Lymphocytic esophagitis
Lymphocytic esophagitis is a rare disorder of the esophagus in which there are an increased number of lymphocytes in the lining of the esophagus, with most cases presenting with normal-appearing esophageal mucosa 1. Lymphocytic esophagitis is a form of chronic esophagitis characterized by edema and lymphocytic infiltration of the epithelial peripapillary fields, without eosinophils 2.
Lymphocytic esophagitis is defined by three histologic characteristics:
- Dense peripapillary lymphocytic infiltrates in the esophageal squamous mucosa (termed Peripapillary Lymphocytosis),
- Little to no intraepithelial granulocytes within these areas of focus and
- The presence of spongiosis, or intercellular edema, of the squamous epithelial cells specifically in the areas of focus.
Lymphocytic esophagitis significance is unclear, as it was previously thought to have little clinical impact. Lymphocytic esophagitis may be related to eosinophilic esophagitis or to gastroesophageal reflux disease (GERD).
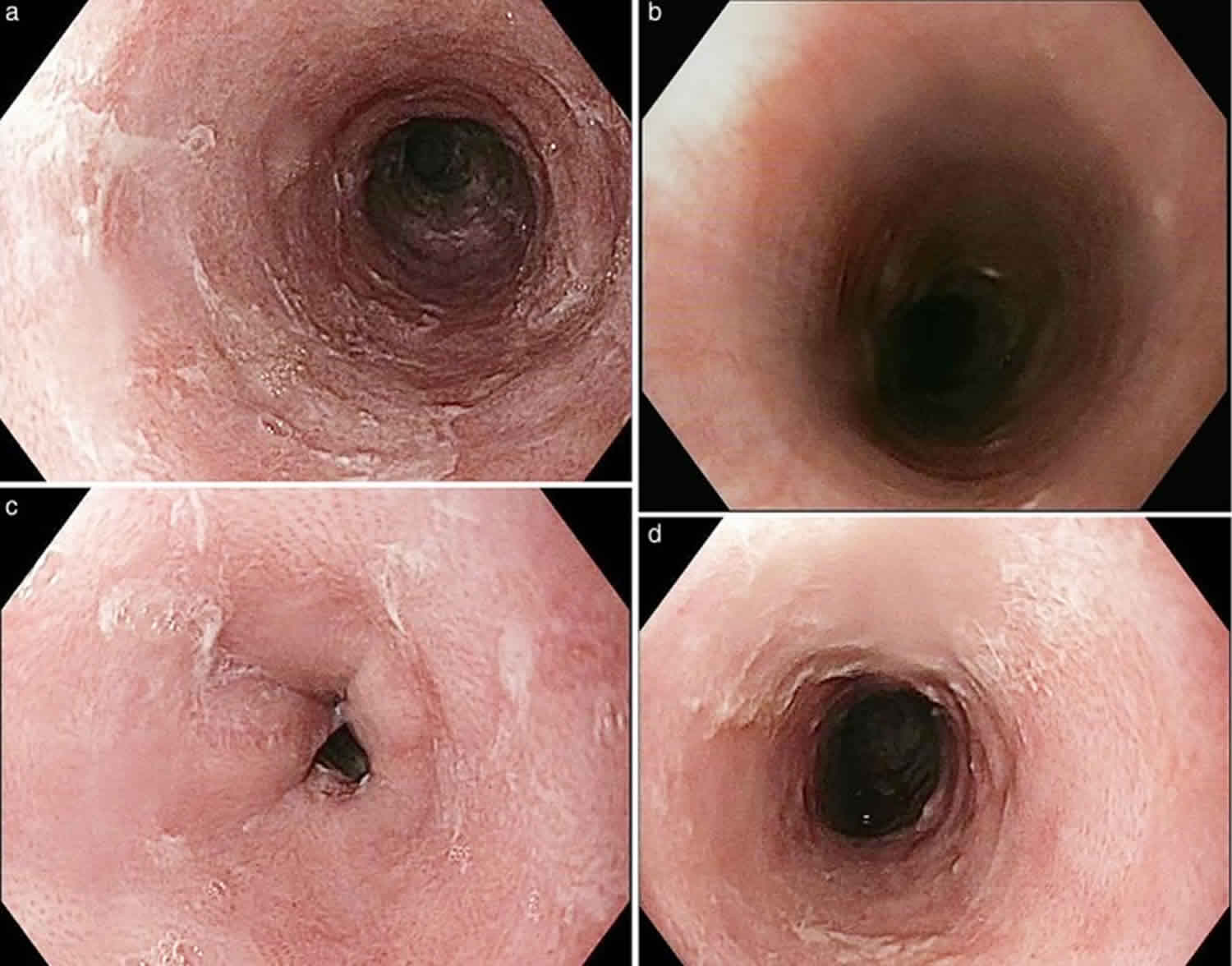
In 2006, however, Rubio and colleagues 3 described 20 unique cases of chronic esophagitis postulating a new entity, lymphocytic esophagitis. Subsequent studies of lymphocytic esophagitis have noted endoscopic features similar to eosinophilic esophagitis, including esophageal rings, esophagitis, esophageal stricture, erythema, nodularity, plaques, furrows and webs 4.
Since its description, there have been numerous publications supporting lymphocytic esophagitis as a distinct clinical entity 5. Few have commented on the presence of esophageal rings.
Overall, lymphocytic esophagitis was detected in ~ 0.1 %, or 1 in 1000, of all patients who underwent esophageal biopsies for any cause 6. Lymphocytic esophagitis predominantly affects older women (60 %) with a median age of 63 2. In contrast, eosinophilic esophagitis tends to affect younger men (66 %) with a median age of 43 6. The majority of lymphocytic esophagitis patients are white (86 %) 7. When compared to patients with normal esophageal biopsies, GERD (gastroesophageal reflux disease) and eosinophilic esophagitis, patients with lymphocytic esophagitis were more likely to use tobacco, had comparable rates of drug and food allergies, and were less likely to have allergic rhinitis 4.
Lymphocytic esophagitis patients typically present with symptoms of dysphagia, chest/ abdominal pain and heartburn 8. Common endoscopic features of lymphocytic esophagitis include esophagitis, strictures and stenosis, but there is a large portion of patients with a normal appearing esophagus. One technique that shows promise in increasing detection and decreasing sampling error is narrow-band imaging magnifying endoscopy (NBI-ME). Currently, there is no research guiding treatment for lymphocytic esophagitis, but proton pump inhibitors and swallowed corticosteroids have been used in the past 8.
Lymphocytic esophagitis cause
Unfortunately, there is not much known regarding the cause of lymphocytic esophagitis. One group of researchers hypothesized that since lymphocytic esophagitis is histologically similar to acute spongiotic dermatitis such as allergic contact dermatitis, irritant contact dermatitis and atopic dermatitis, the cause of lymphocytic esophagitis may have an allergic or irritant component 9. Unfortunately, their study found no statistically significant non-drug allergy or drug allergy association in patients diagnosed with lymphocytic esophagitis 9. Although the origin of lymphocytic esophagitis is currently unknown, further research into this condition will provide a greater insight as to how and why it develops. To date, there have been no specific testing pH monitoring evaluations for gastroesophageal reflux disease (GERD) in this population.
Associated conditions
Of the few studies done focusing on lymphocytic esophagitis, most have tried to find associated conditions but have failed. So far, there has been no associations between lymphocytic esophagitis and allergies (drug and non-drug), H. pylori gastritis or celiac disease 9; the association between lymphocytic esophagitis and Crohn’s disease is still controversial. One of the earlier studies done found that about 40% of patients with lymphocytic esophagitis also had Crohn’s disease 10. Subsequent studies have failed to show a statistically significant association, but interestingly, these studies were all done in adults 11. When looking back at the original study that demonstrated an association, about half of the patients with lymphocytic esophagitis were less than seventeen years old. Two newer studies done focusing on only pediatric patients, both found statistically significant associations between lymphocytic esophagitis and Crohn’s disease 12, 13. One even went as far to say that when lymphocytic esophagitis is found in a pediatric patient with Crohn’s disease, it is more likely to be an upper gastrointestinal manifestation of Crohn’s disease rather than another, separate entity 12.
Lymphocytic esophagitis symptoms
Clinical findings for lymphocytic esophagitis are non-specific. In a study of 119 cases of lymphocytic esophagitis, patients presented with similar symptoms as patients with eosinophilic esophagitis, most commonly dysphagia (lymphocytic esophagitis: 53 % vs. eosinophilic esophagitis: 63 %) 6. Across 4 studies of patients with lymphocytic esophagitis, complaints of dysphagia and heartburn were noted in 43 – 70 % and 18.5 – 47 % of patients respectively 4. Additional symptoms include odynophagia, abdominal pain, chest pain, nausea, vomiting, regurgitation, and heartburn (see Table 1). Notable complications of lymphocytic esophagitis include food impactions 14.
Although not a complete list, the most frequent symptoms of patients who are diagnosed with lymphocytic esophagitis are: dysphagia, chest/abdominal pain and heartburn/reflux 8. The rates of patients presenting with dysphagia and subsequently diagnosed with lymphocytic esophagitis are comparable to patients with dysphagia diagnosed with eosinophilic esophagitis. One difference noted was that lymphocytic esophagitis patients typically didn’t present with food impaction 15. There have been few reports of more serious symptoms in patients with lymphocytic esophagitis. One case report of a 35-year-old female who presented with chest pain, fever and hemodynamic shock was found to have diffuse thickening of the esophagus with bilateral pleural effusions, thought to be due to microperforation of the esophagus. Of note, she endorsed mild odynophagia following her most recent meal along with an acute non-productive cough. Upper endoscopy performed one week after the acute event was unremarkable, but biopsies from the mid and distal esophagus showed large numbers of intramucosal peripapillary lymphocytes along with spongiosis, with a lack of eosinophils, viral inclusions or dysplastic/neoplastic lesions. Workup for autoimmune and chronic inflammatory disease was negative. This case study is one of the first to describe a potentially lethal diagnosis of spontaneous esophageal perforation in which lymphocytic esophagitis might have played a role 16.
Lymphocytic esophagitis diagnosis
There are no serum biomarkers currently known to correlate with lymphocytic esophagitis. A smaller study had found that patients with peripapillary lymphocytosis, one of criteria of lymphocytic esophagitis, were associated with higher inflammatory markers such as erythrocyte sedimentation rate and C-reactive protein 11. More research in this area is needed to determine if these tests would be useful clinically in this setting.
Histopathologic characterization is what defines lymphocytic esophagitis. Histology of lymphocytic esophagitis is characterized by a chronic inflammation of the mid and distal esophagus with epithelial lymphocytic infiltration, particularly involving the peripapillary fields, distinct from other forms of chronic esophagitis where lymphocytes predominate in the interpapillary fields 3. The relative absence of granulocytes is a notable feature of lymphocytic esophagitis.
The overall pattern of inflammation, distribution of lymphocytes within the squamous epithelium, and clinical presentation are important in making a diagnosis 2. Lymphocytic esophagitis is a pathologic diagnosis characterized as having the following:
- Large number of lymphocytes in the peripapillary region of the esophageal squamous epithelium;
- Frequent absence of granulocytes (i. e. eosinophils, neutrophils) 17; and
- Marked intracellular edema/spongiosis 6.
In contrast, biopsies in patients with gastroesophageal reflux disease (GERD) demonstrate minimal, if any, reactive histologic changes. There has been no association identified between lymphocytic esophagitis and lymphocytic infiltration at other sites in the gastrointestinal tract 18.
The lymphocytes identified on biopsy of patients with lymphocytic esophagitis expressed CD3, CD4, CD8 and rarely granzyme B, without T-cell intracytoplasmic antigen 19. No granulocytes were present in 60 % of biopsies meeting diagnostic criteria for lymphocytic esophagitis, and only occasional granulocytes were seen in the remaining 40 %. In contrast, many granulocytes were present in the other forms of esophagitis, including grade 2 and 3 gastroesophageal reflux and Candida albicans esophagitis 17.
Endoscopic features
Patients with lymphocytic esophagitis can have a variety of endoscopic findings (Table 1). Studies have shown that esophagitis was present only 18-24% of the time 9, with strictures appearing 10.1% 20 and narrow-caliber/stenosis seen in 36% of patients with lymphocytic esophagitis 21. Interestingly, about 18 % to 31 % of patients with a normal looking esophagus were ultimately diagnosed with lymphocytic esophagitis 4; this highlights the importance of biopsy in making this diagnosis, and not only relying on gross appearance. Esophageal rings were present in 19.8 % of patients (vs. 18.8 % of patients with eosinophilic esophagitis) 5 . In contrast to eosinophilic esophagitis, patients with lymphocytic esophagitis had fewer esophageal strictures (8.6 % vs. 16.3 %) 7, however the incidence of strictures in lymphocytic esophagitis ranged from 8.6 % to 37 % 4. Overall, based on the endoscopic appearance of the esophagus, eosinophilic esophagitis was suspected by physicians in one third of lymphocytic esophagitis cases 6, demonstrating that these clinically distinct entities have overlapping features.
Trying to differentiate between eosinophilic esophagitis and lymphocytic esophagitis based on a gross view of the esophagus can be challenging. Researchers have reported that about 70% of patients diagnosed with lymphocytic esophagitis had at least 1 feature suggestive of eosinophilic esophagitis on esophagogastroduodenoscopy (felinization with furrows, white plaques and strictures) 21. As mentioned above, gross endoscopic features may not always provide a clear picture as to what is truly occurring within the esophagus. One study recommends using narrow-band imaging magnifying endoscopy (NBI-ME) in order to improve detection rates of lymphocytic esophagitis and eosinophilic esophagitis 21. Narrow-band imaging magnifying endoscopy is commonly used to assess invasiveness of superficial esophageal cancers by visualization of intrapapillary capillary loops. These researchers demonstrated that patients with lymphocytic esophagitis and eosinophilic esophagitis were found to have at least 1 of the following characteristics on narrow-band imaging magnifying endoscopy:
- Beige color (normal color of the esophagus is light green)
- Increased and congested intrapapillary capillary loops (IPCLs), viewed as a dot-like appearance – seen with both white light and narrow band imaging.
- Lack of visible submucosal vessels.
Additionally, these lymphocytic esophagitis patients exhibit the same “crepe paper fragility” of the mucosa, similar to what has been described with eosinophilic esophagitis. Patients with gastroesophageal reflux disease (GERD) did not exhibit any of these features 20. Therefore, this technique could be helpful in differentiating between lymphocytic esophagitis or eosinophilic esophagitis from GERD, and assist with biopsy locations within the esophagus in order to reduce sampling error.
Table 1. Symptoms and endoscopic findings in patients with lymphocytic esophagitis across 4 studies
| Purdy et al. 22 (%) | Haque et al. 6 (%) | Cohen et al. 7 (%) | Pasricha et al. 4 (%) | |
| Clinical manifestations | ||||
| Dysphagia | 43 | 52.9 | 66.7 | 70 |
| Heartburn/reflux | 26 | 18.5 | 46.9 | 26 |
| Abdominal/chest pain | – | – | 44.4 | – |
| Abdominal pain | – | – | – | 15 |
| Chest pain | – | – | – | 19 |
| Nausea/vomiting | – | – | 29.6 | 19 |
| Odynophagia | – | – | 12.3 | 4 |
| All esophageal symptoms | 57 | – | – | – |
| Endoscopic impressions | ||||
| Normal | 31 | 22.6 | 29.6 | 18 |
| Esophagitis | 24 | 18.5 | 16.0 | 33 |
| Eosinophilic esophagitis | – | 33.6 | – | – |
| Esophageal rings | 12 | – | 19.8 | 26 |
| Stricture | – | 10.1 | 8.6 | 37 |
| Motility disorder | – | 5.0 | – | – |
| Schatzki’s ring | – | 2.5 | – | – |
| Candida | – | 1.6 | – | 15 |
| Achalasia | – | 0.8 | – | – |
| Hiatal hernia | 10 | – | – | 26 |
| Narrow caliber | – | – | – | 44 |
Lymphocytic esophagitis pathology
A diagnosis of lymphocytic esophagitis must meet 3 histological characteristics 20:
- Dense peripapillary lymphocytic infiltrates in the esophageal squamous mucosa (termed Peripapillary Lymphocytosis),
- Little to no intraepithelial granulocytes (neutrophils, eosinophils) within these areas of focus and
- The presence of spongiosis, or intercellular edema, of the squamous epithelial cells specifically in the areas of focus.
The exact number of lymphocytes present to satisfy the first requirement has varied across different studies, with minimum cutoffs set as low as 30 per high power field 2 up to 50 per high power field 12. The study that originally used 30 intraepithelial lymphocytes per high power field concluded that having a minimum cutoff could be misleading and would not increase the specificity of making the diagnosis; the researchers felt that that the focus should rather lie in the distribution of the lymphocytes rather than the sheer number 20.
Lymphocytic esophagitis treatment
Treatment options for lymphocytic esophagitis have not been well studied or defined. Given one of the main symptoms of lymphocytic esophagitis being heartburn, proton pump inhibitors (PPI) have been prescribed to patients. One study found that about half of patients diagnosed with lymphocytic esophagitis felt improvement of their initial symptoms after starting a PPI, although it was unsure if there was concomitant GERD that was being treated. It is not clear if there will be a PPI lymphocyte responsive subset of patients as was seen in the PPI responsive eosinophilic esophagitis patients 23. Similarly there has not been a dietary exclusion trial assessment for clinical success as has been shown with eosinophilic esophagitis 24. Others have tried esophageal dilation and topical swallowed steroids (e.g., swallowed fluticasone), which are similar treatments for other forms of esophagitis 25.
Lymphocytic esophagitis prognosis
Although extremely limited data is available, the course of lymphocytic esophagitis appears to be favorable. Almost 90% of patients with lymphocytic esophagitis were still living three years after being diagnosed 15. Most patients report a good quality of life and satisfaction with their health at follow-up visits within three years post diagnosis 15. Only about 33% of patients with lymphocytic esophagitis had visited the emergency room with a gastrointestinal complaint 15. Repeat upper endoscopy on patients with previously diagnosed lymphocytic esophagitis demonstrated persistent lymphocytic esophagitis in 41 % of cases, with the remaining cases showing normal or alternative diagnoses 15. Nearly 60 % of this cohort of lymphocytic esophagitis patients reported improvement in initial symptoms, largely attributed to escalation of medical management (70.6 % of cases), usually with a proton pump inhibitor 15.
- Rouphael C, Gordon IO, Thota PN: Lymphocytic esophagitis: still an enigma a decade later. World J Gastroenterol. 2017, 14:949–956. 10.3748/wjg.v23.i6.949[↩]
- Pleet JL, Taboada S, Rishi A, Milman PJ, Trindade AJ. Rings in the esophagus are not always eosinophilic esophagitis: Case series of ring forming lymphocytic esophagitis and review of the literature. Endosc Int Open. 2017;5(6):E484-E488. doi:10.1055/s-0043-106579 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5451283[↩][↩][↩]
- Rubio CA, Sjödahl K, Lagergren J. Lymphocytic esophagitis: a histologic subset of chronic esophagitis. Am J Clin Pathol. 2006;125(3):432-437.[↩][↩]
- Pasricha S, Gupta A, Reed C C et al.Lymphocytic Esophagitis: An Emerging Clinicopathologic Disease Associated with Dysphagia. Dig Dis Sci. 2016;61:2935–2941.[↩][↩][↩][↩][↩][↩]
- Niewiarowski T J, Stoll L M. Recurrent dysphagia in a patient with chronic lymphocytic esophagitis. Gastrointest Endosc. 2016 doi: 10.1016/j.gie.2016.02.016[↩]
- Haque S, Genta R M. Lymphocytic oesophagitis: clinicopathological aspects of an emerging condition. Gut. 2012;61:1108–1114.[↩][↩][↩][↩][↩][↩]
- Cohen S, Saxena A, Waljee A K et al.Lymphocytic esophagitis: a diagnosis of increasing frequency. J Clin Gastroenterol. 2012;46:828–832.[↩][↩][↩]
- Nayi VR, Johnson DA (2015) Lymphocytic Esophagitis: A Newly Recognized Mimic of Eosinophilic Esophagitis. J Gastrointest Dig Syst 5:357. doi:10.4172/2161-069X.1000357 https://www.omicsonline.org/open-access/lymphocytic-esophagitis-a-newly-recognized-mimic-of-eosinophilicesophagitis-2161-069X-1000357.pdf[↩][↩][↩]
- Purdy JK, Appelman HD, Golembeski CP, McKenna BJ (2008) Lymphocytic esophagitis: a chronic or recurring pattern of esophagitis resembling allergic contact dermatitis. Am J Clin Pathol 130: 508-513.[↩][↩][↩][↩]
- Rubio CA, Sjodahl K, Lagergren J (2006) Lymphocytic esophagitis: a histologic subset of chronic esophagitis. Am J Clin Pathol 125: 432-437.[↩]
- Basseri B, Vasiliauskas EA, Chan O, Wang HL, Basseri RJ1, et al. (2013) Evaluation of peripapillary lymphocytosis and lymphocytic esophagitis in adult inflammatory bowel disease. Gastroenterol Hepatol (N Y) 9: 505-511.[↩][↩]
- Sutton LM, Heintz DD, Patel AS, Weinberg AG (2014) Lymphocytic esophagitis in children. Inflamm Bowel Dis 20: 1324-1328.[↩][↩][↩]
- Ebach DR, Vanderheyden AD, Ellison JM, Jensen CS (2011) Lymphocytic esophagitis: a possible manifestation of pediatric upper gastrointestinal Crohn’s disease. Inflamm Bowel Dis 17: 45-49.[↩]
- Mandaliya R, DiMarino A, Cohen S. Lymphocytic esophagitis mimicking eosinophilic esophagitis. Ann Gastroenterol. 2012;25:355–357.[↩]
- Cohen S, Saxena A, Waljee AK, Piraka C, Purdy J, et al. (2012) Lymphocytic esophagitis: a diagnosis of increasing frequency. J Clin Gastroenterol 46: 828-832.[↩][↩][↩][↩][↩][↩]
- Hendy PJ, Wong DS, Florin TH (2013) Spontaneous oesophageal perforation: an unreported complication of lymphocytic oesophagitis. Gut 62: 1668-1669.[↩]
- Rubio C A, Sjödahl K, Lagergren J. Lymphocytic esophagitis: a histologic subset of chronic esophagitis. Am J Clin Pathol. 2006;125:432–437.[↩][↩]
- Purdy J K, Appelman H D, Golembeski C P et al.Lymphocytic esophagitis: a chronic or recurring pattern of esophagitis resembling allergic contact dermatitis. Am J Clin Pathol. 2008;130:508–513.[↩]
- Dellon E S, Rusin S, Gebhart J H et al.A Clinical Prediction Tool Identifies Cases of Eosinophilic Esophagitis Without Endoscopic Biopsy: A Prospective Study. Am J Gastroenterol. 2015;110:1347–1354.[↩]
- Haque S, Genta RM (2012) Lymphocytic oesophagitis: clinicopathological aspects of an emerging condition. Gut 61: 1108-1114.[↩][↩][↩][↩]
- Tanaka K, Rubio CA, Dlugosz A, Truskaite K, Befrits R, et al. (2013) Narrow-band imaging magnifying endoscopy in adult patients with eosinophilic esophagitis/esophageal eosinophilia and lymphocytic esophagitis. Gastrointest Endosc 78: 659-664.[↩][↩][↩]
- urdy J K, Appelman H D, Golembeski C P et al.Lymphocytic esophagitis: a chronic or recurring pattern of esophagitis resembling allergic contact dermatitis. Am J Clin Pathol. 2008;130:508–513.[↩]
- Dellon ES, Speck O, Woodward K, Gebhart JH, Madanick RD, et al. (2013) Clinical and endoscopic characteristics do not reliably differentiate PPI-responsive esophageal eosinophilia and eosinophilic esophagitis in patients undergoing upper endoscopy: a prospective cohort study. Am J Gastroenterol 108: 1854-1860.[↩]
- Arias A, González-Cervera J, Tenias JM, Lucendo AJ (2014) Efficacy of dietary interventions for inducing histologic remission in patients with eosinophilic esophagitis: a systematic review and meta-analysis. Gastroenterology 146: 1639-1648.[↩]
- Figueiredo PC, Pinto-Marques P, Borralho P, Freitas J (2014) Unusual cause for smoldering dysphagia. Lymphocytic esophagitis. Dysphagia 29: 283-285.[↩]