What is lemon oil
Lemon (Citrus limon L. Burm) is a flowing plant belonging to the Rutaceae family native to Asia. Lemon essential oil has shown antifungal 1 and antioxidative activities 2, as well as improved hippocampus function 3. Citrus essential oil has been identified in different parts of fruits as well as in leaves (particularly present in fruit), showing that limonene, b-myrcene, a-pinene, p-cymene, b-pinene, terpinolene, and other elements are the major aromatic compounds of many citrus species 4. Citrus lemon essential oil is used for many applications such as food, medicines, cosmetics and perfumes, detergents, aromatherapy, pathogen inhibition, and insect control 5.
The bioactive compounds in lemon essential oil are limonene, β-pinene, and γ-terpinene 6. In addition, minor components such as α-pinene, myrcene, and geraniol are present 7. Limonene, a major compound in lemon essential oil, showed lipid-lowering effects via upregulation of peroxisome proliferator-activated receptor alpha and liver X receptor beta in mice with high-fat diet-induced obesity 8. γ-Terpinene also had a lipid-lowering effect 9.
Lemon essential oil is able to accelerate the production of white blood cells, strengthen the immune system, and help in the digestion processes 10. The main constituents of lemon essential oil have demonstrated antiseptic, astringent, and detoxifying properties for blemishes associated with oily skin 11.
Figure 1. Lemon essential oil
Lemon oil production processes
Several processes are used to produce lemon oil. Expressed lemon oil are produced by pressing the outer rind of the ripe lemon fruits by sponge-press (i.e., by hand) or machines 12. Lemon oil may be produced more economically using an integrated juice-oil procedure such as the Brown Oil Extractor 13, where citrus fruit is partially submerged in water and abraded by metal discs. The oil is separated from the juice as a water-emulsion, and further separated using centrifugation to obtain the oil. The yield of expressed lemon oil is approximately 4% for lemons based on the weight of whole fruit 14. Lemon oil is also produced by distillation of expressed oil or direct distillation of fruit 13. Lemon oil is distilled (rectified) for removal of terpenes in order to improve solubility and permit use for flavoring carbonated beverages 14. Lemon oil can also be steam distilled to remove nonvolatile furocoumarins and are subsequently marketed as “psoralen free” (LOLI Global Industries). The type of lemon oil added to cosmetics are not regulated by law, nor limited by recommendations of appropriate trade organizations.
Lemon essential oil phototoxicity
Lemon oil are added as fragrances to a number of perfumed cosmetic products 15. Many of these products are intended for use on sun-exposed skin. Clinical and experimental studies have found that lime oil are phototoxic. A phototoxic reaction may occur when certain chemicals are applied to the skin and subsequently exposed to the sun. Phototoxic reactions result from direct damage to tissue caused by light activation of the photosensitising agent. This is called photocontact dermatitis. Photodermatitis is a self-limited problem that resolves spontaneously once the offending agent is removed or avoided. If avoiding the offending agent is not practical, then affected individuals should follow sun protection strategies, including wearing sun protective clothing and using sunscreen. The U.S. Food and Drug Administration (FDA) has reported the oil from the Persian lime (Citrus aurantifolia, var. Swingle) is presented as a cause of photodermatitis, and 11 cases are reported in which this reaction was observed 16. Because of their phototoxicity, the International Fragrance Association has recommended safe use levels for “leave on products” applied to sun-exposed skin. These safe use levels are intended to avoid short-term phototoxicity. However, long-term effects have not been addressed.oxypeucedanin and
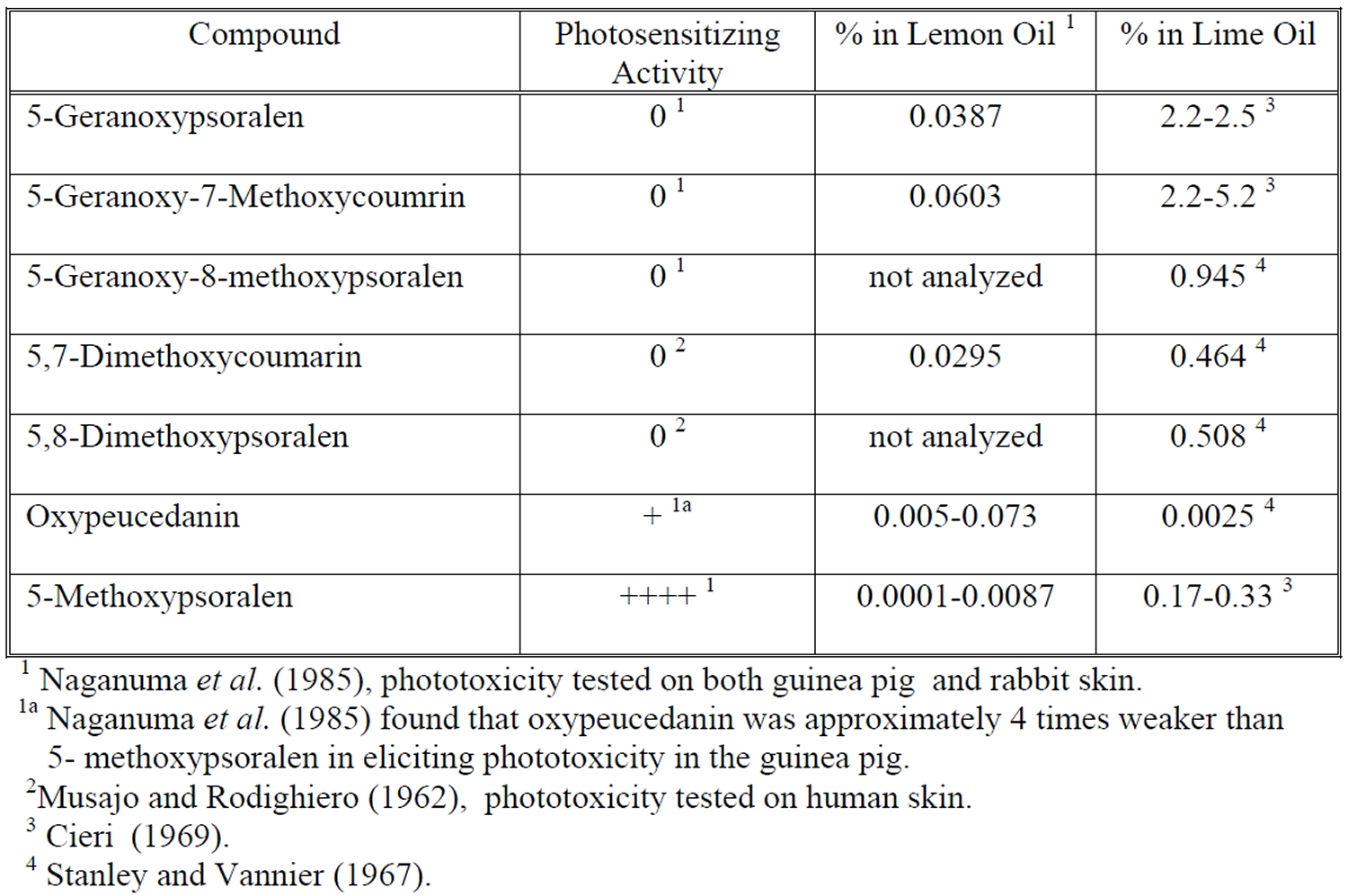
The phototoxicity elicited by lemon oil has been associated with naturally occurring furocoumarins. Two of these furocoumarins, 5-methoxypsoralen (bergapten) and oxypeucedanin, have been identified as the probable phototoxins in both lemon oil and lime oil 15. The levels of these furocoumarins have been found to vary significantly with growing conditions. The phototoxic potency of oxypeucedanin was only one-quarter of that of 5-methoxypsoralen (bergapten). However, the amounts of these two phototoxic compounds present in lemon oils produced in different regions of the world varied by a factor of more than 20 (5-methoxypsoralen, 4-87 ppm; oxypeucedanin, 26-728 ppm), and their ratio was not constant. The two compounds accounted for essentially all of the phototoxic activity of all lemon-oil samples. Among various other citrus-essential oils investigated, lime oil and bitter-orange oil also contained large amounts of oxypeucedanin. Oxypeucedanin was found to elicit photopigmentation on colored-guinea-pig skin without preceding visible erythema.
While the photogenotoxicity of 5-methoxypsoralen is well established, the photogenotoxicity of oxypeucedanin is unknown 15. Bacterial mutagenesis studies indicated that oxypeucedanin is not mutagenic, however photogenotoxicity studies of oxypeucedanin are not available. Additionally, the mutagenicity and photomutagenicity of mixtures of coumarins and furocoumarins in lemon oil and lime oil have not been assessed 15. The photogenotoxicity of oxypeucedanin alone or in mixtures of compounds representative of lemon oil and lime oil has not been assessed. These photogenotoxicity studies are needed. Additional studies on photocarcinogenicity should be conducted if coumarins or furocoumarins are significantly photogenotoxic as individual compounds or in complex mixtures (e.g. lemon or lime oil).
Lemon oil and lime oil were selected by the Center for Food Safety and Applied Nutrition for photogenotoxicity and photocarcinogenesis studies since these phototoxic oils contain furocoumarins. Furocoumarins such as 5-methoxypsoralen have been shown to be photocarcinogenic and are present in lemon and lime oils 15.
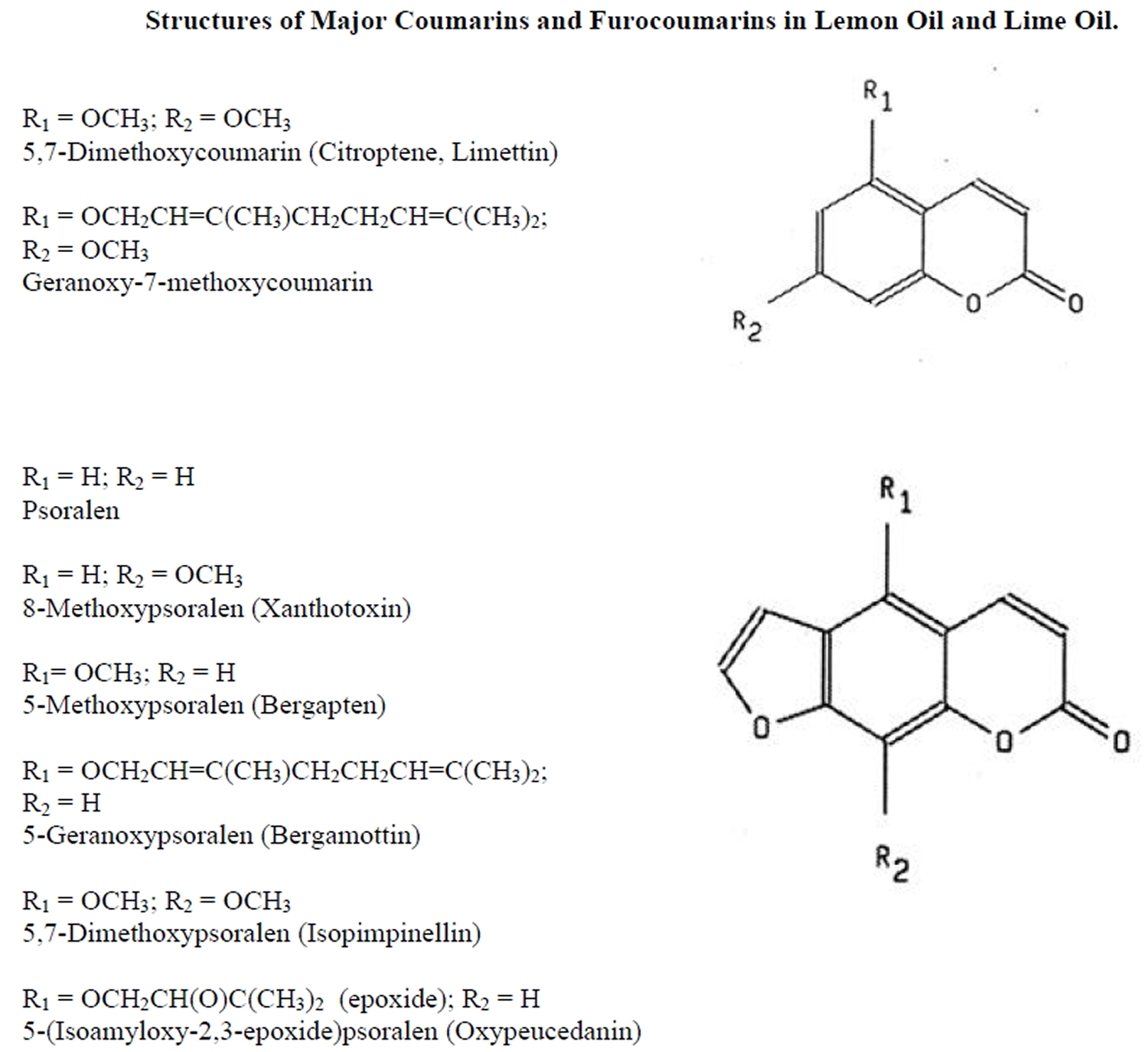
The phototoxicity of lemon oil and lime oil has been demonstrated in both experimental models and humans. Naganuma et al. 17, studied the phototoxicity of lemon oil obtained from different geographic locations. Lemon oil was diluted in ethanol (100% lemon oil, 50% lemon oil, and 20% lemon oil) and applied to backs of shaved, albino guinea pigs. Animals were then exposed to UVA radiation (320-400 nm, 13 J/cm²). Erythema was evaluated at 24, 48 and 72 hr after irradiation. Undiluted lemon oil and 50% lemon oil elicited phototoxicity for most of the samples tested. Lemon oil from Australia and Brazil elicited a weaker phototoxic than lemon oil from the Ivory Coast, Sicily, California, or Argentina. In an effort to identify the phototoxic components in lemon oil, these investigators fractionated samples by solvent extraction and subsequent phototoxicity testing of isolated components. Two phototoxic components were identified: oxypeucedanin and 5-methoxypsoralen (see Figure 2).
Figure 2. Major Coumarins and Furocoumarins in Lemon oil
Forbes et al. 18, have investigated the phototoxicity of a large number of fragrance raw materials including lemon oil and lime oil. Fragrance raw materials were tested using humans, pigs and albino, hairless mice. Several irradiation sources were used including sunlight, a solar simulator and a UVA radiation source (black light). Expressed lime oil was phototoxic in all three species under all three radiation sources. Distilled lime oil usually elicited no phototoxic response in any species. Two samples of distilled lime oil (derived from expressed lime oil), however, were found to be phototoxic. Lemon oil from Greece, Italy and from Florida (Persian limes) was reported to be phototoxic.
Coumarins and Furocoumarins Occurring in Lemon Oil and Lime Oil
Investigators have examined the level of coumarins and furocoumarins in lemon oil and lime oil (Table 1). A large body of evidence indicates that these secondary metabolites are photoactivated phytoalexins, produced to protect plants from bacterial, fungal, parasitic, and insectile infestations 19. Consistent with their protective function, levels of coumarins and furocoumarins vary widely. Naganuma et al. 17, found that levels of oxypeudedanin doubled in lemon oil derived from the same growing area in Australia between 1979 and 1980. The levels of oxypeudedanin in lemon oil from the Ivory Coast, Sicily and California diminished nearly three-fold in the same time period.
The photogenotoxicity and photocarcinogenicity of several linear furocoumarins (psoralens) have been well documented 20. 5-Methoxypsoralen (bergapten), a component of lemon and lime oil, is photogenototic in both prokaryotic and eukaryotic test systems. In addition, 5-methoxypsoralen (bergapten) has been found to be photocarcinogenic in an experimental model 21. Little is known about the photogenotoxicity and photocarcinogenicity of other furocoumarins found in lemon oil and lime oil. The mutagenicity of oxypeucedanin has been assessed in 6 tester strains of Salmonella typhimurium by Uwaifo 22. Oxypeucedanin was not mutagenic, however, photomutagenicity was not assessed.
Table 1. Levels of Major Coumarins and Furocoumarins in Lemon Oil and Lime Oil
Lemon essential oil uses
Lemon (Citrus limon L. Burm) is grown in many parts of the world for juice production and like the other citrus fruits, the peels are considered as waste. Lemon peels are in use in Chinese medicine and African folklore in the management of degenerative conditions, though there is dearth of information on
the scientific basis for the use of lemon peels. Much of the bioactivity of citrus peels have been linked to the phenolic content 24, which are more concentrated in the lemon peels that in the juices and seeds. However, there are a lot of other bioactive constituents, such as essential oils, which also contribute to their observed medicinal uses. Essential oils can be obtained in significant quantities from the peels of citrus fruits and they contain numerous compounds rich in polypohenols and terpene hydrocarbons 25. While, the juice have been shown to possess anticholiensterase and antioxidative properties, there is scarce of information on the medicinal potentials of the lemon peels, especially the essential oils, which are a major component of the peels and can be extracted in significant amount.
Recent trends in the management of neurodegenerative conditions, especially Alzheimer’s disease is to increase brain acetylcholine levels with the use of cholinesterase inhibitors. This is because in Alzheimer’s disease conditions there is elevated cholinesterase activities 26 which serve to break down the neurotransmitters acetylcholine and butyrylcholine thereby giving rise to the symptoms observed in Alzheimer’s disease. However, due to the side effects associated with the use of synthetic cholinesterase inhibitors, attention is been given to natural sources of cholinesterase inhibitors. Some sea weeds 27, citrus juices 28, vegetables and herbs 29 have been shown to be sources of cholinesterase inhibitors. In addition, the use of foods rich in antioxidants have been proposed to be beneficial in the prevention and management of neurodegenerative conditions because the high oxygen consumption of the brain cells and neurons especially, attack by reactive oxygen species 30. This test tube study 31 showed the inhibition of cholinesterases and Fe2+ and quinolinic acid-induced Malondialdehyde production as well as radicals (DPPH*, ABTS*, hydroxyl radicals and nitric oxide) scavenging abilities are possible mechanisms by which lemon peel essential oil could be used in the management and/or prevent neurodegenerative conditions. However, further in vivo experiments and clinical trials are recommended.
The bioactive compounds in lemon essential oil, as analyzed by gas chromatography, are shown in Table 2. The amount of limonene, β-pinene, and γ-terpinene in lemon essential oil was 66.57%, 10.00%, and 9.95%, respectively, indicating that limonene is the major bioactive compound. Besides these, sabinene, α-pinene, myrcene, and geraniol were present in a range of 1~2%. Seven other bioactive compounds were also detected in small quantities.
Plasma lipid levels, particularly cholesterol and LDL “bad” cholesterol levels are positively associated with atherosclerosis development due to hypercholesterolemia. In this animal study 6 , lemon essential oil decreased plasma total cholesterol levels in hypercholesterolemia-induced rabbits, which was significantly higher than the effects from limonene. In addition, the HDL “good” cholesterol level was significantly increased in the limonene group. Moreover, LDL “bad” cholesterol levels in the lemon essential oil and limonene groups were reduced although no significances were found. Limonene had cholesterol-lowering effects in hypercholesterolemia-induced animals 32 via downregulation of PPARα and LXRβ 32. Moreover, γ-terpinene in lemon essential oil, has exhibited cholesterol-lowering effects in animals 33. These effects were greater than those of limonene, a major bioactive compound in lemon essential oil, suggesting that the synergistic effects of bioactive compounds in lemon essential oil were stronger than the effects of limonene alone. Besides limonene, 13 other bioactive compounds (including sabinene, β-pinene, γ-terpinene, α-pinene, myrcene, and geraniol) were detected. In particular, the antioxidative effect of γ-terpinene has been reported to be 3-fold higher than that of limonene 6. Future research studies are needed to elucidate the mechanisms through which lemon essential oil prevents atherosclerosis as well as to determine its effect on inflammation and immune responses.
Table 2. Chemcial composition of lemon essential oil
| Compound | Relative area (%) |
|---|---|
| Limonene | 66.57 |
| β-Pinene | 10.00 |
| γ-Terpinene | 9.95 |
| Sabinene | 1.60 |
| α-Pinene | 1.95 |
| Myrcene | 1.59 |
| geraniol | 1.17 |
| Neral | 0.87 |
| β-Bisabolene | 0.59 |
| Neryl acetate | 0.53 |
| trans-α-Bergamotene | 0.43 |
| Geranyl acetate | 0.35 |
| Terpinolene | 0.29 |
| α-Terpineol | 0.12 |
Lemon, have been shown to be effective against Candida strains 34. However, the reports on the antifungal potential of lemon essential oil against Candida yeasts are ambiguous 35. Some literature data recommend it as highly effective, while other reports that the effects of its use are quite ordinary 36, 37. Devkatte et al. 37 obtained the Minimum Inhibitory Concentration (MIC) value of 0.5 % and the average zone of inhibition of approximately 17.5 mm for the lemon essential oil tested by them, while the zones of inhibition for the selected Candida strains determined by Warnke et al. 36 measured 16–43 mm. Higher mean values related to the zones of inhibition for Candida albicans, ranging from 23 to 45 mm, may be due to the differences in diffusion of particular essential oil ingredients in agar 7 and the concentration of biologically active substances, mainly citral, geraniol and verbenol. Thus, when evaluating the essential oils produced from a particular plant, it is good to know their chemical composition. According to many authors, the essential oil volatile phase composition depends on the conditions in which the plant has grown, its development stage and the way it is stored 7. In the case of citrus fruits, climatic conditions and the region of origin of the fruit are important 38. Differences in potential fungicidal properties of lemon essential oils may be due to variable qualitative and quantitative composition of individual essential oils and are related to the development stage of the fruit prior to extraction, fruit condition and quality as well as plant growing conditions 39.
According to Fisher and Phillips 7, the content of volatile substances in lemon essential oils may range from 85 to 99 %. The qualitative composition—proportions between the content of particular monoterpenes, sesquiterpenes and their oxygenated derivatives—changes as well 38. The highest concentration of volatile substances in lemon essential oils is observed in oils produced from medium-ripe fruit 40. Citrus essential oils are among the oils generally regarded as safe (GRAS) by the US Food and Drug Administration 41.
Lemon essential oils had different chemical compositions, but, except the Aromatic Art essential oil, they contained almost exclusively terpenes and oxygenated terpenes 39. The content of monoterpenes in the tested essential oils is shown in Table 3, monoterpenoids in Table 4 and sesquiterpenes, oxygenated sesquiterpenes and other compounds of the six tested essential oils in Table 5.
Table 3. Monoterpenes in lemon essential oils
| ETJA | Vera-Nord | Avicenna-Oil | Dufti by Gies | Croce Azzurra | Aromatic Art | |
| Area (%) | Area (%) | Area (%) | Area (%) | Area (%) | Area (%) | |
| Aliphatic monoterpenes | ||||||
| ß-myrcene | 4.11 | 1.46 | 0 | 6.94 | 5.42 | 0.37 |
| trans-ocimene | 0.13 | 0.49 | 0 | 0 | 0 | 0 |
| Monocyclic monoterpenes | ||||||
| ß-felandrene | 0 | 0 | 0 | 0.4 | 15.03 | 0 |
| limonene | 48.27 | 23.39 | 42.03 | 63.2 | 38.5 | 22.42 |
| α-terpinene | 0.4 | 0.12 | 0 | 0 | 0.14 | 0 |
| γ-terpinene | 4.85 | 5.87 | 0 | 4.78 | 0.16 | 0 |
| terpinolene | 1.19 | 0.3 | 0 | 0.34 | 0.04 | 0 |
| Bi- and tricyclic monoterpenes | ||||||
| bornylene | 0 | 0 | 0.49 | 0 | 0 | 0 |
| camphene | 0.35 | 0 | 0.17 | 0 | 0 | 0 |
| 3-carene | 0 | 0 | 0 | 0.16 | 0 | 0 |
| α-pinene | 11.06 | 1.44 | 3.42 | 3.61 | 5.56 | 0.16 |
| ß-pinene | 15.14 | 8.93 | 15.15 | 14.31 | 19.98 | 2.21 |
| 3-thujene | 0 | 0.3 | 0.22 | 0.33 | 1.25 | 0 |
| sabinene | 0 | 0.65 | 0 | 0 | 0 | 0.1 |
| tricyclene | 0.21 | 0 | 0 | 0 | 0 | 0 |
| Monoterpenes | 85.7 | 42.96 | 61.49 | 94.07 | 86.07 | 25.27 |
Table 4. Oxygenated monoterpenes in lemon essential oils
| ETJA | Vera-Nord | Avicenna-Oil | Dufti by Gies | Croce Azzurra | Aromatic Art | |
| Area (%) | Area (%) | Area (%) | Area (%) | Area (%) | Area (%) | |
| Oxygenated aliphatic monoterpenes | ||||||
| citral (mix of isomers) | 0 | 0.23 | 0.7 | 1.06 | 2.63 | 0 |
| trans-citral | 7.14 | 15.52 | 0 | 0 | 0.16 | 0.36 |
| cis-citral | 4.3 | 19.41 | 0 | 0.3 | 1.54 | 0 |
| citronellal | 0.2 | 0 | 0 | 0.7 | 0.29 | 0 |
| geranyl acetate | 0.91 | 0.59 | 0 | 0 | 0 | 0 |
| cis-geraniol | 0 | 4.96 | 0 | 0 | 0 | 0 |
| trans-geraniol | 0 | 3.39 | 1.58 | 0 | 0 | 0 |
| Linalool | 0.29 | 0 | 0.63 | 1.73 | 1.51 | 0 |
| linalyl propanoate | 0 | 0.25 | 0 | 0.24 | 0 | 0 |
| bergamol | 0 | 0 | 0 | 0 | 0 | 9.53 |
| neryl acetate | 0.49 | 0.42 | 0 | 0.16 | 1.14 | 0.21 |
| Oxygenated monocyclic monoterpenes | ||||||
| trans-carveol | 0 | 0 | 3.36 | 0 | 0 | 0 |
| carvone | 0 | 0 | 7.28 | 0.1 | 0 | 0 |
| trans-p-2,8-mentadien-1-ol | 0 | 0 | 1.84 | 0 | 0 | 0 |
| menthol | 0,12 | 0 | 0 | 0 | 0 | 0 |
| menthone | 0,06 | 0 | 0 | 0 | 0 | 0 |
| 1-terpinen-4-ol | 0 | 0 | 5.57 | 0 | 0.42 | 0 |
| Oxygenated bi- and tricyclic monoterpenes | ||||||
| pinene oxide | 0.17 | 0 | 0 | 0.65 | 0 | 0 |
| pinocarveol | 0 | 0 | 5.44 | 0 | 0 | 0 |
| verbenol | 0.08 | 0 | 4.9 | 0 | 0 | 0 |
| trans-verbenol | 0 | 0 | 1.5 | 0 | 0 | 0 |
| Oxygenated monoterpenes | 13.76 | 44.76 | 32.8 | 4.93 | 7.67 | 10.09 |
Table 5. Sesquiterpenes, oxygenated sesquiterpenes and others compounds in lemon essential oils
| ETJA | Vera-Nord | Avicenna-Oil | Dufti by Gies | Croce Azzurra | Aromatic Art | |
| Area (%) | Area (%) | Area (%) | Area (%) | Area (%) | Area (%) | |
| Sesquiterpenes | ||||||
| α-bergamotene | 0 | 0.95 | 2.31 | 0 | 0.89 | 0 |
| ß-bergamotene | 0 | 0 | 0 | 0 | 1.54 | 0 |
| ß-bisabolene | 0 | 0.48 | 0 | 0 | 1.82 | 0 |
| α-cadinene | 0 | 0 | 0 | 0.13 | 0 | 0 |
| ß-cadinene | 0 | 0 | 0 | 0.09 | 0 | 0 |
| calarene | 0 | 0 | 0 | 0.15 | 0 | 0 |
| caryophyllene | 0 | 0.24 | 0 | 0.29 | 1.11 | 0.12 |
| α-caryophyllene | 0 | 6.09 | 0 | 0 | 0.06 | 0 |
| ß-farnesene | 0 | 0 | 0 | 0 | 0.06 | 0 |
| germacene B | 0 | 0 | 0 | 0 | 0.38 | 0 |
| valencene | 0 | 0 | 0 | 0.32 | 0 | 0 |
| Sesquiterpenes | 0 | 7.76 | 2.31 | 0.98 | 5.84 | 0.12 |
| Oxygenated sesquiterpenes | ||||||
| caryophyllene oxide | 0 | 0 | 3.2 | 0 | 0 | 0 |
| farnesol | 0 | 4.48 | 0 | 0 | 0 | 6.12 |
| Oxygenated sesquiterpenes | 0 | 4.48 | 3.2 | 0 | 0 | 6.12 |
| Others | ||||||
| hexanoic acid | 0 | 0 | 0 | 0 | 0 | 0.47 |
| hydrocinnamic acid | 0 | 0 | 0 | 0 | 0 | 6.54 |
| ß-ionene | 0 | 0 | 0 | 0 | 0 | 7.81 |
| isopropyl myristate | 0 | 0 | 0 | 0 | 0 | 42.78 |
| 1,1-dimethoxy-2-phenylopropane | 0 | 0 | 0 | 0 | 0 | 0.36 |
| Others | 0 | 0 | 0 | 0 | 0 | 57.6 |
Despite the fact that all tested lemon essential oils contained limonene, their impact on the growth of the tested Candida yeasts was different. The Dufti essential oil, although contained the largest amounts of limonene, did not inhibit the growth of any of the tested yeast strains. The Croce Azzurra essential oil also proved to be ineffective as far as the inhibition of the growth of the tested strains is concerned. Therefore, the inhibitory effect of essential oils does not depend on the concentration of limonene but on the presence of other biologically active substances and the sensitivity of yeasts. Candida glabrata and Candida tropicalis strains are much less sensitive than the Candida albicans strain. Thus, only some essential oils at higher concentrations exhibit the antifungal potential against these yeast strains. Candida glabrata 33 and Candida glabrata 35 strains were the most resistant to the tested lemon essential oils (none of the tested products inhibited their growth). Lemon essential oils with high content of monoterpenoids may be an ingredient of products against candidiasis caused by Candida.
Limonene—a monocyclic terpene—was the main ingredient of the tested lemon essential oils. However, the antimicrobial activity depended on the content of oxygenated monoterpenes—the higher the content, the better fungicidal effects were observed. This is confirmed by the literature data 41. Vera-Nord and Avicenna-Oil essential oils contain 44.8 and 32.8 % of oxygenated monoterpenes, respectively, and exhibit antifungal potential against Candida albicans across the full range of the concentrations used and may be used as antifungal preparations against this yeast strain. The Aromatic Art essential oil, which is less effective as far as the inhibition of the growth of Candida albicans is concerned, contains hydrocinnamic acid and bergamol—compounds known for their antimicrobial properties 38.
Candida glabrata and Candida tropicalis strains were less sensitive to the tested essential oils than the Candida albicans strain—their growth was inhibited by essential oils used at higher concentrations. The Vera-Nord essential oil contains significant amounts of cis- and trans-citral isomers or geraniol and γ-terpinene known for their fungicidal properties 38.
The growth of Candida glabarata was inhibited by two lemon essential oils: Avicenna-Oil and ETJA, which both contains citral and verbenol. Essential oils exhibit antifungal properties when they contain, at the same time, large amounts of one of these oxygenated monoterpenes and small amounts of the second one, may be the reason for the growth inhibition of the tested Candida glabarata yeasts.
The tested essential oils contained also other valuable ingredients, such as carvone, 1-terpinen-4-ol and γ-terpinene—all of these compounds also exhibit antifungal potential, which has already been described in the literature 38.
The use of lemon essential oils in all kinds of candidiasis seems to be an interesting solution because of their documented safety. It should also be noted that some of the authors at the same time emphasize that it is necessary to conduct tests on the toxicity and possible allergenic effects of selected essential oils 42. Therefore, understanding the relationship between the chemical composition of essential oils and their antimicrobial activity is of great importance due to the potential use of lemon essential oil-based products as natural remedies against candidiasis caused by Candida albicans.
Lemon oil for skin
Lemon oil reported uses for skin conditions but are unproven and lacking studies in human for treating abscesses, acne, antiseptic, athlete’s foot, blisters, boils, cellulite, corns, cuts, grazes, greasy and oily conditions, insect bites, mouth ulcers, rosacea, sores,ulcers, viral infections (cold sores, herpes, verrucae,and warts), and wounds 43, 44, 45, 46, 47, 48, 49, 50.
Summary
Regardless of the frequency of the therapeutic claims made for lemon essential oil and the laboratory test tube activity, most evidence of the therapeutic efficacy of lemon essential oil has been published in books about aromatherapy and not in peer-reviewed journals. A few clinical trials have emerged, but their results are rarely confirmed completely to substantiate lemon essential oil effectiveness. More rigorous clinical trials would establish confidence from the medical professionals for the benefits of patients and consumers 51.
- Guerra FQS, Mendes JM, de Oliveira WA, de Souza FS, Trajanov VN, Coutinho HDM, de Oliveira Lima E. Antibacterial activity of the essential oil of Citrus limon against multidrug resistant Acinetobacter strains. Rev Bras Farm. 2013;94:142–147[↩]
- Bertuzzi G, Tirillini B, Angelini P, Venanzoni R. Antioxidative action of Citrus limonum essential oil on skin. Eur J Med Plants. 2013;3:1–9. doi: 10.9734/EJMP/2013/1987[↩]
- Lopes Campêlo LM, Moura Gonçalves FC, Feitosa CM, de Freitas RM. Antioxidant activity of Citrus limon essential oil in mouse hippocampus. Pharm Biol. 2011;49:709–715. doi: 10.3109/13880209.2010.541924 https://www.ncbi.nlm.nih.gov/pubmed/21639684[↩]
- M.F. He´ rent, D.V. Bie, B. Tilquin, Determination of new retention indices for quick identification of essential oils compounds, J. Pharm. Biomed. Anal. 43 (2007) 886–892.[↩]
- D.L. Palazzolo, Electronic cigarettes and vaping: a new challenge in clinical medicine and public health. A literature review, Front Public Health 1 (2013) 56, http://dx.doi.org/10.3389/fpubh.2013.00056[↩]
- Lee H, Woo M, Kim M, Noh JS, Song YO. Antioxidative and Cholesterol-Lowering Effects of Lemon Essential Oil in Hypercholesterolemia-Induced Rabbits. Preventive Nutrition and Food Science. 2018;23(1):8-14. doi:10.3746/pnf.2018.23.1.8. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5894780/[↩][↩][↩][↩]
- Fisher K, Phillips C. Potential antimicrobial uses of essential oils in food: is citrus the answer? Trends Food Sci Technol. 2008;19:156–164. doi: 10.1016/j.tifs.2007.11.006[↩][↩][↩][↩]
- Jing L, Zhang Y, Fan S, Gu M, Guan Y, Lu X, Huang C, Zhou Z. Preventive and ameliorating effects of citrus D-limonene on dyslipidemia and hyperglycemia in mice with high-fat diet-induced obesity. Eur J Pharmacol. 2013;715:46–55. doi: 10.1016/j.ejphar.2013.06.022 https://www.ncbi.nlm.nih.gov/pubmed/23838456[↩]
- Takahashi Y, Inaba N, Kuwahara S, Kuki W. Effects of γ-terpinene on lipid concentrations in serum using Triton WR1339-treated rats. Biosci Biotechnol Biochem. 2003;67:2448–2450. doi: 10.1271/bbb.67.2448 https://www.ncbi.nlm.nih.gov/pubmed/14646206[↩]
- Tisser R., Balacs T. Essential oil safety: a guide for health professionals. Edinburgh: Churchill Livingstone; 1995[↩]
- Tisser R., Young R. Essential oil safety: a guide for health care professional. 2nd. London, UK: Churchill Livingstone; 2013[↩]
- Poucher, W. A. (1991) Poucher’s Perfumes, Cosmetics and Soaps-Vol. 1. The Raw Materials of Perfumery, 9th Edition. Ed.: A. J. Jouhar. Chapman & Hall, NY. pp. 199-201.[↩]
- Swaine, R. L. and R. L. Swaine Jr. (1988) Citrus Oils: Processing, Technology, and Applications. Perfume & Flavorist 13: 2-20.[↩][↩]
- Burdock, G. A. (Ed.) (1995) Fenaroli’s Handbook of Flavor Ingredients, Volume 1, 3rd Edition. CRC Press, Boca Raton, FL[↩][↩]
- Lemon Oil. https://ntp.niehs.nih.gov/ntp/htdocs/chem_background/exsumpdf/lemonlimeoils_508.pdf[↩][↩][↩][↩][↩]
- FDA Poisonous Plant Database. https://www.accessdata.fda.gov/scripts/plantox/detail.cfm?id=23780[↩]
- Naganuma, M., S. Hirose, Y. Nakayama, K. Nakajima and T. Someya (1985) A Study of the Phototoxicity of Lemon Oil. Arch. Dermatol. Res. 278: 31-36.[↩][↩]
- Forbes, P. D., F. Urbach and R. E. Davies (1977) Phototoxicity Testing of Fragrance Raw Materials. Fd. Cosmet. Toxicol. 15: 55-60.[↩]
- Downum, K. R. (1992) Light-Activated Plant Defense. New Phytolog. 122: 401-420.[↩]
- Saffran, W. A. (1988) Genotoxic Effects of Psoralen. In: Psoralen DNA Photobiology. Volume II. Ed.: F. P. Gasparro, CRC Press, Boca Raton, FL. pp 73-86.[↩]
- Zajdela, F. and E. Bisagni (1981) 5-Methoxypsoralen, the Melanogenic Additive in Suntan Preparations, Is Tumorigenic in Mice Exposed to 365 nm UV Radiation. Carcinogenesis 2: 121-127.[↩]
- Uwaifo, A. O. (1984) The Mutagenicities of Seven Coumarin Derivatives and a Furan Derivative (Nimbolide) Isolated from Three Medicinal Plants. J. Toxicol. Environ. Health 13: 521-530.[↩]
- https://ntp.niehs.nih.gov/ntp/htdocs/chem_background/exsumpdf/lemonlimeoils_508.pdf[↩]
- Oboh, G.; Ademosun A. O. Characterization of the antioxidant properties of phenolic extracts from some citrus peels. J. Food Sci. Technol. 49, 729-736;2012[↩]
- Odeh, F.; Rahmo, A.; Alnori, A. S.; Chaty, M. E. The cytotoxic effect of essential oils citrus aurantium peels on human colorectal carcinoma cell line (LIM1863). J. Microbiol. Biotechnol. Food Sci. 1, 1476-1487;2012[↩]
- Rao, A.; Agarwal, S. Role of antioxidant lycopene in cancer and heart disease. J. Ame. Coll. Nutr. 19, 563-569;2000[↩]
- Yoon, N. Y.; Chung, H. Y.; Kim, H. R.; Choi, J. S. Acetyland butyrylcholinesterase inhibitory activities of sterols and phlorotannins from Ecklonia stolonifera. Fish Sci. 74, 200-207;2008[↩]
- Ademosun, A. O.; Oboh G. Inhibition of acetylcholinesterase activity and Fe2+ induced lipid peroxidation in rat brain in vitro by some citrus fruit juices. J. Med. Food. 15, 428-434;2012[↩]
- Mukherjee, P. K.; Kumar, V.; Mal M.; Houghton, P. J. Acetylcholinesterase inhibitors from plants. Phytomed. 14, 289-300;2007[↩]
- Gandhi, S.; Abramov, A. Y. Mechanism of Oxidative Stress in Neurodegeneration. Oxidative Medicine and Cellular Longevity. doi:10.1155/2012/428010;2012[↩]
- Essential Oil from Lemon Peels Inhibit Key Enzymes Linked to Neurodegenerative Conditions and Pro-oxidant Induced Lipid Peroxidation. J. Oleo Sci. 63, (4) 373-381 (2014). https://www.jstage.jst.go.jp/article/jos/63/4/63_ess13166/_pdf/-char/en[↩]
- Jing L, Zhang Y, Fan S, Gu M, Guan Y, Lu X, Huang C, Zhou Z. Preventive and ameliorating effects of citrus D-limonene on dyslipidemia and hyperglycemia in mice with high-fat diet-induced obesity. Eur J Pharmacol. 2013;715:46–55. doi: 10.1016/j.ejphar.2013.06.022[↩][↩]
- Takahashi Y, Inaba N, Kuwahara S, Kuki W. Effects of γ-terpinene on lipid concentrations in serum using Triton WR1339-treated rats. Biosci Biotechnol Biochem. 2003;67:2448–2450. doi: 10.1271/bbb.67.2448[↩]
- Hsu CC, Lai WL, Chuang KC, Lee MH, Tsai YC. The inhibitory activity of linalool against the filamentous growth and biofilm formation in Candida albicans. Med Mycol. 2013;51:473–482. doi: 10.3109/13693786.2012.743051. https://www.ncbi.nlm.nih.gov/pubmed/23210679[↩]
- Carvalhinho S, Costa AM, Coelho AC, Martins E, Sampaio A. Susceptibilities of Candida albicans mouth isolates to antifungal agents. Essential oils and mouth rinses. Mycophatologia. 2012;174:69–76. doi: 10.1007/s11046-012-9520-4[↩]
- Warnke PH, Becker ST, Podschun R, et al. The battle against multi-resistant strains: renaissance of antimicrobial essential oils as a promising force to fight hospital-acquired infections. J Cranio-Maxillofac Surg. 2009;37:392–397. doi: 10.1016/j.jcms.2009.03.017 https://www.ncbi.nlm.nih.gov/pubmed/19473851[↩][↩]
- Devkatte AN, Zore GB, Karuppayil SM. Potential of plant oils as inhibitors of Candida albicans growth. FEMS Yeast Res. 2005;5:867–873. doi: 10.1016/j.femsyr.2005.02.003 https://www.ncbi.nlm.nih.gov/pubmed/15925315[↩][↩]
- Settanni L, Palazzolo E, Guarrasi V, et al. Inhibition of foodborne pathogen bacteria by essential oils extracted from citrus fruits cultivated in Sicily. Food Control. 2012;26:326–330. doi: 10.1016/j.foodcont.2012.01.050[↩][↩][↩][↩][↩]
- Białoń M, Krzyśko-Łupicka T, Koszałkowska M, Wieczorek PP. The Influence of Chemical Composition of Commercial Lemon Essential Oils on the Growth of Candida Strains. Mycopathologia. 2014;177(1-2):29-39. doi:10.1007/s11046-013-9723-3. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3915084/[↩][↩][↩][↩][↩]
- Tirado CB, Stashenko EE, Combariza MY, Martinez JR. Comparative study of Colombian citrus oils by high-resolution gas chromatography and gas chromatography-mass spectrometry. J Chromatogr A. 1995;697:501–513. doi: 10.1016/0021-9673(94)00955-9[↩]
- Espina L, Somolinos M, Lorán S, et al. Chemical composition of commercial citrus fruit essential oils and evaluation of their antimicrobial activity acting alone or in combined processes. Food Control. 2011;22:896–902. doi: 10.1016/j.foodcont.2010.11.021[↩][↩]
- Devkatte AN, Zore GB, Karuppayil SM. Potential of plant oils as inhibitors of Candida albicans growth. FEMS Yeast Res. 2005;5:867–873. doi: 10.1016/j.femsyr.2005.02.003[↩]
- Evans M. Natural Healing: Remedies & Therapies. London, UK: Hermes House; 2010.[↩]
- Lawless J. The Illustrated Encyclopedia of Essential Oils: The Complete Guide to the Use of Oils in Aromatherapy and Herbalism. Rockport, Mass, USA: Element Books; 1995[↩]
- Sellar W. The Directory of Essential Oils. London, UK: C. W. Daniel Company; 1992.[↩]
- Curtis S. Essential Oils. London, UK: Aurum Press; 1996[↩]
- Ark Creative. Just Aromatherapy. Valencia, Calif, USA: Top That! Publishing; 2005[↩]
- Kovac M. A Quick Guide to Essential Oils. Ljubljana, Slovenia: Aromadelavnice s.p.; 2011[↩]
- Meadowbank. Ailments leaflet-find an essential oil for your ailment, 2012[↩]
- Burgess. Burgess and Finch Aromatherapy: Patient Leaflet. 2013[↩]
- Raines D. Wound care. In: Price S., Price L., editors. Aromatherapy for Health Professionals. 4th. chapter 10. London, UK: Churchill Livingstone; 2012.[↩]