What is the mediastinum
The mediastinum is a broad central chest cavity that separates the lungs from the other structures in the chest. Mediastinum extends from the sternum to the bodies of the vertebrae, and from the superior thoracic aperture to the diaphragm. Generally, mediastinum is further divided into three main parts: anterior mediastinum, posterior mediastinum, and middle mediastinum. The borders of the mediastinum include the thoracic inlet superiorly, the diaphragm inferiorly, the spine posteriorly, the sternum anteriorly, and the pleural spaces laterally. The mediastinum contains the thymus gland, the pericardia! sac, the heart, the aorta, esophagus, trachea (windpipe) and the major arteries and veins. Additionally, the mediastinum serves as a passageway for structures such as the esophagus, thoracic duct, and various components of the nervous system as they traverse the thorax on their way to the abdomen.
The thoracic cavity has three parts: (a) two lateral parts, each containing a lung surrounded by a pleural cavity and (b) a central band of organs called the mediastinum (“in the middle”). The medial wall of each pleural cavity is the mediastinum. Two pleural cavities, one on either side of the mediastinum, surround the lungs.
The medial surface of the right lung lies adjacent to a number of important structures in the mediastinum and the root of the neck. These include the:
- heart,
- inferior vena cava,
- superior vena cava,
- azygos vein, and
- esophagus.
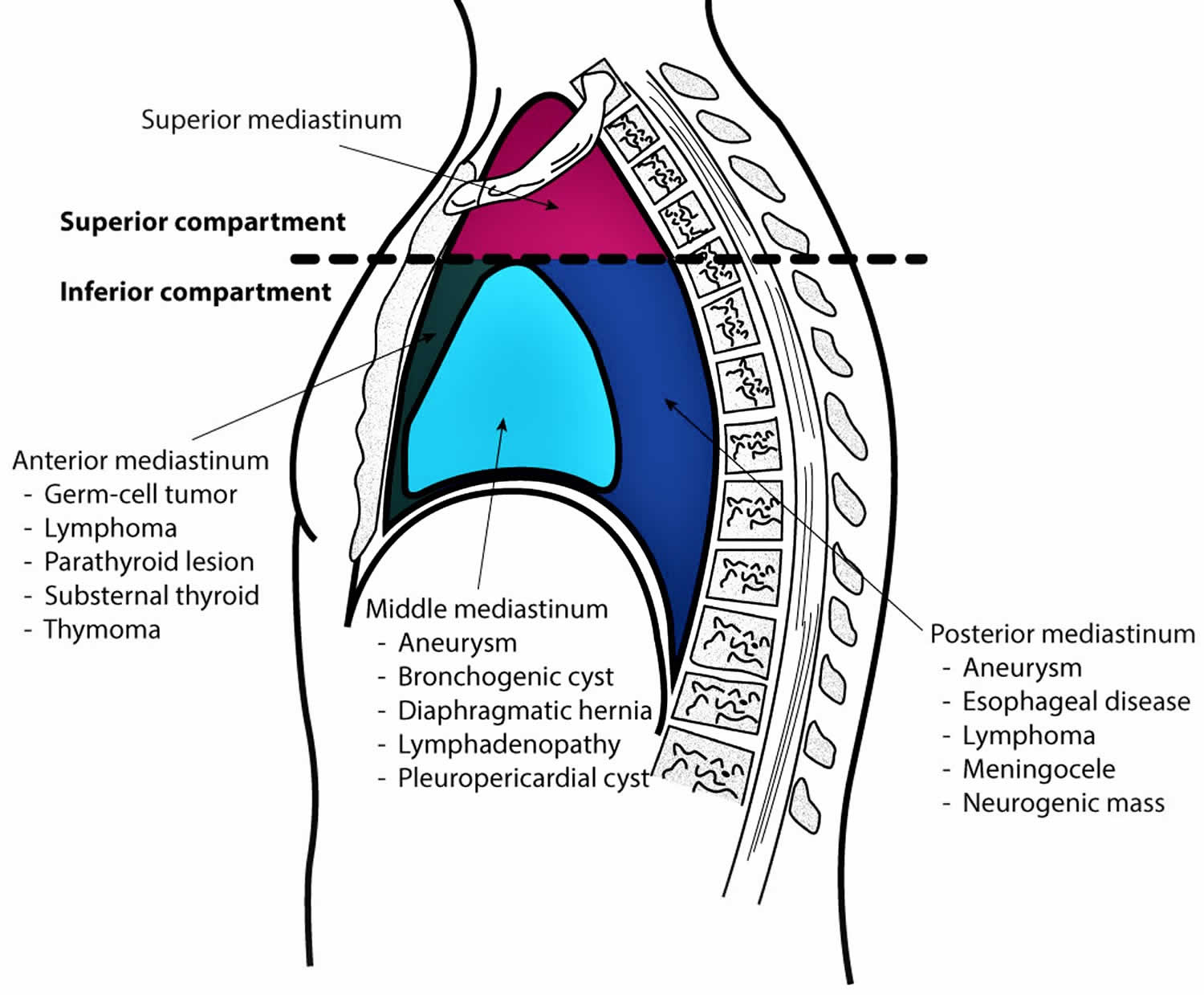
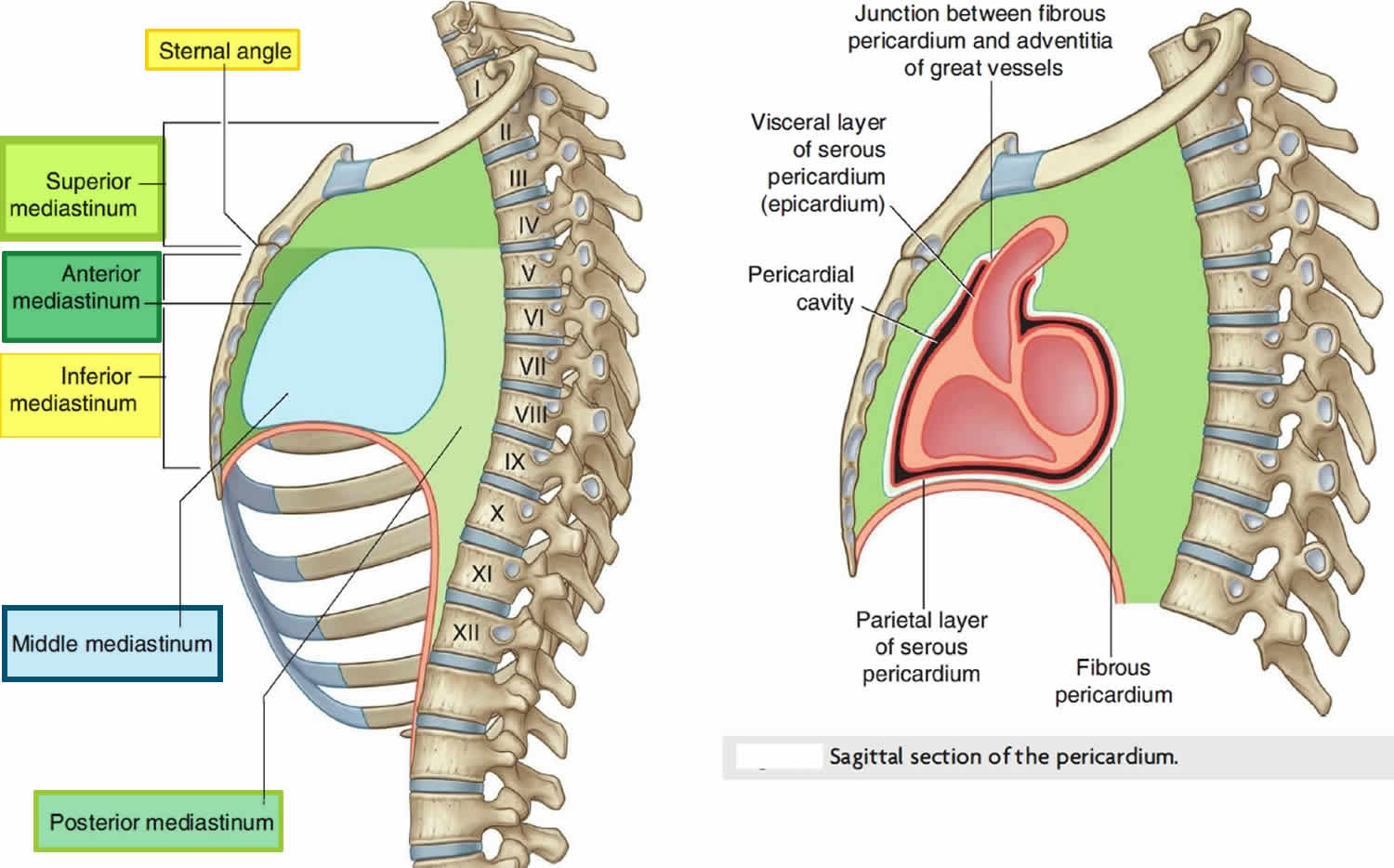
For organizational purposes, the mediastinum is subdivided into several smaller regions. A transverse plane extending from the sternal angle (the junction between the manubrium and the body of the sternum) to the intervertebral disc between vertebrae T4 and T5 separates the mediastinum into the:
- Superior mediastinum, and
- Inferior mediastinum, which is further partitioned into:
- anterior, middle, and posterior mediastinum by the pericardia! sac.
Figure 1. Mediastinum
Figure 2. Mediastinum cavity
Mediastinum function
The mediastinum is constructed in a manner that provides compartments to house and support vital structures within the thorax. Below are the borders of each region of the mediastinum.
- Superior Mediastinum: bordered by the thoracic outlet superiorly, transverse thoracic plane inferiorly, pleural sacs laterally, manubrium of the sternum anteriorly, and first four thoracic vertebral bodies posteriorly
- Anterior Mediastinum: bordered by the pericardium posteriorly, pleural sacs laterally, and bordered anteriorly by the sternum, left transversus thoracis, and fifth, sixth, and seventh left costal cartilages
- Middle Mediastinum: formed by the borders of the pericardial sac
- Posterior Mediastinum: bordered by the pericardium anteriorly, the thoracic surface of the diaphragm inferiorly, the transverse thoracic plane superiorly, the bodies of the fifth to the twelfth thoracic vertebrae posteriorly, and the pleural sacs laterally
Each region of the thoracic mediastinum contains unique structures. Listed below are the different regions of the thoracic mediastinum and a high-level overview of their components.
Each region of the mediastinum can be considered to serve a primary function.
The superior mediastinum is essentially a conduit space which allows for structures to pass between the head, neck, and thorax. The anterior mediastinum is protective in nature and filled with connective and fatty tissue that provides cushioning and support to the thymus as well as the vital cardiac structures just posterior to it. The middle mediastinum is responsible for housing the heart and great vessel roots. The posterior mediastinum, which can be thought of as a continuation of the superior mediastinum, also serves as a conduit which provides space for passage of structures between the thoracic and abdominal cavities.
Although often overlooked, there are many critical muscular structures within the thoracic mediastinum. In the superior thoracic mediastinum is the skeletal and smooth muscle of the esophagus and the left and right inferior oblique part of longus cervicis which assist in mobilizing the cervical spine. In the anterior thoracic mediastinum on the posterolateral sternum originates the transversus thoracis muscles which help in depressing the ribs. In the middle thoracic mediastinum lies arguably one of the most important muscles, the heart. And just posterior to the heart, in the posterior thoracic mediastinum, lies the distal smooth muscle portion of the esophagus. Within blood vessels, especially the large and named vessels such as the aorta, there are many layers of smooth muscle that allow for cardiovascular homeostasis and, although not named as individual muscles, make up a respectable portion of muscular tissue within the mediastinum.
Mediastinum anatomy
Superior mediastinum
Superior mediastinum is posterior to the manubrium of the sternum and anterior to the bodies of the first four thoracic vertebrae (see Figure 1). The superior mediastinum is continuous with the neck above and with the inferior mediastinum below.
- Superior mediastinum superior boundary is an oblique plane passing from the jugular notch upward and posteriorly to the superior border of vertebra T1.
- Inferiorly, a transverse plane passing from the sternal angle to the intervertebral disc between vertebra T4 /T5 separates it from the inferior mediastinum.
- Laterally, it is bordered by the mediastinal part of the parietal pleura on either side.
The major structures found in the superior mediastinum include:
- Organs: thymus, trachea, esophagus
- Arteries: arch of the aorta with its three large branches, brachiocephalic trunk, left common carotid artery, left subclavian artery
- Veins and lymphatics: superior vena cava, right and left brachiocephalic veins, the arch of the azygos, thoracic duct, left superior intercostal vein
- Nerves: left and right vagus nerves, left recurrent laryngeal branch of the left vagus nerve, cardiac nerve, left and right phrenic nerves
Anterior mediastinum
Anterior mediastinum is posterior to the body of the sternum and anterior to the pericardia! sac.
- Anterior mediastinum superior boundary is a transverse plane passing from the sternal angle to the intervertebral disc between vertebra T4 and T5, separating it from the superior mediastinum.
- Anterior mediastinum inferior boundary is the diaphragm.
- Laterally, it is bordered by the mediastinal part of parietal pleura on either side.
The major structures in the anterior mediastinum include:
- Organs: thymus
- Arteries: internal thoracic branches
- Veins and lymphatics: mediastinal branches of the internal thoracic vessels, parasternal lymph nodes
- Nerves: none
Also present are fat, connective tissue and sternopericardial ligaments, which pass from the posterior surface of the body of the sternum to the fibrous pericardium.
Middle mediastinum
Middle mediastinum is centrally located in the thoracic cavity. Middle mediastinum contains the pericardium, heart, origins of the great vessels, various nerves, and smaller vessels.
- Organs: the heart and its great vessel roots
- Arteries: ascending aorta, pulmonary trunk, pericardiacophrenic arteries
- Veins and lymphatics: superior vena cava, pulmonary veins, pericardiacophrenic veins
- Nerve: phrenic nerves, vagus nerves, sympathetic nerves
Posterior mediastinum
Posterior mediastinum is posterior to the pericardial sac and diaphragm and anterior to the bodies of the mid and lower thoracic vertebrae.
Posterior mediastinum superior boundary is a transverse plane passing from the sternal angle to the intervertebral disc between vertebrae T4 and T5.
- Posterior mediastinum inferior boundary is the diaphragm.
- Laterally, it is bordered by the mediastinal part of parietal pleura on either side.
- Superiorly, it is continuous with the superior mediastinum.
Major structures in the posterior mediastinum include:
- Organs: esophagus and its associated nerve plexus
- Arteries: thoracic aorta and its branches
- Veins and lymphatics: azygos vein, hemiazygos vein, thoracic duct and associated lymph nodes
- Nerve: vagus nerve, sympathetic trunks and thoracic splanchnic nerves
Mediastinum cavity clinical significance
The thoracic Mediastinum is a clinically significant region of the human body. Vital cardiopulmonary structures reside within this region. Below is an abbreviated list of significant clinical correlations.
The superior thoracic mediastinum is clinically relevant due to its large vessels and nerves. Penetrating wounds to this area have a high likelihood of affecting important vessels or nerves resulting in critical damage on impact, as well as during extraction 1. The arch of the aorta, the site of a potential aneurysm, runs through the center of the superior thoracic mediastinum and is where the entirety of the of cardiac output, travels before being dispersed throughout the body. An aneurysm in the arch of the aorta is dangerous and if untreated for long enough can dissect and result in near instant death. The superior thoracic mediastinum also houses parts of the esophagus and trachea, which are both conduits that commonly are obstructed and injured by ingestion and inhalation of foreign substances.
The anterior mediastinum may appear clinically benign, but it is responsible for many clinical considerations. The thymus is in the superior portion of the anterior mediastinum, and this organ is notorious for its root in diseases such as myasthenia gravis, pure red cell aplasia, and thymus cancer 2. Additionally, the anterior mediastinum is located directly posterior to the sternum and is therefore vulnerable to trauma to the anterior thorax which can result in an intrathoracic or thymic hematoma.
The middle mediastinum is extremely clinically significant as it contains the pericardium, heart, and great vessel roots. Myocardial infarction, pericardial effusion, cardiac tamponade, Tetralogy of Fallot, and cardiomegaly are just a few cases of pathology which isolate to the middle thoracic mediastinum.
The posterior mediastinum piggybacks a large amount of its clinical importance from the structures descending from the superior thoracic mediastinum. The descending aorta, autonomic nervous networks, the extensive lymphatics, and the esophagus are all capable of causing vast systemic dysfunction in the presence of pathology. Descending thoracic aortic aneurysm, thoracic duct obstruction, and distal esophageal-related dysphagia are problems that can quickly manifest into life-threatening situations.
These examples are just a few of the many clinical correlations of the thoracic mediastinum. These compartments and their structures are often involved in disease and trauma, and therefore understanding the distinct regions of the thoracic mediastinum, the contained structures, landmarks, and physiologic variants are all beneficial to an effective clinician at all levels.
Widened mediastinum
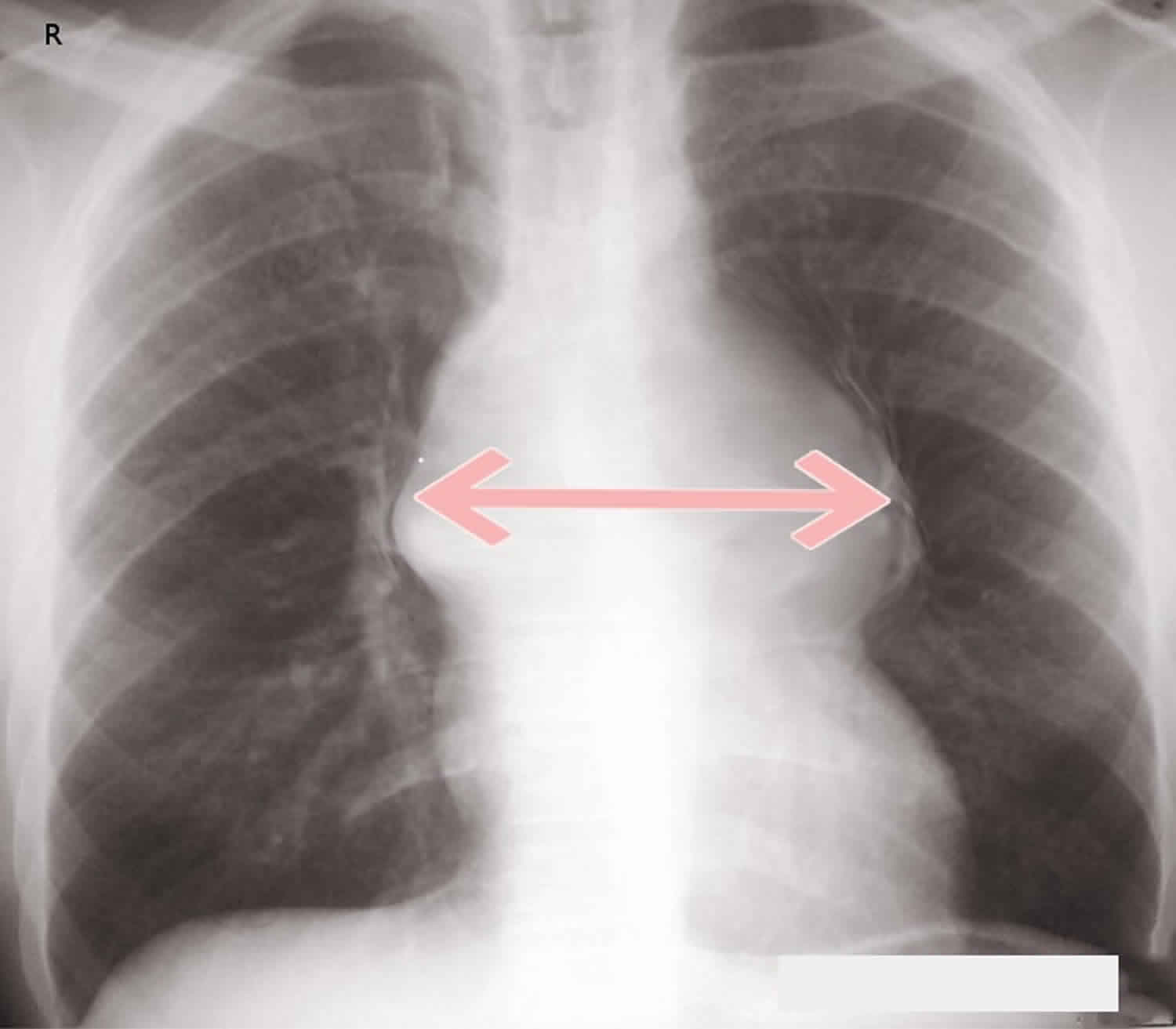
A widened mediastinum is a feature often seen on a plain chest x-ray. When the mediastinum is greater than 6 to 8cm, depending on which source, it is noted to be wide 3. A wide mediastinum has many causes which include the following 3:
- Thoracic aortic aneurysm of the ascending and proximal descending aorta
- Aortic dissection of ascending and proximal descending aorta
- Unfolding of the aorta
- Traumatic aortic rupture
- Hilar lymphadenopathy either infectious or malignant
- Mediastinal masses like lymphoma, seminoma, thymoma
- Mediastinitis
- Cardiac tamponade
- Fractured ribs or thoracic vertebrae
The most significant life-threatening concern associated with a wide mediastinum is an acute aortic rupture or aortic dissection. Additional imaging must be obtained to investigate these diagnoses 4. In a stable patient, CT angiography of the chest is the best study for aortic pathology. However, if there is concern that the patient is unstable, they should not be transported to the radiology suite. In this instance, bedside ultrasound is an option which may reveal aortic pathology.
In all cases, both immediate and potential causes of a widened mediastinum must merit consideration and be ruled out.
Figure 3. Widened mediastinum
[Source 3 ]A widened mediastinum will often be first noted in the emergency department on a chest x-ray. The mediastinal borders are composed of the right heart, ascending aorta, superior vena cava, aortic knob, descending aorta, and the left heart. These structures are all contained between the right and left lungs. The mediastinum can be partitioned into three compartments from anterior to posterior, which is best viewed on the lateral film. The anterior compartment contains the heart, thyroid, and thymus. The middle mediastinal compartment includes the esophagus, trachea, and many lymph nodes. The posterior compartment is the paravertebral area and is primarily composed of the vertebral column 5. The definition of a widened mediastinum is a measured mediastinal width greater than 6 to 8 cm, depending on which source you use 6. This finding is very sensitive for major vascular injury in the setting of trauma but can have numerous additional associated pathologies, as noted above. Pathology causing a widened mediastinum may also compress or displace nearby structures, such as the trachea.
The first consideration when a widened mediastinum is noted on x-ray is the quality of the chest film as poor technique can cause this presentation. The proper distance for taking a standard PA chest x-ray is with 6 feet between the x-ray source and the detector plate. If the distance between the detector plate and the x-ray source is closer than 6 feet, the structures in the film will appear larger and less sharp. Additionally, an AP chest x-ray, typically obtained as a portable chest x-ray, is taken at a closer distance and therefore also reveals larger and less sharp structures. Patient positioning also plays an important role. If the patient is rotated or slouched, the mediastinum may appear falsely widened. After you have ensured the processing of an adequate film, the second consideration is to compare the chest x-ray to prior studies, if available. Typically, more chronic causes of a widened mediastinum, such as a mass or lymphadenopathy, cause focal widening, whereas acute causes of a widened mediastinum, like infection or bleeding, cause diffuse, generalized widening 7. After ensuring that both the execution of the radiograph was with proper technique and the widened mediastinum is a new finding, it is time to pursue this finding further.
Tumor mediastinum
Mediastinal cancers are usually rare. Typically, they are diagnosed in patients aged 30 to 50 years but can develop at any age from any tissue that is located or passes through the mediastinum. Children usually present with cancers in the posterior mediastinum, while adults usually present with cancers in the anterior mediastinum. There is a similar incidence in men and women, but it can vary with the type of cancer present 8. Cancers in the mediastinum can develop from structures that are anatomically located inside the mediastinum or that transverse through the mediastinum during development, and also from metastases or malignancies that originate elsewhere in the body 9.
There are many different types of mediastinal cancers. Their location in the mediastinum can further determine the causes of these cancers 10.
Anterior mediastinum
Thymic carcinoma
Thymic carcinoma is a rare but aggressive, early-metastasizing cancer derived from thymic epithelial cells. It is also the most common cancer of the anterior mediastinum and can be further differentiated into squamous cell carcinoma, basaloid carcinoma, mucoepidermoid carcinoma, lymphoepithelioma-like carcinoma, sarcomatoid carcinoma, clear-cell carcinoma, adenocarcinomas, NUT carcinoma, and undifferentiated carcinoma 11.
Germ cell tumors
The most common location for malignant, germ cell tumors are the gonads; however, they can also arise in extragonadal regions. The mediastinum is the most common known location for extragonadal germ cell tumors. It has been speculated that they occur in these locations due to abnormal migration of germ cells during embryogenesis. In adults, 10% to 15% of all mediastinal tumors are germ cell tumors, while in children, approximately 25% of tumors are germ cell tumors. Thirty percent to 40% of these germ cell tumors are malignant and found mostly in men. When a mediastinal germ cell tumor is discovered, primary testicular or ovarian germ cell tumors should be excluded because the mediastinum could be a site of metastasis. Germ cell tumors include seminomas (termed dysgerminomas in women), embryonal carcinoma, yolk sac tumor, choriocarcinoma, immature teratoma, and mature teratomas. Mature teratomas of the testis are considered cancerous, and it can metastasize to the mediastinum. Other germ cell tumors of the mediastinum include mixed germ cell tumor, germ cell tumor with somatic type solid malignancy, and germ cell tumor with associated hematological malignancy 11.
Lymphoma
Primary mediastinal lymphoma is a relatively uncommon tumor. It is generally observed in senior women and men. The most common types of primary lymphoma which present as a disease in the mediastinum are nodular sclerosing Hodgkin lymphoma and primary mediastinal large B-cell lymphoma. Others include extranodal marginal zone lymphoma of mucosa-associated lymphoid tissue (MALT lymphoma), other mature B-cell lymphomas, T-lymphoblastic lymphoma/leukemia, anaplastic large cell lymphoma, and other rare mature T-cell and NK-cell lymphomas, Hodgkin lymphoma (other types). Secondary lymphomas of the mediastinum, which originate elsewhere in the body and metastasize to the mediastinum are more common than primary mediastinal lymphomas 12.
Mediastinal thyroid mass
Usually benign, these can become cancerous
Posterior mediastinum
Neurogenic mediastinal neoplasms
These are derived from neural cells in any location but are commonly found in the mediastinum. These neurogenic tumors represent more than 60% of the masses found in the posterior mediastinum, are mostly found in children, and can reach a large size before becoming symptomatic. Approximately 30% of neurogenic neoplasms are malignant. They present with a large variety of both clinical and pathological features that are classified by the origin of the cell type. These are the most common cause of neurogenic mediastinal cancers and are classified as nerve sheath neoplasms (Schwannomas and Neurofibromas), these can rarely become malignant. Ganglion cell neoplasms (neuroblastomas and ganglioneuroblastomas) are malignant tumors that occur mostly in children. Paraganglionic cell neoplasms (pheochromocytomas and paragangliomas).
Middle mediastinum
Thyroid cancers
Metastasis from thyroid cancers can spread to the middle mediastinum.
Mediastinal thyroid mass
Usually benign, these can occasionally become cancerous.
Metastatic lung cancers
Lung cancers can metastasize to the mediastinum either by direct extension or lymph node metastasis.
Esophageal cancers
Advanced esophageal cancers can present as a mass in the mediastinum.
Mediastinum tumor diagnosis
Cancers in the mediastinum have a wide diversity of presentation 13. The location of cancer and its composition are paramount to narrowing the differential diagnosis. The tests performed most commonly to diagnose and evaluate mediastinal cancers include:
Chest X-ray
Chest radiographs (posteroanterior and lateral) are usually the first step in the discovery of a mediastinal mass and can help localize the mass is in the anterior, posterior or medial mediastinum to help with the differentials. The mass could be an incidental finding on an asymptomatic patient who has had a chest x-ray before elective surgery or as an evaluation of an unrelated condition. It can also be done as an evaluation in patients who present with symptoms due to the mediastinal mass, or paraneoplastic syndromes. In general, malignant lesions tend to be symptomatic. However, radiography is of very limited value in characterizing mediastinal cancers.
CT Scan of the Chest
To evaluate masses seen on chest x-rays, CT scan of the chest is usually performed with intravenous (IV) contrast, which can often show its exact location, whether the mass is well circumscribed, or if it infiltrates the surrounding structures. In many cases, no further workup is needed for diagnosis. Characterization of the mass on the CT scan is based on specific attenuation of water, air, fat, calcium, soft tissue, and vascular structures. High-resolution multiplanar reformation images demonstrate the detailed anatomical relationship of cancer’s precise location, morphology, as well as its relationship to the adjacent structures.
MRI of the Chest
MRI is indicated when CT finding is equivocal. Recently, special applications of MRI have been developed to identify precisely the tissular components of mediastinal masses. Excellent soft tissue contrast makes MRI an ideal tool to evaluate cancers of the mediastinum. Chemical-shift MRI has been helpful in differentiating normal thymus and thymic hyperplasia from thymic cancers and lymphoma. Diffusion-weighted MRI (DWI), another special application that unveils minute biophysical and metabolic differences between tissues and structures. According to Gumustas et al., the mean apparent diffusion coefficient for malignant mediastinal entities could be substantially lower than that for the benign diseases.
Mediastinoscopy with Biopsy
This test collects cells from the mediastinum to determine the type of mass present for a definitive diagnosis. It is done under general anesthesia. A small incision is made under the sternum, and a small sample of tissue is removed to analyze if cancer is present. This test will help the physician determine the type of cancer present with very high sensitivity and specificity. Establishing the diagnosis of lymphoma generally requires a core biopsy for flow cytometry.
Endobronchial ultrasound (EBUS) and Endoscopic ultrasound (EUS)
Endobronchial ultrasound has become the diagnostic procedure of choice for most of the mediastinal pathologies. Most of the mediastinal lymph node stations including station 2, 4, 7, 10, and 11 can be accessed with the help of an EBUS. Combined with EUS even other stations like 7, 8, and 9 can be accessed. EBUS can be done under conscious sedation or general anesthesia. It is usually a single-day, outpatient procedure with minimal complications. It is found to be safer than mediastinoscopy with almost similar diagnostic efficacy.
Laboratory Studies
Basic Laboratory tests such as complete blood count (CBC) and basic metabolic panel (BMP) can help in the differential diagnosis of lymphomas. In the instances of mediastinal masses, tumor markers can help support a presumptive diagnosis. These include:
- Beta-hCG (associated with germ cell tumors and seminoma)
- LDH (may be elevated in patients with lymphoma)
- AFP (associated with malignant germ cell tumors): Mediastinal nonseminomatous germ cell tumors are more likely to result in distinct elevations of serum AFP and less likely in elevations of beta-hCG compared to gonadal or retroperitoneal nonseminomatous germ cell tumors. This was demonstrated by Bokemeyer et al. Malignant germ cell tumors are closely related to serum tumor markers, especially AFP and beta-hCG and measurement of these serum tumor markers is important in diagnosis, management and follow-up of these patients.
Testicular Ultrasound
In suspected cases of metastasis to the mediastinum from primary testicular germ cell tumors, palpation of the testicles is not sufficient, and ultrasonography of the testicles should be performed in all patients.
Mediastinum tumor treatment
The treatment for mediastinal cancers depends primarily on the type of cancer, its location, aggressiveness, and symptoms it may be causing. Treatments of the most common mediastinal cancers are discussed below 14.
Thymic cancers
Treatment of thymic cancers usually requires surgery, followed by radiation or chemotherapy. Surgery is the initial treatment in patients who present with tumors invading readily resectable structures like the mediastinal pleura, pericardium or adjacent lung. A resected specimen will then be sent for histopathologic examination for staging and to determine if postoperative radiotherapy or chemotherapy are required. The ability to completely resect the thymic carcinoma is dependent on the degree of invasion and/or adherence to surrounding structures. Sometimes to achieve complete resection with histologically negative margins, the pericardium and surrounding lung parenchyma are also removed. Types of surgery which are being performed include thoracoscopy (minimally invasive), mediastinoscopy (minimally invasive), and thoracotomy (performed by making an incision in the chest). In patients for whom complete resection is not feasible as the initial treatment, for example, those with cancer invasion into the innominate vein, phrenic nerve, heart, or great vessels, therapy involving preoperative chemotherapy and postoperative radiotherapy is indicated. If neoadjuvant chemotherapy allows for a partial or complete response, these cancers are considered potentially resectable, and patients should be reevaluated following therapy. If complete resection cannot be accomplished with surgery, these patients should undergo maximum debulking followed by radiotherapy postoperatively if technically feasible. This may control the residual disease. For unresectable cancers, systemic therapy, radiotherapy, or chemoradiotherapy may be recommended in patients who have extensive pleural/pericardial metastases, unreconstructable great vessel invasion, heart, tracheal involvement, or distant metastases. Although there are no clinical trials providing evidence of the benefit of surveillance after treatment, monitoring with thoracic imaging is warranted given early intervention may be more effective. The National Comprehensive Cancer Network recommends a CT scan every 6 months for 2 years, then annually for 5 years. The prognosis for patients with thymic carcinoma depends on the stage of disease, complete resectability, and histologic type 15.
Lymphomas
Treatment depends on the type of lymphoma, its stage, and metastases. Most lymphomas are generally treated with chemotherapy followed by radiation 16.
- Diffuse large B-cell lymphoma are known to grow quickly. Most often, the treatment of choice is chemotherapy with a combination of 4 drugs: cyclophosphamide, doxorubicin, vincristine, and prednisone (known as CHOP), and the monoclonal antibody rituximab. This combination, also called R-CHOP, is commonly given in cycles separated by 3 weeks. Because this regimen includes the drug doxorubicin, which could damage the heart, it is not suitable for patients who have heart problems, and the use of other chemotherapy regimens is recommended.
- Primary mediastinal B-cell lymphoma originates in the mediastinum and is essentially managed like the early-stage of diffuse large B-cell lymphoma. The primary treatment is around 6 courses of CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisone) plus rituximab (R-CHOP). This could be followed by radiation to the mediastinum in suitable patients. A PET/CT scan is usually done after the chemotherapy to see if there are any lymphoma remnants in the chest. If there is no active lymphoma is observed on the PET/CT scan, the patient may be observed without further intervention. However, if the PET/CT scan shows a possible active lymphoma in the mediastinum, radiation may be needed. Often, a biopsy of the chest tumor is ordered by the physician to affirm that the lymphoma is still present before starting radiation.
- Extranodal marginal zone lymphoma of mucosa-associated lymphoid tissue (MALT lymphoma) is rare cancer. It can be treated with radiation therapy to the stomach, rituximab, chemotherapy, chemotherapy plus rituximab, or the targeted drug ibrutinib. Chlorambucil or fludarabine could be used for single-agent chemotherapy or combinations such as CHOP (cyclophosphamide, doxorubicin, vincristine, prednisone) or the combination CVP (cyclophosphamide, vincristine, prednisone) could be used. Hodgkin lymphoma treatment usually starts with chemotherapy. The most common 4-drug combination used in the United States is AVCD: adriamycin (doxorubicin), bleomycin, vinblastine, dacarbazine (DITC), which is given in cycles. Radiation therapy is often given after chemotherapy.
Neurogenic tumors
Surgery is the preferred treatment for malignant neurogenic tumors. Procedures include a post-lateral thoracotomy which can give the surgeon good exposure to the tumor, and more recently video-assisted thoracic surgery (VATS). VATS is a minimally invasive procedure that inserts a thoracoscope and surgical instruments through small incisions in the chest. This procedure decreases the access trauma and gives a good view, especially for smaller posterior mediastinal neurogenic tumors, but is not ideal for giant mediastinal neurogenic tumors because of their huge volume. For dumbbell neurogenic tumors, sufficient preoperative evaluation is required of the intraspinal part of the tumor to prevent uncontrollable hemorrhage during surgery. Cooperation of a thoracic surgeon with a neurosurgeon is recommended for this procedure. For malignant mediastinal neurogenic tumors, the five-year survival rate is low and complete resection is rarely possible. There is frequent use of radiation in conjunction with chemotherapy before surgery to decrease the size of the tumor or after to treat the margin of the resection bed.
Mediastinal germ cell tumors
Management of mediastinal germ cell tumors varies with the type of tumor present. It may include chemotherapy, surgery, radiotherapy, or a combination of these treatments.
- Mediastinal seminoma or dysgerminoma: Treatment essentially depends on the size of the tumor. For smaller sizes, patients usually undergo radiotherapy, and this can eradicate the tumor. For larger tumors, the treatment of choice is chemotherapy. This is most commonly a combination of bleomycin, etoposide, and cisplatin (BEP). Radiation has a higher rate of recurrence compared to chemotherapy. If these treatments fail to get rid of all the tumor mass and the remaining area is smaller than 3 cm, patients are likely to be monitored periodically by the physician to see if the tumor grows again. If the residual tumor is greater than 3 cm after treatment, the physician can opt for routine monitoring or further surgery to remove the tumor.
- Mediastinal nonseminomatous germ cell tumors: Chemotherapy is the first line treatment for primary mediastinal non-seminomatous germ cell tumors. A combination of drugs is traditionally used, the most common being BEP (bleomycin, etoposide, and cisplatin), and standard therapy consists of 4 courses of BEP. Men with nonseminomatous primary mediastinal germ cell tumor have a worse prognosis of survival compared to men with mediastinal seminoma. After chemotherapy, patients may undergo a salvage surgery to remove the remaining tumor. The optimal strategy for long-term survival in patients who present with mediastinal nonseminomatous germ cell tumors remains to be defined. A study in Indiana University described 31 patients with mediastinal nonseminomatous germ cell tumors who received cisplatin, bleomycin, and either vinblastine (20 patients) or etoposide (11 patients) followed by surgical resection; 15 patients (48%) achieved long-term disease-free survival. A retrospective study of 64 patients with mediastinal nonseminomatous germ cell tumors (NSGCTs), who were treated in France from 1983 through 1990, estimated a 2-year overall survival rate of 53%. More recently, a 5-year overall survival rate of 45% was reported in an international analysis of 141 patients with mediastinal NSGCT from 11 cancer centers, treated from 1975 through 1996 in the United States and Europe. Treatment variables that may determine survival outcomes include the duration the patient received chemotherapy, the use of additional anti-cancer drugs such as anthracyclines, alkylating agents, and when the surgery was performed.
- Salhiyyah K, Ashoub A, Diprose P, Barlow C. Knife in the superior mediastinum: Amazing escape. Ann Card Anaesth. 2017 Apr-Jun;20(2):247[↩]
- Yasuda M, Osaki T, Fukuich Y, Kobayashi K, Iwata T, So T. Anterior mediastinal tumor as a solitary lymph node metastasis of occult thyroid carcinoma. J Surg Case Rep. 2019 Feb;2019(2):rjz029[↩]
- Wackerman L, Gnugnoli DM. Widened Mediastinum. [Updated 2019 Mar 12]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2019 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK539890[↩][↩][↩]
- Ong D, Cheung M, Cuenca P, Schauer S. Clinical Utility of Routine Chest X-Rays During the Initial Stabilization of Trauma Patients. South. Med. J. 2019 Jan;112(1):55-59[↩]
- Goizueta AA, Bordoni B. StatPearls [Internet]. StatPearls Publishing; Treasure Island (FL): Jan 13, 2019. Anatomy, Thorax, Lung Pleura And Mediastinum[↩]
- Gutierrez A, Inaba K, Siboni S, Effron Z, Haltmeier T, Jaffray P, Reddy S, Lofthus A, Benjamin E, Dubose J, Demetriades D. The utility of chest X-ray as a screening tool for blunt thoracic aortic injury. Injury. 2016 Jan;47(1):32-6[↩]
- Taghavi S, Askari R. StatPearls [Internet]. StatPearls Publishing; Treasure Island (FL): Jan 13, 2019. Mediastinal Trauma[↩]
- Khatami A, Outhred AC, Britton PN, Huguon E, Lord DJ, Wong M, Charlton A, Kesson AM, Isaacs D. Mediastinal mass in a healthy adolescent at The Children’s Hospital at Westmead, Australia. Thorax. 2015 Feb;70(2):194-7[↩]
- Jilani TN, Siddiqui AH. Cancer, Mediastinum. [Updated 2019 Mar 24]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2019 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK513231[↩]
- Rajdev K, Siddiqui AH, Agarwal S, Ansari Y, Maroun R. Mediastinal recurrence after curative resection of colon cancer: A case report and review of literature. J Oncol Pharm Pract. 2018 Jan 01;:1078155218784744[↩]
- Carter BW, Lichtenberger JP, Benveniste MF. MR Imaging of Thymic Epithelial Neoplasms. Top Magn Reson Imaging. 2018 Apr;27(2):65-71[↩][↩]
- Tang VK, Vijhani P, Cherian SV, Ambelil M, Estrada-Y-Martin RM. Primary pulmonary lymphoproliferative neoplasms. Lung India. 2018 May-Jun;35(3):220-230[↩]
- Hammer MM, Miskin N, Madan R, Hunsaker AR. Predictive Features for Anterior Mediastinal Mass Diagnoses. J Comput Assist Tomogr. 2019 Jan/Feb;43(1):98-103.[↩]
- Rajdev K, Siddiqui AH, Ibrahim U, Patibandla P, Khan T, El-Sayegh D. An Unusually Aggressive Large Cell Carcinoma of the Lung: Undiagnosed until Autopsy. Cureus. 2018 Feb 19;10(2):e2202[↩]
- Mou H, Liao Q, Hou X, Chen T, Zhu Y. Clinical characteristics, risk factors, and outcomes after adjuvant radiotherapy for patients with thymoma in the United States: analysis of the Surveillance, Epidemiology, and End Results (SEER) Registry (1988-2013). Int. J. Radiat. Biol. 2018 May;94(5):495-502[↩]
- PDQ Pediatric Treatment Editorial Board. PDQ Cancer Information Summaries [Internet]. National Cancer Institute (US); Bethesda (MD): Dec 18, 2018. Childhood Non-Hodgkin Lymphoma Treatment (PDQ®): Health Professional Version[↩]