What is omentum
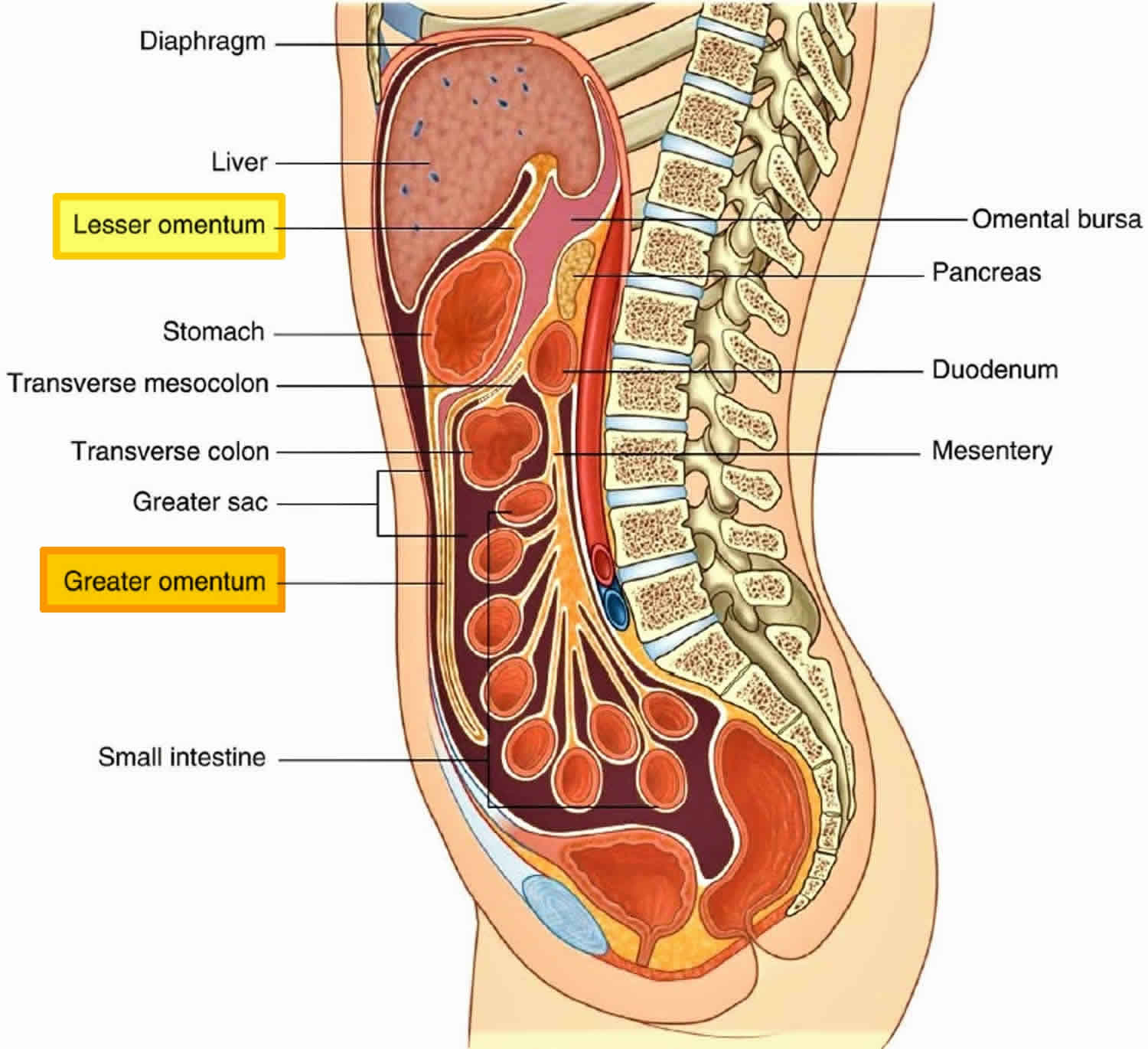
The omentum is part of the mesentery, which consists of two layers of peritoneum, a sheet of two serous membranes fused together that extends from the body wall to the digestive organs (Figures 1 and 2). The omentum pass from the stomach and the first part of the duodenum to other abdominal organs. The size of the omentum varies from 300 gm to 2000 gm with a surface area of 300 cm2 to 1500 cm2 1.
There are two omentum:
- Greater omentum, derived from the dorsal mesentery,
and - Lesser omentum, derived from the ventral mesentery.
The peritoneum lines the walls of the abdominal cavity and covers much of the viscera. The parietal peritoneum lines the walls of the cavity and the visceral peritoneum covers the viscera. Between the parietal and visceral layers of peritoneum is a potential space (the peritoneal cavity). Abdominal viscera either are suspended in the peritoneal cavity by folds of peritoneum (omentum or mesenteries) or are outside the peritoneal cavity. Organs suspended in the cavity are referred to as intraperitoneal; organs outside the peritoneal cavity, with only one surface or part of one surface covered by peritoneum, are retroperitoneal.
In the omentum, the leukocytes aggregate in the perivascular area to form what is termed milky spots 1. The cells derive their origin from the mononucl ear phagocyte system 2 and are arranged around the omental glomeruli that lie directly beneath the mesothelium 3. These structures are supported by delicate networks of reticular fibres which constitute the framework of the organ 4. In humans, milky spots comprise of macrophages (70%), B-lymphocytes (10%), Tlymphocytes (10%), mast cells, and stromal cells. On an ultrastructural level, it has been found that the macrophages are present in different stages of maturation, and that they can readily enter or leave the milky spots 5. The mean number of cells in one milky spot is approximately 600 6. Milky spots develop as specific structures in the greater omentum between the 20th and 35th week of gestation 7. The number of milky spots is highest in infancy and gradually decreases with age 8.
Both the endothelium lining the omental capillaries and the mesothelium overlying the milky spots are specially adapted to facilitate transmigration of leukocytes 9, and for rapid fluid shifts. The endothelial lining of the blood vessels in the milky spots is either discontinuous or contain fenestrations 10. Similarly, there are intercellular pores (the classical stomata of von Recklinghausen) between the mesothelial cells overlying milky spots, and there is an absence of the associated basal lamina in the submesothelial connective tissue 11.
The macrophages in the mature omentum are essentially scavengers. They appear to differentiate from monocytic precursors in the milky spots and are not dependent on precursors derived from the bone marrow 12. They are dendritic in shape and have marked phagocytic abilities. They avidly phagocytose intraperitoneally injected carbon particles and bacteria. When activated, the macrophage precursors in the milky spots proliferate, migrate to the mesothelial surface, and transform into dendritic-shaped macrophages. This process in mice is dependent on macrophage colony stimulating factor (MCSF) being locally produced in the milky spots 12. Interestingly, the omental macrophages, despite their dendritic shape, lack many specific features of true dendritic cells.
The omentum contains large numbers of B and T lymphocytes which are usually located in the periarteriolar area. Following antigen challenge of the peritoneal cavity, the number of lymphocytes in the milky spots may increase up to 40-fold. Although it is unclear whether this increase represents local proliferation or an influx of cells. With such stimulation, the B and T-lymphocytes are found to segregate into distinct areas in situ, and the lymphocytes appear to be associated with stromal cells. Nonetheless, these aggregates do not represent secondary lymphoid organs, because they do not contain interdigitating cells or follicular dendritic cells 5. The omentum appears to be a primary site of B-lymphocyte development 13. In experimental animals, the omentum is a source of unique B-lymphocytes that demonstrate specific surface markers. These B-lymphocytes are predominantly CD5+ (Lyl+), and are common in not only the omentum but also the peritoneum. However, they are rare in the blood, spleen and lymph nodes. Conventional B and T-lymphocytes are not found in the omentum. The CD5+B lymphocytes develop in the omental milky sopts independently from the thymus or bone marrow 14. Hence, the fetal omentum, like the fetal liver and bone marrow, acts as a primary site of B-lymphocyte development 15 and may be considered as a sort of intestinal thymus 16. The function of these CD5+ B lymphocytes remains obscure, nontheless, they are most likely a remnant of a more primitive immune system which is in keeping with the evolutionary origins of the omentum.
Mesothelial cells lining the peritoneal cavity and endothelial cells lining blood vessels share the same mesodermal origin 17. Human omental microvascular endothelial (HOME) and mesothelial (MESO) cells share many phenotypic propertis. In distinguishing between the two cell types, HOME and not MESO cells express a number of specific surface markers (i.e. E-selectin, P-selectin (CD62), and Le-y) and form tube-like structures when cultured on Matrigel. MESO cells differ from HOME cells based upon the expression of cytokeratins; their rapid proliferation in response to platelet-dervied growth factor, and a change from an epitheloid to fibroblast-like morphology in response to tumour necrosis factor and epidermal growth factor. Both HOME and MESO cells express tissue plasminogen activator and plasminogen activator inhibitor, form typical cobblestone monolayers, and are immunoreactive to von Willebrand Factor and Ulex europaeus I lectin 17. Urokinase activity is only expressed by MESO cells 18.
Figure 1. Peritoneal cavity and omentum
Greater omentum
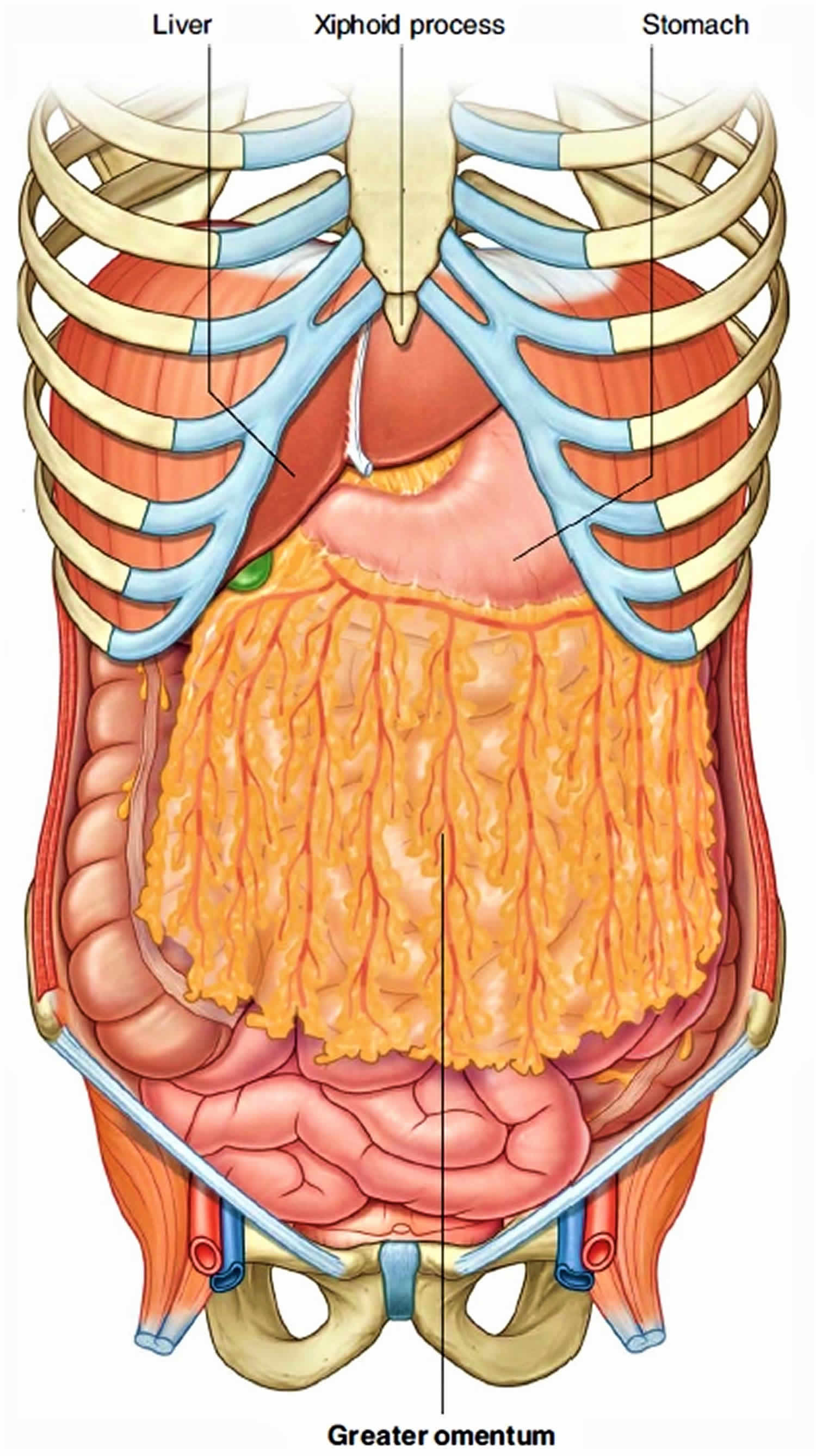
The greater omentum is part of the dorsal mesentery, is a large apron-like fold of peritoneum with a free lower border that attaches to the greater curvature of the stomach and the first part of the duodenum. The greater omentum is the longest peritoneal fold, it drapes over the transverse colon and coils of the jejunum and ileum like a “fatty apron”. The greater omentum is a double sheet that folds back on itself, giving it a total of four layers. Hanging behind the anterior abdominal wall and in front of most of the small intestine, this omentum is usually a conspicuous feature when the peritoneal cavity is opened. Superiorly, the greater omentum attaches to both the transverse colon and the greater curvature of the stomach.
The greater omentum connects the greater curvature of the stomach to the posterior abdominal wall, but in a very roundabout way: Anteriorly, it is tremendously elongated and extends inferiorly to cover the transverse colon and coils of the small intestine like a butterfly net. The left border of the greater omentum wraps around the spleen as the gastrosplenic ligament and continues dorsally as the splenorenal ligament (extending between the spleen and the left kidney) to the posterior body wall.
From attachments along the stomach and duodenum, the greater omentum turning posteriorly, it ascends to associate with, and become adherent to, the peritoneum on the superior surface of the transverse colon and the anterior layer of the transverse mesocolon before arriving at the posterior abdominal wall.
Usually a thin membrane, the greater omentum always contains a considerable amount of adipose tissue, which may become substantial in some individuals. Its adipose tissue content can greatly expand with weight gain, contributing to the characteristic “beer belly” seen in some overweight individuals. The many lymph nodes of the greater omentum contribute macrophages and antibody-producing plasma cells that help combat and contain infections of the gastrointestinal tract. Additionally, there are two arteries and accompanying veins, the right and left gastro-omental vessels, between this double-layered peritoneal apron just inferior to the greater curvature of the stomach.
The long coils of the jejunum and ileum are supported by the mesentery. This sheet fans inferiorly from the posterior abdominal wall like long, pleated curtains. The transverse colon is held to the posterior abdominal wall by the transverse mesocolon, a nearly horizontal sheet that is fused to the underside of the greater omentum, so that it can be viewed only inferiorly. The sigmoid mesocolon is the mesentery that connects the sigmoid colon to the posterior pelvic wall.
Figure 2. Greater omentum
Greater omentum function
When a laparotomy is performed and the peritoneal cavity is opened, the first structure usually encountered is the greater omentum. This fatty double-layered vascular membrane hangs like an apron from the greater curvature of the stomach, drapes over the transverse colon, and lies freely suspended within the abdominal cavity. The greater omentum is often referred to as the “policeman of the abdomen” because of its apparent ability to migrate to any inflamed area and
wrap itself around the organ to wall off inflammation. When a part of bowel becomes inflamed, it ceases peristalsis. This aperistaltic area is referred to as a local
paralytic ileus. The remaining non inflamed part of the bowel continues to move and “massages” the greater omentum to the region where there is no peristalsis. The localized inflammatory reaction spreads to the greater omentum, which then adheres to the diseased area of bowel.
The greater omentum is also an important site for metastatic tumor spread. Direct omental spread by a transcoelomic route is common for carcinoma of the ovary. As the metastases develop within the greater omentum, it becomes significantly thickened. In computed tomography imaging and during laparotmy, the thickened omentum is referred to as an “omental cake.”
Lesser omentum
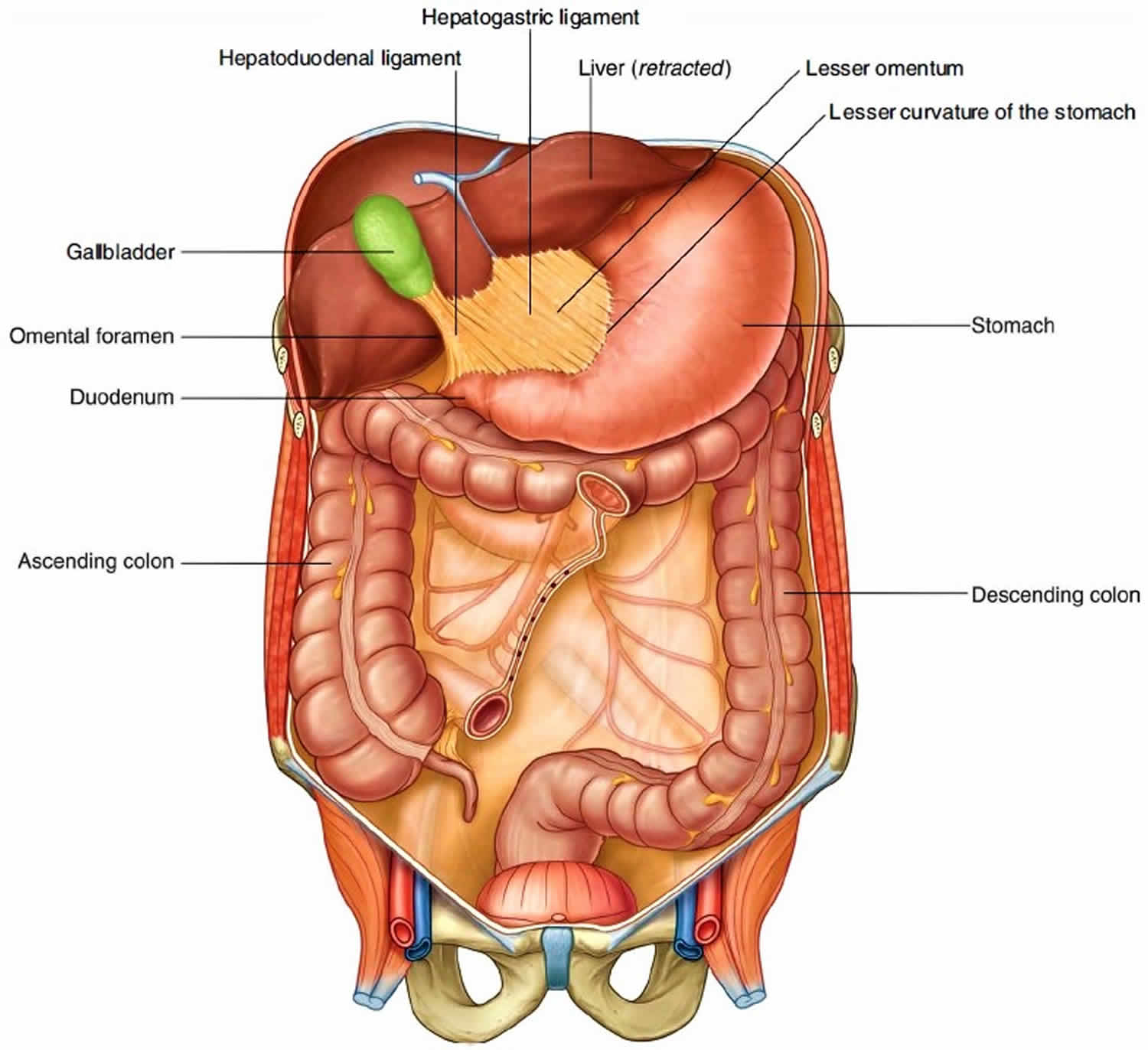
The other two-layered peritoneal omentum is the lesser omentum. The lesser omentum extends from the lesser curvature of the stomach and the first part of the duodenum to the inferior surface of the liver. The lesser omentum connects the stomach and duodenum to the liver. It is the pathway for blood vessels entering the liver and contains the hepatic portal vein, common hepatic artery, and common bile duct, along with some lymph nodes.
A thin membrane continuous with the peritoneal coverings of the anterior and posterior surfaces of the stomach and the first part of the duodenum, the lesser omentum is divided into:
- a medial hepatogastric ligament, which passes between the stomach and liver, and
- a lateral hepatoduodenal ligament, which passes between the duodenum and liver.
The hepatoduodenal ligament ends laterally as a free margin and serves as the anterior border of the omental foramen. Enclosed in this free edge are the hepatic artery proper, the bile duct, and the portal vein. Additionally, the right and left gastric vessels are between the layers of the lesser omentum near the lesser curvature of the stomach.
Figure 3. Lesser omentum
Omentum function
Omentum holds organs in place, store fat, and, most important, provide a route for circulatory vessels and nerves to reach the organs in the peritoneal cavity. Notice that the abdominal aorta is positioned behind the parietal peritoneum. The branches off the aorta that supply the digestive viscera must travel through mesenteries to reach the organs they serve.
If the wall of the alimentary canal becomes infected, cells from the omentum may adhere to the inflamed region, helping to wall off the area. This action prevents spread of the infection to the peritoneal cavity.
The greater omentum contains a great deal of fat and also has a remarkable ability to limit the spread of infections within the peritoneal cavity; for example, it can wrap around and enclose an inflamed appendix.
Peritonitis
The omentum performs a number of functions during episodes of peritonitis. The first of these is the rapid absorption and clearance of bacteria and foreign material from the peritoneal cavity. The omentum is the only site, other than the diaphragmatic stomata, that has a documented ability to absorb particles from the peritoneal cavity 19. But unlike the stomata, the omentum contains potent local effector mechanisms that are mediated by especially macrophages (and probably also B lymphocytes) contained within the milky spots. These macrophages appear to be the principal site for the phagocytosis of particles and bacteria from the peritoneal cavity 8.
The second function of the omentum is to supply leukocytes to the peritoneal cavity. In experimental animals with peritonitis, the omentum appears to be the principal site by which firstly macrophages and then neutrophils migrate into the peritoneal cavity 20. The macrophages are derived from the milky spots which provide the correct microenvironment and growth factors for macrophage proliferation and maturation. The correct microenvironment and growth factors for macrophage proliferation and maturation. The structure of the milky spots and their associated capillary structures aids this process. Because the mesothelium is absent over the milky spots, and the basement membrane is discontinuous, there is rapid exposure of the resident macrophages to intraperitoneal stimulants. this activates the macrophages which then demonstrate marked surface membrane activity and migrate through the stomata of the milky spots into two peritoneal cavity 21.
The omentum also allows for the easy migration of neutrophils from the circulation 22. Due to the structure of the milky spots, there is direct exposure of the postcapillary venules to inflammatory stimuli from the peritoneal cavity 23. The neutrophils are then recruited from the circulation and extravasate via the post-capillary venules in the glomerular tufts into the milky spots and then via the mesothelial stomata into the peritoneal cavity. In one study on mice with peritonitis, the post capillary venules in the milky spots of the omentum were the only abdominal sites detected where plasma extravasation occurred, and the omental milky spots were the major route through which leukocytes migrated into the peritoneal cavity 23. In addition, the omentum was the only abdominal organ which showed an increase in blood flow during peritonitis 23. Milky spots do not seem to serve as a source of dendritic cells 22.
The third function of the omentum is to adhere to and attempt to seal off areas of contamination. The omentum can rapidly produce a layer of fibrin by which to adhere to the contaminated area at the point of contact. In the course of a few days, the fibrin begins to organise with the development of new blood vessels and fibroblasts. In the long term, if the host survives, the contaminated area will be walled of with collagen, and thereby forming dense adhesions 24.
Removal of the omentum impairs peritoneal defence mechanisms 25. In experimental peritonitis, omentectomy has been found to reduce survival[40], and influence a number of peritoneal defence mechanisms 26. In particular, there is a reduction in the total intra peritoneal cell counts, with macrophages being the most effected. There is also a reduction in neutrophil chemotaxis, although their phagocytic activity appeares unchanged 26. In humans, omentectomy also appears to impair the peritoneal defence mechanisms 27. One retrospective analysis compared a group of 406 patients having omentectomy with proctocolectomy and ileoanal anastomosis with a group of 239 patients having a similar procedure without omentectomy 27. The patients were followed up for a mean of 4.3 years. The results showed that the omentectomy group had a significantly higher incidence of postoperative sepsis (4% vs 10%) and sepsis requiring reoperation (3% vs 8%) when compared with patients retaining their omentum. In addition, there was no difference in the incidence of partial or complete small bowel obstruction due to adhesions between the two groups.
Neovascularisation
It has long been recognised that the human omentum can promote angiogenic activity in adjacent structures to which it is applied. Indeed, lipid material obtained from the omentum has been found to induce angiogenesis in rabbit corneas after only a single injection. This angiogenic material obtained from the omentum is abundant in supply 28. Further evaluation of the factors involved in this process have found that the human omental microvascular endothelial cells (HOME cells) express the angiogenic peptide ‘basic fibroblast growth factor 29. This process of neovascularization allows the omentum to provide vascular support to adjacent tissues such as the gut and promote function and healing in ischaemic or inflammed tissue 30. Another example of the angiogenic activity of the omentum has been its ability to support splenic autotransplantations. Although the clinical practice of reimplantation of splenic remnants following splenic injuries has largely been abandoned by surgeons, it is interesting to note that such implants are supported by the omentum and function to a limited capacity 31. The omentum has also be found to be capable of supporting free structures such as the trachea, segments of intestine, sciatic nerve grafts. Such structure can then be used for reconstructive purposes 32.
Foreign bodies
Most surgeons have observed the ability of the omentum to adhere to intra-abdominal foreign bodies such as drains and catheters. In dogs it has been noted that following the placement of various types of drains into the peritoneal cavity, that within seven days all tubes are surrounded and occluded by omentum 33]. This may lead to drainage problems in patients requiring long term catheters placed in the abdominal cavity, such as those for peritoneal dialysis. In such patients, removing the omentum has been found in a number of retrospective and uncontrolled studies to reduce the incidence of catheter blockage and to improve drainage 34. In addition, omentectomy does not appear to alter the peritoneal diffusion capacity 35. Because of this, it has been advocated that partial omentectomy is integral to the surgical technique of peritoneal catheter placement 36. In contrast, Lewis et al 37 argued that the absolute risk reduction with omentectomy in preventing catheter blockage was only 0.18 in a series of 38 patients undergoing 66 catheter placements. It also remains to be clarified whether omentectomy has other deleterious side effects such as an increased incidence of peritonitis. None of the reported trials have adequately addressed this issue.
Omentum cancer
The omentum has been observed to be a frequent site of metastatic disease for many cancers. In animals, malignant cells inoculated into the peritoneal cavity preferentially infiltrate the milky spots in the omentum and grow into distinct metastic 38. The omentum appears capable of supporting not only malignant cells in the milky spots but free intraperitoneal cells. It achieves this due to its intrinsic angiogenic properties. In animals, removing the omentum impacts on the survival of free intraperitoneal malignant cells and there by reduces the rate of local recurrence 39. Because of these observations, the omentum is frequently removed as part of resections for malignancies of various intra abdominal organs 40.
Ovarian cancers, in particular, are characterized by their tendency to spread intraperitoneally and involve the omentum. Hence, there has evolved a general consensus that surgical management of ovarian cancer should include optimal cytoreduction 41. The minimum surgical requirements of this are to perform a total abdominal hysterectomy, bilateral salpingo-oophorectomy, and omentectomy. The apparent value of performing an omentectomy is that it provides staging information and selects patients for adjuvant chemotherapy 42. In addition, in patients with advanced disease, there appears to be a survival advantage in debulking tumour deposits 43. Nonetheless, with borderline ovarian tumours, omentectomy is also frequently advocated but the evidence to support this remains limited 44.
The removal of the omentum in patients with ovarian cancer is not universal. In 1993, a United states national survey of the treatment of patients with ovarian cancer concluded that out of a total of 12, 316 patients with ovarian cancer, the requirements for what were defined as a minimum surgical resection were met in only around 60% of patients (bilateral salpingo-oophorectomy 67%; abdominal hy sterectomy 55%; and omentectomy, 59%) 45. Another analysis of omentectomy in patients with ovarian cancer found that the mean omentectomy size was only 203.5 cm2 (normal omentum = 792 cm2), and secondary ovarian cancer was present in 61%. Thus the optimal extent of omental resection and histological examination remains to be clearly defined 46.
The omentum is intimately associated with the stomach and the gastric lymphatic drainage. Therefore, it is invariably removed as part of a curative resection for gastric cancer 47. Nonetheless, there is no clear evidence to provide guidance as to the extent of such a resection, and whether the entire omentum has to be removed 48. Pseudomyxoma peritonei is a rare neoplasm characterized by mucinous ascites and the mucinous involement of peritoneal surfaces, omentum and bowel loops. Usually pseudomyxoma peritonei is associated with benign or malignant mucinous tumor of the appendix or ovary, and cytoreductive resections, including omentectomy, are advocated as the treatment of choice 49.
- Platell C, Cooper D, Papadimitriou JM, Hall JC. The omentum. World J Gastroenterol. 2000;6(2):169-176. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4723480/[↩][↩]
- Wijffels JF, Hendrickx RJ, Steenbergen JJ, Eestermans IL, Beelen RH. Milky spots in the mouse omentum may play an important role in the origin of peritoneal macrophages. Res Immunol. 1992;143:401–409[↩]
- Shimotsuma M, Shields JW, Simpson-Morgan MW, Sakuyama A, Shirasu M, Hagiwara A, Takahashi T. Morpho-physiological function and role of omental milky spots as omentum-associated lymphoid tissue (OALT) in the peritoneal cavity. Lymphology. 1993;26:90–101[↩]
- Shimotsuma M, Kawata M, Hagiwara A, Takahashi T. Milky spots in the human greater omentum. Macroscopic and histological identification. Acta Anat ( Basel) 1989;136:211–216.[↩]
- Zhu H, Naito M, Umezu H, Moriyama H, Takatsuka H, Takahashi K, Shultz LD. Macrophage differentiation and expression of macrophage colony-stimulating factor in murine milky spots and omentum after macrophage elimination. J Leukoc Biol. 1997;61:436–444[↩][↩]
- Shimotsuma M, Takahashi T, Kawata M, Dux K. Cellular subsets of the milky spots in the human greater omentum. Cell Tissue Res. 1991;264:599–601[↩]
- Krist LF, Koenen H, Calame W, van der Harten JJ, van der Linden JC, Eestermans IL, Meyer S, Beelen RH. Ontogeny of milky spots in the human greater omentum: an immunochemical study. Anat Rec. 1997;249:399–404[↩]
- Shimotsuma M, Simpson-Morgan MW, Takahashi T, Hagiwara A. Ontogeny of milky spots in the fetal lamb omentum. Arch Histol Cytol. 1994;57:291–299[↩][↩]
- Mironov VA, Gusev SA, Baradi AF. Mesothelial stomata overlying omental milky spots: scanning electron microscopic study. Cell Tissue Res. 1979;201:327–330.[↩]
- Krist LF, Eestermans IL, Steenbergen JJ, Hoefsmit EC, Cuesta MA, Meyer S, Beelen RH. Cellular composition of milky spots in the human greater omentum: an immunochemical and ultrastructural study. Anat Rec. 1995;241:163–174[↩]
- Cranshaw ML, Leak LV. Milky spots of the omentum: a source of peritoneal cells in the normal and stimulated animal. Arch Histol Cytol. 1990;53 Suppl:165–177[↩]
- Dux K. Proliferative activity of macrophages in the greater omentum of the mouse in relation to the early postnatal development of the vascular structures. J Leukoc Biol. 1986;40:445–458.[↩][↩]
- Murakami M, Honjo T. Involvement of B-1 cells in mucosal immunity and autoimmunity. Immunol Today. 1995;16:534–539.[↩]
- Kantor AB, Herzenberg LA. Origin of murine B cell lineages. Annu Rev Immunol. 1993;11:501–538.[↩]
- Solvason N, Kearney JF. The human fetal omentum: a site of B cell generation. J Exp Med. 1992;175:397–404.[↩]
- Solvason N, Kearney JF. The human fetal omentum: a site of B cell generation. J Exp Med. 1992;175:397–404[↩]
- Chung-Welch N, Patton WF, Shepro D, Cambria RP. Two-stage isolation procedure for obtaining homogenous populations of microvascular endothelial and mesothelial cells from human omentum. Microvasc Res. 1997;54:121–134[↩][↩]
- Chung-Welch N, Patton WF, Shepro D, Cambria RP. Human omental microvascular endothelial and mesothelial cells: characterization of two distinct mesodermally derived epithelial cells. Microvasc Res. 1997;54:108–120.[↩]
- Shipley PG, Cunningham RS. Studies on the absorption from serous cavities: 1. The omentum as a factor in absorption from the peritoneal cavity. Am J Physiol. 1916;40:75–81.[↩]
- Fukatsu K, Saito H, Han I, Yasuhara H, Lin MT, Inoue T, Furukawa S, Inaba T, Hashiguchi Y, Matsuda T, et al. The greater omentum is the primary site of neutrophil exudation in peritonitis. J Am Coll Surg. 1996;183:450–456[↩]
- Shimotsuma M, Simpson-Morgan MW, Takahashi T, Hagiwara A. Activation of omental milky spots and milky spot macrophages by intraperitoneal administration of a streptococcal preparation, OK-432. Cancer Res. 1992;52:5400–5402[↩]
- Van Vugt E, Van Rijthoven EA, Kamperdijk EW, Beelen RH. Omental milky spots in the local immune response in the peritoneal cavity of rats. Anat Rec. 1996;244:235–245[↩][↩]
- Doherty NS, Griffiths RJ, Hakkinen JP, Scampoli DN, Milici AJ. Post-capillary venules in the “milky spots” of the greater omentum are the major site of plasma protein and leukocyte extravasation in rodent models of peritonitis. Inflamm Res. 1995;44:169–177[↩][↩][↩]
- Konturek SJ, Brzozowski T, Majka I, Pawlik W, Stachura J. Omentum and basic fibroblast growth factor in healing of chronic gastric ulcerations in rats. Dig Dis Sci. 1994;39:1064–1071[↩]
- Hall JC, Heel KA, Papadimitriou JM, Platell C. The pathobiology of peritonitis. Gastroenterology. 1998;114:185–196[↩]
- Agalar F, Sayek I, Cakmakçi M, Hasçelik G, Abbasoglu O. Effect of omentectomy on peritoneal defence mechanisms in rats. Eur J Surg. 1997;163:605–609[↩][↩]
- Ambroze WL, Wolff BG, Kelly KA, Beart RW, Dozois RR, Ilstrup DM. Let sleeping dogs lie: role of the omentum in the ileal pouch-anal anastomosis procedure. Dis Colon Rectum. 1991;34:563–565.[↩][↩]
- Cartier R, Brunette I, Hashimoto K, Bourne WM, Schaff HV. Angiogenic factor: a possible mechanism for neovascularization produced by omental pedicles. J Thorac Cardiovasc Surg. 1990;99:264–268[↩]
- Bikfalvi A, Alterio J, Inyang AL, Dupuy E, Laurent M, Hartmann MP, Vigny L, Raulais D, Courtois Y, Tobelem G. Basic fibroblast growth factor expression in human omental microvascular endothelial cells and the effect of phorbol ester. J Cell Physiol. 1990;144:151–158[↩]
- Williams JK, Carlson GW, Austin GE, Austin ED, Rand RP, Jurkiewicz MJ. Short gut syndrome: treatment by neovascularization of the small intestine. Ann Plast Surg. 1996;37:84–9; discussion 89-90.[↩]
- Weber T, Hanisch E, Baum RP, Seufert RM. Late results of heterotopic autotransplantation of splenic tissue into the greater omentum. World J Surg. 1998;22:883–889[↩]
- Li J, Xu P, Chen H, Yang Z, Zhang Q. Improvement of tracheal autograft survival with transplantation into the greater omentum. Ann Thorac Surg. 1995;60:1592–1596.[↩]
- Agrama HM, Blackwood JM, Brown CS, Machiedo GW, Rush BF. Functional longevity of intraperioneal drains: an experimental evaluation. Am J Surg. 1976;132:418–421.[↩]
- Reissman P, Lyass S, Shiloni E, Rivkind A, Berlatzky Y. Placement of a peritoneal dialysis catheter with routine omentectomy–does it prevent obstruction of the catheter. Eur J Surg. 1998;164:703–707.[↩]
- Selgas R, Muñoz J, Miranda B, Ramos P, Caparros G, Revuelta KL, Gonzalez A, Gallar P, Sanchez Sicilia L. Induced changes of the peritoneal diffusion capacity by smoking, intra abdominal hypertension and omentectomy. Adv Perit Dial. 1989;5:24–27[↩]
- Pumford N, Cassey J, Uttley WS. Omentectomy with peritoneal catheter placement in acute renal failure. Nephron. 1994;68:327–328.[↩]
- Lewis M, Webb N, Smith T, Roberts D. Routine omentectomy is not required in children undergoing chronic peritoneal dialysis. Adv Perit Dial. 1995;11:293–295.[↩]
- Tsujimoto H, Hagiwara A, Shimotsuma M, Sakakura C, Osaki K, Sasaki S, Ohyama T, Ohgaki M, Imanishi T, Yamazaki J, et al. Role of milky spots as selective implantation sites for malignant cells in peritoneal dissemination in mice. J Cancer Res Clin Oncol. 1996;122:590–595[↩]
- Lawrance RJ, Loizidou M, Cooper AJ, Alexander P, Taylor I. Importance of the omentum in the development of intra-abdominal metastases. Br J Surg. 1991;78:117–119.[↩]
- di Re E, Grosso G, Raspagliesi F, Baiocchi G. Fallopian tube cancer: incidence and role of lymphatic spread. Gynecol Oncol. 1996;62:199–202.[↩]
- Kigawa J, Minagawa Y, Itamochi H, Kanamori Y, Ishihara H, Terakawa N. Retroperitoneal lymphadenectomy, including the para-aortic nodes in patients with stage III ovarian cancer. Am J Clin Oncol. 1994;17:230–233[↩]
- Faught W, Lotocki RJ, Heywood M, Krepart GV. Early ovarian cancer: value of a negative staging laparotomy. Eur J Gynaecol Oncol. 1996;17:200–203.[↩]
- Munkarah AR, Hallum AV, Morris M, Burke TW, Levenback C, Atkinson EN, Wharton JT, Gershenson DM. Prognostic significance of residual disease in patients with stage IV epithelial ovarian cancer. Gynecol Oncol. 1997;64:13–17.[↩]
- Tropé C, Kaern J, Vergote IB, Kristensen G, Abeler V. Are borderline tumors of the ovary overtreated both surgically and systemically. A review of four prospective randomized trials including 253 patients with borderline tumors. Gynecol Oncol. 1993;51:236–243[↩]
- Averette HE, Hoskins W, Nguyen HN, Boike G, Flessa HC, Chmiel JS, Zuber K, Karnell LH, Winchester DP. National survey of ovarian carcinoma. I. A patient care evaluation study of the American College of Surgeons. Cancer. 1993;71:1629–1638[↩]
- Steinberg JJ, Demopoulos RI, Bigelow B. The evaluation of the omentum in ovarian cancer. Gynecol Oncol. 1986;24:327–330[↩]
- Santoro E, Garofalo A, Carlini M, Rinaldi G, Santoro E. Early and late results of 100 consecutive total gastrectomies for cancer. Hepatogastroenterology. 1994;41:489–496[↩]
- Hagiwara A, Sawai K, Sakakura C, Shirasu M, Ohgaki M, Yamasaki J, Togawa T, Takahashi T. Complete omentectomy and extensive lymphadenectomy with gastrectomy improves the survival of gastric cancer patients with metastases in the adjacent peritoneum. Hepatogastroenterology. 1998;45:1922–1929[↩]
- Cafiero F, Peressini A, Bertoglio S, Biscaldi E, Queirolo P, Moresco L, Mezaros P, Percivale P. Pseudomixoma peritonei: a case report. Anticancer Res. 1997;17:3901–3905[↩]