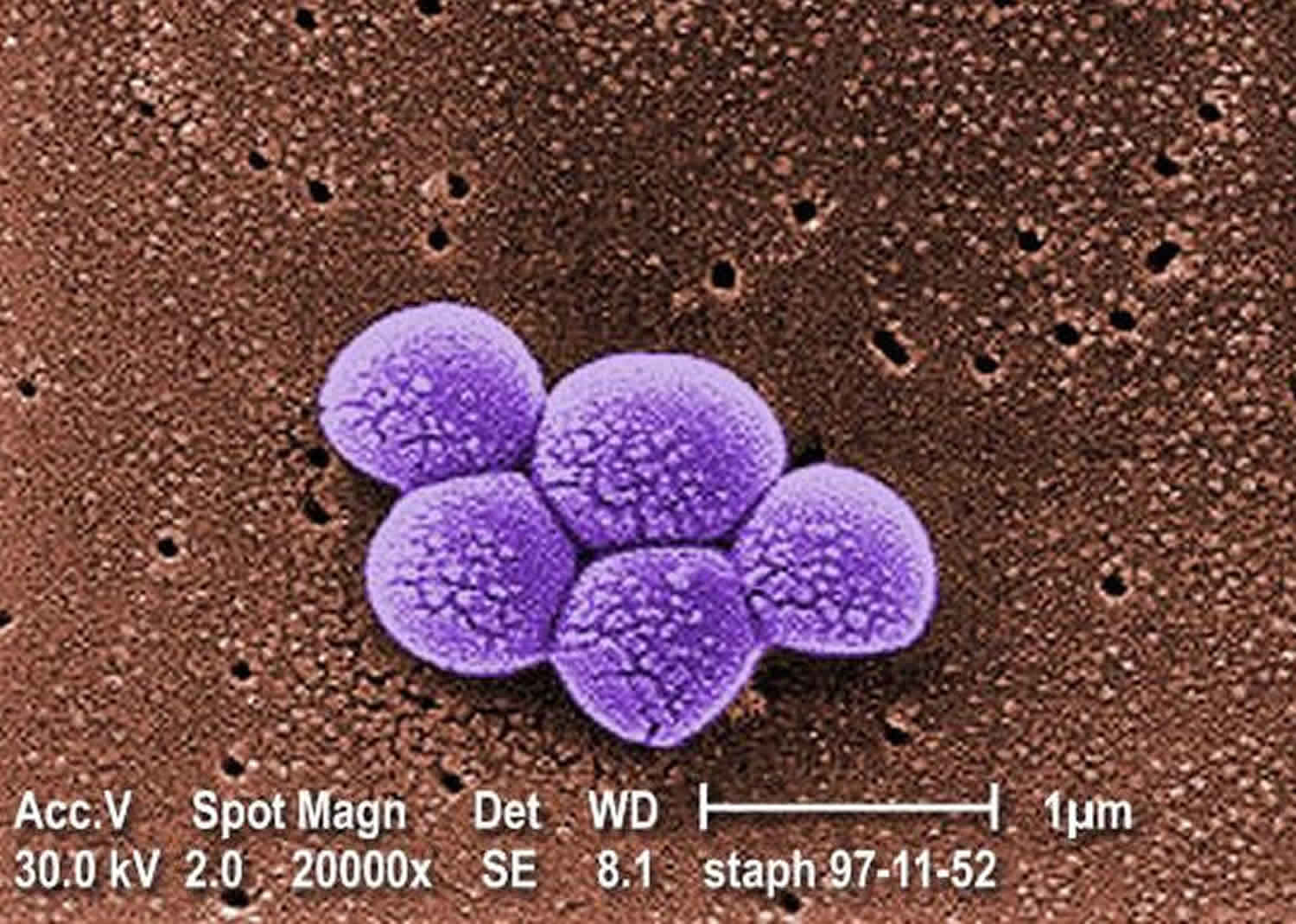
Staphylococcus aureus
Staphylococcus aureus is Gram-positive bacteria (stain purple by Gram stain) that are cocci-shaped and tend to be arranged in clusters that are described as “grape-like” 1. Staphylococcus aureus (staph) is both a commensal bacterium and a human pathogen. Approximately 30% of the human population is colonized with Staphylococcus aureus on the skin and mucous membranes (some people carry Staphylococcus aureus in their noses) 2. Staphylococcus aureus does not normally cause infection on healthy skin; however, if it is allowed to enter the bloodstream or internal tissues, these bacteria may cause a variety of potentially serious infections 3. Transmission is typically from direct contact. However, some infections involve other transmission methods 4. Most of the time, Staphylococcus aureus does not cause any harm; however, sometimes Staphylococcus aureus causes infections. Staphylococcus aureus is also a major human pathogen that causes a wide range of clinical infections. Staphylococcus aureus is a leading cause of bacteremia and infective endocarditis as well as osteoarticular, skin and soft tissue, pleuropulmonary, and device-related infections.
In healthcare settings, these Staphylococcus aureus infections can be serious or fatal, including:
- Bacteremia or sepsis when bacteria spread to the bloodstream.
- Pneumonia, which most often affects people with underlying lung disease including those on mechanical ventilators.
- Infective endocarditis (infection of the heart valves), which can lead to heart failure or stroke.
- Osteomyelitis (bone infection), which can be caused by staph bacteria traveling in the bloodstream or put there by direct contact such as following trauma (puncture wound of foot or intravenous (IV) drug abuse).
Both community-associated and hospital-acquired infections with Staphylococcus aureus have increased in the past 20 years, and the rise in incidence has been accompanied by a rise in antibiotic-resistant strains—in particular, methicillin-resistant Staphylococcus aureus (MRSA) and, more recently, vancomycin-resistant Staphylococcus aureus (VRSA).
Staph aureus infections are caused by several different types of Staphylococcus aureus bacteria, including:
- Methicillin-resistant Staphylococcus aureus (MRSA)
- Methicillin-susceptible Staphylococcus aureus (MSSA)
- Vancomycin-intermediate Staphylococcus aureus (VISA)
- Vancomycin-resistant Staphylococcus aureus (VRSA)
Populations at risk for Staphylococcus aureus infection
Anyone can develop a Staphylococcus aureus infection, although certain groups of people are at greater risk, including people with chronic conditions such as diabetes, cancer, vascular disease, eczema, lung disease, and people who inject drugs. In healthcare facilities, the risk of more serious staph aureus infection is higher because many patients have weakened immune systems or have undergone procedures. In healthcare, the risk of more serious staph infection is higher for patients in intensive care units (ICUs), patients who have undergone certain types of surgeries and patients with medical devices inserted in their bodies.
What puts people at risk for serious Staphylococcus aureus infection
In communities
- Uncovered or draining wounds, especially in high-contact sports or crowded living
- Sharing personal items, such as towels or razors
- Recent stay in a healthcare facility
- Injection drug use like opiods
In hospitals
- Hospital stays or surgery (during and shortly after)
- Exposure to patients carrying or infected with Staphylococcus aureus
- Medical devices in the body, like intravenous (IV) lines
In other healthcare facilities
- Outpatient surgery and procedures like dialysis
- Nursing home stays
- Medical devices in the body, like intravenous (IV) lines
Staphylococcus aureus causes
Skin and soft tissue (impetigo)
Often occurring in young children, impetigo is spread within families and through close physical contact. Impetigo is more prevalent in warm, humid climates because of more opportunities for insect bites and cutaneous trauma. Impetigo may also be a complication of varicella. Diagnosis is usually made based on the characteristic appearance of the lesions. Bullous impetigo may also occur in endemic and epidemic patterns. Nursery outbreaks have been described, and some cases in infants have progressed to scalded skin syndrome or Ritter disease.
Folliculitis, furuncle, and carbuncle
Recurrent staphylococcal skin infections develop in certain patients, such as those with impaired neutrophil function (eg, those with chronic granulomatous disease), patients with atopy and chronic eczema, and those with impaired circulation and diabetes mellitus. However, most patients with recurrent furunculosis are colonized with community-associated MRSA but are otherwise healthy. Thus, an evaluation of the immune system in these individuals is seldom useful.
Bone and joint infections (osteomyelitis)
Osteomyelitis typically occurs in children prior to the age of epiphyseal closure. Osteomyelitis typically originates in the metaphysis of long bones in the region of most rapid growth. Osteomyelitis usually involves (in order of frequency) the lower end of the femur, the upper end of the tibia and humerus, and the radius. Most bone and joint infections result from hematogenous spread, but significant blunt trauma is a preceding event in approximately one third of cases. In addition, penetrating wounds, compound fractures, and orthopedic appliances may introduce microbial infection directly into bone. Notably, the male-to-female ratio of skeletal infections is 2:1, mostly because boys are more likely to experience traumatic events.
Septic arthritis
Staphylococci are frequent etiologic agents of septic arthritis and, since the era of successful vaccination for Haemophilus influenzae, are now more predominant in younger age groups. The USA300 genotype is the most common clone in staphylococcal septic arthritis in children 5. Bacteria can enter the joint space through hematogenous spread, direct inoculation, or contiguous spread of infection. Because the synovial membrane has a high effective blood flow, a large number of bacteria may be delivered to the joint during a period of bacteremia. Inoculation can occur when a joint is punctured with a contaminated object, and many clinical studies reveal that the knee is more likely to be punctured. In the postantibiotic era, contiguous spread has been rare, with the exception of neonatal osteomyelitis.
Endocarditis
Fortunately, Staphylococcus aureus endocarditis is rare in pediatric patients. It often involves adolescent intravenous drug users who do not have antecedent valvular disease. These patients usually present with right-sided disease with evidence of pulmonary disease, such as pulmonary abscesses or shifting infiltrates. In children with preexisting heart disease, endocarditis is often temporally related to cardiac surgery or catheterization. Children with prosthetic valves are particularly vulnerable because of the organism’s propensity to adhere to foreign materials. In addition, patients with indwelling vascular access devices are at risk, because infections from the skin can seed the catheter, resulting in catheter-related blood stream infection and its consequences 6.
Toxic shock syndrome
Infection with toxin-producing Staphylococcus aureus in the absence of protective antibody is an antecedent. Younger patients may be at increased risk because they lack the protective antibodies to the enterotoxins and other exotoxins responsible for producing this clinical syndrome. However, other factors may be involved; Jacobsen et al demonstrated in a small study that not all patients without antibody develop true toxic shock syndrome when infected with a toxin-producing strain of the organism 7.
The erythroderma of toxic shock syndrome depends on preexisting T-cell hypersensitivity and the toxin superantigen for amplification 8.
Approximately 25% of all Staphylococcus aureus strains are toxigenic, and, at any time, roughly 4-10% of healthy individuals are colonized with these strains. In the 1980s, the disease was associated with the use of highly absorbent tampons in women during menstruation. Currently, many cases observed are nonmenstrual (eg, localized infections, surgery, infected varicella lesions, insect bites), and these now account for one third of all cases. These patients carry a higher mortality rate than those with menstrual toxic shock syndrome.
Pneumonia
The primary form occurs without an extrapulmonary focus, presumably through direct inoculation to the lungs, and the secondary form results from hematogenous seeding of the lungs during endocarditis or bacteremia. Predisposing factors include infancy, chronic illness, and viral respiratory disease such as influenza. Patients with head injury and trauma who have nasopharyngeal carriage of S aureus are at increased risk of Staphylococcus aureus pneumonia.
Thrombophlebitis
The causes are associated with infusion, including infected intravenous catheters and needles. The common point of entry for infection related to intravascular devices is the insertion site along the outside of the device.
Deep tissue abscess and infection
These typically result from hematogenous seeding, although myositis or pyomyositis can result from contiguous spread of infection and endophthalmitis can follow trauma (injury or iatrogenic), for example.
Staphylococcus aureus prevention
In a 5-year multicenter study, the introduction of practices to eliminate Staphylococcus aureus significantly reduced the rate of complex Staphylococcus aureus infection at surgical sites, from 0.36% to 0.20%. The study included 38,049 patients who underwent 42,534 operations (cardiac surgery or hip or knee arthroplasty); 28,218 operations were performed before the intervention was implemented and 14,316 were performed during the intervention period 9.
For the intervention, patients were screened for Staphylococcus aureus in the month before surgery. Those with positive screens were asked to apply intranasal mupirocin and to bathe with chlorhexidine for 5 days before surgery. Patients with methicillin-sensitive Staphylococcus aureus received perioperative prophylactic cefazolin, and those with methicillin-resistant Staphylococcus aureus (MRSA) received cefazolin and vancomycin. Patients who were negative for Staphylococcus aureus bathed with chlorhexidine the night before and the morning of surgery, and received cefazolin 10.
A population-based study by Smit et al reported that current statin users had a 27% lower risk of community-acquired Staphylococcus aureus bacteremia and that long-term use was particularly associated with lower risk 11.
Staphylococcus aureus symptoms
Types and presentation of Staphylococcus aureus infection include the following 12:
- Skin and soft tissue (impetigo): A small area of erythema that progresses into bullae (filled with cloudy fluid) that rupture and heal with the formation of a honey-colored crust
- Scalded skin syndrome (Ritter disease): A relatively rare, toxin-mediated disorder with superficial fragile blisters that burst, leaving a tender base; often accompanied by fever and occasionally by mucopurulent eye discharge
- Folliculitis: A tender pustule that involves the hair follicle
- Furuncle: Small abscesses characterized by exuding purulent material from a single opening; involves both the skin and the subcutaneous tissues in areas with hair follicles
- Carbuncle: An aggregate of connected furuncles, with several pustular openings
- Bone infections (osteomyelitis): In children, sudden onset of fever and bony tenderness or a limp; pain may be throbbing and severe; however, presentation in neonates can be subtle
- Septic arthritis: Decreased range of motion, warmth, erythema, and tenderness of the joint with constitutional symptoms and fever; however, these signs may be absent in infants (in whom the hip is the most commonly involved joint)
- Endocarditis: Initially presents as fever and malaise; peripheral emboli may be present; may involve healthy valves
- Toxic shock syndrome: Fever, diffuse macular erythema, and hypotension, with involvement of 3 or more organ systems; can be rapidly progressive in previously healthy individuals
- Pneumonia: Most common in infants, young children, and debilitated patients; a short prodrome of fever followed by rapid onset of respiratory distress; prominent gastrointestinal symptoms may also occur
- Thrombophlebitis: Fever, pain, and occasionally erythema at the insertion site of an intravenous catheter; usually affects hospitalized patients
- Deep tissue abscess and infection: Muscles 13 and organs can become infected, including the parotid gland, eyes, liver, spleen, kidneys, and central nervous system 14; deep abscesses also may occur 15; fever with or without localizing pain is typical.
Staphylococcus aureus diagnosis
Folliculitis, furuncle, and carbuncle
- Diagnosis based on clinical appearance
- Aspiration or incision and culture of purulent material from the lesion occasionally diagnostic
Osteomyelitis
- Cultures of bone aspirate
- Blood culture results positive in only 30-50% of pediatric patients
- C-reactive protein (CRP) levels and erythrocyte sedimentation rate are generally elevated in acute disease
- Bone scan with increased technetium-99m–labeled diphosphonate uptake supports the clinical diagnosis; however, this modality is not as useful in neonates or after trauma or surgery
- Magnetic resonance imaging (MRI) scan is the best imaging modality for defining purulent collections and for planning surgery
- On plain film radiographs, destructive bone changes are usually observed 2 weeks after infection
Septic arthritis
- Gram stain and culture of joint fluid is the primary means of diagnosis
- Direct inoculation of synovial fluid into culture bottles may improve culture yield
- Median white blood cell count in joint fluid is 60.5 × 109, with neutrophil predominance >75%
- Synovial fluid glucose levels are often low
- Plain radiographs show capsular swelling
- MRI or CT scanning is the imaging method of choice for pyogenic sacroiliitis
Endocarditis
- Blood culture is the most important diagnostic procedure
- Inject the blood sample into hypertonic media if the patient has been exposed to antibiotics
- Obtain 3-5 sets of large-volume blood cultures within the first 24 hours
- Echocardiography is a valuable adjunct
Pneumonia
- Blood cultures are more likely to be positive in secondary than primary disease (90% vs 20%)
- An adequate respiratory tract specimen should be obtained prior to initiating therapy; specimens may include endotracheal sampling, pleural fluid, or lung tap
- Sputum specimens are inadequate because upper respiratory tract colonization is common
- No radiologic features are highly specific
- Typical radiographic features are unilateral consolidation in primary staphylococcal pneumonia and bilateral infiltrates in secondary cases
- Early in the disease course, the chest radiograph may reveal minimal infiltrates, but within hours, infiltrates progress rapidly
- Pleural effusion, pneumatoceles, and pneumothorax are also common
- In oncology patients, Staphylococcus aureus may cause pulmonary nodules 16
Thrombophlebitis
- Obtain a blood culture through the intravenous line and a peripheral blood culture and a peripheral blood culture is usually recommended.
Staphylococcus aureus treatment
Because community-associated methicillin-resistant Staphylococcus aureus (CA-MRSA) causes more than one half of all staphylococcal infections in most communities, empiric therapy with penicillins or cephalosporins may be inadequate 16. Some experts recommend combination therapy with a penicillinase-resistant penicillin or cephalosporin (in case the organism is methicillin-sensitive Staphylococcus aureus [MSSA]) 17 and clindamycin or a quinolone. Others suggest use of clindamycin, trimethoprim-sulfamethoxazole (TMP-SMX), rifampin, doxycycline, or a quinolone. Finally, because of concerns about induction of resistance, some recommend using TMP-SMX and rifampin in combination, rather than singly. As data accumulate, clindamycin may become the preferred outpatient antibiotic therapy (compared with TMP-SMX) in regions with a relatively low incidence of clindamycin resistance 18. Recently, treatment guidelines have been published 19.
A randomized, double-blind study by Thwaites et al 20 that included 758 patients with Staphylococcus aureus bacteremia assigned adjunctive rifampicin or a placebo, reported no overall benefit in adjunctive rifampicin over standard antibiotic therapy.
Medical treatment
Impetigo, folliculitis, furuncle and carbuncle
Impetigo and other minor skin infections (ie, superficial or localized infections) may be treated with a topical agent such as mupirocin or retapamulin. However, most community-associated methicillin-resistant Staphylococcus aureus (CA-MRSA) strains are or readily become resistant to mupirocin. More extensive or serious skin disease and bullous impetigo are treated with oral antistaphylococcal agents, as noted above 21.
Scalded skin syndrome (Ritter disease)
As with any Staphylococcus aureus toxin–mediated disease, treatment should aim to eradicate the focus of infection and end toxin production. Administer large doses of intravenous antistaphylococcal agents, such as oxacillin (150 mg/kg/day), or a first-generation cephalosporin, such as cefazolin (100 mg/kg/day). In vitro, clindamycin has been shown to inhibit the synthesis of TSST-1 and is extremely effective in combination with one of the agents mentioned above. Children with denuded skin should be touched as little as possible. Topical antimicrobial agents have little use, because skin damage is self-limited once systemic antibiotics are administered.
Osteomyelitis
Empirically, initiating a semisynthetic penicillin (eg, oxacillin [150 mg/kg/day]) and clindamycin (30-40 mg/kg/day) is a good choice for most cases of community-acquired osteomyelitis. In patients with allergy to penicillin, a first-generation cephalosporin and clindamycin (30-40 mg/kg/day) are an excellent alternative. Use vancomycin or linezolid when the other drugs mentioned are absolutely not tolerated or when resistance or the clinical course dictates. The duration of therapy is a controversial topic in the literature, but the consensus among multiple authors is that the minimum effective treatment time is 4-6 weeks. A switch to oral therapy is acceptable if the child is able to take oral antibiotics, is afebrile, and if he or she has demonstrated a good clinical response to parenteral antibiotics 22.
Septic arthritis
As in osteomyelitis, initiate an appropriate antistaphylococcal drug (eg, oxacillin, which is penicillinase resistant; clindamycin; cefazolin) parenterally. These antibiotics readily reach joint fluid, and the concentration in the joint fluid is 30% of the serum value. Therapy usually continues for at least 4 weeks. Duration of parenteral therapy is often debated. Some authors have demonstrated efficacy with 1 week of parenteral therapy followed with 3 weeks of oral therapy. Consider a switch to oral therapy based on the considerations mentioned above. Joint fluid that reaccumulates should be removed, and a sample should be cultured to assess the efficacy of therapy and to make the patient more comfortable.
Endocarditis
Duration of therapy for endocarditis, which is a life-threatening infection, is at least 4 weeks 23.
The combination of a beta-lactam and an aminoglycoside (usually gentamicin) is advocated, because it increases bacterial killing in vitro and in animal models of endocarditis. In patients with methicillin-resistant Staphylococcus aureus (MRSA), combinations of vancomycin with aminoglycosides should be used. In all cases the aminoglycoside is only added for the first 3 days.
Rifampin, because of its lipid solubility, is another potent agent when used in combination with nafcillin and gentamicin or vancomycin and gentamicin, especially in patients with prosthetic valve endocarditis. Rifampin should never be used alone because resistance can develop.
The response to therapy is usually slow, and patients may continue to have bacteremia, fever, and leukocytosis for at least a week after therapy is initiated.
Some authors recommend obtaining blood cultures after the end of therapy.
Surgical treatment
Skin and soft tissue infections
Drainage of any collections of pus is of paramount importance 24. For small abscesses in afebrile toddlers and children, drainage alone may suffice, since treatment with efficacious and nonefficacious systemic antibacterial therapy was equivalent if adequate drainage had occurred 25. Placement of a subcutaneous drain, rather than formal incision and drainage, has proven successful 26.
Osteomyelitis
Surgery is usually indicated to drain purulent material from the subperiosteal space or if infected foreign material is present.
Septic arthritis
In an infant, septic arthritis of the hip and shoulder is a surgical emergency; these joints should be drained as soon as possible to prevent bony destruction. In addition, if a large amount of fibrin, tissue debris, or loculation is present, preventing adequate drainage with needle aspiration, the joint should be surgically drained.
Endocarditis
If endocarditis occurs in the presence of an intracardiac foreign body, it may require removal.
Toxic shock syndrome
All potential foci of infection should be explored and surgically drained.
Antibiotics, in contrast, play a secondary role in the management of toxic shock syndrome. Because it can block the production of exotoxins by the bacterial ribosome, clindamycin or linezolid is often added to standard antibiotic therapy 27. Intravenous immunoglobulin may also be effective, although clinical evidence of benefit is not well established, and there is less evidence supporting the use of intravenous immunoglobulin to treat staphylococcal toxic shock syndrome than for streptococcal toxic shock syndrome. Nonetheless, it is recommended that intravenous immunoglobulin be considered for patients who have had no clinical response to aggressive supportive therapy within 6 hour 28.
Thrombophlebitis
Remove the infected intravenous line in patients who are immunocompromised or severely ill or when infection is impossible to eradicate medically.
- Taylor TA, Unakal CG. Staphylococcus Aureus. [Updated 2019 Mar 27]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2019 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK441868[↩]
- Wertheim HF, Melles DC, Vos MC, van Leeuwen W, van Belkum A, Verbrugh HA, Nouwen JL. 2005. The role of nasal carriage in Staphylococcus aureus infections. Lancet Infect Dis 5:751–762. doi:10.1016/S1473-3099(05)70295-4.[↩]
- Lowy FD. Staphylococcus aureus infections. N. Engl. J. Med. 1998 Aug 20;339(8):520-32.[↩]
- Rasigade JP, Vandenesch F. Staphylococcus aureus: a pathogen with still unresolved issues. Infect. Genet. Evol. 2014 Jan;21:510-4.[↩]
- Carrillo-Marquez MA, Hulten KG, Hammerman W, Mason EO, Kaplan SL. USA300 is the predominant genotype causing Staphylococcus aureus septic arthritis in children. Pediatr Infect Dis J. 2009 Dec. 28(12):1076-80.[↩]
- Carrillo-Marquez MA, Hulten KG, Mason EO, Kaplan SL. Clinical and Molecular Epidemiology of Staphylococcus aureus Catheter-Related Bacteremia in Children. Pediatr Infect Dis J. 2010 May. 29(5):410-4.[↩]
- Jacobson JA, Kasworm E, Daly JA. Risk of developing toxic shock syndrome associated with toxic shock syndrome toxin 1 following nongenital staphylococcal infection. Rev Infect Dis. 1989 Jan-Feb. 11 Suppl 1:S8-13.[↩]
- John CC, Niermann M, Sharon B, Peterson ML, Kranz DM, Schlievert PM. Staphylococcal toxic shock syndrome erythroderma is associated with superantigenicity and hypersensitivity. Clin Infect Dis. 2009 Dec 15. 49(12):1893-6.[↩]
- Schweizer M, et al. A multicenter intervention to reduce surgical site infections among patients undergoing cardiac operations and total joint arthroplasty (STOP SSI STUDY). Paper presented at IDWeek; October 8-12, 2014; Philadelphia, PA.[↩]
- Staph aureus Scheme Cuts Surgical Infection. https://www.medscape.com/viewarticle/833457[↩]
- Smit J, López-Cortés LE, Thomsen RW, Schønheyder HC, Nielsen H, Frøslev T, et al. Statin Use and Risk of Community-Acquired Staphylococcus aureus Bacteremia: A Population-Based Case-Control Study. Mayo Clin Proc. 2017 Oct. 92 (10):1469-1478.[↩]
- Staphylococcus Aureus Infection Clinical Presentation. https://emedicine.medscape.com/article/971358-clinical[↩]
- Lane JW, Tang J, Taggard D, Byun R. Successful use of daptomycin and linezolid, without surgical intervention, in the treatment of extensive epidural abscess and bacteremia due to methicillin-resistant Staphylococcus aureus (MRSA). Infect Dis Clin Pract. Sept 2011. 19(5):362-364.[↩]
- Abdel-Haq N, Quezada M, Asmar BI. Retropharyngeal Abscess in Children: The Rising Incidence of Methicillin-Resistant Staphylococcus aureus. Pediatr Infect Dis J. 2012 Jul. 31(7):696-9.[↩]
- McNeil JC, Hulten KG, Kaplan SL, Mahoney DH, Mason EO. Staphylococcus aureus Infections in Pediatric Oncology Patients: High Rates of Antimicrobial Resistance, Antiseptic Tolerance and Complications. Pediatr Infect Dis J. 2012 Sep 11.[↩]
- Elliott DJ, Zaoutis TE, Troxel AB, Loh A, Keren R. Empiric Antimicrobial Therapy for Pediatric Skin and Soft-Tissue Infections in the Era of Methicillin-Resistant Staphylococcus aureus. Pediatrics. 2009 Jun. 123(6):e959-66.[↩][↩]
- Lee S, Choe PG, Song KH, Park SW, Kim HB, Kim NJ, et al. Is cefazolin inferior to nafcillin for treatment of methicillin-susceptible Staphylococcus aureus bacteremia?. Antimicrob Agents Chemother. 2011 Nov. 55(11):5122-6.[↩]
- Williams DJ, Cooper WO, Kaltenbach LA, Dudley JA, Kirschke DL, Jones TF, et al. Comparative Effectiveness of Antibiotic Treatment Strategies for Pediatric Skin and Soft-Tissue Infections. Pediatrics. 2011 Aug 15.[↩]
- [Guideline] Liu C, Bayer A, Cosgrove SE, et al. Clinical Practice Guidelines by the Infectious Diseases Society of America for the Treatment of Methicillin-Resistant Staphylococcus Aureus Infections in Adults and Children. Clin Infect Dis. 2011 Feb 1. 52(3):e18-e55.[↩]
- Mejer N, Westh H, Schønheyder HC, Jensen AG, Larsen AR, Skov R, et al. Stable incidence and continued improvement in short term mortality of Staphylococcus aureus bacteraemia between 1995 and 2008. BMC Infect Dis. 2012 Oct 17. 12:260.[↩]
- Duong M, Markwell S, Peter J, Barenkamp S. Randomized, controlled trial of antibiotics in the management of community-acquired skin abscesses in the pediatric patient. Ann Emerg Med. 2010 May. 55(5):401-7.[↩]
- Pääkkönen M, Kallio PE, Kallio MJ, Peltola H. Management of Osteoarticular Infections Caused by Staphylococcus aureus Is Similar to That of Other Etiologies: Analysis of 199 Staphylococcal Bone and Joint Infections. Pediatr Infect Dis J. 2012 May. 31(5):436-8.[↩]
- Karchmer AW. Staphylococcus aureus bacteremia and native valve endocarditis: the role of antimicrobial therapy. Infect Dis Clin Pract. March 2012. 20(2):100-108.[↩]
- Sreeramoju P, Porbandarwalla NS, Arango J, Latham K, Dent DL, Stewart RM, et al. Recurrent skin and soft tissue infections due to methicillin-resistant Staphylococcus aureus requiring operative debridement. Am J Surg. 2011 Feb. 201(2):216-20.[↩]
- Chen AE, Carroll KC, Diener-West M, Ross T, Ordun J, Goldstein MA, et al. Randomized controlled trial of cephalexin versus clindamycin for uncomplicated pediatric skin infections. Pediatrics. 2011 Mar. 127(3):e573-80.[↩]
- McNamara WF, Hartin CW Jr, Escobar MA, Yamout SZ, Lau ST, Lee YH. An alternative to open incision and drainage for community-acquired soft tissue abscesses in children. J Pediatr Surg. 2011 Mar. 46(3):502-6.[↩]
- Successful treatment of staphylococcal toxic shock syndrome with linezolid: a case report and in vitro evaluation of the production of toxic shock syndrome toxin type 1 in the presence of antibiotics. Stevens DL, Wallace RJ, Hamilton SM, Bryant AE. Clin Infect Dis. 2006 Mar 1; 42(5):729-30.[↩]
- Gram-positive toxic shock syndromes. Lappin E, Ferguson AJ. Lancet Infect Dis. 2009 May; 9(5):281-90.[↩]