What is hyperaldosteronism
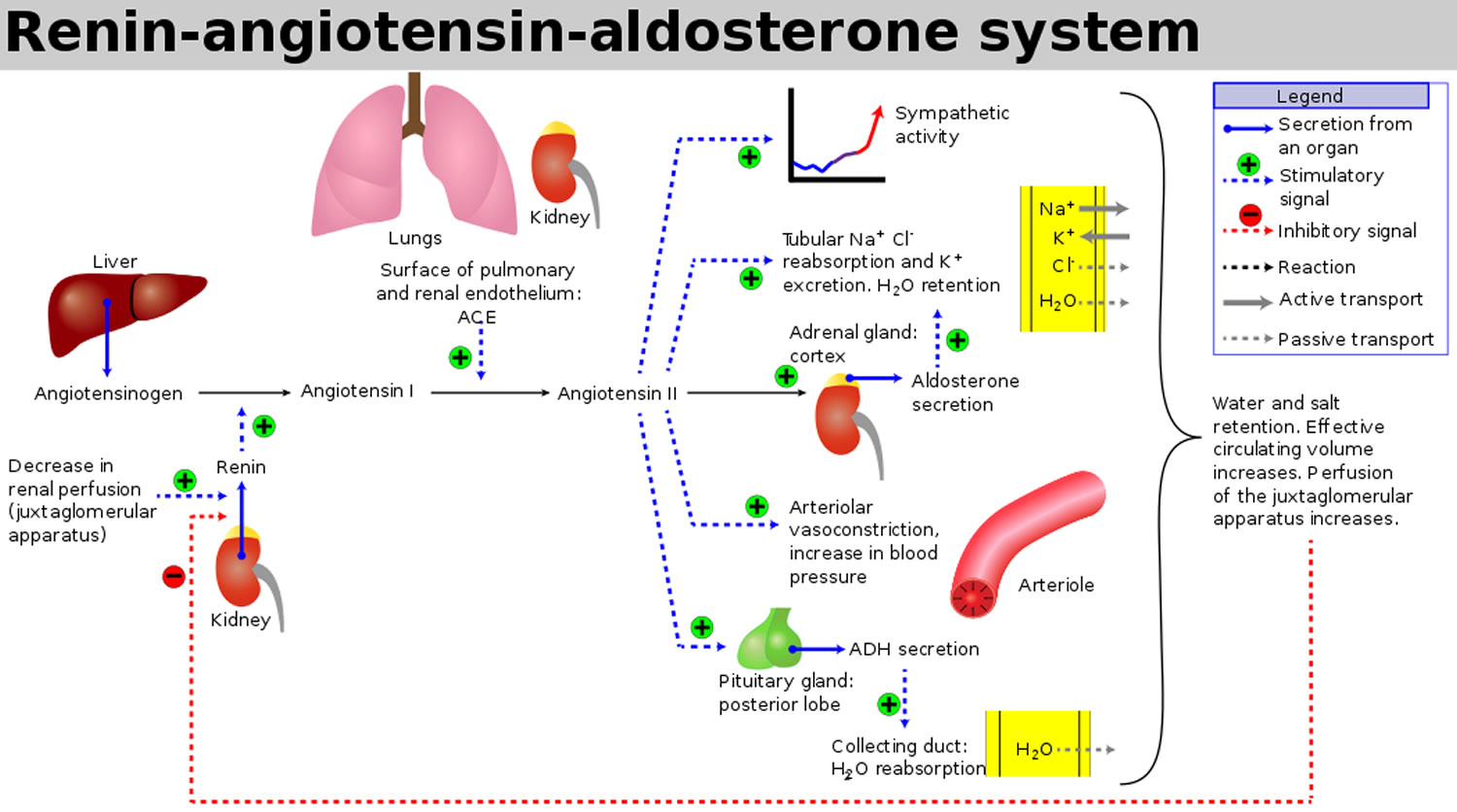
Hyperaldosteronism is a disorder in which the adrenal gland releases too much of the hormone aldosterone into the blood. Aldosterone is produced by the adrenal glands located at the top of each kidney, in their outer portion (called the adrenal cortex). Aldosterone stimulates the retention of sodium (salt) and the elimination of potassium by the kidneys. Renin is produced by the kidneys and controls the activation of the hormone angiotensin, which stimulates the adrenal glands to produce aldosterone. The kidneys release renin when there is a drop in blood pressure or a decrease in sodium chloride concentration in the tubules in the kidney. Renin cleaves the blood protein angiotensinogen to form angiotensin I, which is then converted by a second enzyme to angiotensin II. Angiotensin II causes blood vessels to constrict, and it stimulates aldosterone production. Overall, this raises blood pressure and keeps sodium and potassium at normal levels.
Hyperaldosteronism can initially present as essential and refractory hypertension and can often go undiagnosed 1.
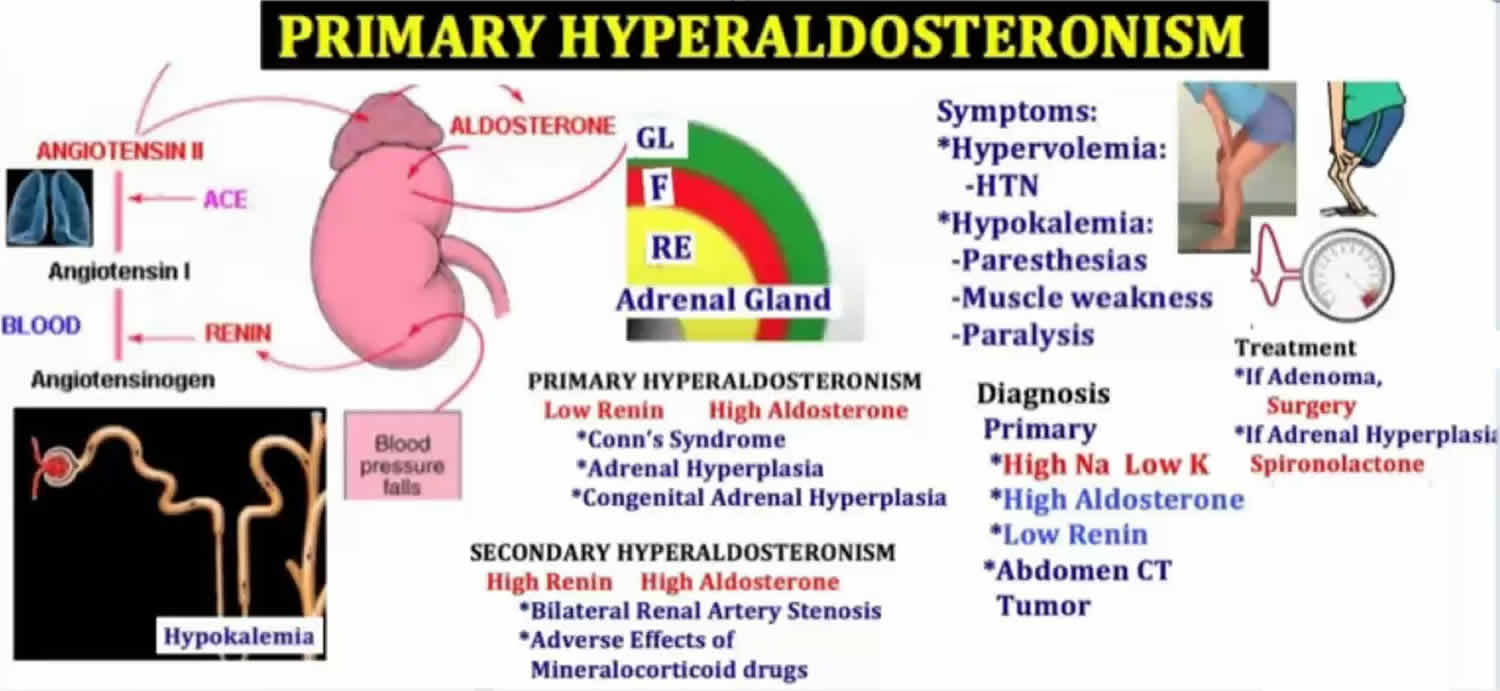
Hyperaldosteronism can be primary or secondary, both presenting similarly but differentiated by a set of lab values and diagnostic studies. The distinction between primary and secondary causes of hyperaldosteronism is of importance, as the manifestations, as well as the subsequent testing and treatment, differ.
Primary hyperaldosteronism is the culprit of less than 1% of hypertensive patients. Although a higher prevalence has been considered, studies have shown an overestimation of cases. Secondary hyperaldosteronism is diagnosed even less often than primary. Both present more frequently in women.
Have your blood pressure checked regularly, especially if you have risk factors for high blood pressure. Ask your doctor about the possibility of having primary hyperaldosteronism if:
- You’re age 45 or older
- You have a family history of high blood pressure
- You have high blood pressure that began at age 44 or younger
- You’re overweight
- You have a sedentary lifestyle
- You use tobacco
- You drink a lot of alcohol
- You have dietary imbalances (too much salt, not enough potassium)
Primary hyperaldosteronism
Primary hyperaldosteronism is due to a problem of the adrenal glands (more specifically the zona glomerulosa) themselves, which causes them to produce too much aldosterone. Primary hyperaldosteronism can present more commonly as a primary tumor in the gland known as Conn syndrome or bilateral hyperplasia 1. Rarer forms are unilateral adrenal hyperplasia, ectopic aldosterone-secreting tumors, aldosterone-producing adrenocortical carcinomas, and familial hyperaldosteronism type 1.
Most cases of primary hyperaldosteronism are caused by a noncancerous (benign) tumor of the adrenal gland. Primary hyperaldosteronism is most common in people 30 to 50 years old.
Common conditions causing the overproduction of aldosterone include:
- A benign growth in an adrenal gland (aldosterone-producing adenoma) — a condition also known as Conn’s syndrome
- Overactivity of both adrenal glands (idiopathic hyperaldosteronism)
In rare cases, primary hyperaldosteronism may be caused by:
- A cancerous (malignant) growth of the outer layer (cortex) of the adrenal gland (adrenal cortical carcinoma)
- A rare type of primary aldosteronism called glucocorticoid-remediable aldosteronism that runs in families and causes high blood pressure in children and young adults
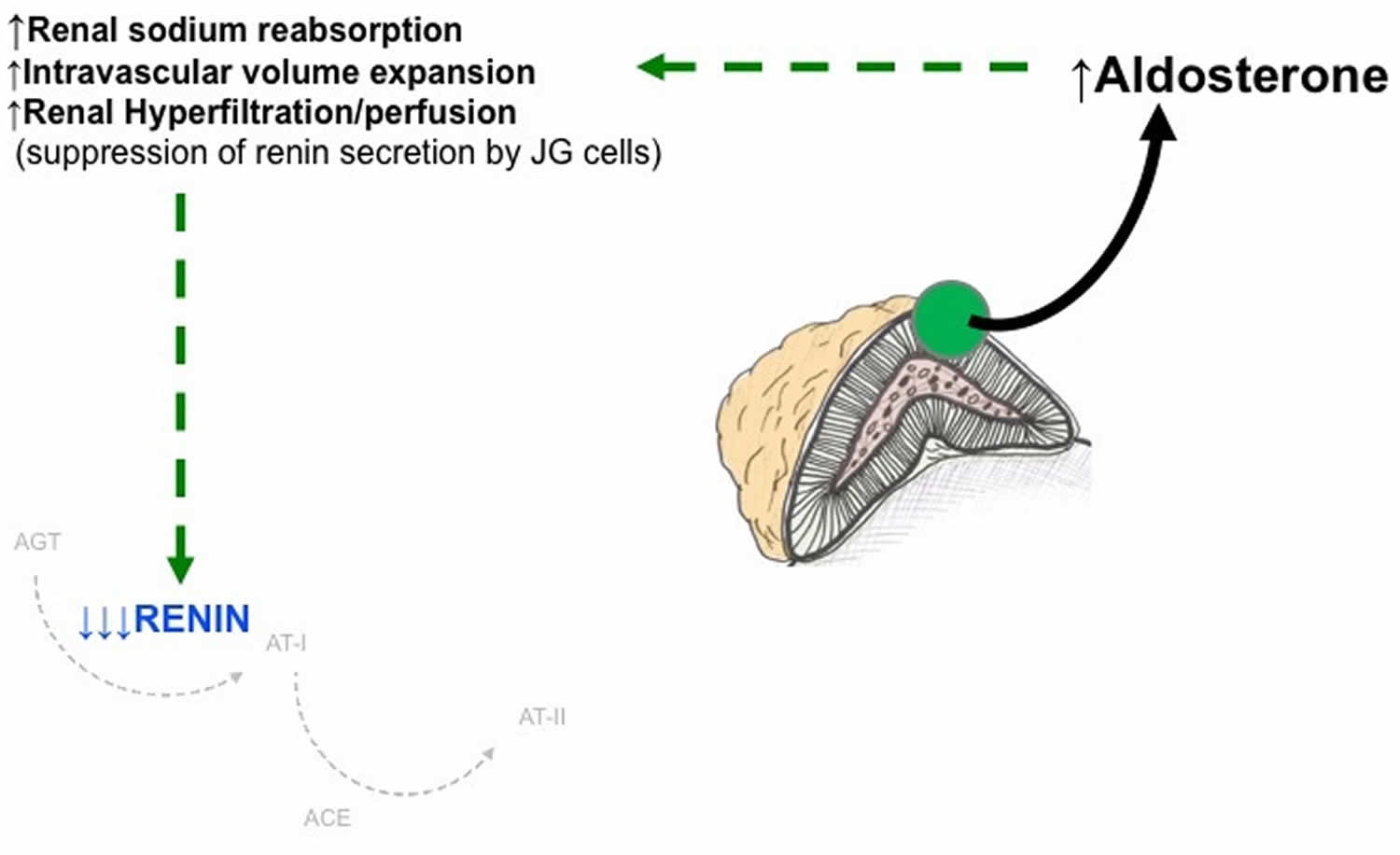
In primary hyperaldosteronism, the plasma renin activity is suppressed (hyporeninemic hyperaldosteronism or renin-independent aldosteronism), and the plasma aldosterone to renin activity ratio is elevated (see Figure 3 below) 2.
Figure 1. Renin angiotensin aldosterone system
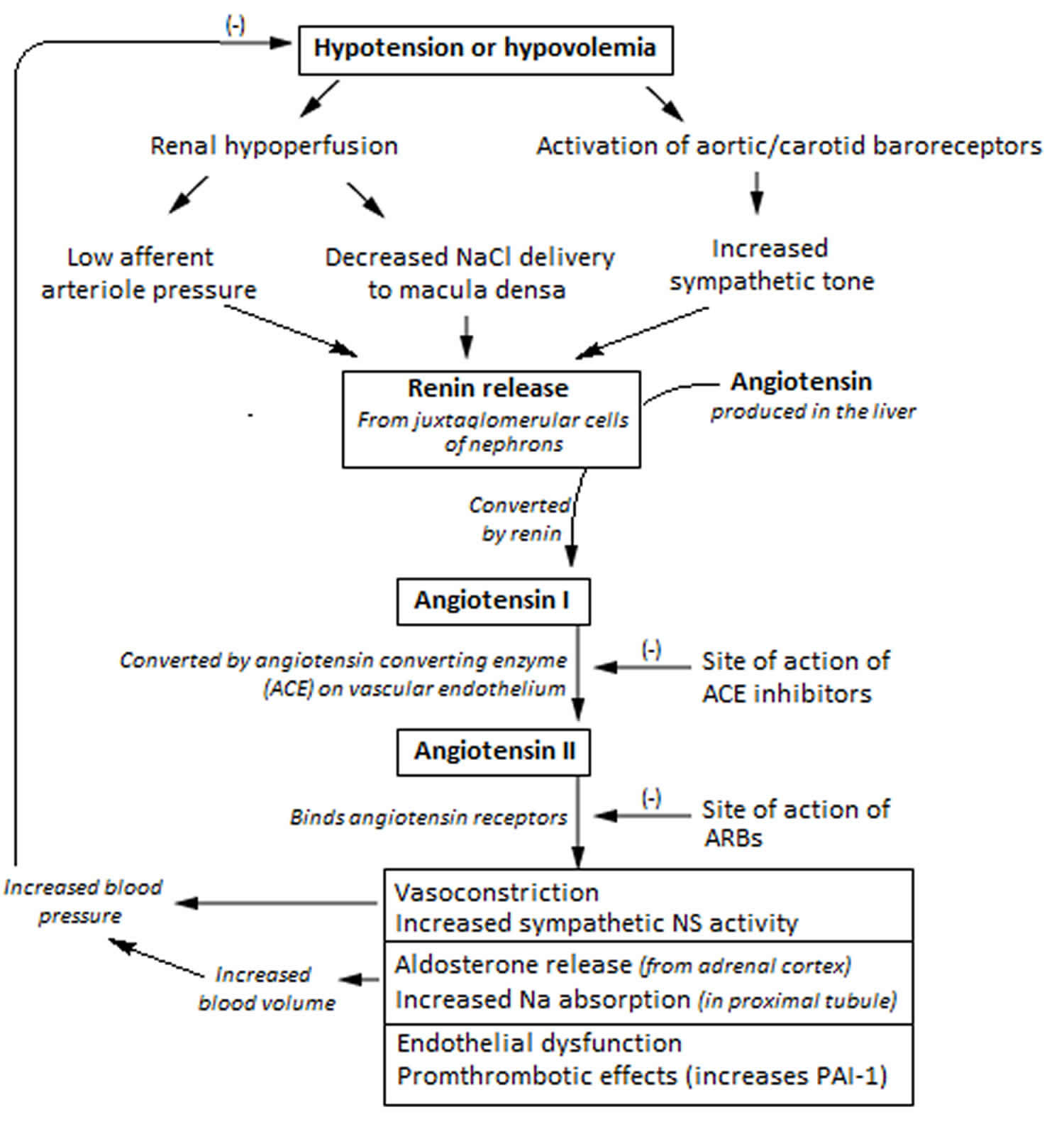
Figure 2. Renin angiotensin aldosterone system mechanism of action
Figure 3. Primary hyperaldosteronism
Secondary hyperaldosteronism
Secondary hyperaldosteronism is caused by a problem elsewhere in the body that causes the adrenal glands to release too much aldosterone. These problems can be with genes, diet, or a medical disorder such as with the heart, liver, kidneys, or high blood pressure. In secondary hyperaldosteronism, increased activation the renin-angiotensin system (RAS) is the initiating event, and activation of the renin-angiotensin system (RAS) then results in excess aldosterone production (hyperreninemic hyperaldosteronism or renin-dependent aldosteronism) 2. Therefore, secondary hyperaldosteronism can be a normal physiologic phenomenon (such as in states of systemic hypovolemia or hypoperfusion), or can manifest as a pathologic entity when activation of the renin-angiotensin system (RAS) is inappropriate relative to the state of the systemic vasculature 2.
Hyperaldosteronism causes
Primary hyperaldosteronism causes
Primary hyperaldosteronism occurs due to the excess aldosterone production by the adrenal gland. The most common cause in two-thirds of the patients occurs due to idiopathic bilateral adrenal hyperplasia 1. In one-third of patients, a tumor in the zona glomerulosa, known as Conn syndrome, can directly cause an increase in aldosterone 1. The clinical picture of these patients can range from asymptomatic to hypertension with hypokalemia.
Aldosterone-producing adenoma, bilateral adrenal hyperplasia and unilateral adrenal hyperplasia
It is currently estimated that aldosterone-producing adenoma or unilateral adrenal hyperplasia account for 30-40% of primary hyperaldosteronism cases, whereas bilateral adrenal hyperplasia accounts for the remaining 60% 3. Definitive diagnosis of the cause of primary hyperaldosteronism can be a challenge in individual patients; however, making the correct diagnosis is of utmost importance, since the treatment for each underlying cause may be different.
Aldosterone-producing adenomas are often small tumors, usually less than 2 cm in diameter. Histopathology of aldosterone-producing adenoma reveals hybrid cells which have histological features of both zona glomerulosa and zona fasciculata cells. Unilateral adrenal hyperplasia, sometimes referred to as primary adrenal hyperplasia, shares many biochemical features with aldosterone-producing adenoma. This diagnosis is often made based on evidence of unilateral production of aldosterone (primarily from adrenal vein sampling) in the absence of a discrete radiographic mass. Similar to aldosterone-producing adenoma, the hypertension and biochemical abnormalities with unilateral adrenal hyperplasia may be cured or substantially ameliorated with unilateral adrenalectomy 4.
Bilateral adrenal hyperplasia probably represents a spectrum of disorders 5. The extent of hyperaldosteronism is often milder in bilateral adrenal hyperplasia compared to aldosterone-producing adenoma, and consequently the severity of hypertension, hypokalemia and suppression of plasma renin activity is often less.
Glucocorticoid-remediable aldosteronism
Glucocorticoid-remediable aldosteronism (also known as familial hyperaldosteronism type 1) is an autosomal dominant disorder characterized by a chimeric duplication, whereby the 5’-promotor region of the 11b-hydroxylase gene (regulated by ACTH) is fused to the coding sequences of the aldosterone synthase gene in a recombination event. The result is that the aldosterone synthase gene (CYP11B2) is under the control of the promoter for the CYP11B1 gene, typically responsible for cortisol production under the regulation of ACTH. Aldosterone synthesis is therefore abnormally and solely regulated by ACTH 6.
Adrenal carcinoma
Adrenal carcinomas are a rare cause of primary aldosteronism. At the time of diagnosis, adrenal carcinomas are usually large (>4 cm) and may be producing one or multiple adrenal cortical hormones, including cortisol, aldosterone, and adrenal androgens.
Congenital adrenal hyperplasia
Another mineralocorticoid-excess state with low plasma renin activity is congenital adrenal hyperplasia (CAH). The most common cause of congenital adrenal hyperplasia is 21-hydroxylase deficiency, which can result in variable insufficiencies of cortisol and aldosterone. However, much rarer forms of congenital adrenal hyperplasia can result in hypermineralocorticoidism. For example, 11b-hydroxylase deficiency and 17a-hydroxylase deficiency can result in excess 11-deoxycorticosterone production and resultant excessive mineralocorticoid receptor activation 7.
Apparent Mineralocorticoid Excess and Liddle’s syndrome
Apparent Mineralocorticoid Excess results from abnormal activation of the Type 1 mineralocorticoid receptor in the kidney by cortisol, secondary to an acquired (licorice ingestion or chewing tobacco) or congenital deficiency of the renal isoform of the enzyme 11b-OH steroid dehydrogenase (11b-HSD). The 11b-HSD2 isoenzyme normally metabolizes cortisol to the inactive compound cortisone in the renal distal convoluted tubule 8. However if there is 11b-OH steroid dehydrogenase (11b-HSD) deficiency, the Type 1 mineralocorticoid receptor is no longer ‘protected’ from activation by cortisol. In Liddle’s syndrome, constitutive activation of the renal epithelial sodium channel (ENaC) results from activating mutations in the ENaC gene. In both Apparent Mineralocorticoid Excess and Liddle’s syndromes, the intrinsic renal abnormalities described lead to unregulated and excessive sodium reabsorption, and therefore a biochemical phenotype of suppressed plasma renin activity, hypokalemia, and undetectable levels of plasma aldosterone 8.
Secondary hyperaldosteronism causes
Secondary hyperaldosteronism occurs due to the excess stimulation of the renin-angiotensin system (RAS). Renal artery stenosis (either in the form of atherosclerosis or fibromuscular dysplasia) causes decreased blood flow through the kidneys, stimulating a false sense of hypovolemia and activating aldosterone secretion 1. In left-sided heart failure and cor pulmonale, a decrease in cardiac output causes aldosterone stimulation 1. Cirrhosis with ascites patients will produce decreased circulating fluid volume, resulting in less perfusion through the kidneys and causing an increase in aldosterone 1. Less common is a renin-producing tumor in the juxtaglomerular cells. Like primary, secondary hyperaldosteronism can present with a wide clinical range.
Usually Normo- or Hypotensive
The most common causes of secondary aldosteronism are medical illnesses that result from a reduction in perceived or effective circulating blood volume, such as congestive heart failure and nephrotic syndrome. Importantly, treatment and correction of the underlying medical illness and volume expansion results in eversal of the activated renin-angiotensin system (RAS). Secondary aldosteronism in a normotensive patient should also raise consideration for Gittleman’s and Barter’s syndrome 2.
Diuretic use can also cause secondary aldosteronism. The findings can mimic those seen in renovascular hypertension, especially in a hypertensive patient. With chronic diuretic use, moderate to severe extracellular and intravascular volume depletion results in renal hypoperfusion, increased release of renin, and subsequently excessive aldosterone production. In rare occasions, surreptitious use of diuretics can produce misleading biochemical findings. A high degree of suspicion should be present in the appropriate setting, such as unexplained hypokalemia in a medical or paramedical worker or an individual attempting to lose weight using pharmacologic methods.
Usually Hypertensive
It is important to distinguish renal vascular disease from renal vascular hypertension. While a large proportion of the adult population may have renal vascular disease (defined as a 50% or greater decrease in renal artery luminal diameter), only a small portion of these patients experience critical and clinically relevant renal hypoperfusion and ischemia 9. Therefore, documentation of both structural and functional abnormalities is required before therapeutic intervention in such patients.
Renovascular hypertension is defined as hypertension associated with either unilateral or bilateral ischemia of the renal parenchyma. There are numerous causes of this disorder. Atherosclerosis of the renal arteries is the most common, accounting for 90% of cases. Fibromuscular dysplasia accounts for less than 10% of cases 9. In these disorders, decreased renal perfusion causes tissue hypoxia and decreased perfusion pressure, thereby stimulating renin release from the juxtaglomerular cells, resulting in secondary aldosterone secretion. Coarctation of the aorta can produce a similar pathophysiology due to renal hypoperfusion.
Although renal vascular hypertension can affect patients of all ages, it is commonly seen in older adults (>50 years) due to the increased prevalence of atherosclerosis in this population. When found in patients less than 50 years of age, renal vascular hypertension is more common in women, usually as a result of fibromuscular dysplasia of one of both of the renal arteries 9.
In very rare cases, juxtaglomerular cell tumors of the kidney that hypersecrete renin have been described 10. Such patients often have severe hypertension, accompanied by marked elevation of renin and aldosterone levels, hypokalemia, and a mass lesion in the kidney. Confirmation includes documentation of unilateral renin secretion in the absence of renal artery stenosis. While very rare, such cases are important to diagnose, as surgical removal of the tumor can be curative.
Hyperaldosteronism symptoms
Primary and secondary hyperaldosteronism have common symptoms, including:
- Moderate to severe high blood pressure
- High blood pressure that takes several medications to control (resistant hypertension)
- High blood pressure along with a low potassium level (hypokalemia)
- Low level of potassium in the blood
- Feeling tired all the time
- Headache
- Muscle weakness
- Numbness
Resistant hypertension are the most common presenting symptoms for patients with hyperaldosteronism. Later on the consequences of hyperaldosteronism develop such as fatigue, headache, polyuria, and polydipsia commonly occur with the hypertension. Metabolic alkalosis is frequently observed in these patients as they are susceptible to the same mechanism that occurs in contraction alkalosis.
Concurrently, hypokalemia historically has been found in a majority of cases, but recent studies have shown a less than 40% correlation 1. Mild hypernatremia and hypomagnesemia also are found in these patients 1.
More rarely, muscle weakness and spasms occur due to the hypokalemia of the hyperaldosteronism. Numbness and paroxysmal paralysis also have been reported.
Signs of hyperaldosteronism:
- Due to hypertension
- Headaches
- Retinopathy (rare)
- Due to hypokalemia
- Neuromuscular symptoms (cramps, paresthesias, weakness)
- Nephrogenic diabetes insipidus
- Cardiac arrhythmia
- Glucose intolerance / impaired insulin secretion
- Due to direct actions of aldosterone on the cardiovascular system
- Cardiac Hypertrophy/Fibrosis
- Vascular smooth muscle hypertrophy
- Due to a reset osmostat
- Mild hypernatremia
Hyperaldosteronism complications
Hyperaldosteronism can lead to high blood pressure and low potassium levels. These complications in turn can lead to other problems. The most common complication and comorbidity associated with hyperaldosteronism patients is the increased risk of cardiovascular mortality. This is associated with excessive aldosterone secretion and can present as atrial fibrillation, left ventricular hypertrophy, myocardial infarction, and stroke.
Primary hyperaldosteronism can cause very high blood pressure, which can damage many organs, including the eyes, kidneys, heart and brain.
Erection problems and gynecomastia (enlarged breasts in men) may occur with long-term use of medicines to block the effect of hyperaldosteronism.
Problems related to high blood pressure
Persistently elevated blood pressure can lead to problems with your heart and kidneys, including:
- Heart attack
- Heart failure
- Left ventricular hypertrophy — enlargement of the muscle that makes up the wall of the left ventricle, one of your heart’s pumping chambers
- Stroke
- Kidney disease or kidney failure
- Premature death
High blood pressure caused by primary hyperaldosteronism carries a higher risk of cardiovascular complications than do other types of high blood pressure. This excess risk is due to the high aldosterone levels, which can cause heart and blood vessel damage independent of complications related to high blood pressure.
Problems related to low potassium levels
Some, but not all, people with primary hyperaldosteronism have low potassium levels (hypokalemia). Mild hypokalemia may not cause any symptoms, but very low levels of potassium can lead to:
- Weakness
- Cardiac arrhythmias
- Muscle cramps
- Excess thirst or urination
Hyperaldosteronism diagnosis
Your doctor will perform a physical exam and ask about your symptoms.
Tests that may be ordered to diagnose hyperaldosteronism include:
- Abdominal CT scan
- ECG
- Blood aldosterone level
- Blood renin activity
- Blood potassium level
- Urinary aldosterone
- Kidney ultrasound
Laboratory findings are typically consistent with hypokalemia (low blood potassium), mild hypernatremia (high blood sodium), and mild hypomagnesemia (low blood magnesium).
Plasma renin concentration (PRC) and plasma renin activity (PRA) are initial laboratory measurements ordered when hyperaldosteronism is suspected. In primary hyperaldosteronism, the plasma renin concentration and plasma renin activity will be decreased (less than 1 ng/mL/hour and undetectable, respectively) as the increased aldosterone originates from the zona glomerulosa itself and not an extrinsic pathway. In secondary hyperaldosteronism, the plasma renin concentration and plasma renin activity will be increased, as renin is the mediator for its hyperaldosteronism effect. These levels are measured in the morning after patients have been out of bed for more than two hours and sitting for at least 5 minutes.
Plasma aldosterone concentration (PAC) to plasma renin activity ratio is used to confirm a suspicion of primary hyperaldosteronism. Specific criteria vary from institution to institution, but studies support an elevated plasma aldosterone concentration/plasma renin activity (greater than 30) and plasma aldosterone concentration (greater than 20 ng/dL) levels with a sensitivity and specificity of 90%. However, a plasma aldosterone concentration/plasma renin activity ratio of greater than 20 and plasma aldosterone concentration greater than 15 ng/dL have been reported to be sufficient. In secondary hyperaldosteronism, both plasma renin activity and PRC are increased, but the plasma aldosterone concentration/plasma renin activity is less than 10.
Twenty-four-hour urine collection is controversial in hyperaldosteronism, but it is used to detect inappropriate potassium wasting (greater than 30 mEq/day). This is primarily used to rule out extrarenal losses and diuretic abuse for explanations of hypokalemia with insignificant to mild aldosterone increases.
Aldosterone suppression testing is typically necessary for confirmation and is achieved with sodium loading and consequent aldosterone measurement. This can be performed with oral sodium loading over 3 days (5000 mg total in diet or 90 mEq of sodium as a tablet) and a urine aldosterone excretion measurement of greater than 12 mcg/day to confirm hyperaldosteronism. Another method is a two-liter intravenous isotonic saline infusion over 4 hours. With the saline infusion, plasma aldosterone concentration levels greater than 10 ng/dL are consistent with hyperaldosteronism; however, saline infusion has a false negative rate of 30%.
Radiological imaging such as CT scan can be used to differentiate adenomas from bilateral hyperplasia, but studies have found that CT cannot differentiate the two reliably.
Adrenal vein sampling. A procedure to insert a catheter into the veins of the adrenal glands may need to be done. This helps check which of the two adrenal glands is making too much aldosterone. Adrenal vein sampling is used to differentiate unilateral from bilateral pathology if a CT scan is not diagnostic. An incongruence in levels (usually a four-fold increase on the side of the adenoma) indicates unilateral origin, whereas equivalent levels indicate bilateral origin.
While histopathology is not a common diagnostic tool for hyperaldosteronism, studies have found a correlation between the immunohistochemical staining of CYP-11B2 in aldosterone-producing adenomas 1. This is primarily used to differentiate the variable types of adenomas such as unilateral micronodules, subtype classifications of adrenal adenomas, multiple aldosterone-producing cell clusters, and aldosterone-producing adenomas.
Who to screen for hyperaldosteronism
The Endocrine Society has published clinical practice guidelines for the diagnosis and treatment of patients with primary hyperaldosteronism 11. The task force recommends screening the following subtypes of patients deemed to be at high-risk for primary hyperaldosteronism:
- Patients with sustained blood pressure >150/100 mmHg on three or more measurements on different days.
- Patients with hypertension resistant to three or more anti-hypertensive medications or patients requiring four or more anti-hypertensive medications to attain blood pressure control.
- Patients with hypertension and sleep apnea.
- Patients with hypertension associated with either spontaneous or diuretic-induced hypokalemia.
- Patients with hypertension and an incidentally discovered adrenal adenoma.
- Patients with hypertension with a family history of early-onset hypertension or cerebrovascular accident at age less than 40 years.
- All hypertensive first-degree relatives of patients with primary hyperaldosteronism, although there is insufficient data from prospective studies to support this recommendation.
Glucocorticoid-remediable aldosteronism should be considered in patients with early-onset hypertension (<20 years of age) in the setting of a suppressed plasma renin activity. A family history of primary hyperaldosteronism or early cerebral hemorrhage (<40 years of age) should also raise suspicion for glucocorticoid-remediable aldosteronism. Screening of glucocorticoid-remediable aldosteronism kindreds has revealed that most affected individuals are not hypokalemic 12.
Confirmation tests
If the aldosterone-renin test suggests that you might have primary hyperaldosteronism, you’ll need another test to confirm the diagnosis, such as one of the following:
- Oral salt loading. You’ll follow a high-sodium diet for three days before your doctor measures aldosterone and sodium levels in your urine.
- Saline infusion test. Your aldosterone levels are tested after sodium mixed with water (saline) is infused into your bloodstream for several hours.
- Fludrocortisone suppression test. After you’ve followed a high-sodium diet and taken fludrocortisone — which mimics the action of aldosterone — for several days, aldosterone levels in your blood are measured.
When prescribing the oral sodium loading test to confirm primary hyperaldosteronism, patients should be instructed to consume a high sodium (200 mmol/day) diet for 4 days. This is best accomplished by adding 4 boullion packets per day to a regular diet (each packet contains 1100 mg, or 48 mmol, of sodium). Sodium chloride tablets can also be used, though in our experience these may be poorly tolerated due to gastrointestinal upset. On the fourth day of high dietary sodium intake, a 24-hour urine collection for urinary aldosterone (or aldosterone excretion rate), creatinine, and sodium is collected. Oral salt loading should result in extra- and intra-vascular volume expansion and renin-angiotensin system (RAS) suppression in normal individuals. Aldosterone excretion greater than 10-12 ug/d in the presence of a urinary sodium excretion greater than 200 mmol/24 hours confirms the diagnosis of primary hyperaldosteronism 11. The advantage of oral sodium loading is that it is easier for both the patient and clinician, as it can be performed on an outpatient basis without using hospital resources. However, this should not be performed on patients with severe uncontrolled blood pressure or moderate to severe, untreated hypokalemia. Blood pressure and potassium levels should be monitored during the testing, as hypertension and hypokalemia can be further precipitated or exacerbated with dietary sodium loading 12.
For the saline suppression test, 2 liters of isotonic saline are infused (500cc/h) over 4 hours. This test should not be performed in patients with compromised cardiac function due to the risk of pulmonary edema. Intravascular volume expansion should suppress the renin-angiotensin system (RAS). In normal subjects, plasma aldosterone concentration decreases below 5 ng/dL at the end of the saline infusion; levels greater than 10 ng/dL are considered diagnostic of autonomous aldosterone production. Values between 6 and 10 ng/dL are considered indeterminate 13.
Table 1. Hyperaldosteronism confirmation tests
| Confirmation Method | Protocol | Interpretation of Results |
|---|---|---|
| Oral Salt Suppression Test | ·Increase sodium intake for 3-4 days via supplemental tablets or dietary sodium to >200 mmol/day · Monitor blood pressure · Provide potassium supplementation to ensure normal serum levels · Measure 24h urinary aldosterone excretion and urinary sodium on 3rd or 4th day | · Primary hyperaldosteronism confirmed: if 24h urinary aldosterone excretion >12 mcg in setting of 24h sodium balance >200 mmol · Primary hyperaldosteronism unlikely: if 24h urinary aldosterone excretion <10mcg |
| Intravenous Saline Infusion Test | · Being infusion of 2L of normal saline after patient lies supine for 1 hour. · Infuse 2L of normal saline over 4 hours (500 mL/h) · Monitor blood pressure, heart rate, potassium · Measure plasma renin and serum aldosterone at time=0h and time=4h | · Primary hyperaldosteronism confirmed: 4h aldosterone level > 10 ng/dL · Primary hyperaldosteronism unlikely: 4h aldosterone level < 5 ng/dL |
| Captopril Challenge Test | · Administer 25-50mg of captopril in the seated position · Measure renin and aldosterone at time=0h and again at time=2h · Monitor blood pressure | · Primary hyperaldosteronism confirmed: serum aldosterone high and renin suppressed* · Primary hyperaldosteronism unlikely: renin elevated and aldosterone suppressed* *varying interpretations without specific validated cut-offs |
| Fludrocortisone Suppression Test | · Administer 0.1 mg fludrocortisone q6h for 4 days · Supplement 75-100 mmol of NaCl daily to ensure a urinary sodium excretion rate of 3 mmol/kg/body weight · Monitor blood pressure · Provide potassium supplementation to ensure normal serum levels · Measure plasma renin and serum aldosterone in the morning of day 4 while seated | · Primary hyperaldosteronism confirmed: Seated serum aldosterone > 6 ng/dL on day 4 with plasma renin activity < 1ng/mL/h · Primary hyperaldosteronism unlikely: suppressed aldosterone < 6 ng/dL |
Hyperaldosteronism treatment
Primary hyperaldosteronism treatment
Treatment for primary hyperaldosteronism depends on the underlying cause, but its basic goal is to normalize or block the effect of high aldosterone levels and prevent the potential complications of high blood pressure and low potassium levels.
Treatment for an adrenal gland tumor
Surgery is the treatment of choice in primary hyperaldosteronism cases where a unilateral adrenal gland tumor is the source of disease. A laparoscopic adrenalectomy is preferred over open adrenalectomy due to fewer complications and a shorter hospital stay. Complete adrenalectomy is preferred over partial adrenalectomy due to greater efficacy and resolution of symptoms. Removing the adrenal tumor may control the symptoms. Even after surgery, some people still have high blood pressure and need to take medicine. But often, the number of medicines or doses can be lowered. Salt restriction (less than 100 mEq/day), alcohol cessation, smoking cessation, maintenance of ideal body weight, and aerobic exercise are all beneficial in the postoperative and posttreatment care.
For nonsurgical candidates, mineralocorticoid receptor antagonists were preferred as a medical therapy.
Medicines to treat hyperaldosteronism include:
- Drugs that block the action of aldosterone (mineralocorticoid receptor antagonists)
- Mineralocorticoid receptor antagonists block the action of aldosterone in your body. Your doctor may first prescribe spironolactone. This medication helps correct high blood pressure and low potassium, but it may cause problems. In addition to blocking aldosterone receptors, spironolactone blocks androgen and progesterone receptors and may inhibit the action of these hormones. Side effects can include male breast enlargement (gynecomastia), decreased sexual desire, impotence, menstrual irregularities and gastrointestinal distress.
- A newer, more expensive mineralocorticoid receptor antagonist called eplerenone acts just on aldosterone receptors, but eliminates the sex hormone side effects associated with spironolactone. Your doctor may recommend eplerenone if you have serious side effects with spironolactone. You may also need other medications for high blood pressure.
- Diuretics (water pills), which help manage fluid buildup in the body
Lifestyle and home remedies
A healthy lifestyle is essential for keeping blood pressure low and maintaining long-term heart health. Here are some healthy lifestyle suggestions:
- Follow a healthy diet. Limit the sodium in your diet by focusing on fresh foods and reduced-sodium products, avoiding condiments, and removing salt from recipes. Diets that also emphasize a healthy variety of foods — including grains, fruits, vegetables and low-fat dairy products — can promote weight loss and help lower blood pressure. Try the Dietary Approaches to Stop Hypertension (DASH) diet — it has proven benefits for your heart.
- Achieve a healthy weight. If your body mass index (BMI) is 25 or more, losing as few as 10 pounds (4.5 kilograms) may reduce your blood pressure.
- Exercise. Regular aerobic exercise can help lower blood pressure. You don’t have to hit the gym — taking vigorous walks most days of the week can significantly improve your health. Try walking with a friend at lunch instead of dining out.
- Don’t smoke. Quitting smoking will improve your overall cardiovascular health. Nicotine in tobacco makes your heart work harder by constricting your blood vessels and increasing your heart rate and blood pressure. Talk to your doctor about medications that can help you stop smoking.
- Limit alcohol and caffeine. Both substances can raise your blood pressure, and alcohol can interfere with the effectiveness of some blood pressure medications. Ask your doctor whether moderate alcohol consumption is safe for you.
Treatment for overactivity of both adrenal glands
For bilateral adrenal hyperplasia (BAH) in primary hyperaldosteronism, mineralocorticoid receptor antagonists are the treatment of choice. Either spironolactone or eplerenone is used, depending on the adverse effect profile exhibited in the patient. A combination of medical therapy, sodium restriction (less than 100 mEq/day), alcohol avoidance, smoking cessation, aerobic exercise, and maintenance of ideal body weight were shown to produce the best results. According to the 2016 Endocrine Society Guidelines 11, the spironolactone starting dose is 12.5 to 25 mg daily and titrated upward every 2 weeks. Eplerenone is started at 50 mg daily and titrated upward. The clinical course ultimately dictates increase and frequency in dosage.
Secondary hyperaldosteronism treatment
For secondary hyperaldosteronism, the treatment of the underlying disease will lead to resolution of the symptoms. Salt restriction also is recommended for better efficiency. Secondary hyperaldosteronism is also treated with medicines (as described above). Surgery is usually not used.
Hyperaldosteronism prognosis
The outlook for primary hyperaldosteronism is good with early diagnosis and treatment.
The outlook for secondary hyperaldosteronism depends on the cause of the condition.
Few studies have been performed on the mortality rates of either form of hyperaldosteronism, but 10-year-survival rates have been reported between 90% to 95% in patients who are treated 1. The most common morbidity associated with this disorder is cardiovascular. Cardiovascular mortality is increased in these patients, but all-cause mortality is not significantly different.
- Dominguez A, Gupta S. Hyperaldosteronism. [Updated 2018 Oct 27]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2018 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK499983[↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Vaidya A, Dluhy R. Hyperaldosteronism. [Updated 2016 Oct 19]. In: De Groot LJ, Chrousos G, Dungan K, et al., editors. Endotext [Internet]. South Dartmouth (MA): MDText.com, Inc.; 2000-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK279065[↩][↩][↩][↩][↩]
- Stowasser M. Update in primary aldosteronism. J Clin Endocrinol Metab 2009;94:3623-30.[↩]
- Meyer A, Brabant G, Behrend M. Long-term follow-up after adrenalectomy for primary aldosteronism. World J Surg 2005;29:155-9.[↩]
- Gordon RD. Mineralocorticoid hypertension. Lancet 1994;344:240-3.[↩]
- Vaidya A, Hamrahian AH, Auchus RJ. Genetics of primary aldosteronism. Endocr Pract 2015;21:400-5.[↩]
- New M. Hypertension in congenital adrenal hyperplasia and apparent mineralocorticoid excess. Ann N Y Acad Sci 2002;970:145-54.[↩]
- Quinkler M, Stewart PM. Hypertension and the cortisol-cortisone shuttle. J Clin Endocrinol Metab 2003;88:2384-92.[↩][↩]
- Safian RD, Textor SC. Renal-artery stenosis. N Engl J Med 2001;344:431-42.[↩][↩][↩]
- Conn JW, Cohen EL, Lucas CP, et al. Primary reninism. Hypertension, hyperreninemia, and secondary aldosteronism due to renin-producing juxtaglomerular cell tumors. Arch Intern Med 1972;130:682-96.[↩]
- Funder JW, Carey RM, Mantero F, et al. The Management of Primary Aldosteronism: Case Detection, Diagnosis, and Treatment: An Endocrine Society Clinical Practice Guideline. J Clin Endocrinol Metab 2016;101:1889-916.[↩][↩][↩]
- Funder JW, Carey RM, Fardella C, et al. Case detection, diagnosis, and treatment of patients with primary aldosteronism: an endocrine society clinical practice guideline. J Clin Endocrinol Metab 2008;93:3266-81.[↩][↩]
- Rossi GP, Belfiore A, Bernini G, et al. Prospective evaluation of the saline infusion test for excluding primary aldosteronism due to aldosterone-producing adenoma. J Hypertens 2007;25:1433-42.[↩]