Plasmacytosis
Plasmacytosis is a rare disorder that occurs mainly in the Asian population specifically in Japanese, Chinese, Korean and Thai populations and it is characterized by a polyclonal proliferation of plasma cells and polyclonal hypergammaglobulinemia 1. Plasma cells, the final products of B‐lymphocyte differentiation, may be mildly elevated in the bone marrow in response to a variety of conditions, including infections, autoimmune diseases, hypersensitivity reactions, toxic exposure, and malignancy 2. Reactive plasmacytosis has been reported in several types of virus infections including Hepatitis A virus, Epstein-Barr virus, Dengue virus, Parvovirus B 19 3.
There are two variants of plasmacytosis 4:
- Cutaneous plasmacytosis with involvement limited to the skin, and characterized by reddish-brown plaques, mainly on the face and trunk. Cutaneous plasmacytosis typically follows a chronic, benign course, but a few cases have beenreported to progress to systemic plasmacytosis 5. The most common extracutaneous manifestations are lymphadenopathy and polyclonal hypergammaglobulinemia 6. Renal amyloidosis, interstitial pneumonia, and hepatosplenomegaly occur lessfrequently 7. Serum immunoglobulin levels greater than 5000 mg/dL and bone marrow plasma cell percentage greater than 7% portend a more aggressive clinical course 7.
- Systemic plasmacytosis is defined as involvement of two or more organ systems.
However, “systemic plasmacytosis” or “cutaneous plasmacytosis”, may not be clearly divided into 2 different entities, as there could be occult systemic involvement in patients with “cutaneous plasmacytosis” 8.
Plasmacytosis is a rare condition, but its prevalence in the Japanese population is relatively high 9. Shimizu et al. 10 reviewed 26 previously published cases with plasmacytosis and found that all but two patients were Japanese. But the precise prevalence of plasmacytosis in Japan, and other countries, remains unclear. In the Caucasian population, there has been one patient with cutaneous plasmacytosis reported from Spain 11 and a patient from Switzerland of unknown race that conforms to the description of Watanabe et al. of systemic plasmacytosis 12.
Cutaneous and systemic plasmacytosis is an exceedingly rare condition characterized by an infiltration of mature plasma cells in various organ systems, which manifests clinically in the form of red-brown macules, papules and plaques 13. Since plasmacytosis regularly occurs at extracutaneous sites, the disorder is thus currently known as ‘cutaneous and systemic plasmacytosis’ 8. Cutaneous and systemic plasmacytosis has been grouped with other nonneoplastic lymphoplasmacytic disorders that include systemic plasmacytosis and some variants of Castleman’s disease. The distinction between these entities is not well defined because many patients with cutaneous plasmacytosis have variable systemic involvement and overlapping features with others 7. The relation between cutaneous and systemic plasmacytosis and Castleman’s disease is controversial. Except for the skin lesions, cutaneous and systemic plasmacytosis and Castleman’s disease share similar clinical and histological features. The histological features of cutaneous and systemic plasmacytosis showed infiltration of mature plasma cells, similar to multicentric plasmacytic Castleman’s disease. Overproduction of IL-6 in the serum has been reported and considered an important factor in the pathogenesis of multicentric plasmacytic Castleman’s disease. IL-6 is also highly expressed in the serum of some patients with cutaneous and systemic plasmacytosis. On the basis of the similar clinical and histological features, it has been suggested that cutaneous and systemic plasmacytosis and cutaneous multicentric plasmacytic Castleman’s disease may be considered a disease continuum rather than distinct entities, which needs further studies to clarify 14.
Cutaneous and systemic plasmacytosis is an uncommon disorder with <100 patients described in the literature thus far, the majority of whom are Japanese 15. Worldwide, cutaneous and systemic plasmacytosis has only been reported in about 10 Caucasian patients 16. Cutaneous and systemic plasmacytosis is usually accompanied by fever, anemia, polyclonal hypergammaglobulinemia and superficial lymphadenopathy 17. A previous study suggested that cutaneous and systemic plasmacytosis has a male to female ratio of 1.0:0.6, and an age of onset between 20 and 62 years with a mean and median age of 37 years 18. Cutaneous and systemic plasmacytosis is usually diagnosed via biopsy; histologically, superficial and deep perivascular and perineural dermatitis with prominent plasma cells is observed 19. Overall, the prognosis of the disorder is favorable, although rare cases have been reported with a more aggressive clinical course, including the development of lymphoma 19. Treatment with immunosuppressive agents of variable potency has been described with a degree of success; however, there is not a common effective treatment 17.
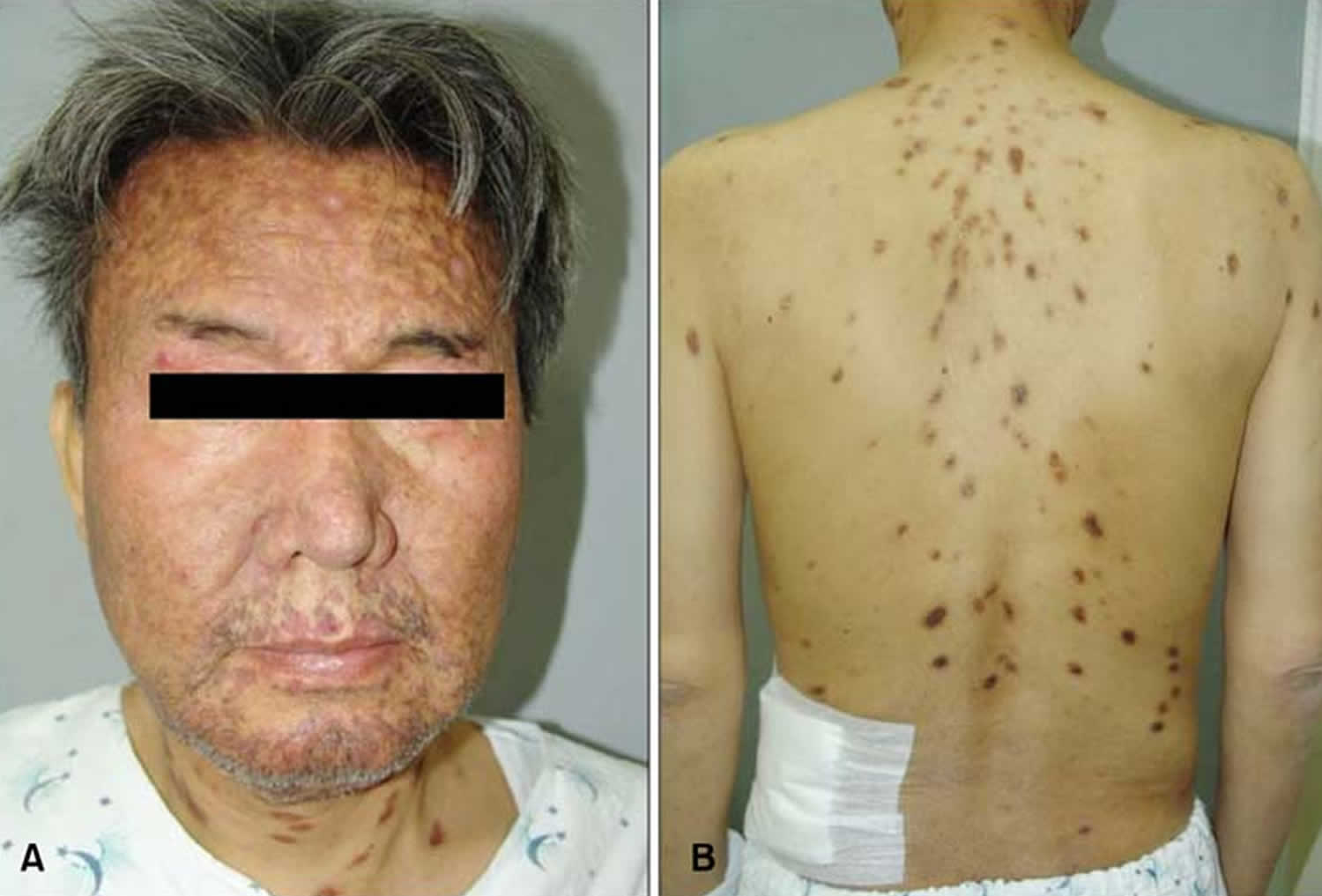
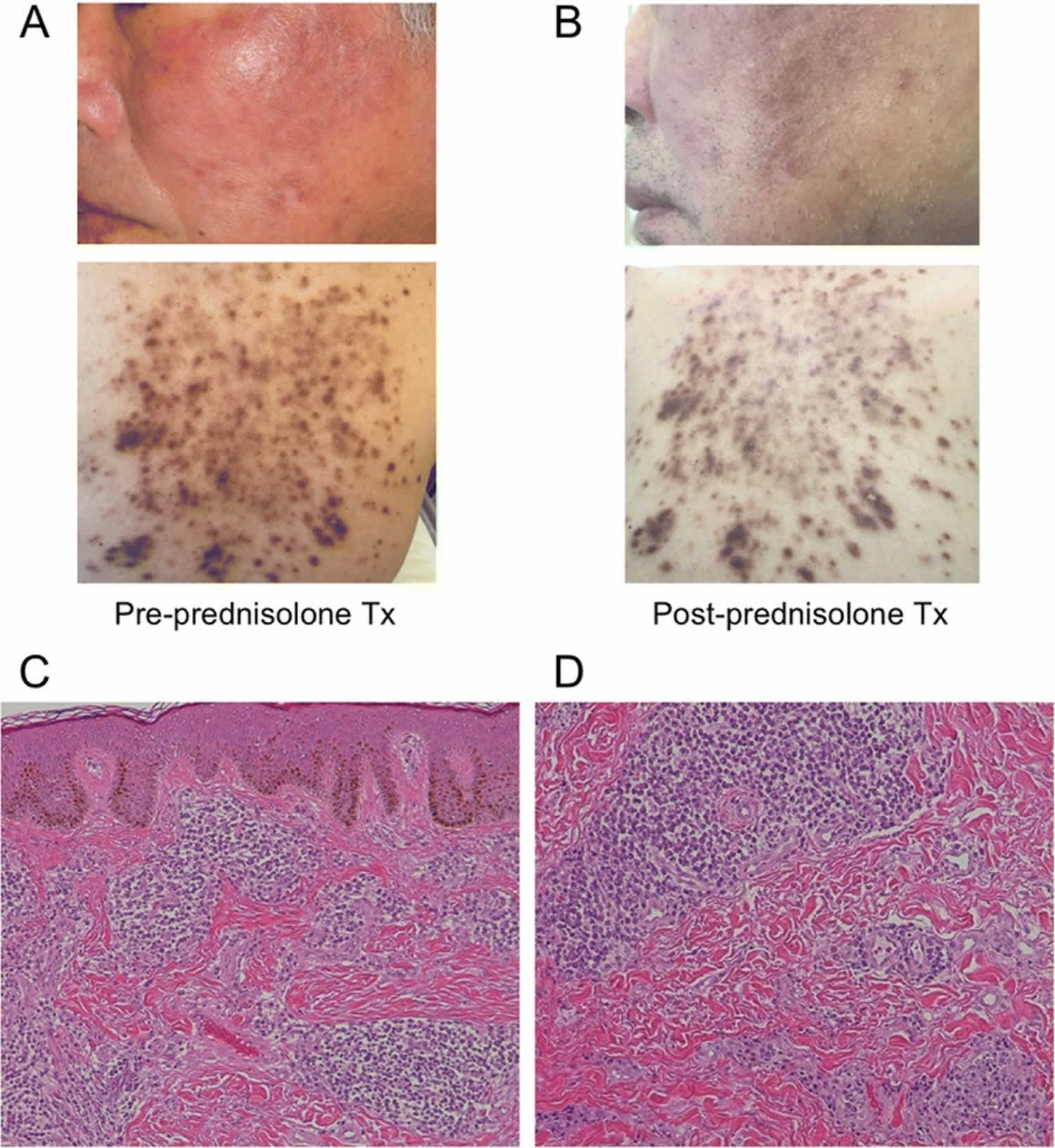
Figure 1. Cutaneous plasmacytosis
Footnotes: (a) The patient presented with red-brown plaques on his face and trunk which were ameliorated by prednisolone therapy. (b) Although the pigmentation of facial lesions remained, redness and swelling improved to some extent. (c, d) Histological examination revealed characteristic findings of cutaneous plasmacytosis. There was no abnormality in either the stratum corneum or epidermis, but plasma cells infiltrated the dermis. Serum and dermal pathological analyses revealed elevated levels of IgG and IgA, without Bence Jones or M protein, and proliferation of mature plasma cells in the dermis. These findings suggested plasmacyotosis rather than multiple myeloma.
Abbreviation: Tx = treatment
[Source 20 ]Plasmacytosis causes
The cause of plasmacytosis is poorly understood and the pathogenesis of plasmacytosis is unknown. It has been proposed that plasmacytosis is a consequence of overreaction to unknown stimuli, which include malignancies 8. Multiple theories have been proposed, including the elevation of cytokine interleukin 6 (IL-6) 21. The variability of humans geographically or IL-6 genetic polymorphisms may possibly explain the frequency of cutaneous and systemic plasmacytosis in individual regions. IL-6 promotes antibody production and drives the differentiation of mature B cells into plasma cells. Importantly, overexpression of IL-6 in mice results in a plasmacytosis 22. Accordingly, serum elevations of IL-6 have been reported in patients with cutaneousand systemic plasmacytosis 23. It is plausible that some type of insult, such as infection, autoimmune dysregulation or genetic mutation, induces a localized or systemic increase in IL-6 that results in plasmacytosis. Furthermore, Haque et al 24 hypothesized that due to the local production of IL-6, long-lived plasma cells originate and survive in the environment of the skin, and that such patients may therefore respond to agents able to interfere with the activity of IL-6.
Plasmacytosis symptoms
The majority of patients with plasmacytosis are asymptomatic, although individuals with systemic involvement may present with constitutional symptoms, including weight loss and fatigue 15. The most common extracutaneous involvement is lymphadenopathy, which occurs in approximately 58% of patients. Lung involvement presents as lymphoid interstitial pneumonia. Liver, spleen, and kidney involvement occur less frequently 25. In the Korean literature, there has been no case of cutaneous and systemic plasmacytosis with involvement of such a variety of organs. Renal involvement in cutaneous and systemic plasmacytosis is highly heterogeneous and may be coincidental in many cases. All cases of renal amyloidosis have been known as secondary amyloidosis 26. Amyloidosis may progress rapidly in cases with high serum amino acids and C-reactive protein. IL-6 may also play a role in amyloid deposition. The overproduction of IL-6 in the serum considered an important factor in the pathogenesis of cutaneous and systemic plasmacytosis, similarly to multicentric plasmacytic Castleman’s disease. It is possible to postulate that a relation exists between IL-6 and amyloid production and deposition in cutaneous and systemic plasmacytosis.
The typical skin lesions are asymptomatic, brownish, irregularly-shaped macules or plaques, which are mostly disseminated on the trunk. The lesions also can additionally involve the face or limbs. Aside from the compulsory cutaneous manifestation, polyclonal hypergammaglobulinemia and lymphadenopathy are the most frequent symptoms of the disease 27.
Plasmacytosis diagnosis
Plasmacytosis should be differentiated from other conditions resulting in the proliferation of plasma cells, including chronic infection, collagen vascular disease, cutaneous marginal B-cell lymphoma, and B-cell pseudolymphoma.
In cutaneous plasmacytosis, histopathological examination shows perivascular and perineural plasma cell infiltrates in the dermis without evidence of folliculitis or other common causes of plasma cell infiltrates. It is also important to demonstrate that the plasma cells are polyclonal. This can be done with kappa and lambda immunohistochemistry or in situ hybridisation. Plasma cells usually revealed a polyclonality expression of both kappa and gamma immunoglobulin light chains 28. Although the plasma cells are usually polyclonal in cutaneous and systemic plasmacytosis, some cases may indeed harbor monoclonal population of plasma cells 29.
Plasmacytosis treatment
Given the rarity of cutaneous and systemic plasmacytosis, no controlled therapeutic trials have thus far been conducted and a commonly effective treatment has not been reported. Generally, the therapy for cutaneous and systemic plasmacytosis has been quite disappointing. For cutaneous plasmacytosis, atrial of topical and systemic corticosteroids, tacrolimus, psoralen ultraviolet A (PUVA), photodynamic therapy and pulsed dye laser are reported with varying success 30. Studies have shown that the most commonly used topical or intralesional therapies performed with corticosteroids result in no, slight or an only transient improvement 31. Topical 0.1% tacrolimus ointment has been shown to help reduce the thickness and pigmentation of skin lesions 32. Tacrolimus and pimecrolimus have been applied as treatment with different effects. Ma et al 33 and Miura et al 34 reported that there was no or only slight benefit when treated with tacrolimus, whereas Hafner et al 35 recorded effective treatment with pimecrolimus in one case. Other available topical treatments for cutaneous plasmacytosis include radiotherapy, photodynamic therapy and psoralen combined with ultraviolet A 36. Photodynamic therapy decreased the thickness of lesions, whereas long-pulse ruby laser helped lighten the pigmentation 32. In individual cases, patient have been treated with cyclophosphamide, vincristine, prednisone and rituximab chemotherapy with only partial, transient resolution of the lesions 37. Lee et al 38 showed that systemic plasmacytosis was improved with melphalan treatment. A recent report described treatment success with a combination of prednisolone and cyclophosphamide. Radiotherapy, phototherapy, and intralesional corticosteroids have been shown to improve cutaneous lesions 25. However, the poor treatment response of this disorder clearly demonstrates the needs for a novel therapeutic method.
Systemic steroids 39, cyclophosphamide 40 and other chemotherapies 41 have been reported to be effective to a certain extent in systemic plasmacytosis.
Thalidomide stimulates the cytotoxic functions of T lymphocytes, thus limiting the immunosuppressive function of regulatory T cells and significantly altering the immunological profile by inhibiting the release of TNFα and IL-6 42. Fang et al 13 hypothesized that through decreased secretion of IL-6, thalidomide may affect the growth of plasma cells, which could be beneficial for the treatment of cutaneous and systemic plasmacytosis. Thalidomide treatment was well tolerated in the present patient 43. The main adverse effects of thalidomide are sedation, fatigue, constipation, peripheral neuropathy and thromboembolic phenomena.
Plasmacytosis prognosis
Most cases of plasmacytosis have followed a chronic and benign clinical course without spontaneous remission 28. The prognosis of systemic plasmacytosis is less favorable than that of cutaneous plasmacytosis, due to systemic complications such as renal failure, respiratory failure, leukemia and malignant lymphoma 8. Therefore, special attention should be given to monitoring for pulmonary and renal involvement.
Although there are reports of patients with plasmacytosis developing malignant lymphoma such as T-cell lymphoma and non-Hodgkin lymphoma, the relation between plasmacytosis and lymphoma is not clear. However, there is a low rate of malignant transformation of cutaneous and systemic plasmacytosis in Asian studies. A single case of T-cell lymphoma arising in a patient with cutaneous and systemic plasmacytosis has been reported; however, whether this was a coincidental finding is unclear 28.
- Systemic plasmacytosis with cutaneous manifestations in a white man: Successful therapy with cyclophosphamide/prednisone. JAAD Volume 38, ISSUE 4, P629-631, April 01, 1998 https://doi.org/10.1016/S0190-9622(98)70131-8[↩]
- Pellat-Deceunynck C, Jego G, Robillard N, Accard F, Amiot M, Bataille R. Reactive plasmacytoses, a model for studying the biology of human plasma cell progenitors and precursors. Hematol J. 2000;1:362–366. doi: 10.1038/sj.thj.6200053[↩]
- Desborough MJ, Grech H. Epstein-Barr virus-driven bone marrow aplasia and plasmacytosis mimicking a plasma cell neoplasm. Br J Haematol. 2014;165:272. doi: 10.1111/bjh.12721[↩]
- Watanabe S, Ohara K, Kukita A, Shigeo M. Systemic plasmacytosis. A syndrome of peculiar multiple skin eruptions, generalized lymphadenopathy, and polyclonal hypergammaglobulinemia. Arch Dermatol. 1986;122:1314–1320. doi: 10.1001/archderm.1986.01660230106022[↩]
- Dautriche, Cula & Driessche, Freya & Chen, Lu & Brinster, Nooshin & Marmon, Shoshana. (2019). Ulcerative cutaneous plasmacytosis. JAAD Case Reports. 5. 540-542. 10.1016/j.jdcr.2019.04.004[↩]
- Wagner, G., Rose, C., Klapper, W. and Sachse, M.M. (2013), Cutaneous and systemic plasmocytosis. JDDG: Journal der Deutschen Dermatologischen Gesellschaft, 11: 1161-1167. https://doi.org/10.1111/ddg.12190[↩]
- Lee TG, Jeong WS, Moon SH, Hwangbo H, Lee SK, Lee DR, Kwon TG. Cutaneous and Systemic Plasmacytosis Associated with Renal Amyloidosis. Ann Dermatol. 2015 Dec;27(6):759-762. https://doi.org/10.5021/ad.2015.27.6.759[↩][↩][↩]
- Tada Y, Komine M, Suzuki S, Kikuchi K, Sasaki M, Kaneko N, Tamaki K. Plasmacytosis: systemic or cutaneous, are they distinct? Acta Derm Venereol. 2000 May;80(3):233-5. doi: 10.1080/000155500750043203 https://www.medicaljournals.se/acta/download/10.1080/000155500750043203[↩][↩][↩][↩]
- Kiniwa Y, Matoba H, Hamano H, Sendo Y, Okuyama R. Cutaneous plasmacytosis associated with autoimmune hemolytic anemia. J Dermatol. 2016;43:343–345. doi: 10.1111/1346-8138.13194[↩]
- Shimizu S, Tanaka M, Shimizu H, Han-yaku H. Is cutaneous plasmacytosis a distinct clinical entity? J Am Acad Dermatol. 1997;36:876–880. doi: 10.1016/S0190-9622(97)70046-X[↩]
- López-Estebaranz JL, Rodriguez-Peralto JL, Ortiz Romero PL, Vanaclocha F, Iglesias Díez L. Cutaneous plasmacytosis: report of a case in a white man. J Am Acad Dermatol. 1994 Nov;31(5 Pt 2):897-900. doi: 10.1016/s0190-9622(94)70255-1[↩]
- Dubouloz M, Nydegger U, Cruchaud A, Harms M, Fabre J. Plasmocytose cutanée et cryo-immunoglobulinémie polyclonale [Cutaneous plasmacytosis and polyclonal cryo-immunoglobulinemia]. Schweiz Med Wochenschr. 1978 Jan 7;108(1):34-9. French.[↩]
- Fang, S., Shan, K., & Chen, A. (2016). Cutaneous and systemic plasmacytosis on the face: Effective treatment of a case using thalidomide. Oncology Letters, 11, 1923-1925. https://doi.org/10.3892/ol.2016.4140[↩][↩]
- Paydas S. Report on 59 patients with renal amyloidosis. Int Urol Nephrol 1999;31:619–631.[↩]
- Honda R, Cerroni L, Tanikawa A, Ebihara T, Amagai M, Ishiko A. Cutaneous plasmacytosis: report of 6 cases with or without systemic involvement. J Am Acad Dermatol. 2013 Jun;68(6):978-85. doi: 10.1016/j.jaad.2012.11.031[↩][↩]
- González-López MA, González-Vela MC, Blanco R, Fernández-Llaca H, Val-Bernal JF. Cutaneous plasmacytosis limited to the extremities in a white patient: an unusual clinical picture. Cutis. 2010 Sep;86(3):143-7.[↩]
- Carey WP, Rico MJ, Nierodzik M, Sidhu G. Systemic plasmacytosis with cutaneous manifestations in a white man: successful therapy with cyclophosphamide/prednisone. J Am Acad Dermatol. 1998 Apr;38(4):629-31. doi: 10.1016/s0190-9622(98)70131-8[↩][↩]
- Leonard AL, Meehan SA, Ramsey D, Brown L, Sen F. Cutaneous and systemic plasmacytosis. J Am Acad Dermatol. 2007 Feb;56(2 Suppl):S38-40. doi: 10.1016/j.jaad.2006.05.019[↩]
- Jayaraman AG, Cesca C, Kohler S. Cutaneous plasmacytosis: A report of five cases with immunohistochemical evaluation for HHV-8 expression. Am J Dermatopathol. 2006 Apr;28(2):93-8. doi: 10.1097/01.dad.0000181107.08791.87[↩][↩]
- Hatano T, Takanashi M, Tsuchihashi H, et al. Myalgia caused by chronic myositis associated with plasmacytosis: a case report. BMC Neurol. 2018;18(1):112. Published 2018 Aug 14. doi:10.1186/s12883-018-1123-3 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6090622[↩]
- Yamamoto T, Katayama I, Nishioka K. Increased plasma interleukin-6 in cutaneous plasmacytoma: the effect of intralesional steroid therapy. Br J Dermatol. 1997 Oct;137(4):631-6. doi: 10.1111/j.1365-2133.1997.tb03801.x[↩]
- [↩]
- IgG1 plasmacytosis in interleukin 6 transgenic mice. Proceedings of the National Academy of Sciences Oct 1989, 86 (19) 7547-7551; DOI: 10.1073/pnas.86.19.7547[↩]
- Haque M, Hou JS, Hisamichi K, Tamada K, Cusack CA, Abdelmalek M, Brown RE, Vonderheid EC. Cutaneous and systemic plasmacytosis vs. cutaneous plasmacytic castleman disease: review and speculations about pathogenesis. Clin Lymphoma Myeloma Leuk. 2011 Dec;11(6):453-61. doi: 10.1016/j.clml.2011.07.004[↩]
- Ma HJ, Liu W, Li Y, Zhao G, Meng RS, Li DG. Cutaneous and systemic plasmacytosis: a Chinese case. J Dermatol 2008;35:536–540.[↩][↩]
- Chen H, Xue Y, Jiang Y, Zeng X, Sun JF. Cutaneous and systemic plasmacytosis showing histopathologic features as mixed-type Castleman disease: a case report. Am J Dermatopathol 2012;34:553–556.[↩]
- Wagner G, Rose C, Klapper W, Sachse MM. Cutaneous and systemic plasmocytosis. J Dtsch Dermatol Ges. 2013 Dec;11(12):1161-7. doi: 10.1111/ddg.12190[↩]
- Amin HM, McLaughlin P, Rutherford CJ, Abruzzo LV, Jones D. Cutaneous and systemic plasmacytosis in a patient of Asian descent living in the United States. Am J Dermatopathol 2002;24:241–245.[↩][↩][↩]
- Honda R, Cerroni L, Tanikawa A, Ebihara T, Amagai M, Ishiko A. Cutaneous plasmacytosis: report of 6 cases with or without systemic involvement. J Am Acad Dermatol 2013;68:978–985.[↩]
- Gilliam AC, Mullen RH, Oviedo G, et al. Isolated benign primarycutaneous plasmacytosis in children: two illustrative cases.Arch Dermatol. 2009;145(3):299-302.[↩]
- Ahn JJ, Yang YS, Shin MK, Lee SW, Kim NI. Case of isolated benign primary cutaneous plasmacytosis in a child. J Dermatol. 2011 Apr;38(4):364-7. doi: 10.1111/j.1346-8138.2010.01111.x[↩]
- Lee JS, Chiam L, Tan KB, Salto-Tellez M, Tan SH, Ong BH, et al. Extensive hyperpigmented plaques in a chinese singaporean woman: a case of cutaneous plasmacytosis. Am J Dermatopathol 2011;33:498–503.[↩][↩]
- Ma HJ, Liu W, Li Y, Zhao G, Meng RS, Li DG. Cutaneous and systemic plasmacytosis: a Chinese case. J Dermatol. 2008 Aug;35(8):536-40. doi: 10.1111/j.1346-8138.2008.00516.x[↩]
- Miura H, Itami S, Yoshikawa K. Treatment of facial lesion of cutaneous plasmacytosis with tacrolimus ointment. J Am Acad Dermatol. 2003 Dec;49(6):1195-6. doi: 10.1016/s0190-9622(03)00860-0[↩]
- Hafner C, Hohenleutner U, Babilas P, Landthaler M, Vogt T. Targeting T cells to hit B cells: successful treatment of cutaneous plasmacytosis with topical pimecrolimus. Dermatology. 2006;213(2):163-5. doi: 10.1159/000093860[↩]
- Tzung TY, Wu KH, Wu JC, Tseng HH. Primary cutaneous plasmacytosis successfully treated with topical photodynamic therapy. Acta Derm Venereol. 2005;85(6):542-3. doi: 10.1080/00015550510027810[↩]
- Amin HM, McLaughlin P, Rutherford CJ, Abruzzo LV, Jones D. Cutaneous and systemic plasmacytosis in a patient of Asian descent living in the United States. Am J Dermatopathol. 2002 Jun;24(3):241-5. doi: 10.1097/00000372-200206000-00009[↩]
- Lee DW, Choi SW, Park JW, Cho BK. Systemic plasmacytosis: a case which improved with melphalan. J Dermatol. 1995 Mar;22(3):205-9. doi: 10.1111/j.1346-8138.1995.tb03372.x[↩]
- Yamamoto T, Katayama I, Nishioka K. Increased plasmainterleukin-6 in cutaneous plasmacytoma: the effect of intrale-sional steroid therapy. Br J Dermatol 1997; 137: 631-636.[↩]
- Carey WP, Rico MJ, Nierodzik M, Sidhu G. Systemicplasmacytosis with cutaneous manifestations in a white man:Successful therapy with cyclophosphamide/prednisone. J AmAcad Dermatol 2000; 38: 629-631.[↩]
- Lee DW, Choi SW, Park JW, Cho BK. Systemic plasmacytosis:a case which improved with melphalan. J Dermatol 1995; 22:205-209.[↩]
- Semeraro M, Vacchelli E, Eggermont A, Galon J, Zitvogel L, Kroemer G, Galluzzi L. Trial Watch: Lenalidomide-based immunochemotherapy. Oncoimmunology. 2013 Nov 1;2(11):e26494. doi: 10.4161/onci.26494[↩]
- Berrebi A, Feldberg E, Spivak I, Shvidel L. Mini-dose of thalidomide for treatment of primary myelofibrosis. Report of a case with complete reversal of bone marrow fibrosis and splenomegaly. Haematologica. 2007 Feb;92(2):e15-6. doi: 10.3324/haematol.10684[↩]