is half glucose and half fructose. High-fructose corn syrup is about 55% fructose and 45% glucose.
It is easy to over-consume sugar in juice and sweet drinks, as they contain mostly water and sugar. One glass of orange juice can contain concentrated sugar from five or six whole oranges. And while it is easy to drink that much sugar, you would be less likely to eat that many oranges in one go.
Fizzy drinks do not make you feel full as quickly as foods do. This makes them easy to over-consume. And a small fizzy drink contains nine teaspoons of added sugar, so drinking just one can means that you have almost reached your recommended maximum intake for that whole day.
The American Heart Association’s guideline is the most frequently used guideline by public health officials. It suggests limiting added sugar to less than 6 teaspoons/day (25 g) for women, 3-6 tsp. (12-25 g) for children, and 9 tsp. (36 g) for men 3. The World Health Organization limit is fairly similar: It suggests that we should be consuming no more than 5% of our daily calories in added sugar, which amounts to about 25 g of added sugar in a 2,000-calorie diet.
A panel convened by the Institute of Medicine recommended the highest dietary limit, that no more than 25% of daily calories should be consumed in added sugar. We do not support this high a limit because studies have shown that consuming sugar at this level could significantly increase your chance of death from heart disease and stroke ,1 as well as affect your metabolism in ways that put you at risk for a range of chronic diseases.2
Glucose
Glucose is an essential nutrient for the human body. It is the major energy source for many cells, which depend on the bloodstream for a steady supply. Blood glucose levels, therefore, are carefully maintained. The liver plays a central role in this process by balancing the uptake and storage of glucose via glycogenesis and the release of glucose via glycogenolysis and gluconeogenesis. The several substrate cycles in the major metabolic pathways of the liver play key roles in the regulation of glucose production.
Looking at glucose first, when food is consumed, there is a corresponding rise and subsequent fall in blood glucose level, as glucose is absorbed from the gastrointestinal tract into the blood and then taken up into the cells in the body.
Glucose in the blood stimulates the pancreas to release insulin, which then triggers uptake of glucose by cells in the body (e.g. muscle cells) causing blood glucose to return to base levels. Insulin will turn off fat burning and promote glucose burning as the body’s primary fuel source. Any excess glucose ends up being stored as glycogen in the muscles, and it can also be stored as lipid in the fat tissue.
Glucose Metabolism
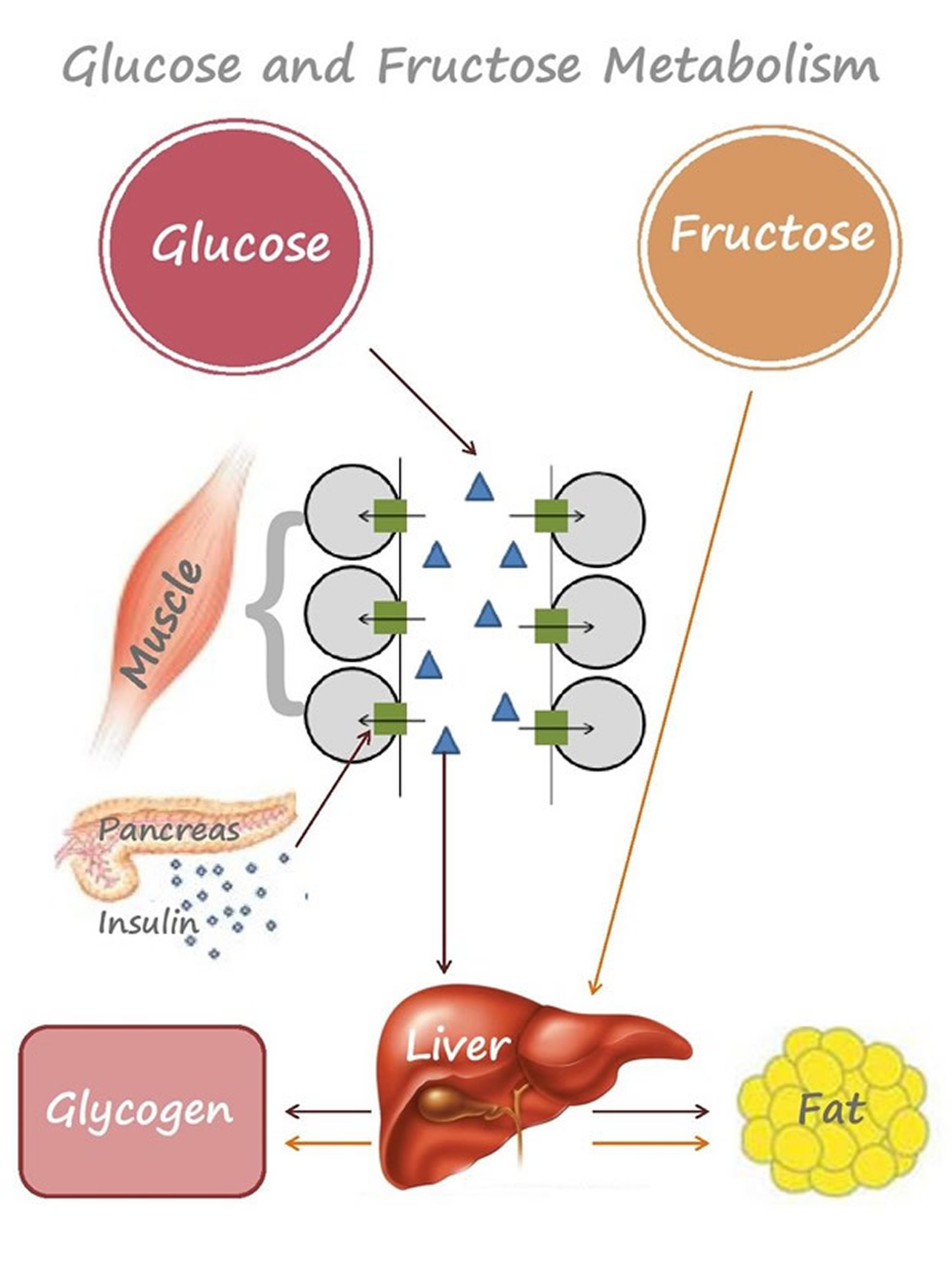
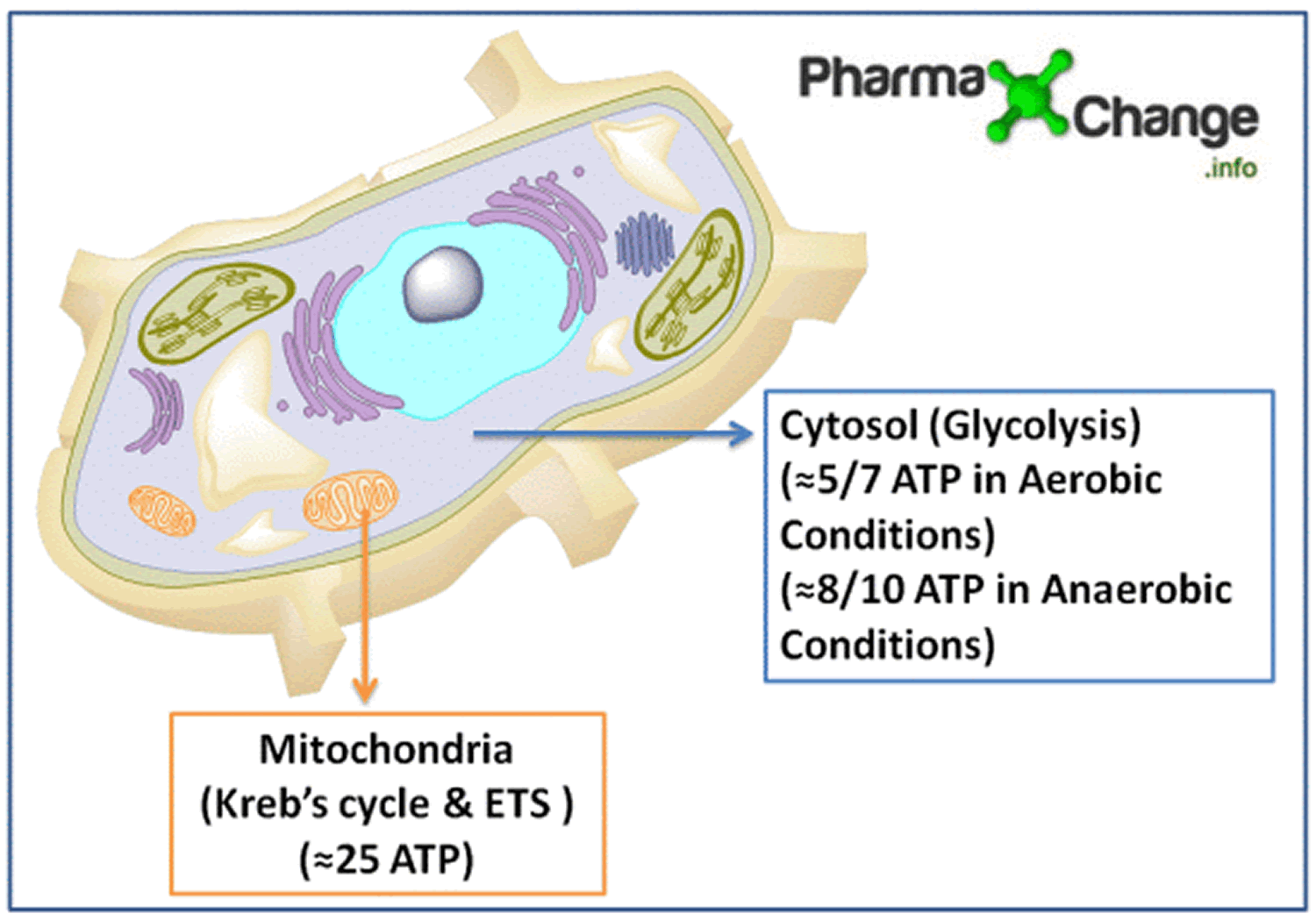
Glucose metabolism (Cellular respiration) can take place either in the presence or absence of oxygen leaving separate end products respectively 4 and it involves 3 stages for the breakdown of glucose –
- Glycolysis,
- Kreb’s cycle and
- The electron transport system.
Glucoregulation is the maintenance of steady levels of glucose in the body and the hormone insulin is the primary regulatory signal in human and when present, it causes many tissue cells to take up glucose from the circulation, causes some cells to store glucose internally in the form of glycogen, causes some cells to take in and hold lipids, and in many cases controls cellular electrolyte balances and amino acid uptake as well. Its absence turns off glucose uptake into cells, reverses electrolyte adjustments, begins glycogen breakdown and glucose release into the circulation by some cells, begins lipid release from lipid storage cells. The level of circulatory glucose (known informally as “blood sugar”) is the most important signal to the insulin-producing cells. Because the level of circulatory glucose is largely determined by the intake of dietary carbohydrates, diet controls major aspects of metabolism via insulin. In humans, insulin is made by beta cells in the pancreas, fat is stored in adipose tissue cells, and glycogen is both stored and released as needed by liver cells. Regardless of insulin levels, no glucose is released to the blood from internal glycogen stores from muscle cells.
The hormone glucagon, on the other hand, has an effect opposite to that of insulin, forcing the conversion of glycogen in liver cells to glucose, which is then released into the blood. Muscle cells, however, lack the ability to export glucose into the blood. The release of glucagon is precipitated by low levels of blood glucose. Other hormones, notably growth hormone, cortisol, and certain catecholamines (such as epinepherine) have glucoregulatory actions similar to glucagon.
Typically, a breakdown of one molecule of glucose by aerobic respiration (i.e. involving both glycolysis and Krebs cycle) is about 33-35 ATP 4. This is categorized as:
- Anaerobic breakdown by Glycolysis – yielding 8-10 ATP
- Aerobic respiration by Krebs cycle – yielding 25 ATP
(Source 5).
Metabolism of glucose leads to its breakdown to pyruvate. This pyruvate can have one of the following three fates:
- Breaks down to lactic acid in the presence of enzyme lactate dehydrogenase in vigorously contracting skeletal muscles, in certain micro-organisms etc. (called lactic acid fermentation)(Anaerobic Respiration)
- Breaks down to ethanol and carbon dioxide in some plant tissues, invertebrates and microorganisms (e.g. yeast) (called alcoholic fermentation) (Anaerobic Respiration)
- Enters aerobic respiration in Kreb’s Cycle to form carbon dioxide and water. (Aerobic Respiration)
These glucose molecules can be either stored as starch or glycogen in the body. When excessive energy is demanded by the body, this reserve is broken down to produce energy aerobically or anaerobically.
- Aerobic Glucose Cellular respiration takes place in three steps:
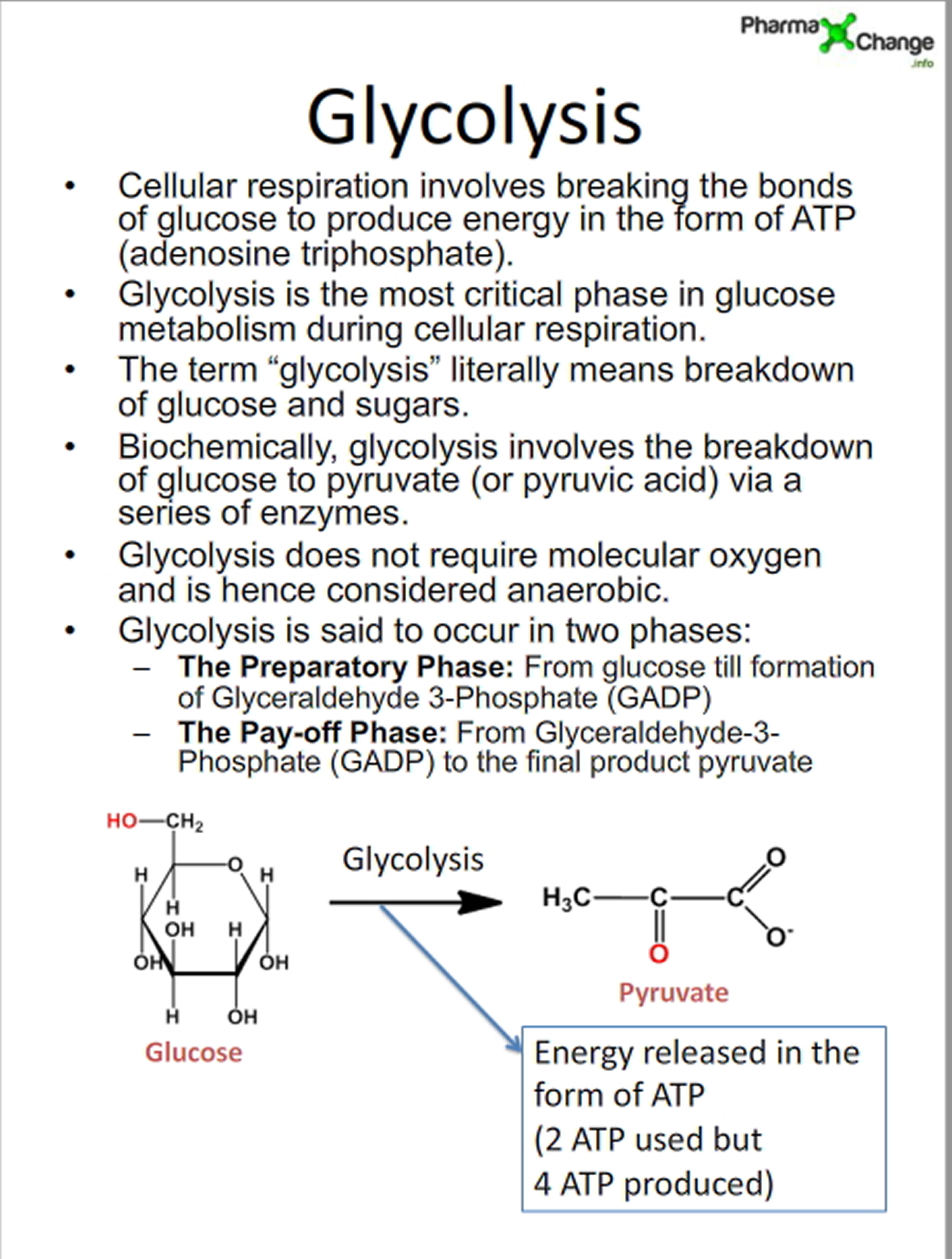
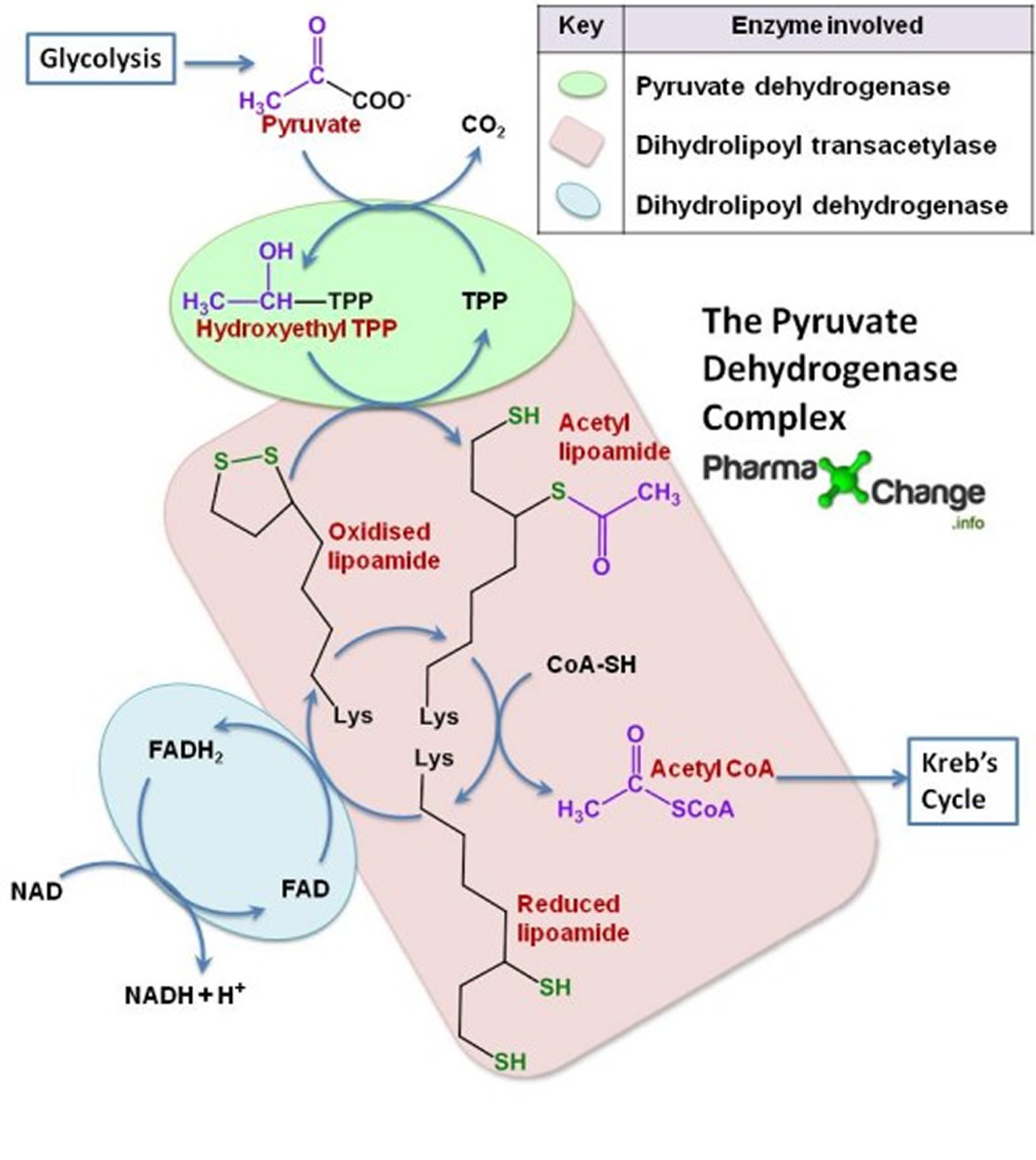
Step 1) Glycolysis (Embden-Meyerhof pathway) : All glycolysis reactions take place in the cytosol as all the enzymes required for the pathway are present here. In glycolysis one glucose molecule is converted to two pyruvate molecules with the release of energy in the form of ATP and NADH. This pathway is divided into two phases the preparatory phase and the pay-off phase. See : Glycolysis Diagram below.
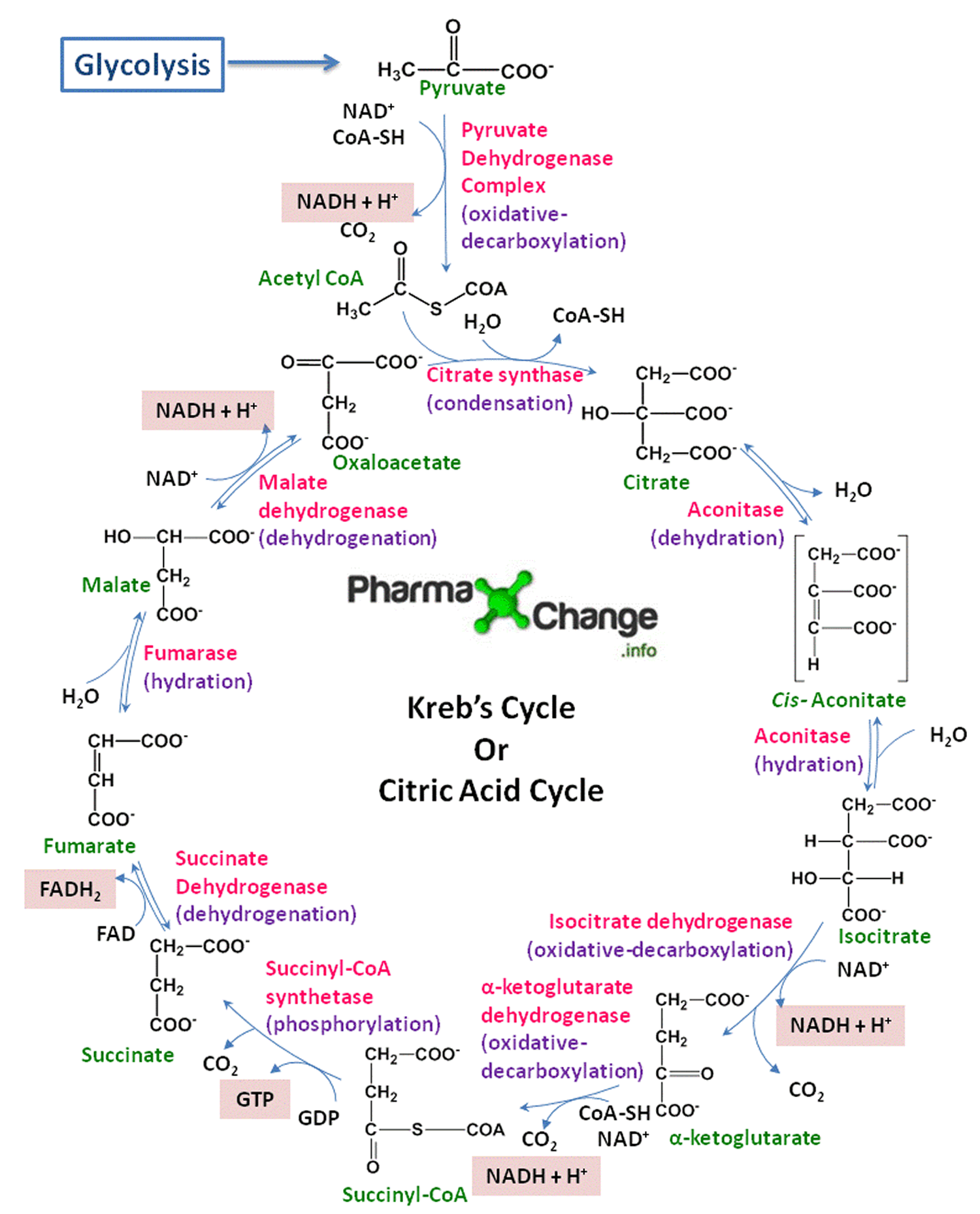
Step 2) Kreb’s Cycle or Citric Acid Cylce or Tricarboxylic Acid Cycle: Glycolysis of 1 molecule of glucose produces 2 molecules of pyruvate. The pyruvate formed in glycolysis then gets converted to acetyl CoA in the mitochondria. The net energy produced by the Kreb’s cycle for each molecule of pyruvate is doubled for each molecule of glucose. In the mitochondria enzymatic reactions occur known as Kreb’s Cycle to breakdown the acetyl CoA into CO2 and H2O with the release of energy in the form of ATP, NADH and FADH2. Kreb’s cycle is a very important step in the metabolic pathway as it produces about 60-70% of ATP for release of energy in the body. It directly or indirectly connects with all the other individual pathways in the body too. It takes place in the mitochondria as all the enzymes and co-enzymes required are present there. See : Kreb’s diagram below.
Thus net energy yield in Kreb’s cycle can be summarized as follows for each molecule of glucose:
| Reaction | Number of ATP or reduced coenzyme formed* | Number of ATP ultimately formed |
| 2 Pyruvate → 2 acetyl CoA | 2 NADH | 5 |
| 2 Isocitrate → 2 α- ketoglutarate | 2 NADH | 5 |
| 2 α- ketoglutarate → 2 succinyl CoA | 2 NADH | 5 |
| 2 Succinyl CoA → 2 succinate | 2 ATP | 2 |
| 2 Succinate → 2 fumarate | 2 FADH2 | 3 |
| 2 Malate → 2 oxaloacetate | 2 NADH | 5 |
| TOTAL | 25 ATP |
(Source 6).
(Source 6).
Step 3) Electron transport system: The reduced coenzymes (NADH and FADH2) formed in glycolysis and Kreb’s cycle are oxidized by giving up the protons and electrons to oxygen present in the mitochondria to synthesize ATP by oxidative phosphorylation.
Fructose
Fructose is also taken up into the blood from the gut, but in this case, the liver serves as a pre-processing organ that can convert fructose to glucose or fat. The liver can release the glucose and fat into the blood or store it as glycogen or fat depots, which, if sugars are consumed in excess, may lead to fatty liver disease and also increase risk for diabetes and cardiovascular disease.
There are also some noted interaction effects between glucose and fructose, in that glucose enables fructose absorption from the gut, while fructose can accelerate glucose uptake and storage in the liver.
If the sugar comes with its inherent fibre (as with whole fruit) then up to 30% of this sugar will not be absorbed. Instead, it will be metabolised by the microbes in the gut, which may improve microbial diversity and help prevent disease. The fibre will also mean a slower rise in blood glucose, which has shown to have positive health effects.
Absorption and Metabolism of Fructose
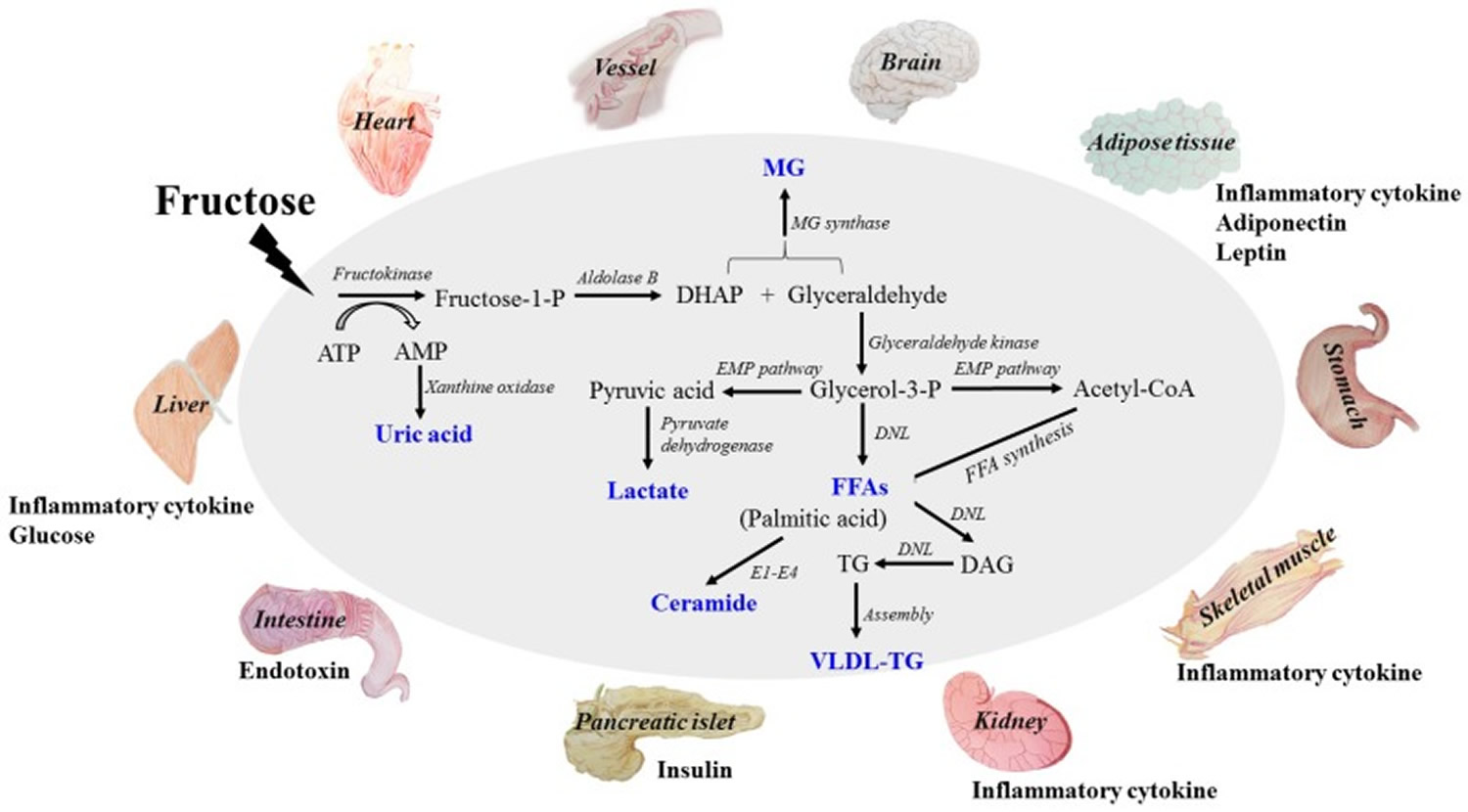
High dietary fructose is a major contributor to insulin resistance and metabolic syndrome, disturbing tissue and organ functions 7. Fructose is mainly absorbed into systemic circulation by glucose transporter 2 (GLUT2) and GLUT5, and metabolized in liver to produce glucose, lactate, triglyceride (TG), free fatty acid (FFA), uric acid (UA) and methylglyoxal (MG). Its extrahepatic absorption and metabolism also take place. High levels of these metabolites are the direct dangerous factors. During fructose metabolism, ATP depletion occurs and induces oxidative stress and inflammatory response, disturbing functions of local tissues and organs to overproduce inflammatory cytokine, adiponectin, leptin and endotoxin, which act as indirect dangerous factors. Fructose and its metabolites directly and/or indirectly cause oxidative stress, chronic inflammation, endothelial dysfunction, autophagy and increased intestinal permeability, and then further aggravate the metabolic syndrome with tissue and organ dysfunctions.
This diagram addresses fructose-induced metabolic syndrome, and the disturbance effects of direct and/or indirect dangerous factors on the functions of liver, adipose, pancreas islet, skeletal muscle, kidney, heart, brain and small intestine. The metabolites of fructose catabolism and the adverse effects of high fructose consumption on tissue and organ functions in a direct and/or indirect manner. Fructose is mainly metabolized in liver to produce glucose, lactate, triglyceride, free fatty acid (FFA), uric acid and methylglyoxal (MG). High levels of these metabolites are the direct dangerous factors. These dangerous factors impair the functions of local tissues and organs to overproduce inflammatory cytokine, adiponectin, leptin and endotoxin, which act as indirect dangerous factors. Meanwhile, glucose, insulin and ghrelin contents in system circulation are also disturbed. Fructose and its metabolites directly and/or indirectly cause oxidative stress, chronic inflammation, endothelial dysfunction, autophagy and increased intestinal permeability, and then further aggravate metabolic syndrome with tissue and organ dysfunctions. DHAP (dihydroxyacetone phosphate) ; TG (triglyceride) ; FFA (free fatty acid) ; UA (uric acid) ; MG (methylglyoxal) ; VLDL-TG (very low-density lipoprotein triglyceride). DNL (de novo lipogenesis). E1: Serine palmitoyl transferase; E2: 3-ketodihydrosphingosine reductase; E3: Ceramide synthase; E4: Dihydroceramide desaturase.
The breakdown of fructose in the liver does more than lead to the buildup of fat. It also:
- elevates triglycerides
- increases harmful LDL (so-called bad cholesterol)
- promotes the buildup of fat around organs (visceral fat)
- increases blood pressure
- makes tissues insulin-resistant, a precursor to diabetes
- increases the production of free radicals, energetic compounds that can damage DNA and cells.
- None of these changes are good for the arteries and the heart.
Researchers have begun looking at connections between fructose, fatty liver disease, and cardiovascular disease. The early results are in line with changes listed above due to the metabolism of fructose.
An article published in 2010 in The New England Journal of Medicine indicated that people with nonalcoholic fatty liver disease are more likely than those without it to have buildups of cholesterol-filled plaque in their arteries. They are also more likely to develop cardiovascular disease or die from it. In fact, people with nonalcoholic fatty liver disease are far more likely to die of cardiovascular disease than liver disease.
A report from the Framingham Heart Study has linked nonalcoholic fatty liver disease with metabolic syndrome, a constellation of changes that is strongly associated with cardiovascular disease. Other studies have linked fructose intake with high blood pressure.
A 6 year follow up study supported by the National Heart, Lung and Blood Institute published in the American Heart Association journal, Circulation, linked regular consumption of sugar-sweetened drinks (such as sodas and fruit drinks) to increased visceral fat, a type of body fat that may contribute to a higher risk of diabetes and heart disease 8.
Summary
Virtually every cell in the body can use glucose for energy. In contrast, only liver cells break down fructose. What happens to fructose inside liver cells is complicated. One of the end products is triglyceride, a form of fat. Uric acid and free radicals are also formed.
Fructose is widely found in natural foods, including fruits, vegetables and honeys, and is added to commercial food additives. Over consumption of fructose is a risk factor for the epidemic of metabolic syndrome, with dysfunctions in multiple tissues and organs including liver, adipose, pancreatic islet, skeletal muscle, kidney, heart, brain and intestine.
Fruit is good for you and is a minor source of fructose for most people.
Experts still have a long way to go to connect the dots between fructose and nonalcoholic fatty liver disease, obesity, diabetes, heart disease, and cancer. Higher intakes of fructose are associated with these conditions, but clinical trials have yet to show that it causes them. There are plenty of reasons to avoid sugary drinks and foods with added sugar, like empty calories, weight gain, and blood sugar swings.
The American Heart Association does not distinguish between types of added sugars — to your body they’re identical. Instead, focus on your total intake because eating too much added sugar leads to being overweight and obese, which are risk factors for diabetes 9. The American Heart Association also recommends that most women get no more than 100 calories a day of added sugar from any source, and that most men get no more than 150 calories a day of added sugar. That’s about 6 teaspoons of added sugar for women and 9 teaspoons for men by limiting the amount of sugar you get from sugar-sweetened drinks, pastries, desserts, breakfast cereals, and more, mainly to avoid gaining weight. The same strategy could also protect your liver and your arteries.
Carbohydrate Metabolism Disorders
Metabolism is the process your body uses to make energy from the food you eat. Food is made up of proteins, carbohydrates, and fats. Chemicals in your digestive system (enzymes) break the food parts down into sugars and acids, your body’s fuel. Your body can use this fuel right away, or it can store the energy in your body tissues. If you have a metabolic disorder, something goes wrong with this process.
Sucrose (table sugar) is made of two simpler sugars called glucose and fructose. Lactose (milk sugar) is made of glucose and galactose. Both sucrose and lactose must be broken down into their component sugars by enzymes before the body can absorb and use them. The carbohydrates in bread, pasta, rice, and other carbohydrate-containing foods are long chains of simple sugar molecules. These longer molecules must also be broken down by the body. If an enzyme needed to process a certain sugar is missing, the sugar can accumulate in the body, causing problems.
Carbohydrate metabolism disorders are a group of metabolic disorders. Normally your enzymes break carbohydrates down into glucose (a type of sugar). If you have one of these disorders, you may not have enough enzymes to break down the carbohydrates. Or the enzymes may not work properly. This causes a harmful amount of sugar to build up in your body. That can lead to health problems, some of which can be serious. Some of the disorders are fatal 10.
These disorders are inherited 10. Newborn babies get screened for many of them, using blood tests. If there is a family history of one of these disorders, parents can get genetic testing to see whether they carry the gene. Other genetic tests can tell whether the fetus has the disorder or carries the gene for the disorder.
Treatments may include special diets, supplements, and medicines. Some babies may also need additional treatments, if there are complications. For some disorders, there is no cure, but treatments may help with symptoms 10.
Hereditary Fructose Intolerance
Hereditary fructose intolerance is caused by lack of the enzyme needed to metabolize fructose. Very small amounts of fructose cause low blood sugar levels and can lead to kidney and liver damage 11.
In this disorder, the body is missing an enzyme that allows it to use fructose, a sugar present in table sugar (sucrose) and many fruits. As a result, a by-product of fructose accumulates in the body, blocking the formation of glycogen and its conversion to glucose for use as energy. Ingesting more than tiny amounts of fructose or sucrose causes low blood sugar levels (hypoglycemia), with sweating, confusion, and sometimes seizures and coma. Children who continue to eat foods containing fructose develop kidney and liver damage, resulting in jaundice, vomiting, mental deterioration, seizures, and death. Chronic symptoms include poor eating, failure to thrive, digestive symptoms, liver failure, and kidney damage. For most types of this disorder, early diagnosis and dietary restrictions started early in infancy can help prevent these more serious problems.
The diagnosis is made when a chemical examination of a sample of liver tissue determines that the enzyme is missing. Treatment involves excluding fructose (generally present in sweet fruits), sucrose, and sorbitol (a sugar substitute) from the diet. Severe attacks of hypoglycemia respond to glucose given by vein. Milder attacks are treated with glucose tablets, which should be carried by anyone who has hereditary fructose intolerance 11.
Mucopolysaccharidoses
Complex sugar molecules called mucopolysaccharides are essential parts of many body tissues. In mucopolysaccharidoses, the body lacks enzymes needed to break down and store mucopolysaccharides. As a result, excess mucopolysaccharides enter the blood and are deposited in abnormal locations throughout the body.
Mucopolysaccharidoses are a group of hereditary disorders in which complex sugar molecules are not broken down normally and accumulate in harmful amounts in the body tissues. The result is a characteristic facial appearance and abnormalities of the bones, eyes, liver, and spleen, sometimes accompanied by intellectual disability 11.
- Mucopolysaccharidoses occur when the body lacks enzymes needed to break down and store complex sugar molecules (mucopolysaccharides).
- Typically, symptoms include short stature, hairiness, stiff finger joints, and coarseness of the face.
- The diagnosis is based on symptoms and a physical examination.
- Although a normal life span is possible, some types cause premature death.
- A bone marrow transplant may help.
During infancy and childhood, short stature, hairiness, and abnormal development become noticeable. The face may appear coarse. Some types of mucopolysaccharidoses cause intellectual disability to develop over several years. In some types, vision or hearing may become impaired. The arteries or heart valves can be affected. Finger joints are often stiff.
A doctor usually bases the diagnosis on the symptoms and a physical examination. The presence of a mucopolysaccharidosis in other family members also suggests the diagnosis. Urine tests may help but are sometimes inaccurate. X-rays may show characteristic bone abnormalities. Mucopolysaccharidoses can be diagnosed before birth by using amniocentesis or chorionic villus sampling.
Prognosis and Treatment
The prognosis depends on the type of mucopolysaccharidosis. A normal life span is possible. Some types, usually those that affect the heart, cause premature death.
In one type of mucopolysaccharidosis, attempts at replacing the abnormal enzyme have had limited, temporary success. Bone marrow transplantation may help some people. However, death or disability often results, and this treatment remains controversial 11.
Disorders of Pyruvate Metabolism
Pyruvate is a substance that is formed in the processing of carbohydrates and proteins and that serves as an energy source for cells. Problems with pyruvate metabolism can limit a cell’s ability to produce energy and allow a buildup of lactic acid, a waste product. Many enzymes are involved in pyruvate metabolism. A hereditary deficiency in any one of these enzymes results in one of a variety of disorders, depending on which enzyme is missing.
Pyruvate metabolism disorders are caused by a lack of the ability to metabolize a substance called pyruvate. These disorders cause a buildup of lactic acid and a variety of neurologic abnormalities 11.
- A deficiency in any one of the enzymes involved in pyruvate metabolism leads to one of many disorders.
- Symptoms include seizures, intellectual disability, muscle weakness, and coordination problems.
- Some of these disorders are fatal.
- Some children are helped by diets that are either high in fat and low in carbohydrates or high in carbohydrates and low in protein.
Symptoms may develop any time between early infancy and late adulthood. Exercise and infections can worsen symptoms, leading to severe lactic acidosis. These disorders are diagnosed by measuring enzyme activity in cells from the liver or skin.
Pyruvate Dehydrogenase Complex Deficiency
This disorder is caused by a lack of a group of enzymes needed to process pyruvate. This deficiency results in a variety of symptoms, ranging from mild to severe. Some newborns with this deficiency have brain malformations. Other children appear normal at birth but develop symptoms, including weak muscles, seizures, poor coordination, and a severe balance problem, later in infancy or childhood. Intellectual disability is common.
This disorder cannot be cured, but some children are helped by a diet that is high in fat and low in carbohydrates 11.
Absence of Pyruvate Carboxylase
Pyruvate carboxylase is an enzyme. A lack of this enzyme causes a very rare condition that interferes with or blocks the production of glucose from pyruvate in the body. Lactic acid and ketones build up in the blood. Often, this disease is fatal. Children who survive have seizures and severe intellectual disability, although there are recent reports of children with milder symptoms. There is no cure, but some children are helped by eating frequent carbohydrate-rich meals and restricting dietary protein 11.
Glycogen Storage Diseases
Glycogen is made of many glucose molecules linked together. The sugar glucose is the body’s main source of energy for the muscles (including the heart) and brain. Any glucose that is not used immediately for energy is held in reserve in the liver, muscles, and kidneys in the form of glycogen and is released when needed by the body.
Glycogen storage diseases occur when there is a defect in the enzymes that are involved in the metabolism of glycogen, resulting in growth abnormalities, weakness, and confusion 11.
- Glycogen storage diseases are caused by lack of an enzyme needed to change glucose into glycogen and break down glycogen into glucose.
- Typical symptoms include weakness, sweating, confusion, kidney stones, and stunted growth.
- The diagnosis is made by examining a piece of tissue under a microscope (biopsy).
Treatment depends on the type of glycogen storage disease and usually involves regulating the intake of carbohydrates.
There are many different glycogen storage diseases (also called glycogenoses), each identified by a roman numeral. These diseases are caused by a hereditary lack of one of the enzymes that is essential to the process of forming glucose into glycogen and breaking down glycogen into glucose. About 1 in 20,000 infants has some form of glycogen storage disease.
Types and Characteristics of Glycogen Storage Diseases
Name | Affected Organs, Tissues, or Cells | Symptoms |
Name | Affected Organs, Tissues, or Cells | Symptoms |
| Type O | Liver or muscle | Episodes of low blood sugar levels (hypoglycemia) during fasting if the liver is affected |
| von Gierke’s disease (type IA) | Liver and kidney | Enlarged liver and kidney, slowed growth, very low blood sugar levels, and abnormally high levels of acid, fats, and uric acid in blood |
| Type IB | Liver and white blood cells | Same as in von Gierke’s disease but may be less severe
Low white blood cell count, recurring infections, and inflammatory bowel disease |
| Pompe’s disease (type II) | All organs | Enlarged liver and heart and muscle weakness |
| Forbes’ disease (type III) | Liver, muscle, and heart | Enlarged liver or cirrhosis, low blood sugar levels, muscle damage, heart damage, and weak bones in some people |
| Andersen’s disease (type IV) | Liver, muscle, and most tissues | Cirrhosis, muscle damage, and delayed growth and development |
| McArdle disease (type V) | Muscle | Muscle cramps or weakness during physical activity |
| Hers’ disease (type VI) | Liver | Enlarged liver
Episodes of low blood sugar during fasting
Often no symptoms |
| Tarui’s disease (type VII) | Skeletal muscle and red blood cells | Muscle cramps during physical activity and red blood cell destruction (hemolysis) |
(Source 11)
Symptoms
Some of these diseases cause few symptoms. Others are fatal. The specific symptoms, age at which symptoms start, and their severity vary considerably among these diseases. For types II, V, and VII, the main symptom is usually weakness. For types I, III, and VI, symptoms are low levels of sugar in the blood and protrusion of the abdomen (because excess or abnormal glycogen may enlarge the liver). Low levels of sugar in the blood cause weakness, sweating, confusion, and sometimes seizures and coma. Other consequences for children may include stunted growth, frequent infections, and sores in the mouth and intestines.
Glycogen storage diseases tend to cause uric acid (a waste product) to accumulate in the joints, which can cause gout, and in the kidneys, which can cause kidney stones. In type I glycogen storage disease, kidney failure is common in the second decade of life or later.
Diagnosis and Treatment
The specific type of glycogen storage disease is diagnosed by examining a piece of muscle or liver tissue under a microscope (biopsy).
Treatment depends on the type of glycogen storage disease. For most types, eating many small carbohydrate-rich meals every day helps prevent blood sugar levels from dropping. For people who have glycogen storage diseases that cause low blood sugar levels, levels are maintained by giving uncooked cornstarch every 4 to 6 hours around the clock. For others, it is sometimes necessary to give carbohydrate solutions through a stomach tube all night to prevent low blood sugar levels from occurring at night.
Galactosemia
Galactose is a sugar that is present in milk and in some fruits and vegetables. A deficient enzyme or liver dysfunction can alter the metabolism, which can lead to high levels of galactose in the blood (galactosemia). There are different forms of galactosemia, but the most common and the most severe form is referred to as classic galactosemia.
Galactosemia (a high blood level of galactose) is caused by lack of one of the enzymes necessary for metabolizing galactose, a sugar present in lactose (milk sugar). A metabolite that is toxic to the liver and kidneys builds up. The metabolite also damages the lens of the eye, causing cataracts 11.
- Galactosemia is caused by lack of one of the enzymes needed to metabolize the sugar in milk.
- Symptoms include vomiting, jaundice, diarrhea, and abnormal growth.
- The diagnosis is based on a blood test.
- Even with adequate treatment, affected children still develop mental and physical problems.
- Treatment involves completely eliminating milk and milk products from the diet.
What causes galactosemia ?
Classic galactosemia is a rare genetic metabolic disorder. The gene defect for Galactosemia is a recessive genetic trait. This faulty gene only emerges when two carriers have children together and pass it to their offspring. For each pregnancy of two such carriers, there is a 25% chance that the child will be born with the disease and a 50% chance that the child will be a carrier for the gene defect. A child born with classic galactosemia inherits a gene for galactosemia from both parents, who are carriers. A child with Duarte galactosemia inherits a gene for classic galactosemia from one parent and a Duarte variant gene from the other parent 12.
Symptoms
Newborns with galactosemia seem normal at first but, within a few days or weeks, lose their appetite, vomit, become jaundiced, have diarrhea, and stop growing normally. White blood cell function is affected, and serious infections can develop. If treatment is delayed, affected children remain short and become intellectually disabled or may die.
Diagnosis
Galactosemia is detectable with a blood test. This test is done as a routine screening test for newborns in all states in the United States. Before conception, adults with a sibling or child known to have the disorder can be tested to find out whether they carry the gene that causes the disease. If two carriers conceive a child, that child has a 1 in 4 chance of being born with the disease.
Prognosis
If galactosemia is recognized at birth and adequately treated, liver and kidney problems do not develop, and initial mental development is normal. However, even with adequate treatment, children with galactosemia may have a lower intelligence quotient (IQ) than their siblings, and they often have speech problems. Girls often have ovaries that do not function, and only a few are able to conceive naturally. Boys, however, have normal testicular function.
Treatment
Galactosemia is treated by completely eliminating milk and milk products—the source of galactose—from an affected child’s diet. Galactose is also present in some fruits, vegetables, and sea products, such as seaweed. Doctors are not sure whether the small amounts in these foods cause problems in the long term. People who have the disorder must restrict galactose intake throughout life.
References- Sugar Science. How The Body Metabolizes Sugar By Royal Society of New Zealand. http://sugarscience.ucsf.edu/sugar-metabolism.html?searched=sugar+metabolism&advsearch=allwords&highlight=ajaxSearch_highlight+ajaxSearch_highlight1+ajaxSearch_highlight2
- Harvard University. Harvard Health Publication. Abundance of fructose not good for the liver, heart. Published: September, 2011. http://www.health.harvard.edu/heart-health/abundance-of-fructose-not-good-for-the-liver-heart
- American Heart Association. Sugar 101. http://www.heart.org/HEARTORG/HealthyLiving/HealthyEating/Nutrition/Sugar-101_UCM_306024_Article.jsp
- Pharma Exchange October 9, 2013. Energetics of Cellular Respiration (Glucose Metabolism). http://pharmaxchange.info/press/2013/10/energetics-of-cellular-respiration-glucose-metabolism/
- Pharma Exchange Info published October 9, 2013. Energetics of Cellular Respiration (Glucose Metabolism) – http://pharmaxchange.info/press/2013/10/energetics-of-cellular-respiration-glucose-metabolism/
- Pharma Exchange Info. Kreb’s Cycle or Citric Acid Cycle or Tricarboxylic Acid Cycle (with Animation) – http://pharmaxchange.info/press/2013/09/krebs-cycle-citric-acid-cycle-tricarboxylic-acid-cycle-animation/
- Nutrients. 2017 Mar 29;9(4). pii: E335. doi: 10.3390/nu9040335. High Dietary Fructose: Direct or Indirect Dangerous Factors Disturbing Tissue and Organ Functions. https://www.ncbi.nlm.nih.gov/pubmed/28353649
- Circulation. 2016;CIRCULATION AHA.115.018704. Sugar-Sweetened Beverage Consumption is Associated With Change of Visceral Adipose Tissue Over 6 Years of Follow-Up. http://circ.ahajournals.org/content/early/2016/01/06/CIRCULATIONAHA.115.018704
- American Heart Association. Is high-fructose corn syrup worse than other added sugars ? http://heartinsight.heart.org/Spring-2016/Is-high-fructose-corn-syrup-worse-than-other-added-sugars/
- U.S. National Library of Medicine. Medline Plus. Carbohydrate Metabolism Disorders. https://medlineplus.gov/carbohydratemetabolismdisorders.html
- Merck Sharp & Dohme Corp. Merck Manual. Disorders of Carbohydrate Metabolism. https://www.merckmanuals.com/home/children-s-health-issues/hereditary-metabolic-disorders/disorders-of-carbohydrate-metabolism
- American Liver Foundation. Galactosemia. http://www.liverfoundation.org/abouttheliver/info/galactosemia/